Introduction
The oral cavity comprises a large and heterogeneous group of malignancies with distinctive histological features, among which oral squamous cell carcinoma (OSCC) is the most common (1). Such heterogeneity is also evident within OSCC at different levels: starting from its etiopathogenesis, where associations with lifestyle habits (smoking, alcohol consumption, exposure to wood and leather dust) and viral infections [human papillomavirus (HPV) and Epstein-Barr virus] have been demonstrated, continuing with its various histopathological variants (such as verrucous carcinoma, basaloid carcinoma, papillary squamous carcinoma, spindle cell carcinoma, acantholytic, or lymphoepithelial carcinoma) and ending with the degree of tumor-associated inflammation (2,3). Although tumor-infiltrating immune cells are recognized as important biomarkers in several malignancies, such as breast, colorectal and lung cancers, their prognostic role in OSCC remains controversial (4,5). Currently, based on the distribution of inflammatory cells within the tumor microenvironment (TME), 'hot' tumors (characterized by a significant infiltrate of tumor-associated immune cells) can be distinguished from 'cold' tumors (showing an almost complete absence of immune cells; Fig. 1) (6).
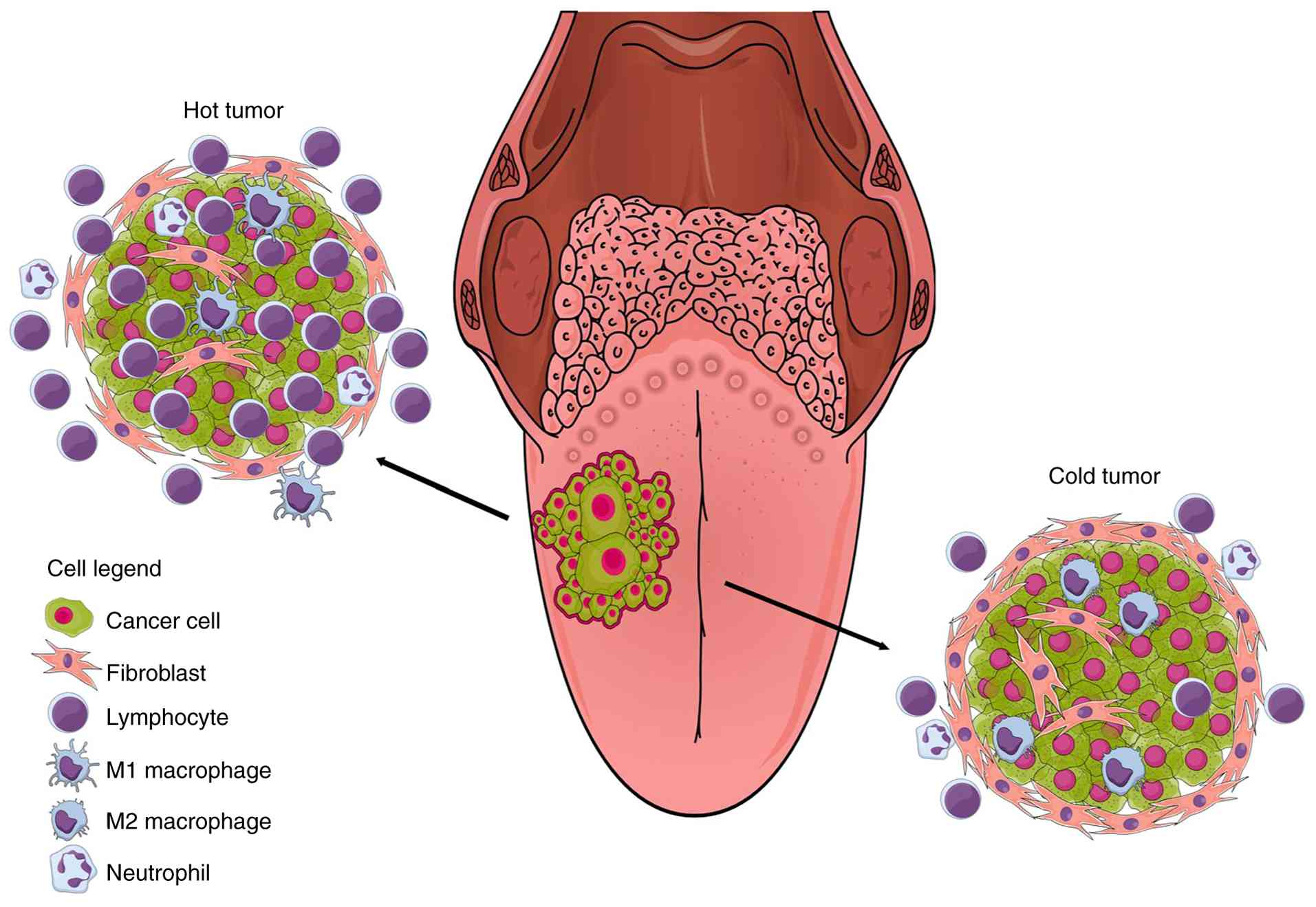 |
Figure 1
Contrasting immune landscapes in oral squamous cell carcinoma: the 'hot' and 'cold' tumor paradigms. The figure illustrates two distinct cancer immune profiles based on the composition of the TME: On the upper left the so-called 'hot tumor' (upper left), while on the bottom right the so-called 'cold tumor' (bottom right). Hot tumors are characterized by a substantial number of immune cells within the TME, primarily consisting of lymphocytes (particularly CD8+ T cells) and M1 macrophages, with low levels of CAFs. This profile includes activated and excluded immunophenotypes and is associated with a improved prognosis, indicating an active role of the immune system in coping cancer growth and spreading. In such cases, IC therapy may yield favorable outcomes, as it can unleash the lymphocytes activity already present around and/or within the tumor. By contrast, cold tumors are characterized by very low levels of lymphocytes, variable number of M2 macrophages and substantial fibrosis. This immuno-desert phenotype highlights the tumor's capacity to evade immune surveillance and correlates with a poorer prognosis. The 'choristers' that shape the TME toward either of these profiles are multiple and not yet fully known, though cytokines and chemokines secreted by cancer cells and other TME components (first of all transforming growth factor β) play a significant role. TME, tumor microenviroment; CAFs, cancer-associated fibroblasts; IC, immune checkpoint.
|
These findings have markedly influenced oncological decision-making in patients with OSCC, particularly in advanced and metastatic disease (4,7). Similar awareness is increasingly emerging across other therapeutic modalities, emphasizing that the cellular constituents of TME exert a substantial effect on patient prognosis and may even serve as therapeutic response predictors (5,7).
The present review provided a comprehensive overview of the principal cellular components of the OSCC TME and discussed their intrinsic involvement in mechanisms of immune evasion, immunosuppression and response to immunotherapy.
Tumor-infiltrating lymphocytes (TILs)
The concept of TILs was first described in 1986 to denote immune cells [mainly T, B and natural killer (NK) lymphocytes] within the TME, where they play a central role in anti-tumor immunity (8). TILs are established prognostic biomarkers in several malignancies, however, their prognostic relevance in OSCC remains controversial (4).
According to the International Immuno-Oncology Biomarker Working Group guidelines (5), TIL density should be reported as the percentage of the stromal area occupied by lymphocytes, a method originally developed for breast cancer and later extended to other tumor types (4). Nonetheless, the literature shows considerable variability in the cut-off values used to distinguish high from low TIL infiltration, with 20 and 30% thresholds being the most frequently adopted in the absence of formal consensus (7).
Furthermore, based on TILs spatial distribution within the TME, solid tumors can be classified into three major immune phenotypes: Immune-inflamed, immune-excluded and immune-desert (Fig. 1). Immune-inflamed tumors are characterized by a dense infiltration of immune cells within the tumor parenchyma. By contrast, immune-excluded tumors display immune cells confined to the peritumoral stroma, without effective infiltration of the tumor core. The immune-desert phenotype ('cold' tumor), exhibits an almost complete absence of immune infiltration. This may result from several mechanisms, including defective recruitment of antigen-presenting cells, impaired T-cell activation or migration and altered cytokine production (5,9). Low Human Leukocyte Antigen (HLA) and interferon-γ (IFN-γ) expression levels have also been reported in such tumors (10). Notably, in a study by Troiano et al (9), all OSCC cases with an immune-desert phenotype were classified as stage IV and exhibited markedly poorer overall survival compared with the other immune profiles.
T lymphocytes
The main component of TILs is represented by T lymphocytes, which migrate from the bloodstream into the tumor as part of the body's immune response to cancer. These cells can be divided into three primary subpopulations: cytotoxic T cells, T helper (Th) cells and regulatory T cells (Treg) (6).
Cytotoxic T-cells, detected by the CD8 marker, can directly kill cancer cells and secrete tumoricidal cytokines (11). In OSCC, CD8+T cells outnumber CD4+T cells, meaning the CD4+/CD8+T ratio is reversed in the TME, compared with the matched peripheral blood (12). A large meta-analysis conducted by Huang et al (13) in 2019 on 2,698 OSCC cases demonstrated that high cytotoxic T-cells infiltration is a strong prognostic factor for improved overall survival (OS), independent of their spatial distribution. Wongpattaraworakul et al (11) also demonstrated that CD3, CD4 and CD8 were associated with OS and progression-free survival (PFS) in univariate analysis, however, none of these markers demonstrate significance in a multivariate analysis.
A higher CD8+/CD4+ T-cell ratio confers superior disease-specific (DSS) and disease-free survival (DFS) and a high CD8+/Forkhead box protein P3 (FOXP3)+ T-cell ratio correlates with OS and DFS (10,14,15). Although effector T cells are abundant in TILs, a number of display an exhausted phenotype [PD-1+, T-cell Immunoglobulin and Mucin-domain containing-3 (TIM-3)+] in tumor tissues, suggesting a partial inability to mount effective immune responses (12).
Th cells, identified by the CD4 marker, play a crucial role in modulating the adaptive immune response against cancer, particularly by promoting cytotoxic T-cell and B-cell proliferation and differentiation (16). However, Huang et al (13) in their meta-analysis demonstrate that most studies report contrasting results regarding their relationship with prognosis in OSCC, probably because CD4+ T cells represent a heterogeneous class divided into multiple subgroups (such as T1, T2, T9, T17, T22, Tregs and Tfh), with distinct and occasionally opposing roles that remain incompletely understood (16).
Tregs are a unique subset of CD4+ T lymphocytes characterized by expression of CD25 and Foxp3 molecules, representing up to 20-30% of the total CD4+ population within the TME. Tregs cells have an anti-inflammatory function through the production of immune-suppressive molecules such as interleukin (IL) 10, IL-35 and transforming growth factor-β (TGF-β), as well as by inhibiting proliferation and activation of other T cell subsets, NK cells, B cells and macrophages, inducing lymphocyte exhaustion via the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) protein (16,17).
Treg levels are associated with prognosis in different malignancies; however, their prognostic significance in OSCC remains inconsistent (18-20). Song et al (19) demonstrated in their study that Th17/Treg ratio was an independent prognostic factor for patient survival with greater predictive accuracy for OS. In contrast. By contrast, Ni et al demonstrates that tumor-infiltrating CD1a+ dendritic cells and CD8+/FOXP3+ ratios are not independent prognostic factors for tongue squamous cell carcinoma patients (14).
Notably, other investigations reported that high FOXP3+ levels are associated with improved prognosis (10,21,22). Several hypotheses have been proposed to explain how high FOXP3+ T-cell numbers may associate with improved outcomes despite their immunosuppressive activity. One possibility is that elevated FOXP3+ TIL expression reflects a generally higher T-cell infiltration, where the cytotoxic effects of CD8+ T cells outweigh Treg-mediated suppression, as suggested by the association between a high CD8+/FOXP3+ ratio and favorable prognosis (22).
Boxberg et al (10) further demonstrated that stromal Tregs (CD4+/FOXP3+) constitute an independent prognostic factor in OSCC. Patients with low stromal Treg density ('FOXP3-non-inflamed' OSCC) had poorer OS, DSS and DFS than those with high-density infiltrates ('FOXP3-inflamed' OSCC). A high FOXP3+/CD4+ ratio, indicating a greater proportion of Tregs among Th cells, was markedly associated with improved outcomes.
Two main hypotheses may justify this finding. First, Tregs may accumulate in a T-cell-inflamed environment with a functional effector T-cell infiltrate driven by IFN type I and II expression, linking to improved prognosis. Alternatively, Tregs might have dual effects: Detrimental when inhibiting effector T cells, but beneficial when reducing protumor chronic inflammation, especially in inflammation-dependent cancers such as OSCC (10). This latter view is indirectly supported by a murine model of carcinogen-induced OSCC, where late-stage Treg depletion did not induce tumor regression but instead promoted rapid tumor growth; early depletion produced no measurable change (23). These observations indicate that Tregs play a complex, context-dependent role in malignancies, varying with tumor type, site, stage and TME factors.
A minor subset of T lymphocytes (<5%) within the TME is represented by γδ T cells (unconventional lymphocytes with a heterodimeric T-cell receptor (TCR) composed of γ and δ chains). Their anti-tumor capabilities are increasingly supported by evidence. In this regard, Lu et al (24) confirmed that γδ T-cell infiltration in tumor tissue can serve as an independent prognostic factor in head and neck squamous cell carcinoma (HNSCC); however, their prognostic role in OSCC remains unproven.
An inverse relationship exists between high T-cell infiltration and low expression of cancer stem cell (CSC) markers (NANOG, SOX2 and nestin) in OSCC, supporting the association between high TIL counts and favorable prognosis (25).
Finally, studies demonstrated that TILs-particularly CD8+ T lymphocytes are abundant in oral premalignant disorders (OPMDs), suggesting that antitumor immune responses develop progressively during carcinogenesis. A significant positive association exists between TIL abundance and dysplasia grade (26,27), due to an increase in CD8+/CD57+ cells during OSCC development (28). Likewise, B-lymphocyte infiltration increases in tongue preneoplastic and neoplastic lesions according to transformation stage (29).
NK lymphocytes
NK cells are a subgroup of lymphocytes that exert anti-tumor activity directly and indirectly. Directly through degranulation of cytotoxic molecules such as perforin and granzyme and also by induction of tumor cell apoptosis via FasL or TNF-related apoptosis-inducing ligand (TRAIL) and indirectly via antibody-dependent cytotoxicity. They also secrete IFN-γ, activating other immune cells within the TME in a proinflammatory and immunostimulatory manner. NK cells constitute only a minor fraction of viable TILs and are identified by expression of CD56 and/or CD57 antigens.
Few studies have assessed their prognostic relevance in HNSCC and OSCC; however, NK cell infiltration consistently correlates with improved survival in both HPV-positive and HPV-negative tumors (13,20,30,31).
B lymphocytes
B cells infiltrating tumor tissues can be classified into three functional categories: Antigen-presenting, antibody-secreting and regulatory B cells. Upon antigen activation and clonal expansion, B cells exert antitumor effects through antigen presentation and antibody secretion; they are also abundant in tertiary lymphoid structures (TLSs). Conversely, regulatory B cells can suppress immune responses and promote tumor progression (32).
In OSCC, high expression of CD20+ B cells is generally associated with improved OS (12,33,34). Notably, a significant reduction in infiltrating CD20+ B cells has been observed in T3-T4 tumors, suggesting that decreased B-cell infiltration may accompany tumor progression. An inverse association between B-lymphocyte count and CSC markers has also been reported (34).
TLSs
TLSs represent ectopic lymphoid aggregates forming in nonlymphoid tissues, including the TME, in response to chronic inflammation and persistent immune stimulation. They have been described in multiple solid tumors, where their presence typically correlates with longer survival. TLSs facilitate germinal center reactions, anti-tumor immune responses mediated by T and B cells and promote lymphocyte recruitment through high endothelial venules (HEVs) (35).
In OSCC, TLS has been proposed as an independent prognostic factor, associated with improved clinical outcomes (32,35,36). TLS abundance follows a gradual increase across immune-desert, immune-excluded and immune-inflamed phenotypes and appears predictive of immunotherapy response (35). The combined presence of high CD8+ and CD57+ TIL densities further enhances prognostic accuracy (36).
TILs in tumor draining lymph nodes (TDLNs)
A few studies have explored the role of T lymphocytes in TDLNs, the natural sites of primary immune response against the OSCC, with noteworthy findings. In a study by Kågedal et al (37), patients with low proportion of CD4+/CD69+ and high proportion of CD8+HLA-DR+ T cells in sentinel lymph nodes showed markedly increased recurrence and decreased DFS compared with those displaying the opposite pattern. CD69 marks recently activated T cells (hours post-activation), while HLA-DR marks longer-activated, potentially exhausted T cells (days post-activation). These data suggest that T-cell exhaustion or impaired activation occurs not only within the tumor but also in TDLNs, potentially contributing to disease progression. Consequently, such patients might benefit from closer clinical monitoring and/or immunotherapy with checkpoint inhibitors.
Similarly, Piersiala et al (38) demonstrated that patients with higher levels of Tregs and immune checkpoints expression (PD-1, TIGIT, CTLA-4 and TIM-3) in TDLNs had the highest recurrence risk, regardless of TNM stage. The authors proposed that these patients might derive the greatest benefit from neoadjuvant immunotherapy.
These findings offer a foundation for investigating the pathobiological mechanisms underlying locoregional metastasis and carcinomas of unknown primary origin, which in the head and neck region frequently localize to laterocervical lymph nodes (39,40).
Macrophages
Macrophages are innate immune cells characterized by remarkable functional plasticity. They can exhibit two main phenotypes: M1 (classically activated) and M2 (alternatively activated). M1 macrophages produce type I proinflammatory cytokines such as IL-1β, IL-1α, IL-12 and TNF-α. They contribute to tumor suppression by presenting tumor-specific antigen and eliminating cancer cells through reactive oxygen species production or antibody dependent cellular cytotoxicity (41).
Conversely, M2 macrophages secrete type II cytokines, including IL-4, IL-6 and IL-10, thereby promoting anti-inflammatory responses. They stimulate tumorigenesis, angiogenesis, matrix remodeling and metastasis and can be identified by surface markers CD163, CD204 and CD206. Macrophages are abundant in the TME of solid tumors, where they are termed tumor-associated macrophages (TAMs); most localize in the tumor stroma and are polarized toward the M2 phenotype (42).
Tumor cells drive M2 polarization of TAMs, while M2 TAMs in turn enhance tumor progression, establishing a positive feedback loop that reinforces malignancy. OSCC cells actively recruit peripheral monocytes to the TME via chemokines such as CCR7 (43) and promote M2 polarization through cytokines such as TGF-β (38), PAI-1 and IL-8 (44), as well as through tumor-derived extracellular vesicles (EVs) (43,45).
TAMs promote cancer progression directly and indirectly. Directly, they secrete growth factors (such as EGF, TGF-β and CCL2) that stimulate cancer cell proliferation and invasion, aided by metalloproteinases (MMPs) (46-48). M2 macrophage-derived EVs enhance OSCC malignancy by delivering miR-23a-3p, which promotes tumor progression via phosphatase and tensin homolog (PTEN) targeting (46). Indirectly, TAMs shape an immunosuppressive milieu by expressing PD-1 ligands (PD-L1 and PD-L2) and immunosuppressive cytokines, thereby inhibiting lymphocyte function, promoting FOXP3+ Treg expansion and reducing immunotherapy efficacy (17).
TAM infiltration is also linked to increased microvessel density (MVD), facilitating angiogenesis, tumor growth and metastasis. TAMs secrete vascular endothelial growth factor (VEGF) and other molecules that promote endothelial cell proliferation, basement membrane degradation and migration, resulting in neovascularization (49,50). Furthermore, TAMs are enriched within lymph node metastasis (LNM), where they remodel the TME by activating fibroblasts and inducing T-cell exhaustion through SPP1-CD44 and CD155-CD226 interactions, aiding metastatic colonization (51).
Fusobacterium nucleatum (Fn), an oral commensal bacterium implicated in chronic periodontitis, also influences macrophage-tumor interactions. Nie et al (52) demonstrated that Fn activates the NFκB pathway in OSCC cells and macrophages, inducing C-X-C motif chemokine ligand 2 secretion that enhances OSCC proliferation, migration and macrophage M2 polarization. Weber et al (53) reported increased macrophage infiltration and M2 polarization in transforming oral leukoplakia (OLP) compared with non-transforming OLP and normal mucosa, suggesting that M2 macrophages facilitate malignant progression by secreting growth factors. Smoking appears to promote this process by inducing M2 polarization (54).
TAMs also contribute to therapy resistance. HPV-negative HNSCC exhibits greater M2 macrophage infiltration than HPV-positive tumors. In this context, TAMs confer radioresistance by releasing heparin-binding EGF-like growth factor (HB-EGF), which activates the non-homologous end-joining (NHEJ) DNA repair pathway, promoting post-radiation survival (55). Similarly, EVs from M2 macrophages can decrease tumor sensitivity to fluorouracil and cisplatin by activating AKT/GSK-3β signaling in cancer cells, leading to apoptosis resistance and enhanced proliferation (56).
Studies have shown that TAM density, particularly CD163+ M2 macrophages, is higher in HNSCC than in normal mucosa, associated with LNM and poor survival (20). A meta-analysis by Chohan et al (57) confirmed that elevated CD163+ TAM levels markedly associate with poor prognosis in OSCC, consistent with several other studies (42,58).
Neutrophils
Neutrophils are innate immune cells frequently found in solid cancers as tumor associated neutrophils (TANs). Similarly to macrophages, TANs may acquire either antitumor (N1), or pro-tumor (N2) activity depending on the cytokines profile of TME. N1 TANs express high levels of IL-12, tumor necrosis factor-α (TNF-α), TRAIL and chemokines CCL3, CXCL9 and CXCL10. Their antitumor effects occur directly, via phagocytosis, degranulation of antimicrobial peptides, generation of reactive oxygen species), or indirectly, through antibody-dependent cytotoxicity and activation of other immune cells (T cells, NK cells, B cells and dendritic cells) in a pro-inflammatory and immunostimulatory manner (59,60).
By contrast, N2 TANs promote tumor growth and dissemination by secreting extracellular matrix (ECM) remodeling enzymes, MMPs and pro-angiogenic factors such as VEGF and immunosuppressive cytokines (IL-10, TGF-β and IL-2). These mediators recruit Tregs, myeloid-derived suppressor cells (MDSCs) and cancer-associated fibroblasts (CAFs), enhancing tumor immune evasion (17,59,60).
TANs typically display an antitumor N1 phenotype at early stages but progressively shift toward a tumor-promoting N2 phenotype as cancer advances, consistent with the immunoediting theory. TGF-β plays a central role in this transition (61). This polarization model, initially identified in mice, has also been demonstrated in human peripheral blood. In cancer patients, high-density neutrophils (HDNs) and low-density neutrophils (LDNs) reflect N1 and N2 phenotypes, respectively. LDNs comprising immature MDSCs and mature HDN-derived cells in a TGF-β-dependent manner, appear transiently during self-limiting inflammation but accumulate persistently in cancer, exhibiting immunosuppressive functions unlike mature HDNs (62).
In OSCC, tumor cells and the TME regulate TAN recruitment, which reciprocally influences tumor growth and TME composition. Chemerin, a multifunctional adipokine secreted by OSCC cells, promotes neutrophil chemotaxis in a concentration-dependent manner. High chemerin expression correlates with increased TAN density, LNM, higher clinical stage, recurrence and reduced OS and DFS, thus serving as an independent prognostic factor (63).
Yu et al (64) demonstrated that TGF-β1 and IL-17A synergistically induce N2 polarization, characterized by high MMP-9 and low CCL3 expression. The main sources of these cytokines are tumor and immune cells within the TME. N2 TANs facilitate OSCC progression by inducing epithelial-mesenchymal transition (EMT) via NF-κB activation and releasing immunosuppressive cytokines. Circulating neutrophils in OSCC patients exhibit a phenotype analogous to TAMs (MMP-9 high/CCL3 low).
Another mechanism contributing to neutrophil-mediated tumor progression is the formation of neutrophil extracellular traps (NETs), web-like DNA structures decorated with histones and enzymes such as myeloperoxidase (MPO), neutrophil elastase (NE)and MMP-9. While MPO may exert cytotoxic effects and histones induce thrombosis (reducing tumor perfusion), NET proteases degrade ECM components, facilitating invasion and metastasis (65,66).
NETs have been detected in OSCC tissues by Garley et al (65,67) and Zhai et al (68) using multiplex immunohistochemistry (mIHC) co-localization of citrullinated histone H3 (citH3) and MPO. NET-rich tumors were associated with larger size and LNM, suggesting a role in disease progression. Also, in vitro, OSCC cells exposed to NET-containing conditioned media exhibited EMT-related gene upregulation, pyroptosis inhibition and increased proliferation and invasion, effects primarily attributed to NE (68).
The oral microbiota also modulates neutrophil-tumor interactions. Streptococcus mutans, which relocates from the mucosal surface to tumor tissue during OSCC development, stimulates cancer cells to produce the oncometabolite kynurenic acid (KYNA). KYNA reprograms the TME by expanding N2 TANs, which release IL-1β, leading to CD8+ T-cell exhaustion and FOXP3+ Treg induction, thereby fostering tumor progression (69).
Across several solid cancers, a high TAN density in histologic sections and an elevated neutrophil-to-lymphocyte ratio (NLR) in peripheral blood are associated with advanced stage, LNM and poor prognosis, making them potential prognostic biomarkers (59,70). Studies in OSCC confirm that increased TAN infiltration and elevated NLR are markedly associated with worse clinical outcomes and reduced OS and PFS (63,70-72).
Mast cells
Mast cells (MCs) are tissue-resident myeloid cells of the innate immune system whose cytoplasm contains basophilic granules rich in histamine, leukotrienes, tryptase, chymase, heparin and inflammatory mediators. MCs are well known for their roles in allergic and autoimmune diseases but are also present in various solid tumors, where they exert tumor-promoting or tumor-suppressive effects depending on tumor type and activation state (73,74).
In OSCC, their role remains controversial. This inconsistency probably arises from variations in case selection, detection methods (such as toluidine blue, Alcian blue-safranin, or anti-tryptase antibodies) (75), MC subtypes (chymase+ vs. chymase-) and tumor site. Some authors reported a positive association between MC density and histological grade, lymphovascular invasion and depth of invasion (76), although its prognostic significance remains uncertain (77,78).
Cai et al (74) demonstrated in their findings that MCs are involved in oral cancer progression and may serve as a potential diagnostic and prognostic marker. Current evidence suggests that MCs are most active in the early stages of OSCC tumorigenesis. Shrestha et al (79) observed the lowest MC count in normal mucosa and significant increases in OPMDs with dyspasia and OSCC, indicating a role in malignant transformation. IL-1 released by MCs promotes epithelial proliferation in OLP, while histamine increases mucosal permeability, facilitating irritant infiltration and enhancing the risk of dysplastic transformation (79,80). Degranulated MCs also release TNF-α, which activates additional MCs, sustaining chronic inflammation and promoting carcinogenesis (81). By contrast, in a systematic review, Tzorakoleftheraki and Koletsa (78) described that some studies propose that mast cells may exert antitumor effects by enhancing immune responses against the cancer cells.
Several studies have reported an increase in MC density in OPMDs compared with OSCC, probably due to TME remodeling and impaired MC migration (79,82). Conversely, Iamaroon et al (83) and Michailidou et al (84) observed persistent or increased MC infiltration in OSCC, particularly within the lamina propria and invasive front, as well as in high MVD areas, supporting their role in invasion and angiogenesis.
MCs facilitate these processes by releasing proteases (tryptase, chymase) that activate MMPs, leading to ECM degradation and metastasis. They also promote angiogenesis through VEGF, tryptase, IL-8 and histamine release (81,85,86), explaining the observed increase in MVD during disease progression (83,87).
Fibroblasts
Fibroblasts are the most abundant non-immune cell type within the TME of solid neoplasms, where they are referred to as CAFs. In OSCC, as in other solid tumors, CAFs predominantly originate from quiescent oral mucosal fibroblasts activated by a variety of tumor-derived cytokines, notably TGF-β, angiopoietin-like protein 3 (ANGPTL3) (88) and platelet-derived growth factor-BB (PDGF-BB) (89). However, pericytes, adipocytes, endothelial cells and bone marrow-derived mesenchymal stem cells have also been identified as potential CAF progenitors in specific contexts (90,91). Most CAFs display a myofibroblastic phenotype, virtually absent in normal oral mucosa (92), similar to that seen in healing wounds and fibrotic disorders. The most commonly used markers for their identification include α-smooth muscle actin (α-SMA), integrin α6 and fibroblast activation protein (FAP) (20). However, these markers lack complete specificity (being expressed by pericytes and smooth muscle cells) and sensitivity (as not all CAF subsets express them) (91).
Attempts to subclassify fibroblasts according to their phenotype, secretory profile, or gene expression have been made, but characterization of CAF heterogeneity in OSCC remains incomplete. CAFs probably represent a subpopulations spectrum with partially overlapping functions and the ability to interconvert (88). In OSCC, CAFs are generally tumor-promoting, with the following main roles: i) Production of cytokines and growth factors that support cancer growth and stimulate fibroblast activation; ii) provision of metabolic support to cancer cells; iii) promotion of tumor invasion; iv) stimulation of angiogenesis; v) facilitation of immune suppression; and vi) induction of therapy resistance (90-92).
Tumor support via soluble factors and extracellular vesicles (EVs)
CAFs promote tumor growth through the secretion of soluble factors such as IL-1β (93), activin A (94), hepatocyte growth factor (HGF) (95), epiregulin (96) and insulin-like growth factor-1 (IGF-1) (97), as well as by releasing EVs that enhance cancer progression. IL-1 secreted by OSCC cells increases CAF proliferation and the expression of secreted cytokines such as CCL7, CXCL1 and IL-8, all of which stimulate tumor cell proliferation in vitro (91). CAFs also produce abundant TGF-β, which drives cancer cell proliferation, invasion and metastasis while inducing the transdifferentiation of normal fibroblasts into CAFs, creating a positive feedback loop (98).
Metabolic reprogramming
OSCC cells profoundly reprogram CAF metabolism, converting them into factories that generate intermediate metabolites for tumor growth. A metabolic coupling arises between tumor cells and CAFs: The latter transfer mitochondria to cancer cells through exosome fusion or cytoplasmic bridges, enhancing oxidative phosphorylation (OXPHOS) in OSCC cells. Concurrently, CAFs undergo aerobic glycolysis ('reverse Warburg effect'), producing pyruvate and lactate that fuel OXPHOS and biosynthetic pathways in cancer cells (91,99-101). Elevated lactate levels induce pseudo-hypoxia in CAFs, leading to caveolin-1 (CAV-1) loss, hypoxia-inducible factor-1α upregulation and secretion of proangiogenic cytokines. Indeed, a glycolytic CAF/tumor-stroma phenotype correlates with higher MVD in OSCC (101). Regarding lipid metabolism, IL-8 produced by cancer cells activates AKT signaling in fibroblasts, leading to ATP citrate lyase phosphorylation and lipid synthesis. These lipids are released and taken up by OSCC cells via CD36, contributing to their metabolic flexibility (102).
Promotion of invasion and ECM remodeling
CAFs facilitate OSCC invasion through ECM remodeling and deposition. They secrete matrix MMPs that degrade type I collagen, enabling cancer cell invasion and produce laminin while reducing fibrillar protein synthesis, thereby strengthening tumor-ECM adhesion via integrins. CAFs also express high levels of hyaluronan synthase 2, which promotes invasion through hyaluronan production (91,103,104). Additionally, they increase ECM stiffness via lysyl oxidase (LOX), which cross-links collagen and elastin. Tumor cells sense this stiffened matrix, activating integrin-FAK signaling and downstream Wnt/β-catenin (105) and YAP pathways (106), promoting EMT and migration. CAFs also contribute to bone invasion by producing receptor activator of NF-κB ligand and promoting osteoclastogenesis (91,107).
Angiogenesis
CAFs are key mediators of neoangiogenesis through VEGF production, Notch pathway activation and recruitment of proangiogenic immune cells such as macrophages (91).
Immune suppression
CAFs promote immune evasion by producing immunosuppressive cytokines (TGF-β, IL-10, IL-6) (20), expressing PD-L1/PD-L2 and creating dense ECM barriers rich in collagen, fibronectin and proteoglycans (hyaluronan, versican) that physically restrict T-cell infiltration. They also induce macrophage polarization toward an M2 phenotype (91,108,109). A subset of OSCC-associated CAFs express tryptophan 2,3-dioxygenase (TDO2), which degrades tryptophan, essential for T-cell proliferation, thereby suppressing CD4+ and CD8+ T-cell activity. TDO2+ CAFs recruit these T cells via CXCL9/10/11 and subsequently induce CD4+ T-cell conversion to Tregs and CD8+ T-cell dysfunction (110).
Therapy resistance
CAFs contribute to cisplatin resistance in HNSCC by forming a collagen barrier that limits drug diffusion (91), sequestering drugs intracellularly (111) and secreting IL-11 (112) and EVs carrying miRNAs that induce chemoresistance-related gene expression in cancer cells (113).
High CAF density is consistently associated with poor prognosis in multiple cancers, including OSCC. Morphologic and immunohistochemistry (IHC) studies show that a high density of α-SMA-positive CAFs correlates with reduced patient survival. Although different cut-offs are used, high CAF density is typically defined by CAF predominance within tumor tissue. A recent meta-analysis by Graizel et al (114) integrating data from 11 studies confirmed high CAF density as an independent negative prognostic factor for OSCC, with particularly adverse effects in younger and female patients. Moreover, Li et al (115) demonstrated that podoplanin-positive CAFs are associated with poorer prognosis, possibly due to enhanced secretion of TGF-β, MMP-2, MMP-9 and exosomal lncRNA FTX, which promotes apoptosis resistance.
Immunotherapy in OSCC
Inflammatory cells represent a principal component of the TME (116) and their interactions with cancer cells constitute a major mechanism enabling carcinomas to evade immune surveillance (5,117). Immunotherapy can be divided into two categories: Active, in which the immune system directly targets tumor cells, and passive, in which costimulatory mechanisms enhance immune activation through cellular receptors, conferring cytotoxic capacity. The discovery of immune checkpoints (ICs), which modulate inflammatory cell activity and consequently tumor progression, has profoundly transformed cancer therapy (118-120).
Among ICs, PD-1 and its ligand PD-L1 are key regulators of T-cell suppression and immune escape (121,122). PD-L1 is a transmembrane protein expressed on tumor and immune cells, including macrophages, dendritic cells and T cells, where it binds to PD-1 on activated lymphocytes to attenuate immune activity. This interaction downregulates TCR signaling, decreases cytokine production and induces T-cell apoptosis or exhaustion, thereby promoting immune tolerance within the TME (123-128). In OSCC, PD-L1 expression has been associated with tumor aggressiveness, advanced clinical stage, LNM and poor prognosis. Hypoxia, oncogenic pathways (such as PI3K/AKT and MAPK) and proinflammatory cytokines like IFN-γ are major inducers of PD-L1 transcription in tumor cells (129).
Importantly, PD-L1 also exhibits non-immune functions that contribute to oncogenesis. Its cytoplasmic domain interacts with downstream signaling molecules that promote proliferation, EMT and resistance to apoptosis. Elevated PD-L1 levels have been associated with increased resistance to radiotherapy and chemotherapy, highlighting its multifaceted role in therapeutic evasion (122). Moreover, PD-L1 expression on exosomes released by tumor cells can suppress immune surveillance at distant sites, enabling systemic immunosuppression and facilitating metastatic dissemination. Thus, PD-L1 serves not only as a predictive biomarker for immune checkpoint blockade but also as a dynamic effector of tumor progression and immune modulation in OSCC (129).
Over the years, in order to identify patients who could benefit from this treatment, researchers have investigated possible cut-offs of PD-L1 expression showing a significant association between their levels and response to treatment (128-131). However, such studies revealed different cut-offs of IHC expression in tumor cells and/or in TME (130-132). Considering the different scoring systems used for the quantification of PD-L1 expression, which represents the gold standard in a number of solid tumors, the combined positive score (CPS), that evaluates through a mathematical formula its expression in tumor cells and immune cells (125), currently is recommended for the suitability for immunotherapy of patients with OSCC (125,126).
The PD-1 blockade agents, pembrolizumab (133) and nivolumab (134), have been approved for metastatic OSCC following important clinical trials. The KEYNOTE-048 phase III trial enrolled 882 patients with recurrent or metastatic HNSCC (339 oral primaries) and showed that pembrolizumab, either alone or combined with platinum and 5-fluorouracil, achieved a statistically significant survival advantage compared with standard therapy (EXTREME regimen) in PD-L1-positive cases (CPS ≥1) (133). After a median follow-up of 45 months, OS improved with pembrolizumab monotherapy in the PD-L1 CPS ≥20 [hazard ratio (HR) 0.61; 95% CI 0.46-0.81)] and CPS ≥1 (HR 0.74; 95% CI 0.61-0.89) groups and was noninferior in the overall population (HR 0.81; 95% CI 0.68-0.97). OS also increased with pembrolizumab-chemotherapy in all CPS categories (≥20, ≥1 and total), with HRs ranging from 0.62-0.71 (135). The CheckMate-141 trial enrolled 361 patients (175 oral cavity primaries) with recurrent HNSCC progressing within six months of platinum-based therapy. Nivolumab markedly improved OS compared to standard agents (HR 0.68; 95% CI 0.54-0.86) after 24.4 months of follow-up (136).
Expanding immunotherapy to earlier disease stages is a current focus. In a multicenter phase II study, pembrolizumab administered preoperatively to 36 patients with locally advanced, non-HPV-related HNSCC improve DFS to 17% at 12 months in high-risk patients. An association between partial tumor response (pTR1-2) and PD-L1 expression was observed, suggesting that pTR could predict favorable outcomes even with a single neoadjuvant dose (137). Another phase II study using two preoperative pembrolizumab cycles reported 1-year DFS rates of 97% (95% CI 71-90%) in intermediate-risk and 66% (95% CI 55-84%) in high-risk groups (138). These findings were confirmed in a subsequent phase III trial (139) and align with results from other solid malignancies (140,141).
Ongoing studies continue to explore combination strategies. Cemiplimab, another PD-1 inhibitor, is being tested with platinum-doublet chemotherapy and cetuximab in locally advanced HNSCC (142). A phase II trial evaluated camrelizumab (anti-PD-1) with nab-paclitaxel and cisplatin as neoadjuvant therapy in 48 patients (16 oral cavity primaries), achieving an overall response rate (ORR) of 89.6% (95% CI 80.9-98.2) (143).
Following the success of PD-1/PD-L1 blockade, novel immune checkpoints are under investigation to further enhance anti-tumor immunity (144,145). Tumor necrosis factor receptor superfamily member 4 (OX40), a T-cell costimulatory receptor, has shown promise: in a phase Ib study, Duhen et al (146) reported increased CD4+ and CD8+ lymphocyte infiltration in peripheral blood and post-surgical specimens following neoadjuvant anti-OX40 (MEDI6469) therapy in locally advanced HNSCC.
Other checkpoint inhibitors under study include antibodies targeting CTLA-4, TIM-3, lymphocyte-activation gene 3 (LAG-3) and IDO, although clinical evidence in OSCC remains limited. CTLA-4 (CD152), expressed on CD4+ and CD8+ T cells, regulates immune homeostasis by inhibiting T-cell activation; its blockade may enhance recognition and elimination of neoplastic cells (146-148). Combination regimens such as nivolumab plus ipilimumab (anti-CTLA-4) and nivolumab plus relatlimab (anti-LAG-3) are currently being evaluated (NCT04080804) (149).
In the context of precision oncology, further modulation of IC pathways, including LAG-3 and TIM-3 blockade, has demonstrated in murine models the capacity to restore CD8+ T-cell activity and suppress tumor growth (146,149). Additional immunostimulatory approaches include cytokine-based therapies using IL-2, IL-1β, IFN-γ and TNF-α, which enhance immune activation and inhibit tumor-induced T-cell apoptosis (150). Monoclonal antibodies targeting Tregs (151) or TAMs (152) also hold potential. These findings are supported by the antitumor activity of IL-2 and IFN-α in enhancing T and NK cell responses within the TME, as demonstrated in other solid tumors (153,154).
Genetic susceptibility may influence immune modulation. For instance, Chien et al (155) found that the IL-23R rs10889677 polymorphism (C allele) conferred a 1.5-fold higher risk of oral cancer and associated with LNM. Furthermore, decreased IFN-γ protein and mRNA expression were shown to negatively are associated with OSCC development (156).
Emerging approaches include adoptive cellular immunotherapy, involving large-scale engineering of tumor-reactive immune cells and therapeutic vaccines. A phase II study evaluating cemiplimab with ISA-101b (targeting HPV16 E6/E7 oncoproteins) in 198 patients demonstrated an ORR of 25.3%, compared with 22.9% in controls. Patients with CPS ≥20 treated with the combination achieved markedly higher response rates and overall survival (157).
Future directions
The growing understanding of the TME in OSCC has transformed the perception of this malignancy from a purely epithelial disorder to a complex ecosystem of cellular and molecular interactions: CAFs, immune cells and ECM components form a dynamic and interdependent network that governs tumor progression, therapeutic resistance and patient outcomes.
Emerging evidence highlights that immune phenotypes ranging from 'immune-inflamed' to 'immune-desert' microenvironments strongly determine prognosis and response to immunotherapy. Patients with high densities of CD8+ T cells, TLSs and NK cells, tend to experience improved OS and favorable responses to PD-1/PD-L1 blockade. Conversely, tumors enriched with M2-polarized macrophages, NETs and CAF-driven fibrosis exhibit immune exclusion, metabolic stress and therapy resistance. Prospective studies on composite biomarkers, should validate integrated scores that combine CPS (PD-L1), immune contexture (CD8+/FOXP3+ ratios, TLS density), stromal load (α-SMA+ CAF index) and systemic inflammation (NLR) to stratify patients for immunotherapy and combination regimens (primary endpoints: OS/DFS; secondary: On-treatment immune remodeling signatures). It may be also possible to pair PD-1/PD-L1 inhibitors with agents that counter macrophage-mediated radioresistance (such as HB-EGF/NHEJ pathway modulators) and with ECM-softening strategies to improve T-cell trafficking. In CAF-directed stroma modulation research, early-phase trials should test inhibitors of TGF-β/IL-6 signaling, LOX-mediated stiffness, or FAP-targeted agents, alone or layered onto PD-1 blockade, to convert immune-excluded tumors into inflamed responders (biopsy-paired spatial readouts recommended). Furthermore, apply single-cell and spatial transcriptomics/proteomics on longitudinal (pre-/post-therapy) specimens to map niches of immune exclusion, CAF heterogeneity (such as TDO2+ subsets) and treatment-induced state shifts that predict durable benefit. In addition, mechanistic and interventional studies on microbiome-immune intervention (such as targeted oral microbiome modulation) should test whether reshaping dysbiosis attenuates N2-skewing and T-cell exhaustion in OSCC.
The summarized data presented in Table I provided an integrated overview of these cellular players, their functional mediators and their prognostic associations within the OSCC microenvironment.
 |
Table I
Key mechanisms, immune phenotypes and prognostic associations in OSCC tumor microenvironment.
|
Table I
Key mechanisms, immune phenotypes and prognostic associations in OSCC tumor microenvironment.
| Component |
Main mechanisms/functions |
Key molecules/markers |
Prognostic/clinical association |
| Tumor cells |
- PD-L1-mediated immune evasion
- Secretion of TGF-β, IL-6, IL-8 promoting CAF activation, EMT, therapy resistance
- Release of EVs carrying PD-L1, miRNAs |
PD-L1, TGF-β, IL-6, IL-8, EVs |
High PD-L1 CPS (≥1 or ≥20) → improved response to pembrolizumab/nivolumab; high PD-L1 heterogeneity limits predictive accuracy |
| TILs |
- CD8+ cytotoxic killing
- CD4+ Th cell modulation
- Tregs suppress immune responses via IL-10, TGF-β, CTLA-4 |
CD3, CD4, CD8, PD-1, CTLA-4 |
High CD8+ and CD8+/+ ratio → ↑ survival Immuneinflamed phenotype → good prognosis Immune-desert phenotype → poor prognosis (Stage IV) |
| B cells/TLSs |
- Antigen presentation, antibody production
- Formation of tertiary lymphoid structures (TLSs) |
CD20, TCL1A |
High CD20+ B cells and TLS abundance → ↑ survival and immunotherapy response |
| NK cells |
- Direct tumor killing (perforin, granzyme)
- Indirect activation of immune cells |
CD56, CD57 |
High NK infiltration → improved OS, both HPV+ and HPV-OSCC |
| Macrophages (TAMs) |
- M1 (IL-12, TNF-α): antitumor
- M2 (IL-10, TGF-β, VEGF): pro-tumor
- Promote EMT, angiogenesis, immune suppression |
CD68, CD163, CD204, CD206 |
High CD163+ M2 TAMs → poor OS, LNM, radioresistance |
| Neutrophils (TANs) |
- N1: cytotoxic and proinflammatory
- N2: immunosuppressive (via TGF-β, IL-17A)
- Form NETs promoting invasion |
MPO, NE, MMP-9, CXCL2 |
High TANs or NLR → poor OS and PFS Chemerin expression → independent poor prognostic factor |
| Mast cells |
- Release histamine, tryptase, VEGF
- Promote angiogenesis and ECM degradation |
Tryptase, Chymase, VEGF |
Controversial: May promote invasion (↑ density in OSCC) or early immune activation in OPMDs |
| Cancer-associated fibroblasts (CAFs) |
- Activated by tumor cytokines (TGF-β, PDGF-BB, ANGPTL3)
- ECM remodeling (MMPs, LOX)
- Reverse Warburg effect (metabolic support)
- Immunosuppression and drug resistance |
α-SMA, FAP, integrin α6, LOX, MMPs, IL-6, TDO2 |
High α-SMA+ CAF density → independent poor prognostic factor Podoplanin+ CAFs → worse OS |
| Immune checkpoints and therapy |
- PD-1/PD-L1 interaction → T-cell exhaustion
- PD-L1 cytoplasmic signaling promotes EMT, proliferation
- New targets: CTLA-4, OX40, LAG-3, TIM-3 |
PD-1, PD-L1, CTLA-4, LAG-3, OX40 |
Pembrolizumab/Nivolumab improve OS in CPS ≥1 tumors (KEYNOTE-048, CheckMate-141) combination regimens under trial |
Together, these data position the CAF-myeloid-PD-L1-oral microbiome-axis as a unifying framework for prognosis and therapeutic design in OSCC.
Conclusions
Translating TME biology into practice will require composite diagnostics and mechanism-based combinations that are tested in biomarker-anchored, biopsy-paired trials. Aligning multi-omics, digital pathology and immuno-oncology within these designs offers the most direct path to durable benefit for patients with OSCC. In conclusion, the OSCC microenvironment represents both a challenge and an opportunity for precision oncology. Targeting the interplay between tumor cells, stromal components and immune effectors holds the potential to overcome therapeutic resistance and improve long-term outcomes. Future studies integrating multi-omics, digital pathology and immuno-oncology will be essential to translate these biological insights into actionable clinical strategies.
Availability of data and materials
Not applicable.
Authors' contributions
Conceptualization was by EC, AB and NS. Review of the literature was by MDM, GC and NS. Writing and original draft preparation was by MDM, SM and EC. Reviewing and editing was by EM, FFVeS, MD and LLM. Methodology was by GC, ELM, MEB and MD. Supervision was by EC, LLM and AB. Critical revision of the manuscript for important intellectual content was by EC, FFVeS and AB. Final approval was by MD, MEB and AB. Data authentication is not applicable. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Acknowledgements
Not applicable.
Funding
The present review received funding from: i) The European Union-NextGeneration EU through the Italian Ministry of University and Research cc under PNRR-M4C2-I1.3 'Health extended alliance for innovative therapies, advanced lab-research and integrated approaches of precision medicine' (acronym HEAL ITALIA). Andrea Ballini and Lorenzo Lo Muzio (grant no. PE_00000019-CUP D73C22001230006). ii) Piano Sviluppo and Coesione Salute, Scheda Monitoraggio Progetti Traiettoria 3 'Medicina rigenerativa, predittiva e personalizzata' Linea di azione 3.1 'Creazione di un programma di medicina di precisione per la mappatura del genoma umano su scala nazionale', project code: T3-AN-13. Project title: Head and neck Oncolgical Personalized outcomE by Artificial Intelligence genoMic analysiS (HOPE-AIMS) (grant no. CUP D73C22000940001).
References
|
1
|
Vieira E, Silva FF, Caponio VCA, Ballini A, Chamorro-Petronacci CM, Lourenzo-Pouso AI, García-García A, Di Domenico M, Suaréz-Peñaranda JM, Pérez-Sayáns M and Padín-Iruegas ME: Smac/DIABLO protein acts as an independent prognostic factor in oral squamous cell carcinoma. Sci Rep. 14:300652024. View Article : Google Scholar
|
|
2
|
Silva FFVE, Caponio VCA, Pérez-Sayáns M, Padín-Iruegas ME, Mascitti M, Chamorro-Petronacci CM, Suaréz-Peñaranda JM and Lorenzo-Pouso AI: Tumor budding is a prognostic factor in head and neck squamous cell carcinoma: A comprehensive meta-analysis and trial sequential analysis. Crit Rev Oncol Hematol. 193:1042022024. View Article : Google Scholar
|
|
3
|
Kim E, Chung M, Jeong HS, Baek CH and Cho J: Histological features of differentiated dysplasia in the oral mucosa: A review of oral invasive squamous cell carcinoma cases diagnosed with benign or low-grade dysplasia on previous biopsies. Hum Pathol. 126:45–54. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Almangush A, Leivo I and Mäkitie AA: Biomarkers for immunotherapy of oral squamous cell carcinoma: Current status and challenges. Front Oncol. 11:6166292021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Salgado R, Denkert C, Demaria S, Sirtaine N, Klauschen F, Pruneri G, Wienert S, Van den Eynden G, Baehner FL, Penault-Llorca F, et al: The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs working group 2014. Ann Oncol. 26:259–271. 2015. View Article : Google Scholar
|
|
6
|
Wu B, Luo D, Wang X, Qiao C, Li R and Liu J: The global trends and distribution in tumor-infiltrating lymphocytes over the past 49 years: bibliometric and visualized analysis. Front Immunol. 15:15118662025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Torri M, Sandell A and Al-Samadi A: The prognostic value of tumor-infiltrating lymphocytes in head and neck squamous cell carcinoma: A systematic review and meta-analysis. Biomed Pharmacother. 180:1175442024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Eberlein TJ, Rosenstein M and Rosenberg SA: Regression of a disseminated syngeneic solid tumor by systemic transfer of lymphoid cells expanded in interleukin 2. J Exp Med. 156:385–397. 1982.PubMed/NCBI
|
|
9
|
Troiano G, Rubini C, Togni L, Caponio VCA, Zhurakivska K, Santarelli A, Cirillo N, Lo Muzio L and Mascitti M: The immune phenotype of tongue squamous cell carcinoma predicts early relapse and poor prognosis. Cancer Med. 9:8333–8344. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Boxberg M, Leising L, Steiger K, Jesinghaus M, Alkhamas A, Mielke M, Pfarr N, Götz C, Wolff KD, Weichert W and Kolk A: Composition and clinical impact of the immunologic tumor microenvironment in oral squamous cell carcinoma. J Immunol. 202:278–291. 2019. View Article : Google Scholar
|
|
11
|
Wongpattaraworakul W, Choi A, Buchakjian MR, Lanzel EA, Kd AR and Simons AL: Prognostic role of tumor-infiltrating lymphocytes in oral squamous cell carcinoma. BMC Cancer. 24:7662024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Quan H, Shan Z, Liu Z, Liu S, Yang L, Fang X, Li K, Wang B, Deng Z, Hu Y, et al: The repertoire of tumor-infiltrating lymphocytes within the microenvironment of oral squamous cell carcinoma reveals immune dysfunction. Cancer Immunol Immunother. 69:465–476. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang Z, Xie N, Liu H, Wan Y, Zhu Y, Zhang M, Tao Y, Zhou H, Liu X, Hou J and Wang C: The prognostic role of tumour-infiltrating lymphocytes in oral squamous cell carcinoma: A meta-analysis. J Oral Pathol Med. 48:788–798. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ni YH, Zhang XX, Lu ZY, Huang XF, Wang ZY, Yang Y, Dong YC, Jing Y, Song Y, Hou YY, et al: Tumor-infiltrating CD1a+ DCs and CD8+/FoxP3+ ratios served as predictors for clinical outcomes in tongue squamous cell carcinoma patients. Pathol Oncol Res. 26:1687–1695. 2020. View Article : Google Scholar
|
|
15
|
Chen WY, Wu CT, Wang CW, Lan KH, Liang HK, Huang BS, Chang YL, Kuo SH and Cheng AL: Prognostic significance of tumor-infiltrating lymphocytes in patients with operable tongue cancer. Radiat Oncol. 13:1572018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Basu A, Ramamoorthi G, Albert G, Gallen C, Beyer A, Snyder C, Koski G, Disis ML, Czerniecki BJ and Kodumudi K: Differentiation and regulation of TH cells: A balancing act for cancer immunotherapy. Front Immunol. 12:6694742021. View Article : Google Scholar
|
|
17
|
Starska-Kowarska K: The role of different immunocompetent cell populations in the pathogenesis of head and neck cancer-regulatory mechanisms of pro- and anti-cancer activity and their impact on immunotherapy. Cancers (Basel). 15:16422023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hayashi T, Yoshikawa K, Suzuki S, Gosho M, Ueda R and Kazaoka Y: Tumor-infiltrating FoxP3+ T cells are associated with poor prognosis in oral squamous cell carcinoma. Clin Exp Dent Res. 8:152–159. 2022. View Article : Google Scholar :
|
|
19
|
Song JJ, Zhao SJ, Fang J, Ma D, Liu XQ, Chen XB, Wang Y, Cheng B and Wang Z: Foxp3 overexpression in tumor cells predicts poor survival in oral squamous cell carcinoma. BMC Cancer. 16:5302016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wondergem NE, Nauta IH, Muijlwijk T, Leemans CR and van de Ven R: The immune microenvironment in head and neck squamous cell carcinoma: on subsets and subsites. Curr Oncol Rep. 22:812020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
de Ruiter EJ, Ooft ML, Devriese LA and Willems SM: The prognostic role of tumor infiltrating T-lymphocytes in squamous cell carcinoma of the head and neck: A systematic review and meta-analysis. Oncoimmunology. 6:e13561482017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fiedler M, Off A, Eichberger J, Spoerl S, Schuderer JG, Taxis J, Bauer RJ, Schreml S, Reichert TE, Ettl T and Weber F: OSCC in never-smokers and never-drinkers is associated with increased expression of tumor-infiltrating lymphocytes and better survival. Cancers (Basel). 15:26882023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chao JL, Korzinkin M, Zhavoronkov A, Ozerov IV, Walker MT, Higgins K, Lingen MW, Izumchenko E and Savage PA: Effector T cell responses unleashed by regulatory T cell ablation exacerbate oral squamous cell carcinoma. Cell Rep Med. 2:1003992021. View Article : Google Scholar
|
|
24
|
Lu Y, Xiang Z, Wang W, Yun B, Yi C, Zhang M, Xie N, Wang C and Zhuang Z: Establishment and validation of a tumor-infiltrating γδT cell related prognostic gene signature in head and neck squamous cell carcinoma. Int Immunopharmacol. 132:1120542024. View Article : Google Scholar
|
|
25
|
Lequerica-Fernández P, Suárez-Canto J, Rodriguez-Santamarta T, Rodrigo JP, Suárez-Sánchez FJ, Blanco-Lorenzo V, Domínguez-Iglesias F, García-Pedrero JM and de Vicente JC: Prognostic relevance of CD4+, CD8+ and FOXP3+ TILs in oral squamous cell carcinoma and correlations with PD-L1 and cancer stem cell markers. Biomedicines. 9:6532021. View Article : Google Scholar
|
|
26
|
Kujan O, Agag M, Smaga M, Vaishnaw Y, Idrees M, Shearston K and Farah CS: PD-1/PD-L1, Treg-related proteins, and tumour-infiltrating lymphocytes are associated with the development of oral squamous cell carcinoma. Pathology. 54:409–416. 2022. View Article : Google Scholar
|
|
27
|
Caponio VCA, Zhurakivska K, Lo Muzio L, Troiano G and Cirillo N: The immune cells in the development of oral squamous cell carcinoma. Cancers (Basel). 15:37792023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
John S, Joseph AP, Raghavan Pillai VB, Ramani P, P J and Ramalingam K: Evaluation of cytotoxic T lymphocytes and natural killer cell distribution in oral squamous cell carcinoma and oral epithelial dysplasia: An immunohistochemical study. Cureus. 16:e563232024.PubMed/NCBI
|
|
29
|
Thomas A, Smitha T, Rao K, Priya NS, Sheethal HS and Chitra S: Expression of CD 20 B-lymphocyte in oral epithelial dysplasia and oral squamous cell carcinoma: A comparative immunohistochemistry study. J Oral Maxillofac Pathol. 27:323–327. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Caruntu A, Moraru L, Lupu M, Vasilescu F, Dumitrescu M, Cioplea M, Popp C, Dragusin A, Caruntu C and Zurac S: Prognostic potential of tumor-infiltrating immune cells in resectable oral squamous cell carcinoma. Cancers (Basel). 13:22682021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kumar S, Chandra A, Agrawal R, Singh P and Pallavi M: Prognostic implications of CD-57 expression in oral squamous cell carcinoma cases. Indian J Pathol Microbiol. 66:815–818. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xie W, Lu J, Chen Y, Wang X, Lu H, Li Q, Jin N, He J, Ou L, Ni J, et al: TCL1A-expressing B cells are critical for tertiary lymphoid structure formation and the prognosis of oral squamous cell carcinoma. J Transl Med. 22:4772024. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sales de Sá R, Miranda Galvis M, Mariz BALA, Leite AA, Schultz L, Almeida OP, Santos-Silva AR, Pinto CAL, Vargas PA, Gollob KJ and Kowalski LP: Increased tumor immune microenvironment CD3+ and CD20+ lymphocytes predict a better prognosis in oral tongue squamous cell carcinoma. Front Cell Dev Biol. 8:6221612021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Suárez-Sánchez FJ, Lequerica-Fernández P, Rodrigo JP, Hermida-Prado F, Suárez-Canto J, Rodríguez-Santamarta T, Domínguez-Iglesias F, García-Pedrero JM and de Vicente JC: Tumor-infiltrating CD20+ B lymphocytes: Significance and prognostic implications in oral cancer microenvironment. Cancers (Basel). 13:3952021. View Article : Google Scholar
|
|
35
|
Liu Z, Meng X, Tang X, Zou W and He Y: Intratumoral tertiary lymphoid structures promote patient survival and immunotherapy response in head neck squamous cell carcinoma. Cancer Immunol Immunother. 72:1505–1521. 2023. View Article : Google Scholar :
|
|
36
|
Li Q, Liu X, Wang D, Wang Y, Lu H, Wen S, Fang J, Cheng B and Wang Z: Prognostic value of tertiary lymphoid structure and tumour infiltrating lymphocytes in oral squamous cell carcinoma. Int J Oral Sci. 12:242020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kågedal Å, Hjalmarsson E, Farrajota Neves da Silva P, Piersiala K, Georén SK, Margolin G, Munck-Wikland E, Winqvist O, Häyry V and Cardell LO: Activation of T helper cells in sentinel node predicts poor prognosis in oral squamous cell carcinoma. Sci Rep. 10:223522020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Piersiala K, Hjalmarsson E, Lagebro V, Farrajota Neves da Silva P, Bark R, Elliot A, Marklund L, Margolin G, Georén SK and Cardell LO: Prognostic value of T regulatory cells and immune checkpoints expression in tumor-draining lymph nodes for oral squamous cell carcinoma. Front Immunol. 15:14554262024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Verginelli F, Pisacane A, Gambardella G, D'Ambrosio A, Candiello E, Ferrio M, Panero M, Casorzo L, Benvenuti S, Cascardi E, et al: Cancer of unknown primary stem-like cells model multi-organ metastasis and unveil liability to MEK inhibition. Nat Commun. 12:24982021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Pisacane A, Cascardi E, Berrino E, Polidori A, Sarotto I, Casorzo L, Panero M, Boccaccio C, Verginelli F, Benvenuti S, et al: Real-world histopathological approach to malignancy of undefined primary origin (MUO) to diagnose cancers of unknown primary (CUPs). Virchows Arch. 482:463–475. 2023. View Article : Google Scholar
|
|
41
|
Takeya M and Komohara Y: Role of tumor-associated macrophages in human malignancies: Friend or foe? Pathol Int. 66:491–505. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shigeoka M, Koma YI, Nishio M, Akashi M and Yokozaki H: Alteration of macrophage infiltrating compartment: A novel view on oral carcinogenesis. Pathobiology. 88:327–337. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhou WH, Wang Y, Yan C, Du WD, Al-Aroomi MA, Zheng L, Lin SF, Gao JX, Jiang S, Wang ZX, et al: CC chemokine receptor 7 promotes macrophage recruitment and induces M2-polarization through CC chemokine ligand 19&21 in oral squamous cell carcinoma. Discov Oncol. 13:672022. View Article : Google Scholar
|
|
44
|
Kai K, Moriyama M, Haque ASMR, Hattori T, Chinju A, Hu C, Kubota K, Miyahara Y, Kakizoe-Ishiguro N, Kawano S and Nakamura S: Oral squamous cell carcinoma contributes to differentiation of monocyte-derived tumor-associated macrophages via PAI-1 and IL-8 production. Int J Mol Sci. 22:94752021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Pang X, Wang SS, Zhang M, Jiang J, Fan HY, Wu JS, Wang HF, Liang XH and Tang YL: OSCC cell-secreted exosomal CMTM6 induced M2-like macrophages polarization via ERK1/2 signaling pathway. Cancer Immunol Immunother. 70:1015–1029. 2021. View Article : Google Scholar
|
|
46
|
Kalogirou EM, Tosios KI and Christopoulos PF: The role of macrophages in oral squamous cell carcinoma. Front Oncol. 11:6111152021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Haque ASMR, Moriyama M, Kubota K, Ishiguro N, Sakamoto M, Chinju A, Mochizuki K, Sakamoto T, Kaneko N, Munemura R, et al: CD206+ tumor-associated macrophages promote proliferation and invasion in oral squamous cell carcinoma via EGF production. Sci Rep. 9:146112019. View Article : Google Scholar
|
|
48
|
Li J, Bao Y, Peng S, Jiang C, Zhu L, Zou S, Xu J and Li Y: M2 macrophages-derived exosomal miRNA-23a-3p promotes the progression of oral squamous cell carcinoma by targeting PTEN. Curr Issues Mol Biol. 45:4936–4947. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yan Q, Liu J, Liu Y, Wen Z, Jin D, Wang F and Gao L: Tumor-associated macrophage-derived exosomal miR21-5p promotes tumor angiogenesis by regulating YAP1/HIF-1α axis in head and neck squamous cell carcinoma. Cell Mol Life Sci. 81:1792024. View Article : Google Scholar
|
|
50
|
Yamagata Y, Tomioka H, Sakamoto K, Sato K, Harada H, Ikeda T and Kayamori K: CD163-positive macrophages within the tumor stroma are associated with lymphangiogenesis and lymph node metastasis in oral squamous cell carcinoma. J Oral Maxillofac Surg. 75:2144–2153. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Dong L, Hu S, Li X, Pei S, Jin L, Zhang L, Chen X, Min A and Yin M: SPP1+ TAM regulates the metastatic colonization of CXCR4+ metastasis-associated tumor cells by remodeling the lymph node microenvironment. Adv Sci (Weinh). 11:e24005242024. View Article : Google Scholar
|
|
52
|
Nie F, Zhang J, Tian H, Zhao J, Gong P, Wang H, Wang S, Yang P and Yang C: The role of CXCL2-mediated crosstalk between tumor cells and macrophages in Fusobacterium nucleatum-promoted oral squamous cell carcinoma progression. Cell Death Dis. 15:2772024. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Weber M, Wehrhan F, Baran C, Agaimy A, Büttner-Herold M, Öztürk H, Neubauer K, Wickenhauser C, Kesting M and Ries J: Malignant transformation of oral leukoplakia is associated with macrophage polarization. J Transl Med. 18:112020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zhu Y, Zhang S, Sun J, Wang T, Liu Q, Wu G, Qian Y, Yang W, Wang Y and Wang W: Cigarette smoke promotes oral leukoplakia via regulating glutamine metabolism and M2 polarization of macrophage. Int J Oral Sci. 13:252021. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Fu E, Liu T, Yu S, Chen X, Song L, Lou H, Ma F, Zhang S, Hussain S, Guo J, et al: M2 macrophages reduce the radiosensitivity of head and neck cancer by releasing HB-EGF. Oncol Rep. 44:698–710. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Tomita R, Sasabe E, Tomomura A and Yamamoto T: Macrophage-derived exosomes attenuate the susceptibility of oral squamous cell carcinoma cells to chemotherapeutic drugs through the AKT/GSK-3β pathway. Oncol Rep. 44:1905–1916. 2020.PubMed/NCBI
|
|
57
|
Chohan MH, Perry M, Laurance-Young P, Salih VM and Foey AD: Prognostic role of CD68+ and CD163+ tumour-associated macrophages and PD-L1 expression in oral squamous cell carcinoma: A meta-analysis. Br J Biomed Sci. 80:110652023. View Article : Google Scholar
|
|
58
|
Alves AM, Diel LF and Lamers ML: Macrophages and prognosis of oral squamous cell carcinoma: A systematic review. J Oral Pathol Med. 47:460–467. 2018. View Article : Google Scholar
|
|
59
|
Masucci MT, Minopoli M and Carriero MV: Tumor associated neutrophils. Their role in tumorigenesis, metastasis, prognosis and therapy. Front Oncol. 9:11462019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Piccard H, Muschel RJ and Opdenakker G: On the dual roles and polarized phenotypes of neutrophils in tumor development and progression. Crit Rev Oncol Hematol. 82:296–309. 2012. View Article : Google Scholar
|
|
61
|
Fridlender ZG and Albelda SM: Tumor-associated neutrophils: Friend or foe? Carcinogenesis. 33:949–955. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Sagiv JY, Michaeli J, Assi S, Mishalian I, Kisos H, Levy L, Damti P, Lumbroso D, Polyansky L, Sionov RV, et al: Phenotypic diversity and plasticity in circulating neutrophil subpopulations in cancer. Cell Rep. 10:562–573. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Hu X, Xiang F, Feng Y, Gao F, Ge S, Wang C, Zhang X and Wang N: Neutrophils promote tumor progression in oral squamous cell carcinoma by regulating EMT and JAK2/STAT3 signaling through chemerin. Front Oncol. 12:8120442022. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Yu T, Tang Q, Chen X, Fan W, Zhou Z, Huang W and Liang F: TGF-β1 and IL-17A comediate the protumor phenotype of neutrophils to regulate the epithelial-mesenchymal transition in oral squamous cell carcinoma. J Oral Pathol Med. 50:353–361. 2021. View Article : Google Scholar
|
|
65
|
Garley M: Unobvious neutrophil extracellular traps signification in the course of oral squamous cell carcinoma: Current understanding and future perspectives. Cancer Control. 30:107327482311593132023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Garley M, Dziemiańczyk-Pakieła D, Grubczak K, Surażyński A, Dąbrowska D, Ratajczak-Wrona W, Sawicka-Powierza J, Borys J, Moniuszko M, Pałka JA and Jabłońska E: Differences and similarities in the phenomenon of NETs formation in oral inflammation and in oral squamous cell carcinoma. J Cancer. 9:1958–1965. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Garley M, Dziemiańczyk-Pakieła D, Ratajczak-Wrona W, Pryczynicz A, Nowak K, Łazarczyk B and Jabłońska E: NETs biomarkers in saliva and serum OSCC patients: One hypothesis, two conclusions. Adv Med Sci. 67:45–54. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Zhai R, Gong Z, Wang M, Ni Z, Zhang J, Wang M, Zhang Y, Zeng F, Gu Z, Chen X, et al: Neutrophil extracellular traps promote invasion and metastasis via NLRP3-mediated oral squamous cell carcinoma pyroptosis inhibition. Cell Death Discov. 10:2142024. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Zhou J, Hu Z, Wang L, Hu Q, Chen Z, Lin T, Zhou R, Cai Y, Wu Z, Zhang Z, et al: Tumor-colonized Streptococcus mutans metabolically reprograms tumor microenvironment and promotes oral squamous cell carcinoma. Microbiome. 12:1932024. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Li C, Zhao L, Wang Q, Ma S, Sun J, Ma C, Liu J, Jing X, Ai D, Nan Z and Qu X: Neutrophils infiltration and its correlation with human papillomavirus status in the oral squamous cell carcinoma. Cancer Manag Res. 11:5171–5185. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Phulari RGS, Rathore RS, Shah AK and Agnani SS: Neutrophil: Lymphocyte ratio and oral squamous cell carcinoma: A preliminary study. J Oral Maxillofac Pathol. 23:78–81. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Mishra V, Giri R, Hota S, Senapati U and Sahu SK: Neutrophil-to-lymphocyte ratio as a prognostic factor in oral squamous cell carcinoma-a single-institutional experience from a developing country. J Oral Maxillofac Pathol. 25:322–326. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Lichterman JN and Reddy SM: Mast cells: A new frontier for cancer immunotherapy. Cells. 10:12702021. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Cai XJ, Peng CR, Zhang JY, Li XF, Wang X, Han Y, Zhang HY, Peng X and Li TJ: Mast cell infiltration and subtype promote malignant transformation of oral precancer and progression of oral cancer. Cancer Res Commun. 4:2203–2214. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Teófilo CR, Ferreira AEC Junior, Batista AC, Fechini Jamacaru FV, Sousa FB, Lima Mota MR, Silva MFE, Barros Silva PG and Alves APNN: Mast cells and blood vessels profile in oral carcinogenesis: An immunohistochemistry study. Asian Pac J Cancer Prev. 21:1097–1102. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Hasan NR, Dehuri P, Jena A and Kanungo S: A correlation of serum histamine and mast cell count with the established prognosticators in oral cancer. J Microsc Ultrastruct. 11:97–102. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Hemmerlein B, Reinhardt L, Wiechens B, Khromov T, Schliephake H and Brockmeyer P: Is CCL2 an important mediator of mast cell-tumor cell interactions in oral squamous cell carcinoma? Int J Mol Sci. 24:36412023. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Tzorakoleftheraki SE and Koletsa T: The complex role of mast cells in head and neck squamous cell carcinoma: A systematic review. Medicina (Kaunas). 60:11732024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Shrestha A, Keshwar S and Raut T: Evaluation of mast cells in oral potentially malignant disorders and oral squamous cell carcinoma. Int J Dent. 2021:56095632021. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Kamal R, Dahiya P, Goyal N, Kumar M, Sharma N and Saini HR: Mast cells and oral pathologies: A review. J Nat Sci Biol Med. 6:35–39. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Salem A and Salo T: Nothing to sneeze at: Histamine and histamine receptors in oral carcinogenesis. Oral Dis. 27:1090–1096. 2021. View Article : Google Scholar
|
|
82
|
Oliveira-Neto HH, Leite AF, Costa NL, Alencar RC, Lara VS, Silva TA, Leles CR, Mendonça FE and Batista AC: Decrease in mast cells in oral squamous cell carcinoma: Possible failure in the migration of these cells. Oral Oncol. 43:484–490. 2007. View Article : Google Scholar
|
|
83
|
Iamaroon A, Pongsiriwet S, Jittidecharaks S, Pattanaporn K, Prapayasatok S and Wanachantararak S: Increase of mast cells and tumor angiogenesis in oral squamous cell carcinoma. J Oral Pathol Med. 32:195–199. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Michailidou EZ, Markopoulos AK and Antoniades DZ: Mast cells and angiogenesis in oral malignant and premalignant lesions. Open Dent J. 2:126–132. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Salem A, Almahmoudi R, Listyarifah D, Siponen M, Maaninka K, Al-Samadi A, Salo T and Eklund KK: Histamine H4 receptor signalling in tongue cancer and its potential role in oral carcinogenesis-a short report. Cell Oncol (Dordr). 40:621–630. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gaje PN, Amalia Ceausu R, Jitariu A, Stratul SI, Rusu LC, Popovici RA and Raica M: Mast cells: Key players in the shadow in oral inflammation and in squamous cell carcinoma of the oral cavity. Biomed Res Int. 2016:92350802016. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Ansari FM, Asif M, Kiani MN, Ara N, Ishaque M and Khan R: Evaluation of mast cell density using CD117 antibody and microvessel density using CD34 antibody in different grades of oral squamous cell carcinoma. Asian Pac J Cancer Prev. 21:3533–3538. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Kim JY, Moon S and Kim D: Oral squamous cell carcinoma-derived ANGPTL3 induces cancer associated fibroblastic phenotypes in surrounding fibroblasts. Asian Pac J Cancer Prev. 23:4315–4322. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Ren X, Li L, Wu J, Lin K, He Y and Bian L: PDGF-BB regulates the transformation of fibroblasts into cancer-associated fibroblasts via the lncRNA LURAP1L-AS1/LURAP1L/IKK/IκB/NF-κB signaling pathway. Oncol Lett. 22:5372021. View Article : Google Scholar
|
|
90
|
Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, Fearon D, Greten FR, Hingorani SR, Hunter T, et al: A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer. 20:174–186. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Bienkowska KJ, Hanley CJ and Thomas GJ: Cancer-associated fibroblasts in oral cancer: A current perspective on function and potential for therapeutic targeting. Front Oral Health. 2:6863372021. View Article : Google Scholar
|
|
92
|
Chaudhary M, Gadbail AR, Vidhale G, Mankar Gadbail MP, Gondivkar SM, Gawande M and Patil S: Comparison of myofibroblasts expression in oral squamous cell carcinoma, verrucous carcinoma, high risk epithelial dysplasia, low risk epithelial dysplasia and normal oral mucosa. Head Neck Pathol. 6:305–313. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Zhang X and Hwang YS: Cancer-associated fibroblast stimulates cancer cell invasion in an interleukin-1 receptor (IL-1R)-dependent manner. Oncol Lett. 18:4645–4650. 2019.PubMed/NCBI
|
|
94
|
Sobral LM, Bufalino A, Lopes MA, Graner E, Salo T and Coletta RD: Myofibroblasts in the stroma of oral cancer promote tumorigenesis via secretion of activin A. Oral Oncol. 47:840–846. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Knowles LM, Stabile LP, Egloff AM, Rothstein ME, Thomas SM, Gubish CT, Lerner EC, Seethala RR, Suzuki S, Quesnelle KM, et al: HGF and c-Met participate in paracrine tumorigenic pathways in head and neck squamous cell cancer. Clin Cancer Res. 15:3740–3750. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Wang Y, Jing Y, Ding L, Zhang X, Song Y, Chen S, Zhao X, Huang X, Pu Y, Wang Z, et al: Epiregulin reprograms cancer-associated fibroblasts and facilitates oral squamous cell carcinoma invasion via JAK2-STAT3 pathway. J Exp Clin Cancer Res. 38:2742019. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Ferreira Mendes JM, de Faro Valverde L, Torres Andion Vidal M, Paredes BD, Coelho P, Allahdadi KJ, Coletta RD, Souza BSF and Rocha CAG: Effects of IGF-1 on proliferation, angiogenesis, tumor stem cell populations and activation of AKT and hedgehog pathways in oral squamous cell carcinoma. Int J Mol Sci. 21:64872020. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Yang W, Zhang S, Li T, Zhou Z and Pan J: Single-cell analysis reveals that cancer-associated fibroblasts stimulate oral squamous cell carcinoma invasion via the TGF-β/Smad pathway. Acta Biochim Biophys Sin (Shanghai). 55:262–273. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Pavlides S, Whitaker-Menezes D, Castello-Cros R, Flomenberg N, Witkiewicz AK, Frank PG, Casimiro MC, Wang C, Fortina P, Addya S, et al: The reverse Warburg effect: Aerobic glycolysis in cancer associated fibroblasts and the tumor stroma. Cell Cycle. 8:3984–4001. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Zhang Z, Gao Z, Rajthala S, Sapkota D, Dongre H, Parajuli H, Suliman S, Das R, Li L, Bindoff LA, et al: Metabolic reprogramming of normal oral fibroblasts correlated with increased glycolytic metabolism of oral squamous cell carcinoma and precedes their activation into carcinoma associated fibroblasts. Cell Mol Life Sci. 77:1115–1133. 2020. View Article : Google Scholar
|
|
101
|
Li X, Jiang E, Zhao H, Chen Y, Xu Y, Feng C, Li J and Shang Z: Glycometabolic reprogramming-mediated proangiogenic phenotype enhancement of cancer-associated fibroblasts in oral squamous cell carcinoma: Role of PGC-1α/PFKFB3 axis. Br J Cancer. 127:449–461. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liu P, Wang Y, Li X, Liu Z, Sun Y, Liu H, Shao Z, Jiang E, Zhou X and Shang Z: Enhanced lipid biosynthesis in oral squamous cell carcinoma cancer-associated fibroblasts contributes to tumor progression: Role of IL8/AKT/p-ACLY axis. Cancer Sci. 115:1433–1445. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Zhang Z, Tao D, Zhang P, Liu X, Zhang Y, Cheng J, Yuan H, Liu L and Jiang H: Hyaluronan synthase 2 expressed by cancer-associated fibroblasts promotes oral cancer invasion. J Exp Clin Cancer Res. 35:1812016. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Matos LL, Menderico GM Junior, Theodoro TR, Pasini FS, Ishikawa MM, Ribeiro AAB, de Mello ES, Pinhal MADS, Moyses RA, Kulcsar MAV, et al: Cancer-associated fibroblast regulation by microRNAs promotes invasion of oral squamous cell carcinoma. Oral Oncol. 110:1049092020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Zhang JY, Zhu WW, Wang MY, Zhai RD, Wang Q, Shen WL and Liu LK: Cancer-associated fibroblasts promote oral squamous cell carcinoma progression through LOX-mediated matrix stiffness. J Transl Med. 19:5132021. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Liu X, Li J, Yang X, Li X, Kong J, Qi D, Zhang F, Sun B, Liu Y and Liu T: Carcinoma-associated fibroblast-derived lysyl oxidase-rich extracellular vesicles mediate collagen crosslinking and promote epithelial-mesenchymal transition via p-FAK/p-paxillin/YAP signaling. Int J Oral Sci. 15:322023. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Elmusrati AA, Pilborough AE, Khurram SA and Lambert DW: Cancer-associated fibroblasts promote bone invasion in oral squamous cell carcinoma. Br J Cancer. 117:867–875. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Evanko SP, Potter-Perigo S, Bollyky PL, Nepom GT and Wight TN: Hyaluronan and versican in the control of human T-lymphocyte adhesion and migration. Matrix Biol. 31:90–100. 2012. View Article : Google Scholar
|
|
109
|
Takahashi H, Sakakura K, Kudo T, Toyoda M, Kaira K, Oyama T and Chikamatsu K: Cancer-associated fibroblasts promote an immunosuppressive microenvironment through the induction and accumulation of protumoral macrophages. Oncotarget. 8:8633–8647. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Hu S, Lu H, Xie W, Wang D, Shan Z, Xing X, Wang XM, Fang J, Dong W, Dai W, et al: TDO2+ myofibroblasts mediate immune suppression in malignant transformation of squamous cell carcinoma. J Clin Invest. 132:e1576492022. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Peltanova B, Liskova M, Gumulec J, Raudenska M, Polanska HH, Vaculovic T, Kalfert D, Grega M, Plzak J, Betka J and Masarik M: Sensitivity to cisplatin in head and neck cancer cells is significantly affected by patient-derived cancer-associated fibroblasts. Int J Mol Sci. 22:19122021. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Tao L, Huang G, Wang R, Pan Y, He Z, Chu X, Song H and Chen L: Cancer-associated fibroblasts treated with cisplatin facilitates chemoresistance of lung adenocarcinoma through IL-11/IL-11R/STAT3 signaling pathway. Sci Rep. 6:384082016. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Kang SH, Oh SY, Lee KY, Lee HJ, Kim MS, Kwon TG, Kim JW, Lee ST, Choi SY and Hong SH: Differential effect of cancer-associated fibroblast-derived extracellular vesicles on cisplatin resistance in oral squamous cell carcinoma via miR-876-3p. Theranostics. 14:460–479. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Graizel D, Zlotogorski-Hurvitz A, Tsesis I, Rosen E, Kedem R and Vered M: Oral cancer-associated fibroblasts predict poor survival: Systematic review and meta-analysis. Oral Dis. 26:733–744. 2020. View Article : Google Scholar
|
|
115
|
Li Y, Ma Z, Li W, Xu X, Shen P, Zhang SE, Cheng B and Xia J: PDPN+ CAFs facilitate the motility of OSCC cells by inhibiting ferroptosis via transferring exosomal lncRNA FTX. Cell Death Dis. 14:7592023. View Article : Google Scholar
|
|
116
|
Maman S and Witz IP: A history of exploring cancer in context. Nat Rev Cancer. 18:359–376. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Giraldo NA, Sanchez-Salas R, Peske JD, Vano Y, Becht E, Petitprez F, Validire P, Ingels A, Cathelineau X, Fridman WH and Sautès-Fridman C: The clinical role of the TME in solid cancer. Br J Cancer. 120:45–53. 2019. View Article : Google Scholar :
|
|
118
|
Harding FA, McArthur JG, Gross JA, Raulet DH and Allison JP: CD28-mediated signalling co-stimulates murine T cells and prevents induction of anergy in T-cell clones. Nature. 356:607–609. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Tivol EA, Borriello F, Schweitzer AN, Lynch WP, Bluestone JA and Sharpe AH: Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 3:541–547. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Keir ME, Butte MJ, Freeman GJ and Sharpe AH: PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol. 26:677–704. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Ferris RL: Immunology and immunotherapy of head and neck cancer. J Clin Oncol. 33:3293–3304. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Hu Y, Lin L, Chen J, Hao K, Zhang S, Guo X, Guo Z, Tian H and Chen X: Highly enhanced antitumor immunity by a three-barreled strategy of the l-arginine-promoted nanovaccine and gene-mediated PD-L1 blockade. ACS Appl Mater Interfaces. 12:41127–41137. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ, Topalian SL, Hwu P, Drake CG, Camacho LH, Kauh J, Odunsi K, et al: Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 366:2455–2465. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Qiao XW, Jiang J, Pang X, Huang MC, Tang YJ, Liang XH and Tang YL: The evolving landscape of PD-1/PD-L1 pathway in head and neck cancer. Front Immunol. 11:17212020. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Paolino G, Pantanowitz L, Barresi V, Pagni F, Munari E, Moretta L, Brunelli M, Bariani E, Vigliar E, Pisapia P, et al: PD-L1 evaluation in head and neck squamous cell carcinoma: Insights regarding specimens, heterogeneity and therapy. Pathol Res Pract. 226:1536052021. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Girolami I, Pantanowitz L, Barberis M, Paolino G, Brunelli M, Vigliar E, Munari E, Satturwar S, Troncone G and Eccher A: Challenges facing pathologists evaluating PD-L1 in head & neck squamous cell carcinoma. J Oral Pathol Med. 50:864–873. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Almangush A, Leivo I and Mäkitie AA: Overall assessment of tumor-infiltrating lymphocytes in head and neck squamous cell carcinoma: Time to take notice. Acta Otolaryngol. 140:246–248. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Cramer JD, Burtness B and Ferris RL: Immunotherapy for head and neck cancer: Recent advances and future directions. Oral Oncol. 99:1044602019. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Moskovitz JM and Ferris RL: Tumor immunology and immunotherapy for head and neck squamous cell carcinoma. J Dent Res. 97:622–626. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Zandberg DP, Algazi AP, Jimeno A, Good JS, Fayette J, Bouganim N, Ready NE, Clement PM, Even C, Jang RW, et al: Durvalumab for recurrent or metastatic head and neck squamous cell carcinoma: Results from a single-arm, phase II study in patients with ≥25% tumour cell PD-L1 expression who have progressed on platinum-based chemotherapy. Eur J Cancer. 107:142–152. 2019. View Article : Google Scholar
|
|
131
|
Rebelatto MC, Midha A, Mistry A, Sabalos C, Schechter N, Li X, Jin X, Steele KE, Robbins PB, Blake-Haskins JA and Walker J: Development of a programmed cell death ligand-1 immunohistochemical assay validated for analysis of non-small cell lung cancer and head and neck squamous cell carcinoma. Diagn Pathol. 11:952016. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Chow LQM, Haddad R, Gupta S, Mahipal A, Mehra R, Tahara M, Berger R, Eder JP, Burtness B, Lee SH, et al: Antitumor activity of pembrolizumab in biomarker-unselected patients with recurrent and/or metastatic head and neck squamous cell carcinoma: Results from the phase Ib KEYNOTE-012 expansion cohort. J Clin Oncol. 34:3838–3845. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Burtness B, Harrington KJ, Greil R, Soulières D, Tahara M, de Castro G Jr, Psyrri A, Basté N, Neupane P, Bratland Å, et al: Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): A randomised, open-label, phase 3 study. Lancet. 394:1915–1928. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Ferris RL, Blumenschein G Jr, Fayette J, Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE, Even C, et al: Nivolumab for recurrent squamous-cell carcinoma of the head and neck. N Engl J Med. 375:1856–1867. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Harrington KJ, Burtness B, Greil R, Soulières D, Tahara M, de Castro G Jr, Psyrri A, Brana I, Basté N, Neupane P, et al: Pembrolizumab with or without chemotherapy in recurrent or metastatic head and neck squamous cell carcinoma: Updated results of the phase III KEYNOTE-048 study. J Clin Oncol. 41:790–802. 2023. View Article : Google Scholar :
|
|
136
|
Ferris RL, Blumenschein G Jr, Fayette J, Guigay J, Colevas AD, Licitra L, Harrington KJ, Kasper S, Vokes EE, Even C, et al: Nivolumab vs investigator's choice in recurrent or metastatic squamous cell carcinoma of the head and neck: 2-Year long-term survival update of CheckMate 141 with analyses by tumor PD-L1 expression. Oral Oncol. 81:45–51. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Uppaluri R, Campbell KM, Egloff AM, Zolkind P, Skidmore ZL, Nussenbaum B, Paniello RC, Rich JT, Jackson R, Pipkorn P, et al: Neoadjuvant and adjuvant pembrolizumab in resectable locally advanced, human papillomavirus-unrelated head and neck cancer: A multicenter, phase II trial. Clin Cancer Res. 26:5140–5152. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Wise-Draper TM, Gulati S, Palackdharry S, Hinrichs BH, Worden FP, Old MO, Dunlap NE, Kaczmar JM, Patil Y, Riaz MK, et al: Phase II clinical trial of neoadjuvant and adjuvant pembrolizumab in resectable local-regionally advanced head and neck squamous cell carcinoma. Clin Cancer Res. 28:1345–1352. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Uppaluri R, Lee NY, Westra W, Cohen EE, Haddad RI, Temam S, Le Tourneau C, Chernock R, Safina S, Klochikhin A, et al: KEYNOTE-689: Phase 3 study of adjuvant and neoadjuvant pembrolizumab combined with standard of care (SOC) in patients with resectable, locally advanced head and neck squamous cell carcinoma. J Clin Oncol. 37(15 Suppl): TPS60902019. View Article : Google Scholar
|
|
140
|
Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, Yokoi T, Chiappori A, Lee KH, de Wit M, et al: Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 377:1919–1929. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Kelly RJ, Ajani JA, Kuzdzal J, Zander T, Van Cutsem E, Piessen G, Mendez G, Feliciano J, Motoyama S, Lièvre A, et al: Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl J Med. 384:1191–1203. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Wong W, Cracchiolo JR, Riaz N, Ganly I, Sherman EJ, Ho AL, Morris L, Ghossein RA, Haque S, Hung KW, et al: Neoadjuvant cemiplimab with platinum-doublet chemotherapy and cetuximab to de-escalate surgery and omit adjuvant radiation in locoregionally advanced head & neck squamous cell carcinoma (HNSCC). J Clin Oncol. 41(16 Suppl): S60192023. View Article : Google Scholar
|
|
143
|
Wu D, Li Y, Xu P, Fang Q, Cao F, Lin H, Li Y, Su Y, Lu L, Chen L, et al: Neoadjuvant chemo-immunotherapy with camrelizumab plus nab-paclitaxel and cisplatin in resectable locally advanced squamous cell carcinoma of the head and neck: A pilot phase II trial. Nat Commun. 15:21772024. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Yu Y and Lee NY: JAVELIN Head and Neck 100: A Phase III trial of avelumab and chemoradiation for locally advanced head and neck cancer. Future Oncol. 15:687–694. 2019. View Article : Google Scholar
|
|
145
|
Wong DJ, Fayette J, Teixeira M, Prabhash K, Mesia R, Kawecki A, Dechaphunkul A, Dinis J, Guo Y, Masuda M, et al: Abstract CT009: IMvoke010: A phase III, double-blind randomized trial of atezolizumab (atezo) after definitive local therapy vs placebo in patients (pts) with high-risk locally advanced (LA) squamous cell carcinoma of the head and neck (SCCHN). Cancer Res. 84(7 Suppl): CT0092024. View Article : Google Scholar
|
|
146
|
Duhen R, Ballesteros-Merino C, Frye AK, Tran E, Rajamanickam V, Chang SC, Koguchi Y, Bifulco CB, Bernard B, Leidner RS, et al: Neoadjuvant anti-OX40 (MEDI6469) therapy in patients with head and neck squamous cell carcinoma activates and expands antigen-specific tumor-infiltrating T cells. Nat Commun. 12:10472021. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Shrikant P, Khoruts A and Mescher MF: CTLA-4 blockade reverses CD8+ T cell tolerance to tumor by a CD4+ T cell- and IL-2-dependent mechanism. Immunity. 11:483–493. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Lee WS, Ye Z, Cheung AMS, Goh YPS, Oh HLJ, Rajarethinam R, Yeo SP, Soh MK, Chan EHL, Tan LK, et al: Effective killing of acute myeloid leukemia by TIM-3 targeted chimeric antigen receptor T cells. Mol Cancer Ther. 20:1702–1712. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Burova E, Hermann A, Dai J, Ullman E, Halasz G, Potocky T, Hong S, Liu M, Allbritton O, Woodruff A, et al: Preclinical development of the anti-LAG-3 antibody REGN3767: Characterization and activity in combination with the anti-PD-1 antibody cemiplimab in human PD-1xLAG-3-knockin mice. Mol Cancer Ther. 18:2051–2062. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Huang J, Barbera L, Brouwers M, Browman G and Mackillop WJ: Does delay in starting treatment affect the outcomes of radiotherapy? A systematic review. J Clin Oncol. 21:555–563. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Maeda S, Murakami K, Inoue A, Yonezawa T and Matsuki N: CCR4 blockade depletes regulatory T cells and prolongs survival in a canine model of bladder cancer. Cancer Immunol Res. 7:1175–1187. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Wolf GT, Liu S, Bellile E, Sartor M, Rozek L, Thomas D, Nguyen A, Zarins K and McHugh JB; INSPIRE Trial Clinical Investigators: Tumor infiltrating lymphocytes after neoadjuvant IRX-2 immunotherapy in oral squamous cell carcinoma: Interim findings from the INSPIRE trial. Oral Oncol. 111:1049282020. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Jiang T, Zhou C and Ren S: Role of IL-2 in cancer immunotherapy. Oncoimmunology. 5:e11634622016. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Tarhini AA, Gogas H and Kirkwood JM: IFN-α in the treatment of melanoma. J Immunol. 189:3789–3793. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Chien MH, Hsin CH, Chou LS, Chung TT, Lin CH, Weng MS, Chou MY, Chen MK and Yang SF: Interleukin-23 receptor polymorphism as a risk factor for oral cancer susceptibility. Head Neck. 34:551–556. 2012. View Article : Google Scholar
|
|
156
|
Tian S, Jiang C, Liu X, Xu S, Zhang Z, Chen H, Zhang Y, Liu Y and Ma D: Hypermethylation of IFN-γ in oral cancer tissues. Clin Oral Investig. 21:2535–2542. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Kong AH, Hesselink MSK, Aguilera B, Adkins D, Even C, Fayette J, Muzaffar J, Visscher S, Melief CJM and Hooftman LW: Phase 2 study of ISA101b (peltopepimut-S) and cemiplimab in patients with advanced HPV16+ oropharyngeal cancer who failed anti-PD1 therapy. J Clin Oncol. 41(16 Suppl): S60282023. View Article : Google Scholar
|















