Introduction
Type 1 diabetes (T1D) is a chronic autoimmune
disorder characterized by the destruction of pancreatic β-cells.
The pathogenesis of T1D is considered to result from a synergistic
interaction among genetic, environmental and immune factors
(1). Currently, insulin
replacement therapy remains the primary treatment for T1D; however,
achieving optimal glycemic control continues to pose significant
challenges (2). Mesenchymal stem
cells (MSCs) are known to attenuate inflammation and modulate the
immune system, although the precise mechanisms underlying
MSC-mediated immunomodulation have yet to be fully elucidated. Due
to their multilineage differentiation capacity and immunomodulatory
properties, MSCs have been extensively researched for treating
degenerative and autoimmune diseases, including T1D. An increasing
body of preclinical and clinical evidence suggests that MSCs can
enhance islet β-cell function in individuals with T1D (3-5).
However, a major caveat is that MSCs from pre-clinical models as
well as T1D patients show gross defects in a number of these
properties and lack the ability to promote protection from T1D,
raising the concern that unaltered autologous MSCs may not have
therapeutic value (6,7). The potential explanation for the
limited efficacy of MSCs therapy in T1D may lie in the nonspecific
nature of MSCs' immunoregulatory functions, coupled with the unique
characteristics of the immune dysregulation observed in this
disease. Consequently, the mere reinfusion of MSCs fails to provide
targeted therapeutic benefits for T1D.
Sialic acid-binding immunoglobulin-like lectins
(Siglecs) constitute a family of glycan-recognizing proteins that
are part of the immunoglobulin superfamily. The Siglec family has
the potential to restore immune tolerance in the context of
autoimmune diseases. A number of Siglecs act as inhibitory
receptors, attenuating activation signals in various immune cells
by binding to sialic acid ligands, which serve as markers of self.
Previous research has demonstrated that Siglec-7 is expressed on
β-cells and is downregulated in both type 1 and type 2 diabetes, as
well as in infiltrating activated immune cells (8). Additionally, Siglec-10 is expressed
on tissue-infiltrating T cells and has been shown to confer
protection against T1D in murine models (9). The research group led by Guo
(10) identified a novel subset
of SIGLEC-1+ monocytes, which may serve as a significant
biomarker for early diagnosis, assessment of disease activity and
monitoring of therapeutic efficacy in T1D.
Siglec-15 is a Siglec family protein that acts as a
significant immune suppressor by inhibiting T-cell activation and
fostering immunosuppressive myeloid cells, thereby suppressing
antigen-specific T cell responses (7,11). Its critical role in tumor immune
evasion is well-documented. Seminal work by Wang et al
(12) established that Siglec-15
directly inhibits tumor-specific CD8+ T cells and its
blockade enhances antitumor immunity. This concept is further
supported by findings from Hu et al (13), which associated high Siglec-15
expression with a non-inflamed, therapy-resistant microenvironment
in bladder cancer. Collectively, these studies indicate that
Siglec-15 suppresses T-cell responses likely via an unidentified
receptor, playing a pivotal role in maintaining immune tolerance.
This function is of particular interest given that disrupted
tolerance is a hallmark of autoimmune pathologies. Nonetheless, the
specific role of Siglec-15 in immune homeostasis and its
involvement in autoimmune diseases, such as T1D, remain poorly
characterized. Our prior research identified that T-cell
immunoglobulin mucin-1 (Tim-1) and T-cell immunoglobulin mucin-4
(Tim-4) are integral to the pathogenesis of T1D (14). Notably, the V-set Ig-like domain
of Siglec-15 exhibits considerable sequence homology with those of
Tim-1 and Tim-4 (15). This
observation implies a potential association of Siglec-15 with the
development of T1D.
The present study examined the presence of Siglec-15
in the serum of patients with T1D and investigated the factors
influencing Siglec-15 expression. Additionally, it explored the
negative regulatory properties of MSCs as a potential tool for
modulating the immune system in T1D. The present study developed
SIGLEC15 gene-transfected MSCs, designated as C3H10/SIGLEC15, which
are murine MSC C3H10 T1/2 cells that stably express SIGLEC15. An
enhanced understanding of the characteristics of SIGLEC15 in C3H10
cells may offer novel insights into stem cell therapy for T1D.
Materials and methods
Patients and controls
Patients were recruited from the Second Affiliated
Hospital of Soochow University. There were 34 patients with newly
diagnosed T1D (diagnosed with the criteria of American Diabetes
Association) (16) and 21
healthy volunteers who were recruited from hospital staff.
Participants were excluded if they had one of the following
conditions: Acute or chronic inflammatory diseases, other
autoimmune diseases, infectious diseases, cancer, or if they were
on antibiotic treatment. Biochemical and clinical data were
obtained from patient medical records. Blood samples were collected
after overnight fasting from patients between March 2021 and March
2024. Blood samples underwent centrifugation at 4°C and 1,800 x g
for a duration of 10 min, after which the resulting cell-free serum
was preserved at -80°C until analysis. Serum concentrations of
Siglec-15 were quantified using an enzyme-linked immunosorbent
assay (ELISA) kit (cat. no. EKN53372; Biomatik). Clinical and
biochemical characteristics of the patients are given in Table I.
 | Table IClinical and biochemical
characteristics of the patients. |
Table I
Clinical and biochemical
characteristics of the patients.
| Characteristic | T1D group | Control group | P-value |
|---|
| Number of
participants | 34 | 21 | |
| Age, years | 33.51±7.6 | 36.48±9.7 | 0.181 |
| Sex.
male/female | 23/11 | 13/8 | 0.189 |
| TC, mmol/l | 4.31±1.09 | 4.00±0.93 | 0.287 |
| TG, mmol/l | 1.29±0.94 | 0.78±0.43 | 0.026 |
| LDL-C, mmol/l | 2.78±0.85 | 2.38±0.76 | 0.082 |
| Uric acid,
µmol/l | 325.18±144.98 | 249.52±82.47 | 0.034 |
| BMI,
kg/m2 | 21.67±3.58 | 20.80±2.15 | 0.321 |
| Diabetes duration,
weeks | 1.45±0.941 | - | - |
| HbA1c, % | 11.06±2.36 | - | - |
| FCP, ng/ml | 0.57±0.46 | - | - |
| 2hCP, ng/ml | 0.97±1.21 | - | - |
| Positive-anti ICA,
(%) | 23 (41.8) | - | - |
| Positive-anti GADA,
(%) | 29 (52.7) | - | - |
| Positive-anti IAA,
(%) | 4 (7.3) | - | - |
| Positive-anti IA2A,
(%) | 13 (23.6) | - | - |
| Positive-anti
ZnT8A, (%) | 0 (0) | - | - |
| Active smoking, n
(%) | 6 (10.9) | - | - |
| Alcohol intake, n
(%) | 1 (1.8) | - | - |
| Siglec15 levels
(ng/ml) | 117.6
(40.69-207.89) | 222
(114.26-649.77) | <0.001 |
Animals
Female non-obese diabetic (NOD) mice, aged 6-8 weeks
and weighing 18-22 g, were purchased from Cavens Biogle and
maintained under pathogen-free conditions at the Model Animal
Research Center of Soochow University. Mice were housed in a room
maintained at 22°C and 50-60% humidity under a 12-h light/dark
cycle (lights off from 08:00 to 20:00). Food and water were
replaced every other day. A total of 18 mice [PBS group (n=6),
Control-MSCs group (n=6), Siglec15-MSCs group (n=6)] were used in
the present experiments. Mice were randomly allocated to the PBS,
Siglec15-MSCs group (overexpression of Siglec-15 in MSC) or
Control-MSCs so that blood glucose means at the start of the
experiment were even. Blinding was not carried out in any of the
experiments. All animal experiments were approved by the
Institutional Animals Ethics Committee at Soochow University,
Suzhou, China (approval no. 202412A0289).
Cell culture
293T cells and the mouse MSC line, C3H10 T1/2, were
originally obtained from the Cell Resource Center, Peking Union
Medical College. Cell lines C3H10 T1/2 were cultured in minimum
essential medium (MEM; Gibco; Thermo Fisher Scientific, Inc.) with
10% fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.)
and 1% Non-Essential Amino Acids (NEAA; Jiangsu KeyGen Biotech Co.,
Ltd.). All cells were incubated at 37°C in a humidified atmosphere
containing 5% CO2.
Reagents
The following antibodies were purchased from
BioLegend, Inc.: APC anti-mouse Perforin Antibody (cat. no.
154404), Brilliant Violet 421 anti-mouse CD11b Antibody (cat. no.
101251), PE anti-mouse CD11b Antibody (cat. no. 101207), APC
anti-mouse CD44 Antibody (cat. no. 103012), FITC anti-mouse CD4
Antibody (cat. no. 100405), Brilliant Violet 421 anti-mouse CD62L
Antibody (cat. no. 104436), APC/Cyanine7 anti-mouse CD8 Antibody
(cat. no. 100714), Brilliant Violet 421 anti-mouse CD8a Antibody
(cat. no. 100753), FITC anti-mouse CD8a Antibody (cat. no. 100705),
PE anti-mouse CD8a Antibody (cat. no. 100707), PE anti-mouse FOXP3
Antibody (cat. no. 320008), FITC anti-mouse IFN-γ Antibody (cat.
no. 505806), Brilliant Violet 421 anti-mouse IL-17A Antibody (cat.
no. 506926), APC anti-mouse CD4 Antibody (cat. no. 100411). The
following antibodies were purchased from Abcam: FITC anti-mouse
IL-4 Antibody (cat. no. ab186716), FITC anti-mouse CD3 Antibody
(cat. no. ab34722), Alexa Fluor 647 Anti-Insulin antibody (cat. no.
ab309368), Alexa Fluor 488 Anti-Glucagon antibody (cat. no.
ab307340). The human Siglec15 ELISA Kit was purchased from Biomatik
(cat. no. EKN53372).
Establishment of a genetically engineered
cell line
The full length of mouse SIGLEC15 cDNA (https://www.ncbi.nlm.nih.gov/nuccore/NM_001101038.2)
was subcloned into lentiviral vector pSLenti-Puro (Shanghai Obio
Technology Corp., Ltd.). Using a second-generation packaging
system, lentiviral particles were generated by co-transfecting 293T
cells (Cell Resource Center, Peking Union Medical College) with 2.0
µg of the recombinant plasmid, 1.5 µg of the
packaging plasmid pHIT60 (Shanghai Obio Technology Corp., Ltd.),
and 0.5 µg of the envelope plasmid pHIT456 (Shanghai Obio
Technology Corp., Ltd.) at a 4:3:1 ratio using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.). After 48 h of incubation at 37°C, viral
supernatants were collected. C3H10 T1/2 cells were then transduced
with the viral supernatant at a multiplicity of infection (MOI) of
20 in the presence of 8 µg/ml polybrene (cat. no. C0351;
Beyotime Biotechnology). Following 24 h of transduction, the
virus-containing medium was replaced with fresh complete medium. At
48 h post-transduction, cells were subjected to selection with 3.5
µg/ml puromycin (cat. no. ST551; Beyotime Biotechnology) for
1 week to establish stable polyclonal populations. The puromycin
concentration was subsequently reduced to 1 µg/ml for
maintenance. The resulting puromycin-resistant C3H10 T1/2 cell line
stably expressing SIGLEC15 was designated Siglec15-MSCs.
Treatment of NOD mice with engineered
MSCs
Pre-diabetic (10-12-week-old) NOD mice from multiple
cages were pooled and randomly allocated into three treatment
groups. Each mouse received two intravenous injections (on day 0
and day 3) at the following doses per injection: The PBS group
received 200 µl of PBS alone; the Control-MSCs group
received 5×105 control MSCs resuspended in 200 µl
of PBS; and the Siglec15-MSCs group received 5×105
Siglec15-MSCs in 200 µl of PBS. Three groups of mice were
monitored for hyperglycemia by testing for blood glucose levels
twice a week. Blood glucose was measured from the tail vein in the
ad libitum-fed state, with 5-10 µl of blood collected
for each measurement. Blood glucose was assayed using Yuwell 580
(Yuwell). NOD mice with glucose levels <5.6 mmol/l were
considered pre-diabetic. Diabetes was defined as two consecutive
readings above 13.9 mmol/l (17). Cohorts of mice were euthanized at
week 8 to determine the degree of insulitis, immune cell phenotype
and diabetes incidence. Mice were sacrificed by cervical
dislocation after being deeply anesthetized with 5% isoflurane
inhalation and loss of respiration and reflexes was confirmed
before tissue collection.
Flow cytometry
The surface epitopes of the cells were analyzed by
flow cytometry using a series of anti-mouse monoclonal antibodies
(mAbs; eBioscience; Thermo Fisher Scientific, Inc.). For the direct
immunofluorescence assay, cells were stained with fluorescein
isothiocyanate (FITC)- or phycoerythrin (PE)-conjugated mAbs for 30
min at 4°C in the dark. For intracellular staining, the cells were
fixed with 2% paraformaldehyde for 30 min at room temperature,
permeabilized with 0.5% saponin and stained with FITC- or
PE-conjugated rat anti-mouse mAbs for 30 min at 4°C in the dark.
Rat isotype immunoglobulin (Ig)-PE or Ig-FITC served as negative
controls. A BD FACSCanto II flow cytometer (BD Biosciences) was
used for data acquisition, and the data were analyzed using FlowJo
v10.10.0 (BD Biosciences).
Histochemistry
Mouse pancreas samples were dissected, fixed in 4%
paraformaldehyde in PBS at 4°C and embedded in paraffin. The
sections (2 µm thick) were deparaffinized and stained with
hematoxylin and eosin (H&E) for general morphological
assessment. The deparaffinized sections were also treated with
citrate buffer (TRS; pH 6.0; Wuhan Servicebio Technology Co., Ltd.)
using a high pressure oven for 30 sec at 121°C and 10 sec at 90°C.
The sections were blocked for 30 min at room temperature using a
blocking buffer consisting of 3% BSA. Islets were stained for
insulin (cat. no. I2018; 1:100; R&D Systems) and glucagon (cat.
no. ab10988; 1:100; Abcam). Primary antibody incubation was carried
out overnight at 4°C. Cell nuclei were counterstained with DAPI.
The stained sections were analyzed by a FLUOVIEW FV3000 laser
scanning confocal microscope (Nikon Corporation) for
high-resolution micro-imaging.
Reverse transcription-quantitative (RT-q)
PCR
Total RNA was extracted from MSCs, Control-MSCs, and
Siglec15-MSCs cultured to 70-90% confluence using
TRIzol® Reagent (cat. no. 15596018; Thermo Fisher
Scientific, Inc.) A total of 3 µg of RNA was used for
reverse transcription using the PrimeScript II 1st Strand cDNA
Synthesis Kit (cat. no. 6210A; Takara Biotechnology Co., Ltd.)
according to the manufacturer's protocol. The cDNA was further used
to conduct qPCR analysis per the BeyoFast SYBR Green qPCR Mix (cat.
no. D7260; Beyotime Biotechnology). The thermocycling protocol was:
Initial denaturation at 95°C for 2 min, followed by 40 cycles of
95°C for 15 sec, 55°C for 20 sec and 72°C for 30 sec. A melting
curve analysis was performed from 60°C to 95°C to verify
amplification specificity. Gene expression levels were calculated
using the 2-ΔΔCq method (18). All reactions were performed in
triplicate. The according primer pairs for the target gene and
housekeeping gene were as follows: Siglec-15 (Forward: TGC TGC TGC
TTG GCA TTC TGG, Reverse: CCT GAG CCT GAG ACC GTG GAG) and β-actin
(Forward: AGG TCA TCA CTA TTG GCA ACG AGC, Reverse: AGA GGT CTT TAC
GGA TGT CAA CGTC).
Statistical analysis
Data are expressed as the mean ± SEM. Differences
between the indicated two groups were analyzed by unpaired
Student's t-test. The data of RT-qPCR and flow cytometry were
evaluated using one-way ANOVA followed by Tukey's test for multiple
group comparisons. For analysis of diabetes-free rate, the log-rank
test was performed between the indicated two groups. Correlations
were evaluated using Spearman's correlation test and Mann-Whitney U
test. Blood glucose levels were evaluated using mixed ANOVA
followed by Sidak's test for multiple group comparisons. Fisher's
exact test was used to compare the frequency of insulitis grades.
All statistical analyses were performed using SPSS (version 27.0;
IBM Corp.) and GraphPad Prism 10 (Dotmatics). P<0.05 was
considered to indicate a statistically significant difference.
Results
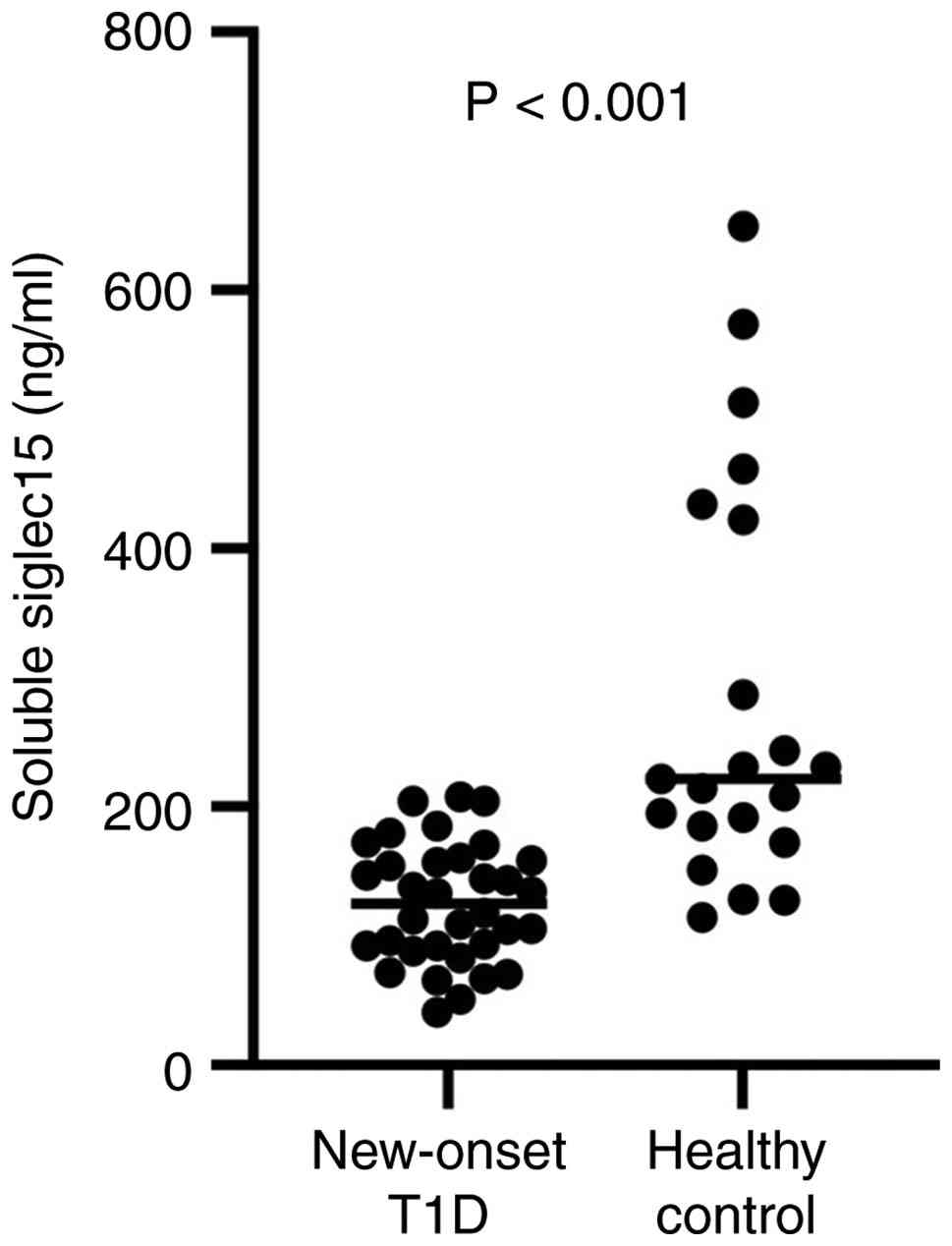
Soluble Siglec-15 in new-onset T1D and
healthy controls
The present study enrolled a total of 56
participants, categorized into a new-onset T1D group consisting of
34 individuals and a healthy control group comprising 21
individuals. The clinical characteristics of the present study
population are detailed in Table
I. Soluble Siglec-15 was detectable in both the T1D cohort and
the healthy controls. Notably, serum levels of Siglec-15 were
markedly reduced in the new-onset T1D patients [117.6
(40.69-207.89) ng/ml] compared with the healthy control group [222
(114.26-649.77) ng/ml], with a statistical significance of
P<0.001 (Fig. 1). However,
within the new-onset T1D group, no significant correlations were
observed between the titers of soluble Siglec-15 and various
clinical parameters, including fasting C-peptide, random blood
glucose levels, age, disease duration, HbA1c, autoantibodies
(AAbs), creatinine (Cr), blood urea nitrogen (BUN), uric acid (UA),
alanine aminotransferase (ALT), aspartate aminotransferase (AST),
total cholesterol (TC), triglycerides (TG), low-density lipoprotein
(LDL) and high-density lipoprotein (HDL; Tables SI and SII).
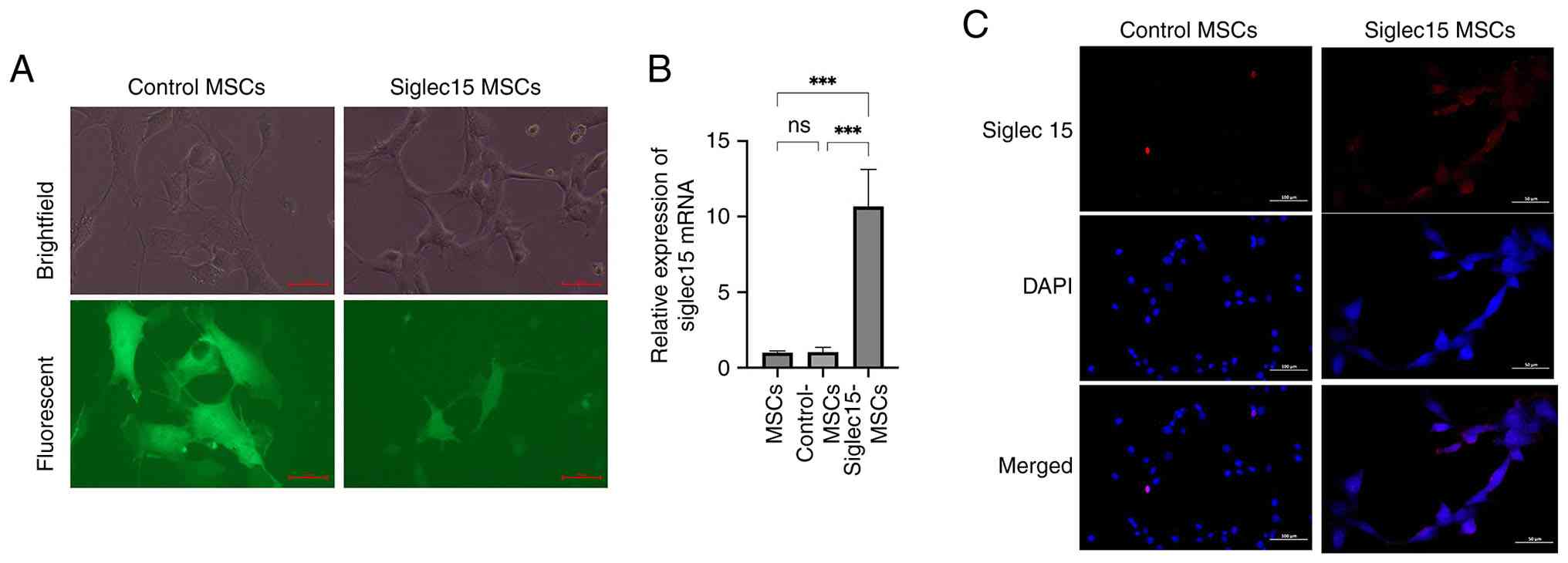
Expression of SIGLEC15 gene in C3H10 T1/2
cell line
To generate SIGLEC15-expressing MSCs, C3H10 T1/2
cells were transduced with either control or SIGLEC15 lentivirus
particles, both incorporating a GFP reporter. The efficiency of
transduction, as indicated by GFP expression, was verified through
fluorescence microscopy (Fig.
2A). RT-qPCR analysis confirmed that SIGLEC15 transcript levels
were markedly elevated in Siglec15-MSCs compared with Control-MSCs
(Fig. 2B). The immunofluorescent
staining results demonstrated a similar trend (Fig. 2C), indicating that C3H10 T1/2
cells can be effectively engineered to express SIGLEC15.
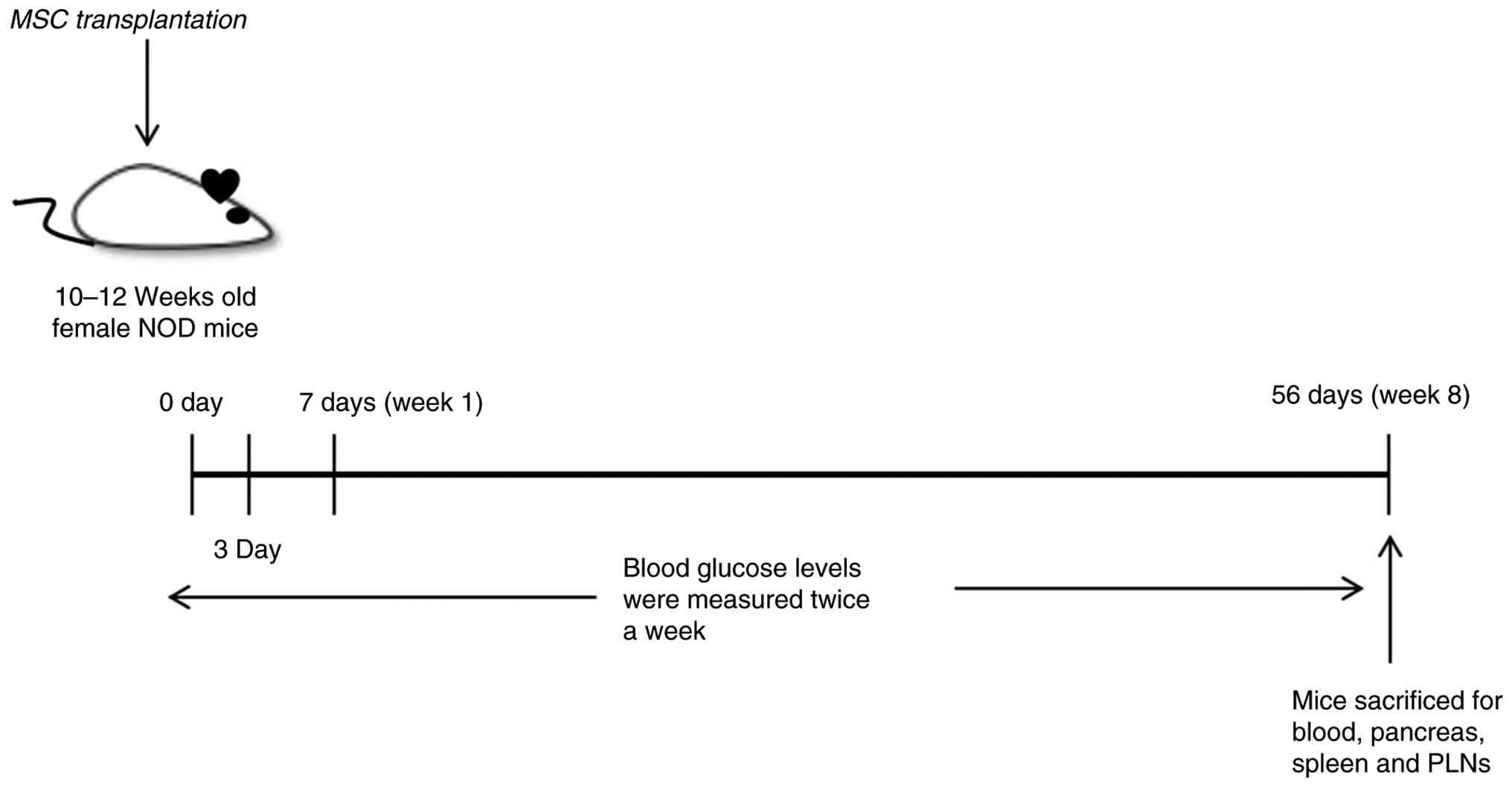
Siglec15-MSCs treatment at pre-diabetic
stage results in diminished insulitis and prevention of T1D in NOD
mice
To evaluate the impact of Siglec15-MSCs treatment on
insulitis and the incidence of T1D, pre-diabetic female NOD mice
were administered Siglec15-MSCs, Control-MSCs, or PBS twice during
the first week (Fig. 3). The
cohorts were subsequently monitored for hyperglycemia by measuring
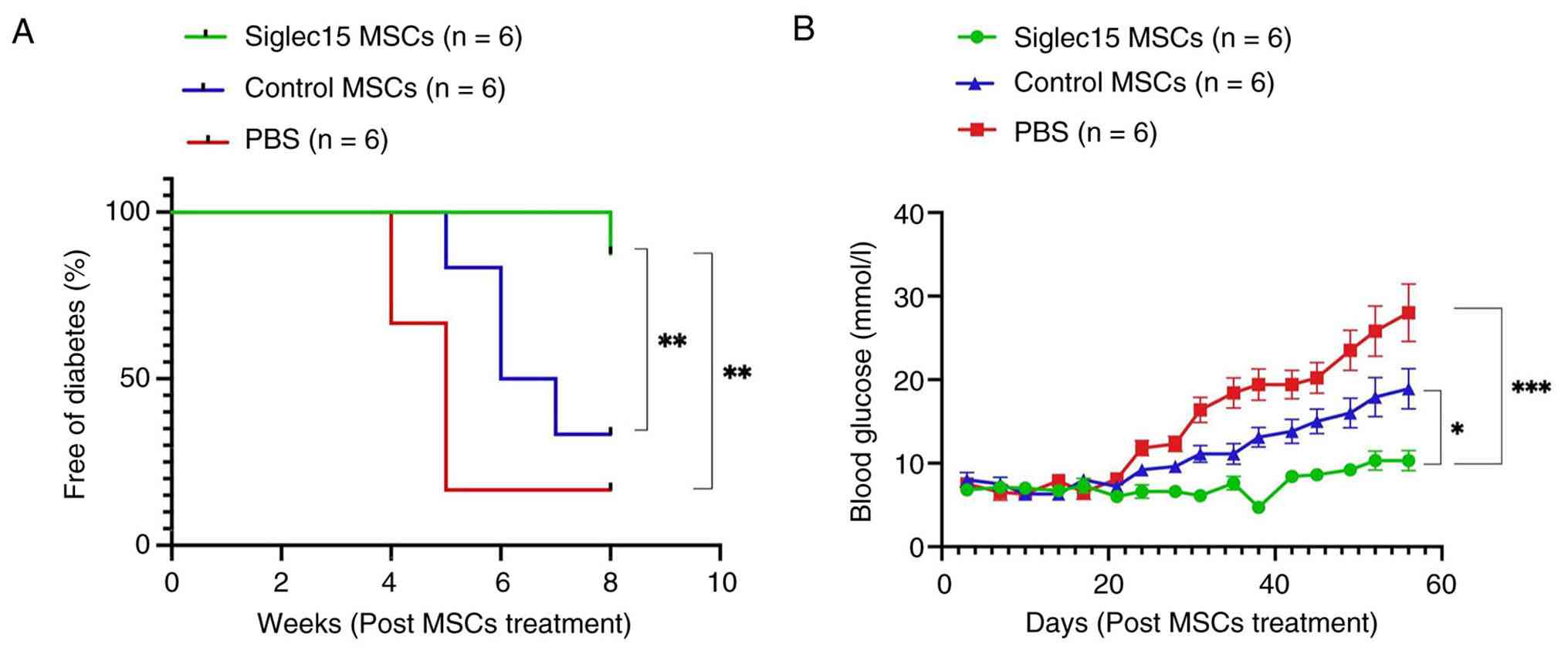
blood glucose levels biweekly. As illustrated in Fig. 4A, administration of Siglec15-MSCs
resulted in a reduced incidence of overt diabetes, with 83% of the
Siglec15-MSCs-treated mice maintaining euglycemia for at least 8
weeks post-treatment. By contrast, more than 17% of the untreated
(PBS) group and 33% of the Control-MSCs-treated group developed
hyperglycemia during this period. Furthermore, Siglec15-MSCs
administration led to markedly lower mean blood glucose levels
compared with the untreated and Control-MSCs-treated group
(Fig. 4B).
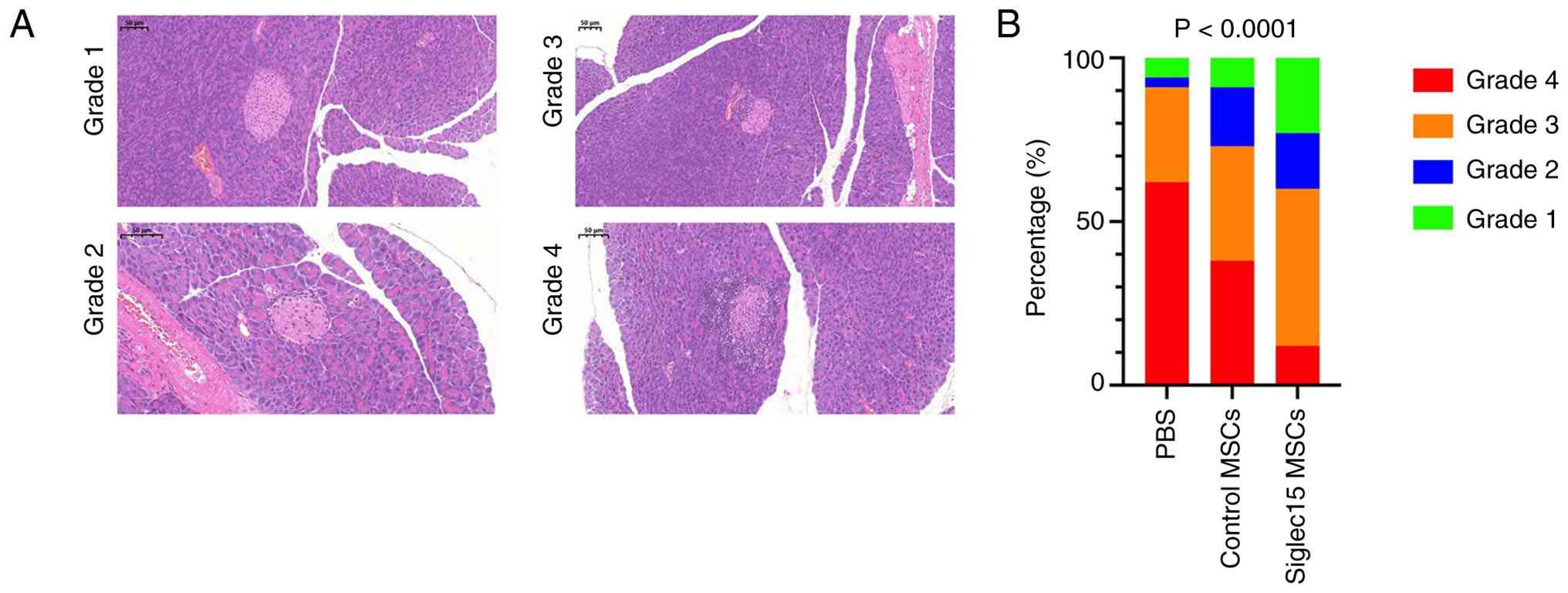
The degree of insulitis was assessed after 49 days
by subjecting pancreatic sections to H&E staining and grading
for insulitis severity. As illustrated in Fig. 5A, the grading scale reflects the
severity of islet lymphocyte infiltration, with higher grades
indicating increased infiltration (1=peri-islet infiltration
(<5%), 2=5-25% islet infiltration, 3=25-50% islet infiltration
and 4=>50% islet infiltration). Fig. 5B demonstrated that a total of 100
areas (25 islet areas per pancreas) were analyzed across three or
more intermittent sections for each experimental group. Statistical
significance was evaluated using Fisher's exact test to compare the
relative frequencies of islets with insulitis grades ≤2 and ≥3
between the groups. Notably, NOD mice treated with Siglec15-MSCs
exhibited markedly reduced insulitis compared with those treated
with control-MSCs or PBS. These findings suggested that a single
administration of Siglec15-MSCs can preserve a substantial number
of intact islets or islets with less severe insulitis, thereby
preventing hyperglycemia for a considerable period.
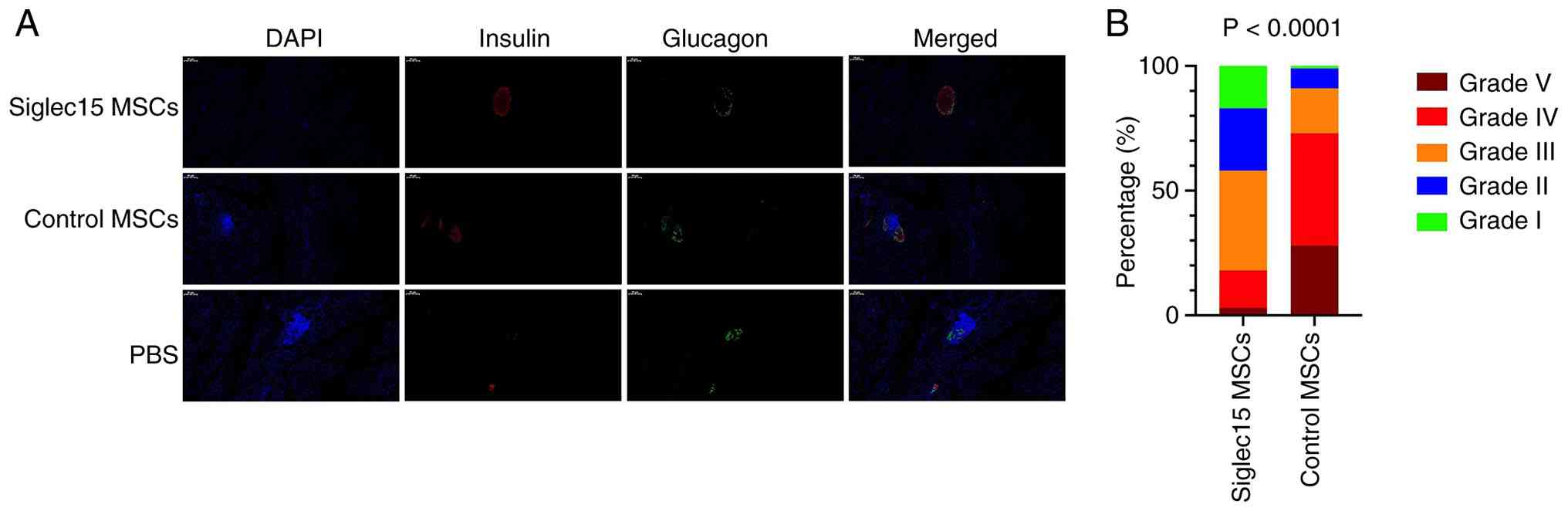
Preservation of insulin-secreting islets
by Siglec15-MSCs treatment at the pre-diabetic stage
To assess the effect of Siglec15-MSCs treatment on
the abundance of functional islets, the frequency of
insulin-positive islets was measured in mice treated with
Siglec15-MSCs, control-MSCs, or PBS, seven weeks post-treatment. As
illustrated in Fig. 6A, the
majority of islets in the Siglec15-MSCs group exhibited mild
perinsulitis, while maintaining well-preserved insulin and glucagon
staining. By contrast, the islets in the control and PBS groups
demonstrated poorly preserved insulin and glucagon staining. The
characteristics of the islets were graded on a scale from I to V,
based on the extent of immune cell infiltration (determined by DAPI
staining) and insulin presence (positive or negative): Grade I
(<5% infiltration/insulin+), Grade II (5-25%
infiltration/insulin+), Grade III (25-50% infiltration/insulin+),
Grade IV (50-100% infiltration/insulin+) and Grade V (50-100%
infiltration/insulin-). A total of 40 areas (10 islet areas per
pancreas) from three or more intermittent sections were analyzed
for each group. The P-value was calculated using Fisher's exact
test to compare the relative numbers of islets with insulitis
grades ≤III and ≥IV between the Siglec15-MSCs and Control MSCs
groups (Fig. 6B). These findings
suggested that the prevention of T1D in mice treated with
Siglec15-MSCs is associated with the preservation of
insulin-producing islet mass and the restoration of insulin
expression in non-functional β-cells.
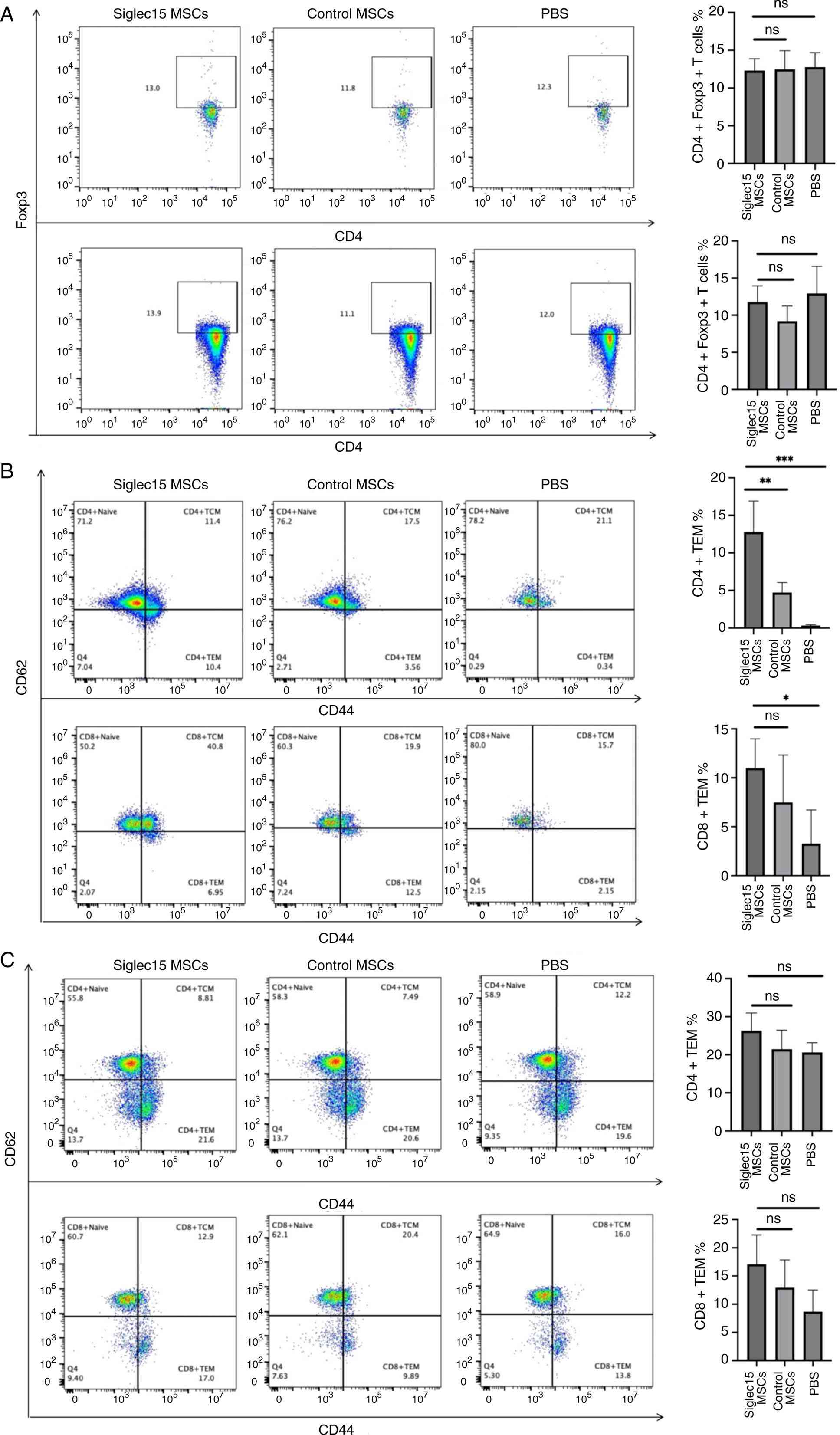
Siglec15-MSCs ameliorate autoimmunity via
memory T cells
The present study investigated the alterations in
memory T-cell and regulatory T-cell (Treg) populations within the
pancreas-draining lymph nodes and spleen following treatment with
Siglec15-MSCs. As depicted in Fig.
7A, there was no significant difference in the frequency of
Treg cells in the pancreas-draining lymph nodes and spleen across
the three experimental groups. Flow cytometric analysis
demonstrated that Siglec15-MSCs therapy markedly increased the
percentages of CD4+ effector memory T cells (TEMs)
(P<0.001) in the draining lymph nodes compared with both the
control and PBS groups. Additionally, Siglec15-MSCs therapy
resulted in a significant increase in the percentages of
CD8+ TEM cells (P<0.05) relative to the PBS group;
however, no significant difference was observed between the
Siglec15-MSCs and control groups (Fig. 7B). The frequencies of
CD4+ and CD8+ memory T cells in the spleen
did not differ markedly among the Siglec15-MSCs, Control-MSCs and
PBS-treated mice (Fig. 7C).
Discussion
Siglec15 is a type I transmembrane protein
characterized by two immunoglobulin-like domains and is highly
conserved across vertebrate species, indicating its significant and
fundamental role in immune regulation (19). In contrast to a number of other
Siglecs, which contain immunoreceptor tyrosine-based inhibitory
motif and transmit inhibitory signals, Siglec15 interacts with
activating adaptor proteins such as DAP12, thereby initiating
activating immune signals (15).
Wang et al (12)
identified Siglec15 as a critical immune suppressor within the
tumor microenvironment, where it is upregulated on human cancer
cells and tumor-associated macrophages. Due to its distinct
immunosuppressive properties and non-redundant function compared
with PD-1/PD-L1, Siglec15 has emerged as a promising target for
cancer immunotherapy. Nevertheless, the role of Siglec15 in
autoimmune diseases has been largely overlooked. In this study, we
provide a comprehensive elucidation of the multifaceted role of
Siglec15 and MSCs engineered to express SIGLEC15 in the modulation
of T1D.
The present study initially demonstrated that
soluble Siglec15 serum levels are markedly reduced in patients with
new-onset T1D compared with healthy controls, indicating a
potential dysregulation of Siglec15-mediated immune modulation
during the early stages of the disease. Importantly, soluble
Siglec15 levels did not exhibit an association with traditional
clinical parameters such as C-peptide, HbA1c, or autoantibody
titers. This suggested that Siglec15 may operate independently of,
or upstream from, overt β-cell dysfunction and glycemic control.
This observation is consistent with existing literature that
highlights the critical roles of Siglec family members in immune
tolerance and homeostasis (20).
Nonetheless, the proposed causal relationship requires further
validation. The observed reduction in Siglec15 levels in patients
with new-onset T1D prompts consideration of two potential
possibilities. One possibility is that decreased secretion of
Siglec15 by patient cells contributes directly to disease
pathogenesis. In this scenario, diminished Siglec15 secretion leads
to impaired immunoregulatory capacity, which, in combination with
other predisposing factors, results in autoimmune destruction of
pancreatic islets. Alternatively, the reduction in circulating
Siglec15 may be consequential rather than causal. During the
progression of T1D, as autoreactive T cells attack islet β-cells,
Siglec15 may be continuously consumed in an effort to suppress the
ongoing autoimmune response. In this context, Siglec15 could play
an immunosuppressive role and its progressive depletion may reflect
the intensity and persistence of the autoimmune attack. When
Siglec15 levels fall below the threshold required to maintain
immune tolerance equilibrium, the clinical onset of T1D may occur.
Regardless of whether the reduction in Siglec15 is a cause or a
consequence of the autoimmune process, this early immunological
phase may represent a critical therapeutic window for interventions
aimed at restoring immune balance and preserving islet
function.
Moreover, it has been demonstrated that MSCs can
mitigate tissue damage and enhance function following lung injury,
kidney disease and diabetes (21). The inherent ability of MSCs to
migrate within the body has also been documented (22,23). T1D is an autoimmune disorder
characterized by the immune-mediated destruction of pancreatic
β-cells. Studies have explored the use of MSCs as a potential
therapeutic approach for T1D; however, the outcomes have often been
inconsistent and suboptimal (24,25). The heterogeneity observed in
clinical outcomes may be attributed to the diversity of MSCs
sources used across studies. MSCs can be readily isolated from
several tissues, such as bone marrow, adipose tissue and umbilical
cord, each of which exhibits distinct biological properties. In
clinical practice, bone marrow-derived MSCs (BM-MSCs) and umbilical
cord-derived MSCs (UC-MSCs) are among the most frequently employed
(26). A comparative study by
Zhang et al (3) in NOD
mice demonstrated that both UC-MSCs and BM-MSCs yielded comparable
therapeutic effects, including reduced blood glucose levels,
preserved β-cell function and similar immunomodulatory outcomes,
such as mitigating insulitis, decreasing Th17 cell populations and
elevating regulatory T cell numbers. Thus, given that no single MSC
source has shown definitive superiority for T1D treatment, research
efforts are increasingly directed toward engineering MSCs to
enhance their therapeutic potential for this specific condition
(27-29).
The present study demonstrated that engineering MSCs
to overexpress SIGLEC15 markedly improved their therapeutic potency
in the NOD mouse model. Administration of Siglec15-MSCs during the
pre-diabetic stage notably delayed the onset of diabetes and
preserved insulin-positive islets, while also reducing the severity
of insulitis, surpassing the performance of control MSCs and PBS
groups. These findings supported the hypothesis that engineering
MSCs to express immunomodulatory molecules can address the inherent
deficiencies observed in syngeneic MSCs derived from diabetic
models (29,30). This effect was associated with
the selective expansion of CD4+ effector memory T cells
in the pancreas-draining lymph nodes.
Effector memory T cells are integral to the
pathogenesis of T1D, an autoimmune disorder characterized by the T
cell-mediated destruction of pancreatic β-cells. Various
immunotherapeutic approaches for T1D have been shown to modulate
both the frequency and phenotype of memory T cells. Alefacept, for
instance, selectively depletes CD2high TEMs and central
memory T cells (TCMs), while preserving naïve and Regulatory T
cells (Tregs). Additionally, it promotes the expansion of a subset
of exhausted-like
TIGIT+PD1+CD4+TEMs, which exhibit
reduced production of pro-inflammatory cytokines. This alteration
in TEM phenotype is associated with diminished β-cell destruction
and clinical improvement in individuals with new-onset T1D
(31). Furthermore, stem cell
educator therapy entails the ex vivo exposure of a patient's
lymphocytes to cord blood-derived multipotent stem cells (CB-SCs),
which function to 'educate' the immune cells. This intervention
results in a decreased proportion of TEMs in peripheral blood,
enhanced CCR7 expression and the conversion of TEMs into TCMs or
naïve T cells, thereby restoring immune homeostasis (32). Tolerogenic dendritic cells or
ethylene carbodiimide-fixed splenocytes presenting islet antigens
facilitate antigen-specific anergy or functional inactivation in
pathogenic effector/memory T cells. These therapeutic approaches
reduce the frequency and activity of TEMs, promote the expansion of
regulatory T cells and restore immune tolerance to β-cell antigens,
thereby preventing or delaying the progression of T1D (33,34).
In contrast to these treatments, the present study
revealed no alteration in the proportion of TEMs in peripheral
sites, such as the spleen. However, a significant increase in
CD4+ TEMs was specifically noted in the pancreatic
draining lymph nodes. The pancreatic lymph nodes (pLNs) serve as
critical sites for the initiation and regulation of autoimmune
responses against pancreatic β-cells in T1D. Unlike peripheral
blood or distal lymphoid tissues, pLNs are strategically positioned
to sample pancreatic antigens and coordinate local immune
responses. Consequently, alterations within the pLNs are of
paramount significance. A previous study identified significant
alterations in CD4+TEMs within pLNs at various stages of
T1D development. In both pre-T1D and T1D individuals, the frequency
of CD4+TEMs was markedly reduced in pLNs compared with
non-diabetic controls (35).
Through modulation of the immune environment within the pLNs,
treatment with Siglec15-MSCs reversed the alterations in
CD4+ TEMs in NOD mice, thereby preventing the
progression of T1D.
However, the molecular mechanism by which Siglec15
interacts with CD4+TEMs remains unclear and its role in
autoimmunity has not been explored, as previous research has
primarily focused on types of cancer. A prior study has
demonstrated that Siglec15 may act as a central glyco-immune
checkpoint by promoting osteoclastogenesis and suppressing T cell
immunity through sialic acid-mediated interactions, thereby
facilitating breast cancer bone metastasis and the spread to
secondary organs (36).
Furthermore, Siglec15 can suppress T cell activity by binding to
sialylated glycans on CD11b via its V-set domain and this
interaction can be inhibited by antibodies targeting its
glycan-binding site (37). The
Kyn/AhR/Siglec15 axis has been identified as a novel immune escape
mechanism in head and neck squamous cell carcinoma, wherein
Siglec15 functions as an immunosuppressive checkpoint that inhibits
CD8+T cell activity, independent of the PD-1/PD-L1
pathway. Notably, these findings offer valuable insights for future
research into the specific role of Siglec15 in T1D (38).
Recent advances in the immunological understanding
of T1D are facilitating the development of innovative therapeutic
strategies that hold the potential for achieving a cure with an
acceptable safety profile. However, current monotherapies are
constrained by their limited efficacy and transient effects.
Combination immunotherapy presents a promising approach to
overcoming these limitations by synergistically targeting multiple
disease pathways. Through clinical data analyses, Siglec15 has been
identified as a novel therapeutic target in T1D. Consequently, the
present study engineered MSCs to overexpress this immunomodulatory
molecule. The resulting Siglec15-MSCs, representing a novel
combination immunotherapy strategy, exhibited markedly enhanced
immunoregulatory capacity and superior therapeutic efficacy
compared with control MSCs in NOD mice. Looking ahead, the
administration of Siglec15-expressing MSCs, potentially in
conjunction with other islet-regenerative or immunomodulatory
agents, represents a promising strategy to enhance therapeutic
efficacy and promote sustained euglycemia, even in patients with
established disease.
The present study was subject to several
limitations. First, the sample size of participants was limited and
there is an absence of data concerning the long-term safety and
efficacy of the intervention. Second, the specific mechanisms
through which Siglec15 modulates T-cell autoimmunity and maintains
β-cell function were not explored. Additionally, although the
animal experiments did not demonstrate tumorigenesis, previous
studies have linked elevated Siglec15 expression with tumor immune
suppression, underscoring the need for comprehensive safety
assessments to exclude any potential oncogenic risks associated
with this therapeutic target (38,40). Consequently, future research
involving larger cohorts and extended observation periods is
essential to validate these findings and address the current
limitations.
The present study is the first to report a
significant reduction in circulating soluble Siglec15 levels in
patients with newly diagnosed T1D compared with healthy controls.
Although no correlation with clinical parameters was detected
within the patient cohort, functional analyses demonstrated that
mesenchymal stem cells engineered to overexpress SIGLEC15 exert a
robust protective effect. In NOD mice, administration of
Siglec15-MSCs during the pre-diabetic stage markedly reduced
diabetes incidence, mitigated the severity of insulitis and
preserved functional, insulin-producing islet mass. The therapeutic
mechanism appears to involve immunomodulation, specifically through
the expansion of memory T-cell populations in pancreatic-draining
lymph nodes. Collectively, these findings underscored the potential
of Siglec15-MSCs-based therapy as a novel strategy to modulate
autoimmunity and prevent the onset of T1D.
Supplementary Data
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
HG conceptualized the present study and analyzed the
data, performed the investigation, the visualization and validation
and designed the methodology; he also wrote the original draft and
reviewed and edited the manuscript. YT, SL, YG, YH and CF performed
the investigation, visualization and data validation. CF and JH
conceptualized the present study and wrote, reviewed and edited the
manuscript. They were also responsible for the funding acquisition
and the supervision. JH and CF confirm the authenticity of all the
raw data. All authors reviewed and approved the final
manuscript.
Ethics approval and consent to
participate
All patients provided signed, informed consent for
their tissues to be used for scientific research. Ethical approval
for the present study was obtained from the Second Affiliated
Hospital of Soochow University (Suzhou, China; approval no.
JD-LK2025059-IO1). The in vivo experimental procedures
performed in the present study were approved by the Institutional
Animal Care and Use Committee of Soochow University (Suzhou, China;
approval no. 202412A0289.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
Dr Chen Fang ORCID: 0000-0003-4329-9471
Acknowledgments
Not applicable.
Funding
The present study received funding from National Natural Science
Foundation of China (grant no. 82100881 to Heming Guo); The Suzhou
Science and Education Strengthening Health Youth Project (grant no.
KJXW2022014 to Ying Gu and grant no. KJXW2022013 to Yiting
Huang).
References
|
1
|
Quattrin T, Mastrandrea LD and Walker LSK:
Type 1 diabetes. Lancet. 401:2149–2162. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Subramanian S, Khan F and Hirsch IB: New
advances in type 1 diabetes. BMJ. 384:e0756812024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang W, Ling Q, Wang B, Wang K, Pang J,
Lu J, Bi Y and Zhu D: Comparison of therapeutic effects of
mesenchymal stem cells from umbilical cord and bone marrow in the
treatment of type 1 diabetes. Stem Cell Res Ther. 13:4062022.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Primavera R, Regmi S, Yarani R, Levitte S,
Wang J, Ganguly A, Chetty S, Guindani M, Ricordi C, Meyer E and
Thakor AS: Precision delivery of human bone marrow-derived
mesenchymal stem cells into the pancreas via intra-arterial
injection prevents the onset of diabetes. Stem Cells Transl Med.
13:559–571. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Madani S, Amanzadi M, Aghayan HR, Setudeh
A, Rezaei N, Rouhifard M and Larijani B: Investigating the safety
and efficacy of hematopoietic and mesenchymal stem cell
transplantation for treatment of T1DM: A systematic review and
meta-analysis. Syst Rev. 11:822022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
de Lima KA, de Oliveira GL, Yaochite JN,
Pinheiro DG, de Azevedo JT, Silva WA Jr, Covas DT, Couri CE, Simões
BP, Voltarelli JC, et al: Transcriptional profiling reveals
intrinsic mRNA alterations in multipotent mesenchymal stromal cells
isolated from bone marrow of newly-diagnosed type 1 diabetes
patients. Stem Cell Res Ther. 7:922016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Carlsson PO, Schwarcz E, Korsgren O and Le
Blanc K: Preserved β-cell function in type 1 diabetes by
mesenchymal stromal cells. Diabetes. 64:587–592. 2015. View Article : Google Scholar
|
|
8
|
Dharmadhikari G, Stolz K, Hauke M, Morgan
NG, Varki A, de Koning E, Kelm S and Maedler K: Siglec-7 restores
β-cell function and survival and reduces inflammation in pancreatic
islets from patients with diabetes. Sci Rep. 7:453192017.
View Article : Google Scholar
|
|
9
|
Bandala-Sanchez E, Zhang Y, Reinwald S,
Dromey JA, Lee BH, Qian J, Böhmer RM and Harrison LC: T cell
regulation mediated by interaction of soluble CD52 with the
inhibitory receptor Siglec-10. Nat Immunol. 14:741–748. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Guo M, Guo H, Zhu J, Wang F, Chen J, Wan
C, Deng Y, Wang F, Xu L, Chen Y, et al: A novel subpopulation of
monocytes with a strong interferon signature indicated by SIGLEC-1
is present in patients with in recent-onset type 1 diabetes.
Diabetologia. 67:623–640. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
He F, Wang N, Li J, He L, Yang Z, Lu J,
Xiong G, Yu C and Wang S: High affinity monoclonal antibody
targeting Siglec-15 for cancer immunotherapy. J Clin Transl Res.
7:739–749. 2021.
|
|
12
|
Wang J, Sun J, Liu LN, Flies DB, Nie X,
Toki M, Zhang J, Song C, Zarr M, Zhou X, et al: Siglec-15 as an
immune suppressor and potential target for normalization cancer
immunotherapy. Nat Med. 25:656–666. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hu J, Yu A, Othmane B, Qiu D, Li H, Li C,
Liu P, Ren W, Chen M, Gong G, et al: Siglec15 shapes a non-inflamed
tumor microenvironment and predicts the molecular subtype in
bladder cancer. Theranostics. 11:3089–3108. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guo H, Shen Y, Kong YH, Li S, Jiang R, Liu
C, Fang C and Hu J: The expression of Tim-1 and Tim-4 molecules in
regulatory T cells in type 1 diabetes. Endocrine. 68:64–70. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Angata T and Varki A: Discovery,
classification, evolution and diversity of siglecs. Mol Aspects
Med. 90:1011172023. View Article : Google Scholar
|
|
16
|
American Diabetes Association: 2.
Classification and diagnosis of diabetes: Standards of medical care
in diabetes-2021. Diabetes Care. 44:S15–S33. 2021. View Article : Google Scholar
|
|
17
|
Ansari MJ, Salama AD, Chitnis T, Smith RN,
Yagita H, Akiba H, Yamazaki T, Azuma M, Iwai H, Khoury SJ, et al:
The programmed death-1 (PD-1) pathway regulates autoimmune diabetes
in nonobese diabetic (NOD) mice. J Exp Med. 198:63–69. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-delta delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
19
|
Hiruma Y, Hirai T and Tsuda E: Siglec-15,
a member of the sialic acid-binding lectin, is a novel regulator
for osteoclast differentiation. Biochem Biophys Res Commun.
409:424–429. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Du H, Tang J, Li X, Wang X, Wu L, Zhang R,
Hu P and Yang Y: Siglec-15 is an immune suppressor and potential
target for immunotherapy in the pre-metastatic lymph node of
colorectal cancer. Front Cell Dev Biol. 9:6919372021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kucia M, Reca R, Jala VR, Dawn B,
Ratajczak J and Ratajczak MZ: Bone marrow as a home of heterogenous
populations of nonhematopoietic stem cells. Leukemia. 19:1118–1127.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ji JF, He BP, Dheen ST and Tay SS:
Interactions of chemokines and chemokine receptors mediate the
migration of mesenchymal stem cells to the impaired site in the
brain after hypoglossal nerve injury. Stem Cells. 22:415–427. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ratajczak J, Wysoczynski M, Zuba-Surma E,
Wan W, Kucia M, Yoder MC and Ratajczak MZ: Adult murine bone
marrow-derived very small embryonic-like stem cells differentiate
into the hematopoietic lineage after coculture over OP9 stromal
cells. Exp Hematol. 39:225–237. 2011. View Article : Google Scholar
|
|
24
|
Izadi M, Sadr Hashemi Nejad A, Moazenchi
M, Masoumi S, Rabbani A, Kompani F, Hedayati Asl AA, Abbasi
Kakroodi F, Jaroughi N, Mohseni Meybodi MA, et al: Mesenchymal stem
cell transplantation in newly diagnosed type-1 diabetes patients: A
phase I/II randomized placebo-controlled clinical trial. Stem Cell
Res Ther. 13:2642022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wu H and Mahato RI: Mesenchymal stem
cell-based therapy for type 1 diabetes. Discov Med. 17:139–143.
2014.PubMed/NCBI
|
|
26
|
Zhou T, Yuan Z, Weng J, Pei D, Du X, He C
and Lai P: Challenges and advances in clinical applications of
mesenchymal stromal cells. J Hematol Oncol. 14:242021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gou W, Hua W, Swaby L, Cui W, Green E,
Morgan KA, Strange C and Wang H: Stem cell therapy improves human
islet graft survival in mice via regulation of macrophages.
Diabetes. 71:2642–2655. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Özçelik H, Yazır Y, Duruksu G, Kılıç KC
and Öztürk A: Membrane-engineered Wharton's Jelly derived
mesenchymal stem cells with anti-CD2 antibody coating modulate
activated CD3+ T-cell responses. Tissue Cell. 99:1032742026.
View Article : Google Scholar
|
|
29
|
Khazaei M, Khazaei F, Niromand E and
Ghanbari E: Tissue engineering approaches and generation of
insulin-producing cells to treat type 1 diabetes. J Drug Target.
31:14–31. 2023. View Article : Google Scholar
|
|
30
|
Luque-Campos N, Contreras-López RA, Jose
Paredes-Martínez M, Torres MJ, Bahraoui S, Wei M, Espinoza F,
Djouad F, Elizondo-Vega RJ and Luz-Crawford P: Mesenchymal stem
cells improve rheumatoid arthritis progression by controlling
memory T cell response. Front Immunol. 10:7982019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Higdon LE, Cooney LA, Serti E, Suwannasaen
D, Muir VS, Wiedeman AE, Harris KM, Pardo J, Anderson MS, Speake C,
et al: Early expansion of TIGIT+PD1+ effector memory CD4 T cells
via agonistic effect of alefacept in new-onset type 1 diabetes. J
Immunol. 214:12–22. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhao Y, Knight CM, Jiang Z, Delgado E, Van
Hoven AM, Ghanny S, Zhou Z, Zhou H, Yu H, Hu W, et al: Stem cell
educator therapy in type 1 diabetes: From the bench to clinical
trials. Autoimmun Rev. 21:1030582022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Passeri L, Andolfi G, Bassi V, Russo F,
Giacomini G, Laudisa C, Marrocco I, Cesana L, Di Stefano M, Fanti
L, et al: Tolerogenic IL-10-engineered dendritic cell-based therapy
to restore antigen-specific tolerance in T cell mediated diseases.
J Autoimmun. 138:1030512023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Luo X, Pothoven KL, McCarthy D, DeGutes M,
Martin A, Getts DR, Xia G, He J, Zhang X, Kaufman DB and Miller SD:
ECDI-fixed allogeneic splenocytes induce donor-specific tolerance
for long-term survival of islet transplants via two distinct
mechanisms. Proc Natl Acad Sci USA. 105:14527–14532. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Golden GJ, Wu VH, Hamilton JT, Amses KR,
Shapiro MR, Sada Japp A, Liu C, Pampena MB, Kuri-Cervantes L, Knox
JJ, et al: Immune perturbations in human pancreas lymphatic tissues
prior to and after type 1 diabetes onset. Nat Commun. 16:46212025.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang Y, Xu Z, Wu KL, Yu L, Wang C, Ding H,
Gao Y, Sun H, Wu YH, Xia M, et al: Siglec-15/sialic acid axis as a
central glyco-immune checkpoint in breast cancer bone metastasis.
Proc Natl Acad Sci USA. 121:e23129291212024. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lenza MP, Egia-Mendikute L,
Antoñana-Vildosola A, Soares CO, Coelho H, Corzana F, Bosch A,
Manisha P, Quintana JI, Oyenarte I, et al: Structural insights into
Siglec-15 reveal glycosylation dependency for its interaction with
T cells through integrin CD11b. Nat Commun. 14:34962023. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang XY, Shi JB, Jin SF, Wang RJ, Li MY,
Zhang ZY, Yang X and Ma HL: Metabolic landscape of head and neck
squamous cell carcinoma informs a novel kynurenine/Siglec-15 axis
in immune escape. Cancer Commun (Lond). 44:670–694. 2024.
View Article : Google Scholar : PubMed/NCBI
|