Introduction
Diabetic retinopathy (DR) is one of the most
prevalent complications of diabetes mellitus, taking a marked toll
on the quality of life of patients, with a global prevalence of
22.27% (95% CI, 19.73-25.03) among patients with diabetes; the
global number of DR cases is estimated to increase from 103.12
million in 2020 to 160.50 million by 2045(1). Sustained hyperglycemia subjects
retinal pigment epithelial (RPE) cells to prolonged high glucose
(HG) exposure. As key cells in the outer retina, RPE cells serve a
central role in physiological processes, such as maintaining
retinal structural integrity, visual cycle and immune regulation
(2,3). HG can induce metabolic disorders in
RPE cells, increase oxidative stress, activate inflammatory
responses and lead to pyroptosis, ultimately damaging the normal
structure and function of the retina (4-6).
Therefore, inhibiting RPE cell dysfunction or death under HG
conditions may help alleviate DR progression.
Autophagy, as a cellular self-protection mechanism,
maintains internal homeostasis by degrading and recycling damaged
components (7). Autophagy
activation can eliminate HG-induced toxic substances to preserve
cellular stability, making it a key therapeutic target for DR
(8,9). The balance between autophagy and
pyroptosis is important in the pathological context of
DR-associated injury. Cell death represents a tightly regulated,
evolutionarily conserved process, key in maintaining tissue
homeostasis and driving disease progression, with its diverse
execution mechanisms ranging from apoptosis and necrosis to
necroptosis and autophagic cell death (10). Pyroptosis, an inflammatory subtype
of programmed necrosis, participates in DR through the
overexpression of pyroptosis-related molecules such as gasdermin D
(GSDMD) and gasdermin E (GSDME). This overexpression drives
cellular perforation and the secretion of pro-inflammatory
cytokines, ultimately culminating in inflammatory cell death
(1,11,12).
Accumulating evidence has suggested the existence of an intimate
regulatory association between autophagy and pyroptosis, with
bidirectional effects mediated by key molecules and pathways.
Autophagy suppresses pyroptosis via lysosomal degradation of the
NLRP3 inflammasome, thereby inhibiting caspase-1/GSDMD activation
and IL-1β/IL-18 release; by contrast, impaired autophagy promotes
pyroptotic cell death. This interaction is further modulated by the
mTOR/ULK1 axis and STAT3-dependent signaling, highlighting the
complexity of their regulatory network (13-15).
Therefore, balancing the activities of autophagy and pyroptosis is
central to the pathological progression of DR.
Maresin 1 (MaR1) is a specific pro-resolving lipid
mediator derived from ω-3 fatty acid metabolism, possessing
anti-inflammatory, antioxidant and tissue repair properties
(16-18).
Previous studies have demonstrated that MaR1 promotes autophagic
flux in LPS-stimulated human periodontal ligament cells (19). Additionally, MaR1 has been shown to
inhibit the activation of the NLRP3 inflammasome and subsequent
pyroptosis in rat models of neuropathic pain and mouse models of
liver ischemia-reperfusion injury (19-21).
It has been reported that MaR1 blocks HG-induced ferroptosis in
ARPE-19 cells, with this inhibition mediated through activation of
the nuclear factor erythroid 2-related factor 2/heme
oxygenase-1/glutathione peroxidase 4 pathway (22), findings that point to its potential
utility in DR prevention and treatment. However, the precise
mechanisms governing the regulation of autophagy and pyroptosis by
MaR1 in RPE cells remain poorly understood.
The present study aimed to explore the effect of
MaR1 on autophagy and pyroptosis in HG-stimulated ARPE-19 cells,
along with the mechanisms driving these effects, in order to lay a
theoretical foundation for the development and application of MaR1
in DR therapy.
Materials and methods
Cell culture and treatment
ARPE-19 (cat. no. iCell-h020; iCell Bioscience,
Inc.), a human RPE cell line, was maintained in DMEM/F12 medium
(Thermo Fisher Scientific, Inc.) supplemented with 10% FBS
(Invitrogen; Thermo Fisher Scientific, Inc.) and 1%
penicillin/streptomycin. Cultures were incubated at 37˚C in a
humidified atmosphere containing 5% CO2. To mimic DR
in vitro, ARPE-19 cells were subjected to 25 mmol/l HG for
48 h at 37˚C, with cells treated with 5.5 mmol/l normal glucose
(NG) serving as the control group.
Following the guidance of previous literature,
ARPE-19 cells were pretreated with MaR1 (cat. no. HY-116429;
MedChemExpress), dissolved in dimethyl sulfoxide (DMSO), at
concentrations of 1, 10 and 100 nmol/l for 30 min at 37˚C before HG
exposure for 24 h (22-24).
To investigate the role of autophagy and sirtuin 1
(SIRT1)/peroxisome proliferator-activated receptor-γ (PPAR-γ)
signaling, ARPE-19 cells were treated with 10 µM of the SIRT1
inhibitor selisistat (EX527;
C13H13ClN2O; cat. no. HY-15452;
MedChemExpress) or 2.5 µM of the autophagy inhibitor
3-methyladenine (3-MA; C6H7N5;
cat. no. HY-19312; MedChemExpress), both dissolved in DMSO, for 2 h
at 37˚C before HG exposure. The dose and duration of EX527 and 3-MA
administration were determined in relation to previous studies
(25,26).
MTT assay
An MTT cell proliferation and cytotoxicity assay kit
(cat. no. C0009S; Beyotime Biotechnology) was used to assess
ARPE-19 cell viability. Briefly, 1x104 ARPE-19 cells
were seeded into each well of a 96-well plate and treated as
aforementioned. Following 48 h of treatment, the cells were
incubated with 10 µl MTT solution (5 mg/ml) for 4 h at 37˚C.
Subsequently, 150 µl DMSO was added to solubilize the formazan
crystals and absorbance was measured at a wavelength of 570 nm.
ELISA
The culture medium was collected and centrifuged at
1,000 x g for 20 min at 4˚C to remove cell debris. Following this,
the supernatant from ARPE-19 cells was collected. TNF-α (cat. no.
97072ES), IL-1β (cat. no. 97028ES) and IL-6 (cat. no. 97068ES)
levels were quantified using corresponding ELISA kits from Shanghai
Yeasen Biotechnology Co., Ltd., according to the manufacturer's
protocols. All ELISA results were normalized by first subtracting
the absorbance values of blank control wells (containing only assay
diluent without cell supernatant) to eliminate non-specific
background signals. The concentrations of cytokines in the cell
supernatants were determined using ELISA kits according to the
manufacturer's instructions. The results are expressed as pg/ml. To
ensure consistency, the total protein concentration of each sample
was also monitored using a BCA protein assay kit. All measurements
were performed in triplicate wells and the coefficient of variation
for replicate measurements was <15%.
Monodansylcadaverine (MDC)
staining
MDC staining (cat. no. C3018S; Beyotime
Biotechnology) was used to assess autophagy levels in ARPE-19
cells. ARPE-19 cells were seeded at a density of 2x105
cells/well in 6-well culture plates. Once ARPE-19 cells reached
70-80% confluency, they were washed with PBS and incubated with 1
ml freshly prepared MDC solution at 37˚C for 30 min. Following
incubation, the MDC solution was aspirated and the cells were
washed with PBS to eliminate any unbound dye. Finally, the cells
were visualized using the CKX53 fluorescence microscope (Evident
Corporation).
Immunofluorescence staining
ARPE-19 cells were seeded at a density of
2x104 cells/well in 24-well culture plates. After
reaching the appropriate confluency, the cells were fixed in 4%
paraformaldehyde (cat. no. P0099; Beyotime Biotechnology) for 20
min at room temperature, and then permeabilized with 0.1% Triton
X-100 (cat. no. P0096; Beyotime Biotechnology) at room temperature
for 15 min. Subsequently, the cells were blocked with 1% BSA
solution (cat. no. ST023; Beyotime Biotechnology) at room
temperature for 1 h, followed by overnight incubation at 4˚C with
diluted anti-GSDMD N-terminal (GSDMD-N) primary antibody (1:100;
cat. no. ER1901-37; HUABIO). The next day, the cells were further
incubated with a FITC conjugated goat anti-rabbit IgG secondary
antibody (1:1,000; cat. no. GB22303; Wuhan Servicebio Technology
Co., Ltd.) at room temperature for 2 h in the dark. Finally, the
cells were stained with a DAPI solution (1 µg/ml; cat. no. C1002;
Beyotime Biotechnology) for 10 min at room temperature in the dark
before images were captured using a CKX53 fluorescence
microscope.
Western blotting
Protein extracts were prepared from ARPE-19 cells
using RIPA buffer (cat. no. P0013B; Beyotime Biotechnology), and
protein concentrations were further quantified using the same BCA
protein assay kit as described in the ELISA section. A total of 30
µg protein per sample was used in SDS-PAGE for separation, before
the separated proteins were transferred to PVDF membranes. After
blocking with 5% non-fat milk at room temperature for 1 h, the
membranes were probed with primary antibodies overnight at 4˚C.
Following primary antibody incubation, the membranes were washed
incubated with HRP-conjugated goat anti-rabbit IgG (1:5,000; cat.
no. GB23303; Wuhan Servicebio Technology Co., Ltd.) or
HRP-conjugated goat anti-mouse IgG (1:5,000; cat. no. GB23301;
Wuhan Servicebio Technology Co., Ltd.) at room temperature for 2 h,
depending on the host species of the primary antibodies. Blots were
developed in the dark using an enhanced chemiluminescence (ECL)
reagent (cat. no. WBKLS0100; MilliporeSigma), and protein bands
were visualized using a gel documentation system (ChemiDoc MP;
Bio-Rad Laboratories, Inc.). The densitometric analysis of the
protein bands was performed using ImageJ software, version 1.53t
(National Institutes of Health). The primary antibodies used in the
present study included the following rabbit antibodies: Anti-beclin
1 (1:1,000; cat. no. AF5128; Affinity Biosciences, Ltd.), anti-p62
(1:1,000; cat. no. AF5384; Affinity Biosciences, Ltd.), anti-LC3
(1:1,000; cat. no. 14600-1-AP; Proteintech Group, Inc.),
anti-caspase-1 (1:1,000; cat. no. 22915-1-AP; Proteintech Group,
Inc.), anti-cleaved-caspase-1 (1:1,000; cat. no. AF4005; Affinity
Biosciences, Ltd.), anti-GSDMD (1:1,000; cat. no. 20770-1-AP;
Proteintech Group, Inc.), anti-GSDMD-N (1:1,000; cat. no.
ER1901-37; HUABIO), anti-NLR family pyrin domain containing 3
(NLRP3; 1:1,000; cat. no. DF7438; Affinity Biosciences, Ltd.),
anti-apoptosis-associated speck-like protein containing a CARD
(ASC; 1:1,000; cat. no. DF6304; Affinity Biosciences, Ltd.),
anti-IL-18 (1:1,000; cat. no. DF6252; Affinity Biosciences, Ltd.)
and anti-β-actin (1:5,000; cat. no. AF7018; Affinity Biosciences,
Ltd.). Mouse antibodies included: Anti-SIRT1 (1:2,000; cat. no.
60303-1-Ig; Proteintech Group, Inc.) and anti-PPAR-γ (1:2,000; cat.
no. 66936-1-Ig; Proteintech Group, Inc.).
Statistical analysis
SPSS (version 24.0; IBM Corp.) was used for
statistical analysis. Data were obtained from at least three
independent experiments and are presented as the mean ± SD.
Comparisons among multiple groups were analyzed using one-way ANOVA
followed by Tukey's post hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
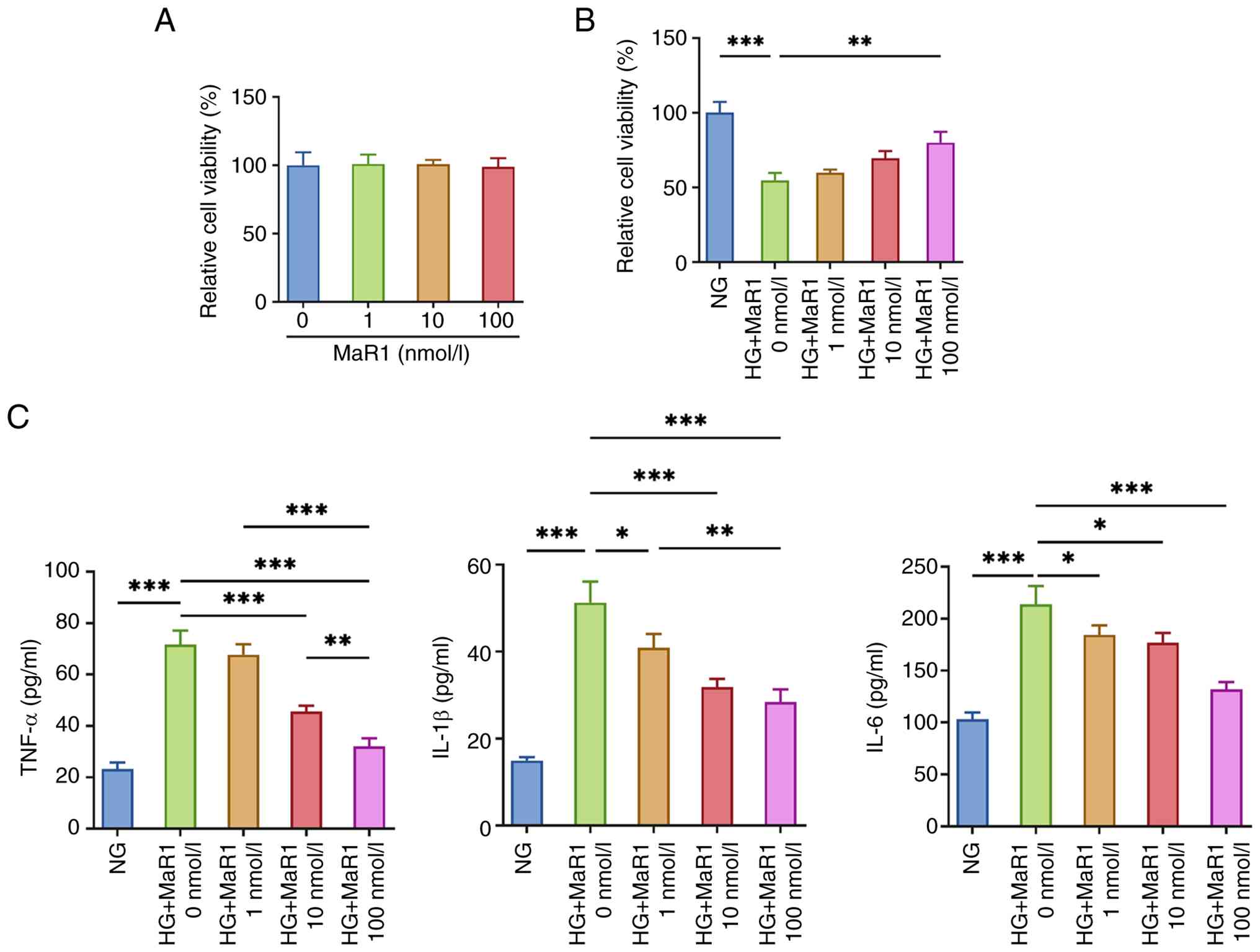
MaR1 ameliorates HG-induced ARPE-19
cell damage
As shown in Fig.
1A, MaR1 treatment at 1, 10 and 100 nmol/l did not
significantly affect the viability of ARPE-19 cells under control
conditions. However, under HG conditions, cell viability dropped
significantly. By contrast, MaR1 reversed this viability reduction
in a dose-dependent manner (Fig.
1B). ELISA assays revealed higher TNF-α, IL-1β and IL-6 levels
in the supernatant of HG-exposed cells compared with those in the
NG group. MaR1 treatment significantly mitigated the secretion of
these inflammatory factors. Specifically, the 100 nmol/l dose
exhibited the most potent anti-inflammatory effect, significantly
suppressing cytokine levels compared with the lower doses (1 and 10
nmol/l), indicating a dose-dependent inhibition (Fig. 1C).
MaR1 ameliorates HG-induced pyroptosis
in ARPE-19 cells
Immunofluorescence staining revealed that the
expression of the pyroptosis marker GSDMD-N increased following HG
exposure, as assessed through fluorescence intensity (Fig. 2A). However, MaR1 treatment
significantly reduced this HG-induced fluorescence intensity.
Western blotting further demonstrated that caspase-1 and GSDMD
expression displayed no significant changes across different
treatment groups. By contrast, ARPE-19 cells in the HG group
exhibited markedly higher levels of GSDMD-N, cleaved caspase-1,
NLRP3, ASC and IL-18 compared with those in the NG group (Fig. 2B). Crucially, MaR1 treatment
reversed the upregulation of these pyroptosis-related proteins.
These findings suggested that HG stimulation induced pyroptosis in
ARPE-19 cells and that MaR1 dose-dependently inhibited the
HG-induced increase in the expression of pyroptosis-related
proteins.
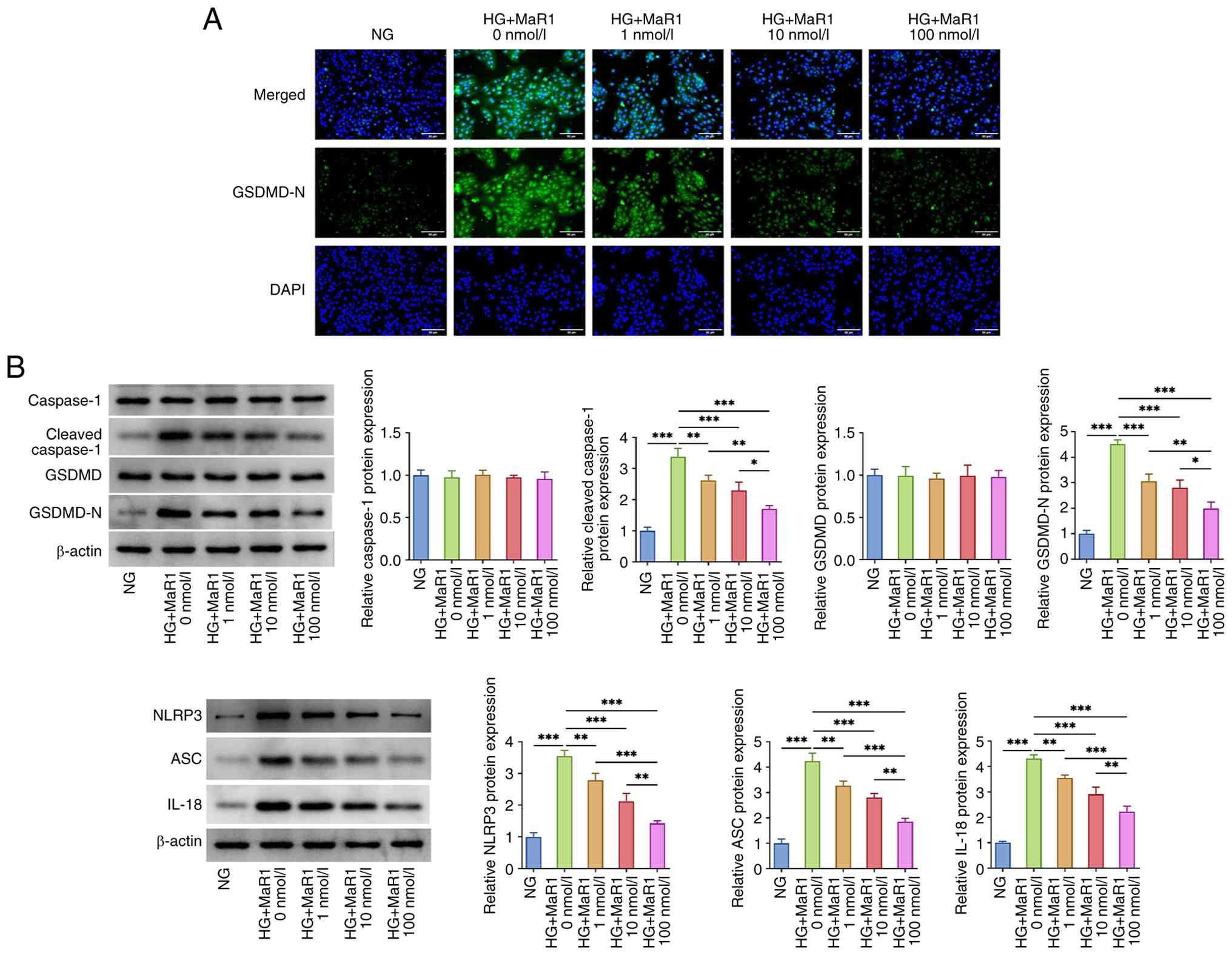 | Figure 2MaR1 ameliorates HG-induced
pyroptosis in ARPE-19 cells. (A) Effect of MaR1 treatment at
varying concentrations on the fluorescence intensity of GSDMD-N in
HG-induced ARPE-19 cells. Scale bar, 50 µm. (B) Effect of MaR1
treatment at varying concentrations on the expression of
pyroptosis-related proteins (caspase-1, GSDMD, GSDMD-N, cleaved
caspase-1, NLRP3, ASC and IL-18) in HG-induced ARPE-19
cells.*P<0.05, **P<0.01 and
***P<0.001. MaR1, maresin 1; HG, high glucose; NG,
normal glucose; GSDMD, gasdermin D; GSDMD-N, GSDMD N-terminal;
NLRP3, NLR family pyrin domain containing 3; ASC,
apoptosis-associated speck-like protein containing a CARD. |
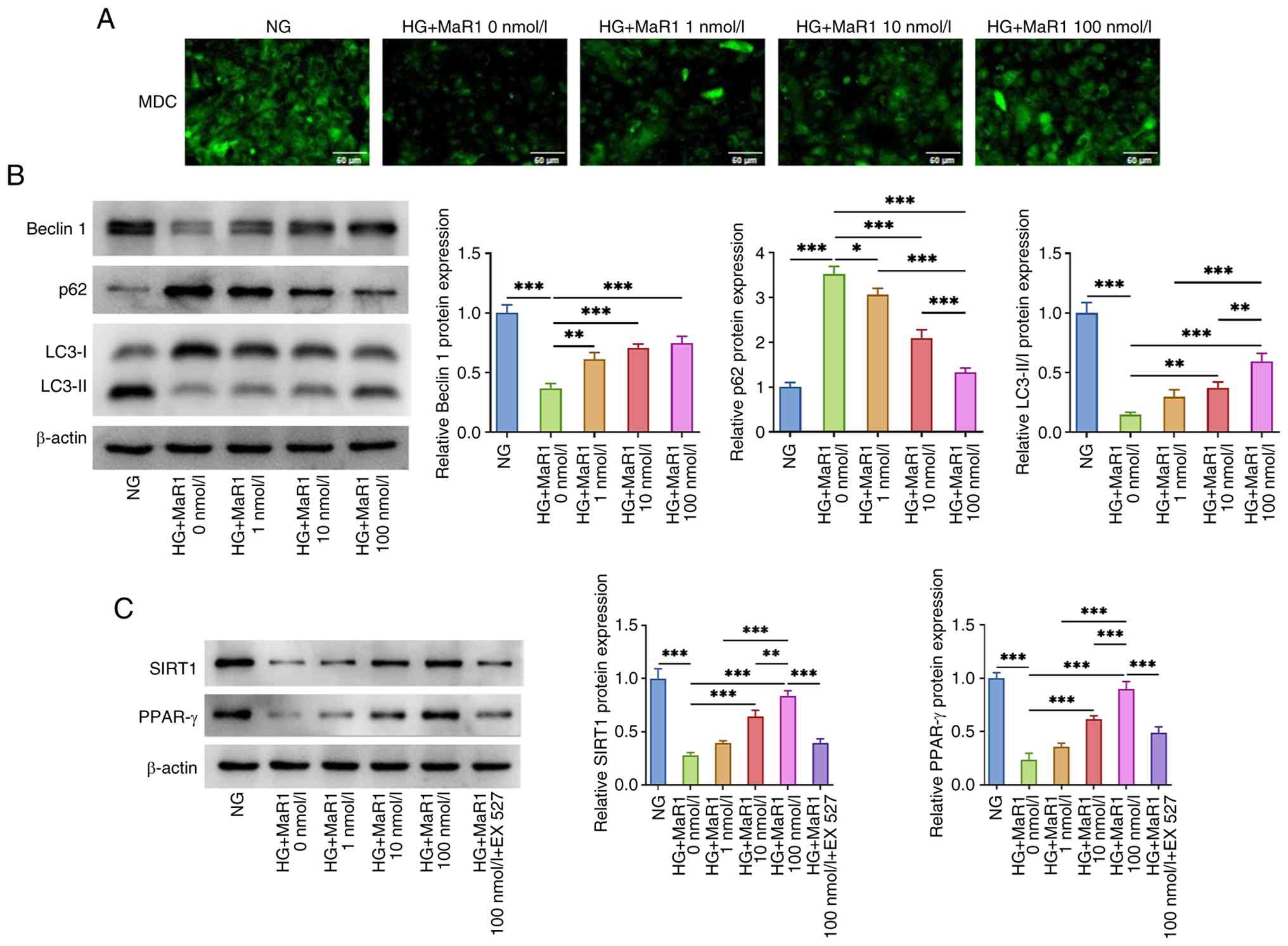
MaR1 improves HG-induced autophagy in
ARPE-19 cells and regulates SIRT1/PPAR-γ signaling
MDC staining revealed that ARPE-19 cells in the HG
group exhibited lower MDC fluorescence intensity compared with that
in the NG group, suggesting that HG conditions led to a reduction
in autophagic flux in ARPE-19 cells (Fig. 3A). Western blot results further
corroborated this finding, as evidenced by the decreased expression
of beclin 1, decreased LC3-II/I ratio and increased expression of
p62 in the HG-treated group compared with the corresponding levels
in the NG group (Fig. 3B).
However, MaR1 reinstated the autophagic flux in HG-stimulated
ARPE-19 cells in a concentration-dependent manner. Furthermore, it
was observed that HG exposure reduced the expression levels of
SIRT1 and PPAR-γ in ARPE-19 cells, whereas MaR1 reversed this
reduction. Notably, the expression of SIRT1 and PPAR-γ
significantly increased with higher doses of MaR1 (Fig. 3C). By contrast, the addition of the
SIRT1 inhibitor EX527 effectively abolished the MaR1-induced
upregulation of both SIRT1 and PPAR-γ, confirming the involvement
of the SIRT1 pathway (Fig.
3C).
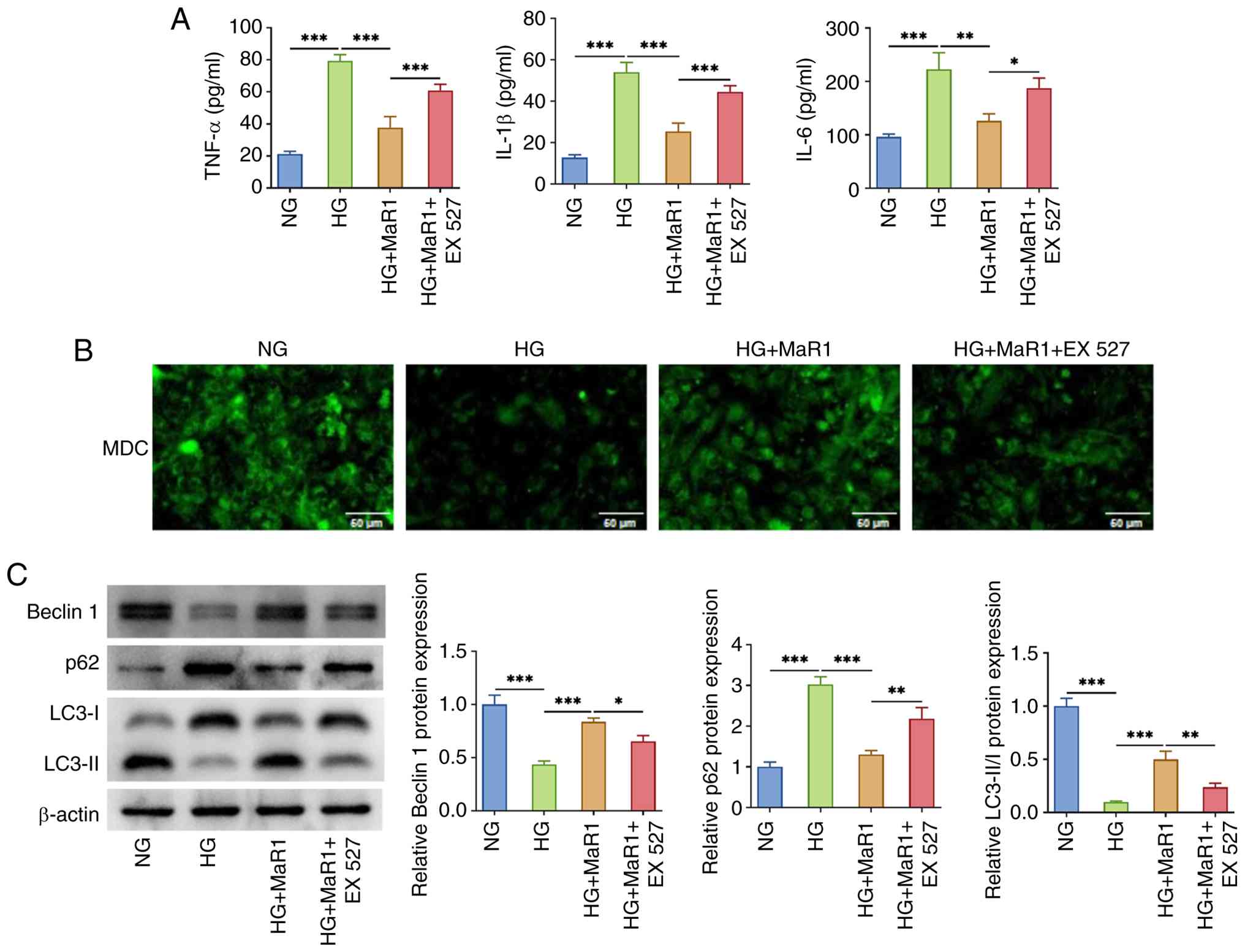
MaR1 ameliorates HG-induced
inflammatory response and autophagy in ARPE-19 cells through
SIRT1/PPAR-γ signaling
To explore the association between the SIRT1/PPAR-γ
signaling pathway and the action of MaR1, the SIRT1 inhibitor EX527
was used to pretreat ARPE-19 cells before exposure to HG. Based on
the preliminary findings that 100 nmol/l MaR1 exhibited the most
potent protective and anti-inflammatory effects (Fig. 1, Fig.
2 and Fig. 3), this
concentration was selected for all subsequent rescue experiments.
ELISA results indicated that although MaR1 effectively inhibited
the expression of inflammatory factors induced by HG in ARPE-19
cells, the administration of EX527 partially reversed this
inhibition (Fig. 4A). In addition,
EX527 reduced the ability of MaR1 to restore the autophagic flux
(Fig. 4B), upregulate beclin 1 and
LC3-II/I expression levels or reduce p62 levels (Fig. 4C) in HG-stimulated ARPE-19
cells.
MaR1 mediates autophagy through
SIRT1/PPAR-γ signaling to ameliorate HG-induced pyroptosis in
ARPE-19 cells
To further examine how SIRT1/PPAR-γ
signaling-mediated autophagy is associated with the effects of
MaR1, ARPE-19 cells were pretreated with the SIRT1 inhibitor EX527.
Additionally, to rigorously confirm the specific involvement of the
autophagy pathway in MaR1-mediated protection, the autophagy
inhibitor 3-MA was used before subjecting the cells to HG. The
findings showed that MaR1 markedly decreased GSDMD-N fluorescence
intensity in ARPE-19 cells (Fig.
5A) and reduced the expression levels of GSDMD-N, cleaved
caspase-1, NLRP3, ASC and IL-18 compared with those in the HG group
(Fig. 5B). However, both EX527 and
3-MA significantly reversed the protective effects of MaR1, leading
to the re-elevation of these pyroptosis-related markers. Notably,
the 3-MA treatment resulted in significantly higher protein
expression levels compared with the EX527 treatment, suggesting
that direct autophagy inhibition had a more pronounced effect on
blocking MaR1 activity than SIRT1 inhibition in this context.
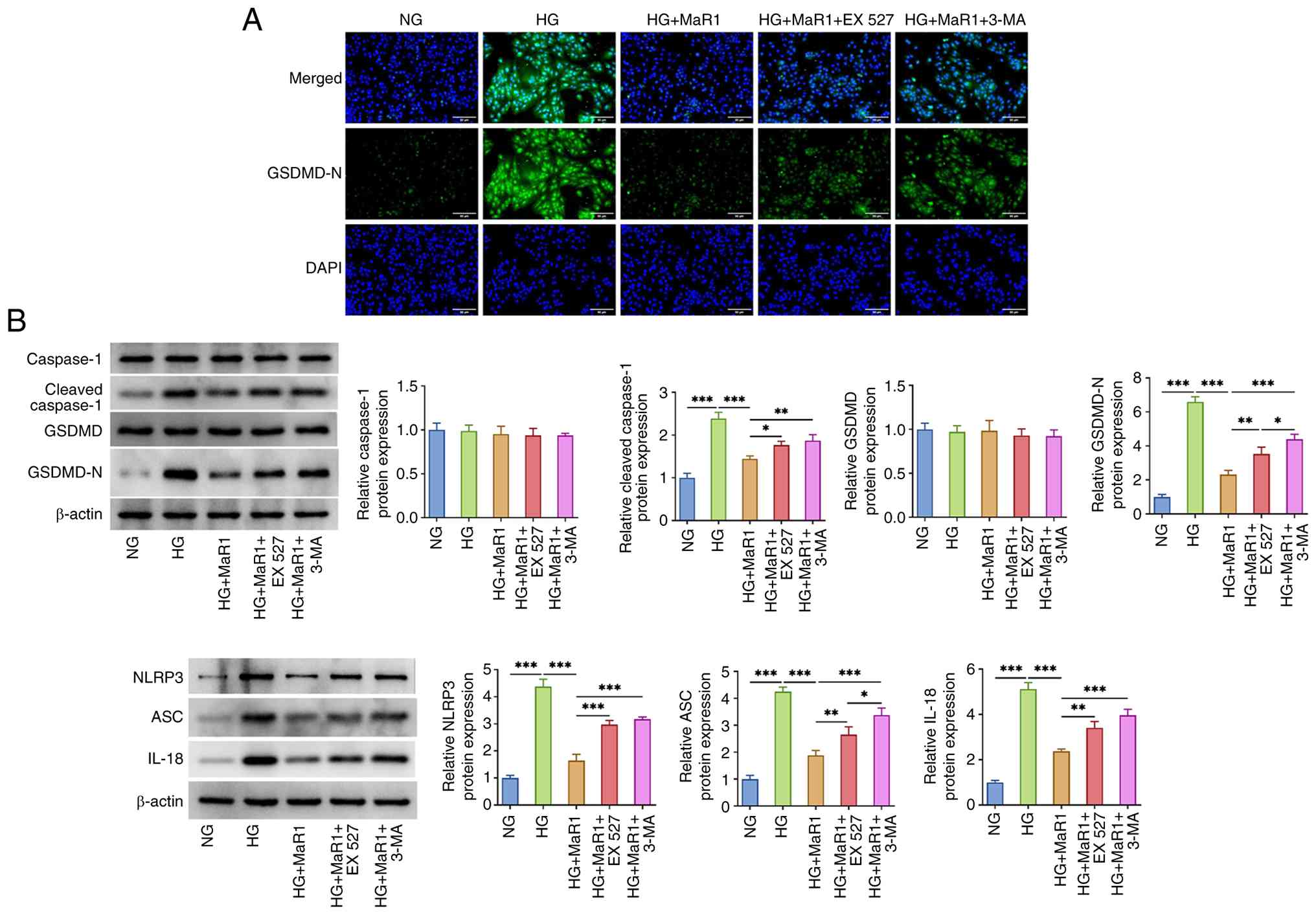 | Figure 5MaR1 mediates autophagy through
SIRT1/peroxisome proliferator-activated receptor-γ signaling to
ameliorate HG-induced pyroptosis in ARPE-19 cells. (A) SIRT1
inhibitor EX527 and autophagy inhibitor 3-MA enhanced the
fluorescence intensity of GSDMD-N in ARPE-19 cells treated with HG
and MaR1. Scale bar, 50 µm. (B) EX527 and 3-MA upregulated the
expression of GSDMD-N, cleaved caspase-1, NLRP3, ASC and IL-18 in
ARPE-19 cells treated with HG and MaR1. *P<0.05,
**P<0.01 and ***P<0.001. MaR1, maresin
1; HG, high glucose; NG, normal glucose; SIRT1, sirtuin 1; GSDMD,
gasdermin D; GSDMD-N, GSDMD N-terminal; NLRP3, NLR family pyrin
domain containing 3; ASC, apoptosis-associated speck-like protein
containing a CARD; 3-MA, 3-methyladenine. |
Discussion
In the pathological progression of DR, an
association exists between autophagy and pyroptosis, which is key
in regulating the fate of retinal cells and the development of DR.
As a notable pathogenic factor of DR, the HG environment induces
metabolic disorders in RPE cells, including mitochondrial
dysfunction, thereby triggering a series of cellular stress
responses that contribute to HG-induced retinal damage. HG impairs
mitochondrial respiratory chain function, particularly by
suppressing the activities of complexes I and III, which in turn
results in the overproduction of reactive oxygen species (ROS)
within mitochondria. These ROS further damage the mitochondrial
membrane structure and function, hindering the normal metabolism
and energy supply of mitochondria. If damaged mitochondria are not
promptly cleared, they will continuously generate ROS, forming a
vicious cycle that exacerbates intracellular oxidative stress
levels. Accumulating evidence indicates that oxidative and carbonyl
stress, fueled by overproduction of ROS and reactive carbonyl
species (such as methylglyoxal), act as a drivers of diabetic
complications, including retinopathy, neuropathy and nephropathy
(27,28). Therefore, inhibiting ROS production
and scavenging excess ROS have been suggested as potential
therapeutic strategies for DR (29).
Pyroptosis is an inflammatory type of programmed
necrosis that regulates a number of cellular pathophysiological
processes. During the pathological progression of DR, upstream
pyroptosis-related molecules, such as GSDMD and GSDME are
overexpressed, leading to cellular perforation, this process causes
ruptured cells to release potent pro-inflammatory cytokines,
specifically IL-1β, IL-18 and high mobility group box 1, thereby
triggering inflammatory cell death and amplifying the chronic
inflammatory response in the retina (11-13).
NLRP3 inflammasome activation is associated with pyroptosis
occurrence. In an HG environment, NLRP3 inflammasome, a
multiprotein complex primarily composed of NLRP3 and the adaptor
protein ASC and caspase-1, is activated by various signals. These
include ROS produced by damaged mitochondria, as well as other
cellular stress signals, such as potassium (K+) efflux,
and endoplasmic reticulum stress (30,31).
Once activated, the NLRP3 inflammasome processes procaspase-1 into
active caspase-1. Active caspase-1 in turn cleaves the GSDMD
protein to produce the GSDMD-N fragment. This fragment forms pores
on the cell membrane, leading to the release of cellular contents
and subsequent inflammatory responses and pyroptosis (32-34).
Research has shown that in HG-induced RPE cell models, NLRP3
inflammasome activation increases markedly, alongside a notable
rise in GSDMD-N levels, demonstrating that HG-induced pyroptosis
occurs in these cells (35).
Autophagy, as an important intracellular
self-protection mechanism, serves a key role in responding to
mitochondrial damage (36,37). When damaged mitochondria accumulate
in cells, autophagy is activated to encapsulate the damaged
mitochondria into autophagosomes, which subsequently fuse with
lysosomes, leading to the degradation of damaged mitochondria.
Furthermore, 14S-hydroperoxydocosahexaenoic acid (14S-HpDHA) is an
intermediate in the MaR synthesis pathway and previous studies have
shown that 14S-HpDHA reaches a peak during the resolution of
inflammation, suggesting that MaR is involved in inhibiting
inflammation (24,38). MaR1 is a chemical isomer of MaR and
growing evidence indicates that MaR1 can alleviate inflammatory
responses and inhibit ROS production in various models, including
LPS-stimulated human periodontal ligament cells, as well as rodent
models of neuropathic pain and liver ischemia-reperfusion injury
(21,39). The present study demonstrated that
MaR1 supports autophagic recovery by upregulating autophagy-related
genes and proteins. Beclin 1, a known initiator of autophagy,
showed increased expression after MaR1 treatment, thereby
indicating increased autophagosome formation. An elevated LC3-II/I
ratio suggested greater autophagosome formation and turnover,
implying that MaR1 enhanced autophagic flux by modulating LC3
conversion. Furthermore, reduced expression of p62, an autophagic
degradation substrate, served as an indirect marker of enhanced
autophagic activity (40). This
suggests that MaR1 promotes efficient p62 degradation, reinforcing
the role of MaR1in restoring autophagy in ARPE-19 cells.
Simultaneously, the results suggested that 3-MA, an autophagy
inhibitor, may abrogate the regulatory effect of MaR1 on
pyroptosis, further demonstrating the key role of autophagy in
MaR1-mediated attenuation of HG-induced cellular pyroptosis.
Autophagy and pyroptosis exhibit a negative
regulatory association, in which autophagy can suppress pyroptosis.
Mitophagy-mediated clearance of damaged mitochondria reduces ROS
production, which in turn lowers NLRP3 inflammasome activation and
suppresses pyroptosis. This provides an important protective
mechanism for maintaining the homeostasis and function of retinal
cells. Impaired autophagic function fails to efficiently clear
damaged mitochondria or prevent NLRP3 inflammasome activation,
triggering excessive pyroptosis, worsening retinal cell damage and
death, as well as further driving the progression of DR (34,41).
Therefore, further research on the pathological importance of the
autophagy-pyroptosis crosstalk holds important theoretical and
clinical value for understanding the pathogenesis of DR and
identifying effective therapeutic targets.
SIRT1 is a NAD+-dependent deacetylase
that performs a number of biological functions, such as regulating
cellular metabolism via PGC-1α, oxidative stress responses and
inflammatory processes through FOXO1 and Nrf2, and inflammatory
processes by inhibiting the NF-κB signaling pathway (42). Previous studies have shown that
activation of SIRT1 not only inhibits oxidative stress-induced
apoptosis but also alleviates vascular dysfunction, exerting a
protective effect against diabetes-induced retinal vascular leakage
(43). PPAR-γ is a nuclear
receptor that serves as a key regulator of lipid metabolism,
inflammation modulation and cellular differentiation. SIRT1 exerts
its functions through activating PPAR-γ (44). With regard to the regulatory
mechanism of SIRT1 on PPAR-γ, two possible pathways exis.t: i)
Direct deacetylation; and ii) indirect regulation through other
intermediate molecules. In terms of direct deacetylation, studies
have demonstrated that SIRT1 physically interacts with PPAR-γ and
specifically mediates its deacetylation at key lysine residues
(45,46). For instance, SIRT1 can deacetylate
and activate PGC-1α, which serves as a potent co-activator for
PPAR-γ, thereby enhancing its transcriptional activity (47). Additionally, SIRT1-mediated
deacetylation of the NF-κB p65 subunit inhibits its transcriptional
activity, which in turn alleviates the NF-κB-dependent
transcriptional repression of PPAR-γ. This restoration of PPAR-γ
signaling maintains mitochondrial homeostasis and antioxidant
defenses in ARPE-19 cells under stress, as evidenced by the
upregulation of downstream targets such as PGC-1α and SOD2
(48,49). The present study found that MaR1
pretreatment could restore the expression of SIRT1 and PPAR-γ in
HG-induced ARPE-19 cells. This suggests that MaR1-mediated
upregulation of the SIRT1/PPAR-γ signaling pathway may be a key
potential mechanism underlying its effects in restoring autophagy,
inhibiting pyroptosis and attenuating inflammatory responses. This
hypothesis was further supported by employing a SIRT1 inhibitor.
The results revealed that the SIRT1 inhibitor could simultaneously
attenuate the regulatory effects of MaR1 on inflammation,
pyroptosis and autophagy. This finding highlights the importance of
the SIRT1/PPAR-γ signaling pathway in MaR1-mediated regulation of
cellular pyroptosis and autophagy, contributing towards a more
comprehensive understanding of the mechanism of action of MaR1.
Therefore, future research should focus on elucidating the
molecular association between SIRT1 and PPAR-γ, while also further
exploring their synergy with regard to the regulation of autophagy
and suppression of pyroptosis by MaR1, thus providing a clearer
understanding of the regulatory mechanisms of this signaling
pathway in DR.
In the present study, it was demonstrated that MaR1
treatment effectively restored the viability of ARPE-19 cells
exposed to an HG environment. The results showed that under HG
conditions, the metabolic and physiological functions of ARPE-19
cells were markedly impaired, leading to a significant decreased in
cell viability. However, MaR1 treatment reversed these effects.
Specifically, the data suggested that MaR1 might enhance cell
viability by activating the SIRT1/PPAR-γ signaling pathways.
Additionally, it was observed that MaR1 significantly reduced the
levels of inflammatory factors in HG-exposed ARPE-19 cells, further
supporting its protective role.
The present study demonstrated that MaR1 restored
autophagy, potentially by upregulating the SIRT1/PPAR-γ signaling
pathway, thereby effectively attenuating HG-induced pyroptosis and
inflammatory responses in ARPE-19 cells (Fig. 6). However, a limitation of the
present study is that the experiments were conducted using ARPE-19
cells. In vitro cell models offer advantages, such as easy
control of experimental conditions and relatively simple operation,
facilitating the investigation of intracellular molecular
mechanisms and signaling pathways. However, notable differences
exist between in vitro cell models and the complex
physiological environment observed in vivo. Cells in
vivo are influenced by numerous factors, including cell-cell
interactions, the tissue microenvironment and systemic metabolic
status, which are difficult to fully simulate through in
vitro models.
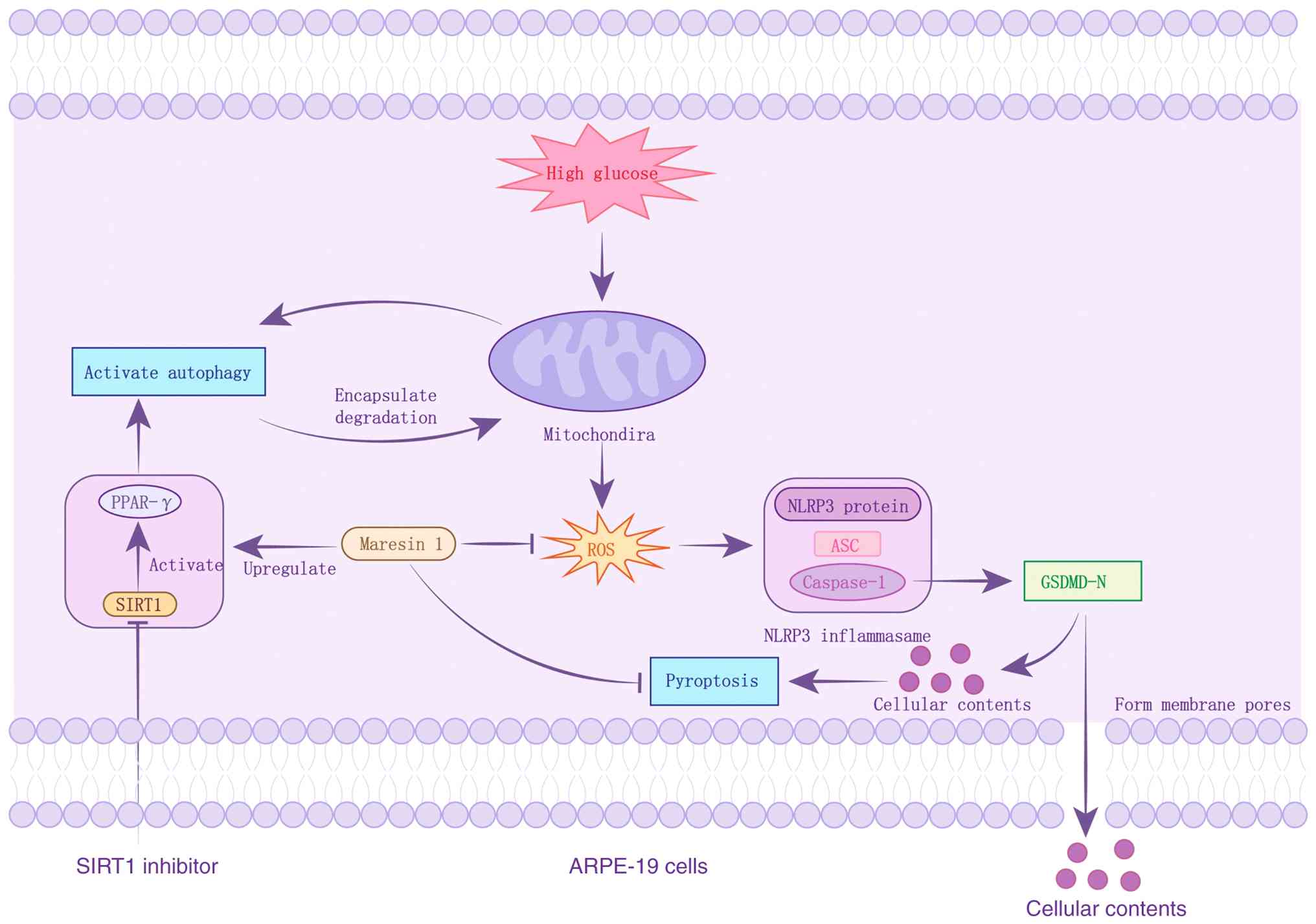 | Figure 6Schematic illustration of the
protective mechanism of MaR1 against HG-induced pyroptosis in
ARPE-19 cells. High glucose stimulation triggers the overproduction
of ROS, leading to the activation of the NLRP3 inflammasome complex
(comprising NLRP3, ASC and Caspase-1). This activation results in
the cleavage of GSDMD, which forms membrane pores (GSDMD-N) and
promotes the release of pro-inflammatory cytokines, ultimately
inducing pyroptosis. However, MaR1 treatment effectively
upregulates SIRT1 expression, which in turn enhances PPAR-γ levels
and restores autophagic flux. The restored autophagy facilitates
the clearance of damaged mitochondria and ROS, thereby inhibiting
the NLRP3/GSDMD signaling pathway and protecting ARPE-19 cells from
HG-induced inflammatory damage. SIRT1, sirtuin 1; PPAR-γ,
peroxisome proliferator-activated receptor-γ; ROS, reactive oxygen
species; GSDMD-N, gasdermin D N-terminal; NLRP3, NLR family pyrin
domain containing 3; ASC, apoptosis-associated speck-like protein
containing a CARD. |
To comprehensively verify the findings of the
present study, further research in diabetic animal models is
required. Currently, commonly used models include the
streptozotocin (STZ)-induced diabetic rat model and the db/db
diabetic mouse models, among others (50). Based on these models, diabetes can
be established by intraperitoneal injection of STZ or using the
gene knockout technology, followed by MaR1 intervention to observe
the therapeutic effect on DR. Detecting autophagy and
pyroptosis-related markers in retinal tissues, such as LC3 and
GSDMD-N protein expression levels, along with inflammatory factor
release, will allow for the assessment of whether the regulation of
autophagy and pyroptosis by MaR1 extends to in vivo
contexts. This will offer more robust theoretical evidence and
experimental support for the use of MaR1 in DR therapy, while also
advancing its translation from basic research to clinical
practice.
Acknowledgements
Not applicable.
Funding
Funding: The present study was funded by the Medical Science
Research Project of Hebei (grant no. 20240063) and the 2024
Government-Funded Training Program for Outstanding Clinical
Medicine Talents (grant no. ZF2024074).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
JZ collected and collated research data, drafted the
initial manuscript and revised it based on peer review feedback. DZ
designed the research methods, analyzed/interpreted key
experimental results and helped refine the discussion section to
enhance the academic depth of the manuscript. YZ conducted the
literature review (systematic study screening and background
summary), performed data analysis, and contributed to the
interpretation of the results, ensuring the reliability and
scientific rigor of the research foundation.. HaoS assisted with
experimental procedures and the analysis and interpretation of the
acquired data. ZY participated in the statistical analysis of
experimental data (through professional software for data
processing/validation) and manuscript proofreading (formatting and
language error correction). HaiS conceived and designed the
research framework, supervised the entire manuscript preparation
process, provided critical revisions on intellectual content,
approved the final publishable version and is accountable for all
work aspects, ensuring proper investigation/resolution of issues
related to research accuracy or integrity. JZ and HaiS confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Teo ZL, Tham YC, Yu M, Chee ML, Rim TH,
Cheung N, Bikbov MM, Wang YX, Tang Y, Lu Y, et al: Global
prevalence of diabetic retinopathy and projection of burden through
2045: systematic review and Meta-analysis. Ophthalmology.
128:1580–1591. 2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kim DH, Kim JH, Hwangbo H, Kim SY, Ji SY,
Kim MY, Cha HJ, Park C, Hong SH, Kim GY, et al: Spermidine
attenuates oxidative Stress-induced apoptosis via blocking Ca2+
overload in retinal pigment epithelial cells independently of ROS.
Int J Mol Sci. 22(1361)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Schäfer N, Wolf HN, Enzbrenner A, Schikora
J, Reichenthaler M, Enzmann V and Pauly D: Properdin modulates
complement component production in stressed human primary retinal
pigment epithelium cells. Antioxidants (Basel).
9(793)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Zhang Q, Li HS, Li R, Du JH and Jiao C:
Autophagy dysregulation mediates the damage of high glucose to
retinal pigment epithelium cells. Int J Ophthalmol. 14:805–811.
2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Liang GH, Luo YN, Wei RZ, Yin JY, Qin ZL,
Lu LL and Ma WH: CircZNF532 knockdown protects retinal pigment
epithelial cells against high glucose-induced apoptosis and
pyroptosis by regulating the miR-20b-5p/STAT3 axis. J Diabetes
Investig. 13:781–795. 2022.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Huang HW, Yang CM and Yang CH: Fibroblast
growth factor type 1 ameliorates High-Glucose-induced oxidative
stress and neuroinflammation in retinal pigment epithelial cells
and a Streptozotocin-induced diabetic rat model. Int J Mol Sci.
22(7233)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hu Z, Wang X, Hu Q and Chen X: Exploring
the protective effects of herbal monomers against diabetic
retinopathy based on the regulation of autophagy and apoptosis: A
review. Medicine (Baltimore). 102(e35541)2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Adornetto A, Gesualdo C, Laganà ML, Trotta
MC, Rossi S and Russo R: Autophagy: A novel pharmacological target
in diabetic retinopathy. Front Pharmacol. 12(695267)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Feng L, Liang L, Zhang S, Yang J, Yue Y
and Zhang X: HMGB1 downregulation in retinal pigment epithelial
cells protects against diabetic retinopathy through the
Autophagy-lysosome pathway. Autophagy. 18:320–339. 2022.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mustafa M, Ahmad R, Tantry IQ, Ahmad W,
Siddiqui S, Alam M, Abbas K, Moinuddin Hassan MI, Habib S and Islam
S: Apoptosis: A comprehensive overview of signaling pathways,
morphological changes, and physiological significance and
therapeutic implications. Cells. 13(1838)2024.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Zheng X, Wan J and Tan G: The mechanisms
of NLRP3 inflammasome/pyroptosis activation and their role in
diabetic retinopathy. Front Immunol. 14(1151185)2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Al Mamun A, Mimi AA, Zaeem M, Wu Y,
Monalisa I, Akter A, Munir F and Xiao J: Role of pyroptosis in
diabetic retinopathy and its therapeutic implications. Eur J
Pharmacol. 904(174166)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wu C, Chen H, Zhuang R, Zhang H, Wang Y,
Hu X, Xu Y, Li J, Li Y, Wang X, et al: Betulinic acid inhibits
pyroptosis in spinal cord injury by augmenting autophagy via the
AMPK-mTOR-TFEB signaling pathway. Int J Biol Sci. 17:1138–1152.
2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Cai D, Li C, Zhang Y, He S, Guo Y, Liao W,
Liao Y, Bin J and He X: CircHipk3 serves a dual role in macrophage
pyroptosis by promoting NLRP3 transcription and inhibition of
autophagy to induce abdominal aortic aneurysm formation. Clin
Transl Med. 14(e70102)2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zheng Q, Wang D, Lin R and Xu W:
Pyroptosis, ferroptosis, and autophagy in spinal cord injury:
Regulatory mechanisms and therapeutic targets. Neural Regen Res.
20:2787–2806. 2025.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yin Z, Zhang J, Zhao M, Peng S, Ye J, Liu
J, Xu Y, Xu S, Pan W, Wei C, et al: Maresin-1 ameliorates
hypertensive vascular remodeling through its receptor LGR6. MedComm
(2020). 5(e491)2024.PubMed/NCBI View
Article : Google Scholar
|
|
17
|
Saito-Sasaki N, Sawada Y and Nakamura M:
Maresin-1 and inflammatory disease. Int J Mol Sci.
23(1367)2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Rodríguez MJ, Sabaj M, Tolosa G, Herrera
Vielma F, Zúñiga MJ, González DR and Zúñiga-Hernández J: Maresin-1
prevents liver fibrosis by targeting Nrf2 and NF-κB, reducing
oxidative stress and inflammation. Cells. 10(3406)2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhao M, Xian W, Liu W, Chen D, Wang S and
Cao J: Maresin1 alleviates neuroinflammation by inhibiting
caspase-3/GSDME-mediated pyroptosis in mice cerebral
ischemia-reperfusion model. J Stroke Cerebrovasc Dis.
33(107789)2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Du L, Li Y and Liu W: Maresin 1 regulates
autophagy and inflammation in human periodontal ligament cells
through glycogen synthase kinase-3β/β-catenin pathway under
inflammatory conditions. Arch Oral Biol. 87:242–247.
2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yin P, Wang X, Wang S, Wei Y, Feng J and
Zhu M: Maresin 1 improves cognitive decline and ameliorates
inflammation in a mouse model of Alzheimer's disease. Front Cell
Neurosci. 13(466)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Li Y, Liu J, Ma X and Bai X: Maresin-1
inhibits high glucose induced ferroptosis in ARPE-19 cells by
activating the Nrf2/HO-1/GPX4 pathway. BMC Ophthalmol.
23(368)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Wang Y, Li R, Chen L, Tan W, Sun Z, Xia H,
Li B, Yu Y, Gong J, Tang M, et al: Maresin 1 inhibits
Epithelial-to-Mesenchymal transition in vitro and attenuates
bleomycin induced lung fibrosis in vivo. Shock. 44:496–502.
2015.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhang P, Yin Y, Wang T, Li W, Li C, Zeng
X, Yang W, Zhang R, Tang Y, Shi L, et al: Maresin 1 mitigates
concanavalin A-induced acute liver injury in mice by inhibiting
ROS-mediated activation of NF-κB signaling. Free Radic Biol Med.
147:23–36. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wang G and Lin N: NAD-Dependent protein
deacetylase Sirtuin-1 mediated mitophagy regulates early brain
injury after subarachnoid hemorrhage. J Inflamm Res. 17:1971–1981.
2024.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Sheng Y, Sun B, Guo WT, Zhang YH, Liu X,
Xing Y and Dong DL: 3-Methyladenine induces cell death and its
interaction with chemotherapeutic drugs is independent of
autophagy. Biochem Biophys Res Commun. 432:5–9. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Baynes JW and Thorpe SR: Role of oxidative
stress in diabetic complications: A new perspective on an old
paradigm. Diabetes. 48:1–9. 1999.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Islam S, Mir AR, Raghav A, Khan F, Alam K,
Ali A and Uddin M: Neo-Epitopes generated on hydroxyl radical
modified GlycatedIgG have role in immunopathology of diabetes type
2. PLoS One. 12(e0169099)2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kang Q and Yang C: Oxidative stress and
diabetic retinopathy: Molecular mechanisms, pathogenetic role and
therapeutic implications. Redox Biol. 37(101799)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Muñoz-Planillo R, Kuffa P, Martínez-Colón
G, Smith BL, Rajendiran TM and Núñez G: K+ efflux is the
common trigger of NLRP3 inflammasome activation by bacterial toxins
and particulate matter. Immunity. 38:1142–1153. 2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Bronner DN, Abuaita BH, Chen X, Fitzgerald
KA, Nuñez G, He Y, Yin XM and O'Riordan MX: Endoplasmic reticulum
stress activates the inflammasome via NLRP3- and Caspase-2-driven
mitochondrial damage. Immunity. 43:451–462. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Huang C, Qi P, Cui H, Lu Q and Gao X:
CircFAT1 regulates retinal pigment epithelial cell pyroptosis and
autophagy via mediating m6A reader protein YTHDF2 expression in
diabetic retinopathy. Exp Eye Res. 222(109152)2022.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wang JJ, Chen ZL, Wang DD, Wu KF, Huang WB
and Zhang LQ: linc00174 deteriorates the pathogenesis of diabetic
retinopathy via miR-26a-5p/PTEN/Akt signalling cascade-mediated
pyroptosis. Biochem Biophys Res Commun. 630:92–100. 2022.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wang N, Wei L, Liu D, Zhang Q, Xia X, Ding
L and Xiong S: Identification and validation of Autophagy-related
genes in diabetic retinopathy. Front Endocrinol (Lausanne).
13(867600)2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wang P, Chen F, Wang W and Zhang XD:
Hydrogen sulfide attenuates high Glucose-induced human retinal
pigment epithelial cell inflammation by inhibiting ROS formation
and NLRP3 inflammasome activation. Mediators Inflamm.
2019(8908960)2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Qi X, Mitter SK, Yan Y, Busik JV, Grant MB
and Boulton ME: Diurnal rhythmicity of autophagy is impaired in the
diabetic retina. Cells. 9(905)2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Chang HC and Guarente L: SIRT1 and other
sirtuins in metabolism. Trends Endocrinol Metab. 25:138–145.
2014.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Abdulnour RE, Dalli J, Colby JK,
Krishnamoorthy N, Timmons JY, Tan SH, Colas RA, Petasis NA, Serhan
CN and Levy BD: Maresin 1 biosynthesis during platelet-neutrophil
interactions is organ-protective. Proc Natl Acad Sci USA.
111:16526–16531. 2014.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Chatterjee A, Sharma A, Chen M, Toy R,
Mottola G and Conte MS: The pro-resolving lipid mediator maresin 1
(MaR1) attenuates inflammatory signaling pathways in vascular
smooth muscle and endothelial cells. PLoS One.
9(e113480)2014.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Bjørkøy G, Lamark T, Brech A, Outzen H,
Perander M, Overvatn A, Stenmark H and Johansen T: p62/SQSTM1 forms
protein aggregates degraded by autophagy and has a protective
effect on huntingtin-induced cell death. J Cell Biol. 171:603–614.
2005.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zhou R, Yazdi AS, Menu P and Tschopp J: A
role for mitochondria in NLRP3 inflammasome activation. Nature.
469:221–225. 2011.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Imai S and Guarente L: NAD+ and sirtuins
in aging and disease. Trends Cell Biol. 24:464–471. 2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Mortuza R, Feng B and Chakrabarti S: SIRT1
reduction causes renal and retinal injury in diabetes through
endothelin 1 and transforming growth factor β1. J Cell Mol Med.
19:1857–1867. 2015.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Althagafy HS, Ali FEM, Hassanein EHM,
Mohammedsaleh ZM, Kotb El-Sayed MI, Atwa AM, Sayed AM and Soubh AA:
Canagliflozin ameliorates ulcerative colitis via regulation of
TLR4/MAPK/NF-κB and Nrf2/PPAR-γ/SIRT1 signaling pathways. Eur J
Pharmacol. 960(176166)2023.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Qiang L, Wang L, Kon N, Zhao W, Lee S,
Zhang Y, Rosenbaum M, Zhao Y, Gu W, Farmer SR and Accili D: Brown
remodeling of white adipose tissue by SirT1-dependent deacetylation
of Pparγ. Cell. 150:620–632. 2012.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Picard F, Kurtev M, Chung N, Topark-Ngarm
A, Senawong T, Machado De Oliveira R, Leid M, McBurney MW and
Guarente L: Sirt1 promotes fat mobilization in white adipocytes by
repressing PPAR-gamma. Nature. 429:771–776. 2004.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Rius-Pérez S, Torres-Cuevas I, Millán I,
Ortega ÁL and Pérez S: PGC-1α, inflammation, and oxidative stress:
An integrative view in metabolism. Oxid Med Cell Longev.
2020(1452696)2020.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Yang H, Zhang W, Pan H, Feldser HG, Lainez
E, Miller C, Leung S, Zhong Z, Zhao H, Sweitzer S, et al: SIRT1
activators suppress inflammatory responses through promotion of p65
deacetylation and inhibition of NF-κB activity. PLoS One.
7(e46364)2012.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Guo Q, Jin Y, Chen X, Ye X, Shen X, Lin M,
Zeng C, Zhou T and Zhang J: NF-κB in biology and targeted therapy:
New insights and translational implications. Signal Transduct
Target Ther. 9(53)2024.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Furman BL: Streptozotocin-induced diabetic
models in mice and rats. Curr Protoc. 1(e78)2021.PubMed/NCBI View Article : Google Scholar
|