Introduction
Acute pulmonary embolism (APE) is a common cause of
acute cardiovascular failure and is associated with high morbidity
and mortality rates (1,2). The embolism causing obstruction of
the pulmonary vasculature usually leads to pulmonary hypertension
(PH) and cardiac failure. Studies have demonstrated that pulmonary
artery smooth muscle cells (PASMCs) excessively proliferate and
migrate to the pulmonary artery intima following APE, which results
in reconstruction of the pulmonary vasculature and thus increases
pulmonary vascular resistance (3,4).
MicroRNAs (miRNAs/miRs) are ~22 nucleotide-long
non-coding RNAs, which serve as post-transcriptional modulators of
the expression of target genes through binding to the 3′
untranslated regions (UTRs) of their respective target genes.
miRNAs affect mRNA expression levels through inhibiting translation
or inducing mRNA degradation (5).
Various miRNAs have been shown to regulate cell proliferation,
death and other physiological processes. Dysregulated expression of
various miRNAs is associated with several diseases, including
cardiovascular diseases, such as PH. Studies have shown that
abnormal expression of miRNAs contribute to the pathogenesis of
hypoxia-induced PH (6), such as
miR-31a-5p through targeting of TP53 (7), miR-135a through regulation of bone
morphogenetic protein receptor type-2 (BMPR2) levels (8) and miR-17 through targeting of
mitofusin-2 (9). Yue et al
(10) showed that miR-143/145
promotes hypoxia-induced proliferation and migration of PASMCs, and
improves hypoxia-induced PH through targeting ABCA1. Courboulin
et al (11) demonstrated
that miR-204 serves a significant role in decreasing proliferation,
vascular remodeling and regulating pulmonary artery blood pressure
in PH, through targeting of SHP2 (11). Several miRNAs have been identified
as biomarkers for chronic thromboembolic pulmonary hypertension
(CTEPH) and APE. miR-759, Let-7d, Let-7b and miR-22 have been
demonstrated to modulate fibrinolysis, which contributes to the
development of CTEPH (12).
Let-7d suppresses proliferation of PASMC and Let-7b targets TGBFR1
and endothelin-1 reducing migration of pulmonary artery endothelial
cells and PASMCs (13,14). Recent studies have shown that
expression of miR-23a, miR-221, miR-27a/b, miR-1233 and miR-28-3p
are significantly increased in the plasma of patients with APE
compared with healthy individuals, and may thus serve as potential
biomarkers for APE (1,15–18). Zhang et al (19) demonstrated that miR-23a controls
the proliferation and migration of human PASMCs by targeting
BMPR2/Smad1 signaling (19).
However, the specific mechanisms of several miRNAs remain to be
determined in APE. To further understand the pathophysiological
mechanisms underlying APE, additional studies examining the effects
of miRNAs on APE required.
Li et al (20) demonstrated that miR-106b-5p binds
to the 3′-UTR of Angiopoietin 2 (Angpt2) to induce migration and
tube formation of HUVECs, and human cholesteatoma perimatrix
fibroblasts (hCPFs)-exosomes transports miR-106b-5p to endothelial
cells and promotes angiogenesis by upregulating expression of
Angpt2 (20). miR-106b-5p is
pivotal in regulating cell proliferation and migration. Thus, it
was hypothesized that miR-106b-5p may be closely associated with
excessive proliferation and migration of PASMCs following APE. As a
member of the NR4A subfamily of nuclear receptors, NOR-1 activity
is sustained at a relatively low levels in healthy vascular
endothelial cells and is upregulated when affected by external
stimuli (21,22). NOR-1 is an effector of
inflammation, growth factors, lipoproteins and thrombin, that
controls the spreading, migration and proliferation of vascular
cells (23–26). In the present study, miR-106b-5p
was downregulated in PDGF-induced PASMCs and in an APE mouse model.
Furthermore, miR-106b-5p targeted the 3′ UTR of NOR-1 mRNA. The
functional roles of miR-106b-5p in PDGF-induced PASMCs and in an
APE mouse model were evaluated and the underlying molecular
mechanisms were determined.
Materials and methods
Mouse model of APE
Male C57BL/6 mice (weighing 20±2 g; n=48), were
purchased from the animal center of Xi’an Jiaotong University and
kept at 22±2°C with a relative humidity of 40–70%, allowed to
freely forage, with a 12-h light/dark cycle and ad libitum
access to food and water. All animal experiments were performed
according to the National Institutes of Health’s Guide for the Care
and Use of Laboratory Animals and approved by the Institutional
Animal Care and Use Committee of Xi’an Jiaotong University. The APE
model was established through self-blood coagulum, as previously
described (27). Briefly, 100
μl blood was drawn from the tail veins of the mice. After
coagulation, blood was clotted in a 37°C water bath for 30 min and
diced into 1×1 mm sections. A total of 30 autologous blood clots
were injected followed by 0.4 ml saline to establish the model. In
the sham group, mice were injected with 0.4 ml saline. Mice were,
respectively, treated with 10 mg/kg agomiR-106b-5p
(5′-UAAAGUGCUGACAGUGCAGAU-3′) or agomiR-NC
(5′-UUCUCCGAACGUGUCACGU-3′) for 3 consecutive days (Guangzhou
RiboBio Co., Ltd.) through tail vein intravenous injections,
beginning 15 min after establishment of the model. Following the
operation, mice were given ad libitum access to water and
food. The mortality of the mice in each group was monitored. After
7 days of treatment, mice were euthanized by carbon dioxide
asphyxiation (flow rate displacing no more than 30% of the chamber
volume/minute, mice were kept in carbon dioxide asphyxiation for
2–3 min, followed by respiratory and cardiac arrest for another 1
min in the box), and lung tissue was obtained to analyze the lung
index: Lung index = lung weight (mg)/body weight (g) ×100%; and to
perform subsequent experiments.
PDGF-induced PASMCs model
Mouse PASMCs were purchased from ScienCell Research
Laboratories, Inc. PASMCs were cultured in DMEM (Gibco; Thermo
Fisher Scientific, Inc.) supplemented with 10% FBS (Gibco; Thermo
Fisher Scientific, Inc.). Once confluence had reached 80%, PASMCs
were treated with 10, 20 or 40 ng/ml PDGF (Sigma-Aldrich; Merck
KGaA), and then treated with agomiR-106b-5p, antagomiR-106b-5p
(5′-AUCUGCACUGUCAGCACUUUA-3′), agomiR-NC or antagomiR-NC
(5′-UUCUCCGAACGUGUCACGU-3′), respectively, for 24 h.
Transfection
NOR1 lentiviral activation particles (cat. no.
sc-421926-LAC; Santa Cruz Biotechnology, Inc.) were used to
overexpress NOR1. PASMCs at 80% confluence were treated with the
lentiviral particles, incubated at 37°C for 6 h and subsequently
the media was replaced. PASMCs were further cultured for 48 h
before subsequent experiments were performed.
Cell proliferation assay
Proliferation of PASMCs was assessed using a Cell
Counting Kit-8 (CCK8; Dojindo Molecular Technologies, Inc.),
according to the manufacturer’s protocol. A total of
3×104 cells/well were plated in 96-well plates.
Following treatment, 10 μl CCK8 solution was added to the
culture medium and the cultures were incubated for 1–3 h at 37°C
with 5% CO2. Absorbance was measured at 450 nm with a
microplate reader (Invitrogen; Thermo Fisher Scientific, Inc.).
Transwell invasion assays
Transwell invasion assays were performed using
Transwell chambers (8 μm) coated with Matrigel (BD
Biosciences; Becton, Dickinson and Company). In the bottom chamber,
750 μl DMEM supplemented with 10% FBS was added and 250
μl cell suspension at a density of 1×105/ml was
added to the upper chamber without FBS. After culturing for 24 h,
the cells which had not invaded were removed and the remaining
cells were fixed using 75% alcohol for 10 min at room temperature,
stained with 0.5% crystal violet for 5 min at room temperature, and
imaged in four randomly selected fields under an inverted optical
light microscope (magnification, ×10). The number of invaded cells
in each group was counted.
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was obtained from the PASMCs of lung
tissue samples and cells using TRIzol® reagent
(Invitrogen; Thermo Fisher Scientific, Inc.). RT was performed in a
20 μl mixture containing 1 μg total RNA using a
miRcute Plus miRNA First-Strand cDNA kit (Tiangen Biotech Co. Ltd.)
or QuantScript RT kit (Tiangen Biotech Co. Ltd.) according to the
manufacturer’s protocols (37.0°C for 15 min, 95.0°C for 5 sec and
4.0°C for 60 min). qPCR experiments were performed using a miRcute
Plus miRNA qPCR kit (Tiangen Biotech Co. Ltd.) or a SuperReal
PreMix Plus (SYBR Green) (Tiangen Biotech Co. Ltd.) according to
the manufacturer’s protocols (95°C for 2 min, followed by 35 cycles
of 95°C for 15 sec, 55°C for 30 sec and 72°C for 10 sec; final
elongation at 72°C for 10 min). U6 and GAPDH were used as internal
controls for miRNA and mRNA, respectively. The sequences of the
primers used were: NOR1 forward, 5′-TGCTTTACGTCCTAGACCAG-3′ and
reverse, 5′-GTCTCGTCCACCTGATGTAT-3′; GAPDH forward,
5′-AACTTTGGCATTGTGGAAGG-3′ and reverse, 5′-GGATGCAGGGATGATGTTCT-3′;
miR-106b-5p, 5′-UAAAGUGCUGACAGUGCAGAU-3′; and U6.
5′-CAAATTCGTGAAGCGTTCCATAT-3′. Relative gene expression data were
analyzed using reverse transcription-quantitative PCR and the
2−ΔΔCq method (28).
The level of mRNA was determined by calculating the intensity ratio
of NOR1 mRNA/GAPDH mRNA or miR-106b-5p/U6.
Western blotting
Total proteins were obtained from the PASMCs of lung
tissue samples and cells using RIPA lysis buffer (Beyotime
Institute of Biotechnology). Protein concentrations were quantified
using a bicinchoninic acid protein kit (Invitrogen; Thermo Fisher
Scientific, Inc.). Protein (40 μg/lane) was loaded onto 10%
SDS-PAGE gels. Following SDS-PAGE and transfer to a PVDF membrane
(EMD Millipore), membranes were blocked in 5% BSA (Sigma-Aldrich;
Merck KGaA). Membranes were incubated with the primary antibodies
at 4°C overnight. The primary antibodies used were anti-NOR1 (cat.
no. ab94507; 1:1,000; Abcam), anti-proliferating cell nuclear
antigen (PCNA; cat. no. ab92552; 1:1,000; Abcam) and anti-GAPDH
(cat. no. 5174S; 1:2,000; CST Biological Reagents Co., Ltd.).
Subsequently, membranes were incubated with the Horseradish
peroxidase, Goat Anti-Rabbit IgG secondary antibody (cat. no.
A21020; 1:20,000; Abbkine Scientific Co., Ltd.) for 2 h at room
temperature. Signals were visualized using Clarity Western ECL
Substrate (Bio-Rad Laboratories, Inc.) and detected using a MiVnt
image analysis system (Bio-Rad Laboratories, Inc.). Optical
densities of the bands were calculated using Image Lab v5.2.1
(Bio-Rad Laboratories, Inc.).
Luciferase assays
Bioinformatics analysis using TargetScan v7.2
(http://www.targetscan.org/vert_72/),
PicTar (http://www.pictar.org/), and miRanda
(http://www.microrna.org/microrna/home.do) predicted
that NOR-1 was a potential target gene of miR-106b-5p (29,30). Plasmids containing a portion of
wide type (WT) or mutated (Mut) 3-UTR of the NOR1 gene were
synthesized by Guangzhou RiboBio Co., Ltd. A total of
4×104 PASMCs/well were plated and co-transfected with
200 ng luciferase vector (Promega Corporation) and 100 nM
agomiR-106b-5p or agomiR-NC using Lipofectamine® 3000
reagent (Invitrogen; Thermo Fisher Scientific, Inc.). After 48 h,
PASMCs were lysed and a Dual-Luciferase Reporter Assay (Promega
Corporation) was then performed as follows: Luciferase Assay
Reagent II was injected, the relative light unit (RLU) of Firefly
luciferase activity was measured, Stop and Glo® reagent
was injected, the RLU of Renilla luciferase activity was
measured and the level of RLU Firefly/RLU Renilla was
analyzed according to the manufacturer’s protocol.
RNA immunoprecipitation (RIP)
RIP of miRNA ribonucleoprotein complex with
anti-Argonaute 1 (Ago2; Abcam) or immunoglobulin G (IgG;
Sigma-Aldrich; Merck KGaA) was performed as previously reported
(17). When PASMCs reached 80%
confluence, they were transfected with 100 nM agomiR-106b-5p or
agomiR-NC for 24 h. Cell lysates were harvested using RIP buffer,
incubated with magnetic beads bound with anti-Ago2 or IgG
antibodies. After digestion with proteinase K, the precipitated
complexes were collected and the RNA was extracted using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.). Subsequently RT-qPCR was performed to confirm the
miR-106b-5p target was NOR1.
Statistical analysis
Data are presented as the mean ± standard error of
the mean of four experimental repeats. All statistical analyses
were performed using GraphPad Prism version 7.0 (GraphPad Prism
Software, Inc.). Differences between groups were compared using a
Student’s t-test or a one-way ANOVA followed by a post-hoc Tukey’s
test. P<0.05 was considered to indicate a statistically
significant difference.
Results
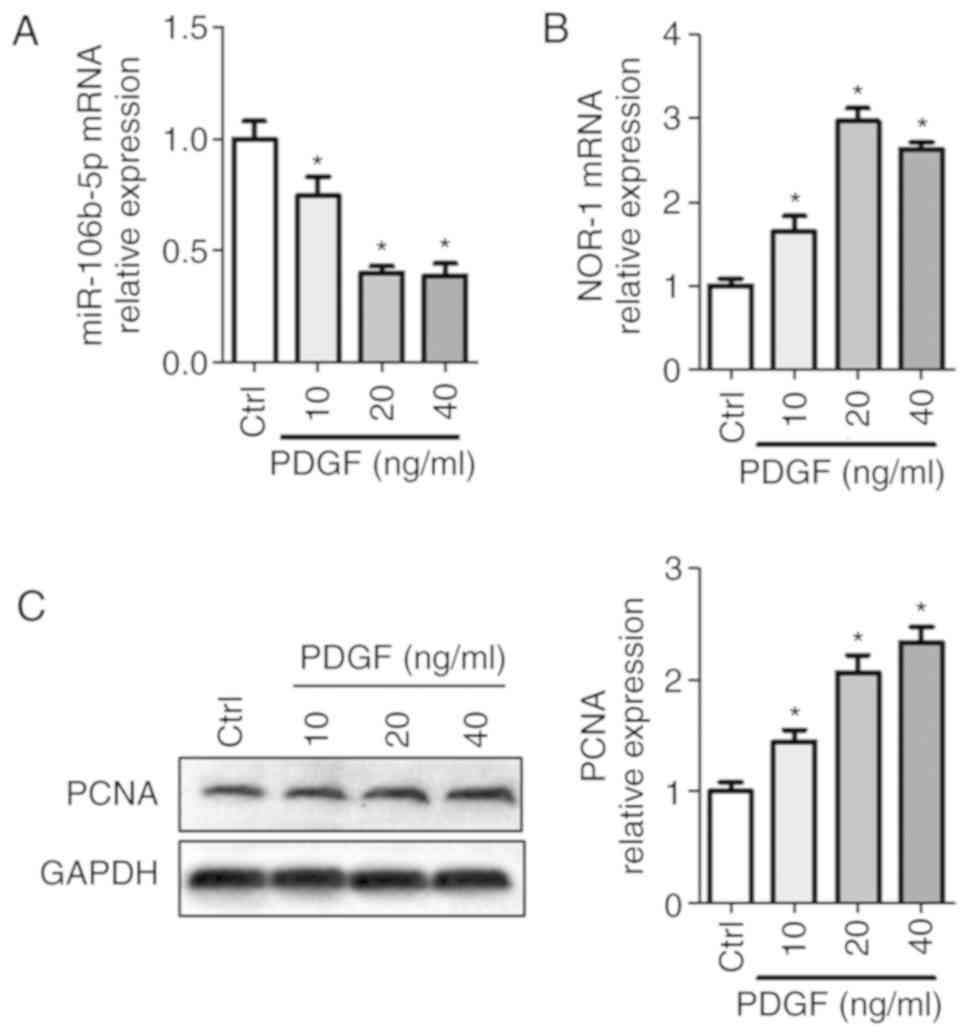
PDGF reduces miR-106b-5p expression and
increases activity of the NOR-1-PCNA signaling pathway in
PASMCs
To examine the roles of miR-106b-5p on PASMCs,
miR-106b-5p levels and NOR-1-PCNA pathway activity were detected
following treatment with 0, 10, 20 or 40 ng/ml PDGF in PASMCs. As
shown in Fig. 1, PDGF
significantly decreased miR-106b-5p levels, but increased NOR-1
mRNA and PCNA expression levels in a dose-dependent manner.
miR-106b-5p levels were decreased 0.75-fold, 0.4-fold and 0.39-fold
when treated with 10, 20 and 40 ng/ml PDGF, respectively, compared
with the control group (Fig. 1A).
NOR-1 mRNA expression levels were increased 1.65-fold, 2.97-fold
and 2.63-fold when treated with 10, 20 and 40 ng/ml PDGF treatment,
respectively, compared with the control group (Fig. 1B). PCNA expression levels were
increased 1.45-fold, 2.07-fold and 2.33-fold when treated with 10,
20 and 40 ng/ml PDGF treatment, respectively, compared with the
control group (Fig. 1C). Thus,
PDGF treatment reduced miR-106b-5p expression levels and increased
proliferation of PASMCs in a dose-dependent manner. For subsequent
experiments, 20 ng/ml PDGF was used.
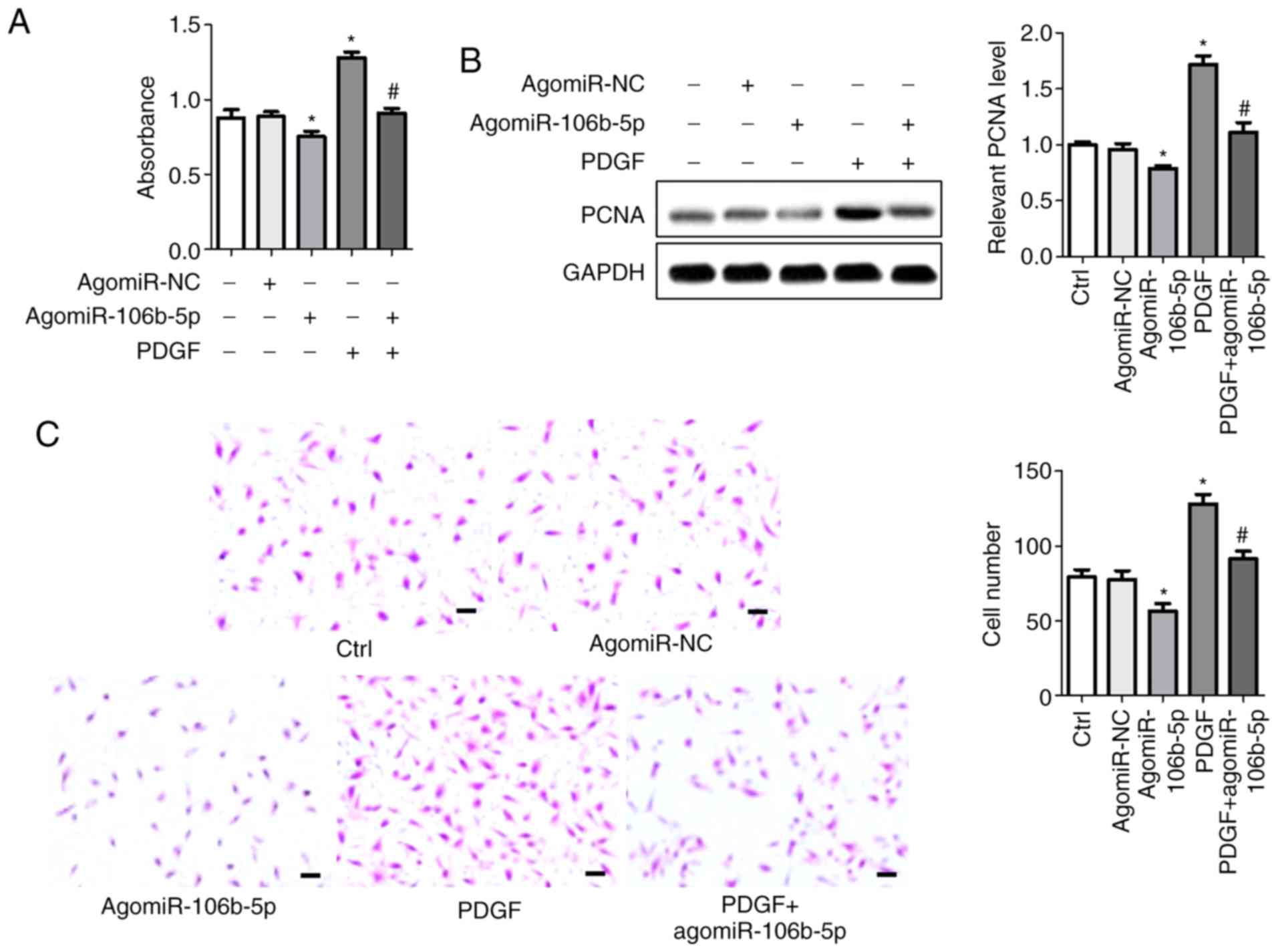
miR-106b-5p reverses PDGF-induced
proliferation and migration of PASMCs
To determine the effect of miR-106b-5p on the
proliferation and migration of PASMCs, agomiR-106b-5p and
antagomiR-106b-5p were used to induce or suppress miR-106b-5p
expression in PASMCs, respectively, and the levels of miR-106b-5p
was assessed using RT-qPCR (Fig.
S1A). CCK8 assays were used to assess cell viability.
Consistent with previous results, PDGF significantly increased the
viability of cells and agomiR-106b-5p inhibited the viability of
cells compared with the control group (P<0.05). AgomiR-106b-5p
reversed the PDGF-induced increase in viability in PASMCs compared
with the PDGF group (P<0.05; Fig.
2A). PCNA protein expression was measured using western
blotting (Fig. 2B). Protein bands
and densitometry analysis showed that agomiR-106b-5p significantly
abrogated the PDGF-induced increase in PCNA expression compared
with the PDGF group (P<0.05). Cell invasion was assessed using a
Transwell invasion assay (Fig.
2C). Following agomiR-106b-5p treatment, the number of invasive
cells was significantly reduced, whereas PDGF increased the
invasiveness of PASMCs. The number of invasive cells in the
control, agomiR-NC, agomiR-106b-5p, PDGF and PDGF + agomiR-106b-5p
groups were 79.33±4.84, 77.67±5.78, 56.67±4.91, 128.30±6.06 and
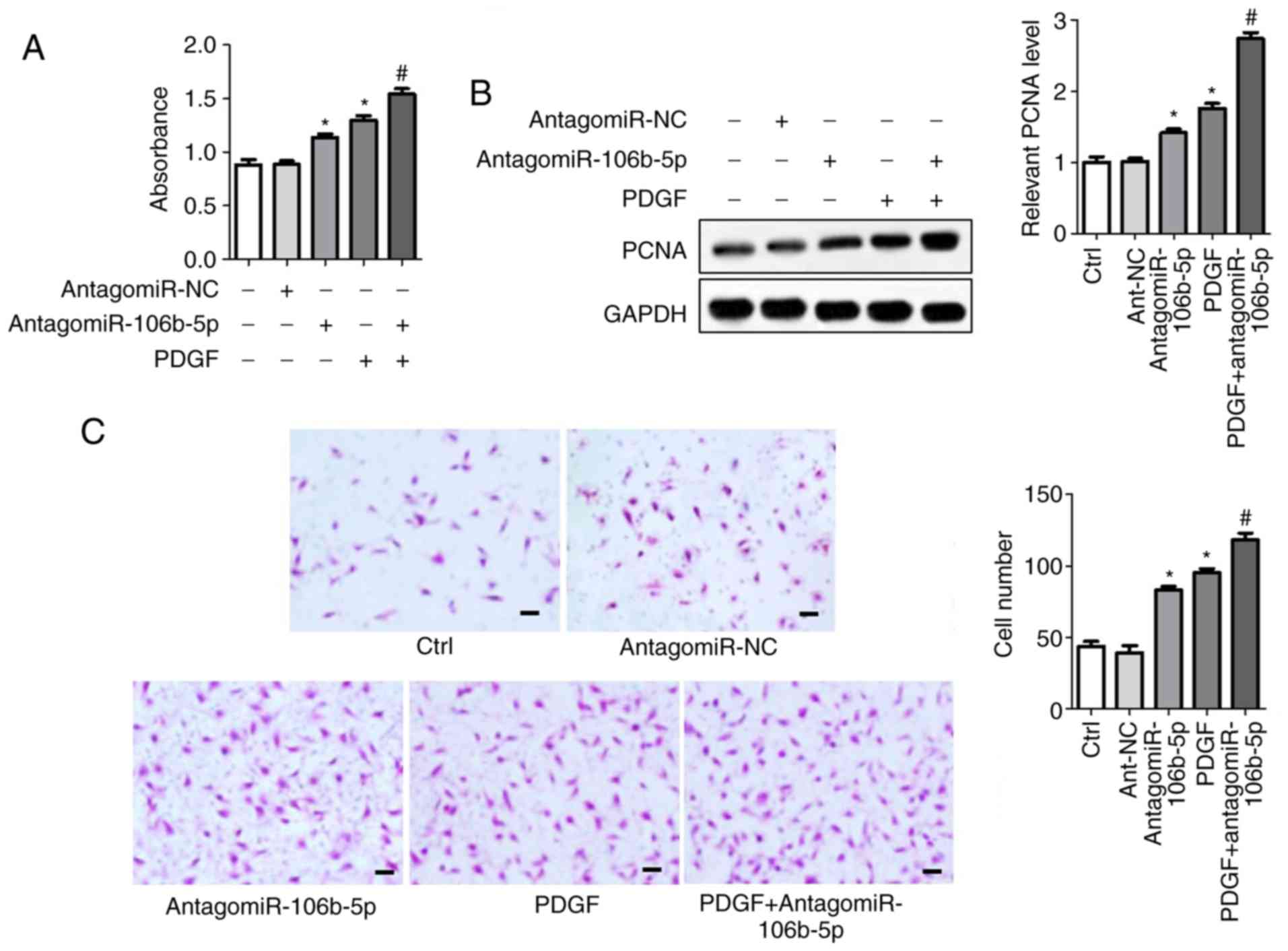
91.33±5.57, respectively. Conversely, antagomiR-106b-5p inhibited
miR-106b-5p expression in PASMCs, which facilitated cell
proliferation (cell viability and PCNA expression) and migration
compared with the control group (Fig.
3). AntagomiR-106b-5p significantly enhanced the effects of
PDGF on the combined treatment group compared with PDGF alone
(Fig. 3).
miR-106b-5p targets the 3′UTR of NOR-1
and reduces NOR-1 expression
Bioinformatics analysis was performed to predict the
potential target genes of miR-106b-5p and NOR-1 was shown to be a
potential target of miR-106b-5p (Fig.
4A). To validate this prediction, the specificity of
miR-106b-5p to NOR1 mRNA was assessed using two methods: Ago2-IP
and 3′UTR luciferase reporter assays. The RIP assay results
demonstrated that there was an ~5-fold enrichment of NOR-1 mRNA
obtained from the Ago2-IP of the agomiR-106b-5p group compared with
the NC group (P<0.05), suggesting that miR-106b-5p and NOR-1
were located in the same RNA-induced silencing complex (Fig. 4B). As shown in Fig. 4C, after transfection with
WT-NOR-1, there was an ~0.47-fold decrease in the relative
luciferase activity in the agomiR-106b-5p group compared with the
NC group (P<0.05), and an ~1.56-fold increase in luciferase
activity in the antagomiR-106b-5p group compared with the NC group
(P<0.05). There was no difference in the luciferase activity
between the groups following transfection with the Mut NOR-1 UTR
(Fig. 4C). These data suggest
that miR-106b-5p may target the 3′-UTR of NOR-1, which was also
confirmed by the results in Fig.
4D. The mRNA expression levels of NOR-1 were significantly
decreased following agomiR-106b-5p treatment and increased
following treatment with antagomiR-106b-5p and/or PDGF, and
agomiR-106b-5p also suppressed PDGF-induced NOR-1 expression levels
(P<0.05; Fig. 4D). Thus, these
data demonstrate that miR-106b-5p negatively regulates NOR-1
expression via binding to the 3′ UTR.
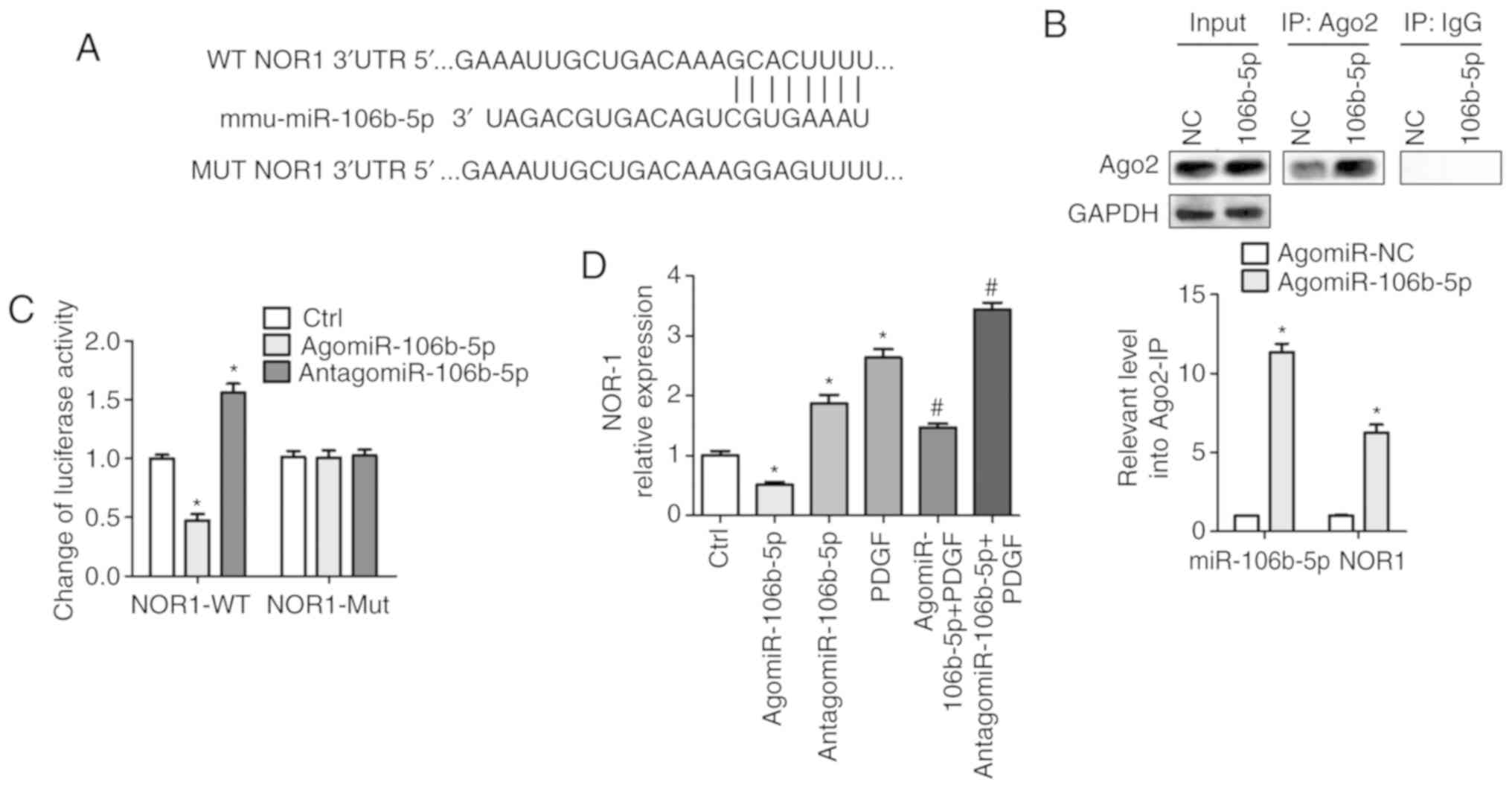 | Figure 4miR-106b-5p targeting of NOR-1 is
confirmed using Ago2 immunoprecipitation and luciferase reporter
assays. (A) Bioinformatics analysis using TargetScan, PicTar and
miRanda predicted that NOR-1 was a potential target gene of
miR-106b-5p. (B) PASMCs were transfected with 100 nM agomiR-106b-5p
or agomiR-NC for 24 h, the cells were collected and
immunoprecipitated using Ago2 or IgG and the fold-changes in NOR-1
transcript levels were calculated. *P<0.05 vs.
agomiR-NC group. (C) After transfection with WT-NOR-1 or Mut-NOR-1
and treatment with agomiR-106b-5p or antagomiR-106b-5p in PASMCs,
luciferase assays were performed to demonstrate miR-106b-5p
directly targeted NOR-1. *P<0.05 vs. Ctrl group. (D)
After treatment with agomiR-106b-5p, antagomiR-106b-5p or PDGF in
PASMCs, the mRNA expression levels of NOR-1 were measured.
*P<0.05 vs. Ctrl group. #P<0.05 vs.
PDGF group. miR, microRNA; PDGF, platelet-derived growth factor;
PASMC, pulmonary artery smooth muscle cell; NC, negative control;
WT, wild-type; Mut, mutated; Ctrl, control. |
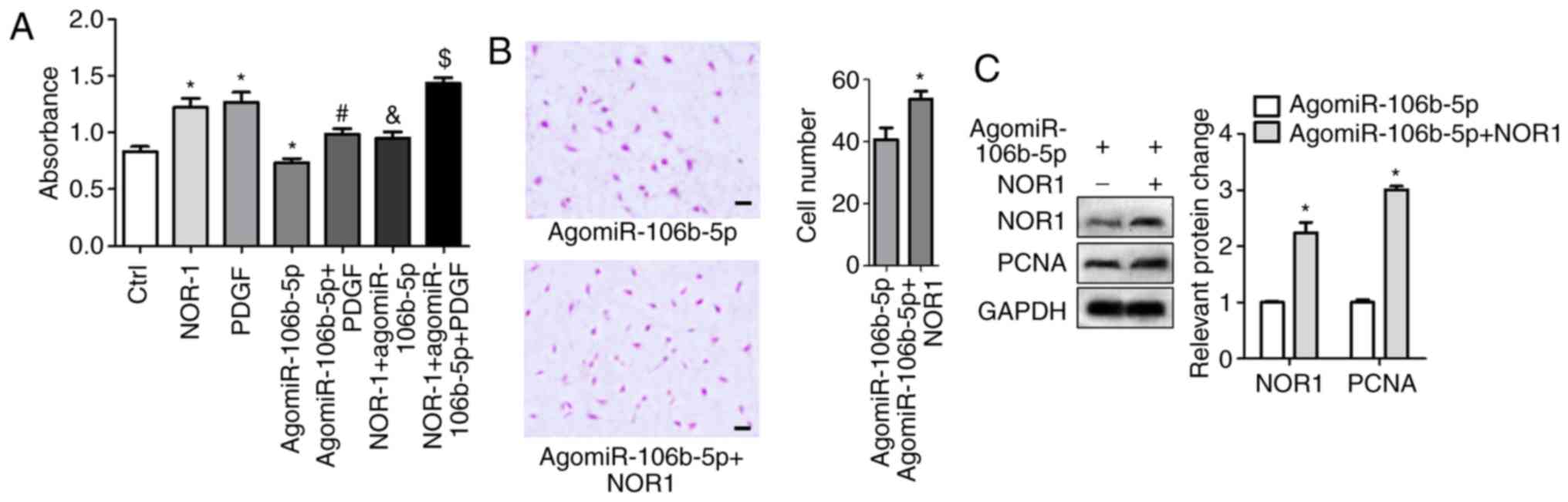
Activated NOR-1 reduces
miR-106b-5p-inhibition of PASMC proliferation and migration
After 48 h transfection with NOR1 lentiviral
activation particles or NC particles in PASMCs, transfection
efficiency for NOR1 overexpression are shown in Fig. S1B and C. NOR1 lentiviral
activation particles induced NOR1 mRNA and expression. Then cells
were treated with agomiR-106b-5p and/or PDGF, and cell viability
and migration were measured. NOR-1 and PDGF significantly increased
cell viability and agomiR-106b-5p decreased cell viability, but the
effect of agomiR-106b-5p was significantly reduced in NOR-1
activated PASMCs (Fig. 5A).
Activated NOR-1 increased cell invasion in the agomiR-106b-5p
treatment group, the invasive cell count was 40.7±3.84 and
53.7±2.60 in the agomiR-106b-5p group and the NOR-1 +
agomiR-106b-5p group, respectively (Fig. 5B). Western blotting results showed
that NOR-1 activation significantly increased PCNA expression in
the NOR-1 + agomiR-106b-5p group compared with the agomiR-106b-5p
group (P<0.05; Fig. 5C). Thus,
NOR-1 activation reversed the inhibition of miR-106b-5p on
proliferation and migration of PASMCs.
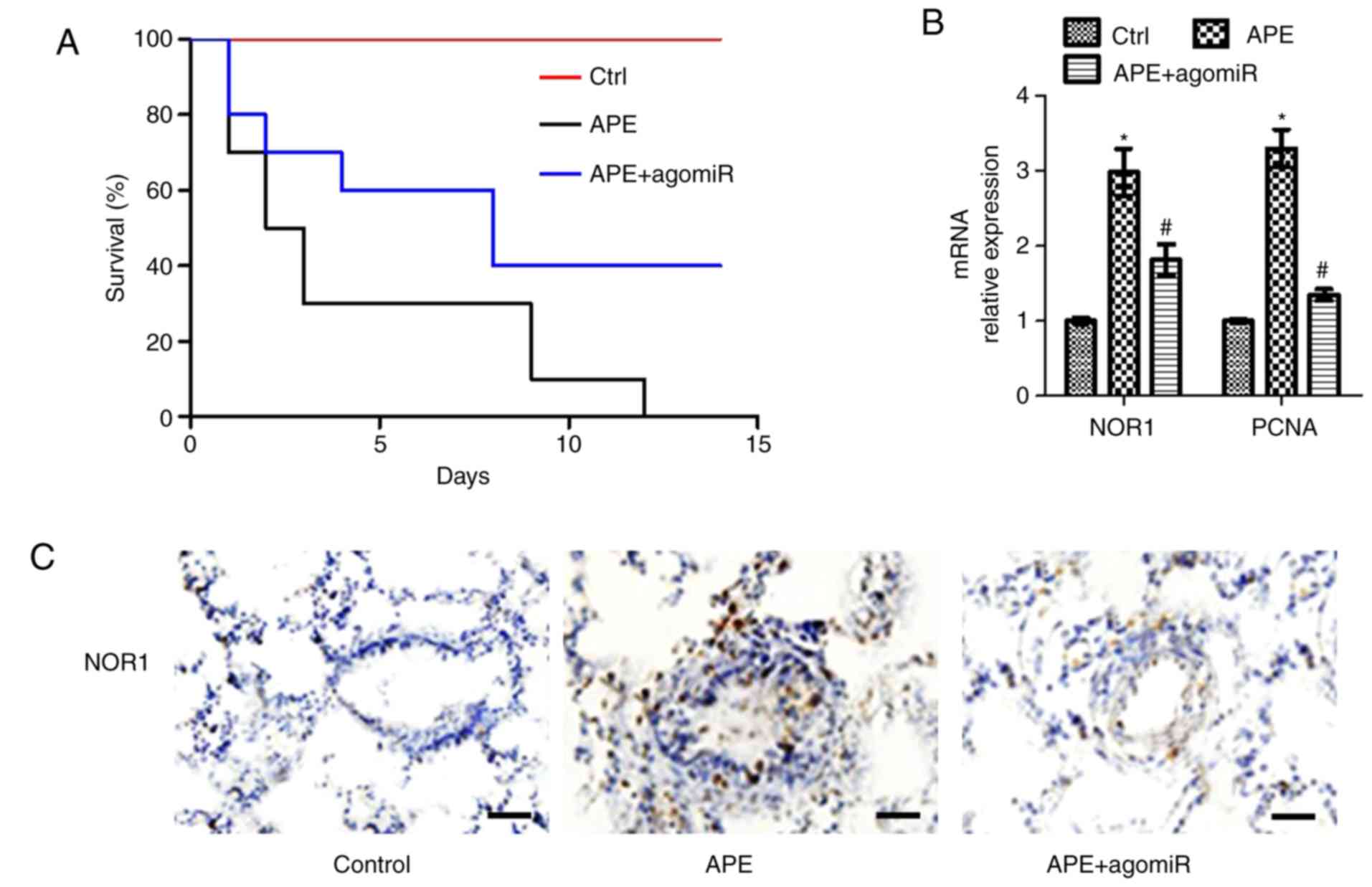
AgomiR-106b-5p improves APE-induced
mortality and increases pulmonary vascular proliferation in
mice
An APE mouse model was established for investigating
the positive effect of miR-106b-5p in vivo. Following
agomiR-106b-5p treatment, some mice were maintained for 15 days to
monitor the survival and some of these mice were euthanized on day
7 to assess pulmonary vascular proliferation. Survival of the
APE-induced mice after 7 days was 30% and all mice were dead after
12 days. AgomiR-106b-5p reduced the number of early deaths in
APE-mice and 40% of mice were still alive at the end of the study
(Fig. 6A). Considering the
importance of NOR-1 and PCNA in vascular proliferation, the mRNA
expression levels of NOR-1 and PCNA were assessed in the pulmonary
artery. APE significantly increased the levels of NOR-1 and PCNA
mRNA, whereas agomiR-106b-5p treatment significantly reduced the
mRNA expression levels (both P<0.05; Fig. 6B). As shown in the
immunohistochemistry results, NOR-1 expression was higher in the
narrowed blood vessels of APE-mice and lower in the neointima of
pulmonary vasculature in agomiR-106b-5p-treated APE-mice (Fig. 6C). These results suggest that
agomiR-106b-5p reduced APE-induced lung injury via inhibition of
NOR-1 in the pulmonary vasculature of mice.
Discussion
Numerous studies have shown that the excessive
proliferation and migration of PASMCs is involved in the
reconstruction of pulmonary vasculature and the increase in
pulmonary vascular resistance in APE (3,4).
In the present study, it was demonstrated that miR-106b-5p
expression was down-regulated in PDGF-induced PASMCs and in an APE
mouse model. miR-106b-5p suppressed the proliferation and migration
of PASMCs in the APE mouse model and in the PDGF-induced model, by
targeting NOR-1. Furthermore, NOR-1 overexpression reversed the
reduction of proliferation and migration in miR-106b-5p
agomiR-treated PASMCs. AgomiR-106b-5p improved APE-induced
mortality and pulmonary vascular proliferation in mice.
As technologies have improved, a large amount of
data regarding the expression of various miRNAs in APE have been
obtained. Using an Affymetrix miRNA array, Miao et al
(27) showed that 24 miRNAs were
upregulated and 22 miRNAs were downregulated miRNAs (including
miR-3148) when comparing a CTEPH group with control samples, and
the number of target genes co-regulated by miR-3148 and other
miRNAs was the highest of all miRNAs (27). Guo et al (14) found that miR-1260, miR-602,
miR-129-5p, miR-1908 and miR-483-5p were upregulated, and
miR-140-3p, miR-93, miR-22, miR-106b and let-7b were downregulated
in patients with CTEPH compared with the healthy controls, using a
miRCURY LNA Array. As an anti-tumor factor or tumor promoter,
miR-106b-5p, the matured product of miR-106b, has been reported to
serve an important role in multiple types of cancer. miR-106b-5p
targets CTSA to suppress the invasion and metastasis of colorectal
cancer (31), but miR-106b-5p
promotes stem cell-like properties of hepatocellular carcinoma
cells through targeting PTEN via a PI3K/Akt signaling pathway
(32). Li et al (20) demonstrated that miR-106b-5p binds
the 3′-UTR of Angpt2 to induce migration and tube formation of
HUVECs, and hCPFs-exosome transport to endothelial cells expressing
relatively low amounts of miR-106b-5p, promoting angiogenesis
through upregulation of Angpt2 (20). miR-106b-5p is pivotal in
regulating cell proliferation and migration. Studies have
demonstrated that PASMCs excessively proliferate and migrate to the
pulmonary artery intima following APE, resulting in the
reconstruction of the pulmonary vasculature and increasing
pulmonary vascular resistance (3,4).
Thus, it was hypothesized that miR-106b-5p may be closely
associated with excessive proliferation and migration of PASMCs
following APE. In the present study, the expression of miR-106b-5p
in patients with APE and in PDGF-induced PASMCs was determined, and
was shown to be decreased compared with the control. In
atherosclerotic plaques, Zhang et al (33) showed that the levels of
miR-106b-5p were downregulated compared with normal vascular
tissues. TNF-α downregulates miR-106b-5p levels and increases
caspase-3 activation and cell DNA fragmentation levels, and
upregulation of miR-106b-5p relieves TNF-α-induced apoptosis
through targeting PTEN in HUVECs (33). In the present study, it was shown
that NOR-1 was a direct target of miR-106b-5p, PDGF treatment
increased NOR-1 expression and proliferation of human PASMCs, but
miR-106b-5p agomiR reduced proliferation of PASMCs through
suppression of NOR-1 expression. miR-106b-5p agomiR ameliorated the
thrombus-induced intimal hyperplasia and NOR-1 expression in
neointimal SMCs of APE mice.
NOR-1 is an effector of inflammation, growth
factors, lipoproteins and thrombin, which regulates the migration
and proliferation of vascular cells (23–26). PDGF and thrombin induce rapid
upregulation of NOR-1 expression, via multiple pathways, including
alterations of cytosolic [Ca2+], via the ERK-mitogen
associated protein kinase signaling pathway and downstream
activation of cAMP response element binding protein (25,34). A previous study demonstrated that
NOR1 increases S phase kinase-associated protein 2 expression
during the proliferation which underlies neointima formation, which
itself is involved in vascular proliferation (35). NOR1 also mediates the expression
of several genes and proteins during vascular survival, such as
cellular inhibitor of apoptosis 2, Cyclin D1, antiproteinase α-2
macroglobulin, matrix metalloproteinase (MMP)-2 and MMP-9 (36–38). In a guidewire-induced arterial
injury mouse model, NOR1 deletion reduced neointima formation
during arterial injury (39),
whereas, NOR1-overexpression in SMCs increased mitogenic activity
and upregulated expression of embryonic smooth muscle myosin heavy
chain, resulting in increased neointima formation in transgenic
mice (40). In the present study,
NOR1 expression levels were increased in PDGF-induced PASMCs and
the neointimal SMCs of APE mice. PASMC viability and intimal
hyperplasia were significantly reduced following administration of
miR-106-5p agomiR via inhibition of NOR1. Rodriguez-Calvo et
al (41) showed that the
increased transcriptional activity of Cyclin D2 and increased
proliferation in NOR1-overexpressing aortic SMCs and carotid artery
ligation resulted in more severe neointimal formation and
hemadostenosis in NOR1-transgenic mice compared with the wild-type
mice. In the present study it was also shown that NOR-1
overexpression reversed the inhibition of proliferation in
miR-106b-5p agomiR-treated PASMCs. Furthermore, NOR1 has been
confirmed to be a downstream target of miR-638 (42,43). miR-638 expression was reduced in
PDGF-induced human SMCs, in a dose and time-dependent manner. NOR1
is a downstream target gene of miR-638 and NOR1 inhibition is
pivotal for miR-638-mediated suppression of PDGF-induced cyclin D1
expression and cell proliferation in human aortic SMCs (42). miR-638 also regulates aberrant
proliferation and migration of airway SMCs via targeting of NOR1
and Cyclin D1, which contributes to enhanced airway smooth muscle
mass associated with asthma.
In conclusion, the role of miR-106-5p in APE has not
been reported previously, to the best of our knowledge and it was
confirmed that miR-106-5p was a novel regulator of PASMC
proliferation and pulmonary vascular remodeling via targeting of
NOR1. miR-106-5p expression levels were decreased in PDGF-induced
PASMCs and overexpression of miR-106-5p decreased proliferation and
migration of PASMCs following PDGF stimulation. NOR1 overexpression
reversed the effects of miR-106-5p on PDGF-induced PASMCs,
suggesting that miR-106-5p serves a significant role in PASMCs
through mediating NOR1 activity. Together, these results suggest a
novel molecular mechanism underlying proliferation and migration of
PASMCs, improving understanding of the pathogenesis of APE and the
development of novel therapeutic targets.
Supplementary Information
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors’ contributions
QM, JZ and YM performed the experiments. HC and LP
were responsible for analysis and interpretation of the results.
HC, LP and HT designed the experiments, and HC wrote the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All animal experiments were performed according to
NIH Guide for the Care and Use of Laboratory Animals and approved
by the Institutional Animal Care and Use Committee of Xi’an
Jiaotong University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Liu T, Kang J and Liu F: Plasma levels of
microRNA-221 (miR-221) are increased in patients with acute
pulmonary embolism. Med Sci Monit. 24:8621–8626. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shi Y, Zhang Z, Cai D, Kuang J, Jin S, Zhu
C, Shen Y, Feng W, Ying S and Wang L: Urokinase attenuates
pulmonary thromboembolism in an animal model by inhibition of
inflammatory response. J Immunol Res. 2018:6941368. 2018.
View Article : Google Scholar
|
|
3
|
Zhou B, Sun G, Mei F and Xu H: The effects
of low-molecular-weight heparin on lung and pulmonary artery
injuries in acute pulmonary embolism rat model via platelet-derived
growth factor-β. Saudi Pharm J. 25:564–569. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Xu X, Shi L, Ma X, Su H, Ma G, Wu X, Ying
K and Zhang R: RhoA-Rho associated kinase signaling leads to
renin-angiotensin system imbalance and angiotensin converting
enzyme 2 has a protective role in acute pulmonary embolism. Thromb
Res. 176:85–94. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Caruso P, MacLean MR, Khanin R, McClure J,
Soon E, Southgate M, MacDonald RA, Greig JA, Robertson KE, Masson
R, et al: Dynamic changes in lung microRNA profiles during the
development of pulmonary hypertension due to chronic hypoxia and
monocrotaline. Arterioscler Thromb Vasc Biol. 30:716–723. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Feng Q, Tian T, Liu J, Zhang L, Qi J and
Lin X: Deregulation of microRNA-31a-5p is involved in the
development of primary hypertension by suppressing apoptosis of
pulmonary artery smooth muscle cells via targeting TP53. Int J Mol
Med. 42:290–298. 2018.PubMed/NCBI
|
|
8
|
Lee HW and Park SH: Elevated microRNA-135a
is associated with pulmonary arterial hypertension in experimental
mouse model. Oncotarget. 8:35609–35618. 2017.PubMed/NCBI
|
|
9
|
Lu Z, Li S, Zhao S and Fa X: Upregulated
miR-17 regulates hypoxia-mediated human pulmonary artery smooth
muscle cell proliferation and apoptosis by targeting mitofusin 2.
Med Sci Monit. 22:3301–3308. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yue Y, Zhang Z, Zhang L, Chen S, Guo Y and
Hong Y: miR-143 and miR-145 promote hypoxia-induced proliferation
and migration of pulmonary arterial smooth muscle cells through
regulating ABCA1 expression. Cardiovasc Pathol. 37:15–25. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Courboulin A, Paulin R, Giguère NJ,
Saksouk N, Perreault T, Meloche J, Paquet ER, Biardel S, Provencher
S, Côté J, et al: Role for miR-204 in human pulmonary arterial
hypertension. J Exp Med. 208:535–548. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Opitz I and Kirschner MB: Molecular
research in chronic thromboembolic pulmonary hypertension. Int J
Mol Sci. 20:pii: E784. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang L, Guo LJ, Liu J, Wang W, Yuan JX,
Zhao L, Wang J and Wang C: MicroRNA expression profile of pulmonary
artery smooth muscle cells and the effect of let-7d in chronic
thromboembolic pulmonary hypertension. Pulm Circ. 3:654–664. 2013.
View Article : Google Scholar
|
|
14
|
Guo L, Yang Y, Liu J, Wang L, Li J, Wang
Y, Liu Y, Gu S, Gan H, Cai J, et al: Differentially expressed
plasma microRNAs and the potential regulatory function of Let-7b in
chronic thromboembolic pulmonary hypertension. PLoS One.
9:e1010552014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sarrion I, Milian L, Juan G, Ramon M,
Furest I, Carda C, Cortijo Gimeno J and Mata Roig M: Role of
circulating miRNAs as biomarkers in idiopathic pulmonary arterial
hypertension: Possible relevance of miR-23a. Oxid Med Cell Longev.
2015:792846. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang Q, Ma J, Jiang Z, Wu F, Ping J and
Ming L: Diagnostic value of circulating microRNA-27a/b in patients
with acute pulmonary embolism. Int Angiol. 37:19–25. 2018.
|
|
17
|
Kessler T, Erdmann J, Vilne B, Bruse P,
Kurowski V, Diemert P, Schunkert H and Sager HB: Serum
microRNA-1233 is a specific biomarker for diagnosing acute
pulmonary embolism. J Transl Med. 14:1202016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhou X, Wen W, Shan X, Qian J, Li H, Jiang
T, Wang W, Cheng W, Wang F, Qi L, et al: MiR-28-3p as a potential
plasma marker in diagnosis of pulmonary embolism. Thromb Res.
138:91–95. 2016. View Article : Google Scholar
|
|
19
|
Zhang Y, Peng B and Han Y: MiR-23a
regulates the proliferation and migration of human pulmonary artery
smooth muscle cells (HPASMCs) through targeting BMPR2/Smad1
signaling. Biomed Pharmacother. 103:1279–1286. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li Y, Liang J, Hu J, Ren X and Sheng Y:
Down-regulation of exosomal miR-106b-5p derived from cholesteatoma
perimatrix fibroblasts promotes angiogenesis in endothelial cells
by overexpression of Angiopoietin 2. Cell Biol Int. 42:1300–1310.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhao Y and Bruemmer D: NR4A orphan nuclear
receptors: Transcriptional regulators of gene expression in
metabolism and vascular biology. Arterioscler Thromb Vasc Biol.
30:1535–1541. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Marti-Pamies I, Cañes L, Alonso J,
Rodriguez C and Martinez-Gonzalez J: The nuclear receptor
NOR-1/NR4A3 regulates the multifunctional glycoprotein vitronectin
in human vascular smooth muscle cells. FASEB J. 31:4588–4599. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Martinez-Gonzalez J, Rius J, Castelló A,
Cases-Langhoff C and Badimon L: Neuron-derived orphan receptor-1
(NOR-1) modulates vascular smooth muscle cell proliferation. Circ
Res. 92:96–103. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rius J, Martinez-Gonzalez J, Crespo J and
Badimon L: Involvement of neuron-derived orphan receptor-1 (NOR-1)
in LDL-induced mitogenic stimulus in vascular smooth muscle cells:
Role of CREB. Arterioscler Thromb Vasc Biol. 24:697–702. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Martorell L, Martinez-Gonzalez J, Crespo
J, Calvayrac O and Badimon L: Neuron-derived orphan receptor-1
(NOR-1) is induced by thrombin and mediates vascular endothelial
cell growth. J Thromb Haemost. 5:1766–1773. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Thakar RG, Cheng Q, Patel S, Chu J, Nasir
M, Liepmann D, Komvopoulos K and Li S: Cell-shape regulation of
smooth muscle cell proliferation. Biophys J. 96:3423–3432. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Miao R, Wang Y, Wan J, Leng D, Gong J, Li
J, Zhang Y, Pang W, Zhai Z and Yang Y: Microarray analysis and
detection of MicroRNAs associated with chronic thromboembolic
pulmonary hypertension. Biomed Res Int. 2017:8529796. 2017.
View Article : Google Scholar
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
29
|
Liu X, Qu J, Xue W, He L, Wang J, Xi X,
Liu X, Yin Y and Qu Y: Bioinformatics-based identification of
potential microRNA biomarkers in frequent and non-frequent
exacerbators of COPD. Int J Chron Obstruct Pulmon Dis.
13:1217–1228. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Latonen L, Afyounian E, Jylhä A, Nättinen
J, Aapola U, Annala M, Kivinummi KK, Tammela TTL, Beuerman RW,
Uusitalo H, et al: Integrative proteomics in prostate cancer
uncovers robustness against genomic and transcriptomic aberrations
during disease progression. Nat Commun. 9:11762018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ni S, Weng W, Xu M, Wang Q, Tan C, Sun H,
Wang L, Huang D, Du X and Sheng W: miR-106b-5p inhibits the
invasion and metastasis of colorectal cancer by targeting CTSA.
Onco Targets Ther. 11:3835–3845. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shi DM, Bian XY, Qin CD and Wu WZ:
miR-106b-5p promotes stem cell-like properties of hepatocellular
carcinoma cells by targeting PTEN via PI3K/Akt pathway. Onco
Targets Ther. 11:571–585. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang J, Li SF, Chen H and Song JX:
MiR-106b-5p inhibits tumor necrosis factor-α-induced apoptosis by
targeting phosphatase and tensin homolog deleted on chromosome 10
in vascular endothelial cells. Chin Med J (Engl). 129:1406–1412.
2016. View Article : Google Scholar
|
|
34
|
Nomiyama T, Nakamachi T, Gizard F, Heywood
EB, Jones KL, Ohkura N, Kawamori R, Conneely OM and Bruemmer D: The
NR4A orphan nuclear receptor NOR1 is induced by platelet-derived
growth factor and mediates vascular smooth muscle cell
proliferation. J Biol Chem. 281:33467–33476. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gizard F, Zhao Y, Findeisen HM, Qing H,
Cohn D, Heywood EB, Jones KL, Nomiyama T and Bruemmer D:
Transcriptional regulation of S phase kinase-associated protein 2
by NR4A orphan nuclear receptor NOR1 in vascular smooth muscle
cells. J Biol Chem. 286:35485–35493. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Alonso J, Galán M, Marti-Pamies I, Romero
JM, Camacho M, Rodríguez C and Martinez-Gonzalez J: NOR-1/NR4A3
regulates the cellular inhibitor of apoptosis 2 (cIAP2) in vascular
cells: Role in the survival response to hypoxic stress. Sci Rep.
6:340562016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang CG, Lei W, Li C, Zeng DX and Huang
JA: Neuron-derived orphan receptor 1 promoted human pulmonary
artery smooth muscle cells proliferation. Exp Lung Res. 41:208–215.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rodriguez-Calvo R, Ferrán B, Alonso J,
Marti-Pamies I, Aguiló S, Calvayrac O, Rodríguez C and
Martinez-Gonzalez J: NR4A receptors up-regulate the antiproteinase
alpha-2 macroglobulin (A2M) and modulate MMP-2 and MMP-9 in
vascular smooth muscle cells. Thromb Haemost. 113:1323–1334. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nomiyama T, Zhao Y, Gizard F, Findeisen
HM, Heywood EB, Jones KL, Conneely OM and Bruemmer D: Deficiency of
the NR4A neuron-derived orphan receptor-1 attenuates neointima
formation after vascular injury. Circulation. 119:577–586. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Rodriguez-Calvo R, Guadall A, Calvayrac O,
Navarro MA, Alonso J, Ferrán B, de Diego A, Muniesa P, Osada J,
Rodríguez C and Martinez-Gonzalez J: Over-expression of
neuron-derived orphan receptor-1 (NOR-1) exacerbates neointimal
hyperplasia after vascular injury. Hum Mol Genet. 22:1949–1959.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Rodriguez-Calvo R, Guadall A, Calvayrac O,
Alonso J, Ferran B, Marti I, Navarro MÁ, de Diego A, Osada J,
Rodríguez C and Martinez-Gonzalez J: The nuclear receptor NOR-1
regulates the activation of vascular cells and vascular remodelling
in response to hemodynamic stress. Clin Investig Arterioscler.
26:66–75. 2014.(In Spanish). PubMed/NCBI
|
|
42
|
Li P, Liu Y, Yi B, Wang G, You X, Zhao X,
Summer R, Qin Y and Sun J: MicroRNA-638 is highly expressed in
human vascular smooth muscle cells and inhibits PDGF-BB-induced
cell proliferation and migration through targeting orphan nuclear
receptor NOR1. Cardiovasc Res. 99:185–193. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang H, Yao H, Yi B, Kazama K, Liu Y,
Deshpande D, Zhang J and Sun J: MicroRNA-638 inhibits human airway
smooth muscle cell proliferation and migration through targeting
cyclin D1 and NOR1. J Cell Physiol. 234:369–381. 2018. View Article : Google Scholar : PubMed/NCBI
|