Introduction
Follistatin-like protein 1 (FSTL1) is a 315-amino
acid secreted glycoprotein comprising an N-terminal signal peptide,
a follistatin-like domain and a calcium-binding EF-hand domain
(1,2). This structural configuration allows
FSTL1 to engage with multiple proteins, thereby participating in
intricate biological regulatory processes (3). FSTL1 is widely expressed in tissues
such as the heart, lungs, skeletal muscle and synovial membranes
(4-7). Under normal physiological
conditions, FSTL1 maintains stable expression levels, supporting
growth, development and cellular proliferation. However, its
expression is notably upregulated in inflammatory states (8,9),
and FSTL1 serves a role in inflammatory diseases, autoimmune
responses and tumour progression by regulating various signalling
pathways (10,11). Gene knockout models have
highlighted the essential function of FSTL1 in multi-system
development, with FSTL1-deficient mice exhibiting early mortality
due to developmental defects (12,13). At the cellular level, FSTL1
primarily operates through paracrine and autocrine mechanisms,
interacting with receptors such as activin and TGF-β receptors to
regulate cellular processes (1,14). The differential expression of
FSTL1 in different tissues is closely linked to its specific
biological functions (15,16). As a novel inflammatory regulator,
FSTL1 exhibits therapeutic potential in autoimmune diseases,
cardiovascular disorders and tissue regeneration (7,17,18). Further investigations into its
mechanisms of action may enhance understanding of the molecular
regulatory network underlying inflammation (19,20), identify new targets for
anti-inflammatory drug development (21), and improve diagnostic and
prognostic approaches for diseases such as inflammatory diseases
and fibrosis (4,22).
Role of FSTL1 in inflammatory
regulation
FSTL1 is an extracellular matrix glycoprotein that
serves a pivotal role in various inflammatory diseases. Research
has demonstrated that FSTL1 modulates inflammatory responses
through distinct signalling pathways (16,23), positioning it as a potential
future therapeutic target for certain inflammatory conditions.
Role of FSTL1 in cell proliferation,
differentiation and apoptosis
FSTL1 regulates cellular proliferation through
complex mechanisms that vary across different cell types. This
regulatory activity is particularly evident in tumour cell lines
(24,25). For example, in MDA-MB-231 breast
cancer cells, FSTL1 acts as a cell proliferation inhibitor, whereas
in gastric cancer cells, it promotes cellular proliferation
(26,27). This cell type-specific regulation
highlights the functional diversity of FSTL1.
Regulatory mechanisms of FSTL1 in
inflammatory responses
The role of FSTL1 in inflammatory responses is
multifaceted, involving complex and precisely coordinated
mechanisms (28,29). In terms of immune cell
regulation, FSTL1 modulates immune cell infiltration and
activation, maintaining immune system homeostasis by balancing
pro-inflammatory and anti-inflammatory responses (27,30,31). By inhibiting hyperactivation of
neutrophils and macrophages, FSTL1 helps prevent sustained
inflammatory damage (31,32).
Inflammatory signal transduction is central to the
regulatory functions of FSTL1. By modulating pathways such as NF-κB
and MAPK, FSTL1 is involved in both the initiation and resolution
of inflammation, serving a key role in innate and adaptive immune
responses (33,34). Notably, FSTL1 interacts with the
Toll-like receptor (TLR) system, particularly TLR4, to orchestrate
inflammatory processes (Fig. 1).
Through TLR4 engagement, FSTL1 activates the MyD88-dependent
signalling cascade, triggering the downstream activation of NF-κB
and MAPK pathways, which regulate the production of
pro-inflammatory cytokines and chemokines across various cell types
(2,35). The TLR4/MyD88/NF-κB axis
represents a critical mechanism by which FSTL1 fine-tunes
inflammatory responses in both physiological and pathological
contexts. In certain scenarios, FSTL1 also exhibits
anti-inflammatory properties by inhibiting TLR4-mediated
inflammatory signalling, highlighting its context-dependent
immunomodulatory functions. These unique regulatory mechanisms
enable FSTL1 to orchestrate immune network interactions, serving an
essential role in maintaining immune balance (30).
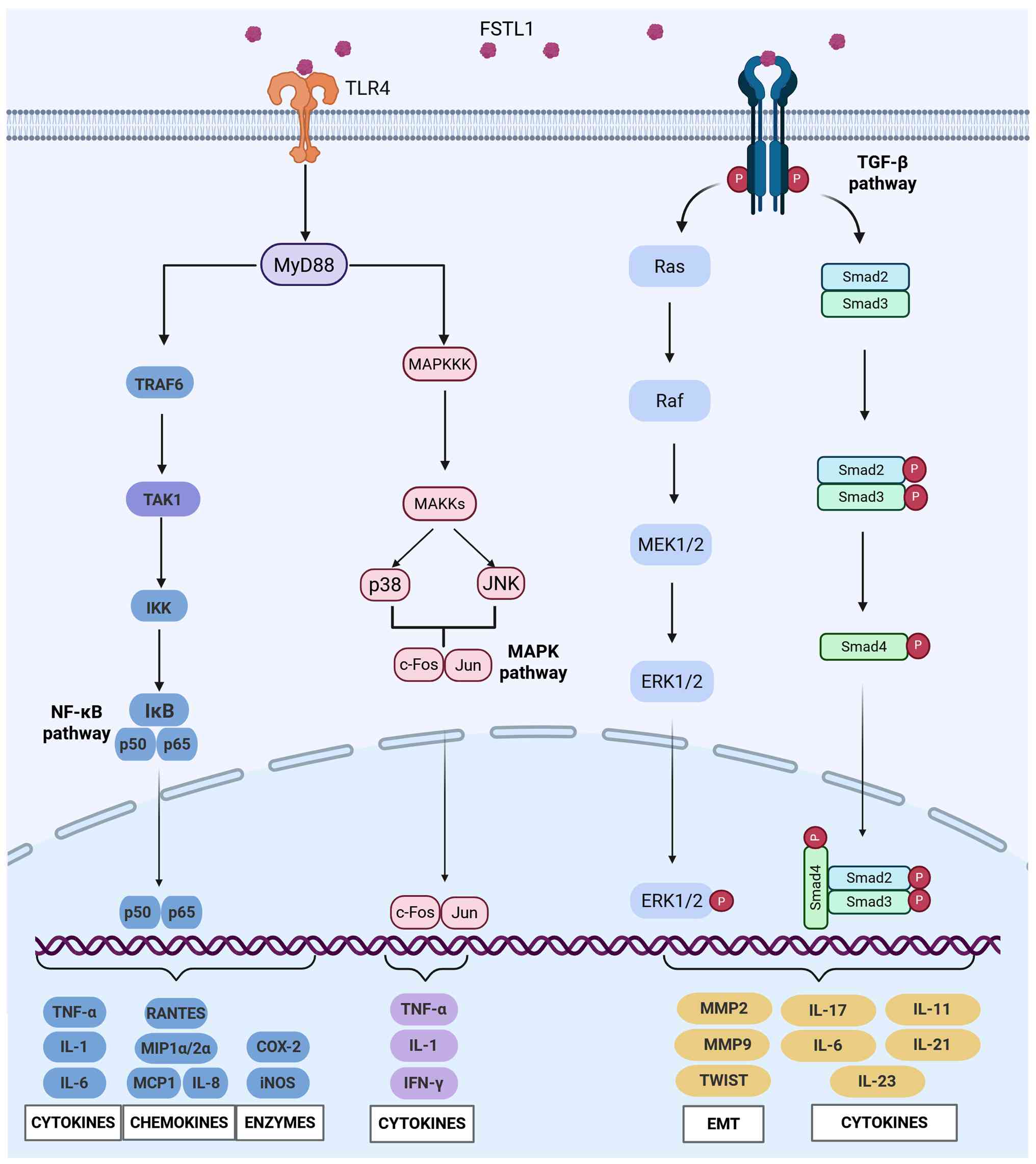 | Figure 1FSTL1-mediated crosstalk between TLR4
and TGF-β signalling pathways in inflammation and fibrosis. This
schematic diagram illustrates the molecular mechanisms by which
FSTL1 orchestrates crosstalk between TLR4 and TGF-β signalling
pathways. Upon binding to cell surface receptors, FSTL1
simultaneously activates multiple downstream signalling cascades.
TLR4 pathway activation: FSTL1 binding to TLR4 triggers
MyD88-dependent signalling, leading to two major downstream
branches: i) TRAF6-TAK1-IKK axis resulting in NF-κB pathway
activation through IκB degradation and nuclear translocation of
p50/p65 heterodimers; ii) MAPKKK-MAPKs cascade activating p38 and
JNK, which converge on AP-1 (c-Fos/Jun) transcription factors.
TGF-β pathway enhancement: FSTL1 promotes TGF-β receptor complex
formation and enhances canonical Smad signalling through Smad2/3
phosphorylation and Smad4 nuclear translocation, while also
activating non-canonical pathways, including the Ras-Raf-MEK-ERK
cascade. Transcriptional outcomes: The convergence of these
pathways results in coordinated transcriptional programs producing:
i) Pro-inflammatory mediators, chemokines and enzymes; ii) fibrotic
mediators including MMPs, pro-fibrotic cytokines and EMT-associated
factors. Pathway interactions: Phosphorylated ERK1/2 and activated
transcription factors (Smad2/3/4 complex, NF-κB, AP-1) coordinate
nuclear gene expression programs that promote both inflammatory
responses and tissue remodelling processes. The diagram
demonstrates how FSTL1 serves as a critical hub protein integrating
immune activation and fibrotic responses through simultaneous
modulation of multiple signalling networks. Created with BioRender.com (Created in BioRender. Deng, Z. (2025)
https://BioRender.com/3o0ydwz). EMT,
epithelial-mesenchymal transition; FSTL1, follistatin-like protein
1; IKK, IκB kinase; iNOS, inducible nitric oxide synthase; MCP1,
monocyte chemoattractant protein-1; MIP, macrophage inflammatory
protein; MMP, matrix metalloproteinase; TLR4, Toll-like receptor
4. |
Regulation of articular cartilage by
FSTL1 pathways
FSTL1 is a key regulator in cartilage biology,
influencing chondrocyte behaviour and tissue homeostasis (7,36,37). In chondrocyte metabolism, FSTL1
promotes chondrocyte proliferation and differentiation, regulates
cartilage matrix synthesis, and participates in complex cartilage
tissue remodelling processes (38-40), all of which are essential for
maintaining cartilage integrity and biomechanical function.
Emerging evidence has highlighted the specific
pathological mechanisms of FSTL1 in joint disorders (41,42). In conditions such as
osteoarthritis, FSTL1 contributes to disease onset and progression
by modulating inflammatory responses, influencing chondrocyte
apoptosis and survival, and regulating cartilage matrix protein
expression through the TGF-β signalling pathway (2,43,44). Cartilage development and
regeneration represent critical areas of FSTL1 research, given its
potential in regulating cartilage-specific transcription factors,
and promoting cartilage regeneration and repair (45,46).
Major signalling pathways and their
mechanisms
FSTL1 modulates key signalling pathways that govern
cellular homeostasis, inflammatory regulation and oncogenesis. The
following chapter summarises the main signalling pathways involving
FSTL1.
TGF-β and Raf/MEK/ERK signalling
pathways
FSTL1 regulates TGF-β superfamily signalling through
intricate molecular mechanisms, serving a central role in cellular
biological processes (Fig. 1).
The TGF-β pathway is essential for regulating cell proliferation,
differentiation, migration and tissue development, with FSTL1
acting as a precise modulator in this context (47,48). Previous studies have established
FSTL1 as a TGF-β-inducible gene encoding a secreted glycoprotein,
creating a bidirectional regulatory relationship between FSTL1 and
TGF-β signalling (44,49).
During mesenchymal cell development and tissue
remodelling, FSTL1 influences TGF-β signal transduction through
multiple mechanisms (39). One
key mechanism involves the direct regulation of Smad protein
phosphorylation, which impacts cell fate decisions (41). In 2025, research demonstrated
that FSTL1 accelerates cellular apoptosis and extracellular matrix
degeneration by activating the TGF-β/Smad2/3 pathway, promoting
dose-dependent increases in phosphorylated (p)-Smad2/Smad2 and
p-Smad3/Smad3 ratios, leading to nuclear translocation of these
transcription factors (44). In
processes such as cardiac regeneration, FSTL1 stimulates
cardiomyocyte proliferation and differentiation via the TGF-β/Smad
pathway, thereby facilitating tissue repair and functional recovery
(44,50). FSTL1 enhances TGF-β non-Smad
signalling by promoting phosphorylation-driven activation of the
Raf/MEK/ERK cascade, thereby amplifying associated fibrogenic and
proliferative effects (51).
Moreover, exercise-induced FSTL1 has been shown to promote cardiac
angiogenesis through the disco-interacting protein 2 homolog A
(DIP2A)-Smad2/3 pathway following myocardial infarction,
highlighting its therapeutic potential in cardiovascular
regeneration (21,52-55).
FSTL1 regulates the TGF-β pathway at multiple
molecular levels (56,57). In this context, FSTL1 modulates
the phosphorylation levels of Smad2/3 proteins, alters their
binding affinity with receptor complexes, and influences their
nuclear translocation and transcriptional activity (40). A 2025 study confirmed the direct
regulatory relationship between FSTL1 and TGF-β/Smad signalling,
showing that TGF-β pathway inhibition using SB-431542 may reverse
FSTL1-induced Smad2 and Smad3 phosphorylation (44). Additionally, FSTL1 regulates the
spatial configuration of type I and II TGF-β receptors, affecting
the stability of their interactions and modulating signal
transduction efficiency (58).
In inflammatory microenvironments, FSTL1 expression
is modulated by inflammatory cytokines such as TGF-β1, IL-1β and
TNF-α (52,59). Its expression is subject to dose-
and time-dependent regulation. Recent findings have suggested that
plasma FSTL1 levels can serve as a non-invasive diagnostic
biomarker for inflammatory and fibrotic diseases (60,61). By controlling the release of
inflammatory mediators and modulating immune cell infiltration and
activation, FSTL1 helps maintain inflammatory homeostasis (31,32).
FSTL1 and TGF-β signalling form a bidirectional
regulatory network that is essential for tissue homeostasis and
remodelling (52,62,63). FSTL1, as a TGF-β-inducible gene,
is upregulated by TGF-β1 through Smad3-dependent transcriptional
activation. In turn, secreted FSTL1 functions as a matricellular
protein that facilitates TGF-β receptor complex formation and
amplifies downstream signalling, creating a positive feedback loop.
At the molecular level, FSTL1 directly regulates Smad2/3
phosphorylation, nuclear translocation and transcriptional
activity, with TGF-β inhibitors reversing FSTL1-induced Smad
phosphorylation (44,57,63). This TGF-β/Smad-FSTL1 axis
orchestrates various physiological processes, including mesenchymal
cell differentiation, extracellular matrix synthesis, cardiac
regeneration and fibrotic responses (63,64). In inflammatory microenvironments,
FSTL1 expression is modulated by cytokines such as TGF-β1, IL-1β
and TNF-α (52,62,63). These multifaceted mechanisms
position FSTL1 as a key amplifier in TGF-β-mediated signalling
networks.
NF-κB signalling pathway
The NF-κB signalling pathway is a key regulator of
cellular inflammation and immune responses (Fig. 1), with a critical role in
inflammatory diseases, immune modulation and cell survival
(65,66).
FSTL1 interacts with the NF-κB pathway in a
bidirectional, context-dependent manner, with effects varying
across tissue types and disease models. Recent studies have shown
that in kidney inflammation, FSTL1 derived from renal tubular
epithelial cells inhibits NF-κB activation, protecting against
kidney fibrosis by reducing renal epithelial inflammation (67,68). Mechanistically, FSTL1 suppresses
TNF-α-induced phosphorylation and nuclear translocation of NF-κB
p65, thereby decreasing IL-1β, IL-6 and ICAM-1 expression, and
limiting leukocyte infiltration (67,68). By contrast, in pulmonary
fibrosis, FSTL1 promotes NF-κB activation, driving
epithelial-mesenchymal transition and inflammation. FSTL1
expression is induced by TGF-β1, leading to NF-κB phosphorylation
and establishing a pro-fibrotic feedback loop. Quercetin alleviates
pulmonary fibrosis by downregulating FSTL1 expression and
modulating NF-κB signalling (68).
FSTL1 is also essential for the initial activation
of NF-κB signalling in specific disease contexts (31,35). By regulating the activity of the
IκB kinase complex, FSTL1 directly affects the phosphorylation and
degradation of NF-κB (31,35,69). In neuroinflammatory models, FSTL1
knockdown reduces microglial activation by suppressing NF-κB
signalling, thereby protecting against neurological damage in
Parkinson's disease models (70). In nucleus pulposus cells, FSTL1
accelerates cellular senescence and intervertebral disc
degeneration (IVDD) through TLR4/NF-κB pathway activation (2). In macrophages and other immune
cells, FSTL1 regulates NF-κB nuclear translocation and
transcriptional activity, influencing the expression and release of
inflammatory cytokines to maintain inflammatory homeostasis
(35).
Oxidative stress is a key trigger for the activation
of the NF-κB signalling pathway, with FSTL1 exerting complex
regulatory effects on intracellular reactive oxygen species (ROS)
production. In high glucose-induced oxidative stress models, FSTL1
exacerbates oxidative stress and cellular transdifferentiation
injury in renal tubular epithelial cells, thereby promoting NF-κB
activation and subsequent inflammatory responses (71).
Regulating inflammatory cytokine expression is a
central mechanism through which FSTL1 modulates the NF-κB pathway
(31,72). FSTL1 precisely controls the
expression of inflammatory cytokines such as TNF-α, IL-6 and IL-1β,
with its regulatory direction being tissue-specific: In renal
epithelial cells, FSTL1 upregulation decreases TNF-α-induced
secretion of IL-1β and IL-6 through NF-κB inhibition (67), whereas in pulmonary epithelial
cells, FSTL1 enhances the secretion of these cytokines via NF-κB
activation (2). Bioinformatics
analyses have confirmed notable positive associations between FSTL1
and key NF-κB pathway components (RELA and NFKB1), as well as
downstream cytokines, in patients with pulmonary fibrosis (68). In various inflammatory disease
models, FSTL1 demonstrates marked anti-inflammatory effects by
inhibiting NF-κB signalling, thereby reducing the intensity and
duration of inflammatory responses (31,73).
Cell apoptosis and survival, central to NF-κB
regulation, are also influenced by FSTL1, which impacts cell
survival decisions through NF-κB-mediated modulation of
apoptosis-related proteins and anti-apoptotic factors (74,75). In neurodegenerative disease
models, particularly Parkinson's disease, inhibition of HOXA11-AS
protects mice from neuroinflammation and neuronal apoptosis through
the microRNA-124-3p-FSTL1-NF-κB axis, illustrating the therapeutic
potential of targeting FSTL1-NF-κB signalling (70). In autoimmune disease models,
FSTL1 has emerged as both a potential therapeutic target and a
biomarker of inflammation, with its expression associated with
disease activity in rheumatic diseases (74,76,77).
The regulation of the NF-κB pathway by FSTL1 is
tissue- and disease-specific (27,78), with its regulatory patterns
varying across cell types and disease models. In neuroinflammation,
it may exert notable inhibitory effects, whereas in tumour
microenvironments, the regulatory mechanisms are more complex
(75,79). Specifically, in tumours, FSTL1
modulates tumour progression and metastasis by regulating the
inflammatory polarisation of tumour-associated macrophages,
offering potential targets for tumour immunotherapy (78,79).
MAPK signalling pathway
FSTL1 regulates a wide range of biological
processes, including cellular stress responses, proliferation,
survival and inflammation, primarily through the MAPK signalling
pathway (Fig. 1) (17,80). Recent research has revealed that
FSTL1 can function as both an activator and an antagonist of MAPK
signalling, with its effects dependent on the cellular context
(81).
Regulation of the MAPK pathway by FSTL1 is highly
cell- and tissue-specific, and its mechanisms vary across different
tumour cell types. In 2024, pioneering research demonstrated that
FSTL1 acts as an antagonist of ERK1/2 phosphorylation during
ciliogenesis and preadipocyte differentiation, revealing an
inhibitory role in MAPK signalling that contrasts with its
activating effects in other contexts (81). In the breast cancer cell line
MDA-MB-231 and in cervical cancer cells, FSTL1 inhibits cell
proliferation by regulating the phosphorylation and activity of
MAPK family members, such as ERK, JNK and p38, thus influencing
downstream gene expression and cell fate (82,83). In cervical cancer, FSTL1
upregulation markedly reduces cell proliferation, migration and
invasion, while promoting apoptosis through downregulation of the
insulin-like growth factor 1 receptor/PI3K/AKT pathway, which
intersects with MAPK signalling.
FSTL1 serves a central role in regulating the
inflammatory response, primarily mediating inflammatory cascades
through the MAPK and NF-κB pathways. In cardiac fibrosis models,
FSTL1 promotes profibrotic mechanisms by activating MAPK signalling
via TGF-β1. FSTL1 upregulation enhances TGF-β signalling, leading
to increased activation of ERK, JNK and p38 pathways. By contrast,
depletion of FSTL1 reduces the levels of ERK, JNK and p38, while
blocking JNK and p38 markedly impairs fibroblast proliferation,
differentiation and migration (84). Furthermore, glycosylated FSTL1
specifically induces cardiac fibroblast proliferation through
ERK1/2 activation, whereas non-glycosylated FSTL1 may exert
distinct effects (85-87).
FSTL1-induced regulation of inflammation exhibits
notable heterogeneity across different tissues (37). In nucleus pulposus cells and in
IVDD, FSTL1 accelerates cell senescence and promotes inflammation
via TLR4/NF-κB pathway activation, rather than directly through
MAPK signalling. FSTL1 expression is upregulated in degenerative
disc tissues, and FSTL1 small interfering RNA (siRNA) notably
inhibits IVDD progression by reducing inflammatory responses
(2,44,88).
Overall, FSTL1 serves diverse regulatory roles in
cell proliferation and inflammatory responses through complex MAPK
signalling pathways. Findings have revealed that FSTL1 can act as
both an activator and an inhibitor of ERK1/2 phosphorylation,
depending on the cellular differentiation state and tissue context.
In cardiac tissue, FSTL1 activates MAPK pathways (ERK, JNK and p38)
through TGF-β1-mediated mechanisms, promoting fibroblast activation
and fibrosis. Conversely, during adipocyte differentiation and
ciliogenesis, FSTL1 acts as an ERK1/2 antagonist, inhibiting MAPK
signalling. These regulatory mechanisms exhibit marked cell- and
tissue-specificity, offering potential novel strategies for tumour
treatment and inflammatory disease research (33,89). Understanding the regulatory role
of FSTL1 in signal transduction may facilitate the development of
targeted therapies (17,58,71,90).
AKT/glycogen synthase kinase (GSK)-3β
signalling pathway
The AKT/GSK-3β signalling pathway is crucial for
regulating cell survival, proliferation, differentiation and
apoptosis (17,91). FSTL1 modulates this pathway
through various molecular mechanisms (41,92). At the cellular level, FSTL1
primarily influences signal transduction by modulating AKT protein
phosphorylation and activity (Fig.
2), a process involving intricate protein interactions and
conformational changes (93,94).
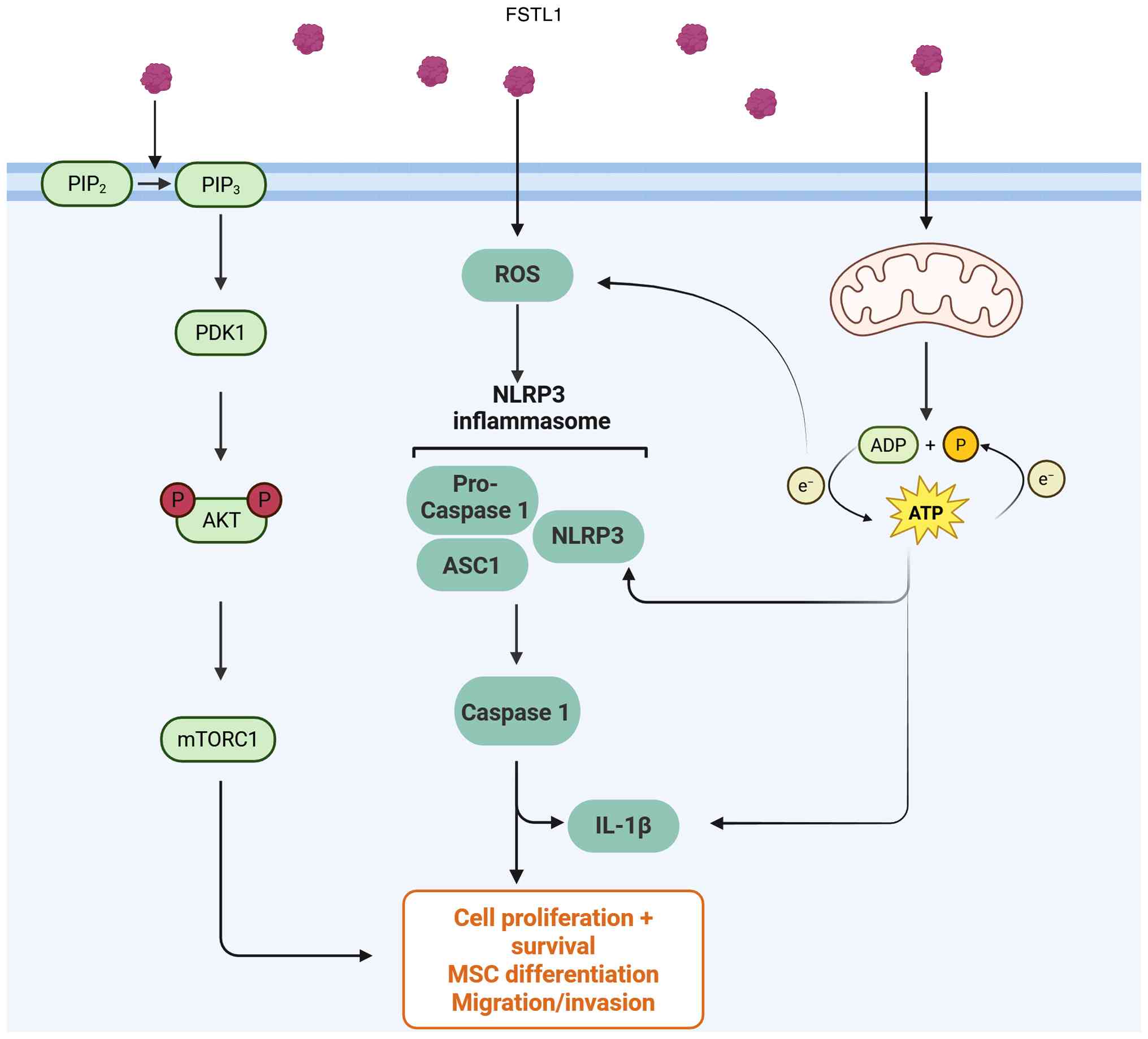 | Figure 2FSTL1 integrates metabolic,
inflammatory and differentiation signalling through the
PI3K/AKT/mTOR and NLRP3 inflammasome pathways. This diagram
delineates the intracellular signalling mechanisms by which FSTL1
regulates cellular metabolism, inflammatory responses and cell fate
decisions. PI3K/AKT/mTORC1 Axis: FSTL1 signalling facilitates
PIP2 to PIP3 at the plasma membrane, a
critical step indicative of PI3K activation. PIP3
recruits and activates PDK1, which in turn phosphorylates and fully
activates AKT. Activated AKT (phosphorylated-AKT) serves as a
central signalling hub, driving the activation of the mTORC1
pathway, a master regulator of anabolic processes. NLRP3
inflammasome activation: Concurrently, FSTL1 stimulation induces
the generation of ROS. Both FSTL1 and ROS contribute to the
assembly and activation of the NLRP3 inflammasome complex. This
multi-protein complex, consisting of NLRP3, the adaptor protein ASC
and pro-caspase-1, leads to the cleavage and activation of
caspase-1. Active caspase-1 then catalyses the maturation and
secretion of the potent pro-inflammatory cytokine IL-1β. FSTL1
enhances electron transport chain activity, which promotes NLRP3
inflammasome activation and IL-1β secretion. Integrated functional
outcomes: The convergence of these FSTL1-initiated signals
orchestrates key cellular responses: Cell proliferation and
survival, MSC differentiation and migration/invasion. Solid arrows
indicate direct activation or conversion processes; tapered lines
represent contributory pathways or functional outcomes. Created
with BioRender.com (Created in BioRender.
Deng, Z. (2025) https://BioRender.com/w34o3gm). ASC,
apoptosis-associated speck-like protein containing a CARD; FSTL1,
follistatin-like protein 1; MSC, mesenchymal stem cell; NLRP3, NLR
family pyrin domain-containing 3; ROS, reactive oxygen species. |
FSTL1 signalling facilitates the conversion of
PIP2 to PIP3 at the plasma membrane, a
critical step indicative of PI3K activation. FSTL1 activates the
PI3K/AKT signalling pathway to regulate cell survival and
proliferation (41,94). In different disease models, FSTL1
modulates the AKT/GSK-3β pathway in a tissue- and disease-specific
manner. In tumour cells, FSTL1 enhances cell proliferation,
migration and invasion by promoting AKT pathway activation, whereas
in neuroprotective contexts, it exhibits inhibitory and protective
effects (91,95).
GSK-3β, a key downstream target of AKT, is also
regulated by FSTL1. In neurodegenerative and inflammatory disease
models, FSTL1 inhibits GSK-3β activity by regulating its
phosphorylation, thereby reducing cellular inflammation and
oxidative stress damage (95).
This mechanism serves a critical role in neuroprotection and
inflammatory regulation, suggesting potential therapeutic
applications.
Oxidative stress is a major pathological mechanism
in various diseases. FSTL1 exerts potent antioxidant effects
through the AKT/GSK-3β signalling pathway. In models of oxidative
stress induced by high glucose and inflammation, FSTL1 activates
the AKT pathway, inhibiting GSK-3β activity and effectively
reducing cellular oxidative damage (96). This mechanism is critical for
preventing and treating diabetic complications and
neurodegenerative diseases. In multiple disease models, FSTL1
exhibits finely tuned regulatory functions through this pathway,
highlighting its essential role in maintaining cellular homeostasis
(92,96).
NLR family pyrin domain-containing 3
(NLRP3) inflammasome signalling pathway
FSTL1 serves a pivotal role in regulating the NLRP3
inflammasome signalling pathway, with its complex molecular
mechanisms being critical for the development and progression of
inflammatory diseases, such as acute pancreatitis, gout and
neuroinflammatory disorders (19,69,97). As a key inflammatory regulatory
complex in the innate immune system, the activation of the NLRP3
inflammasome is tightly controlled at multiple levels by FSTL1
(69,98,99).
During the initiation of inflammation, FSTL1
regulates the release of inflammatory mediators by modulating NLRP3
protein conformational changes and aggregation (Fig. 2) (19,69,100). In immune cells, such as
macrophages and dendritic cells, FSTL1 inhibits excessive
activation of the NLRP3 inflammasome, thereby modulating the
intensity and duration of inflammatory responses and helping to
maintain immune homeostasis (37,100,101).Oxidative stress is a key trigger
for NLRP3 inflammasome activation, and FSTL1 serves an essential
regulatory role in this process (101). By regulating the production and
clearance of ROS within cells, FSTL1 directly influences the
activation threshold of the NLRP3 inflammasome (19,98). In models of oxidative stress
induced by high glucose and inflammation, FSTL1 mitigates aberrant
NLRP3 inflammasome activation by inhibiting excessive ROS
production, thereby reducing inflammatory damage (102,103).
Cellular apoptosis, a critical mechanism in NLRP3
inflammasome regulation, is influenced by FSTL1 through its
modulation of caspase-1 activity and apoptotic pathways (97). In models of neurodegenerative and
autoimmune diseases, FSTL1 effectively reduces cellular
inflammatory damage (17,69,100).
Notably, FSTL1-induced regulation of the NLRP3
inflammasome signalling pathway exhibits notable tissue and disease
specificity. Its regulatory mechanisms are complex and variable
across different cell types and disease models, offering novel
research perspectives for a deeper understanding of the role of
FSTL1 in inflammatory regulation (19,37).
Other signalling pathways
FSTL1-mitochondria interactions
FSTL1 regulates inflammatory responses through its
direct localisation to the mitochondria and involvement in
mitochondrial quality control (MQC). By localising to macrophage
mitochondria, FSTL1 enhances electron transport chain activity,
which promotes NLRP3 inflammasome activation and IL-1β secretion
(Fig. 2) (19,23). Mitochondrial dysfunction triggers
inflammatory cascades through various mechanisms. Mitochondrial ROS
(mtROS) generated by reverse electron transport activate NLRP3
inflammasomes (104), while the
release of mitochondrial DNA (mtDNA) activates cGAS-STING pathways,
driving interferon responses and contributing to cardiovascular
inflammation (105,106). In ischemic cardiomyopathy,
impaired MQC results in excessive mitochondrial fission,
compromised mitophagy, and the release of damage-associated
molecular patterns, which activate inflammatory pathways (107). FSTL1 preserves mitochondrial
integrity by regulating dynamics and calcium homeostasis. During
myocardial ischemia-reperfusion injury, FSTL1 may counteract
nuclear receptor subfamily 4 group A member 1-mediated fission and
promote FUN14 domain-containing 1-mediated mitophagy, helping to
maintain mitochondrial networks (108). FSTL1 also modulates
mitochondrial calcium homeostasis through interactions between
transmembrane BAX inhibitor motif-containing 6 (TMBIM6) and
voltage-dependent anion channel 1 (VDAC1), reducing calcium
overload-induced necroptosis (109). Mitochondrial-induced vascular
endothelial injury is another critical pathway linking
mitochondrial dysfunction with cardiovascular inflammation
(110). Pharmacological
interventions have shown therapeutic potential: Quercetin preserves
DNA-dependent protein kinase catalytic subunit-sirtuin 5-mediated
MQC to inhibit necroptosis (111), while ginsenoside Rb1
ameliorates heart failure through mitochondrial and inflammatory
regulation (112). FSTL1
further influences macrophage polarisation to promote liver
fibrosis (31) and mitigates
acute pancreatitis by regulating NLRP3-mediated pyroptosis
(98). Targeting the
FSTL1-mitochondria-inflammation axis offers a promising therapeutic
strategy.
Endothelial nitric oxide synthase
(eNOS) signalling pathway regulation
FSTL1 is crucial for endothelial cell function and
promotes revascularisation of ischemic tissue by regulating the
eNOS signalling pathway (16,41). Its mechanisms primarily involve
modulating eNOS activity and expression, enhancing endothelial cell
migration and tubule formation, which are vital for tissue repair
and vascular regeneration (113). In cardiovascular and ischemic
diseases, FSTL1 represents a potential therapeutic target by
inhibiting endothelial cell inflammatory cytokine expression and
reducing endothelial cell inflammatory responses (21,90).
AMPK signalling pathway
regulation
As a cellular energy sensor, AMPK regulates
cellular metabolism, proliferation and survival (94,114). FSTL1 influences mitochondrial
biogenesis, fatty acid oxidation and glucose metabolism through the
activation of the AMPK signalling pathway (16,115). In metabolic diseases and tumour
progression, FSTL1 regulates cellular energy metabolism via the
AMPK pathway, impacting cellular survival and proliferation
(116,117).
Specificity of regulatory
mechanisms
The roles of FSTL1 in eNOS and AMPK signalling
exhibit notable tissue and cell specificity (16,118). Its regulatory patterns vary
under different physiological and pathological conditions,
highlighting FSTL1 as a unique multifunctional signalling molecule
the activity of which is tightly controlled by multiple factors
(119). Overall, FSTL1
regulates endothelial cell function and energy metabolism through
eNOS and AMPK signalling pathways, showcasing its versatility in
diverse biological processes (113,120,121).
Conclusion
FSTL1 regulates cell functions and inflammatory
responses through multiple pathways, including the TGF-β, NF-κB,
MAPK and AKT/GSK-3β signalling pathways, the NLRP3 inflammasome and
mitochondria (17,33,91). These pathways are critical in
inflammation, tissue development and tumour progression,
positioning FSTL1 as a key target for studying inflammation, and
cellular proliferation and development (57,92). A deeper understanding of the
mechanisms of FSTL1 in physiological and pathological processes
will provide valuable insights into its complex role in disease
development and progression, paving the way for new therapeutic
strategies.
Advances in FSTL1-associated therapeutic
strategies
FSTL1, a central regulator of inflammation and
fibrosis, has emerged as a potential therapeutic target across
various diseases (63,64,122). Extensive research into the
molecular functions and signalling pathways of FSTL1 has led to the
development of several FSTL1-targeted therapeutic strategies,
showing notable potential in multiple inflammatory disease models
such as osteoarthritis and rheumatoid arthritis (8,36,123).
FSTL1 exhibits pleiotropic biological effects in
inflammatory microenvironments (123,124). Notably, FSTL1 expression is
markedly elevated in the synovial tissues and serum of patients
with rheumatoid arthritis and osteoarthritis, and it is positively
associated with disease severity (123,125,126). FSTL1 forms a key signalling
axis with TGF-β, regulating Smad3 phosphorylation and SOX9
transcription factor activity, and directly influences chondrocyte
differentiation and function (36,44). In osteoarthritis models, FSTL1
enhances inflammation through TLR4/MyD88/NF-κB and MAPK pathways,
increasing the production of inflammatory cytokines such as IL-6,
IL-8 and monocyte chemoattractant protein-1 (37,123,127). Additionally, FSTL1 exerts dual
regulatory effects on extracellular matrix metabolism; moderate
levels of FSTL1 promote cartilage matrix synthesis, whereas higher
concentrations drive cartilage degradation by upregulating
metalloproteinase expression (36,37). These molecular mechanisms provide
a theoretical foundation for the development of FSTL1-targeted
therapeutic strategies.
Monoclonal neutralising antibodies against FSTL1
are among the most extensively studied therapeutic strategies
(63,122). In collagen-induced arthritis
models, FSTL1-neutralising antibodies can markedly reduce joint
swelling, synovial inflammation and bone erosion (8,63,128). In fibrotic disease models, such
as pulmonary fibrosis and skin scar hyperplasia, these antibodies
alleviate tissue fibrosis by inhibiting fibroblast activation and
extracellular matrix deposition (63,99,122). Notably, FSTL1-neutralising
antibodies exhibit synergistic effects when combined with
traditional antirheumatic drugs, potentially enhancing efficacy
while minimising adverse reactions (63).
siRNA and gene silencing technologies offer an
alternative strategy for precise FSTL1-targeted therapy (2,129,130). Using nanoparticle delivery
systems, such as liposomes, optimally designed siRNAs can reduce
FSTL1 expression by >90% with high specificity (129,130). In IVDD models, FSTL1
siRNA-treated mesenchymal stem cells (MSCs) can improve disc tissue
structure and function. MRI and histological examinations have
confirmed a reduction in nucleus pulposus cell apoptosis and
improved extracellular matrix homeostasis in response to FSTL1
siRNA (2,131).
The MSC-FSTL1 interaction presents an innovative
therapeutic direction (36,37,132). FSTL1 demonstrates
concentration-dependent effects: Low concentrations enhance the
chondrogenic and osteogenic differentiation abilities of MSCs,
increasing the expression of markers such as SOX9, collagen type II
α1 chain and aggrecan (133),
whereas high concentrations may inhibit cartilage formation and
trigger inflammatory responses (37). FSTL1 also influences the
immunomodulatory properties of MSCs by regulating their paracrine
functions (30,134). In osteoarthritis models, MSCs
with low FSTL1 expression exhibit stronger anti-inflammatory and
cartilage-protective effects (135).
In disease-specific applications, FSTL1 displays
microenvironment-dependent functions (37,136). In osteoarthritis, early-stage
FSTL1 protects chondrocytes through TGF-β signalling, whereas at
later stages, elevated FSTL1 concentrations accelerate cartilage
degradation (137). FSTL1
regulates disease progression through cross-talk between the
TGF-β/Smad, bone morphogenetic protein and Wnt/β-catenin pathways
(127,137,138). In rheumatoid arthritis, high
FSTL1 expression in synovial tissues and serum provides key
insights into disease progression (6,126), and targeted inhibition of FSTL1
markedly alleviates joint inflammation and tissue damage (28,63). In fibrosis-related diseases,
neutralising antibodies have shown efficacy in alleviating lung and
skin fibrosis induced by bleomycin or TGF-β1, presenting a
promising strategy for treating progressive organ fibrosis
(122,139). Collectively, FSTL1-targeted
therapies, as key modulators of inflammation and fibrosis, have
broad therapeutic potential.
In the future, FSTL1 therapeutic strategies may
evolve toward multi-target inhibition and precision medicine
(128,140). Researchers are investigating
the combined use of FSTL1 inhibitors with different mechanisms of
action, such as pairing neutralising antibodies with siRNA
technology (140) or combining
FSTL1 targeting with traditional antirheumatic drugs for
synergistic effects (17,141).
Personalised treatment approaches based on the FSTL1 expression
profiles and microenvironment characteristics of patients are also
being developed. Assessing FSTL1 levels, and related signalling
molecules in serum and tissues, may help identify the most
effective interventions (123).
The development of nanodelivery systems, including liposomes and
polymer nanoparticles targeting cartilage or synovium, is
progressing rapidly (142,143). These systems can markedly
enhance drug accumulation in target tissues while minimising
systemic side effects (123).
Integrating genomics, proteomics and metabolomics will provide a
comprehensive understanding of the dynamic and functional networks
of FSTL1 across disease stages, laying the groundwork for precise
interventions (17).
Despite the potential of FSTL1-targeted therapies,
their clinical translation faces several challenges. First, since
FSTL1 serves a role in various physiological processes, long-term
inhibition may carry potential risks, particularly in the
cardiovascular system, where FSTL1 has protective effects. Systemic
inhibition could potentially affect cardiac function (16,87). Second, the
concentration-dependent bidirectional regulatory effects of FSTL1
and its microenvironment-dependent functions add complexity to
targeted therapy. Achieving precise control over FSTL1
concentration and activity in target tissues will be a key
challenge (115,119,144). Moreover, to enable precision
treatment, predictive biomarkers related to FSTL1 need to be
developed to screen suitable patient populations and assess
treatment efficacy (27,145,146). Finally, while FSTL1 inhibition
has shown positive results in animal models, large-scale human
clinical trial data are still lacking; multi-centre, randomised
controlled trials will be crucial to evaluate the efficacy and
safety of FSTL1-targeted drugs (52).
In conclusion, FSTL1 is a critical regulator in
inflammatory and fibrotic diseases, and represents a highly
promising therapeutic target (17,63). Through diverse strategies, such
as monoclonal antibodies, siRNA-induced gene silencing and MSC
modulation, FSTL1-targeted therapy offers novel, precise treatment
options for chronic inflammatory diseases such as osteoarthritis
and rheumatoid arthritis (6,8,30,92). This multi-faceted, multi-level
research approach has suggested broad prospects for the application
of FSTL1 in treating inflammatory diseases (147). As basic research deepens and
translational medicine advances, FSTL1-targeted therapy is poised
for clinical application in the near future, offering more
effective and personalised treatment options for patients.
Conclusion and discussion
Multifaceted regulatory roles of FSTL1 in
disease pathophysiology
FSTL1 has emerged as a multifunctional glycoprotein
that orchestrates a wide range of biological processes in both
physiological and pathological contexts. Its widespread expression
in the heart, lungs, skeletal muscle and joint synovium highlights
its critical role in regulating cellular proliferation,
differentiation and apoptosis (4-7,17,118). The structural composition of
FSTL1, consisting of an N-terminal signal peptide, a
follistatin-like domain and a calcium-binding EF-hand domain,
enables complex protein-protein interactions that mediate its
diverse functions (1-3). Studies have revealed the dual roles
of FSTL1 in disease progression, demonstrating both protective and
pathogenic effects depending on tissue context, disease stage and
microenvironmental factors (17,62,87,118).
In osteoarthritis, the upregulation of FSTL1 in
synovial fluid accelerates chondrocyte apoptosis and mitophagy
through activation of the TGF-β/Smad2/3 signalling pathways, thus
driving disease progression (8,39,44,96). By contrast, in cardiovascular
contexts, FSTL1 exhibits cardioprotective properties by promoting
angiogenesis following myocardial infarction, acting as a
cardiokine that aids cardiac regeneration (2,50,52-55,96).
Molecular mechanisms and signalling
networks
FSTL1 exerts its diverse biological functions
through the intricate regulation of multiple interconnected
signalling pathways. The TGF-β/Smad signalling axis is a
cornerstone of FSTL1 function, where it serves as both a
TGF-β-inducible gene and a modulator of TGF-β receptor complex
formation (56-58). Previous mechanistic studies have
shown that FSTL1 directly regulates Smad2/3 phosphorylation in a
dose-dependent manner, with TGF-β pathway inhibitors effectively
reversing FSTL1-induced Smad activation (44,49,148). This bidirectional regulatory
network creates a positive feedback loop that amplifies
TGF-β-mediated responses in processes ranging from cardiac
regeneration to fibrotic pathology.
The NF-κB signalling pathway is regulated in a
tissue-specific manner by FSTL1 (27,78), exhibiting context-dependent
pro-inflammatory and anti-inflammatory effects. In renal tubular
epithelial cells, FSTL1 inhibits NF-κB activation by reducing
TNF-α-induced p65 phosphorylation and nuclear translocation,
offering protection against kidney fibrosis (67,68). By contrast, in pulmonary and
intervertebral disc tissues, FSTL1 positively regulates NF-κB
signalling through TLR4/MyD88-dependent pathways, driving
inflammatory cytokine production and promoting tissue degeneration
(2,68).
The regulation of MAPK signalling by FSTL1 has been
refined by recent research, which has demonstrated that FSTL1
functions as an ERK1/2 antagonist during ciliogenesis and
preadipocyte differentiation (81), in contrast to its activating role
in cardiac fibrosis, where it promotes ERK, JNK and p38 pathway
activation via TGF-β1-mediated mechanisms (85-87). This functional plasticity
highlights the critical role of cellular differentiation states in
determining the signalling outcomes of FSTL1.
The AKT/GSK-3β pathway is another key regulatory
node where FSTL1 exerts context-dependent effects on cell survival
and stress responses (17,91,149). FSTL1 activates PI3K/AKT
signalling, modulating downstream GSK-3β activity through
phosphorylation-dependent mechanisms (94). This axis exhibits bidirectional
functions: It promotes cell proliferation and invasion in tumour
contexts, while providing neuroprotection and antioxidant effects
in neurodegenerative and metabolic diseases by inhibiting GSK-3β
activity (91,95).
FSTL1 orchestrates innate immune responses by
directly regulating the NLRP3 inflammasome (69,98). It modulates NLRP3 protein
oligomerisation and controls the release of inflammatory mediators
in immune cells. This regulatory mechanism exhibits notable tissue
and disease specificity, providing protective effects in
inflammatory and autoimmune conditions (100,102,103).
FSTL1 also regulates inflammatory responses through
its direct localisation to mitochondria and modulation of MQC. By
enhancing electron transport chain activity, FSTL1 promotes NLRP3
inflammasome activation (19,23), while mtROS and released mtDNA
trigger inflammatory cascades via the NLRP3 and cGAS-STING pathways
(105,106). On the other hand, FSTL1
maintains mitochondrial integrity by regulating mitochondrial
dynamics, promoting mitophagy and modulating calcium homeostasis
through TMBIM6-VDAC1 interactions, preventing necroptosis (109). This dual regulatory effect
highlights the complex roles of the mitochondrial-inflammatory
axis.
FSTL1 coordinates vascular and metabolic
homeostasis through the integrated regulation of the eNOS and AMPK
pathways (16). It activates the
AKT-eNOS axis to enhance nitric oxide-mediated angiogenesis and
endothelial function in ischemic tissues, while stimulating AMPK
signalling to promote glucose uptake, mitochondrial biogenesis and
fatty acid oxidation (16).
These tissue-specific regulatory mechanisms link exercise-induced
myokine secretion to systemic metabolic and cardiovascular
benefits.
Therapeutic applications and clinical
translation
The therapeutic landscape of FSTL1-targeted
interventions includes monoclonal antibody strategies, RNA
interference technologies and cell-based therapies. Monoclonal
neutralising antibodies are the most clinically advanced approach,
demonstrating efficacy in collagen-induced arthritis models by
blocking FSTL1-receptor interactions and attenuating downstream
signalling. In fibrotic disease models, FSTL1-neutralising
antibodies markedly reduce tissue fibrosis by inhibiting fibroblast
activation (63,64,122).
siRNA-mediated gene silencing offers precise
temporal control of FSTL1 expression, with nanoparticle delivery
systems achieving >90% suppression with high target specificity
(130). In IVDD models, FSTL1
siRNA-treated MSCs markedly improve disc morphology and function
while reducing nucleus pulposus cell apoptosis (2,44). Disease-specific applications have
shown promising preclinical results, with recent 2025
investigations expanding the role of FSTL1 as a non-invasive
diagnostic biomarker for advanced liver fibrosis (44,60). Plasma FSTL1 levels have
demonstrated diagnostic utility in chronic liver diseases (60).
Challenges and future perspectives
Despite notable progress, the development of
FSTL1-targeted therapeutics faces several challenges that must be
addressed for successful clinical translation. The tissue-specific
and context-dependent nature of FSTL1 regulation presents a
fundamental complexity, as identical interventions may produce
opposing outcomes depending on the cellular context and disease
stage. The concentration-dependent bidirectional regulatory effects
of FSTL1 highlight the need for precise dosing and pharmacokinetic
control in target tissues. Developing controlled-release
formulations, tissue-targeted nanodelivery systems and
feedback-regulated expression platforms may help address these
pharmacological challenges.
A major translational gap is the lack of validated
biomarkers for patient stratification and treatment response
monitoring. While plasma FSTL1 levels are associated with disease
activity in several conditions, comprehensive biomarker panels that
integrate FSTL1 with downstream pathway components are necessary to
enable precision medicine approaches (27,60,145).
The absence of large-scale clinical trial data
remains the most important barrier to the clinical application of
FSTL1-targeted therapeutics. Current evidence largely stems from
preclinical models and small-scale human biomarker studies,
highlighting the need for rigorously designed clinical trials to
establish safety and efficacy. Future research should focus on
mechanistic investigations that elucidate tissue-specific
regulatory mechanisms and identify context-dependent molecular
switches controlling the pro-inflammatory roles of FSTL1 compared
with its anti-inflammatory roles. The development of conditional
knockout models and inducible expression systems will allow for
precise temporal and spatial control, enabling the dissection of
stage-specific roles in disease progression.
Concluding remarks
FSTL1 is a multifaceted molecular regulator at the
core of inflammation, fibrosis and tissue remodelling processes.
Its intricate regulation of pathways such as TGF-β, NF-κB, MAPK,
AKT/GSK-3β and the NLRP3 inflammasome, alongside its novel role in
MQC, positions FSTL1 as a master coordinator of cellular
homeostasis and stress responses. Therapeutic strategies targeting
FSTL1, including monoclonal antibodies, RNA interference and
cell-based approaches, have shown promising efficacy in preclinical
models of osteoarthritis, rheumatoid arthritis, fibrotic diseases
and cardiovascular disorders. The emergence of FSTL1 as both a
diagnostic and prognostic biomarker across various disease contexts
further highlights its clinical relevance. However, successful
clinical translation is dependent on overcoming key challenges,
including the complexity of tissue-specific regulation,
cardiovascular safety concerns and the development of reliable
biomarkers. The integration of multi-omics technologies, precision
medicine strategies and advanced drug delivery systems holds the
potential to address these obstacles and unlock the therapeutic
promise of FSTL1. As mechanistic understanding improves and
translational research progresses, FSTL1-targeted therapies are
poised to become transformative precision treatments for
inflammatory and fibrotic diseases.
Availability of data and materials
Not applicable.
Authors' contributions
All authors made substantial contributions to this
work. CM and JLi were responsible for the core writing work of this
review, including the integration and analysis of literature
content, as well as the writing and organization of the full text.
WJ, XC, JLiu and XT participated in the construction of the
structural framework of this review, clarified the overall logic
and chapter layout of the review, undertook the collection,
collation and provision of relevant references, and completed the
writing of the application section of the manuscript. WL, ZD and ZZ
were responsible for the review and proofreading of this review,
comprehensively examining the academic rigor, content accuracy,
logical consistency and expression standardization of the review,
and putting forward revision suggestions. CM and JL contributed
equally to this article. Data authentication is not applicable. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Natural Science
Foundation of China (grant nos. 81800785, 81972085 and 82172465),
the Natural Science Foundation of Guangdong Province (grant nos.
2023A1515010102 and 2024A1515220060), the Guangdong Provincial Key
Clinical Discipline-Orthopaedics (grant no. 2000005), the Guangdong
Province Medical Science and Technology Research Foundation Project
(grant no. A2024359), the Sanming Project of Shenzhen Health and
Family Planning Commission (grant no. SZSM202311008), Shenzhen
Science and Technology Planning (grant nos. JCYJ20230807115121043,
JCYJ20240813141041053, JCYJ20240813141011015), the Municipal
Financial Subsidy of Shenzhen Medical Key Discipline Construction
(grant no. SZXK025), the Team-based Medical Science Research
Program (grant no. 2024YZZ13), the Shenzhen Portion of the
Shenzhen-Hong Kong Science and Technology Innovation Cooperation
Zone (grant no. HTHZQSWS-KCCYB-2023060), and the Shenzhen Second
People's Hospital Clinical Research Fund of Shenzhen High-level
Hospital Construction Project (grant no. 20253357005).
References
|
1
|
Tanaka M, Murakami K, Ozaki S, Imura Y,
Tong XP, Watanabe T, Sawaki T, Kawanami T, Kawabata D, Fujii T, et
al: DIP2 disco-interacting protein 2 homolog A (Drosophila) is a
candidate receptor for follistatin-related
protein/follistatin-like1-analysis of their binding with TGF-β
superfamily proteins. FEBS J. 277:4278–4289. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yan X, Ding JY, Zhang RJ, Zhang HQ, Kang
L, Jia CY, Liu XY and Shen CL: FSTL1 accelerates nucleus pulposus
cell senescence and intervertebral disc degeneration through
TLR4/NF-κB pathway. Inflammation. 47:1229–1247. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Adams D, Larman B and Oxburgh L:
Developmental expression of mouse Follistatin-like 1 (Fstl1):
Dynamic regulation during organogenesis of the kidney and lung.
Gene Expr Patterns. 7:491–500. 2007. View Article : Google Scholar
|
|
4
|
Lara-Pezzi E, Felkin LE, Birks EJ,
Sarathchandra P, Panse KD, George R, Hall JL, Yacoub MH, Rosenthal
N and Barton PJ: Expression of Follistatin-related genes is altered
in heart failure. Endocrinology. 149:5822–5827. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chan QK, Ngan HY, Ip PP, Liu VW, Xue WC
and Cheung AN: Tumor suppressor effect of follistatin-like 1 in
ovarian and endometrial carcinogenesis: A differential expression
and functional analysis. Carcinogenesis. 30:114–121. 2009.
View Article : Google Scholar
|
|
6
|
Chaly Y, Marinov AD, Oxburgh L, Bushnell
DS and Hirsch R: FSTL1 promotes arthritis in mice by enhancing
inflammatory cytokine/chemokine expression. Arthritis Rheum.
64:1082–1088. 2012. View Article : Google Scholar
|
|
7
|
Peters MC, Di Martino S, Boelens T, Qin J,
van Mil A, Doevendans PA, Chamuleau SAJ, Sluijter JPG and Neef K:
Follistatin-like 1 promotes proliferation of matured human hypoxic
iPSC-cardiomyocytes and is secreted by cardiac fibroblasts. Mol
Ther Methods Clin Dev. 25:3–16. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Clutter SD, Wilson DC, Marinov AD and
Hirsch R: Follistatin-like protein 1 promotes arthritis by
up-regulating IFN-gamma. J Immunol. 182:234–239. 2009. View Article : Google Scholar
|
|
9
|
Kawabata D, Tanaka M, Fujii T, Umehara H,
Fujita Y, Yoshifuji H, Mimori T and Ozaki S: Ameliorative effects
of follistatin-related protein/TSC-36/FSTL1 on joint inflammation
in a mouse model of arthritis. Arthritis Rheum. 50:660–668. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li X, Fang W, Hu Y, Wang Y and Li J:
Characterization of fibronectin type III domain-containing protein
5 (FNDC5) gene in chickens: Cloning, tissue expression, and
regulation of its expression in the muscle by fasting and cold
exposure. Gene. 570:221–229. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu S, Wang L, Wang W, Lin J, Han J, Sun
H, Guo H, Sun R and Wu Q: TSC-36/FRP inhibits vascular smooth
muscle cell proliferation and migration. Exp Mol Pathol.
80:132–140. 2006. View Article : Google Scholar
|
|
12
|
Gawlitta D, van Rijen MHP, Schrijver EJM,
Alblas J and Dhert WJA: Hypoxia impedes hypertrophic chondrogenesis
of human multipotent stromal cells. Tissue Eng Part A.
18:1957–1966. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wilson DC, Marinov AD, Blair HC, Bushnell
DS, Thompson SD, Chaly Y and Hirsch R: Follistatin-like protein 1
is a mesenchyme-derived inflammatory protein and may represent a
biomarker for systemic-onset juvenile rheumatoid arthritis.
Arthritis Rheum. 62:2510–2516. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hayakawa S, Ohashi K, Shibata R, Kataoka
Y, Miyabe M, Enomoto T, Joki Y, Shimizu Y, Kambara T, Uemura Y, et
al: Cardiac Myocyte-derived Follistatin-like 1 prevents renal
injury in a subtotal nephrectomy model. J Am Soc Nephrol.
26:636–646. 2015. View Article : Google Scholar
|
|
15
|
Widera C, Horn-Wichmann R, Kempf T,
Bethmann K, Fiedler B, Sharma S, Lichtinghagen R, Leitolf H,
Ivandic B, Katus HA, et al: Circulating concentrations of
Follistatin-like 1 in healthy individuals and patients with acute
coronary syndrome as assessed by an immunoluminometric sandwich
assay. Clin Chem. 55:1794–1800. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shimano M, Ouchi N, Nakamura K, van Wijk
B, Ohashi K, Asaumi Y, Higuchi A, Pimentel DR, Sam F, Murohara T,
et al: Cardiac myocyte follistatin-like 1 functions to attenuate
hypertrophy following pressure overload. Proc Natl Acad Sci USA.
108:E899–E906. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mattiotti A, Prakash S, Barnett P and van
den Hoff MJB: Follistatin-like 1 in development and human diseases.
Cell Mol Life Sci. 75:2339–2354. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Murphy N, Gaynor KU, Rowan SC, Walsh SM,
Fabre A, Boylan J, Keane MP and McLoughlin P: Altered expression of
bone morphogenetic protein accessory proteins in murine and human
pulmonary fibrosis. Am J Pathol. 186:600–615. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chaly Y, Fu Y, Marinov A, Hostager B, Yan
W, Campfield B, Kellum JA, Bushnell D, Wang Y, Vockley J and Hirsch
R: Follistatin-like protein 1 enhances NLRP3 inflammasome-mediated
IL-1β secretion from monocytes and macrophages. Eur J Immunol.
44:1467–1479. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sundaram GM, Common JE, Gopal FE, Srikanta
S, Lakshman K, Lunny DP, Lim TC, Tanavde V, Lane EB and Sampath P:
'See-saw' expression of microRNA-198 and FSTL1 from a single
transcript in wound healing. Nature. 495:103–106. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ogura Y, Ouchi N, Ohashi K, Shibata R,
Kataoka Y, Kambara T, Kito T, Maruyama S, Yuasa D, Matsuo K, et al:
Therapeutic impact of Follistatin-like 1 on myocardial ischemic
injury in preclinical models. Circulation. 126:1728–1738. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
El-Armouche A, Ouchi N, Tanaka K, Doros G,
Wittköpper K, Schulze T, Eschenhagen T, Walsh K and Sam F:
Follistatin-like 1 in chronic systolic heart failure: A marker of
left ventricular remodeling. Circ Heart Fail. 4:621–627. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chaly Y, Hostager B, Smith S and Hirsch R:
Follistatin-like protein 1 and its role in inflammation and
inflammatory diseases. Immunol Res. 59:266–272. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Torres S, Bartolomé RA, Mendes M, Barderas
R, Fernandez-Aceñero MJ, Peláez-García A, Peña C, Lopez-Lucendo M,
Villar-Vázquez R, de Herreros AG, et al: Proteome profiling of
Cancer-associated fibroblasts identifies novel proinflammatory
signatures and prognostic markers for colorectal cancer. Clin
Cancer Res. 19:6006–6019. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou X, Xiao X, Huang T, Du C, Wang S, Mo
Y, Ma N, Murata M, Li B, Wen W, et al: Epigenetic inactivation of
follistatin-like 1 mediates tumor immune evasion in nasopharyngeal
carcinoma. Oncotarget. 7:16433–16444. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu Y, Tan X, Liu W, Chen X, Hou X, Shen
D, Ding Y, Yin J, Wang L, Zhang H, et al: Follistatin-like protein
1 plays a tumor suppressor role in clear-cell renal cell carcinoma.
Chin J Cancer. 37:22018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li L, Huang S, Yao Y, Chen J, Li J, Xiang
X, Deng J and Xiong J: Follistatin-like 1 (FSTL1) is a prognostic
biomarker and correlated with immune cell infiltration in gastric
cancer. World J Surg Oncol. 18:3242020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li G, Ren H, Wu X, Hu Q, Hong Z, Wang G,
Gu G, Ren J and Li J: Follistatin like protein-1 modulates
macrophage polarization and aggravates dextran sodium
sulfate-induced colitis. Int Immunopharmacol. 83:1064562020.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhang Y, Wang Y, Zheng G, Liu Y, Li J,
Huang H, Xu C, Zeng Y, Zhang X, Qin J, et al: Follistatin-like 1
(FSTL1) interacts with Wnt ligands and Frizzled receptors to
enhance Wnt/β-catenin signaling in obstructed kidneys in vivo. J
Biol Chem. 298:1020102022. View Article : Google Scholar
|
|
30
|
Zheng X, Zhou X, Ma G, Yu J, Zhang M, Yang
C, Hu Y, Ma S, Han Z, Ning W, et al: Endogenous Follistatin-like 1
guarantees the immunomodulatory properties of mesenchymal stem
cells during liver fibrotic therapy. Stem Cell Res Ther.
13:4032022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rao J, Wang H, Ni M, Wang Z, Wang Z, Wei
S, Liu M, Wang P, Qiu J, Zhang L, et al: FSTL1 promotes liver
fibrosis by reprogramming macrophage function through modulating
the intracellular function of PKM2. Gut. 71:2539–2550. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu T, Liu Y, Miller M, Cao L, Zhao J, Wu
J, Wang J, Liu L, Li S, Zou M, et al: Autophagy plays a role in
FSTL1-induced epithelial mesenchymal transition and airway
remodeling in asthma. Am J Physiol Lung Cell Mol Physiol.
313:L27–L40. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jin YK, Li XH, Wang W, Liu J, Zhang W,
Fang YS, Zhang ZF, Dai HP, Ning W and Wang C: Follistatin-Like 1
Promotes Bleomycin-induced pulmonary fibrosis through the
transforming growth factor beta 1/Mitogen-activated protein kinase
signaling pathway. Chin Med J (Engl). 131:1917–1925. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Murakami K, Tanaka M, Usui T, Kawabata D,
Shiomi A, Iguchi-Hashimoto M, Shimizu M, Yukawa N, Yoshifuji H,
Nojima T, et al: Follistatin-related protein/follistatin-like 1
evokes an innate immune response via CD14 and toll-like receptor 4.
FEBS Lett. 586:319–324. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xiao X, Zhang H, Ning W, Yang Z, Wang Y
and Zhang T: Knockdown of FSTL1 inhibits microglia activation and
alleviates depressive-like symptoms through modulating
TLR4/MyD88/NF-κB pathway in CUMS mice. Exp Neurol. 353:1140602022.
View Article : Google Scholar
|
|
36
|
Chaly Y, Hostager B, Smith S and Hirsch R:
The Follistatin-like protein 1 pathway is important for maintaining
healthy articular cartilage. ACR Open Rheumatol. 2:407–414. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li W, Alahdal M, Deng Z, Liu J, Zhao Z,
Cheng X, Chen X, Li J, Yin J, Li Y, et al: Molecular functions of
FSTL1 in the osteoarthritis. Int Immunopharmacol. 83:1064652020.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Tanaka K, Valero-Muñoz M, Wilson RM,
Essick EE, Fowler CT, Nakamura K, van den Hoff M, Ouchi N and Sam
F: Follistatin-Like 1 regulates hypertrophy in heart failure with
preserved ejection fraction. JACC Basic Transl Sci. 1:207–221.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Prakash S, Borreguero LJJ, Sylva M, Flores
Ruiz L, Rezai F, Gunst QD, de la Pompa JL, Ruijter JM and van den
Hoff MJB: Deletion of fstl1 (Follistatin-Like 1) From the
Endocardial/Endothelial lineage causes mitral valve disease.
Arterioscler Thromb Vasc Biol. 37:e116–e130. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chaly Y, Blair HC, Smith SM, Bushnell DS,
Marinov AD, Campfield BT and Hirsch R: Follistatin-like protein 1
regulates chondrocyte proliferation and chondrogenic
differentiation of mesenchymal stem cells. Ann Rheum Dis.
74:1467–1473. 2015. View Article : Google Scholar
|
|
41
|
Ouchi N, Oshima Y, Ohashi K, Higuchi A,
Ikegami C, Izumiya Y and Walsh K: Follistatin-like 1, a secreted
muscle protein, promotes endothelial cell function and
revascularization in ischemic tissue through a Nitric-oxide
Synthase-dependent mechanism. J Biol Chem. 283:32802–32811. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liu S, Yang Y, Jiang S, Xu H, Tang N, Lobo
A, Zhang R, Liu S, Yu T and Xin H: MiR-378a-5p Regulates
proliferation and migration in vascular smooth muscle cell by
targeting CDK1. Front Genet. 10:222019. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hambrock HO, Kaufmann B, Müller S, Hanisch
FG, Nose K, Paulsson M, Maurer P and Hartmann U: Structural
characterization of TSC-36/Flik: Analysis of two charge isoforms. J
Biol Chem. 279:11727–11735. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yan X, Ding JY, Zhang RJ, Wang YX, Zhou
LP, Zhang HQ, Kang L, Jia CY, Liu XY and Shen CL: FSTL1 accelerates
nucleus Pulposus-derived mesenchymal stem cell apoptosis in
intervertebral disc degeneration by activating TGF-β-mediated
Smad2/3 phosphorylation. J Transl Med. 23:2322025. View Article : Google Scholar
|
|
45
|
Javaheri B, Poulet B, Aljazzar A, de Souza
R, Piles M, Hopkinson M, Shervill E, Pollard A, Chan B, Chang YM,
et al: Stable sulforaphane protects against gait anomalies and
modifies bone microarchitecture in the spontaneous STR/Ort model of
osteoarthritis. Bone. 103:308–317. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Rosas IO, Richards TJ, Konishi K, Zhang Y,
Gibson K, Lokshin AE, Lindell KO, Cisneros J, Macdonald SD, Pardo
A, et al: MMP1 and MMP7 as potential peripheral blood biomarkers in
idiopathic pulmonary fibrosis. PLoS Med. 5:e932008. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Bai Y, Liu YJ, Wang H, Xu Y, Stamenkovic I
and Yu Q: Inhibition of the hyaluronan-CD44 interaction by merlin
contributes to the tumor-suppressor activity of merlin. Oncogene.
26:836–850. 2007. View Article : Google Scholar
|
|
48
|
Gorelik L, Gilbride K, Dobles M, Kalled
SL, Zandman D and Scott ML: Normal B cell homeostasis requires B
cell activation factor production by Radiation-resistant cells. J
Exp Med. 198:937–945. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhang XH, Chen Y, Li B, Liu JY, Yang CM
and Ma MZ: Blocking follistatin-like 1 attenuates liver fibrosis in
mice by regulating transforming growth factor-beta signaling. Int J
Clin Exp Pathol. 11:1112–1122. 2018.PubMed/NCBI
|
|
50
|
Song CAL, Adili A, Kari A and Abuduhaer A:
FSTL1 aggravates sepsis-induced acute kidney injury through
regulating TLR4/MyD88/NF-κB pathway in newborn rats. Signa Vitae.
17:167–173. 2021.
|
|
51
|
Bevivino G, Sedda S, Franzè E, Stolfi C,
Di Grazia A, Dinallo V, Caprioli F, Facciotti F, Colantoni A,
Ortenzi A, et al: Follistatin-like protein 1 sustains colon cancer
cell growth and survival. Oncotarget. 9:31278–31290. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Karisa P, Sylviana N, Fitria N and
Setiawan S: FSTL-1 as a novel cardiokine of cardiac angiogenesis: A
systematic review. Vasc Health Risk Manag. 21:437–449. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Xi Y, Hao M, Liang Q, Li Y, Gong DW and
Tian Z: Dynamic resistance exercise increases skeletal
muscle-derived FSTL1 inducing cardiac angiogenesis via
DIP2A-Smad2/3 in rats following myocardial infarction. J Sport
Health Sci. 10:594–603. 2021. View Article : Google Scholar :
|
|
54
|
Alteköester AK and Harvey RP:
Bioengineered FSTL1 patches restore cardiac function following
myocardial infarction. Trends Mol Med. 21:731–733. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Kon M, Ebi Y and Nakagaki K: Effects of
acute sprint interval exercise on Follistatin-like 1 and apelin
secretions. Arch Physiol Biochem. 127:223–227. 2021. View Article : Google Scholar
|
|
56
|
Shang H, Liu X and Guo H: Knockdown of
Fstl1 attenuates hepatic stellate cell activation through the
TGF-β1/Smad3 signaling pathway. Mol Med Rep. 16:7119–7123. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zheng X, Qi C, Zhang S, Fang Y and Ning W:
TGF-β1 induces Fstl1 via the Smad3-c-Jun pathway in lung
fibroblasts. Am J Physiol Lung Cell Mol Physiol. 313:L240–L251.
2017. View Article : Google Scholar
|
|
58
|
Sylva M, Moorman AF and van den Hoff MJB:
Follistatin-like 1 in vertebrate development. Birth Defects Res C
Embryo Today. 99:61–69. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Talavera-Adame D, Wu G, He Y, Ng TT, Gupta
A, Kurtovic S, Hwang JY, Farkas DL and Dafoe DC: Endothelial cells
in co-culture enhance embryonic stem cell differentiation to
pancreatic progenitors and insulin-producing cells through BMP
signaling. Stem Cell Rev Rep. 7:532–543. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Li W, Chi Y, Xiao X, Li J, Sun M, Sun S,
Xu W, Zhang L, Li X, Cheng F, et al: Plasma FSTL-1 as a noninvasive
diagnostic biomarker for patients with advanced liver fibrosis.
Hepatology. 82:669–682. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Tang Y, Ke Y and Ma H: Non-invasive
assessment techniques for renal fibrosis: Advances and
perspectives. Ren Fail. 47:25556852025. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Du R, Li K, Guo K, Chen Z, Han L and Bian
H: FSTL1: A double-edged sword in cancer development. Gene.
906:1482632024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Li X, Fang Y, Jiang D, Dong Y, Liu Y,
Zhang S, Guo J, Qi C, Zhao C, Jiang F, et al: Targeting FSTL1 for
multiple fibrotic and systemic autoimmune diseases. Mol Ther.
29:347–364. 2021. View Article : Google Scholar
|
|
64
|
Xu XY, Du Y, Liu X, Ren Y, Dong Y, Xu HY,
Shi JS, Jiang D, Xu X, Li L, et al: Targeting follistatin like 1
ameliorates liver fibrosis induced by carbon tetrachloride through
TGF-β1-miR29a in mice. Cell Commun Signal. 18:1512020. View Article : Google Scholar
|
|
65
|
Guo Q, Jin Y, Chen X, Ye X, Shen X, Lin M,
Zeng C, Zhou T and Zhang J: NF-κB in biology and targeted therapy:
New insights and translational implications. Signal Transduct
Target Ther. 9:532024. View Article : Google Scholar
|
|
66
|
Mussbacher M, Derler M, Basílio J and
Schmid JA: NF-κB in monocytes and macrophages-an inflammatory
master regulator in multitalented immune cells. Front Immunol.
14:11346612023. View Article : Google Scholar
|
|
67
|
Niu Z, Guo J, Liu X, Chen M, Jin Y, Yao M,
Li X, Che Q, Li S, Zhang C, et al: Renal tubular epithelial-derived
follistatin-like 1 protects against UUO-induced renal fibrosis in
mice via inhibiting NF-κB-mediated epithelial inflammation.
Theranostics. 15:2413–2427. 2025. View Article : Google Scholar
|
|
68
|
Lan Y, Dong C, Wu M, Yuan R, Yang K, Yang
Z, Chen Y, Zhang J, Qi B and Lu X: Quercetin ameliorates
epithelial-mesenchymal transition and inflammation by targeting
FSTL1 and modulating the NF-κB pathway in pulmonary fibrosis. Front
Pharmacol. 16:15947572025. View Article : Google Scholar
|
|
69
|
Chen L and Liu Z: Downregulation of FSTL-1
attenuates the inflammation injury during Streptococcus pneumoniae
infection by inhibiting the NLRP3 and TLR4/NF-κB signaling pathway.
Mol Med Rep. 20:5345–5352. 2019.PubMed/NCBI
|
|
70
|
Yang Q, Guo W, Wang L, Zhang Y, Tian Y,
Ming D, Xiao X and Yang J: Effects of Fstl1 on neuroinflammation
and microglia activation in lipopolysaccharide-induced acute
depression-like mice. Behav Brain Res. 493:1156962025. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Zhang B, Geng H, Zhao K, Omorou M, Liu S,
Ye Z, Zhang F, Luan H and Zhang X: FSTL1 aggravates high
Glucose-induced oxidative stress and transdifferentiation in HK-2
cells. Sci Rep. 15:4342025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Fan N, Sun H, Wang Y, Wang Y, Zhang L, Xia
Z, Peng L, Hou Y, Shen W, Liu R, et al: Follistatin-like 1: A
potential mediator of inflammation in obesity. Mediators Inflamm.
2013:7525192013. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Parola M: Follistatin-like protein 1 and
chronic liver disease progression: A novel pro-inflammatory and
pro-fibrogenic mediator? J Hepatol. 75:1014–1016. 2021.
|
|
74
|
Ni X, Cao X, Wu Y and Wu J: FSTL1
suppresses tumor cell proliferation, invasion and survival in
non-small cell lung cancer. Oncol Rep. 39:13–20. 2018.
|
|
75
|
Dai D, Xie J, Zheng Y, Chen F, Zhao B and
Miao L: H3K27 acetylation-induced FSTL1 upregulation by P300/RUNX1
co-activation exacerbated autophagy-mediated neuronal damage and
NF-κB-stimulated inflammation in Alzheimer's disease.
Cytotechnology. 75:449–460. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Cao H, Han X, Jia Y and Zhang B:
Inhibition of long non-coding RNA HOXA11-AS against
neuroinflammation in Parkinson's disease model via targeting
miR-124-3p mediated FSTL1/NF-κB axis. Aging (Albany NY).
13:11455–11469. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Velikova T, Bakopoulou K, Gulinac M,
Manova E, Valkov H, Miteva D and Shumnalieva R: Emerging
therapeutic and inflammation biomarkers: The role of Meteorin-Like
(Metrnl) and Follistatin-Like 1 (FSTL1) in inflammatory diseases.
Int J Mol Sci. 26:97112025. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Kudo-Saito C, Ishida A, Shouya Y, Teramoto
K, Igarashi T, Kon R, Saito K, Awada C, Ogiwara Y and Toyoura M:
Blocking the FSTL1-DIP2A Axis Improves Anti-tumor immunity. Cell
Rep. 24:1790–1801. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Lau MC, Ng KY, Wong TL, Tong M, Lee TK,
Ming XY, Law S, Lee NP, Cheung AL, Qin YR, et al: FSTL1 promotes
metastasis and chemoresistance in esophageal squamous cell
carcinoma through NFκB-BMP signaling Cross-talk. Cancer Res.
77:5886–5899. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Qu Y, Liu Y and Li R: FSTL1 promotes
inflammatory reaction and cartilage catabolism through interplay
with NFκB signaling pathways in an in vitro ONFH Model.
Inflammation. 42:1491–1503. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Santos L, Guggeri L, Escande C, Badano JL
and Prieto-Echagüe V: FSTL1 is an antagonist of ERK1/2
phosphorylation during ciliogenesis and preadipocyte
differentiation. bioRxiv. Feb 21–2024.Epub ahead of print.
View Article : Google Scholar
|
|
82
|
Liu Z, Zhang H and Hu X: Analysis of the
expression and mechanism of follistatin-like protein 1 in cervical
cancer. Oncol Rep. 50:2152023. View Article : Google Scholar
|
|
83
|
Zhang Y, Xu X, Yang Y, Ma J, Wang L, Meng
X, Chen B, Qin L, Lu T and Gao Y: Deficiency of Follistatin-like
protein 1 accelerates the growth of breast cancer cells at lung
metastatic sites. J Breast Cancer. 21:267–276. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Kuna J, Chmielewski G, Jaśkiewicz Ł,
Knapik M and Krajewska-Włodarczyk M: The role of selected myokines
in the development of cardiovascular diseases, and their
involvement in developing heart failure in rheumatoid arthritis
patients. Int J Mol Sci. 26:81942025. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Xu C, Jiang T, Ni S, Chen C, Li C, Zhuang
C, Zhao G, Jiang S, Wang L, Zhu R, et al: FSTL1 promotes nitric
oxide-induced chondrocyte apoptosis via activating the
SAPK/JNK/caspase3 signaling pathway. Gene. 732:1443392020.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Le Luduec JB, Condamine T, Louvet C,
Thebault P, Heslan JM, Heslan M, Chiffoleau E and Cuturi MC: An
immunomodulatory role for Follistatin-like 1 in heart allograft
transplantation. Am J Transplant. 8:2297–2306. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Horak M, Fairweather D, Kokkonen P, Bednar
D and Bienertova-Vasku J: Follistatin-like 1 and its paralogs in
heart development and cardiovascular disease. Heart Fail Rev.
27:2251–2265. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Wang S, Wei J, Shi J, He Q, Zhou X, Gao X
and Cheng L: Follistatin-like 1 attenuation suppresses
intervertebral disc degeneration in mice through interacting with
TNF-α and Smad signaling pathway. Oxid Med Cell Longev.
2021:66407512021. View Article : Google Scholar
|
|
89
|
Kim HJ, Kang WY, Seong SJ, Kim SY, Lim MS
and Yoon YR: Follistatin-like 1 promotes osteoclast formation via
RANKL-mediated NF-κB activation and M-CSF-induced precursor
proliferation. Cell Signal. 28:1137–1144. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Guo J, Liang W, Li J and Long J: Knockdown
of FSTL1 inhibits oxLDL-induced inflammation responses through the
TLR4/MyD88/NF-κB and MAPK pathway. Biochem Biophys Res Commun.
478:1528–1533. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Yang W, Wu Y, Wang C, Liu Z, Xu M and
Zheng X: FSTL1 contributes to tumor progression via attenuating
apoptosis in a AKT/GSK-3β-dependent manner in hepatocellular
carcinoma. Cancer Biomark. 20:75–85. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Shen H, Cui G, Li Y, Ye W, Sun Y, Zhang Z,
Li J, Xu G, Zeng X, Zhang Y, et al: Follistatin-like 1 protects
mesenchymal stem cells from hypoxic damage and enhances their
therapeutic efficacy in a mouse myocardial infarction model. Stem
Cell Res Ther. 10:172019. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Ou M, Li C, Tang D, Xue W, Xu Y, Zhu P, Li
B, Xie J, Chen J, Sui W, et al: Genotyping, generation and
proteomic profiling of the first human autosomal dominant
osteopetrosis type II-specific induced pluripotent stem cells. Stem
Cell Res Ther. 10:2512019. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Rodrigues Gomes J: A narrative review on
the FSTL-1 protein and its current known impact on cardiovascular
ischaemic disease. Int J Med Students. 12:457–464. 2024. View Article : Google Scholar
|
|
95
|
Liang X, Hu Q, Li B, McBride D, Bian H,
Spagnoli P, Chen D, Tang J and Zhang JH: Follistatin-like 1
attenuates apoptosis via disco-interacting protein 2 homolog A/Akt
pathway after middle cerebral artery occlusion in rats. Stroke.
45:3048–3054. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Oshima Y, Ouchi N, Sato K, Izumiya Y,
Pimentel DR and Walsh K: Follistatin-like 1 is an Akt-regulated
cardioprotective factor secreted by the heart. Circ Res.
103:337–346. 2008.
|
|
97
|
Han YB, Zhou G, Zheng SF, Li G and Zhao T:
Follistatin-like protein 1 promotes mechanical ventilation-induced
NLRP3 inflammasome activation in human pulmonary microvascular
endothelial cells. Inflamm Res. 72:311–323. 2023.
|
|
98
|
Wang L, Wang N, Shi G and Sun S:
Follistatin-like 1 ameliorates severe acute pancreatitis associated
lung injury via inhibiting the activation of NLRP3 inflammasome and
NF-κB pathway. Am J Transl Res. 14:4310–4320. 2022.
|
|
99
|
Fang Y, Zhang S, Li X, Jiang F, Ye Q and
Ning W: Follistatin like-1 aggravates silica-induced mouse lung
injury. Sci Rep. 7:3992017. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Zhou H, Wang L, Lv W and Yu H: The NLRP3
inflammasome in allergic diseases: Mechanisms and therapeutic
implications. Clin Exp Med. 24:2312024. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Ramachandran R, Manan A, Kim J and Choi S:
NLRP3 inflammasome: A key player in the pathogenesis of life-style
disorders. Exp Mol Med. 56:1488–1500. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Zhang Y and Xia Y: Mon-210 Role of
follistatin-like1 (Fstl1) in chronic kidney disease. Kidney Int
Rep. 4(Suppl 1): S3872019. View Article : Google Scholar
|
|
103
|
Zhao Y, Sun J, Zhang W, et al:
Follistatin-like 1 protects against doxorubicin-induced
cardiomyopathy through upregulation of Nrf2. Free Radic Biol Med.
195:164–176. 2023.
|
|
104
|
Casey AM, Ryan DG, Prag HA, Chowdhury SR,
Marques E, Turner K, Gruszczyk AV, Yang M, Wolf DM, Miljkovic JL,
et al: Pro-inflammatory macrophages produce mitochondria-derived
superoxide by reverse electron transport at complex I that
regulates IL-1β release during NLRP3 inflammasome activation. Nat
Metab. 7:493–507. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Marques E, Kramer R and Ryan DG:
Multifaceted mitochondria in innate immunity. NPJ Metab Health Dis.
2:62024. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Zhang Y, Zou M, Wu H, Zhu J and Jin T: The
cGAS-STING pathway drives neuroinflammation and neurodegeneration
via cellular and molecular mechanisms in neurodegenerative
diseases. Neurobiol Dis. 202:1067102024. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Li K, Zhang W, Niu L, Ye Y, Ren J and Liu
C: Lead-free cesium manganese halide nanocrystals embedded glasses
for X-Ray imaging. Adv Sci (Weinh). 10:e22048432023. View Article : Google Scholar :
|
|
108
|
Wang J, Zhuang H, Jia L, He X, Zheng S, Ji
K, Xie K, Ying T, Zhang Y, Li C and Chang X: Nuclear receptor
subfamily 4 group A member 1 promotes myocardial
ischemia/reperfusion injury through inducing mitochondrial fission
factor-mediated mitochondrial fragmentation and inhibiting FUN14
domain containing 1-depedent mitophagy. Int J Biol Sci.
20:4458–4475. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Chang X, Zhou S, Liu J, Wang Y, Guan X, Wu
Q, Liu Z and Liu R: Zishenhuoxue decoction-induced myocardial
protection against ischemic injury through TMBIM6-VDAC1-mediated
regulation of calcium homeostasis and mitochondrial quality
surveillance. Phytomedicine. 132:1553312024. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Pang B, Dong G, Pang T, Sun X, Liu X, Nie
Y and Chang X: Emerging insights into the pathogenesis and
therapeutic strategies for vascular endothelial injury-associated
diseases: Focus on mitochondrial dysfunction. Angiogenesis.
27:623–639. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Chang X, Zhang Q, Huang Y, Liu J, Wang Y,
Guan X, Wu Q, Liu Z and Liu R: Quercetin inhibits necroptosis in
cardiomyocytes after ischemia-reperfusion via
DNA-PKcs-SIRT5-orchestrated mitochondrial quality control.
Phytother Res. 38:2496–2517. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Pu X, Zhang Q, Liu J, Wang Y, Guan X, Wu
Q, Liu Z, Liu R and Chang X: Ginsenoside Rb1 ameliorates heart
failure through DUSP-1-TMBIM-6-mediated mitochondrial quality
control and gut flora interactions. Phytomedicine. 132:1558802024.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Li MY, Gao RP, Zhu Q, Chen Y, Tao BB and
Zhu YC: Skeletal muscle-derived FSTL1 starting up angiogenesis by
regulating endothelial junction via activating Src pathway can be
upregulated by hydrogen sulfide. Am J Physiol Cell Physiol.
325:C1252–C1266. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Garcia D and Shaw RJ: AMPK: Mechanisms of
cellular energy sensing and restoration of metabolic balance. Mol
Cell. 66:789–800. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Seki M, Powers JC, Maruyama S, Zuriaga MA,
Wu CL, Kurishima C, Kim L, Johnson J, Poidomani A, Wang T, et al:
Acute and chronic increases of circulating FSTL1 normalize energy
substrate metabolism in Pacing-induced heart failure. Circ Heart
Fail. 11:e0044862018. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Wang N, Wang B, Maswikiti EP, Yu Y, Song
K, Ma C, Han X, Ma H, Deng X, Yu R and Chen H: AMPK-a key factor in
crosstalk between tumor cell energy metabolism and immune
microenvironment? Cell Death Discov. 10:2372024. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Li JP, He WB, Xu SW, Liu JZ, Huang ZQ, Li
CP, Huang CL, Mai PB, Geng DF, Qiu XF, et al: FSTL1 protects
against acute aortic dissection by suppressing vascular smooth
muscle cell phenotypic switching and degradation of the
extracellular matrix. J Mol Cell Cardiol. 198:60–73. 2025.
View Article : Google Scholar
|
|
118
|
Tania NP, Maarsingh H, Bos IS, Mattiotti
A, Prakash S, Timens W, Gunst QD, Jimenez-Borreguero LJ, Schmidt M,
van den Hoff MJB and Gosens R: Endothelial follistatin-like-1
regulates the postnatal development of the pulmonary vasculature by
modulating BMP/Smad signaling. Pulm Circ. 7:219–231. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Ahsan M, Garneau L and Aguer C: The
bidirectional relationship between AMPK pathway activation and
myokine secretion in skeletal muscle: How it affects energy
metabolism. Front Physiol. 13:10408092022. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Lee HJ, Lee JO, Lee YW, Kim SA, Park SH
and Kim HS: Kalirin, a GEF for Rac1, plays an important role in
FSTL-1-mediated glucose uptake in skeletal muscle cells. Cell
Signal. 29:150–157. 2017. View Article : Google Scholar
|
|
121
|
Chen J, Zong J, Su S, Ji X, Wang L, Han X
and Zhao M: Cardiovascular system is influenced by skeletal
Muscle-derived extracellular vesicles, myokines and MicroRNAs based
on inter-organ communication: A systematic review. Int J Med Sci.
22:2382–2397. 2025. View Article : Google Scholar
|
|
122
|
Dong Y, Geng Y, Li L, Li X, Yan X, Fang Y,
Li X, Dong S, Liu X, Li X, et al: Blocking follistatin-like 1
attenuates bleomycin-induced pulmonary fibrosis in mice. J Exp Med.
212:235–252. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Ni S, Miao K, Zhou X, Xu N, Li C, Zhu R,
Sun R and Wang Y: The involvement of follistatin-like protein 1 in
osteoarthritis by elevating NF-κB-mediated inflammatory cytokines
and enhancing fibroblast like synoviocyte proliferation. Arthritis
Res Ther. 17:912015. View Article : Google Scholar
|
|
124
|
Miyamae T, Marinov AD, Sowders D, Wilson
DC, Devlin J, Boudreau R, Robbins P and Hirsch R: Follistatin-like
Protein-1 is a novel proinflammatory molecule. J Immunol.
177:4758–4762. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Wang Y, Li D, Xu N, Tao W, Zhu R, Sun R,
Fan W, Zhang P, Dong T and Yu L: Follistatin-like protein 1: A
serum biochemical marker reflecting the severity of joint damage in
patients with osteoarthritis. Arthritis Res Ther. 13:R1932011.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Li D, Wang Y, Xu N, Wei Q, Wu M, Li X,
Zheng P, Sun S, Jin Y, Zhang G, et al: Follistatin-like protein 1
is elevated in systemic autoimmune diseases and correlated with
disease activity in patients with rheumatoid arthritis. Arthritis
Res Ther. 13:R172011. View
Article : Google Scholar : PubMed/NCBI
|
|
127
|
Parfenova OK, Kukes VG and Grishin DV:
Follistatin-like proteins: Structure, functions and biomedical
importance. Biomedicines. 9:9992021. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
796. Suppression of collagen-induced
arthritis by neutralizing follistatin-like protein-1. Mol Ther.
16(Suppl 1): S2982008. View Article : Google Scholar
|
|
129
|
Taina-González L and de la Fuente M: The
potential of nanomedicine to unlock the limitless applications of
mRNA. Pharmaceutics. 14:4602022. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Evers MJW, Du W, Yang Q, Kooijmans SAA,
Vink A, van Steenbergen M, Vader P, de Jager SCA, Fuchs SA,
Mastrobattista E, et al: Delivery of modified mRNA to damaged
myocardium by systemic administration of lipid nanoparticles. J
Control Release. 343:207–216. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Liu C, Kong N, Liu H, Zhang Y, Qin W, Zhao
W, Yang X, Wang Y, Cao X, Liu T, et al: FSTL1 and TLR4 interact
with PEDV structural proteins to promote virus adsorption to host
cells. J Virol. 99:e01837242025. View Article : Google Scholar :
|
|
132
|
Nakamura K: Key mediators of the efficacy
of mesenchymal stem cells on in vivo disease models. Cell
Transplant. Jun 25–2025.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Chaly Y, Smith S, Bushnell D, Campfield B,
Blair H and Hirsch R: Follistatin-like protein 1 is an important
regulator of chondrogenesis. In: 2013 ACR/ARHP Annual Meeting; 66.
2013
|
|
134
|
Zheng X, Tian S, Li T, Zhang S, Zhou X,
Liu Y, Su R, Zhang M, Li B, Qi C, et al: Host FSTL1 defines the
impact of stem cell therapy on liver fibrosis by potentiating the
early recruitment of inflammatory macrophages. Signal Transduct
Target Ther. 10:812025. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Wu Y, Zhou S and Smas CM: Downregulated
expression of the secreted glycoprotein follistatin-like 1 (Fstl1)
is a robust hallmark of preadipocyte to adipocyte conversion. Mech
Dev. 127:183–202. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Li F, Qian W, Wang J and Gao M: FSTL1 can
be a promising target in TMJ osteoarthritis via regulating
chondrocyte mitophagy and apoptosis. J Craniofac Surg.
36:1401–1407. 2025. View Article : Google Scholar :
|
|
137
|
Melas IN, Chairakaki AD, Chatzopoulou EI,
Messinis DE, Katopodi T, Pliaka V, Samara S, Mitsos A, Dailiana Z,
Kollia P and Alexopoulos LG: Modeling of signaling pathways in
chondrocytes based on phosphoproteomic and cytokine release data.
Osteoarthritis Cartilage. 22:509–518. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Jia S, Yu Z and Bai L: Exerkines and
osteoarthritis. Front Physiol. 14:13027692023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Liu Y, Xu J, Liu T, Wu J, Zhao J, Wang J,
Zou M, Cao L, Liu X, Pan Y, et al: FSTL1 aggravates cigarette
smoke-induced airway inflammation and airway remodeling by
regulating autophagy. BMC Pulm Med. 21:452021. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Campfield BT, Eddens T, Henkel M, Majewski
M, Horne W, Chaly Y, Gaffen SL, Hirsch R and Kolls JK:
Follistatin-like protein 1 modulates IL-17 signaling via IL-17RC
regulation in stromal cells. Immunol Cell Biol. 95:656–665. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Sisto M, Ribatti D, Ingravallo G and Lisi
S: The expression of Follistatin-like 1 protein is associated with
the activation of the EMT program in Sjögren's syndrome. J Clin
Med. 11:53682022. View Article : Google Scholar
|
|
142
|
Hwang B, Korsnick L, Shen M, Jin L, Singh
Y, Abdalla M, Bauser-Heaton H and Serpooshan V: FSTL-1 loaded 3D
bioprinted vascular patch regenerates the ischemic heart tissue.
iScience. 27:1107702024. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Wang X, Wu DH and Senyo SE: mRNA therapy
for myocardial infarction: A review of targets and delivery
vehicles. Front Bioeng Biotechnol. 10:10370512022. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Fang D, Shi X, Lu T, Ruan H and Gao Y: The
glycoprotein follistatin-like 1 promotes brown adipose
thermogenesis. Metabolism. 98:16–26. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Lang H, Zhang H, Ma M, Wan X, Chen Y, Sun
Q and Cao C: Serum Fstl1, a novel biomarker screened based on
protein array technology, predict acute kidney injury and major
renal adverse events after cardiac surgery: A prospective cohort
study. Clin Chim Acta. 539:79–86. 2023. View Article : Google Scholar
|
|
146
|
Kim DK, Kang SH, Kim JS, Kim YG, Lee YH,
Lee DY, Ahn SY, Moon JY, Lee SH, Jeong KH and Hwang HS: Clinical
implications of circulating follistatin-like protein-1 in
hemodialysis patients. Sci Rep. 13:66372023. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Elsadek BEM, Abdelghany AA, Abd El-Aziz
MA, Madkor HR, Abd Elrady Ahmed A, Abd-Elghaffar SK and Elsadek
AAM: Validation of the diagnostic and prognostic values of ADAMTS5
and FSTL1 in osteoarthritis rat model. Cartilage. 13(2_Suppl):
1263S–1273S. 2021. View Article : Google Scholar
|
|
148
|
Geng Y, Dong Y, Yu M, Zhang L, Yan X, Sun
J, Qiao L, Geng H, Nakajima M, Furuichi T, et al: Follistatin-like
1 (Fstl1) is a bone morphogenetic protein (BMP) 4 signaling
antagonist in controlling mouse lung development. Proc Natl Acad
Sci USA. 108:7058–7063. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Chen F, Hu Q, Huang H, Chen B, Xia Y and
Liu W: Follistatin-like protein 1 increases transepithelial
resistance in kidney epithelial cells through Akt signaling. Mol
Med Rep. 16:4341–4347. 2017. View Article : Google Scholar : PubMed/NCBI
|
















