Introduction
Stroke, especially ischemic stroke, is a disorder
that results in high rates of disability and mortality (1,2).
High incidence and recurrence rates are also characteristics,
resulting in a serious threat to human health (3). The poor prognosis is partially due
to the subsequent release of cytotoxic factors and the resultant
apoptosis (4-6). Apoptosis caused by focal ischemia of
a short duration (30-60 min) is a prominent feature of the ischemic
stroke region and penumbra border region, where neuronal
degeneration and necrosis occur (7,8).
Thus, the development of anti-apoptotic drugs able to improve the
prognosis of stroke is necessary (9).
Previous studies have demonstrated that B cell
lymphoma-2 (Bcl-2) and caspase-3 are involved in apoptosis
regulation (9-11). Bcl-2 overexpression inhibits the
activation of caspase-3 and subsequent apoptosis (10). Caspase-3 is the most important
terminal cleavage enzyme in the process of apoptosis, and serves a
major function in the induction of apoptosis (10,12). P53 also increases the expression
of BCL2 associated X, apoptosis regulator (Bax), which is an
apoptosis promoter belonging to the BCL-2 gene family, and its
overexpression antagonizes Bcl-2 (13). Bcl-2 downregulation also promotes
apoptosis (14). Thus, Bcl-2/Bax,
caspase-3 and p53 may serve an important function in apoptosis,
including neuronal apoptosis.
Glutamate (GLU) is an excitatory neurotransmitter in
the central nervous system, but its excessive accumulation in the
extracellular matrix may result in ischemic brain damage (15). Lactate dehydrogenase (LDH) is
present in the cytoplasm of normal cells and is released when the
cells are damaged. Thus, its activity reflects the degree of cell
damage and cytotoxicity (16).
Midazolam (MDZ) is a typical benzodiazepine drug
with several functions, including anti-anxiety, sedation and
hypnosis. Intramuscular or intravenous injection produces short
anterograde memory loss, since the drug is capable of quickly
crossing the blood-brain barrier (17,18). MDZ does not appear to induce drug
resistance, abstinence symptoms or rebound (17). MDZ has previously been
demonstrated to inhibit caspase-3 activation (19). Furthermore, MDZ inhibits the
apoptosis of astrocytes induced by oxygen and glucose deprivation
(20). Therefore, MDZ may exert a
protective effect on neuronal apoptosis induced by cerebral
ischemia.
Thus, the hypothesis of the present study was that
MDZ regulates the expression of apoptosis-associated genes to
inhibit apoptosis, together with the inhibition of cytotoxicity to
protect neurons and confer recovery-promoting effects.
To validate this hypothesis, GLU-induced neuronal
injury was performed in vitro. LDH release and the
expression of apoptosis-associated genes were measured to evaluate
cytotoxicity and apoptosis. In the constructed transient middle
cerebral artery occlusion (tMCAO) animal model, neuronal protection
was evaluated by an LDH release assay and tissue staining.
Materials and methods
Neuron isolation and culture
Neurons were obtained from 20 male Sprague Dawley
rats (8-12 weeks old; weight, 300-500 g) purchased from the Animal
Experimental Center of Hainan Medical University, The Second
Affiliated Hospital (Haikou, China). Rats had free access to water
and food and they were kept under the standard laboratory
conditions (22-25°C; 65±5% humidity; 12 h light/dark cycle).
Following sacrifice, the brain was carefully removed from the skull
and placed on ice. The meninges were removed under a dissecting
light microscope and the cortical tissue was collected, placed in a
centrifuge tube, mixed with 0.125% trypsin (Beyotime Institute of
Biotechnology, Haimen, China) and digested at 37°C for 30 min.
Dulbecco’s modified Eagle’s medium (DMEM; Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) containing 5% fetal bovine
serum (FBS; Gibco; Thermo Fisher Scientific, Inc.) was added to the
digested tissue and the mixture was carefully pipetted. Then, the
tube was left to stand at 37°C. Fluid without large particles was
transferred into another centrifuge tube and the cell suspension
was carefully pipetted several times to obtain a suspension with no
visible tissue. Fluid was pipetted in a six-well plate and the
supernatant was transferred to another plate containing 5% FBS, 2%
B27 (Gibco; Thermo Fisher Scientific, Inc.) and neurobasal medium
(Gibco; Thermo Fisher Scientific, Inc.) after 2 h (21,22).
MTT assay
Neuron viability was detected by MTT assay. A total
of 104 neurons/well were seeded in 96-well plates
containing 200 µl DMEM with 10% FBS to evaluate neuron
proliferation at different concentrations of MDZ (Shanghai Guyan
Biological Technology Co., Ltd., Shanghai, China) and GLU
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). MTT was added and
incubated for 4 h at 37°C (Cytotoxicity Assay kit; Beyotime
Institute of Biotechnology). DMSO (Sigma-Aldrich; Merck KGaA) was
used to dissolve the formazan crystals. Obtical density was
measured at a wavelength of 570 nm. Neurons without MDZ or GLU
treatment represented control group 1, while groups 2-7 were
simultaneously treated with GLU (final concentration, 100
µM) and MDZ at concentrations of 0, 0.1, 0.3, 0.5, 0.7 or
0.9 mg/l at 37°C for 4 h (23,24)
LDH release assay
Neurons were seeded as described in the previous
paragraph, and incubated with GLU and MDZ at different
concentrations as aforementioned. LDH was measured using an LDH
assay kit (Nanjing KeyGen Biotech Co., Ltd., Nanjing, China)
according to the manufacturer’s protocol (16,25).
Apoptosis evaluation by flow
cytometry
Neurons were seeded as previously described and
incubated with 0.7 mM MDZ and GLU (final concentration, 100
µM). In group 2, only GLU was added (final concentration,
100 µM), while the control group lacked any treatment. The
medium was removed, neurons were washed with PBS and EGTA-free
trypsin was added. When neurons were detached, PBS was added, and
neurons were collected in a 15 ml tube and centrifuged at 1,000 x g
for 3 min at room temperature. Next, neurons were washed with PBS
and centrifuged at 1,000 x g for 3 min at room temperature.
Annexin-V from a Fluorescein Isothiocyanate-Annexin V Apoptosis
Detection kit (BD Biosciences, San Jose, CA, USA) was used
according to the manufacturer’s protocol and apoptosis was measured
by MoFlo Astrios EQ flow cytometry and corresponding Summit version
6.2 software was used to analyze the results (Beckman Coulter,
Brea, CA, USA) (26,27).
Western blot
Neurons were cultured in serum-free DMEM medium for
24 h. NP40 cell lysis buffer (Gibco; Thermo Fisher Scientific,
Inc.) was used to lyse neurons on ice for 5 min, and sonication
(20-24 kHz) was used to further cell cleavage at 0°C (8 times
within 2 min, 5 sec each time and pauses of 10 sec). Cell debris
was removed by centrifugation at 1,000 x g for 1 min at 0°C and
total protein was obtained and detected by Bicinchoninic acid (BCA)
method using BCA Protein Assay kit (Beyotime Institute of
Biotechnology) according to the manufacturer’s protocol.
Graded 4-15% polyacrylamide gels were used (Bio-Rad
Laboratories, Inc., Hercules, CA, USA) and 50 µg protein
were loaded per lane, followed by electrophoresis to separate the
target proteins. Then, proteins were transferred onto Immobilin-P
membranes (Sigma-Aldrich; Merck KGaA), and membranes were blocked
by 5% non-fat milk for 1 h at room temperature. Samples were
incubated overnight with antibodies against Bcl-2 (cat. no. ab692;
1:500), Bax (cat. no. ab32503; 1:500), p53 (cat. no. ab26;
1:1,000), caspase-3 (cat. no. ab13585; 1:500) and GAPDH (cat. no.
ab8245; 1:500) all from Abcam (Cambridge, MA, USA), diluted in 5%
skim milk powder. Following washing twice with PBS and Dulbecco’s
PBS, samples were treated with goat anti-mouse immunoglobulin G
H&L (cat. no. ab6785; 1:10,000; Sigma-Aldrich; Merck KGaA) and
incubated at room temperature for 1 h. Immunoreactive bands were
cut and an Odyssey SA infrared imaging system (LI-COR Biosciences,
Lincoln, NE, USA) was used to observe the results (28,29).
Semi quantitative reverse
transcription-polymerase chain reaction (RT-PCR)
Semi quantitative RT-PCR amplification was used to
evaluate the expression of apoptotic genes with the A1250 Access
RT-PCR system (Promega Corporation, Madison, WI, USA). Neurons were
digested with 5% trypsin (Beyotime Institute of Biotechnology) and
divided into single cells under a light microscope. Then, cells
were lysed with NP40 cell lysis buffer in PCR tubes for 2 h on ice.
MgSO4 (25 mM), nuclease-free water, dNTP mix, AMV/Tfl 5X
Reaction Buffer, SuperScript® IV reverse transcriptase
(all Invitrogen; Thermo Fisher Scientific, Inc.), and specific
upstream and downstream primers for GAPDH, p53, caspase-3, Bcl-2
and Bax were added to a 0.5 ml reaction tube. The thermocycling
conditions for the PCR were set according to the manufacturer’s
protocol of SuperScript™ One-Step RT-PCR System with Platinum™ Taq
DNA Polymerase included in the Prime Script TM RT reagent kit
(Invitrogen; Thermo Fisher Scientific, Inc.). PCR products were
detected by 1.2% agar gel electrophoresis (0.5 µg/ml
ethidium bromide) and observed under ultra violet light, as
previously described (30,31).
The results were analyzed by Quantity One software (version 4.62;
Bio-Rad Laboratories, Inc.). The primers used were the following:
GAPDH forward, 5′-ACC ACA GTC CAT GCC ATC AC-3′ and reverse, 5′-TCC
ACC ACC CTG TTG CTG TA-3′; p53 forward, 5′-ACC TAT GGA AAC TAC TTC
CTG AAA-3′ and reverse, 5′-CTG GCA TTC TGG GAG CTT CA-3′; caspase-3
forward, 5′-TGG AAC AAA TGG ACC TGT TGA CC-3′ and reverse; 5′-AGG
ACT CAA ATT CTG TTG CCA CC-3′; Bcl-2 forward, 5′-TTC TTT GAG TTC
GGT GGG GTC-3′ and reverse, 5′-TGC ATA TTT GTT TGG GGC AGG-3′; and
Bax forward, 5′-TCC ACC AAG AAG CTG AGC GAG-3′ and reverse; 5′-GTC
CAG CCC ATG ATG GTT CT-3′.
Animal model
All animal procedures followed the Guide for the
Care and Use of Laboratory Animals (33) and were approved by the
Institutional Clinical Experiments Committee of the Second
Affiliated Hospital of Hainan Medical University (Haikou, China).
Sprague Dawley rats (male, 3 months old, 300-500 g) were randomly
divided into 4 groups as follows (n=40; n=10 in each group): Sham
group, model group, MDZ (2 mg/kg) group, and MDZ (5 mg/kg) group.
Rats had free access to water and food and they were housed under
stable conditions (22-25°C; 0.03% CO2; 65±5% humidity;
12 h light/dark cycle). Sodium pentobarbital 2.5% (36 mg/kg;
Sigma-Aldrich; Merck KGaA) was used to anesthetize the rats. The
common carotid artery, internal carotid artery and external carotid
artery were isolated, while the wing palate artery and the cranial
branches of the mentioned arteries were separated. Arterial clips
were used to clamp the common carotid artery and the internal
carotid artery. The distal end of the external carotid artery was
ligated and cut. A 5-cm-long nylon filament (diameter, 0.24-0.28
mm) was then inserted into the middle cerebral artery for 2 h.
Sham-operated rats were subjected to the same surgical procedure as
the rats from the tMCAO groups (34), except for the occlusion of the
middle cerebral artery. Rats were treated with a sublingual
intravenous injection of MDZ or the same volume of normal saline 10
min following ischemia (34,35).
Tetrazolium chloride (TTC) staining and
toluidine blue staining
A total of 24 and 72 h following tMCAO induction,
rats were sacrificed under deep anesthesia induced by sodium
pentobarbital injection. The number of animals used for each group
was 10. Rats randomly selected from each group were used for TTC
staining (Beyotime Institute of Biotechnology). Brains were
carefully removed and cut into six 2.0-mm-thick coronal sections.
Sections were stained with 2% TTC in normal saline for 30 min at
37°C, and fixed in 4% paraformaldehyde solution overnight at 4°C
(34).
Brain tissues were stained for 10 min at room
temperature with 0.04% w/v toluidine blue in 0.1 M sodium acetate
buffer, pH 4.0, to visualize the glycosaminoglycans, followed by 2
min counterstaining in 0.1% w/v fast green FCF at room temperature
(37).
Immunofluorescence
A total of 24 and 72 h following tMCAO induction,
rats were terminally anesthetized by 1.5% halothane (Shenzhen
YunXing Biological Technology Co., Ltd., Shenzhen, China) in air
and perfused with saline through the ascending aorta and next with
4% paraformaldehyde. Neurons cultured in vitro were also
subjected to immunofluorescence staining. Sections of terminally
anesthetized tMCAO rats were cut (10-20 µm thickness) using
a cryomicrotome and incubated with 3% goat serum (R&D Systems,
Inc., Minneapolis, MN, USA) at 37°C for 40 min. Sections were then
incubated with rabbit polyclonal primary antibody against RNA
binding fox-1 homolog 3 (1:500; cat. no. ab177487; Abcam) or rat
polyclonal primary antibody against neuron filament protein (1:500;
cat. no. ab4666; Abcam) and Alzheimer-associated neuronal thread
protein (1:500; cat. no. ab49385; Abcam) overnight at 4°C, followed
by incubation with secondary antibody (Alexa Fluor 568-donkey
anti-mouse immunoglobulin G; 1:1,000; cat. no. ab175700; Abcam) for
2 h at 25°C. Subsequently, nuclei were counterstained using DAPI
(Abcam) at room temperature for 20 min. The stained sections were
examined with a Leica fluorescence microscope (Leica Microsystems
GmbH, Wetzlar, Germany) (38,39). The number of rats used for each
group was 10.
Behavioral analysis
The BBB behavior score was used to assess the effect
of MDZ on behavior of tMCAO rats (39). Rats were divided into four groups
as described above. The assessment was performed at 0, 24 and 72 h
following treatment with MDZ. Movement, balance and coordination of
limbs, joints, and the whole body of rats was assessed and scored
0-21. Scoring 0-7 points indicated that rats exhibited few or no
hindlimb joint movements, 8-14 points indicated ataxic gait and
15-21 points indicated that rats could perform fine movements
including toe and tail movements.
Statistical analysis
Statistical analysis was performed using SPSS 13.0
software package (SPSS, Inc., Chicago, IL, USA). Results are
expressed as the mean ± standard deviation, and one-way analysis of
variance with Dunnett’s t-test was used to evaluate significance.
P<0.05 was considered to indicate a statistically significant
difference.
Results
MDZ reduces GLU-induced neuronal
cytotoxicity and apoptosis
To begin, extracted neurons were identified and
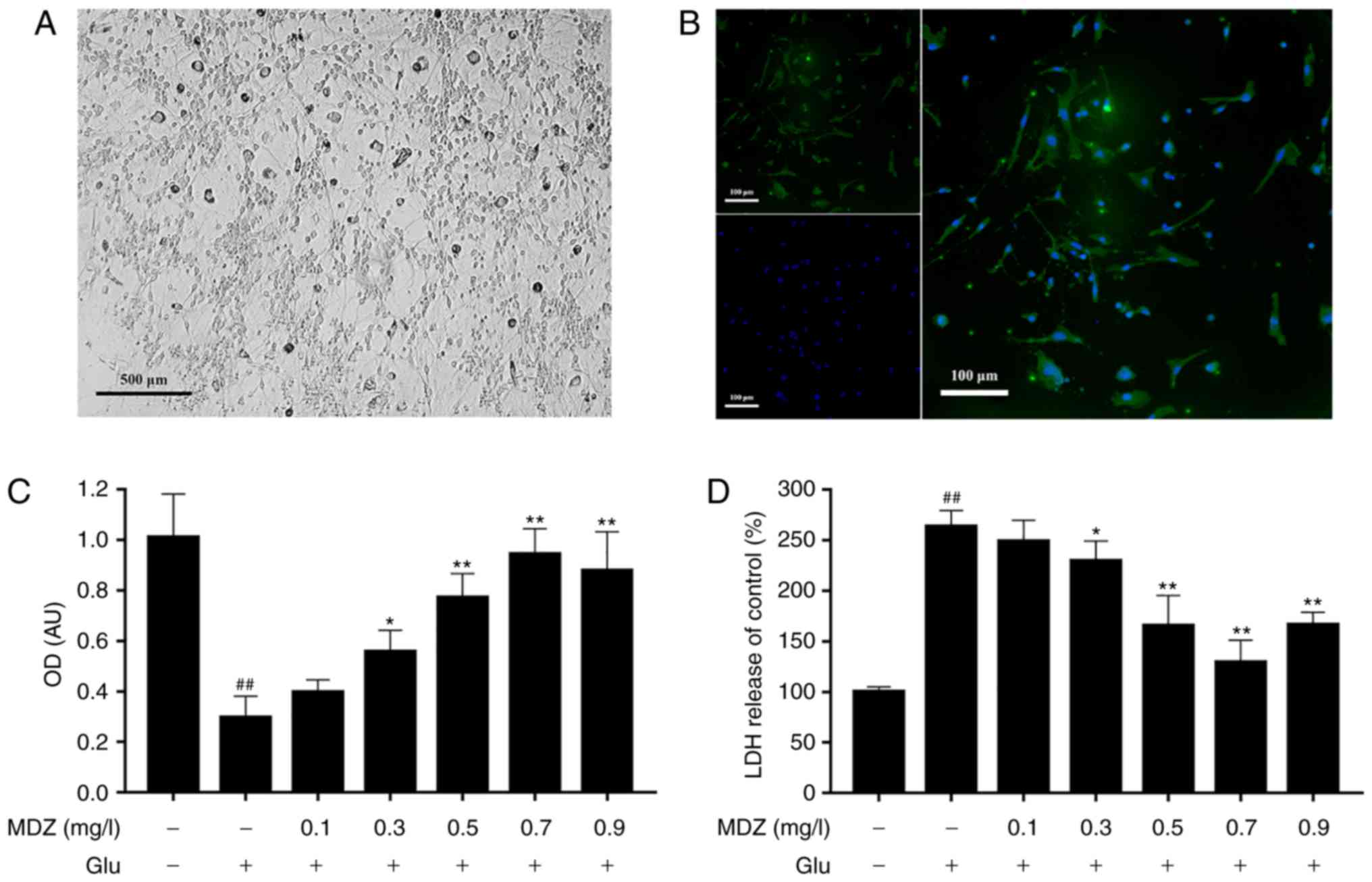
neuron purity was tested. Neurons were observed under a microscope
and were revealed to have normal morphology and clear nerve fiber
growth (Fig. 1A). In addition,
immunofluorescent staining revealed the purity of the extracted
neurons (Fig. 1B). Thus, they
were used for further experiments.
Next, the function of GLU in the induction of
cytotoxicity and apoptosis was demonstrated in vitro. The
change in staining absorbance with or without GLU treatment was
measured. The MTT assay results revealed that the number of cells
was reduced to 1/3 following GLU treatment (Fig. 1C). Furthermore, LDH release was
increased to 257% compared with the control (Fig. 1D). A significant increase in
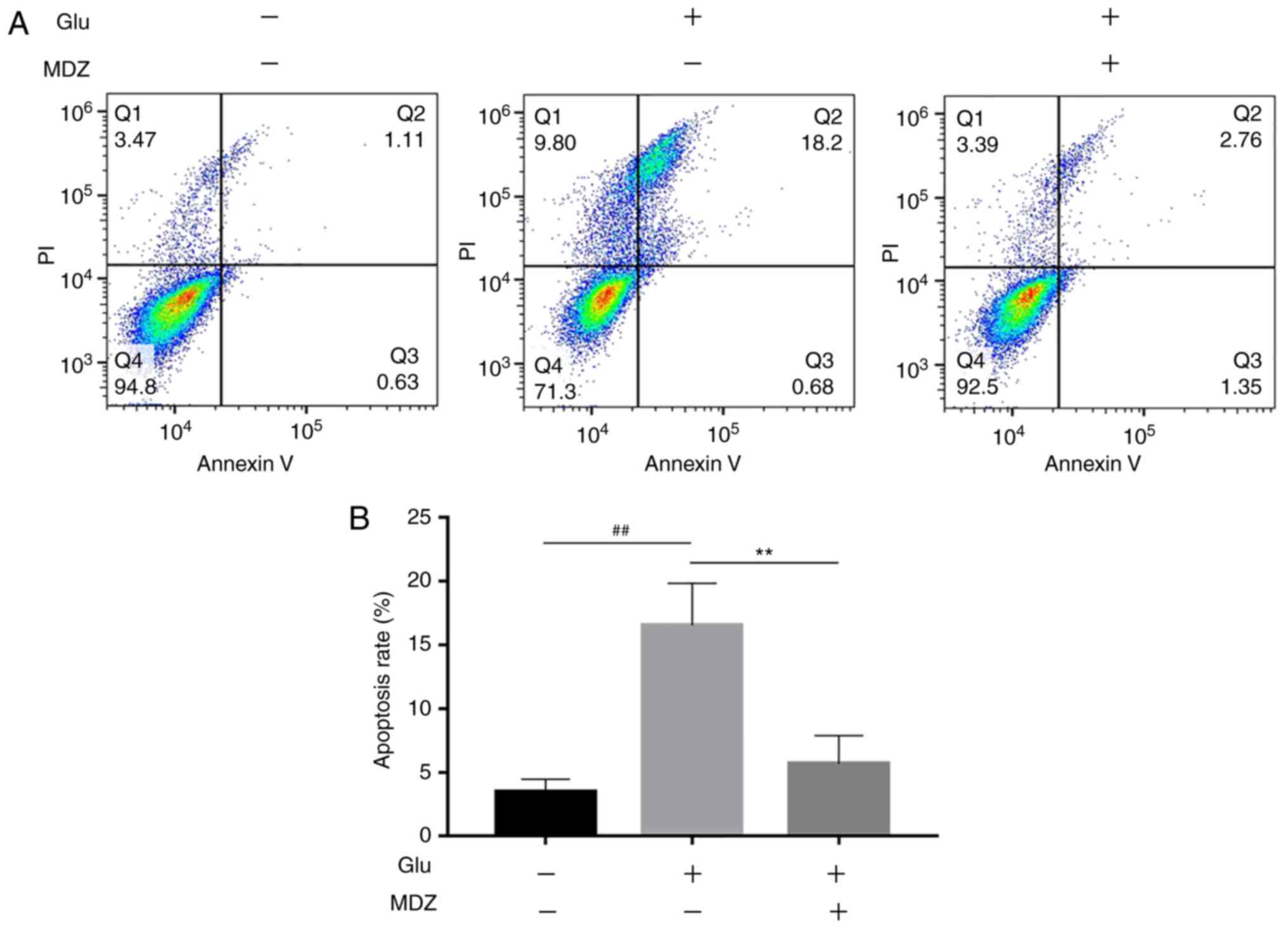
apoptotic cells following GLU treatment was observed using flow
cytometry (Fig. 2A).
Subsequently, the effect of MDZ was evaluated,
demonstrating a reduced cytotoxicity and apoptosis in vitro.
MDZ reduced neuronal death and LDH release, and the protective
effect was concentration-dependent (Fig. 1C and D), reaching an optimal
effect at 0.7 mg/ml. At this concentration, neuron density was
close to the density of the control group, with LDH release of
134%, and was thus considerably reduced compared with group 2.
Furthermore, flow cytometry results revealed that the effect of MDZ
on apoptosis (Fig. 2) was also
significant, since GLU increased apoptosis from 3.8 to 16.9%, but
apoptosis was reduced to ~5.8% following MDZ addition.
MDZ protects against GLU-induced changes
in apoptosis- associated gene expression
Next, the anti-apoptotic mechanism of MDZ treatment
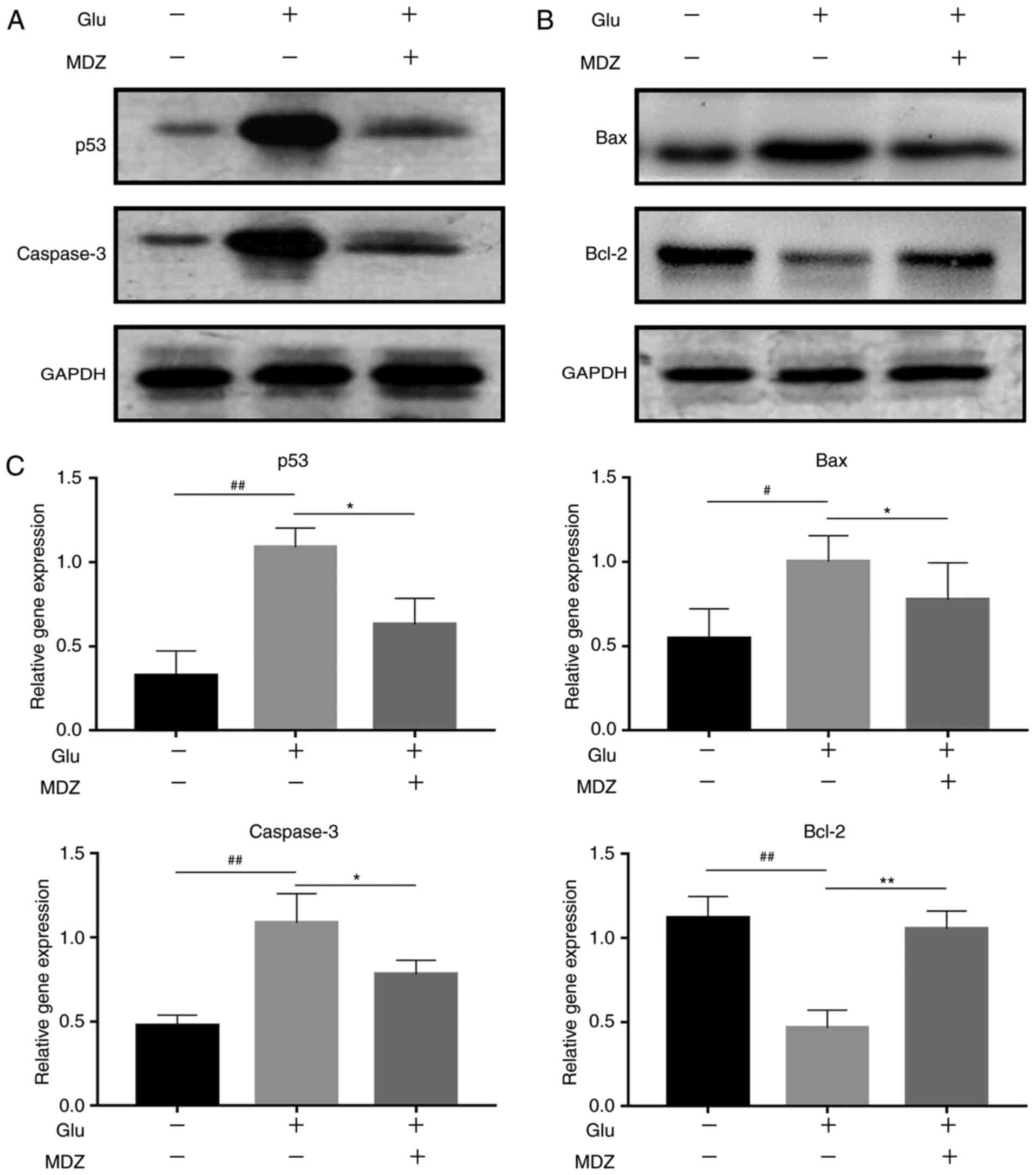
was investigated. p53 and caspase3 protein expression were
noticeably increased following GLU treatment, which may increase
cell apoptosis, and the expression of these proteins was
significantly inhibited following MDZ treatment (Fig. 3A). Furthermore, BCL-2 gene
expression was inhibited, and the expression of the apoptosis
promoter gene Bax was increased following GLU treatment. MDZ
also inhibited this effect (Fig.
3B). The PCR results confirmed the western blotting results,
and the difference in the expression of apoptosis-associated genes
between experimental groups was statistically significant
(P<0.05; Fig. 3C). Therefore,
GLU induced the expression of p53, caspase-3 and Bax, and inhibited
Bcl-2 expression, while MDZ treatment reversed these effects.
MDZ reduces LDH release from brain tissue
into the serum
Clinical use of MDZ primarily occurs through
peripheral intravenous injection, since MDZ passes through the
blood-brain barrier to exert its effects relatively quickly. The
involvement of MDZ in the reduction of cytotoxicity in peripheral
blood vessels or brain tissue was confirmed by animal experiments.
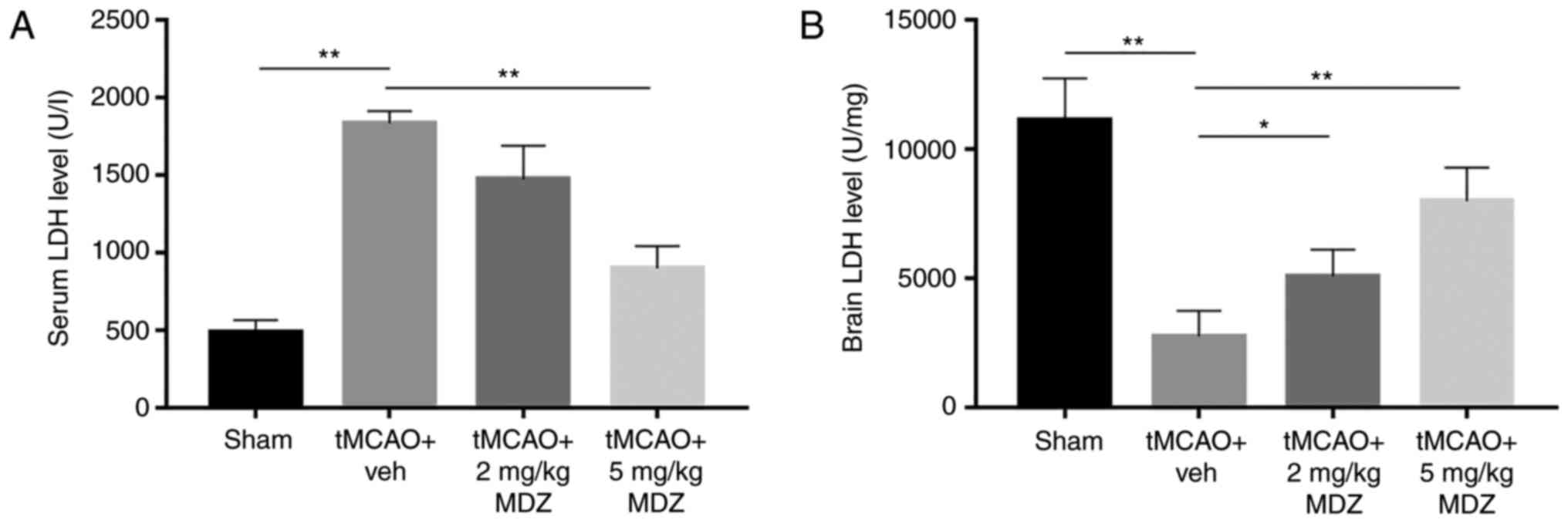
LDH concentrations in the peripheral plasma and brain tissue were
measured in different tMCAO experimental animal groups. The results
revealed that serum LDH concentration in the tMCAO animal model
significantly increased from 502±24 to 1,866±71 U/l, and decreased
following MDZ treatment in a dose-dependent manner (Fig. 4A). LDH concentration in the brain
tissue decreased from 11,073±1,631 to 2,337±276 U/l in the tMCAO
animal model, and increased with MDZ treatment in a dose-dependent
manner (Fig. 4B).
MDZ protects neurons from tMCAO damage in
the ischemic core and boundary zone
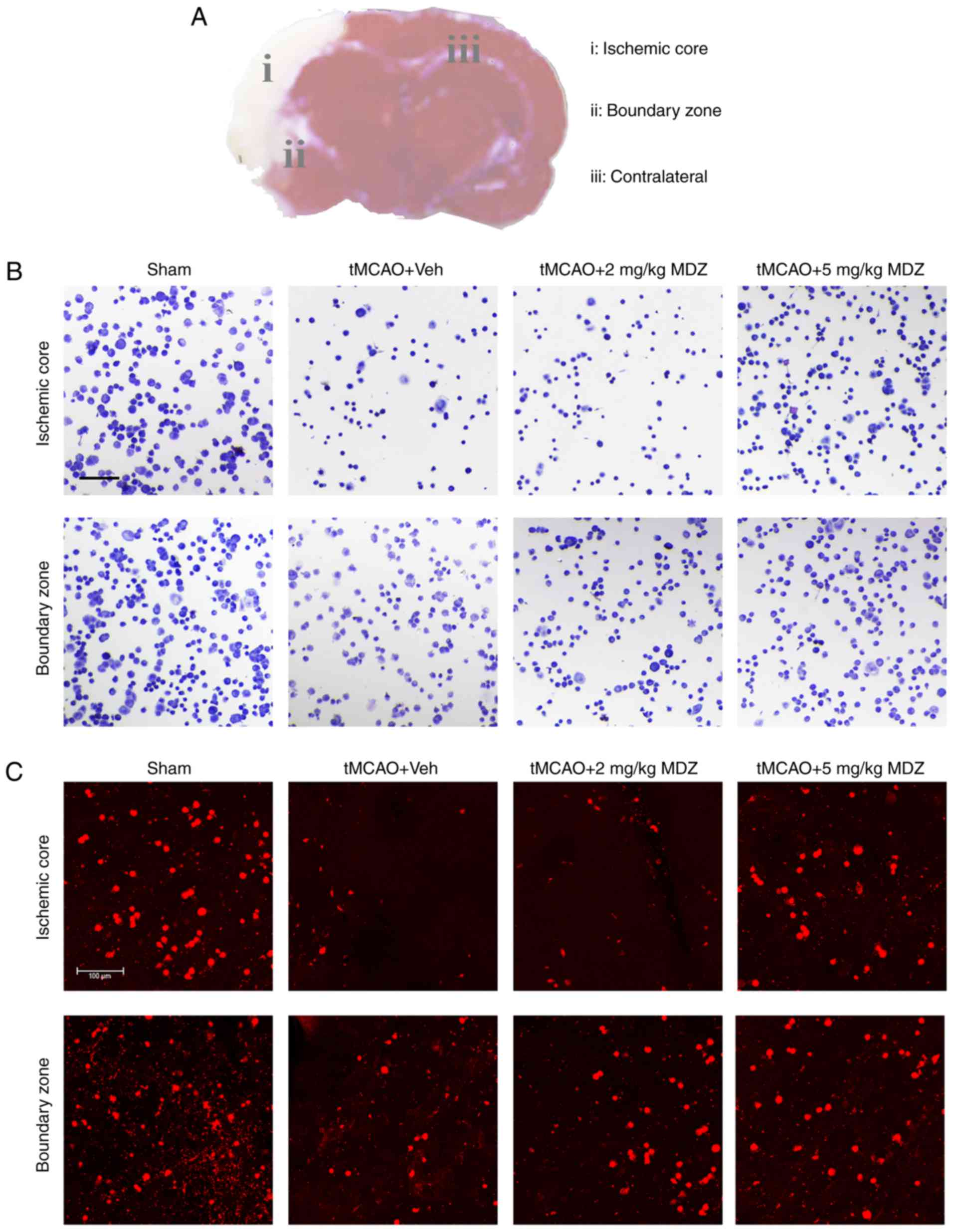
TTC staining in rat brain tissue revealed that the
tMCAO experimental animal model was successfully constructed in the
present study, as demonstrated by the ischemic and boundary areas
in rat brain tissue, which were consistent with the physiological
characteristics of cerebral infarction (Fig. 5A). After 24 and 72 h, the brain of
the corresponding treated animals was subjected to morphological
analysis and histological staining. The results of toluidine blue
staining revealed that the number of neuron in the tMCAO
experimental animals was decreased compared with the Sham group,
but was increased following MDZ treatment compared with the tMCAO +
vehicle group, in a dose-dependent manner (Fig. 5B). Immunofluorescent staining
showed similar results: tMCAO produced a remarkable neuronal
damage, while MDZ could significantly play a role in protecting
neurons. Furthermore, 5 mg/kg achieved improved results compared
with 2 mg/kg MDZ (Fig. 5C).
Furthermore, the staining results revealed that this effect
occurred in the boundary core. The number of neurons in the
boundary core also increased under MDZ treatment. At 24 and 72 h,
neuron density in the ischemic and boundary areas was statistically
analyzed. The results revealed a clear increase in cell density
following MDZ treatment compared with the tMCAO, although this
density was not considerably different between 24 and 72 h
(Fig. 5D and E). In the ischemic
core, cell density was decreased from 1.08±0.11×103 to
0.31±0.03×103 cells/mm2 in tMCAO rats treated
with the vehicle, and increased to 0.74±0.08×103
cells/mm2 following 5 mg/kg MDZ treatment within 24 h.
Furthermore, in the boundary zone, MDZ achieved a similar effect;
increasing cell density from 0.42±0.02×103 to
0.83±0.08×103 cells/mm2. The behavior of
tMCAO rats was also analyzed. The BBB method was used to assess
rats’ behavior, including movement, coordination and fine movement
of the hind limbs. The rats in the control group and sham group
were all given a score of 21. Following tMCAO operation all rats
were assessed at 7-10. A total of 3 rats were assessed 12-14
following 24 h of treatment with MDZ (2 mg/kg) and only one of them
scored 17 following 72 h of treatment with MDZ (2 mg/kg). In
addition, in the MDZ (5 mg/kg) group only two rats scored 16 at 24
h and 17 and 18 at 72 h following treatment with MDZ (data not
shown). The results indicate that BBB scores of behaviors improved
in certain rats, but not all following treatment with MDZ.
Discussion
Apoptosis is not only the main pathological change
following ischemic cerebral infarction, but is also the main cause
of poor prognosis. Cytotoxicity mediated by cytotoxic factors also
damage neurons to a certain extent. However, there is no effective
anti-apoptotic drug treatment against cerebral infarction currently
being used in clinical practice (41). In the present study, the
involvement of MDZ in the regulation of apoptosis was investigated
in an attempt to identify an optimal dose.
The results revealed that MDZ protected rat cortical
neurons from GLU-induced tMCAO models leading to neuronal damage.
This protective effect was achieved by inhibiting apoptosis and
reducing LDH release. LDH levels in the serum and brain tissue
reflected the rapid release of LDH from neurons and released from
the brain tissue into the peripheral plasma to mediate
cytotoxicity. MDZ inhibited the release of LDH and simultaneously
reduced its release from the brain tissue to the peripheral plasma
to inhibit cytotoxicity. Reducing Bcl-2 activity and increasing the
expression of p53, caspase-3 and Bax are potential mechanisms to
regulate apoptosis. As aforementioned, GLU oxidative toxicity
serves a key function in the development and progression of
neuronal death induced by cerebral ischemia. MDZ has previously
been demonstrated to inhibit the apoptosis of astrocytes (20). Consistent with previous studies,
the results of the present study suggested that MDZ increased Bcl-2
activation and inhibited apoptosis by decreasing the expression of
p53, caspase-3 and Bax. In addition, MDZ inhibited LDH release to
reduce cytotoxicity and apoptosis. MDZ treatment in the tMCAO model
also reduced neuronal damage.
The results of the present study did not show a
significant difference between 24 and 72 h, suggesting that MDZ had
a rapid and prolonged neuroprotective effect. In other words, MDZ
achieved its effect within 24 h, protecting neurons by reducing the
cytotoxic effect and apoptosis, and this effect increased with MDZ
concentration. The ability of MDZ to rapidly cross the blood-brain
barrier results in a rapid response to treatment, fewer
contraindications and no withdrawal symptoms, providing a
foundation for the clinical application against cerebral infarction
(41). The present study also
investigated the optimum MDZ concentration in vitro, and
demonstrated that MDZ achieved optimal neuroprotective effects at
0.7 mg/l. However, the ideal administration route and dose to
achieve a local 0.7 mg/l concentration in the brain requires
further study.
The present study demonstrated that the
neuroprotective function of MDZ was achieved by reducing LDH
release and regulating the expression of apoptosis-associated
genes. The inhibition of astrocyte apoptosis due to oxidative
glucose deprivation has been demonstrated to be associated with the
Janus kinase 2-signaling transducer and activator of transcription
3 signaling pathway (20).
However, this aspect was not analyzed in the present study. In
addition, MDZ appeared potentially effective in ameliorating
behavioral abnormalities of rats. Therefore, the effect of MDZ on
behavioral abnormalities and the mechanisms underlying its
inhibition of GLU-induced LDH release and neuronal apoptosis
require further study.
In conclusion, MDZ achieves its neuroprotective
effects by inhibiting neuronal apoptosis, providing a potential
novel treatment for stroke and may improve prognosis.
Acknowledgments
Not applicable.
Funding
The present study was supported by the 2016
Cultivation Fund of Hainan Medical University (grant no. HY2016-07)
and the Industry Research Project of Hainan Health and Family
Planning Commission (grant no. 1601032054A2001).
Availability of data and materials
The analyzed data sets generated during the study
are available from the corresponding author on reasonable
request.
Authors’ contributions
HY took part in all experiments and was the major
contributor in writing the manuscript. XW completed the cell
experiments and took part in data analysis. FK was responsible for
the cell culture and took part in data analysis. ZC helped complete
the cell experiments and other experiments in vivo. YM took
part in animal experiments and manuscript writing. MD was in charge
of the overall planning and gave substantial advice for experiments
and manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
All animal procedures followed the Guide for the
Care and Use of Laboratory Animals and were approved by the
Institutional Clinical Experiments Committee of the Second
Affiliated Hospital of Hainan Medical University (Haikou,
China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Shichita T, Ito M, Morita R, Komai K,
Noguchi Y, Ooboshi H, Koshida R, Takahashi S, Kodama T and
Yoshimura A: MAFB prevents excess inflammation after ischemic
stroke by accelerating clearance of damage signals through MSR1.
Nat Med. 23:723–732. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Amarenco P, Albers GW, Denison H, Easton
JD, Evans SR, Held P, Hill MD, Jonasson J, Kasner SE, Ladenvall P,
et al: Efficacy and safety of ticagrelor versus aspirin in acute
stroke or transient ischaemic attack of atherosclerotic origin: A
subgroup analysis of SOCRATES, a randomised, double-blind,
controlled trial. Lancet Neurol. 16:301–310. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Compter A, van der Worp HB, Schonewille
WJ, Vos JA, Boiten J, Nederkoorn PJ, Uyttenboogaart M, Lo RT and
Algra A: Stenting versus medical treatment in patients with
symptomatic vertebral artery stenosis: A randomised open-label
phase 2 trial. Lancet Neurol. 14:606–614. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen Y, Won SJ, Xu Y and Swanson RA:
Targeting microglial activation in stroke therapy: Pharmacological
tools and gender effects. Curr Med Chem. 21:2146–2155. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kravchick DO, Karpova A, Hrdinka M,
Lopez-Rojas J, Iacobas S, Carbonell AU, Iacobas DA, Kreutz MR and
Jordan BA: Synaptonuclear messenger PRR7 inhibits c-Jun
ubiquitination and regulates NMDA-mediated excitotoxicity. EMBO J.
35:1923–1934. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stonesifer C, Corey S, Ghanekar S,
Diamandis Z, Acosta SA and Borlongan CV: Stem cell therapy for
abrogating stroke-induced neuroinflammation and relevant secondary
cell death mechanisms. Prog Neurobiol. 158:94–131. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shuros AC, Salo RW, Florea VG, Pastore J,
Kuskowski MA, Chandrashekhar Y and Anand IS: Ventricular
preexcitation modulates strain and attenuates cardiac remodeling in
a swine model of myocardial infarction. Circulation. 116:1162–1169.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sun F, Li X, Duan WQ, Tian W, Gao M, Yang
J, Wu XY, Huang D, Xia W, Han YN, et al: Transforming growth
factor-β receptor III is a potential regulator of ischemia-induced
cardiomyocyte apoptosis. J Am Heart Assoc. 6:pii:e0053572017.
View Article : Google Scholar
|
|
9
|
Wang C and Youle RJ: Predominant
requirement of Bax for apoptosis in HCT116 cells is determined by
Mcl-1’s inhibitory effect on Bak. Oncogene. 31:3177–3189. 2012.
View Article : Google Scholar
|
|
10
|
Arlet JB, Ribeil JA, Guillem F, Negre O,
Hazoume A, Marcion G, Beuzard Y, Dussiot M, Moura IC, Demarest S,
et al: HSP70 sequestration by free α-globin promotes ineffective
erythropoiesis in β-thalassaemia. Nature. 514:242–246. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li H, Lv B, Kong L, Xia J, Zhu M, Hu L,
Zhen D, Wu Y, Jia X, Zhu S and Cui H: Nova1 mediates resistance of
rat pheochromocytoma cells to hypoxia-induced apoptosis via the
Bax/Bcl-2/caspase-3 pathway. Int J Mol Med. 40:1125–1133. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mathieu J, Zhou W, Xing Y, Sperber H,
Ferreccio A, Agoston Z, Kuppusamy KT, Moon RT and Ruohola-Baker H:
Hypoxia-inducible factors have distinct and stage-specific roles
during reprogramming of human cells to pluripotency. Cell Stem
Cell. 14:592–605. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Llambi F, Wang YM, Victor B, Yang M,
Schneider DM, Gingras S, Parsons MJ, Zheng JH, Brown SA, Pelletier
S, et al: BOK is a non-canonical BCL-2 family effector of apoptosis
regulated by ER-associated degradation. Cell. 165:421–433. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Grinkevich VV, Nikulenkov F, Shi Y, Enge
M, Bao W, Maljukova A, Gluch A, Kel A, Sangfelt O and Selivanova G:
Ablation of key oncogenic pathways by RITA-reactivated p53 is
required for efficient apoptosis. Cancer Cell. 31:724–726. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Andrabi SA, Kang HC, Haince JF, Lee YI,
Zhang J, Chi Z, West AB, Koehler RC, Poirier GG, Dawson TM and
Dawson VL: Iduna protects the brain from glutamate excitotoxicity
and stroke by interfering with poly(ADP-ribose) polymer-induced
cell death. Nat Med. 17:692–699. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Weber NC, Riedemann I, Smit KF, Zitta K,
van de Vondervoort D, Zuurbier CJ, Hollmann MW, Preckel B and
Albrecht M: Plasma from human volunteers subjected to remote
ischemic preconditioning protects human endothelial cells from
hypoxia-induced cell damage. Basic Res Cardiol. 110:172015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Drake-Brockman TF, Ramgolam A, Zhang G,
Hall GL and von Ungern-Sternberg BS: The effect of endotracheal
tubes versus laryngeal mask airways on perioperative respiratory
adverse events in infants: A randomised controlled trial. Lancet.
389:701–708. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Scott RC: Buccal midazolam as rescue
therapy for acute seizures. Lancet Neurol. 4:592–593. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Osterop SF, Virtanen MA, Loepke JR, Joseph
B, Loepke AW and Vutskits L: Developmental stage-dependent impact
of midazolam on calbindin, calretinin and parvalbumin expression in
the immature rat medial prefrontal cortex during the brain growth
spurt. Int J Dev Neurosci. 45:19–28. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu L, You Q, Tu Y, Li Q, Zheng L, Li X,
Gu J and Wang G: Midazolam inhibits the apoptosis of astrocytes
induced by oxygen glucose deprivation via targeting JAK2-STAT3
signaling pathway. Cell Physiol Biochem. 35:126–136. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Makinodan M, Rosen KM, Ito S and Corfas G:
A critical period for social experience-dependent oligodendrocyte
maturation and myelination. Science. 337:1357–1360. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Marion E, Song OR, Christophe T, Babonneau
J, Fenistein D, Eyer J, Letournel F, Henrion D, Clere N, Paille V,
et al: Mycobacterial toxin induces analgesia in buruli ulcer by
targeting the angiotensin pathways. Cell. 157:1565–1576. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Novoa-Her ran S, Umaña-Perez A, Canals F
and Sanchez-Gomez M: Serum depletion induces changes in protein
expression in the trophoblast-derived cell line HTR-8/SVneo. Cell
Mol Biol Lett. 21:222016. View Article : Google Scholar
|
|
24
|
Zou ZK, Huang YQ, Zou Y, Zheng XK and Ma
XD: Silencing of LSD1 gene modulates histone methylation and
acetylation and induces the apoptosis of JeKo-1 and MOLT-4 cells.
Int J Mol Med. 40:319–328. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chaudhari U, Nemade H, Gaspar JA,
Hescheler J, Hengstler JG and Sachinidis A: MicroRNAs as early
toxicity signatures of doxorubicin in human-induced pluripotent
stem cell-derived cardiomyocytes. Arch Toxicol. 90:3087–3098. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kumamaru H, Ohkawa Y, Saiwai H, Yamada H,
Kubota K, Kobayakawa K, Akashi K, Okano H, Iwamoto Y and Okada S:
Direct isolation and RNA-seq reveal environment-dependent
properties of engrafted neural stem/progenitor cells. Nat Commun.
3:11402012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Huang WH, Liao WR and Sun RX: Astragalus
polysaccharide induces the apoptosis of human hepatocellular
carcinoma cells by decreasing the expression of Notch1. Int J Mol
Med. 38:551–557. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang SJ, Xu TT, Li L, Xu YM, Qu ZL, Wang
XC, Huang SQ, Luo Y, Luo NC, Lu P, et al: Bushen-Yizhi formula
ameliorates cognitive dysfunction through SIRT1/ER stress pathway
in SAMP8 mice. Oncotarget. 8:49338–49350. 2017.PubMed/NCBI
|
|
29
|
Yu Z, Rayile A, Zhang X, Li Y and Zhao Q:
Ulinastatin protects against lipopolysaccharide-induced cardiac
microvascular endothelial cell dysfunction via downregulation of
lncRNA MALAT1 and EZH2 in sepsis. Int J Mol Med. 39:1269–1276.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Starokadomskyy P, Gemelli T, Rios JJ, Xing
C, Wang RC, Li H, Pokatayev V, Dozmorov I, Khan S, Miyata N, et al:
DNA polymerase-α regulates the activation of type I interferons
through cytosolic RNA:DNA synthesis. Nat Immunol. 17:495–504. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang XD, Baladandayuthapani V, Lin H,
Mulligan G, Li B, Esseltine DL, Qi L, Xu J, Hunziker W, Barlogie B,
et al: Tight junction protein 1 modulates proteasome capacity and
proteasome inhibitor sensitivity in multiple myeloma via
EGFR/JAK1/STAT3 signaling. Cancer Cell. 29:639–652. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shen Z, Zheng Y, Wu J, Chen Y, Wu X, Zhou
Y, Yuan Y, Lu S, Jiang L, Qin Z, et al: PARK2-dependent mitophagy
induced by acidic postconditioning protects against focal cerebral
ischemia and extends the reperfusion window. Autophagy. 13:473–485.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
National Research Council (US) Institute
for Laboratory Animal Research: Guide for the Care and Use of
Laboratory Animals. National Academies Press (US); Washington, DC:
1996
|
|
34
|
Chen B, Tao J, Lin Y, Lin R, Liu W and
Chen L: Electro-acupuncture exerts beneficial effects against
cerebral ischemia and promotes the proliferation of neural
progenitor cells in the cortical peri-infarct area through the
Wnt/β-catenin signaling pathway. Int J Mol Med. 36:1215–1222. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
van Hout GP, Bosch L, Ellenbroek GH, de
Haan JJ, van Solinge WW, Cooper MA, Arslan F, de Jager SC,
Robertson AA, Pasterkamp G and Hoefer IE: The selective
NLRP3-inflammasome inhibitor MCC950 reduces infarct size and
preserves cardiac function in a pig model of myocardial infarction.
Eur Heart J. 38:828–836. 2017.
|
|
36
|
Alvarez-Saavedra M, De Repentigny Y,
Lagali PS, Raghu Ram EV, Yan K, Hashem E, Ivanochko D, Huh MS, Yang
D, Mears AJ, et al: Snf2h-mediated chromatin organization and
histone H1 dynamics govern cerebellar morphogenesis and neural
maturation. Nat Commun. 5:41812014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kaminski MM, Tosic J, Kresbach C, Engel H,
Klockenbusch J, Müller AL, Pichler R, Grahammer F, Kretz O, Huber
TB, et al: Direct reprogramming of fibroblasts into renal tubular
epithelial cells by defined transcription factors. Nat Cell Biol.
18:1269–1280. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lee JS, Chang EH, Koo OJ, Jwa DH, Mo WM,
Kwak G, Moon HW, Park HT, Hong YB and Choi BO: Pmp22 mutant
allele-specific siRNA alleviates demyelinating neuropathic
phenotype in vivo. Neurobiol Dis. 100:99–107. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang H, Li P, Xu N, Zhu L, Cai M, Yu W and
Gao Y: Paradigms and mechanisms of inhalational anesthetics
mediated neuroprotection against cerebral ischemic stroke. Med Gas
Res. 6:194–205. 2016. View Article : Google Scholar
|
|
40
|
Basso DM, Beattie MS and Bresnahan JC: A
sensitive and reliable locomotor rating scale for open field
testing in rats. J Neurotrauma. 12:1–21. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Matsuo N and Morita T: Efficacy, safety,
and cost effectiveness of intravenous midazolam and flunitrazepam
for primary insomnia in terminally ill patients with cancer: A
retrospective multicenter audit study. J Palliat Med. 10:1054–1062.
2007. View Article : Google Scholar : PubMed/NCBI
|