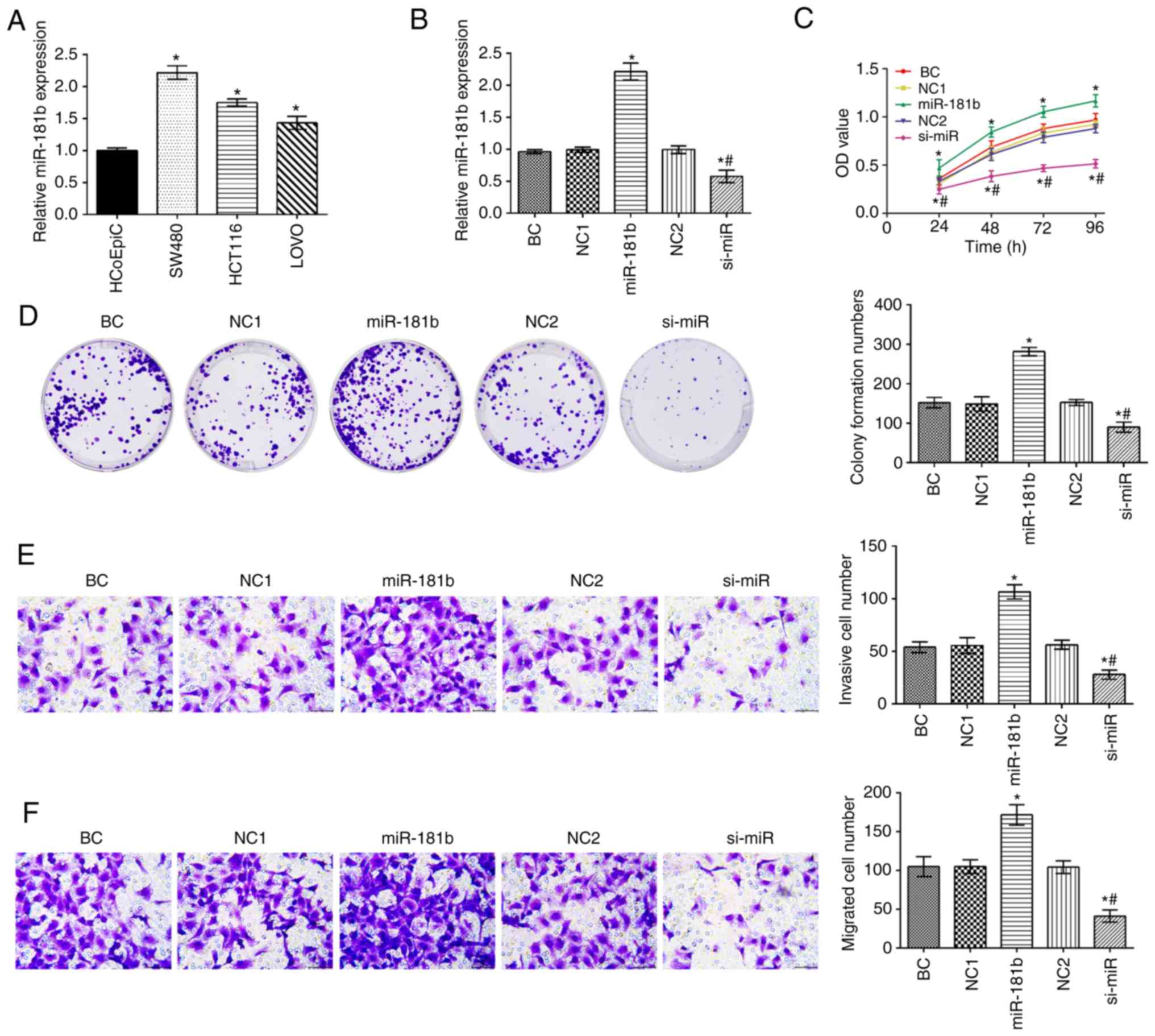
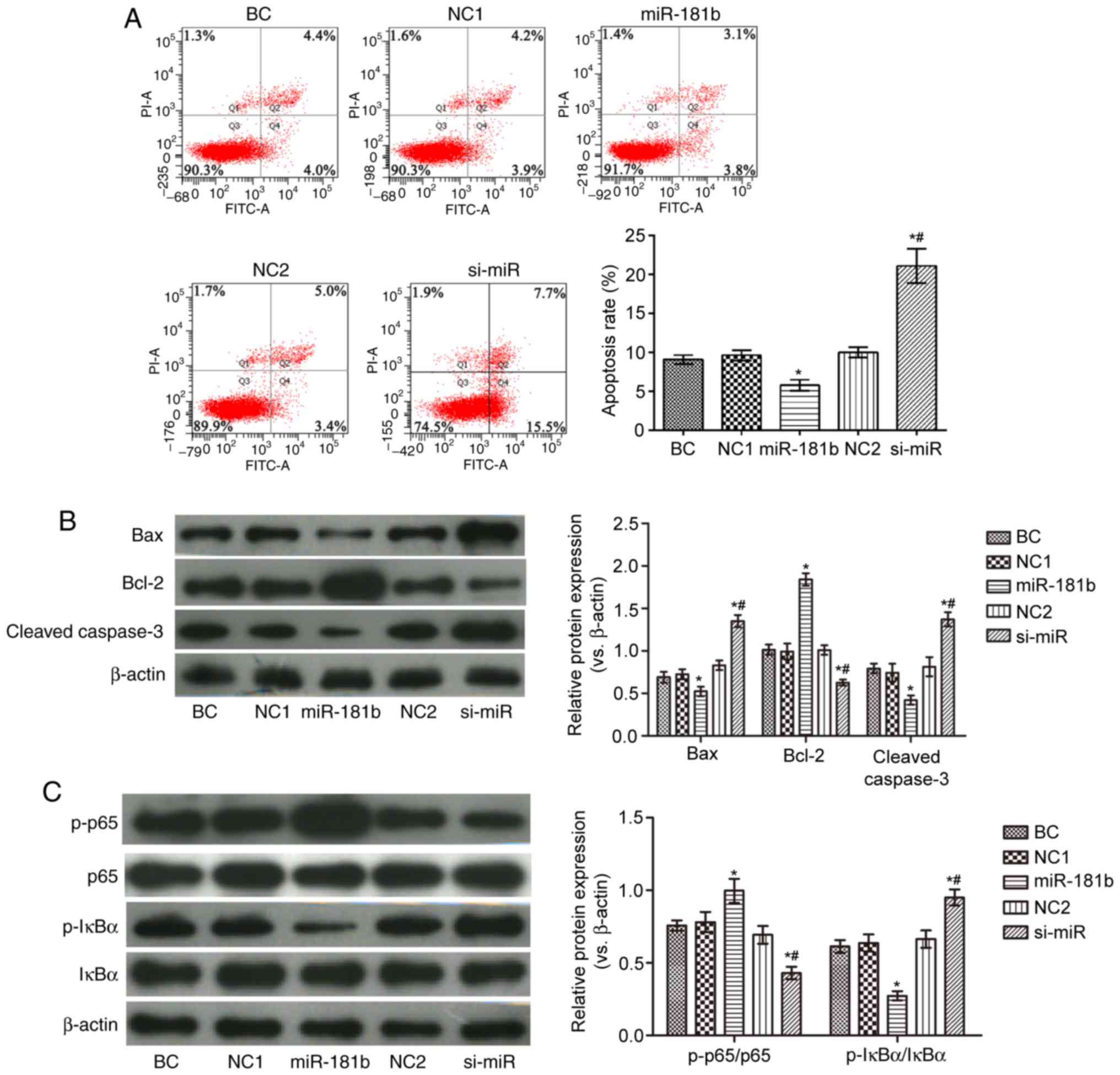
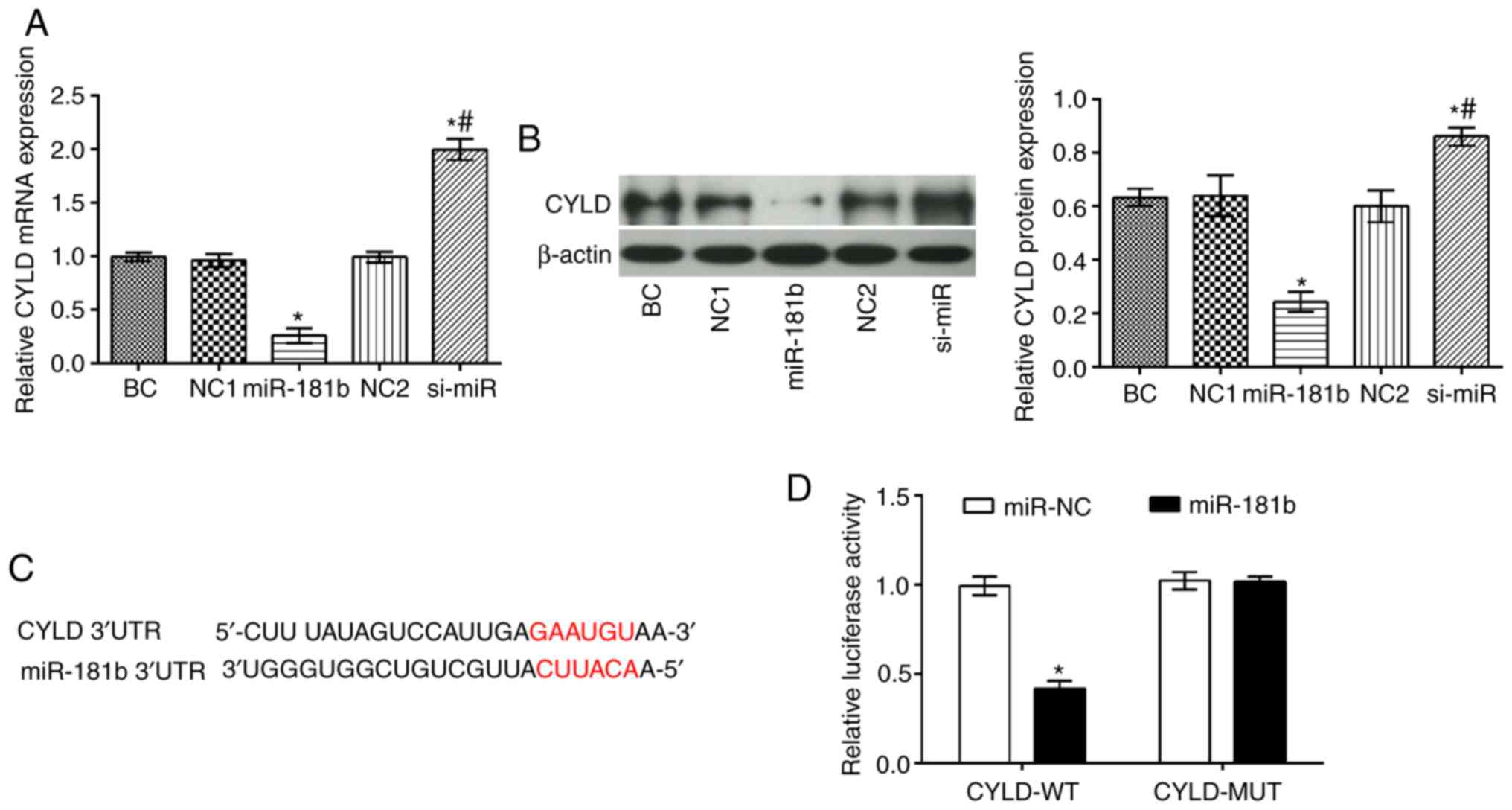
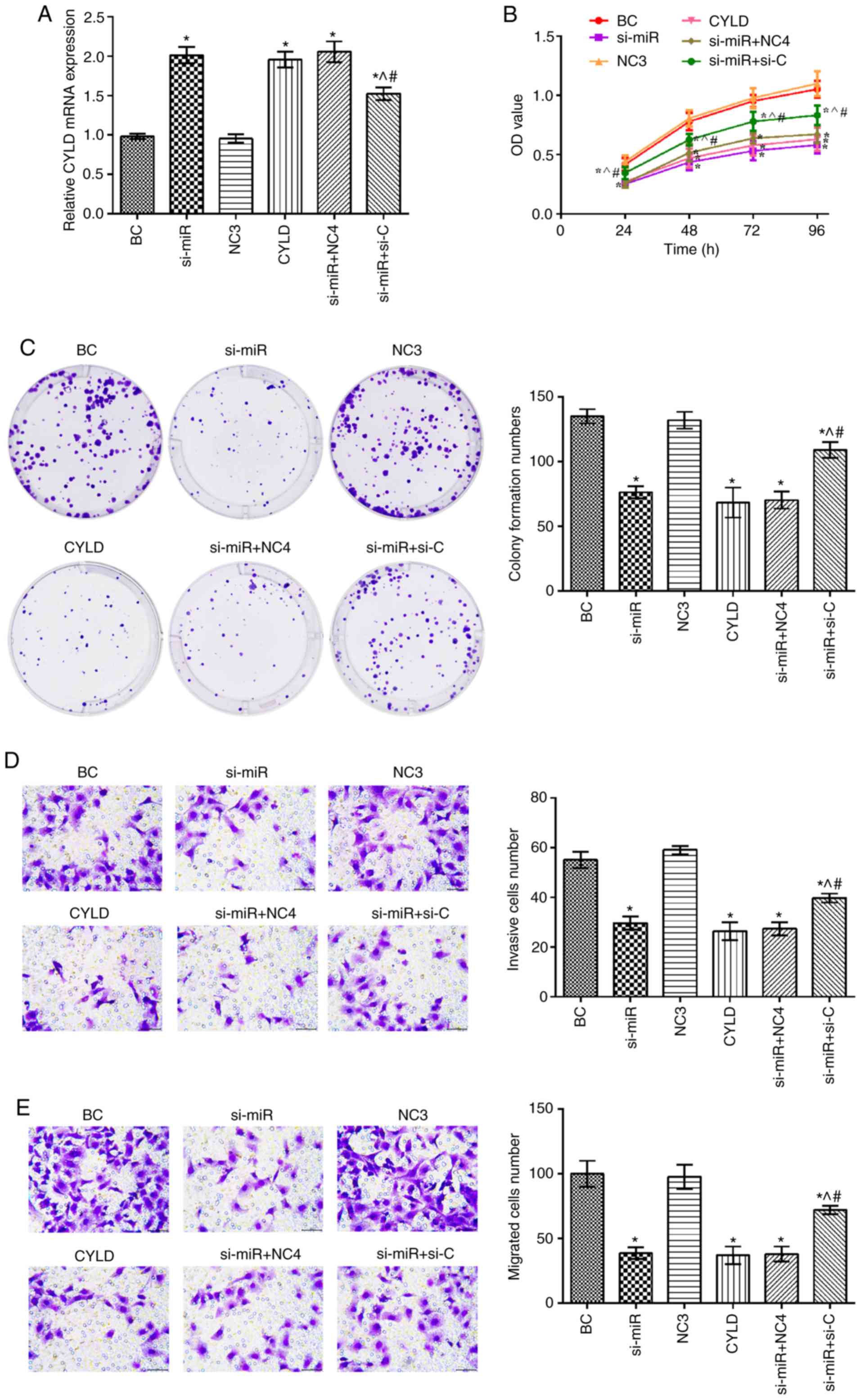
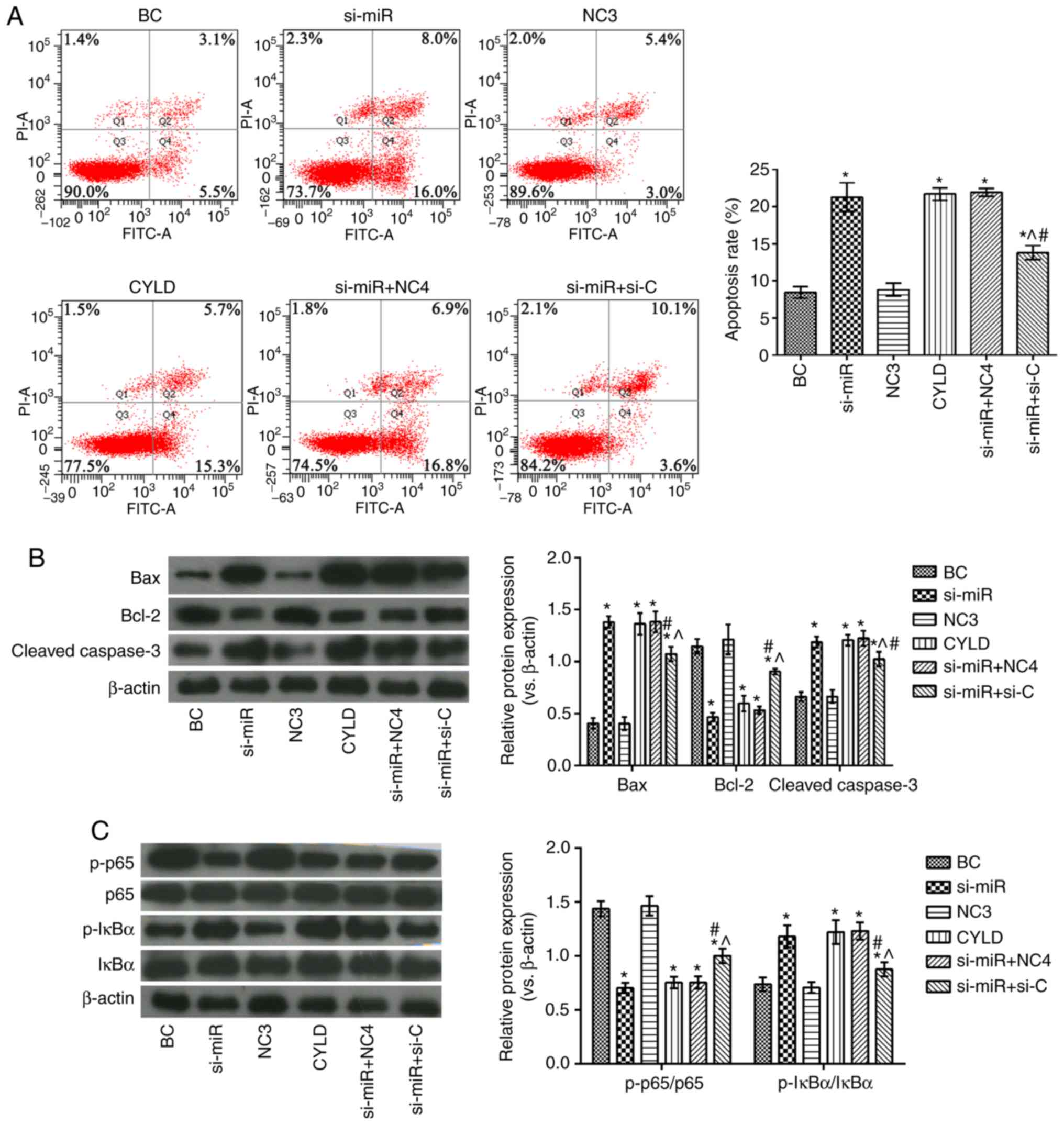
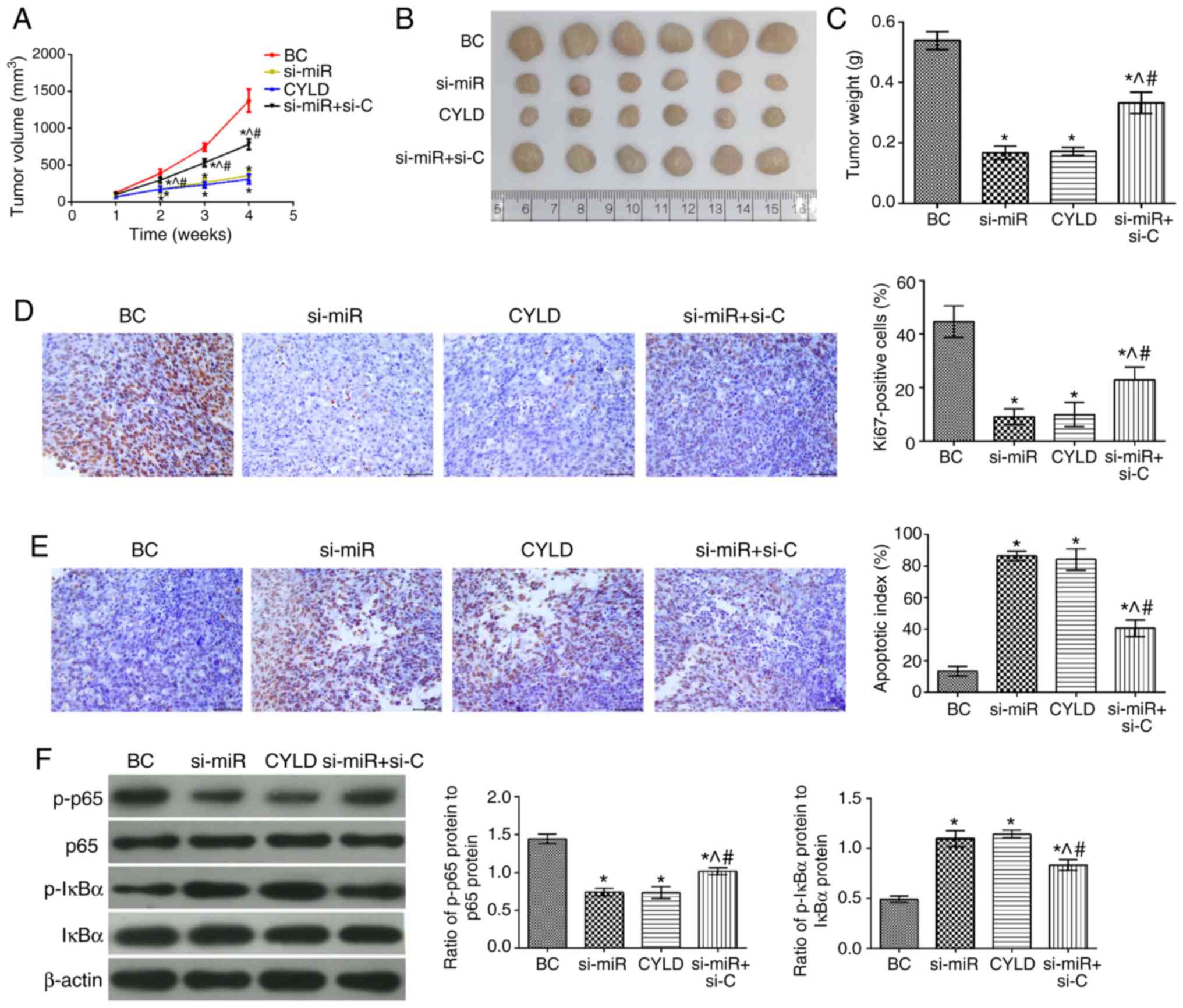
|
1
|
Orangio GR: The economics of colon cancer.
Surg Oncol Clin N Am. 27:327–347. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Labianca R, Beretta GD, Kildani B, Milesi
L, Merlin F, Mosconi S, Pessi MA, Prochilo T, Quadri A, Gatta G, et
al: Colon cancer. Crit Rev Oncol Hematol. 74:106–133. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yang H, Wu J, Zhang J, Yang Z, Jin W, Li
Y, Jin L, Yin L, Liu H and Wang Z: Integrated bioinformatics
analysis of key genes involved in progress of colon cancer. Mol
Genet Genomic Med. 7:e005882019. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sadun RE, Sachsman SM, Chen X, Christenson
KW, Morris WZ, Hu P and Epstein AL: Immune signatures of murine and
human cancers reveal unique mechanisms of tumor escape and new
targets for cancer immunotherapy. Clin Cancer Res. 13:4016–4025.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Indrieri A, Carrella S, Carotenuto P,
Banfi S and Franco B: The pervasive role of the miR-181 family in
development, neurode-generation, and cancer. Int J Mol Sci.
21:20922020. View Article : Google Scholar
|
|
6
|
Ebert MS and Sharp PA: Roles for microRNAs
in conferring robustness to biological processes. Cell.
149:515–524. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhao LD, Zheng WW, Wang GX, Kang XC, Qin
L, Ji JJ and Hao S: Epigenetic silencing of miR-181b contributes to
tumorigenicity in colorectal cancer by targeting RASSF1A. Int J
Oncol. 48:1977–1984. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xia Y and Gao Y: MicroRNA-181b promotes
ovarian cancer cell growth and invasion by targeting LATS2. Biochem
Biophys Res Commun. 447:446–451. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang L, Wang YL, Liu S, Zhang PP, Chen Z,
Liu M and Tang H: miR-181b promotes cell proliferation and reduces
apoptosis by repressing the expression of adenylyl cyclase 9 (AC9)
in cervical cancer cells. FEBS Lett. 588:124–130. 2014. View Article : Google Scholar
|
|
10
|
Rezaei T, Amini M, Hashemi ZS, Mansoori B,
Rezaei S, Karami H, Mosafer J, Mokhtarzadeh A and Baradaran B:
microRNA-181 serves as a dual-role regulator in the development of
human cancers. Free Radic Biol Med. 152:432–454. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin Z, Li D, Cheng W, Wu J, Wang K and Hu
Y: MicroRNA-181 functions as an antioncogene and mediates NF-κB
pathway by targeting RTKN2 in ovarian cancers. Reprod Sci.
26:1071–1081. 2019. View Article : Google Scholar
|
|
12
|
Xia Y, Shen S and Verma IM: NF-κB, an
active player in human cancers. Cancer Immunol Res. 2:823–830.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Xing M: Molecular pathogenesis and
mechanisms of thyroid cancer. Nat Rev Cancer. 13:184–199. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hoesel B and Schmid JA: The complexity of
NF-κB signaling in inflammation and cancer. Mol Cancer. 12:862013.
View Article : Google Scholar
|
|
15
|
Sanches JGP, Xu Y, Yabasin IB, Li M, Lu Y,
Xiu X, Wang L, Mao L, Shen J, Wang B, et al: miR-501 is upregulated
in cervical cancer and promotes cell proliferation, migration and
invasion by targeting CYLD. Chem Biol Interact. 285:85–95. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li X, Abdel-Mageed AB, Mondal D and Kandil
E: The nuclear factor kappa-B signaling pathway as a therapeutic
target against thyroid cancers. Thyroid. 23:209–218. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
18
|
Wang X, Meng Q, Qiao W, Ma R, Ju W, Hu J,
Lu H, Cui J, Jin Z, Zhao Y and Wang Y: miR-181b/Notch2 overcome
chemoresis-tance by regulating cancer stem cell-like properties in
NSCLC. Stem Cell Res Ther. 9:3272018. View Article : Google Scholar
|
|
19
|
Xu DD, Zhou PJ, Wang Y, Zhang L, Fu WY,
Ruan BB, Xu HP, Hu CZ, Tian L, Qin JH, et al: Reciprocal activation
between STAT3 and miR-181b regulates the proliferation of
esophageal cancer stem-like cells via the CYLD pathway. Mol Cancer.
15:402016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pan X, Feng J, Zhu Z, Yao L, Ma S, Hao B
and Zhang G: A positive feedback loop between miR-181b and STAT3
that affects Warburg effect in colon cancer via regulating PIAS3
expression. J Cell Mol Med. 22:5040–5049. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li Y, Huang W and Xu Y, Zhou L, Liang Y,
Gao C, Long Y and Xu Y: CYLD deubiquitinase negatively regulates
high glucose-induced NF-κB inflammatory signaling in mesangial
cells. Biomed Res Int. 2017:39829062017. View Article : Google Scholar
|
|
22
|
Xu RX, Liu RY, Wu CM, Zhao YS, Li Y, Yao
YQ and Xu YH: DNA damage-induced NF-κB activation in human
glioblastoma cells promotes miR-181b expression and cell
proliferation. Cell Physiol Biochem. 35:913–925. 2015. View Article : Google Scholar
|
|
23
|
Zehavi L, Schayek H, Jacob-Hirsch J, Sidi
Y, Leibowitz-Amit R and Avni D: MiR-377 targets E2F3 and alters the
NF-kB signaling pathway through MAP3K7 in malignant melanoma. Mol
Cancer. 14:682015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Iliopoulos D, Jaeger SA, Hirsch HA, Bulyk
ML and Struhl K: STAT3 activation of miR-21 and miR-181b-1 via PTEN
and CYLD are part of the epigenetic switch linking inflammation to
cancer. Mol Cell. 39:493–506. 2010. View Article : Google Scholar : PubMed/NCBI
|