Introduction
Glioblastoma multiforme (GBM) is the most malignant
primary brain tumor in humans and the most lethal cancer of the
central nervous system, with an annual incidence of 3.19 cases per
100,000 persons (1-3). Despite significant advances in
tumor resection, radiation therapy, and chemotherapy, the prognosis
of GBM remains poor. Treatment failure and continued disease
progression results in a median overall survival of approximately
12-15 months, with a 5-year survival rate of less than 10%
(4). Studies have revealed that
the rapid proliferation and high invasiveness of GBM cells lead to
treatment failure and tumor recurrence (5,6).
Therefore, it is urgent to develop an effective treatment for
GBM.
Src-family kinases (SFKs) are non-receptor tyrosine
kinases. The family of proteins contains nine members (Fyn, c-Src,
Yes, Lyn, Lck, Blk, Hck, Fgr and Yrk), five of which (Fyn, c-Src,
Yes, Lyn and Lck) are expressed in human gliomas (7-10). SFKs are frequently overexpressed
and/or activated in numerous human cancers (11-13), where they play a role in tumor
invasion, proliferation, metastasis, survival, and angiogenesis
(14). By knocking down
individual SFKs in cultured cells (LN229, SF767, and GBM8),
Lewis-Tuffin et al (7)
determined that reduced c-Src, Fyn, and Yes expression also reduced
growth and migration and altered motility-related protein
phosphorylation patterns, while reduced Lyn expression had little
effect on growth and migration. Other in vitro studies have
revealed that Fyn knockdown is associated with decreased glioma
cell proliferation and migration (15,16). Fyn tyrosine kinase, a downstream
target of the oncogenic receptor tyrosine kinase pathway, is rarely
mutated, yet significantly overexpressed, in human GBM (17,18). The mechanisms of Fyn tyrosine
kinase overexpression in human glioma cells are poorly known yet
they are important since small-molecule inhibitors of Fyn may be
therapeutic options for glioma.
Rosmarinic acid (RA) is a natural phenolic compound
that acts as a Fyn inhibitor by homology modeling of the human Fyn
structure (19). RA can be found
in species of the Boraginaceae and Lamiaceae families, mainly in
the leaves of Rosmarinus officinalis, from which it can be
easily isolated; it can also be found in peppermint, lemon balm,
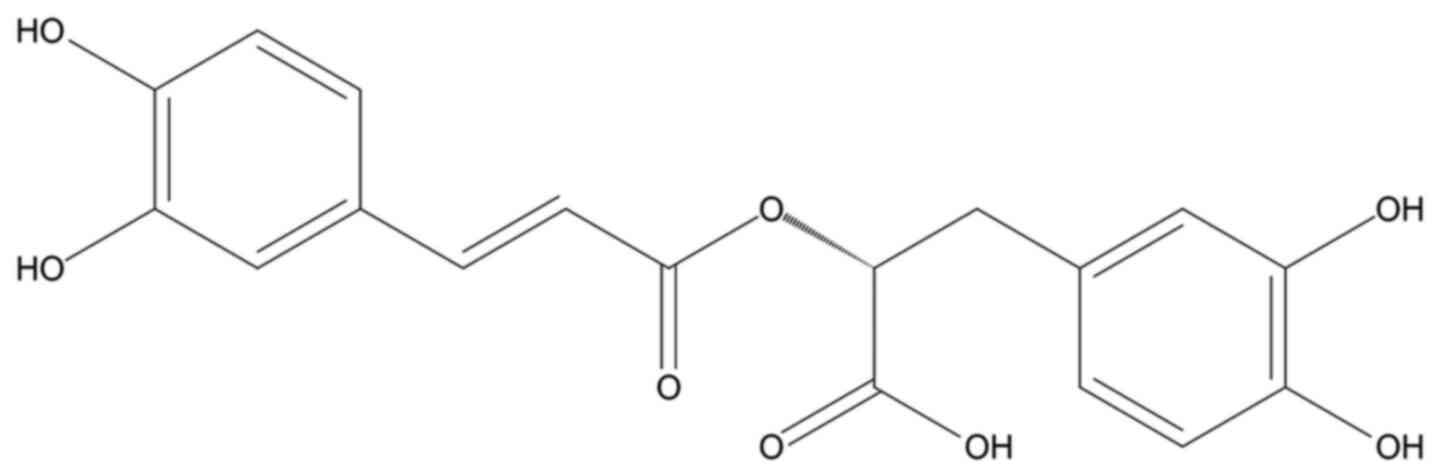
oregano, sage, and thyme (20,21). The molecular structure of RA
(chemical formula: C18H16O8)
contains two benzene rings located at the extremities of the
molecule and a pair of ortho-hydroxyl groups in each benzene ring
(Fig. 1). It has been reported
that RA exerts a variety of beneficial biological properties,
mainly antioxidant (22),
anti-inflammatory (23),
pro-apoptotic (24), and
neuroprotective (25) effects.
Furthermore, recent studies have revealed that RA has
antineoplastic activity in leukemia, hepatocellular carcinoma,
gastric carcinoma, colorectal cancer, breast cancer, and small-cell
carcinoma of the lung (26-29). However, the effects of RA on
tumor biological characteristics, such as proliferation, migration,
and invasion of human glioma cells and their mechanisms, have not
been clearly reported.
The phosphatidylinositol 3-kinase (PI3K)/Akt/nuclear
factor-κB (NF-κB) signaling pathway is an important pathway in the
regulation of tumorigenesis, and is significantly activated in
glioma (30). The activation of
PI3K/Akt/NF-κB signaling cascades is inhibited by Fyn knockdown in
primary astrocytes (31).
Therefore, it was hypothesized that RA may have an anti-glioma
effect by inhibiting the PI3K/Akt/NF-κB signaling pathway.
In the present study, the effects of RA on glioma
growth, migration, invasion, and apoptosis in vitro were
explored, and the findings suggest that RA may be a potential
therapeutic agent for glioma.
Materials and methods
Reagents and antibodies
RA was purchased from Aladdin (cat. no. R109804).
LY294002 and 740 Y-P were purchased from MedChemExpress (cat. nos.
HY-10108 and HY-P0175, respectively). The following primary
antibodies were used: Fyn (product code ab125016; 1:2,000) was
purchased from Abcam, PI3K (product no. 4249S; 1:1,000), MMP-2
(product no. 87809S; 1:1,000), MMP-9 (product no. 13667S; 1:1,000),
Bcl-2 (product no. 3498S; 1:1,000), Bax (product no. 2772S;
1:1,000), cleaved caspase-3 (product no. 9661S; 1:1,000),
fibrillarin (product no. 2639S; 1:1,000), and caspase-3 (product
no. 9662S; 1:1,000), were all from Cell Signaling Technology, Inc.,
and phosphorylated Akt (p-Akt) (cat. no. sc-514032; 1:500), Akt
(cat. no. sc-81434; 1:1,000), NF-κB p65 (cat. no. sc-8008;
1:1,000), β-actin (cat. no. sc-47778; 1:2,000), and
glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (cat. no.
sc-47724; 1:2,000) were purchased from Santa Cruz Biotechnology,
Inc.
Cell culture
Human-derived glioma cell lines (U251 and U343) and
the normal human astrocyte (NHA) cell line were purchased from the
American Type Culture Collection (ATCC). All cells were maintained
under 5% CO2 at 37°C in Dulbecco's modified Eagle's
medium (DMEM) (cat. no. 11995040) containing 100 U/ml penicillin,
100 µg/ml streptomycin (cat. no. 15070063), and 10% fetal
bovine serum (FBS) (cat. no. 10100147; all from Gibco; Thermo
Fisher Scientific, Inc.) in water-jacketed humidity-controlled
incubators.
Cell viability
Cell proliferation was calculated using Cell
Counting Kit-8 (CCK-8) assay (Dojindo Molecular Technologies, Inc.)
as per the manufacturer's instructions. Briefly, cells were seeded
in 96-well microplates at 2×103 cells/well and medium
containing various concentrations of RA (0, 100, 200 and 400
µM) were added to the wells. After incubation for 24 or 48
h, the medium was replaced with 10 µl CCK-8 solution. After
further incubation for 1 h at 37°C, the absorbance of each well was
measured at 450 nm using a microplate reader (Multiskan Go 1510;
Thermo Fisher Scientific, Inc.). In addition, cell viability
assessed by CCK-8 assay, was also performed on cells that were
treated with or without RA (200 µM) for 24 h after PI3K
agonist (740 Y-P, 25 µg/ml) or inhibitor (LY294002, 30
µM) pretreatment for 2 h.
Immunofluorescence
U251 and U343 cells were seeded on a cover slide in
a 24-well plate at a density of 5×104 cells/well and
maintained in a CO2 incubator for overnight growth.
Then, the cells were treated with RA (0, 100, 200 and 400
µM) or vehicle for 24 h. Subsequently, the cells were washed
three times with PBS and blocked for 1 h in PBS containing 0.2%
Triton X-100 and 10% skimmed milk powder at 37°C. Immunodetection
was performed by incubation with a Fyn-specific antibody at 37°C
for 1 h, followed by overnight incubation at 4°C. After washing
three times in PBS, the cover slides were incubated for 1 h at 37°C
with an anti-rabbit Alexa Fluor 488-conjugated secondary antibody
(1:200; Invitrogen; Thermo Fisher Scientific, Inc.). The cells were
then washed three times with PBS and mounted using ProLong™ Gold
antifade reagent (P36930; Thermo Fisher Scientific, Inc.). Stained
cells were analyzed at a magnification of ×200 using a confocal
microscope (LSM 800; Zeiss AG).
Wound healing assay
Cell migration was evaluated using the wound healing
assay as previously described (32). Briefly, U251 and U343 cells were
seeded in 24-well plates at 5×104 cells/well and allowed
to adhere. Then, a wound/scratch was produced using a 200-µl
pipette tip. After washing off the separated cells with
phosphate-buffered solution (PBS), serum-free medium containing
various concentrations of RA (0, 100, 200, and 400 µM) was
added to the wells. The wound was observed at regular intervals
between 0 and 48 h. Randomly selected areas were photographed at a
magnification of ×100 using a phase-contrast microscope (CKX41;
Olympus Corporation) and the wound area was calculated by ImageJ
software (version 1.8.0; National Institutes of Health).
Invasion assay
The invasion assay was conducted using
Corning® BioCoat™ Matrigel® Invasion Chambers
(product no. 354480; Corning, Inc.) with 8-µm pore chambers
inserted into 24-well plates. U251 and U343 cells (5×104
cells) were cultured in 500 µl of serum-free DMEM with RA in
the inserts and 500 µl of DMEM containing 15% FBS in the
bottom of the wells. After 24 h of incubation, the inserts were
washed three times with PBS and the cells were fixed with 4%
paraformaldehyde at room temperature for 10 min. After washing with
PBS, the cells were stained with 0.1% crystal violet at room
temperature for 15 min. Following another wash with PBS, the inner
sides of the chamber were wiped with a cotton swab and images of
the cells that invaded through the Matrigel® were
obtained using a phase-contrast microscope (magnification, ×200).
Finally, the number of invading cells was counted.
TUNEL and DAPI staining
U251 and U343 cells were seeded on a cover slide in
a 24-well plate at a density of 3×104 cells/well and
maintained in a CO2 incubator for overnight growth.
Cells were treated with various concentrations of RA (0, 100, 200,
and 400 µM) or vehicle for 24 h. Terminal deoxynucleotidyl
transferase (TdT) dUTP nick-end labeling (TUNEL) staining was
performed according to the manufacturer's instructions (product no.
C1086; Beyotime Institute of Biotechnology). Briefly, the cells
were fixed with 4% paraformaldehyde at room temperature for 30 min,
incubated for 5 min with 0.3% Triton X-100 in PBS, and then
incubated with a reaction mixture containing terminal
deoxynucleotidyl transferase and fluorescent labeling solution for
60 min at 37°C according to the manufacturer's protocol. The cells
were then stained with 4′,6-diamidino-2-phenylindole (DAPI) for 5
min at 37°C and mounted using ProLong™ Gold antifade reagent.
Stained cells were analyzed using a confocal microscope
(magnification, ×200).
Flow cytometry
Cell apoptosis was detected using an Annexin V-FITC
apoptosis detection kit (product no. C1062L; Beyotime Institute of
Biotechnology) according to the manufacturer's instructions. In
brief, cells were treated with RA (0, 100, 200 and 400 µM)
for 24 h and then resuspended in binding buffer containing Annexin
V and PI. After incubation at 25°C in the dark for 15 min,
apoptotic cells were evaluated immediately using a FACScan flow
cytometer (BD Biosciences). Data analysis was performed using
FlowJo v10.0.7 software (Tree Star, Inc.).
Western blot analysis
The cells were lysed in RIPA buffer (cat. no. 89901;
Thermo Fisher Scientific, Inc.) supplemented with complete protease
inhibitor cocktail and phosphatase inhibitor (Roche Diagnostics).
Lysate protein concentrations were determined using the BCA Protein
Assay kit (Beyotime Institute of Biotechnology). For some
experiments, Nuclear Protein Extraction kit (product no. P0027;
Beyotime Institute of Biotechnology) was used to isolate the
nuclear proteins in accordance with the manufacturer's
instructions. Proteins (20 µg) from each sample was
separated by 10% SDS-polyacrylamide gel electrophoresis (SDS-PAGE)
and transferred to polyvinylidene difluoride (PVDF) membranes (EMD
Millipore). After blocking with 5% w/v non-fat milk in TBST for 1 h
at room temperature, the membranes were incubated overnight at 4°C
with primary antibodies. Then, the membranes were washed with TBST
and horseradish peroxidase-linked IgG secondary antibodies
(anti-mouse; product no. 7076; 1:4,000; and anti-rabbit; product
no. 7074; 1:4,000; Cell Signaling Technology, Inc.) were added for
1 h at room temperature. The membranes were visualized with Western
Bright™ reagent (cat. no. K-12043-D10; Advansta, Inc.). The band
signals were captured by The ChemiDoc MP system (Bio-Rad
Laboratories, Inc.). ImageJ software (version 1.8.0; National
Institutes of Health) was used for densitometric analysis. The
experiments were repeated at least three times independently.
Statistical analyses
The data are expressed as the mean ± standard
deviation (SD) of at least three independent experiments, and
statistical differences of cell viability were determined using
two-way analysis of variance (ANOVA) followed by Dunnett's multiple
comparison test; other comparisons were performed using one-way
ANOVA followed by the Dunnett's multiple comparison test. All
statistical analyses were computed by SPSS statistical analysis
software version 22.0 (IBM Corp.). Values of P<0.05 were
considered to indicate statistically significant differences.
Results
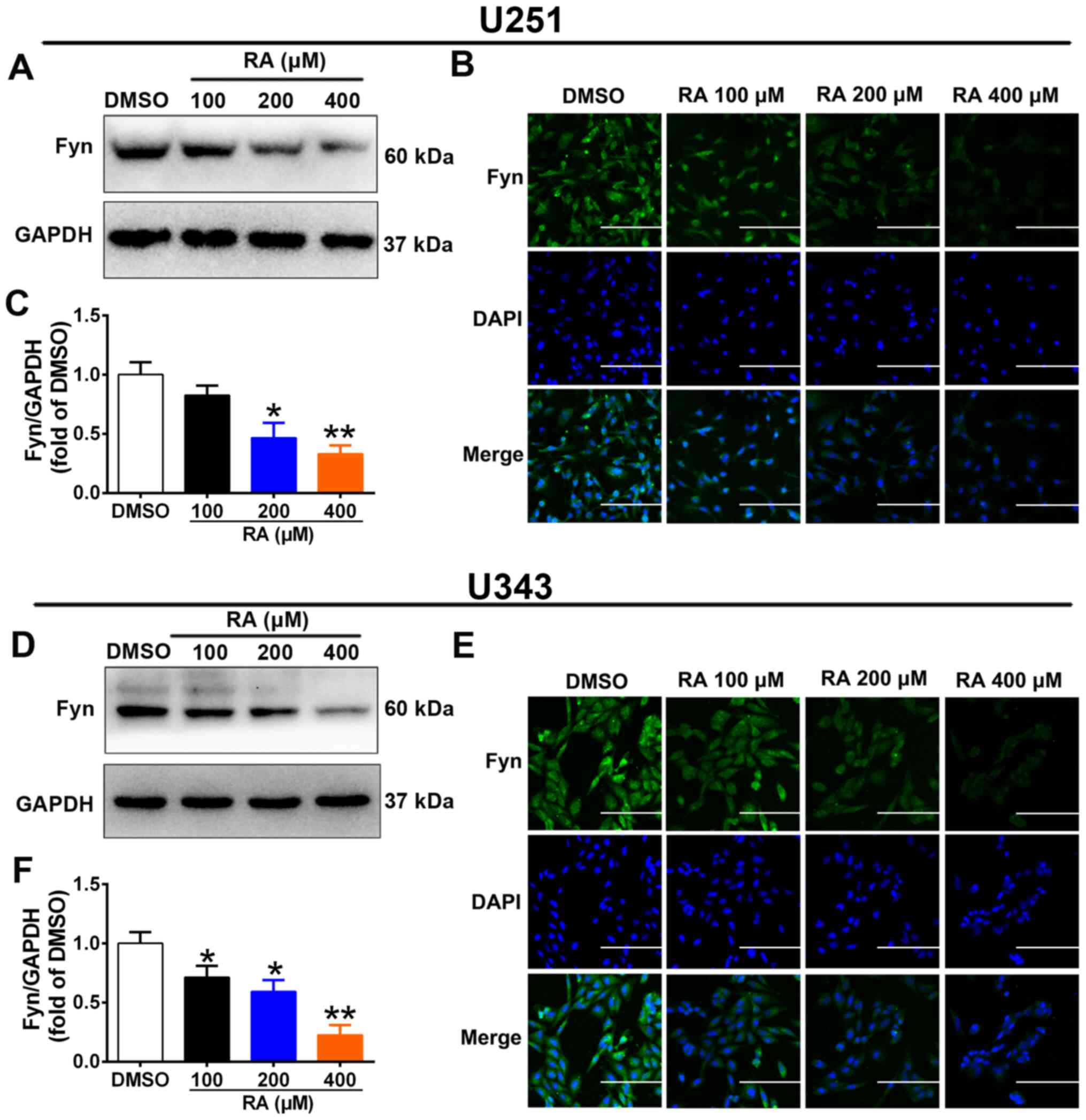
RA inhibits Fyn expression in vitro
First, the effect of RA on the expression of Fyn was
examined in glioma cell lines (Fig.
2). RA treatment reduced Fyn expression in U251 and U343 cells
in a dose-dependent manner (Fig. 2A,
C, D and F). Immunofluorescence staining confirmed that RA
inhibited Fyn expression in U251 and U343 cells (Fig. 2B and E).
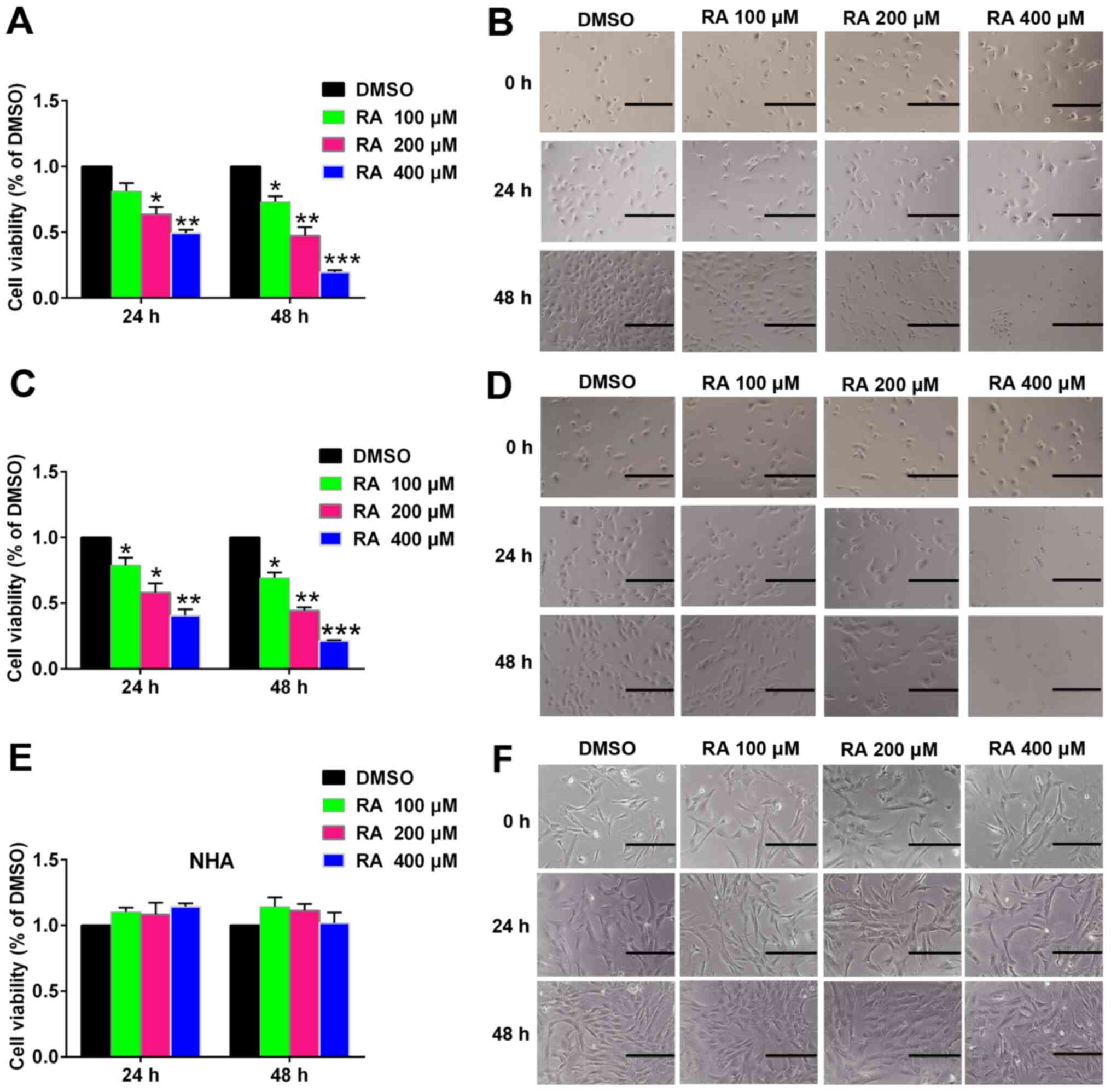
RA inhibits glioma cell
proliferation
Next, the effect of RA on glioma cell proliferation
was examined using the CCK-8 assay (Fig. 3). RA treatment inhibited cell
proliferation in a time- and dose-dependent manner in U251 and U343
cells (Fig. 3A and C).
RA-induced cytotoxicity was also observed under a microscope
(Fig. 3B and D). The images
revealed that as the concentration of RA increased, the density of
glioma cells decreased, and more cells shrank and died. These
observations were in line with the results of the CCK-8 assay.
As a control, the effect of RA on NHA was also
examined. RA treatment for 24 h or 48 h did not significantly
affect NHA cell viability and morphology (Fig. 3E and F). These results indicated
that RA specifically inhibited the proliferation of glioma cells,
with no significant effect on the proliferation of NHA cells.
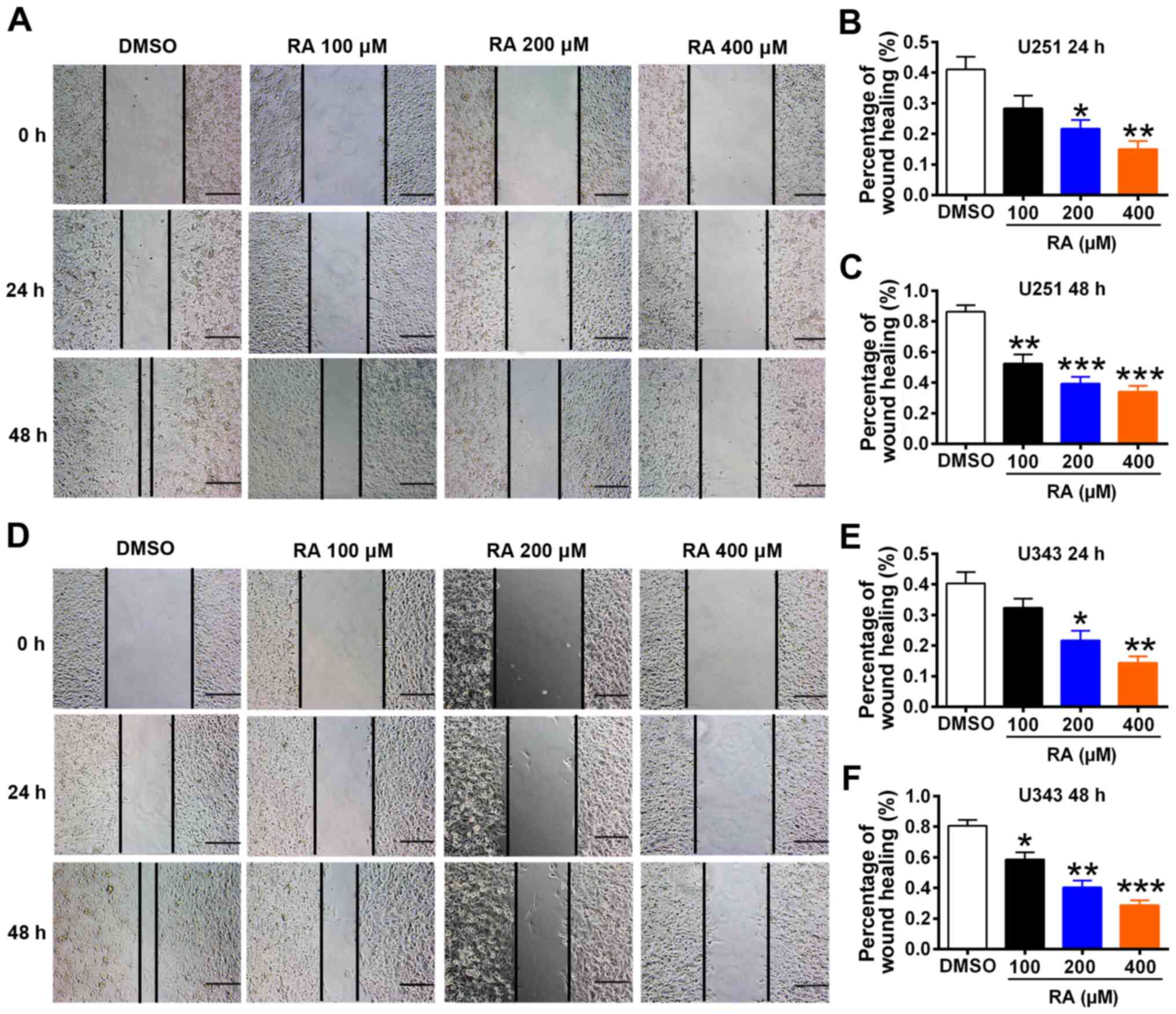
RA inhibits glioma cell migration
Metastasis of rapidly migrating tumor cells is the
main cause of mortality for most patients with cancer (33). Therefore, inhibiting migration
could be an important strategy to prevent tumor metastasis. the
effect of RA on glioma cell migration was examined (Fig. 4). Both U251 and U343 cells
exhibited reduced migration after treatment with RA for 24 or 48 h
at different concentrations (Fig. 4A
and D). Specifically, in the control group of the U251 cell
line, wound healing reached 41.0% after 24 h, while it only reached
28.3, 21.6, and 15.0% after treatment with 100, 200, and 400
µM RA, respectively (Fig.
4B). In the control group of the U343 cell line, wound healing
reached 40.3% after 24 h, while it only reached 32.3, 21.7, and
14.3% after treatment with 100, 200, and 400 µM RA,
respectively (Fig. 4E). At 48 h,
wound healing of control U251 and U343 cells reached 86.3% and
80.6%, respectively; and 52.3, 39.3, and 34.0% (U251 cells;
Fig. 4C) and 58.7, 40.3, and
28.7% (U343 cells; Fig. 4F)
after treatment with 100, 200, and 400 µM RA,
respectively.
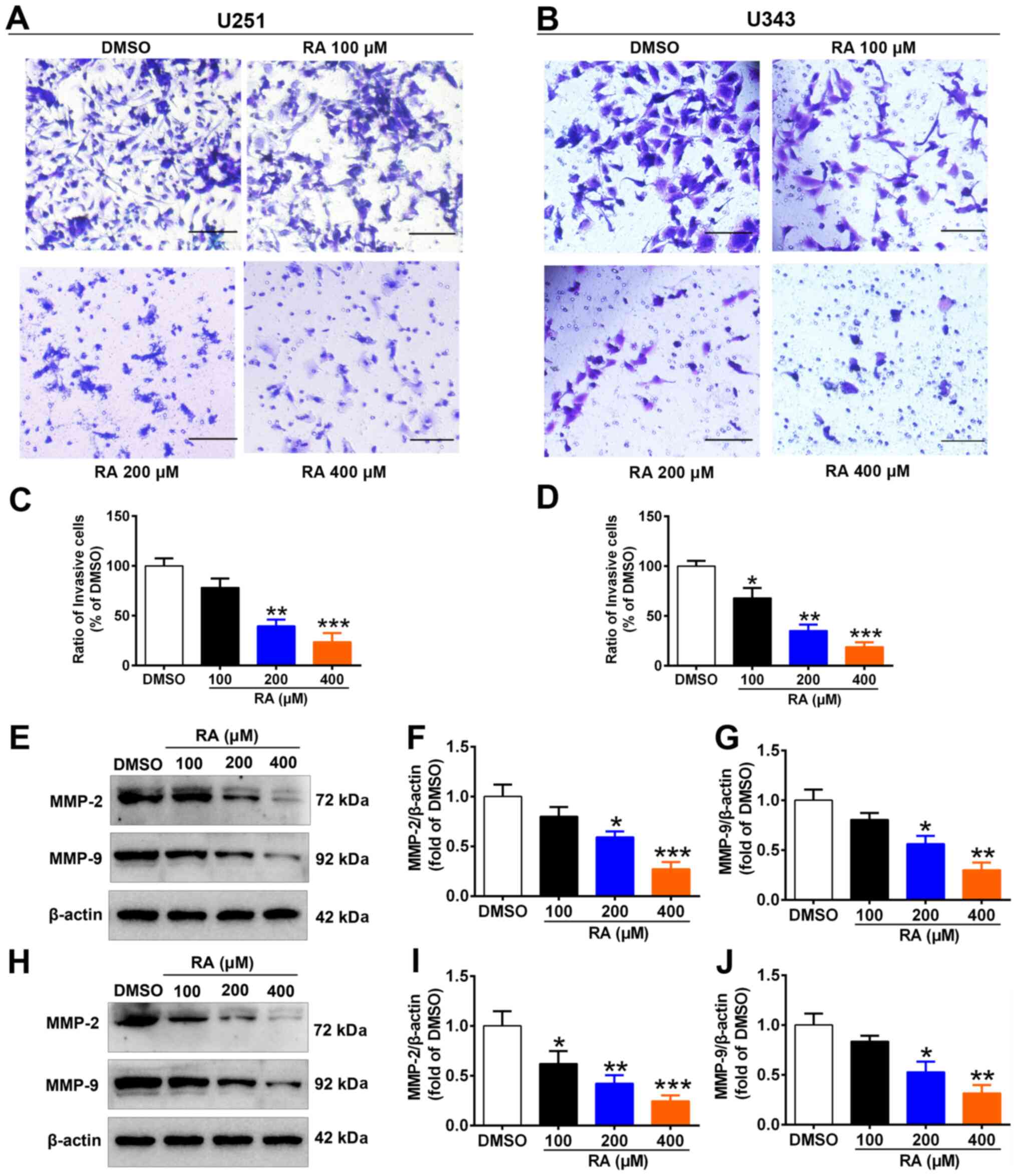
RA inhibits glioma cell invasion
The high invasion capability of glioma cells is the
main reason for the high refractory and recurrence rates of glioma
(34). Therefore, the effect of
RA on glioma cell invasion was assessed using a
Matrigel® invasion assay (Fig. 5). RA significantly inhibited
invasion through the Matrigel® (Fig. 5A and B). Compared with the
control group, the percentage of invading U251 cells was decreased
by 21.7, 60.3, and 76.4% after treatment with 100, 200, and 400
µM RA for 24 h, respectively (Fig. 5C); similarly, the percentage of
invading U343 cells was decreased by 32, 64.7, and 81%,
respectively (Fig. 5D).
Previous studies have reported the role of matrix
metalloproteinase-2 (MMP-2) and MMP-9 in cancer development,
including tumor cell growth, migration, invasion, and metastasis,
and particularly so in glioma (35-37). The expression of MMP-2 and MMP-9
were detected by western blotting. RA dose-dependently inhibited
the expression of MMP-2 and MMP-9 in U251 and U343 cells (Fig. 5E-J). Compared with the control
group, the expression of MMP-2 in U251 cells was decreased by 20.0,
40.7, and 62.7% (Fig. 5F), and
that of MMP-9 was decreased by 19.9, 43.7, and 70% (Fig. 5G), after treatment with 100, 200,
and 400 µM for 24 h, respectively. Similarly, the expression
of MMP-2 in U343 cells decreased by 38.0, 57.7, and 75.3% (Fig. 5I), and that of MMP-9 was
decreased by 16.3, 47.0, and 68.3% (Fig. 5J), after treatment with 100, 200,
and 400 µM for 24 h, respectively. These results indicated
that RA inhibited glioma cell invasion and migration by reducing
the expression of MMPs, thereby providing pathways for the invasion
and metastasis of glioma cells.
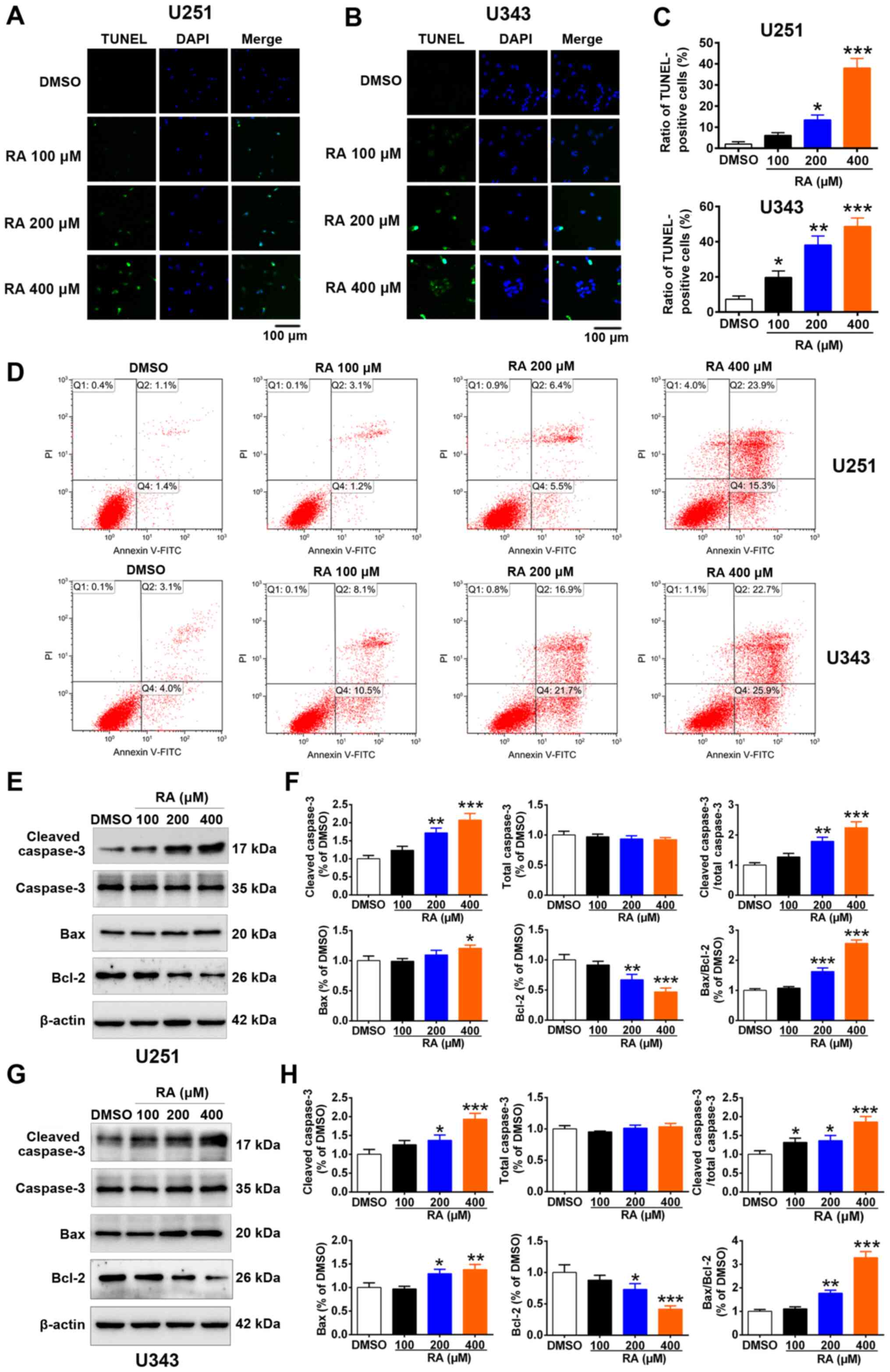
RA increases glioma cell apoptosis
Apoptosis plays a crucial role in cancer treatment
(38). Therefore, whether RA
could induce apoptosis was investigated. A TUNEL assay revealed
that RA significantly induced apoptosis in a dose-dependent manner
in both U251 and U343 cells (Fig.
6A-C). Annexin V/PI dual-staining assays by flow cytometry were
also conducted to detect apoptosis after treatment with RA. As
revealed in Fig. 6D, compared
with the control group, the apoptotic rates of the U251 and U343
cells treated with RA were increased. The apoptosis suppressor
protein Bcl-2 and the pro-apoptotic proteins Bax, cleaved
caspase-3, and caspase-3 are key in the process of apoptosis in
glioma cells (39,40). Caspase-3 is the effector caspase
that initiates cell degradation during the final stage of
apoptosis, and cleaved caspase3 is the active form of caspase3;
Bcl-2, a survival-promoting protein, and Bax, a pro-apoptotic
protein, both are members of the Bcl family that play important
roles in the regulation of intrinsic apoptotic signaling (39,41). It was determined that RA
treatment increased the expression of cleaved caspase-3 and Bax and
reduced the expression of Bcl-2 in both U251 and U343 cells
(Fig. 6E and G). In addition, RA
treatment increased the relative ratio of Bax/Bcl-2 as well as
cleaved caspase-3/caspase-3, indicating RA-induced apoptosis in
glioma cells. Compared with the control group, the relative ratio
of Bax/Bcl-2 in U251 cells was increased by 7.6, 63.0, and 156.3%,
and that in U343 cells was increased by 10.7, 77.7, 228.6%, after
treatment with 100, 200, and 400 µM RA for 24 h,
respectively (Fig. 6F and H). In
addition, the relative ratio of cleaved caspase-3/caspase-3 in U251
cells was increased by 27.3, 79.6, and 124.3%, and that in U343
cells was increased by 29.3, 36.3, and 86%, after treatment with
100, 200, and 400 µM RA for 24 h (Fig. 6F and H).
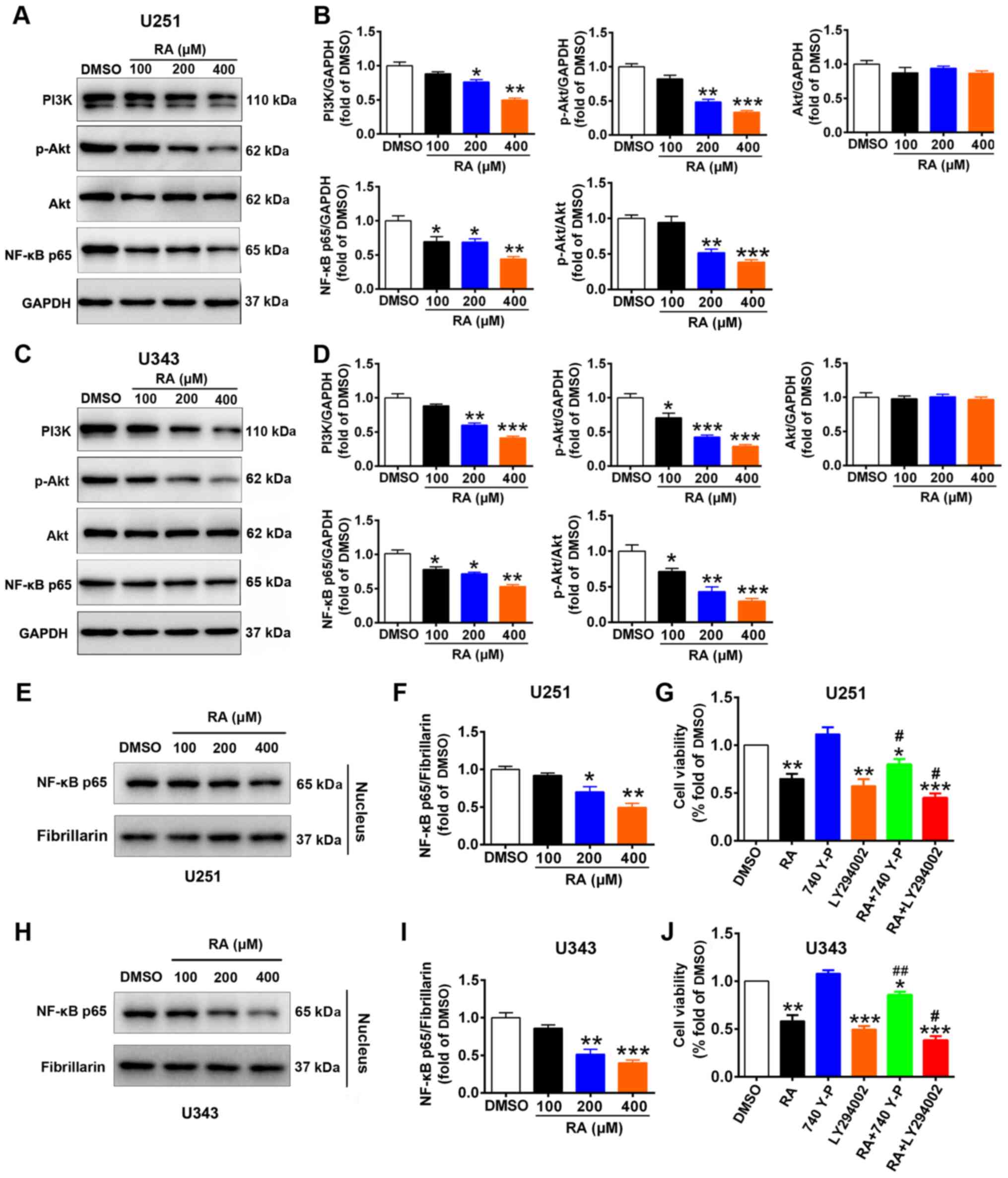
RA inhibits the PI3K/Akt/NF-κB signaling
pathway in glioma cells
PI3K/Akt/NF-κB signaling plays a vital role in cell
proliferation, survival, and metabolism, and is constitutively
activated in most tumors, including glioma (42-44). Therefore, the effect of RA on the
PI3K/Akt/NF-κB signaling pathway was examined. It was revealed that
treatment with 100, 200, and 400 µM RA for 24 h
significantly decreased the protein expression of PI3K, p-Akt, and
NF-κB in U251 and U343 cells compared to control cells (Fig. 7A-D). Specifically, the expression
of PI3K was decreased by 11.7, 24, and 50.3% in U251 cells and by
12.0, 40, and 59% in U343 cells, respectively. In U251 cells, the
expression of p-Akt was decreased by 18.0, 51.7, and 66.7% and that
of NF-κB p65 by 30.7, 31.4, and 56%, respectively. Similarly, in
U343 cells, the expression of p-Akt was decreased by 29.4, 57.7,
and 71.3% and that of NF-κB p65 by 22.0, 28.4, and 47.0%,
respectively. Considering that NF-κB p65 serves as a transcription
factor and oncogene (45), the
effect of RA on nuclear translocation of NF-κB p65 was also
investigated. The results revealed that the expression level of
NF-κB p65 in the nucleus was significantly inhibited following
treatment with RA in U251 and U343 glioma cells (Fig. 7E, F, H and I). To further
investigate the mechanisms underlying RA-mediated inhibition of
U251 and U343 cells, cell viability was performed. The results
revealed that RA significantly inhibited the cell viability of U251
and U343 cells. Moreover, treatment with RA combined with 740 Y-P
reversed the RA-induced deccrease in cell viability, and treatment
with RA combined with LY294002 enhanced the effects of RA on cell
viability (Fig. 7G and J).
Collectively, these results indicated that RA may inhibit
proliferation of glioma cells via the PI3K/Akt/NF-κB signaling
pathway.
Discussion
In the present study, the effects of RA were
explored on the biological characteristics of two glioma cell
lines, U251 and U343. It was revealed that RA inhibited
proliferation, migration, and invasion and induced apoptosis in
both U251 and U343 cells. In general, the effects were
dose-dependent. It was also determined that RA significantly
reduced the protein levels of MMP-2 and MMP-9, which promote
migration and induce invasion, and of the apoptosis suppressor
protein Bcl-2; and increased the levels of pro-apoptotic proteins,
such as cleaved caspase-3 and Bax in both U251 and U343 cells.
Previous research has reported that the PI3K/Akt/NF-κB signaling
pathway plays an important role in the growth, proliferation,
migration, and invasion of glioma (46). The present study further
recognized the importance of PI3K/Akt/NF-κB signaling in
glioma.
Glioma, one of the most common intracranial tumors,
is characterized by rapid growth and high aggressiveness (47). To date, neurosurgery is the
optimal and most direct treatment for gliomas. However,
neurosurgery often fails to completely remove the tumor tissue due
to the highly invasive nature of the glioma, and subsequent
treatment requires a combination of drug chemotherapy and
radiotherapy (48). Radiotherapy
of brain tissue often has great side effects, and chemotherapy
plays a crucial role in the treatment and prognosis of patients
with this cancer (49). However,
the efficacy of current chemotherapy options is not often
satisfactory due to side effects and the development of drug
resistance. Therefore, the identification and development of more
efficient and less toxic drugs for the prevention and treatment of
glioma is essential.
In recent years, the use of traditional Chinese
medicine to treat tumors has attracted increasing attention from
researchers owing to the significant curative efficacy and
relatively fewer side effects (37,50,51). RA is a medicinal herb that not
only is easy to isolate, but can also be ingested from food or tea
(52). RA has been reported to
have antioxidant, antiviral, and anti-inflammatory effects and has
been used in an Egyptian herbal tea for the prevention of cancer
(53). Furthermore, RA was
revealed to significantly improve the efficiency of radiotherapy by
exerting a radiosensitizing effect on tumor cells (54). In addition, it enhanced
chemosensitivity of resistant gastric carcinoma cells to 5-Fu by
downregulating miR-642a-3p and miR-6785-5p and increasing
FOXO4 expression (28). Various studies have revealed that
RA has antineoplastic activity in leukemia, hepatocellular
carcinoma, gastric carcinoma, colorectal cancer, breast cancer, and
small-cell carcinoma of the lung (26-29). However, the effects of RA on the
biological characteristics of glioma is limited.
The present study revealed that RA inhibited the
expression of Fyn in a dose-dependent manner in U251 and U343
cells. This provides a theoretical basis for its antitumor effects
in glioma. High invasion and rapid proliferation are the main
reasons for refractory glioma and poor prognosis of glioma
(34). Cell viability assay and
morphological observation revealed that RA inhibited the growth of
glioma in a time- and dose-dependent manner. Concurrently, it had
little effect on NHA cell proliferation and morphology. This
suggested that RA may be a promising antitumor drug based on its
weak toxicity. The present study also demonstrated that RA
inhibited glioma cell invasion. Since MMP-2 and MMP-9 are important
regulators of invasion, the effect of RA on their expression levels
was also examined. Western blotting revealed that treatment with RA
decreased MMP-2 and MMP-9 expression in glioma cells. The main
feature of tumor metastasis is the migration of cancer cells from
the initial tumor site to the circulatory system or lymphatic
system (55). Therefore,
inhibiting tumor cell migration could reduce metastasis. In the
present study, RA inhibited migration of U251 and U343 cells.
Considering that apoptosis is a crucial antitumor mechanism
(56), it was investigated
whether RA could induce apoptosis in glioma cells. The percentage
of apoptotic U251 and U343 cells was significantly increased after
treatment with RA. As Bcl-family proteins are main regulators of
apoptosis (57), the effect of
RA on the expression of Bax and Bcl-2 was also examined. RA
increased Bax expression and decreased Bcl-2 expression in glioma
cells. Correspondingly, the expression of cleaved (activated)
caspase-3, but not that of total caspase-3, was increased by RA. In
addition, the Bax/Bcl-2 and cleaved caspase-3/caspase-3 ratios were
increased by RA, thus providing a mechanistic basis for the
induction of apoptosis by RA in glioma cells. These results are
similar to the reported role of RA in colorectal cancer (37). Numerous studies have revealed
that the PI3K/Akt/NF-κB pathway is closely related to glioma
progression (46,58,59). In line with these studies, the
present results revealed that the PI3K/Akt/NF-κB signaling pathway
was involved in the antitumor effects of RA in glioma.
Collectively, these findings indicated that the
antitumor effects of RA in glioma may be mediated by Fyn
inhibition. The detailed mechanisms warrant further investigation
in vitro and in vivo.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
BH and GH conceived and designed the study. YL, XX,
HT and YP performed the experiments. YL, XX and HT analyzed the
data. YL, BH and GH wrote the manuscript. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no conflicts of
interest.
Acknowledgments
Not applicable.
Funding
The present research was funded by China Postdoctoral Science
Foundation (grant no. 2019M663104) and the Science and Technology
Planning Project of Guangdong Province (grant no.
2017A020215089).
References
|
1
|
Lapointe S, Perry A and Butowski NA:
Primary brain tumours in adults. Lancet. 392:432–446. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Senders JT, Harary M, Stopa BM, Staples P,
Broekman MLD, Smith TR, Gormley WB and Arnaout O: Information-based
medicine in glioma patients: A clinical perspective. Comput Math
Methods Med. 2018:85720582018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Batash R, Asna N, Schaffer P, Francis N
and Schaffer M: Glioblastoma multiforme, diagnosis and treatment;
recent literature review. Curr Med Chem. 24:3002–3009. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stupp R, Mason WP, van den Bent MJ, Weller
M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn
U, et al: Radiotherapy plus concomitant and adjuvant temozolomide
for glioblastoma. N Engl J Med. 352:987–996. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang S, Liao K, Miao Z, Wang Q, Miao Y,
Guo Z, Qiu Y, Chen B, Ren L, Wei Z, et al: CircFOXO3 promotes
glioblastoma progression by acting as a competing endogenous RNA
for NFAT5. Neuro Oncol. 21:1284–1296. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
El-Sehemy A, Selvadurai H, Ortin-Martinez
A, Pokrajac N, Mamatjan Y, Tachibana N, Rowland K, Lee L, Park N,
Aldape K, et al: Norrin mediates tumor-promoting and -suppressive
effects in glioblastoma via Notch and Wnt. J Clin Invest.
130:3069–3086. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lewis-Tuffin LJ, Feathers R, Hari P,
Durand N, Li Z, Rodriguez FJ, Bakken K, Carlson B, Schroeder M,
Sarkaria JN and Anastasiadis PZ: Src family kinases differentially
influence glioma growth and motility. Mol Oncol. 9:1783–1798. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Irby RB and Yeatman TJ: Role of Src
expression and activation in human cancer. Oncogene. 19:5636–5642.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Summy JM and Gallick GE: Src family
kinases in tumor progression and metastasis. Cancer Metastasis Rev.
22:337–358. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Frame MC: Src in cancer: Deregulation and
consequences for cell behaviour. Biochim Biophys Acta.
1602:114–130. 2002.PubMed/NCBI
|
|
11
|
Tornillo G, Knowlson C, Kendrick H, Cooke
J, Mirza H, Aurrekoetxea-Rodríguez I, Vivanco MDM, Buckley NE,
Grigoriadis A and Smalley MJ: Dual mechanisms of LYN kinase
dysregulation drive aggressive behavior in breast cancer cells.
Cell Rep. 25:3674–3692.e10. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Emaduddin M, Bicknell DC, Bodmer WF and
Feller SM: Cell growth, global phosphotyrosine elevation, and c-Met
phosphorylation through Src family kinases in colorectal cancer
cells. Proc Natl Acad Sci USA. 105:2358–2362. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Masaki T, Okada M, Tokuda M, Shiratori Y,
Hatase O, Shirai M, Nishioka M and Omata M: Reduced C-terminal Src
kinase (Csk) activities in hepatocellular carcinoma. Hepatology.
29:379–384. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yadav V and Denning MF: Fyn is induced by
Ras/PI3K/Akt signaling and is required for enhanced
invasion/migration. Mol Carcinog. 50:346–352. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lu KV, Zhu S, Cvrljevic A, Huang TT,
Sarkaria S, Ahkavan D, Dang J, Dinca EB, Plaisier SB, Oderberg I,
et al: Fyn and SRC are effectors of oncogenic epidermal growth
factor receptor signaling in glioblastoma patients. Cancer Res.
69:6889–6898. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Han X, Zhang W, Yang X, Wheeler CG,
Langford CP, Wu L, Filippova N, Friedman GK, Ding Q,
Fathallah-Shaykh HM, et al: The role of Src family kinases in
growth and migration of glioma stem cells. Int J Oncol. 45:302–310.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Comba A, Dunn PJ, Argento AE, Kadiyala P,
Ventosa M, Patel P, Zamler DB, Núñez FJ, Zhao L, Castro MG and
Lowenstein PR: Fyn tyrosine kinase, a downstream target of receptor
tyrosine kinases, modulates antiglioma immune responses. Neuro
Oncol. 22:806–818. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bowman RL, Wang Q, Carro A, Verhaak RG and
Squatrito M: GlioVis data portal for visualization and analysis of
brain tumor expression datasets. Neuro Oncol. 19:139–141. 2017.
View Article : Google Scholar :
|
|
19
|
Jelić D, Mildner B, Kostrun S, Nujić K,
Verbanac D, Culić O, Antolović R and Brandt W: Homology modeling of
human Fyn kinase structure: Discovery of rosmarinic acid as a new
Fyn kinase inhibitor and in silico study of its possible binding
modes. J Med Chem. 50:1090–1100. 2007. View Article : Google Scholar
|
|
20
|
Şengelen A and Önay-Uçar E: Rosmarinic
acid and siRNA combined therapy represses Hsp27 (HSPB1) expression
and induces apoptosis in human glioma cells. Cell Stress
Chaperones. 23:885–896. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Juskowiak B, Bogacz A, Wolek M, Kamiński
A, Uzar I, Seremak-Mrozikiewicz A and Czerny B: Expression
profiling of genes modulated by rosmarinic acid (RA) in MCF-7
breast cancer cells. Ginekol Pol. 89:541–545. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang SY, Hong CO, Lee GP, Kim CT and Lee
KW: The hepatoprotection of caffeic acid and rosmarinic acid, major
compounds of Perilla frutescens, against t-BHP-induced oxidative
liver damage. Food Chem Toxicol. 55:92–99. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chu X, Ci X and He J, Jiang L, Wei M, Cao
Q, Guan M, Xie X, Deng X and He J: Effects of a natural prolyl
oligopeptidase inhibitor, rosmarinic acid, on
lipopolysaccharide-induced acute lung injury in mice. Molecules.
17:3586–3598. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lin CS, Kuo CL, Wang JP, Cheng JS, Huang
ZW and Chen CF: Growth inhibitory and apoptosis inducing effect of
Perilla frutescens extract on human hepatoma HepG2 cells. J
Ethnopharmaco. 112:557–567. 2007. View Article : Google Scholar
|
|
25
|
Kelsey NA, Wilkins HM and Linseman DA:
Nutraceutical antioxidants as novel neuroprotective agents.
Molecules. 15:7792–7814. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wu CF, Hong C, Klauck SM, Lin YL and
Efferth T: Molecular mechanisms of rosmarinic acid from Salvia
miltiorrhiza in acute lymphoblastic leukemia cells. J
Ethnopharmaco. 176:55–68. 2015. View Article : Google Scholar
|
|
27
|
Xavier CP, Lima CF, Fernandes-Ferreira M
and Pereira-Wilson C: Salvia fruticosa, Salvia officinalis, and
rosmarinic acid induce apoptosis and inhibit proliferation of human
colorectal cell lines: The role in MAPK/ERK pathway. Nutr Cancer.
61:564–571. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yu C, Chen DQ, Liu HX, Li WB, Lu JW and
Feng JF: Rosmarinic acid reduces the resistance of gastric
carcinoma cells to 5-fluorouracil by downregulating FOXO4-targeting
miR-6785-5p. Biomed Pharmacother. 109:2327–2334. 2019. View Article : Google Scholar
|
|
29
|
Yesil-Celiktas O, Sevimli C, Bedir E and
Vardar-Sukan F: Inhibitory effects of rosemary extracts, carnosic
acid and rosmarinic acid on the growth of various human cancer cell
lines. Plant Foods Hum Nutr. 65:158–163. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li W, Du Q, Li X, Zheng X, Lv F, Xi X,
Huang G, Yang J and Liu S: Eriodictyol Inhibits Proliferation,
Metastasis and induces apoptosis of glioma cells via PI3K/Akt/NF-κB
signaling pathway. Front Pharmacol. 11:1142020. View Article : Google Scholar
|
|
31
|
Ko HM, Lee SH, Kim KC, Joo SH, Choi WS and
Shin CY: The role of TLR4 and fyn interaction on
lipopolysaccharide-stimulated PAI-1 expression in astrocytes. Mol
Neurobiol. 52:8–25. 2015. View Article : Google Scholar
|
|
32
|
Zhang T, Ji D, Wang P, Liang D, Jin L, Shi
H, Liu X, Meng Q, Yu R and Gao S: The atypical protein kinase RIOK3
contributes to glioma cell proliferation/survival,
migration/invasion and the AKT/mTOR signaling pathway. Cancer Lett.
415:151–163. 2018. View Article : Google Scholar
|
|
33
|
Yachida S, Jones S, Bozic I, Antal T,
Leary R, Fu B, Kamiyama M, Hruban RH, Eshleman JR, Nowak MA, et al:
Distant metastasis occurs late during the genetic evolution of
pancreatic cancer. Nature. 467:1114–1117. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Marín-Ramos NI, Thein TZ, Cho HY, Swenson
SD, Wang W, Schönthal AH, Chen TC and Hofman FM: NEO212 inhibits
migration and invasion of glioma stem cells. Mol Cancer Ther.
17:625–637. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pan H, Xue W, Zhao W and Schachner M:
Expression and function of chondroitin 4-sulfate and chondroitin
6-sulfate in human glioma. FASEB J. 34:2853–2868. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Szabo E, Schneider H, Seystahl K, Rushing
EJ, Herting F, Weidner KM and Weller M: Autocrine VEGFR1 and VEGFR2
signaling promotes survival in human glioblastoma models in vitro
and in vivo. Neuro Oncol. 18:1242–1252. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Han YH, Kee JY and Hong SH: Rosmarinic
Acid Activates AMPK to inhibit metastasis of colorectal cancer.
Front Pharmacol. 9:682018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Niknejad H, Yazdanpanah G and Ahmadiani A:
Induction of apoptosis, stimulation of cell-cycle arrest and
inhibition of angiogenesis make human amnion-derived cells
promising sources for cell therapy of cancer. Cell Tissue Res.
363:599–608. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang Y, Tang H, Zhang Y, Li J, Li B, Gao
Z, Wang X, Cheng G and Fei Z: Saponin B, a novel cytostatic
compound purified from Anemone taipaiensis, induces apoptosis in a
human glioblastoma cell line. Int J Mol Med. 32:1077–1084. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Su CC, Wang MJ and Chiu TL: The
anti-cancer efficacy of curcumin scrutinized through core signaling
pathways in glioblastoma. Int J Mol Med. 26:217–224.
2010.PubMed/NCBI
|
|
41
|
Zhang WF, Yang Y, Li X, Xu DY, Yan YL, Gao
Q, Jia AL and Duan MH: Angelica polysaccharides inhibit the growth
and promote the apoptosis of U251 glioma cells in vitro and in
vivo. Phytomedicine. 33:21–27. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yang SX, Polley E and Lipkowitz S: New
insights on PI3K/AKT pathway alterations and clinical outcomes in
breast cancer. Cancer Treat Rev. 45:87–96. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Arlt A, Gehrz A, Müerköster S, Vorndamm J,
Kruse ML, Fölsch UR and Schäfer H: Role of NF-kappaB and Akt/PI3K
in the resistance of pancreatic carcinoma cell lines against
gemcitabine-induced cell death. Oncogene. 22:3243–3251. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tomar VS, Patil V and Somasundaram K:
Temozolomide induces activation of Wnt/β-catenin signaling in
glioma cells via PI3K/Akt pathway: Implications in glioma therapy.
Cell Biol Toxicol. 36:273–278. 2020. View Article : Google Scholar
|
|
45
|
Song L, Chen X, Mi L, Liu C, Zhu S, Yang
T, Luo X, Zhang Q, Lu H and Liang X: Icariin-induced inhibition of
SIRT6/NF-κB triggers redox mediated apoptosis and enhances
anti-tumor immunity in triple-negative breast cancer. Cancer Sci.
111:4242–4256. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Fahey JM, Korytowski W and Girotti AW:
Upstream signaling events leading to elevated production of
pro-survival nitric oxide in photodynamically-challenged
glioblastoma cells. Free Radic Biol Med. 137:37–45. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Furnari FB, Fenton T, Bachoo RM, Mukasa A,
Stommel JM, Stegh A, Hahn WC, Ligon KL, Louis DN, Brennan C, et al:
Malignant astrocytic glioma: Genetics, biology, and paths to
treatment. Genes Dev. 21:2683–2710. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Karachi A, Dastmalchi F, Mitchell DA and
Rahman M: Temozolomide for immunomodulation in the treatment of
glioblastoma. Neuro Oncol. 20:1566–1572. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Patil SA, Hosni-Ahmed A, Jones TS, Patil
R, Pfeffer LM and Miller DD: Novel approaches to glioma drug design
and drug screening. Expert Opin Drug Discov. 8:1135–1151. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xie JH, Lai ZQ, Zheng XH, Xian YF, Li Q,
Ip SP, Xie YL, Chen JN, Su ZR, Lin ZX and Yang XB: Apoptosis
induced by bruceine D in human non-small-cell lung cancer cells
involves mitochondrial ROS-mediated death signaling. Int J Mol Med.
44:2015–2026. 2019.PubMed/NCBI
|
|
51
|
Ong SKL, Shanmugam MK, Fan L, Fraser SE,
Arfuso F, Ahn KS, Sethi G and Bishayee A: Focus on formononetin:
Anticancer potential and molecular targets. Cancers. 11:6112019.
View Article : Google Scholar :
|
|
52
|
Petersen M and Simmonds MS: Rosmarinic
acid. Phytochemistry. 62:121–125. 2003. View Article : Google Scholar
|
|
53
|
Elansary HO and Mahmoud EA: Egyptian
herbal tea infusions' antioxidants and their antiproliferative and
cytotoxic activities against cancer cells. Nat Prod Res.
29:474–479. 2015. View Article : Google Scholar
|
|
54
|
Alcaraz M, Alcaraz-Saura M, Achel DG,
Olivares A, López- Morata JA and Castillo J: Radiosensitizing
effect of rosmarinic acid in metastatic melanoma B16F10 cells.
Anticancer Res. 34:1913–1921. 2014.PubMed/NCBI
|
|
55
|
McCall B, McPartland CK, Moore R,
Frank-Kamenetskii A and Booth BW: Effects of astaxanthin on the
proliferation and migration of breast cancer cells in vitro.
Antioxidants (Basel). 7:1352018. View Article : Google Scholar
|
|
56
|
Shin J, Song MH, Oh JW, Keum YS and Saini
RK: Pro-oxidant actions of carotenoids in triggering apoptosis of
cancer cells: A review of emerging evidence. Antioxidants (Basel).
9:5322020. View Article : Google Scholar
|
|
57
|
Chowdhury I, Thompson WE, Welch C, Thomas
K and Matthews R: Prohibitin (PHB) inhibits apoptosis in rat
granulosa cells (GCs) through the extracellular signal-regulated
kinase 1/2 (ERK1/2) and the Bcl family of proteins. Apoptosis.
18:1513–1525. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Yan J, Xu C, Li Y, Tang B, Xie S, Hong T
and Zeng E: Long non-coding RNA LINC00526 represses glioma
progression via forming a double negative feedback loop with AXL. J
Cell Mol Med. 23:5518–5531. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Nakabayashi H and Shimizu K: Involvement
of Akt/NF-κB pathway in antitumor effects of parthenolide on
glioblastoma cells in vitro and in vivo. BMC Cancer. 12:4532012.
View Article : Google Scholar
|