Introduction
The growth plate and its primary cell type, the
chondrocyte, are integral to endochondral ossification and thus the
linear growth of the long bones (1). The continuing development of in
vitro chondrocyte cell lines has furthered our understanding of
the underlying mechanisms of endochondral ossification.
The ATDC5 cell line, which was first isolated from
the differentiating teratocarcinoma stem cell line AT805, is
commonly used as a model for in vitro chondrocyte research
(2). To date, the ATDC5 cell line
has been utilised in approximately 300 studies. Previous studies
have detailed a well-characterised method of ATDC5 differentiation
and mineralisation, initially by Shukunami et al (3). This method has provided a reliable
model of in vitro chondrocyte mineralisation for a number of
years and has been widely used in the field since its publication;
however it does contain some drawbacks. For example, mineralisation
studies require a culture time of at least 34 days and a change of
culture conditions. Both the cell culture medium and the
CO2 concentration have to be altered after 21 days of
culture to facilitate extracellular matrix (ECM) mineralisation 13
days later. Since its publication, a number of groups have
attempted to simplify the culture method. For example, the addition
of inorganic phosphate to ATDC5 cultures has been shown to increase
differentiation and the rate of ECM mineralisation (4,5).
Another study has detailed that the addition of ascorbic acid
shortened the proliferation phase of the ATDC5 cells from 21 to 7
days (6); while the temporal
expression of markers of chondrogenic differentiation was examined,
the ECM mineralisation capability of the ATDC5 cells under these
culture conditions was not.
Therefore, our aim was to develop a culture model
for ATDC5 cells which produced both consistent chondrogenesis and
physiological ECM mineralisation in a reduced time period for in
vitro experimentation. In this study, β-glycerophosphate (βGP)
was added throughout the culture period. βGP is cleaved by alkaline
phosphatase (ALP) and other phosphohydrolases produced by the
chondrocytes once they have reached hypertrophy to release
inorganic phosphate, thus mimicking the phosphate availability
in vivo (3,7,8).
It was hypothesised that this strategy would facilitate an
incremental increase in mineral deposition once an appropriate ECM
had been deposited. This would thereby increase the rate of
mineralisation compared with previous methods while retaining the
expected stages of chondrogenic differentiation as well as
crucially, the formation of physiological mineral.
Materials and methods
Cell culture
Chondrogenic ATDC5 cells (Riken Cell Bank, Ibaraki,
Japan) (3) were cultured in a
differentiation medium [DMEM/F-12 (1:1) with GlutaMAX I containing
5% FBS, 1% insulin transferrin and selenium, 1% sodium pyruvate and
0.5% gentamicin (Invitrogen, Paisley, UK)] at a density of 6,000
cells/cm2 in multi-well plates (Iwaki Cell Biology;
Sterilin, Feltham, UK) (9,10).
Cells were left for 6 days to reach confluency at which point the
medium was supplemented with 10 mM βGP and 50 μg/ml
L-ascorbate-2-phosphate (ascorbic acid). Cells were incubated in a
humidified atmosphere (37°C, 5% CO2) for up to 41 days
and the medium was changed every second or third day. For
levamisole experiments, ATDC5 cells were cultured in varying
concentrations of levamisole (Sigma, Gillingham, UK) (0–1,000 μM)
for up to 15 days.
Histochemical staining
Calcium deposition in ATDC5 cells was evaluated by
Alizarin red staining as described previously (11). Briefly, cells were fixed in 4%
paraformaldehyde and then 2% Alizarin red (Sigma, pH 4.2) was added
to the cell layers for 5 min at room temperature. Cells were washed
with distilled water (dH20) and images were captured.
Alizarin red-stained cultures were extracted with 10%
cetylpyridinium chloride for 10 min and the optical density (OD) of
the digests was measured at 570 nm by spectrophotometry (Multiskan
Ascent; Thermo Electron Corporation, Vantaa, Finland). Proteoglycan
synthesis was evaluated by staining the cell layers with Alcian
blue (Sigma). Cells were fixed in 95% methanol for 20 min and
stained with 1% Alcian blue 8GX in 0.1 M HCl overnight. Cells were
washed in dH20 and images were captured. Alcian
blue-stained cultures were extracted with 6 M guanidine-HCl for 6 h
at room temperature and the OD was determined at 630 nm by
spectrophotometry (11).
Real-time quantitative PCR (qRT-PCR)
RNA was extracted using the RNeasy Mini kit (Qiagen
Ltd., Crawley, West Sussex, UK), according to the manufacturer’s
instructions. For each sample, total RNA content was assessed by
absorbance at 260 nm and purity by A260/A280 ratios, and then
reverse-transcribed. cDNA was diluted to 10 ng/μl in nuclease-free
water (Sigma), and stored at −20°C. qRT-PCR reactions were
conducted with a MX3000P qPCR machine (Stratagene, Stockport, UK)
using a SYBR-Green detection method. Primers were designed in-house
and synthesised by MWG Eurofins, London, UK. Reactions were run in
triplicate and routinely normalised against GAPDH. Primer
sequences: Col2a1, forward, 5′-CGGTCCTACGGTGTCAGG-3′ and
reverse, 5′-GCAGAGGACATTCCCAGTGT-3′; Col10a1, for wa rd,
5′-CATA A AG G GCCCACT TGCTA-3′ and
reverse,5′-CAGGAATGCCTTGTTCTCCT-3′; GAPDH forward,
5′-TGAGGCCGGTGCTGAGTATGTCG-3′ and reverse,
5′-CCACAGTCTTCTGGGTGGCAGTG-3′. qRT-PCR products were sequenced by
the GenePool, University of Edinburgh.
Transmission electron microscopy
(TEM)
ATDC5 cells were cultured at 6,000
cells/cm2 on nitrocellulose discs (Nunc, Roskilde,
Denmark) in mineralising conditions for 15 days. Cells were fixed
in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer at 37°C
for 1 h. During processing, the cell monolayers were washed in 0.1
M sodium cacodylate, post-fixed in 1% osmium tetroxide and
dehydrated through graded alcohols (35, 70, 95 and 100%). The
monolayers were then processed to Epon in a vacuum oven at 60°C.
Monolayers were viewed using a Phillips CMIRO TEM (FEI Vic Ltd.,
Cambridge, UK) and images were captured on Gatan Orius ICD camera
(Gatan, Oxford, UK).
Fourier transform-infrared spectroscopy
(FTIR)
ATDC5 cells were cultured for 41 days in
mineralising conditions as previously described. Cell monolayers
were fixed in 95% methanol and embedded in LR White. Spectral
images of 2 μm-thick culture sections were collected using a
Spectrum Spotlight 100 system (Perkin-Elmer, Waltham, MA, USA) with
a spectral resolution of 4 μm and 6.25 μm pixel size in
transmission mode. The collected spectra were truncated, base-lined
and the contribution of LR White was spectrally subtracted using
ISYS software (Spectral Dimensions, Olney, MD, USA) and then
analysed using ISys Chemical Imaging software. Spectra extracted
from these images were analysed using Grams/32 software (Thermo
Electron Corporation, Waltham, MA, USA). The parameters measured
included mineral/matrix ratio, carbonate/phosphate ratio,
crystallinity and collagen maturity (12).
Statistics
Data were analysed by one-way analysis of variance
(ANOVA), with Tukey simultaneous tests used to identify differences
between individual time-points, using SigmaPlot 11.0 software
(Systat Software UK Ltd., London, UK). Cell culture experiments
were repeated at least twice and P<0.05 was considered
statistically significant.
Results
ATDC5 cells undergo the expected stages
of chondrocyte differentiation
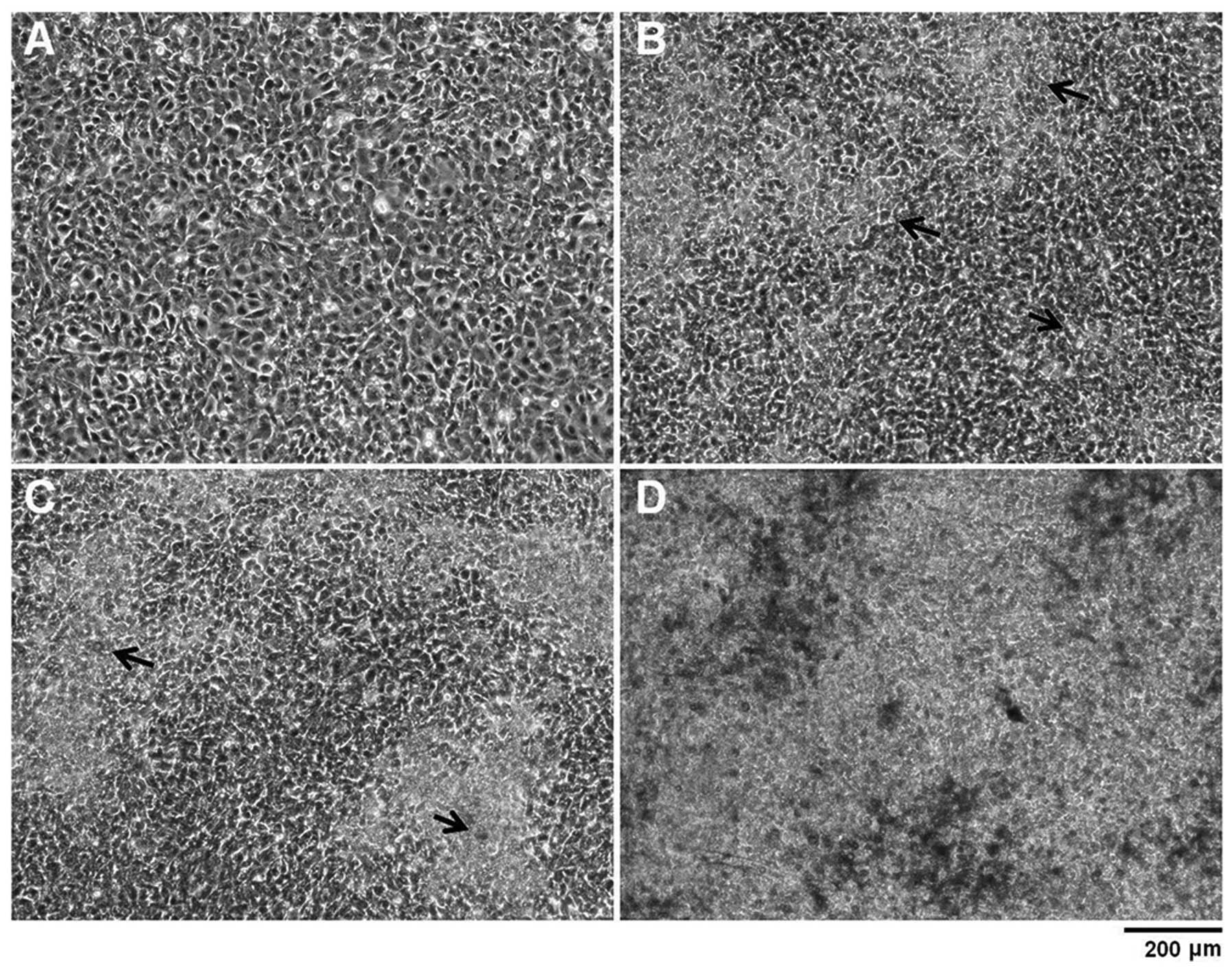
Images collected by light microscopy over a 34-day
time-course indicated comparable differentiation to previously
characterised ATDC5 cultures (3,4).
ATDC5 cell cultures reached confluency 6 days after seeding with no
extensive ECM formation (Fig.
1A). At this point, ATDC5 cells were then cultured in the
presence of 10 mM βGP and 50 μg/ml ascorbic acid. This facilitates
cell differentiation and the secretion of an extensive ECM which
assembles around the cells as visualised at Day 13 of culture by
the phase contrast images (Fig.
1B) and by Alcian blue staining (data not shown). As
differentiation continues, these nodules increased in area and
began to conjoin (Fig. 1C). A
large proportion of each ATDC5 monolayer was Alcian blue-positive
by Day 34 of culture, indicating that ATDC5 cells produced a
glycosaminoglycan (GAG)-rich ECM and underwent chondrogenesis
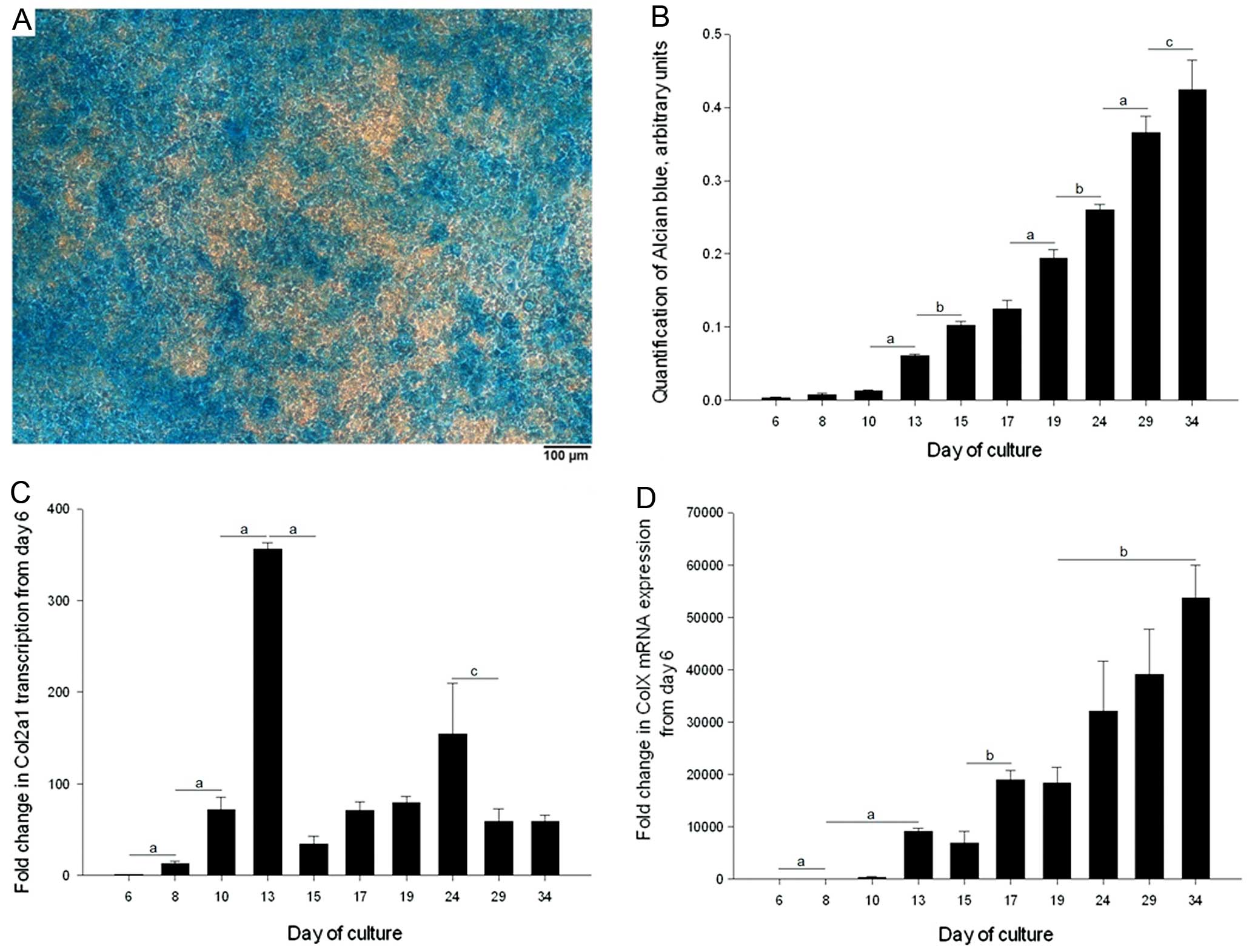
(Fig. 2A). The temporal increase
in GAG-deposition over a 34-day time-course was established by
quantifying Alcian blue staining (Fig. 2B). GAG-deposition progressively
increased from Day 6, such that there were significant increases in
Alcian blue staining between various time-points within the culture
period.
To examine the process of chondrogenesis and
hypertrophic differentiation of the ATDC5 cells during monolayer
culture, Col2a1 and Col10a1 gene transcription at
specific time-points was analysed by qRT-PCR. Col2a1
transcription increased significantly between each time-point from
Day 6 to 13; transcription then decreased but even at Day 34,
transcription was greater compared to Day 6 (by >58-fold,
P<0.001) (Fig. 2C).
Col10a1 transcription also increased significantly over the
first 4 time-points up to Day 13 over a 10,000-fold range, which by
Day 34 progressed to a greater than 50,000-fold increase in
transcription compared to that of Day 6 (Fig. 2D).
ATDC5 cells mineralise their surrounding
ECM, producing physiological mineral
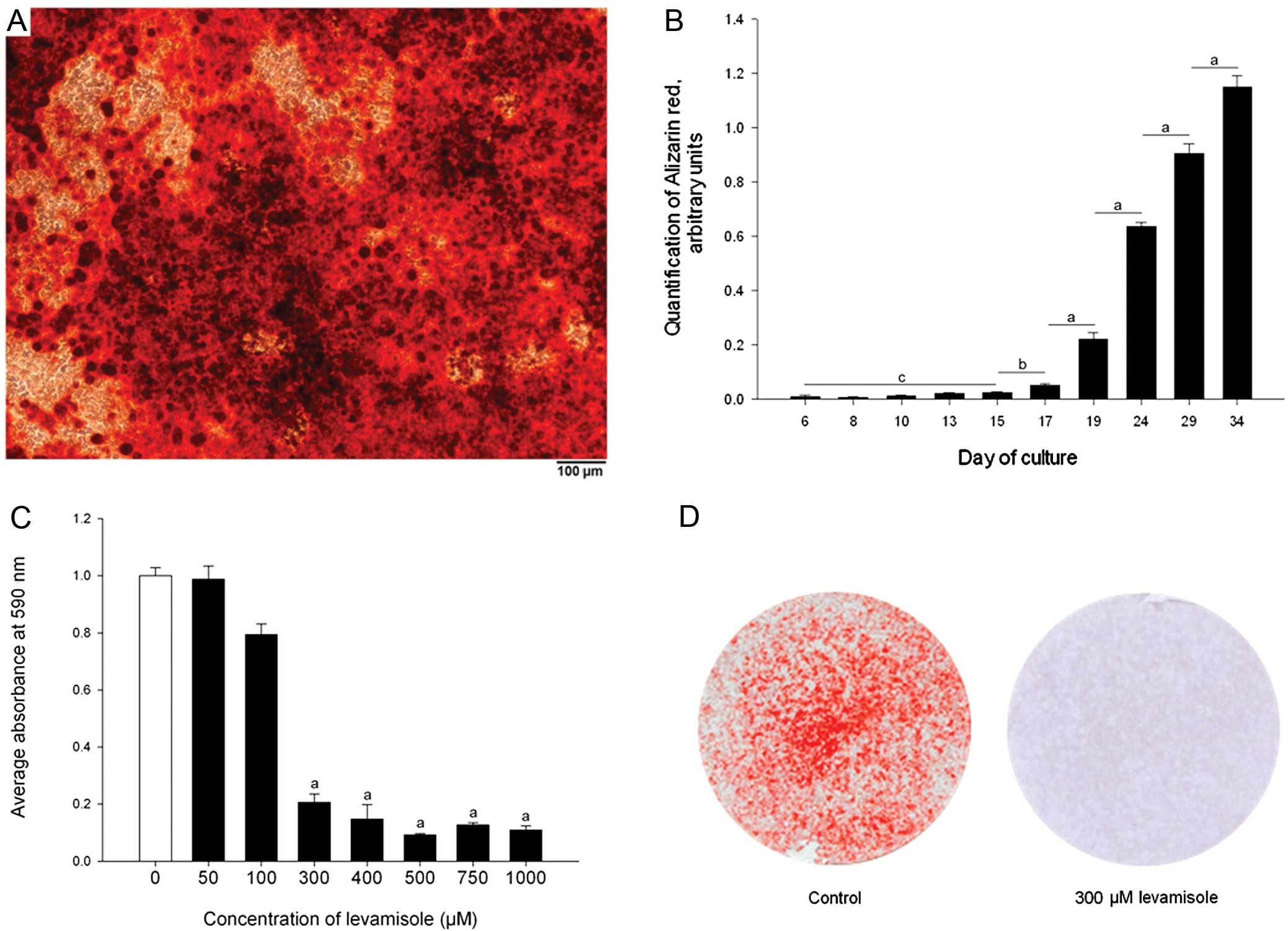
Phase contrast images indicated mineralisation of
the ATDC5 ECM from Day 14 onwards and this was confirmed by
Alizarin red staining over a 34-day time-course (Fig. 1C and D, Fig. 3A and B). Quantification of this
staining indicated that after an initial delay, presumably while
early differentiation stages were occurring, calcium accumulation
increased rapidly from Day 17 to 34 (Fig. 3B) (P<0.001).
Levamisole, a well established inhibitor of ALP,
inhibited ATDC5 ECM mineralisation at Day 15 of culture at
concentrations in excess of 300 μM (P<0.001) (Fig. 3C) with no apparent alterations in
the morphology of the ATDC5 cells (Fig. 3D) (13). This indicated that the enzyme ALP
is required, and therefore that chondrogenic differentiation of the
ATDC5 cells is necessary for effective mineralisation.
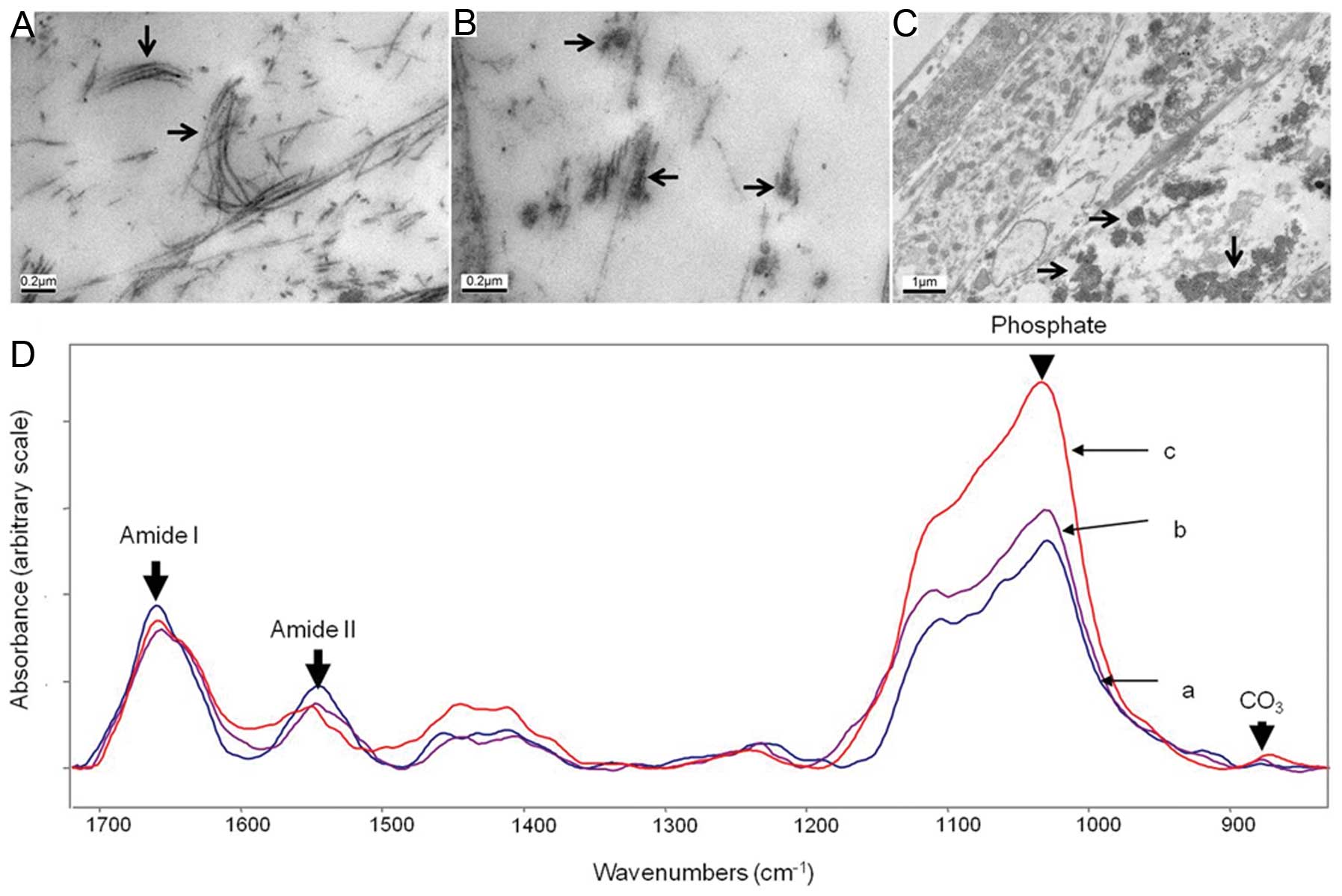
FTIR and TEM were adopted as two well recognised
methods to determine whether the properties of the mineral formed
in culture is similar to that which is formed by mineralised
cartilage in vivo (14).
ATDC5 cells were shown to produce a collagenous ECM by TEM in which
banded fibers, synonymous with collagen fibers, were present in the
ECM (Fig. 4A) (15). Along some of the collagen fibers,
electron-dense regions were present, indicative of the onset of
mineralisation (Fig. 4B). In some
discrete regions of the ECM, electron-dense spheres of ∼200–500 nm
were present which were also associated with the collagenous fibers
(Fig. 4C); these are possibly
mineralised matrix vesicles (MVs) (1,8,9).
These results suggest that ATDC5 cells produce a collagenous ECM
and that the mineral formed is in alignment with the collagen
fibrils, as is observed in endochondral ossification. ATDC5 cells
were cultured for 41 days for FTIR analysis (Fig. 4D-a). The FTIR spectra were
compared with those of E14 embryonic mouse bone (Fig. 4D-b) and 4-month-old cortical mouse
bone (Fig. 4D-c), and were used
to generate numerical parameters which may be compared with in
vivo samples (Table I). The
resulting data strongly suggest that the mineralisation of the
ATDC5 monolayers resembles that of embryonic mouse bone.
 | Table I.Mineralisation parameters from FTIR
samples. |
Table I.
Mineralisation parameters from FTIR
samples.
| Sample | Mineral-to-matrix
ratio |
Carbonate-to-mineral ratio | Crystallinity |
|---|
| A | 3.000±0.917 | 0.008±0.005 | 1.128±0.009 |
| B | 2.200±2.500 | 0.005±0.002 | 1.072±0.062 |
| C | 6.500±0.900 | 0.006±0.001 | 1.130±0.030 |
Discussion
Attempts to unravel the underlying mechanisms of
endochondral ossification have been limited by current models. The
data presented in this manuscript characterise a novel, rapid
culture method for studying physiological chondrocyte ECM
mineralisation using ATDC5 cells that we observed to be highly
reproducible. A mineralisation method for ATDC5 cell culture was
first described by Shukunami et al (3), however its drawbacks have been
identified by several other groups and thus the method has been
gradually developed with time. In this study, we cultured ATDC5
cells in the presence of ascorbic acid and 10 mM βGP.
ATDC5 cells have previously been cultured with
ascorbic acid, which facilitates collagen synthesis. This reduces
the proliferation phase of the cells and promotes their
differentiation (6,16). Ascorbic acid has also been shown
to promote the hypertrophic differentiation of cultured primary
chick chondrocytes (17). In the
present study, cells were cultured in the presence of 50 μg/ml
ascorbic acid from when they reached confluency and in concurrence
with previous studies, this promoted ECM formation. The increased
mRNA expression of the chondrogenic marker Col2a1 correlated
with the onset of Alcian blue-stained cartilaginous nodules and the
increased mRNA expression of Col10a1 with the
differentiation of the cells to a hypertrophic phenotype. The
delayed onset of Col10a1 transcription at Day 10 is
consistent with the 2 stages of differentiation that must occur
from Day 6 for the cells to become hypertrophic. The observation
that Col10a1 expression preceded the first observations of
mineral formation provides further evidence that the model is able
to mimic the in vivo endochondral ossification. During
differentiation the histology of the cultures was similar to that
described by Shukunami et al (3).
In addition to ascorbic acid, an exogenous phosphate
source is routinely added to cell cultures to induce and stimulate
mineralisation of the ECM. βGP is a preferential exogenous organic
phosphate source as it is a substrate for ALP and therefore the
cells directly dictate when it is cleaved to release inorganic
phosphate with their differentiation to a hypertrophic phenotype.
In this study we cultured ATDC5 cells in the presence of 10 mM βGP
and observed mineral formation from Day 15 of culture upon collagen
fibrils and within MVs. However, in a number of osteoblast and
chondrocyte cultures, the growth of cells in the presence of βGP
has been shown to lead to the formation of sporadic mineral
formation on the cell surface and in the culture medium and not
upon collagen fibrils which is regarded to be dystrophic
mineralisation and not physiological hydroxyapatite (18,19).
In the present study we showed that the presence of
ALP is necessary for βGP-induced ATDC5 mineralisation. This is
consistent with previous studies in which the activity of ALP has
been investigated in ATDC5 cells (3). Furthermore, the addition of
levamisole, a potent inhibitor of ALP, to ATDC5 cultures inhibited
their ECM mineralisation. Mineralisation was also inhibited in
cells cultured in the presence of βGP and in the absence of
insulin, which is required for their differentiation (data not
shown) (20). This result,
therefore, suggests that mineral formation is dependent upon both
chondrogenic differentiation and the subsequent presence of ALP.
Additionally, the inhibition of ECM mineralisation when ATDC5 cells
were cultured without insulin further emphasises that the mineral
formed is not dystrophic and is dependent on their differentiation
status.
Although routinely used as indicators of
mineralisation, Alizarin red and von Kossa staining are not
sufficient to conclude that mineralisation is physiological since
the presence of calcium and/or phosphate does not indicate HA
formation per se (21).
For this reason, we adopted FTIR and TEM to examine whether or not
the mineral formed in culture is physiological (14).
Shukunami et al (3) have previously used TEM for ATDC5
analysis and reported the presence of extremely dark
calcium-containing spherites which they identified as mineralised
MVs in discrete regions between the cells, associated with
collagenous fibers. In the present study, structures were noted in
our TEM analysis which were indistinguishable in shape and size
from those reported by Shukunami et al (3). The MVs derived from ATDC5 cells are
of a similar size and appearance to MVs derived from in vivo
tissues including chicken growth plate chondrocytes and rat
epiphyseal hypertrophic chondrocytes (22). These results indicate that the
ultrastructure of the collagenous fibrils appears as expected and
that mineralisation of ATDC5 cultures appears to form in a
physiological manner.
Furthermore, in this study we showed that the
spectra of the ATDC5 monolayer more closely resembled that of the
developing embryonic bone compared to the fully developed cortical
bone. The mineral-to-matrix ratio of the ATDC5 cultures, a key
determinant of mineral composition is similar to the values for
mineralised embryonic bone, E14, which is the earliest point at
which mineralisation occurs in the mouse (23). The mineral-matrix ratios in other
publications provide additional comparisons: in 10-day-old and
10-week-old wild-type mouse calcified growth plate cartilage the
ratios have been calculated as 2.7 and 5.48, respectively (24,25). There is a considerable variation
in these parameters, but the mineral-matrix ratio within the ATDC5
monolayer is within the expected region. The ATDC5 monolayer model
characterised by Shukunami et al (3) was analysed by FTIR and spectra were
compared with those of cultured primary rabbit chondrocytes,
resulting in spectra which were almost super-imposable. These
spectra show that the absorbance is greater in the amide-range
compared to the phosphate-range, thus although the mineral-to
matrix ratio is not provided, it is certainly less than those
reported in the present study. Therefore, ECM mineralisation is
greater in this ATDC5 model compared to the method generated by
Shukunami et al (3). If
mineralisation in the cultures is ectopic and mineral is
accumulated simply due to ALP cleavage of βGP, the ratio of
mineral-to-matrix would be expected to be extremely high, which is
not the case. A study by Huitema et al (26), demonstrated this; inorganic
phosphate was added to medium conditioned by ATDC5 cells which
generated flat, mineralised structures, with extremely small
amide-I peaks, relative to phosphate peaks.
The mineralised product of the ATDC5 monolayer
produced a phosphate peak with a clear shoulder at approximately
1,130 cm−1. This is characteristic of the hydroxyapatite
containing acid phosphate which is gradually lost as the crystal
matures; thus both the ATDC5 monolayer and the embryonic bone
contain this peak, which is absent in the mature hydroxyapatite
sample (27,28). The values obtained from the
carbonate substitution and crystallinity are within the range of
biologically relevant in vivo samples, which also indicates
that this ATDC5 model generates physiologically relevant mineral
(24,29).
The development and characterisation of a rapidly
mineralising chondrocyte model has the potential to assist us in
better understanding the underpinning molecular mechanisms
responsible for poor linear bone growth which is observed in a
number of chronic diseases such as cystic fibrosis, chronic kidney
disease, rheumatological conditions and inflammatory bowel disease.
Chondrocyte models, including the ATDC5 cell line, have proved
invaluable for determining the effects of pro-inflammatory
cytokines and glucocorticoids on chondrocyte proliferation,
differentiation and gene expression (9,10).
However, the absence of a practical and accessible in vitro
chondrocyte mineralisation model has hindered a fuller appreciation
of how cartilage mineralisation and endochondral ossification are
disrupted by factors e.g. cytokines and drugs, that are responsible
for impaired linear bone growth in children.
In conclusion, in this study we developed and
characterised an improved and rapid method of ATDC5 differentiation
which develops a physiologic mineralised ECM 15 days after seeding.
To our knowledge, this is the earliest report of mineralisation in
which physiological attributes of the mineral have been
characterised.
Acknowledgements
The authors thank Steve Mitchell,
University of Edinburgh, for assisting with the TEM technique. This
project was funded by the Biotechnology and Biological Sciences
Research Council (BBSRC), a scholarship award from the UK (P.N. and
K.S.), the Institute Strategic Programme Grant Funding (C.F. and
V.M.) and the Institute Career Path Fellowship Funding (V.M.). A.B.
and L.S. received funding from NIH grant AR046121.
References
|
1.
|
HM KronenbergDevelopmental regulation of
the growth plateNature423332336200310.1038/nature0165712748651
|
|
2.
|
T AtsumiY MiwaK KimataY IkawaA
chondrogenic cell line derived from a differentiating culture of
AT805 teratocarcinoma cellsCell Differ
Dev30109116199010.1016/0922-3371(90)90079-C2201423
|
|
3.
|
C ShukunamiK IshizekiT AtsumiY OhtaF
SuzukiY HirakiCellular hypertrophy and calcification of embryonal
carcinoma-derived chondrogenic cell line ATDC5 in vitroJ Bone Miner
Res1211741188199710.1359/jbmr.1997.12.8.11749258747
|
|
4.
|
D MagneG BluteauC FaucheuxG PalmerC
Vignes-ColombeixP PiletT RouillonJ CaverzasioP WeissG DaculsiJ
GuicheuxPhosphate is a specific signal for ATDC5 chondrocyte
maturation and apoptosis-associated mineralization: possible
implication of apoptosis in the regulation of endochondral
ossificationJ Bone Miner
Res1814301442200310.1359/jbmr.2003.18.8.1430
|
|
5.
|
T FujitaT MeguroN IzumoC YasutomiR
FukuyamaH NakamutaM KoidaPhosphate stimulates differentiation and
mineralization of the chondroprogenitor clone ATDC5Jpn J
Pharmacol85278281200110.1254/jjp.85.27811325020
|
|
6.
|
FM AltafTM HeringNH KazmiJU YooB
JohnstoneAscorbate-enhanced chondrogenesis of ATDC5 cellsEur Cell
Mater126470200617096313
|
|
7.
|
MR CoeTA SummersSJ ParsonsAL BoskeyG
BalianMatrix mineralization in hypertrophic chondrocyte cultures.
Beta glycerophosphate increases type X collagen messenger RNA and
the specific activity of pp60c-src kinaseBone Miner18911061992
|
|
8.
|
T MatsuzawaHC AndersonPhosphatases of
epiphyseal cartilage studied by electron microscopic cytochemical
methodsJ Histochem Cytochem19801808197110.1177/19.12.8014335252
|
|
9.
|
VE MacRaeC FarquharsonSF AhmedThe
restricted potential for recovery of growth plate chondrogenesis
and longitudinal bone growth following exposure to pro-inflammatory
cytokinesJ Endocrinol189319328200610.1677/joe.1.0660916648299
|
|
10.
|
HC OwenJN MinerSF AhmedC FarquharsonThe
growth plate sparing effects of the selective glucocorticoid
receptor modulator, AL-438Mol Cell
Endocrinol264164170200710.1016/j.mce.2006.11.00617182172
|
|
11.
|
VE MacRaeMG DaveyL McTeirS NarisawaMC
YadavJL MillanC FarquharsonInhibition of PHOSPHO1 activity results
in impaired skeletal mineralization during limb development of the
chickBone4611461155201010.1016/j.bone.2009.12.01820053388
|
|
12.
|
S Gourion-ArsiquaudPA WestAL BoskeyFourier
transform-infrared microspectroscopy and microscopic imagingMethods
Mol Biol455293303200810.1007/978-1-59745-104-8_2018463826
|
|
13.
|
BH VanAlkaline phosphatase. I. Kinetics
and inhibition by levamisole of purified isoenzymes from humansClin
Chem229729761976
|
|
14.
|
AL BoskeyR RoyCell culture systems for
studies of bone and tooth mineralizationChem
Rev10847164733200810.1021/cr078247318800815
|
|
15.
|
LC HughesCW ArcherI ap GwynnThe
ultrastructure of mouse articular cartilage: collagen orientation
and implications for tissue functionality. A polarised light and
scanning electron microscope study and reviewEur Cell
Mater968842005
|
|
16.
|
TM TemuKY WuPA GruppusoC PhornphutkulThe
mechanism of ascorbic acid-induced differentiation of ATDC5
chondrogenic cellsAm J Physiol Endocrinol
Metab299E325E334201020530736
|
|
17.
|
PS LeboyL VaiasB UschmannE GolubSL AdamsM
PacificiAscorbic acid induces alkaline phosphatase, type X
collagen, and calcium deposition in cultured chick chondrocytesJ
Biol Chem26417281172861989
|
|
18.
|
M RohdeH MayerExocytotic process as a
novel model for mineralization by osteoblasts in vitro and in vivo
determined by electron microscopic analysisCalcif Tissue
Int80323336200710.1007/s00223-007-9013-517406769
|
|
19.
|
G GronowiczFN WoodielMB McCarthyLG RaiszIn
vitro mineralization of fetal rat parietal bones in defined
serum-free medium: effect of beta-glycerol phosphateJ Bone Miner
Res4313324198910.1002/jbmr.56500403052763870
|
|
20.
|
M BorgersThe cytochemical application of
new potent inhibitors of alkaline phosphatasesJ Histochem
Cytochem21812824197310.1177/21.9.8124741290
|
|
21.
|
LF BonewaldSE HarrisJ RosserMR DallasSL
DallasNP CamachoB BoyanA Boskeyvon Kossa staining alone is not
sufficient to confirm that mineralization in vitro represents bone
formationCalcif Tissue
Int72537547200310.1007/s00223-002-1057-y12724828
|
|
22.
|
LN WuBR GengeDG DunkelbergerRZ LeGerosB
ConcannonRE WuthierPhysicochemical characterization of the
nucleational core of matrix vesiclesJ Biol
Chem27244044411199710.1074/jbc.272.7.44049020163
|
|
23.
|
AI CaplanBone developmentCiba Found
Symp1363211988
|
|
24.
|
AL BoskeySB DotyD StinerI BindermanViable
cells are a requirement for in vitro cartilage calcificationCalcif
Tissue Int5817718519968852573
|
|
25.
|
HC AndersonJB SipeL HessleR DhanyamrajuE
AttiNP CamachoJL MillanImpaired calcification around matrix
vesicles of growth plate and bone in alkaline phosphatase-deficient
miceAm J
Pathol164841847200410.1016/S0002-9440(10)63172-014982838
|
|
26.
|
LF HuitemaPR van WeerenBW van BalkomT
VisserCH van de LestA BarneveldJB HelmsAB VaandragerSoluble factors
released by ATDC5 cells affect the formation of calcium phosphate
crystalsBiochim Biophys
Acta177411081117200710.1016/j.bbapap.2007.06.00517669701
|
|
27.
|
GR SauerRE WuthierFourier transform
infrared characterization of mineral phases formed during induction
of mineralization by collagenase-released matrix vesicles in vitroJ
Biol Chem26313718137241988
|
|
28.
|
RZ LeGerosPreparation of octacalcium
phosphate (OCP): a direct fast methodCalcif Tissue
Int37194197198510.1007/BF025548413924374
|
|
29.
|
EP PaschalisO JacenkoB OlsenB
deCrombruggheAL BoskeyThe role of type X collagen in endochondral
ossification as deduced by Fourier transform infrared microscopy
analysisConnect Tissue
Res35371377199610.3109/030082096090292149084677
|