The Spt-Ada-Gcn5-acetyltransferase (SAGA) complex, a
highly conserved 19-subunit machinery, spans evolutionary lineages
from yeast to mammals (1,2).
It harbors a tetrameric deubiquitinase module (DUBm) composed of
ubiquitin-specific peptidase 22 (USP22), Ataxin 7/SAGA-associated
factor 73 (ATXN7/Sgf73), Ataxin 7-like protein 3 (ATXN7L3/Sgf11)
and ENY2 transcription/export complex subunit, which together form
a tightly associated tetrameric DUBm (3,4).
The DUBm is an independently folding subcomplex that connects to
the rest of the SAGA via the C-terminal portion of Sgf73 (3). As a core component of SAGA, USP22
belongs to the ubiquitin-specific processing proteases (USPs); they
make up the largest deubiquitinase subfamily and are known in yeast
as UBPs (5). Structurally, USP22
features an N-terminal zinc finger domain and a C-terminal
catalytic domain, which enable its enzymatic activity (6).
USP22 exerts robust protumorigenic effects across
various malignancies by orchestrating key cellular processes
(5,7). In patients with non-small cell lung
cancer, USP22 promotes cell proliferation, invasion and stemness
via murine double minute X/p53 pathway (8) inducing epithelial-mesenchymal
transition (EMT) (9) and
stabilizing murine double minute 2 to suppress ferroptosis
(10). Ferroptosis is an iron
dependent, nonapoptotic form of cell death driven by membrane lipid
peroxidation (11). In patients
with hepatocellular carcinoma, it facilitates tumor progression and
drug resistance by stabilizing E2F transcription factor 6 (E2F6) to
activate RAC-alpha serine/threonine-protein kinase (AKT) signaling
(12), maintaining peroxisome
proliferator-activated receptor γ (PPARγ)-mediated lipid anabolism
(13), and engaging the silent
mating-type information regulation 2 homologue 1
(SIRT1)/AKT/multidrug resistance-associated protein 1 (MRP1) axis
(14). In patients with prostate
cancer, USP22 functions as an oncogenic driver by modulating cell
proliferation and DNA damage repair (15) In patients with breast cancer, it
enhances human epidermal growth factor receptor 2-driven invasive
phenotypes by inhibiting the unfolded protein response (16). Additionally, in patients with
colorectal cancer, USP22 promotes programmed death-ligand 1 (PD-L1)
stability through deubiquitination (17), whereas in those with
osteosarcoma, its upregulation is associated with tumor progression
(18). Mechanistically, it also
regulates necroptosis by modulating receptor-interacting protein
kinase 3 stability (19).
Notably, while substantial research has focused on
the role of USP22 in cancer stemness (20), emerging evidence has revealed its
critical functions in nontumor pathologies. However, investigations
into physiologic and pathogenic processes across various cell
types, tissues and organ systems, remain fragmented, necessitating
a comprehensive synthesis of its involvement in disease a etiology,
functional roles and regulatory networks. Therefore, the present
study reviewed the roles and regulatory mechanisms of USP22 across
diverse biological processes and pathological conditions, including
embryonic survival, skin wound healing, cellular and tissue damage
and repair following ischemia-reperfusion, inflammatory and immune
regulation, fibrosis and tissue remodeling. The detailed substrates
of USP22 involved therein and their functions are summarized in
Tables I-IV.
The SAGA complex harbors two enzymatic activities:
the general control nonderepressible 5 (Gcn5) histone
acetyltransferase and the USP22 deubiquitinase (21). Previous studies have shown that
loss of GCN5 affects early embryogenesis, neural stem cell
differentiation, retinoic acid signaling and fibroblast growth
factor signaling (22,23). Additionally, USP22 is critical
for regulating the differentiation of embryonic stem cells
(24,25). Studies have shown that mice
carrying a hypomorphic allele of Usp22 exhibit reduced body
size (26) whereas Usp22
deletion leads to embryonic lethality in mice (27). This is mainly because USP22
exerts a deubiquitinating effect to stabilize the SIRT1 protein in
embryonic cells, thereby inhibiting p53 acetylation-induced
apoptosis and maintaining embryonic cell survival (27). Placental blood vessels also play
a crucial role in embryonic development; abnormal placental
vascular development and vascular dysfunction may result in
uteroplacental perfusion disorders, intrauterine malnutrition and
fetal growth restriction (28,29). Fetal growth restriction (FGR)
refers to the failure of a fetus to achieve its growth potential in
the uterus and is a common obstetric complication that can lead to
various adverse outcomes (30,31). USP22 is essential for placental
angiogenesis and embryonic survival because it regulates the
functions of endothelial cells and pericytes. USP22 expression is
downregulated in the placental tissues of patients with FGR,
leading to inhibited proliferation, increased apoptosis and
impaired angiogenesis in human umbilical vein endothelial cells and
its mechanism may involve downregulation of the PI3K/AKT signaling
pathway (28). Another study
indicated that Usp22 deletion causes placental vascular
development defects and subsequent early embryonic mortality, which
is associated with impaired TGF-β receptor and RTK signaling
pathways (32). The
aforementioned mechanisms involving USP22 collectively contribute
to poor embryonic development or even mortality, revealing the
multiple protective roles of USP22 in embryonic development.
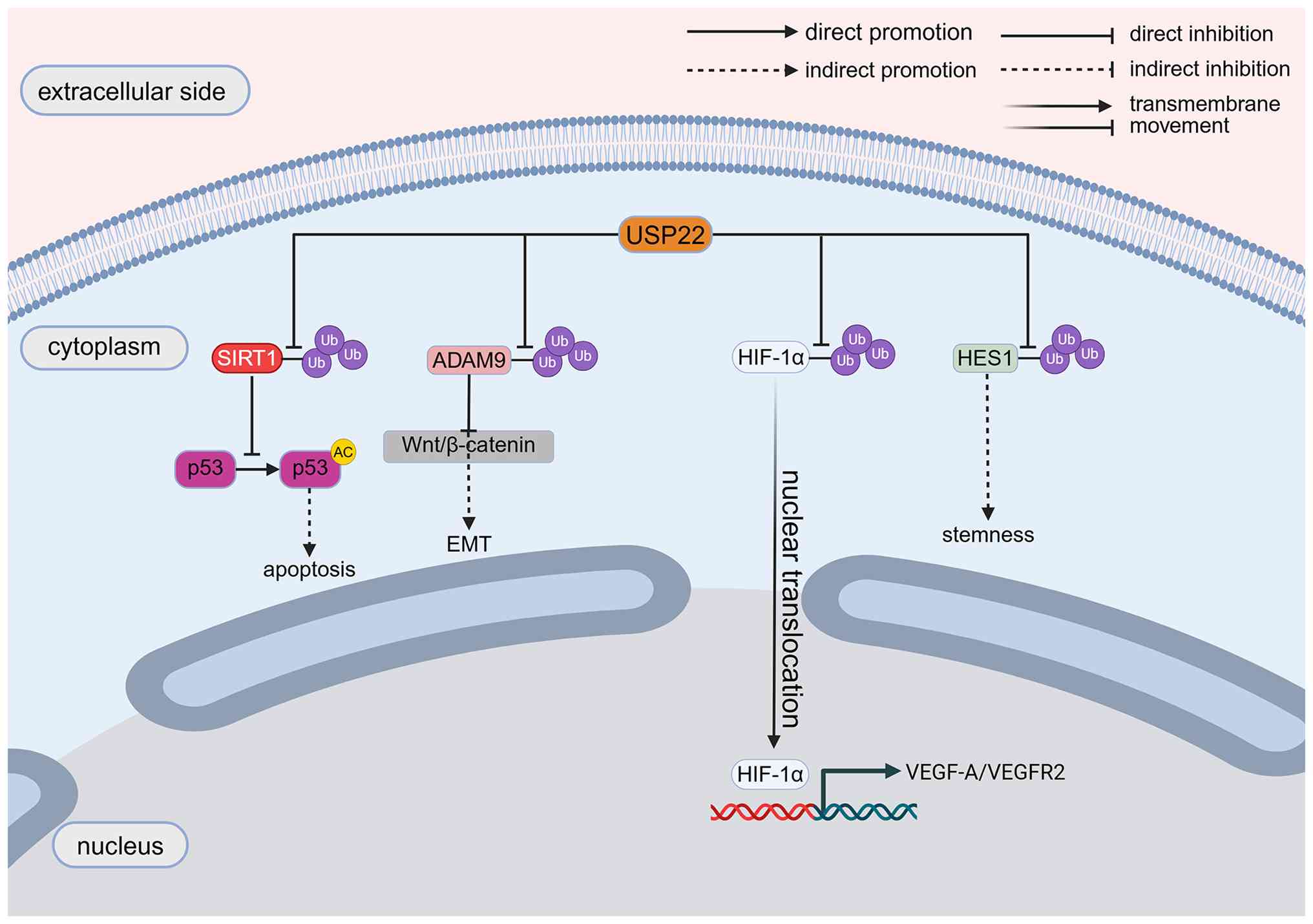
In contrast to that in FGR, upregulated USP22 in
placental trophoblasts from patients with preeclampsia (PE)
deubiquitinates and stabilizes a disintegrin and metalloprotease 9
(ADAM9), thereby inhibiting the activity of the Wnt/β-catenin
pathway. This suppresses the proliferation, migration, invasion and
EMT of trophoblast cells and induces their apoptosis. This process
may cause shallow trophoblast invasion and spiral artery rupture,
leading to PE (33) (Table I; Fig. 1).
Although existing studies have revealed the multiple
roles and related pathways of USP22 in embryonic survival,
placental angiogenesis and pregnancy complications (FGR, PE),
significant research gaps remain. The expression patterns and
functional outcomes of USP22 differ between FGR and PE and further
investigation is needed to clarify the regulatory mechanisms
underlying these divergent phenotypes in different cell types, as
well as their potential associations. The mouse models and
single-cell lines used in recent studies cannot fully replicate the
complexity of human placental development; additionally,
mechanistic research has not effectively linked clinical diagnostic
biomarkers and targeted intervention strategies and the crosstalk
between various regulatory pathways remains to be further
elucidated. These issues provide key directions for future
research.
USP22 sustains neural progenitor/stem cell (NSC)
pluripotency by prolonging the half-life of the hairy and enhancer
of split 1 (Hes1), after which USP22 expression knockdown alters
the abundance of cells but not the shape of the small intestine,
indicating that USP22 not only preserves the number of stem cells
but also modulates cell differentiation.
NSCs residing in the adult mammalian brain
contribute to neurodevelopment and repair via proliferative and
differentiation functions (34,35), with the dynamic Hes1 gene
expression playing a pivotal role in normal developmental timing
and fate determination (36,37). USP22 interacts with the
transcription factor Hes1 to prolong its half-life and inhibit
neuronal differentiation while maintaining cell stemness by
sustaining the periodic oscillation of Hes1 (38). In patients with traumatic brain
injury (TBI), overexpression of USP22 promotes the proliferation of
NSCs in the hippocampal dentate gyrus region and inhibits their
differentiation into mature neurons, thereby improving
hippocampus-dependent cognitive impairment. USP22 is expected to be
a potential therapeutic target for post-TBI treatment (39). Proper regulation of gene
expression patterns serves a vital role in determining cell fate
and regulating cell differentiation (40-42). Usp22 is widely expressed in most
embryonic tissues at embryonic Day 15.5 (E15.5). Systemic knockdown
of Usp22 expression does not affect the overall morphology of the
mouse small intestine or the number of Olfactomedin4-positive
(Olfm4+) stem cells but rather causes changes in the
abundance of specific differentiated cell populations (goblet cells
and Paneth cells) (26). These
findings indicate that USP22 primarily affects the differentiation
stage rather than the size of the stem cell pool and that these
changes are independent of H2B monoubiquitination (Table I; Fig. 1).
Currently, research on the regulation of stem cell
function by USP22 remains notably inadequate. Hes1 is a key gene
related to intestinal epithelial cell proliferation and
differentiation and is highly expressed in stem cells and
progenitor cells (43). However,
the finding that USP22 expression knockdown does not affect the
number of intestinal stem cells contradicts their regulatory
relationship in NSCs. This discrepancy may be associated with
differences in the basal expression of USP22 or Hes1 in different
cell types; the underlying tissue-specific regulatory network
crosstalk still requires further elucidation.
Cutaneous wounds secondary to traumatic injuries,
thermal burns, surgical procedures and diabetic complications
constitute common clinical entities (44). Mesenchymal stem cells derived
from adipose tissue are referred to as adipose-derived stem cells
(ADSCs) (45). Under hypoxic
conditions, USP22 in ADSCs and their exosomes is transferred to
vascular endothelial cells, where it directly interacts with and
stabilizes hypoxia inducible factor 1 subunit alpha (HIF-1α)
protein, promoting its nuclear translocation to activate the
downstream vascular endothelial growth factor A (VEGF-A)/vascular
endothelial growth factor receptor 2 (VEGFR2) pathway (Fig. 1), thereby enhancing angiogenesis
and collagen synthesis (46).
This mechanism reveals a novel strategy targeting the
USP22/HIF-1α/VEGF axis for trauma repair (47). Notably, investigating the
relationship between the homeostatic maintenance of this process
and scarring represents an intriguing research direction (Table I; Fig. 1).
The regulatory role of USP22 in cell death differs
across organs: In renal podocytes, upregulated USP22 expression
exacerbates apoptosis and inflammatory responses by interacting
with SIRT1 or stabilizing high mobility group box 1 (HMGB1);
whereas in pancreatic β-cells, downregulated USP22 expression
triggers ubiquitin-mediated degradation of SIRT1, activating
mitochondrial apoptosis and ferroptosis pathways.
Glomerular podocytes are indispensable for
maintaining the structural and functional integrity of the
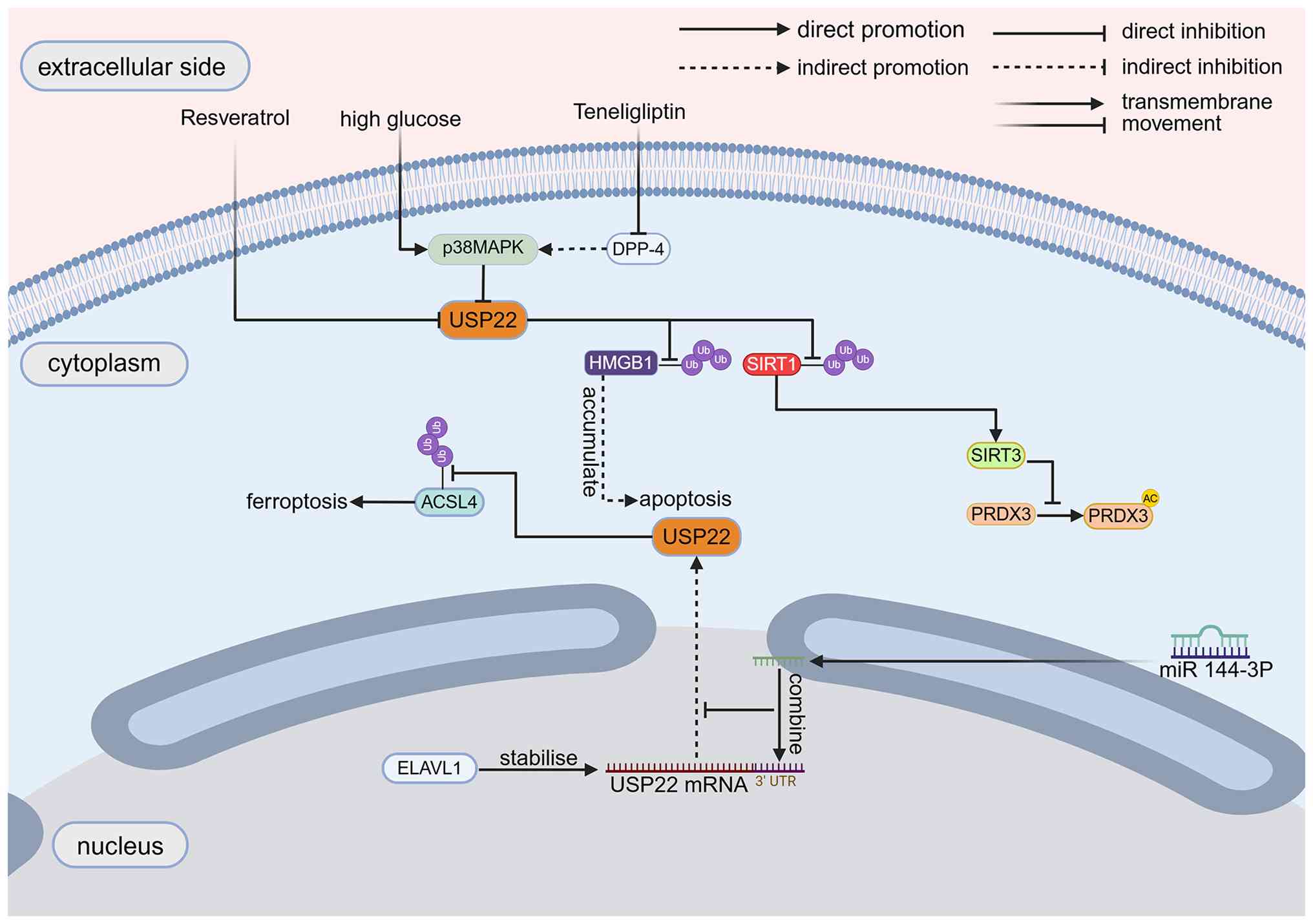
glomerular filtration barrier (48). In high-glucose environments,
Usp22 expression is spontaneously upregulated in podocytes,
promoting podocyte depletion, whereas Usp22 expression knockdown
protects podocytes from high-glucose induced injury (49). Resveratrol mitigates the
progression of diabetic nephropathy by inhibiting USP22 expression,
which may be associated with increased SIRT1 expression (27,50). Findings from another study
(51) may help elucidate the
underlying cause of this phenomenon: Under high-glucose conditions,
embryonic lethal abnormal vision-like 1 (ELAVL1) binds to and
stabilizes USP22 mRNA, thereby upregulating USP22 protein
expression. USP22 subsequently inhibits the proteasomal degradation
of acyl-CoA synthetase long-chain family member 4 (ACSL4) through
deubiquitination. Excessive ACSL4 expression promotes intracellular
lipid peroxidation in podocytes, ultimately triggering ferroptosis
(51). HMGB1, a
damage-associated molecular pattern, is sensed by Toll-like
receptors (TLRs) (52). In
patients with hypertensive nephropathy (HN), USP22 is significantly
upregulated in podocytes, leading to HMGB1 deubiquitination,
thereby inhibiting its proteasomal degradation. The accumulation of
HMGB1 promotes apoptosis and inflammatory factor release and
activates inflammatory pathways in renal tubular epithelial cells
via the paracrine effect of podocytes, further inducing renal
injury (53).
In the pancreas of patients with type 2 diabetes
mellitus, inflammatory, senescent and oxidative stress processes
may coexist (54). Given the
weak antioxidant defense of pancreatic islet β-cells, chronic
hyperglycemic exposure triggers oxidative stress-mediated β-cell
dysfunction (55,56). Hyperglycemia-induced p38
mitogen-activated protein kinase (p38MAPK) hyperactivation in
β-cells downregulates USP22 expression, promoting mitochondrial
superoxide accumulation via the SIRT1/SIRT3/peroxiredoxin 3 (PRDX3)
axis to elicit β cell apoptosis through NADPH oxidase (NOX)/c-Jun
N-terminal kinase (JNK)/66 kDa src homology and collagen
domain-containing protein (p66Shc) signalosome activation. The
dipeptidyl peptidase-4 (DPP-4) inhibitor teneligliptin protects
pancreatic islet β-cells by suppressing p38MAPK to upregulate USP22
expression (57). Additionally,
lipid peroxidation induces ferroptosis (58) via miR-144-3p: This miRNA
upregulates and targets the USP22 3' UTR, promoting SIRT1
proteasomal degradation to activate the ferroptotic pathway
(59). Therefore, USP22/SIRT1
clearly serves a crucial roles in the functional maintenance and
cell survival of pancreatic islet β-cells (Table II; Fig. 2).
These bidirectional effects in podocytes and
β-cells, mediated by tissue-specific stress signals and
USP22-mediated differential regulation of core targets,
collectively highlight USP22's tissue-dependent function and the
essential role of SIRT1 deubiquitination in cell survival.
I/R injury constitutes an inevitable form of damage
incurred during revascularization and is commonly encountered in
clinical scenarios such as trauma, arterial thrombotic or embolic
disorders, shock, major surgical procedures and organ
transplantation (60-63). USP22 serves a pivotal regulatory
role in I/R injury, mediating pathophysiology in cardiac,
intestinal and cerebral tissues via distinct molecular mechanisms.
Its dysregulated expression and function are intimately linked to
cell death and cell cycle control, positioning it as a potential
therapeutic target for ischemic diseases.
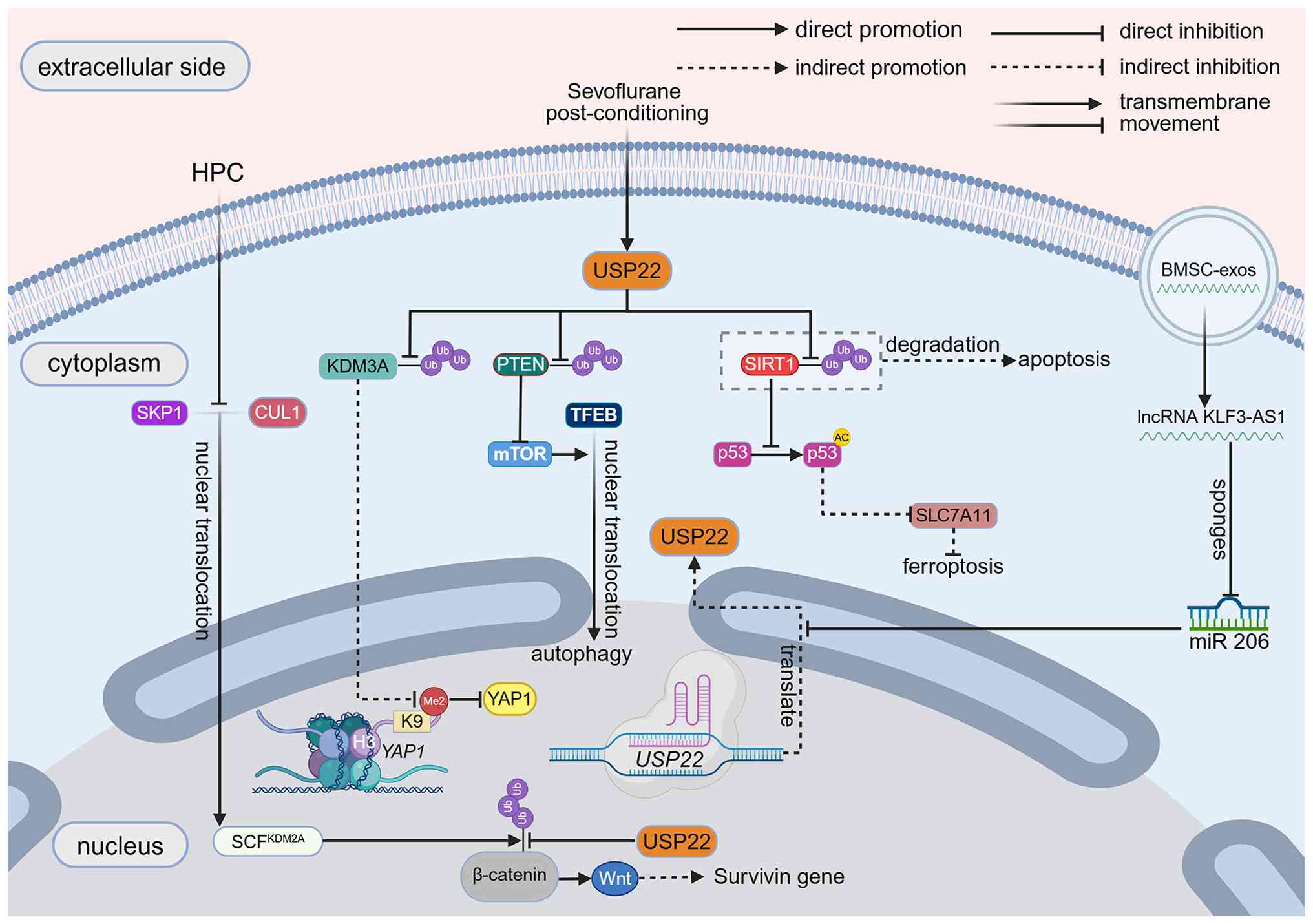
Myocardial I/R (MI/R) injury is a critical
determinant of postinfarction complications (64), involving core mechanisms such as
ferroptosis, oxidative stress and apoptosis (65-67). USP22 deubiquitinates and
stabilizes SIRT1, reducing p53 protein expression and acetylation
levels to relieve the transcriptional repression of solute carrier
family 7 member 11 (SLC7A11), thereby decreasing iron-dependent
lipid peroxidation and cardiomyocyte ferroptosis (68). Additionally, sevoflurane
postconditioning upregulates USP22 expression to stabilize
lysine-specific demethylase 3A (KDM3A), reducing H3K9me2
methylation at the Yes-associated protein 1 (YAP1) promoter to
promote YAP1 transcription, thus mitigating cardiomyocyte apoptosis
and improving cardiac function (69). These studies reveal that USP22
serves dual protective roles in MI/R injury by regulating the
SIRT1/p53/SLC7A11 and KDM3A/YAP1 axes (70,71).
Intestinal I/R lesions are mechanisms that maintain
a number of diseases and are early factors in systemic inflammatory
response systems, in clinical practice dysfunction syndromes of
various organs are associated with high mortality rates (72-74). During the 0-24 h post-I/R period,
USP22 expression reaches a nadir at 6 h and is positively
associated with the G1-phase marker cyclin D1 and
proliferation marker proliferating cell nuclear antigen (PCNA).
USP22 deletion impairs post-I/R cell proliferation, induced
G1 arrest and blocked S-phase entry, whereas USP22
overexpression reverse these phenotypes (20).
Acute ischemic stroke is a major health hazard that
causes physical disability, cognitive impairment and even mortality
and affects millions of individuals worldwide each year (75,76). However, the available treatment
strategies for stroke are limited (77). Therefore, identifying potential
brain protectors or initiating endogenous neuroprotective
mechanisms against acute cerebral ischemia is highly important. The
long non-coding (lnc)RNA KLF3-AS1 carried by bone marrow
mesenchymal stem cell-derived exosomes (BMSC-exos) sponges microRNA
(miR)-206 to upregulate USP22 mRNA expression. It subsequently
stabilizes the SIRT1 protein via its deubiquitinating activity,
alleviates cerebral I/R injury through the anti-inflammatory and
antioxidant effects of SIRT1 and promotes neurological function
recovery (78). In contrast to
the aforementioned neuroprotective role of USP22 after I/R, USP22
is highly expressed in cerebral I/R injury. It stabilizes the
phosphatase and tensin homolog (PTEN) protein through direct
interaction and deubiquitination, inhibits the mTOR/TFEB pathway
and promotes excessive autophagy and oxidative stress (79). The contradictory role of USP22 in
cerebral I/R injury essentially reflects multidimensional-dependent
functional heterogeneity; its function is not inherently determined
but is jointly regulated by substrate specificity, cell type,
ischemic time window, microenvironmental signals, expression level
and subcellular localization (80). Briefly, during cerebral I/R
injury, USP22 has distinct effects on microglia and neurons. This
contradiction also suggests that USP22 is not merely a 'protective
factor' or 'damaging factor' but rather a 'hub molecule in the
signaling network' in cerebral I/R injury whose functional
orientation depends on the dynamic needs of the pathological
microenvironment.
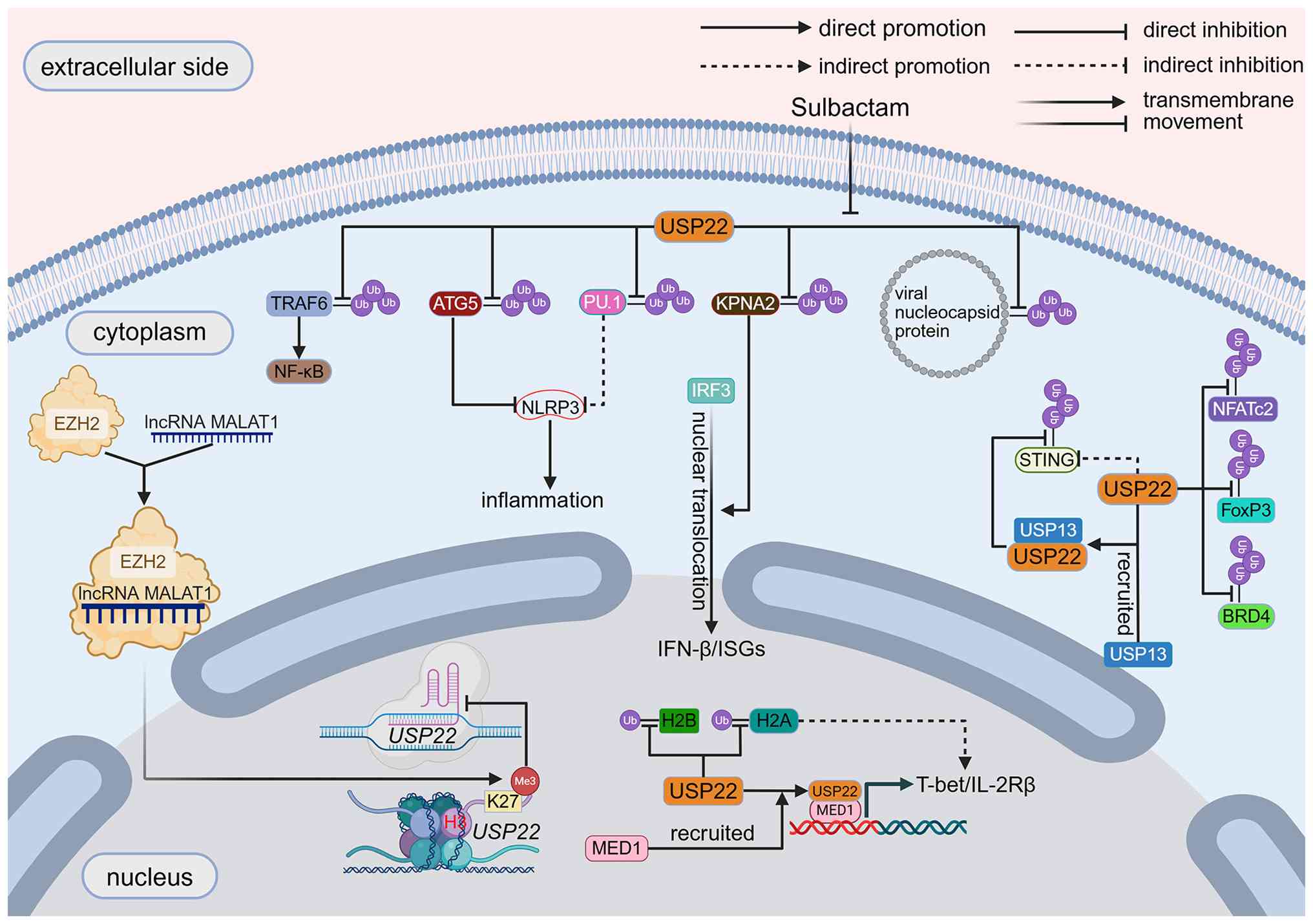
USP22 exerts paradoxical regulatory effects during
viral infections, whose function depends on the specific viral type
and the host cellular microenvironment. USP22 can regulate both
type I IFNs and type III interferon (IFN-λ) expression, indicating
that USP22 is a broad-spectrum antiviral regulator across
interferon types (Fig. 3).
In antiviral immunity, USP22 stabilizes karyopherin
subunit alpha 2 (KPNA2) to promote interferon regulatory factor 3
(IRF3) nuclear translocation, activating IFN-β and
interferon-stimulated genes for antiviral defense (83). Conversely, in severe acute
respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, USP22
deubiquitinates nucleocapsid protein (NP) to increase viral
replication. Sulbactam inhibits infection by downregulating USP22
expression and promoting ubiquitination-mediated degradation of NP
(84). USP22 expression knockout
in epithelial cells increases IFN-λ secretion and suppressing viral
replication (85). Thus, USP22
may contribute to the replication of certain viruses and serve as a
key node in the regulation of mucosal immunity regulation (Table III; Fig. 4).
In various disease models such as sepsis-induced
myocardial dysfunction, sepsis and keratitis, USP22 performs dual
regulatory functions and promotes inflammatory responses under
specific pathological conditions. Its specific role depends on
various factors including the cellular microenvironment, substrate
protein interactions and pathogen types.
Sepsis-induced myocardial dysfunction (SIMD) is
intimately linked to inflammation, oxidative stress and other
pathological processes (86).
During SIMD, the expression of the cardiac lncRNA,
metastasis-associated lung adenocarcinoma transcript 1 increases
significantly, promoting H3K27me3 methylation at the USP22 promoter
via enhancer of zeste 2 polycomb repressive complex 2 subunit
binding to repress USP22 expression. This exacerbates myocardial
oxidative stress, increases proinflammatory cytokines expression,
promotes apoptosis, reduces anti-inflammatory factors and worsens
cardiac function (87). In
sepsis models, USP22 deubiquitinates and stabilizes
autophagy-related 5 (ATG5) to promote autophagosome formation,
inhibit NLR family pyrin domain containing 3 (NLRP3) inflammasome
activation and lead to lysosomal degradation, thereby regulating
various inflammatory diseases (88). Notably, USP22 modulates NLRP3
expression via alternative mechanisms, such as indirectly
regulating NLRP3 expression in microglia by inhibiting Spi-1
proto-oncogene during neuroinflammation (88-90). This discovery enables novel
therapeutic strategies for NLRP3-associated diseases including
gout, atherosclerosis, type 2 diabetes and Alzheimer's disease
(88,91-93). USP22 suppresses inflammation not
only by reducing inflammasome activation but also by inhibiting the
stimulator of interferon response cGAMP interactor 1 (STING)
pathway to decrease inflammatory cytokine expression (94). In intestinal epithelial cells,
USP22 negatively regulates the STING pathway to suppress excessive
IFN-λ activation and maintain epithelial immune homeostasis
(85). Additionally, USP22
recruits ubiquitin-specific protease 13 (USP13) to remove
K27-linked ubiquitin chains from STING, further inhibiting aberrant
type I interferon activation and preventing excessive
immunopathological damage (90,95). Collectively, these findings
demonstrate the anti-inflammatory role of USP22.
Existing studies have mentioned only influencing
factors such as the cellular microenvironment and pathogen type,
but none of these studies have clarified how upstream signals alter
functional bias. Across various inflammatory models, it remains
unknown why USP22 selects ATG5, STING, TRAF6, or BRD4 as substrates
as well as the nature of the underlying substrate competition
mechanism and molecular switches.
USP22 serves multidimensional roles in immune cell
regulation, dynamically modulating emergency hematopoiesis
(100), microglial polarization
(89), T cell function (110-117) and macrophage lipid metabolism
(118-120) via epigenetic modification,
signaling pathway activation and transcription factor
stabilization. Its functional diversity, closely linked to
cell-type-specific substrate interactions, offers potential
therapeutic targets for immune-related diseases such as
autoimmunity and atherosclerosis.
Emergency hematopoiesis, a crisis state of the
immune system, serves two primary functions: i) Increasing blood
cell production to compensate for massive losses during
inflammation and infection (100) and ii) 'preactivating' immunity
to facilitate faster clearance of infectious agents (101). Pan-hematopoietic-specific
knockout of Usp22 expression increases H2Bub1 levels and activates
the expression of inflammation-related genes, myeloid-associated
transcription factors and mitochondrial respiratory genes to drive
progenitor proliferation and differentiation. This manifests as
systemic emergency hematopoiesis, significantly enhancing the
innate immune capacity against Listeria infection (102).
Alzheimer's disease (AD) represents the foremost
cause of neurodegenerative dementia on a global scale (103). Major depressive disorder is a
primary cause of physical and mental disability globally and
affects hundreds of millions of individuals (104). The two diseases are intricately
linked to microglia, which, as the first line of immune defense in
the central nervous system, elicit inflammation that is associated
with neurodegenerative diseases and depressive-like behaviors
(105). USP22 acts as a
deubiquitinase for PU.1, positively regulating PU.1 stability
(106) to promote microglial
polarization towards the M2 phenotype (89), a process involved in
AD-associated inflammatory responses (107,108). Similarly, USP22 can stabilize
its downstream substrate KAT2A through K48-linked deubiquitination,
thereby promoting microglial mitochondrial damage and oxidative
stress and exacerbating central inflammation and depressive-like
behaviors. A study identified the material basis underlying mental
disorders, providing a supplementary theoretical foundation for the
pharmacotherapy of mental illnesses (109).
USP22 exerts multifaceted functions in T cell
regulation, with these activities only beginning to be
characterized (110-114). In Jurkat T cells, USP22
directly associates with nuclear factor of activated T cells 2
(NFATc2) to sustain the protein, thereby augmenting the
transcription of interleukin-2 (IL-2) (110). In regulatory T cells (Tregs),
USP22 preserves immune homeostasis by maintaining forkhead box P3
(Foxp3) expression; Treg-specific USP22 knockout elicits autoimmune
predisposition, whereas Usp22 ablation alleviates Treg-mediated
suppression of antitumor immunity (102,115). In innate invariant natural
killer T (iNKT) cells, USP22 drives T-bet/IL-2Rβ expression and
iNKT maturation through H2A deubiquitination or interaction with
coactivator mediator complex subunit 1 (116). Furthermore, USP22 sustains
PD-L1 expression to curb excessive CD8+ T cell activation; and
USP22 deficiency exacerbates T cell-mediated immunopathological
injury (117).
Macrophage internalization of oxidized low-density
lipoprotein to form foam cells constitutes a pathophysiological
signature of atherosclerosis (118). In macrophages, the
C1q/TNF-related protein 9-mediated USP22/SIRT1 axis hinders
atherosclerotic advancement by increasing autophagic flux,
decreasing lipid deposition and repressing foam cell
formation-related gene expression (119). USP22 is expressed at low levels
in macrophages in atherosclerotic lesions. Its overexpression can
deubiquitinate and stabilize PPARγ protein and upregulate the
expression of efferocytosis-related molecules, thereby inhibiting
foam cell formation and improving plaque stability. Conversely, the
application of USP22 inhibitors exacerbates the progression of
atherosclerosis (120). These
findings collectively suggest that USP22 may serve as a potential
therapeutic target for the treatment of atherosclerosis (Table III; Fig. 4).
USP22 serves pivotal regulatory roles in various
fibrotic disorders. In patients with diabetic kidney disease (DKD)
and HN, it contributes to renal fibrogenesis via distinct
mechanisms, including the inhibition of glomerular mesangial
fibrosis (121,122) and the promotion of
tubulointerstitial fibrosis (123) and podocyte apoptosis (53,106). In patients with pneumoconiosis,
USP22 facilitates EMT and extracellular matrix (ECM) deposition
(Fig. 4). These insights offer
potential therapeutic targets for treating fibrotic diseases
(124).
DKD, a major microvascular complication of diabetes,
is a leading cause of end-stage renal disease (125). In glomerular mesangial cells,
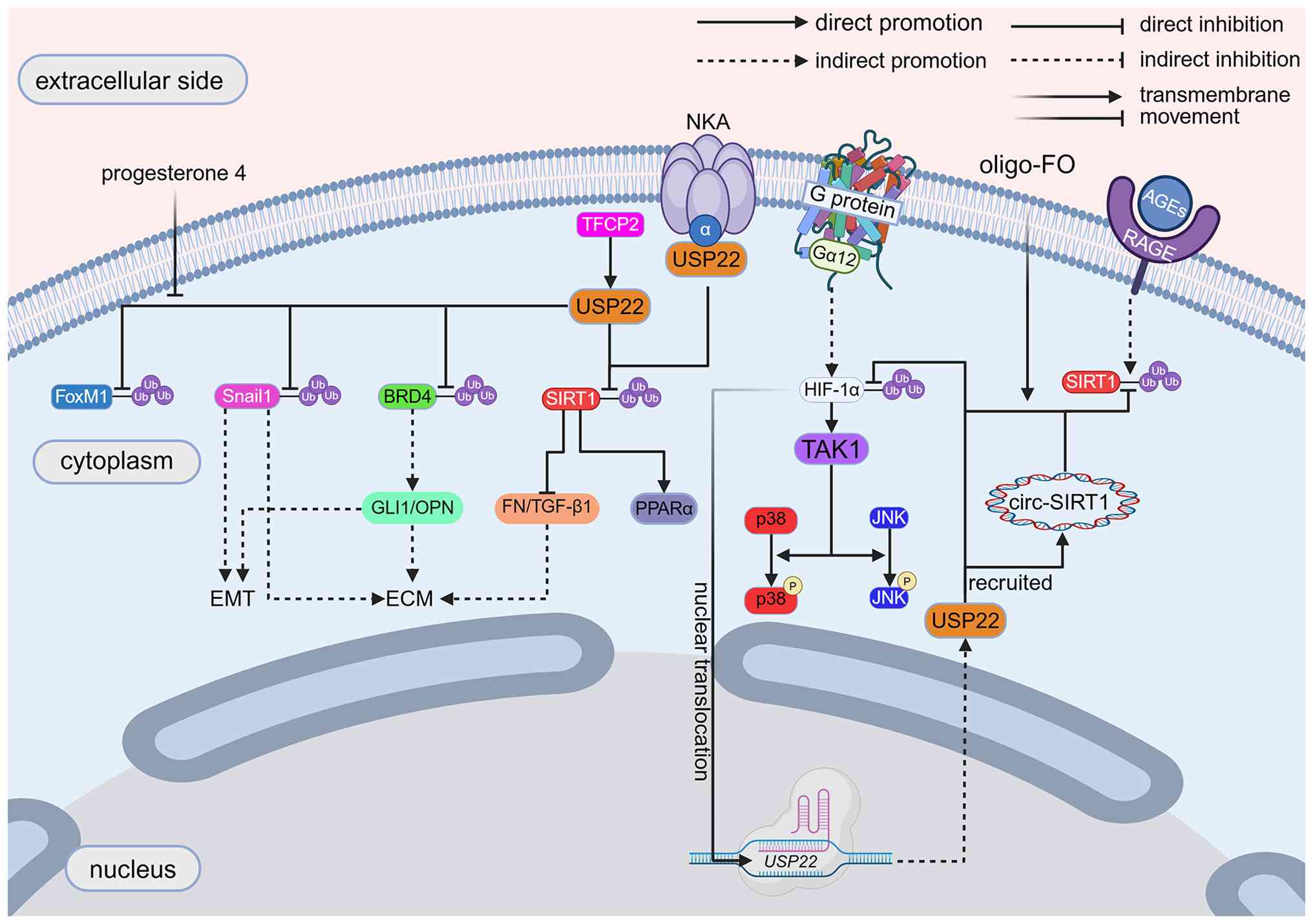
advanced glycation products (AGEs) promote SIRT1 degradation via
the ubiquitin-proteasome pathway, whereas USP22 specifically
removes K48-linked ubiquitin chains from SIRT1 to stabilize its
expression, suppress fibronectin (FN)/TGF-β1 production and reduce
ECM deposition (50,121,122). Similarly, low-molecular-weight
fucoidan blocks AGE/RAGE pathway-induced ECM accumulation by
activating the USP22/SIRT1 axis, significantly ameliorating renal
fibrosis in diabetic conditions (121,126,127).
EMT serves a critical role in promoting pulmonary
fibrosis in patients with pneumoconiosis and tubulointerstitial
fibrosis related to DKD (123,124,128). Pneumoconiosis, which has been
identified as a major global public health threat, is caused by
prolonged inhalation of mineral dust leading to extensive pulmonary
fibrosis (124,129). Integrated analysis of radiomics
and transcriptomics revealed that high expression of transcription
factor CP2 (TFCP2) is positively associated with pulmonary fibrosis
severity. Following mineral dust inhalation, enhanced TFCP2/USP22
interaction in lung epithelial cells activates the PI3K/AKT
signaling pathway, thereby promoting EMT and ECM deposition
(124). As a key EMT
transcription factor, Snail Homolog 1 (Snail1) is deubiquitinated
and stabilized by USP22 in renal tubular epithelial cells, driving
their transition to mesenchymal cells and the gradual replacement
of normal tubules and/or the surrounding matrix by matrix proteins
(123,130,131). Quercetin ameliorates the
pathological process of tubulointerstitial fibrosis by reducing
USP22-Snail1 binding and abolishing the deubiquitination-mediated
stabilization of Snail1 (132).
In patients with HN, the accumulation of paracrine HMGB1 in
podocytes induces fibrotic marker expression in tubular epithelial
cells, exacerbating renal interstitial inflammation and fibrosis
(53) (Table IV; Fig. 5).
Nonalcoholic fatty liver disease (NAFLD), renamed
metabolic fatty liver disease (MAFLD), has become the most common
liver disease in the world (133,134). The activity of Na+/K+-ATPase
(NKA), a classic ubiquitous ion pump (135), is significantly reduced and the
expression of the NKAα1 subunit is decreased in hepatocytes under
hyperglycemic/hyperinsulinemia conditions (136,137). Decreased NKA activity in liver
tissues of patients with NAFLD and HFD-fed mice diminishes the
formation of membrane-associated NKAα1/USP22 complexes, impairing
the recruitment and deubiquitination-mediated stabilization of
SIRT1 to disrupt fatty acid oxidation (FAO) (138). Gα12, a recently identified G12
family member of G protein α subunits, is widely expressed in the
liver (139-141) and Gα12 deficiency predisposes
the liver to lipid accumulation (142). Gα12 induces USP22 transcription
via HIF-1α, which maintains SIRT1 stability through
deubiquitination to activate peroxisome proliferator-activated
receptor alpha-mediated mitochondrial FAO gene expression (142,143). Taken together, these findings
reveal a pivotal regulatory role of USP22 in maintaining hepatic
lipid metabolic homeostasis. The interaction between NKAα1 and
USP22 provides a new paradigm for understanding the regulatory
cascade of 'metabolic environment-membrane protein complex-cellular
metabolism'. Future exploration can be conducted on how to
indirectly regulate USP22 by improving NKA function, which may
offer new insights for the intervention of metabolic liver diseases
(Table IV; Fig. 5).
Asthma, a common heterogeneous disease characterized
by chronic airway inflammation and remodeling (144,145), features airway smooth muscle
cell (ASMC) proliferation and collagen deposition as remodeling
hallmarks (146). TGF-β1
induces USP22 expression upregulation in ASMCs, stabilizing BRD4 to
activate the Hedgehog pathway effectors GLI family zinc finger 1
(GLI1) and osteopontin, thereby driving ASMC proliferation and
airway collagen deposition (147).
Myocardial hypertrophy, a compensatory response of
the heart to physiological or pathological pressure overload, can
progress to heart failure (70,71,148). In the hypertrophic myocardium,
Circ-SIRT1 expression is significantly downregulated.
Overexpression of Circ-SIRT1 results in the recruitment of USP22 to
remove ubiquitin modifications from SIRT1, stabilizing its protein
level to promote autophagy and inhibit cardiomyocyte hypertrophy
and cardiac remodeling (149).
Paradoxically, USP22 protein expression is upregulated in
myocardial hypertrophy. Upregulated USP22 stabilizes
hypoxia-inducible factor 1α, thereby activating transforming growth
factor-β activated kinase 1 to phosphorylate downstream p38 and
JNK1/2, promoting myocardial hypertrophy, fibrosis and dysfunction
(150). These findings suggest
that downregulated Circ-SIRT1 and upregulated USP22 expression may
synergistically drive myocardial hypertrophy, whereas Circ-SIRT1
overexpression counteracts USP22-mediated effects to inhibit
hypertrophic phenotypes. Additional studies (151) have explained the upregulation
of USP22 expression in cardiomyocytes following chronic heart
failure: POZ/BTB and AT hook containing zinc finger 1
transcriptionally activate USP22 expression by binding to its
promoter (151).
Human endometrial stromal cells (hESCs), which act
as endometrial mesenchymal/stem cells, participate in monthly
regeneration (152).
Endometritis disrupts hESC function (153,154), leading to decidualization
failure and an increased risk of early miscarriage (155). Decidualization, a prerequisite
for blastocyst implantation and placental development (156,157), is regulated by ovarian hormones
(158). USP22 influences the
pathological progression of endometritis by modulating hESC
inflammatory responses, ferroptosis and decidualization.
Endometritis reduces USP22 expression in hESCs, whereas USP22
overexpression alleviates inflammation, suppresses ferroptosis and
ameliorates decidualization failure (159). USP22 expression is
significantly decreased in the endometrium of patients with
recurrent implantation failure, accompanied by enhanced proteasomal
degradation of forkhead box protein M1, leading to reduced
endometrial receptivity (160).
During early pregnancy and uterine decidualization, progesterone
stimulates USP22 upregulation in mouse endometrial stromal cells,
indicating that progesterone induces USP22 expression via the
progesterone/progesterone receptor pathway to promote
decidualization (161)
(Table IV; Fig. 5).
As a core deubiquitinase in the SAGA complex, USP22
exerts pleiotropic regulatory effects through two major molecular
mechanisms and serves as a key node in biological and pathological
networks.
First, the USP22-SIRT1-centred regulatory axis has
extensive pandisease relevance and acts as a core regulatory module
across tissues and diseases. By deubiquitinating and stabilizing
SIRT1, USP22 regulates diverse downstream signaling pathways in
different physiological and pathological contexts to maintain
cellular function and tissue homeostasis. In metabolism-related
diseases, this axis regulates FAO and metabolic balance and
functions as a common driver of metabolic syndrome-associated
pathological processes. In patients with fibrotic diseases such as
diabetic nephropathy, the USP22-SIRT1 axis inhibits the expression
of fibrosis-related factors, reduces extracellular matrix
deposition and exerts antifibrotic effects. In I/R injury of
tissues such as the myocardium and brain, the axis protects cells
through anti-inflammatory, antioxidative and antiapoptotic effects.
Additionally, it is involved in physiological processes such as
pancreatic β-cell survival and hepatic lipid metabolic homeostasis,
with its dysfunction closely linked to the development and
progression of various diseases. Systematic validation of the
regulatory patterns of this axis in various disease cohorts is
expected to establish it as a universal biomarker for metabolic
disorders and oxidative stress-related diseases, laying the
foundation for the development of cross-disease therapeutic
agents.
Second, USP22 forms a microenvironment-specific
regulatory network through dynamic interactions with diverse
proteins and the core mechanism underlying its functional duality
can be summarized as the crosstalk between substrate competition
and its own posttranslational modifications. The functional
tendency of USP22 is not fixed but is jointly shaped by its
posttranslational modification status and the competitive binding
of downstream substrates. This dynamic balance enables it to adapt
to the signal requirements of different microenvironments, thereby
exhibiting contrasting effects such as anti-inflammatory or
proinflammatory effects and protective or damaging effects.
Moreover, as a core component of the SAGA complex, the functions of
USP22 are finely regulated by upstream signaling networks and its
regulatory scope and precision are further expanded through
crosstalk with key signaling pathways. Notably, USP22 can also
target disease-specific substrates, which not only serve as key
mediators of its functions but also act as molecular markers to
distinguish different pathological states. This provides a basis
for precise disease classification and addresses the lack of
specific biomarkers for the clinical diagnosis of certain
diseases.
The clinical translation of the functions of USP22
focuses on the precise expansion of drug development and the
clinical implementation of diagnostic biomarkers and the
exploration of more targeted translational pathways on the basis of
existing research foundations. In terms of drug development, the
combined application of drug repurposing has become a key
direction: Combining DPP-4 inhibitors with inflammation-regulating
inhibitors can synergistically regulate USP22-related axes in
metabolic diseases, simultaneously improving metabolic disorders
and inflammatory responses to enhance intervention efficacy. The
optimization potential of natural products is worthy of
exploration; enhancing the bioavailability of quercetin,
oligofucoidan and other compounds through nanocarrier technology
can provide new options for low-cost adjuvant therapy. To address
the double-edged sword effect of USP22, a disease stage-subcellular
localization dual-guided strategy can reduce off-target effects and
combining this strategy with artificial intelligence-assisted
design of substrate-specific inhibitors is expected to overcome the
limitations of traditional drugs.
In the field of diagnostic biomarkers,
USP22-substrate modules exhibit prominent clinical value:
USP22-ADAM9 can distinguish preeclampsia from fetal growth
restriction and USP22-Snail1 can assess the progression of renal
fibrosis. Following validation in clinical cohorts, these modules
are expected to be transformed into diagnostic or prognostic
assessment tools. Single-cell spatial multiomics technology can
further screen specific biomarker panels to improve diagnostic
accuracy. Additionally, the potential value of USP22 in the
intervention of aging-related diseases and the enhancement of stem
cell transplantation efficacy has opened up new scenarios for its
clinical application and future research should focus on exploring
the translational feasibility of these directions.
Not applicable.
JNX and CYZ wrote the main manuscript text, YDZ
consulted research materials and XX and SBL assisted with topic
selection and guided writing. Data authentication is not
applicable. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present study was supported by the National Natural Science
Foundation of China (grant no. 82273270).
|
1
|
Spedale G, Timmers HT and Pijnappel WW:
ATAC-king the complexity of SAGA during evolution. Genes Dev.
26:527–541. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Huang J, Dai W, Xiao D, Xiong Q, Liu C, Hu
J, Ge F, Yu X and Li S: Acetylation-dependent SAGA complex
dimerization promotes nucleosome acetylation and gene
transcription. Nat Struct Mol Biol. 29:261–273. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Morgan MT, Haj-Yahya M, Ringel AE, Bandi
P, Brik A and Wolberger C: Structural basis for histone H2B
deubiquitination by the SAGA DUB module. Science. 351:725–728.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Feng T, Ling S, Xu C, Ying L, Su D and Xu
X: Ubiquitin-specific peptidase 22 in cancer. Cancer Lett.
514:30–37. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Melo-Cardenas J, Zhang Y, Zhang DD and
Fang D: Ubiquitin-specific peptidase 22 functions and its
involvement in disease. Oncotarget. 7:44848–44856. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bonnet J, Romier C, Tora L and Devys D:
Zinc-finger UBPs: Regulators of deubiquitylation. Trends Biochem
Sci. 33:369–375. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yadav P, Subbarayalu P, Medina D, Nirzhor
S, Timilsina S, Rajamanickam S, Eedunuri VK, Gupta Y, Zheng S,
Abdelfattah N, et al: M6A RNA methylation regulates histone
ubiquitination to support cancer growth and progression. Cancer
Res. 82:1872–1889. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ding F, Bao C, Tian Y, Xiao H, Wang M, Xie
X, Hu F and Mei J: USP22 promotes NSCLC tumorigenesis via MDMX
up-regulation and subsequent p53 inhibition. Int J Mol Sci.
16:307–320. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hu J, Yang D, Zhang H, Liu W, Zhao Y, Lu
H, Meng Q, Pang H, Chen X, Liu Y and Cai L: USP22 promotes tumor
progression and induces epithelial-mesenchymal transition in lung
adenocarcinoma. Lung Cancer. 88:239–245. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lu P, Li Z and Xu H: USP22 promotes
gefitinib resistance and inhibits ferroptosis in non-small cell
lung cancer by deubiquitination of MDM2. Thorac Cancer.
15:2260–2271. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dixon SJ and Olzmann JA: The cell biology
of ferroptosis. Nat Rev Mol Cell Biol. 25:424–442. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jing T, Wang B, Yang Z and Liu Y, Xu G, Xu
X, Jiao K, Chen Z, Xiang L, Zhang L and Liu Y: Deubiquitination of
the repressor E2F6 by USP22 facilitates AKT activation and tumor
growth in hepatocellular carcinoma. Cancer Lett. 518:266–277. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ning Z, Guo X, Liu X, Lu C, Wang A, Wang
X, Wang W, Chen H, Qin W, Liu X, et al: USP22 regulates lipidome
accumulation by stabilizing PPARγ in hepatocellular carcinoma. Nat
Commun. 13:21872022. View Article : Google Scholar
|
|
14
|
Ling S, Li J, Shan Q, Dai H, Lu D, Wen X,
Song P, Xie H, Zhou L, Liu J, et al: USP22 mediates the multidrug
resistance of hepatocellular carcinoma via the SIRT1/AKT/MRP1
signaling pathway. Mol Oncol. 11:682–695. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
McCann JJ, Vasilevskaya IA, Poudel Neupane
N, Shafi AA, McNair C, Dylgjeri E, Mandigo AC, Schiewer MJ,
Schrecengost RS, Gallagher P, et al: USP22 functions as an
oncogenic driver in prostate cancer by regulating cell
proliferation and DNA repair. Cancer Res. 80:430–443. 2020.
View Article : Google Scholar
|
|
16
|
Prokakis E, Dyas A, Grün R, Fritzsche S,
Bedi U, Kazerouni ZB, Kosinsky RL, Johnsen SA and Wegwitz F: USP22
promotes HER2-driven mammary carcinoma aggressiveness by
suppressing the unfolded protein response. Oncogene. 40:4004–4018.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Huang J, Yin Q, Wang Y, Zhou X, Guo Y,
Tang Y, Cheng R, Yu X, Zhang J, Huang C, et al: EZH2 inhibition
enhances PD-L1 protein stability through USP22-Mediated
deubiquitination in colorectal cancer. Adv Sci (Weinh).
11:e23080452024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang D, Jiang F, Wang X and Li G:
Downregulation of ubiquitin-specific protease 22 inhibits
proliferation, invasion, and epithelial-mesenchymal transition in
osteosarcoma cells. Oncol Res. 25:743–751. 2017. View Article : Google Scholar
|
|
19
|
Roedig J, Kowald L, Juretschke T,
Karlowitz R, Ahangarian Abhari B, Roedig H, Fulda S, Beli P and van
Wijk SJ: USP22 controls necroptosis by regulating
receptor-interacting protein kinase 3 ubiquitination. EMBO Rep.
22:e501632021. View Article : Google Scholar :
|
|
20
|
Ji AL, Li T, Zu G, Feng DC, Li Y, Wang GZ,
Yao JH and Tian XF: Ubiquitin-specific protease 22 enhances
intestinal cell proliferation and tissue regeneration after
intestinal ischemia reperfusion injury. World J Gastroenterol.
25:824–836. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Koutelou E, Hirsch CL and Dent SY:
Multiple faces of the SAGA complex. Curr Opin Cell Biol.
22:374–382. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu W, Edmondson DG, Evrard YA, Wakamiya M,
Behringer RR and Roth SY: Loss of Gcn5l2 leads to increased
apoptosis and mesodermal defects during mouse development. Nat
Genet. 26:229–232. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wilde JJ, Siegenthaler JA, Dent SY and
Niswander LA: Diencephalic size is restricted by a novel interplay
between GCN5 acetyltransferase activity and retinoic acid
signaling. J Neurosci. 37:2565–2579. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xiong J, Gong Z, Zhou X, Liu J, Jiang HE,
Wu P and Li W: p38 mitogen-activated protein kinase inhibits USP22
transcription in HeLa cells. Biomed Rep. 3:461–467. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sussman RT, Stanek TJ, Esteso P, Gearhart
JD, Knudsen KE and McMahon SB: The epigenetic modifier
ubiquitin-specific protease 22 (USP22) regulates embryonic stem
cell differentiation via transcriptional repression of
sex-determining region Y-box 2 (SOX2). J Biol Chem.
288:24234–24246. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kosinsky RL, Wegwitz F, Hellbach N,
Dobbelstein M, Mansouri A, Vogel T, Begus-Nahrmann Y and Johnsen
SA: Usp22 deficiency impairs intestinal epithelial lineage
specification in vivo. Oncotarget. 6:37906–37918. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin Z, Yang H, Kong Q, Li J, Lee SM, Gao
B, Dong H, Wei J, Song J, Zhang DD and Fang D: USP22 antagonizes
p53 transcriptional activation by deubiquitinating Sirt1 to
suppress cell apoptosis and is required for mouse embryonic
development. Mol Cell. 46:484–494. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu Z, Wang J, Gao Y, Guo Y, Zhu Y, Sun Y
and Yang H: USP22 regulates the formation and function of placental
vasculature during the development of fetal growth restriction.
Placenta. 111:19–25. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Burton GJ and Jauniaux E: Pathophysiology
of placental-derived fetal growth restriction. Am J Obstet Gynecol.
218:S745–S761. 2018. View Article : Google Scholar
|
|
30
|
Colella M, Frérot A, Novais ARB and Baud
O: Neonatal and long-term consequences of fetal growth restriction.
Curr Pediatr Rev. 14:212–218. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Giabicani E, Pham A, Brioude F, Mitanchez
D and Netchine I: Diagnosis and management of postnatal fetal
growth restriction. Best Pract Res Clin Endocrinol Metab.
32:523–534. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Koutelou E, Wang L, Schibler AC, Chao HP,
Kuang X, Lin K, Lu Y, Shen J, Jeter CR, Salinger A, et al: USP22
controls multiple signaling pathways that are essential for
vasculature formation in the mouse placenta. Development.
146:dev1740372019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu J, Wang Y, Zhang S, Sun L and Shi Y:
ADAM9 deubiquitination induced by USP22 suppresses proliferation,
migration, invasion, and epithelial-mesenchymal transition of
trophoblast cells in preeclampsia. Placenta. 146:50–57. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kempermann G and Gage FH: New nerve cells
for the adult brain. Sci Am. 280:48–53. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bond AM, Ming GL and Song H: Adult
mammalian neural stem cells and neurogenesis: Five decades later.
Cell Stem Cell. 17:385–395. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kobayashi T, Mizuno H, Imayoshi I,
Furusawa C, Shirahige K and Kageyama R: The cyclic gene Hes1
contributes to diverse differentiation responses of embryonic stem
cells. Genes Dev. 23:1870–1875. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Imayoshi I, Isomura A, Harima Y, Kawaguchi
K, Kori H, Miyachi H, Fujiwara T, Ishidate F and Kageyama R:
Oscillatory control of factors determining multipotency and fate in
mouse neural progenitors. Science. 342:1203–1208. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kobayashi T, Iwamoto Y, Takashima K,
Isomura A, Kosodo Y, Kawakami K, Nishioka T, Kaibuchi K and
Kageyama R: Deubiquitinating enzymes regulate Hes1 stability and
neuronal differentiation. FEBS J. 282:2411–2423. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang Z, Li J, Wang B, Hou C, Liu Q, Wang
W, Zhao Y, Yin Q, Yang S, Zhang H and Yang X: Ubiquitin-specific
protease 22 promotes neural stem cells stemness maintenance and
adult hippocampal neurogenesis, contributing to cognitive recovery
following traumatic brain injury. Neuroscience. 496:219–229. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xie W, Schultz MD, Lister R, Hou Z,
Rajagopal N, Ray P, Whitaker JW, Tian S, Hawkins RD, Leung D, et
al: Epigenomic analysis of multilineage differentiation of human
embryonic stem cells. Cell. 153:1134–1148. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hawkins RD, Hon GC, Lee LK, Ngo Q, Lister
R, Pelizzola M, Edsall LE, Kuan S, Luu Y, Klugman S, et al:
Distinct epigenomic landscapes of pluripotent and lineage-committed
human cells. Cell Stem Cell. 6:479–491. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhu J, Adli M, Zou JY, Verstappen G, Coyne
M, Zhang X, Durham T, Miri M, Deshpande V, De Jager PL, et al:
Genome-wide chromatin state transitions associated with
developmental and environmental cues. Cell. 152:642–654. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sheaffer KL, Kim R, Aoki R, Elliott EN,
Schug J, Burger L, Schübeler D and Kaestner KH: DNA methylation is
required for the control of stem cell differentiation in the small
intestine. Genes Dev. 28:652–664. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bielefeld KA, Amini-Nik S and Alman BA:
Cutaneous wound healing: recruiting developmental pathways for
regeneration. Cell Mol Life Sci. 70:2059–2081. 2013. View Article : Google Scholar :
|
|
45
|
Lee SC, Jeong HJ, Lee SK and Kim SJ:
Hypoxic conditioned medium from human adipose-derived stem cells
promotes mouse liver regeneration through JAK/STAT3 signaling. Stem
Cells Transl Med. 5:816–825. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Christman KL, Vázquez-Dorbatt V, Schopf E,
Kolodziej CM, Li RC, Broyer RM, Chen Y and Maynard HD: Nanoscale
growth factor patterns by immobilization on a heparin-mimicking
polymer. J Am Chem Soc. 130:16585–16591. 2008. View Article : Google Scholar
|
|
47
|
Qian L, Li B, Pi L, Fang B and Meng X:
Hypoxic adipose stem cell-derived exosomes carrying high-abundant
USP22 facilitate cutaneous wound healing through stabilizing HIF-1α
and upregulating lncRNA H19. FASEB J. 38:e236532024. View Article : Google Scholar
|
|
48
|
Wang J, Yue G, Chen W, Li R, Han X, Cai W,
Liu Y and Xu J: CDK9-mediated transcriptional activation of NEK7
promotes NLRP3-dependent podocyte Pyroptosis in diabetic kidney
disease. Int Immunopharmacol. 171:1161722026. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Shi JX, Wang QJ, Li H and Huang Q:
Silencing of USP22 suppresses high glucose-induced apoptosis, ROS
production and inflammation in podocytes. Mol Biosyst.
12:1445–1456. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ao N and Liu Y, Feng H, Bian X, Li Z, Gu
B, Zhao X and Liu Y: Ubiquitin-specific peptidase USP22 negatively
regulates the STAT signaling pathway by deubiquitinating SIRT1.
Cell Physiol Biochem. 33:1863–1875. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang X, Wang W, Han M, Zhang J and Li Y:
ELAVL1-stabilized USP22 promotes diabetic nephropathy progression
via mediating podocyte injury and death by triggering ACSL4
deubiquitination. Transpl Immunol. 93:1022802025. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Xu X, Piao HN, Aosai F, Zeng XY, Cheng JH,
Cui YX, Li J, Ma J, Piao HR, Jin X and Piao LX: Arctigenin protects
against depression by inhibiting microglial activation and
neuroinflammation via HMGB1/TLR4/NF-ĸB and TNF-α/TNFR1/NF-ĸB
pathways. Br J Pharmacol. 177:5224–5245. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Peng Z, Huang X, Pan Y, Li W, Hu H, Chen
X, Zhang Z, Hu J, Qi Y, Chen W, et al: USP22 promotes angiotensin
II-induced podocyte injury by deubiquitinating and stabilizing
HMGB1. Cell Signal. 131:1117712025. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Halim M and Halim A: The effects of
inflammation, aging and oxidative stress on the pathogenesis of
diabetes mellitus (type 2 diabetes). Diabetes Metab Syndr.
13:1165–1172. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Evans JL, Goldfine ID, Maddux BA and
Grodsky GM: Oxidative stress and stress-activated signaling
pathways: A unifying hypothesis of type 2 diabetes. Endocr Rev.
23:599–622. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lytrivi M, Castell AL, Poitout V and Cnop
M: Recent insights into mechanisms of β-Cell lipo- and
glucolipotoxicity in type 2 diabetes. J Mol Biol. 432:1514–1534.
2020. View Article : Google Scholar
|
|
57
|
Elumalai S, Karunakaran U, Moon JS and Won
KC: High glucose-induced PRDX3 acetylation contributes to
glucotoxicity in pancreatic β-cells: Prevention by Teneligliptin.
Free Radic Biol Med. 160:618–629. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Stockwell BR, Friedmann Angeli JP, Bayir
H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascón S, Hatzios SK,
Kagan VE, et al: Ferroptosis: A regulated cell death nexus linking
metabolism, redox biology, and disease. Cell. 171:273–285. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zhang S, Liu X, Wang J, Yuan F and Liu Y:
Targeting ferroptosis with miR-144-3p to attenuate pancreatic β
cells dysfunction via regulating USP22/SIRT1 in type 2 diabetes.
Diabetol Metab Syndr. 14:892022. View Article : Google Scholar
|
|
60
|
Tong L, Liu R, Yang Y, Zhao J, Ye S, Wang
X and Qin Y: Ghrelin protects against ischemia/reperfusion-induced
hepatic injury via inhibiting Caspase-11-mediated noncanonical
pyroptosis. Transpl Immunol. 80:1018882023. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Tendler DA: Acute intestinal ischemia and
infarction. Semin Gastrointest Dis. 14:66–76. 2003.PubMed/NCBI
|
|
62
|
Blikslager AT, Moeser AJ, Gookin JL, Jones
SL and Odle J: Restoration of barrier function in injured
intestinal mucosa. Physiol Rev. 87:545–564. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zhang MX, Zhao Q and He XS: Research
progress of ischemia-free liver transplantation. Hepatobiliary
Pancreat Dis Int. 24:18–22. 2025. View Article : Google Scholar
|
|
64
|
Neri M, Riezzo I, Pascale N, Pomara C and
Turillazzi E: Ischemia/reperfusion injury following acute
myocardial infarction: A critical issue for clinicians and forensic
pathologists. Mediators Inflamm. 2017:70183932017. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Ye J, Lyu TJ, Li LY, Liu Y, Zhang H, Wang
X, Xi X, Liu ZJ and Gao JQ: Ginsenoside Re attenuates myocardial
ischemia/reperfusion induced ferroptosis via miR-144-3p/SLC7A11.
Phytomedicine. 113:1546812023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Bulluck H, Rosmini S, Abdel-Gadir A, White
SK, Bhuva AN, Treibel TA, Fontana M, Ramlall M, Hamarneh A, Sirker
A, et al: Residual myocardial iron following intramyocardial
hemorrhage during the convalescent phase of reperfused
ST-Segment-elevation myocardial infarction and adverse left
ventricular remodeling. Circ Cardiovasc Imaging. 9:e0049402016.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Xiang Q, Yi X, Zhu XH, Wei X and Jiang DS:
Regulated cell death in myocardial ischemia-reperfusion injury.
Trends Endocrinol Metab. 35:219–234. 2024. View Article : Google Scholar
|
|
68
|
Ma S, Sun L, Wu W, Wu J, Sun Z and Ren J:
USP22 protects against myocardial ischemia-reperfusion injury via
the SIRT1-p53/SLC7A11-dependent inhibition of ferroptosis-induced
cardiomyocyte death. Front Physiol. 11:5513182020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Song S, Wang Y, Wang HY and Guo LL: Role
of sevoflurane in myocardial ischemia-reperfusion injury via the
ubiquitin-specific protease 22/lysine-specific demethylase 3A axis.
Bioengineered. 13:13366–13383. 2022. View Article : Google Scholar :
|
|
70
|
Oka T, Akazawa H, Naito AT and Komuro I:
Angiogenesis and cardiac hypertrophy: Maintenance of cardiac
function and causative roles in heart failure. Circ Res.
114:565–571. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Bernardo BC, Weeks KL, Pretorius L and
McMullen JR: Molecular distinction between physiological and
pathological cardiac hypertrophy: Experimental findings and
therapeutic strategies. Pharmacol Ther. 128:191–227. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Zhou W, Yao J, Wang G, Chen Z, Li Z, Feng
D, Li Y, Qasim W, Tan W, Ning S and Tian X: PKCζ phosphorylates
TRAF2 to protect against intestinal ischemia-reperfusion-induced
injury. Cell Death Dis. 8:e29352017. View Article : Google Scholar
|
|
73
|
Eltzschig HK and Eckle T: Ischemia and
reperfusion-from mechanism to translation. Nat Med. 17:1391–1401.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Yoshinaga-Itano C and Thomson V: The work
of the Village: Creating a new world for children with hearing loss
and their families. Int J Audiol. 47(Suppl 1): S14–S22. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Zhang L, Zhang RL, Jiang Q, Ding G, Chopp
M and Zhang ZG: Focal embolic cerebral ischemia in the rat. Nat
Protoc. 10:539–547. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Mazuryk J, Puchalska I, Koziński K,
Ślusarz MJ, Ruczyński J, Rekowski P, Rogujski P, Płatek R,
Wiśniewska MB, Piotrowski A, et al: PTD4 peptide increases neural
viability in an in vitro model of acute ischemic stroke. Int J Mol
Sci. 22:60862021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Yanev P, Seevinck PR, Rudrapatna US, Bouts
MJ, van der Toorn A, Gertz K, Kronenberg G, Endres M, van Tilborg
GA and Dijkhuizen RM: Magnetic resonance imaging of local and
remote vascular remodelling after experimental stroke. J Cereb
Blood Flow Metab. 37:2768–2779. 2017. View Article : Google Scholar
|
|
78
|
Xie X, Cao Y, Dai L and Zhou D: Bone
marrow mesenchymal stem cell-derived exosomal lncRNA KLF3-AS1
stabilizes Sirt1 protein to improve cerebral ischemia/reperfusion
injury via miR-206/USP22 axis. Mol Med. 29:32023. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Li Y, Gao J, Liu C, Bu N, Zhan S, Wu H,
Zhang R, Sun H and Fan H: USP22 knockdown protects against cerebral
ischemia/reperfusion injury via destabilizing PTEN protein and
activating the mTOR/TFEB pathway. Naunyn Schmiedebergs Arch
Pharmacol. 396:3163–3175. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Qin X, Zhu J, Lu H, Yi M, Zhao Z, Zhang W
and Cheng J: Research progress of deubiquitinating enzymes in
cerebral ischemia-reperfusion injury. Front Aging Neurosci.
17:15889202025. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wen H, Liu L, Zhan L, Liang D, Li L, Liu
D, Sun W and Xu E: Neuroglobin mediates neuroprotection of hypoxic
postconditioning against transient global cerebral ischemia in rats
through preserving the activity of Na(+)/K(+) ATPases. Cell Death
Dis. 9:6352018. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Zuo Y, Xue J, Wen H, Zhan L, Chen M, Sun W
and Xu E: Inhibition of SCF(KDM2A)/USP22-dependent nuclear
β-catenin ubiquitylation mediates cerebral ischemic tolerance.
Commun Biol. 8:2142025. View Article : Google Scholar
|
|
83
|
Cai Z, Zhang MX, Tang Z, Zhang Q, Ye J,
Xiong TC, Zhang ZD and Zhong B: USP22 promotes IRF3 nuclear
translocation and antiviral responses by deubiquitinating the
importin protein KPNA2. J Exp Med. 217:e201911742020. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Xiao X, Li S, Zheng Z, Ji Y, Du Q, Zuo Y,
Miao Y, Yuan Y, Zheng H, Huang F and Wang J: Targeting USP22 to
promote K63-linked ubiquitination and degradation of SARS-CoV-2
nucleocapsid protein. J Virol. 99:e02234242025. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Karlowitz R, Stanifer ML, Roedig J,
Andrieux G, Bojkova D, Bechtel M, Smith S, Kowald L, Schubert R,
Boerries M, et al: USP22 controls type III interferon signaling and
SARS-CoV-2 infection through activation of STING. Cell Death Dis.
13:6842022. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Lin Y, Xu Y and Zhang Z: Sepsis-induced
myocardial dysfunction (SIMD): The pathophysiological mechanisms
and therapeutic strategies targeting mitochondria. Inflammation.
43:1184–1200. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Xu H, Ye W and Shi B: LncRNA MALAT1
regulates USP22 expression through EZH2-Mediated H3K27me3
modification to accentuate sepsis-induced myocardial dysfunction.
Cardiovasc Toxicol. 22:813–830. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Di Q, Zhao X, Tang H, Li X, Xiao Y, Wu H,
Wu Z, Quan J and Chen W: USP22 suppresses the NLRP3 inflammasome by
degrading NLRP3 via ATG5-dependent autophagy. Autophagy.
19:873–885. 2023. View Article : Google Scholar :
|
|
89
|
Yu MC, Li XL, Ning ML, Yan ZZ and Yu WT:
USP22 inhibits microglial M1 polarization by regulating the
PU.1/NLRP3 inflammasome pathway. Brain Res Bull. 220:1111572025.
View Article : Google Scholar
|
|
90
|
Sun H, Zhang Q, Jing YY, Zhang M, Wang HY,
Cai Z, Liuyu T, Zhang ZD, Xiong TC, Wu Y, et al: USP13 negatively
regulates antiviral responses by deubiquitinating STING. Nat
Commun. 8:155342017. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Strowig T, Henao-Mejia J, Elinav E and
Flavell R: Inflammasomes in health and disease. Nature.
481:278–286. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Davis BK, Wen H and Ting JP: The
inflammasome NLRs in immunity, inflammation, and associated
diseases. Annu Rev Immunol. 29:707–735. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Wen H, Ting JP and O'Neill LA: A role for
the NLRP3 inflammasome in metabolic diseases-did Warburg miss
inflammation? Nat Immunol. 13:352–357. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Tang Y, Wang T, Gu L, Xu Y, Yang Z, Zhu W,
Zhang Q, Luo J, Cao J and Jiao Y: USP11 exacerbates
radiation-induced pneumonitis by activating endothelial cell
inflammatory response via OTUD5-STING signaling. Int J Radiat Oncol
Biol Phys. 119:1261–1274. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Liu Q, Wu Y, Qin Y, Hu J, Xie W, Qin FX
and Cui J: Broad and diverse mechanisms used by deubiquitinase
family members in regulating the type I interferon signaling
pathway during antiviral responses. Sci Adv. 4:eaar28242018.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Chen D, Song D, Ma Y, Lu W, Qiu J and Wang
Y: USP22 promotes pro-inflammatory responses in Pseudomonas
aeruginosa-induced keratitis by targeting TRAF6. Mol Med Rep.
25:1492022. View Article : Google Scholar
|
|
97
|
Asrani SK, Devarbhavi H, Eaton J and
Kamath PS: Burden of liver diseases in the world. J Hepatol.
70:151–171. 2019. View Article : Google Scholar
|
|
98
|
Grant BF, Chou SP, Saha TD, Pickering RP,
Kerridge BT, Ruan WJ, Huang B, Jung J, Zhang H, Fan A and Hasin DS:
Prevalence of 12-Month alcohol use, high-risk drinking, and DSM-IV
alcohol use disorder in the United States, 2001-2002 to 2012-2013:
Results from the National epidemiologic survey on alcohol and
related conditions. JAMA Psychiatry. 74:911–923. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Yan R, Chu J, Zhou Y, Shan W, Hu Y, Lin M,
Zhao Y, Sun R, Wang Z, Lv L, et al: Ubiquitin-specific protease 22
ameliorates chronic alcohol-associated liver disease by regulating
BRD4. Pharmacol Res. 168:1055942021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Boettcher S and Manz MG: Regulation of
inflammation- and infection-driven hematopoiesis. Trends Immunol.
38:345–357. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Brook B, Harbeson DJ, Shannon CP, Cai B,
He D, Ben-Othman R, Francis F, Huang J, Varankovich N, Liu A, et
al: BCG vaccination-induced emergency granulopoiesis provides rapid
protection from neonatal sepsis. Sci Transl Med. 12:eaax45172020.
View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Cortez JT, Montauti E, Shifrut E,
Gatchalian J, Zhang Y, Shaked O, Xu Y, Roth TL, Simeonov DR, Zhang
Y, et al: CRISPR screen in regulatory T cells reveals modulators of
Foxp3. Nature. 582:416–420. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Vossel K, Ranasinghe KG, Beagle AJ, La A,
Ah Pook K, Castro M, Mizuiri D, Honma SM, Venkateswaran N, Koestler
M, et al: Effect of levetiracetam on cognition in patients with
alzheimer disease with and without epileptiform activity: A
randomized clinical trial. JAMA Neurol. 78:1345–1354. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Zheng P, Wu J, Zhang H, Perry SW, Yin B,
Tan X, Chai T, Liang W, Huang Y, Li Y, et al: The gut microbiome
modulates gut-brain axis glycerophospholipid metabolism in a
region-specific manner in a nonhuman primate model of depression.
Mol Psychiatry. 26:2380–2392. 2021. View Article : Google Scholar :
|
|
105
|
Xu Y, Jin MZ, Yang ZY and Jin WL:
Microglia in neurodegenerative diseases. Neural Regen Res.
16:270–280. 2021. View Article : Google Scholar :
|
|
106
|
Melo-Cardenas J, Xu Y, Wei J, Tan C, Kong
S, Gao B, Montauti E, Kirsammer G, Licht JD, Yu J, et al: USP22
deficiency leads to myeloid leukemia upon oncogenic Kras activation
through a PU.1-dependent mechanism. Blood. 132:423–434. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Cakir B, Tanaka Y, Kiral FR, Xiang Y,
Dagliyan O, Wang J, Lee M, Greaney AM, Yang WS, duBoulay C, et al:
Expression of the transcription factor PU.1 induces the generation
of microglia-like cells in human cortical organoids. Nat Commun.
13:4302022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Pimenova AA, Herbinet M, Gupta I, Machlovi
SI, Bowles KR, Marcora E and Goate AM: Alzheimer's-associated PU.1
expression levels regulate microglial inflammatory response.
Neurobiol Dis. 148:1052172021. View Article : Google Scholar
|
|
109
|
Lu Y, Zheng Y, Sun X, Song Y, Zhou Y, Shen
F, Huang R and Xu Z: Knockdown of USP22 alleviates LPS-induced
microglial inflammation and mouse depressive-like behaviors via
KAT2A. Neuropsychopharmacology. 50:1904–1912. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Gao Y, Lin F, Xu P, Nie J, Chen Z, Su J,
Tang J, Wu Q, Li Y, Guo Z, et al: USP22 is a positive regulator of
NFATc2 on promoting IL2 expression. FEBS Lett. 588:878–883. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Liu X, Li H, Zhong B, Blonska M,
Gorjestani S, Yan M, Tian Q, Zhang DE, Lin X and Dong C: USP18
inhibits NF-κB and NFAT activation during Th17 differentiation by
deubiquitinating the TAK1-TAB1 complex. J Exp Med. 210:1575–1590.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Zhong B, Liu X, Wang X, Chang SH, Liu X,
Wang A, Reynolds JM and Dong C: Negative regulation of
IL-17-mediated signaling and inflammation by the ubiquitin-specific
protease USP25. Nat Immunol. 13:1110–1117. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
van Loosdregt J, Fleskens V, Fu J,
Brenkman AB, Bekker CP, Pals CE, Meerding J, Berkers CR, Barbi J,
Gröne A, et al: Stabilization of the transcription factor Foxp3 by
the deubiquitinase USP7 increases Treg-cell-suppressive capacity.
Immunity. 39:259–271. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Zhang J, Chen C, Hou X, Gao Y, Lin F, Yang
J, Gao Z, Pan L, Tao L, Wen C, et al: Identification of the E3
deubiquitinase ubiquitin-specific peptidase 21 (USP21) as a
positive regulator of the transcription factor GATA3. J Biol Chem.
288:9373–9382. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Yu X, Lao Y, Teng XL, Li S, Zhou Y, Wang
F, Guo X, Deng S, Chang Y, Wu X, et al: SENP3 maintains the
stability and function of regulatory T cells via BACH2
deSUMOylation. Nat Commun. 9:31572018. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Zhang Y, Wang Y, Gao B, Sun Y, Cao L,
Genardi SM, Wang CR, Li H, Sun Z, Yang Y and Fang D: USP22 controls
iNKT immunity through MED1 suppression of histone H2A
monoubiquitination. J Exp Med. 217:e201822182020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Friebus-Kardash J, Christ TC, Dietlein N,
Elwy A, Abdelrahman H, Holnsteiner L, Hu Z, Rodewald HR and Lang
KS: Usp22 deficiency leads to downregulation of PD-L1 and
pathological activation of CD8(+) T cells and causes
immunopathology in response to acute LCMV infection. Vaccines
(Basel). 11:15632023. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Chen SG, Xiao J, Liu XH, Liu MM, Mo ZC,
Yin K, Zhao GJ, Jiang J, Cui LB, Tan CZ, et al: Ibrolipim increases
ABCA1/G1 expression by the LXRα signaling pathway in THP-1
macrophage-derived foam cells. Acta Pharmacol Sin. 31:1343–1349.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Zeng M, Yang Y, Wang Z, Zhao X, Zhu D,
Wang M, Chen Y and Wei X: CTRP9 prevents atherosclerosis
progression through changing autophagic status of macrophages by
activating USP22 mediated-de-ubiquitination on Sirt1 in vitro. Mol
Cell Endocrinol. 584:1121612024. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Tang S, Lu C, Meng Z, Ye Z, Qin Y, Na N,
Xian S, Huang F and Zeng Z: USP22 enhances atherosclerotic plaque
stability and macrophage efferocytosis by stabilizing PPARγ. Commun
Biol. 8:6782025. View Article : Google Scholar
|
|
121
|
Huang KP, Chen C, Hao J, Huang JY, Liu PQ
and Huang HQ: AGEs-RAGE system down-regulates Sirt1 through the
ubiquitin-proteasome pathway to promote FN and TGF-β1 expression in
male rat glomerular mesangial cells. Endocrinology. 156:268–279.
2015. View Article : Google Scholar
|
|
122
|
Huang K, Huang J, Xie X, Wang S, Chen C,
Shen X, Liu P and Huang H: Sirt1 resists advanced glycation end
products-induced expressions of fibronectin and TGF-beta1 by
activating the Nrf2/ARE pathway in glomerular mesangial cells. Free
Radic Biol Med. 65:528–540. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Zhao X, He X, Wei W and Huang K: USP22
aggravated diabetic renal tubulointerstitial fibrosis progression
through deubiquitinating and stabilizing Snail1. Eur J Pharmacol.
947:1756712023. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Liu Y, Wu J, Zhou J, Guo J, Ren D, Bai Y
and Hu D: TFCP2 is recognized as a dynamic monitoring index of
pneumoconiosis by combining radiomics with transcriptomics. Int
Immunopharmacol. 153:1144572025. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Kanwar YS, Sun L, Xie P, Liu FY and Chen
S: A glimpse of various pathogenetic mechanisms of diabetic
nephropathy. Annu Rev Pathol. 6:395–423. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Yu WC, Huang RY and Chou TC:
Oligo-fucoidan improves diabetes-induced renal fibrosis via
activation of Sirt-1, GLP-1R, and Nrf2/HO-1: An in vitro and in
vivo study. Nutrients. 12:30682020. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Meng XM, Tang PM, Li J and Lan HY:
TGF-β/Smad signaling in renal fibrosis. Front Physiol. 6:822015.
View Article : Google Scholar
|
|
128
|
Xiao Y, Jiang X, Peng C, Zhang Y, Xiao Y,
Liang D, Shi M, Wang Y, Zhang F and Guo B: BMP-7/Smads-induced
inhibitor of differentiation 2 (Id2) upregulation and Id2/Twist
interaction was involved in attenuating diabetic renal
tubulointerstitial fibrosis. Int J Biochem Cell Biol.
116:1056132019. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Qi XM, Luo Y, Song MY, Liu Y, Shu T, Liu
Y, Pang JL, Wang J and Wang C: Pneumoconiosis: Current status and
future prospects. Chin Med J (Engl). 134:898–907. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Lee JH, Jung SM, Yang KM, Bae E, Ahn SG,
Park JS, Seo D, Kim M, Ha J, Lee J, et al: A20 promotes metastasis
of aggressive basal-like breast cancers through
multi-monoubiquitylation of Snail1. Nat Cell Biol. 19:1260–1273.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Miao H, Wu XQ, Zhang DD, Wang YN, Guo Y,
Li P, Xiong Q and Zhao YY: Deciphering the cellular mechanisms
underlying fibrosis-associated diseases and therapeutic avenues.
Pharmacol Res. 163:1053162021. View Article : Google Scholar
|
|
132
|
Zhao X, Wang S, He X, Wei W and Huang K:
Quercetin prevents the USP22-Snail1 signaling pathway to ameliorate
diabetic tubulointerstitial fibrosis. Food Funct. 15:11990–12006.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Wu MY and Fan JG: Gut microbiome and
nonalcoholic fatty liver disease. Hepatobiliary Pancreat Dis Int.
22:444–451. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Heida A, Gruben N, Catrysse L, Koehorst M,
Koster M, Kloosterhuis NJ, Gerding A, Havinga R, Bloks VW,
Bongiovanni L, et al: The hepatocyte IKK:NF-κB axis promotes liver
steatosis by stimulating de novo lipogenesis and cholesterol
synthesis. Mol Metab. 54:1013492021. View Article : Google Scholar
|
|
135
|
Cui X and Xie Z: Protein Interaction and
Na/K-ATPase-mediated signal transduction. Molecules. 22:9902017.
View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Iannello S, Milazzo P and Belfiore F:
Animal and human tissue Na,K-ATPase in normal and insulin-resistant
states: Regulation, behaviour and interpretative hypothesis on NEFA
effects. Obes Rev. 8:231–251. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Obradovic M, Bjelogrlic P, Rizzo M,
Katsiki N, Haidara M, Stewart AJ, Jovanovic A and Isenovic ER:
Effects of obesity and estradiol on Na+/K+-ATPase and their
relevance to cardiovascular diseases. J Endocrinol. 218:R13–R23.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Sun HJ, Tan JX, Shan XD, Wang ZC, Wu ZY,
Bian JS and Nie XW: DR region of NKAα1 is a target to ameliorate
hepatic lipid metabolism disturbance in obese mice. Metabolism.
145:1555792023. View Article : Google Scholar
|
|
139
|
Strathmann MP and Simon MI: G alpha 12 and
G alpha 13 subunits define a fourth class of G protein alpha
subunits. Proc Natl Acad Sci USA. 88:5582–5586. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Xu N, Bradley L, Ambdukar I and Gutkind
JS: A mutant alpha subunit of G12 potentiates the eicosanoid
pathway and is highly oncogenic in NIH 3T3 cells. Proc Natl Acad
Sci USA. 90:6741–6745. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Chan AM, Fleming TP, McGovern ES, Chedid
M, Miki T and Aaronson SA: Expression cDNA cloning of a
transforming gene encoding the wild-type G alpha 12 gene product.
Mol Cell Biol. 13:762–768. 1993.PubMed/NCBI
|
|
142
|
Kim TH, Yang YM, Han CY, Koo JH, Oh H, Kim
SS, You BH, Choi YH, Park TS, Lee CH, et al: Gα12 ablation
exacerbates liver steatosis and obesity by suppressing
USP22/SIRT1-regulated mitochondrial respiration. J Clin Invest.
128:5587–5602. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Leone TC, Weinheimer CJ and Kelly DP: A
critical role for the peroxisome proliferator-activated receptor
alpha (PPARalpha) in the cellular fasting response: the
PPARalpha-null mouse as a model of fatty acid oxidation disorders.
Proc Natl Acad Sci USA. 96:7473–7478. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Agache I, Palmer E, Sanver D, Kirtland M
and Shamji MH: Molecular allergology approach to allergic asthma.
Mol Aspects Med. 85:1010272022. View Article : Google Scholar
|
|
145
|
Papi A, Brightling C, Pedersen SE and
Reddel HK: Asthma. Lancet. 391:783–800. 2018. View Article : Google Scholar
|
|
146
|
Camoretti-Mercado B and Lockey RF: Airway
smooth muscle pathophysiology in asthma. J Allergy Clin Immunol.
147:1983–1995. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Chen H, Liu J, Zhang J, Chen Y, Wang Y,
Qiu Y, Hu H, Chai L, Zhang Q, Wang Q and Li M: USP22/BRD4 mediated
hedgehog pathway activation contributes to airway remodeling in
asthma. Int Immunopharmacol. 153:1145382025. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Driver JA, Djoussé L, Logroscino G,
Gaziano JM and Kurth T: Incidence of cardiovascular disease and
cancer in advanced age: Prospective cohort study. BMJ.
337:a24672008. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Wang W, Wang L, Yang M, Wu C, Lan R, Wang
W and Li Y: Circ-SIRT1 inhibits cardiac hypertrophy via activating
SIRT1 to promote autophagy. Cell Death Dis. 12:10692021. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Akimana C, Al-Khodor S and Abu Kwaik Y:
Host factors required for modulation of phagosome biogenesis and
proliferation of Francisella tularensis within the cytosol. PLoS
One. 5:e110252010. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Luo Y, Zhou F, Chen Q, Cai H, Yao K and
Deng X: Knockdown of PATZ1 alleviates chronic heart failure through
the USP22/HIF-1α axis. Sci Rep. 15:405662025. View Article : Google Scholar
|
|
152
|
Li T, Chan RWS, Li RHW, Ng EHY, Zhang S
and Yeung WSB: Endometrial mesenchymal stromal/stem cells improve
regeneration of injured endometrium in mice. Biol Res. 57:62024.
View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Yu J, Berga SL, Zou W, Yook DG, Pan JC,
Andrade AA, Zhao L, Sidell N, Bagchi IC, Bagchi MK and Taylor RN:
IL-1β inhibits connexin 43 and disrupts decidualization of human
endometrial stromal cells through ERK1/2 and p38 MAP kinase.
Endocrinology. 158:4270–4285. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Jiang J, Yu K, Jiang Z and Xue M: IL-37
affects the occurrence and development of endometriosis by
regulating the biological behavior of endometrial stromal cells
through multiple signaling pathways. Biol Chem. 399:1325–1337.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Yang HL, Yang TY, Gowrisankar YV, Liao CH,
Liao JW, Huang PJ and Hseu YC: Suppression of LPS-induced
inflammation by chalcone flavokawain a through activation of
Nrf2/ARE-mediated antioxidant genes and inhibition of ROS/NFκB
signaling pathways in primary splenocytes. Oxid Med Cell Longev.
2020:34762122020. View Article : Google Scholar
|
|
156
|
Dey SK, Lim H, Das SK, Reese J, Paria BC,
Daikoku T and Wang H: Molecular cues to implantation. Endocr Rev.
25:341–373. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Weimar CH, Macklon NS, Post Uiterweer ED,
Brosens JJ and Gellersen B: The motile and invasive capacity of
human endometrial stromal cells: Implications for normal and
impaired reproductive function. Hum Reprod Update. 19:542–557.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Strowitzki T, Germeyer A, Popovici R and
von Wolff M: The human endometrium as a fertility-determining
factor. Hum Reprod Update. 12:617–630. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Xing X, Zhang G, Yi F and Xu X:
Overexpression of USP22 ameliorates LPS-induced endometrial stromal
cells inflammation and modulates cells decidualization by
inhibiting ferroptosis. Reprod Biol. 24:1009132024. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Zhou M, Gao Y, Wu S, Wang Y and Yang J:
USP22 is required for human endometrial stromal cell proliferation
and decidualization by deubiquitinating FoxM1. Cell Signal.
121:1112652024. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Wang Y, Gao Y, Zhou C, Kong S, Wang H and
Yang J: Usp22 is expressed in mouse uterus during early pregnancy
and involved in endometrial stromal cell decidualization. Cells
Dev. 166:2036812021. View Article : Google Scholar : PubMed/NCBI
|