Introduction
Systemic sclerosis (SSc) is a disease characterized
by vasculopathy, activation of the immune system and exaggerated
deposition of extracellular matrix (ECM), resulting in stiff skin
and fibrosis of internal organs. Skin fibrosis in scleroderma may
be dramatic, severely affecting the mobility and quality of life of
a patient, while lung complications are responsible for the
majority of deaths. Several therapeutic options are available for
SSc patients, including immunomodulatory agents, but their efficacy
in reversing fibrosis is controversial, being considered at most
only effective in halting the progression of the skin and lung
disease. Therefore, the development of alternative therapeutic
agents is warranted.
Ciprofloxacin is a broad-spectrum antibiotic of the
fluoroquinolone class that targets bacterial DNA gyrase, with
satisfactory tissue distribution (1). Previous in vivo studies in
animal models of fibrosis have suggested an antifibrotic role for
ciprofloxacin. Thus, ciprofloxacin treatment significantly
decreased hepatic fibrogenesis in bile duct ligated and carbon
tetrachloride/ethanol cirrhotic rats (2,3).
Furthermore, topical ciprofloxacin increased the incidence of
corneal perforations, significantly delaying corneal wound healing
(4,5) and in a separate study prolonged
tympanic membrane perforation healing (6). Increased matrix metalloproteinase
(MMP) synthesis in response to ciprofloxacin treatment has been
reported in several cell types, including tenocytes (7–10),
chondrocytes (11), corneal
epithelial cells and corneal stromal keratocytes (4).
A recent double blind randomized clinical trial
compared changes in skin fibrosis in placebo and
ciprofloxacin-treated scleroderma patients. Using the modified
Rodnan skin score (MRSS), investigators demonstrated that after six
months of treatment there was a significant decrease in MRSS in
patients treated with ciprofloxacin when compared with the
placebo-treated group (58 vs. 18%). Importantly, no significant
side effects were reported, suggesting that long-term use of this
drug may be safe in SSc patients (12). While this report suggests that
ciprofloxacin has antifibrotic effects on SSc skin, the mechanism
of action in dermal fibroblasts is completely unknown.
Friend leukemia integration factor 1 (Fli1) is a
member of the Ets family of transcription factors that is
preferentially expressed in hematopoietic cell lineages (13). Although expressed at low levels in
dermal fibroblasts, Fli1 plays a pivotal role in the regulation of
ECM genes, including type I collagen (14–16) and the profibrotic matrix protein
connective tissue growth factor (CCN2) (17). Fli1 is a potent inhibitor of
collagen gene expression in dermal fibroblasts and the
downregulation of Fli1 protein in dermal fibroblasts from the
affected skin of SSc patients correlates with elevated collagen
deposition, thus suggesting a role of Fli-1 in SSc fibrosis
(15).
The present study was undertaken to examine the
effects of ciprofloxacin on cultured human dermal fibroblasts from
SSc patients and the normal controls. We demonstrate that
ciprofloxacin reduces the expression of the fibrotic markers
collagen type I, CCN2 and cartilage oligomeric matrix protein
(COMP) and that it upregulates matrix metalloproteinase 1 (MMP1)
gene expression via an Erk1/2 dependent mechanism. Our study also
provides evidence that SSc fibroblasts are more sensitive to the
antifibrotic effects of ciprofloxacin, presumably via DNA
methyltransferase 1 (Dnmt1)-induced derepression of Fli1.
Additionally, we demonstrate that ciprofloxacin has potent
antifibrotic effects on lung fibroblasts isolated from SSc patients
with interstitial lung disease (ILD).
Materials and methods
Reagents
The following antibodies were used: monoclonal
β-actin (Sigma-Aldrich, St. Louis, MO, USA), anti-CTGF, anti-lamin
A/C (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA), goat
anti-type-1 collagen (Southern Biotech, Birmingham, AL, USA),
anti-phospho-ERK1/2 (T202/Y204), anti-ERK1/2, anti-SMAD2/3,
anti-phospho-SMAD2/3 (S465/467), anti-SMAD1/5/8,
anti-phospho-SMAD1/5 (S463/465)/SMAD8 (S426/428), monoclonal
anti-Dnmt1 (Cell Signaling Technology, Inc., Beverly, MA, USA) and
monoclonal anti-MMP1 (Millipore, Billerica, MA, USA). Polyclonal
rabbit anti-Fli-1 was purchased from Aviva Systems Biology (Cat #:
ARP38096_T020; San Diego, CA, USA). Ciprofloxacin was purchased
from LKT Laboratories (St. Paul, MN, USA). Dulbecco’s modified
Eagle’s medium (DMEM) and 100X antibiotic-antimycotic solution
(penicillin streptomycin and amphotericin B) were obtained from
Gibco-BRL (Grand Island, NY, USA). Fetal bovine serum (FBS) was
purchased from HyClone (Logan, UT, USA). The ERK1/2 inhibitor UO126
was purchased from Cell Signaling Technology, Inc. Enhanced
chemiluminescence reagent and bovine serum albumin (BSA) protein
assay reagent were obtained from Pierce Biotechnology, Inc.
(Rockford, IL, USA). Tri reagent was purchased from the Molecular
Research Center (Cincinnati, OH, USA). Primers were purchased from
Operon (Huntsville, AL, USA).
Cell culture
Human dermal fibroblast cultures were established
from biopsy specimens obtained from the dorsal forearms of SSc
patients with diffuse cutaneous disease and from age-, race- and
gender-matched healthy donors, upon informed consent and in
compliance with the Institutional Review Board. Dermal fibroblasts
were cultured from the biopsy specimens as previously described
(24). Normal and SSc skin
fibroblasts were cultured in DMEM supplemented with 10% FBS and 1%
antibiotic-antimycotic solution. Primary lung fibroblast cell
cultures were developed using the explant method immediately after
open lung biopsy, upon informed consent and in compliance with the
Institutional Review Board. The lung specimens were sectioned in
small fragments and transferred to a large 25-cm2 flask
(Costar, USA) containing DMEM supplemented with 20% FBS, 1%
L-glutamine and 1% penicillin + streptomycin. The specimens were
incubated at 37°C in a 5% CO2 atmosphere until cells
achieved confluence. After cell detachment with 0.2% trypsin in
phosphate-buffered saline (PBS), the cells were transferred to a
75-cm2 flask and grown in DMEM with 20% FBS.
Fibroblasts were grown to confluence, changed to
serum-free media and treated with indicated doses of ciprofloxacin
or 0.1 M HCI for 24, 48 or 96 h before mRNA or protein was
collected. For the inhibition of ERK1/2, UO126 was applied (1
μM) in the serum-free media 1 h prior to the beginning of
ciprofloxacin treatment and the cells were harvested after 48
h.
Western blot analysis
Cells were collected and washed with PBS. Cell
pellets were suspended in lysis buffer containing 20 mM Tris-HCl,
pH 7.5, 15 mM NaCl, 1 mM EDTA, 1 mM EGTA, 1% Triton X-100, 2.5 mM
sodium pyrophosphate and 1 mM glycerophosphate with freshly added
phosphatase inhibitors (5 mM sodium fluoride and 1 mM
Na3VO4) and a protease inhibitor mixture
(Sigma-Aldrich). Protein concentration was quantified using the BCA
Protein assay kit (Thermo Scientific, Rockford, IL, USA). Equal
amounts of total protein for each sample were separated via
SDS-PAGE and transferred to nitrocellulose membranes (Bio-Rad,
Hercules, CA, USA). Membranes were blocked in 2% milk in TBST for 1
h and incubated with the primary Ab overnight at 4°C. After TBST
washes, membranes were probed with HRP-conjugated secondary Ab
against the appropriate species for 1 h at room temperature.
Protein was visualized using an ECL reagent (Amersham Biosciences,
Piscataway, NJ, USA) and quantified using Image J densitometry
software. For nuclear extraction, the NE-PER nuclear and
cytoplasmic extraction kit was used according to the manufacturer’s
instructions. For the loading controls, blots were stripped with
Restore Western Blot Stripping buffer (all were from Thermo
Scientific) and reprobed with antibodies to β-actin or lamin A/C,
for whole cell or nuclear lysates, respectively.
Real-time PCR
Total RNA was isolated from dermal fibroblasts using
Tri reagent according to the manufacturer’s instructions. RNA (1
μg) was reverse transcribed in a 20-μl reaction using
random primers and Transcriptor First Strand Synthesis kit (Roche
Applied Science, Indianapolis, IN, USA). cDNA was diluted 10-fold
and quantitative (q)PCR was performed using IQ SYBR Green mix on an
iCycler PCR machine (all were from Bio-Rad) using 1 μl of
cDNA in triplicate, with β-actin as the internal control. The
primers used were as follows: β2-microglobulin, forward (GCC GTG
TGA ACC ATG TGA CTT T) and reverse (CCA AAT GCG GCA TCT TCA AA);
MMP1, forward (TCT GGG GTG TGG TGT CTA) and reverse (GCC TCC CAT
CAT TCT CAG GTT); COL1A1, forward (CCA GAA GAA CTG GTA CAT CAG CA)
and reverse (CGC CAT ACT CGA ACT GGG AAT); COL1A2, forward (GAT GTT
GAA CTT GTT GCT GAG G) and reverse (TCT TTC CCC ATT CAT TTG TCT T);
Fli-1, forward (GGA TGG CAA GGA ACT GTG TAA) and reverse (GGT TGT
ATA GGC CAG CAG); COMP, forward (GCA CCG ACG TCA ACG AGT) and
reverse (TGG TGT TGA TAC AGC GGA CT); CCN2, forward (TTG CGA AGC
TGA CCT GGA AGA GAA) and reverse (AGC TCG GTA TGT CTT CAT GCT
GGT).
Results
Increased sensitivity of SSc fibroblasts
to the antifibrotic effects of ciprofloxacin
In order to compare the effects of ciprofloxacin on
SSc and normal dermal fibroblasts, quiescent SSc and control cells
were treated with increasing concentrations of ciprofloxacin, and
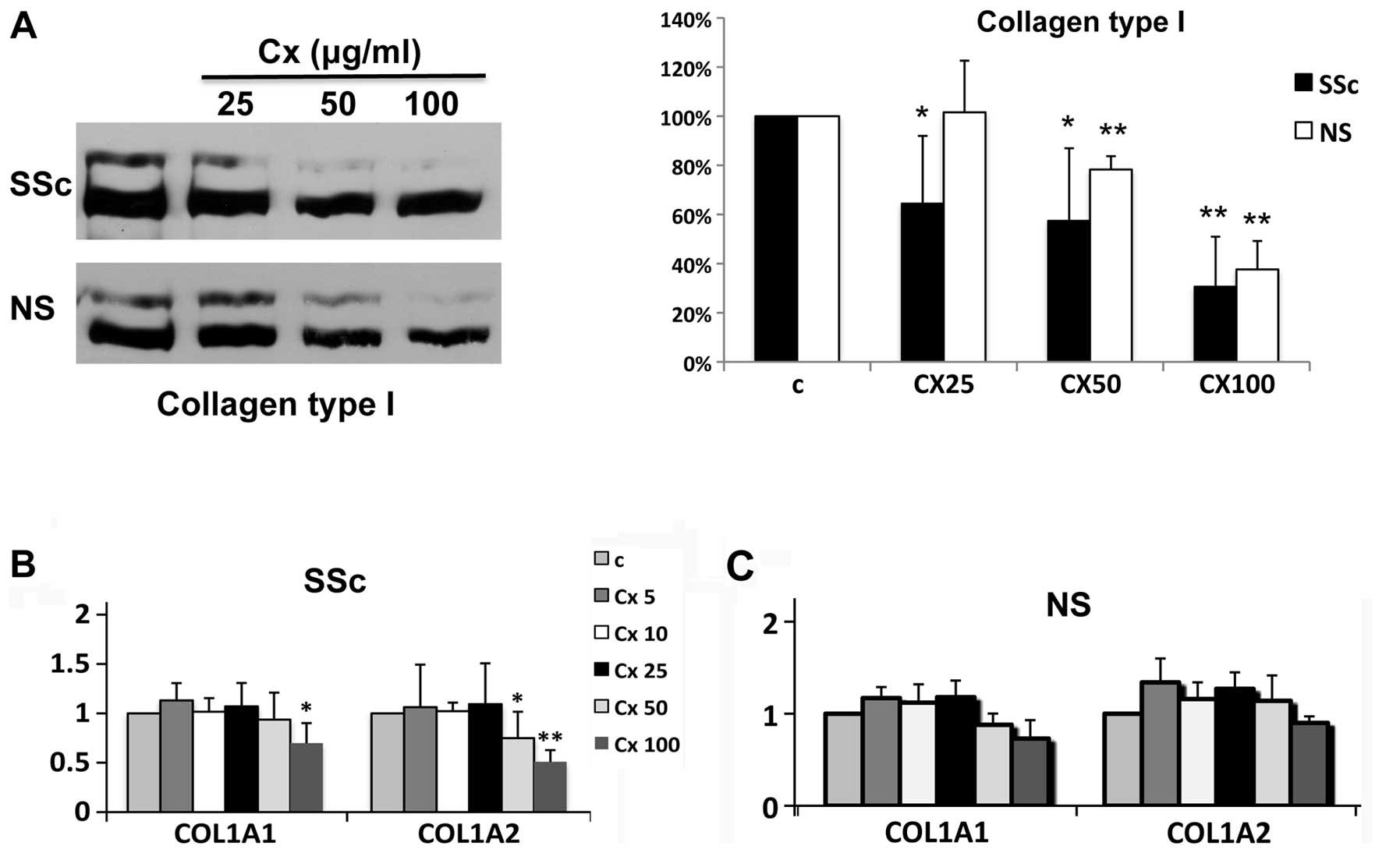
collagen protein levels were analyzed in the culture supernatants.
Ciprofloxacin more potently decreased collagen type I secretion in
SSc cells compared to normal fibroblasts (Fig. 1A). Thus, there was a statistically
significant decrease in secreted collagen starting with 25
μg/ml of ciprofloxacin (∼40%, right panel), while the same
dose had no effect on collagen type I production in normal control
fibroblasts. However, at the highest dose tested (100
μg/ml), ciprofloxacin potently downregulated collagen type I
protein (>60%) in both normal and SSc dermal fibroblasts.
Ciprofloxacin also inhibited mRNA expression of COL1A1 and COL1A2,
displaying a similar dose response (Fig. 1B and C). Comparable to the results
at the protein levels, there was a statistically significant
decrease in both collagen type I gene expression in SSc cells, with
COL1A2 being more responsive to ciprofloxacin treatment (Fig. 1B). Ciprofloxacin only modestly
inhibited COL1A1 and COL1A2 mRNA expression by normal dermal
fibroblasts, not reaching statistical significance (Fig. 1C). These data suggest that SSc
dermal fibroblasts are more sensitive to the antifibrotic effects
of ciprofloxacin compared to the healthy dermal fibroblasts.
Ciprof loxacin downregulates CCN2 and
COMP and increases MMP1 in human dermal fibroblasts
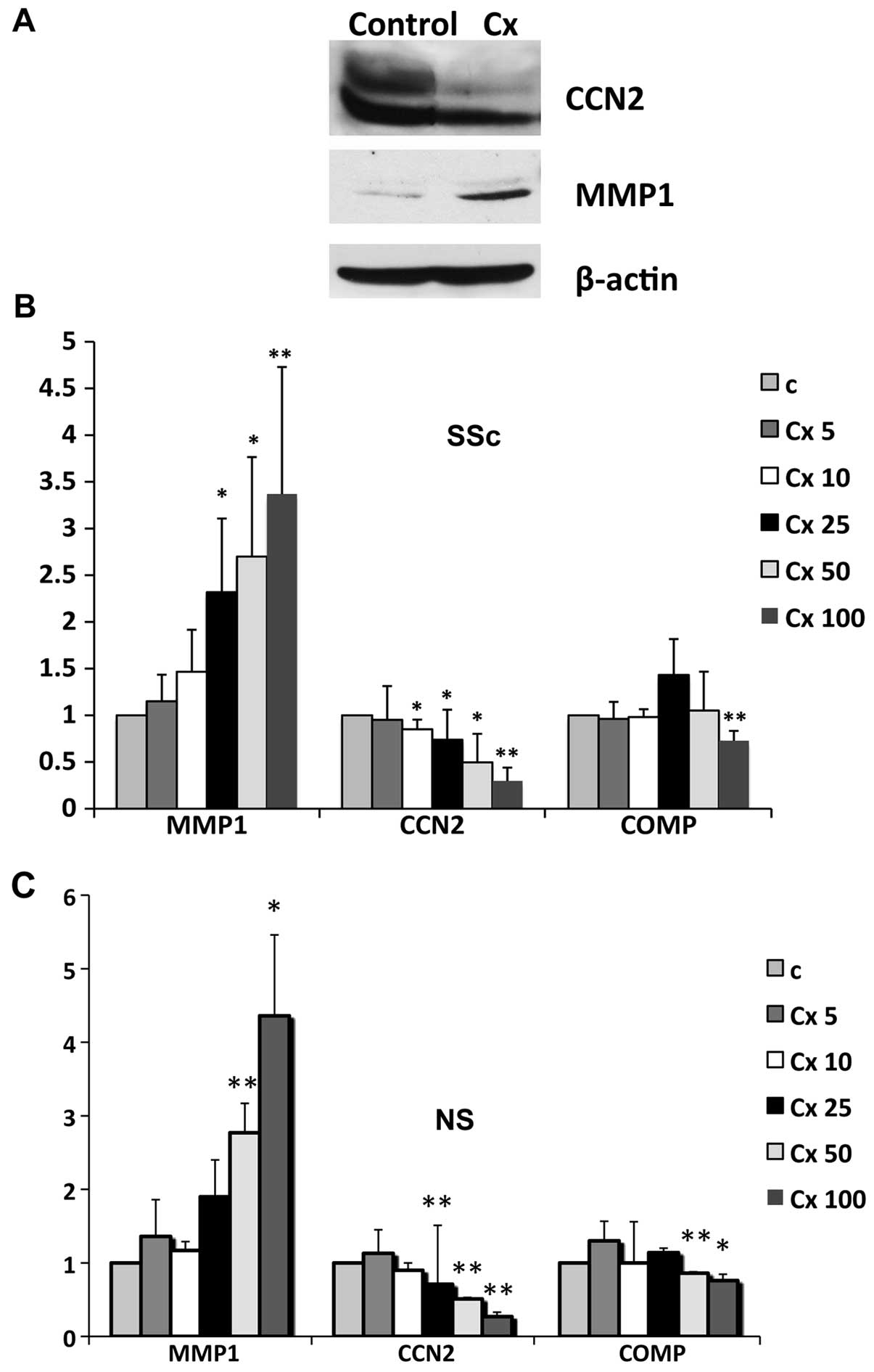
We next examined the effects of ciprofloxacin
treatment on other fibrotic markers that have been implicated in
SSc pathogenesis. Protein levels of the profibrotic gene CCN2 and
of matrix degrading metalloproteinase MMP1 were analyzed in cell
lysates after antibiotic treatment. As previously reported in other
cell types, ciprofloxacin increased MMP1 production in human dermal
fibroblasts. Additionally, there was a significant decrease in CCN2
protein levels after antibiotic treatment, an effect that has not
been previously described in any other cell types (Fig. 2A). To further investigate
potential differences in response to ciprofloxacin treatment
between SSc and healthy cells, we analyzed mRNA levels of MMP1,
CCN2 and the cartilage oligomeric matrix protein (COMP). MMP1 gene
expression was induced in a dose-dependent manner in both SSc and
normal cells, starting with the smallest dose tested and to a
similar extent in both cell types. Opposite effects were observed
for CCN2 and COMP, which were decreased at the mRNA levels in both
SSc and normal cells (Fig. 2B and
C).
Ciprofloxacin induces MMP1 gene
expression via an Erk1/2-dependent mechanism
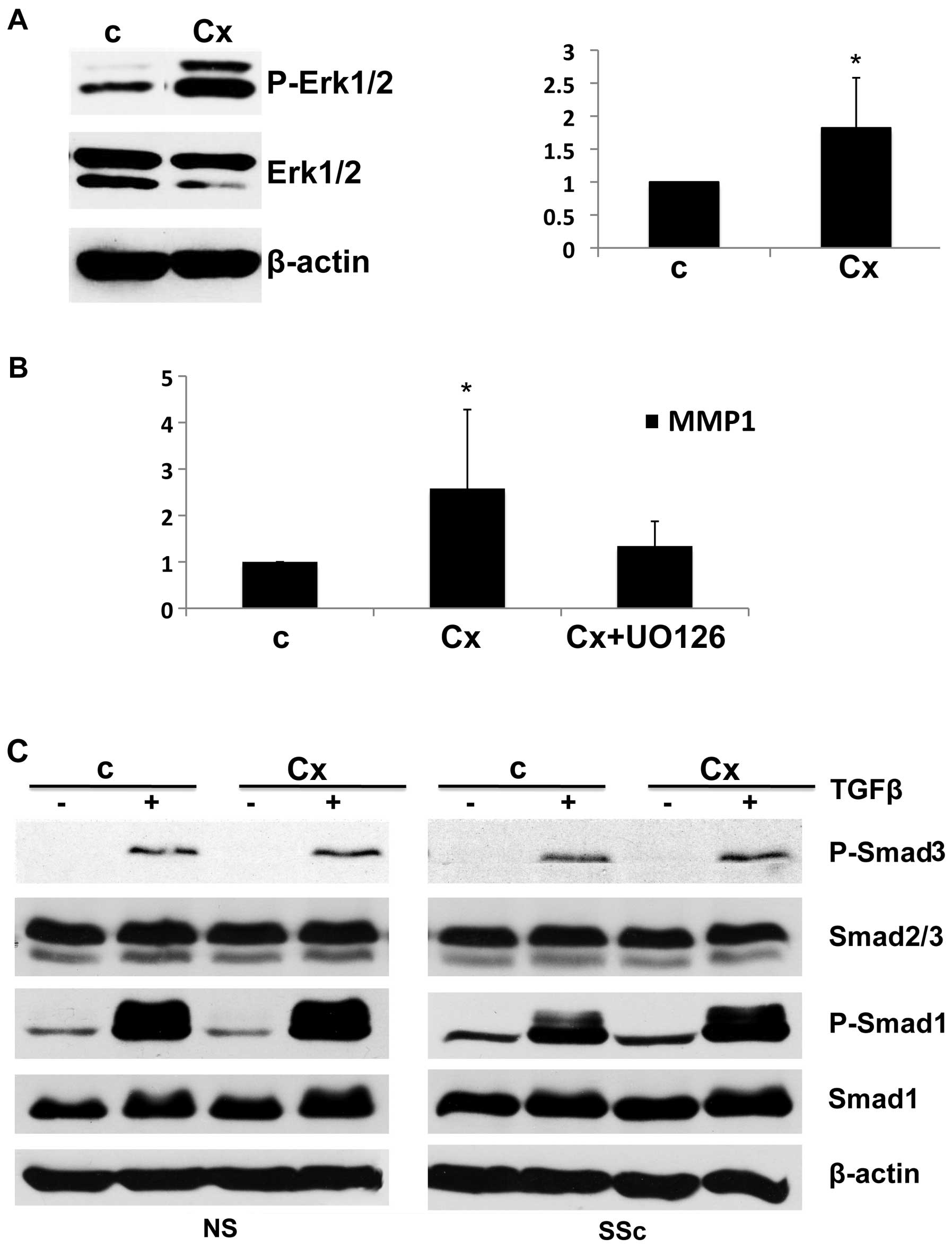
Previous studies have demonstrated that in human
dermal fibroblasts MMP1 gene expression is mainly controlled via an
Erk1/2-dependent mechanism (18–23). To investigate the mechanism of
ciprofloxacin-induced MMP1 gene upregulation we examined the effect
of ciprofloxacin on phosphorylated Erk1/2 (P-Erk1/2). Ciprofloxacin
significantly induced Erk1/2 phosphorylation in human dermal
fibroblasts, with the levels of P-Erk1/2 almost doubling (Fig. 3A, right panel), suggesting that
Erk1/2 may be involved in ciprofloxacin-induced MMP1 gene
expression. To further confirm this we pretreated cells with a
pharmacologic inhibitor of Erk1/2 (UO126) before ciprofloxacin
treatment. Pretreatment with Erk1/2 inhibitor completely abolished
ciprofloxacin-induced MMP1 upregulation (Fig. 3B). Together, these findings
strongly support the notion that the activation of the Erk1/2
pathway contributes to ciprofloxacin-induced MMP1 upregulation in
human dermal fibroblasts.
Effects of ciprofloxacin on signaling
pathways deregulated in SSc
Aberrant activation of several signaling pathways
implicated in the pathogenesis of SSc may serve as a target for
ciprofloxacin treatment. These include the upregulation of the
major profibrotic TGFβ pathway, as well as the PI3K/Akt and
PKCδ/c-abl/Fli1 pathways (24,25). TGFβ signaling plays a central role
in SSc pathogenesis and is evidenced by an increased expression of
pSmad1 in SSc skin and cultured fibroblasts (26), elevated phosphorylated Smad2/3
levels and increased nuclear localization of phosphorylated Smad2/3
in these cells (27).
To investigate the mechanism behind the antifibrotic
actions of ciprofloxacin we evaluated its effects on TGFβ
signaling. Pretreatment with ciprofloxacin had no effect on
TGFβ-induced phosphorylation of Smad3 or Smad1 in both SSc and
healthy cells, suggesting that the antifibrotic effects of
ciprofloxacin are not mediated through the TGFβ/Smad pathway
(Fig. 3C).
Akt activation has been previously linked to the
regulation of collagen, MMP1 and CCN2 gene expression (19,28) and constitutive Akt activation has
been reported in SSc fibroblasts (24). Similar to the results in TGFβ/Smad
activation, ciprofloxacin treatment had no effects on Akt
phosphorylation, suggesting that this pathway is not involved (data
not shown).
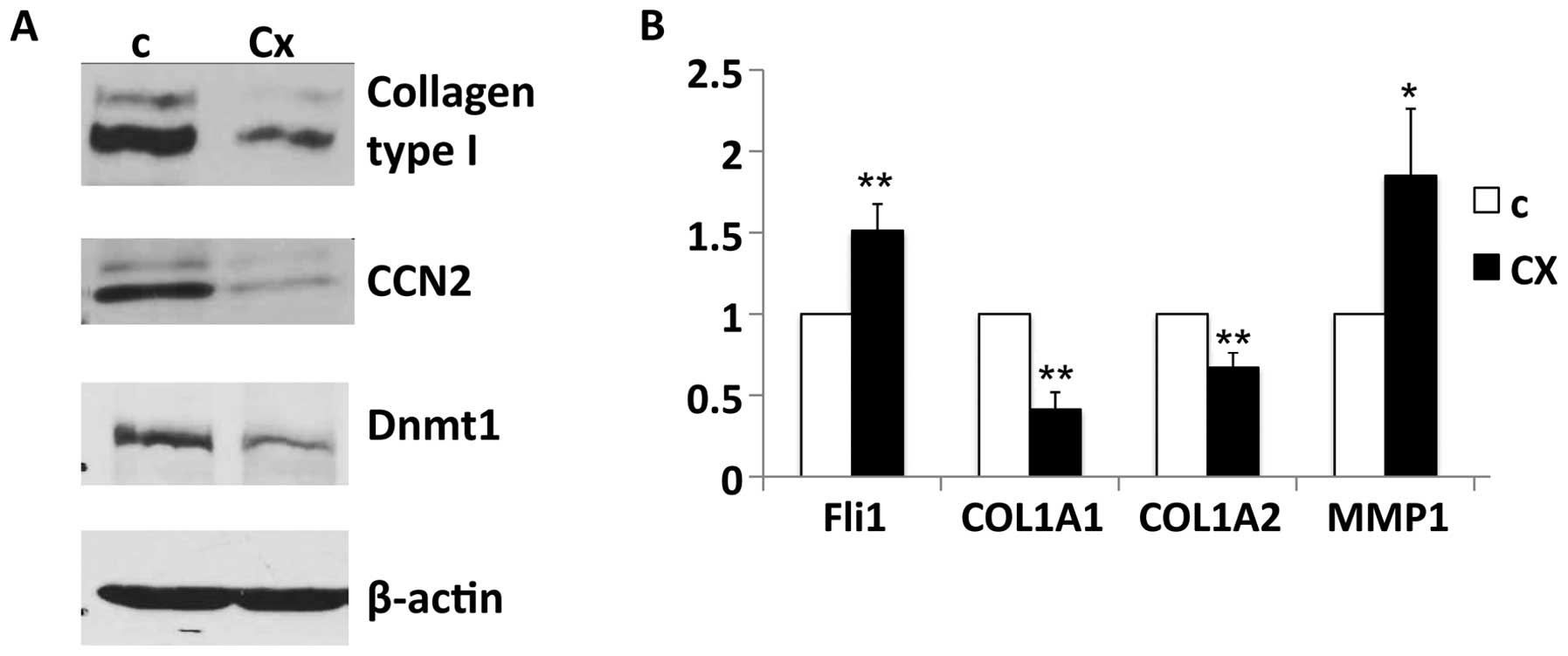
Ciprofloxacin increases Fli1 levels in
SSc fibroblasts
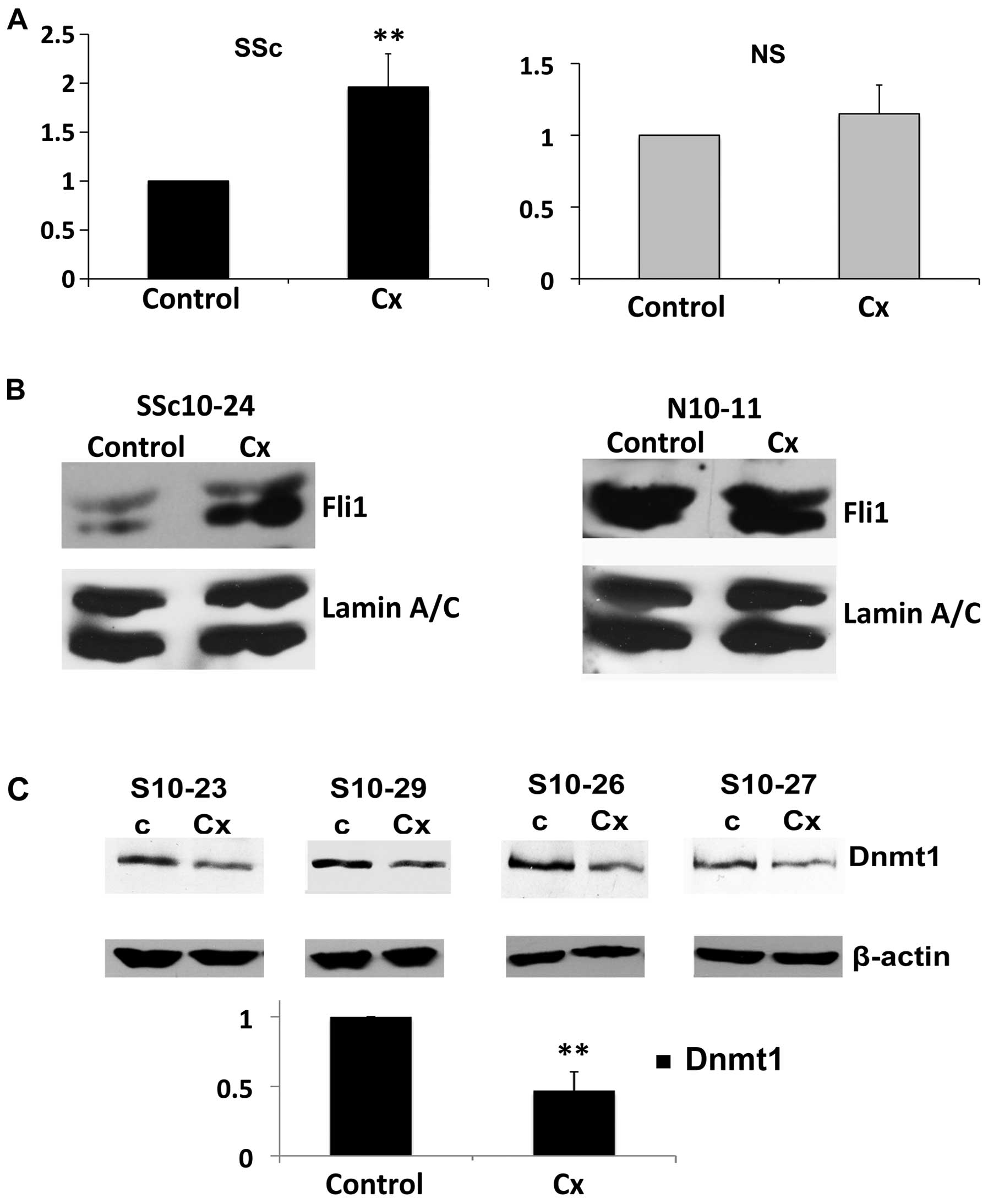
We evaluated the effects of ciprofloxacin on Fli1
levels in normal and SSc dermal fibroblasts. Treatment of SSc
fibroblasts with ciprofloxacin resulted in a statistically
significant increase in Fli1 mRNA, leading to a 2-fold increase
compared to untreated cells. However, when ciprofloxacin was added
to the normal healthy fibroblasts, there was no effect on the mRNA
levels of Fli1 (Fig. 4A).
Essentially the same results were obtained at the protein levels,
with a significant increase in Fli1 expression in SSc fibroblasts
but with no change in the healthy cells after ciprofloxacin
treatment (Fig. 4B).
Epigenetic repression of the Fli1 gene via
methylation of cytosine nucleotides in the non-coding regions has
been previously demonstrated to contribute to SSc fibrosis. Thus, a
recent report demonstrated that the levels of epigenetic mediators,
including the expression of methyltransferase Dnmt1, were altered
in SSc fibroblasts (29). To
examine whether epigenetic changes may be implicated in the
ciprofloxacin-induced Fli1 upregulation in SSc dermal fibroblasts,
four different SSc cell lines were treated with ciprofloxacin and
the protein levels of Dnmt1 were then analyzed in cell lysates. In
all cell lines there was a significant decrease in protein levels
of Dnmt1 after ciprofloxacin treatment (>50%, P<0.0001;
Fig. 4C).
Ciprofloxacin has antifibrotic effects on
lung fibroblasts isolated from SSc patients with ILD
Since one of the major complications in SSc is
pulmonary fibrosis, we next evaluated whether ciprofloxacin also
reduces fibrotic markers in SSc lung fibroblasts isolated from
patients with ILD. Ciprofloxacin potently downregulated collagen
type I protein levels and decreased mRNA expression of both COL1A1
and COL1A2 chains (Fig. 5A and
B). Similar to the results obtained in dermal fibroblasts, the
levels of the profibrotic marker CCN2 were also downregulated after
ciprofloxacin treatment, while MMP1 gene expression was enhanced.
Furthermore, ciprofloxacin inhibited the levels of Dnmt1, while it
upregulated Fli1 gene expression, thus suggesting that a similar
mechanism accounts for the antifibrotic effects of ciprofloxacin in
both dermal and lung fibroblasts isolated from SSc patients
(Fig. 5). Based on these results,
we concluded that ciprofloxacin has antifibrotic effects on human
lung fibroblasts from SSc patients with ILD.
Discussion
Skin and lung fibrosis in SSc are serious
complications of the disease, which lack effective treatment.
Intensive efforts have been made to discover new therapies that may
ameliorate fibrosis in this disease. However, drugs that potently
reduce fibrosis in vitro and in animal models have failed to
provide reproducible antifibrotic effects in clinical trials for
SSc patients. Intriguing preliminary data from a small clinical
trial considering the effects of ciprofloxacin versus a placebo on
skin fibrosis in SSc suggest that this antibiotic may be an
effective treatment for skin fibrosis (12). In this study, we demonstrated that
ciprofloxacin has dual antifibrotic effects on SSc dermal and lung
fibroblasts by upregulating MMP1 and downregulating CCN2 and
collagen type I levels. Furthermore, we provide evidence that Fli1
is upregulated after ciprofloxacin treatment only in SSc
fibroblasts, but not in healthy cells, which were less responsive
to the antifibrotic effects of antibiotic treatment. Additionally
our study reveals that ciprofloxacin-induced MMP1 upregulation is
mediated via the activation of the Erk1/2 pathway.
Tendinopathies, including tendon ruptures, have been
described as rare but severe complications of ciprofloxacin
treatment, with the main risk factors being old age, systemic
steroid therapy, dialysis and strenuous physical activity. Although
the exact pathologic mechanisms underlying this severe complication
are poorly understood, several mechanisms have been proposed,
including the upregulation of matrix metalloproteinases (MMPs)
followed by type I collagen degradation, inhibition of cell
proliferation by the downregulation of cyclin B and
cyclin-dependent kinase 1 (Cdk1) and the inhibition of tenocyte
migration by the downregulation of focal adhesion kinase
phosphorylation (7,8,30–32). Thus, ciprofloxacin-induced
collagen downregulation in tendon cells is the result of increased
ECM degradation due to the upregulation of various MMPs. Fibrosis
in SSc is the result of the uncontrolled deposition of ECM that is
presumably due to increased synthesis and to decreased degradation
of matricellular components. Ciprofloxacin may affect SSc fibrosis
by regulating the aberrant expression of MMPs, leading to increased
collagen turnover. MMP1 is the only enzyme capable of initiating
the breakdown of interstitial collagens, including collagen type I,
and published data indicates that this enzyme is downregulated in
SSc cells (33). Our study
demonstrates that, in addition to the effects on MMP1,
ciprofloxacin treatment may directly block collagen synthesis in
SSc fibroblasts, but not in healthy controls, by upregulating Fli1
levels.
The underexpression of Fli1 has been previously
reported in fibrotic conditions such as SSc (15), cardiac fibrosis (34) and wound healing (35). Fli1 expression is markedly
downregulated in lesional fibroblasts of patients with scleroderma,
and a recent study has linked epigenetic regulation to the
repression of the Fli1 gene in scleroderma skin in vivo.
Thus, increased methylation of the Fli1 promoter was observed in
SSc skin, while the authors did not discover any detectable
methylation in the healthy control skin, suggesting Fli1
hypermethylation is specific for SSc fibroblasts (29). DNA methylation is an epigenetic
regulation mechanism, resulting in transcriptional silencing of
genes. This process is important for normal development but errors
leading to promoter hypermethylation may play a major role in human
disease. DNA methylation occurs by the addition of methyl groups to
cytosine residues in DNA, catalyzed by a family of enzymes called
DNA methyltransferases. Of the three types of Dnmt observed in
mammalian cells, Dnmt1 was upregulated up to 3-fold in SSc
fibroblasts, while Dnmt3a and Dnmt3b levels remained unchanged
(29). In our study ciprofloxacin
treatment potently decrease Dnmt1 levels in SSc dermal and lung
fibroblasts, while upregulating Fli1 gene expression and
downregulating COL1A1 and COL1A2 mRNA. Of note, ciprofloxacin had
no effects on Fli1 levels in normal dermal fibroblasts, which also
showed no reduction in collagen mRNA levels, suggesting that
ciprofloxacin regulates Fli1 levels in SSc fibroblasts via an
epigenetic mechanism that involves Dnmt1 downregulation.
Recent studies (19,25) from our laboratory demonstrated
that the reduction of Fli1 levels in dermal fibroblasts results in
an increased synthesis of type I collagen and CCN2 and a reduction
in MMP1 production. In this study we revealed that ciprofloxacin
treatment has opposite effects on collagen, CCN2 and MMP1. While
transcriptional regulation of collagen type I genes in response to
ciprofloxacin treatment is mainly controlled via Fli1, other
factors may contribute to ciprofloxacin-induced MMP1 upregulation
and CCN2 repression, since the mRNA levels of these genes are
altered in both SSc and normal fibroblasts.
Erk1/2 activates the Ets1 transcription factor that
in turn cooperates with the AP-1 proteins c-Fos and c-Jun in
inducing the MMP1 promoter (19).
Our present study demonstrates that ciprofloxacin induces Erk1/2
phosphorylation, and that the specific Erk1/2 inhibitor UO126
completely prevents ciprofloxacin-induced MMP1 upregulation, thus
revealing that Erk1/2 is required for this process.
CCN2 and COMP are matricellular proteins that are
over-expressed in SSc and are potently induced by TGFβ (36–38). Our previously published study
showed that SSc fibroblasts have constitutive activation of Smad1
pathway, and that this pathway is directly involved in the
upregulation of CCN2 gene expression (25). Additionally, Smad3 is required for
the TGFβ induction of CCN2 (39).
In our study, ciprofloxacin had no effects on TGFβ-induced
phosphorylation of Smad1 or Smad3, thus suggesting that the
downregulation of CCN2 and COMP in response to antibiotic treatment
is independent of the TGFβ/Smad pathway. To the best of our
knowledge, this is the first report to demonstrate that
ciprofloxacin downregulates CCN2 and COMP in fibroblasts; however,
further studies are required to elucidate the exact mechanism for
these effects.
ILD in SSc is a potentially lethal complication with
modest therapeutic options. Our study demonstrated that, similar to
dermal fibroblasts, ciprofloxacin has antifibrotic effects on
cultured SSc ILD lung fibroblasts, by upregulating Fli1 and MMP1
levels and downregulating collagen type I and CCN2 gene expression.
The present data suggest that ciprofloxacin may also be an
attractive antifibrotic therapy for SSc ILD.
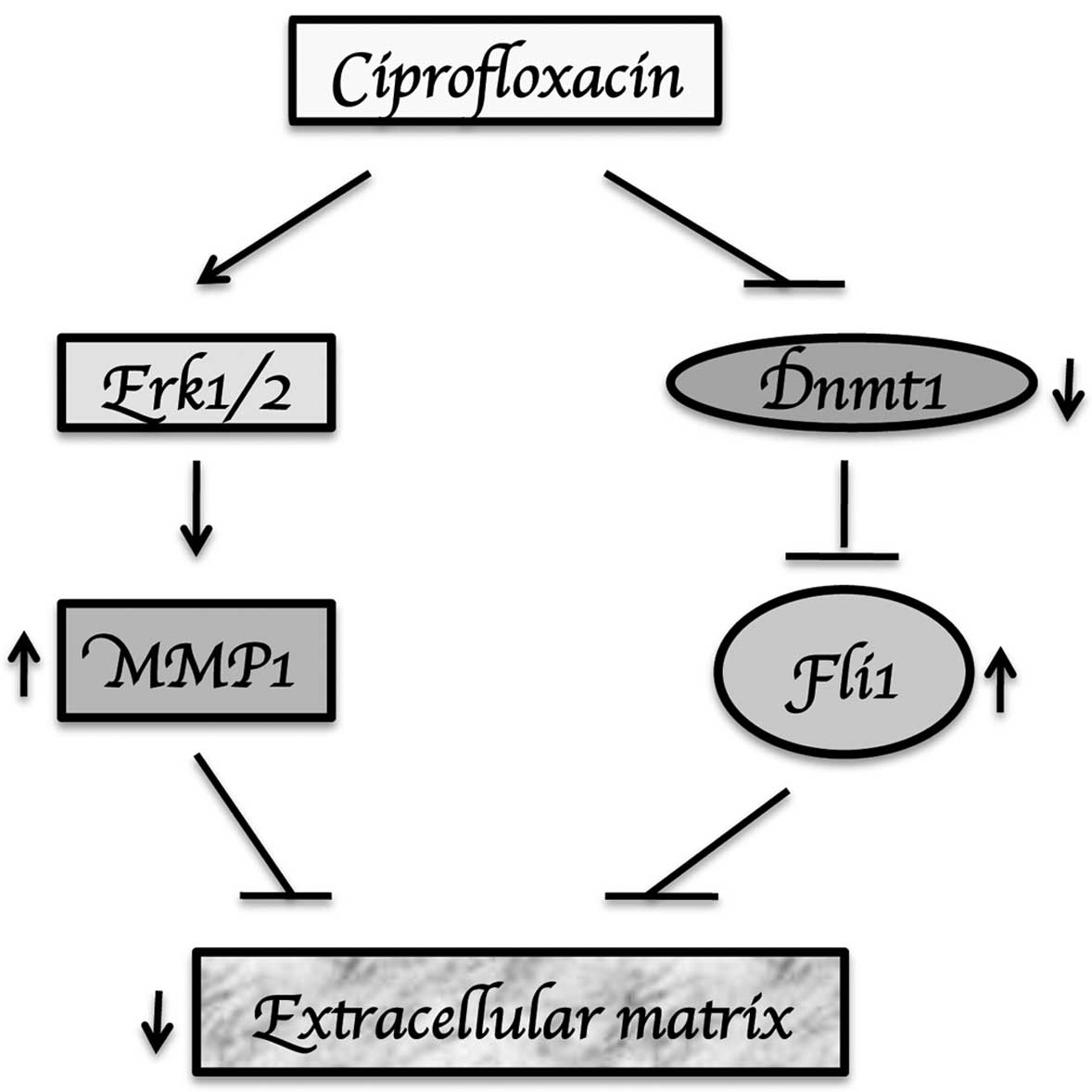
In summary, our study showed that ciprofloxacin has
antifibrotic actions in SSc dermal and lung fibroblasts via
downregulation of Dnmt1, upregulation of Fli1 and induction of MMP1
gene expression via an Erk1/2-dependent mechanism (Fig. 6). While these results provide
evidence to support the use of ciprofloxacin in SSc, larger
randomized clinical trials are warranted to confirm whether this
may be a new treatment modality for SSc skin and lung fibrosis.
References
|
1.
|
A De SarroG De SarroAdverse reactions to
fluoroquinolones. an overview on mechanistic aspectsCurr Med
Chem8371384200111172695
|
|
2.
|
HC LinYY YangTH TsaiThe relationship
between endotoxemia and hepatic endocannabinoids in cirrhotic rats
with portal hypertensionJ
Hepatol5411451153201110.1016/j.jhep.2010.09.02621145843
|
|
3.
|
M ZhangG SongGY MinukEffects of hepatic
stimulator substance, herbal medicine, selenium/vitamin E, and
ciprofloxacin on cirrhosis in the
ratGastroenterology11011501155199610.1053/gast.1996.v110.pm86130048613004
|
|
4.
|
VE ReviglioMA HakimJK SongTP O’BrienEffect
of topical fluoroquinolones on the expression of matrix
metalloproteinases in the corneaBMC
Ophthalmol310200310.1186/1471-2415-3-1014529574
|
|
5.
|
C SharmaT VelpandianS Baskar SinghN Ranjan
BiswasR Bihari VajpayeeS GhoseEffect of fluoroquinolones on the
expression of matrix metalloproteinase in debrided cornea of
ratsToxicol Mech
Methods21612201110.3109/15376516.2010.52918321058936
|
|
6.
|
J BuytenG KaufmanM RyanEffects of
ciprofloxacin/dexamethasone and ofloxacin on tympanic membrane
perforation healingOtol
Neurotol28887890200710.1097/MAO.0b013e3180dca1a3
|
|
7.
|
WC TsaiCC HsuCP ChenCiprofloxacin
up-regulates tendon cells to express matrix metalloproteinase-2
with degradation of type I collagenJ Orthop
Res296773201110.1002/jor.2119620602464
|
|
8.
|
AN CorpsRL HarrallVA CurrySA FenwickBL
HazlemanGP RileyCiprofloxacin enhances the stimulation of matrix
metalloproteinase 3 expression by interleukin-1beta in human
tendon-derived cells. A potential mechanism of
fluoroquinolone-induced tendinopathyArthritis
Rheum4630343040200210.1002/art.10617
|
|
9.
|
AN CorpsRL HarrallVA CurryBL HazlemanGP
RileyContrasting effects of fluoroquinolone antibiotics on the
expression of the collagenases, matrix metalloproteinases (MMP)-1
and -13, in human tendon-derived cellsRheumatology
(Oxford)4415141517200510.1093/rheumatology/kei08716148020
|
|
10.
|
J SendzikM ShakibaeiM Schafer-KortingH
LodeR StahlmannSynergistic effects of dexamethasone and quinolones
on human-derived tendon cellsInt J Antimicrob
Agents35366374201010.1016/j.ijantimicag.2009.10.00920034766
|
|
11.
|
K GotoK YabeT SuzukiK TakasunaT JindoS
ManabeGene expression profiles in the articular cartilage of
juvenile rats receiving the quinolone antibacterial agent
ofloxacinToxicology249204213200810.1016/j.tox.2008.05.00518572299
|
|
12.
|
EC RubenVR ManuelOR AgustinM HuertaFM
AntonioDE IvanCiprofloxacin utility as antifibrotic in the skin of
patients with sclerodermaJ
Dermatol37323329201010.1111/j.1346-8138.2010.00826.x20507401
|
|
13.
|
AH TruongY Ben-DavidThe role of Fli-1 in
normal cell function and malignant
transformationOncogene1964826489200010.1038/sj.onc.120404211175364
|
|
14.
|
J Czuwara-LadykowskaF ShirasakiP JackersDK
WatsonM TrojanowskaFli-1 inhibits collagen type I production in
dermal fibroblasts via an Sp1-dependent pathwayJ Biol
Chem2762083920848200110.1074/jbc.M01013320011278621
|
|
15.
|
M KuboJ Czuwara-LadykowskaO
MoussaPersistent down-regulation of Fli1, a suppressor of collagen
transcription, in fibrotic scleroderma skinAm J
Pathol163571581200310.1016/S0002-9440(10)63685-112875977
|
|
16.
|
Y AsanoM MarkiewiczM KuboG SzalaiDK
WatsonM TrojanowskaTranscription factor Fli1 regulates collagen
fibrillogenesis in mouse skinMol Cell
Biol29425434200910.1128/MCB.01278-0819001092
|
|
17.
|
SS NakerakantiB KapanadzeM YamasakiM
MarkiewiczM TrojanowskaFli1 and Ets1 have distinct roles in
connective tissue growth factor/CCN2 gene regulation and induction
of the profibrotic gene programJ Biol
Chem2812525925269200610.1074/jbc.M60046620016829517
|
|
18.
|
P HainesGH SamuelH CohenM TrojanowskaAM
BujorCaveolin-1 is a negative regulator of MMP-1 gene expression in
human dermal fibroblasts via inhibition of Erk1/2/Ets1 signaling
pathwayJ Dermatol
Sci64210216201110.1016/j.jdermsci.2011.08.00521925842
|
|
19.
|
AM BujorS NakerakantiE MorrisFN HantM
TrojanowskaAkt inhibition up-regulates MMP1 through a
CCN2-dependent pathway in human dermal fibroblastsExp
Dermatol19347354201010.1111/j.1600-0625.2010.01065.x20201953
|
|
20.
|
S BuM YamanakaH PeiDihydrosphingosine
1-phosphate stimulates MMP1 gene expression via activation of
ERK1/2-Ets1 pathway in human fibroblastsFASEB
J20184186200616278291
|
|
21.
|
M JinninH IhnY MimuraY AsanoK YamaneK
TamakiMatrix metalloproteinase-1 up-regulation by hepatocyte growth
factor in human dermal fibroblasts via ERK signaling pathway
involves Ets1 and Fli1Nucleic Acids
Res3335403549200510.1093/nar/gki64815972796
|
|
22.
|
Y AsanoH IhnK YamaneM JinninY MimuraK
TamakiIncreased expression of integrin alpha(v)beta3 contributes to
the establishment of autocrine TGF-beta signaling in scleroderma
fibroblastsJ
Immunol17577087718200510.4049/jimmunol.175.11.770816301681
|
|
23.
|
Y MimuraH IhnM JinninY AsanoK YamaneK
TamakiEpidermal growth factor affects the synthesis and degradation
of type I collagen in cultured human dermal fibroblastsMatrix
Biol25202212200610.1016/j.matbio.2005.12.00216413767
|
|
24.
|
JB JunM KuechleJ MinScleroderma
fibroblasts demonstrate enhanced activation of Akt (protein kinase
B) in situJ Invest
Dermatol124298303200510.1111/j.0022-202X.2004.23559.x15675946
|
|
25.
|
AM BujorY AsanoP HainesR LafyatisM
TrojanowskaThe c-Abl tyrosine kinase controls protein kinase
Cdelta-induced Fli-1 phosphorylation in human dermal
fibroblastsArthritis
Rheum6317291737201110.1002/art.3028421321929
|
|
26.
|
J PannuY AsanoS NakerakantiSmad1 pathway
is activated in systemic sclerosis fibroblasts and is targeted by
imatinib mesylateArthritis
Rheum5825282537200810.1002/art.2369818668566
|
|
27.
|
Y MoriSJ ChenJ VargaExpression and
regulation of intracellular SMAD signaling in scleroderma skin
fibroblastsArthritis
Rheum4819641978200310.1002/art.1115712847691
|
|
28.
|
AM BujorJ PannuS BuEA SmithRC
Muise-HelmericksM TrojanowskaAkt blockade downregulates collagen
and upregulates MMP1 in human dermal fibroblastsJ Invest
Dermatol12819061914200810.1038/jid.2008.3918323784
|
|
29.
|
Y WangPS FanB KahalehAssociation between
enhanced type I collagen expression and epigenetic repression of
the FLI1 gene in scleroderma fibroblastsArthritis
Rheum5422712279200610.1002/art.2194816802366
|
|
30.
|
RJ Williams IIIE AttiaTL WickiewiczJA
HannafinThe effect of ciprofloxacin on tendon, paratenon, and
capsular fibroblast metabolismAm J Sports
Med28364369200010843129
|
|
31.
|
WC TsaiCC HsuFT TangAM WongYC ChenJH
PangCiprofloxacin-mediated cell proliferation inhibition and G2/M
cell cycle arrest in rat tendon cellsArthritis
Rheum5816571663200810.1002/art.2351818512786
|
|
32.
|
WC TsaiCC HsuHC ChenCiprofloxacin-mediated
inhibition of tenocyte migration and down-regulation of focal
adhesion kinase phosphorylationEur J
Pharmacol6072326200910.1016/j.ejphar.2009.02.00619232343
|
|
33.
|
K TakedaA HatamochiH UekiM NakataY
OishiDecreased collagenase expression in cultured systemic
sclerosis fibroblastsJ Invest
Dermatol103359363199410.1111/1523-1747.ep123949368077701
|
|
34.
|
J ElkarehSM PeriyasamyA
ShidyakMarinobufagenin induces increases in procollagen expression
in a process involving protein kinase C and Fli-1: implications for
uremic cardiomyopathyAm J Physiol Renal
Physiol296F1219F1226200910.1152/ajprenal.90710.2008
|
|
35.
|
A SakthianandeswarenJM CurtisC ElsoFine
mapping of Leishmania major susceptibility Locus lmr2 and evidence
of a role for Fli1 in disease and wound healingInfect
Immun7827342744201010.1128/IAI.00126-1020368343
|
|
36.
|
D AbrahamConnective tissue growth factor:
growth factor, matricellular organizer, fibrotic biomarker or
molecular target for anti-fibrotic therapy in SSc?Rheumatology
(Oxford)47Suppl 5v8v9200810.1093/rheumatology/ken27818784153
|
|
37.
|
G FarinaR LemaireP PancariJ BayleRL WidomR
LafyatisCartilage oligomeric matrix protein expression in systemic
sclerosis reveals heterogeneity of dermal fibroblast responses to
transforming growth factor betaAnn Rheum
Dis68435441200910.1136/ard.2007.086850
|
|
38.
|
G FarinaD LafyatisR LemaireR LafyatisA
four-gene biomarker predicts skin disease in patients with diffuse
cutaneous systemic sclerosisArthritis
Rheum62580588201010.1002/art.2722020112379
|
|
39.
|
A HolmesDJ AbrahamS SaX ShiwenCM BlackA
LeaskCTGF and SMADs, maintenance of scleroderma phenotype is
independent of SMAD signalingJ Biol
Chem2761059410601200110.1074/jbc.M01014920011152469
|