Primary Sjögren's syndrome (pSS) is a chronic
autoimmune disease characterized by lymphocyte infiltration of
exocrine glands, resulting in glandular dysfunction and
irreversible tissue damage, which may manifest as dry mouth and
eyes (1). If extraductal
involvement occurs, manifestations involving the lungs, joints,
skin, muscles, kidneys and nervous system may appear (1-3).
The pathogenesis of pSS is unclear at present. Scholars have
proposed various pathogenic mechanisms for this disease, including
genetic susceptibility (4),
infection (5), the role of
salivary gland (SG) epithelial cells (6), immune cells [B, T and dendritic
cells (DCs)] (7-9) and the combined action of cytokines
(10). These factors causing
immune disorders jointly lead to pro-inflammatory consequences,
resulting in damage to glands and extraglandular organs. Although
understanding of the pathogenesis of this disease is advancing,
there is currently no specific treatment for pSS. The treatment is
limited to symptom relief. Certain targeted drugs, such as
belimumab and rituximab that target B cells, have been used in the
treatment of pSS, but they are only used for severe and refractory
systemic disease (11). However,
certain patients have poor response to drugs and the occurrence of
adverse reactions, so it is necessary to find targeted drugs that
can be applied to a range of patients with pSS.
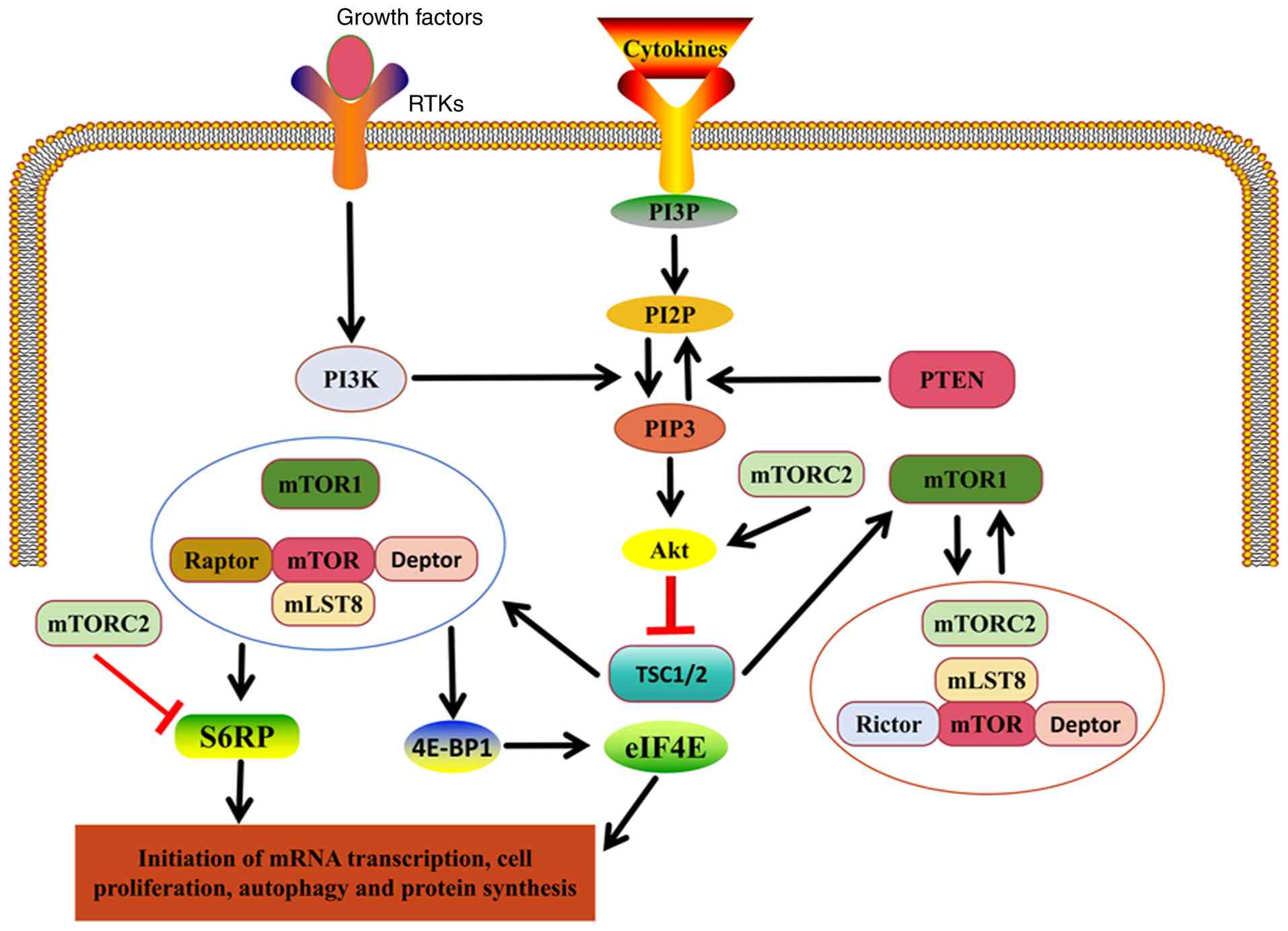
The mTOR signaling pathway serves a crucial role in
cellular metabolism, proliferation and survival. Studies in mouse
psoriasis models have shown that excessive activation of mTOR is
associated with autoimmune diseases, such as psoriasis (12,13). In 2013, the role of mTOR
signaling pathway activation in SS was first described (14), but the mechanism of its specific
occurrence and development in pSS has not yet been fully
elucidated. The excessive activation of mTOR is associated with
immune disorders (15), namely
the activation of immune cells and the secretion of
pro-inflammatory factors, which disrupts the balance of the immune
system. This may be associated with the occurrence and development
of pSS. PSS has a notable impact on patient health and quality of
life, as well as causing a social and economic burden. Therefore,
by reviewing the potential role of the mTOR signaling pathway in
pSS, the present study aimed to identify the potential pathogenesis
of pSS and therapeutic targets.
In 1991, TOR was identified during the screening of
rapamycin-resistant yeast mutants (16). Rapamycin, used as an
immunosuppressant and anticancer drug, interacts with TOR to
regulate cell proliferation and metabolism (16). In 1994, its mammalian homolog,
mTOR, was discovered (17). mTOR
is a serine/threonine kinase that belongs to the
phosphatidylinositol 3-kinase (PI3K)-related kinase family
(18). It is a highly conserved
protein composed of 2,549 amino acids, with a molecular weight of
~289 kDa (19). mTOR forms two
distinct complexes, mTOR complex (mTORC)1 and 2 (Fig. 1) (20). mTORC1 contains three core
components: mTOR, the mammalian lethal Sec13 protein 8 and the mTOR
regulatory-associated protein (Raptor), along with DEP
domain-containing mTOR-interacting protein (Deptor). By contrast,
mTORC2 lacks Raptor but contains Rictor (20). These two complexes differ in
composition and function. mTORC1 regulates protein and lipid
synthesis, suppresses catabolic processes such as lysosomal
biogenesis and autophagy, promotes cell proliferation and survival
and responds to environmental cues such as nutrients, stress,
oxygen levels and growth factors (21). By contrast, mTORC2 primarily
influences cell survival, metabolism and cytoskeletal organization
and is involved in regulating cell chemotaxis and migration
(22).
Upstream of mTOR, various growth factors and
signaling molecules, including PI3K and the tuberous sclerosis
complex (TSC1/2), regulate its activation (Fig. 1) (23). The downstream targets of the PI3K
pathway include serine/threonine kinases such as Akt, which
participate in metabolic regulation, cell survival and
proliferation (24). Thus, mTOR
serves as a central hub connecting multiple signaling pathways and
regulating key cellular functions. Among these, the PI3K/Akt/mTOR
signaling pathway plays a critical role in controlling metabolism
and determining cell fate processes, including proliferation,
differentiation and apoptosis (25,26).
Downstream of mTOR, the primary effectors include
ribosomal protein S6 kinase (S6K) and eukaryotic translation
initiation factor 4E-binding protein (20). Once mTORC activates S6K, it
transmits upstream signals to multiple effectors, regulating mRNA
translation, promoting protein and lipid synthesis and inhibiting
autophagy. These processes collectively enhance cell proliferation
(Fig. 1) (27,28). Several regulatory molecules also
modulate the mTOR signaling pathway, including TSC1/2 and PTEN,
which serve as negative regulators of mTOR activity, preventing
excessive activation and uncontrolled cell proliferation (Fig. 1) (29). Decreased mTOR activation impairs
lymphocyte development, proliferation and migration, leading to
immune deficiency. Conversely, hyperactivation of mTOR is
associated with autoimmune disease (30,31).
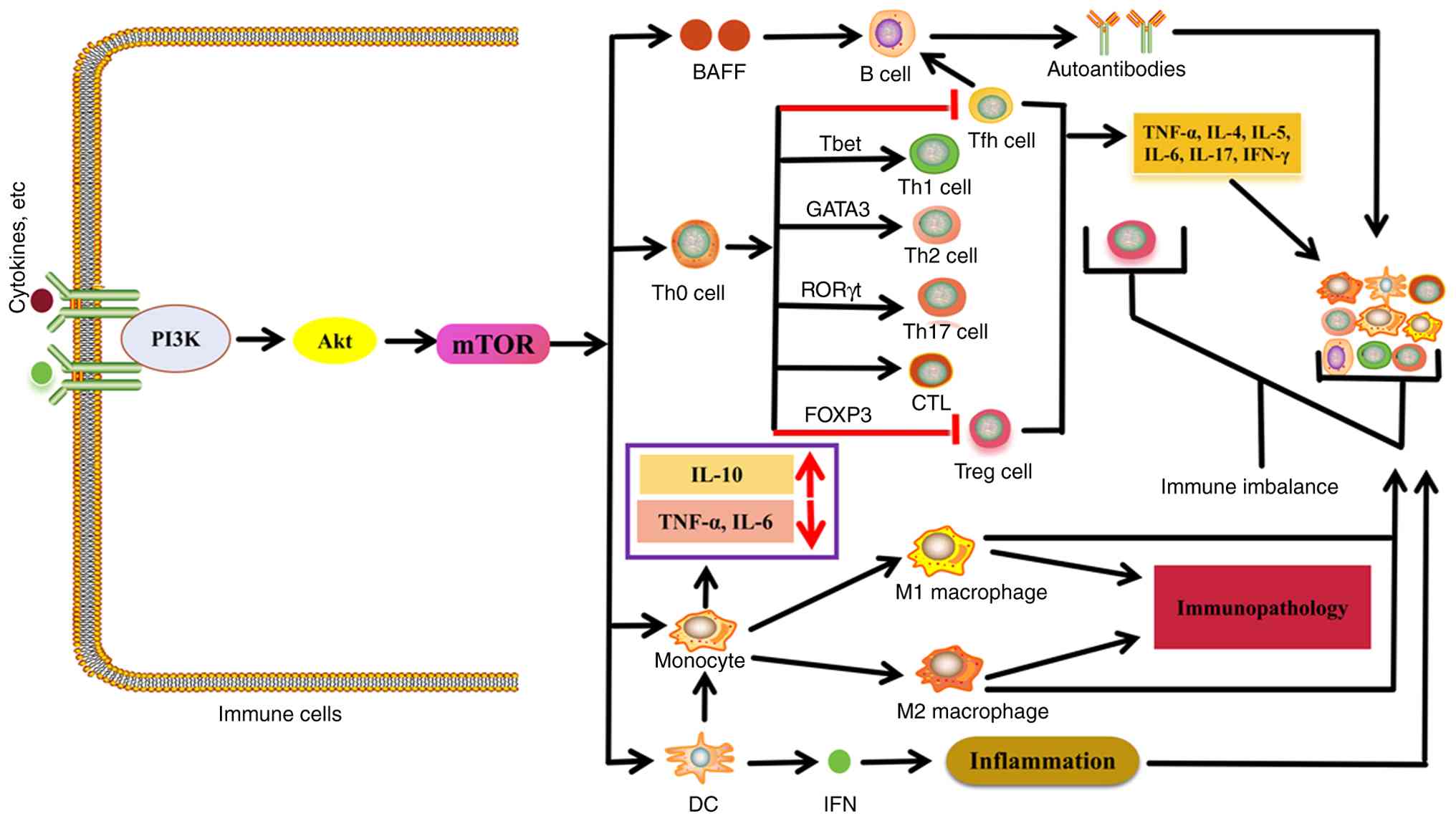
The mTOR signaling pathway serves a pivotal role in
regulating the activation, proliferation and differentiation of
immune cells, thereby influencing their functional properties.
Dysregulation of this pathway can lead to immune imbalance
(32-66) (Fig. 2). Due to its critical role in
immune cell development and function, mTOR signaling has garnered
increasing research attention.
DCs are specialized immune cells responsible for
initiating innate immune responses by capturing, processing and
presenting antigens to naïve T cells. They serve a vital role in
bridging innate and adaptive immunity by promoting T cell
activation (32). Studies have
demonstrated that the mTOR signaling pathway is key for regulating
the development and differentiation of DCs (33,34). In both NOD/B6.SJL mice and
humans, inhibition of mTORC impairs DC expansion induced by growth
factors (34,35). Conversely, loss of PTEN activates
the PI3K/Akt/mTOR signaling pathway, thereby promoting DC
proliferation and expansion (34). Under the regulation of
transcription factors IFN regulatory factors 5 and 7, mTORC1
activation enhances the expression of type I IFN in DCs (36). IL-4 activates mTORC1 in
monocytes, inducing their differentiation into DCs, whereas
inhibition of mTOR promotes DC apoptosis (37,38).
Monocytes and macrophages are phagocytic immune
cells that initiate and regulate inflammation through diverse
signaling mechanisms. They migrate to inflammatory sites, where
they differentiate into macrophages with distinct functional
phenotypes (39,40). The mTOR signaling pathway plays a
central role in monocyte activation and differentiation into
heterogeneous macrophage subsets, thereby maintaining immune
homeostasis. Dysregulation of this pathway disturbs immune balance
and has been implicated in various inflammatory diseases, such as
gout and sepsis (42,43). Deletion of the TSC2 gene in
monocytes results in mTORC1 activation, which enhances IL-10
production while suppressing pro-inflammatory cytokines such as
IL-6 and tumor necrosis factor (TNF)-α following lipopolysaccharide
stimulation (44). Additionally,
activation of the PI3K/Akt/mTOR signaling pathway promotes
polarization of monocytes toward the M2 macrophage phenotype
(45). Conversely, inhibition of
this pathway favors polarization toward the M1 phenotype (45,46). Persistent mTORC1 activation in
macrophages induces hypertrophy and proliferation, contributes to
immune pathology and is associated with granuloma formation
(47).
T cells are key components of the adaptive immune
system. Upon antigen recognition by the T cell receptor and the
presence of co-stimulatory molecules and cytokines, CD28-mediated
co-stimulation activates the PI3K/Akt/mTOR signaling pathway. This
activation supports metabolic activity and initiates the
activation, proliferation, and differentiation of CD4+
and CD8+ T cells (48). When mTOR activity is low,
immature T cells remain in a quiescent state (49). Conversely, hyperactivation of
mTOR enhances IL-12-mediated signal transducer and activator of
transcription 4 signaling, leading to increased expression of IFN-γ
and T-box transcription factor TBX21, which promotes
CD4+ T cell differentiation into the T helper (Th)1
phenotype (50). Overactivation
of mTORC2 in T cells induces high expression of the transcription
factor GATA3 and promotes differentiation into Th2 cells,
accompanied by the production of IL-4, IL-5 and IL-13 (51). In the presence of transforming
growth factor (TGF)-β and IL-6, excessive activation of mTORC1
upregulates the transcription factor retinoic acid-related orphan
receptor (ROR)γt, driving CD4+ T cell differentiation
into Th17 cells (51-52). Conversely, inhibition of mTOR
favors the differentiation of regulatory T (Treg) cells and T
follicular helper (Tfh) cells (48,53). Experimental evidence shows that
knockout of the mTOR gene in mice increases the population of
forkhead box P3 (FOXP3)+ Treg cells (54-55). By contrast, increased mTORC1
activity disrupts Treg stability, resulting in elevated production
of IL-17 and IL-1β and enhancing inflammatory responses (54-55). Moreover, loss of TSC2 leads to
increased generation of effector CD8+ T cells,
indicating that mTORC activity is essential for effector
CD8+ T cell differentiation, whereas memory
CD8+ T cell formation is markedly reduced (56,57). In summary, the mTOR signaling
pathway serves as a central regulatory hub that coordinates
multiple aspects of T cell biology, including development,
activation, differentiation, survival and memory formation.
B cells are essential components of the immune
system. The differentiation fate of immature B cells depends on the
integration of signals from B cell receptors (BCRs), co-stimulatory
molecules and cytokines (58).
Following BCR activation, AKT is recruited and phosphorylated,
leading to activation of the PI3K/Akt/mTOR signaling pathway. This
activation promotes protein synthesis, proliferation and
differentiation in B cells and amplifies BCR and CD40 signaling,
enabling naïve B cells to rapidly differentiate into
antibody-secreting cells via the extrafollicular (EF) pathway and
migrate to germinal centers (59). Decreased mTORC1 activation in B
cells impairs early development and decreases the formation of
antibody-secreting plasma cells (60,61). Conversely, hyperactivation of
mTORC1 accelerates EF plasma cell differentiation (62). Disruption of mTOR transcription
hinders B cell development and proliferation at multiple stages
(63). For example, the
survival, proliferation and metabolic activity, both oxidative and
glycolytic, of pre-B cells are diminished, while peripheral B cell
populations are depleted. This results in plasma cell dysfunction
and a failure to complete the preparatory processes required for
antibody production (54-65).
Furthermore, mTOR activation enhances the stimulatory effects of B
cell activation factor (BAFF), promoting uncontrolled B cell
proliferation and contributing to pathological autoimmunity
(66). These findings indicate
that excessive mTOR signaling promotes dysregulated B cell
proliferation, potentially leading to autoimmune disease.
According to a global epidemiological study based on
PubMed and Embase data, the overall incidence of pSS was 6.92
cases/100,000 person-years, and the overall prevalence was 60.82
cases/100,000 people in 2013 (67). pSS is more common in middle-aged
female patients, with a male-to-female incidence ratio of ~9:1
(68). The diagnostic age of pSS
is generally ~50 years, with an average diagnostic age ranging from
51.6±13.8 to 62.0±13.0 years old (69,70). The initial symptoms typically
appear before diagnosis, because patients often ignore dry mouth
and eyes (69,70). In addition, the incidence of pSS
increases with age. The incidence rate peaks in female patients
aged between 55 and 64 years and in male patients aged between 65
and 74 years (71). The
prevalence of pSS varies in different regions. Research has found
that the prevalence of pSS among patients with a European
background is twice as high as that among non-European patients
(72). There are also
differences between ethnicities. Black patients are diagnosed with
SS at an earlier age, and the male-to-female ratio of Asian
patients is 27:1 (73). In a
systematic review and meta-analysis, it was found that the
standardized mortality rate of patients with pSS is 1.38 (74). The primary causes of death are
cardiovascular diseases, solid organ and lymphoid malignancy and
infections (74). In addition,
multiple risk factors [such as male sex, older age at diagnosis,
extraductal involvement, vasculitis, anti-Sjögren syndrome B
antibody (SSB) positivity, hypocomplementemia and cryoglobulinemia]
are associated with an increased mortality rate (74).
In Th17 cells from patients with pSS, the mRNA
levels of PI3K, Akt, mTOR and IL-17 are significantly higher than
those in healthy controls (80,81). This suggests activation of the
PI3K/Akt/mTOR signaling pathway contributes to the proliferation
and differentiation of CD4+ T cells into Th17 cells and
promotes IL-17 secretion (80,81). Activation of the mTOR pathway
facilitates the nuclear translocation of RORγt, promotes Th17
differentiation and inhibits expansion of the Treg population,
resulting in immune dysregulation and an imbalance between Th17 and
Treg cells (82,83). IL-17 produced by Th17 cells
induces the secretion of pro-inflammatory cytokines such as TNF-α
and IL-1β. It also attracts immune cells to the SG by stimulating
IL-8 and chemokine release, thereby amplifying glandular
inflammation (84). In addition,
Th17 cells secrete IL-22, which recruits B cells and enhances
autoantibody production via cytokine-mediated stimulation (85). Inhibition of mTOR decreases RORγt
protein expression and Th17 polarization, while increasing FOXP3
expression and Treg cell numbers, thereby restoring the Treg/Th17
balance (86). These
observations indicate that the Th17/Treg imbalance driven by mTOR
pathway activation serves a key role in initiating and promoting
pSS progression.
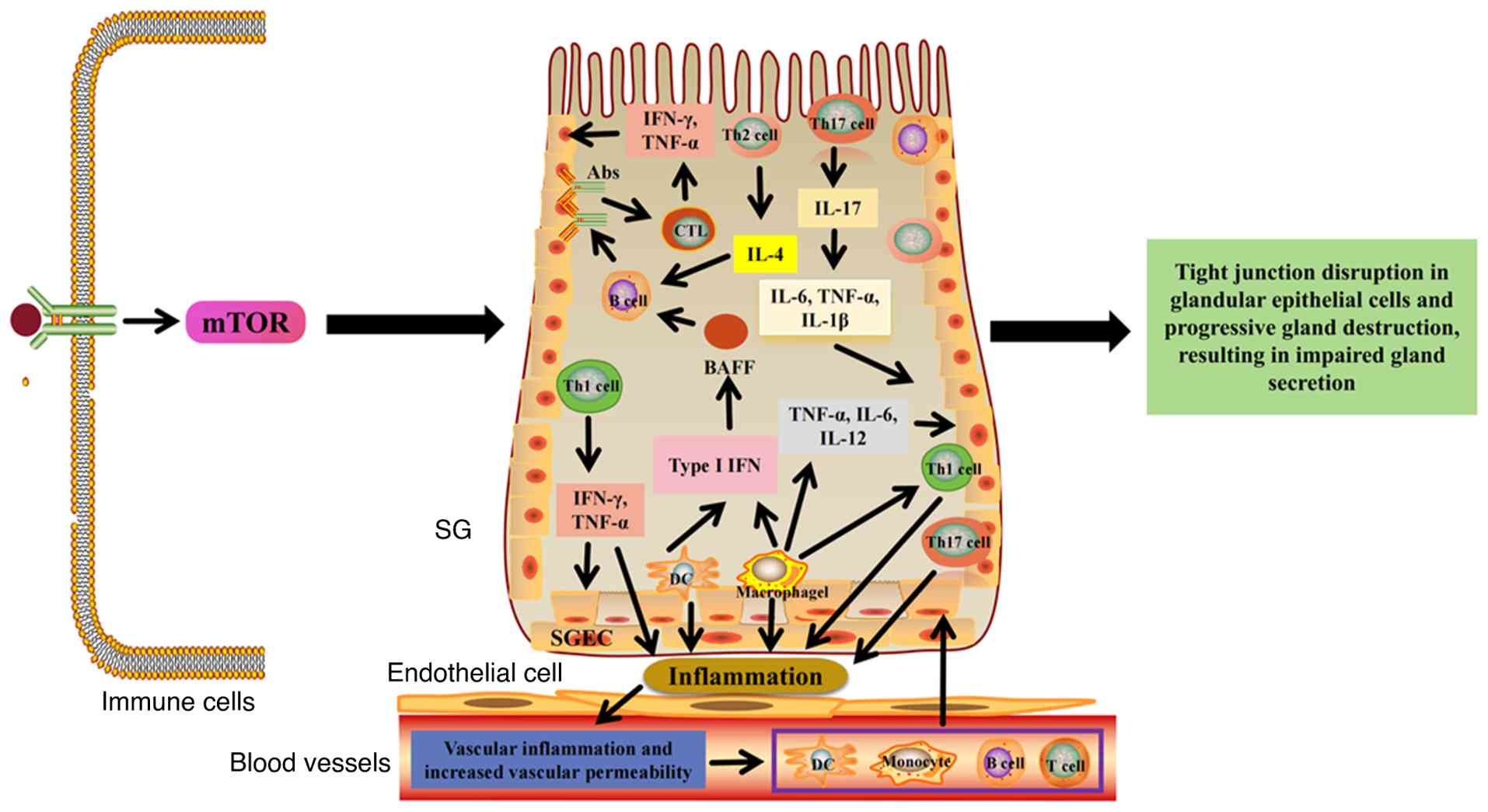
As aforementioned, activation of the mTORC signaling
pathway also induces the proliferation and differentiation of
CD4+ T cells into Th1 and Th2 subsets. The immune
response in the SG of patients with pSS is predominantly mediated
by Th1 cells, and Th1-associated cytokines such as IFN-γ and TNF-α
are significantly elevated in SG tissue (87,88). These cytokines disrupt the
integrity of tight junctions in SG epithelial cells and decrease
the number of vesicle cells, thereby impairing glandular secretory
function (88,89). Th2 cells contribute to B cell
activation through cytokines such as IL-4 (90). The mTORC signaling pathway also
activates macrophages, monocytes and DCs, which produce large
quantities of type I IFN and IFN-γ, amplifying the inflammatory
response and inducing elevated expression of BAFF. Elevated BAFF
levels are observed in the SG of patients with pSS (91). The interaction between BAFF and
its receptor promotes B cell activation, maturation and
proliferation, leading to aberrant antibody production, including
anti-Sjögren's-syndrome-related antigen (SS)A and anti-SSB
autoantibodies. These autoantibodies bind glandular epithelial
cells and transmit activating signals to T cells, driving their
differentiation into CD8+ cytotoxic T lymphocytes (CTLs)
(88). CTLs accumulate within
the SG, where they secrete high levels of IFN-γ and TNF-α,
sustaining glandular inflammation and contributing to the
pathogenesis of pSS (88).
Additionally, type I IFN stimulates macrophages to produce the
chemokine C-X-C motif ligand 13 (CXCL13), which promotes B cell
aggregation within the SG, thereby intensifying the inflammatory
response (92). In this
proinflammatory milieu, the mTOR signaling pathway activates
macrophages to polarize toward the M1 phenotype, leading to the
release of proinflammatory cytokines such as TNF-α, IL-6 and IL-12
(93,94). These cytokines accelerate
extracellular matrix degradation and cell apoptosis, promote T cell
differentiation into Th1 cells and exacerbate glandular
inflammation, resulting in tissue destruction (93,94). As pSS progresses, this chronic
inflammation gradually subsides, and M1 macrophages polarize toward
the M2 phenotype under the influence of TGF-β1 signaling (88,95). M2 macrophages secrete
anti-inflammatory mediators, including IL-10 and TGF-β, which
further reinforce M2 polarization and signaling, thereby promoting
glandular fibrosis (88,95). Under this sustained inflammatory
state, vascular endothelial cells in the SG become damaged.
Intercellular adhesion molecule-1 facilitates the adhesion of
immune cells to endothelial surfaces, promoting endothelial cell
activation. Consequently, vascular inflammatory responses increase
vascular permeability, creating a favorable environment for the
migration of immune cells, such as DCs, macrophages and
lymphocytes, from the periphery into the SG, thereby enhancing
immune cell infiltration (75,96,97). Therefore, in pSS, activation of
the mTOR signaling pathway primarily influences immune cell
proliferation and differentiation, contributes to immune imbalance,
such as Th17/Treg disequilibrium and M1/M2 polarization imbalance,
promotes the production of inflammatory cytokines and chemokines
and facilitates immune cell infiltration into the SG. The resulting
inflammatory response disrupts the tight junctions of glandular
epithelial cells, progressively destroying glandular architecture
via atrophy, ductal dilation and narrowing, leading to glandular
damage and secretory dysfunction (Fig. 3).
At present, the specific treatment plan for pSS is
unclear, and there is no radical cure. Symptomatic treatment is the
primary approach, with the aim of alleviating the symptoms of
exocrine diseases and controlling extraductal manifestations. The
first-line treatment drugs for SS-associated dry eye syndrome
include artificial tear drops and lubricating ointments (99). Topical cyclosporine, when used
alone or in combination with topical steroids, as well as oral
muscarinic receptor agonists such as rutin, serve as lacrimal gland
secretants and help alleviate dry eye symptoms (100). For dry mouth symptoms, patients
drink water frequently during the day and chew sugar-free gum to
increase saliva flow and maintain oral lubrication (99,100). At the same time, it is
recommended that patients maintain oral hygiene and have regular
dental check-ups due to the risk of tooth decay. If necessary,
muscarinic agonists should be used to promote SG secretion
(101). There are also
challenges in the treatment of pSS outside the gland. Typically,
the management strategies of closely related systemic autoimmune
diseases, such as systemic lupus erythematosus and rheumatoid
arthritis, are used. These drugs include non-steroidal
anti-inflammatory drugs, glucocorticoids, immunosuppressants (such
as methotrexate, leflunomide, sulfasalazine or cyclosporine) and
biological agents (such as belimumab and rituximab) (101). When pSS involves some organ
complications, such as lung diseases, decreased blood cell (such as
white blood cells and platelets) levels, vasculitis, kidney disease
and central nervous system and musculoskeletal disorder, these
drugs (such as methotrexate, leflunomide, sulfasalazine or
cyclosporine) need to be used in combination (99,102). Depending on the overall organ
involvement, it may be necessary to consult specialists and adopt a
comprehensive multidisciplinary approach. Although these drugs have
achieved certain therapeutic effects, they cannot cure the disease
and there are some adverse reactions, such as infections,
osteoporosis, gastrointestinal and cardiovascular complications,
gonadal toxicity, bone marrow suppression, fetal malformation and
miscarriages (103). This also
highlights the need for potential therapies.
As aforementioned, the immune dysregulation
resulting from excessive activation of multiple immune cell types
by the mTOR signaling pathway in patients with pSS contributes to
disease onset and progression. Beyond SG involvement, this
dysregulation may also affect extraglandular organs, leading to
complications that impair organ function and, in severe cases,
threaten patient survival (74).
Therefore, targeting the mTOR signaling pathway represents a key
therapeutic strategy for improving clinical outcomes in patients
with pSS.
Immunohistochemical analysis of SGs from patients
with pSS has shown increased expression of phosphorylated Akt and
mTOR in infiltrating lymphocytes, suggesting that activation of the
Akt/mTOR signaling pathway may be associated with pSS pathogenesis
(104). In vitro,
treatment with fangchinoline, an mTOR pathway inhibitor, suppresses
Akt/mTOR signaling in B cells, leading to decreased cell
proliferation, cell cycle arrest and enhanced apoptosis (104). Mesencephalic astrocyte-derived
neurotrophic factor (MANF), an evolutionarily conserved protein
secreted by brain astrocytes, is upregulated during inflammation
and is implicated in the pathogenesis of inflammatory disease
(105). When MANF is
co-cultured with lymphocytes from patients with pSS in vitro
study, it downregulates Akt and mTOR phosphorylation and promotes
autophagy in CD4+ T cells (106). Moreover, MANF decreases
lymphocytic infiltration in the SG and tripartite motif-containing
21/SSA expression (106).
Similarly, co-culture of rapamycin (100 nM) with Th and B cells
from patients with pSS in vitro inhibits mTOR activity,
thereby reducing the activation and proliferation of these cells
and suppressing IFN-γ and immunoglobulin G production (80). Another in vitro study
using iguratimod to treat CD4+ T cells from patients
with pSS showed that the drug targets pyruvate dehydrogenase kinase
1, inhibiting the Akt/mTOR signaling pathway (107). This inhibition suppresses the
activation, proliferation and differentiation of Tfh cells, further
impairing B cell activation and differentiation (107). Collectively, these in
vitro studies support the therapeutic potential of targeting
the mTOR signaling pathway in the treatment of pSS.
In a mouse model of pSS, treatment with a rapamycin
nanoparticle formulation (250 μM) downregulates mTOR gene
expression and significantly decreases lymphocyte infiltration in
the lacrimal glands (LGs), while simultaneously minimizing systemic
toxicity (14). Compared with
intraglandular delivery, the residence time of rapamycin within the
LGs is prolonged. Consequently, a lower total drug dose is
sufficient to suppress LG inflammation and improve tear secretion,
with no evidence of necrosis or fibrosis in the glands (108). These findings suggest that
rapamycin represents a promising therapeutic candidate for
pSS-associated lacrimal gland inflammation, achieving efficacy
through localized delivery that allows dose reduction and minimizes
systemic side effects. Runzaoling (RZL), a traditional Chinese
medicine clinical prescription with immunomodulatory properties,
administered via continuous gavage to non-obese diabetic (NOD) mice
for 10 weeks inhibits the mTOR signaling pathway, downregulates
IL-17 expression in Th17 cells of the submandibular gland and
decreases CD4+ T cell differentiation into Th17 cells
(81). In addition, metformin
has demonstrated immunoregulatory and anti-inflammatory effects
(109). In a pSS mouse model,
oral administration of metformin for 9 weeks enhances Treg cell
populations through activation of the AMPK/mTOR signaling pathway
(110). It also suppresses Th1,
Th17 and Tfh cell differentiation and decreases B cell activation,
thereby lowering the levels of IL-6, TNF-α and IL-17 (110). In a in vivo study, the
combined use of artemisinin salts and metformin demonstrated a
greater improvement in salivary secretion compared with artemisinin
salts alone (111). This
combination primarily regulates the PI3K/Akt pathway, inhibiting
apoptosis and autophagy in SGs of patients with type 2
diabetes-associated xerostomia, thereby alleviating SG damage
(111). These findings
highlight the potential of metformin to target the mTOR signaling
pathway for the treatment of pSS. Another compound with
antirheumatic properties, fangchinoline, a biologically active
dibenzylisoquinoline alkaloid, activates the Akt/mTOR signaling
pathway in NOD mice (104).
Fangchinoline effectively inhibits B lymphocyte proliferation,
decreases lymphocytic lesions in the submandibular gland and
increases salivary secretion in mice (104). Seletalisib, a potent and
selective oral PI3K inhibitor that modulates the Akt/mTOR signaling
pathway, significantly decreases phosphorylation of ribosomal
protein S6 in pSS mice (112).
Treatment with seletalisib also decreases the numbers of B and T
lymphocytes and plasma cells in the SG, leading to reduced
autoantibody titers and improved salivary secretion (112). Similarly, oral administration
of parsaclisib in pSS mice ameliorates SG inflammation, decreases
BAFF expression in saliva and lowers levels of autoreactive B cells
and autoantibodies (113).
PI3Kδ inhibitor AS2819899 suppresses T cell-dependent antibody
production, markedly reduced anti-double-stranded DNA antibody
titers and improves renal dysfunction in spontaneous systemic lupus
erythematosus (SLE) mice (114). Similarly, alpelisib, a PI3Kα
inhibitor, modulates B and T cells in a murine model of SLE
nephritis by inhibiting PI3Kα activity, which decreases
proinflammatory cytokine and autoantibody production, diminishes
glomerular complement deposition and improves renal histopathology
and function (115).
Regarding clinical studies, available data remain
limited. In a two-stage randomized, double-blind clinical study,
treatment of patients with pSS using seletalisib led to decreased B
and T cell numbers within the SG and shrinkage of glandular
lesions, demonstrating the therapeutic potential of targeting the
PI3K/mTOR signaling pathway in pSS management (116). However, adverse events were
reported, including diarrhea, abnormal hepatic and renal function
and allergic dermatitis (116).
In a single-center, single-arm phase II clinical study involving
patients with connective tissue disease-associated immune
thrombocytopenia (ITP), oral administration of the mTOR inhibitor
sirolimus (6-15 ng/ml) for 6 months produced no therapeutic
response in pSS-associated ITP (pSS-ITP) cases and no significant
changes were observed in lymphocyte subsets; no serious adverse
effects were detected during the study period (117). Conversely, another clinical
study reported that 75% of patients with pSS-ITP who receive
sirolimus achieve overall remission (118). Of two patients with SS and SLE,
one attained complete remission after 6 months and no severe
drug-related toxicity was observed, and the other patient had no
significant response (118).
Similarly, in a clinical study of seven patients with pSS-ITP
treated with sirolimus for 6 months, the complete, partial and
non-response rates were 42.9, 42.9 and 14.3%, respectively
(119). Treatment with
sirolimus increased the proportion of Treg cells while
significantly decreasing IL-8 and IL-17A levels (119). These findings suggest the
therapeutic potential of sirolimus in pSS, however, larger studies
are required to confirm its efficacy and safety. Notably, numerous
patients experience adverse effects during treatment, including
oral ulcers, hypercholesterolemia, gastrointestinal reactions and
fungal infections (119). In
other autoimmune diseases, sirolimus has also demonstrated
favorable therapeutic efficacy: In a real-world clinical study
involving 52 patients with SLE treated with sirolimus, complement
levels improved more markedly at 3 and 6 months compared with those
in the tacrolimus group (120).
Moreover, a higher proportion of patients in the sirolimus group
maintained prednisone doses ≤7.5 mg/day (120). In the treatment of lupus
nephritis, sirolimus produces greater improvements in complement
levels at 3, 6 and 12 months compared with mycophenolate mofetil
therapy (121). The
aforementioned studies reported mild adverse effects, including
infection, leukopenia, gastrointestinal discomfort, rash and mild
renal dysfunction. However, no serious adverse events or
discontinuations of sirolimus therapy were observed (120,121). A clinical study found that
belamcainide treatment of patients with pSS causes a transient
decrease in total B cell counts in peripheral blood, whereas memory
B cell levels remain elevated for a prolonged period before
declining below baseline with continued therapy (122). The concentration of CXCL13, an
alternative biomarker (B lymphocyte stimulator) reflecting SG
immune activity, is decreased, however, no significant improvement
in xerostomia symptoms is observed compared with controls (122). A preliminary clinical study
reported upregulation of the PI3K/Akt/mTOR signaling pathway, which
may be associated with inadequate clinical response to belamcainide
(123).
pSS is an autoimmune disease that not only affects
the quality of life of patients but also increases economic burden.
The immune disorder caused by mTOR activation is a potentially
important factor in the pathogenesis of SS. Targeting the mTOR
signaling pathway regulates the balance between immune cells.
Therefore, mTOR signaling pathway inhibitors are promising and
beneficial drugs in the treatment of pSS, but associated adverse
reactions are noted. At present, there is still a lack of research
on the activation of the mTOR signaling pathway and its targeting
in pSS. The specific mechanisms involved in this signaling pathway
and at the molecular level have not yet been clarified. Large-scale
prospective studies are required to confirm the efficacy in
reducing inflammation or raising platelets) and mechanisms. Optimal
drug dosage and safety of mTOR inhibitors in pSS should be explored
to provide novel therapeutic windows for pSS.
Not applicable.
RXH, CCW and YTY wrote the manuscript. DLM, YY and
JYL performed the literature review and constructed figures. XXH
conceptualized and designed the study. All authors have read and
approved the final manuscript. Data authentication is not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
No funding was received.
|
1
|
Manfrè V, Chatzis LG, Cafaro G, Fonzetti
S, Calvacchi S, Fulvio G, Navarro Garcia IC, La Rocca G, Ferro F,
Perricone C, et al: Sjögren's syndrome: One year in review 2022.
Clin Exp Rheumatol. 40:2211–2224. 2022.
|
|
2
|
Stefanski AL, Tomiak C, Pleyer U, Dietrich
T, Burmester GR and Dörner T: The diagnosis and treatment of
Sjögren's syndrome. Dtsch Arztebl Int. 114:354–361. 2017.PubMed/NCBI
|
|
3
|
Koh JH, Park Y, Lee J, Jeon H, Moon SJ,
Kim YH, Min JK, Park SH and Kwok SK: Long-term outcome of
interstitial lung disease in patients with primary Sjögren's
syndrome: A retrospective observational study. Korean J Intern Med.
40:148–159. 2025. View Article : Google Scholar :
|
|
4
|
Lessard CJ, Li H, Adrianto I, Ice JA,
Rasmussen A, Grundahl KM, Kelly JA, Dozmorov MG, Miceli-Richard C,
Bowman S, et al: Variants at multiple loci implicated in both
innate and adaptive immune responses are associated with Sjögren's
syndrome. Nat Genet. 45:1284–1292. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu Z and Chu A: Sjögren's syndrome and
viral infections. Rheumatol Ther. 8:1051–1059. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kapsogeorgou EK and Tzioufas AG:
Interaction of human salivary gland epithelial cells with B
lymphocytes: Implications in the pathogenesis of Sjögren's
syndrome. Mediterr J Rheumatol. 31:424–426. 2020. View Article : Google Scholar
|
|
7
|
Haacke EA, Bootsma H, Spijkervet FKL,
Visser A, Vissink A, Kluin PM and Kroese FGM: FcRL4+
B-cells in salivary glands of primary Sjögren's syndrome patients.
J Autoimmun. 81:90–98. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tanaka Y, Sotome T, Inoue A, Mukozu T,
Kuwabara T, Mikami T, Kowhi-Shigematsu T and Kondo M: SATB1
conditional knockout results in Sjögren's syndrome in mice. J
Immunol. 199:4016–4022. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
van Blokland SC, van Helden-Meeuwsen CG,
Wierenga-Wolf AF, Drexhage HA, Hooijkaas H, van de Merwe JP and
Versnel MA: Two different types of sialoadenitis in the NOD- and
MRL/lpr mouse models for Sjögren's syndrome: A differential role
for dendritic cells in the initiation of sialoadenitis? Lab Invest.
80:575–585. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tian Y, Yang H, Liu N, Li Y and Chen J:
Advances in pathogenesis of Sjögren's syndrome. J Immunol Res.
2021:59282322021. View Article : Google Scholar
|
|
11
|
Ramos-Casals M, Brito-Zerón P, Bombardieri
S, Bootsma H, De Vita S, Dörner T, Fisher BA, Gottenberg JE,
Hernandez-Molina G, Kocher A, et al: EULAR recommendations for the
management of Sjögren's syndrome with topical and systemic
therapies. Ann Rheum Dis. 79:3–18. 2020. View Article : Google Scholar
|
|
12
|
Dorjsembe B, Joo H, Nho C, Ham J and Kim
JC: Aruncus dioicus var. kamtschaticus extract ameliorates
psoriasis-like skin inflammation via Akt/mTOR and JAK2/STAT3
signaling pathways in a murine model. Nutrients. 14:50942022.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Roy T, Banang-Mbeumi S, Boateng ST, Ruiz
EM, Chamcheu RN, Kang L, King JA, Walker AL, Nagalo BM, Kousoulas
KG, et al: Dual targeting of mTOR/IL-17A and autophagy by fisetin
alleviates psoriasis-like skin inflammation. Front Immunol.
13:10758042023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shah M, Edman MC, Janga SR, Shi P,
Dhandhukia J, Liu S, Louie SG, Rodgers K, Mackay JA and
Hamm-Alvarez SF: A rapamycin-binding protein polymer nanoparticle
shows potent therapeutic activity in suppressing autoimmune
dacryoadenitis in a mouse model of Sjögren's syndrome. J Control
Release. 171:269–279. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shan J, Jin H and Xu Y: T cell metabolism:
A new perspective on Th17/treg cell imbalance in systemic lupus
erythematosus. Front Immunol. 11:10272020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Heitman J, Movva NR and Hall MN: Targets
for cell cycle arrest by the immunosuppressant rapamycin in yeast.
Science. 253:905–909. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sabatini DM, Erdjument-Bromage H, Lui M,
Tempst P and Snyder SH: RAFT1: A mammalian protein that binds to
FKBP12 in a rapamycin-dependent fashion and is homologous to yeast
TORs. Cell. 78:35–43. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mita MM, Mita A and Rowinsky EK: Mammalian
target of rapamycin: A new molecular target for breast cancer. Clin
Breast Cancer. 4:126–137. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Keith CT and Schreiber SL: PIK-related
kinases: DNA repair, recombination, and cell cycle checkpoints.
Science. 270:50–51. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang M, Lu Y, Piao W and Jin H: The
translational regulation in mTOR pathway. Biomolecules. 12:8022022.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kim YC and Guan KL: mTOR: A pharmacologic
target for autophagy regulation. J Clin Invest. 125:25–32. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sun Y, Wang H, Qu T, Luo J, An P, Ren F,
Luo Y and Li Y: mTORC2: A multifaceted regulator of autophagy. Cell
Commun Signal. 21:42023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zoncu R, Efeyan A and Sabatini DM: mTOR:
From growth signal integration to cancer, diabetes and ageing. Nat
Rev Mol Cell Biol. 12:21–35. 2011. View Article : Google Scholar
|
|
24
|
Nunnery SE and Mayer IA: Targeting the
PI3K/AKT/mTOR pathway in hormone-positive breast cancer. Drugs.
80:1685–1697. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yilmaz OH and Morrison SJ: The PI-3kinase
pathway in hematopoietic stem cells and leukemia-initiating cells:
A mechanistic difference between normal and cancer stem cells.
Blood Cells Mol Dis. 41:73–76. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Korets SB, Czok S, Blank SV, Curtin JP and
Schneider RJ: Targeting the mTOR/4E-BP pathway in endometrial
cancer. Clin Cancer Res. 17:7518–7528. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Magnuson B, Ekim B and Fingar DC:
Regulation and function of ribosomal protein S6 kinase (S6K) within
mTOR signalling networks. Biochem J. 441:1–21. 2012. View Article : Google Scholar
|
|
28
|
Alves CL and Ditzel HJ: Drugging the
PI3K/AKT/mTOR pathway in ER+ breast cancer. Int J Mol Sci.
24:45222023. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Grabiner BC, Nardi V, Birsoy K, Possemato
R, Shen K, Sinha S, Jordan A, Beck AH and Sabatini DM: A diverse
array of cancer-associated MTOR mutations are hyperactivating and
can predict rapamycin sensitivity. Cancer Discov. 4:554–563. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Marnett LJ: Lipid peroxidation-DNA damage
by malondialdehyde. Mutat Res. 424:83–95. 1999.PubMed/NCBI
|
|
31
|
Lai ZW, Kelly R, Winans T, Marchena I,
Shadakshari A, Yu J, Dawood M, Garcia R, Tily H, Francis L, et al:
Sirolimus in patients with clinically active systemic lupus
erythematosus resistant to, or intolerant of, conventional
medications: A single-arm, open-label, phase 1/2 trial. Lancet.
391:1186–1196. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shortman K and Liu YJ: Mouse and human
dendritic cell subtypes. Nat Rev Immunol. 2:151–161. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sukhbaatar N, Hengstschläger M and
Weichhart T: mTOR-mediated regulation of dendritic cell
differentiation and function. Trends Immunol. 37:778–789. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sathaliyawala T, O'Gorman WE, Greter M,
Bogunovic M, Konjufca V, Hou ZE, Nolan GP, Miller MJ, Merad M and
Reizis B: Mammalian target of rapamycin controls dendritic cell
development downstream of Flt3 ligand signaling. Immunity.
33:597–606. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
van de Laar L, Buitenhuis M, Wensveen FM,
Janssen HL, Coffer PJ and Woltman AM: Human CD34-derived myeloid
dendritic cell development requires intact phosphatidylinositol
3-kinase-protein kinase B-mammalian target of rapamycin signaling.
J Immunol. 184:6600–6611. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Schmitz F, Heit A, Dreher S, Eisenächer K,
Mages J, Haas T, Krug A, Janssen KP, Kirschning CJ and Wagner H:
Mammalian target of rapamycin (mTOR) orchestrates the defense
program of innate immune cells. Eur J Immunol. 38:2981–2992. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Haidinger M, Poglitsch M, Geyeregger R,
Kasturi S, Zeyda M, Zlabinger GJ, Pulendran B, Hörl WH, Säemann MD
and Weichhart T: A versatile role of mammalian target of rapamycin
in human dendritic cell function and differentiation. J Immunol.
185:3919–3931. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Katholnig K, Linke M, Pham H,
Hengstschläger M and Weichhart T: Immune responses of macrophages
and dendritic cells regulated by mTOR signalling. Biochem Soc
Trans. 41:927–933. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Auffray C, Sieweke MH and Geissmann F:
Blood monocytes: Development, heterogeneity, and relationship with
dendritic cells. Annu Rev Immunol. 27:669–692. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Shi C and Pamer EG: Monocyte recruitment
during infection and inflammation. Nat Rev Immunol. 11:762–774.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Italiani P and Boraschi D: From monocytes
to M1/M2 macrophages: Phenotypical vs. functional differentiation.
Front Immunol. 5:5142014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shalova IN, Lim JY, Chittezhath M,
Zinkernagel AS, Beasley F, Hernández-Jiménez E, Toledano V,
Cubillos-Zapata C, Rapisarda A, Chen J, et al: Human monocytes
undergo functional re-programming during sepsis mediated by
hypoxia-inducible factor-1α. Immunity. 42:484–498. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chung YH, Kim DH and Lee WW: Monosodium
urate crystal-induced pro-interleukin-1β production is
post-transcriptionally regulated via the p38 signaling pathway in
human monocytes. Sci Rep. 6:345332016. View Article : Google Scholar
|
|
44
|
Weichhart T, Costantino G, Poglitsch M,
Rosner M, Zeyda M, Stuhlmeier KM, Kolbe T, Stulnig TM, Hörl WH,
Hengstschläger M, et al: The TSC-mTOR signaling pathway regulates
the innate inflammatory response. Immunity. 29:565–577. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rauh MJ, Ho V, Pereira C, Sham A, Sly LM,
Lam V, Huxham L, Minchinton AI, Mui A and Krystal G: SHIP represses
the generation of alternatively activated macrophages. Immunity.
23:361–374. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Araki K, Ellebedy AH and Ahmed R: TOR in
the immune system. Curr Opin Cell Biol. 23:707–715. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Linke M, Pham HT, Katholnig K, Schnöller
T, Miller A, Demel F, Schütz B, Rosner M, Kovacic B, Sukhbaatar N,
et al: Chronic signaling via the metabolic checkpoint kinase mTORC1
induces macrophage granuloma formation and marks sarcoidosis
progression. Nat Immunol. 18:293–302. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Panwar V, Singh A, Bhatt M, Tonk RK,
Azizov S, Raza AS, Sengupta S, Kumar D and Garg M: Multifaceted
role of mTOR (mammalian target of rapamycin) signaling pathway in
human health and disease. Signal Transduct Target Ther. 8:3752023.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang R, Dillon CP, Shi LZ, Milasta S,
Carter R, Finkelstein D, McCormick LL, Fitzgerald P, Chi H, Munger
J and Green DR: The transcription factor Myc controls metabolic
reprogramming upon T lymphocyte activation. Immunity. 35:871–882.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kusaba H, Ghosh P, Derin R, Buchholz M,
Sasaki C, Madara K and Longo DL: Interleukin-12-induced
interferon-gamma production by human peripheral blood T cells is
regulated by mammalian target of rapamycin (mTOR). J Biol Chem.
280:1037–1043. 2005. View Article : Google Scholar
|
|
51
|
Waickman AT and Powell JD: mTOR,
metabolism, and the regulation of T-cell differentiation and
function. Immunol Rev. 249:43–58. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mafi S, Mansoori B, Taeb S, Sadeghi H,
Abbasi R, Cho WC and Rostamzadeh D: mTOR-mediated regulation of
immune responses in cancer and tumor microenvironment. Front
Immunol. 12:7741032022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Delgoffe GM, Kole TP, Cotter RJ and Powell
JD: Enhanced interaction between Hsp90 and raptor regulates mTOR
signaling upon T cell activation. Mol Immunol. 46:2694–2698. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Delgoffe GM, Kole TP, Zheng Y, Zarek PE,
Matthews KL, Xiao B, Worley PF, Kozma SC and Powell JD: The mTOR
kinase differentially regulates effector and regulatory T cell
lineage commitment. Immunity. 30:832–844. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Lee WH, Kim GE, Hong KJ, Kim HS and Lee
GR: Insulin receptor substrate 1 signaling inhibits Foxp3
expression and suppressive functions in Treg cells through the
mTORC1 pathway. Int J Mol Sci. 24:25512023. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Pollizzi KN, Patel CH, Sun IH, Oh MH,
Waickman AT, Wen J, Delgoffe GM and Powell JD: mTORC1 and mTORC2
selectively regulate CD8+ T cell differentiation. J Clin
Invest. 125:2090–2108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zeng H and Chi H: mTOR signaling in the
differentiation and function of regulatory and effector T cells.
Curr Opin Immunol. 46:103–111. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Tangye SG, Nguyen T, Deenick EK, Bryant VL
and Ma CS: Inborn errors of human B cell development,
differentiation, and function. J Exp Med. 220:e202211052023.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Staniek J and Rizzi M: Signaling
activation and modulation in extrafollicular B cell responses.
Immunol Rev. 330:e700042025. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Jones DD, Gaudette BT, Wilmore JR,
Chernova I, Bortnick A, Weiss BM and Allman D: mTOR has distinct
functions in generating versus sustaining humoral immunity. J Clin
Invest. 126:4250–4261. 2026. View Article : Google Scholar
|
|
61
|
Tsui C, Martinez-Martin N, Gaya M,
Maldonado P, Llorian M, Legrave NM, Rossi M, MacRae JI, Cameron AJ,
Parker PJ, et al: Protein kinase C-β dictates B cell fate by
regulating mitochondrial remodeling, metabolic reprogramming, and
heme biosynthesis. Immunity. 48:1144–1159.e5. 2018. View Article : Google Scholar
|
|
62
|
Benhamron S, Pattanayak SP, Berger M and
Tirosh B: mTOR activation promotes plasma cell differentiation and
bypasses XBP-1 for immunoglobulin secretion. Mol Cell Biol.
35:153–166. 2015. View Article : Google Scholar
|
|
63
|
Zhang S, Readinger JA, DuBois W,
Janka-Junttila M, Robinson R, Pruitt M, Bliskovsky V, Wu JZ,
Sakakibara K, Patel J, et al: Constitutive reductions in mTOR alter
cell size, immune cell development, and antibody production. Blood.
117:1228–1238. 2011. View Article : Google Scholar
|
|
64
|
Iwata TN, Ramírez JA, Tsang M, Park H,
Margineantu DH, Hockenbery DM and Iritani BM: Conditional
disruption of raptor reveals an essential role for mTORC1 in B cell
development, survival, and metabolism. J Immunol. 197:2250–2260.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Gaudette BT, Jones DD, Bortnick A, Argon Y
and Allman D: mTORC1 coordinates an immediate unfolded protein
response-related transcriptome in activated B cells preceding
antibody secretion. Nat Commun. 11:7232020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Zeng Q, Qin S, Zhang H, Liu B, Qin J, Wang
X, Zhang R, Liu C, Dong X, Zhang S, et al: Rapamycin attenuates
BAFF-extended proliferation and survival via disruption of mTORC1/2
signaling in normal and neoplastic B-lymphoid cells. J Cell
Physiol. 233:516–529. 2018. View Article : Google Scholar
|
|
67
|
Qin B, Wang J, Yang Z, Yang M, Ma N, Huang
F and Zhong R: Epidemiology of primary Sjögren's syndrome: A
systematic review and meta-analysis. Ann Rheum Dis. 74:1983–1989.
2015. View Article : Google Scholar
|
|
68
|
Mavragani CP and Moutsopoulos HM: The
geoepidemiology of Sjögren's syndrome. Autoimmun Rev. 9:A305–A310.
2010. View Article : Google Scholar
|
|
69
|
Baldini C, Pepe P, Quartuccio L, Priori R,
Bartoloni E, Alunno A, Gattamelata A, Maset M, Modesti M, Tavoni A,
et al: Primary Sjogren's syndrome as a multi-organ disease: impact
of the serological profile on the clinical presentation of the
disease in a large cohort of Italian patients. Rheumatology
(Oxford). 53:839–844. 2014. View Article : Google Scholar
|
|
70
|
Brito-Zerón P, Theander E, Baldini C,
Seror R, Retamozo S, Quartuccio L, Bootsma H, Bowman SJ, Dörner T,
Gottenberg JE, et al: Early diagnosis of primary Sjögren's
syndrome: EULAR-SS task force clinical recommendations. Expert Rev
Clin Immunol. 12:137–156. 2016. View Article : Google Scholar
|
|
71
|
Weng MY, Huang YT, Liu MF and Lu TH:
Incidence and mortality of treated primary Sjogren's syndrome in
Taiwan: A population-based study. J Rheumatol. 38:706–708. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Maldini C, Seror R, Fain O, Dhote R,
Amoura Z, De Bandt M, Delassus JL, Falgarone G, Guillevin L, Le
Guern V, et al: Epidemiology of primary Sjögren's syndrome in a
French multiracial/multiethnic area. Arthritis Care Res (Hoboken).
66:454–463. 2014. View Article : Google Scholar
|
|
73
|
Brito-Zerón P, Acar-Denizli N, Zeher M,
Rasmussen A, Seror R, Theander E, Li X, Baldini C, Gottenberg JE,
Danda D, et al: Influence of geolocation and ethnicity on the
phenotypic expression of primary Sjögren's syndrome at diagnosis in
8310 patients: A cross-sectional study from the big data Sjögren
project consortium. Ann Rheum Dis. 76:1042–1050. 2017. View Article : Google Scholar
|
|
74
|
Singh AG, Singh S and Matteson EL: Rate,
risk factors and causes of mortality in patients with Sjögren's
syndrome: A systematic review and meta-analysis of cohort studies.
Rheumatology (Oxford). 55:450–460. 2016.
|
|
75
|
Ren Y, Cui G and Gao Y: Research progress
on inflammatory mechanism of primary Sjögren syndrome. Zhejiang Da
Xue Xue Bao Yi Xue Ban. 50:783–794. 2021.
|
|
76
|
Mavragani CP and Moutsopoulos HM:
Sjögren's syndrome: Old and new therapeutic targets. J Autoimmun.
110:1023642020. View Article : Google Scholar
|
|
77
|
Cai Y, Sun R, Wang R, Ren JG, Zhang W,
Zhao YF and Zhao JH: The activation of Akt/mTOR pathway by
bleomycin in Epithelial-to-mesenchymal transition of human
submandibular gland cells: A treatment mechanism of bleomycin for
mucoceles of the salivary glands. Biomed Pharmacother. 90:109–115.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Soypaçacı Z, Gümüş ZZ, Çakaloğlu F, Özmen
M, Solmaz D, Gücenmez S, Gercik Ö and Akar S: Role of the mTOR
pathway in minor salivary gland changes in Sjogren's syndrome and
systemic sclerosis. Arthritis Res Ther. 20:1702018. View Article : Google Scholar
|
|
79
|
Assinder SJ, Dong Q, Kovacevic Z and
Richardson DR: The TGF-beta, PI3K/Akt and PTEN pathways:
Established and proposed biochemical integration in prostate
cancer. Biochem J. 417:411–421. 2009. View Article : Google Scholar
|
|
80
|
Blokland SLM, Hillen MR, Wichers CGK,
Zimmermann M, Kruize AA, Radstake TRDJ, Broen JCA and van Roon JAG:
Increased mTORC1 activation in salivary gland B cells and T cells
from patients with Sjögren's syndrome: mTOR inhibition as a novel
therapeutic strategy to halt immunopathology? RMD Open.
5:e0007012019. View Article : Google Scholar
|
|
81
|
Zeng P, Jiang Z, Huang Z, Huang Y, Xu H,
Chen C and Ma W: PI3K/AKT/mTOR signaling pathway is downregulated
by runzaoling (RZL) in Sjögren's syndrome. Mediators Inflamm.
2022:72361182022. View Article : Google Scholar
|
|
82
|
Shields CA, McCalmon M, Ibrahim T, White
DL, Williams JM, LaMarca B and Cornelius DC: Placental
ischemia-stimulated T-helper 17 cells induce
preeclampsia-associated cytolytic natural killer cells during
pregnancy. Am J Physiol Regul Integr Comp Physiol. 315:R336–R343.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Wang W, Sung N, Gilman-Sachs A and
Kwak-Kim J: T helper (Th) cell profiles in pregnancy and recurrent
pregnancy losses: Th1/Th2/Th9/Th17/Th22/Tfh cells. Front Immunol.
11:20252020. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Veldhoen M: Interleukin 17 is a chief
orchestrator of immunity. Nat Immunol. 18:612–621. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Barone F, Nayar S, Campos J, Cloake T,
Withers DR, Toellner KM, Zhang Y, Fouser L, Fisher B, Bowman S, et
al: IL-22 regulates lymphoid chemokine production and assembly of
tertiary lymphoid organs. Proc Natl Acad Sci USA. 112:11024–11029.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gao L, Dong Y, Lin R, Meng Y, Wu F and Jia
L: The imbalance of Treg/Th17 cells induced by perinatal bisphenol
A exposure is associated with activation of the PI3K/Akt/mTOR
signaling pathway in male offspring mice. Food Chem Toxicol.
137:1111772020. View Article : Google Scholar
|
|
87
|
Singh N and Cohen PL: The T cell in
Sjogren's syndrome: Force majeure, not spectateur. J Autoimmun.
39:229–233. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Qi W, Tian J, Wang G, Yan Y, Wang T, Wei
Y, Wang Z, Zhang G, Zhang Y and Wang J: Advances in cellular and
molecular pathways of salivary gland damage in Sjögren's syndrome.
Front Immunol. 15:14051262024. View Article : Google Scholar
|
|
89
|
Sisto M, Lisi S, Lofrumento DD, Frassanito
MA, Cucci L, D'Amore S, Mitolo V and D'Amore M: Induction of
TNF-alpha-converting enzyme-ectodomain shedding by pathogenic
autoantibodies. Int Immunol. 21:1341–1349. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Crotty S: A brief history of T cell help
to B cells. Nat Rev Immunol. 15:185–189. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Nocturne G and Mariette X: B cells in the
pathogenesis of primary Sjögren syndrome. Nat Rev Rheumatol.
14:133–145. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Zhao J, Kubo S, Nakayamada S, Shimajiri S,
Zhang X, Yamaoka K and Tanaka Y: Association of plasmacytoid
dendritic cells with B cell infiltration in minor salivary glands
in patients with Sjögren's syndrome. Mod Rheumatol. 26:716–724.
2016. View Article : Google Scholar
|
|
93
|
Baturone R, Soto MJ, Márquez M, Macías I,
de Oca MM, Medina F, Chozas N, García-Pérez S and Girón-González
JA: Health-related quality of life in patients with primary
Sjögren's syndrome: Relationship with serum levels of
proinflammatory cytokines. Scand J Rheumatol. 38:386–389. 2009.
View Article : Google Scholar
|
|
94
|
Zong Y, Yang Y, Zhao J, Li L, Luo D, Hu J,
Gao Y, Wei L, Li N and Jiang L: Characterisation of macrophage
infiltration and polarisation based on integrated transcriptomic
and histological analyses in Primary Sjögren's syndrome. Front
Immunol. 14:12921462023. View Article : Google Scholar
|
|
95
|
Lu X, Li N, Zhao L, Guo D, Yi H, Yang L,
Liu X, Sun D, Nian H and Wei R: Human umbilical cord mesenchymal
stem cells alleviate ongoing autoimmune dacryoadenitis in rabbits
via polarizing macrophages into an anti-inflammatory phenotype. Exp
Eye Res. 191:1079052020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Caprio M, Newfell BG, la Sala A, Baur W,
Fabbri A, Rosano G, Mendelsohn ME and Jaffe IZ: Functional
mineralocorticoid receptors in human vascular endothelial cells
regulate intercellular adhesion molecule-1 expression and promote
leukocyte adhesion. Circ Res. 102:1359–1367. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Błochowiak KJ, Olewicz-Gawlik A,
Trzybulska D, Nowak-Gabryel M, Kocięcki J, Witmanowski H and
Sokalski J: Serum ICAM-1, VCAM-1 and E-selectin levels in patients
with primary and secondary Sjögren's syndrome. Adv Clin Exp Med.
26:835–842. 2017. View Article : Google Scholar
|
|
98
|
Silver N, Proctor GB, Arno M and Carpenter
GH: Activation of mTOR coincides with autophagy during
ligation-induced atrophy in the rat submandibular gland. Cell Death
Dis. 1:e142010. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Maleki-Fischbach M, Kastsianok L, Koslow M
and Chan ED: Manifestations and management of Sjögren's disease.
Arthritis Res Ther. 26:432024. View Article : Google Scholar
|
|
100
|
Price EJ, Rauz S, Tappuni AR, Sutcliffe N,
Hackett KL, Barone F, Granata G, Ng WF, Fisher BA, Bombardieri M,
et al: The British society for rheumatology guideline for the
management of adults with primary Sjögren's syndrome. Rheumatology
(Oxford). 56:e24–e48. 2017. View Article : Google Scholar
|
|
101
|
Negrini S, Emmi G, Greco M, Borro M,
Sardanelli F, Murdaca G, Indiveri F and Puppo F: Sjögren's
syndrome: A systemic autoimmune disease. Clin Exp Med. 22:9–25.
2022. View Article : Google Scholar :
|
|
102
|
Yura Y and Hamada M: Outline of salivary
gland pathogenesis of Sjögren's syndrome and current therapeutic
approaches. Int J Mol Sci. 24:111792023. View Article : Google Scholar
|
|
103
|
Su X, Yu H, Lei Q, Chen X, Tong Y, Zhang
Z, Yang W, Guo Y and Lin L: Systemic lupus erythematosus:
Pathogenesis and targeted therapy. Mol Biomed. 5:542024. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Shao Y, Fu J, Zhan T, Ye L and Yu C:
Fangchinoline inhibited proliferation of neoplastic B-lymphoid
cells and alleviated Sjögren's syndrome-like responses in NOD/Ltj
mice via the Akt/mTOR pathway. Curr Mol Pharmacol. 15:969–979.
2022. View Article : Google Scholar
|
|
105
|
Zhou KG, Huang YB, Zhu ZW, Jiang M, Jin
LJ, Guan Q, Tian LL and Zhang JX: Mesencephalic astrocyte-derived
neurotrophic factor inhibits neuroinflammation through
autophagy-mediated α-synuclein degradation. Arch Gerontol Geriatr.
131:1057382025. View Article : Google Scholar
|
|
106
|
Cheng D, Zhou T, Liu H, Li L, Xuan Y,
Huang L, Liu Y, Zhang X, Wei W and Wu H: MANF inhibits Sjögren's
syndrome salivary gland epithelial cell apoptosis and antigen
expression of Ro52/SSA through endoplasmic reticulum
stress/autophagy pathway. Int Immunopharmacol. 122:1105822023.
View Article : Google Scholar
|
|
107
|
Lyu T, Jiang H, Zeng L, Liu S, He C, Luo
C, Qiao L, Zhao Y and Chen H: Iguratimod suppresses Tfh cell
differentiation in primary Sjögren's syndrome patients through
inhibiting Akt/mTOR/STAT3 signaling. Arthritis Res Ther.
25:1522023. View Article : Google Scholar
|
|
108
|
Ju Y, Edman MC, Guo H, Janga SR, Peddi S,
Louie SG, Junge JA, MacKay JA and Hamm-Alvarez SF: Intralacrimal
sustained delivery of rapamycin shows therapeutic effects without
systemic toxicity in a mouse model of autoimmune dacryoadenitis
characteristic of Sjögren's syndrome. Biomacromolecules.
22:1102–1114. 2021. View Article : Google Scholar
|
|
109
|
Kim J, Kim YS and Park SH: Metformin as a
treatment strategy for Sjögren's syndrome. Int J Mol Sci.
22:72312021. View Article : Google Scholar
|
|
110
|
Kim JW, Kim SM, Park JS, Hwang SH, Choi J,
Jung KA, Ryu JG, Lee SY, Kwok SK, Cho ML and Park SH: Metformin
improves salivary gland inflammation and hypofunction in murine
Sjögren's syndrome. Arthritis Res Ther. 21:1362019. View Article : Google Scholar
|
|
111
|
Zhang S and Li J, Nong X, Zhan Y, Xu J,
Zhao D, Ma C, Wang Y, Li Y, Li Z and Li J: Artesunate combined with
metformin ameliorate on diabetes-induced xerostomia by mitigating
superior salivatory nucleus and salivary glands injury in type 2
diabetic rats via the PI3K/AKT pathway. Front Pharmacol.
12:7746742021. View Article : Google Scholar
|
|
112
|
Nayar S, Campos J, Smith CG, Iannizzotto
V, Gardner DH, Colafrancesco S, Pipi E, Kollert F, Hunter KJ,
Brewer C, et al: Phosphatidylinositol 3-kinase delta pathway: A
novel therapeutic target for Sjögren's syndrome. Ann Rheum Dis.
78:249–260. 2019. View Article : Google Scholar :
|
|
113
|
Scuron MD, Fay BL, Connell AJ, Oliver J
and Smith PA: The PI3Kδ inhibitor parsaclisib ameliorates pathology
and reduces autoantibody formation in preclinical models of
systemic lupus erythematosus and Sjӧgren's syndrome. Int
Immunopharmacol. 98:1079042021. View Article : Google Scholar
|
|
114
|
Kaneko Y, Fukahori H, Yamagami K,
Kawashima T, Ito M, Akamatsu M, Marui T, Kato K, Takahashi F and
Morokata T: Effects of AS2819899, a novel selective PI3Kδ
inhibitor, in a NZB/W F1 mouse lupus-like nephritis model. Int
Immunopharmacol. 87:1067642020. View Article : Google Scholar
|
|
115
|
Yamaguchi J, Isnard P, Robil N, de la
Grange P, Hoguin C, Schmitt A, Hummel A, Megret J, Goudin N, Luka
M, et al: PIK3CA inhibition in models of proliferative
glomerulonephritis and lupus nephritis. J Clin Invest.
134:e1764022024. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Juarez M, Diaz N, Johnston GI, Nayar S,
Payne A, Helmer E, Cain D, Williams P, Devauchelle-Pensec V, Fisher
BA, et al: A phase 2 randomized, double-blind, placebo-controlled,
proof-of-concept study of oral seletalisib in primary Sjögren's
syndrome. Rheumatology (Oxford). 60:1364–1375. 2021. View Article : Google Scholar
|
|
117
|
Wu C, Wang Q, Xu D, Li M and Zeng X:
Sirolimus for patients with connective tissue disease-related
refractory thrombocytopenia: A single-arm, open-label clinical
trial. Rheumatology (Oxford). 60:2629–2634. 2021. View Article : Google Scholar
|
|
118
|
Du H, Su W, Su J, Hu J, Wu D, Long W and
Zhu J: Sirolimus for the treatment of patients with refractory
connective tissue disease-related thrombocytopenia: A pilot study.
Rheumatology (Oxford). 63:79–84. 2024. View Article : Google Scholar
|
|
119
|
Li X, Su L, Sun X, Li X, Tian Z, Liao Q,
Sun W and Zhao Y: Effect of sirolimus on connective tissue disease
related refractory thrombocytopenia: Clinical efficacy and
immunomodulatory mechanism. Postepy Dermatol Alergol. 42:494–501.
2025.PubMed/NCBI
|
|
120
|
Jiang N, Li M, Zhang H, Duan X, Li X, Fang
Y, Li H, Yang P, Luo H, Wang Y, et al: Sirolimus versus tacrolimus
for systemic lupus erythematosus treatment: Results from a
real-world CSTAR cohort study. Lupus Sci Med. 9:e0006172022.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Bai W, Peng L, Gui Y, Chen Y, Duan X, Li
X, Zhang H, Huo Y, Xu J, Yang P, et al: Sirolimus versus
mycophenolate mofetil for the treatment of lupus nephritis: Results
from a real-world CSTAR cohort study. Rheumatol Immunol Res.
6:80–89. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Mariette X, Barone F, Baldini C, Bootsma
H, Clark KL, De Vita S, Gardner DH, Henderson RB, Herdman M, Lerang
K, et al: A randomized, phase II study of sequential belimumab and
rituximab in primary Sjögren's syndrome. JCI Insight.
7:e1630302022. View Article : Google Scholar
|
|
123
|
Moysidou GS, Garantziotis P, Sentis G,
Nikoleri D, Malissovas N, Nikoloudaki M, Stergioti EM, Polia S,
Paschalidis N, Filia A, et al: Molecular basis for the
disease-modifying effects of belimumab in systemic lupus
erythematosus and molecular predictors of early response: Blood
transcriptome analysis implicates the innate immunity and DNA
damage response pathways. Ann Rheum Dis. 84:262–273. 2025.
View Article : Google Scholar : PubMed/NCBI
|