Renal fibrosis is a key pathological process driving
the progression of chronic kidney disease (CKD) to end-stage renal
disease, ultimately resulting in renal failure (1,2).
The rate and severity of fibrotic progression critically influence
patient survival and quality of life (3-5).
However, no direct and effective treatment for renal fibrosis
currently exists aside from kidney transplantation and renal
dialysis (6-8). Therefore, elucidating the
underlying mechanisms of renal fibrosis may offer novel insights
for developing potential therapeutic strategies (9).
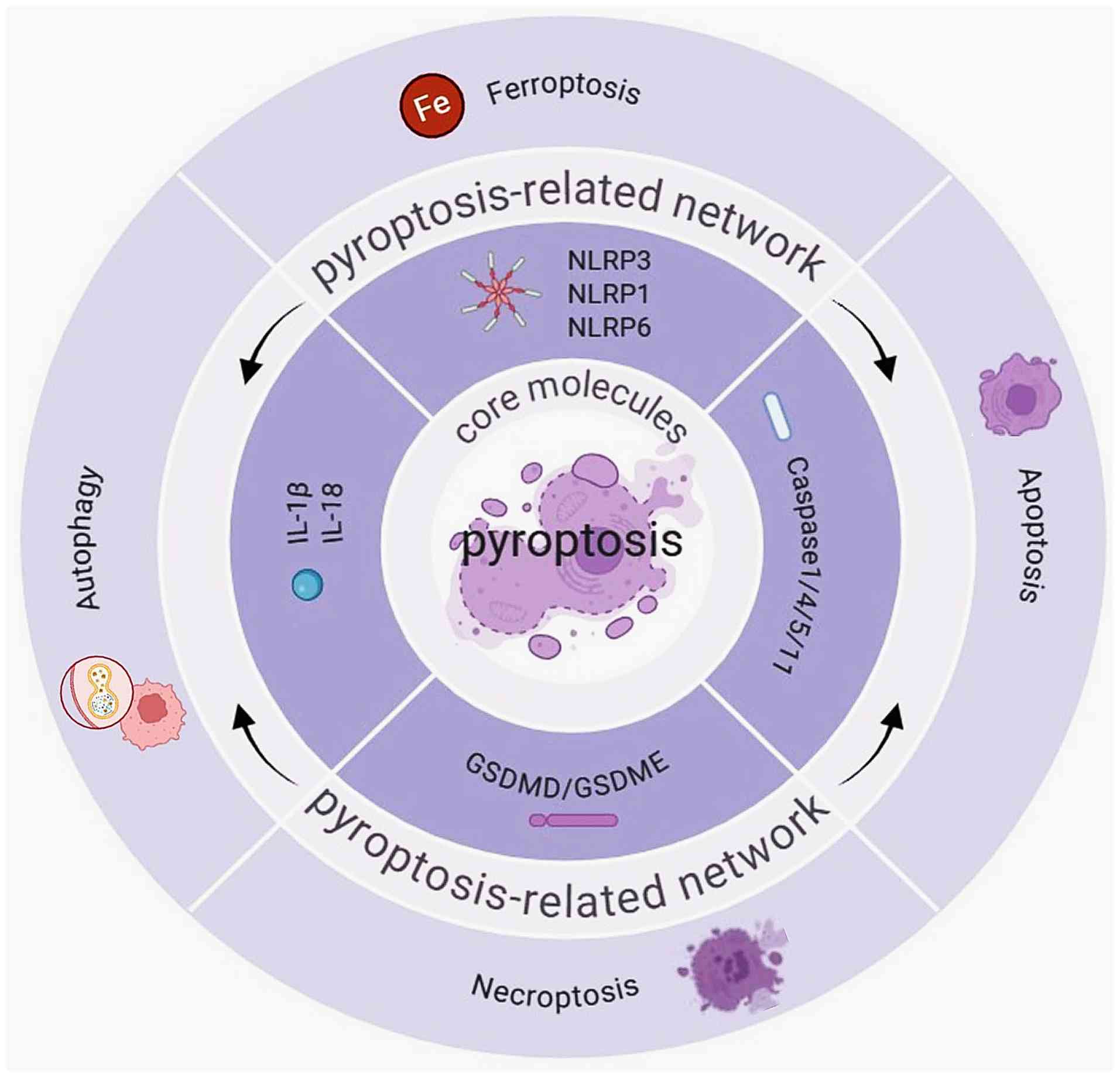
Pyroptosis is a highly inflammatory form of
programmed cell death characterized primarily by inflammasome
formation, activation of cysteine aspartic acid-specific proteases
(caspases), gasdermin (GSDM) dependence, and the release of
cellular contents and lactate dehydrogenase (10-13). Under physiological conditions,
pyroptosis functions as an innate immune response that eliminates
damaged cells and maintains in vivo homeostasis (14-16). Accumulating evidence indicates
that pyroptosis is closely associated with organ fibrosis,
including renal fibrosis, liver fibrosis and pulmonary fibrosis
(17,18). A substantial body of research has
demonstrated that sustained inflammation driven by pyroptosis is a
pivotal contributor to the development of renal fibrosis (19). Mediators released by pyroptotic
cells, such as interleukin (IL)-1β, IL-18 and damage-associated
molecular patterns (DAMPs), not only recruit and activate immune
cells such as M1 macrophages but also induce further pyroptosis in
neighboring cells, thereby establishing a self-amplifying
'inflammation-pyroptosis' vicious cycle (20). In this context, pyroptosis acts
not merely as an amplifier of inflammation but also as a critical
initiator of the chronic pro-fibrotic inflammatory microenvironment
(21-23). Notably, core effector molecules
involved in pyroptosis have been robustly validated as promoters of
renal fibrosis (24,25).
Inflammasomes serve as pyroptosis sentinels and are
large multiprotein complexes; among them, the nod-like receptor
protein 3 (NLRP3) inflammasome has garnered particular attention in
the context of renal fibrosis due to its central role (17,26). Activation of NLRP3 promotes the
release of pro-fibrotic factors (26,27). Gasdermin D (GSDMD) is the first
GSDM family member identified as essential for pyroptosis (25,28). Upon disruption of its
autoinhibitory conformation, GSDMD acquires pore-forming activity
on the cell membrane, leading to cell swelling and rupture
(29). As the principal
executioner of pyroptosis, GSDMD holds considerable research
potential in renal fibrosis (30). Genetic ablation of GSDMD has been
shown to ameliorate renal fibrosis (31). Moreover, clinical studies have
demonstrated that the severity of renal fibrosis in patients with
diabetic nephropathy (DN) is positively correlated with the
expression levels of both NLRP3 and GSDMD (32). Collectively, activation of these
key molecules and the interconnectivity of pyroptosis signaling
pathways coordinately regulate the progression of renal fibrosis
(33,34).
In the pathological process of pyroptosis and renal
fibrosis, crosstalk between pyroptosis and apoptosis, necroptosis,
ferroptosis and autophagy promotes renal fibrosis and accelerates
renal failure (35-38). For example, GSDMD can promote
ferroptosis by increasing cellular iron accumulation (39). Ferroptosis inhibitors, such as
Ferrostatin-1, indirectly attenuates the activity of the NLRP3
inflammasome (40). In addition,
PANoptosis, a cross-regulation of pyroptosis, apoptosis and
necroptosis (41,42), has been shown to be associated
with the development of renal fibrosis (43,44). Despite these insights, the
mechanisms underlying these interactions in renal fibrosis remain
unclear. The pathological effects of the interactive network formed
by pyroptosis and other forms of cell death undoubtedly exacerbate
renal fibrosis. Crucially, this regulatory machinery extends beyond
the kidney and is integrated into a complex network of inter-organ
crosstalk (45). The gut-kidney
axis acts as a pivotal remote switch, where gut-derived toxins or
beneficial metabolites calibrate the activation threshold of the
renal NLRP3 inflammasome (46,47). Therefore, elucidation of the
mechanisms of multi-target synergistic regulation is becoming
increasingly important in renal fibrosis (38,39) (Fig. 1).
At present, numerous studies have investigated the
relationship between pyroptosis and renal fibrosis (5), but the regulatory mechanisms by
which pyroptosis influences renal fibrosis remain under active
investigation. Emerging evidence suggests that inhibiting
pyroptosis may represent a potential therapeutic strategy to
alleviate renal fibrosis (11).
In the present review, the regulatory mechanisms of pyroptosis were
summarized and its role in renal fibrosis was discussed.
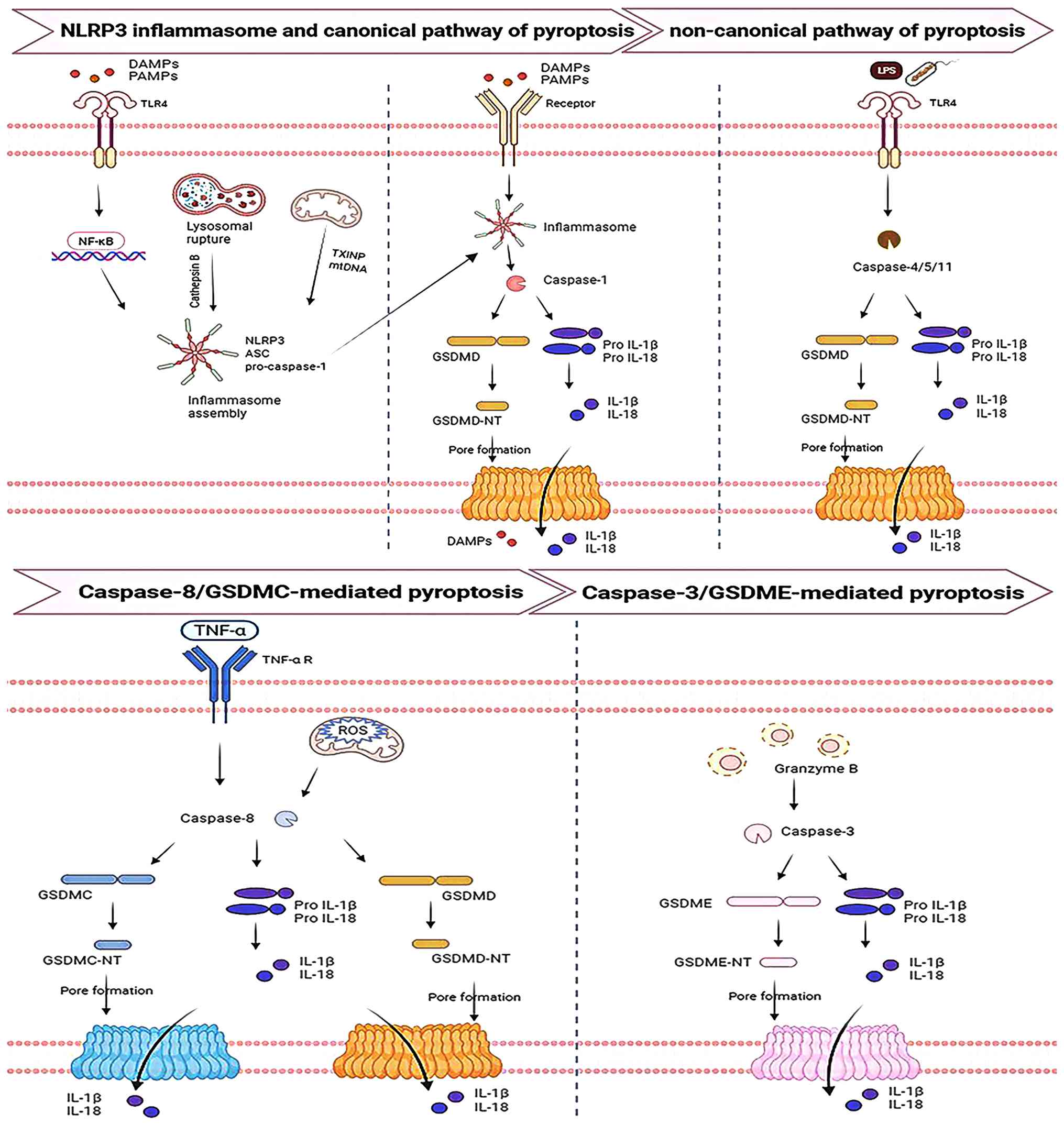
When cells or tissues are damaged, inflammasomes
assemble upon sensing DAMPs or pathogen-associated molecular
patterns (PAMPs) (48,49). The assembled inflammasomes
recruit and activate procaspase-1, enabling its autocatalytic
conversion into mature caspase-1 through autolysis. On the one
hand, activated caspase-1 cleaves GSDMD to generate its N-terminal
fragments (GSDMD-NT). Oligomerization of these fragments forms
10-14-nm-diameter plasma membrane pores, leading to rupture of
renal tubular epithelial cells and the release of abundant DAMPs
(26). On the other hand,
caspase-1 mediates the maturation and secretion of pro-IL-1β and
pro-IL-18 (50). Pyroptosis also
recruits and polarizes macrophages toward the M1 phenotype through
persistent pro-inflammatory signaling, amplifying inflammation and
promoting the release of pro-fibrotic factors such as transforming
growth factor-β (TGF-β) (20).
Upstream cellular events such as oxidative stress,
mitochondrial damage, or defective autophagy can also prime NLRP3
inflammasome assembly (56-58). Liao et al (34) demonstrated that inhibiting the
reactive oxygen species (ROS)/NLRP3 axis effectively alleviates
renal fibrosis in 5/6 nephrectomy (5/6 NX) rats. Under high-glucose
conditions, mitochondrial damage induces massive ROS production and
triggers cytosolic release of mitochondrial DNA (mtDNA), which
synergistically initiates NLRP3 assembly (59). Furthermore, impaired autophagic
flux and defective autophagolysosomal degradation result in CTSB
release, activating NLRP3 and pyroptosis, thereby facilitating
fibrosis development (57).
Although research in this field is extensive, current
investigations remain largely phenomenological, primarily
characterizing empirical associations between specific stimuli and
pyroptotic phenotypes. The substantial heterogeneity among these
upstream signals complicates identification of a convergent
molecular node, which is critical for developing precision
therapeutic targets capable of counteracting multiple pathogenic
triggers across various etiologies.
Notably, epigenetic regulation also plays a pivotal
role: N6-methyladenosine modification of NLRP3 enhances its
stability and induces pyroptosis and inflammation in the kidney
(60). Additionally, palmitate
reinforces NLRP3 activation by enhancing its S-palmitoylation in
synergy with lipopolysaccharide (LPS) stimulation (61). However, despite the substantial
potential of these emerging regulatory mechanisms, current research
remains at an early stage, with existing evidence primarily based
on molecular expression correlations. Moreover, the scarcity of
clinical-grade small-molecule inhibitors targeting these specific
epigenetic and post-translational modifications continues to limit
translational maturity and clinical applicability (Fig. 2).
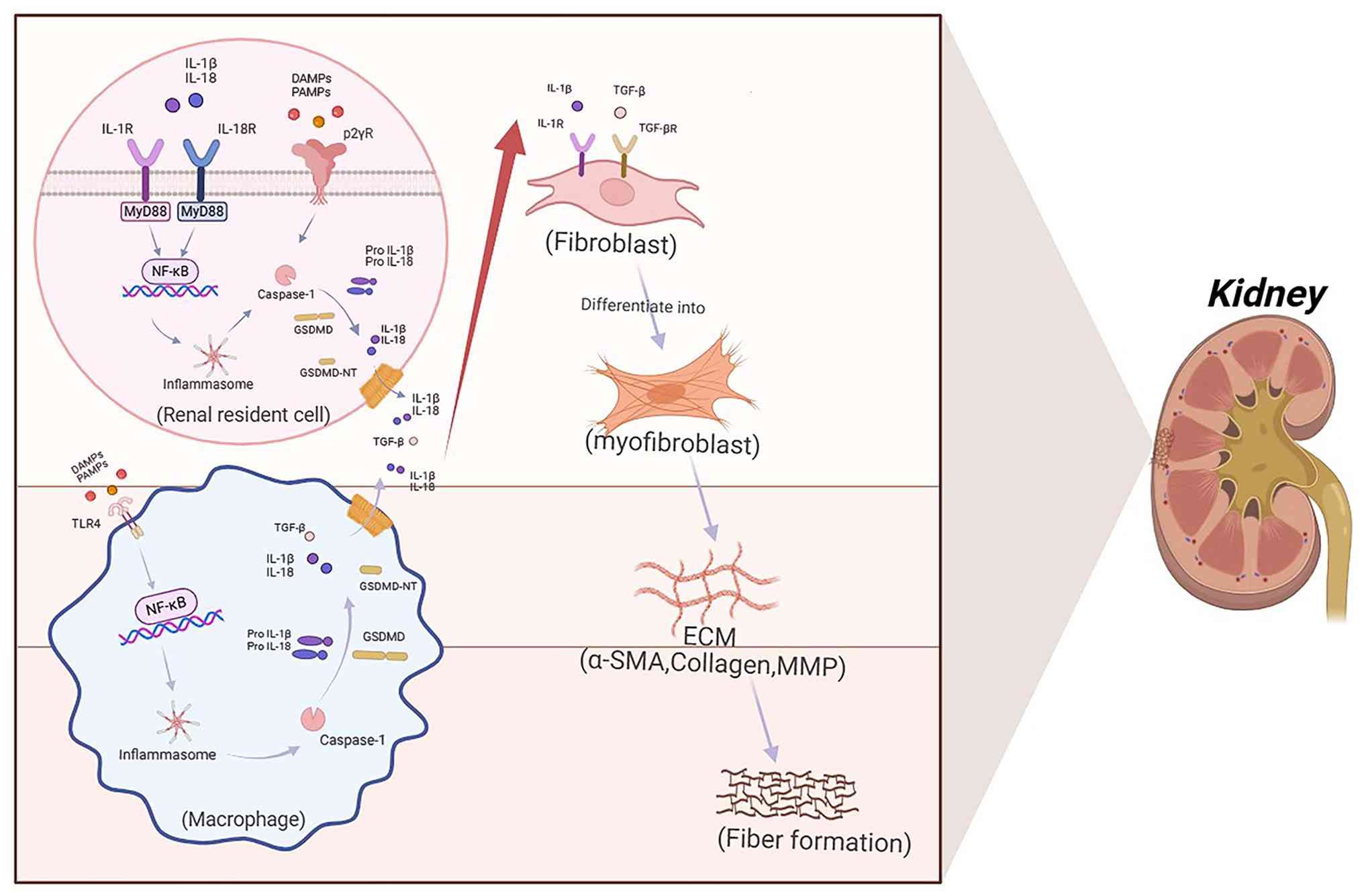
The release of IL-1β and IL-18 during pyroptosis
directly activates renal interstitial fibroblasts via the
IL-1R/MyD88 and IL-18R/NF-κB signaling pathways (62), promoting collagen deposition and
myofibroblast differentiation (63,64). Moreover, pyroptosis releases
DAMPs, including mtDNA and adenosine triphosphate (ATP), which
activate macrophages through TLR9/P2X purinoceptor 7 receptors and
fibroblasts via purinergic signaling, thereby enhancing the TGF-β
signaling pathway and driving extracellular matrix deposition
through upregulation of fibronectin (FN) and α-smooth muscle actin
(α-SMA) (65-67) (Fig. 3).
The non-canonical pathway involves the direct
activation of caspase-4/5 (human) or caspase-11 (mouse) by LPS from
Gram-negative bacteria, leading to GSDMD cleavage (26) and initiating a downstream cascade
similar to that of the canonical pathway (28,68). GSDMD-mediated pyroptosis,
executed through caspase-dependent cleavage, promotes inflammation
and fibrosis (69,70). Surprisingly, research has shown
that the conserved cysteine residue at position 191 (Cys191) in
human GSDMD undergoes S-palmitoylation, which promotes
GSDMD-mediated pyroptosis and cytokine release (71). Additionally, Cys191 mutations or
treatment with palmitoyl-transferase inhibitors suppress GSDMD
palmitoylation, its membrane localization and subsequent pyroptosis
or IL-1β secretion (72).
However, from a critical perspective, the role of
non-canonical pathways in renal fibrosis may be underestimated.
Direct activation of caspase-11 by intracellular LPS triggers
potassium (K+) efflux, which facilitates secondary NLRP3
inflammasome assembly and amplifies the pyroptotic signaling
cascade. Crucially, this intracellular event is intricately linked
to systemic regulation via the gut-kidney axis. Intestinal
dysbiosis in the context of CKD compromises gut barrier integrity,
leading to systemic accumulation and translocation of gut-derived
LPS and uremic toxins into renal tissues. These remote pathogenic
signals serve as pivotal triggers that initiate and sustain chronic
renal inflammation and the pyroptotic cascade, representing a
critical systemic mechanism driving the progression of renal
fibrosis (45,73) (Fig. 4).
Chemotherapeutic agents such as cisplatin or
granzyme B can induce caspase-3 activation, which cleaves GSDME to
release the GSDME-NT fragment. GSDME-NT oligomerizes to form pores
in the plasma membrane, thereby inducing pyroptosis. Li et
al (74) found that
caspase-3/GSDME-mediated pyroptosis of renal parenchymal cells
contributes to fibrosis development following ureteral obstruction.
Further investigations by Wu et al (75) team revealed that GSDME deficiency
significantly attenuates renal fibrosis and dysfunction in both
unilateral ureteral obstruction (UUO) and 5/6 NX models.
Conversely, overexpression of GSDME-NT exacerbates fibrotic
responses in UUO kidneys and TGF-β1-treated renal tubular
epithelial cells (75). Notably,
GSDME and GSDMD exhibit synergistic regulatory roles in the
progression of pyroptosis and fibrosis (76). A previous study indicated that
combined deletion of these two genes confers greater kidney
protection than deletion of either gene alone (77).
The metabolite α-ketoglutarate is upregulated by
ROS, oxidizing death receptor 6 (DR6) and prompting its endocytosis
and formation of the DR6 receptosome. Within this complex,
caspase-8 undergoes autocleavage and activation, subsequently
cleaving GSDMC to release GSDMC-NT, which forms membrane pores and
induces pyroptosis (78).
Moreover, under specific conditions such as activation of tumor
necrosis factor-α (TNF-α) signaling, caspase-8 can cleave GSDMD to
generate GSDMD-NT, leading to pyroptosis (29).
The central pathogenic hub through which pyroptosis
drives renal fibrosis lies in GSDMD-mediated lytic rupture of the
cell membrane. This pivotal execution step directly causes
explosive release of pro-inflammatory factors and DAMPs, thereby
initiating and perpetuating a pro-fibrotic tissue microenvironment.
While single-target inhibition of the upstream sensor NLRP3 or the
executioner GSDMD has shown therapeutic potential in preclinical
models, inhibitors targeting GSDME, which may act as a compensatory
executor, also present significant therapeutic prospects.
Overall, the core pathogenic hub of
pyroptosis-driven renal fibrosis centers on GSDMD-mediated
cytolytic rupture. This critical execution step directly triggers
explosive release of pro-inflammatory cytokines and DAMPs, thereby
initiating and sustaining a self-perpetuating pro-fibrotic tissue
microenvironment (11). While
single-target inhibition of the upstream sensor NLRP3 or the
executioner protein GSDMD has demonstrated significant therapeutic
potential in preclinical models, the existence of potential
compensatory mechanisms involving other GSDMs, such as GSDME,
suggests that inhibitors targeting these alternative executioners
also hold substantial therapeutic promise.
Recent evidence indicates that pyroptosis does not
occur in isolation but rather engages in sophisticated crosstalk
with other programmed cell death modalities, such as apoptosis and
necroptosis. This complex regulatory network amplifies initial
damage signals and dictates cellular fate, ultimately driving the
progression of renal fibrosis (Fig.
4).
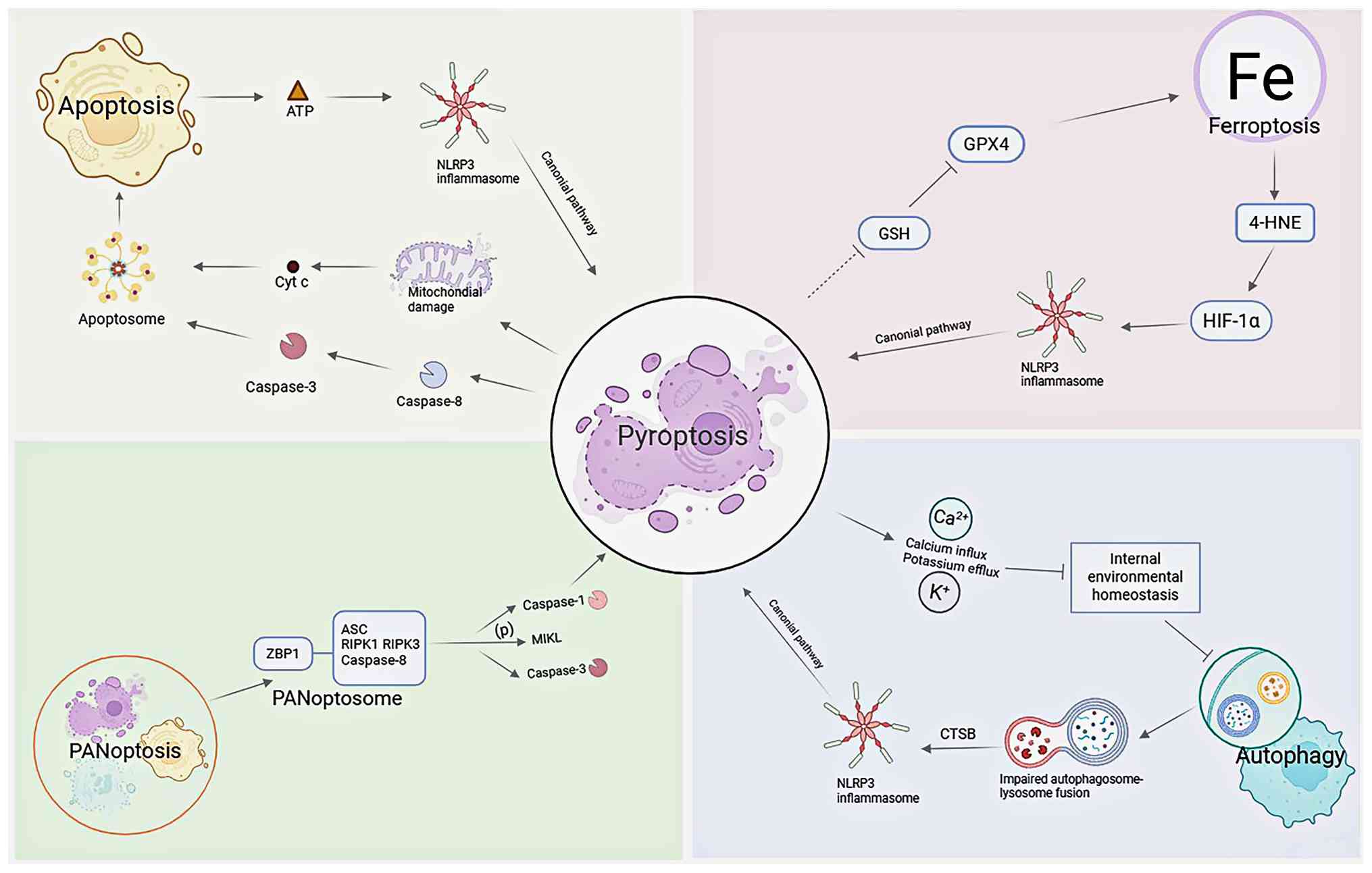
Pyroptosis and apoptosis interact synergistically
through the dual cleavage function of caspase-8 (79). Triggered by TNF-α signaling,
caspase-8 functions as a molecular switch that initiates classical
apoptosis via the caspase-3 cascade while simultaneously cleaving
GSDMD or GSDME to execute inflammatory pyroptosis (29). This crosstalk is amplified under
hypoxic conditions, where the GSDMC-mediated pyroptosis pathway
accelerates tubular epithelial injury (80). Moreover, these pathways establish
a self-perpetuating vicious cycle: Pyroptosis-induced mitochondrial
dysfunction facilitates cytochrome c release and
caspase-9/3-dependent apoptosis, whereas early apoptotic cells
release ATP to feedback-activate the NLRP3 inflammasome, thereby
intensifying the fibrotic response (79,81).
PANoptosis is an integrated programmed cell death
pathway that coordinates signaling from pyroptosis, apoptosis and
necroptosis through assembly of a multi-protein scaffold called the
PANoptosome. Comprising key sensors and adaptors, including Z-DNA
binding protein 1 (ZBP1), ASC, caspase-8 and receptor-interacting
serine/threonine-protein kinase 1/3, this complex is primarily
triggered by TNF-α to drive inflammatory tissue damage (43,82). Furthermore, RIPK3 facilitates
renal fibrosis progression in UUO models by phosphorylating NLRP3,
thereby enhancing inflammasome activity and inflammatory signaling
(83,84). Single-cell RNA sequencing
analysis by Zhuang et al (83) and Chen et al (85) confirmed that PANoptosis in
proximal tubular epithelial cells significantly exacerbates renal
injury. Consequently, targeting PANoptosis, particularly by
inhibiting its activation in macrophages, represents a promising
therapeutic strategy to mitigate systemic inflammation and chronic
kidney damage (86,87).
Pyroptosis and ferroptosis synergistically drive
kidney disease progression through a complex molecular interplay
(39,40). Pyroptotic stress facilitates
ferroptosis by perturbing intracellular redox homeostasis and
impairing the glutathione/glutathione peroxidase 4 axis (39,88). Conversely, lipid peroxidation
end-products such as 4-hydroxynonenal accumulated during
ferroptosis serve as endogenous danger signals that directly
aggravate oxidative stress and potentiate, either directly or
indirectly, activation of the NLRP3 inflammasome, thereby further
amplifying the inflammatory response (89). Ultimately, this reciprocal
coupling establishes a self-amplifying loop of oxidative stress and
inflammation, positioning this crosstalk as a central driver of
renal fibrosis (90).
The crosstalk between autophagy and pyroptosis is
complex, exhibiting bidirectional regulation in the pathological
process. Selective mitophagy provides a protective mechanism by
clearing damaged mitochondria and ROS, thereby suppressing NLRP3
inflammasome activation and negatively regulating pyroptosis
(91). Conversely, pathological
stress can induce lysosomal membrane permeabilization, leading to
CTSB leakage into the cytoplasm and exacerbating pyroptotic cell
death (92). Under persistent
stimuli such as uric acid, the autophagic response may shift from
an early adaptive defense to a late-stage dysregulated state
(93). Consequently, resolving
autophagic dysfunction and suppressing pyroptosis-driven
inflammation are essential therapeutic strategies for mitigating
renal fibrosis.
In conclusion, given the inherent complexity of
pyroptosis and its interactive network, as well as the substantial
functional redundancy among distinct programmed cell death
modalities, targeting a single pathway may yield limited
therapeutic efficacy due to compensatory cellular escape
mechanisms. Consequently, multi-target synergistic intervention has
emerged as a transformative therapeutic paradigm. Specifically,
strategies targeting convergent terminal executioners (for example,
dual GSDMD/E blockade) or upstream integrative hubs (for example,
PANoptosome-targeted intervention) represent promising preclinical
paradigms with high translational potential. Although challenges
such as cell-type specificity, safety profiling and
kidney-selective delivery remain to be addressed, these approaches
constitute emerging therapeutic frameworks for interrupting the
maladaptive acute-to-chronic kidney disease (AKI-to-CKD) transition
and effectively halting the progression of renal fibrosis.
In exploring the pathological role of pyroptosis in
driving renal fibrosis, the strategic selection of animal models is
fundamental for assessing mechanistic reliability and clinical
translational potential.
While LPS-induced models serve as the 'gold
standard' for validating canonical and non-canonical pyroptotic
pathways via rapid GSDMD cleavage, they exhibit inherent
limitations in mimicking chronic fibrotic evolution, as they
primarily simulate systemic sepsis or acute inflammatory responses
rather than primary renal pathology, often resulting in transient
functional shifts that fail to capture the sustained, progressive
nature of clinical CKD. By contrast, the ischemia-reperfusion
injury (IRI) model is more suited for capturing the pyroptotic
cascade during the AKI-to-CKD transition, though its utility is
frequently compromised by high technical sensitivity to factors
such as core temperature and a narrow injury threshold that risks
either rapid tissue repair or premature animal mortality. For
mechanistic studies focused on interstitial fibrosis, UUO remains
the core methodology for investigating the pressure-triggered
NLRP3/Caspase-1 axis due to its superior reproducibility, albeit
its purely mechanical etiology lacks clinical systemic
comorbidities, and its compressed, irreversible pathological course
limits its effectiveness in evaluating therapies aimed at
regression of fibrosis. By comparison, the adenine-induced model
provides an ideal setting for studying secondary pyroptosis
triggered by crystalline-induced metabolic disturbances.
Furthermore, the cisplatin model offers unique advantages in
elucidating Caspase-3/GSDME-mediated pyroptosis, facilitating a
deeper understanding of the cross-talk between apoptosis and
pyroptosis, notwithstanding that traditional high-dose regimens
typically induce severe acute necrosis and high lethality, thereby
diverging from clinical repeated low-dose protocols and obscuring
the window for longitudinal fibrotic assessment (94,95).
Despite these significant contributions, a gap
persists between these experimental models and the complex
pathophysiology of human CKD. Although5/6 NX and DN models offer
higher translational value by simulating hemodynamic fluctuations
and metabolic aberrations, their extended induction periods and
complex background signals often complicate the precise
quantification of specific contribution of pyroptosis (96). Consequently, cross-validating
findings across multiple models to balance experimental stability
with clinical relevance is an essential prerequisite for advancing
pyroptosis research toward therapeutic intervention (Table I).
While no single experimental model can fully
encapsulate the intricate landscape of pyroptosis in human CKD, the
integrated application of models such as UUO, IRI and DN has
successfully identified the core pyroptotic signaling molecules
driving renal fibrogenesis. Building upon the mechanistic
breakthroughs derived from these experimental systems, a diverse
array of targeted interventions, specifically those modulating
inflammasome assembly, caspase activation and GSDM pore formation,
has emerged, delineating a promising therapeutic blueprint for
halting renal fibrosis progression and advancing clinical
translation (Table II).
Small-molecule inhibitors such as MCC950, a classic
NLRP3 inhibitor, block NLRP3 ATPase activity, prevent inflammasome
assembly (97,98), and reduce renal fibrosis
(99,100). Although MCC950 demonstrated
robust efficacy in animal studies, its clinical development was
hindered by potential hepatotoxicity (101).
In the context of clinical translation, tranilast,
an anti-fibrotic drug approved in Japan, has been shown to bind
directly to the NACHT domain of NLRP3, thereby inhibiting its
oligomerization, suppressing inflammasome assembly, and attenuating
renal fibrosis (102,103).
Similarly, BAY 11-7082, a sulfonic acid derivative,
suppresses NLRP3 activation by inhibiting both NLRP3 ATPase
activity and NF-κB signaling (104). In diabetic rat models, it has
demonstrated the potential to restore blood glucose and creatinine
levels while ameliorating renal damage (105). However, due to its high
electrophilicity, this compound is prone to broad non-specific
off-target effects, and its systemic toxicity remains a significant
barrier to clinical translation. CY-09, a novel NLRP3
inflammasome-specific inhibitor, directly binds the NLRP3 NACHT
domain and inhibits NLRP3 ATPase activity (106). Nevertheless, current evidence
for CY-09 is derived primarily from rodent studies, and it lacks
essential human pharmacokinetic data and clinical validation
regarding its long-term safety profile (107).
Natural compounds and metabolic derivatives offer
diverse mechanistic pathways for intervening in pyroptosis.
β-hydroxybutyrate, a ketone metabolite, downregulates NLRP3 by
preventing K+ efflux and reducing ASC oligomerization
and speck formation (108),
thereby ameliorating renal fibrosis in a streptozotocin-induced
diabetic model (109). However,
its clinical utility is limited by a lack of significant lifespan
extension in late-stage disease and a biphasic effect wherein
concentrations >10 mM become cytotoxic (110). Pterostilbene (PT,
trans-3,5-dimethoxy-4-hydroxystilbene) (111,112), berberine (BBR) (113,114), phloretin (115) and baicalein (22) attenuate renal fibrosis by
inhibiting NLRP3 activation through various mechanisms, including
inhibition of histone deacetylases (for example, HDAC3), induction
of autophagy, or scavenging of mitochondrial ROS. Despite their
therapeutic potential, the clinical translation of these natural
compounds is hindered by unfavorable pharmacokinetic profiles.
Specifically, BBR is limited by <1% bioavailability due to its
quaternary ammonium structure and P-glycoprotein-mediated efflux,
while PT, notwithstanding its lipophilicity, suffers from metabolic
instability and poor renal targeting. Similarly, phloretin remains
constrained by low bioavailability (~8.67%) and rapid systemic
clearance. Furthermore, baicalein's efficacy is compromised by the
mandatory intestinal hydrolysis of its glycoside form and
subsequent rapid hepatic glucuronidation, which collectively limit
effective drug accumulation in renal tissues.
Butyrate, a short-chain fatty acid (SCFA),
suppresses NLRP3-mediated pyroptosis in an adenine-induced CKD
mouse model and reduces pyroptosis-related proteins (IL-1β,
caspase-1 and GSDMD), thereby delaying CKD progression (52). It also modulates epigenetic
modifications through inhibition of histone deacetylases such as
HDAC3 to inhibit pyroptosis (116,117). Clinical trials show that oral
butyrate supplementation (4 g/day) reduces inflammatory markers in
patients with CKD (118).
Although butyrate has demonstrated potential in reducing
inflammatory markers in small-scale clinical trials, these natural
metabolites generally face significant translational bottlenecks,
such as low bioavailability and short half-lives.
Fucoidan targets NLRP3-mediated podocyte pyroptosis
through L-selectin binding, reducing GSDMD-NT expression and
proteinuria in DN (119).
However, its high molecular weight may limit renal bioavailability
(120). Pyrroloquinoline
quinone inhibits the NLRP3-caspase-1-GSDMD pathway and scavenges
mitochondrial ROS, attenuating pyroptosis in high-glucose-treated
HK-2 cells and DN mice and decreasing fibrosis markers (α-SMA and
collagen IV) (121). However,
its optimal dosage and safety profile remain to be established.
Dapansutrile (OLT1177) selectively inhibits the
NLRP3 inflammasome by binding to its NACHT domain and blocking
ATPase activity. In folic acid-induced models, it significantly
lowered serum creatinine and reduced tubulointerstitial fibrosis.
Its clinical efficacy is being evaluated in the DAPAN-DIA Phase II
trial involving 300 diabetic patients with systemic inflammation.
However, its clinical maturity is constrained by a lack of
long-term human safety data, potential infection risks, and limited
efficacy as a monotherapy during the NF-κB priming phase (122,123).
Traditional Chinese medicine (TCM) and its bioactive
constituents offer a sophisticated, multi-targeted approach to
alleviating renal fibrosis and facilitating tissue repair (124). For example, Shizhifang and
Qizhi Jiangtang Capsule suppress pyroptosis via the
NLRP3/caspase-1/GSDMD pathway, thereby improving renal injury
(125,126).
VX-765, a non-toxic small-molecule caspase-1
inhibitor, has shown promising results in the treatment of epilepsy
but remains clinically unproven for nephropathy (127,128). Ac-YVAD-cmk (129), a caspase-1 inhibitor, reduces
pyroptosis-related proteins and inflammatory factors by inhibiting
the active site of caspase-1. Quercetin (130), a natural compound, displays
potential to alleviate renal fibrosis by inhibiting the
caspase-1-mediated canonical pyroptosis pathway. Similarly, both
Ac-YVAD-cmk and quercetin show efficacy in animal models; however,
the former lacks human data, and the latter is constrained by rapid
metabolism and a bioavailability of <2% (131).
GSDMD-mediated pyroptosis plays a critical role in
the pathogenesis of renal fibrosis, positioning GSDMD inhibitors as
a potential therapeutic strategy.
Although NSA inhibits GSDMD oligomerization, its
clinical potential is limited by the complexity of its inhibitory
mechanism (136,137). Additionally, metformin
indirectly modulates the GSDMD pathway via AMP-activated protein
kinase activation, reducing collagen deposition in animal models
(138,139). Although these studies provide a
robust mechanistic foundation, most inhibitors remain in the
experimental stage, and their maturity and safety in the context of
renal disease require careful validation through dedicated clinical
trials.
Direct IL-1β inhibitors, such as the IL-1 receptor
antagonist anakinra and the monoclonal antibody canakinumab, have
shown promise in reducing fibrosis. Anakinra reduces blood pressure
and renal fibrosis in hypertensive mice but has minimal effects on
inflammation (140). The
clinical evidence for canakinumab was more extensively established
in the landmark Canakinumab Anti-inflammatory Thrombosis Outcomes
Study (CANTOS). A prespecified analysis of the CANTOS sub-cohort
with moderate CKD [baseline estimated glomerular filtration rate
(eGFR) <60 ml/min/1.73 m2] revealed that canakinumab
reduced the risk of major adverse cardiovascular events by 18% (HR
0.82), with the most pronounced clinical benefits observed in
'biologic responders' who achieved on-treatment high-sensitivity
C-reactive protein levels <2 mg/l. Although the trial did not
demonstrate a statistically significant preservation of eGFR or
reduction in albuminuria compared with placebo, the findings
underscore the pivotal role of IL-1β in driving the 'residual
inflammatory risk' prevalent in CKD populations and provide a
safety-validated framework for anti-inflammatory strategies in
cardiorenal syndromes (141).
Targeting upstream pathogenic signals, specifically
oxidative stress and pro-inflammatory priming factors, represents a
critical research frontier and therapeutic strategy for inhibiting
pyroptosis and mitigating renal fibrosis. Syringaresinol (SYR)
effectively clears ROS via the NRF2-mediated pathway, leading to
reductions in pyroptosis-associated proteins and interstitial
fibrosis in diabetic models (142). This effect is complemented by
N-acetyl-L-cysteine, an established ROS scavenger that prevents
oxidative stress-driven inflammasome activation and subsequent cell
death (37).
Furthermore, TCM and its bioactive constituents
offer a sophisticated, multi-targeted approach to modulating the
upstream microenvironment. Poria cocos has demonstrated
clinical potential as an adjunctive therapy for diabetic kidney
disease and nephrotic syndrome by regulating the NRF2 and
NF-κB/NOX4 axes via its triterpenoid and polysaccharide
constituents to alleviate proteinuria and renal damage (143). Rhubarb (Rheum palmatum)
(144) and its primary
anthraquinone derivatives (145) (for example, rhein and emodin)
exert potent effects by inhibiting NF-κB-mediated transcriptional
priming while synergistically downregulating the TGF-β/Smad and
Wnt/β-catenin pathways. These mechanisms are integrated within
formulas such as the Bushen Huoxue decoction and compounds such as
bicyclol, which collectively suppress inflammatory priming and
NLRP3 activation (34).
Concurrently, Qufeng Tongluo Decoction (146) has been shown to effectively
alleviate podocyte injury by restoring impaired autophagic flux,
thereby preserving the stability of cytoskeletal proteins such as
nephrin. In summary, pharmacological modulation of these upstream
triggers provides a robust framework for alleviating renal
pathological injury at its metabolic and inflammatory origins.
As aforementioned, the intricate crosstalk between
pyroptosis and other regulated cell death (RCD) modes drives a
pathological cascade that accelerates renal fibrosis, providing a
mechanistic rationale for targeting multiple RCD processes.
Targeting PANoptosis to inhibit organ fibrosis is
now a promising research direction. Licochalcone B and baicalin
attenuate fibrosis by inhibiting ZBP1-dependent PANoptosome
assembly, while the medicinal plant Achyranthes aspera has
been shown to improve renal function and tubular injury by
mitigating PANoptosis (87,147,148). Necrostatin-1, a RIPK1
inhibitor, suppresses overexpression of RIPK1-RIPK3-MLKL proteins
and pyroptosis-related cytokines and decreases apoptotic cell death
to reduce renal fibrosis (149). Moreover, the clinical relevance
of this mechanism is underscored by the verified upregulation of
ACSL4 in the renal tubules of patients with various CKDs, including
IgA nephropathy and DN (150).
Beyond mitigating lipid peroxidation, the ferroptosis inhibitor
liproxstatin-1 also regulates PANoptosis, effectively inhibiting
hepatic apoptosis, pyroptosis and necroptosis (151). At the transcriptional level,
the ubiquitin-specific protease 11 inhibitor mitoxantrone prevents
stabilization of the transcription factor Krüppel-like factor 4
(KLF4), thereby blocking the KLF4-induced dual pyroptotic pathways
(Caspase-1/GSDMD and Caspase-3/GSDME) to alleviate renal fibrosis
(152).
The rapid evolution of biotechnological
interventions has positioned gene editing and stem cell-derived
therapies as pioneering frontiers for disrupting the pyroptotic
cascade and addressing renal fibrosis.
CRISPR-Cas9 technology is widely used to construct
mouse models with gene knock-out or knock-in modifications to study
the role of specific genes in renal fibrosis, such as GSDMD
knock-out (153). CRISPR-Cas9
also facilitates genome-wide association studies for kidney
function to identify risk genes associated with fibrotic kidney
disease (154). A recent study
found that an improved CRISPR-Cas9 system revives gene expression
by specifically targeting methylated, silenced anti-fibrotic genes
such as RASAL1 and Klotho, thereby improving renal fibrosis
(155). However, translational
challenges remain regarding the long-term safety and delivery
efficiency of gene editing. Stem cell therapy, particularly
exosome-based therapy, represents a novel therapeutic strategy
against renal fibrosis. For example, overexpression of exosomal
miR-342-3p suppresses high glucose-induced renal interstitial
fibrosis (156). By elevating
exosomal miR-342-3p levels, pyroptosis of renal tubular epithelial
cells can be inhibited and renal function can be improved (157). Additionally,
nanotechnology-based delivery platforms such as PEGylated liposomes
could overcome the bioavailability limitations of natural compounds
such as quercetin (158).
Liposomal encapsulation of such compounds enhances therapeutic
efficacy and reduces systemic toxicity in renal fibrosis (159). Nickel-cobalt alloy magnetic
nanocrystals (an inorganic nanomaterial, exhibits broad-spectrum
inflammasome inhibitory effects, simultaneously suppressing the
activation of three inflammasomes, NLRP3, NLR family CARD domain
containing 4 and absent in melanoma 2. This property renders them
both anti-inflammatory agents and ideal delivery vehicles for
anti-inflammatory drugs (160).
In summary, this strategic direction is catalyzing a clinical
paradigm shift from palliative symptom management toward targeted
molecular repair.
In conclusion, GSDMD-mediated cytolytic rupture
serves as the central pathogenic hub in renal fibrosis, acting as
the terminal executioner that perpetuates the pro-fibrotic milieu.
However, successful clinical translation requires moving beyond
descriptive research to address several strategic gaps. Current
investigations fail to elucidate how the renal microenvironment,
specifically chronic hypoxia, electrolyte flux, and uremic toxins,
differentially modulates pyroptosis activation thresholds across
distinct nephron segments. Furthermore, the long-term safety of
suppressing innate immune sentinels such as NLRP3 remains poorly
defined; given its critical role in host defense, systemic
inhibition may heighten susceptibility to opportunistic fungal or
viral infections in immunocompromised patients with CKD. Standard
experimental models also exhibit substantial translatability gaps,
including functional compensation by the contralateral kidney in
UUO models and strain-dependent resistance to fibrosis (for
example, in C57BL/6 mice) in 5/6 NX models, factors that can
obscure true therapeutic efficacy. Future research must prioritize
multi-target synergistic interventions, particularly those directed
at integrative molecular scaffolds such as the PANoptosome, to
circumvent cellular escape mechanisms. The translational roadmap
necessitates longitudinal functional validation using GFR-based
metrics, identification of non-invasive urinary biomarkers for
patient stratification, and development of renal-targeted precision
delivery systems to maximize anti-fibrotic potency while minimizing
systemic toxicity.
Not applicable.
YT wrote the original draft, visualized data, and
wrote, reviewed and edited the manuscript. QM wrote the original
draft and visualized data. HF and HS wrote the original draft. JL
and QZ wrote, reviewed and edited the manuscript, supervised the
study and acquired funding. All authors read and approved the final
version of the manuscript. Data authentication is not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present study was supported by the Sichuan Science and
Technology Program (grant no. 2026NSFSC0641), the Southwest Medical
University Technology Program (grant no. 2023QN019), the National
Natural Science Foundation of China (grant no. 82205002), the
Science and Technology Research Special Project of Sichuan
Administration of Traditional Chinese Medicine (grant no.
2024MS524) and the Special project of integrated Chinese and
Western medicine, Southwest Medical University (grant no.
2023ZYQJ04).
|
1
|
Romagnani P, Remuzzi G, Glassock R, Levin
A, Jager KJ, Tonelli M, Massy Z, Wanner C and Anders HJ: Chronic
kidney disease. Nat Rev Dis Primers. 3:170882017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liyanage T, Toyama T, Hockham C, Ninomiya
T, Perkovic V, Woodward M, Fukagawa M, Matsushita K,
Praditpornsilpa K, Hooi LS, et al: Prevalence of chronic kidney
disease in Asia: a systematic review and analysis. BMJ Glob Health.
7:e0075252022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Glassock RJ, Warnock DG and Delanaye P:
The global burden of chronic kidney disease: Estimates, variability
and pitfalls. Nat Rev Nephrol. 13:104–114. 2017. View Article : Google Scholar
|
|
4
|
Youhua L: Kidney Fibrosis: Fundamental
questions, challenges, and perspectives. Integr Med Nephrol Androl.
11:e24–00027. 2024.
|
|
5
|
Liu Y, Lei H, Zhang W, Xing Q, Liu R, Wu
S, Liu Z, Yan Q, Li W, Liu X and Hu Y: Pyroptosis in renal
inflammation and fibrosis: current knowledge and clinical
significance. Cell Death Dis. 14:4722023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kok HM, Falke LL, Goldschmeding R and
Nguyen TQ: Targeting CTGF, EGF and PDGF pathways to prevent
progression of kidney disease. Nat Rev Nephrol. 10:700–711. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Humphreys BD: Mechanisms of renal
fibrosis. Annu Rev Physiol. 80:309–326. 2018. View Article : Google Scholar
|
|
8
|
Nastase MV, Zeng-Brouwers J, Wygrecka M
and Schaefer L: Targeting renal fibrosis: Mechanisms and drug
delivery systems. Adv Drug Deliv Rev. 129:295–307. 2018. View Article : Google Scholar
|
|
9
|
Khwaja A, El Kossi M, Floege J and El
Nahas M: The management of CKD: A look into the future. Kidney Int.
72:1316–1323. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cuevas S and Pelegrín P: Pyroptosis and
redox balance in kidney diseases. Antioxid Redox Signal. 35:40–60.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Song Z, Gong Q and Guo J: Pyroptosis:
Mechanisms and links with fibrosis. Cells. 10:35092021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hsu SK, Li CY, Lin IL, Syue WJ, Chen YF,
Cheng KC, Teng YN, Lin YH, Yen CH and Chiu CC: Inflammation-related
pyroptosis, a novel programmed cell death pathway, and its
crosstalk with immune therapy in cancer treatment. Theranostics.
11:8813–8835. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kim YG, Kim SM, Kim KP, Lee SH and Moon
JY: The role of inflammasome-dependent and inflammasome-independent
NLRP3 in the kidney. Cells. 8:13892019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Du T, Gao J, Li P, Wang Y, Qi Q, Liu X, Li
J, Wang C and Du L: Pyroptosis, metabolism, and tumor immune
microenvironment. Clin Transl Med. 11:e4922021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fu H, Gu YH, Tan J, Yang YN and Wang GH:
CircACTR2 in macrophages promotes renal fibrosis by activating
macrophage inflammation and epithelial-mesenchymal transition of
renal tubular epithelial cells. Cell Mol Life Sci. 79:2532022.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cheng P, Li S and Chen H: Macrophages in
lung injury, repair, and fibrosis. Cells. 10:4362021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xiao Y, Zhao C, Tai Y, Li B, Lan T, Lai E,
Dai W, Guo Y, Gan C, Kostallari E, et al: STING mediates hepatocyte
pyroptosis in liver fibrosis by Epigenetically activating the NLRP3
inflammasome. Redox Biol. 62:1026912023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liang Q, Cai W, Zhao Y, Xu H, Tang H, Chen
D, Qian F and Sun L: Lycorine ameliorates bleomycin-induced
pulmonary fibrosis via inhibiting NLRP3 inflammasome activation and
pyroptosis. Pharmacol Res. 158:1048842020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu P, Zhang Z and Li Y: Relevance of the
pyroptosis-related inflammasome pathway in the pathogenesis of
diabetic kidney disease. Front Immunol. 12:6034162021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yin W, Wang JH, Liang YM, Liu KH and Chen
Y and Chen Y: Neferine targeted the NLRC5/NLRP3 pathway to inhibit
M1-type polarization and pyroptosis of macrophages to improve
hyperuricemic nephropathy. Curr Mol Med. 25:90–111. 2025.
View Article : Google Scholar
|
|
21
|
Cheng Y, Lu Z, Mao T, Song Y, Qu Y, Chen
X, Chen K, Liu K and Zhang C: Magnoflorine ameliorates chronic
kidney disease in high-fat and high-fructose-fed mice by promoting
parkin/PINK1-Dependent mitophagy to inhibit
NLRP3/Caspase-1-mediated pyroptosis. J Agric Food Chem.
72:12775–12787. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Feng M, Luo F, Wu H, Chen Y, Zuo J, Weng
X, Chen G and Zhong J: Network pharmacology analysis and
machine-learning models confirmed the ability of YiShen HuoXue
decoction to alleviate renal fibrosis by inhibiting pyroptosis.
Drug Des Devel Ther. 17:3169–3192. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang L, Liu W, Li S, Wang J, Sun D, Li H,
Zhang Z, Hu Y and Fang J: Astragaloside IV alleviates renal
fibrosis by inhibiting renal tubular epithelial cell pyroptosis
induced by urotensin II through regulating the cAMP/PKA signaling
pathway. PLoS One. 19:e03043652024. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Islamuddin M and Qin X: Renal macrophages
and NLRP3 inflammasomes in kidney diseases and therapeutics. Cell
Death Discov. 10:2292024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li Y and Guo B: GSDMD-mediated pyroptosis:
Molecular mechanisms, diseases and therapeutic targets. Mol Biomed.
6:112025. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lorenz G, Darisipudi MN and Anders HJ:
Canonical and non-canonical effects of the NLRP3 inflammasome in
kidney inflammation and fibrosis. Nephrol Dial Transplant.
29:41–48. 2014. View Article : Google Scholar
|
|
27
|
Zhang H and Wang Z: Effect and regulation
of the NLRP3 inflammasome during renal fibrosis. Front Cell Dev
Biol. 7:3792020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gao H, Xie T, Li Y, Xu Z, Song Z, Yu H,
Zhou H, Li W, Yun C, Guan B, et al: Role of gasdermins in chronic
kidney disease. Front Immunol. 16:15577072025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Burdette BE, Esparza AN, Zhu H and Wang S:
Gasdermin D in pyroptosis. Acta Pharm Sin B. 11:2768–2782. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Elias EE, Lyons B and Muruve DA:
Gasdermins and pyroptosis in the kidney. Nat Rev Nephrol.
19:337–350. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yang X, Luo Q, Wu Z, Wang C, Yang Y, Zheng
L, Li K, Zhao L and Jurong Y: Tanshinone IIA reduces
tubulointerstitial fibrosis by suppressing GSDMD-mediated
pyroptosis. Pharm Biol. 63:364–373. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zuo Y, Chen L, Gu H, He X, Ye Z, Wang Z,
Shao Q and Xue C: GSDMD-mediated pyroptosis: A critical mechanism
of diabetic nephropathy. Expert Rev Mol Med. 23:e232021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li S, Sun Y, Song M, Song Y, Fang Y, Zhang
Q, Li X, Song N, Ding J, Lu M and Hu G:
NLRP3/caspase-1/GSDMD-mediated pyroptosis exerts a crucial role in
astrocyte pathological injury in mouse model of depression. JCI
Insight. 6:e1468522021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liao L, Tao P, Xu Q, Chen J, Liu W, Hu J
and Lu J: Bushen Huoxue formula protects against renal fibrosis and
pyroptosis in chronic kidney disease by inhibiting
ROS/NLRP3-mediated inflammasome activation. Ren Fail.
46:23544442024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chung SD, Lai TY, Chien CT and Yu HJ:
Activating Nrf-2 signaling depresses unilateral ureteral
obstruction-evoked mitochondrial stress-related autophagy,
apoptosis and pyroptosis in kidney. PLoS One. 7:e472992012.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hao J, Wang S, Guo X, Guo X and Hao L: The
axis of miR-30a/AIF-1/TRPC6/calcineurin A/NFAT2 regulated the death
modalities and inflammation of renal tubular epithelial cells in
diabetic kidney disease via exosome. Life Sci. 377:1237602025.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liao L, Tao P, Xu Q, Chen W, Chen J, Liu
W, Liu W, Hu J and Lu J: TRIM6 promotes ROS-mediated inflammasome
activation and pyroptosis in renal tubular epithelial cells via
ubiquitination and degradation of GPX3 protein. Front Biosci
(Landmark Ed). 29:582024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jiang YJ, Jin J, Nan QY, Ding J, Cui S,
Xuan MY, Piao MH, Piao SG, Zheng HL, Jin JZ, et al: Coenzyme Q10
attenuates renal fibrosis by inhibiting RIP1-RIP3-MLKL-mediated
necroinflammation via Wnt3α/β-catenin/GSK-3β signaling in
unilateral ureteral obstruction. Int Immunopharmacol.
108:1088682022. View Article : Google Scholar
|
|
39
|
Wu Y, Pi D, Zhou S, Yi Z, Dong Y, Wang W,
Ye H, Chen Y, Zuo Q and Ouyang M: Ginsenoside Rh3 induces
pyroptosis and ferroptosis through the Stat3/p53/NRF2 axis in
colorectal cancer cells. Acta Biochim Biophys Sin (Shanghai).
55:587–600. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Huang Y, Xu W and Zhou R: NLRP3
inflammasome activation and cell death. Cell Mol Immunol.
18:2114–2127. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Tang R, Xu J, Zhang B, Liu J, Liang C, Hua
J, Meng Q, Yu X and Shi S: Ferroptosis, necroptosis, and pyroptosis
in anticancer immunity. J Hematol Oncol. 13:1102020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Frank D and Vince JE: Pyroptosis versus
necroptosis: Similarities, differences, and crosstalk. Cell Death
Differ. 26:99–114. 2019. View Article : Google Scholar
|
|
43
|
Zheng M and Kanneganti TD: The regulation
of the ZBP1-NLRP3 inflammasome and its implications in pyroptosis,
apoptosis, and necroptosis (PANoptosis). Immunol Rev. 297:26–38.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Guo Y, Zhao Y, Qiao Y, Xing Y, Fang Y,
Zhao Y, Yang H, Chen Y and Yang B: Targeting panoptosis: A
narrative review of its therapeutic potential in kidney disease.
BMC Nephrol. 26:5452025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
He X, Yan G and Yingyong Z: Targeting gut
microbiota as a promising therapy for chronic kidney disease.
Integr Med Nephrol Androl. 12:e25–00026. 2025. View Article : Google Scholar
|
|
46
|
Li XJ, Shan QY, Wu X, Miao H and Zhao YY:
Gut microbiota regulates oxidative stress and inflammation: A
double-edged sword in renal fibrosis. Cell Mol Life Sci.
81:4802024. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang J, Jiang M, Li X, Ye Y, Xie Y, Wu T,
Chen Y, Yu H, Wu H, Yang Z and Zhou E: Inulin supplementation
alleviates ochratoxin A-induced kidney injury through modulating
intestinal microbiota. J Agric Food Chem. 72:18682–18696. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Miao EA, Leaf IA, Treuting PM, Mao DP,
Dors M, Sarkar A, Warren SE, Wewers MD and Aderem A:
Caspase-1-induced pyroptosis is an innate immune effector mechanism
against intracellular bacteria. Nat Immunol. 11:1136–1142. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhang X, Qu H, Yang T, Kong X and Zhou H:
Regulation and functions of NLRP3 inflammasome in cardiac fibrosis:
Current knowledge and clinical significance. Biomed Pharmacother.
143:1122192021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shi J, Zhao Y, Wang K, Shi X, Wang Y,
Huang H, Zhuang Y, Cai T, Wang F and Shao F: Cleavage of GSDMD by
inflammatory caspases determines pyroptotic cell death. Nature.
526:660–665. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ram C, Gairola S, Verma S, Mugale MN,
Bonam SR, Murty US and Sahu BD: Biochanin A ameliorates nephropathy
in High-Fat diet/streptozotocin-induced diabetic rats: Effects on
NF-kB/NLRP3 axis, pyroptosis, and fibrosis. Antioxidants (Basel).
12:10522023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Tian X, Zeng Y, Tu Q, Jiao Y, Yao S, Chen
Y, Sun L, Xia Q, Luo Y, Yuan L and Jiang Q: Butyrate alleviates
renal fibrosis in CKD by regulating NLRP3-mediated pyroptosis via
the STING/NF-κB/p65 pathway. Int Immunopharmacol. 124(Pt B):
1110102023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Al Mamun A, Ara Mimi A, Wu Y, Zaeem M,
Abdul Aziz M, Aktar Suchi S, Alyafeai E, Munir F and Xiao J:
Pyroptosis in diabetic nephropathy. Clin Chim Acta. 523:131–143.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Martinon F, Pétrilli V, Mayor A, Tardivel
A and Tschopp J: Gout-associated uric acid crystals activate the
NALP3 inflammasome. Nature. 440:237–241. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Xu Z, Tang C, Song X, Liu Z, Zhou J, Shi
Q, Yu C and Xu C: High uric acid exacerbates nonalcoholic
steatohepatitis through NLRP3 inflammasome and gasdermin D-mediated
pyroptosis. J Biol Chem. 301:1102492025. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zheng J, Hu Q, Zou X, Xu G and Cao Y:
Uranium induces kidney cells pyroptosis in culture involved in
ROS/NLRP3/caspase-1 signaling. Free Radic Res. 56:40–52. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wei Y, You Y, Zhang J, Ban J, Min H, Li C
and Chen J: Crystalline silica-induced macrophage pyroptosis
interacting with mitophagy contributes to pulmonary fibrosis via
modulating mitochondria homeostasis. J Hazard Mater.
454:1315622023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Liu H, Xiang X, Shi C, Guo J, Ran T, Lin
J, Dong F, Yang J and Miao H: Oxidative stress and inflammation in
renal fibrosis: Novel molecular mechanisms and therapeutic targets.
Chem Biol Interact. 421:1117842025. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Li JM, Song ZH, Li Y, Chen HW, Li H, Yuan
L, Li J, Lv WY, Liu L and Wang N: NR4A1 silencing alleviates
high-glucose-stimulated HK-2 cells pyroptosis and fibrosis via
hindering NLRP3 activation and PI3K/AKT pathway. World J Diabetes.
16:975442025.PubMed/NCBI
|
|
60
|
Lan J, Xu B, Shi X, Pan Q and Tao Q:
WTAP-mediated N6-methyladenosine modification of NLRP3 mRNA in
kidney injury of diabetic nephropathy. Cell Mol Biol Lett.
27:512022. View Article : Google Scholar
|
|
61
|
Zheng S, Que X, Wang S, Zhou Q, Xing X,
Chen L, Hou C, Ma J, An P, Peng Y, et al: ZDHHC5-mediated NLRP3
palmitoylation promotes NLRP3-NEK7 interaction and inflammasome
activation. Mol Cell. 83:4570–4585.e7. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Bani-Hani AH, Leslie JA, Asanuma H,
Dinarello CA, Campbell MT, Meldrum DR, Zhang H, Hile K and Meldrum
KK: IL-18 neutralization ameliorates obstruction-induced
epithelial-mesenchymal transition and renal fibrosis. Kidney Int.
76:500–511. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Liu X, Long X, Liu W, Zhao Y, Hayashi T,
Yamato M, Mizuno K, Fujisaki H, Hattori S, Tashiro SI, et al: Type
I collagen induces mesenchymal cell differentiation into
myofibroblasts through YAP-induced TGF-β1 activation. Biochimie.
150:110–130. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Park JY, Kim JH, Park CH, Kim SH, Kim IH
and Cho WG: Polyhexamethylene guanidine phosphate induces
pyroptosis via reactive oxygen species-regulated mitochondrial
dysfunction in bronchial epithelial cells. Toxicology.
505:1538272024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Ramos-Tovar E and Muriel P: Molecular
mechanisms that link oxidative stress, inflammation, and fibrosis
in the liver. Antioxidants (Basel). 9:12792020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Wu P, Liao T, Ma Z, Wei Y, Yin S, Huang Z
and Mao J: Macrophage pyroptosis promotes synovial fibrosis through
the HMGB1/TGF-β1 axis: An in vivo and in vitro study. In Vitro Cell
Dev Biol Anim. 59:289–299. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Sun J, Ge X, Wang Y, Niu L, Tang L and Pan
S: USF2 knockdown downregulates THBS1 to inhibit the TGF-β
signaling pathway and reduce pyroptosis in sepsis-induced acute
kidney injury. Pharmacol Res. 176:1059622022. View Article : Google Scholar
|
|
68
|
Li Z, Ji S, Jiang ML, Xu Y and Zhang CJ:
The regulation and modification of GSDMD signaling in diseases.
Front Immunol. 13:8939122022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
He W, Wan H, Hu L, Chen P, Wang X, Huang
Z, Yang ZH, Zhong CQ and Han J: Gasdermin D is an executor of
pyroptosis and required for interleukin-1β secretion. Cell Res.
25:1285–1298. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Wang Y, Li Y, Chen Z, Yuan Y, Su Q, Ye K,
Chen C, Li G, Song Y, Chen H and Xu Y: GSDMD-dependent neutrophil
extracellular traps promote macrophage-to-myofibroblast transition
and renal fibrosis in obstructive nephropathy. Cell Death Dis.
13:6932022. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Du G, Healy LB, David L, Walker C, El-Baba
TJ, Lutomski CA, Goh B, Gu B, Pi X, Devant P, et al: ROS-dependent
S-palmitoylation activates cleaved and intact gasdermin D. Nature.
630:437–446. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Liu Z, Li S, Wang C, Vidmar KJ, Bracey S,
Li L, Willard B, Miyagi M, Lan T, Dickinson BC, et al:
Palmitoylation at a conserved cysteine residue facilitates
gasdermin D-mediated pyroptosis and cytokine release. Proc Natl
Acad Sci USA. 121:e24008831212024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Rühl S and Broz P: Caspase-11 activates a
canonical NLRP3 inflammasome by promoting K(+) efflux. Eur J
Immunol. 45:2927–2936. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Li Y, Yuan Y, Huang ZX, Chen H, Lan R,
Wang Z, Lai K, Chen H, Chen Z, Zou Z, et al: GSDME-mediated
pyroptosis promotes inflammation and fibrosis in obstructive
nephropathy. Cell Death Differ. 28:2333–2350. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Wu M, Xia W, Jin Q, Zhou A, Wang Q, Li S,
Huang S, Zhang A, Zhang Y, Li Y and Jia Z: Gasdermin E deletion
attenuates ureteral obstruction- and 5/6 nephrectomy-induced renal
fibrosis and kidney dysfunction. Front Cell Dev Biol. 9:7541342021.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Chen Z, Chen C, Lai K, Wu C, Wu F, Chen Z,
Ye K, Xie J, Ma H, Chen H, et al: GSDMD and GSDME synergy in the
transition of acute kidney injury to chronic kidney disease.
Nephrol Dial Transplant. 39:1344–1359. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Kang L, Dai J, Wang Y, Shi P, Zou Y, Pei
J, Tian Y, Zhang J, Buranasudja VC, Chen J, et al: Blocking
Caspase-1/Gsdmd and Caspase-3/-8/Gsdme pyroptotic pathways rescues
silicosis in mice. PLoS Genet. 18:e10105152022. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Zhang JY, Zhou B, Sun RY, Ai YL, Cheng K,
Li FN, Wang BR, Liu FJ, Jiang ZH, Wang WJ, et al: The metabolite
α-KG induces GSDMC-dependent pyroptosis through death receptor
6-activated caspase-8. Cell Res. 31:980–997. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Fritsch M, Günther SD, Schwarzer R, Albert
MC, Schorn F, Werthenbach JP, Schiffmann LM, Stair N, Stocks H,
Seeger JM, et al: Caspase-8 is the molecular switch for apoptosis,
necroptosis and pyroptosis. Nature. 575:683–687. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Ai Y, Meng Y, Yan B, Zhou Q and Wang X:
The biochemical pathways of apoptotic, necroptotic, pyroptotic, and
ferroptotic cell death. Mol Cell. 84:170–179. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Zhang H, Huang C, Zhang D and Zhu Y:
Pioglitazone protects against hypoxia-induced cardiomyocyte
apoptosis through inhibiting NLRP3/Caspase-1 pathway in vivo and in
vitro. Int Heart J. 63:893–903. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Hou Y, Feng Q, Wei C, Cao F, Liu D, Pan S,
Shi Y, Liu Z and Liu F: Emerging role of PANoptosis in kidney
diseases: Molecular mechanisms and therapeutic opportunities.
Apoptosis. 30:579–596. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zhuang S, Li F, Wang L, Lai Z, Li D, Wu H,
Wu J, Qu J, Zhang X, Zhang M, et al: Neutrophil extracellular
trap-derived double-stranded RNA aggravates PANoptosis in renal
ischemia reperfusion injury. Cell Commun Signal. 23:1402025.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Guo C, Fu R, Zhou M, Wang S, Huang Y, Hu
H, Zhao J, Gaskin F, Yang N and Fu SM: Pathogenesis of lupus
nephritis: RIP3 dependent necroptosis and NLRP3 inflammasome
activation. J Autoimmun. 103:1022862019. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Chen Q, He H, Wang G, Huang L, Yang J, Sun
Y, Zhang Z and Liao X: Integrated transcriptomic analysis
identifies TAP1 as a key regulator of PANoptosis in diabetic kidney
disease tubular injury. Inflammation. 49:802026. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Ren L, Yang Y, Li W, Zheng X, Liu J, Li S,
Yang H, Zhang Y, Ge B, Zhang S, et al: CDK1 serves as a therapeutic
target of adrenocortical carcinoma via regulating
epithelial-mesenchymal transition, G2/M phase transition, and
PANoptosis. J Transl Med. 20:4442022. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
You YP, Yan L, Ke HY, Li YP, Shi ZJ, Zhou
ZY, Yang HY, Yuan T, Gan YQ, Lu N, et al: Baicalin inhibits
PANoptosis by blocking mitochondrial Z-DNA formation and
ZBP1-PANoptosome assembly in macrophages. Acta Pharmacol Sin.
46:430–447. 2025. View Article : Google Scholar
|
|
88
|
Zeng YL, Liu LY, Ma TZ, Liu Y, Liu B, Liu
W, Shen QH, Wu C and Mao ZW: Iridium(III) photosensitizers induce
simultaneous pyroptosis and ferroptosis for multi-network
synergistic tumor immunotherapy. Angew Chem Int Ed Engl.
63:e2024108032024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Hsu CG, Chávez CL, Zhang C, Sowden M, Yan
C and Berk BC: The lipid peroxidation product 4-hydroxynonenal
inhibits NLRP3 inflammasome activation and macrophage pyroptosis.
Cell Death Differ. 29:1790–1803. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Torrico S, Hotter G, Muñoz Á, Calle P,
García M, Poch E and Játiva S: PBMC therapy reduces cell death and
tissue fibrosis after acute kidney injury by modulating the pattern
of monocyte/macrophage survival in tissue. Biomed Pharmacother.
178:1171862024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Zhang Q, Zhang X, Su H, An N, Gong S, Chen
X, Mahyar K, Huafeng L and Chen Y: Selective autophagy in kidney
diseases: The tip of the Iceberg. Integr Med Nephrol Androl.
12:e24–00036. 2025. View Article : Google Scholar
|
|
92
|
An Z and Ding W: Syntaxin17 restores
lysosomal function and inhibits pyroptosis caused by acinetobacter
baumannii. J Microbiol. 62:315–325. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Hu Y, Shi Y, Chen H, Tao M, Zhou X, Li J,
Ma X, Wang Y and Liu N: Blockade of autophagy prevents the
progression of hyperuricemic nephropathy through inhibiting NLRP3
inflammasome-mediated pyroptosis. Front Immunol. 13:8584942022.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Bao YW, Yuan Y, Chen JH and Lin WQ: Kidney
disease models: Tools to identify mechanisms and potential
therapeutic targets. Zool Res. 39:72–86. 2018.PubMed/NCBI
|
|
95
|
Eddy AA, López-Guisa JM, Okamura DM and
Yamaguchi I: Investigating mechanisms of chronic kidney disease in
mouse models. Pediatr Nephrol. 27:1233–1247. 2012. View Article : Google Scholar
|
|
96
|
Kim K, Anderson EM, Thome T, Lu G, Salyers
ZR, Cort TA, O'Malley KA, Scali ST and Ryan TE: Skeletal myopathy
in CKD: A comparison of adenine-induced nephropathy and 5/6
nephrectomy models in mice. Am J Physiol Renal Physiol.
321:F106–F119. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zahid A, Li B, Kombe AJK, Jin T and Tao J:
Pharmacological Inhibitors of the NLRP3 Inflammasome. Front
Immunol. 10:25382019. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Coll RC, Hill JR, Day CJ, Zamoshnikova A,
Boucher D, Massey NL, Chitty JL, Fraser JA, Jennings MP, Robertson
AAB and Schroder K: MCC950 directly targets the NLRP3
ATP-hydrolysis motif for inflammasome inhibition. Nat Chem Biol.
15:556–559. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Zhang C, Zhu X, Li L, Ma T, Shi M, Yang Y
and Fan Q: A small molecule inhibitor MCC950 ameliorates kidney
injury in diabetic nephropathy by inhibiting NLRP3 inflammasome
activation. Diabetes Metab Syndr Obes. 12:1297–1309. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Østergaard JA, Jha JC, Sharma A, Dai A,
Choi JSY, de Haan JB, Cooper ME and Jandeleit-Dahm K: Adverse renal
effects of NLRP3 inflammasome inhibition by MCC950 in an
interventional model of diabetic kidney disease. Clin Sci (Lond).
136:167–180. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Li H, Guan Y, Liang B, Ding P, Hou X, Wei
W and Ma Y: Therapeutic potential of MCC950, a specific inhibitor
of NLRP3 inflammasome. Eur J Pharmacol. 928:1750912022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Huang Y, Jiang H, Chen Y, Wang X, Yang Y,
Tao J, Deng X, Liang G, Zhang H, Jiang W and Zhou R: Tranilast
directly targets NLRP3 to treat inflammasome-driven diseases. EMBO
Mol Med. 10:e86892018. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Kaneyama T, Kobayashi S, Aoyagi D and
Ehara T: Tranilast modulates fibrosis, epithelial-mesenchymal
transition and peritubular capillary injury in unilateral ureteral
obstruction rats. Pathology. 42:564–573. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Chiazza F, Couturier-Maillard A, Benetti
E, Mastrocola R, Nigro D, Cutrin JC, Serpe L, Aragno M, Fantozzi R,
Ryffel B, et al: Targeting the NLRP3 inflammasome to reduce
diet-induced metabolic abnormalities in mice. Mol Med.
21:1025–1037. 2016. View Article : Google Scholar :
|
|
105
|
Kolati SR, Kasala ER, Bodduluru LN,
Mahareddy JR, Uppulapu SK, Gogoi R, Barua CC and Lahkar M: BAY
11-7082 ameliorates diabetic nephropathy by attenuating
hyperglycemia-mediated oxidative stress and renal inflammation via
NF-κB pathway. Environ Toxicol Pharmacol. 39:690–699. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Jiang H, He H, Chen Y, Huang W, Cheng J,
Ye J, Wang A, Tao J, Wang C, Liu Q, et al: Identification of a
selective and direct NLRP3 inhibitor to treat inflammatory
disorders. J Exp Med. 214:3219–3238. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Yang M and Zhao L: The selective
NLRP3-inflammasome inhibitor CY-09 ameliorates kidney injury in
diabetic nephropathy by inhibiting NLRP3-inflammasome activation.
Curr Med Chem. 30:3261–3270. 2023. View Article : Google Scholar
|
|
108
|
Youm YH, Nguyen KY, Grant RW, Goldberg EL,
Bodogai M, Kim D, D'Agostino D, Planavsky N, Lupfer C, Kanneganti
TD, et al: The ketone metabolite β-hydroxybutyrate blocks NLRP3
inflammasome-mediated inflammatory disease. Nat Med. 21:263–269.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Aisyah R, Katsuya S, Miyata K, Chen Z,
Horii M, Kudo A, Kumrungsee T, Tsubota J and Yanaka N:
β-hydroxybutyrate attenuates diabetic renal and muscular
pathologies in a streptozotocin-induced diabetic model. Nutrition.
137:1127922025. View Article : Google Scholar
|
|
110
|
Schreier LP, Zhu Z, Kusunoki Y, Li C, Ku
J, Klaus M and Anders HJ: Oral Ketone β -hydroxybutyrate supplement
retards the loss of GFR in alport mice on dual renin-angiotensin
system/sodium-glucose transporter 2 blockade. Kidney360.
6:1085–1095. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wang YJ, Chen YY, Hsiao CM, Pan MH, Wang
BJ, Chen YC, Ho CT, Huang KC and Chen RJ: Induction of autophagy by
pterostilbene contributes to the prevention of renal fibrosis via
attenuating NLRP3 inflammasome activation and
epithelial-mesenchymal transition. Front Cell Dev Biol. 8:4362020.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Pan J, Shi M, Li L, Liu J, Guo F, Feng Y,
Ma L and Fu P: Pterostilbene, a bioactive component of blueberries,
alleviates renal fibrosis in a severe mouse model of hyperuricemic
nephropathy. Biomed Pharmacother. 109:1802–1808. 2019. View Article : Google Scholar
|
|
113
|
Li Z and Zhang W: Protective effect of
berberine on renal fibrosis caused by diabetic nephropathy. Mol Med
Rep. 16:1055–1062. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Ma Z, Zhu L, Wang S, Guo X, Sun B, Wang Q
and Chen L: Berberine protects diabetic nephropathy by suppressing
epithelial-to-mesenchymal transition involving the inactivation of
the NLRP3 inflammasome. Ren Fail. 44:923–932. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Cui D, Liu S, Tang M, Lu Y, Zhao M, Mao R,
Wang C, Yuan Y, Li L, Chen Y, et al: Phloretin ameliorates
hyperuricemia-induced chronic renal dysfunction through inhibiting
NLRP3 inflammasome and uric acid reabsorption. Phytomedicine.
66:1531112020. View Article : Google Scholar
|
|
116
|
Wu SE, Hashimoto-Hill S, Woo V, Eshleman
EM, Whitt J, Engleman L, Karns R, Denson LA, Haslam DB and Alenghat
T: Microbiota-derived metabolite promotes HDAC3 activity in the
gut. Nature. 586:108–112. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Yan M, Li X, Sun C, Tan J, Liu Y, Li M, Qi
Z, He J, Wang D and Wu L: Sodium butyrate attenuates AGEs-induced
oxidative stress and inflammation by inhibiting autophagy and
affecting cellular metabolism in THP-1 cells. Molecules.
27:87152022. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Li HB, Xu ML, Xu XD, Tang YY, Jiang HL, Li
L, Xia WJ, Cui N, Bai J, Dai ZM, et al: Faecalibacterium
prausnitzii Attenuates CKD via Butyrate-Renal GPR43 Axis. Circ Res.
131:e120–e134. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Wang MZ, Wang J, Cao DW, Tu Y, Liu BH,
Yuan CC, Li H, Fang QJ, Chen JX, Fu Y, et al: Fucoidan alleviates
renal fibrosis in diabetic kidney disease via inhibition of NLRP3
inflammasome-mediated podocyte pyroptosis. Front Pharmacol.
13:7909372022. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Zahan MS, Hasan A, Rahman MH, Meem KN,
Moni A, Hannan MA and Uddin MJ: Protective effects of fucoidan
against kidney diseases: Pharmacological insights and future
perspectives. Int J Biol Macromol. 209:2119–2129. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Qu X, Zhai B, Liu Y, Chen Y, Xie Z, Wang
Q, Wu Y, Liu Z, Chen J, Mei S, et al: Pyrroloquinoline quinone
ameliorates renal fibrosis in diabetic nephropathy by inhibiting
the pyroptosis pathway in C57BL/6 mice and human kidney 2 cells.
Biomed Pharmacother. 150:1129982022. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Bai F, Wang D and Wu Y: Dapansutrile in
multidisciplinary therapeutic applications: mechanisms and clinical
perspectives. Front Pharmacol. 16:17311652025. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Elsayed MS, Abu-Elsaad NM and Nader MA:
The NLRP3 inhibitor dapansutrile attenuates folic acid induced
nephrotoxicity via inhibiting inflammasome/caspase-1/IL axis and
regulating autophagy/proliferation. Life Sci. 285:1199742021.
View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Song L, Zhang W, Tang SY, Luo SM, Xiong
PY, Liu JY, Hu HC, Chen YQ, Jia B, Yan QH, et al: Natural products
in traditional Chinese medicine: Molecular mechanisms and
therapeutic targets of renal fibrosis and state-of-the-art drug
delivery systems. Biomed Pharmacother. 170:1160392024. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Zhou J, Wang C, Zhang X, Wu Z, Wu Y, Li D
and Gao J: Shizhifang ameliorates pyroptosis of renal tubular
epithelial cells in hyperuricemia through inhibiting NLRP3
inflammasome. J Ethnopharmacol. 317:1167772023. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Su S, Guo Z, Yang H, Liu H, Tang J and
Jiang X: Mechanism of Qizhi Jiangtang capsule inhibits podocyte
pyroptosis to improve kidney injury in diabetes nephropathy by
regulating NLRP3/caspase-1/GSDMD pathway. Xi Bao Yu Fen Zi Mian Yi
Xue Za Zhi. 41:204–210. 2025.In Chinese. PubMed/NCBI
|
|
127
|
Flores J, Noël A, Foveau B, Lynham J,
Lecrux C and LeBlanc AC: Caspase-1 inhibition alleviates cognitive
impairment and neuropathology in an Alzheimer's disease mouse
model. Nat Commun. 9:39162018. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Wen S, Deng F, Li L, Xu L, Li X and Fan Q:
VX-765 ameliorates renal injury and fibrosis in diabetes by
regulating caspase-1-mediated pyroptosis and inflammation. J
Diabetes Investig. 13:22–33. 2022. View Article : Google Scholar :
|
|
129
|
Yang M, Fang JT, Zhang NS, Qin LJ, Zhuang
YY, Wang WW, Zhu HP, Zhang YJ, Xia P and Zhang Y:
Caspase-1-Inhibitor AC-YVAD-CMK inhibits pyroptosis and ameliorates
acute kidney injury in a model of sepsis. Biomed Res Int.
2021:66366212021. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Liu X, Liang Q, Qin Y, Chen Z and Yue R:
Advances and perspectives on the anti-fibrotic mechanisms of the
quercetin. Am J Chin Med. 53:1411–1440. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Wang W and Wu W: Therapeutic effects of
quercetin on renal fibrosis and injury. Integr Med Nephrol Androl.
12:e24–00051. 2025. View Article : Google Scholar
|
|
132
|
Hu JJ, Liu X, Xia S, Zhang Z, Zhang Y,
Zhao J, Ruan J, Luo X, Lou X, Bai Y, et al: FDA-approved disulfiram
inhibits pyroptosis by blocking gasdermin D pore formation. Nat
Immunol. 21:736–745. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Silva CMS, Wanderley CWS, Veras FP, Sonego
F, Nascimento DC, Gonçalves AV, Martins TV, Cólon DF, Borges VF,
Brauer VS, et al: Gasdermin D inhibition prevents multiple organ
dysfunction during sepsis by blocking NET formation. Blood.
138:2702–2713. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Zhang Y, Zhang R and Han X: Disulfiram
inhibits inflammation and fibrosis in a rat unilateral ureteral
obstruction model by inhibiting gasdermin D cleavage and
pyroptosis. Inflamm Res. 70:543–552. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Teschke R: Top-ranking drugs out of 3312
drug-induced liver injury cases evaluated by the Roussel Uclaf
Causality Assessment Method. Expert Opin Drug Metab Toxicol.
14:1169–1187. 2018.PubMed/NCBI
|
|
136
|
Rathkey JK, Zhao J, Liu Z, Chen Y, Yang J,
Kondolf HC, Benson BL, Chirieleison SM, Huang AY, Dubyak GR, et al:
Chemical disruption of the pyroptotic pore-forming protein
gasdermin D inhibits inflammatory cell death and sepsis. Sci
Immunol. 3:eaat27382018. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Li N, Wang Y, Wang X, Sun N and Gong YH:
Pathway network of pyroptosis and its potential inhibitors in acute
kidney injury. Pharmacol Res. 175:1060332022. View Article : Google Scholar
|
|
138
|
Zhou LH, Zou H, Hao JY, Huang Y, Zhang JN,
Xu XH and Li J: Metformin inhibits ovarian granular cell pyroptosis
through the miR-670-3p/NOX2/ROS pathway. Aging (Albany NY).
15:4429–4443. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Han YC, Tang SQ, Liu YT, Li AM, Zhan M,
Yang M, Song N, Zhang W, Wu XQ, Peng CH, et al: AMPK agonist
alleviate renal tubulointerstitial fibrosis via activating
mitophagy in high fat and streptozotocin induced diabetic mice.
Cell Death Dis. 12:9252021. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Ling YH, Krishnan SM, Chan CT, Diep H,
Ferens D, Chin-Dusting J, Kemp-Harper BK, Samuel CS, Hewitson TD,
Latz E, et al: Anakinra reduces blood pressure and renal fibrosis
in one kidney/DOCA/salt-induced hypertension. Pharmacol Res.
116:77–86. 2017. View Article : Google Scholar
|
|
141
|
Cherney DZI, Lytvyn Y and McCullough PA:
Cardiovascular risk reduction in patients with chronic kidney
disease: Potential for targeting inflammation with canakinumab. J
Am Coll Cardiol. 71:2415–2418. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Li G, Liu C, Yang L, Feng L, Zhang S, An
J, Li J, Gao Y, Pan Z, Xu Y, et al: Syringaresinol protects against
diabetic nephropathy by inhibiting pyroptosis via NRF2-mediated
antioxidant pathway. Cell Biol Toxicol. 39:621–639. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Guo ZY, Wu X, Zhang SJ, Yang JH, Miao H
and Zhao YY: Poria cocos: Traditional uses, triterpenoid components
and their renoprotective pharmacology. Acta Pharmacol Sin.
46:836–851. 2025. View Article : Google Scholar
|
|
144
|
Miao H, Wang KE, Li P and Zhao YY:
Rhubarb: Traditional uses, phytochemistry, multiomics-based novel
pharmacological and toxicological mechanisms. Drug Des Devel Ther.
19:9457–9480. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Feng HY, Wang YQ, Yang J, Miao H, Zhao YY
and Li X: Anthraquinones from rheum officinale ameliorate renal
fibrosis in acute kidney injury and chronic kidney disease. Drug
Des Devel Ther. 19:5739–5760. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Ruan Z, Liu J, Liu W and Huang W: Qufeng
tongluo decoction may alleviate podocyte injury induced by high
glucose and hydrogen peroxide by regulating autophagy. Integr Med
Nephrol Androl. 11:e24–00023. 2024. View Article : Google Scholar
|
|
147
|
Ren H, Zhao C, Zhou L, Ke Q, Chen Y, Chen
Z, He J, Chen D, He X, Quan M, et al: Licochalcone B attenuates
pulmonary fibrosis via inhibiting ZBP1-dependent PANoptosis. J
Ethnopharmacol. 349:1199402025. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Lin SY, Chang CL, Liou KT, Kao YK, Wang
YH, Chang CC, Kuo TBJ, Huang HT, Yang CCH, Liaw CC and Shen YC: The
protective role of Achyranthes aspera extract against
cisplatin-induced nephrotoxicity by alleviating oxidative stress,
inflammation, and PANoptosis. J Ethnopharmacol. 319:1170972024.
View Article : Google Scholar
|
|
149
|
Xuan MY, Piao SG, Ding J, Nan QY, Piao MH,
Jiang YJ, Zheng HL, Jin JZ and Li C: Dapagliflozin alleviates renal
fibrosis by inhibiting RIP1-RIP3-MLKL-Mediated necroinflammation in
unilateral ureteral obstruction. Front Pharmacol. 12:7983812022.
View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Wang B, Yang LN, Yang LT, Liang Y, Guo F,
Fu P and Ma L: Fisetin ameliorates fibrotic kidney disease in mice
via inhibiting ACSL4-mediated tubular ferroptosis. Acta Pharmacol
Sin. 45:150–165. 2024. View Article : Google Scholar
|
|
151
|
Tong J, Lan XT, Zhang Z, Liu Y, Sun DY,
Wang XJ, Ou-Yang SX, Zhuang CL, Shen FM, Wang P and Li DJ:
Ferroptosis inhibitor liproxstatin-1 alleviates metabolic
dysfunction-associated fatty liver disease in mice: Potential
involvement of PANoptosis. Acta Pharmacol Sin. 44:1014–1028. 2023.
View Article : Google Scholar
|
|
152
|
Wang X, Xie X, Ni JY, Li JY, Sun XA, Xie
HY, Yang NH, Guo HJ, Lu L, Ning M, et al: USP11 promotes renal
tubular cell pyroptosis and fibrosis in UUO mice via inhibiting
KLF4 ubiquitin degradation. Acta Pharmacol Sin. 46:159–170. 2025.
View Article : Google Scholar
|
|
153
|
Shao Q, Sun C, Zhang Q, Liu J, Xia Y, Jin
B and Qian X: Macrophages regulates the transition of pericyte to
peritoneal fibrosis through the GSDMD/IL-1β axis. Int
Immunopharmacol. 101:1083232021. View Article : Google Scholar
|
|
154
|
Doke T, Huang S, Qiu C, Liu H, Guan Y, Hu
H, Ma Z, Wu J, Miao Z, Sheng X, et al: Transcriptome-wide
association analysis identifies DACH1 as a kidney disease risk gene
that contributes to fibrosis. J Clin Invest. 131:e1418012021.
View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Xu X, Tan X, Tampe B, Wilhelmi T, Hulshoff
MS, Saito S, Moser T, Kalluri R, Hasenfuss G, Zeisberg EM and
Zeisberg M: High-fidelity CRISPR/Cas9-based gene-specific
hydroxymethylation rescues gene expression and attenuates renal
fibrosis. Nat Commun. 9:35092018. View Article : Google Scholar
|
|
156
|
Jiang ZH, Tang YZ, Song HN, Yang M, Li B
and Ni CL: miRNA-342 suppresses renal interstitial fibrosis in
diabetic nephropathy by targeting SOX6. Int J Mol Med. 45:45–52.
2020.
|
|
157
|
Yang J, Liao D, He L and Fan W: Puerarin
raises exosomal miR-342 - 3p by inhibiting lncRNA NEAT1 in
umbilical cord mesenchymal stem cells to alleviate renal tubular
epithelial cell pyroptosis in chronic renal failure. Naunyn
Schmiedebergs Arch Pharmacol. 398:14219–14238. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Tang L, Li K, Zhang Y, Li H, Li A, Xu Y
and Wei B: Quercetin liposomes ameliorate streptozotocin-induced
diabetic nephropathy in diabetic rats. Sci Rep. 10:24402020.
View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Li R, Li Y, Zhang J, Liu Q, Wu T, Zhou J,
Huang H, Tang Q, Huang C, Huang Y, et al: Targeted delivery of
celastrol to renal interstitial myofibroblasts using
fibronectin-binding liposomes attenuates renal fibrosis and reduces
systemic toxicity. J Control Release. 320:32–44. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Lin J, Dong L, Liu YM, Hu Y, Jiang C, Liu
K, Liu L, Song YH, Sun M, Xiang XC, et al: Nickle-cobalt alloy
nanocrystals inhibit activation of inflammasomes. Natl Sci Rev.
10:nwad1792023. View Article : Google Scholar : PubMed/NCBI
|