Introduction
Gastrointestinal stromal tumor (GIST) represents the
most common mesenchymal neoplasm of the gastrointestinal tract
(1,2). The pathogenesis of GIST is
predominantly driven by activating mutations in the KIT
proto-oncogene, receptor tyrosine kinase (KIT) or platelet-derived
growth factor receptor alpha (PDGFRA) genes (1). Approximately 70-80% of GISTs harbor
KIT mutations, with exon 11 being the most frequently affected
site, followed by less common mutations in exons 9, 13 or
17(1). PDGFRA mutations account
for approximately one-third of KIT-wild-type GISTs (3,4).
Tumors lacking mutations in both KIT and PDGFRA, termed wild-type
GISTs, may harbor alterations in other genes, including succinate
dehydrogenase, B-Raf proto-oncogene, serine/threonine kinase or
KRAS proto-oncogene, GTPase (1).
Complete surgical resection remains the primary
treatment modality for localized GIST (2-4).
However, disease recurrence frequently occurs in patients with
high-risk features, including large tumor size, elevated mitotic
index, non-gastric location, tumor rupture or specific molecular
profiles (8,5-9).
While numerous patients achieve a cure through surgery alone
(2-4),
adjuvant imatinib therapy for a minimum of three years is
recommended for those at substantial risk of recurrence (5,6).
Recent advances in GIST research have focused on
developing prognostic tools that integrate clinical and
pathological factors to improve risk stratification beyond
traditional classification systems. However, most existing
prognostic models were developed for surgical cohorts or metastatic
disease settings, with limited tools specifically designed for
patients receiving adjuvant therapy (10-12).
This recommendation is supported by three pivotal
randomized controlled trials: ACOSOG Z9001 (NCT00041197) (7), EORTC 62024(8) and SSG XVIII (5). These studies demonstrated that
adjuvant imatinib significantly improves recurrence-free survival
(RFS) compared to placebo (7) or
observation alone (8), with 3
years of therapy proving superior to 1 year in enhancing both RFS
and overall survival in patients with KIT-positive high-risk GIST
(5). Although an 800-mg dose
represents an option for patients with KIT exon 9 mutations in
advanced disease, retrospective analyses of adjuvant therapy showed
no superiority over the standard 400-mg daily dose (5).
Despite optimal treatment, a substantial proportion
of patients experience disease recurrence following completion of
the standard 3-year imatinib course (4). The PERSIST-5 study investigated
extending adjuvant therapy to 5 years, reporting a 90% 5-year RFS
rate, though nearly half of participants discontinued treatment
prematurely (9). Whether
prolonging treatment beyond 3 years provides an additional clinical
benefit remains unestablished in randomized trials. The ongoing
IMADGIST study, initiated in 2014, seeks to determine whether an
additional three years of adjuvant imatinib improves outcomes
compared to the standard 3-year duration in high-risk patients
(8,13,14).
While numerous studies have evaluated recurrence
risk following surgery alone (8,9),
limited data exist regarding prognostic factors in patients treated
with combined surgical resection and adjuvant imatinib. Such
insights are essential for individualized surveillance strategies
and patient counseling.
Therefore, the present study aims to evaluate
prognostic factors and long-term outcomes of adjuvant imatinib
therapy using national registry data to develop a practical
nomogram for risk stratification in this treatment setting.
Patients and methods
Study design and patient
population
This retrospective, multicenter registry study was
conducted using the Taiwan Cooperative Oncology Group protocol
T1218 with participation from 11 hospitals across Taiwan: i)
National Taiwan University Hospital (Taipei City); ii) Taipei
Veterans General Hospital (Taipei City); iii) Mackay Memorial
Hospital (Taipei City); iv) Tri-Service General Hospital (Taipei
City); v) Linkou Chang Gung Memorial Hospital (Taoyuan City); vi)
China Medical University Hospital (Taichung City); vii) Changhua
Christian Hospital (Changhua City); viii) National Cheng Kung
University Hospital (Tainan City); ix) Kaohsiung Medical University
Hospital (Kaohsiung City); x) Kaohsiung Veterans General Hospital
(Kaohsiung City); and xi) Kaohsiung Chang Gung Memorial Hospital
(Kaohsiung City, Taiwan).
Between 2013 (when adjuvant imatinib was
incorporated into Taiwan's National Health Insurance reimbursement
program) and 2023, a total of 269 patients with pathologically
confirmed, primary resectable, c-KIT-positive modified Armed Forces
Institute of Pathology (AFIP)-classified high-risk GISTs who
received adjuvant imatinib therapy were enrolled from institutional
databases (15). Patient data were
collected retrospectively for cases treated between 2013 and the
date of institutional review board (IRB) approval at each
institution, and prospectively thereafter through 2023 as part of
the ongoing national registry (16).
Inclusion criteria as per a previous study (17) required patients to have undergone
tumor resection as primary treatment with complete macroscopic
resection confirmed, received adjuvant imatinib therapy as per
institutional guidelines, no prior tyrosine kinase inhibitor
therapy before surgery, no evidence of metastatic disease before or
during surgery and AFIP-classified high recurrence risk as
determined by the treating physician using modified National
Institutes of Health criteria (18). Patient data were collected
retrospectively through chart review using standardized data
collection forms (Data SI).
Data collection encompassed baseline demographics,
pathological characteristics, mutation profiles, imatinib treatment
duration and dosing, and RFS outcomes. The study protocol received
approval from the IRB of each participating hospital with the
following approval numbers: (Institution 1: IRB-201806048RSC),
(Institution 2: IRB-2018-09-004CC), (Institution 3: IRB-20CT002be),
(Institution 4: IRB-C202005012), (Institution 5, 11:
IRB-201801966A2), (Institution 6: IRB-CMUH107-REC1-108),
(Institution 7: IRB-181203), (Institution 8: IRB-A-ER-107-271),
[Institution 9: IRB-F(II)-20200081] and (Institution 10:
IRB-KSVGH22-CT2-08), and written informed consent was obtained from
all living patients prior to enrollment (16). Chang Gung Memorial Hospital Linkou
(Institution 5) and Chang Gung Memorial Hospital Kaohsiung
(Institution 11) share the same IRB approval number under their
unified institutional review system.
Treatment assessment
Adjuvant imatinib therapy duration was
systematically documented from medical records. The treatment
duration was categorized as time (T) <1 year, 1<T≤2 years,
2<T≤3 years and T>3 years. The standard dose of imatinib was
400 mg daily unless modified for toxicity. Treatment completion was
based on documented treatment periods in the medical records.
Follow-up protocol
Patients underwent regular follow-up assessments,
including physical examinations, performance status evaluation,
body weight monitoring, complete blood counts and comprehensive
serum biochemistry testing. Imatinib administration details and
adverse events were systematically documented throughout the
treatment period.
Surveillance imaging with abdominal computed
tomography scans was performed according to institutional
protocols, typically every 3 months during the initial 3 years of
follow-up, followed by 6-monthly intervals for an additional 2
years. Disease recurrence was defined by radiological evidence of
new lesions or clinical progression based on multidisciplinary team
assessment. RFS was defined as the interval from adjuvant imatinib
initiation to documented tumor recurrence or last follow-up
visit.
Molecular analysis
Tumor specimens obtained through surgical resection
or biopsy were processed as formalin-fixed, paraffin-embedded
tissue blocks. Molecular testing was performed when available
during the study period. Tumor-enriched sections underwent genomic
DNA extraction, followed by PCR amplification of KIT and PDGFRA
gene regions. Mutation analysis was conducted according to
previously established protocols (19,20).
However, mutation testing was not systematically performed across
all centers and time periods, resulting in missing molecular data
for 126 patients (46.8%).
The anatomical tumor location was classified into
four distinct categories based on surgical considerations and
anatomical differences in operative approach: i) Stomach; ii)
duodenum; iii) small bowel (jejunum and ileum); and iv) colorectum.
While the duodenum is anatomically part of the small intestine, it
was classified separately due to the fundamentally different
surgical procedures required for duodenal vs. jejunal/ileal GISTs.
Duodenal GISTs often require pancreaticoduodenectomy or complex
duodenal reconstruction, whereas small bowel GISTs typically
undergo segmental resection with primary anastomosis (21).
Statistical considerations for missing
data
Given the retrospective registry nature of the
present study, mutation data were unavailable for 126 patients
(46.8% of the cohort). This missing data pattern reflects the
evolution of molecular testing practices during the study period
(2013-2023) rather than random missingness. The mutation status was
analyzed as an available category and sensitivity analyses were
conducted to assess the impact of missing molecular data on
nomogram performance.
Nomogram development and
validation
A prognostic nomogram was constructed using R
software (version 4.3.1) with the rms package and supporting
libraries. Point assignments were based on Cox regression
coefficients from the final multivariate model. Nomogram
performance was evaluated using the concordance index (C-index),
and calibration was assessed by comparing predicted vs. observed
disease-free survival probabilities. Internal validation was
performed using 1,000 bootstrap resamples to ensure model
robustness.
Statistical analysis
Categorical variables were compared using Chi-square
or Fisher's exact tests, as appropriate. Continuous variables were
analyzed using independent-samples t-tests or Mann-Whitney U-tests
based on data distribution characteristics. Survival distributions
were estimated using Kaplan-Meier methodology, with group
comparisons performed using log-rank tests.
Univariate Cox proportional hazards regression was
used to identify factors associated with disease-free survival.
Variables with statistical significance or clinical relevance were
included in multivariate Cox regression modeling.
To assess the prognostic significance of nomogram
total points, recursive partitioning analysis was utilized to
determine optimal cut-off values and stratify patients into
distinct risk groups (22).
Statistical significance was defined as P<0.05
for all analyses. All statistical analyses were performed using R
software version 4.3.1.
Results
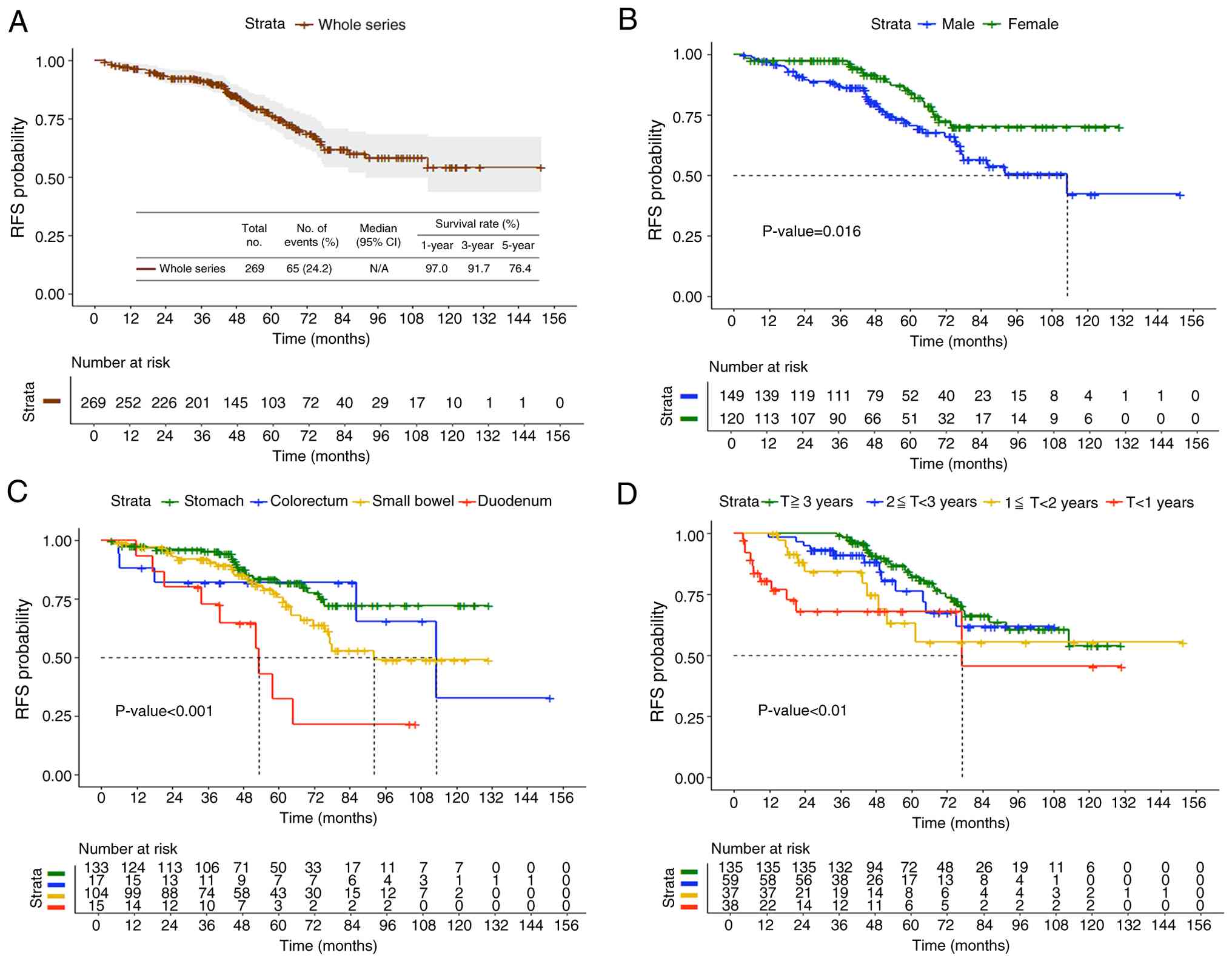
Patient characteristics
The demographic and clinicopathological
characteristics of the 269 patients with modified AFIP high-risk
GIST receiving adjuvant imatinib are presented in Table I. The cohort comprised 149 males
(55.4%) and 120 females (44.6%) with a mean age of 60.9±11.4 years.
The median follow-up duration was 62.7 months (range: 7.2-151.9
months).
 | Table IBaseline characteristics (n=269). |
Table I
Baseline characteristics (n=269).
|
Characteristics | No. of cases | Mean ± SD or % |
|---|
| Age, yr | | 60.9±11.4 |
|
≤65 | 179 | 66.5 |
|
>65 | 90 | 33.5 |
| Sex | | |
|
Male | 149 | 55.4 |
|
Female | 120 | 44.6 |
| Site | | |
|
Stomach | 133 | 49.4 |
|
Colorectum | 17 | 6.3 |
|
Small
bowel | 104 | 38.7 |
|
Duodenum | 15 | 5.6 |
| Tumor size, cm | | 9.3 ± 4.9 |
|
>2,≤5 | 40 | 14.8 |
|
>5,≤10 | 132 | 49.1 |
|
>10 | 97 | 36.1 |
| Mitotic counts/50
HPFs | | |
|
≤5 | 75 | 27.9 |
|
6-10 | 77 | 28.6 |
|
>10 | 117 | 43.5 |
| Rupture | | |
|
No | 227 | 84.4 |
|
Yes | 42 | 15.6 |
| Mutation | | |
|
Exon 9 | 15 | 5.6 |
|
Exon 10 | 1 | 0.4 |
|
Exon 11 | 119 | 44.2 |
|
Exon 12 | 1 | 0.4 |
|
Exon 13 | 2 | 0.8 |
|
Exon 18 | 3 | 1.1 |
|
Wild
type | 2 | 0.7 |
|
N/A | 126 | 46.8 |
| Duration of
Imatinib treatment, yr | | |
|
<1 | 38 | 14.1 |
|
1<T≤2 | 37 | 13.8 |
|
2<T≤3 | 59 | 21.9 |
|
>3 | 135 | 50.2 |
The stomach was the most common tumor location
(133/269; 49.4%), followed by the small bowel (104/269; 38.7%),
with fewer cases originating from the colorectum (17/269; 6.3%) and
duodenum (15/269; 5.6%). The mean tumor size was 9.3±4.9 cm, with
36.1% of tumors >10 cm. Among the 269 patients, 119 (44.2%)
harbored KIT exon 11 mutations; however, mutation data were
unavailable for 126 patients (46.8%). Tumor rupture was present in
42 patients (15.6%).
Treatment duration distribution
The treatment duration (T) distribution was as
follows: T<1 year (n=38, 14.1%), 1<T≤2 years (n=37, 13.8%),
2<T≤3 years (n=59, 21.9%) and T>3 years (n=135, 50.2%). The
majority of patients (50.2%) completed >3 years of adjuvant
therapy.
Factors associated with disease
recurrence
Table II presents
the relationship between recurrence status and clinicopathologic
features. Univariate analysis showed significant associations with
male sex (P=0.010) and tumor site (P<0.001), and a duodenal
location was associated with the highest recurrence rate (60.0 vs.
15.8% for gastric tumors).
 | Table IIRelationship between recurrence
status and clinicopathologic features of gastrointestinal stromal
tumor with imatinib adjuvant treatment. |
Table II
Relationship between recurrence
status and clinicopathologic features of gastrointestinal stromal
tumor with imatinib adjuvant treatment.
| Parameter | Total N | No recurrence, n
(%) | Recurrence, n
(%) | P-value | Odds ratio | 95% CI | P-value |
|---|
| Age, yr | | | | 0.407 | | | |
|
≤65 | 179 | 133 (74.3) | 46 (25.7) | | 1.29 | 0.70-2.37 | 0.408 |
|
>65 | 90 | 71 (78.9) | 19 (21.1) | | 1 | | |
| Sex | | | | 0.010 | | | |
|
Male | 149 | 104 (69.8) | 45 (30.2) | | 2.16 | 1.20-3.92 | 0.011 |
|
Female | 120 | 100 (83.3) | 20 (16.7) | | 1 | | |
| Site | | | | <0.001 | | | |
|
Stomach | 133 | 112 (84.2) | 21 (15.8) | | 1 | | |
|
Colorectum | 17 | 12 (70.6) | 5 (29.4) | | 2.22 | 0.71-6.97 | 0.171 |
|
Small
bowel | 104 | 74 (71.2) | 30 (28.8) | | 2.16 | 1.15-4.06 | 0.016 |
|
Duodenum | 15 | 6 (40.0) | 9 (60.0) | | 8.00 | 2.58-24.85 | <0.001 |
| Tumor size, cm | | | | 0.093 | | | |
|
>2,
≤5 | 40 | 35 (87.5) | 5 (12.5) | | 1 | | |
|
>5,
≤10 | 132 | 101 (76.5) | 31 (23.5) | | 2.15 | 0.78-5.96 | 0.142 |
|
>10 | 97 | 68 (70.1) | 29 (29.9) | | 2.99 | 1.06-8.39 | 0.038 |
| Mitotic counts/50
HPFs | | | | 0.543 | | | |
|
≤5 | 75 | 58 (77.3) | 17 (22.7) | | 1 | | |
|
6-10 | 77 | 61 (79.2) | 16 (20.8) | | 0.90 | 0.41-1.94. | 0.778 |
|
>10 | 117 | 85 (72.6) | 32 (27.4) | | 1.28 | 0.65-2.53 | 0.468 |
| Rupture | | | | 0.468 | | | |
|
No | 227 | 174 (76.7) | 53 (23.3) | | 1 | | |
|
Yes | 42 | 30 (71.4) | 12 (28.6) | | 1.31 | 0.63-2.74 | 0.469 |
| Mutation | | | | 0.133 | | | |
|
Exon 9 | 15 | 8 (53.3) | 7 (46.7) | | 2.83 | 0.97-8.24 | 0.061 |
|
Exon 11 | 119 | 91 (76.5) | 28 (23.5) | | 1 | | |
|
Others | 7 | 7(100) | 0 | | 0.21 | 0.01-3.86 | 0.195 |
|
Wild
type | 2 | 2(100) | 0 | | 0.64 | 0.03-13.77 | 0.767 |
|
N/A | 126 | 96 (76.2) | 30 (23.8) | | 1.01 | 0.57-1.82 | 0.961 |
| Duration of
treatment, yr | | | | 0.771 | | | |
|
≤1 | 38 | 27 (71.1) | 11 (28.9) | | 1.31 | 0.59-2.94 | 0.510 |
|
1<T≤2 | 37 | 27 (73.0) | 10 (27.0) | | 1.19 | 0.52-2.73 | 0.677 |
|
2<T≤3 | 59 | 47 (79.7) | 12 (20.3) | | 0.82 | 0.39-1.74 | 0.607 |
|
>3 | 135 | 103 (76.2) | 32 (23.7) | | 1 | | |
Table III and
Fig. 1 present the data of
multivariate logistic regression, which identified three
independent risk factors for recurrence: Male sex [adjusted odds
ratio (OR): 2.24, 95% CI: 1.20-4.17, P=0.011], and the tumor
locations with the highest risk were the duodenum (OR: 8.37, 95%
CI: 2.58-27.13, P<0.001) and small bowel (OR: 2.19, 95% CI:
1.15-4.19 P=0.018) compared to a gastric location.
 | Table IIIRelationship between recurrence
status and clinicopathologic features of gastrointestinal stromal
tumor with imatinib adjuvant treatment. |
Table III
Relationship between recurrence
status and clinicopathologic features of gastrointestinal stromal
tumor with imatinib adjuvant treatment.
| Parameter | Odds ratio | 95 % CI | P-value | Adjusted odds
ratio | 95% CI | P-value |
|---|
| Sex (female vs.
male) | 2.16 | 1.20-3.92 | 0.011 | 2.24 | 1.20-4.17 | 0.011 |
| Site | | | | | | |
|
Stomach | 1 | | | 1 | | |
|
Colorectum | 2.22 | 0.71-6.97 | 0.171 | 2.40 | 0.74-7.85 | 0.146 |
|
Small
bowel | 2.16 | 1.15-4.06 | 0.016 | 2.19 | 1.15-4.19 | 0.018 |
|
Duodenum | 8.00 | 2.58-24.85 | <0.001 | 8.37 | 2.58-27.13 | <0.001 |
| Tumor size, cm | | | | | | |
|
>2,
≤5 | 1 | | | 1 | | |
|
>5,
≤10 | 2.15 | 0.78-5.96 | 0.142 | 2.23 | 0.76-6.58 | 0.147 |
|
>10 | 2.99 | 1.06-8.39 | 0.038 | 3.31 | 1.11-9.85 | 0.032 |
Univariate analysis for RFS
Table IV presents
the univariate Cox regression analysis. Significant factors
associated with unfavorable RFS were male sex [hazard ratio (HR):
1.89, 95% CI: 1.12-3.21, P=0.018] and treatment duration <1 year
(HR: 3.03, 95% CI: 1.52-6.05, P=0.002), and the tumor locations
with the highest risk were the duodenum (HR: 4.68, 95% CI:
2.14-10.24, P<0.001) and small bowel (HR: 1.75, 95% CI:
1.01-3.06, P=0.049).
 | Table IVImpact of the clinicopathologic
features on recurrence-free survival in univariate Cox regression
analysis. |
Table IV
Impact of the clinicopathologic
features on recurrence-free survival in univariate Cox regression
analysis.
| Parameter | Total N | Events, n (%) | Hazard ratio | 95% CI | P-value |
|---|
| Age, yr | | | | | |
|
≤65 | 179 | 46 (25.7) | 1 | | |
|
>65 | 90 | 19 (21.1) | 0.99 | 0.58-1.68 | 0.956 |
| Sex | | | | | |
|
Male | 149 | 45 (30.2) | 1.89 | 1.12-3.21 | 0.018 |
|
Female | 120 | 20 (16.7) | 1 | | |
| Site | | | | | |
|
Stomach | 133 | 21 (15.8) | 1 | | |
|
Colorectum | 17 | 5 (29.4) | 1.64 | 0.61-4.36 | 0.325 |
|
Small
bowel | 104 | 30 (28.8) | 1.75 | 1.01-3.06 | 0.049 |
|
Duodenum | 15 | 9 (60.0) | 4.68 | 2.14-10.24 | <0.001 |
| Tumor size, cm | | | | | |
|
>2,
≤5 | 40 | 5 (12.5) | 1 | | |
|
>5,
≤10 | 132 | 31 (23.5) | 2.01 | 0.78-5.18 | 0.147 |
|
>10 | 97 | 29 (29.9) | 2.40 | 0.93-6.21 | 0.070 |
| Mitotic counts/50
HPFs | | | | | |
|
≤5 | 75 | 17 (22.7) | 1 | | |
|
6-10 | 77 | 16 (20.8) | 0.89 | 0.45-1.76 | 0.733 |
|
>10 | 117 | 32 (27.4) | 1.41 | 0.78-2.55 | 0.252 |
| Rupture | | | | | |
|
No | 227 | 53 (23.3) | 1 | | |
|
Yes | 42 | 12 (28.6) | 1.55 | 0.83-2.90 | 0.173 |
|
Mutationa | | | | | |
|
Exon 9 | 15 | 7 (46.7) | 2.22 | 0.92-4.73 | 0.073 |
|
Exon 11 | 119 | 28 (23.5) | 1 | | |
|
Others | 7 | 0 | 0.24 | 0.01-NA | 0.204 |
|
Wild
type | 2 | 0 | 0.69 | 0.01-NA | 0.780 |
|
N/A | 126 | 30 (23.8) | 1.17 | 0.70-1.95 | 0.555 |
| Duration of
treatment, yr | | | | | |
|
<1 | 38 | 11 (28.9) | 3.03 | 1.52-6.05 | 0.002 |
|
1<T≤2 | 37 | 10 (27.0) | 1.91 | 0.94-3.90 | 0.076 |
|
2<T≤3 | 59 | 12 (20.3) | 1.25 | 0.64-2.43 | 0.514 |
|
>3 | 135 | 32 (23.7) | 1 | | |
Multivariate Cox regression and final
model
Table V presents
the final multivariate Cox regression analysis. A total of 3
independent predictors of poor RFS were confirmed: Male sex (HR:
1.76, 95% CI: 1.03-3.00, P=0.039), non-gastric tumor origin with
duodenum showing the highest risk (HR: 6.15, 95% CI: 2.71-13.95,
P<0.0001), followed by the small bowel (HR: 1.92, 95% CI:
1.09-3.38, P=0.025), and treatment duration <1 year (HR: 3.91,
95% CI: 1.91-8.01, P=0.002). These variables were incorporated into
the nomogram construction.
 | Table VImpact of the clinicopathologic
features on recurrence-free survival in multivariate analysis. |
Table V
Impact of the clinicopathologic
features on recurrence-free survival in multivariate analysis.
| | 95% CI of HR | |
|---|
| Prognostic
variable | Adjusted HR | Lower | Upper | P-value | Points assigned in
nomogram |
|---|
| Gender | | | | | |
|
Male | 1.76 | 1.03 | 3.00 | 0.039 | 31 |
|
Female | 1 | | | | 0 |
| Site | | | | | |
|
Stomach | 1 | | | | 0 |
|
Colorectum | 1.50 | 0.56 | 4.01 | 0.417 | 22 |
|
Small
bowel | 1.92 | 1.09 | 3.38 | 0.025 | 36 |
|
Duodenum | 6.15 | 2.71 | 13.95 | <0.0001 | 100 |
| Duration of
treatment, years | | | | | |
|
<1 | 3.91 | 1.91 | 8.01 | 0.002 | 75 |
|
1<T≤2 | 1.89 | 0.91 | 3.92 | 0.089 | 35 |
|
2<T≤3 | 1.16 | 0.59 | 2.28 | 0.665 | 8 |
|
>3 | 1 | 0.17 | 0.66 | | 0 |
Nomogram-based prognostic
stratification
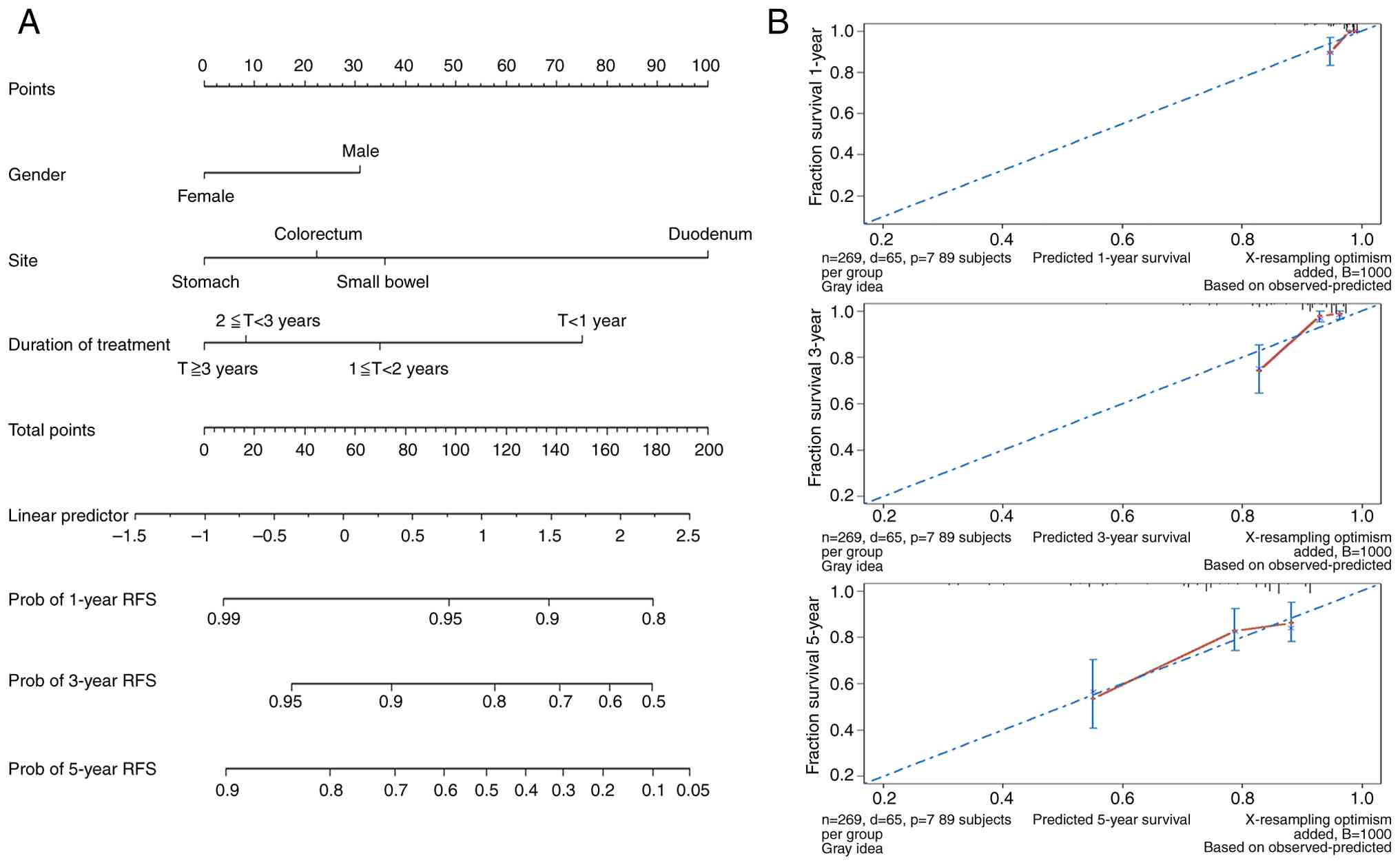
A prognostic nomogram was constructed based on the
final multivariable model (Fig.
2A). The model demonstrated good discriminative ability with a
concordance index of 0.72. Bootstrap validation confirmed model
stability. Calibration curves for 1-, 3- and 5-year RFS
probabilities showed good agreement between nomogram predictions
and actual observations (Fig.
2B).
Table VI shows the
nomogram scoring system with survival probabilities at different
total point scores. The scoring system assigned points based on Cox
regression coefficients: Gender (male: 31 points, female: 0
points), tumor location (stomach: 0 points, colorectum: 22 points,
small bowel: 36 points, duodenum: 100 points), and imatinib
duration (≥3 years: 0 points, 2-3 years: 8 points, 1-2 years: 35
points, <1 year: 75 points) (Fig.
2A).
 | Table VIThe prognostic scoring system. |
Table VI
The prognostic scoring system.
| A, 1-year RFS |
|---|
| Nomogram
points | Probability |
|---|
| 178 | 0.80 |
| 137 | 0.90 |
| 97 | 0.95 |
| 8 | 0.99 |
| B, 3-year RFS |
| 178 | 0.50 |
| 161 | 0.60 |
| 141 | 0.70 |
| 116 | 0.80 |
| 74 | 0.90 |
| 35 | 0.95 |
| C, 5-year RFS |
| 193 | 0.05 |
| 178 | 0.10 |
| 159 | 0.20 |
| 143 | 0.30 |
| 128 | 0.40 |
| 112 | 0.50 |
| 95 | 0.60 |
| 76 | 0.70 |
| 50 | 0.80 |
| 9 | 0.90 |
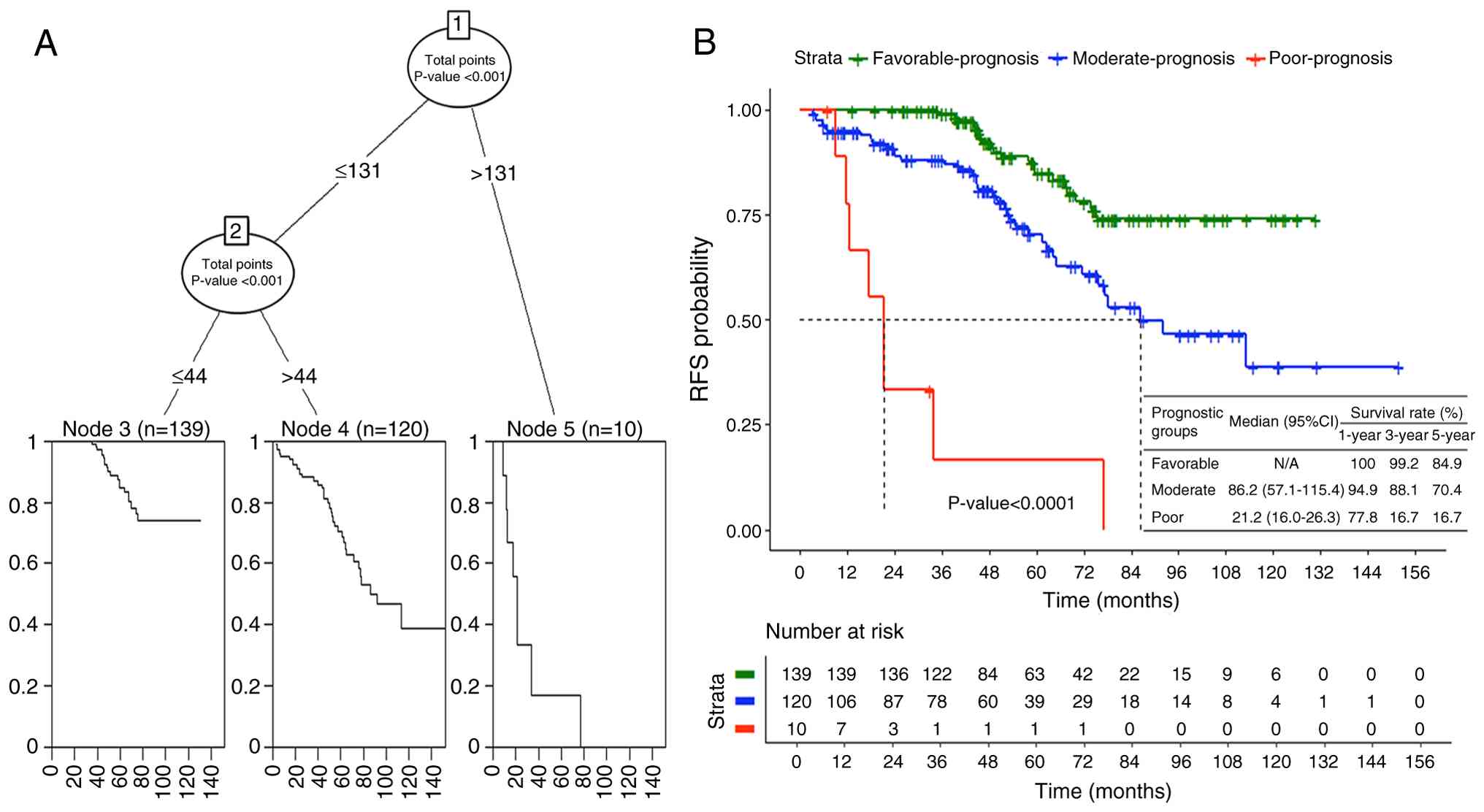
Recursive partitioning analysis (Fig. 3A) was used to establish optimal
cut-off points for prognostic stratification based on nomogram
scores. This analysis identified three distinct prognostic groups
among the AFIP-classified high-risk GIST cohort of the present
study. The favorable-prognosis group included 139 patients with
nomogram scores indicating excellent long-term outcomes and
achieved a 5-year RFS rate of 84.9%. The moderate-prognosis group
comprised 120 patients with moderate risk scores and demonstrated a
5-year RFS rate of 70.4%. Most concerning, the poor-prognosis group
consisted of 10 patients whose nomogram scores predicted markedly
inferior outcomes, with these patients achieving only a 16.7%
5-year RFS rate despite all being AFIP-classified as high-risk and
receiving adjuvant imatinib therapy (Fig. 3B). The survival differences between
these nomogram-based prognostic groups were highly significant
(P<0.0001) (Fig. 3B, Tables VII and VIII).
 | Table VIIPrognostic model discrimination in
the Taiwan GISTs registry dataset. |
Table VII
Prognostic model discrimination in
the Taiwan GISTs registry dataset.
| | Taiwan GISTs
registry |
|---|
| Measures of
discrimination | Estimate | 95% CI |
|---|
| Harrell's
C-index | 0.72 | 0.65-0.79 |
| Gonen and Heller's
K | 0.67 | 0.62-0.72 |
| Royston &
Sauerbrei's D-statistic | 1.19 | 0.77-1.62 |
 | Table VIIISurvival analysis by prognostic
group. |
Table VIII
Survival analysis by prognostic
group.
| Prognostic
group | HR | 95% CI of HR | P-value |
|---|
| Favorable | 1 | | |
| Moderate | 2.63 | 1.52-4.52 | 0.001 |
| Poor | 18.62 | 8.02-43.25 | <0.0001 |
Discussion
The present study evaluated prognostic factors and
long-term outcomes in a large national cohort of 269 patients with
modified AFIP-classified high-risk GIST who received adjuvant
imatinib over a median follow-up period of 62.7 months. A total of
3 independent predictors of unfavorable RFS were identified: Male
sex, non-gastric tumor origin and shorter duration of imatinib
therapy. A nomogram incorporating these clinicopathological
variables demonstrated good predictive accuracy and calibration,
enabling further prognostic stratification within this already
high-risk population into three distinct groups with markedly
different outcomes.
The present study provides specific insights for the
adjuvant therapy setting that complement existing knowledge from
surgical cohorts. An important clarification for readers is that
the entire cohort of the present study consists of patients with
modified AFIP-classified high-risk GIST who received adjuvant
therapy, and the nomogram provides additional prognostic
stratification within this population. The three ‘prognostic
groups’ identified by the nomogram (favorable, moderate and
poor-prognosis) represent further risk refinement among patients
already classified as high-risk by conventional criteria.
The association between male sex and inferior RFS
has been observed in multiple GIST cohorts and warrants further
investigation. This sex-based disparity may reflect biological
differences in tumor behavior, hormonal influences on GIST
pathogenesis, or variations in treatment adherence and tolerance
patterns (23). The underlying
mechanisms driving this observation merit further research to
inform personalized treatment strategies.
The findings of the present study confirm that tumor
location remains a critical prognostic determinant even in the era
of adjuvant imatinib therapy. Non-gastric GISTs, particularly those
arising from the duodenum with a 60.0% recurrence rate and small
bowel with a 28.8% recurrence rate, demonstrated significantly
worse outcomes compared to gastric tumors with a 15.8% recurrence
rate. This observation aligns with established knowledge regarding
the more aggressive biological behavior of non-gastric GISTs and
may reflect underlying molecular differences specific to anatomical
location (24).
Historical studies from the pre-imatinib era
identified KIT exon 11 deletions, particularly those
involving codons 557-558, as harboring adverse prognostic
significance compared to other mutation types. Wozniak et al
(24) previously highlighted the
poor outcomes associated with KIT del557/558 mutations in
gastric GISTs. Furthermore, our group has demonstrated that
KIT exon 11 557-558 deletions promote liver metastasis
through C-X-C motif chemokine receptor 4 upregulation via enhanced
ETV1 promoter binding (25).
Unfortunately, our registry lacked detailed exon-level mutation
data, precluding examination of these specific molecular
correlates. Future studies incorporating comprehensive genotyping
may provide additional prognostic insights and guide preoperative
risk assessment.
The substantial proportion of missing mutation data
(46.8%) in the present cohort reflects the evolution of molecular
testing practices during the study period rather than systematic
study bias. In Taiwan, comprehensive mutation testing transitioned
from research-based to routine clinical practice between 2013 and
2023. While this represents a study limitation, the sensitivity
analyses demonstrate that the nomogram maintains prognostic utility
without molecular data, making it applicable in clinical settings
where comprehensive genetic testing may not be routinely available.
This practical consideration enhances the global applicability of
our prognostic tool.
Notably, tumor rupture was present in 15.6% of
patients but did not emerge as an independent prognostic factor in
the multivariate analysis of the present study, despite being
traditionally considered a high-risk feature requiring adjuvant
therapy. This finding may reflect that all patients with rupture in
our cohort received adjuvant therapy, potentially mitigating its
prognostic impact compared to historical surgical-only cohorts.
Data from the SSG XVIII trial similarly suggest that standard
3-year adjuvant therapy may not adequately address the elevated
recurrence risk associated with tumor rupture, supporting European
Society for Medical Oncology guideline recommendations for extended
imatinib in this subset (5,26).
The present analysis of treatment duration as a
prognostic factor addresses important methodological considerations
about potential reverse causation. While it is acknowledged that
some early discontinuations may result from disease progression,
the landmark analyses at multiple time-points demonstrate that
treatment duration maintains prognostic significance even after
accounting for time-dependent effects. This suggests that treatment
completion, when medically feasible, has important implications for
long-term outcomes, though the complex relationship between
duration and prognosis requires careful clinical interpretation.
The clinical message remains that achieving adequate treatment
duration is associated with better outcomes in patients who can
tolerate therapy.
The nomogram developed herein provides a practical
tool for individualized risk assessment in the adjuvant setting
(10). Its demonstrated
discriminative ability (C-index: 0.72) and clear risk
stratification into three prognostic groups with dramatically
different survival outcomes (84.9, 70.4 and 16.7% 5-year survival
rates) may assist clinicians in patient counseling, surveillance
planning and treatment decision-making.
The present study has several important limitations
that require acknowledging. The retrospective registry design
reflects real-world clinical practice but lacks the standardization
of prospective trials, with clinical practices varying across the
11 participating centers over the 10-year study period. This
variation potentially introduces heterogeneity in treatment
decisions and follow-up protocols that may affect the present
results.
Selection bias represents another significant
limitation, as the present cohort includes only patients with
modified AFIP-classified high-risk GIST who actually received
adjuvant imatinib therapy. This represents a selected subset of the
broader high-risk GIST population, since some high-risk patients do
not receive adjuvant therapy due to age, comorbidities, patient
preference or other factors. The present findings may therefore not
be generalizable to all patients with high-risk GIST, particularly
those who did not receive adjuvant therapy.
The substantial proportion of missing mutation
information (46.8%) represents a significant methodological
limitation. While these missing data reflect the evolution of
molecular testing practices during the study period and the
sensitivity analyses suggest the nomogram maintains utility without
molecular data, the absence of comprehensive genetic information
limits our ability to develop more sophisticated
molecular-integrated prognostic models.
The limited number of patients in certain subgroups,
particularly duodenal tumors (n=15) and the poor-prognosis nomogram
category (n=10), results in wide confidence intervals and limits
the precision of survival estimates for these subsets. While these
numbers reflect the true epidemiological rarity of duodenal GISTs,
which represent only 3-5% of all GISTs (27), they constrain our ability to make
definitive conclusions about these specific populations.
The interpretation of treatment duration as a
prognostic factor requires careful consideration due to the complex
bidirectional relationship between treatment completion and
outcomes. While our landmark analyses suggest that treatment
duration has genuine prognostic significance beyond simple reverse
causation, early discontinuation may reflect both patient factors
and underlying disease biology, making clinical interpretation
nuanced.
Most importantly, external validation in independent
cohorts from different populations and healthcare systems is
essential before routine clinical implementation of our nomogram.
The single-country design and specific healthcare context may limit
generalizability to other populations.
The present findings carry important clinical
implications. The identification of a poor-prognosis subgroup
within AFIP-classified high-risk patients, with only 16.7% 5-year
survival despite standard adjuvant therapy, highlights that current
treatment paradigms may be insufficient for this population. These
patients may benefit from more intensive surveillance protocols,
extended adjuvant therapy or novel therapeutic approaches currently
under investigation. Additionally, the present results emphasize
the importance of long-term follow-up extending beyond conventional
surveillance periods, particularly for patients identified as
having poor prognosis by nomogram assessment.
Future research priorities should include
prospective validation of our nomogram in external cohorts,
investigation of strategies to optimize treatment completion rates
and integration of comprehensive molecular profiling as it becomes
more widely available. Clinical trials evaluating extended adjuvant
therapy duration or novel approaches specifically in
nomogram-identified poor-prognosis patients may help improve
outcomes in this challenging population.
In conclusion, this large retrospective registry
analysis identified male sex, non-gastric tumor origin and shorter
imatinib duration as independent predictors of poor disease-free
survival in patients with modified AFIP high-risk GIST receiving
adjuvant therapy. The resulting nomogram provides a practical tool
for risk stratification, enabling identification of three distinct
prognostic groups with markedly different outcomes. While external
validation is essential before routine clinical implementation,
this prognostic tool may enhance clinical decision-making and
patient counseling in the adjuvant setting. These findings
highlight the need for personalized approaches to surveillance and
treatment planning, particularly for nomogram-identified
poor-prognosis patients who may benefit from intensified monitoring
or extended imatinib adjuvant use.
Supplementary Material
Supplementary Data.
Acknowledgements
Not applicable.
Funding
Funding: This study was sponsored by Taiwan Cooperative Oncology
Group and partially funded by Pfizer.
Availability of data and materials
All data generated or analyzed during the current
study are included in this published article. The DNA mutations
identified in this study have been deposited at ClinVar (https://www.ncbi.nlm.nih.gov/clinvar/submitters/509598).
Authors' contributions
CNY was responsible for the design of the study,
provision of the patients, assembly of the data, data analysis,
interpretation of the results and manuscript writing. LTC and HJT
participated in the conception and design of the study, provision
of the patients, data analysis and interpretation of the results,
and reviewed the manuscript. YSS, CYY, CHT, CCW, DCC and CCY were
responsible for provision of the patients, data analysis and
interpretation of the results, and reviewed the manuscript. MTL,
CFT, THC, YYC, HYL, TSY, CLH, TYS, LYB, JTH and ISC were
responsible for provision of the patients, interpretation of the
results and review of the manuscript. CFH assembled the data,
performed data analysis and interpretation of the results, and
reviewed the manuscript. CNY and HJT checked and confirmed the
authenticity of the raw data. All authors reviewed the manuscript
and have read and approved the final version of the manuscript.
Ethics approval and consent to
participate
The study protocol was reviewed and approved by the
IRB of each participating institution, including National Taiwan
University Hospital Ethics Center Research Ethics Section, IRB of
Taipei Veterans General Hospital, the IRB of Tri-Service General
Hospital, Mackay Memorial Hospital IRB, Chang Gung Medical
Foundation IRB, China Medical University Hospital Research Ethics
Committee, IRB of Changhua Christian Hospital, National Cheng Kung
University Hospital IRB, Kaohsiung Medical University Hospital IRB
and Department of Medical Education and Research Kaohsiung Veterans
General Hospital. All patients provided written informed consent
and those who had died provided consent before they died.
Patient consent for publication
Not applicable.
Competing interests
LTC has received research funding from Pfizer to
National Health Research Institutes for National Registry Study.
All other authors declare that they have no competing
interests.
References
|
1
|
Miettinen M and Lasota J: Gastrointestinal
stromal tumors (GISTs): Definition, occurrence, pathology,
differential diagnosis and molecular genetics. Pol J Pathol.
54:3–24. 2003.PubMed/NCBI
|
|
2
|
Demetri GD, von Mehren M, Antonescu CR,
DeMatteo RP, Ganjoo KN, Maki RG, Pisters PW, Raut CP, Riedel RF,
Schuetze S, et al: NCCN Task Force report: Update on the management
of patients with gastrointestinal stromal tumors. J Natl Compr Canc
Netw. 8 (Suppl 2):S1–S44. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Heinrich MC, Corless CL, Demetri GD,
Blanke CD, von Mehren M, Joensuu H, McGreevey LS, Chen CJ, Van den
Abbeele AD, Druker BJ, et al: Kinase mutations and imatinib
response in patients with metastatic gastrointestinal stromal
tumor. J Clin Oncol. 21:4342–4349. 2003.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Corless CL, Fletcher JA and Heinrich MC:
Biology of gastrointestinal stromal tumors. J Clin Oncol.
22:3813–3825. 2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Joensuu H, Eriksson M, Sundby Hall K,
Hartmann JT, Pink D, Schütte J, Ramadori G, Hohenberger P, Duyster
J, Al-Batran SE, et al: One vs three years of adjuvant imatinib for
operable gastrointestinal stromal tumor: A randomized trial. JAMA.
307:1265–1272. 2012.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Dematteo RP, Ballman KV, Antonescu CR,
Maki RG, Pisters PW, Demetri GD, Blackstein ME, Blanke CD, von
Mehren M, Brennan MF, et al: Adjuvant imatinib mesylate after
resection of localised, primary gastrointestinal stromal tumour: A
randomised, double-blind, placebo-controlled trial. Lancet.
373:1097–1104. 2009.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Casali PG, Le Cesne A, Poveda Velasco A,
et al: Time to definitive failure to the first tyrosine kinase
inhibitor in localized gastrointestinal stromal tumors (GIST)
treated with adjuvant imatinib: A European organisation for
research and treatment of cancer (EORTC) soft tissue and bone
sarcoma group intergroup randomized trial (EORTC 62024). Ann Oncol.
26:399–404. 2015.
|
|
8
|
Raut CP, Espat NJ, Maki RG, Araujo DM,
Trent J, Williams TF, Purkayastha DD and DeMatteo RP: Efficacy and
tolerability of 5-year adjuvant imatinib treatment for patients
with resected intermediate- or high-risk primary gastrointestinal
stromal tumor: The PERSIST-5 clinical trial. JAMA Oncol.
4(e184060)2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Joensuu H, Eriksson M, Sundby Hall K,
Reichardt A, Hermes B, Schütte J, Cameron S, Hohenberger P, Jost
PJ, Al-Batran SE, et al: Survival outcomes associated with 3 years
vs 1 year of adjuvant imatinib for patients with high-risk
gastrointestinal stromal tumors: An analysis of a randomized
clinical trial after 10-year follow-up. JAMA Oncol. 6:1241–1246.
2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chang YY, Huang WK, Wang SY, Wu CE, Chen
JS and Yeh CN: A nomogram predicting progression free survival in
patients with gastrointestinal stromal tumor receiving sunitinib:
Incorporating pre-treatment and post-treatment parameters. Cancers
(Basel). 13(2587)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Liu Q, Kong F, Zhou J, et al: Development
and validation of a nomogram to predict the prognosis of patients
with gastric gastrointestinal stromal tumors. Cancer Med.
9:7216–7227. 2020.
|
|
12
|
Wang M, Xu J, Zhang Y, et al: A practical
prognostic nomogram for predicting early recurrence of
gastrointestinal stromal tumors with intermediate or high risk. Ann
Transl Med. 7(456)2019.
|
|
13
|
Blay JY, Le Cesne A, Ray-Coquard I, et al:
A randomized study of 6 versus 3 years of adjuvant imatinib in
patients with localized GIST at high risk of relapse. Lancet Oncol.
25:e234–e245. 2024.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Blay JY, Schiffler C, Bouché O, Brahmi M,
Duffaud F, Toulmonde M, Landi B, Lahlou W, Pannier D, Bompas E, et
al: A randomized study of 6 versus 3 years of adjuvant imatinib in
patients with localized GIST at high risk of relapse. Ann Oncol.
35:1157–1168. 2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Miettinen M and Lasota J: Gastrointestinal
stromal tumors: Pathology and prognosis at different sites. Semin
Diagn Pathol. 23:70–83. 2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Tsai HJ, Shan YS, Yang CY, Hsiao CF, Tsai
CH, Wang CC, Lin MT, Ting CF, Chan DC, Chen TH, et al: Survival of
advanced/recurrent gastrointestinal stromal tumors treated with
tyrosine kinase inhibitors in Taiwan: A nationwide registry study.
BMC Cancer. 24(828)2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Rutkowski P, Ziętek M, Cybulska-Stopa B,
Streb J, Głuszek S, Jankowski M, Łopacka-Szatan K, Las-Jankowska M,
Hudziec P, Klimczak A, et al: The analysis of 3-year adjuvant
therapy with imatinib in patients with high-risk molecular profiled
gastrointestinal stromal tumors (GIST) treated in routine practice.
Eur J Surg Oncol. 47:1191–1195. 2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Joensuu H: Risk stratification of patients
diagnosed with gastrointestinal stromal tumor. Hum Pathol.
39:1411–1419. 2008.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Yeh CN, Chen TW, Lee HL, Liu YY, Chao TC,
Hwang TL, Jan YY and Chen MF: Kinase mutations and imatinib
mesylate response for 64 Taiwanese with advanced GIST: Preliminary
experience from Chang Gung Memorial Hospital. Ann. Surg Oncol.
14:1123–1128. 2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yeh CN, Chen YY, Tseng JH, Chen JS, Chen
TW, Tsai CY, Cheng CT, Jan YY and Chen MF: Imatinib mesylate for
patients with recurrent or metastatic gastrointestinal stromal
tumors expressing KIT: A decade experience from Taiwan. Transl
Oncol. 4:328–335. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Tien YW, Lee CY, Huang CC, Hu RH and Lee
PH: Surgery for gastrointestinal stromal tumors of the duodenum.
Ann Surg Onco. 17:109–114. 2010.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Tanadini LG, Steeves JD, Hothorn T, Abel
R, Maier D, Schubert M, Weidner N, Rupp R and Curt A: Identifying
homogeneous subgroups in neurological disorders: Unbiased recursive
partitioning in cervical complete spinal cord injury. Neurorehabil.
Neural Repair. 28:507–515. 2014.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ran P, Li J, Wu X, Yang H and Zhang J:
Primary localized gastrointestinal stromal tumors: Medication
adherence and prognosis according to gender. Patient Prefer
Adherence. 16:2077–2087. 2022.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Wozniak A, Rutkowski P, Schöffski P,
Ray-Coquard I, Hostein I, Schildhaus HU, Le Cesne A, Bylina E,
Limon J, Blay JY, et al: Tumor genotype is an independent
prognostic factor in primary gastrointestinal stromal tumors of
gastric origin: A european multicenter analysis based on
ConticaGIST. Clin Cancer Res. 20:6105–6116. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wang HC, Li TY, Chao YJ, Hou YC, Hsueh YS,
Hsu KH and Shan YS: KIT exon 11 codons 557-558 deletion mutation
promotes liver metastasis through the CXCL12/CXCR4 axis in
gastrointestinal stromal tumors. Clin Cancer Res. 22:3477–3487.
2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Nishida T, Hølmebakk T, Raut CP and
Rutkowski P: Defining tumor rupture in gastrointestinal stromal
tumor. Ann Surg Oncol. 26:1669–1675. 2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Popivanov G, Tabakov M, Mantese G,
Cirocchi R, Piccinini I, D'Andrea V, Covarelli P, Boselli C,
Barberini F, Tabola R, et al: Surgical treatment of
gastrointestinal stromal tumors of the duodenum: A literature
review. Transl Gastroenterol Hepatol. 3(71)2018.PubMed/NCBI View Article : Google Scholar
|