Introduction
High mobility group box 1 (HMGB1) was initially
characterized as a non-histone nucleoprotein; it is an ubiquitously
expressed, highly conserved DNA-binding protein. It regulates gene
transcription, stabilizes the structure of nucleosomes, mediates
inflammation, and facilitates nerve growth and tumor metastasis. It
is localized in the nucleus, but HMGB1 is also found in the
cytoplasm and membrane of various cells (1–4).
Mesenchymal stem cells (MSCs) are one of the primary
stem cell populations involved in fracture repair; their migration
into the fracture microenvironment and osteogenic differentiation
play an important role in healing (5). Previous studies have suggested that
inflammatory factors released by necrotic cells play a significant
role in MSC migration and differentiation in the inflammatory
microenvironment of fractures (6,7).
HMGB1 is released into the extracellular space when it is secreted
from active cells and is passively released from necrotic cells
following a fracture, resulting in a marked increase in the
concentration of HMGB1 outside cells (8).
It is well known that HMGB1 acts as a
chemoattractant for MSCs (9). In
the fracture microenvironment, the expression of almost all HMGB1
receptors is significantly upregulated, including the receptor for
advanced glycation end products (RAGE), Toll-like receptor (TLR)2
and TLR4. RAGE is currently understood to be the main high-affinity
receptor for HMGB1 (10). When
HMGB1 binds to RAGE, the mitogen-activated protein kinase (MAPK)
signaling pathway is activated (11). It has been shown that the p38 MAPK
signaling pathway plays an important role in the osteoblastic
differentiation of MSCs (12).
Moreover, the intracellular nuclear factor-κB (NF-κB) pathway,
mediated by TLR2/4 (10), is
known to facilitate cell proliferation and differentiation
(10,13,14).
To date, it remains unknown as to whether HMGB1
promotes the differentiation of MSCs, and which of the
aforementioned signal transduction pathways may be involved in any
effect of HMGB1 on MSCs. Thus, in the present study, we
investigated the role that extracellular HMGB1 plays in promoting
the differentiation of MSCs. Furthermore, the extracellular
receptors and intracellular signaling pathways that mediate the
promotion of MSC differentiation by HMGB1 were investigated, using
receptor neutralizing antibodies and signaling pathway
inhibitors.
Materials and methods
Reagents
Recombinant human HMGB1 protein was purchased from
Sigma (St. Louis, MO, USA). Graded concentrations of HMGB1, ranging
from 12.5 to 200 ng/ml, were used in the experiments described
below, as serum levels of HMGB1 in septic patients have previously
been reported to be approximately 30–150 ng/ml (15). For RAGE receptor blocking, RAGE
neutralizing antibody (No. Q15109; R&D Systems, Minneapolis,
MN, USA) was used at the concentration of 5 µg/ml, for TLR2
receptor blocking, TLR2 neutralizing antibody (No. O60603; R&D
Systems) was used at the concentration of 0.5 µg/ml and for
TLR4 receptor blocking, TLR4 neutralizing antibody (No. O00206;
R&D Systems) was used at the concentration of 1.5 µg/ml.
p38 inhibitor (SB203580; Cell Signaling Technology, Inc., Danvers,
MA, USA) was used at the concentration of 0.5 µM, and NF-κB
inhibitor (BAY 11-7082; Cell Signaling Technology, Inc.) was used
at the concentration of 0.1 µM. All of the above-mentioned
reagents were used according to the respective manufacturer's
instructions.
Culture and expansion of human bone
marrow-derived MSCs
MSCs were purchased from Cyagen Biosciences, Inc.
(Guangzhou, China). The MSCs were trypsinized and passaged after
the cell confluence reached approximately 80%, and cells at passage
3–5 were used in the experiments described below.
Transwell migration assay
Cell migration was examined using Transwell chambers
(pore size, 8 µm diameter; Corning Costar, Acton, MA, USA).
Complete medium containing 0.1% fetal calf serum (FCS) was added to
the wells of a 24-well plate, and serum-starved 1×105
MSCs were then suspended in a volume of 100 µl complete
medium containing 0.1% FCS were added to the upper chamber. Prior
to the addition of HMGB1, the Transwell plate (with MSCs in the
upper chamber and medium containing 0.1% FCS only in the lower
chamber) was first incubated at 37°C for 1 h. Following the
addition of HMGB1, the plate was incubated at 37°C for 3 h,
followed by membrane fixation with 4% paraformaldehyde and staining
with 0.1% crystal violet (both from Cyagen Biosciences (Guangzhou)
Inc., Guangzhou, China. The membranes were then washed, and the
cells on the underside of the membranes were observed under a light
microscope (Leica DMI LM; Leica Microsystems, Wetzlar, Germany).
Subsequently, the numbers of cells were counted in 5–10 random
fields for each membrane.
Assays for osteogenic
differentiation
In order to induce osteogenic differentiation, the
cells were cultured in standard osteoinductive (OSI) medium
containing 0.1 µM dexamethasone (DEX), 10 mM
glycerophosphate disodium and 0.5 mM ascorbic acid (Sigma), as
previously described (16). In
addition, HMGB1- and RAGE-blocking antibodies, TLR2/4
receptor-blocking antibody and p38 and NF-κB inhibitors in some
instances were utilized in OSI medium in order to observe the
osteoinductive effects of HMGB1.
Total RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
In order to observe MSC differentiation following
exposure to HMGB1, the cells were cultured in osteogenic induction
medium (Cyagen Biosciences, Inc.) or HMGB-1 at 50 ng/ml for 5 days.
Total RNA was extracted using TRIzol reagent (Invitrogen Life
Technologies, Carlsbad, CA, USA) according to the manufacturer's
instructions. RT-qPCR) was carried out in order to observe the
expression of osteoblastic markers. Quantitative PCR (qPCR) was
carried out using a StepOnePlus Real-Time PCR system and SYBR-Green
reagents (Roche Diagnostics, Indianapolis, IN, USA) as a
double-strand DNA-specific binding dye. Primers of the selected
genes selected for PCR reactions were synthesized as shown in
Table I. PCR reactions were
performed in triplicate for each run. The mRNA expression levels of
target genes and the housekeeping gene, GAPDH, were quantified in
separate tubes. The cycling conditions were as follows: 95°C for 30
sec and 40 cycles of 95°C for 5 sec, 60°C for 35 sec. The value of
2−ΔCt represents the relative level of target gene
expression.
 | Table IPrimers used for RT-qPCR. |
Table I
Primers used for RT-qPCR.
| Gene name | Primer |
|---|
| OSTERIX | F:
5′-CCTGCGACTGCCCTAATT-3′ |
| R:
5′-GCGAAGCCTTGCCATACA-3′ |
| BMP-2 | F:
5′-CATGCCATTGTTCAGACG-3′ |
| R:
5′-TGTACTAGCGACACCCAC-3′ |
| ALP | F:
5′-TTGACCTCCTCGGAAGACACTCTG-3′ |
| R:
5′-CGCCTGGTAGTTGTTGTGAGCATAG-3′ |
| RAGE | F:
5′-TGTGTGGCCACCCATTCCA-3′ |
| R:
5′-TGTGTGGCCACCCATTCCA-3′ |
| TLR2 | F:
5′-TGACTCCCAGGAGCTCTTAG-3′ |
| R:
5′-CTTCCTTGGAGAGGCTGATG-3′ |
| TLR4 | F:
5′-CCTGGACCTGAGCTTTAATC-3′ |
| R:
5′-AAAGGCTCCCAGGGCTAAAC-3′ |
| GAPDH | F:
5′-TGACGCTGGGGCTGGCATTG-3′ |
| R:
5′-GGCTGGTGGTCCAGGGGTCT-3′ |
Alkaline phosphatase (ALP) and Alizarin
red staining
The human MSCs (104/cm2) were
seeded into 24-well plates and cultured in osteogenic induction
medium (Cyagen Biosciences, Inc.). After 7 days, ALP activity was
assayed using a BCIP/NBT Alkaline Phosphatase Color Development kit
(Beyotime Biotech, Jiangsu, China). DAPI (Beyotime Biotech) was
used to stain the nuclei. The ALP-positive cells were counted under
a light microscope (Leica DMI LM) in 5 randomly selected fields.
Calcium deposits were detected by staining with 2% Alizarin red (pH
4.2; Sigma). To quantify the number of stained nodules, the stain
was solubilized with 0.5 ml 5% sodium dodecyl sulphate (SDS) in 0.5
N HCl for 30 min at room temperature. The solubilized stain (0.15
ml) was transferred to the wells of a 96-well plate, and the
absorbance was measured at 405 nm. Data are presented as the means
± SD, n=3.
Western blot analysis
The cells were harvested and lysed in RIPA buffer
containing protease inhibitors. After measuring the concentration,
protein samples were separated by 10% SDS-polyacrylamide gel
electrophoresis (SDS-PAGE) and transferred onto polyvinylidene
difluoride (PVDF) membranes (Millipore, Billerica, MA, USA). The
membranes were blocked in 5% skim milk for 1 h and incubated with
antibodies against ALP (sc-98652; Santa Cruz Biotechnology, Inc.,
Santa Cruz, CA, USA), OSTERIX (ab94744; Abcam, Cambridge, MA, USA),
bone morphogenetic protein-2 (BMP-2; sc-6895; Santa Cruz
Biotechnology, Inc.), RAGE (ab3611; Abcam), TLR2 (sc-10739), TLR4
(sc-293072) (both from Santa Cruz Biotechnology, Inc.), p38
(#9212), phosphorylated (p-)p38 (9215) (both from Cell Signaling
Technology, Inc.), NF-κB (sc-372; Santa Cruz Biotechnology, Inc.),
p-NF-κB (#3039; Cell Signaling Technology, Inc.) and β-actin
(sc-130301; Santa Cruz Biotechnology, Inc.) separately at 4°C
overnight. Following incubation with peroxidase-linked secondary
antibodies, immunoreactive proteins were visualized with ECL
reagent (Thermo Fisher Scientific, Inc., Waltham, MA, USA). The
relative quantification of the bands was performed using Image J
software.
Statistical analysis
Statistical significance was determined using a
two-tailed Student's t-test, assuming equal variances. The
Chi-square test was used to compare rates. P-values <0.05, and
<0.01 were considered to indicate statistically significant
differences.
Results
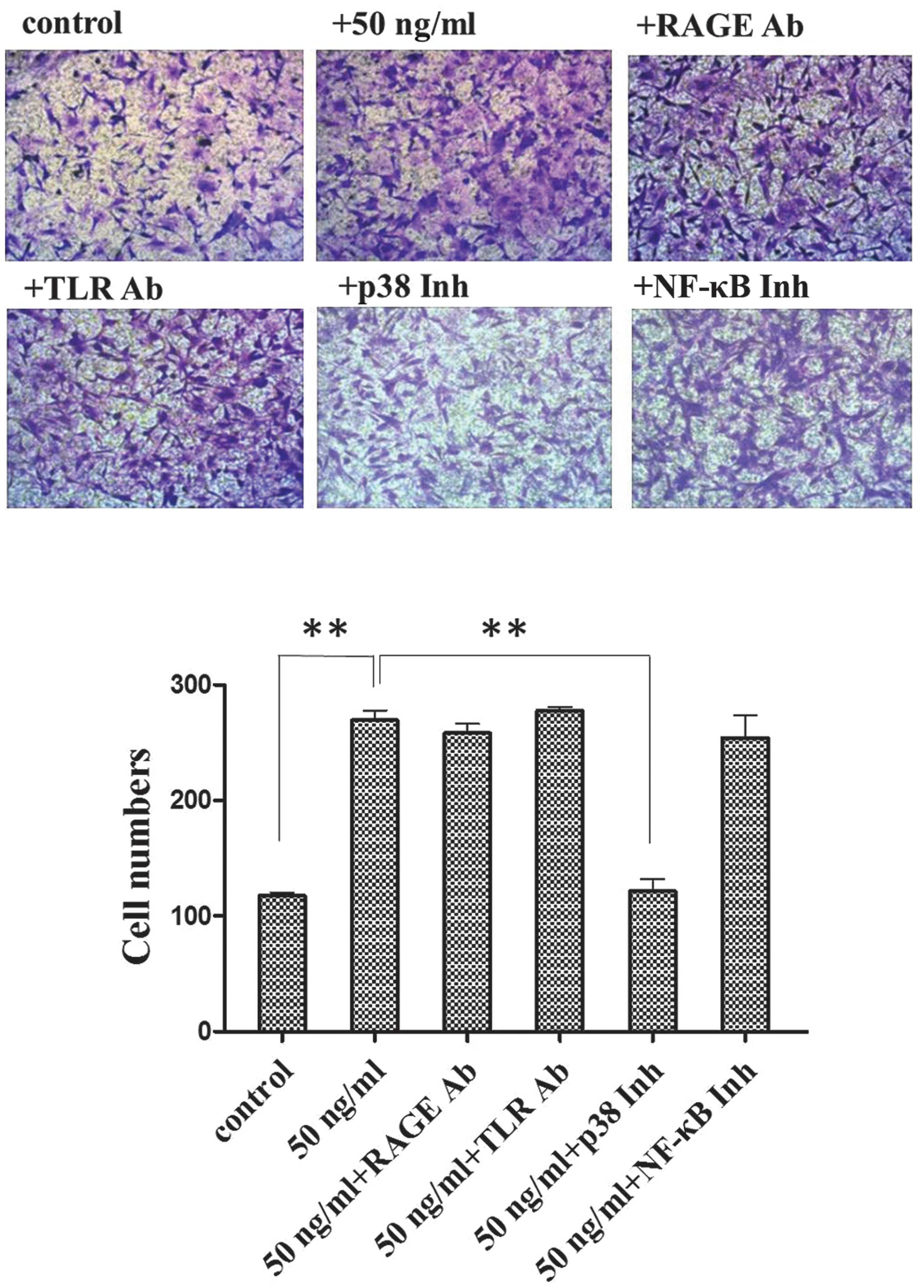
Effects of HMGB1 on the migration of
MSCs
Following treatment with HMGB1 at a concentration of
50 ng/ml, there was a significant increase in the number of
migrating cells compared to the untreated control group (Fig. 1). The cells were treated with
HMGB1 (50 ng/ml) and either RAGE neutralizing antibody (Fig. 1; 50 ng/ml + RAGE Ab), TLR2/4
neutralizing antibody (Fig. 1; 50
ng/ml + TLR Ab), or NF-κB inhibitor (Fig. 1; 50 ng/ml + NF-κB Inh). In each
case, there was a significant increase in cell migration compared
to the untreated control group; however, the difference was not
significant compared to the cells treated with HMGB1 alone. There
was a decrease in the number of migrating cells in the p38
inhibitor-treated group (Fig. 1;
50 ng/ml + p38 Inh) compared to the population of cells treated
with HMGB1 alone.
Effects of HMGB1 on the osteoblastic
differentiation of MSCs
Alizarin red staining and quantitative analysis
revealed that HMGB1 (12.5 to 50 ng/ml) significantly promoted the
formation of calcified nodules in the MSCs. Moreover, ALP staining
of the MSCs (7 days) demonstrated that HMGB1 (12.5 to 50 ng/ml)
significantly stimulated the synthesis of ALP in the MSCs (Fig. 2).
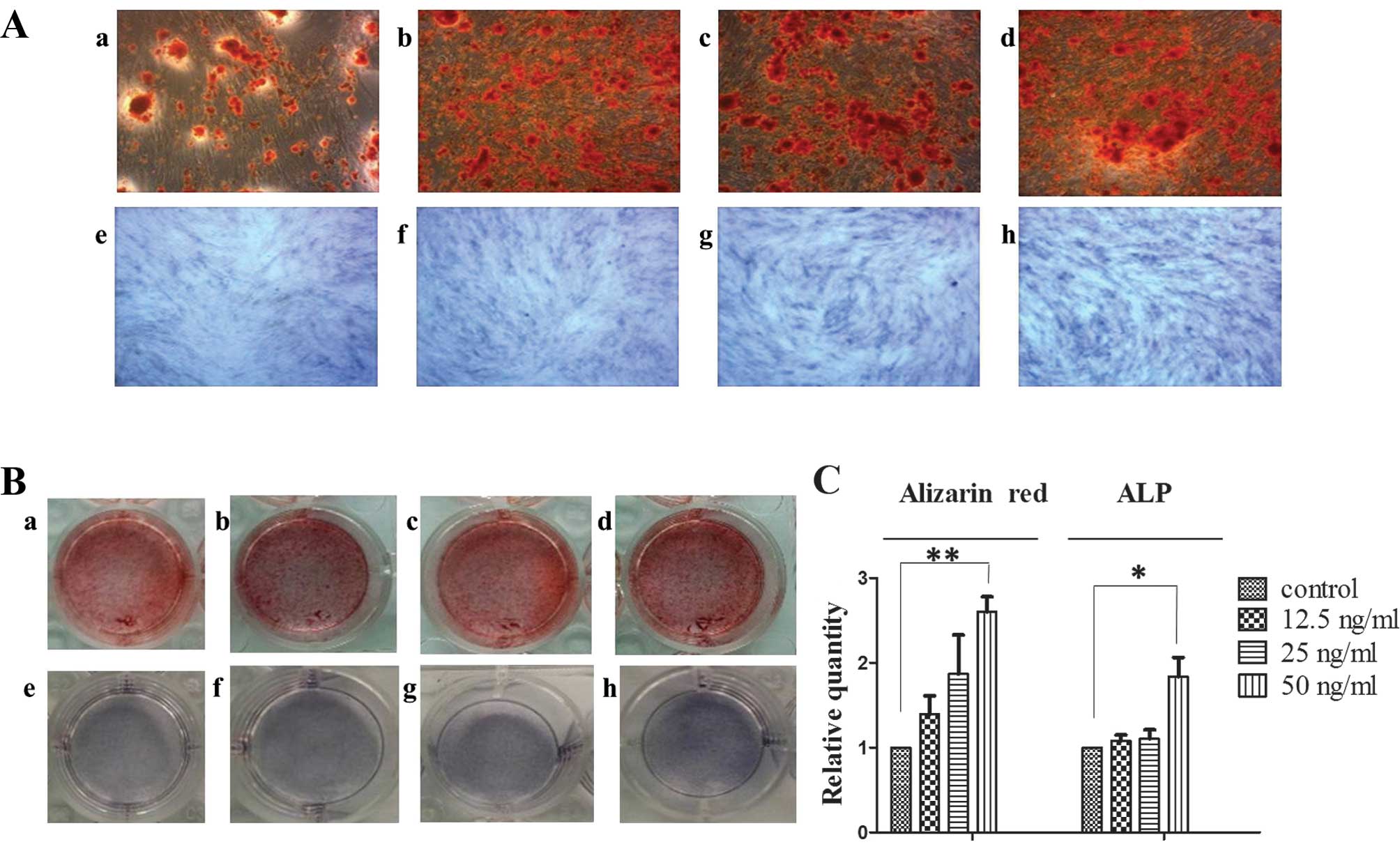 | Figure 2High mobility group box 1 (HMGB1)
promotes the formation of calcified nodules and alkaline
phosphatase (ALP) activity. Mesenchymal stem cells (MSCs) were
maintained in standard osteoinductive (OSI) complete medium alone
(A and B, panel a) or with HMGB1 (A and B, panels b–d: 12.5, 25 and
50 ng/ml, respectively) for 14 days. Cells were stained with
Alizarin red (A and B, panels a–d). MSCs were maintained in OSI
complete medium alone (A and B, panel e) or with HMGB1 (A and B,
panels f–h: 12.5, 25 and 50 ng/ml, respectively) for 7 days. Cells
were stained with Alizarin red (A and B, panels a–d) or with ALP (A
and B, panels e–h). The number of stained nodules and ALP activity
in MSCs treated with graded concentrations of HMGB1, or OSI medium
alone was quantified. (C) Results were obtained from 3 independent
experiments and are expressed as the means ± SD (n=3 in each
experiment). *P<0.05 and **P<0.01. |
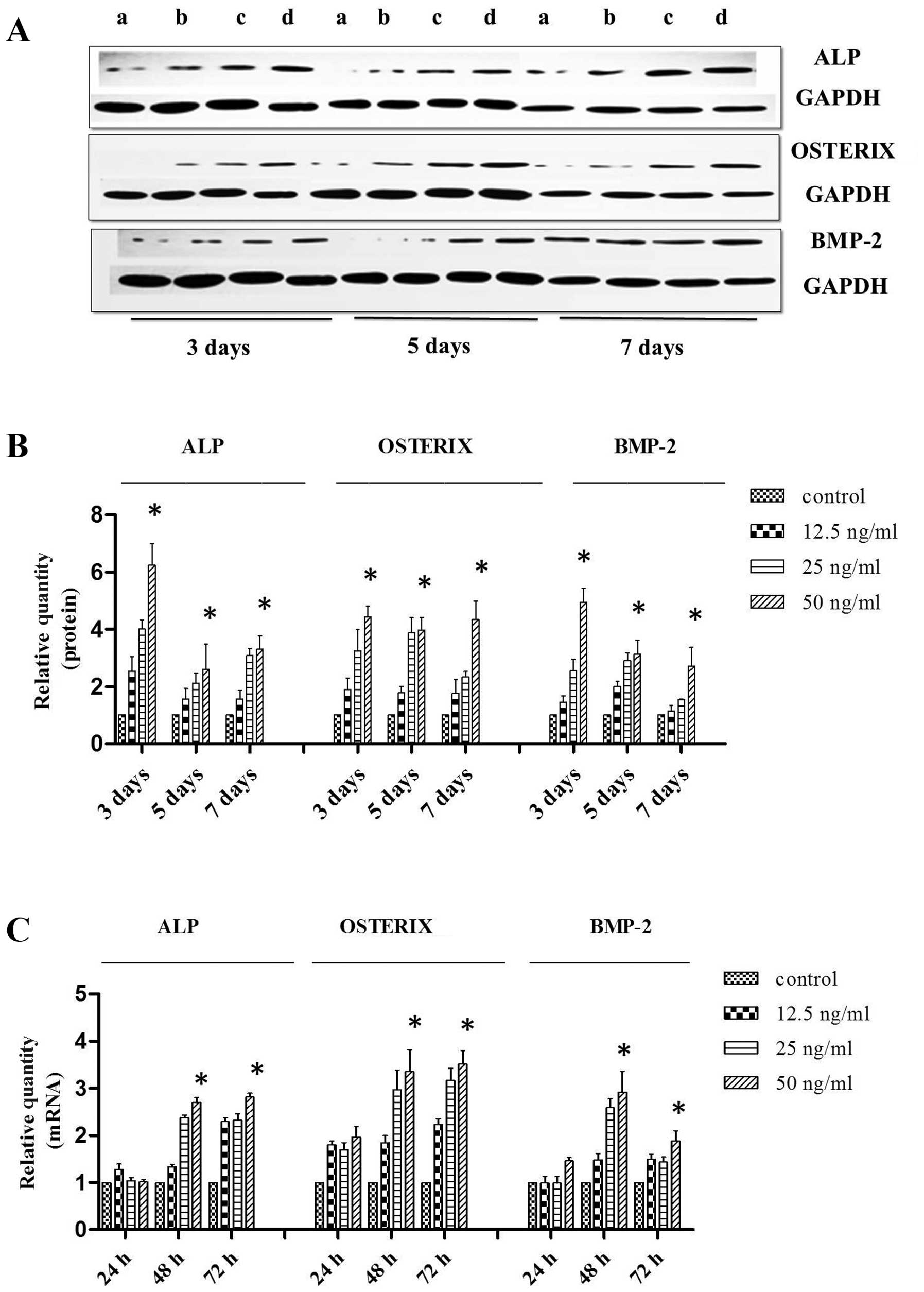
Effects of HMGB1 on the mRNA and protein
expression levels of ALP, OSTERIX and BMP-2 in MSCs
The MSCs were maintained in OSI complete medium
alone (Fig. 3A, lane a) or with
HMGB1 (Fig. 3A, lanes b–d: 12.5,
25 and 50 ng/ml, respectively) for 3, 5 and 7 days. Western blot
analysis was used to determine the extent of osteoblastic
differentiation (Fig. 3B). Our
results revealed that treatment with HMGB1 (12.5 to 50 ng/ml)
markedly increased the protein expression levels of OSTERIX, ALP
and BMP-2 during the osteoblastic differentiation of MSCs. The
results of RT-qPCR (24, 48 and 72 h), which was used to detect the
expression of the corresponding mRNA transcripts, also revealed the
upregulation of gene expression in response to treatment with HMGB1
(Fig. 3C).
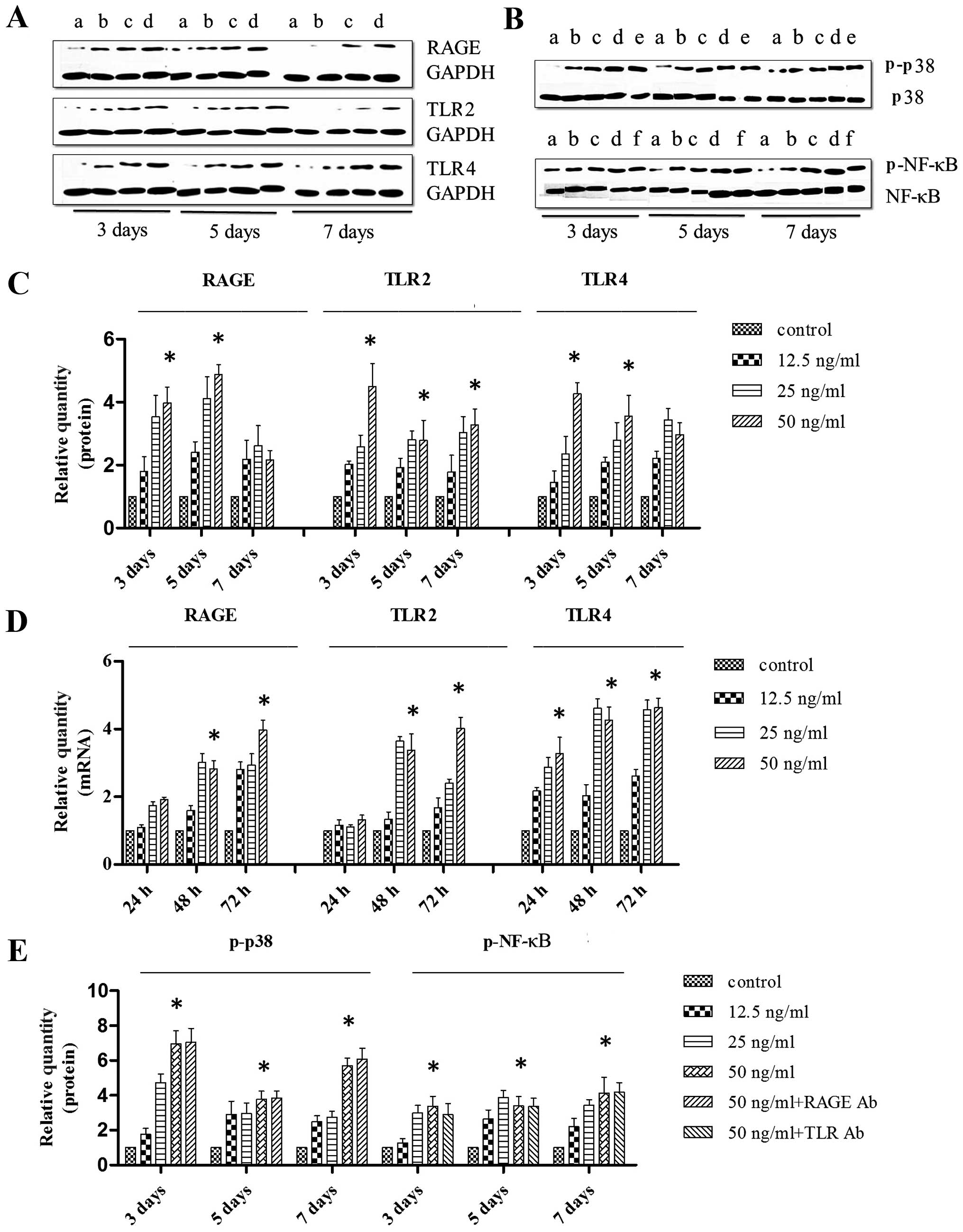
Effects of HMGB1 on the mRNA and protein
expression levels of RAGE, TLR2/4, and mediators of the p38 MAPK
and NF-κB signaling pathways in MSCs
The MSCs were maintained in OSI complete medium
alone (Fig. 4A, lane a) or with
HMGB1 (Fig. 4A, lanes b–d: 12.5,
25 and 50 ng/ml, respectively) for 3, 5 and 7 days. Western blot
analysis was used to determine the protein expression levels of
RAGE and TLR2/4 (Fig. 4C). Our
results revealed that treatment with HMGB1 (12.5 andto 50 ng/ml)
markedly increased the expression of RAGE and TLR2/4 during the
osteoblastic differentiation of MSCs. The results of RT-qPCR (24,
48 and 72 h), which was used to detect expression of the
corresponding mRNA transcripts, also revealed the upregulation of
gene expression in response to treatment with HMGB1 (Fig. 4D).
The MSCs were maintained in OSI complete medium
alone (Fig. 4B, lane a) or with
HMGB1 (Fig 4B, lanes b–d: 12.5,
25 and 50 ng/ml, respectively). The MSCs were also maintained in
OSI complete medium with HMGB1 (50 ng/ml) and RAGE neutralizing
antibody (Fig. 4B, lane e) or TLR
2/4 neutralizing antibody (Fig.
4B, lane f). There was a dose-dependent increase in the
expression of p-p38 and p-NF-κB in response to HMGB1 treatment.
Treatment with the RAGE neutralizing antibody and the TLR2/4
neutralizing antibody had no significant effect on the
HMGB1-induced upregulation of p-p38 and p-NF-κB (Fig. 4E).
Effects of neutralizing antibodies or
inhibitors on the HMGB1-induced osteoblastic differentiation of
MSCs
Statistical analysis indicated that treatment of the
MSCs with the TLR2/4 neutralizing antibody or p38 inhibitors
attenuated the formation of calcified nodules (Fig. 5A and B, panels a–f). Similar
results were observed following ALP staining (7 days) (Fig. 5A and B, panels g–l). However,
treatment of the MSCs with NF-κB inhibitors did not significantly
inhibit the formation of calcified nodules or ALP activity compared
to the cells treated with 50 ng/ml HMGB1 alone (Fig. 5).
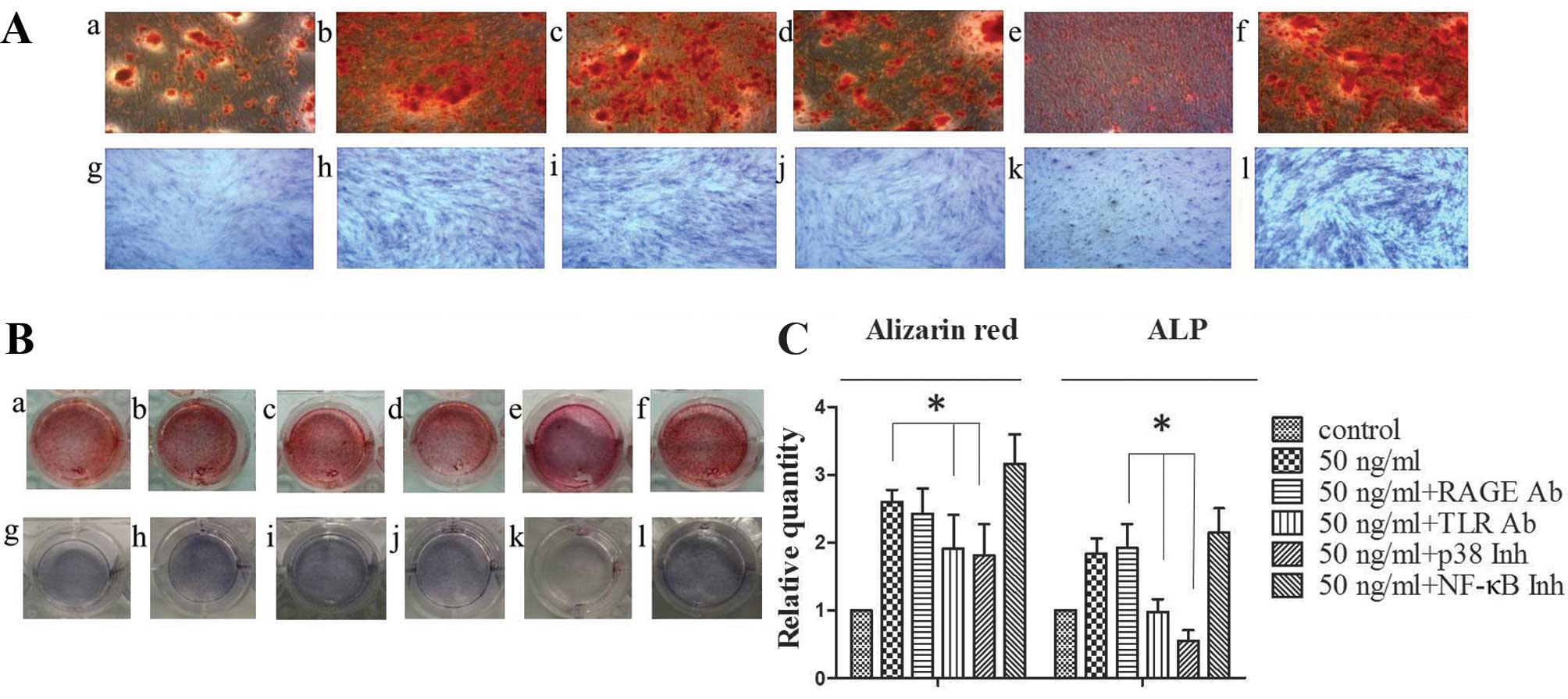 | Figure 5Toll-like receptor (TLR) 2/4
neutralizing antibody or p38 inhibitors inhibit the high mobility
group box 1 (HMGB1)-induced osteoblastic differentiation of
mesenchymal stem cells (MSCs). MSCs were maintained in standard
osteoinductive (OSI) complete medium alone (A and B, panels a and
g) or with HMGB1 (50 ng/ml) (A and B, panels b and h). MSCs were
also maintained in OSI complete medium with HMGB1 (50 ng/ml) and
receptor for advanced glycation end products (RAGE) neutralizing
antibody (A and B, panels c and i) TLR2/4 neutralizing antibody (A
and B, panels d and j), p38 inhibitors (A and B, panels e and k) or
nuclear factor-κB (NF-κB) inhibitors (A and B, panels f and l).
Cells were treated with alizarin red stain for 14 days (A and B,
panels a–f) or stained with alkaline phosphatase (ALP) stain for 7
days (A and B, panels g–l). The number of stained nodules and ALP
activity in MSCs treated with or without neutralizing antibody or
inhibitors were quantified (C). Results were obtained from 3
independent experiments and are expressed as the means ± SD (n=3 in
each experiment). Ab, antibody; Inh, inhibitor.
*P<0.05. |
Effects of neutralizing antibodies or
inhibitors on the HMGB1-induced upregulation of ALP, OSTERIX and
BMP-2 mRNA and protein expression in MSCs
Treatment of the MSCs with RAGE neutralizing
antibody and 50 ng/ml HMGB1 significantly altered the expression of
OSTERIX, ALP and BMP-2 compared to the untreated control group, but
not compared to the cells treated with HMGB1 alone. Treatment of
the MSCs with TLR2/4 neutralizing antibody or p38 inhibitor
resulted in a marked decrease in expression compared to the cells
treated with 50 ng/ml HMGB1 alone. By contrast, the cells treated
with the NF-κB inhibitor exhibited a marked increase in expression
compared to the cells treated with 50 ng/ml HMGB1 (Fig. 6).
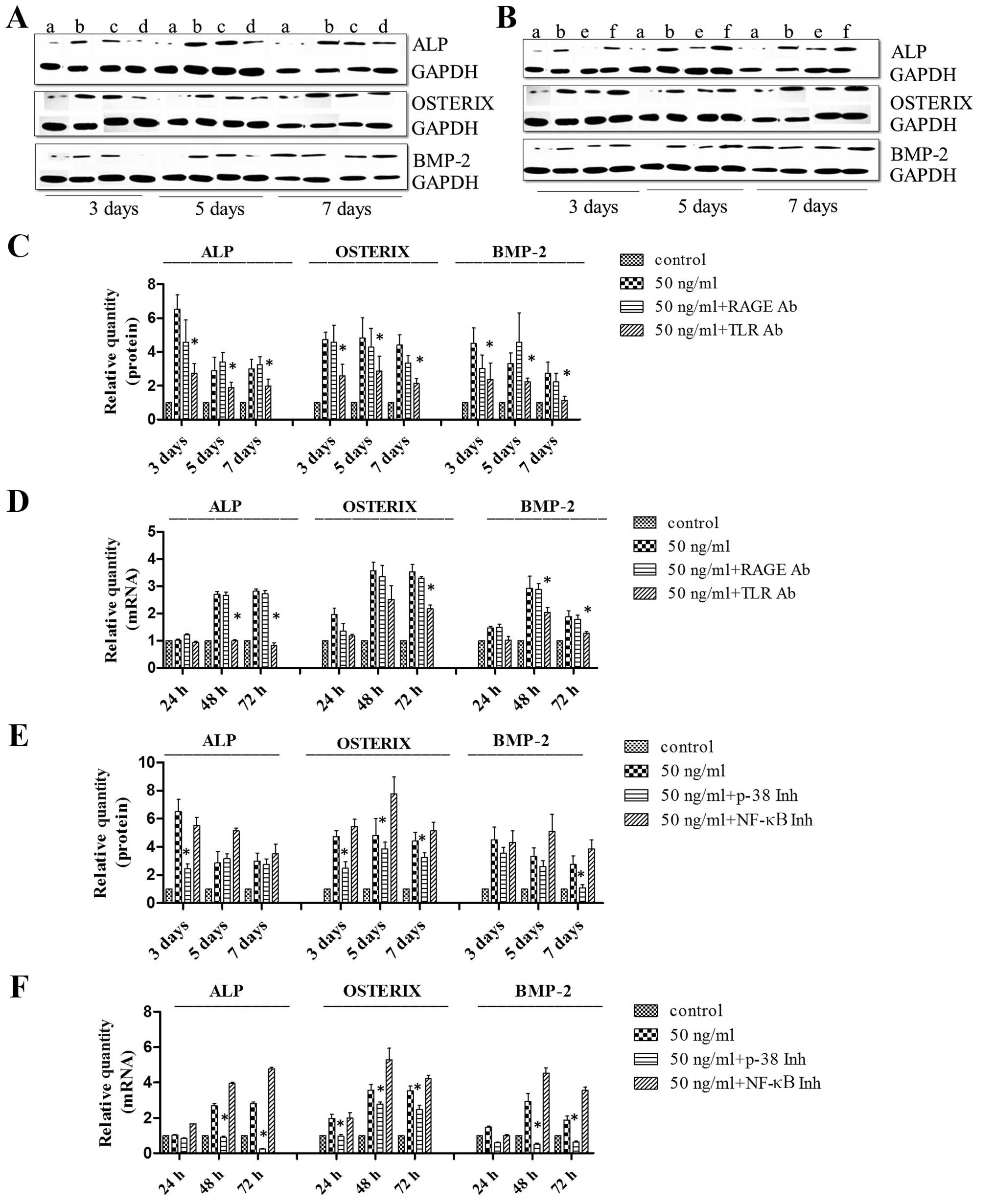 | Figure 6Toll-like receptor (TLR2/4)
neutralizing antibody or p38 inhibitors inhibit the high mobility
group box 1 (HMGB1)-induced upregulation of OSTERIX, alkaline
phosphatase (ALP) and bone morphogenetic protein 2 (BMP-2) protein
expression detected by western blot analysis and RT-qPCR.
Mesenchymal stem cells (MSCs) were maintained in standard
osteoinductive (OSI) complete medium alone (A and B, lane a) or
with HMGB1 (50 ng/ml) (A and B, lane b). MSCs were also maintained
in OSI complete medium with HMGB1 (50 ng/ml) and receptor for
advanced glycation end products (RAGE) neutralizing antibody (A,
lane c), TLR2/4 neutralizing antibody (A, lane d), p38 inhibitors
(B, lane e) or nuclear factor-κB (NF-κB) inhibitors (B, lane f).
The degree of osteoblastic differentiation was determined using
western blot analysis (C and E) and RT-qPCR (D and F) to detect the
protein and mRNA expression levels of ALP, OSTERIX and BMP-2 in
MSCs. Results were obtained from 3 independent experiments and are
expressed as the means ± SD (n=3 in each experiment). Ab, antibody;
Inh, inhibitor. *P<0.05. |
Discussion
HMGB1 is a widely expressed and multifunctional
inflammatory factor, the biological characteristics of which have
been studied in depth. It is expressed in almost all adult tissues,
and is involved in the development, maintenance and repair of
connective tissue, in processes such as vasculogenesis and
angiogenesis (1,4,8,17),
neurite outgrowth (18),
hematopoietic stem cell proliferation (19), embryonic development (20), and tumorigenesis (21). Notably, MSCs suppress both innate
and adaptive immune responses (22). In the present study, we
demonstrated that HMGB1 significantly promoted the migration of
MSCs through the p38 MAPK signaling pathway. Additionally, HMGB1
promoted the osteoblastic differentiation of MSCs, and increased
the gene and protein expression levels of RAGE and TLR2/4, which
bind with HMGB1. The downstream signaling pathways p38 MAPK and
NF-κB have previously been shown to regulate cell proliferation and
differentiation (10–14). In this study, we found that these
signaling pathways were activated. We therefore hypothesized that
HMGB1 acts through RAGE and TLR 2/4, activating the p38 MAPK and
NF-κB signaling pathways, and thus promoting the osteoblastic
differentiation of MSCs.
In this study, treatment with a neutralizing
antibody against RAGE had no significant effect on MSC
differentiation following treatment with HMGB1, or the activation
of the p38 MAPK signaling pathway. Moreover, blockade of the p38
MAPK signaling pathway resulted in a marked decrease in the
osteoblastic differentiation of MSCs. The inhibition of the TLR 2/4
receptor attenuated the HMGB1-induced MSC differentiation, but had
no significant effect on NF-κB signaling pathway activation. The
NF-κB inhibitor had no significant effect on HMGB1-induced MSC
differentiation.
Taken together, the results of the present study
suggest that HMGB1 promotes MSC differentiation by activating the
TLR2/4 receptor and p38 MAPK signaling pathway. Given that the p38
MAPK signaling pathway is not activated through the RAGE receptor,
this result was to be expected. The RAGE receptor primarily
mediates the effects of immunosuppressors on immune cell activation
and promotes the synthesis of cytokines (23). In the inflammatory
microenvironment of a fracture, the RAGE receptor is synthesized in
large quantities and is bound to HMGB1. Its main function is likely
associated with the activation of other signaling pathways to
promote inflammation (9), rather
than the signaling pathways tested in this study. Given that the
target of the NF-κB inhibitor (BAY 11-7082) is IκB, the data may
indicate that HMGB1 induces NF-κB activation prior to IκB
phosphorylation. In the present study, we did not examine the
effects of HMGB1 on IκB kinase activity, and thus the increased
activity of IκB kinase caused by HMGB1 may be likely the mechanism.
This will be our next research focus.
Certain studies have described the role of these
receptors in the regulation of MSCs by HMGB1 (10,11). In the present study we
demonstrated that activation of the p38 MAPK signaling pathway is
one of the mechanisms through which HMGB1 promotes MSC migration.
The c-Jun N-terminal kinase (JNK) and extracellular
signal-regulated kinase (ERK) signaling pathways, which are also
members of the MAPK family, are both known to significantly
influence cell migration (24).
However, whether these two pathways are involved in the mechanisms
through which HMGB1 promotes MSC migration remains to be
determined. In the present study, we demonstrted that the
activation of the TLR2/4 receptor and the p38 MAPK signaling
pathway is one of the mechanisms through which HMGB1 induces MSC
differentiation. Although this study demonstrated that the p38 MAPK
signaling pathway was not activated by the RAGE receptor, we
hypothesized that there may be another type of HMGB1 receptor that
activates this signaling pathway, or perhaps HMGB1 may activate
this pathway through signaling crosstalk. This is a worthy topic
for future research. On the other hand, previous research has shown
that the activation of TLR2/4 promotes the synthesis of interleukin
(IL)-6 (25), which is also
capable of regulating cell proliferation and differentiation
(26). Thus, the role of IL-6
should be a key focus of subsequent research. Previous research has
also shown that TLRs can inhibit stem cell differentiation
(27). This inhibition likely
results from the activation of different TLRs activating NF-κB or
IL-6, depending on the cell line and culture conditions used, but
the actual mechanisms remain to be elucidated. Increasing evidence
suggests that the Wnt signaling pathway is important in the
regulation of the differentiation of various stem cell populations
(28). Therefore, a potential
future research could focus on whether HMGB1 influences the Wnt
signaling pathway, and whether there is an HMGB1 receptor that
activates this signaling pathway, which may be involved in the
differentiation of MSCs.
Acknowledgments
This study was supported by a grant from the
National Natural Science Foundation of China (Nos. 81271973 and
81201397), the Zhejiang Provincial Natural Science Foundation of
China (No. LY15H060001; No. LY15H060002 and No. LY13H060002) and
Zhejiang Medical and Health Science and Technology Plan Project
(No. 2011ZDA011).
References
|
1
|
Degryse B, Bonaldi T, Scaffidi P, Müller
S, Resnati M, Sanvito F, Arrigoni G and Bianchi ME: The high
mobility group (HMG) boxes of the nuclear protein HMG1 induce
chemotaxis and cytoskeleton reorganization in rat smooth muscle
cells. J Cell Biol. 152:1197–1206. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Palumbo R, Sampaolesi M, De Marchis F,
Tonlorenzi R, Colombetti S, Mondino A, Cossu G and Bianchi ME:
Extracellular HMGB1, a signal of tissue damage, induces
mesoangioblast migration and proliferation. J Cell Biol.
164:441–449. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Andersson U, Erlandsson-Harris H, Yang H
and Tracey KJ: HMGB1 as a DNA-binding cytokine. J Leukoc Biol.
72:1084–1091. 2002.PubMed/NCBI
|
|
4
|
Yang H, Wang H, Czura CJ and Tracey KJ:
HMGB1 as a cytokine and therapeutic target. J Endotoxin Res.
8:469–472. 2002. View Article : Google Scholar
|
|
5
|
Granero-Moltó F, Weis JA, Miga MI, Landis
B, Myers TJ, O'Rear L, Longobardi L, Jansen ED, Mortlock DP and
Spagnoli A: Regenerative effects of transplanted mesenchymal stem
cells in fracture healing. Stem Cells. 27:1887–1898. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Glass GE, Chan JK, Freidin A, Feldmann M,
Horwood NJ and Nanchahal J: TNF-alpha promotes fracture repair by
augmenting the recruitment and differentiation of muscle-derived
stromal cells. Proc Natl Acad Sci USA. 108:1585–1590. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Marsell R and Einhorn TA: The biology of
fracture healing. Injury. 42:551–555. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Naglova H and Bucova M: HMGB1 and its
physiological and pathological roles. Bratisl Lek Listy.
113:163–171. 2012.PubMed/NCBI
|
|
9
|
Meng E, Guo Z, Wang H, Jin J, Wang J, Wang
H, Wu C and Wang L: High mobility group box 1 protein inhibits the
proliferation of human mesenchymal stem cells and promotes their
migration and differentiation along osteoblastic pathway. Stem
Cells Dev. 17:805–813. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Park JS, Svetkauskaite D, He Q, Kim JY,
Strassheim D, Ishizaka A and Abraham E: Involvement of toll-like
receptors 2 and 4 in cellular activation by high mobility group box
1 protein. J Biol Chem. 279:7370–7377. 2004. View Article : Google Scholar
|
|
11
|
Nakajima Y, Inagaki Y, Kido J and Nagata
T: Advanced glycation end products increase expression of S100A8
and A9 via RAGE-MAPK in rat dental pulp cells. Oral Dis.
21:328–334. 2015. View Article : Google Scholar
|
|
12
|
Hu Y, Chan E, Wang SX and Li B: Activation
of p38 mitogen-activated protein kinase is required for osteoblast
differentiation. Endocrinology. 144:2068–2074. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chang CP, Su YC, Lee PH and Lei HY:
Targeting NFKB by autophagy to polarize hepatoma-associated
macrophage differentiation. Autophagy. 9:619–621. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Delhalle S, Blasius R, Dicato M and
Diederich M: A beginner's guide to NF-kappaB signaling pathways.
Ann N Y Acad Sci. 1030:1–13. 2004. View Article : Google Scholar
|
|
15
|
Wang H, Vishnubhakat JM, Bloom O, Zhang M,
Ombrellino M, Sama A and Tracey KJ: Proinflammatory cytokines
(tumor necrosis factor and interleukin 1) stimulate release of high
mobility group protein-1 by pituicytes. Surgery. 126:389–392. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guo Z, Li H, Li X, Yu X, Wang H, Tang P
and Mao N: In vitro characteristics and in vivo immunosuppressive
activity of compact bone-derived murine mesenchymal progenitor
cells. Stem Cells. 24:992–1000. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yuk JM, Yang CS, Shin DM, Kim KK, Lee SK,
Song YJ, Lee HM, Cho CH, Jeon BH and Jo EK: A dual regulatory role
of apurinic/apyrimidinic endonuclease 1/redox factor-1 in
HMGB1-induced inflammatory responses. Antioxid Redox Signal.
11:575–588. 2009. View Article : Google Scholar
|
|
18
|
Kim JB, Sig Choi J, Yu YM, Nam K, Piao CS,
Kim SW, Lee MH, Han PL, Park JS and Lee JK: HMGB1, a novel
cytokine-like mediator linking acute neuronal death and delayed
neuroinflammation in the postischemic brain. J Neurosci.
26:6413–6421. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang XB, Chen X, Song KD, Wang J, Yao W,
Liu HL and Sun ZM: Effects of HMGB1 on human cord blood CD34(+)
hematopoietic stem cells proliferation and differentiation in
vitro. Zhonghua Xue Ye Xue Za Zhi. 31:88–91. 2010.In Chinese.
PubMed/NCBI
|
|
20
|
Polanská E, Dobšáková Z, Dvořáčková M,
Fajkus J and Štros M: HMGB1 gene knockout in mouse embryonic
fibroblasts results in reduced telomerase activity and telomere
dysfunction. Chromosoma. 121:419–431. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tang Q, Li J, Zhu H, Li P, Zou Z and Xiao
Y: Hmgb1-IL-23-IL-17-IL-6-Stat3 axis promotes tumor growth in
murine models of melanoma. Mediators Inflamm. 2013:7138592013.
View Article : Google Scholar
|
|
22
|
Bianchi ME and Manfredi AA: High-mobility
group box 1 (HMGB1) protein at the crossroads between innate and
adaptive immunity. Immunol Rev. 220:35–46. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sun W, Jiao Y, Cui B, Gao X, Xia Y and
Zhao Y: Immune complexes activate human endothelium involving the
cell-signaling HMGB1-RAGE axis in the pathogenesis of lupus
vasculitis. Lab Invest. 93:626–638. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang M, Sun L, Wang X, Chen S, Jia Q, Liu
N, Chen Y, Kong Y, Zhang L and Zhang AL: Activin B promotes
BM-MSC-mediated cutaneous wound healing by regulating cell
migration via the JNK-ERK signaling pathway. Cell Transplant.
23:1061–1073. 2014. View Article : Google Scholar
|
|
25
|
Akira S, Hirano T, Taga T and Kishimoto T:
Biology of multifunctional cytokines: IL 6 and related molecules
(IL 1 and TNF). FASEB J. 4:2860–2867. 1990.PubMed/NCBI
|
|
26
|
Schilling D, Thomas K, Nixdorff K, Vogel
SN and Fenton MJ: Toll-like receptor 4 and Toll-IL-1 receptor
domain-containing adapter protein (TIRAP)/myeloid differentiation
protein 88 adapter-like (Mal) contribute to maximal IL-6 expression
in macrophages. J Immunol. 169:5874–5880. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Pevsner-Fischer M, Morad V, Cohen-Sfady M,
Rousso-Noori L, Zanin-Zhorov A, Cohen S, Cohen IR and Zipori D:
Toll-like receptors and their ligands control mesenchymal stem cell
functions. Blood. 109:1422–1432. 2007. View Article : Google Scholar
|
|
28
|
Liu Y, Han D, Wang L and Feng H:
Down-regulation of Wnt10a affects odontogenesis and proliferation
in mesenchymal cells. Biochem Biophys Res Commun. 434:717–721.
2013. View Article : Google Scholar : PubMed/NCBI
|