Introduction
Essential oils (EOs) are natural volatile complex
compounds that are characterized by a strong scent and produced by
aromatic plants as secondary metabolites (1). EOs have largely been employed for
their well-known natural antitumor, anti-inflammatory, antioxidant
and anti-bacterial properties (2). Currently, approximately 3,000 EOs
have been identified, 300 of which are commercially important,
particularly in the pharmaceutical, agronomic, food, sanitary,
cosmetic and perfume industries. EOs and some of their components
are used in the perfume and cosmetic industry, as well as in the
manufacture of sanitary products, dentistry and agriculture; they
have also been employed as food preservatives, additives and
natural remedies (3,4). EOs are also used in massages as
mixtures with vegetable oil, or in baths. Their most frequent use
is in aromatherapy (1). Some EOs
may have particular medicinal properties that are thought to cure
certain organ dysfunctions or systemic disorders. EOs have long
been used as the components of fragrances. Over past few years,
there is a renewed interest in understanding their biological
properties due to their potential novel applications in human
health, agriculture and the environment.
The prevalence of allergic diseases, such as
allergic rhinitis, atopic dermatitis, asthma and food allergies has
increased in the majority of countries (5). Immunologically active mast cells and
basophils express the high-affinity receptor for immunoglobulin E
(IgE) on their surface and play critical roles in various
biological processes associated with allergic diseases (6). The interaction of multivalent
antigens with surface-bound IgE triggers the secretion of the
mediators stored in cytoplasmic granules and causes a de
novo synthesis of cytokines (7), that in turn activate the migration
of neutrophils and macrophages, causing tissue inflammation
(8).
Inflammation is an initial host immune reaction
mediated by inflammatory cytokines, such as tumor necrosis factor-α
(TNF-α) (9). Macrophages play an
important role in the host defense against noxious substances
(10). Macrophage activation by
lipopolysaccharide (LPS), a major component of Gram-negative
bacteria outer membranes, leads to the increased production of
pro-inflammatory cytokines, thereby mediating the major cytotoxic
and pro-apoptotic mechanisms that participate in the innate
response in many mammals (11).
However, the cytokine overproduction by activated macrophages has
been implicated in the pathophysiology of several inflammatory
diseases, including rheumatoid arthritis, atherosclerosis, chronic
hepatitis, pulmonary fibrosis and inflammatory brain diseases
(11). Therefore, LPS-stimulated
macrophages serve as a useful model for studying inflammation and
the potential mechanisms of action of anti-inflammatory
compounds.
In this study, the anti-allergic and
anti-inflammatory properties of 20 different types of EOs isolated
from herbal plants and citrus fruits were examined. Lemongrass EO
exhibited the strongest anti-allergic and anti-inflammatory
properties and therefore, the major chemical constituents of this
EO were evaluated by gas chromatography (GC)-mass spectrometry
(MS). These results are of great significance in recognizing the
novel future applications of EOs derived from these plants.
Materials and methods
Materials
The 20 commercially available EOs were donated by
Nagaoka Perfumery Co., Ltd. (Osaka, Japan). The 17 types of
hydrodistilled EOs used in the study were from herbal plants,
including basil (Ocimum basilicum L., produced in the Union
of the Comoros), caraway (Carum carvi L., Europe), carrot
seed (Daucus carota L., France), celery seed (Apium
graveolens L., India), chamomile (Matricaria chamomilla
L., Egypt), citronella (Cymbopogon winterianus Jowitt,
Indonesia), clary sage (Salvia sclarea L., USA), clove
(Syzygium aromaticum L., Madagascar), cumin (Cuminum
cyminum L., India), eucalyptus (Eucalyptus globulus L.,
China), lemongrass [Cymbopogon citratus (DC.) Stapf, India],
marjoram (Majorana hortensis Moench., France), nutmeg
(Myristica fragrans Houtt., Indonesia), sage (Salvia
officinalis L., Albania), sandalwood (Santalum album L.,
India), spearmint (Mentha spicata L., USA), and thyme
(Thymus vulgaris L., India). The cold-pressed EOs were
derived from 3 species of citrus fruits: lemon (Citrus limon
L., USA), lime (C. aurantifolia Swingle, Mexico) and orange
(C. sinensis L., Portugal). Five terpenoids, citral
(3,7-dimethyl-2,6-octadien-1-al), geraniol, geranyl acetate,
linalool and camphene were purchased from Sigma-Aldrich, Inc. (St.
Louis, MO, USA). Tranilast and glycyrrhetinic acid, anti-allergic
and anti-inflammatory controls, respectively, were also obtained
from Sigma-Aldrich, Inc. Geranial was prepared by the oxidation of
geraniol with manganese dioxide, as previously described (12). For western blot analyses,
anti-nuclear factor-κB (NF-κB) p65 and anti-inhibitor of NF-κB
(IκB)-α antibodies were purchased from Santa Cruz Biotechnology,
Inc. (Santa Cruz, CA, USA). Horseradish peroxidase-conjugated
anti-rabbit immunoglobulin G (IgG) antibody was obtained from
Thermo Scientific (Kanagawa, Japan). All other reagents were of
analytical grade and were obtained from Nacalai Tesque, Inc.
(Kyoto, Japan).
Cells
RBL-2H3 rat basophilic leukemia and RAW264.7 murine
macrophage cell lines were obtained from the American Type Culture
Collection (ATCC; Manassas, VA, USA). The RBL-2H3 cells were
cultured in Eagle’s minimum essential medium supplemented with 4.5
g glucose/l plus 10% fetal bovine serum (FBS), 5 mM L-glutamine, 50
U/ml penicillin and 50 U/ml streptomycin. The RAW264.7 cells were
cultured in Dulbecco’s modified Eagle’s medium supplemented with
4.5 g glucose/l plus 10% FBS, 5 mM L-glutamine, 50 U/ml penicillin
and 50 U/ml streptomycin. All cells were cultured at 37°C in their
respective media in a humidified atmosphere of 5%
CO2/95% air.
Measurement of β-hexosaminidase
release
Mast cell degranulation-mediated histamine release
is accompanied by the release of β-hexosaminidase (13). Therefore, we performed a
β-hexosaminidase release assay, as previously described (14) with slight modifications to
determine the degranulation of mast cells as a measure of the
anti-allergenic activity of the examined compounds. Briefly, the
RBL-2H3 cells at 8×104 cells/well in 24-well plates were
washed with Tyrode’s buffer (137 mM NaCl, 5.6 mM glucose, 11.9 mM
NaHCO3, 2.7 mM KCl and 0.32 mM
NaH2PO4) containing 1 mM CaCl2 and
0.5 mM MgCl2, and the EOs (in 0.5% DMSO) were then added
to the culture wells at a final concentration of 100 μg/ml. The
cells were then stimulated with 5 μM A23187 and incubated for 30
min. The cell supernatant and total cell lysate dissolved in 2%
Triton X-100 were collected and mixed with substrate solution (2 mM
p-nitrophenyl-N-acetyl-β-D-glucosaminide in 0.1 M
sodium citrate buffer, pH 4.5). The mixture was incubated for 90
min at 37°C and the reaction was then terminated by the addition of
a stopping buffer containing a 0.2 M glycine buffer at pH 11.0.
Absorbance at 405 nm was measured using a microplate reader
(Vmax-K; Molecular Devices, LLC, Sunnyvale, CA, USA). The
EO-mediated inhibition of β-hexosaminidase release and activity was
expressed as the percentage inhibition and calculated using the
following formula: % activity = [(β-hexosaminidase release in the
absence of EO − β-hexosaminidase release in the presence of
EO)/β-hexosaminidase release in the absence of EO] ×100.
Measurement of the EO-mediated inhibition
of TNF-α
The RAW264.7 cells were placed in a 12-well plate at
5×104 cells/well and incubated for 24 h. The cells were
then pre-treated with each EO at a final concentration of 100 μg/ml
in 0.5% DMSO for 30 min prior to the addition of LPS (100 ng/ml).
Following LPS stimulation for 24 h, the cell culture medium was
collected to quantify the secreted TNF-α using a commercially
available enzyme-linked immunosorbent assay (ELISA) development
system (Bay Bioscience Co., Ltd., Kobe, Japan), in accordance with
the manufacturer’s instructions. The EO-mediated inhibition of
TNF-α production was expressed as the percentage inhibition,
calculated using the following formula: % activity = [(TNF-α
production in the absence of EO − TNF-α production in the presence
of EO)/TNF-α production in the absence of EO] ×100.
GC-MS
GC-MS was performed using an Agilent 6890 gas
chromatograph coupled with an Agilent 5972A MSD mass spectrometer
(Agilent Technologies Japan, Ltd., Tokyo, Japan). The chromatograph
was fitted with an InertCap WAX (polyethylene glycol)-fused silica
capillary column (60 m × 0.25 mm i.d.; film thickness, 0.25 μm; GL
Sciences, Inc., Tokyo, Japan), with purified helium (≥99.99995%)
carrier gas at 149 kPa inlet pressure, in split/splitless mode and
at 1.2 ml/min. The oven temperature was programmed as follows: 5
min at 70°C and then 3°C increments/min to 240°C, with both the
injector and transfer line held at 240°C. The mass spectrometer
operated at a 70 eV electron impact, with an ion source temperature
of 150°C, and the data collected in full scan mode over a mass scan
range revealed an m/z of 27–400. Each 0.6 μl sample was
injected in a split ratio of 1:60. A qualitative analysis was
performed by similarity searches of private and commercial mass
spectral databases (Wiley 275 and the National Institute of
Standards and Technology 02).
Cell treatment and preparation of nuclear
and whole cellular lysates and western blot analysis
The RAW264.7 cells were pre-treated with 10 μg/ml of
a test compound for 30 min followed by treatment with 100 ng/ml
LPS. After 30 min, the nuclear proteins and whole cell lysates were
collected, using the methods described in the study by Nishiumi
et al (15). The protein
concentrations of the nuclear proteins and whole cell lysates were
measured using the BCA™ Protein Assay kit (Pierce, Rockford, IL,
USA), according to the manufacturers instructions. Briefly, 25 μl
of each sample or 2 mg/ml BSA solution (as a standard) were added
to a 96-well microplate well, and then 200 μl of BCA Working
Reagent were added to each well. The microplate was incubated at
37°C for 30 min, and the protein concentration was determined by
measuring the absorbance at 575 nm. The nuclear proteins and whole
cell lysates were subjected to western blot analysis to evaluate
the nuclear translocation of NF-κB and the protein expression of
IκB-α.
The nuclear proteins (30–50 μg of protein/sample)
and whole cell lysates (50 μg of protein/sample) were boiled in a
quarter volume of sample buffer [1 M Tris-HCl, pH 7.5, 640 mM
2-mercaptoethanol, 0.2% bromphenol blue, 4% sodium dodecyl sulfate
(SDS) and 20% glycerol] and then separated on 10%
SDS-polyacrylamide gels. The proteins in the gels were transferred
onto a PVDF membrane. The membrane was blocked with 1% skimmed milk
(for NF-κB) or 5% bovine serum albumin in TBS-T (10 mM Tris-HCl,
100 mM NaCl and 0.5% Tween-20) (for IκB-α) and probed with an
anti-NF-κB p65 (1:1,000) or anti-IκB-α (1:1,000) antibody and
subsequently with the horseradish peroxidase-conjugated secondary
antibody. The protein-antibody complex was detected using
ChemiLumi-ONE (Nacalai Tesque, Inc.) and an Image Reader
(ImageQuant Imager 350/350Lumi; GE Healthcare Life Sciences,
Uppsala, Sweden). The intensity of each band was analyzed using
ImageJ software, which was developed at the National Institutes of
Health. The results were expressed as a percentage of the mean
protein expression ratio of the test compound-treated samples
compared with that of the control group (i.e., the cells incubated
in the absence of the test compound).
Animals
Female 6-week-old imprinting control region (ICR)
mice (25–27 g body weight) were obtained from Japan SLC, Inc.
(Hamamatsu, Japan), maintained on a standard moderate fat (MF) diet
(Oriental Yeast Co., Ltd., Osaka, Japan), and provided with water
ad libitum. All animal experiments were approved by the Kobe
Gakuin University Animal Committee (Kobe, Japan) according to the
university guidelines for the care and use of laboratory
animals.
Measurement of anti-anaphylactic
effects
The passive cutaneous anaphylaxis (PCA) reaction was
measured according to a previous study (14). Mice were sensitized by an
intradermal injection of 0.1 μg of anti-dinitrophenyl (DNP)-IgE in
the ear and intravenously challenged 4 h later with 0.2 ml (1
mg/ml) of a DNP-labeled human serum albumin solution containing 2%
Evans blue dye. In a series of experiments, a test compound (100
mg/kg) was administered orally 2 h prior to antigen challenge. The
animals in the control group received saline. The mice were
subsequently sacrificed and the ears removed and weighed 30 min
after the challenge. After dissolution of the ears in 200 μl of 1 N
potassium hydroxide (KOH), they were incubated overnight at 37°C
for the measurement of the amount of Evans blue dye present in the
exudates. For this purpose, the dissolved tissue solution was added
to 400 μl of a mixture of acetone and 0.6 N phosphoric acid (5/13,
v/v) and the optical density at 620 nm was measured. The amount of
dye in the exudates was calculated from an Evans blue standard
curve and the results expressed as a percentage of the mean exudate
dye amount in the ear samples of the treated test mice compared
with that of the controls.
Measurement of anti-inflammatory
activity
The mouse inflammatory test was performed according
to the method described in the study by Gschwendt et al
(16). Briefly, an acetone
solution containing a test compound at 500 μg/20 μl or a vehicle
control of 20 μl of acetone was applied to the inner section of a
mouse ear and, 30 min later, an acetone solution of
12-O-tetradecanoylphorbol-13-acetate (TPA, a chemical edema
inducer) at 0.5 μg/20 μl was applied to the same part of the ear;
acetone, followed by a TPA application, served as a control. After
7 h, a 6-mm diameter disk was obtained from the ear and weighed.
The anti-inflammatory activity was determined by comparing the ear
disk weight of the test group and the control group animals. The
results were expressed as a percentage and were derived using the
following formula: percentage activity = [(TPA only) − (tested
compound plus TPA)]/[(TPA only) − (vehicle)] ×100.
Statistical analysis
All the results are expressed as the means ±
standard deviation (SD) of at least 3 independent determinations
for each experiment. Statistical significance between each
experimental group was analyzed using the Student’s t-test, and a
probability value of 0.01 and 0.05 was used as the criterion of
significance.
Results
EO-induced inhibition of β-hexosaminidase
release in mast cells
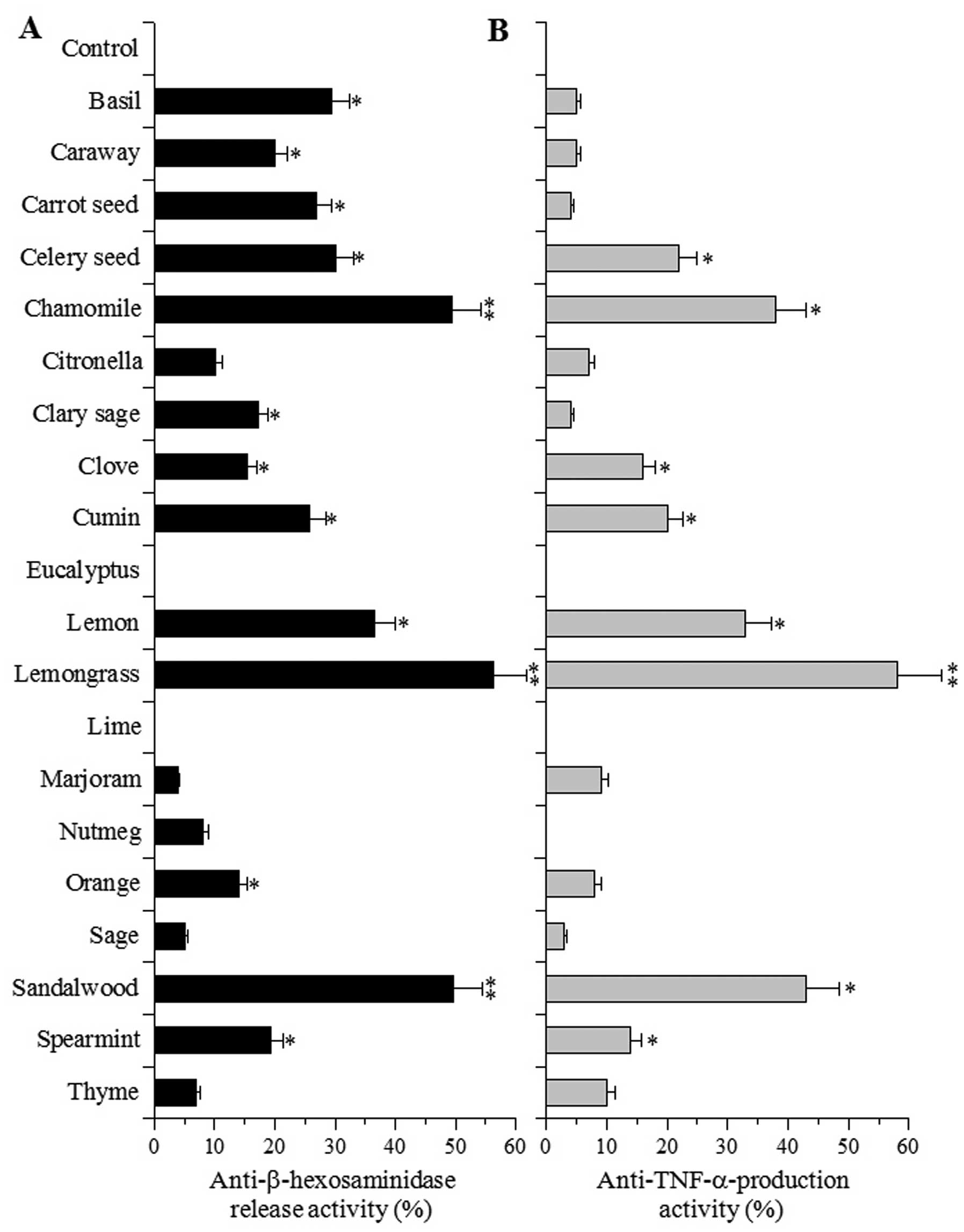
First, the inhibitory effects of 20 species of EOs
on mast cell degranulation were investigated. β-hexosaminidase
release is widely used as an indicator for evaluating the extent of
degranulation by mast cells and the corresponding release of
histamine and other chemical mediators, which is an important
process in initiating the immediate type of hypersensitive
reaction. The effects of the EOs (at 100 μg/ml) on the release of a
chemical mediator (β-hexosaminidase) was investigated in the
RBL-2H3 cells treated with the calcium ionophore, A23187. The EOs
did not influence the growth of RBL-2H3 cells and did not inhibit
β-hexosaminidase enzyme activity (data not shown). The degree of
degranulation was calculated by assessing the β-hexosaminidase
activity in the supernatant and cell lysate. Among the 20 EOs
examined, treatment with EOs from chamomile, lemongrass and
sandalwood produced >40% inhibition of mast cell degranulation,
as assessed by β-hexosaminidase release. Moreover, of the EOs
examined, lemongrass EO showed the strongest inhibitory activity
(Fig. 1A). On the other hand, EOs
from eucalyptus and lime did not influence β-hexosaminidase
release. A23187 is known to induce a calcium flux, which may
further induce protein kinase C (PKC) activation, a process
essential for mast cell degranulation (8). It is hence plausible that the
β-hexosaminidase inhibition induced by treatment with EOs may
affect PKC activity. The mechanisms through which EOs inhibit mast
cell degranulation require further investigation.
EO-induced inhibition of TNF-α production
in macrophages
We then investigated whether plant EOs, at 100
μg/ml, inhibit the production of TNF-α induced by LPS stimulation
in cultured cells. The inflammatory cytokine, TNF-α, activates the
NF-κB signaling pathway by binding to the TNF-α receptor, thereby
initiating an inflammatory response implicated in various
inflammatory diseases (17). In
cultured RAW264.7 murine macrophage cells, none of the compounds
examined showed cytotoxicity at 100 μg/ml (data not shown); the EOs
had no effect on the proliferation of peritoneal macrophages. The
RAW264.7 cells produced 693 pg/ml of TNF-α following stimulation
with LPS. The rate of TNF-α production in the controls was set at
0%. Among the EOs examined, those derived from lemongrass showed
the strongest effect on TNF-α production (>50% inhibition) and
those from chamomile, lemon and sandalwood displayed the second
strongest effects (30–50% inhibition) (Fig. 1B). EOs derived from eucalyptus,
lime and nutmeg failed to affect TNF-α production (0%
inhibition).
Association between the EO-induced
inhibition of β-hexosaminidase release by mast cells and TNF-α
production by macrophages
The possible association between the observed
inhibition of β-hexosaminidase in mast cells and TNF-α inhibition
in macrophages was confirmed by comparing the effects of the 20 EOs
on these biological activities. The inhibition of β-hexosaminidase
release showed a high correlation with TNF-α inhibition, with a
significant correlation coefficient (R2=0.783). These
results led us to speculate that the anti-allergic activity related
to the inhibition of β-hexosaminidase release and the
anti-inflammatory effects induced by the suppression of TNF-α may
be EO-related events within a shared biological pathway.
Among the 20 EOs examined, lemongrass EO produced
the strongest inhibitory activities in the cultured cells (Fig. 1); thereafter we focused on
lemongrass EO.
Identification of lemongrass EO
components
The association between the biological activity of
lemongrass EO and EO composition was clarified by an analysis of
the primary EO components by GC-MS. In total, 138 compounds were
detected in the EO, of which 40 components were identified, and the
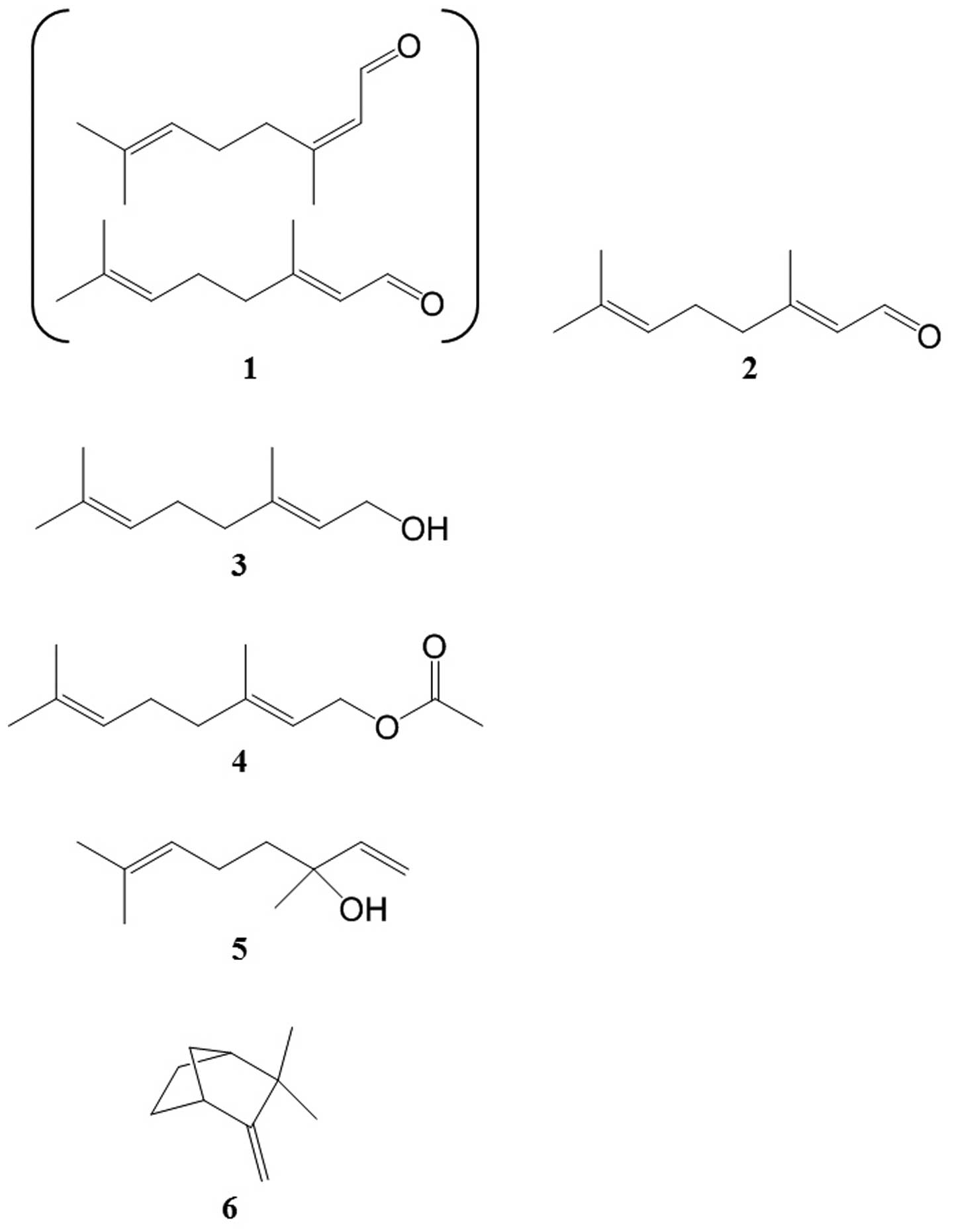
15 major compounds are listed in Table I. The 2 main compounds
representing >30% of the total content were geranial and neral
(40.16 and 34.24%, respectively). A mixture of these stereoisomic
monoterpene aldehydes is referred to as citral. Of the identified
components, 5 compounds, citral [1], geraniol [3], geranyl acetate
[4], linalool [5] and camphene [6], were commercially available.
Geranial [2] was chemically synthesized. Preparations of compounds
1–6 contain terpenoids of low molecular weight, and their
corresponding chemical structures are shown in Fig. 2.
 | Table IChemical composition of lemongrass
essential oil (EO). |
Table I
Chemical composition of lemongrass
essential oil (EO).
| Components | Composition
(%) |
|---|
| Geranial [1]
[2] | 40.16 |
| Neral [1] | 34.24 |
| Geraniol [3] | 5.11 |
| Geranyl acetate
[4] | 2.89 |
| Linalool [5] | 1.45 |
|
Methylheptenone | 1.44 |
| Camphene [6] | 1.39 |
| 4-Nonanone | 1.38 |
|
β-Caryophyllene | 1.09 |
| γ-Cadinene | 0.96 |
| Limonene | 0.33 |
| α-Pinene | 0.28 |
| Tricyclene | 0.18 |
| Acetone | 0.13 |
| 4-Heptanone | 0.06 |
| Others | 8.91 |
Effects of major lemongrass EO components
on β-hexosaminidase release and TNF-α production in cultured
cells
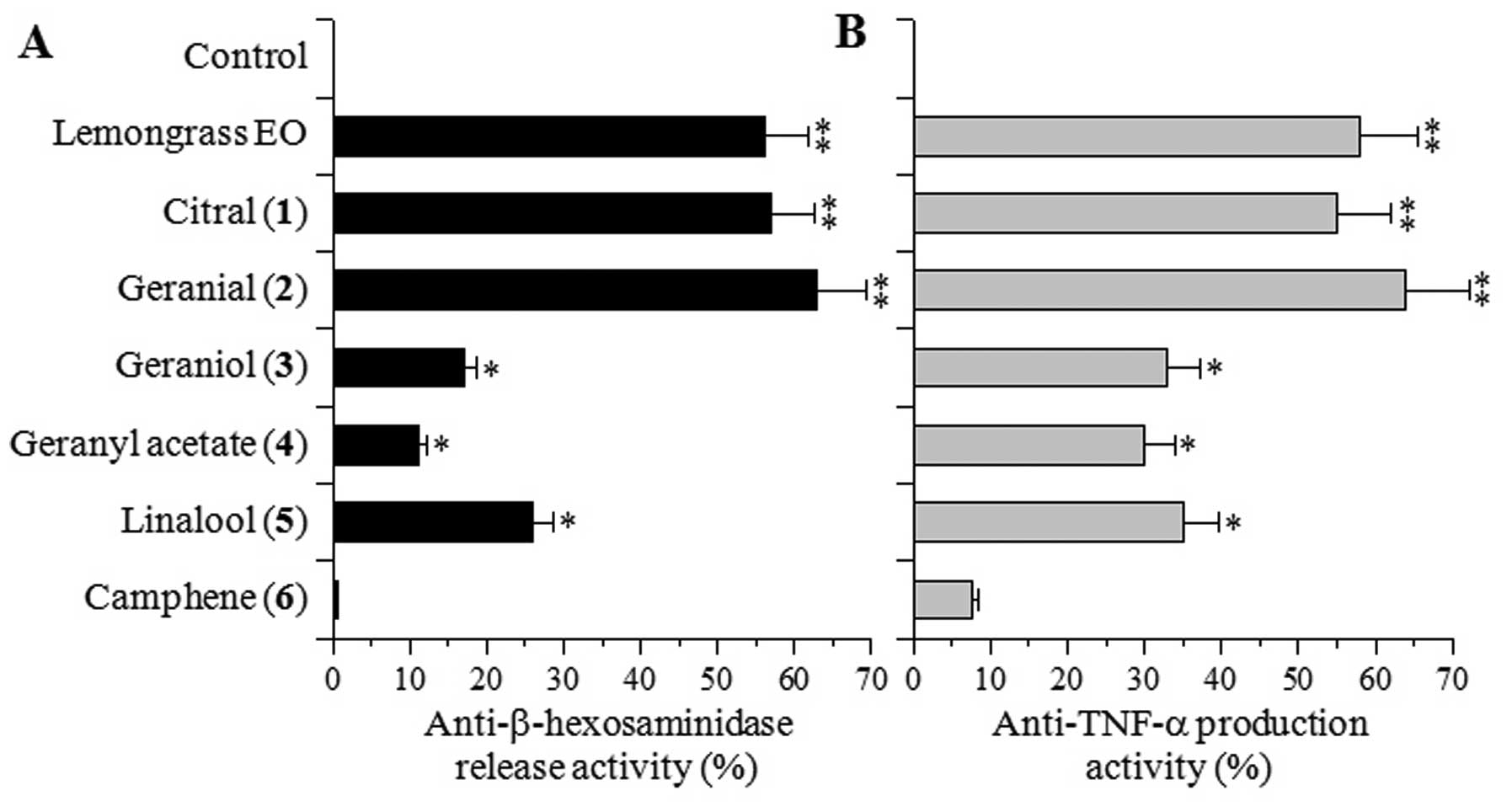
Based on the above results, the inhibitory effects
of 100 μg/ml of lemongrass EO and its major components, compounds
1–6, on β-hexosaminidase release by mast cells and TNF-α production
by macrophages were investigated. In the RBL-2H3 and RAW264.7
cells, these compounds did not show cytotoxicity at 100 μg/ml (data
not shown).
Citral [1] (a mixture of neral and geranial) and
geranial [2], which is a trans-isomer of citral, markedly
inhibited the release of β-hexosaminidase induced by the calcium
ionophore, A23187, and this inhibitory effect was similar to that
elicited by lemongrass EO (Fig.
3A). On the other hand, 4 other components, geraniol [3],
geranyl acetate [4], linalool [5] and camphene [6], exerted weak or
moderate effects, and the inhibitory effect on the release of
β-hexosaminidase elicited by compounds 3–6 was 2-fold weaker than
that elicited by compounds 1–2 or lemongrass EO.
The effects of compounds 1–6 on TNF-α production
were similar to those on β-hexosaminidase release, and compounds
1–2 and lemongrass EO exerted stronger inhibitory effects than
compounds 3–6 (Fig. 3B). These
results suggest that citral [1] and/or geranial [2] are the
bioactive components conferring the biological activities to
lemongrass EO. Therefore, we sought to explore compounds 1–2
further.
The inhibitory activity of citral [1] and
geranial [2] against LPS-induced inflammatory responses in cultured
macrophages
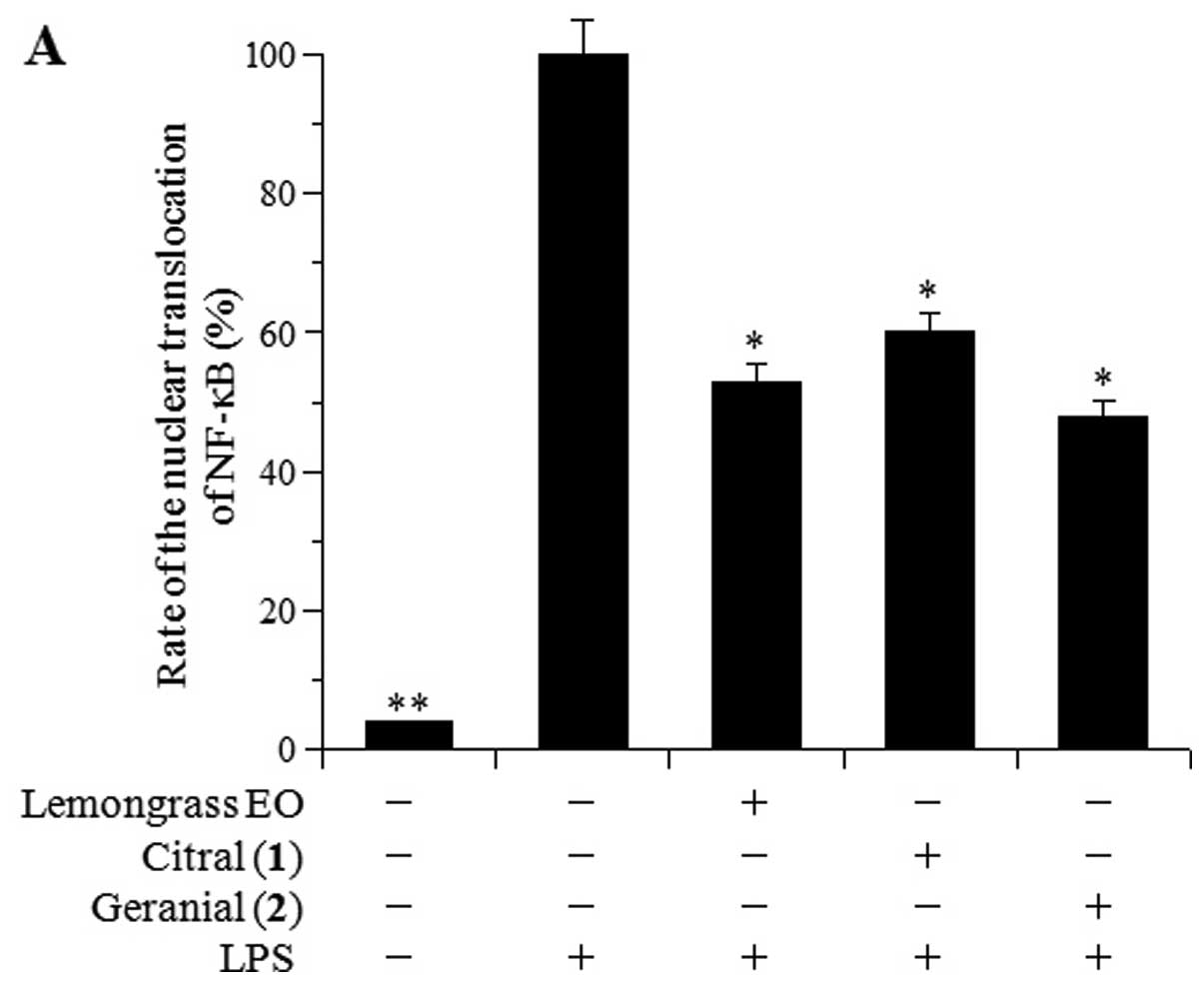
NF-κB activation is known to be the rate-controlling
factor during an inflammatory response. Therefore, the inhibitory
effects of lemongrass EO and its major components, citral [1] and
geranial [2], on the LPS-induced nuclear translocation of NF-κB
were examined in RAW264.7 cells (Fig.
4A). Western blot analysis revealed that 10 μg/ml of citral [1]
and geranial [2] inhibited the LPS-induced NF-κB nuclear
translocation, with 40 and 52% inhibition, respectively. Lemongrass
EO, at 10 μg/ml, also inhibited the nuclear translocation of NF-κB,
and this inhibitory activity was similar to that of compounds
1–2.
Stimulation with LPS results in the activation of
Toll-like receptor 4 and that of downstream IκB kinases (IKKs),
which in turn phosphorylate IκB, resulting in IκB degradation and
the translocation of NF-κB into the nucleus (18). IκB-α inhibits NF-κB by masking the
nuclear localization signals (NLS) of NF-κB proteins and
maintaining them in a sequestered and inactive state in the
cytoplasm (19). In addition,
IκB-α blocks the ability of the NF-κB transcription factor to bind
to DNA, which is required for the proper functioning of NF-κB
(20). Therefore, the effects of
lemongrass EO and compounds 1–2 on the LPS-induced decrease in the
protein expression of IκB-α were examined in the RAW264.7 cells
(Fig. 4B). IκB-α protein
expression was reduced by 59% following LPS stimulation (Fig. 4B). The amount of IκB-α protein in
the cells treated with compounds 1–2 (10 μg/ml each) was almost the
same as that in the LPS-treated cells (the IκB-α protein level was
approximately 35% compared to that in the control untreated cells),
suggesting that compounds 1–2 did not influence the protein
expression of IκB-α. These results demonstrate that lemongrass EO
and its major components suppress the NF-κB nuclear translocation
through an IκB-α-independent mechanism. The effects of these
compounds at other points in the NF-κB signaling pathway required
further investigation.
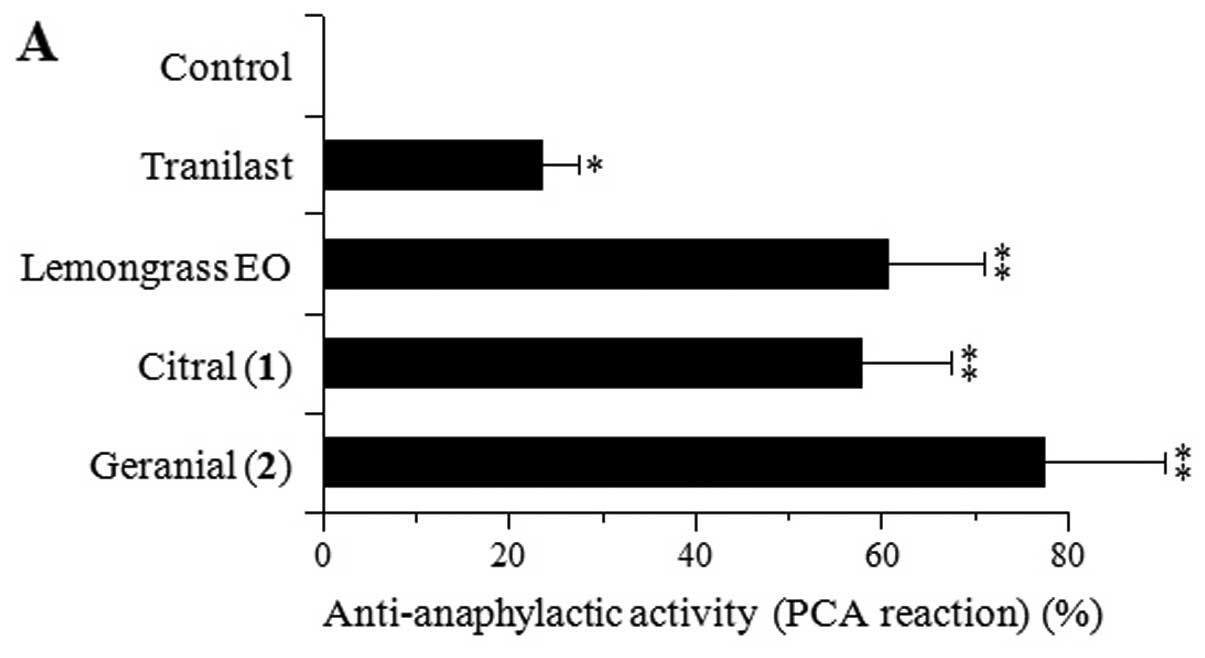
In vivo anti-allergic and
anti-inflammatory activities of citral [1] and geranial [2]
The IgE-mediated PCA reaction in vivo is a
method used for studying the mechanisms of the immediate
hypersensitivity reaction. As shown in Fig. 5A, 100 mg/kg of tranilast, a
commonly used anti-allergic drug that targets mast cell
degranulation and inhibits the PCA reaction, was used as a positive
control. Lemongrass EO and its major components, citral [1] and
geranial [2], at 100 mg/kg also inhibited the PCA reaction in the
mice by 60.7, 57.7 and 77.4%, respectively. The inhibitory effects
of lemongrass EO and compounds 1–2 were >2-fold stronger than
those of tranilast, suggesting a great potential for use as
anti-allergic compounds.
We examined the anti-inflammatory activity of
lemongrass EO, citral [1] and geranial [2] by examining their
effects on TPA-induced inflammatory edema of the mouse ear in
vivo. The application of TPA (0.5 μg) to the mouse ears induced
edema, resulting in a 241% increase in the weight of the ear disk 7
h after application. Pre-treatment with 500 μg/ear of these
compounds significantly suppressed inflammation, and the strength
of the inhibitory effect was ranked as follows: geranial [2] >
lemongrass EO > citral [1] (Fig.
5B). Therefore, the in vivo anti-inflammatory effects of
these compounds displayed the same order as their in vivo
anti-allergic effects (Fig. 5A).
These results suggest that the anti-allergic effects of the
components of lemongrass EO positively correlate with the
anti-inflammatory activity observed. Glycyrrhetinic acid (500
μg/ear), a known anti-inflammatory agent, had a 40.5%
anti-inflammatory effect in this assay system, and these compounds
were found to be approximately 1.5-fold more effective than this
agent.
The biological effects elicited by compounds 1–2
were similar to those produced by almost the same concentrations of
lemongrass EO (Figs. 3–5), suggesting that the anti-allergy and
anti-inflammatory effects of lemongrass EO are largely due to its 2
major components, citral [1] and/or geranial [2].
Discussion
Over the years, EOs have attracted much attention in
scientific research and are increasingly used in pharmaceutical,
nutritional and cosmetic industrial applications (21,22), primarily due to their potent
anti-microbial (23), antioxidant
(24), anticancer (25) and anti-inflammatory activities
(26) of EOs. The infection
process induces inflammation, during which inflammatory mediators,
such as cytokines are released from phagocytes (27). As various skin disorders,
including atopic dermatitis (28)
and acne vulgaris (29), are
associated with infection-induced inflammation, the presence of
anti-inflammatory agents may explain the effectiveness of certain
plant-derived EOs in the treatment of these syndromes. In several
regions worldwide, aromatic herbs play major roles in primary
health care, particularly in rural areas (30). Thus, an understanding of herbal
biological activities may further the effectiveness and the use of
many of their functional components, particularly as beneficial
additives in medical, nutritional and cosmetic products.
In the present study, 20 EOs were selected as known
herbal plant- and citrus fruit-derived EOs. Among the plant-derived
EOs worldwide, the compounds selected represent the most well known
and most commonly used materials in perfume preparations. These EOs
were isolated by hydrodistillation or cold-pressing and were
confirmed to be free of any contamination, such as heavy metals and
pesticides by GC-MS analyses (data not shown). Among these 20 EOs,
lemongrass EO exhibited the strongest inhibitory activity on mast
cell degranulation, which was assessed by quantifying
β-hexosaminidase release from RBL-2H3 cells treated with the
calcium ionophore, A23187 (Fig.
1A) and the production of TNF-α, a pleiotropic inflammatory
cytokine, by RAW264.7 murine macrophages treated with LPS (Fig. 1B). We observed that individual
constituents of lemongrass EO, including citral [1], which is a
mixture of stereoisomers, geranial (trans-isomer) [2] and
neral (cis-isomer), comprising 74.40% of lemongrass EO
(Table I), inhibited NF-κB
nuclear translocation by LPS, and suppressed TNF-α production in
macrophage cell culture experiments in vitro (Figs. 3–5). These compounds also exhibited potent
anti-allergic/anti-inflammatory activity in vivo. Lemongrass
EO and its major components, such as citral [1] and geranial [2]
exhibited anti-inflammatory activity in a TPA-induced mouse model
of ear edema, although the underlying molecular mechanisms remain
unelucidated (Fig. 5B). Since the
activation of NF-κB has been observed in a model of TPA-induced ear
edema (31), we hypothesized that
the anti-inflammatory effects may be, at least in part, dependent
on the inhibition of NF-κB activation. At present, citral [1] and
geranial [2] are regarded as promising anti-inflammatory agents.
Our study indicates that these compounds are useful as NF-κB
inhibitors and may be potent chemopreventive agents against
inflammation. Among the lemongrass EO components, citral [1] and
geranial [2] inhibited the release of β-hexosaminidase and the
production of TNF-α, while geraniol [3] and geranyl acetate [4] did
not have any effects (Fig. 3),
suggesting that the ketone group of citral may be important for
these observed inhibitory activities.
Lemongrass is native to India and tropical Asia. It
is widely used as an herb in Asian cuisine. It has a subtle citrus
flavor and can be dried and powdered, or used fresh. Lemongrass is
commonly used in teas, soups and curries. It is also suitable for
poultry, fish, beef and seafood. It is often used as a tea in
African countries, such as Togo and the Democratic Republic of
Congo, as well as in Latin American countries, such as Mexico.
Among the components of lemongrass, geranial [2] has a strong lemon
odor. The lemon odor of neral is less intense, but sweeter. Citral
[1], on the other hand, is an aromatic compound used in perfumery
for its citrus effect. Citral [1] is also used as a flavor and for
fortifying lemon oil. This plant containing citral [1] is used in
folk medicine as an anti-spasmodic, hypotensive, anti-convulsant,
analgesic, anti-emetic, antitussive, anti-rheumatic, antiseptic,
anti-bacterial, anti-diarrheal and antioxidant agent, as well as in
the treatment of nervous and gastrointestinal disorders and fevers
(32). In this study, lemongrass
EO was found to have anti-allergic and anti-inflammatory
activities; however, the mechanisms behind these different
bioactivities were not examined in detail. The association between
the above-mentioned biological activities and the blocking of the
nuclear translocation of NF-κB and TNF-α production requires
further investigation.
Modern aromatherapy for allergic and inflammatory
diseases has been developed primarily based on clinical trials of
EOs by several pioneers; however, scientific research on the
physiological role of these EOs against allergic and inflammatory
responses is still at a primitive stage. The elucidation of the
pharmacological functions of lemongrass EO in vivo may
provide a rationale for the clinical application of this EO as an
anti-allergic and anti-inflammatory substance. In conclusion, our
data provide strong scientific evidence and highlight the benefits
of lemongrass EO and its constituents, citral [1] and geranial [2],
for use in medical and/or cosmetic applications.
Acknowledgements
This study was supported in part by the Ministry of
Education, Culture, Sports, Science and Technology (MEXT,
Japan)-Supported Program for the Strategic Research Foundation at
Private Universities, 2012–2016. Y.M. received Grants-in-Aid for
Scientific Research (C) (no. 24580205) from MEXT and the Hyogo
Science and Technology Association (Japan). I.K. received a
Grant-in-Aid for Young Scientists (B) (no. 23710262) from MEXT.
Abbreviations:
|
EOs
|
essential oils
|
|
TNF-α
|
tumor necrosis factor-α
|
|
TPA
|
12-O-tetradecanoylphorbol-13-acetate
|
|
IgE
|
immunoglobulin E
|
|
LPS
|
lipopolysaccharide
|
|
GC
|
gas chromatography
|
|
MS
|
mass spectrometry
|
|
NF-κB
|
nuclear factor-κB
|
|
IκB
|
inhibitor of NF-κB
|
|
IgG
|
immunoglobulin G
|
|
FBS
|
fetal bovine serum
|
|
ELISA
|
enzyme-linked immunosorbent assay
|
|
SDS
|
sodium dodecyl sulfate
|
|
PCA
|
passive cutaneous anaphylaxis
|
|
DNP
|
dinitrophenyl
|
|
SD
|
standard deviation
|
|
PKC
|
protein kinase C
|
References
|
1
|
Bakkali F, Averbeck S, Averbeck D and
Idaomar M: Biological effects of essential oils-a review. Food Chem
Toxicol. 46:446–475. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Woollard AC, Tatham KC and Barker S: The
influence of essential oils on the processå of wound healing: a
review of the current evidence. J Wound Care. 16:255–257. 2007.
|
|
3
|
Prabuseenivasan S, Jayakumar M and
Ignacimuthu S: In vitro antibacterial activity of some plant
essential oils. BMC Complement Altern Med. 6:392006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Clarke JO and Mullin GE: A review of
complementary and alternative approaches to immunomodulation. Nutr
Clin Pract. 23:49–62. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wüthrich B: Epidemiology of the allergic
diseases: are they really on the increase? Int Arch Allergy Appl
Immunol. 90(Suppl 1): S3–S10. 1989.PubMed/NCBI
|
|
6
|
Stevens RL and Austen KF: Recent advances
in the cellular and molecular biology of mast cells. Immunol Today.
10:381–386. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Plaut M, Pierce JH, Watson CJ, Hanley-Hyde
J, Nordan RP and Paul WE: Mast cell lines produce lymphokines in
response to cross-linkage of Fc epsilon RI or to calcium
ionophores. Nature. 339:64–67. 1989. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gordon JR, Burd PR and Galli SJ: Mast
cells as a source of multifunctional cytokines. Immunol Today.
11:458–464. 1990.PubMed/NCBI
|
|
9
|
Ronis MJ, Butura A, Korourian S, Shankar
K, Simpson P, Badeaux J, Albano E, Ingelman-Sundberg M and Badger
TM: Cytokine and chemokine expression associated with
steatohepatitis and hepatocyte proliferation in rats fed ethanol
via total enteral nutrition. Exp Biol Med (Maywood). 233:344–355.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pierce GF: Macrophages: important
physiologic and pathologic sources of polypeptide growth factors.
Am J Respir Cell Mol Biol. 2:233–234. 1990.PubMed/NCBI
|
|
11
|
Boscá L, Zeini M, Través PG and Hortelano
S: Nitric oxide and cell viability in inflammatory cells: a role
for NO in macrophage function and fate. Toxicology. 208:249–258.
2005.PubMed/NCBI
|
|
12
|
Hirano M, Yakabe S, Chikamori H, Clark JH
and Morimoto T: Oxidation by chemical manganese dioxide. Part 3.
Oxidation of benzylic and allylic alcohols, hydroxyarenes and
aminoarenes. J Chem Res (S). 770–771. 1998. View Article : Google Scholar
|
|
13
|
Razin E, Mencia-Huerta JM, Stevens RL,
Lewis RA, Liu FT, Corey E and Austen KF: IgE-mediated release of
leukotriene C4, chondroitin sulfate E proteoglycan,
beta-hexosaminidase, and histamine from cultured bone
marrow-derived mouse mast cells. J Exp Med. 157:189–201. 1983.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sato H, Kobayashi Y, Hattori A, Suzuki T,
Shigekawa M and Jippo T: Inhibitory effects of water-soluble
low-molecular-weight β-(1,3-1,6) D-glucan isolated from
Aureobasidium pullulans 1A1 strain black yeast on mast cell
degranulation and passive cutaneous anaphylaxis. Biosci Biotechnol
Biochem. 76:84–88. 2012.
|
|
15
|
Nishiumi S, Yamamoto N, Kodoi R, Fukuda I,
Yoshida K and Ashida H: Antagonistic and agonistic effects of
indigoids on the transformation of an aryl hydrocarbon receptor.
Arch Biochem Biophys. 470:187–199. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gschwendt M, Kittstein W, Fürstenberger G
and Marks F: The mouse ear edema: a quantitatively evaluable assay
for tumor promoting compounds and for inhibitors of tumor
promotion. Cancer Lett. 25:177–185. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Aggarwal BB: Signalling pathways of the
TNF superfamily: a double-edged sword. Nat Rev Immunol. 3:745–756.
2003. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hashimoto T, Nonaka Y, Minato K, Kawakami
S, Mizuno M, Fukuda I, Kanazawa K and Ashida H: Suppressive effect
of polysaccharides from the edible and medicinal mushrooms,
Lentinus edodes and Agaricus blazei, on the
expression of cytochrome P450s in mice. Biosci Biotechnol Biochem.
66:1610–1614. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jacobs MD and Harrison SC: Structure of an
IkappaBalpha/NF-kappaB complex. Cell. 95:749–758. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Verma IM, Stevenson JK, Schwarz EM, Van
Antwerp D and Miyamoto S: Rel/NF-kappa B/I kappa B family: intimate
tales of association and dissociation. Genes Dev. 9:2723–2735.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cheng SS, Lin HY and Chang ST: Chemical
composition and antifungal activity of essential oils from
different tissues of Japanese Cedar (Cryptomeria japonica).
J Agric Food Chem. 53:614–619. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ksouri R, Falleh H, Megdiche W, Trabelsi
N, Mhamdi B, Chaieb K, Bakrouf A, Magné C and Abdelly C:
Antioxidant and antimicrobial activities of the edible medicinal
halophyte Tamarix gallica L. and related polyphenolic
constituents. Food Chem Toxicol. 47:2083–2091. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lo Cantore P, Shanmugaiah V and Iacobellis
NS: Antibacterial activity of essential oil components and their
potential use in seed disinfection. J Agric Food Chem.
57:9454–9461. 2009.PubMed/NCBI
|
|
24
|
Dutra RC, Leite MN and Barbosa NR:
Quantification of phenolic constituents and antioxidant activity of
Pterodon emarginatus vogel seeds. Int J Mol Sci. 9:606–614.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kumar A, Malik F, Bhushan S, Sethi VK,
Shahi AK, Kaur J, Taneja SC, Qazi GN and Singh J: An essential oil
and its major constituent isointermedeol induce apoptosis by
increased expression of mitochondrial cytochrome c and apical death
receptors in human leukaemia HL-60 cells. Chem Biol Interact.
171:332–347. 2008. View Article : Google Scholar
|
|
26
|
Chao LK, Hua KF, Hsu HY, Cheng SS, Liu JY
and Chang ST: Study on the antiinflammatory activity of essential
oil from leaves of Cinnamomum osmophloeum. J Agric Food
Chem. 53:7274–7278. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lourens AC, Reddy D, Başer KH, Viljoen AM
and Van Vuuren SF: In vitro biological activity and essential oil
composition of four indigenous South African Helichrysum
species. J Ethnopharmacol. 95:253–258. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tsukahara H, Shibata R, Ohshima Y,
Todoroki Y, Sato S, Ohta N, Hiraoka M, Yoshida A, Nishima S and
Mayumi M: Oxidative stress and altered antioxidant defenses in
children with acute exacerbation of atopic dermatitis. Life Sci.
72:2509–2516. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Grange PA, Chéreau C, Raingeaud J, Nicco
C, Weill B, Dupin N and Batteux F: Production of superoxide anions
by keratinocytes initiates P. acnes-induced inflammation of
the skin. PLoS Pathog. 5:e10005272009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kamatou GP, Viljoen AM, Gono-Bwalya AB,
van Zyl RL, van Vuuren SF, Lourens AC, Başer KH, Demirci B, Lindsey
KL, van Staden J and Steenkamp P: The in vitro pharmacological
activities and a chemical investigation of three South African
Salvia species. J Ethnopharmacol. 102:382–390. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Medeiros R, Otuki MF, Avellar MC and
Calixto JB: Mechanisms underlying the inhibitory actions of the
pentacyclic triterpene alpha-amyrin in the mouse skin inflammation
induced by phorbol ester
12-O-tetradecanoylphorbol-13-acetate. Eur J Pharmacol.
559:227–235. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shah G, Shri R, Panchal V, Sharma N, Singh
B and Mann AS: Scientific basis for the therapeutic use of
Cymbopogon citratus, stapf (Lemon grass). J Adv Pharm
Technol Res. 2:3–8. 2011. View Article : Google Scholar
|