Introduction
Adenomyosis (AM) is a gynecological disorder
characterized by the infiltration of endometrial tissue into the
myometrium, clinically manifesting as dysmenorrhea, menorrhagia and
infertility (1). Epidemiological
data indicate an incidence of ~21.9% (2), with 5-25% of cases occurring in
women under the age of 39. Although advances in imaging techniques
such as transvaginal ultrasound and magnetic resonance imaging
(MRI) have significantly improved the diagnostic rate of AM in
women of reproductive age (3),
the increasing trend of delayed childbearing has made the disease a
major contributing factor to secondary infertility, with chronic
inflammatory response and immune dysregulation emerging as the core
drivers linking the pathological progression of AM to reproductive
impairment. Moreover, due to its insidious nature, early-stage and
mild cases are often underdiagnosed, suggesting that the actual
prevalence may be underestimated.
Numerous studies have confirmed that women with
infertility frequently present with sonographic features of AM and
exhibit significant immune dysfunction (4,5).
AM has increasingly become a critical factor impacting both female
fertility and the outcomes of assisted reproductive technology
(ART) (6,7). The abnormal infiltration of
tissue-specific immune cell subsets, such as macrophages, natural
killer (NK) cells and mast cells, combined with an imbalance
between pro-inflammatory cytokines [such as interleukin (IL)-6,
IL1B and C-X-C motif chemokine ligand 8 (CXCL8)] and
anti-inflammatory cytokines (such as IL-4 and IL-10), contributes
to pathological angiogenesis, impaired endometrial decidualization
and reduced endometrial receptivity (ER). These changes ultimately
lead to embryo implantation failure (8), thereby resulting in adverse
pregnancy outcomes. Elevated levels of inflammatory markers in both
serum and peritoneal fluid confirm that chronic inflammation and
immune dysregulation are not isolated local phenomena but systemic
disruptions of the immune-inflammatory network in AM (9). Therefore, elucidating the unique
immunoregulatory network centered on chronic inflammation and
immune dysregulation in AM, identifying diagnostic inflammatory
cytokines and developing targeted intervention strategies are
essential to improving reproductive outcomes in affected
patients.
Pathogenic mechanisms and clinical evidence
of AM-associated reproductive impairment
AM-induced reproductive dysfunction arises from the
interplay of three interconnected pathological dimensions:
Structural derangements of the uterus and fallopian tubes,
functional impairments of ovarian and myometrial physiology and
molecular dysregulation of the endometrial microenvironment, with
persistent inflammatory signaling serving as the core mediator that
links these dimensions into a self-reinforcing pathological loop
(10). Studies have consistently
confirmed that these multifaceted abnormalities collectively
compromise both natural conception and ART outcomes (11), as elaborated below.
Pathogenic mechanisms
The pathogenesis of AM-related infertility begins
with structural anomalies that disrupt the physiological
architecture of the reproductive tract. Structural assessments
through hysterosalpingography reveal significantly higher uterine
cavity distortion rates in patients with AM compared with controls:
78% in diffuse AM and 54% in focal AM vs. 37% in non-AM populations
(6). Anatomical distortion of
the uterine wall, characterized by myometrial hypertrophy and
endometrial invagination into the myometrium, not only destroys the
spatial integrity required for embryo implantation but also exerts
mechanical traction on the fallopian tubes, impairing their
transport dynamics (11). Using
T2-weighted MRI and hysterosalpingoscintigraphy, Kissler et
al (12) demonstrated a
significantly higher incidence of impaired tubal transport in
patients with diffuse AM compared with those with focal AM and
healthy controls. This tubal dysfunction compromises the timely
meeting of gametes and the subsequent transport of embryos to the
uterine cavity, directly hindering fertilization. Concurrently, the
structural derangement of the uterus exacerbates functional
impairments: Aberrant myometrial contractility patterns (marked by
increased frequency and uncoordinated contractions) disrupt embryo
positioning within the uterus and reduce blood perfusion to the
junctional zone (JZ), a critical region for implantation (13). Additionally, reduced ovarian
responsiveness in patients with AM is closely linked to local and
systemic inflammatory cytokines [such as tumor necrosis factor-α
(TNF-α) and IL-6], which interfere with granulosa cell function and
folliculogenesis, diminishing oocyte quality and quantity (11).
Underpinning these structural and functional
abnormalities is molecular and immunological dysregulation, with
the persistent inflammatory microenvironment acting as a central
driver. Spatial transcriptomic analyses have revealed that during
ectopic endometrial invasion in AM, SFRP5+ epithelial
cells promote endometrial proliferation and angiogenesis through
activation of the Indian hedgehog signaling molecule signaling
pathway, initiating basal layer invagination, while
ESR1+ smooth muscle cells (SMCs) facilitate invasive
tract formation via collagen degradation (13). These processes not only reinforce
uterine structural distortion but also sustain inflammation, which
further suppresses implantation-related molecular pathways.
Specifically, elevated levels of pro-inflammatory cytokines (such
as IL-6 and TNF-α) and oxidative stress markers in the adenomyotic
microenvironment downregulate key implantation genes such as
Homeobox (HOX)A10, HOXA11 and leukemia inhibitory factor (LIF),
compromising ER and inhibiting decidualization (6). A genomic study has further
confirmed the dysregulation of 34 fertility-associated genes during
the implantation window, including imbalances in the matrix
metalloproteinases (MMPs)/tissue inhibitors (TIMPs) system, an
essential regulator of decidualization and trophoblast invasion
(14).
Notably, these pathological processes are mutually
reinforcing; structural distortion amplifies inflammatory signaling
by disrupting tissue integrity, while inflammation exacerbates
structural and functional damage through fibrosis (driven by
CNN1+ stromal fibroblasts) and sustained tissue injury
(13). This loop ultimately
creates an intrauterine environment hostile to embryo survival and
implantation.
Clinical evidence
Clinical data consistently validate the link between
the pathological features of AM and poor reproductive outcomes,
with severity-dependent effects clearly observed. In terms of ART
outcomes, meta-analysis data has revealed that patients with AM
have a clinical pregnancy rate of 40.5% following in vitro
fertilization (IVF)/intracytoplasmic sperm injection compared with
49.8% in controls [risk ratio (RR), 0.72; 95% confidence interval
(CI), 0.55-0.95], coupled with significantly elevated miscarriage
rates (31.9 vs. 14.1%; RR, 2.12; 95% CI 1.20-3.75) (15). Further supporting this
severity-dependent effect, a prospective multicenter study by
Mavrelos et al (16)
demonstrated a stepwise decline in clinical pregnancy rates: 42.7%
(95% CI, 37.1-48.3) in women without sonographic AM features, 22.9%
(95% CI, 13.4-32.6) in those with four features and further to
13.0% (95% CI, 2.2-23.9) in those with all seven features,
indicating a strong association between disease severity and
reduced fertility. Epidemiological data further highlight the role
of AM in reproductive failure: The prevalence of AM is 24.4% among
infertile populations, rising to 38.2% in recurrent miscarriage
cohorts and 34.7% in recurrent implantation failure (RIF) groups
(17), a finding consistent with
the notion that persistent inflammatory and molecular dysregulation
disproportionately affects women with repeated reproductive losses.
Even in oocyte recipient cycles, AM independently increases early
miscarriage risk and lowers term pregnancy rates, likely due to JZ
dysfunction impairing trophoblast development and early
placentation (18).
Collectively, this confirms that intrauterine pathological changes
(rather than ovarian factors alone) drive poor reproductive
outcomes (11).
Synthesizing the aforementioned mechanisms and
clinical evidence, we consider that while anatomical derangements
and myometrial dysfunction represent relatively fixed pathological
endpoints, the aberrant inflammatory milieu (functioning as the
core mediator of the pathological loop) constitutes a tractable
therapeutic target. Elucidating the key signaling pathways and
immunomodulatory networks underlying inflammation-driven damage
could inform the development of novel interventional strategies to
disrupt this pathological loop, restore ER and ultimately improve
fertility outcomes in patients with AM. This framework also directs
future translational research toward validating these targets in
clinical trials.
Abnormal expression of inflammatory
cytokines in AM promotes infertility
Pro-inflammatory cytokine IL-6 enhances
proliferation and invasion of the eutopic endometrium
IL-6, predominantly secreted by activated
macrophages (19), exhibits
elevated expression in endometrial stromal cells (ESCs) in
macrophage co-culture systems (20). Spatial transcriptomics analysis
has identified an IL-6-centered protein-protein interaction
subnetwork within adenomyotic lesions, forming a synergistic
pro-inflammatory axis with cytokines such as CXCL8 and TNF-α, which
activates local inflammatory cascades via the Toll-like receptor
(TLR)4/NF-κB signaling pathway (21). Mechanistically, canonical IL-6
signaling involves complex formation with the soluble IL-6 receptor
and gp130 co-receptor (22),
triggering Janus kinase (JAK)2/signal transducer and activator of
transcription (STAT)3 pathway activation characterized by STAT3
phosphorylation and nuclear translocation (23). Dysregulation of the
IL-6/JAK2/STAT3 pathway has been implicated in the pathogenesis of
autoimmune diseases, chronic inflammation and certain malignancies
(24). In AM, endometrial
cell-derived exosomes have been shown to activate this signaling
pathway, thereby promoting ESC proliferation, migration and cell
cycle progression. Tocilizumab, an IL-6 receptor inhibitor, was
able to effectively reverse these effects (25). Moreover, IL-6 trans-signaling
activates the PI3K/AKT/mTOR pathway, which suppresses the
autophagic death of ectopic endometrial cells, thereby promoting
lesion establishment (26).
Concurrently, activation of the RAS/RAF/MEK/ERK pathway enhances
cellular motility, accelerating disease progression in AM (27).
In reproductive physiology, IL-6 plays key
regulatory roles in embryo implantation, fetal development and
other pregnancy-associated processes (28). Experimental evidence demonstrates
that blastocyst development is impaired in IL-6 knockout mice,
thereby adversely affecting pregnancy outcomes. IL-6 not only
induces the secretion of monocyte chemoattractant protein-1 (MCP-1)
by decidual stromal cells, promoting macrophage recruitment and the
formation of a localized inflammatory response that impairs embryo
implantation (20), but also
disrupts ER and affects embryo implantation through activation of
the JAK/STAT3 signaling pathway (29). Furthermore, IL-6 mediates
vascular and immune dysregulation at the maternal-fetal interface
(placenta/decidua) and has been proposed as a potential diagnostic
or biomarker candidate for endometriosis (EMs)-associated
infertility (30).
Pro-inflammatory cytokine CXCL8 induces
proliferation and angiogenesis in the eutopic endometrium
As a key member of the α-chemokine family, CXCL8
mediates neutrophil chemotaxis and activation via specific
interactions with cognate receptors CXCR1 and CXCR2. In patients
with AM, CXCL8 mRNA and protein expression are markedly upregulated
in both ectopic lesions and the eutopic endometrium (31), paralleled by proliferative-phase
upregulation of CXCR1/CXCR2 receptors on endometrial epithelial
cells (32). Through paracrine
signaling, the CXCL8-CXCR1 axis is activated, thereby promoting the
invasive growth of ectopic endometrial tissue (33).
Physiologically, CXCL8 expression in normal
endometrium peaks during the late secretory phase, facilitating
neutrophil recruitment and stromal proliferation to prepare for
menstruation (32). However, in
AM pathophysiology, loss of CXCL8 cyclical rhythmicity results in
defective postmenstrual repair and pathological angiogenesis within
the eutopic endometrium. Activation of the TLR4/p38/ERK pathway can
induce CXCR1 expression and promote CXCL8 secretion (34). Clinical analyses reveal
diminished CXCL8 concentrations in the cervical mucus of patients
with AM-associated infertility (35), while elevated serum CXCL8 levels
and autoantibody positivity highlight its potential as a diagnostic
biomarker for immune-mediated infertility (36). Singh et al (37) demonstrated inverse correlations
between CXCL8 levels and both oocyte maturity and embryo quality. A
further study has confirmed the dual role of CXCL8 in regulating
early pregnancy outcomes through endometrial paracrine signaling
and mediating embryo implantation via CXCR1 binding in patients
with AM (38).
IL1B regulates inflammatory cascades,
immune homeostasis and hormonal crosstalk in AM-associated
infertility
IL1B, a prototypical pro-inflammatory cytokine,
initiates biological responses through binding to IL-1 receptor
type I (IL-1R1) and recruiting IL-1 receptor accessory protein
(IL-1RAcP) to form a high-affinity receptor complex (39). IL1B undergoes canonical
activation by lipopolysaccharide (LPS) binding to TLRs. This
binding event induces TLRs to recruit the adaptor protein MyD88 via
their intracellular Toll/IL-1 receptor domain, culminating in IL1B
transcriptional activation (40). Elevated IL1B expression is
consistently observed in the eutopic and ectopic endometrium of
patients with AM (19), where it
serves as a critical initiator of pathological inflammatory
cascades. MyD88 can also activates IRAK kinases and the IκB kinase
(IKK) complex, leading to inhibitor of NF-κB (IκB) degradation and
NF-κB nuclear translocation (41), thereby initiating downstream
inflammatory cascades via the classical NF-κB pathway.
Furthermore, upregulation of IL1B has been
implicated in disrupting the T helper cell 17 (Th17)/regulatory T
cells (Treg) balance, leading to immune intolerance at the
maternal-fetal interface, which is a key mechanism underlying
implantation failure (42). The
function of regulatory factors involved in embryo-maternal
communication, such as IL-1RAcP and IL-1R antagonist (IL-1RA), may
also be impaired. The ratio of IL-1RA to IL1B has been proposed as
a potential predictive marker for adverse pregnancy outcomes in
patients with AM (43).
In addition, Wang et al (44) demonstrated a significant positive
correlation between IL1B expression and serum levels of estradiol
(E2), progesterone and endometrial thickness (P<0.05),
suggesting that IL1B may synergize with E2 and progesterone to
improve ER and could serve as a predictive indicator of pregnancy
outcomes. Lastly, IL1B has been reported to impair sperm binding to
the zona pellucida in EMs (45),
suggesting that this mechanism may also contribute to infertility
in patients with AM.
Anti-inflammatory cytokine IL10
facilitates immune tolerance in ectopic endometrial tissues
As a representative anti-inflammatory cytokine,
IL-10 exerts its effects by forming a functional heterodimeric
receptor complex composed of IL-10 receptor 1 and IL-10 receptor 2
(46). On one hand, IL-10
suppresses immune responses by inhibiting Th1 cell activity and
disrupting the Th1/Th2 balance; on the other hand, it can also
enhance immune responses by stimulating the functions of
CD8+ T cells, NK cells and B cells (47).
Multiple studies have confirmed that IL-10
expression is significantly elevated in both the eutopic and
ectopic endometrial epithelial tissues of patients with AM compared
with normal endometrium (48-50). Mechanistically, ectopic
endometrial tissues in AM exhibit tumor-like invasive and immune
escape properties as they must evade host immune surveillance to
colonize and proliferate within the myometrium (51). This process is highly consistent
with the immunosuppressive function of IL10. In tumor cells, IL-10
has been shown to inhibit antigen-presenting cell function and T
cell activation by inducing the expression of human leukocyte
antigen-G (HLA-G) and downregulating the levels of classical MHC
class I and II antigens, thereby reducing pro-inflammatory cytokine
production and achieving immune escape (52). This suggests that IL-10 may play
a similar role in AM, promoting immune escape and enabling ectopic
endometrial cells to invade the myometrium and establish local
immune tolerance (53).
At the maternal-fetal interface, IL-10 critically
maintains immune tolerance through STAT3/HOXA10-mediated regulation
of ER (54). During the
implantation window, IL-10 contributes to the establishment of a
tolerogenic immune microenvironment by reducing the proportion and
functional activity of uterine NK (uNK) cells at the
decidual-placental interface. Abnormally low IL-10 levels in the
endometrium or placental tissues have been observed in
pregnancy-related complications such as recurrent miscarriage and
preeclampsia (55). During the
implantation window, significantly reduced local endometrial IL-10
levels have been observed in patients with AM, which may represent
a key factor contributing to their lower embryo implantation rates
compared with women with normal endometrium (20).
Role of other inflammatory cytokines in
AM-related infertility COX-2 mediates cell proliferation,
angiogenesis and multifaceted pathways to drive AM-associated
reproductive dysfunction
COX-2 is a key enzyme involved in the conversion of
arachidonic acid into various endogenous prostaglandins. While
typically undetectable in normal tissues, COX-2 can be induced by
inflammatory stimuli, earning it the label of a 'stress gene'
(56). In AM, elevated COX-2
expression has been observed in both the eutopic and ectopic
endometrium; it contributes to disease pathogenesis by regulating
inflammatory pathways linked to cell proliferation, angiogenesis
and immune suppression (57).
Moreover, COX-2 has been proposed as a potential molecular
biomarker or therapeutic target for dysmenorrhea in patients with
early-stage AM (58). An in
vitro study has shown that COX-2 inhibitors significantly
suppress the migration and invasion of endometrial mesenchymal stem
cells (59), supporting its
potential as a novel therapeutic target in AM.
COX-2 exacerbates AM-associated infertility through
multiple mechanisms. First, its upregulation increases
prostaglandin E2 (PGE2) production, driving chronic inflammation
that leads to ovarian apoptosis, pelvic adhesions, fallopian tube
dysfunction and luteal phase defects. Second, COX-2-derived PGE2
activates aromatase, boosting local estrogen biosynthesis and
creating a positive feedback loop between estrogen and inflammation
that accelerates ectopic lesion growth. In addition, the COX-2/PGE2
signaling pathway downregulates progesterone receptor (PR)
expression, inducing progesterone resistance and impairing
endometrial decidualization (60). Furthermore, COX-2 may disrupt the
function of stromal telocytes, compromising structural integrity at
the utero-tubal junction and affecting gamete transport and the
embryo implantation microenvironment (61). Through these coordinated effects
on hormonal balance, tissue structure and immune-endocrine
crosstalk, COX-2 acts as a central regulator of reproductive
dysfunction in AM.
NF-κB mediates the pathogenesis of AM via
regulating inflammatory cytokine networks, immune tolerance and
progesterone resistance
NF-κB serves as a central regulator of inflammatory
responses. Upon stimulation by pro-inflammatory factors such as
LPS, TNF-α or IL-1, IKKβ mediates the phosphorylation of IκB,
leading to the release of the active p50/p65 heterodimer. This
complex translocates into the nucleus and initiates the
transcription of pro-inflammatory cytokines such as IL-6, TNF-α and
CXCL8, as well as COX-2 (62). A
preclinical study has validated the pathogenic role of NF-κB in AM,
where endometrial microenvironmental aberrations sustain NF-κB
activation, generating cytotoxic inflammation that impairs
embryonic viability and endometrial epithelial integrity,
ultimately diminishing implantation rates, effects reversible
through NF-κB inhibition in murine AM models (63).
In terms of pregnancy, the TLR4/NF-κB pathway
regulates maternal-fetal immune tolerance by modulating the
Th17/Treg cell ratio. This balance is crucial for proper
trophoblast invasion and angiogenesis at the placental interface,
which are essential for ensuring adequate fetal blood supply,
nutrient delivery and successful pregnancy outcomes (64). Furthermore, NF-κB engages in
reciprocal crosstalk with PR isoforms: Chronic inflammatory
signaling induces constitutive NF-κB activation that selectively
downregulates PR-B expression, fostering progesterone resistance.
This molecular disruption impairs endometrial decidualization and
creates a non-receptive implantation milieu. Fig. 1 illustrates the regulatory
networks of multiple inflammation-related cytokines (including
IL-1, IL-6, IL-8 and IL-10) and their downstream signaling pathways
(such as NF-κB and STAT3) involved in the aforementioned processes
(65).
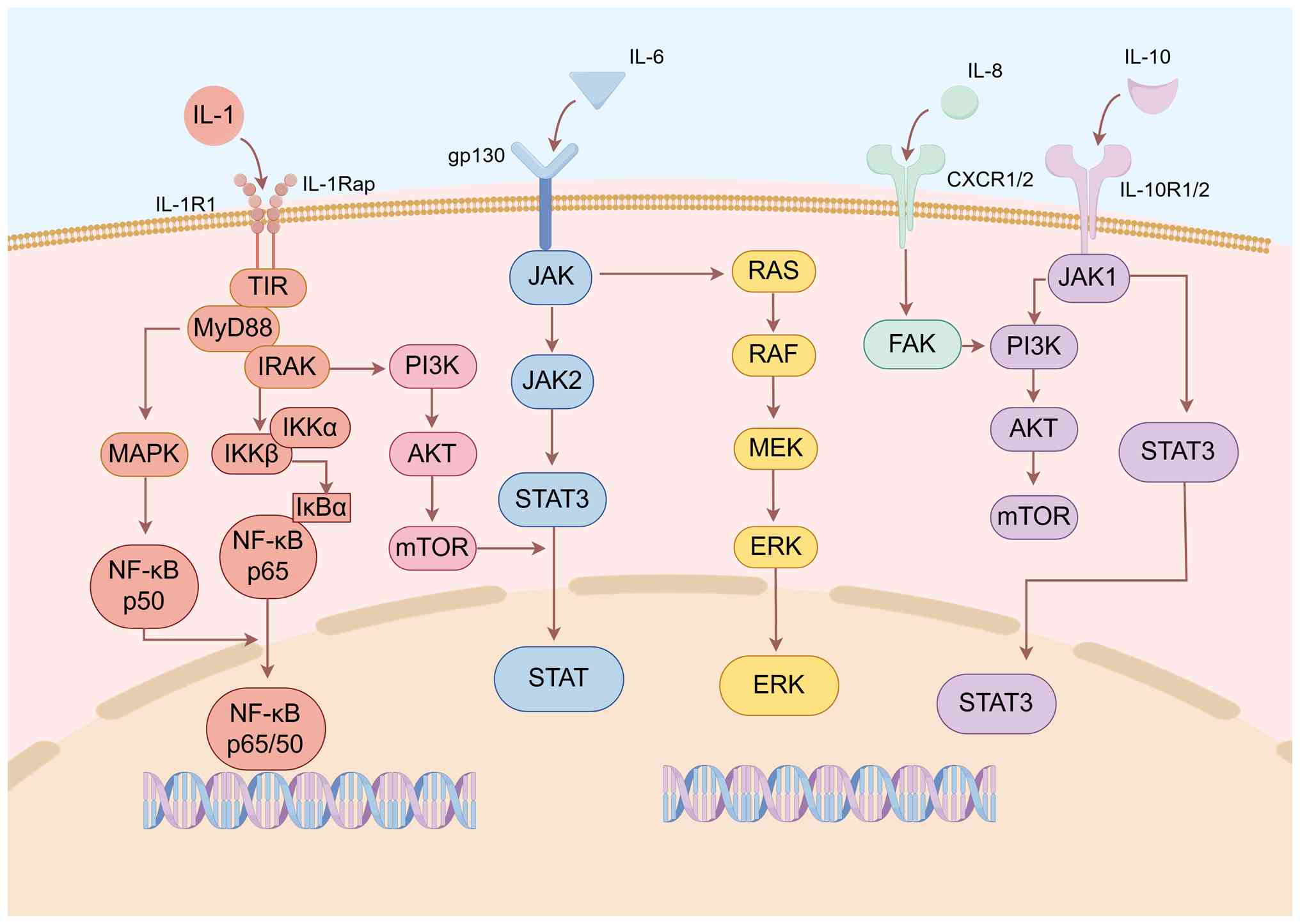 | Figure 1Main inflammatory pathways through
which inflammatory cytokines promote the occurrence and development
of adenomyosis. IL-1, IL-6, CXCL8 and IL10 are the inflammatory
cytokines mainly described in the present review. Their promotion
of the establishment and development of adenomyosis mainly involves
signal pathways such as JAK/STAT, PI3K/AKT/mTOR, RAS/RAF/MEK/ERK
and NK-κB. AKT, protein kinase B; CXCR1/2, C-X-C chemokine receptor
1/2; ERK, extracellular signal-regulated kinase; FAK, focal
adhesion kinase; gp130, glycoprotein 130; IKKa/b, IκB kinase a/b;
IκBa, inhibitor of nuclear factor κB a; IL, interleukin; IL-1R1,
interleukin-1 receptor 1; IL-1Rap, interleukin-1 receptor accessory
protein; IRAK, interleukin-1 receptor-associated kinase; JAK, Janus
kinase; MAPK, mitogen-activated protein kinase; MEK,
mitogen-activated protein kinase kinase; mTOR, mammalian target of
rapamycin; MyD88, myeloid differentiation primary response 88;
NF-κB, nuclear factor κ-light-chain-enhancer of activated B Cells;
PI3K, phosphatidylinositol 3-kinase; RAF, rapidly accelerated
fibrosarcoma; RAS, rat sarcoma viral oncogene homolog; STAT3,
signal transducer and activator of transcription 3; TIR,
Toll/interleukin-1 receptor domain. |
Immune cell-derived inflammatory
cytokines lead to infertility
Immune cells serve as the primary source of
inflammatory cytokines, and their dysregulated activation and
crosstalk disrupt the endometrial immune microenvironment and
maternal-fetal interface homeostasis, ultimately contributing to
AM-associated infertility.
Macrophages
Macrophages are critical regulators of tissue
inflammation and repair, and their phenotypic and functional
abnormalities are closely linked to AM pathogenesis. Studies
consistently demonstrate significantly elevated macrophage density
in both the eutopic and ectopic endometrial tissues of patients
with AM compared with normal endometrium (66,67), with a phenotypic shift:
Pro-inflammatory M1 macrophages decrease, while anti-inflammatory
M2 macrophages increase, this shift creates an immunosuppressive
microenvironment that favors ectopic lesion survival.
The pathogenic effects of M2 macrophages in AM are
multifaceted: First, excessive M2-derived cytokines (such as IL-10
and TGF-β) promote angiogenesis, accelerate ectopic endometrial
cell proliferation and induce pathological tissue fibrosis. Second,
M2 macrophages activate the STAT3 signaling pathway in ectopic
endometrial cells, further enhancing their growth and invasive
capacity (68), Third, the
accumulation of M2 macrophages alters myometrial contractility,
disrupting embryo transport through the fallopian tube and
impairing the stability of the implantation microenvironment
(69).
Mechanistically, an in vitro study has
confirmed that AM-derived ESCs secrete chemokine CCL2, which
recruits monocytes via the estrogen receptor β (ERβ)/NF-κB
signaling pathway (70); these
recruited monocytes then undergo excessive M2 polarization under
the influence of the CCL2-CCR2 axis. In addition, ESCs release
exosomal microRNA (miRNA/miR)-301a-3p, which mediates M2
polarization via the PTEN/PI3K pathway (71), forming a vicious cycle.
Upregulated Legumain pseudogene 1 not only promotes M2 polarization
but also serves as a non-invasive biomarker for predicting lesion
recurrence (72). The resultant
M1/M2 disequilibrium disrupts implantation-phase inflammatory
homeostasis at the maternal-fetal interface, impairing embryo
attachment competence (73).
NK cells
uNK cells are the most abundant lymphocyte subset in
the endometrium, accounting for 30-50% of endometrial lymphocytes
and 70% of decidual lymphocytes. Under physiological conditions,
uNK cells play indispensable roles in regulating ER, trophoblast
invasion and placental vascular remodeling, functions mediated by
their secretion of cytokines such as TNF-α, IL-10, IL-1β and LIF
(74). The number and function
of uNK cells exhibit dynamic changes throughout the menstrual
cycle: Their counts remain low during the proliferative phase,
increase significantly after ovulation and peak during early
pregnancy (75). In the
secretory phase, uNK cells participate in spiral artery remodeling;
after embryo implantation, they promote early placental development
by facilitating trophoblast invasion and optimizing spiral artery
dilation, thereby ensuring efficient maternal-fetal exchange of
nutrients and oxygen (76).
Additionally, uNK cells eliminate infected maternal decidual cells
during early pregnancy, maintaining local immune surveillance
without damaging the embryo (77). Abnormal uNK cell function has
been implicated in multiple reproductive disorders, including
recurrent miscarriage and repeated implantation failure,
highlighting their critical role in reproductive success (78).
Although current studies have not confirmed
significant differences in NK cell expression in women with AM, one
key study has reported increased uNK cell counts in the eutopic
endometrium of patients with severe AM (diffuse or adenomyoma
type). More notably, regardless of quantity, the functional defect
of uNK cells in AM [specifically, reduced expression of killer
immunoglobulin-like receptors (KIRs)] is a major factor impairing
embryo implantation (79).
Furthermore, uNK cells collaborate with other endometrial/decidual
immune cells to establish a Th2-dominant immune environment, a
hallmark of maternal-fetal immune tolerance. In AM, abnormal uNK
cell subsets or dysregulated uNK-derived cytokine networks disrupt
this Th2-dominant state, increasing the risk of maternal immune
rejection of the embryo and impeding successful pregnancy (80).
Mast cells and T cells
Aberrant activation of immune cells such as mast
cells and T cells adversely affects reproductive function by
modulating the inflammatory microenvironment. Table SI summarizes the key immune cell
subsets (including mast cells, T cells and other relevant
populations), their associated pro-inflammatory/profibrotic
mediators and downstream pathological effects on the endometrium
and reproductive outcomes. Specifically, activated mast cells
release profibrotic mediators such as TGF-β1 and fibroblast growth
factor (FGF), which accelerate endometrial fibrosis (81); this not only distorts the
endometrial structure (compromising ER) but also enhances the
invasive capacity of ectopic lesions. Moreover, mast cell-derived
inflammatory mediators (such as histamine and prostaglandins)
disrupt endometrial epithelial integrity and alter local blood
flow, creating an unfavorable microenvironment for embryo
implantation (82).
T cell subsets (particularly Th17 cells and Treg
cells) play a central role in maintaining immune homeostasis at the
maternal-fetal interface. In AM, this balance is severely
disrupted: The number of pro-inflammatory Th17 cells increases,
while the number and functional activity of immunosuppressive Treg
cells decrease (83), This
Th17/Treg imbalance intensifies local endometrial inflammation
(such as elevated IL-17 secretion) and impairs maternal-fetal
immune tolerance.
Inflammatory microenvironment in AM
affects pregnancy outcomes
The tumor microenvironment, often referred to as the
'soil' for malignant tumor growth, plays a critical role in
coordinating tumor invasion and immune evasion through inflammatory
mediators, immune cell infiltration, angiogenesis and matrix
remodeling. Notably, a highly analogous dynamic system exists in
AM. Although considered a benign condition, AM lesions exhibit
several hallmarks of malignancy. Specifically, ectopic endometrial
cells are capable of degrading the basement membrane via MMP-2 and
MMP-9, infiltrating the myometrium in an asymmetric pattern,
disrupting the vascular and lymphatic systems and facilitating the
implantation of endometrial tissue within the myometrial layer,
processes that closely resemble the local invasion behavior of
malignant tumors (84).
Furthermore, lesion cells can evade apoptosis by upregulating the
anti-apoptotic protein Bcl-2 through the activation of NF-κB
signaling (85). In addition,
persistent activation of hypoxia-inducible factor-1α (HIF-1α)
drives a shift toward glycolytic metabolic reprogramming, resulting
in excessive lactate production. This not only supports cellular
energy homeostasis but also creates an acidic microenvironment
similar to that observed in tumors, thus establishing a vicious
cycle that confers survival advantages (86). Notably, chronic inflammatory
stimulation has been shown to induce the expression of
epithelial-mesenchymal transition (EMT) markers such as Snail and
vimentin, which enhances the invasive potential of lesion cells and
significantly increases the risk of malignant transformation of
ectopic endometrial lesions (87).
The immune dysregulation associated with AM markedly
contributes to infertility; it impairs ovulatory function, induces
tubal dysfunction or adhesions and disrupts the transport of sperm
and embryos (88). Additionally,
the immunosuppressive microenvironment, characterized by the
secretion of immunoinhibitory factors, interferes with the immune
support required for normal embryo development, leading to
pregnancy failure (Fig. 2)
(89). Some patients may also
produce autoantibodies against endometrial tissue or embryos (such
as antiphospholipid antibodies and anti-endometrial antibodies),
directly damaging the endometrium or targeting the embryo, thereby
impairing implantation (90).
Moreover, abnormal expression of chemokines and adhesion molecules
may disrupt embryo-endometrial signaling, reducing ER. A recent
single-cell RNA sequencing study has revealed altered intercellular
communication among epithelial cells, stromal cells, perivascular
cells, endothelial cells and immune cells within ectopic lesions.
These alterations lead to changes in pathways associated with cell
proliferation, angiogenesis and immune responses, underscoring the
dynamic remodeling of the AM microenvironment. This spatiotemporal
dysregulation ultimately impairs embryo implantation (91).
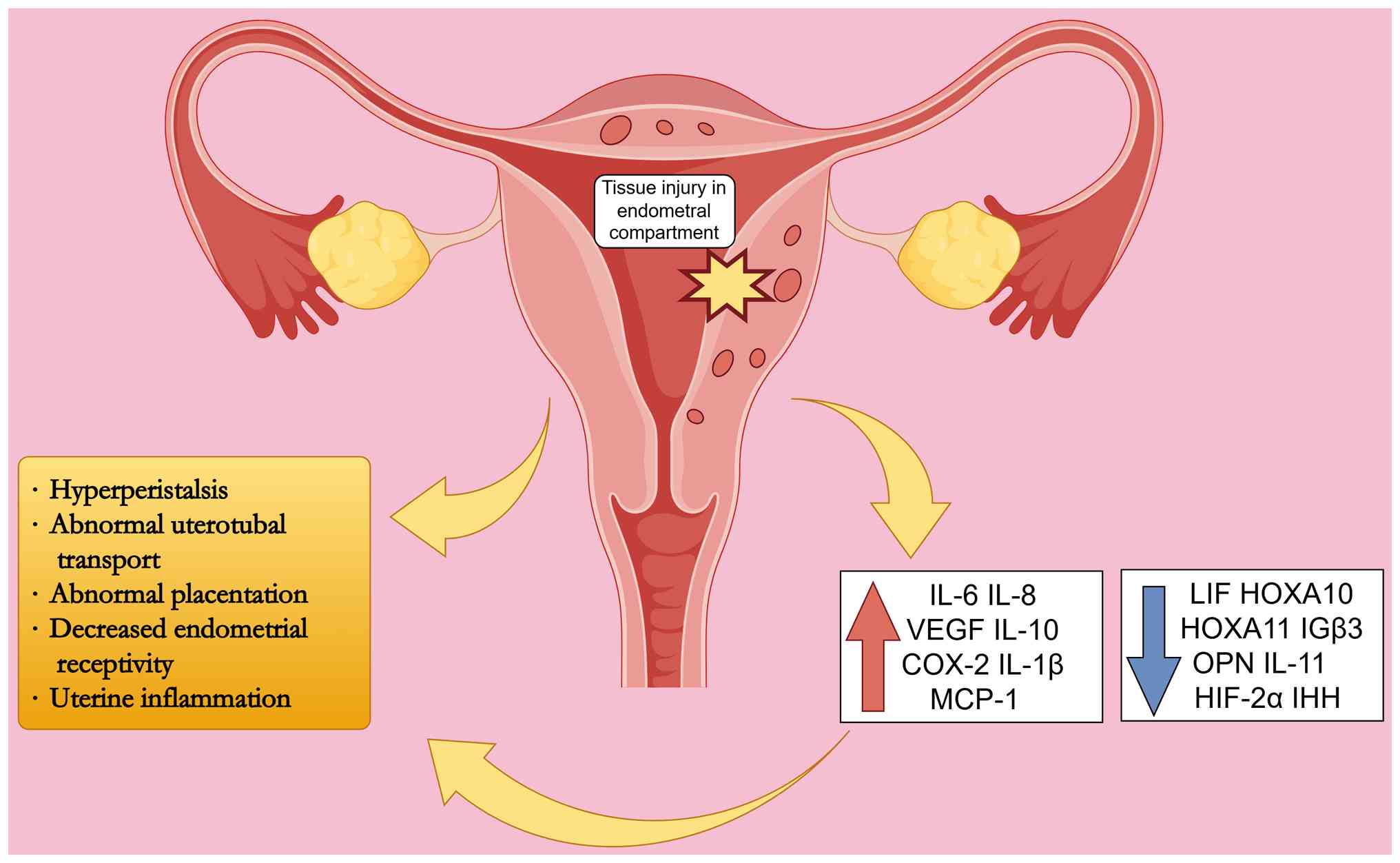 | Figure 2Abnormal aggregation of inflammatory
cytokines affects the inflammatory microenvironment and interferes
with multiple stages of embryo implantation. Tissue damage at the
endometrium-myometrium interface is the key trigger for abnormal
aggregation of inflammatory cytokines. Among them, IL-6, CXCL8,
VEGF, IL10, COX-2, IL1B and MCP-1 are upregulated, while LIF,
HOXA10, HOXA11, IGβ3, OPN, IL11, HIF-2α and IHH are downregulated.
The abnormal inflammatory microenvironment leads to
hyperperistalsis of the uterus, decreased fallopian tube transport
function, abnormal placental formation and decreased endometrial
receptivity. COX-2, cyclooxygenase-2; HIF-2α, Hypoxia-Inducible
Factor-2α; HOXA10, homeobox A10; HOXA11, homeobox A11; IHH, Indian
hedgehog; IGβ3, integrin β3; IL, interleukin; LIF, leukemia
inhibitory factor; MCP-1, monocyte chemoattractant protein-1; OPN,
osteopontin; VEGF, vascular endothelial growth factor. |
Mechanisms of inflammatory cytokine
regulation in myometrial peristalsis, tissue damage and repair and
EMT in AM
Inflammatory cytokines disrupt uterine
peristalsis by altering the immune microenvironment
Under physiological conditions, the JZ (a
transitional region between the endometrium and myometrium) serves
two core functions; it exhibits periodic contractile patterns to
regulate menstrual shedding, gamete transport and embryo
implantation (92), while also
acting as a physiological immune regulatory barrier that maintains
genital tract microbial homeostasis and optimizes the embryo
implantation microenvironment via immune cells (such as macrophages
and NK cells) and cytokine networks (93).
In MRI diagnostics, JZ thickening (>12 mm) is
explicitly a pathognomonic feature of AM (94). Maubon et al (95) confirmed a threshold-dependent
association between JZ thickness and implantation rate:
Implantation rates of 45% when JZ thickness is <10 mm, a sharp
decline to 16% at 10-12 mm, and a further reduction to 5% in cases
with JZ thickness >12 mm. They further summarized that
MRI-quantified JZ thickness is the primary negative predictor of
implantation failure; specifically, beyond the critical threshold
of 7 mm, the risk of implantation failure increases exponentially
(95). This inverse
thickness-implantation relationship persists across diagnostic
modalities. Notably, Tellum et al (96) emphasized that JZ morphological
heterogeneity, particularly focal asymmetric thickening secondary
to myometrial cysts or adenomyoma formation, should be incorporated
into the differential diagnostic criteria for AM.
JZ dysfunction in patients with AM impairs
placentation. Impaired decidual transformation leads to trophoblast
retention within the JZ, hindering spiral artery remodeling and
causing defective deep placentation (17). Single-cell RNA sequencing has
further confirmed a reduced number of uNK cells and an insufficient
secretion of angiogenic factors in JZ-thickened regions, which
directly limits the depth of trophoblast invasion and exacerbates
defective deep placentation (16). Therefore, the abnormally
thickened JZ observed in AM may hinder embryo transport and
preimplantation positioning, while also disrupting the
microenvironmental homeostasis at the endometrial-myometrial
junction (EMJ) during decidualization (12).
Physiologically, uterine peristalsis exhibits
phase-specific dynamics. Antegrade peristaltic waves mediate
menstrual shedding during the proliferative phase, retrograde
contractions facilitate sperm migration during the ovulatory window
and quiescent contractions in the luteal phase create a favorable
microenvironment for embryo implantation (97). Thus, physiological uterine
peristalsis is indispensable for coordinating intrauterine sperm
and embryo translocation. In patients with AM, speckle-tracking
transvaginal ultrasonography reveals characteristic pathological
contractile patterns, including a disorganized contractile
direction during the ovulatory phase and hypercontractility in the
luteal phase (98). The core
mechanism underlying this pathological contractility is linked to
the excessive local production of prostaglandin F2α (PGF2α) and
IL-6 in the JZ (99). These
factors collectively drive luteal-phase uterine hyperperistalsis,
impairing embryo positioning and adhesion. Chronic
hypercontractility further exacerbates JZ microtrauma, sustains
inflammatory cascades and establishes an 'inflammation-estrogen'
feedforward loop that accelerates AM pathogenesis. Clinically,
these dysregulations manifest as obstetric sequelae, including
uterine atony, retained placenta and postpartum hemorrhage,
highlighting the central role of JZ dysfunction in both the
pathological mechanisms and clinical consequences of AM (100).
Mechanisms of tissue injury and repair
(TIAR)
The TIAR hypothesis is a central framework for
explaining AM pathogenesis, as it highlights inflammation and
immune dysregulation as key drivers of disease initiation and
progression, with elevated inflammatory mediators and a disrupted
endometrial immune niche ultimately impairing embryo implantation
(101). This subsection
systematically elaborates on the triggers of TIAR, its mediated
pathological cascades and the subsequent amplifying loops that
sustain AM development.
TIAR activation in AM arises from two primary
sources of mechanical stress. Endogenously, supraphysiological
uterine peristalsis and increased intrauterine pressure (a hallmark
of AM) directly damage myocytes and fibroblasts within the JZ, a
critical physical barrier between the endometrium and myometrium
(102), and this mechanical
damage initiates the TIAR cascade. Exogenously, iatrogenic injuries
(such as curettage, childbirth, induced abortion and cesarean
section) impose supraphysiological mechanical strain on the uterus,
directly inducing COX-2 and CXCL8 expression in fibroblasts
(4). Even mild mechanical
stretching of cultured fibroblasts has been shown to activate
COX-2, promote PGE2 and CXCL8 production and thereby trigger TIAR
(4). Notably, iatrogenic damage
often impairs the JZ (103); as
the JZ serves as a barrier to prevent endometrial invasion, its
disruption further facilitates endometrial penetration into the
myometrium and increases AM risk following intrauterine procedures
(104).
Once triggered, the TIAR cascade drives AM
progression through a sequential pathological process. First,
TIAR-induced microtrauma leads to myometrial fissures, enabling the
ectopic displacement of endometrial fragments (105). These ectopic fragments are
recognized by the immune system as damaged tissue-initiating
lesion-specific inflammatory priming characterized by IL1B/IL-6
upregulation, platelet aggregation and HIF-1α stabilization, which
propagates local-to-systemic inflammatory cascades (102). At the molecular level, IL1B
plays a central role in amplifying inflammation; it activates COX-2
transcription via a mitogen-activated protein kinase
(MAPK)-dependent pathway, which enhances COX-2 binding to the cAMP
response element, recruiting immune cells and further amplifying
the inflammatory cascade (106), and it activates the ERK1/2 and
NF-κB signaling pathways to induce COX-2 expression; elevated COX-2
then promotes PGE2 synthesis (9), which in turn upregulates IL-6 and
CXCL8 expression, creating a positive feedback loop to sustain
inflammation.
Beyond inflammatory amplification, the TIAR-mediated
response interacts with estrogen metabolism to form a vicious cycle
that accelerates AM chronicity. PGE2 upregulates the expression of
steroidogenic acute regulatory protein and CYP19A1 to enhance local
E2 biosynthesis (67); E2
further activates estrogen receptors (ERα/ERβ) to promote cell
proliferation while simultaneously trans-activating the COX-2 gene,
thus increasing PGE2 production and perpetuating inflammation.
Notably, ectopic lesions (predominantly localized to the
anterior/posterior uterine walls and fundal-cornual regions with
concentrated mechanical stress) exacerbate uterine
hyperperistalsis, which reinforces this TIAR-inflammation-estrogen
loop and in turn drives disease progression (107).
EMT, fibroblast-to-myofibroblast
transdifferentiation (FMT) and fibrosis: Pathogenic roles in AM and
impacts on pregnancy
EMT is a biological process in which epithelial
cells lose polarity and intercellular junctions, gaining
mesenchymal characteristics, including enhanced motility and
invasiveness (108). Hallmarks
of EMT include downregulation of E-cadherin and upregulation of
N-cadherin and vimentin, resulting in the loss of epithelial
polarity and increased invasive potential. In AM, EMT serves as a
key driver of ectopic lesion formation by enabling endometrial
cells to traverse the EMJ and establish adenomyotic lesions
(67), while also exacerbating
disease progression by promoting fibrosis and uterine
dysfunction.
Multiple cytokines and signaling pathways
orchestrate EMT in AM. CXCL8 activates the FAK/PI3K/AKT pathway to
enhance the proliferation, migration and invasiveness of eutopic
ESCs (34), supporting their
survival in ectopic environments (59). Macrophage-induced IL-6
overactivation in human endometrial cells suppresses E-cadherin
promoter activity and upregulates N-cadherin/vimentin via the
JAK2/STAT3 pathway, directly inducing EMT (67). IL1B activates the Wnt/β-catenin
pathway, a key regulator of EMT, with COX-2 further amplifying this
effect to drive AM progression (109,110). Hypoxic conditions in the
ectopic microenvironment also induce HIF-1α expression, which
upregulates IL-6, CXCL8 and CXCL12. CXCL12 promotes angiogenesis
via its receptor CXCR4, while IL-6 and CXCL8 amplify inflammatory
and pro-fibrotic signals (103).
Additionally, Notch1 is upregulated in ectopic
endometrium and indirectly promotes EMT by modulating pathways such
as NF-κB, leading to N-cadherin/Snail/Slug upregulation and
E-cadherin loss; downregulation of Numb, a negative regulator of
Notch, may further exacerbate EMT-driven fibrosis and lesion
invasion (111). Although the
direct role of IL-10 in AM-associated EMT remains unstudied, its
EMT-promoting effects in cancer and renal cells suggest potential
relevance (112); notably,
thymus-expressed chemokine derived from embryonic stem cells and
macrophages upregulates Tregs in the ectopic microenvironment,
promoting IL-10/TGF-β1 secretion that suppresses NK cell
cytotoxicity and Th1-type immunity, creating an immunosuppressive
niche that protects ectopic endometrial cells from clearance
(113). Beyond driving ectopic
lesions, EMT collaborates with fibrosis to worsen AM pathogenesis,
as fibrosis in AM arises from dysregulated wound healing. Unlike
the regenerative phase of normal tissue repair (where injured cells
are replaced without scarring), AM is characterized by a fibrotic
phase in which parenchymal tissue is replaced by fibrotic
connective tissue, accompanied by excessive extracellular matrix
(ECM) deposition and remodeling imbalance (114).
EMT and FMT are central to this process, driving
lesion stabilization and myometrial thickening (115), while ectopic ESCs directly
interact with the myometrium to induce SMC fusion and abnormal
expansion, altering uterine morphology and function. As key
fibrotic regulators, macrophages orchestrate EMT, FMT and SMC
modulation via M1-M2 polarization and activation of the canonical
TGF-β/Smad3 pathway, synergistically promoting ECM collagen
synthesis; during phagocytic clearance of cellular debris, they
also regulate ECM turnover via the dynamic balance between MMPs and
their TIMPs, mediating the degradation-deposition cycle of ECM
reconstruction (84,116), a pathway that further
cross-talks with PI3K/AKT and MAPK signaling to amplify
pro-fibrotic effects. Under TIAR mechanisms, ectopic endometrial
cells undergo cyclic hemorrhage-induced chronic inflammation,
triggering EMT, FMT and SMC modulation to form fibrotic scars, with
fibrosis markers such as α-smooth muscle actin and type I collagen
positively correlating with uterine volume, linking fibrosis
severity to disease progression (114).
From a reproductive perspective, EMT and fibrosis
disrupt AM-associated reproductive function via multiple pathways:
They cause structural disruption at the EMJ, interfering with
embryo implantation and placental development; induce uterine
hyperperistalsis, impairing embryo and gamete transport (117); and promote excessive scar
formation that increases intrauterine adhesion risk, ultimately
contributing to infertility (118). The integrated regulatory
network linking these key pathogenic processes (inflammatory
cytokine signaling, TIAR cascades, EMT, FMT and fibrosis) to
AM-related reproductive dysfunction is systematically summarized in
Fig. 3.
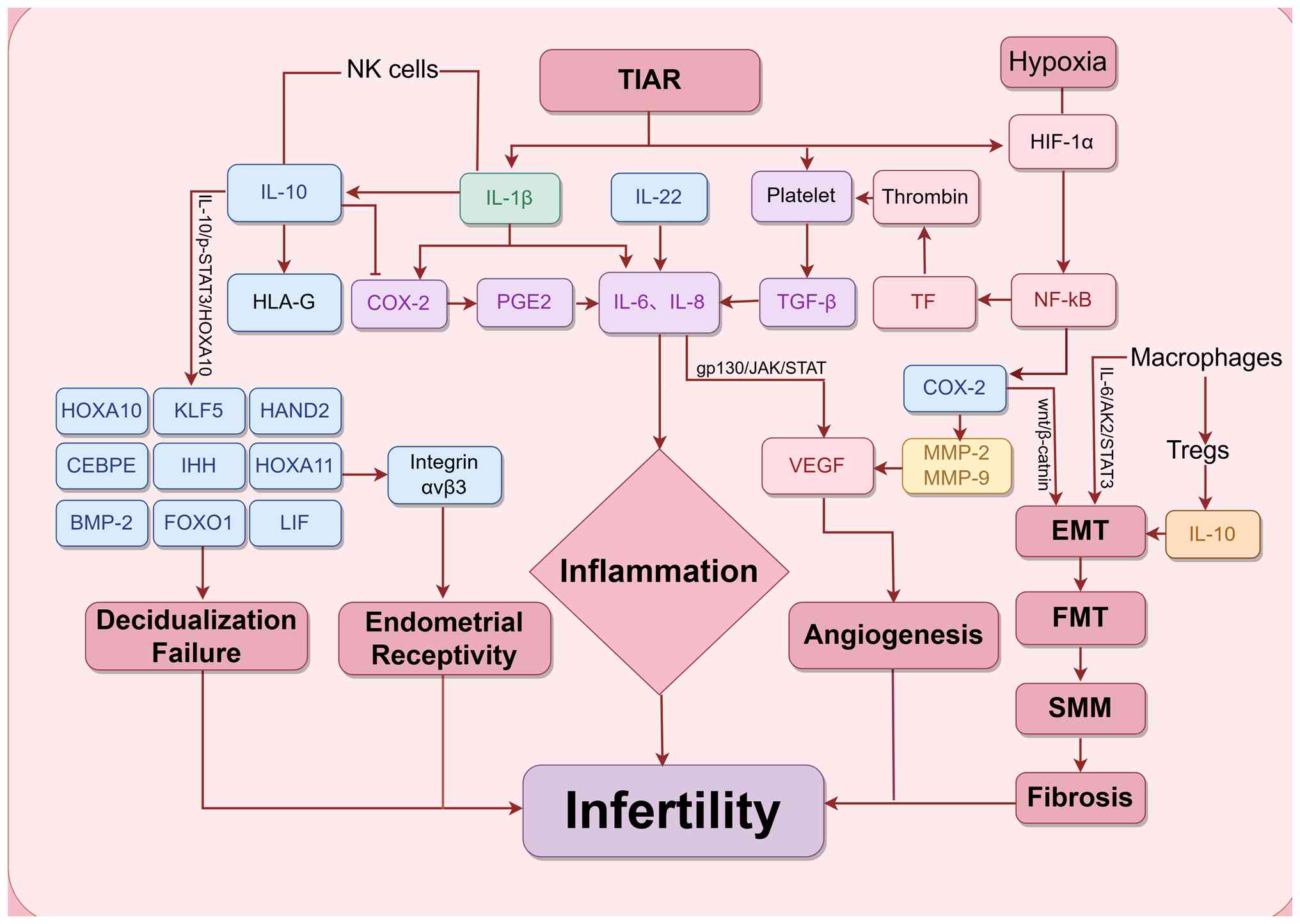 | Figure 3Overview of interactions between
inflammatory cytokines in adenomyosis-related infertility in the
present review. However, these interactions do not necessarily
represent the actual or complete biological pathways. NF-κB,
nuclear factor κB; HIF1A, hypoxia-inducible factor 1α; VEGF,
vascular endothelial growth factor; TGF-β, transforming growth
factor β; MMP, matrix metalloproteinase; HLA-G, human leucocyte
antigen-G; COX2, cyclooxygenase2; PGE2, prostaglandin E2; TF,
tissue factor; BMP-2, bone morphogenetic protein-2; CEBPE,
CCAAT/enhancer-binding protein ε; EMT, epithelial-mesenchymal
transition; FMT, fibroblast-myofibroblast transition; FOXO1,
forkhead box O1; HAND2, heart and Neural Crest Derivatives
Expressed 2; HOXA10, homeobox A10; HOXA11, homeobox A11; IHH,
Indian hedgehog; IL, interleukin; JAK, Janus kinase; KLF5,
Kruppel-like factor 5; LIF, leukemia inhibitory factor; NK cells,
natural killer cells; gp130, glycoprotein 130; SMM, smooth muscle
metaplasia; STAT, signal transducer and activator of transcription;
TIAR, tissue injury and repair; Tregs, regulatory T cells. |
Impact of inflammatory cytokines on
pregnancy outcomes in AM
Impact of neovascularization on
pregnancy
In AM, aberrantly elevated pro-angiogenic factors,
such as VEGF and FGF, and reduced anti-angiogenic activity drive
excessive neovascularization. This not only provides nutritional
support for ectopic lesions [facilitates their invasion, survival
and proliferation to form a vicious cycle (119)] but also causes structural
abnormalities in neovessels. These fragile vessels are prone to
rupture, leading to abnormal uterine bleeding, disrupted
endometrial homeostasis and impaired ER (120). The aberrant vascular network
further interferes with embryo positioning and adhesion,
contributing to implantation failure.
Pathological angiogenesis in AM is primarily
triggered by TIAR, closely linked to vascular damage from cyclical
endometrial bleeding (121).
Under physiological conditions, endometrial repair follows a
four-phase process: Hemostasis, inflammation, proliferation and
remodeling (122). This ordered
process ensures proper tissue recovery without persistent damage.
However, in pathological states, this repair process is disrupted.
The disorganized repair leads to vascular structures, which in turn
exacerbate local inflammation and fibrosis and perpetuate a vicious
cycle of tissue injury and dysregulated repair.
In the initial phase of repair, activated platelets
drive hemostasis (123) and
release bioactive molecules [such as thromboxane A2,
5-hydroxytryptamine, PDGF and epidermal growth factor (EGF)] that
upregulate pro-inflammatory genes including VEGF, COX-2 and MMP-9,
thereby creating a pro-angiogenic microenvironment (124). Notably, VEGF, a key specific
marker of angiogenesis, not only directly promotes endothelial cell
proliferation but is also elevated in the follicular fluid of
patients with EMs, and clinically increased VEGF levels are
significantly associated with poor ovarian response, reduced oocyte
maturation rates and higher miscarriage risk (125).
Moreover, platelets secrete platelet-derived growth
factor, EGF and FGF to directly stimulate endothelial proliferation
and recruit macrophages and neutrophils to the lesion site; these
immune cells subsequently release inflammatory cytokines (such as
IL-6, IL-10, CXCL8, TGF-β, IL-22 and NF-κB) to amplify the local
inflammatory response (Fig. 4)
(126,127). Platelets also upregulate tissue
factor, which binds to circulating factor VIIa to activate the
coagulation cascade. The resultant thrombin generation promotes
sustained platelet activation, forming a 'coagulation-inflammation'
positive feedback loop (128).
Salim et al (129)
confirmed the upregulation of these angiogenesis- and
inflammation-related factors in adenomyotic lesions, supporting
increased pathological neovascularization. Growing evidence
indicates that platelets are not merely hemostatic cells but also
key immunoregulatory agents (130) whose interaction with ectopic
lesions fosters a microenvironment supporting lesion progression
and fibrosis (131).
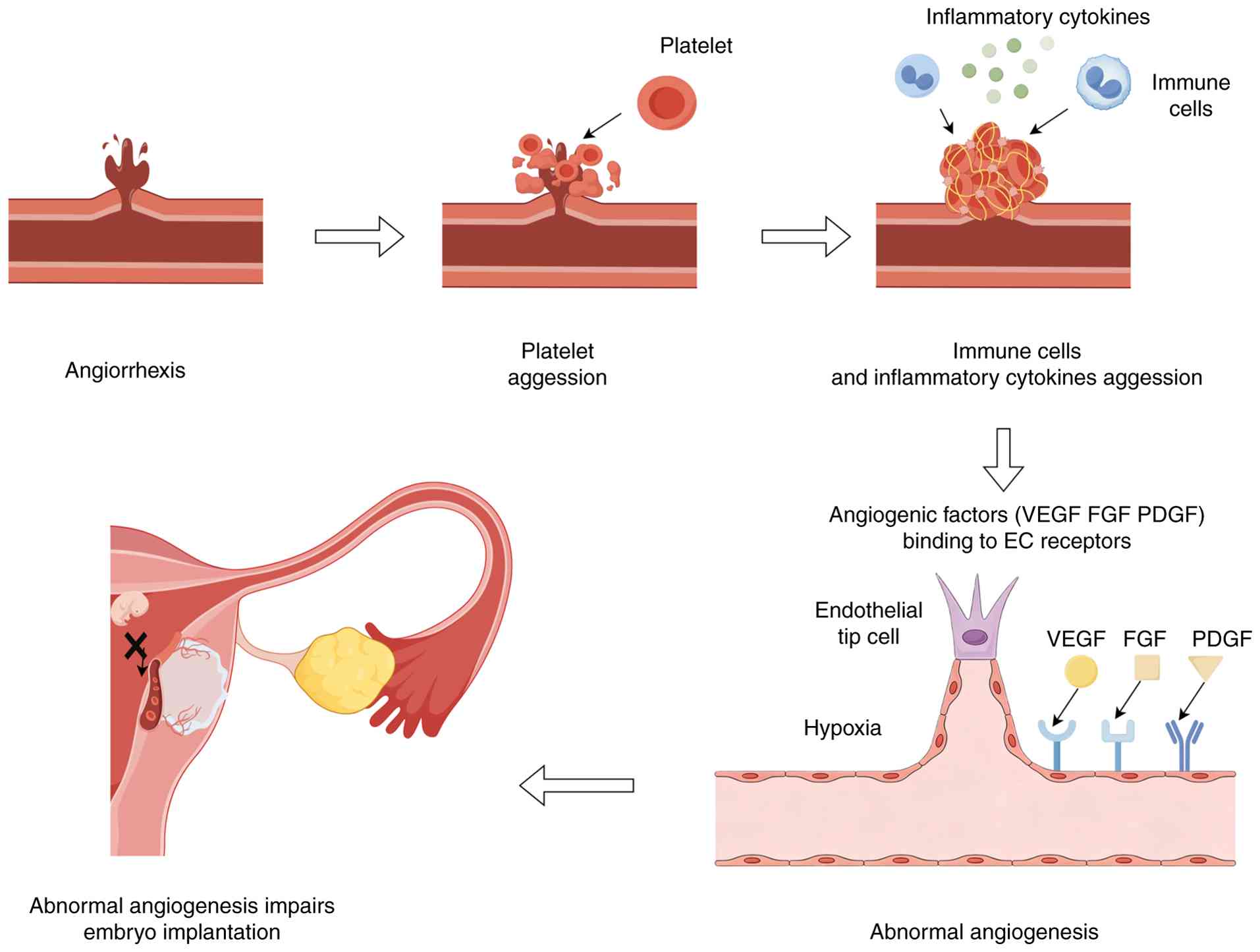 | Figure 4Main process of angiogenesis. First,
endometrial bleeding occurs periodically as a sign of tissue
damage. Second, platelet adhesion and aggregation participate in
hemostasis. Third, platelets attract inflammatory cytokines and
immune cells to gather at the wound. Fourth, inflammatory cytokines
act as angiogenic factors to induce neovascularization of ectopic
endometrium and upregulate factors directly related to angiogenesis
such as VEGF, FGF and PDGF. Among them, hypoxia is a crucial
inducing factor. Fifth, abnormal angiogenesis leads to decreased
endometrial receptivity and affects embryo implantation. EC,
endothelial cell; FGF, fibroblast growth factor; PDGF,
platelet-derived growth factor; VEGF, vascular endothelial growth
factor. |
The interplay between angiogenesis, immune cells and
inflammation is well-recognized (132). Angiogenesis is modulated by
immune cells and inflammatory mediators (133-135) and reciprocally influences
immune responses. Wei et al (136) demonstrated that IL-6 promotes
pathological angiogenesis by upregulating VEGF via activating the
STAT3 signaling pathway. Clinical data reveal a strong positive
correlation between serum IL-6 levels and lesion microvessel
density, indicating the potential of IL-6 as a biomarker for
disease progression (137).
Thymic stromal lymphopoietin (TSLP) induces CXCL8 secretion in
cervical cancer cells via the ERK1/2 pathway to stimulate human
umbilical vein endothelial cell proliferation and angiogenesis, and
this mechanism appears to be recapitulated in the ectopic
endometrium of AM, suggesting TSLP as a potential therapeutic
target (138,139). IL1B enhances CXCL8 and VEGF
expression to promote invasive migration of ESCs and facilitate
immune cell extravasation by upregulating ICAM-1 and VCAM-1,
thereby driving tissue remodeling (140). Silencing the COX-2 gene
significantly reduces VEGF and MMP-9 expression in ectopic
endometrium, underscoring the pivotal role of COX-2 as an upstream
regulator in AM (141).
Additionally, clinical specimens show a strong correlation between
COX-2 positivity, dysmenorrhea severity and lesion volume (58). As a central transcription factor,
NF-κB directly regulates COX-2, VEGF, MMPs and the plasminogen
activator system (142,143), and mediates endothelial
inflammation induced by IL1B and TNF-α (144), highlighting the central role of
the COX-2/NF-κB axis.
Compared with normal endometrium, IL-10 expression
in adenomyotic tissues is reduced by 62%, which impairs its
regulatory effect on COX-2, MMP-2 and MMP-9 and contributes to
uncontrolled angiogenesis (145); this dysregulation is closely
associated with impaired ER due to an abnormal IL-10/IL-6 ratio
during the implantation window. IL-22, a member of the IL-10
cytokine family, enhances stromal cell invasiveness by upregulating
VEGF, IL-6 and CXCL8 expression (146), and clinical data show that
serum IL-22 levels are significantly elevated in infertile patients
and inversely correlated with embryo implantation rates during IVF
cycles (147). Fischer et
al (148) confirmed by
immunohistochemistry that eutopic endometrial cells in AM exhibit
heightened sensitivity to IL-1, with increased expression of CXCL8,
CXCL8 receptor and VEGF, indicating that excessive
neovascularization impairs ER. The expression of angiogenic markers
in both eutopic and ectopic endometrium suggests that angiogenesis
not only facilitates invasion of normal endometrial tissue but also
supports the maintenance of ectopic lesions, thereby serving as a
critical factor in implantation failure (128).
Furthermore, tissue injury-induced vascular
hyperpermeability, extravasation of blood components and increased
interstitial fluid volume (149) may contribute to common clinical
symptoms in patients with AM such as abnormal uterine bleeding and
infertility (150). Perfusion
loss and subsequent hypoxia from vascular injury activate the
HIF-1α/NF-κB pathway, which robustly upregulates VEGF expression
(151) and drives the
COX-2-PGE2-P450 aromatase positive feedback loop, leading to
abnormal uterine peristalsis; clinical data show a strong
correlation between COX-2 and VEGF expression (152). During this process, upregulated
PGE2 activates P450 aromatase to promote local estrogen synthesis,
which in turn induces EMT and increases vascular permeability,
driving ectopic endometrial cells into a pro-angiogenic state
(67,153). Additionally, extravasated
platelets may provide a protective barrier for ectopic stromal
cells, shielding them from physical clearance and immune
surveillance (133).
Regulation of ER impairment in AM
Regulatory basis of normal ER and
hormonal-immune imbalance in AM
Embryo implantation depends on the dynamic
hormonal-immune balance established at the maternal-embryo
interface (154). The ability
of the endometrium to support normal embryo implantation, referred
to as ER, is primarily governed by the precisely timed and
transient window of implantation (WOI) during the mid-secretory
phase. During this period, an immunosuppressive state dominated by
NK cells helps prevent maternal immune rejection of the
semi-allogeneic embryo (155).
However, in patients with AM, this hormonal-immune balance is
disrupted, characterized by aberrant expression of pro-inflammatory
cytokines, chemokines and immunoregulatory factors. This disruption
leads to impaired ER and reduced sensitivity to progesterone
(156). Furthermore, delayed
embryo implantation beyond the WOI is perceived as embryo
malposition by the maternal system, increasing the risk of placenta
previa, abnormal placentation and placental insufficiency, all of
which can result in preterm birth, intrauterine growth retardation
and even preeclampsia (157).
Immune-cytokine alterations and key
gene dysregulation in the AM endometrium
Both in natural and stimulated cycles, the
endometrium of patients with AM demonstrates characteristic
alterations in immune cell infiltration and cytokine profiles,
including increased macrophage infiltration, elevated IL-6
expression and reduced expression of IL-11 and its receptor, key
regulators of implantation (158). Notably, IL-6 receptors are
widely expressed on blastocyst trophectoderm and endometrial
epithelial cells, indicating that chronic local inflammation may
impair embryo-endometrium crosstalk and directly compromise
receptivity (159). Moreover,
COX-2 is markedly upregulated in perivascular regions of the
endometrium; it aggravates inflammation via the induction of IL-6
and CXCL8, while also promoting aberrant angiogenesis, collectively
disrupting the microenvironment required for implantation. These
findings position COX-2 as a potential biomarker of impaired ER
(160). Immune tolerance during
the WOI also depends on adequate IL-10 secretion; however,
significantly reduced IL-10 levels have been observed in the
decidua of patients with AM and this deficiency has been associated
with recurrent miscarriage and preeclampsia (161). Additionally, uNK cells exhibit
functional abnormalities, including decreased secretion of VEGF and
placental growth factor, which may impair trophoblast migration and
angiogenesis at the implantation site (162). Other inflammatory mediators,
such as IL1B and corticotropin-releasing hormone, are also
abnormally elevated in the endometrium of affected patients,
further contributing to implantation failure (19).
Beyond immune cell and cytokine dysregulation,
intrinsic molecular regulators of ER, specifically homeobox genes,
are also significantly disrupted in AM. Homeobox genes HOXA10 and
HOXA11 serve as essential regulators of ER; they promote embryo
adhesion by regulating β3-integrin expression and the formation of
stromal cell pinopodes (163).
A clinical study has shown that patients with implantation failure
often exhibit downregulation of HOXA10 and HOXA11 expression
(164). In AM, both HOXA10 and
HOXA11 are significantly downregulated, while HIF-2α is
upregulated. Pharmacological inhibition of HIF-2α has been shown to
restore HOX gene expression (165), suggesting that a hypoxic
microenvironment impairs ER through the HIF-2α-HOX axis (166). Notably, this link between HOX
genes and ER is further supported by animal studies. Specifically,
Hoxa10/A11-deficient mice exhibit reduced endometrial gland
formation and decreased LIF secretion (164).
LIF, a member of the IL-6 cytokine family, is
essential for blastocyst development, decidualization and
trophoblast differentiation (167) and it regulates implantation via
the STAT3/ERK signaling pathway (168). In AM, aberrant LIF expression
levels, together with dysregulated IL-11, contributes to impaired
gp130-mediated signal transduction (157). Furthermore, disruption of the
IL-10/phosphorylated-STAT3/HOXA10 signaling axis has been strongly
linked to infertility and early pregnancy loss (54). The IL-33/ST2 pathway also
regulates HOXA10 expression and participates in the decidualization
process (156). Additionally,
the expression of β3-integrin and its ligand osteopontin (OPN),
both essential for blastocyst adhesion, is significantly reduced in
the endometrium of patients with AM (169). This reduction may be driven by
IL-6/CXCL8-mediated downregulation of αvβ3 integrin expression
(170). HOXA10 directly
promotes β3-integrin expression, while OPN binds to CD44 receptors
to enhance blastocyst adhesion. Impaired function of both molecules
further exacerbates implantation failure.
ER-related signaling pathway
dysregulation and potential therapeutic targets
Aberrant activation of the NF-κB signaling pathway
in AM induces the production of TNF-α, IL1B and IL-6, molecules
that damage the endometrial epithelium and reduce implantation
rates (171). In animal models,
NF-κB activation during the WOI has been shown to interfere with
LIF expression, thereby impairing ER and adversely affecting
pregnancy outcomes (172).
Although the direct connection between ER and endometrial fibrosis
remains unclear, a preclinical study has highlighted a potential
therapeutic direction: Treatment with anti-TGF-β1 improves both
fibrosis and pregnancy outcomes in AM models (173). This suggests that TGF-β1 may
serve as a potential therapeutic target for restoring receptivity
as it can modulate both fibrosis and the immune microenvironment
(174).
Abnormal decidualization of the
endometrium
Normal decidualization process and
core defects in AM
Decidualization is a critical process for embryo
implantation and placental development (175); it refers to the transformation
of ESCs into specialized decidual cells with secretory functions
under progesterone regulation, which further establishes an
immunotolerant microenvironment, participates in embryonic nutrient
supply and maintains maternal-fetal interface homeostasis (157). Physiologically, decidualization
follows a two-phase dynamic progression: The early acute
inflammatory phase relies on chemokine-mediated immune cell
recruitment to support stromal remodeling, while the subsequent
anti-inflammatory phase establishes maternal-fetal immune tolerance
(157). Transcriptomic analyses
by Salker et al (176)
showed that during the initiation phase (day 2) of decidualization,
70 cytokines/receptors were significantly upregulated, whereas only
12 key regulators (including IL-11Rα and LIFR) remained highly
expressed in the terminal differentiation phase (day 8).
However, in patients with AM, decidualization is
frequently impaired, primarily driven by pathological factors such
as chronic inflammation, oxidative stress and immune dysregulation,
all of which are core mechanisms underlying infertility and adverse
pregnancy outcomes (177). A
hallmark pathological feature of AM, uterine JZ disruption, induces
upregulation of pro-inflammatory cytokines (such as IL-6, IL1B and
CXCL family chemokines) and accumulation of reactive oxygen species
(ROS); this disrupts the pro-inflammatory/anti-inflammatory balance
essential for normal decidualization (178,179). Notably, the aforementioned
temporal dysregulation of cytokine expression further exacerbates
decidualization defects in AM.
Key pathogenic mechanisms of
decidualization impairment in AM
Abnormalities in inflammatory mediators are central
to AM-associated decidualization defects. IL-6 is highly expressed
in decidual cells of adenomyotic tissues (180); it promotes ectopic endometrial
proliferation via the gp130/JAK/STAT3 axis while suppressing
progesterone-induced decidualization genes such as KLF5 and FOXO1,
thereby inhibiting stromal cell differentiation (181). The STAT3 pathway, critical for
embryo adhesion, decidual transformation (68), ovarian steroidogenesis,
follicular development, endometrial decidualization and early
events of embryo implantation (182), exhibits aberrant activation in
AM, potentially reducing fertility and increasing miscarriage risk
(183). IL1B is physiologically
required for embryo implantation by upregulating integrin αvβ3 and
mediating adhesion through the COX-2/PTGS2/PGE2 axis, but it is
abnormally elevated in AM (184,185). This elevation triggers
NF-κB-driven hyperinflammation, which impairs early
decidual-trophoblast and chorionic-decidual interactions (186), reduces ER and contributes to
placental insufficiency, a mechanism that also links EMs to adverse
pregnancy outcomes (187).
Additionally, chemokines such as MCP-1, CXCL1/2/3 and MIP-3α are
aberrantly upregulated in AM (188). This upregulation induces
excessive uNK cell infiltration and disrupts T-cell polarization,
which in turn impairs embryo selection and implantation failure
(189). MCP-1, in particular,
acts as a major immune effector in this process (66).
Beyond abnormal inflammatory mediators, the
expression of multiple key genes regulating decidualization is
significantly downregulated in the endometrium of patients with AM.
Reduced expression of embryo implantation-related genes such as
FOXO1, KLF5, CEBPB and HAND2 directly impairs decidual cell
differentiation and lowers ER (180). Diminished bone morphogenetic
protein 2 secretion decreases the expression of HOXA10 and LIF in
ESCs, affecting embryo positioning and trophoblast invasion
(190). Inadequate expression
of EGF and heparin-binding EGF-like growth factor (HB-EGF) impairs
β3 integrin expression, weakens EMT and disrupts paracrine
regulation during the early phase of decidualization (191). Moreover, co-culture of
decidualized human ESCs with developmentally arrested embryos
reduces the secretion of key implantation modulators (including
IL1B, HB-EGF, IL-6 and IL-10) (192). Since decidual factor secretion
is critical for screening viable embryos, this finding suggests
that patients with AM and recurrent miscarriage may lack the
ability to select viable embryos.
Immune homeostasis imbalance at the maternal-fetal
interface is another key mechanism underlying impaired
decidualization in AM. Trophoblast cells lack classical MHC-I
antigens to avoid maternal T-cell attack, but this renders them
susceptible to surveillance by decidual uNK and
lymphokine-activated killer cells (193). In AM, reduced infiltration of
decidual Treg cells disrupts immune balance, enhancing the activity
of cytotoxic T cell and NK cells (194) and increasing the risk of
recurrent miscarriage. uNK cells, which are essential for
regulating trophoblast invasion into the JZ and spiral artery
remodeling (195), rely on
HLA-C/KIR interactions for maternal recognition of the
semi-allogenic fetus (193).
Decidualization defects in AM alter HLA-C expression and disrupt
the ligand-receptor balance, thereby impairing immune tolerance.
Additionally, cytokines critical for decidualization and immune
regulation (such as IL-11, IL-15 and IL-33) are abnormally
expressed in AM (176,196,197). Dysregulation of IL-11 impairs
the decidualization process, while upregulated IL-15 drives
abnormal uNK cell proliferation and exacerbates inflammation at
ectopic endometrial lesions (198). The imbalance between MMPs and
their TIMPs in the decidua of patients with AM also contributes to
infertility and decidualization defects (199).
Additionally, elevated thrombin levels, which are
observed in patients with AM and menorrhagia, are also closely
associated with impaired decidualization. Thrombin exerts multiple
deleterious effects on decidualization; it upregulates
pro-inflammatory chemokines, induces endothelial dysfunction and
hyperpermeability, promotes platelet aggregation, suppresses
prolactin secretion (a marker of decidualization) and alters the
morphology of decidualized stromal cells (200). Thrombin also activates genes
involved in ECM degradation and chemokine production, further
exacerbating decidualization defects. This effect is part of the
broader multi-pathway regulatory network (encompassing tissue
injury and repair cascades, macrophage polarization, EMT,
angiogenesis dysregulation and reduced endometrial receptivity)
that converges to drive infertility in AM, as systematically
summarized in Fig. 5.
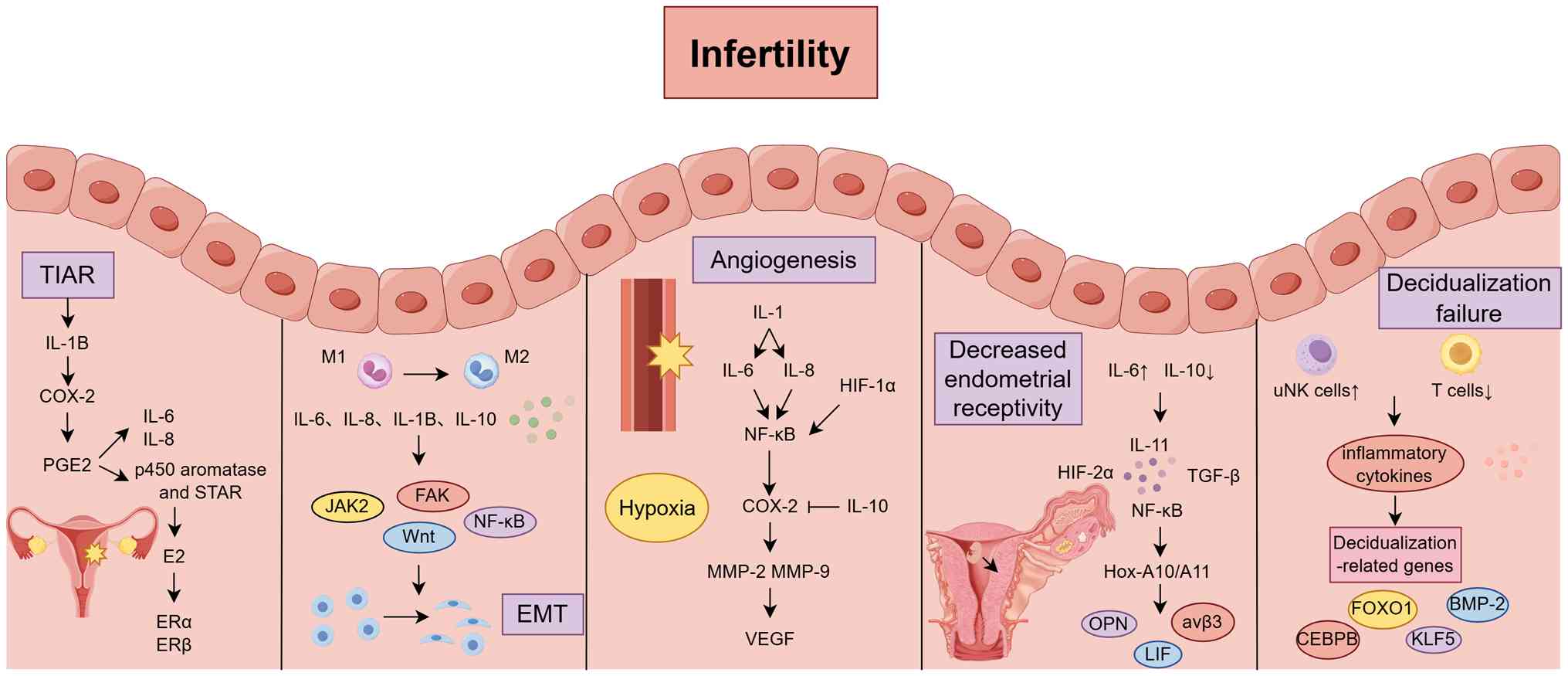 | Figure 5Main pathogenesis of
adenomyosis-related female infertility. Inflammatory cytokines
mainly promote the occurrence and development of adenomyosis by
promoting TIAR and EMT and interfere with the normal embryo
implantation process by promoting angiogenesis, leading to
decreased endometrial receptivity and impaired decidualization.
BMP-2, bone morphogenetic protein-2; CEBPB, CCAAT/enhancer-binding
protein β; COX-2, cyclooxygenase-2; E2, estradiol; EMT,
epithelial-mesenchymal transition; ERα, Estrogen Receptor Alpha;
ERβ, Estrogen Receptor β; FAK, Focal Adhesion Kinase; FOXO1,
Forkhead Box O1; HIF-1α, hypoxia-inducible factor-1α; Hox-A10/A11,
homeobox A10/A11; IL, interleukin; JAK2, Janus kinase 2; KLF5,
Kruppel-like factor 5; LIF, leukemia inhibitory factor; MMP, matrix
metalloproteinase; OPN, osteopontin; PGE2, prostaglandin E2; STAR,
steroidogenic acute regulatory protein; TGF-β, transforming growth
factor-β; TIAR, tissue injury and repair; uNK cells, uterine
natural killer cells; VEGF, vascular endothelial growth factor. |
Clinical medications targeting inflammatory
cytokines for the treatment of AM-related infertility
Non-steroidal anti-inflammatory drugs
(NSAIDs)
NSAIDs exert therapeutic effects in AM primarily by
inhibiting COX activity and subsequently reducing prostaglandin
synthesis (201). Studies have
demonstrated that NSAIDs can decrease the depth of endometrial
infiltration into the myometrium, inhibit the migration and
invasion of ESCs and induce apoptosis (202,203). In addition, NSAIDs suppress key
pathological processes such as fibrosis, angiogenesis and EMT
(204). Therefore, NSAIDs are
considered promising pharmacological candidates for the clinical
management of AM.
Celecoxib, a highly selective COX-2 inhibitor,
significantly reduces inflammatory responses and the depth of
lesion infiltration in EMs by suppressing the expression of
pro-inflammatory cytokines such as IL1B, IL-6 and TNF-α, as well as
the transcription factor NF-κB (205). Studies have suggested that in
ART, localized inflammatory responses may impair embryo
implantation by inducing abnormal uterine contractility and
reducing ER via prostaglandin-mediated mechanisms (10,206). Theoretically, NSAIDs may
improve ART outcomes by mitigating the detrimental effects of
prostaglandins; however, this hypothesis requires further
experimental validation (207).
Hormonotherapy
Gonadotropin-releasing hormone
(GnRH)
GnRH is a hypothalamic peptide hormone that acts on
the anterior pituitary via the hypophyseal portal system to
regulate the synthesis and pulsatile secretion of
follicle-stimulating hormone and luteinizing hormone. A study by
Zippl et al (89)
reported no significant differences in endometrial immune
characteristics between women with AM and those with recurrent
pregnancy loss or RIF. The study also found that GnRH-a suppress
the production of inflammatory cytokines such as TNF-α and IL1B by
modulating the mononuclear phagocyte system, thereby inhibiting
inflammation and angiogenesis. Similarly, Khan et al
(208) demonstrated that GnRH-a
treatment downregulates the secretion of cytokines (such as IL1B,
MCP-1 and VEGF) from adenomyotic endometrial cells, attenuates the
immune response of von Willebrand factor and improves the
inflammatory microenvironment, ultimately enhancing ER.
Furthermore, GnRH-a therapy has been shown to
upregulate the expression of key receptivity markers in the
endometrium, including HOXA10, HOXA11, integrin β3 and LIF, thereby
promoting embryo implantation (209). Several case reports have
confirmed successful pregnancies in previously infertile women with
AM following GnRH-a treatment (210,211). Notably, the combination of
GnRH-a with surgical intervention appears to yield superior
outcomes. In a large prospective study, 55 out of 165 women with AM
conceived after surgery alone or surgery followed by GnRH-a
therapy, achieving a clinical pregnancy rate of 77.5%, with 69.0%
delivering successfully by the end of a 2-year follow-up period
(212).
GnRH antagonists (GnRH-As)
GnRH-As exert their effects by competitively
blocking GnRH receptors in the pituitary gland, thereby directly
inhibiting the secretion of gonadotropins. Compared with GnRH-a,
GnRH-As offer the advantage of avoiding the initial 'flare-up
effect' (a transient surge in gonadotropins at the onset of
treatment) and allow for a more rapid recovery of ovarian function
after drug withdrawal, making them particularly suitable for
patients desiring fertility preservation (213). Currently available GnRH-As
include elagolix, relugolix and linzagolix. A clinical study has
shown that the administration of GnRH-A prior to ART procedures or
surgical intervention may improve embryo transfer outcomes and
increase surgical success rates (209).
Lessey et al (214) demonstrated that treatment with
elagolix for 2 months led to the upregulation of several
anti-inflammatory mediators, including IL-10, IL-13, CCL18 and
TWIST2. Simultaneously, miRNA-mediated downregulation of
inflammatory components such as NOTCH1, NF-κB, T cells and NK cells
was observed, suggesting that elagolix significantly modulates the
systemic inflammatory milieu. Moreover, elagolix has been
associated with higher pregnancy and live birth rates compared with
oral contraceptive pills, although further research is warranted to
confirm these findings (215).
Dienogest (DNG)
DNG is a progestin with high selectivity for PRs
that exerts mild ovulation-inhibiting effects; it can directly
suppress the proliferation of AM stromal cells and induce
apoptosis. As the primary target of DNG, the activation of the PR
also regulates inflammation in the endometrium (216). A prospective study confirmed
that dienogest and danazol can alleviate the production of
TNF-α-induced IL-6 and CXCL8 in endometriotic stromal cells by
inactivating NF-κB (217). Oral
administration of dienogest also increases the number of
infiltrating NK cells in ectopic endometrial glands, benefiting
pregnancies that occur after cessation of treatment in terms of
embryo implantation and fetal protection (218).
Immunomodulators
Immunomodulators represent a promising therapeutic
strategy for preserving fertility as they target inflammatory
responses, modulate the immune microenvironment and inhibit the
growth of ectopic lesions. Most notably, these agents can improve
tubal patency, enhance oocyte quality and restore the pelvic
microenvironment without interfering with estrogen secretion or
ovulatory cycles, making them an optimal choice for patients
seeking to retain reproductive potential (219).
IL-targeted therapies
A study has demonstrated that tocilizumab, an IL-6
receptor antagonist, can effectively inhibit ectopic lesion cell
proliferation and migration induced by endometrial cell-derived
exosomes (220). Tocilizumab
induces G1/S cell cycle arrest and significantly suppresses
activation of the IL-6/JAK2/STAT3 signaling axis, thereby exerting
potent anti-inflammatory and antiproliferative effects (25).
AMY109, a long-acting circulating antibody
targeting CXCL8, has shown promising antitumor activity by reducing
fibrosis and immunosuppression in cancer models (221). In a non-human primate model of
EMs, AMY109 significantly reduced the volume of nodular lesions and
improved fibrosis and adhesions; its mechanism is associated with
the inhibition of neutrophil recruitment and MCP-1 production, and
notably, it does not impair ovarian function (222).
The classic antidiabetic drug metformin has
demonstrated therapeutic potential in AM (223); it exerts beneficial effects by
modulating oxidative stress, inflammation and apoptosis.
Specifically, metformin downregulates pro-inflammatory cytokines
(including IL-6 and CXCL8), angiogenic factors (including VEGF and
MMP-9), and simultaneously upregulates LIF and HOXA10, thereby
enhancing ER and supporting implantation (224). A clinical study has confirmed
that the combination of high-intensity focused ultrasound and
metformin significantly reduces uterine volume and lowers
recurrence rates in AM, largely due to the suppression of
inflammatory mediators (225).
Anakinra, an IL-1 receptor antagonist that
competitively binds to IL-1R1, has been shown in a clinical study
to significantly reduce visual analogue scale (VAS) pain scores and
improve quality of life in patients with EMs, without adversely
affecting ovulatory function or menstrual regularity during
treatment (226).
Lipoxin A4, an anti-inflammatory and pro-resolving
lipid mediator, significantly reduces the size of endometriotic
lesions by downregulating pro-inflammatory cytokines (including
IL1B and IL-6) and inhibiting the expression of COX-2 and PGE2 in
both lesions and peritoneal fluid cells (227). Additionally, it suppresses the
expression of angiogenic factors (such as VEGF, HIF-1α and MMP-9),
thereby inhibiting neovascularization, and regulates TGF-β isoform
expression, reducing adhesion and invasive properties of
endometriotic cells (228).
These effects collectively contribute to lesion regression and
potential improvements in fertility.
NF-κB inhibitors
Disulfiram, a derivative of thiamine, has recently
been shown to exert anti-endometriotic effects. An animal study has
demonstrated that disulfiram significantly reduces serum TNF-α and
IL1B levels and inhibits angiogenesis within ectopic lesions by
downregulating VEGF-A and MMP-2 expression via suppression of NF-κB
nuclear translocation (229).
Moreover, disulfiram alleviates postoperative inflammatory
adhesions associated with EMs, thereby helping to prevent
infertility caused by adhesion formation (230).
Animal experiments have also shown that melatonin
exerts therapeutic effects in AM by blocking the NF-κB signaling
pathway. Melatonin downregulates ROS and pro-inflammatory cytokines
such as TNF-α and IL1B, while upregulating VEGF, thereby improving
the endometrial microenvironment, reducing apoptosis of endometrial
cells and restoring ER. These effects ultimately improve pregnancy
outcomes in AM mouse models (231). Furthermore, melatonin
alleviates uterine oxidative stress and improves uteroplacental
hemodynamics, creating a favorable environment for embryo
implantation and development (232).
TNF-α inhibitors
Etanercept, a TNF inhibitor, effectively blocks
TNF-α/β-mediated NF-κB activation (233). Experimental studies have shown
that administration of etanercept in baboon models of EMs
significantly reduces both the number and volume of endometriotic
lesions (234). In mouse
models, treatment with etanercept results in reduced lesion size
along with downregulation of VEGF, IL-6 and TNF-α levels (235). A retrospective study involving
68 infertile women with EMs reported that a single intramuscular
injection of 50 mg etanercept on day 2 of the menstrual cycle in
the treatment group (n=19) led to improved clinical pregnancy and
embryo implantation rates (236).
Pentoxifylline, a non-selective phosphodiesterase
inhibitor, has been shown to suppress the secretion of
pro-inflammatory cytokines such as TNF-α and IL1B, while also
downregulating key angiogenic factors (237). A randomized controlled trial
confirmed that postoperative treatment with pentoxifylline
significantly reduced VAS pain scores and improved long-term
outcomes in patients with EMs (238). Another randomized controlled
trial demonstrated that in patients with mild or asymptomatic EMs,
the pregnancy rate in the pentoxifylline-treated group was twice as
high as that in the control group (239).
Emerging technologies
Specialized targeted materials
Intralipid, an intravenous lipid emulsion derived
from soybean polyunsaturated fatty acids and egg yolk
phospholipids, has gained significant attention in reproductive
medicine due to its immunomodulatory properties. Basic research
indicates that intralipid exerts broad anti-inflammatory and immune
homeostasis effects by specifically suppressing NK cell activity
(240). In patients with RIF
complicated by NK cell hyperactivity, intralipid treatment has been
associated with live birth rates ranging from 33 to 42%, while
clinical observations in patients with recurrent pregnancy loss
report even higher live birth rates of 75 to 91%, markedly
improving adverse pregnancy outcomes (241). Consequently, lipid-based
intralipid administration could be considered as a novel delivery
route for patients with AM (242).
Recent advances in nanoparticle-based precision
diagnostics and therapeutics have shown breakthrough potential. A
digital droplet enzyme-linked immunosorbent assay system
constructed with SiO2 nanoparticles achieved femtomolar
sensitivity for detecting inflammatory markers such as OPN, IL-6
and IL-10 in menstrual blood, unveiling for the first time the
molecular-level immune-inflammatory signatures in patients with EMs
and providing reliable non-invasive biomarkers (243). Furthermore, 10 nm magnetic iron
oxide nanoparticles enable in situ visualization and
localization of ectopic lesions via MRI, markedly enhancing
diagnostic accuracy (244).
Outer membrane vesicle-modified poly
(lactic-co-glycolic acid) nanoparticles have been developed to
deliver IL-4 plasmids specifically to macrophages, successfully
reversing M2 polarization and markedly inhibiting lesion
progression without compromising ovarian reserve (245). In addition, cerium dioxide
nanoparticles have been shown to reduce oxidative stress and
inhibit angiogenesis, thereby alleviating induced endometrial
lesions in mouse models while preserving both the quantity and
quality of oocytes (246).
Precision therapeutics targeting
non-coding RNAs (ncRNAs)
miRNAs, small ncRNAs that suppress target gene
translation by binding mRNAs, regulate angiogenesis and
inflammatory responses during EMs pathogenesis. Elevated levels of
angiogenic and inflammatory factors such as VEGF-A, IL-6 and CXCL8
in ectopic lesions are closely linked to aberrant activation of
miRNA regulatory networks, including miR-21 and miR-135b (247). Notably, miR-199a targets and
suppresses IKKβ in ESCs, inhibiting NF-κB activation and CXCL8
production, thereby attenuating ESC adhesion, migration and
invasiveness (248), miR-25-3p,
delivered via EMs-derived extracellular vesicles (EMs-EVs), targets
the PTEN/AKT pathway, inducing collagen deposition and inhibiting
decidualization, ultimately decreasing ER (220). The U.S. FDA has approved RNA
interference therapeutics such as patisiran, which employs lipid
nanoparticles to silence pathogenic RNAs in lesions (249). Additionally, exosomes have been
explored as natural carriers for therapeutic ncRNAs (such as
miR-141-3p or let-7b), enabling targeted delivery to ectopic
lesions and enhancing local uptake (250). Patients with AM exhibit
aberrant expression of 21 fertility-related miRNAs in EMs-EVs
secreted by the endometrium, potentially underlying infertility
mechanisms (251).
Small-molecule drugs targeting EMs-associated miRNAs (such as
inhibitors of miR-135 or miR-196) have entered phase II clinical
trials, highlighting ncRNA-based therapies as promising novel
strategies for fertility preservation in patients with EMs.
Artificial intelligence (AI)-driven
novel target identification
The AI-powered drug target discovery platform
PandaOmics (https://pandaomics.cn/access) has recently made
breakthrough progress in EMs research. Through intelligent
algorithmic screening, the platform identified two novel
immune-regulatory therapeutic targets, guanylate binding protein 2
Gene (GBP2) and hemopoietic cell kinase (HCK). Key validation
experiments demonstrated that gene silencing of GBP2 or HCK
significantly inhibited EMs cell proliferation and induced
apoptosis, confirming their potential as therapeutic targets
(252). Therapeutically, outer
membrane vesicle-functionalized poly(lactic-co-glycolic acid)
nanoparticles precisely targeting macrophages modulate the immune
microenvironment, markedly suppressing lesion progression without
impairing ovarian reserve, offering a novel non-hormonal treatment
option for reproductive-aged patients (245).
Traditional Chinese Medicine
(TCM)
Comprehensive TCM therapies, including oral herbal
formulations, acupuncture and topical applications, have been shown
to reduce inflammatory cytokine levels and demonstrate unique
advantages in the diagnosis and treatment of AM. For instance,
Shaoyao-Gancao decoction and its active ingredient paeoniflorin
significantly decrease PGE2 and PGF2α levels, alleviating
AM-associated dysmenorrhea (253).
Resveratrol, a polyphenolic compound, has both
anti-inflammatory and antioxidant properties. Animal studies have
demonstrated that it can effectively inhibit myometrial
infiltration and alleviate pain-related characteristics without
disrupting hormonal homeostasis (254). Clinical trials have
demonstrated that a daily dose of 30 mg resveratrol combined with
oral contraceptives significantly reduces the expression of COX-2
and aromatase expression, potentially via suppression of
prostaglandin biosynthesis pathways, thereby diminishing the
invasive potential of ectopic endometrial tissue (255).
Oleuropein, a novel natural product, selectively
inhibits ERβ activity without affecting ERα. As ERβ can activate
the NLRP3 inflammasome, potentiate IL-1β-mediated proliferation and
modulate signaling cascades such as TNFα/NF-κB, ROS and
IL-6/JAK/STAT3, selective inhibition by oleuropein may impede
disease progression (256).
Experiments have further indicated that oleuropein restores
defective endometrial decidualization and attenuates excessive
inflammatory responses, ultimately improving pregnancy rates in EMs
models (257).
Quercetin, a natural flavonoid polyphenol, exhibits
antioxidant, anti-inflammatory, antiproliferative and antitumor
activities; it dose-dependently suppresses the migration and
invasion of ectopic ESCs by downregulating invasion-related
proteins including Fascin, Ezrin and MMP-2/MMP-9 (110), aligning with the natural
bioactive compound-based interventions featured in the multi-modal
therapeutic framework of Fig.
6.
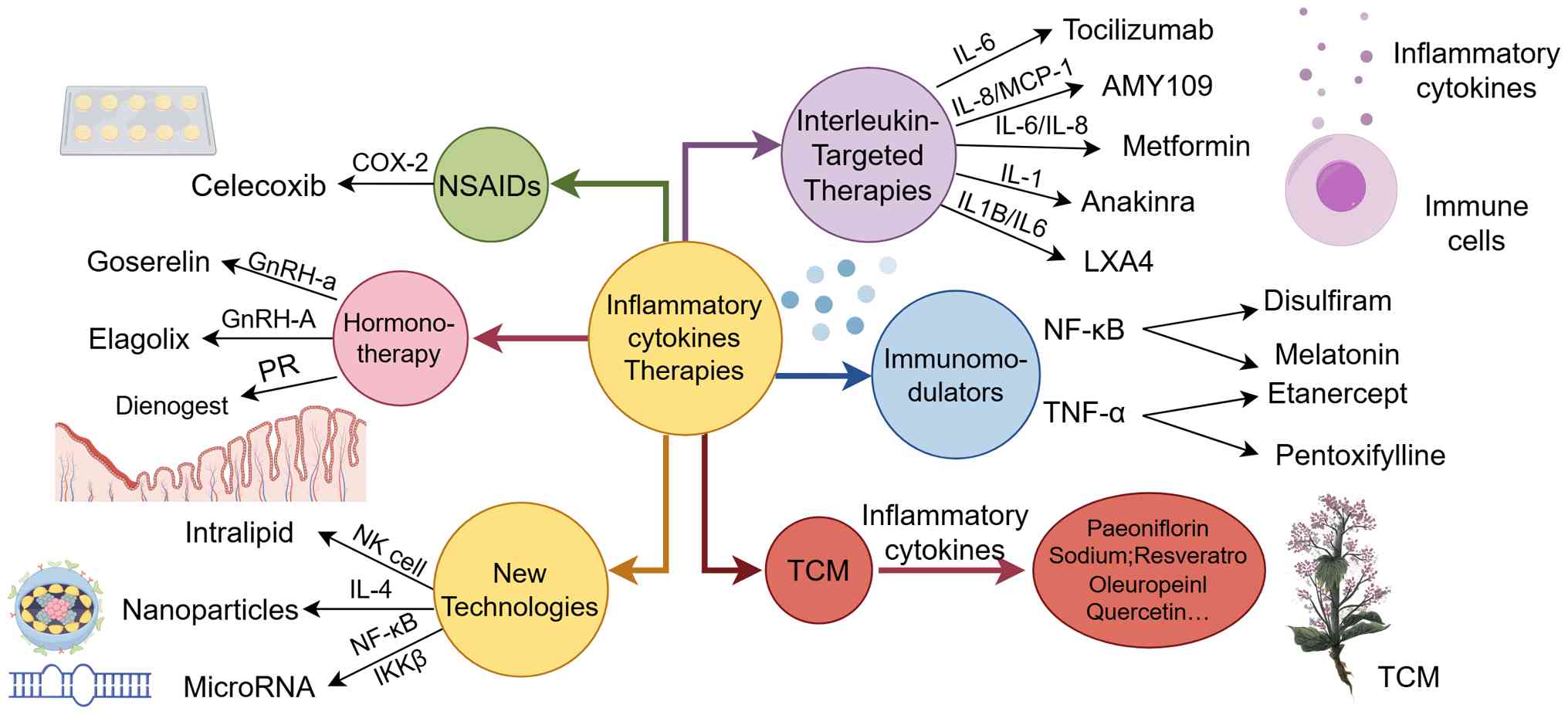 | Figure 6Main therapeutic strategies for
adenomyosis-related female infertility. Taking inflammatory
cytokines as the core regulatory target, this figure covers
multiple therapeutic modalities including NSAIDs, hormonal therapy,
interleukin-targeted therapies, immunomodulators, TCM and emerging
therapeutic technologies and displays the corresponding specific
drugs and their action targets for each modality. These therapeutic
strategies can modulate inflammatory cytokines, thereby
ameliorating adenomyosis-associated pathological processes, further
enhancing endometrial receptivity and reversing decidualization
dysfunction, which contributes to improving the infertility
outcomes in women with adenomyosis. TCM, Traditional Chinese
Medicine; GnRH-a, gonadotropin-releasing hormone agonist; GnRH-A,
gonadotropin-releasing hormone antagonist; IKKβ, IκB kinase β; IL,
interleukin; MCP-1, monocyte chemoattractant protein-1; NSAIDs,
non-steroidal anti-inflammatory drugs; PR, progesterone
receptor. |
Furthermore, sodium tanshinone IIA sulfonate
inhibits platelet aggregation and suppresses the TGF-β1/Smad3 and
NF-κB signaling pathways, thereby attenuating fibrosis (258). Oral emodin dose-dependently
inhibits IL1B-induced NF-κB expression and Wnt/β-catenin signaling
activation to treat AM (109).
Berberine significantly reduces LPS-induced endometrial invasion
and downregulates IL-6 and CXCL8 expression, promoting apoptosis of
proliferative ESCs (259).
Collectively, a growing body of evidence supports these findings.
Guided by TCM principles of syndrome differentiation and
individualized therapy, patients can achieve symptomatic relief and
improved fertility, with robust clinical efficacy. These
TCM-derived bioactive components are included in Table SII, which comprehensively
summarizes the full spectrum of medications for AM treatment. These
agents target inflammatory cytokines while preserving female
reproductive capacity.
Discussion
AM is commonly regarded as a chronic inflammatory
immune disease. We consider that chronic inflammation acts as the
upstream inducer of immunological abnormalities in AM, with the
subsequent imbalance between proinflammatory and anti-inflammatory
signals, serving as a downstream pathological consequence;
together, this cascade constitutes the critical core factors
affecting fertility in affected women. Notably, this pathogenic
process originates from persistent TIAR at the EMJ: Repeated
menstrual microtrauma at the EMJ triggers sustained release of
proinflammatory cytokines, which in turn initiates chronic
inflammation. This chronic inflammatory milieu further induces EMT
in eutopic endometrial cells, enhancing their invasiveness to
infiltrate the myometrium, while simultaneously disrupting local
immune homeostasis to cause immune dysregulation, establishing a
reciprocal amplification loop between inflammation and immune
dysfunction that underpins AM pathogenesis.
Inflammatory cytokines, key immunological markers,
abnormally accumulate in endometrial lesions, serum and peritoneal
fluid and play a central role in AM-related infertility. Driven by
chronic inflammation and synergized by immune dysregulation, their
dysregulated expression exerts multi-stage reproductive impairment.
On the one hand, they sustain an abundant blood supply to ectopic
lesions by promoting neovascularization and the inflammatory
changes at the EMJ (exacerbated by EMT-mediated tissue remodeling)
lead to reduced ER and impaired decidualization. On the other hand,
immune dysregulation disturbs the maternal-fetal immune tolerance
required for embryo implantation.
Together, these effects ultimately hinder embryo
implantation, causing infertility, IVF failure and recurrent
pregnancy loss. In addition, disturbances in inflammatory cytokine
levels are associated with adverse immune environments during
childbirth and postpartum, contributing to risks such as preterm
birth, postpartum hemorrhage and premature rupture of membranes.
Other symptoms of AM, including menorrhagia and dysmenorrhea, are
also rooted in this chronic inflammation-immune dysregulation
cascade, as proinflammatory factors sensitize pain pathways and
disrupt endometrial hemostasis.
At present, the clinical diagnosis of AM relies
primarily on non-invasive methods, such as transvaginal ultrasound
or MRI, with CA-125 as the only routinely measured biomarker.
However, CA-125 has limited diagnostic potential, with insufficient
sensitivity for detecting early and mild AM, and lacks the capacity
for timely diagnosis and prevention of chronic pain and infertility
associated with AM (260).
Therefore, the development of novel serum biomarkers is essential
for patients with AM who have long-term fertility plans. Research
has demonstrated that inflammatory cytokines possess diagnostic
sensitivity and specificity (261), with IL-6, CXCL8 and IL1B
showing the most notable potential for clinical translation. Serum
IL-6 is a reliable marker for the early diagnosis of infertility
related to EMs. CXCL8 may serve as a specific predictor for
evaluating embryo and oocyte quality in patients with AM undergoing
IVF and can independently assess pretransfer embryonic
developmental potential (238).
Elevated IL1B levels may also act as a highly sensitive and
specific potential indicator for early AM diagnosis (262). Additionally, the
neutrophil-to-lymphocyte ratio in peripheral blood has been
recognized as a valuable marker for infertility in women with EMs,
while increased levels of anti-inflammatory cytokines, such as
IL10, IL4 and IL-1RA, have been linked to the disease prognosis
(263). Endometrial
autoantibodies also offer a novel, non-invasive diagnostic approach
(264). However, to enable
effective diagnosis or treatment of AM based on inflammatory
cytokines, high-sensitivity cytokine arrays and multivariable
statistical analyses (accounting for inter-individual phenotypic
variability) are necessary, highlighting the need for optimized
diagnostic tools. When multiple biomarkers are evaluated,
multivariable statistical analyses are needed, with careful
consideration of the phenotypic variability of EMs across different
individuals. Thus, further advancements in diagnostic tools and
techniques are crucial.
The present review also discussed recent advances
in pharmacological therapies targeting inflammatory cytokines such
as IL1B, IL-6, CXCL8, IL-10, NF-κB and COX-2. As treatment
strategies for AM often depend on the specific patient's symptoms,
a number of medications effective for dysmenorrhea and menorrhagia
may compromise fertility, with some significantly compromising
reproductive function (242).
Therefore, prior to treatment, individualized assessment of the
inflammatory and immune status of the patient is essential for
determining the most appropriate therapeutic approach, which may
support the development of more effective, safer management options
for AM-associated infertility.
Future research should focus on three specific
priorities to advance the field: First, large-scale prospective
studies should be conducted to validate the diagnostic cut-off
values of IL-6 and IL1B for early AM and AM-related infertility and
establish combined biomarker panels to improve diagnostic accuracy;
second, clinical trials should be launched to evaluate the efficacy
of IL-6-targeted therapies (such as tocilizumab) for improving IVF
implantation rates in patients with AM and explore the synergistic
effects of combining NF-κB inhibitors with IL1B antagonists; third,
the crosstalk mechanism between chronic inflammation-induced EMT
and immune dysregulation at the EMJ should be clarified, to
identify new intervention nodes that simultaneously target lesion
invasion and immune tolerance for fertility preservation.
In conclusion, chronic inflammation-driven immune
dysregulation is the core pathogenic link underlying AM-related
reproductive impairment and IL-6, CXCL8 and IL-1β represent the
most promising targets for diagnostic optimization and therapeutic
innovation. Further research into these directions will provide
critical support for improving reproductive outcomes in patients
with AM.
Conclusion
The present review summarizes the role of
inflammatory cytokines in the pathogenesis of AM-related
infertility, particularly highlighting that the pathological
progression and fertility impairment of AM are largely dependent on
the regulation of inflammatory cytokines. These cytokines directly
or indirectly affect the fertility of patients with AM by
modulating multiple signaling pathways to regulate critical
physiological processes, including cell proliferation and invasion,
EMT, TIAR, angiogenesis and ER. Given their irreplaceable and core
role in disease progression, these inflammatory cytokines hold
great promise as important biomarkers for the early diagnosis and
targeted treatment of AM-related infertility. However, current
research on their clinical application value is still in the
exploratory stage. Further studies are needed to conduct targeted
investigations to further explore their potential in optimizing
diagnosis methods and therapeutic strategies.
Supplementary Data
Availability of data and materials
Not applicable.
Authors' contributions
BY wrote the manuscript and constructed the tables
and figures. YS, FL, JJ and GC provided critical insights and
contributed to the manuscript writing. ZW and MN refined the
research ideas, reviewed the initial draft and revised the final
manuscript. ZL and WS reviewed and provided critical feedback for
the manuscript. All the authors read and approved the final version
of the manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
The present review was supported by National Natural Science
Foundation of China (grant nos. 82575124, 82474559 and 82505636),
Natural Science Foundation of Shandong Province, China (grant nos.
ZR2025MS1375 and ZR2023LZY024) and Traditional Chinese Medicine
Science and Technology Development Plan Project of Shandong
Province (grant nos. M-2023259 and M20241836).
References
|
1
|
Orazov MR, Radzinsky VE, Nosenko EN,
Khamoshina MB, Dukhin AO and Lebedeva MG: Immune-inflammatory
predictors of the pelvic pain syndrome associated with adenomyosis.
Gynecol Endocrinol. 33(Suppl 1): S44–S46. 2017. View Article : Google Scholar
|
|
2
|
Upson K and Missmer SA: Epidemiology of
adenomyosis. Semin Reprod Med. 38:89–107. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vannuccini S and Petraglia F: Recent
advances in understanding and managing adenomyosis. F1000Res.
8:F1000 Faculty Rev-2832019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kobayashi H: Molecular targets for
nonhormonal treatment based on a multistep process of adenomyosis
development. Reprod Sci. 30:743–760. 2023. View Article : Google Scholar
|
|
5
|
Puente JM, Fabris A, Patel J, Patel A,
Cerrillo M, Requena A and Garcia-Velasco JA: Adenomyosis in
infertile women: Prevalence and the role of 3D ultrasound as a
marker of severity of the disease. Reprod Biol Endocrinol.
14:602016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vannuccini S, Tosti C, Carmona F, Huang
SJ, Chapron C, Guo SW and Petraglia F: Pathogenesis of adenomyosis:
An update on molecular mechanisms. Reprod Biomed Online.
35:592–601. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nirgianakis K, Kalaitzopoulos DR, Schwartz
ASK, Spaanderman M, Kramer BW, Mueller MD and Mueller M: Fertility,
pregnancy and neonatal outcomes of patients with adenomyosis: A
systematic review and meta-analysis. Reprod Biomed Online.
42:185–206. 2021. View Article : Google Scholar
|
|
8
|
Xing Z, Af khami S, Bavananthasivam J,
Fritz DK, D'Agostino MR, Vaseghi-Shanjani M, Yao Y and Jeyanathan
M: Innate immune memory of tissue-resident macrophages and trained
innate immunity: Re-vamping vaccine concept and strategies. J
Leukoc Biol. 108:825–834. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Guo SW: The pathogenesis of adenomyosis
vis-à-vis endometriosis. J Clin Med. 9:4852020. View Article : Google Scholar
|
|
10
|
Szubert M, Koziróg E, Olszak O,
Krygier-Kurz K, Kazmierczak J and Wilczynski J: Adenomyosis and
infertility-review of medical and surgical approaches. Int J
Environ Res Public Health. 18:12352021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vigano P, Corti L and Berlanda N: Beyond
infertility: Obstetrical and postpartum complications associated
with endometriosis and adenomyosis. Fertil Steril. 104:802–812.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kissler S, Hamscho N, Zangos S, Wiegratz
I, Schlichter S, Menzel C, Doebert N, Gruenwald F, Vogl TJ, Gaetje
R, et al: Uterotubal transport disorder in adenomyosis and
endometriosis-a cause for infertility. BJOG. 113:902–908. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li B, Qi J, Cao Y, Long Y, Wei Z, Wang WS,
Hu S, Wang Y, Zhu Q, Hu X, et al: From invaginating site to deep
lesion: Spatial transcriptomics unravels ectopic endometrial
penetration features in adenomyosis. Adv Sci (Weinh).
12:e24117522025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Martínez-Conejero JA, Morgan M, Montesinos
M, Fortuño S, Meseguer M, Simón C, Horcajadas JA and Pellicer A:
Adenomyosis does not affect implantation, but is associated with
miscarriage in patients undergoing oocyte donation. Fertil Steril.
96:943–950. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Vercellini P, Consonni D, Dridi D, Bracco
B, Frattaruolo MP and Somigliana E: Uterine adenomyosis and in
vitro fertilization outcome: A systematic review and meta-analysis.
Hum Reprod. 29:964–977. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mavrelos D, Holland TK, O'Donovan O,
Khalil M, Ploumpidis G, Jurkovic D and Khalaf Y: The impact of
adenomyosis on the outcome of IVF-embryo transfer. Reprod Biomed
Online. 35:549–554. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Moffett A and Colucci F: Uterine NK cells:
Active regulators at the maternal-fetal interface. J Clin Invest.
124:1872–1879. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cozzolino M, Cosentino M, Loiudice L,
Martire FG, Galliano D, Pellicer A and Exacoustos C: Impact of
adenomyosis on in vitro fertilization outcomes in women undergoing
donor oocyte transfers: A prospective observational study. Fertil
Steril. 121:480–488. 2024. View Article : Google Scholar
|
|
19
|
Carrarelli P, Yen CF, Funghi L, Arcuri F,
Tosti C, Bifulco G, Luddi A, Lee CL and Petraglia F: Expression of
inflammatory and neurogenic mediators in adenomyosis. Reprod Sci.
24:369–375. 2017. View Article : Google Scholar
|
|
20
|
Zhihong N, Yun F, Pinggui Z, Sulian Z and
Zhang A: Cytokine profiling in the eutopic endometrium of
adenomyosis during the implantation window after ovarian
stimulation. Reprod Sci. 23:124–133. 2016. View Article : Google Scholar
|
|
21
|
Gan L, Li Y, Chen Y, Huang M, Cao J, Cao
M, Wang Z, Wan G and Gui T: Transcriptome analysis of eutopic
endometrial stromal cells in women with adenomyosis by
RNA-sequencing. Bioengineered. 13:12637–12649. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Baran P, Hansen S, Waetzig GH, Akbarzadeh
M, Lamertz L, Huber HJ, Ahmadian MR, Moll JM and Scheller J: The
balance of interleukin (IL)-6, IL-6•soluble IL-6 receptor (sIL-6R),
and IL-6•sIL-6R•sgp130 complexes allows simultaneous classic and
trans-signaling. J Biol Chem. 293:6762–6775. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Candido S, Tomasello BMR, Lavoro A,
Falzone L, Gattuso G and Libra M: Novel insights into epigenetic
regulation of IL6 pathway: In silico perspective on inflammation
and cancer relationship. Int J Mol Sci. 22:101722021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Neurath MF and Finotto S: IL-6 signaling
in autoimmunity, chronic inflammation and inflammation-associated
cancer. Cytokine Growth Factor Rev. 22:83–89. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jiang X and Chen X: Endometrial
cell-derived exosomes facilitate the development of adenomyosis via
the IL-6/JAK2/STAT3 pathway. Exp Ther Med. 26:5262023. View Article : Google Scholar
|
|
26
|
Zhang K, Zhou Z, Wang C, Yu M, Zhang Y,
Shi Y, Wang X, Liu Y, Xu L, Shi W and Liu Z: Mechanism study of
cinnamomi ramulus and Paris polyphylla sm. drug pair in the
treatment of adenomyosis by network pharmacology and experimental
validation. Evid Based Complement Alternat Med.
2022:26244342022.PubMed/NCBI
|
|
27
|
Samson SC, Khan AM and Mendoza MC: ERK
signaling for cell migration and invasion. Front Mol Biosci.
9:9984752022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Smith SK, Charnock-Jones DS and Sharkey
AM: The role of leukemia inhibitory factor and interleukin-6 in
human reproduction. Hum Reprod. 13(Suppl 3): S237–S246. 1998.
View Article : Google Scholar
|
|
29
|
Xu Y, Wu F, Qin C and Lin Y: Paradoxical
role of phosphorylated STAT3 in normal fertility and the
pathogenesis of adenomyosis and endometriosis†. Biol Reprod.
110:5–13. 2024. View Article : Google Scholar
|
|
30
|
Incognito GG, Di Guardo F, Gulino FA,
Genovese F, Benvenuto D, Lello C and Palumbo M: Interleukin-6 as a
useful predictor of endometriosis-associated infertility: A
systematic review. Int J Fertil Steril. 17:226–230. 2023.PubMed/NCBI
|
|
31
|
Liu XN and Cheng ZP: Expression of
high-mobility group box-1 in eutopic/ectopic endometrium and
correlations with inflammation-related factors in adenomyosis.
Gynecol Endocrinol. 39:22692652023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ulukus EC, Ulukus M, Seval Y, Zheng W and
Arici A: Expression of interleukin-8 and monocyte chemotactic
protein-1 in adenomyosis. Hum Reprod. 20:2958–2963. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li MQ, Luo XZ, Meng YH, Mei J, Zhu XY, Jin
LP and Li DJ: CXCL8 enhances proliferation and growth and reduces
apoptosis in endometrial stromal cells in an autocrine manner via a
CXCR1-triggered PTEN/AKT signal pathway. Hum Reprod. 27:2107–2116.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zheng D, Duan H, Wang S, Xu Q, Gan L, Li J
and Dong Q: FAK regulates epithelial-mesenchymal transition in
adenomyosis. Mol Med Rep. 18:5461–5472. 2018.PubMed/NCBI
|
|
35
|
Kataranovski M, Radojcić L, Prokić V and
Vojvodić D: Presence of interleukin-8 and the IL-1 receptor
antagonist in the cervical mucus of fertile and infertile women.
Vojnosanit Pregl. 61:359–364. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
An LF, Zhang XH, Sun XT, Zhao LH, Li S and
Wang WH: Unexplained infertility patients have increased serum
IL-2, IL-4, IL-6, IL-8, IL-21, TNFα, IFNγ and increased Tfh/CD4 T
cell ratio: Increased Tfh and IL-21 strongly correlate with
presence of autoantibodies. Immunol Invest. 44:164–173. 2015.
View Article : Google Scholar
|
|
37
|
Singh AK, Dutta M, Chattopadhyay R,
Chakravarty B and Chaudhury K: Intrafollicular interleukin-8,
interleukin-12, and adrenomedullin are the promising prognostic
markers of oocyte and embryo quality in women with endometriosis. J
Assist Reprod Genet. 33:1363–1372. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhu D: Expression and clinical
significance of IL-8 and CXCR1 in adenomyosis (Dissertation).
Central South University; 2010
|
|
39
|
Boraschi D, Italiani P, Weil S and Martin
MU: The family of the interleukin-1 receptors. Immunol Rev.
281:197–232. 2018. View Article : Google Scholar
|
|
40
|
De Nardo D: Toll-like receptors:
Activation, signalling and transcriptional modulation. Cytokine.
74:181–189. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Surai PF, Kochish II and Kidd MT: Redox
homeostasis in poultry: Regulatory roles of NF-κB. Antioxidants
(Basel). 10:1862021. View Article : Google Scholar
|
|
42
|
Dinarello CA: Immunological and
inflammatory functions of the interleukin-1 family. Annu Rev
Immunol. 27:519–550. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Equils O, Kellogg C, McGregor J, Gravett
M, Neal-Perry G and Gabay C: The role of the IL-1 system in
pregnancy and the use of IL-1 system markers to identify women at
risk for pregnancy complications†. Biol Reprod. 103:684–694. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wang H, Shi G, Li M, Fan H, Ma H and Sheng
L: Correlation of IL-1 and HB-EGF with endometrial receptivity. Exp
Ther Med. 16:5130–5136. 2018.PubMed/NCBI
|
|
45
|
Faber BM, Chegini N, Mahony MC and
Coddington CC III: Macrophage secretory products and sperm zona
pellucida binding. Obstet Gynecol. 98:668–673. 2001.PubMed/NCBI
|
|
46
|
Saraiva M and O'Garra A: The regulation of
IL-10 production by immune cells. Nat Rev Immunol. 10:170–181.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
O'Garra A, Barrat FJ, Castro AG, Vicari A
and Hawrylowicz C: Strategies for use of IL-10 or its antagonists
in human disease. Immunol Rev. 223:114–131. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Qin X, Zhang H, Wang F, Xue J and Wen Z:
Expression and possible role of interleukin-10 receptors in
patients with adenomyosis. Eur J Obstet Gynecol Reprod Biol.
161:194–198. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Bourdon M, Santulli P, Jeljeli M,
Vannuccini S, Marcellin L, Doridot L, Petraglia F, Batteux F and
Chapron C: Immunological changes associated with adenomyosis: A
systematic review. Hum Reprod Update. 27:108–129. 2021. View Article : Google Scholar
|
|
50
|
Gui T, Chen C, Zhang Z, Tang W, Qian R, Ma
X, Cao P and Wan G: The disturbance of TH17-Treg cell balance in
adenomyosis. Fertil Steril. 101:506–514. 2014. View Article : Google Scholar
|
|
51
|
Antero MF, Ayhan A, Segars J and Shih IM:
Pathology and pathogenesis of adenomyosis. Semin Reprod Med.
38:108–118. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Thellin O, Coumans B, Zorzi W, Igout A and
Heinen E: Tolerance to the foeto-placental 'graft': Ten ways to
support a child for nine months. Curr Opin Immunol. 12:731–737.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wang F, Li H, Yang Z, Du X, Cui M and Wen
Z: Expression of interleukin-10 in patients with adenomyosis.
Fertil Steril. 91:1681–1685. 2009. View Article : Google Scholar
|
|
54
|
Wang J, Huang C, Jiang R, Du Y, Zhou J,
Jiang Y, Yan Q, Xing J, Hou X, Zhou J, et al: Decreased endometrial
IL-10 impairs endometrial receptivity by downregulating HOXA10
expression in women with adenomyosis. Biomed Res Int.
2018:25497892018. View Article : Google Scholar
|
|
55
|
Sowmya S, Sri Manjari K, Ramaiah A,
Sunitha T, Nallari P, Jyothy A and Venkateshwari A: Interleukin 10
gene promoter polymorphisms in women with early-onset
pre-eclampsia. Clin Exp Immunol. 178:334–341. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Singer II, Kawka DW, Schloemann S, Tessner
T, Riehl T and Stenson WF: Cyclooxygenase 2 is induced in colonic
epithelial cells in inflammatory bowel disease. Gastroenterology.
115:297–306. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Li C, Chen R, Jiang C, Chen L and Cheng Z:
Correlation of LOX-5 and COX-2 expression with inflammatory
pathology and clinical features of adenomyosis. Mol Med Rep.
19:727–733. 2019.
|
|
58
|
Zhang J, Shi L, Duan J, Li M and Li C:
Proteomic detection of COX-2 pathway-related factors in patients
with adenomyosis. PeerJ. 12:e167842024. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Chen YJ, Li HY, Chang YL, Yuan CC, Tai LK,
Lu KH, Chang CM and Chiou SH: Suppression of migratory/invasive
ability and induction of apoptosis in adenomyosis-derived
mesenchymal stem cells by cyclooxygenase-2 inhibitors. Fertil
Steril. 94:1972–1979. e1–e4. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang P and Wang G: Progesterone
resistance in endometriosis: Current evidence and putative
mechanisms. Int J Mol Sci. 24:69922023. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yang XJ, Yang J, Liu Z, Yang G and Shen
ZJ: Telocytes damage in endometriosis-affected rat oviduct and
potential impact on fertility. J Cell Mol Med. 19:452–462. 2015.
View Article : Google Scholar :
|
|
62
|
Sakamoto Y, Harada T, Horie S, Iba Y,
Taniguchi F, Yoshida S, Iwabe T and Terakawa N: Tumor necrosis
factor-alpha-induced interleukin-8 (IL-8) expression in
endometriotic stromal cells, probably through nuclear factor-kappa
B activation: Gonadotropin-releasing hormone agonist treatment
reduced IL-8 expression. J Clin Endocrinol Metab. 88:730–735. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Wu M and Zhang Y: MiR-182 inhibits
proliferation, migration, invasion and inflammation of endometrial
stromal cells through deactivation of NF-κB signaling pathway in
endometriosis. Mol Cell Biochem. 476:1575–1588. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Xu L, Yu Y, Sang R, Ge B, Wang M, Zhou H
and Zhang X: Inonotus obliquus polysaccharide protects against
adverse pregnancy caused by Toxoplasma gondii infection through
regulating Th17/Treg balance via TLR4/NF-κB pathway. Int J Biol
Macromol. 146:832–840. 2020. View Article : Google Scholar
|
|
65
|
Chen CC, Montalbano AP, Hussain I, Lee WR
and Mendelson CR: The transcriptional repressor GATAD2B mediates
progesterone receptor suppression of myometrial contractile gene
expression. J Biol Chem. 292:12560–12576. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Tremellen KP and Russell P: The
distribution of immune cells and macrophages in the endometrium of
women with recurrent reproductive failure. II: Adenomyosis and
macrophages. J Reprod Immunol. 93:58–63. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
An M, Li D, Yuan M, Li Q, Zhang L and Wang
G: Interaction of macrophages and endometrial cells induces
epithelial-mesenchymal transition-like processes in adenomyosis.
Biol Reprod. 96:46–57. 2017.PubMed/NCBI
|
|
68
|
Itoh F, Komohara Y, Takaishi K, Honda R,
Tashiro H, Kyo S, Katabuchi H and Takeya M: Possible involvement of
signal transducer and activator of transcription-3 in cell-cell
interactions of peritoneal macrophages and endometrial stromal
cells in human endometriosis. Fertil Steril. 99:1705–1713. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Kim H, Wang SY, Kwak G, Yang Y, Kwon IC
and Kim SH: Exosome-guided phenotypic switch of M1 to M2
macrophages for cutaneous wound healing. Adv Sci (Weinh).
6:19005132019. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Gou Y, Li X, Li P, Zhang H, Xu T, Wang H,
Wang B, Ma X, Jiang X and Zhang Z: Estrogen receptor β upregulates
CCL2 via NF-κB signaling in endometriotic stromal cells and
recruits macrophages to promote the pathogenesis of endometriosis.
Hum Reprod. 34:646–658. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Huang Y, Zhu L, Li H, Ye J, Lin N, Chen M,
Pan D and Chen Z: Endometriosis derived exosomal miR-301a-3p
mediates macrophage polarization via regulating PTEN-PI3K axis.
Biomed Pharmacother. 147:1126802022. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Sun SG, Guo JJ, Qu XY, Tang XY, Lin YY,
Hua KQ and Qiu JJ: The extracellular vesicular pseudogene LGMNP1
induces M2-like macrophage polarization by upregulating LGMN and
serves as a novel promising predictive biomarker for ovarian
endometriosis recurrence. Hum Reprod. 37:447–465. 2022. View Article : Google Scholar
|
|
73
|
Zhang D, Yu Y, Duan T and Zhou Q: The role
of macrophages in reproductive-related diseases. Heliyon.
8:e116862022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Le Bouteiller P and Tabiasco J: Killers
become builders during pregnancy. Nat Med. 12:991–992. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Ścieżyńska A, Komorowski M, Soszyńska M
and Malejczyk J: NK cells as potential targets for immunotherapy in
endometriosis. J Clin Med. 8:14682019. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Huhn O, Zhao X, Esposito L, Moffett A,
Colucci F and Sharkey AM: How do uterine natural killer and innate
lymphoid cells contribute to successful pregnancy? Front Immunol.
12:6076692021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Szekeres-Bartho J: Regulation of NK cell
cytotoxicity during pregnancy. Reprod Biomed Online. 16:211–217.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Díaz-Hernández I, Alecsandru D,
García-Velasco JA and Domínguez F: Uterine natural killer cells:
From foe to friend in reproduction. Hum Reprod Update. 27:720–746.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Shi J, Xu Q, Yu S and Zhang T:
Perturbations of the endometrial immune microenvironment in
endometriosis and adenomyosis: Their impact on reproduction and
pregnancy. Semin Immunopathol. 47:162025. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Mei J, Zhou WJ, Zhu XY, Lu H, Wu K, Yang
HL, Fu Q, Wei CY, Chang KK, Jin LP, et al: Suppression of autophagy
and HCK signaling promotes PTGS2high FCGR3−
NK cell differentiation triggered by ectopic endometrial stromal
cells. Autophagy. 14:1376–1397. 2018. View Article : Google Scholar
|
|
81
|
Wang X, Zhang P, Tang Y, Chen Y, Zhou E
and Gao K: Mast cells: A double-edged sword in inflammation and
fibrosis. Front Cell Dev Biol. 12:14664912024. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Niu W, Zhang Y, Liu H, Liang N, Xu L, Li
Y, Yao W, Shi W and Liu Z: Single-cell profiling uncovers the roles
of endometrial fibrosis and microenvironmental changes in
adenomyosis. J Inflamm Res. 16:1949–1965. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Hoe E, Anderson J, Nathanielsz J, Toh ZQ,
Marimla R, Balloch A and Licciardi PV: The contrasting roles of
Th17 immunity in human health and disease. Microbiol Immunol.
61:49–56. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Zhang H, Li C, Li W, Xin W and Qin T:
Research advances in adenomyosis-related signaling pathways and
promising targets. Biomolecules. 14:14022024. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Li J, Yanyan M, Mu L, Chen X and Zheng W:
The expression of Bcl-2 in adenomyosis and its effect on
proliferation, migration, and apoptosis of endometrial stromal
cells. Pathol Res Pract. 215:1524772019. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Mao C, Liu X and Guo S: Decreased
glycolysis at menstruation is associated with increased menstrual
blood loss. Reprod Sci. 30:928–951. 2023. View Article : Google Scholar
|
|
87
|
Anderson NM and Simon MC: The tumor
microenvironment. Curr Biol. 30:R921–R925. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Leyendecker G, Kunz G, Wildt L, Beil D and
Deininger H: Uterine hyperperistalsis and dysperistalsis as
dysfunctions of the mechanism of rapid sperm transport in patients
with endometriosis and infertility. Hum Reprod. 11:1542–1551. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Zippl AL, Kyvelidou C, Frank M, Gapp E,
Reiser E, Braun AS, Feil K, Schuchter S, Rockenschaub P, Toth B and
Seeber B: The impact of GnRH agonists on endometrial immune cells
in patients with adenomyosis: A prospective cohort study. BMC Med.
23:3382025. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Ota H, Igarashi S, Hatazawa J and Tanaka
T: Is adenomyosis an immune disease? Hum Reprod Update. 4:360–367.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Chen T, Xu Y, Xu X, Wang J, Qiu Z, Yu Y,
Jiang X, Shao W, Bai D, Wang M, et al: Comprehensive
transcriptional atlas of human adenomyosis deciphered by the
integration of single-cell RNA-sequencing and spatial
transcriptomics. Protein Cell. 15:530–546. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Bulletti C, de Ziegler D, Polli V,
Diotallevi L, Del Ferro E and Flamigni C: Uterine contractility
during the menstrual cycle. Hum Reprod. 15(Suppl 1): S81–S89. 2000.
View Article : Google Scholar
|
|
93
|
Leyendecker G, Bilgicyildirim A, Inacker
M, Stalf T, Huppert P, Mall G, Böttcher B and Wildt L: Adenomyosis
and endometriosis. Re-visiting their association and further
insights into the mechanisms of auto-traumatisation. An MRI study.
Arch Gynecol Obstet. 291:917–932. 2015. View Article : Google Scholar :
|
|
94
|
Dueholm M: Uterine adenomyosis and
infertility, review of reproductive outcome after in vitro
fertilization and surgery. Acta Obstet Gynecol Scand. 96:715–726.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Maubon A, Faury A, Kapella M, Pouquet M
and Piver P: Uterine junctional zone at magnetic resonance imaging:
A predictor of in vitro fertilization implantation failure. J
Obstet Gynaecol Res. 36:611–618. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Tellum T, Matic GV, Dormagen JB, Nygaard
S, Viktil E, Qvigstad E and Lieng M: Diagnosing adenomyosis with
MRI: A prospective study revisiting the junctional zone thickness
cutoff of 12 mm as a diagnostic marker. Eur Radiol. 29:6971–6981.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Rees CO, de Boer A, Huang Y, Wessels B,
Blank C, Kuijsters N, Huppelschoten A, Zizolfi B, Foreste V, Di
Spiezio Sardo A, et al: Uterine contractile activity in healthy
women throughout the menstrual cycle measured using a novel
quantitative two-dimensional transvaginal ultrasound speckle
tracking method. Reprod Biomed Online. 46:115–122. 2023. View Article : Google Scholar
|
|
98
|
Huang Y, Rees C, Sammali F, Blank C,
Schoot D and Mischi M: Characterization of uterine peristaltic
waves by ultrasound strain analysis. IEEE Trans Ultrason
Ferroelectr Freq Control. 69:2050–2060. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Leimert KB, Verstraeten BSE, Messer A,
Nemati R, Blackadar K, Fang X, Robertson SA, Chemtob S and Olson
DM: Cooperative effects of sequential PGF2α and IL-1β on IL-6 and
COX-2 expression in human myometrial cells†. Biol Reprod.
100:1370–1385. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Zhu L, Xiao L, Che HS, Li YP and Liao JT:
Uterine peristalsis exerts control over fluid migration after mock
embryo transfer. Hum Reprod. 29:279–285. 2014. View Article : Google Scholar
|
|
101
|
Lai ZZ, Yang HL, Ha SY, Chang KK, Mei J,
Zhou WJ, Qiu XM, Wang XQ, Zhu R, Li DJ and Li MQ: Cyclooxygenase-2
in endometriosis. Int J Biol Sci. 15:2783–2797. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Bergeron C, Amant F and Ferenczy A:
Pathology and physiopathology of adenomyosis. Best Pract Res Clin
Obstet Gynaecol. 20:511–521. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Kusama K, Fukushima Y, Yoshida K,
Sakakibara H, Tsubata N, Yoshie M, Kojima J, Nishi H and Tamura K:
Endometrial epithelial-mesenchymal transition (EMT) by
menstruation-related inflammatory factors during hypoxia. Mol Hum
Reprod. 27:gaab0362021. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Acloque H, Adams MS, Fishwick K,
Bronner-Fraser M and Nieto MA: Epithelial-mesenchymal transitions:
The importance of changing cell state in development and disease. J
Clin Invest. 119:1438–1449. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Wu MH, Wang CA, Lin CC, Chen LC, Chang WC
and Tsai SJ: Distinct regulation of cyclooxygenase-2 by
interleukin-1beta in normal and endometriotic stromal cells. J Clin
Endocrinol Metab. 90:286–295. 2005. View Article : Google Scholar
|
|
106
|
Yang G, Im HJ and Wang JHC: Repetitive
mechanical stretching modulates IL-1beta induced COX-2, MMP-1
expression, and PGE2 production in human patellar tendon
fibroblasts. Gene. 363:166–172. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Leyendecker G, Wildt L and Mall G: The
pathophysiology of endometriosis and adenomyosis: Tissue injury and
repair. Arch Gynecol Obstet. 280:529–538. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Bilyk O, Coatham M, Jewer M and Postovit
LM: Epithelial-to-mesenchymal transition in the female reproductive
tract: From normal functioning to disease pathology. Front Oncol.
7:1452017. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Feng T, Wei S, Wang Y, Fu X, Shi L, Qu L
and Fan X: Rhein ameliorates adenomyosis by inhibiting NF-κB and
β-Catenin signaling pathway. Biomed Pharmacother. 94:231–237. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Xu W, Song Y, Li K, Zhang B and Zhu X:
Quercetin inhibits adenomyosis by attenuating cell proliferation,
migration and invasion of ectopic endometrial stromal cells. Drug
Des Devel Ther. 14:3815–3826. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Qi S, Zhao X, Li M, Zhang X, Lu Z, Yang C,
Zhang C, Zhang H and Zhang N: Aberrant expression of
Notch1/numb/snail signaling, an epithelial mesenchymal transition
related pathway, in adenomyosis. Reprod Biol Endocrin. 13:962015.
View Article : Google Scholar
|
|
112
|
Liu CY, Xu JY, Shi XY, Huang W, Ruan TY,
Xie P and Ding JL: M2-polarized tumor-associated macrophages
promoted epithelial-mesenchymal transition in pancreatic cancer
cells, partially through TLR4/IL-10 signaling pathway. Lab Invest.
93:844–854. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Li MQ, Wang Y, Chang KK, Meng YH, Liu LB,
Mei J, Wang Y, Wang XQ, Jin LP and Li DJ: CD4+Foxp3+ regulatory T
cell differentiation mediated by endometrial stromal cell-derived
TECK promotes the growth and invasion of endometriotic lesions.
Cell Death Dis. 5:e14362014. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Liu X, Shen M, Qi Q, Zhang H and Guo SW:
Corroborating evidence for platelet-induced epithelial-mesenchymal
transition and fibroblast-to-myofibroblast transdifferentiation in
the development of adenomyosis. Hum Reprod. 31:734–749. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Allavena P, Sica A, Solinas G, Porta C and
Mantovani A: The inflammatory micro-environment in tumor
progression: The role of tumor-associated macrophages. Crit Rev
Oncol Hemat. 66:1–9. 2008. View Article : Google Scholar
|
|
116
|
Bourdon M, Santulli P, Doridot L, Jeljeli
M, Chêne C, Chouzenoux S, Nicco C, Marcellin L, Chapron C and
Batteux F: Immune cells and Notch1 signaling appear to drive the
epithelial to mesenchymal transition in the development of
adenomyosis in mice. Mol Hum Reprod. 27:gaab0532021. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Guo SW: Cracking the enigma of
adenomyosis: An update on its pathogenesis and pathophysiology.
Reproduction. 164:R101–R121. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Zhou Z, Wang H, Zhang X, Song M, Yao S,
Jiang P, Liu D, Wang Z, Lv H, Li R, et al: Defective autophagy
contributes to endometrial epithelial-mesenchymal transition in
intrauterine adhesions. Autophagy. 18:2427–2442. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Liu ZL, Chen HH, Zheng LL, Sun LP and Shi
L: Angiogenic signaling pathways and anti-angiogenic therapy for
cancer. Signal Transduct Target Ther. 8:1982023. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Burton GJ, Watson AL, Hempstock J, Skepper
JN and Jauniaux E: Uterine glands provide histiotrophic nutrition
for the human fetus during the first trimester of pregnancy. J Clin
Endocrinol Metab. 87:2954–2959. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Guo SW: Fibrogenesis resulting from cyclic
bleeding: The Holy Grail of the natural history of ectopic
endometrium. Hum Reprod. 33:353–356. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Shaw TJ and Martin P: Wound repair: A
showcase for cell plasticity and migration. Curr Opin Cell Biol.
42:29–37. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Shaw TJ and Martin P: Wound repair at a
glance. J Cell Sci. 122:3209–3213. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Sreeramkumar V, Adrover JM, Ballesteros I,
Cuartero MI, Rossaint J, Bilbao I, Nácher M, Pitaval C, Radovanovic
I, Fukui Y, et al: Neutrophils scan for activated platelets to
initiate inflammation. Science. 346:1234–1238. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Campo S, Campo V and Benagiano G:
Infertility and adenomyosis. Obstet Gynecol Int. 2012:7861322012.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Gear ARL and Camerini D: Platelet
chemokines and chemokine receptors: Linking hemostasis,
inflammation, and host defense. Microcirculation. 10:335–350. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Koupenova M, Clancy L, Corkrey HA and
Freedman JE: Circulating platelets as mediators of immunity,
inflammation, and thrombosis. Circ Res. 122:337–351. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Levi M and van der Poll T: The role of
natural anticoagulants in the pathogenesis and management of
systemic activation of coagulation and inflammation in critically
ill patients. Semin Thromb Hemost. 34:459–468. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Salim R, Riris S, Saab W, Abramov B,
Khadum I and Serhal P: Adenomyosis reduces pregnancy rates in
infertile women undergoing IVF. Reprod Biomed Online. 25:273–277.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Collins CE and Rampton DS: Review article:
Platelets in inflammatory bowel disease-pathogenetic role and
therapeutic implications. Aliment Pharmacol Ther. 11:237–247. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Ding D, Liu X, Duan J and Guo SW:
Platelets are an unindicted culprit in the development of
endometriosis: Clinical and experimental evidence. Hum Reprod.
30:812–832. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Griffioen AW and Molema G: Angiogenesis:
Potentials for pharmacologic intervention in the treatment of
cancer, cardiovascular diseases, and chronic inflammation.
Pharmacol Rev. 52:237–268. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Seliger B, Cabrera T, Garrido F and
Ferrone S: HLA class I antigen abnormalities and immune escape by
malignant cells. Semin Cancer Biol. 12:3–13. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Dings RPM, Vang KB, Castermans K, Popescu
F, Zhang Y, Oude Egbrink MGA, Mescher MF, Farrar MA, Griffioen AW
and Mayo KH: Enhancement of T-cell-mediated antitumor response:
Angiostatic adjuvant to immunotherapy against cancer. Clin Cancer
Res. 17:3134–3145. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Sica A, Larghi P, Mancino A, Rubino L,
Porta C, Totaro MG, Rimoldi M, Biswas SK, Allavena P and Mantovani
A: Macrophage polarization in tumour progression. Semin Cancer
Biol. 18:349–355. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Wei LH, Kuo ML, Chen CA, Chou CH, Lai KB,
Lee CN and Hsieh CY: Interleukin-6 promotes cervical tumor growth
by VEGF-dependent angiogenesis via a STAT3 pathway. Oncogene.
22:1517–1527. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Raghunathachar Sahana K, Akila P, Prashant
V, Sharath Chandra B and Nataraj Suma M: Quantitation of vascular
endothelial growth factor and interleukin-6 in different stages of
breast cancer. Rep Biochem Mol Biol. 6:33–39. 2017.PubMed/NCBI
|
|
138
|
Chang KK, Liu LB, Li H, Mei J, Shao J, Xie
F, Li MQ and Li DJ: TSLP induced by estrogen stimulates secretion
of MCP-1 and IL-8 and growth of human endometrial stromal cells
through JNK and NF-κB signal pathways. Int J Clin Exp Pathol.
7:1889–1899. 2014.
|
|
139
|
Xie F, Meng YH, Liu LB, Chang KK, Li H, Li
MQ and Li DJ: Cervical carcinoma cells stimulate the angiogenesis
through TSLP promoting growth and activation of vascular
endothelial cells. Am J Reprod Immunol. 70:69–79. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Su LC, Xu WD and Huang AF: IRAK family in
inflammatory autoimmune diseases. Autoimmun Rev. 19:1024612020.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Liang S, Shi LY, Duan JY, Liu HH, Wang TT
and Li CY: Celecoxib reduces inflammation and angiogenesis in mice
with adenomyosis. Am J Transl Res. 13:2858–2866. 2021.PubMed/NCBI
|
|
142
|
Yi KW, Kim SH, Ihm HJ, Oh YS, Chae HD, Kim
CH and Kang BM: Increased expression of p21-activated kinase 4 in
adenomyosis and its regulation of matrix metalloproteinase-2 and -9
in endometrial cells. Fertil Steril. 103:1089–1097.e2. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Li B, Chen M, Liu X and Guo SW:
Constitutive and tumor necrosis factor-α-induced activation of
nuclear factor-κB in adenomyosis and its inhibition by
andrographolide. Fertil Steril. 100:568–577. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Park H, Kim SH, Cho YM, Ihm HJ, Oh YS,
Hong SH, Chae HD, Kim CH and Kang BM: Increased expression of
nuclear factor kappa-B p65 subunit in adenomyosis. Obstet Gynecol
Sci. 59:123–129. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Hoesel B and Schmid JA: The complexity of
NF-κB signaling in inflammation and cancer. Mol Cancer. 12:862013.
View Article : Google Scholar
|
|
146
|
Wang Q, Wang L, Shao J, Wang Y, Jin LP, Li
DJ and Li MQ: L-22 enhances the invasiveness of endometrial stromal
cells of adenomyosis in an autocrine manner. Int J Clin Exp Pathol.
7:5762–5771. 2014.PubMed/NCBI
|
|
147
|
Heidari Z, Moudi B, Sheibak N, Asemi-Rad
A, Keikha N, Mahmoudzadeh-Sagheb H and Ghasemi M: Interleukin 22
expression during the implantation window in the endometrium of
women with unexplained recurrent pregnancy loss and unexplained
infertility compared to healthy parturient individuals. J
Interferon Cytokine Res. 41:461–468. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Fischer CP, Kayisili U and Taylor HS:
HOXA10 expression is decreased in endometrium of women with
adenomyosis. Fertil Steril. 95:1133–1136. 2011. View Article : Google Scholar
|
|
149
|
Griffioen AW, Mans LA, de Graaf AMA,
Nowak-Sliwinska P, de Hoog CLMM, de Jong TAM, Vyth-Dreese FA, van
Beijnum JR, Bex A and Jonasch E: Rapid angiogenesis onset after
discontinuation of sunitinib treatment of renal cell carcinoma
patients. Clin Cancer Res. 18:3961–3971. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Harmsen MJ, Arduç A, Bleeker MCG,
Juffermans LJM, Griffioen AW, Jordanova ES and Huirne JAF:
Increased angiogenesis and lymphangiogenesis in adenomyosis
visualized by multiplex immunohistochemistry. Int J Mol Sci.
23:84342022. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
van Uden P, Kenneth NS, Webster R, Müller
HA, Mudie S and Rocha S: Evolutionary conserved regulation of
HIF-1β by NF-κB. PLoS Genet. 7:e10012852011. View Article : Google Scholar
|
|
152
|
Fujiwaki R, Iida K, Kanasaki H, Ozaki T,
Hata K and Miyazaki K: Cyclooxygenase-2 expression in endometrial
cancer: Correlation with microvessel count and expression of
vascular endothelial growth factor and thymidine phosphorylase. Hum
Pathol. 33:213–219. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Horng HC, Chang WH, Yeh CC, Huang BS,
Chang CP, Chen YJ, Tsui KH and Wang PH: Estrogen effects on wound
healing. Int J Mol Sci. 18:23252017. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Altmäe S, Reimand J, Hovatta O, Zhang P,
Kere J, Laisk T, Saare M, Peters M, Vilo J, Stavreus-Evers A and
Salumets A: Research resource: Interactome of human embryo
implantation: Identification of gene expression pathways,
regulation, and integrated regulatory networks. Mol Endocrinol.
26:203–217. 2012. View Article : Google Scholar
|
|
155
|
Lee JY, Lee M and Lee SK: Role of
endometrial immune cells in implantation. Clin Exp Reprod Med.
38:119–125. 2011. View Article : Google Scholar
|
|
156
|
He B, Teng XM, Hao F, Zhao M, Chen ZQ, Li
KM and Yan Q: Decreased intracellular IL-33 impairs endometrial
receptivity in women with adenomyosis. Front Endocrinol (Lausanne).
13:9280242022. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Gellersen B and Brosens JJ: Cyclic
decidualization of the human endometrium in reproductive health and
failure. Endocr Rev. 35:851–905. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Kuivasaari P, Hippelainen M, Anttila M and
Heinonen S: Effect of endometriosis on IVF/ICSI outcome: Stage
III/IV endometriosis worsens cumulative pregnancy and live-born
rates. Hum Reprod. 20:3130–3135. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Bennett WA, Lagoo-Deenadayalan S, Brackin
MN, Hale E and Cowan BD: Cytokine expression by models of human
trophoblast as assessed by a semiquantitative reverse
transcription-polymerase chain reaction technique. Am J Reprod
Immunol. 36:285–294. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Blitek A, Mendrzycka AU, Bieganska MK,
Waclawik A and Ziecik AJ: Effect of steroids on basal and
LH-stimulated prostaglandins F(2alpha) and E(2) release and
cyclooxygenase-2 expression in cultured porcine endometrial stromal
cells. Reprod Biol. 7:73–88. 2007.PubMed/NCBI
|
|
161
|
Viganò P, Somigliana E, Mangioni S,
Vignali M, Vignali M and Di Blasio AM: Expression of interleukin-10
and its receptor is up-regulated in early pregnant versus cycling
human endometrium. J Clin Endocrinol Metab. 87:5730–5736. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Lee SK, Kim CJ, Kim DJ and Kang JH: Immune
cells in the female reproductive tract. Immune Netw. 15:16–26.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Du H and Taylor HS: The role of hox genes
in female reproductive tract development, adult function, and
fertility. Cold Spring Harb Perspect Med. 6:a230022015.
|
|
164
|
Taylor HS, Igarashi P, Olive DL and Arici
A: Sex steroids mediate HOXA11 expression in the human
peri-implantation endometrium. J Clin Endocrinol Metab.
84:1129–1135. 1999.PubMed/NCBI
|
|
165
|
Guo S, Zhang D, Lu X, Zhang Q, Gu R, Sun B
and Sun Y: Hypoxia and its possible relationship with endometrial
receptivity in adenomyosis: A preliminary study. Reprod Biol
Endocrinol. 19:72021. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Taylor CT and Colgan SP: Regulation of
immunity and inflammation by hypoxia in immunological niches. Nat
Rev Immunol. 17:774–785. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Salleh N and Giribabu N: Leukemia
inhibitory factor: Roles in embryo implantation and in nonhormonal
contraception. ScientificWorldJournal. 2014:2015142014. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Yen CF, Liao SK, Huang SJ, Tabak S, Arcuri
F, Lee CL, Arici A, Petraglia F, Wang HS and Kayisli UA: Decreased
endometrial expression of leukemia inhibitory factor receptor
disrupts the STAT3 signaling in adenomyosis during the implantation
window. Reprod Sci. 24:1176–1186. 2017. View Article : Google Scholar
|
|
169
|
Xiao Y, Li T, Xia E, Yang X, Sun X and
Zhou Y: Expression of integrin β3 and osteopontin in the eutopic
endometrium of adenomyosis during the implantation window. Eur J
Obstet Gynecol Reprod Biol. 170:419–422. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Xiao Y, Sun X, Yang X, Zhang J, Xue Q, Cai
B and Zhou Y: Leukemia inhibitory factor is dysregulated in the
endometrium and uterine flushing fluid of patients with adenomyosis
during implantation window. Fertil Steril. 94:85–89. 2010.
View Article : Google Scholar
|
|
171
|
Wu H, Jiang K, Guo S, Yang J, Zhao G, Qiu
C and Deng G: IFN-τ mediated control of bovine major
histocompatibility complex class I expression and function via the
regulation of bta-miR-148b/152 in bovine endometrial epithelial
cells. Front Immunol. 9:1672018. View Article : Google Scholar
|
|
172
|
Nakamura H, Kimura T, Ogita K, Nakamura T,
Takemura M, Shimoya K, Koyama S, Tsujie T, Koyama M and Murata Y:
NF-kappaB activation at implantation window of the mouse uterus. Am
J Reprod Immunol. 51:16–21. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Kay N, Huang CY, Shiu LY, Yu YC, Chang Y,
Schatz F, Suen JL, Tsai EM and Huang SJ: TGF-β1 neutralization
improves pregnancy outcomes by restoring endometrial receptivity in
mice with adenomyosis. Reprod Sci. 28:877–887. 2021. View Article : Google Scholar
|
|
174
|
Wang T, Yuan J, Zhang J, Tian R, Ji W,
Zhou Y, Yang Y, Song W, Zhang F and Niu R: Anxa2 binds to STAT3 and
promotes epithelial to mesenchymal transition in breast cancer
cells. Oncotarget. 6:30975–30992. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Zohni KM, Gat I and Librach C: Recurrent
implantation failure: A comprehensive review. Minerva Ginecol.
68:653–667. 2016.PubMed/NCBI
|
|
176
|
Salker MS, Nautiyal J, Steel JH, Webster
Z, Sućurović S, Nicou M, Singh Y, Lucas ES, Murakami K, Chan YW, et
al: Disordered IL-33/ST2 activation in decidualizing stromal cells
prolongs uterine receptivity in women with recurrent pregnancy
loss. PLoS One. 7:e522522012. View Article : Google Scholar
|
|
177
|
Reis FM, Petraglia F and Taylor RN:
Endometriosis: Hormone regulation and clinical consequences of
chemotaxis and apoptosis. Hum Reprod Update. 19:406–418. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Burney RO, Talbi S, Hamilton AE, Vo KC,
Nyegaard M, Nezhat CR, Lessey BA and Giudice LC: Gene expression
analysis of endometrium reveals progesterone resistance and
candidate susceptibility genes in women with endometriosis.
Endocrinology. 148:3814–3826. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Tamaresis JS, Irwin JC, Goldfien GA,
Rabban JT, Burney RO, Nezhat C, DePaolo LV and Giudice LC:
Molecular classification of endometriosis and disease stage using
high-dimensional genomic data. Endocrinology. 155:4986–4999. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Peng Y, Jin Z, Liu H and Xu C: Impaired
decidualization of human endometrial stromal cells from women with
adenomyosis†. Biol Reprod. 104:1034–1044. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Kraakman MJ, Kammoun HL, Allen TL,
Deswaerte V, Henstridge DC, Estevez E, Matthews VB, Neill B, White
DA, Murphy AJ, et al: Blocking IL-6 trans-signaling prevents
high-fat diet-induced adipose tissue macrophage recruitment but
does not improve insulin resistance. Cell Metab. 21:403–416. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Ahn SH, Edwards AK, Singh SS, Young SL,
Lessey BA and Tayade C: IL-17A contributes to the pathogenesis of
endometriosis by triggering proinflammatory cytokines and
angiogenic growth factors. J Immunol. 195:2591–2600. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Jiang Y, Liao Y, He H, Xin Q, Tu Z, Kong
S, Cui T, Wang B, Quan S, Li B, et al: FoxM1 directs STAT3
expression essential for human endometrial stromal decidualization.
Sci Rep. 5:137352015. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
van Mourik MS, Macklon NS and Heijnen CJ:
Embryonic implantation: Cytokines, adhesion molecules, and immune
cells in establishing an implantation environment. J Leukoc Biol.
85:4–19. 2009. View Article : Google Scholar
|
|
185
|
Geisert R, Fazleabas A, Lucy M and Mathew
D: Interaction of the conceptus and endometrium to establish
pregnancy in mammals: Role of interleukin 1β. Cell Tissue Res.
349:825–838. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Petraglia F, Arcuri F, de Ziegler D and
Chapron C: Inflammation: A link between endometriosis and preterm
birth. Fertil Steril. 98:36–40. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Marcellin L, Santulli P, Gogusev J,
Lesaffre C, Jacques S, Chapron C, Goffinet F, Vaiman D and Méhats
C: Endometriosis also affects the decidua in contact with the fetal
membranes during pregnancy. Hum Reprod. 30:392–405. 2015.
View Article : Google Scholar
|
|
188
|
Fraccaroli L, Alfieri J, Leiros CP and
Ramhorst R: Immunomodulatory effects of chemokines during the early
implantation window. Front Biosci (Elite Ed). 1:288–298.
2009.PubMed/NCBI
|
|
189
|
Lukassen HGM, Joosten I, van Cranenbroek
B, van Lierop MJC, Bulten J, Braat DDM and van der Meer A: Hormonal
stimulation for IVF treatment positively affects the
CD56bright/CD56dim NK cell ratio of the endometrium during the
window of implantation. Mol Hum Reprod. 10:513–520. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Sinclair DC, Mastroyannis A and Taylor HS:
Leiomyoma simultaneously impair endometrial BMP-2-mediated
decidualization and anticoagulant expression through secretion of
TGF-β3. J Clin Endocrinol Metab. 96:412–421. 2011. View Article : Google Scholar
|
|
191
|
Lee KY, Jeong JW, Wang J, Ma L, Martin JF,
Tsai SY, Lydon JP and DeMayo FJ: Bmp2 is critical for the murine
uterine decidual response. Mol Cell Biol. 27:5468–5478. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Teklenburg G, Salker M, Molokhia M, Lavery
S, Trew G, Aojanepong T, Mardon HJ, Lokugamage AU, Rai R, Landles
C, et al: Natural selection of human embryos: Decidualizing
endometrial stromal cells serve as sensors of embryo quality upon
implantation. PLoS One. 5:e102582010. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Makrigiannakis A, Petsas G, Toth B,
Relakis K and Jeschke U: Recent advances in understanding
immunology of reproductive failure. J Reprod Immunol. 90:96–104.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Arruvito L, Sotelo AI, Billordo A and
Fainboim L: A physiological role for inducible FOXP3(+) Treg cells.
Lessons from women with reproductive failure. Clin Immunol.
136:432–441. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Salmeri N, Farina A, Candiani M, Dolci C,
Bonavina G, Poziello C, Viganò P and Cavoretto PI: Endometriosis
and impaired placentation: A prospective cohort study comparing
uterine arteries doppler pulsatility index in pregnancies of
patients with and without moderate-severe disease. Diagnostics
(Basel). 12:10242022. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Godbole G and Modi D: Regulation of
decidualization, interleukin-11 and interleukin-15 by homeobox A 10
in endometrial stromal cells. J Reprod Immunol. 85:130–139. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Ashkar AA, Black GP, Wei Q, He H, Liang L,
Head JR and Croy BA: Assessment of requirements for IL-15 and IFN
regulatory factors in uterine NK cell differentiation and function
during pregnancy. J Immunol. 171:2937–2944. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Chegini N, Roberts M and Ripps B:
Differential expression of interleukins (IL)-13 and IL-15 in
ectopic and eutopic endometrium of women with endometriosis and
normal fertile women. Am J Reprod Immunol. 49:75–83. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Visse R and Nagase H: Matrix
metalloproteinases and tissue inhibitors of metalloproteinases:
Structure, function, and biochemistry. Circ Res. 92:827–839. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Vannuccini S, Clifton VL, Fraser IS,
Taylor HS, Critchley H, Giudice LC and Petraglia F: Infertility and
reproductive disorders: Impact of hormonal and inflammatory
mechanisms on pregnancy outcome. Hum Reprod Update. 22:104–115.
2016. View Article : Google Scholar
|
|
201
|
Oladosu FA, Tu FF and Hellman KM:
Nonsteroidal antiinflammatory drug resistance in dysmenorrhea:
Epidemiology, causes, and treatment. Am J Obstet Gynecol.
218:390–400. 2018. View Article : Google Scholar
|
|
202
|
Carrarelli P, Funghi L, Bruni S, Luisi S,
Arcuri F and Petraglia F: Naproxen sodium decreases prostaglandins
secretion from cultured human endometrial stromal cells modulating
metabolizing enzymes mRNA expression. Gynecol Endocrinol.
32:319–322. 2016. View Article : Google Scholar
|
|
203
|
Jin Z, Wu X, Liu H and Xu C: Celecoxib, a
selective COX-2 inhibitor, markedly reduced the severity of
tamoxifen-induced adenomyosis in a murine model. Exp Ther Med.
19:3289–3299. 2020.PubMed/NCBI
|
|
204
|
Achache H and Revel A: Endometrial
receptivity markers, the journey to successful embryo implantation.
Hum Reprod Update. 12:731–746. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Olivares C, Bilotas M, Buquet R, Borghi M,
Sueldo C, Tesone M and Meresman G: Effects of a selective
cyclooxygenase-2 inhibitor on endometrial epithelial cells from
patients with endometriosis. Hum Reprod. 23:2701–2708. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Harmsen MJ, Juffermans LJM, Kroon MO,
Griffioen AW and Huirne JAF: Anti-angiogenic therapy as potential
treatment for adenomyosis. Angiogenesis. 28:122025. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Nyachieo A, Siristatidis CS and Vaidakis
D: Nonsteroidal anti-inflammatory drugs for assisted reproductive
technology. Cochrane Database Syst Rev. 10:CD0076182019.PubMed/NCBI
|
|
208
|
Khan KN, Kitajima M, Hiraki K, Fujishita
A, Sekine I, Ishimaru T and Masuzaki H: Changes in tissue
inflammation, angiogenesis and apoptosis in endometriosis,
adenomyosis and uterine myoma after GnRH agonist therapy. Hum
Reprod. 25:642–653. 2010. View Article : Google Scholar
|
|
209
|
Guo S, Li Z, Yan L, Sun Y and Feng Y: GnRH
agonist improves pregnancy outcome in mice with induced adenomyosis
by restoring endometrial receptivity. Drug Des Devel Ther.
12:1621–1631. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Harada T, Khine YM, Kaponis A, Nikellis T,
Decavalas G and Taniguchi F: The impact of adenomyosis on women's
fertility. Obstet Gynecol Surv. 71:557–568. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Niu Z, Chen Q, Sun Y and Feng Y: Long-term
pituitary downregulation before frozen embryo transfer could
improve pregnancy outcomes in women with adenomyosis. Gynecol
Endocrinol. 29:1026–1030. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Wang PH, Liu WM, Fuh JL, Cheng MH and Chao
HT: Comparison of surgery alone and combined surgical-medical
treatment in the management of symptomatic uterine adenomyoma.
Fertil Steril. 92:876–885. 2009. View Article : Google Scholar
|
|
213
|
Al-Inany HG, Youssef MA, Aboulghar M,
Broekmans F, Sterrenburg M, Smit J and Abou-Setta AM: GnRH
antagonists are safer than agonists: An update of a Cochrane
review. Hum Reprod Update. 17:4352011. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Lessey BA, Dong A, Deaton JL, Angress D,
Savaris RF and Walker SJ: Inflammatory changes after medical
suppression of suspected endometriosis for implantation failure:
Preliminary results. Int J Mol Sci. 25:68522024. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Barretta M, Vignali M, La Marca A and
Grandi G: The oral GnRH antagonists, a new class of drugs in
gynecology: From pharmacokinetics to possible clinical
applications. Expert Opin Drug Metab Toxicol. 1–13. 2024.Epub ahead
of print. PubMed/NCBI
|
|
216
|
McCormack PL: Dienogest: A review of its
use in the treatment of endometriosis. Drugs. 70:2073–2088. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Horie S, Harada T, Mitsunari M, Taniguchi
F, Iwabe T and Terakawa N: Progesterone and progestational
compounds attenuate tumor necrosis factor alpha-induced
interleukin-8 production via nuclear factor kappa B inactivation in
endometriotic stromal cells. Fertil Steril. 83:1530–1535. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Prathoomthong S, Tingthanatikul Y,
Lertvikool S, Rodratn N, Waiyaput W, Dittharot K, Sroyraya M and
Sophonsritsuk A: The effects of dienogest on macrophage and natural
killer cells in adenomyosis: A randomized controlled study. Int J
Fertil Steril. 11:279–286. 2018.
|
|
219
|
de Mattos RM, Machado DE, Perini JA,
Alessandra-Perini J, Meireles da Costa NDO, Wiecikowski AFDRDO,
Cabral KMDS, Takiya CM, Carvalho RS and Nasciutti LE: Galectin-3
plays an important role in endometriosis development and is a
target to endometriosis treatment. Mol Cell Endocrinol. 486:1–10.
2019. View Article : Google Scholar
|
|
220
|
Zhu Y, Zheng B, Zhang Y, Li M, Jiang Y,
Zhou J, Zhang Y, Kang N, Wu M, Yan Y, et al: Ectopic endometrial
stromal cell-derived extracellular vesicles encapsulating
microRNA-25-3p induce endometrial collagen I deposition impairing
decidualization in endometriosis. Mol Hum Reprod. 30:gaae0422024.
View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Yamamoto N, Kitano S, Koyama T, Ikeda M,
Mizugaki H, Narikiyo T, Yamaguchi Y, Ishida T, Takubo R, Ogami C,
et al: Phase I study of the safety and clinical activity of the
interleukin-8 inhibitor AMY109 combined with atezolizumab in
patients with advanced solid cancers. J Immunother Cancer.
12:e0092622024. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Nishimoto-Kakiuchi A, Sato I, Nakano K,
Ohmori H, Kayukawa Y, Tanimura H, Yamamoto S, Sakamoto Y, Nakamura
G, Maeda A, et al: A long-acting anti-IL-8 antibody improves
inflammation and fibrosis in endometriosis. Sci Transl Med.
15:eabq58582023. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Xue J, Zhang H, Liu W, Liu M, Shi M, Wen Z
and Li C: Metformin inhibits growth of eutopic stromal cells from
adenomyotic endometrium via AMPK activation and subsequent
inhibition of AKT phosphorylation: A possible role in the treatment
of adenomyosis. Reproduction. 146:397–406. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Cheng J, Li C, Ying Y, Lv J, Qu X, McGowan
E, Lin Y and Zhu X: Metformin alleviates endometriosis and
potentiates endometrial receptivity via decreasing VEGF and MMP9
and increasing leukemia inhibitor factor and HOXA10. Front
Pharmacol. 13:7502082022. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Hou Y, Qin Z, Fan K, Xu Y and Huang X:
Combination therapeutic effects of high intensity focused
ultrasound and Metformin for the treatment of adenomyosis. Exp Ther
Med. 15:2104–2108. 2018.PubMed/NCBI
|
|
226
|
Sullender RT, Agarwal RK, Jacobs MB,
Wessels JM, Foster WG and Agarwal SK: Pilot study of IL-1
antagonist anakinra for treatment of endometriosis. Int J Womens
Health. 16:1583–1593. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Xu Z, Zhao F, Lin F, Chen J and Huang Y:
Lipoxin A4 inhibits the development of endometriosis in mice: The
role of anti-inflammation and anti-angiogenesis. Am J Reprod
Immunol. 67:491–497. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Kumar R, Clerc AC, Gori I, Russell R,
Pellegrini C, Govender L, Wyss JC, Golshayan D and Canny GO:
Lipoxin A4 prevents the progression of de novo and established
endometriosis in a mouse model by attenuating prostaglandin E2
production and estrogen signaling. PLoS One. 9:e897422014.
View Article : Google Scholar
|
|
229
|
Celik O, Ersahin A, Acet M, Celik N,
Baykus Y, Deniz R, Ozerol E and Ozerol I: Disulfiram, as a
candidate NF-κB and proteasome inhibitor, prevents endometriotic
implant growing in a rat model of endometriosis. Eur Rev Med
Pharmacol Sci. 20:4380–4389. 2016.PubMed/NCBI
|
|
230
|
Akdaş Reis Y, Tapisiz OL, Göktolga Ü,
Şimşek G, Erten Ö, Kiykaç Altinbaş Ş and Erkaya S: The effect of
disulfiram in the prevention of postoperative adhesion formation in
an experimental rat uterine horn model. Reprod Sci. 28:2650–2660.
2021. View Article : Google Scholar
|
|
231
|
Guan X, Liu D, Zhou H, Dai C, Wang T, Fang
Y, Jia Y and Li K: Melatonin improves pregnancy outcomes in
adenomyosis mice by restoring endometrial receptivity via
NF-κB/apoptosis signaling. Ann Transl Med. 10:13172022. View Article : Google Scholar
|
|
232
|
Chuffa LGDA, Lupi LA, Cucielo MS, Silveira
HS, Reiter RJ and Seiva FRF: Melatonin promotes uterine and
placental health: Potential molecular mechanisms. Int J Mol Sci.
21:3002019. View Article : Google Scholar
|
|
233
|
Wong M, Ziring D, Korin Y, Desai S, Kim S,
Lin J, Gjertson D, Braun J, Reed E and Singh RR: TNFalpha blockade
in human diseases: Mechanisms and future directions. Clin Immunol.
126:121–136. 2008. View Article : Google Scholar
|
|
234
|
Barrier BF, Bates GW, Leland MM, Leach DA,
Robinson RD and Propst AM: Efficacy of anti-tumor necrosis factor
therapy in the treatment of spontaneous endometriosis in baboons.
Fertil Steril. 81(Suppl 1): S775–S779. 2004. View Article : Google Scholar
|
|
235
|
Zulfikaroglu E, Kilic S, Islimye M, Aydin
M, Zergeroglu S and Batioglu S: Efficacy of anti-tumor necrosis
factor therapy on endometriosis in an experimental rat model. Arch
Gynecol Obstet. 283:799–804. 2011. View Article : Google Scholar
|
|
236
|
Önalan G, Tohma YA and Zeyneloğlu HB:
Effect of etanercept on the success of assisted reproductive
technology in patients with endometrioma. Gynecol Obstet Inves.
83:358–364. 2018. View Article : Google Scholar
|
|
237
|
Etemadi S, Abtahi Froushani SM, Hashemi
Asl SM and Mahmoudian A: Combined atorvastatin and pentoxifylline
in ameliorating inflammation induced by complete Freund's adjuvant.
Inflammopharmacology. 30:935–944. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Kamencic H and Thiel JA: Pentoxifylline
after conservative surgery for endometriosis: A randomized,
controlled trial. J Minim Invasive Gynecol. 15:62–66. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Creus M, Fábregues F, Carmona F, del Pino
M, Manau D and Balasch J: Combined laparoscopic surgery and
pentoxifylline therapy for treatment of endometriosis-associated
infertility: A preliminary trial. Hum Reprod. 23:1910–1916. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
240
|
Roussev RG, Ng SC and Coulam CB: Natural
killer cell functional activity suppression by intravenous
immunoglobulin, intralipid and soluble human leukocyte antigen-G.
Am J Reprod Immunol. 57:262–269. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Coulam CB: Intralipid treatment for women
with reproductive failures. Am J Reprod Immunol. 85:e132902021.
View Article : Google Scholar
|
|
242
|
Etrusco A, Barra F, Chiantera V, Ferrero
S, Bogliolo S, Evangelisti G, Oral E, Pastore M, Izzotti A, Venezia
R, et al: Current medical therapy for adenomyosis: From bench to
bedside. Drugs. 83:1595–1611. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Wang H, Gan Z, Wang Y, Hu D, Zhang L, Ye F
and Duan P: A noninvasive menstrual blood-based diagnostic platform
for endometriosis using digital droplet enzyme-linked immunosorbent
assay and single-cell RNA sequencing. Research (Wash D C).
8:06522025.PubMed/NCBI
|
|
244
|
Moses AS, Taratula OR, Lee H, Luo F, Grenz
T, Korzun T, Lorenz AS, Sabei FY, Bracha S, Alani AWG, et al:
Nanoparticle-based platform for activatable fluorescence imaging
and photothermal ablation of endometriosis. Small. 16:e19069362020.
View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Wu N, Han Z, Lv W, Huang Y, Zhu J, Deng J
and Xue Q: Reprogramming peritoneal macrophages with outer membrane
vesicle-coated PLGA nanoparticles for endometriosis prevention.
Biomaterials. 319:1231982025. View Article : Google Scholar : PubMed/NCBI
|
|
246
|
Yuxue J, Ran S, Minghui F and Minjia S:
Applications of nanomaterials in endometriosis treatment. Front
Bioeng Biotechnol. 11:11841552023. View Article : Google Scholar : PubMed/NCBI
|
|
247
|
Marí-Alexandre J, Barceló-Molina M,
Belmonte-López E, García-Oms J, Estellés A, Braza-Boïls A and
Gilabert-Estellés J: Micro-RNA profile and proteins in peritoneal
fluid from women with endometriosis: Their relationship with
sterility. Fertil Steril. 109:675–684.e2. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
248
|
Dai L, Gu L and Di W: MiR-199a attenuates
endometrial stromal cell invasiveness through suppression of the
IKKβ/NF-κB pathway and reduced interleukin-8 expression. Mol Hum
Reprod. 18:136–145. 2012. View Article : Google Scholar
|
|
249
|
Kaczmarek JC, Kowalski PS and Anderson DG:
Advances in the delivery of RNA therapeutics: From concept to
clinical reality. Genome Med. 9:602017. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Abdulmonem WA, Ahsan M, Mallick AK,
Mohamed AH, Waggiallah HA, Shafie A, Alzahrani HS, Ashour AA, Rab
SO, Mirdad MT and Ali HTO: The role of exosomal miRNAs in female
infertility: Therapeutic potential and mechanisms of action. Stem
Cell Rev Rep. 21:1141–1159. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Juárez-Barber E, Segura-Benítez M,
Carbajo-García MC, Bas-Rivas A, Faus A, Vidal C, Giles J and
Labarta E: Extracellular vesicles secreted by adenomyosis
endometrial organoids contain miRNAs involved in embryo
implantation and pregnancy. Reprod Biomed Online. 46:470–481. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
252
|
Liu BHM, Lin Y, Long X, Hung SW, Gaponova
A, Ren F, Zhavoronkov A, Pun FW and Wang CC: Utilizing AI for the
Identification and validation of novel therapeutic targets and
repurposed drugs for endometriosis. Adv Sci (Weinh).
12:e24065652025. View Article : Google Scholar :
|
|
253
|
Guan YG, Liao JB, Li KY, Li YC, Song Y,
Ling J and Su ZR: Potential mechanisms of an antiadenomyosis
Chinese herbal formula shaoyao-gancao decoction in primary cell
culture model. Evid Based Complement Alternat Med. 2014:9829132014.
View Article : Google Scholar : PubMed/NCBI
|
|
254
|
Zhu B, Chen Y, Zhang H, Liu X and Guo SW:
Resveratrol reduces myometrial infiltration, uterine hyperactivity,
and stress levels and alleviates generalized hyperalgesia in mice
with induced adenomyosis. Reprod Sci. 22:1336–1349. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Maia H Jr, Haddad C, Pinheiro N and Casoy
J: Advantages of the association of resveratrol with oral
contraceptives for management of endometriosis-related pain. Int J
Womens Health. 4:543–549. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Han SJ, Jung SY, Wu SP, Hawkins SM, Park
MJ, Kyo S, Qin J, Lydon JP, Tsai SY, Tsai MJ, et al: Estrogen
receptor β modulates apoptosis complexes and the inflammasome to
drive the pathogenesis of endometriosis. Cell. 163:960–974. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
257
|
Park Y, Cho YJ, Sung N, Park MJ, Guan X,
Gibbons WE, O'Malley BW and Han SJ: Oleuropein suppresses
endometriosis progression and improves the fertility of mice with
endometriosis. J Biomed Sci. 29:1002022. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Jiang X, Chen Y, Zhu H, Wang B, Qu P, Chen
R and Sun X: Sodium tanshinone IIA sulfonate ameliorates bladder
fibrosis in a rat model of partial bladder outlet obstruction by
inhibiting the TGF-β/Smad pathway activation. PLoS One.
10:e1296552015.
|
|
259
|
Liu L, Chen L, Jiang C, Guo J, Xie Y, Kang
L and Cheng Z: Berberine inhibits the LPS-induced proliferation and
inflammatory response of stromal cells of adenomyosis tissues
mediated by the LPS/TLR4 signaling pathway. Exp Ther Med.
14:6125–6130. 2017.PubMed/NCBI
|
|
260
|
Wu L, Yang Z, Chen H and Piao Y:
Diagnostic accuracy of combination of CA125 for endometriosis: A
network meta-analysis. Int J Gynaecol Obstet. Sep 9–2025.Epub ahead
of print. View Article : Google Scholar
|
|
261
|
Rižner TL: Noninvasive biomarkers of
endometriosis: myth or reality? Expert Rev Mol Diagn. 14:365–385.
2014. View Article : Google Scholar
|
|
262
|
Mu F, Harris HR, Rich-Edwards JW,
Hankinson SE, Rimm EB, Spiegelman D and Missmer SA: A prospective
study of inflammatory markers and risk of endometriosis. Am J
Epidemiol. 187:515–522. 2018. View Article : Google Scholar
|
|
263
|
Măluţan AM, Drugan T, Ciortea R,
Mocan-Hognogi RF, Bucuri C, Rada MP and Mihu D: Serum
anti-inflammatory cytokines for the evaluation of inflammatory
status in endometriosis. J Res Med Sci. 20:668–674. 2015.
View Article : Google Scholar :
|
|
264
|
Inagaki J, Sugiura-Ogasawara M, Nomizu M,
Nakatsuka M, Ikuta K, Suzuki N, Kaihara K, Kobayashi K, Yasuda T,
Shoenfeld Y, et al: An association of IgG anti-laminin-1
autoantibodies with endometriosis in infertile patients. Hum
Reprod. 18:544–549. 2003. View Article : Google Scholar : PubMed/NCBI
|




















