Obesity is generally defined as excessive body fat
that impairs health. The primary measure of obesity is the ratio of
weight to height squared (kg/m2), known as body mass
index (BMI) (1). The body weight
classification criteria issued by the World Health Organization
define a BMI >30 kg/m2 as obesity and a BMI >40
kg/m2 as severe obesity, wherein both being overweight
or obese are associated with increased mortality (2,3).
A number of studies have confirmed that obesity can trigger chronic
low-grade inflammation and metabolic dysfunction in multiple organs
(such as adipose tissue, skeletal muscle and the liver),
significantly increasing the risk of type 2 diabetes (T2DM) and
metabolic dysfunction-associated steatotic liver disease (MASLD)
(4-6). Although research has demonstrated
the participation of immune cells, hepatocytes and myocytes as well
as inflammatory factors, adipokines and epigenetic regulation in
obesity-related metabolic disorders, the precise molecular
mechanisms and regulatory networks involved remain to be fully
elucidated (7-11).
Adipose tissue is a crucial metabolic and endocrine
organ whose functions extend far beyond simple energy storage.
Owing to differences in structure and function, adipose tissue can
be divided into white adipose tissue (WAT) and brown adipose tissue
(BAT). WAT is composed mainly of white adipocytes, which store
excess energy in the form of triglycerides (TGs) (12). BAT is composed mainly of brown
adipocytes, which are rich in mitochondria and uncoupling protein
(UCP)1 and can actively generate heat (13). Additionally, beige adipose tissue
and pink adipose tissue exist. Beige adipose tissue originates
through the transformation of WAT, a process termed 'adipose
browning'. Like brown fat, beige adipocytes exhibit high
mitochondrial density and express thermogenesis-related genes,
enabling energy dissipation by non-shivering thermogenesis
(14). Moreover, pink adipose
tissue has the ability to synthesize and secrete milk (15). Adipose tissue functions as an
active endocrine organ and regulates the metabolic-inflammatory
balance through the secretion of adipokines (such as leptin and
adiponectin) and inflammatory cytokines [such as IL-8, tumour
necrosis factor-α (TNF-α) and monocyte chemoattractant protein-1
(MCP-1). These bioactive molecules can be autonomously secreted by
adipocytes or produced by infiltrating immune cells within the
tissue (16). However, in the
context of obesity, adipocyte hypertrophy occurs, accompanied by
impaired tissue angiogenesis and the extensive infiltration of
immune cells (such as T cells and macrophages) into adipose tissue;
these cells exhibit polarization towards proinflammatory phenotypes
and increased secretion of proinflammatory cytokines (17,18). Moreover, enlarged fat cells also
exhibit insulin resistance and increased production of
proinflammatory cytokines and adipokines (19,20). These phenomena affect the limited
healthy expansion of adipose tissue and increase various adverse
metabolic consequences. Thus, the induction of adipose tissue
establishes a long-term low-grade inflammatory state and has been
shown to have a significant effect on obesity-induced metabolic
diseases such as T2DM, MASLD and atherosclerosis (21-24).
Obesity is the ultimate result of an imbalance
between energy intake and energy expenditure. Excess lipids
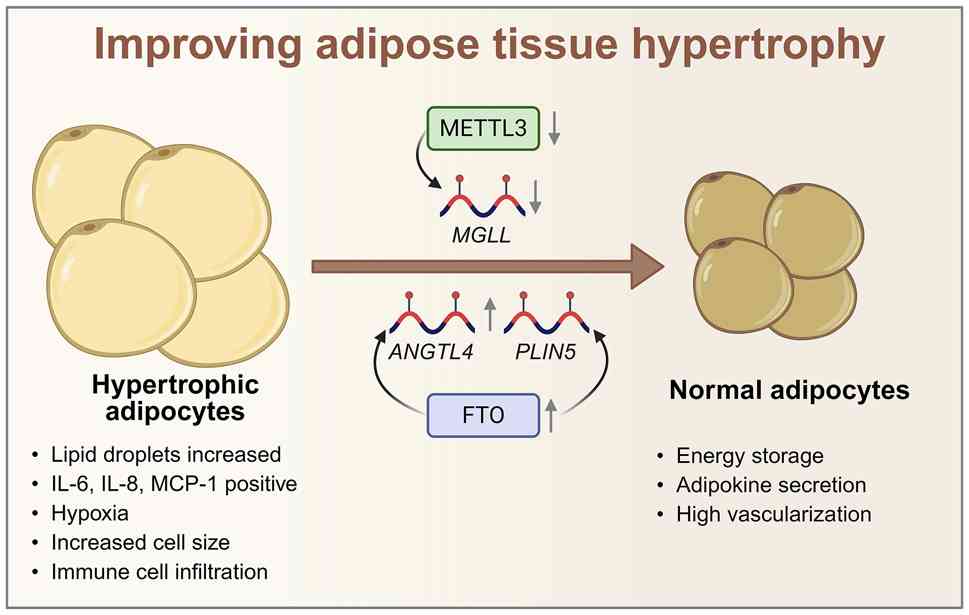
accumulate mainly in adipose tissue, leading to increased adipocyte
size, which is known as hypertrophy. This is an important
pathological mechanism of adipocyte expansion and occurs mainly in
post-development (adult) WAT (43). Compared with smaller adipocytes,
larger hypertrophic adipocytes exhibit increased secretion of
interleukin (IL)-6, IL-8, MCP-1 and other inflammatory cytokines
and decreased secretion of IL-10, an anti-inflammatory cytokine
(19). In addition, a study has
shown that in individuals with obesity, the capillaries between
hypertrophic fat cells become thinner (44). Limited by the sparse organization
of and mechanical stress on the capillaries, fat tissue does not
expand as fat cells hypertrophy. Therefore, hypertrophic fat cells
experience continuous hypoxia, some of which die, which causes
macrophages to accumulate in the adipose tissue, especially near
dead fat cells, thereby further increasing adipose tissue
inflammation (45). If the
hypertrophy of fat cells can be inhibited or the production of
hypertrophic fat cells can be reduced, a direct therapeutic effect
on adipose tissue inflammation can be achieved.
Adipogenesis is a complex process in which stem
cells differentiate into adipocytes and form adipose tissue. The
whole process consists of two steps: The commitment stage and the
terminal differentiation stage (56). Lipogenesis occurs mainly during
body development (that is, in adolescents and children), and the
number of fat cells in adults is strictly regulated, even in
individuals with obesity (57).
However, a study has demonstrated that, during adipose tissue
expansion in obese adult animals, adipose tissue dysplasia caused
by adipocyte proliferation can also be alleviated by promoting
de novo adipocyte differentiation to ensure safe energy
storage in WAT (58).
Insufficient adipogenesis in adipose tissue leads to persistent,
chronic inflammation in adipose tissue, which further inhibits the
differentiation of adipogenic precursors, creating a vicious cycle
(56,59). Angiogenesis occurs prior to
hyperplastic expansion to provide sufficient blood to the
developing tissue. Therefore, ensuring adequate angiogenesis during
adipogenesis plays a crucial role in the composition and function
of adipose tissue. As such, obesity can promote the formation of
new fat cells (lipogenesis) to distribute excess fat and replace
hypertrophic adipocytes, which may have a positive effect on
obesity and adipose tissue inflammation. Studies have confirmed
that m6A and its related factors play important roles in
these two main pathways of adipogenesis and in angiogenesis, with
the specific regulatory mechanisms described below.
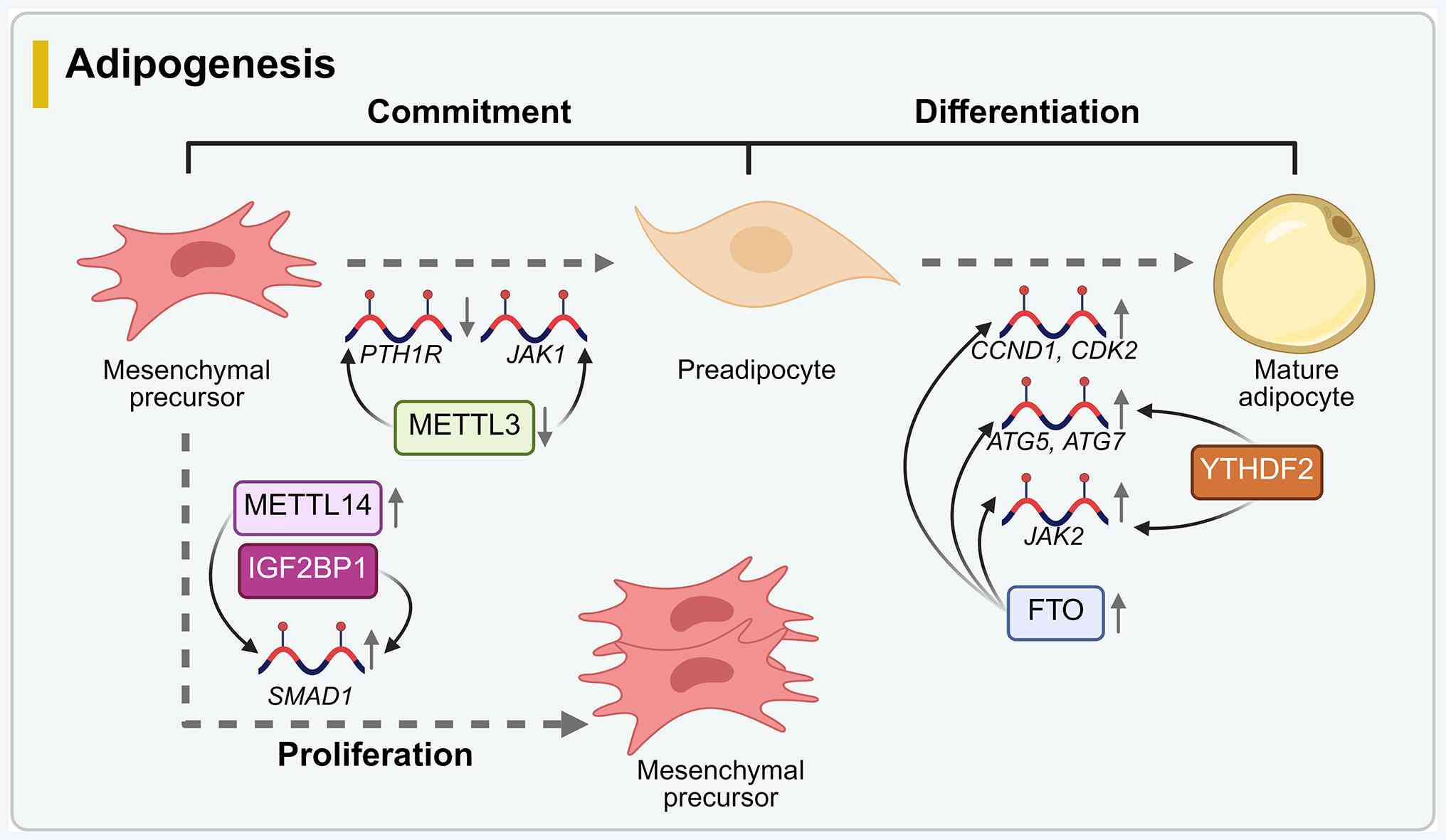
Adipogenesis commitment is a phase in which
mesenchymal precursor cells are induced to form precursor
adipocytes under the influence of signals from bone morphogenetic
protein (BMP), hormones and insulin, during which time the cells do
not undergo morphological changes (56). In an in vivo study using
obese mouse models, METTL3 knockdown promoted the differentiation
of bone marrow mesenchymal stem cells (BMSCs) into adipocytes by
decreasing the translation efficiency of the parathyroid hormone 1
receptor (60). Moreover, an
in vitro study using BMSCs obtained from porcine bone marrow
revealed that METTL3 depletion in pig BMSCs reduces the
m6A levels of Janus kinase (JAK)1 and increases its
expression, resulting in an increase in signal transducer and
activator of transcription (STAT)5 expression, which significantly
regulates the protein levels of CCAAT/enhancer binding protein bate
(C/EBPβ) and stimulates the lipogenic differentiation of BMSCs
(61). Furthermore, in a study
involving in vivo and in vitro experiments using the
ovariectomized mouse model and human BMSCs, as well as research on
human skeletal tissue samples, METTL14 silencing was shown to
downregulate SMAD1 expression through an IGF2BP1-dependent
m6A mechanism, which inhibits BMSC proliferation and
osteogenic differentiation (62). It is therefore reasonable to
speculate that m6A may affect adipogenesis by modulating
BMSC proliferation.
Studies on the differentiation of BMSCs into
adipocyte precursors have focused on METTL3, but whether other
m6A-related proteins are involved in the regulation of
this process remains to be confirmed. The process of adipogenic
commitment is regulated mainly by factors related to those that
promote adipogenesis, such as BMP2/4 (63). Thus, investigating whether such
regulatory factors can have a more efficient effect on adipocyte
production under the influence of m6A is valuable. In
addition, research has focused mainly on the regulation of
m6A-mediated commitment to adipogenesis in BMSCs, and
research on whether adipose-derived stem cells (ADSCs), another
major source of adipocytes, are regulated by m6A during
the process of adipocyte differentiation is insufficient.
In the terminal differentiation stage of
adipogenesis, the precursor adipocytes formed during the commitment
stage first undergo expansion through mitotic cloning, in which
stagnant preadipocytes undergo several rounds of mitosis (64). After undergoing mitotic clone
expansion, preadipocytes become mature adipocytes wherein they lose
their fibroblast form and accumulate cytoplasmic TGs (56). Studies have shown that
m6A also plays a regulatory role at this stage. For
example, transcriptome analysis of mouse fat cell
m6A-sequencing data showed that FTO can target thousands
of m6A-modified genes that are closely related to
obesity and adipogenesis (35).
Furthermore, an in vitro study on mouse 3T3-L1 preadipocytes
confirmed that after FTO knockout, YTHDF2 targets autophagy-related
(ATG)5 and ATG7 transcripts, reducing the expression of ATG5 and
ATG7 and resulting in reduced expression of C/EBPβ and autophagy
inhibition, thus decreasing autophagy and adipogenesis. The
overexpression of FTO promotes autophagy, which promotes
adipogenesis (32). Moreover, an
in vitro study using mouse 3T3-L1 preadipocytes demonstrated
that a lack of FTO also inhibits the expression of JAK2, resulting
in the phosphorylation-mediated inactivation of STAT3, thus
inhibiting the transcription and expression of C/EBPβ and
inhibiting adipogenesis (33).
In addition, for mitotic clone amplification (MCE) in the early
stage of terminal differentiation, the inhibition of FTO expression
in 3T3-L1 cells leads to increased m6A methylation
levels of cyclin D1 (CCND1) and cyclin-dependent kinase 2, the
protein expression of which is reduced after recognition by YTHDF2,
resulting in blockade of the MCE process and in turn the inhibition
of lipogenesis (65) (Fig. 2).
Angiogenesis is the process through which new
capillaries are formed. Healthy adipose tissue is surrounded by a
dense network of capillaries that supply the nutrients and oxygen
necessary for the normal growth of adipocytes. This vascular
network also facilitates the transport of lipids, such as fatty
acids, into and out of adipocytes, serving as a crucial barrier
that helps maintain tissue homeostasis and enables crosstalk with
other organs. In the context of obesity, the expansion of adipose
tissue requires marked angiogenesis to establish a vast vascular
network that can provide ample oxygen and nutrients. During this
process, adipocytes typically secrete a variety of mitogens
specific to endothelial cells and other angiogenic growth factors
to stimulate angiogenesis (69).
However, when influenced by certain pathological factors, the rate
of angiogenesis may match the rapid expansion of adipose tissue. If
the formation of new blood vessels cannot adequately support the
needs of excessive adipose tissue enlargement, local hypoxia may
occur, exacerbating the inflammatory response. Angiogenesis is
regulated primarily by various factors, including proangiogenic
factors such as VEGF, basic fibroblast growth factor and apoptosis
antigen 1 (70,71), and antiangiogenic factors, such
as total suspended particulates, VEGF-A165b and platelet factor 4
(72-74), and the functions of endothelial
cells (75). Endothelial cells
are primarily located in the innermost layer of blood vessels,
where they are responsible for regulating local vascular tone and
permeability; they also coordinate with neighbouring cells to
modulate immune/inflammatory responses and blood supply. The mutual
balance of these factors maintains angiogenesis within the normal
physiological range. Angiogenesis is a complex process that
involves a variety of factors. It is essential to explore new
molecular regulatory networks that can restore or enhance capillary
formation in adipose tissue affected by obesity. Improving the
function of endothelial cells is particularly important, as this
can help alleviate hypoxia in hypertrophic adipocytes and reduce
the local inflammatory microenvironment. Research in this area is
important for understanding and addressing the complications
associated with obesity.
Researchers have developed a model of endothelial
cell proliferation by specifically knocking out the phosphatase and
tensin homologue gene in endothelial cells, to examine the
mechanisms underlying the interaction between endothelial cells and
adipose tissue. Their findings revealed that endothelial cells
communicate with adipocytes by secreting polyamines, which promote
angiogenesis in adipose tissue and help alleviate obesity (86). Therefore, m6A may play
a role in the communication between the endothelium and adipocytes,
thereby influencing angiogenesis in adipose tissue. In addition to
angiogenesis, the structural changes and permeability of blood
vessels also significantly regulate inflammation in adipose tissue.
Research has shown that long non-coding-small nucleolar RNA host
gene 5 can interact with IGF2BP2 in breast cancer-associated
fibroblasts, increasing the stability of zinc finger protein 281
mRNA in an m6A-dependent manner. This interaction
subsequently stimulates both angiogenesis and vascular permeability
(87). However, relatively few
studies on adipose tissue exist in this context, and systematic
conclusions are lacking. Moreover, tumour blood vessels and adipose
tissue blood vessels have different structures and functions.
Therefore, elucidating the role of m6A in the tumour
vascular system may not be directly applicable to the adipose
tissue vascular system, which indicates some limitations of current
research. Therefore, there is great potential for future research
in this direction. In addition, considering that the functional
regulation and angiogenesis of endothelial cells depend on changes
in the local microenvironment, it is difficult to form new
capillaries in adipose tissue in individuals with obesity.
Therefore, investigating whether m6A, as the main
modification that is regulated by the local microenvironment,
mediates angiogenesis by sensing changes in the local
microenvironment may become a new potential research direction with
the aim of regulating adipose tissue angiogenesis and relieving
inflammation in adipose tissue.
In mammals, WAT, which stores energy primarily in
the form of triacylglycerol, mobilizes when needed in the form of
fatty acids (lipolysis). The main function of BAT is to utilize
glucose and lipids to maintain body temperature (thermogenesis)
owing to the specific expression of UCP1, which uncouples the
electron transport chain to produce heat instead of ATP (88). An in vivo study using
mouse models has shown that, on the basis of the plasticity of WAT
and in the context of obesity or high ambient temperature, there is
a lack of leptin receptors, β-adrenergic conduction dysfunction in
brown fat cells and lipase deficiency in the body, resulting in the
gradual transformation of brown fat cells into white unilocular
cells in individuals with obesity (89,90). The proportion of BAT in total
body adipose tissue and activity of BAT decrease, whereas
macrophage infiltration, brown fat cell death and crowd-like
structure (CLS) formation increase, aggravating the inflammatory
response in adipose tissue (89). However, under cold stimulation,
increased body movement and the use of adrenergic receptor β
receptor agonists, AMPK modulators (cordycepin or liraglutide),
sirtuin activators and sodium-glucose cotransporter 2 inhibitors
(empagliflozin) increase the content of BAT or transform some white
adipose cells into beige adipose cells with similar characteristics
and functions; that is, after WAT browning/beiging, the
inflammatory response of adipose tissue and the body is
significantly reduced (91-93). Therefore, the targeted inhibition
of BAT whitening or the promotion of brown adipose cell generation
or WAT browning are highly important for the treatment of adipose
tissue inflammation and related metabolic diseases.
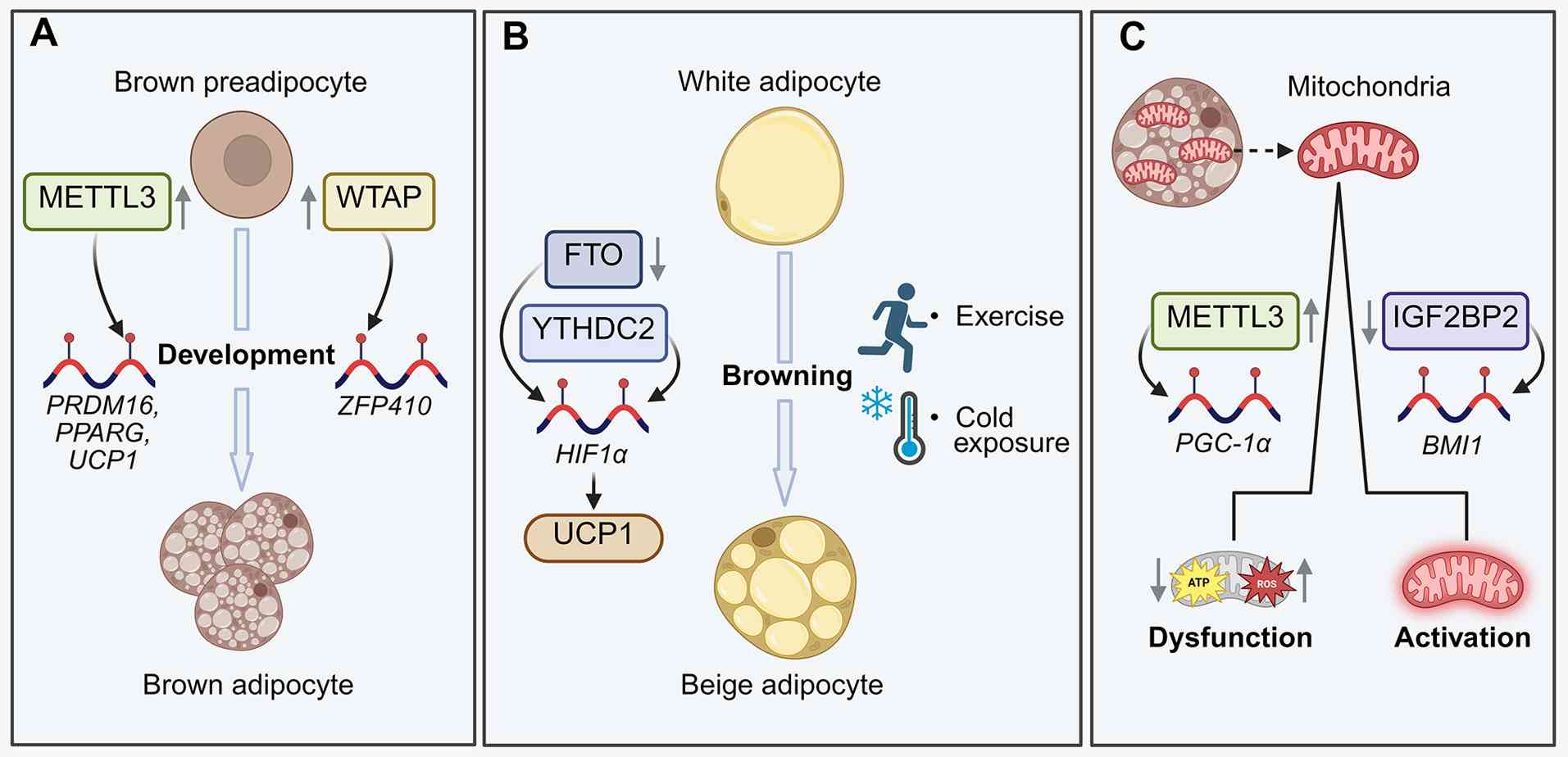
The browning of WAT enhances its thermogenic
capacity by increasing UCP1 mRNA expression in white adipocytes.
This mechanism not only synergizes with non-pharmacological
interventions such as cold exposure, dietary modulation and
physical exercise but also has significant therapeutic potential
for ameliorating metabolic disorders and combating obesity
(96,97). Studies have confirmed that
m6A can effectively regulate the browning of WAT in
adults. For example, in an in vivo study using mouse models,
FTO deletion in white adipose cells increased the m6A
level of HIF1A mRNA, which is recognized by YTHDC2 and increases
the protein expression of HIF1A. This approach also activated the
transcription of PPARγ coactivator-1α (PGC-1α) and other
thermogenesis-related genes and promoted the expression of UCP1 in
and the browning of white fat cells (38).
Additionally, as mitochondria are among the main
components of WAT and BAT, changes in mitochondrial content and
function are crucial for WAT browning. A number of studies have
established that m6A modulates mitochondrial function by
regulating mitochondrial activity, dysfunction and biogenesis. For
example, an in vitro study using the THP-1 cell model has
discovered that, during inflammation, METTL3 increases the
m6A methylation of PGC-1α mRNA and promotes reactive
oxygen species accumulation in monocytes, thereby exacerbating
mitochondrial dysfunction (98).
Moreover, an in vitro and in vivo study using mouse
models has demonstrated that the knockdown of IGF2BP2 increases
mitochondrial activity in haematopoietic stem cells (HSCs) by
promoting the attenuation of B lymphoma Mo-MLV insertion region 1
mRNA, leading to the reactivation of mitochondria-related genes
(99). Evidence from in
vivo and in vitro studies using mouse models and
relevant cell lines demonstrates that exosomes derived from ADSCs
promote the differentiation of beige adipocytes and the browning of
WAT, thereby ameliorating metabolic disorders in mice with
diet-induced obesity (100,101). However, the exact exosomal
components that mediate WAT browning and thermogenesis remain
unknown and require further study. Therefore, whether
m6A affects WAT browning by regulating the crosstalk
between adipocytes and other cells remains to be further confirmed.
In summary, m6A influences the browning of WAT, the
development of BAT and the associated metabolic homeostasis by
modifying key genes and regulating intercellular communication; its
multiple roles in adipose tissue development and functional
regulation are summarized in Fig.
3.
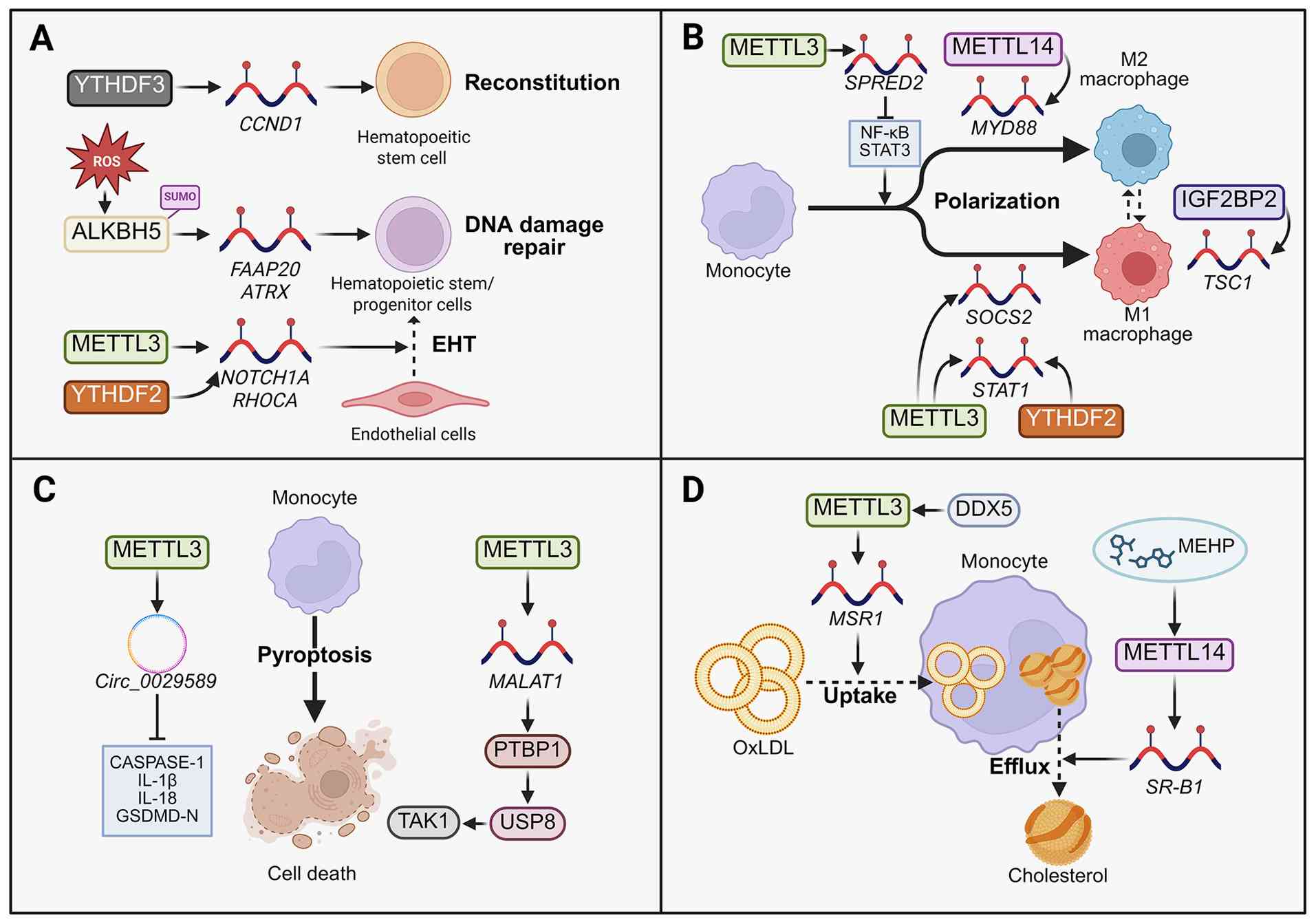
Classical tissue-resident macrophages originate
mainly from the yolk sac or foetal liver during the embryonic
period, are maintained in adulthood through self-proliferation, and
are independent of monocytes. However, under inflammatory
conditions, the primary tissue-resident macrophages that infiltrate
adipose tissue differentiated from monocytes (102). The main function of these
phagocytic cells is to engulf cell debris and pathogens and
activate lymphocytes or other immune cells. Additionally, they are
essential for innate immunity and play notable roles in
inflammatory responses. There are two main types of macrophages: M1
and M2. M1 macrophages are typically activated by interferon-γ and
lipopolysaccharide and release proinflammatory factors. M2
macrophages are activated by type 2 helper T cell-derived cytokines
such as IL-4 and immune complexes, which help suppress inflammatory
factors. This activity contributes to the inhibition of
inflammatory responses and the promotion of tissue repair (103,104). In the context of obesity,
overnutrition leads to adipocyte dysfunction, the induction of
local hypoxia and endoplasmic reticulum stress, the secretion of
numerous chemokines and inflammatory signals, the recruitment of
bone marrow-derived mononuclear macrophages and adipose tissue
macrophage (ATM) infiltration. As an inflammatory microenvironment
forms, the number of macrophages increases, as does the M1/M2
macrophage ratio (9). M1
macrophages are a subpopulation of cells with a predominantly
proinflammatory phenotype; they accumulate around dead adipocytes
to form a CLS and secrete various proinflammatory cytokines
(17,18). Additionally, in a
high-fat-diet-induced animal model of obesity, ATMs in the adipose
tissue of obese mice exhibit increased accumulation of
intracellular lipid droplets (LDs) (105). Based on analysis of tissue
samples from obese patients, compared with normal ATMs, ATMs in
obese individuals have a unique gene expression profile, which is
significantly correlated to insulin resistance (106). Ultimately, the combination of
these factors results in ATMs being closely involved in increasing
the inflammatory response of local adipose tissue and further
increasing the inflammatory response throughout the body.
As aforementioned, obesity-induced inflammation of
adipose tissue is a primary contributor to various metabolic
diseases, including T2DM and MASLD. Inflammatory adipose tissue
releases free fatty acids and inflammatory cytokines, which promote
insulin resistance. These changes notably impact tissue homeostasis
and the progression of metabolic diseases. Recent research has
highlighted the role of m6A modification and its
associated regulatory proteins in various biological functions. The
level of m6A modification, along with its corresponding
effects, largely depends on the regulation of the local
microenvironment. A study has shown that m6A levels
increase or decrease in conjunction with the development of obesity
and related metabolic disorders (10). Furthermore, m6A
modifications are significantly altered in the context of T2DM
(21), MASLD (22) and atherosclerosis (23) and play roles in the regulation of
adipose tissue dysfunction, insulin resistance, liver fibrosis and
other related pathological processes (36,50,67,127). The discovery of how
m6A influences adipose tissue inflammation has gradually
elucidated its potential to alleviate MASLD and T2DM. In this
section, the regulatory functions of m6A-mediated
epigenetic modifications in metabolic diseases are further
explored, with a particular focus on adipose tissue inflammation
and its implications for the treatment of T2DM and MASLD.
MASLD refers to a range of liver conditions that
occur when >5% of liver cells become fatty, typically in
individuals who consume little or no alcohol (128). MASLD is a multisystem disease
that affects various organs outside the liver as well as different
regulatory pathways within the body (129). If left untreated, MASLD can
progress to non-alcoholic steatohepatitis (NASH), which is
characterized by the inflammation and swelling of liver cells. This
condition can further lead to cirrhosis and hepatocellular
carcinoma (HCC), both of which pose serious threats to human health
(130). According to
epidemiological statistics and analyses, the global prevalence of
MASLD is ~25% and is continuously increasing, and the prevalence of
NASH in these patients is also gradually increasing (131,132). Compared with other liver
diseases, HCC is more common in patients with NASH (133,134). Current treatment methods for
MASLD include several mechanism-based approaches. Key classes of
pharmacological agents in clinical development include
glucagon-like peptide-1 receptor agonists such as semaglutide
(135,136), liver-directed thyroid hormone
receptor-β agonists such as resmetirom (137,138) and farnesoid X receptor agonists
such as obeticholic acid (139).
Adipose tissue is a crucial metabolic organ in the
body, influencing the metabolic state of the liver and the
development and progression of MASLD through various pathways. One
major factor involved in this process is the inflammation of
adipose tissue caused by obesity, which significantly contributes
to insulin resistance (140).
In cases of obesity and insulin resistance, lipolysis is increased,
leading to increased production of free fatty acids. Additionally,
the efficiency of fatty acid release from adipose tissue and fatty
acid uptake by the liver is increased in most patients with MASLD.
As these fatty acids enter liver cells (hepatocytes), they promote
the liver accumulation of TGs, which contributes to increased lipid
deposition. This accumulation of lipids is a key factor in the
development of MASLD (5).
Moreover, previous studies and database analyses have revealed
several inflammatory cytokines that play a role in the onset and
progression of MASLD, including IL-1β, IL-6, TNF-α, C-reactive
protein and intercellular adhesion molecule-1 (141-143). Under conditions of adipose
tissue inflammation, the release of inflammatory cytokines such as
TNF-α and IL-6 from adipose tissue increases. This promotes insulin
resistance and contributes to the occurrence and progression of
MASLD (4,7,8,144). Additionally, adipose tissue
plays a role in regulating liver metabolism and insulin resistance
through the secretion of numerous adipokines, including leptin and
adiponectin (145-147). From the above, it can be
observed that inflammation in adipose tissue plays significant
roles in the onset and progression of MASLD. Elucidating
communication between adipose tissue inflammation and liver
metabolic function holds considerable research significance.
Diabetes and its complications pose notable threats
to global health. According to the International Diabetes
Federation, the global prevalence of diabetes was estimated to be
9.3% (463 million people) in 2019; this figure is projected to
increase to 10.2% (578 million people) by 2030 and further to 10.9%
(700 million people) by 2045 (155). At present, T2DM accounts for
>90% of all diabetes cases, making it a major global health
issue (156,157). Key factors contributing to the
development of T2DM include increasing rates of obesity, the lack
of physical activity, the consumption of high-calorie diets and an
ageing population (158).
Adipose tissue inflammation plays a role in influencing the onset
and progression of T2DM through various mechanisms. For instance,
adipose tissue induces insulin resistance through the release of
adipokines under chronic inflammatory conditions. The insulin
secreted by pancreatic β cells is not sufficient to adequately
counteract this insulin resistance, leading to pancreatic β-cell
dysfunction, glucose intolerance and ultimately T2DM (6,159). Understanding the mechanisms
that connect adipose tissue inflammation to T2DM, particularly
concerning the regulation of m6A, is highly
important.
Insulin resistance plays a crucial role in the
development of T2DM. In particular, the cytokines and inflammatory
factors (such as TNF-α and IL-6) secreted from adipose tissue cells
due to inflammation can significantly affect insulin sensitivity
and fat metabolism (164,165). It is therefore important to
further investigate how m6A modification in inflammatory
adipose tissue regulates the transcription and translation of these
factors. Additionally, future research should further investigate
the molecular networks and dynamic changes involved in
m6A modification during adipose tissue inflammation and
its role in the pathogenesis of T2DM. Such studies should focus
specifically on how m6A modification functions in
different types of adipose tissues (white, brown and beige) and how
it interacts synergistically with other metabolic organs, such as
the liver, muscle and pancreas. Such investigations could provide a
new perspective on our understanding of the pathogenesis of T2DM.
Additionally, an in vivo study using type 2 diabetes cardiac
fibroblast-specific NOTCH1 conditional knockout mouse models has
shown that m6A is closely involved in the complications
of diabetes, specifically diabetic cardiomyopathy. This is
primarily reflected in how ALKBH5 deficiency increases NOTCH1
methylation to promote mitochondrial fission (166). Finally, examining the specific
regulatory molecules that influence m6A
modification-related enzymes (including METTL3, METTL14 and WTAP)
could lead to the development of potential therapeutic strategies
aimed at alleviating adipose tissue inflammation and improving
insulin resistance. The development of small molecule inhibitors or
activators in this area holds notable research value.
Despite advancements in recent research, a number
of questions remain unanswered regarding the specific roles of
m6A in adipose tissue inflammation and related metabolic
diseases. First, how m6A modification influences the
inflammatory response and metabolic functions of adipose tissue
through the regulation of gene expression in various types of
adipocytes and immune cells remains unclear. Second, the dynamic
changes in m6A regulators in the context of obesity and
metabolic diseases, along with the complexity of their regulatory
networks, necessitate further experimental data to provide clarity.
Additionally, there may be notable differences in m6A
modifications and their regulatory mechanisms across different
tissues and cell types. Moreover, current insights into the roles
of m6A in adipose tissue inflammation and associated
metabolic disorders are predominantly derived from mouse or pig
models. While these animal studies are instrumental for elucidating
underlying mechanisms, certain differences in fat distribution,
immune responses and the m6A regulatory network exist in
these species. Therefore, the translation of these findings into
human clinical applications requires careful consideration and
further validation.
Not applicable.
XY and HT conceived the review. XW and ST wrote the
first version of the manuscript. XY and HT jointly oversaw the
writing of all versions following the initial draft. XZ and KY
revised the manuscript. XY was responsible for all revisions and
refinements after the manuscript was submitted. All authors read
and approved the final version of the manuscript. Data
authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
This review was supported by the Natural Science Foundation of
the Guangxi Zhuang Autonomous Region (grant no. 2024GXNSFAA010154),
the National Natural Sciences Foundation of China (grant nos.
82570989 and 82370463), the China Postdoctoral Science Foundation
(grant no. 2024M761313) and the Guangdong Basic and Applied Basic
Research Foundation (grant no. 2025A1515012522).
|
1
|
No authors listed. Correction to: Obesity
phenotypes, diabetes, and cardiovascular diseases. Circ Res.
127:e1072020.PubMed/NCBI
|
|
2
|
Ma H, Wang X, Heianza Y, Manson JE and Qi
L: Proteomic signature of BMI and risk of cardiovascular disease.
Clin Chem. 70:1474–1484. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Caballero B: Humans against obesity: Who
will win? Adv Nutr. 10(Suppl 1): S4–S9. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kawai T, Autieri MV and Scalia R: Adipose
tissue inflammation and metabolic dysfunction in obesity. Am J
Physiol Cell Physiol. 320:C375–C391. 2021. View Article : Google Scholar
|
|
5
|
Donnelly KL, Smith CI, Schwarzenberg SJ,
Jessurun J, Boldt MD and Parks EJ: Sources of fatty acids stored in
liver and secreted via lipoproteins in patients with nonalcoholic
fatty liver disease. J Clin Invest. 115:1343–1351. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kahn SE, Hull RL and Utzschneider KM:
Mechanisms linking obesity to insulin resistance and type 2
diabetes. Nature. 444:840–846. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fontana L, Eagon JC, Trujillo ME, Scherer
PE and Klein S: Visceral fat adipokine secretion is associated with
systemic inflammation in obese humans. Diabetes. 56:1010–1013.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Senn JJ, Klover PJ, Nowak IA, Zimmers TA,
Koniaris LG, Furlanetto RW and Mooney RA: Suppressor of cytokine
signaling-3 (SOCS-3), a potential mediator of
interleukin-6-dependent insulin resistance in hepatocytes. J Biol
Chem. 278:13740–13746. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lumeng CN, Bodzin JL and Saltiel AR:
Obesity induces a phenotypic switch in adipose tissue macrophage
polarization. J Clin Invest. 117:175–184. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rønningen T, Dahl MB, Valderhaug TG, Cayir
A, Keller M, Tönjes A, Blüher M and Böttcher Y: m6A regulators in
human adipose tissue-depot-specificity and correlation with
obesity. Front Endocrinol (Lausanne). 12:7788752021. View Article : Google Scholar
|
|
11
|
Wu H and Ballantyne CM: Skeletal muscle
inflammation and insulin resistance in obesity. J Clin Invest.
127:43–54. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Becher T, Palanisamy S, Kramer DJ, Eljalby
M, Marx SJ, Wibmer AG, Butler SD, Jiang CS, Vaughan R, Schöder H,
et al: Brown adipose tissue is associated with cardiometabolic
health. Nat Med. 27:58–65. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Villarroya F, Cereijo R, Gavaldà-Navarro
A, Villarroya J and Giralt M: Inflammation of brown/beige adipose
tissues in obesity and metabolic disease. J Intern Med.
284:492–504. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chaturvedi S, Chaturvedi P, Gupta PC,
Awasthi SK and Kalani A: Molecular epigenetics in the transition of
white to brown fat. Pathol Res Pract. 272:1560732025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fève B, Cinti S, Beaupère C, Vatier C,
Vigouroux C, Vali A, Capeau J, Grosfeld A and Moldes M: Pink
adipose tissue: A paradigm of adipose tissue plasticity. Ann
Endocrinol (Paris). 85:248–251. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Curat CA, Miranville A, Sengenès C, Diehl
M, Tonus C, Busse R and Bouloumié A: From blood monocytes to
adipose tissue-resident macrophages: Induction of diapedesis by
human mature adipocytes. Diabetes. 53:1285–1292. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Weisberg SP, McCann D, Desai M, Rosenbaum
M, Leibel RL and Ferrante AW Jr: Obesity is associated with
macrophage accumulation in adipose tissue. J Clin Invest.
112:1796–1808. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Haase J, Weyer U, Immig K, Klöting N,
Blüher M, Eilers J, Bechmann I and Gericke M: Local proliferation
of macrophages in adipose tissue during obesity-induced
inflammation. Diabetologia. 57:562–571. 2014. View Article : Google Scholar
|
|
19
|
Skurk T, Alberti-Huber C, Herder C and
Hauner H: Relationship between adipocyte size and adipokine
expression and secretion. J Clin Endocrinol Metab. 92:1023–1033.
2007. View Article : Google Scholar
|
|
20
|
Giordano A, Murano I, Mondini E, Perugini
J, Smorlesi A, Severi I, Barazzoni R, Scherer PE and Cinti S: Obese
adipocytes show ultrastructural features of stressed cells and die
of pyroptosis. J Lipid Res. 54:2423–2436. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang Y, Shen F, Huang W, Qin S, Huang JT,
Sergi C, Yuan BF and Liu SM: Glucose is involved in the dynamic
regulation of m6A in patients with type 2 diabetes. J Clin
Endocrinol Metab. 104:665–673. 2019. View Article : Google Scholar
|
|
22
|
Qin Y, Li B, Arumugam S, Lu Q, Mankash SM,
Li J, Sun B, Li J, Flavell RA, Li HB and Ouyang X: m6A
mRNA methylation-directed myeloid cell activation controls
progression of NAFLD and obesity. Cell Rep. 37:1099682021.
View Article : Google Scholar
|
|
23
|
Jian D, Wang Y, Jian L, Tang H, Rao L,
Chen K, Jia Z, Zhang W, Liu Y, Chen X, et al: METTL14 aggravates
endothelial inflammation and atherosclerosis by increasing FOXO1
N6-methyladeosine modifications. Theranostics. 10:8939–8956. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Long H, Yu Y, Ouyang J, Lu H and Zhao G:
Insights into RNA N6-methyladenosine and programmed cell death in
atherosclerosis. Mol Med. 30:1372024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang X, Feng J, Xue Y, Guan Z, Zhang D,
Liu Z, Gong Z, Wang Q, Huang J, Tang C, et al: Structural basis of
N(6)-adenosine methylation by the METTL3-METTL14 complex. Nature.
534:575–578. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ping XL, Sun BF, Wang L, Xiao W, Yang X,
Wang WJ, Adhikari S, Shi Y, Lv Y, Chen YS, et al: Mammalian WTAP is
a regulatory subunit of the RNA N6-methyladenosine
methyltransferase. Cell Res. 24:177–189. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jiang X, Liu B, Nie Z, Duan L, Xiong Q,
Jin Z, Yang C and Chen Y: The role of m6A modification in the
biological functions and diseases. Signal Transduct Target Ther.
6:742021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ye M, Chen J, Lu F, Zhao M, Wu S, Hu C, Yu
P, Kan J, Bai J, Tian Y and Tang Q: Down-regulated FTO and ALKBH5
co-operatively activates FOXO signaling through m6A methylation
modification in HK2 mRNA mediated by IGF2BP2 to enhance glycolysis
in colorectal cancer. Cell Biosci. 13:1482023. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang Q, Chen C, Ding Q, Zhao Y, Wang Z,
Chen J, Jiang Z, Zhang Y, Xu G, Zhang J, et al: METTL3-mediated
m6A modification of HDGF mRNA promotes gastric cancer
progression and has prognostic significance. Gut. 69:1193–1205.
2020. View Article : Google Scholar
|
|
30
|
Huo FC, Zhu ZM, Du WQ, Pan YJ, Jiang X,
Kang MJ, Liu BW, Mou J and Pei DS: HPV E7-drived ALKBH5 promotes
cervical cancer progression by modulating m6A modification of PAK5.
Pharmacol Res. 195:1068632023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wu T, Liao L, Wu T, Chen S, Yi Q and Xu M:
IGF2BP2 promotes glycolysis and hepatocellular carcinoma stemness
by stabilizing CDC45 mRNA via m6A modification. Cell Cycle.
22:2245–2263. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang X, Wu R, Liu Y, Zhao Y, Bi Z, Yao Y,
Liu Q, Shi H, Wang F and Wang Y: m6A mRNA methylation
controls autophagy and adipogenesis by targeting Atg5 and Atg7.
Autophagy. 16:1221–1235. 2020. View Article : Google Scholar :
|
|
33
|
Wu R, Guo G, Bi Z, Liu Y, Zhao Y, Chen N,
Wang F, Wang Y and Wang X: m6A methylation modulates
adipogenesis through JAK2-STAT3-C/EBPβ signaling. Biochim Biophys
Acta Gene Regul Mech. 1862:796–806. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kobayashi M, Ohsugi M, Sasako T, Awazawa
M, Umehara T, Iwane A, Kobayashi N, Okazaki Y, Kubota N, Suzuki R,
et al: The RNA methyltransferase complex of WTAP, METTL3, and
METTL14 regulates mitotic clonal expansion in adipogenesis. Mol
Cell Biol. 38:e00116–18. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhao X, Yang Y, Sun BF, Shi Y, Yang X,
Xiao W, Hao YJ, Ping XL, Chen YS, Wang WJ, et al: FTO-dependent
demethylation of N6-methyladenosine regulates mRNA splicing and is
required for adipogenesis. Cell Res. 24:1403–1419. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Merkestein M, Laber S, McMurray F, Andrew
D, Sachse G, Sanderson J, Li M, Usher S, Sellayah D, Ashcroft FM
and Cox RD: FTO influences adipogenesis by regulating mitotic
clonal expansion. Nat Commun. 6:67922015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang CY, Shie SS, Wen MS, Hung KC, Hsieh
IC, Yeh TS and Wu D: Loss of FTO in adipose tissue decreases
Angptl4 translation and alters triglyceride metabolism. Sci Signal.
8:ra1272015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wu R, Chen Y, Liu Y, Zhuang L, Chen W,
Zeng B, Liao X, Guo G, Wang Y and Wang X: m6A methylation promotes
white-to-beige fat transition by facilitating Hif1a translation.
EMBO Rep. 22:e523482021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhao Y, Hu J, Sun X, Yang K, Yang L, Kong
L, Zhang B, Li F, Li C, Shi B, et al: Loss of m6A demethylase
ALKBH5 promotes post-ischemic angiogenesis via post-transcriptional
stabilization of WNT5A. Clin Transl Med. 11:e4022021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang H, Hu X, Huang M, Liu J, Gu Y, Ma L,
Zhou Q and Cao X: Mettl3-mediated mRNA m6A methylation
promotes dendritic cell activation. Nat Commun. 10:18982019.
View Article : Google Scholar
|
|
41
|
Yang Z, Wang T, Wu D, Min Z, Tan J and Yu
B: RNA N6-methyladenosine reader IGF2BP3 regulates cell cycle and
angiogenesis in colon cancer. J Exp Clin Cancer Res. 39:2032020.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yao MD, Jiang Q, Ma Y, Liu C, Zhu CY, Sun
YN, Shan K, Ge HM, Zhang QY, Zhang HY, et al: Role of
METTL3-dependent N6-methyladenosine mRNA modification in
the promotion of angiogenesis. Mol Ther. 28:2191–2202. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang QA, Tao C, Gupta RK and Scherer PE:
Tracking adipogenesis during white adipose tissue development,
expansion and regeneration. Nat Med. 19:1338–1344. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Spencer M, Unal R, Zhu B, Rasouli N,
McGehee RE Jr, Peterson CA and Kern PA: Adipose tissue
extracellular matrix and vascular abnormalities in obesity and
insulin resistance. J Clin Endocrinol Metab. 96:E1990–E1998. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Trayhurn P: Hypoxia and adipose tissue
function and dysfunction in obesity. Physiol Rev. 93:1–21. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wei D, Sun Q, Li Y, Li C, Li X and Sun C:
Leptin reduces Plin5 m6A methylation through FTO to
regulate lipolysis in piglets. Int J Mol Sci. 22:106102021.
View Article : Google Scholar
|
|
47
|
Xiao Y, Jiang T, Qi X, Zhou J, Pan T, Liao
Q, Liu S, Zhang H, Wang J, Yang X, et al: PROTAC-mediated FTO
protein degradation effectively alleviates diet-induced obesity and
hepatic steatosis. Int J Biol Macromol. 285:1382922025. View Article : Google Scholar
|
|
48
|
Huang X, Huang X, Guo H, Li J, Zhou C,
Huang Y, Lai C, Zeng W, Tan X, Niu L, et al: Intermittent
hypoxia-induced METTL3 downregulation facilitates MGLL-mediated
lipolysis of adipocytes in OSAS. Cell Death Discov. 8:3522022.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yang Z, Yu GL, Zhu X, Peng TH and Lv YC:
Critical roles of FTO-mediated mRNA m6A demethylation in regulating
adipogenesis and lipid metabolism: Implications in lipid metabolic
disorders. Genes Dis. 9:51–61. 2021. View Article : Google Scholar
|
|
50
|
Kang Q, Zhu X, Ren D, Ky A, MacDougald OA,
O'Rourke RW and Rui L: Adipose METTL14-elicited N6
-methyladenosine promotes obesity, insulin resistance, and NAFLD
through suppressing β adrenergic signaling and lipolysis. Adv Sci
(Weinh). 10:e23016452023. View Article : Google Scholar
|
|
51
|
Guo H, Wang B, Xu K, Nie L, Fu Y, Wang Z,
Wang Q, Wang S and Zou X: m6A reader HNRNPA2B1 promotes
esophageal cancer progression via up-regulation of ACLY and ACC1.
Front Oncol. 10:5530452020. View Article : Google Scholar
|
|
52
|
Wu W, Wang S, Liu Q, Shan T, Wang X, Feng
J and Wang Y: AMPK facilitates intestinal long-chain fatty acid
uptake by manipulating CD36 expression and translocation. FASEB J.
34:4852–4869. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lan J, Xu B, Shi X, Pan Q and Tao Q:
WTAP-mediated N6-methyladenosine modification of NLRP3
mRNA in kidney injury of diabetic nephropathy. Cell Mol Biol Lett.
27:512022. View Article : Google Scholar
|
|
54
|
Liu BH, Tu Y, Ni GX, Yan J, Yue L, Li ZL,
Wu JJ, Cao YT, Wan ZY, Sun W and Wan YG: Total flavones of
abelmoschus manihot ameliorates podocyte pyroptosis and injury in
high glucose conditions by targeting METTL3-dependent
m6A modification-mediated NLRP3-inflammasome activation
and PTEN/PI3K/Akt signaling. Front Pharmacol. 12:6676442021.
View Article : Google Scholar
|
|
55
|
Yuan X, Li T, Shi L, Miao J, Guo Y and
Chen Y: Human umbilical cord mesenchymal stem cells deliver
exogenous miR-26a-5p via exosomes to inhibit nucleus pulposus cell
pyroptosis through METTL14/NLRP3. Mol Med. 27:912021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Ghaben AL and Scherer PE: Adipogenesis and
metabolic health. Nat Rev Mol Cell Biol. 20:242–258. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Spalding KL, Arner E, Westermark PO,
Bernard S, Buchholz BA, Bergmann O, Blomqvist L, Hoffstedt J,
Näslund E, Britton T, et al: Dynamics of fat cell turnover in
humans. Nature. 453:783–787. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Shao M, Vishvanath L, Busbuso NC, Hepler
C, Shan B, Sharma AX, Chen S, Yu X, An YA, Zhu Y, et al: De novo
adipocyte differentiation from Pdgfrβ+ preadipocytes
protects against pathologic visceral adipose expansion in obesity.
Nat Commun. 9:8902018. View Article : Google Scholar
|
|
59
|
Wernstedt Asterholm I, Tao C, Morley TS,
Wang QA, Delgado-Lopez F, Wang ZV and Scherer PE: Adipocyte
inflammation is essential for healthy adipose tissue expansion and
remodeling. Cell Metab. 20:103–118. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Wu Y, Xie L, Wang M, Xiong Q, Guo Y, Liang
Y, Li J, Sheng R, Deng P, Wang Y, et al: Mettl3-mediated
m6A RNA methylation regulates the fate of bone marrow
mesenchymal stem cells and osteoporosis. Nat Commun. 9:47722018.
View Article : Google Scholar
|
|
61
|
Yao Y, Bi Z, Wu R, Zhao Y, Liu Y, Liu Q,
Wang Y and Wang X: METTL3 inhibits BMSC adipogenic differentiation
by targeting the JAK1/STAT5/C/EBPβ pathway via an
m6A-YTHDF2-dependent manner. FASEB J. 33:7529–7544.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Huang C and Wang Y: Downregulation of
METTL14 improves postmenopausal osteoporosis via IGF2BP1 dependent
posttranscriptional silencing of SMAD1. Cell Death Dis. 13:9192022.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Huang H, Song TJ, Li X, Hu L, He Q, Liu M,
Lane MD and Tang QQ: BMP signaling pathway is required for
commitment of C3H10T1/2 pluripotent stem cells to the adipocyte
lineage. Proc Natl Acad Sci USA. 106:12670–12675. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Patel YM and Lane MD: Mitotic clonal
expansion during preadipocyte differentiation: Calpain-mediated
turnover of p27. J Biol Chem. 275:17653–17660. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Liao X, Liu J, Chen Y, Liu Y, Chen W, Zeng
B, Liu Y, Luo Y, Huang C, Guo G, et al: Metformin combats obesity
by targeting FTO in an m6A-YTHDF2-dependent manner. J
Drug Target. 30:983–991. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Wang X, Sun B, Jiang Q, Wu R, Cai M, Yao
Y, Liu Q, Shi H, Feng J and Wang Y: mRNA m6A plays
opposite role in regulating UCP2 and PNPLA2 protein expression in
adipocytes. Int J Obes (Lond). 42:1912–1924. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Li Y, Zhang Q, Cui G, Zhao F, Tian X, Sun
BF, Yang Y and Li W: m6A regulates liver metabolic
disorders and hepatogenous diabetes. Genomics Proteomics
Bioinformatics. 18:371–383. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Xu Z, Qin Y, Lv B, Tian Z and Zhang B:
Intermittent fasting improves high-fat diet-induced obesity
cardiomyopathy via alleviating lipid deposition and apoptosis and
decreasing m6A methylation in the heart. Nutrients. 14:2512022.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Herold J and Kalucka J: Angiogenesis in
adipose tissue: The interplay between adipose and endothelial
cells. Front Physiol. 11:6249032021. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ferrara N and Adamis AP: Ten years of
anti-vascular endothelial growth factor therapy. Nat Rev Drug
Discov. 15:385–403. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Edelman ER, Mathiowitz E, Langer R and
Klagsbrun M: Controlled and modulated release of basic fibroblast
growth factor. Biomaterials. 12:619–626. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Kazerounian S, Yee KO and Lawler J:
Thrombospondins in cancer. Cell Mol Life Sci. 65:700–712. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Karki S, Ngo DTM, Farb MG, Park SY,
Saggese SM, Hamburg NM, Carmine B, Hess DT, Walsh K and Gokce N:
WNT5A regulates adipose tissue angiogenesis via antiangiogenic
VEGF-A165b in obese humans. Am J Physiol Heart Circ
Physiol. 313:H200–H206. 2017. View Article : Google Scholar
|
|
74
|
Bikfalvi A: Platelet factor 4: An
inhibitor of angiogenesis. Semin Thromb Hemost. 30:379–385. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Leung SWS and Shi Y: The glycolytic
process in endothelial cells and its implications. Acta Pharmacol
Sin. 43:251–259. 2022. View Article : Google Scholar :
|
|
76
|
Shen W, Pu J, Zuo Z, Gu S, Sun J, Tan B,
Wang L, Cheng J and Zuo Y: The RNA demethylase ALKBH5 promotes the
progression and angiogenesis of lung cancer by regulating the
stability of the LncRNA PVT1. Cancer Cell Int. 22:3532022.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Zhi S, Li J, Kong X, Xie X, Zhang Q and
Fang G: Insulin-like growth factor 2 mRNA binding protein 2
regulates proliferation, migration, and angiogenesis of
keratinocytes by modulating heparanase stability. Bioengineered.
12:11267–11276. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Zhang G, Wang T, Huang Z, Chen Y, Sun L,
Xia X, He F, Fan C, Wang S and Liu W: METTL3 dual regulation of the
stability of LINC00662 and VEGFA RNAs promotes colorectal cancer
angiogenesis. Discov Oncol. 13:892022. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Jiang L, Li Y, He Y, Wei D, Yan L and Wen
H: Knockdown of m6A reader IGF2BP3 inhibited hypoxia-induced cell
migration and angiogenesis by regulating hypoxia inducible
factor-1α in stomach cancer. Front Oncol. 11:7112072021. View Article : Google Scholar
|
|
80
|
Ma YS, Shi BW, Guo JH, Liu JB, Yang XL,
Xin R, Shi Y, Zhang DD, Lu GX, Jia CY, et al: microRNA-320b
suppresses HNF4G and IGF2BP2 expression to inhibit angiogenesis and
tumor growth of lung cancer. Carcinogenesis. 42:762–771. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wang J, Tan L, Yu X, Cao X, Jia B, Chen R
and Li J: lncRNA ZNRD1-AS1 promotes malignant lung cell
proliferation, migration, and angiogenesis via the miR-942/TNS1
axis and is positively regulated by the m6A reader
YTHDC2. Mol Cancer. 21:2292022. View Article : Google Scholar
|
|
82
|
Rudnicki M, Abdifarkosh G, Nwadozi E,
Ramos SV, Makki A, Sepa-Kishi DM, Ceddia RB, Perry CG, Roudier E
and Haas TL: Endothelial-specific FoxO1 depletion prevents
obesity-related disorders by increasing vascular metabolism and
growth. Elife. 7:e397802018. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Dong G, Yu J, Shan G, Su L, Yu N and Yang
S: N6-methyladenosine methyltransferase METTL3 promotes
angiogenesis and atherosclerosis by upregulating the JAK2/STAT3
pathway via m6A reader IGF2BP1. Front Cell Dev Biol. 9:7318102021.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Wang LJ, Xue Y, Li H, Huo R, Yan Z, Wang
J, Xu H, Wang J, Cao Y and Zhao JZ: Wilms' tumour 1-associating
protein inhibits endothelial cell angiogenesis by m6A-dependent
epigenetic silencing of desmoplakin in brain arteriovenous
malformation. J Cell Mol Med. 24:4981–4991. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Wang LJ, Xue Y, Huo R, Yan Z, Xu H, Li H,
Wang J, Zhang Q, Cao Y and Zhao JZ: N6-methyladenosine
methyltransferase METTL3 affects the phenotype of cerebral
arteriovenous malformation via modulating Notch signaling pathway.
J Biomed Sci. 27:622020. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Monelli E, Villacampa P, Zabala-Letona A,
Martinez-Romero A, Llena J, Beiroa D, Gouveia L, Chivite I, Zagmutt
S, Gama-Perez P, et al: Angiocrine polyamine production regulates
adiposity. Nat Metab. 4:327–343. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Zeng H, Hou Y, Zhou X, Lang L, Luo H, Sun
Y, Wan X, Yuan T, Wang R, Liu Y, et al: Cancer-associated
fibroblasts facilitate premetastatic niche formation through lncRNA
SNHG5-mediated angiogenesis and vascular permeability in breast
cancer. Theranostics. 12:7351–7370. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Gong D, Lei J, He X, Hao J, Zhang F, Huang
X, Gu W, Yang X and Yu J: Keys to the switch of fat burning:
Stimuli that trigger the uncoupling protein 1 (UCP1) activation in
adipose tissue. Lipids Health Dis. 23:3222024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Kotzbeck P, Giordano A, Mondini E, Murano
I, Severi I, Venema W, Cecchini MP, Kershaw EE, Barbatelli G,
Haemmerle G, et al: Brown adipose tissue whitening leads to brown
adipocyte death and adipose tissue inflammation. J Lipid Res.
59:784–794. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Peng Y, Zhao L, Li M, Liu Y, Shi Y and
Zhang J: Plasticity of adipose tissues: Interconversion among
white, brown, and beige fat and its role in energy homeostasis.
Biomolecules. 14:4832024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Aldiss P, Betts J, Sale C, Pope M, Budge H
and Symonds ME: Exercise-induced 'browning' of adipose tissues.
Metabolism. 81:63–70. 2018. View Article : Google Scholar :
|
|
92
|
Kuryłowicz A and Puzianowska-Kuźnicka M:
Induction of adipose tissue browning as a strategy to combat
obesity. Int J Mol Sci. 21:62412020. View Article : Google Scholar
|
|
93
|
Lim S, Honek J, Xue Y, Seki T, Cao Z,
Andersson P, Yang X, Hosaka K and Cao Y: Cold-induced activation of
brown adipose tissue and adipose angiogenesis in mice. Nat Protoc.
7:606–615. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Wang Y, Gao M, Zhu F, Li X, Yang Y, Yan Q,
Jia L, Xie L and Chen Z: METTL3 is essential for postnatal
development of brown adipose tissue and energy expenditure in mice.
Nat Commun. 11:16482020. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Tao X, Du R, Guo S, Feng X, Yu T, OuYang
Q, Chen Q, Fan X, Wang X, Guo C, et al: PGE2-EP3 axis
promotes brown adipose tissue formation through stabilization of
WTAP RNA methyltransferase. EMBO J. 41:e1104392022. View Article : Google Scholar
|
|
96
|
Wang CH, Tsuji T, Wu LH, Yang CY, Huang
TL, Sato M, Shamsi F and Tseng YH: Endothelin 3/EDNRB signaling
induces thermogenic differentiation of white adipose tissue. Nat
Commun. 15:72152024. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wang G, Meyer JG, Cai W, Softic S, Li ME,
Verdin E, Newgard C, Schilling B and Kahn CR: Regulation of UCP1
and mitochondrial metabolism in brown adipose tissue by reversible
succinylation. Mol Cell. 74:844–857.e7. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Zhang X, Li X, Jia H, An G and Ni J: The
m6A methyltransferase METTL3 modifies PGC-1α mRNA
promoting mitochondrial dysfunction and oxLDL-induced inflammation
in monocytes. J Biol Chem. 297:1010582021. View Article : Google Scholar
|
|
99
|
Yin R, Chang J, Li Y, Gao Z, Qiu Q, Wang
Q, Han G, Chai J, Feng M, Wang P, et al: Differential
m6A RNA landscapes across hematopoiesis reveal a role
for IGF2BP2 in preserving hematopoietic stem cell function. Cell
Stem Cell. 29:149–159.e7. 2022. View Article : Google Scholar
|
|
100
|
Jung YJ, Kim HK, Cho Y, Choi JS, Woo CH,
Lee KS, Sul JH, Lee CM, Han J, Park JH, et al: Cell reprogramming
using extracellular vesicles from differentiating stem cells into
white/beige adipocytes. Sci Adv. 6:eaay67212020. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Zhao H, Shang Q, Pan Z, Bai Y, Li Z, Zhang
H, Zhang Q, Guo C, Zhang L and Wang Q: Exosomes from
adipose-derived stem cells attenuate adipose inflammation and
obesity through polarizing M2 macrophages and beiging in white
adipose tissue. Diabetes. 67:235–247. 2018. View Article : Google Scholar
|
|
102
|
Chen Y and Zhang X: Pivotal regulators of
tissue homeostasis and cancer: Macrophages. Exp Hematol Oncol.
6:232017. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Randolph GJ: Immunology. No need to coax
monocytes. Science. 332:1268–1269. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Risser GE, Machour M, Hernaez-Estrada B,
Li D, Levenberg S and Spiller KL: Effects of Interleukin-4
(IL-4)-releasing microparticles and adoptive transfer of
macrophages on immunomodulation and angiogenesis. Biomaterials.
296:1220952023. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Li Y, Du Y, Xu Z, He Y, Yao R, Jiang H, Ju
W, Qiao J, Xu K, Liu TM and Zeng L: Intravital lipid droplet
labeling and imaging reveals the phenotypes and functions of
individual macrophages in vivo. J Lipid Res. 63:1002072022.
View Article : Google Scholar
|
|
106
|
Li C, Menoret A, Farragher C, Ouyang Z,
Bonin C, Holvoet P, Vella AT and Zhou B: Single cell
transcriptomics based-MacSpectrum reveals novel macrophage
activation signatures in diseases. JCI Insight. 5:e1264532019.
View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Zhang C, Chen Y, Sun B, Wang L, Yang Y, Ma
D, Lv J, Heng J, Ding Y, Xue Y, et al: m6A modulates
haematopoietic stem and progenitor cell specification. Nature.
549:273–276. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Zhang X, Cong T, Wei L, Zhong B, Wang X,
Sun J, Wang S, Xu MM, Zhu P, Jiang H and Wang J: YTHDF3 modulates
hematopoietic stem cells by recognizing RNA m6A
modification on Ccnd1. Haematologica. 107:2381–2394. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Yu F, Wei J, Cui X, Yu C, Ni W, Bungert J,
Wu L, He C and Qian Z: Post-translational modification of RNA m6A
demethylase ALKBH5 regulates ROS-induced DNA damage response.
Nucleic Acids Res. 49:5779–5797. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Yin H, Zhang X, Yang P, Zhang X, Peng Y,
Li D, Yu Y, Wu Y, Wang Y, Zhang J, et al: RNA m6A methylation
orchestrates cancer growth and metastasis via macrophage
reprogramming. Nat Commun. 12:13942021. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Zheng Y, Li Y, Ran X, Wang D, Zheng X,
Zhang M, Yu B, Sun Y and Wu J: Mettl14 mediates the inflammatory
response of macrophages in atherosclerosis through the NF-κB/IL-6
signaling pathway. Cell Mol Life Sci. 79:3112022. View Article : Google Scholar
|
|
112
|
Huangfu N, Zheng W, Xu Z, Wang S, Wang Y,
Cheng J, Li Z, Cheng K, Zhang S, Chen X and Zhu J: RBM4 regulates
M1 macrophages polarization through targeting STAT1-mediated
glycolysis. Int Immunopharmacol. 83:1064322020. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Liu Y, Liu Z, Tang H, Shen Y, Gong Z, Xie
N, Zhang X, Wang W, Kong W, Zhou Y and Fu Y: The
N6-methyladenosine (m6A)-forming enzyme
METTL3 facilitates M1 macrophage polarization through the
methylation of STAT1 mRNA. Am J Physiol Cell Physiol.
317:C762–C775. 2019. View Article : Google Scholar
|
|
114
|
Zhong C, Tao B, Yang F, Xia K, Yang X,
Chen L, Peng T, Xia X, Li X and Peng L: Histone demethylase JMJD1C
promotes the polarization of M1 macrophages to prevent glioma by
upregulating miR-302a. Clin Transl Med. 11:e4242021. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Wang X, Ji Y, Feng P, Liu R, Li G, Zheng
J, Xue Y, Wei Y, Ji C, Chen D and Li J: The m6A reader IGF2BP2
regulates macrophage phenotypic activation and inflammatory
diseases by stabilizing TSC1 and PPAR γ. Adv Sci (Weinh).
8:21002092021. View Article : Google Scholar
|
|
116
|
Guo M, Yan R, Ji Q, Yao H, Sun M, Duan L,
Xue Z and Jia Y: IFN regulatory factor-1 induced macrophage
pyroptosis by modulating m6A modification of circ_0029589 in
patients with acute coronary syndrome. Int Immunopharmacol.
86:1068002020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Shu B, Zhou YX, Li H, Zhang RZ, He C and
Yang X: The METTL3/MALAT1/PTBP1/USP8/TAK1 axis promotes pyroptosis
and M1 polarization of macrophages and contributes to liver
fibrosis. Cell Death Discov. 7:3682021. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Zhao W, Wang Z, Sun Z, He Y, Jian D, Hu X,
Zhang W and Zheng L: RNA helicase DDX5 participates in
oxLDL-induced macrophage scavenger receptor 1 expression by
suppressing mRNA degradation. Exp Cell Res. 366:114–120. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Park MH, Jeong E and Choudhury M:
Mono-(2-Ethylhexyl) phthalate regulates cholesterol efflux via
MicroRNAs regulated m6A RNA methylation. Chem Res
Toxicol. 33:461–469. 2020. View Article : Google Scholar
|
|
120
|
Kratz M, Coats BR, Hisert KB, Hagman D,
Mutskov V, Peris E, Schoenfelt KQ, Kuzma JN, Larson I, Billing PS,
et al: Metabolic dysfunction drives a mechanistically distinct
proinflammatory phenotype in adipose tissue macrophages. Cell
Metab. 20:614–625. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Gao X, Salomon C and Freeman DJ:
Extracellular vesicles from adipose tissue-a potential role in
obesity and type 2 diabetes? Front Endocrinol (Lausanne).
8:2022017. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Pan Y, Hui X, Hoo RLC, Ye D, Chan CYC,
Feng T, Wang Y, Lam KSL and Xu A: Adipocyte-secreted exosomal
microRNA-34a inhibits M2 macrophage polarization to promote
obesity-induced adipose inflammation. J Clin Invest. 129:834–849.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Zhang Y, Mei H, Chang X, Chen F, Zhu Y and
Han X: Adipocyte-derived microvesicles from obese mice induce M1
macrophage phenotype through secreted miR-155. J Mol Cell Biol.
8:505–517. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Caesar R, Reigstad CS, Bäckhed HK,
Reinhardt C, Ketonen M, Lundén GÖ, Cani PD and Bäckhed F:
Gut-derived lipopolysaccharide augments adipose macrophage
accumulation but is not essential for impaired glucose or insulin
tolerance in mice. Gut. 61:1701–1707. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Dong J, Dong Y, Dong Y, Chen F, Mitch WE
and Zhang L: Inhibition of myostatin in mice improves insulin
sensitivity via irisin-mediated cross talk between muscle and
adipose tissues. Int J Obes (Lond). 40:434–442. 2016. View Article : Google Scholar :
|
|
126
|
Liu Y, Wang X, Huang M, Luo A, Liu S, Cai
M, Li W, Yuan S, Zheng Z, Liu X and Tang C: METTL3 facilitates
kidney injury through promoting IRF4-mediated plasma cell
infiltration via an m6A-dependent manner in systemic lupus
erythematosus. BMC Med. 22:5112024. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Zhou B, Liu C, Xu L, Yuan Y, Zhao J, Zhao
W, Chen Y, Qiu J, Meng M, Zheng Y, et al:
N6-methyladenosine reader protein YT521-B homology
domain-containing 2 suppresses liver steatosis by regulation of
mRNA stability of lipogenic genes. Hepatology. 73:91–103. 2021.
View Article : Google Scholar
|
|
128
|
Sanyal AJ, Brunt EM, Kleiner DE, Kowdley
KV, Chalasani N, Lavine JE, Ratziu V and McCullough A: Endpoints
and clinical trial design for nonalcoholic steatohepatitis.
Hepatology. 54:344–353. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Armstrong MJ, Adams LA, Canbay A and Syn
WK: Extrahepatic complications of nonalcoholic fatty liver disease.
Hepatology. 59:1174–1197. 2014. View Article : Google Scholar
|
|
130
|
Sayiner M, Koenig A, Henry L and Younossi
ZM: Epidemiology of nonalcoholic fatty liver disease and
nonalcoholic steatohepatitis in the united states and the rest of
the world. Clin Liver Dis. 20:205–214. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Younossi ZM, Koenig AB, Abdelatif D, Fazel
Y, Henry L and Wymer M: Global epidemiology of nonalcoholic fatty
liver disease-meta-analytic assessment of prevalence, incidence,
and outcomes. Hepatology. 64:73–84. 2016. View Article : Google Scholar
|
|
132
|
Younossi ZM, Blissett D, Blissett R, Henry
L, Stepanova M, Younossi Y, Racila A, Hunt S and Beckerman R: The
economic and clinical burden of nonalcoholic fatty liver disease in
the United States and Europe. Hepatology. 64:1577–1586. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Mittal S, El-Serag HB, Sada YH, Kanwal F,
Duan Z, Temple S, May SB, Kramer JR, Richardson PA and Davila JA:
Hepatocellular carcinoma in the absence of cirrhosis in united
states veterans is associated with nonalcoholic fatty liver
disease. Clin Gastroenterol Hepatol. 14:124–131.e1. 2016.
View Article : Google Scholar
|
|
134
|
Dyson J, Jaques B, Chattopadyhay D, Lochan
R, Graham J, Das D, Aslam T, Patanwala I, Gaggar S, Cole M, et al:
Hepatocellular cancer: The impact of obesity, type 2 diabetes and a
multidisciplinary team. J Hepatol. 60:110–117. 2014. View Article : Google Scholar
|
|
135
|
Newsome PN, Buchholtz K, Cusi K, Linder M,
Okanoue T, Ratziu V, Sanyal AJ, Sejling AS and Harrison SA;
NN9931-4296 Investigators: A placebo-controlled trial of
subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J
Med. 384:1113–1124. 2021. View Article : Google Scholar
|
|
136
|
Newsome PN, Sejling AS and Sanyal AJ:
Semaglutide or placebo for nonalcoholic steatohepatitis. Reply. N
Engl J Med. 385:e62021.PubMed/NCBI
|
|
137
|
Harrison SA and Taub R: A phase 3 trial of
resmetirom in NASH with liver fibrosis. Reply. N Engl J Med.
390:1632–1633. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Harrison SA, Bedossa P, Guy CD,
Schattenberg JM, Loomba R, Taub R, Labriola D, Moussa SE, Neff GW,
Rinella ME, et al: A phase 3, randomized, controlled trial of
resmetirom in NASH with liver fibrosis. N Engl J Med. 390:497–509.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Younossi ZM, Ratziu V, Loomba R, Rinella
M, Anstee QM, Goodman Z, Bedossa P, Geier A, Beckebaum S, Newsome
PN, et al: Obeticholic acid for the treatment of non-alcoholic
steatohepatitis: Interim analysis from a multicentre, randomised,
placebo-controlled phase 3 trial. Lancet. 394:2184–2196. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Burhans MS, Hagman DK, Kuzma JN, Schmidt
KA and Kratz M: Contribution of adipose tissue inflammation to the
development of type 2 diabetes mellitus. Compr Physiol. 9:1–58.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Auguet T, Bertran L, Binetti J, Aguilar C,
Martínez S, Sabench F, Lopez-Dupla JM, Porras JA, Riesco D, Del
Castillo D and Richart C: Relationship between IL-8 circulating
levels and TLR2 hepatic expression in women with morbid obesity and
nonalcoholic steatohepatitis. Int J Mol Sci. 21:41892020.
View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Ponziani FR, Bhoori S, Castelli C,
Putignani L, Rivoltini L, Del Chierico F, Sanguinetti M, Morelli D,
Paroni Sterbini F, Petito V, et al: Hepatocellular carcinoma is
associated with gut microbiota profile and inflammation in
nonalcoholic fatty liver disease. Hepatology. 69:107–120. 2019.
View Article : Google Scholar
|
|
143
|
Duan Y, Pan X, Luo J, Xiao X, Li J,
Bestman PL and Luo M: Association of inflammatory cytokines with
non-alcoholic fatty liver disease. Front Immunol. 13:8802982022.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Méndez-García LA, Trejo-Millán F,
Martínez-Reyes CP, Manjarrez-Reyna AN, Esquivel-Velázquez M,
Melendez-Mier G, Islas-Andrade S, Rojas-Bernabé A, Kzhyshkowska J
and Escobedo G: Infliximab ameliorates tumor necrosis
factor-alpha-induced insulin resistance by attenuating PTP1B
activation in 3T3L1 adipocytes in vitro. Scand J Immunol.
88:e127162018. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Zhang Q, Wang J, Huang F, Yao Y and Xu L:
Leptin induces NAFLD progression through infiltrated CD8+ T
lymphocytes mediating pyroptotic-like cell death of hepatocytes and
macrophages. Dig Liver Dis. 53:598–605. 2021. View Article : Google Scholar
|
|
146
|
Yamauchi T, Nio Y, Maki T, Kobayashi M,
Takazawa T, Iwabu M, Okada-Iwabu M, Kawamoto S, Kubota N, Kubota T,
et al: Targeted disruption of AdipoR1 and AdipoR2 causes abrogation
of adiponectin binding and metabolic actions. Nat Med. 13:332–339.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
147
|
Balmer ML, Joneli J, Schoepfer A, Stickel
F, Thormann W and Dufour JF: Significance of serum adiponectin
levels in patients with chronic liver disease. Clin Sci (Lond).
119:431–436. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Luo Y, Zhang Z, Xiang L, Zhou B, Wang X,
Lin Y, Ding X, Liu F, Lu Y and Peng Y: Analysis of
N6-methyladenosine methylation modification in fructose-induced
non-alcoholic fatty liver disease. Front Endocrinol (Lausanne).
12:7806172021. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Peng Z, Gong Y, Wang X, He W, Wu L, Zhang
L, Xiong L, Huang Y, Su L, Shi P, et al:
METTL3-m6A-Rubicon axis inhibits autophagy in
nonalcoholic fatty liver disease. Mol Ther. 30:932–946. 2022.
View Article : Google Scholar
|
|
150
|
Chen A, Chen X, Cheng S, Shu L, Yan M, Yao
L, Wang B, Huang S, Zhou L, Yang Z and Liu G: FTO promotes SREBP1c
maturation and enhances CIDEC transcription during lipid
accumulation in HepG2 cells. Biochim Biophys Acta Mol Cell Biol
Lipids. 1863:538–548. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Tang Z, Sun C, Yan Y, Niu Z, Li Y, Xu X,
Zhang J, Wu Y, Li Y, Wang L, et al: Aberrant elevation of FTO
levels promotes liver steatosis by decreasing the m6A methylation
and increasing the stability of SREBF1 and ChREBP mRNAs. J Mol Cell
Biol. 14:mjac0612023. View Article : Google Scholar :
|
|
152
|
Wang H, Wang Y, Lai S, Zhao L, Liu W, Liu
S, Chen H, Wang J, Du G and Tang B: LINC01468 drives NAFLD-HCC
progression through CUL4A-linked degradation of SHIP2. Cell Death
Discov. 8:4492022. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Liu Y, Sun L, Guo H, Zhou S, Wang C, Ji C,
Meng F, Liang S, Zhang B, Yuan Y, et al: Targeting SLP2-mediated
lipid metabolism reprograming restricts proliferation and
metastasis of hepatocellular carcinoma and promotes sensitivity to
Lenvatinib. Oncogene. 42:374–388. 2023. View Article : Google Scholar
|
|
154
|
Chen L, Xia S, Wang F, Zhou Y, Wang S,
Yang T, Li Y, Xu M, Zhou Y, Kong D, et al: m6A
methylation-induced NR1D1 ablation disrupts the HSC circadian clock
and promotes hepatic fibrosis. Pharmacol Res. 189:1067042023.
View Article : Google Scholar
|
|
155
|
Saeedi P, Petersohn I, Salpea P, Malanda
B, Karuranga S, Unwin N, Colagiuri S, Guariguata L, Motala AA,
Ogurtsova K, et al: Global and regional diabetes prevalence
estimates for 2019 and projections for 2030 and 2045: Results from
the international diabetes federation diabetes atlas, 9th edition.
Diabetes Res Clin Pract. 157:1078432019. View Article : Google Scholar
|
|
156
|
Holman N, Young B and Gadsby R: Current
prevalence of type 1 and type 2 diabetes in adults and children in
the UK. Diabet Med. 32:1119–1120. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Bruno G, Runzo C, Cavallo-Perin P,
Merletti F, Rivetti M, Pinach S, Novelli G, Trovati M, Cerutti F
and Pagano G; Piedmont Study Group for Diabetes Epidemiology:
Incidence of type 1 and type 2 diabetes in adults aged 30-49 years:
The population-based registry in the province of Turin, Italy.
Diabetes Care. 28:2613–2619. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Chatterjee S, Khunti K and Davies MJ: Type
2 diabetes. Lancet. 389:2239–2251. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Siersbaek R, Nielsen R and Mandrup S:
PPARgamma in adipocyte differentiation and metabolism-novel
insights from genome-wide studies. FEBS Lett. 584:3242–3249. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Chen C, Xiang Q, Liu W, Liang S, Yang M
and Tao J: Co-expression network revealed roles of RNA
m6A methylation in human β-Cell of type 2 diabetes
mellitus. Front Cell Dev Biol. 9:6511422021. View Article : Google Scholar
|
|
161
|
Wu X, Wang W, Fan S, You L, Li F, Zhang X,
Wu H, Tang J, Qi Y, Feng W, et al: U-shaped association between
serum IGF2BP3 and T2DM: A cross-sectional study in Chinese
population. J Diabetes. 15:349–361. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Xiao L, De Jesus DF, Ju CW, Wei JB, Hu J,
DiStefano-Forti A, Tsuji T, Cero C, Männistö V, Manninen SM, et al:
m6A mRNA methylation in brown fat regulates systemic
insulin sensitivity via an inter-organ prostaglandin signaling axis
independent of UCP1. Cell Metab. 36:2207–2227.e9. 2024. View Article : Google Scholar
|
|
163
|
Huang X, He W, Fan S, Li H and Ye G:
IGF2BP3-mediated enhanced stability of MYLK represses MSC
adipogenesis and alleviates obesity and insulin resistance in HFD
mice. Cell Mol Life Sci. 81:172024. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Hotamisligil GS: Inflammation and
metabolic disorders. Nature. 444:860–867. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Shoelson SE, Lee J and Goldfine AB:
Inflammation and insulin resistance. J Clin Invest. 116:1793–1801.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Liu ZY, Lin LC, Liu ZY, Song K, Tu B, Sun
H, Zhou Y, Mao S, Zhang Y, Li R, et al:
N6-Methyladenosine-mediated phase separation suppresses
NOTCH1 expression and promotes mitochondrial fission in diabetic
cardiac fibrosis. Cardiovasc Diabetol. 23:3472024. View Article : Google Scholar
|
|
167
|
Roberts TC, Langer R and Wood MJA:
Advances in oligonucleotide drug delivery. Nat Rev Drug Discov.
19:673–694. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Nuñez JK, Chen J, Pommier GC, Cogan JZ,
Replogle JM, Adriaens C, Ramadoss GN, Shi Q, Hung KL, Samelson AJ,
et al: Genome-wide programmable transcriptional memory by
CRISPR-based epigenome editing. Cell. 184:2503–2519.e17. 2021.
View Article : Google Scholar : PubMed/NCBI
|