Introduction
Inflammation is the attempt of the body to remove
harmful stimuli and initiate the healing process. However, chronic
inflammatory processes are involved in the pathogenesis of common
inflammation-associated diseases (1,2).
Various infectious agents, such as bacteria and viruses, cause
inflammation and macrophages play a critical role in initiating and
propagating inflammatory responses by releasing pro-inflammatory
mediators, such as nitric oxide (NO), prostaglandin E2
(PGE2) and cytokines to promote the inflammatory
response (3,4).
Lipopolysaccharide (LPS), a major component of the
outer membrane of Gram-negative bacteria, activates a number of
major inflammatory cellular effects by inducing several
interconnecting mechanisms through specific surface molecules known
as pathogen-associated molecular patterns, which bind to Toll-like
receptors (TLRs), particularly TLR4, leading to the release of a
wide variety of inflammatory inducers (5,6).
The LPS-initiated signaling cascade occurs by binding of LPS to its
receptor complex, and this leads to activation of the nuclear
factor-κB (NF-κB) pathway (7,8).
In normal cells, NF-κB is constitutively localized in the cytosol
as a homodimer or heterodimer, which is associated with the NF-κB
inhibitor, inhibitory-κBα (IκB). Upon stimulation with LPS, IκB
proteins are phosphorylated, ubiquitinated and rapidly degraded by
the ubiquitin-proteasome pathway, and the resulting free NF-κB
migrates into the nucleus and activates transcription of
NF-κB-dependent inflammatory enzymes and cytokines (5,9,10).
These observations indicate that antagonistic agents of TLR4 can
inactivate pro-inflammatory downstream signaling pathways by
suppressing the differential target gene expression and cellular
responses.
Several epidemiological studies have shown that
consuming allium vegetables is associated with a decreased risk of
various diseases (11,12). Among them, garlic (Allium
sativam L.) has long been used for culinary and medicinal
purposes in numerous cultures. Accumulating studies have
demonstrated that garlic has a wide range of biological activities
against a number of chronic diseases, including cardiovascular
problems, diabetes, infections and even cancer (13–15). Garlic is a particularly rich
source of organosulfur compounds (OSCs), such as diallyl sulfide
(DAS), diallyl disulfide (DADS) and diallyl trisulfide (DATS),
which are believed to be responsible for its flavor and aroma, as
well as its potential health benefits (16–18). In particular, studies have shown
that the antioxidant potential is in the order DATS > DADS >
DAS (19–21). Na et al (22) found the same order for the
anticancer potential, suggesting that the number of sulfur atoms
plays a vital role in the biological activities of OSCs. Although
DATS has been suggested recently as the strongest inhibitor of
cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS)
among OSCs (21,23), the molecular mechanisms underlying
the anti-inflammatory effects of DATS have not been extensively
explored.
Therefore, the present study was carried out to
further investigate the potential anti-inflammatory effect and
underlying molecular mechanism of DATS using a murine RAW 264.7
macrophage model, which can be stimulated with LPS to mimic a
status of inflammation. The results suggest that DATS effectively
suppresses LPS-induced inflammatory signaling by inactivating the
NF-κB pathway and inhibiting TLR4.
Materials and methods
Cell culture
The murine RAW 264.7 macrophage cell line was
obtained from the American Type Culture Collection (Manassas, VA,
USA) and was cultured in Dulbecco’s modified Eagle’s medium
(Gibco-BRL, Grand Island, NY, USA) supplemented with 10% fetal
bovine serum (Gibco-BRL), 100 U/ml penicillin, 100 μg/ml
streptomycin, and 2 mM L-glutamine, at 37°C in a 5% CO2
humidified air environment.
DATS treatment and MTT assay
DATS was obtained from LKT Laboratories (St. Paul,
MN, USA) and was dissolved in dimethyl sulfoxide (DMSO;
Sigma-Aldrich, St. Louis, MO, USA) as a 100 mM stock solution and
was stored in aliquots at −20°C. RAW 264.7 cells were incubated
with different concentrations of DATS or 100 ng/ml LPS
(Sigma-Aldrich) alone, or pretreated with DATS for 1 h before LPS
for the cell viability assay. After 24 h, the medium was removed
and the cells were incubated with 0.5 mg/ml
3-(4,5-dimethylthiazol-2-yl)-2,5-di-phenyltetrazolium bromide (MTT;
Sigma-Aldrich) solution for 2 h. The supernatant was discarded and
the formazan blue, which was formed in the cells, was dissolved in
DMSO. Optical density was measured at 540 nm with a microplate
reader (Dynatech Laboratories, Chantilly, VA, USA) and growth
inhibition was assessed as the percent viability, in which
vehicle-treated cells were considered as 100% viable.
Measurement of NO production
Concentrations of NO in the culture supernatants
were determined by measuring nitrite, a stable oxidation product of
nitric oxide, using the Griess reagent (Sigma-Aldrich). Briefly,
cells (5×105 cells/ml) were stimulated in 24-well plates
with DATS and/or LPS for 24 h. Subsequently, 100 μl of each
culture supernatant was mixed with an equal volume of Griess
reagent. After 10 min incubation at room temperature, absorbance
was measured at 540 nm and nitrite production was determined with
an NaNO2 standard curve (24).
Determination of PGE2, tumor
necrosis factor-α (TNF-α) and interleukin (IL)-1β production
The cells were incubated with DATS in either the
presence or absence of LPS (100 ng/ml) for 24 h to measure the
quantity of PGE2. A 100 μl aliquot of culture
medium supernatant was collected and the concentration (pg/ml) of
PGE2 in the cell culture medium was calculated based on
the concentrations of the standard solution using a PGE2
enzyme-linked immunosorbent assay (ELISA) kit following the
manufacturer’s instructions (Cayman Chemical Co., Ann Arbor, MI,
USA). The levels of TNF-α and IL-1β were also measured with ELISA
kits (R&D Systems, Minneapolis, MN, USA) according to the
manufacturer’s instructions (25).
RNA isolation and reverse
transcriptase-polymerase chain reaction (RT-PCR)
Total RNA was isolated using TRIzol reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA) according to the
manufacturer’s instructions, and reverse-transcribed using M-MLV
reverse transcriptase (Promega, Madison, WI, USA) to produce cDNAs.
RT-generated cDNAs encoding iNOS, COX-2, TNF-α
and IL-1β genes were amplified by PCR using selective
primers, which were purchased from Bioneer (Seoul, Korea). The PCR
primers were as follows: mouse iNOS (5′-ATG TCC GAA GCA AAC
ATC AC-3′ and 5′-TAA TGT CCA GGA AGT AGG TG-3′), COX-2
(5′-CAG CAA ATC CTT GCT GTT CC-3′ and 5′-TGG GCA AAG AAT GCA AAC
ATC-3′), TNF-α (5′-ATG AGC ACA GAA AGC ATG ATC-3′ and 5′-TAC
AGG CTT GTC ACT CGA ATT-3′) and IL-1β (5′-ATG GCA ACT GTT
CCT GAA CTC AAC T-3′ and 5′-TTT CCT TTC TTA GAT ATG GAC AGG AC-3′).
Following amplification, the PCR reactants were electrophoresed in
1% agarose gels and visualized by ethidium bromide (Sigma-Aldrich)
staining. In a parallel experiment, glyceraldehyde-3-phosphate
dehydrogenase was used as an internal control.
Protein extraction and western
blotting
The cells were harvested and lysed with lysis buffer
[20 mM sucrose, 1 mM EDTA, 20 μM Tris-Cl (pH 7.2), 1 mM
dithiothreitol, 10 mM KCl, 1.5 mM MgCl2 and 5
μg/ml aprotinin] for 1 h. In a parallel experiment, nuclear
and cytosolic proteins were prepared using nuclear extraction
reagents (Pierce, Rockford, IL, USA) according to the
manufacturer’s instructions. Protein concentration was measured
using a Bio-Rad protein assay (Bio-Rad Laboratories, Hercules, CA,
USA) according to the manufacturer’s instructions. For western blot
analysis, equivalent amounts of proteins were separated by
electrophoresis on sodium dodecyl sulfate-polyacrylamide gels and
transferred to nitrocellulose membranes (Schleicher and Schuell,
Keene, NH, USA). After blocking with 5% skimmed milk, the membranes
were incubated with protein-specific antibodies for 1 h,
subsequently incubated with appropriate enzyme-linked secondary
antibodies [mouse IgG, HRP-linked whole antibody (NA931) and rabbit
IgG, HRP-linked whole antibody (NA934)] (Amersham Corp., Arlington
Heights, IL, USA) and visualized by enhanced chemiluminescence
(Amersham Corp.) according to the manufacturer’s instructions.
Anti-iNOS (#610330) and anti-COX-2 (#160106) antibodies were
purchased from BD Biosciences (San Jose, CA, USA) and Cayman
Chemical Co., respectively. Anti-IL-1β (SC7884), anti-TLR4
(SC10741), anti-MyD88 (SC11356), anti-NF-κB p65 (SC109), anti-actin
(SC1616) and anti-GAPDH (SC32233) antibodies were obtained from
Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). Anti-TNF-α
(#3737) and anti-IκB (#4812) antibodies were purchased from Cell
Signaling Technology, Inc. (Danvers, MA, USA). Anti-nucleolin
(AB22758) antibody was obtained from Abcam (Cambridge, MA,
USA).
Electrophoretic mobility assay
(EMSA)
EMSA was performed with the nuclear extract.
Synthetic complementary NF-κB (5′-AGT TGA GGG GAC TTT CCC AGG C-3′)
binding oligonucleotides (Santa Cruz Biotechnology, Inc.) were
3′-biotinylated using the biotin 3′-end DNA labeling kit (Pierce)
according to the manufacturer’s instructions, and annealed for 30
min at room temperature. Assays were loaded onto native 4%
polyacrylamide gels pre-electrophoresed for 60 min in 0.5X Tris
borate/EDTA before being transferred onto a positively charged
nylon membrane (Hybond™-N+) in 0.5X Tris borate/EDTA at
100 V for 30 min. The transferred DNAs were cross-linked to the
membrane at 120 mJ/cm2. Horseradish
peroxidase-conjugated streptavidin was used according to the
manufacturer’s instructions to detect the transferred DNA.
Immunofluorescence staining
The RAW 264.7 cells were cultured on glass
coverslips in 6-well plates for 24 h, stimulated with LPS in the
presence or absence of DATS, fixed with 4% paraformaldehyde in
phosphate-buffered saline (PBS) for 10 min at room temperature, and
permeabilized with 100% MeOH for 10 min at 20°C. The anti-NF-κB p65
antibody was applied for 1 h followed by a 1 h incubation with
fluorescein isothiocyanate (FITC)-conjugated donkey anti-rabbit IgG
(Jackson ImmunoResearch Laboratories, Inc., West Grove, PA, USA).
Subsequent to washing with PBS, nuclei were stained with
4,6-diamidino-2-phenyllindile (Sigma-Aldrich), and fluorescence was
visualized using a fluorescence microscope (Carl Zeiss, Oberkochen,
Germany). RAW 264.7 cells were stimulated with Alexa Fluor
488-conjugated LPS (100 ng/ml, AF-LPS; Invitrogen) for 30 min in
the presence or absence of DATS for the LPS/TLR4 complex formation
assay. The cells were fixed, stained with anti-TLR4 antibody for 90
min at 4°C and then incubated with secondary antibodies conjugated
with Alexa Fluor 594 (1:200; Invitrogen) for 1 h. The stained cells
were observed under a fluorescence microscope.
Measurement of TLR4 expression on cell
surface
RAW 264.7 cells were incubated with AF-LPS in the
presence or absence of DATS for 1 h. The cells were washed twice
with PBS, harvested with 0.005% EDTA and analyzed by flow
cytometry. Alexa 488 was excited using a 488 argon-ion laser line
and detected on a channel FL1 using a 530 nm emission filter. The
fluorescence emission of samples was recorded by a flow cytometer
(26).
Statistical analysis
Data are presented as mean ± standard deviation.
Statistical significance was determined using an analysis of
variance followed by Student’s t-test. P<0.05 was considered to
indicate a statistically significant difference.
Results
Effects of DATS and LPS on RAW 264.7
macrophage viability
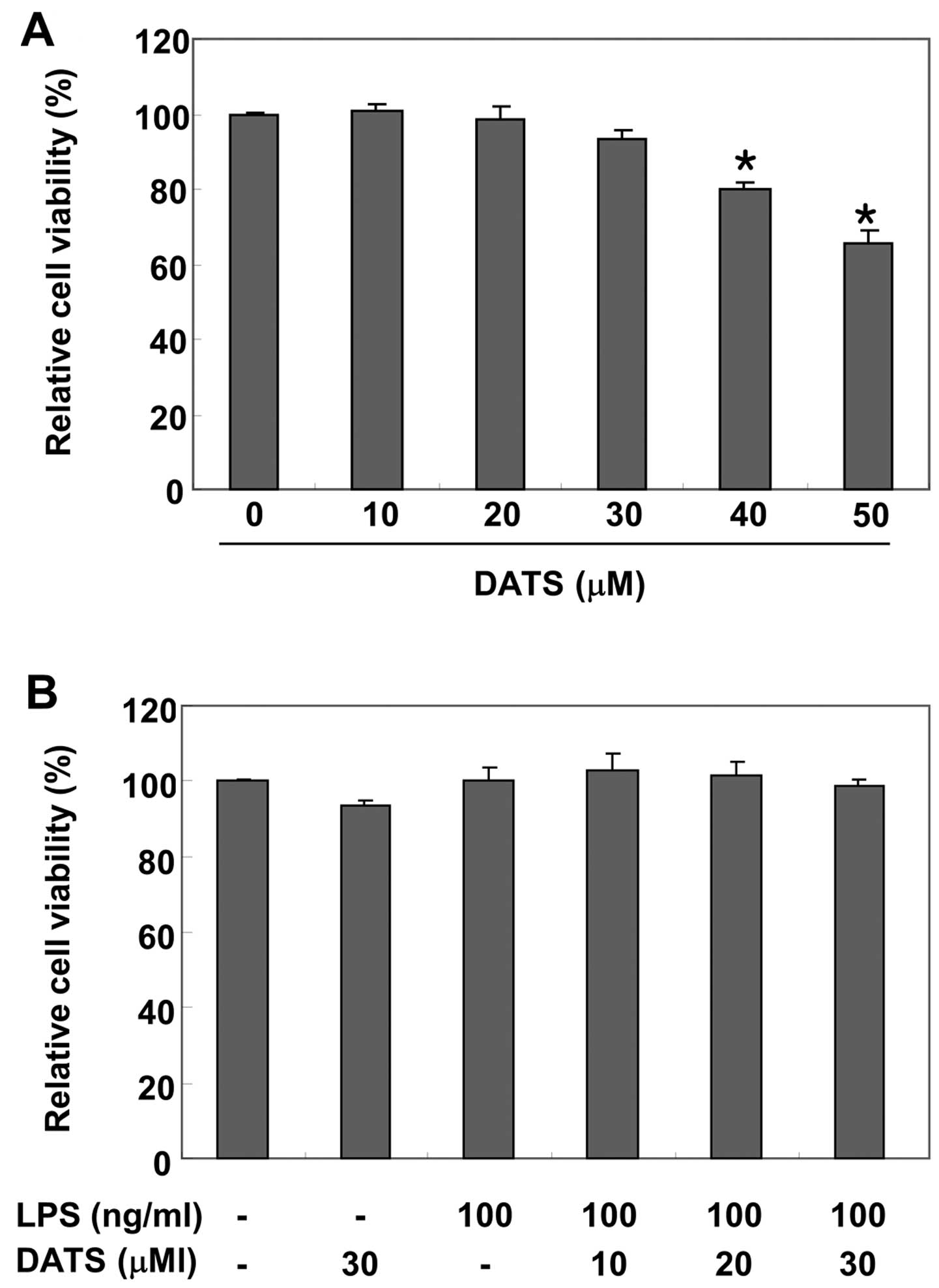
To examine whether DATS is cytotoxic to RAW 264.7
macrophages, the cells were exposed to DATS for 24 h in the
presence or absence of LPS, and cell viability was measured by the
MTT assay. The results showed no effect of DATS on cell viability
at concentrations of 10, 20 or 30 μM (Fig. 1A); however, at higher
concentrations the MTT assay suggested an adverse influence of DATS
on cell viability. Furthermore, no significant cytotoxic effects
were observed at ≤30 μM DATS in the presence of LPS
(Fig. 1B).
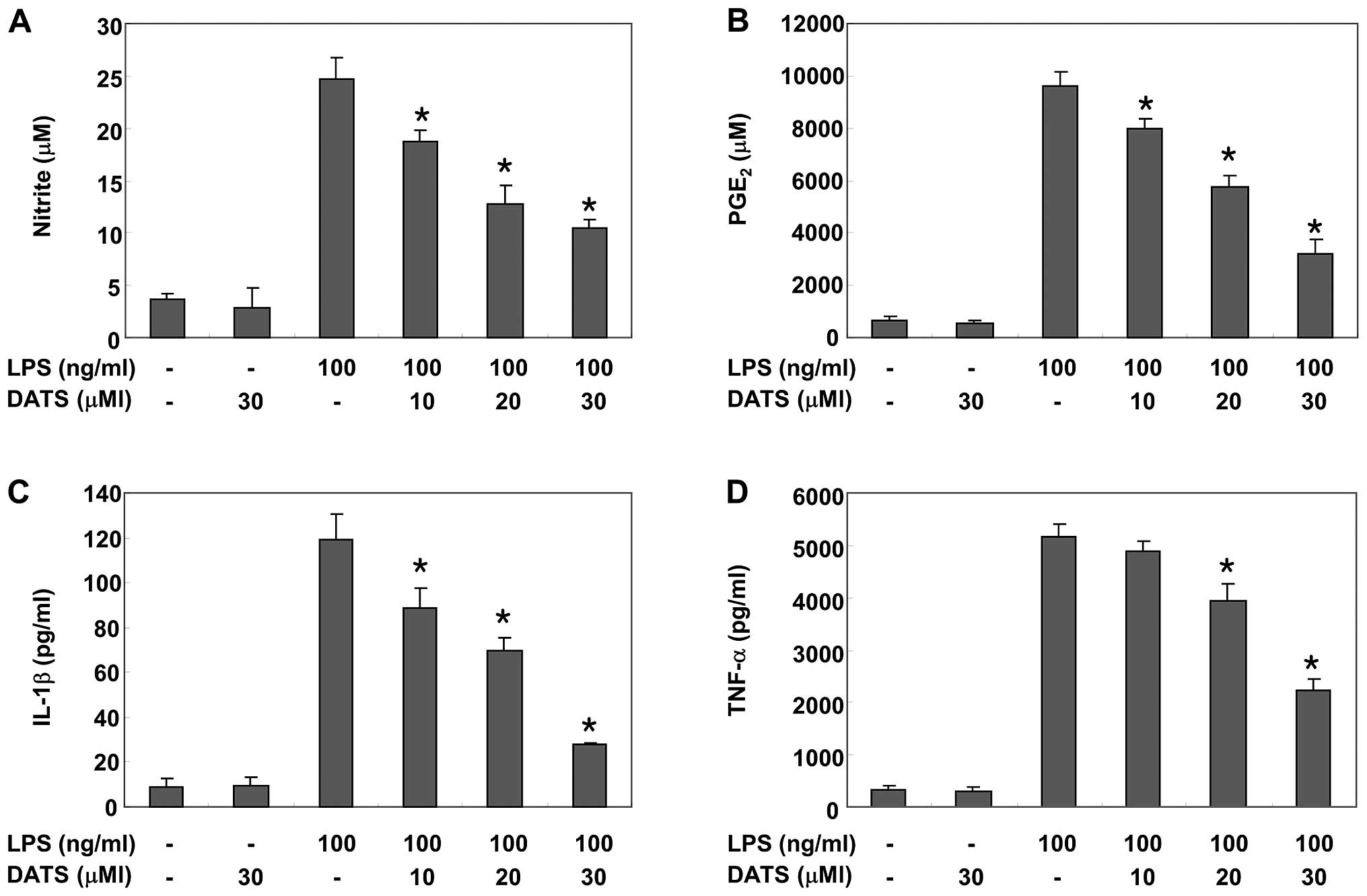
DATS reduces LPS-induced production of
pro-inflammatory mediators and cytokines
Pro-inflammatory mediators including NO and
PGE2, and pro-inflammatory cytokines, such as IL-1β and
TNF-α, that were released into the culture medium were measured
using the Griess reagent and ELISA to examine the inhibitory effect
of DATS on LPS-induced inflammatory responses. According to the NO
detection assay, LPS alone markedly induced NO production compared
to that generated by the control. However, pretreatment with DATS
significantly repressed the levels of NO production in
LPS-stimulated RAW 264.7 cells in a concentration-dependent manner
≤30 μM (Fig. 2A). The
levels of PGE2 and tested cytokines also increased
significantly in the culture media of LPS-stimulated RAW 264.7
cells, but the increases were significantly attenuated in a
concentration-dependent manner by DATS pretreatment (Fig. 2B–D).
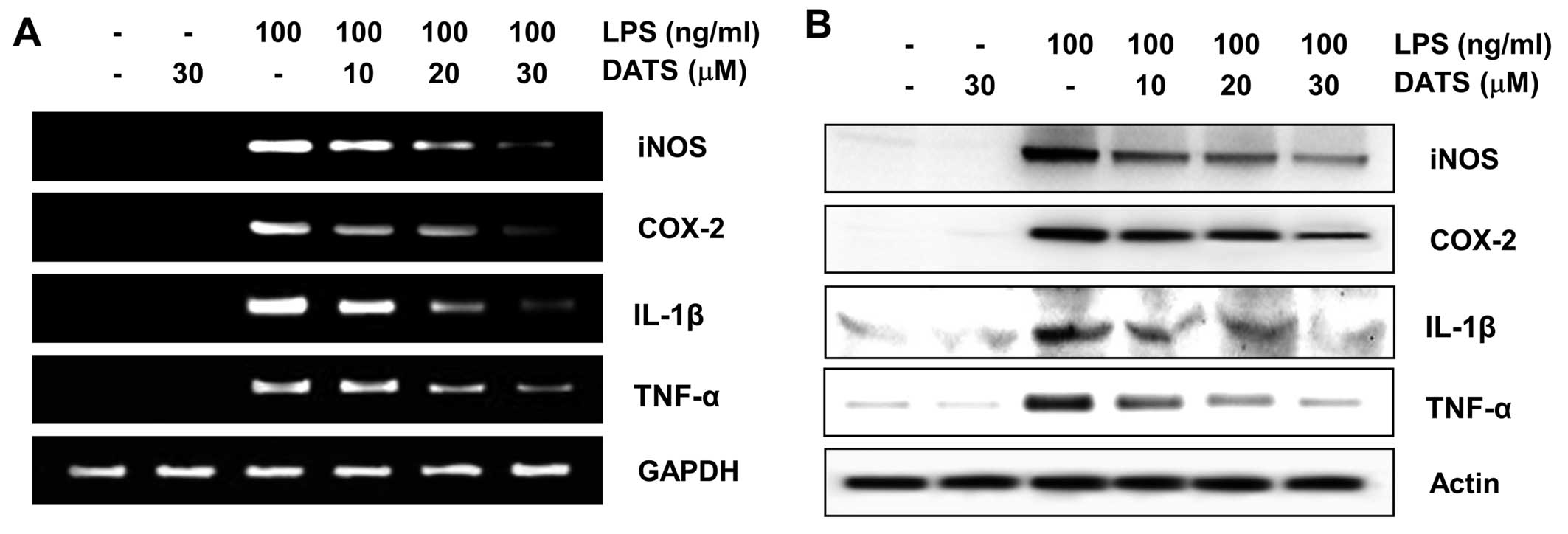
DATS inhibits LPS-induced expression of
pro-inflammatory enzymes and cytokines
The effects of DATS on the levels of iNOS, COX-2,
TNF-α and IL-1β mRNA and protein expression was measured by RT-PCR
and western blot analyses to elucidate the mechanism involved in
the inhibition of pro-inflammatory mediators and cytokines
generated by DATS in LPS-stimulated RAW 264.7 cells. mRNA and
protein expression in unstimulated RAW 264.7 cells was undetectable
or extremely low. However, expression increased markedly in
response to LPS, which was significantly inhibited by pretreatment
with DATS (Fig. 3). These results
indicate that the reduced expression of pro-inflammatory enzymes
and cytokines at the transcriptional levels contributed to the
inhibitory effect of DATS on LPS-induced NO, PGE2, IL-1β
and TNF-α production.
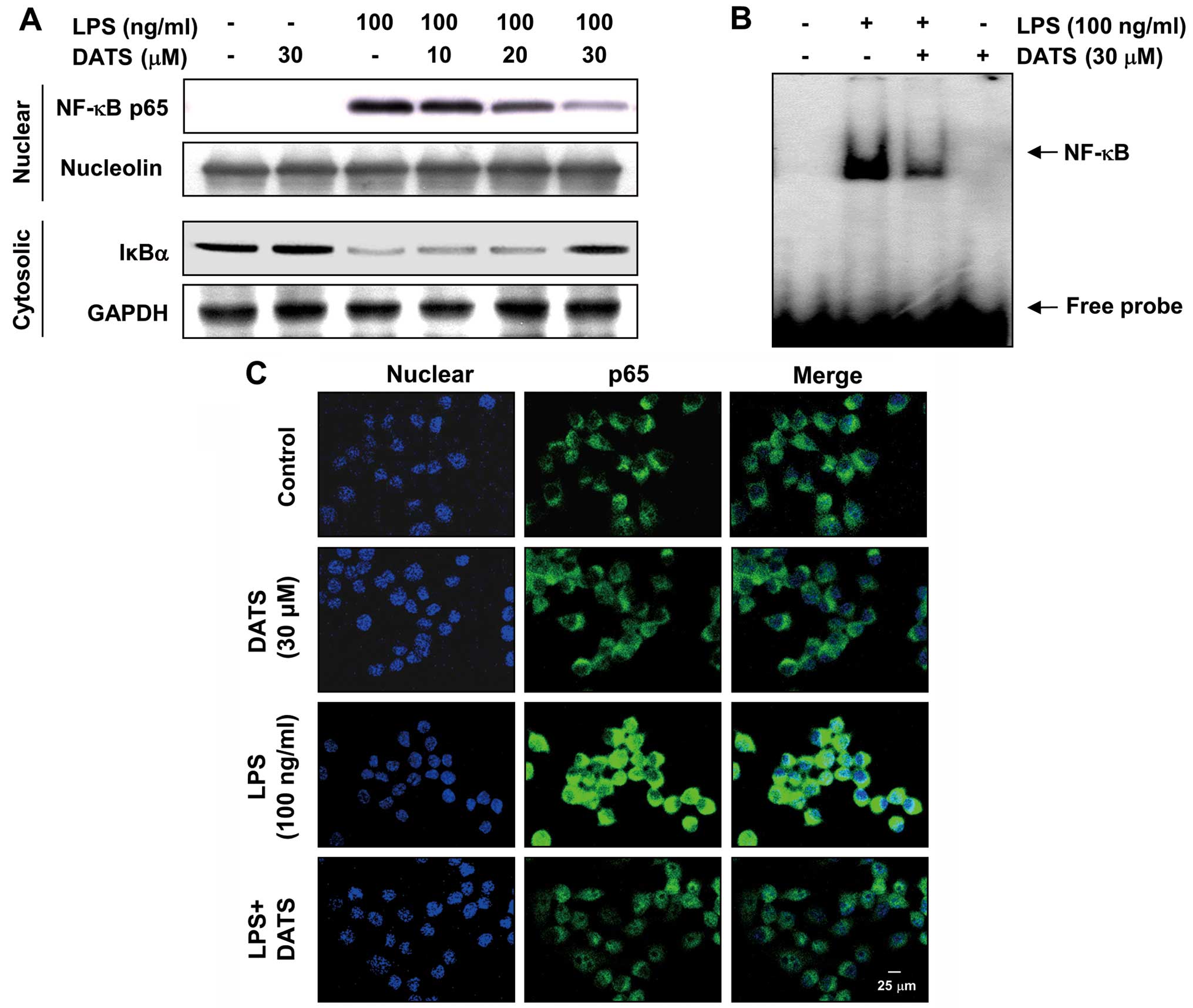
DATS blocks LPS-induced nuclear
translocation of NF-κB p65 and degradation of IκBα
As previous studies suggested that NF-κB is an
important transcription factor regulating the expression of
pro-inflammatory enzymes and cytokines (5,9,10),
whether DATS blocked the NF-κB signaling pathway was explored. The
western blot analysis results showed that the amount of NF-κB p65
in the nucleus was markedly increased within 30 min of exposure to
LPS alone, concomitant with degradation of IκBα in the cytosol.
However, LPS-induced NF-κB p65 levels in the nuclear fractions
decreased markedly by DATS pretreatment, and LPS-induced IκBα
degradation was clearly blocked in a concentration-dependent manner
by pretreatment with DATS (Fig.
4A). EMSA also showed that treatment with LPS causes an
increase in NF-κB DNA-binding activity at 30 min, while
pretreatment of the cells with DATS for 1 h resulted in a
significant reduction in the DNA-binding activity of NF-κB
(Fig. 4B). In parallel with
immunoblotting data, the immunofluorescence images revealed that
the nuclear accumulation of NF-κB p65 was not induced in the cells
following treatment with DATS alone in the absence of LPS
stimulation; however, it was strongly induced following LPS
stimulation, and the shift of NF-κB to the nucleus was completely
abolished subsequent to pretreating the cells with DATS (Fig. 4C). Taken together, these results
suggest that DATS treatment inhibits LPS-induced NF-κB
translocation by attenuating the IκBα degradation.
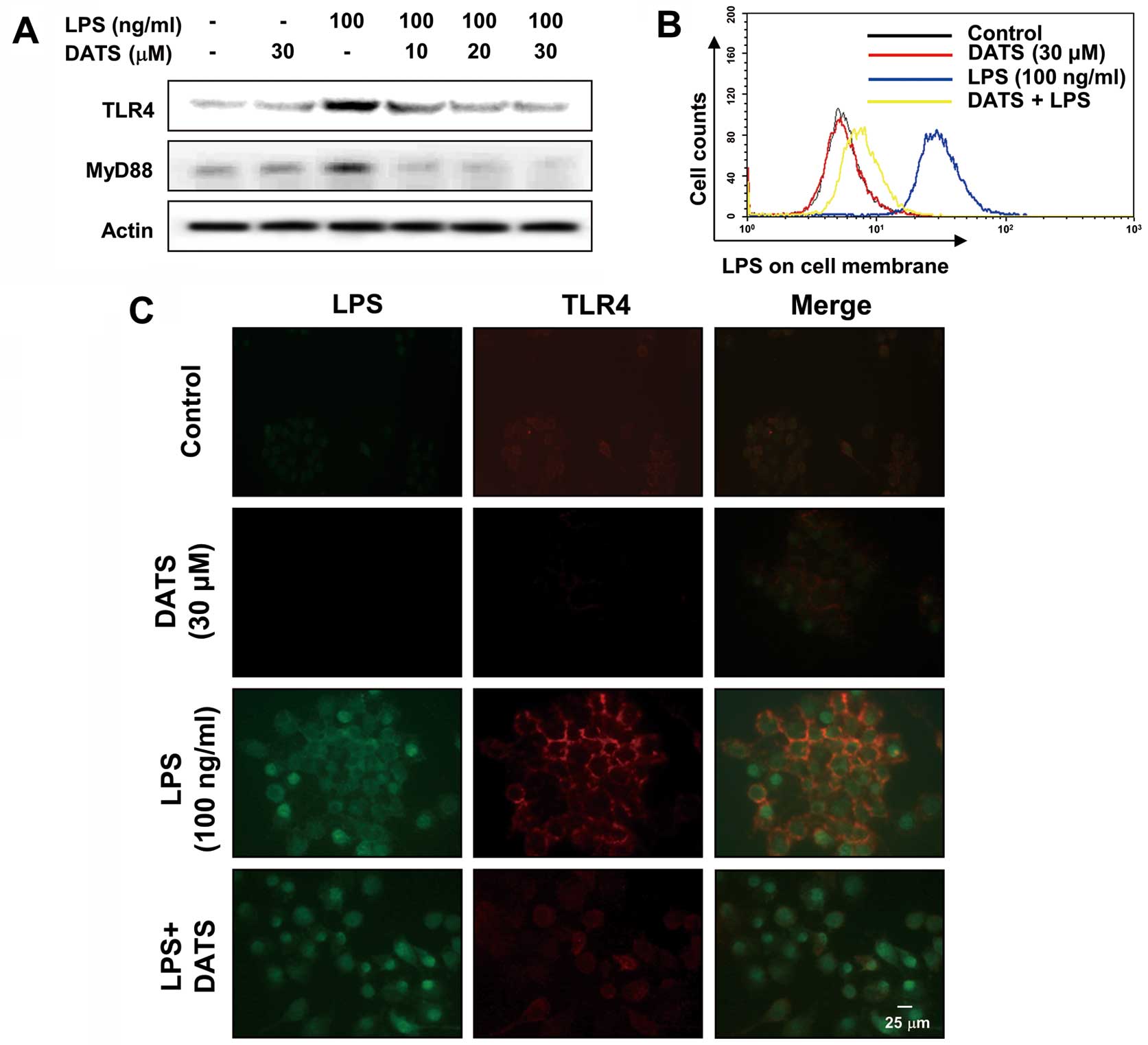
DATS inhibits LPS-induced TLR4 and
myeloid differentiation factor 88 (MyD88) expression, and the
interaction between LPS and TLR4
The effects of DATS was assessed on the expression
of LPS-induced TLR4 and MyD88 (a TLR4-associated molecule)
expression to determine the involvement of the TLR4 signaling
pathway in the DATS-mediated anti-inflammatory potential. The
increased levels of TLR4 and MyD88 proteins in LPS-treated RAW
264.7 cells were completely blocked in a concentration-dependent
fashion by pretreatment with DATS (Fig. 5A). Furthermore, when cells were
treated with AF-LPS alone, fluorescence of LPS and TLR4 were
observed outside the cell membrane by the immunofluorescence assay
(Fig. 5B). However, the
fluorescence intensity of TLR4 was markedly attenuated in the
presence of DATS, suggesting that DATS may inhibit the interaction
between LPS and TLR4 on the RAW 264.7 cell surface. In addition,
treatment with LPS in the presence of DATS significantly prevented
the binding of LPS to the RAW 264.7 cell surface (Fig. 5C), indicating that DATS may
interfere with the clustering of TLR4 with MyD88.
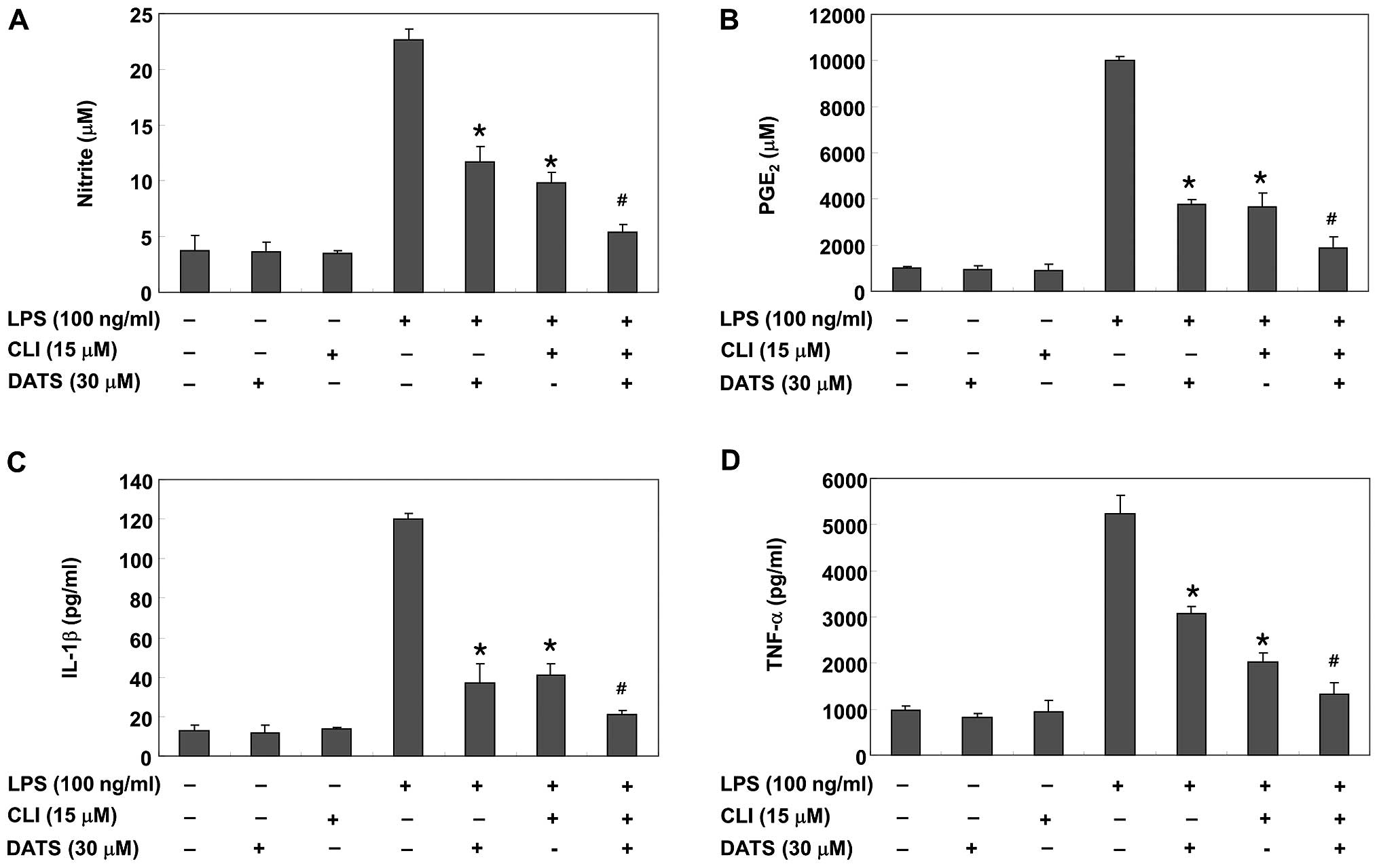
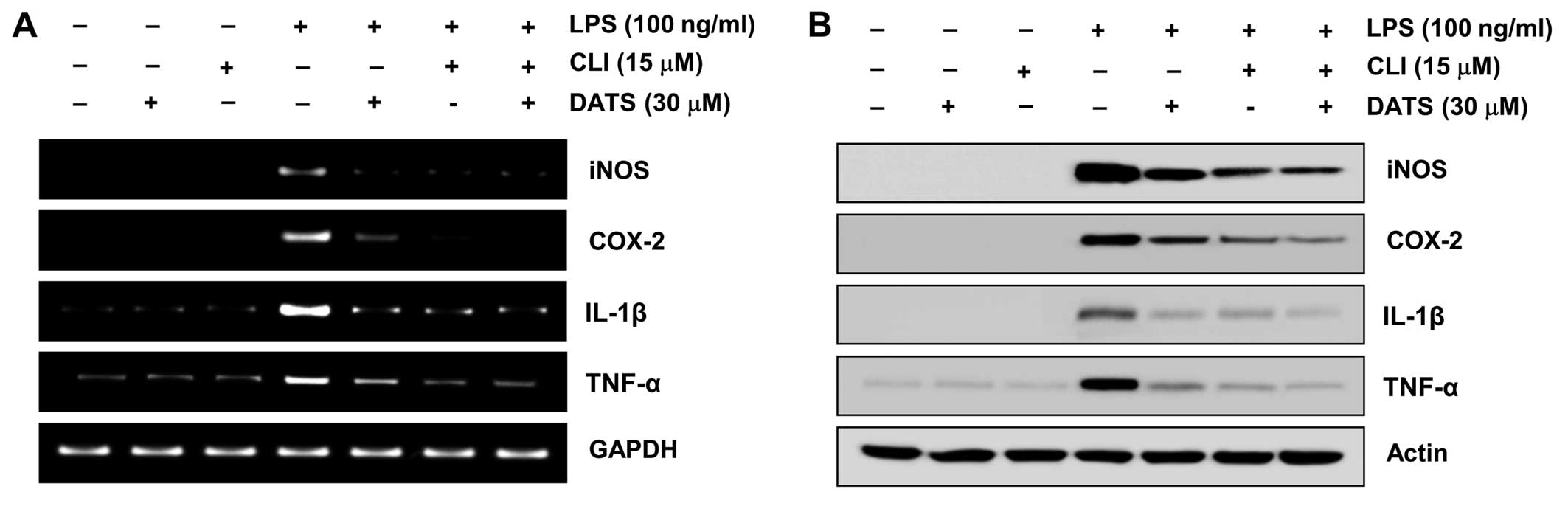
CLI-095, a TLR4 inhibitor, increases the
anti-inflammatory potential of DATS
The effects of CLI-095, which blocks TLR4-mediated
signaling, were evaluated to further confirm the involvement of the
TLR4 signaling pathway in the DATS-mediated anti-inflammatory
potential. CLI-095 reduced the LPS-induced production of
pro-inflammatory mediators and cytokines by suppressing the
transcriptional levels of their corresponding genes (Fig. 6). Furthermore, DATS and CLI-095
co-treatment synergistically inhibited LPS-induced synthesis of NO
and PGE2, as well as expression of iNOS and COX-2. DATS
and CLI-095 co-treatment synergistically inhibited LPS-induced
release and expression of IL-1β and TNF-α (Fig. 7). These results verify that the
inhibitory effects of DATS on the LPS-induced inflammatory
responses may result from suppression of the TLR4 signaling
pathway.
Discussion
In the present study, DATS was shown to be suitable
for reducing LPS-induced NO and PGE2 synthesis in RAW
264.7 macrophages. This suppression was possibly due to inhibiting
the upregulation of iNOS and COX-2 at the transcriptional level
during macrophage activation by LPS. Parallel inhibitory effects on
synthesis and expression of TNF-α and IL-1β were also observed in
LPS-stimulated RAW 264.7 cells.
During the inflammatory process, LPS induces the
overproduction of pro-inflammatory mediators, such as NO and
PGE2, which are generated by the inducible enzymes iNOS
and COX-2, respectively (3,4).
NO is controlled by NO synthase and iNOS is markedly upregulated in
inflammatory disorders. Another enzyme that plays a pivotal role in
mediating inflammation is COX-2, which catalyzes the rate-limiting
step in the synthesis of PGE2 from arachidonic acid. In
addition to NO and PGE2, numerous inflammatory cytokines
have been recently demonstrated to be initiators and mediators of
the inflammatory response. Among these, TNF-α and IL-1β are the
major pro-inflammatory cytokines produced by activated macrophages,
and their excessive production has been linked to the development
of chronic inflammatory diseases (27,28). Additionally, TNF-α and IL-1β
production is critical for the synergistic induction of NO and
PGE2 production in LPS-stimulated macrophage cells
(27,29). Therefore, overproduction of these
inflammatory factors is a histopathological characteristic of
various inflammation-mediated diseases, and the selective
inhibition of their production and function may be effectively
therapeutic in the control of inflammatory disorders.
Consistent with the findings of the present study
(Figs. 2 and 3), DATS inhibited LPS-induced NO and
iNOS expression, as well as intracellular reactive oxygen species
(ROS) generation, and the inhibitory effect of DATS on LPS-induced
iNOS expression is possibly attributed to its antioxidant potential
to inhibit NF-κB activation in RAW 264.7 macrophages (21). DATS has been reported to reduce
the levels of LPS-induced PGE2 synthesis and several
cytokines, including TNF-α, by inactivating mitogen-activated
protein kinases and NF-κB signaling pathways in the same cell line
(23). This study also revealed
that DATS reduces LPS-induced ROS and activates transcription
factor NF-E2 p45-related factor 2-mediated expression of
heme oxygenase-1 and NAD(P)H quinine oxidoreductase 1,
demonstrating that cross-talk between antioxidant and
anti-inflammation in the inhibition of LPS-induced inflammation by
DATS. In addition, DATS inhibits TNF-α production by inhibiting its
transcriptional activity and downregulating NF-κB activation in
inflamed mucosa of ulcerative colitis (30). Additionally, DATS ameliorates
dextran sulfate sodium-induced mouse colitis presumably by blocking
inflammatory signaling mediated by NF-κB and signal transducer and
activator of transcription 3 transcription factors, as well as
expression of their target proteins, including COX-2 and iNOS
(31). A previous study has also
suggested that DATS suppresses high glucose-induced cardiomyocyte
apoptosis by inhibiting NADPH oxidase-related ROS and downstream
c-Jun N-terminal kinase/NF-κB signaling (32). A previous study showed that DATS
has a anti-cancer effect by inhibiting the levels of certain
critical genes involved in cancer cell growth and metastasis by
suppressing NF-κB activation (33). In agreement with these previous
observations, the present data also revealed that the
anti-inflammatory effects of DATS appeared to involve inhibiting
NF-κB activation by blocking LPS-stimulated IκBα degradation and
nuclear translocation of the NF-κB p65 protein (Fig. 4). Based on the present study
findings and previous studies, DATS may exert its anti-inflammatory
effect by suppressing the production of these pro-inflammatory
mediators and cytokines by modulating the NF-κB signaling
pathway.
By contrast, increasing evidence suggests that
signaling pathways downstream of TLR4, a pattern recognition
receptor that generates innate immune responses to pathogens such
as LPS, play an important role in the pathogenesis of inflammation
(6). TLR4 has a common TIR motif
in the cytoplasmic domain and shares different adaptor proteins,
including MyD88 for NF-kB activation (34). During infection, LPS directly
binds with cellular TLR4 and induces a cascade of inflammatory
events by triggering overproduction of inflammatory inducers
through receptor dimerization and the recruitment of adapter
molecules (35,36). Several studies have reported that
certain anti-inflammatory agents compete with LPS binding to TLR4,
resulting in the suppression of downstream signaling pathways
(37,38). In addition, TLR4 and MyD88 are
upregulated in various inflammatory processes induced by LPS
(39,40). Consequently, the pharmacological
reduction of LPS-inducible inflammatory regulators through
inhibiting the association between LPS and TLR4 is regarded as an
attractive therapeutic strategy for numerous acute and chronic
inflammatory diseases. In the present study, increased expression
of TLR4 and MyD88 proteins was concentration-dependently reduced in
the presence of DATS (Fig. 5A).
The data also indicate that DATS has inhibitory effects on the
binding of LPS with TLR4 (Fig. 5B and
C), suggesting a possible antagonistic effect of DATS against
TLR4. In addition, combined pretreatment of DATS with CLI-095, a
specific TLR4 signaling inhibitor (41,42), synergistically attenuated the
release of NO and PGE2, as well as expression of
iNOS and COX-2 mRNA and protein (Fig. 6). In accordance with these
results, co-treatment also reduced the production and expression of
TNF-α and IL-1β compared to those in the DATS or CLI-095 alone
treated groups in LPS-treated RAW 264.7 macrophages (Fig. 7). These observations suggest that
DATS may inhibit the initiation of intracellular inflammatory
signaling cascades by attenuating the binding of LPS to TLR4 on
macrophages, which is a pivotal upstream signal for NF-κB
activation. Therefore, the antagonistic function of DATS against
TLR4 may be responsible for the anti-inflammatory effects of DATS
in LPS-stimulated RAW 264.7 macrophages.
In conclusion, the present data reveal that DATS
inhibited LPS-induced iNOS, COX-2, TNF-α and IL-1β expression,
which was accompanied by reducing the corresponding transcriptional
gene expression through interfering with the clustering of LPS with
TLR4. This resulted in blocking the activation of NF-κB signaling
pathway in RAW 264.7 macrophages. These findings provide a further
partial molecular description of the mechanism that underlies the
anti-inflammatory properties of DATS.
Acknowledgments
The present study was supported by a grant (no.
7-19-42) from the Rural Development Administration and the National
Research Foundation of Korea grant funded by the Korea government
(no. 2008-0062611 and 2012046358), Republic of Korea.
References
|
1
|
Lind L: Circulating markers of
inflammation and atherosclerosis. Atherosclerosis. 169:203–214.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bertolini A, Ottani A and Sandrini M: Dual
acting anti-inflammatory drugs: a reappraisal. Pharmacol Res.
44:437–450. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kanno S, Shouji A, Tomizawa A, Hiura T,
Osanai Y, Ujibe M, Obara Y, Nakahata N and Ishikawa M: Inhibitory
effect of naringin on lipopolysaccharide (LPS)-induced endotoxin
shock in mice and nitric oxide production in RAW 264.7 macrophages.
Life Sci. 78:673–681. 2006. View Article : Google Scholar
|
|
4
|
Poltorak A, He X, Smirnova I, Liu MY, Van
Huffel C, Du X, Birdwell D, Alejos E, Silva M, Galanos C,
Freudenberg M, Ricciardi-Castagnoli P, Layton B and Beutler B:
Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations
in Tlr4 gene. Science. 282:2085–2088. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Takeda K and Akira S: Microbial
recognition by Toll-like receptors. J Dermatol Sci. 34:73–82. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shuto T, Kato K, Mori Y, Viriyakosol S,
Oba M, Furuta T, Okiyoneda T, Arima H, Suico MA and Kai H:
Membrane-anchored CD14 is required for LPS-induced TLR4 endocytosis
in TLR4/MD-2/CD14 overexpressing CHO cells. Biochem Biophys Res
Commun. 338:1402–1409. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chan ED and Riches DW: IFN-gamma + LPS
induction of iNOS is modulated by ERK, JNK/SAPK, and p38(mapk) in a
mouse macrophage cell line. Am J Physiol Cell Physiol.
280:C441–C450. 2001.PubMed/NCBI
|
|
8
|
Hattori Y, Hattori S and Kasai K:
Lipopolysaccharide activates Akt in vascular smooth muscle cells
resulting in induction of inducible nitric oxide synthase through
nuclear factor-kappa B activation. Eur J Pharmacol. 481:153–158.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Anwar MA, Basith S and Choi S: Negative
regulatory approaches to the attenuation of Toll-like receptor
signaling. Exp Mol Med. 45:e112013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Datla P, Kalluri MD, Basha K, Bellary A,
Kshirsagar R, Kanekar Y, Upadhyay S, Singh S and Rajagopal V:
9,10-dihydro-2,5-dime-thoxyphenanthrene-1,7-diol, from Eulophia
ochreata, inhibits inflammatory signalling mediated by Toll-like
receptors. Br J Pharmacol. 160:1158–1170. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tattelman E: Health effects of garlic. Am
Fam Physician. 72:103–106. 2005.PubMed/NCBI
|
|
12
|
Moyad MA: Bladder cancer recurrence: Part
II. What do I tell my patients about lifestyle changes and dietary
supplements? Curr Opin Urol. 13:379–383. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yi L and Su Q: Molecular mechanisms for
the anti-cancer effects of diallyl disulfide. Food Chem Toxicol.
57:362–370. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Padiya R and Banerjee SK: Garlic as an
anti-diabetic agent: recent progress and patent reviews. Recent Pat
Food Nutr Agric. 5:105–127. 2013. View Article : Google Scholar
|
|
15
|
Capasso A: Antioxidant action and
therapeutic efficacy of Allium sativum L. Molecules. 18:690–700.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Iciek M, Kwiecień I and Włodek L:
Biological properties of garlic and garlic-derived organosulfur
compounds. Environ Mol Mutagen. 50:247–265. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lea MA: Organosulfur compounds and cancer.
Adv Exp Med Biol. 401:147–154. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Amagase H, Petesch BL, Matsuura H, Kasuga
S and Itakura Y: Intake of garlic and its bioactive components. J
Nutr. 131:955S–962S. 2001.PubMed/NCBI
|
|
19
|
Chen C, Pung D, Leong V, Hebbar V, Shen G,
Nair S, Li W and Kong AN: Induction of detoxifying enzymes by
garlic organosulfur compounds through transcription factor Nrf2:
effect of chemical structure and stress signals. Free Radic Biol
Med. 37:1578–1590. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fukao T, Hosono T, Misawa S, Seki T and
Ariga T: The effects of allyl sulfides on the induction of phase II
detoxification enzymes and liver injury by carbon tetrachloride.
Food Chem Toxicol. 42:743–749. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liu KL, Chen HW, Wang RY, Lei YP, Sheen LY
and Lii CK: DATS reduces LPS-induced iNOS expression, NO
production, oxidative stress, and NF-kappaB activation in RAW 264.7
macrophages. J Agric Food Chem. 54:3472–3478. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Na HK, Kim EH, Choi MA, Park JM, Kim DH
and Surh YJ: Diallyl trisulfide induces apoptosis in human breast
cancer cells through ROS-mediated activation of JNK and AP-1.
Biochem Pharmacol. 84:1241–1250. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
You S, Nakanishi E, Kuwata H, Chen J,
Nakasone Y, He X, Liu X, Zhang S, Zhang B and Hou DX: Inhibitory
effects and molecular mechanisms of garlic organosulfur compounds
on the production of inflammatory mediators. Mol Nutr Food Res.
57:2049–2060. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kyung J, Kim D, Park D, Yang YH, Choi EK,
Lee SP, Kim TS, Lee YB and Kim YB: Synergistic anti-inflammatory
effects of Laminaria japonica fucoidan and Cistanche tubulosa
extract. Lab Anim Res. 28:91–97. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lee SH, Kim DW, Eom SA, Jun SY, Park M,
Kim DS, Kwon HJ, Kwon HY, Han KH, Park J, Hwang HS, Eum WS and Choi
SY: Suppression of 12-O-tetradecanoylphorbol-13-acetate
(TPA)-induced skin inflammation in mice by transduced Tat-Annexin
protein. BMB Rep. 45:354–359. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Joh EH and Kim DH: Lancemaside A inhibits
lipopolysac-charide-induced inflammation by targeting LPS/TLR4
complex. J Cell Biochem. 111:865–871. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Aggarwal BB and Natarajan K: Tumor
necrosis factors: developments during the last decade. Eur Cytokine
Netw. 7:93–124. 1996.PubMed/NCBI
|
|
28
|
Brennan FM and McInnes IB: Evidence that
cytokines play a role in rheumatoid arthritis. J Clin Invest.
118:3537–3545. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Blatteis CM, Li S, Li Z, Perlik V and
Feleder C: Signaling the brain in systemic inflammation: the role
of complement. Front Biosci. 9:915–931. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bai AP, Ouyang Q and Hu RW: Diallyl
trisulfide inhibits tumor necrosis factor-alpha expression in
inflammed mucosa of ulcerative colitis. Dig Dis Sci. 50:1426–1431.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lee HJ, Lee HG, Choi KS, Surh YJ and Na
HK: Diallyl trisulfide suppresses dextran sodium sulfate-induced
mouse colitis: NF-κB and STAT3 as potential targets. Biochem
Biophys Res Commun. 437:267–273. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kuo WW, Wang WJ, Tsai CY, Way CL, Hsu HH
and Chen LM: Diallyl trisufide (DATS) suppresses high
glucose-induced cardiomyocyte apoptosis by inhibiting JNK/NFκB
signaling via attenuating ROS generation. Int J Cardiol.
168:270–280. 2013. View Article : Google Scholar
|
|
33
|
Lai KC, Hsu SC, Kuo CL, Yang JS, Ma CY, Lu
HF, Tang NY, Hsia TC, Ho HC and Chung JG: Diallyl sulfide, diallyl
disulfide, and diallyl trisulfide inhibit migration and invasion in
human colon cancer colo 205 cells through the inhibition of matrix
metalloproteinase-2, -7, and -9 expressions. Environ Toxicol.
28:479–488. 2013. View Article : Google Scholar
|
|
34
|
Akira S and Takeda K: Toll-like receptor
signalling. Nat Rev Immunol. 4:499–511. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Fitzgerald KA, Palsson-McDermott EM, Bowie
AG, Jefferies CA, Mansell AS, Brady G, Brint E, Dunne A, Gray P,
Harte MT, McMurray D, Smith DE, Sims JE, Bird TA and O’Neill LA:
Mal (MyD88-adapter-like) is required for Toll-like receptor-4
signal transduction. Nature. 413:78–83. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jawan B, Kao YH, Goto S, Pan MC, Lin YC,
Hsu LW, Nakano T, Lai CY, Sun CK, Cheng YF, Tai MH, Eng HL, Wang
CS, Huang CJ, Lin CR and Chen CL: Propofol pretreatment attenuates
LPS-induced granulocyte-macrophage colony-stimulating factor
production in cultured hepatocytes by suppressing MAPK/ERK activity
and NF-kappaB translocation. Toxicol Appl Pharmacol. 229:362–373.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jung JS, Shin KO, Lee YM, Shin JA, Park
EM, Jeong J, Kim DH, Choi JW and Kim HS: Anti-inflammatory
mechanism of exogenous C2 ceramide in lipopolysaccharide-stimulated
microglia. Biochim Biophys Acta. 1831:1016–1026. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lee JY, Ye J, Gao Z, Youn HS, Lee WH, Zhao
L, Sizemore N and Hwang DH: Reciprocal modulation of Toll-like
receptor-4 signaling pathways involving MyD88 and
phosphati-dylinositol 3-kinase/AKT by saturated and polyunsaturated
fatty acids. J Biol Chem. 278:37041–37051. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Islam MA, Cinar MU, Uddin MJ, Tholen E,
Tesfaye D, Looft C and Schellander K: Expression of Toll-like
receptors and downstream genes in lipopolysaccharide-induced
porcine alveolar macrophages. Vet Immunol Immunopathol. 146:62–73.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Cario E and Podolsky DK: Differential
alteration in intestinal epithelial cell expression of toll-like
receptor 3 (TLR3) and TLR4 in inflammatory bowel disease. Infect
Immun. 68:7010–7017. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ii M, Matsunaga N, Hazeki K, Nakamura K,
Takashima K, Seya T, Hazeki O, Kitazaki T and Iizawa Y: A novel
cyclohexene derivative, ethyl
(6R)-6-[N-(2-Chloro-4-fluorophenyl)sulfamoyl]
cyclohex-1-ene-1-carboxylate (TAK-242), selectively inhibits
toll-like receptor 4-mediated cytokine production through
suppression of intracellular signaling. Mol Pharmacol.
69:1288–1295. 2006. View Article : Google Scholar
|
|
42
|
Takashima K, Matsunaga N, Yoshimatsu M,
Hazeki K, Kaisho T, Uekata M, Hazeki O, Akira S, Iizawa Y and Ii M:
Analysis of binding site for the novel small-molecule TLR4 signal
transduction inhibitor TAK-242 and its therapeutic effect on mouse
sepsis model. Br J Pharmacol. 157:1250–1262. 2009. View Article : Google Scholar : PubMed/NCBI
|