1. Introduction
Prostate cancer (PCa) is the second most common
cancer among men worldwide (1-3).
The latest estimated data from the International Agency for
Research on Cancer as of April 2025 (from the GLOBOCAN 2022
database) clearly predicts that the number of cases will double by
2040(1). Early-stage PCa typically
presents no obvious clinical symptoms, leading numerous patients to
seek medical attention only when the disease has progressed to a
middle or advanced stage. Thus, early detection of PCa is key in
effective treatment and improved patient prognosis. Clinical
screening for PCa primarily involves digital rectal examination,
prostate-specific antigen (PSA) testing and imaging studies.
Prostate biopsy remains the most established method for diagnosing
PCa. Current guidelines from The European Association of Urology,
The National Comprehensive Cancer Network and The American
Urological Association (4-7)
all designate prostate biopsy as the ‘gold standard’ for diagnosing
this cancer subtype. When conducted under standardized procedures,
prostate biopsy is considered a safe diagnostic approach. Prior to
biopsy, multi-modal imaging assessment is recommended (8).
The prostate imaging reporting and data system
(PI-RADS) (9) serves as a
standardized scoring system for evaluating prostate MRI results.
The decision to proceed with a biopsy should be based on the
PI-RADS score in conjunction with the individual circumstances of
the patient and other examination findings, such as the tumor
marker PSA test result (10). The
PI-RADS score ranges from 1-5, with higher scores indicating a
greater likelihood of PCa. A score of 3 typically indicates
moderate suspicion and does not definitively rule out malignant
lesions. The necessity for a biopsy should be determined based on a
comprehensive assessment of the clinical symptoms of the patient
(such as frequent urination and difficulty urinating), serum PSA
levels, age, family history and other factors. If PSA levels are
elevated or symptoms are severe, a biopsy may be recommended for
pathological diagnosis. In cases where the initial biopsy result is
benign but there are signs of disease progression, a repeat biopsy
may be warranted. Conversely, if the patient is in poor health,
close observation and regular follow-up may be preferred. When the
PI-RADS score is ≥4, the risk of PCa markedly increases, making
biopsy strongly advisable for a definitive diagnosis. The combined
biopsy strategy-utilizing both systematic and targeted biopsy-has
become a mainstream approach (11).
Studies have indicated that this combined method can
notably enhance the detection rate of clinically significant PCa
(csPCa), achieving rates as high as 68%, while also reducing the
likelihood of missed diagnoses (10,12-14).
As a result, improving the accuracy of biopsy equipment to elevate
the positive detection rate and facilitate early PCa diagnosis has
become a priority in the field. While transrectal ultrasound
(TRUS)-guided prostate biopsy remains a traditional method, new
imaging technologies have emerged, though they often face
challenges such as insufficient detection accuracy and low positive
biopsy rates. Recently, the introduction of prostate biopsy robots
has further advanced the field. By integrating imaging, image
fusion and artificial intelligence, these technologies simplify the
biopsy process, enhance feasibility and improve the accuracy of
outcomes. Previously published literature (15-17)
has discussed a number of possible ultrasound technologies for
prostate biopsy. The present review aimed to summarize the latest
advancements in new ultrasound technologies for targeted prostate
biopsy and to compare new technologies, whilst including
contradictory findings and cost-benefit analyses.
2. Micro-ultrasound (MUS)-guided targeted
prostate biopsy
MUS is a novel technology that provides high
resolution images, notably improving the visualization of tissue
details. Operating at a frequency of 29 MHz, MUS achieves a spatial
resolution of 70 µm, which is comparable to the size of prostate
ducts. This represents a 300% increase in spatial resolution
compared with the traditional ultrasound (18). The high resolution of the MUS
system allows for detailed visualization of the catheter anatomy
and cellular density, thereby enhancing the understanding of
prostate anatomy. Preliminary research has demonstrated that MUS
exhibits higher sensitivity in detecting PCa, establishing itself
as a promising diagnostic imaging technique for the disease. For
example, a study was conducted involving 67 patients who underwent
prostate biopsy with the ExactVu™ 29 MHz MUS system,
revealing detection rates of 56.7% for real-time MUS-guided biopsy
compared with 44.8% for traditional biopsy (19). A meta-analysis by Dariane et
al (20) further determined
that MUS-guided biopsy outperformed systematic biopsy in detecting
csPCa while identifying a number of non-csPCA cases. Therefore, MUS
represents a viable biopsy method. Future prospects for the novel
29-MHz high-resolution MUS include the development of a scoring
system, known as prostate risk identification-MUS, which aims to
enhance precision and accuracy in a PCa setting (19,21).
This scoring system assesses the severity based on the observed
structural damage, echo characteristics and boundary morphology of
the glands under high-frequency (29 MHz) MUS, ranging from 1 point
(very low risk) to 5 points (very high risk); it is a standardized
5-point Likert scale specifically designed for MUS. This system
could assist urologists in achieving accurate and reproducible
results for PCa detection, comparable to those obtained using
MRI-based PI-RADS scoring.
3. Contrast-enhanced ultrasound
(CEUS)-guided targeted prostate biopsy
CEUS involves the injection of microbubbles of
contrast agent into the peripheral veins of patients to observe the
filling of the prostate with the contrast agent, which reflects the
perfusion of prostate microvessels. Targeted biopsy of the prostate
guided by CEUS can enhance the detection rate of PCa (22). A commonly used ultrasound contrast
agent in clinical practice is SonoVue (Bracco), also known as
Sonofo Micro-Sulfur Hexafluoride Bubbles. With a diameter of ~2.5
µm, it is similar in size to red blood cells in the blood. After
being injected through the median vein in the elbow, it can freely
pass through the blood and reach the target organ or tissue.
SonoVue is currently the most widely used ultrasound contrast
microbubble system and is a pure blood pool contrast agent that
does not penetrate the extravascular space. This characteristic
allows it to more accurately reflect the morphological features and
blood perfusion of tumors. Qi et al (23) found that contrast-enhanced TRUS was
markedly more accurate compared with traditional grayscale imaging
in measuring the size of prostate tumors, with a mean
underestimation of ~3.9 mm for grayscale imaging compared with 0.6
mm for contrast-enhanced transrectal ultrasonography. In addition,
Wang et al (24) discovered
that molecular imaging of in situ PCa using nano-bubble
ultrasound contrast agents targeting membrane PSA demonstrated
notable advantages in diagnosing PCa, such as reduced radiation,
lower costs and reduced patient waiting time. Additional studies
have indicated that CEUS not only increases the detection rate of
PCa but also aids in the grading and staging of the disease
(25-28).
However, the accuracy of the examination can vary based on the
experience of the physician, the obesity level of the patient and
the shape of the prostate.
4. Ultrasound elastography-guided targeted
prostate biopsy
Ultrasound elastography is an innovative ultrasonic
diagnostic technology that measures changes in the amplitude of
echo signals before and after tissue compression, allowing for the
assessment of tissue hardness. This technique effectively
differentiates between diseased and normal tissue based on the
varying elastic coefficients of different tissues (29). A notable advantage of ultrasound
elastography is its ability to detect lesions through changes in
tissue hardness, thereby improving the detection rate of lesions
compared with traditional 2D gray-scale ultrasound imaging-guided
puncture biopsy. Its application in the medical field has been
gradually integrated (30). PCa
tumor cells proliferate rapidly and are invasive, resulting in a
higher number of cancer cells and a harder texture in the lesion
area compared with the surrounding normal tissue. Notably, changes
in tissue hardness occur earlier compared with alterations in
anatomical structure (31) and
ultrasound elastography can identify the hardness of internal
prostate tissues, thereby revealing lesions. In a prospective study
conducted by Brock et al (32), 231 patients with suspected PCa were
randomly assigned to either a transrectal real-time
elastography-guided biopsy group or a transrectal gray-scale
ultrasound-guided biopsy group. The findings indicated a
significantly higher cancer detection rate (P=0.007) in patients
undergoing biopsy with real-time elastography (40.5%) compared with
the gray-scale group (23.8%). Sensitivity and specificity for
detecting PCa across 1,386 prostate sectors were 53.5 and 70.5% for
real-time elastography, compared with 11.7 and 93.7% for gray-scale
ultrasound, respectively. This demonstrated a significant
improvement in the accuracy of PCa detection under the guidance of
real-time elastography compared with traditional gray-scale
ultrasound.
Currently, the main applications of prostate
ultrasound elastography include strain elastography (SE) and shear
wave elastography (SWE), both of which can guide targeted prostate
biopsies. SE involves manually applying pressure to the tissue with
a probe. Due to differences in hardness, the tissue undergoes
varying deformations, which are visualized in different colors on
the imaging, with harder tissue appearing blue and softer tissue
appearing red (33). Although SE
results cannot be quantitatively analyzed, semi-quantitative
information can be obtained by measuring the strain rate between
the region of interest and the surrounding normal tissue. SE can
perform real-time imaging in any area within the penetration range
and targeted biopsies guided by SE, when combined with systematic
biopsies, can markedly enhance the detection rate of PCa. Salomon
et al (34) found detection
rates of 39.1 and 29.0% for systematic and SE-targeted biopsies,
respectively, with a combined detection rate of 46.2%. Furthermore,
a study by Boehm et al (35) demonstrated that real-time
elastography-targeted biopsy achieved a higher overall Gleason
assignment accuracy (68.3 vs. 56.7%; P=0.008) compared with
systematic biopsy alone. However, the main drawbacks of SE include
the need for manual pressurization, poor repeatability and a high
dependence on the skill and experience of the operator (36).
By contrast, SWE measures the propagation speed of
shear waves within tissues to calculate the Young's modulus value,
providing a real-time quantitative graph of the elastic properties
of soft tissues. In SWE imaging, harder tissues appear red, while
softer tissues appear blue. SWE offers a number of advantages, as
it does not require manual pressurization, allows for quantitative
measurements, provides objective values, has high repeatability and
is less dependent on the experience of the operator (37). A meta-analysis by Anbarasan et
al (38) indicated that SWE
exhibited high sensitivity (77%) and specificity (84%) in detecting
csPCa. Fu et al (39)
conducted SWE on three sections of the prostate, namely the level
of the base, mid-gland and apex of the prostate peripheral zone,
along with suspected lesions identified by 2D ultrasound in a study
exploring peripheral PCa. The results revealed that with a critical
threshold of 42 kPa, SWE demonstrated high sensitivity,
specificity, positive predictive value, negative predictive value
and accuracy in differentiating between benign and malignant
lesions. Furthermore, as the Gleason score and prognosis grade
increased, there was a trend toward a higher Young's modulus values
in PCa lesions. Therefore, in addition to identifying PCa lesions,
SWE holds marked potential for reducing unnecessary biopsies and
guiding the acquisition of more valuable tissue during puncture, as
well as predicting the pathological score and prognosis of cancer
foci.
5. Multiple parameter MRI-TRUS cognitive
fusion-guided targeted prostate biopsy
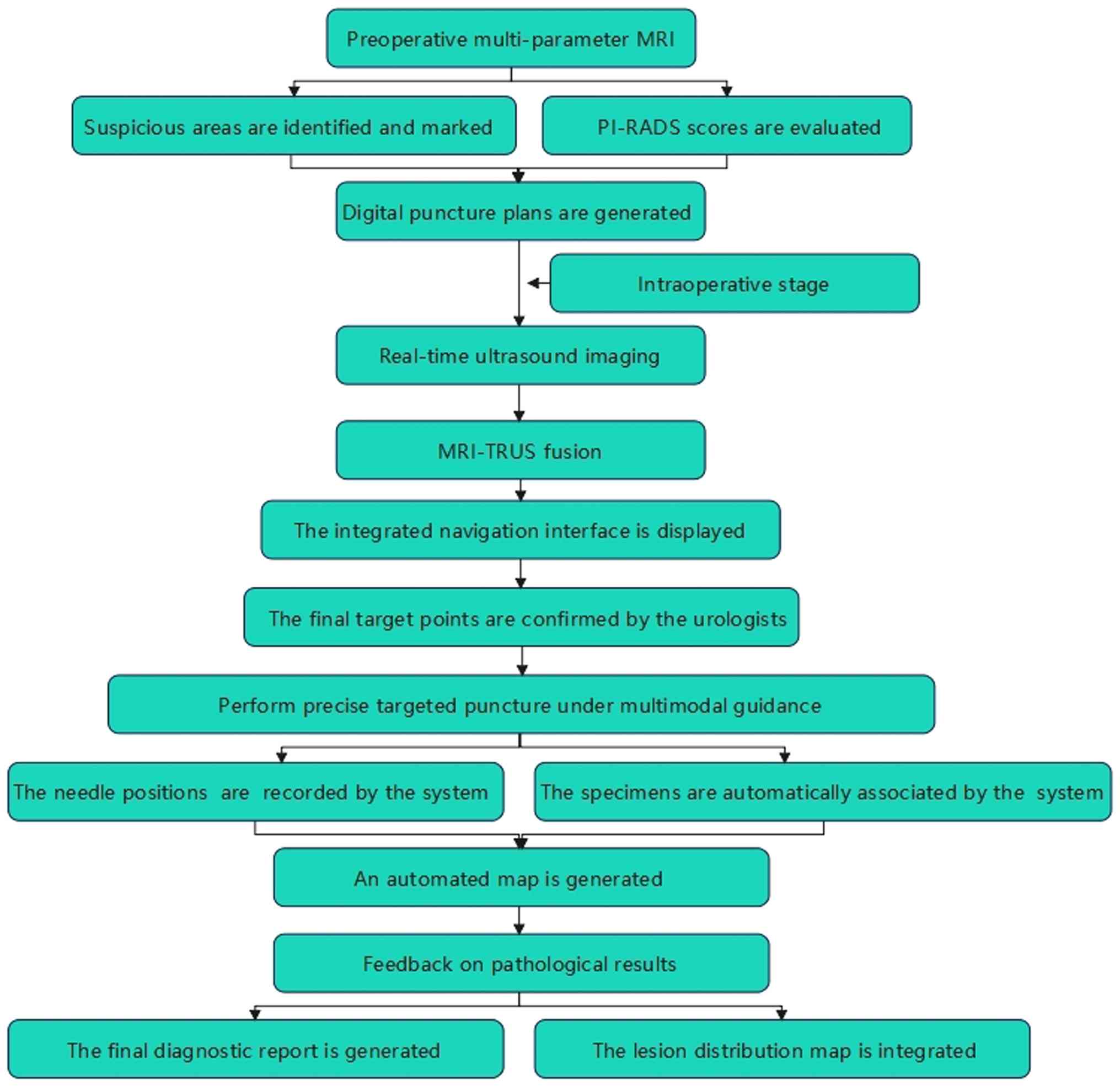
MRI-TRUS cognitive fusion image-guided biopsy
involves integrating a number of MRI scans prior to the procedure
with real-time TRUS examination images captured during the biopsy.
The target lesion is identified using the MRI images and the biopsy
is performed under the guidance of TRUS images (40). This technique requires the
physician performing the biopsy to possess strong film
interpretation skills. Its advantages include real-time visibility
of the lesion during the procedure, accurate targeting for the
biopsy and elimination of the need to scan numerous MRI sequences
(Fig. 1). Additionally, random
systematic biopsies of the prostate can be conducted simultaneously
under the guidance of TRUS (41).
A study by Drăgoescu et al (42) regarding MRI cognitive
fusion-targeted perineal prostate biopsy indicated that this
approach outperformed systematic biopsy in detecting overall PCa in
larger patient populations and was particularly effective in
identifying csPCa. This improvement helped one-fifth of patients
avoid missing necessary treatment for PCa. Kuliš et al
(43) compared cognitive
fusion-targeted biopsy and systematic prostate biopsy in patients
with repeated negative systematic biopsies but ongoing clinical
suspicion of PCa. The findings revealed that cognitive targeted
prostate biopsy based on numerous MRI scans was superior in
diagnosing patients, particularly those with persistently elevated
PSA levels.
However, this technique relies heavily on the
experience of the puncture operator, which introduces a degree of
subjectivity. The biopsy results may exhibit marked uncertainty
when performed by less experienced practitioners (44). It is also important to consider the
potential drawbacks associated with MRI, including costs and the
occurrence of artifacts (42-44).
MRI equipment is expensive and the examination cost is high, and
its accessibility is limited in some areas. Besides, MRI scans take
a long time and have complex sequences, requiring high patient
cooperation. Gas in the rectum and metal implants (such as hip
joint prostheses) can cause uneven local magnetic fields, resulting
in artifacts, severe image distortion or signal loss.
6. Multiple MRI-TRUS software
integration-guided targeted prostate biopsy
Multiple MRI-TRUS software integration-guided
targeted prostate biopsy involves storing prostate MRI image data
in specialized equipment that is fused with intraoperative
real-time ultrasound images. This fusion is achieved automatically
and intelligently by the machines, clearly identifying the target
area, tracking the TRUS probe and determining the optimal puncture
biopsy path (45). This approach
reduces the reliance on the ability of the puncture physician to
interpret films. The combination of precise positioning from
prostate MRI with real-time ultrasound imaging technology meets the
requirements for accurately locating and tracking prostate lesion
sites. This technology notably enhances the ability to identify
prostate lesions during procedures and improves the accuracy of PCa
punctures (46).
A prospective study conducted by Fiard et al
(47) showed that 30 patients with
suspected PCa who underwent MRI examinations also received routine
12-needle systematic biopsies, along with two additional targeted
biopsies of suspicious areas. When comparing the results of
systematic and targeted biopsies, both methods demonstrated a
sensitivity of 91% in detecting cancer; however targeted biopsies
required fewer needles. In an additional study by Yarlagadda et
al (48), which included both
prospective and retrospective analyses, patients underwent
systematic biopsies guided by TRUS and biopsies guided by the
fusion of multiple MRI-TRUS software. The findings indicated that,
while maintaining the same cancer detection rate, significantly
fewer needle cores were sampled during the MRI-TRUS software-guided
biopsy, with a reduction of 63% in cores taken (P<0.001).
Currently, numerous types of multiple MRI-TRUS
software integration-guided biopsy navigation systems are available
on the market, differing in aspects such as software interface,
image fusion methods and biopsy path selection (49). These platforms differ in terms of
software interface, volumetric-USG acquisition technique, needle
tracking method, image fusion algorithm and biopsy route. However,
the high cost of these devices limits their widespread adoption in
a short timeframe, restricting their overall application.
7. Robot-assisted ultrasound-guided
biopsy
Robot-assisted ultrasound-guided biopsies utilize a
mechanical arm to accurately position the biopsy needle in 3D,
determining the direction of the needle, puncture depth and biopsy
location while allowing for the removal of all biopsy tissues from
a single incision. This system can automatically set the puncture
depth, enhance needle placement accuracy and reduce operation time
(50). While ultrasound offers
low-cost, dynamic real-time imaging, it exhibits marked
limitations, as cancer lesions often remain invisible, requiring
urologists to manually adjust the TRUS probe during biopsy
procedures to achieve clearer ultrasound images. In addition,
manual operation of the TRUS probe and biopsy needle poses safety
concerns. Targeted prostate biopsies risk needle deformation due to
device imaging errors or variations in physician skill, which may
lead to incorrect targeting and excessive needle insertions,
resulting in complications, such as septic complications and acute
urinary retention (51). These
issues highlight the current limitations in puncture methods and
accuracy.
With advancements in artificial intelligence,
researchers are beginning to integrate engineering, minimally
invasive surgery and imaging to develop robot-assisted puncture
systems (52-54)
aimed at improving puncture accuracy, reducing the number of biopsy
cores needed and alleviating patient pain. Compared with manual
biopsies, robot-assisted ultrasound-guided prostate biopsies allow
for more precise needle placement with minimum reliance on
physician experience or imaging examinations (55).
Robot-assisted prostate biopsy can alleviate the
workload for urologists. An advantage of this system is that
numerous biopsies can be performed at a single puncture site;
however, the accuracy of the biopsies still heavily depends on the
proficiency of the urologist. Phee et al (56) introduced a prototype robotic system
designed for the accurate and consistent insertion of biopsy
needles into the prostate. This system employed a TRUS probe to
capture a series of 2D images of the prostate, which are then used
to create a 3D computer model for the organ. Urologists utilize
this model to determine biopsy points within the prostate, after
which the robotic system calculates the necessary needle insertion
positions, allowing for the actual biopsy to be performed. Ho et
al (57) designed an
ultrasound-guided perineal double-cone concept
six-degree-of-freedom prostate biopsy robot system, which has
demonstrated safety and accuracy in achieving precise prostate
biopsies without harming the urethra of the patient.
Lim et al (55) proposed a robot-assisted system for
TRUS-guided prostate biopsy, notable for its needle aiming accuracy
of ~1 mm, establishing it as a feasible and safe option for
assisting prostate biopsies. However, one limitation of this system
is the lack of a supporting structure to maintain the leg posture
of the patient, increasing the uncertainty of patient position
changes during the procedure, which may affect needle tip
positioning accuracy. Yan et al (58) introduced an eight-degree-of-freedom
robot system designed for the operation, positioning and insertion
of ultrasound probes, aiding physicians in achieving ultrasound
probe scanning, needle position adjustments and needle insertions.
This structure boasts high rigidity and compactness, facilitating
the required movements of both the probe and needle while
minimizing the risk of conflict between the robot and the
patient.
8. Conclusion and outlook
MUS, CEUS and ultrasound elastography are cheaper
and easier to obtain compared with other novel technologies.
MRI-TRUS cognitive fusion-guided targeted prostate biopsy is not
routinely used due to its high cost, time-consumption and
complicated operational procedures. Patients with negative TRUS but
positive MRI can undergo MRI-TRUS in suspicious malignant areas.
MRI-TRUS image fusion technology allows urologists to progress from
blind, systematic biopsy to targeted and tracked biopsy. Multiple
MRI-TRUS software integration-guided targeted biopsy requires
pre-biopsy multiparametric MRI data, which are obtained and stored
in a specific storage hard device. This enables the real-time TRUS
imaging and serves a complementary role in systematic biopsy,
providing an objective basis for the development of clinical
diagnosis and treatment plans. The use of a fusion device makes it
costlier compared with systematic biopsy. Multiple MRI-TRUS
software integration is convenient, fast and enables real-time
imaging and is likely to show high clinical application prospects.
With regard to cost-benefit analysis, it has been established that
MRI-TRUS cognitive fusion and multiple MRI-TRUS software
integration-guided targeted prostate biopsy exhibit improved
benefits, with costs decreasing annually and the positive rate of
puncture increasing. However, there are still certain problems with
MRI-TRUS software benefits. The software sale cost is relatively
high and the sales growth rate is not as fast as the cost growth
rate, resulting in poor sales benefits. Human error is inevitable
in systematic biopsy and the accuracy of diagnosis has not reached
the desired effect (59). The
application of robot-assisted ultrasound may represent a novel
solution to the current dilemma of PCa diagnosis. Robot-assisted
ultrasound can not only analyze the imaging information of PCa but
also integrate any other diagnosis or treatment information
regarding the patient, thus improving the accuracy of
prostate-targeted biopsy and effectively monitoring the progress of
PCa. However, robot-assisted ultrasound still needs to overcome its
limitations, including the lack of extensive multi-center testing,
unified industry standards, as well as issues with sharing and
privacy, such as data transmission security, protection of
sensitive information and legal compliance responsibilities. Yet,
overall, the development of robot-assisted ultrasound should bring
about promising changes to the diagnosis process (8).
MUS, contrast-enhanced ultrasound, ultrasound
elastography and MRI-TRUS fusion imaging serve key roles in
targeted prostate biopsy, although each method exhibits limitations
and deficiencies (Table I).
Robot-assisted targeted biopsy of the prostate not only improves
detection accuracy and stability but also reduces operation time
and the incidence of surgical complications, thereby decreasing the
labor intensity for physicians. However, further research is
required regarding robot-assisted prostate biopsy systems. Future
investigations should aim to focus on refining and enhancing new
ultrasound technologies that guide targeted punctures.
 | Table IComparisons between modalities. |
Table I
Comparisons between modalities.
| Technology | Basic
principles | Advantages | Main
limitations | Clinical
applications | Disadvantages | (Refs.) |
|---|
| MUS | The operating
frequency is 29 MHz, with a spatial resolution of 70 µm, which is
300% higher compared with that of traditional ultrasound. It can
clearly display the anatomical structure and cell density of the
prostate ducts. | It exhibits high
spatial resolution and a higher sensitivity for PCa detection. It
is superior to systematic biopsy in detecting csPCa and can reduce
the detection of non-csPCa. | As a relatively new
technology, its popularity and long-term clinical data may still be
accumulating. | As a promising
diagnostic imaging technology, it offers a new high-resolution
option for targeted prostate biopsy. | The imaging quality
is uneven at the apex and base of the prostate, which may indicate
sampling bias; it has relatively low sensitivity for non-glandular
tumors (such as sarcomas). | (18-21) |
| CEUS | Intravenous
infusion of microbubble contrast agent (such as SonoVue) is
utilized to determine the filling status of the contrast agent
within the prostate to reflect micro-vascular perfusion, thereby
identifying tumor lesions. | It accurately
measures the size of prostate tumors; the targeted PSMA nano-bubble
contrast agent has pronounced advantages in molecular imaging
diagnosis; it may be helpful for the grading and staging of
PCa. | The use of contrast
agents increases the number of procedures and costs; the
interpretation of blood perfusion requires certain experience. | By reflecting the
hemodynamic characteristics of the tumor, the positive detection
rate of PCa is improved, providing a functional basis for targeted
puncture. | It is markedly
affected by benign lesions such as prostatitis and hyper-plasia and
has a relatively high false positive rate; the contrast agent shows
poor imaging in low-blood-flow tumors. | (22-28) |
| Ultrasound
elastography | With reference to
the differences in the elastic coefficients of numerous tissues,
the deformation of the tissues under pressure is converted into a
color image, thereby providing information on tissue hardness to
distinguish between diseased and normal tissues. | SE can be imaged in
real time and combined with system biopsy to notably improve the
detection rate. SWE can quantitatively measure, has strong
objectivity and high repeatability. It has a high sensitivity (77%)
and specificity (84%) for the detection of csPCa and the Young's
modulus value is positively associated with the Gleason score. | SE requires manual
pressure application, has poor repeatability and relies heavily on
the operator's experience. SWE has further requirements for
equipment and technology. | By detecting
changes in tissue hardness (PCa tissues are usually harder), SWE
shows great potential in predicting the pathological score and
prognosis of cancer lesions. | Insufficient
imaging stability in deep prostate tissue (especially the posterior
lobe); prone to misjudging fibrotic or calcified areas as
malignant. | (29-39) |
| MRI-TRUS
fusion | The preoperative
high-resolution MRI images are fused with the real-time TRUS images
during the operation. The target area is precisely located using
MRI and the puncture is completed under ultrasound guidance. | By combining the
precise positioning capabilities of MRI with the real-time imaging
advantages of ultrasound, the software fusion requires minimal
operator reading skills while exhibiting a high degree of
intelligence. This approach can markedly reduce the number of
puncture needles needed, while maintaining the same detection
rate. | Cognitive fusion is
highly dependent on the experience of the operator, with a strong
subjective element. Software fusion equipment is expensive and it
cannot be widely adopted in the short term. | It can increase the
detection rate of csPCa. Notably, in patients with negative
systematic biopsy but with ongoing clinical suspicion, its
diagnostic value is even higher. It is an important development
direction for current precise puncture techniques. | The fusion error
(especially the deformation of the prostate during the operation)
remains a notable challenge; the accuracy of locating atypical or
diffuse lesions is further compromised. | (40-49) |
| Robot assisted
ultrasound | Utilizing the
robotic arm for precise 3D positioning, automatically planning the
puncture path and depth and combining AI and imaging fusion
technology to guide the puncture. | The puncture
accuracy is high (~1 mm level), which can reduce the reliance on
the operator, lower the surgical complications and has the
potential for remote operation. | The equipment is
costly, the technology integration is still in its early stages and
the lack of supporting structures may affect the stability of the
patient position, thereby impacting the accuracy. | It represents a
future development direction of puncture technology, aiming to
achieve full automation and intelligence, ultimately enhancing the
accuracy, stability and safety of puncture. | Recent research
indicates that there are still algorithmic limitations in its
multi-target area and multi-angle puncture path planning; the
learning curve is steep and the popularization and promotion are
slow. | (50-58) |
In addition, it is important to improve upon the
ability of the robot in accurate imaging fusion of real-time and 3D
images, as well as to facilitate intelligent interaction through
voice and gesture commands. This would enhance the efficiency of
physician operations, enable remote control capabilities and
provide patients with convenient access to medical care, even if
the patients are far away from the urologists (60). Enhancing the self-decision-making
abilities of robots, ensuring they avoid collisions with patients
during surgeries and preventing accidental punctures of key
Urologists should select the appropriate technology for patients
based on their specific conditions. For example, for patients with
obesity, MRI navigation or robot-assisted surgery is recommended
due to the clear operating field and high flexibility of the
instruments. In addition, patients who are allergic to ultrasound
contrast agents should opt for non-contrast imaging techniques,
such as MUS, ultrasound elastography or MRI navigation and
robot-assisted surgery (10,19,22,30).
For those with a number of underlying diseases, it is important to
consider the opinions of different disciplines and balance the
feasibility and safety of the chosen technology (61). For example, for patients with
severe cardiovascular diseases and the use of anticoagulant drugs,
a cardiologist is required to assess the risks of discontinuing
antiplatelet drugs and the urologist needs to evaluate the
necessity of biopsy and the risk of bleeding. Urologists should
reasonably determine the technical approach based on their
experience and the resources available to them.
Overall, to facilitate clinical translation, the
following suggestions are proposed: i) Establish a standardized
multi-modal imaging acquisition and fusion operation procedure; ii)
provide specialized training for urologists in robot-assisted
targeted biopsy; and iii) integrate personalized biopsy plans into
clinical pathway guidelines for the early diagnosis of PCa. With
advancements in medicine and new ultrasound technologies, the
application of multi-modal fusion and robot-assisted
ultrasound-guided targeted prostate biopsy will become increasingly
valuable when paired with personalized plans.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
TJ and LW wrote and edited the present manuscript.
SZ revised the manuscript draft. All authors have read and approved
the final version of the manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dee EC, Iyengar R, Narayan A, Feliciano
EJG, Wu JF, Ho FDV, Ng K, Willmann J, Cabaero MLL, Tan AKNG, et al:
National cancer system characteristics and prostate cancer
outcomes: An analysis of global data. Prostate. 85:947–953.
2025.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Bergengren O, Pekala KR, Matsoukas K,
Fainberg J, Mungovan SF, Bratt O, Bray F, Brawley O, Luckenbaugh
AN, Mucci L, et al: 2022 Update on prostate cancer epidemiology and
risk factors-A systematic review. Eur Urol. 84:191–206.
2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Yuan H, Jiang Y, Tan Y and Xiang Y:
Current status and time trends of cancer incidence and mortality
worldwide. Cancer Rese Prev Treat. 48:642–646. 2021.
|
|
4
|
Cornford P, Tilki D, van den Bergh RCN,
Eberli D, Fonteyne V, Gandaglia G, Gillessen S, Henry AM, van
Leenders GJLH, Oldenburg J, et al: EAU-EANM-ESTRO-ESUR-ISUP-SIOG
guidelines on prostate cancer. European Association of Urology,
Arnhem, The Netherlands, 2025.
|
|
5
|
National Comprehensive Cancer Network
(NCCN): NCCN clinical practice guidelines in oncology (NCCN
Guidelines®): Prostate cancer version 2.2025. NCCN, Plymouth
Meeting, PA, 2025.
|
|
6
|
Wei JT, Barocas D, Carlsson S, Coakley F,
Eggener S, Etzioni R, Fine SW, Han M, Kim SK, Kirkby E, et al:
Early detection of prostate cancer: AUA/SUO guideline part I:
Prostate cancer screening. J Urol. 210:46–53. 2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Eastham JA, Auffenberg GB, Barocas DA,
Chou R, Crispino T, Davis JW, Eggener S, Horwitz EM, Kane CJ,
Kirkby E, et al: Clinically localized prostate cancer: AUA/ASTRO
guideline, part I: Introduction, risk assessment, staging, and
risk-based management. J Urol. 208:10–18. 2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Aymaz S, Oğuz NK, Aymaz Ş, Aydın HR,
Okatan AE, Kadıoğlu ME and Bulut E: Adaptive ensemble learning for
prostate cancer classification on multi-modal MRI: Reducing
unnecessary biopsies. BMC Med Imaging. 26(76)2026.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bischoff LM, Endler C, Krausewitz P,
Ellinger J, Klümper N, Isaak A, Mesropyan N, Kravchenko D, Kuetting
D, Sprinkart AM, et al: Acquisition, image quality, and PI-RADS
agreement of ultrahigh-gradient DWI in prostate 3-T MRI. Eur Radiol
Exp. 10(17)2026.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Trappe S, Schimmöller L, Althoff P, Schero
KJ, Berg S, Radtke JP, Esposito I, Roghmann F, Albers P, Antoch G,
et al: Diagnostic value of prostate magnetic resonance imaging in
men with prostate-specific antigen levels ≥15 ng/ml for biopsy
decision-making. Insights Imaging. 17(57)2026.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Falagario UG, Sanguedolce F, Dovey Z,
Carbonara U, Crocerossa F, Papastefanou G, Autorino R, Recchia M,
Ninivaggi A, Busetto GM, et al: Prostate cancer biomarkers: A
practical review based on different clinical scenarios. Crit Rev
Clin Lab Sci. 59:297–308. 2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gann PH, Stackhouse N, Gastala N, Ma W,
Wright ME, Watson K, Stepping C, King-Lee P, Xu Z, Patel T and
Abern MR: A trial of risk-adapted prostate cancer screening in a
federally supported health center network serving a high-risk
population. Cancer. 132(e70340)2026.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Nabaasa S, Mutakooha MM, Amadile L,
Bagenda CN, Ninsiima JL, Birungi A, Atwine R, Wasswa H, Kasadha R,
Lauben T and Ssedyabane F: Correlating histological results and
total serum levels of the prostate-specific antigen among patients
in southwestern Uganda. Prostate Cancer.
2026(9924021)2026.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Matsumoto S and Takasu S: Postmortem serum
prostate-specific antigen as a potential marker for prostatic
disease: A forensic exploratory study. Cureus.
18(e101818)2026.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Liang Y, Yang C, Zhuo Y, Li Y, Zhang L and
Huang J: Advances in application of new ultrasound technologies in
prostate-targeted biopsy. J Radiol Med Imaging. 3(1034)2020.
|
|
16
|
Liu Y, Xiang L, Xu G, Zhang Y and Xu H:
Recent advances of multimoda ultrasound in image-guided
prostate-targeted biopsy. J Interv Med. 5:117–121. 2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kang H and Hu M: Recent advances in
targeted prostate cancer diagnosis and therapy using ultrasound
imaging combined with novel nanocarriers:facilitating early
detection and effective treatment of prostate cancer. Cancer Treat
Res Commun. 45(100994)2025.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Al-Qurri A, Thaher A and Almekkawy MK:
Enhanced deep neural network for prostate segmentation in
micro-ultrasound images. Sensors (Basel). 25(6815)2025.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ghai S, Eure G, Fradet V, Hyndman ME,
McGrath T, Wodlinger B and Pavlovich CP: Assessing cancer risk on
novel 29 MHz micro-ultrasound images of the prostate: Creation of
the micro-ultrasound protocol for prostate risk identification. J
Urol. 196:562–569. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Dariane C, Ploussard G, Barret E, Beauval
JB, Brureau L, Créhange G, Fromont G, Gauthé M, Mathieu R,
Renard-Penna R, et al: Micro-ultrasound-guided biopsies versus
systematic biopsies in the detection of prostate cancer: A
systematic review and meta-analysis. World J Urol. 41:641–651.
2023.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Diana P, Lughezzani G, Saita A, Uleri A,
Frego N, Contieri R, Buffi N, Balzarini L, D'Orazio F, Piergiuseppe
C, et al: Head-to-head comparison between high-resolution
microultrasound imaging and multiparametric MRI in detecting and
local staging of bladder cancer: The BUS-MISS protocol. Bladder
Cancer. 8:119–127. 2022.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Wink M, Frauscher F, Cosgrove D, Chapelon
JY, Palwein L, Mitterberger M, Harvey C, Rouvière O, de la Rosette
J and Wijkstra H: Contrast-enhanced ultrasound and prostate cancer;
a multicentre European research coordination project. Eur Urol.
54:982–992. 2008.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Qi TY, Chen YQ, Jiang J, Zhu YK, Yao XH
and Qi J: Contrast-enhanced transrectal ultrasonography:
Measurement of prostate cancer tumor size and correlation with
radical prostatectomy specimens. Int J Urol. 20:1085–1091.
2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Wang Y, De Leon AC, Perera R, Abenojar E,
Gopalakrishnan R, Basilion JP, Wang X and Exner AA: Molecular
imaging of orthotopic prostate cancer with nanobubble ultrasound
contrast agents targeted to PSMA. Sci Rep. 11(4726)2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Pallwein L, Mitterberger M, Gradl J,
Aigner F, Horninger W, Strasser H, Bartsch G, zur Nedden D and
Frauscher F: Value of contrast-enhanced ultrasound and elastography
in imaging of prostate cancer. Curr Opin Urol. 17:39–47.
2007.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Liu B, He H, Zhao Y, Cui Y and Wang J: The
diagnostic value of two-dimensional ultrasound score,
contrast-enhanced ultrasound score and ultrasound elastography
score in prostate cancer. Transl Androl Urol. 13:1805–1813.
2024.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Pauchard F, Kramer F, Kirmayr M and
Escobar M: Stage at diagnosis of prostate cancer in an
institutional hospital. Review and comparison of national and
international data. Rev Med Chil. 151:711–716. 2023.PubMed/NCBI View Article : Google Scholar : (In Spanish).
|
|
28
|
Safdar H, Sardar M, Tekchandani N,
Iftikhar A, Iftikhar H, Ghumman F and Burki J: Clinical utility of
contrast-enhanced ultrasound (CEUS) in urology: A multisystem
review. Cureus. 17(e94690)2025.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Liang J and Zhao Y: Ultrasound
elastography in prostate diseases: Current status and future
directions-a review. J Clin Ultrasound. 53:2117–2129.
2025.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wang J, Tong M, Lu H, et al: The
application of ultrasound elastography technology in the diagnosis
of prostate cancer. Mod Biomed Prog. 14:3797–3794. 2014.
|
|
31
|
Lu J, Zhu J, Zhang SK, Qi P, Shen P, Liu G
and Ge W: Comparison of diagnostic value of different ultrasound
techniques for prostate cancer with different gleason scores. Chin
J Ultrasound Med. 37:197–200. 2021.(In Chinese).
|
|
32
|
Brock M, Eggert T, Löppenberg B, Braun K,
Roghmann F, Palisaar RJ, Noldus J and von Bodman C: Value of
real-time elastography to guide the systematic prostate biopsy in
men with normal digital rectal exam. Aktuelle Urol. 44:40–44.
2013.PubMed/NCBI View Article : Google Scholar : (In German).
|
|
33
|
Urban M, Vasconcelos L, Brom K, Dave J and
Kijanka P: Shear wave elastography primer for the abdominal
radiologist. Abdom Radiol (NY). 50:3744–3763. 2025.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Salomon G, Drews N, Autier P, Beckmann A,
Heinzer H, Hansen J, Michl U, Schlomm T, Haese A, Steuber T, et al:
Incremental detection rate of prostate cancer by real-time
elastography targeted biopsies in combination with a conventional
10-core biopsy in 1024 consecutive patients. BJU Int. 113:548–553.
2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Boehm K, Tennstedt P, Beyer B, Schiffmann
J, Beckmann A, Michl U, Beyersdorff D, Budäus L, Graefen M,
Karakiewicz PI and Salomon G: Additional elastography-targeted
biopsy improves the agreement between biopsy Gleason grade and
Gleason grade at radical prostatectomy. World J Urol. 34:805–810.
2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Ferraioli G, Barr RG, Farrokh A, Radzina
M, Cui XW, Dong Y, Rocher L, Cantisani V, Polito E, D'Onofrio M, et
al: How to perform shear wave elastography. Part I. Med Ultrason.
24:95–106. 2022.PubMed/NCBI View
Article : Google Scholar
|
|
37
|
Keskin ET, Kaplanoglu V, Senocak C, Basar
H and Bozkurt OF: Transrectal shear wave elastography for detection
of prostate cancer. Urologia. 90:230–235. 2023.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Anbarasan T, Wei C, Bamber JC, Barr RG and
Nabi G: Characterisation of prostate lesions using transrectal
shear wave elastography (SWE) ultrasound imaging: A systematic
review. Cancers Basel. 13(122)2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Fu S, Tang Y, Tan S, Zhao Y and Cui L:
Diagnostic value of transrectal shear wave elastography for
prostate cancer detection in peripheral zone: Comparison with
magnetic resonance imaging. J Endourol. 34:558–566. 2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Li Y, Zhou S, Chen J, Mao F, Niu XB, Sun
L, Xu M and Liu JT: PI-RADS v2.1 score combined with PSA density
for diagnosis of clinically significant prostate cancer in the PSA
grey zone by MRI-TRUS cognitivefusion-guided transperineal targeted
prostate biopsy. Zhonghua Nan Ke Xue. 31:50–54. 2025.PubMed/NCBI(In Chinese).
|
|
41
|
Li D, Liu Y, Li Z, et al: A single-center
clinical study of 614 cases of transrectal targeted prostate biopsy
guided by the cognitive fusion of magnetic resonance and
transrectal ultrasound. J Biomed Eng. 37:225–229. 2020.
|
|
42
|
Drăgoescu PO, Drocaș AI, Drăgoescu AN,
Pădureanu V, Pănuș A, Stănculescu AD, Radu MA, Florescu LM, Gheonea
IA, Mirea C and Mitroi G: Transperineal prostate biopsy targeted by
magnetic resonance imaging cognitive fusion. Diagnostics (Basel).
13(1373)2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Kuliš T, Zekulić T, Alduk AM, Lušić M,
Bulimbašić S, Ferenčak V, Mokos I, Hudolin T and Kaštelan Ž:
Targeted prostate biopsy using a cognitive fusion of
multiparametric magnetic resonance imaging and transrectal
ultrasound in patients with previously negative systematic biopsies
and non-suspicious digital rectal exam. Croat Med J. 61:49–54.
2020.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Yin S, Wang J and Shan L: Research
progress in application of multi-parametric magnetic resonance
imaging and transrectal ultrasound cognitive fusion technology in
targeted prostate biopsy. J Clin Med Pract. 27:130–133. 2023.
|
|
45
|
Ito M, Yonese I, Toide M, Ikuta S,
Kobayashi S and Koga F: Superior detection of significant prostate
cancer by transperineal prostate biopsy using MRI-transrectal
ultrasound fusion image guidance over cognitive registration. Int J
Clin Oncol. 28:1545–1553. 2023.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Li Y and Zeng T: Research progress of
robot and imaging techniques in prostate biopsy. J Clin Urol.
38:315–320. 2023.
|
|
47
|
Fiard G, Hohn N, Descotes JL, Rambeaud JJ,
Troccaz J and Long JA: Targeted MRI-guided prostate biopsies for
the detection of prostate cancer: Initial clinical experience with
real-time 3-dimensional transrectal ultrasound guidance and
magnetic resonance/transrectal ultrasound image fusion. Urology.
81:1372–1378. 2013.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Yarlagadda VK, Lai WS, Gordetsky JB,
Porter KK, Nix JW, Thomas JV and Rais-Bahrami S: MRI/US
fusion-guided prostate biopsy allows for equivalent cancer
detection with significantly fewer needle cores in biopsy-naive
men. Diagn Interv Radiol. 24:115–120. 2018.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Das CJ, Razik A, Sharma S and Verma S:
Prostate biopsy: When and how to perform. Clin Radiol. 74:853–864.
2019.
|
|
50
|
Kroenig M, Schaal K, Benndorf M,
Soschynski M, Lenz P, Krauss T, Drendel V, Kayser G, Kurz P, Werner
M, et al: Diagnostic accuracy of robot-guided, software based
transperineal MRI/TRUS fusion biopsy of the prostate in a high risk
population of previously biopsy negative men. Biomed Res Int.
2016(2384894)2016.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Sivaraman A, Ramasamy V, Aarthy P, Sankar
V and Sivaraman PB: Safety and feasibility of freehand
transperineal prostate biopsy under local anesthesia: Our initial
experience. Indian J Urol. 38:34–41. 2022.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Zhang W, Xia P, Liu S, Huang X, Zhao X,
Liu Z, Dang H, Li X and Niu G: A coordinate positioning puncture
method under robot-assisted CT-guidance: Phantom and animal
experiments. Minim Invasive Ther Allied Technol. 31:206–215.
2022.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Zou Y, Lan T, Zhang Y, Lai X, Zhou Z and
Li C: Parallel cable-driven prostate puncture robot: Design and
performance evaluation. Int J Med Robot. 21(e70102)2025.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Chen X, Yan Y, Li A, Wang T and Wang Y:
Robot-assisted needle insertion for CT-guided puncture:
Experimental study with a phantom and animals. Cardiovasc Intervent
Radiol. 46:128–135. 2023.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Lim S, Jun C, Chang D, Petrisor D, Han M
and Stoianovici D: Robotic transrectal ultrasound guided prostate
biopsy. IEEE Trans Biomed Eng. 66:2527–2537. 2019.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Phee L, Yuen J, Xiao D, Chan CF, Ho H,
Thng CH, Tan PH, Cheng C and Ng WS: Ultrasound guided robotic
biopsy of the prostate. Int J Humanoid Robot. 3:463–483. 2006.
|
|
57
|
Ho HSS, Mohan P, Lim ED, Li DL, Yuen JSP,
Ng WS, Lau WKO and Cheng CWS: Robotic ultrasound-guided prostate
intervention device: System description and results from phantom
studies. Int J Med Robot. 5:51–58. 2009.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Yan J, Pan B and Fu Y: Ultrasound-guided
prostate percutaneous intervention robot system and calibration by
informative particle swarm optimization. Front Mech Eng.
17(3)2022.
|
|
59
|
Raychaudhuri R, Lin DW and Montgomery RB:
Prostate cancer: A review. JAMA. 333:1433–1446. 2025.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Monsky WL, James RC, Unni Krishnan A and
Seslar SP: Remote and telerobotic ultrasound imaging. Methodist
Debakey Cardiovasc J. 21:60–70. 2025.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Dias AB, O'Brien C, Correas JM and Ghai S:
Multiparametric ultrasound and micro-ultrasound in prostate cancer:
A comprehensive review. Br J Radiol. 95(20210633)2022.PubMed/NCBI View Article : Google Scholar
|