Introduction
Angiogenesis plays an important role in the recovery
of the microvascular bed and prevents further apoptosis and
necrosis of ischemic myocardial tissues, which may be injured,
stunned or hibernating post-myocardial infarction (MI) (1). Therefore, angiogenesis is a key
factor in the recovery of the ischemic myocardium and myocardial
remodeling in the early stages of MI (1,2).
Previous studies have demonstrated that infiltration by dendritic
cells (DCs) is significantly increased in the infarcted myocardium
(3-5), while angiogenesis is impaired and
cardiac function is deteriorated in DC ablation mice following MI
(6). These results suggest that
DCs play an important role in angio-genesis and cardiac healing
after MI. However, to the best of our knowledge, the mechanism
underlying the effects of DCs on MI has yet to be fully
elucidated.
Exosomes (EXs) are small double-membrane vesicles
(30-100 nm) containing a wide range of functional proteins, mRNAs
and microRNAs (miRNAs or miRs), which are secreted via exocytosis
by different types of cells, including DCs, lymphocytes and
platelets, as well as cells originating from other tissues, under
physiological or pathological conditions (7-9).
Previous studies have demonstrated that EXs may act as mediators of
cell-to-cell communication and play a key role in cardiac
remodeling after MI (5,10). Furthermore, it was observed in our
previous study that EXs derived from DCs (DEXs) may improve cardiac
function after MI (10). However,
to the best of our knowledge, whether DEXs improve cardiac function
by promoting angiogenesis via paracrine signaling remains
unknown.
miRNAs are short, non-coding RNAs that regulate gene
expression through translational repression or degradation of
target mRNAs (9). Over the last
decade, miRNAs have emerged as key regulators of several cellular
processes, including angiogenesis, and certain miRNAs, such as
miR-132, miR-126, miR-451a, miR-494-3p, miR-16a-5p and miR-23a-3p,
have been shown to play key roles in the vasculature in both
endothelial and perivascular cells (11,12). In addition, previous studies have
demonstrated that EXs contain a large number of miRNAs, and EXs may
deliver these miRNAs to their target cells (13-15). It was previously reported that DC
infiltration is significantly increased in the infarcted myocardium
and plays an important role in angiogenesis and cardiac healing
after MI (6). Furthermore, our
previous study demonstrated that DEXs also contain abundant miRNAs
that are associated with angiogenesis (16). Therefore, it may be inferred that
the DCs infiltrating the infarcted myocardium may promote
angiogenesis via secretion of exosomal miRNAs post-MI.
The aim of the present study was to investigate the
expression and enrichment of miR-494-3p in EXs secreted from DCs
post-MI, and determine whether it can promote angiogenesis in
vitro and in vivo.
Materials and methods
Study design
An outline of the design of the present study is
shown in Fig. 1. In brief,
C57BL/6 mice served as donors for the culture of bone
marrow-derived DCs (BMDCs) and as MI models. Then, samples were
collected from the supernatants of necrotic HL-1 cells, which were
incubated with BMDCs for 24 h as the MI group. Samples from the
supernatants of normal cells served as the control group, and from
the culture medium of BMDCs as the negative group. Subsequently,
DEXs from different groups were isolated and added to primary
cultures of rat cardiac microvascular endothelial cells (CMECs),
confocal microscopy was performed to observe the interaction of
CMECs with DEXs, and angiogenesis was evaluated through measuring
tube formation and vascular endothelial growth factor (VEGF)
expression. For the in vivo experiments, DEXs from different
groups were injected into the infarcted myocardium of MI model
mice, and angiogenesis was evaluated by measuring the expression of
VEGF and CD31 in the infarcted myocardium and the mean number of
CD31-positive capillaries were counted in the infarcted area under
an optical microscope. Finally, the expression profile of miRNAs in
splenic DCs of MI model mice was analyzed by Affymetrix miRNA 4.0
chip assays, and those miRNAs associated with angiogenesis that
were significantly upregulated in the Affymetrix miRNA 4.0 chip
were also certified in DCs and DEXs by reverse
transcription-quantitative PCR (RT-qPCR) analysis. Subsequently, it
was evaluated whether the significantly upregulated and highly
enriched DEX-miR-494-3p could enhance tube formation in
vitro and angiogenesis in vivo.
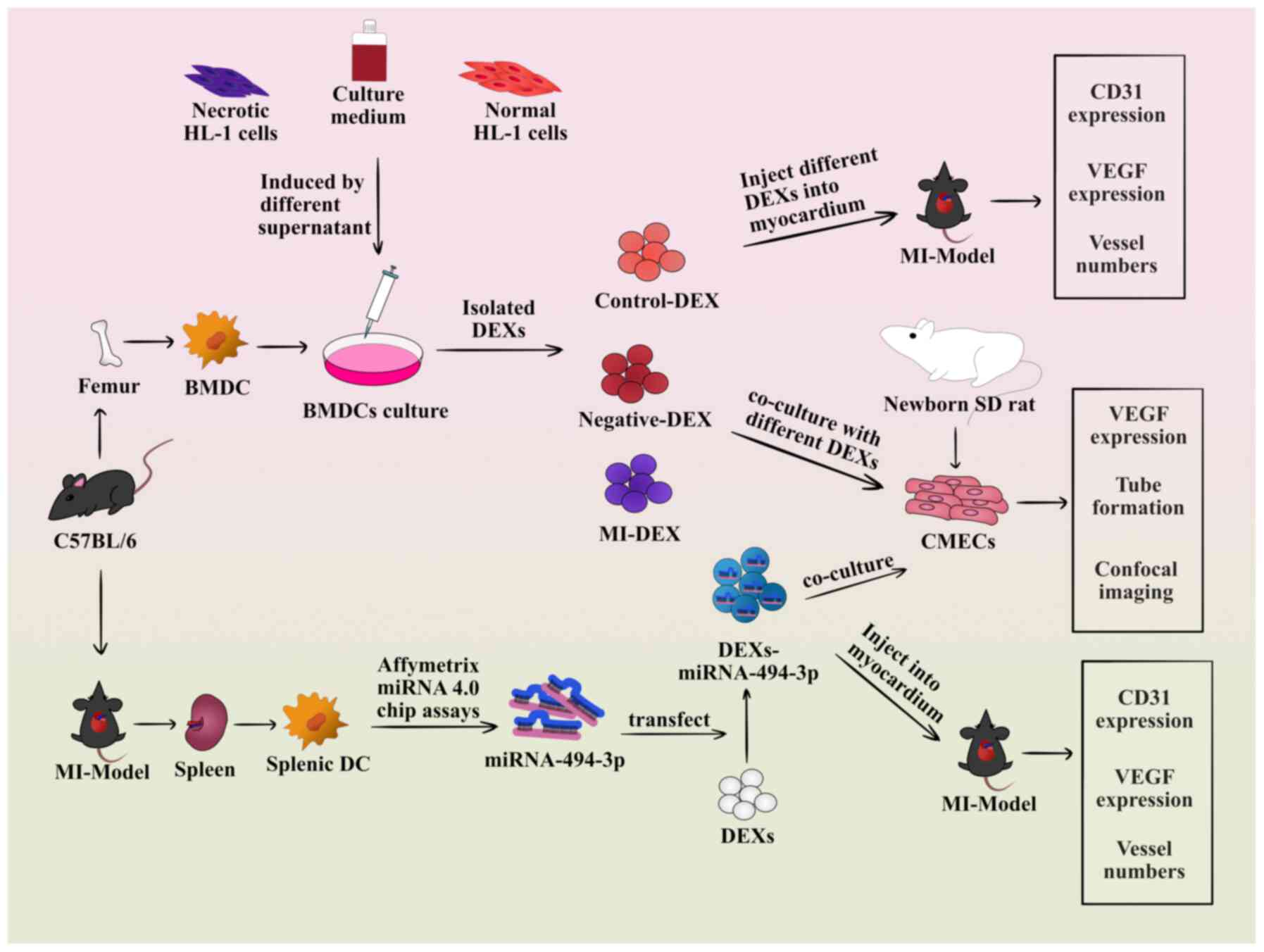 | Figure 1Flowchart of the design of the
present study. In brief, C57BL/6 mice served as donors for the
culture of BMDCs and as MI models. Samples were collected from the
supernatants of necrotic HL-1 cells, which were incubated with
BMDCs for 24 h as the MI group. Samples from the supernatants of
normal cells served as the control group, and samples from the
culture medium of BMDCs served as the negative group. Subsequently,
DEXs from different groups were isolated and added to primary
cultures of rat CMECs, followed by angiogenesis evaluation through
measuring tube formation and VEGF expression. Confocal microscopy
was performed to observe the interaction of CMECs with DEXs. For
the in vivo experiments, DEXs from different groups were
injected into the infarcted myocardium of MI model mice, and
angiogenesis was evaluated by measuring the expressions of VEGF and
CD31 in the infarcted myocardium and the mean number of vessel
counts in the infarcted area. Finally, the expression profile of
miRNAs in splenic DCs of MI model mice was analyzed by Affymetrix
miRNA 4.0 chip assays, and those miRNAs associated with
angiogenesis that were significantly upregulated in the Affymetrix
miRNA 4.0 chip were also certified in DCs and DEXs by reverse
transcription-quantitative PCR analysis. Subsequently, it was
evaluated whether the significantly upregulated and highly enriched
miR-494-3p could enhance tube formation in vitro and
angiogenesis in vivo. DEXs, dendritic cell-derived exosomes;
BMDC, bone marrow-derived dendritic cell; MI, myocardial
infarction; DC, dendritic cell; SD, Sprague-Dawley; miRNA-494-3p,
microRNA-494-3p; CMECs, cardiac microvascular endothelial cells;
VEGF, vascular endothelial growth factor. |
Animals
Male, wild-type mice (C57BL/6) and newborn male
Sprague-Dawley rats were purchased from the Shanghai Laboratory
Animal Center. Male, 6-week-old C57BL/6 mice (weight, 16-20 g;
n=30) were used for culture of BMDCs and the newborn rats (age, 1-2
days; weight, 5-6 g; n=20) were used for culture of CMECs. In
addition, 8-week-old male C57BL/6 mice (weight, 22-28 g, n=30) were
used to construct the MI models and were subjected to DEX
injections. All the mice and rats were housed under pathogen-free
conditions in a standard laboratory with a controlled room
temperature (22±1°C) and humidity (65-70%), and a 12:12-h
light-dark cycle, with free access to food and water. Animal care
and treatment complied with the standards approved by the
Institutional Review Board of Zhongshan Hospital of Fudan
University and the Shanghai Institutes of Biological Sciences-CAS
(A5894-01).
Culture of BMDCs and CMECs
BMDCs were isolated from C57BL/6 mice. BMDCs were
cultured with a non-serum medium (X-VIVO 15; Lonza Group, Ltd.) to
eliminate the interference of exosomes from FBS, as described
previously (17). Briefly, the
mice were sacrificed by cervical dislocation and the femurs were
isolated. The bone marrow cells were washed out from the femurs and
cultured in non-serum medium containing 10 ng/ml
granulocyte-macrophage colony-stimulating factor and 1 ng/ml IL-4
(both from R&D Systems, Inc.). The medium was changed every
other day. On day 7, the BMDCs were collected and treated as
previously described (17). CMECs
were isolated from newborn rats. Briefly, the rats were sacrificed
by cervical dislocation and immersed in 75% ethanol for 10 min.
Then, the hearts were excised and rinsed with 4°C Hank's Balanced
Salt Solution (HyClone; Cytiva), followed by resecting the left
ventricular tissue and removing the epicardium and endocardium. The
remaining tissues were cut into 1-mm3 pieces and
inoculated into 10-cm culture dishes with 1 ml FBS (Gibco; Thermo
Fisher Scientific, Inc.). The tissues were then cultured in a 37°C,
5% CO2 incubator for 4 h, and for a further 72 h in
high-glucose DMEM (Invitrogen; Thermo Fisher Scientific, Inc.)
supplemented with 20% FBS. When the endothelial cells reached a
confluence of 90%, the tissues were removed carefully and digested
with 0.125% trypsin (Gibco; Thermo Fisher Scientific, Inc.). The
cells were transferred to another culture dish and the second
generation of cells were used for subsequent experiments. All the
experiments were performed under sterile conditions.
Simulation of post-MI cardiomyocyte
microenvironment in vitro
Three types of samples were used to mimic the MI
microenvironment: i) The supernatants of necrotic HL-1 cells; ii)
the supernatants of hypoxic primary cardiomyocytes; and iii) the
supernatants of infarcted mouse myocardium, which have been
described in detail in our previous study (10). The results of the previous study
suggested that treatment with the supernatants of necrotic HL-1
cells or hypoxic primary cardiomyocytes in vitro activates
DCs in mice to a similar extent as MI. In the present study, the
supernatants of necrotic HL-1 cells were used to mimic the MI
microenvironment. In brief, 100-µl samples, from which the
cell membrane particles were removed by centrifugation at 1,500 × g
at room temperature for 30 min, were collected from the
supernatants of the necrotic HL-1 cells (MI group) and added to
1×106 BMDCs for 24 h. The same conditions were used with
the supernatant from normal cells (control group) or 100 µl
of culture medium from the culture of DCs (negative group). All the
experiments were performed under sterile conditions.
DEX isolation
The ExoQuick-TC™ Exosome Precipitation Solution
(System Biosciences Inc.) was used to isolate DEXs, according to
the protocol recommended by the manufacturer. Briefly, 1/5 volume
of ExoQuick-TC™ Exosome Precipitation Solution was added to 10 ml
supernatant of DCs and refrigerated overnight at 4°C. Then, the
samples were centrifuged at 1,500 × g at room temperature for 30
min and the supernatant was removed. The DEXs were re-suspended in
1 ml PBS (HyClone; Cytiva). DEXs were divided into 3 groups as
follows: i) MI DEX group: DEXs isolated from BMDCs stimulated by
the supernatant of necrotic HL-1 cells; ii) control DEX group: DEXs
isolated from BMDCs stimulated by the supernatant of normal HL-1
cells; and iii) negative DEX group: DEXs isolated by adding the
same amount of BMDC medium. The details were described in our
previous study (10).
Interaction of CMECs with DEXs by
confocal laser scanning microscopy
DEXs were labeled with PKH67 at room temperature for
10 min (green; Sigma-Aldrich; Merck KGaA) and co-cultured with
CMECs, and confocal laser scanning micros-copy was used to observe
the interaction between CMECs and DEXs. Briefly, 25,000 CMECs were
co-cultured with 100 µl PKH67-labeled DEXs for 24 h. Then,
the supernatant was discarded, followed by washing twice with PBS,
fixation for 10 min at room temperature with 4% paraformaldehyde
(Sangon Biotech Co., Ltd.) and staining for 15 min at room
temperature with phalloidin-TRITC (Thermo Fisher Scientific, Inc.)
and DAPI (Invitrogen; Thermo Fisher Scientific, Inc.).
Subsequently, the cells were observed using a confocal laser
scanning microscope (magnification, ×630; Leica TCS SP8; Leica
Microsystems GmbH; 40× or 60×/1.30 NA HCX PL APO CS OIL objective
lens) and images were captured using the Leica application suite
Advanced Fluorescence Lite and analyzed using LAS AF Lite V4.3.1
software (National Institutes of Health).
Tube formation assay
Matrigel™ basement membrane matrix (growth factor
reduced) (BD Biosciences) was thawed at 4°C overnight, and then
transferred into precooled 24-well plates and incubated at 37°C for
30 min. Subsequently, CMECs, which had been divided into four
groups [unstimulated group, negative DEX group (addition of
negative DEXs, 100 µl), control DEX group (addition of
control DEXs, 100 µl) and MI DEX group (addition of MI DEXs,
100 µl)], were seeded onto the 24 well-plates (25,000 cells
per well, three wells per group), then cultured in a 37°C, 5%
CO2 incubator with 1% FBS-DMEM. Finally, images were
captured at 3, 6, 9, 12 and 24 h using the BH-2 inverted optical
microscope (magnification, ×50; Olympus Corporation) for the
enclosed networks of complete tubes. The tube formation images were
analyzed by the WimTube Image Analysis V 7.02 software (Ibidi
GmbH).
Detection of VEGF expression in CMECs in
vitro
After co-culturing negative, control and MI DEXs
with CMECs for 24 h, the expression of VEGF in CMECs was detected
by western blotting. Briefly, CMECs were collected and lysed in
RIPA lysis buffer (Sigma-Aldrich; Merck KGaA) containing 1%
protease inhibitors (cOmplete Protease Inhibitor Cocktail; Roche
Diagnostic, Inc.). Following centrifugation at 14,000 × g for 20
min at 4°C and discarding the sediment, the lysates were loaded in
SDS buffer containing 50 mM DTT (Invitrogen; Thermo Fisher
Scientific, Inc.) and heated at 100°C for 5 min. Then, proteins (10
µg/lane) were separated by 12% SDS-PAGE (Sigma-Aldrich;
Merck KGaA) and trans-ferred to PVDF membranes (EMD Millipore).
After blocking with 5% BSA (Sigma-Aldrich; Merck KGaA) for 2 h at
room temperature, the membranes were incubated with mouse anti-VEGF
monoclonal antibody (Abcam; cat. no. ab69479; 1:500) or mouse
anti-GAPDH (Abcam; cat. no. ab8245; 1:1,000) overnight at 4°C.
After the membranes were washed with TBST (Sigma-Aldrich; Merck
KGaA) three times, they were incubated with goat anti-mouse IgG
horseradish peroxidase-conjugated secondary monoclonal antibody
(Abcam; cat. no. ab205719; 1:2,000) at room temperature for 2 h,
followed by washing in TBST. Finally, the membranes were exposed
using the LAS-3000 imager system (Fujifilm Corporation), and the
image analysis and the densitometric analysis to semi-quantify the
protein expression data were performed using Quantity One V 4.6.6
software (Bio-Rad Laboratories, Inc.).
MI model induction and injection of
DEXs
Coronary artery ligation was used to induce the MI
model (18). Briefly, mice were
anaesthetized by 2% isoflurane inhalation (RWD Life Science) with
an isoflurane delivery system (Harvard Bioscience, Inc.). A small
incision was made on the skin of the left chest, followed by
dissecting and retracting the major and minor pectoral muscles.
Next, a clamp was used to create a small hole in the 4th
intercostal space and open the pleural membrane. The clamp was then
opened slightly and the heart was extracted. The left coronary
artery (LCA) was ligated with a 6-0 silk suture and the heart was
immediately placed back into the intrathoracic cavity. Finally, the
air was evacuated and the skin was sutured. Mice undergoing the
same surgical procedure, but without LCA ligation, were used as the
sham group. After successfully constructing the MI model, 10
µg negative, control or MI DEXs were injected into the left
ventricle in five sites around the infarct area, as described
previously (19).
Detection of the expression of VEGF and
CD31 in the infarcted myocardium
On day 7, MI model mice were sacrificed by cervical
dislocation and the expression of VEGF and CD31 was determined by
immunohistochemistry, as previously described (19). Briefly, tissue sections from heart
samples were fixed with 4% paraformaldehyde at room temperature for
24-48 h. Then, paraffin-embedded murine heart samples were cut into
5-µm-thick tissue sections, and dewaxed in xylene (Sangon
Biotech Co., Ltd.) and rehydrated using ethanol. Then, antigen
retrieval was performed with boiling 0.01 M citrate buffer (Sangon
Biotech Co., Ltd.) and, after using 3% hydrogen peroxide (Sangon
Biotech Co., Ltd.) to eliminate the endogenous peroxidase activity,
the sections were incubated with goat serum blocking solution
(Abcam) for 15 min at room temperature. Next, the sections were
incubated overnight at 4°C with anti-CD31 (Abcam; cat. no. ab9498;
1:2,000) and anti-VEGF (Abcam; cat. no. ab1316; 1:200). After
washing three times with PBS, sections were incubated with
horseradish peroxidase-labeled goat anti-rabbit IgG (Abcam; cat.
no. ab5879; 1:500) for 15 min at 37°C. After DAB color reaction
(using the DAB Horseradish Peroxidase Color Development Kit; Sangon
Biotech Co., Ltd.) and hematoxylin (Sangon Biotech Co., Ltd.)
counterstaining for 1 min at room temperature, the sections were
sealed with neutral gum and observed under an optical microscope
(magnification, ×400; Olympus Corporation).
miRNA expression profile in splenic DCs
of MI mice
At 24 h and on day 7, MI model mice were sacrificed
by cervical dislocation and the spleens were removed and crushed to
prepare the splenic single-cell suspension. Subsequently, the
method of positive selection was used with anti-CD11c+
microbeads (Miltenyi Biotec GmbH) according to the manufacturers'
instructions to obtain purified CD11c+ DCs. Next,
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) was used to extract total RNA from DCs, according to the
manufacturers' instructions. Finally, the total RNA was sent to
Beijing Boao Biological Co., Ltd. for quality testing, and
Affymetrix miRNA 4.0 microarray for miRNAs screening was performed
after passing the quality testing.
Detection of miRNA expression within
DEXs
miRcute miRNA extraction and isolation kit (Tiangen
Biochemical Technology Co., Ltd.) was used to extract miRNA in DEXs
following the manufacturer's instructions. Then, the miRcute miRNA
First-Strand cDNA Synthesis Kit (Tiangen) was used to generate cDNA
from miRNA according to the manufacturer's instructions. The
miRcute miRNA qPCR Detection Kit (SYBR Green; Tiangen) was used for
qPCR following the manufacturer's instructions. The reactions were
incubated in a 96-well plate at 94°C for 2 min, followed by 40
cycles of 94°C for 20 sec and 60°C for 34 sec on an Applied
Biosystems 7500 Real-time PCR System (Thermo Fisher Scientific,
Inc.). To calculate the relative fold change values, the Cq values
were normalized to U6 miRNA as internal control. Analysis was
performed using the 2−ΔΔCq method (20). Primers were synthesized by Tiangen
Biochemical Technology Co., Ltd. (Hsa-miR-494-3p; stock no.
MIMAT0002816; Hsa-U6; stock no. CD201-0145). The mature sequence of
miR-494-3p was 5′-UGA AAC AUA CAC GGG AAA CCU C-3′.
miR-494-3p transfection into DEXs
miR-494-3p mimic, inhibitor and their scrambled
negative controls were purchased from Qiagen (cat. nos. 219600,
219300, 1027281 and 1027271, respectively) and transfected into
normal cultured BMDCs in vitro. Briefly, BMDCs were plated
in 6-well plates and cultured overnight. Then, the BMDCs were
transfected with 10 nM miR-494-3p mimic, 50 nM inhibitor or 10 nM
scram-bled sequence using riboFECTTM CP Reagent (Guangzhou RiboBio
Co., Ltd.) according to the manufacturer's protocol. Successful
transfection was confirmed by RT-qPCR assessment of the expression
of miR-494-3p. BMDCs only treated with riboFECTTM CP Reagent
without miRNAs were considered as the blank group and those
transfected with the scrambled negative controls were used as the
negative control (NC) group. Following transfection, BMDCs were
cultured with a non-serum medium (X-VIVO 15; Lonza Group, Ltd.) in
a 37°C, 5% CO2 incubator for 24 h. Then, DEXs were
isolated as described above. Finally, DEXs were co-cultured with
CMECs and tube formation assay was performed, or DEX-miR-494-3p
were injected into the infarcted myocardium in MI model mice, as
described above.
Statistical analysis
All results presented are from at least three
independent experiments for each condition. Data are expressed as
the mean ± standard deviation. Statistical analysis was performed
using GraphPad Prism 5 software (GraphPad Prism Software Inc.).
Differences among groups were examined using either One-way ANOVA
(parametric method) or Kruskal-Wallis test (non-parametric method)
according to the results of normality and homogeneity tests of
variance. If there was a statistically significant difference among
groups, a post hoc test was applied to evaluate the difference
between groups. In this case, Bonferroni's test was used for
parametric data and Dunn's test was used for non-parametric data.
P<0.05 was considered to indicate a statistically significant
difference.
Results
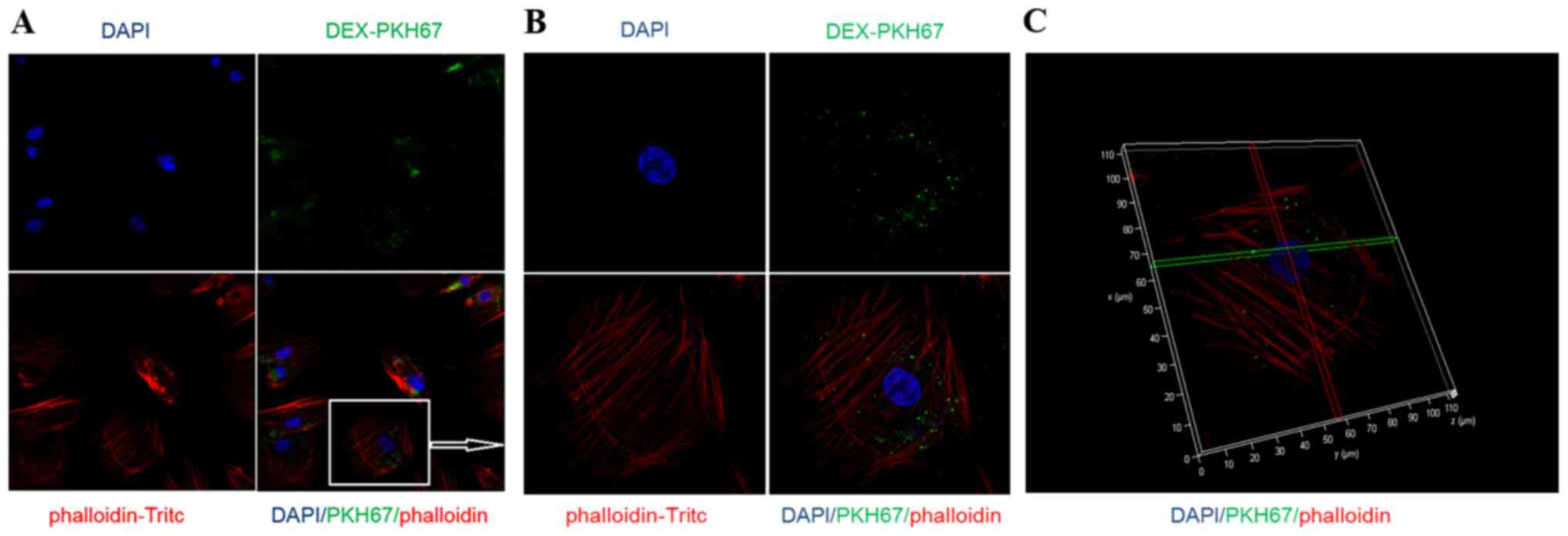
DEXs are taken up by CMECs
After co-culturing PKH67-labeled DEXs with CMECs,
confocal laser scanning microscopy was used to examine whether DEXs
were taken up by CMECs, and it was observed that CMECs were able to
directly take up DEXs (Fig.
2).
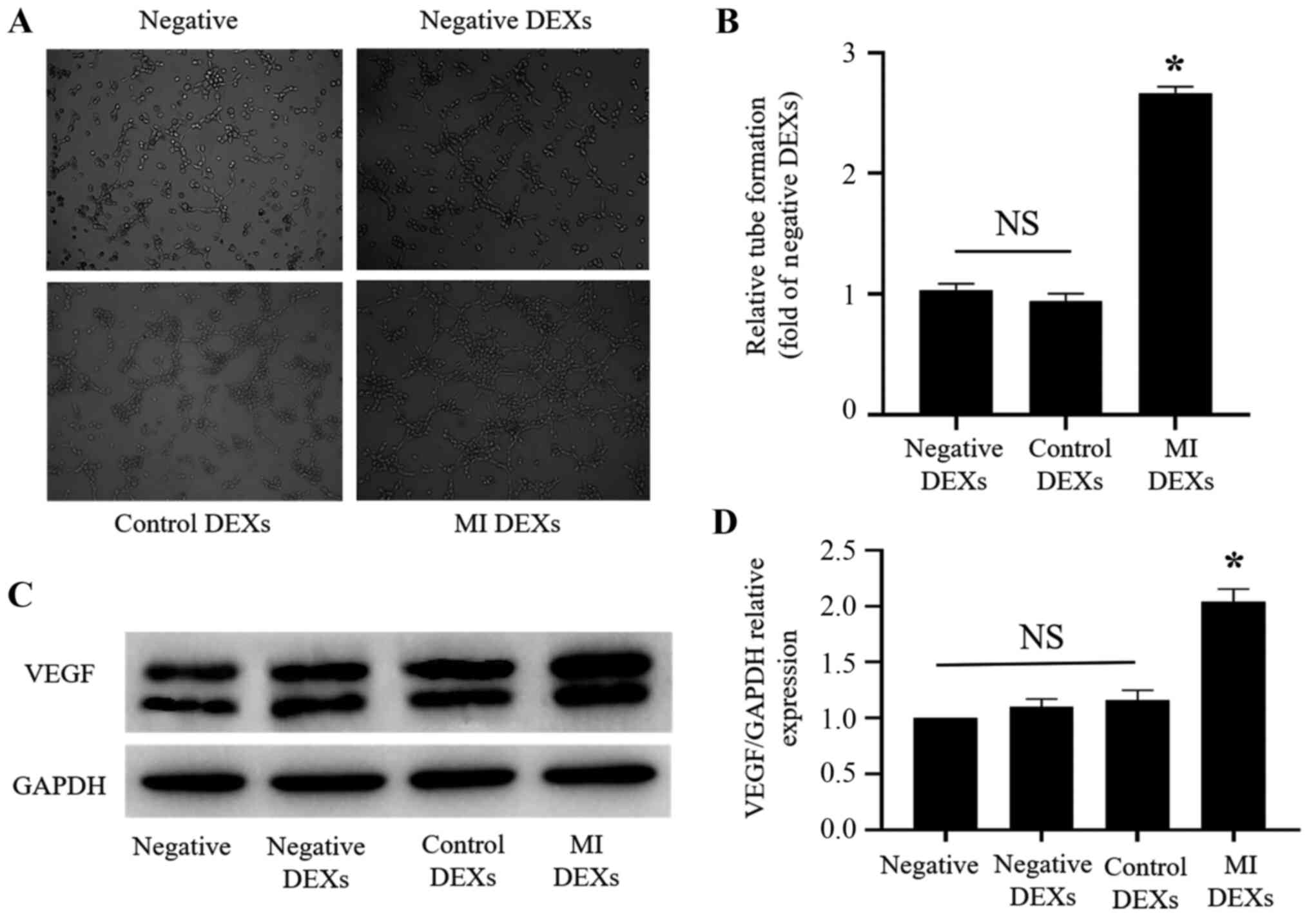
Effects of DEXs on CMECs
After determining, using confocal laser scanning
microscopy, that DEXs could be taken up by CMECs, the effects of
DEXs on CMECs were examined. Matrigel was used to perform a tube
formation assay with CMECs and explore the effects of negative,
control and MI DEXs on tube formation by CMECs. It was observed
that MI DEXs, compared with negative and control DEXs,
significantly promoted the tube formation ability of CMECs at 6 h
(P<0.05; Fig. 3A and B). After
co-culturing negative, control and MI DEXs with CMECs for 24 h, the
expression of VEGF in CMECs was detected by western blotting. It
was observed that MI DEXs significantly upregulated VEGF expression
in CMECs compared with negative, negative DEXs and control DEXs
(Fig. 3C and D).
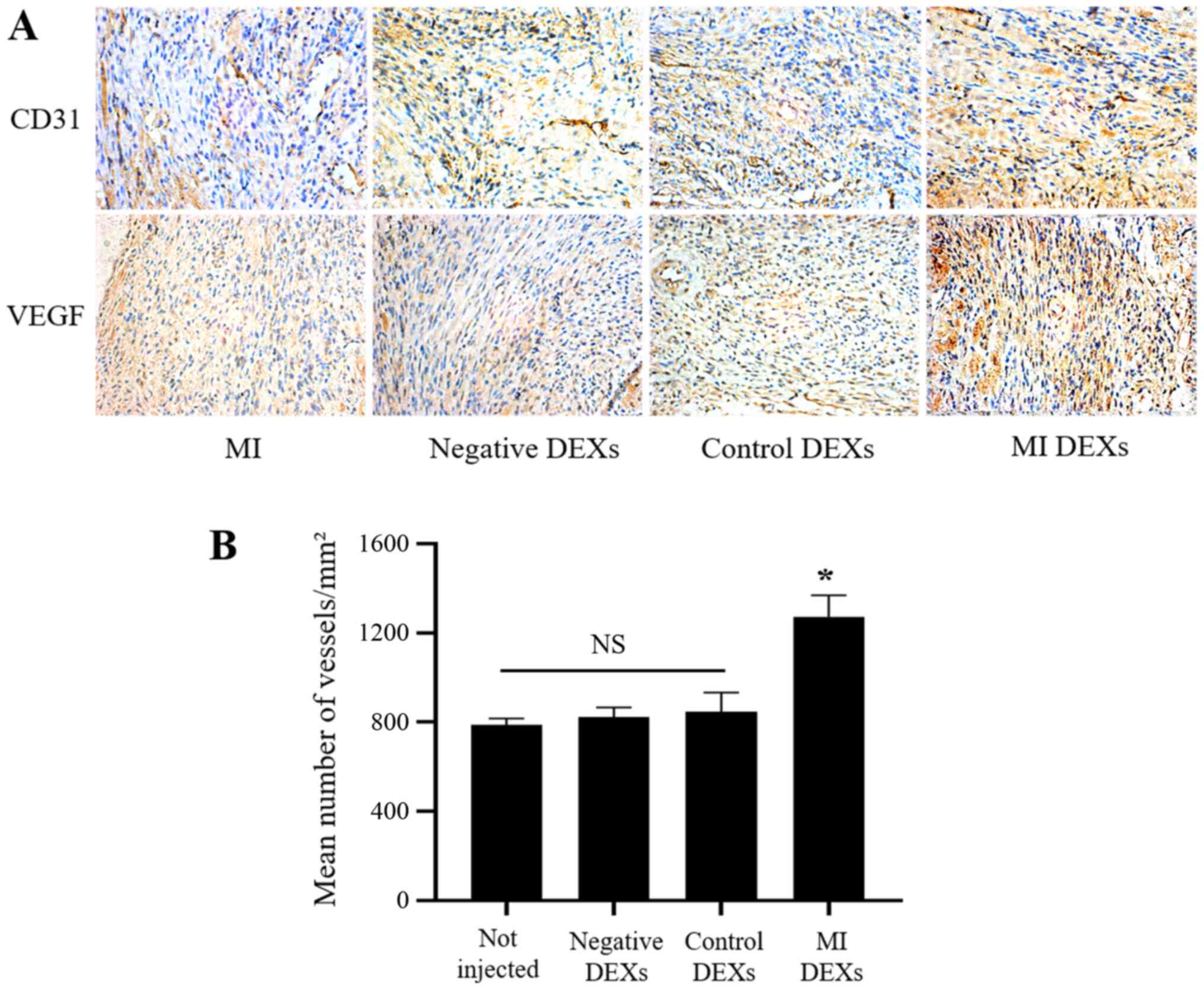
MI DEXs promote angiogenesis in mice
after MI
After the in vitro experiments demonstrated
that DEXs affected CMECs, the effects of DEXs on the myocardium
in vivo were investigated. The immunohistochemistry results
revealed that the expression of CD31 and VEGF in the infarcted
myocardium of MI model mice was markedly increased in the MI DEXs
compared with the MI, negative DEXs and control DEXs (Fig. 4A). Furthermore, the mean number of
vessels in the infarcted myocardium was significantly increased in
the MI DEX compared with non-injected, negative DEXs and control
DEXs (Fig. 4B). These results
suggested that injection of MI DEXs promoted angiogenesis after MI
in mice.
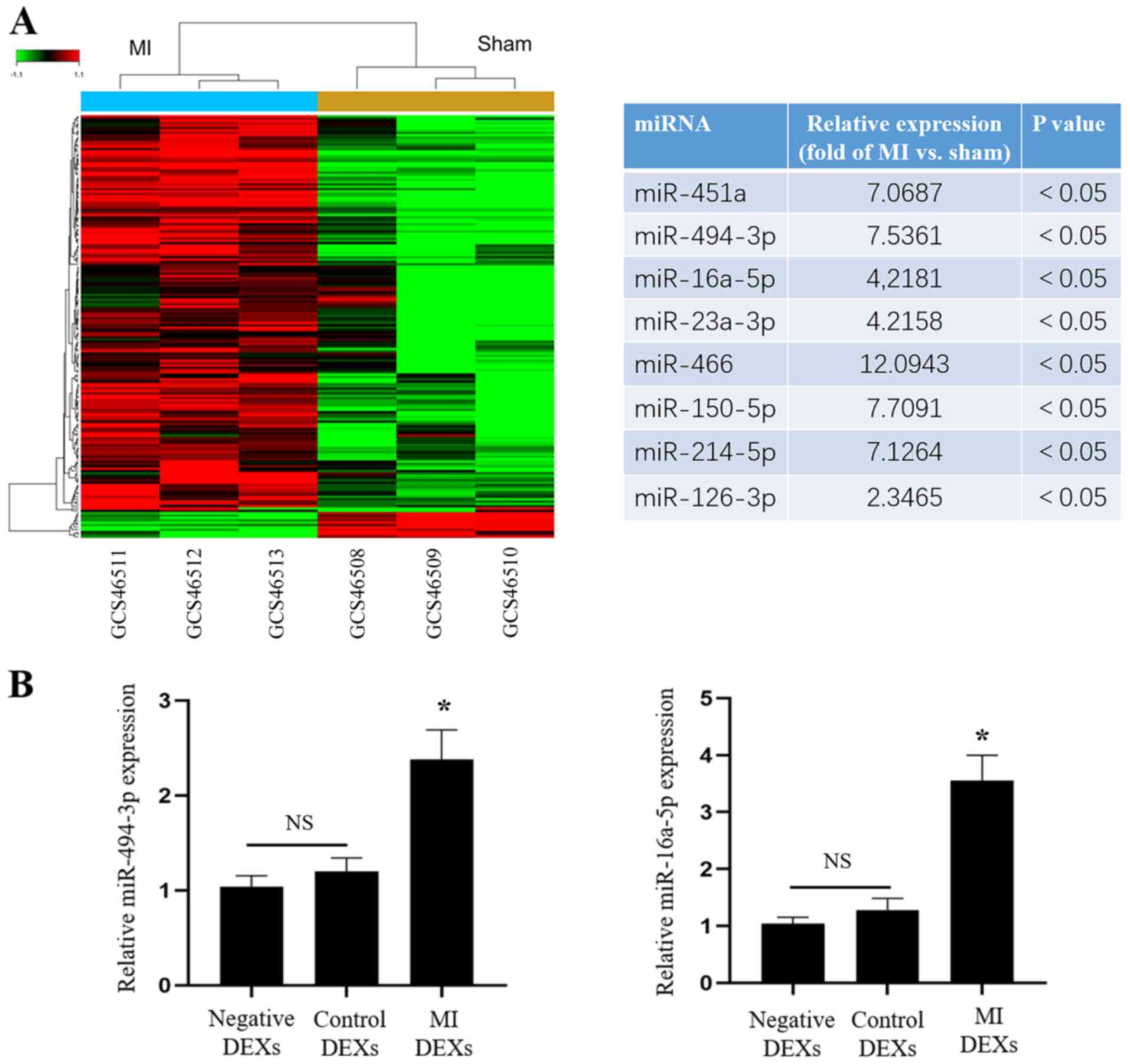
miRNA expression in DCs from mice after
MI
Our previous experiments demonstrated that DEXs
could enhance angiogenesis in vitro as well as in
vivo, but the mechanisms through which DEXs exert their effects
remained unknown. A large number of studies have shown that EXs
contain various miRNAs, some of which are associated with
angiogenesis. Therefore, gene chip screening for the expression of
miRNAs in splenic DCs of MI model mice was performed, and revealed
that the expression levels of miR-451a, miR-494-3p, miR-16a-5p
miR-23a-3p, miR-466, miR-150-5p, miR-214-5p and miR-126-3p which
are associated with angiogenesis, were significantly increased
compared with that in sham mice (P<0.05; Fig. 5A). Then, RT-qPCR was used to
detect the expression of angiogenesis-related miRNAs, and the
expression of miR-494-3p and miR-16a-5p was also found to be
significantly increased in MI DEXs compared with that in negative
DEXs and control DEXs (P<0.05; Fig. 5B), but other miRNAs were not
significantly increased. The enhancement of tube formation by
miR-16a-5p in vitro was not observed in our previous
experiments, therefore miR-494-3p was selected for further
experiments in the present study.
DEX-miR-494-3p enhances tube formation in
CMECs in vitro
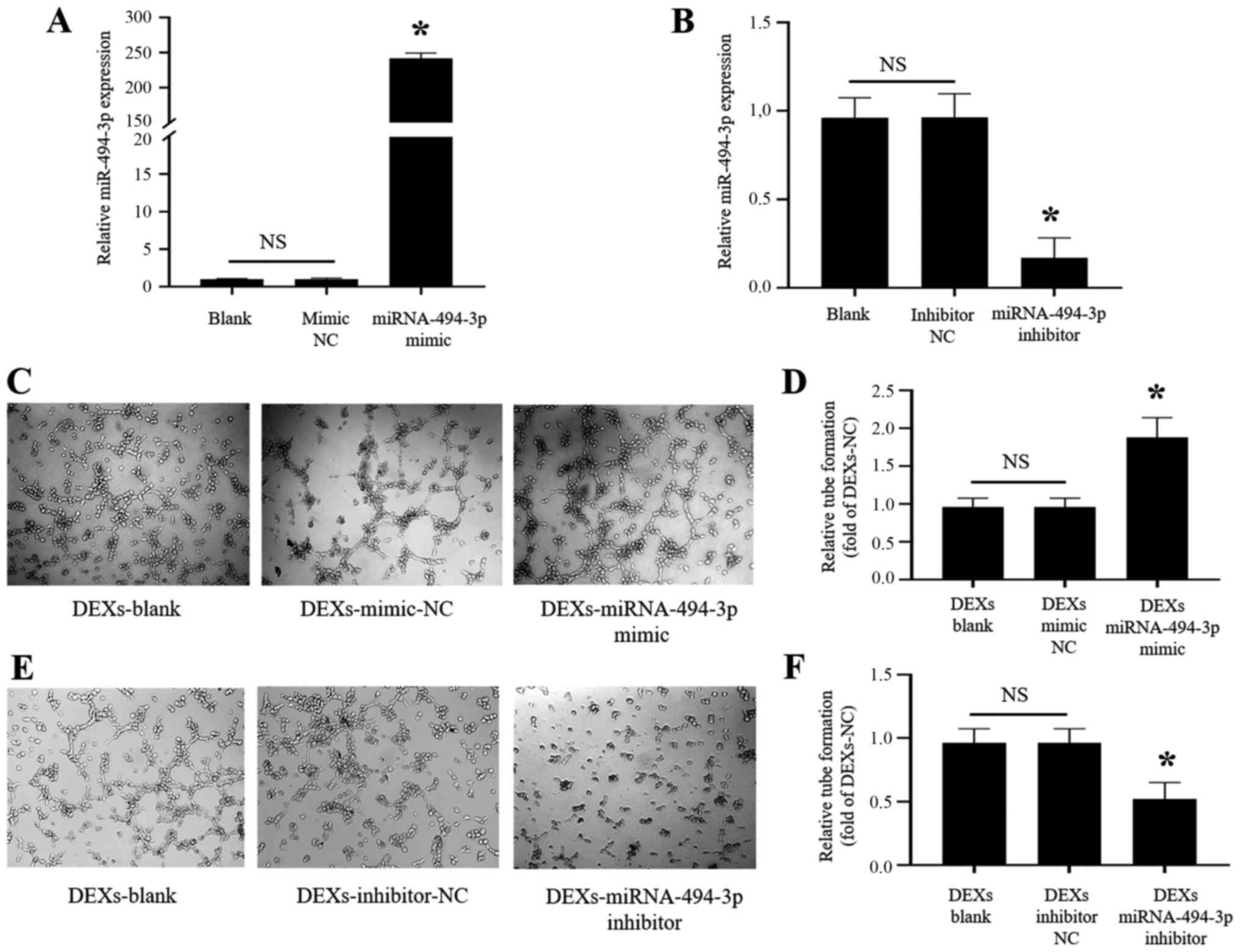
miR-494-3p mimic and inhibitor were transfected into
BMDCs, and successful transfection was confirmed by RT-qPCR
analysis of miR-494-3p expression, which was significantly
increased in the mimic and significantly reduced in the inhibitor
compared with the blank and NC (Fig.
6A and B). It was observed that DEX-miR-494-3p mimic
significantly enhanced the tube formation by CMECs at 6 h
(P<0.05; Fig. 6C and D), while
DEX-miR-494-3p inhibitor significantly inhibited tube formation by
CMECs compared with DEXs-blank and DEXs-NC (P<0.05; Fig. 6E and F).
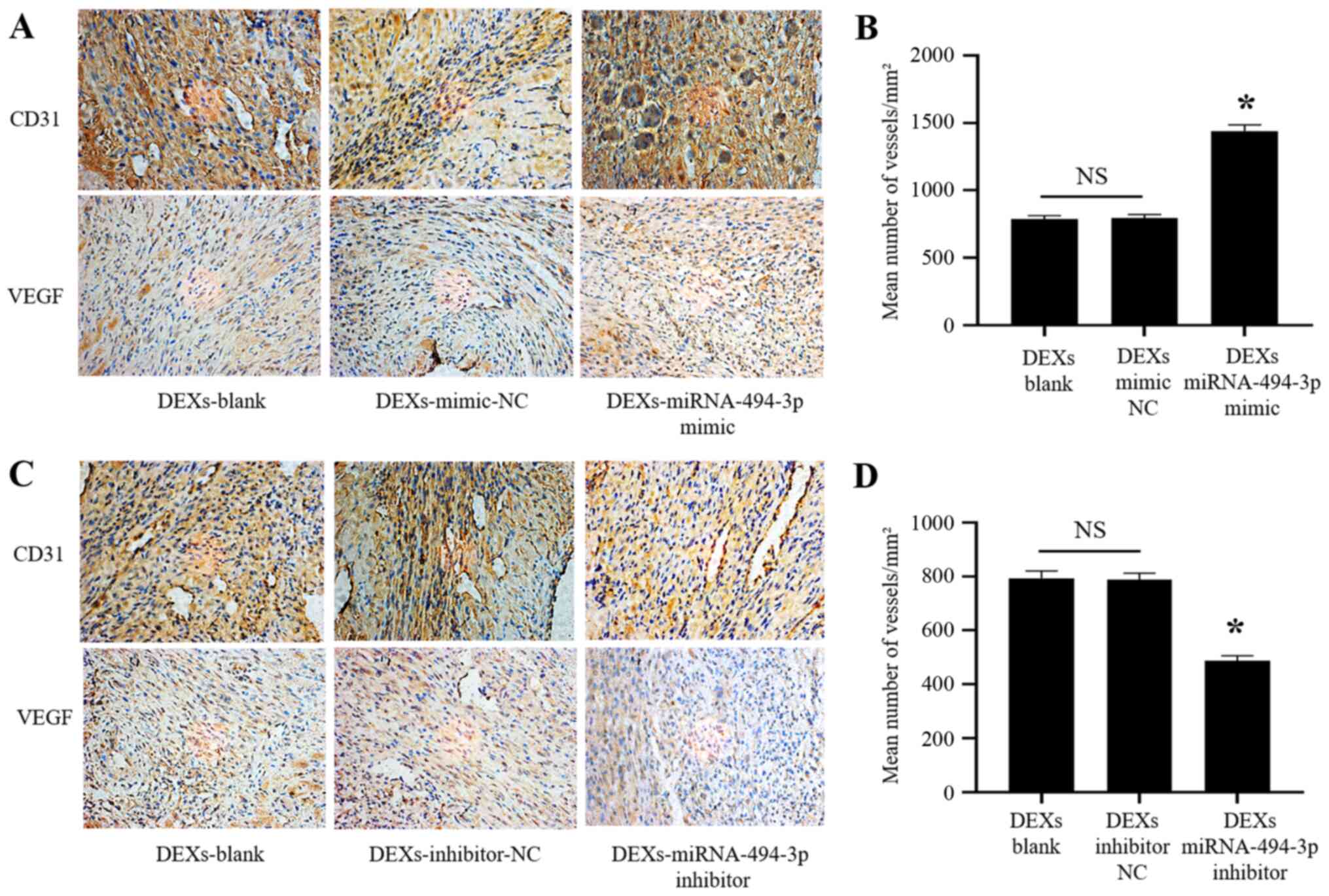
DEX-miR-494-3p promotes angiogenesis
after MI in mice
Our previous experiments revealed that
DEX-miR-494-3p enhanced tube formation by CMECs in vitro. It
was next investigated whether DEX-miR-494-3p mimic induced
angiogenesis in vivo. DEXs injected into the infarcted
myocardium in MI model mice were used, and it was observed that
DEX-miR-494-3p mimic markedly increased the expression of CD31 and
VEGF in the infarcted myocardium, while DEX-miR-494-3p inhibitor
markedly decreased CD31 and VEGF expression compared with the DEXs
blank and DEXs NC (Fig. 7A and
C). In addition, the mean number of vessels in the infarcted
myocardium was significantly increased in the DEX-miR-494-3p mimic
group and significantly decreased in the DEX-miR-494-3p inhibitor
group compared with the DEXs blank and DEXs NC groups (Fig. 7B and D).
Discussion
In the present study, it was demonstrated that EXs
secreted by DCs can be directly taken up by CMECs. Furthermore, MI
DEXs significantly upregulated VEGF expression in CMECs and
enhanced tube formation and angiogenesis in the infarcted
myocardium of MI model mice. Gene chip screening and RT-qPCR
validation revealed that the expressions of miR-494-3p and
miR-16a-5p, which are associated with angiogenesis, were
significantly increased in MI DEXs, and further studies revealed
that transfer of miR-494-3p mimic into DEXs significantly enhanced
tube formation by CMECs and angiogenesis in the infarcted
myocardium of MI model mice.
Our previous study demonstrated that MI DEXs could
significantly improve cardiac function in mice after MI (10). The present study demonstrated that
MI DEXs could increase the number of vessels and the expression of
CD31 and VEGF in the infarcted myocardium, which indicated that EXs
derived from DCs may improve cardiac function by inducing
myocardial angiogenesis via paracrine signaling post-MI.
A large number of studies have demonstrated that EXs
are the mediators of cell-to-cell communication. EXs secreted from
cells can directly act on or be taken up by other cells and play a
role in regulating the function of target cells (21-23). Previous studies have also reported
that EXs play an important role in myocardial healing and cardiac
remodeling after MI (8,17,21,24). Khan et al (21) observed that EXs extracted from
embryonic stem cells could promote the repair of injured myocardial
tissue and improve cardiac function and survival rate of mice after
MI. Teng et al (25)
demonstrated that bone marrow mesenchymal stem cell-derived EXs
enhanced angiogenic capacity and significantly improved cardiac
function after MI in rats. However, the majority of previous
studies focused on the role of EXs secreted by stem cells,
cardiomyocytes or fibroblasts (21,26), while to the best of our knowledge,
there is still no relevant report on the role of EXs secreted by
immune inflammatory cells in the repair of myocardial injury after
MI.
It has been demonstrated that immune inflammatory
cells, including DCs, play a key role in cardiac remodeling and are
implicated in cardiac angiogenesis after MI (6,27,28); however, whether DCs promote
angiogenesis by secreting DEXs remains unknown, to the best of our
knowledge. The present study demonstrated that MI DEXs
significantly upregulated the expression of VEGF in CMECs and
enhanced tube formation and angiogenesis in the infarcted
myocardium of MI model mice, which indicated that the DCs
infiltrating the infarcted myocardium post-MI may promote
angiogenesis by secreting DEXs, and the angiogenesis following MI
plays an important role in the recovery of the injured myocardium
and maintenance of cardiac function (29).
Regarding the mechanism through which DEXs promote
angiogenesis post-MI, previous studies demonstrated that EXs
secreted by cells contain a large number of miRNAs (21,26), some of which have been confirmed
by several studies to be associated with angiogenesis (11,12,30,31). Therefore, the miRNAs in splenic
DCs of MI model mice were screened using Affymetrix miRNA 4.0
microarrays, and the expression levels of miR-451a, miR-494-3p and
miR-16a-5p, which are associated with angiogenesis, were found to
be significantly increased compared with that in sham mice. Then,
these miRNAs were examined in MI DEXs by RT-qPCR and the expression
of miR-494-3p and miR-16a-5p was also found to be significantly
increased in MI DEXs. miR-494-3p was then transferred into DEXs,
and DEX-miR-494-3p was used to induce tube formation by CMECs and
angiogenesis in MI model mice. The results demonstrated that
DEX-miR-494-3p significantly enhanced tube formation by CMECs and
angiogenesis in MI model mice post-MI, which indicated that the
effect of DEXs on myocardium is partially mediated by the high
expression of miRNAs within DEXs.
Previous studies have reported the association
between miR-494 and angiogenesis, but the conclusions have been
inconsistent. Mao et al (32) found that tumor-derived miR-494
could promote angiogenesis in cell carcinoma. However, Welten et
al observed that inhibition of miRNA clusters in the 14q32
region, including miR-494, increased angiogenesis as well as
recanalization after peripheral vascular ischemia (33). In the present study, miR-494-3p
significantly enhanced tube formation by CMECs in vitro and
angiogenesis in MI model mice. Thus, the role of miR-494 in
angiogenesis is not entirely consistent across different disease
models and requires further in-depth investigation.
In conclusion, the present study demonstrated that
DCs can promote angiogenesis by secreting DEXs after MI, which is
partially achieved by delivery of highly expressed miRNAs in DEXs.
These findings indicate a novel DEX-miRNA-based approach to MI
treatment. There are some limitations in the present study; it did
not identify the target of miR-494-3p and how this affects the
angiogenic pathways post-MI and the cell signaling events
downstream. Therefore, the underlying molecular mechanisms require
further elucidation by future studies.
Abbreviations:
|
MI
|
myocardial infarction
|
|
DCs
|
dendritic cells
|
|
BMDCs
|
bone marrow-derived dendritic
cells
|
|
DEXs
|
dendritic cell-derived exosomes
|
|
CMECs
|
cardiac microvascular endothelial
cells
|
|
VEGF
|
vascular endothelial growth factor
|
Acknowledgments
The authors would like to thank Dr Qiao Ke
(Institute of Metabolic and Integrative Biology, Fudan University,
Shanghai, China) for providing help with confocal imaging, Dr Zhao
Zhonghua (Department of Pathology, School of Basic Medical Science,
Fudan University, Shanghai, China) for providing help with
immunohistochemistry, and Dr Ma Leilei (Shanghai Institute of
Cardiovascular Diseases, Zhongshan Hospital, Fudan University,
Shanghai, China) for his help with the construction of the MI mouse
model.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant nos. 81770350, 81570237,
81870197 and 81870200), the Key Disciplines Group Construction
Project of Pudong Health Bureau of Shanghai (grant no.
PWZxq2017-05), and the Top-level Clinical Discipline Project of
Shanghai Pudong District (grant no. PWYgf2018-02).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HL performed the cell culture, exosome isolation and
tube formation assay. YZ determined microRNA expression by reverse
transcription-quantitative PCR analysis. JY performed cell
transfection. HL, YZ and JY wrote the manuscript. WG and XZ
constructed the myocardial infarction mouse model and performed the
injection of DEXs. KY performed confocal laser scanning microscopy
examination. LL discussed the results, commented on the manuscript
and analyzed the data. JG designed the experiments and analyzed the
data. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Institutional
Review Board of Zhongshan Hospital of Fudan University and the
Shanghai Institutes of Biological Sciences-CAS (approval no.
A5894-01).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
van der Laan AM, Piek JJ and van Royen N:
Targeting angiogenesis to restore the microcirculation after
reperfused MI. Nat Rev Cardiol. 6:515–523. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shah AM and Mann DL: In search of new
therapeutic targets and strategies for heart failure: Recent
advances in basic science. Lancet. 378:704–712. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Frangogiannis NG: Regulation of the
inflammatory response in cardiac repair. Circ Res. 110:159–173.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yan X, Anzai A, Katsumata Y, Matsuhashi T,
Ito K, Endo J, Yamamoto T, Takeshima A, Shinmura K, Shen W, et al:
Temporal dynamics of cardiac immune cell accumulation following
acute myocardial infarction. J Mol Cell Cardiol. 62:24–35. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Montecalvo A, Larregina AT, Shufesky WJ,
Stolz DB, Sullivan ML, Karlsson JM, Baty CJ, Gibson GA, Erdos G,
Wang Z, et al: Mechanism of transfer of functional microRNAs
between mouse dendritic cells via exosomes. Blood. 119:756–766.
2012. View Article : Google Scholar :
|
|
6
|
Anzai A, Anzai T, Nagai S, Maekawa Y,
Naito K, Kaneko H, Sugano Y, Takahashi T, Abe H, Mochizuki S, et
al: Regulatory role of dendritic cells in postinfarction healing
and left ventricular remodeling. Circulation. 125:1234–1245. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang D, Lee H, Zhu Z, Minhas JK and Jin
Y: Enrichment of selective miRNAs in exosomes and delivery of
exosomal miRNAs in vitro and in vivo. Am J Physiol Lung Cell Mol
Physiol. 312:L110–L121. 2017. View Article : Google Scholar :
|
|
8
|
Emanueli C, Shearn AIU, Angelini GD and
Sahoo S: Exosomes and exosomal miRNAs in cardiovascular protection
and repair. Vascul Pharmacol. 71:24–30. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vlassov AV, Magdaleno S, Setterquist R and
Conrad R: Exosomes: Current knowledge of their composition,
biological functions, and diagnostic and therapeutic potentials.
Biochim Biophys Acta. 1820:940–948. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu H, Gao W, Yuan J, Wu C, Yao K, Zhang
L, Ma L, Zhu J, Zou Y and Ge J: Exosomes derived from dendritic
cells improve cardiac function via activation of CD4+ T
lymphocytes after myocardial infarction. J Mol Cell Cardiol.
91:123–133. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tiwari A, Mukherjee B and Dixit M:
MicroRNA key to angiogenesis regulation: MiRNA biology and therapy.
Curr Cancer Drug Targets. 18:266–277. 2018. View Article : Google Scholar
|
|
12
|
Sun P, Zhang K, Hassan SH, Zhang X, Tang
X, Pu H, Stetler RA, Chen J and Yin KJ: Endothelium-targeted
deletion of microRNA-15a/16-1 promotes post-stroke angiogenesis and
improves long-term neurological recovery. Circ Res. 126:1040–1057.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lin Y, Zhang C, Xiang P, Shen J, Sun W and
Yu H: Exosomes derived from HeLa cells break down vascular
integrity by triggering endoplasmic reticulum stress in endothelial
cells. J Extracell Vesicles. 9:17223852020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu S, Chen J, Shi J, Zhou W, Wang L, Fang
W, Zhong Y, Chen X, Chen Y, Sabri A and Liu S: M1-like
macrophage-derived exosomes suppress angiogenesis and exacerbate
cardiac dysfunction in a myocardial infarction microenvironment.
Basic Res Cardiol. 115:222020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kang JY, Park H, Kim H, Mun D, Park H, Yun
N and Joung B: Human peripheral bloodderived exosomes for microRNA
delivery. Int J Mol Med. 43:2319–2328. 2019.PubMed/NCBI
|
|
16
|
Liu H, Yuan J, Gao W, Wu C, Yao K, Zhang
L, Guo X, Yu W, Zou Y and Ge J: Exosomes secreted from dendritic
cells enhance tube formation in cardiac microvascular endothelial
cells after myocardial infarction. European Heart Journal. 36(Suppl
1): 19582015.
|
|
17
|
Alexander M, Hu R, Runtsch MC, Kagele DA,
Mosbruger TL, Tolmachova T, Seabra MC, Round JL, Ward DM and
O'Connell RM: Exosome-delivered microRNAs modulate the inflammatory
response to endotoxin. Nat Commun. 6:73212015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gao E, Lei YH, Shang X, Huang ZM, Zuo L,
Boucher M, Fan Q, Chuprun JK, Ma XL and Koch WJ: A novel and
efficient model of coronary artery ligation and myocardial
infarction in the mouse. Circ Res. 107:1445–1453. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Huang K, Liu J, Zhang H, Wang J and Li H:
Intramyocardial injection of siRNAs can efficiently establish
myocardial tissue-specific renalase knockdown mouse model. Biomed
Res Int. 2016:12675702016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
21
|
Khan M, Nickoloff E, Abramova T, Johnson
J, Verma SK, Krishnamurthy P, Mackie AR, Vaughan E, Garikipati VN,
Benedict C, et al: Embryonic stem cell-derived exosomes promote
endogenous repair mechanisms and enhance cardiac function following
myocardial infarction. Circ Res. 117:52–64. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bang C, Batkai S, Dangwal S, Gupta SK,
Foinquinos A, Holzmann A, Just A, Remke J, Zimmer K, Zeug A, et al:
Cardiac fibroblast-derived microRNA passenger strand-enriched
exosomes mediate cardiomyocyte hypertrophy. J Clin Invest.
124:2136–2146. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sun X, He S, Wara AK, Icli B, Shvartz E,
Tesmenitsky Y, Belkin N, Li D, Blackwell TS, Sukhova GK, et al:
Systemic delivery of microRNA-181b inhibits nuclear factor-κB
activation, vascular inflammation, and atherosclerosis in
apolipoprotein E-deficient mice. Circ Res. 114:32–40. 2014.
View Article : Google Scholar
|
|
24
|
Xiao X, Lu Z, Lin V, May A, Shaw DH, Wang
Z, Che B, Tran K, Du H and Shaw PX: MicroRNA miR-24-3p reduces
apoptosis and regulates Keap1-Nrf2 pathway in mouse cardiomyocytes
responding to ischemia/reperfusion injury. Oxid Med Cell Longev.
2018:70421052018. View Article : Google Scholar
|
|
25
|
Teng X, Chen L, Chen W, Yang J, Yang Z and
Shen Z: Mesenchymal stem cell-derived exosomes improve the
microenvironment of infarcted myocardium contributing to
angiogenesis and anti-inflammation. Cell Physiol Biochem.
37:2415–2424. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gao W, Liu H, Yuan J, Wu C, Huang D, Ma Y,
Zhu J, Ma L, Guo J, Shi H, et al: Exosomes derived from mature
dendritic cells increase endothelial inflammation and
atherosclerosis via membrane TNF-α mediated NF-κB pathway. J Cell
Mol Med. 20:2318–2327. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hofmann U and Frantz S: Role of T-cells in
myocardial infarction. Eur Heart J. 37:873–879. 2016. View Article : Google Scholar
|
|
28
|
Hofmann U and Frantz S: Role of
lymphocytes in myocardial injury, healing, and remodeling after
myocardial infarction. Circ Res. 116:354–367. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nishida M, Carley WW, Gerritsen ME,
Ellingsen O, Kelly RA and Smith TW: Isolation and characterization
of human and rat cardiac microvascular endothelial cells. Am J
Physiol. 264:H639–H652. 1993.PubMed/NCBI
|
|
30
|
Zeng Z, Li Y, Pan Y, Lan X, Song F, Sun J,
Zhou K, Liu X, Ren X, Wang F, et al: Cancer-derived exosomal
miR-25-3p promotes pre-metastatic niche formation by inducing
vascular permeability and angiogenesis. Nat Commun. 9:53952018.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sun Z, Shi K, Yang S, Liu J, Zhou Q, Wang
G, Song J, Li Z, Zhang Z and Yuan W: Effect of exosomal miRNA on
cancer biology and clinical applications. Mol Cancer. 17:1472018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mao G, Liu Y, Fang X, Liu Y, Fang L, Lin
L, Liu X and Wang N: Tumor-derived microRNA-494 promotes
angiogenesis in non-small cell lung cancer. Angiogenesis.
18:373–382. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Welten SM, Bastiaansen AJ, De Jong RC, de
Vries MR, Peters EA, Boonstra MC, Sheikh SP, Monica NL, Kandimalla
ER, Quax PH and Nossent AY: Inhibition of 14q32 MicroRNAs miR-329,
miR-487b, miR-494, and miR-495 increases neovascularization and
blood flow recovery after ischemia. Circ Res. 115:696–708. 2014.
View Article : Google Scholar : PubMed/NCBI
|