Orthopedic rehabilitation has long relied on
mechanical interventions, such as physical therapy, exercise and
load-bearing activities, to promote tissue repair and functional
recovery (1,2). However, the biological mechanisms
underlying these therapies have only recently been elucidated
through advances in understanding mechanotransduction, the process
by which cells convert mechanical stimuli into biochemical signals
(3,4). This intricate interplay between
physical forces and cellular responses plays a pivotal role in
tissue regeneration, particularly in the bone, cartilage, tendons
and ligaments (5,6). As regenerative medicine continues
to evolve, understanding mechanotransduction offers new
opportunities to enhance healing, optimize rehabilitation protocols
and develop novel bioengineered therapies (7,8).
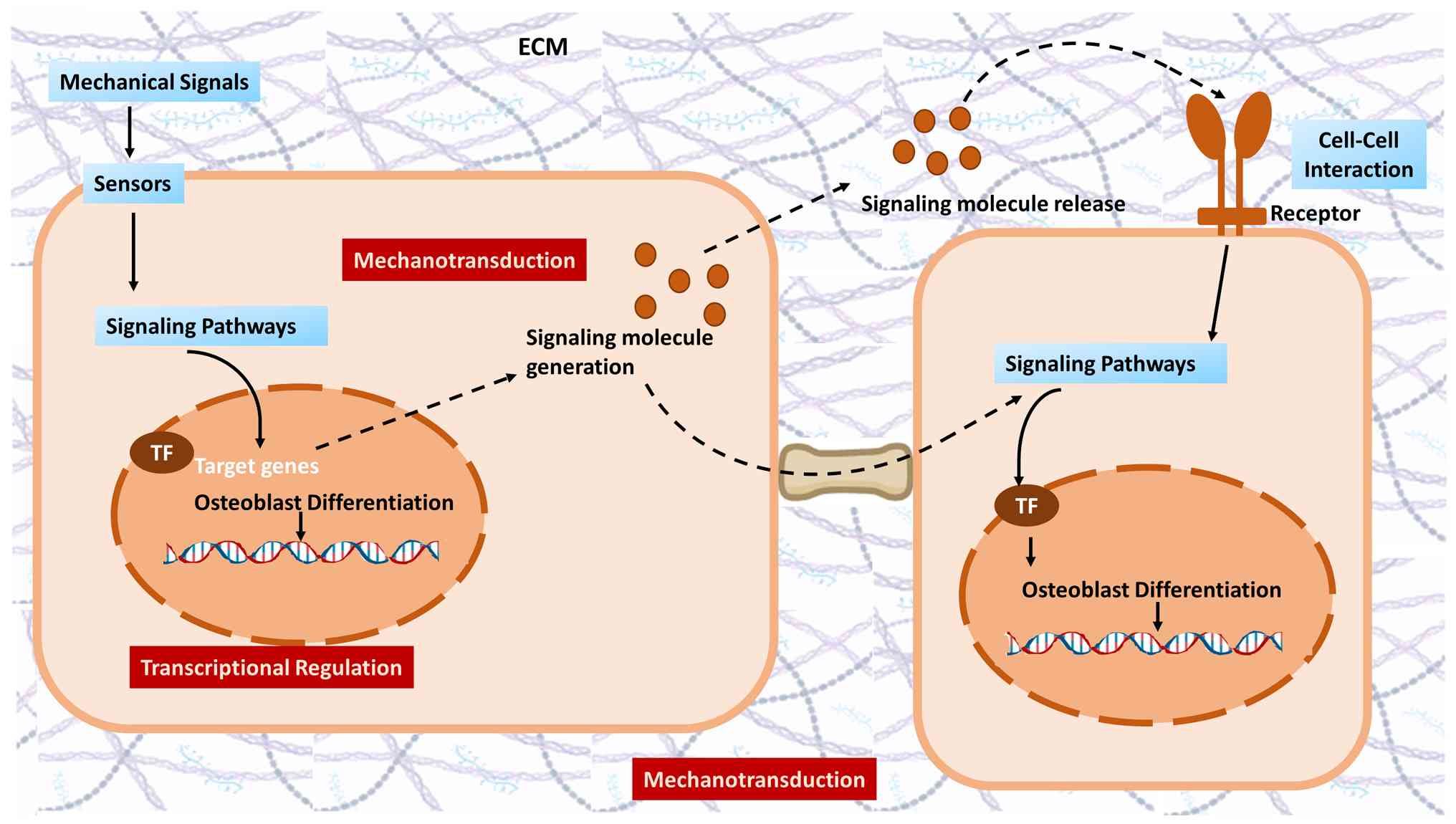
Mechanotransduction is a fundamental biological
phenomenon that enables cells to sense and respond to mechanical
cues such as tension, compression and shear stress (9,10). In orthopedic tissues, specialized
cells including osteocytes, chondrocytes, tenocytes and mesenchymal
stem cells (MSCs) possess mechanosensitive receptors (such as
integrins, ion channels and primary cilia) that detect
extracellular mechanical forces (11,12). These signals trigger
intracellular cascades, such as the activation of Yes-associated
protein (YAP)/transcriptional coactivator with PDZ-binding motif
(TAZ), Wnt/β-catenin and mitogen-activated protein kinase (MAPK)
pathways, ultimately influencing gene expression, extracellular
matrix (ECM) remodeling and tissue adaptation (13-15). The ECM itself acts as a dynamic
scaffold that transmits and amplifies mechanical signals, further
modulating cellular behavior (16,17).
Distraction histogenesis is the biological process
that regenerates bone and soft tissue (18). In bone regeneration, mechanical
loading stimulates the osteogenic differentiation of MSCs and
osteoblasts while inhibiting osteoclast activity, thereby promoting
bone formation and preventing resorption (19,20). Low-magnitude high-frequency
vibration and controlled cyclic loading have shown promise in
accelerating fracture healing and mitigating osteoporosis-related
bone loss (21,22). Similarly, in cartilage repair,
chondrocytes respond to dynamic compression by upregulating
anabolic factors (such as aggrecan and collagen type II) while
suppressing catabolic enzymes [such as matrix metalloproteinases
(MMPs)] (23,24). However, excessive or aberrant
loading can induce degenerative changes, highlighting the need for
precise mechanotherapeutic strategies (25). Tendon and ligament healing, often
hindered by poor vascularity and slow ECM turnover, also depends on
mechanotransduction (26,27).
Controlled mechanical stimulation enhances collagen alignment and
tensile strength, whereas immobilization leads to tissue atrophy
and fibrosis (28). Emerging
evidence suggests that tendon stem/progenitor cells exhibit
load-dependent differentiation, offering potential targets for
regenerative interventions (29). The integration of
mechanotransduction principles into regenerative medicine has led
to innovative approaches in orthopedic rehabilitation (7,25).
Biomechanically optimized scaffolds, embedded with
growth factors and designed to mimic native tissue mechanics,
enhance stem cell recruitment and differentiation (30,31). Additionally, dynamic bioreactor
systems apply physiologically relevant mechanical stimuli to
engineered tissues, improving their functional maturation before
implantation (32). Clinically,
mechanotherapy, the therapeutic application of mechanical forces,
has gained traction. Techniques such as extracorporeal shockwave
therapy (ESWT) and pulsed electromagnetic fields (PEMFs) harness
mechanotransduction to stimulate tissue repair, while personalized
rehabilitation protocols leverage patient-specific loading regimens
to maximize recovery (3,33). The optimal magnitude, frequency
and duration of mechanical stimuli vary across tissues and
individuals, necessitating further research into precision
mechanotherapies. Additionally, the crosstalk between mechanical
and biochemical signaling pathways must be deciphered to develop
synergistic treatment strategies. Future advancements in
biofabrication, smart biomaterials and artificial intelligence
(AI)-driven biomechanical modeling hold promise for tailoring
regenerative therapies to individual patient needs (34).
The present review comprehensively examines the
fundamental mechanisms of mechanotransduction, detailing how
specialized sensors convert physical forces into biochemical
signals that direct cellular behavior. The present review explores
the critical role of the ECM as a dynamic mediator of mechanical
signaling and investigates tissue-specific responses in bone,
cartilage, tendon and ligament regeneration. The discussion extends
to the notable impact of mechanical loading on stem cell
differentiation and the development of innovative biomechanical
strategies in regenerative medicine, including advanced
biomaterials and bioreactor systems. Clinically, the present review
focuses on translating these principles into effective
mechanotherapy protocols and personalized rehabilitation
approaches. Finally, the prevailing challenges in defining optimal
loading parameters are addressed and future directions are
explored, emphasizing the potential of emerging technologies such
as smart biomaterials and AI-driven modeling to create precise,
biologically-driven interventions. By synthesizing these elements,
the present review aims to highlight the transformative potential
of integrating mechanobiology with regenerative medicine to advance
orthopedic rehabilitation outcomes.
The conversion of mechanical forces into biochemical
signals, a process fundamental to tissue repair, is initiated by
specialized cellular structures known as mechanosensors. These
sensors detect physical cues including compression, tension and
fluid shear stress within the musculoskeletal environment (35). Principal among these are
integrins, transmembrane receptors that form focal adhesion
complexes, creating a critical link between the ECM and the
intracellular cytoskeleton. These complexes act as primary force
transduction hubs, sensing deformation and matrix stiffness.
Additionally, stretch-activated ion channels (such as Piezo1)
embedded in the cell membrane respond to mechanical perturbation by
rapidly altering ion flux, particularly Ca2+, to
initiate immediate electrochemical signaling (36). On the surface of cells such as
osteocytes and chondrocytes, primary cilia project as non-motile
antennae, exquisitely tuned to sense subtle changes in fluid flow
and pressure. Force detection by these sensors triggers a
sophisticated cascade of intracellular signaling pathways. The
mechanical signal is first propagated through the dynamic
cytoskeleton, a network that distributes tension from the membrane
to the nucleus (37). This
mechanical energy is then converted into chemical signals through
the activation of key mediators.
Mechanotransduction is the sophisticated process
through which cells perceive external mechanical forces and convert
them into intracellular biochemical responses (4,9).
This fundamental mechanism is initiated by mechanosensors, which
are specialized cellular structures that detect mechanical
perturbations. Key sensors include integrins, which tether the
intracellular cytoskeleton to the ECM, forming focal adhesion
complexes that act as primary force transduction hubs (38,39). Additionally, stretch-activated
ion channels (such as Piezo1) rapidly alter ion flux upon membrane
deformation, while primary cilia on chondrocytes and osteocytes
function as cellular antennae, sensing fluid shear stress and
compression (40). Force
detection triggers a cascade of intracellular signaling pathways.
The mechanical signal is propagated via the cytoskeleton, a dynamic
network that distributes tension throughout the cell (37). This leads to the activation of
key mediators such as the Hippo pathway effectors YAP and TAZ,
which translocate to the nucleus to regulate genes responsible for
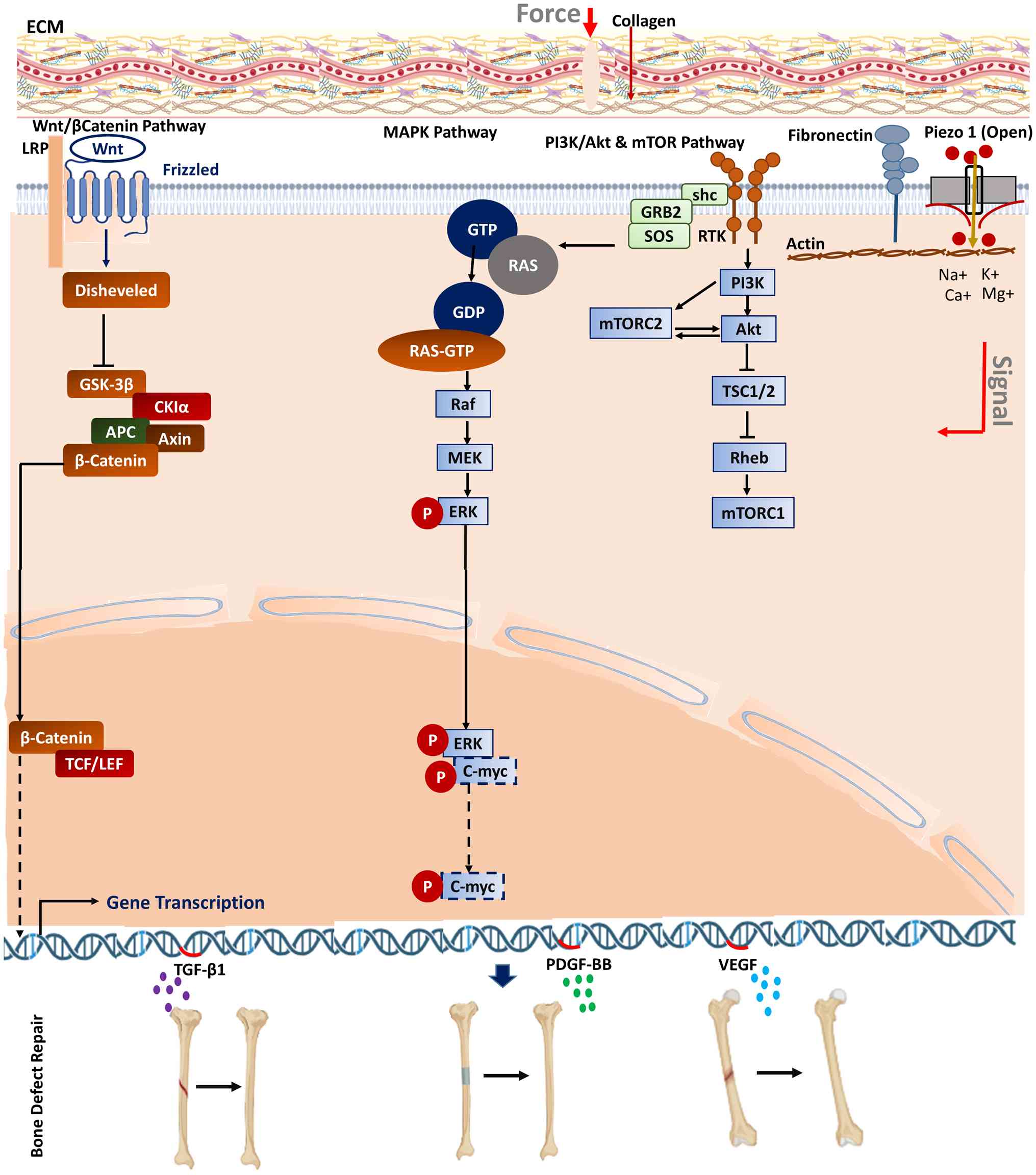
proliferation and matrix synthesis (41,42). Fig. 1 depicts the key signaling
cascades (such as the Wnt/β-catenin and MAPK pathways) activated by
mechanosensitive ion channels, which further modulate cell fate
decisions, including differentiation and apoptosis (43,44). Crucially, mechanotransduction is
bidirectional. Cells not only respond to forces but also actively
exert contractile forces on their surroundings through actomyosin
activity, a concept known as mechanoreciprocity (45,46). This continuous dialogue between
cells and their biomechanical environment is essential for
maintaining tissue homeostasis and is a critical target for guiding
regenerative outcomes in orthopedic tissues (47). Key cellular mechanosensors and
signaling pathways are shown in Table I (48-56).
The ECM is far more than a passive structural
scaffold; it is a dynamic and active mediator essential for
cellular mechanical sensing. The composition, architecture and
physical properties of the ECM fundamentally govern how mechanical
forces are transmitted, attenuated or amplified before reaching
cellular mechanosensors (57).
The stiffness of the ECM, or elastic modulus, provides a critical
physical cue that directly influences cell fate. For instance, MSCs
can sense this rigidity through integrin-mediated adhesions, a
process known as durotaxis, which directs them toward osteogenic
differentiation on stiffer, bone-mimetic substrates or adipogenesis
on softer substrates (58).
Beyond static properties, the viscoelasticity of the ECM, its
ability to exhibit both elastic solid and viscous fluid behaviors,
allows it to absorb and distribute energy from dynamic loading
(59). This time-dependent
response protects cells from sudden, damaging impacts while
facilitating the transfer of beneficial, rhythmic strains.
Furthermore, the molecular organization of the ECM is pivotal. The
specific arrangement of fibrillar collagens, proteoglycans and
glycoproteins creates a unique architectural landscape that filters
mechanical signals (60). This
organized network ensures that forces such as tension or
compression are not merely felt as blunt pressure but are
translated into specific, spatially guided biochemical
instructions.
Crucially, the matrix acts as a biochemical
reservoir that works in concert with mechanical inputs. Embedded
growth factors and bioactive peptides are often sequestered within
the ECM and can be released or activated in response to mechanical
deformation (61). The
mechanosensitive signaling triggers the production of growth
factors including TGF-β1, platelet-derived growth factor-BB and
VEGF, which are released into the circulation and transported to
the injury site to promote bone lengthening and regeneration
(62). This process, termed
mechano-chemo transduction, creates a synergistic effect where
physical forces directly modulate the local biochemical
microenvironment. For example, mechanical strain can liberate TGF-β
from its latent binding proteins in the matrix, thereby
simultaneously providing a mechanical and a chemical stimulus for
tissue repair (63). Therefore,
the ECM is an indispensable partner in mechanotransduction; it
functions as a sophisticated signal processor that contextualizes
external mechanical loads, ensuring that the subsequent
intracellular signaling cascades and transcriptional responses are
appropriate for maintaining tissue homeostasis or initiating
regeneration (60). This central
role makes the rational design of ECM-mimetic biomaterials a
paramount strategy in regenerative orthopedics.
The regenerative processes in bone and cartilage are
guided by mechanotransduction, although the specific cellular
responses differ due to the distinct physiological demands of each
tissue (64-66). In bone regeneration, mechanical
loading is a potent anabolic stimulus (67). Osteocytes, embedded within the
mineralized matrix, act as the primary mechanosensors, detecting
interstitial fluid flow shear stress generated during loading
(68,69). This detection inhibits sclerostin
expression, thereby unleashing the Wnt/β-catenin signaling pathway.
This cascade promotes osteoblastic bone formation and suppresses
osteoclastic resorption, making targeted mechanical stimulation a
critical therapeutic strategy for enhancing fracture healing and
combating osteoporosis (53,70,71). By contrast, cartilage
regeneration presents a more complex mechanobiological challenge
due to its avascular nature and low cellularity (72). Chondrocytes within the
proteoglycan-rich ECM respond optimally to dynamic compression and
hydrostatic pressure, which upregulate anabolic genes for type II
collagen and aggrecan (73,74). However, the response is dependent
on the nature, magnitude and frequency of the load. While
physiological, dynamic loading promotes matrix synthesis and the
chondrogenesis of MSCs, aberrant loading such as high-impact shear
or prolonged static compression induces a catabolic state
characterized by the release of inflammatory cytokines and
matrix-degrading enzymes such as MMP-13, accelerating degeneration
(74). This nuanced
understanding is directly applied in rehabilitative medicine. For
bone, low-magnitude high-frequency vibration and controlled
weight-bearing protocols are used to stimulate healing (75). For cartilage, motion therapies
and continuous passive motion devices are designed to provide
beneficial dynamic compression while avoiding detrimental shear
forces, thereby creating a pro-regenerative mechanical
microenvironment (76).
Mechanical loading is a fundamental regulator of
bone mass and architecture, with osteogenic responses following a
well-established principle whereby bone forms in areas of high
stress and resorbs in areas of disuse (77,78). This adaptive process, governed by
mechanotransduction, is crucial for fracture healing and preventing
osteoporosis (79). Osteocytes,
comprising >90% of bone cells and entombed within lacunae, act
as the orchestrators of this response; they detect minute
deformations of the bone matrix, which cause interstitial fluid to
flow within the canalicular network, generating shear stress across
their extensive dendritic processes (69,80). This mechanical stimulation
triggers a rapid biochemical response. Osteocytes downregulate the
secretion of sclerostin, a key inhibitor of the Wnt/β-catenin
signaling pathway (81,82). The subsequent activation of Wnt
signaling in pre-osteoblasts and lining cells promotes their
proliferation, differentiation and ultimately, bone formation
(83). Concurrently, mechanical
signals suppress osteocyte-supported receptor activator of nuclear
factor κ-B ligand expression, thereby inhibiting osteoclastogenesis
and bone resorption (84).
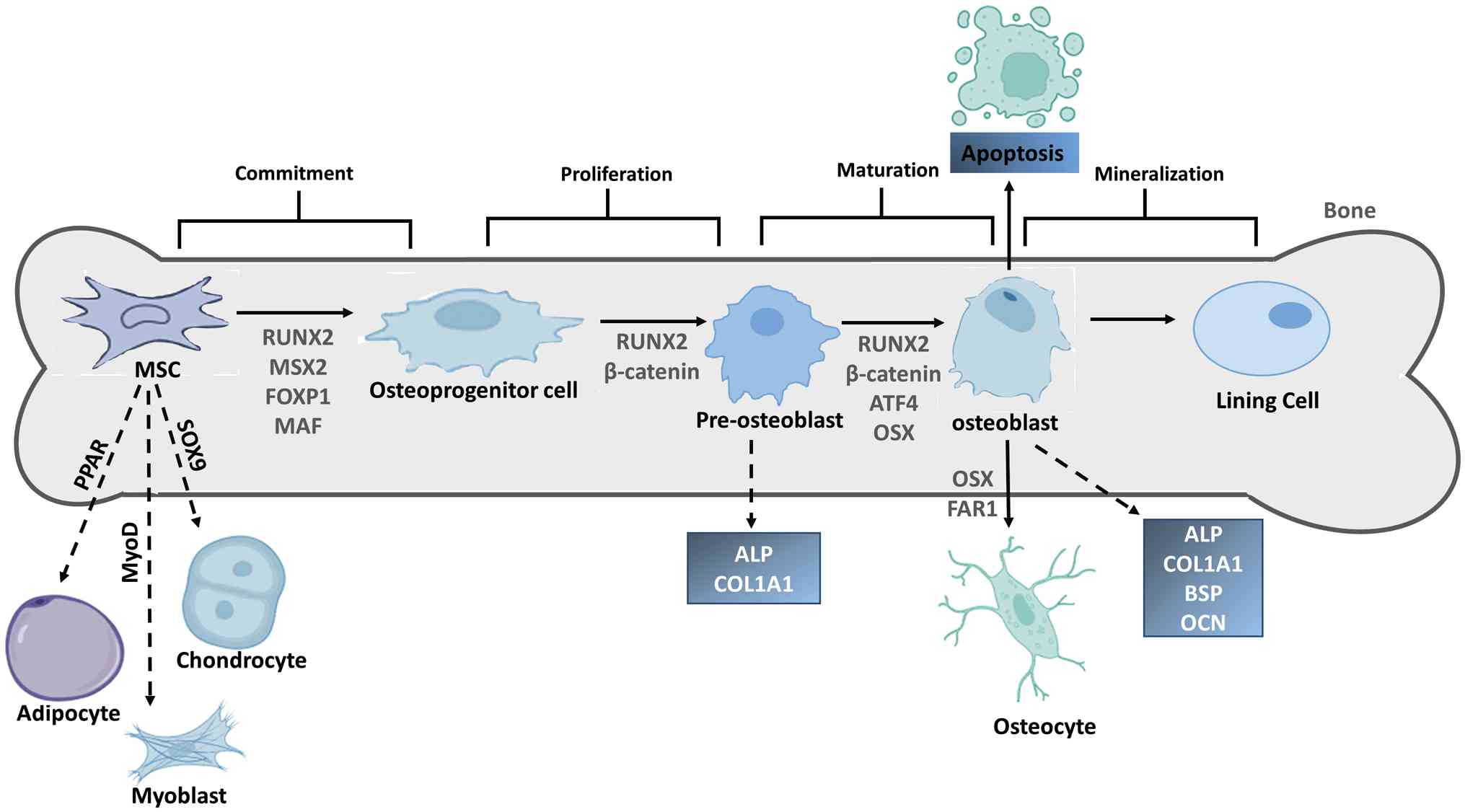
Mechanical stimulation directs MSCs toward becoming bone-forming
osteoblasts. This occurs by activating osteogenic transcription
factors [such as β-catenin and Runt-related transcription factor 2
(RUNX2)] and suppressing regulators of other cell fates (such as
fat or cartilage). As the cells mature from precursors into
functional osteoblasts, they sequentially express specific marker
genes (such as alkaline phosphatase, collagen type I α1 chain and
osteocalcin). The final outcome for an osteoblast is either
programmed cell death or embedding into bone as a lining cell. This
mechanically driven process is essential for bone growth and
healing in rehabilitation (85-87) (Fig. 2). The net result is a powerful
anabolic shift favoring net bone deposition. Therapeutic strategies
in rehabilitation leverage this knowledge. Controlled, dynamic
loading regimens such as those achieved through specific
weight-bearing exercises or low-magnitude, high-frequency vibration
are designed to exceed the minimal effective strain threshold
needed to initiate this anabolic cascade. This targeted
'mechanotherapy' provides a non-pharmacological means to accelerate
fracture callus maturation, enhance bone density around implants
and counteract the bone loss associated with immobilization, making
it a cornerstone of modern orthopedic rehabilitation (88,89).
Chondrocyte mechanobiology is a critical determinant
of success or failure in cartilage repair, presenting a unique
therapeutic paradox (90,91).
Residing within an avascular, aneural ECM, chondrocytes are
sensitive to their mechanical environment (92). The application of physiological
dynamic compression and hydrostatic pressure, mimicking joint
loading during movement, promotes an anabolic response (67). This stimulates the synthesis of
essential matrix components such as aggrecan and type II collagen,
crucial for restoring the load-bearing functionality of the tissue
(93,94). Such mechanical cues are vital for
guiding the chondrogenic differentiation of implanted MSCs in
tissue engineering strategies (95,96). However, the beneficial effects
are critically dependent on load characteristics. Deviations into
abnormal loading patterns, such as high-magnitude impact, shear
stress or prolonged static compression, trigger a starkly
different, catabolic fate. These detrimental forces activate
inflammatory pathways (such as the NF-κB pathway) and upregulate
matrix-degrading enzymes (MMPs and ADAMTS), leading to the
breakdown of the very matrix regenerative therapies aim to build
(97,98). This dichotomy underscores the
importance of precise rehabilitative loading. Protocols employing
motion therapy and continuous passive motion are designed to
deliver pro-anabolic stimuli while meticulously avoiding the
destructive shear and inflammatory stress that hinder repair and
accelerate post-traumatic osteoarthritis.
Tendon and ligament healing are a mechanosensitive
process where the precise application of load is paramount for
restoring functional strength and preventing dysfunctional scar
tissue (99-101). These densely collagenous,
hypovascular tissues rely on mechanotransduction to guide repair
(102). Tenocytes and ligament
fibroblasts possess an array of mechanosensors, including integrins
and stretch-activated ion channels, which detect changes in tension
and strain during movement (103). Early, controlled mechanical
loading stimulates the production and organized alignment of
collagen fibrils, enhancing the tensile properties of the repair
and promoting a more regenerative rather than purely scar-forming
outcome (104,105). Conversely, the absence of load
(immobilization) leads to tissue atrophy, matrix disorganization
and adhesion formation (104).
As shown in Fig. 3, physical
force is converted into a biochemical signal by bone cells through
mechanotransduction, and surface sensors stimulate the
intracellular cascade that activates transcription factors to
upregulate osteogenic gene expression. However, excessive or
premature loading can be equally detrimental, provoking reinjury,
inflammation and metaplasia (106). The therapeutic window is
narrow. Therefore, rehabilitation protocols are designed to
leverage mechanotransduction carefully. Techniques such as early
controlled motion and progressive loading regimens apply precise
biomechanical cues to activate pro-reparative signaling pathways in
tenocytes and resident stem cells. This promotes collagen synthesis
and maturation while steering the healing process away from the
weak, fibrotic scar tissue that characterizes poor functional
recovery, making mechanotherapy a cornerstone of effective tendon
and ligament rehabilitation (107,108).
Mechanical loading is a potent regulator of stem
cell fate, serving as a critical determinant in their commitment to
specific lineages essential for musculoskeletal repair (30). The differentiation of MSCs is not
solely governed by biochemical cues; the physical forces present in
their microenvironment provide instructive signals that can
override soluble factors (109). For instance, substrate
stiffness is a primary mechanical cue. MSCs cultured on substrates
mimicking the stiffness of bone tissue tend to undergo
osteogenesis, upregulating RUNX2 and osteocalcin expression
(110). By contrast, softer
substrates that resemble brain or fat tissue promote neurogenesis
or adipogenesis, respectively (111). This phenomenon, known as
durotaxis, highlights how cells sense and migrate along stiffness
gradients, a principle vital for designing biomaterials in tissue
engineering (112,113). Beyond static stiffness, dynamic
mechanical forces such as cyclic tensile strain, compression and
fluid shear stress directly activate mechanosensitive pathways that
dictate lineage specification (114,115). Applied cyclic strain promotes
tenogenic and osteogenic differentiation by activating pathways
such as focal adhesion kinase/MAPK and RhoA/Rho-associated
coiled-coil-containing protein kinase, which influence cytoskeletal
tension and nuclear translocation of transcription factors
(116). Fluid shear stress,
crucial in vascular and bone environments, enhances osteogenesis by
stimulating prostaglandin release and activating Wnt/β-catenin
signaling (117,118). Even low-intensity vibrations
have been shown to promote osteogenic differentiation while
suppressing adipogenesis, illustrating the finely tuned nature of
mechanical input (119).
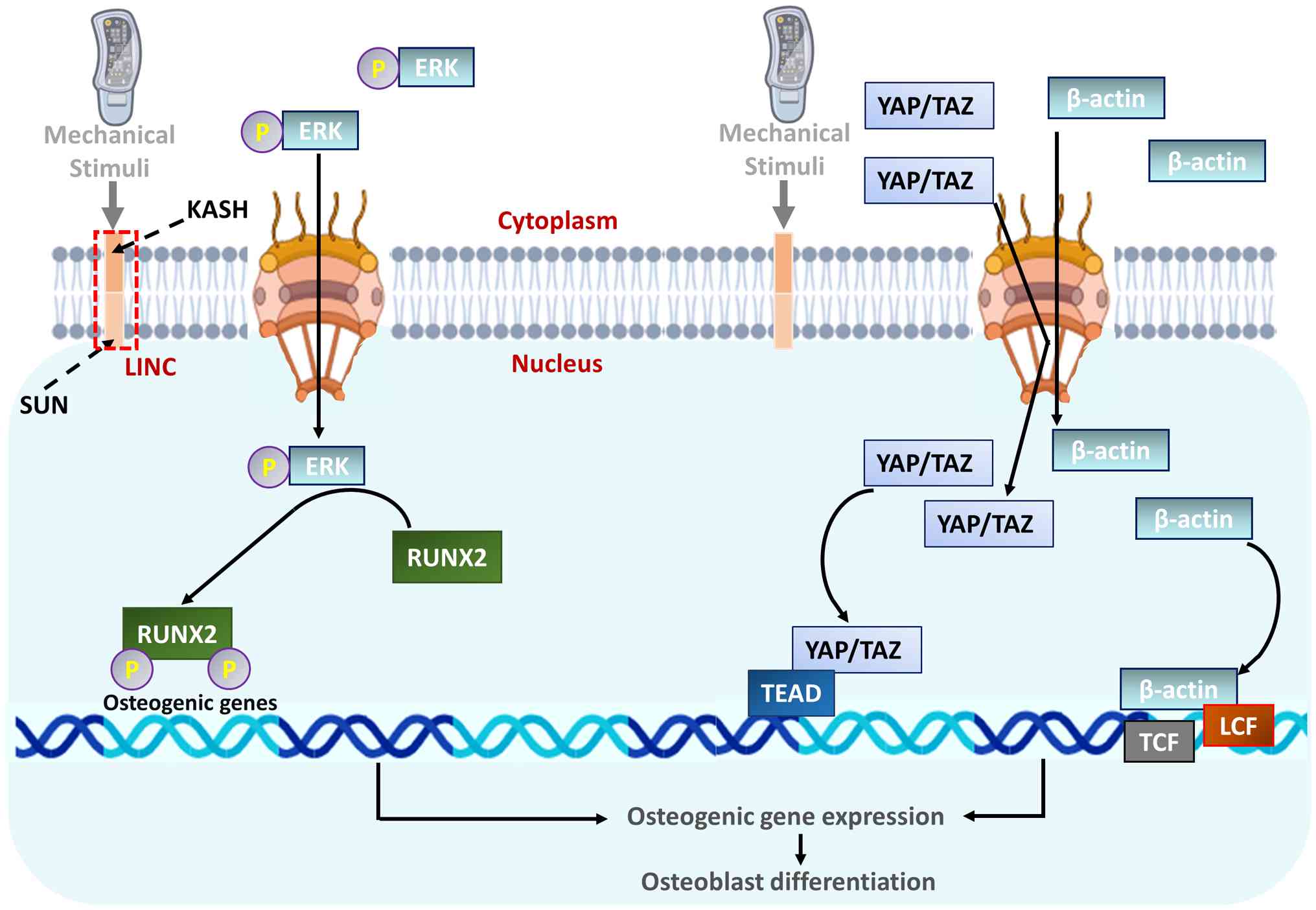
Mechanical forces are transmitted from the cell cytoskeleton
(F-actin) to the nucleus through the linker of nucleoskeleton and
cytoskeleton complex, causing the nucleus to deform (120). This strain increases the
permeability of nuclear pores, allowing for faster import of
critical transcription factors (120). As a result, mechanosensitive
regulators such as YAP/TAZ and β-catenin accumulate in the nucleus
(121). There, they initiate
osteogenic genetic programs. At the same time, phosphorylated RUNX2
binds to DNA, prompting chromatin to remodel into an open state.
This open conformation further activates the transcription of genes
that are essential for bone formation (Fig. 4). The implications for
regenerative medicine are profound. In bioreactors for tissue
engineering, mechanical conditioning such as cyclic stretching of
tendon grafts or fluid flow perfusion in bone scaffolds is used to
pre-condition stem cell-seeded constructs, promoting
differentiation and matrix maturation before implantation (122). In clinical rehabilitation,
understanding how specific exercise-induced loading regimens
influence endogenous stem cell pools can lead to targeted therapies
that harness mechanical cues to guide tissue repair, offering a
non-invasive strategy to enhance regenerative outcomes in
orthopedic healing (123,124).
The integration of mechanobiology principles into
regenerative medicine has given rise to innovative biomechanical
strategies designed to orchestrate tissue repair by harnessing the
power of mechanical forces (125,126). These approaches move beyond
passive structural support, aiming to actively direct cellular
behavior through precisely controlled physical cues. A central
strategy involves the development of smart biomaterial scaffolds.
These are not inert structures but are engineered with specific
mechanical properties such as tunable stiffness, viscoelasticity
and microtopography that mimic the native ECM of the target tissue
(127,128). For instance, a scaffold
designed for bone regeneration is designed to be rigid to promote
osteogenesis, while a cartilage scaffold requires a compliant,
hydrogel-based environment to support chondrogenesis (129). Furthermore, these scaffolds can
be functionalized with tethered bioactive molecules that are
mechanically activated upon cell adhesion or scaffold stretching,
creating a dynamic feedback loop with resident cells (130,131). Beyond static design, dynamic
bioreactor systems are a cornerstone of in vitro tissue
engineering. These devices apply biomimetic mechanical stimuli
including cyclic compression, tensile strain and fluid shear stress
to cell-seeded constructs during cultivation (132,133). This process of mechanical
preconditioning promotes stem cell differentiation, enhances ECM
synthesis and organization and yields a more functional and robust
tissue graft prior to implantation (134). For example, tensile bioreactors
are used to generate aligned collagen fibers in engineered
ligaments, significantly improving their ultimate tensile strength
(135,136). Translating these principles to
the clinic, advanced mechanotherapy is revolutionizing
rehabilitation. Techniques such as ESWT and low-intensity pulsed
ultrasound (LIPUS) deliver targeted mechanical energy to injury
sites, activating pro-regenerative mechanotransduction pathways,
enhancing angiogenesis and stimulating stem cell recruitment
(137,138). These biomechanical strategies,
which work in concert with biological cues, represent a paradigm
shift from merely replacing damaged tissue to actively instructing
the innate healing mechanisms of the body, thereby significantly
improving functional outcomes in orthopedic rehabilitation
(139,140).
Physical therapies represent the deliberate clinical
application of mechanotransduction principles, utilizing controlled
mechanical stimuli to directly influence cellular behavior and
guide tissue repair (25,141).
Therapeutic exercise is not merely about strengthening muscles; it
is a precise modality that delivers targeted biomechanical cues to
injured bones, cartilage, tendons and ligaments (25,142). Each movement, whether it is
weight-bearing, resistance training or dynamic motion, generates
specific forces that are detected by cellular mechanosensors, such
as integrins and ion channels (1). This initiates intracellular
signaling cascades that promote anabolic processes, including
collagen synthesis, matrix organization and stem cell
differentiation (143,144). The efficacy of these
interventions hinges on the careful calibration of mechanical
dosing. Rehabilitation protocols are designed to apply loads within
a therapeutic window that stimulate repair without exacerbating
damage. For instance, eccentric loading of tendons promotes aligned
collagen fibril formation, while controlled motion following
cartilage procedures delivers essential dynamic compression that
enhances chondrocyte activity and nutrient diffusion (23,145). By harnessing the body's innate
responsiveness to physical forces, exercise-based therapies provide
a powerful, non-invasive strategy to optimize the regenerative
microenvironment, making them a cornerstone of modern orthopedic
rehabilitation.
The principles of mechanotransduction are directly
applied in orthopedic rehabilitation to enhance healing and
functional recovery (7,25). Clinicians utilize controlled
mechanical loading through tailored exercise regimens to stimulate
cellular repair processes across various tissues. Following
fracture fixation, progressive weight-bearing is prescribed to
generate osteogenic fluid shear stress, promoting callus formation
and bone remodeling (146,147). This approach harnesses the
mechanosensitivity of osteocytes to guide structural adaptation. In
soft tissue injuries, specific loading protocols are fundamental
(141,148). For tendinopathies, eccentric
strengthening exercises apply controlled tensile strains that
upregulate collagen production in tenocytes, improving tendon
fibril alignment and tensile strength (149). Similarly, postoperative
rehabilitation after cartilage repair procedures incorporates
continuous passive motion and carefully graded active exercises
(150,151). These interventions deliver
essential dynamic compression and hydrostatic pressure to
chondrocytes, supporting matrix synthesis while preventing the
formation of adhesions and fibrous tissue (23,152). These clinical strategies
exemplify mechanotherapy, where externally applied forces are
translated into biochemical signals that drive anabolic cellular
activity. By modulating the intensity, frequency and type of
mechanical stimulus, rehabilitation specialists can optimize the
tissue microenvironment to support regeneration, reduce recovery
time and improve long-term functional outcomes for patients with
musculoskeletal injuries (Table
II) (153-158).
Mechanotherapy is a targeted therapeutic approach
that applies controlled mechanical forces to directly influence the
biological process of fracture repair (159,160). Following a fracture, the
carefully timed introduction of specific mechanical stimuli is
crucial for guiding callus formation, mineralization and eventual
remodeling. This strategy harnesses the innate mechanosensitivity
of bone cells, particularly osteocytes, which act as primary
sensors of changes in their mechanical environment. Clinical
applications begin with an initial period of relative stabilization
to allow early callus formation, followed by the progressive
introduction of load (161).
Controlled weight-bearing and resistance exercises are prescribed
to generate intermittent hydrostatic pressure and fluid shear
stress within the porous network of the bone (162,163). These mechanical cues are
detected by osteocytes, triggering intracellular signaling cascades
that downregulate sclerostin expression. The subsequent activation
of the Wnt/β-catenin pathway promotes osteoblast differentiation
and activity, accelerating bone formation while simultaneously
inhibiting osteoclastic bone resorption (53,164). Advanced modalities such as
LIPUS and PEMFs provide non-invasive mechanical and electrical
stimulation to the fracture site (165). These techniques enhance
cellular proliferation, angiogenesis and matrix synthesis,
particularly in cases of delayed union or non-union. By precisely
modulating the mechanical microenvironment, mechanotherapy offers a
powerful, non-pharmacological method to optimize the innate healing
capacity of the body, reduce recovery time and improve structural
outcomes in fracture management.
Mechanically assisted tissue engineering represents
a paradigm shift in regenerative medicine, moving beyond passive
scaffolds to dynamic systems that actively instruct cellular
behavior through applied physical forces. This approach recognizes
that mechanical cues are as critical as biochemical signals in
directing stem cell differentiation and fostering the development
of functional, load-bearing tissues. In vitro, this is
achieved through the use of bioreactors that deliver biomimetic
mechanical conditioning such as cyclic strain for tendons, fluid
shear for bone and dynamic compression for cartilage to cell-seeded
constructs (166). This
preconditioning promotes ECM synthesis, improves structural
organization and enhances the mechanical properties of the
engineered tissue before implantation. The principles extend to
smart scaffold design, where materials are engineered with specific
mechanical properties such as tailored stiffness, elasticity and
degradability that mimic the native tissue environment and provide
ongoing mechanical cues in vivo (167). These scaffolds can be designed
to respond to body movements, thereby continuously stimulating
integrated cells post-implantation. By harnessing
mechanotransduction to guide cellular activity at every stage,
mechanically assisted tissue engineering creates more robust and
biologically integrated grafts, significantly improving their
functional outcomes and success rates in orthopedic repair and
rehabilitation (125,168).
The future of orthopedic rehabilitation lies in
personalizing interventions based on individual mechanobiological
profiles. Personalized mechanotransduction-based therapies move
beyond one-size-fits-all protocols by accounting for
patient-specific factors such as age, genetics, tissue viability
and biomechanics (169).
Advanced imaging and diagnostic technologies enable clinicians to
assess the unique mechanical microenvironment and cellular
responsiveness of the patient, creating a foundation for tailored
rehabilitation strategies. For instance, real-time feedback systems
and wearable sensors can monitor load distribution and movement
patterns during therapeutic exercises. This data, combined with
genetic profiling that identifies variations in mechanosensitive
pathways, allows for the optimization of mechanical dosing
prescribing specific intensities, frequencies and types of loading
that are most likely to stimulate anabolic responses in that
individual (67). In tissue
engineering, this approach translates to 3D-bioprinted scaffolds
customized to match the anatomical and mechanical requirements of
the patient, potentially seeded with autologous cells primed ex
vivo using patient-specific mechanical conditioning (170). By aligning therapeutic
mechanical inputs with the cellular responsiveness of the
individual, these precision interventions maximize regenerative
potential, minimize the risk of re-injury and significantly improve
functional recovery, heralding a new era of truly personalized
orthopedic medicine.
Despite significant advances, translating
mechanotransduction research into clinical practice faces notable
challenges. A primary hurdle is defining the optimal mechanical
dosage, such as the precise intensity, frequency and duration of
loading, required to stimulate anabolic repair without provoking
catabolic damage or inflammation (171). This therapeutic window varies
significantly between tissues, individuals and even stages of
healing. Furthermore, the complex, interdependent nature of
mechanosignaling pathways makes it difficult to isolate specific
therapeutic targets (65).
Future progress hinges on developing more sophisticated smart
biomaterials that can dynamically respond to in vivo
mechanical cues and deliver bioactive factors in a
feedback-controlled manner (172).
The ultimate objective is to create a closed-loop
system where AI-driven algorithms analyze this multifaceted data to
prescribe and dynamically adjust mechanical dosing in real-time
(173). This will ensure that
the stimulus remains within the patient-specific therapeutic window
throughout the healing process, which evolves from the inflammatory
phase to remodeling. Furthermore, this principle extends to ex
vivo tissue engineering, where bioreactors can apply
patient-specific mechanical conditioning to stem cell-seeded
constructs, pre-adapting them to the mechanical demands they will
encounter upon implantation (174). By moving beyond a
one-size-fits-all model, these optimized, data-driven protocols
will maximize regenerative potential, minimize the risk of
re-injury and significantly accelerate functional recovery,
heralding a new era of precision orthopedics.
The present review established mechanotransduction
as the pivotal mechanism linking mechanical forces to cellular
regeneration in orthopedic rehabilitation. The sophisticated
interplay between cellular sensors, signaling pathways and the ECM
enables physical stimuli to direct tissue repair and adaptation.
The translation of these principles into mechanotherapy and
biomechanically-informed biomaterials represents a notable
advancement beyond traditional rehabilitation. Looking forward, the
field must overcome the challenge of defining optimal, personalized
mechanical dosing. The future lies in integrating real-time
biomechanical monitoring with patient-specific profiling to create
dynamic, adaptive treatment protocols. The convergence of smart
biomaterials, AI-driven modeling and a deeper systems-level
understanding of mechanobiological networks will enable truly
predictive and personalized regenerative interventions. This
evolution towards precision mechanotherapy promises to
revolutionize musculoskeletal care by optimally harnessing the
innate healing mechanisms of the body. This will ultimately enable
clinicians to precisely harness the innate mechanoresponsive
capacity, offering more effective, non-invasive strategies to
restore function and revolutionize patient outcomes in
musculoskeletal medicine.
Not applicable.
BW and XZ conceived the review, designed the
manuscript writing structure and drafted the manuscript. HL, LL and
TL edited and revised the manuscript. YL, QF, YC and BD
participated in the literature search and analysis of literature
content to be included in the review. All authors read and approved
the final version of the manuscript. Data authentication is not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
No funding was received.
|
1
|
Kacprzak B and Stańczak M: Knee Joint
Response to MechanicalLoading: Bounding Mechanotransduction with
Rehabilitation. 2024, Available from: https://www.preprints.org/manuscript/202409.0995/v1.
|
|
2
|
Sueki D and Brechter J: Orthopedic
Rehabilitation Clinical Advisor. 1st edtion. Elsevier Health
Sciences; Amsterdam: 2009
|
|
3
|
d'Agostino M, Craig K, Tibalt E and
Respizzi S: Shock wave as biological therapeutic tool: From
mechanical stimulation to recovery and healing, through
mechanotransduction. Int J Surg. 24:147–153. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang N: Review of cellular
mechanotransduction. J Phys D Appl Phys. 50:2330022017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yang Y, Wu Y, Zhou K, Wu D, Yao X, Heng
BC, Zhou J, Liu H and Ouyang H: Interplay of forces and the immune
response for functional tendon regeneration. Front Cell Dev Biol.
9:6576212021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Huang X, Das R, Patel A and Duc Nguyen T:
Physical stimulations for bone and cartilage regeneration. Regen
Eng Transl Med. 4:216–237. 2018. View Article : Google Scholar
|
|
7
|
Glatt V, Evans CH and Stoddart MJ:
Regenerative rehabilitation: The role of mechanotransduction in
orthopaedic regenerative medicine. J Orthop Res. 37:1263–1269.
2019. View Article : Google Scholar
|
|
8
|
Gulrandhe P, Acharya S, Phansopkar P and
Naqvi W: Exploring the dynamic concept of mechanobiology in
regenerative rehabilitation: A narrative review. J Clin Diagn Res.
18:KE01–KE04. 2024.
|
|
9
|
Martino F, Perestrelo AR, Vinarský V,
Pagliari S and Forte G: Cellular mechanotransduction: From tension
to function. Front Physiol. 9:8242018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
White CR and Frangos JA: The shear stress
of it all: The cell membrane and mechanochemical transduction.
Philos Trans R Soc Lond B Biol Sci. 362:1459–1467. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Raman N, Imran SAM, Ahmad Amin Noordin KB,
Zaman WSWK and Nordin F: Mechanotransduction in mesenchymal stem
cells (MSCs) differentiation: A review. Int J Mol Sci. 23:45802022.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu BY, Jin Y, Ma XH, Wang CY, Guo Y and
Zhou D: The potential role of mechanically sensitive ion channels
in the physiology, injury, and repair of articular cartilage. J
Orthop Surg (Hong Kong). 28:23094990209502622020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Piersma B, Bank RA and Boersema M:
Signaling in fibrosis: TGF-β, WNT, and YAP/TAZ converge. Front Med
(Lausanne). 2:592015.
|
|
14
|
Heng BC, Zhang X, Aubel D, Bai Y, Li X,
Wei Y, Fussenegger M and Deng X: An overview of signaling pathways
regulating YAP/TAZ activity. Cell Mol Life Sci. 78:497–512. 2021.
View Article : Google Scholar
|
|
15
|
Jiang L, Li J, Zhang C, Shang Y and Lin J:
YAP-mediated crosstalk between the Wnt and Hippo signaling pathways
(review). Mol Med Rep. 22:4101–4106. 2020.PubMed/NCBI
|
|
16
|
Xie W, Wei X, Kang H, Jiang H, Chu Z, Lin
Y, Hou Y and Wei Q: Static and dynamic: Evolving biomaterial
mechanical properties to control cellular mechanotransduction. Adv
Sci (Weinh). 10:22045942023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang T, Nanda SS, Papaefthymiou GC and Yi
DK: Mechanophysical cues in extracellular matrix regulation of cell
behavior. Chembiochem. 21:1254–1264. 2020. View Article : Google Scholar
|
|
18
|
Li G: Novel applications of distraction
histogenesis. Orthop Proc. 107-B(Suppl 9): S852025. View Article : Google Scholar
|
|
19
|
Sun Y, Wan B, Wang R, Zhang B, Luo P, Wang
D, Nie JJ, Chen D and Wu X: Mechanical stimulation on mesenchymal
stem cells and surrounding microenvironments in bone regeneration:
Regulations and applications. Front Cell Dev Biol. 10:8083032022.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ma Q, Miri Z, Haugen HJ, Moghanian A and
Loca D: Significance of mechanical loading in bone fracture
healing, bone regeneration, and vascularization. J Tissue Eng.
14:204173142311725732023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pagnotti GM, Styner M, Uzer G, Patel VS,
Wright LE, Ness KK, Guise TA, Rubin J and Rubin CT: Combating
osteoporosis and obesity with exercise: Leveraging cell
mechanosensitivity. Nat Rev Endocrinol. 15:339–355. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yu Y, Feng T, Qiu H, Gu Y, Chen Q, Zuo C
and Ma H: Simultaneous photoacoustic and ultrasound imaging: A
review. Ultrasonics. 139:1072772024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Anderson DE and Johnstone B: Dynamic
mechanical compression of chondrocytes for tissue engineering: A
critical review. Front Bioeng Biotechnol. 5:762017. View Article : Google Scholar
|
|
24
|
Li Y, Frank EH, Wang Y, Chubinskaya S,
Huang HH and Grodzinsky AJ: Moderate dynamic compression inhibits
pro-catabolic response of cartilage to mechanical injury, tumor
necrosis factor-α and interleukin-6, but accentuates degradation
above a strain threshold. Osteoarthritis Cartilage. 21:1933–1941.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Thompson WR, Scott A, Loghmani MT, Ward SR
and Warden SJ: Understanding mechanobiology: Physical therapists as
a force in mechanotherapy and musculoskeletal regenerative
rehabilitation. Phys Ther. 96:560–569. 2016. View Article : Google Scholar :
|
|
26
|
Chatterjee M, Muljadi PM and
Andarawis-Puri N: The role of the tendon ECM in
mechanotransduction: Disruption and repair following overuse.
Connect Tissue Res. 63:28–42. 2022. View Article : Google Scholar
|
|
27
|
Kjaer M: Role of extracellular matrix in
adaptation of tendon and skeletal muscle to mechanical loading.
Physiol Rev. 84:649–698. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kong W, Lyu C, Liao H and Du Y: Collagen
crosslinking: Effect on structure, mechanics and fibrosis
progression. Biomed Mater. 16:0620052021. View Article : Google Scholar
|
|
29
|
Ren C, Liu F, Zhang S, Xu H and Yang P:
The role of collagen matrix in the development and progression of
heterotopic ossification in tendon and its biological mechanisms.
FASEB J. 39:e708732025. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Vining KH and Mooney DJ: Mechanical forces
direct stem cell behaviour in development and regeneration. Nat Rev
Mol Cell Biol. 18:728–742. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liao S, Chan CK and Ramakrishna S: Stem
cells and biomimetic materials strategies for tissue engineering.
Mater Sci Eng C. 28:1189–1202. 2008. View Article : Google Scholar
|
|
32
|
Castro N, Ribeiro S, Fernandes M, Ribeiro
C, Cardoso V, Correia V, Minguez R and Lanceros-Mendez S:
Physically active bioreactors for tissue engineering applications.
Adv Biosyst. 4:e20001252020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rosso F, Bonasia DE, Marmotti A, Cottino U
and Rossi R: Mechanical stimulation (pulsed electromagnetic fields
'PEMF' and extracorporeal shock wave therapy 'ESWT') and tendon
regeneration: A possible alternative. Front Aging Neurosci.
7:2112015. View Article : Google Scholar
|
|
34
|
Nosrati H and Nosrati M: Artificial
intelligence in regenerative medicine: Applications and
implications. Biomimetics (Basel). 8:4422023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xiao R, Liu J and Xu XS: Mechanosensitive
GPCRs and ion channels in shear stress sensing. Curr Opin Cell
Biol. 84:1022162023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Poole K: The diverse physiological
functions of mechanically activated ion channels in mammals. Annu
Rev Physiol. 84:307–329. 2022. View Article : Google Scholar
|
|
37
|
Janmey PA: The cytoskeleton and cell
signaling: Component localization and mechanical coupling. Physiol
Rev. 78:763–781. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Schwartz MA: Integrins and extracellular
matrix in mechanotransduction. Cold Spring Harb Perspect Biol.
2:a0050662010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kechagia JZ, Ivaska J and Roca-Cusachs P:
Integrins as biomechanical sensors of the microenvironment. Nat Rev
Mol Cell Biol. 20:457–473. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Verbruggen SW, Sittichokechaiwut A and
Reilly GC: Osteocytes and primary cilia. Curr Osteoporos Rep.
21:719–730. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Varelas X: The Hippo pathway effectors TAZ
and YAP in development, homeostasis and disease. Development.
141:1614–1626. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kwon H, Kim J and Jho Eh: Role of the
Hippo pathway and mechanisms for controlling cellular localization
of YAP/TAZ. FEBS J. 289:5798–5818. 2022. View Article : Google Scholar
|
|
43
|
Garcin CL and Habib SJ: A comparative
perspective on Wnt/β-catenin signalling in cell fate determination.
Asymmetric Cell Division in Development, Differentiation and
Cancer, Results and Problems in Cell Differentiation. Springer
International Publishing; Cham: pp. 323–350. 2017, View Article : Google Scholar
|
|
44
|
Shah N, Morsi Y and Manasseh R: From
mechanical stimulation to biological pathways in the regulation of
stem cell fate. Cell Biochem Funct. 32:309–325. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Van Helvert S, Storm C and Friedl P:
Mechanoreciprocity in cell migration. Nat Cell Biol. 20:8–20. 2018.
View Article : Google Scholar
|
|
46
|
Murrell M, Oakes PW, Lenz M and Gardel ML:
Forcing cells into shape: The mechanics of actomyosin
contractility. Nat Rev Mol Cell Biol. 16:486–498. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Butler DL, Goldstein SA, Guldberg RE, Guo
XE, Kamm R, Laurencin CT, McIntire LV, Mow VC, Nerem RM, Sah RL, et
al: The impact of biomechanics in tissue engineering and
regenerative medicine. Tissue Eng Part B Rev. 15:477–484. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ramage L: Integrins and extracellular
matrix in mechanotransduction. Cell Health Cytoskelet. 4:1–9.
2012.
|
|
49
|
Kanchanawong P and Calderwood DA:
Organization, dynamics and mechanoregulation of integrin-mediated
cell-ECM adhesions. Nat Rev Mol Cell Biol. 24:142–161. 2023.
View Article : Google Scholar
|
|
50
|
Li J, Huang S and Chen H: Advances in
imaging techniques for mammalian/human ciliated cell's cilia:
Insights into structure, function, and dynamics. Biology (Basel).
14:5212025.PubMed/NCBI
|
|
51
|
Abou Alaiwi WA, Lo ST and Nauli SM:
Primary cilia: Highly sophisticated biological sensors. Sensors
(Basel). 9:7003–7020. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Piccolo S, Dupont S and Cordenonsi M: The
biology of YAP/TAZ: Hippo signaling and beyond. Physiol Rev.
94:1287–1312. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hu L, Chen W, Qian A and Li YP:
Wnt/β-catenin signaling components and mechanisms in bone
formation, homeostasis, and disease. Bone Res. 12:392024.
View Article : Google Scholar
|
|
54
|
Duan P and Bonewald L: The role of the
wnt/β-catenin signaling pathway in formation and maintenance of
bone and teeth. Int J Biochem Cell Biol. 77:23–29. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Etienne-Manneville S: Actin and
microtubules in cell motility: Which one is in control? Traffic.
5:470–477. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hohmann T and Dehghani F: The
cytoskeleton-a complex inter-acting meshwork. Cells. 8:3622019.
View Article : Google Scholar
|
|
57
|
Mierke CT: Extracellular matrix cues
regulate mechanosensing and mechanotransduction of cancer cells.
Cells. 13:962024. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Vincent LG, Choi YS, Alonso-Latorre B, del
Álamo JC and Engler AJ: Mesenchymal stem cell durotaxis depends on
substrate stiffness gradient strength. Biotechnol J. 8:472–484.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Chaudhuri O, Cooper-White J, Janmey PA,
Mooney DJ and Shenoy VB: Effects of extracellular matrix
viscoelasticity on cellular behaviour. Nature. 584:535–546. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Berdiaki A, Neagu M, Tzanakakis P,
Spyridaki I, Pérez S and Nikitovic D: Extracellular matrix
components and mechanosensing pathways in health and disease.
Biomolecules. 14:11862024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Teixeira SPB, Domingues RMA, Shevchuk M,
Gomes ME, Peppas NA and Reis RL: Biomaterials for sequestration of
growth factors and modulation of cell behavior. Adv Funct Mater.
30:19090112020. View Article : Google Scholar
|
|
62
|
Cipitria A and Salmeron-Sanchez M:
Mechanotransduction and growth factor signalling to engineer
cellular microenvironments. Adv Healthc Mater. 6:17000522017.
View Article : Google Scholar
|
|
63
|
Hinz B: The extracellular matrix and
transforming growth factor-β1: Tale of a strained relationship.
Matrix Biol. 47:54–65. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Selig M, Lauer JC, Hart ML and Rolauffs B:
Mechanotransduction and stiffness-sensing: Mechanisms and
opportunities to control multiple molecular aspects of cell
phenotype as a design cornerstone of cell-instructive biomaterials
for articular cartilage repair. Int J Mol Sci. 21:53992020.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Hodgkinson T, Kelly DC, Curtin CM and
O'Brien FJ: Mechanosignalling in cartilage: An emerging target for
the treatment of osteoarthritis. Nat Rev Rheumatol. 18:67–84. 2022.
View Article : Google Scholar
|
|
66
|
Zhao Z, Li Y, Wang M, Zhao S, Zhao Z and
Fang J: Mechanotransduction pathways in the regulation of cartilage
chondrocyte homoeostasis. J Cell Mol Med. 24:5408–5419. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ozcivici E, Luu YK, Adler B, Qin YX, Rubin
J, Judex S and Rubin CT: Mechanical signals as anabolic agents in
bone. Nat Rev Rheumatol. 6:50–59. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Klein-Nulend J and Bakker AD: Osteocytes:
Mechanosensors of bone and orchestrators of mechanical adaptation.
Clin Rev Bone Miner Metab. 5:195–209. 2007. View Article : Google Scholar
|
|
69
|
Ganesh T, Laughrey LE, Niroobakhsh M and
Lara-Castillo N: Multiscale finite element modeling of mechanical
strains and fluid flow in osteocyte lacunocanalicular system. Bone.
137:1153282020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Wu Z, Li W, Jiang K, Lin Z, Qian C, Wu M,
Xia Y, Li N, Zhang H, Xiao H, et al: Regulation of bone
homeostasis: Signaling pathways and therapeutic targets. MedComm
(2020). 5:e6572024. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Gao Y, Chen N, Fu Z and Zhang Q: Progress
of Wnt signaling pathway in osteoporosis. Biomolecules. 13:4832023.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Nguyen MT, Gronthos S, Zhao Y,
Chandrakanthan V, Truong VK and Vasilev K: Overcoming challenges in
cartilage regeneration: The role of chondrogenic inducers. Bioeng
Transl Med. e700792025. View Article : Google Scholar
|
|
73
|
Guilak F and Hung CT: Physical regulation
of cartilage metabolism. Basic Orthopaedic Biomechanics &
Mechano-Biology. Mow VC and Huiskes R: Lippincott Williams &
Wilkins; Philadelphia, PA: pp. 2592005
|
|
74
|
Iseki T, Rothrauff BB, Kihara S, Sasaki H,
Yoshiya S, Fu FH, Tuan RS and Gottardi R: Dynamic compressive
loading improves cartilage repair in an in vitro model of
microfracture: Comparison of 2 mechanical loading regimens on
simulated microfracture based on fibrin gel scaffolds encapsulating
connective tissue progenitor cells. Am J Sports Med. 47:2188–2199.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Shi HF, Cheung WH, Qin L, Leung AHC and
Leung KS: Low-magnitude high-frequency vibration treatment augments
fracture healing in ovariectomy-induced osteoporotic bone. Bone.
46:1299–1305. 2010. View Article : Google Scholar
|
|
76
|
Frank C, Akeson WH, Woo SL, Amiel D and
Coutts RD: Physiology and therapeutic value of passive joint
motion. Clin Orthop Relat Res. 113–125. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Mellon SJ and Tanner K: Bone and its
adaptation to mechanical loading: A review. Int Mater Rev.
57:235–255. 2012. View Article : Google Scholar
|
|
78
|
Pivonka P, Park A and Forwood MR:
Functional adaptation of bone: The mechanostat and beyond.
Multiscale Mechanobiology of Bone Remodeling and Adaptation. CISM
International Centre for Mechanical Sciences; Pivonka P: 578.
Springer, Heidelberg: pp. 1–60. 2017
|
|
79
|
Augat P, Simon U, Liedert A and Claes L:
Mechanics and mechano-biology of fracture healing in normal and
osteoporotic bone. Osteoporos Int. 16(Suppl 2): S36–S43. 2005.
View Article : Google Scholar
|
|
80
|
Yu W, Ou R, Hou Q, Li C, Yang X, Ma Y, Wu
X and Chen W: Multiscale interstitial fluid computation modeling of
cortical bone to characterize the hydromechanical stimulation of
lacunar-canalicular network. Bone. 193:1173862025. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Marini F, Giusti F, Palmini G and Brandi
ML: Role of Wnt signaling and sclerostin in bone and as therapeutic
targets in skeletal disorders. Osteoporos Int. 34:213–238. 2023.
View Article : Google Scholar
|
|
82
|
Burgers TA and Williams BO: Regulation of
Wnt/β-catenin signaling within and from osteocytes. Bone.
54:244–249. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Arya PN, Saranya I and Selvamurugan N:
Crosstalk between Wnt and bone morphogenetic protein signaling
during osteogenic differentiation. World J Stem Cells. 16:102–113.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Honma M, Ikebuchi Y, Kariya Y and Suzuki
H: Regulatory mechanisms of RANKL presentation to osteoclast
precursors. Curr Osteoporos Rep. 12:115–120. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Sun Y, Yuan Y, Wu W, Lei L and Zhang L:
The effects of locomotion on bone marrow mesenchymal stem cell
fate: Insight into mechanical regulation and bone formation. Cell
Biosci. 11:882021. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Chan WCW, Tan Z, To MKT and Chan D:
Regulation and role of transcription factors in osteogenesis. Int J
Mol Sci. 22:54452021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
James AW: Review of signaling pathways
governing MSC osteogenic and adipogenic differentiation.
Scientifica (Cairo). 2013:6847362013.
|
|
88
|
Ozdemir F, Zateri C and Murat S:
Evaluation of the efficacy of therapeutic ultrasound on bone
mineral density in postmenopausal period. Rheumatol Int.
28:361–365. 2008. View Article : Google Scholar
|
|
89
|
Popa M, Cursaru A, Cretu B, Iordache S,
Iacobescu GL, Spiridonica R, Serban B and Cirstoiu C: Enhancing
osteoporosis management: A thorough examination of surgical
techniques and their effects on patient outcomes. Cureus.
16:e596812024.PubMed/NCBI
|
|
90
|
Muthu S, Korpershoek JV, Novais EJ, Tawy
GF, Hollander AP and Martin I: Failure of cartilage regeneration:
Emerging hypotheses and related therapeutic strategies. Nat Rev
Rheumatol. 19:403–416. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Oliveira S, Hinckel BB, Silva FS, Carvalho
Ó and Leal A: A guide to articular cartilage functioning: A
comprehensive review, current challenges and mechanobiological
solutions. Prog Biomed Eng (Bristol). 7:2025.
|
|
92
|
Statham P, Jones E, Jennings LM and Fermor
HL: Reproducing the biomechanical environment of the chondrocyte
for cartilage tissue engineering. Tissue Eng Part B Rev.
28:405–420. 2022. View Article : Google Scholar
|
|
93
|
Kjær M, Langberg H, Heinemeier K, Bayer
ML, Hansen M, Holm L, Doessing S, Kongsgaard M, Krogsgaard MR and
Magnusson SP: From mechanical loading to collagen synthesis,
structural changes and function in human tendon. Scand J Med Sci
Sports. 19:500–510. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Responte DJ, Natoli RM and Athanasiou KA:
Collagens of articular cartilage: Structure, function, and
importance in tissue engineering. Crit Rev Biomed Eng. 35:363–411.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Fahy N, Alini M and Stoddart MJ:
Mechanical stimulation of mesenchymal stem cells: Implications for
cartilage tissue engineering. J Orthop Res. 36:52–63. 2018.
View Article : Google Scholar
|
|
96
|
Kelly DJ and Jacobs CR: The role of
mechanical signals in regulating chondrogenesis and osteogenesis of
mesenchymal stem cells. Birth Defects Res C Embryo Today. 90:75–85.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Fang T, Zhou X, Jin M, Nie J and Li X:
Molecular mechanisms of mechanical load-induced osteoarthritis. Int
Orthop. 45:1125–1136. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Li T, Peng J, Li Q, Shu Y, Zhu P and Hao
L: The mechanism and role of ADAMTS protein family in
osteoarthritis. Biomolecules. 12:9592022. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Jiang F, Zhao H, Zhang P, Bi Y, Zhang H,
Sun S, Yao Y, Zhu X, Yang F, Liu Y, et al: Challenges in
tendon-bone healing: emphasizing inflammatory modulation mechanisms
and treatment. Front Endocrinol (Lausanne). 15:14858762024.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Killian ML, Cavinatto L, Galatz LM and
Thomopoulos S: The role of mechanobiology in tendon healing. J
Shoulder Elbow Surg. 21:228–237. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Woo SLY, Nguyen TD, Papas N and Liang R:
Tissue mechanics of ligaments and tendons. Biomechanics in
ergonomics. 2nd edition. CRC Press; pp. 127–148. 2007
|
|
102
|
Fu S, Panayi A, Fan J, Mayer HF, Daya M,
Khouri RK, Gurtner GC, Ogawa R and Orgill DP: Mechanotransduction
in wound healing: from the cellular and molecular level to the
clinic. Adv Skin Wound Care. 34:67–74. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Li Y, Wu T and Liu S: Identification and
distinction of tenocytes and tendon-derived stem cells. Front Cell
Dev Biol. 9:6295152021. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Montgomery J: Building a better scar:
Re-engineering extracellular matrix structure in dermal scars.
Virginia Polytechnic Institute; 2020
|
|
105
|
Buff-Lindner AH: The Role of Poly N Acetyl
Glucosamine Nanofibers in Cutaneous Wound Healing. Baishideng
Publishing Group Inc.; Pleasanton, CA: 2014
|
|
106
|
Aicale R, Tarantino D and Maffulli N:
Overuse injuries in sport: A comprehensive overview. J Orthop Surg
Res. 13:3092018. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
El Ayadi A, Jay JW and Prasai A: Current
approaches targeting the wound healing phases to attenuate fibrosis
and scarring. Int J Mol Sci. 21:11052020. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Sephel GC and Woodward SC: Repair,
Regeneration, and Fibrosis. Lippincott, Williams & Wilkins;
Baltimore: pp. 84–117. 2001
|
|
109
|
Huang C, Dai J and Zhang XA: Environmental
physical cues determine the lineage specification of mesenchymal
stem cells. Biochim Biophys Acta. 1850:1261–1266. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
El-Rashidy AA, El Moshy S, Radwan IA, Rady
D, Abbass MMS, Dörfer CE and Fawzy El-Sayed KM: Effect of polymeric
matrix stiffness on osteogenic differentiation of mesenchymal
stem/progenitor cells: Concise review. Polymers (Basel).
13:29502021. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Lee J, Abdeen AA, Tang X, Saif TA and
Kilian KA: Matrix directed adipogenesis and neurogenesis of
mesenchymal stem cells derived from adipose tissue and bone marrow.
Acta Biomater. 42:46–55. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Espina JA, Marchant CL and Barriga EH:
Durotaxis: The mechanical control of directed cell migration. FEBS
J. 289:2736–2754. 2022. View Article : Google Scholar :
|
|
113
|
Aubry D, Gupta M, Ladoux B and Allena R:
Mechanical link between durotaxis, cell polarity and anisotropy
during cell migration. Phys Biol. 12:0260082015. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Fang Y, Wu D and Birukov KG:
Mechanosensing and mechanoregulation of endothelial cell functions.
Compr Physiol. 9:873–904. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Chen JC and Jacobs CR: Mechanically
induced osteogenic lineage commitment of stem cells. Stem Cell Res
Ther. 4:1072013. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Niu H, Lin D, Tang W, Ma Y, Duan B, Yuan Y
and Liu C: Surface topography regulates osteogenic differentiation
of MSCs via crosstalk between FAK/MAPK and ILK/β-catenin pathways
in a hierarchically porous environment. ACS Biomater Sci Eng.
3:3161–3175. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Jia YY, Li F, Geng N, Gong P, Huang SJ,
Meng LX, Lan J and Ban Y: Fluid flow modulates the expression of
genes involved in the Wnt signaling pathway in osteoblasts in 3D
culture conditions. Int J Mol Med. 33:1282–1288. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Alfieri R, Vassalli M and Viti F:
Flow-induced mechanotransduction in skeletal cells. Biophys Rev.
11:729–743. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Baskan O, Mese G and Ozcivici E:
Low-intensity vibrations normalize adipogenesis-induced
morphological and molecular changes of adult mesenchymal stem
cells. Proc Inst Mech Eng H. 231:160–168. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Kloc M and Wosik J: Mechanical forces,
nucleus, chromosomes, and chromatin. Biomolecules. 15:3542025.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Cai X, Wang KC and Meng Z:
Mechanoregulation of YAP and TAZ in cellular homeostasis and
disease progression. Front Cell Dev Biol. 9:6735992021. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Ahata B, Kan T, Serefoglu Gun B, Tanyeri
Y, Oktay B, Oktay A and Koc RC: Bioreactors for tissue engineering.
Biomaterials and Tissue Engineering. Gunduz O, Egles C, Pérez RA,
Ficai D and Ustundag CB: Springer International Publishing; Cham:
pp. 259–303. 2023, View Article : Google Scholar
|
|
123
|
Chen J, Zhou R, Feng Y and Cheng L:
Molecular mechanisms of exercise contributing to tissue
regeneration. Signal Transduct Target Ther. 7:3832022. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Head PL: Rehabilitation considerations in
regenerative medicine. Phys Med Rehabil Clin N Am. 27:1043–1054.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Kim S, Uroz M, Bays JL and Chen CS:
Harnessing mechanobiology for tissue engineering. Dev Cell.
56:180–191. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Shafiq M, Ali O, Han SB and Kim DH:
Mechanobiological strategies to enhance stem cell functionality for
regenerative medicine and tissue engineering. Front Cell Dev Biol.
9:7473982021. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Xia T, Liu W and Yang L: A review of
gradient stiffness hydrogels used in tissue engineering and
regenerative medicine. J Biomed Mater Res A. 105:1799–1812. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Janson IA and Putnam AJ: Extracellular
matrix elasticity and topography: Material-based cues that affect
cell function via conserved mechanisms. J Biomed Mater Res A.
103:1246–1258. 2015. View Article : Google Scholar
|
|
129
|
Song J, Li L, Fang L, Zhang E, Zhang Y,
Zhang Z, Vangari P, Huang Y, Tian F, Zhao Y, et al: Advanced
strategies of scaffolds design for bone regeneration. BMEMat.
1:e120462023. View Article : Google Scholar
|
|
130
|
Kim TG, Shin H and Lim DW: Biomimetic
scaffolds for tissue engineering†. Adv Funct Mater. 22:2446–2468.
2012. View Article : Google Scholar
|
|
131
|
Monemian Esfahani A, Rosenbohm J, Reddy K,
Jin X, Bouzid T, Riehl B, Kim E, Lim JY and Yang R: Tissue
regeneration from mechanical stretching of cell-cell adhesion.
Tissue Eng Part C Methods. 25:631–640. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Huang G, Li F, Zhao X, Ma Y, Li Y, Lin M,
Jin G, Lu TJ, Genin GM and Xu F: Functional and biomimetic
materials for engineering of the three-dimensional cell
microenvironment. Chem Rev. 117:12764–12850. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Badekila AK, Kini S and Jaiswal AK:
Fabrication techniques of biomimetic scaffolds in three-dimensional
cell culture: A review. J Cell Physiol. 236:741–762. 2021.
View Article : Google Scholar
|
|
134
|
Schumann D, Kujat R, Nerlich M and Angele
P: Mechanobiological conditioning of stem cells for cartilage
tissue engineering. Biomed Mater Eng. 16(Suppl 4): S37–S52.
2006.PubMed/NCBI
|
|
135
|
Wang T, Gardiner BS, Lin Z, Rubenson J,
Kirk TB, Wang A, Xu J, Smith DW, Lloyd DG and Zheng MH: Bioreactor
design for tendon/ligament engineering. Tissue Eng Part B Rev.
19:133–146. 2013. View Article : Google Scholar
|
|
136
|
Lim WL, Liau LL, Ng MH, Chowdhury SR and
Law JX: Current progress in tendon and ligament tissue engineering.
Tissue Eng Regen Med. 16:549–571. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Qin H, Du L, Luo Z, He Z, Wang Q, Chen S
and Zhu YL: The therapeutic effects of low-intensity pulsed
ultrasound in musculoskeletal soft tissue injuries: Focusing on the
molecular mechanism. Front Bioeng Biotechnol. 10:10804302022.
View Article : Google Scholar :
|
|
138
|
Lei L, Zhang Q, Du M and Li L:
Mechanoregulation of cell fate by low-intensity pulsed ultrasound:
Mechanisms and advances in regenerative medicine. BIO Integr.
6:1–18. 2025. View Article : Google Scholar
|
|
139
|
Malik S: The physics of the human body:
Biomechanics and beyond. Worldw J Phys. 1:53–63. 2020.
|
|
140
|
Glatt V, Evans CH and Tetsworth K: A
concert between biology and biomechanics: The influence of the
mechanical environment on bone healing. Front Physiol. 7:6782017.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Ng JL, Kersh ME, Kilbreath S and Knothe
Tate M: Establishing the basis for mechanobiology-based physical
therapy protocols to potentiate cellular healing and tissue
regeneration. Front Physiol. 8:3032017. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Khan KM and Scott A: Mechanotherapy: How
physical therapists' prescription of exercise promotes tissue
repair. Br J Sports Med. 43:247–252. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Xiao Z and Quarles LD: Physiological
mechanisms and therapeutic potential of bone mechanosensing. Rev
Endocr Metab Disord. 16:115–129. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Mobasheri A, Carter SD, Martín-Vasallo P
and Shakibaei M: Integrins and stretch activated ion channels;
putative components of functional cell surface mechanoreceptors in
articular chondrocytes. Cell Biol Int. 26:1–18. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Lambrianides Y: Temporal Dynamics of
Muscle and Tendon Adaptation to Mechano-Metabolic Stimuli. London
South Bank University; 2024
|
|
146
|
Beeharry MW and Ahmad B: Principles of
fracture healing and fixation: A literature review. Cureus.
16:e762502024.PubMed/NCBI
|
|
147
|
Thakur AJ: The Elements of Fracture
Fixation. 4th edition. Elsevier Health Sciences; India: 2019
|
|
148
|
Qin YX and Zhao J: Mechanobiology in
cellular, molecular, and tissue adaptation. Mechanobiol Med.
1:1000222023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Pourshafie S, Ashnagar Z, Jalaie S and
Bashardoust Tajali S: Effects of eccentric exercises with and
without dry needling approaches at the patients with chronic
rotator cuff tendinopathy. J Bodyw Mov Ther. 42:976–981. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Fazalare JA, Griesser MJ, Siston RA and
Flanigan DC: The use of continuous passive motion following knee
cartilage defect surgery: A systematic review. Orthopedics.
33:8782010. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Howard JS, Mattacola CG, Romine SE and
Lattermann C: Continuous passive motion, early weight bearing, and
active motion following knee articular cartilage repair: Evidence
for clinical practice. Cartilage. 1:276–286. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Elder BD and Athanasiou KA: Hydrostatic
pressure in articular cartilage tissue engineering: From
chondrocytes to tissue regeneration. Tissue Eng Part B Rev.
15:43–53. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Bonewald LF and Johnson ML: Osteocytes,
mechanosensing and Wnt signaling. Bone. 42:606–615. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
154
|
ur Rehman S, Iqbal S, Shahid MU, Jahangir
MS and Malik AL: Cartilage: Structure, function, and the
pathogenesis of osteoarthritis. Advancements in Synovial Joint
Science-Structure, Function, and Beyond. IntechOpen; London: 2024,
View Article : Google Scholar
|
|
155
|
Houpt JB, Gahunia HK and Pritzker KPH:
Physical and rehabilitative therapy for knee articular cartilage
injury and disease. Articular Cartilage of the Knee: Health,
Disease and Therapy. Springer; New York, NY: pp. 235–251. 2020,
View Article : Google Scholar
|
|
156
|
Galloway MT, Lalley AL and Shearn JT: The
role of mechanical loading in tendon development, maintenance,
injury, and repair. J Bone Joint Surg Am. 95:1620–1628. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Gonçalves AI, Righelli L, Reis RL, El Haj
AJ and Gomes ME: Understanding degeneration and healing pathways
for tissue engineered treatment strategies in tendinopathy. Cells
Tissues Organs. 214:459–476. 2025.
|
|
158
|
Burdick JA and Vunjak-Novakovic G:
Engineered microenvironments for controlled stem cell
differentiation. Tissue Eng Part A. 15:205–219. 2009. View Article : Google Scholar :
|
|
159
|
McKay J, Nasb M and Hafsi K:
Mechanobiology-based physical therapy and rehabilitation after
orthobiologic interventions: A narrative review. Int Orthop.
46:179–188. 2022. View Article : Google Scholar
|
|
160
|
Benulič Č, Canton G, Rasio N, Murena L and
Kristan A: Mechanobiology of indirect bone fracture healing under
conditions of relative stability: A narrative review for the
practicing clinician. Acta Biomed. 92:e20215822022.
|
|
161
|
Claes L, Eckert-Hübner K and Augat P: The
effect of mechanical stability on local vascularization and tissue
differentiation in callus healing. J Orthop Res. 20:1099–1105.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Zhang B, Mateus J and Hargens A:
Osteoporosis, circulation, and fluid dynamics. Skeletal Circulation
in Clinical Practice. World Scientific; pp. 253–282. 2016,
View Article : Google Scholar
|
|
163
|
Zernicke R, MacKay C and Lorincz C:
Mechanisms of bone remodeling during weight-bearing exercise. Appl
Physiol Nutr Metab. 31:655–660. 2006. View Article : Google Scholar
|
|
164
|
Wang X, Tian Y, Liang X, Yin C, Huai Y,
Zhao Y, Huang Q, Chu X, Wang W and Qian A: Bergamottin promotes
osteoblast differentiation and bone formation via activating the
Wnt/β-catenin signaling pathway. Food Funct. 13:2913–2924. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Walker NA, Denegar CR and Preische J:
Low-intensity pulsed ultrasound and pulsed electromagnetic field in
the treatment of tibial fractures: A systematic review. J Athl
Train. 42:530–535. 2007.
|
|
166
|
Li K, Zhang C, Qiu L, Gao L and Zhang X:
Advances in application of mechanical stimuli in bioreactors for
cartilage tissue engineering. Tissue Eng Part B Rev. 23:399–411.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Carotenuto F, Politi S, Ul Haq A, De
Matteis F, Tamburri E, Terranova ML, Teodori L, Pasquo A and Di
Nardo P: From soft to hard biomimetic materials: Tuning
micro/nano-architecture of scaffolds for tissue regeneration.
Micromachines (Basel). 13:7802022. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Rajendran AK, Sankar D, Amirthalingam S,
Kim HD, Rangasamy J and Hwang NS: Trends in mechanobiology guided
tissue engineering and tools to study cell-substrate interactions:
A brief review. Biomater Res. 27:552023. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Han HM, Kim SY and Kim DH:
Mechanotransduction for therapeutic approaches: Cellular aging and
rejuvenation. APL Bioeng. 9:0215022025. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Akilbekova D and Turlybekuly A:
Patient-specific 3D bioprinting for in situ tissue engineering and
regenerative medicine. 3D Printing in Medicine. Elsevier; New York,
NY: pp. 149–178. 2023, View Article : Google Scholar
|
|
171
|
Behringer M, Heinrich C and Franz A:
Anabolic signals and muscle hypertrophy-significance for strength
training in sports medicine. Sports Orthop Traumatol. 41:9–18.
2025. View Article : Google Scholar
|
|
172
|
Raman R and Langer R: Biohybrid design
gets personal: New materials for patient-specific therapy. Adva
Mater. 32:e19019692020. View Article : Google Scholar
|
|
173
|
Soumya MAAK: AI-driven insights:
Revolutionizing health diagnostics and treatment. Budha
Publication; Tamil, Nadu: 2024
|
|
174
|
Gantenbein B, Frauchiger DA, May RD,
Bakirci E, Rohrer U and Grad S: Developing bioreactors to host
joint-derived tissues that require mechanical stimulation.
Encyclopedia of Tissue Engineering and Regenerative Medicine.
Elsevier; New York, NY: pp. 261–280. 2019
|