Introduction
Plant polyphenols are natural compounds that are
widely distributed in plants and have been reported to possess
antioxidant, antitumor and antiaging properties (1). However, the potential application of
plant polyphenols is rather limited, due to their low
bioavailability caused by poor cell membrane penetration (2). It was recently reported that
acylation enhances biological activity, possibly due to the
improvement of membrane penetration (3). Acetylated
(−)-epigallocate-chin-3-O-gallate, isovitexin and isoorientin
exhibited greater lipophilicity compared with their precursors
(4). The enzymatic acylation of
isoquercitrin and isorhamnetin-3-O-glucoside has been found to
improve their antiproliferative effect on Caco-2 cancer cells
(5,6). Additionally, the acetylated
quercetin ester exhibited marked antiproliferative activity against
human cervical cancer cells and murine fibroblast NIH-3T3 cells
(7).
Arbutin (hydroquinone-O-β-D-glucopyranoside), a
natural polyphenol isolated from the bearberry plant
Arctostaphylos uvaursi, has been traditionally used as a
whitening agent (8,9). Arbutin has been found to reduce the
melanin content and tyrosinase activity in cultured human
melanocytes, B16 murine melanoma cells and HMV-II cells (10–14). It has been reported that certain
arbutin derivatives exert more prominent inhibitory effects on
melanin content and tyrosinase activity compared with those of
arbutin (12,15,16). Furthermore, arbutin has been
confirmed to exert a pro-apoptotic effect on human bladder cancer
TCCSUP and human melanoma A375 cells (17,18). However, the effects of arbutin and
its acetylated derivative on B16 murine melanoma cells have not yet
been reported.
Apoptosis is a complex programmed cell death,
manifesting as cell shrinkage, chromatin condensation and
internucleosomal DNA fragmentation (19). Extrinsic and intrinsic pathways
play important roles in cellular functions, and mitochondrial
function is considered to be a therapeutic target for cancer
treatment (20).
In the present study, acetylated arbutin was
prepared in order to improve the biological effects of arbutin, and
the effects of arbutin and its acetylated derivative on melanin
synthesis, tyrosinase activity and apoptosis of B16 murine melanoma
cells were investigated. Flow cytometry was used to detect the
effects of arbutin and acetylated arbutin on cell cycle and
apoptosis, and the wound healing assay was used to measure cell
migration ability. The potential underlying mechanism was
investigated by western blot analysis and fluorescence
microscopy.
Materials and methods
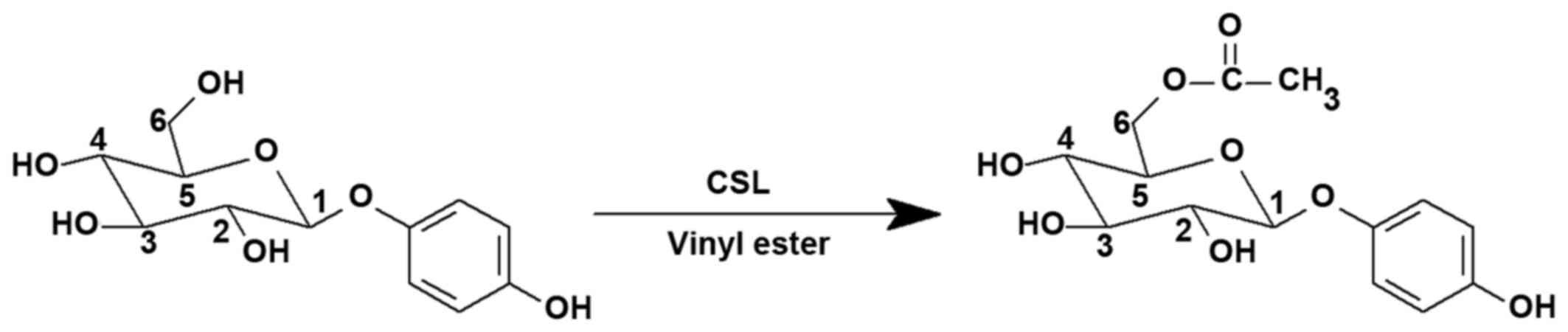
Synthesis of acetylated arbutin
Arbutin was purchased from Wuhan Fude Chemical Co.,
Ltd. (Wuhan, China), and its acetylated derivative was first
synthesized by our laboratory (Fig.
1) (21). The reaction
conditions were as follows: Arbutin (0.73 mmol), vinyl acetate (30
mmol), Candida sp. (CSL) (100 mg) and tetrahydrofuran (THF;
20 ml) were incubated with shaking (100 rpm) at 40°C for 24 h
[water activity (aw) = 0.63]. The nuclear magnetic resonance (NMR)
analysis indicated that the C-6′ position in the glucose moiety of
arbutin was modified. The purity (>99%) was confirmed by
high-performance liquid chromatography.
Cell culture
The murine melanoma cells B16 (American Type Culture
Collection, Manassas, VA, USA) were cultured in complete RPMI-1640
medium with 10% (v/v) inactivated fetal bovine serum, penicillin G
and streptomycin (100 mg/l) at 37°C in 5% CO2. All the
agents for cell culture were obtained from Gibco, Thermo Fisher
Scientific (Grand Island, NY, USA).
Cell viability assay
A total of 5×103 cells per well were
plated onto 96-well plates, and various doses of arbutin and its
acetylated derivative were added into each well, stimulating the
cells for 24, 48 or 72 h. A total of 20 µl of
3-(4,5-dimethylthi-azol-2-yl)-2, 5-diphenyltetrazoliumbromide (MTT;
Amresco, Solon, OH, USA) solution (5 mg/ml) was added to each well
and then removed after a 4-h incubation at 37°C in the dark. Purple
formazan crystals were dissolved in 150 µl dimethylsulfoxide
(DMSO) and quantitatively detected by measuring the absorbance at
540 nm using a microplate reader (F200 pro; Bio-Rad, Hercules, CA,
USA). Each test was performed in quadruplicate, and each experiment
was repeated twice.
Melanin content assay
The melanin content was measured as previously
described, with minor modifications (22). B16 cells were seeded at a density
of 5×105 cells per well in 6-well plates and incubated
for 24 h. After treatment with arbutin and its acetylated
derivative for 48 h, the cells were washed with phosphate-buffered
saline (PBS) and incubated with 1 M NaOH containing 1% DMSO at 37°C
for 1 h. After dilution with 400 µl distilled water, the
absorbance at 490 nm was measured by a UV-2700 spectrophotometer
(Shimadzu Corp., Kyoto, Japan).
Tyrosinase activity assay
Tyrosinase activity was measured based on a
previously described method using L-DOPA as a substrate (23). A total of 5×105 cells
were seeded into 6-well plates and incubated for 24 h, and then
treated with arbutin and its acetylated derivative. After 48 h, the
cells were washed with PBS and lysed with 1% Triton X-100 (200
µl). Cells were disrupted by repeated freezing and thawing,
and then 100 µl of the mixture and 50 µl 0.5% L-DOPA
were added into a 96-well plate at 37°C for 3 h. The absorbance was
measured at 490 nm by a spectrophotometer (Shimadzu Corp.).
Cell cycle and apoptosis analysis
Cell cycle and apoptosis were determined by flow
cytometry (24). A total of
5×105 cells were seeded into 6-well plates and then
treated with arbutin and its acetylated derivative. For cell cycle
analysis, after 24 h, the cells were harvested, fixed in ice-cold
70% ethanol and then stored at 4°C overnight. The cells were washed
twice with PBS, suspended in 0.5 ml of cold solution containing 25
g/ml RNase A and 50 g/ml propidium iodide (PI; Sigma-Aldrich, Merck
KGaA, St. Louis, MO, USA), and incubated at 37°C for an additional
30 min in the dark. For cell apoptosis analysis, the cells were
harvested, washed with cold PBS twice, and resuspended in binding
buffer according to the manufacturer's protocol. Cells were then
incubated with 5 µl Annexin V-fluorescein isothiocyanate
(FITC) and 10 µl PI (50 µg/ml)(Becton Dickinson,
Miami, FL, USA) mixed and incubated at room temperature for 10 min
in the dark. The cell cycle and apoptosis were determined by
analyzing 15,000 ungated cells using FACSCalibur (BD Biosciences,
Mountain View, CA, USA) and Cell Quest software
(Becton-Dickinson).
Migration assay
The wound healing assay was performed to evaluate
the effect of arbutin and its acetylated derivative on the
migration of B16 cells (25). A
total of 5×105 cells were seeded into a 6-well plate and
incubated for 24 h. The cell monolayer was subjected to a
mechanical scratch wound using a syringe needle, and then treated
with arbutin and its acetylated derivative for 48 h. Digitized
images of the wound area were captured with BioTek Cytation™ 3 Cell
Imaging Multi-Mode Reader (BioTek Instruments Inc., Winooski, VT,
USA). The width of the wound was expressed as a percentage of the
control group.
Assessment of mitochondrial membrane
potential (MMP)
Cells were incubated with arbutin and its acetylated
derivative for 24 h, and then incubated with 2 µM
5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolylcarbocyanine
iodide (JC-1; Sigma-Aldrich, Merck KGaA) at 37°C for 5 min in the
dark. Following three washes with PBS, the changes in fluorescent
color were examined using a fluorescence microscope (magnification,
x20; CCD camera, TE2000; Nikon, Tokyo, Japan). The mean ratio of
red (590 nm) to green (540 nm) fluorescent intensity of each cell
in different groups was calculated by Image J software (National
Institutes of Health, Bethesda, WA, USA). The values of the treated
cells were expressed as a percentage of those from the
corresponding control cells.
Western blot analysis
The cells were seeded into 6-well plates at a
density of 5×105 cells per well and treated with arbutin
and its acetylated derivative for 24 h. Treated cells were
harvested, washed with cold PBS and lysed with RIPA buffer
containing 1% protease inhibitor cocktail and 2%
phenylmethanesulfonyl fluoride (all from Sigma-Aldrich, Merck
KGaA). Protein concentration was measured using a bicinchoninic
acid protein assay kit (Bio-Rad). A total of 30 µl protein
was separated using a 10–12% SDS-PAGE gel and transferred
electrophoretically onto PVDF membranes. The transferred membranes
were blocked in 5% bovine serum albumin for 4 h, and then blotted
with the following primary antibodies at 4°C overnight at dilution
of 1:1,000: B-cell lymphoma (Bcl)-2, Bcl-xL and GAPDH (all from
Cell Signaling Technology, Inc., Danvers, MA, USA). Binding was
detected using an enhanced chemiluminescence kit (GE Healthcare,
Buckinghamshire, UK). The intensity of the bands was quantified by
Image J software (National Institutes of Health).
Statistical analysis
Data were collected from at least three individual
experiments for each assay and are presented as means ± standard
deviation. The difference between two independent samples was
analyzed by a one-way analysis of variance to detect statistical
significance followed by post-hoc multiple comparisons (Dunn's
test) using SPSS 16.0 software (IBM corp., Armonk, NY, USA).
P<0.05 was considered to indicate statistically significant
differences.
Results
Antiproliferative activity of arbutin and
acetylated arbutin
Arbutin and acetylated arbutin inhibited the
viability of B16 murine melanoma cells in a time- and
dose-dependent manner (P<0.05; Fig. 2). The 72 h IC50 of the
acetylated arbutin was ~3.85 mM, which exhibited a higher efficacy
in terms of cell proliferation compared with arbutin. Acetylated
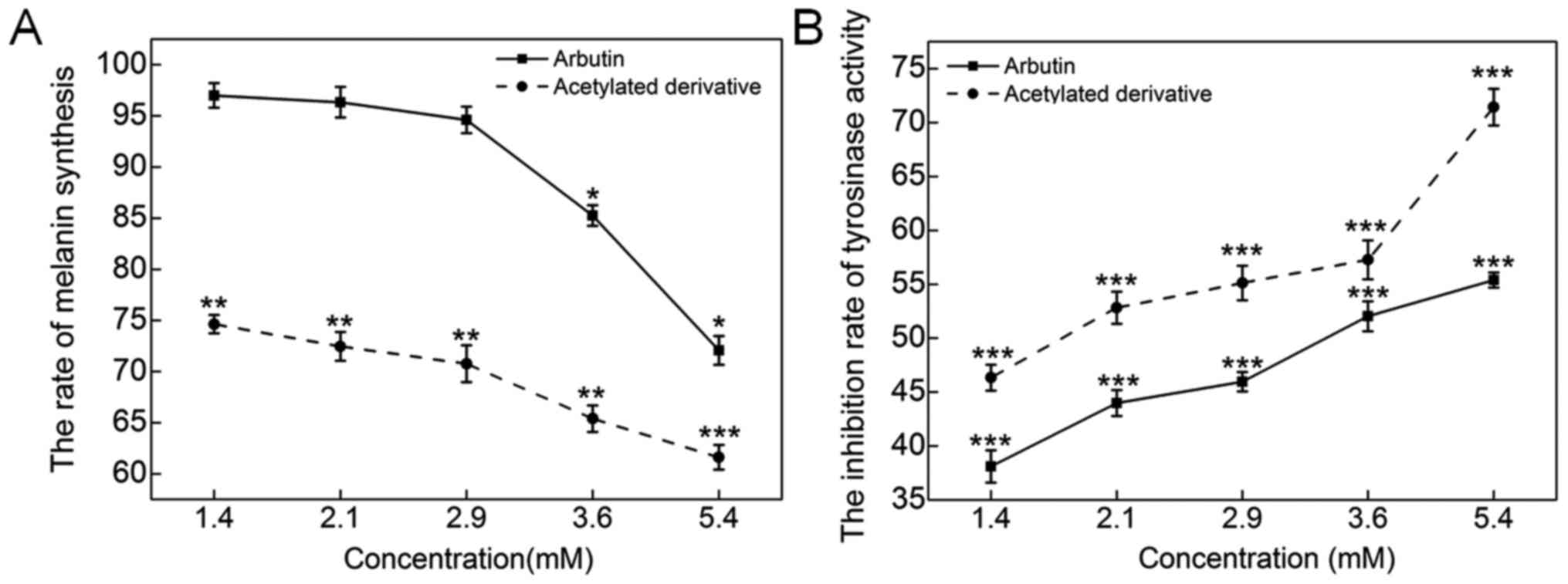
arbutin significantly inhibited melanogenesis up to 89.9% compared
with the 45.8% inhibition by arbutin at 5.4 mM (P<0.05; Fig. 3A). Tyrosinase is known to be the
key enzyme in the process of melanin synthesis. Arbutin and
acetylated arbutin at 5.4 mM suppressed tyrosinase activity by 55.4
and 71.4%, respectively, after a 48-h incubation (P<0.01;
Fig. 3B).
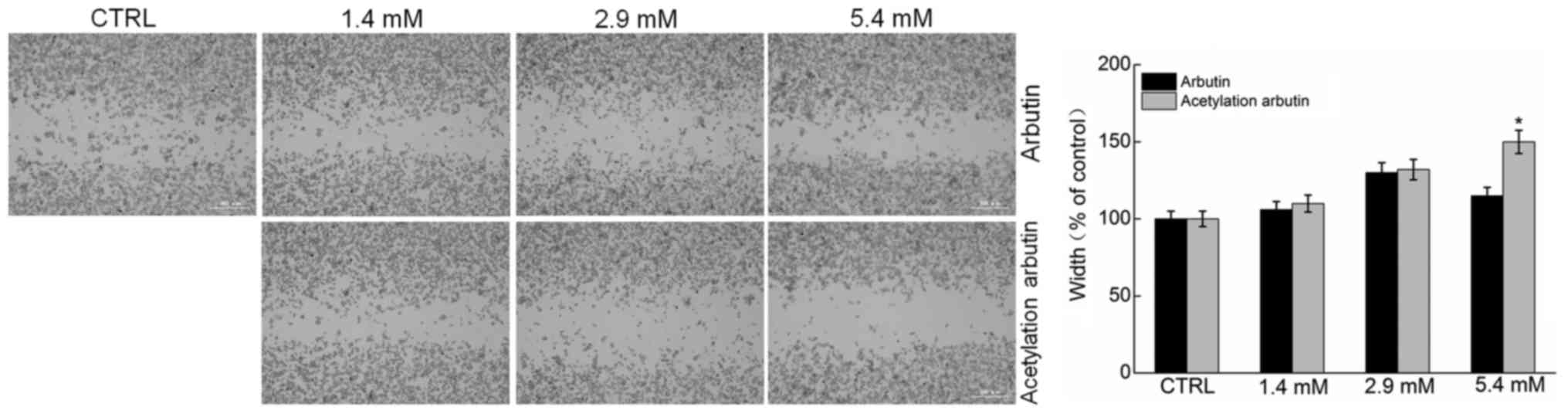
The wound healing assay is considered to be an
effective method for analysing cell migration (24). Comparatively, at the selected
dose, only acetylated arbutin significantly suppressed the
migration ability of B16 cells (P<0.05; Fig. 4). Therefore, at the same dose,
acetylated arbutin achieved a better effect compared with
arbutin.
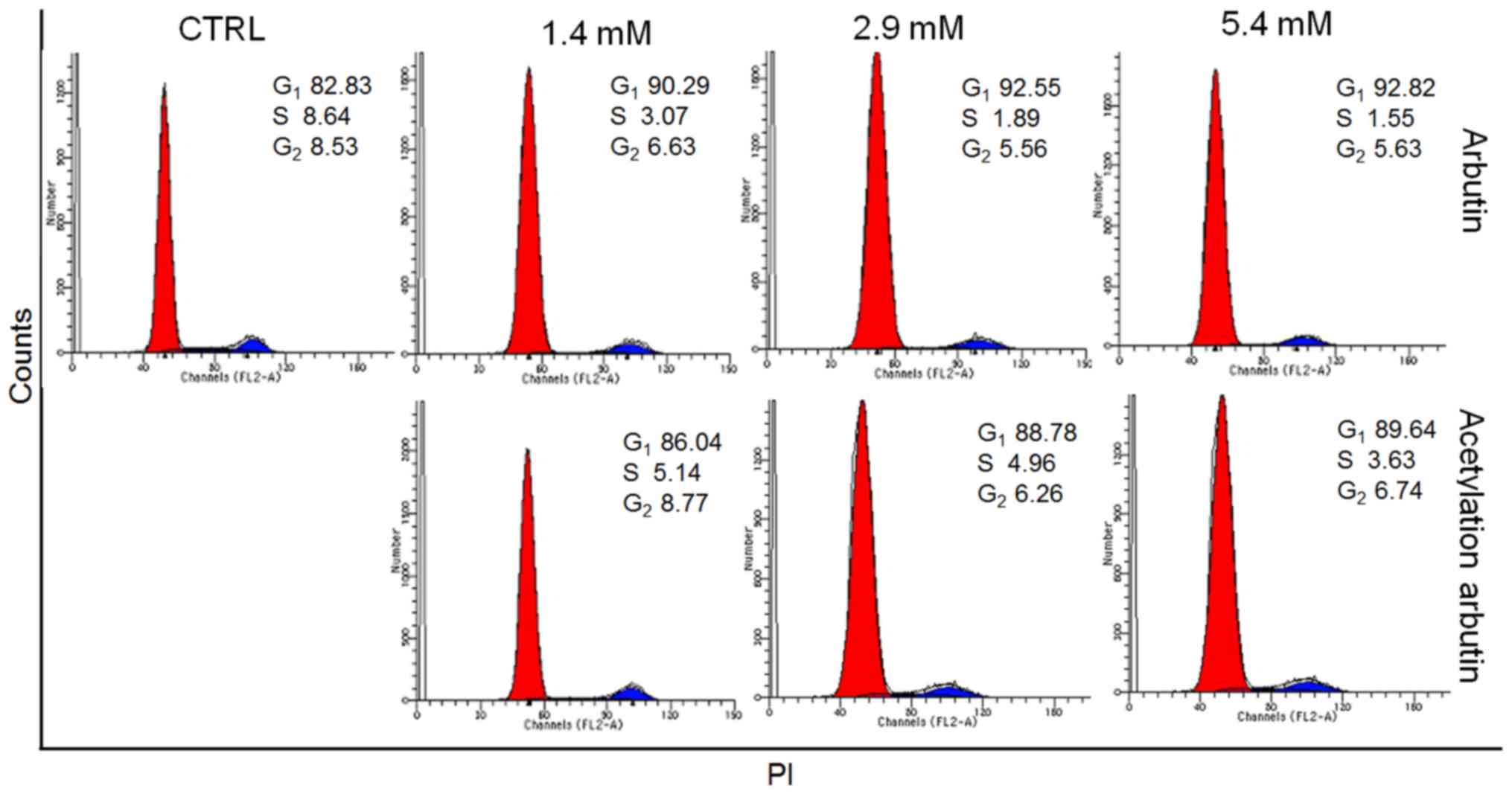
Arbutin and acetylated arbutin caused increased G1
cell cycle arrest compared with control cells (92.8 and 89.6 vs.
82.8%, respectively) in B16 murine melanoma cells after 24 h of
treatment, indicating their inhibitory activity on cell growth
(Fig. 5).
Pro-apoptotic effects of arbutin and
acetylated arbutin
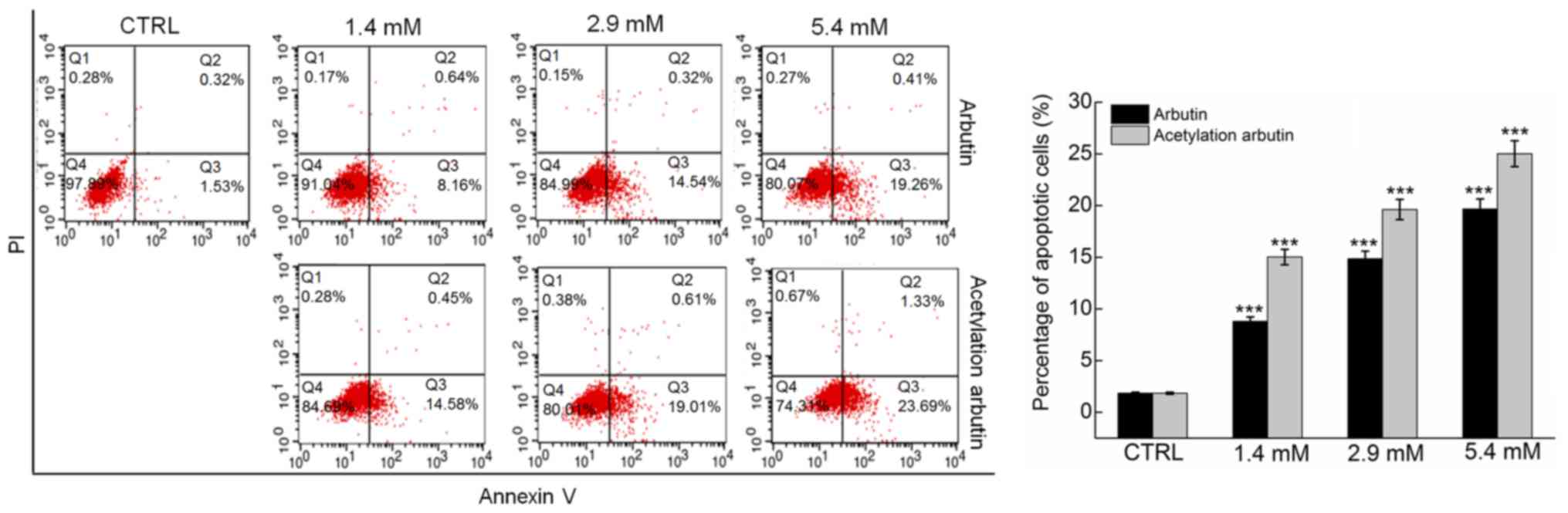
The apoptosis rate following arbutin and acetylated
arbutin treatment was detected by flow cytometry using Annexin
V-FITC labeling for the detection of phosphatidylserine
externalization, occurring as an early step during apoptosis.
Compared with control cells (1.5% of early apoptosis), arbutin and
acetylated arbutin caused apoptosis in 19.3 and 23.7% of the cells,
respectively, after 24 h of treatment at 5.4 mM (P<0.001;
Fig. 6).
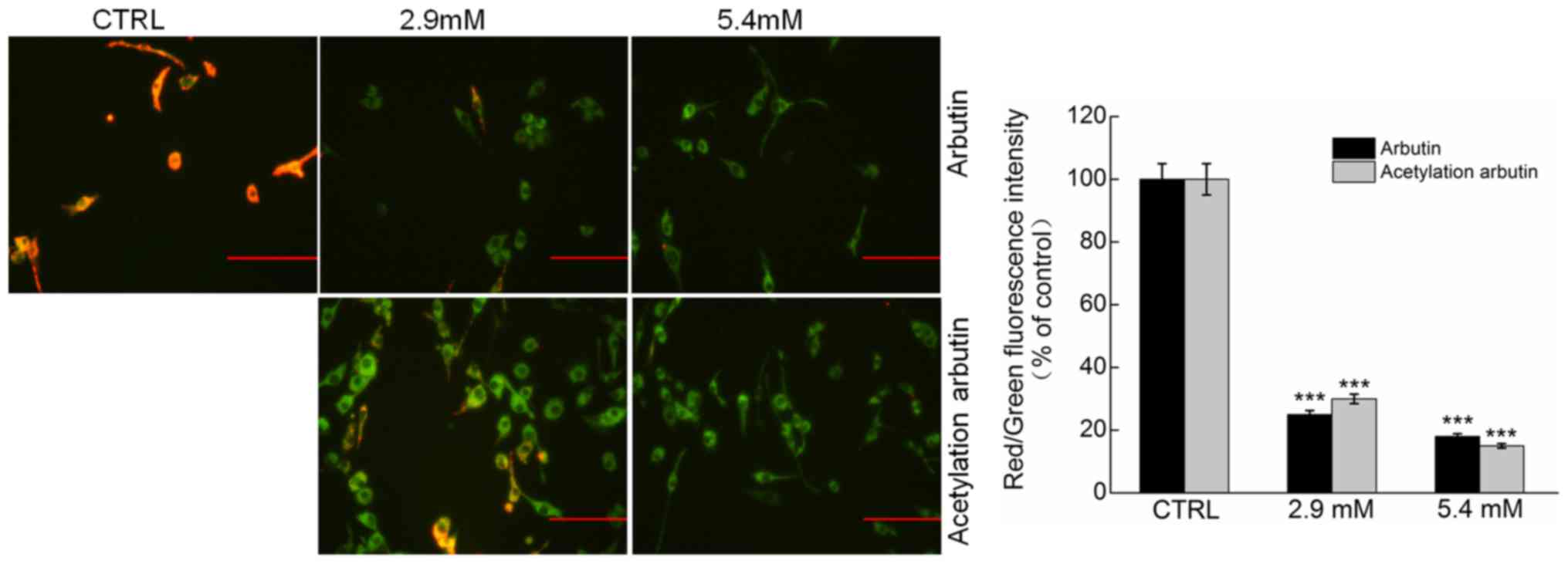
Arbutin and acetylated arbutin cause
mitochondrial dysfunction
Mitochondria play a crucial role in regulating the
intrinsic and extrinsic apoptotic pathways. Arbutin and acetylated
arbutin caused marked MMP loss in B16 murine melanoma cells, as
indicated by the augmentation in green fluorescence and the
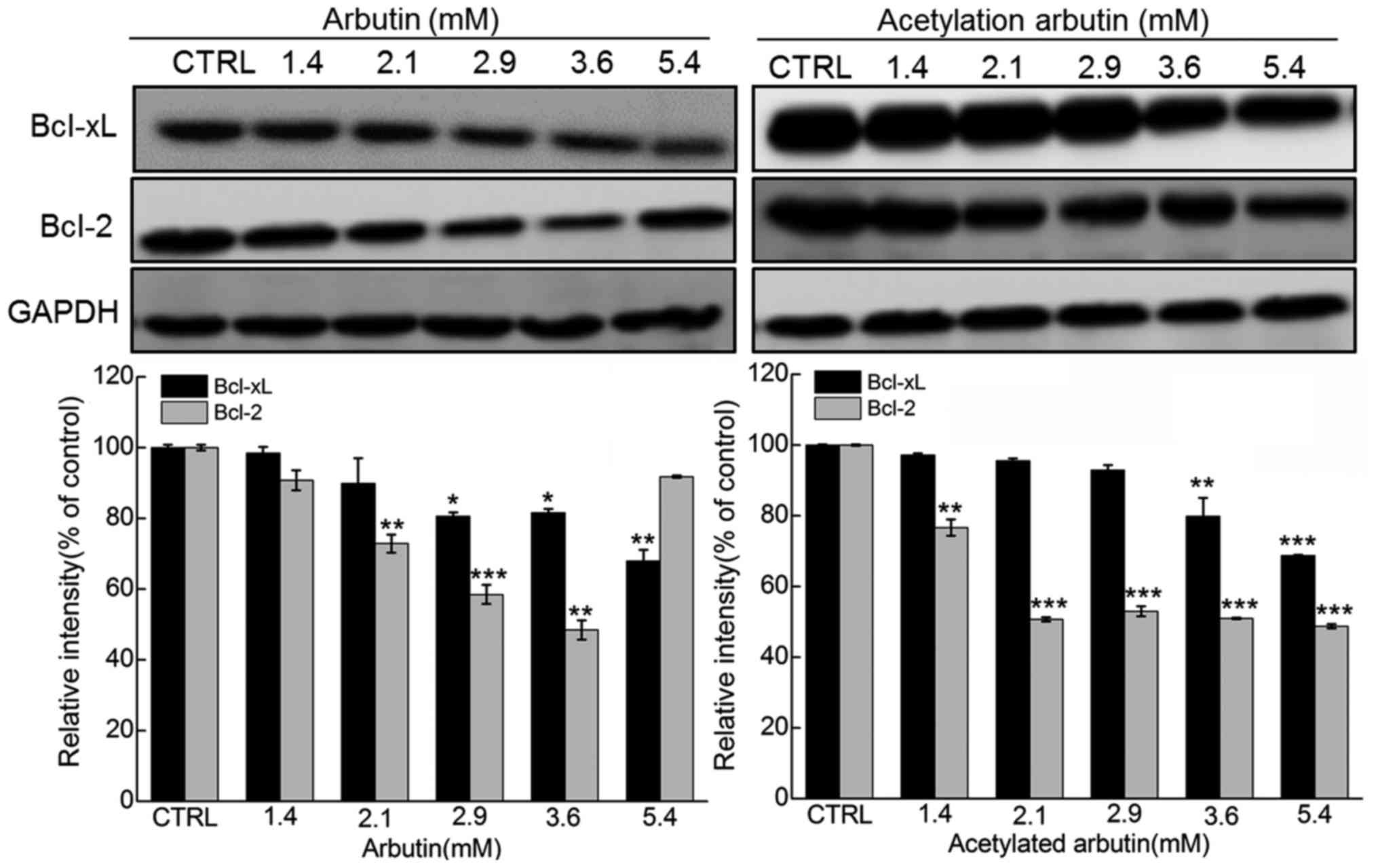
reduction in red fluorescence (P<0.001; Fig. 7). Moreover, a striking reduction
in Bcl-2 and Bcl-xL expression was noted in arbutin- and acetylated
arbutin-treated cells (P<0.05; Fig. 8).
Discussion
The accumulation of melanin pigment has become a
major cosmetic concern in humans. Arbutin, a naturally occurring
hydroquinone that has a similar structure to the substrate that
causes inhibition of tyrosinase, has been widely used in Japan
(26,27). Tyrosinase is a multifunctional
copper-containing polyphenol oxidative enzyme, considered to be a
key enzyme in melanin biosynthesis and melanization in animals. The
inhibition of tyrosinase activity is an important target for the
treatment of pigmentation disorders (28). Several derivatives of arbutin have
been synthesized, including deoxyarbutin, in an attempt to overcome
the limitation of penetration through the skin (29). Acetylated derivatives of arbutin
exhibit higher antimelanogenetic and antityrosinase activity
compared with the parent compound. An arbutin derivative caused a
50% higher inhibition of tyrosinase activity, compared with free
arbutin at the same concentration (30). Following incubation with arbutin
6′-undecenoate, melanin production decreased by ~70% in B16
melanoma cells compared with that in control cells (31). In the present study, the
acetylated arbutin derivative exhibited a higher efficacy in terms
of tyrosinase activity inhibition and reduction of melanin
production compared with arbutin, which may be associated with the
increased solubility in oil-based systems and the improvement of
membrane penetration (10-12).
Arbutin has also been reported to exert inhibitory
effects on the proliferation of cancer cells, including A375 human
malignant melanoma cells (17)
and HCT-15 human TCCSUP cells (18). Our study demonstrated that arbutin
and its acetylated derivative reduced cell viability, inhibited
cell migration, enhanced apoptotic rate, caused G1 phase arrest and
induced mitochondrial disruption in B16 melanoma cells.
Accumulating evidence has confirmed the mitochondrial role in cell
apoptosis (32), which may serve
as a target for cancer treatment. The mitochondrial pathway is
regulated by the Bcl-2 family of anti-apoptotic proteins, including
Bcl-2 and Bcl-xL, and the pro-apoptotic proteins (33). The anti-apoptotic proteins
regulate apoptosis by blocking the release of mitochondrial
molecules, whereas imbalances in the expression of Bcl-2 family
proteins, particularly the Bcl-2 and Bax proteins, lead to
permeabilization of the mitochondrial outer membrane followed by
release of proteins, such as cytochrome c, from mitochondria to the
cytoplasm, which may play an important role in subsequent cell
death (34). Arbutin and its
acetylated derivative decreased the expression of Bcl-2 and Bcl-xL,
indicating that the mitochondrial apoptotic pathway may be involved
in this pro-apoptotic effect.
It has been reported that oxidative stress induces
apoptosis through DNA damage and mitochondrial dysfunction
(35,36). Arbutin exerts strong antioxidant
effects, comparable or even superior to that of its precursor,
hydroquinone, as skin-lightening agent. Arbutin protects U973 cells
from radiation-induced apoptosis by decreasing intracellular
hydroxyl radical production (37). Although our data indicated that
the mitochondrial apoptotic pathway is responsible for arbutin and
its acetylated derivative causing cell apoptosis, the precise role
of the oxidative system requires further investigation.
In summary, arbutin and its acetylated derivative
markedly reducing melanin content, suppressed tyrosinase activity
and induced apoptosis in B16 melanoma cells, which may be mediated
through modulation on the mitochondrial apoptotic pathway. It was
also revealed that acetylated arbutin exhibited a higher efficacy
compared with that of parent arbutin. Additionally, the control of
melanin formation is crucial for the treatment of abnormal skin
pigmentation, which may increase the risk of malignant melanoma.
Tyrosinase is a key enzyme in melanin biosynthesis and its
expression is closely correlated with melanogenesis (28). Therefore, arbutin and acetylated
arbutin may be potential candidates not only for skin whitening,
but also for the treatment of malignant melanoma.
Acknowledgments
The present study was supported by grants from the
National Natural Science Foundation of China (nos. 21172093,
31070708, 81402955 and 21072075), the Special Fund for Basic
Scientific Research of Jilin University (nos. 450060326007 and
450060491559), the Scientific Research Project of Liaoning
Educational Committee (no. L2012431), and the Natural Science
Foundation of Jilin Province in China (nos. 20140101141JC and
201115038).
References
|
1
|
Yazaki K, Sasaki K and Tsurumaru Y:
Prenylation of aromatic compounds, a key diversification of plant
secondary metabolites. Phytochemistry. 70:1739–1745. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang RL, Li N, Li RF, Smith TJ and Zong
MH: A highly regioselective route to arbutin esters by immobilized
lipase from Penicillium expansum. Bioresour Technol. 101:1–5. 2010.
View Article : Google Scholar
|
|
3
|
Trier S, Linderoth L, Bjerregaard S,
Andresen TL and Rahbek UL: Acylation of Glucagon-like peptide-2:
Interaction with lipid membranes and in vitro intestinal
permeability. PLoS One. 9:e1099392014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ma X, Yan R, Yu S, Lu Y, Li Z and Lu H:
Enzymatic acylation of isoorientin and isovitexin from bamboo-leaf
extracts with fatty acids and antiradical activity of the acylated
derivatives. J Agric Food Chem. 60:10844–10849. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Salem JH, Humeau C, Chevalot I,
Harscoat-Schiavo C, Vanderesse R, Blanchard F and Fick M: Effect of
acyl donor chain length on isoquercitrin acylation and biological
activities of corresponding esters. Process Biochem. 45:382–389.
2010. View Article : Google Scholar
|
|
6
|
Salem JH, Chevalot I, Harscoat-Schiavo C,
Paris C, Fick M and Humeau C: Biological activities of flavonoids
from Nitraria retusa (Forssk.) Asch and their acylated derivatives.
Food Chem. 124:486–494. 2011. View Article : Google Scholar
|
|
7
|
Danihelová M, Veverka M, Sturdík E and
Jantová S: Antioxidant action and cytotoxicity on HeLa and NIH-3T3
cells of new quercetin derivatives. Interdiscip Toxicol. 6:209–216.
2013. View Article : Google Scholar
|
|
8
|
Sakuma K, Ogawa M, Sugibayashi K, Yamada K
and Yamamoto K: Relationship between tyrosinase inhibitory action
and oxidation-reduction potential of cosmetic whitening ingredients
and phenol derivatives. Arch Pharm Res. 22:335–339. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hu ZM, Zhou Q, Lei TC, Ding SF and Xu SZ:
Effects of hydroquinone and its glucoside derivatives on
melanogenesis and antioxidation: Biosafety as skin whitening
agents. J Dermatol Sci. 55:179–184. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tomita Y, Maeda K and Tagami H:
Melanocyte-stimulating properties of arachidonic acid metabolites:
possible role in postinflammatory pigmentation. Pigment cell
research/sponsored by the European Society for Pigment Cell
Research and the International. Pigment Cell Society. 5:357–361.
1992. View Article : Google Scholar
|
|
11
|
Nakajima M, Shinoda I, Fukuwatari Y and
Hayasawa H: Arbutin increases the pigmentation of cultured human
melanocytes through mechanisms other than the induction of
tyrosinase activity. Pigment cell research/sponsored by the
European Society for Pigment Cell Research and the International
Pigment Cell Society. 11:12–17. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sugimoto K, Nishimura T, Nomura K,
Sugimoto K and Kuriki T: Inhibitory effects of alpha-arbutin on
melanin synthesis in cultured human melanoma cells and a
three-dimensional human skin model. Biol Pharm Bull. 27:510–514.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Akiu S, Suzuki Y, Asahara T, Fujinuma Y
and Fukuda M: Inhibitory effect of arbutin on
melanogenesis-biochemical study using cultured B16 melanoma cells.
Nihon Hifuka Gakkai Zasshi. 101:609–613. 1991.PubMed/NCBI
|
|
14
|
Maeda K and Fukuda M: In vitro
effectiveness of several whitening cosmetic components in human
melanocytes. J Soc Cosmet Chem. 42:261–268. 1991.
|
|
15
|
Sugimoto K, Nishimura T, Nomura K,
Sugimoto K and Kuriki T: Syntheses of arbutin-alpha-glycosides and
a comparison of their inhibitory effects with those of
alpha-arbutin and arbutin on human tyrosinase. Chem Pharm Bull
(Tokyo). 51:798–801. 2003. View Article : Google Scholar
|
|
16
|
Sugimoto K, Nomura K, Nishimura T, Kiso T,
Sugimoto K and Kuriki T: Syntheses of
alpha-arbutin-alpha-glycosides and their inhibitory effects on
human tyrosinase. J Biosci Bioeng. 99:272–276. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cheng SL, Liu RH, Sheu JN, Chen ST,
Sinchaikul S and Tsay GJ: Toxicogenomics of A375 human malignant
melanoma cells treated with arbutin. J Biomed Sci. 14:87–105. 2007.
View Article : Google Scholar
|
|
18
|
Li H, Jeong YM, Kim SY, Kim MK and Kim DS:
Arbutin inhibits TCCSUP human bladder cancer cell proliferation via
up-regulation of p21. Pharmazie. 66:306–309. 2011.PubMed/NCBI
|
|
19
|
Reed JC: Dysregulation of apoptosis in
cancer. J Clin Oncol. 17:2941–2953. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kurokawa M and Kornbluth S: Caspases and
kinases in a death grip. Cell. 138:838–854. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jiang L, Xie X, Yue H, Wu Z, Wang H, Yang
F, Wang L and Wang Z: Highly efficient and regioselective acylation
of arbutin catalyzed by lipase from Candida sp. Process Biochem.
50:789–792. 2015. View Article : Google Scholar
|
|
22
|
Fujii T and Saito M: Inhibitory effect of
quercetin isolated from rose hip (Rosa canina L.) against
melanogenesis by mouse melanoma cells. Biosci Biotechnol Biochem.
73:1989–1993. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kubo I, Nitoda T and Nihei K: Effects of
quercetin on mushroom tyrosinase and B16-F10 melanoma cells.
Molecules. 12:1045–1056. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang J, Wu D, Xing Z, Liang S, Han H, Shi
H, Zhang Y, Yang Y and Li Q: N-Isopropylacrylamide-modified
polyethylenimine-mediated p53 gene delivery to prevent the
proliferation of cancer cells. Colloids Surf B Biointerfaces.
129:54–62. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xing Z, Gao S, Duan Y, Han H, Li L, Yang Y
and Li Q: Delivery of DNAzyme targeting aurora kinase A to inhibit
the proliferation and migration of human prostate cancer. Int J
Nanomedicine. 10:5715–5727. 2015.PubMed/NCBI
|
|
26
|
Maeda K and Fukuda M: Arbutin: Mechanism
of its depigmenting action in human melanocyte culture. J Pharmacol
Exp Ther. 276:765–769. 1996.PubMed/NCBI
|
|
27
|
Hori I, Nihei K and Kubo I: Structural
criteria for depigmenting mechanism of arbutin. Phytother Res.
18:475–479. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kang KH, Lee B, Son S, Yun HY, Moon KM,
Jeong HO, Kim DH, Lee EK, Choi YJ, Kim H, et al: (Z)-2-(Benzo[d]
thiazol-2-ylamino)-5-(substituted benzylidene)thiazol-4(5H)-one
Derivatives as Novel Tyrosinase Inhibitors. Biol Pharm Bull.
38:1227–1233. 2015. View Article : Google Scholar
|
|
29
|
Yan Q, Cao R, Yi W, Yu L, Chen Z, Ma L and
Song H: Synthesis and evaluation of
5-benzylidene(thio)barbiturate-beta-D-glycosides as mushroom
tyrosinase inhibitors. Bioorg Med Chem Lett. 19:4055–4058. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tokiwa Y, Kitagawa M and Raku T: Enzymatic
synthesis of arbutin undecylenic acid ester and its inhibitory
effect on mushroom tyrosinase. Biotechnol Lett. 29:481–486. 2007.
View Article : Google Scholar
|
|
31
|
Tokiwa Y, Kitagawa M, Raku T, Yanagitani S
and Yoshino K: Enzymatic synthesis of arbutin undecylenic acid
ester and its inhibitory effect on melanin synthesis. Bioorg Med
Chem Lett. 17:3105–3108. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lee CS, Kim YJ, Lee MS, Han ES and Lee SJ:
18beta-Glyc-yrrhetinic acid induces apoptotic cell death in SiHa
cells and exhibits a synergistic effect against antibiotic
anti-cancer drug toxicity. Life Sci. 83:481–489. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kang MH and Reynolds CP: Bcl-2 inhibitors:
targeting mitochondrial apoptotic pathways in cancer therapy. Clin
cancer res. 15:126–1132. 2009. View Article : Google Scholar
|
|
34
|
Kinnally KW and Antonsson B: A tale of two
mitochondrial channels, MAC and PTP, in apoptosis. Apoptosis.
12:857–868. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wochna A, Niemczyk E, Kurono C, Masaoka M,
Kedzior J, Słomińska E, Lipiński M and Wakabayashi T: A possible
role of oxidative stress in the switch mechanism of the cell death
mode from apoptosis to necrosis--studies on rho0 cells.
Mitochondrion. 7:119–124. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Denning TL, Takaishi H, Crowe SE, Boldogh
I, Jevnikar A and Ernst PB: Oxidative stress induces the expression
of Fas and Fas ligand and apoptosis in murine intestinal epithelial
cells. Free Radic Biol Med. 33:1641–1650. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wu LH, Li P, Zhao QL, Piao JL, Jiao YF,
Kadowaki M and Kondo T: Arbutin, an intracellular hydroxyl radical
scavenger, protects radiation-induced apoptosis in human lymphoma
U937 cells. Apoptosis. 19:1654–1663. 2014. View Article : Google Scholar : PubMed/NCBI
|