Introduction
Rheumatoid arthritis (RA) is a common systemic and
autoimmune inflammatory disease of the joints, characterized by
inflammation, synovial hyperplasia and abnormal immune responses
(1-3). Fibroblast-like synoviocytes (FLSs)
in the RA synovium (referred to as RA-FLSs) may affect the vascular
endothelium (4). Furthermore,
accumulating evidence has suggested that RA-FLSs serve crucial
roles in the destruction process of the joints and the development
of RA (5-7). RA-FLSs have aggressive properties
similar to those exhibited by cancer cells, including
hyperproliferation, defective apoptosis and invasiveness (8). Thus, the prevention of RA-FLS
proliferation and induction of apoptosis may be an effective
therapeutic strategy for the treatment of RA.
Protein kinase B (Akt) is a serine/threonine kinase
and downstream effector of phosphoinositide 3-kinase (PI3K).
Activated PI3K increases the intracellular content of
phosphatidylinoside-4,5-diphosphate and
phosphati-dylinoside-3,4,5-triphosphate, positively inducing Akt
phosphorylation (9). The
mitogen-activated protein kinase (MAPK), extracellular
signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK) and
p38 signaling pathways are constructed around a three-tiered
phosphorylation cascade. The MAPK signaling pathway-associated
proteins are known to be involved in the mediation of inflammation,
and serve a critical role in the excessive proliferation and
invasion of synovial fibroblasts of RA (10-12). Janus kinase (JAK) activation leads
to the activation of downstream substrates and of signal transducer
and activator of transcription (STAT) proteins, followed by their
nuclear translocation and subsequent activation of target genes
(13). The STAT3 signaling
pathway is responsible for transmitting the outer signals of the
cell to the nucleus and expressing the effects of biological
stimulation through the induction of target gene transcription.
Dysfunctional JAK/STAT signaling has been implicated in various
hematological and immunological disorders, as well as other
pathological inflammatory conditions (14-16). Reactive oxygen species (ROS),
including oxygen ions, peroxides and oxygen-free radicals, are the
main molecules that the body produces during oxidative stress.
Highly reactive oxygen-free radicals are considered to be involved
in the pathogenesis of RA (17-21).
The use of traditional Chinese medicine is promising
for adjuvant therapy due to the low cost and low toxicity of the
formulations used (22).
Cryptotanshinone (CT) is a fat-soluble anthraquinone derivative and
a major tanshinone found in Salvia miltiorrhiza Bunge
(Lamiaceae) (17). It has a wide
range of pharmacological properties, such as antitumor,
anti-bacterial and anti-inflammatory properties (22-25). A number of studies have reported
that CT inhibits the growth of certain types of cancer cells or
induces cancer cell apoptosis, including in lung, breast and
prostate cancer (26-29). However, the effects of CT on the
proliferation and apoptosis of RA-FLSs, and the underlying
molecular mechanisms remain unclear.
In the present study, the effects of CT on RA-FLS
proliferation and apoptosis were evaluated, and the associated
molecular pathways were examined. In addition, the roles of the
MAPK, Akt and STAT3 signaling pathways associated with ROS
generation were investigated in RA-FLSs.
Materials and methods
Chemicals and reagents
CT (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany)
was dissolved (20 mM CT in 100% dimethyl sulfoxide) to obtain a
stock solution and stored at −20°C. The stock solution was diluted
with cell culture media prior to further use.
Patients and samples
Informed consent was obtained from RA patients prior
to inclusion in the present study, and the procedure was approved
by the Ethical Committee of the Fifth Affiliated Hospital of Harbin
Medical University (Daqing, China). Synovial tissue samples were
obtained from RA patients (including 3 females and 3 males aged
53-66 years) undergoing synovectomy or joint replacement in the
Fifth Affiliated Hospital of Harbin Medical University. The
synovial tissues were minced into small pieces and digested with 2
mg/ml collagenase type I at 37°C for 4 h. Subsequent to the
enzymatic dispersion of synovial tissues, synovial cells were
pelleted by centrifugation (1,200 × g for 3 min at room
temperature) and cultured in Dulbecco’s modified Eagle’s medium
(DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) at
37°C in a humidified atmosphere containing 5% CO2. After
24 h of incubation, the adherent cells were collected and cultured
in DMEM, and are referred to as the primary RA-FLS cells.
Cell lines and culture
The HFLS-RA (cat. no. CBR130803), MH7A (cat. no.
CBR130742) and normal synovial HFLS (cat. no. CBR130948) cells were
purchased from Saiqi Biological Engineering Co., Ltd. (Shanghai,
China). Cells were cultured in DMEM supplemented with 10% fetal
bovine serum (Gibco; Thermo Fisher Scientific, Inc.), 100
µg/ml penicillin and 100 U/ml streptomycin in a humidified
5% CO2 incubator at 37°C.
Cell Counting kit-8 (CCK-8) proliferation
test
Primary RA-FLS, HFLS-RA, MH7A and HFLS cells were
seeded on 96-well plates (1×104 cells/well) and treated
with different concentrations of CT (1, 3, 10, 30 and 100
µmol/l) for 24 h. Prior to proliferation detection, 10
µl CCK-8 reagent (Beyotime Institute of Biotechnology,
Shanghai, China) was added into each well and incubated at 37°C for
1.5 h. Finally, the absorbance was measured by a microplate reader
(A490) and the cell survival rates were calculated.
Fluorescence microscopy
A total of 5×105 HFLS-RA and MH7A
cells/well were seeded in 6-well plates and cultured overnight.
Next, the cells were treated with 5 µmol/l CT for different
time durations (0, 3, 6, 12 and 24 h). According to the
manufacturer’s manual that was provided with the Apoptosis
Detection kit (Beyotime Institute of Biotechnology), cells were
stained with 10 µl Annexin V-FITC and 5 µl propidium
iodide (PI) for 15 min, and the fluorescence intensity of each
sample was determined by fluorescence microscopy (Leica
Microsystem, Wetzlar, Germany).
Flow cytometry
A total of 5×105 HFLS-RA were seeded in
6-well plates and cultured overnight. HFLS-RA cells were pretreated
with N-acetyl cysteine (NAC, 5 mmol/l, Sigma-Aldrich; Merck KGaA)
for 30 min and incubated with CT (5 µmol/l) for 24 h. Then,
cells were stained with 10 µl Annexin V-FITC and 5 µl
PI for 15 min, and the number of apoptotic cells were detected by
flow cytometry (Beckman Coulter, Inc., Brea, CA, USA).
In order to measure the ROS levels, HFLS-RA cells
were treated with CT (5 µmol/l) for 0, 3, 6, 12 and 24 h,
following which the cells were incubated with
2′,7′-dichlorofluorescein-diacetate (DCFH-DA; Merck, Shanghai,
China) for 30 min at 37°C. Next, cells were washed 2-3 times with
PBS and harvested by centrifugation at 1,200 × g for 5 min at room
temperature. The intracellular ROS levels were subsequently
analyzed by flow cytometry.
Western blot analysis
Cells were lysed in ice-cold
radioimmunoprecipitation buffer (50 mmol/l Tris (pH 7.4), 150
mmol/l NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, 20
mg/ml AEBSF, 0.5 mg/ml pepstatin, 0.5 mg/ml leupeptin and 2 mg/ml
aprotinin; (Beyotime Institute of Biotechnology) at 4°C for 30 min
by shaking every 5 min and then separated by centrifugation at 4°C
and 12,000 × g for 30 min. The protein concentrations were
determined using Bradford reagent (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA). Next, 5X buffer was added to the total
supernatants (30 µg), and proteins were resolved by 12%
sodium dodecyl sulfate-polyacrylamide gel electrophoresis.
Subsequently, the proteins were transferred to nitrocellulose (NC)
membranes, which were blocked in 5% skim milk for 2 h at room
temperature. The NC membranes were then incubated overnight at 4°C
with the following primary antibodies (all purchased from Santa
Cruz Biotechnology, Inc., Dallas, TX, USA): Mouse monoclonal
antibodies against α-tubulin (1:2,500; cat. no. sc-8035), B-cell
lymphoma 2 (Bcl-2; 1:1,500; cat. no. sc-7382), Bcl-2-associated
death promoter (Bad; 1:1,500; cat. no. sc-8044), cleaved caspase-3
(1:1,500; cat. no. sc-373730), cleaved poly (ADP-ribose) polymerase
(PARP; 1:1,500; cat. no. sc-8007), phosphorylated ERK [p-ERK (Tyr
204); 1:1,500; cat. no. sc-8059], p-JNK (Thr 183 and Tyr 185,
1:1,500; cat. no. sc-6254), JNK (1:1,500; cat. no. sc-7345), p-p38
(1:1,500; cat. no. sc-7973), p-STAT3 (Tyr 705, 1:1,500; cat. no.
sc-8059) and STAT3 (1:1,500; cat. no. sc-8019); and rabbit
polyclonal antibodies against ERK2 (1:1,500; cat. no. sc-154),
p38α/β (1:1,500; cat. no. sc-7972), Akt1/2/3 (1:1,500; cat. no.
sc-8312) and p-Akt1/2/3 (Ser 473, 1:2,500; cat. no. sc-7985-R).
Following washing with Tris-buffered saline/Tween 20 (TBST), the NC
membranes were incubated with horseradish peroxidase (HRP)-labeled
goat anti-rabbit IgG (1:5,000; cat. no. ZB-2301) and goat anti-rat
IgG (1:5,000; cat. no. ZB-2305) were used as secondary antibodies
for 2 h at room temperature. After the membranes were washed again
with TBST, an enhanced chemiluminescence reagent (Thermo Fisher
Scientific, Inc.) was used to detect the relative intensities of
proteins using Amersham imager 600 (AI600; GE Healthcare, Chicago,
IL, USA). The band intensities were analyzed by ImageJ software
(National Institutes of Health, Bethesda, MD, USA), and the
relative intensity of protein expression levels was calculated by
SPSS version 18.0 statistical software (SPSS, Inc., Chicago, IL,
USA).
Statistical analysis
Data are presented as the mean ± standard deviation,
and all the experiments were replicated three times. Continuous
data were analyzed by one-way analysis of variance followed by
Tukey’s post hoc tests using SPSS version 18.0 statistical
software. P<0.05 was considered to indicate a difference that
was statistically significant.
Results
CT exhibits cytotoxic effects in primary
RA-FLS, HFLS-RA and MH7A cells
To determine the effects of CT on RA-FLS survival,
primary RA-FLS, HFLS-RA, MH7A and normal HFLS cells were treated
with different concentrations of CT (1, 3, 10, 30 and 100
µmol/l) for 24 h, and the cell viabilities were evaluated by
the CCK-8 assay. As shown in Fig.
1A-C, CT significantly decreased the viabilities of primary
RA-FLS, HFLS-RA and MH7A cells in a dose-dependent manner. The half
maximal inhibitory concentration (IC50) values of
primary RA-FLS, HFLS-RA and MH7A cells were 6.69±0.97, 2.56±0.42
and 7.10±0.94 µmol/l, respectively. Furthermore, CT
exhibited low cytotoxic effects on HFLS cells, which had an
IC50 value >100 µmol/l; however, significance
was observed compared with the control (Fig. 1D).
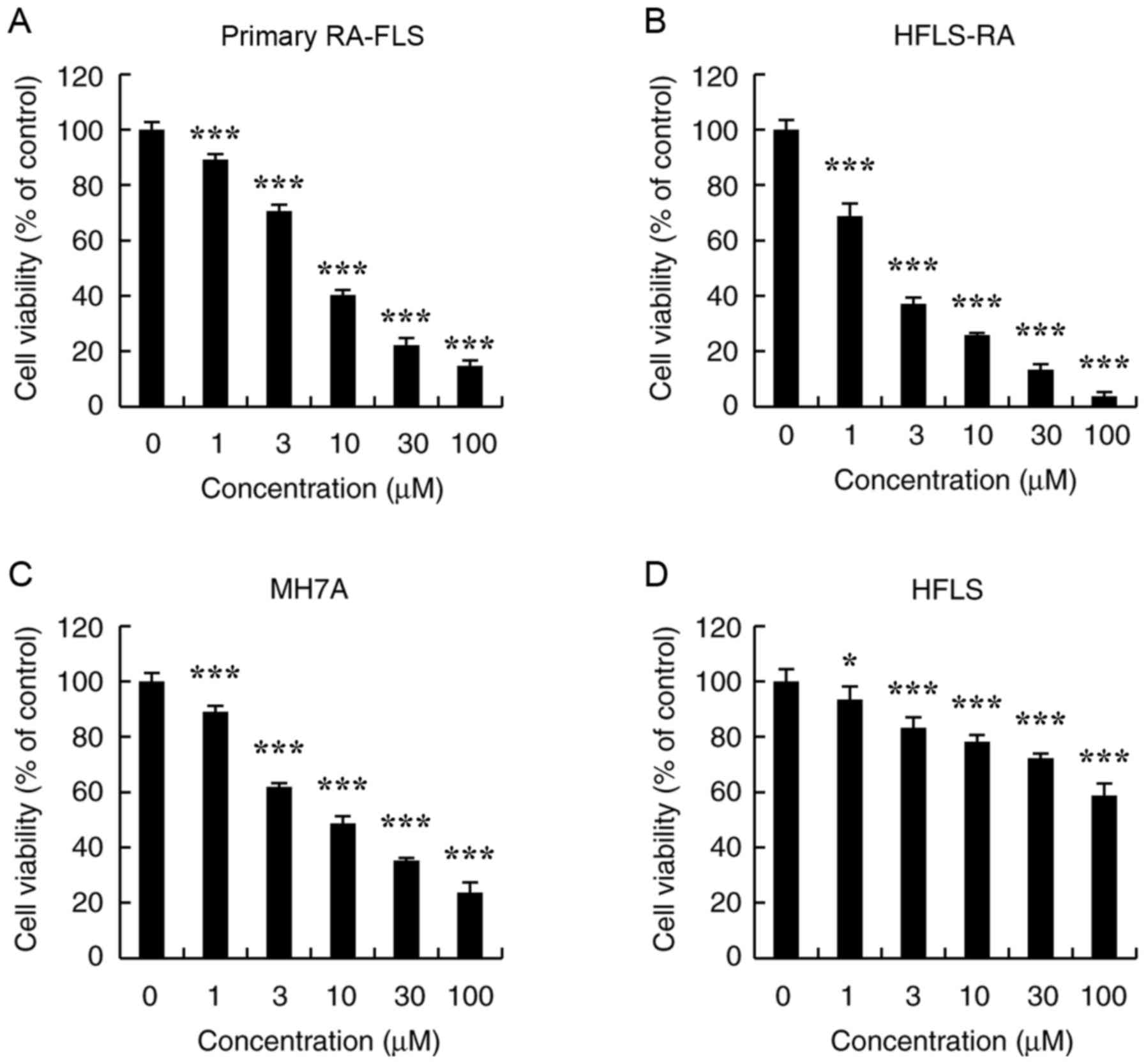 | Figure 1Effects of CT on the proliferation of
primary RA-FLS, HFLS-RA, MH7A and HFLS cells. Cells were treated
with different concentrations of CT (0, 1, 3, 10, 30 and 100
µmol/l) for 24 h, following which the cell viability was
evaluated by a Cell Counting kit-8 assay. The cytotoxic effects of
CT on (A) Primary RA-FLS, (B) HFLS-RA, (C) MH7A and (D) HFLS cell
viability are displayed. *P<0.05 and
***P<0.001, vs. control (0 µmol/l CT) group.
CT, cryptotanshinone. |
CT induces apoptosis by regulating the
mitochondrial pathways in HFLS-RA cells
To determine whether CT induced apoptosis in
RA-FLSs, HFLS-RA and MH7A cells were treated with 5 µmol/l
CT for different time points (0, 3, 6, 12 and 24 h), and apoptosis
was examined by Annexin V-FITC/PI double staining and flow
cytometry. As shown in Fig. 2A-D,
the fluorescence intensity significantly increased with the
increase in the duration of CT treatment. In addition, apoptosis
gradually increased in HFLS-RA and MH7A cells in a time-dependent
manner (Fig. 2E-H). These results
suggest that CT may be a potential inducer of apop-tosis in
RA-FLSs.
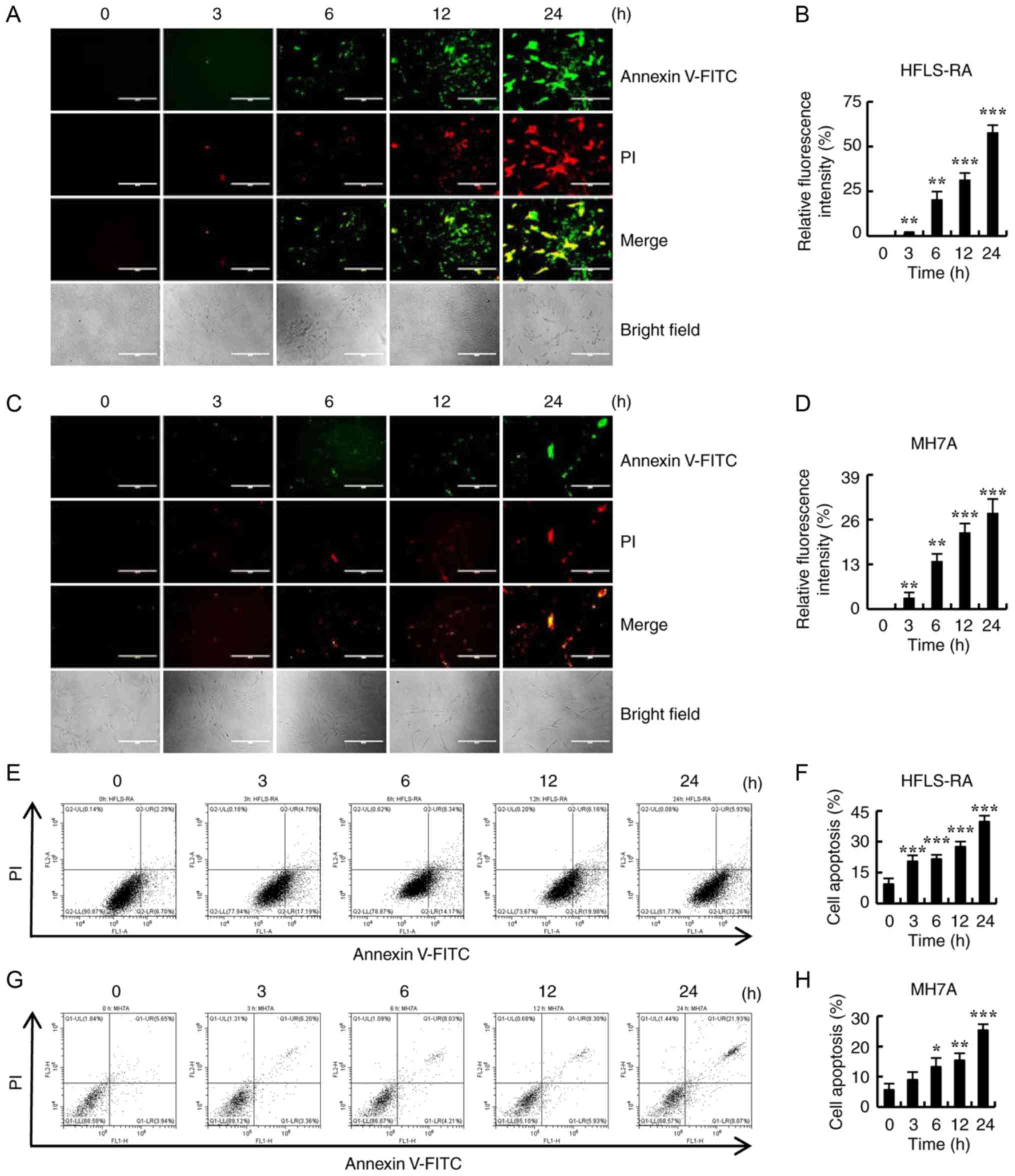 | Figure 2Effects of CT on the apoptosis of
HFLS-RA and MH7A cells. Cells were treated with 5 µmol/l CT
for different time durations (0, 3, 6, 12 and 24 h). (A)
Fluorescence microscopic images (magnification, ×400; scale bars,
200 µm) and (B) quantified fluorescence intensity of HFLS-RA
cells stained with Annexin V-FITC/PI. (C) Fluorescence microscopic
images (magnification, ×400; scale bars, 200 µm) and (D)
quantified fluorescence intensity of MH7A cells stained with
Annexin V-FITC/PI. (E) Flow cytometry results and (F) quantified
cell apoptosis rate in HFLS-RA cells subsequent to staining with
Annexin V-FITC/PI. (G) Flow cytometry results and (H) quantified
cell apoptosis rate in MH7A cells subsequent to staining with
Annexin V-FITC/PI. *P<0.05, **P<0.01
and ***P<0.001, vs. control (0 h) group. CT,
cryptotanshinone. |
CT-induced apoptosis was more notable in HFLS-RA
cells compared with in MH7A cells; thus, HFLS-RA cells were used as
the representative RA-FLS cells in subsequent experiments. As shown
in Fig. 3A and B, CT treatment
markedly downregulated the expression of Bcl-2, while it
upregulated the protein expression levels of Bad, cleaved caspase-3
and cleaved PARP. These results revealed that CT induced apoptosis
through the mitochondrial pathway.
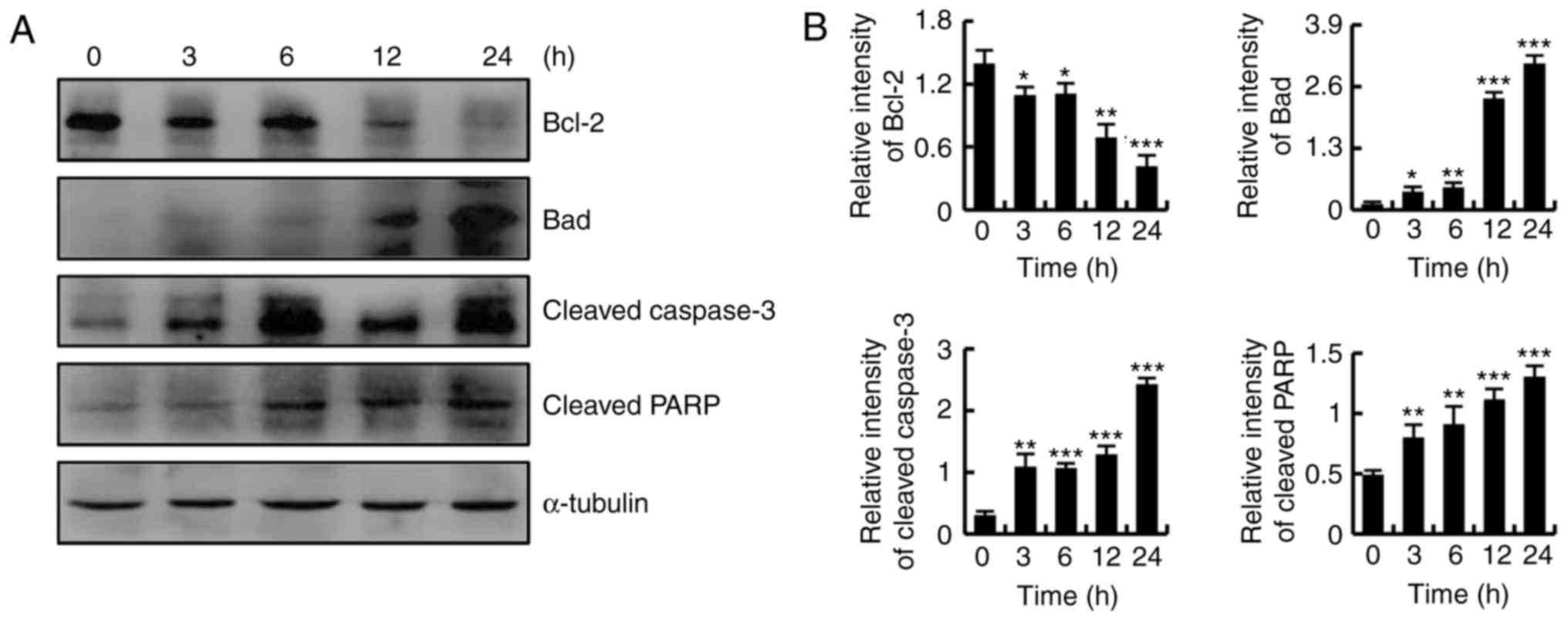 | Figure 3CT induced apoptosis by regulating
the mitochondrial pathway in HFLS-RA cells. Cells were treated with
5 µmol/l CT for various time durations (0, 3, 6, 12 and 24
h). (A) Western blot analysis results and (B) quantified protein
expression levels of Bcl-2, Bad, cleaved caspase-3 and cleaved PARP
in HFLS-RA cells. *P<0.05, **P<0.01 and
***P<0.001, vs. control (0 h) group. CT,
cryptotanshinone; Bcl-2, B-cell lymphoma 2; Bad, Bcl-2-associated
death promoter; PARP, poly (ADP-ribose) polymerase. |
CT induces apoptosis by regulating the
MAPK, Akt and STAT3 signaling pathways in HFLS-RA cells
To determine whether CT induced apoptosis by
regulating the Akt, MAPK and STAT3 signaling pathways, HFLS-RA
cells were treated with 5 µmol/l CT for different time
points (0, 3, 6, 12 and 24 h), and protein expression levels were
detected by western blotting. As shown in Fig. 4A and B, CT exposure increased the
protein expression levels of p-p38 and p-JNK, and downregulated the
expression levels of p-Akt, p-ERK and p-STAT3, in a time-dependent
manner. These results revealed that CT induced apoptosis in HFLS-RA
cells via the MAPK, Akt and STAT3 signaling pathways.
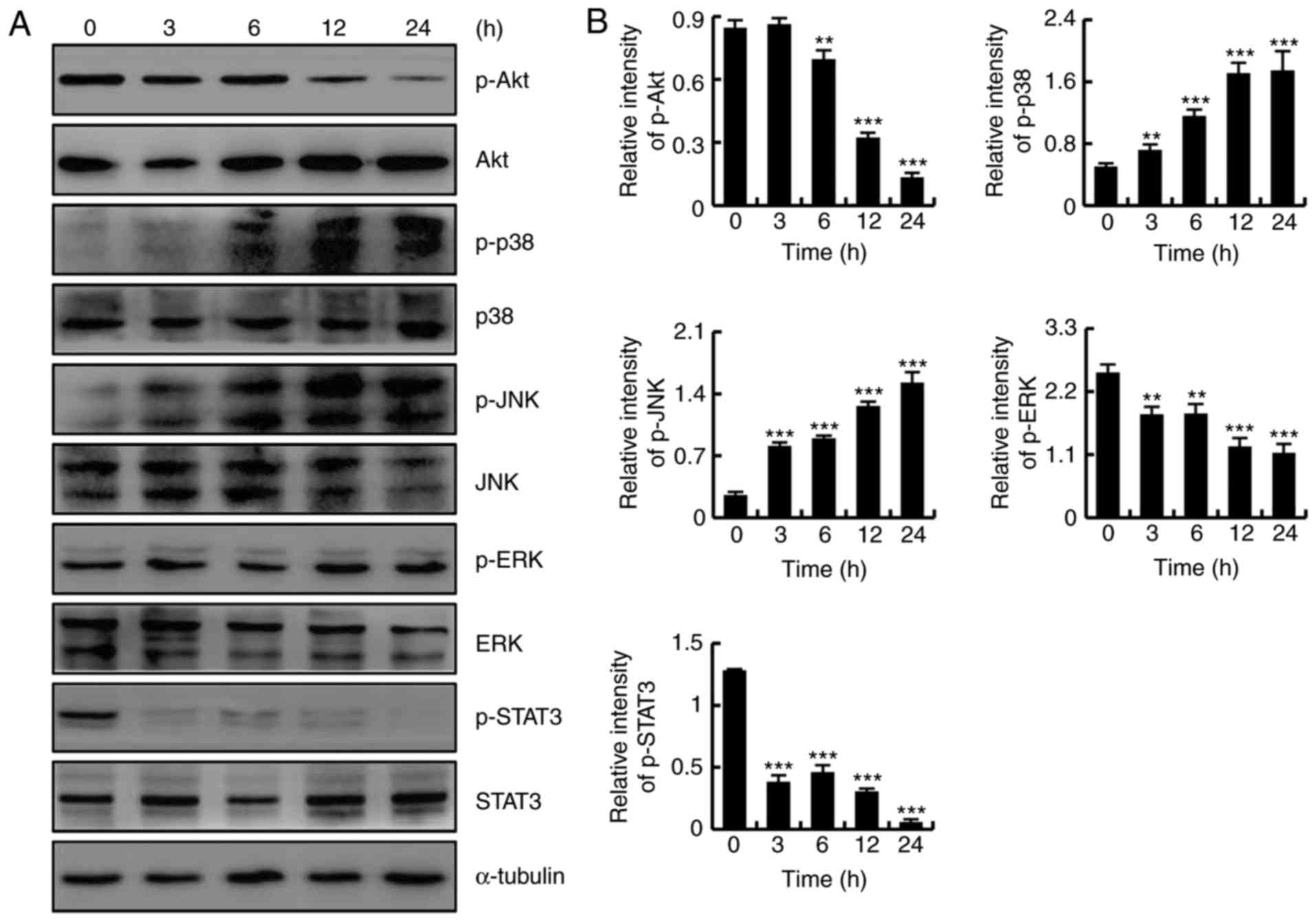 | Figure 4CT induced apoptosis by regulating
the Akt, mitogen-activated protein kinase and STAT3 signaling
pathways in HFLS-RA cells. Cells were treated with 5 µmol/l
CT for various time durations (0, 3, 6, 12 and 24 h). (A) Western
blot analysis results and (B) quantified protein expression levels
of Akt, p38, JNK, ERK and STAT3. **P<0.01 and
***P<0.001, vs. control (0 h) group. CT,
cryptotanshinone; Akt, protein kinase B; JNK, c-Jun N-terminal
kinase; ERK, extracellular signal-regulated kinase; STAT3, signal
transducer and activator of transcription-3; p-,
phosphorylated. |
CT induces apoptosis by regulating
intracellular ROS levels in HFLS-RA cells
To determine whether CT induced intracellular ROS
generation, the HFLS-RA cells were treated with 5 µmol/l CT
for different durations (0, 3, 6, 12 and 24 h), after which the
levels of ROS were detected by flow cytom-etry. As shown in
Fig. 5A and B, CT treatment
significantly increased ROS generation in a time-dependent
manner.
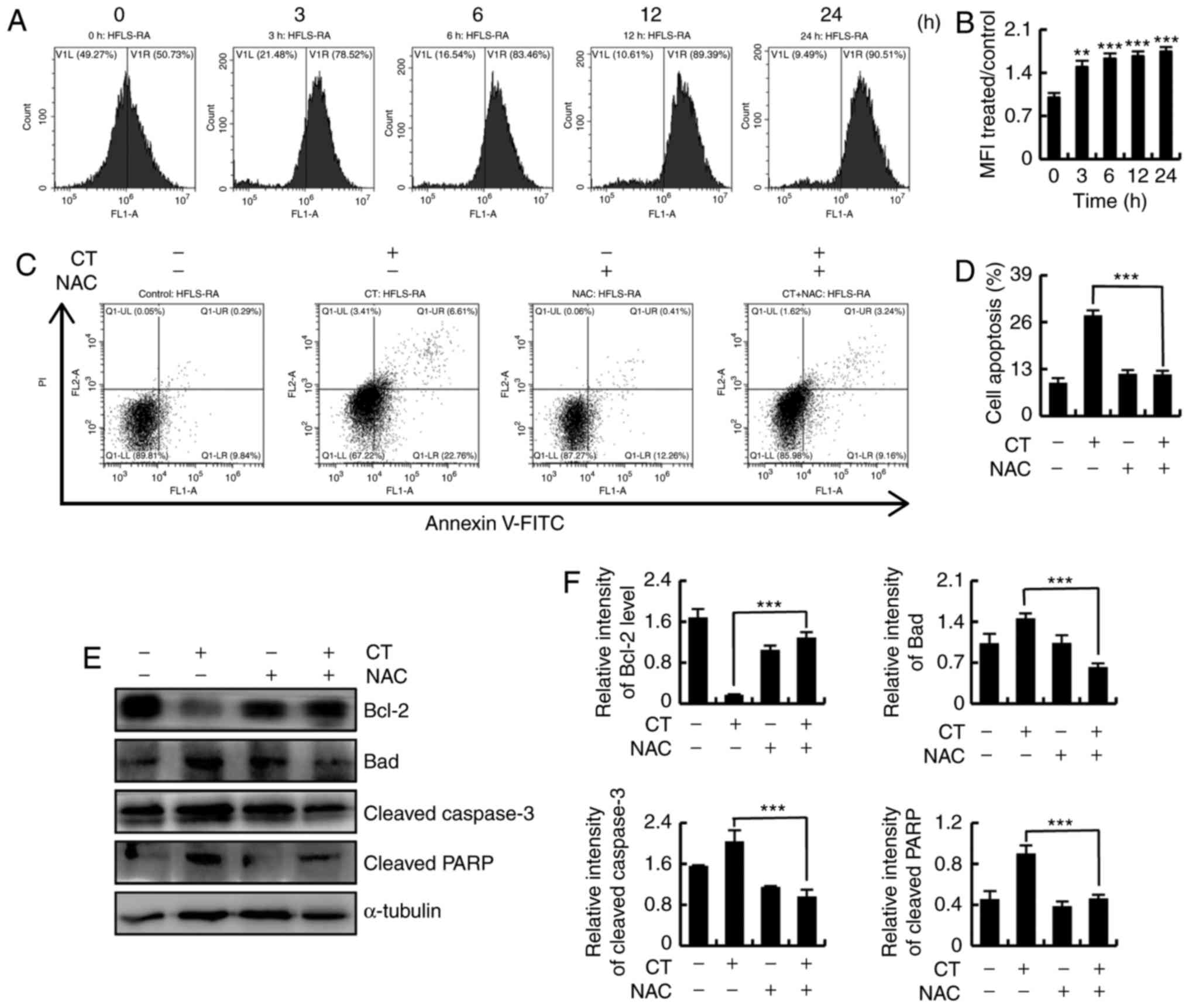 | Figure 5CT induced apoptosis via ROS-mediated
signaling pathways in HFLS-RA cells. (A) Flow cytometry and (B)
quantified levels of intracellular ROS in HFLS-RA cells incubated
with 5 µmol/l CT for the indicated time durations (0, 3, 6,
12 and 24 h) and then stained with DCFH-DA (10 µmol/l) for
30 min. (C) Flow cytometry and (D) quantification of apoptosis in
HFLS-RA cells pretreated with NAC (100 µmol/l) for 30 min
and then stimulated with 5 µmol/l CT for 24 h. (E) Western
blot analysis and (F) quantified protein expression levels of
Bcl-2, Bad, cleaved caspase-3 and cleaved PARP in HFLS-RA cells
treated with NAC (100 µmol/l) and/or 5 µmol/l CT.
**P<0.01 and ***P<0.001, vs. untreated
control group. CT, cryptotanshinone; ROS, reactive oxygen species;
NAC, N-acetyl cysteine; Bcl-2, B-cell lymphoma 2; Bad,
Bcl-2-associated death promoter; PARP, poly (ADP-ribose)
polymerase. |
To further investigate whether CT-induced apoptosis
was mediated by intracellular ROS, the cells were divided into four
groups, as follows: Control, CT, NAC and CT+NAC. As shown in
Fig. 5C and D, CT-induced
apoptosis was significantly suppressed after scavenging of
intracellular ROS by treatment with NAC. Changes in the expression
levels of apoptosis-associated proteins were detected by western
blotting, which demonstrated that the expression levels of Bcl-2,
Bad, cleaved caspase-3 and cleaved PARP were recovered following
intracellular ROS scavenging (Fig. 5E
and F). These results indicated that the increased levels of
ROS were involved in CT-induced apoptosis.
CT induces apoptosis through ROS-mediated
Akt, MAPK and STAT3 signaling pathways in HFLS-RA cells
To investigate whether CT induced apoptosis through
regulating ROS-mediated Akt, MAPK and STAT3 signaling pathways,
HFLS-RA cells were cultured overnight, treated with CT or NAC for
24 h, and divided into four groups as follows: Control, CT, NAC and
CT+NAC. Next, the protein expression levels were detected by
western blotting. As shown in Fig. 6A
and B, as compared with the CT treatment group, upregulation of
p-Akt, p-ERK and p-STAT3 levels, and downregulation of p-p38 and
p-JNK were detected in the CT+NAC treatment group. These data
indicated that CT induced apoptosis through ROS-mediated MAPK, Akt
and STAT3 signaling pathways.
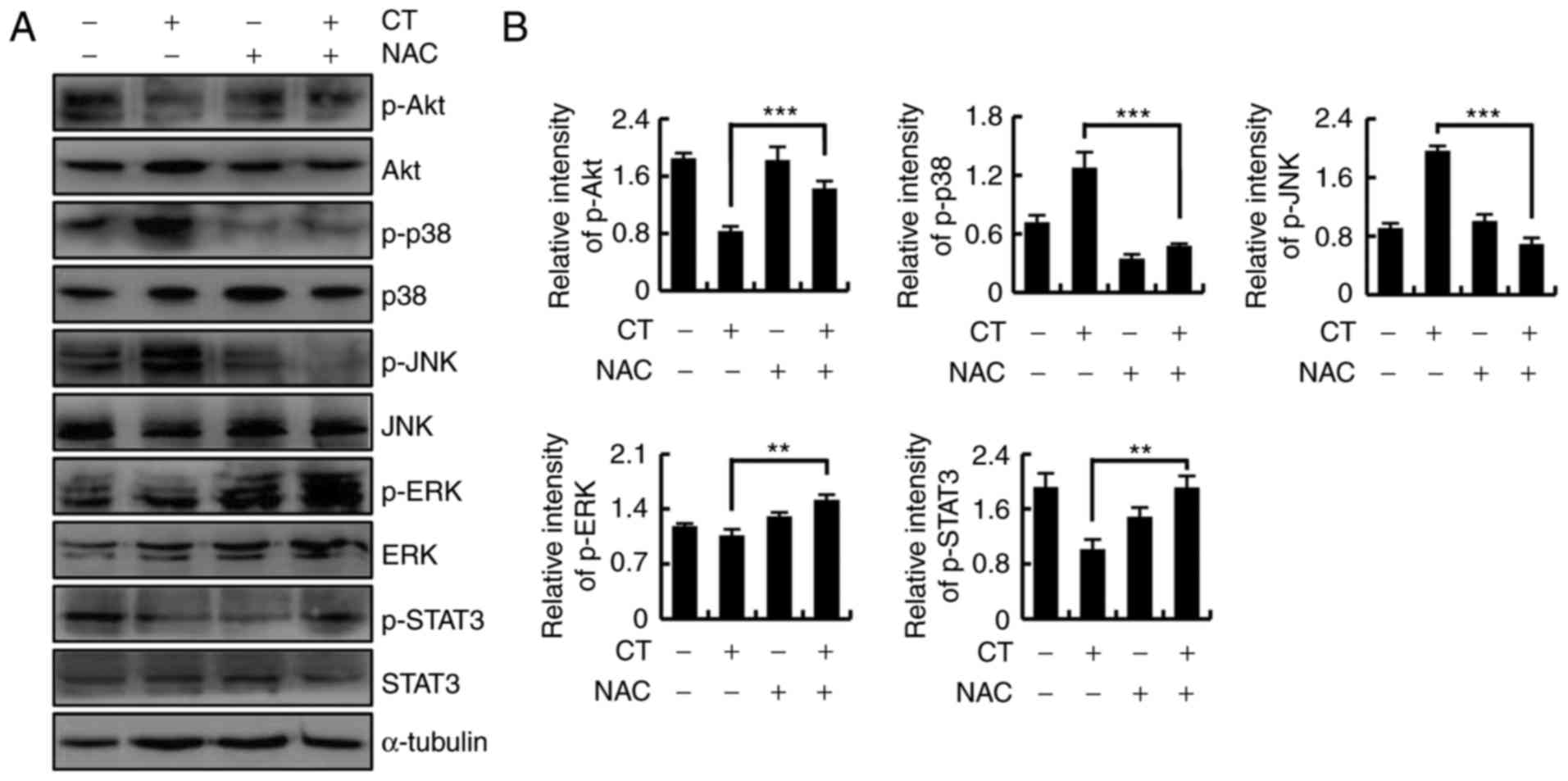 | Figure 6CT induced ROS-mediated apoptosis via
the Akt, mitogen-activated protein kinase and STAT3 signaling
pathways. Cells were pretreated with NAC (100 µmol/l) for 30
min and then stimulated with 5 µmol/l CT for 24 h. (A)
Western blots and (B) quantified protein expression levels of Akt,
p38, JNK, ERK and STAT3 are shown. **P<0.01 and
***P<0.001, vs. untreated control group. CT,
cryptotanshinone; ROS, reactive oxygen species; NAC, N-acetyl
cysteine; Akt, protein kinase B; JNK, c-Jun N-terminal kinase; ERK,
extracellular signal-regulated kinase; STAT3, signal transducer and
activator of transcription-3; p-, phosphorylated. |
Discussion
RA-FLSs are characterized by the massive imbalance
between excessive cell proliferation and apoptosis, and induced
cells apoptosis may be a possible treatment for patients with RA
(30). Apoptosis is regulated by
the mitochondrial pathway via the imbalance of pro-apoptotic (such
as Bad) and anti-apoptotic proteins (such as Bcl-2) (31). Caspase-3 is the key executive
mediator of apoptosis and a common downstream effector of a number
of apoptotic signaling pathways (32). The present study demonstrated that
the anti-proliferative and pro-apoptotic effects of CT in primary
RA-FLS, HFLS-RA and MH7A cells. In addition, the results revealed
that the CT-mediated apoptosis of RA-FLSs may be causally linked to
the upregulation of the pro-apoptotic protein Bad and
downregulation of the anti-apoptotic protein Bcl-2, as well as the
activation of caspase-3 and PARP. These findings indicated that
activation of the caspase cascade contributed to the apoptosis of
RA-FLSs.
The Akt signaling pathway generally restrains
chondrogenesis and apoptosis mediated by Akt, and has been
considered a potential target for RA treatment (33). In addition, the MAPK signaling
pathway controls the proliferation, differentiation, survival and
migration of various cell types, thereby serving a crucial role in
cell differentiation, apoptosis, stress response, and occurrence
and development of various human diseases (34,35). Thus, blocking the MAPK signaling
pathway as a tool for identifying a novel drug for RA treatment has
been the focus of certain research studies (36,37). Furthermore, the STAT3 signaling
pathway is a signal transduction pathway that was identified in
recent years to function in response to cytokine stimulation. It
participates in numerous important biological processes, such as
cell proliferation, differentiation, apoptosis and immune
regulation (38). STAT3 has also
been detected in the synovial lining of RA patients and animal
models of RA, and STAT3 blockade promoted apoptosis in RA-FLSs
(39). In the current study, the
molecular mechanisms underlying CT-induced apoptosis were
evaluated, and the results indicated that CT decreased the
expression levels of Akt, ERK and STAT3, and increased the
expression levels of JNK and p38. Thus, CT induced apoptosis via
the Akt, MAPK and STAT3 signaling pathways in HFLS-RA cells.
ROS is a group of highly reactive molecular oxygen
species produced by mitochondrial respiration, including oxygen
ions, hydrogen peroxide and oxygen-free radicals (17). ROS generation is considered to be
one of the primary cytotoxic mechanisms that induce cell apoptosis
or necrosis (19). In normal
conditions, high levels of ROS are involved in the pathogenesis of
RA and may be an important mechanism for cell death (40). The results of the present study
demonstrated that CT increased the levels of ROS, which led to an
increase in the number of apoptotic cells, while apoptosis was
suppressed by intracellular ROS scavenging. In addition, the Akt,
MAPK and STAT3 signaling pathways were rescued after the scavenging
of intracellular ROS. These results revealed that CT-induced
apoptosis was regulated by ROS-mediated Akt, MAPK and STAT3
signaling pathways in HFLS-RA cells. In future research, the
effects of CT in vivo should be evaluated.
In conclusion, the results of the present study
demonstrated the mechanisms underlying the function of CT in
HFLS-RA cells. Specifically, CT inhibited the proliferation and
induced the apoptosis of HFLS-RA cells through ROS-mediated Akt,
MAPK and STAT3 signaling pathways. These data provide evidence that
CT may be a potential therapeutic agent for the treatment of
RA.
Funding
The present study was funded by the Multigrain
Production And Processing Characteristic Discipline Construction
Project (grant no. 2042070010), the Natural Science Foundation of
Heilongjiang Province of China (grant no. LC2015036) and the
Postdoctoral Scientific Research Foundation of Heilongjiang
Province of China (grant no. LBH-Q13132).
Availability of data and materials
The analyzed datasets generated during the study are
available from the corresponding author on reasonable request.
Authors’ contributions
HNS, YHL, LQM, DJZ and CHJ conceived and designed
the experiments. HNS, YHL and LQM performed the experiments. XJP,
YW, JRW, HW, YiZ, JQL, WTX and YL performed the CCK-8 assay. YuZ,
TZ, YHH and MHJ performed sample preparations for western blotting.
GNS, YQZ and LKC performed sample preparations for flow cytometry.
DJZ and CHJ wrote the paper. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethical
Committee of the Fifth Affiliated Hospital of Harbin Medical
University (Daqing, China). Written informed consent was obtained
from each patient involved in the current study.
Patient consent for publication
Consent for publication was obtained from the
participants.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Xu H, He Y, Yang X, Liang L, Zhan Z, Ye Y,
Yang X, Lian F and Sun L: Anti-malarial agent artesunate inhibits
TNF-alpha- induced production of proinflammatory cytokines via
inhibition of NF-kappaB and PI3 kinase/Akt signal pathway in human
rheumatoid arthritis fibroblast-like synoviocytes. Rheumatology
(Oxford). 46:920–926. 2007. View Article : Google Scholar
|
|
2
|
Muthana M, Hawtree S, Wilshaw A, Linehan
E, Roberts H, Khetan S, Adeleke G, Wright F, Akil M, Fearon U, et
al: C5orf30 is a negative regulator of tissue damage in rheumatoid
arthritis. Proc Natl Acad Sci USA. 112:11618–11623. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Taylor JS: Thrombotic Thrombocytopenic
Purpura. Pediatric Surgery. Springer International Publishing;
Cham, Switzerland: pp. 303–304. 2014
|
|
4
|
Firth J: Rheumatoid arthritis: Diagnosis
and multidisciplinary management. Br J Nurs. 20:1179–1180. 2011.
View Article : Google Scholar
|
|
5
|
Ota F, Maeshima A, Yamashita S, Ikeuchi H,
Kaneko Y, Kuroiwa T, Hiromura K, Ueki K, Kojima I and Nojima Y:
Activin A induces cell proliferation of fibroblast-like
synoviocytes in rheumatoid arthritis. Arthritis Rheum.
48:2442–2449. 2003. View Article : Google Scholar
|
|
6
|
Kalkan A, Hallert E, Carlsson P, Roback K
and Sjöwall C: Individual variations in treatment decisions by
Swedish rheumatologists regarding biological drugs for rheumatoid
arthritis. Scand J Rheumatol. 44:265–270. 2015.
|
|
7
|
Puppo F, Murdaca G, Ghio M and Indiveri F:
Emerging biologic drugs for the treatment of rheumatoid arthritis.
Autoimmun Rev. 4:537–541. 2005. View Article : Google Scholar
|
|
8
|
Messori A, Fadda V, Maratea D, Trippoli S,
Gatto R, De Rosa M and Marinai C: Biological drugs for the
treatment of rheumatoid arthritis by the subcutaneous route:
Interpreting efficacy data to assess statistical equivalence. Ther
Adv Musculoskelet Dis. 6:207–214. 2014. View Article : Google Scholar
|
|
9
|
Tian J, Chen JW, Gao JS, Li L and Xie X:
Resveratrol inhibits TNF-α-induced IL-1β, MMP-3 production in human
rheumatoid arthritis fibroblast-like synoviocytes via modulation of
PI3kinase/Akt pathway. Rheumatol Int. 33:1829–1835. 2013.
View Article : Google Scholar
|
|
10
|
Westra J and Limburg PC: p38
mitogen-activated protein kinase (MAPK) in rheumatoid arthritis.
Mini Rev Med Chem. 6:867–874. 2006. View Article : Google Scholar
|
|
11
|
Hammaker DR, Boyle DL, Inoue T and
Firestein GS: Regulation of the JNK pathway by TGF-β activated
kinase 1 in rheumatoid arthritis synoviocytes. Arthritis Res Ther.
9:R572007. View
Article : Google Scholar
|
|
12
|
Valère A, Garnotel R, Villena I, Guenounou
M, Pinon JM and Aubert D: Activation of the cellular
mitogen-activated protein kinase pathways ERK, P38 and JNK during
Toxoplasma gondii invasion. Parasite. 10:59–64. 2003. View Article : Google Scholar
|
|
13
|
Reynolds G, Cooles FA, Isaacs JD and
Hilkens CM: Emerging immunotherapies for rheumatoid arthritis. Hum
Vaccin Immunother. 10:822–837. 2014. View
Article : Google Scholar
|
|
14
|
Migita K, Izumi Y, Torigoshi T, Satomura
K, Izumi M, Nishino Y, Jiuchi Y, Nakamura M, Kozuru H, Nonaka F, et
al: Inhibition of Janus kinase/signal transducer and activator of
transcription (JAK/STAT) signaling pathway in rheumatoid synovial
fibroblasts using small molecule compounds. Clin Exp Immunol.
174:356–363. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ahmad SF, Ansari MA, Zoheir KM, Bakheet
SA, Korashy HM, Nadeem A, Ashour AE and Attia SM: Regulation of
TNF-α and NF-κB activation through the JAK/STAT signaling pathway
downstream of histamine 4 receptor in a rat model of LPS-induced
joint inflammation. Immunobiology. 220:889–898. 2015. View Article : Google Scholar
|
|
16
|
Bugatti S, Manzo A, Caporali R and
Montecucco C: Inflammatory lesions in the bone marrow of rheumatoid
arthritis patients: A morphological perspective. Arthritis Res
Ther. 14:2292012. View
Article : Google Scholar
|
|
17
|
Rahman N, Jeon M, Song HY and Kim YS:
Cryptotanshinone, a compound of Salvia miltiorrhiza inhibits
pre-adipocytes differentiation by regulation of
adipogenesis-related genes expression via STAT3 signaling.
Phytomedicine. 23:58–67. 2016. View Article : Google Scholar
|
|
18
|
Rahman T, Hosen I, Islam MM and Shekhar
HU: Oxidative stress and human health. Adv Biosci Biotechnol.
3:997–1019. 2012. View Article : Google Scholar
|
|
19
|
Domej W, Oettl K and Renner W: Oxidative
stress and free radicals in COPD-implications and relevance for
treatment. Int J Chron Obstruct Pulmon Dis. 9:1207–1224. 2014.
View Article : Google Scholar
|
|
20
|
Mateen S, Moin S, Khan AQ, Zafar A and
Fatima N: Increased reactive oxygen species formation and oxidative
stress in rheumatoid arthritis. PLoS One. 11:e01529252016.
View Article : Google Scholar
|
|
21
|
Griendling KK, Touyz RM, Zweier JL,
Dikalov S, Chilian W, Chen YR, Harrison DG and Bhatnagar A;
American Heart Association Council on Basic Cardiovascular
Sciences: Measurement of reactive oxygen species, reactive nitrogen
species, and redox-dependent signaling in the cardiovascular
system: A scientific statement from the american heart association.
Circ Res. 119:e39–e75. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen L, Wang HJ, Xie W, Yao Y, Zhang YS
and Wang H: Cryptotanshinone inhibits lung tumorigenesis and
induces apoptosis in cancer cells in vitro and in vivo. Mol Med
Rep. 9:2447–2452. 2014. View Article : Google Scholar
|
|
23
|
Wu CF, Klauck SM and Efferth T: Anticancer
activity of cryptotanshinone on acute lymphoblastic leukemia cells.
Arch Toxicol. 90:2275–2286. 2016. View Article : Google Scholar
|
|
24
|
Chen W, Pan Y, Wang S, Liu Y, Chen G, Zhou
L, Ni W, Wang A and Lu Y: Cryptotanshinone activates AMPK-TSC2 axis
leading to inhibition of mTORC1 signaling in cancer cells. BMC
Cancer. 17:342017. View Article : Google Scholar :
|
|
25
|
Chen W, Lu Y, Chen G and Huang S:
Molecular evidence of cryptotanshinone for treatment and prevention
of human cancer. Anticancer Agents Med Chem. 13:979–987. 2013.
View Article : Google Scholar :
|
|
26
|
Li XH, Yang XM and Wu XK: Effects of
cryptotanshinone in lowering androgens synthesis for the prenatally
androgenized male rats. Zhongguo Zhong Xi Yi Jie He Za Zhi.
28:1001–1004. 2008.In Chinese.
|
|
27
|
Cha JD, Lee JH, Choi KM, Choi SM and Park
JH: Synergistic effect between cryptotanshinone and antibiotics
against clinic methicillin and vancomycin-resistant staphylococcus
aureus. Evid Based Complement Alternat Med. 2014:4505722014.
View Article : Google Scholar
|
|
28
|
Li W, Saud SM, Young MR, Colburn NH and
Hua B: Cryptotanshinone, a Stat3 inhibitor, suppresses colorectal
cancer proliferation and growth in vitro. Mol Cell Biochem.
406:63–73. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lin L, Wang D, Li L, Ding X and Ma H:
Dehydroepiandrosterone inhibits cell proliferation and improves
viability by regulating S phase and mitochondrial permeability in
primary rat leydig cells. Mol Med Rep. 14:705–714. 2016. View Article : Google Scholar
|
|
30
|
Ye T, Zhu S, Zhu Y, Feng Q, He B, Xiong Y,
Zhao L, Zhang Y, Yu L and Yang L: Cryptotanshinone induces melanoma
cancer cells apoptosis via, ROS-mitochondrial apoptotic pathway and
impairs cell migration and invasion. Biomed Pharmacother.
82:319–326. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Du J, Chen C, Sun Y, Zheng L and Wang W:
Ponicidin suppresses HT29 cell growth via the induction of G1 cell
cycle arrest and apoptosis. Mol Med Rep. 12:5816–5820. 2015.
View Article : Google Scholar
|
|
32
|
Zhou L, Luan H, Dong X and Li Y:
Activation of the PI3K/Akt and MAPK signaling pathways antagonizes
adriamycin-induced HL-60 leukemia cell apoptosis. Mol Med Rep.
3:641–644. 2010. View Article : Google Scholar
|
|
33
|
Li S, Chen JW, Xie X, Tian J, Deng C, Wang
J, Gan HN and Li F: Autophagy inhibitor regulates apoptosis and
proliferation of synovial fibroblasts through the inhibition of
PI3K/AKT pathway in collagen-induced arthritis rat model. Am J
Transl Res. 9:2065–2076. 2017.
|
|
34
|
Zhai H, Hu S, Liu T, Wang F, Wang X, Wu G,
Zhang Y, Sui M, Liu H and Jiang L: Nitidine chloride inhibits
proliferation and induces apoptosis in colorectal cancer cells by
suppressing the ERK signaling pathway. Mol Med Rep. 13:2536–2542.
2016. View Article : Google Scholar
|
|
35
|
Hao W, Zhang X, Zhao W, Zhu H, Liu ZY, Lu
J and Chen X: Cryptotanshinone induces pro-death autophagy through
JNK signaling mediated by reactive oxygen species generation in
lung cancer cells. Anticancer Agents Med Chem. 16:593–600. 2016.
View Article : Google Scholar
|
|
36
|
Park KR, Yun HM, Quang TH, Oh H, Lee DS,
Auh QS and Kim EC: 4-Methoxydalbergione suppresses growth and
induces apoptosis in human osteosarcoma cells in vitro and in vivo
xenograft model through down-regulation of the JAK2/STAT3 pathway.
Oncotarget. 7:6960–6971. 2016.PubMed/NCBI
|
|
37
|
Cheng G, Gao F, Sun X, Bi H and Zhu Y:
Paris saponin VII suppresses osteosarcoma cell migration and
invasion by inhibiting MMP-2/9 production via the p38 MAPK
signaling pathway. Med Rep. 14:3199–3205. 2016. View Article : Google Scholar
|
|
38
|
Miao D and Zhang L: Leptin modulates the
expression of catabolic genes in rat nucleus pulposus cells through
the mitogen-activated protein kinase and Janus kinase 2/signal
transducer and activator of transcription 3 pathways. Mol Med Rep.
12:1761–1768. 2015. View Article : Google Scholar
|
|
39
|
Yang Y, Dong Q and Li R: Matrine induces
the apoptosis of fibroblast-like synoviocytes derived from rats
with collagen- induced arthritis by suppressing the activation of
the JAK/STAT signaling pathway. Int J Mol Med. 39:307–316. 2017.
View Article : Google Scholar
|
|
40
|
Zheng R, You Z, Jia J, Lin S, Han S, Liu
A, Long H and Wang S: Curcumin enhances the antitumor effect of
ABT-737 via activation of the ROS-ASK1-JNK pathway in
hepatocellular carcinoma cells. Mol Med Rep. 13:1570–1576. 2016.
View Article : Google Scholar
|




















