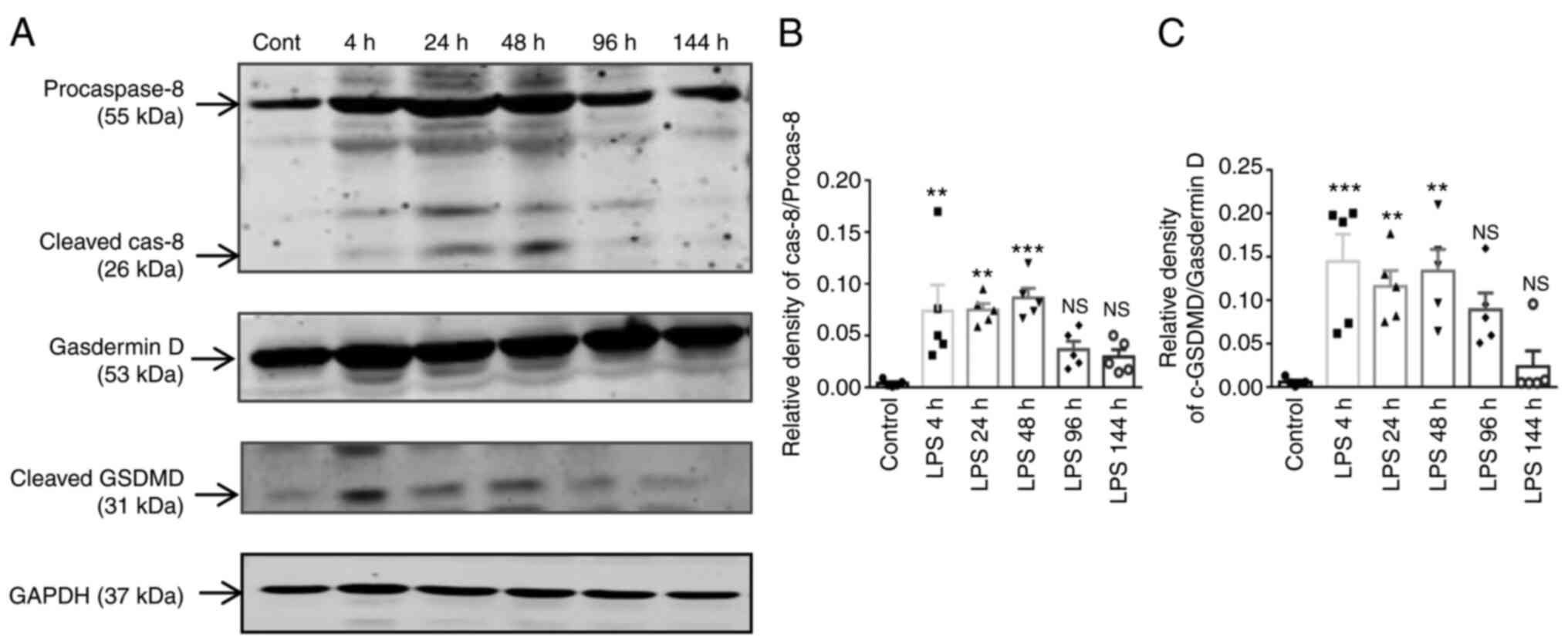
|
1
|
Ranieri VM, Rubenfeld GD, Thompson BT,
Ferguson ND, Caldwell E, Fan E, Camporota L and Slutsky AS: Acute
respiratory distress syndrome: The Berlin definition. JAMA.
307:2526–2533. 2012.PubMed/NCBI
|
|
2
|
Kushimoto S, Endo T, Yamanouchi S,
Sakamoto T, Ishikura H, Kitazawa Y, Taira Y, Okuchi K, Tagami T,
Watanabe A, et al: Relationship between extravascular lung water
and severity categories of acute respiratory distress syndrome by
the Berlin definition. Crit Care. 17:R1322013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Abdulnour REE and Levy BD: Acute lung
injury and the acute respiratory distress syndrome. Med Manag Surg
Patient A Textb Perioper Med Fifth Ed. 154–171. 2010.
|
|
4
|
Johnston LK, Rims CR, Gill SE, McGuire JK
and Manicone AM: Pulmonary macrophage subpopulations in the
induction and resolution of acute lung injury. Am J Respir Cell Mol
Biol. 47:417–426. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang A, Pan W, Lv J and Wu H: Protective
effect of amygdalin on LPS-induced acute lung injury by inhibiting
NF-κB and NLRP3 signaling pathways. Inflammation. 40:745–751. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tianzhu Z and Shumin W: Esculin inhibits
the inflammation of LPS-induced acute lung injury in mice via
regulation of TLR/NF-κB pathways. Inflammation. 38:1529–1536. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Park EJ, Kim YM, Kim HJ and Chang KC:
Luteolin activates ERK1/2- and Ca2+-dependent HO-1 induction that
reduces LPS-induced HMGB1, iNOS/NO, and COX-2 expression in
RAW264.7 cells and mitigates acute lung injury of endotoxin mice.
Inflamm Res. 67:445–453. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Marsland BJ, Nembrini C, Grün K, Reissmann
R, Kurrer M, Leipner C and Kopf M: TLR ligands act directly upon T
cells to restore proliferation in the absence of protein kinase
C-theta signaling and promote autoimmune myocarditis. J Immunol.
178:3466–3473. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mandraju R, Murray S, Forman J and Pasare
C: Differential ability of surface and endosomal TLRs to induce CD8
T cell responses in vivo. J Immunol. 192:4303–4315. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Imam F, Al-Harbi NO, Al-Harbi MM, Ansari
MA, Zoheir KMA, Iqbal M, Anwer MK, Hoshani ARA, Attia SM and Ahmad
SF: Diosmin downregulates the expression of T cell receptors,
pro-inflammatory cytokines and NF-κB activation against LPS-induced
acute lung injury in mice. Pharmacol Res. 102:1–11. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Boorsma CE, Draijer C and Melgert BN:
Macrophage heterogeneity in respiratory diseases. Mediators
Inflamm. 2013:7692142013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Byrne AJ, Mathie SA, Gregory LG and Lloyd
CM: Pulmonary macrophages: Key players in the innate defence of the
airways. Thorax. 70:1189–1196. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lee JW, Chun W, Lee HJ, Min JH, Kim SM,
Seo JY, Ahn KS and Oh SR: The role of macrophages in the
development of acute and chronic inflammatory lung diseases. Cells.
10:8972021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nakajima T, Suarez CJ, Lin KW, Jen KY,
Schnitzer JE, Makani SS, Parker N, Perkins DL and Finn PW: T cell
pathways involving CTLA4 contribute to a model of acute lung
injury. J Immunol. 184:5835–5841. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Eberl G: RORγt, a multitask nuclear
receptor at mucosal surfaces. Mucosal Immunol. 10:27–34. 2017.
View Article : Google Scholar
|
|
16
|
Noack M and Miossec P: Th17 and regulatory
T cell balance in autoimmune and inflammatory diseases. Autoimmun
Rev. 13:668–677. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Connors TJ, Ravindranath TM, Bickham KL,
Bickham KL, Gordon CL, Zhang F, Levin B, Baird JS and Farber DL:
Airway CD8+ T cells are associated with lung injury during infant
viral respiratory tract infection. Am J Respir Cell Mol Biol.
54:822–830. 2016. View Article : Google Scholar :
|
|
18
|
Wong JJM, Leong JY, Lee JH, Albani S and
Yeo JG: Insights into the immuno-pathogenesis of acute respiratory
distress syndrome. Ann Transl Med. 7:5042019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kumar V: Toll-like receptors in
sepsis-associated cytokine storm and their endogenous negative
regulators as future immunomodulatory targets. Int Immunopharmacol.
89:1070872020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Soy M, Keser G, Atagündüz P, Tabak F,
Atagündüz I and Kayhan S: Cytokine storm in COVID-19: Pathogenesis
and overview of anti-inflammatory agents used in treatment. Clin
Rheumatol. 39:2085–2094. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hennig P, Garstkiewicz M, Grossi S,
Filippo MD, French LE and Beer HD: The crosstalk between Nrf2 and
Inflammasomes. Int J Mol Sci. 19:5622018. View Article : Google Scholar :
|
|
22
|
Garstkiewicz M, Strittmatter GE, Grossi S,
Sand J, Fenini G, Werner S, French LE and Beer HD: Opposing effects
of Nrf2 and Nrf2-activating compounds on the NLRP3 inflammasome
independent of Nrf2-mediated gene expression. Eur J Immunol.
47:806–817. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Maier NK, Leppla SH and Moayeri M: The
cyclopentenone prostaglandin 15d-PGJ 2 inhibits the NLRP1 and NLRP3
inflammasomes. J Immunol. 194:2776–2785. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tonelli C, Chio IIC and Tuveson DA:
Transcriptional regulation by Nrf2. Antioxid Redox Signal.
29:1727–1745. 2018. View Article : Google Scholar :
|
|
25
|
Kelley N, Jeltema D, Duan Y and He Y: The
NLRP3 inflammasome: An overview of mechanisms of activation and
regulation. Int J Mol Sci. 20:33282019. View Article : Google Scholar :
|
|
26
|
Possomato-Vieira, José S and Khalil RAK:
Mechanism and regulation of NLRP3 inflammasome activation. Physiol
Behav. 176:139–148. 2016.
|
|
27
|
Zhao C, Gillette DD, Li X, Zhang Z and Wen
H: Nuclear factor E2-related factor-2 (Nrf2) is required for NLRP3
and AIM2 inflammasome activation. J Biol Chem. 289:17020–17029.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sarhan J, Liu BC, Muendlein HI, Li P,
Nilson R, Tang AY, Rongvaux A, Bunnell SC, Shao F, Green DR and
Poltorak A: Caspase-8 induces cleavage of gasdermin D to elicit
pyroptosis during Yersinia infection. Proc Natl Acad Sci USA.
115:E10888–E10897. 2018. View Article : Google Scholar :
|
|
29
|
Muendlein HI, Jetton D, Connolly WM,
Eidell KP, Magri Z, Smirnova I and Poltorak A: CFLIPL protects
macrophages from LPS-induced pyroptosis via inhibition of complex
II formation. Science. 367:1379–1384. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xia S, Hollingsworth LR and Wu H:
Mechanism and regulation of gasdermin-mediated cell death. Cold
Spring Harb Perspect Biol. 12:1–14. 2020. View Article : Google Scholar
|
|
31
|
Cen M, Ouyang W, Zhang W, Yang L, Lin X,
Dai M, Hu H, Tang H, Liu H, Xia J and Xu F: MitoQ protects against
hyper-permeability of endothelium barrier in acute lung injury via
a Nrf2-dependent mechanism. Redox Biol. 41:1019362021. View Article : Google Scholar
|
|
32
|
Tseng TL, Chen MF, Tsai MJ, Hsu YH, Chen
CP and Lee TJF: Oroxylin-a rescues LPS-induced acute lung injury
via regulation of NF-κB signaling pathway in rodents. PLoS One.
7:e474032012. View Article : Google Scholar
|
|
33
|
Matute-Bello G, Downey G, Moore BB,
Groshong SD, Matthay MA, Slutsky AS and Kuebler WM: An official
american thoracic society workshop report: Features and
measurements of experimental acute lung injury in animals. Am J
Respir Cell Mol Biol. 44:725–738. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
35
|
Socci DJ, Bjugstad KB, Jones HC, Pattisapu
JV and Arendash GW: Evidence that oxidative stress is associated
with the pathophysiology of inherited hydrocephalus in the H-Tx rat
model. Exp Neurol. 155:109–117. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Roberts CA, Dickinson AK and Taams LS: The
interplay between monocytes/macrophages and CD4+ T cell subsets in
rheumatoid arthritis. Front Immunol. 6:5712015. View Article : Google Scholar :
|
|
37
|
Dagvadorj J, Shimada K, Chen S, Jones HD,
Tumurkhuu G, Zhang W, Wawrowsky KA, Crother TR and Arditi M:
Lipopolysaccharide induces alveolar macrophage necrosis via CD14
and the P2x7 receptor leading to Interleukin-1α release. Immunity.
42:640–653. 2016. View Article : Google Scholar
|
|
38
|
Altemeier WA, Zhu X, Berrington WR, Harlan
JM and Liles WC: Fas (CD95) induces macrophage proinflammatory
chemokine production via a MyD88-dependent, caspase-independent
pathway. J Leukoc Biol. 82:721–728. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hatanaka E, Monteagudo PT, Marrocos MSM
and Campa A: Neutrophils and monocytes as potentially important
sources of proinflammatory cytokines in diabetes. Clin Exp Immunol.
146:443–447. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
An SJ, Pae HO, Oh GS, Choi BM, Jeong S,
Jang SI, Oh H, Kwon TO, Song CE and Chung HT: Inhibition of TNF-α,
IL-1β, and IL-6 productions and NF-κB activation in
lipopolysaccharide-activated RAW 264.7 macrophages by catalposide,
an iri glycoside isolated from Catalpa ovata G. Don (Bignoniaceae).
Int Immunopharmacol. 2:1173–1181. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Bosnar M, Bošnjak B, Čužić S, Hrvacic B,
Marjanovic N, Glojnaric I, Culic O, Parnham MJ and Haber VE:
Azithromycin and clarithromycin inhibit lipopolysaccharide-induced
murine pulmonary neutrophilia mainly through effects on
macrophage-derived granulocyte-macrophage colony-stimulating factor
and interleukin-1β. J Pharmacol Exp Ther. 331:104–113. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Risso K, Kumar G, Ticchioni M, Sanfiorenzo
C, Dellamonica J, Guillouet-de Salvador F, Bernardin G, Marquette
CH and Roger PM: Early infectious acute respiratory distress
syndrome is characterized by activation and proliferation of
alveolar T-cells. Eur J Clin Microbiol Infect Dis. 34:1111–1118.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Janssen WJ, Barthel L, Muldrow A,
Oberley-Deegan RE, Kearns MT, Jakubzick C and Henson PM: Fas
determines differential fates of resident and recruited macrophages
during resolution of acute lung injury. Am J Respir Crit Care Med.
184:547–560. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Chen X, Tang J, Shuai W, Meng J, Feng J
and Han Z: Macrophage polarization and its role in the pathogenesis
of acute lung injury/acute respiratory distress syndrome. Inflamm
Res. 69:883–895. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Williams MA, Rangasamy T, Bauer SM,
Killedar S, Karp M, Kensler TW, Yamamoto M, Breysse P, Biswal S and
Georas SN: Disruption of the transcription factor Nrf2 promotes
pro-oxidative dendritic cells that stimulate Th2-like
immunoresponsiveness upon activation by ambient particulate matter.
J Immunol. 181:4545–4559. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
D'Souza NB, Mandujano FJ, Nelson S, Summer
WR and Shellito JE: CD4+ T lymphocyte depletion attenuates
lipopolysaccharide-induced tumor necrosis factor secretion by
alveolar macrophages in the mouse. Lymphokine Cytokine Res.
13:359–366. 1994.PubMed/NCBI
|
|
47
|
Crowe CR, Chen K, Pociask DA, Alcorn JF,
Krivich C, Enelow RI, Ross TM, Witztum JL and Kolls JK: Critical
role of IL-17RA in immunopathology of influenza infection. J
Immunol. 183:5301–5310. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chai YS, Chen YQ, Lin SH, Xie K, Wang CJ,
Yang YZ and Xu F: Curcumin regulates the differentiation of naïve
CD4+T cells and activates IL-10 immune modulation against acute
lung injury in mice. Biomed Pharmacother. 125:1099462020.
View Article : Google Scholar
|
|
49
|
Philippakis GE, Lazaris AC, Papathomas TG,
Zissis C, Agrogiannis G, Thomopoulou G, Nonni A, Xiromeritis K,
Nikolopoulou-Stamati P, Bramis J, et al: Adrenaline attenuates the
acute lung injury after intratracheal lipopolysaccharide
instillation: An experimental study. Inhal Toxicol. 20:445–453.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Haring JS, Badovinac VP and Harty JT:
Inflaming the CD8+ T cell response. Immunity. 25:19–29. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ilatovskaya DV, Pitts C, Clayton J,
Domondon M, Troncoso M, Pippin S and DeLeon-Pennell KY: CD8+
T-cells negatively regulate inflammation post-myocardial
infarction. Am J Physiol Hear Circ Physiol. 317:H581–H596. 2019.
View Article : Google Scholar
|
|
52
|
den Haan JMM, Kraal G and Bevan MJ:
Cutting edge: Lipopolysaccharide induces IL-10-producing regulatory
CD4 + T cells that suppress the CD8 + T cell response. J Immunol.
178:5429–5433. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hussein MR, Hassan HI, Hofny ERM, Elkholy
M, Fatehy NA, Elmoniem AEA, El-Din AME, Afifi OA and Rashed HG:
Alterations of mononuclear inflammatory cells, CD4/CD8+ T cells,
interleukin 1β, and tumour necrosis factor α in the bronchoalveolar
lavage fluid, peripheral blood, and skin of patients with systemic
sclerosis. J Clin Pathol. 58:178–184. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Cabrera-Perez J, Condotta SA, Badovinac VP
and Griffith TS: Impact of sepsis on CD4 T cell immunity. J Leukoc
Biol. 96:767–777. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hori S, Nomura T and Sakaguchi S: Control
of regulatory T cell development by the transcription factor Foxp3.
J Immunol. 198:981–985. 2017.PubMed/NCBI
|
|
56
|
Ivanov II, McKenzie BS, Zhou L, Tadokoro
CE, Lepelley A, Lafaille JJ, Cua DJ and Littman DR: The orphan
nuclear receptor RORγt directs the differentiation program of
proinflammatory IL-17+ T helper cells. Cell. 126:1121–1133. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Griffin GK, Newton G, Tarrio ML, Bu DX,
Maganto-Garcia E, Azcutia V, Alcaide P, Grabie N, Luscinskas FW,
Croce KJ and Lichtman AH: IL-17 and TNFα sustain neutrophil
recruitment during inflammation through synergistic effects on
endothelial activation1. J Immunol. 188:6287–6299. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wen H, Miao EA and Ting JPY: Mechanisms of
NOD-like receptor-associated inflammasome activation. Immunity.
39:432–441. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Abais JM, Xia M, Zhang Y, Boini KM and Li
PL: Redox regulation of NLRP3 inflammasomes: ROS as trigger or
effector? Antioxidants Redox Signal. 22:1111–1129. 2015. View Article : Google Scholar
|
|
60
|
Zhou R, Tardivel A, Thorens B, Choi I and
Tschopp J: Thioredoxin-interacting protein links oxidative stress
to inflammasome activation. Nat Immunol. 11:136–140. 2010.
View Article : Google Scholar
|
|
61
|
Zhou R, Yazdi AS, Menu P and Tschopp J: A
role for mitochondria in NLRP3 inflammasome activation. Nature.
469:221–226. 2011. View Article : Google Scholar
|
|
62
|
Vomund S, Schäfer A, Parnham MJ, Brüne B
and Von Knethen A: Nrf2, the master regulator of anti-oxidative
responses. Int J Mol Sci. 18:27722017. View Article : Google Scholar
|
|
63
|
Hornung V, Bauernfeind F, Halle A, Samstad
EO, Kono H, Rock KL, Fitzgerald KA and Latz E: Silica crystals and
aluminum salts activate the NALP3 inflammasome through phagosomal
destabilization. Nat Immunol. 9:847–856. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Jais A, Einwallner E, Sharif O, Gossens K,
Lu TTH, Soyal SM, Medgyesi D, Neureiter D, Paier-Pourani J,
Dalgaard K, et al: Heme oxygenase-1 drives metaflammation and
insulin resistance in mouse and man. Cell. 158:25–40. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Tronel C, Rochefort GY, Arlicot N, Bodard
S, Chalon S and Antier D: Oxidative stress is related to the
deleterious effects of heme oxygenase-1 in an in vivo
neuroinflammatory rat model. Oxid Med Cell Longev. 2013:2649352013.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Raghawan AK, Sripada A, Gopinath G,
Pushpanjali P, Kumar Y, Radha V and Swarup G: A disease-associated
mutant of NLRC4 shows enhanced interaction with SUG1 leading to
constitutive fadddependent caspase-8 activation and cell death. J
Biol Chem. 292:1218–1230. 2017. View Article : Google Scholar
|
|
67
|
Pierini R, Juruj C, Perret M, Jones CL,
Mangeot P, Weiss DS and Henry T: AIM2/ASC triggers
caspase-8-dependent apoptosis in francisella-infected
caspase-1-deficient macrophages. Cell Death Differ. 19:1709–1721.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Antonopoulos C, Russo HM, El Sanadi C,
Martin BN, Li X, Kaiser WJ, Mocarski ES and Dubyak GR: Caspase-8 as
an effector and regulator of NLRP3 inflammasome signaling. J Biol
Chem. 290:20167–20184. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Robinson N, Ganesan R, Hegedűs C, Kovács
K, Kufer TA and Virág L: Programmed necrotic cell death of
macrophages: Focus on pyroptosis, necroptosis, and parthanatos.
Redox Biol. 26:1012392019. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Haitao Lee PP: Neutrophil extracellular
traps promoted alveolar macrophages pyroptosis in LPS induced
ALI/ARDS. Eur Respir J. 52:PA42842018.
|
|
71
|
Orning P, Weng D, Starheim K, Ratner D,
Best Z, Lee B, Brooks A, Xia S, Wu H, Kelliher MA, et al: Pathogen
blockade of TAK1 triggers caspase-8-dependent cleavage of gasdermin
D and cell death. Science. 362:1064–1069. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Shi J, Zhao Y, Wang K, Shi X, Wang Y,
Huang H, Zhuang Y, Cai T, Wang F and Shao F: Cleavage of GSDMD by
inflammatory caspases determines pyroptotic cell death. Nature.
526:660–665. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Kayagaki N, Stowe IB, Lee BL, O'Rourke K,
Anderson K, Warming S, Cuellar T, Haley B, Roose-Girma M, Phung QT,
et al: Caspase-11 cleaves gasdermin D for non-canonical
inflammasome signalling. Nature. 526:666–671. 2015. View Article : Google Scholar : PubMed/NCBI
|