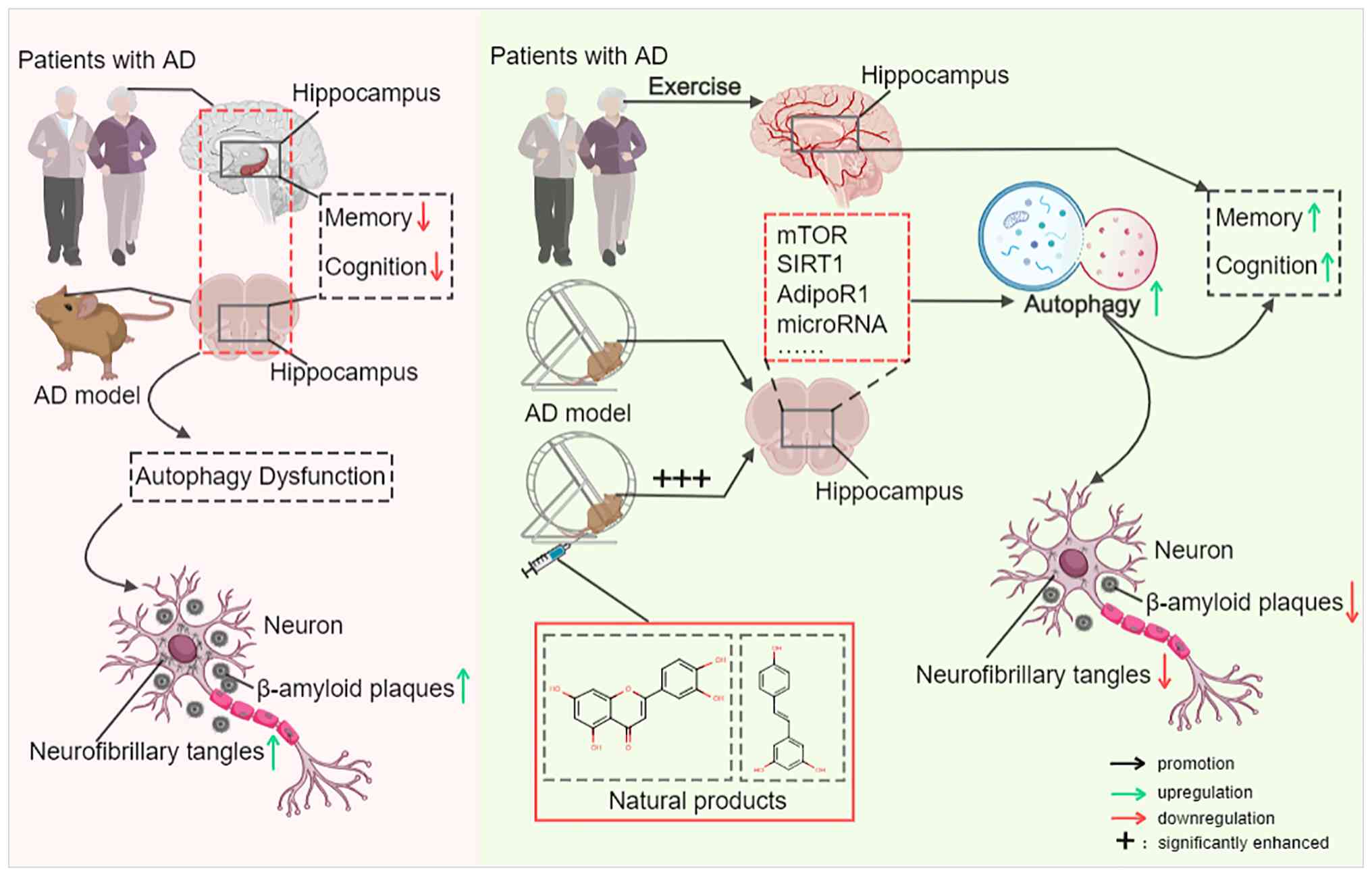
|
1
|
Scheltens P, De Strooper B, Kivipelto M,
Holstege H, Chételat G, Teunissen CE, Cummings J and van der Flier
WM: Alzheimer's disease. Lancet. 397:1577–1590. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Reitz C, Pericak-Vance MA, Foroud T and
Mayeux R: A global view of the genetic basis of Alzheimer disease.
Nat Rev Neurol. 19:261–277. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Di Meco A, Curtis ME, Lauretti E and
Praticò D: Autophagy dysfunction in Alzheimer's Disease:
Mechanistic insights and new therapeutic opportunities. Biol
Psychiatry. 87:797–807. 2020. View Article : Google Scholar
|
|
4
|
Hardy J and Selkoe DJ: The amyloid
hypothesis of Alzheimer's disease: Progress and problems on the
road to therapeutics. Science. 297:353–356. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
De Strooper B and Karran E: The cellular
phase of Alzheimer's disease. Cell. 164:603–615. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Iqbal K, Liu F and Gong CX: Tau and
neurodegenerative disease: The story so far. Nat Rev Neurol.
12:15–27. 2016. View Article : Google Scholar
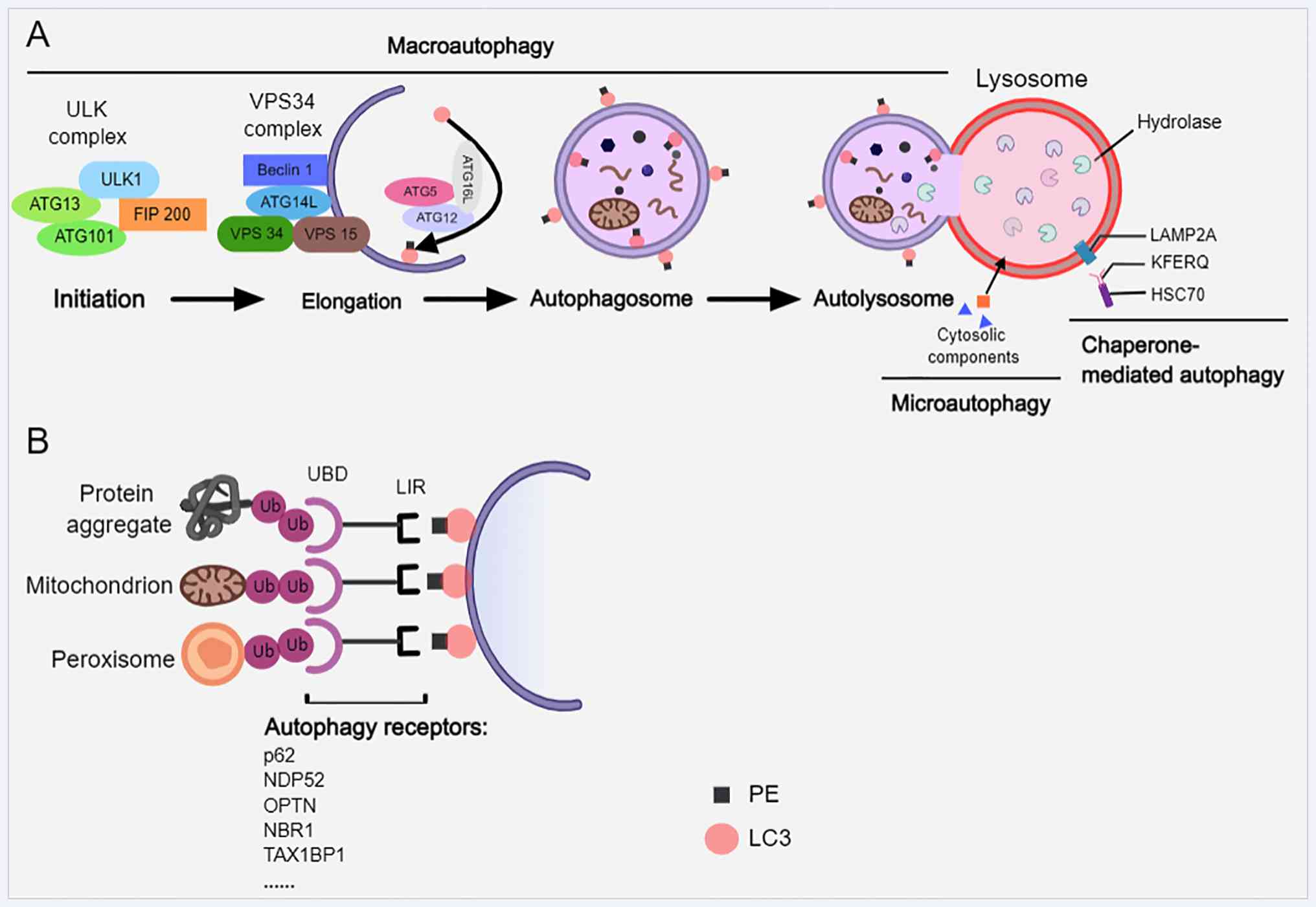
|
|
7
|
Tong BC, Wu AJ, Huang AS, Dong R,
Malampati S, Iyaswamy A, Krishnamoorthi S, Sreenivasmurthy SG, Zhu
Z, Su C, et al: Lysosomal TPCN (two pore segment channel)
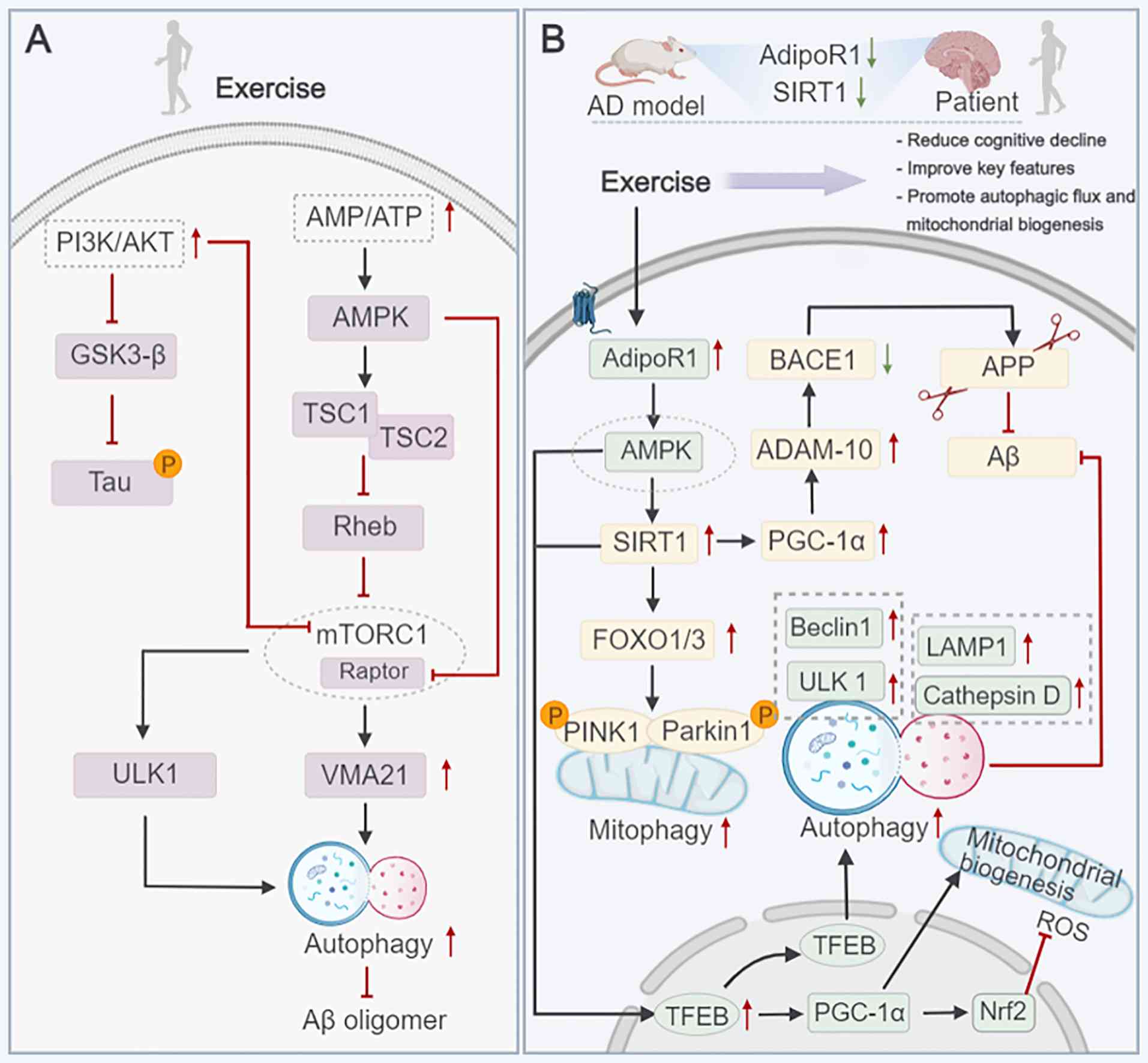
inhibition ameliorates beta-amyloid pathology and mitigates memory
impairment in Alzheimer disease. Autophagy. 18:624–642. 2022.
View Article : Google Scholar :
|
|
8
|
Nixon RA: Autophagy-lysosomal-associated
neuronal death in neurodegenerative disease. Acta Neuropathol.
148:422024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tzioras M, McGeachan RI, Durrant CS and
Spires-Jones TL: Synaptic degeneration in Alzheimer disease. Nat
Rev Neurol. 19:19–38. 2023. View Article : Google Scholar
|
|
10
|
Butterfield DA and Halliwell B: Oxidative
stress, dysfunctional glucose metabolism and Alzheimer disease. Nat
Rev Neurosci. 20:148–160. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Deng Z, Dong Y, Zhou X, Lu JH and Yue Z:
Pharmacological modulation of autophagy for Alzheimer's disease
therapy: Opportunities and obstacles. Acta Pharm Sin B.
12:1688–1706. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lee JH and Nixon RA: Autolysosomal
acidification failure as a primary driver of Alzheimer disease
pathogenesis. Autophagy. 18:2763–2764. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rubinsztein DC, Codogno P and Levine B:
Autophagy modulation as a potential therapeutic target for diverse
diseases. Nat Rev Drug Discov. 11:709–730. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
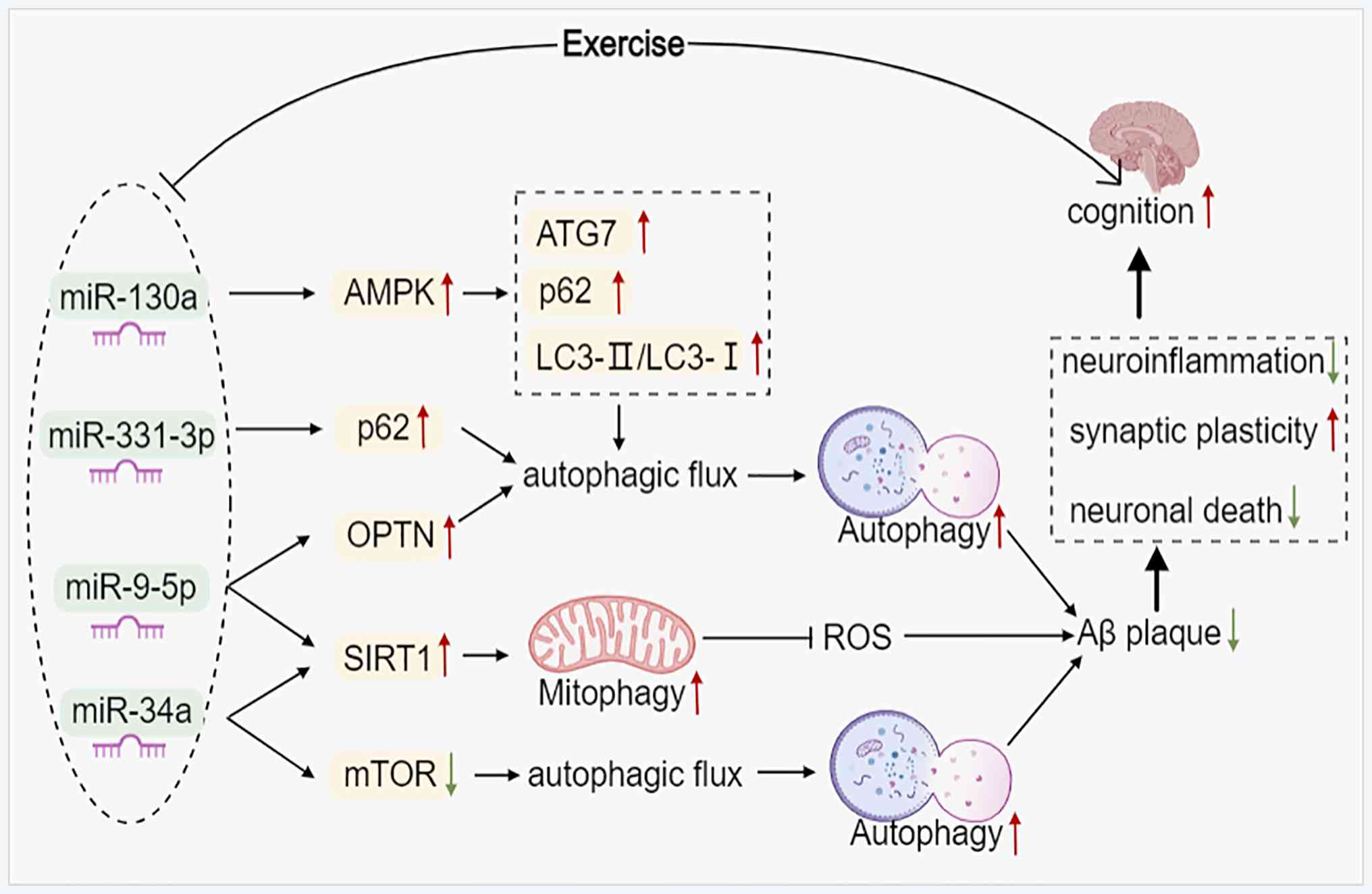
14
|
Levine B and Kroemer G: Biological
functions of autophagy genes: A disease perspective. Cell.
176:11–42. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yu L, Chen Y and Tooze SA: Autophagy
pathway: Cellular and molecular mechanisms. Autophagy. 14:207–215.
2018. View Article : Google Scholar :
|
|
16
|
Kaushik S, Tasset I, Arias E, Pampliega O,
Wong E, Martinez-Vicente M and Cuervo AM: Autophagy and the
hallmarks of aging. Ageing Res Rev. 72:1014682021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lee JH, Yang DS, Goulbourne CN, Im E,
Stavrides P, Pensalfini A, Chan H, Bouchet-Marquis C, Bleiwas C,
Berg MJ, et al: Faulty autolysosome acidification in Alzheimer's
disease mouse models induces autophagic build-up of Aβ in neurons,
yielding senile plaques. Nat Neurosci. 25:688–701. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
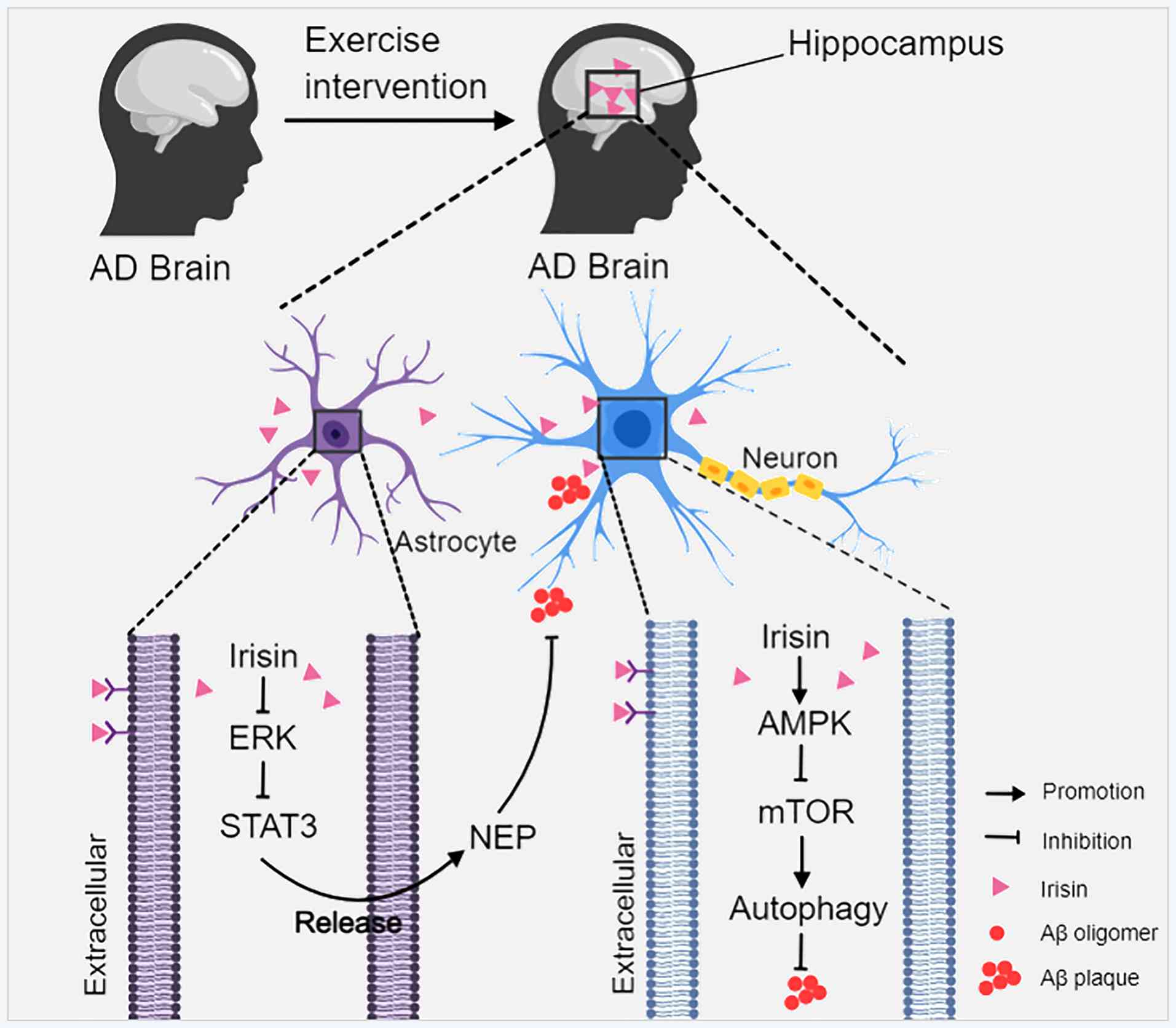
|
Uddin MS, Stachowiak A, Mamun AA, Tzvetkov
NT, Takeda S, Atanasov AG, Bergantin LB, Abdel-Daim MM and
Stankiewicz AM: Autophagy and Alzheimer's Disease: From molecular
mechanisms to therapeutic implications. Front Aging Neurosci.
10:042018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nixon RA: The role of autophagy in
neurodegenerative disease. Nat Med. 19:983–997. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Palmer JE, Wilson N, Son SM, Obrocki P,
Wrobel L, Rob M, Takla M, Korolchuk VI and Rubinsztein DC:
Autophagy, aging, and age-related neurodegeneration. Neuron.
113:29–48. 2025. View Article : Google Scholar
|
|
21
|
Fang EF, Hou Y, Palikaras K, Adriaanse BA,
Kerr JS, Yang B, Lautrup S, Hasan-Olive MM, Caponio D, Dan X, et
al: Mitophagy inhibits amyloid-β and tau pathology and reverses
cognitive deficits in models of Alzheimer's disease. Nat Neurosci.
22:401–412. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Menzies FM, Fleming A, Caricasole A, Bento
CF, Andrews SP, Ashkenazi A, Füllgrabe J, Jackson A, Jimenez
Sanchez M, Karabiyik C, et al: Autophagy and neurodegeneration:
Pathogenic mechanisms and therapeutic opportunities. Neuron.
93:1015–1034. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sharif K, Watad A, Bragazzi NL, Lichtbroun
M, Amital H and Shoenfeld Y: Physical activity and autoimmune
diseases: Get moving and manage the disease. Autoimmun Rev.
17:53–72. 2018. View Article : Google Scholar
|
|
24
|
Zhu C, Ma H, He A, Li Y, He C and Xia Y:
Exercise in cancer prevention and anticancer therapy: Efficacy,
molecular mechanisms and clinical information. Cancer Lett.
544:2158142022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Andersen LL: Health promotion and chronic
disease prevention at the workplace. Annu Rev Public Health.
45:337–357. 2024. View Article : Google Scholar
|
|
26
|
Halon-Golabek M, Borkowska A,
Herman-Antosiewicz A and Antosiewicz J: Iron Metabolism of the
skeletal muscle and neurodegeneration. Front Neurosci. 13:1652019.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
López-Ortiz S, Pinto-Fraga J, Valenzuela
PL, Martín-Hernández J, Seisdedos MM, García-López O, Toschi N, Di
Giuliano F, Garaci F, Mercuri NB, et al: Physical exercise and
Alzheimer's disease: Effects on pathophysiological molecular
pathways of the disease. Int J Mol Sci. 22:28972021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mahalakshmi B, Maurya N, Lee SD and
Bharath Kumar V: Possible neuroprotective mechanisms of physical
exercise in neurodegeneration. Int J Mol Sci. 21:58952020.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sujkowski A, Hong L, Wessells RJ and Todi
SV: The protective role of exercise against age-related
neurodegeneration. Ageing Res Rev. 74:1015432022. View Article : Google Scholar
|
|
30
|
De la Rosa A, Olaso-Gonzalez G,
Arc-Chagnaud C, Millan F, Salvador-Pascual A, García-Lucerga C,
Blasco-Lafarga C, Garcia-Dominguez E, Carretero A, Correas AG, et
al: Physical exercise in the prevention and treatment of
Alzheimer's disease. J Sport Health Sci. 9:394–404. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fox FAU, Diers K, Lee H, Mayr A, Reuter M,
Breteler MMB and Aziz NA: Association between accelerometer-derived
physical activity measurements and brain structure: A
Population-based cohort study. Neurology. 99:e1202–e1215. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yau WW, Kirn DR, Rabin JS, Properzi MJ,
Schultz AP, Shirzadi Z, Palmgren K, Matos P, Maa C, Pruzin JJ, et
al: Physical activity as a modifiable risk factor in preclinical
Alzheimer's disease. Nat Med. 31:4075–4083. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cotman CW, Berchtold NC and Christie LA:
Exercise builds brain health: Key roles of growth factor cascades
and inflammation. Trends Neurosci. 30:464–472. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kang J, Liu M, Yang Q, Dang X, Li Q, Wang
T, Qiu B, Zhang Y, Guo X, Li X, et al: Exercise training exerts
beneficial effects on Alzheimer's disease through multiple
signaling pathways. Front Aging Neurosci. 17:15580782025.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nixon RA and Rubinsztein DC: Mechanisms of
autophagy-lysosome dysfunction in neurodegenerative diseases. Nat
Rev Mol Cell Biol. 25:926–946. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mizushima N and Komatsu M: Autophagy:
Renovation of cells and tissues. Cell. 147:728–741. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kaur J and Debnath J: Autophagy at the
crossroads of catabolism and anabolism. Nat Rev Mol Cell Biol.
16:461–472. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nakatogawa H: Mechanisms governing
autophagosome biogenesis. Nat Rev Mol Cell Biol. 21:439–458. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nazio F and Cecconi F: Autophagy up and
down by outsmarting the incredible ULK. Autophagy. 13:967–968.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gui X, Yang H, Li T, Tan X, Shi P, Li M,
Du F and Chen ZJ: Autophagy induction via STING trafficking is a
primordial function of the cGAS pathway. Nature. 567:262–266. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li WW, Li J and Bao JK: Microautophagy:
Lesser-known self-eating. Cell Mol Life Sci. 69:1125–1136. 2012.
View Article : Google Scholar
|
|
42
|
Bourdenx M, Martín-Segura A, Scrivo A,
Rodriguez-Navarro JA, Kaushik S, Tasset I, Diaz A, Storm NJ, Xin Q,
Juste YR, et al: Chaperone-mediated autophagy prevents collapse of
the neuronal metastable proteome. Cell. 184:2696–2714.e25. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kerr JS, Adriaanse BA, Greig NH, Mattson
MP, Cader MZ, Bohr VA and Fang EF: Mitophagy and Alzheimer's
disease: Cellular and molecular mechanisms. Trends Neurosci.
40:151–166. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sulkshane P, Ram J, Thakur A, Reis N,
Kleifeld O and Glickman MH: Ubiquitination and receptor-mediated
mitophagy converge to eliminate oxidation-damaged mitochondria
during hypoxia. Redox Biol. 45:1020472021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang Z, Yang X, Song YQ and Tu J:
Autophagy in Alzheimer's disease pathogenesis: Therapeutic
potential and future perspectives. Ageing Res Rev. 72:1014642021.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Deng P, Fan T, Gao P, Peng Y, Li M, Li J,
Qin M, Hao R, Wang L, Li M, et al: SIRT5-mediated desuccinylation
of RAB7A protects against Cadmium-induced Alzheimer's disease-like
pathology by restoring autophagic flux. Adv Sci (Weinh).
11:e24020302024. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Xie ZS, Zhao JP, Wu LM, Chu S, Cui ZH, Sun
YR, Wang H, Ma HF, Ma DR, Wang P, et al: Hederagenin improves
Alzheimer's disease through PPARα/TFEB-mediated autophagy.
Phytomedicine. 112:1547112023. View Article : Google Scholar
|
|
48
|
Shafei MA, Harris M and Conway ME:
Divergent metabolic regulation of autophagy and mTORC1-early events
in Alzheimer's disease? Front Aging Neurosci. 9:1732017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang X and Jia J: Magnolol improves
Alzheimer's disease-like pathologies and cognitive decline by
promoting autophagy through activation of the AMPK/mTOR/ULK1
pathway. Biomed Pharmacother. 161:1144732023. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Zhang XW, Zhu XX, Tang DS and Lu JH:
Targeting autophagy in Alzheimer's disease: Animal models and
mechanisms. Zool Res. 44:1132–1145. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Salminen A, Kaarniranta K, Kauppinen A,
Ojala J, Haapasalo A, Soininen H and Hiltunen M: Impaired autophagy
and APP processing in Alzheimer's disease: The potential role of
Beclin 1 interactome. Prog Neurobiol. 106-107:33–54. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
O'Brien CE and Wyss-Coray T: Sorting
through the roles of beclin 1 in microglia and neurodegeneration. J
Neuroimmune Pharmacol. 9:285–292. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Longobardi A, Catania M, Geviti A, Salvi
E, Vecchi ER, Bellini S, Saraceno C, Nicsanu R, Squitti R, Binetti
G, et al: Autophagy markers are altered in Alzheimer's disease,
dementia with lewy bodies and frontotemporal dementia. Int J Mol
Sci. 25:11252024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Peña-Martinez C, Rickman AD and Heckmann
BL: Beyond autophagy: LC3-associated phagocytosis and endocytosis.
Sci Adv. 8:eabn17022022. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Heckmann BL, Teubner BJW, Tummers B,
Boada-Romero E, Harris L, Yang M, Guy CS, Zakharenko SS and Green
DR: LC3-Associated Endocytosis facilitates β-Amyloid clearance and
mitigates neurodegeneration in murine Alzheimer's disease. Cell.
178:536–551.e14. 2019. View Article : Google Scholar
|
|
56
|
Udayar V, Chen Y, Sidransky E and Jagasia
R: Lysosomal dysfunction in neurodegeneration: Emerging concepts
and methods. Trends Neurosci. 45:184–199. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Chae CW, Yoon JH, Lim JR, Park JY, Cho JH,
Jung YH, Choi GE, Lee HJ and Han HJ: TRIM16-mediated lysophagy
suppresses high-glucose-accumulated neuronal Aβ. Autophagy.
19:2752–2768. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Qian XH, Ding GY, Chen SY, Liu XL, Zhang M
and Tang HD: Blood cathepsins on the risk of Alzheimer's disease
and related pathological biomarkers: Results from observational
cohort and mendelian randomization study. J Prev Alzheimers Dis.
11:1834–1842. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Almeida MF, Bahr BA and Kinsey ST:
Endosomal-lysosomal dysfunction in metabolic diseases and
Alzheimer's disease. Int Rev Neurobiol. 154:303–324. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Mançano ASF, Pina JG, Froes BR and Sciani
JM: Autophagy-lysosomal pathway impairment and cathepsin
dysregulation in Alzheimer's disease. Front Mol Biosci.
11:14902752024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Cecarini V, Bonfili L, Gogoi O, Lawrence
S, Venanzi FM, Azevedo V, Mancha-Agresti P, Drumond MM, Rossi G,
Berardi S, et al: Neuroprotective effects of p62(SQSTM1)-engineered
lactic acid bacteria in Alzheimer's disease: A pre-clinical study.
Aging. 12:15995–16020. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Thal DR, Gawor K and Moonen S: Regulated
cell death and its role in Alzheimer's disease and amyotrophic
lateral sclerosis. Acta Neuropathol. 147:692024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zheng X, Lin W, Jiang Y, Lu K, Wei W, Huo
Q, Cui S, Yang X, Li M, Xu N, et al: Electroacupuncture ameliorates
beta-amyloid pathology and cognitive impairment in Alzheimer
disease via a novel mechanism involving activation of TFEB
(transcription factor EB). Autophagy. 17:3833–3847. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Settembre C, Fraldi A, Medina DL and
Ballabio A: Signals from the lysosome: A control centre for
cellular clearance and energy metabolism. Nat Rev Mol Cell Biol.
14:283–296. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Morais GP, de Sousa Neto IV, Marafon BB,
Ropelle ER, Cintra DE, Pauli JR and Silva A: The dual and emerging
role of physical exercise-induced TFEB activation in the protection
against Alzheimer's disease. J Cell Physiol. 238:954–965. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Nnah IC, Wang B, Saqcena C, Weber GF,
Bonder EM, Bagley D, De Cegli R, Napolitano G, Medina DL, Ballabio
A, et al: TFEB-driven endocytosis coordinates MTORC1 signaling and
autophagy. Autophagy. 15:151–164. 2019. View Article : Google Scholar :
|
|
67
|
Zhang J, Zhang Y, Wang J, Xia Y, Zhang J
and Chen L: Recent advances in Alzheimer's disease: Mechanisms,
clinical trials and new drug development strategies. Signal
Transduct Target Ther. 9:2112024. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Dewanjee S, Chakraborty P, Bhattacharya H,
Chacko L, Singh B, Chaudhary A, Javvaji K, Pradhan SR, Vallamkondu
J, Dey A, et al: Altered glucose metabolism in Alzheimer's disease:
Role of mitochondrial dysfunction and oxidative stress. Free Radic
Biol Med. 193:134–157. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Chen WT, Lu A, Craessaerts K, Pavie B,
Sala Frigerio C, Corthout N, Qian X, Laláková J, Kühnemund M,
Voytyuk I, et al: Spatial transcriptomics and in situ sequencing to
study Alzheimer's disease. Cell. 182:976–991.e19. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Martini-Stoica H, Xu Y, Ballabio A and
Zheng H: The Autophagy-lysosomal pathway in neurodegeneration: A
TFEB perspective. Trends Neurosci. 39:221–234. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Wu JJ, Yu H, Bi SG, Wang ZX, Gong J, Mao
YM, Wang FZ, Zhang YQ, Nie YJ and Chai GS: Aerobic exercise
attenuates autophagy-lysosomal flux deficits by ADRB2/β2-adrenergic
receptor-mediated V-ATPase assembly factor VMA21 signaling in
APP-PSEN1/PS1 mice. Autophagy. 20:1015–1031. 2024. View Article : Google Scholar :
|
|
72
|
Bi SG, Yu H, Gao TL, Wu JJ, Mao YM, Gong
J, Wang FZ, Yang L, Chen J, Lan ZC, et al: Aerobic exercise
attenuates Autophagy-lysosomal flux deficits via β2-AR-Mediated
ESCRT-III Subunit CHMP4B in mice with human MAPT P301L. Aging Cell.
24:e701842025. View Article : Google Scholar
|
|
73
|
Kou X, Chen D and Chen N: Physical
activity alleviates cognitive dysfunction of Alzheimer's disease
through regulating the mTOR signaling pathway. Int J Mol Sci.
20:15912019. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Li HY, Rong SS, Hong X, Guo R, Yang FZ,
Liang YY, Li A and So KF: Exercise and retinal health. Restor
Neurol Neurosci. 37:571–581. 2019.PubMed/NCBI
|
|
75
|
Ungvari Z, Fazekas-Pongor V, Csiszar A and
Kunutsor SK: The multifaceted benefits of walking for healthy
aging: From Blue Zones to molecular mechanisms. Geroscience.
45:3211–3239. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Cassilhas RC, Tufik S and de Mello MT:
Physical exercise, neuroplasticity, spatial learning and memory.
Cell Mol Life Sci. 73:975–983. 2016. View Article : Google Scholar
|
|
77
|
Rocchi A, Yamamoto S, Ting T, Fan Y,
Sadleir K, Wang Y, Zhang W, Huang S, Levine B, Vassar R and He C: A
Becn1 mutation mediates hyperactive autophagic sequestration of
amyloid oligomers and improved cognition in Alzheimer's disease.
PLoS Genet. 13:e10069622017. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Li X, He Q, Zhao N, Chen X, Li T and Cheng
B: High intensity interval training ameliorates cognitive
impairment in T2DM mice possibly by improving PI3K/Akt/mTOR
Signaling-regulated autophagy in the hippocampus. Brain Res.
1773:1477032021. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Chen J, Zhu T, Yang X, Yang Z, Shen M, Gu
B, Wang D, Zhang Y, Zhang M, Sun S, et al: Treadmill exercise
alleviates STING-mediated microglia pyroptosis and polarization via
activating mitophagy post-TBI. Free Radic Biol Med. 239:155–176.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Sun L, Wu L, Xu Z, Zeng W and Wang Y:
Running exercise alleviates depressive-like behaviors through the
activation of PINK1-Parkin mediated mitophagy in mice exposed to
chronic social defeat stress. Psychiatry Res. 352:1167142025.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Zhang Y, Liao B, Hu S, Pan SY, Wang GP,
Wang YL, Qin ZH and Luo L: High intensity interval training induces
dysregulation of mitochondrial respiratory complex and mitophagy in
the hippocampus of middle-aged mice. Behav Brain Res.
412:1133842021. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Tan ZX, Dong F, Wu LY, Feng YS and Zhang
F: The beneficial role of exercise on treating Alzheimer's disease
by Inhibiting β-amyloid peptide. Mol Neurobiol. 58:5890–5906. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Gratuze M, Julien J, Morin F, Marette A
and Planel E: Differential effects of voluntary treadmill exercise
and caloric restriction on tau pathogenesis in a mouse model of
Alzheimer's disease-like tau pathology fed with Western diet. Prog
Neuropsychopharmacol Biol Psychiatry. 79:452–461. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Xu L, Li M, Wei A, Yang M, Li C, Liu R,
Zheng Y, Chen Y, Wang Z, Wang K, et al: Treadmill exercise promotes
E3 ubiquitin ligase to remove amyloid β and P-tau and improve
cognitive ability in APP/PS1 transgenic mice. J Neuroinflammation.
19:2432022. View Article : Google Scholar
|
|
85
|
Morais GP, de Sousa Neto IV, Veras ASC,
Teixeira GR, Paroschi LO, Pinto AP, Dos Santos JR, Alberici LC,
Cintra DEC, Pauli JR, et al: Chronic exercise protects against
cognitive deficits in an Alzheimer's disease model by enhancing
autophagy and reducing mitochondrial abnormalities. Mol Neurobiol.
62:12791–12810. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Ohia-Nwoko O, Montazari S, Lau YS and
Eriksen JL: Long-term treadmill exercise attenuates tau pathology
in P301S tau transgenic mice. Mol Neurodegener. 9:542014.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Hussain MS, Agrawal N, Ilma B, M MR, Nayak
PP, Kaur M, Khachi A, Goyal K, Rekha A, Gupta S, et al: Autophagy
and cellular senescence in Alzheimer's disease: Key drivers of
neurodegeneration. CNS Neurosci Ther. 31:e705032025. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Saxton RA and Sabatini DM: mTOR signaling
in growth, metabolism, and disease. Cell. 168:960–976. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Kim J, Kundu M, Viollet B and Guan KL:
AMPK and mTOR regulate autophagy through direct phosphorylation of
Ulk1. Nat Cell Biol. 13:132–141. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Norwitz NG and Querfurth H: mTOR
Mysteries: Nuances and questions about the mechanistic target of
rapamycin in neurodegeneration. Front Neurosci. 14:7752020.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Gourmaud S, Stewart DA, Irwin DJ, Roberts
N, Barbour AJ, Eberwine G, O'Brien WT, Vassar R, Talos DM and
Jensen FE: The role of mTORC1 activation in seizure-induced
exacerbation of Alzheimer's disease. Brain. 145:324–339. 2022.
View Article : Google Scholar :
|
|
92
|
Babygirija R, Sonsalla MM, Mill J, James
I, Han JH, Green CL, Calubag MF, Wade G, Tobon A, Michael J, et al:
Protein restriction slows the development and progression of
pathology in a mouse model of Alzheimer's disease. Nat Commun.
15:52172024. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Watson K and Baar K: mTOR and the health
benefits of exercise. Semin Cell Dev Biol. 36:130–139. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Kang EB and Cho JY: Effect of treadmill
exercise on PI3K/AKT/mTOR, autophagy, and Tau hyperphosphorylation
in the cerebral cortex of NSE/htau23 transgenic mice. J Exerc
Nutrition Biochem. 19:199–209. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Deneubourg C, Ramm M, Smith LJ, Baron O,
Singh K, Byrne SC, Duchen MR, Gautel M, Eskelinen EL, Fanto M, et
al: The spectrum of neurodevelopmental, neuromuscular and
neurodegenerative disorders due to defective autophagy. Autophagy.
18:496–517. 2022. View Article : Google Scholar :
|
|
96
|
Shen K, Liu X, Chen D, Chang J, Zhang Y
and Kou X: Voluntary wheel-running exercise attenuates brain aging
of rats through activating miR-130a-mediated autophagy. Brain Res
Bull. 172:203–211. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wątroba M and Szukiewicz D: The role of
sirtuins in aging and age-related diseases. Adv Med Sci. 61:52–62.
2016. View Article : Google Scholar
|
|
98
|
Wang J, Zhou F, Xiong CE, Wang GP, Chen
LW, Zhang YT, Qi SG, Wang ZH, Mei C, Xu YJ, et al: Serum sirtuin1:
A potential blood biomarker for early diagnosis of Alzheimer's
disease. Aging (Albany NY). 15:9464–9478. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Liu L, Dai WZ, Zhu XC and Ma T: A review
of autophagy mechanism of statins in the potential therapy of
Alzheimer's disease. J Integr Neurosci. 21:462022. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Surya K, Manickam N, Jayachandran KS,
Kandasamy M and Anusuyadevi M: Resveratrol mediated regulation of
hippocampal neuroregenerative plasticity via SIRT1 pathway in
synergy with wnt signaling: Neurotherapeutic implications to
mitigate memory loss in Alzheimer's disease. J Alzheimers Dis.
94(Suppl): S125–S140. 2023. View Article : Google Scholar :
|
|
101
|
Mehramiz M, Porter T, O'Brien EK,
Rainey-Smith SR and Laws SM: A potential role for Sirtuin-1 in
Alzheimer's disease: Reviewing the biological and environmental
evidence. J Alzheimers Dis Rep. 7:823–843. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Zhao N, Xia J and Xu B: Physical exercise
may exert its therapeutic influence on Alzheimer's disease through
the reversal of mitochondrial dysfunction via
SIRT1-FOXO1/3-PINK1-Parkin-mediated mitophagy. J Sport Health Sci.
10:1–3. 2021. View Article : Google Scholar :
|
|
103
|
Koo JH, Kang EB, Oh YS, Yang DS and Cho
JY: Treadmill exercise decreases amyloid-β burden possibly via
activation of SIRT-1 signaling in a mouse model of Alzheimer's
disease. Exp Neurol. 288:142–152. 2017. View Article : Google Scholar
|
|
104
|
Han R, Liu Y, Li S, Li XJ and Yang W:
PINK1-PRKN mediated mitophagy: Differences between in vitro and in
vivo models. Autophagy. 19:1396–1405. 2023. View Article : Google Scholar :
|
|
105
|
Pallanck LJ: Culling sick mitochondria
from the herd. J Cell Biol. 191:1225–1227. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Zhao N, Zhang X, Li B, Wang J, Zhang C and
Xu B: Treadmill exercise improves PINK1/Parkin-Mediated mitophagy
activity against Alzheimer's disease pathologies by upregulated
SIRT1-FOXO1/3 Axis in APP/PS1 mice. Mol Neurobiol. 60:277–291.
2023. View Article : Google Scholar
|
|
107
|
Huang J, Wang X, Zhu Y, Li Z, Zhu YT, Wu
JC, Qin ZH, Xiang M and Lin F: Exercise activates lysosomal
function in the brain through AMPK-SIRT1-TFEB pathway. CNS Neurosci
Ther. 25:796–807. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Tyagi A and Pugazhenthi S: A Promising
strategy to treat neurodegenerative diseases by SIRT3 Activation.
Int J Mol Sci. 24:16152023. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Khoramipour K, Chamari K, Hekmatikar AA,
Ziyaiyan A, Taherkhani S, Elguindy NM and Bragazzi NL: Adiponectin:
Structure, physiological functions, role in diseases, and effects
of nutrition. Nutrients. 13:11802021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Bloemer J, Pinky PD, Govindarajulu M, Hong
H, Judd R, Amin RH, Moore T, Dhanasekaran M, Reed MN and
Suppiramaniam V: Role of adiponectin in central nervous system
disorders. Neural Plast. 2018:45935302018. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Rehman IU, Park JS, Choe K, Park HY, Park
TJ and Kim MO: Overview of a novel osmotin abolishes abnormal
metabolic-associated adiponectin mechanism in Alzheimer's disease:
Peripheral and CNS insights. Ageing Res Rev. 100:1024472024.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Ali T, Rehman SU, Khan A, Badshah H, Abid
NB, Kim MW, Jo MH, Chung SS, Lee HG, Rutten BPF and Kim MO:
Adiponectin-mimetic novel nonapeptide rescues aberrant neuronal
metabolic-associated memory deficits in Alzheimer's disease. Mol
Neurodegener. 16:232021. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Liu B, Liu J, Wang JG, Liu CL and Yan HJ:
AdipoRon improves cognitive dysfunction of Alzheimer's disease and
rescues impaired neural stem cell proliferation through
AdipoR1/AMPK pathway. Exp Neurol. 327:1132492020. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Sun F, Wang J, Meng L, Zhou Z, Xu Y, Yang
M, Li Y, Jiang T, Liu B and Yan H: AdipoRon promotes amyloid-β
clearance through enhancing autophagy via nuclear GAPDH-induced
sirtuin 1 activation in Alzheimer's disease. Br J Pharmacol.
181:3039–3063. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Jian Y, Yuan S, Yang J, Lei Y, Li X and
Liu W: Aerobic exercise alleviates abnormal autophagy in brain
cells of APP/PS1 mice by upregulating AdipoR1 levels. Int J Mol
Sci. 23:99212022. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Lu TX and Rothenberg ME: MicroRNA. J
Allergy Clin Immunol. 141:1202–1207. 2018. View Article : Google Scholar
|
|
117
|
Kou X and Chen N: Resveratrol as a natural
autophagy regulator for prevention and treatment of Alzheimer's
disease. Nutrients. 9:9272017. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Qi W, Ying Y, Wu P, Dong N, Fu W, Liu Q,
Ward N, Dong X, Zhao RC and Wang J: Inhibition of miR-4763-3p
expression activates the PI3K/mTOR/Bcl2 autophagy signaling pathway
to ameliorate cognitive decline. Int J Biol Sci. 20:5999–6017.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Madadi S, Schwarzenbach H, Saidijam M,
Mahjub R and Soleimani M: Potential microRNA-related targets in
clearance pathways of amyloid-β: Novel therapeutic approach for the
treatment of Alzheimer's disease. Cell Biosci. 9:912019. View Article : Google Scholar
|
|
120
|
Kou X, Chen D and Chen N: The regulation
of microRNAs in Alzheimer's sisease. Front Neurol. 11:2882020.
View Article : Google Scholar
|
|
121
|
Zhang H, Liang J and Chen N: The potential
role of miRNA-Regulated autophagy in Alzheimer's disease. Int J Mol
Sci. 23:77892022. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Kou X, Li J, Liu X, Chang J, Zhao Q, Jia
S, Fan J and Chen N: Swimming attenuates d-galactose-induced brain
aging via suppressing miR-34a-mediated autophagy impairment and
abnormal mitochondrial dynamics. J Appl Physiol (1985).
122:1462–1469. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Chen P, Chen F, Lei J, Li Q and Zhou B:
Activation of the miR-34a-Mediated SIRT1/mTOR signaling pathway by
urolithin a attenuates D-Galactose-induced brain aging in mice.
Neurotherapeutics. 16:1269–1282. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Chen ML, Hong CG, Yue T, Li HM, Duan R, Hu
WB, Cao J, Wang ZX, Chen CY, Hu XK, et al: Inhibition of miR-331-3p
and miR-9-5p ameliorates Alzheimer's disease by enhancing
autophagy. Theranostics. 11:2395–2409. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Wang Y, Sun X, He B and Yu S: Ginsenoside
Rg1 downregulates miR-9-5p expression to modulate SIRT1-Mediated
mitochondrial dysfunction and Ameliorate Alzheimer's disease. Mol
Neurobiol. 62:13044–13059. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Boström P, Wu J, Jedrychowski MP, Korde A,
Ye L, Lo JC, Rasbach KA, Boström EA, Choi JH, Long JZ, et al: A
PGC1-α-dependent myokine that drives brown-fat-like development of
white fat and thermogenesis. Nature. 481:463–468. 2012. View Article : Google Scholar
|
|
127
|
Lourenco MV, Frozza RL, de Freitas GB,
Zhang H, Kincheski GC, Ribeiro FC, Gonçalves RA, Clarke JR, Beckman
D, Staniszewski A, et al: Exercise-linked FNDC5/irisin rescues
synaptic plasticity and memory defects in Alzheimer's models. Nat
Med. 25:165–175. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Zhang H, Wu X, Liang J, Kirberger M and
Chen N: Irisin, an exercise-induced bioactive peptide beneficial
for health promotion during aging process. Ageing Res Rev.
80:1016802022. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
de Freitas GB, Lourenco MV and De Felice
FG: Protective actions of exercise-related FNDC5/Irisin in memory
and Alzheimer's disease. J Neurochem. 155:602–611. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Ratne N, Jari S, Tadas M, Katariya R, Kale
M, Kotagale N, Madia D, Umekar M and Taksande B: Neurobiological
role and therapeutic potential of exercise-induced irisin in
Alzheimer's disease management. Ageing Res Rev. 105:1026872025.
View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Islam MR, Valaris S, Young MF, Haley EB,
Luo R, Bond SF, Mazuera S, Kitchen RR, Caldarone BJ, Bettio LEB, et
al: Exercise hormone irisin is a critical regulator of cognitive
function. Nat Metab. 3:1058–1070. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Lourenco MV: Irisin limits amyloid-β
buildup in Alzheimer's disease. Trends Endocrinol Metab. 35:94–96.
2024. View Article : Google Scholar
|
|
133
|
Kim E, Kim H, Jedrychowski MP, Bakiasi G,
Park J, Kruskop J, Choi Y, Kwak SS, Quinti L, Kim DY, et al: Irisin
reduces amyloid-β by inducing the release of neprilysin from
astrocytes following downregulation of ERK-STAT3 signaling. Neuron.
111:3619–3633.e8. 2023. View Article : Google Scholar
|
|
134
|
Bellettini-Santos T, Batista-Silva H,
Marcolongo-Pereira C, Quintela-Castro FCA, Barcelos RM, Chiepe K,
Rossoni JV Jr, Passamani-Ambrosio R, da Silva BS, Chiarelli-Neto O,
et al: Move your body toward healthy aging: Potential
neuroprotective mechanisms of irisin in Alzheimer's disease. Int J
Mol Sci. 24:124402023. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Bujak AL, Crane JD, Lally JS, Ford RJ,
Kang SJ, Rebalka IA, Green AE, Kemp BE, Hawke TJ, Schertzer JD and
Steinberg GR: AMPK activation of muscle autophagy prevents
fasting-induced hypoglycemia and myopathy during aging. Cell Metab.
21:883–890. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Sánchez B, Muñoz-Pinto MF and Cano M:
Irisin enhances longevity by boosting SIRT1, AMPK, autophagy and
telomerase. Expert Rev Mol Med. 25:e42022. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Rahman MM, Islam MR and Emran TB:
Clinically important natural products for Alzheimer's disease. Int
J Surg. 104:1068072022. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
He Z, Zhang H, Li X, Tu S, Wang Z, Han S,
Du X, Shen L, Li N and Liu Q: The protective effects of
Esculentoside A through AMPK in the triple transgenic mouse model
of Alzheimer's disease. Phytomedicine. 109:1545552023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Fan Y, Wang N, Rocchi A, Zhang W, Vassar
R, Zhou Y and He C: Identification of natural products with
neuronal and metabolic benefits through autophagy induction.
Autophagy. 13:41–56. 2017. View Article : Google Scholar :
|
|
140
|
Thakral S, Yadav A, Singh V, Kumar M,
Kumar P, Narang R, Sudhakar K, Verma A, Khalilullah H, Jaremko M
and Emwas AH: Alzheimer's disease: Molecular aspects and treatment
opportunities using herbal drugs. Ageing Res Rev. 88:1019602023.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Agah S, Akbari A, Sadeghi E, Morvaridzadeh
M, Basharat Z, Palmowski A and Heshmati J: Resveratrol
supplementation and acute pancreatitis: A comprehensive review.
Biomed Pharmacother. 137:1112682021. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Li C, Wang N, Zheng G and Yang L: Oral
administration of Resveratrol-Selenium-Peptide nanocomposites
alleviates Alzheimer's disease-like pathogenesis by inhibiting Aβ
aggregation and regulating gut microbiota. ACS Appl Mater
Interfaces. 13:46406–46420. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Islam F, Nafady MH, Islam MR, Saha S,
Rashid S, Akter A, Or-Rashid MH, Akhtar MF, Perveen A, Md Ashraf G,
et al: Resveratrol and neuroprotection: An insight into prospective
therapeutic approaches against Alzheimer's disease from bench to
bedside. Mol Neurobiol. 59:4384–4404. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Chen JY, Zhu Q, Zhang S, OuYang D and Lu
JH: Resveratrol in experimental Alzheimer's disease models: A
systematic review of preclinical studies. Pharmacol Res.
150:1044762019. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Rahman MH, Akter R, Bhattacharya T,
Abdel-Daim MM, Alkahtani S, Arafah MW, Al-Johani NS, Alhoshani NM,
Alkeraishan N, Alhenaky A, et al: Resveratrol and neuroprotection:
Impact and its therapeutic potential in Alzheimer's disease. Front
Pharmacol. 11:6190242020. View Article : Google Scholar
|
|
146
|
Turner RS, Thomas RG, Craft S, van Dyck
CH, Mintzer J, Reynolds BA, Brewer JB, Rissman RA, Raman R and
Aisen PS: A randomized, double-blind, placebo-controlled trial of
resveratrol for Alzheimer disease. Neurology. 85:1383–1391. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Broderick TL, Rasool S, Li R, Zhang Y,
Anderson M, Al-Nakkash L, Plochocki JH, Geetha T and Babu JR:
Neuroprotective effects of chronic resveratrol treatment and
exercise training in the 3xTg-AD mouse model of Alzheimer's
disease. Int J Mol Sci. 21:73372020. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Kou JJ, Shi JZ, He YY, Hao JJ, Zhang HY,
Luo DM, Song JK, Yan Y, Xie XM, Du GH and Pang XB: Luteolin
alleviates cognitive impairment in Alzheimer's disease mouse model
via inhibiting endoplasmic reticulum stress-dependent
neuroinflammation. Acta Pharmacol Sin. 43:840–849. 2022. View Article : Google Scholar
|
|
149
|
Vongthip W, Nilkhet S, Boonruang K,
Sukprasansap M, Tencomnao T and Baek SJ: Neuroprotective mechanisms
of luteolin in glutamate-induced oxidative stress and
autophagy-mediated neuronal cell death. Sci Rep. 14:77072024.
View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Ali F and Siddique YH: Bioavailability and
Pharmaco-therapeutic potential of luteolin in overcoming
Alzheimer's disease. CNS Neurol Disord Drug Targets. 18:352–365.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
He Z, Li X, Wang Z, Cao Y, Han S, Li N,
Cai J, Cheng S and Liu Q: Protective effects of luteolin against
amyloid beta-induced oxidative stress and mitochondrial impairments
through peroxisome proliferator-activated receptor γ-dependent
mechanism in Alzheimer's disease. Redox Biol. 66:1028482023.
View Article : Google Scholar
|
|
152
|
Tao X, Zhang R, Wang L, Li X and Gong W:
Luteolin and exercise combination therapy ameliorates Amyloid-β1-42
Oligomers-Induced cognitive impairment in AD Mice by mediating
neuroinflammation and autophagy. J Alzheimers Dis. 92:195–208.
2023. View Article : Google Scholar
|
|
153
|
Tao X, Wang L and Gong W: Untargeted
metabolomics reveals the mechanisms of luteolin and exercise
combination treatment against cognitive impairments in AD mice
through modulating autophagy. J Nutr Biochem. 145:1100112025.
View Article : Google Scholar : PubMed/NCBI
|