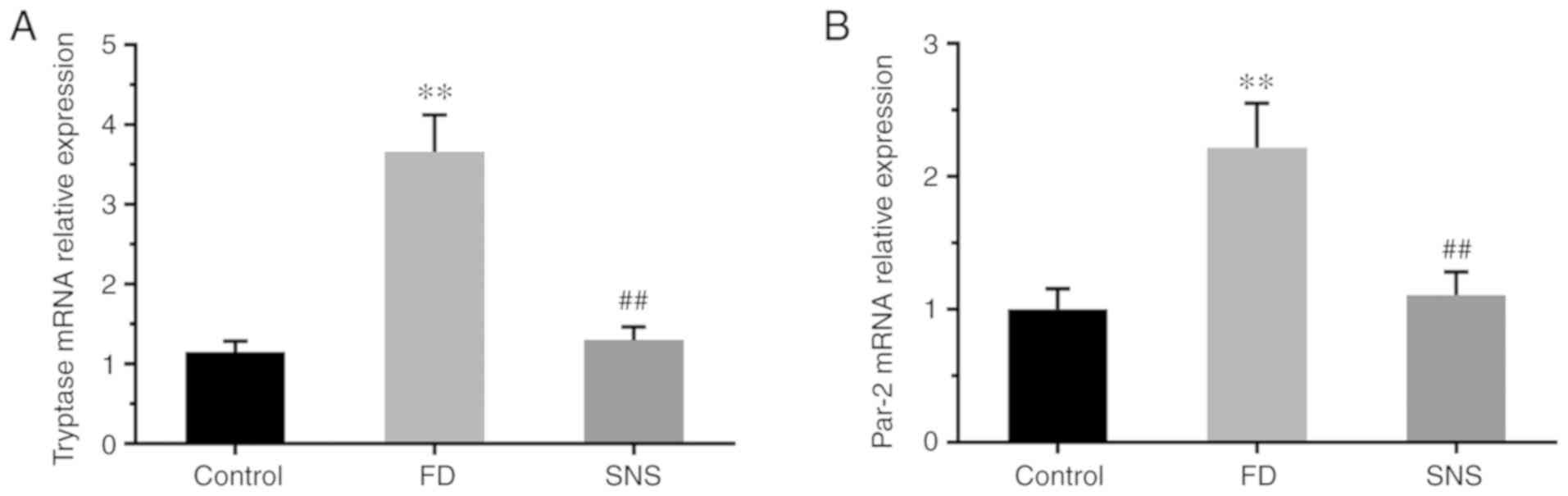
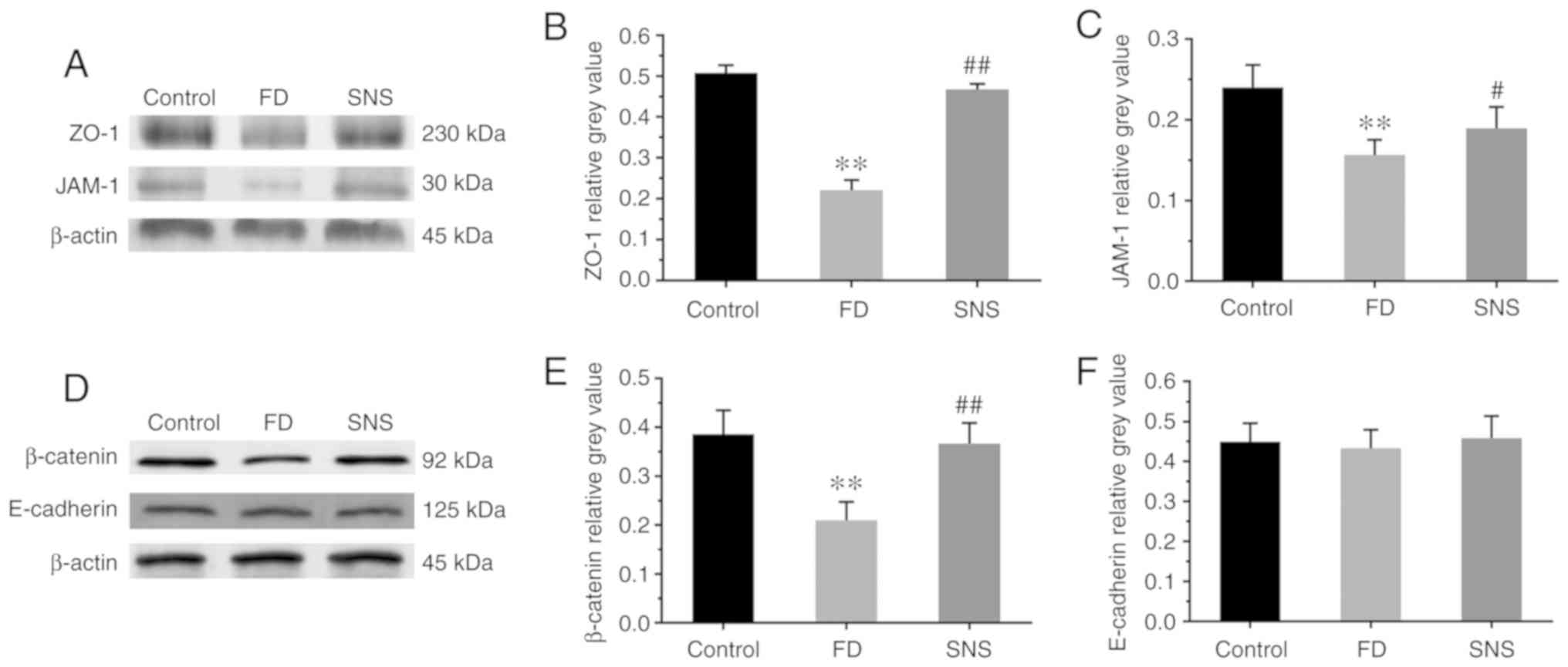
|
1
|
Tack J, Talley NJ, Camilleri M, Holtmann
G, Hu P, Malagelada JR and Stanghellini V: Functional
gastroduodenal disorders. Gastroenterology. 130:1466–1479. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stanghellini V, Chan FK, Hasler WL,
Malagelada JR, Suzuki H, Tack J and Talley NJ: Gastroduodenal
disorders. Gastroenterology. 150:1380–1392. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Konturek SJ, Konturek PC, Pawlik T,
Sliwowski Z, Ochmański W and Hahn EG: Duodenal mucosal protection
by bicarbonate secretion and its mechanism. J Physiol Pharmacol.
55(Suppl 2): S5–S17. 2014.
|
|
4
|
Vanheel H, Vicario M, Vanuytsel T, Van
Oudenhove L, Martinez C, Keita ÅV, Pardon N, Santos J, Söderholm
JD, Tack J and Farré R: Impaired duodenal mucosal integrity and
low-grade inflammation in functional dyspepsia. Gut. 63:262–271.
2014. View Article : Google Scholar
|
|
5
|
Ishigami H, Matsumura T, Kasamatsu S,
Hamanaka S, Taida T, Okimoto K, Saito K, Minemura S, Maruoka D,
Nakagawa T, et al: Endoscopy-guided evaluation of duodenal mucosal
permeability in functional dyspepsia. Clin Transl Gastroenterol.
8:e832017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Koloski NA, Jones M, Kalantar J, Weltman
M, Zaguirre J and Talley NJ: The brain-gut pathway in functional
gastrointestinal disorders is bidirectional: A 12-year prospective
population-based study. Gut. 61:1284–1290. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stengel A and Taché YF: Activation of
brain somatostatin signaling suppresses CRF receptor-mediated
stress response. Front Neurosci. 11:2312017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hagiwara SI, Kaushal E, Paruthiyil S,
Pasricha PJ, Hasdemir B and Bhargava A: Gastric
corticotropin-releasing factor influences mast cell infiltration in
a rat model of functional dyspepsia. PLoS One. 13:e2037042018.
View Article : Google Scholar
|
|
9
|
Wouters MM, Vicario M and Santos J: The
role of mast cells in functional GI disorders. Gut. 65:155–168.
2016. View Article : Google Scholar
|
|
10
|
Jacob C, Yang PC, Darmoul D, Amadesi S,
Saito T, Cottrell GS, Coelho AM, Singh P, Grady EF, Perdue M and
Bunnett NW: Mast cell tryptase controls paracellular permeability
of the intestine. Role of protease-activated receptor 2 and
beta-arrestins. J Biol Chem. 280:31936–31948. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Feng He and Qin Ling: Clinical efficacy of
Sini powder and western medicine in the treatment of mixed cold and
heat functional dyspepsia. Acta Chin Med Pharmacol. 42:148–150.
2014.
|
|
12
|
Han YY and Wang HB: Clinical observation
of modified sini powder in the treatment of 30 cases of functional
dyspepsia with depression. Beijing J Trad Chin Med. 30:457–458.
2011.
|
|
13
|
Li Y, Sun Y, Ma X, Xue X, Zhang W, Wu Z,
Ouyang Y, Chen J and Wang W, Guo S and Wang W: Effects of Sini san
used alone and in combination with fluoxetine on central and
peripheral 5-HT levels in a rat model of depression. J Tradit Chin
Med. 33:674–681. 2013. View Article : Google Scholar
|
|
14
|
Chang X, Zhao L, Wang J, Lu X and Zhang S:
Sinisan improves duodenal tight junction integrity in a rat model
of functional dyspepsia. BMC Complement Altern Med. 17:4322017.
View Article : Google Scholar
|
|
15
|
Chinese Pharmacopoeia Commission:
Pharmacopoeia of the People's Republic of China. 1. China Medical
Science Press; pp. 86–280. 2015
|
|
16
|
Wu ZY, Zhang SS, Li PC, Lu XF, Wang ZF,
Wang JJ and Li XL: Rat model of functional dyspepsia resulting from
iodoacet-amide-treated and tail-squeezed. Chin J Integrated
Traditional Western Med Digestion. 23:462–466. 2015.
|
|
17
|
Taché Y and Perdue MH: Role of peripheral
CRF signalling pathways in stress-related alterations of gut
motility and mucosal function. Neurogastroenterol Motil. 16(Suppl
1): S137–S142. 2004. View Article : Google Scholar
|
|
18
|
Taché Y, Martinez V, Million M and Rivier
J: Corticotropin-releasing factor and the brain-gut motor response
to stress. Can J Gastroenterol. 13(Suppl A): A18–A25. 1999.
View Article : Google Scholar
|
|
19
|
Walker MM and Talley NJ: The role of
duodenal inflammation in functional dyspepsia. J Clin
Gastroenterol. 51:12–18. 2017. View Article : Google Scholar
|
|
20
|
Neilan NA, Garg UC, Schurman JV and
Friesen CA: Intestinal permeability in children/adolescents with
functional dyspepsia. BMC Res Notes. 7:2752014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Larson D and Mitre E: Histamine release
and surface CD200R1 staining as sensitive methods for assessing
murine mast cell activation. J Immunol Methods. 379:15–22. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fernández-Blanco JA, Hollenberg MD,
Martinez V and Vergara P: PAR-2-mediated control of barrier
function and motility differs between early and late phases of
postinfectious gut dysfunction in the rat. Am J Physiol
Gastrointest Liver Physiol. 304:G390–G400. 2013. View Article : Google Scholar
|
|
23
|
Li S, Guan J, Ge M, Huang P, Lin Y and Gan
X: Intestinal mucosal injury induced by tryptase-activated
protease-activated receptor 2 requires β-arrestin-2 in vitro. Mol
Med Rep. 12:7181–7187. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Camilleri M, Madsen K, Spiller R,
Greenwood-Van Meerveld B and Verne GN: Intestinal barrier function
in health and gastrointestinal disease. Neurogastroenterol Motil.
24:503–512. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Stremmel W, Staffer S, Schneider MJ,
Gan-Schreier H, Wannhoff A, Stuhrmann N, Gauss A, Wolburg H,
Mahringer A, Swidsinski A and Efferth T: Genetic mouse models with
intestinal-specific tight junction deletion resemble an ulcerative
colitis phenotype. J Crohns Colitis. 11:1247–1257. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Landy J, Ronde E, English N, Clark SK,
Hart AL, Knight SC, Ciclitira PJ and Al-Hassi HO: Tight junctions
in inflammatory bowel diseases and inflammatory bowel disease
associated colorectal cancer. World J Gastroenterol. 22:3117–3126.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Martinez C, Vicario M, Ramos L, Lobo B,
Mosquera JL, Alonso C, Sánchez A, Guilarte M, Antolín M, de Torres
I, et al: The jejunum of diarrhea-predominant irritable bowel
syndrome shows molecular alterations in the tight junction
signaling pathway that are associated with mucosal pathobiology and
clinical manifestations. Am J Gastroenterol. 107:736–746. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Boeckxstaens G, Camilleri M, Sifrim D,
Houghton LA, Elsenbruch S, Lindberg G, Azpiroz F and Parkman HP:
Fundamentals of neurogastroenterology:
Physiology/motility-sensation. Gastroenterology. Feb. 18–2016.Epub
ahead of print. View Article : Google Scholar
|
|
29
|
Pastorelli L, De Salvo C, Mercado JR,
Vecchi M and Pizarro TT: Central role of the gut epithelial barrier
in the pathogenesis of chronic intestinal inflammation: Lessons
learned from animal models and human genetics. Front Immunol.
4:2802013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hamada K, Shitara Y, Sekine S and Horie T:
Zonula occludens-1 alterations and enhanced intestinal permeability
in methotrexate-treated rats. Cancer Chemother Pharmacol.
66:1031–1038. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Naik UP and Eckfeld K: Junctional adhesion
molecule 1 (JAM-1). J Biol Regul Homeost Agents. 17:341–347.
2003.
|
|
32
|
Lee HS, Kim DK, Kim YB and Lee KJ: Effect
of acute stress on immune cell counts and the expression of tight
junction proteins in the duodenal mucosa of rats. Gut Liver.
7:190–196. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tian X, Liu Z, Niu B, Zhang J, Tan TK, Lee
SR, Zhao Y, Harris DC and Zheng G: E-cadherin/β-catenin complex and
the epithelial barrier. J Biomed Biotechnol. 2011:5673052011.
View Article : Google Scholar
|
|
34
|
Yuan HP, Li Z, Zhang Y, Li XP, Li FK and
Li YQ: Anxiety and depression are associated with increased counts
and degranulation of duodenal mast cells in functional dyspepsia.
Int J Clin Exp Med. 8:8010–8014. 2015.PubMed/NCBI
|
|
35
|
Vanheel H, Vicario M, Boesmans W,
Vanuytsel T, Salvo-Romero E, Tack J and Farré R: Activation of
eosinophils and mast cells in functional dyspepsia: An
ultrastructural evaluation. Sci Rep. 8:53832018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Powell N, Walker MM and Talley NJ: The
mucosal immune system: Master regulator of bidirectional gut-brain
communications. Nat Rev Gastroenterol Hepatol. 14:143–159. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
De la Roca-Chiapas JM, Solís-Ortiz S,
Fajardo-Araujo M, Sosa M, Córdova-Fraga T and Rosa-Zarate A: Stress
profile, coping style, anxiety, depression, and gastric emptying as
predictors of functional dyspepsia: A case-control study. J
Psychosom Res. 68:73–81. 2010. View Article : Google Scholar
|
|
38
|
Overman EL, Rivier JE and Moeser AJ: CRF
induces intestinal epithelial barrier injury via the release of
mast cell proteases and TNF-α. PLoS One. 7:e399352012. View Article : Google Scholar
|
|
39
|
Wallon C, Yang PC, Keita AV, Ericson AC,
McKay DM, Sherman PM, Perdue MH and Söderholm JD:
Corticotropin-releasing hormone (CRH) regulates macromolecular
permeability via mast cells in normal human colonic biopsies in
vitro. Gut. 57:50–58. 2008. View Article : Google Scholar
|
|
40
|
Nozu T and Okumura T:
Corticotropin-releasing factor receptor type 1 and type 2
interaction in irritable bowel syndrome. J Gastroenterol.
50:819–830. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ayyadurai S, Gibson AJ, D'Costa S, Overman
EL, Sommerville LJ, Poopal AC, Mackey E, Li Y and Moeser AJ:
Frontline science: Corticotropin-releasing factor receptor subtype
1 is a critical modulator of mast cell degranulation and
stress-induced pathophysiology. J Leukoc Biol. 102:1299–1312. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
D'Costa S, Ayyadurai S, Gibson AJ, Mackey
E, Rajput M, Sommerville LJ, Wilson N, Li Y, Kubat E, Kumar A, et
al: Mast cell corticotropin-releasing factor subtype 2 suppresses
mast cell degranulation and limits the severity of anaphylaxis and
stress-induced intestinal permeability. J Allergy Clin Immunol.
143:1865–1877. 2019. View Article : Google Scholar
|