Introduction
Pemphigus vulgaris (PV) is a life threatening
autoimmune blistering disease characterized by loss of cell-cell
adhesion of keratinocytes (acantholysis) and the formation of
non-healing suprabasal intraepidermal blisters (1,2).
Its annual incidence ranges from 0.098-5 patients per 100,000
individuals (3). Although the
use of corticosteroids and immunosuppressive agents has markedly
improved clinical outcomes in patients with PV, prolonged
administration of these drugs increases the risk of serious adverse
events, with infections being particularly notable (4). Growing evidence indicates that
autoantibody-induced keratinocyte acantholysis involves multiple
mechanisms. Autoantibodies binding to PV antigens not only directly
disrupt desmosomal adhesive function but also activate
intracellular kinase signaling pathways downstream of ligated
antigens. These include epidermal growth factor receptor (EGFR),
Src, mammalian target of rapamycin (mTOR), p38 mitogen-activated
protein kinases (MAPK), as well as elevated intracellular calcium
and activation of cell death cascades, leading to the dissociation
of keratinocyte and formation of acantholysis (5). Therefore, uncovering the mechanisms
by which autoantibodies trigger acantholysis and cell death is
crucial for the development of targeted therapeutics.
Recently, increasing evidence has demonstrated that
interleukins play significant roles and represent promising
therapeutic targets in skin-related diseases (6,7).
Our previous study found that interleukin-37 (IL-37) inhibits
keratinocyte dissociation and desmoglein-3 (Dsg3) endocytosis
through upregulating caveolin-1 and inhibiting signal transducer
and activator of transcription 3 (STAT3) pathways (8). IL-37 is predominantly expressed in
circulating monocytes, macrophages, dendritic cells, tonsillar B
cells, and plasma cells, as well as in epithelial cells of the skin
and gut in response to inflammation (9). In psoriasis, IL-37 expression has
been reported to be decreased (10); however, findings in atopic
dermatitis remain inconsistent (9,11,12).
As a member of interleukin-1 superfamily, IL-37
functions as a natural suppressor of inflammatory and immune
responses (13,14). It modulates both innate and
acquired immunity by maintaining the cytokine balance away from
excessive inflammation (15,16). Notably, IL-37 binds to the
IL-18Rα and recruits the co-receptor IL-1R8 to form a tripartite
complex, triggering a signaling cascade that involves mTOR, MAPK
and NF-kB pathways (17).
Johnston et al (18)
identified synergistic interactions between IL-1 and EGFR, a
pivotal regulator of epithelial homeostasis, in enhancing
keratinocyte antimicrobial defense mechanisms. Furthermore, it has
been demonstrated that PV-IgG induces EGFR activation, and clinical
use of EGFR inhibitors such as lapatinib prevents keratinocyte
blister formation (19). These
findings led to the hypothesis that IL-37 may regulate
EGFR-mediated keratinocyte acantholysis and cell death.
The present study stimulated HaCaT keratinocytes
with anti-Dsg3 antibody to establish an in vitro PV model
and co-cultured these cells with IL-37 to investigate its role and
underlying mechanisms in PV pathogenesis. The results revealed that
IL-37 treatment markedly reduced keratinocyte dissociation and
apoptosis in anti-Dsg3 antibodies-treated-keratinocytes through the
IL-1R8/TNF-alpha-converting enzyme (TACE/ADAM-17)/EGFR pathway.
Collectively, these findings identified IL-37 as a pivotal
regulator of EGFR activation and keratinocyte dissociation in PV,
highlighting its potential as a therapeutic target.
Materials and methods
Collection of serum and skin tissues
Between June 2019 and June 2020, serum and skin
tissues were collected from 15 patients with PV (8 males and 7
females; mean age 46.00±12.63 years, range 21-70) undergoing
treatment in the Department of Dermatology in People's Hospital of
Xinjiang Uygur Autonomous Region. Healthy control serum (15
samples, 7 males and 8 females; mean age 45.20±12.03 years, range
25-63) and skin tissues (5 samples) were collected from
participants undergoing skin plastic surgery at the same hospital.
The present study was approved by the Ethics Committee of the
People's Hospital of Xinjiang Uygur Autonomous Region (approval no.
2019030616). The procedures followed were in accordance with the
Helsinki Declaration of 1975, as revised in 1983.
Cell culture
HaCaT cells were obtained from Cell Bank of the
Chinese Academy of Sciences (cat. no. SCSP-5091). The cell line has
tested negative for mycoplasma, bacteria and fungi and has been
verified by STR profiling. HaCaT cells were cultured in DMEM
(Gibco; Thermo Fisher Scientific, Inc. Massachusetts, USA.)
containing 10% fetal bovine serum (FBS; Gibco; Thermo Fisher
Scientific, Inc.) at 37°C and 5% CO2 incubator.
CCK-8 assay
HaCaT cells (2×103 cell/well) were
cultured in 96-well plate and treated with different concentration
of IL-37 recombinant protein (0, 25, 50, 100, and 200 ng/ml). Then,
10 μl of CCK-8 (Beyotime Biotechnology) were added and
cultured for 1 h, the absorbance were detected at 450 nm.
Construction of PV-cell model
To establish the in vitro model of
antibody-induced acantholysis, HaCaT cells placed in 6-well plates
were treated with mouse monoclonal anti-Dsg3 antibody AK23 (cat.
no. MA5-51877; Thermo Fisher Scientific, Inc.) at a dilution of
1:2,000 or 1:1,000 for 24 h at 37°C. Subsequently, the cells were
treated with IL-37 recombinant protein (isoform b; 100 ng/ml; cat.
no. 10155-HNAE; Sino Biological, Inc.) for 24 h at 37°C.
Cell transfection and treatment
To knockdown the expression of IL-1R8, IL-18Rα and
ADAM17, HaCaT cells were transfected with 50 nM of IL-1R8-,
IL-18Rα- and ADAM17-specific small interference RNA (si-RNA;
Shanghai GenePharma Co., Ltd.) using Lipofectamine® 3000
(Invitrogen; Thermo Fisher Scientific, Inc.) for 48 h at 37°C
according to the manufacturer's instructions. The knockdown
efficiency was detected by western blot assay. Subsequently, HaCaT
cells were treated with AK23 for 24 h and 100 ng/ml of IL-37
recombinant protein for another 24 h. The sequences of siRNAs were
as follows: si-IL-1R8 #1: 5'-GCAAGUUCGUGAACUUCAUCC-3', and
5'-AUGAAGUUCACGAACUUGCGG-3'; si-IL-1R8 #2:
5'-GCGUCGCUCCUCUGCUCAACA-3', and 5'-UUGAGCAGAGGAGCGACGCCG';
si-IL-1R8 #3: 5'-GGGAGAGUUUGCUCCCAAAGG-3', and
5'-UUUGGGAGCAAACUCUCCCCU-3'; si-IL-18R #1:
5'-GAGUGAGAUUGUCAGUGUAAG-3' and 5'-UACACUGACAAUCUCACUCUU-3';
si-IL-18R #2: 5'-GAGUCUUAAUCUUCAGAAACA-3' and
5'-UUUCUGAAGAUUAAGACUCGG-3'; si-IL-18R #3:
5'-GAGAAGAGAUGAAACAUUAAC-3' and 5'-UAAUGUUUCAUCUCUUCUCGU-3';
si-ADAM17 #1: 5'-GAUUUGACCUACAAAUCAAUC-3' and
5'-UUGAUUUGUAGGUCAAAUCUA-3'; si-ADAM17 #2:
5'-AGAGAUCUACAGACUUCAACA-3' and 5'-UUGAAGUCUGUAGAUCUCUUU-3';
si-ADAM17 #3: 5'-GAAUCGUGUUGACAGCAAAGA-3' and
5'-UUUGCUGUCAACACGAUUCUG-3'; si-NC: 5'-UUCUCCGAACGUGUCACGUTT-3' and
5'-ACGUGACACGUUCGGAGAATT-3'.
For EGFR inhibition assay, HaCaT cells were
pre-treated with 1 μM of EGFR inhibitor AG1478
(MedChemExpress) at 37°C for 30 min followed by treatment with AK23
for 24 h at 37°C.
Enzyme-linked immunosorbent assay
(ELISA)
The levels of IL-37 (cat. no. PI652), IL-6 (cat. no.
PI325), IL-10 (cat. no. PI536), and IL-17 (cat. no. PI550) in the
serum or cellular supernatant were measured using ELISA kits
(Beyotime Biotechnology) according to the manufacturer's
instructions. Briefly, the serum (50 μl sample analysis
buffer and 50 μl serum/well) or cellular supernatant (100
μl/well) was added and cultured for 120 min at room
temperature. Then, biotinylated antibody (100 μl/well) was
added and cultured for 60 min at room temperature. Following this,
streptavidin (100 μl/well) labeled with horseradish
peroxidase was added and incubated at room temperature for 20 min
away from light. Then, a color-developing agent (TMB solution; 100
μl/well) was added, and the reaction wells were sealed using
a sealing plate film (white), and incubated at room temperature for
15 min away from light. Finally, a termination solution (50
μl/well) was added and the absorbance was measure at 450 nm
immediately after mixing.
Immunohistochemical staining
Skin tissues were fixed with 4% paraformaldehyde for
15 min at room temperature, and then permeabilized with 0.5% Triton
X-100 for 20 min at room temperature. After blocking with normal
goat serum in PBS containing 0.1% Tween for 30 min at room
temperature, the tissue sections were incubated with IL-37 antibody
(cat. no. ab278499; Abcam) overnight at 4°C. The next day, the skin
tissues were incubated with biotin-labeled goat anti-rabbit IgG
(H+L) antibody (Thermo Fisher Scientific, Inc.; 1:200) for 15 min
at room temperature. They were then counterstained with hematoxylin
for 5 min at room temperature. Finally, images of the skin tissue
sections were captured using a light microscope (Leica DM4000 B
LED; Leica Microsystems GmbH).
Dsg3 immunocytofluorescence
HaCaT keratinocytes cultured with or without mouse
anti-Dsg3 antibody (1:1,000 dilution) were fixed with 4%
paraformaldehyde, incubated with 0.1% Triton X-100 for 10 min at
room temperature and then blocked with 5% goat serum for 1 h at
room temperature. Subsequently, mouse anti-Dsg3 antibodies (1:100
dilution; cat. no. NBP1-78984; Novus Biologicals; Bio-Techne) were
added and incubated at 4°C. The next day, Alexa Fluor 594-labelled
goat anti-rabbit IgG (cat. no. 33112ES60; Shanghai Yeasen
Biotechnology Co., Ltd.) was added and incubated for another 2 h at
37°C in the dark. The nuclei were stained with DAPI at room
temperature for 5 min (1:5,000 dilution; Beyotime Biotechnology).
Images of fluorescence staining were captured using a fluorescence
microscope.
Reverse transcription-quantitative (RT-q)
PCR
Cells (1×106 cell) were collected and
lysed with TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.), after which their RNA was extracted. RNA (1
μg) was reverse transcribed into the cDNA using the
PrimeScript RT Master Mix kit (Takara Biotechnology Co., Ltd.)
following the manufacturer's instructions. The mRNA levels of these
samples were then detected using SYBR® Premix Ex Taq II
(Qiagen China Co., Ltd.) according to the manufacturer's
instructions. The reactions program was as follows: 95°C for 5 min,
followed by 40 cycles at 95°C for 5 sec and 60°C for 34 sec and
finally extended at 72°C for 10 min. Gene expression levels were
quantified using the 2−ΔΔCq method (20), with β-actin serving as the
endogenous reference gene. The primers used were: IL-37: Forward:
5'-GACCAGGATCACAAAGTACTGG-3', Reverse:
5'-GAGCTCAAGGATGAGGCTAATG-3'; IL-10: Forward:
5'-GCTGGAGGACTTTAAGGGTTAC-3', Reverse: 5'-GATGTCTGGGTCTTGGTTCTC-3';
IL-6: Forward: 5'-CACTCACCTCTTCAGAACGAAT-3', Reverse:
5'-GCTGCTTTCACACATGTTACTC-3'; IL-17: Forward:
5'-GAAATCCAGGATGCCCAAATTC-3', Reverse:
5'-GAGGTGGATCGGTTGTAGTAATC-3'; β-actin: Forward:
5'-AGCCTTCCTTCCTGGGCAT-3', Reverse: 5'-TGATCTTCATTGTGCTGGGTG-3'.
All experiments were performed in three independent biological
replicates.
Western blotting
All the cell samples from all the groups and the
skin tissue samples from patients with PV were lysed with RIPA
buffer (cat. no. P0013C; Beyotime Biotechnology,) for 10 min at
4°C. Protein concentration was determined using the Bradford
Protein Concentration Assay Kit (Beyotime Biotechnology). Total
protein (20 μg) were electrophoresed on 6, 8, 10 or 12%
SDS-PAGE gel and subsequently transferred to a PVDF membrane. The
membranes were then blocked using a blocking buffer (Beyotime
Biotechnology) for 1 h at room temperature. Following this, the
membranes were incubated with the following specific primary
antibodies overnight at 4°C: Rabbit anti-IL-37 monoclonal antibody
(cat. no. ab278499; Abcam), rabbit anti-Bax monoclonal antibody
(cat. no. ab32503; Abcam), rabbit anti-Bcl-2 polyclonal antibody
(cat. no. ab59348; Abcam), rabbit anti-caspase 3 antibody (cat. no.
9662; Cell Signaling Technology, Inc.), rabbit anti-cleaved
caspase3 antibody (cat. no. 9661; Cell Signaling Technology, Inc.),
mouse anti-IL-1R8 antibody (cat. no. sc-271864; Santa Cruz
Biotechnology, Inc.), mouse anti-IL-18Rα antibody (cat. no.
PA5-115404; Thermo Fisher Scientific, Inc.), rabbit anti-EGFR
phospho Y1173 antibody (cat. no. ab5652; Abcam), rabbit anti-EGFR
antibody (cat. no. ab52894; Abcam), rabbit anti-ADAM17 polyclonal
antibody (cat. no. ab39162; Abcam), anti-phosphorylated (p-) ERK1/2
antibody (cat. no. ab201015; Abcam), anti-ERK1/2 antibody (cat. no.
ab184699; Abcam), anti-p-AKT (Ser473) antibody (cat. no. 9271; Cell
Signaling Technology, Inc.), anti-AKT antibody (cat. no. 4691; Cell
Signaling Technology, Inc.), anti-p-STAT3 antibody (cat. no.
ab76315; Abcam), and anti-STAT3 antibody (cat. no. ab68153; Abcam).
Then, the appropriate HRP-conjugated goat anti-rabbit IgG or goat
anti-mouse antibodies were added and incubated for 1 h at room
temperature. Finally, positive bands were visualized using an
imaging system (Bio-Rad Laboratories, Inc.). Densitometric analysis
was performed using ImageJ software (version 1.53; National
Institutes of Health).
Dispase-based dissociation assay
HaCaT keratinocytes were cultivated on 24-well
plates at a density of 1×106 per well. The cells were
then stimulated with or without anti-Dsg3 antibody in the presence
or absence of IL-37 and/or EGFR activator NSC228155 for 15 min at
37°C. After two washes using phosphate-buffered saline (PBS), each
well was treated with 2 U/ml of Dispase II (MilliporeSigma) and
incubated at 37°C for 30 min. Dispase II was then gently removed,
and 1 ml of PBS was added. The cells were then pipetted up and down
10 times using a 1 ml electric pipette at a constant speed to
disrupt the integrity of the monolayer cells and dissociate the
cells. Finally, cell debris was stained with 0.01% crystal violet
(MilliporeSigma) at room temperature for 1 min and the number of
fragments was measured using an inverted microscope. The experiment
was performed three times.
Mitochondrial membrane potential
The PV-cell model was treated with IL-37 recombinant
protein for 24 h, after which it was washed twice with PBS. The
cells were then stained with 1.0 mM of JC-1 (cat. no. C2005;
Beyotime Biotechnology) at 37°C for 10 min. The fluorescence
intensity of the cells was detected using a fluorescence
microscope.
Flow cytometry
All the groups of cells were washed with PBS for
three times and then double-stained with annexin V-FITC and
propidium iodide at 37°C for 20 min. The apoptosis of each sample
group was analyzed using a fluorescence-activated cell sorter
(FACS; CytoFLEX; Beckman Coulter, Inc.). Data analysis was
conducted using FlowJo software (version 10.8.1; Becton, Dickinson
& Company, Oregon, USA). Apoptotic rates were calculated as the
sum of the percentage of early and late apoptotic cells.
Co-immunoprecipitation assays
Co-immunoprecipitation assays were conducted
according to the manufacturer's instructions (cat. no. 88804,
Thermo Fisher Scientific, Inc.) The HaCaT cells were washed with
PBS and lysed with lysis buffer (0.025M pH=7.4 Tris, 0.15M NaCl,
0.001M EDTA, 1% NP40 and 5% glycerol) supplemented with a protease
inhibitor cocktail (100: 1 (v/v)). After being centrifuged at
13,680 × g for 15 min at 4°C, the supernatant was collected, and 20
μl of the collected supernatant was used as input. The
remaining cell lysate supernatant (0.5 mg per reaction) was
incubated with 2 μg of IL-37 antibody (cat. no. ab278499;
Abcam, IgG antibody was used as control) at 4°C overnight. The
immunocomplex was captured using 10 μl of protein A+G beads
at 37°C for 1 h. After incubation, the complex was separated on a
magnetic rack for 10 sec, and then washed thrice using wash buffer
(0.025 M pH=7.4 Tris, 0.15 M NaCl, 0.001 M EDTA, 1% NP40 and 5%
glycerol) supplemented with a protease inhibitor cocktail. Western
blotting was used for further analysis.
Statistical analysis
The data were analyzed using SPSS 18.0 (SPSS, Inc.)
and GraphPad Prism 6 (Dotmatics) and expressed as mean ± SD.
Differences between two groups were estimated by independent sample
t test. Differences among multiple groups were estimated using
one-way ANOVA with Bonferroni's post-hoc test. The correlations
between serum IL-37 and IL-10, IL-6, and IL-17, respectively, were
assessed using Pearson's correlation analysis. All experiments were
conducted with three independent biological replicates. A post hoc
power analysis was conducted using G*Power 3.1.9.2
(Heinrich Heine University Düsseldorf, Düsseldorf, Germany) to
evaluate the sufficiency of the clinical sample size. Given α=0.05,
n=15 per group, and IL-37 concentrations of 4.20±0.98 pg/ml
(healthy control) vs. 10.52±2.87 pg/ml (PV), the achieved power was
1.00, which exceeds the conventional threshold of 0.9. P<0.05
was considered to indicate a statistically significant
difference.
Results
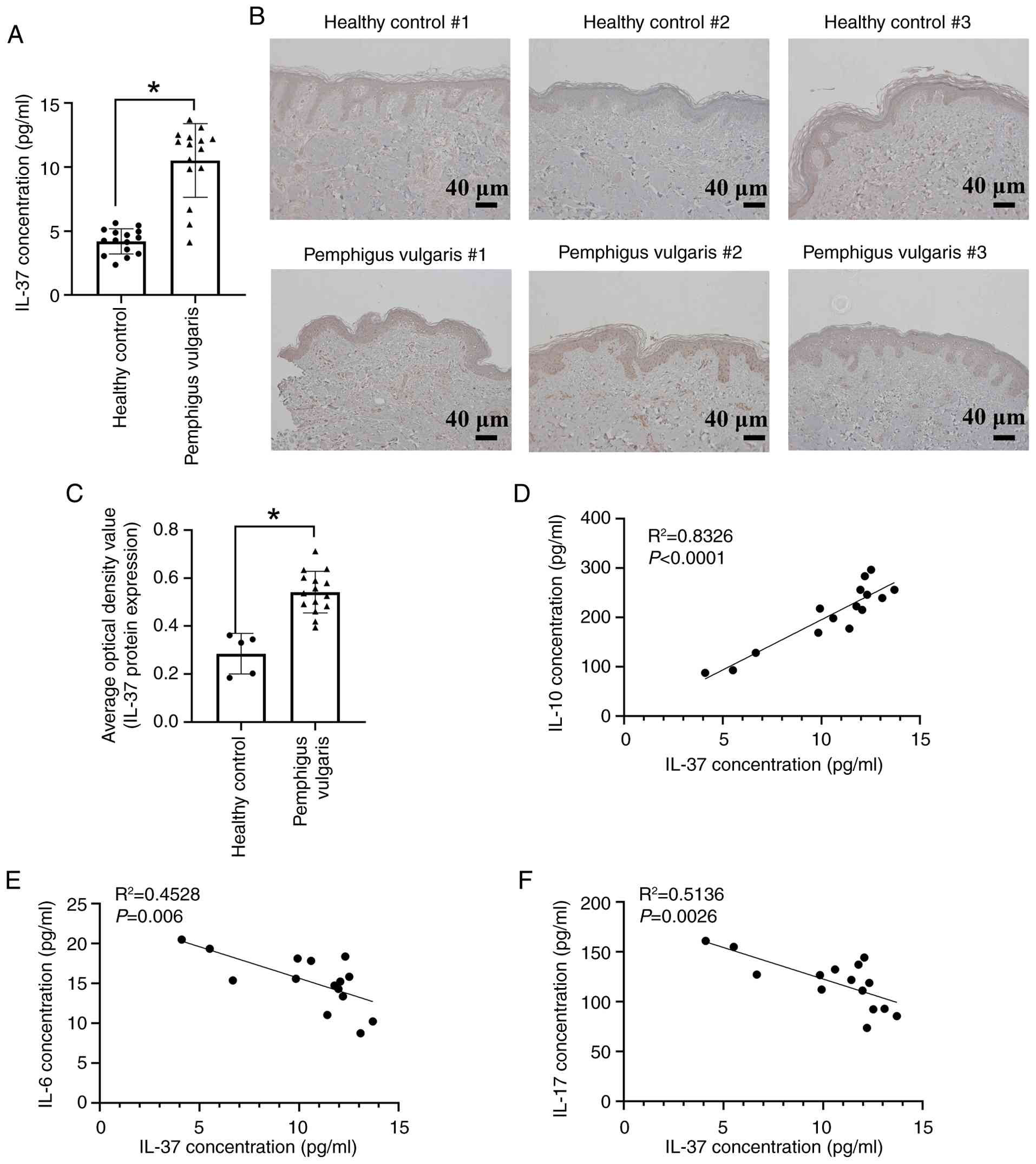
IL-37 is augmented in PV and associated
with inflammatory factors
To elucidate the pathophysiological role of IL-37 in
PV, the present study analyzed the concentration of IL-37 in the
serum and skin tissues from healthy controls and. Serum IL-37
levels were markedly higher in patients with PV compared with
healthy controls (Fig. 1A).
Similarly, IL-37 expression was markedly elevated in the skin
tissues of patients with PV than in healthy controls, as indicated
by immunohistochemistry assays (Fig.
1B and C). Furthermore, serum IL-37 levels showed a positive
correlation with the anti-inflammatory factor IL-10 (Fig. 1D), and a negative correlation
with the pro-inflammatory cytokines IL-6 and IL-17 (Fig. 1E and F).
IL-37 protects HaCaT cells from
Dsg3-induced keratinocyte dissociation and apoptosis
To investigate the functional role of IL-37 in PV,
an in vitro PV model was established by treating HaCaT
keratinocytes with the anti-Dsg3 antibody AK23. Immunofluorescence
staining revealed that under control or with a low anti-Dsg3
antibody (1:2,000 dilution) treated conditions, Dsg3 exhibited a
linear, continuous staining along the cell borders. By contrast, a
higher anti-Dsg3 antibody concentration (1:1,000 dilution) resulted
in markedly reduced Dsg3 staining, indicating impaired cell
adhesion and successful modeling of PV-like acantholysis (Fig. 2A). HaCaT cells were treated with
different concentration of IL-37 recombinant protein (0, 25, 50,
100 and 200 ng/ml). As shown in Fig.
2B, IL-37 at concentrations of 100 ng/ml and below had no
effect on cell viability, whereas 200 ng/ml IL-37 markedly reduced
cell viability. Therefore, 100 ng/ml IL-37 was selected for
subsequent experiments. The anti-Dsg3 antibody treatment markedly
increased the IL-37 levels (Fig. 2B
and C). Co-treatment with 100 ng/ml of IL-37 and anti-Dsg3
antibody further enhanced IL-37 expression compared with anti-Dsg3
antibody alone (Fig. 2C and D).
Additionally, the concentration and mRNA expression of IL-10 were
markedly increased (Fig. 2E and
F), while the concentration and mRNA expression of IL-6
(Fig. 2G and H) and IL-17
(Fig. 2I and J) were markedly
decreased in IL-37 in combination with the anti-Dsg3 treated HaCaT
group.
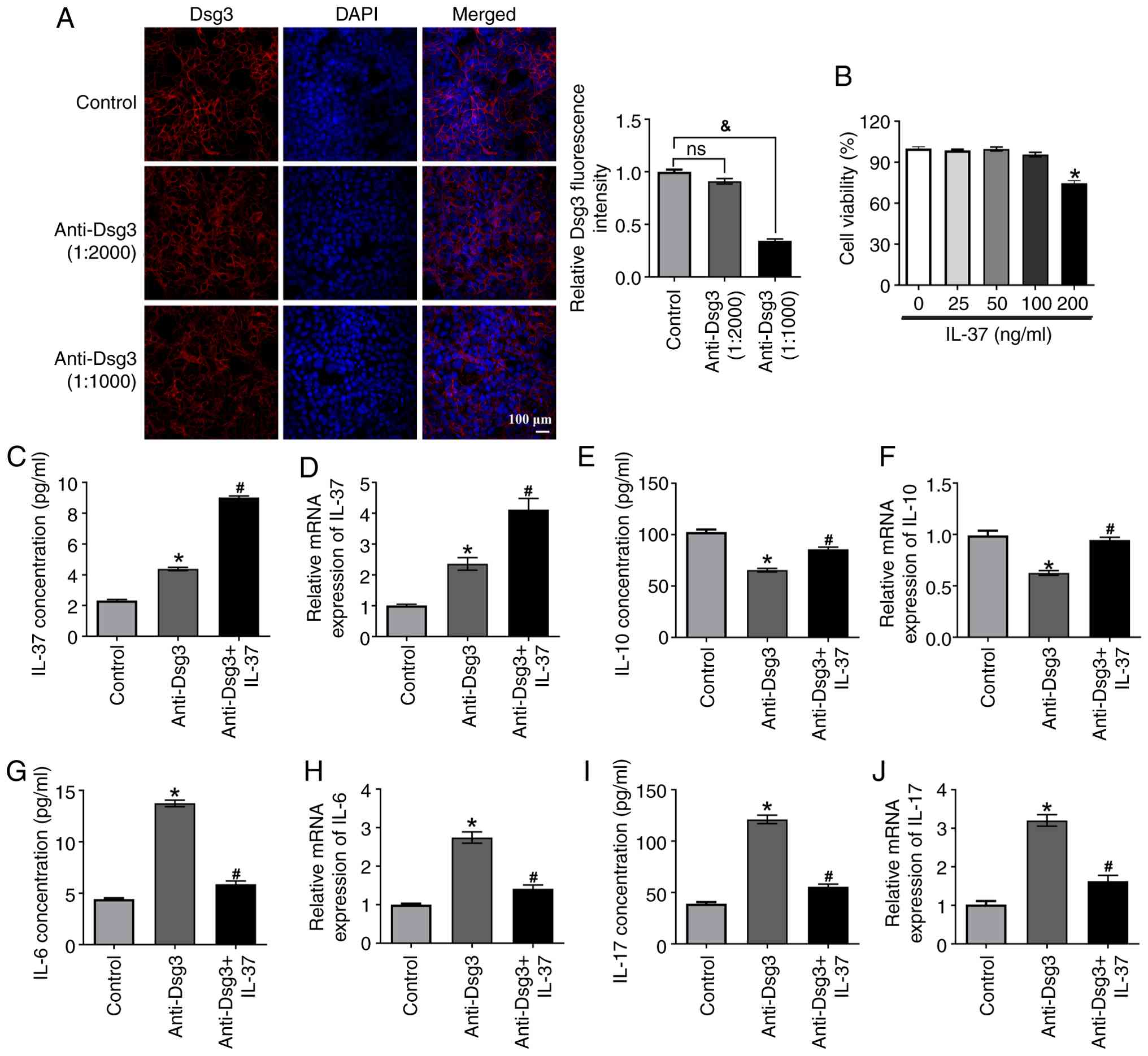 | Figure 2IL-37 protects HaCaT cells from
Dsg3-induced keratinocyte dissociation and apoptosis. (A) The
effect of different dilutions of Dsg-3 antibodies on cell
dissociation was analyzed by immunofluorescence. (B) HaCaT cells
were treated with different concentration of IL-37 recombinant
protein (0, 25, 50, 100, and 200 ng/ml). Cell viability was
detected by CCK-8 assay. (C) HaCaT cells were treated with the
Dsg-3 antibodies, followed by administration of the IL-37
recombinant protein. The concentration of IL-37 was analyzed using
an ELISA assay. (D) The relative mRNA expression of IL-37 was
detected using RT-qPCR. The (E) concentration and (F) relative mRNA
expression of IL-10 were measured using an IL-10 ELISA kit and
RT-qPCR, respectively. The (G) oncentration and (H) relative mRNA
expression of IL-6 were measured by the IL-6 ELISA kit and RT-qPCR,
respectively. The (I) concentration and (J) relative mRNA
expression of IL-17 were measured by the IL-17 ELISA kit and
RT-qPCR, respectively. &P<0.05;
*P<0.05 vs. Control group; #P<0.05 vs.
anti-Dsg3 group. Data are presented as mean ± SD, n=3 biological
independent replicates. One-way ANOVA with Bonferroni's post-hoc
test was used for multiple group comparisons. IL, interleukin;
Dsg3, desmoglein-3; ELISA, enzyme-linked immunosorbent assay;
RT-qPCR, reverse transcription-quantitative PCR. |
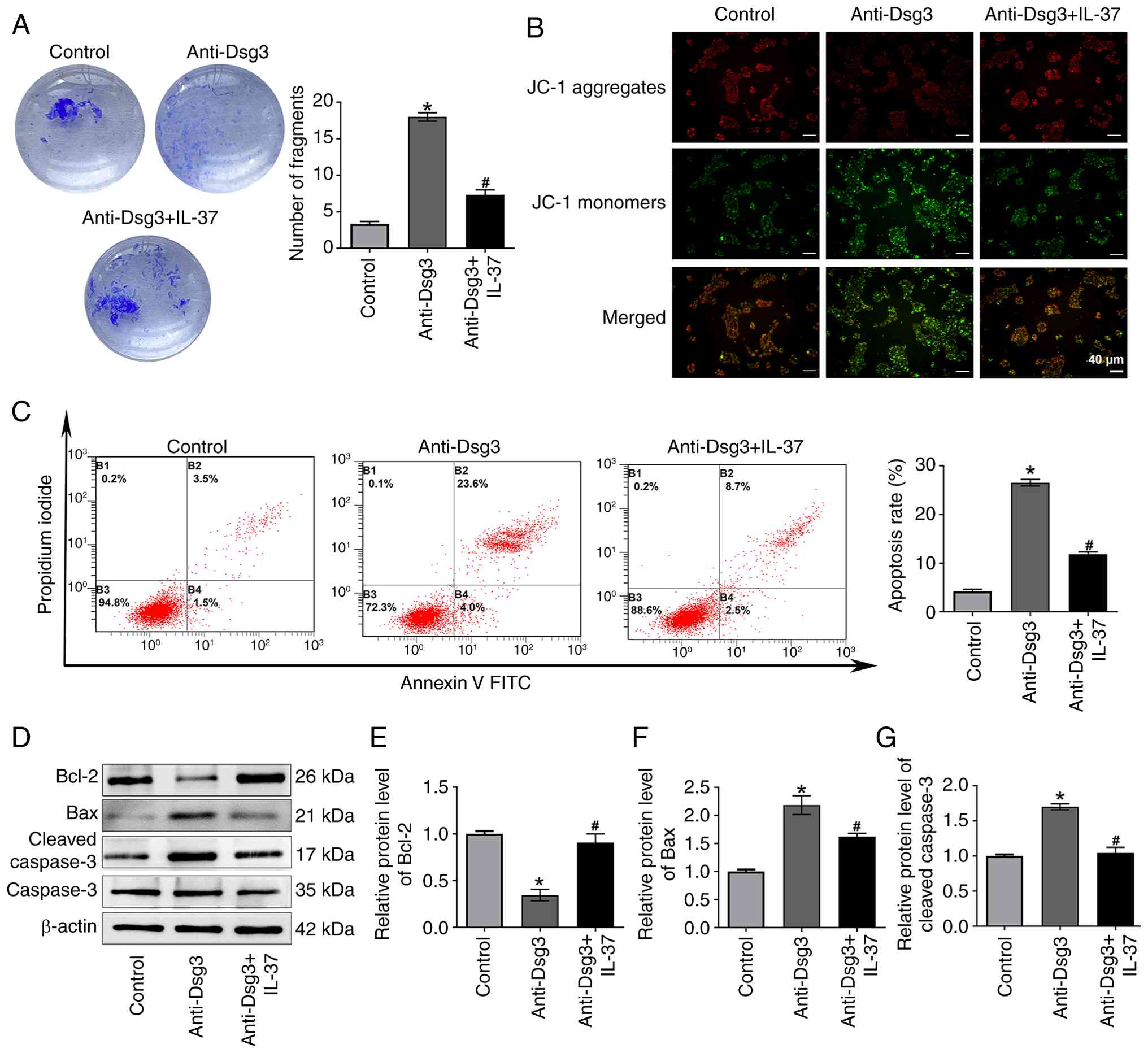
The dispase-based dissociation assays demonstrated a
significant increase in the number of fragments in the anti-Dsg3
group compared with the control group (Fig. 3A). By contrast, IL-37 treatment
reduced the number of fragments compared with the anti-Dsg3 group
(Fig. 3A). Given that PV
pathogenesis may be initiated by pro-apoptotic proteins and that
the apoptotic pathway are activated prior to morphological signs of
acantholysis (21,22), the present study further analyzed
the apoptosis of the HaCaT cells. JC-1 assay revealed enhanced
green fluorescence and reduced red fluorescence in the anti-Dsg3
group compared with the control group, indicating a transition from
red to green fluorescence, characteristic of early apoptosis in the
anti-Dsg3 group (Fig. 3B).
However, IL-37 decreased the intensity of green fluorescence and
increased the red fluorescence intensity, suggesting that IL-37
treatment increased mitochondrial membrane potential and inhibited
cell apoptosis (Fig. 3B).
Consistent with these findings, the flow cytometry analysis
demonstrated that the apoptosis of HaCaT cells was increased in the
anti-Dsg3 group compared with the control group, whereas the
apoptosis of HaCaT cells in the anti-Dsg3+IL-37 group was decreased
compared with that in the anti-Dsg3 group (Fig. 3C). Further analysis of
apoptosis-related markers showed that anti-Dsg3 antibody exposure
markedly decreased Bcl-2 expression and increased expression of Bax
and cleaved-caspase 3 compared with the control group (Fig. 3D-G). IL-37 treatment reversed
these changes, upregulating Bcl-2 and downregulating Bax and
cleaved-caspase 3 expression (Fig.
3D-G).
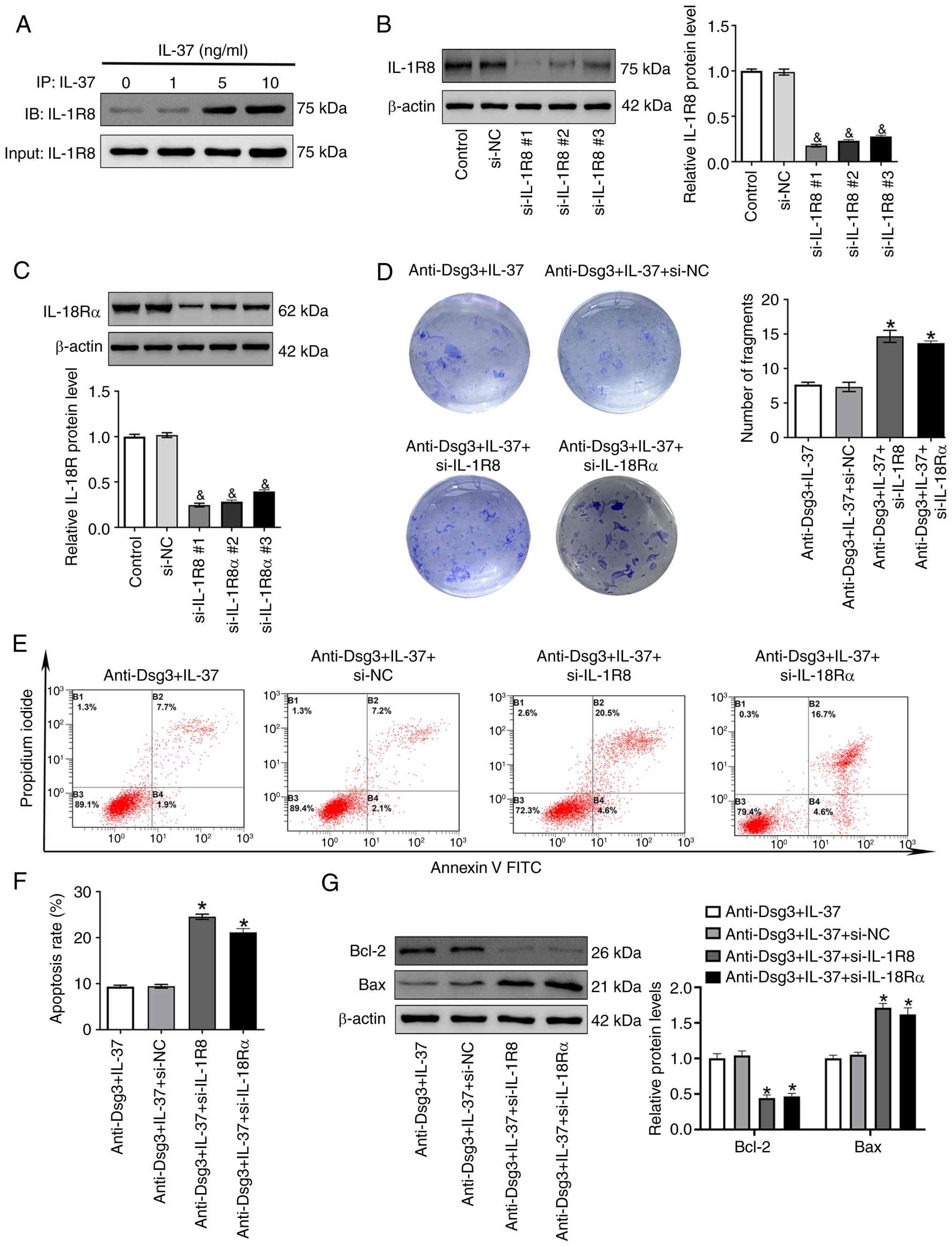
IL-37 protects HaCaT cells from
keratinocyte dissociation and apoptosis through IL-1R8
As a ligand protein, IL-37 functions by binding to
its receptor IL-1R8 (23). The
co-immunoprecipitation assay confirmed the interaction of IL-37 and
IL-1R8 in the HaCaT cells (Fig.
4A). To further investigate the functional role of this
interaction, IL-1R8 and IL-18Rα were knocked down in the HaCaT
cells through transfection with IL-1R8-specific siRNA and
IL-18Rα-specific siRNA, respectively. IL-1R8 and IL-18Rα expression
were markedly decreased in IL-1R8 siRNA (Fig. 4B) or IL-18Rα siRNA (Fig. 4C) transfected HaCaT cells.
Knockdown of either IL-1R8 or IL-18Rα resulted in an increased
number of cellular fragments compared with the negative control
siRNA transfected HaCaT cells (Fig.
4D). Additionally, the apoptosis rate was markedly higher in
the anti-Dsg3 + IL-37 + si-IL-1R8 group and anti-Dsg3 + IL-37 +
si-IL-18Rα group than in the anti-Dsg3 + IL-37 + si-NC group
(Fig. 4E and F). Consistent with
these findings, the Bax expression was upregulated, while the Bcl-2
expression was downregulated in the anti-Dsg3 + IL-37 + si-IL-1R8
group and anti-Dsg3 + IL-37 + si-IL-18Rα group relative to the
anti-Dsg3 + IL-37 + si-NC group (Fig. 4G).
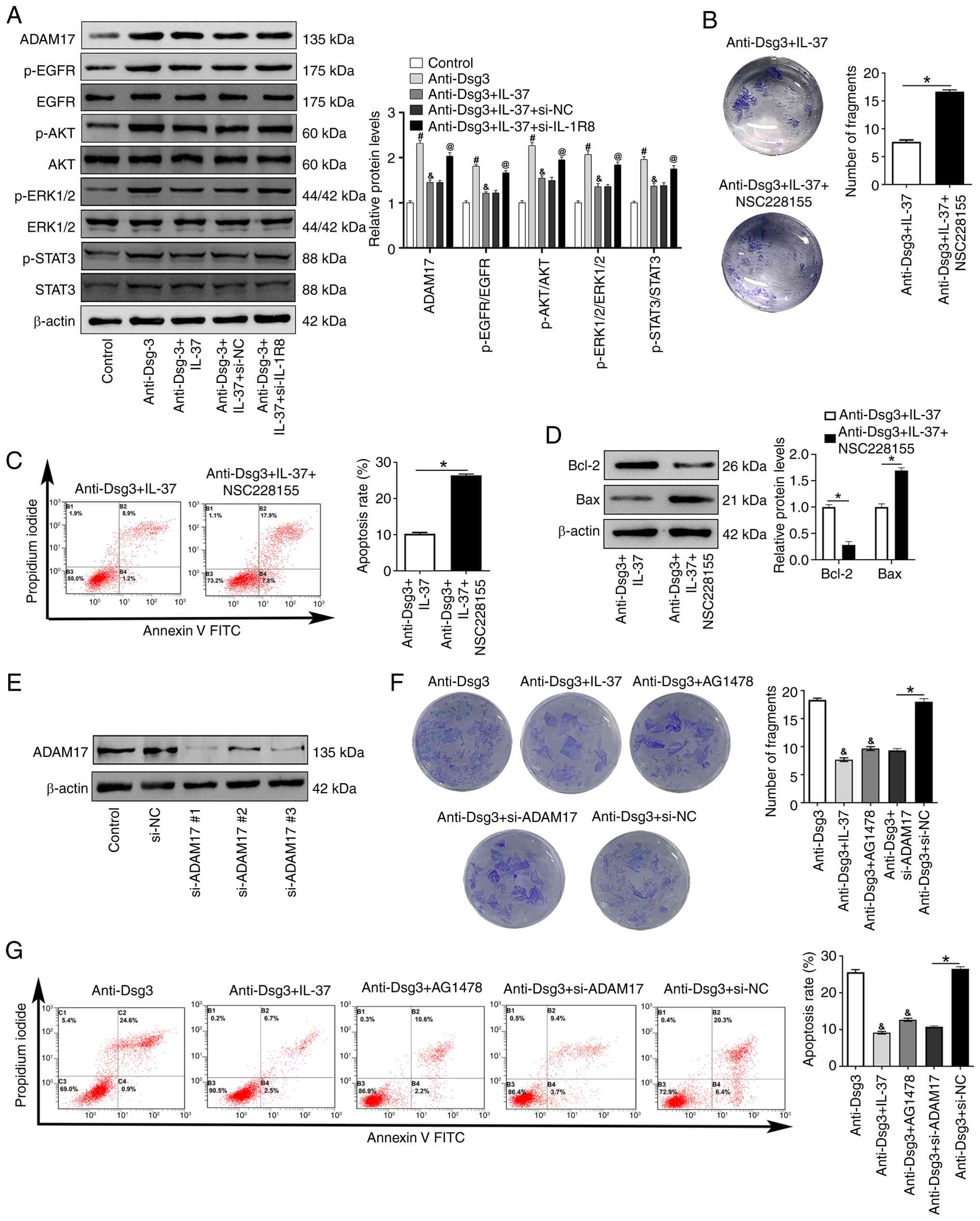
IL-37/IL-1R8 protects HaCaT cells from
keratinocyte dissociation and apoptosis through the ADAM17/EGFR
pathway
Following PV-IgG treatment, EGFR
auto-phosphorylation, internalization and acantholysis were induced
(24). It has been reported that
IL-1R deficiency upregulates the expression of ADAM-17, a
metalloproteinase responsible for the shedding and release of EGFR
ligands (25). To further
explore the potential mechanisms by which IL-37 administration
induces keratinocyte dissociation and apoptosis, the present study
examined the expression of ADAM17 and EGFR phosphorylation.
Compared with the anti-Dsg3 group, ADAM17 expression, p-EGFR as
well as EGFR downstream protein p-AKT, p-ERK1/2, and p-STAT3 levels
were markedly decreased in the anti-Dsg-3 + IL-37 group, indicating
a suppression of canonical EGFR signaling cascades (Fig. 5A). Furthermore, knockdown of
IL-1R8 markedly increased the expression of ADAM17, p-EGFR,
p-ERK1/2, p-AKT, and p-STAT3 levels in the anti-Dsg3 + IL-37 +
si-IL-1R8 group when compared with that in the anti-Dsg3 + IL-37 +
si-NC group (Fig. 5A).
Pharmacologically activation of EGFR using the EGFR
activator NSC228155 enhanced epidermal fragment formation and
apoptosis rates (Fig. 5B and C).
Additionally, Bcl-2 expression was markedly decreased, whereas Bax
protein expression was markedly increased in the anti-Dsg3 + IL-37
+ NSC228155 group compared with the anti-Dsg3 + IL-37 group
(Fig. 5D).
Subsequently, HaCaT was transfected with ADAM17
siRNA and western blotting showed a downregulation of ADAM17 after
ADAM17 siRNA transfection, with ADAM17 siRNA #1 demonstrating the
most pronounced reduction in protein level (Fig. 5E). This ADAM17 knockdown markedly
decreased both epidermal fragment formation (Fig. 5F) and apoptosis rates (Fig. 5G) compared with anti-Dsg3+si-NC
group. Notably, compared with the anti-Dsg3 group, EGFR inhibitor
AG1478 also decreased epidermal fragment formation and apoptosis
rates (Fig. 5F and G).
Discussion
The present study demonstrated that IL-37 is highly
expressed in the serum and skin tissue of patients with PV. IL-37
regulates HaCaT cell dissociation and apoptosis in an
IL-1R8-dependent manner. Furthermore, the IL-37/IL-1R8 axis
inhibits HaCaT cell dissociation and apoptosis by negatively
regulating the ADAM17/EGFR pathway.
It is well established that desmoglein-1 (Dsg1) and
Dsg3, calcium-dependent cell adhesion molecules, serve as the
primary autoantibodies targets in the pathogenesis of PV (26). Several non-Dsg antigens,
including desmocollin 1 and 3, mitochondrial proteins, human
leukocyte antigen molecules and various subtypes of nicotinic and
muscarinic acetylcholine receptor, have also been implicated in PV
(27), indicating a
multifactorial etiology. In a previous study, we found that IL-37
inhibited keratinocyte dissociation and Dsg3 endocytosis by
upregulating caveolin-1 and inhibiting the STAT3 pathway (8). The present study discovered that
IL-37 recombination protein led to a loss of cell dissociation and
a decrease in cell apoptosis. As a ligand protein, IL-37 functions
by binding to its receptors, IL-1R8 and IL-18R. Previous reports
indicate that IL-37b exerts anti-inflammatory effects through
IL-1R8 by inhibiting the p38, ERK, JNK, and NF-κB pathways, and
that IL-1R8 knockdown impairs the anti-inflammatory activity of
IL-37 (28,29). Using a co-immunoprecipitation
assay, the present study confirmed the interaction between IL-37
and IL-1R8. Knockdown of IL-1R8 reversed the protective effect of
IL-37 on cell dissociation and apoptosis, suggesting that IL-37
acts through an IL-1R8-dependent mechanism.
Following PV IgG treatment, EGFR
autophosphorylation, internalization and acantholysis are induced
(24). In keratinocytes,
inhibition of EGFR blocked PV IgG-triggered Dsg3 endocytosis,
keratinocyte dissociation and blister formation, indicating
cross-talk between EGFR and Dsg3 (24). Various factors, including
G-protein-coupled receptor agonists and cytokines (such as IL-1β),
can transactivate EGFR. This transactivation requires ADAM-17, a
metalloproteinase that mediates the shedding and release of EGFR
ligands (30). Previous studies
have demonstrated that ADAM family members are associated with cell
adhesion (31) and,
specifically, ADAM10 and ADAM17 contribute to Dsg2 turnover
(32). Additionally, ADAM17 can
be activated by interleukin-15 (33). Notably, Wang et al
(25) reported that IL-1R
deficiency increased ADAM17 expression levels in a rat model of
Alzheimer's disease. The present study found that IL-37 markedly
decreased both ADAM17 expression and EGFR phosphorylation.
Furthermore, activation of EGFR reversed the effects of IL-37 on
HaCaT cell dissociation and apoptosis. It is well established that
patients with PV exhibit a distinct autoantibody profile, leading
to variations in intracellular signaling patterns (27,34). For instance, patients with PV and
with anti-Dsg3 and anti-Dsg1 antibodies, but not anti-desmocollin3
antibodies, show increased levels of EGFR ligands such as EGF.
These patients can benefit from treatment with EGF or EGFR
inhibitors (34). Thus, patients
with PV and with DSG1/3 antibodies might respond favorably to IL-37
therapy. Importantly, the function of IL-37 is
concentration-dependent; at high concentrations, it forms dimers
that inactivate it, thus inhibiting its anti-inflammatory function
(35). This crucial mechanism
underscores that its function is not linear. In the present study,
the observed IL-37 levels appeared to fall within a therapeutic
window where its net effect remained protective. Future studies are
warranted to delineate the exact concentration threshold for
dimerization in PV and to explore IL-37 as a potential therapeutic
agent, where strategies to stabilize its active, monomeric form
could be highly beneficial. Therefore, the concentration of IL-37
must be carefully considered in clinical applications aiming to
inhibit EGFR in PV.
The present study used an anti-Dsg3 antibody (AK23)
induced HaCaT cell model. However, this model does not fully
recapitulate the complexity of PV, which involves multiple
autoantibodies and patient heterogeneity. Validating key findings
in a murine model of PV or patient-derived samples is needed in
further studies.
In conclusion, the present study demonstrated that
IL-37 inhibited ADAM17-mediated EGFR activation via its receptor
IL-1R8, thereby inhibiting keratinocyte acantholysis and apoptosis.
These findings provide new perspective on the treatment of
pemphigus, suggesting that therapeutic strategies may extend beyond
small-molecule EGFR inhibitors to include targeting the
IL-37/IL-1R8 pathway.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Author' contributions
FH was responsible for formal analysis, methodology,
supervision, original draft preparation, writing, reviewing and
editing. WC was responsible for formal analysis, supervision,
original draft preparation, writing, reviewing and editing. QW was
responsible for formal analysis and methodology. XZ was responsible
for formal analysis and methodology. FX was responsible for formal
analysis and methodology. JZ was responsible for conceptualization,
supervision, writing, reviewing and editing. JL was responsible for
conceptualization, supervision, writing, reviewing and editing.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the People's Hospital of Xinjiang Uygur Autonomous
Region (approval no. 2019030616). The procedures followed were in
accordance with the Helsinki Declaration of 1975, as revised in
1983. Serum and skin tissues were collected from 15 patients with
PV who were undergoing treatment in the Department of Dermatology
in People's Hospital of Xinjiang Uygur Autonomous Region. Serum (15
samples) and skin tissue (5 samples) from healthy control were
collected from participants who were undergoing skin plastic
surgery in plastic surgery in the same hospital.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
The present study was supported by National Natural Science
Foundation of China (grant no. 82260621), Natural Sciences of
Xinjiang Uygur Autonomous Region (grant no. 2022D01C623) and Basic
and Clinical Research Project of Stem Cell Biotherapy-People's
Hospital of Xinjiang Uygur Autonomous Region (grant no.
GXBZX-2023004).
References
|
1
|
Vafaeian A, Mahmoudi H and Daneshpazhooh
M: What is novel in the clinical management of pemphigus vulgaris?
Expert Rev Clin Pharmacol. 17:489–503. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Schmidt E, Kasperkiewicz M and Joly P:
Pemphigus. Lancet. 394:882–894. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rosi-Schumacher M, Baker J, Waris J,
Seiffert-Sinha K and Sinha AA: Worldwide epidemiologic factors in
pemphigus vulgaris and bullous pemphigoid. Front Immunol.
14:11593512023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhao W, Wang J, Zhu H and Pan M:
Comparison of guidelines for management of pemphigus: A review of
systemic corticosteroids, rituximab, and other immunosuppressive
therapies. Clin Rev Allergy Immunol. 61:351–362. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Brescacin A, Baig Z, Bhinder J, Lin S,
Brar L and Cirillo N: What protein kinases are crucial for
acantholysis and blister formation in pemphigus vulgaris? A
systematic review. J Cell Physiol. 237:2825–2837. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fukaura R and Akiyama M: Targeting IL-36
in inflammatory skin diseases. BioDrugs. 37:279–293. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mesas-Fernández A, Bodner E, Hilke FJ,
Meier K, Ghoreschi K and Solimani F: Interleukin-21 in autoimmune
and inflammatory skin diseases. Eur J Immunol. 53:e22500752023.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liang J, Hu F, Mao L, Qiu Y, Jiang F, Wang
Q, Abulikemu K, Hong Y, Ge X and Kang X: Interleukin-37 inhibits
desmoglein-3 endocytosis and keratinocyte dissociation via
upregulation of Caveolin-1 and inhibition of the STAT3 pathway. J
Eur Acad Dermatol Venereol. 37:1920–1927. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhou J, Gemperline DC, Turner MJ, Oldach
J, Molignano J, Sims JT and Stayrook KR: Transcriptomic analysis of
healthy and atopic dermatitis samples reveals the role of IL-37 in
human skin. Immunohorizons. 5:830–843. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rønholt K, Nielsen AL, Johansen C,
Vestergaard C, Fauerbye A, López-Vales R, Dinarello CA and Iversen
L: IL-37 expression is downregulated in lesional psoriasis skin.
Immunohorizons. 4:754–761. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hou T, Tsang MS, Chu IM, Kan LL, Hon KL,
Leung TF, Lam CW and Wong CK: Skewed inflammation is associated
with aberrant interleukin-37 signaling pathway in atopic
dermatitis. Allergy. 76:2102–2114. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fujita H, Inoue Y, Seto K, Komitsu N and
Aihara M: Interleukin-37 is elevated in subjects with atopic
dermatitis. J Dermatol Sci. 69:173–175. 2013. View Article : Google Scholar
|
|
13
|
Gu M, Jin Y, Gao X, Xia W, Xu T and Pan S:
Novel insights into IL-37: An anti-inflammatory cytokine with
emerging roles in anti-cancer process. Front Immunol.
14:12785212023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Borgia F, Custurone P, Li Pomi F, Vaccaro
M, Alessandrello C and Gangemi S: IL-33 and IL-37: A possible axis
in skin and allergic diseases. Int J Mol Sci. 24:3722022.
View Article : Google Scholar
|
|
15
|
Cavalli G and Dinarello CA: Suppression of
inflammation and acquired immunity by IL-37. Immunol Rev.
281:179–190. 2018. View Article : Google Scholar
|
|
16
|
Schröder A, Lunding LP, Zissler UM, Vock
C, Webering S, Ehlers JC, Orinska Z, Chaker A, Schmidt-Weber CB,
Lang NJ, et al: IL-37 regulates allergic inflammation by
counterbalancing pro-inflammatory IL-1 and IL-33. Allergy.
77:856–869. 2022. View Article : Google Scholar
|
|
17
|
Pan Y, Wen X, Hao D, Wang Y, Wang L, He G
and Jiang X: The role of IL-37 in skin and connective tissue
diseases. Biomed Pharmacother. 122:1097052020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Johnston A, Gudjonsson JE, Aphale A,
Guzman AM, Stoll SW and Elder JT: EGFR and IL-1 signaling
synergistically promote keratinocyte antimicrobial defenses in a
differentiation-dependent manner. J Invest Dermatol. 131:329–337.
2011. View Article : Google Scholar :
|
|
19
|
Sayar BS, Rüegg S, Schmidt E, Sibilia M,
Siffert M, Suter MM, Galichet A and Müller EJ: EGFR inhibitors
erlotinib and lapatinib ameliorate epidermal blistering in
pemphigus vulgaris in a non-linear, V-shaped relationship. Exp
Dermatol. 23:33–38. 2014. View Article : Google Scholar
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
21
|
Ramani P, Ravikumar R, Pandiar D, Monica
K, Krishnan RP, Ramasubramanian A and Sukumaran G: Apoptolysis: A
less understood concept in the pathogenesis of Pemphigus Vulgaris.
Apoptosis. 27:322–328. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hutchison DM, Hosking AM, Hong EM and
Grando SA: Mitochondrial autoantibodies and the role of apoptosis
in pemphigus vulgaris. Antibodies (Basel). 11:552022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sánchez-Fernández A, Zandee S,
Amo-Aparicio J, Charabati M, Prat A, Garlanda C, Eisenmesser EZ,
Dinarello CA and López-Vales R: IL-37 exerts therapeutic effects in
experimental autoimmune encephalomyelitis through the receptor
complex IL-1R5/IL-1R8. Theranostics. 11:1–13. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bektas M, Jolly PS, Berkowitz P, Amagai M
and Rubenstein DS: A pathophysiologic role for epidermal growth
factor receptor in pemphigus acantholysis. J Biol Chem.
288:9447–9456. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang H, Peng G, Wang B, Yin H, Fang X, He
F, Zhao D, Liu Q and Shi L: IL-1R(-/-) alleviates cognitive
deficits through microglial M2 polarization in AD mice. Brain Res
Bull. 157:10–17. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schmitt T and Waschke J:
Autoantibody-specific signalling in pemphigus. Front Med
(Lausanne). 8:7018092021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Amber KT, Valdebran M and Grando SA:
Non-desmoglein antibodies in patients with pemphigus vulgaris.
Front Immunol. 9:11902018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Luo P, Feng C, Jiang C, Ren X, Gou L, Ji P
and Xu J: IL-37b alleviates inflammation in the temporomandibular
joint cartilage via IL-1R8 pathway. Cell Prolif. 52:e126922019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jia C, Zhuge Y, Zhang S, Ni C, Wang L, Wu
R, Niu C, Wen Z, Rong X, Qiu H and Chu M: IL-37b alleviates
endothelial cell apoptosis and inflammation in Kawasaki disease
through IL-1R8 pathway. Cell Death Dis. 12:5752021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Schumacher N and Rose-John S: ADAM17
orchestrates Interleukin-6, TNFα and EGF-R signaling in
inflammation and cancer. Biochim Biophys Acta Mol Cell Res.
1869:1191412022. View Article : Google Scholar
|
|
31
|
Cirillo N and Prime SS: A scoping review
of the role of metalloproteinases in the pathogenesis of autoimmune
pemphigus and pemphigoid. Biomolecules. 11:15062021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Klessner JL, Desai BV, Amargo EV, Getsios
S and Green KJ: EGFR and ADAMs cooperate to regulate shedding and
endocytic trafficking of the desmosomal cadherin desmoglein 2. Mol
Biol Cell. 20:328–337. 2009. View Article : Google Scholar :
|
|
33
|
Mishra HK, Dixon KJ, Pore N, Felices M,
Miller JS and Walcheck B: Activation of ADAM17 by IL-15 limits
human NK Cell proliferation. Front Immunol. 12:7116212021.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ivars M, España A, Alzuguren P, Pelacho B,
Lasarte JJ and López-Zabalza MJ: The involvement of ADAM10 in
acantholysis in mucocutaneous pemphigus vulgaris depends on the
autoantibody profile of each patient. Br J Dermatol. 182:1194–1204.
2020. View Article : Google Scholar
|
|
35
|
Mei Y and Liu H: IL-37: An
anti-inflammatory cytokine with antitumor functions. Cancer Rep
(Hoboken). 2:e11512019.PubMed/NCBI
|