Introduction
Hypertensive disorders emerge during pregnancy,
including gestational hypertension, pre-eclampsia (PE), eclampsia,
chronic hypertension complicated with PE, and chronic hypertension
(1). PE, which is mainly
characterized by maternal proteinuria and hypertension, is one of
the main causes of maternal and perinatal mortality (2,3).
The morbidity rate due to PE is increasing, although the etiology
and pathogenesis of PE are not fully understood; therefore,
clinical treatment strategies available to treat the disease are
currently limited (4). According
to the latest published articles, the proliferation, invasion and
migration of trophoblast cells into the uterus serve important
roles in human placenta formation and embryo implantation (5,6).
Decreased invasive ability, and increased apoptosis, of trophoblast
cells are two major causes of PE during the critical stage of
placental implantation (7). Once
certain changes in the regulatory factors have taken place,
trophoblast cells will greatly proliferate and develop into
choriocarcinoma, which causes damage to the tissues and blood
vessels surrounding the uterus, and distant metastasis at an early
stage. Therefore, regulating the proliferation, migration and
invasion of trophoblast cells has a positive significance for the
treatment of PE and related diseases.
Previous studies (8,9)
have highlighted that the oxidation of low-density lipoprotein
(LDL) leads to upregulation of the levels of inflammatory factors,
including tumor necrosis factor-α and interleukin-6, in the body,
and may decrease the cell invasive ability of trophoblasts and
promote cell apoptosis, thereby resulting in abnormal development
of placenta and the occurrence of PE. According to the National
Center for Biotechnology Information (NCBI) database, LDL
receptor-related protein 6 (LRP6) is highly expressed in placental
tissues. Previous studies have confirmed that LRP6 serves a
critical role in promoting the migration and invasion of numerous
types of cancers; for example, Liu et al (10) reported that LRP6 is upregulated in
breast cancer tissues, and that its overexpression is an important
factor contributing to cancer cell migration and invasion.
Therefore, it was possible to hypothesize that LRP6 may be involved
in regulating the migration and invasive abilities of trophoblast
cells.
The current study aimed to determine the expression
levels of LRP6 in PE tissues and trophoblast cell lines, to explore
the effects of upregulated and downregulated LRP6 on the
proliferation, migration and invasion of trophoblast cells, and to
further probe the relevant targets and mechanisms. Furthermore, the
mechanism of migration and invasion of trophoblast cells was also
explored so as to provide a probable therapeutic target for the
treatment of PE and other related diseases.
Materials and methods
Participants
A total of 40 patients diagnosed with PE (11) who attended the Qilu Hospital of
Shandong University from January to December 2018 were enrolled for
the present study. The patients were pregnant and primipara, aged
between 24 and 29 years old, and their gestational ages ranged from
34-39 weeks. Patients receiving reproductive technical assistance,
a history of repeated abortions and pregnancy complications (such
as hypertension, gestational diabetes and/or heart disease) were
excluded. As healthy controls, 22 corresponding normal pregnant
women were selected for comparison, whose characteristics were
similar to those of the patients with PE in terms of their
gestational age and body mass index. All participants underwent
cesarean sections. The study was approved by the Ethics Committee
of Qilu Hospital of Shandong University (grant approval number:
SDUQLH20180121), and written informed consent was obtained from all
participants.
Tissue acquisition
Tissue masses measuring 1×1×1 cm were collected
within 3 min after the delivery of placenta by Cesarean section
(avoiding bleeding, infarction and calcification areas). Extracted
tissues were dried using a dry gauze, frozen in liquid nitrogen,
and stored in a refrigerator at −70°C. All the above procedures
were performed under aseptic conditions. The relationship between
LRP6 and placental tissue was assessed using the NCBI database
(https://www.ncbi.nlm.nih.gov/gene/4040).
Cell culture
The trophoblast cell line B6Tert-1 was purchased
from the Institute of Zoology, State Key Laboratory of Reproductive
Biology, Chinese Academy of Sciences, and the trophoblast cell line
HTR8/SVneo was obtained from American Type Culture Collection
(ATCC). The gestational choriocarcinoma cell line JEG-3 (also
purchased from ATCC) served as a control. B6Tert-1 cells were
cultured in Invitrogen F12/Dulbecco's modified Eagle's medium
(DMEM; 1:1) containing 2 mmol/l glutamine, 10 mg/ml insulin, 10
ng/ml epidermal growth factor, and 0.1% bovine serum albumin
(Invitrogen; Thermo Fisher Scientific, Inc.). HTR8/SVneo and JEG-3
cells were maintained in Gibco Roswell Park Memorial Institute
(RPMI)-1640 medium (Thermo Fisher Scientific, Inc.) that contained
100 µg/ml streptomycin, 100 units/ml penicillin (Invitrogen;
Thermo Fisher Scientific, Inc.), and 10% HyClone™ fetal bovine
serum (GE Healthcare Life Sciences). All the cells were incubated
in a humidified environment at 37°C under an atmosphere of 5%
CO2, and the medium was changed daily.
Cell transfection
To investigate the effects of LRP6 on trophoblast
cells, the LRP6 sequence were synthesized by Guangzhou RiboBio Co.,
Ltd. and cloned into the pcDNA3.1 vector (cat. no. V79020; Thermo
Fisher Scientific, Inc.). siLRP6 (20 pmol; cat. no. #8650, Cell
Signaling Technology, Inc.) was transfected into JEG-3 cells
(3x105 cells), whereas the HTR8/SVneo cell line
(3x105 cells) was transfected with the LRP6
overexpression plasmid (20 pmol) using Invitrogen
Lipofectamine® 2000 transfection reagent (Thermo Fisher
Scientific, Inc.). For comparison, untransfected cells served as
blank controls, whereas JEG-3 cells transfected with scrambled
sequence served as a negative control (siNC; 20 pmol; cat. no.
1022076; Qiagen, Inc.). The siNC sequence was: Sense, 5′-UUC UCC
GAA CGU GUC ACG U-3′ and antisense, 5′-ACG UGA CAC GUU CGG AGA
A-3′. HTR8/SVneo cells were transfected with an empty pcDNA3.1
vector (20 pmol) as the mock group.
In order to explore the role of microRNA
(miRNA/miR)-346 in JEG-3 and HTR8/SVneo cells, 20 pmol mimics
control (cat. no. 4464059; Thermo Fisher Scientific, Inc.), miR-346
mimics (cat. no. 4464066; Thermo Fisher Scientific, Inc.),
inhibitor control (cat. no. 4464079; Thermo Fisher Scientific,
Inc.), and miR-346 inhibitor (cat. no. HLTUD0507; Sigma-Aldrich;
Merck KGaA) were respectively transfected into the two cell lines
using Lipofectamine® 2000 transfection reagent. JEG-3
cells (3×105 cells) were co-transfected with 20 pmol
siLRP6 and 20 pmol miR-346 inhibitor, whereas HTR8/SVneo cells
(3×105 cells) were co-transfected with 20 pmol LRP6 and
20 pmol miR-346 mimics. In addition, 20 pmol inhibitor control and
20 pmol siNC were co-transfected into JEG-3 cells (3×105
cells), and HTR8/SVneo cells (3×105 cells) were
co-transfected with mimics control and empty pcDNA3.1 vector
(mock), and served as negative controls for comparison. In the cell
transfection process, 2 µl Lipofectamine® 2000
reagent were mixed with 50 pmol RNA to generate liposome-enclosed
nucleotides.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) assay
Total RNA from tissues and cells was isolated using
Invitrogen reagent (Thermo Fisher Scientific, Inc.), and the
quality and integrity of the total RNAs were analyzed using a
NanoDrop™ 2000c spectrophotometer (Thermo Fisher Scientific, Inc.)
and 1% modified agarose gel electrophoresis. First-strand cDNA was
synthesized from the isolated RNA (1 µg) using PrimeScript
RT Master mix (Perfect Real-Time) (Takara Bio Inc.), according to
the manufacturer's protocol. The RT-qPCR assay was performed using
an ABI Prism 7500 Fast Real-time PCR system (Applied Biosystems;
Thermo Fisher Scientific, Inc.), and the thermocycling reaction
conditions were set as follows: 40 cycles at 50°C for 10 min and
95°C for 10 min, then 95°C for 15 sec and 45°C for 45 sec. The
corresponding mRNA expression levels were normalized against the
expression of β-actin or U6, and the data were processed using the
comparative 2-ΔΔCq method (12). The primers were synthesized by
Shanghai GenePharma Co., Ltd., and their sequences are shown in
Table I.
 | Table IPrimer sequences. |
Table I
Primer sequences.
| Gene | Forward
(5′-3′) | Reverse
(5′-3′) |
|---|
| LRP6 |
TTGTTGCTTTATGCAAACAGACGC |
GTTTAATGGCTTCTTCGCTGAC |
| miR-346 |
GATGCTATGTCTGCCCGCAT |
ATACCAACAAGTGCTGAGGAAGTG |
| MMP-2 |
GTGATCTTGACCAGAATACC |
GCCAATGATCCTGTATGTG |
| MMP-9 |
AAGGATGGGAAGTACTGG |
GCCCAGAGAAGAAGAAAG |
| TIMP-1 C |
ACCTTATACCAGCGTTATG |
TTTCCAGCAATGAGAAACTC |
| TIMP-2 |
GGCCTGATAAGGATATAGAG |
CTTTCCTGCAATGAGATATTCC |
| β-actin CC |
AACCGCGAGAAGATGA |
CCAGAGGCGTACAGGGATAG |
| U6 C |
TCGCTTCGGCAGCACA |
AACGCTTCACGAATTTGCGT |
Cell Counting Kit-8 (CCK-8) assay
Viability of the transfected cells was determined
using a CCK-8 kit (Beyotime Institute of Biotechnology). In brief,
after transfection had been allowed to proceed for 24 and 48 h, 10
µl CCK-8 reagents were added into the transfected cells and
co-cultured at 37°C under an atmosphere of 5% CO2 for 2
h. The optical density (OD) values of the cells were measured using
an ELX800 BioTek microplate reader (Biotek Instruments, Inc.) at
450 nm, and the OD values of the cells was measured at 24, 48, 72
and 96 h, respectively.
Wound-healing assay
The migration distances of the trans-fected cells
were measured by performing a wound-healing assay. For detection,
and prior to the assay, the transfected cells were cultured in
serum-free medium in 24-well plates to reach 90-100% confluence. A
200 µl sterile pipette tip was used to scratch a wound
through the cells; subsequently, the wounds were observed, and
images were captured, under an inverted microscope (Eclipse TS-100;
Nikon Corporation) at 0 and 24 h.
Transwell invasion assay
The invasive abilities of the transfected cells were
determined using Costar™ Transwell invasion chambers (Corning
Inc.). In the experiment, the upper chambers were pre-coated with
Matrigel™ (BD Biosciences), whereas the lower chambers contained
the medium with 10% FBS. The transfected cells
(3x105/ml) were suspended in serum-free medium, and
transferred into the upper chamber (200 µl/well). After
incubation for 24 h, non-invaded cells were removed by cotton
swabs, whereas the invaded cells were fixed in 4% paraformaldehyde
and stained with 0.1% crystal violet solution for 20 min. The
invaded cells were observed and counted under an inverted
microscope (Eclipse TS-100; Nikon Corporation).
Western blot (WB) analysis
WB analysis was performed to detect the expression
levels of proteins of interest in the cells. Total proteins were
extracted using RIPA lysis buffer (Beijing Solarbio Science &
Technology Co., Ltd.), and the concentrations of proteins were
determined using a bicinchoninic acid (BCA) protein assay (Pierce;
Thermo Fisher Scientific, Inc.). Total proteins (50 µg) were
separated by SDS-PAGE (Beyotime Institute of Biotechnology) using
10% gels, and then transferred onto a polyvinylidene difluoride
(PVDF) membrane (Merck Millipore). After blocking the membranes
with 5% non-fat milk for 1 h, the membranes were co-cultured with
primary antibodies [anti-LRP6 (1:2,000; cat. no. ab134146),
anti-MMP-2 (1:1,000; cat. no. ab37150), anti-MMP-9 (1:1,000; cat.
no. ab73734), anti-TIMP-1 (1:500; cat. no. ab61224), anti-TIMP-2
(1:500; cat. no. ab180630) all antibodies purchased from Abcam]
overnight at 4°C; β-actin (1:5,000; cat. no. ab8226; Abcam) was
used as the internal reference. Subsequently, homologous secondary
antibodies [rabbit anti-mouse IgG H&L (HRP), cat. no. ab3728,
1:7,000; and goat anti-rabbit IgG H&L (HRP), cat. no. ab6721,
1:7,000; both from Abcam] were added and incubated with the
membranes for 2 h at 37°C. The protein blots were developed using
ECL detection reagent (cat. no. 345818; Merck Millipore) and
scanned using a sensitive multifunctional imager (Amersham Imager
600; GE Healthcare).
Double-luciferase reporter analysis
LRP6 was shown to be the potential target for
miR-346 using TargetScan 7.2 (http://http://www.targetscan.org/) as the prediction
method, which was subsequently verified by double-luciferase
reporter analysis. For the assay experiment, either the wild-type
or the mutant sequence of the LRP6 3′-untranslated region (3′-UTR)
was inserted into the reporter plasmid (Promega Corporation) to
construct the corresponding carriers. miR-346 mimics and blank
control were respectively co-transfected into HTR8/SVneo and JEG-3
cells containing the constructed carriers with wild-type and mutant
LRP6 3′-UTR for 48 h using Lipofectamine™ 2000 transfection
reagent. The luciferase activity of the cells was determined by
using the Luciferase Assay systems of Promega, following the
manufacturer's protocol (Promega Corporation).
Statistical analysis
Statistical Package of the Social Sciences 20.0
software (SPSS, Inc.) was used for data analysis. The data are
shown as the mean ± standard deviation (SD). Comparisons between
groups were performed by Student's t-test or one-way analysis of
variance (ANOVA) followed by post hoc test (Tukey's). All
experiments were repeated in triplicate. P<0.05 was considered
to indicate a statistically significant value.
Results
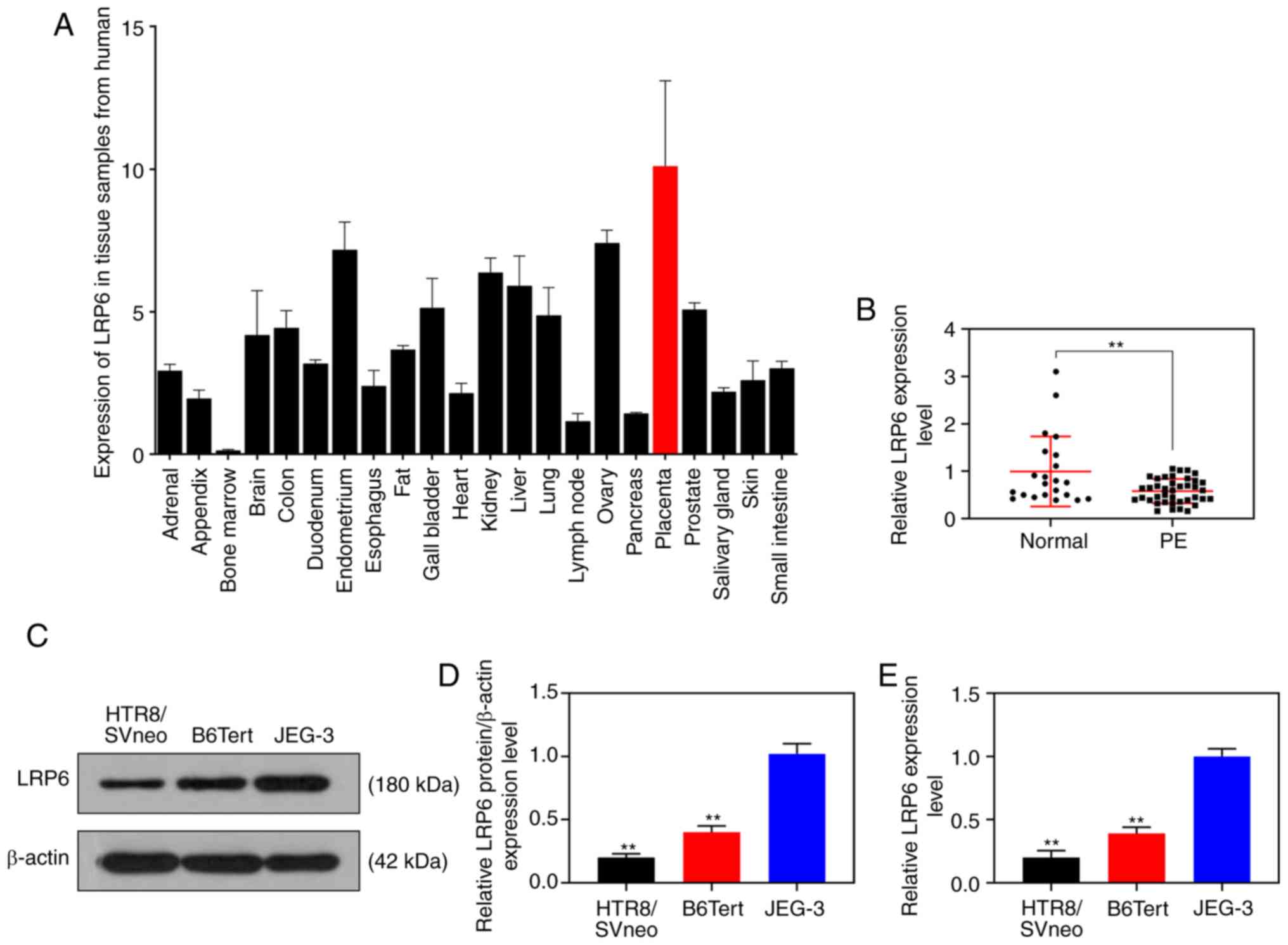
Expression of LRP6 is decreased in
trophoblast cells
The NCBI database (https://www.ncbi.nlm.nih.gov/gene/4040) revealed that
LRP6 was relatively highly expressed in placental tissues (Fig. 1A). In the current study, the
RT-qPCR results revealed that the expression level of LRP6 in
placental tissues derived from patients with PE was measurably
reduced compared with that in normal pregnant women (P<0.001;
Fig. 1B). As for the cell
experiments, compared with the JEG-3 gestational choriocarcinoma
cells, LRP6 was weakly expressed in the trophoblast cell lines,
B6Tert-1 and HTR8/SVneo (P<0.001; Fig. 1C-E).
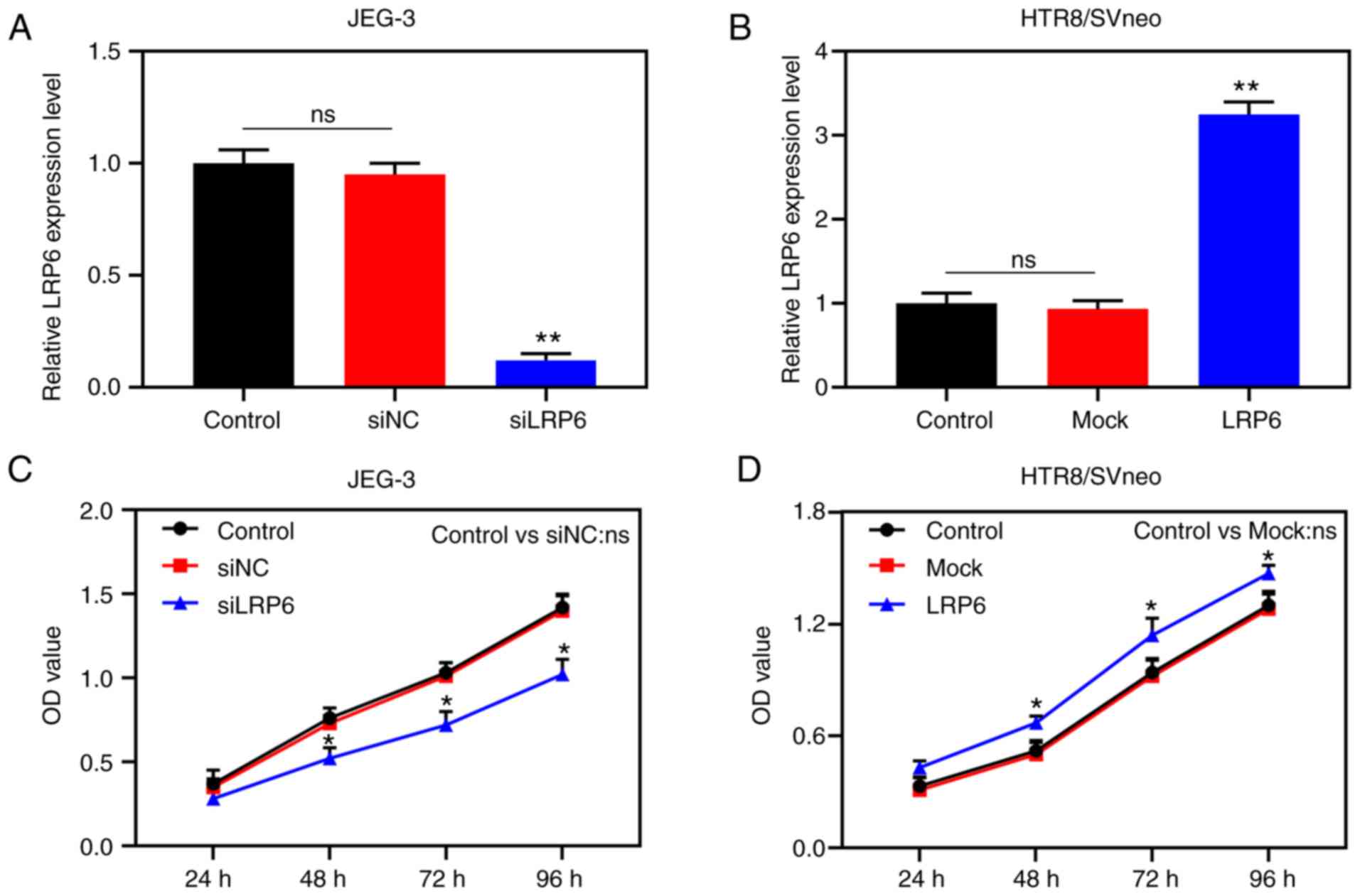
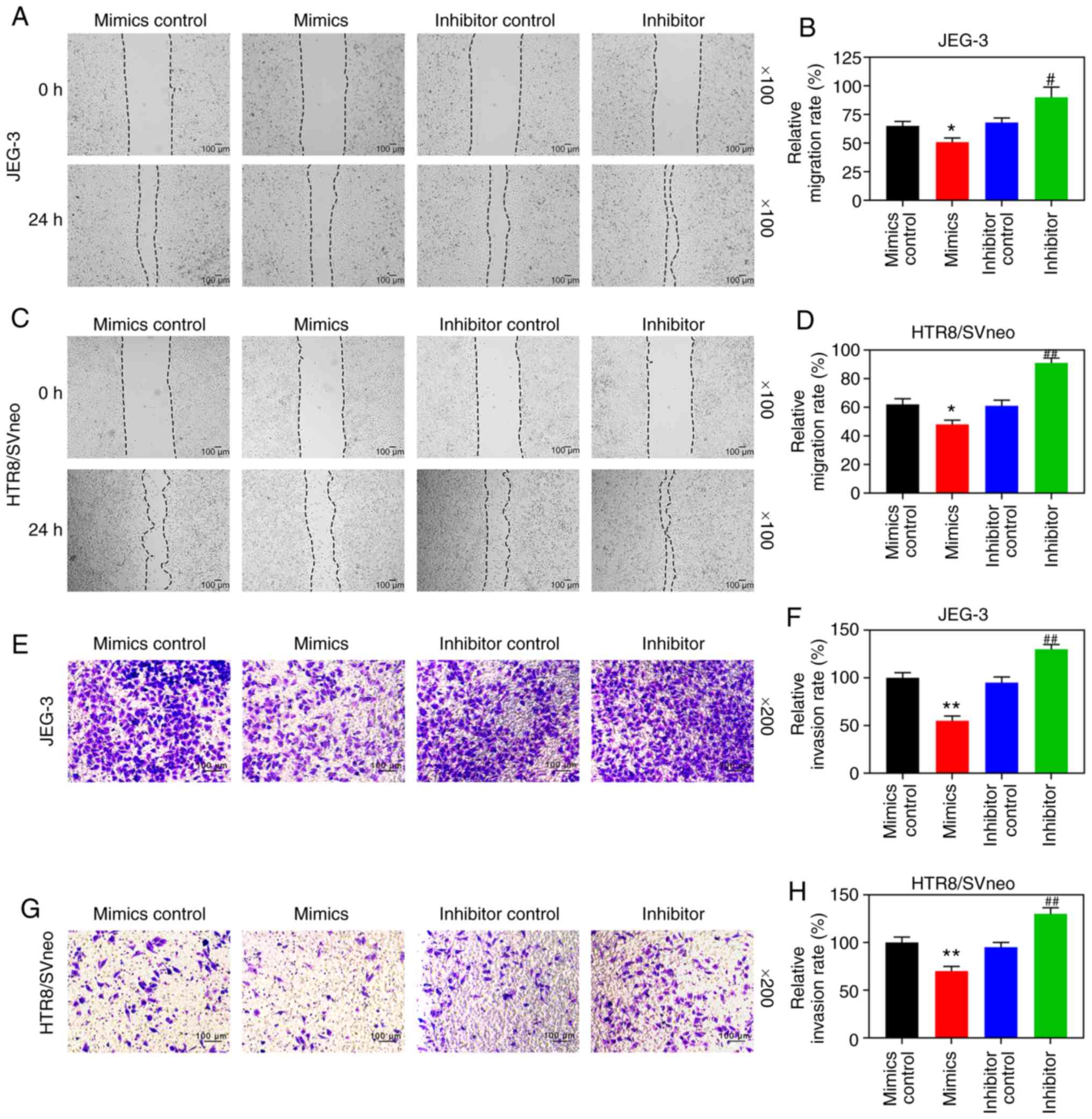
Overexpression of LRP6 promotes the
proliferation, migration and invasion of trophoblast cells
Transfection results demonstrated that LRP6
expression was successfully inhibited in JEG-3 cells, although it
was clearly upregulated in HTR8/SVneo cells (P<0.001; Fig. 2A and B), suggesting that the
transfection had been successfully conducted. In the subsequent
experiments, according to the CCK-8 assay, siLRP6 inhibited the
activity of JEG-3 cells, whereas overex-pressed LRP6 promoted the
cell activity of the HTR8/SVneo cell lines (P<0.05; Fig. 2C and D). Moreover, microscopic
images and quantitative analysis revealed that overexpressed LRP6
led to a measurable increase in the migratory distance and invasion
rate of the HTR8/SVneo cells, while the effects of poorly expressed
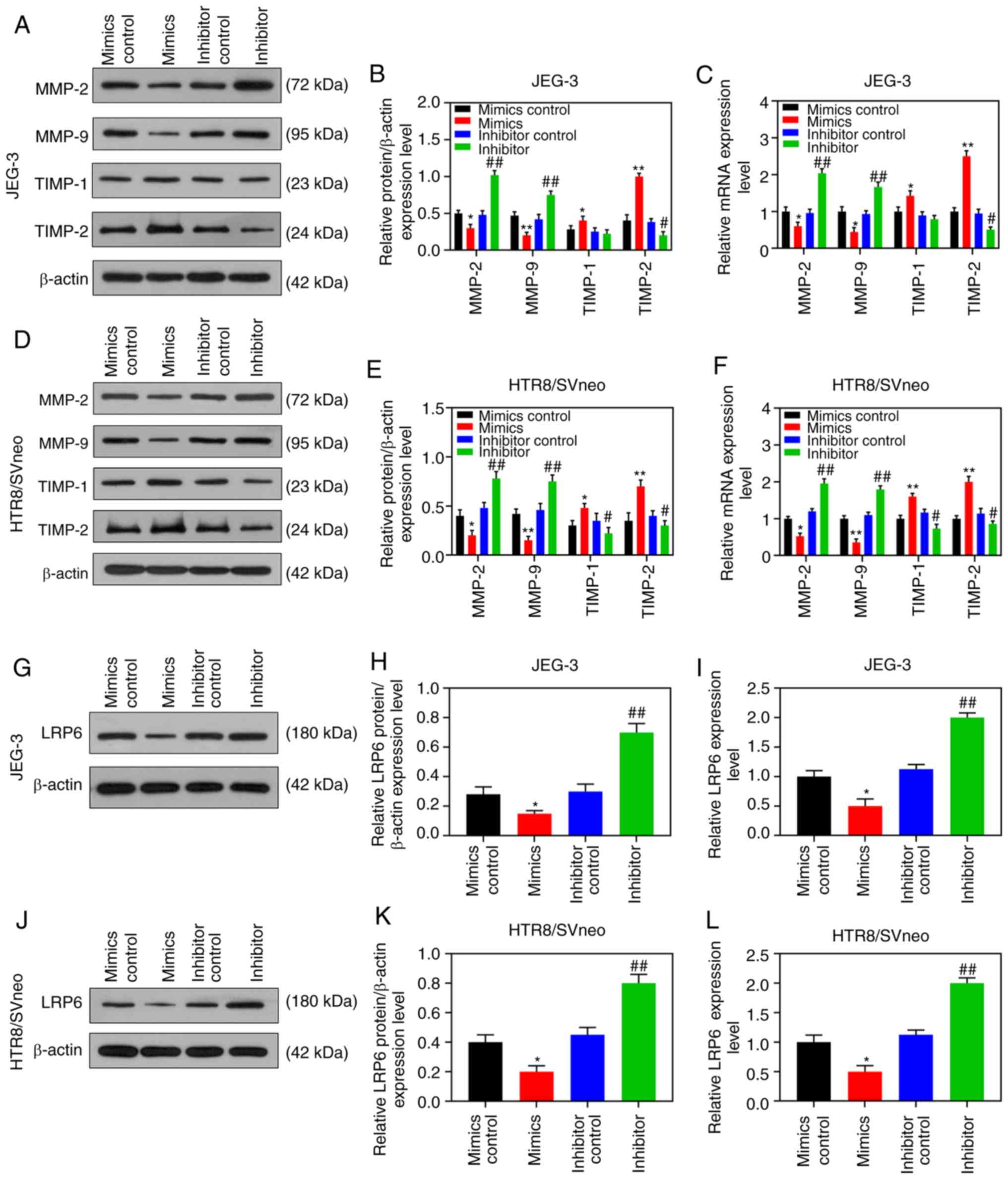
LRP6 on JEG-3 cells followed an opposite trend (P<0.05; Fig. 3A-D). Furthermore, the RT-qPCR and
WB analyses revealed that upregulating LRP6 led to promotion of the
expression levels of MMP-2 and MMP-9, although the expression
levels of TIMP-1 and TIMP-2 in HTR8/SVneo cells were suppressed.
Again, it was noted that the effects of siLRP6 on JEG-3 cells
followed an opposite trend compared with the effects of
overexpressed LRP6 (P<0.001; Fig.
3G-L).
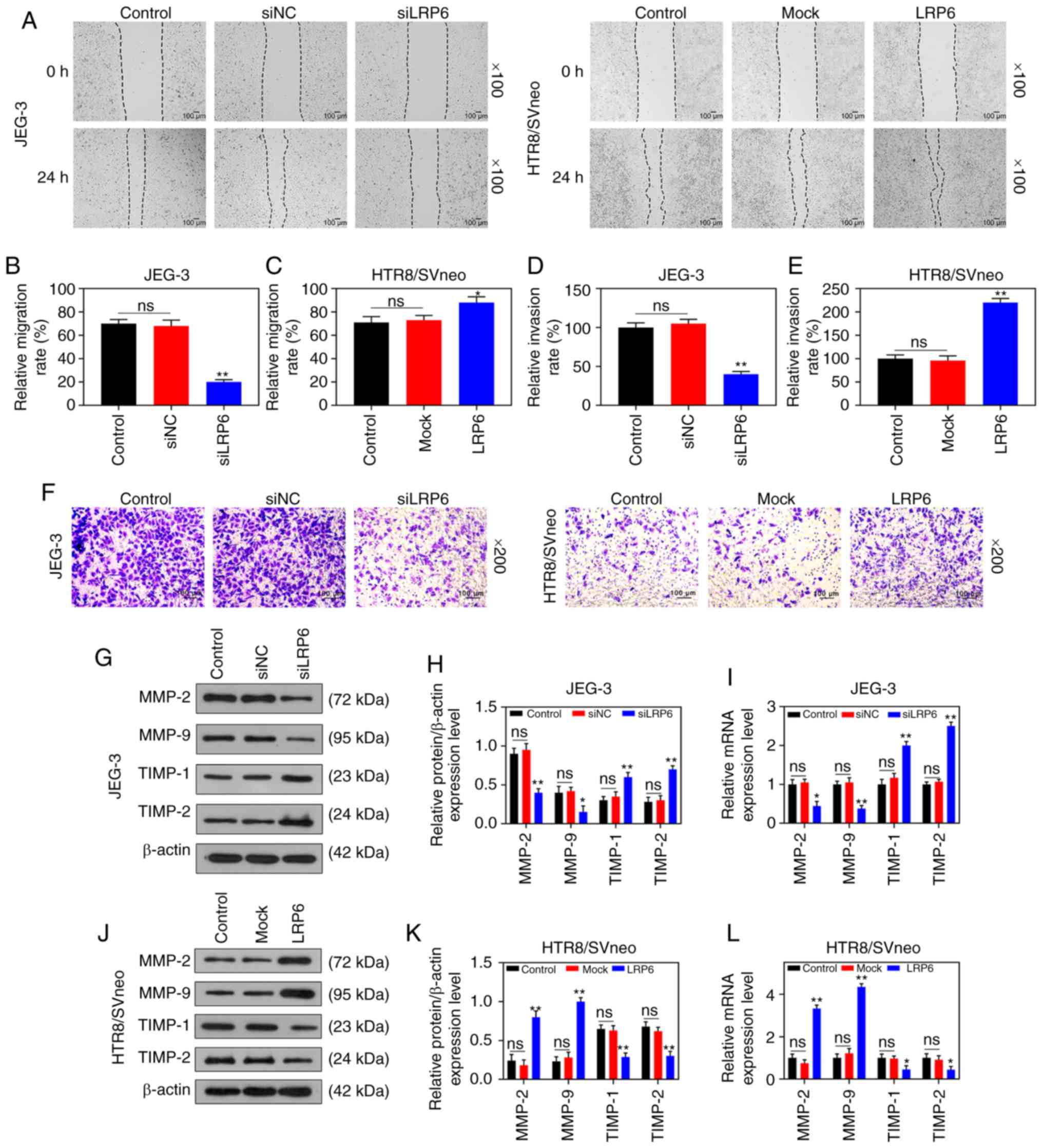 | Figure 3Overexpression of LRP6 enhances
migration and invasion of trophoblast cells. Negative control
(siNC) and siLRP6 were transfected into JEG-3 cells, whereas
HTR8/SVneo cells were transfected with LRP6 or empty pcDNA3.1
vector (Mock), and un-transfected cells served as a control for
comparison. (A-F) Microscopic images and quantitative analysis of
wound healing and Transwell assays in cells following transfection.
(G-L) Western blot analysis and RT-qPCR analyses were performed to
determine the expression levels of genes and proteins associated
with migration and invasion (i.e., MMP-2, MMP-9, TIMP-1, TIMP-2).
The magnification of the images (either x100 or ×200) is indicated
in the Figure parts, where applicable.**P<0.001,
*P<0.05 vs. siNC or Mock (n=3). ns, not significant;
LRP6, low-density-lipoprotein receptor-related protein 6; MMP,
matrix metalloproteinase; TIMP, tissue inhibitor of
metal-loproteinase. |
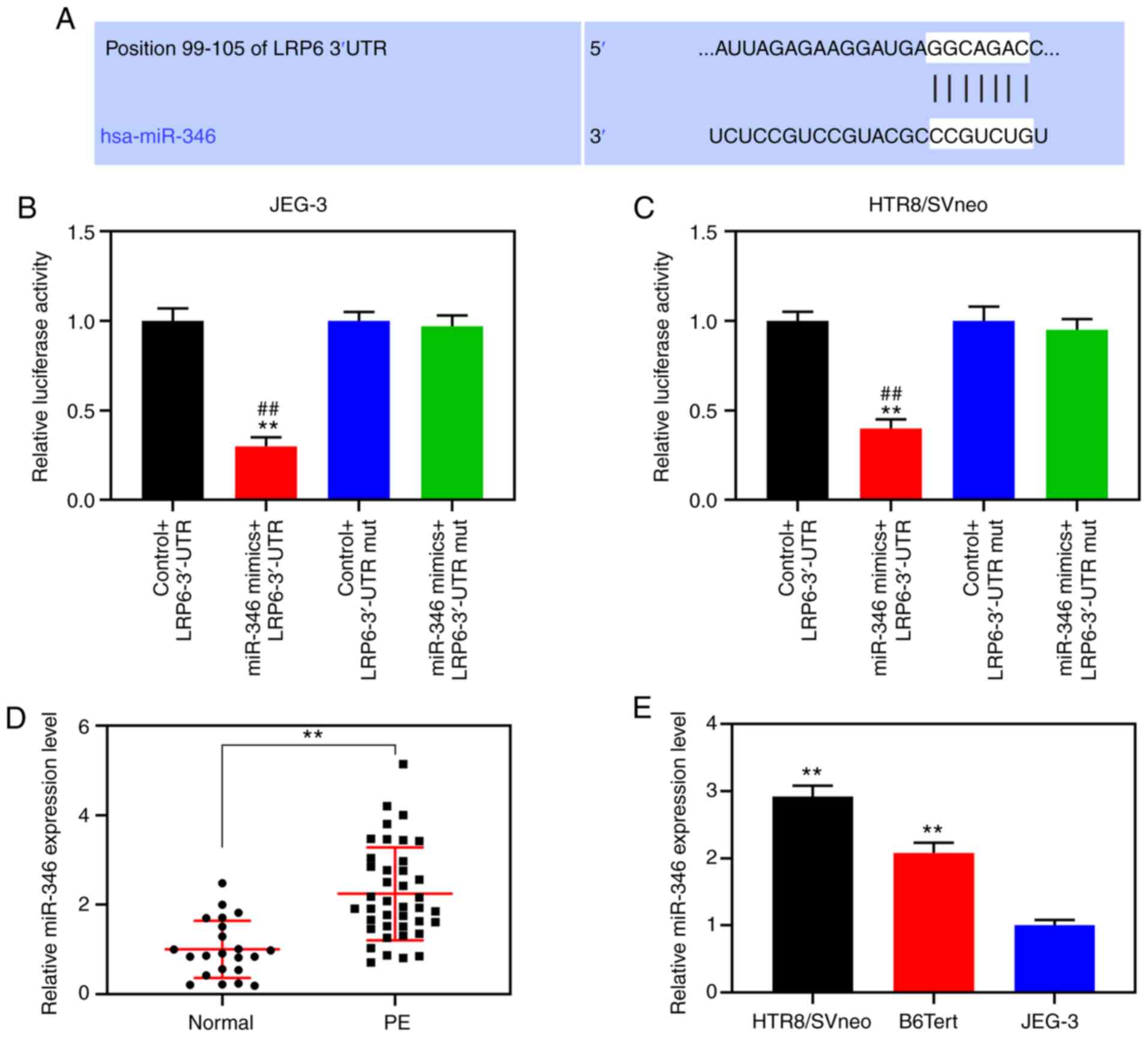
Downregulated miR-346 promotes the
proliferation, migration and invasion of trophoblast cells
According to the TargetScan prediction, nt positions
99-105 of the LRP6 3′-UTR are able to bind to miR-346 (Fig. 4A). For further verification,
double-luciferase reporter analysis was performed, and the data
demonstrated that luciferase activity was notably inhibited in
cells co-transfected with miR-346 and the LRP6 3′-UTR (P<0.001;
Fig. 4B and C). RT-qPCR was
subsequently performed to detect miR-346 expression in the placenta
of patients with PE and normal pregnant women and trophoblastic
cells, and the results showed that the expression level of miR-346
in placental tissues derived from the PE patients was measurably
increased compared with that from normal pregnant women
(P<0.001; Fig. 4D). Moreover,
compared with the JEG-3 gestational choriocarcinoma cells, miR-346
was highly expressed in the trophoblast cell lines, B6Tert-1 and
HTR8/SVneo (P<0.001; Fig. 4E).
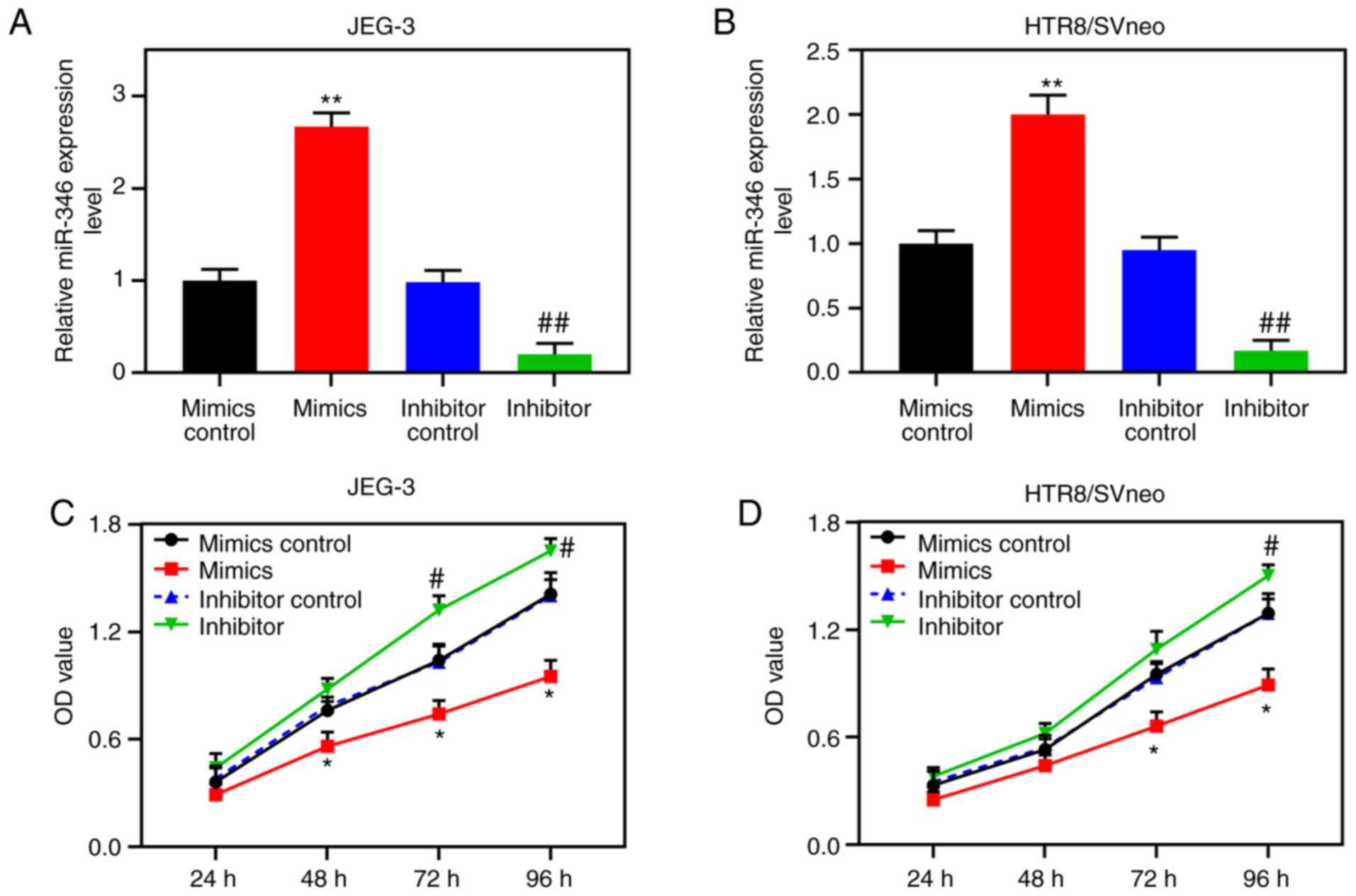
Following transfection, miR-346 was shown to be highly expressed in
JEG-3 cells, and poorly expressed in HTR8/SVneo cells (P<0.001;
Fig. 5A and B). CCK-8 assays
confirmed that upregulated miR-346 led to suppression of the
activity of the HTR8/SVneo cells, whereas poorly expressed miR-346
increased the activities of these cells (Fig. 5C and D).
Wound healing and Transwell experiments were
subsequently performed. The results revealed that upregulated
miR-346 reduced the cell migration distance and reduced invasion
rates of the JEG-3 and HTR8/SVneo cells, whereas downregulating
miR-346 resulted in increased rates of cell migration and invasion
(P<0.05; Fig. 6). Accordingly,
RT-qPCR and WB analysis revealed that upregulated miR-346 could
increase the expression levels of TIMP-1 and TIMP-2, although the
expression levels of MMP-2 and MMP-9 in the two cell lines were
decreased, and these results showed the opposite trend to those
performed with downregulated miR-346 (P<0.05; Fig. 7A-F).
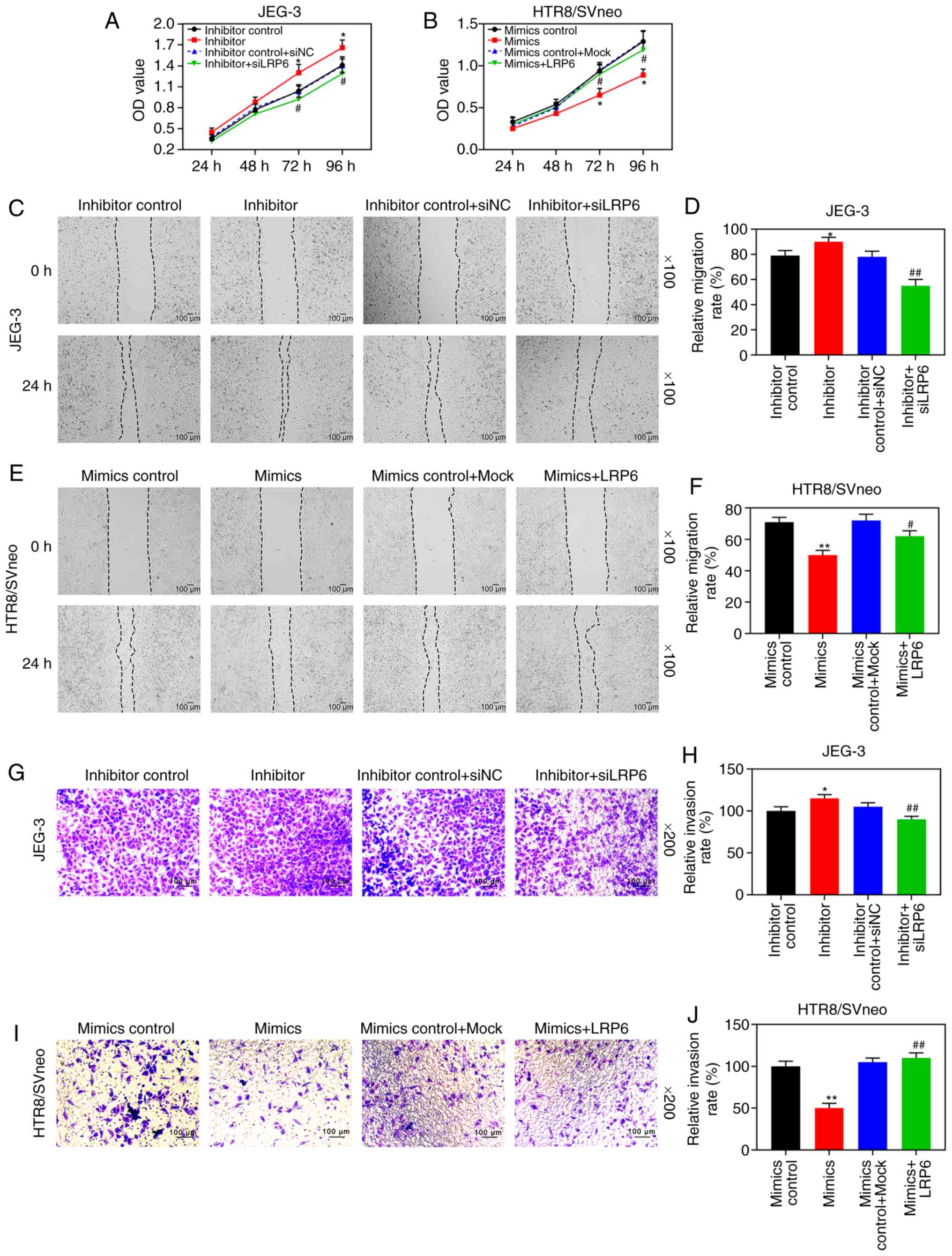
LRP6 reverses the inhibitory effect of
miR-346 on the proliferation, migration and invasion of trophoblast
cells
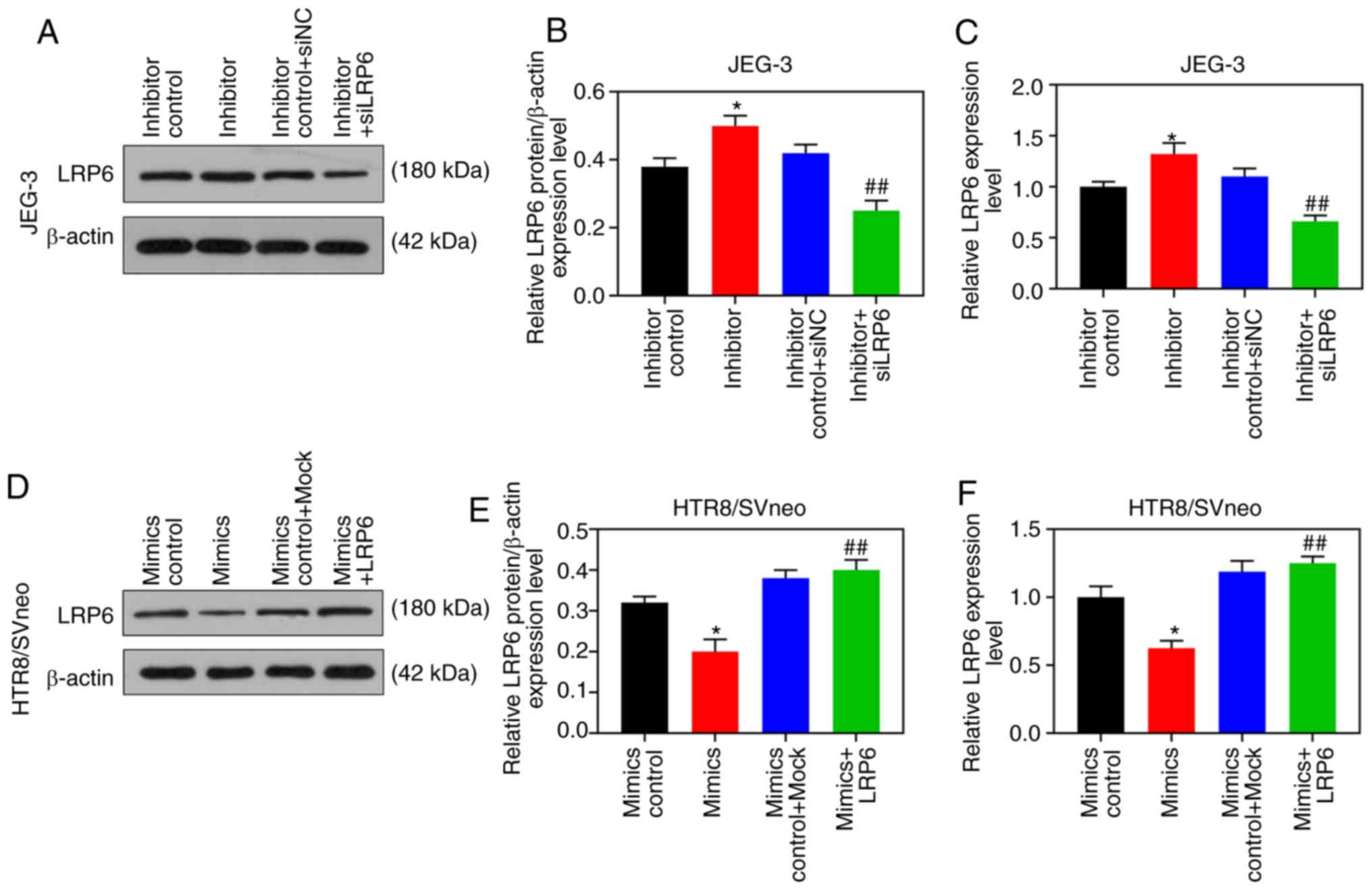
According to the results of RT-qPCR and WB analysis,
the expression level of LRP6 was gradually downregulated by the
upregulation of miR-346, but it was markedly upregulated by
downregulating miR-346 (P<0.05; Fig. 7G-L). After the co-transfection,
CCK-8 assays revealed that siLRP6 could reverse the effects of
poorly expressed miR-346 on JEG-3 cell activity, whereas LRP6 could
reverse the inhibitory effect of overexpressed miR-346 on
HTR8/SVneo cell activity (Fig. 8A and
B). Furthermore, the experimental images and quantitative
analysis from wound healing and Transwell experiments indicated
that siLRP6 suppressed the effects of poorly expressed miR-346 on
cell migration and invasion, whereas LRP6 overexpression relieved
the inhibitory effects of overexpressed miR-346 on cell migration
and invasion (P<0.05; Fig.
8C-J). In addition, the expression levels of LRP6 were
significantly downregulated in HTR8/SVneo cells that were
transfected with miR-346 mimics (P<0.05; Fig. 9).
Discussion
Embryo implantation, which is one of the decisive
factors for a successful pregnancy, refers to the process of the
embryo gradually burying itself in the endometrial epithelium after
entering the uterine cavity, and the invasion of trophoblast cells
has a crucially important role during the process (13). Trophoblast cells are the main cell
type in placental tissues, and abnormal migration and invasion
abilities of the cells leads to the occurrence of PE, gestational
choriocarcinoma and/or other related diseases (14,15). In the present study, it has been
shown that LRP6 was suppressed in PE tissues and different
trophoblast cell lines, although it was expressed at relatively
high levels in gestational choriocarcinoma cells. In a previously
published study, Wang et al (16) demonstrated that downregulated LRP6
could inhibit the growth, invasion and migration of retinoblastoma
cells, findings that were similar to the results in the current
study. Oppositely to the effect of downregulated LRP6, the present
study has also demonstrated that overexpressing LRP6 can measurably
promote cell activity and enhance the migration distance and the
invasion rate of trophoblast cells, suggesting that upregulated
LRP6 may slow down the progression of PE. As for the role of LRP6
in other diseases, Mao et al (17) demonstrated that LRP6 was
upregulated in human triple negative breast cancer cell lines, and
the metastatic ability of the cancer cells was increased via the
Wnt/β-catenin protein signaling pathway. LRP6 was also found to
promote metastasis and invasion of colorectal cancer through
cytoskeleton dynamics (18).
These findings further confirmed the role exerted by LRP6
overexpression in terms of promoting cell proliferation, migration
and invasion, and therefore LRP6 may be a potential therapeutic
target for treating trophoblast-cell-associated diseases.
Degradation of the extracellular matrix by members
of the MMP family is a critical process in cell migration and
invasion (19). TIMPs are a type
of tissue inhibitor of metal-loproteinases with multiple
physiological functions, and they are able to bind with MMPs
noncovalently to maintain the activity of MMPs, and to participate
in the maintenance and stability of extracellular matrix components
(20). On the one hand, MMP-2 and
MMP-9 degrade the extracellular matrix, allowing trophoblast cells
to continuously invade the endometrium with the gradual formation
of chorion, which eventually allows trophoblast cells to penetrate
the decidua and blood vessels during placental formation (21,22). On the other hand, specific binding
of TIMP-1 to MMP-9, and of TIMP-2 to MMP-2, inhibits the invasion
of MMPs, so that the invasive behavior and proliferation of
trophoblast cells can be properly controlled, thereby leading to
the maintenance of moderate levels of cell invasion (23). The present study has shown that
overexpression of LRP6 not only promoted the expression of MMP-2
and MMP-9, but also inhibited the secretion of TIMP-1 and TIMP-2,
in trophoblast cells, whereas transfection with siLRP6 led to
entirely the opposite effects on gestational choriocarcinoma cells.
These results suggest that the upregulation of LRP6 may control
cell migration and invasion by regulating the expression levels of
MMPs and TIMPs, thereby alleviating the conditions of PE and
gestational choriocarcinoma.
miRNAs have been demonstrated to be involved in
embryo implantation through regulating target genes (24,25). miRNAs control the expression of
target genes, and 60% of them are predicted to be regulators of
protein-encoding genes in humans (26). Our study demonstrated that miR-346
is able to target and negatively regulate the expression of LRP6,
suggesting that upregulated miR-346 inhibited LRP6 expression.
Moreover, miR-346 was highly expressed in tissues and trophoblast
cell lines; the gestational choriocarcinoma cell line JEG-3 served
as a control. LRP6 is known to promote metastasis in numerous types
of cancer; however, to the best of our knowledge, there are no
reports of its role in trophoblasts. Therefore, JEG-3 cells were
selected as a control group to study the effect of LRP6 on the
normal trophoblast cell line B6Tert-1. In addition, the current
study has demonstrated that overexpressed miR-346 is able to
suppress the proliferation, migration and invasion of tropho-blast
cells and JEG-3 cell, a finding that was reversed by LRP6.
Similarly, the downregulation of miR-346 led to an activation of
the proliferation, migration and invasion of gestational
choriocarcinoma cells, and this phenomenon was also be reversed by
siLRP6 treatment. miR-346 is known to have a tumor-promoter
function in a variety of tumor cells, including in liver cancer
(27), nasopharyngeal cancer
(28), and breast cancer
(29). However, contrary to
previous findings, the present study revealed that downregulated
miR-346 promoted the proliferation, migration and invasion of
trophoblast cells. It was speculated that miR-346 might have
cell-specific effects; therefore, miR-346 may have different
effects on various cell types. In addition, miR-346 inhibited
migration and invasion of HTR8/SVneo cells. Therefore, it is
possible to conclude that miR-346 was involved in the
proliferation, migration and invasion of the trophoblast cells,
suggesting that miR-346 may also have a critical role in PE and
gestational choriocarcinoma.
Furthermore, Su et al (30) reported that miR-346 regulated
endocrine gland-vascular endothelial growth factor
(EG-VEGF)-induced trophoblast invasion by inhibiting MMP-2 and
MMP-9, although it had only a limited effect on TIMP-1 and TIMP-2.
Consistently in the present study, it was found that the effect of
downregulated miR-346 on invasion-associated enzymes was similar to
that of upregulated LRP6, which was found to promote the expression
of MMP-2 and MMP-9 and to inhibit the expression of TIMP-1 and
TIMP-2. Taken together, these results suggested that the effects of
miR-346 on cell migration and invasion are mainly realized by
regulating the expression of MMPs and inhibiting TIMPs.
In conclusion, the present study has demonstrated
that LRP6 is downregulated in patients with PE, whereas the
upregulation of LRP6 promoted the proliferation and migration of
trophoblast cells, which is opposite to the effect of downregulated
LRP6 in gestational choriocarcinoma cells. Moreover, LRP6 has been
shown to be involved in the occurrence of PE and gestational
choriocarcinoma through regulating the activity, migratory and
invasive abilities of cancer cells through miR-346, suggesting that
LRP6 might be a possible therapeutic target for the treatment of PE
and gestational choriocarcinoma, and that a putative therapy
involving LRP6 treatment may decelerate the disease process.
However, the effect of miR-346 on the invasion and proliferation of
trophoblast cells will be further confirmed in our future studies.
In addition, the expression of LRP6 in tissues of patients with
choriocarcinoma needs to be investigated further. Furthermore,
in vivo animal experiments should also help in subsequent
investigations of the expression levels of LRP6, and its effects,
in animals.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LZ made the most substantial contributions to the
paper in terms of its conception and design of the experiments; HL,
ML, WZ, ZY, and SZ were involved with data acquisition, data
analysis and interpretation; and LZ was responsible for drafting
the article and critically revising it for important intellectual
content. All authors have approved the final approval of the
version, and agree to be held accountable for all aspects of the
work in ensuring that questions related to the accuracy or
integrity of the work are appropriately investigated and
resolved.
Ethics approval and consent to
participate
All procedures performed in studies involving human
participants were in accordance with the ethical standards of the
institutional and/or national research committee, and with the 1964
Helsinki declaration and its later amendments or comparable ethical
standards. Specifically, the present study was approved by the
Ethics Committee of Qilu Hospital of Shandong University (grant
approval number: SDUQLH20180121), and the informed consent from all
participants was obtained.
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no conflicts of
interests.
Acknowledgments
Not applicable.
References
|
1
|
Turner K and Hameed AB: Hypertensive
disorders in pregnancy current practice review. Curr Hypertens Rev.
13:80–88. 2017.PubMed/NCBI
|
|
2
|
Mol BWJ, Roberts CT, Thangaratinam S,
Magee LA, de Groot CJM and Hofmeyr GJ: Pre-eclampsia. Lancet.
387:999–1011. 2016. View Article : Google Scholar
|
|
3
|
von Dadelszen P and Magee LA:
Pre-eclampsia: An update. Curr Hypertens Rep. 16:4542014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zakiyah N, Postma MJ, Baker PN and van
Asselt AD; IMPROvED Consortium: Pre-eclampsia diagnosis and
treatment options: A review of published economic assessments.
Pharmacoeconomics. 33:1069–1082. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Robins JC: Implantation:
Trophoblast-endometrial interactions. Semin Reprod Med. 34:3–4.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gupta SK, Malhotra SS, Malik A, Verma S
and Chaudhary P: Cell signaling pathways involved during invasion
and syncytialization of trophoblast cells. Am J Reprod Immunol.
75:361–371. 2016. View Article : Google Scholar
|
|
7
|
McNally R, Alqudah A, Obradovic D and
McClements L: Elucidating the pathogenesis of pre-eclampsia using
in vitro models of spiral uterine artery remodelling. Curr
Hypertens Rep. 19:932017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang Y, Hu X, Gao G, Wang Y, Chen P and
Ye Y: Autophagy protects against oxidized low density
lipoprotein-mediated inflammation associated with preeclampsia.
Placenta. 48:136–143. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Arifin R, Kyi WM, Che Yaakob CA and Yaacob
NM: Increased circulating oxidised low-density lipoprotein and
antibodies to oxidised low-density lipoprotein in preeclampsia. J
Obstet Gynaecol. 37:580–584. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu CC, Prior J, Piwnica-Worms D and Bu G:
LRP6 overexpression defines a class of breast cancer subtype and is
a target for therapy. Proc Natl Acad Sci USA. 107:5136–5141. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ramos JGL, Sass N and Costa SHM:
Preeclampsia. Rev Bras Ginecol Obstet. 39:496–512. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rao X, Huang X, Zhou Z and Lin X: An
improvement of the 2^(-delta delta CT) method for quantitative
real-time polymerase chain reaction data analysis. Biostat
Bioinforma Biomath. 3:71–85. 2013.PubMed/NCBI
|
|
13
|
Godbole G, Suman P, Malik A, Galvankar M,
Joshi N, Fazleabas A, Gupta SK and Modi D: Decrease in expression
of HOXA10 in the decidua after embryo implantation promotes
trophoblast invasion. Endocrinology. 158:2618–2633. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li P, Guo W, Du L, Zhao J, Wang Y, Liu L,
Hu Y and Hou Y: microRNA-29b contributes to pre-eclampsia through
its effects on apoptosis, invasion and angiogenesis of trophoblast
cells. Clin Sci (Lond). 124:27–40. 2013. View Article : Google Scholar
|
|
15
|
Chen H, Meng T, Liu X, Sun M, Tong C, Liu
J, Wang H and Du J: Long non-coding RNA MALAT-1 is downregulated in
preeclampsia and regulates proliferation, apoptosis, migration and
invasion of JEG-3 trophoblast cells. Int J Clin Exp Pathol.
8:12718–12727. 2015.
|
|
16
|
Wang J, Wang X, Li Z, Liu H and Teng Y:
MicroRNA-183 suppresses retinoblastoma cell growth, invasion and
migration by targeting LRP6. FEBS J. 281:1355–1365. 2014.
View Article : Google Scholar
|
|
17
|
Mao Z, Li H, Du B, Cui K, Xing Y, Zhao X
and Zai S: LncRNA DANCR promotes migration and invasion through
suppression of lncRNA-LET in gastric cancer cells. Biosci Rep.
37:BSR201710702017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yao Q, An Y, Hou W, Cao YN, Yao MF, Ma NN,
Hou L, Zhang H, Liu HJ and Zhang B: LRP6 promotes invasion and
metastasis of colorectal cancer through cytoskeleton dynamics.
Oncotarget. 8:109632–109645. 2017. View Article : Google Scholar
|
|
19
|
Zhong T, Chen J, Ling Y, Yang B, Xie X, Yu
D, Zhang D, Ouyang J and Kuang H: Down-regulation of neuropathy
target esterase in preeclampsia placenta inhibits human trophoblast
cell invasion via modulating MMP-9 levels. Cell Physiol Biochem.
45:1013–1022. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Arpino V, Brock M and Gill SE: The role of
TIMPs in regulation of extracellular matrix proteolysis. Matrix
Biol. 44-46:247–254. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zong L, Wei X, Gou W, Huang P and Lv Y:
Zinc improves learning and memory abilities of fetal growth
restriction rats and promotes trophoblast cell invasion and
migration via enhancing STAT3-MMP-2/9 axis activity. Oncotarget.
8:115190–115201. 2017. View Article : Google Scholar
|
|
22
|
Yu Y, Wang L, Liu T and Guan H:
MicroRNA-204 suppresses trophoblast-like cell invasion by targeting
matrix metallopro-teinase-9. Biochem Biophys Res Commun.
463:285–291. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nissi R, Talvensaari-Mattila A, Kotila V,
Niinimaki M, Jarvela I and Turpeenniemi-Hujanen T: Circulating
matrix metalloproteinase MMP-9 and MMP-2/TIMP-2 complex are
associated with spontaneous early pregnancy failure. Reprod Biol
Endocrinol. 11:22013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zendjabil M, Favard S, Tse C, Abbou O and
Hainque B: The microRNAs as biomarkers: What prospects? C R Biol.
340:114–131. 2017.In French. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liang J, Wang S and Wang Z: Role of
microRNAs in embryo implantation. Reprod Biol Endocrinol.
15:902017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tufekci KU, Oner MG, Meuwissen RL and Genc
S: The role of microRNAs in human diseases. Methods Mol Biol.
1107:33–50. 2014. View Article : Google Scholar
|
|
27
|
Yu Q, Yang X, Duan W, Li C, Luo Y and Lu
S: miRNA-346 promotes proliferation, migration and invasion in
liver cancer. Oncol Lett. 14:3255–3260. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yan HL, Li L, Li SJ, Zhang HS and Xu W:
miR-346 promotes migration and invasion of nasopharyngeal carcinoma
cells via targeting BRMS1. J Biochem Mol Toxicol. 30:602–607. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang F, Luo LJ, Zhang L, Wang DD, Yang SJ,
Ding L, Li J, Chen D, Ma R, Wu JZ and Tang JH: MiR-346 promotes the
biological function of breast cancer cells by targeting SRCIN1 and
reduces chemosensitivity to docetaxel. Gene. 600:21–28. 2017.
View Article : Google Scholar
|
|
30
|
Su MT, Tsai PY, Tsai HL, Chen YC and Kuo
PL: miR-346 and miR-582-3p-regulated EG-VEGF expression and
trophoblast invasion via matrix metalloproteinases 2-9. Biofactors.
43:210–219. 2017. View Article : Google Scholar
|