Diabetes mellitus is a group of metabolic disorders
characterized by hyperglycemia resulting from defects in insulin
secretion, insulin action, or both (1). Clinically, diabetes mellitus is
primarily categorized into Type 1 diabetes (T1DM, accounting for
5-10%) and Type 2 diabetes (T2DM, accounting for 90-95%). T1DM is
chiefly caused by absolute insulin deficiency resulting from
autoimmune-mediated destruction of pancreatic islet β-cells;
whereas T2DM is characterized by insulin resistance accompanied by
relative insufficiency in insulin secretion. Diabetes has become an
epidemic disease (2), with
China's prevalence projected to rise to 592 million by 2035
(3). Chronic hyperglycemia also
disrupts insulin secretion and/or function and is associated with
long-term damage and dysfunction in various tissues and organs
(eyes, kidneys, nerves, heart and blood vessels) as well as cancer
(4). The management of diabetes
and its complications remains a significant challenge, particularly
diabetic kidney disease (DKD). Existing biomarkers as therapeutic
targets only partially mitigate DKD progression and provide rough
predictions of disease advancement. When diabetes coexists with
cardiovascular disease (CVD), mortality nearly doubles (5). Even after restoring normal blood
glucose and metabolic balance, the effects of hyperglycemia on
diabetes and its complications do not immediately reverse; a
phenomenon termed 'metabolic memory' (6-8).
Metabolic memory primarily manifests as epigenetic alterations in
target cells under diabetic conditions (9). Since epigenetic modifications are
often reversible, this offers therapeutic opportunities to improve
cellular dysfunction and mitigate or 'erase' metabolic memory
(10).
Epigenetics refers to the study of heritable
phenotypic changes that do not involve alterations in DNA sequences
(11-13). Research in epigenetics has shown
a rapid growth trend (14). DNA
methylation, histone modifications and non-coding RNA regulation
are three major research directions in epigenetics (15). Epigenetic research not only
provides new perspectives for understanding gene expression
regulation but also offers novel approaches for disease prevention
and treatment (16). For
instance, by modulating epigenetic modifications, novel therapeutic
strategies can be developed to combat environmentally induced
diseases such as CVD, diabetes and cancer. Furthermore, epigenetic
markers serve as diagnostic biomarkers for diseases, underpinning
personalized treatment and precision medicine (17).
Emerging epigenetic tools can be used for the
prevention, diagnosis and treatment of diabetes and its
complications (18). Therefore,
investigating epigenetic variations under high-sugar environments
and their regulatory roles in diabetes progression holds
significant potential for clinical applications in diabetes
diagnosis and therapy (19). The
present review primarily introduced the epigenetic regulatory
mechanisms of high-sugar microenvironments across different cell
types and disease models, offering novel insights for treating
diabetes and its complications through epigenetic modulation. The
present review uniquely integrates findings across pancreatic,
hepatic, vascular and renal systems to provide a holistic view of
the epigenetic landscape reshaped by hyperglycemia.
Histones are proteins around which DNA is wound,
forming the fundamental units of chromatin (45,46). Modifications of histones include
methylation, acetylation, phosphorylation, ubiquitylation,
ADP-ribosylation, SUMOylation and other novel modifications, which
can directly influence chromatin structure (47). Different histone modification
states are closely associated with gene activity levels (48). For example, acetylation is
typically linked to transcriptional activation (49), while methylation can lead to
transcriptional repression (50).
In high-sugar environments, the most common histone
modification type includes histone acetylation. Histone acetylation
occurs when acetyltransferases (such as CBP and p300) add acetyl
groups to histone lysine residues. This modification leads to
chromatin relaxation, making DNA more accessible to transcription
factors and other regulatory proteins, thereby promoting gene
transcription (51). Biernacka
et al (52) discovered
that high-glucose-treated LNCaP cells (a human prostate cancer cell
line) exhibited markedly increased histone H3 acetylation
associated with the insulin-like growth factor binding protein 2
gene, leading to chemotherapy resistance in prostate cancer
(PCa). High glucose also induces histone H3 methylation (53). Histone methylation refers to the
addition of methyl groups to lysine or arginine residues on histone
proteins. Depending on methylation levels, histone methylation can
either promote or suppress gene transcription. For example,
tri-methylation of lysine 4 on histone H3 catalyzed by histone
methyltransferases (H3K4me3) is typically associated with gene
activation, whereas H3K27me3 is often linked to gene silencing
(54,55). Ishikawa et al (56) discovered that in type 2 diabetes
mellitus (T2DM), increased expression and decreased methylation of
the cyclin-dependent kinase inhibitor 1A and
phosphodiesterase 7B in T2DM exhibit increased expression
and reduced methylation. This combination impairs
glucose-stimulated insulin release by inhibiting β-cell
proliferation, promoting apoptosis and disrupting cAMP signaling
pathways (56). In a study of
Drosophila oocytes, Sun et al (57) found that under high-sugar
conditions, histone H3 methylation levels (such as, H3K9 and H3K27)
increase, suppressing cyclin D1 gene expression and
disrupting cell cycle progression, thereby impairing early
embryonic development.
Lactylation is a newly discovered post-translational
modification of proteins. It uses lactate, the end product of
glycolysis, as a substrate to covalently attach a lactyl group to
lysine residues on both histone and non-histone proteins (58). In high-glucose microenvironments,
cellular metabolic pathways undergo significant alterations that
directly influence lactylation levels, targets and functions,
thereby driving disease progression in conditions including cancer
and diabetic complications (59).
The high-glucose microenvironment drives cellular
metabolic reprogramming via the Warburg Effect, causing cells to
preferentially metabolize glucose through efficient glycolysis even
under oxygen-sufficient conditions. This process leads to a
substantial increase in intracellular lactate production. Research
confirms that high-glucose conditions specifically upregulate
L-lactic acid levels, while D-lactic acid remains unaffected. This
indicates that hyperglycemia primarily rewrites the cellular
modification profile through the L-lactic acid pathway (60).
In a high-sugar environment, lactylated histone
modifications do not simply increase overall but exhibit
site-specific enrichment and functional reprogramming. The
hyperglycemic microenvironment induces elevated levels at specific
histone lactylation sites, with H3K18la and H3K9la being the most
characteristic sites (61). For
instance, in diabetic retinopathy models, hyperglycemia/hypoxia
triggers explosive increases in H3K9la and H3K18la in retinal
endothelial cells, thereby activating transcription programs of
pro-angiogenic genes. Evidence also shows that, Alanyl-tRNA
synthetase 1 acts as a lactyltransferase to modulate H3K18, thereby
regulating elongase-5 transcription and mediating ferroptosis in
individuals with DKD (62).
Research indicates that elevated glucose levels
induce abnormal expression of certain histone modification enzymes
(63). Altered activity of these
enzymes directly modifies histone modification states, thereby
affecting gene transcriptional activity (64). For example, Sánchez-Ceinos et
al (65) found that the
histone lysine N-methyltransferase (enhancer of Zeste 2 polycomb
repressive complex 2 subunit) increases under high glucose
conditions in both human aortic endothelial cells (HAECs) exposed
to high glucose in vitro and those isolated from individuals
with diabetes (D-HAECs). This leads to elevated H3K27me3 levels and
the oxidative stress driven by H3K27me3 enhances nuclear factor κB
p65 (NF-κB p65) activity, ultimately causing endothelial
dysfunction (65). Furthermore,
Miao et al (66) found
that high-glucose-treated monocytes exhibited increased
transcriptional activity of histone acetyltransferases CBP
(CREB-binding protein) and p/CAF (p300/CBP-associated factor),
leading to elevated levels of HH3 (histone H3) acetylation at TNF-α
and COX-2 promoters in monocytes in human blood monocytes from type
1 and type 2 diabetic. This alteration promotes the expression of
metabolism- and inflammation-related genes such as NF-κB (66).
Changes in histone modifications induced by
high-sugar environments directly influence gene expression. For
example, Sun et al (67)
found P/CAF acetylated lysine residues 328 and lysine 450, in liver
cells from mice treated with high glucose, leading to proteasomal
degradation and increased blood glucose and hepatic glucose output.
Yamazaki et al (68) also
found that H3K9 acetylation (H3K9ac) is elevated in the promoter
region of the high glycated hemoglobin group in blood lymphocytes
and monocytes from type 1 diabetes mellitus patients, according to
the report on histone modification status. H3K9ac also influences
NF-κB expression, with studies indicating that increased H3K9ac
correlates with inflammation-induced progression of DKD (69). A high-sugar environment not only
affects individual gene expression but also, as research shows, a
high-sugar diet can alter the epigenetic state of pregnant women,
leading to genomic changes in their offspring (70). For example, leptin gene
(LEP) is a gene regulating energy balance. Allard et
al (71) demonstrated
through a Mendelian randomization study that maternal hyperglycemia
reduces LEP DNA methylation levels in offspring and
correlates with elevated umbilical cord blood leptin levels. This
suggests that in utero-induced epigenetic regulation may
contribute to increased obesity rates later in life (71-73). Table II describes the histone
modifications induced by high sugar in certain genes (65,74-78).
Non-coding RNAs refer to RNA molecules that do not
encode proteins, including long non-coding RNAs (lncRNAs) and
microRNAs (miRNAs) (79). They
play crucial roles in regulating gene expression. Non-coding RNAs
possess several important functions, including: i) Transcription
regulation: Non-coding RNAs influence gene transcription levels by
modulating the activity of transcription factors (80); ii) Splicing functions: Certain
lncRNAs can influence mRNA splicing processes, thereby altering the
final protein product type (81); iii) Stabilizing mRNA molecules:
Non-coding RNAs can bind to mRNA, affecting its stability and
degradation rate; iv) Regulating translation: By binding to
translation-related factors, ncRNAs can modulate mRNA translation
efficiency.
Research indicates that under high-glucose/diabetes
conditions, the expression of certain non-coding RNAs undergoes
significant alterations (82).
These changes may be mediated through the following mechanisms: i)
Modulating transcription factor activity: High glucose levels
induce alterations in the activity of specific transcription
factors, which in turn regulate the expression of particular
lncRNAs. For instance, Zhang et al (83) observed that in genetically
diabetic C57BKS mouse cells, signal transducer and activator of
transcription 1 phosphorylation markedly upregulates
metastasis-associated lung adenocarcinoma transcript 1 (MALAT1)
expression and downregulates miR-205 expression through the
JAK-STAT signaling pathway, disrupting epithelial-mesenchymal
transition (EMT) and impairing wound healing in patients with
diabetes. ii) Epigenetic modifications: A high-sugar environment
may also indirectly regulate non-coding RNA expression by affecting
DNA methylation and histone modifications. For example, in a study
on gestational diabetes mellitus (GDM), Li et al (84) found that the NADPH oxidase
5 gene exhibited higher methylation levels in the peripheral
blood of GDM pregnant women, indirectly leading to the lncRNA
RPL13P5 forming a co-expression network with the TSC
complex subunit 2 gene through the PI3K/AKT signaling pathway,
thereby inhibiting pancreatic islet β-cell growth and reducing
insulin secretion under both hypoglycemic and hyperglycemic
conditions.
In high-glucose environments, specific non-coding
RNAs have been identified as closely associated with insulin
signaling and glucose/lipid metabolism. For instance, maternally
expressed gene 3 (Meg3), an lncRNA located on human
chromosome 14q32, is upregulated under hyperglycemic conditions. It
induces ferroptosis by activating the p53 pathway (85). Abnormal Meg3 expression may be
closely linked to diabetes development. In diabetic mice, the
lncRNA H19 exhibits reduced expression levels in hepatocytes. It
enhances hepatic gluconeogenesis and glucose output by regulating
the MAPK, PI3K/Akt and mTOR signaling pathways (86). Wu et al (87) further discovered that in
high-glucose-induced mouse hepatocytes, downregulated miR-206
impairs lipogenesis and promotes insulin signaling by regulating
the PTPN1-INSR/IRS and PTPN1-PP2A-SP1-Srebp1c pathways. Table III describes the effects of
high glucose on the expression of selected ncRNAs (88-94).
A high-sugar environment influences gene expression
in different cell types through multiple epigenetic mechanisms,
specifically manifested in alterations such as DNA methylation,
histone modifications and non-coding RNAs. These changes may lead
to abnormal cellular functions, thereby affecting organismal
health.
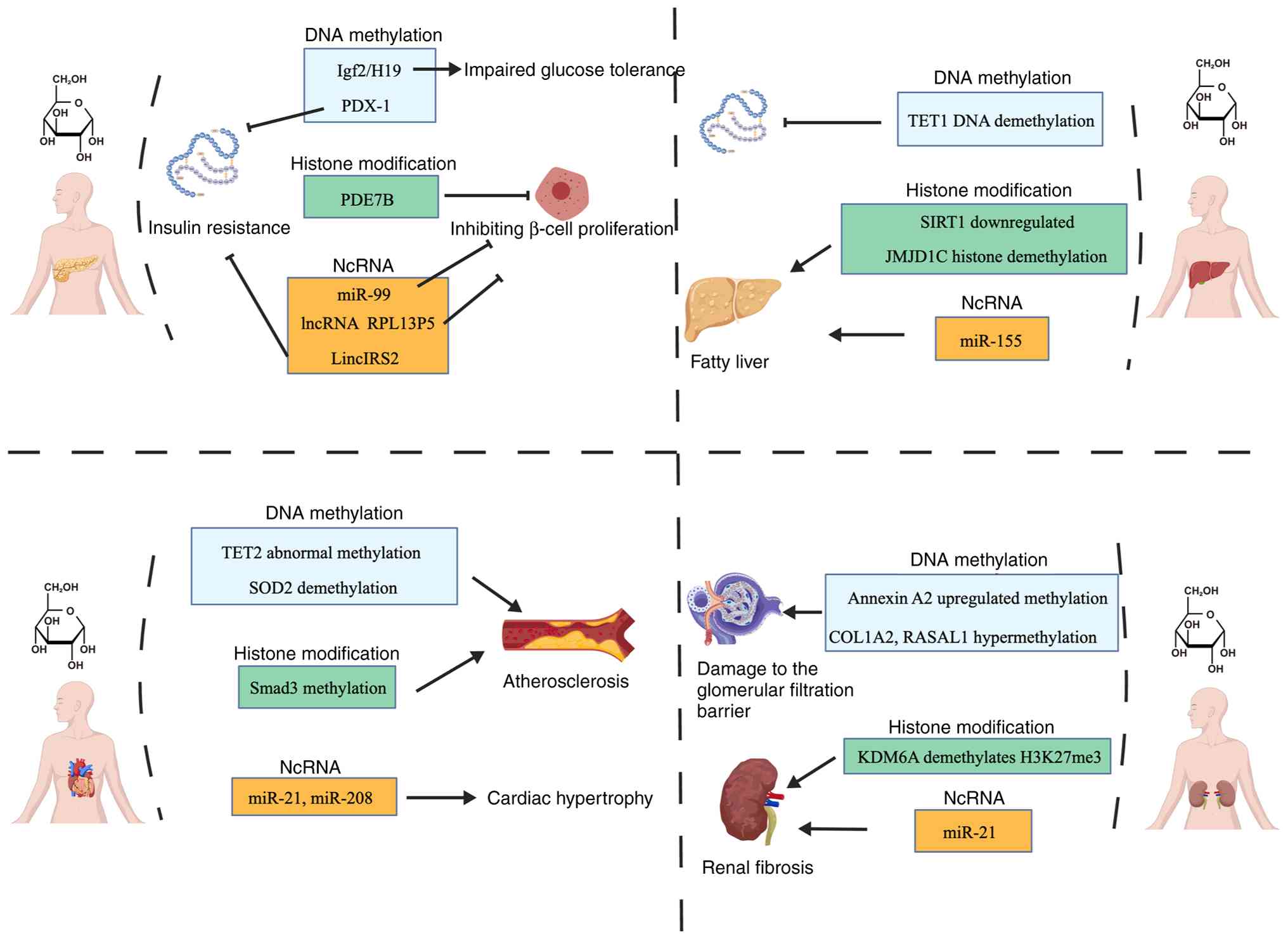
In pancreatic islet cells from patients with T2DM,
epigenetic variations cause β-cell dysfunction, manifesting as
reduced insulin secretion and insulin resistance (95). Research indicates the existence
of functionally distinct β-cell subtypes within islets, which may
possess different epigenetic backgrounds and regulatory
specificities, playing a crucial role in insulin secretion and
blood glucose regulation (96).
In pancreatic islet cells, high glucose regulates
epigenetic variation through the following mechanisms: i) Enhancing
transcriptional activity: For example, Bevacqua et al
(97) discovered in an adult
pancreatic islet study that exposure to high glucose concentrations
activates the histone methyltransferase SET domain containing 7,
histone lysine methyltransferase in pancreatic β cells. This enzyme
catalyzes the H3K4me1 in the PDX-1 gene promoter region. This
epigenetic modification leads to chromatin relaxation, facilitating
PDX-1 binding to the promoter and enhancing its transcriptional
activity. This binding further induced methylation modifications on
the PDX-1 protein itself (such as K91 site methylation), forming a
positive feedback loop that impaired glucose-stimulated insulin
secretion (GSIS) (97). ii)
Regulation of signaling pathways: Hyperglycemia can influence the
epigenetic characteristics of pancreatic islet cells through
multiple signaling pathways. For example, hyperglycemia regulates
mitochondrial autophagy via the PI3K/Akt/mTOR pathway, thereby
impairing pancreatic β-cell function. Under fluctuating
hyperglycemic conditions, elevated DNMT activity causes
hypomethylation in the miR-99 gene promoter region (CpG
island demethylation), releasing transcriptional repression of
miR-99. Elevated miR-99 suppresses the PI3K/Akt/mTOR pathway in
pancreatic β-cells, leading to increased mitochondrial autophagy
and consequently inhibiting β-cell proliferation and insulin
secretion (98).
A high-glucose environment may regulate hepatocyte
function by influencing epigenetic mechanisms. For example,
elevated glucose concentrations promote O-GlcNAc glycosylation
modification at serine and threonine residues of the
transcriptional regulatory protein tet-methylcytosine dioxygenase 1
(TET1), thereby activating TET1 protein function. TET1 catalyzes
the conversion of 5-methylcytosine to 5-hydroxymethylcytosine,
initiating DNA demethylation processes that promote inflammatory
responses and oxidative stress in hepatocytes (99,100). High glucose also affects
hepatocyte function by inducing epigenetic variations that alter
cellular signaling pathways. Wang et al (101) discovered that SIRT1
(NAD-dependent histone deacetylase) is downregulated in HG-treated
hepatocytes. This deficiency leads to suppressed mTORC2 activity
and Akt dephosphorylation, resulting in upregulation of
gluconeogenic genes (glucose-6-phosphatase, catalytic subunit,
phosphoenolpyruvate carboxykinase 1) and insulin resistance in
mice (102).
High concentrations of glucose can activate multiple
transcription factors in liver cells, which play crucial roles in
glucose and lipid metabolism. For example, Yun et al
(103) found that elevated
glucose levels activate NF-κB in human monocytic (THP-1) cells, a
key inflammatory transcription factor. Activation of NF-κB induces
the expression of miR-146 and miR-155. miR-146 contributes to
insulin resistance by regulating gene expression in the insulin
signaling pathway, while miR-155 promotes fatty liver development
by modulating suppressor of cytokine signaling 1 (SOCS1) expression
(103). Multiple transcription
factors also interact with each other under high glucose
conditions. The glucose-responsive transcription factor
carbohydrate response element binding protein (ChREBP) is a key
regulator of hepatic lipogenesis. Together with upstream
stimulating factor 1 (USF1), it activates the transcription of
lipogenic genes, thereby regulating lipid synthesis in the liver
(102). When glucose levels
rise, ChREBP is activated by acetylation at the K672 site by p300
(an acetyltransferase). It then synergizes with USF1 to influence
the conversion of fatty acids into triglycerides for storage via
the PI3K/AKT pathway, thereby enhancing lipid synthesis in
hepatocytes (103). This
interaction holds significant importance in both glucose and lipid
metabolism. Liver X receptor alpha (LXRα) in THP-1 is another key
transcription factor that positively regulates GLUT4 expression.
Under high-glucose conditions, LXRα expression is upregulated. It
interacts with JMJD1C (an H3K9 histone demethylase), leading to
H3K9 demethylation and subsequent promotion of GLUT4 expression,
thereby enhancing hepatic lipogenesis (103).
High glucose-induced DNA methylation in hepatocytes
is another common epigenetic phenomenon. Studies indicate that high
glucose affects the methylation status of certain genes in
hepatocytes, potentially leading to disorders in glucose and lipid
metabolism. For example, Krause et al (104) demonstrated that in T2DM, sterol
regulatory element-binding protein 1c (SREBP-1c, a key
transcription factor that binds to the SRE sterol regulatory
element within the promoters of lipogenic genes to enhance their
transcription) exhibits hypomethylation at CpG sites, impairing its
transcription. This enhances IRS-2 expression and induces excessive
lipidsynthesis via the PI3K/Akt pathway, closely associated with
hepatic steatosis (104,105).
Epigenetic alterations induced by high glucose
concentrations in human cells markedly affect metabolic functions
in muscle and fat cells. These epigenetic changes not only
influence gene expression but are also closely associated with the
development of T2DM and obesity.
In a high-glucose environment, the metabolic
function of muscle cells is markedly impaired. Research indicates
that high glucose levels induce insulin resistance, thereby
reducing glucose uptake in muscle and adipose tissue while
promoting hepatic glucose release. Friedrichsen et al
discovered that in the muscles of patients with T2DM, genes
associated with mitochondrial function, such as ubiquinone
oxidoreductase subunit B6 and cytochrome c oxidase subunit
7A1 exhibit elevated DNA methylation levels, impairing NADH
oxidation. This subsequently blocks the PI3K/Akt pathway, leading
to insulin resistance in muscle cells (106). The acetylation or deacetylation
status of histones in muscle cells is also affected by high
glucose. Research indicates that increased histone acetylation
correlates with enhanced muscle cell metabolic activity, whereas
high glucose environments activate deacetylases, thereby
suppressing muscle cell metabolism and reducing glucose utilization
efficiency (107). High glucose
also modulates muscle cell signaling pathways, inducing epigenetic
alterations. For instance, activation of the mTOR pathway typically
drives cell growth by promoting anabolic processes and inhibiting
catabolism. Saha et al (108) discovered that under high
glucose conditions, mTORC1 phosphorylates the T505 site of the
JMJD1C protein and inhibits LC3-II activity. LC3-II is a hallmark
protein for autophagosome formation; its suppressed activity
reduces cellular autophagy levels, leading to abnormal
proliferation of muscle cells. Since JMJD1C is established as a
histone demethylase, its phosphorylation specifically enhances
H3K9me2 demethylation in muscle cells, particularly in promoter
regions of muscle differentiation-related genes (such as
MyoD). This epigenetic modification promotes the
upregulation of myogenic transcription factors such as MyoD,
ultimately accelerating muscle cell differentiation and enhancing
glucose metabolism capacity (108).
Adipocytes play a crucial role in energy storage and
metabolic regulation. Under high glucose concentrations, the
epigenetic regulation of adipocytes also exhibits significant
alterations. In a high-glucose environment, adipocytes promote
lipogenesis by modifying epigenetic modifications. For instance,
Stegemann and Buchner (109)
discovered that methylation occurs in the promoter region of the
peroxisome proliferator-activated receptor gamma (PPARγ)
gene under high-sugar conditions, thereby promoting lipogenesis
(109-112). High glucose also disrupts
insulin signaling pathways in adipocytes, a disruption often
associated with epigenetic alterations. For example, miR-150
regulates lipogenesis in bovine preadipocytes via the mTOR
signaling pathway (111). A
high-glucose environment can also induce inflammatory responses
within adipocytes through epigenetic mechanisms. For instance, Zhao
et al (91) found that
Gm4419 (a long non-coding RNA) regulates mesangial cell
inflammatory factor expression via the NF-κB signaling pathway
under high-glucose conditions, thereby promoting chronic low-grade
inflammation and damaging adipose tissue, a process closely linked
to obesity and metabolic syndrome (113,114).
Hyperglycemia, a clinically prevalent metabolic
abnormality, can trigger multiple diseases including diabetes, DKD,
diabetic CVD and diabetic retinopathy. Under identical high-sugar
conditions, the cellular behavioral changes induced in different
diseases vary, exhibiting diverse molecular mechanisms and
epigenetic alterations.
The present review focused on the direct epigenetic
effects of hyperglycemia, it is crucial to acknowledge the inherent
complexity of diabetes as a metabolic disorder. Epigenetic
alterations observed in clinical and preclinical studies are likely
attributable to the synergistic effects of the diabetic
environment, encompassing hyperglycemia, insulin resistance,
dyslipidaemia and elevated levels of advanced glycation end
products. The epigenetic modifications observed in patients with
diabetes or animal models are therefore the result of a concerted
action of these multiple factors. Although in vitro studies
using high-glucose treatment can isolate the effects of
hyperglycemia, translating these findings to the in vivo
context requires caution. Future mechanistic studies should aim to
dissect the individual and synergistic contributions of each
metabolic abnormality to the overall epigenetic landscape.
Abnormal glucose metabolism is a hallmark feature of
diabetes patients. The effects of a high-sugar environment on the
epigenetics of diabetes patients include abnormal DNA methylation
(see High glucose and cellular DNA methylation), disruption
of histone modification homeostasis (see Histone modifications
in high-sugar environments) and disordered non-coding RNA
regulatory networks (see Effects of high-glucose environment on
non-coding RNA expression and consequences). The effect of
hyperglycemic symptoms on pancreatic function in patients with
diabetes (see Epigenetic regulation of pancreatic islet cells by
high glucose) primarily involves the regulation of
transcription factors and signaling pathways. These aspects will
not be elaborated upon further here. It is particularly worth
noting that epigenetic alterations are often closely associated
with nutritional metabolism (115-117). Jiang et al (118) first revealed that
hypermethylation of the CpG island in the promoter region of
hepatic glycogen synthase kinase β (GSK-β) suppresses
transcription of this gene. By activating the downstream PI3K-Akt
pathway, this leads to reduced hepatic glycogen synthesis capacity
and elevated fasting blood glucose levels. This epigenetic
regulatory mechanism has been demonstrated to be markedly
associated with the development of T2DM. In subsequent studies, the
team further discovered in a high-fat diet-induced obese mouse
model that liver GSK-β promoter methylation levels in the
experimental group were 2.3 times higher than in the control group,
accompanied by a 68% decrease in GSK-β mRNA expression. This
abnormal methylation positively correlated with the insulin
resistance index, suggesting that nutritional metabolic stress can
exacerbate diabetes progression through epigenetic reprogramming
(117,118).
In modern societies, dietary shifts have led to
markedly increased sugar intake. This high-sugar diet not only
affects metabolism but also induces CVD through epigenetic
alterations (119). Research
indicates that hyperglycemia is a major CVD risk factor (120-122). Hyperglycemia not only directly
causes vascular damage but also accelerates CVD progression through
multiple pathways. For instance, persistent hyperglycemia leads to
atherosclerosis, increased inflammatory responses and impaired
vascular endothelial function: All key pathological features of CVD
(123).
A high-sugar environment can induce a series of gene
expression changes through epigenetic regulation, thereby affecting
cardiovascular system function. Under hyperglycemic conditions, the
expression levels of genes encoding NF-κB subunits (such as
RELA and NFKB1) and the SOD2 gene increase in
cardiomyocytes. This may be associated with the demethylation of
specific CpG islands in the promoter regions of these genes. This
epigenetic regulation may lead to enhanced NF-κB-mediated
inflammatory responses and oxidative stress imbalance, subsequently
triggering cardiomyocyte dysfunction and promoting atherosclerosis
progression (122,123). Hao et al (124) observed increased H3K4me3 levels
in the Smad3 gene promoter region during their study of
cardiomyocytes from patients with diabetes. Smad3, a key regulator
of myocardial fibrosis, undergoes methylation that leads to either
overexpression or silencing of SOD2, inducing cardiovascular
dysfunction. Hyperglycemia also affects the expression of
non-coding RNAs (such as miRNAs), playing crucial roles in CVD
pathogenesis (125). Specific
miRNAs influence cardiomyocyte proliferation, apoptosis and
inflammatory responses by regulating target gene expression.
Studies indicate that miR-1 expression is suppressed in
cardiomyocytes under high-sugar conditions, while its appropriate
expression can prevent diabetic-induced cellular oxidative damage
(126). Furthermore, altered
expression of miR-21 and miR-208 is implicated in
high-sugar-induced cardiac hypertrophy and dysfunction (127). High-sugar-induced epigenetic
changes also elevate inflammatory cytokine expression in
endothelial cells, thereby promoting atherosclerosis formation.
Overexpression of inflammatory factors damages vascular
endothelium, further exacerbating CVD risk (128). A high-sugar environment also
induces epigenetic alterations in vascular smooth muscle cells
(VSMCs), such as abnormal methylation of the tet methylcytosine
dioxygenase 2 gene, which drives phenotypic shifts in VSMCs.
This promotes cell proliferation and migration, leading to vascular
narrowing and dysfunction (129).
DKD is one of the common complications in patients
with diabetes, primarily manifested as declining renal function
that may ultimately lead to renal failure (130). Increasing evidence indicates
that epigenetic regulation plays a crucial role in the onset and
progression of DKD (131). In
DKD, abnormal DNA methylation patterns have been found to be
closely associated with functional impairment of renal cells
(115,132). For instance, Chen et al
(133) discovered markedly
upregulated methylation of Annexin A2 (ANXA2) in patients
with DKD, leading to disruption of podocyte cytoskeletal
structures, increased renal cell apoptosis and detachment and
subsequent impairment of the glomerular filtration barrier,
resulting in reduced renal function (133). Studies indicate that in
diabetic kidney tissue, DNMTs mediate hypermethylation of COL1A2,
which activates TGF-β1, IL-6, TNF-α and IL-1β, exacerbating renal
inflammation and fibrosis. This disrupts extracellular matrix (ECM)
structure and releases damage-associated molecular patterns, such
as fragmented collagen peptides, thereby perpetuating the
inflammatory response (134,135). Methylation may also impair
transcription factor binding capacity. Certain transcription
factors require binding to unmethylated DNA to function and this
binding may be inhibited in DKD. As noted by Bechtel et al
(136), hypermethylation of
RASAL1 in patients with DKD promotes Ras gene activation in
fibroblasts, preventing its activation of GAP and consequently
accelerating renal fibrosis. Certain genes promoting cell
proliferation and fibrosis may exhibit hyperacetylation in diabetic
kidneys, thereby driving pathological processes (137-139). For instance, Lazar et al
(140) found that activation of
the histone acetyltransferase p300/CBP in diabetic mouse kidneys
enhanced ROS production and promoted renal inflammation. Briest
et al (141) also
revealed that under high-glucose conditions, KDM6A, acting as a
histone demethylase, reduces H3K27me3 levels and accelerates
glomerular inflammation progression.
Certain miRNAs and lncRNAs are up- or downregulated
in patients with DKD, affecting the expression of genes associated
with kidney injury (142). For
example, miR-21 has been demonstrated to be upregulated in DKD.
miR-21 suppresses PTEN expression by directly binding to its 3'-UTR
region. PTEN serves as a negative regulator of the PI3K/Akt/mTOR
signaling pathway; its downregulation leads to enhanced Akt
phosphorylation, activating the downstream mTOR pathway. The
activated Akt/mTOR pathway further stimulates TGF-β1 secretion,
inducing EMT in renal tubular epithelial cells and promoting ECM
deposition (such as type I collagen, fibronectin), thereby
facilitating tubular cell proliferation and fibrosis (143-145). As noted by Sur et al
(146), in high-glucose-treated
renal cells, hypermethylation of the miRNA-29b promoter region and
enhanced DNMT3B activity elevate promoter methylation levels of
anti-fibrotic genes (such as TIMP3, MMP9), silencing
their expression. This epigenetic dysregulation exacerbates
glomerular fibrosis in patients with DKD. lncRNAs also play a
crucial role in DKD progression. For instance, Dieter et al
(147) discovered that PVT1
expression increases in high-glucose-induced human glomerular
mesangial cells, accompanied by significant elevations in levels of
major ECM fibronectin and type IV collagen α1, as well as TGF-β1
and plasminogen activator inhibitor-1 (PAI-1), thereby inducing
renal inflammatory responses and glomerular mesangial cell
apoptosis.
The reversibility of epigenetic marks transforms
them from mere diagnostic indicators into promising therapeutic
targets in modern medicine. Their role is particularly significant
in various metabolic diseases such as diabetes, cancer and CVD
(149-153). Since epigenetic markers reflect
cellular states under specific environmental conditions, they are
considered potential disease diagnostic indicators (154,155). Epigenetic markers can be used
for early screening of various diseases, providing earlier warning
signals than traditional methods. For instance, methylation of
MALAT1 is commonly observed in patients with diabetes, enabling its
clinical application for early diagnosis (156). Epigenetic markers also
facilitate disease progression monitoring and treatment response
assessment. By periodically analyzing patients' epigenetic
profiles, clinicians can improve the evaluation of disease
progression and adjust treatment strategies as needed (157). Epigenetic characteristics
further support prognostic evaluation, as certain markers directly
correlate with disease outcomes. For example, in patients with
diabetes, elevated GLP-1R methylation risk correlates with
increased complication risk, providing a basis for personalized
treatment (158). Regarding
epigenetic regulation-based therapies for diabetes and related
diseases, Liu et al (134) detailed therapeutic approaches
for DKD. The present study primarily supplemented how epigenetics
can be used to treat diabetes and its associated CVD.
Epigenetic changes are reversible, thus offering new
possibilities for diabetes treatment. Researchers are exploring
strategies to reverse diabetes and its complications through
epigenetic therapies. This includes utilizing small-molecule drugs
or gene editing technologies to modulate epigenetic marks, thereby
restoring normal gene expression patterns (159,160). Such modulation can be achieved
through several key mechanisms, including: i) DNA methylation
regulation: Hyperglycemia can cause abnormal methylation in key
genes, such as those involved in insulin signaling pathways (such
as the PIK3R1 promoter region), affecting insulin secretion
and sensitivity (161). For
instance, inhibiting DNMTs can reverse abnormal methylation in
certain genes, restore β-cell function and improve insulin
resistance. 5-Aza-cytidine (5-AzaC), a chemical inhibitor,
suppresses DNMT activity (162). Filip et al (163) shows that treating diabetic
mouse models with 5-AzaC restores expression of key insulin
secretion genes (such as Ins1 and Ins2), thereby improving
β-cell function and lowering blood glucose levels. ii) Histone
modification intervention: Reduced histone acetylation correlates
with insulin resistance. Histone deacetylase inhibitors (HDACi),
such as sodium valproate, increase histone acetylation, promote
GLUT4 gene expression and improve glucose metabolism
(164). iii) Non-coding RNA
regulation: miRNAs (such as miR-29, miR-375) exhibit abnormal
expression in diabetes, targeting insulin secretion and
inflammatory responses. Modulating their levels via antisense
oligonucleotides or miRNA mimics improves pancreatic function and
reduces β-cell apoptosis. For example, miR-34a is a known miRNA
associated with β-cell apoptosis. Research from Wang et al
(165) indicates that using
miR-34a inhibitors (such as antisense oligonucleotides) effectively
reduces miR-34a expression. This suppression increases insulin
secretion in β-cells and reduces cell apoptosis, thereby improving
β-cell survival and function (165).
Epigenetic interventions offer a novel perspective
for CVD therapy and their potential is exemplified by the following
approaches: i) Regulation of vascular inflammation and metabolic
memory: Hyperglycemia-induced 'metabolic memory' leads to
persistent vascular inflammation through epigenetic mechanisms
(such as sustained DNA methylation or histone modifications).
Inhibiting DNMTs or HDACs can reduce inflammatory responses in
vascular endothelial cells and delay atherosclerosis progression.
ii) Myocardial fibrosis and cardiac remodeling: In diabetic
cardiomyopathy, abnormal methylation or histone modifications of
pro-fibrotic genes such as TGF-β drive myocardial fibrosis.
Epigenetic drugs (such as HDACi) suppress fibrosis-related gene
expression, improving cardiac function. Vorinostat, an FDA-approved
HDAC inhibitor, has been shown to suppress expression of type I and
III collagen genes (such as COL1A1 and COL3A1) and
improve cardiovascular function by modulating fibrosis-related
signaling pathways (such as the TGF-β pathway) (166). iii) Endothelial function
repair: non-coding RNAs (such as lncRNA MALAT1) play regulatory
roles in endothelial cell injury. Studies indicate that inhibiting
MALAT1 expression enhances survival capacity in human microvascular
endothelial cells and reduces apoptosis induced by hyperglycemia or
oxidative stress. Research indicates that MALAT1 regulates the
expression of miR-199a-5p target genes, phosphatase and
vesicle-associated genes (such as RAP1A and RAP1B), by
competing for binding. Targeting MALAT1 increases miR-199a-5p
expression, thereby promoting endothelial functional recovery and
reducing inflammation and thrombosis (167). Table IV illustrates the progress of
drugs targeting epigenetic regulation for treating diseases induced
by hyperglycemia (161,162,164-167).
The epigenetic regulatory mechanisms triggered by
high-sugar environments (including hyperglycemia, or high glucose
treatment) are increasingly becoming a research focus to elucidate
the pathogenesis of diabetes and its complications (such as
cardiovascular disease, nephropathy and neuropathy). In clinic,
hyperglycemia not only alters cellular metabolic states but also
induces persistent changes in cellular function by affecting
multiple epigenetic modifications, including DNA methylation,
histone modifications and non-coding RNA expression.
Hyperglycemia-induced epigenetic variations trigger functional
alterations across multiple organs and precipitate diverse
diseases. The present review highlighted only well-researched
cellular, organ and disease models; numerous other disease models,
such as diabetic retinopathy and diabetic foot, due to the scarcity
of research materials, remain incompletely elucidated.
Despite the abundance of existing research on
epigenetic alterations in high-sugar environments and diabetes
(including its complications), numerous uncharted territories
warrant further exploration. It is therefore recommend to
strengthen research in the following three areas: i) Mechanistic
studies: Deepening our understanding of the specific roles played
by different epigenetic mechanisms in diabetes pathogenesis. For
instance, analyzing the epigenetic characteristics of vascular
cells in patients with diabetes via high-throughput sequencing
techniques. ii) Intervention strategy research: Investigating how
various environmental factors (such as diet and exercise) influence
epigenetic alterations. iii) Epigenetics and metabolic disease
association studies: Further exploring the links between epigenetic
modifications and other metabolic disorders to uncover broader
biological mechanisms.
Regarding the application of epigenetics tools for
disease diagnosis and treatment, the following areas warrant
greater emphasis: i) Therapeutic strategies targeting epigenetic
modifications: Developing drugs and tools (such as
CRISPR/Cas9-based modification tools) that target specific
epigenetic markers to restore normal cellular function; ii)
Application of personalized medicine: Analyzing patients'
epigenetic profiles to formulate tailored treatment plans, which
holds promise for enhancing therapeutic outcomes; Changes in
epigenetic markers under hyperglycemic conditions carry significant
implications for disease diagnosis and monitoring. As research
progresses, these markers may emerge as a new generation of
diagnostic indicators, providing clinicians with more sensitive and
specific detection methods, ultimately improving patient treatment
outcomes and quality of life.
Not applicable.
DY and WW contributed to the conception and design
of the article. HZ drafted the manuscript. XL and YL revised the
manuscript. Data authentication is not applicable. All authors read
and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present study was supported by Jilin Provincial Health
Talent Program (grant no. JLSWSRCZX2025 to WW).
|
1
|
Harreiter J and Roden M: Diabetes
mellitus-Definition, classification, diagnosis, screening and
prevention (Update 2019). Wien Klin Wochenschr. 131(Suppl 1):
S6–S15. 2019.In German. View Article : Google Scholar
|
|
2
|
Menke A, Casagrande S, Geiss L and Cowie
CC: Prevalence of and trends in diabetes among adults in the United
States, 1988-2012. JAMA. 314:1021–1029. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Forouhi NG and Wareham NJ: Epidemiology of
diabetes. Medicine (Abingdon). 42:698–702. 2014.
|
|
4
|
Association AD: Diagnosis and
classification of diabetes mellitus. Diabetes Care. 36(Suppl 1):
S67–S74. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Schmidt AM: Diabetes Mellitus and
Cardiovascular Disease. Arteriosclerosis, Thrombosis, and Vascular
Biology. 2019. View Article : Google Scholar
|
|
6
|
Berezin A: Metabolic memory phenomenon in
diabetes mellitus: Achieving and perspectives. Diabetes Metab
Syndr. 10(Suppl 1): S176–S183. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vasishta S, Umakanth S, Adiga P and Joshi
MB: Extrinsic and intrinsic factors influencing metabolic memory in
type 2 diabetes. Vascul Pharmacol. 142:1069332022. View Article : Google Scholar
|
|
8
|
Kato M and Natarajan R: Epigenetics and
epigenomics in diabetic kidney disease and metabolic memory. Nat
Rev Nephrol. 15:327–345. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Intine RV and Sarras MP Jr: Metabolic
memory and chronic diabetes complications: Potential role for
epigenetic mechanisms. Curr Diab Rep. 12:551–559. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cavalli G and Heard E: Advances in
epigenetics link genetics to the environment and disease. Nature.
571:489–499. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Peixoto P, Cartron PF, Serandour AA and
Hervouet E: From 1957 to nowadays: A brief history of epigenetics.
Int J Mol Sci. 21:75712020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sapienza C and Issa JP: Diet, nutrition,
and cancer epigenetics. Annu Rev Nutr. 36:665–681. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gayon J: From Mendel to epigenetics:
History of genetics. C R Biol. 339:225–230. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Grunau C, Le Luyer J, Laporte M and Joly
D: The epigenetics dilemma. Genes (Basel). 11:232019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang L, Lu Q and Chang C: Epigenetics in
health and disease. Adv Exp Med Biol. 1253:3–55. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kaliman P: Epigenetics and meditation.
Curr Opin Psychol. 28:76–80. 2019. View Article : Google Scholar
|
|
17
|
Villanueva L, Álvarez-Errico D and
Esteller M: The contribution of epigenetics to cancer
immunotherapy. Trends Immunol. 41:676–691. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Recillas-Targa F: Cancer epigenetics: An
overview. Arch Med Res. 53:732–740. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dawson MA and Kouzarides T: Cancer
epigenetics: From mechanism to therapy. Cell. 150:12–27. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Huang W, Li H, Yu Q, Xiao W and Wang DO:
LncRNA-mediated DNA methylation: An emerging mechanism in cancer
and beyond. J Exp Clin Cancer Res. 41:1002022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bird A, Taggart M, Frommer M, Miller OJ
and Macleod D: A fraction of the mouse genome that is derived from
islands of nonmethylated, CpG-rich DNA. Cell. 40:91–99. 1985.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Meng H, Cao Y, Qin J, Song X, Zhang Q, Shi
Y and Cao L: DNA methylation, its mediators and genome integrity.
Int J Biolo Sci. 11:604–617. 2015. View Article : Google Scholar
|
|
23
|
Takai D and Jones PA: Comprehensive
analysis of CpG islands in human chromosomes 21 and 22. Proc Natl
Acad Sci USA. 99:3740–3745. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhao SG, Chen WS, Li H, Foye A, Zhang M,
Sjöström M, Aggarwal R, Playdle D, Liao A, Alumkal JJ, et al: DNA
methylation landscapes in advanced prostate cancer. Nat Genet.
52:778–789. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sina AAI, Carrascosa LG, Liang Z, Grewal
YS, Wardiana A, Shiddiky MJA, Gardiner RA, Samaratunga H, Gandhi
MK, Scott RJ, et al: Epigenetically reprogrammed methylation
landscape drives the DNA self-assembly and serves as a universal
cancer biomarker. Nat Commun. 9:49152018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Riggs AD: X chromosome inactivation,
differentiation, and DNA methylation revisited, with a tribute to
Susumu Ohno. Cytogenet Genome Res. 99:17–24. 2002. View Article : Google Scholar
|
|
27
|
Chen ACH, Huang W, Fong SW, Chan C, Lee
KC, Yeung WSB and Lee YL: Hyperglycemia altered DNA methylation
status and impaired pancreatic differentiation from embryonic stem
cells. Int J Mol Sci. 22:107292021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ginno PA, Gaidatzis D, Feldmann A, Hoerner
L, Imanci D, Burger L, Zilbermann F, Peters AHFM, Edenhofer F,
Smallwood SA, et al: A genome-scale map of DNA methylation turnover
identifies site-specific dependencies of DNMT and TET activity. Nat
Commun. 11:26802020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Charlton J, Jung EJ, Mattei AL, Bailly N,
Liao J, Martin EJ, Giesselmann P, Brändl B, Stamenova EK, Müller
FJ, et al: TETs compete with DNMT3 activity in pluripotent cells at
thousands of methylated somatic enhancers. Nat Genet. 52:819–827.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Law PP and Holland ML: DNA methylation at
the crossroads of gene and environment interactions. Essays
Biochem. 63:717–726. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Łoboś P and Regulska-Ilow B: Link between
methyl nutrients and the DNA methylation process in the course of
selected diseases in adults. Rocz Panstw Zakl Hig. 72:123–136.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Qu Y, Dang S and Hou P: Gene methylation
in gastric cancer. Clin Chim Acta. 424:53–65. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vigorelli V, Resta J, Bianchessi V, Lauri
A, Bassetti B, Agrifoglio M, Pesce M, Polvani G, Bonalumi G,
Cavallotti L, et al: Abnormal DNA methylation induced by
hyperglycemia reduces CXCR 4 gene expression in CD 34+ stem cells.
J Am Heart Assoc. 8:e0100122019. View Article : Google Scholar
|
|
34
|
de Mello VDF, Pulkkinen L, Lalli M,
Kolehmainen M, Pihlajamäki J and Uusitupa M: DNA methylation in
obesity and type 2 diabetes. Ann Med. 46:103–113. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gill AM, Leiter EH, Powell JG, Chapman HD
and Yen TT: Dexamethasone-induced hyperglycemia in obese Avy/a
(viable yellow) female mice entails preferential induction of a
hepatic estrogen sulfotransferase. Diabetes. 43:999–1004. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jain M, LoGerfo FW, Guthrie P and Pradhan
L: Effect of hyperglycemia and neuropeptides on interleukin-8
expression and angiogenesis in dermal microvascular endothelial
cells. J Vasc Surg. 53:1654–1660.e2. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hamad M, Mohammed AK, Hachim MY,
Mukhopadhy D, Khalique A, Laham A, Dhaiban S, Bajbouj K and Taneera
J: Heme Oxygenase-1 (HMOX-1) and inhibitor of differentiation
proteins (ID1, ID3) are key response mechanisms against
iron-overload in pancreatic β-cells. Mol Cell Endocrinol.
538:1114622021. View Article : Google Scholar
|
|
38
|
Chen YL, Rosa RH, Kuo L and Hein TW:
Hyperglycemia augments Endothelin-1-Induced constriction of human
retinal venules. Transl Vis Sci Technol. 9:12020. View Article : Google Scholar
|
|
39
|
Jurado-Aguilar J, Barroso E, Bernard M,
Zhang M, Peyman M, Rada P, Valverde ÁM, Wahli W, Palomer X and
Vázquez-Carrera M: GDF15 activates AMPK and inhibits
gluconeogenesis and fibrosis in the liver by attenuating the
TGF-β1/SMAD3 pathway. Metabolism. 152:1557722024. View Article : Google Scholar
|
|
40
|
Tewari S, Zhong Q, Santos JM and Kowluru
RA: Mitochondria DNA replication and DNA methylation in the
metabolic memory associated with continued progression of diabetic
retinopathy. Invest Ophthalmol Vis Sci. 53:4881–4888. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Park JH, Stoffers DA, Nicholls RD and
Simmons RA: Development of type 2 diabetes following intrauterine
growth retardation in rats is associated with progressive
epigenetic silencing of Pdx1. J Clin Invest. 118:2316–2324.
2008.PubMed/NCBI
|
|
42
|
Ding GL, Wang FF, Shu J, Tian S, Jiang Y,
Zhang D, Wang N, Luo Q, Zhang Y, Jin F, et al: Transgenerational
glucose intolerance with Igf2/H19 epigenetic alterations in mouse
islet induced by intrauterine hyperglycemia. Diabetes.
61:1133–1142. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhang T, Ouyang H, Mei X, Lu B, Yu Z, Chen
K, Wang Z and Ji L: Erianin alleviates diabetic retinopathy by
reducing retinal inflammation initiated by microglial cells via
inhibiting hyperglycemia-mediated ERK1/2-NF-κB signaling pathway.
FASEB J. 33:11776–11790. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Gunes A, Schmitt C, Bilodeau L, Huet C,
Belblidia A, Baldwin C, Giard JM, Biertho L, Lafortune A, Couture
CY, et al: IL-6 Trans-signaling is increased in diabetes, impacted
by glucolipotoxicity, and associated with liver stiffness and
fibrosis in fatty liver disease. Diabetes. 72:1820–1834. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Millán-Zambrano G, Burton A, Bannister AJ
and Schneider R: Histone post-translational modifications-cause and
consequence of genome function. Nat Rev Genet. 23:563–580. 2022.
View Article : Google Scholar
|
|
46
|
Zhou LQ and Dean J: Reprogramming the
genome to totipotency in mouse embryos. Trends Cell Biol. 25:82–91.
2015. View Article : Google Scholar
|
|
47
|
Lawrence M, Daujat S and Schneider R:
Lateral thinking: How histone modifications regulate gene
expression. Trends Genet. 32:42–56. 2016. View Article : Google Scholar
|
|
48
|
Zhang Y, Sun Z, Jia J, Du T, Zhang N, Tang
Y, Fang Y and Fang D: Overview of histone modification. Adv Exp Med
Biol. 1283:1–16. 2021. View Article : Google Scholar
|
|
49
|
Shen Y, Wei W and Zhou DX: Histone
acetylation enzymes coordinate metabolism and gene expression.
Trends Plant Sci. 20:614–621. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gong F and Miller KM: Histone methylation
and the DNA damage response. Mutat Res Rev Mutat Res. 780:37–47.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
He W, Li Q and Li X: Acetyl-CoA regulates
lipid metabolism and histone acetylation modification in cancer.
Biochim Biophys Acta Rev Cancer. 1878:1888372023. View Article : Google Scholar
|
|
52
|
Biernacka KM, Uzoh CC, Zeng L, Persad RA,
Bahl A, Gillatt D, Perks CM and Holly JM: Hyperglycaemia-induced
chemoresistance of prostate cancer cells due to IGFBP2. Endocr
Relat Cancer. 20:741–751. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Hong H, Li YM, Meng XM, Deng T and Zhu BM:
Histone methylation and diabetic cardiomyopathy. Sheng Li Xue Bao.
74:461–468. 2022.In Chinese. PubMed/NCBI
|
|
54
|
Brasacchio D, Okabe J, Tikellis C,
Balcerczyk A, George P, Baker EK, Calkin AC, Brownlee M, Cooper ME
and El-Osta A: Hyperglycemia induces a dynamic cooperativity of
histone methylase and demethylase enzymes associated with
gene-activating epigenetic marks that coexist on the lysine tail.
Diabetes. 58:1229–1236. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yuan Y, Zhu C, Wang Y, Sun J, Feng J, Ma
Z, Li P, Peng W, Yin C, Xu G, et al: α-Ketoglutaric acid
ameliorates hyperglycemia in diabetes by inhibiting hepatic
gluconeogenesis via serpina1e signaling. Sci Adv. 8:eabn28792022.
View Article : Google Scholar
|
|
56
|
Ishikawa K, Tsunekawa S, Ikeniwa M,
Izumoto T, Iida A, Ogata H, Uenishi E, Seino Y, Ozaki N, Sugimura
Y, et al: Long-term pancreatic beta cell exposure to high levels of
glucose but not palmitate induces DNA methylation within the
insulin gene promoter and represses transcriptional activity. PLoS
One. 10:e01153502015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Sun Y, Ma X and Su Y: Research progress on
methylation of histones H3K9 and H3K27 in early-stage embryos.
Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 37:1296–1300. 2020.In Chinese.
PubMed/NCBI
|
|
58
|
Zhang D, Tang Z, Huang H, Zhou G, Cui C,
Weng Y, Liu W, Kim S, Lee S, Perez-Neut M, et al: Metabolic
regulation of gene expression by histone lactylation. Nature.
574:575–580. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zheng B, Pan Y, Qian F, Liu D, Ye D, Yu B,
Zhong S, Zheng W, Wang X, Zhou B, et al: High sugar induced RCC2
lactylation drives breast cancer tumorigenicity through
upregulating MAD2L1. Adv Sci (Weinh). 12:e24155302025. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Ren H, Tang Y and Zhang D: The emerging
role of protein L-lactylation in metabolic regulation and cell
signaling. Nat Metab. 7:647–664. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Raychaudhuri D, Singh P, Chakraborty B,
Hennessey M, Tannir AJ, Byregowda S, Natarajan SM, Trujillo-Ocampo
A, Im JS and Goswami S: Histone lactylation drives CD8+ T cell
metabolism and function. Nat Immunol. 25:2140–2151. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Hong J, Xu H, Yu L, Yu Z, Chen X, Meng Z,
Zhu J, Li J and Zhu M: AARS1-mediated lactylation of H3K18 and
STAT1 promotes ferroptosis in diabetic nephropathy. Cell Death
Differ. Sep 23–2025.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Gao L, Yu W, Song P and Li Q: Non-histone
Methylation of SET7/9 and its Biological Functions. Recent Pat
Anticancer Drug Discov. 17:231–243. 2022. View Article : Google Scholar
|
|
64
|
Pradhan S, Chin HG, Estève PO and Jacobsen
SE: SET7/9 mediated methylation of non-histone proteins in
mammalian cells. Epigenetics. 4:383–387. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Sánchez-Ceinos J, Hussain S, Khan AW,
Zhang L, Almahmeed W, Pernow J and Cosentino F: Repressive H3K27me3
drives hyperglycemia-induced oxidative and inflammatory
transcriptional programs in human endothelium. Cardiovasc Diabetol.
23:1222024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Miao F, Gonzalo IG, Lanting L and
Natarajan R: In vivo chromatin remodeling events leading to
inflammatory gene transcription under diabetic conditions. J Biol
Chem. 279:18091–18097. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Sun C, Wang M, Liu X, Luo L, Li K, Zhang
S, Wang Y, Yang Y, Ding F and Gu X: PCAF improves glucose
homeostasis by suppressing the gluconeogenic activity of PGC-1α.
Cell Rep. 9:2250–2262. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yamazaki T, Mimura I, Tanaka T and Nangaku
M: Treatment of diabetic kidney disease: Current and future.
Diabetes Metab J. 45:11–26. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Singh R, Chandel S, Dey D, Ghosh A, Roy S,
Ravichandiran V and Ghosh D: Epigenetic modification and
therapeutic targets of diabetes mellitus. Biosci Rep.
40:BSR202021602020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Dalfrà MG, Burlina S, Del Vescovo GG and
Lapolla A: Genetics and epigenetics: New insight on gestational
diabetes mellitus. Front Endocrinol (Lausanne). 11:6024772020.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Allard C, Desgagné V, Patenaude J, Lacroix
M, Guillemette L, Battista MC, Doyon M, Ménard J, Ardilouze JL,
Perron P, et al: Mendelian randomization supports causality between
maternal hyperglycemia and epigenetic regulation of leptin gene in
newborns. Epigenetics. 10:342–351. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Ling C and Rönn T: Epigenetics in human
obesity and type 2 diabetes. Cell Metab. 29:1028–1044. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Chen B, Du YR, Zhu H, Sun ML, Wang C,
Cheng Y, Pang H, Ding G, Gao J, Tan Y, et al: Maternal inheritance
of glucose intolerance via oocyte TET3 insufficiency. Nature.
605:761–766. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Malmgren S, Spégel P, Danielsson AP,
Nagorny CL, Andersson LE, Nitert MD, Ridderstråle M, Mulder H and
Ling C: Coordinate changes in histone modifications, mRNA levels,
and metabolite profiles in clonal INS-1 832/13 β-cells accompany
functional adaptations to lipotoxicity. J Biol Chem.
288:11973–11987. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Liao W, Xu N, Zhang H, Liao W, Wang Y,
Wang S, Zhang S, Jiang Y, Xie W and Zhang Y: Persistent high
glucose induced EPB41L4A-AS1 inhibits glucose uptake via GCN5
mediating crotonylation and acetylation of histones and
non-histones. Clin Transl Med. 12:e6992022. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Lee HA, Kang SH, Kim M, Lee E, Cho HM,
Moon EK and Kim I: Histone deacetylase inhibition ameliorates
hypertension and hyperglycemia in a model of Cushing's syndrome. Am
J Physiol Endocrinol Metab. 314:E39–E52. 2018. View Article : Google Scholar
|
|
77
|
Horitani K, Iwasaki M, Kishimoto H, Wada
K, Nakano M, Park H, Adachi Y, Motooka D, Okuzaki D and Shiojima I:
Repetitive spikes of glucose and lipid induce senescence-like
phenotypes of bone marrow stem cells through H3K27me3
demethylase-mediated epigenetic regulation. Am J Physiol Heart Circ
Physiol. 321:H920–H932. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Qiu AW, Cao X, Zhang WW and Liu QH: IL-17A
is involved in diabetic inflammatory pathogenesis by its receptor
IL-17RA. Exp Biol Med (Maywood). 246:57–65. 2021. View Article : Google Scholar
|
|
79
|
Zogg H, Singh R and Ro S: Current Advances
in RNA Therapeutics for Human Diseases. Int J Mol Sci. 23:27362022.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Hombach S and Kretz M: Non-coding RNAs:
Classification, biology and functioning. Adv Exp Med Biol.
937:3–17. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Mattick JS and Makunin IV: Non-coding RNA.
Hum Mol Genet. 15:R17–R29. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Dong Y, Wan G, Peng G, Yan P, Qian C and
Li F: Long non-coding RNA XIST regulates hyperglycemia-associated
apoptosis and migration in human retinal pigment epithelial cells.
Biomed Pharmacother. 125:1099592020. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zhang L, Hung GCC, Meng S, Evans R and Xu
J: LncRNA MALAT1 Regulates Hyperglycemia Induced EMT in
Keratinocyte via miR-205. Noncoding RNA. 9:142023.PubMed/NCBI
|
|
84
|
Li J, Du B, Geng X and Zhou L: lncRNA
SNHG17 is downregulated in gestational diabetes mellitus (GDM) and
has predictive values. Diabetes Metab Syndr Obes. 14:831–838. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Chen C, Huang Y, Xia P, Zhang F, Li L,
Wang E, Guo Q and Ye Z: Long noncoding RNA Meg3 mediates
ferroptosis induced by oxygen and glucose deprivation combined with
hyperglycemia in rat brain microvascular endothelial cells, through
modulating the p53/GPX4 axis. Eur J Histochem. 65:32242021.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Goyal N, Tiwary S, Kesharwani D and Datta
M: Long non-coding RNA H19 inhibition promotes hyperglycemia in
mice by upregulating hepatic FoxO1 levels and promoting
gluconeogenesis. J Mol Med (Berl). 97:115–126. 2019. View Article : Google Scholar
|
|
87
|
Wu H, Zhang T, Pan F, Steer CJ, Li Z, Chen
X and Song G: MicroRNA-206 prevents hepatosteatosis and
hyperglycemia by facilitating insulin signaling and impairing
lipogenesis. J Hepatology. 66:816–824. 2017. View Article : Google Scholar
|
|
88
|
Pradas-Juni M, Hansmeier NR, Link JC,
Schmidt E, Larsen BD, Klemm P, Meola N, Topel H, Loureiro R,
Dhaouadi I, et al: A MAFG-lncRNA axis links systemic nutrient
abundance to hepatic glucose metabolism. Nat Commun. 11:6442020.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Cui X, Tan J, Shi Y, Sun C, Li Y, Ji C, Wu
J, Zhang Z, Chen S, Guo X and Liu C: The long non-coding RNA
Gm10768 activates hepatic gluconeogenesis by sequestering
microRNA-214 in mice. J Biol Chem. 293:4097–4109. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Li M, Niu M, Fan X, Chen F, Cao H, Liu Q,
Gan S, Yue P and Gao J: LncRNA MIR181A2HG inhibits keratinocytes
proliferation through miR-223-3p/SOX6 axis. Aging (Albany NY).
16:9846–9858. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Zhao G, Hailati J, Ma X, Bao Z, Bakeyi M
and Liu Z: LncRNA Gm4419 regulates myocardial Ischemia/Reperfusion
injury through targeting the miR-682/TRAF3 axis. J Cardiovasc
Pharmacol. 76:305–312. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Liu B, Gu Y, Luo JH, Fan YS and Feng ZY:
Effects of inhibiting the expression of lncRNA PVT1 on the
proliferation, apoptosis and oxidative stress of vascular
endothelial cells induced by hyperglycemia. Zhongguo Ying Yong
Sheng Li Xue Za Zhi. 37:561–565. 2021.In Chinese. PubMed/NCBI
|
|
93
|
He L, Zhu C, Jia J, Hao XY, Yu XY, Liu XY
and Shu MG: ADSC-Exos containing MALAT1 promotes wound healing by
targeting miR-124 through activating Wnt/β-catenin pathway. Biosci
Rep. 40:BSR201925492020. View Article : Google Scholar
|
|
94
|
Heydari N, Sharifi R and Nourbakhsh M,
Golpour P and Nourbakhsh M: Long non-coding RNAs TUG1 and MEG3 in
patients with type 2 diabetes and their association with
endoplasmic reticulum stress markers. J Endocrinol Invest.
46:1441–1448. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Golson ML and Kaestner KH: Epigenetics in
formation, function, and failure of the endocrine pancreas. Mol
Metab. 6:1066–1076. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Dror E, Fagnocchi L, Wegert V, Apostle S,
Grimaldi B, Gruber T, Panzeri I, Heyne S, Höffler KD, Kreiner V, et
al: Epigenetic dosage identifies two major and functionally
distinct β cell subtypes. Cell Metabolism. 35:821–836.7. 2023.
View Article : Google Scholar
|
|
97
|
Bevacqua RJ, Zhao W, Merheb E, Kim SH,
Marson A, Gloyn AL and Kim SK: Multiplexed CRISPR gene editing in
primary human islet cells with Cas9 ribonucleoprotein. bioRxiv. Sep
17–2023.PubMed/NCBI
|
|
98
|
Brown MR and Matveyenko AV: It's what and
when you eat: An overview of transcriptional and epigenetic
responses to dietary perturbations in pancreatic islets. Front
Endocrinol (Lausanne). 13:8426032022. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Klimontov VV, Saik OV and Korbut AI:
Glucose variability: How does it work? Int J Mol Sci. 22:77832021.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Ayodeji SA, Bao B, Teslow EA, Polin LA,
Dyson G, Bollig-Fischer A and Fehl C: Hyperglycemia and O-GlcNAc
transferase activity drive a cancer stem cell pathway in
triple-negative breast cancer. Cancer Cell Int. 23:1022023.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Wang RH, Kim HS, Xiao C, Xu X, Gavrilova O
and Deng CX: Hepatic Sirt1 deficiency in mice impairs mTorc2/Akt
signaling and results in hyperglycemia, oxidative damage, and
insulin resistance. J Clin Invest. 121:4477–4490. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Xue W, Huang J, Chen H, Zhang Y, Zhu X, Li
J, Zhang W, Yuan Y, Wang Y, Zheng L and Huang K: Histone
methyltransferase G9a modulates hepatic insulin signaling via
regulating HMGA1. Biochim Biophys Acta Mol Basis Dis. 1864:338–346.
2018. View Article : Google Scholar
|
|
103
|
Yun JM, Jialal I and Devaraj S: Epigenetic
regulation of high glucose-induced proinflammatory cytokine
production in monocytes by curcumin. J Nutr Biochem. 22:450–458.
2011. View Article : Google Scholar
|
|
104
|
Krause C, Geißler C, Tackenberg H, El
Gammal AT, Wolter S, Spranger J, Mann O, Lehnert H and Kirchner H:
Multi-layered epigenetic regulation of IRS2 expression in the liver
of obese individuals with type 2 diabetes. Diabetologia.
63:2182–2193. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Benichou E, Seffou B, Topçu S, Renoult O,
Lenoir V, Planchais J, Bonner C, Postic C, Prip-Buus C, Pecqueur C,
et al: The transcription factor ChREBP Orchestrates liver
carcinogenesis by coordinating the PI3K/AKT signaling and cancer
metabolism. Nat Commun. 15:18792024. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Friedrichsen M, Ribel-Madsen R, Mortensen
B, Hansen CN, Alibegovic AC, Højbjerre L, Sonne MP, Wojtaszewski
JF, Stallknecht B, Dela F and Vaag A: Muscle inflammatory signaling
in response to 9 days of physical inactivity in young men with low
compared with normal birth weight. Eur J Endocrinol. 167:829–838.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Yucel N, Wang YX, Mai T, Porpiglia E, Lund
PJ, Markov G, Garcia BA, Bendall SC, Angelo M and Blau HM: Glucose
metabolism drives histone acetylation landscape transitions that
dictate muscle stem cell function. Cell Rep. 27:3939–3955.e6. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Saha S, Fang X, Green CD and Das A: mTORC1
and SGLT2 Inhibitors-A therapeutic perspective for diabetic
cardiomyopathy. Int J Mol Sci. 24:150782023. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Stegemann R and Buchner DA:
Transgenerational inheritance of metabolic disease. Semin Cell Dev
Biol. 43:131–140. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Lee HH, An SM, Ye BJ, Lee JH, Yoo EJ,
Jeong GW, Kang HJ, Alfadda AA, Lim SW, Park J, et al: TonEBP/NFAT5
promotes obesity and insulin resistance by epigenetic suppression
of white adipose tissue beiging. Nat Commun. 10:35362019.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Hansen NS, Strasko KS, Hjort L, Kelstrup
L, Houshmand-Øregaard A, Schrölkamp M, Schultz HS, Scheele C,
Pedersen BK, Ling C, et al: Fetal hyperglycemia changes human
preadipocyte function in adult life. J Clin Endocrinol Metab.
102:1141–1150. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Houde AA, Hivert MF and Bouchard L: Fetal
epigenetic programming of adipokines. Adipocyte. 2:41–46. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Ishida T, Morisawa S, Iizuka M, Fujita H,
Jobu K, Morita Y and Miyamura M: Juzentaihoto extract suppresses
adipocyte hypertrophy and improves hyperglycemia in KKAy mice.
Pharmazie. 75:191–194. 2020.PubMed/NCBI
|
|
114
|
Villarroya F, Cereijo R, Gavaldà-Navarro
A, Villarroya J and Giralt M: Inflammation of brown/beige adipose
tissues in obesity and metabolic disease. J Intern Med.
284:492–504. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Yoshimoto N, Hayashi K, Hishikawa A,
Hashiguchi A, Nakamichi R, Sugita-Nishimura E, Yoshida-Hama E,
Azegami T, Nakayama T and Itoh H: Significance of podocyte DNA
damage and glomerular DNA methylation in CKD patients with
proteinuria. Hypertens Res. 46:1000–1008. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Su M, Yu T, Yu Y, Cheng Q, Zheng Y, Liao R
and Zeng Z: hsa-miR-607, lncRNA TUG1 and hsa_circ_0071106 can be
combined as biomarkers in type 2 diabetes mellitus. Exp Biol Med
(Maywood). 247:1609–1618. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Trang K and Grant SFA: Genetics and
epigenetics in the obesity phenotyping scenario. Rev Endocr Metab
Disord. 24:775–793. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Jiang Z, Zhao M, Voilquin L, Jung Y, Aikio
MA, Sahai T, Dou FY, Roche AM, Carcamo-Orive I, Knowles JW, et al:
Isthmin-1 is an adipokine that promotes glucose uptake and improves
glucose tolerance and hepatic steatosis. Cell Metab.
33:1836–1852.e11. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Kannel WB and McGee DL: Diabetes and
cardiovascular disease. The framingham study. JAMA. 241:2035–2038.
1979. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Lowe LP, Liu K, Greenland P, Metzger BE,
Dyer AR and Stamler J: Diabetes, asymptomatic hyperglycemia, and
22-year mortality in black and white men. The Chicago heart
association detection project in industry study. Diabetes Care.
20:163–669. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Gu K, Cowie CC and Harris MI: Mortality in
adults with and without diabetes in a national cohort of the U.S.
population, 1971-1993. Diabetes Care. 21:1138–1145. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Morgan CL, Currie CJ and Peters JR:
Relationship between diabetes and mortality: A population study
using record linkage. Diabetes Care. 23:1103–1107. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Wei M, Gaskill SP, Haffner SM and Stern
MP: Effects of diabetes and level of glycemia on all-cause and
cardiovascular mortality. The San Antonio heart study. Diabetes
Care. 21:1167–1172. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Hao J and Liu Y: Epigenetics of
methylation modifications in diabetic cardiomyopathy. Front
Endocrinol (Lausanne). 14:11197652023. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Prandi FR, Lecis D, Illuminato F, Milite
M, Celotto R, Lerakis S, Romeo F and Barillà F: Epigenetic
modifications and Non-coding RNA in Diabetes-mellitus-induced
coronary artery disease: Pathophysiological link and new
therapeutic frontiers. Int J Mol Sci. 23:45892022. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Zhou Y, Suo W, Zhang X, Yang Y, Zhao W, Li
H and Ni Q: Targeting epigenetics in diabetic cardiomyopathy:
Therapeutic potential of flavonoids. Biomed Pharmacother.
157:1140252023. View Article : Google Scholar
|
|
127
|
Temple NJ: Fat, sugar, whole grains and
heart disease: 50 years of confusion. Nutrients. 10:392018.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Silveira Rossi JL, Barbalho SM, Reverete
de Araujo R, Bechara MD, Sloan KP and Sloan LA: Metabolic syndrome
and cardiovascular diseases: Going beyond traditional risk factors.
Diabetes Metab Res Rev. 38:e35022022. View Article : Google Scholar
|
|
129
|
Henry RM, Kostense PJ, Dekker JM, Nijpels
G, Heine RJ, Kamp O, Bouter LM and Stehouwer CD: Carotid arterial
remodeling: A maladaptive phenomenon in type 2 diabetes but not in
impaired glucose metabolism: The Hoorn study. Stroke. 35:671–676.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Samsu N: Diabetic nephropathy: Challenges
in pathogenesis, diagnosis, and treatment. Biomed Res Int.
2021:14974492021. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Papadopoulou-Marketou N, Paschou SA,
Marketos N, Adamidi S, Adamidis S and Kanaka-Gantenbein C: Diabetic
nephropathy in type 1 diabetes. Minerva Med. 109:218–228. 2018.
View Article : Google Scholar
|
|
132
|
Wong YH, Wong SH, Wong XT, Yap QY, Yip KY,
Wong LZ, Chellappan DK, Bhattamisra SK and Candasamy M: Genetic
associated complications of type 2 diabetes mellitus. Panminerva
Med. 64:274–288. 2022. View Article : Google Scholar
|
|
133
|
Chen J, Liu Y, Xia S, Ye X and Chen L:
Annexin A2 (ANXA2) regulates the transcription and alternative
splicing of inflammatory genes in renal tubular epithelial cells.
BMC Genomics. 23:5442022. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Liu Z, Liu J, Wang W, An X, Luo L, Yu D
and Sun W: Epigenetic modification in diabetic kidney disease.
Front Endocrinol (Lausanne). 14:11339702023. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Kushwaha K, Garg SS and Gupta J: Targeting
epigenetic regulators for treating diabetic nephropathy. Biochimie.
202:146–158. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Bechtel W, McGoohan S, Zeisberg EM, Müller
GA, Kalbacher H, Salant DJ, Müller CA, Kalluri R and Zeisberg M:
Methylation determines fibroblast activation and fibrogenesis in
the kidney. Nat Med. 16:544–550. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Bates J, Vijayakumar A, Ghoshal S,
Marchand B, Yi S, Kornyeyev D, Zagorska A, Hollenback D, Walker K,
Liu K, et al: Acetyl-CoA carboxylase inhibition disrupts metabolic
reprogramming during hepatic stellate cell activation. J Hepatol.
73:896–905. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Lu J, Li XQ, Chen PP, Zhang JX, Liu L,
Wang GH, Liu XQ, Jiang TT, Wang MY, Liu WT, et al: Activation of
acetyl-CoA synthetase 2 mediates kidney injury in diabetic
nephropathy. JCI Insight. 8:e1658172023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Li X, Li C and Sun G: Histone acetylation
and its modifiers in the pathogenesis of diabetic nephropathy. J
Diabetes Res. 2016:40653822016. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Lazar AG, Vlad ML, Manea A, Simionescu M
and Manea SA: Activated histone acetyltransferase p300/CBP-Related
signalling pathways Mediate Up-Regulation of NADPH oxidase,
inflammation, and fibrosis in diabetic kidney. Antioxidants
(Basel). 10:13562021. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Briest F, Noerenberg D, Hennch C, Yoshida
K, Hablesreiter R, Nimo J, Sasca D, Kirchner M, Mansouri L, Inoue
Y, et al: Frequent ZNF217 mutations lead to transcriptional
deregulation of interferon signal transduction via altered
chromatin accessibility in B cell lymphoma. Leukemia. 37:2237–2249.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Feng X, Yang X, Zhong Y and Cheng X: The
role of ncRNAs-mediated pyroptosis in diabetes and its vascular
complications. Cell Biochem Funct. 42:e39682024. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Guo J: Effect of miR-21 on renal fibrosis
induced by Nano-SiO2 in diabetic nephropathy rats via
PTEN/AKT pathway. J Nanosci Nanotechnol. 21:1079–1084. 2021.
View Article : Google Scholar
|
|
144
|
Dhas Y, Arshad N, Biswas N, Jones LD and
Ashili S: MicroRNA 21 silencing in diabetic nephropathy: Insights
on therapeutic strategies. Biomedicines. 11:25832023. View Article : Google Scholar
|
|
145
|
Liu S, Wu W, Liao J, Tang F, Gao G, Peng
J, Fu X, Zhan Y, Chen Z, Xu W and Zhao S: MicroRNA 21: A critical
pathogenic factor of diabetic nephropathy. Front Endocrinol
(Lausanne). 13:8950102022. View Article : Google Scholar
|
|
146
|
Sur S, Steele R, Shi X and Ray RB:
miRNA-29b inhibits prostate tumor growth and induces apoptosis by
increasing bim expression. Cells. 8:14552019. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Dieter C, Lemos NE, Girardi E, Ramos DT,
Pellenz FM, Canani LH, Assmann TS and Crispim D: The rs3931283/PVT1
and rs7158663/MEG3 polymorphisms are associated with diabetic
kidney disease and markers of renal function in patients with type
2 diabetes mellitus. Mol Biol Rep. 50:2159–2169. 2023. View Article : Google Scholar
|
|
148
|
Jiang S, Li H, Zhang L, Mu W, Zhang Y,
Chen T, Wu J, Tang H, Zheng S, Liu Y, et al: Generic diagramming
platform (GDP): A comprehensive database of high-quality biomedical
graphics. Nucleic Acids Res. 53:D1670–D1676. 2025. View Article : Google Scholar :
|
|
149
|
Johnstone SE, Gladyshev VN, Aryee MJ and
Bernstein BE: Epigenetic clocks, aging, and cancer. Science.
378:1276–1277. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
López J, Añazco-Guenkova AM,
Monteagudo-García Ó and Blanco S: Epigenetic and epitranscriptomic
control in prostate cancer. Genes (Basel). 13:3782022. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Garcia-Martinez L, Zhang Y, Nakata Y, Chan
HL and Morey L: Epigenetic mechanisms in breast cancer therapy and
resistance. Nat Commun. 12:17862021. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Hanahan D: Hallmarks of cancer: New
dimensions. Cancer Discovery. 12:31–46. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Fan H, Yang F, Xiao Z, Luo H, Chen H, Chen
Z, Liu Q and Xiao Y: Lactylation: Novel epigenetic regulatory and
therapeutic opportunities. Am J Physiol Endocrinol Metab.
324:E330–E338. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Nagaraju GP, Dariya B, Kasa P, Peela S and
El-Rayes BF: Epigenetics in hepatocellular carcinoma. Semin Cancer
Biol. 86:622–632. 2022. View Article : Google Scholar
|
|
155
|
Panera N, Mandato C, Crudele A, Bertrando
S, Vajro P and Alisi A: Genetics, epigenetics and transgenerational
transmission of obesity in children. Front Endocrinol.
13:10060082022. View Article : Google Scholar
|
|
156
|
Galicia-Garcia U, Jebari S, Larrea-Sebal
A, Uribe KB, Siddiqi H, Ostolaza H, Benito-Vicente A and Martín C:
Statin Treatment-induced development of type 2 diabetes: From
clinical evidence to mechanistic insights. Int J Mol Sci.
21:47252020. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Grady WM, Yu M and Markowitz SD:
Epigenetic alterations in the gastrointestinal tract: Current and
emerging use for biomarkers of cancer. Gastroenterology.
160:690–709. 2021. View Article : Google Scholar
|
|
158
|
Li X, Lu L, Hou W, Huang T, Chen X, Qi J,
Zhao Y and Zhu M: Epigenetics in the pathogenesis of diabetic
nephropathy. Acta Biochim Biophys Sin (Shanghai). 54:163–172. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Khan RMM, Chua ZJY, Tan JC, Yang Y, Liao Z
and Zhao Y: From Pre-diabetes to diabetes: Diagnosis, treatments
and translational research. Medicina (Kaunas). 55:5462019.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Ling C, Bacos K and Rönn T: Epigenetics of
type 2 diabetes mellitus and weight change-a tool for precision
medicine? Nat Rev Endocrinol. 18:433–448. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Asano T, Fujishiro M, Kushiyama A, Nakatsu
Y, Yoneda M, Kamata H and Sakoda H: Role of phosphatidylinositol
3-kinase activation on insulin action and its alteration in
diabetic conditions. Biol Pharm Bull. 30:1610–1616. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Chen X, Pan X, Zhang W, Guo H, Cheng S, He
Q, Yang B and Ding L: Epigenetic strategies synergize with
PD-L1/PD-1 targeted cancer immunotherapies to enhance antitumor
responses. Acta Pharm Sin B. 10:723–733. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Filip K, Lewińska A, Adamczyk-Grochala J,
Marino Gammazza A, Cappello F, Lauricella M and Wnuk M:
5-azacytidine inhibits the activation of senescence program and
promotes cytotoxic autophagy during trdmt1-mediated oxidative
stress response in insulinoma β-TC-6 cells. Cells. 11:12132022.
View Article : Google Scholar
|
|
164
|
Thakur V, Gonzalez MA, Parada M, Martinez
RD and Chattopadhyay M: Role of histone deacetylase inhibitor in
diabetic painful neuropathy. Mol Neurobiol. 61:2283–2296. 2024.
View Article : Google Scholar
|
|
165
|
Wang Y, Zhou F, Li M, Zhang Y, Li N and
Shao L: MiR-34a-5p promotes hepatic gluconeogenesis by suppressing
SIRT1 expression. Exp Cell Res. 420:1133362022. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Özel M, Baskol M, Akalın H and Baskol G:
Suberoylanilide hydroxamic acid (SAHA) reduces fibrosis markers and
deactivates human stellate cells via the epithelial-mesenchymal
transition (EMT). Cell Biochem Biophys. 79:349–357. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Samidurai A, Olex AL, Ockaili R,
Kraskauskas D, Roh SK, Kukreja RC and Das A: Integrated asnalysis
of lncRNA-miRNA-mRNA regulatory network in rapamycin-induced
cardioprotection against ischemia/reperfusion injury in diabetic
Rabbits. Cells. 12:28202023. View Article : Google Scholar
|