Introduction
Liver fibrosis is the advanced stage of liver
disease accompanying by hepatocyte necrosis and excessive or
uncontrolled deposition of extracellular matrix (ECM) under
constant stimulation of a variety of pathogenic factors (viral
hepatitis, alcohol abuse, metabolic disease and immune injury)
(1–3). Great progress has been made in the
mechanisms and cell biology of liver fibrosis. Numerous small
molecules and biologics have been identified that are reaching
preclinical testing of anti-fibrotic agents and strategies, but the
effective anti-fibrotic drugs approved for clinical use in advanced
liver fibrosis still are scarce (4–6).
Although liver transplantation is the only effective treatment to
cure liver cirrhosis at present, it is limited by organ donor
shortage, surgery-related complications, immunological rejection,
and high cost worldwide (7,8).
In recent years, increasing research (9–14)
has suggested that stem cell transplantation is an effective
alternative therapy for liver fibrosis/cirrhosis. The stem cells,
including embryonic stem cells, induced pluripotent stem cells, and
adult stem cells, have the potential of differentiation into
hepatocyte-like cells both in vivo and in vitro
(15–19). In these stem cells, bone
marrow-derived mesenchymal stem cells (BMSCs) are the most abundant
source and is most widely used in animal experiments and clinical
trials. BMSCs have several advantages, such as easy acquisition,
strong proliferative capacities, and immune-modulatory property
that are able to migrate to damaged tissues (20). BMSC transplantation therapy alone
may not attenuate liver fibrosis completely (21), since it cannot degrade the ECM and
fiber scar effectively in cirrhotic tissue which may prevent
proliferation of BMSCs, suggesting that the therapeutic efficacy of
BMSCs needs improvement. According to recent studies (22–25), BMSCs could be used as a potent
ideal vehicle for gene delivery. Gene modified stem cells may
maintain the direct differentiation characteristics and secrete
exogenous cytokines for the purpose of anti-fibrogenic therapy.
Matrix metalloproteinase (MMP) is the main enzyme
responsible for ECM degradation and tissue inhibitor of
metalloproteinases (TIMPs) has the ability to inhibit MMPs
(26). MMPs secreted by HSCs and
Kupffer cells participating in the degradation of ECM, is
endogenous proteolytic enzyme family of zinc-calcium ions (27). MMP is the strongest enzyme to
degrade collagen fibers, which are the main component of ECM and
play an important role in physiological and pathological process.
Although some studies and cell culture findings suggest that MMP2
promotes hepatic fibrogenesis (28). Moreover, some evidence suggests
that MMP2 may be anti-fibrotic in liver disease, which is capable
of cleaving type I collagen in vitro and limiting HSC
activity after liver injury (29–31). MMP1, called fibroblasts type, is
the main human interstitial collagenase and reversed liver fibrosis
process by degrading collagen type I and III in ECM (32). It has been reported that imbalance
between too few MMP1 and too much TIMP1 is an important mechanism
of liver fibrosis (33). Iimuro
et al tried to improve this imbalance by upregulating MMP1
expression in rat and observed liver fibrosis attenuation to some
extent (34). Yang et al
(35) also found that enhancement
of the expression of MMP1 in liver tissues of
CCl4-induced hepatic fibrotic rats, which may result in
its elevated activity that contributes to fighting against hepatic
fibrosis. In the present study, we investigated the therapeutic
efficacy of BMSCs overexpressing MMP1 in a rat model of liver
fibrosis induced by CCl4. To assess therapeutic
effectiveness, we evaluated changes in liver function, liver
histopathology and fibrous protein [hepatic hydroxyproline and
α-smooth muscle actin (α-SMA)] after transplantation. We show that
therapy with BMSCs/MMP1 resulted in an improved therapeutic effect
compared with BMSCs alone, probably because of the sustainably
expressed MMP1 level in the liver. Our findings indicate BMSCs/MMP1
transplantation not only improved biochemical parameters but also
attenuated progression of liver fibrosis, suggesting that BMSCs may
be a potential cell source in preventing liver fibrosis and MMP1
gene may enhance the anti-fibrotic effect of BMSCs.
Materials and methods
Animals
Male Sprague-Dawley (SD) rats were obtained from the
Institute of Zoology at the Third Military Medical university
(Chongqing, China). The animals were housed in air-conditioned
rooms, with controlled temperature and humidity with 12 h
light-dark cycles. Food and water were available ad libitum.
Animal welfare and experimental procedures were carried out in
accordance with the Guide for the Care and Use of Laboratory
Animals (Ministry of Science and Technology of China, 2006). The
Ethics Committee of Chengdu Military General Hospital approved all
of the animal experiments.
Construction of recombinant adenovirus
vector
Constructing the recombinant adenovirus vector
containing hMMP1 gene with Gateway™ Clone Technology as previously
described (36). Briefly, the
full-length gene hMMP1 was amplified by using PCR from the pcDNA3.1
plasmid, then it was cut down and connected to the entry vector
pENTER™ 1A (both from Invitrogen, Carlsbad, CA, USA). The entry
clone and the destination vectors pJTI™ R4 the Dest CMV-IRES/eGFP
pA vector (Invitrogen) recombine using the LR reaction to form the
expression clone pAd-hMMP1-IRES/eGFP. The linear
pAd-hMMP1-IRES/eGFP transfected into HEK293A cells packaging the
Ad-hMMP1-IRES/eGFP. The target protein expression was detected by
RT-PCR and western blot assay. The adenovirus titre was measured by
TCID50 method, and stored at −80°C in the phosphate-buffered saline
(PBS).
BMSCs isolation, culture and gene
transduction
Rats BMSCs were isolated from bone marrow and
expanded in culture according to previous studies (37,38). For adenoviral transduction, the
BMSCs were washed with serum-free Dulbecco's modified Eagle's
medium (DMEM) three times and exposed to fresh medium containing
Ad-MMP1-eGFP (1.8×1010 pfu/ml) and Ad-eGFP
(1.0×1010 pfu/ml) in 5 ml DMEM at 37°C for 4 h,
according to the multiplicity of infection 50, 100, 200 and 300
(pfu number/cell). The medium was removed, and the cells were
washed once with DMEM and re-cultured in normal medium for 24 h,
after which cell transplantation was performed.
Cell surface labeling
BMSCs and MMP1-BMSC phenotypes were analyzed by flow
cytometry using a FACSCalibur (Becton-Dickinson Biosciences, Ann
Arbor, MI, USA). The cells were re-suspended in phosphate-buffered
saline (PBS) at a concentration of 1×106 cells/ml, and
were incubated with following fluorescent anti-human antibodies:
fluorescein isothiocyanate (FITC)-conjugated CD45 and CD90,
phycoerythrin (PE)-conjugated CD105, CD14, CD34 and CD79a (Santa
Cruz Biotechnology, Inc., Santa Cruz, CA, USA). The rat
immunoglobulin IgG-FITC and IgG-PE was used as the isotype-matched
control. The cells were tagged 45 min away from light at room
temperature, washed three times with PBS and detected with
FACSCalibur flow cytometer.
CCl4-induced liver fibrosis
model and BMSCs transplantation
Seventy male rats were divided into two groups
randomly, group A: liver fibrosis model (n=60) and group B: control
(n=10). Liver fibrosis was induced by subcutaneous injection of
CCl4 oil solution (1:1 olive oil; Sigma-Aldrich,
Steinheim, Germany) at a dose of 1 ml/kg body weight twice per week
for 8 weeks. The same volume of saline solution was applied to
control rats. The rats were sacrificed to assess the extent of
liver fibrosis after withdrawing injection of CCl4. For
cell implantation, CCl4 treated rats were classified
into five groups (n=10): normal control group, rats were treated
with olive oil infused with saline; model control group, rats were
treated with CCl4 infused with olive oil; BMSCs group,
rats were treated with CCl4 infused with saline
containing untreated BMSCs (3×106 cells); eGFP/BMSCs
group, rats were treated with CCl4 infused with saline
containing BMSCs transduced with Ad-eGFP for 24 h (3×106
cells); MMP1-eGFP/BMSCs group, rats treated with CCl4
were infused with saline containing BMSCs transduced with Ad
MMP1-eGFP for 24 h (3×106 cells); transplantation was
administered as a single dose. Rats were sacrificed 2 or 4 weeks
post-implantation, liver tissue was obtained to observe the
expression of GFP by frozen section. Blood was collected from
celiac artery for analysis. Liver tissue was fixed in 10% neutral
buffered formalin for histopathological and immunohistochemical
examination, or stored at −80°C for future use.
Enzyme-linked immunosorbent assay (ELISA)
for MMP1 secreted by adenoviral transducted BMSCs and TIMP1 in the
liver
Enzyme-linked immunosorbent assay for MMP1 secreted
in both MMP1/BMSCs and BMSCs (2×106 cells) were
transfected with Ad-MMP1-eGFP at optimum MOI of 300 puf/cell in
6-well plates and cultured for 72 h, the culture supernants were
centrifuged at 10,000 × g/min for 10 min and collected for
analysis. In addition, the media from untreated BMSCs were
collected as the control. Commercial MMP1 ELISA kits (R&D
Systems, Minneapolis, MN, USA) were used to detect the content of
MMP1 in each group.
The levels of MMP1 and tissue inhibitor of
metal-loproteinases-1 (TIMP1) in the liver tissue of rats 4 weeks
after transplantation were measured using ELISA kits (R&D
Systems). Wet liver tissue (100 mg/sample) was homogenized in 1 ml
PBS in the presence of 1% protease inhibitors (Sigma-Aldrich). The
supernatant fraction of liver homogenate was used to measure MMP1
and TIMP1 levels.
Enzymatic activity of MMP1 in vitro and
in vivo
The enzymatic activity of human matrix
metalloproteinase 1 secreted in MMP1 gene modified BMSCs and in
liver after implantation was tested by fluorescence quantitative
kits of MMP1 enzyme activity (GEM, China). The protein of MMP1 was
collected from the supernatants in cultured BMSCs and from liver
tissue as above. Enzyme activity was performed according to the
manufacturer's instructions. Fluorescence microplate reader (Japan)
the setting was: excitation wavelength 330 nm, distribution
wavelength 400 nm, 37°C. The data were expressed as the mean
[nmol/(mg•min)] ± SD, with n=10/group.
Liver function test
The blood samples were obtained from large artery
according to experimental design, and stored at −80°C. Alanine
aminotransferase (ALT), aspartate aminotransferase (AST), total
bilirubin (TBIL), albumin (ALB) and prothrombin time (PT) were
measured with automatic biochemical analyzer.
Liver histology
The Masson staining method was to detect collagen
fibers, the blue areas are considered the collagen area. The livers
were harvested at sacrifice, washed in PBS, and fixed in 10%
formalin overnight at 4°C. Tissues embedded in paraffin were cut
into 5-µm-thin sections. For Masson's trichrome stain,
sectioned samples were placed in Bouin's solution at waterbath of
60°C for 1 h and washed in running tap water for 5 min, stained in
succession with Weigert's working hematoxylin solution for 10 min,
Biebrich Scarlet solution for 5 min,
phosphomo-lybdic/phosphotungstic-acid for 10 min, transferred
directly into Aniline blue for 5 min, 1% acetic acid for 1 min,
dehydrate, clear, and coverslip. Sections were examined with a
microscope (IX70; Olympus, Tokyo, Japan). For H&E analysis,
sectioned samples were stained with Mayer's hematoxylin solution
(Sigma-Aldrich) for 5 min followed by Eosin Y (Deventer, The
Netherlands) for 5 min.
Hepatic hydroxyproline determination
Hepatic hydroxyproline was tested by improvement of
Kivirikko method. Simply, liver samples of rats were obtained from
sacrifice, and 20 mg of liver tissue were weighed, frozen and cut
into homogeneity. Liver tissue was hydrolyzed with 3 ml 6 N HCl for
24 h at 100°C. The mixture was centrifuged at 2,000 rpm, 4°C for 5
min. The content of hydroxyproline (HYP) was determined by
colorimetric assay at a wavelength of 560 nm. The quantity of HYP
was calculated against a calibration curve obtained using HYP
standards. Finally, the HYP content in each sample was quantified
with µg/g (liver wet dry).
Western blot analysis for α-SMA of the
liver tissue
Liver tissues harvested from rats per group were
lysed with RIPA peptide lysis buffer (Shenerg Biocolor, Shanghai,
China) with 1% protease inhibitors at 4°C. Lysate containing 20
µg of protein was separated by electrophoresis on 10%
acrylamide sodium dodecyl sulfate (SDS) gels. After
electrophoresis, the protein was transferred onto polyvinylidene
difluoride membranes (PVDF; Millipore, Billerica, MA, USA). The
membrane was incubated with mouse anti-rat α-SMA monoclonal
antibody (1:1,000 dilution; Sigma-Aldrich) overnight at 4°C and
horseradish peroxidase (HRP)-conjugated goat anti-mouse IgG
(1:5,000 dilution; Santa Cruz Biotechnology, Inc.) for 2 h at room
temperature in a gyratory shaker. After adequate washes, the
membrane was processed using SuperSignal West Pico chemiluminescent
substrate (Pierce, Rockford, IL, USA). The mouse anti-rat β-actin
monoclonal antibody (1:500 dilution; Santa Cruz Biotechnology,
Inc.) as an internal standard. The intensities of α-SMA and β-actin
were measured using the quantity.
Statistical analysis
The data are presented as the means ± standard
deviation (SD). The differences between mean values of each group
were compared by a one-way analysis of variance (ANOVA) and
considered to be statistically significant when the adjusted
P<0.05. All analyses were performed using SPSS version 16.0
statistical software (SPSS, Inc., Chicago, IL, USA). ImageJ was
used to analyze the images.
Results
Characteristics of BMSCs and MMP1
gene-transduced BMSCs
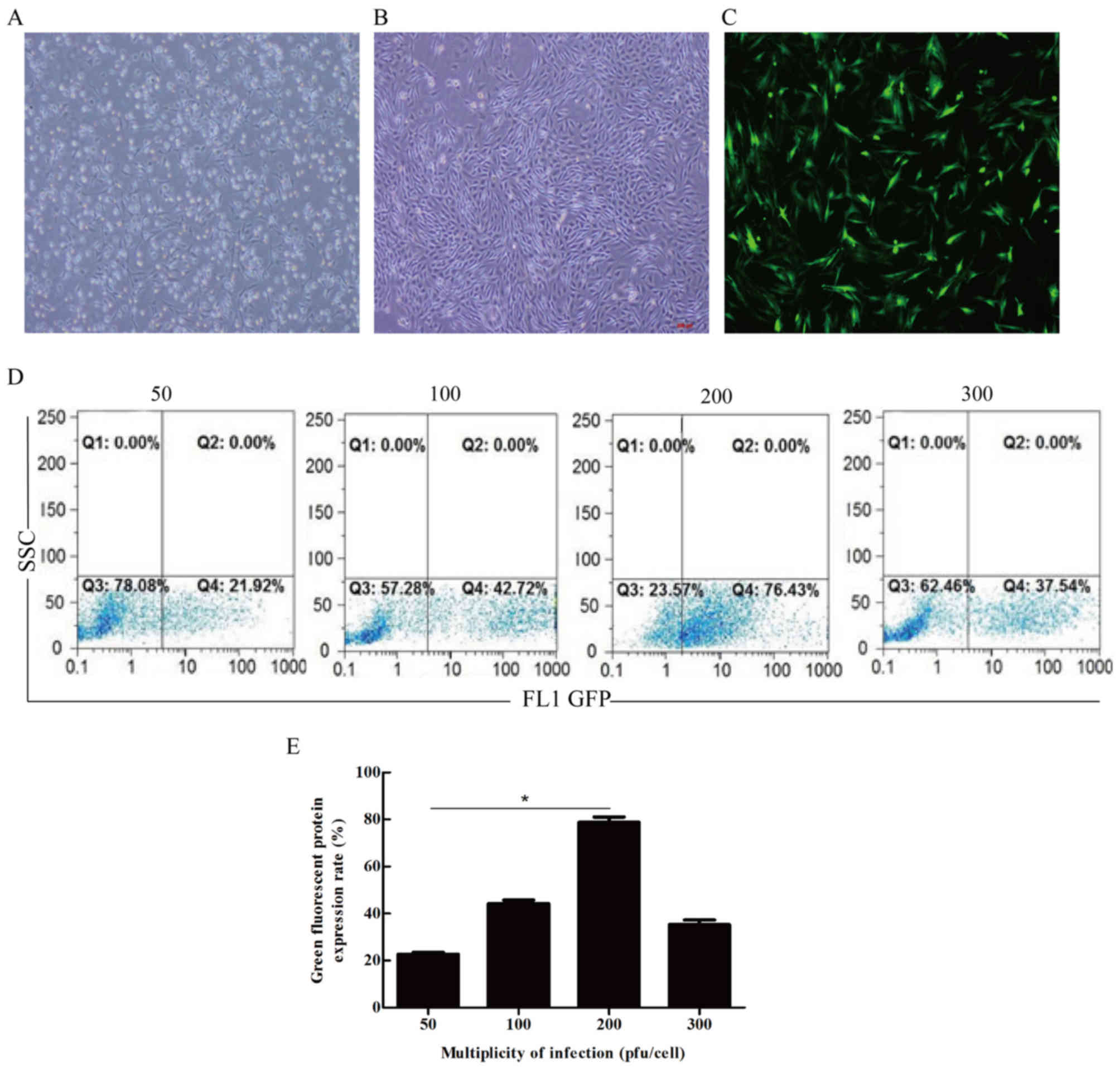
The isolated BMSCs presented uniform morphology,
grew in spindles, and arranged in whirlpool shape (Fig. 1A and B). BMSCs were then
transfected by recombinant adenovirus at the multiplicity of
infection (MOI) of 50, 100, 200 or 300 pfu/cell, respectively. We
found that BMSCs/MMP1 expressed green fluorescence from 24 h to 21
days after infection. The fluorescence intensity increased in level
with MOI and reached its highest level of 76.43% at MOI 200
pfu/cell 72 h after the infection (Fig. 1C–E).
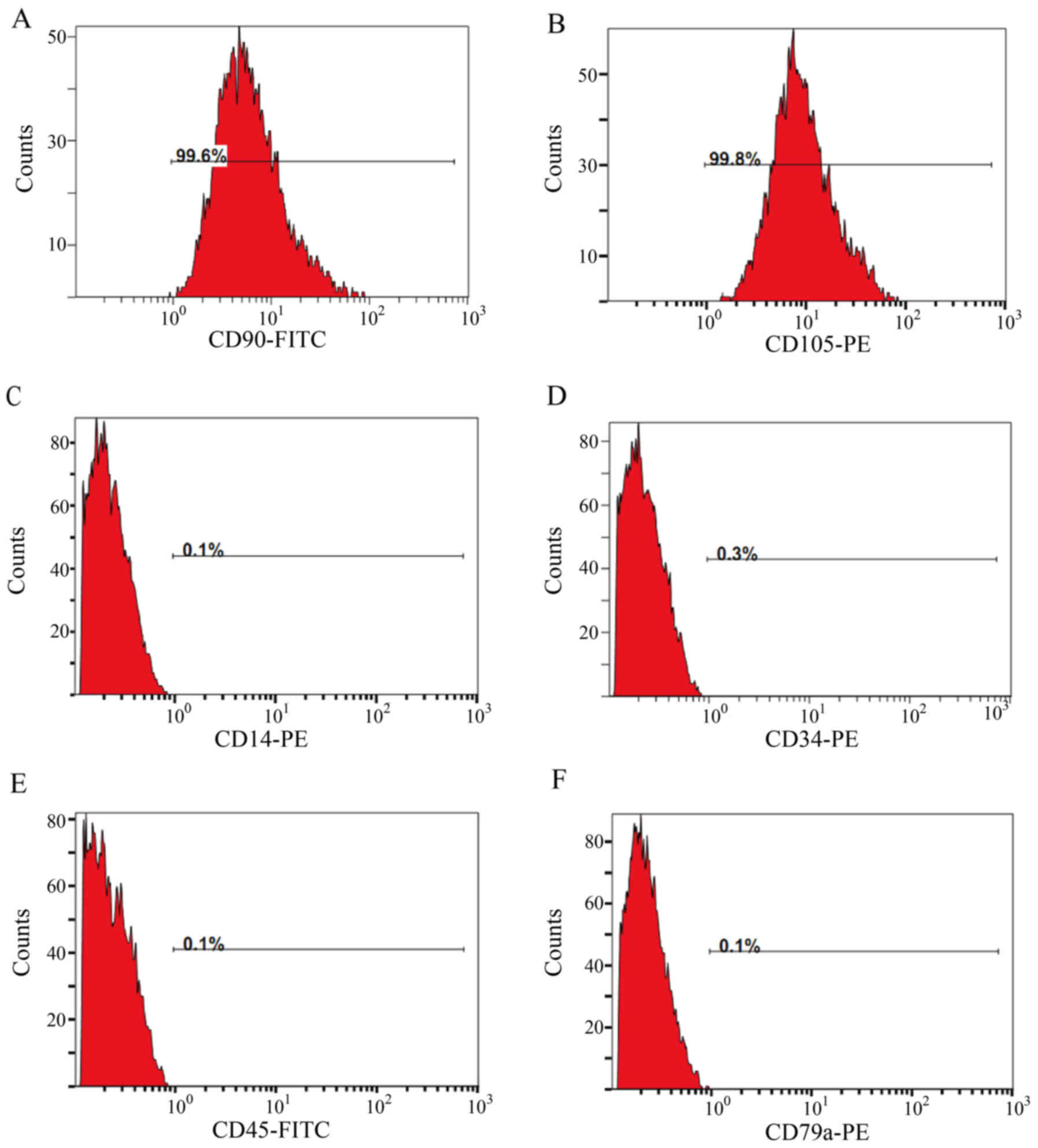
To identify the origin of these cells, we next
detected the expression of both BMSC markers CD90 and CD105 and
hematopoietic cell markers CD34, CD45, CD14 and CD79a by flow
cytometry. The results showed that BMSCs/MMP1 were 99.6% positive
for CD90 and 99.8% for CD105, while only 0.1% positive for CD45,
CD14 and CD79a, and 0.3% positive for CD34 (Fig. 2), which indicated that the bone
marrow-derived and MMP1 gene modified cells were BMSCs.
BMSCs/MMP1 alleviated
CCl4-induced liver fibrosis
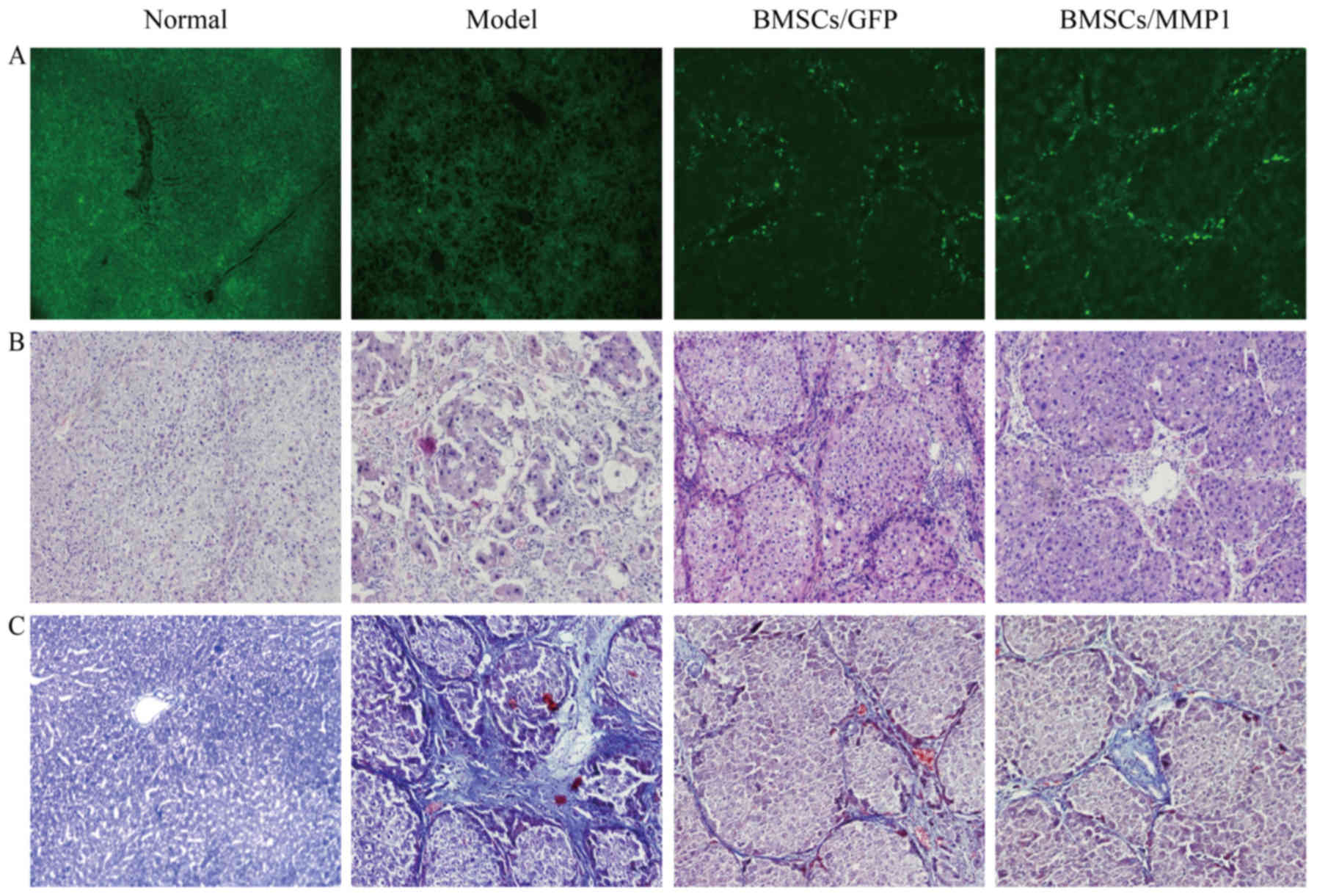
After transplantation of BMSCs, the green
fluorescence positive cells distributed around the hepatic
vascular, hepatic sinusoid, and hepatic lobule of implantation rats
by fluorescent microscope (Fig.
3A), indicating that BMSCs/MMP1 were implanted successfully in
the liver. To address the therapeutic effect of BMSCs/MMP1 on liver
fibrosis, we injected saline, BMSCs, or BMSCs/MMP1 into rats via
the tail vein. In CCl4-induced fibrotic liver, there
were evidently much pseudolobuli surrounded by fibrotic septa
joining the central area and was slightly decreased by
transplantation of BMSCs, while it was significantly reduced by
BMSCs/MMP1 transplantation (Fig.
3B).
Masson staining were performed 4 weeks after cell
transplantation to investigate the collagen content in fibrotic
liver. The collagen stained area slightly decreased by
transplantation of BMSCs and was strongly reduced by BMSCs/MMP1
transplantation, which was consistent with the histological changes
(Fig. 3C and D). Intra-hepatic
hydroxyproline levels, another indicator of tissue collagen
content, showed a similar pattern (Fig. 3E). These results clearly
demonstrated that BMSC transplantation degraded hepatic collagen to
a certain degree, while BMSCs/MMP1 may enhance the anti-fibrotic
effect significantly in liver fibrosis.
The expression of α-SMA represents the activation of
hepatic HSCs, a main profibrogenic factor during liver fibrosis.
Western blot analysis results showed that expression of α-SMA
significantly increased in the model group compared with those of
normal group. Transplantation of BMSCs decreased the expression of
α-SMA, and transplantation of BMSCs/MMP1 decreased the α-SMA level
to a further low level (Fig. 3F and
G). Taken together, these data indicated that BMSCs/MMP1 were
significantly more effective than BMSCs alone as a therapy for
liver fibrosis in rats.
BMSCs/MMP1 attenuated
CCl4-induced liver injury
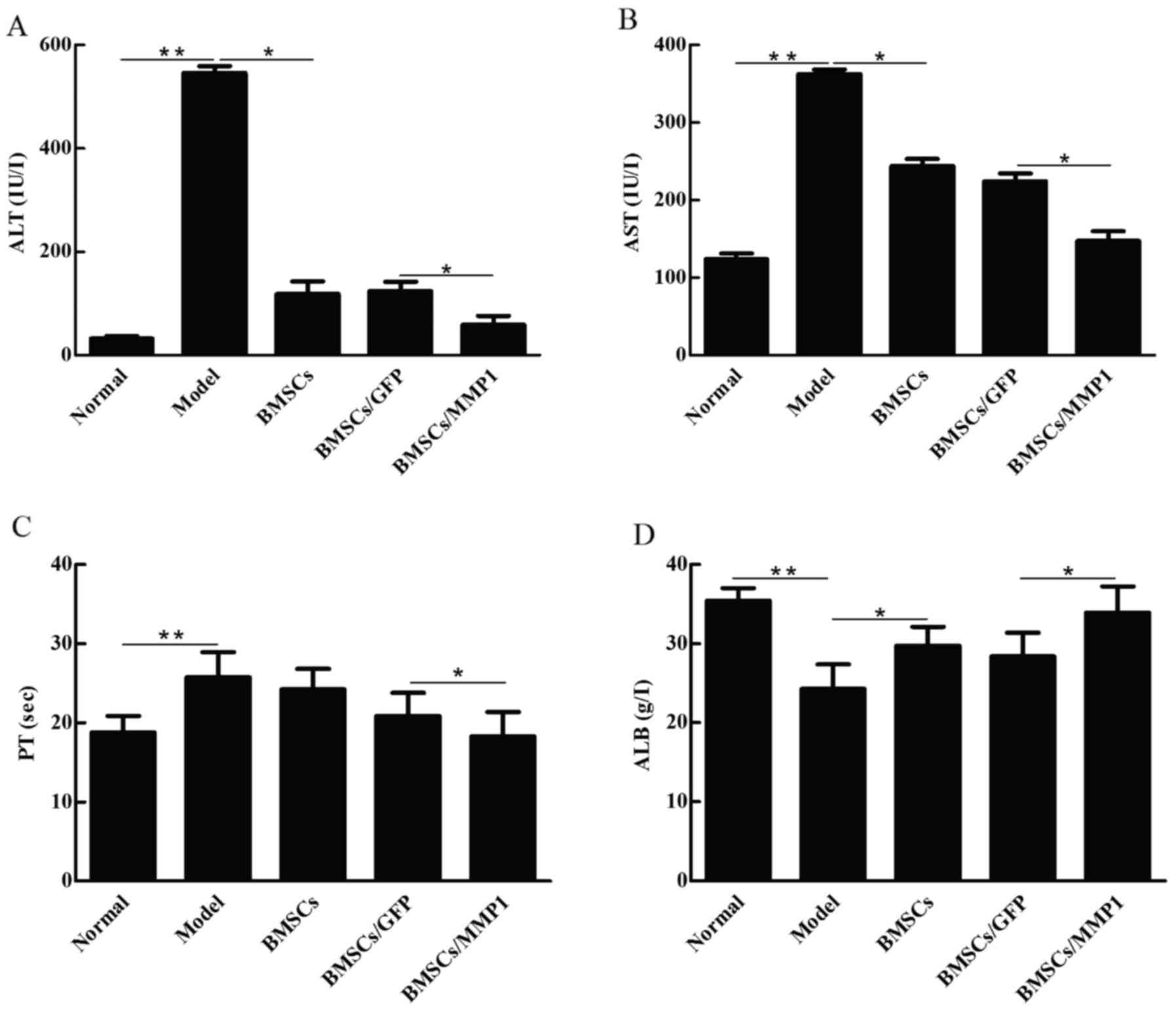
We subsequently evaluated the effects of cell
transplantation on liver injury and liver function. As shown in
Fig. 4, levels of ALT (Fig. 4A) and AST (Fig. 4B), which are indicators of liver
damage, and PT (Fig. 4C)
significantly increased in the CCl4 model group, and ALB
(Fig. 4D), which is a parameter
of liver function, markedly decreased compared with those of the
normal controls. Transplantations of BMSCs slightly improved these
parameters, while BMSCs/MMP1 decreased levels of ALT, AST and PT,
and increased ALB level. These results indicated that BMSCs/MMP1
was more effective than BMSCs alone with respect to the attenuation
of liver injury and recovery of liver function.
Hepatic MMP1 and TIMP1 levels after
transplantation of BMSCs/MMP1
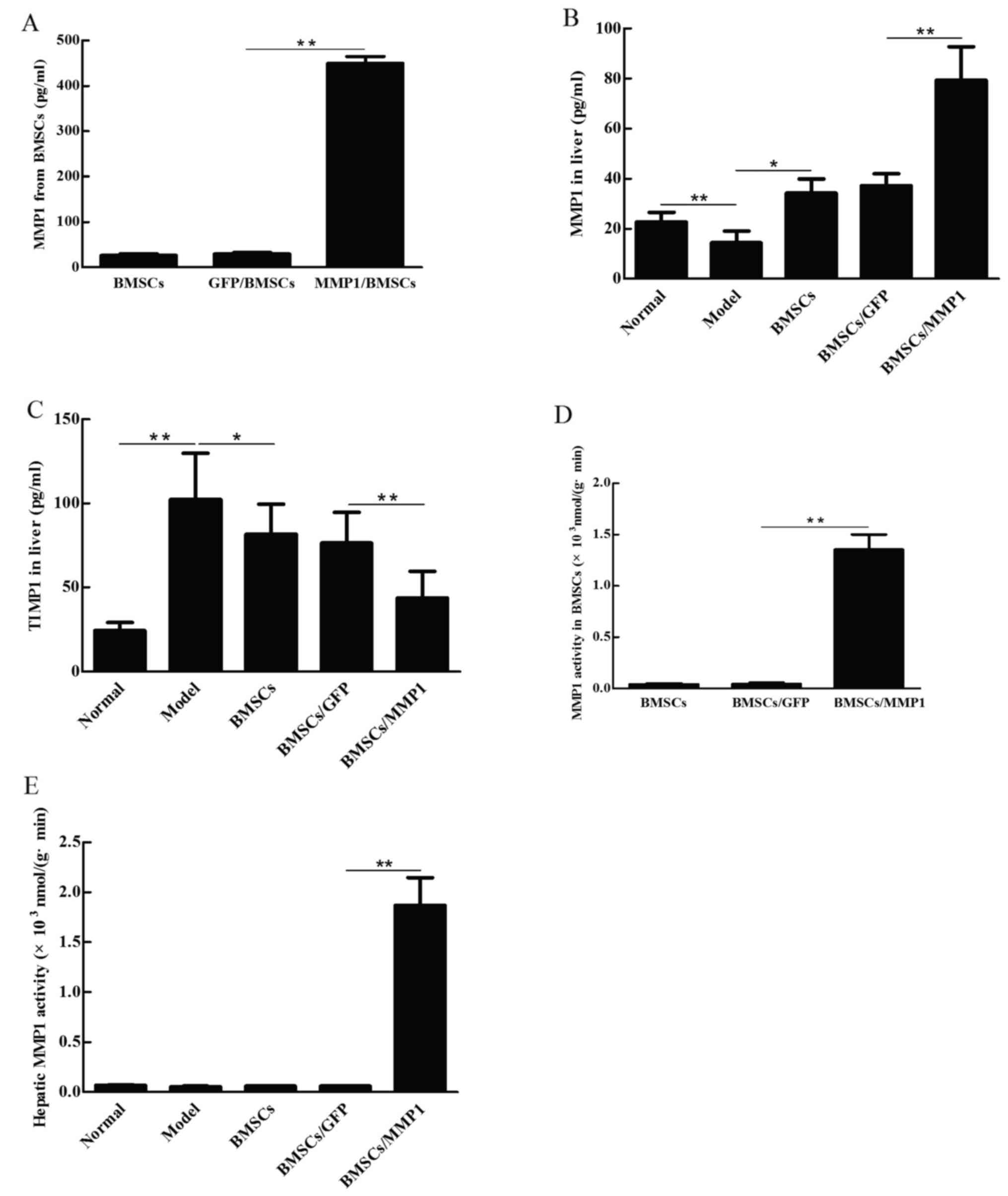
The amount of MMP1 produced by BMSCs/MMP1 was
assessed by ELISA. The results showed that the amount of MMP1 from
BMSCs/MMP1 increased more than 100 times higher than the amount
secreted by BMSCs (Fig. 5A). To
investigate the effect of BMSCs/MMP1 on MMP1 and TIMP1 secretion in
CCl4-induced liver fibrosis, we next transplanted the
BMSCs/MMP1 in these rats. The results showed that MMP1 level
significantly increased in BMSCs/MMP1 group compared with those of
BMSC group (Fig. 5B), while TMIP1
level was significantly suppressed in the BMSCs/MMP1 group compared
with either model group or normal group (Fig. 5C). In addition, the ratio of the
MMP1 to TIMP1 level in model group was lower than that of normal
group (data not shown). Transplantation of BMSCs improved the
imbalance, while transplantation of BMSCs/MMP1 increased the ratio
to a further high level compared with that of BMSCs alone.
We next investigated the enzyme activity of MMP1
produced by BMSCs/MMP1 before and after cell transplantation. The
enzyme activity of MMP1, either 72 h after gene transfection or 2
weeks after transplantation was detected. The results showed that
enzyme activity of MMP1 [1.3528×10−3 nmol/(g•min)] was
higher in BMSCs/MMP1 than that of BMSC group (Fig. 5D). After cell transplantation, the
enzyme activity of MMP1 was higher in livers of BMSCs/MMP1 injected
animals than that of BMSC group (Fig.
5E). These data demonstrated that not only the quantity but
also the biological activity of MMP1 produced by BMSCs/MMP1 was
elevated either in vitro or in the liver.
Discussion
Liver fibrosis is a worldwide disease that may lead
to irreversible end-stage liver diseases. There is still no
effective drug to reverse liver cirrhosis. Stem cells have the
capacity of self-renew and differentiation into various cell lines,
including hepatocyte-like cells under proper treatments or in the
presence of a suitable hepatic microenvironment, and therefore,
throw light on therapy in liver diseases (20,39–42). It has been reported that
transplantation of stem cells is an effective therapy for hepatic
diseases, and BMSCs could improve impaired liver function and
participate in the reconstruction of liver architecture (43–46). Nevertheless, other studies
indicated that transplantation with BMSCs in liver fibrosis has its
limitations due to the imbalance of synthesis and degradation of
ECM in liver fibrosis and cirrhosis (21). In cirrhotic liver, recovery of
liver functions is extensively inhibited by fiber tissue which
limits hepatocyte regeneration (47). Recent study showed, although BMSCs
transplantation can reduce the production of collagen partially by
inhibiting the activation of hepatic stellate cells or increasing
MMP9, it could not degrade the collagen in fibrotic liver
effectively (45). There are
still excessive collagens in liver fibrosis after BMSC
transplantation, as was also found in the present study.
Degradation of ECM is mainly induced by MMPs, which
consequently may free up space for hepatic cell proliferation.
Iimuro et al (34)
injected the recombinant adenovirus containing MMP1 gene (Ad-MMP1)
to the thioacetamide-induced liver fibrosis in rats and showed that
the number of activated HSCs decreased, collagen obviously
degraded, hepatocyte partly proliferated, rat liver fibrosis
significantly reduced, and liver function improved consequently.
Garcia-Banuelos et al (48) transplanted hMMP8 gene-modified
recombinant adenovirus (Ad-hMMP8) into liver fibrosis of rats
induced by CCl4 injection and bile duct ligation,
respectively. The results showed that the degree of liver fibrosis
was alleviated and MMP2, MMP3, MMP9 and HGF expression in liver
tissue were increased significantly, while transforming growth
factor-β1 (TGF-β1) expression was reduced, accompanying by the
decrease of the volume of ascites, improvement of liver function,
and disappearance of gastric varices. These results imply that
therapy with upregulated expression of MMP genes targeted to the
liver may be useful as a therapeutic strategy even in advanced
liver fibrosis or liver cirrhosis.
With the development of gene therapy, genetically
engineered BMSC transplantation has been reported to be beneficial
for treatment of bone disease (49), cardiovascular disease (23) and neurological diseases (22). Novel approaches including gene
modified BMSCs have been supposed to reverse established liver
cirrhosis. In the present study, we transplanted BMSCs/MMP1 into
CCl4-induced liver fibrosis in rats for the first time.
Our results showed that exogenous MMP1 stably expressed BMSCs/MMP1
and these cells actively proliferated in vitro. Moreover,
tracing BMSCs by GFP in vivo, we observed that most of the
BMSCs planted in the liver successfully after transplantation,
distributing around the hepatic vasculature, hepatic sinusoid, and
hepatic lobule of implantation rats. BMSCs mainly concentrated in
the liver because of its specific homing capacity to the injured
organ. Regarding the mechanism of BMSC homing in liver, it was
regulated by a variety of molecules, such as Sry-related
high-mobility group box 11 (Sox11) (50), stromal-derived factor-1 (SDF-1)
(51), vascular endothelial
growth factor (VEGF) (52), basic
fibroblast growth factor (bFGF) (53), and fibroblast activation protein
(FAP) (54). Due to its ease to
express exogenous gene and low immunogenicity (55,56), BMSCs may be used as an ideal
target for gene therapy and may play an important role in treatment
of advanced liver fibrosis or even liver cirrhosis.
In the present study, although inhibition of HSC
activation was observed and content of collagen determined both by
the Masson staining and hydroxyproline evaluation in liver fibrosis
was partially degraded after BMSC transplantation, the hepatic
histology was not improved significantly. Transplantation of
BMSCs/MMP1 was more effective than BMSCs alone as a therapy for
liver fibrosis in rats. BMSCs differentiated into hepatocytes under
the fibrotic liver microenvironment, inhibited HSC activation to
reduce collagen deposit, and subsequently improved the liver
function. On the other hand, BMSCs/MMP1 sustainably secreted MMP1
to degrade the excessive hepatic collagens. In this study, Masson
staining and HE staining showed that collagens were effectively
degraded in the liver and distorted architecture of cirrhotic liver
was improved obviously after BMSCs/MMP1 transplantation.
In conclusion, the present study evaluated
BMSCs/MMP1 transfusion in established liver fibrosis. We concluded
that MMP1 gene sustainably expressed both in vivo and in
vitro, transplanted BMSCs/MMP1 mainly concentrated in fibrotic
liver, and consequently both biochemical parameters and hepatic
architecture improved, suggesting that BMSCs may be a potential
cell source and MMP1 gene may be a target for gene-modified BMSC
therapy in chronic liver disease. Although these findings are
encouraging for the further development of gene therapeutic
approaches in liver cirrhosis, research should be undertaken to
investigate mechanisms that may account for it.
Acknowledgments
We would like to thank the staff of the Department
of Gastroenterology and Hepatology of Chengdu Military General
Hospital for their assistance.
Notes
[1]
Funding
This study was supported by the National Natural
Science Foundation of China (no. 81702931 to CD) and the Grand of
Chengdu Military General Hospital (no. 2011YG-A07).
[2] Authors'
contributions
CD contributed to the data interpretation, drafting,
revision and finalization of the manuscript, and funding
application. MJ and XW contributed to the data acquisition,
analysis and manuscript drafting. JQ, HX and YW contributed to the
data acquisition and analysis. YZ and DZ contributed to the
conception of the study and manuscript editing. HX contributed to
the data acquisition. WZ and SZ contributed to the study concept,
experimental design and supervision.
[3] Availability
of data and material
We declared that materials described in the
manuscript, including all relevant raw data, will be freely
available to any scientist wishing to use them for non-commercial
purposes, without breaching participant confidentiality.
[4] Ethics
approval and consent to participate
All animal experiments were approved by the Ethics
Committee of Chengdu Military General Hospital.
[5] Consent for
publication
Not applicable.
[6] Competing
interests
The authors declare that they have no competing
interests.
References
|
1
|
Friedman SL: Molecular regulation of
hepatic fibrosis, an integrated cellular response to tissue injury.
J Biol Chem. 275:2247–2250. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pinzani M, Romanelli RG and Magli S:
Progression of fibrosis in chronic liver diseases: Time to tally
the score. J Hepatol. 34:764–767. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Malhi H and Gores GJ: Cellular and
molecular mechanisms of liver injury. Gastroenterology.
134:1641–1654. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen RJ, Wu HH and Wang YJ: Strategies to
prevent and reverse liver fibrosis in humans and laboratory
animals. Arch Toxicol. 89:1727–1750. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yoon YJ, Friedman SL and Lee YA:
Antifibrotic therapies: Where are we now? Semin Liver Dis.
36:87–98. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schuppan D: Liver fibrosis: Common
mechanisms and antifibrotic therapies. Clin Res Hepatol
Gastroenterol. 39(Suppl 1): S51–S59. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Toniutto P, Zanetto A, Ferrarese A and
Burra P: Current challenges and future directions for liver
transplantation. Liver Int. 37:317–327. 2017. View Article : Google Scholar
|
|
8
|
Jadlowiec CC and Taner T: Liver
transplantation: Current status and challenges. World J
Gastroenterol. 22:4438–4445. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chang YJ, Liu JW, Lin PC, Sun LY, Peng CW,
Luo GH, Chen TM, Lee RP, Lin SZ, Harn HJ, et al: Mesenchymal stem
cells facilitate recovery from chemically induced liver damage and
decrease liver fibrosis. Life Sci. 85:517–525. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sakaida I, Terai S, Yamamoto N, Aoyama K,
Ishikawa T, Nishina H and Okita K: Transplantation of bone marrow
cells reduces CCl4-induced liver fibrosis in mice.
Hepatology. 40:1304–1311. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wu LM, Li LD, Liu H, Ning KY and Li YK:
Effects of Guiyuanfang and autologous transplantation of bone
marrow stem cells on rats with liver fibrosis. World J
Gastroenterol. 11:1155–1160. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li TZ, Kim JH, Cho HH, Lee HS, Kim KS, Lee
SW and Suh H: Therapeutic potential of bone-marrow-derived
mesenchymal stem cells differentiated with growth-factor-free
coculture method in liver-injured rats. Tissue Eng Part A.
16:2649–2659. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang M, Zhang X, Xiong XI, Yang Z, Li P,
Wang J, Sun YU, Yang Z and Hoffman RM: Bone marrow mesenchymal stem
cells reverse liver damage in a carbon tetrachloride-induced mouse
model of chronic liver injury. In Vivo. 30:187–193. 2016.PubMed/NCBI
|
|
14
|
Truong NH, Nguyen NH, Le TV, Vu NB, Huynh
N, Nguyen TV, Le HM, Phan NK and Pham PV: Comparison of the
treatment efficiency of bone marrow-derived mesenchymal stem cell
transplantation via tail and portal veins in
CCl4-induced mouse liver fibrosis. Stem Cells Int.
2016:57204132016. View Article : Google Scholar
|
|
15
|
Shu SN, Wei L, Wang JH, Zhan YT, Chen HS
and Wang Y: Hepatic differentiation capability of rat bone
marrow-derived mesenchymal stem cells and hematopoietic stem cells.
World J Gastroenterol. 10:2818–2822. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Si-Tayeb K, Noto FK, Nagaoka M, Li J,
Battle MA, Duris C, North PE, Dalton S and Duncan SA: Highly
efficient generation of human hepatocyte-like cells from induced
pluripotent stem cells. Hepatology. 51:297–305. 2010. View Article : Google Scholar :
|
|
17
|
di Bonzo LV, Ferrero I, Cravanzola C,
Mareschi K, Rustichell D, Novo E, Sanavio F, Cannito S, Zamara E,
Bertero M, et al: Human mesenchymal stem cells as a two-edged sword
in hepatic regenerative medicine: Engraftment and hepatocyte
differentiation versus profibrogenic potential. Gut. 57:223–231.
2008. View Article : Google Scholar
|
|
18
|
Aurich I, Mueller LP, Aurich H,
Luetzkendorf J, Tisljar K, Dollinger MM, Schormann W, Walldorf J,
Hengstler JG, Fleig WE, et al: Functional integration of
hepatocytes derived from human mesenchymal stem cells into mouse
livers. Gut. 56:405–415. 2007. View Article : Google Scholar
|
|
19
|
Feng Z, Li C, Jiao S, Hu B and Zhao L: In
vitro differentiation of rat bone marrow mesenchymal stem cells
into hepatocytes. Hepatogastroenterology. 58:2081–2086. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Eom YW, Shim KY and Baik SK: Mesenchymal
stem cell therapy for liver fibrosis. Korean J Intern Med.
30:580–589. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shackel N and Rockey D: In pursuit of the
'Holy Grail' - stem cells, hepatic injury, fibrogenesis and repair.
Hepatology. 41:16–18. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Onda T, Honmou O, Harada K, Houkin K,
Hamada H and Kocsis JD: Therapeutic benefits by human mesenchymal
stem cells (hMSCs) and Ang-1 gene-modified hMSCs after cerebral
ischemia. J Cereb Blood Flow Metab. 28:329–340. 2008. View Article : Google Scholar :
|
|
23
|
Zeng B, Chen H, Zhu C, Ren X, Lin G and
Cao F: Effects of combined mesenchymal stem cells and heme
oxygenase-1 therapy on cardiac performance. Eur J Cardiothorac
Surg. 34:850–856. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Siller-López F, Sandoval A, Salgado S,
Salazar A, Bueno M, Garcia J, Vera J, Gálvez J, Hernández I, Ramos
M, et al: Treatment with human metalloproteinase-8 gene delivery
ameliorates experimental rat liver cirrhosis. Gastroenterology.
126:1122–1133. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hu JJ, Sun C, Lan L, Chen YW and Li DG:
Therapeutic effect of transplanting beta(2)m(−)/Thy1(+) bone
marrow-derived hepatocyte stem cells transduced with
lentiviral-mediated HGF gene into CCl(4)-injured rats. J Gene Med.
12:244–254. 2010.PubMed/NCBI
|
|
26
|
Robert S, Gicquel T, Victoni T, Valença S,
Barreto E, Bailly-Maître B, Boichot E and Lagente V: Involvement of
matrix metalloproteinases (MMPs) and inflammasome pathway in
molecular mechanisms of fibrosis. Biosci Rep. 36:e003602016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Brinckerhoff CE and Matrisian LM: Matrix
metalloproteinases: A tail of a frog that became a prince. Nat Rev
Mol Cell Biol. 3:207–214. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Takahara T, Furui K, Funaki J, Nakayama Y,
Itoh H, Miyabayashi C, Sato H, Seiki M, Ooshima A and Watanabe A:
Increased expression of matrix metalloproteinase-II in experimental
liver fibrosis in rats. Hepatology. 21:787–795. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Radbill BD, Gupta R, Ramirez MCM, DiFeo A,
Martignetti JA, Alvarez CE, Friedman SL, Narla G, Vrabie R, Bowles
R, et al: Loss of matrix metalloproteinase-2 amplifies murine
toxin-induced liver fibrosis by upregulating collagen I expression.
Dig Dis Sci. 56:406–416. 2011. View Article : Google Scholar
|
|
30
|
Issa R, Zhou X, Constandinou CM,
Fallowfield J, Millward-Sadler H, Gaca MDA, Sands E, Suliman I,
Trim N and Knorr A: Spontaneous recovery from micronodular
cirrhosis: Evidence for incomplete resolution associated with
matrix cross-linking. Gastroenterology. 126:1795–1808. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Preaux AM, D'Ortho MP, Bralet MP, Laperche
Y and Mavier P: Apoptosis of human hepatic myofibroblasts promotes
activation of matrix metalloproteinase-2. Hepatology. 36:615–622.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Visse R and Nagase H: Matrix
metalloproteinases and tissue inhibitors of metalloproteinases:
Structure, function, and biochemistry. Circ Res. 92:827–839. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nagase H, Visse R and Murphy G: Structure
and function of matrix metalloproteinases and TIMPs. Cardiovasc
Res. 69:562–573. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Iimuro Y, Nishio T, Morimoto T, Nitta T,
Stefanovic B, Choi SK, Brenner DA and Yamaoka Y: Delivery of matrix
metal-loproteinase-1 attenuates established liver fibrosis in the
rat. Gastroenterology. 124:445–458. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yang Q, Xie RJ, Geng XX, Luo XH, Han B and
Cheng ML: Effect of Danshao Huaxian capsule on expression of matrix
metalloproteinase-1 and tissue inhibitor of metalloproteinase-1 in
fibrotic liver of rats. World J Gastroenterol. 11:4953–4956. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Du C, Jiang MD, Zeng WZ and Zheng SM:
Construction of recombinant adenovirus vector for human matrix
metalloproteinase-1 gene and detection of collagen type III
degradation in vitro. Chin J Tissue Eng Res. 7995–8000. 2014.In
Chinese.
|
|
37
|
Nadri S, Soleimani M, Hosseni RH, Massumi
M, Atashi A and Izadpanah R: An efficient method for isolation of
murine bone marrow mesenchymal stem cells. Int J Dev Biol.
51:723–729. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Soleimani M and Nadri S: A protocol for
isolation and culture of mesenchymal stem cells from mouse bone
marrow. Nat Protoc. 4:102–106. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shiota G and Itaba N: Progress in stem
cell-based therapy for liver disease. Hepatol Res. May 18–2016.Epub
ahead of print. PubMed/NCBI
|
|
40
|
Haldar D, Henderson NC, Hirschfield G and
Newsome PN: Mesenchymal stromal cells and liver fibrosis: A
complicated relationship. FASEB J. 30:3905–3928. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Matsumoto T, Takami T and Sakaida I: Cell
transplantation as a non-invasive strategy for treating liver
fibrosis. Expert Rev Gastroenterol Hepatol. 10:639–648. 2016.
View Article : Google Scholar
|
|
42
|
Raafat N, Abdel AS, Abdo FK and El GN:
Mesenchymal stem cells: In vivo therapeutic application ameliorates
carbon tetrachloride induced liver fibrosis in rats. Int J Biochem
Cell Biol. 68:109–118. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cho KA, Lim GW, Joo SY, Woo SY, Seoh JY,
Cho SJ, Han HS and Ryu KH: Transplantation of bone marrow cells
reduces CCl4-induced liver fibrosis in mice. Liver Int.
31:932–939. 2011. View Article : Google Scholar
|
|
44
|
Meier RP, Mahou R, Morel P, Meyer J,
Montanari E, Muller YD, Christofilopoulos P, Wandrey C,
Gonelle-Gispert C and Bühler LH: Microencapsulated human
mesenchymal stem cells decrease liver fibrosis in mice. J Hepatol.
62:634–641. 2015. View Article : Google Scholar
|
|
45
|
Kim MD, Kim SS, Cha HY, Jang SH, Chang DY,
Kim W, Suh-Kim H and Lee JH: Therapeutic effect of hepatocyte
growth factor-secreting mesenchymal stem cells in a rat model of
liver fibrosis. Exp Mol Med. 46:e1102014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Irfan A and Ahmed I: Could stem cell
therapy be the cure in liver cirrhosis? J Clin Exp Hepatol.
5:142–146. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Mormone E, George J and Nieto N: Molecular
pathogenesis of hepatic fibrosis and current therapeutic
approaches. Chem Biol Interact. 193:225–231. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Garcia-Banuelos J, Siller-Lopez F, Miranda
A, Aguilar LK, Aguilar-Cordova E and Armendariz-Borunda J:
Cirrhotic rat livers with extensive fibrosis can be safely
transduced with clinical-grade adenoviral vectors. Evidence of
cirrhosis reversion. Gene Ther. 9:127–134. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Dong SW, Ying DJ, Duan XJ, Xie Z, Yu ZJ,
Zhu CH, Yang B and Sun JS: Bone regeneration using an acellular
extracellular matrix and bone marrow mesenchymal stem cells
expressing Cbfa1. Biosci Biotechnol Biochem. 73:2226–2233. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xu L, Huang S, Hou Y, Liu Y, Ni M, Meng F,
Wang K, Rui Y, Jiang X and Li G: Sox11-modified mesenchymal stem
cells (MSCs) accelerate bone fracture healing: Sox11 regulates
differentiation and migration of MSCs. FASEB J. 29:1143–1152. 2015.
View Article : Google Scholar
|
|
51
|
Yuan L, Sakamoto N, Song G and Sato M:
Low-level shear stress induces human mesenchymal stem cell
migration through the SDF-1/CXCR4 axis via MAPK signaling pathways.
Stem Cells Dev. 22:2384–2393. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Schichor C, Birnbaum T, Etminan N, Schnell
O, Grau S, Miebach S, Aboody K, Padovan C, Straube A, Tonn JC, et
al: Vascular endothelial growth factor A contributes to
glioma-induced migration of human marrow stromal cells (hMSC). Exp
Neurol. 199:301–310. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Schmidt A, Ladage D, Schinkothe T,
Klausmann U, Ulrichs C, Klinz FJ, Brixius K, Arnhold S, Desai B,
Mehlhorn U, et al: Basic fibroblast growth factor controls
migration in human mesenchymal stem cells. Stem Cells.
24:1750–1758. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Chung KM, Hsu SC, Chu YR, Lin MY, Jiaang
WT, Chen RH and Chen X: Fibroblast activation protein (FAP) is
essential for the migration of bone marrow mesenchymal stem cells
through RhoA activation. PLoS one. 9:e887722014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gebler A, Zabel O and Seliger B: The
immunomodulatory capacity of mesenchymal stem cells. Trends Mol
Med. 18:128–134. 2012. View Article : Google Scholar
|
|
56
|
Holmes C and Stanford WL: Concise review:
stem cell antigen-1: expression, function, and enigma. Stem Cells.
25:1339–1347. 2007. View Article : Google Scholar : PubMed/NCBI
|