Introduction
Idiopathic pulmonary fibrosis (IPF) is part of a
large group of diffuse parenchymal lung diseases, that are
characterized by progressive parenchymal fibrosis, without any
known cause and poor prognosis. Nogee et al conducted a
genetic analysis of a mother (onset age, 1 year) with desquamative
interstitial pneumonia (DIP) and a child (onset age, 6 weeks) with
non-specific interstitial pneumonia (NSIP), and identified a
heterozygous mutation of the surfactant protein C (SP-C) gene
(deletion of exon 4, SP-CΔexon4) for the first time
(1). SP-C is a component of the
pulmonary surfactant, which is a heterogeneous complex of lipids
and proteins that lowers lung-surface tension and contributes to
the host defense system. The SP-C gene is located on the short arm
of chromosome 8, and comprises 6 exons and 5 introns (2). The precursor of SP-C, proSP-C,
consists of 197 amino acids and is transformed into mature SP-C
(consisting of 24–58 amino acids) through intracellular cleavage
and modification (3,4). ProSP-CΔexon4 has been
found to induce endoplasmic reticulum (ER) stress (5,6).
Fibrosis is defined by the accumulation of excess
extracellular matrix (ECM) components, such as collagen, in and
around inflamed or damaged tissue (7,8).
Fibrosis can develop in the majority of tissue types, and advanced
fibrosis causes fibrotic diseases, such as pulmonary fibrosis,
liver cirrhosis and renal fibrosis, resulting in organ dysfunction
and eventually death. According to recent studies, synoviolin/HRD1
is involved in liver fibrosis (9)
and collagen synthesis in the kidneys (10). Synoviolin, which we have
previously cloned (11), is an
ER-resident E3 ubiquitin ligase with a RING finger domain that
activates the ER-associated degradation (ERAD) pathway (11). Synoviolin is ubiquitously
expressed in all organs of the body, particularly in the liver,
pancreas and skeletal muscle (12). Synoviolin is upregulated in
response to ER stress and the overexpression of human
inositol-requiring kinase 1 (IRE1) and activating transcription
factor 6 (ATF6) (12,13). We have previously demonstrated
that heterozygous synoviolin knockout mice are resistant to
CCl4-induced liver fibrosis. In addition, we have
demonstrated that collagen secretion is reduced by synoviolin
deficiency in mouse embryonic fibroblasts, suggesting the
involvement of synoviolin in collagen secretion (9). Li et al (10) demonstrated that synoviolin
expression was increased in a mouse model of renal interstitial
fibrosis caused by unilateral ureteral obstruction (UUO). In
addition, they demonstrated that collagen secretion is reduced by
synoviolin deletion in NRK-49F cells (normal rat kidney
interstitial fibroblasts) (10).
These data suggest that synoviolin expression is increased by ER
stress and that synoviolin promotes collagen synthesis. Thus, we
hypothesized that synoviolin is involved in lung fibrosis, and in
particular, in ER stress-related lung fibrosis. In order to confirm
our hypothesis, in the present study, we investigated the
involvement of synoviolin and synoviolin inhibitor, LS-102, in
collagen secretion in an in vitro model of IPF by
transfecting A549 cells (a human lung adenocarcinoma epithelial
cell line) with SP-C lacking exon 4.
To investigate our hypothesis that synoviolin is
involved in lung fibrosis and that synoviolin inhibitors are
candidate antifibrotic agents, we investigated the association
between synoviolin and collagen secretion in an in vitro
model, in which A549 cells were transfected with
SP-CΔexon4, which has been reported to induce ER stress
(5,6). In the present study, we did not use
any chemical or physical method to induce fibrosis; instead, we
focused on an SP-C mutation, SP-CΔexon4, which has been
identified in parent-child cases of familial interstitial pneumonia
(1).
Materials and methods
Cell culture
The A549 human lung adenocarcinoma cell line was
purchased from the American Type Culture Collection (ATCC,
Manassas, VA, USA). The A549 cells were cultured in minimum
essential medium (Sigma-Aldrich, St. Louis, MO, USA) supplemented
with 1% penicillin, 1% streptomycin and 10% (heat-inactivated)
fetal bovine serum (FBS).
Transfection of cells with plasmid
constructs and siRNA
The wild-type proSP-C expression vector was
constructed using pcDNA3-FLAG (Invitrogen, Carlsbad, CA, USA) by
the insertion of wild-type proSP-C cDNA that was isolated from a
human lung cDNA library (Takara Bio Co., Tokyo, Japan).
SP-CΔexon4 (deletion of exon 4) was
constructed according to previously published techniques (14). All constructs were confirmed by
DNA sequencing. The A549 cells were grown for 24 h and transfected
with wild-type SP-C or mutant SP-C constructs using Lipofectamine
2000 (Invitrogen) as per the manufacturer’s instructions.
The sequences of the synoviolin siRNA and scrambled
synoviolin siRNA were identical to those described in a previous
study (15), and were chemically
synthesized at Hokkaido System Science (Hokkaido, Japan). The A549
cells were seeded at 40–50% confluency in 6-well plates and
cultured for 24 h. Subsequently, 50 nmol of annealed RNA duplex was
transfected using Lipofectamine 2000 according to the
manufacturer’s instructions.
Antibodies
The antibodies used in this study were as follows:
anti-synoviolin/HRD1 monoclonal antibody as previously described
(11), anti-FLAG antibody
(anti-FLAG M2 monoclonal antibody, F3165; Sigma-Aldrich),
anti-hemagglutinin-A (HA) 3F10 monoclonal antibody (1867423; Roche
Applied Science) and anti-β-actin monoclonal antibody (A5441;
Sigma-Aldrich).
Preparation of cellular protein
Cell cultures were harvested and proteins were
extracted using a cell extraction buffer containing 0.05 M Tris-HCl
(pH 8.0), 0.15 M NaCl, 5.0 mM ethylenediaminetetraacetic acid
(EDTA), 1% NP-40 and protease inhibitors (1 μg/ml leupeptin, 1
μg/ml dithiothreitol (DTT), 1 μg/ml pepstatin and 1.5 μg/ml
aprotinin) at 4°C.
Reporter assay
The A549 cells were plated on 24-well plates
(5×104 cells/well). The synoviolin promoter
(SyG-2055/+845 PGV-B2) was prepared as described in a previous
study (16). For transient
transfection into the A549 cells, 20 ng/well synoviolin promoter
and 50 ng/well test plasmids (SP-C1–197 or
SP-CΔexon4) were co-transfected using Lipofectamine 2000
(Invitrogen) according to the manufacturer’s recommendations. After
24 h, the cultures were aspirated and the cells were added to 100
μl of passive lysis buffer (Promega, Madison, WI, USA). The cell
debris was pelleted and 5 μl of supernatant were collected and
immediately analyzed for luciferase activity using the Promega
Luciferase assay system (Promega) and a microplate luminometer
(Centro XS3 LB 960; Berthold Technologies, Wildbad, Germany). The 5
μl supernatant samples were used to measure the protein
concentration in each well using a multi-spectrophotometer (Viento
XS; Dainippon Sumitomo Pharma, Tokyo, Japan). The assay was
performed in triplicate and the mean values were calculated for
each sample. The luciferase activity results were normalized to the
protein concentration in each sample.
Western blot analysis
The A549 cells were seeded at 40–50% confluency in
100-mm plates and harvested 18 h following transfection.
Tunicamycin-treated (10 μg/ml) A549 cells were prepared as a model
of chemical induction of ER stress. Cellular protein extracts were
resolved by 10% SDS-PAGE, transferred onto a nitrocellulose
membrane and immunoblotted with anti-synoviolin/HRD1 monoclonal
antibodies followed by horseradish peroxidase-conjugated secondary
antibodies. The antigen-antibody complexes were visualized using an
ECL detection system (Promega). β-actin expression was used as an
internal control. Immunoblots were scanned and band densities were
quantified using ImageJ software.
Real-time polymerase chain reaction
(PCR)
The A549 cells were grown in 100-mm culture plates.
Forty-eight hours following transfection, total RNA from the A549
cells was isolated using Isogen II (Nippon Gene, Tokyo, Japan) and
cDNA was synthesized using RiverTra Ace (Toyobo, Tokyo, Japan)
according to the manufacturer’s instructions. Real-time PCR
relative quantification analysis was performed to measure the mRNA
expression levels of transforming growth factor-β (TGF-β),
connective tissue growth factor (CTGF), prolyl 4-hydroxylase
subunit alpha 1 (P4HA1), X-box binding protein 1 (XBP1) and
synoviolin using the FastStart Universal Probe Master mix (ROX;
Roche Applied Science, Indianapolis, IN, USA), primers from the
Universal Probe Library Assay Design Center and probes from the
universal probe library set (both from Roche Applied Science,
Tokyo, Japan) with the Step One Plus Detection System and software
(Applied Biosystems, Life Technologies Japan, Tokyo, Japan). The
primers and probes used are as follows: TGF-β forward,
GCAGCACGTGGAGCTGTA and reverse, CAGCCGGTTGC TGAGGTA (probe:
Universal probe library probe #72); CTGF forward,
AGCTGACCTGGAAGAGAACATT and reverse, GCTCGGTATGTCTTCATGCTG (probe:
Universal probe library probe #71); P4HA1 forward, TCGTCAAAGACCTAG
CAAAACC and reverse, CCGTCTCCAAGTCTCCTGTTA (probe: Universal probe
library probe #30); XBP1 forward, GGAGTTAAGACAGCGCTTGG and reverse,
CACTGG CCTCACTTCATTCC (probe: Universal probe library probe #37);
synoviolin forward, TTCGTCAGCCACGCCTAT and reverse,
GAGCACCATCGTCATCAGG (probe: Universal probe library probe #89). The
protocol included 1 cycle at 95°C for 10 min, followed by 40 cycles
at 95°C for 1 sec and 60°C for 20 sec, according to the
manufacturer’s recommendations. The mRNA level was normalized
relative to the amount of the transcript coding for 18S rRNA.
Measurement of soluble secreted
collagen
The A549 cells were seeded at 40–50% confluency in
6-well plates. Twelve hours following transfection, the medium was
replaced with serum-free Opti-MEM (Gibco, Life Technologies Japan,
Tokyo, Japan) to eliminate the effects of serum on collagen
measurement. At 30 h following transfection, the total soluble
collagen in the culture supernatants was measured using the Sircol
collagen assay method (Biocolor, County Antrim, UK) according to
the manufacturer’s instructions. The assay was performed in
triplicate and the mean values of each sample were calculated.
Immunoprecipitation
The cells were plated on 100-mm tissue culture
dishes and MG132 was added at a concentration of 20 μM 24 h
following transfection. The cells were co-transfected with
hemagglutinin (HA)-tagged ubiquitin (Ub) and an SP-C wild-type,
SP-C mutant, or empty vector. Co-transfected with the synoviolin
gene were also prepared. Twenty-seven hours following transfection,
the cells were harvested and lysed with 200 μl of lysis buffer [50
mM HEPES-KOH (pH 7.9), 150 mM KCl, 1% Triton X-100, 1 mM
phenylmethylsulfonyl fluoride (PMSF), 10% glycerol, 2 μg/ml
aprotinin, 2 μg/ml leupeptin, 2 μg/ml pepstatin A and 20 μM MG132
(a proteasome inhibitor)]. Following centrifugation at 5,000 rpm
for 10 min, 150 μl of the clear supernatant was incubated with 5 μl
of normal mouse IgG (Santa Cruz Biotechnology, Inc., Santa Cruz,
CA, USA), 30 μl of protein G-Sepharose (GE Healthcare, Tokyo,
Japan) and 350 μl of lysis buffer at 4°C with rotation for 30 min.
The supernatant was incubated with 2 μl of anti-FLAG M2 monoclonal
antibody at 4°C with rotation for 2 h, after which 40 μl of protein
G were added, followed by incubation at 4°C with rotation for 1 h.
The immunoprecipitates were solubilized in SDS sample buffer [0.078
M Tris-HCl (pH 6.8), 2% SDS, 10% glycerol, 5% 2-mercaptoethanol and
0.003% bromophenol blue] and then subjected to 4–15% gradient
SDS-PAGE (Bio-Rad, Hercules, CA, USA).
Synoviolin inhibitor, LS-102
Synoviolin inhibitor, LS-102, was identified by
screening approximately 4 million compounds for potential
synoviolin enzyme inhibitors as described in a previous study
(5). Dimethyl sulfoxide (DMSO) is
used as solvent for LS-102; thus, we used DMSO as a control. Twelve
hours following transfection, Opti-MEM (no serum added) was
replaced, and LS-102 (5 μM) or DMSO (5 μM) was added to the medium
to evaluate the effect of LS-102.
Statistical analysis
All data are expressed as the means ± standard
deviation (SD). Differences between groups were examined using the
Student’s t-test and considered statistically significant at
P<0.05. All results were derived from at least 3 independent
experiments.
Results
Overexpression of SP-CΔexon4
promotes collagen secretion in A549 cells
Studies have found an association between mutations
in the SP-C gene and familial pulmonary fibrosis (1,17,18). Therefore, in this study we focused
on the SP-CΔexon4 mutation. To assess the association
between the SP-CΔexon4 mutation and fibrosis in the
current experimental system, we measured the mRNA expression levels
of transforming growth factor-β (TGF-β), connective tissue growth
factor (CTGF) and prolyl 4-hydroxylase subunit alpha 1 (P4HA1),
which are responsible for enhancing the production of collagen
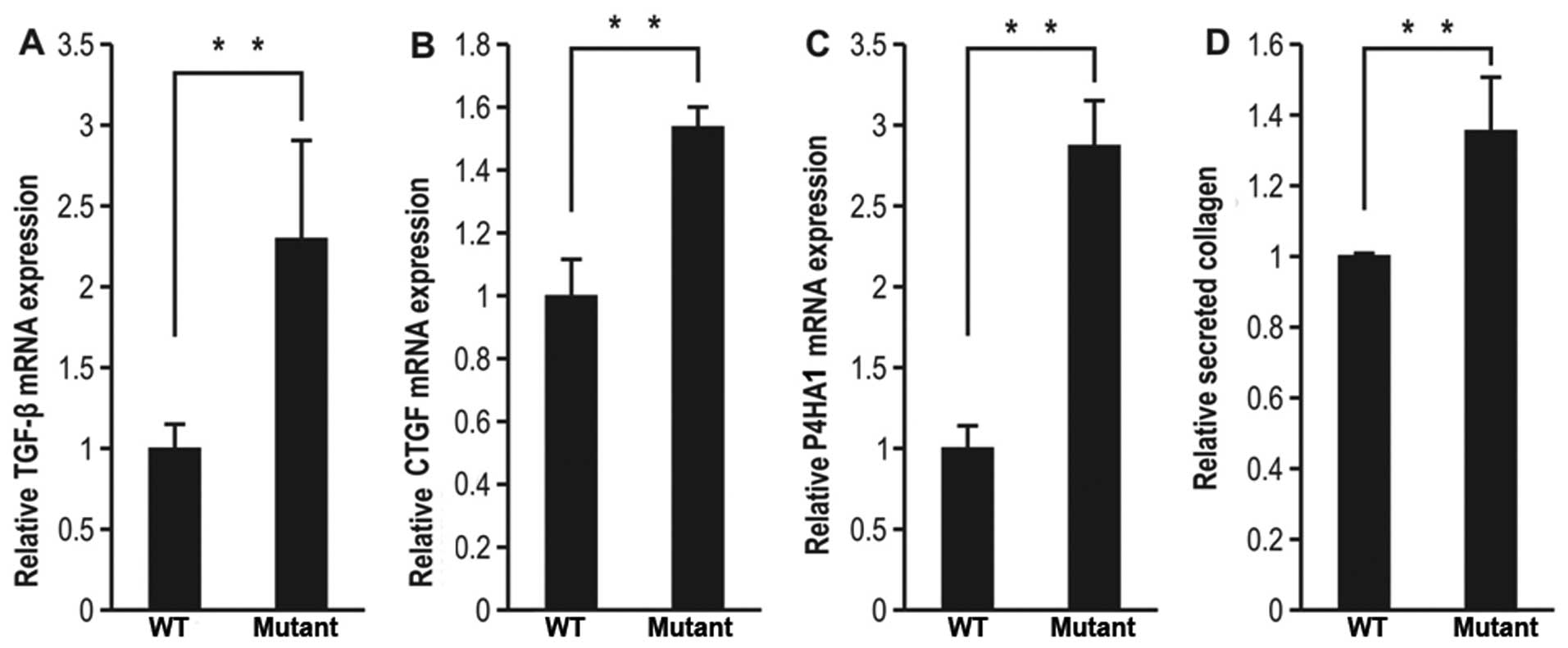
protein, a typical ECM component involved in fibrosis (19–21). Transfection of the cells with the
mutant SP-C gene increased the mRNA expression levels of TGF-β,
CTGF and P4HA1 by approximately 2.3-, 1.5- and 2.9-fold,
respectively, compared to the wild-type (Fig. 1A–C).
Fibrosis is attributed to the excess deposition of
ECM components in and around inflamed or damaged tissue (8). Collagen is a major component of the
ECM; therefore, we measured collagen secretion following
transfection of the cells with mutant SP-C. To confirm that mutant
SP-C promoted collagen secretion from mutant SP-C-expressing A549
cells, collagen secretion was measured following transfection with
a wild-type or mutant SP-C gene. The A549 cells transfected with
the mutant SP-C gene secreted approximately 1.3-fold more collagen
into the medium than the cells transfected with wild-type SP-C
(Fig. 1D).
SP-CΔexon4 increases the
expression of synoviolin
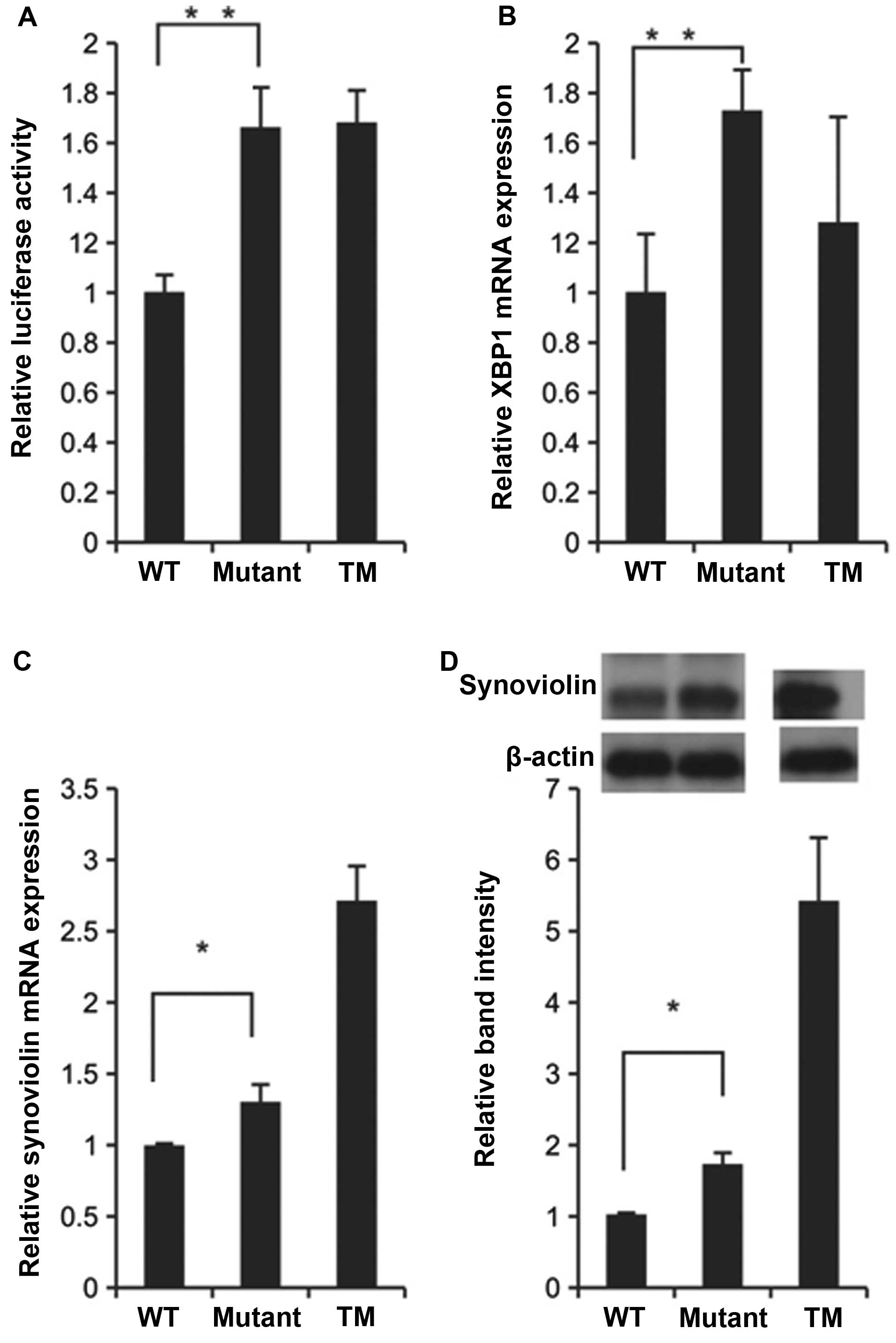
To examine the hypothesis that synoviolin plays a
role in fibrosis induced by SP-CΔexon4, we evaluated the
level of synoviolin expression in SP-CΔexon4-expressing
A549 cells. First, we constructed a plasmid carrying a luciferase
gene with expression regulated by a synoviolin promoter, which was
co-transfected together with the wild-type SP-C or mutant SP-C gene
expression plasmid into the A549 cells. Twenty-four hours following
transfection, a luciferase assay was conducted. The A549 cells
expressing mutant SP-C showed 1.6-fold greater luciferase activity
than the wild-type SP-C-expressing cells (Fig. 2A). These data suggest that the
transcriptional activity of synoviolin was enhanced in the
SP-CΔexon4 gene-transfected cells.
Subsequently, to confirm that ER stress is induced
in this experimental system and that the mRNA expression of
synoviolin is activated in the mutant SP-C-transfected cells, we
examined the expression levels of X-box binding protein 1 (XBP1)
and synoviolin in the A549 cells by real-time PCR. The mRNA
expression levels of XBP1 and synoviolin were increased in the
mutant SP-C-expressing A549 cells in comparison to the wild-type
SP-C-expressing cells (Fig. 2B and
C).
Finally, to assess the protein expression of
synoviolin, we performed western blot analysis with an
anti-synoviolin antibody. Transfection of the A549 cells with
mutant SP-C increased the protein expression of synoviolin compared
with the cells transfected with wild-type SP-C (Fig. 2D). Thus, the overexpression of
SP-CΔexon4 in the A549 cells increased the
transcriptional activity of synoviolin and the expression of
synoviolin at the mRNA and protein levels.
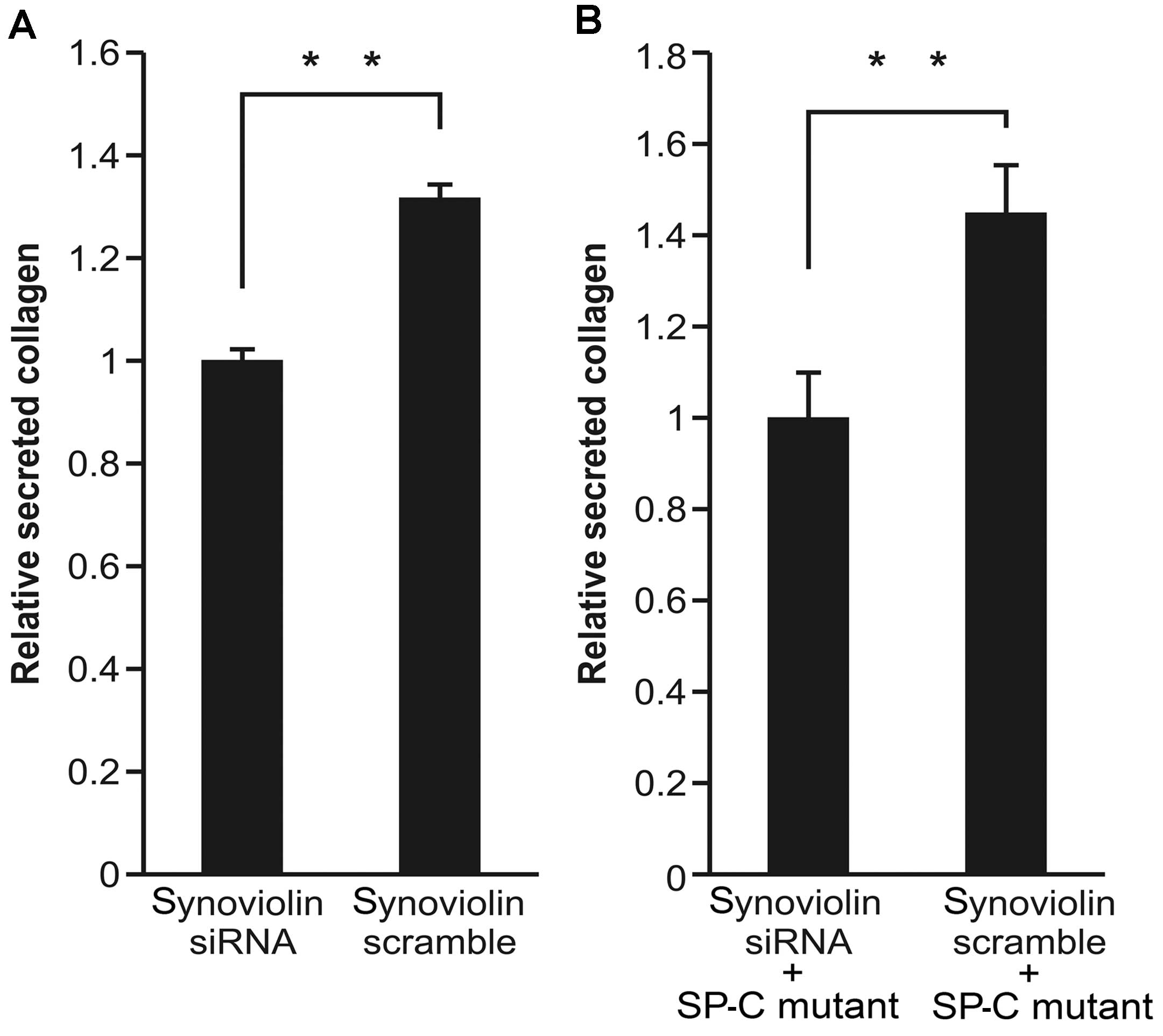
Synoviolin knockdown decreases collagen
secretion in A549 cells transfected with SP-CΔexon4
To explore the role of synoviolin in collagen
secretion in the A549 cells, we evaluated collagen production in
A549 cells following transfection with siRNA targeting synoviolin.
We found that the level of soluble secreted collagen in the
supernatant of A549 cells after the knockdown of synoviolin was
approximately 20% less than that produced by the scrambled
siRNA-transfected A549 cells (Fig.
3A). Furthermore, we analyzed the effects of synoviolin siRNA
on collagen secretion in SP-CΔexon4-expressing A549
cells. The A549 cells were co-transfected with
SP-CΔexon4 and either synoviolin siRNA or scrambled
siRNA. The amount of soluble secreted collagen in the supernatant
of the synoviolin siRNA-transfected
SP-CΔexon4-expressing A549 cells was approximately 30%
less than that produced by scrambled siRNA-transfected
SP-CΔexon4-expressing A549 cells (Fig. 3B).
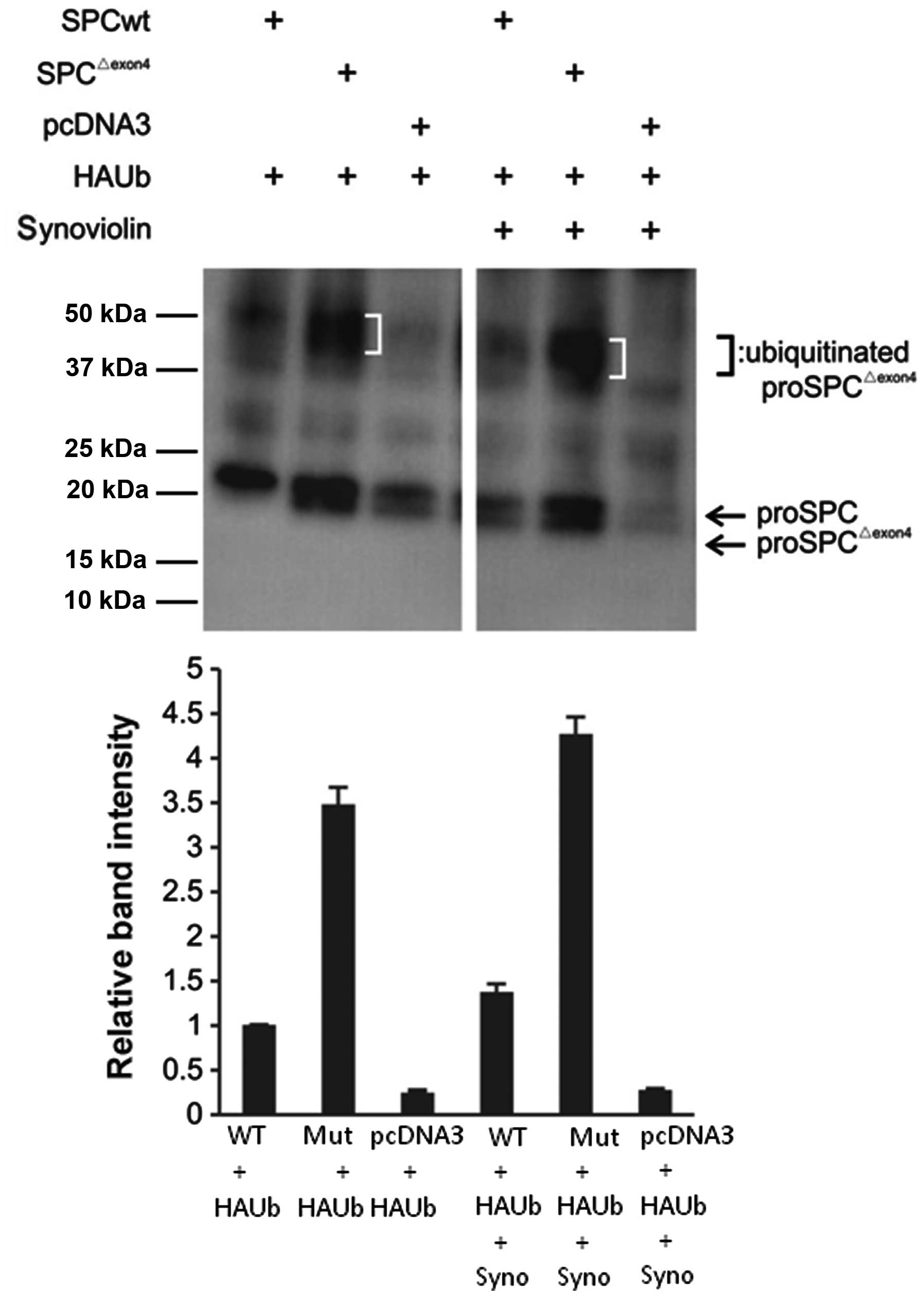
Synoviolin ubiquitinates
proSP-CΔexon4 as a substrate
To demonstrate the ubiquitination of
proSP-CΔexon4, the A549 cells were co-transfected with
hemagglutinin (HA)-tagged ubiquitin (Ub) and an SP-C wild-type,
SP-C mutant, or empty vector. In addition, to determine whether
synoviolin ubiquitinates proSP-CΔexon4 as a substrate,
cells co-transfected with the synoviolin gene were also prepared.
Following immunoprecipitation with an anti-FLAG antibody, western
blot analysis was performed using an anti-HA antibody. The cells
transfected with SP-CΔexon4 showed a ladder pattern,
suggesting ubiquitination. The synoviolin-containing cells showed a
more intense band (Fig. 4). These
results suggested that the proSP-CΔexon4 protein was
ubiquitinated in this experimental system and that synoviolin may
participate in this ubiquitination.
Collagen secretion is suppressed by the
synoviolin inhibitor, LS-102
To investigate the effects of synoviolin inhibition
on collagen secretion in A549 cells, we performed experiments using
the synoviolin inhibitor, LS-102, as described in a previous study
of ours (22), in which we
identified LS-102 by screening approximately 4 million compounds
for potential synoviolin enzyme inhibitors. As a result, LS-101 and
LS-102 were identified as compounds that inhibited the
autoubiquitination activity of synoviolin. These compounds were
administered in a mouse model of collagen-induced arthritis, in
which they demonstrated protective effects against arthritis.
LS-101 had more potent inhibitory effects on the synoviolin enzyme
than LS-102. However, LS-102 was used in this study as it is more
selective for synoviolin than LS-101. Twelve hours following
transfection, Opti-MEM (no serum added) was replaced, and LS-102 or
DMSO (5 μM; as a control) was added to the medium. The tunicamycin
(10 μg/ml)-treated A549 cells were also prepared as a positive
control for ER stress induction. LS-102 significantly decreased
collagen secretion into the medium (Fig. 5).
Discussion
In the present study, we demonstrated that the
expression of synoviolin and collagen secretion were enhanced by
the overexpression of SP-CΔexon4 in A549 cells and that
collagen secretion was suppressed by the use of the synoviolin
inhibitor, LS-102.
The pathogenic mechanisms of fibrosis have recently
attracted the attention of researches, and studies have indicated
that diverse factors, pathways and systems are involved in the
progression of fibrosis. However, the detailed mechanisms involved
are not yet fully understood. Wynn et al (8), Tanjore et al (23,24) and Zhong et al (25) reported that various pathways are
involved in fibrosis, including the ER-stress pathway, and that ER
stress leads to fibrosis. In the present study, we did not use any
chemical or physical method to induce fibrosis, but instead we
focused on a mutation in SP-C that was identified in mother-child
cases of familial interstitial pneumonia, and which produced ER
stress in A549 cells. Considering our results in the context of the
dual role of synoviolin as an E3 ubiquitin ligase and collagen
synthesis promoter, this study suggests the existence of a novel
pathway: ER stress increases synoviolin synthesis, which promotes
collagen synthesis, contributing to fibrosis. This result confirm
the association between ER stress and fibrosis that has been
previously reported (23,25). Further studies are required to
fully determine the association between synoviolin and
fibrosis.
Excessive fibrosis affects various organs and
tissues, including the endocardium, the heart, the liver, the
kidneys, the skin, the bone marrow, the retroperitoneum and the
lungs. Advanced fibrosis is a serious disorder that causes organ
dysfunction and death; accordingly, antifibrotic drugs are in
development. Various factors [TGF-β, CTGF, tumor necrosis factor
(TNF), endothelin, chemokines, chemokine receptors, and matrix
metalloproteinases (MMPs)] and effector cells are involved in
fibrosis (19,20,26–28). As a result, there are several
potential therapeutic vectors which may be used in the treatment of
fibrosis, and thus a wide range of candidate therapeutic agents is
available (8).
In the present study, we demonstrate that synoviolin
expression and collagen secretion are enhanced by transfecting A549
cells with SP-CΔexon4 and that collagen secretion is
suppressed by transfection with synoviolin siRNA or the synoviolin
inhibitor, LS-102. Based on these findings and the recent studies
indicating that synoviolin is involved in liver fibrosis (9) and renal fibrosis (10), our data suggest that the
synoviolin inhibitor, LS-102 represents a candidate antifibrotic
agent for the treatment of interstitial pneumonia; however, further
studies, including in vivo studies, are required in order to
confirm this therapeutic potential.
Acknowledgements
We thank the staff at the Future Medical Science at
the Institute of Medical Science and the Integrated Gene Editing
Section of Tokyo Medical University for their assistance. This
study was funded in part by grants from the Naito Foundation,
Natural Science Scholarship Daiichi-Sankyo Foundation of Life
Science, Mitsubishi Tanabe Pharma Corporation, Bureau of Social
Welfare and Public Health, The Ministry of Health Labour and
Welfare, The Ministry of Education, Culture, Sports, Science and
Technology, Japan Society for the Promotion of Science, Health
Labour Sciences Research Grant, Takeda Science Foundation,
AstraZeneca R&D Grant 2013, Grant-in-Aid for Exploratory
Research, Grant-in-Aid for Young Scientists (B).
References
|
1
|
Nogee LM, Dunbar AE III, Wert SE, Askin F,
Hamvas A and Whitsett JA: A mutation in the surfactant protein C
gene associated with familial interstitial lung disease. N Engl J
Med. 344:573–579. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Beers MF and Mulugeta S: Surfactant
protein C biosynthesis and its emerging role in conformational lung
disease. Annu Rev Physiol. 67:663–696. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mulugeta S and Beers MF: Surfactant
protein C: its unique properties and emerging immunomodulatory role
in the lung. Microbes Infect. 8:2317–2323. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nogee LM: Genetics of the hydrophobic
surfactant proteins. Biochim Biophys Acta. 1408:323–333. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mulugeta S, Nguyen V, Russo SJ, Muniswamy
M and Beers MF: A surfactant protein C precursor protein BRICHOS
domain mutation causes endoplasmic reticulum stress, proteasome
dysfunction, and caspase 3 activation. Am J Respir Cell Mol Biol.
32:521–530. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Maguire JA, Mulugeta S and Beers MF:
Endoplasmic reticulum stress induced by surfactant protein C
BRICHOS mutants promotes proinflammatory signaling by epithelial
cells. Am J Respir Cell Mol Biol. 44:404–414. 2011. View Article : Google Scholar :
|
|
7
|
Wynn TA: Cellular and molecular mechanisms
of fibrosis. J Pathol. 214:199–210. 2008. View Article : Google Scholar
|
|
8
|
Wynn TA and Ramalingam TR: Mechanisms of
fibrosis: therapeutic translation for fibrotic disease. Nat Med.
18:1028–1040. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hasegawa D, Fujii R, Yagishita N, et al:
E3 ubiquitin ligase synoviolin is involved in liver fibrogenesis.
PLoS One. 5:e135902010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li Shen LY, Ding Y, Liu Y, Su D and Liang
X: Hrd1 participates in the regulation of collagen I synthesis in
renal fibrosis. Mol Cell Biochem. 386:35–44. 2014. View Article : Google Scholar
|
|
11
|
Amano T, Yamasaki S, Yagishita N, et al:
Synoviolin/Hrd1, an E3 ubiquitin ligase, as a novel pathogenic
factor for arthropathy. Genes Dev. 17:2436–2449. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kaneko M, Ishiguro M, Niinuma Y, Uesugi M
and Nomura Y: Human HRD1 protects against ER stress-induced
apoptosis through ER-associated degradation. FEBS Lett.
532:147–152. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kaneko M and Nomura Y: ER signaling in
unfolded protein response. Life Sci. 74:199–205. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang WJ, Mulugeta S, Russo SJ, et al:
Deletion of exon 4 from human surfactant protein C results in
aggresome formation and generation of a dominant negative. J Cell
Sci. 116:683–692. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yamasaki S, Yagishita N, Sasaki T, et al:
Cytoplasmic destruction of p53 by the endoplasmic
reticulum-resident ubiquitin ligase ‘Synoviolin’. EMBO J.
26:113–122. 2007. View Article : Google Scholar
|
|
16
|
Tsuchimochi K, Yagishita N, Yamasaki S, et
al: Identification of a crucial site for synoviolin expression. Mol
Cell Biol. 25:7344–7356. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nogee LM, Dunbar AE III, Wert S, Askin F,
Hamvas A and Whitsett JA: Mutations in the surfactant protein C
gene associated with interstitial lung disease. Chest.
121:S20S–S21. 2002. View Article : Google Scholar
|
|
18
|
Thomas AQ, Lane K, Phillips J III, et al:
Heterozygosity for a surfactant protein C gene mutation associated
with usual interstitial pneumonitis and cellular nonspecific
interstitial pneumonitis in one kindred. Am J Respir Crit Care Med.
165:1322–138. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Border WA and Noble NA: Transforming
growth factor beta in tissue fibrosis. N Engl J Med. 331:1286–1292.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bonniaud P, Margetts PJ, Kolb M,
Haberberger T, Kelly M, Robertson J and Gauldie J: Adenoviral gene
transfer of connective tissue growth factor in the lung induces
transient fibrosis. Am J Respir Crit Care Med. 168:770–778. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Myllyharju J: Prolyl 4-hydroxylases, the
key enzymes of collagen biosynthesis. Matrix Biol. 22:15–24. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yagishita N, Aratani S, Leach C, et al:
RING-finger type E3 ubiquitin ligase inhibitors as novel candidates
for the treatment of rheumatoid arthritis. Int J Mol Med.
30:1281–1286. 2012.PubMed/NCBI
|
|
23
|
Tanjore H, Cheng DS, Degryse AL, Zoz DF,
Abdolrasulnia R, Lawson WE and Blackwell TS: Alveolar epithelial
cells undergo epithelial-to-mesenchymal transition in response to
endoplasmic reticulum stress. J Biol Chem. 286:30972–30980. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tanjore H, Lawson WE and Blackwell TS:
Endoplasmic reticulum stress as a pro-fibrotic stimulus. Biochim
Biophys Acta. 1832:940–947. 2013. View Article : Google Scholar :
|
|
25
|
Zhong Q, Zhou B, Ann DK, et al: Role of
endoplasmic reticulum stress in epithelial-mesenchymal transition
of alveolar epithelial cells: effects of misfolded surfactant
protein. Am J Respir Cell Mol Biol. 45:498–509. 2011. View Article : Google Scholar :
|
|
26
|
Hogaboam CM, Steinhauser ML, Chensue SW
and Kunkel SL: Novel roles for chemokines and fibroblasts in
interstitial fibrosis. Kidney Int. 54:2152–2159. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Arai M, Ikawa Y, Chujo S, et al: Chemokine
receptors CCR2 and CX3CR1 regulate skin fibrosis in the mouse model
of cytokine-induced systemic sclerosis. J Dermatol Sci. 69:250–258.
2013. View Article : Google Scholar
|
|
28
|
Fernandez IE and Eickelberg O: New
cellular and molecular mechanisms of lung injury and fibrosis in
idiopathic pulmonary fibrosis. Lancet. 380:680–688. 2012.
View Article : Google Scholar : PubMed/NCBI
|