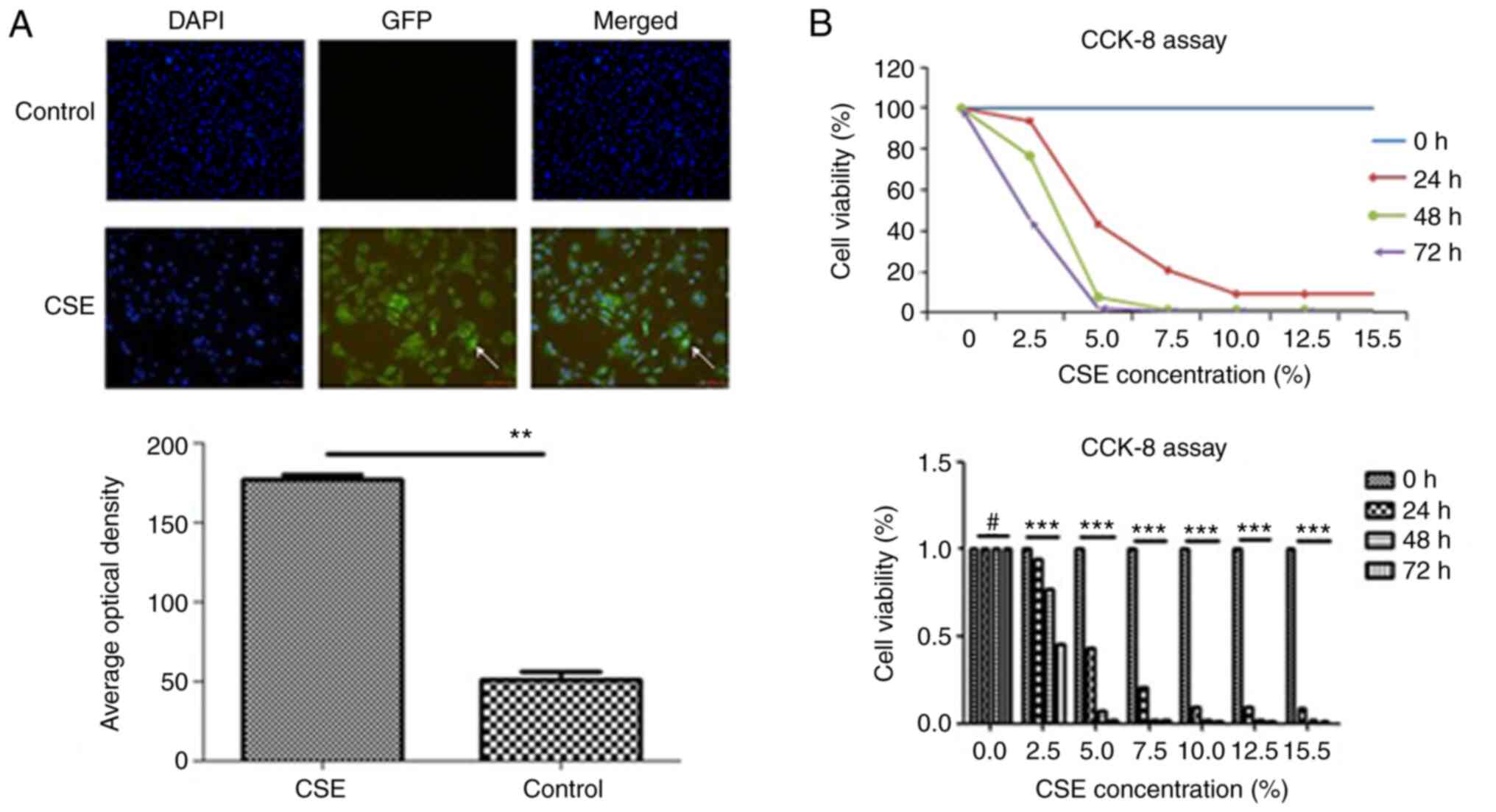
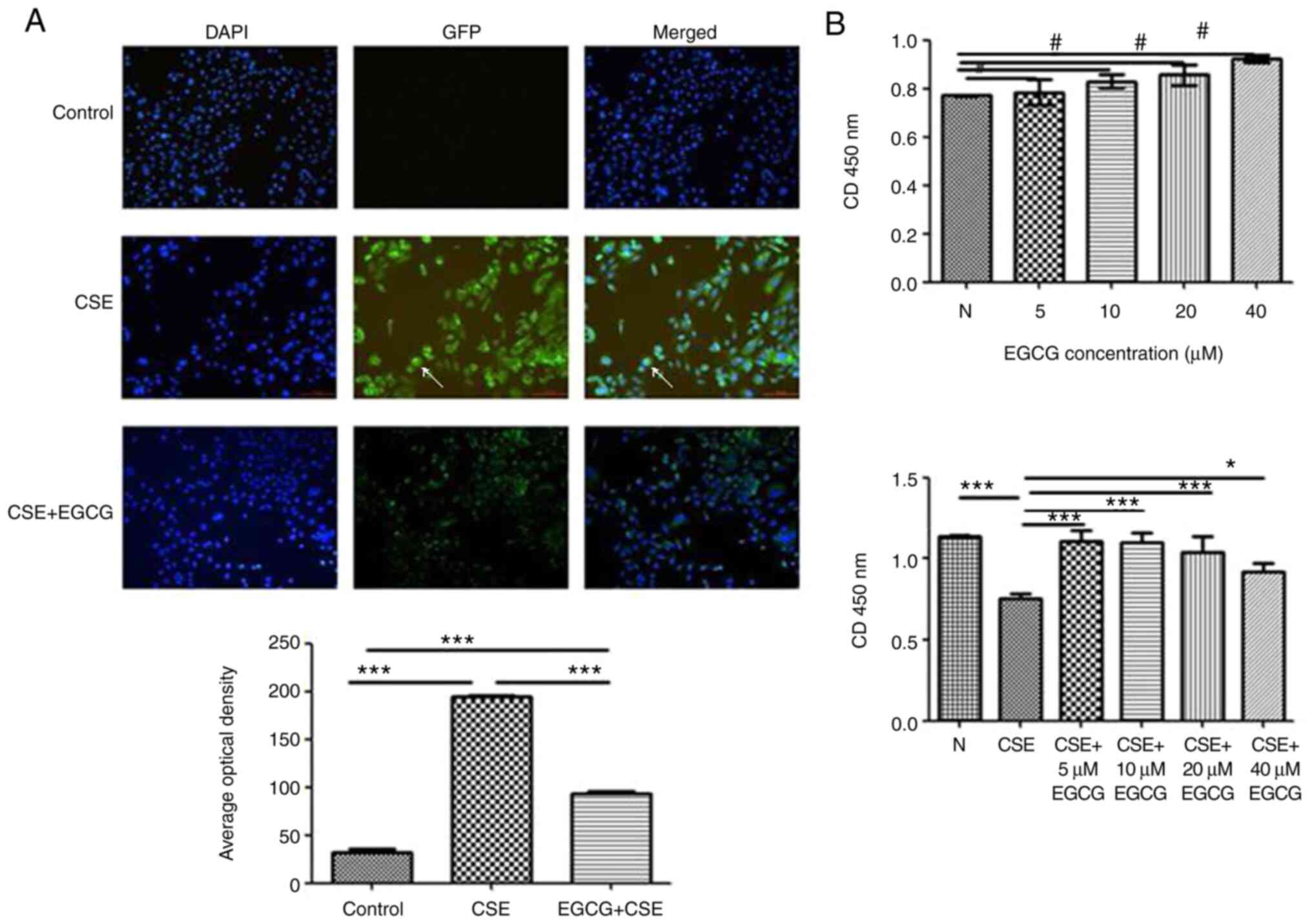
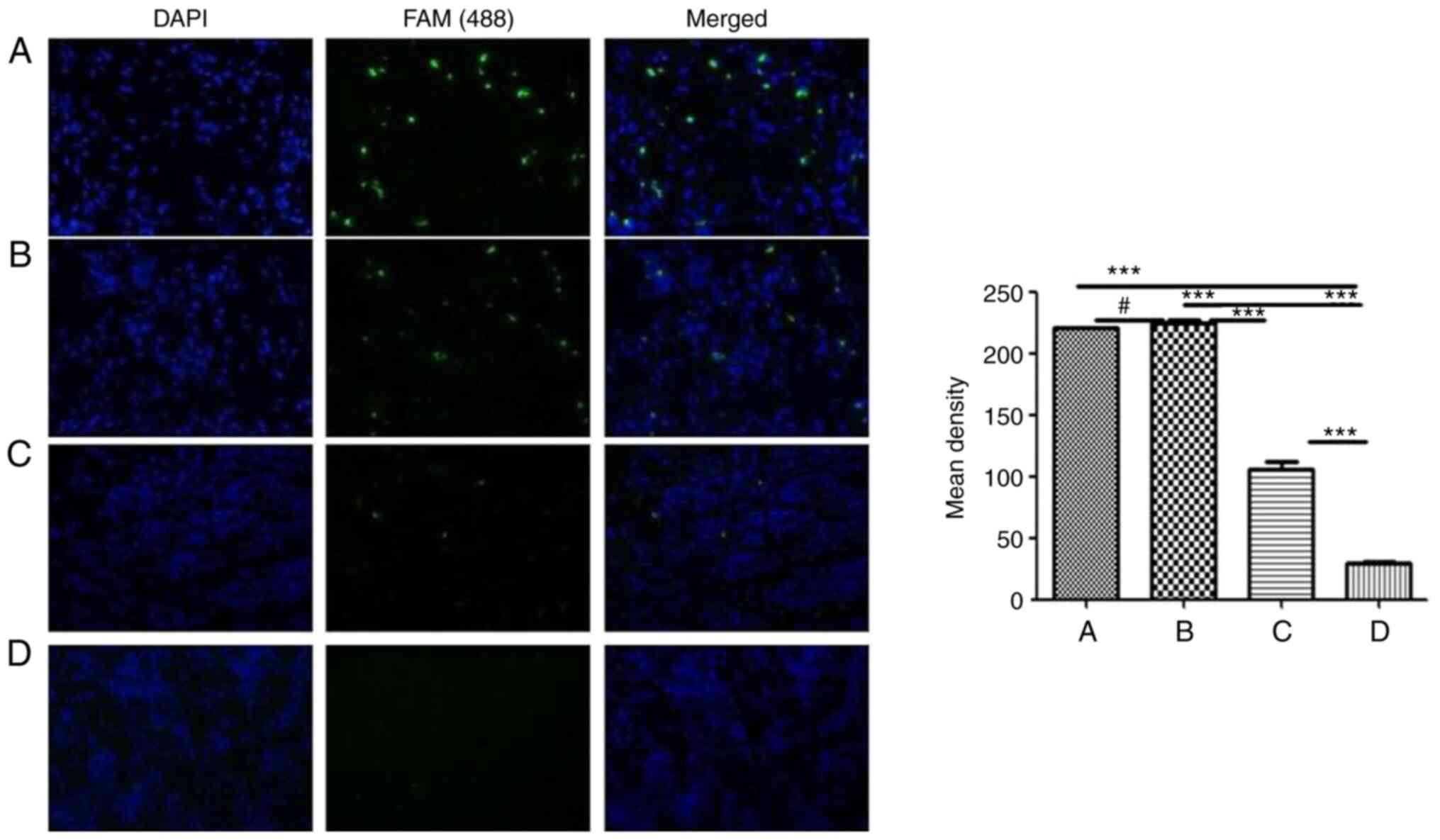
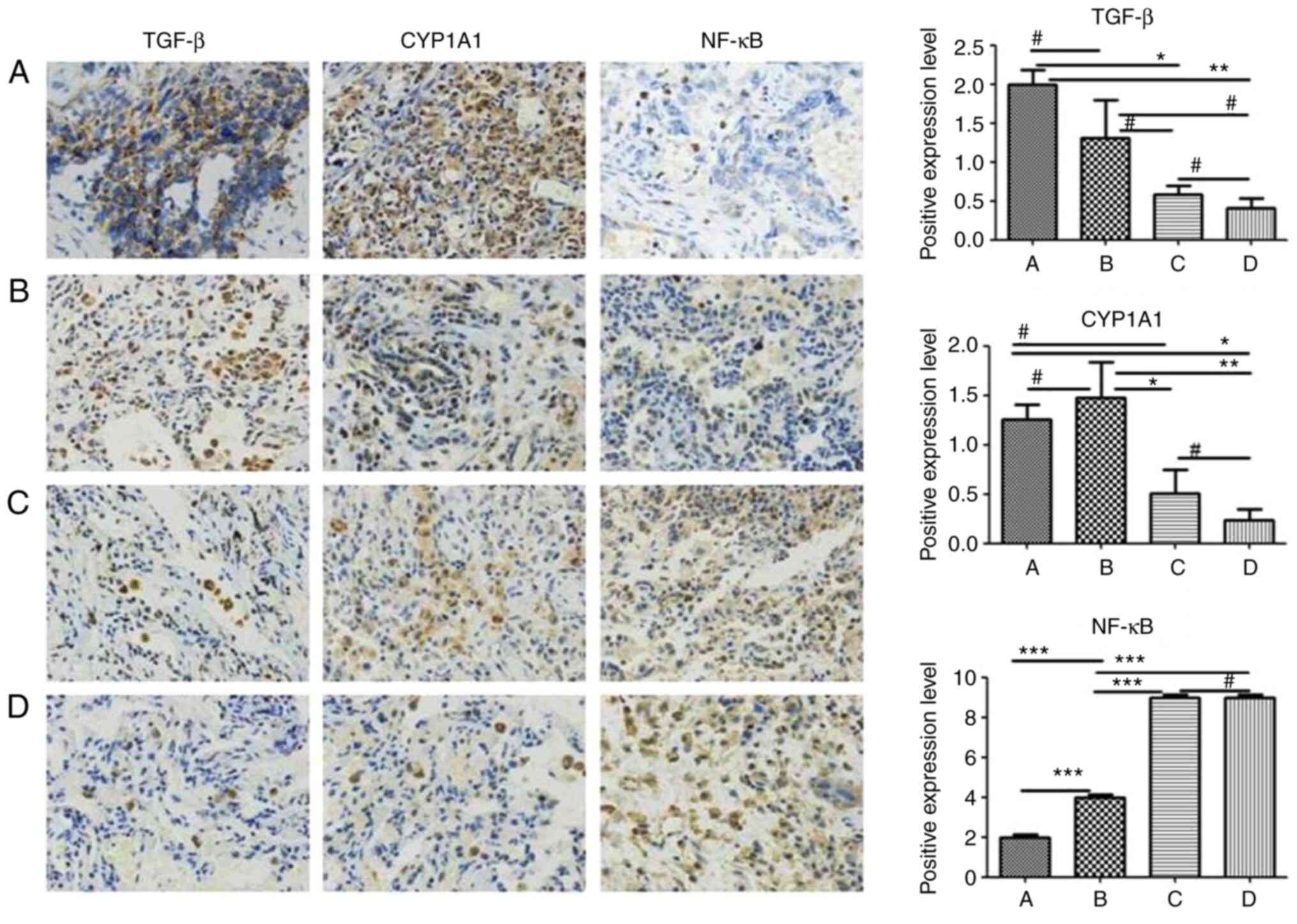
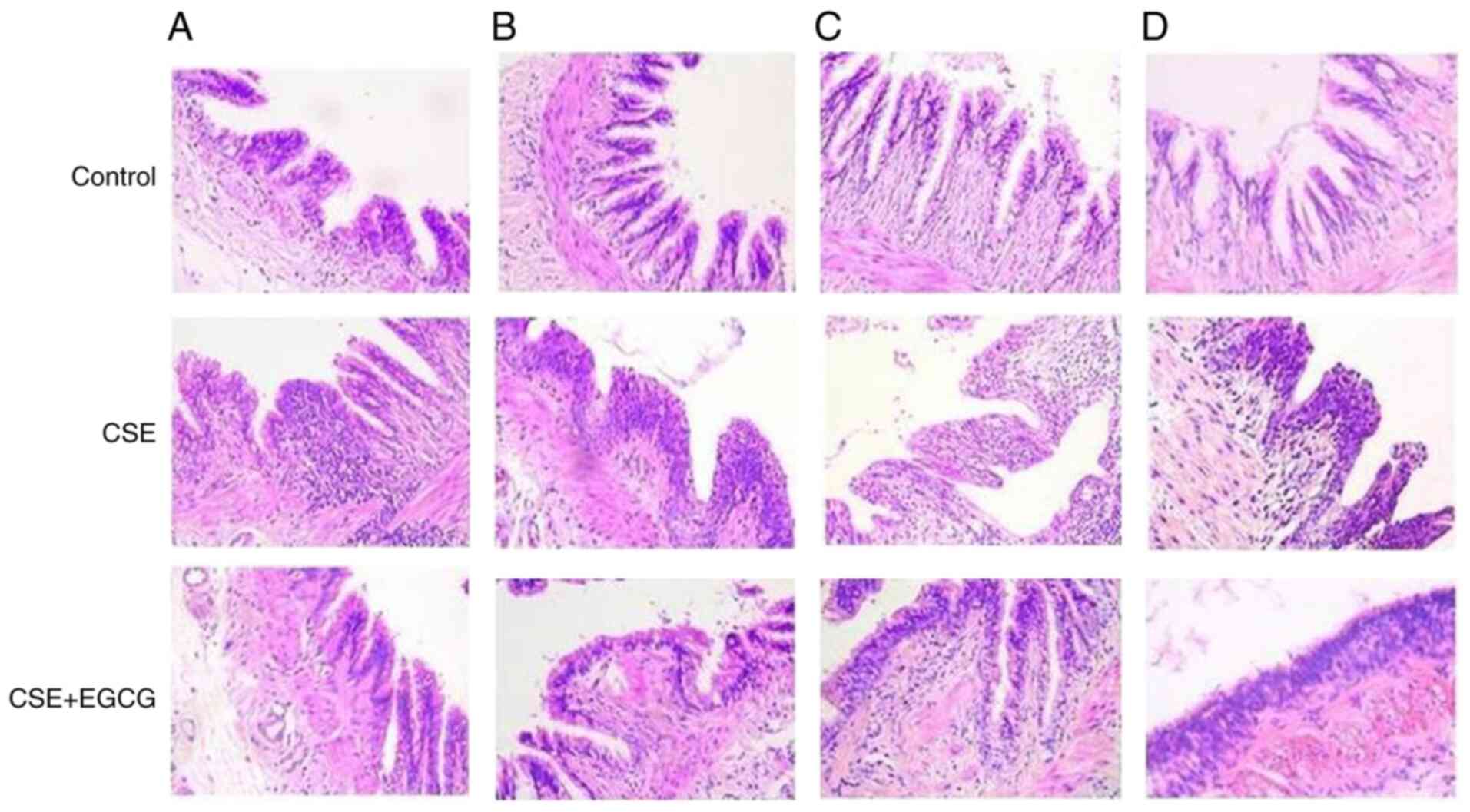
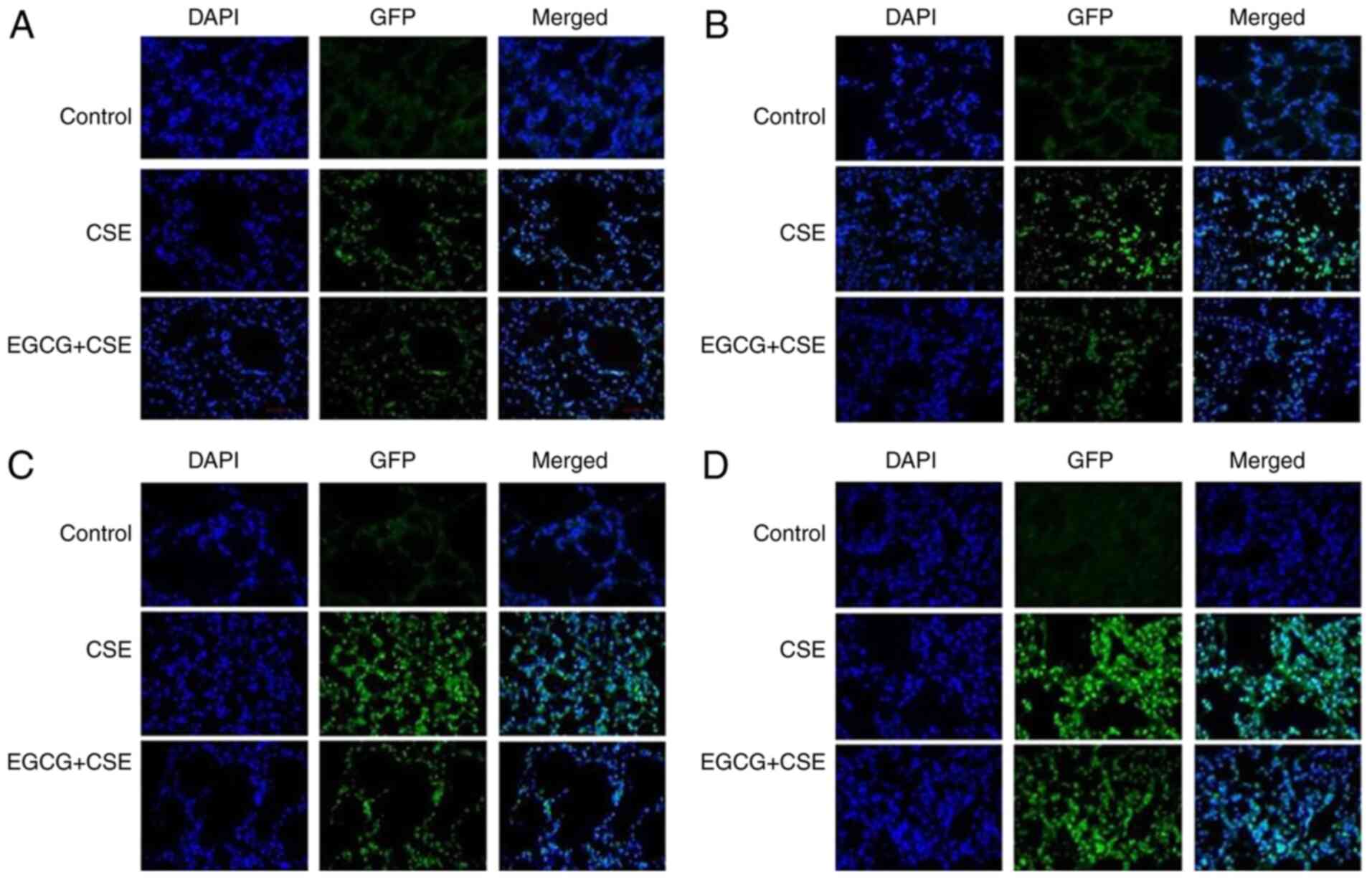
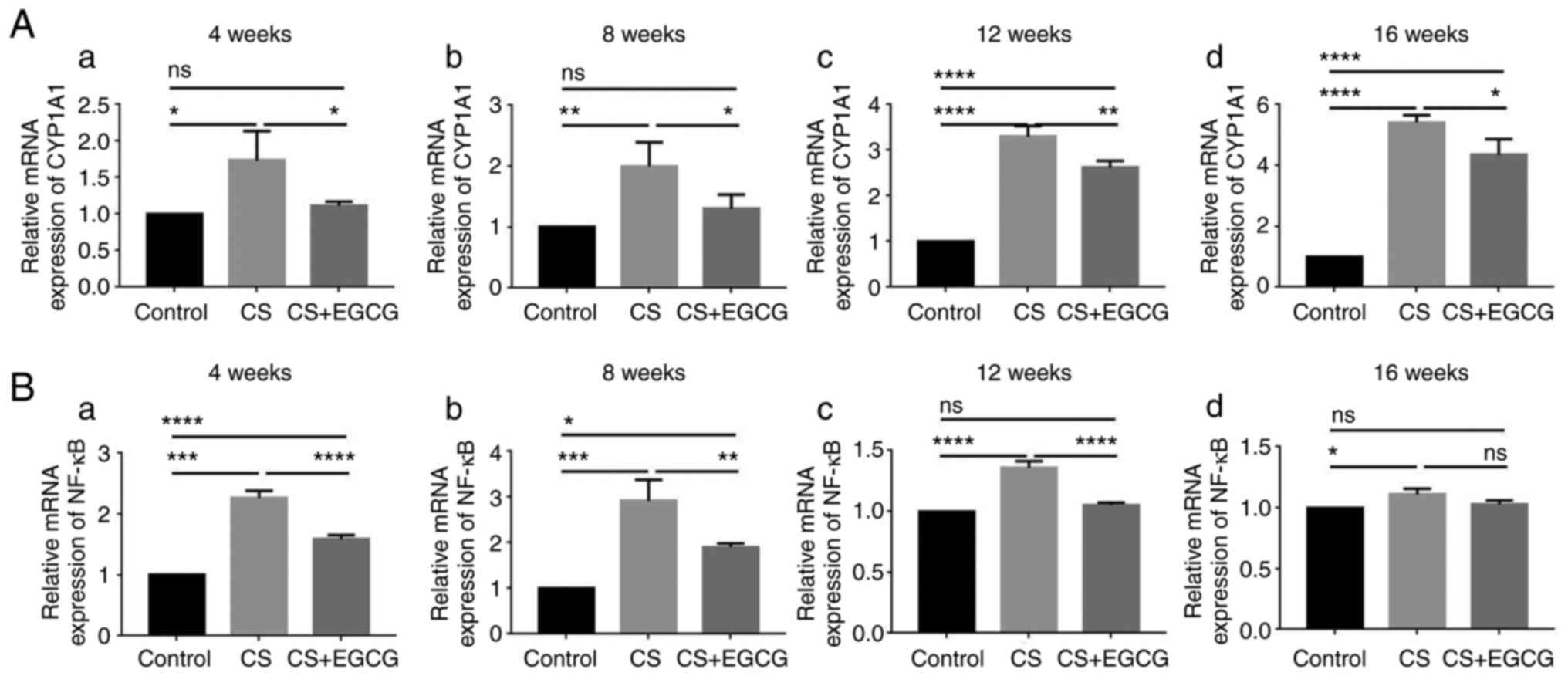
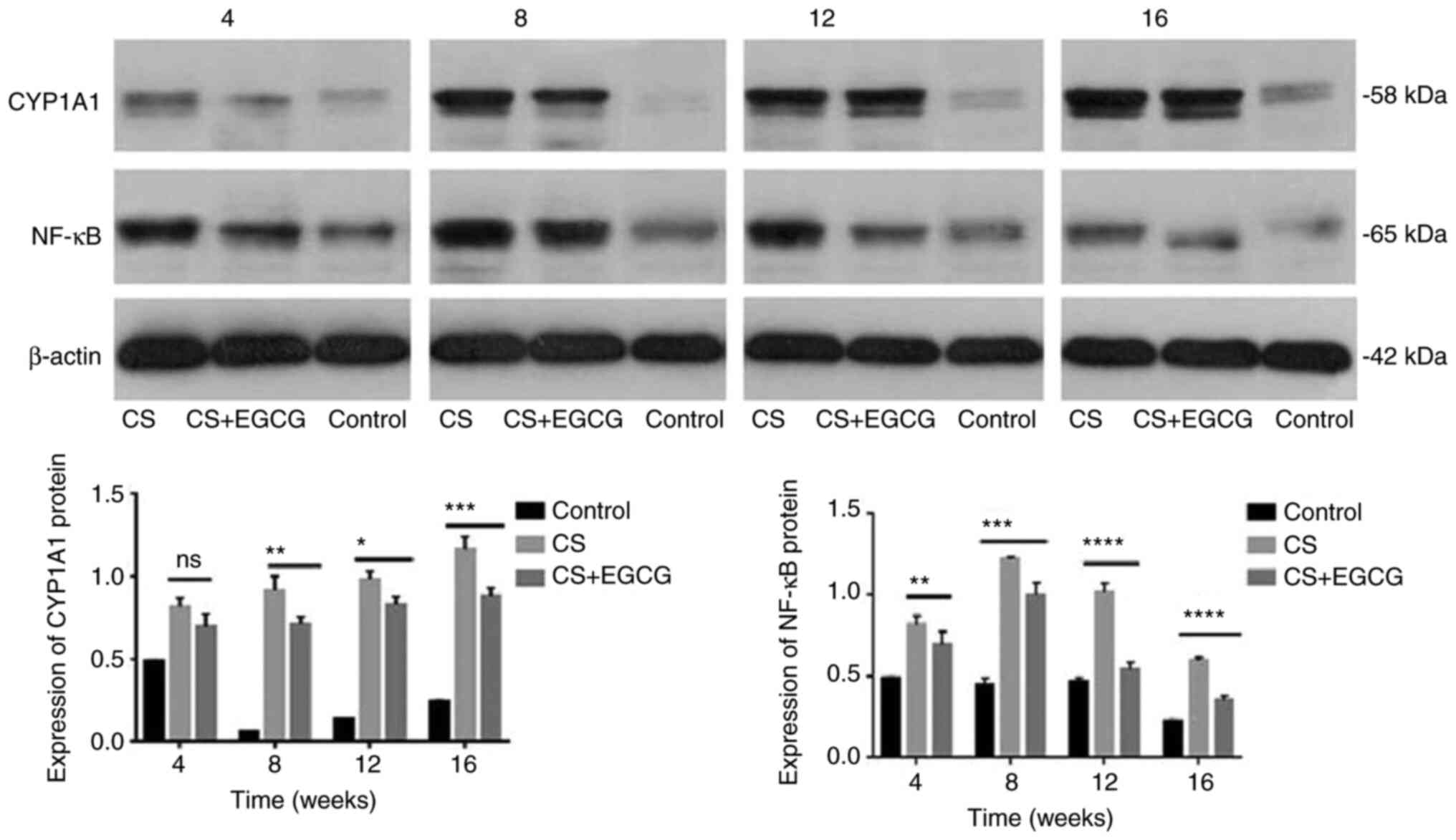
|
1
|
Tu CY, Cheng FJ, Chen CM, Wang SL, Hsiao
YC, Chen CH, Hsia TC, He YH, Wang BW, Hsieh IS, et al: Cigarette
smoke enhances oncogene addiction to c-MET and desensitizes
EGFR-expressing non-small cell lung cancer to EGFR TKIs. Mol Oncol.
12:705–723. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Raja R, Sahasrabuddhe NA, Radhakrishnan A,
Syed N, Solanki HS, Puttamallesh VN, Balaji SA, Nanjappa V, Datta
KK, Babu N, et al: Chronic exposure to cigarette smoke leads to
activation of p21 (RAC1)-activated kinase 6 (PAK6) in non-small
cell lung cancer cells. Oncotarget. 7:61229–61245. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Thai P, Statt S, Chen CH, Liang E,
Campbell C and Wu R: Characterization of a novel long noncoding
RNA, SCAL1, induced by cigarette smoke and elevated in lung cancer
cell lines. Am J Respir Cell Mol Biol. 49:204–211. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Billatos E, Faiz A, Gesthalter Y, LeClerc
A, Alekseyev YO, Xiao X, Liu G, Ten Hacken NHT, Heijink IH, Timens
W, et al: Impact of acute exposure to cigarette smoke on airway
gene expression. Physiol Genomics. 50:705–713. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gu Q, Hu C, Chen N and Qu J: A comparison
between lung carcinoma and a subcutaneous malignant tumor induced
in rats by a 3,4-benzopyrene injection. Int J Clin Exp Pathol.
11:3934–3942. 2018.PubMed/NCBI
|
|
6
|
Sá VK, Rocha TP, Moreira A, Soares FA,
Takagaki T, Carvalho L, Nicholson AG and Capelozzi VL:
Hyaluronidases and hyaluronan synthases expression is inversely
correlated with malignancy in lung/bronchial pre-neoplastic and
neoplastic lesions, affecting prognosis. Braz J Med Biol Res.
48:1039–1047. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rushing BR and Selim MI: Aflatoxin B1: A
review on metabolism, toxicity, occurrence in food, occupational
exposure, and detoxification methods. Food Chem Toxicol.
124:81–100. 2019. View Article : Google Scholar
|
|
8
|
Gavish M, Cohen S and Nagler R: Cigarette
smoke effects on TSPO and VDAC expression in a cellular lung cancer
model. Eur J Cancer Prev. 25:361–367. 2016. View Article : Google Scholar
|
|
9
|
Nagler R, Cohen S and Gavish M: The effect
of cigarette smoke on the translocator protein (TSPO) in cultured
lung cancer cells. J Cell Biochem. 116:2786–2792. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Meng X, Meng C, Yang B, Zhao L, Sun X, Su
Y, Liu H, Fan F, Liu X and Jia L: AP-2α downregulation by cigarette
smoke condensate is counteracted by p53 in human lung cancer cells.
Int J Mol Med. 34:1094–1100. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Filosto S, Becker CR and Goldkorn T:
Cigarette smoke induces aberrant EGF receptor activation that
mediates lung cancer development and resistance to tyrosine kinase
inhibitors. Mol Cancer Ther. 11:795–804. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Faiz A, Heijink IH, Vermeulen CJ, Guryev
V, van den Berge M, Nawijn MC and Pouwels SD: Cigarette smoke
exposure decreases CFLAR expression in the bronchial epithelium,
augmenting susceptibility for lung epithelial cell death and DAMP
release. Sci Rep. 8:124262018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gu Q, Hu C, Chen Q and Xia Y: Tea
polyphenols prevent lung from preneoplastic lesions and effect p53
and bcl-2 gene expression in rat lung tissues. Int J Clin Exp
Pathol. 6:1523–1531. 2013.PubMed/NCBI
|
|
14
|
Sundar IK and Rahman I: Gene expression
profiling of epigenetic chromatin modification enzymes and histone
marks by cigarette smoke: Implications for COPD and lung cancer. Am
J Physiol Lung Cell Mol Physiol. 311:L1245–L1258. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hayakawa S, Ohishi T, Miyoshi N, Oishi Y,
Nakamura Y and Isemura M: Anti-cancer effects of green tea
epigallocatchin-3-gallate and coffee chlorogenic acid. Molecules.
25:45532020. View Article : Google Scholar :
|
|
16
|
Singh BN, Shankar S and Srivastava RK:
Green tea catechin, epigallocatechin-3-gallate (EGCG): Mechanisms,
perspectives and clinical applications. Biochem Pharmacol.
82:1807–1821. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang L, Chen W, Tu G, Chen X, Lu Y, Wu L
and Zheng D: Enhanced chemotherapeutic efficacy of
PLGA-encapsulated epigallocatechin gallate (EGCG) against human
lung cancer. Int J Nanomedicine. 15:4417–4429. 2020.PubMed/NCBI
|
|
18
|
Tang N, Wu Y, Zhou B, Wang B and Yu R:
Green tea, black tea consumption and risk of lung cancer: A
meta-analysis. Lung Cancer. 65:274–283. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fritz H, Seely D, Kennedy DA, Fernandes R,
Cooley K and Fergusson D: Green tea and lung cancer: A systematic
review. Integr Cancer Ther. 12:7–24. 2013. View Article : Google Scholar
|
|
20
|
Lu Y, Yao R, Yan Y, Wang Y, Hara Y, Lubet
RA and You M: A gene expression signature that can predict green
tea exposure and chemopreventive efficacy of lung cancer in mice.
Cancer Res. 66:1956–1963. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gu Q, Hu C, Chen Q, Xia Y, Feng J and Yang
H: Development of a rat model by 3,4-benzopyrene intra-pulmonary
injection and evaluation of the effect of green tea drinking on p53
and bcl-2 expression in lung carcinoma. Cancer Detect Prev.
32:444–451. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou H, Chen JX, Yang CS, Yang MQ, Deng Y
and Wang H: Gene regulation mediated by microRNAs in response to
green tea polyphenol EGCG in mouse lung cancer. BMC Genomics.
15(Suppl 11): S32014. View Article : Google Scholar
|
|
23
|
Huang J, Chen S, Shi Y, Li CH, Wang XJ, Li
FJ, Wang CH, Meng QH, Zhong JN, Liu M and Wang ZM: Epigallocatechin
gallate from green tea exhibits potent anticancer effects in A-549
non-small lung cancer cells by inducing apoptosis, cell cycle
arrest and inhibition of cell migration. J BUON. 22:1422–1427.
2017.
|
|
24
|
Oya Y, Mondal A, Rawangkan A, Umsumarng S,
Iida K, Watanabe T, Kanno M, Suzuki K, Li Z, Kagechika H, et al:
Down-regulation of histone deacetylase 4, -5 and -6 as a mechanism
of synergistic enhancement of apoptosis in human lung cancer cells
treated with the combination of a synthetic retinoid, Am80 and
green tea catechin. J Nutr Biochem. 42:7–16. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Milligan SA, Burke P, Coleman DT, Bigelow
RL, Steffan JJ, Carroll JL, Williams BJ and Cardelli JA: The green
tea polyphenol EGCG potentiates the antiproliferative activity of
c-Met and epidermal growth factor receptor inhibitors in non-small
cell lung cancer cells. Clin Cancer Res. 15:4885–4894. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li M, Li JJ, Gu QH, An J, Cao LM, Yang HP
and Hu CP: EGCG induces lung cancer A549 cell apoptosis by
regulating Ku70 acetylation. Oncol Rep. 35:2339–2347. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang L, Xie J, Gan R, Wu Z, Luo H, Chen
X, Lu Y, Wu L and Zheng D: Synergistic inhibition of lung cancer
cells by EGCG and NF-κB inhibitor BAY11-7082. J Cancer.
10:6543–6556. 2019. View Article : Google Scholar :
|
|
28
|
Jiang P, Xu C, Zhang P, Ren J, Mageed F,
Wu X, Chen L, Zeb F, Feng Q and Li S: Epigallocatechin-3-gallate
inhibits self-renewal ability of lung cancer stem-like cells
through inhibition of CLOCK. Int J Mol Med. 46:2216–2224. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Deng YT and Lin JK: EGCG inhibits the
invasion of highly invasive CL1-5 lung cancer cells through
suppressing MMP-2 expression via JNK signaling and induces G2/M
arrest. J Agric Food Chem. 59:13318–13327. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gu JJ, Qiao KS, Sun P, Chen P and Li Q:
Study of EGCG induced apoptosis in lung cancer cells by inhibiting
PI3K/Akt signaling pathway. Eur Rev Med Pharmacol Sci.
22:4557–4563. 2018.PubMed/NCBI
|
|
31
|
Yu C, Jiao Y, Xue J, Zhang Q, Yang H, Xing
L, Chen G, Wu J, Zhang S, Zhu W and Cao J: Metformin sensitizes
non-small cell lung cancer cells to an epigallocatechin-3-gallate
(EGCG) treatment by suppressing the Nrf2/HO-1 signaling pathway.
Int J Biol Sci. 13:1560–1569. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhu J, Jiang Y, Yang X, Wang S, Xie C, Li
X, Li Y, Chen Y, Wang X, Meng Y, et al: Wnt/β-catenin pathway
mediates (-)-Epigallocatechin-3-gallate (EGCG) inhibition of lung
cancer stem cells. Biochem Biophys Res Commun. 482:15–21. 2017.
View Article : Google Scholar
|
|
33
|
Wei R, Wirkus J, Yang Z, Machuca J,
Esparza Y and Mackenzie GG: EGCG sensitizes
chemotherapeutic-induced cytotoxicity by targeting the ERK pathway
in multiple cancer cell lines. Arch Biochem Biophys.
692:1085462020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen A, Jiang P, Zeb F, Wu X, Xu C, Chen L
and Feng Q: EGCG regulates CTR1 expression through its
pro-oxidative property in non-small-cell lung cancer cells. J Cell
Physiol. 235:7970–7981. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Doukas SG, Vageli DP, Lazopoulos G,
Spandidos DA, Sasaki CT and Tsatsakis A: The effect of NNK, a
tobacco smoke carcinogen, on the miRNA and mismatch dna repair
expression profiles in lung and head and neck squamous cancer
cells. Cells. 9:10312020. View Article : Google Scholar :
|
|
36
|
Hu DL, Wang G, Yu J, Zhang LH, Huang YF,
Wang D and Zhou HH: Epigallocatechin-3-gallate modulates long
non-coding RNA and mRNA expression profiles in lung cancer cells.
Mol Med Rep. 19:1509–1520. 2019.PubMed/NCBI
|
|
37
|
Bhardwaj V and Mandal AKA: Next-generation
sequencing reveals the role of epigallocatechin-3-gallate in
regulating putative novel and known microRNAs which target the MAPK
pathway in non-small-cell lung cancer a549 cells. Molecules.
24:3682019. View Article : Google Scholar :
|
|
38
|
Chen Y, Pan Y, Ji Y, Sheng L and Du X:
Network analysis of differentially expressed smoking-associated
mRNAs, lncRNAs and miRNAs reveals key regulators in
smoking-associated lung cancer. Exp Ther Med. 16:4991–5002.
2018.PubMed/NCBI
|
|
39
|
Torkashvand J, Farzadkia M, Sobhi HR and
Esrafili A: Littered cigarette butt as a well-known hazardous
waste: A comprehensive systematic review. J Hazard Mater.
383:1212422020. View Article : Google Scholar
|
|
40
|
Hecht SS: Approaches to chemoprevention of
lung cancer based on carcinogens in tobacco smoke. Environ Health
Perspect. 105(Suppl 4): S955–S963. 1997.
|
|
41
|
Hoffmann D, Rivenson A, Chung FL and Hecht
SS: Nicotine-derived N-nitrosamines (TSNA) and their relevance in
tobacco carcinogenesis. Crit Rev Toxicol. 21:305–311. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Li GX, Chen YK, Hou Z, Xiao H, Jin H, Lu
G, Lee MJ, Liu B, Guan F, Yang Z, et al: Pro-oxidative activities
and dose-response relationship of (-)-epigallocatechin-3-gallate in
the inhibition of lung cancer cell growth: A comparative study in
vivo and in vitro. Carcinogenesis. 31:902–910. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Thomassen DG, Chen BT, Mauderly JL,
Johnson NF and Griffith WC: Inhaled cigarette smoke induces
preneoplastic changes in rat tracheal epithelial cells.
Carcinogenesis. 10:2359–2361. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bjermer L, Cai Y, Nilsson K, Hellström S
and Henriksson R: Tobacco smoke exposure suppresses
radiation-induced inflammation in the lung: A study of
bronchoalveolar lavage and ultrastructural morphology in the rat.
Eur Respir J. 6:1173–1180. 1993.PubMed/NCBI
|
|
45
|
Xue Y, Harris E, Wang W and Baybutt RC:
Vitamin A depletion induced by cigarette smoke is associated with
an increase in lung cancer-related markers in rats. J Biomed Sci.
22:842015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
47
|
Yuan C, Xiang L, Bai R, Cao K, Gao Y,
Jiang X, Zhang N, Gong Y and Xie C: MiR-195 restrains lung
adenocarcinoma by regulating CD4+ T cell activation via
the CCDC88C/Wnt signaling pathway: A study based on the cancer
genome atlas (TCGA), gene expression omnibus (GEO) and
bioinformatic analysis. Ann Transl Med. 7:2632019. View Article : Google Scholar
|
|
48
|
Chen Y and Wang X: miRDB: An online
database for prediction of functional microRNA targets. Nucleic
Acids Res. 48D:D127–D131. 2020. View Article : Google Scholar
|
|
49
|
Vlachos IS, Zagganas K, Paraskevopoulou
MD, Georgakilas G, Karagkouni D, Vergoulis T, Dalamagas T and
Hatzigeorgiou AG: DIANA-miRPath v3.0: Deciphering microRNA function
with experimental support. Nucleic Acids Res. 43W:W460–W466. 2015.
View Article : Google Scholar
|
|
50
|
Freis A, Keller A, Ludwig N, Meese E,
Jauckus J, Rehnitz J, Capp E, Strowitzki T and Germeyer A: Altered
miRNA-profile dependent on ART outcome in early pregnancy targets
Wnt-pathway. Reproduction. 154:799–805. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang C, Wu R, Sargsyan D, Zheng M, Li S,
Yin R, Su S, Raskin I and Kong AN: CpG methyl-seq and RNA-seq
epigenomic and transcriptomic studies on the preventive effects of
Moringa isothiocyanate in mouse epidermal JB6 cells induced by the
tumor promoter TPA. J Nutr Biochem. 68:69–78. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhao X, Liang M, Li X, Qiu X and Cui L:
Identification of key genes and pathways associated with osteogenic
differentiation of adipose stem cells. J Cell Physiol.
233:9777–9785. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Heinrich U, Muhle H, Takenaka S, Ernst H,
Fuhst R, Mohr U, Pott F and Stöber W: Chronic effects on the
respiratory tract of hamsters, mice and rats after long-term
inhalation of high concentrations of filtered and unfiltered diesel
engine emissions. J Appl Toxicol. 6:383–395. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Melnick RL, Huff JE, Roycroft JH, Chou BJ
and Miller RA: Inhalation toxicology and carcinogenicity of
1,3-butadiene in B6C3F1 mice following 65 weeks of exposure.
Environ Health Perspect. 86:27–36. 1990.PubMed/NCBI
|
|
55
|
Ceppi M, Munnia A, Cellai F, Bruzzone M
and Peluso MEM: Linking the generation of DNA adducts to lung
cancer. Toxicology. 390:160–166. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Munnia A, Giese RW, Polvani S, Galli A,
Cellai F and Peluso MEM: Bulky DNA adducts, tobacco smoking,
genetic susceptibility, and lung cancer risk. Adv Clin Chem.
81:231–277. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Avino P, Scungio M, Stabile L, Cortellessa
G, Buonanno G and Manigrasso M: Second-hand aerosol from tobacco
and electronic cigarettes: Evaluation of the smoker emission rates
and doses and lung cancer risk of passive smokers and vapers. Sci
Total Environ. 642:137–147. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Duan S, Wang N, Huang L, Shao H, Zhang P,
Wang W, Wu Y, Wang J, Liu H, Zhang Q and Feng F: NLRP3 inflammasome
activation involved in LPS and coal tar pitch extract-induced
malignant transformation of human bronchial epithelial cells.
Environ Toxicol. 34:585–593. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Vaz M, Hwang SY, Kagiampakis I, Phallen J,
Patil A, O'Hagan HM, Murphy L, Zahnow CA, Gabrielson E, Velculescu
VE, et al: Chronic cigarette smoke-induced epigenomic changes
precede sensitization of bronchial epithelial cells to single-step
transformation by KRAS mutations. Cancer Cell. 32:360–376.e6. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Chen C, Jiang X, Gu S and Zhang Z:
MicroRNA-155 regulates arsenite-induced malignant transformation by
targeting Nrf2-mediated oxidative damage in human bronchial
epithelial cells. Toxicol Lett. 278:38–47. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Fujiki H, Watanabe T, Sueoka E, Rawangkan
A and Suganuma M: Cancer prevention with green tea and its
principal constituent, EGCG: From early investigations to current
focus on human cancer stem cells. Mol Cells. 41:73–82.
2018.PubMed/NCBI
|
|
62
|
Moorthy B, Chu C and Carlin DJ: Polycyclic
aromatic hydrocarbons: From metabolism to lung cancer. Toxicol Sci.
145:5–15. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Lawther PJ and Waller RE: Coal fires,
industrial emissions and motor vehicles as sources of environmental
carcinogens. IARC Sci Publ. 6:27–40. 1976.
|
|
64
|
Perera F: Carcinogenicity of airborne fine
particulate benzo(a) pyrene: An appraisal of the evidence and the
need for control. Environ Health Perspect. 42:163–185. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Knize MG, Salmon CP, Pais P and Felton JS:
Food heating and the formation of heterocyclic aromatic amine and
polycyclic aromatic hydrocarbon mutagens/carcinogens. Adv Exp Med
Biol. 459:179–193. 1999. View Article : Google Scholar : PubMed/NCBI
|