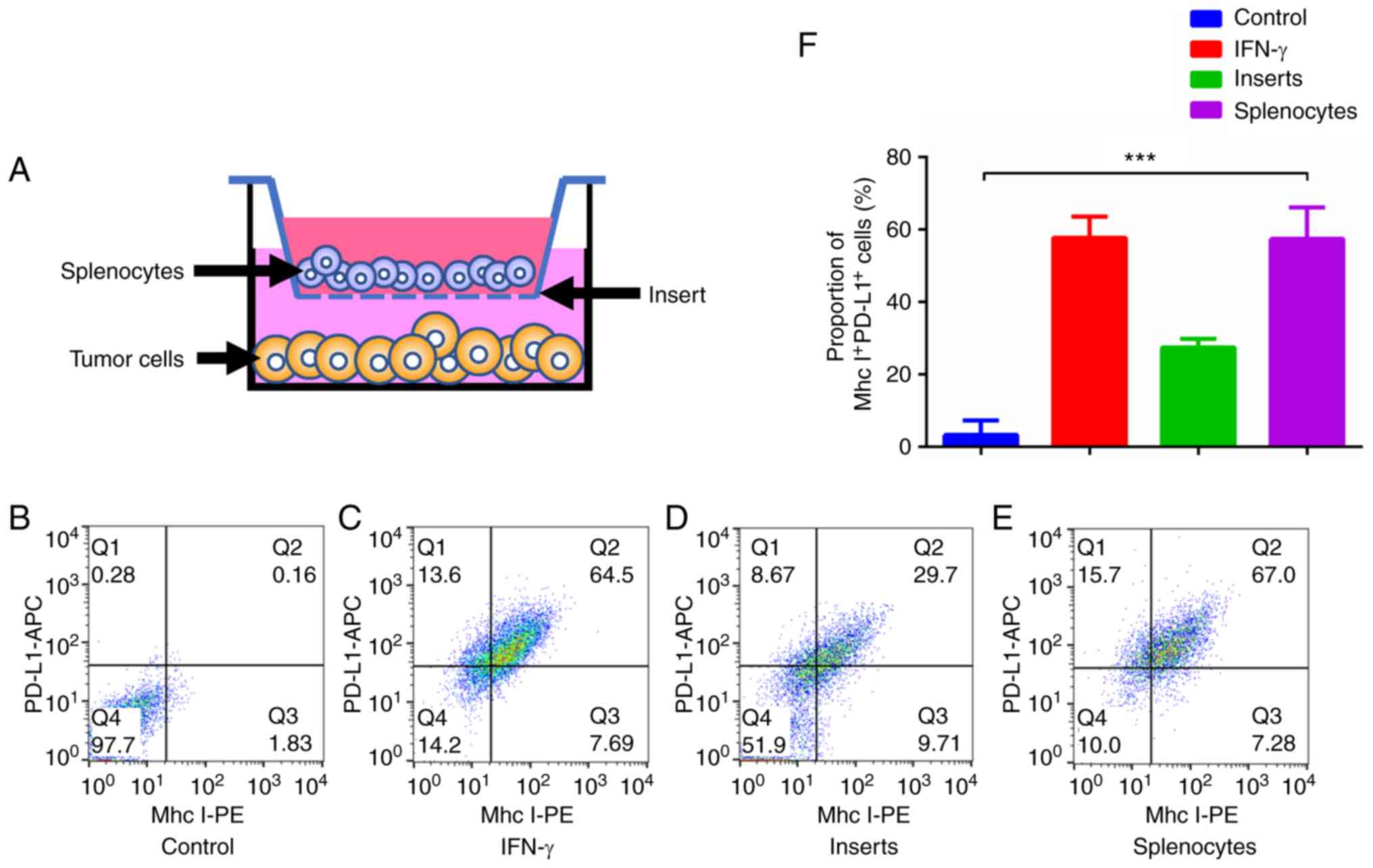
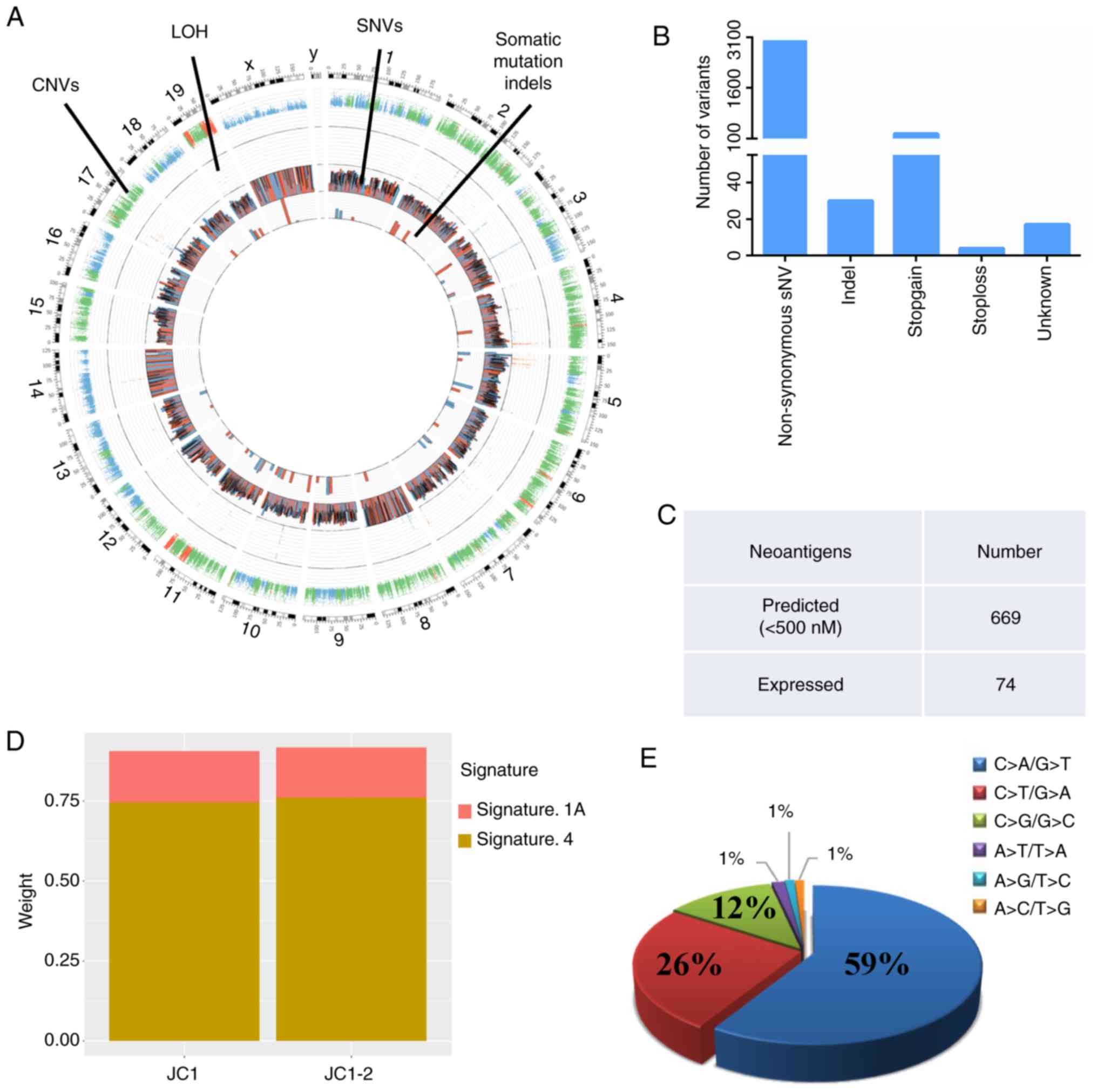
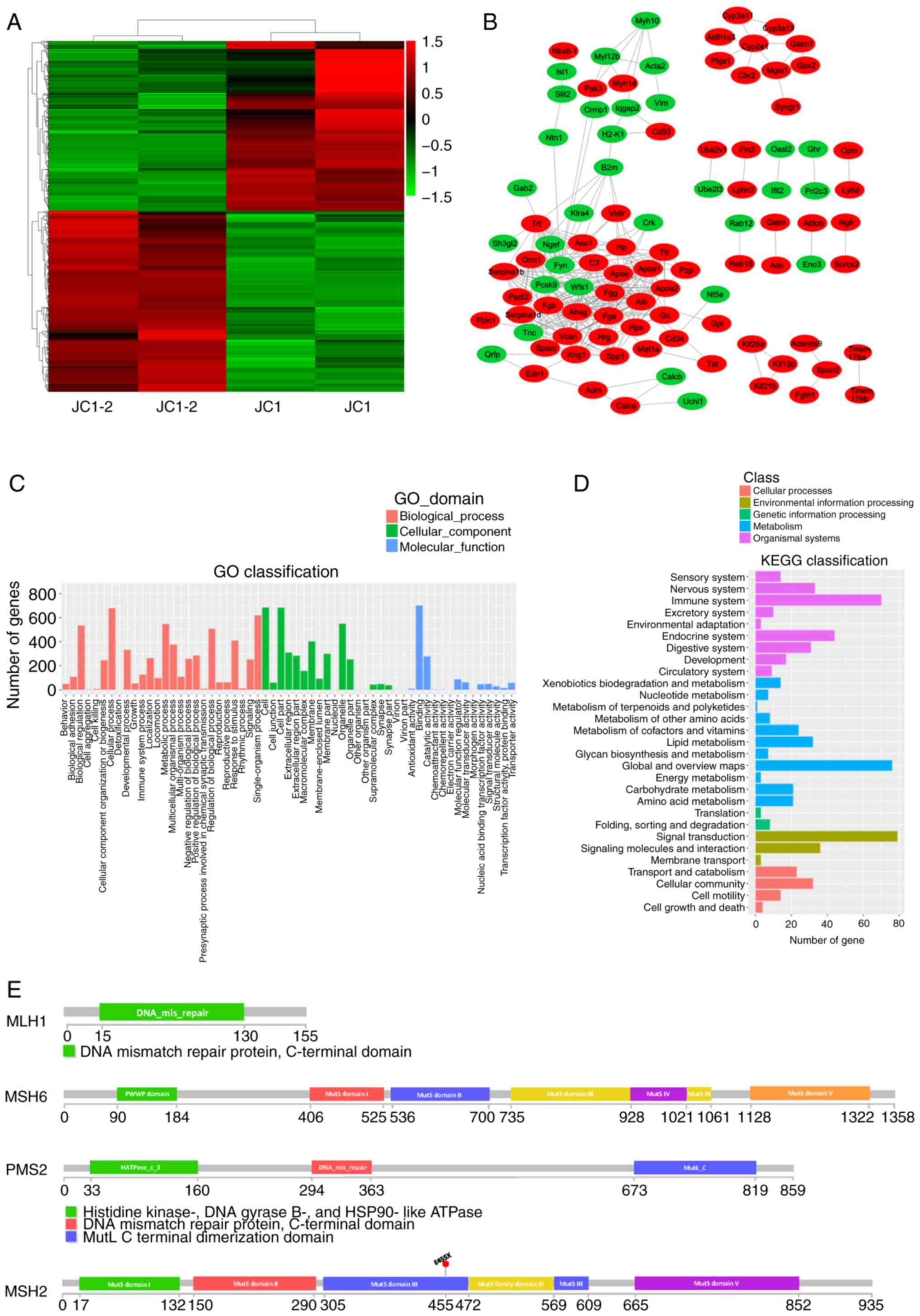
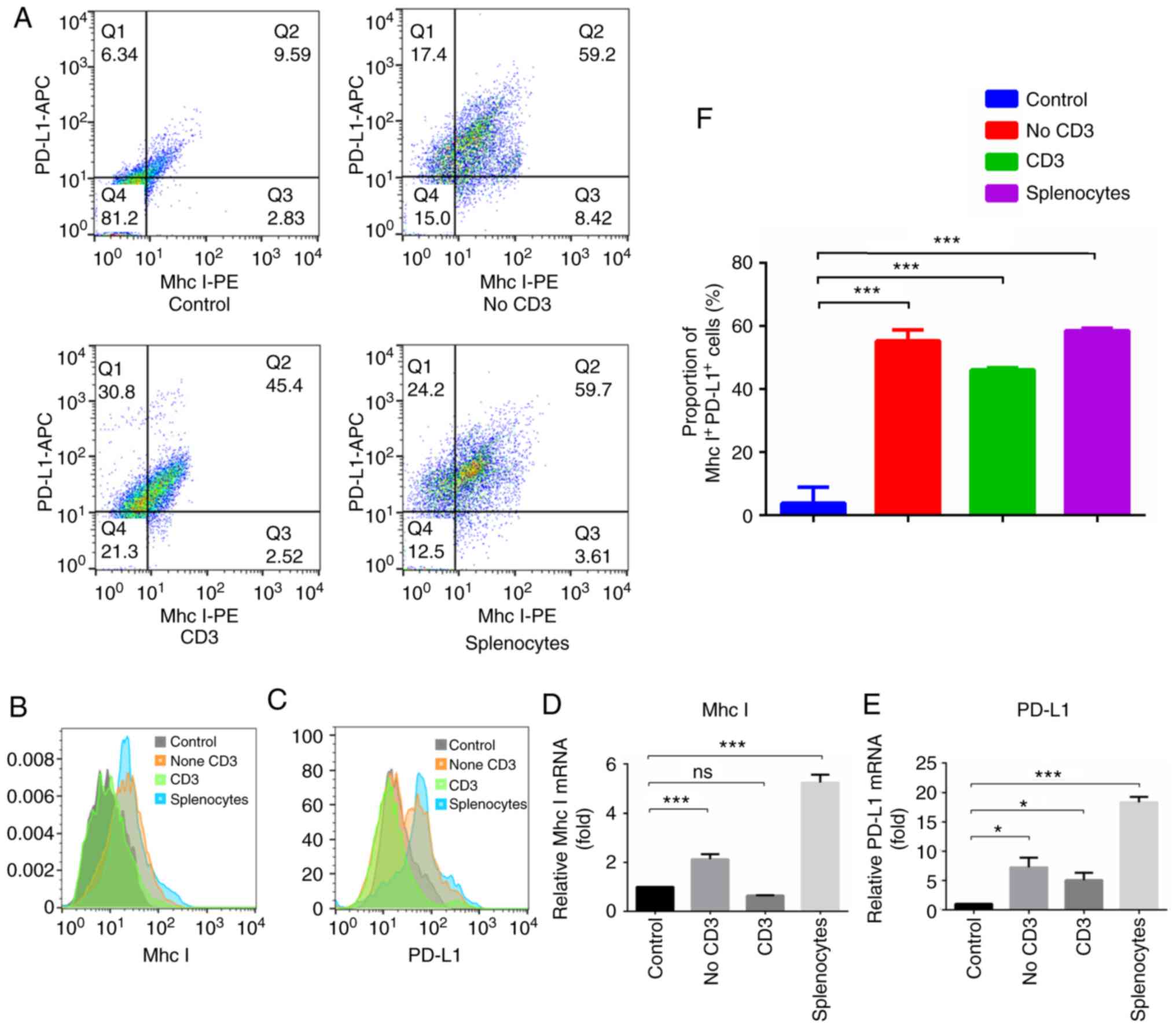
|
1
|
De Moerlooze L, Spencer-Dene B, Revest JM,
Hajihosseini M, Rosewell I and Dickson C: An important role for the
IIIb isoform of fibroblast growth factor receptor 2 (FGFR2) in
mesenchymal-epithelial signalling during mouse organogenesis.
Development. 127:483–492. 2000.PubMed/NCBI
|
|
2
|
Kang H, Kiess A and Chung CH: Emerging
biomarkers in head and neck cancer in the era of genomics. Nat Rev
Clin Oncol. 12:11–26. 2015. View Article : Google Scholar
|
|
3
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Beenken A and Mohammadi M: The FGF family:
Biology, pathophysiology and therapy. Nat Rev Drug Discov.
8:235–253. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rothschild U, Muller L, Lechner A,
Schlösser HA, Beutner D, Läubli H, Zippelius A and Rothschild SI:
Immunotherapy in head and neck cancer-scientific rationale, current
treatment options and future directions. Swiss Med Wkly.
148:w146252018.
|
|
6
|
Gould SE, Junttila MR and de Sauvage FJ:
Translational value of mouse models in oncology drug development.
Nat Med. 21:431–439. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
de Visscher SA, Witjes MJ, van der Vegt B,
de Bruijn HS, van der Ploegvan den Heuvel A, Amelink A, Sterenborg
HJ, Roodenburg JL and Robinson DJ: Localization of liposomal mTHPC
formulations within normal epithelium, dysplastic tissue, and
carcinoma of oral epithelium in the 4NQO-carcinogenesis rat model.
Lasers Surg Med. 45:668–678. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Miki K, Orita Y, Gion Y, Takao S, Ohno K,
Takeuchi M, Ito T, Minoura A, Tachibana T, Marunaka H, et al:
Tumor-associated macrophages in the development of
4-nitro-quinoline-1-oxide-induced tongue squamous cell carcinoma in
a mouse model. Oncology. 93:204–212. 2017. View Article : Google Scholar
|
|
9
|
Young MR: Use of carcinogen-induced
premalignant oral lesions in a dendritic cell-based vaccine to
stimulate immune reactivity against both premalignant oral lesions
and oral cancer. J Immunother. 31:148–156. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tseng SH, Yang CC, Yu EH, Chang C, Lee YS,
Liu CJ, Chang KW and Lin SC: K14-EGFP-miR-31 transgenic mice have
high susceptibility to chemical-induced squamous cell tumorigenesis
that is associating with Ku80 repression. Int J Cancer.
136:1263–1275. 2015. View Article : Google Scholar
|
|
11
|
Miki K, Orita Y, Gion Y, Takao S, Ohno K,
Takeuchi M, Ito T, Hanakawa H, Tachibana T, Marunaka H, et al:
Regulatory T cells function at the early stage of tumor progression
in a mouse model of tongue squamous cell carcinoma. Cancer Immunol
Immunother. 65:1401–1410. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang Y: Cancer immunotherapy: Harnessing
the immune system to battle cancer. J Clin Invest. 125:3335–3337.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lu X, Horner JW, Paul E, Shang X, Troncoso
P, Deng P, Jiang S, Chang Q, Spring DJ, Sharma P, et al: Effective
combinatorial immunotherapy for castration-resistant prostate
cancer. Nature. 543:728–732. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Olson B, Li Y, Lin Y, Liu ET and Patnaik
A: Mouse models for cancer immunotherapy research. Cancer Discov.
8:1358–1365. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Efremova M, Rieder D, Klepsch V,
Charoentong P, Finotello F, Hackl H, Hermann-Kleiter N, Löwer M,
Baier G, Krogsdam A and Trajanoski Z: Targeting immune checkpoints
potentiates immunoediting and changes the dynamics of tumor
evolution. Nat Commun. 9:322018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tang XH, Knudsen B, Bemis D, Tickoo S and
Gudas LJ: Oral cavity and esophageal carcinogenesis modeled in
carcinogen-treated mice. Clin Cancer Res. 10:301–313. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
DePristo MA, Banks E, Poplin R, Garimella
KV, Maguire JR, Hartl C, Philippakis AA, del Angel G, Rivas MA,
Hanna M, et al: A framework for variation discovery and genotyping
using next-generation DNA sequencing data. Nat Genet. 43:491–498.
2011. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
McKenna A, Hanna M, Banks E, Sivachenko A,
Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly
M and DePristo MA: The Genome Analysis Toolkit: A MapReduce
framework for analyzing next-generation DNA sequencing data. Genome
Res. 20:1297–1303. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
20
|
Schumacher TN and Schreiber RD:
Neoantigens in cancer immunotherapy. Science. 348:69–74. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Alexandrov LB, Nik-Zainal S, Wedge DC,
Campbell PJ and Stratton MR: Deciphering signatures of mutational
processes operative in human cancer. Cell Rep. 3:246–259. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Alexandrov LB, Nik-Zainal S, Wedge DC,
Aparicio SA, Behjati S, Biankin AV, Bignell GR, Bolli N, Borg A,
Børresen-Dale AL, et al: Signatures of mutational processes in
human cancer. Nature. 500:415–421. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sun S, Schiller JH and Gazdar AF: Lung
cancer in never smokers-a different disease. Nat Rev Cancer.
7:778–790. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Le Calvez F, Mukeria A, Hunt JD, Kelm O,
Hung RJ, Tanière P, Brennan P, Boffetta P, Zaridze DG and Hainaut
P: TP53 and KRAS mutation load and types in lung cancers in
relation to tobacco smoke: Distinct patterns in never, former, and
current smokers. Cancer Res. 65:5076–5083. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Collura A, Lagrange A, Svrcek M, Marisa L,
Buhard O, Guilloux A, Wanherdrick K, Dorard C, Taieb A, Saget A, et
al: Patients with colorectal tumors with microsatellite instability
and large deletions in HSP110 T17 have improved response to
5-fluorouracil-based chemotherapy. Gastroenterology. 146:401–411.
2014. View Article : Google Scholar
|
|
26
|
Tajima A, Hess MT, Cabrera BL, Kolodner RD
and Carethers JM: The mismatch repair complex hMutS alpha
recognizes 5-fluorouracil-modified DNA: Implications for
chemosensitivity and resistance. Gastroenterology. 127:1678–1684.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hemminki A, Mecklin JP, Jarvinen H,
Aaltonen LA and Joensuu H: Microsatellite instability is a
favorable prognostic indicator in patients with colorectal cancer
receiving chemo-therapy. Gastroenterology. 119:921–928. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Baretti M and Le DT: DNA mismatch repair
in cancer. Pharmacol Ther. 189:45–62. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gao J, Shi LZ, Zhao H, Chen J, Xiong L, He
Q, Chen T, Roszik J, Bernatchez C, Woodman SE, et al: Loss of IFN-γ
pathway genes in tumor cells as a mechanism of resistance to
anti-CTLA-4 therapy. CELL. 167:397–404. 2016. View Article : Google Scholar
|
|
30
|
Hynes NE and Lane HA: ERBB receptors and
cancer: The complexity of targeted inhibitors. Nat Rev Cancer.
5:341–354. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mattox AK, Lee J, Westra WH, Pierce RH,
Ghossein R, Faquin WC, Diefenbach TJ, Morris LG, Lin DT, Wirth LJ,
et al: PD-1 expression in head and neck squamous cell carcinomas
derives primarily from functionally anergic CD4(+) TILs in the
presence of PD-L1(+) TAMs. Cancer Res. 77:6365–6374. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou G, Sprengers D, Boor PPC, Doukas M,
Schutz H, Mancham S, Pedroza-Gonzalez A, Polak WG, de Jonge J,
Gaspersz M, et al: Antibodies against immune checkpoint molecules
restore functions of tumor-infiltrating T cells in hepatocellular
carcinomas. Gastroenterology. 153:1107–1119. 2017. View Article : Google Scholar
|
|
33
|
Bi Y, Deng J, Murry DJ and An G: A
whole-body physiologically based pharmacokinetic model of gefitinib
in mice and scale-up to humans. AAPS J. 18:228–238. 2016.
View Article : Google Scholar :
|
|
34
|
Wherry EJ and Kurachi M: Molecular and
cellular insights into T cell exhaustion. Nat Rev Immunol.
15:486–499. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Topalian SL, Taube JM, Anders RA and
Pardoll DM: Mechanism-driven biomarkers to guide immune checkpoint
blockade in cancer therapy. Nat Rev Cancer. 16:275–287. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Karwacz K, Bricogne C, MacDonald D, Arce
F, Bennett CL, Collins M and Escors D: PD-L1 co-stimulation
contributes to ligand-induced T cell receptor down-modulation on
CD8+ T cells. EMBO Mol Med. 3:581–592. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chen G, Huang AC, Zhang W, Zhang G, Wu M,
Xu W, Yu Z, Yang J, Wang B, Sun H, et al: Exosomal PD-L1
contributes to immunosuppression and is associated with anti-PD-1
response. Nature. 560:382–386. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Flies DB and Chen L: The new B7s: Playing
a pivotal role in tumor immunity. J Immunother. 30:251–260. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Vermeer DW, Coppock JD, Zeng E, Lee KM,
Spanos WC, Onken MD, Uppaluri R, Lee JH and Vermeer PD: Metastatic
model of HPV+ oropharyngeal squamous cell carcinoma demonstrates
heterogeneity in tumor metastasis. Oncotarget. 7:24194–24207. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Stack EC, Wang C, Roman KA and Hoyt CC:
Multiplexed immunohistochemistry, imaging, and quantitation: A
review, with an assessment of Tyramide signal amplification,
multispectral imaging and multiplex analysis. Methods. 70:46–58.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Feng Z, Puri S, Moudgil T, Wood W, Hoyt
CC, Wang C, Urba WJ, Curti BD, Bifulco CB and Fox BA: Multispectral
imaging of formalin-fixed tissue predicts ability to generate
tumor-infiltrating lymphocytes from melanoma. J Immunother Cancer.
3:472015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Feng Z, Jensen SM, Messenheimer DJ, Farhad
M, Neuberger M, Bifulco CB and Fox BA: Multispectral imaging of T
and B cells in murine spleen and tumor. J Immunol. 196:3943–3950.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liu J, Yuan Y, Chen W, Putra J,
Suriawinata AA, Schenk AD, Miller HE, Guleria I, Barth RJ, Huang YH
and Wang L: Immune-checkpoint proteins VISTA and PD-1
nonredundantly regulate murine T-cell responses. Proc Natl Acad Sci
USA. 112:6682–6687. 2015. View Article : Google Scholar : PubMed/NCBI
|