|
1
|
Meng Y, Liu Y, Duan R, Liu B, Lin Z, Ma Y,
Jiang L, Qin Z and Li T: Global, regional, and national
epidemiology of vision impairment due to diabetic retinopathy among
working-age population, 1990-2021. J Diabetes. 17:e701212025.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen Y, Tang S, Huang Y, Deng J, Chen X,
Qi Y, Xiao H, Li Y, Li H and Guan H: Global burden of blindness or
visually impairment attributable to diabetic retinopathy in the
adults aged 70 years and older, 1990-2021: Results from the global
burden of disease study in 2021. Diabetes Res Clin Pract.
226:1123832025. View Article : Google Scholar
|
|
3
|
Antonetti DA, Silva PS and Stitt AW:
Current understanding of the molecular and cellular pathology of
diabetic retinopathy. Nat Rev Endocrinol. 17:195–206. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tang Y, Shi Y and Fan Z: The mechanism and
therapeutic strategies for neovascular glaucoma secondary to
diabetic retinopathy. Front Endocrinol (Lausanne). 14:11023612023.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yue T, Shi Y, Luo S, Weng J, Wu Y and
Zheng X: The role of inflammation in immune system of diabetic
retinopathy: Molecular mechanisms, pathogenetic role and
therapeutic implications. Front Immunol. 13:10550872022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kuroiwa DAK, Malerbi FK and Regatieri CVS:
New insights in resistant diabetic macular edema. Ophthalmologica.
244:485–494. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tatsumi T: Current treatments for diabetic
macular edema. Int J Mol Sci. 24:95912023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sweeney MD, Ayyadurai S and Zlokovic BV:
Pericytes of the neurovascular unit: Key functions and signaling
pathways. Nat Neurosci. 19:771–783. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhou L, Xu Z, Lu H, Cho H, Xie Y, Lee G,
Ri K and Duh EJ: Suppression of inner blood-retinal barrier
breakdown and pathogenic Müller glia activation in ischemia
retinopathy by myeloid cell depletion. J Neuroinflammation.
21:2102024. View Article : Google Scholar
|
|
10
|
Bora K, Kushwah N, Maurya M, Pavlovich MC,
Wang Z and Chen J: Assessment of inner blood-retinal barrier:
Animal models and methods. Cells. 12:24432023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
O'Leary F and Campbell M: The blood-retina
barrier in health and disease. FEBS J. 290:878–891. 2023.
View Article : Google Scholar
|
|
12
|
Klaassen I, Van Noorden CJF and
Schlingemann RO: Molecular basis of the inner blood-retinal barrier
and its breakdown in diabetic macular edema and other pathological
conditions. Prog Retin Eye Res. 34:19–48. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fletcher EL, Dixon MA, Mills SA and
Jobling AI: Anomalies in neurovascular coupling during early
diabetes: A review. Clin Exp Ophthalmol. 51:81–91. 2023. View Article : Google Scholar
|
|
14
|
Kugler EC, Greenwood J and MacDonald RB:
The 'neuro-glial-vascular' unit: The role of glia in neurovascular
unit formation and dysfunction. Front Cell Dev Biol. 9:7328202021.
View Article : Google Scholar
|
|
15
|
Grimes WN, Berson DM, Sabnis A, Hoon M,
Sinha R, Tian H and Diamond JS: Layer-specific anatomical and
physiological features of the retina's neurovascular unit. Curr
Biol. 35:109–120.e4. 2025. View Article : Google Scholar :
|
|
16
|
Ivanova E, Alam NM, Prusky GT and
Sagdullaev BT: Blood-retina barrier failure and vision loss in
neuron-specific degeneration. JCI Insight. 5:e1267472019.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sheng X, Zhang C, Zhao J, Xu J, Zhang P,
Ding Q and Zhang J: Microvascular destabilization and intricated
network of the cytokines in diabetic retinopathy: From the
perspective of cellular and molecular components. Cell Biosci.
14:852024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rudraraju M, Narayanan SP and Somanath PR:
Regulation of blood-retinal barrier cell-junctions in diabetic
retinopathy. Pharmacol Res. 161:1051152020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Daneman R, Zhou L, Kebede AA and Barres
BA: Pericytes are required for blood-brain barrier integrity during
embryogenesis. Nature. 468:562–566. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Resnikoff HA, Miller CG and Schwarzbauer
JE: Implications of fibrotic extracellular matrix in diabetic
retinopathy. Exp Biol Med (Maywood). 247:1093–1102. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Park DY, Lee J, Kim J, Kim K, Hong S, Han
S, Kubota Y, Augustin HG, Ding L, Kim JW, et al: Plastic roles of
pericytes in the blood-retinal barrier. Nat Commun. 8:152962017.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Leonardo ED, Hinck L, Masu M, Keino-Masu
K, Ackerman SL and Tessier-Lavigne M: Vertebrate homologues of C.
elegans UNC-5 are candidate netrin receptors. Nature. 386:833–838.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Serafini T, Colamarino SA, Leonardo ED,
Wang H, Beddington R, Skarnes WC and Tessier-Lavigne M: Netrin-1 is
required for commissural axon guidance in the developing vertebrate
nervous system. Cell. 87:1001–1014. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Navankasattusas S, Whitehead KJ, Suli A,
Sorensen LK, Lim AH, Zhao J, Park KW, Wythe JD, Thomas KR, Chien CB
and Li D: The netrin receptor UNC5B promotes angiogenesis in
specific vascular beds. Development. 135:659–667. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Larrivée B, Freitas C, Trombe M, Lv X,
Delafarge B, Yuan L, Bouvrée K, Bréant C, Del Toro R, Bréchot N, et
al: Activation of the UNC5B receptor by netrin-1 inhibits sprouting
angiogenesis. Genes Dev. 21:2433–2447. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tu T, Zhang C, Yan H, Luo Y, Kong R, Wen
P, Ye Z, Chen J, Feng J, Liu F, et al: CD146 acts as a novel
receptor for netrin-1 in promoting angiogenesis and vascular
development. Cell Res. 25:275–287. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yu Y, Zou J, Han Y, Quyang L, He H, Hu P,
Shao Y and Tu P: Effects of intravitreal injection of netrin-1 in
retinal neovascularization of streptozotocin-induced diabetic rats.
Drug Des Devel Ther. 9:6363–6377. 2015.PubMed/NCBI
|
|
28
|
Yang Y, Zou L, Wang Y, Xu KS, Zhang JX and
Zhang JH: Axon guidance cue netrin-1 has dual function in
angiogenesis. Cancer Biol Ther. 6:743–748. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Oshitari T: Neurovascular impairment and
therapeutic strategies in diabetic retinopathy. Int J Environ Res
Public Health. 19:4392021. View Article : Google Scholar
|
|
30
|
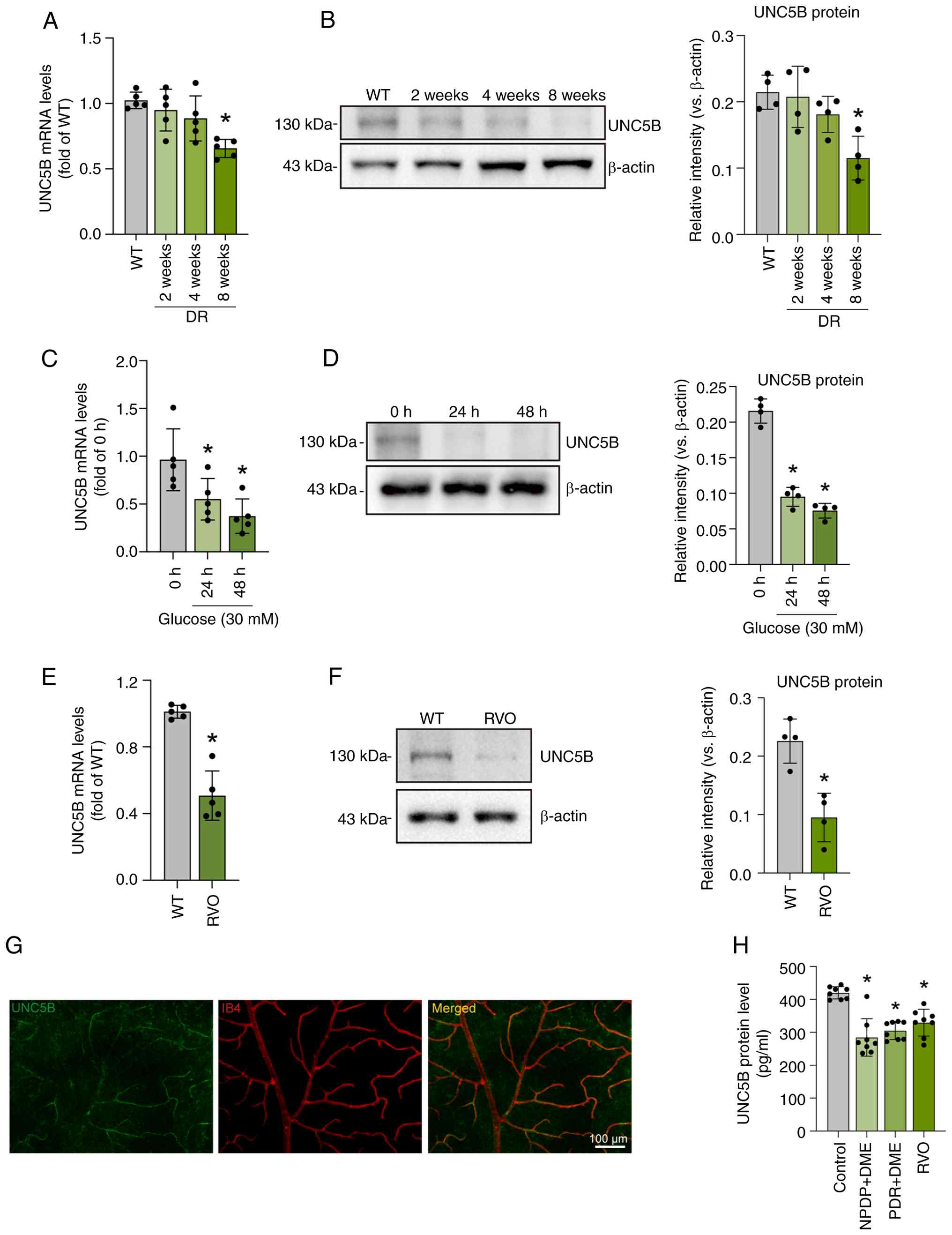
Ma J, Liu X, Qin Y, Qu Y, Liu Q and Li J:
Canagliflozin inhibits NLRP3 inflammasome activation to protect the
retinal neurovascular unit in a mouse retinal vein occlusion model.
Exp Eye Res. 263:1108032026. View Article : Google Scholar
|
|
31
|
Lu X, Le Noble F, Yuan L, Jiang Q, De
Lafarge B, Sugiyama D, Bréant C, Claes F, De Smet F, Thomas JL, et
al: The netrin receptor UNC5B mediates guidance events controlling
morphogenesis of the vascular system. Nature. 432:179–186. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kaur S, Abu-Asab MS, Singla S, Yeo SY and
Ramchandran R: Expression pattern for unc5b, an axon guidance gene
in embryonic zebrafish development. Gene Expr. 13:321–327. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sun L, Wang R, Hu G, Liu H, Lv K, Duan Y,
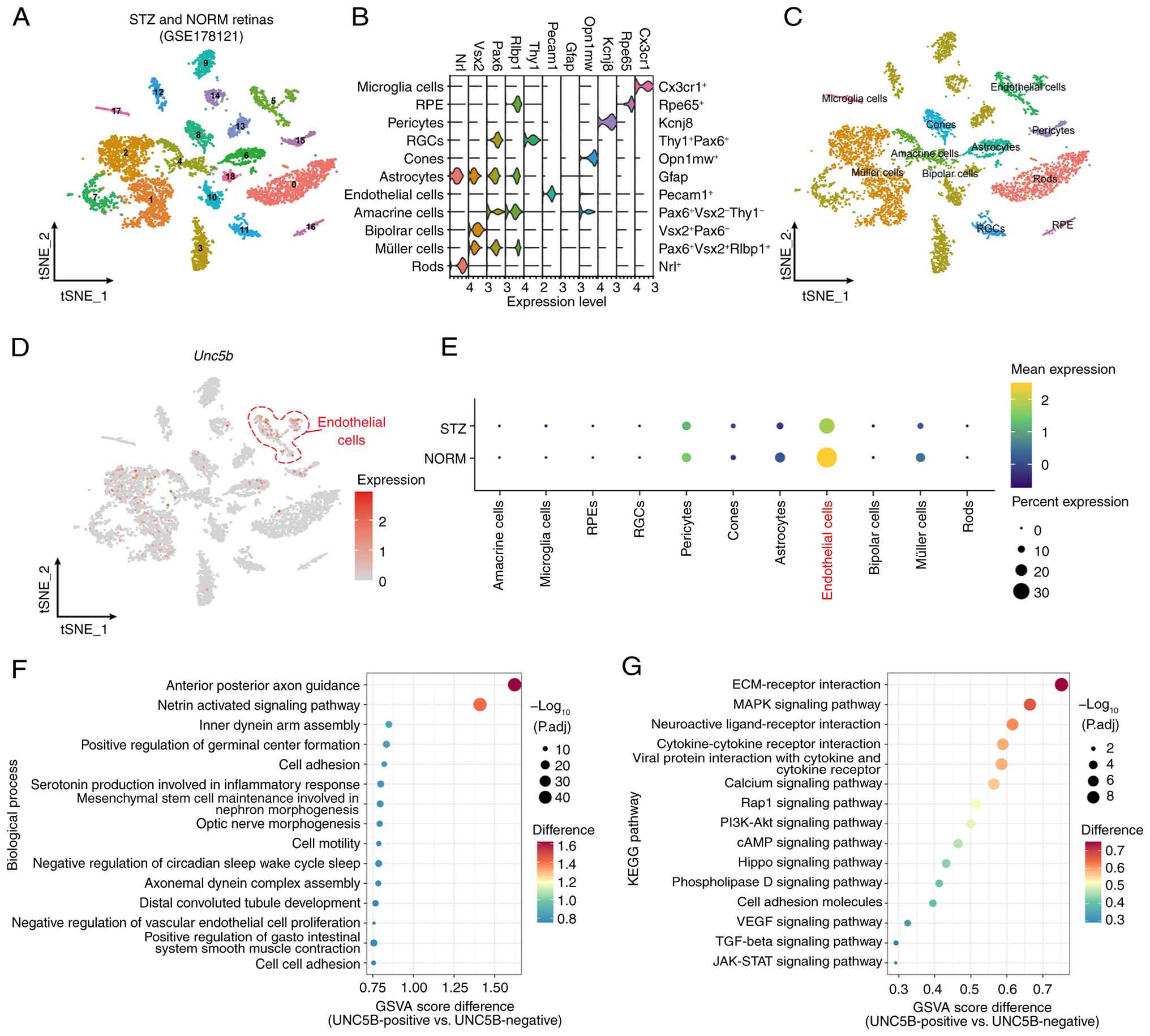
Shen N, Wu J, Hu J, Liu Y, et al: Single cell RNA sequencing
(scRNA-Seq) deciphering pathological alterations in
streptozotocin-induced diabetic retinas. Exp Eye Res.
210:1087182021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Binet F, Cagnone G, Crespo-Garcia S, Hata
M, Neault M, Dejda A, Wilson AM, Buscarlet M, Mawambo GT, Howard
JP, et al: Neutrophil extracellular traps target senescent
vasculature for tissue remodeling in retinopathy. Science.
369:eaay53562020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Siddiqui SS, Siddiqui ZK and Malik AB:
Albumin endocytosis in endothelial cells induces TGF-beta receptor
II signaling. Am J Physiol Lung Cell Mol Physiol. 286:L1016–L1026.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang N, Zhang D, Sun G, Zhang H, You Q,
Shao M and Yue Y: Lipopolysaccharide-induced caveolin-1
phosphorylation-dependent increase in transcellular permeability
precedes the increase in paracellular permeability. Drug Des Devel
Ther. 9:4965–4977. 2015.PubMed/NCBI
|
|
37
|
Adzraku SY, Wang G, Cao C, Bao Y, Wang Y,
Smith AO, Du Y, Wang H, Li Y, Xu K, et al: Robo4 inhibits gamma
radiation-induced permeability of a murine microvascular
endothelial cell by regulating the junctions. Cell Mol Biol Lett.
28:22023. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xia M, Jiao L, Wang XH, Tong M, Yao MD, Li
XM, Yao J, Li D, Zhao PQ and Yan B: Single-cell RNA sequencing
reveals a unique pericyte type associated with capillary
dysfunction. Theranostics. 13:2515–2530. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fresta CG, Fidilio A, Caruso G, Caraci F,
Giblin FJ, Leggio GM, Salomone S, Drago F and Bucolo C: A New human
blood-retinal barrier model based on endothelial cells, pericytes,
and astrocytes. Int J Mol Sci. 21:16362020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
41
|
Chen S, Zhou Y, Chen Y and Gu J: fastp: An
ultra-fast all-in-one FASTQ preprocessor. Bioinformatics.
34:i884–i890. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Oosuka S, Kida T, Oku H, Horie T,
Morishita S, Fukumoto M, Sato T and Ikeda T: Effects of an
aquaporin 4 inhibitor, TGN-020, on murine diabetic retina. Int J
Mol Sci. 21:23242020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chou JC, Rollins SD and Fawzi AA: Trypsin
digest protocol to analyze the retinal vasculature of a mouse
model. J Vis Exp. e504892013.PubMed/NCBI
|
|
44
|
Zhao Y, Ge Z, Guo T, Liu H, Zhou Y, Chen
J, Xu H and Chen Z: Complement C3/C3aR signaling pathway inhibition
ameliorates retinal damage in experimental retinal vein occlusion.
Invest Ophthalmol Vis Sci. 66:22025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Avrutsky MI, Chen CW, Lawson JM, Snipas
SJ, Salvesen GS and Troy CM: Caspase-9 inhibition confers stronger
neuronal and vascular protection compared to VEGF neutralization in
a mouse model of retinal vein occlusion. Front Neurosci.
17:12095272023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Minshall RD, Sessa WC, Stan RV, Anderson
RG and Malik AB: Caveolin regulation of endothelial function. Am J
Physiol Lung Cell Mol Physiol. 285:L1179–L1183. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Avrutsky MI, Ortiz CC, Johnson KV,
Potenski AM, Chen CW, Lawson JM, White AJ, Yuen SK, Morales FN,
Canepa E, et al: Endothelial activation of caspase-9 promotes
neurovascular injury in retinal vein occlusion. Nat Commun.
11:31732020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wei L, Gao J, Wang L, Tao Q and Tu C:
Hippo/YAP signaling pathway: A new therapeutic target for diabetes
mellitus and vascular complications. Ther Adv Endocrinol Metab.
14:204201882312201342023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lei Y, Liu Q, Chen B, Wu F, Li Y, Dong X,
Ma N, Wu Z, Zhu Y, Wang L, et al: Protein O-GlcNAcylation coupled
to Hippo signaling drives vascular dysfunction in diabetic
retinopathy. Nat Commun. 15:93342024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chaqour B: CCN-Hippo YAP signaling in
vision and its role in neuronal, glial and vascular cell function
and behavior. J Cell Commun Signal. 17:255–262. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ahmad US, Uttagomol J and Wan H: The
regulation of the Hippo pathway by intercellular junction proteins.
Life (Basel). 12:17922022.PubMed/NCBI
|
|
52
|
Park JA and Kwon YG: Hippo-YAP/TAZ
signaling in angiogenesis. BMB Rep. 51:157–162. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Gu Y, Hao S, Liu K, Gao M, Lu B, Sheng F,
Zhang L, Xu Y, Wu D, Han Y, et al: Airborne fine particulate matter
(PM2.5) damages the inner blood-retinal barrier by
inducing inflammation and ferroptosis in retinal vascular
endothelial cells. Sci Total Environ. 838:1565632022. View Article : Google Scholar
|
|
54
|
Haydinger CD, Ferreira LB, Williams KA and
Smith JR: Mechanisms of macular edema. Front Med (Lausanne).
10:11288112023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhou LY, Liu ZG, Sun YQ, Li YZ, Teng ZQ
and Liu CM: Preserving blood-retinal barrier integrity: A path to
retinal ganglion cell protection in glaucoma and traumatic optic
neuropathy. Cell Regen. 14:132025. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Raza Q, Nadeem T, Youn SW, Swaminathan B,
Gupta A, Sargis T, Du J, Cuervo H, Eichmann A, Ackerman SL, et al:
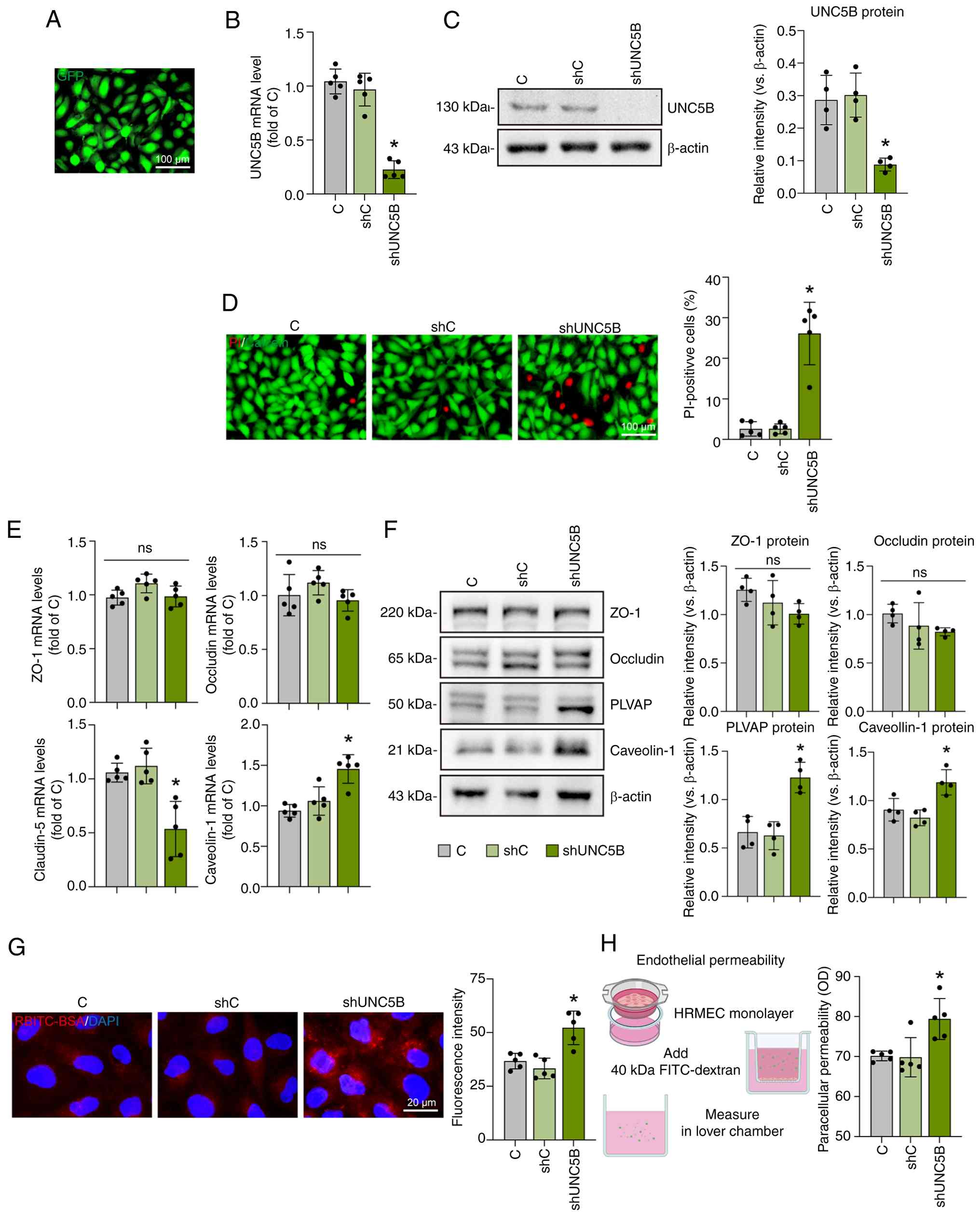
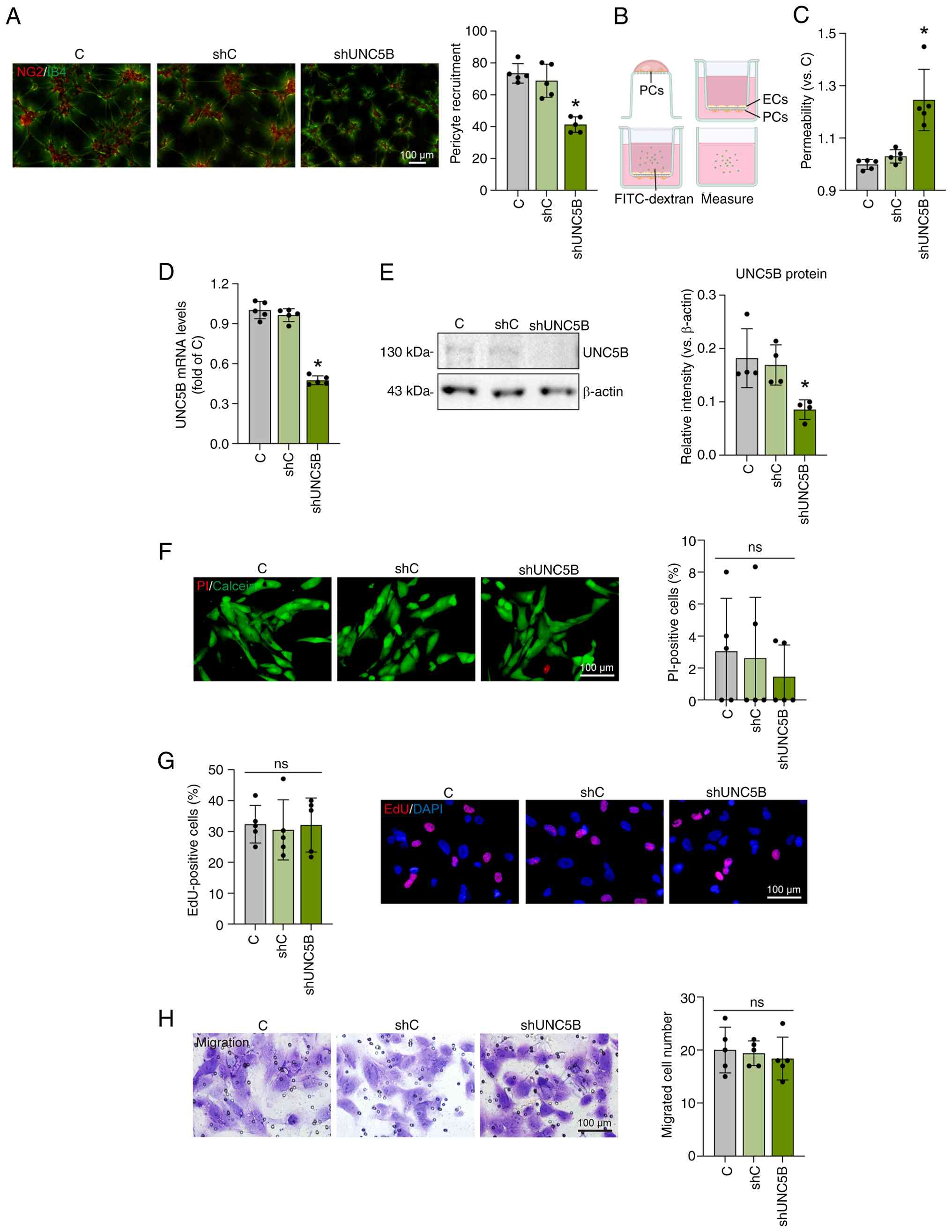
Notch signaling regulates UNC5B to suppress endothelial
proliferation, migration, junction activity, and retinal plexus
branching. Sci Rep. 14:136032024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Boyé K, Geraldo LH, Furtado J,
Pibouin-Fragner L, Poulet M, Kim D, Nelson B, Xu Y, Jacob L, Maissa
N, et al: Endothelial Unc5B controls blood-brain barrier integrity.
Nat Commun. 13:11692022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Furtado J, Geraldo LH, Leser FS, Poulet M,
Park H, Pibouin-Fragner L, Eichmann A and Boyé K: Netrin-1 binding
to Unc5B regulates blood-retina barrier integrity. bioRxiv
[Preprint]: 2023.01.21.525006. 2023.
|
|
59
|
Furtado J, Geraldo LH, Leser FS,
Bartkowiak B, Poulet M, Park H, Robinson M, Pibouin-Fragner L,
Eichmann A and Boyé K: Interplay between netrin-1 and Norrin
controls arteriovenous zonation of blood-retina barrier integrity.
Proc Natl Acad Sci USA. 121:e24086741212024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang CL, Ma JJ, Li X, Yan HQ, Gui YK, Yan
ZX, You MF and Zhang P: The role of transcytosis in the
blood-retina barrier: From pathophysiological functions to drug
delivery. Front Pharmacol. 16:15653822025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Armulik A, Abramsson A and Betsholtz C:
Endothelial/pericyte interactions. Circ Res. 97:512–523. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Nabeel Mustafa A, Salih Mahdi M, Ballal S,
Chahar M, Verma R, Ali Al-Nuaimi AM, Kumar MR, Kadhim A, Al-Hussein
R, Adil M and Jasem Jawad M: Netrin-1: Key insights in neural
development and disorders. Tissue Cell. 93:1026782025. View Article : Google Scholar
|
|
63
|
Li Y, Chai JL, Shi X, Feng Y, Li JJ, Zhou
LN, Cao C and Li KR: Gαi1/3 mediate netrin-1-CD146-activated
signaling and angiogenesis. Theranostics. 13:2319–2336. 2023.
View Article : Google Scholar
|
|
64
|
Roy S and Kim D: Retinal capillary
basement membrane thickening: Role in the pathogenesis of diabetic
retinopathy. Prog Retin Eye Res. 82:1009032021. View Article : Google Scholar
|
|
65
|
Boopathy GTK and Hong W: Role of Hippo
pathway-YAP/TAZ signaling in angiogenesis. Front Cell Dev Biol.
7:492019. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Islam MS, Afrin S, Singh B, Jayes FL,
Brennan JT, Borahay MA, Leppert PC and Segars JH: Extracellular
matrix and Hippo signaling as therapeutic targets of antifibrotic
compounds for uterine fibroids. Clin Transl Med. 11:e4752021.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Lv H and Ai D: Hippo/yes-associated
protein signaling functions as a mechanotransducer in regulating
vascular homeostasis. J Mol Cell Cardiol. 162:158–165. 2022.
View Article : Google Scholar
|
|
68
|
Weiss EL: Hippo unleashed! Proteome-scale
analysis reveals new views of Hippo pathway biology. Sci Signal.
6:pe362013. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Ji L, Tian H, Webster KA and Li W:
Neurovascular regulation in diabetic retinopathy and emerging
therapies. Cell Mol Life Sci. 78:5977–5985. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Meng C, Gu C, He S, Su T, Lhamo T, Draga D
and Qiu Q: Pyroptosis in the retinal neurovascular unit: New
insights into diabetic retinopathy. Front Immunol. 12:7630922021.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Ahn EH, Kang SS, Qi Q, Liu X and Ye K:
Netrin1 deficiency activates MST1 via UNC5B receptor, promoting
dopaminergic apoptosis in Parkinson's disease. Proc Natl Acad Sci
USA. 117:24503–24513. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Qi Q, Li DY, Luo HR, Guan KL and Ye K:
Netrin-1 exerts oncogenic activities through enhancing
Yes-associated protein stability. Proc Natl Acad Sci USA.
112:7255–7260. 2015. View Article : Google Scholar : PubMed/NCBI
|