Introduction
Acute lung injury (ALI) is a clinical syndrome
induced by several factors, including severe extrapulmonary
infection, non-chest trauma, acute pancreatitis and massive blood
transfusion. ALI is a common cause of respiratory failure in
patients with severe disease and has a high incidence rate and
mortality (1). The mechanism of
ALI is complex and has not yet been fully elucidated; however, it
appears to involve the activation of neutrophils and macrophages,
which produce and release numerous inflammatory mediators (such as
IL-6 and IL-1β) and aggregate on alveolar and pulmonary capillary
endothelial cells to induce inflammatory cascades. Subsequently,
these inflammatory cascades increase the permeability of the
barrier between the pulmonary capillary endothelium and alveolar
epithelial cells, causing edema in the alveoli and pulmonary
interstitium, ultimately resulting in respiratory failure (2). Currently, glucocorticoids
(including methylprednisolone, hydrocortisone and mometasone
furoate) are widely used to treat ALI, even though they have a poor
therapeutic effect and often cause side effects, such as severe
infection secondary to immunosuppression, gastrointestinal
bleeding, elevated blood glucose levels and osteoporosis (3). Due to this unsatisfactory clinical
application of glucocorticoids, there is an urgent need to develop
novel strategies and therapeutics to treat ALI.
Ferroptosis, a recently discovered type of cell
death, is involved in the pathogenesis of diseases caused by
inflammation and oxidative stress (4-6).
The most prominent manifestation of ALI is an uncontrollable
inflammatory response. Recent studies have shown that ferroptosis
is involved in ALI, and the inhibition of ferroptosis can
effectively improve the symptoms of ALI. For example, Yang et
al (7) reported that STAT6
alleviates lipopolysaccharide (LPS)-induced ALI by inhibiting
ferroptosis via the regulation of the p53/SLC7A11 pathway. AU-rich
element-binding factor 1 protects against ferroptosis and
alleviates sepsis-induced ALI by regulating nuclear factor
erythroid 2-related factor 2 (Nrf2) and activating transcription
factor 3 (8). In addition, a
specific ferroptosis inhibitor (ferrostatin-1) has been shown to
effectively alleviate LPS-induced ALI both in vivo and in
vitro (9). These studies have demonstrated that ferroptosis
occurs in ALI and that inhibition of ferroptosis may provide a
novel therapeutic target for ALI, thereby prompting the
identification of potential drugs that inhibit ferroptosis and
ALI.
Nrf2 is the main antioxidant transcription factor
responsible for the regulation of intracellular redox imbalance by
controlling its downstream target gene expression, such as heme
oxygenase 1 and glutathione peroxidase 4 (Gpx4) (10). Studies have shown that Nrf2 is a
vital regulator of ferroptosis in several diseases, including ALI
(8,11-13). Nrf2 is activated by sirtuin 1
(Sirt1), which is also involved in cellular response to stress,
including inflammatory reactions and oxidative stress. The
activation of the Sirt1/Nrf2 signaling pathway can inhibit
ferroptosis and alleviate sepsis-induced acute kidney injury
(14) and acetaminophen-induced
liver injury (15). Gpx4), a
downstream target of Nrf2, is a membrane peroxidase in mammals that
is critical for ferroptosis (10). However, the role of the
Sirt1/Nrf2/Gpx4 signaling pathway in LPS-induced ferroptosis and
ALI remains unclear.
Quercetin, a well-studied flavonoid extensively
found in fruits and vegetables, exhibits anti-fibrotic (16), anti-inflammatory (17) and antioxidative (18) activities. Previous studies have
indicated that quercetin also inhibits ferroptosis in vivo
and in vitro. For example, quercetin exerts beneficial
effects on type 2 diabetes mellitus, potentially by inhibiting
pancreatic β-cell ferroptosis (19). Quercetin also exhibits a
protective role in acute kidney injury via inhibiting ferroptosis
(20). Quercetin alleviates
acrylamide-induced liver injury by inhibiting autophagy-dependent
ferroptosis (21). Furthermore,
quercetin has been found to attenuate ALI in several animal models,
including LPS-induced ALI (22),
cigarette smoke-induced ALI (23) and sepsis-induced ALI (24). However, whether quercetin can
protect against LPS-induced ALI by inhibiting ferroptosis and its
potential mechanisms require further elucidation. Considering that
quercetin is also an effective Sirt1 activator (24,25), the present study investigated
whether quercetin inhibits ferroptosis and alleviates LPS-induced
ALI by activating the Sirt1/Nrf2/Gpx4 pathway.
Materials and methods
Reagents
Primary antibodies against occludin (cat. no.
27260-1-AP), Sirt1 (cat. no. 13161-1-AP) and GAPDH (cat. no.
60004-1-Ig), as well as HRP-conjugated goat anti-mouse (cat. no.
SA00001-1), goat anti-rabbit (cat. no. SA00001-2) and rabbit
anti-goat (cat. no. SA00001-4) secondary antibodies were purchased
from Wuhan Sanying Biotechnology. Primary antibodies against zonula
occludens-1 (ZO-1) (cat. no. ab190085), Gpx4 (cat. no. ab125066),
Nrf2 (cat. no. ab92946) and 4-hydroxynonenal (4-HNE; cat. no.
ab46545) were purchased from Abcam. Quercetin, EX527 (Sirt1
inhibitor) and ferrostatin-1 were obtained from Selleck Chemicals.
LPS (055: B5) was obtained from Beijing Solarbio Science &
Technology Co., Ltd.
Cell culture and transfection
Primary mouse type II alveolar epithelial (AT2)
cells (cat. no. CP-M003) were obtained from Procell Life Science
& Technology Co., Ltd. Cells were cultured in DMEM/F-12
containing 10% FBS and maintained at 37°C in a humidified 5%
CO2 incubator. The Sirt1 small interfering (si)RNA and
negative control (NC) sequences were designed and synthesized by
Genomeditech (Shanghai) Co., Ltd. The siRNA sequences were as
follows: siSirt1#1 sense, 5′-GCU GAG GUA UAU UCA GAC U-3′ and
antisense, 5′-AGU CUG AAU AUA CCU CAG C-3′; siSirt1#2 sense, 5′-GAC
UCA AGU UCA CCU GAA AGA-3′ and antisense, 5′-UCU UUC AGG UGA ACU
UGA GUC-3′; siSirt1#3 sense, 5′-CCU CAA GCG AUG UUU GAU A-3′ and
antisense, 5′-UAU CAA ACA UCG CUU GAG G-3′; and siNC sense, 5′-UUC
UCC GAA CGU GUC ACG U-3′ and antisense, 5′-ACG UGA CAC GUU CGG A
GAA-3′. Cell transfection was performed according to the
manufacturer's instructions of Lipofectamine 3000 (Invitrogen;
Thermo Fisher Scientific, Inc.). The final concentration of siRNA)
was 20 nM. After 6 h of culture at 37°C in a humidified 5%
CO2 incubator, the medium that transfection medium was
replaced with fresh complete culture medium. Subsequent experiments
were performed after 36 h.
LPS-induced AT2 cell injury model
To explore the protective effects of quercetin in
vitro, an LPS-induced AT2 cell injury model was established.
Briefly, AT2 cells were seeded in 96-well plates (5,000 cells/well
or 6-well plates (1×105 cells/well), pretreated with
quercetin (0, 5, 10 and 20 μM) for 2 h, and then LPS (5, 10
or 20 μg/ml) was used to induce the acute cell injury model.
The cells were cultured at 37°C in a 5% CO2 incubator.
Cells were collected at 6, 12 and 24 h for subsequent
experiments.
Cell Counting Kit-8 (CCK-8), cell death
and lactate dehydrogenase (LDH) release assays
CCK-8, cell death and LDH release assays were
performed to measure cell injury as previously described (26). CCK-8 (cat. no. C0039), LDH (cat.
no. C0016) and cell death reagents (7-AAD; cat. no. C1053S) were
purchased from Beyotime Institute of Biotechnology. AT2 cells were
seeded in 96-well plates (5,000 cells/well) for CCK-8 and LDH
assays or 6-well plates (1×105 cells/well) for cell
death detection. For the CCK-8 assay, 10 μl CCK-8 reagent
was added to each well, incubated at 37°C for 1 h, and the optical
density value was measured at 450 nm. For cell death detection,
after the AT2 cells were treated as aforementioned, the cells were
digested with trypsin without EDTA, and then the cells were
harvested and washed twice with PBS. A total of 500 μl 7-AAD
working solution was added to each well and incubated at 37°C for
10 min. Cell death was detected using a flow cytometer
(FACSCalibur; Becton, Dickinson and Company). CytExpert 2.4
software (Beckman Coulter, Inc.) was used to analyze the data.
Reverse transcription-quantitative PCR
(RT-qPCR)
RT-qPCR was performed as previously described
(27). Total RNA was extracted
from AT2 cells according to the instructions of the Total RNA
Extraction Kit (Beijing Solarbio Science & Technology Co.,
Ltd.). RNA samples were reverse transcribed using the iScript cDNA
Synthesis Kit (Bio-Rad Laboratories, Inc.) according to the
manufacturer's instructions. The cDNAs were then added to the
reaction with the indicated primers using SYBR Green Supermix
(Bio-Rad Laboratories, Inc.) according to the manufacturer's
instructions and amplified using qPCR, performed at 95°C for 2 min,
followed by 40 cycles of amplification at 95°C for 10 sec, 62°C for
30 sec and 72°C for 30 sec, using a CFX96 Real-Time PCR Detection
System (Bio-Rad Laboratories, Inc.). Data were analyzed with
2-ΔΔCq method (28),
using β-actin for normalization. The following primers were used:
Sirt1-forward, 5′-TCC TTC AGT GTC ATG GTT CCT TTG C-3′ and reverse,
5′-CTC CAC GAA CAG CTT CAC AAT CAA C-3′; and β-actin forward,
5′-GTG CTA TGT TGC TCT AGA CTT CG-3′ and reverse, 5′-ATG CCA CAG
GAT TCC ATA CC-3′.
Animals, experimental design and animal
welfare
A total of 102 male C57BL/6 mice (age, 6-8 weeks;
weight, 18-22 g) were purchased from Chengdu Dossy Experimental
Animals Co., Ltd. (https://www.cd-dossy.cn/). All mice were housed in
cages with free access to food and water at 25°C with a 12/12-h
light/dark cycle and ~55% relative humidity, and were quarantined
and acclimatized before the experiment. The mice were randomly
divided into five groups (n=6 mice/group): Control (saline), LPS (5
mg/kg, dissolved in saline) and LPS + quercetin (20, 40 or 60
mg/kg, dissolved in DMSO) for lung injury detection. The ALI model
was established based on previous studies (29). Briefly, quercetin (20, 40 or 60
mg/kg) was administered via oral gavage. For ferroptosis detection,
the mice were randomly divided into four groups (n=6 mice/group):
Control (saline), LPS (5 mg/kg, dissolved in saline), quercetin (40
mg/kg, dissolved in DMSO), LPS + quercetin. Inhibition of Sirt1
in vivo, mice were divided into four groups (n=6
mice/group): Control (saline), LPS (5 mg/kg), LPS + quercetin (40
mg/kg) and LPS + quercetin + EX527 (5 mg/kg). For alveolar
epithelial barrier integrity detection, the mice were randomly
divided into four groups (n=6 mice/group): Control (saline), LPS (5
mg/kg), LPS + quercetin (40 mg/kg), LPS + ferrostatin-1 (5 mg/kg).
The use of ferrostatin-1 as a ferroptosis inhibitor was based on
our previous study (27). After
1 h of treatment with quercetin, Sirt1 inhibitor (EX527) or
ferrostatin-1, the mice were anesthetized with sodium pentobarbital
(intraperitoneal injection, 50 mg/kg), and LPS was intratracheally
injected to induce lung injury. A total of 12 h after LPS
administration, the mice were euthanized by intraperitoneal
injection of 120 mg/kg sodium pentobarbital, and the lung tissue
and bronchoalveolar lavage fluid (BALF) were collected. Death was
verified by observing the cardiac arrest and spontaneous
respiratory arrest.
During the experimental process, the health and
behavior of the animals were monitored hourly. Humane endpoints
included indicators, such as huddled posture, immobility, ruffled
fur, failure to eat, hypothermia (colonic temperature of <34°C),
weight loss >20%, inability to stand, agonal breathing, severe
muscular atrophy, severe ulceration or uncontrolled bleeding. There
was no accidental death of mice during the present study, and all
the mice were euthanized at the end of the experiment.
Immunohistochemical (IHC) analysis,
immunofluorescence (IF), western blotting and hematoxylin and eosin
(H&E) staining
IHC, IF and western blot assays were used to
evaluate the protein levels of Sirt1, Nrf2, ZO-1, occludin, 4-HNE
and Gpx4. For IHC, mouse lung tissues were fixed overnight with 4%
paraformaldehyde at room temperature, followed by paraffin
embedding and slicing (5 μm). The lung tissue sections were
waxed at 65°C for 2 h, dewaxed twice in xylene (5 min each time),
immersed in 100, 95, 85 and 75 % ethanol for 2 min each, and then
immersed in PBS for 2 min. Subsequently, the tissue sections were
immersed in EDTA Antigen Retrieval solution (1:50, diluted in
deionized water; cat. no. P0085; Beyotime Institute of
Biotechnology) in a pressure cooker for 10 min, cooled naturally at
room temperature, and washed twice with PBS. After incubation in 3%
H2O2 for 10 min, the sections were washed
three times with PBS. Blocking with 5% goat serum (cat. no. C0265;
Beyotime Institute of Biotechnology) was performed at room
temperature for 30 min, following by primary antibody addition at
4°C overnight. On the next day, the sections were washed twice with
PBS, followed by incubation with the secondary antibody
(HRP-labelled; ready-to-use; cat. no. PV-9000; OriGene
Technologies, Inc.) was added to cover the lung tissue and
incubated at 37°C for 20 min, and the sections were washed twice
with PBS (5 min each). DAB (cat. no. ZLI-9017; OriGene
Technologies, Inc.) was used to stain the sections. Finally, the
sections were incubated with hematoxylin staining solution for 1.5
min at room temperature.
For IF, AT2 cells were fixed with 4%
paraformaldehyde at room temperature for 15 min, incubated with
0.1% Triton X-100 for 5 min to permeate the cell membrane, and
blocked with 5% goat serum at room temperature for 30 min, followed
by primary antibody incubation (1:100) overnight at 4°C. The
secondary antibody, Cy3-labeled goat anti-rabbit (cat. no. A0516;
Beyotime Institute of Biotechnology) or Cy3-labeled donkey
anti-goat (cat. no. A0502; Beyotime Institute of Biotechnology) was
diluted 1:300 and incubated at 37°C in the dark for 1 h. After
washing with PBS three times, DAPI (1:5,000; diluted in PBS; cat.
no. C0060; Beijing Solarbio Science & Technology Co., Ltd.) was
added at 37°C in the dark for 5 min.
For WB, AT2 cells or lung tissue proteins were
extracted using RIPA lysis Buffer (cat. no. P0013C; Beyotime
Institute of Biotechnology,) and protein determination was
performed with BCA. Protein samples (30 μg) were separated
on 10% gels using SDS-PAGE and transferred to 0.2-μm
polyvinylidene fluoride membranes. The membranes were blocked using
Tris-buffered saline containing 0.1% Tween-20 and 5% skimmed milk
for 1 h at 25°C. The membranes were incubated with the primary
antibodies overnight at 4°C. After washing, the membranes were
incubated with corresponding HRP-conjugated secondary antibodies
(1:5,000) at 25°C for 1.5 h. The bands were visualized in
conjunction with ECL Detection Reagents (EMD Millipore) using the
Quantity One 5.2 software (Bio-Rad Laboratories, Inc.). All primary
antibodies were used at a 1:100 dilution for IHC and IF and 1:1,000
dilution for western blotting.
H&E staining was performed as previously
described (30). The hematoxylin
staining solution was incubated for 1.5 min and the eosin dye
solution for 30 sec at room temperature. Random images for IHC, IF,
and H&E were captured at a magnification of ×200 or ×400 using
a fluorescence microscope (DM4000B; Leica Microsystems, Inc.) and
analyzed using ImageJ v1.48 software (National Institutes of
Health). The severity of lung injury was scored was evaluated based
on the alveolar wall thickness, the amount of cellular infiltration
and the level of hemorrhaging, as previously described with minor
modifications (31). Lung injury
scores were graded on a scale of 0-8 as follows: 0, no injury; 2,
mild injury (up to 25% injury of the field of view); 4, moderate
injury (up to 50% injury of the field of view); 6, severe injury
(up to 75% injury of the field of view); and 8, extremely severe
injury (diffused injury).
ELISA
The levels of IL-6 (cat. no. ml098430), IL-1β (cat.
no. ml098416) and TNF-α (cat. no. mIC50536-1) in mouse BALF were
detected using ELISA kits (Shanghai Enzyme-linked Biotechnology
Co., Ltd.) according to the manufacturer's protocols. A
myeloperoxidase (MPO) activity assay kit (cat. no. RXSH0539;
Quanzhou Ruixin Biotechnology Co., Ltd.) was used to measure MPO
activity in lung tissues.
Measurement of reactive oxygen species
(ROS), glutathione (GSH), iron and lipid peroxidation levels
Dihydroethidium (DHE; Molecular Probes; Thermo
Fisher Scientific, Inc.) staining was used to determine ROS levels
in the lung tissues. Briefly, the DHE probe was diluted with PBS
(1:5,000) and added to the lung tissue sections at room temperature
for 10 min. ROS levels in AT2 cells were measured using a ROS Assay
kit (cat. no. S0033S; Beyotime Institute of Biotechnology) by flow
cytometry as previously described (23). Briefly, AT2 cells, seeded in
6-well plates (1×105/well), were treated as
aforementioned and incubated with RPMI-1640 medium containing
dichlorodihydrofluorescein diacetate (10 μM; included in the
ROS kit) at 37°C for 20 min. The cells were then washed three times
with PBS, and fluorescence intensity was determined with a flow
cytometer (FACSCalibur; Becton, Dickinson and Company). CytExpert
2.4 software (Beckman Coulter, Inc.) was to analyze the data. GSH
levels in AT2 cells and lung tissues were measured using a GSH
assay kit (cat. no. BC1170; Beijing Solarbio Science &
Technology Co., Ltd.). Lipid peroxidation levels and relative iron
concentrations in AT2 cells and lung tissues were evaluated by
measuring malondialdehyde (MDA) concentrations using a lipid
peroxidation assay kit (cat. no. S0131M; Beyotime Institute of
Biotechnology) and an Iron Assay Kit (cat. no. E1042; Applygen
Technologies, Inc.), respectively. All kits were used according to
the manufacturers' instructions.
Lung wet/dry (W/D) weight
measurement
The W/D ratio measurement of lung samples were
conducted as previously described (27). For W/D ratio measurement, after
the mice were sacrificed the whole lung tissue was excised and
quickly rinsed with pre-cooled normal saline. A filter paper was
used to remove excess water, and the weight of the lung tissue of
each group of mice was immediately weighed. At this point, the
measurement data was recorded as W weight. Subsequently, the lung
tissue was dried in a constant temperature oven at 65°C for 48 h,
and then weighed. The measured data was recorded as D weight.
5-Ethynyl-2′-deoxyuridine (EDU)
The EDU assay was conducted as previously described
(27). Briefly, AT2 cells,
seeded in 24-well plates (2×104/well), were treated as
aforementioned and incubated with EDU (cat. no. C0071S; Beyotime
Institute of Biotechnology) working solution at 37°C for 30 min.
After washing with PBS three times, DAPI (1:5,000, diluted in PBS)
was added at 37°C in the dark for 5 min. Random images were
captured at ×200 magnification using a fluorescence microscope
(Leica Microsystems, Inc.).
Transmission electron microscopy
(TEM)
The mitochondrial morphology of AT2 cells was
analyzed via TEM (FEIG2; Thermo Fisher Scientific, Inc.) as
previously described (27).
Briefly, the cells were first fixed in 2.5% glutaraldehyde and then
incubated in 0.1 M osmium tetroxide (prepared in 0.1 M PBS, pH 7.4)
for 2 h or longer at room temperature. Following further
dehydration, permeation and embedding (in araldite) steps,
ultra-thin sections (65 nm) were obtained and viewed with the
transmission electron microscope.
Statistical analysis
All in vitro experiments were performed
independently at least three times, and all animals were randomly
assigned to their experimental groups. Data are presented as the
mean ± standard deviation or as the median + interquartile range
and were analyzed using GraphPad Prism 7 software (GraphPad
Software; Dotmatics). Statistical significance was determined using
one-way ANOVA followed by Tukey's post hoc test or Kruskal-Wallis
followed by Dunn's post hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
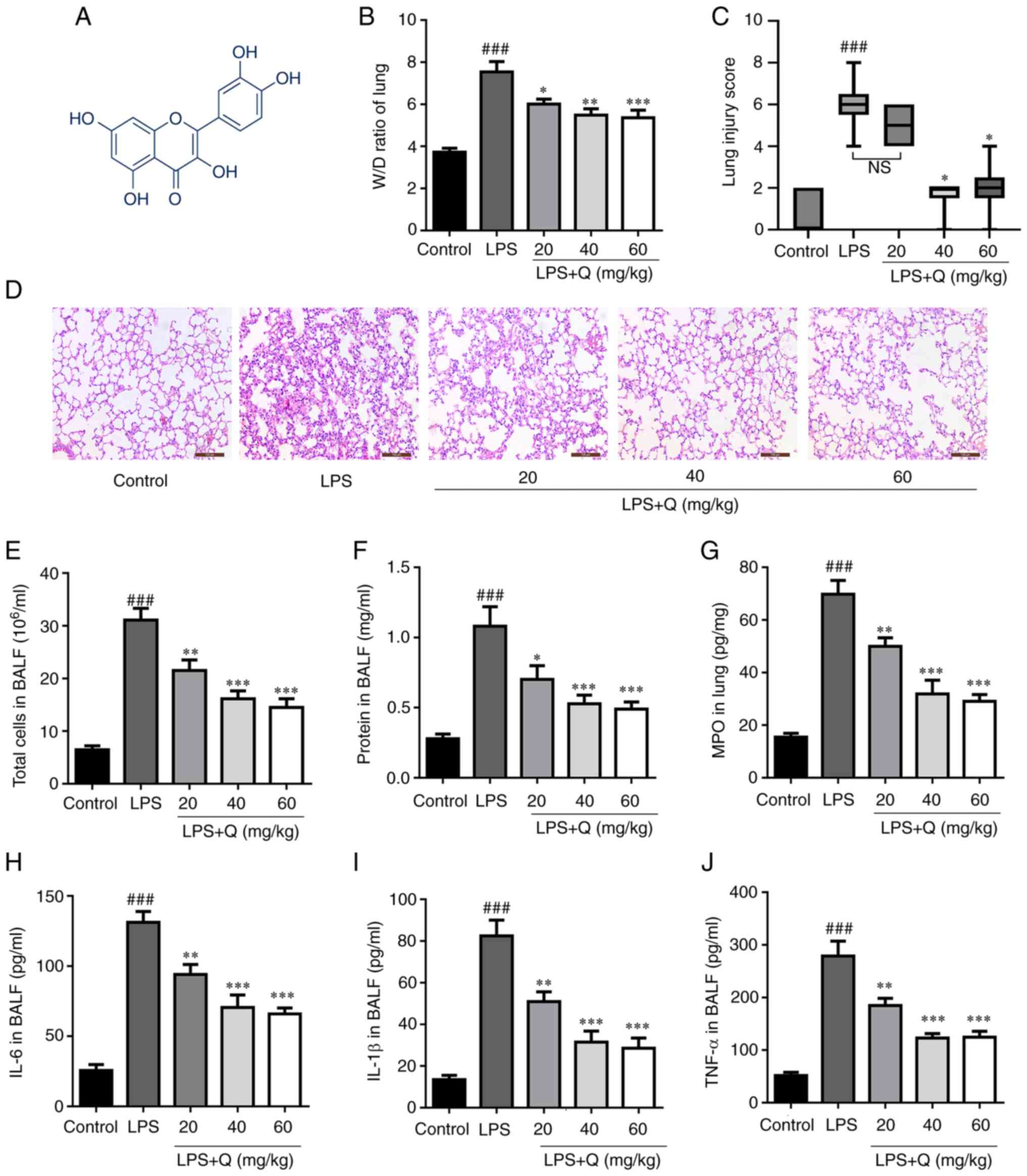
Treatment with quercetin alleviates
LPS-induced ALI in mice
Firstly, LPS was used to establish a mouse model of
ALI and examine the potential protective effects of quercetin
(Fig. 1A) against ALI. The
results showed that quercetin reduced LPS-induced edema (Fig. 1B) and decreased the alveolar wall
thickness and the amount of cellular infiltration induced by LPS
(Fig. 1C and D), as measured by
the lung injury score. Furthermore, the total cell count and
protein concentrations in BALF were markedly increased after LPS
treatment compared with the control group (Fig. 1E and F). However, pretreatment
with quercetin markedly decreased the total number of cells and
protein leakage in BALF. In addition, quercetin significantly
decreased MPO activity compared with the LPS-only group (Fig. 1G). Moreover, quercetin
pretreatment effectively reduced TNF-α, IL-6 and IL-1β levels in
mouse BALF (Fig. 1H-J).
Collectively, the present results indicate that quercetin
significantly alleviated the pathological changes and inflammatory
response in the lung tissues of LPS-induced mice.
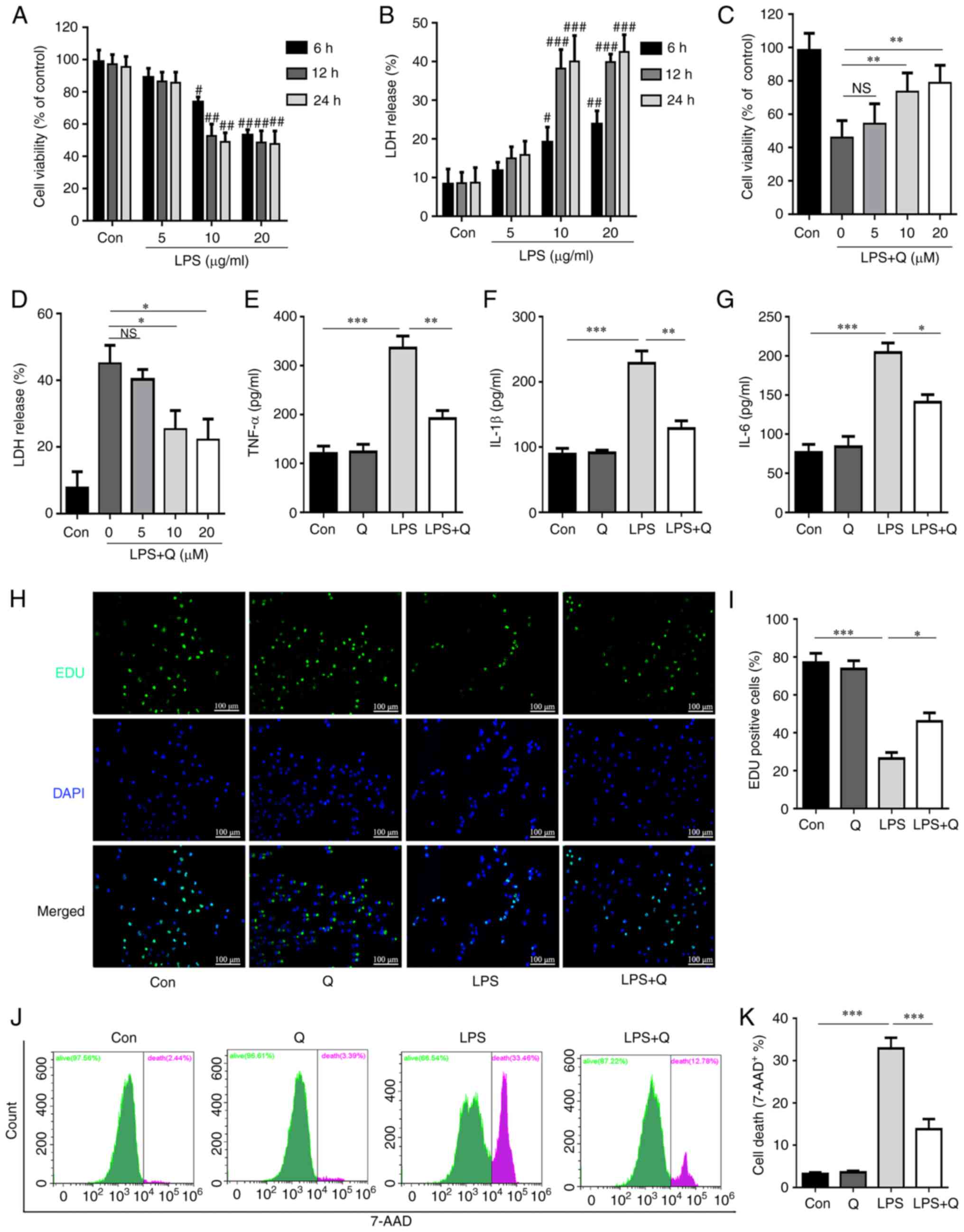
Quercetin alleviates LPS-induced alveolar
epithelial cell injury
To further evaluate the effects of quercetin on
LPS-induced alveolar epithelial cell injury, an LPS-induced AT2
cell injury model was established. The CCK-8 and LDH release assays
showed that LPS treatment markedly inhibited the proliferation of
AT2 cells and increased the LDH release in a time- and
dose-dependent manner (Fig. 2A and
B). Next, to determine the most effective quercetin
concentration, AT2 cells were separated into five groups: Control
(saline), LPS (10 μM) and LPS + quercetin (5, 10 or 20
μM). CCK-8 and LDH release assays were subsequently
performed (Fig. 2C and D). The
results indicated that 10 and 20 μM quercetin had similar
protective effects on the viability and LDH release of AT2 cells.
Based on these data, a 10 μM concentration for both LPS and
quercetin, as well as the 12-h time point were selected for
subsequent cell experiments. The results demonstrated that
quercetin treatment significantly reduced inflammatory cytokine
secretion (Fig. 2E-G), promoted
cell proliferation (Fig. 2H and
I) and inhibited cell death (Fig. 2J and K) after LPS treatment.
Collectively, these results suggest that quercetin exhibited
protective effects against LPS-induced alveolar epithelial cell
injury.
Quercetin inhibits ferroptosis in the
LPS-induced ALI mouse model via the Sirt1/Nrf2/Gpx4 pathway
To investigate whether quercetin inhibits
ferroptosis in ALI, ferroptosis parameters were evaluated in the
LPS-induced ALI mouse model. Quercetin decreased the alveolar wall
thickness and the amount of cellular infiltration in LPS-treated
mice, as evaluated through H&E staining (Fig. 3A) and the lung injury score
(Fig. 3B). In addition,
quercetin significantly attenuated LPS-induced ferroptosis in the
lungs of mice with ALI. Specifically, quercetin reversed the
decrease in the levels of GSH (Fig.
3C) and Gpx4 (Fig. 3H and J)
and reduced the levels of MDA (Fig.
3D), iron (Fig. 3E), ROS
(Fig. 3F and G) and 4-HNE
(Fig. 3I and J) in LPS-treated
mice. To explore whether quercetin modulated the Sirt1/Nrf2/Gpx4
signaling pathway in LPS-induced ALI, the protein expression levels
of Sirt1, Nrf2 and Gpx4 were detected. As shown in Fig. 3K-N, the protein expression of
Sirt1, Nrf2 and Gpx4 was upregulated in the LPS + quercetin group
compared with that in the LPS group. Collectively, these results
indicate that quercetin inhibited LPS-induced ferroptosis via the
Sirt1/Nrf2/Gpx4 pathway.
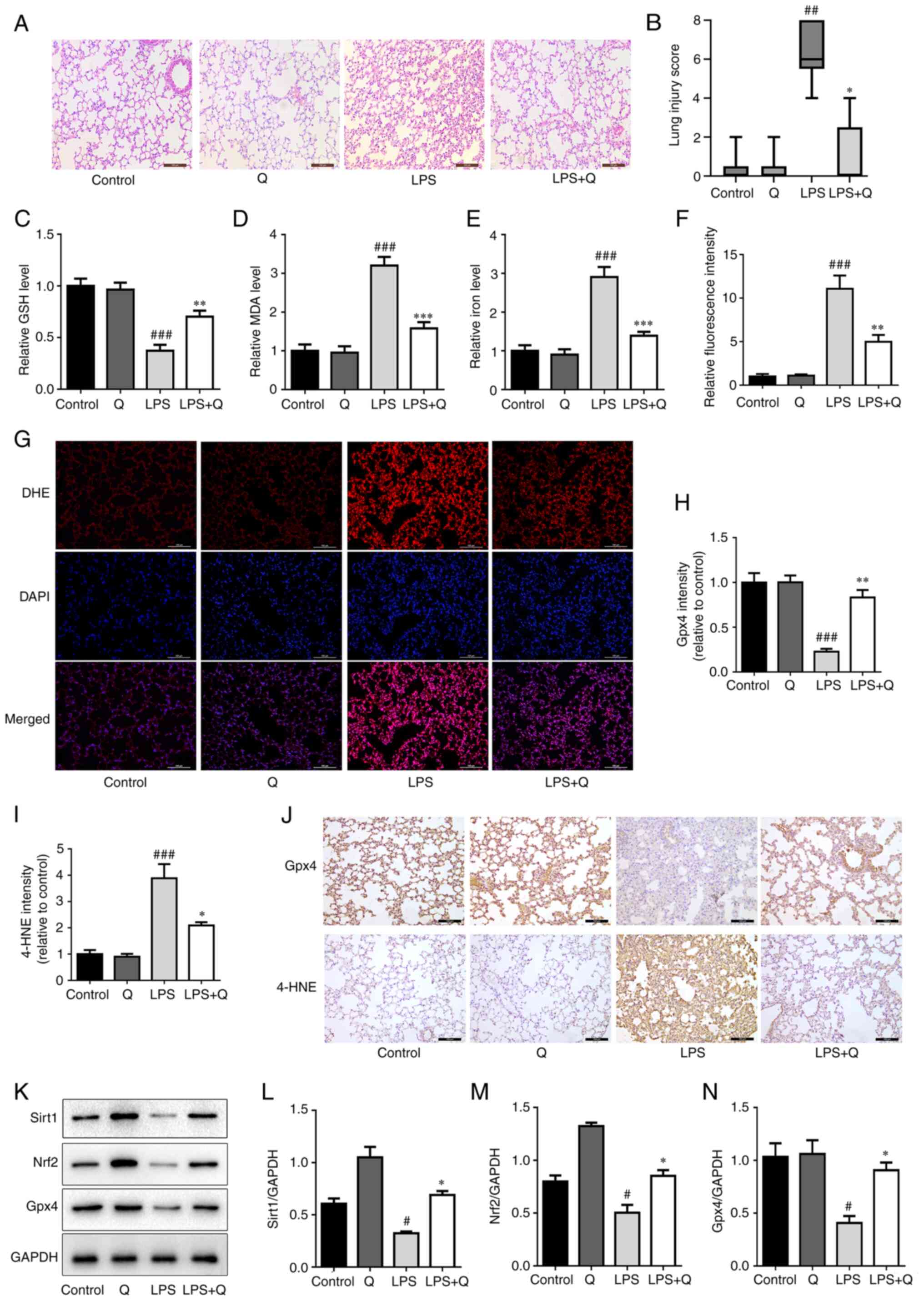 | Figure 3Quercetin inhibits ferroptosis in the
LPS-induced acute lung injury mouse model via the Sirt1/Nrf2/Gpx4
pathway. (A) Representative hematoxylin and eosin-stained images of
lung tissues (scale bar, 100 μm). (B) Lung injury score.
Levels of (C) GSH, (D) MDA and (E) iron in mouse lung tissues. (F)
Quantitative results DHE. (G) Levels of reactive oxygen species in
mouse lung tissues were assessed by DHE staining (scale bar, 100
μm). Quantitative results of IHC images of (H) Gpx4 and (I)
4-HNE in mouse lung tissues. (J) Representative IHC images of Gpx4
and 4-HNE in mouse lung tissues (scale bar, 100 μm). (K)
Protein expression levels of Sirt1, Nrf2 and Gpx4 in mouse lung
tissues detected through western blotting. Relative protein
expression quantitative analysis of (L) Sirt1, (M) Nrf2 and (N)
Gpx4. #P<0.05, ##P<0.01 and
###P<0.001 vs. control; *P<0.05,
**P<0.01 and ***P<0.001 vs. LPS. GSH,
glutathione; MDA, malondialdehyde; LPS, lipopolysaccharide; Q,
quercetin; Gpx4, glutathione peroxidase 4; 4-HNE, 4-hydroxynonenal;
Nrf2, nuclear factor erythroid 2-related factor 2; Sirt1, sirtuin
1; DHE, dihydroethidium; IHC, immunohistochemistry. |
Quercetin inhibits ferroptosis in the
LPS-induced alveolar epithelial cell injury model via the
Sirt1/Nrf2/Gpx4 pathway
Consistent with the in vivo results,
quercetin inhibited the decrease in the levels of GSH (Fig. 4A) and Gpx4 (Fig. 4E and F) and reduced the levels of
MDA (Fig. 4B), iron (Fig. 4C), ROS (Fig. 4D) and 4-HNE (Fig. 4E and G) in the LPS-induced AT2
cell model. Moreover, quercetin inhibited the LPS-induced
mitochondrial shrinkage and decreased the mitochondrial cristae
(Fig. 4H). Similarly, the
expression levels of Sirt1, Nrf2 and Gpx4 were significantly
decreased after LPS treatment compared with the control group, and
quercetin effectively reversed this decrease (Fig. 4I-L). These data suggest that
quercetin inhibited LPS-induced ferroptosis in alveolar epithelial
cells via the Sirt1/Nrf2/Gpx4 pathway.
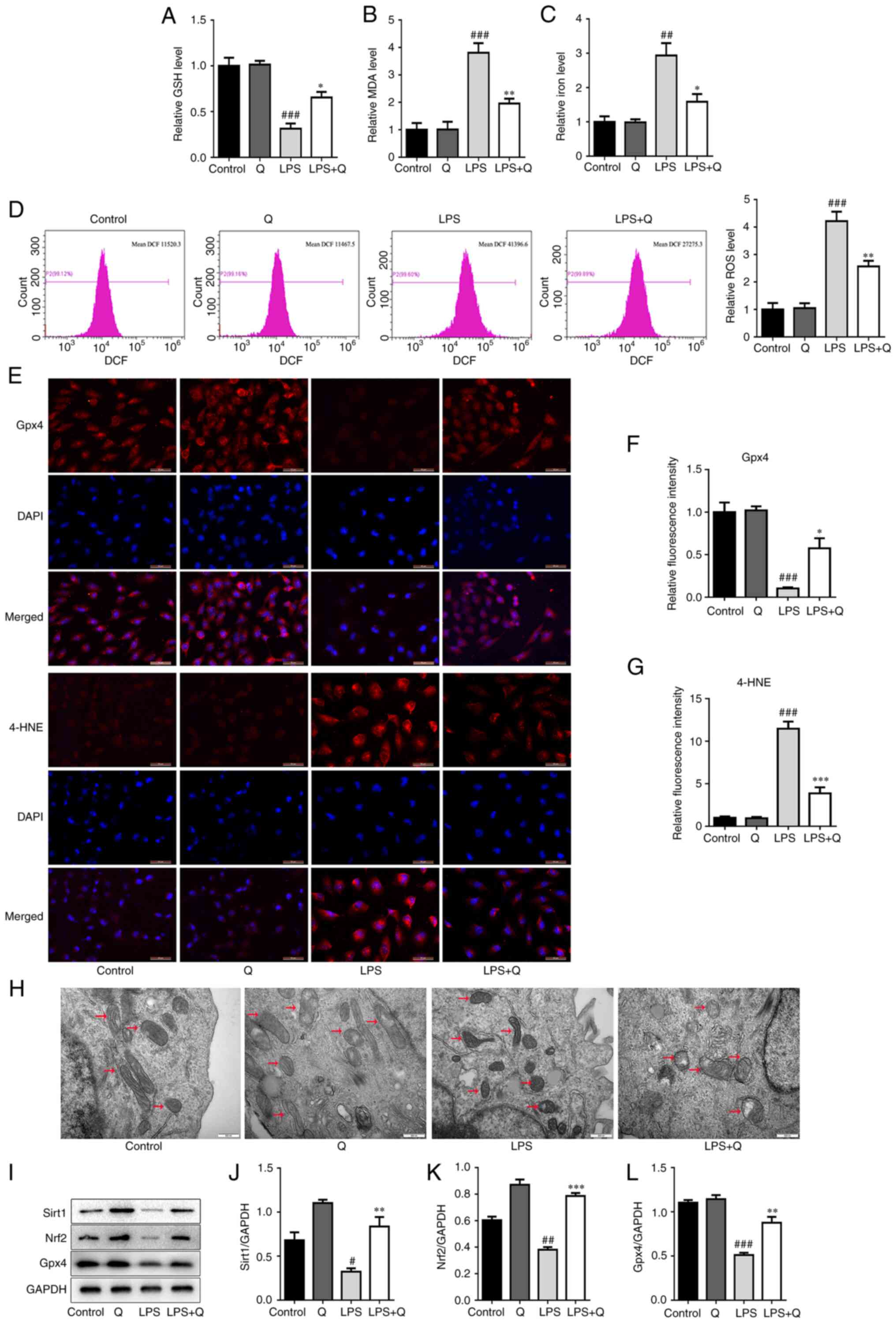 | Figure 4Quercetin inhibits ferroptosis in the
LPS-induced alveolar epithelial cell injury model via the
Sirt1/Nrf2/Gpx4 pathway. Levels of (A) GSH, (B) MDA, (C) iron in in
AT2 cells. (D) Representative flow cytometry images (left) and
quantitative analysis (right) of ROS in AT2 cells. (E)
Representative immunofluorescence images (scale bar, 50 μm)
and relative fluorescence intensity of (F) Gpx4 and (G) 4-HNE in
alveolar epithelial cells. (H) Transmission electron microscopy
images of representative mitochondrial structures (indicated by
arrows) in AT2 cells (scale bar, 500 nm). (I) Western blotting and
quantitative analysis of the protein levels of (J) Sirt1, (K) Nrf2
and (L) Gpx4 in alveolar epithelial cells. #P<0.05,
##P<0.01 and ###P<0.001 vs. control;
*P<0.05, **P<0.01 and
***P<0.001 vs. LPS. GSH, glutathione; MDA,
malondialdehyde; ROS, reactive oxygen species; LPS,
lipopolysaccharide; Q, quercetin; Gpx4, glutathione peroxidase 4;
4-HNE, 4-hydroxynonenal; Nrf2, nuclear factor erythroid 2-related
factor 2; Sirt1, sirtuin 1. |
Inhibition of Sirt1 expression reverses
the protective effect of quercetin against LPS-induced ALI and
ferroptosis
To further confirm that quercetin alleviates ALI by
inhibiting ferroptosis via the Sirt1 pathway, in vivo
experiments were performed using the Sirt1 inhibitor EX527 (5
mg/kg) based on a previous study (32). EX527 abolished the protective
effect of quercetin on LPS-induced ALI (Fig. 5A-D, F and G) and ferroptosis
(Fig. 5E and H-L). The results
indicated that quercetin markedly reduced LPS-induced edema
(Fig. 5A), alveolar wall
thickness and the amount of cellular infiltration (Fig. 5F and G) and MPO activity in lung
tissue (Fig. 5D) and decreased
the total number of cells (Fig.
5B) and protein leakage (Fig.
5C) in BALF. However, EX527 reversed these protective effects
of quercetin. Furthermore, EX527 reversed the effect of quercetin
on inhibiting Gpx4 degradation (Fig.
5J and K) and reducing the levels of iron (Fig. 5E), ROS (Fig. 5H and I) and 4-HNE (Fig. 5J and L) in LPS-treated mice.
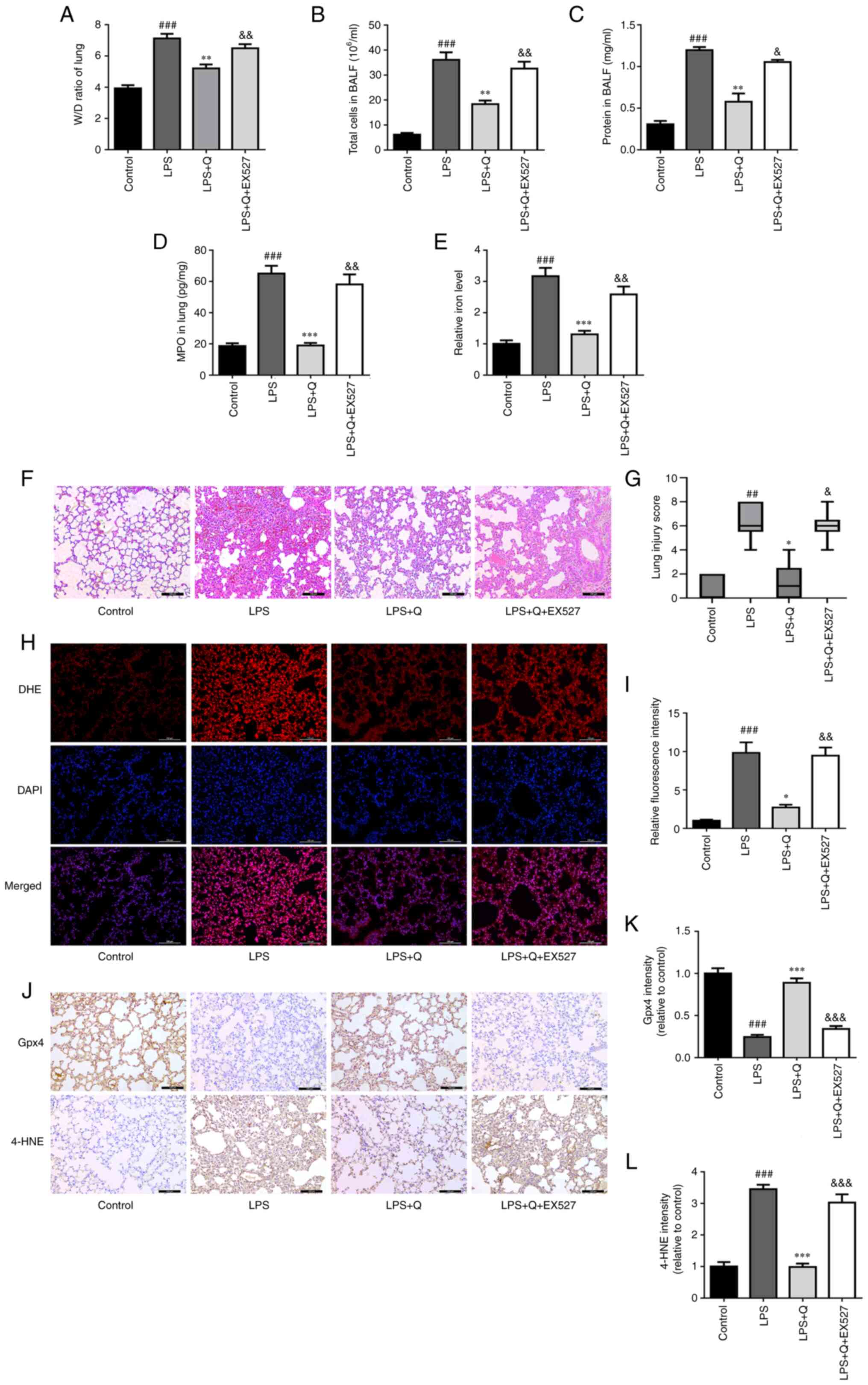 | Figure 5Sirtuin 1 inhibition reverses the
protective effect of quercetin against LPS-induced acute lung
injury and ferroptosis. (A) Lung W/D ratio. (B) Total cell and (C)
protein concentration in BALF. (D) MPO and (E) relative iron levels
in the lungs. (F) Representative hematoxylin and eosin-stained
images of lung tissues (scale bar, 100 μm). (G) Lung injury
score. (H) Representative DHE fluorescence images of mouse lung
tissues (scale bar, 100 μm). (I) Relative DHE fluorescence
intensity. (J) Representative immunohistochemistry images (scale
bar, 100 μm) and quantitative analysis of (K) Gpx4 and (L)
4-HNE in lung tissues. ##P<0.01 and
###P<0.001 vs. control; *P<0.05,
**P<0.01 and ***P<0.001 vs. LPS;
&P<0.05, &&P<0.01 and
&&&P<0.001 vs. LPS + Q. W/D, wet/dry;
BALF, bronchoalveolar lavage fluid; MPO, myeloperoxidase; LPS,
lipopolysaccharide; Q, quercetin; Gpx4, glutathione peroxidase 4;
4-HNE, 4-hydroxynonenal; DHE, dihydroethidium. |
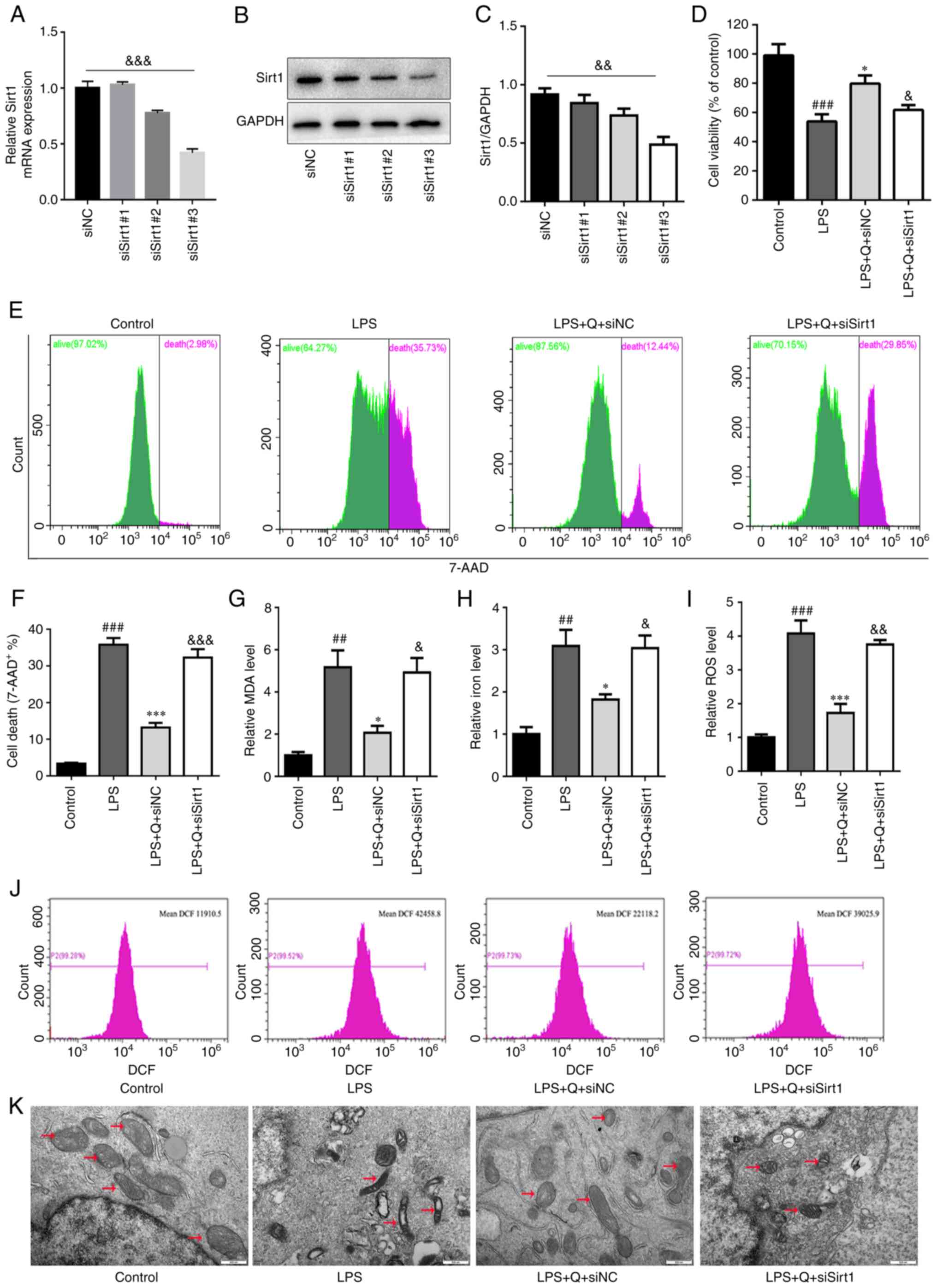
Sirt1 knockdown abrogates the
quercetin-induced inhibition of ferroptosis in vitro
To further verify the role of Sirt1 in the
quercetin-induced inhibition of ferroptosis in vitro, we
downregulated Sirt1 expression was knocked down in primary mouse
AT2 cells using siRNA. The knockdown efficiency was confirmed by
RT-qPCR and western blotting (Fig.
6A-C), and siSirt1#3 was selected for subsequent experiments.
Sirt1 knockdown reduced the protective effect of quercetin against
LPS-induced cell injury, including reducing the viability and
increasing the death of AT2 cells (Fig. 6D-F). Moreover, Sirt1 knockdown
abolished the protective effect of quercetin on LPS-induced
ferroptosis in AT2 cells, including increasing the levels of ROS
(Fig. 6I and J), MDA (Fig. 6G) and iron (Fig. 6H), and inducing mitochondrial
shrinkage (Fig. 6K), confirming
the previous findings.
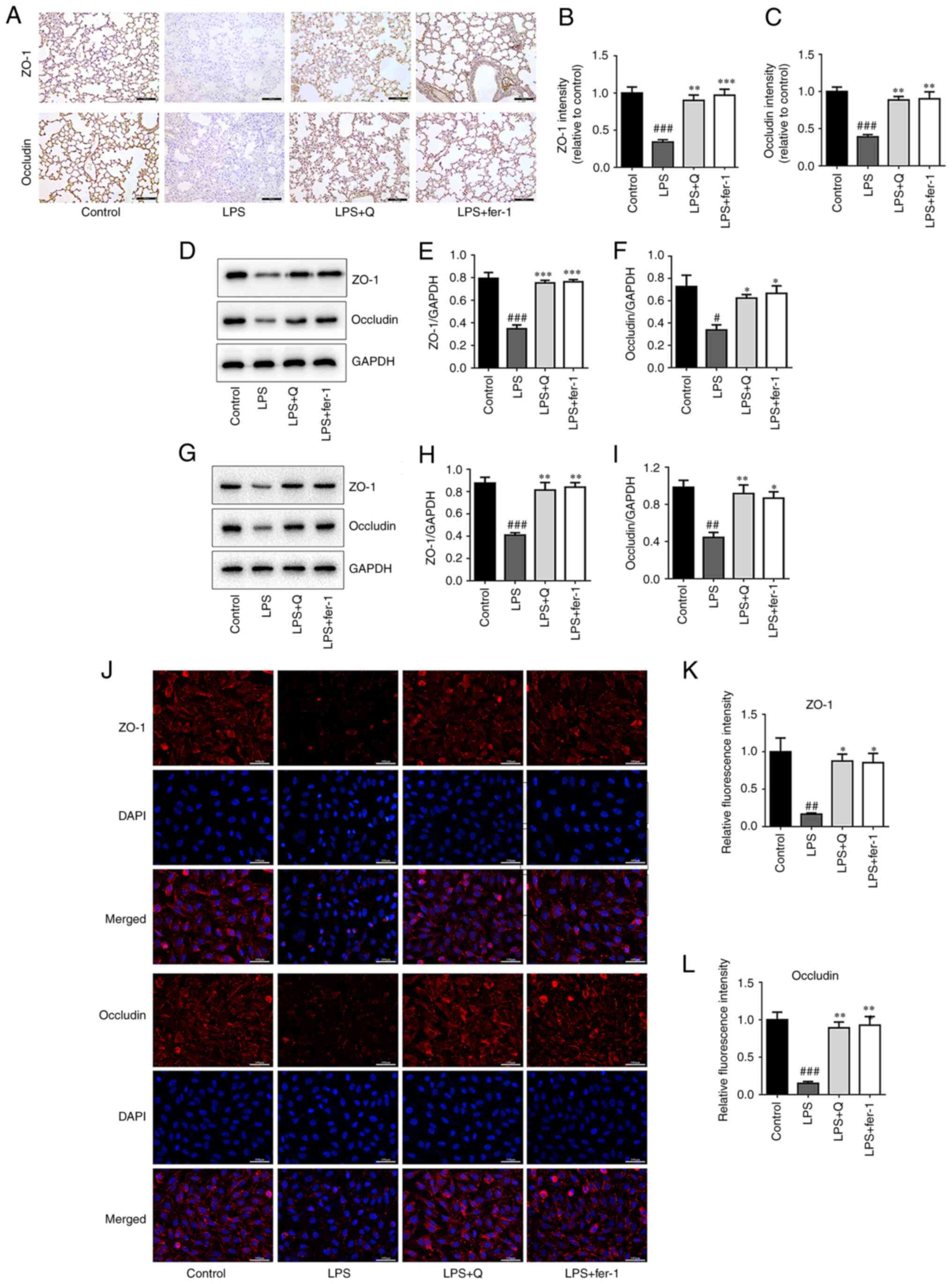
Quercetin attenuates the reduction in
tight junction protein levels following LPS treatment in vivo and
in vitro
Using LPS-induced ALI in vivo and in
vitro models, the tight junction protein expression levels of
ZO-1 and occludin were evaluated using IHC (Fig. 7A-C), western blotting (Fig. 7D-I) and IF (Fig. 7J-L). The expression of these
proteins was significantly decreased following LPS treatment. To
demonstrate the role of ferroptosis in LPS-induced epithelial
barrier destruction, a ferroptosis inhibitor was used, the dose of
which was selected based on our previous study (27). The results showed that both
quercetin and ferrostatin-1, significantly enhanced the protein
expression of ZO-1 and occludin in LPS-treated mice and cells.
Together, these results indicate that quercetin attenuated the
LPS-induced reduction of tight junction proteins both in
vivo and in vitro by preventing ferroptosis.
Discussion
ALI has received increased public attention since
the onset of coronavirus disease 2019 (33). However, sepsis remains the main
cause of ALI (34), rendering it
the leading cause of death in patients with sepsis. The molecular
mechanism of sepsis-induced ALI is complex and has not been fully
elucidated. Excessive oxidative stress-mediated programmed cell
death, such as ferroptosis and pyroptosis, plays a crucial role in
ALI pathogenesis (4,8,29,32). The bacterial endotoxin LPS is a
major trigger of acute inflammation in sepsis-induced ALI (35) and is most commonly used in animal
models of ALI induced by sepsis (36,37). The present study demonstrated
that quercetin alleviated LPS-induced ALI by inhibiting ferroptosis
in vivo and in vitro.
Ferroptosis is a lipid peroxidation, iron-dependent
cell death (4). During ALI,
alveolar epithelial cells and alveolar macrophages exhibit
significant ferroptosis, which accelerates the progression of the
disease (38). GSH is an
important antioxidant in cells, which is negatively associated with
ferroptosis (39). By contrast,
MDA is the end product of lipid oxidation during ferroptosis, which
is positively associated with ferroptosis (39). Mitochondria are important
organelles of oxidative metabolism and play a crucial role in
ferroptosis (39). Abnormal
mitochondrial metabolism significantly leads to rapid consumption
of glutathione and subsequent lipid ROS production and ferroptosis
(39). In the present study,
changes in the aforementioned mediators were detected, thereby
confirming that ferroptosis participates in LPS-induced ALI.
Quercetin has been widely studied because of its
strong anti-inflammatory and antioxidant properties, including
inhibiting the production of inflammatory factors, tuning immune
cell infiltration and suppressing oxidative stress) (17-24). However, the role of quercetin in
ALI has not yet been fully investigated. In the current study,
quercetin significantly attenuated LPS-induced lung edema, tissue
damage and inflammation. Quercetin effectively alleviated
LPS-induced alveolar epithelial cell injury in vitro and
LPS-induced ALI in vivo. Considering that ferroptosis is
induced by lipid peroxidation (40) and is involved in the pathogenesis
of ALI (8,9,29), we hypothesized that quercetin
could inhibit ferroptosis and alleviate LPS-induced ALI. Therefore,
characteristics of ferroptosis were examined through various assays
to evaluate the effect of quercetin on LPS-induced ferroptosis. The
results indicated that quercetin decreased LPS-induced ROS
generation, GSH depletion and MDA formation both in vivo and
in vitro.
It has been previously shown that LPS can induce
alveolar epithelial cells and capillary endothelial cells to
release a large number of inflammatory mediators and chemokines,
including IL-1β, IL-6 and TNF-α, resulting in neutrophil
infiltration and increased inflammation (2). Uncontrolled inflammation
subsequently leads to diffuse alveolar capillary basement membrane
damage, increased permeability of the pulmonary capillary
endothelium and alveolar epithelial cell barrier and edema in the
alveolar and pulmonary interstitium, which result in ALI (41). Tight junctions, which are
distributed between epithelial and endothelial cells throughout the
body, create a regulated paracellular channel for the transport of
water, solutes and immune cells, and are also a key component of
the alveolar epithelial barrier (42). Tight junction proteins are the
main components of the intercellular junctions that play a critical
role in epithelial cells. Proteins ZO-1 and occludin are
responsible for maintaining the integrity of tight junctions and
alveolar epithelial barrier function (43,44). Thus, the expression levels of
these two proteins were evaluated and it was demonstrated that
quercetin, as a ferroptosis inhibitor, increased the levels of
these proteins in alveolar epithelial cells following exposure to
LPS. However, the present study also had certain limitations.
Firstly, the spontaneous sepsis model (induced by cecal ligation
puncture) (24) and the
exogenous endotoxin (LPS) model are commonly used in the study of
sepsis-induced ALI; however, only the LPS model was used in the
current study, and the results may need to be verified in another
in vivo experimental model of ALI. Secondly, there may be
other mechanisms via which quercetin regulates ferroptosis, which
need to be explored further in future studies. Lastly, the
occurrence of ferroptosis was not verified in clinical ALI
samples.
In conclusion, the present study demonstrated that
quercetin administration exerted significant protective effects
against LPS-induced ALI in both in vitro and in vivo
models. The mechanisms underlying these effects in alveolar
epithelial cells may involve ferroptosis inhibition via the
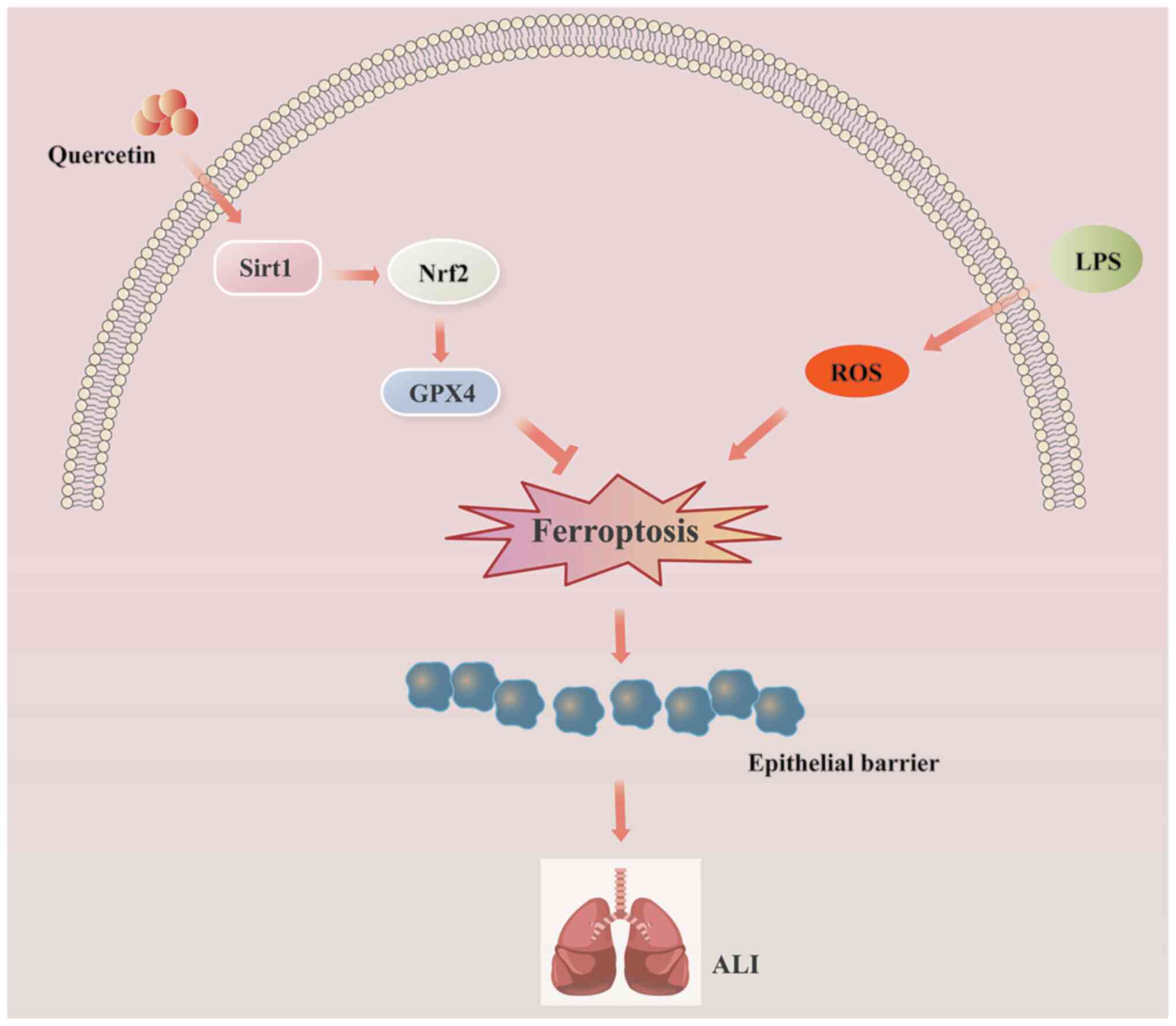
Sirt1/Nrf2/Gpx4 signaling pathway (Fig. 8). The present findings identified
a novel role for quercetin as a potential therapy for ALI
prevention.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SD, JL and LL conducted the majority of the
experiments, performed the statistical analysis, and wrote,
reviewed and edited the original manuscript. SD and SL revised the
manuscript. SL, YY, TL, TZ and GX provided research materials and
developed the methodology. YX and DW conceived and designed the
study, and wrote, reviewed and edited the original manuscript. SD
and YX confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
All animal experiments were approved by the
Laboratory Animal Ethics Committee of Chengdu Medical College
(Chengdu, China; approval no. 2021-085).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Abbreviations:
|
ALI
|
acute lung injury
|
|
AT2
|
type II alveolar epithelial
|
|
BALF
|
bronchoalveolar lavage fluid
|
|
Gpx4
|
glutathione peroxidase 4
|
|
GSH
|
glutathione
|
|
H&E
|
hematoxylin and eosin
|
|
4-HNE
|
4-hydroxynonenal
|
|
IF
|
immunofluorescence
|
|
IHC
|
immunohistochemistry
|
|
LDH
|
lactate dehydrogenase
|
|
LPS
|
lipopolysaccharide
|
|
MDA
|
malondialdehyde
|
|
MPO
|
myeloperoxidase
|
|
Nrf2
|
nuclear factor erythroid 2-related
factor 2
|
|
ROS
|
reactive oxygen species
|
|
Sirt1
|
sirtuin 1
|
|
TEM
|
transmission electron microscopy
|
|
W/D
|
wet/dry
|
|
ZO-1
|
zonula occludens-1
|
Acknowledgments
Not applicable.
Funding
The present study was funded by the National Natural Science
Foundation of China (grant nos. 81972977, 82273574 and 82273433),
Foundation of Health Commission of Sichuan Province (grant no.
20ZD016), Foundation of Health Commission of Chengdu (grant no.
2021001), Foundation of Chengdu Science and Technology Bureau
(grant no. 2021-YF05-00291-SN), Foundation of Sichuan Science and
Technology Agency (grant no. 2019YJ0589), Foundation of the First
Affiliated Hospital of Chengdu Medical College (grant nos.
CYFY202LNZD01, CYFY2020YB05 and CYFY2019ZD06), Disciplinary
Construction Innovation Team Foundation of Chengdu Medical College
(grant no. CMC-XK-2103), Foundation of Chengdu Municipal Health
Commission (grant no. 2022333), Graduate Innovation Fund Project of
Chengdu Medical College (grant nos. YCX2022-03-01 and
YCX2022-03-02), Foundation of Sichuan Medical and Health Promotion
Association (grant no. KY2022QN0313) and Foundation of Chengdu
Medical College (grant no. CYZYB20-09).
References
|
1
|
Xia L, Zhang C, Lv N, Liang Z, Ma T, Cheng
H, Xia Y and Shi L: AdMSC-derived exosomes alleviate acute lung
injury via transferring mitochondrial component to improve
homeostasis of alveolar macrophages. Theranostics. 12:2928–2947.
2022.
|
|
2
|
Ware LB and Matthay MA: The acute
respiratory distress syndrome. N Engl J Med. 342:1334–1349.
2000.
|
|
3
|
Vandewalle J, Luypaert A, De Bosscher K
and Libert C: Therapeutic mechanisms of glucocorticoids. Trends
Endocrinol Metab. 29:42–54. 2018.
|
|
4
|
Guo W, Wu Z, Chen J, Guo S, You W, Wang S,
Ma J, Wang H, Wang X, Wang H, et al: Nanoparticle delivery of
miR-21-3p sensitizes melanoma to anti-PD-1 immunotherapy by
promoting ferroptosis. J Immunother Cancer. 10:e0043812022.
|
|
5
|
Gupta U, Ghosh S, Wallace CT, Shang P, Xin
Y, Nair AP, Yazdankhah M, Strizhakova A, Ross MA, Liu H, et al:
Increased LCN2 (lipocalin 2) in the RPE decreases autophagy and
activates inflammasome-ferroptosis processes in a mouse model of
dry AMD. Autophagy. 19:92–111. 2023.
|
|
6
|
Wang Y, Chen D, Xie H, Jia M, Sun X, Peng
F, Guo F and Tang D: AUF1 protects against ferroptosis to alleviate
sepsis-induced acute lung injury by regulating NRF2 and ATF3. Cell
Mol Life Sci. 79:2282022.
|
|
7
|
Yang Y, Ma Y, Li Q, Ling Y, Zhou Y, Chu K,
Xue L and Tao S: STAT6 inhibits ferroptosis and alleviates acute
lung injury via regulating P53/SLC7A11 pathway. Cell Death Dis.
13:5302022.
|
|
8
|
Wang Y, Yuan Y, Wang W, He Y, Zhong H,
Zhou X, Chen Y, Cai XJ and Liu LQ: Mechanisms underlying the
therapeutic effects of Qingfeiyin in treating acute lung injury
based on GEO datasets, network pharmacology and molecular docking.
Comput Biol Med. 145:1054542022.
|
|
9
|
Liu P, Feng Y, Li H, Chen X, Wang G, Xu S,
Li Y and Zhao L: Ferrostatin-1 alleviates
lipopolysaccharide-induced acute lung injury via inhibiting
ferroptosis. Cell Mol Biol Lett. 25:102020.
|
|
10
|
Kerins MJ and Ooi A: The roles of NRF2 in
modulating cellular iron homeostasis. Antioxid Redox Signal.
29:1756–1773. 2018.
|
|
11
|
Dodson M, Castro-Portuguez R and Zhang DD:
NRF2 plays a critical role in mitigating lipid peroxidation and
ferroptosis. Redox Biol. 23:1011072019.
|
|
12
|
Li Y, Cao Y, Xiao J, Shang J, Tan Q, Ping
F, Huang W, Wu F, Zhang H and Zhang X: Inhibitor of
apoptosis-stimulating protein of p53 inhibits ferroptosis and
alleviates intestinal ischemia/reperfusion-induced acute lung
injury. Cell Death Differ. 27:2635–2650. 2020.
|
|
13
|
Hu J, Gu W, Ma N, Fan X and Ci X:
Leonurine alleviates ferroptosis in cisplatin-induced acute kidney
injury by activating the Nrf2 signalling pathway. Br J Pharmacol.
179:3991–4009. 2022.
|
|
14
|
Qiongyue Z, Xin Y, Meng P, Sulin M, Yanlin
W, Xinyi L and Xuemin S: Post-treatment with irisin attenuates
acute kidney injury in sepsis mice through anti-ferroptosis via the
SIRT1/Nrf2 pathway. Front Pharmacol. 13:8570672022.
|
|
15
|
Wang C, Liu T, Tong Y, Cui R, Qu K, Liu C
and Zhang J: Ulinastatin protects against acetaminophen-induced
liver injury by alleviating ferroptosis via the SIRT1/NRF2/HO-1
pathway. Am J Transl Res. 13:6031–6042. 2021.
|
|
16
|
Geng F, Xu M, Zhao L, Zhang H, Li J, Jin
F, Li Y, Li T, Yang X, Li S, et al: Quercetin alleviates pulmonary
fibrosis in mice exposed to silica by inhibiting macrophage
senescence. Front Pharmacol. 13:9120292022.
|
|
17
|
Li J, Sun Z, Luo G, Wang S, Cui H, Yao Z,
Xiong H, He Y, Qian Y and Fan C: Quercetin attenuates
trauma-induced heterotopic ossification by tuning immune cell
infiltration and related inflammatory insult. Front Immunol.
12:6492852021.
|
|
18
|
Farag MR, Moselhy AAA, El-Mleeh A,
Aljuaydi SH, Ismail TA, Di Cerbo A, Crescenzo G and Abou-Zeid SM:
Quercetin alleviates the immunotoxic impact mediated by oxidative
stress and inflammation induced by doxorubicin exposure in rats.
Antioxidants (Basel). 10:19062021.
|
|
19
|
Li D, Jiang C, Mei G, Zhao Y, Chen L, Liu
J, Tang Y, Gao C and Yao P: Quercetin alleviates ferroptosis of
pancreatic β cells in type 2 diabetes. Nutrients. 12:29542020.
|
|
20
|
Wang Y, Quan F, Cao Q, Lin Y, Yue C, Bi R,
Cui X, Yang H, Yang Y, Birnbaumer L, et al: Quercetin alleviates
acute kidney injury by inhibiting ferroptosis. J Adv Res.
28:231–243. 2020.
|
|
21
|
Huang T, Zhang K, Wang J, He K, Zhou X and
Nie S: Quercetin alleviates acrylamide-induced liver injury by
inhibiting autophagy-dependent ferroptosis. J Agric Food Chem.
71:7427–7439. 2023.
|
|
22
|
Huang R, Zhong T and Wu H: Quercetin
protects against lipopolysaccharide-induced acute lung injury in
rats through suppression of inflammation and oxidative stress. Arch
Med Sci. 11:427–432. 2015.
|
|
23
|
da Silva Araújo NP, de Matos NA, Leticia
Antunes Mota S, Farias de Souza AB, Dantas Cangussú S, Cunha Alvim
de Menezes R and Silva Bezerra F: Quercetin attenuates acute lung
injury caused by cigarette smoke both in vitro and in vivo. COPD.
17:205–214. 2020.
|
|
24
|
Sang A, Wang Y, Wang S, Wang Q, Wang X, Li
X and Song X: Quercetin attenuates sepsis-induced acute lung injury
via suppressing oxidative stress-mediated ER stress through
activation of SIRT1/AMPK pathways. Cell Signal. 96:1103632022.
|
|
25
|
Cui Z and Zhao X, Amevor FK, Du X, Wang Y,
Li D, Shu G, Tian Y and Zhao X: Therapeutic application of
quercetin in aging-related diseases: SIRT1 as a potential
mechanism. Front Immunol. 13:9433212022.
|
|
26
|
Deng S, Wu D, Li L, Li J and Xu Y: TBHQ
attenuates ferroptosis against 5-fluorouracil-induced intestinal
epithelial cell injury and intestinal mucositis via activation of
Nrf2. Cell Mol Biol Lett. 26:482021.
|
|
27
|
Li J, Deng SH, Li J, Li L, Zhang F, Zou Y,
Wu DM and Xu Y: Obacunone alleviates ferroptosis during
lipopolysaccharide-induced acute lung injury by upregulating
Nrf2-dependent antioxidant responses. Cell Mol Biol Lett.
27:292022.
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
|
|
29
|
Yang HH, Duan JX, Liu SK, Xiong JB, Guan
XX, Zhong WJ, Sun CC, Zhang CY, Luo XQ, Zhang YF, et al: A
COX-2/sEH dual inhibitor PTUPB alleviates
lipopolysaccharide-induced acute lung injury in mice by inhibiting
NLRP3 inflammasome activation. Theranostics. 10:4749–4761.
2020.
|
|
30
|
Yu Y, Wu DM, Li J, Deng SH, Liu T, Zhang
T, He M, Zhao YY and Xu Y: Bixin attenuates experimental autoimmune
encephalomyelitis by suppressing TXNIP/NLRP3 inflammasome activity
and activating NRF2 signaling. Front Immunol. 11:5933682020.
|
|
31
|
Zhang R, Guo N, Yan G, Wang Q, Gao T,
Zhang B and Hou N: Ginkgolide C attenuates
lipopolysaccharide-induced acute lung injury by inhibiting
inflammation via regulating the CD40/NF-κB signaling pathway. Int J
Mol Med. 47:622021.
|
|
32
|
Zhu Y, Wang K, Ma Z, Liu D, Yang Y, Sun M,
Wen A, Hao Y, Ma S, Ren F, et al: SIRT1 activation by butein
attenuates sepsis-induced brain injury in mice subjected to cecal
ligation and puncture via alleviating inflammatory and oxidative
stress. Toxicol Appl Pharmacol. 363:34–46. 2019.
|
|
33
|
Li JP, Wu KH, Chao WR, Lee YJ, Yang SF and
Chao YH: Immunomodulation of mesenchymal stem cells in acute lung
injury: From preclinical animal models to treatment of severe
COVID-19. Int J Mol Sci. 23:81962020.
|
|
34
|
Li J, Bai Y, Tang Y, Wang X, Cavagnaro MJ,
Li L, Li Z, Zhang Y and Shi J: A 4-benzene indol derivative
alleviates LPS-induced acute lung injury through inhibiting the
NLRP3 inflammasome. Front Immunol. 13:8121642022.
|
|
35
|
Zhang B, Liu ZY, Li YY, Luo Y, Liu ML,
Dong HY, Wang YX, Liu Y, Zhao PT, Jin FG and Li ZC:
Antiinflammatory effects of matrine in LPS-induced acute lung
injury in mice. Eur J Pharm Sci. 44:573–579. 2011.
|
|
36
|
Lv H, Liu Q, Wen Z, Feng H, Deng X and Ci
X: Xanthohumol ameliorates lipopolysaccharide (LPS)-induced acute
lung injury via induction of AMPK/GSK3β-Nrf2 signal axis. Redox
Biol. 12:311–324. 2017.
|
|
37
|
Fukatsu M, Ohkawara H, Wang X, Alkebsi L,
Furukawa M, Mori H, Fukami M, Fukami SI, Sano T, Takahashi H, et
al: The suppressive effects of Mer inhibition on inflammatory
responses in the pathogenesis of LPS-induced ALI/ARDS. Sci Signal.
15:eabd25332022.
|
|
38
|
Tang X, Liu J, Yao S, Zheng J, Gong X and
Xiao B: Ferulic acid alleviates alveolar epithelial barrier
dysfunction in sepsis-induced acute lung injury by activating the
Nrf2/HO-1 pathway and inhibiting ferroptosis. Pharm Biol.
60:2286–2294. 2022.
|
|
39
|
Zhang X, Hou L, Guo Z, Wang G, Xu J, Zheng
Z, Sun K and Guo F: Lipid peroxidation in osteoarthritis: Focusing
on 4-hydroxynonenal, malondialdehyde, and ferroptosis. Cell Death
Discov. 9:3202023.
|
|
40
|
Liang D, Minikes AM and Jiang X:
Ferroptosis at the intersection of lipid metabolism and cellular
signaling. Mol Cell. 82:2215–2227. 2022.
|
|
41
|
Li Y, Huang X, Huang S, He H, Lei T,
Saaoud F, Yu XQ, Melnick A, Kumar A, Papasian CJ, et al: Central
role of myeloid MCPIP1 in protecting against LPS-induced
inflammation and lung injury. Signal Transduct Target Ther.
2:170662017.
|
|
42
|
Buckley A and Turner JR: Cell biology of
tight junction barrier regulation and mucosal disease. Cold Spring
Harb Perspect Biol. 10:a0293142018.
|
|
43
|
You K, Xu X, Fu J, Xu S, Yue X, Yu Z and
Xue X: Hyperoxia disrupts pulmonary epithelial barrier in newborn
rats via the deterioration of occludin and ZO-1. Respir Res.
13:362012.
|
|
44
|
Englert JA, Macias AA, Amador-Munoz D,
Pinilla Vera M, Isabelle C, Guan J, Magaoay B, Suarez Velandia M,
Coronata A, Lee A, et al: Isoflurane ameliorates acute lung injury
by preserving epithelial tight junction integrity. Anesthesiology.
123:377–388. 2015.
|