1. Introduction
Garlic (Allium sativum) is widely
acknowledged for its notable antiviral properties, primarily
derived from organosulfur components such as allicin (1-3).
These bioactive agents such as diallyl disulfide (DADS), diallyl
trisulfide (DATS) and ajoene have been found to suppress viral
replication and strengthen the immune response of the host
(1). Therefore, garlic may be a
useful adjunct in managing respiratory viral infections,
particularly in mitigating virus-induced oxidative stress (1,2,4,5).
Current antiviral drugs, including nucleoside analogs (such as
acyclovir for herpes viruses and oseltamivir for influenza) and
protease inhibitors (such as ritonavir for HIV), exhibit certain
disadvantages, including limited effectiveness against emerging
viral strains and the development of drug-resistant viruses
(6-8).
This positions natural options such as garlic as a promising
direction for further research and highlights that the development
of novel antiviral drugs is key, especially during pandemics, as
the coronavirus disease 19 (COVID-19) crisis has shown. Studies
have demonstrated that garlic-derived compounds can inhibit severe
acute respiratory syndrome coronavirus 2 (SARS-CoV-2) replication
in vitro (9,10), block viral entry into host cells by
interfering with the spike protein-angiotensin-converting enzyme 2
receptor interaction (11), and
reduce virus-induced inflammation by suppressing pro-inflammatory
cytokines such as IL-6 and IL-8(12). Furthermore, a randomized controlled
trial suggested that garlic extract as adjunctive therapy may
reduce the need for supplemental oxygen in hospitalized patients
with COVID-19(13). These findings
underscore the relevance of exploring garlic-based interventions in
the context of emerging viral pandemics.
Over the past century, natural products have been a
primary source for drug development, with 60-80% of antibiotics and
anticancer drugs being derived from natural products or their
derivatives (14). Garlic is known
for its diverse pharmacological effects, such as antibacterial,
anti-inflammatory and antiviral activities (15). The antiviral capacity of this plant
is mainly associated with organosulfur constituents, including
DATS, ajoene and allicin (16).
The latter, a sulfur-rich bioactive molecule, is produced upon
crushing garlic through the enzymatic action of alliinase (17). Allicin exhibits a range of
biological activities, including antimicrobial effects,
immunomodulatory actions (such as enhancing natural killer cell
activity, activating macrophages and regulating cytokine
production) and direct antiviral effects (18-20).
These antiviral functions are mediated through its ability to
obstruct viral entry into host cells, suppress viral replication
and modulate host immune responses to fight infection (21).
The pharmacokinetic profile of allicin demonstrates
its efficient absorption and rapid metabolism, underpinning its
potential as an antiviral agent (22,23).
However, its use in medicine is limited as it breaks down easily
and exhibits a strong smell, which has prompted a search for
improved delivery methods (24).
In response, research has shifted toward allicin-loaded
nano-carriers designed to boost its bioavailability and shelf-life
(23). Encapsulation of allicin in
nanoparticles improves targeted delivery to infection sites,
markedly strengthening its antiviral efficacy and reducing
non-specific toxicity (23,25).
These innovative systems improve the solubility and cellular
permeability of allicin, enabling controlled release and expanding
its potential applications in antiviral therapy, including enhanced
targeting to infection sites and prolonged therapeutic activity
(23,26).
Preclinical and virological investigations have
revealed the broad-spectrum antiviral potential of allicin and
other garlic-derived components, including ajoene, DATS, DADS,
saponins and garlic extracts, as summarized in Tables I and II (1,21,27-37).
In addition, to represent a translational perspective, Table III summarizes key human clinical
studies involving garlic-based interventions for infection-related
conditions (20,34,37-43).
Against enveloped viruses such as influenza, allicin has been shown
to disrupt viral integrity by binding to and oxidizing thiol groups
in viral envelope proteins, thereby inhibiting viral entry into
host cells (1). In studies
conducted on the common rhinovirus pathogen, allicin and garlic
extract have been shown to reduce viral RNA load, suppress
virus-induced pro-inflammatory cytokine release (IL-6 and IL-8) and
prevent epithelial barrier dysfunction in human bronchial
epithelial cells (3,21). Furthermore, research regarding the
hepatitis C virus model has highlighted the ability of allicin and
its derivatives to modulate the cellular redox state and influence
signaling pathways involved in antiviral defense (18,44).
Allicin also exhibits marked antiviral activity against influenza A
virus by effectively inhibiting viral replication by targeting and
disrupting viral neuraminidase protein function, which is key in
the release of new viral particles from infected cells (45). Finally, allicin has been shown to
exhibit direct virucidal effects, meaning it can inactivate virus
particles upon contact. This occurs primarily through binding to
and oxidizing thiol groups in viral surface proteins (such as the
hemagglutinin of influenza virus and envelope glycoproteins of
herpes viruses), leading to protein denaturation and loss of viral
infectivity (21,46).
 | Table IIn vitro studies on the
potential antiviral effects and mechanisms of garlic-derived
components. |
Table I
In vitro studies on the
potential antiviral effects and mechanisms of garlic-derived
components.
| First author/s,
year | Bioactive
compound | Virus studied and
model | Key findings | Proposed mechanism
of action | (Refs.) |
|---|
| Klenk et al,
2008 | Allicin | Influenza A (H1N1,
H3N2 and H9N2); MDCK cells | Markedly reduced
viral titers and CPE. Showed direct virucidal activity. | Direct virucidal
effect: Disrupted the integrity of the viral envelope and key
proteins. Inhibited viral replication: May interfere with viral
polymerase function. Immuno-modulation: Induced expression of
anti-viral cytokines. | (31) |
| Petrovska and
Cekovska, 2010; Jakobsen et al, 2012 | Ajoene | HCMV; HHV-1; MRC-5
cell | Potently inhibited
viral replication. Showed synergistic effects with standard
antivirals (ganciclovir and acyclovir). | Blocked viral entry
and cell-cell transmission, primarily by targeting the
integrin/NF-κB signaling pathway. Inhibited late stages of
replication such as viral assembly and egress. Induced cellular
autophagy: This autophagic response was | (27,28) |
| Tsai et al,
1985; Borlinghaus et al, 2021 | DATS/DADS | HCV; Huh7.5 cells;
influenza virus; MDCK cells | Suppressed HCV RNA
replication and infectious particle production. Protected cells
from influenza-induced CPE. | harnessed to
degrade viral components. Induced HO-1 expression and activity:
This antioxidant enzyme created an anti-viral state in the cell.
Direct virucidal activity. | (29,30) |
| Rouf et al,
2020 | Allicin and garlic
extract | HRV; human
bronchial epithelial cells (BEAS-2B) | Reduced viral RNA
load and cytokine release (IL-6 and IL-8). Prevented virus-induced
barrier dysfunction. | Immunomodulation:
Suppressed virus-induced pro-inflammatory responses and oxidative
stress. Preserved epithelial barrier integrity by preventing tight
junction disruption. | (1) |
| Thuy et al,
2020 | Garlic extract | SARS-CoV-2; Vero E6
cells | Showed
dose-dependent inhibition of SARS-CoV-2 replication. | Direct virucidal
activity: Incubation with the extract inactivated viral particles.
Inhibited viral entry: May interfere with spike protein binding to
the ACE2 receptor. | (32) |
| Tatarintsev et
al, 1992 | Saponins (from
garlic) | HSV-1 and HSV-2;
Vero cells | Exhibited strong
inhibition of viral adsorption and penetration into host
cells. | Direct virucidal
effect: Interacted with the viral envelope, destabilizing it.
Blocked viral attachment: May compete with the virus for host cell
binding sites. | (33) |
 | Table IIPotential antiviral effects and
mechanisms of garlic-derived components based on in vivo
studies. |
Table II
Potential antiviral effects and
mechanisms of garlic-derived components based on in vivo
studies.
| First author/s,
year | Garlic-derived
preparation | Virus | Study model | Key antiviral
effects | Proposed
mechanisms | (Refs.) |
|---|
| Nantz et al,
2012 | AGE | Influenza A (H1N1)
virus | Mice | Reduced mortality;
less severe lung pathology; decreased lung viral titers. | Enhanced cellular
immunity: Increased activation and cytolytic activity of NK cells
and γδ-T cells in the lungs. Immunomodulation: Modulated cytokine
production. | (34) |
| Guo et al,
1993 | Allicin | CMV | Mice | Increased survival
rate; reduced viral load in the salivary glands; protected against
splenic atrophy. | Immunoenhancement:
Reversed the virus-induced suppression of NK cell activity.
Modulated cytokine expression (increased IL-2 and IFN-γ expression,
as well as decreased IL-10 expression). | (35) |
| Weber et al,
1992 | Ajoene (a stable
derivative) | Influenza A (H1N1)
virus | Mice | Reduced lung virus
titers | Altered viral
entry: Shown in vitro to interfere with the early stage of
the viral life cycle, potentially by disrupting viral fusion. The
in vivo effect is perhaps a combination of direct antiviral
action and immunomodulation. | (21) |
| Fadlalla and
Bakhiet, 2010 | Fresh garlic clove
extract | NDV | Chickens | Enhanced humoral
immune response; increased antibody titers against NDV
post-vaccination. | Immunomodulation:
Acting as an immunostimulant/adjuvant, potentiating the immune
response to vaccines. | (36) |
| Choi and Park,
2012 | Garlic essential
oil | PRRSV | Pigs | Improved average
daily weight gain; reduced lung lesions and viral load in
serum. | Immunomodulation:
Increased CD4+/CD8+ T cell ratio; modulation
of cytokine production. | (37) |
 | Table IIISummary of RCTs investigating
garlic-based interventions in viral infection management. |
Table III
Summary of RCTs investigating
garlic-based interventions in viral infection management.
| First author/s,
year | Design and
population | Garlic
intervention | Control | Key findings | Limitations | (Refs.) |
|---|
| Josling, 2001 | RCT, parallel
groups; 146 healthy adults (mean age, 52 years) | Garlic supplement
(allicin), 1 capsule/day for 8 days | Placebo | Marked reduction in
cold/flu incidence (24 vs. 65 cases). Shorter symptom duration in
garlic group. | Self-reported
symptoms. Whole garlic extract, not purified allicin. | (49) |
| Andrianova et
al, 2003 | Two-phase RCT;
phase I: 640 children (6-17 years); phase II: 115 children (10-12
years) | Garlic tablets,
300-600 mg/day for 150 days | Placebo | Phase II: 50% lower
risk of ARVI (RR=0.50). Milder disease course in garlic group | Pediatric
population only. Sustained-release formulation used. | (38) |
| Nantz et al,
2012; Percival, 2016 | RCT, parallel
groups; 120 healthy adults (mean age, 26 years) | AGE, 2.56 g/day for
90 days | Placebo | A 61% reduction in
cold/flu severity. Enhanced NK cell and γδ-T cell function. | Immune markers
measured; no viral load data. | (20,34) |
| Kenawy et
al, 2014 | RCT, parallel
groups; 50 patients with warts (mean age, 25 years) | LGE, applied
topically twice daily for 28 days | Saline | A 96% complete
clearance of warts. Increased serum TNF-α levels. | Non-randomized
design in part. Small sample size. | (39) |
| Lee et al,
2012 | RCT, 4-arm
dose-response; 79 patients with chronic hepatitis | DDB and garlic oil
capsules (25 + 50 mg), 2-6 caps/day for 42 days | Placebo | Decreased ALT/AST
levels. Reduced HBV DNA levels in combination group. | Open-label design.
Garlic oil composition variable. | (40) |
| Hiltunen et
al, 2007 | RCT, parallel
groups; 52 patients (mean age, 38 years) | Intranasal
cellulose and PGE spray, once daily per nostril | Cellulose only | Lower respiratory
infection incidence (OR=0.30). | Physical barrier
mechanism, not direct antiviral. | (41) |
| Mousavi et
al, 2018 | RCT, split-body
design; 35 men with genital warts (mean age, 33 years) | 10% garlic extract,
topical application vs. cryotherapy | PEG base | Garlic: 93%
clearance vs. cryotherapy: 73% clearance. Lower recurrence with
garlic. | Split-body design
limits blinding. | (42) |
|
Gołebiowska-Wawrzyniak et al,
2005 | RCT, parallel
groups; 30 immunodeficient children (3-15 years) | Inosiplex and
garlic extract (50 mg/kg/day), for 10 days | Garlic extract
only | Enhanced
CD3+/CD4+ T cell counts Clinical improvement
in viral infections. | Small sample size.
Combined immuno-modulator intervention. | (43) |
Previous research has broadened the general
understanding of the therapeutic potential of allicin beyond its
direct antiviral activity. Investigations have been increasingly
exploring the synergistic effects between allicin and conventional
antiviral drugs to enhance treatment efficacy and combat the
emergence of drug-resistant viral strains (33,47,48).
Furthermore, a number of epidemiological and dietary studies have
indicated that regular intake of garlic, a primary source of
allicin, may be associated with a lower incidence and severity of
viral respiratory infections, including the common cold (primarily
caused by rhinoviruses, coronaviruses and respiratory syncytial
virus) and influenza (34,49,50).
Due to its multi-faceted mechanism of action and favorable safety
profile, allicin represents a compelling candidate for use in
complementary antiviral regimens. However, comprehensive clinical
trials are necessary to determine its safety, optimal dosing and
long-term benefits in therapeutic applications.
The present review aims to summarize the latest
evidence on the clinical potential of allicin in antiviral therapy,
discussing progress in novel delivery systems and synergistic drug
combinations, and outlining future directions for developing
allicin-based antiviral strategies. With continued investigation,
allicin is poised to become a valuable natural therapeutic agent
for improving the management of viral infections.
2. Methods
In the present study, literature regarding the
antiviral traits and delivery methods of allicin was systematically
searched, concentrating on three areas: i) The compound itself
(‘allicin’ and ‘garlic extract’); ii) its biological activity
(‘antiviral’ and ‘virus inhibition’); and iii) its technological
applications (‘nanoparticle’ and ‘drug delivery’). Comprehensive
searches were conducted across the PubMed (https://pubmed.ncbi.nlm.nih.gov/), Embase (www.embase.com) and Web of Science (https://www.webofscience.com) databases. The reference
lists of the identified articles were also manually checked to
ensure that all relevant studies were included. Original research
articles investigating the antiviral activity, mechanisms, delivery
systems and pharmacokinetics of allicin were included, while
non-English publications, studies lacking allicin characterization
and non-research articles (such as editorials) were excluded.
Priority was given to studies directly addressing the antiviral
mechanisms or advanced formulations of allicin, while relevant
reviews and foundational papers were also considered to provide
broader context.
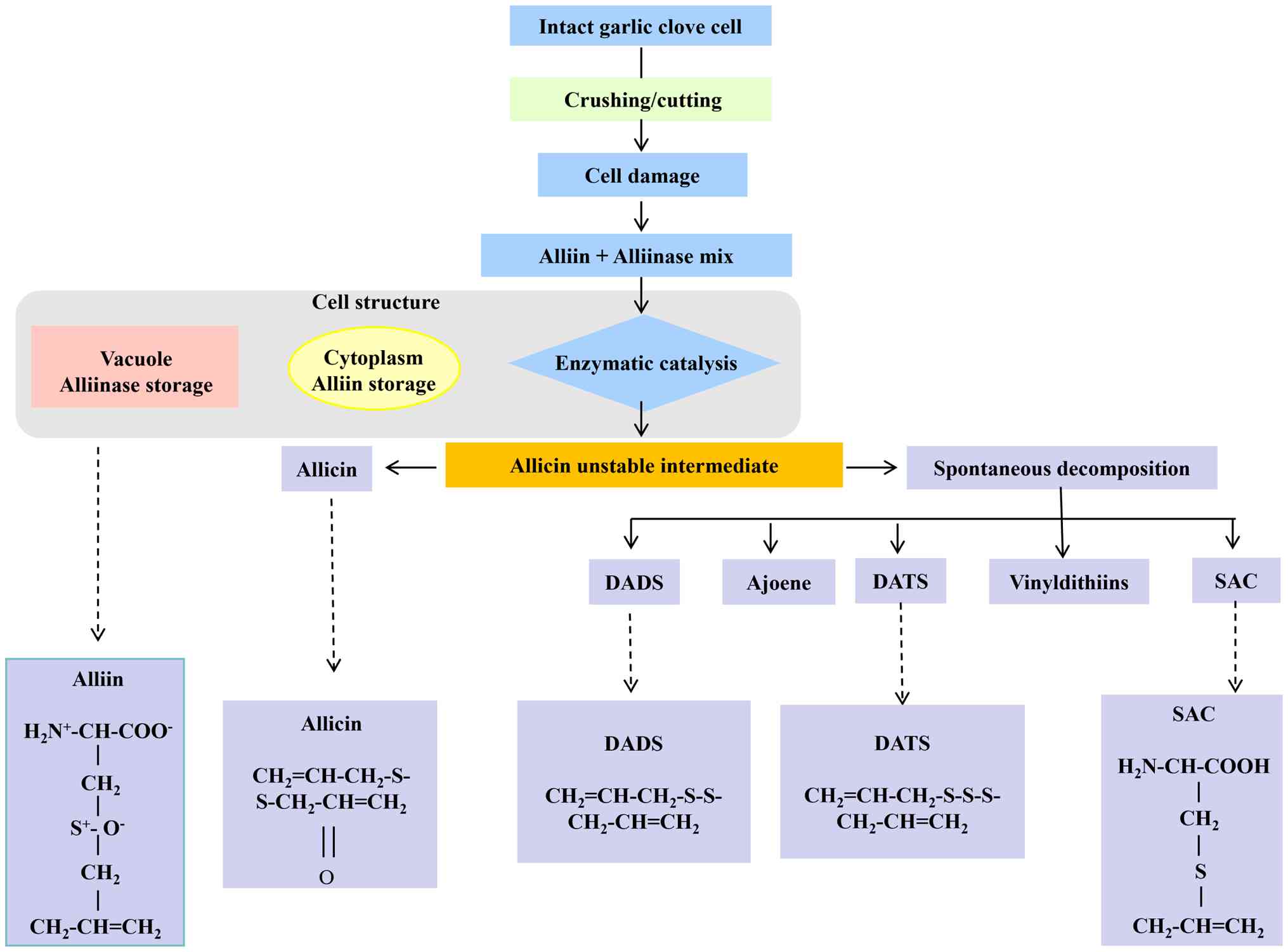
3. Chemistry and pharmacokinetics
Allicin (diallyl thiosulfinate), the principal
bioactive agent derived from garlic, is not intrinsically present
in intact cloves. Its formation is triggered by mechanical
disruption (crushing or cutting), which initiates an enzymatic
process mediated by alliinase (51). This enzyme catalyzes the conversion
of the stable precursor alliin (S-allyl-L-cysteine sulfoxide) into
allyl sulfenic acid. Subsequently, two molecules of allyl sulfenic
acid spontaneously condense to form allicin, a reaction that occurs
almost instantaneously and releases the characteristic pungent odor
of fresh garlic (52). Chemically,
allicin is defined by a thiosulfinate group
(S+-S-) and two allyl moieties, with the
molecular formula C6H10OS2
(52). A key challenge in its
therapeutic use, particularly in antiviral applications, is its
inherent chemical instability (53). Allicin is highly reactive and
rapidly degrades into a number of oil-soluble sulfur compounds,
including ajoene, dithiins and diallyl polysulfides (54). This underscores the critical need
for advanced stabilization and delivery strategies to fully harness
the antiviral potential of allicin.
The pharmacokinetic profile of allicin is
characterized by rapid absorption and extensive metabolism. Upon
oral administration, it is readily absorbed from the
gastrointestinal tract; however, its high reactivity leads to rapid
metabolic conversion (51). Given
that allicin is metabolized quickly and extensively before it
enters the bloodstream, very little of the ingested dose reaches
systemic circulation in its intact form. Lawson and Hunsaker
(55) reported that the relative
bioavailability of allicin from garlic supplements ranges from 36
to 111% depending on formulation, with values >100% indicating
higher absorption than the reference standard used in the study. A
radiolabeling study in rats estimated ≥65% absorption of
allicin-derived material, although unchanged allicin was absent
from urine, confirming extensive presystemic metabolism (56). This low and unpredictable
bioavailability is a key barrier to its use as a medicine (51). Evidence has indicated that allicin
can permeate cell membranes to exert its biological effects,
including intracellular antiviral actions, within infected host
cells (57). A notable challenge
in pharmacokinetic analysis is the rapid decomposition of the
compound, which has limited the development of reliable methods for
its direct quantification in human plasma (22). Allicin is not present in intact
garlic cloves but is formed upon tissue disruption. Within the
intact garlic clove, the stable precursor alliin is stored in the
cytoplasm, while the enzyme alliinase is compartmentalized within
vacuoles (58,59). Mechanical damage from crushing or
cutting brings these components into contact, initiating enzymatic
catalysis. Alliinase rapidly converts alliin to allyl sulfenic
acid, which spontaneously condenses to form allicin (46,60).
Allicin itself is an unstable intermediate that undergoes
spontaneous decomposition into various bioactive sulfur-containing
metabolites, including DADS, DATS, ajoene, vinyldithiins and
S-allyl-cysteine (18,61,62).
These metabolites, particularly DADS, DATS, ajoene and
vinyldithiins, are considered to markedly contribute to the
observed antiviral effects of garlic-derived preparations (1) (Fig.
1). Investigations in rodent models have demonstrated that
these metabolites, particularly the lipophilic 1,2-vinyldithiin,
exhibit prolonged detectability in serum and tissues such as fat
and kidneys for ≤24 h, suggesting a potential for sustained
antiviral activity (56,63).
Following its rapid metabolism, primary derivatives
of allicin, including DADS and allyl methyl sulfide (AMS), enter
systemic circulation (64). These
sulfur-bearing metabolites are subsequently processed in the liver
through phase I and II metabolic reactions, such as oxidation and
glucuronidation, being ultimately eliminated through renal
excretion and exhalation (63).
Distribution studies utilizing radiolabeling techniques have
revealed that these organosulfur compounds accumulate in a number
of key tissues, including the liver, lung parenchyma and
respiratory tract mucosa, exhibiting a particular affinity for
cartilage-rich areas (56,63). The preferential accumulation of
allicin-derived compounds in respiratory tissues supports their
potential use against respiratory viral infections (56).
Research has indicated that allicin can modulate key
drug metabolism pathways, primarily through interactions with
cytochrome P450 (CYP) enzymes. Specifically, allicin has been shown
to inhibit the activity of CYP2C19, a mechanism that could
potentially elevate systemic levels of co-administered antiviral
drugs metabolized by this enzyme, including nelfinavir (an HIV
protease inhibitor) and certain anti-hepatitis C virus agents such
as dasabuvir (65,66). By contrast, allicin exhibits
minimal inhibitory activity against CYP3A4, indicating a degree of
enzymatic selectivity in its drug interaction profile (65,66).
Understanding these inhibitory effects is key in predicting and
managing potential herb-drug interactions in patients receiving
allicin-containing supplements alongside specific antiviral
regimens that are substrates for CYP2C19.
Clinical investigations into garlic supplements have
revealed notable variability in the oral bioavailability of
allicin, a key determinant for its systemic antiviral efficacy
(55,62,67).
This variability is heavily influenced by pharmaceutical
formulation and concomitant food intake. For example, the
bioavailability from enteric-coated preparations can vary, but is
markedly diminished when administered with high-protein meals,
perhaps due to prolonged gastric retention (62). Collectively, these factors
contribute to the overarching challenge of achieving predictable
and effective systemic concentrations of intact allicin following
oral administration, severely constraining its reliable clinical
translation (68). The
pharmacokinetic profile and systemic exposure of allicin-derived
metabolites are commonly monitored by quantifying AMS in exhaled
breath and urine, serving as a practical surrogate for assessing
the handling of these bioactive compounds by the body (62). Understanding how formulation, food
intake and metabolic conversion affect allicin bioavailability is
essential for designing dosing regimens that achieve
therapeutically relevant concentrations in vivo (55,67).
The metabolic fate of allicin involves its rapid
conversion into numerous organosulfur metabolites, which can be
detected in biological fluids and exhaled breath, providing
insights into its systemic distribution and bioavailability
(62). Both animal and human
studies have determined that allicin is unstable and rapidly
metabolized, leading to poor bioavailability (55,56,67,69).
To overcome the inherent instability of allicin, research efforts
have been directed toward advanced delivery platforms.
Nanotechnology has emerged as an important strategy to address
these limitations, leading to the development of
allicin-encapsulating nanoparticles, liposomes and other carrier
systems. These advanced delivery platforms are designed to shield
the labile compound from premature degradation and facilitate its
targeted delivery to sites of viral infection in a therapeutically
active form (23,26). For example, encapsulating allicin
within chitosan-based nanocarriers has been shown to enhance its
stability, enable a sustained release profile, and consequently,
augment its in vivo antiviral efficacy in preclinical
studies (70-72).
Parallel strategies include prodrug designs, where stable
biosynthetic precursors are administered and subsequently converted
into active allicin in situ (73-75).
These innovative approaches collectively address the primary
pharmacokinetic limitations of allicin, namely, its instability,
low oral bioavailability and pungent odor, thereby refining its
profile as a more viable candidate for clinical development against
viral pathogens.
4. Nano-delivery strategies to overcome the
limitations of allicin
Although allicin exhibits strong antiviral activity
in laboratory studies, its clinical development is limited by
physicochemical hurdles, including instability and poor
bioavailability (55,56). The extreme chemical instability of
the compound in biological environments, coupled with rapid
metabolism and poor bioavailability, severely limits its
therapeutic utility (23,24,76).
Nanotechnology offers a promising solution to close this divide
between laboratory results and real-world medical use (77,78).
Researchers have been developing sophisticated nanoscale delivery
systems to protect allicin from degradation, control its release
kinetics, enhance its solubility and bioavailability, and
potentially direct it towards specific sites of viral infection,
thereby maximizing therapeutic efficacy while minimizing systemic
side effects (23,24,75,79).
Design and fabrication of nanocarriers
for allicin
Selection of the right nanocarrier is key, and
depends on both how the drug will be administered and on the
properties of allicin itself. A number of systems have shown
potential. Lipid nanoparticles, such as solid lipid nanoparticles
(SLNs) and nanostructured lipid carriers, use body-friendly lipids
to create a protective shell surrounding the water-repelling
allicin (26,80). This shield prevents degradation and
allows the drug to be released slowly over time (81). Liposomal structures, characterized
by their dual-layer phospholipid composition, can harbor allicin
within their lipid membranes, isolating it from aqueous biological
environments (23). Among the
polymeric options, biodegradable poly(lactic-co-glycolic acid)
(PLGA) particles permit fine-tuned release kinetics by adjusting
polymer properties, such as molecular weight, lactic-to-glycolic
acid ratio and copolymer composition, making them ideal for
extended therapeutic action (82,83).
Chitosan-based nanoparticles adhere well to mucosal surfaces and
help drugs penetrate tissues. This makes them particularly
promising for treating respiratory viral infections (84,85).
Nanoemulsions have also been studied to improve how well allicin
dissolves and is absorbed in the gut (24,86).
Production, analysis and stability
enhancement
Development of these nanoformulations requires
reliable production techniques and detailed physical
characterization. Standard methods comprise high-shear
homogenization for lipid-based carriers, solvent evaporation
processes for polymer-based systems and high-energy dispersion for
nanoemulsions (81,87). Detailed analysis is key as it
ensures the product is consistent and helps predict how it will
perform in the body. Important parameters to consider include
particle size and size distribution, which determine circulation
time in the bloodstream, cellular uptake efficiency and
biodistribution, with an optimal size range of 80 to 200 nm
typically reported for intravenous delivery (81,88).
Surface electrical properties (ζ potential) influence particle
stability and their engagement with biological surfaces (84). The most important measures include
encapsulation success and drug payload, which indicate how
effectively allicin is incorporated. Studies have shown that
allicin in SLNs can achieve encapsulation efficiencies >75%.
This greatly improves its stability, protecting the majority of the
compound in gut-like conditions where free allicin would otherwise
break down quickly (26,86).
Enhanced antiviral performance of
nanoformulated allicin
Comparative studies across in vitro and in
vivo models have robustly demonstrated the superior antiviral
efficacy of nano-encapsulated allicin compared with its free form
(77,89). This improvement is primarily
attributed to the protective effect of nanocarriers on the fragile
allicin molecule. For example, engineered delivery systems, such as
whey protein isolate/chitosan (WPI/CS) complexes and zein-sodium
caseinate nanoparticles, have been shown to markedly enhance the
stability and encapsulation efficiency of allicin, creating a
reservoir of the active compound shielded against rapid degradation
in physiological environments (90,91).
This protection directly translates into stronger and more
sustained intracellular antiviral activity (92).
In vitro evidence has further demonstrated
this advantage. In nanomedicine, it is well-established that
encapsulation can notably improve the cellular uptake and
bioavailability of poorly soluble or unstable drugs, thereby
enhancing their pharmacological activity (93-96).
This principle has been validated across numerous drug classes,
including antitumor agents and antibiotics (97). Furthermore, the potential of
nano-encapsulation to boost the antiviral potency of bioactive
compounds such as allicin is supported by precedents with other
molecules. For example, nanoformulations have shown efficacy in
overcoming the poor solubility and stability of curcumin and
resveratrol, leading to marked improvements in their in
vitro and in vivo bioactivity (98). This evidence provides a compelling
rationale for applying analogous strategies for allicin. The
principles demonstrated by WPI/CS and zein-caseinate systems,
including efficient encapsulation, protection from degradation and
controlled release, underpin these outcomes, as stable
encapsulation is a prerequisite for sustained intracellular
delivery and action (90,91).
These laboratory-based advantages, including
enhanced stability, prolonged circulation and improved cellular
uptake, have been consistently corroborated in animal infection
models (78). The improved in
vivo outcomes of nanoformulated allicin are attributed to the
enhanced permeability and retention (EPR) effect, a passive
targeting mechanism whereby nanoparticles accumulate selectively at
sites of inflammation or infection due to leaky vasculature and
impaired lymphatic drainage (99,100). This EPR-like phenomenon has been
directly demonstrated in infectious disease models, including
tuberculosis granulomas (101),
confirming its relevance for targeted antimicrobial delivery
(102). As a key principle in
nanoparticle drug delivery to diseased sites, the EPR effect, a
well-documented concept in tumor biology and infectious
inflammation, facilitates the passive accumulation of nano-sized
carriers at sites with leaky vasculature (101-104).
This principle has been successfully leveraged to improve the
delivery to sites of infection and inflammation and efficacy of
numerous nanomedicines, including liposomal amphotericin B
(AmBisome®) for fungal infections, liposomal antibiotics
for intracellular bacteria and polymeric nanoparticles for
tuberculosis treatment (105-107).
Therefore, designing allicin nanoformulations represents a logical
approach to enhance its targeting and accumulation at viral
infection sites. The improved therapeutic results can be attributed
to two main factors: i) The nanocarriers stabilize allicin during
systemic circulation; and ii) they utilize mechanisms such as the
EPR effect to promote its accumulation at the site of infection
(90,101).
Improved pharmacokinetics and tissue
distribution
Nanocarriers effectively address the core
pharmacokinetic challenge of allicin, namely its low oral
bioavailability, by enhancing its stability and absorption
(23,79). Nanoparticulate systems have been
demonstrated to enhance the oral bioavailability of challenging
drugs by protecting them from gastrointestinal degradation and
facilitating absorption through lymphatic transport (108). This strategy has been extensively
validated, including through the use of curcumin, the oral
bioavailability and therapeutic efficacy of which have been
increased by numerous orders of magnitude through
nanoencapsulation, markedly transforming its translational
potential (109-111).
Such successful case studies form a compelling precedent for
addressing the analogous delivery challenges posed by allicin
(23,79,112). Specifically, encapsulating
allicin within delivery systems such as nanoemulsions has been
shown to markedly enhance its intestinal absorption and systemic
exposure levels (1). This
improvement in bioavailability is key in realizing the full
spectrum of documented health benefits of allicin, including its
antimicrobial and antioxidant properties (23).
With regard to systemic administration, the use of
nanocarriers, particularly those coated with polyethylene glycol
(PEG), is an effective strategy to prolong the circulation time and
favorably alter pharmacokinetic profiles, a concept well-documented
in the literature (113-116).
PEGylation is a well-established technique to evade immune
clearance, as evidenced by its success in numerous U.S. Food and
Drug Administration-approved nanomedicines, including
Doxil® (pegylated liposomal doxorubicin),
Oncaspar® (pegylated L-asparaginase),
Neulasta® (pegylated granulocyte colony-stimulating
factor), Pegasys® (pegylated interferon alfa-2a) and
Cimzia® (pegylated antibody fragment) (117-119).
Consistent with this, allicin packaged in PLGA nanoparticles
exhibits prolonged detectability and increased systemic exposure
compared with the free molecule, with studies reporting an up to
4.2-fold higher area under the curve and extended half-life for
various drugs encapsulated in PLGA nanoparticles (120,121).
Beyond enhancing systemic levels, nanocarriers can
redirect drug distribution within the body. Nanoparticles can be
engineered to achieve modified biodistribution, leading to
increased drug accumulation in target tissues such as the liver,
spleen and sites of inflammation or infection, thereby improving
the therapeutic index (122).
Active targeting strategies employing specific ligands further
validate the ability to direct nanoformulations to particular cell
types, a principle readily applicable to targeting infected tissues
(123). Investigations using
tracking methods have shown that nanoparticles can promote the
preferential accumulation of allicin in organs with active immune
activity or altered vascular permeability, such as the lungs and
liver, which are common sites of viral pathology (87,124,125). This redirected distribution not
only elevates drug concentrations at sites of infection but also
reduces the risk of off-target effects, including toxicity to
healthy tissues such as the heart, kidneys and bone marrow, as well
as minimizing non-specific immune activation and inflammation
(126,127).
Conclusions
In conclusion, the strategic application of
nanoscale drug delivery technologies represents a marked
advancement in elucidating the therapeutic potential of allicin
(79). Encapsulation of this
chemically unstable compound within rationally designed
nanocarriers has enabled improved stabilization, enhanced efficacy,
and optimized pharmacokinetic behavior and favorable tissue
targeting (71,128). Collectively, current evidence
indicates that nanodelivery systems constitute a key strategy in
transforming allicin from a promising natural compound into a
clinically viable therapeutic agent (23,79).
Future research is expected to focus on the development of more
sophisticated delivery platforms, including stimulus-responsive
nanocarriers capable of releasing allicin selectively under
pathological conditions such as reduced pH, as well as actively
targeted systems functionalized with ligands that recognize
virus-infected cells (128). The
continued integration of advanced materials engineering with the
broad-spectrum antiviral properties of allicin holds promise for
the development of novel effective antiviral therapies.
5. Allicin as a natural antiviral
therapeutic agent
Viral infections continue to pose a notable and
evolving threat to global public health, with both emerging and
re-emerging pathogens contributing to morbidity and mortality
worldwide (44). The limitations
of existing antiviral therapies, including narrow-spectrum
activity, the emergence of drug-resistant viral strains and high
treatment costs, underscore the urgent need for novel antiviral
agents (129). In this context,
scientific attention has been increasingly directed toward natural
products as valuable resources for drug discovery. Allicin, a
bioactive organosulfur compound formed when garlic tissue is
crushed or damaged, has attracted considerable interest due to its
broad-spectrum antimicrobial properties, with increasing evidence
supporting its antiviral potential (1,3,52,130).
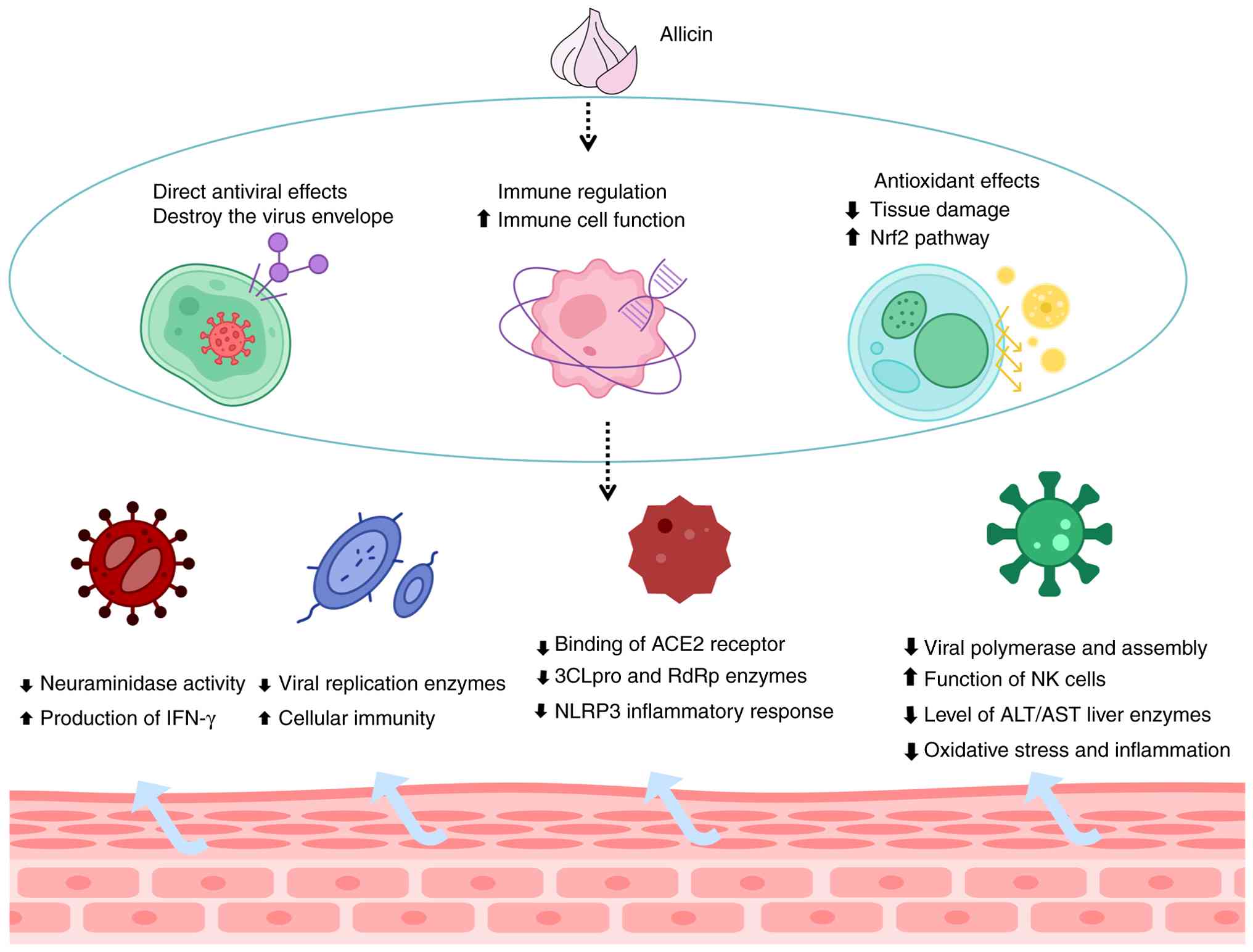
As illustrated in Fig.
2, garlic-derived organosulfur compounds, particularly allicin,
exhibit multifaceted antiviral mechanisms. These include direct
inactivation of viral particles and interference with viral entry,
replication and assembly (131,132). Molecular docking studies have
further demonstrated that allicin and its derivatives (such as
diallyl sulfide, diallyl disulfide, diallyl trisulfide and ajoene)
inhibit key viral enzymes, including the SARS-CoV-2 3C-like
protease and RNA-dependent RNA polymerase, thereby suppressing
viral replication (133-135).
Additionally, allicin modulates host inflammatory responses by
suppressing the NLR family pyrin domain containing 3 inflammasome
pathway, reducing excessive inflammation and associated tissue
damage (136). These combined
direct antiviral and immunomodulatory actions contribute to the
broad-spectrum therapeutic potential of allicin against viral
infections (131,132). Notably, the antiviral effects of
allicin exhibit a clear dose-response relationship. For example,
Weber et al (21)
demonstrated concentration-dependent virucidal activity against
herpes simplex virus types 1 and 2, providing classic
pharmacological evidence regarding the dose-dependent antiviral
action of allicin (21).
Mechanistic studies have suggested that allicin exerts
context-dependent effects on the cellular redox status. In
virus-infected cells, allicin induces oxidative stress through
S-thioallylation of protein thiols and glutathione depletion,
leading to redox-mediated alterations that promote the degradation
of viral components and create an unfavorable environment for viral
replication (46,57,137). By contrast, in uninfected cells
and tissues, allicin and its metabolites activate the nuclear
factor erythroid 2-related factor 2 (Nrf2) antioxidant pathway,
enhancing cellular antioxidant defenses and reducing oxidative
stress and inflammation, thereby mitigating virus-associated tissue
damage (21,33,57,138-143)
(Fig. 2; Table IV). This dual mechanism, selective
pro-oxidant action in infected cells coupled with cytoprotective
antioxidant effects in healthy tissues, contributes to the
therapeutic selectivity of allicin against viral infections
(137). In addition, allicin has
been shown to modulate host cell signaling pathways, including the
Nrf2 pathway, which serves an important role in regulating cellular
antioxidant defenses. Activation of this pathway by allicin induces
antioxidant enzymes that create an intracellular environment
unfavorable for viral replication, thereby suppressing viral
propagation (44,138,144). Notably, such modulation occurs
within an optimal concentration range rather than in a linear
manner, as emphasized in reviews of the immunomodulatory effects of
garlic-derived compounds (145-147).
Furthermore, allicin has been reported to act synergistically with
conventional antiviral drugs in certain contexts, including
enhanced inhibition of respiratory syncytial virus when combined
with moroxydine and α-interferon, potentially enhancing therapeutic
efficacy while reducing the required drug dosage and associated
toxicity (4,148). This synergistic effect may
additionally contribute to minimizing adverse effects and limiting
the emergence of drug-resistant viral strains (149,150).
 | Table IVSummary of the main antiviral
mechanisms of garlic-derived compounds. |
Table IV
Summary of the main antiviral
mechanisms of garlic-derived compounds.
| First author/s,
year | Mechanism
category | Pathway/target
involved | Primary action | Core antiviral
function | (Refs.) |
|---|
| Weber et al,
1992 | Direct antiviral
action | Viral
particles/enzymes (such as neuraminidase) |
Inhibition/inactivation | Blocking numerous
stages of the viral life cycle. | (21) |
| Miron et al,
2000; Nan et al, 2021 | Host cellular state
modulation | Oxidative and ER
stress | Induction | Creating an
unfavorable environment for viral replication; degrading viral
components. | (57,140) |
| Zeng et al,
2013; Zhang et al, 2022 | | Nrf2 signaling
pathway | Activation | Enhancing cellular
antioxidant defense, maintaining homeostasis | (141,142) |
| Tatarintsev et
al, 1992; Bouyahya et al, 2025 | Host immunity and
fate regulation | NF-κB pathway | Inhibition | Limiting viral
exploitation of host resources; mitigating excessive
inflammation. | (33,143) |
In addition to allicin, other garlic-derived
organosulfur compounds also exhibit broad-spectrum antiviral
activity through diverse mechanisms. Alliin, the biosynthetic
precursor of allicin, has been shown to suppress viral replication
in infected cells while exhibiting minimal cytotoxicity toward
uninfected host cells, suggesting a degree of selectivity in its
antiviral action (1). This
selective activity implies a favorable therapeutic window, a key
principle of dose-response pharmacology (3). Comparative analyses, including those
summarized by Ankri and Mirelman (3), indicate that the concentrations
required for antimicrobial (including antiviral) effects are often
markedly lower compared with those associated with toxicity in
mammalian cells, thereby defining a beneficial selective index.
Specifically, this selectivity translates into a
favorable therapeutic window. In vitro studies have
demonstrated that allicin exhibits concentration-dependent
antiviral activity with a quantifiable safety margin (21,132,137). Effective concentrations against
numerous viruses are often observed in the low micromolar range,
whereas cytotoxic concentrations are notably higher. As a result,
reported selectivity indices frequently exceed a score of 3, as
shown in comparative bioactivity analyses (3,21).
However, this therapeutic window is not fixed and may vary
depending on the viral species, host cell type and experimental
conditions (21). Notably,
translating in vitro potency into safe and effective human
dosing requires systematic investigation, as achieving and
maintaining therapeutic plasma or tissue concentrations in
vivo involves complex pharmacokinetic and bioavailability
factors that remain incompletely characterized (51).
DADS, a stable transformation product of allicin,
has been shown to enhance host antiviral defenses by activating the
Nrf2 signaling pathway. This upregulates a network of antioxidant
and cytoprotective genes that can restrict viral replication
(141). Similarly, DATS
interferes with numerous stages of the viral life cycle,
potentially through disruption of viral entry and assembly
processes (44). The antiviral
activity of DATS and related compounds is explicitly
concentration-dependent, with efficacy increasing sharply across a
narrow dose range in experimental models (151). Furthermore, the induction of
caspase-3 and caspase-8 activity in virus-infected cells indicates
that these compounds can promote apoptosis of infected cells,
thereby limiting viral spread (57). This pro-apoptotic effect itself
requires a threshold concentration to initiate the apoptotic
cascade, reflecting a key principle of dose-response pharmacology
(57). In addition, secondary
metabolites such as S-allylmercaptocysteine and S-allylcysteine
contribute to overall antiviral efficacy by inhibiting viral
replication and facilitating the clearance of infected cells
(21).
The proposed mechanism underlying the antiviral
activity of allicin is summarized in Fig. 2. Following cellular uptake in
virus-infected cells, allicin primarily induces intracellular
oxidative stress through the generation of reactive oxygen species,
which can promote degradation of viral components (57). A key aspect of its antiviral action
involves modulation of host cell signaling pathways important for
viral replication. Allicin has been shown to influence the NF-κB
pathway, a central regulator of inflammation and cell survival that
is frequently exploited by viruses (131,152). In addition, allicin affects MAPK
signaling cascades, which viruses commonly manipulate to establish
a favorable intracellular environment (131,153). Collectively, these mechanisms,
including induction of oxidative stress and disruption of pro-viral
signaling networks, result in inhibition of viral replication and
facilitate elimination of infected cells, potentially through
enhanced caspase-mediated apoptosis (154,155).
Beyond modulation of the cellular redox state,
allicin activates key host defense signaling pathways, such as the
ERK/MAPK pathway, thereby promoting an intracellular environment
unfavorable for viral replication (156). Unlike the aforementioned direct
oxidative stress mechanisms, which degrade viral components through
chemical modification, these signaling pathways regulate immune
gene expression and cellular antiviral responses (132,156). Studies have shown that allicin
stimulates the JNK and p38 MAPK signaling cascades, both of which
serve important roles in initiating intrinsic antiviral responses
(19,140,156). Activation of these pathways
promotes the upregulation of immunomodulatory receptors and
facilitates caspase activation, which are processes that contribute
to the programmed elimination of virus-infected cells (30). As a result, viral production and
spread are effectively curtailed (1). This multifaceted engagement of host
signaling networks highlights the ability of allicin to disrupt the
viral life cycle through both direct antiviral effects and indirect
host-mediated mechanisms.
Allicin further combats viral infection by inducing
programmed cell death in infected host cells through a number of
molecular pathways. This process includes disruption of
mitochondrial integrity, leading to the cytosolic release of
pro-apoptotic factors that subsequently activate key effector
enzymes (57). In addition,
allicin can induce endoplasmic reticulum stress in infected cells,
a mechanism known to interfere with viral protein synthesis and
folding. This disruption contributes to suppression of viral
replication and promotes clearance of infected cells (26). Collectively, these complementary
mechanisms enhance overall antiviral efficacy by eliminating
cellular niches that support viral replication. To clarify how
these multifaceted actions operate in concert, the core antiviral
pathways and mechanisms of allicin are summarized in Table IV (21,33,57,140-143).
Beyond its direct antiviral effects, allicin may
also serve a protective role in mitigating virus-associated tissue
damage and modulating the host immune response. For example,
activation of cellular defense pathways such as the Nrf2-mediated
antioxidant response has been shown to reduce oxidative stress and
inflammatory injury in experimental models of viral hepatitis,
thereby contributing to liver protection (23). In addition, the immunomodulatory
properties of allicin suggest potential benefits in attenuating
excessive inflammatory responses commonly observed in severe viral
infections (19). Although
numerous in vivo and preclinical studies support these
protective effects (30,137,138), further clinical investigations
are required to determine their relevance and applicability in the
management of human viral diseases (157,158). Overall, available evidence
underscores the role of garlic-derived compounds in modulating
inflammatory profiles in chronic pathological conditions.
6. Safety aspects
Careful evaluation is warranted when considering
the safety profile of allicin in relation to its application as an
antiviral therapeutic agent. Despite garlic having a long history
of dietary use, the potent biological activity of allicin
necessitates thorough assessment of potential adverse effects
(22,159). A major limitation in current
safety evaluations is the heavy reliance on animal toxicology
studies, accompanied by a lack of comprehensive human safety trials
and long-term surveillance data needed to fully characterize risks
under therapeutic conditions (160,161).
Clinical observations indicate that high-dose
allicin supplementation may cause gastrointestinal discomfort,
including nausea, heartburn and diarrhea, as well as systemic
effects such as headache, tachycardia and mild hypotension in
susceptible individuals (49).
However, existing human data are largely derived from
efficacy-oriented studies with limited safety monitoring periods.
These studies lack the systematic, longitudinal design required to
detect rare or delayed adverse events (22,160,162). Consequently, there is a clear
need for prospective safety studies specifically designed to
evaluate the risk profile of allicin in defined patient populations
across extended treatment durations.
Experimental studies have demonstrated a clear
dose-dependent toxicity profile for allicin (163,164). At low to moderate doses, allicin
enhances antioxidant capacity in animal models. By contrast,
excessive administration (for example, 1,000 mg/kg/day in rats) has
been associated with hepatorenal toxicity, as evidenced by
histopathological alterations in liver and kidney tissues (165). The molecular basis of this
differential effect may be attributed to the characteristics of
allicin as a reactive sulfur species. Allicin readily penetrates
biological membranes and oxidizes intracellular thiols, including
glutathione and protein cysteine residues. Such oxidative reactions
can induce structural modifications through disulfide bond
formation, thereby disrupting normal cellular function and
potentially resulting in cytotoxic effects (57).
Chronic high-dose exposure studies have identified
additional safety concerns. Prolonged administration of
concentrated garlic preparations has been associated with
hematological abnormalities, including oxidative hemolysis of
erythrocytes, as well as body weight loss and impaired
spermatogenesis in rodent models (165,166,167). In addition, toxicological
investigations of pulmonary and hepatic systems indicate that
sustained consumption of high doses of garlic juice can induce
adverse changes in these organs (168,169). Collectively, these findings
emphasize the importance of careful dose optimization and duration
control in the development of allicin-based antiviral
formulations.
Overall, preclinical evidence supports a
dose-dependent safety profile for allicin, in which beneficial or
neutral effects are observed at lower concentrations [for example,
5-20 mg/kg/day in mice (170) and
15-45 mg/kg/day in rats (171)],
whereas sustained exposure to doses exceeding the therapeutic range
may result in adverse organ effects, including signs of hepatorenal
stress in rodent models (171).
Translation of these toxicological findings to human risk
assessment requires cautious interpretation due to well-documented
interspecies differences in drug metabolism and physiological
responses (172). Consequently, a
central objective in translational development is to empirically
define the human therapeutic window by identifying dose ranges that
achieve antiviral efficacy while maintaining an acceptable safety
margin. This process necessitates systematic phase I clinical
trials designed to characterize pharmacokinetics, assess
tolerability and establish the maximum tolerated dose in human
subjects (173). Therefore,
although existing non-clinical data identify exposure thresholds
associated with toxicological risk, definitive demonstration of the
safety of allicin at clinically relevant doses must rely on
prospective, well-controlled studies in human populations (68).
Beyond the intrinsic toxicity profile of allicin
and garlic-derived preparations, their potential to interact with
conventional medications represents an additional key safety
consideration. The most extensively documented interaction involves
anticoagulant and antiplatelet therapies (174-176).
Allicin and other garlic organosulfur compounds exhibit
antiplatelet activity, perhaps through inhibition of thromboxane
synthesis and platelet aggregation. As a result, these compounds
may potentiate the effects of anticoagulant and antiplatelet drugs
such as warfarin, aspirin and clopidogrel, thereby increasing the
risk of bleeding, particularly with high-dose or prolonged garlic
supplementation (174,177). Documented interactions between
warfarin and concentrated garlic products have been reported in the
literature, including cases of elevated international normalized
ratios and bleeding events (178-180).
Such inhibition may increase the systemic exposure
and toxicity of drugs that are substrates of these enzymes,
including CYP2C19 substrates such as proton pump inhibitors (such
as omeprazole), antiplatelet drugs (such as clopidogrel) and
certain antiviral agents (such as nelfinavir) (65,66).
This effect is especially relevant in antiviral therapy, as
numerous protease inhibitors used in the treatment of HIV and
hepatitis C rely heavily on CYP3A4-mediated metabolism (181). Conversely, evidence has indicated
that chronic garlic consumption may induce CYP enzyme activity,
underscoring the complexity and dose- and duration-dependent nature
of these interactions (182).
In addition, given the reported hypoglycemic
effects of garlic supplementation, including reductions in fasting
blood glucose and hemoglobin A1c in patients with type 2 diabetes
(183,184), and its antihypertensive effects,
including modest but significant reductions in both systolic and
diastolic blood pressure in individuals with hypertension (185,186), potential interactions with
antihypertensive and antidiabetic medications warrant
consideration, although supporting clinical evidence remains
limited (187). Consequently, for
allicin to be safely incorporated into antiviral treatment
regimens, particularly as part of combination therapies, careful
patient selection, monitoring of coagulation parameters and drug
concentrations (when applicable), and clear guidance regarding the
use of standardized formulations vs. dietary garlic preparations
are key.
7. Conclusion and future perspectives
Allicin, a principal organosulfur compound derived
from garlic, exhibits potential as a broad-spectrum antiviral agent
based on extensive preclinical research. Its antiviral activity
arises from numerous complementary mechanisms, including direct
virucidal effects, disruption of viral replication processes and
modulation of host cellular signaling pathways.
Current clinical evidence, summarized in Table III, suggests that garlic-based
interventions may provide therapeutic benefits for certain viral
infections, including reductions in cold/flu incidence and
severity, as well as improved clinical outcomes in viral hepatitis
and cutaneous warts. However, as shown in Table III, these studies predominantly
used multi-component garlic preparations (such as whole extract,
aged extract and garlic oil) rather than purified allicin, and
exhibited considerable heterogeneity in study design, dosing and
clinical endpoints (20,34,38-43,49).
This variability highlights a gap between controlled experimental
findings and validated clinical application.
Notable progress has been made in overcoming the
physicochemical limitations of allicin, particularly through the
development of engineered delivery systems such as nanocarriers,
which enhance stability and systemic bioavailability. Although
these technological advances represent key prerequisites,
definitive evidence of antiviral efficacy, optimized dosing
regimens and comprehensive safety profiles for purified or
formulated allicin in humans must still be established through
clinical investigation.
Future research should aim to prioritize clinical
validation of these advanced delivery platforms. The most important
subsequent steps include conducting phase-appropriate clinical
trials using standardized allicin formulations to characterize
pharmacokinetics, defining dose-response relationships and
assessing safety, including the potential for drug-drug
interactions, in relevant patient populations, including
individuals with acute viral infections [such as influenza or
COVID-19(157)], those at high
risk of viral complications [including immunocompromised or elderly
patients (188)] and patients
receiving concurrent antiviral or other medications that may
interact with allicin (188,189). In addition, research efforts
should continue to elucidate pathogen-specific mechanisms of action
and host-directed immunomodulatory effects.
In summary, allicin represents a scientifically
well-established antiviral candidate supported by robust
preclinical evidence and emerging, albeit preliminary, clinical
observations. Its successful translation into a viable antiviral
therapeutic will depend on coordinated research strategies designed
to systematically bridge existing evidence gaps through targeted
clinical studies and formulation-focused innovation.
Acknowledgements
Not applicable.
Funding
Funding: The present review was supported by the Construction
Task Book of Hangzhou Biomedical and Health Industry Development
Support Science and Technology Special Project (Phase 14; grant no.
2024WJC135), the Zhejiang Traditional Chinese Medicine Science and
Technology Project (grant no. 2023ZL565) and the Construction Fund
of Key Medical Disciplines of Hangzhou (grant no. HWB-2025-10).
Availability of data and materials
Not applicable.
Authors' contributions
MJY and ZM conceptualized the study and wrote the
original draft of the manuscript. YZ and WZ were involved in the
screening, extraction and analysis of the literature data, based on
which they designed the figures and tables. QPX and ZM contributed
to the writing, review and revision of the manuscript. HMW
contributed to the construction of the theoretical framework, the
interpretation of data and the critical review of key sections of
the manuscript. Data authentication is not applicable. All authors
have read and approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Rouf R, Uddin SJ, Sarker DK, Islam MT, Ali
ES, Shilpi JA, Nahar L, Tiralongo E and Sarker SD: Antiviral
potential of garlic (Allium sativum) and its organosulfur
compounds: A systematic update of pre-clinical and clinical data.
Trends Food Sci Technol. 104:219–234. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
El-Saber Batiha G, Magdy Beshbishy A, G
Wasef L, Elewa YHA, A Al-Sagan A, Abd El-Hack ME, Taha AE, M
Abd-Elhakim Y and Prasad Devkota H: Chemical constituents and
pharmacological activities of garlic (Allium sativum L.): A
review. Nutrients. 12(872)2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ankri S and Mirelman D: Antimicrobial
properties of allicin from garlic. Microbes Infect. 1:125–129.
1999.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Khubber S, Hashemifesharaki R, Mohammadi M
and Gharibzahedi SMT: Garlic (Allium sativum L.): A
potential unique therapeutic food rich in organosulfur and
flavonoid compounds to fight with COVID-19. Nutr J.
19(124)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Chung LY: The antioxidant properties of
garlic compounds: Allyl cysteine, alliin, allicin, and allyl
disulfide. J Med Food. 9:205–213. 2006.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Strasfeld L and Chou S: Antiviral drug
resistance: Mechanisms and clinical implications. Infect Dis Clin
North Am. 24:413–437. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Piret J and Boivin G: Antiviral drug
resistance in herpesviruses other than cytomegalovirus. Rev Med
Virol. 24:186–218. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hejran AB, Sarwari A, Yadav VK, Hassand MS
and Niazi P: Analyzing and critically evaluating the problems of
antiviral chemotherapy. Br J Pharm Pharm Sci. 1:47–59. 2024.
|
|
9
|
Majeed M, Nagabhushanam K, Lawrence L,
Prakasan P and Mundkur L: The mechanism of anti-viral activity of a
novel, hydroponically selenium-enriched garlic powder
(SelenoForce®) against SARS-CoV-2 virus. Glob Adv Integr
Med Health. 13(27536130241268100)2024.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Smith T, Purcell D, Diamantopoulos N and
McAuley J: Inhibitory effects of SARS-CoV-2 penetration of host
cells by garlic oil and juice extract are cultivar specific. J
Functi Foods. 121(106393)2024.
|
|
11
|
Atoum MF, Padma KR and Don KR: Paving new
roads using Allium sativum as a repurposed drug and
analyzing its antiviral action using artificial intelligence
technology. Iran J Pharm Res. 21(e131577)2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gasparello J, Papi C, Marzaro G, Macone A,
Zurlo M, Finotti A, Agostinelli E and Gambari R: Aged garlic
extract (AGE) and its constituent S-Allyl-cysteine (SAC) inhibit
the expression of pro-inflammatory genes induced in bronchial
epithelial IB3-1 cells by exposure to the SARS-CoV-2 spike protein
and the BNT162b2 vaccine. Molecules. 29(5938)2024.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Taghavi MR, Tavanaei Tamanaei T, Oghazian
MB, Tavana E, Mollazadeh S, Niloofar P, Oghazian S, Hoseinzadeh A,
Hesari A, Ansari Mohseni M, et al: Effectiveness of fortified
garlic extract oral capsules as adjuvant therapy in hospitalized
patients with coronavirus disease 2019: A triple-blind randomized
controlled clinical trial. Curr Ther Res Clin Exp.
98(100699)2023.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sofi FA and Tabassum N: Natural product
inspired leads in the discovery of anticancer agents: An update. J
Biomol Struct Dyn. 41:8605–8628. 2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Verma T, Aggarwal A, Dey P, Chauhan AK,
Rashid S, Chen KT and Sharma R: Medicinal and therapeutic
properties of garlic, garlic essential oil, and garlic-based snack
food: An updated review. Front Nutr. 10(1120377)2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Singh N, Gusain A, Nigam M and Mishra AP:
The pharmacological and therapeutic versatility of Allium species:
A comprehensive exploration of bioactive constituents and
biological activities. Discov Appl Sci. 7(349)2025.
|
|
17
|
Ilić DP, Nikolić V, Nikolić LB, Stankovic
MZ, Stanojević LP and Cakić MD: Allicin and related compounds:
Biosynthesis, synthesis and pharmacological activity†.
Facta Univ Phys Chem Technol. 9:9–20. 2011.
|
|
18
|
Iciek M, Kwiecień I and Włodek L:
Biological properties of garlic and garlic-derived organosulfur
compounds. Environ Mol Mutagen. 50:247–265. 2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Arreola R, Quintero-Fabián S, López-Roa
RI, Flores-Gutiérrez EO, Reyes-Grajeda JP, Carrera-Quintanar L and
Ortuño-Sahagún D: Immunomodulation and anti-inflammatory effects of
garlic compounds. J Immunol Res. 2015(401630)2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Percival SS: Aged garlic extract modifies
human immunity. J Nutr. 146:433S–436S. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Weber ND, Andersen DO, North JA, Murray
BK, Lawson LD and Hughes BG: In vitro virucidal effects of
Allium sativum (garlic) extract and compounds. Planta Med.
58:417–423. 1992.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Salehi B, Zucca P, Orhan IE, Azzini E,
Adetunji CO, Mohammed SA, Banerjee SK, Sharopov F, Rigano D,
Sharifi-Rad J, et al: Allicin and health: A comprehensive review.
Trends Food Sci Technol. 86:502–516. 2019.
|
|
23
|
Deng Y, Ho CT, Lan Y, Xiao J and Lu M:
Bioavailability, health benefits, and delivery systems of allicin:
A review. J Agric Food Chem. 71:19207–19220. 2023.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Bala R, Madaan R, Chauhan S, Gupta M,
Dubey AK, Zahoor I, Brijesh H, Calina D and Sharifi-Rad J:
Revitalizing allicin for cancer therapy: Advances in formulation
strategies to enhance bioavailability, stability, and clinical
efficacy. Naunyn Schmiedebergs Arch Pharmacol. 397:703–724.
2024.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ditta SA, Yaqub A, Ullah R, Tanvir F and
Ali S: Allicin-capped silver nanoparticles (AgNPs): Synthesis,
profiling, antioxidant, and biomedical properties. Nano Biomed Eng.
16:370–385. 2024.
|
|
26
|
Alyasiri FJ, Ghobeh M and Tabrizi MH:
Preparation and characterization of allicin-loaded solid lipid
nanoparticles surface-functionalized with folic acid-bonded
chitosan: In vitro anticancer and antioxidant activities. Front
Biosci (Landmark Ed). 28(135)2023.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Petrovska BB and Cekovska S: Extracts from
the history and medical properties of garlic. Pharmacogn Rev.
4:106–110. 2010.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Jakobsen TH, van Gennip M, Phipps RK,
Shanmugham MS, Christensen LD, Alhede M, Skindersoe ME, Rasmussen
TB, Friedrich K, Uthe F, et al: Ajoene, a sulfur-rich molecule from
garlic, inhibits genes controlled by quorum sensing. Antimicrob
Agents Chemother. 56:2314–2325. 2012.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Tsai Y, Cole LL, Davis LE, Lockwood SJ,
Simmons V and Wild GC: Antiviral properties of garlic: In vitro
effects on influenza B, herpes simplex and coxsackie viruses.
Planta Med. 51:460–461. 1985.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Borlinghaus J, Foerster Née Reiter J,
Kappler U, Antelmann H, Noll U, Gruhlke MCH and Slusarenko AJ:
Allicin, the odor of freshly crushed garlic: A review of recent
progress in understanding Allicin's effects on cells. Molecules.
26(1505)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Klenk HD, Matrosovich M and Stech J: Avian
influenza: Molecular mechanisms of pathogenesis and host range. In:
Animal Viruses: Molecular Biology. Mettenleiter TC and Sobrino F
(eds). Caister Academic Press, UK, pp253-303, 2008.
|
|
32
|
Thuy BTP, My TTA, Hai NTT, Hieu LT, Hoa
TT, Thi Phuong Loan H, Triet NT, Anh TTV, Quy PT, Tat PV, et al:
Investigation into SARS-CoV-2 resistance of compounds in garlic
essential oil. ACS Omega. 5:8312–8320. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Tatarintsev AV, Vrzhets PV, Ershov DE,
Shchegolev AA, Turgiev AS, Karamov EV, Kornilaeva GV, Makarova TV,
Fedorov NA and Varfolomeev SD: The ajoene blockade of
integrin-dependent processes in an HIV-infected cell system. Vestn
Ross Akad Med Nauk. 6–10. 1992.PubMed/NCBI(In Russian).
|
|
34
|
Nantz MP, Rowe CA, Muller CE, Creasy RA,
Stanilka JM and Percival SS: Supplementation with aged garlic
extract improves both NK and γδ-T cell function and reduces the
severity of cold and flu symptoms: a randomized, double-blind,
placebo-controlled nutrition intervention. Clin Nutr. 31:337–344.
2012.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Guo NL, Lu DP, Woods GL, Reed E, Zhou GZ,
Zhang LB and Waldman RH: Demonstration of the anti-viral activity
of garlic extract against human cytomegalovirus in vitro. Chin Med
J (Engl). 106:93–96. 1993.PubMed/NCBI
|
|
36
|
Fadlalla IMT, Mohammed BH and Bakhiet AO:
Effect of feeding garlic on the performance and immunity of
broilers. Asian J Poult Sci. 4:182–189. 2010.
|
|
37
|
Choi YH and Park HY: Anti-inflammatory
effects of spermidine in lipopolysaccharide-stimulated BV2
microglial cells. J Biomed Sci. 19(31)2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Andrianova IV, Sobenin IA, Sereda EV,
Borodina LI and Studenikin MI: Effect of long-acting garlic tablets
‘allicor’ on the incidence of acute respiratory viral infections in
children. Ter Arkh. 75:53–56. 2003.PubMed/NCBI(In Russian).
|
|
39
|
Kenawy S, Mohammed GF, Younes S and
Elakhras AI: Evaluation of TNF-α serum level in patients with
recalcitrant multiple common warts, treated by lipid garlic
extract. Dermatol Ther. 27:272–277. 2014.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lee MH, Kim YM and Kim SG: Efficacy and
tolerability of diphenyl-dimethyl-dicarboxylate plus garlic oil in
patients with chronic hepatitis. Int J Clin Pharmacol Ther.
50:778–786. 2012.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Hiltunen R, Josling PD and James MH:
Preventing airborne infection with an intranasal cellulose powder
formulation (Nasaleze travel). Adv Ther. 24:1146–1153.
2007.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Mousavi ZB, Mehrabian A, Golfakhrabadi F
and Namjoyan F: A clinical study of efficacy of garlic extract
versus cryotherapy in the treatment of male genital wart. Dermatol
Sin. 36:196–199. 2018.
|
|
43
|
Gołebiowska-Wawrzyniak M, Markiewicz K,
Kozar A, Derentowicz P, Czerwińska-Kartowicz I, Jastrzebska-Janas
K, Wacławek J, Wawrzyniak ZM and Siwińska-Gołebiowska H:
Immunological and clinical study on therapeutic efficacy of inosine
pranobex. Pol Merkur Lekarski. 19:379–382. 2005.PubMed/NCBI(In Polish).
|
|
44
|
Zhu Y, Peng S, Peng S, Chen X, Zou L,
Liang R, Ruan R, Dai L and Liu W: Fiber complex-stabilized
high-internal-phase emulsion for allicin encapsulation:
Microstructure, stability, and thermal-responsive properties. J Sci
Food Agric. 105:1116–1125. 2025.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Reiter J, Hübbers AM, Albrecht F, Leichert
LIO and Slusarenko AJ: Allicin, a natural antimicrobial defence
substance from garlic, inhibits DNA gyrase activity in bacteria.
Int J Med Microbiol. 310(151359)2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Rabinkov A, Miron T, Konstantinovski L,
Wilchek M, Mirelman D and Weiner L: The mode of action of allicin:
Trapping of radicals and interaction with thiol containing
proteins. Biochim Biophys Acta. 1379:233–244. 1998.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Berginc K, Milisav I and Kristl A: Garlic
flavonoids and organosulfur compounds: Impact on the hepatic
pharmacokinetics of saquinavir and darunavir. Drug Metab
Pharmacokinet. 25:521–530. 2010.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Ayaz M, Ullah F, Sadiq A, Ullah F, Ovais
M, Ahmed J and Devkota HP: Synergistic interactions of
phytochemicals with antimicrobial agents: Potential strategy to
counteract drug resistance. Chem Biol Interact. 308:294–303.
2019.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Josling P: Preventing the common cold with
a garlic supplement: A double-blind, placebo-controlled survey. Adv
Ther. 18:189–193. 2001.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Lissiman E, Bhasale AL and Cohen M: Garlic
for the common cold. Cochrane Database Syst Rev.
2014(CD006206)2014.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Lawson LD and Wang ZJ: Allicin and
allicin-derived garlic compounds increase breath acetone through
allyl methyl sulfide: Use in measuring allicin bioavailability. J
Agric Food Chem. 53:1974–1983. 2005.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Borlinghaus J, Albrecht F, Gruhlke MCH,
Nwachukwu ID and Slusarenko AJ: Allicin: Chemistry and biological
properties. Molecules. 19:12591–12618. 2014.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Fujisawa H, Suma K, Origuchi K, Seki T and
Ariga T: Thermostability of allicin determined by chemical and
biological assays. Biosci Biotechnol Biochem. 72:2877–2883.
2008.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Miron T, Bercovici T, Rabinkov A, Wilchek
M and Mirelman D: [3H]Allicin: Preparation and applications. Anal
Biochem. 331:364–369. 2004.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Lawson LD and Hunsaker SM: Allicin
bioavailability and bioequivalence from garlic supplements and
garlic foods. Nutrients. 10(812)2018.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Lachmann G, Lorenz D, Radeck W and Steiper
M: The pharmacokinetics of the S35 labeled labeled garlic
constituents alliin, allicin and vinyldithiine.
Arzneimittelforschung. 44:734–743. 1994.PubMed/NCBI(In German).
|
|
57
|
Miron T, Rabinkov A, Mirelman D, Wilchek M
and Weiner L: The mode of action of allicin: Its ready permeability
through phospholipid membranes may contribute to its biological
activity. Biochim Biophys Acta. 1463:20–30. 2000.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Lawson LD: Garlic: A review of its
medicinal effects and indicated active compounds. In:
Phytomedicines of Europe: Chemistry and Biological Activity. Lawson
L and Bauer R (eds). American Chemical Society, Washington DC,
pp177-209, 1998.
|
|
59
|
Ellmore GS and Feldberg RS: Alliin lyase
localization in bundle sheaths of the garlic clove (Allium
sativum). Am J Bot. 81:89–94. 1994.
|
|
60
|
Stoll A and Seebeck E: About Alliin, the
genuine mother substance of garlic oil. Experientia.
3(114)1947.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Block E: The chemistry of garlic and
onions. Sci Am. 252:114–119. 1985.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Lawson LD and Gardner CD: Composition,
stability, and bioavailability of garlic products used in a
clinical trial. J Agric Food Chem. 53:6254–6261. 2005.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Egen-Schwind C, Eckard R, Jekat FW and
Winterhoff H: Pharmacokinetics of vinyldithiins, transformation
products of allicin. Planta Med. 58:8–13. 1992.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Nakamoto M, Kunimura K and Ohtani M:
Pharmacokinetics of sulfur-containing compounds in aged garlic
extract: S-Allylcysteine, S-1-propenylcysteine, S-methylcysteine,
S-allylmercaptocysteine and others (Review). Exp Ther Med.
29(102)2025.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Yang LJ, Fan L, Liu ZQ, Mao YM, Guo D, Liu
LH, Tan ZR, Peng L, Han CT, Hu DL, et al: Effects of allicin on
CYP2C19 and CYP3A4 activity in healthy volunteers with different
CYP2C19 genotypes. Eur J Clin Pharmacol. 65:601–608.
2009.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Zou L, Harkey MR and Henderson GL: Effects
of herbal components on cDNA-expressed cytochrome P450 enzyme
catalytic activity. Life Sci. 71:1579–1589. 2002.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Lawson LD and Wang ZJ: Low allicin release
from garlic supplements: A major problem due to the sensitivities
of alliinase activity. J Agric Food Chem. 49:2592–2599.
2001.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Ried K and Fakler P: Potential of garlic
(Allium sativum) in lowering high blood pressure: Mechanisms
of action and clinical relevance. Integr Blood Press Control.
7:71–82. 2014.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Egen-Schwind C, Eckard R and Kemper FH:
Metabolism of garlic constituents in the isolated perfused rat
liver. Planta Med. 58:301–305. 1992.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Jafernik K, Ładniak A, Blicharska E,
Czarnek K, Ekiert H, Wiącek AE and Szopa A: Chitosan-based
nanoparticles as effective drug delivery systems-a review.
Molecules. 28(1963)2023.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Hashemy SI, Amiri H, Hosseini H,
Sadeghzadeh F, Jaseem MMM and Tabrizi MH: PEGylated
lecithin-chitosan-folic acid nanoparticles as nanocarriers of
allicin for in vitro controlled release and anticancer effects.
Appl Biochem Biotechnol. 195:4036–4052. 2023.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Liu Y, Song R, Zhang X and Zhang D:
Enhanced antimicrobial activity and pH-responsive sustained release
of chitosan/poly (vinyl alcohol)/graphene oxide nanofibrous
membrane loading with allicin. Int J Biol Macromol. 161:1405–1413.
2020.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Miron T, Wilchek M, Shvidel L, Berrebi A
and Arditti FD: S-allyl derivatives of 6-mercaptopurine are highly
potent drugs against human B-CLL through synergism between
6-mercaptopurine and allicin. Leuk Res. 36:1536–1540.
2012.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Appel E, Rabinkov A, Neeman M, Kohen F and
Mirelman D: Conjugates of daidzein-alliinase as a targeted pro-drug
enzyme system against ovarian carcinoma. J Drug Target. 19:326–335.
2011.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Krest I and Keusgen M: Stabilization and
pharmaceutical use of alliinase. Pharmazie. 54:289–293.
1999.PubMed/NCBI
|
|
76
|
Yang Y, Li M, Bao HY, Li XX and Li JG:
Advances in cardiovascular protection effects and mechanisms of
allicin. Zhongguo Zhong Yao Za Zhi. 48:3409–3420. 2023.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
77
|
Mozafari ES, Baei MT and Lemeski ET:
Computational study of the therapeutic properties of allicin and
its nanocomplexes using DFT and molecular docking techniques. Sci
Rep. 15(23034)2025.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Xu Q, Wu J, Zhao Y, Zhou Z, Liu Y, Song Z,
Chen L and Han H: Macrophage membrane-coated nanoscale allicin for
the targeted treatment of drug-resistant bacterial pneumonia. ACS
Appl Nano Mater. 8:935–943. 2025.
|
|
79
|
Najafi F, Farrokhzad N, Ghaemi A, Khezri
DA, Hajiabbas M, Khanizadeh A, Farsani NK, Khoramipour M,
Fatemipayam N, Zadeh ES, et al: Advancement in nanocarrier-mediated
delivery of herbal bioactives: From bench to beside. Nat Prod
Bioprospect. 15(69)2025.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Kharazmi F, Neamati A and Tabrizi MH:
Evaluation of the anticancer effects of allicin-loaded solid lipid
nanoparticles in the A549 lung cancer cell line. BioNanoSci.
15(49)2025.
|
|
81
|
Mendoza-Muñoz N, Urbán-Morlán Z,
Leyva-Gómez G, Zambrano-Zaragoza ML, Piñón-Segundo E and
Quintanar-Guerrero D: Solid lipid nanoparticles: An approach to
improve oral drug delivery. J Pharm Pharm Sci. 24:509–532.
2021.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Yang J, Zeng H, Luo Y, Chen Y, Wang M, Wu
C and Hu P: Recent applications of PLGA in drug delivery systems.
Polymers (Basel). 16(2606)2024.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Danhier F, Ansorena E, Silva JM, Coco R,
Le Breton A and Préat V: PLGA-based nanoparticles: An overview of
biomedical applications. J Control Release. 161:505–522.
2012.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Abruzzo A, Giordani B, Miti A, Vitali B,
Zuccheri G, Cerchiara T, Luppi B and Bigucci F: Mucoadhesive and
mucopenetrating chitosan nanoparticles for glycopeptide antibiotic
administration. Int J Pharm. 606(120874)2021.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Jiang H, Zhang S, Chen Y, Wang F and Jiang
W: Preparation and characterization of curdlan-chitosan conjugate
nanoparticles as mucosal adjuvants for intranasal influenza H1N1
subunit vaccine. Int J Biol Macromol. 266(131289)2024.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Ma C, Li S, Yin Y, Xu W, Xue T, Wang Y,
Liu X and Liu F: Preparation, characterization, formation mechanism
and stability of allicin-loaded emulsion gel. LWT.
161(113389)2022.
|
|
87
|
Su Y, Liu J, Tan S, Liu W, Wang R and Chen
C: PLGA sustained-release microspheres loaded with an insoluble
small-molecule drug: Microfluidic-based preparation, optimization,
characterization, and evaluation in vitro and in vivo. Drug Deliv.
29:1437–1446. 2022.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Kong W, Wei Y, Dong Z, Liu W, Zhao J,
Huang Y, Yang J, Wu W, He H and Qi J: Role of size, surface charge,
and PEGylated lipids of lipid nanoparticles (LNPs) on intramuscular
delivery of mRNA. J Nanobiotechnology. 22(553)2024.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Kalkal A, Allawadhi P, Pradhan R, Khurana
A, Bharani KK and Packirisamy G: Allium sativum derived
carbon dots as a potential theranostic agent to combat the COVID-19
crisis. Sens Int. 2(100102)2021.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Hu L, Zhao P, Wei Y, Lei Y, Guo X, Deng X
and Zhang J: Preparation and characterization study of zein-sodium
caseinate nanoparticle delivery systems loaded with allicin. Foods.
13(3111)2024.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Ding S, Zhao J, Jiang Z, Mu J, Huang L and
Dai C: Fabrication of whey protein isolate/chitosan complexes and
its protective effect on allicin. J Appl Polym Sci.
140(e53576)2023.
|
|
92
|
NANOTEC: Allicin from garlic is a
remarkable bioactive that Thai researchers are advancing through
nanotechnology. NANOTEC, Pathum Thani, 2024.
|
|
93
|
Petros RA and DeSimone JM: Strategies in
the design of nanoparticles for therapeutic applications. Nat Rev
Drug Discov. 9:615–627. 2010.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Thakur S, Godela R, Mandava K and Kolure
R: Advances in nanocarrier technology for drug encapsulation: A
comprehensive overview. Discov Mater. 5(124)2025.
|
|
95
|
Abdul Azeez N, Venkateshwaran K, Kandasamy
R, Saravanan M and Deepa VS: Importance of nano-carriers,
surfactant system, and their loading mechanism to improve the
absorption and bio-availability of drugs: A review. Adv Nat Sci
Nanosci Nanotechnol. 15(033002)2024.
|
|
96
|
Yanamadala Y, Muthumula CMR, Khare S and
Gokulan K: Strategies to enhance nanocrystal formulations for
overcoming physiological barriers across diverse routes of
administration. Int J Nanomedicine. 20:367–402. 2025.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Peer D, Karp JM, Hong S, Farokhzad OC,
Margalit R and Langer R: Nanocarriers as an emerging platform for
cancer therapy. Nat Nanotechnol. 2:751–760. 2007.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Odriozola-Serrano I, Oms-Oliu G and
Martín-Belloso O: Nanoemulsion-based delivery systems to improve
functionality of lipophilic components. Front Nutr.
1(24)2014.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Maeda H, Nakamura H and Fang J: The EPR
effect for macromolecular drug delivery to solid tumors:
Improvement of tumor uptake, lowering of systemic toxicity, and
distinct tumor imaging in vivo. Adv Drug Deliv Rev. 65:71–79.
2013.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Wu J: The enhanced permeability and
retention (EPR) effect: The significance of the concept and methods
to enhance its application. J Pers Med. 11(771)2021.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Fenaroli F, Repnik U, Xu Y, Johann K, Van
Herck S, Dey P, Skjeldal FM, Frei DM, Bagherifam S, Kocere A, et
al: Enhanced permeability and retention-like extravasation of
nanoparticles from the vasculature into tuberculosis granulomas in
zebrafish and mouse models. ACS Nano. 12:8646–8661. 2018.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Azzopardi EA, Ferguson EL and Thomas DW:
The enhanced permeability retention effect: A new paradigm for drug
targeting in infection. J Antimicrob Chemother. 68:257–274.
2013.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Matsumura Y and Maeda H: A new concept for
macromolecular therapeutics in cancer chemotherapy: Mechanism of
tumoritropic accumulation of proteins and the antitumor agent
smancs. Cancer Res. 46:6387–6392. 1986.PubMed/NCBI
|
|
104
|
Maeda H: Vascular permeability in cancer
and infection as related to macromolecular drug delivery, with
emphasis on the EPR effect for tumor-selective drug targeting. Proc
Jpn Acad Ser B Phys Biol Sci. 88:53–71. 2012.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Adler-Moore J and Proffitt RT: AmBisome:
Liposomal formulation, structure, mechanism of action and
pre-clinical experience. J Antimicrob Chemother. 49 (Suppl
1):S21–S30. 2002.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Moghimi SM, Hunter AC and Murray JC:
Nanomedicine: Current status and future prospects. FASEB J.
19:311–330. 2005.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Jacobo-Delgado YM, Navarro-Tovar G and
Rivas-Santiago B: Potential use of liposomes in tuberculosis
treatment. Rev Med Inst Mex Seguro Soc. 61:661–669. 2023.PubMed/NCBI View Article : Google Scholar
|
|
108
|
des Rieux A, Fievez V, Garinot M,
Schneider YJ and Préat V: Nanoparticles as potential oral delivery
systems of proteins and vaccines: A mechanistic approach. J Control
Release. 116:1–27. 2006.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Anand P, Kunnumakkara AB, Newman RA and
Aggarwal BB: Bioavailability of curcumin: Problems and promises.
Mol Pharm. 4:807–818. 2007.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Zhang J, Peng Y, Hu S, Xu J, Yu C, Hua L,
Zhou S and Liu Q: Therapeutic application of curcumin and its
nanoformulation in dentistry: Opportunities and challenges. AIMS
Mol Sci. 12:148–172. 2025.
|
|
111
|
Liu Y, Yu T, Zhang C, Yang Z, Yu D, He B
and Liang Y: Nanostructured delivery systems for curcumin:
Improving bioavailability and plaque-targeting efficacy in
atherosclerosis. Pharmaceutics. 17(1465)2025.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Alshammari EM: Curcumin-based
biocompatible nanocarriers: A contemporary perspective in
functional foods and biomedical applications. Discover Nano.
20(226)2025.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Knop K, Hoogenboom R, Fischer D and
Schubert US: Poly(ethylene glycol) in drug delivery: Pros and cons
as well as potential alternatives. Angew Chem Int Ed Engl.
49:6288–6308. 2010.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Makharadze D, Del Valle LJ, Katsarava R
and Puiggalí J: The art of PEGylation: From simple polymer to
sophisticated drug delivery system. Int J Mol Sci.
26(3102)2025.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Krishnan J, Poomalai P, Ravichandran A,
Reddy A and Sureshkumar R: A concise review on effect of PEGylation
on the properties of lipid-based nanoparticles. Assay Drug Dev
Technol. 22:246–264. 2024.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Rattan R, Bhattacharjee S, Zong H, Swain
C, Siddiqui MA, Visovatti SH, Kanthi Y, Desai S, Pinsky DJ and
Goonewardena SN: Nanoparticle-macrophage interactions: A balance
between clearance and cell-specific targeting. Bioorg Med Chem.
25:4487–4496. 2017.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Suk JS, Xu Q, Kim N, Hanes J and Ensign
LM: PEGylation as a strategy for improving nanoparticle-based drug
and gene delivery. Adv Drug Deliv Rev. 99:28–51. 2016.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Gabizon AA, Gabizon-Peretz S,
Modaresahmadi S and La-Beck NM: Thirty years from FDA approval of
pegylated liposomal doxorubicin (Doxil/Caelyx): An updated analysis
and future perspective. BMJ Oncol. 4(e000573)2025.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Santhanakrishnan KR, Koilpillai J and
Narayanasamy D: PEGylation in pharmaceutical development: Current
status and emerging trends in macromolecular and immunotherapeutic
drugs. Cureus. 16(e66669)2024.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Kashyap S, Singh A, Mishra A and Singh V:
Enhanced sustained release of furosemide in long circulating
chitosan-conjugated PLGA nanoparticles. Res Pharm Sci. 14:93–106.
2019.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Abdullah SA, Ansari MJ, Anwer MK, Ganaie
MA, Iqbal M, Alshahrani SM, Alalaiwe AS, Alsulays BB, Alshehri S
and Sulta AS: Enhanced oral bioavailability of ibrutinib
encapsulated poly (lactic-co-glycolic acid) nanoparticles:
pharmacokinetic evaluation in rats. Curr Pharm Anal. 15:661–668.
2019.
|
|
122
|
Wilhelm S, Tavares AJ, Dai Q, Ohta S,
Audet J, Dvorak HF and Chan WCW: Analysis of nanoparticle delivery
to tumours. Nat Rev Mater. 1(16014)2016.
|
|
123
|
Bertrand N, Wu J, Xu X, Kamaly N and
Farokhzad OC: Cancer nanotechnology: The impact of passive and
active targeting in the era of modern cancer biology. Adv Drug
Deliv Rev. 66:2–25. 2014.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Shraddha P, Gorakhnath B, Aniket S, Tanuja
G and Vishakha K: Recent advances in nanoparticles-based drug
delivery systems. Int J Sci R Tech. 2:53–61. 2025.
|
|
125
|
Kumar T, Pandit V and Singh R: Development
and evaluation of PLGA nanoparticles loaded allicin for breast
cancer. J Chem Health Risks. 13:871–889. 2023.
|
|
126
|
Lim M, Fletcher NL, Saunus JM, McCart Reed
AE, Chittoory H, Simpson PT, Thurecht KJ and Lakhani SR: Targeted
hyperbranched nanoparticles for delivery of doxorubicin in breast
cancer brain metastasis. Mol Pharm. 20:6169–6183. 2023.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Cheng B, Ahn HH, Nam H, Jiang Z, Gao FJ,
Minn I and Pomper MG: A unique core-shell structured, glycol
chitosan-based nanoparticle achieves cancer-selective gene delivery
with reduced off-target effects. Pharmaceutics.
14(373)2022.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Li M, Wu J, Yang T, Zhao Y, Ren P, Chang
L, Shi P, Yang J, Liu Y, Li X, et al: Engineered biomimetic
nanoparticles-mediated targeting delivery of allicin against
myocardial ischemia-reperfusion injury by inhibiting ferroptosis.
Int J Nanomedicine. 19:11275–11292. 2024.PubMed/NCBI View Article : Google Scholar
|
|
129
|
De Clercq E: New nucleoside analogues for
the treatment of hemorrhagic fever virus infections. Chem Asian J.
14:3962–3968. 2019.PubMed/NCBI View Article : Google Scholar
|
|
130
|
Nakamoto M, Kunimura K, Suzuki JI and
Kodera Y: Antimicrobial properties of hydrophobic compounds in
garlic: Allicin, vinyldithiin, ajoene and diallyl polysulfides. Exp
Ther Med. 19:1550–1553. 2020.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Hu J, Li C, Zhou Y, Ding J, Li X and Li Y:
Allicin inhibits porcine reproductive and respiratory syndrome
virus infection in vitro and alleviates inflammatory responses.
Viruses. 15(1050)2023.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Jikah AN and Edo GI: Mechanisms of action
by sulphur compounds in Allium sativum. A review. Pharmacol
Res Mod Chin Med. 9(100323)2023.
|
|
133
|
Dhote AM, Patil VR, Lokwani DK, Amnerkar
ND, Ugale VG, Charbe NB, Bhongade BA and Khadse SC: Strategic
analyses to identify key structural features of
antiviral/antimalarial compounds for their binding interactions
with 3CLpro, PLpro and RdRp of SARS-CoV-2: In silico molecular
docking and dynamic simulation studies. J Biomol Struct Dyn.
40:11914–11931. 2022.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Ashraf H, Dilshad E, Afsar T, Almajwal A,
Shafique H and Razak S: Molecular screening of bioactive compounds
of garlic for therapeutic effects against COVID-19. Biomedicines.
11(643)2023.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Rajendran M, Roy S, Ravichandran K, Mishra
B, Gupta DK, Nagarajan S, Arul Selvaraj RC and Provaznik I: In
silico screening and molecular dynamics of phytochemicals from
Indian cuisine against SARS-CoV-2 MPro. J Biomol Struct
Dyn. 40:3155–3169. 2022.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Wu H, Pang MM, Li YL, Hong JH, Liu PM,
Bian M and Yang JJ: Allicin aplealleviates gouty arthritis by
regulating the gut-joint axis, reducing XOD activity, inhibiting
oxidative stress, and suppressing NLRP3 inflammasome activation.
Drug Des Devel Ther. 19:7887–7907. 2025.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Mösbauer K, Fritsch VN, Adrian L,
Bernhardt J, Gruhlke MCH, Slusarenko AJ, Niemeyer D and Antelmann
H: The effect of allicin on the proteome of SARS-CoV-2 infected
calu-3 cells. Front Microbiol. 12(746795)2021.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Mocayar Marón FJ, Camargo AB and Manucha
W: Allicin pharmacology: Common molecular mechanisms against
neuroinflammation and cardiovascular diseases. Life Sci.
249(117513)2020.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Jiang H, Wang X, Lei W, Song P and Cheng
J: Allicin alleviates cerebral ischemia/reperfusion injury by
inhibiting neuronal mitochondrial oxidative stress via OTUD1/NRF2
axis. Phytomedicine. 148(157468)2025.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Nan B, Yang C, Li L, Ye H, Yan H, Wang M
and Yuan Y: Allicin alleviated acrylamide-induced NLRP3
inflammasome activation via oxidative stress and endoplasmic
reticulum stress in Kupffer cells and SD rats liver. Food Chem
Toxicol. 148(111937)2021.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Zhang XN, Zhao N, Guo FF, Wang YR, Liu SX
and Zeng T: Diallyl disulfide suppresses the
lipopolysaccharide-driven inflammatory response of macrophages by
activating the Nrf2 pathway. Food Chem Toxicol.
159(112760)2022.PubMed/NCBI View Article : Google Scholar
|
|
142
|
Zeng T, Zhang CL, Song FY, Zhao XL, Yu LH,
Zhu ZP and Xie KQ: The activation of HO-1/Nrf-2 contributes to the
protective effects of diallyl disulfide (DADS) against
ethanol-induced oxidative stress. Biochim Biophys Acta.
1830:4848–4859. 2013.PubMed/NCBI View Article : Google Scholar
|
|
143
|
Bouyahya A, Kamari F, El Omari N, Touhtouh
J, Mssillou I, Aanniz T, Benali T, Khalid A, Abdalla AN, Iesa MAM,
et al: Unlocking the natural chemical sources, nutritional
properties, biological effects, and molecular actions of Ajoene. J
Funct Foods. 127(106714)2025.
|
|
144
|
Jin X, Wang C, Chen C, Hai S, Rahman SU,
Zhao C, Huang W, Feng S and Wang X: Allicin attenuates the
oxidative damage induced by Aflatoxin B1 in dairy cow
hepatocytes via the Nrf2 signalling pathway. Ecotoxicol Environ
Saf. 280(116527)2024.PubMed/NCBI View Article : Google Scholar
|
|
145
|
Xu C, Mathews AE, Rodrigues C, Eudy BJ,
Rowe CA, O'Donoughue A and Percival SS: Aged garlic extract
supplementation modifies inflammation and immunity of adults with
obesity: A randomized, double-blind, placebo-controlled clinical
trial. Clin Nutr ESPEN. 24:148–155. 2018.PubMed/NCBI View Article : Google Scholar
|
|
146
|
Patya M, Zahalka MA, Vanichkin A, Rabinkov
A, Miron T, Mirelman D, Wilchek M, Lander HM and Novogrodsky A:
Allicin stimulates lymphocytes and elicits an antitumor effect: A
possible role of p21ras. Int Immunol. 16:275–281. 2004.PubMed/NCBI View Article : Google Scholar
|
|
147
|
Schäfer G and Kaschula CH: The
immunomodulation and anti-inflammatory effects of garlic
organosulfur compounds in cancer chemoprevention. Anticancer Agents
Med Chem. 14:233–240. 2014.PubMed/NCBI View Article : Google Scholar
|
|
148
|
Zhou G, Han S, Shi Y and Sui H: Inhibition
of respiratory syncytial virus (RSV) by moroxydine, allicin,
alpha-interferon and their joint application. Zhonghua Shi Yan He
Lin Chuang Bing Du Xue Za Zhi. 11:286–289. 1997.PubMed/NCBI(In Chinese).
|
|
149
|
Srivastava R, Dubey NK, Sharma M, Kharkwal
H, Bajpai R and Srivastava R: Boosting the human antiviral response
in conjunction with natural plant products. Front Nat Prod.
3(2024)2025.
|
|
150
|
Gideon M: Transformative natural
product-drug combinations: Advancing techniques to enhance efficacy
against drug-resistant pathogens. INNOSC Theranostics Pharmacol
Sci. 8:87–95. 2025.
|
|
151
|
Block E: Garlic and other alliums: The
lore and the science. The Royal Society of Chemistry: Cambridge,
U.K., pp454, 2010.
|
|
152
|
Xu N, Han W, Yun G and Shi L: Allicin
protects renal function, improves oxidative stress and lipid
peroxidation in rats with chronic renal failure. Iran J Kidney Dis.
17:135–140. 2023.PubMed/NCBI
|
|
153
|
Bauer D, Redmon N, Mazzio E, Taka E,
Reuben JS, Day A, Sadrud-Din S, Flores-Rozas H, Soliman KF and
Darling-Reed S: Diallyl disulfide inhibits TNFα induced CCL2
release through MAPK/ERK and NF-kappa-B signaling. Cytokine.
75:117–126. 2015.PubMed/NCBI View Article : Google Scholar
|
|
154
|
Raza ZA, Khalil S, Ayub A and Banat IM:
Recent developments in chitosan encapsulation of various active
ingredients for multifunctional applications. Carbohydrate Res.
492(108004)2020.PubMed/NCBI View Article : Google Scholar
|
|
155
|
Mašková L, Závišová L, Kašpar O, Knejzlík
Z, Rimpelová S and Tokárová V: Design and evaluation of composite
films for in situ synthesis and antibacterial activity of allicin
vapour. J Mater Sci. 59:13614–13631. 2024.
|
|
156
|
Wang L, Jiao H, Zhao J, Wang X, Sun S and
Lin H: Allicin alleviates reticuloendotheliosis virus-induced
immunosuppression via ERK/mitogen-activated protein kinase pathway
in specific pathogen-free chickens. Front Immunol.
8(1856)2017.PubMed/NCBI View Article : Google Scholar
|
|
157
|
Yaghoubian H, Niktale H, Yazdi AP, Ghorani
V, Rashed MM and Hashemian AM: Evaluate the therapeutic effect of
allicin (L-cysteine) on clinical presentation and prognosis in
patients with COVID-19. Eur J Transl Myol. 31(9518)2021.PubMed/NCBI View Article : Google Scholar
|
|
158
|
Nahas R and Balla A: Complementary and
alternative medicine for prevention and treatment of the common
cold. Can Fam Physician. 57:31–36. 2011.PubMed/NCBI
|
|
159
|
Bülbül EN and Serdaroğlu KE: The toxic
effect ways of allicin on different cell lines. J Neuro Sci.
10:49–56. 2023.
|
|
160
|
Cascajosa-Lira A, Andreo-Martínez P,
Prieto AI, Baños A, Guillamón E, Jos A and Cameán AM: In vitro
toxicity studies of bioactive organosulfur compounds from Allium
spp. with potential application in the agri-food industry: A
review. Foods. 11(2620)2022.PubMed/NCBI View Article : Google Scholar
|
|
161
|
Ayrle H, Mevissen M, Kaske M, Nathues H,
Gruetzner N, Melzig M and Walkenhorst M: Medicinal
plants-prophylactic and therapeutic options for gastrointestinal
and respiratory diseases in calves and piglets? A systematic
review. BMC Vet Res. 12(89)2016.PubMed/NCBI View Article : Google Scholar
|
|
162
|
Bhuker S, Kaur A, Rajauria K, Tuli HS,
Saini AK, Saini RV and Gupta M: Allicin: A promising modulator of
apoptosis and survival signaling in cancer. Med Oncol.
41(210)2024.PubMed/NCBI View Article : Google Scholar
|
|
163
|
Narain NM, Amoako DG, Somboro AM, Arhin I,
Mhlongo NN, Kumalo HM and Khan RB: Allicin induces
post-translational modifications of p53, DNA damage, and oxidative
stress in human embryonic kidney cells. J Biotech Res. 13:90–102.
2022.
|
|
164
|
Yu CQ, Cai ZL, Yan YB, Zhang YN, Pan RP
and Ling CQ: Inhibitory effect of allicin on growth of human ovary
cancer cell line HO8910. J Anhui Tradit Chin Med Coll. 20:34–36.
2001.
|
|
165
|
Mohammed MO, Muhamad FA and Dyary HO:
Allicin attenuates doxorubicin-induced testicular and systemic
toxicity in rats via antioxidant and anti-apoptotic mechanisms.
Reprod Toxicol. 138(109082)2025.PubMed/NCBI View Article : Google Scholar
|
|
166
|
Banerjee SK, Mukherjee PK and Maulik SK:
Garlic as an antioxidant: The good, the bad and the ugly. Phytother
Res. 17:97–106. 2003.PubMed/NCBI View Article : Google Scholar
|
|
167
|
Sultan SA, Khawaji MH, Alsughayyir J,
Alfhili MA, Alamri HS and Alrfaei BM: Antileukemic activity of
sulfoxide nutraceutical allicin against THP-1 cells is associated
with premature phosphatidylserine exposure in human erythrocytes.
Saudi J Biol Sci. 27:3376–3384. 2020.PubMed/NCBI View Article : Google Scholar
|
|
168
|
Rana SV, Pal R, Vaiphei K and Singh K:
Garlic hepatotoxicity: Safe dose of garlic. Trop Gastroenterol.
27:26–30. 2006.PubMed/NCBI
|
|
169
|
Alnaqeeb MA, Thomson M, Bordia T and Ali
M: Histopathological effects of garlic on liver and lung of rats.
Toxicol Lett. 85:157–164. 1996.PubMed/NCBI View Article : Google Scholar
|
|
170
|
Wang ET, Chen DY, Liu HY, Yan HY and Yuan
Y: Protective effect of allicin against glycidamide-induced
toxicity in male and female mice. Gen Physiol Biophys. 34:177–187.
2015.PubMed/NCBI View Article : Google Scholar
|
|
171
|
Orabi SH, Abd Eldaium D, Hassan A, Sabagh
HSE and Abd Eldaim MA: Allicin modulates diclofenac sodium induced
hepatonephro toxicity in rats via reducing oxidative stress and
caspase 3 protein expression. Environ Toxicol Pharmacol.
74(103306)2020.PubMed/NCBI View Article : Google Scholar
|
|
172
|
Martignoni M, Groothuis GMM and de Kanter
R: Species differences between mouse, rat, dog, monkey and human
CYP-mediated drug metabolism, inhibition and induction. Expert Opin
Drug Metab Toxicol. 2:875–894. 2006.PubMed/NCBI View Article : Google Scholar
|
|
173
|
U.S. Food and Frug Administration (FDA):
S9 nonclinical evaluation for anticancer pharmaceuticals. FDA,
Rockville, MD, 2010.
|
|
174
|
Borrelli F, Capasso R and Izzo AA: Garlic
(Allium sativum L.): Adverse effects and drug interactions
in humans. Mol Nutr Food Res. 51:1386–1397. 2007.PubMed/NCBI View Article : Google Scholar
|
|
175
|
Tattelman E: Health effects of garlic. Am
Fam Physician. 72:103–106. 2005.PubMed/NCBI
|
|
176
|
Ho BE, Shen DD, McCune JS, Bui T, Risler
L, Yang Z and Ho RJY: Effects of garlic on cytochromes P450 2C9-
and 3A4-mediated drug metabolism in human hepatocytes. Sci Pharm.
78:473–481. 2010.PubMed/NCBI View Article : Google Scholar
|
|
177
|
Rahman K and Lowe GM: Garlic and
cardiovascular disease: A critical review. J Nutr. 136 (3
Suppl):736S–740S. 2006.PubMed/NCBI View Article : Google Scholar
|
|
178
|
Izzo AA and Ernst E: Interactions between
herbal medicines and prescribed drugs: A systematic review. Drugs.
61:2163–2175. 2001.PubMed/NCBI View Article : Google Scholar
|
|
179
|
Vaes LP and Chyka PA: Interactions of
warfarin with garlic, ginger, ginkgo, or ginseng: Nature of the
evidence. Ann Pharmacother. 34:1478–1482. 2000.PubMed/NCBI View Article : Google Scholar
|
|
180
|
Chen XW, Serag ES, Sneed KB, Liang J, Chew
H, Pan SY and Zhou SF: Clinical herbal interactions with
conventional drugs: From molecules to maladies. Curr Med Chem.
18:4836–4850. 2011.PubMed/NCBI View Article : Google Scholar
|
|
181
|
Piscitelli SC, Burstein AH, Welden N,
Gallicano KD and Falloon J: The effect of garlic supplements on the
pharmacokinetics of saquinavir. Clin Infect Dis. 34:234–238.
2002.PubMed/NCBI View
Article : Google Scholar
|
|
182
|
Kansara MB and Jani AJ: Possible
interactions between garlic and conventional drugs: A review. Pharm
Biol Eval. 4:73–81. 2017.
|
|
183
|
Wang J, Zhang X, Lan H and Wang W: Effect
of garlic supplement in the management of type 2 diabetes mellitus
(T2DM): A meta-analysis of randomized controlled trials. Food Nutr
Res. 61(1377571)2017.PubMed/NCBI View Article : Google Scholar
|
|
184
|
Hou LQ, Liu YH and Zhang YY: Garlic intake
lowers fasting blood glucose: Meta-analysis of randomized
controlled trials. Asia Pac J Clin Nutr. 24:575–582.
2015.PubMed/NCBI View Article : Google Scholar
|
|
185
|
Xiong XJ, Wang PQ, Li SJ, Li XK, Zhang YQ
and Wang J: Garlic for hypertension: A systematic review and
meta-analysis of randomized controlled trials. Phytomedicine.
22:352–361. 2015.PubMed/NCBI View Article : Google Scholar
|
|
186
|
Ried K: Garlic lowers blood pressure in
hypertensive individuals, regulates serum cholesterol, and
stimulates immunity: An updated meta-analysis and review. J Nutr.
146:389S–396S. 2016.PubMed/NCBI View Article : Google Scholar
|
|
187
|
Izzo AA, Hoon-Kim S, Radhakrishnan R and
Williamson EM: A critical approach to evaluating clinical efficacy,
adverse events and drug interactions of herbal remedies. Phytother
Res. 30:691–700. 2016.PubMed/NCBI View Article : Google Scholar
|
|
188
|
Olivera EJ and Palacios C: Use of
supplements in Puerto Rican older adults residing in an elderly
project. P R Health Sci J. 31:213–219. 2012.PubMed/NCBI
|
|
189
|
Berginc K and Kristl A: The mechanisms
responsible for garlic-drug interactions and their in vivo
relevance. Curr Drug Metab. 14:90–101. 2013.PubMed/NCBI
|