Introduction
Diabetic nephropathy (DN) is a common complication
of diabetes mellitus, and 30–40% of all diabetic patients will
ultimately develop DN (1). The
destructive role of renal tubular epithelial cell (RTEC) apoptosis
in DN progression has been reported by clinical and basic research
(2,3). Therefore, inhibition of RTEC damage
may be an effective treatment strategy to block the occurrence and
development of DN.
MicroRNAs (miRNAs/miRs) are a class of
single-stranded, small non-coding RNAs (21–25 nucleotides), which
degrade or inhibit their target genes at the post-transcriptional
level (4). Numerous studies have
reported the involvement of miRNAs in the progression of diabetes
complications, especially DN (5–7).
For instance, miR-93 can prevent the progression of DN by
downregulating the promoter of the host minichromosome maintenance
complex component 7 gene (8).
Wang et al (9) observed
that miR-21 contributed to renal fibrosis by targeting MMP-9 in a
mouse model of DN. Furthermore, several studies have revealed that
miRNAs are involved in high glucose (HG)-mediated apoptosis in
RTEC. For example, Li et al (10) found that miR-25 was associated
with RTEC apoptosis via the PTEN/AKT pathway. Moreover,
overexpression of miR-23c inhibits the apoptosis of RTEC by
targeting ELAV like RNA binding protein 1 (11). Since the central role of RTEC
injury in DN has been extensively studied (12), identifying additional miRNAs that
are involved in the regulation of the RTEC injury could provide
further in-depth understanding of the pathogenesis of DN.
In the present study, the miRNA expression profile
was examined in peripheral blood from patients with DN using a
microarray assay, and the most downregulated of these, miR-199a-3p,
was selected for further analysis. Using a HG-induced RTEC injury
model, the functional role of miR-199a-3p in RTEC injury and the
underlying mechanisms were investigated.
Materials and methods
Tissue samples
Peripheral blood samples (10 ml) were collected from
30 patients with DN and 30 healthy controls at the Department of
Nephrology, Huaihe Hospital of Henan University between May 2017
and June 2018. The patients with DN included 13 men and 17 women
with a mean age of 34.2 years (age range, 21–62 years), while the
healthy controls included 11 men and 19 women with a mean age of
32.1 years (age range, 20–60 years). The study was approved by the
Research Ethics Committee of Huaihe Hospital of Henan University.
Written informed consent was obtained from all patients.
Cell culture and treatment
HK-2 cells were obtained from the American Type
Culture Collection, and were cultured in DMEM (Thermo Fisher
Scientific, Inc.) with 8% FBS (Gibco; Thermo Fisher Scientific,
Inc.) and 1% penicillin-streptomycin (Beyotime Institute of
Biotechnology) in a 5% CO2 incubator at 37°C. When HK-2
cells reached 90% confluence, the serum was withdrawn for 24 h and
the media were changed to serum-free DMEM containing 5.5 mmol/l
glucose or 30 mM glucose (Sigma-Aldrich; Merck KGaA) (13–15). At 6, 12, 18 and 24 h after
treatment, the expression of miR-199a-3p in HK-2 cells was analyzed
using reverse transcription-quantitative (RT-q)PCR.
miRNA microarray
The microRNA array was performed by Kangcheng
Bio-Tech, Inc. Total RNA was isolated from peripheral blood of
patients with DN and controls using a miRNeasy kit (Qiagen, Inc.).
The RNA purity was determined via NanoDrop ND-1000
spectrophotometry (Thermo Fisher Scientific, Inc.) and the RNA
quality was determined using 1% agarose-formaldehyde denaturing gel
electrophoresis. After RNA quantitation, the samples were assessed
using the miRCURY LNA™ Array v. 16.0 (Exiqon A/S; Qiagen, Inc.)
according to the manufacturer's protocol. The procedure and imaging
processes were performed as described previously (16). The microarray data were analyzed
using Agilent Feature Extraction software (version 10.7; Agilent
Technologies, Inc.) (17).
Differentially expressed miRNAs were screened with an unpaired
t-test (P<0.05) combined with a significant threshold value of a
fold change [FC; (log2 (FC) >2 for upregulated, and log2 (FC)
≤-2 for downregulated]. The microarray data that support the
findings of this study are available from the corresponding author
upon reasonable request.
RT-qPCR
Total RNA was extracted from peripheral blood
samples or cells using TRIzol® reagent (Thermo Fisher
Scientific, Inc.). cDNA was synthesized using PrimeScript One Step
RT-PCR kit (Takara Biotechnology Co., Ltd.) for 60 min at 42°C.
RT-qPCR was performed using the SYBR Green PCR kit (Toyobo Life
Science) on an ABI 7500 system (Thermo Fisher Scientific, Inc.).
The primers for RT-qPCR analysis were as follows: miR-199a-3p
forward, 5′-TACAGGACCGGTCGACTAGG-3′ and reverse,
5′-CTCGCTTCGGCAGCACA-3′; U6 forward, 5′-TGCGGGTGCTCGCTTCGCAGC-3′
and reverse, 5′-CCAGTGCAGGGTCCGAGGT-3′; IKKβ forward,
5′-AGCTCTGGAACCTCCTGAAGA-3′ and reverse,
5′-AGCTCCAGTCTAGGGTCGTGA-3′; and GAPDH forward,
5′-AGGTCGGTGTGAACGGATTTG-3′ and reverse:
5′-TGTAGACCATGTAGTTGAGGTCA-3′. The reaction mixtures were denatured
at 95°C for 3 min, followed by 40 two-step cycles of 95°C for 10
sec and 60°C for 30 sec. Relative quantification was determined via
normalization to U6 or GAPDH. The relative expression levels were
calculated based on the 2−ΔΔCq method (18).
Cell transfection
HK-2 cells (5×105/well) were seeded in a
6-well plate overnight, and then cells were transfected with 20 nM
miR-199a-3p mimics, 20 nM mimics negative control (NC), 20 nM
miR-199a-3p inhibitor, 20 nM inhibitor NC, 2 μg pcDNA-IKKβ
or pcDNA3.1-vector (Shanghai GenePharma Co., Ltd.) using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.). The sequences were as follows: miR-199a-3p
mimics, 5′-ACAGUAGUCUGCACAUUGGUUA-3′; mimics NC,
5′-GGACCAAATCTCGAGATTTGG-3′; miR-199a-3p inhibitor,
5′-UAACCAAUGUGCAGACUACUGU-3′; and inhibitor NC,
5′-TTCTCCGAACGTGTCACGTTTC-3′. After 24 h transfection, HK-2 cells
were stimulated with HG (30 mM) for 24 h at 37°C, and then protein
and RNA were extracted for analyses.
Cell Counting Kit-8 (CCK-8) assay
After 24 h transfection, HK-2 cells
(5×103) were stimulated with HG (30 mM) for 24 h at
37°C, and then cell viability in 96-well plates was evaluated using
CCK-8 assay according to the manufacturer's instructions. Briefly,
10 μl CCK-8 solution (Dojindo Molecular Technologies, Inc.)
was added to each well and incubated at 37°C for another 2 h. The
absorbance at 450 nm was measured using a microplate reader
(Bio-Rad Laboratories, Inc.).
Flow cytometry assay
After 24 h transfection, HK-2 cells were stimulated
with HG (30 mM) at 37°C for 24 h, and then apoptosis was evaluated
using Annexin V/PI apoptosis-detection kit (Nanjing KeyGen Biotech
Co., Ltd.), according to the manufacturer's protocols. The cells
were harvested by ice-cold PBS and stained with FITC-Annexin V and
PI in binding buffer for 15 min at room temperature in the dark.
Then, cell apoptosis were detected with an EPICS XL-MCL FACScan
flow cytometer (Becton, Dickinson and Company) and analyzed using
FlowJo 8.7.1 software (FlowJo LLC). The results indicated healthy
viable cells in the lower left quadrant (Q4) on the scatter plot as
(FITC-/PI-). The lower right quadrant (Q3) represented the early
stage apoptotic cells as (FITC+/PI−). The
upper right quadrant (Q2) represented necrotic cells and late stage
apoptotic cells as (FITC+/PI+). Apoptotic
rate = percentage of early stage apoptotic cells (Q3) + percentage
of late stage apoptotic cells (Q2). The experiment was repeated
three times independently.
Caspase-3 activity assay
After 24 h transfection, HK-2 cells were stimulated
with HG (30 mM) at 37°C for 24 h, and cells (5×103/well)
were collected and lysed using RIPA lysis buffer (Beyotime
Institute of Biotechnology). Then, caspase-3 activity in was
determined using a caspase-3 Activity Assay kit (Beyotime Institute
of Biotechnology), according to the manufacturer's protocol. The
optical density was measured at a wavelength of 450±2 nm using a
microplate reader (Bio-Rad Laboratories, Inc.).
ELISA assay
After 24 h transfection, HK-2 cells were stimulated
with HG (30 mM) at 37°C for 24 h, and then the levels of IL-6 (cat.
no. p1330), IL-8 (cat. no. p1640) and TNF-α (cat. no. pt518) were
evaluated using commercial ELISA kits (Beyotime Institute of
Biotechnology).
Vector construction
Luciferase reporters were generated based on the
firefly luciferase expressing vector pGL3-control (Promega
Corporation). To construct pGL3-IKKβ-3′ untranslated region (UTR),
a partial 3′UTR of the IKKβ segment of human IKKβ mRNA containing
the putative miR-199a-3p binding sites was amplified and cloned
into the vector pGL3-control. Mutations within the potential
miR-199a-3p binding sites were introduced using a QuikChange
Site-Directed Mutagenesis kit (Thermo Fisher Scientific, Inc.).
Following sequencing, the recombinant segment of the correct clone
was incised by BamHI and XbaI (Takara Bio, Inc.). The
recombinant segment was inserted into pGL3 vector, which was
incised by the same two restriction endonucleases. The clones were
sequenced, and the correct clones were amplified and identified
before transfection.
Luciferase assays
TargetScan 7.0 (http://www.targetscan.org) and PicTar (https://pictar.mdc-berlin.de/; release 2007) were used
to search for the putative targets of miR-199a-3p. The
dual-luciferase reporter assay was performed as described
previously (19). HK-2 cells were
transfected with 20 nM miR-199a-3p or inhibitor and the luciferase
reporter plasmids using Lipofectamine® 2000 (Invitrogen;
Thermo Fisher Scientific, Inc.). After 48 h, luciferase activity
was assessed using the dual luciferase reporter kit (Beyotime
Institute of Biotechnology). Renilla activity was used to
normalize firefly luciferase activity.
Western blot analysis
After 24 h transfection, HK-2 cells were stimulated
with HG (30 mM) at 37°C for 24 h. Then, total protein was obtained
using RIPA lysis buffer (Beyotime Institute of Biotechnology) and
quantified with a BCA protein assay kit (Pierce; Thermo Fisher
Scientific, Inc.). Next, the proteins (40 μg/lane) in the
lysates were separated via 12% SDS-PAGE and transferred to PVDF
membranes (Cytiva). After being blocked with a 5% skim milk
solution for 1 h at room temperature, the specific primary
antibodies were incubated in the membranes at 4°C overnight,
including IKKβ (cat. no. 8943; 1:1,000), IL-6 (cat. no. 12912;
1:1,000), TNF-α (cat. no. 11948; 1:1,000), Bax (cat. no. 5023;
1:1,000), Bcl-2 (cat. no. 3498; 1:1,000), cleaved-poly(ADP-ribose)
polymerase 1 (PARP; cat. no. 5625; 1:1,000), total PARP (cat. no.
9532; 1:2,000), cleaved-caspase-3 (cat. no. 9654; 1:1,000), total
caspase-3 (cat. no. 14220; 1:1,000), phosphorylated (p)-IκB-α (cat.
no. 2859, 1:1,000), IκB-α (cat. no. 4814; 1:1,000), nuclear p-p65
(cat. no. 3033, 1:1,000), Histone H3 (cat. no. 9728; 1:1,000) and
β-actin (cat. no. 4970; 1:2,000). All antibodies were obtained from
Cell Signaling Technology, Inc., while IL-8 (cat. no. ab110727;
1:1,000) was obtained from Abcam. Subsequently, the corresponding
anti-mouse or anti-rabbit secondary horseradish
perxoidase-conjugated antibodies (cat. nos. 7076 and 7074; 1:2,000)
were added into the membranes for 2 h at room temperature. The
protein bands were visualized using an ECL detection system (Thermo
Fisher Scientific, Inc.). Semi-quantification was performed using
ImageJ version 1.46 (National Institutes of Health).
Statistical analysis
Statistical analysis was performed using GraphPad
Prism (Version 5.0; GraphPad Software, Inc.). Data are presented as
the mean ± SD. Differences between groups were analyzed using
unpaired Student's t-test or one-way ANOVA followed by Bonferroni
post hoc test. Pearson's analyses were used for correlation
analysis between miR-199a-3p and IKKβ expression levels. P<0.05
was considered to indicate a statistically significant difference.
The experiment was repeated three times independently.
Results
miR-199a-3p is downregulated in
peripheral blood of patients with DN
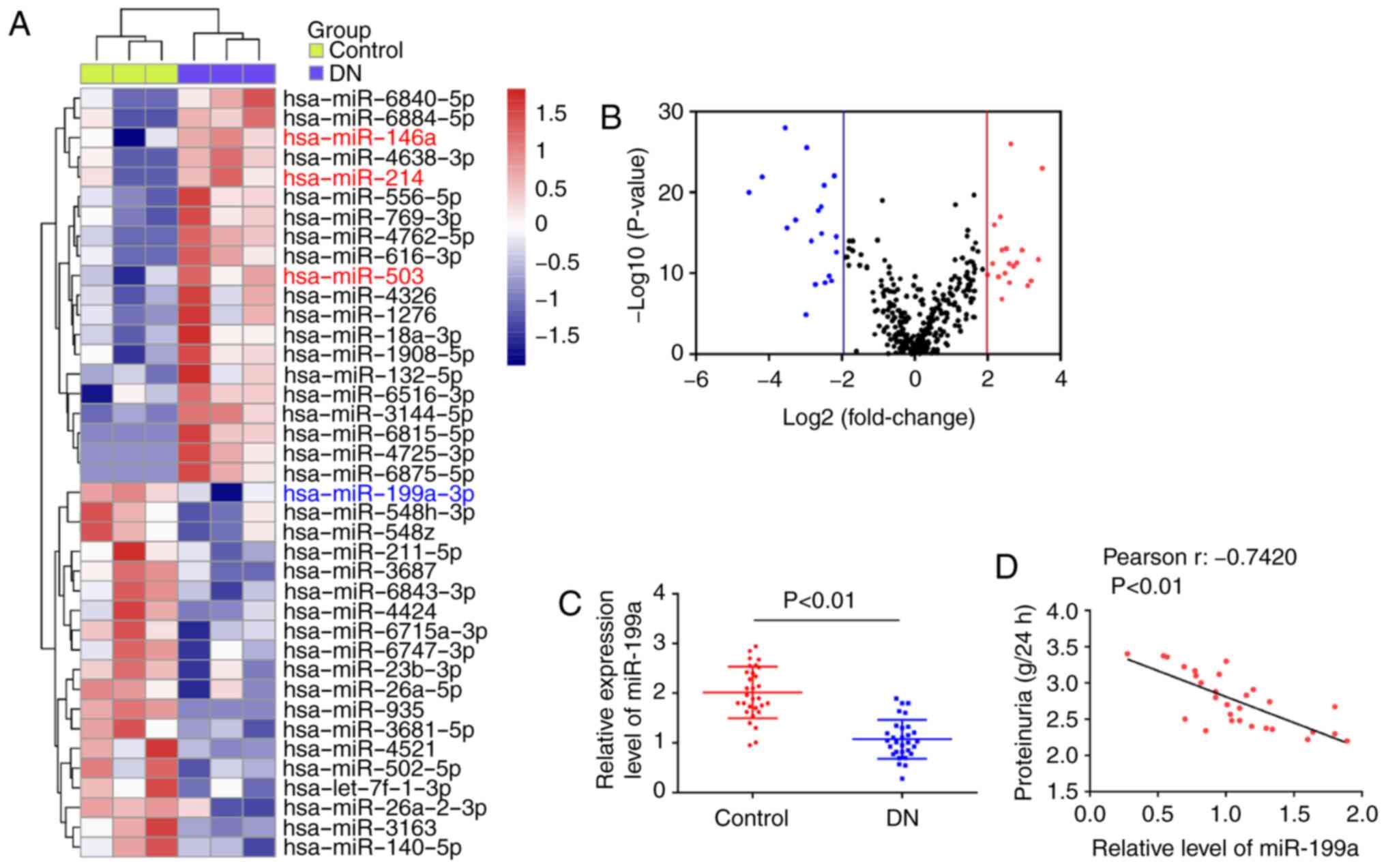
Microarray was used to compare the miRNA patterns
between peripheral blood of patients with DN and healthy controls.
The miRNA microarray identified 20 miRNAs (e.g miR-146a, miR-214
and miR-503) that were upregulated and 19 miRNAs (e.g miR-199a-3p)
that were downregulated by >4-fold in the peripheral blood of
patients with DN (Fig. 1A). The
volcano plot demonstrates all of the differentially expressed miR
between peripheral blood from patients with DN and healthy controls
(Fig. 1B). Among these aberrantly
expressed miRNAs, miR-146a, miR-214 and miR-503 were increased,
which was consistent with previous reports (6,20,21), indicating the reliability of the
present microarray. Of the downregulated miRNAs, miR-199a-3p was
identified as one of the most significantly downregulated.
Next, RT-qPCR was performed to further detect
miR-199a-3p expression in clinical samples (n=30). It was
identified that miR-199a-3p expression was significantly
downregulated in the DN group, compared with the control group
(Fig. 1C). Moreover, a negative
correlation was observed between miR-199a-3p and proteinuria in
patients with DN (Fig. 1D).
Therefore, miR-199a-3p may be a useful biomarker in DN diagnosis
and evaluating its severity.
Overexpression of miR-199a-3p suppresses
HG-induced the apoptosis in HK-2 cells
As an initial step, an in vitro model was
established via HG (30 mM) treatment in HK-2 cells, a human RTEC
cell line that is widely used for the DN research (22). Then, the expression of miR-199a-3p
in HK-2 cells was analyzed at different times (6, 12, 18 and 24 h)
using RT-qPCR. Consistent with the results in the clinical samples,
HG treatment time-dependently decreased the expression of
miR-199a-3p in HK-2 cells (Fig.
2A).
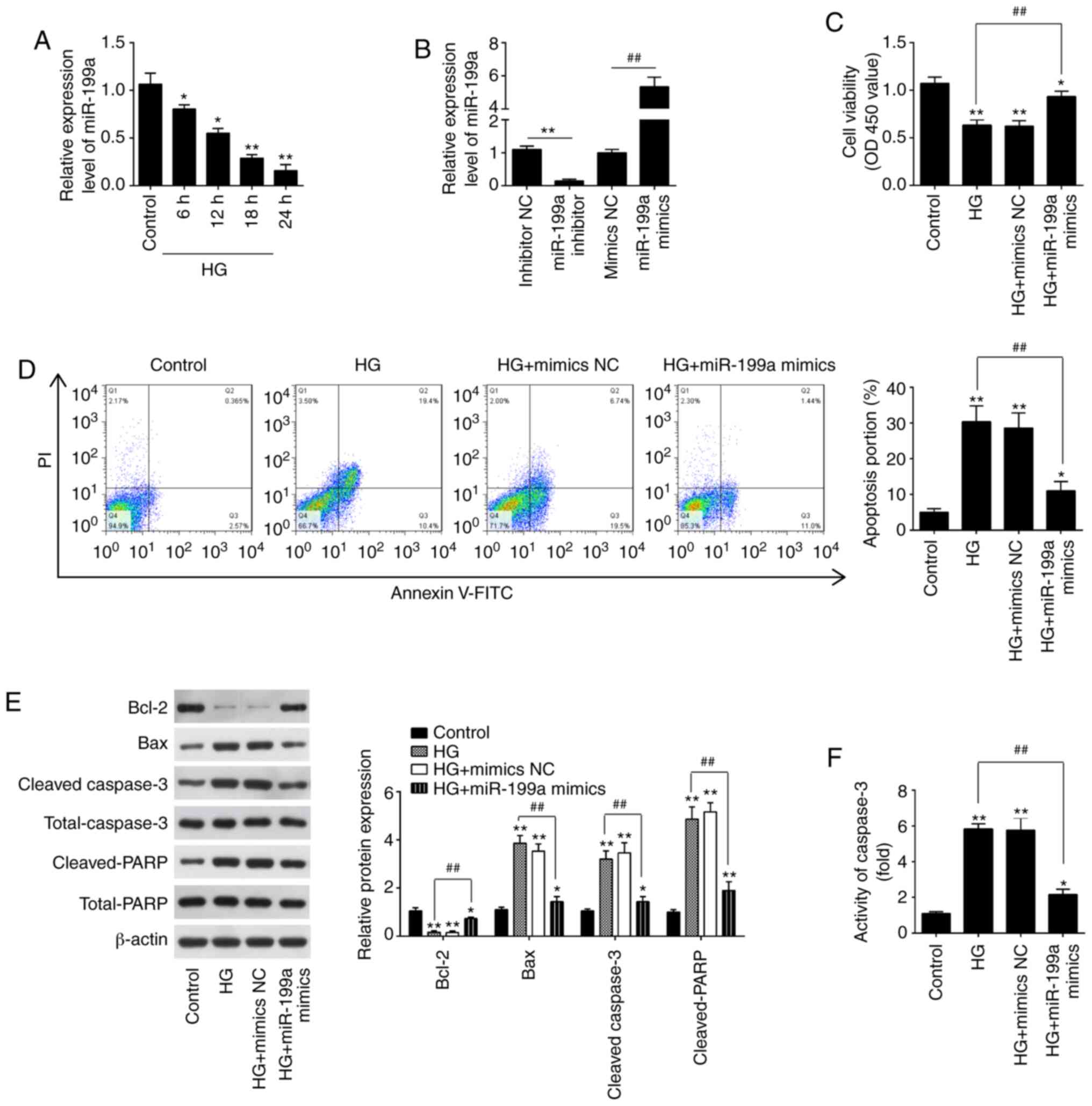 | Figure 2Overexpression of miR-199a-3p
inhibits HG-induced HK-2 cell apoptosis. (A) Expression of
miR-199a-3p was determined via RT-qPCR analysis at 6, 12, 18 and 24
h after HG treatment. *P<0.05, **P<0.01
vs. Control group. (B) Expression of miR-199a-3p was determined via
RT-qPCR analysis 24 h after miR-199a-3p mimics and miR-199a-3p
inhibitor transfection. **P<0.01 vs. mimics NC group;
##P<0.01 vs. inhibitor NC group. (C) HK-2 cells were
transfected with miR-199a-3p mimics for 24 h, followed by treatment
with 30 mM HG for another 24 h, and then cells were harvested for
subsequent experiments. Cell viability was assessed using Cell
Counting Kit-8 assay. (D) Apoptosis was measured using flow
cytometry. (E) Protein expression levels of Bax, Bcl-2, cleaved
caspase-3 and cleaved-PARP were measured via western blotting. (F)
Activity of caspase-3 was measured using a caspase-3 Activity Assay
kit. Data are presented as the mean ± SD of three independent
experiments. *P<0.05, **P<0.01 vs.
control group; ##P<0.01 vs. HG group. HG, high
glucose; miR, microRNA; NC, negative control; PARP,
poly(ADP-ribose) polymerase 1; OD, optical density; RT-qPCR,
reverse transcription-quantitative PCR. |
Subsequently, miR-199a-3p mimics and miR-199a-3p
inhibitor were transfected into the cultured HK-2 cells, and the
effects on cell viability and apoptosis were examined. It was
demonstrated that miR-199-3p expression was significantly increased
after miR-199a-3p mimics transfection, and decreased after
miR-199-3p inhibitor transfection in HK-2 cells (Fig. 2B). According to the results of
CCK-8 assay, HG treatment led to a significant decline in the cell
viability, compared with the control group; however, the viability
was significantly increased after miR-199a-3p mimics transfection
(Fig. 2C). Moreover, miR-199a-3p
overexpression weakened the HG-induced apoptosis of HK-2 cells
(Fig. 2D). It was also found that
HG significantly increased the expression levels of Bax,
cleaved-caspase-3 and cleaved-PARP, but decreased the expression of
Bcl-2 compared with the control group in HK-2 cells; these effects
induced by HG were significantly attenuated by miR-199a-3p
overexpression (Fig. 2E).
Furthermore, miR-199a-3p overexpression weakened the HG-induced
caspase-3 activity (Fig. 2F).
Collectively, these data suggested that miR-199a-3p could relieve
the HK-2 cell apoptosis caused by HG.
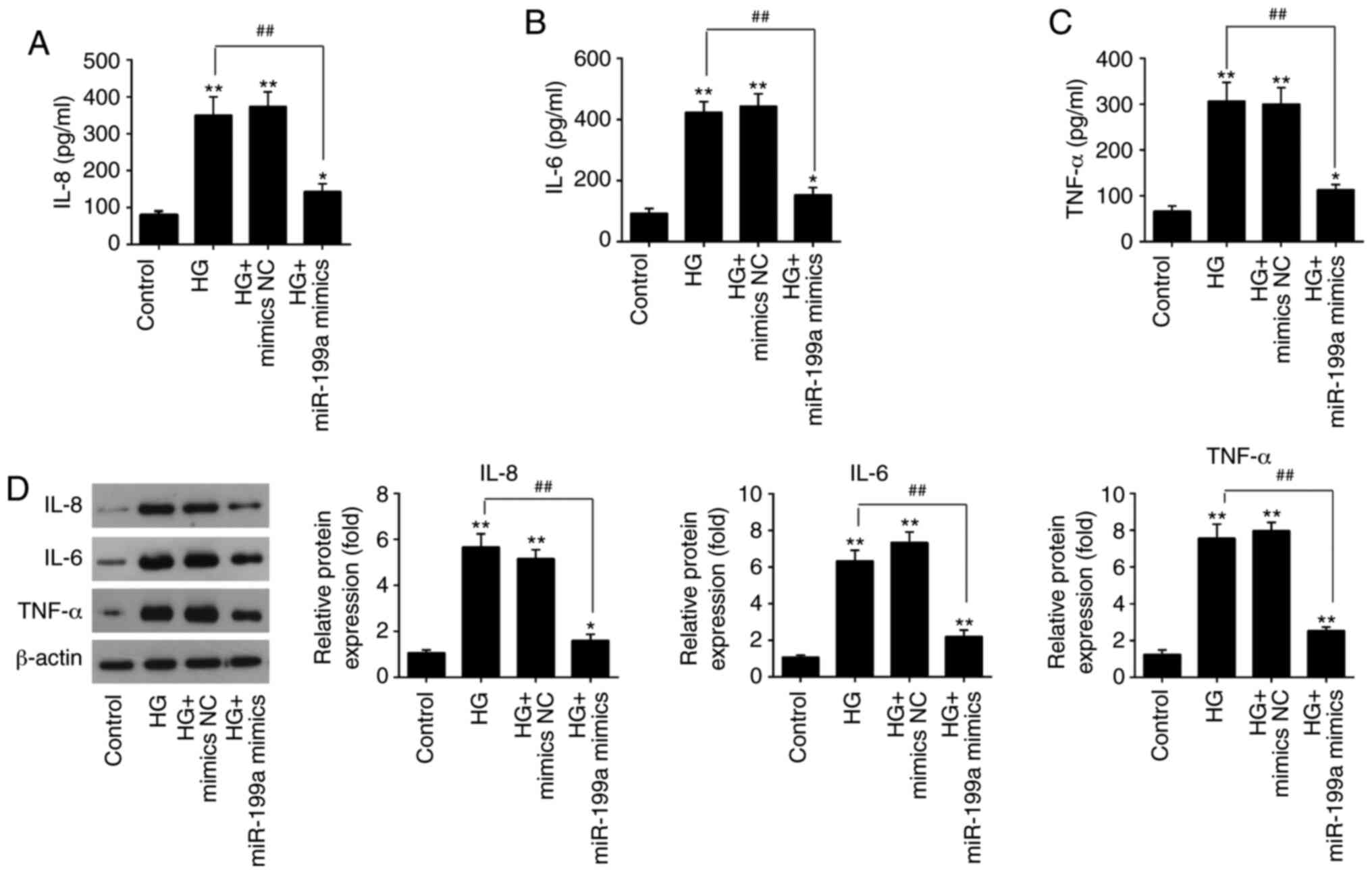
Overexpression of miR-199a-3p attenuates
the HG-induced inflammatory response in HK-2 cells
It has been reported that the inflammatory response
is correlated with DN progression and is an important pathological
bases of DN (23). Therefore, the
present study examined the influences of miR-199a-3p on the
releases of inflammatory cytokines in HG-treated HK-2 cells. The
levels of IL-8 (Fig. 3A), IL-6
(Fig. 3B) and TNF-α (Fig. 3C) were significantly increased
after HG stimulation, compared with the control group, but
overexpression of miR-199a-3p significantly decreased the release
of these cytokines in HG-treated HK-2 cells. The protein expression
levels of these pro-inflammatory cytokines were also detected via
western blotting. Compared with the control group, HG treatment
significantly elevated these protein expression levels in HK-2
cells; however, miR-199a-3p overexpression abolished HG-induced
effects on protein expression levels (Fig. 3D). Thus, it was indicated that
miR-199a-3p inhibited the HG-induced inflammatory response in HK-2
cells.
miR-199a-3p blocks the activation of the
NF-κB pathway in HK-2 cells
In DN, NF-κB is constitutively active and is
involved in promoting tubular cell injury, which suggests that
inhibiting the activity of NF-κB may constitute a promising
therapeutic approach to prevent DN (24,25). Therefore, the influence of
miR-199a-3p on the expression levels of key components in the NF-κB
pathway were examined in HK-2 cells. HG treatment significantly
upregulated the expression levels of IKKβ, p-IκBα and nuclear
p-p65, compared with the control group. However, compared with HG
group, miR-199a-3p overexpression significantly downregulated these
expression levels (Fig. 4A and
B). These data demonstrated that miR-199a-3p blocked the
activation of the NF-κB signaling pathway, which may be involved in
the inhibition of inflammatory mediator release in HG-induced HK-2
cells.
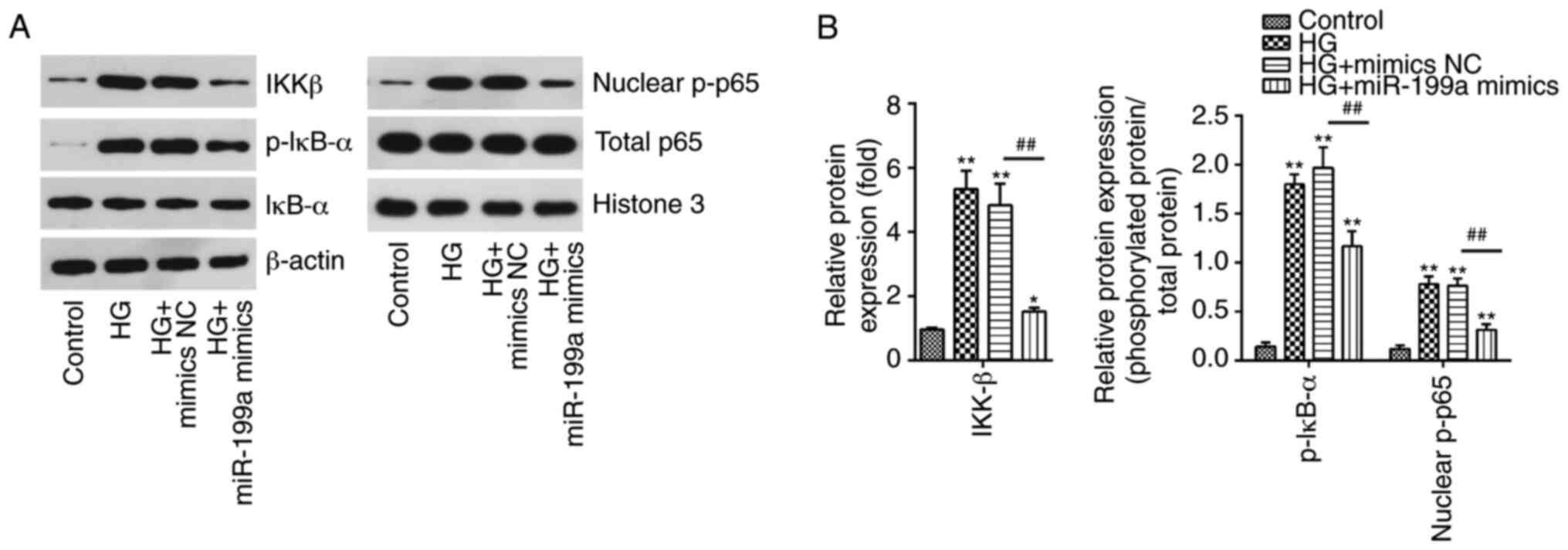 | Figure 4miR-199a-3p blocks the HG-induced
activation of the NF-κB pathway. HK-2 cells were transfected with
miR-199a-3p mimics for 24 h, followed by treatment with 30 mM HG
for another 24 h, and then cells were harvested for subsequent
experiments. (A) Protein expression levels of IKKβ, IκBα, p-IκB-α,
nuclear p-p65 and total p65 were assessed using western blotting.
(B) Bands were semi-quantitatively analyzed using ImageJ software,
and normalized to β-actin or Histone H3 density. Data are presented
as the mean ± SD of three independent experiments.
*P<0.05, **P<0.01 vs. Control group;
##P<0.01 vs. HG + mimics NC group. HG, high glucose;
miR, microRNA; NC, negative control; p-, phosphorylated. |
IKKβ is a direct target of miR-199a-3p in
HK-2 cells
To evaluate how miR-199a-3p regulates the NF-κB
pathway in HG-induced cellular injury in vitro, two
bioinformatics databases, TargetScan 7.0 (http://www.targetscan.org) and PicTar (https://pictar.mdc-berlin.de/; release 2007), were
utilized to predict the targets of miR-199a-3p. IKKβ, one of the
upstream molecules of the NF-κB signaling pathway, may be a
potential target of miR-199a-3p (Fig.
5A). Moreover, it was identified that IKKβ mRNA expression was
significantly downregulated by miR-199a-3p mimics transfection, but
upregulated by miR-199a-3p inhibitor transfection (Fig. 5B). To experimentally validate
whether IKKβ was a direct target of miR-199a-3p, a luciferase
reporter assay was performed. The luciferase activity of the
IKKβ-3′UTR wild-type was decreased in HK-2 cells after miR-199a-3p
mimics transfection, whereas it was increased by miR-199a-3p
inhibitor transfection, compared with NC group (Fig. 5C). However, the luciferase
activity of IKKβ-3′UTR mutant reporter plasmid demonstrated no
significant change.
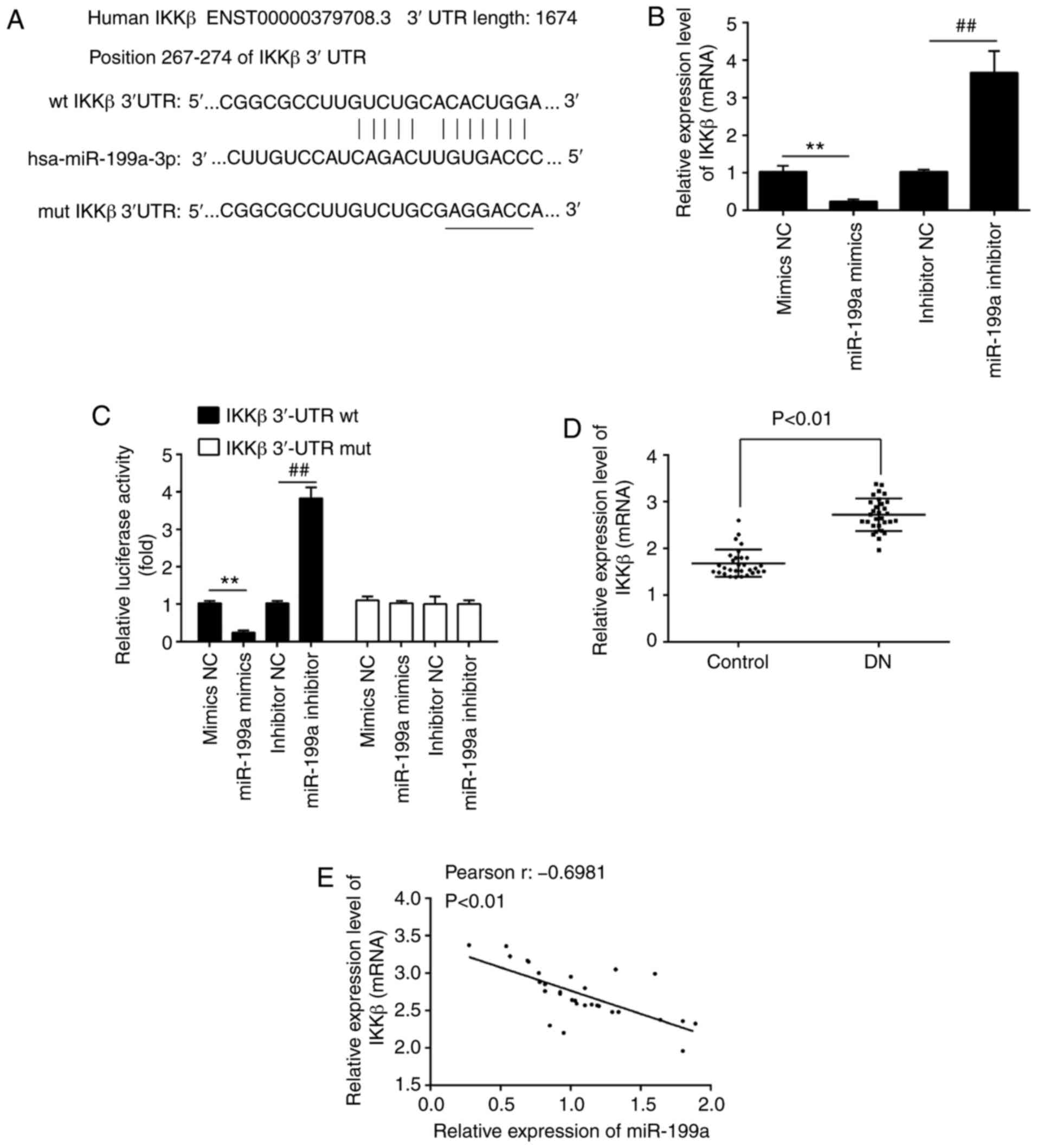 | Figure 5IKKβ is a direct target of
miR-199a-3p in HK-2 cells. (A) Putative binding site of miR-199a-3p
and IKKβ. (B) RT-qPCR analysis was used to assess the expression of
IKKβ in HK-2 cells after miR-199a-3p mimics or miR-199a-3p
inhibitor transfection. (C) HK-2 cells were co-transfected with
firefly luciferase constructs containing the IKKβ wt or mut 3′UTRs
and miR-199a-3p mimics, mimics NC, miR-199a-3p inhibitor or
inhibitor NC. Then, the luciferase activity was analyzed using a
dual-luciferase reporter assay system. Data are presented the mean
± SD of three independent experiments. **P<0.01 vs.
mimics NC group; ##P<0.01 vs. inhibitor NC group. (D)
RT-qPCR analysis was conducted to assess the expression of IKKβ in
30 peripheral blood samples from patients with DN and 30 peripheral
blood samples from healthy controls. (E) Pearson's correlation was
used to assess the correlation between miR-199a-3p and IKKβ
expression levels in peripheral blood samples from patients with DN
(r, -0.6981; P<0.01). RT-qPCR, reverse
transcription-quantitative PCR; HG, high glucose; miR, microRNA;
NC, negative control; wt, wild-type; mut, mutant; UTR, untranslated
region. |
The expression of IKKβ was measured in the
aforementioned clinical samples, and it was found that IKKβ was
significantly upregulated in the DN group compared with the control
group (Fig. 5D). Furthermore, a
moderate negative correlation between miR-199a-3p and IKKβ
expression levels in peripheral blood samples of patients with DN
was observed (Fig. 5E). These
data indicated that IKKβ was a functional target of miR-199a-3p in
HK-2 cells.
miR-199a-3p inhibits HG-induced apoptosis
and inflammatory response by targeting IKKβ
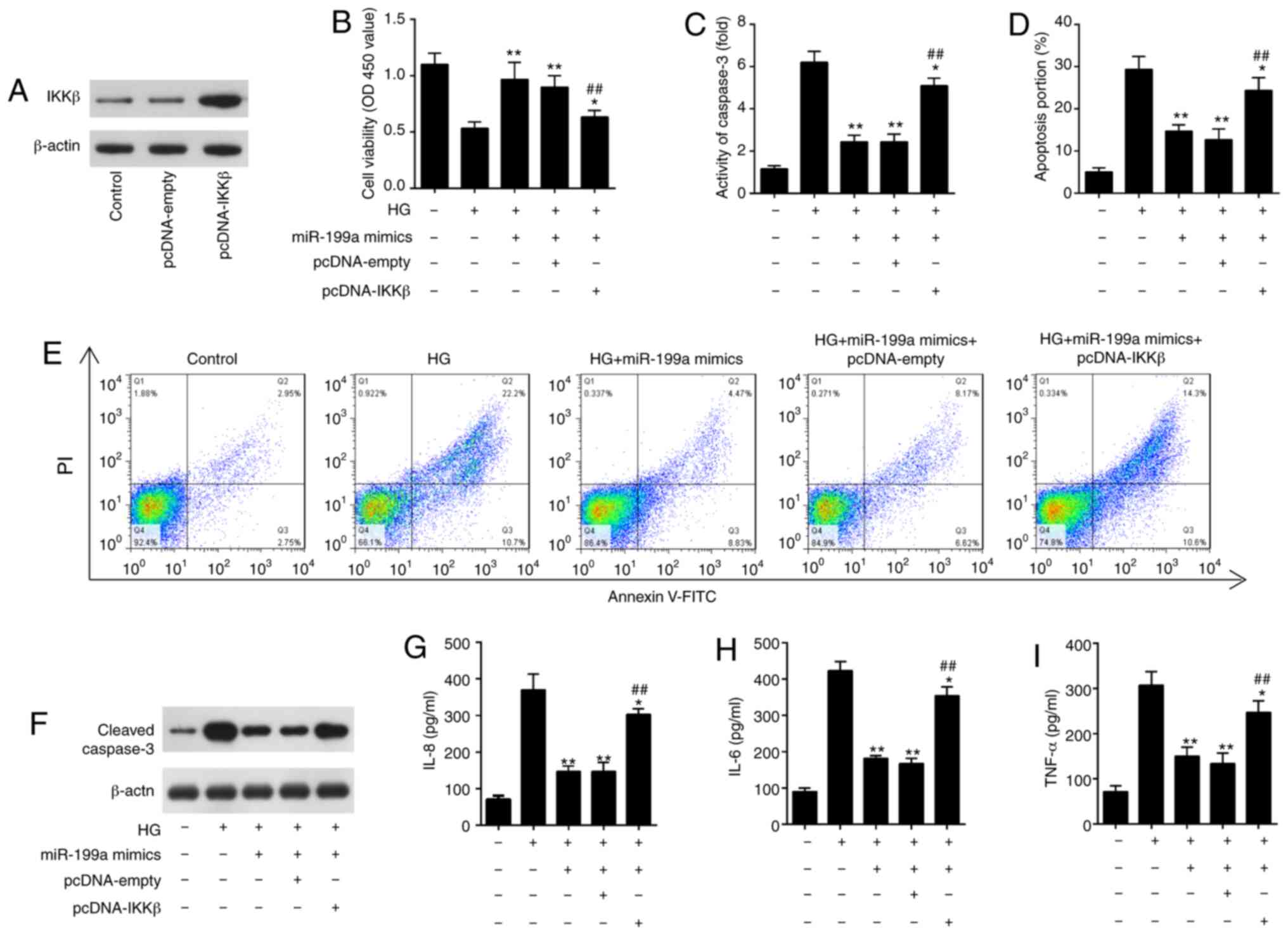
To investigate whether IKKβ was involved in the
protective effects of miR-199a-3p against HG-induced inflammatory
response and apoptosis, HK-2 cells were transfected with pcDNA-IKKβ
plasmid together with miR-199a-3p mimics, followed by HG
stimulation. IKKβ expression was markedly increased in HK-2 cells
after pcDNA-IKKβ transfection, as determined via western blotting
(Fig. 6A). It was demonstrated
that IKKβ overexpression attenuated the increased cell viability
mediated by miR-199a-3p overexpression in HG treated HK-2 cells
(Fig. 6B). It was also found that
overexpression of IKKβ reversed the inhibition of cell apoptosis
and activity of caspase-3 mediated by miR-199a-3p mimics in HG
treated HK-2 (Fig. 6C-E). In
addition, the increased expression of cleaved-caspase-3 caused by
HG was attenuated by miR-199a-3p mimics, which was reversed by
overexpression of IKKβ (Fig. 6F).
The inhibitory effects of miR-199a-3p on HG-induced the releases of
IL-8, IL-6 and TNF-α were also reversed by IKKβ overexpression
(Fig. 6G-I). Collectively, these
data suggested that miR-199a-3p exerted its anti-apoptotic and
anti-inflammatory abilities by targeting IKKβ.
Discussion
In the present study, miR-199a-3p expression was
downregulated in the peripheral blood samples of patients with DN,
and was negatively correlated with the severity of DN. Moreover, it
was observed that miR-199a-3p overexpression inhibited the
apoptosis and inflammatory response by targeting the IKKβ/NF-κB
pathway in vitro. These data suggested that miR-199a-3p may
act as a promising therapeutic target for DN.
Increasing evidence has revealed that miRNAs serve
critical roles in the pathogenesis of DN. For example, Wang et
al (26) reported that
miR-424 overexpression prevented the occurrence and progression of
DN by targeting RPTOR independent companion of MTOR complex 2 in
rats. Moreover, Yang et al (27) observed that miR-214 suppressed
oxidative stress in DN via the reactive oxygen species/Akt/mTOR
signaling pathway in proximal tubular cells. Bai et al
(28) also found that miR-130b
attenuated renal tubulointerstitial fibrosis via the repression of
Snail in HG-treated NRK-52E cells. These studies indicated that
targeting miRNAs may be an effective approach for DN treatment. In
the present study, using a miRNA microarray assay, miR-199a-3p
expression was identified to be downregulated in the peripheral
blood samples of patients with DN. Furthermore, its expression was
negatively correlated with proteinuria in patients with DN. These
findings suggested that miR-199a-3p may serve important role in the
pathogenesis of DN.
miR-199a-3p has been observed to be frequently
down-regulated in several cancerous tissues and to influence
various malignant processes, such as tumor growth, invasion and
metastasis. For example, miR-199a-3p decreases esophageal cancer
cell proliferation by targeting p21 activated kinase 4 (29). Liu et al (30) also found that overexpression of
miR-199a-3p suppressed the cellular proliferation, colony
formation, invasion and migration in clear cell renal cell
carcinoma. However, other studies have reported increased
expression of miR-199a-3p in various cancer types, including
prostate cancer (31) and
hepatocellular carcinoma (32),
which contradicts the tumor suppressive role of this miRNA
(33). In addition, several
studies have demonstrated that miR-199a-3p has protective role in
multiple types of cell injuries. For example, Dai et al
(34) revealed that elevated
miR-199-5p expression disrupted sustained endoplasmic reticulum
stress and prevented hepatocytes from undergoing bile acid- or
thapsigargin-induced cell death. Furthermore, Tao et al
(35) found that miR-199a-3p
promoted cardiomyocyte proliferation by inhibiting CD151
expression. Therefore, targeting miR-199a-3p may be benefit for the
treatment of human diseases, but the precise mechanisms vary in
different cell types.
Previous studies have reported that miR-199a-3p
exerts anti-inflammatory and anti-apoptotic effects in various
diseases (36,37). For example, Liu et al
(38) revealed that miR-199a-3p
decreased pro-inflammatory cytokines expression levels in alveolar
macrophages and septic lung tissues of mice. In addition, Wang
et al (39) observed that
evaluated miR-199a-3p attenuated TNF-α-induced apoptosis in human
nucleus pulposus cells by targeting MAP3K5. It has also been
demonstrated that miR-199a-3p overexpression improves cerebral
ischemic injury via the inhibition of neuronal apoptosis and
suppression of the inflammatory response in rats (40). Notably, miR-199a-3p has previously
been reported to be down-regulated in the urine of patients with DN
(22). Additionally, it has been
revealed that miR-199a-3p exerts anti-apoptotic and
anti-inflammatory roles in various types of cells, such as human
nucleus pulposus cells, kidney cells, hepatocytes, liver sinusoidal
endothelial cells and retinal microglia cells (23–28). miR-199a-3p has been previously
reported to be decreased in the urine from the patients with DN,
which suggests that urine miR-199a-3p could be used as a potential
biomarker for DN (41). However,
to the best of our knowledge, the function of miR-199a-3p in the
apoptosis and inflammatory response of RTEC in DN has not been
previously reported. Thus, the present study aimed to investigate
the effect of miR-199a-3p in DN. Using a HG-induced HK-2 cell
injury model, the present study demonstrated that miR-199a-3p
attenuated HG-induced the apoptosis and release of pro-inflammatory
factors in HK-2 cells. These results suggested that miR-199a-3p
exerted its protective role against HG-induced RTEC injury via the
suppression of apoptosis and the inflammatory response.
The NF-κB pathway serves a pivotal role in
HG-induced injury by regulating the inflammatory response (42–44). With regards to the pathological
progression in DN, Kuhad and Chopra (45) reported that suppression of the
NF-κB pathway using NF-κB inhibitors protected renal functions via
the suppression of the inflammatory response in diabetic rats. Xie
et al (46) have also
shown that Carnosic acid improved DN by inhibiting the NF-κB
pathway in streptozotocin-induced diabetic mice. Moreover, Ohga
et al (47) demonstrated
that thiazolidinedione (a PPAR-γ agonist) treatment ameliorated
renal injury in experimental diabetic rats via anti-inflammatory
effects mediated by inhibiting NF-κB activation. In addition to
being an inflammatory regulator, the NF-κB pathway also controls
the expression levels of key genes involved in apoptosis, such as
Bax and Bcl-2 (48,49). For example, it has been reported
that suppression of the NF-κB pathway contributes to a decrease in
apoptosis induced by HG in HK-2 cells (50). The present findings suggested that
miR-199a-3p overexpression suppressed HG-induced activation of the
NF-κB pathway in HK-2 cells, indicating that miR-199a-3p exerted
its anti-apoptotic and anti-inflammation activities via the
regulation of the NF-κB pathway. However, the mechanism of
miR-199a-3p-mediated regulation of NF-κB is yet to be fully
elucidated.
The NF-κB signaling pathway is one of the most
important pathways mediating the generation of inflammatory
factors, including TNF-α, IL-6 and IL-8, in various types of cells,
such as bronchial epithelial cells, periodontal ligament cells,
endothelial cells and colorectal cancer cells (51–54). In the present study, decreased
TNF-α, IL-6 and IL-8 expression levels were observed after
miR-199a-3p transfection, and were accompanied with reduced NF-κB
activation. These results suggested that miR-199a-3p may function,
at least in part, via the NF-κB pathway to downregulate the
expression levels of inflammatory factors.
IKKβ, one of the catalytic subunits of the IKK
complex, is the major kinase controlling the canonical pathway of
NF-κB activation, in which phosphorylation of IκB by IKK releases
NF-κB to enter nucleus, where it binds to cognate sequences in the
promoter region of multiple genes (55). It has been reported that IKKβ acts
as a downstream molecule of certain miRNAs to mediate the role of
the miRNAs in various cell types, including miR-199a (56). For example, miR-199a-3p suppresses
IKKβ to inhibit NF-κB activity, which reduces the malignancy of
oral squamous cell carcinoma cells (57). Dai et al (58) also revealed that miR-199a
attenuated the invasive capability of endometrial stromal cells via
the suppression of IKKβ, which inhibited the activation of the
NF-κB pathway. Moreover, Chen et al (59) reported that miR-199a could affect
NF-κB activity in ovarian cancer cells by targeting IKKβ. Previous
studies have also shown that miR-199a inhibits the activation of
the NF-κB signaling pathway in cancer cells and renal cells
(59,60). Therefore, it was suggested that
miR-199a-3p regulates the NF-κB pathway in DN. In the current
study, IKKβ was identified to be targeted by miR-199a-3p. Further
experimental results indicated that IKKβ expression in HK-2 cells
was upregulated, and was negatively correlated with miR-199a-3p in
patients with DN. It was demonstrated that IKKβ overexpression
alleviated the inhibitory effects of miR-199a-3p overexpression on
inflammation and apoptosis in HG-treated HK-2 cells. Collectively,
the present findings indicated that miR-199a-3p exerted its
anti-inflammatory and anti-apoptotic effects via the IKKβ/NF-κB
pathway in HG-treated HK-2 cells.
However, there are limitations to the present study.
DN is a complex pathological process involving numerous miRNAs and
target genes. The main limitation of this study is the sole focus
on the IKKβ/NF-κB pathway. The underlying relationship between this
pathway and other related pathways requires further
investigations.
In conclusion, the current findings demonstrated
that the miR-199a-3p was downregulated in patients with DN, and
miR-199a-3p overexpression improved HG-induced inflammation and
apoptosis by blocking the IKKβ/NF-κB pathway. These findings
support the hypothesis that enhanced miR-199a-3p expression may
serve as a novel therapeutic approach for the treatment of DN.
Acknowledgments
Not applicable.
Funding
No funding was received.
Availability of data and materials
The miRNA microarray datasets generated and/or
analyzed during the current study are not publicly available due to
other research on the microarray that is under way, but are
available from the corresponding author on reasonable request.
Other data generated or analyzed during the present study are
included in this published article.
Authors' contributions
Conceived and designed the experiments: JS.
Performed the experiments: RZ and LQ. Analyzed the data: RZ and LQ.
Contributed reagents, materials and analysis tools: JS. Wrote the
paper: RZ and JS. All authors have read and approved the final
version of manuscript.
Ethics approval and consent to
participate
All individuals provided informed consent for the
use of human specimens for clinical research. The present study was
approved by the Huaihe Hospital of Henan University Ethics
Committees.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Umanath K and Lewis JB: Update on diabetic
nephropathy: Core curriculum 2018. Am J Kidney Dis. 71:884–895.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Verzola D, Gandolfo MT, Ferrario F,
Rastaldi MP, Villaggio B, Gianiorio F, Giannoni M, Rimoldi L,
Lauria F, Miji M, et al: Apoptosis in the kidneys of patients with
type II diabetic nephropathy. Kidney Int. 72:1262–1272. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Magri CJ and Fava S: The role of tubular
injury in diabetic nephropathy. Eur J Intern Med. 20:551–555. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li T and Cho WC: MicroRNAs: Mechanisms,
functions and progress. Genomics Proteomics Bioinformatics.
10:237–238. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhu FX, Wu HL, Chen JX, Han B and Guo YF:
Dysregulation of microRNA-181b and TIMP3 is functionally involved
in the pathogenesis of diabetic nephropathy. J Cell Physiol.
234:18963–18969. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bhatt K, Lanting LL, Jia Y, Yadav S, Reddy
MA, Magilnick N, Boldin M and Natarajan R: Anti-inflammatory role
of MicroRNA-146a in the pathogenesis of diabetic nephropathy. J Am
Soc Nephrol. 27:2277–2288. 2016. View Article : Google Scholar :
|
|
7
|
Deshpande SD, Putta S, Wang M, Lai JY,
Bitzer M, Nelson RG, Lanting LL, Kato M and Natarajan R:
Transforming growth factor-β-induced cross talk between p53 and a
microRNA in the pathogenesis of diabetic nephropathy. Diabetes.
62:3151–3162. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Long J, Wang Y, Wang W, Chang BH and
Danesh FR: Identification of microRNA-93 as a novel regulator of
vascular endothelial growth factor in hyperglycemic conditions. J
Biol Chem. 285:23457–23465. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang J, Gao Y, Ma M, Li M, Zou D, Yang J,
Zhu Z and Zhao X: Effect of miR-21 on renal fibrosis by regulating
MMP-9 and TIMP1 in kk-ay diabetic nephropathy mice. Cell Biochem
Biophys. 67:537–546. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li H, Zhu X, Zhang J and Shi J:
MicroRNA-25 inhibits high glucose-induced apoptosis in renal
tubular epithelial cells via PTEN/AKT pathway. Biomed Pharmacother.
96:471–479. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li X, Zeng L, Cao C, Lu C, Lian W, Han J,
Zhang X, Zhang J, Tang T and Li M: Long noncoding RNA MALAT1
regulates renal tubular epithelial pyroptosis by modulated miR-23c
targeting of ELAVL1 in diabetic nephropathy. Exp Cell Res.
350:327–335. 2017. View Article : Google Scholar
|
|
12
|
Lau GJ, Godin N, Maachi H, Lo CS, Wu SJ,
Zhu JX, Brezniceanu ML, Chénier I, Fragasso-Marquis J, Lattouf JB,
et al: Bcl-2-modifying factor induces renal proximal tubular cell
apoptosis in diabetic mice. Diabetes. 61:474–484. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang YF, Zhang Y, Liu CX, Huang J and
Ding GH: microRNA-125b contributes to high glucose-induced reactive
oxygen species generation and apoptosis in HK-2 renal tubular
epithelial cells by targeting angiotensin-converting enzyme 2. Eur
Rev Med Pharmacol Sci. 20:4055–4062. 2016.PubMed/NCBI
|
|
14
|
Su J, Ren J, Chen H and Liu B:
MicroRNA-140-5p ameliorates the high glucose-induced apoptosis and
inflammation through suppressing TLR4/NF-κB signaling pathway in
human renal tubular epithelial cells. Biosci Rep.
40:BSR201923842020. View Article : Google Scholar
|
|
15
|
Lv L, Zhang J, Tian F, Li X, Li D and Yu
X: Arbutin protects HK-2 cells against high glucose-induced
apoptosis and autophagy by up-regulating microRNA-27a. Artif Cells
Nanomed Biotechnol. 47:2940–2947. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang GJ, Jiao BP, Liu YJ, Li YR and Deng
BB: Reactivation of microRNA-506 inhibits gastric carcinoma cell
metastasis through ZEB2. Aging (Albany NY). 11:1821–1831. 2019.
View Article : Google Scholar
|
|
17
|
Kohl M, Wiese S and Warscheid B:
Cytoscape: Software for visualization and analysis of biological
networks. Methods Mol Biol. 696:291–303. 2011. View Article : Google Scholar
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
19
|
Ding Y, Wang L, Zhao Q, Wu Z and Kong L:
MicroRNA93 inhibits chondrocyte apoptosis and inflammation in
osteoarthritis by targeting the TLR4/NFkB signaling pathway. Int J
Mol Med. 43:779–790. 2019.
|
|
20
|
Wang X, Shen E, Wang Y, Li J, Cheng D,
Chen Y, Gui D and Wang N: Cross talk between miR-214 and PTEN
attenuates glomerular hypertrophy under diabetic conditions. Sci
Rep. 6:315062016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zha F, Bai L, Tang B, Li J, Wang Y, Zheng
PX, Ji T and Bai S: MicroRNA-503 contributes to podocyte injury via
targeting E2F3 in diabetic nephropathy. J Cell Biochem.
120:12574–12581. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen YY, Peng XF, Liu GY, Liu JS, Sun L,
Liu H, Xiao L and He LY: Protein arginine methyltranferase-1
induces ER stress and epithelial-mesenchymal transition in renal
tubular epithelial cells and contributes to diabetic nephropathy.
Biochim Biophys Acta Mol Basis Dis. 1865:2563–2575. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ni WJ, Tang LQ and Wei W: Research
progress in signalling pathway in diabetic nephropathy. Diabetes
Metab Res Rev. 31:221–233. 2015. View Article : Google Scholar
|
|
24
|
Yu C, Qi D, Sun JF, Li P and Fan HY: Rhein
prevents endotoxin-induced acute kidney injury by inhibiting NF-κB
activities. Sci Rep. 5:118222015. View Article : Google Scholar
|
|
25
|
Yang X, Wang Y and Gao G: High glucose
induces rat mesangial cells proliferation and MCP-1 expression via
ROS-mediated activation of NF-κB pathway, which is inhibited by
eleutheroside E. J Recept Signal Transduct Res. 36:152–157. 2016.
View Article : Google Scholar
|
|
26
|
Wang G, Yan Y, Xu N, Hui Y and Yin D:
Upregulation of microRNA-424 relieved diabetic nephropathy by
targeting rictor through mTOR complex2/protein kinase B signaling.
J Cell Physiol. 234:11646–11653. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang S, Fei X, Lu Y, Xu B, Ma Y and Wan H:
miRNA-214 suppresses oxidative stress in diabetic nephropathy via
the ROS/Akt/mTOR signaling pathway and uncoupling protein 2. Exp
Ther Med. 17:3530–3538. 2019.PubMed/NCBI
|
|
28
|
Bai X, Geng J, Zhou Z, Tian J and Li X:
MicroRNA-130b improves renal tubulointerstitial fibrosis via
repression of Snail-induced epithelial-mesenchymal transition in
diabetic nephropathy. Sci Rep. 6:204752016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Phatak P, Burrows WM, Chesnick IE,
Tulapurkar ME, Rao JN, Turner DJ, Hamburger AW, Wang JY and Donahue
JM: MiR-199a-3p decreases esophageal cancer cell proliferation by
targeting p21 activated kinase 4. Oncotarget. 9:28391–28407. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liu J, Liu B, Guo Y, Chen Z, Sun W, Gao W,
Wu H and Wang Y: MiR-199a-3p acts as a tumor suppressor in clear
cell renal cell carcinoma. Pathol Res Pract. 214:806–813. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Qu F, Zheng J, Gan W, Lian H, He H, Li W,
Yuan T, Yang Y, Li X, Ji C, et al: MiR-199a-3p suppresses
proliferation and invasion of prostate cancer cells by targeting
Smad1. Oncotarget. 8:52465–52473. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Callegari E, D'Abundo L, Guerriero P,
Simioni C, Elamin BK, Russo M, Cani A, Bassi C, Zagatti B,
Giacomelli L, et al: miR-199a-3p modulates MTOR and PAK4 pathways
and inhibits tumor growth in a hepatocellular carcinoma transgenic
mouse model. Mol Ther Nucleic Acids. 11:485–493. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li L, Mou YP, Wang YY, Wang HJ and Mou XZ:
miR-199a-3p targets ETNK1 to promote invasion and migration in
gastric cancer cells and is associated with poor prognosis. Pathol
Res Pract. 215:1525112019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Dai BH, Geng L, Wang Y, Sui CJ, Xie F,
Shen RX, Shen WF and Yang JM: microRNA-199a-5p protects hepatocytes
from bile acid-induced sustained endoplasmic reticulum stress. Cell
Death Dis. 4:e6042013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tao Y, Zhang H, Huang S, Pei L, Feng M,
Zhao X, Ouyang Z, Yao S, Jiang R and Wei K: miR-199a-3p promotes
cardiomyocyte proliferation by inhibiting Cd151 expression. Biochem
Biophys Res Commun. 516:28–36. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rane S, He M, Sayed D, Vashistha H,
Malhotra A, Sadoshima J, Vatner DE, Vatner SF and Abdellatif M:
Downregulation of miR-199a derepresses hypoxia-inducible
factor-1alpha and Sirtuin 1 and recapitulates hypoxia
preconditioning in cardiac myocytes. Circ Res. 104:879–886. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zuo Y, Wang Y, Hu H and Cui W:
Atorvastatin protects myocardium against ischemia-reperfusion
injury through inhibiting miR-199a-5p. Cell Physiol Biochem.
39:1021–1030. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu Y, Guan H, Zhang JL, Zheng Z, Wang HT,
Tao K, Han SC, Su LL and Hu D: Acute downregulation of miR-199a
attenuates sepsis-induced acute lung injury by targeting SIRT1. Am
J Physiol Cell Physiol. 314:C449–C455. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang W, Guo Z, Yang S, Wang H and Ding W:
Upregulation of miR-199 attenuates TNF-α-induced human nucleus
pulposus cell apoptosis by downregulating MAP3K5. Biochem Biophys
Res Commun. 505:917–924. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li M, Luan L, Liu Q, Liu Y, Lan X, Li Z
and Liu W: MiRNA-199a-5p protects against cerebral ischemic injury
by down-regulating DDR1 in rats. World Neurosurg. 131:e486–e494.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Meng L, Li G, Liu X, Jiang J, Zhu M and
Sun Y: Decreased urine miR-199-3p may be a potential biomarker for
diabetic nephropathy via targeting zinc finger e-box-bindingprotein
1. Clin Lab. 64:1177–1182. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gu J, Huang W, Zhang W, Zhao T, Gao C, Gan
W, Rao M, Chen Q, Guo M, Xu Y and Xu YH: Sodium butyrate alleviates
high-glucose-induced renal glomerular endothelial cells damage via
inhibiting pyroptosis. Int Immunopharmacol. 75:1058322019.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kang Z, Zeng J, Zhang T, Lin S, Gao J,
Jiang C, Fan R and Yin D: Hyperglycemia induces NF-κB activation
and MCP-1 expression via downregulating GLP-1R expression in rat
mesangial cells: Inhibition by metformin. Cell Biol Int.
43:940–953. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li J, Tang Y and Cai D: IKKβ/NF-κB
disrupts adult hypo-thalamic neural stem cells to mediate a
neurodegenerative mechanism of dietary obesity and pre-diabetes.
Nat Cell Biol. 14:999–1012. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kuhad A and Chopra K: Attenuation of
diabetic nephropathy by tocotrienol: Involvement of NFkB signaling
pathway. Life Sci. 84:296–301. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Xie Z, Zhong L, Wu Y, Wan X, Yang H, Xu X
and Li P: Carnosic acid improves diabetic nephropathy by activating
Nrf2/ARE and inhibition of NF-κB pathway. Phytomedicine.
47:161–173. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ohga S, Shikata K, Yozai K, Okada S, Ogawa
D, Usui H, Wada J, Shikata Y and Makino H: Thiazolidinedione
ameliorates renal injury in experimental diabetic rats through
anti-inflammatory effects mediated by inhibition of NF-kappaB
activation. Am J Physiol Renal Physiol. 292:F1141–F1150. 2007.
View Article : Google Scholar
|
|
48
|
Leibowitz B and Yu J: Mitochondrial
signaling in cell death via the Bcl-2 family. Cancer Biol Ther.
9:417–422. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Nam MS, Jung DB, Seo KH, Kim BI, Kim JH,
Kim JH, Kim B, Baek NI and Kim SH: Apoptotic effect of sanggenol L
via caspase activation and inhibition of NF-κB signaling in ovarian
cancer cells. Phytother Res. 30:90–96. 2016. View Article : Google Scholar
|
|
50
|
Lu J, Peng J, Xiang M, He L, Wang D, Xiong
G and Li S: Trichosanthes kirilowii lectin alleviates diabetic
nephropathy by inhibiting the LOX1/NF-κB/caspase-9 signaling
pathway. Biosci Rep. 38:BSR201800712018. View Article : Google Scholar
|
|
51
|
Abolhassani M, Aloulou N, Chaumette MT,
Aparicio T, Martin-Garcia N, Mansour H, Gouvello SL, Delchier JC
and Sobhani I: Leptin receptor-related immune response in
colorectal tumors: The role of colonocytes and interleukin-8.
Cancer Res. 68:9423–9432. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Fang Y, Shi C, Manduchi E, Civelek M and
Davies PF: MicroRNA-10a regulation of proinflammatory phenotype in
athero-susceptible endothelium in vivo and in vitro. Proc Natl Acad
Sci USA. 107:13450–13455. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kanoh S, Tanabe T and Rubin BK: Dapsone
inhibits IL-8 secretion from human bronchial epithelial cells
stimulated with lipopolysaccharide and resolves airway inflammation
in the ferret. Chest. 140:980–990. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Chi XP, Ouyang XY and Wang YX: Hydrogen
sulfide synergistically upregulates porphyromonas gingivalis
lipopoly-saccharide-induced expression of IL-6 and IL-8 via NF-κB
signalling in periodontal fibroblasts. Arch Oral Biol. 59:954–961.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Caamaño J and Hunter CA: NF-kappaB family
of transcription factors: Central regulators of innate and adaptive
immune functions. Clin Microbiol Rev. 15:414–429. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Bardin P, Marchal-Duval E, Sonneville F,
Blouquit-Laye S, Rousselet N, Rouzic PL, Corvol H and Tabary O:
Small RNA and transcriptome sequencing reveal the role of
miR-199a-3p in inflammatory processes in cystic fibrosis airways. J
Pathol. 245:410–420. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wei D, Shen B, Wang W, Zhou Y, Yang X, Lu
G, Yang J and Shao Y: MicroRNA199a5p functions as a tumor
suppressor in oral squamous cell carcinoma via targeting the
IKKβ/NFkB signaling pathway. Int J Mol Med. 43:1585–1596.
2019.PubMed/NCBI
|
|
58
|
Dai L, Gu L and Di W: MiR-199a attenuates
endometrial stromal cell invasiveness through suppression of the
IKKβ/NF-κB pathway and reduced interleukin-8 expression. Mol Hum
Reprod. 18:136–145. 2012. View Article : Google Scholar
|
|
59
|
Chen R, Alvero AB, Silasi DA, Kelly MG,
Fest S, Visintin I, Leiser A, Schwartz PE, Rutherford T and Mor G:
Regulation of IKKbeta by miR-199a affects NF-kappaB activity in
ovarian cancer cells. Oncogene. 27:4712–4723. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Wu C, Lv C, Chen F, Ma X, Shao Y and Wang
Q: The function of miR-199a-5p/Klotho regulating TLR4/NF-κB
p65/NGAL pathways in rat mesangial cells cultured with high glucose
and the mechanism. Mol Cell Endocrinol. 417:84–93. 2015. View Article : Google Scholar : PubMed/NCBI
|