Long-term epidemiological studies have found that
the incidence of LC is increasing (14,16); this could be a real increase or
the result of an increased awareness of the necessity of examining
colonic biopsy samples in patients with diarrhea without obvious
etiology (14). In Örebro county
(Sweden) and Olmsted county (MN, USA) where these studies were
conducted, the awareness of LC has been high and constant during
all of the investigation period, making it unlikely that this
factor played a role in the observed increase in LC incidence.
However, change in the age distribution during the long observation
periods (>15 years) may have an effect on this increase, since
official Swedish statistics indicate a considerable increase in the
proportion of individuals >60 years of age in Örebro county.
The reported rate of spontaneous symptomatic
remission in LC in follow-up studies has varied between 59 and 93%
(23,27). Histopathological normalization
reportedly occurs in 82% of patients with LC after approximately 38
months of follow-up (23). A
follow-up study over a 6.4-year period also found a high rate of
symptomatic remission in patients with LC (28). Moreover, the risk of colon cancer
or inflammatory bowel disease is not increased in patients with LC
(29).
The results of a physical examination, laboratory
tests, small and large bowel radiology and colonoscopy are normal
in LC, with the diagnosis instead being based on histological
changes in colonic biopsy samples. The typical histological changes
include an increase in intraepithelial lymphocytes (≥20/100
enterocytes) and infiltration of inflammatory cells (mainly
lymphocytes and plasma cells) in the lamina propria (21,30). Although surface epithelial damage
can be observed, the architecture of the mucosa is normal (Fig. 1) (21,30). In clinical practice, the increase
in intraepithelial lymphocytes is usually clearly evident with
manual counting only required in cases with a borderline number of
lymphocytes (21). The surface
mucosa overlying lymphoid aggregates always contains higher numbers
of intraepithelial lymphocytes; thus, these areas should be avoided
when an LC diagnosis is being made (21).
While the histopathological diagnosis of LC is
straightforward, it is unclear whether the histopathological
changes observed in LC are diffusely distributed throughout the
colon or limited to a certain segment(s). Certain studies have
found the histopathological changes to be more prominent and easier
to detect in the right colon (cecum and the ascending and right
transverse colon) (11,31,32), while other authors have maintained
that biopsies obtained from the left colon (sigmoid and descending
colon) are reliable for the diagnosis of LC (10,33,34). These controversial results
question whether colonoscopy with a segmental biopsy is required
for the diagnosis of LC, or whether sigmoidoscopy is sufficient.
Solving this issue is important for clinical practice, since the
risk of complications, the clinicians labor and the economic burden
are higher for colonoscopy than for sigmoidoscopy. Since this
matter has not yet been resolved, several gastroenterologists
perform colonoscopy with a segmental colonic biopsy to ensure an
accurate diagnosis.
Chromogranin A is a 68-kDa protein comprising 439
amino acid residues, which was isolated from secretory granules of
the adrenal medulla (35).
Chromogranin A is co-stored and co-released with monoamines and
peptide hormones present in the adrenal medulla, pituitary gland,
parathyroid, thyroid C-cells, pancreatic islets, endocrine cells of
the gastrointestinal tract and sympathetic nerves (35–37). Chromogranin A is therefore
considered to be a general marker for all endocrine cells. Some of
the patients in a cohort of irritable bowel syndrome (IBS), showed
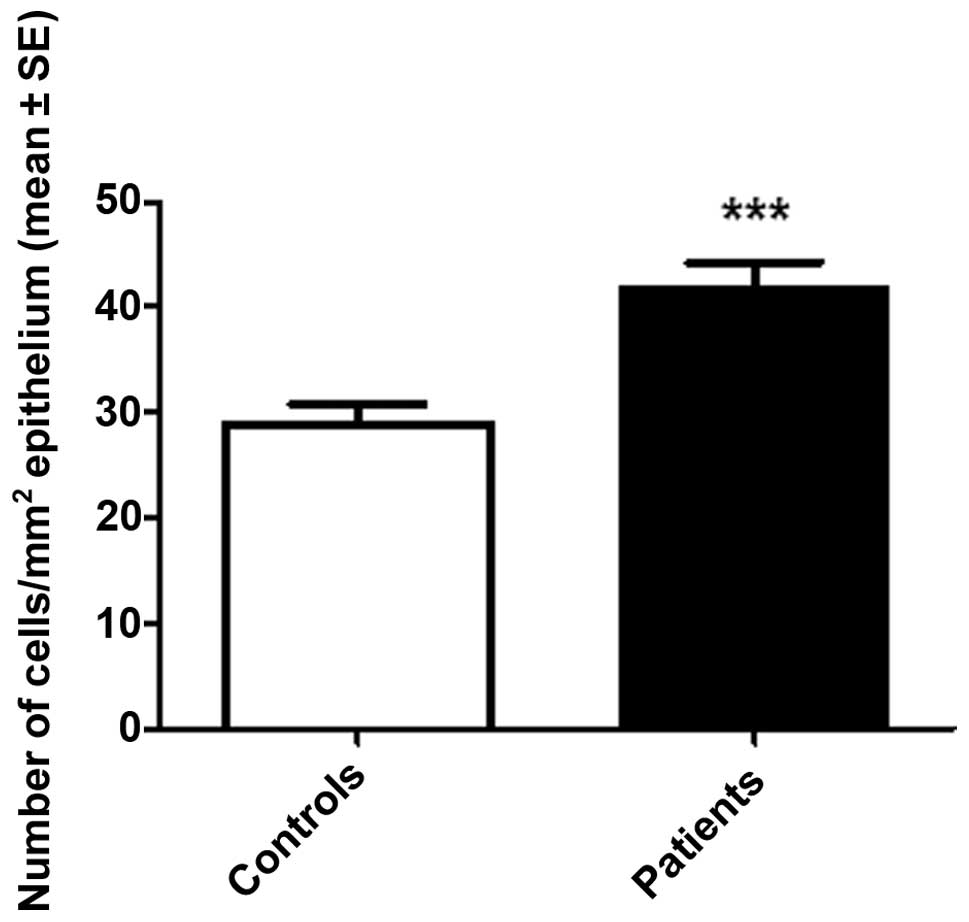
an extremely high density of colonic chromogranin A-positive cells
(Fig. 2), and were subsequently
identified as having LC (38).
This unexpected observation was confirmed in a larger cohort of
patients with LC, and led to the colonic chromogranin A-positive
cell density being proposed as a diagnostic marker for LC (Figs. 3 and 4) (39). The use of the chromogranin
A-positive cell density as a marker for the diagnosis of LC has a
high sensitivity (97 and 100%, in the right and left colon,
respectively) and specificity (98 and 94%, in the right and left
colon, respectively) (39). The
time and cost associated with the use of this marker are the same
as those for the detection of lymphocytes. Using the chromogranin
A-positive cell density as a marker has several advantages: i) it
has a high sensitivity and specificity in the left colon, thus
sigmoidoscopy with biopsy can be used in the diagnosis; ii) it is
not affected by the artifact caused by lymphoid aggregates and is
thus useful in borderline cases where lymphocyte infiltration does
not completely fulfill the definition of LC; and iii) it can be
used to differentiate patients with LC from those with IBS,
refractory celiac disease, lymphocytic enterocolitis, autoimmune
enteropathy and lymphoma.
LC and IBS have similar symptoms and normal
radiological and endoscopic appearances of the colon (40), which means that patients with LC
are at risk of being misdiagnosed as IBS (24,40–43). The task of differentiating between
LC and IBS is made even more difficult by the fact that some
patients with IBS exhibit an increase in the number of
intraepithelial lymphocytes (40,44). The use of colonic chromogranin
A-positive cell density as a marker for LC may prove to be
beneficial in such cases, since the density of this marker in the
colon is low in patients with IBS (45).
Celiac disease has been reported to be associated
with LC; between 4.3 and 31% of patients with celiac disease
exhibit histopathological changes typical of LC (46–48). Approximately 7% of patients with
LC have celiac disease (6,10,23,25,49–51).
It has been speculated that colonic lymphocyte infiltration
observed in the colon of patients with celiac disease is part of
the same autoimmune pathogenesis observed in the small intestine.
The coexistence of celiac disease and LC makes it necessary to
further examine patients with celiac disease who do not respond to
a gluten-free diet. Thus, one of the first tasks in clinical
practice is to exclude the possibility of celiac disease in
patients exhibiting LC symptomology.
It is generally accepted that LC is secondary to an
abnormal immune reaction in predisposed individuals.
Gastrointestinal infections, drugs and/or autoimmunity possibely
trigger this reaction. An abnormality in the colon neuroendocrine
system has also been reported and seems to play a role in the
pathophysiology of LC.
Gastrointestinal infection as an etiological factor
of LC is supported by a case report of a 19-year-old patient who
developed LC following Campylobacter infection (52), and by a significant seasonal
pattern in the incidence of LC (53). Associations with Clostridium
difficile, Yersinia enterocolitica and Campylobacter
jejuni have been observed in collagenous colitis, which is a
disease related to LC (54–56). The initial inflammatory response
following gastrointestinal infection shows an increase in
CD3+ lymphocytes, CD8 intraepithelial lymphocytes and
calprotectin-positive macrophages (57). These changes rapidly diminish in
most subjects; however, a small number of patients with persistent
symptoms fail to show this decline (57). This process can commence
irrespective of the type of infectious agents (i.e. virus, bacteria
or protozoa). Consequently, a report of no association being found
between anti-Yersinia antibodies and LC in 19 patients does
not exclude an infectious genesis for LC (58). The issue still remains however, of
why only a proportion of patients that suffer gastroenteritis
develop LC. Genetic factors may be of importance, since variants of
the matrix metalloproteinase-9 (MMP-9) gene have been observed in
collagenous colitis (59).
A role for gastrointestinal infections in the
pathogenesis of LC has been supported by the striking similarity
between LC and post-infectious IBS (PI-IBS). These conditions have
a similar clinical presentation and both can regress spontaneously
(57,60). Both LC and PI-IBS show an
intra-epithelial and submucosal infiltration of lymphocytes and
mast cells, and exhibit a high density of colonic serotonin and
peptide YY (PYY) cells (40,61–64). These similarities have prompted
suggestions that LC and PI-IBS are the same disorder (62).
Several studies have demonstrated an association
between LC and autoimmune diseases, such as celiac disease,
diabetes mellitus, arthritis and thyroid diseases (21,70). Moreover, patients with LC exhibit
positivity in the antinuclear antibody (ANA) test (21). Associations between microscopic
colitis and the TNF2 allele, HLA A1 allele and cytokine gene
polymorphism in the IL-6–174 GG genotype have been reported
(71–73).
Colonic nitric oxide (NO) levels are markedly
increased in patients with LC, which is due to an increase in NO
synthase levels in the colonic epithelium (74–76). The colonic serotonin (Figs. 4 and 5) and PYY cell densities (Figs. 6 and 7) have been reported to be extremely
high in patients with LC (62).
NO increases intestinal motility and stimulates the intestinal
secretion of water and electrolytes (77,78). Serotonin activates the submucosal
sensory branch of the enteric nervous system, and controls
gastrointestinal motility and chloride secretion via inter-neurons
and motor neurons (79,80). PYY stimulates the absorption of
water and electrolytes, and is a major regulator of the ‘ileal
brake’ (81). Furthermore, PYY
inhibits prostaglandin E2 and vasoactive intestinal polypeptide
(VIP) actiation, which stimulate intestinal fluid secretion
(82–84). The administration of PYY has been
shown to inhibit diarrhea in experimental mouse models by reducing
intestinal fluid secretion and delaying colonic transit (85).
Serotonin secretion by enterochromaffin (EC) cells
can be enhanced or attenuated by the secretory products of immune
cells, such as CD4+ T cells (86). Furthermore, serotonin modulates
the immune response (86). EC
cells are in contact with or very close to CD3+ and
CD20+ lymphocytes and several serotonergic receptors
have been characterized in lymphocytes, monocytes, macrophages and
dendritic cells (87–91). It is therefore conceivable that
the increase in colonic serotonin cell density in patients with LC
results from the interaction between lymphocytes and EC cells.
Serotonin stimulates NO secretion, which is partly responsible for
its effects on intestinal motility and thus secretion is partly due
to the activation of NO (78,88–91). It is reasonable to assume that the
increase in colonic NO levels is caused by the increase in
serotonin levels observed in patients with LC. The increase in
colonic serotonin and NO levels in patients with LC accelerates
intestinal motility and water and electrolyte secretion observed in
LC diarrhea, which is of secretory character (92). Accelerated intestinal motility and
secretion may be the cause of the compensatory secondary increase
in PYY levels in the colon of patients with LC. This compensatory
mechanism may explain the occurrence of intermittent diarrhea in
some patients with LC, as an increase in PYY levels can compensate
for the increased secretion and motility for only short periods ot
time (Fig. 8).
The drugs commonly used in the treatment of LC are
loperamide, 5-aminosalicylates, budesonide, prednisolone, bismuth
subsalicylate, immunosuppression and anti-TNF-α therapies (17,93–95) (Fig.
9). Cholestyramine has also been recommended for the treatment
of LC, but its clinical response was possibly due to the fact that
the patients treated suffered from bile acid malabsorption
(93). Loperamide, which is a
symptomatic non-specific anti-diarrheal agent, is often the
first-line therapy prescribed. It is administered as a tablet of 2
mg (up to 8 tablets taken daily if necessary). A complete or
partial response to loperamide has been observed in 70–73% of
patients (11,22). 5-Aminosalicylates, such as
sulfasalazine and mesalamine are a common therapy for LC in
clinical practice due to positive responses and tolerance in the
majority of patients (22,96).
However, there have been several reports of 5-aminosalicylates
inducing remission in <50% of patients (25,97). Budesonide is the most documented
drug used in the treatment of LC. It is a glucocorticoid with no
significant adverse events that are known to occur in systemic
treatments with corticosteroids. This is due to its limited
systemic availability caused by the extensive first-pass hepatic
metabolism by cytochrome P-450 enzymes. Clinical remission has been
reported in 82–86% of patients with LC following 6-weeks of
treatment with 9 mg of budesonide daily (11,98). Relapse after cessation of
budesonide treatment has been shown to occur in 14–44% of patients
after 1–7 months (11,98). The relapsed patients responded to
new treatment and remained in clinical remission with a maintenance
therapy of a median daily budesonide dose of 3 mg (11,98). Four weeks of treatment with
prednisolone at 40 mg daily tapered by 5 mg/week resulted in
complete or partial clinical remission in 87–94% of patients with
LC (11,99). However, the majority of these
patients relapsed after termination of the therapy (11,22). Bismuth subsalicylate at a dose of
2 or 3 tablets (262 mg each) 3 or 4 times daily for 8 weeks induced
remission in the majority of patients which was sustained up to
7–23 months (22,100). No toxicity was absorbed despite
the high dose of bismuth, which may have been due to the short
treatment duration or low bismuth absorption. However, treatment
with bismuth is not without any toxic effects and its availability
is limited in certain countries. Treatment with azathioprine at
2–2.5 mg/kg/day, with 6-mercaptopurine at 1 mg/kg/day both
indefinitely, and anti-TNF-α have produced good results in patients
with refractory LC (94,99,101). The indications for surgical
therapy are limited (102).
Concomitant drug use should be assessed and drugs
that potentially cause drug-induced LC should be discontinued, if
possible. Considering the possibility that LC and PI-IBS can be the
same disorder, diet, regular exercise and probiotic intake
recommendations provided to patients with IBS (103–105) should also be recommended for
patients with LC. The benign course and high rate of spontaneous
remission of LC denotes that drugs with severe side-effects should
be avoided if possible. Furthermore, the drug cost and drug
coverage may be limiting factors for some patients. These aspects
should be taken into account when selecting treatment. An
algorithmic approach to the treatment of LC is recommended
(Fig. 9).
LC is not a rare disorder as previously thought.
Although LC is considered a diarrheal disorder, it can present with
other symptoms, such as constipation or abdominal pain. Colonic
biopsy samples in patients presenting with symptoms other than
diarrhea and with a macroscopically normal colon should be taken
more frequently than routinely done at present. The density of
colonic chromogranin A-positive cells may be used as a diagnostic
tool for LC. The cause of LC may be a low-grade inflammation with
an interaction with the colonic neuroendocrine system. There are
good reasons to suspect that LC and PI-IBS are one and the same
disorder. The benign course and high rate of spontaneous remission,
as well as the drug cost and drug coverage should be taken into
accout when making decisions regarding treatment. An algorithmic
approach to the treatment of LC is preferable.
|
1
|
Read NW, Krejs GJ, Read MG, Santa Ana CA,
Morawski SG and Fordtran JS: Chronic diarrhea of unknown origin.
Gastroenterology. 78:264–271. 1980.PubMed/NCBI
|
|
2
|
Chande N, MacDonald JK and McDonald JW:
Interventions for treating microscopic colitis: a Cochrane
Inflammatory Bowel Disease and Functional Bowel Disorders Review
Group systematic review of randomized trials. Am J Gastroenterol.
104:235–241; quiz 234, 242, 2009.
|
|
3
|
Pardi DS: Microscopic colitis: an update.
Inflamm Bowel Dis. 10:860–870. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pardi DS and Kelly CP: Microscopic
colitis. Gastroenterology. 140:1155–1165. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yen EF and Pardi DS: Review of the
microscopic colitides. Curr Gastroenterol Rep. 13:458–464. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Olesen M, Eriksson S, Bohr J, Jarnerot G
and Tysk C: Microscopic colitis: a common diarrhoeal disease. An
epidemiological study in Orebro, Sweden, 1993–1998. Gut.
53:346–350. 2004.PubMed/NCBI
|
|
7
|
Guagnozzi D, Lucendo AJ, Angueira-Lapena
T, Gonzalez-Castillo S and Tenias Burillo JM: Prevalence and
incidence of microscopic colitis in patients with diarrhoea of
unknown aetiology in a region in central Spain. Dig Liver Dis.
44:384–388. 2012. View Article : Google Scholar
|
|
8
|
Fernandez-Banares F, Salas A, Forne M,
Esteve M, Espinos J and Viver JM: Incidence of collagenous and
lymphocytic colitis: a 5-year population-based study. Am J
Gastroenterol. 94:418–423. 1999.PubMed/NCBI
|
|
9
|
Fernandez-Banares F, Salas A, Esteve M, et
al: Evolution of the incidence of collagenous colitis and
lymphocytic colitis in Terrassa, Spain: a population-based study.
Inflamm Bowel Dis. 17:1015–1020. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bjornbak C, Engel PJ, Nielsen PL and Munck
LK: Microscopic colitis: clinical findings, topography and
persistence of histopathological subgroups. Aliment Pharmacol Ther.
34:1225–1234. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Agnarsdottir M, Gunnlaugsson O, Orvar KB,
et al: Collagenous and lymphocytic colitis in Iceland. Dig Dis Sci.
47:1122–1128. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pardi DS, Loftus EV Jr, Smyrk TC, et al:
The epidemiology of microscopic colitis: a population based study
in Olmsted County, Minnesota. Gut. 56:504–508. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Williams JJ, Kaplan GG, Makhija S, et al:
Microscopic colitis-defining incidence rates and risk factors: a
population-based study. Clin Gastroenterol Hepatol. 6:35–40. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Rasmussen MA and Munck LK: Systematic
review: are lymphocytic colitis and collagenous colitis two
subtypes of the same disease - microscopic colitis? Aliment
Pharmacol Ther. 36:79–90. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fernandez-Banares F, Esteve M and Viver
JM: Epidemiology of microscopic colitis. Gut. 56:10332007.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brown WR and Tayal S: Microscopic colitis.
A review. J Dig Dis. 14:277–281. 2013. View Article : Google Scholar
|
|
17
|
Yen EF and Pardi DS: Review article:
microscopic colitis - lymphocytic, collagenous and ‘mast cell’
colitis. Aliment Pharmacol Ther. 34:21–32. 2011.
|
|
18
|
Mohamed N, Marais M and Bezuidenhout J:
Microscopic colitis as a missed cause of chronic diarrhea. World J
Gastroenterol. 17:1996–2002. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pascua MF, Kedia P, Weiner MG, Holmes J,
Ellenberg J and Lewis JD: Microscopic colitis and medication use.
Clin Med Insights Gastroenterol. 2010:11–19. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Munch A, Aust D, Bohr J, et al:
Microscopic colitis: Current status, present and future challenges:
statements of the European Microscopic Colitis Group. J Crohns
Colitis. 6:932–945. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mahajan D, Goldblum JR, Xiao SY, Shen B
and Liu X: Lymphocytic colitis and collagenous colitis: a review of
clinicopathologic features and immunologic abnormalities. Adv Anat
Pathol. 19:28–38. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pardi DS, Ramnath VR, Loftus EV Jr,
Tremaine WJ and Sandborn WJ: Lymphocytic colitis: clinical
features, treatment, and outcomes. Am J Gastroenterol.
97:2829–2833. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mullhaupt B, Guller U, Anabitarte M,
Guller R and Fried M: Lymphocytic colitis: clinical presentation
and long term course. Gut. 43:629–633. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Barta Z, Mekkel G, Csipo I, et al:
Microscopic colitis: a retrospective study of clinical presentation
in 53 patients. World J Gastroenterol. 11:1351–1355. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Olesen M, Eriksson S, Bohr J, Jarnerot G
and Tysk C: Lymphocytic colitis: a retrospective clinical study of
199 Swedish patients. Gut. 53:536–541. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Koskela RM, Niemela SE, Karttunen TJ and
Lehtola JK: Clinical characteristics of collagenous and lymphocytic
colitis. Scand J Gastroenterol. 39:837–845. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Baert F, Wouters K, D'Haens G, et al:
Lymphocytic colitis: a distinct clinical entity? A
clinicopathological confrontation of lymphocytic and collagenous
colitis. Gut. 45:375–381. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sveinsson OA, Orvar KB, Birgisson S,
Agnarsdottir M and Jonasson JG: Clinical features of microscopic
colitis in a nation-wide follow-up study in Iceland. Scand J
Gastroenterol. 43:955–960. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kao KT, Pedraza BA, McClune AC, et al:
Microscopic colitis: a large retrospective analysis from a health
maintenance organization experience. World J Gastroenterol.
15:3122–3127. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liszka L, Woszczyk D and Pajak J:
Histopathological diagnosis of microscopic colitis. J Gastroenterol
Hepatol. 21:792–797. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Veress B, Lofberg R and Bergman L:
Microscopic colitis syndrome. Gut. 36:880–886. 1995. View Article : Google Scholar
|
|
32
|
Thijs WJ, van Baarlen J, Kleibeuker JH and
Kolkman JJ: Microscopic colitis: prevalence and distribution
throughout the colon in patients with chronic diarrhoea. Neth J
Med. 63:137–140. 2005.PubMed/NCBI
|
|
33
|
Fine KD, Seidel RH and Do K: The
prevalence, anatomic distribution, and diagnosis of colonic causes
of chronic diarrhea. Gastrointest Endosc. 51:318–326. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Matteoni CA, Wang N, Goldblum JR,
Brzezinski A, Achkar E and Soffer EE: Flexible sigmoidoscopy for
the detection of microscopic colitis. Am J Med. 108:416–418. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Taupenot L, Harper KL and O'Connor DT: The
chromogranin-secretogranin family. N Engl J Med. 348:1134–1149.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wiedenmann B and Huttner WB: Synaptophysin
and chromogranins/secretogranins - widespread constituents of
distinct types of neuroendocrine vesicles and new tools in tumor
diagnosis. Virchows Arch B Cell Pathol Incl Mol Pathol. 58:95–121.
1989. View Article : Google Scholar
|
|
37
|
Deftos LJ: Chromogranin A: its role in
endocrine function and as an endocrine and neuroendocrine tumor
marker. Endocr Rev. 12:181–187. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
El-Salhy M, Lomholt-Beck B and Gundersen
D: High chromogranin A cell density in the colon of patients with
lymphocytic colitis. Mol Med Rep. 4:603–605. 2011.PubMed/NCBI
|
|
39
|
El-Salhy M, Gundersen D, Hatlebakk JG and
Hausken T: Chromogranin A cell density as a diagnostic marker for
lymphocytic colitis. Dig Dis Sci. 57:3154–3159. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
El-Salhy M, Gundersen D, Hatlebakk JG and
Hausken T: Irritable Bowel Syndrome: Diagnosis Pathogenesis and
Treatment Options. Nova Science Publisher; New York: 2012
|
|
41
|
El-Salhy M, Halwe J, Lomholt-Beck B and
Gundersen D: The prevalence of inflammatory bowel diseases and
microscopic colitis and colorectal cancer in patients with
irritable bowel syndrome. Gastroenterology Insights. 3:7–10. 2011.
View Article : Google Scholar
|
|
42
|
Madisch A, Bethke B, Stolte M and Miehlke
S: Is there an association of microscopic colitis and irritable
bowel syndrome - a subgroup analysis of placebo-controlled trials.
World J Gastroenterol. 11:64092005.PubMed/NCBI
|
|
43
|
Rahman MA, Raihan AS, Ahamed DS, et al:
Symptomatic overlap in patients with diarrhea predominant irritable
bowel syndrome and microscopic colitis in a sub group of
Bangladeshi population. Bangladesh Med Res Counc Bull. 38:33–38.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Arevalo F, Aragon V, Montes P, Guzman E
and Monge E: Increase of intraepithelial lymphocytes in patients
with irritable bowel syndrome. Rev Gastroenterol Peru. 31:315–318.
2011.(In Spanish).
|
|
45
|
El-Salhy M, Lomholt-Beck B and Hausken T:
Chromogranin A as a possible tool in the diagnosis of irritable
bowel syndrome. Scand J Gastroenterol. 45:1435–1439. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Dewar DH, Donnelly SC, McLaughlin SD,
Johnson MW, Ellis HJ and Ciclitira PJ: Celiac disease: management
of persistent symptoms in patients on a gluten-free diet. World J
Gastroenterol. 18:1348–1356. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wolber R, Owen D and Freeman H: Colonic
lymphocytosis in patients with celiac sprue. Hum Pathol.
21:1092–1096. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Green PH, Yang J, Cheng J, Lee AR, Harper
JW and Bhagat G: An association between microscopic colitis and
celiac disease. Clin Gastroenterol Hepatol. 7:1210–1216. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Fraser AG, Warren BF, Chandrapala R and
Jewell DP: Microscopic colitis: a clinical and pathological review.
Scand J Gastroenterol. 37:1241–1245. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Abdulkarim AS and Murray JA: Celiac
disease. Curr Treat Options Gastroenterol. 5:27–38. 2002.
View Article : Google Scholar
|
|
51
|
Ciclitira PJ, King AL and Fraser JS: AGA
technical review on Celiac Sprue. American Gastroenterological
Association Gastroenterology. 120:1526–1540. 2001.PubMed/NCBI
|
|
52
|
Perk G, Ackerman Z, Cohen P and Eliakim R:
Lymphocytic colitis: a clue to an infectious trigger. Scand J
Gastroenterol. 34:110–112. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
LaSala PR, Chodosh AB, Vecchio JA, Schned
LM and Blaszyk H: Seasonal pattern of onset in lymphocytic colitis.
J Clin Gastroenterol. 39:891–893. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Erim T, Alazmi WM, O'Loughlin CJ and
Barkin JS: Collagenous colitis associated with Clostridium
difficile: a cause effect? Dig Dis Sci. 48:1374–1375. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Bohr J, Nordfelth R, Jarnerot G and Tysk
C: Yersinia species in collagenous colitis: a serologic
study. Scand J Gastroenterol. 37:711–714. 2002. View Article : Google Scholar
|
|
56
|
Makinen M, Niemela S, Lehtola J and
Karttunen TJ: Collagenous colitis and Yersinia
enterocolitica infection. Dig Dis Sci. 43:1341–1346. 1998.
View Article : Google Scholar
|
|
57
|
Spiller RC, Jenkins D, Thornley JP, et al:
Increased rectal mucosal enteroendocrine cells, T lymphocytes, and
increased gut permeability following acute Campylobacter
enteritis and in post-dysenteric irritable bowel syndrome. Gut.
47:804–811. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Tamboli CP, Good MR, Reynolds EM, Sharma P
and Mitros FA: Anti-Yersinia antibodies are not associated
with microscopic colitis in an American case-control study. Scand J
Gastroenterol. 46:1442–1448. 2011.
|
|
59
|
Madisch A, Hellmig S, Schreiber S, Bethke
B, Stolte M and Miehlke S: Allelic variation of the matrix
metalloproteinase-9 gene is associated with collagenous colitis.
Inflamm Bowel Dis. 17:2295–2298. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Spiller R and Garsed K: Postinfectious
irritable bowel syndrome. Gastroenterology. 136:1979–1988. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Dunlop SP, Jenkins D, Neal KR and Spiller
RC: Relative importance of enterochromaffin cell hyperplasia,
anxiety, and depression in postinfectious IBS. Gastroenterology.
125:1651–1659. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
El-Salhy M, Gundersen D, Hatlebakk JG and
Hausken T: High densities of serotonin and peptide YY cells in the
colon of patients with lymphocytic colitis. World J Gastroenterol.
18:6070–6075. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Park JH, Rhee PL, Kim G, et al:
Enteroendocrine cell counts correlate with visceral
hypersensitivity in patients with diarrhoea-predominant irritable
bowel syndrome. Neurogastroenterol Motil. 18:539–546. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Kim HS, Lim JH, Park H and Lee SI:
Increased immunoendocrine cells in intestinal mucosa of
postinfectious irritable bowel syndrome patients 3 years after
acute Shigella infection - an observation in a small case
control study. Yonsei Med J. 51:45–51. 2010.PubMed/NCBI
|
|
65
|
Beaugerie L and Pardi DS: Review article:
drug-induced microscopic colitis - proposal for a scoring system
and review of the literature. Aliment Pharmacol Ther. 22:277–284.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Fernandez-Banares F, Esteve M, Espinos JC,
et al: Drug consumption and the risk of microscopic colitis. Am J
Gastroenterol. 102:324–330. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Bjarnason I, Hayllar J, MacPherson AJ and
Russell AS: Side effects of nonsteroidal anti-inflammatory drugs on
the small and large intestine in humans. Gastroenterology.
104:1832–1847. 1993.PubMed/NCBI
|
|
68
|
Leung WK, Bjarnason I, Wong VW, Sung JJ
and Chan FK: Small bowel enteropathy associated with chronic
low-dose aspirin therapy. Lancet. 369:6142007. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Takeuchi K, Smale S, Premchand P, et al:
Prevalence and mechanism of nonsteroidal anti-inflammatory
drug-induced clinical relapse in patients with inflammatory bowel
disease. Clin Gastroenterol Hepatol. 4:196–202. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Tysk C, Bohr J, Nyhlin N, Wickbom A and
Eriksson S: Diagnosis and management of microscopic colitis. World
J Gastroenterol. 14:7280–7288. 2008. View Article : Google Scholar
|
|
71
|
Giardiello FM, Lazenby AJ, Yardley JH, et
al: Increased HLA A1 and diminished HLA A3 in lymphocytic colitis
compared to controls and patients with collagenous colitis. Dig Dis
Sci. 37:496–499. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Koskela RM, Karttunen TJ, Niemela SE,
Lehtola JK, Ilonen J and Karttunen RA: Human leucocyte antigen and
TNFalpha polymorphism association in microscopic colitis. Eur J
Gastroenterol Hepatol. 20:276–282. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Koskela RM, Karttunen TJ, Niemela SE,
Lehtola JK, Bloigu RS and Karttunen RA: Cytokine gene polymorphism
in microscopic colitis association with the IL-6–174 GG genotype.
Eur J Gastroenterol Hepatol. 23:607–613. 2011.PubMed/NCBI
|
|
74
|
Lundberg JO, Herulf M, Olesen M, et al:
Increased nitric oxide production in collagenous and lymphocytic
colitis. Eur J Clin Invest. 27:869–871. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Olesen M, Middelveld R, Bohr J, et al:
Luminal nitric oxide and epithelial expression of inducible and
endothelial nitric oxide synthase in collagenous and lymphocytic
colitis. Scand J Gastroenterol. 38:66–72. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Kolios G, Rooney N, Murphy CT, Robertson
DA and Westwick J: Expression of inducible nitric oxide synthase
activity in human colon epithelial cells: modulation by T
lymphocyte derived cytokines. Gut. 43:56–63. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Mourad FH, Turvill JL and Farthing MJ:
Role of nitric oxide in intestinal water and electrolyte transport.
Gut. 44:143–147. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Kuwahara A, Kuramoto H and Kadowaki M:
5-HT activates nitric oxide-generating neurons to stimulate
chloride secretion in guinea pig distal colon. Am J Physiol.
275:G829–G834. 1998.PubMed/NCBI
|
|
79
|
Gershon MD: Plasticity in serotonin
control mechanisms in the gut. Curr Opin Pharmacol. 3:600–607.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Kellum JM, Albuquerque FC, Stoner MC and
Harris RP: Stroking human jejunal mucosa induces 5-HT release and
Cl- secretion via afferent neurons and 5-HT4 receptors. Am J
Physiol. 277:G515–G520. 1999.PubMed/NCBI
|
|
81
|
Walsh JH: Gastrointestinal hormones.
Physiology of the Gastrointestinal Tract. Johnson LR, Alpers DH,
Christensen J, Jacobson ED and Walsh JH: Raven Press; New York: pp.
1–128. 1994
|
|
82
|
Goumain M, Voisin T, Lorinet AM, et al:
The peptide YY- preferring receptor mediating inhibition of small
intestinal secretion is a peripheral Y(2) receptor: pharmacological
evidence and molecular cloning. Mol Pharmacol. 60:124–134.
2001.
|
|
83
|
Souli A, Chariot J, Voisin T, et al:
Several receptors mediate the antisecretory effect of peptide YY,
neuropeptide Y, and pancreatic polypeptide on VIP-induced fluid
secretion in the rat jejunum in vivo. Peptides. 18:551–557. 1997.
View Article : Google Scholar
|
|
84
|
Whang EE, Hines OJ, Reeve JR Jr, et al:
Antisecretory mechanisms of peptide YY in rat distal colon. Dig Dis
Sci. 42:1121–1127. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Moriya R, Shirakura T, Hirose H, Kanno T,
Suzuki J and Kanatani A: NPY Y2 receptor agonist PYY(3–36) inhibits
diarrhea by reducing intestinal fluid secretion and slowing colonic
transit in mice. Peptides. 31:671–675. 2010.
|
|
86
|
Khan WI and Ghia JE: Gut hormones:
emerging role in immune activation and inflammation. Clin Exp
Immunol. 161:19–27. 2010.PubMed/NCBI
|
|
87
|
Yang GB and Lackner AA: Proximity between
5-HT secreting enteroendocrine cells and lymphocytes in the gut
mucosa of rhesus macaques (Macaca mulatta) is suggestive of
a role for enterochromaffin cell 5-HT in mucosal immunity. J
Neuroimmunol. 146:46–49. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Briejer MR, Akkermans LM, Meulemans AL,
Lefebvre RA and Schuurkes JA: Nitric oxide is involved in
5-HT-induced relaxations of the guinea-pig colon ascendens in
vitro. Br J Pharmacol. 107:756–761. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Stoner MC, Scherr AM, Lee JA, Wolfe LG and
Kellum JM: Nitric oxide is a neurotransmitter in the chloride
secretory response to serotonin in rat colon. Surgery. 128:240–245.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Stoner MC and Kellum JM: Both serotonin
and a nitric-oxide donor cause chloride secretion in rat
colonocytes by stimulating cGMP. Surgery. 130:236–241. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Kadowaki M, Gershon MD and Kuwahara A: Is
nitric oxide involved in 5-HT-induced fluid secretion in the gut?
Behav Brain Res. 73:293–296. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Protic M, Jojic N, Bojic D, et al:
Mechanism of diarrhea in microscopic colitis. World J
Gastroenterol. 11:5535–5539. 2005.PubMed/NCBI
|
|
93
|
Stroehlein JR: Microscopic colitis. Curr
Treat Options Gastroenterol. 10:231–236. 2007. View Article : Google Scholar
|
|
94
|
Esteve M, Mahadevan U, Sainz E, Rodriguez
E, Salas A and Fernandez-Banares F: Efficacy of anti-TNF therapies
in refractory severe microscopic colitis. J Crohns Colitis.
5:612–618. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Chande N: Microscopic colitis: an approach
to treatment. Can J Gastroenterol. 22:686–688. 2008.PubMed/NCBI
|
|
96
|
Fernandez-Banares F, Salas A, Esteve M,
Espinos J, Forne M and Viver JM: Collagenous and lymphocytic
colitis: evaluation of clinical and histological features, response
to treatment, and long-term follow-up. Am J Gastroenterol.
98:340–347. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Calabrese C, Fabbri A, Areni A, Zahlane D,
Scialpi C and Di Febo G: Mesalazine with or without cholestyramine
in the treatment of microscopic colitis: randomized controlled
trial. J Gastroenterol Hepatol. 22:809–814. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Miehlke S, Madisch A, Karimi D, et al:
Budesonide is effective in treating lymphocytic colitis: a
randomized double-blind placebo-controlled study. Gastroenterology.
136:2092–2100. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Pardi DS, Loftus EV Jr, Tremaine WJ and
Sandborn WJ: Treatment of refractory microscopic colitis with
azathioprine and 6-mercaptopurine. Gastroenterology. 120:1483–1484.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Fine KD and Lee EL: Efficacy of open-label
bismuth subsalicylate for the treatment of microscopic colitis.
Gastroenterology. 114:29–36. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Vennamaneni SR and Bonner GF: Use of
azathioprine or 6-mercaptopurine for treatment of steroid-dependent
lymphocytic and collagenous colitis. Am J Gastroenterol.
96:2798–2799. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Varghese L, Galandiuk S, Tremaine WJ and
Burgart LJ: Lymphocytic colitis treated with proctocolectomy and
ileal J-pouch-anal anastomosis: report of a case. Dis Colon Rectum.
45:123–126. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Ostgaard H, Hausken T, Gundersen D and
El-Salhy M: Diet and effects of diet management on quality of life
and symptoms in patients with irritable bowel syndrome. Mol Med
Rep. 5:1382–1390. 2012.PubMed/NCBI
|
|
104
|
El-Salhy M, Ostgaard H, Gundersen D,
Hatlebakk JG and Hausken T: The role of diet in the pathogenesis
and management of irritable bowel syndrome (Review). Int J Mol Med.
29:723–731. 2012.PubMed/NCBI
|
|
105
|
El-Salhy M, Lillebø E, Reinemo A, Salmelid
L and Hausken T: Effects of a health program comprising
reassurance, diet management, probiotics administration and regular
exercise on symptoms and quality of life in patients with irritable
bowel syndrome. Gastroenterology Insights. 2:21–26. 2010.
View Article : Google Scholar
|