Introduction
Moyamoya disease (MMD) is a rare, chronic and
progressive cerebrovascular occlusive disorder of unknown etiology.
MMD is characterized by bilateral stenosis or occlusion of the
terminal portions of the internal carotid arteries and the proximal
anterior and middle cerebral arteries, accompanied by the formation
of a fragile collateral network at the base of the brain, the
so-called ‘moyamoya’ (meaning ‘puff of smoke’ in Japanese) vessels
(1,2). The disease has a bimodal age
distribution, with peaks in childhood and in adults in their fourth
decade, and exhibits a notable predominance in East Asian
populations (3,4). Clinically, MMD manifests primarily as
cerebral ischemia in children and adults <40 years old, whereas
intracranial hemorrhage becomes more common in older adults
(5). In adult patients with the
ischemic phenotype, the persistent hypoperfusion leads to transient
ischemic attacks (TIAs) or cerebral infarctions, resulting in the
accumulation of neurological deficits, cognitive decline and a
notable deterioration in quality of life, posing a substantial
burden on individuals, families and society (6,7).
Currently, there are no effective pharmacological treatments to
halt or reverse the underlying pathological progression of MMD. The
mainstay of management is revascularization surgery, which aims to
augment cerebral blood flow by establishing collateral pathways
from the external carotid circulation (8,9).
Indirect revascularization techniques, such as
encephaloduroarteriosynangiosis (EDAS), are widely employed,
particularly for pediatric patients and in cases where direct
anastomosis is technically challenging (10). This procedure involves placing
vascularized tissues from the scalp or dura onto the ischemic brain
surface, inducing a natural and gradual neovascularization response
over weeks to months. Compared with direct bypass, indirect
revascularization offers advantages including relative technical
simplicity, shorter operative time, lower risk of perioperative
hyperperfusion syndrome, and a more tempered, self-regulating blood
supply restoration (11,12).
Nevertheless, the therapeutic efficacy of indirect
revascularization is entirely dependent on postoperative de
novo collateral formation, as this procedure provides no
immediate blood flow restoration unlike direct bypass. A
substantial number of adult patients fail to develop adequate
collateral circulation, which perpetuates the risk of ischemic
events (13,14). This limitation highlights the
urgent need for adjuvant strategies to enhance surgical
neovascularization, the core determinant of indirect
revascularization efficacy. The pathogenesis of MMD, which involves
inflammatory dysregulation and impaired vascular remodeling,
provides a compelling rationale for the use of atorvastatin, a
3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor
(statin) conventionally indicated for hypercholesterolemia and
atherosclerotic cardio-cerebrovascular disease prevention (15,16).
Beyond its lipid-lowering properties, atorvastatin exhibits
pleiotropic effects, including endothelial protection,
anti-inflammatory action and the promotion of functional
angiogenesis, which are theoretically synergistic with indirect
revascularization (17-20).
Large-scale clinical studies have confirmed that good
neovascularization (Matsushima grade, Good) after indirect
revascularization is an independent predictor of reduced long-term
ischemic event risk in adult MMD (12,14).
Although some evidence has suggested that statins may improve
collateral circulation in ischemic cerebrovascular disease
(21,22), robust prospective evidence for its
specific use in adult patients with MMD undergoing indirect
revascularization is lacking (23). The current prospective, randomized
study selected 6-month angiographic neovascularization grade as the
primary endpoint, with the aim of directly assessing the effects of
atorvastatin on the core determinant of indirect revascularization
efficacy.
Materials and methods
Study design and patient
population
The present single-center, prospective, randomized,
open-label, blinded endpoint study was conducted at the Department
of Neurosurgical Trauma I, Cangzhou Central Hospital (Cangzhou,
China). Consecutive adult patients (aged 18-65 years) who were
diagnosed with ischemic-type MMD and scheduled to undergo indirect
revascularization surgery between March 2023 and August 2024 were
assessed for eligibility.
Inclusion criteria
The diagnosis of MMD was confirmed using digital
subtraction angiography (DSA) based on the diagnostic criteria
established by the Japanese Ministry of Health and Welfare
(24). The inclusion criteria were
as follows: i) Aged between 18 and 65 years. ii) Radiologically
confirmed ischemic-type MMD (presence of TIAs and/or cerebral
infarction). iii) Planned treatment with indirect revascularization
surgery. iv) Ability to provide written informed consent.
Exclusion criteria. Patients were excluded
from the present study based on the following criteria: i)
Hemorrhagic-onset MMD or a history of intracranial hemorrhage. ii)
Presence of notable comorbidities, including severe cardiac,
hepatic or renal insufficiency (defined as alanine aminotransferase
or aspartate aminotransferase levels >3 times the upper limit of
normal, or an estimated glomerular filtration rate <30
ml/min/1.73 m2). iii) Known allergy or contraindication
to statin therapy. iv) Pregnancy or lactation. v) Preoperative
modified Rankin Scale (mRS) score >3(25), indicating severe disability. vi)
Concurrent conditions requiring long-term anticoagulation therapy.
vii) History of statin use within 3 months prior to enrollment.
viii) MMD associated with other conditions such as atherosclerosis,
autoimmune diseases or cranial radiotherapy.
A total of 80 patients who met the inclusion
criteria were finally enrolled and randomly assigned in a 1:1 ratio
to either the control group or the atorvastatin group.
Randomization, blinding and
intervention
Randomization: Eligible patients were randomly
assigned in a 1:1 ratio to either the control group or the
atorvastatin group. The random allocation sequence was generated by
an independent statistician using computer-generated random
numbers. The sequence was concealed using sequentially numbered,
opaque, sealed envelopes. After a patient provided written informed
consent and prior to surgery, the attending neurosurgeon opened the
next envelope in the sequence to reveal the group assignment.
Blinding: Given the nature of the pharmacological intervention,
this was an open-label study for the patients and treating
physicians to ensure practical feasibility and patient safety.
However, a blinded endpoint assessment was strictly implemented to
minimize bias. The neuroradiologists who evaluated the
postoperative DSA results and the clinicians who performed the mRS
assessments at follow-up were completely unaware of the group
assignments of the patients. All imaging data were anonymized
before being presented to the evaluators. Intervention: All
patients underwent indirect revascularization surgery (EDAS)
performed by senior neurosurgeons with extensive experience in
cerebrovascular surgery.
The patients in the control group (n=40) received
standard postoperative medical therapy. This included a single
antiplatelet agent [either aspirin (100 mg, daily) or clopidogrel
(75 mg, daily)] and management of vascular risk factors (for
example, antihypertensive drugs) as clinically indicated.
The patients in the atorvastatin group (n=40)
received identical standard postoperative medical therapy to the
control group, but also received oral atorvastatin calcium tablets
(20 mg, once daily). This dose was selected based on its
well-established safety profile in Chinese patients with adult
cerebrovascular disease, its consistency with domestic clinical
practice guidelines, and due to prior prospective evidence
supporting its pro-angiogenic effect in MMD (26,27).
The atorvastatin administration was initiated on postoperative day
1, provided the condition of the patient was stable, and was
continued for ≥6 months. No dose-escalation regimen, higher-dose
(40 mg/day) group or predefined management protocol for concomitant
potent CYP3A4 inhibitors was included in this trial design, as
enrolled patients excluded those requiring long-term use of such
medications.
Outcome measures and data
collection
The primary outcome of the present study was the
rate of good neovascularization at the 6-month follow-up, assessed
by DSA. Angiographic collateral formation was evaluated according
to the Matsushima grading system (28), which classifies the extent of
revascularization into three categories: Poor, Fair and Good. For
the purpose of the current study, a Matsushima grade of ‘Good’ was
defined as a good outcome, whereas ‘Fair’ and ‘Poor’ were defined
as poor outcomes.
The secondary outcomes were assessed at 6 months
postoperatively and included: i) Functional outcome: An improvement
of ≥1 point on the mRS compared with the preoperative baseline. ii)
Ischemic event recurrence: The recurrence of any ischemic event,
defined as a composite of TIA or new cerebral infarction, confirmed
by clinical assessment and neuroimaging (MRI). Prespecified root
cause analysis was performed for all recorded ischemic events to
explore the association between event occurrence and postoperative
neovascularization grade. iii) Hemodynamic improvement: Changes in
cerebral hemodynamics were evaluated using computed tomography
perfusion (CTP). Parameters of interest included cerebral blood
flow (CBF) and mean transit time (MTT) in the middle cerebral
artery territory of the operated hemisphere. iv) Safety outcomes:
The incidence of all adverse events, with specific attention paid
to statin-related adverse events, such as liver enzyme elevation,
myalgia and creatine kinase (CK) elevation. No patient-reported
Health-Related Quality of Life (HRQoL) data (29), blood-based biomarkers, or
full-cycle medical cost data for cost-effectiveness analysis were
collected as prespecified outcomes in the trial.
Regarding data collection and follow-up, baseline
demographic data, clinical presentation, vascular risk factors,
Suzuki angiographic stage (30)
and preoperative mRS scores were recorded for all patients upon
enrollment. Follow-up assessments were conducted at 6 weeks (via
telephone) and 6 months (via outpatient visit). The 6-week
follow-up primarily documented adherence to medication, adverse
events and the occurrence of TIA. No routine 1-3 month imaging
[including transcranial Doppler (TCD) or interim CTP] was
prescheduled in the trial design. The 6-month follow-up served as
the primary efficacy endpoint, with all patients completing
clinical assessment (mRS), cerebral perfusion imaging (CTP) and DSA
(the reference standard for neovascularization grading). Routine
follow-up reminders were provided for all patients, but no specific
adherence-boosting interventions for asymptomatic/minimally
symptomatic patients were prespecified in this trial.
Regarding endpoint adjudication, to ensure
objectivity, all DSA and CTP images were analyzed by two
independent, experienced neuroradiologists who were blinded to the
clinical data and group assignments. Any discrepancies in the
assessment were resolved by consensus or by a third senior
neuroradiologist. Clinical mRS scores at follow-up were assessed by
a trained neurologist who was also blinded to the treatment
allocation.
Statistical analysis
All statistical analyses were performed using SPSS
(version 26.0; IBM Corp.), with two-sided P<0.05 considered to
indicate a statistically significant difference. Baseline
characteristics were compared using independent samples t-test,
Mann-Whitney U tests or χ2/Fisher's exact tests, as
appropriate. Efficacy analyses followed the intention-to-treat
principle. For binary outcomes (for example, good
neovascularization rate), the risk ratio (RR) with 95% confidence
interval (CI) was calculated and groups were compared using the
χ2 test. For continuous outcomes (such as CBF), the mean
difference (MD) with 95% CI was calculated, and intergroup
comparisons were performed using independent samples t-tests. A
multivariate logistic regression model was used to identify
independent predictors, reporting adjusted odds ratio (aOR) with
95% CI. The consistency of the treatment effect across prespecified
subgroups was explored via subgroup analysis, with formal
interaction tests performed by adding a treatment-by-subgroup
interaction term to the multivariate logistic regression model,
with significance assessed via the Wald test.
Results
Patient characteristics
Between March 2023 and August 2024, a total of 80
eligible adult patients with ischemic-type MMD were enrolled and
randomly allocated to the control group (n=40) or the atorvastatin
group (n=40). All patients received their assigned treatment and
were included in the final analysis. The baseline demographic and
clinical characteristics were well-balanced between the two groups,
as detailed in Table I
 | Table IBaseline characteristics of the
patients (n=80). |
Table I
Baseline characteristics of the
patients (n=80).
| Characteristic | Control group
(n=40) | Atorvastatin group
(n=40) | Test statistic | P-value |
|---|
| Age, years, mean ±
SD | 44.8±11.2 | 46.1±10.5 | t=-0.552 | 0.584 |
| Sex, n (%) | | |
χ2=0.205 | 0.651 |
|
Male | 24 (60.0) | 22 (55.0) | | |
|
Female | 16 (40.0) | 18 (45.0) | | |
| Body mass index,
kg/m2, mean ± SD | 24.5±3.0 | 24.3±2.9 | t=0.473 | 0.642 |
| Clinical
presentation/qualifying event, n (%) | | |
χ2=0.503 | 0.478 |
|
Cerebral
infarction | 25 (62.5) | 28 (70.0) | | |
|
TIA
only | 15 (37.5) | 12 (30.0) | | |
| Preoperative mRS
score, median (IQR) | 2 (1-2) | 2 (1-2) | Z=-0.114 | 0.915 |
| Suzuki stage, n
(%) | | |
χ2=0.063 | 0.969 |
|
Stage
III | 14 (35.0) | 13 (32.5) | | |
|
Stage
IV | 19 (47.5) | 20 (50.0) | | |
|
Stage V | 7 (17.5) | 7 (17.5) | | |
| Involved side, n
(%) | | |
χ2=0.313 | 0.576 |
|
Unilateral | 7 (17.5) | 9 (22.5) | | |
|
Bilateral | 33 (82.5) | 31 (77.5) | | |
| Operated side, n
(%) | | |
χ2=0.201 | 0.654 |
|
Left | | 18 (45.0) | 20 (50.0) | |
|
Right | 22 (55.0) | 20 (50.0) | | |
| Comorbidities, n
(%) | | | | |
|
Hypertension | 19 (47.5) | 18 (45.0) |
χ2=0.050 | 0.823 |
|
Diabetes
mellitus | 11 (27.5) | 9 (22.5) |
χ2=0.052 | 0.819 |
|
Hyperlipidemia | 26 (65.0) | 23 (57.5) |
χ2=0.474 | 0.491 |
| Laboratory values,
mean ± SD | | | | |
|
Total
cholesterol, mmol/l | 4.6±0.9 | 4.5±0.8 | t=0.537 | 0.598 |
|
LDL-C,
mmol/l | 2.8±0.7 | 2.7±0.6 | t=0.706 | 0.485 |
Primary efficacy outcomes
The impact of atorvastatin on the primary efficacy
endpoints at the 6-month follow-up is summarized in Table II. A significantly higher
proportion of patients in the atorvastatin group achieved good
neovascularization, as defined by a Matsushima grade of ‘Good’,
compared with the control group [65.0% (26/40) vs. 37.5% (15/40)].
This difference was statistically significant, with an RR of 1.73
(95% CI, 1.10-2.74; P=0.014). This corresponded to an absolute risk
reduction (ARR) of 27.5% for the primary endpoint of good
neovascularization, with a number needed to treat (NNT) of 4
(calculated as the reciprocal of the ARR: 1/0.275=3.64). Analysis
of the full distribution of Matsushima grades revealed a
significant shift toward better angiographic outcomes in the
atorvastatin group (P=0.031 for trend). Specifically, the
atorvastatin group had nearly double the number of ‘Good’ responses
and fewer ‘Poor’ responses [5.0% (2/40)] compared with in the
control group [17.5% (7/40)]. Notably, all 6 postoperative ischemic
events in the cohort occurred in patients with Poor or Fair
neovascularization, providing risk-based evidence for treatment
escalation decisions in non-responders.
 | Table IIPrimary efficacy outcomes at 6-month
follow-up. |
Table II
Primary efficacy outcomes at 6-month
follow-up.
| Outcome
measure | Control group
(n=40) | Atorvastatin group
(n=40) | Effect size (95%
CI) | P-value |
|---|
| Angiographic
outcomes | | | | |
|
Good
neovascularizationa,
n (%) | 15 (37.5) | 26 (65.0) | RR: 1.73
(1.10-2.74) | 0.014 |
|
Matsushima
grade distribution, n (%) | | | - | 0.031 |
|
Good | 15 (37.5) | 26 (65.0) | | |
|
Fair | 18 (45.0) | 12 (30.0) | | |
|
Poor | 7 (17.5) | 2 (5.0) | | |
| Hemodynamic
outcomes | | | | |
|
Improvement
in CBF, ml/100 g/min, mean ± SD | 8.5±4.2 | 12.8±5.1 | MD: 4.3
(2.2-6.4) | 0.001 |
|
Reduction in
MTT, sec, mean ± SD | -1.2±0.6 | -1.9±0.7 | MD: -0.7 (-0.9 to
-0.5) | 0.001 |
Consistent with the angiographic findings,
postoperative hemodynamic improvements were significantly greater
in the atorvastatin group. The mean improvement in CBF was 12.8±5.1
ml/100 g/min in the atorvastatin group, compared with 8.5±4.2
ml/100 g/min in the control group, resulting in a MD of 4.3 ml/100
g/min (95% CI: 2.2-6.4; P<0.001). Similarly, the reduction in
MTT was more pronounced in the atorvastatin group (-1.9±0.7 seco)
than in the control group (-1.2±0.6 sec), with a MD of -0.7 sec
(95% CI: -0.9 to -0.5; P<0.001).
Secondary clinical and safety
outcomes
Outcomes related to clinical function and event
recurrence are presented in Table
III. An improvement of ≥1 point on the mRS scale was observed
in 29 patients (72.5%) in the atorvastatin group, compared with 20
patients (50.0%) in the control group (RR: 1.45; 95% CI: 1.03-2.04;
P=0.039). The composite endpoint of TIA or stroke recurrence
occurred in 1 patient (2.5%) in the atorvastatin group and 5
patients (12.5%) in the control group; this difference was not
statistically significant (P=0.201). Root cause analysis confirmed
all 6 events occurred in patients with poor neovascularization on
6-month DSA, with statistical analysis limited by the small number
of events.
 | Table IIISecondary clinical and safety
outcomes at 6-month follow-up. |
Table III
Secondary clinical and safety
outcomes at 6-month follow-up.
| Outcome
measure | Control group
(n=40) | Atorvastatin group
(n=40) | Effect size (95%
CI) | P-value |
|---|
| Secondary clinical
outcomes | | | | |
|
mRS
improvement ≥1, n (%) | 20 (50.0) | 29 (72.5) | RR: 1.45
(1.03-2.04) | 0.039 |
|
TIA/stroke
recurrence, n (%) | 5 (12.5) | 1 (2.5) | - | 0.201a |
| Safety
outcomes | | | | |
|
Any adverse
event, n (%) | 17 (42.5) | 22 (55.0) | - | 0.263 |
|
Liver enzyme
elevation, n (%) | 0 (0.0) | 3 (7.5) | - | 0.241a |
|
Myalgia, n
(%) | 1 (2.5) | 2 (5.0) | - | 1.000a |
The frequency of adverse events is also detailed in
Table III. The proportion of
patients experiencing any adverse event was 55.0% in the
atorvastatin group and 42.5% in the control group (P=0.263). Liver
enzyme elevation was observed in 3 patients (7.5%) in the
atorvastatin group and in none of the control group patients
(0.0%); this difference was not statistically significant
(P=0.241). The incidence of myalgia was similar between groups (5.0
vs. 2.5%, P=1.000).
Multivariate analysis of predictors
for good neovascularization
To determine whether atorvastatin treatment was an
independent predictor of good neovascularization after adjusting
for potential confounding variables, a multivariate logistic
regression analysis was performed. The model included treatment
group, age, sex, Suzuki stage and qualifying event type as
covariates (Table IV). After
adjustment for these baseline characteristics, treatment with
atorvastatin remained significantly associated with a higher
likelihood of achieving good neovascularization (aOR: 3.125; 95%
CI: 1.281-7.624; P=0.012). None of the other variables included in
the model, age, sex, Suzuki stage or qualifying event type,
demonstrated a statistically significant association with the
primary outcome (all P>0.05).
 | Table IVMultivariate logistic regression
analysis of predictors for good neovascularization. |
Table IV
Multivariate logistic regression
analysis of predictors for good neovascularization.
| Variable | Adjusted odds
ratio | 95% CI | P-value |
|---|
| Treatment
group | | | |
|
Control | 1.000
(Reference) | | |
|
Atorvastatin | 3.125 | 1.281-7.624 | 0.012 |
| Age | | | |
|
≤45
years | 1.000
(Reference) | | |
|
>45
years | 0.812 | 0.334-2.007 | 0.652 |
| Sex | | | |
|
Female | 1.000
(Reference) | | |
|
Male | 1.291 | 0.522-3.188 | 0.583 |
| Suzuki stage | | | |
|
III-IV | 1.000
(Reference) | | |
|
V | 0.615 | 0.186-2.109 | 0.436 |
| Qualifying
event | | | |
|
TIA
only | 1.000
(Reference) | | |
|
Cerebral
infarction | 0.702 | 0.285-1.763 | 0.448 |
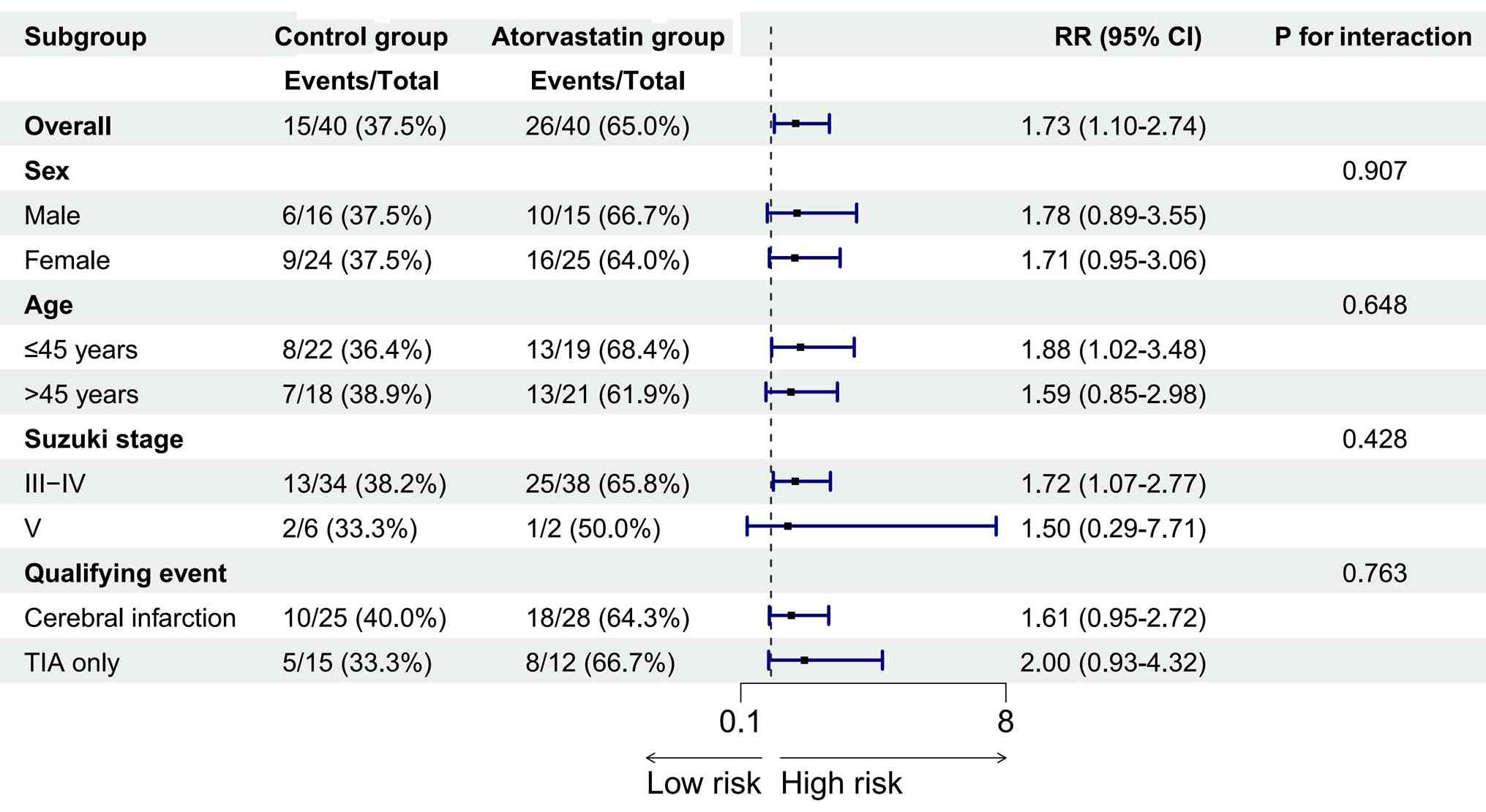
Subgroup analysis
An exploratory subgroup analysis was conducted to
evaluate the consistency of the treatment effect of atorvastatin on
the primary outcome of good neovascularization across key baseline
patient characteristics. The results are presented in Fig. 1. The beneficial effect of
atorvastatin was consistently observed across all predefined
subgroups, including sex, age, Suzuki stage and qualifying event
type. As shown in Fig. 1, all
point estimates of the RR favored the atorvastatin group, with all
values >1 and falling to the right of the line of no effect
(RR=1) in the forest plot. Formal Wald tests for interaction were
not statistically significant for any subgroup (all P-values for
interaction >0.05), indicating that the treatment effect did not
significantly differ based on these baseline characteristics.
Discussion
The present prospective, randomized trial
demonstrates that adjunctive low-dose atorvastatin significantly
enhances the core efficacy determinant of indirect
revascularization in adult patients with ischemic MMD. The
principal finding was a markedly higher rate of good
neovascularization (Matsushima grade, Good) in the atorvastatin
group, which is an established independent predictor of reduced
long-term ischemic event risk, supported by improved cerebral
hemodynamics and functional recovery. The 27.5% absolute increase
in good neovascularization (65.0 vs. 37.5%) and the aOR of 3.125
underscored a substantial treatment effect. The favorable outcomes
observed with atorvastatin may be explained by three evidence-based
mechanisms aligned with ischemic MMD pathogenesis: i)
Endothelial-protective effects to rescue the endothelial
dysfunction driving MMD progression; ii) anti-inflammatory actions
to mitigate the chronic inflammatory dysregulation that inhibits
physiological angiogenesis in MMD; iii) direct pro-angiogenic
effects, including endothelial nitric oxide synthase upregulation
and endothelial progenitor cell mobilization, which augment
collateral formation induced by indirect revascularization
(31,32). The present results are consistent
with those of Wang et al (27), whose prospective study also
identified atorvastatin as an independent predictor of good
collateral formation post-EDAS. However, the current study advances
this evidence by employing a randomized design, which minimizes
confounding bias and provides a higher level of evidence than the
previous observational cohort.
The hemodynamic improvements observed in the present
study, namely, greater increases in CBF and greater reductions in
MTT, provide direct functional physiological evidence corresponding
to the superior angiographic neovascularization in the atorvastatin
group, indicating that the newly formed collaterals are
functionally competent. This functional benefit translated into a
significantly higher proportion of patients achieving functional
independence (mRS improvement ≥1: 72.5 vs. 50.0%). While the
reduction in TIA/stroke recurrence (2.5 vs. 12.5%) was not
statistically significant, likely due to sample size constraints,
the direction of effect is clinically encouraging and warrants
further investigation in larger trials. A notable strength of the
study is its randomized design, which addresses a key limitation of
the earlier prospective trial by Wang et al (27) where treatment allocation was
non-random. Furthermore, the present findings provide key
preliminary clinical evidence supporting the core scientific
hypothesis of the ongoing large-scale, double-blind randomized
controlled trial [whose formal trial protocol was published by Gao
et al in 2023(33)], which
aims to definitively confirm the efficacy of atorvastatin in this
patient population. The positive results of the present study
provide preliminary support for the hypothesis being tested in that
larger trial. The multivariate and subgroup analyses strengthen the
conclusions, confirming atorvastatin as an independent predictor of
good neovascularization and demonstrating a consistent treatment
effect across key patient subgroups. Regarding safety, adjunctive
atorvastatin was well-tolerated. The incidence of transient liver
enzyme elevation (7.5%) was consistent with the known safety
profile of low-dose statins and similar to that reported in
previous studies (27,34,35),
with no serious adverse events leading to drug discontinuation.
For clinical decision-making, this effect size,
combined with a favorable safety profile, supports postoperative
atorvastatin as a recommended adjuvant option for eligible
patients; however, current single-center data are insufficient to
establish it as a universal standard of care. For patients with
Poor or Fair neovascularization (non-responders) at 6 months, we
recommend comprehensive re-evaluation of cerebral hemodynamics and
ischemic risk. Complementary direct bypass surgery may be
considered for those with persistent hypoperfusion or recurrent
ischemic symptoms, although the efficacy of this escalated strategy
was not formally evaluated in the present study. Given the
exclusion of patients with concurrent atherosclerosis or other
cerebrovascular comorbidities in this trial, the current
conclusions cannot be directly extrapolated to patients with mixed
pathologies. For this population, more intensive safety monitoring
(including regular lipid, liver enzyme and CK testing) is
recommended, and further targeted studies are required to confirm
treatment efficacy. For patients achieving Good neovascularization
at 6 months, statin discontinuation may be considered for those
without atherosclerotic risk factors and stable cerebral perfusion,
while long-term maintenance therapy is recommended for patients
with underlying vascular risk factors to sustain endothelial
protection and anti-inflammatory benefits. This recommendation is
based on the established pleiotropic effects of statins, as the
current study did not collect long-term data to compare outcomes
between maintenance and discontinuation. In line with global statin
clinical guidelines, for patients requiring short-term concomitant
use of potent CYP3A4 inhibitors (for example, clarithromycin and
amiodarone), temporary suspension of atorvastatin for the full
duration of inhibitor use is recommended; for those requiring
long-term use, switching to a statin not metabolized by the CYP3A4
pathway may be considered. This recommendation is based on
established drug interaction specifications (36), as the present trial did not enroll
patients requiring such concomitant medications.
Preclinical and clinical studies have confirmed that
VEGF and inflammatory cytokines are key mediators of
neovascularization after indirect revascularization, with potential
value for guiding personalized statin use (37-39);
however, the present trial did not prospectively explore such
blood-based biomarkers. Biomarker-guided treatment stratification
will be a core focus of our subsequent prospective studies. For
primary care settings without routine DSA access, a
guideline-aligned alternative monitoring pathway combining regular
clinical assessment for new ischemic events or neurological
decline, and serial TCD is recommended for preliminary efficacy and
safety screening. Prompt referral is recommended for definitive
angiographic evaluation for patients with abnormal findings;
however, this non-invasive strategy cannot fully replace DSA as the
reference standard for neovascularization grading. For asymptomatic
or minimally symptomatic patients on long-term statin therapy,
guideline-aligned adherence-improving measures are recommended,
including standardized patient education on treatment benefits,
regular follow-up reminders and simplified medication regimens,
although the efficacy of these measures was not formally evaluated
in the current trial. Given the 6-month short-term follow-up and
lack of prespecified full medical cost collection in this
single-center small-sample trial, a robust decision-tree model and
formal Incremental Cost-Effectiveness Ratio estimation cannot be
reliably constructed with the current data. A formal
cost-effectiveness analysis will be conducted as a core secondary
analysis in our upcoming large-scale multi-center confirmatory
trial.
No formal guideline revision recommendations are
proposed at this stage, as the findings of the present
single-center small-sample trial require further validation in
large-scale multi-center prospective studies. The prespecified
primary endpoint of the present trial was 6-month postoperative
efficacy; we aim to initiate 2- to 5-year extended follow-up of
this cohort to assess long-term stroke recurrence and cognitive
outcomes. Mechanistically, preclinical evidence suggests potential
synergistic pro-angiogenic effects between atorvastatin and
collateral-promoting drugs such as cilostazol (40); the current single-agent trial
provides a preliminary clinical basis for subsequent combination
therapy trials. The findings of the present study on the
pro-neovascularization efficacy of statins provide preliminary
human clinical evidence from a treatment response perspective,
supporting the core role of vascular endothelial dysfunction and
inflammation in the pathogenesis of MMD. For the upcoming
multi-center confirmatory trial, we aim to involve patient
representatives in the study design process, to optimize
patient-centric outcome measures. and improve recruitment and
implementation efficiency.
The present study has several limitations that
should be considered. First, as a single-center trial, the patient
population and surgical expertise may reflect a specific
institutional context, which could affect the generalizability of
the results. Second, although the sample size was adequate to
detect a significant difference in the primary angiographic
endpoint, it was relatively modest, which limited statistical power
for subgroup analyses and precludes definitive conclusions
regarding the clinical efficacy of atorvastatin, especially for
less frequent endpoints such as ischemic event recurrence, as
reflected by the wide CIs in some subgroup analyses. Third,
clinical or imaging data were not collected at 1-3 months
postoperatively, which prevented the identification of early
predictors of final neovascularization outcomes; such data are
critical for dynamic, individualized treatment adjustment and will
be a core focus of subsequent research. Fourth, the 6-month
follow-up period was sufficient to evaluate angiographic maturation
as the primary outcome, but is too short to assess the long-term
stability of newly formed collateral vessels or the sustained
impact on preventing lifelong disability or fatal stroke. Fifth,
while randomization ensured baseline comparability and multivariate
regression adjusted for known confounders, residual confounding
from unmeasured factors, such as subtle differences in intrinsic
angiogenic potential, cannot be entirely excluded. Sixth, the
strict exclusion of patients with MMD related to atherosclerosis or
other concurrent cerebrovascular diseases restricts the
generalizability of the findings to patients with mixed
pathological mechanisms. Seventh, functional recovery was evaluated
only using the physician-assessed mRS, without collection of
patient-reported HRQoL data, limiting the comprehensive assessment
of the overall clinical value of the treatment. HRQoL assessment
will be included as a core secondary endpoint in our future
large-scale confirmatory trials. Eighth, the present trial only
investigated the efficacy and safety of a fixed 20 mg/day
atorvastatin regimen, with no prespecified dose-escalation design
or exploration of higher-dose (40 mg/day) therapy. The preplanned
subgroup analyses only evaluated the consistency of the 20 mg/day
effect across baseline subgroups and cannot support a potential
greater benefit of 40 mg/day in specific populations (for example,
younger patients without liver disease); dose optimization across
subgroups will be a key focus of our future prospective studies.
Ninth, the study was designed with only a 6-month treatment course
and follow-up endpoint, without predefined long-term evaluation of
statin maintenance versus discontinuation after achieving good
neovascularization. Long-term follow-up to clarify optimal
treatment duration and sustained clinical benefits will be a
central aim of our subsequent research.
In conclusion, the present single-center prospective
randomized trial provides preliminary evidence that adjunctive
atorvastatin 20 mg/day is associated with higher good
neovascularization rate, improved cerebral hemodynamics and better
functional recovery at 6 months after indirect revascularization in
adult ischemic MMD, with a manageable safety profile. Further
large-scale multi-center studies are warranted to validate these
findings and to determine the definitive clinical role of
atorvastatin for this patient population.
Acknowledgements
Not applicable.
Funding
Funding: This manuscript was funded by the Self-funded Project
under the Key Research and Development Program of Cangzhou City
(grant no. 222106093).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
DW conceptualized and designed the study, developed
the study methodology, including the randomization and endpoint
assessment protocols, performed the clinical investigation, led
data curation, including prospective data collection and quality
control, drafted the original manuscript and administered the
project. JD performed the formal statistical analysis, created all
data visualizations, and critically reviewed and edited the
manuscript. SL participated in the clinical investigation and
patient follow-up, and provided essential study resources,
including clinical site and operating platform support. YL
participated in the clinical investigation and patient recruitment,
and provided essential study resources, including neuroimaging data
acquisition and technical support. XX contributed to the study
methodology, including the statistical analysis plan, and performed
validation of all endpoint data and statistical results via
independent blinded review. LH participated in the clinical
investigation, and assisted with data curation, including
standardized data entry and source verification against original
medical records. QP made substantial contributions to the
conception and design of the study, led the interpretation of core
study results, secured funding for the research, provided critical
supervision for all study procedures, critically revised the
manuscript for important intellectual content, and oversaw final
approval of the manuscript for publication. DW and QP confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript, and agree to be accountable for all
aspects of the work in full accordance with ICMJE authorship
guidelines.
Ethics approval and consent to
participate
The current study was performed in accordance with
The Declaration of Helsinki and was approved by the local ethics
committee of the Cangzhou Central Hospital [approval no.
2023-235-01(z); Cangzhou, China]. Each patient provided written
informed consent for participation.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ihara M, Yamamoto Y, Hattori Y, Clausen T,
Bang OY, Steinberg GK, Tournier-Lasserve E and Koizumi A: Moyamoya
disease: Diagnosis and interventions. Lancet Neurol. 21:747–758.
2022.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gonzalez NR, Amin-Hanjani S, Bang OY,
Coffey C, Du R, Fierstra J, Fraser JF, Kuroda S, Tietjen GE and
Yaghi S: American Heart Association Stroke Council; Council on
Cardiovascular and Stroke Nursing; and Council on Clinical
Cardiology. Adult moyamoya disease and syndrome: Current
perspectives and future directions: A scientific statement from the
American Heart Association/American stroke association. Stroke.
54:e465–e479. 2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Asselman C, Hemelsoet D, Eggermont D,
Dermaut B and Impens F: Moyamoya disease emerging as an
immune-related angiopathy. Trends Mol Med. 28:939–950.
2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Fujimura M, Bang OY and Kim JS: Moyamoya
disease. Front Neurol Neurosci. 40:204–220. 2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Chen T, Wei W, Yu J, Xu S, Zhang J, Li X
and Chen J: The progression of pathophysiology of moyamoya disease.
Neurosurgery. 93:502–509. 2023.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Cheng X, Cao Y, Duan J, Zhou M, Ye S and
Zhu Y: Moyamoya disease: Epidemiology, clinical features,
pathogenesis, diagnosis and therapeutic interventions. Mol Biomed.
6(76)2025.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kaur S, Parikh P, Khatri J and Jaber W:
Coronary vasculopathy due to moyamoya disease. J Nucl Cardiol.
30:2229–2232. 2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
He S, Zhou Z, Cheng MY, Hao X, Chiang T,
Wang Y, Zhang J, Wang X, Ye X, Wang R, et al: Advances in moyamoya
disease: Pathogenesis, diagnosis, and therapeutic interventions.
MedComm (2020). 6(e70054)2025.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Shang S, Zhou D, Ya J, Li S, Yang Q, Ding
Y, Ji X and Meng R: Progress in moyamoya disease. Neurosurg Rev.
43:371–382. 2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Fiaschi P, Scala M, Piatelli G, Tortora D,
Secci F, Cama A and Pavanello M: Limits and pitfalls of indirect
revascularization in moyamoya disease and syndrome. Neurosurg Rev.
44:1877–1887. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yang RM, Hao FB, Zhao B, Zhang Q, Yu D,
Zou ZX, Gao G, Guo QB, Shen XX, Fu HG, et al: Natural course and
risk factors of moyamoya disease with unruptured intracranial
aneurysm. Front Neurol. 14(1115909)2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Li JJ, Wang XP, Wang QN, Bao XY, Guo QB,
Kong ZQ, Gao G, Wang MJ, Liu SM, Fu HG, et al: Long-term outcomes
after conservative and EDAS treatment for 111 elderly patients with
moyamoya disease: Longitudinal and cross-sectional study. J
Neurosurg. 140:800–808. 2023.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Karunanithi K, Han C, Lee CJ, Shi W, Duan
L and Qian Y: Identification of a hemodynamic parameter for
assessing treatment outcome of EDAS in Moyamoya disease. J Biomech.
48:304–309. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bao XY, Wang QN, Wang XP, Yang RM, Zou ZX,
Zhang Q, Li DS and Duan L: Recognition of the effect of indirect
revascularization for moyamoya disease: The balance between the
stage progression and neoangiogenesis. Front Neurol.
13(861187)2022.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Liu JJ and Steinberg GK: Direct versus
indirect bypass for moyamoya disease. Neurosurg Clin N Am.
28:361–374. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kolb B, Fadel H, Rajah G, Saber H, Luqman
A and Rangel-Castilla L: Effect of revascularization on cognitive
outcomes in intracranial steno-occlusive disease: A systematic
review. Neurosurg Focus. 46(E14)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Khokhar SA, Rehman RM and Masood S:
Comparison of efficiency between Rosuvastatin and Atorvastatin in
reducing low-density lipoprotein (LDL-C) in patients with diabetes
mellitus. J Pak Med Assoc. 72:2288–2290. 2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Ryan TE, Torres MJ, Lin CT, Clark AH,
Brophy PM, Smith CA, Smith CD, Morris EM, Thyfault JP and Neufer
PD: High-dose atorvastatin therapy progressively decreases skeletal
muscle mitochondrial respiratory capacity in humans. JCI Insight.
9(e174125)2024.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ki YJ, Kim W, Lee KH, Han SJ, Kim YH, Doh
JH, Kim TN, Chung CH, Kim DY, Cho JM, et al: Lipid-lowering effect
and safety of ezetimibe and atorvastatin 5 mg in patients with
primary hypercholesterolemia or mixed dyslipidemia: A randomized,
double-blind, parallel, multicenter, phase 3 clinical trial. Clin
Cardiol. 48(e70138)2025.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Al-Qaaneh AM, Obaid WT, Al-Mohammadi OS,
Al-Qaaneh AM, Rabaan AA and Mustafa SM: Dose-dependent atorvastatin
associated with angioedema. Int J Clin Pharmacol Ther. 60:106–110.
2022.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Zhu J, Ma M, Fang J, Bao J, Dong S, Chen
N, Guo Y and He L: Prestroke statin use enhances collateralization
in acute ischemic stroke patients. Restor Neurol Neurosci.
38:311–321. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Miura S and Saku K: Regulation of
angiogenesis and angiogenic factors by cardiovascular medications.
Curr Pharm Des. 13:2113–2117. 2007.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Williams F, Martin K, Scott TR, Clark D,
Amell MF, Spratt NJ, Beard DJ and Coupland KG: Effects of
simvastatin treatment on leptomeningeal collateral vessels:
Resistance, number and diameter. Vascul Pharmacol.
160(107518)2025.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kuroda S and Houkin K: Moyamoya disease:
Current concepts and future perspectives. Lancet Neurol.
7:1056–1066. 2008.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Birkeland P, Hansen V, Tharmabalan V,
Lauritsen J, Nielsen T, Truelsen T, Rosenbaum S and von
Weitzel-Mudersbach P: Long-Term outcome of moyamoya disease. Acta
Neurochir Suppl. 136:93–97. 2025.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Liu L, Li Z, Zhou H, Duan W, Huo X, Xu W,
Li S, Nie X, Liu H, Liu J, et al: Chinese Stroke Association
guidelines for clinical management of ischaemic cerebrovascular
diseases: Executive summary and 2023 update. Stroke Vasc Neurol.
6(e3)2023.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wang QN, Bao XY, Zou ZX, Wang XP, Zhang Q,
Li DS, Zhao YQ and Duan L: The role of atorvastatin in collateral
circulation formation induced by encephaloduroarteriosynangiosis: A
prospective trial. Neurosurg Focus. 51(E9)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Li W, Zhao M, Liu X, Wang P, Zhu H, Zhang
Q, Zhu C, Zhang Q, Zhao J and Zhang Y: A novel system for
evaluating collateralization of the external carotid artery after
cerebral revascularization in moyamoya disease. J Neurosurg.
140:1707–1715. 2023.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Bersano A, Khan N, Fuentes B, Acerbi F,
Canavero I, Tournier-Lasserve E, Vajcoczy P, Zedde ML, Hussain S,
Lémeret S, et al: European stroke organisation (ESO) guidelines on
moyamoya angiopathy endorsed by vascular european reference network
(VASCERN). Eur Stroke J. 1:55–84. 2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Yamamoto S, Kashiwazaki D, Akioka N,
Kuwayama N, Noguchi K and Kuroda S: Progressive shrinkage of
involved arteries in parallel with disease progression in moyamoya
disease. World Neurosurg. 122:e253–e261. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zinellu A and Mangoni AA: An updated
systematic review and meta-analysis of the effect of statins on
asymmetric dimethylarginine. Nitric Oxide. 120:26–37.
2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kronborg TM, Schierwagen R, Trošt K, Gao
Q, Moritz T, Bendtsen F, Gantzel RH, Andersen ML, Teisner AS,
Grønbæk H, et al: Atorvastatin for patients with cirrhosis. A
randomized, placebo-controlled trial. Hepatol Commun.
7(e0332)2023.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Gao G, Wang QN, Hao FB, Wang XP, Liu SM,
Wang MJ, Han C, Bao XY and Duan L: To explore the effectiveness of
atorvastatin in the postoperative formation of collateral blood
vessels after encephaloduroarteriosynangiosis in patients with
moyamoya disease: A prospective double-blind randomized controlled
study. Front Neurol. 14(1169253)2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Zhou S, Chen R, Liu J, Guo Z, Su L, Li Y,
Zhang X, Luo F, Gao Q, Lin Y, et al: comparative effectiveness and
safety of atorvastatin versus rosuvastatin : A multi-database
cohort study. Ann Intern Med. 12:1641–1651. 2024.PubMed/NCBI View
Article : Google Scholar
|
|
35
|
Zhao J, Li X, Xia X, Tian X, Xu Q, Zhang
X, Tian R, Meng X and Wang A: Comparative effectiveness and safety
of atorvastatin versus rosuvastatin among patients with ischemic
stroke or transient ischemic attack: A national registry-based
observational study. EClinicalMedicine. 86(103381)2025.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Denisov IG, Baylon JL, Grinkova YV,
Tajkhorshid E and Sligar SG: Drug-Drug interactions between
atorvastatin and dronedarone mediated by monomeric CYP3A4.
Biochemistry. 5:805–816. 2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Araújo FA, Rocha MA, Mendes JB and Andrade
SP: Atorvastatin inhibits inflammatory angiogenesis in mice through
down regulation of VEGF, TNF-alpha and TGF-beta1. Biomed
Pharmacother. 64:29–34. 2010.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Tian Y, Zhang F, Qiu Y, Wang S, Li F, Zhao
J, Pan C, Tao Y, Yu D and Wei W: Reduction of choroidal
neovascularization via cleavable VEGF antibodies conjugated to
exosomes derived from regulatory T cells. Nat Biomed Eng.
5:968–982. 2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Awad IA, Alcazar-Felix RJ, Stadnik A,
Kinkade S, Jhaveri A, Lee J, Hage S, Iqbal J, Polster SP, Shenkar
R, et al: Safety and efficacy of atorvastatin for rebleeding in
cerebral cavernous malformations (AT CASH EPOC): A phase 1/2a,
randomised placebo-controlled trial. Lancet Neurol. 24:295–304.
2025.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Chiu HW, Shih CC, Huang HJ, Huang TH and
Sung LC: Cilostazol attenuates vascular inflammation via the
regulation of TICAM1/IRF3 signaling pathway. Biomed Pharmacother.
189(118275)2025.PubMed/NCBI View Article : Google Scholar
|