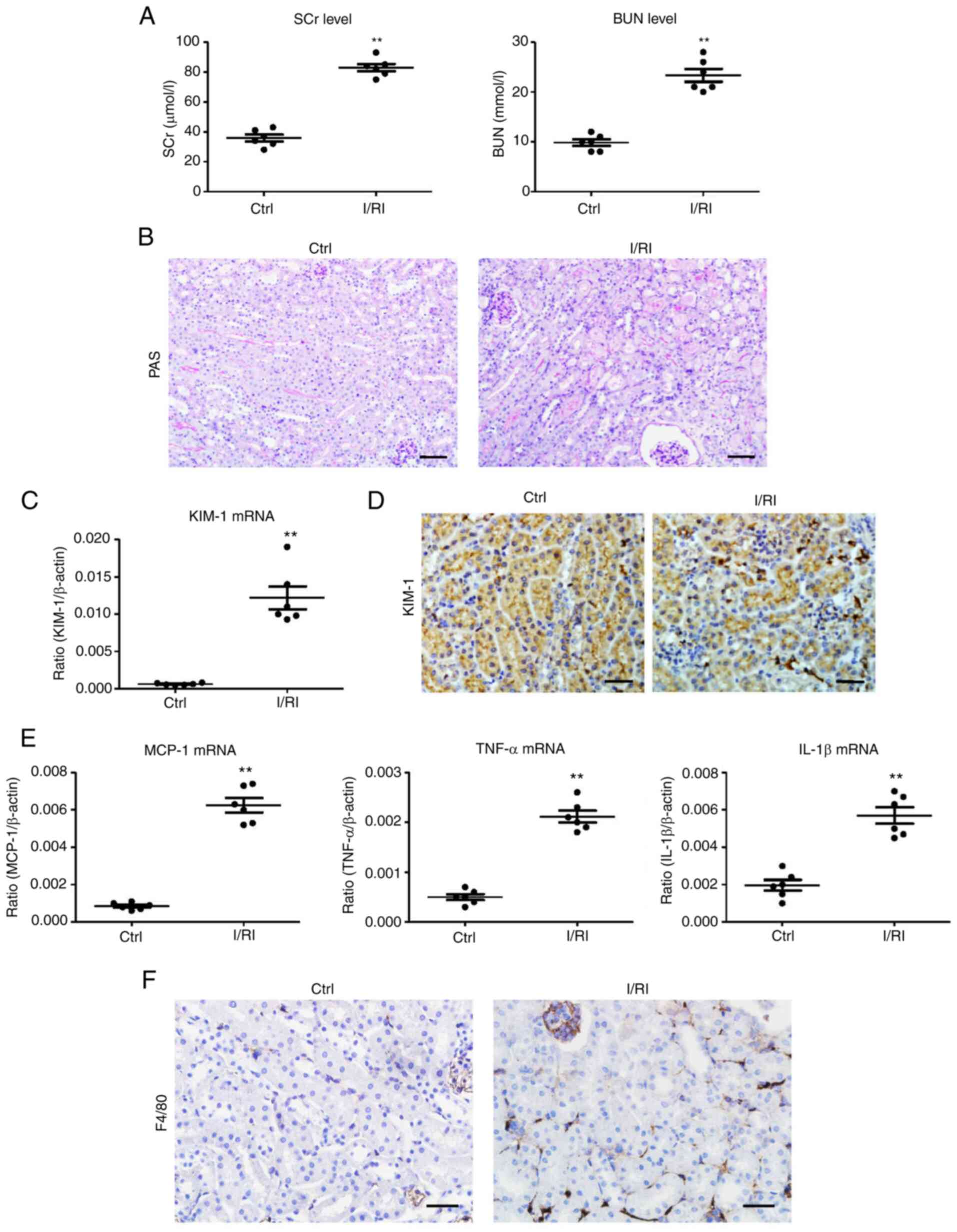
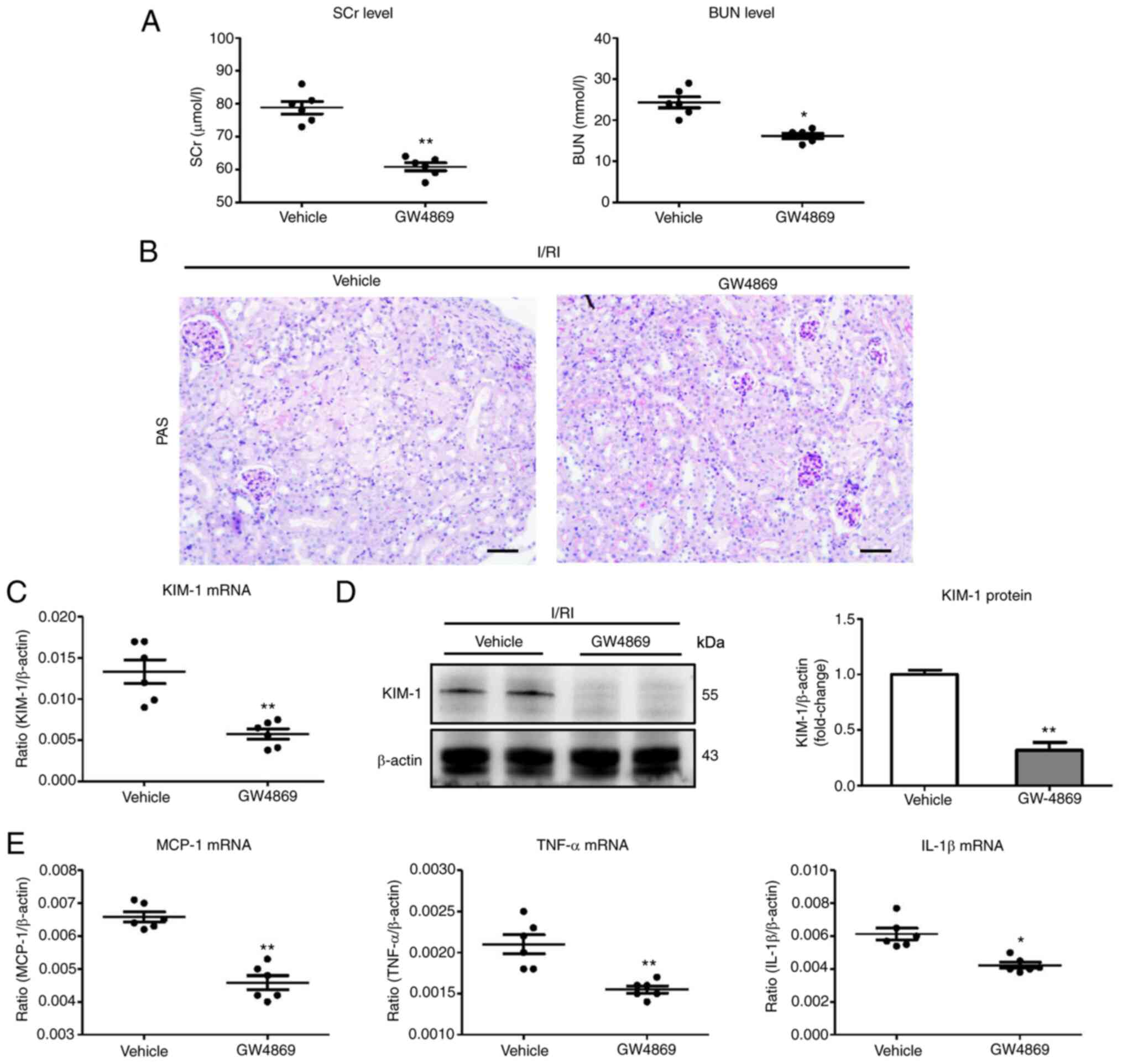
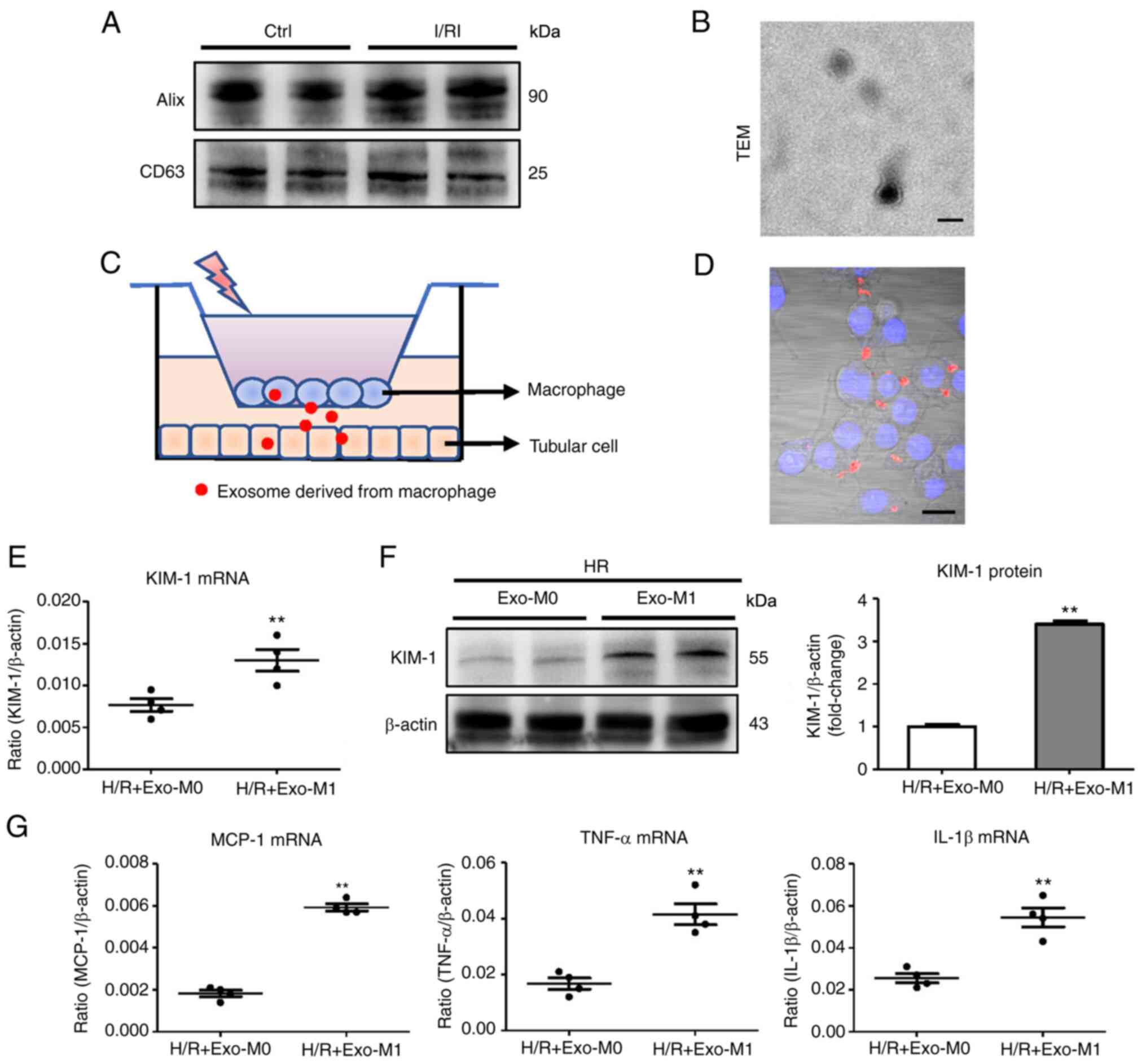
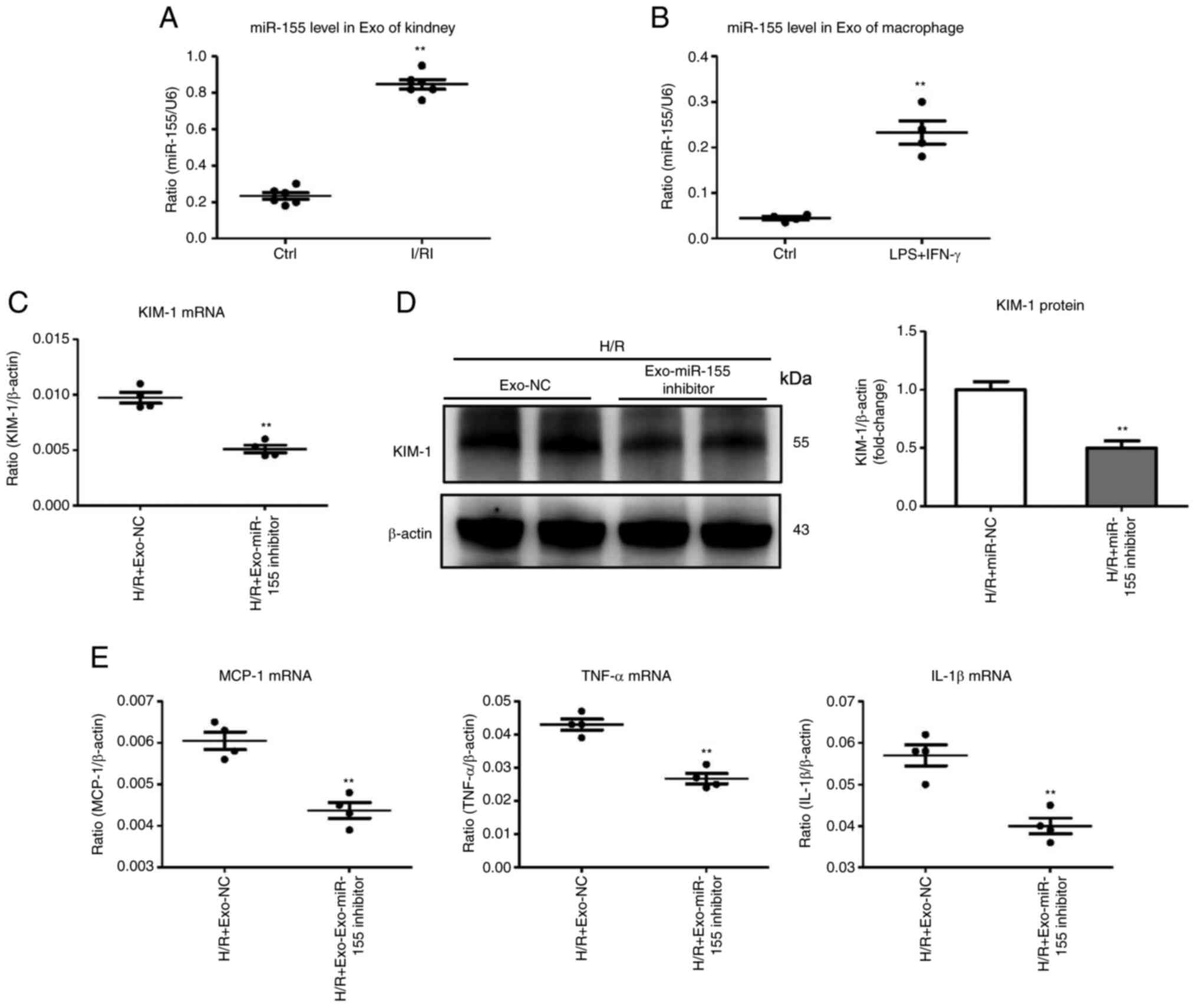
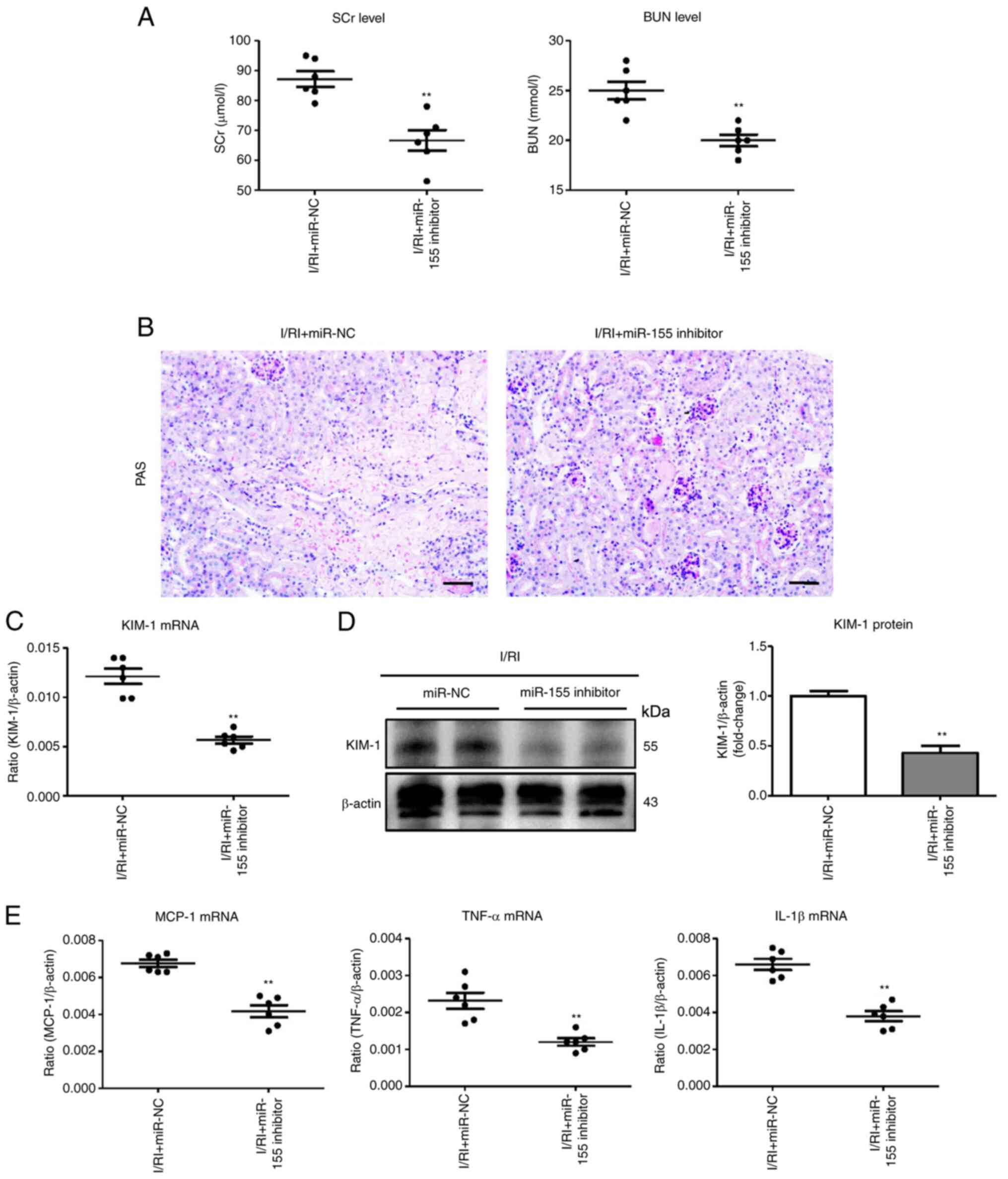
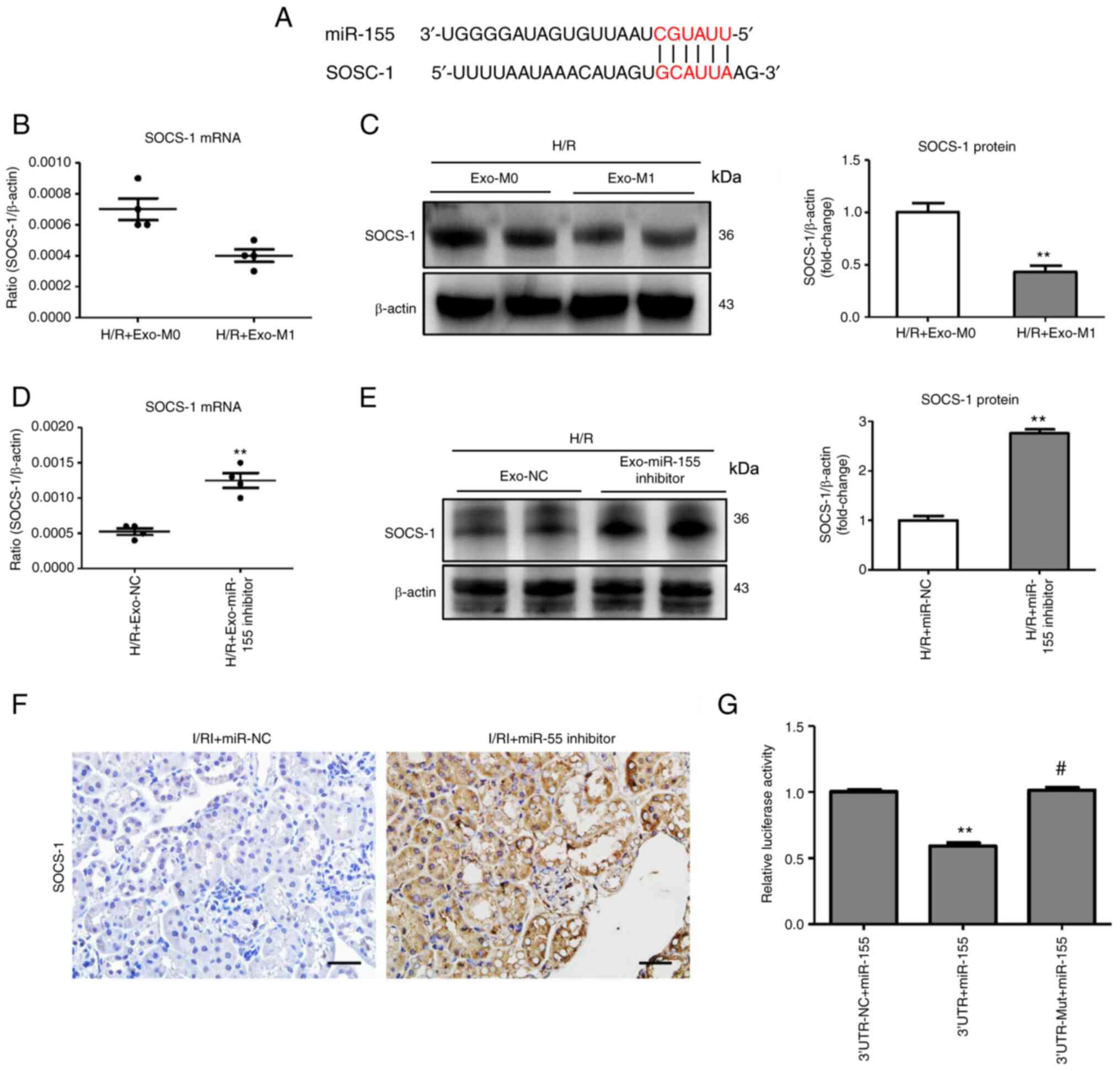
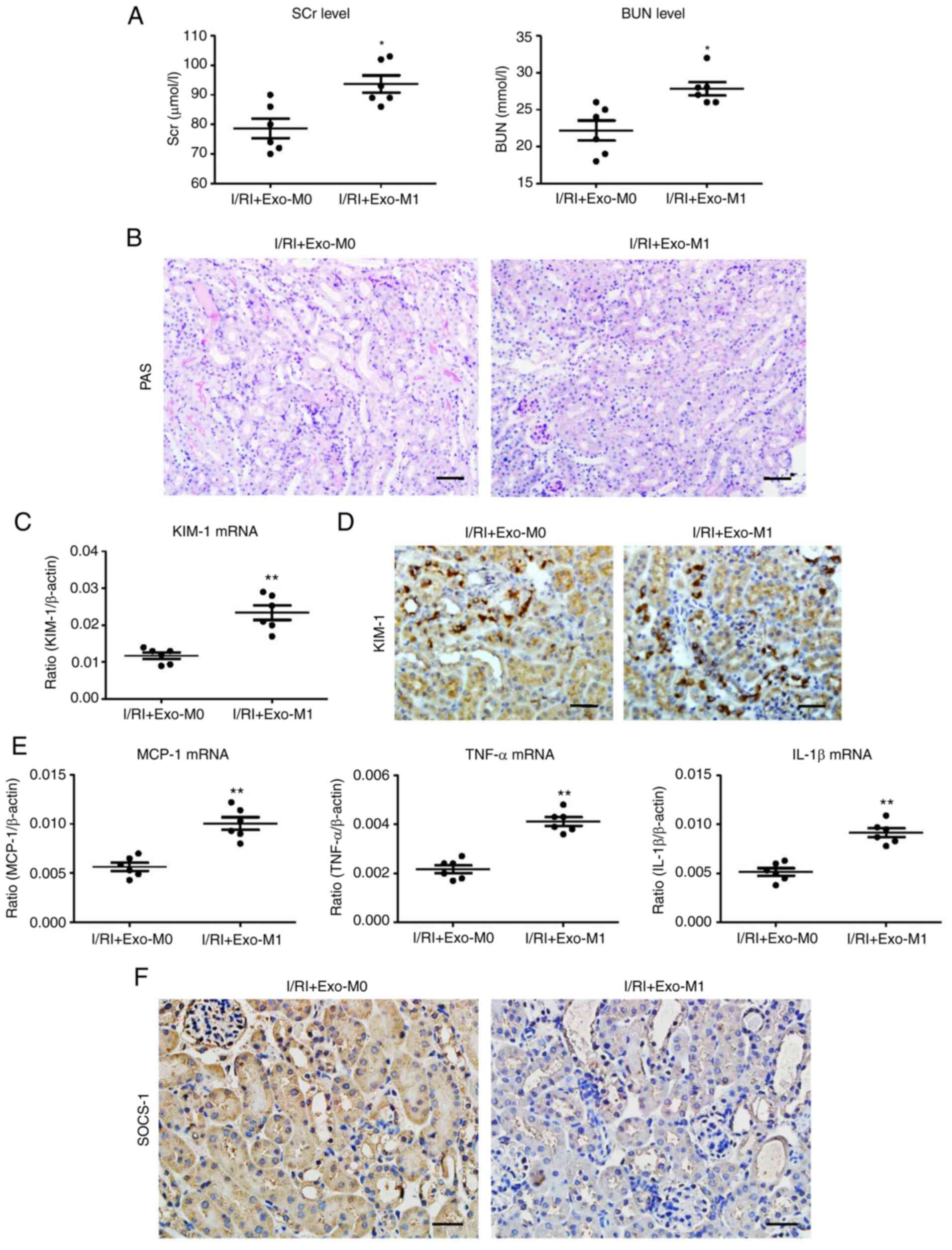
|
1
|
Ronco C, Bellomo R and Kellum JA: Acute
kidney injury. Lancet. 394:1949–1964. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Takaori K, Nakamura J, Yamamoto S, Nakata
H, Sato Y, Takase M, Nameta M, Yamamoto T, Economides AN, Kohno K,
et al: Severity and frequency of proximal tubule injury determines
renal prognosis. J Am Soc Nephrol. 27:2393–2406. 2016. View Article : Google Scholar :
|
|
3
|
Cao JY, Wang B, Tang TT, Wen Y, Li ZL,
Feng ST, Wu M, Liu D, Yin D, Ma KL, et al: Exosomal miR-125b-5p
deriving from mesenchymal stem cells promotes tubular repair by
suppression of p53 in ischemic acute kidney injury. Theranostics.
11:5248–5266. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tang PM, Nikolic-Paterson DJ and Lan HY:
Macrophages: Versatile players in renal inflammation and fibrosis.
Nat Rev Nephrol. 15:144–158. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Huen SC and Cantley LG: Macrophages in
renal injury and repair. Annu Rev Physiol. 79:449–469. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mathieu M, Martin-Jaular L, Lavieu G and
Théry C: Specificities of secretion and uptake of exosomes and
other extracellular vesicles for cell-to-cell communication. Nat
Cell Biol. 21:9–17. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ding H, Li LX, Harris PC, Yang J and Li X:
Extracellular vesicles and exosomes generated from cystic renal
epithelial cells promote cyst growth in autosomal dominant
polycystic kidney disease. Nat Commun. 12:45482021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mori MA, Ludwig RG, Garcia-Martin R,
Brandão BB and Kahn CR: Extracellular miRNAs: From biomarkers to
mediators of physiology and disease. Cell Metab. 30:656–673. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
van Niel G, D'Angelo G and Raposo G:
Shedding light on the cell biology of extracellular vesicles. Nat
Rev Mol Cell Biol. 19:213–228. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu X, Miao J, Wang C, Zhou S, Chen S, Ren
Q, Hong X, Wang Y, Hou FF, Zhou L and Liu Y: Tubule-derived
exosomes play a central role in fibroblast activation and kidney
fibrosis. Kidney Int. 97:1181–1195. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lv LL, Feng Y, Wen Y, Wu WJ, Ni HF, Li ZL,
Zhou LT, Wang B, Zhang JD, Crowley SD and Liu BC: Exosomal CCL2
from tubular epithelial cells is critical for albumin-induced
tubulointerstitial inflammation. J Am Soc Nephrol. 29:919–935.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
de Couto G, Gallet R, Cambier L,
Jaghatspanyan E, Makkar N, Dawkins JF, Berman BP and Marbán E:
Exosomal MicroRNA transfer into macrophages mediates cellular
postconditioning. Circulation. 136:200–214. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li ZL, Lv LL, Tang TT, Wang B, Feng Y,
Zhou LT, Cao JY, Tang RN, Wu M, Liu H, et al: HIF-1α inducing
exosomal microRNA-23a expression mediates the cross-talk between
tubular epithelial cells and macrophages in tubulointerstitial
inflammation. Kidney Int. 95:388–404. 2019. View Article : Google Scholar
|
|
14
|
Yu W, Zeng H, Chen J, Fu S, Huang Q, Xu Y,
Xu A, Lan HY and Tang Y: miR-20a-5p is enriched in hypoxia-derived
tubular exosomes and protects against acute tubular injury. Clin
Sci (Lond). 134:2223–2234. 2020. View Article : Google Scholar
|
|
15
|
Wang C, Zhang C, Liu L, A X, Chen B, Li Y
and Du J: Macrophage-derived mir-155-containing exosomes suppress
fibroblast proliferation and promote fibroblast inflammation during
cardiac injury. Mol Ther. 25:192–204. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ge X, Tang P, Rong Y, Jiang D, Lu X, Ji C,
Wang J, Huang C, Duan A, Liu Y, et al: Exosomal miR-155 from
M1-polarized macrophages promotes EndoMT and impairs mitochondrial
function via activating NF-κB signaling pathway in vascular
endothelial cells after traumatic spinal cord injury. Redox Biol.
41:1019322021. View Article : Google Scholar
|
|
17
|
Wei Q, Liu Y, Liu P, Hao J, Liang M, Mi
QS, Chen JK and Dong Z: MicroRNA-489 induction by hypoxia-inducible
factor-1 protects against ischemic kidney injury. J Am Soc Nephrol.
27:2784–2796. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gusella GL, Fedorova E, Hanss B, Marras D,
Klotman ME and Klotman PE: Lentiviral gene transduction of kidney.
Hum Gene Ther. 13:407–414. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Weidemann A, Bernhardt WM, Klanke B,
Daniel C, Buchholz B, Câmpean V, Amann K, Warnecke C, Wiesener MS,
Eckardt KU and Willam C: HIF activation protects from acute kidney
injury. J Am Soc Nephrol. 19:486–494. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li ZL, Wang B, Lv LL, Tang TT, Wen Y, Cao
JY, Zhu XX, Feng ST, Crowley SD and Liu BC: FIH-1-modulated HIF-1α
C-TAD promotes acute kidney injury to chronic kidney disease
progression via regulating KLF5 signaling. Acta Pharmacol Sin.
42:2106–2119. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lv LL, Feng Y, Wu M, Wang B, Li ZL, Zhong
X, Wu WJ, Chen J, Ni HF, Tang TT, et al: Exosomal miRNA-19b-3p of
tubular epithelial cells promotes M1 macrophage activation in
kidney injury. Cell Death Differ. 27:210–226. 2020. View Article : Google Scholar :
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
23
|
Catalano M and O'Driscoll L: Inhibiting
extracellular vesicles formation and release: A review of EV
inhibitors. J Extracell Vesicles. 9:17032442019. View Article : Google Scholar
|
|
24
|
Chevalier RL: The proximal tubule is the
primary target of injury and progression of kidney disease: Role of
the glomerulotubular junction. Am J Physiol Renal Physiol.
311:F145–F161. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu BC, Tang TT, Lv LL and Lan HY: Renal
tubule injury: A driving force toward chronic kidney disease.
Kidney Int. 93:568–579. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Baek JH, Zeng R, Weinmann-Menke J,
Valerius MT, Wada Y, Ajay AK, Colonna M and Kelley VR: IL-34
mediates acute kidney injury and worsens subsequent chronic kidney
disease. J Clin Invest. 125:3198–3214. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wen Y and Crowley SD: The varying roles of
macrophages in kidney injury and repair. Curr Opin Nephrol
Hypertens. 29:286–292. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Meng XM, Tang PM, Li J and Lan HY:
Macrophage phenotype in kidney injury and repair. Kidney Dis
(Basel). 1:138–146. 2015. View Article : Google Scholar
|
|
29
|
Wang Y, Zhao M, Liu S, Guo J, Lu Y, Cheng
J and Liu J: Macrophage-derived extracellular vesicles: Diverse
mediators of pathology and therapeutics in multiple diseases. Cell
Death Dis. 11:9242020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wu R, Gao W, Yao K and Ge J: Roles of
exosomes derived from immune cells in cardiovascular diseases.
Front Immunol. 10:6482019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu J, Wu F and Zhou H: Macrophage-derived
exosomes in cancers: Biogenesis, functions and therapeutic
applications. Immunol Lett. 227:102–108. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Huang H, Liu H, Tang J, Xu W, Gan H, Fan Q
and Zhang W: M2 macrophage-derived exosomal miR-25-3p improves high
glucose-induced podocytes injury through activation autophagy via
inhibiting DUSP1 expression. IUBMB Life. 72:2651–2662. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ying W, Riopel M, Bandyopadhyay G, Dong Y,
Birmingham A, Seo JB, Ofrecio JM, Wollam J, Hernandez-Carretero A,
Fu W, et al: Adipose tissue macrophage-derived exosomal miRNAs can
modulate in vivo and in vitro insulin sensitivity. Cell.
171:372–384.e12. 2017. View Article : Google Scholar
|
|
34
|
Wang B, Wang ZM, Ji JL, Gan W, Zhang A,
Shi HJ, Wang H, Lv L, Li Z, Tang T, et al: Macrophage-derived
exosomal Mir-155 regulating cardiomyocyte pyroptosis and
hypertrophy in uremic cardiomyopathy. JACC Basic Transl Sci.
5:148–166. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yoshimura A, Naka T and Kubo M: SOCS
proteins, cytokine signalling and immune regulation. Nat Rev
Immunol. 7:454–465. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Park SH, Kim KE, Hwang HY and Kim TY:
Regulatory effect of SOCS on NF-kappaB activity in murine
monocytes/macrophages. DNA Cell Biol. 22:131–139. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Markó L, Vigolo E, Hinze C, Park JK, Roël
G, Balogh A, Choi M, Wübken A, Cording J, Blasig IE, et al: Tubular
epithelial NF-κB activity regulates ischemic AKI. J Am Soc Nephrol.
27:2658–2669. 2016. View Article : Google Scholar
|
|
38
|
Heymans S, Corsten MF, Verhesen W, Carai
P, van Leeuwen RE, Custers K, Peters T, Hazebroek M, Stöger L,
Wijnands E, et al: Macrophage microRNA-155 promotes cardiac
hypertrophy and failure. Circulation. 128:1420–1432. 2013.
View Article : Google Scholar : PubMed/NCBI
|