Introduction
Retinopathy of prematurity (ROP) is a proliferative
retinal vascular disorder that predominantly affects premature
infants, representing one of the leading preventable causes of
childhood blindness worldwide (1,2). ROP
arises from abnormal retinal vascularization in preterm neonates,
where the incompletely developed retinal vasculature at birth
renders them particularly vulnerable to various pathophysiological
insults (2,3).
The epidemiological relevance of ROP has intensified
in parallel with advances in neonatal intensive care medicine.
While marked improvements in perinatal and neonatal care have
notably reduced mortality rates among premature infants, these same
advances have led to increased survival rates of extremely low
birth weight and very low gestational age infants, populations at
the highest risk for ROP development (4,5);
this has resulted in an increased prevalence of ROP. ROP remains a
leading cause of preventable childhood blindness worldwide, and can
lead to irreversible blindness if severe and untreated. China
accounts for 4,000 of these cases, which represents 1.9% of the
prevalence of blindness among children <5 years old (6).
The clinical spectrum of ROP encompasses a broad
range of severity, from mild self-resolving forms to severe
progressive disease requiring immediate intervention. Severe ROP,
characterized by advanced retinal pathology including
neovascularization, vitreoretinal proliferation and potential
retinal detachment, poses particularly notable challenges for both
neonatologists and ophthalmologists (7). These cases are associated with
substantial treatment complexity, marked healthcare costs and poor
visual prognosis despite optimal therapeutic interventions. The
irreversible nature of severe ROP-related vision loss underscores
the critical importance of early identification, risk
stratification and timely therapeutic intervention (7).
Current screening protocols for ROP rely heavily on
established demographic risk factors, primarily gestational age
(<32 weeks) and birth weight (<1,500 g), which serve as
fundamental parameters for determining screening eligibility and
frequency (8). However, the
pathogenesis of ROP is multifactorial, involving complex
interactions between genetic predisposition, environmental factors,
systemic inflammatory responses and various perinatal and neonatal
complications (9,10). Research has increasingly focused on
identifying additional biomarkers and risk factors that may enhance
the ability to predict severe ROP development beyond traditional
parameters (11).
Understanding the comprehensive risk factor profile
associated with severe ROP development is key for optimizing
clinical management strategies, enabling more precise risk
stratification and facilitating early intervention in high-risk
populations (10,12). Such insights could lead to the
development of more sophisticated predictive models that complement
existing screening protocols and potentially improve long-term
visual outcomes in severe ROP.
Therefore, the present retrospective case-control
study aimed to systematically investigate the associations between
multiple clinical parameters, including general demographic
characteristics, perinatal complications, hematological indices,
biochemical markers and maternal factors during pregnancy, and the
development of severe ROP in premature infants. By identifying
novel risk factors and validating known predictors, the present
study aimed to provide new perspectives on the pathogenesis of
severe ROP, and contribute to the development of more comprehensive
risk assessment strategies for early identification and prevention
of severe ROP in clinical practice.
Materials and methods
Study design and population
The present retrospective case-control study was
conducted at Hebei General Hospital (Shijiazhuang, China),
analyzing clinical data from preterm infants admitted to the
Neonatal and Ophthalmology Departments between September 2018 and
July 2023. All enrolled infants underwent comprehensive fundus
examination for ROP screening according to established protocols
(13).
The present study was conducted in accordance with
The Declaration of Helsinki and approved by the Institutional
Review Board of Hebei General Hospital [approval no. 115 (2024)].
Given the retrospective nature of the study utilizing existing
medical records, informed consent requirements were waived by the
ethics committee, with appropriate measures taken to ensure patient
confidentiality and data protection.
Participants. Inclusion criteria
Case group (severe ROP): Preterm infants with birth
weight <3,000 g, gestational age <37 weeks and diagnosis of
severe ROP confirmed by fundus screening. Control group (mild ROP):
Preterm infants with birth weight <3,000 g, gestational age
<37 weeks and diagnosis of mild ROP confirmed by fundus
screening.
Control infants were restricted to those who
developed any stage of ROP that subsequently resolved without
treatment; neonates who completed screening but never developed ROP
were excluded. The present design intentionally compared two ends
of the ROP spectrum (mild vs. severe) rather than ‘ROP vs. no-ROP’,
allowing for the identification of factors that drive progression
rather than initial disease occurrence. Consequently, the inclusion
of relatively more mature neonates (32-36 weeks gestational age) in
the mild ROP group may have attenuated the observed effect sizes of
classical risk factors; this limitation is addressed in the
discussion.
Exclusion criteria. Infants were excluded if
they had: i) Gestational age ≥37 weeks or birth weight ≥3,000 g;
ii) incomplete clinical data or medical records; iii) inability to
ensure adequate follow-up until retinal vascularization reached
zone III in both eyes without pre-threshold or threshold lesions;
iv) systemic diseases markedly affecting hematological and
biochemical parameters (such as hematological disorders, genetic
and metabolic diseases); and v) concurrent ocular diseases or
systemic conditions that could independently cause ocular
pathology.
ROP diagnosis and classification
ROP diagnosis and classification were performed
according to the International Classification of Retinopathy of
Prematurity, Third Edition criteria (13). Based on fundus screening results
and clinical course, participants were stratified into two groups:
Mild ROP group included infants diagnosed with ROP during screening
that did not meet treatment criteria and subsequently resolved
spontaneously during follow-up; severe ROP group included infants
requiring therapeutic intervention [retinal laser photocoagulation
or intravitreal anti-vascular endothelial growth factor (VEGF)
injection]. Severe ROP includes type I threshold lesions and
rapidly progressive ROP, whereas other types of lesions were
considered mild ROP (13).
Fundus examination
All examinations were performed by experienced
ophthalmologists using the RetCam 3 digital imaging system (Clarity
Medical Systems). Initial examination was conducted at 4-6 weeks
postnatal age or 31-32 weeks corrected gestational age, whichever
occurred later. Subsequent follow-up intervals were determined
based on initial findings and disease progression, continuing until
complete retinal vascularization to zone III or 45 weeks corrected
gestational age without threshold lesions. Pupil dilation was
achieved using compound tropicamide eye drops instilled bilaterally
at 5-10 min intervals, repeated ≥4 times until maximum pupillary
dilation was achieved; topical anesthesia was provided with
proparacaine hydrochloride eye drops. Following appropriate infant
restraint and head stabilization, sterile pediatric eyelid specula
were used to maintain adequate eyelid separation. Gatifloxacin
ophthalmic gel was applied to the eyelid margins, and comprehensive
digital fundus photography was performed using RetCam 3 system to
document retinal findings.
Overall, 94 of 102 eligible infants (92.2%)
completed follow-up until zone III vascularisation or 45 weeks
corrected age; 22 infants were excluded for insufficient
documentation and 80 were included in the final analysis.
Data collection
Comprehensive clinical data were systematically
collected for each participant. Neonatal characteristics included
sex, gestational age, birth weight, mode of delivery and 1-min
Apgar score. Perinatal complications assessed included neonatal
pneumonia, neonatal bronchopulmonary dysplasia (NBPD), neonatal
sepsis, neonatal respiratory distress syndrome (NRDS) and
requirement for blood transfusion.
Clinical laboratory data, including routine blood
test results, were also collected. Hematological and biochemical
analyses were performed at two time points: Within 24 h of birth
and at 1-week postnatal age. Complete blood count included white
blood cell count (WBC; x109/l), neutrophil count (N;
x109/l), lymphocyte count (L; x109/l),
monocyte count (M; x109/l), platelet count (PLT;
x109/l), mean platelet volume (MPV; fl) and hemoglobin
(Hb; g/l). Biochemical profile included total cholesterol (TC),
triglycerides (TG), low-density lipoprotein (LDL), apolipoprotein
(Apo)B, high-density lipoprotein (HDL) and ApoA1. Based on
laboratory values, the following inflammatory indices were
calculated: Neutrophil-to-lymphocyte ratio (NLR),
lymphocyte-to-monocyte ratio (LMR), platelet-to-lymphocyte ratio
(PLR) and systemic immune-inflammation index (SII=platelet count ×
Neutrophil count/Lymphocyte count).
Maternal clinical characteristics during the third
trimester were assessed, including gestational diabetes mellitus,
gestational hypertension and TC, TG, LDL, ApoB, HDL and ApoA1.
Statistical analysis
Statistical analyses were conducted using SPSS
version 24.0 (IBM Corp.). Normally distributed data are presented
as the mean ± SD and differences between two groups were analyzed
using a two-tailed unpaired Student's t-test. Non-normally
distributed data are presented as the median with interquartile
range [M (Q1, Q3)] and were compared using
the Mann-Whitney U test. Categorical data are presented as
frequencies and percentages [n (%)] and were compared using
χ2 test. Comparison of neonatal sepsis and blood
transfusion was performed with Fisher's exact test. Variables
demonstrating statistical significance (P<0.05) in univariate
analyses were included in multivariate binary logistic regression
models to identify independent risk factors for severe ROP
development. To avoid multicollinearity, pairwise Pearson
correlation and variance inflation factor (VIF) were examined
between continuous variables; when VIF ≥5 or r ≥0.7, the variable
with weaker clinical utility was excluded before the final model
was fitted. Receiver operating characteristic (ROC) curves were
constructed for independent risk factors to evaluate their
predictive capacity for severe ROP. The area under the curve (AUC)
with 95% confidence intervals (CIs) was calculated. Optimal cut-off
values were determined using Youden's index, with corresponding
sensitivity and specificity values calculated. P<0.05 was
considered to indicate a statistically significant difference.
Results
Study population characteristics
The present retrospective case-control study
included 80 preterm infants who were admitted to the Neonatal and
Ophthalmology Departments of Hebei General Hospital between
September 2018 and July 2023, and underwent comprehensive fundus
examination for ROP screening. Based on fundus screening results
and clinical course, participants were stratified into two groups:
42 infants in the mild ROP group and 38 infants in the severe ROP
group.
Demographic and clinical
characteristics
Comparative analysis of demographic and clinical
parameters between the mild and severe ROP groups revealed several
significant differences (Table I).
Infants with severe ROP demonstrated significantly lower
gestational age (median 29.00 vs. 32.00 weeks; Z=-4.886;
P<0.001) and birth weight (median 1,137.5 vs. 1,645.0 g;
Z=-4.972; P<0.001) compared with those with mild ROP, and the
1-min Apgar score was significantly lower in the severe ROP group
(median 9 vs. 10; Z=-3.512; P<0.001).
 | Table IComparison of general data in preterm
infants with different severity of ROP. |
Table I
Comparison of general data in preterm
infants with different severity of ROP.
| Parameter | Mild ROP group,
n=42 | Severe ROP group,
n=38 | Statistical
quantity | P-value |
|---|
| Sex (%) | | |
χ2=0.005 | 0.946 |
|
Male | 24 (57.14) | 22 (57.89) | | |
|
Female | 18 (42.86) | 16 (42.11) | | |
| GA, weeks | 32.00 (4.25) | 29.00 (2.75) | Z=-4.886 | <0.001 |
| BW, g | 1,645.00
(804.25) | 1,137.50
(497.50) | Z=-4.972 | <0.001 |
| Mode of
delivery | | |
χ2=2.759 | 0.097 |
|
Caesarean
section (%) | 26 (61.90) | 30 (78.95) | | |
|
Natural
birth,n (%) | 16 (38.10) | 8 (21.05) | | |
| Gestational
diabetes, n (%) | 8 (19.05) | 9 (23.68) |
χ2=0.346 | 0.556 |
| Gestational
hypertension, n (%) | 9 (21.43) | 13 (34.21) |
χ2=0.529 | 0.467 |
| Lipid parameters in
the third trimester of pregnancy | | | | |
|
TC,
mmol/l | 6.34 (1.17) | 6.15 (1.07) | Z=-1.053 | 0.292 |
|
TG,
mmol/l | 3.28 (1.22) | 3.09 (0.64) | Z=-0.416 | 0.678 |
|
LDL,
mmol/l | 3.37 (0.60) | 3.60 (0.70) | Z=-1.517 | 0.129 |
|
ApoB,
g/l | 1.14 (0.13) | 1.04 (0.27) | Z=-1.779 | 0.058 |
|
HDL,
mmol/l | 1.81 (0.33) | 1.65 (0.31) | Z=-1.759 | 0.079 |
|
ApoA1,
g/l | 1.93 (0.32) | 1.89 (0.47) | Z=-0.846 | 0.398 |
| 1-min Apgar score,
points | 10 (0) | 9(3) | Z=-3.512 | <0.001 |
| Neonatal pneumonia
(%) | 7 (16.67) | 13 (34.21) |
χ2=1.671 | 0.196 |
| NBPD, n (%) | 11 (26.19) | 32 (84.21) |
χ2=27.015 | <0.001 |
| Neonatal sepsis, n
(%) | 5 (11.90) | 14 (36.84) |
χ2=6.851 | 0.009 |
| NRDS, n (%) | 15 (35.71) | 26 (68.42) |
χ2=8.542 | 0.003 |
| Blood transfusion,
n (%) | 18 (42.86) | 37 (97.37) |
χ2=15.402 | <0.001 |
Regarding perinatal complications, significant
differences were observed between groups; NBPD was significantly
more prevalent in the severe ROP group compared with in the mild
ROP group (84.21 vs. 26.19%; χ²=27.015; P<0.001). Similarly,
NRDS occurred more frequently in the severe ROP group (68.42 vs.
35.71%; χ²=8.542; P=0.003), as did neonatal sepsis (36.84 vs.
11.90%; χ²=6.851; P=0.009). Blood transfusion requirements were
also significantly higher in the severe ROP group (97.37 vs.
42.86%; χ²=15.402; P<0.001) compared with in the mild ROP
group.
No statistically significant differences were
observed between groups regarding sex distribution, mode of
delivery, maternal gestational diabetes, maternal gestational
hypertension or neonatal pneumonia incidence.
Maternal third-trimester lipid
parameters
Analysis of maternal lipid profiles during the third
trimester revealed no significant differences between groups for
any measured parameters, including TC, TG, LDL, ApoB, HDL and ApoA1
(all P>0.05; Table I).
Hematological parameters within 24 h of
birth. Comparison of hematological parameters obtained within
24 h of birth demonstrated selective differences between groups
(Table II). For example, the L
was significantly higher in the severe ROP group compared with that
in the mild ROP group (median 4.59 vs. 3.90x109/l;
Z=-2.144; P=0.032). In addition, the LMR was significantly lower in
the severe ROP group (median 6.17 vs. 8.21; Z=-2.441; P=0.015).
Other hematological parameters including WBC, N, PLT, MPV, Hb, NLR,
PLR and SII showed no statistically significant differences between
groups at this time point (all P>0.05).
 | Table IIComparison of hematological
parameters within 24 h of birth in preterm infants with ROP of
different severities. |
Table II
Comparison of hematological
parameters within 24 h of birth in preterm infants with ROP of
different severities.
| Blood
parameter | Mild ROP group,
n=42 | Severe ROP group,
n=38 | Z statistic | P-value |
|---|
| WBC,
x109/l | 9.38 (5.71) | 9.10 (5.97) | -8.029 | 0.407 |
| N,
x109/l | 3.34 (3.67) | 3.47 (6.66) | -1.060 | 0.916 |
| L,
x109/l | 3.90 (2.83) | 4.59 (2.98) | -2.144 | 0.032 |
| PLT,
x109/l | 222.5 (83.00) | 249.0 (79.80) | -1.595 | 0.110 |
| MPV, fl | 10.35 (1.125) | 10.10 (1.125) | -1.124 | 0.260 |
| Hb, g/l | 167.50 (24.00) | 164.50 (27.50) | -0.328 | 0.261 |
| NLR | 0.71 (0.76) | 0.91 (2.78) | -1.098 | 0.272 |
| LMR | 8.21 (5.21) | 6.17 (5.42) | -2.441 | 0.015 |
| PLR | 56.00±23.56 | 58.62 (47.44) | -1.252 | 0.210 |
| SII | 172.40
(181.34) | 206.10
(519.10) | -0.723 | 0.470 |
Hematological parameters at 1 week of
age
Analysis of hematological parameters obtained at 1
week postnatal age revealed additional significant differences
(Table III); the MPV was
significantly lower in the severe ROP group compared with that in
the mild ROP group (median 11.46 vs. 11.87 fl; Z=-1.988; P=0.047)
and the LMR remained significantly lower in the severe ROP group
(median 2.25 vs. 3.50; Z=-2.582; P=0.010).
 | Table IIIComparison of hematological
parameters at 1 week of life in preterm infants with ROP of
different severities. |
Table III
Comparison of hematological
parameters at 1 week of life in preterm infants with ROP of
different severities.
| Blood
parameter | Mild ROP group,
n=42 | Severe ROP group,
n=38 | Z statistic | P-value |
|---|
| WBC,
x109/l | 8.80 (4.81) | 9.55 (4.73) | -1.147 | 0.252 |
| N,
x109/l | 4.06 (2.78) | 6.49 (5.66) | -1.325 | 0.185 |
| L,
x109/l | 200.00
(159.25) | 225.50
(120.25) | -1.233 | 0.217 |
| PLT,
x109/l | 3.00 (1.92) | 3.63 (1.83) | -1.113 | 0.266 |
| MPV, fl) | 11.87 (1.20) | 11.46 (0.93) | -1.988 | 0.047 |
| Hb, g/l | 136.50 (29.45) | 139.62 (28.75) | -0.892 | 0.373 |
| NLR | 1.26 (0.57) | 1.78 (1.56) | -1.310 | 0.190 |
| LMR | 3.50 (1.34) | 2.25 (2.10) | -2.582 | 0.010 |
| PLR | 66.61 (36.91) | 62.76 (34.89) | -0.366 | 0.714 |
| SII | 292.74
(260.64) | 344.22
(400.67) | -0.318 | 0.751 |
| TC, mmol/l | 3.00 (0.85) | 2.99 (0.91) | -0.337 | 0.736 |
| TG, mmol/l | 0.68 (0.55) | 0.70 (0.74) | -0.458 | 0.647 |
| LDL, mmol/l | 2.06 (0.59) | 1.84 (0.96) | -0.790 | 0.429 |
| ApoB, g/l | 0.45 (0.12) | 0.46 (0.22) | -0.762 | 0.446 |
| HDL, mmol/l | 0.84 (0.30) | 0.84 (0.29) | -1.571 | 0.116 |
| ApoA1, g/l | 0.77 (0.22) | 0.88 (0.31) | -1.688 | 0.091 |
Biochemical parameters measured at 1 week, including
lipid profiles (TC, TG, LDL, ApoB, HDL, ApoA1), did not demonstrate
significant differences between groups (all P>0.05). Similarly,
other hematological parameters and inflammatory indices (WBC, N,
PLT, Hb, NLR, PLR and SII) showed no significant differences at
this time point (all P>0.05).
Multivariate analysis of risk
factors
To identify independent risk factors for severe ROP
development, multivariate binary logistic regression analysis was
performed using all variables that demonstrated statistical
significance (P<0.05) in univariate analyses as independent
variables, with ROP severity as the dependent variable (Table IV). Neonatal pneumonia was also
included due to its clinical relevance, despite being
non-significant in the univariate analysis.
 | Table IVUnivariate and multivariate logistic
regression analysis of retinopathy of prematurity severity. |
Table IV
Univariate and multivariate logistic
regression analysis of retinopathy of prematurity severity.
| | Univariate
analysis | Multivariate
analysis | |
|---|
| Variable | Category/unit | OR (95% CI) | P-value | Regression
coefficient | Standard error | Wald | df | OR (95% CI) | P-value |
|---|
| Gestational
age | Per 1 week
increase | 0.52
(0.40-0.68) | <0.001 | -0.545 | 0.131 | 17.264 | 1 | 0.580
(0.4-0.7) | <0.001 |
| Birth weight | Per 100 g
increase | 5.60
(3.10-10.2) | <0.001 | - | - | - | - | - | - |
| 1-min Apgar
score | Per 1-point
increase | 3.42
(1.40-8.37) | <0.001 | - | - | - | - | - | - |
| Sex | Male vs.
female | 0.97
(0.41-2.29) | 0.946 | - | - | - | - | - | - |
| Mode of
delivery | Cesarean vs.
vaginal | 0.43
(0.16-1.17) | 0.097 | - | - | - | - | - | - |
| Maternal
gestational diabetes | Yes vs. no | 1.05
(0.37-3.00) | 0.556 | - | - | - | - | - | - |
| Maternal
gestational hypertension | Yes vs. no | 1.48
(0.56-3.91) | 0.467 | - | - | - | - | - | - |
| Neonatal
pneumonia | Yes vs. no | 2.32
(0.83-6.48) | 0.196 | - | - | - | - | - | - |
| NBPD | Yes vs. no | 14.5
(4.8-43.7) | <0.001 | - | - | - | - | - | - |
| NRDS | Yes vs. no | 3.90
(1.60–9.50) | 0.003 | - | - | - | - | - | - |
| Neonatal
sepsis | Yes vs. no | 4.30
(1.40-13.5) | 0.009 | - | - | - | - | - | - |
| Blood
transfusion | Yes vs. no | 33.1 (4.3-256) | <0.001 | - | - | - | - | - | - |
| LMR within 24
h | Per 1-unit
increase | 4.29
(2.02-9.12) | 0.015 | - | - | - | - | - | - |
| LMR at 1 week | Per 1-unit
increase | 4.30
(2.02-9.15) | 0.010 | - | - | - | - | - | - |
| MPV at 1 week | Per 1 fl
increase | 0.49
(0.25-0.96) | 0.047 | - | - | - | - | - | - |
The analysis included the following variables:
Gestational age, birth weight, 1-min Apgar score, neonatal
pneumonia, NBPD, neonatal sepsis, NRDS, blood transfusion
requirements,24 h LMR, 1 week MPV and 1 week LMR.
Although NBPD, NRDS, sepsis, blood transfusion, LMR
and MPV were significant in univariate analysis, they lost
independent significance after adjustment for gestational age and
were therefore not retained in the final model. Univariate logistic
regression was first performed for each potential risk factor;
variables with P<0.05 were entered into the multivariate model.
For dichotomous exposures, the reference category was always the
group with the presumed lowest risk (such as female for sex,
vaginal for delivery mode, no for morbidities). Continuous
variables were analyzed per-unit increase. Multivariate logistic
regression analysis identified gestational age as the sole
independent risk factor for severe ROP development (β=-0.545;
Wald=17.264; P<0.001; odds ratio=0.580; 95% CI: 0.4-0.7;
Table IV). The OR indicates that
for each additional week of gestational age, the risk of developing
severe ROP decreased by ~42%. Due to high collinearity between
gestational age and birth weight (r=0.78, VIF=8.9), birth weight
was excluded from the final model; consequently, gestational age
remained the only independent predictor.
Predictive performance of gestational
age
To assess the early predictive value of gestational
age for severe ROP development, ROC curve analysis was performed
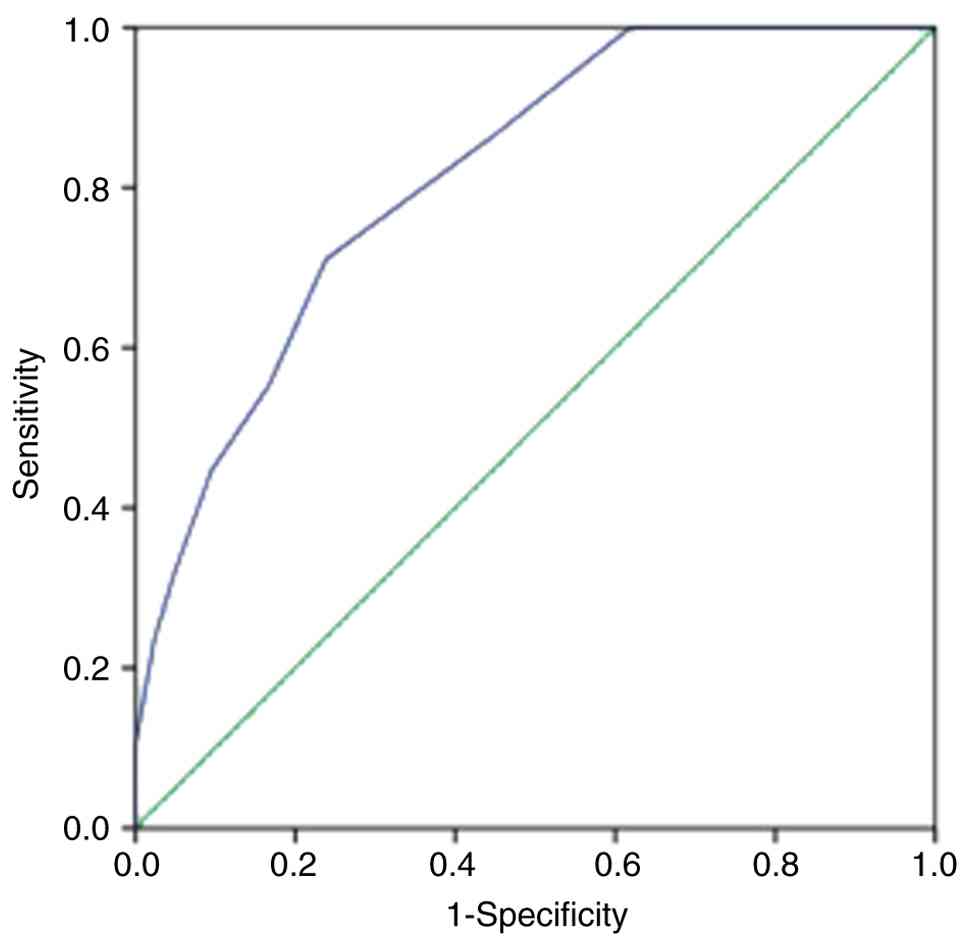
(Table V; Fig. 1) and the AUC was 0.815 (95% CI:
0.725-0.906; P<0.001), indicating good discriminatory ability.
Using Youden's index to determine the optimal threshold, a
gestational age of 30.5 weeks was identified as the cut-off value,
yielding a sensitivity of 76.2% and specificity of 71.1% for
predicting severe ROP development. This threshold suggested that
infants born at or before 30.5 weeks of gestational age have a
significantly increased risk for developing severe ROP requiring
therapeutic intervention.
 | Table VEarly predictive value of gestational
age in severe retinopathy of prematurity. |
Table V
Early predictive value of gestational
age in severe retinopathy of prematurity.
| Indicator | AUC | Sensitivity | Specificity | Optimal threshold,
weeks | 95% CI | P-value |
|---|
| Gestational
age | 0.815 | 0.762 | 0.711 | 30.5 | 0.725-0.906 | <0.001 |
Discussion
The present retrospective case-control study
provided comprehensive insights into the risk factors associated
with severe ROP in preterm infants. The present findings
corroborated established knowledge while unveiling novel
associations that may enhance the understanding of severe ROP
pathogenesis and improve clinical risk stratification
strategies.
Gestational age and birth weight are universally
recognized as the most notable risk factors for ROP development and
progression (4,13). The present study reinforces this
relationship, demonstrating that infants with severe ROP had
significantly lower gestational age and birth weight compared with
those with mild ROP; these findings align with extensive literature
documenting the inverse association between gestational age/birth
weight and ROP severity (14,15).
The pathophysiological basis for this association lies in the
incomplete retinal vascularization at birth in extremely preterm
infants, whose immature organ systems render them particularly
vulnerable to oxidative stress and inflammatory insults that
disrupt normal retinal angiogenesis (16).
Multivariate logistic regression analysis identified
gestational age as an independent risk factor for severe ROP, with
an optimal threshold of 30.5 weeks demonstrating 76.2% sensitivity
and 71.1% specificity for predicting severe ROP development. This
threshold aligns closely with current screening guidelines that
emphasize heightened fundus examinations for infants born <32
weeks gestation (8). A recent
large-scale prospective cohort study by Mhina et al
(17) involving 193 Tanzanian
preterm infants similarly identified gestational age <32 weeks
as a critical threshold, with affected infants demonstrating a
6.8-fold increased risk for severe ROP. The consistency of these
findings across diverse populations underscores the robust
predictive value of gestational age in severe ROP risk
assessment.
The present study identified several perinatal
complications significantly associated with severe ROP development,
including NBPD, NRDS and neonatal sepsis. These associations
highlight the multifactorial nature of ROP pathogenesis, where
systemic inflammatory responses and prolonged oxygen exposure
contribute synergistically to abnormal retinal vascularization
(10,16,18).
The association between NBPD and severe ROP observed
in the present study is particularly notable, as it reflects the
complex interplay between pulmonary and ocular vascular development
in preterm infants. NBPD pathogenesis involves dysregulated
expression of multiple angiogenic factors, including VEGF,
insulin-like growth factor-1 (IGF-1) and transforming growth
factor-β, which may directly influence retinal neovascularization
(19,20). Furthermore, infants with NBPD
typically require prolonged oxygen supplementation, exposing them
to fluctuating oxygen tensions that promote oxidative stress and
subsequent ROP development (21).
The significant association between neonatal sepsis
and severe ROP in the present cohort aligns with emerging evidence
implicating systemic inflammation in ROP pathogenesis (22). During sepsis, elevated levels of
pro-inflammatory cytokines such as IL-6, IL-8 and tumor necrosis
factor-α (TNF-α) can directly damage retinal vascular endothelium
and induce retinal hypoxia, thereby stimulating pathological VEGF
release and neovascularization (23,24).
Meta-analysis evidence by Wang et al (25) demonstrated that sepsis increases
ROP risk by ~2.5-fold, with early severe systemic inflammatory
responses particularly disruptive to normal retinal vascular
development.
The present investigation of hematological
parameters revealed several novel associations that warrant further
exploration. The LMR emerged as a potentially protective biomarker,
with significantly higher values observed in the mild ROP group
both within 24 h of birth and at 1 week of age. This finding
suggests that a balanced inflammatory response, characterized by
preserved lymphocyte populations relative to activated monocytes,
may confer protection against severe ROP development (26).
The LMR is regarded as a reliable inflammatory
biomarker across various diseases due to its stability,
cost-effectiveness, and ease of calculation compared with
traditional inflammatory mediators (27). In the context of ROP, elevated LMR
may reflect a more controlled inflammatory response that minimizes
collateral damage to developing retinal vasculature. A recent study
by Xu et al (28) similarly
identified LMR as a prognostic indicator in patients with sepsis,
suggesting its broader applicability as an inflammatory
biomarker.
The simultaneous increase in absolute L and the fall
in LMR are not contradictory. LMR is a ratio; in the severe ROP
cohort the median L value rose from 3.90 to 4.59x109/l
at 24 h, whereas the concomitant median M value increased from 0.48
to 0.77x109/l (data not shown); since the relative
expansion of monocytes exceeded that of lymphocytes, LMR declined
from 8.21 to 6.17. This pattern, absolute lymphocytosis coupled
with a proportionally larger monocyte mobilization, mirrored an
enhanced systemic inflammatory response, as circulating monocytes
are primary sources of TNF-α, IL-6 and VEGF that drive retinal
neovascularization (20).
Consequently, the lower LMR reflected not lymphocyte depletion, but
an over-proportionate increase in monocytes, supporting the
hypothesis that dysregulated inflammation contributes to severe ROP
development.
Notably, the present study also identified MPV as
significantly lower in the severe ROP group at 1 week of age. This
association may reflect enhanced platelet activation and altered
hemostatic function in infants at risk of developing severe ROP
(29). Platelets contain α
granules storing diverse angiogenic regulatory factors, including
both pro-angiogenic agents (VEGF, IGF-1) and anti-angiogenic
factors (endostatin), which are released in response to various
stimuli to modulate retinal neovascularization (30). The observed increase in MPV may
indicate heightened platelet activation and degranulation,
potentially contributing to the dysregulated angiogenic environment
characteristic of severe ROP (31).
The 1-min Apgar score demonstrated a significant
association with ROP severity in the present study, corroborating
previous findings by Ali et al (32) and Marinov et al (33), which identified low Apgar scores as
predictors of severe ROP development. The Apgar score provides a
rapid assessment of neonatal adaptation immediately after birth,
with lower scores reflecting compromised physiological status that
may predispose infants to subsequent complications (34), including severe ROP. However, in
the present multivariate analysis, Apgar score did not emerge as an
independent predictor, suggesting that its influence on ROP
development may be mediated through other factors such as increased
susceptibility to sepsis, prolonged ventilatory support or
hemodynamic instability (35).
The requirement for blood transfusion was
significantly more common in the severe ROP group, reflecting both
the overall medical complexity of these infants and potential
direct effects of transfusion on ROP development. Blood
transfusions in preterm infants have been associated with increased
oxidative stress due to iron overload and altered oxygen-carrying
capacity, potentially contributing to retinal vascular damage
(36). Additionally, frequent
transfusions often indicate underlying medical instability,
prolonged hospitalization and exposure to multiple risk factors
that collectively increase severe ROP risk (37).
Notably, the present study did not identify
significant associations between maternal third-trimester lipid
parameters and severe ROP development, despite theoretical
considerations suggesting potential relationships. Maternal lipid
metabolism during pregnancy influences fetal development, including
retinal vascularization, through effects on placental function and
nutrient transfer (10). However,
the complex interplay between maternal metabolic factors and
neonatal ROP development may be overshadowed by more potent
postnatal risk factors in extremely preterm infants. Future studies
with larger sample sizes and more detailed maternal metabolic
profiling may be needed to detect subtle associations between
maternal factors and severe ROP risk.
The identification of multiple independent risk
factors for severe ROP development has important clinical
implications for risk stratification and screening protocols. While
current guidelines primarily rely on gestational age and birth
weight criteria, the present findings suggested that incorporating
additional parameters such as systemic inflammatory markers,
specific perinatal complications and early hematological indices
could potentially enhance predictive accuracy (36). The development of composite risk
scores incorporating multiple biomarkers may enable more precise
identification of high-risk infants requiring intensive
surveillance and early intervention.
Furthermore, the recognition of modifiable risk
factors such as sepsis prevention, optimal oxygen management and
inflammatory response modulation provides opportunities for
targeted interventions that may reduce severe ROP incidence
(11). Enhanced infection control
measures, use of oxygen therapy and consideration of
anti-inflammatory strategies in high-risk infants represent
potential areas for clinical improvement.
The present control group comprised only infants who
developed any stage of ROP that subsequently regressed, which
introduces selection bias inherent to the case-control design;
infants who never developed ROP were excluded. This approach
intentionally compared two points on the same disease spectrum
(mild vs. severe) rather than comparing ‘disease’ with ‘no
disease’. The advantage is that constitutional determinants of
prematurity per se are partially neutralized, allowing the
research to focus on factors that drive progression from mild to
severe ROP. However, this has limitations: First, effect sizes of
classical risk factors (gestational age and birth weight) may be
diluted because numerous controls were still relatively mature
(median gestational age, 32 weeks); second, the baseline risk of
developing ROP cannot be quantified, only the risk of escalating
within the ROP phenotype. Consequently, the ORs reported here are
specific to ‘progression’ rather than ‘incidence’ and extrapolation
to the broader <37-week population should be made cautiously.
Future studies limited to infants born at ≤30 weeks gestation and
explicitly including an ROP-free cohort, will be needed to
re-estimate both incidence and progression effects without this
spectrum bias.
The present study has several other limitations that
should be acknowledged: The retrospective design limits the ability
to establish causal relationships and may introduce selection bias;
the relatively small sample size may have limited the power to
detect subtle associations, particularly regarding maternal
factors. Additionally, the single-center design may limit the
generalizability of the present findings to other populations with
different demographic characteristics or clinical practices.
In conclusion, the present study identified
gestational age ≤30.5 weeks as the strongest predictor of severe
ROP, whereas a persistently low LMR emerged as a potential novel
biomarker for early risk stratification. Enhanced surveillance and
timely intervention targeting high-risk infants, particularly those
with very low gestational age, systemic complications and abnormal
inflammatory profiles, are essential for improving clinical
outcomes and preserving vision in this vulnerable population.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Hebei Province
Medical Applicable Technology Tracking Project (grant no.
GZ2020094).
Availability of data and materials
The data generated in the present study are not
publicly available due to restrictions imposed by the institutional
review ethics committee concerning participant confidentiality and
privacy, but may be requested from the corresponding author.
Authors' contributions
QM and XL were involved in the study conception and
design, data acquisition, analysis and interpretation, and drafting
of the manuscript. JW performed the statistical analysis, prepared
the figures and provided critical revision for intellectual
content. KL and WT were involved in patient recruitment, data
collection and technical support. QM and XL confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript, and agree to be accountable for all aspects of
the work.
Ethics approval and consent to
participate
The present study protocol was reviewed and approved
by the Medical Ethics Committee of Hebei General Hospital [approval
no. 115 (2024)]. Given the retrospective nature of the study
utilizing existing medical records, informed consent requirements
were waived by the ethics committee, with appropriate measures
taken to ensure patient confidentiality and data protection.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Karmouta R, Strawbridge JC, Langston S,
Altendahl M, Khitri M, Chu A and Tsui I: Neurodevelopmental
outcomes in infants screened for retinopathy of prematurity. JAMA
Ophthalmol. 141:1125–1132. 2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sabri K, Ells AL, Lee EY, Dutta S and
Vinekar A: Retinopathy of prematurity: A global perspective and
recent developments. Pediatrics. 150(e2021053924)2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Dammann O, Hartnett ME and Stahl A:
Retinopathy of prematurity. Dev Med Child Neurol. 65:625–631.
2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Han G, Lim DH, Kang D, Cho J, Guallar E,
Chang YS, Chung TY, Kim SJ and Park WS: Association between
retinopathy of prematurity in very-low-birth-weight infants and
neurodevelopmental impairment. Am J Ophthalmol. 244:205–215.
2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wu T, Zhang L, Tong Y, Qu Y, Xia B and Mu
D: Retinopathy of prematurity among very low-birth-weight infants
in China: Incidence and perinatal risk factors. Invest Ophthalmol
Vis Sci. 59:757–763. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Xu S, Liang Z, Du Q, Li Z, Tan G, Nie C,
Yang Y, Lv X, Zhang C and Luo X: A systematic study on the
prevention and treatment of retinopathy of prematurity in China.
BMC Ophthalmol. 18(44)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hong EH, Shin YU and Cho H: Retinopathy of
prematurity: A review of epidemiology and current treatment
strategies. Clin Exp Pediatr. 65:115–126. 2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Al Amro SA, Al Aql F, Al Hajar S, Al Dhibi
H, Al Nemri A, Mousa A and Ahmad J: Practical guidelines for
screening and treatment of retinopathy of prematurity in Saudi
Arabia. Saudi J Ophthalmol. 32:222–226. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zonda GI, Mogos R, Melinte-Popescu AS,
Adam AM, Harabor V, Nemescu D, Socolov D, Harabor A,
Melinte-Popescu M, Hincu MA, et al: Hematologic risk factors for
the development of retinopathy of prematurity-a retrospective
study. Children (Basel). 10(567)2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Tang W, Zhang Y, Zhang H, Li K, Zhao Z, Ma
H, Jiang X, Jia Z and Ma Q: Progress in the study of association
between hematological indicators and retinopathy of prematurity
(Review). Biomed Rep. 21(111)2024.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hartnett ME and Penn JS: Mechanisms and
management of retinopathy of prematurity. N Engl J Med.
367:2515–2526. 2012.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Celik K, Ekinci D, Asena M and Matur NO:
Can hematological parameters be a indicator risk factor in the
development of retinopathy of prematurity? Klin Padiatr.
233:216–220. 2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chiang MF, Quinn GE, Fielder AR, Ostmo SR,
Paul Chan RV, Berrocal A, Binenbaum G, Blair M, Peter Campbell J,
Capone A Jr, et al: International classification of retinopathy of
prematurity, third edition. Ophthalmology. 128:e51–e68.
2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bhatnagar A, Skrehot HC, Bhatt A, Herce H
and Weng CY: Epidemiology of retinopathy of prematurity in the US
from 2003 to 2019. JAMA Ophthalmol. 141:479–485. 2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Hu X, Zhang J, Zhang M, Chen X, Han S and
Zhu J: Incidence and risk factors for retinopathy of prematurity in
a tertiary hospital in China. Clin Ophthalmol. 17:3189–3194.
2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wang J, Li M, Geng Z, Khattak S, Ji X, Wu
D and Dang Y: Role of oxidative stress in retinal disease and the
early intervention strategies: A review. Oxid Med Cell Longev.
2022(7836828)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Mhina C, Mtogo Y, Mafwiri M, Sanyiwa A,
Mosenene NS and Malik ANJ: Prevalence and associated factors for
retinopathy of prematurity at a tertiary hospital in Dar es Salaam,
Tanzania. Eye (Lond). 39:1476–1480. 2025.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kim SJ, Port AD, Swan R, Campbell JP, Chan
RVP and Chiang MF: Retinopathy of prematurity: A review of risk
factors and their clinical significance. Surv Ophthalmol.
63:618–637. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Podraza W, Michalczuk B, Jezierska K,
Domek H, Kordek A, Łoniewska B, Modrzejewska M and Kot J:
Correlation of retinopathy of prematurity with bronchopulmonary
dysplasia. Open Med (Wars). 13:67–73. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Rivera JC, Holm M, Austeng D, Morken TS,
Zhou TE, Beaudry-Richard A, Sierra EM, Dammann O and Chemtob S:
Retinopathy of prematurity: inflammation, choroidal degeneration,
and novel promising therapeutic strategies. J Neuroinflammation.
14(165)2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Stoll BJ, Hansen NI, Bell EF, Walsh MC,
Carlo WA, Shankaran S, Laptook AR, Sánchez PJ, Van Meurs KP,
Wyckoff M, et al: Trends in care practices, morbidity, and
mortality of extremely preterm neonates, 1993-2012. JAMA.
314:1039–1051. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Almutairi M, Chechalk K, Deane E, Fox R,
Janes A, Maguire-Henry T, McCabe D, O'Connor C, Quirk J, Swan E, et
al: Biomarkers in retinopathy of prematurity: A systematic review
and meta-analysis. Front Pediatr. 12(1371776)2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Tremblay S, Miloudi K, Chaychi S, Favret
S, Binet F, Polosa A, Lachapelle P, Chemtob S and Sapieha P:
Systemic inflammation perturbs developmental retinal angiogenesis
and neuroretinal function. Invest Ophthalmol Vis Sci. 54:8125–8139.
2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kıran Yenice E, Kara C, Karsli Türkoglu T,
Ulubaş Işık D and Çelik İH: Predictive value of serum inflammatory
markers in retinopathy of prematurity. Eye (Lond). 38:2822–2826.
2024.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wang X, Tang K, Chen L, Cheng S and Xu H:
Association between sepsis and retinopathy of prematurity: A
systematic review and meta-analysis. BMJ Open.
9(e025440)2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Bujoreanu Bezman L, Tiutiuca C, Bujoreanu
FC, Cârneciu N, Crăescu M, Dimofte F, Niculeț E and Nechita A: From
blood count parameters to ROP risk: Early hematological predictors
in preterm infants. Medicina (Kaunas). 61(1581)2025.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Oruz O, Dervişoğulları MS, Öktem ME and
İncekaş C: Predictive role of systemic immune-inflammation index
and neutrophil/lymphocyte ratio values in infants with retinopathy
of prematurity. Graefes Arch Clin Exp Ophthalmol. 262:3125–3134.
2024.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Xu T, Song S, Zhu K, Yang Y, Wu C, Wang N
and Lu S: Systemic inflammatory response index improves prognostic
predictive value in intensive care unit patients with sepsis. Sci
Rep. 15(1908)2025.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Choręziak A, Szpecht D,
Chmielarz-Czarnocińska A, Pawłowska I and Gotz-Więckowska A: The
association of platelet counts with development and treatment for
retinopathy of prematurity-is thrombocytopenia a risk factor? Arch
Med Sci. 18:400–405. 2022.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Parrozzani R, Nacci EB, Bini S, Marchione
G, Salvadori S, Nardo D and Midena E: Severe retinopathy of
prematurity is associated with early post-natal low platelet count.
Sci Rep. 11(891)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Deger I, Erturul S, Kaya IK, Yılmaz ST and
Yolbaş I: Can platelet count, platelet mass index and mean platelet
volume be parameters in retinopathy of prematurity? East J Med.
27:513–518. 2022.
|
|
32
|
Ali YF, El-Morshedy S, Imam AA,
Abdelrahman NI, Elsayed RM, Alkholy UM, Abdalmonem N and Shehab MM:
The role of serum apelin in retinopathy of prematurity. Clin
Ophthalmol. 11:387–392. 2017.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Marinov VG, Koleva-Georgieva DN, Sivkova
NP and Krasteva MB: The 5-min apgar score as a prognostic factor
for development and progression of retinopathy of prematurity.
Folia Med (Plovdiv). 59:78–83. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Furness A, Fair F, Higginbottom G, Oddie S
and Soltani H: A review of the current policies and guidance
regarding Apgar scoring and the detection of jaundice and cyanosis
concerning black, Asian and ethnic minority neonates. BMC Pediatr.
24(198)2024.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Góralska A, Puskarz-Gąsowska JE, Bujnowski
P and Bokiniec R: Assessing the risk of retinopathy of prematurity
in preterm neonates using the expanded Apgar score. Pediatr Med
Rodz. 20:402–408. 2024.
|
|
36
|
Stutchfield CJ, Jain A, Odd D, Williams C
and Markham R: Foetal haemoglobin, blood transfusion, and
retinopathy of prematurity in very preterm infants: A pilot
prospective cohort study. Eye (Lond). 31:1451–1455. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Wang X, Rao R, Li H, Lei X and Dong W: Red
blood cell transfusion for incidence of retinopathy of prematurity:
Prospective multicenter cohort study. JMIR Pediatr Parent.
7(e60330)2024.PubMed/NCBI View
Article : Google Scholar
|