Introduction
Prostate cancer (PCa) is one of the major types of
malignancy in men, with a high incidence rate (1). Currently, the primary treatment
strategies for these tumors are surgical castration combined with
androgen deprivation therapy or antiandrogens, radiotherapy,
chemotherapy, immunotherapy and other combined treatments (2,3).
In patients with PCa, the aim of these treatments is initiation of
apoptosis in tumor cells, thereby inhibiting cancer growth and
spread.
Cantharidin, a type of terpenoid, is extracted from
the blister beetles (Mylabris genus) and is used in
Traditional Chinese Medicine (4,5).
Cantharidin possesses antibiotic, antiviral and antitumor
qualities, and can affect immune responses (4,6).
Cantharidin has been used as a medicinal agent for >2,000 years
and has a number of applications, including treatment of edema and
warts (4). Cantharidin treatment
initiates cell cycle arrest and attenuates apoptosis in various
types of carcinomas including those of the bladder (7), breast (8), liver (9,10),
and colon (11), buccal mucosa
and leukemia (12,13).
Tumor necrosis factor (TNF)-related
apoptosis-inducing ligand (TRAIL) is the only identified protein
that can specifically initiate apoptosis of tumor cells without
affecting noncancerous cells (3).
TRAIL, a type II transmembrane protein, is a part of the TNF
superfamily of cytokines identified in 1995 by Wiley et al
(14). TRAIL is able to bind the
death receptors (DR)-4 and DR-5, promoting the death-inducing
signaling complexes activity of caspase-3 and caspase-8, thereby
initiating tumor cell death (15). However, downregulation of the
death receptors and pro-apoptotic proteins, and upregulation of the
anti-apoptotic proteins such as cellular FLICE-like inhibitory
protein (c-FLIP), leads to TRAIL-mediated apoptosis resistance
(16-19). Therefore, identification of TRAIL
sensitizers is may help to overcome the barrier of TRAIL
resistance.
Autophagy is a multi-step cellular process in which
cytoplasmic constituents are targeted to lysosomes for degradation,
and is associated with several diseases including cancer (20). Recent studies have revealed that
autophagy has a dual mechanism, having either pro-survival or
pro-death characteristics depending on the cell type and stimulus
strength (21-23). Autophagy acts in a pro-survival
manner through cytoprotective events that favor cell survival and
decrease the incidence of cell death. Conversely, autophagy can
augment a pro-death signaling system and ultimately result in cell
death in cancer (24). The
complete autophagic flux engages microtubule-associated proteins
1A/1B light chain 3B (LC3), which serves an important role in cargo
elimination, phagophore elongation and autophagosome composition,
and the sequestosome 1 (p62)-cargo system is selectively attached
to autophagosomes through the interaction of p62 and LC3 (20).
In the present study, the effectiveness of
cantharidin in sensitization of PCa cells to TRAIL was examined. We
aimed to elucidate the molecular pathways underlying the effect of
cantharidin and synergistic effect of cantharidin and TRAIL in
DU145 cells. It was identified that cantharidin treatment
sensitizes DU145 tumor cells to TRAIL-mediated apoptosis via
autophagy flux. The combined treatment of cantharidin and TRAIL may
represent a viable therapeutic strategy for certain cases of
TRAIL-resistant cancer.
Materials and methods
Cell culture
Human prostate DU145 and LNCaP cell lines were
maintained in RPMI-1640 medium (Gibco; Thermo Fisher Scientific,
Inc.) containing 10% fetal bovine serum (FBS) and 1%
penicillin/streptomycin. During the experiments, the culture medium
was changed to RPMI-1640 containing 1% FBS. In the present study,
TRAIL treatment intervals of 1 and 2 h were used for the western
blot analysis and the cell viability assay, respectively. It was
identified that a 2 h treatment period for the combination of TRAIL
and cantharidin induced a high level of cell death that affected
the detection of certain proteins. The cells were maintained at
37°C and 5% CO2 in a humidified incubator.
Reagents
Cantharidin was acquired from Sigma-Aldrich; Merck
KGaA, while TRAIL was attained from AbFrontier Co., Ltd.
Cell viability assay
The DU145 and LNCaP cells were plated
2×105 cells/well in 12-well plates and treated with
cantharidin (0, 0.25, 0.50 and 1 μM) for 18 h and were
further exposed to TRAIL (200 ng/ml) for an additional 2 h. For
autophagy inhibition, cells were treated 10 μM chloroquine
(CQ) 1 h prior to cantharidin or TRAIL treatment at 37°C. Cell
morphology was assessed under a light microscope (Nikon
Corporation) at magnification, ×400. Cell viability was determined
by crystal violet staining method as previously described (25).
Trypan blue exclusion assay
Cell viability was evaluated by trypan blue
exclusion assay using a hemocytometer. The DU145 and LNCaP cells
were seeded 1×105 cells/well in 24-well plate, after
treatment the cells were dissociated with trypsin-EDTA are were
then suspended in 1 ml PBS per well and 1 ml trypan blue solution
(Sigma-Aldrich; Merck KGaA). After 5 min at 20°C, the cells were
counted using a hemocytometer (Marienfeld Corp.) under a light
microscope (Nikon Corp.) at ×400 magnification. Untreated cells
were used as control, and the cell viability was compared to the
control. Each treatment was performed in triplicate.
Immunofluorescence staining
DU145 cells were cultured on poly-L-lysine-coated
coverslips. Following cantharidin treatment with or without CQ or
ATG5 siRNA, the cells were were fixed with 4% paraformaldehyde at
4°C for 10 min and permeabilized with 0.1% Triton X-100. The cells
were then incubated for 1 h at room temperature in a blocking
solution (5% FBS in TBS) followed by overnight incubation at 4°C
with anti p62 antibody (1:250; cat. no. PAS-20839; Invitrogen;
Thermo Fisher Scientific, Inc.). Following washing with PBS, the
cells were incubated with the Alexa Fluor®
488-conjugated donkey polyclonal anti-rabbit secondary antibody
(1:500; cat. no. A-21206; Thermo Fisher Scientific, Inc.) for 2 h
in the dark. Finally, immunostaining was visualized under a
fluorescence microscope at magnification, ×200.
Western blot analysis
Western blot analysis was performed as described
previously (26).
Immunoprecipitation assay buffer (Qiagen, Inc.) was used for total
protein extraction. The supernatant was collected by centrifugation
at 11,200 × g and 4°C for 10 min. The protein concentration was
determined using a BCA protein assay kit (Thermo Fisher Scientific;
Inc.). Protein (30 μg) were separated on 10% SDS-PAGE gels
and transferred to polyvinylidene fluoride membranes. The membranes
were blocked with 5% non-fat dried milk at 25°C for 1 h. Then
membranes were incubated with primary antibodies for 1 h at 25°C,
the following antibodies were used: DR-5 (1:1,000; cat. no.
ab181846; Abcam); β-actin (1:10,000; cat. no. A5441; Sigma-Aldrich;
Merck KGaA); c-FLIP (1:1,000; cat. no. ADI-AAP-440; Enzo life
sciences, USA); LC3A/B (1:1,000; cat. no. 4108; Cell Signaling
Technology, Inc.); p62 (1:250; cat. no. PAS-20839; Invitrogen;
Thermo Fisher Scientific, Inc.); ATG5 (1:1,000; cat. no. c.s12994s;
Cell Signaling Technology, Inc.); and cleaved caspase-8 (1:1,000;
cat. no. 551242; BD Pharmingen; BD Biosciences). Then membranes
were incubated with horseradish peroxidase-conjugated secondary
antibodies (1:5,000; cat. nos. ADI-SAB-100 and ADI-SAB-300; Enzo
Life Science, Inc.) at 25°C for 1 h. The immune reactive protein
bands were visualized using enhanced chemiluminescence detection
system (GE Healthcare Life Sciences) and detected with
chemiluminiscence imaging system (Fusion FX7; Viber Lourmat). The
intensities of the protein bands were determined using Image J Java
1.8.0 software (National Institutes of Health).
Transmission electron microscopy (TEM)
analysis
Following the fixation of cells in 2% glutaraldehyde
(Electron Microscopy Sciences) and 2% paraformaldehyde (Electron
Microscopy Sciences) in 0.05 M sodium cacodylate (pH 7.2; Electron
Microscopy Sciences) for 2 h at 4°C, the specimens were fixed in 1%
osmium tetroxide (Electron Microscopy Sciences) for 1 h at 4°C,
dehydrated in increasing ethanol concentration (25, 50, 70, 90 and
100%) for 5 min each, and embedded in Embed 812 epoxy resin
(Electron Microscopy Sciences) for 48 h at 60°C according to the
manufacturers' protocol. Ultrathin sections (60 nm) were prepared
using a LKBIII ultratome (Leica Microsystems GmbH) and stained with
0.5% uranyl acetate (Electron Microscopy Sciences) for 20 min and
0.1% lead citrate (Electron Microscopy Sciences) for 7 min at room
temperature. Images were captured using a Hitachi H7650 electron
microscope (Hitachi, Ltd.) at magnification, ×10,000, installed at
the Center for University-Wide Research Facilities (CURF), Jeonbuk
National University.
Small interfering RNA transfection
DU145 cells were seeded 1×105 cells/well
in 24-well plates for 24 h and then transfected with Silencer
Select small interfering RNA (ATG5 siRNA; oligo ID HSS114103;
Sequence GGU UUG GAC GAA UUC CAA CUU GUU U; Invitrogen; Thermo
Fisher Scientific, Inc.) using Lipofectamine® 2000
(Thermo Fisher Scientific, Inc.) according to the manufacturer's
protocol. Concomitantly, non-targeting siRNA was transfected as a
negative control. The cells were incubated with ATG5 siRNA or
negative control siRNA for 6 h and the medium was then changed to
RPMI-1640 with 10% FBS for 24 h. The cells were then treated with
neferine, or neferine in combination with TRAIL.
Statistical analysis
Statistical analyses were performed using GraphPad
v.5.01 software (GraphPad Software Inc.). All experiments were
performed in triplicate, and the data are expressed as the mean ±
standard error of the mean. Significant differences between control
and treated samples were analyzed by one-way analysis of variance
followed by Tukey's post hoc test.
Results
Effect of cantharidin on TRAIL-mediated
apoptosis in PCa cells
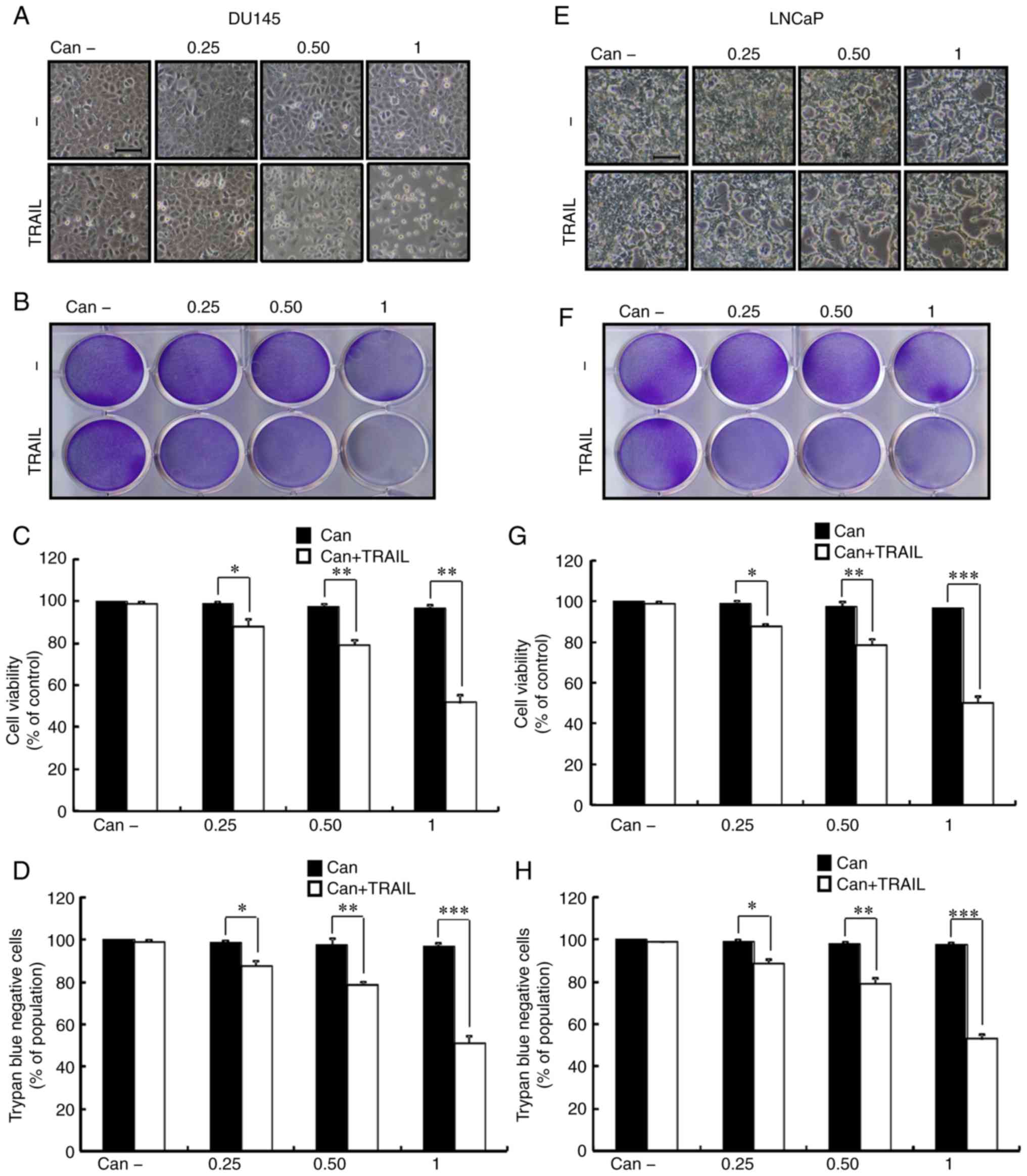
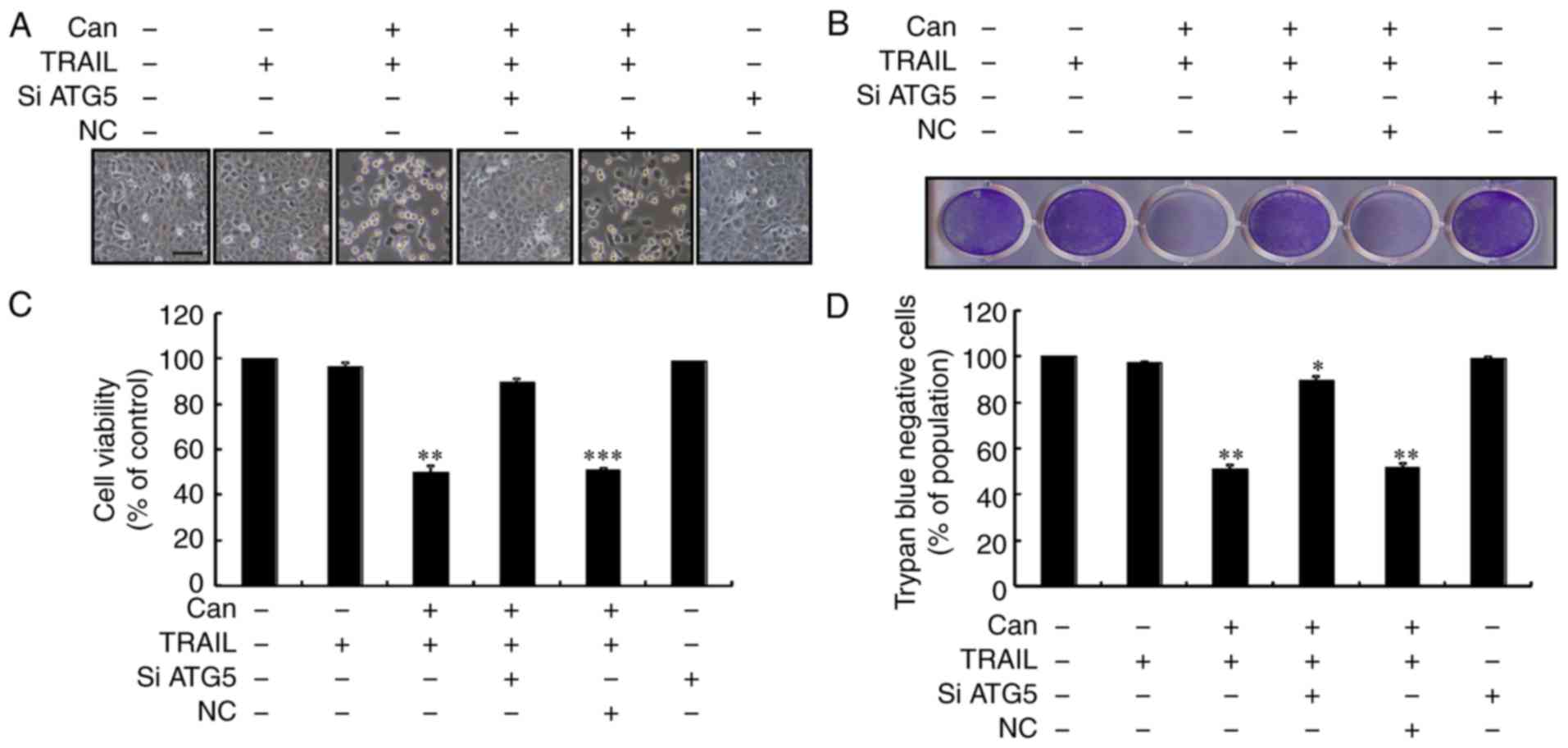
To observe the potential suppressive effect of
cantharidin on PCa cell growth, DU145 and LNCaP cells were treated
consecutively with cantharidin at different concentrations for 18 h
and then TRAIL for an additional 2 h. As shown in Fig. 1, TRAIL or cantharidin alone only
marginally induced cell death, and no morphological changes were
observed. In addition, the combined regimen of cantharidin and
TRAIL significantly attenuated cell viability, whereas treatment
with cantharidin or TRAIL alone had no effect on apoptosis
(Fig. 1A-H). The results
suggested that cantharidin effectively sensitized human PCa cells
to TRAIL-mediated apoptosis.
Cantharidin induced autophagy and
enhanced cells to TRAIL-mediated apoptosis
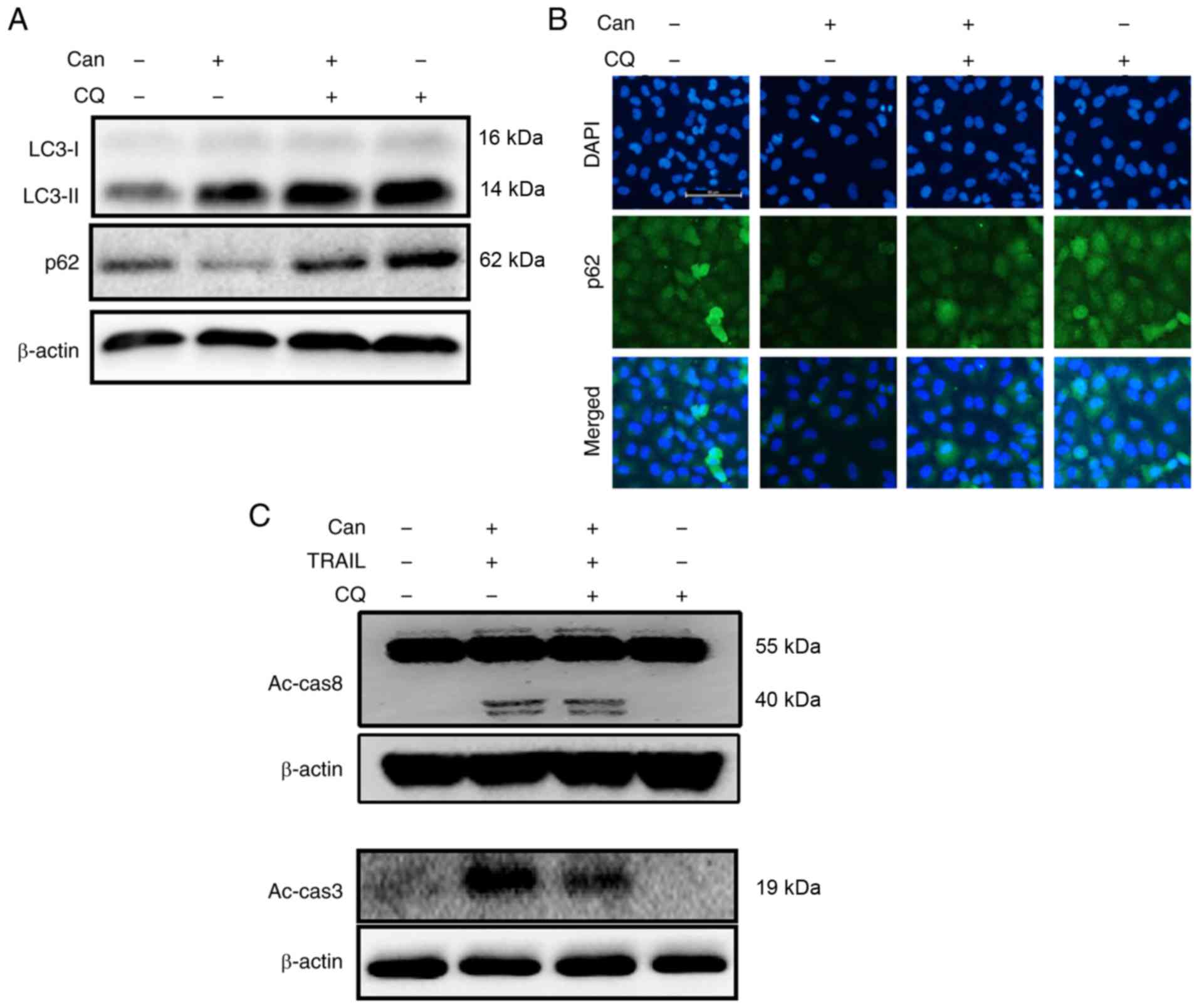
In response to cantharidin treatment, DU145 cells
exhibited significantly upregulated LC3-II expression levels and
attenuated p62 expression levels (Figs. 2A, and S1A and B). The protein levels assayed
by immunofluorescence staining were consistent with the results of
western blot analysis (Figs. 2B
and S1C). TEM demonstrated that
both autophagic and vacant vacuoles were secreted in
cantharidin-treated cells (Fig.
2C). Cells treated with cantharidin and TRAIL in combination
exhibited increased expression of activated caspase (Ac-cas)-3 and
Ac-cas8, respectively (Fig. 2D).
These data suggest that cantharidin can initiate autophagy in DU145
cells.
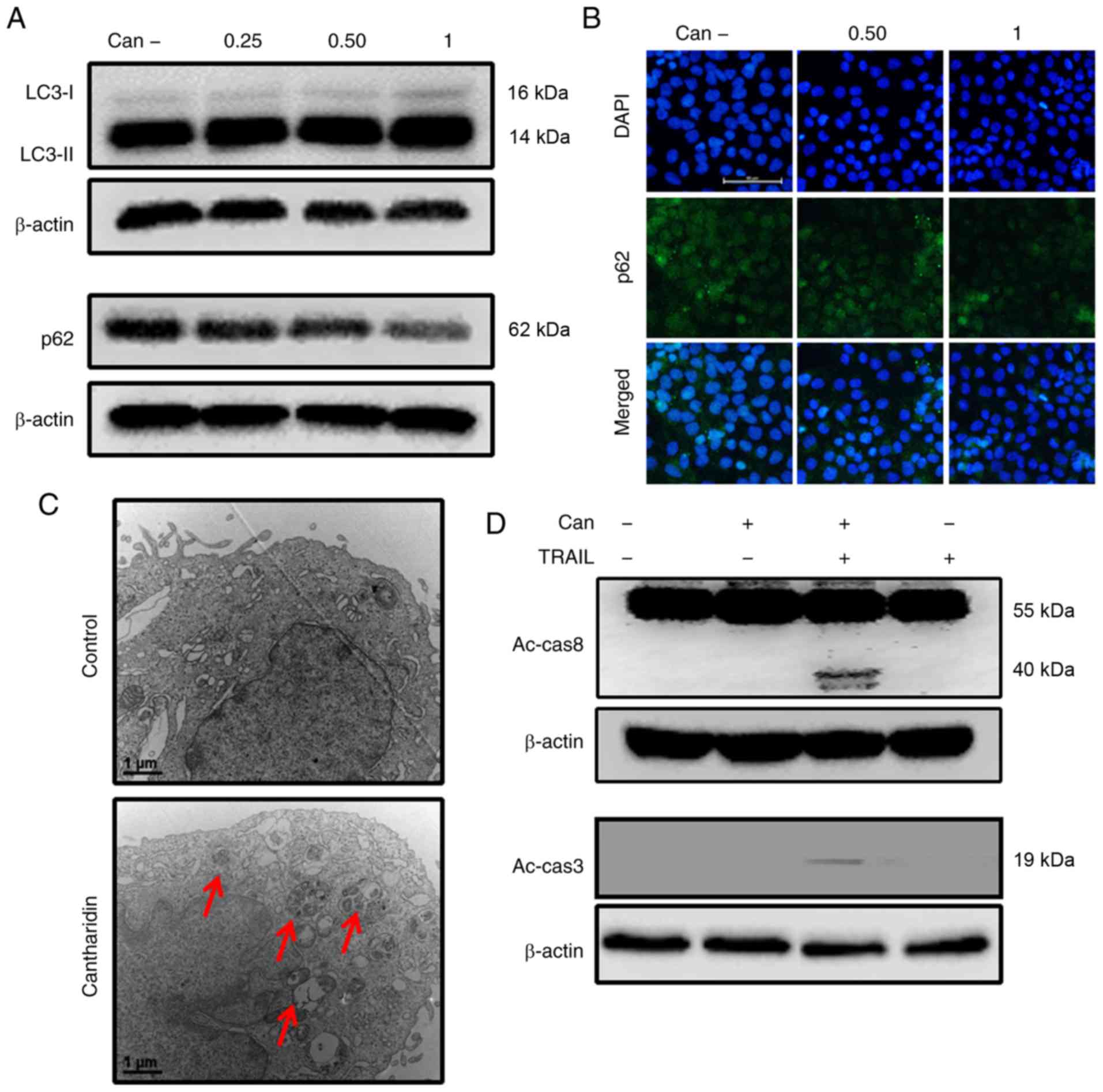 | Figure 2Can induces autophagy and enhances
TRAIL-mediated apoptosis of cells. DU145 cells were treated with
Can (0, 0.25, 0.50 and 1 μM) for 18 h. (A) The levels of
LC3-II and p62 were assessed by western blot analysis. (B) DU145
cells were treated with Can (0, 0.50 and 1 μM) for 18 h.
Cells were then immunostained with p62 (green) and evaluated for
fluorescence (scale bar=50 μm; magnification, ×200). (C) The
formation of autophagosomes in treated cells was examined by
transmission electron microscopy. Red arrows indicate
autophagosomes. Scale bar=1 μm. (D) DU145 cells were treated
with Can (1 μM) for 18 h and then TRAIL for an additional 1
h. The levels of Ac-cas3 and Ac-cas8 were assessed by western blot
analysis. β-actin was used as the control. TRAIL, tumor necrosis
factor-related apoptosis-inducing ligand; can, cantharidin; Ac-cas,
activated caspase; p62, sequestosome 1; LC3-I, cytoplasmic
microtubule-associated proteins 1A/1B light chain 3B; LC3-II,
lipid-modified microtubule-associated proteins 1A/1B light chain
3B. |
Autophagy inhibition rescues
TRAIL-mediated apoptosis sensitization through cantharidin
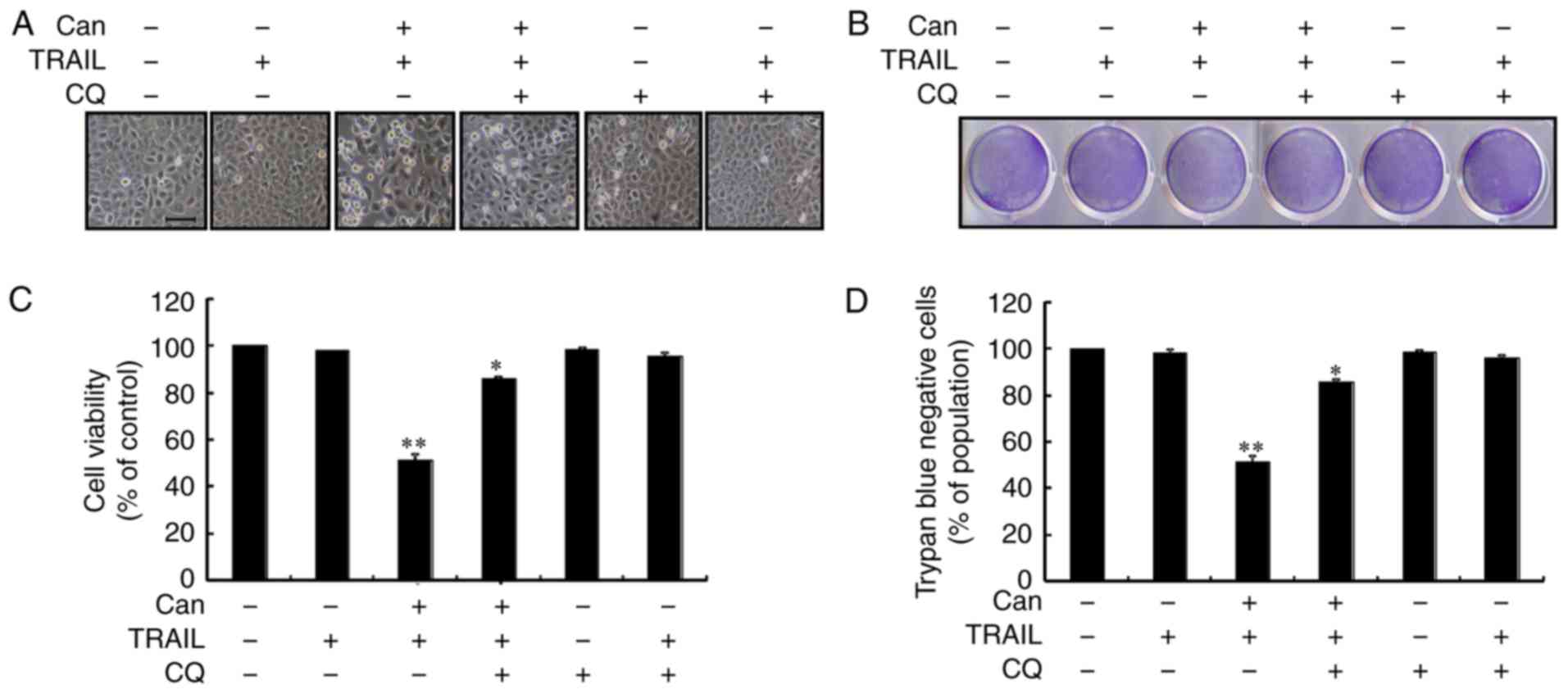
Autophagy control was further confirmed by examining
the autophagy flux following treatment with CQ. CQ promoted the
accumulation of membrane-bound LC3-II and led to an increase in p62
levels (Figs. 3A, and S2A and B). Immunofluorescence staining
confirmed the increase in p62 expression (Figs. 3B and S2C). However, co-treatment with
cantharidin, TRAIL and CQ attenuated the upregulation of Ac-cas3
and Ac-cas8 (Fig. 3C). The
effects of cantharidin on the viability of the DU145 cells in
response to CQ were also investigated. Cell morphology analysis
confirmed that CQ treatment more effectively attenuated the cell
death compared with the combined regimen of cantharidin and TRAIL
(Fig. 4A). Co-treatment with
cantharidin, TRAIL and CQ markedly increased the viability of DU145
cells (Fig. 4B-D). This suggests
that cantharidin sensitized TRAIL-mediated apoptosis may be
attenuated through CQ by blocking autophagy flux.
Genetic attenuation of autophagy rescues
cantharidin-sensitized TRAIL-mediated apoptosis
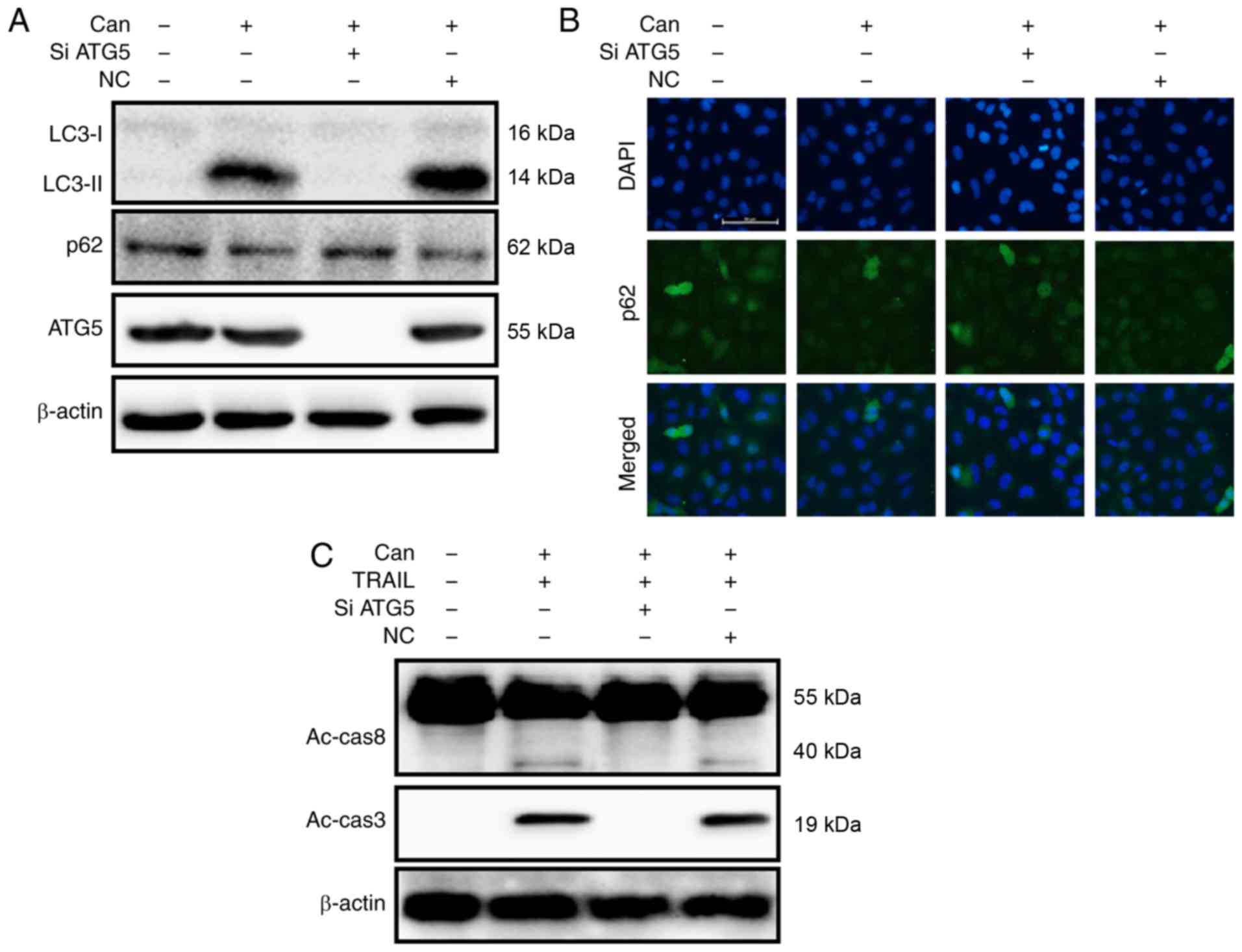
To investigate the inhibition of autophagic flux,
the autophagy-related 5 (ATG5) gene was knocked down. ATG5
knockdown decreased cantharidin-initiated expression of LC3-II and
markedly increased p62 protein levels (Figs. 5A and S3A). The protein levels of p62,
determined by immunofluorescence staining, were consistent with
those determined by western blot analysis (Figs. 5B and S3B). However, co-treatment with
cantharidin, ATG5 siRNA and TRAIL attenuated the increase in
Ac-cas3 and Ac-cas8 expression (Fig.
5C). Cell morphology analysis confirmed that the ATG5 siRNA
regimen regulated cell death more effectively compared with
combined treatment of cantharidin and TRAIL (Fig. 6A). Co-treatment with cantharidin,
TRAIL and ATG5 siRNA markedly increased the viability of DU145
cells and significantly attenuated cell death (Fig. 6B-D). These data indicate that
cantharidin-sensitized prolongation of TRAIL-mediated apoptosis can
be facilitated by genetic attenuation of autophagy and induction of
autophagy flux.
Downregulation of c-FLIP and upregulation
of DR-5 by cantharidin rescues TRAIL resistance
The results from the western blot analysis revealed
that cantharidin attenuated the expression of c-FLIP and
upregulated the expression of DR-5 (Figs. 7A and S4A and B). The combined regimen of
canthar-idin and TRAIL decreased c-FLIP and increased DR-5 levels
(Figs. 7B and S4C and D). c-FLIP expression was
increased and DR-5 upregulation was partially inhibited by combined
treatment with CQ and cantharidin (Fig. 7C). Furthermore, a combined regimen
of ATG5 siRNA and cantharidin increased c-FLIP levels while DR-5
upregulation was partially inhibited (Fig. 7D). These data suggest that
cantharidin down-regulated c-FLIP expression and upregulated DR-5
expression and thereby sensitized TRAIL-mediated apoptosis through
the initiation of autophagy flux.
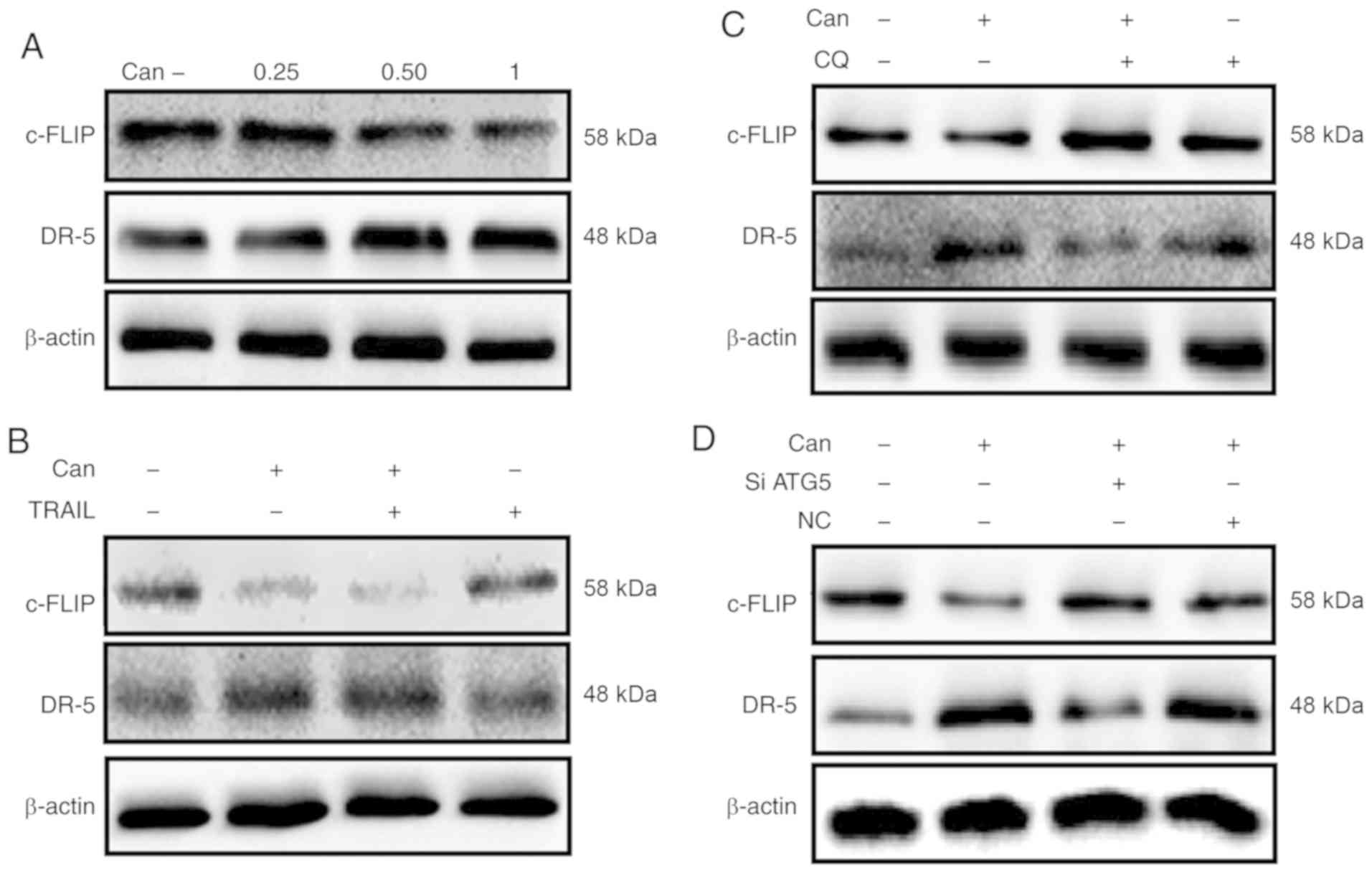 | Figure 7Downregulation of c-FLIP and
upregulation of DR-5 by Can attenuates TRAIL resistance. DU145
cells were treated with Can (0, 0.25, 0.50 and 1 μM) for 18
h. (A) The levels of c-FLIP and DR-5 were assessed by western blot
analysis. DU145 cells were treated with Can (1 μM) for 18 h
and then TRAIL (200 ng/ml) for an additional 1 h. (B) The levels of
c-FLIP and DR-5 were assessed by western blot analysis. DU145 cells
were pretreated with CQ (10 μM) for 1 h prior to exposure to
Can (1 μM) for 18 h. (C) The levels of c-FLIP and DR-5 were
assessed by western blot analysis. DU145 cells were pretreated with
ATG5 siRNA or negative control siRNA for 24 h prior to their
exposure to Can (1 μM) for 18 h. (D) The levels of c-FLIP
and DR-5 assessed by western blot analysis. c-FLIP, cellular
FLICE-like inhibitory protein; DR-5, death receptor 5; TRAIL, tumor
necrosis factor-related apoptosis-inducing ligand; ATG5,
autophagy-related 5; siRNA, small interfering RNA; Can,
cantharidin; CQ, chloroquine; NC, negative control. |
Discussion
The present study investigated the mechanism of
cantharidin-mediated TRAIL sensitization. Cantharidin treatment
induced autophagy flux, decreased c-FLIP expression and increased
DR-5 expression. It also enhanced the cleavage of caspase-8 and
caspase-3 leading to the induction of apoptosis. Furthermore,
cantharidin treatment sensitized TRAIL-mediated apoptosis in
multiple cancer cells. Therefore, the results suggested that
cantharidin treatment may be used for suppression of
TRAIL-resistance.
TRAIL can specifically initiate apoptosis in human
malignant and transformed cells (27), whereas normal cells can resist
this cell death process. The precise understanding of the function
of TRAIL may present a novel therapy regimen for cancer treatment
(28). Although various types of
tumor cells are sensitive to TRAIL-mediated apoptosis, other cells
including PCa are resistant (29). Cantharidin is a known inhibitor of
protein phosphatases 1 and 2A (PP1 and PP2A) (30). It is a terpenoid that has been
reported to exhibit promising biological and clinical effects
(30). Cantharidin sensitized
TRAIL-mediated apoptosis in prostate cancer cells. In the present
study, the combined treatment of cantharidin and TRAIL enhanced
cancer cell death both in DU145 and LNCaP cell lines, suggesting
that cantharidin has the ability to decrease TRAIL-resistance in
prostate cancer cells.
Certain studies have reported that cantharidin
attenuates tumor cell propagation and induction of autophagy
(31-34). Autophagy has been widely
investigated in response to various stimuli, including particular
anticancer agents (35). In the
present study, cantharidin treatment induced an increase in LC-3II
expression and a decrease in p62 expression, which indicated that
cantharidin treatment enhanced autophagy flux. However, cantharidin
treatment alone cannot induce cancer cell apoptosis; the combined
treatment of cantharidin and TRAIL led to cleavage of caspase-8 and
caspase-3. The autophagy mechanism is controlled by evolutionarily
conserved autophagy-associated ATG proteins, which are engaged in
the enucleation and elongation of autophagosome composition
(36). The present study also
confirmed that a diacritic pharmacological and genetic autophagy
inhibitor can rescue cantharidin-sensitized apoptosis initiation
through TRAIL. These data indicated that cantharidin treatment
sensitizes TRAIL-mediated activation of caspase through the
activation of autophagy.
TRAIL has 2 death receptors, DR-4 and DR-5, which
initiate cancer cell death signals (15). Previous studies have demonstrated
that the drug-initiated downregulation of c-FLIP and upregulation
of DR-5 promotes apoptosis in tumor cells (37-39). The results of the present study
also indicated that cantharidin mediated the downregulation of
c-FLIP and upregulation of DR-5, and that this effect could be
rescued by pharmacological and genetic suppression of autophagy.
These results indicated that cantharidin-induced autophagy
sensitized TRAIL-mediated cancer cell apoptosis through the
downregulation of c-FLIP and upregulation of DR-5.
In summary, the downregulation of c-FLIP and
upregulation of DR-5 by cantharidin treatment sensitized
TRAIL-mediated apoptosis in DU145 tumor cells via autophagy flux.
The combined regimen of cantharidin and TRAIL may become an
adequate therapeutic strategy for the treatment of certain cases of
TRAIL-resistant cancer.
Supplementary Data
Funding
The present study was supported by a grant from the
National Research Foundation of Korea funded by the Ministry of
Education (grant no. 2019R1A2B5B02069765).
Availability of data and materials
All data generated or analyzed during the present
study are included in this published article or are available from
the corresponding author upon reasonable request.
Authors' contributions
UMN, HY and SYP designed, executed the study and
analyzed the data. UMN and HY wrote the manuscript. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Jemal A, Siegel R, Xu J and Ward E: Cancer
statistics, 2010. CA Cancer J Clin. 60:277–300. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gamat M and McNeel DG: Androgen
deprivation and immunotherapy for the treatment of prostate cancer.
Endocr Relat Cancer. 24:T297–T310. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hao L, Zhao Y, Li ZG, He HG, Liang Q,
Zhang ZG, Shi ZD, Zhang PY and Han CH: Tumor necrosis
factor-related apoptosis-inducing ligand inhibits proliferation and
induces apoptosis of prostate and bladder cancer cells. Oncol Lett.
13:3638–3640. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dorn DC, Kou CA, Png KJ and Moore MA: The
effect of cantharidins on leukemic stem cells. Int J Cancer.
124:2186–2199. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nickolls LC and Teare D: Poisoning by
cantharidin. Br Med J. 2:1384–1386. 1954. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Huh JE, Kang KS, Chae C, Kim HM, Ahn KS
and Kim SH: Roles of p38 and JNK mitogen-activated protein kinase
pathways during cantharidin-induced apoptosis in U937 cells.
Biochem Pharmacol. 67:1811–1818. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huan SK, Lee HH, Liu DZ, Wu CC and Wang
CC: Cantharidin-induced cytotoxicity and cyclooxygenase 2
expression in human bladder carcinoma cell line. Toxicology.
223:136–143. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Williams LA, Moller W, Merisor E, Kraus W
and Rosner H: In vitro anti-proliferation/cytotoxic activity of
cantharidin (Spanish Fly) and related derivatives. West Indian Med
J. 52:10–13. 2003.PubMed/NCBI
|
|
9
|
Xiong X, Wu M, Zhang H, Li J, Lu B, Guo Y,
Zhou T, Guo H, Peng R, Li X, et al: Atg5 siRNA inhibits autophagy
and enhances norcantharidin-induced apoptosis in hepatocellular
carcinoma. Int J Oncol. 47:1321–1328. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen YN, Chen JC, Yin SC, Wang GS, Tsauer
W, Hsu SF and Hsu SL: Effector mechanisms of norcantharidin-induced
mitotic arrest and apoptosis in human hepatoma cells. Int J Cancer.
100:158–165. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kok SH, Cheng SJ, Hong CY, Lee JJ, Lin SK,
Kuo YS, Chiang CP and Kuo MY: Norcantharidin-induced apoptosis in
oral cancer cells is associated with an increase of proapoptotic to
antiapoptotic protein ratio. Cancer Lett. 217:43–52. 2005.
View Article : Google Scholar
|
|
12
|
Su CC, Lee KI, Chen MK, Kuo CY, Tang CH
and Liu SH: Cantharidin induced oral squamous cell carcinoma cell
apoptosis via the JNK-regulated mitochondria and endoplasmic
reticulum stress-related signaling pathways. PLoS One.
11:e01680952016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yi SN, Wass J, Vincent P and Iland H:
Inhibitory effect of norcantharidin on K562 human myeloid leukemia
cells in vitro. Leuk Res. 15:883–886. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wiley SR, Schooley K, Smolak PJ, Din WS,
Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C, Smith CA,
et al: Identification and characterization of a new member of the
TNF family that induces apoptosis. Immunity. 3:673–682. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Srivastava RK: TRAIL/Apo-2L: Mechanisms
and clinical applications in cancer. Neoplasia. 3:535–546. 2001.
View Article : Google Scholar
|
|
16
|
Walczak H, Bouchon A, Stahl H and Krammer
PH: Tumor necrosis factor-related apoptosis-inducing ligand retains
its apoptosis-inducing capacity on Bcl-2- or Bcl-xL-overexpressing
chemotherapy-resistant tumor cells. Cancer Res. 60:3051–3057.
2000.PubMed/NCBI
|
|
17
|
Kelly MM, Hoel BD and Voelkel-Johnson C:
Doxorubicin pretreatment sensitizes prostate cancer cell lines to
TRAIL induced apoptosis which correlates with the loss of c-FLIP
expression. Cancer Biol Ther. 1:520–527. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ng CP, Zisman A and Bonavida B: Synergy is
achieved by complementation with Apo2L/TRAIL and actinomycin D in
Apo2L/TRAIL-mediated apoptosis of prostate cancer cells: Role of
XIAP in resistance. Prostate. 53:286–299. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang Y and Zhang B: TRAIL resistance of
breast cancer cells is associated with constitutive endocytosis of
death receptors 4-5. Mol Cancer Res. 6:1861–1871. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Levine B, Mizushima N and Virgin HW:
Autophagy in immunity and inflammation. Nature. 469:323–335. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kondo Y, Kanzawa T, Sawaya R and Kondo S:
The role of autophagy in cancer development and response to
therapy. Nat Rev Cancer. 5:726–734. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kondo Y and Kondo S: Autophagy and cancer
therapy. Autophagy. 2:85–90. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Moretti L, Yang ES, Kim KW and Lu B:
Autophagy signaling in cancer and its potential as novel target to
improve anticancer therapy. Drug Resist Updat. 10:135–143. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Eisenberg-Lerner A, Bialik S, Simon HU and
Kimchi A: Life and death partners: Apoptosis, autophagy and the
cross-talk between them. Cell Death Differ. 16:966–975. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nazim UM, Moon JH, Lee JH, Lee YJ, Seol
JW, Eo SK, Lee JH and Park SY: Activation of autophagy flux by
metformin downregulates cellular FLICE-like inhibitory protein and
enhances TRAIL- induced apoptosis. Oncotarget. 7:23468–23481. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nazim UM, Jeong JK and Park SY:
Ophiopogonin B sensitizes TRAIL-induced apoptosis through
activation of autophagy flux and downregulates cellular FLICE-like
inhibitory protein. Oncotarget. 9:4161–4172. 2017. View Article : Google Scholar
|
|
27
|
Elmallah MI and Micheau O: Marine drugs
regulating apoptosis induced by tumor necrosis factor-related
apoptosis-inducing ligand (TRAIL). Mar Drugs. 13:6884–6909. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wei RJ, Zhang XS and He DL:
Andrographolide sensitizes prostate cancer cells to TRAIL-induced
apoptosis. Asian J Androl. 20:200–204. 2018. View Article : Google Scholar :
|
|
29
|
Stuckey DW and Shah K: TRAIL on trial:
Preclinical advances in cancer therapy. Trends Mol Med. 19:685–694.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Honkanen RE: Cantharidin, another natural
toxin that inhibits the activity of serine/threonine protein
phosphatases types 1 and 2A. FEBS Lett. 330:283–286. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li HC, Xia ZH, Chen YF, Yang F, Feng W,
Cai H, Mei Y, Jiang YM, Xu K and Feng DX: Cantharidin inhibits the
growth of triple-negative breast cancer cells by suppressing
autophagy and inducing apoptosis in vitro and in vivo. Cell Physiol
Biochem. 43:1829–1840. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ren Y, Zhang SW, Xie ZH, Xu XM, Chen LL,
Lou ZG, Weng GB and Yao XP: Cantharidin induces G2/M arrest and
triggers apoptosis in renal cell carcinoma. Mol Med Rep.
14:5614–5618. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hsiao YP, Tsai CH, Wu PP, Hsu SC, Liu HC,
Huang YP, Yang JH and Chung JG: Cantharidin induces G2/M phase
arrest by inhibition of Cdc25c and Cyclin A and triggers apoptosis
through reactive oxygen species and the mitochondriadependent
pathways of A375.S2 human melanoma cells. Int J Oncol.
45:2393–2402. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Han Z, Li B, Wang J, Zhang X, Li Z, Dai L,
Cao M and Jiang J: Norcantharidin inhibits SK-N-SH neuroblastoma
cell growth by induction of autophagy and apoptosis. Technol Cancer
Res Treat. 16:33–44. 2017. View Article : Google Scholar :
|
|
35
|
Poornima P, Weng CF and Padma VV: Neferine
from Nelumbo nucifera induces autophagy through the inhibition of
PI3K/Akt/mTOR pathway and ROS hyper generation in A549 cells. Food
Chem. 141:3598–3605. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Klionsky DJ and Emr SD: Autophagy as a
regulated pathway of cellular degradation. Science. 290:1717–1721.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Park MH, Kim JH, Chung YH and Lee SH:
Bakuchiol sensitizes cancer cells to TRAIL through ROS- and
JNK-mediated upregulation of death receptors and downregulation of
survival proteins. Biochem Biophys Res Commun. 473:586–592. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu YJ, Lin YC, Lee JC, Kuo SC, Ho CT,
Huang LJ, Kuo DH and Way TD: CCT327 enhances TRAIL-induced
apoptosis through the induction of death receptors and
downregulation of cell survival proteins in TRAIL-resistant human
leukemia cells. Oncol Rep. 32:1257–1264. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sophonnithiprasert T, Nilwarangkoon S,
Nakamura Y and Watanapokasin R: Goniothalamin enhances
TRAIL-induced apoptosis in colorectal cancer cells through DR5
upregulation and cFLIP downregulation. Int J Oncol. 47:2188–2196.
2015. View Article : Google Scholar : PubMed/NCBI
|