The expression and activity of protein are tightly
controlled by epigenetic processes, particularly post-translational
modifications. Among these, ubiquitination is a highly conserved
and reversible modification that covalently attaches ubiquitin
molecules to target proteins, thereby modulating their stability,
activity, localization and interactions (1). Ubiquitination is essential in
diverse physiological and pathological processes, including protein
degradation, cell cycle regulation, signal transduction, gene
expression and autophagy (2,3).
The ubiquitin-proteasome system (UPS) is the principal pathway for
intracellular protein degradation and regulation. It consists of
ubiquitin, ubiquitin-activating enzymes (E1), conjugating enzymes
(E2), ligases (E3), deubiquitinating enzymes (DUBs) and the 26S
proteasome. In eukaryotes, 80-90% of protein degradation depends on
the UPS, which is mediated through a sequential enzymatic cascade
(4). Ubiquitin contains seven
lysine residues (K6, K11, K27, K29, K33, K48 and K63) and one
N-terminal methionine residue (Met1), which can be used to form
ubiquitin chains. Notably, ubiquitin can form monomer ubiquitin
chains or polyubiquitin chains (such as K48 and K63). Different
types of ubiquitin chains can regulate different cellular processes
(5). For instance, the K48 chain
is usually labeled with protein for degradation (6), while the K63 chain is involved in
signal transduction (7). K6
chain are associated with autophagy and DNA damage response. K11
chain participates in cell-cycle regulation and proteasomal
degradation. K27 chain is implicated in protein secretion and
innate immunity. K29 chain has a role in neurodegenerative
disorders. K33 chain can influence protein trafficking (8,9).
Notably, in contrast to the well-characterized K48 and K63-linked
chains, these atypical ubiquitin linkages remain comparatively
understudied. Initially, ubiquitin is activated by E1 in an
ATP-dependent manner, transferred to the cysteine residue of E2,
and subsequently conjugated to substrate proteins via E3 ligases,
resulting in substrate ubiquitination (10).
DUBs represent a major branch of the ubiquitin
system. They contain ubiquitin-binding motifs that facilitate the
recognition and recruitment of ubiquitinated proteins, thereby
ensuring specificity and precise regulation (11). While ubiquitin attachment is
mediated by the E1/E2/E3 enzymatic cascade, DUBs reverse this
process by cleaving peptide or isopeptide bonds between ubiquitin
and its substrates. This deubiquitination not only stabilizes
proteins but also fine-tunes signaling cascades, preventing
excessive activation and maintaining ubiquitin system homeostasis
(12,13). There are ~100 DUBs known in
humans and they are categorized into seven families:
Ubiquitin-specific proteases (USPs), ovarian tumor proteases,
Josephins and JAB1/MPN/Mov34 metalloenzymes, Machado-Joseph disease
protein domain proteases, ubiquitin carboxy-terminal hydrolases
(UCHs), motif-interacting with ubiquitin-containing novel DUB
family and the zinc finger-containing ubiquitin peptidase 1
(14). USPs are indispensable
for regulating critical biological functions such as DNA damage and
repair, metabolism, cellular differentiation, epigenetic modulation
and protein stability (15-18). USP10 dysregulation has been
associated with multiple human diseases, including inflammatory
disorders and infections (19-21), cardiovascular disease (22-24) and neurodegeneration (25-27). Alterations in the expressions of
USPs have also been observed in cancers, which suggests that these
proteins are involved in tumorigenic mechanisms (28-31).
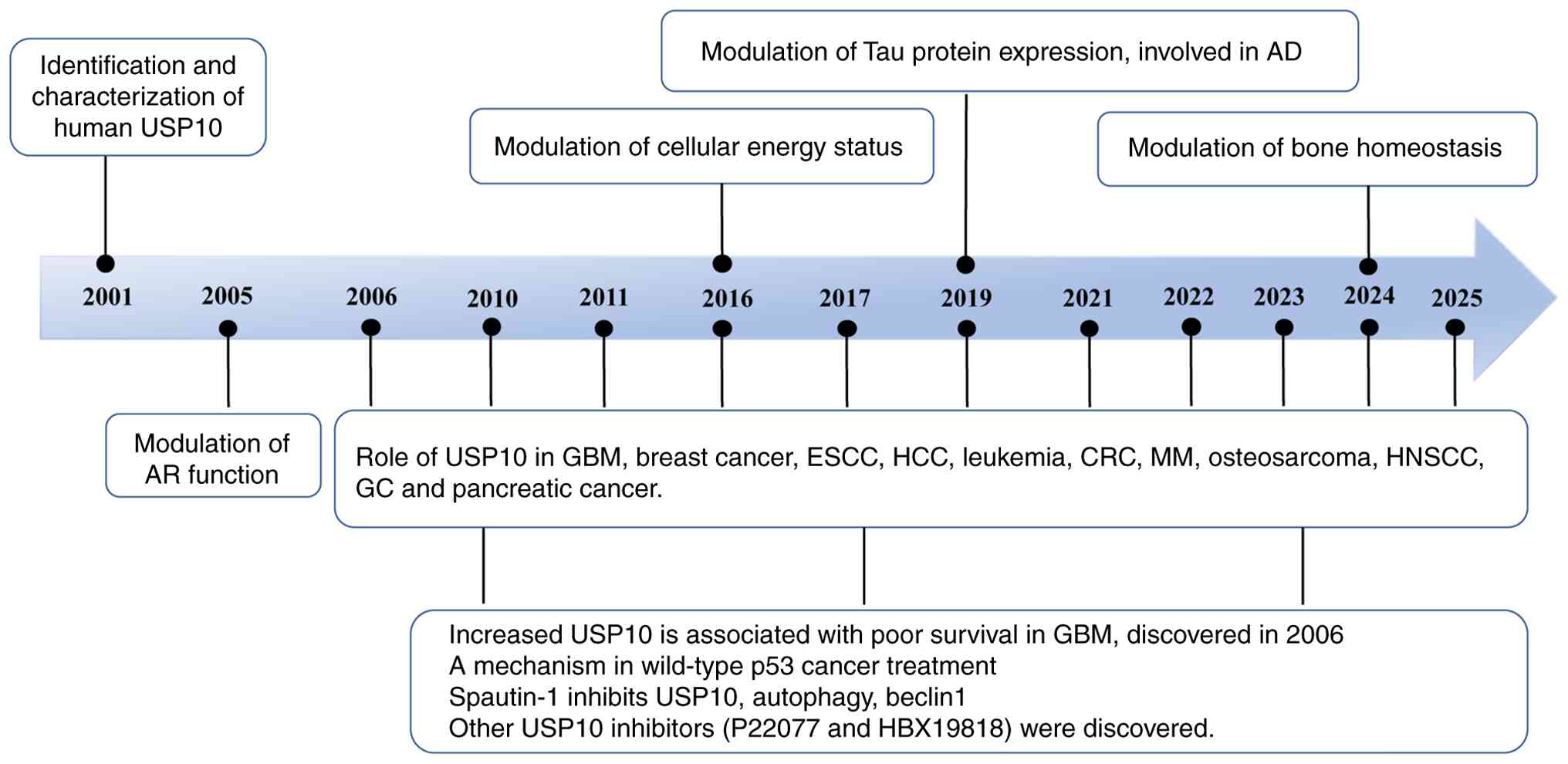
USP10, a deubiquitinase that interacts with Ras-GAP
SH3-domain-binding proteins, was first described by Soncini et
al (32) in 2001, and
subsequent research has progressively revealed its diverse
biological functions (Fig. 1).
There have been reports that USP10 modulates p53 stability
(33,34), autophagy (35,36), DNA damage response (DDR)
(37-39) and cellular energy metabolism
(40,41). While early studies primarily
focused on its oncogenic roles in specific cancers, more recent
findings highlight its broader biological functions, regulatory
mechanisms and pathological relevance across multiple diseases. The
present review offers a comprehensive overview of USP10 biology,
with particular emphasis on its mechanistic links to human diseases
and its potential as a therapeutic target, offering new
perspectives for cancer and beyond.
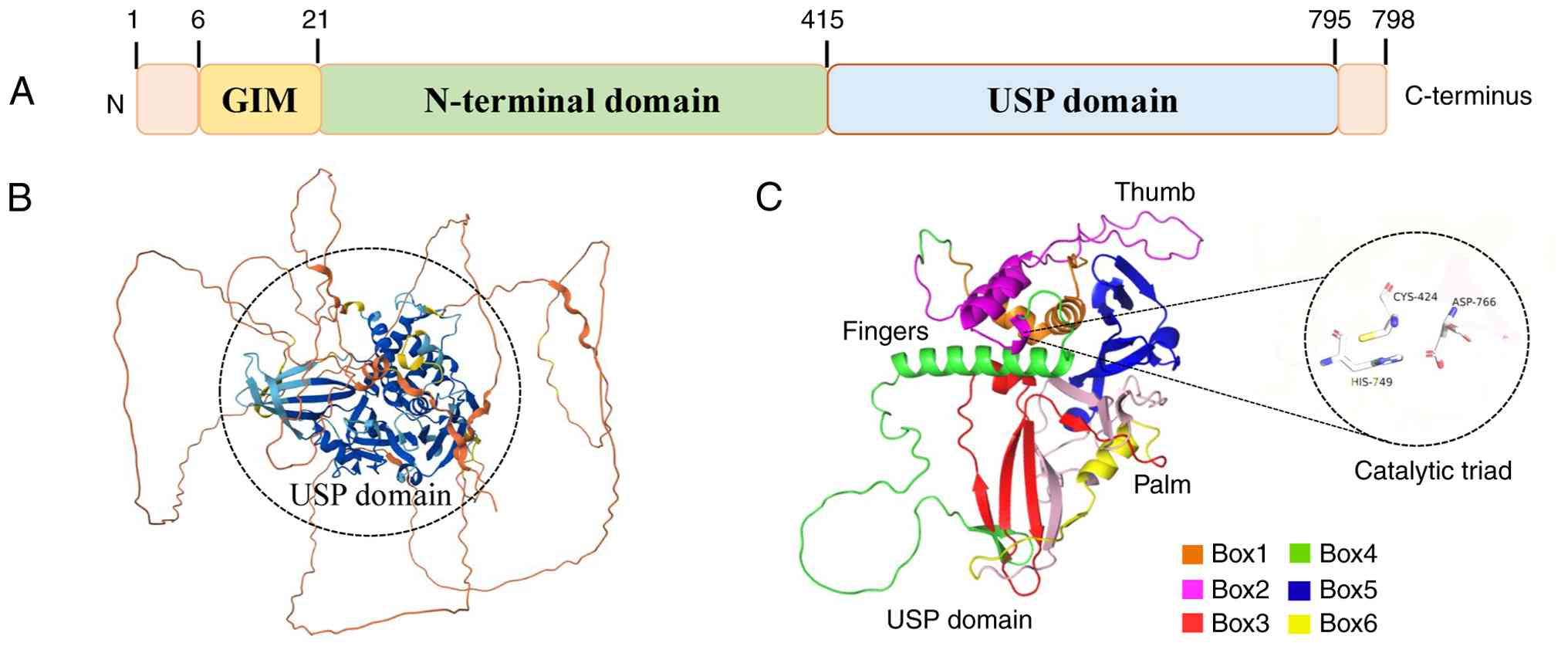
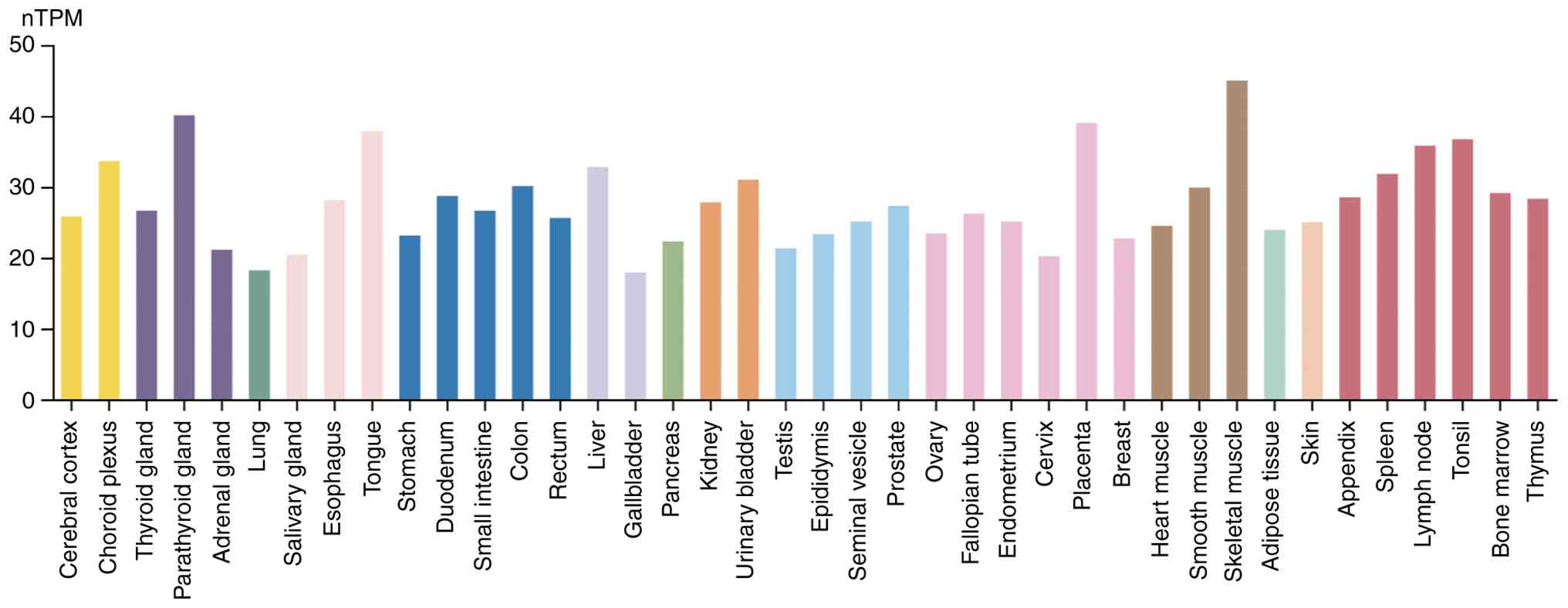
USP10 is evolutionarily conserved, sharing ~99% AA
sequence homology with its mouse counterparts, and is broadly
expressed in both the nucleus and cytoplasm of most cell types.
Expression of USP10 has been detected in multiple human tissues,
such as brain, kidney, adrenal gland, breast and stomach (47). The highest RNA expression of
USP10 is observed in skeletal muscle (Fig. 3) according to the Human Protein
Atlas (https://www.proteinatlas.org).
Abnormal upregulation of USP10 has been reported in several cancer
types, including osteosarcoma (36), breast cancer (48), pancreatic ductal adenocarcinoma
(PDAC) (49), colorectal cancer
(CRC) (50), prostate cancer
(51), non-small cell lung
cancer (NSCLC) (52), ovarian
cancer (52), glioblastoma (GBM)
(53), esophageal squamous cell
carcinoma (ESCC) (54) and head
and neck squamous cell carcinoma (55). Notably, USP10 expression is also
found in lower eukaryotes such as yeast (56), where its homolog, UBP3, is
essential for DNA repair (57).
The subcellular localization of USP10 is highly
dynamic and context-dependent. Under non-stressed conditions, USP10
is predominantly cytoplasmic, where it functions as a
deubiquitinase to stabilize and enhance p53 activity. Following DNA
damage and ATM serine/threonine kinase (ATM) activation, USP10
becomes phosphorylated and translocates to the nucleus, where it
cooperates with USP7 to deubiquitinate and stabilize p53 (34). This nucleo-cytoplasmic shuttling
is essential for USP10 to access and regulate compartment-specific
substrates such as nuclear p53. Additional research has
demonstrated that AKT-mediated phosphorylation of USP10 at Thr764
within its nuclear localization signal promotes nuclear
translocation (58). More
recently, Liu et al (38)
demonstrated that oxaliplatin treatment markedly increases the
nuclear expression of USP10 in CRC cells. Nuclear USP10 enhances
CRC proliferation, oxaliplatin resistance and DNA repair by
stabilizing XPA binding protein 2 (XAB2). Notably, although USP10
nuclear translocation can be induced by ATM-mediated
phosphorylation (Thr42/Ser337) following stress and by AKT-mediated
phosphorylation (Thr764) under chemotherapeutic exposure, current
evidence does not support a sequential or hierarchical relationship
between these pathways. Instead, ATM and AKT appear to regulate
USP10 localization through distinct, context-dependent mechanisms,
and it remains unclear whether they interact or compensate for each
other in specific disease settings such as oxaliplatin-treated CRC.
In summary, these findings indicate that USP10 localization is
highly plastic and sensitive to external stimuli, including DNA
damage and chemotherapeutic stress.
DDR is a sophisticated protein network that
coordinates DNA repair with cell-cycle check-points to protect
cells from genomic insults (59). Increasing evidence indicates that
USPs are key regulators of DDR, acting through deubiquitination of
critical mediators such as p53, checkpoint kinase 2 (CHK2) and
SWI/SNF-related matrix-associated actin-dependent regulator of
chromatin subfamily A member 5 (60-64). For example, Zhu et al
(65) reported that USP33
stabilizes p53 via deubiquitination, thereby modulating the DDR.
Similarly, USP39 enhances DNA damage repair and radioresistance by
stabilizing CHK2 (66).
MutS homolog 2 (MSH2), a central DNA mismatch repair
protein, is essential for genomic stability. A previous study
showed that USP10 deubiquitinates and stabilizes MSH2, thereby
influencing cellular sensitivity to DNA damage (37). Recent findings revealed that
USP10 binds to XAB2 and removes ubiquitin at Lys593, preventing
proteasome-mediated degradation and stabilizing the protein.
Stabilized XAB2 upregulates annexin A2 (ANXA2) transcription by
binding to its promoter, thereby facilitating CRC cell growth, DDR
activation, oxaliplatin-induced DNA repair and chemoresistance
(38). Additional evidence
further links USP10 to therapeutic resistance. Zhao et al
(67) showed that astrocyte
elevated gene-1 (AEG-1) mitigates radiation-induced DNA damage and
enhances radioresistance in ESCC. Mechanistically, AEG-1 recruits
USP10 by removing K48-linked polyubiquitin chains from
poly(ADP-ribose) polymerase 1 (PARP1) at Lys425 to prevent its
proteasomal degradation. This stabilization facilitates homologous
recombination-mediated repair of DNA double-strand breaks,
ultimately reducing DNA damage and conferring resistance to
irradiation (67).
Autophagy is an essential mechanism for preserving
cellular homeostasis, contributing to environmental adaptation,
pathogen clearance, aging delay and energy stress responses. USP10
participates in autophagy activation by modulating the activity of
autophagy-related molecules such as microtubule-associated protein
1 light chain 3b (LC3B) and Beclin1. Under starvation stress in
neonatal mice, USP10 promotes survival by removing ubiquitin
modifications from murine double minute 2, thereby enhancing its E3
ligase activity toward p53 and inducing autophagy (33). USP10 also deubiquitinates LC3B,
resulting in increased LC3B levels and enhanced autophagy (35). Furthermore, Liu et al
(68) demonstrated that USP10
stabilizes Beclin1 by preventing its ubiquitin-mediated
degradation, thereby facilitating autophagy.
SGs are cytoplasmic condensates in eukaryotic cells,
with G3BP1/2 identified as critical regulators of their assembly.
USP10 is essential to SG dynamics by interacting with G3BPs, thus
regulating their formation and disassembly (69). Recently, Tahmasebinia et
al (70) showed that the
USP10-G3BP1 complex functions as a central hub interfacing with
pathways such as energy metabolism, enabling fine-tuning of
mitochondrial homeostasis and rapid adaptation to environmental
stimuli.
A previous study identified USP10 as a
transcriptional coactivator of androgen receptor (AR)-regulated
genes, where it binds AR and enhances androgen-responsive promoter
activity (71). Recent findings
revealed that USP10 and sirtuin 6 (SIRT6) work together to preserve
spermatogonial proliferation and DNA repair capacity, thereby
providing critical protection against spermatogenic defects induced
by microcystin-LR (72). USP10
also regulates the stability of G3BP1, a core component of SGs,
thereby influencing their assembly and disassembly. This mechanism
helps maintain SG homeostasis in supporting cells and ensures
normal spermatogenesis. Furthermore, USP10 modulates SG dynamics in
the presence of pathological protein variants, such as fused in
sarcomaR521C, linking USP10 to male fertility and the
pathogenesis of related reproductive disorders (73).
There is evidence that USP10 serves a notable role
in regulating immune and inflammatory responses (74,75). In a mouse model of cerebral
ischemia-reperfusion (I/R) injury, USP10 was shown to promote
neuroinflammation through activation of the NF-κB signaling pathway
(74). Conversely, USP10 binds
the nuclear T-box transcription factor T-bet, stabilizing it
through deubiquitination and thereby alleviating type 2 T-helper
cell-dominant inflammation in asthma (76). Retinoic acid inducible gene-I
(RIG-I) recognizes viral RNA and triggers robust antiviral
responses by inducing type I interferon production. To transmit
RIG-I-mediated signaling, the mitochondrial adaptor mitochondrial
antiviral signaling protein (MAVS) assembles into prion-like
aggregates that activate downstream kinases and transcription
factors (77). Liu et al
(78) demonstrated that USP10
removes unanchored K63-linked polyubiquitin chains from MAVS,
thereby suppressing RIG-I-mediated MAVS aggregation. Consistently,
USP10-deficient mice displayed increased resistance to RNA virus
infection. Previously, USP10 has been shown to regulate humoral
immunity. By deubiquitinating activation-induced cytidine deaminase
(AID), USP10 modulates neutralizing antibody production following
nanoparticle vaccination against severe acute respiratory syndrome
coronavirus 2 and human immunodeficiency virus type 1 (58).
Cell signaling is the process by which chemical or
physical cues are transmitted through a cascade of molecular
events, ultimately contributing to the expression of specific genes
and consequent biological responses such as proliferation and
apoptosis (84). Numerous
signaling pathways have been identified to be regulated by USP10,
including Notch (85), TGF-β
(86), NF-κB (87,88), Wnt (89), Hippo (90,91) and PI3K/AKT (92). Specifically, USP10 removes
ubiquitin chains from αv-integrin, resulting in its accumulation on
the cell surface, subsequent activation of TGFβ1 signaling and
pathological differentiation of myofibroblasts (93). The role of USP10 in modulating
these signaling pathways has been comprehensively reviewed by Chen
et al (94). Taken
together, USP10 exerts critical regulatory functions across diverse
cellular processes, including DNA damage repair, autophagy, immune
responses and signal transduction.
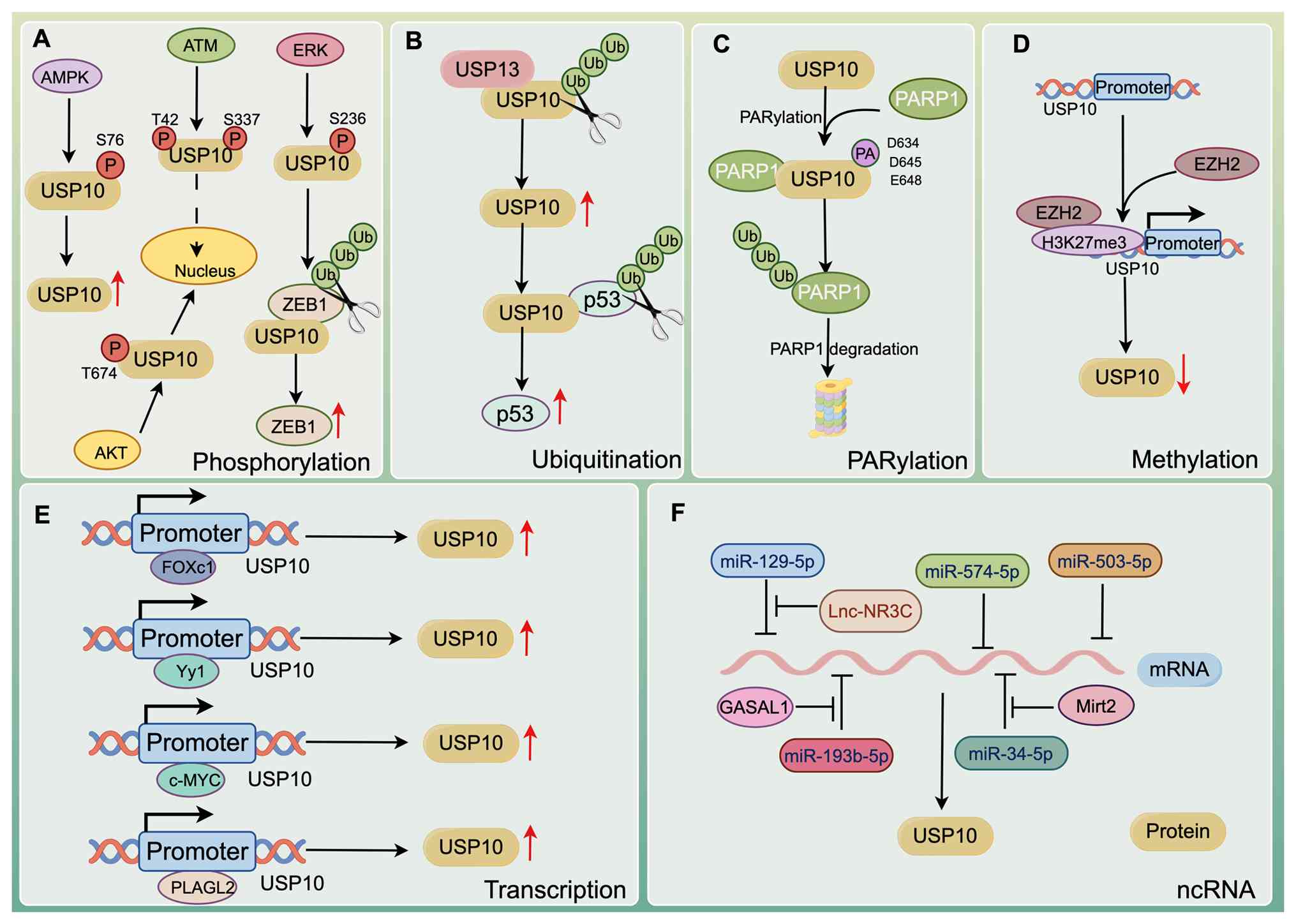
Such as most deubiquitinases, USP activity may be
modulated through numerous mechanisms at both transcriptional and
post-translational levels. Similarly, USP10 expression is tightly
controlled by phosphorylation, histone methylation, ubiquitination,
PARylation and transcriptional regulation (Fig. 4). Elucidating these regulatory
mechanisms may provide critical insights for the development of
novel therapeutic strategies targeting USP10.
Transcription is essential for gene expression and
cellular function. As key regulatory molecules, transcription
factors modulate gene expression by binding DNA sequences and
influencing transcriptional activity through their effector
domains. Dysregulation of transcription can alter gene expression
patterns, which can aid in the onset and progression of various
diseases. The transcriptional activity of USP10 is regulated by
factors such as Yin Yang-1 (Yy1), c-Myc, FOXC1 and pleomorphic
adenoma gene like-2 (PLAGL2). Yy1 downregulates USP10
transcription, aggravating pathological cardiac hypertrophy
(95). Conversely, c-Myc induces
USP10 transcription, which stabilizes p14ARF protein; USP10, in
turn, deubiquitinates p14ARF, preventing its proteasome-dependent
degradation and accelerating mouse embryonic fibroblast
hyperproliferation (96). FOXC1
transcriptionally upregulates USP10 mRNA, thereby activating Wnt
signaling and promoting PDAC progression (89). PLAGL2 enhances the transcription
of the USP10 promoter by interacting with it, while USP10
deubiquitinates and stabilizes PLAGL2 protein, forming a positive
feedback loop that mutually reinforces transcriptional activation
(97).
USP10 activity is regulated in part through
phosphorylation of its N-terminal domain. Yuan et al
(34) demonstrated that,
following DNA damage, ATM phosphorylates USP10 at Thr42 and Ser337,
promoting its nuclear translocation. In the nucleus, the N-terminal
region of USP10 interacts with p53 and prevents it from becoming
ubiquitinated. Similarly, another study indicated that
co-stimulation of B cells express BCR (a surface receptor for
antigen) and Toll-like receptor (TLR)1/2 triggers AKT-dependent
phosphorylation of USP10 at Thr674, leading to nuclear
translocation and stabilization of AID (58). Under energy stress, AMP-activated
protein kinase (AMPK) phosphorylates USP10 at Ser76, enhancing
USP10-mediated deubiquitination of AMPK. In turn, USP10 promotes
AMPK activity through K63-linked deubiquitination, establishing a
positive feedback loop to respond to fluctuations in cellular
energy metabolism (40).
Likewise, it has been shown that activated AMPK phosphorylates
USP10 at Ser76 to enhance deubiquitination and stability of the
scaffold protein Axin1 (98).
This phosphorylation also strengthens USP10/β-catenin interaction
and supports β-catenin phase separation, thereby inhibiting tumor
growth (98). Conversely, Sun
et al (99) revealed that
ERK phosphorylates USP10 at Ser236, which weakens the association
of USP with zinc finger E-box binding homeobox 1 (ZEB1), enhancing
ZEB1 protein stability and promoting colorectal cancer
metastasis.
Enhancer of zeste homolog 2 (EZH2) is the catalytic
subunit of the polycomb repressive complex 2 and a highly conserved
histone methyltransferase. EZH2 modulates downstream gene
expression by catalyzing trimethylation of lysine 27 on histone H3
(H3K27me3) (100). Lei et
al (101) demonstrated that
EZH2 modulates protein stability in malignant cells by recruiting
USP7, thereby influencing neuronal gene expression. Huang et
al (102) demonstrated that
EZH2 promotes USP22 transcription by modulating H3K27me3 at the
USP22 promoter, which in turn enhances programmed death-ligand 1
(PD-L1) protein stability. Recently, EZH2 was shown to suppress
USP10 expression by increasing H3K27me3 at the USP10 promoter. This
repression enhanced ubiquitin-mediated degradation of glutathione
peroxidase 4, ultimately promoting ferroptosis in alveolar
epithelial cells during sepsis (103).
USP13 stabilizes USP10 by removing ubiquitin chains
through its deubiquitinase activity, therefore averting
proteasome-mediated destruction (68). Notably, Beclin1 can modulate the
deubiquitination activity of both USP10 and USP13, controlling
their protein stability (68).
Mechanistically, Beclin1 functions as a scaffold within the Vps34
complex, maintaining the integrity and deubiquitinating capacity of
USP10 and USP13. Notably, by interacting with and stabilizing
USP13, Beclin1 indirectly facilitates USP13-mediated
deubiquitination of USP10, thereby contributing to maintaining
USP10 stability (68).
PARylation, a post-translational modification required for
initiation of the DDR, also regulates USP10. PARP1, a key enzyme in
the mammalian (ADP-ribosyl) transferase family, catalyzes
PARylation (104). A recent
study revealed that PARP1-mediated PARylation of USP10 at D634,
D645 and E648 enhances its deubiquitination activity, thereby
amplifying the DDR (39).
MicroRNAs (miRNA/miR) bind sequence-specifically to
the 3'-untranslated region (3'-UTR) of target mRNAs, promoting
degradation or preventing translation. For instance, Peng et
al (105) identified USP10
as a direct target of miR-503-5p, which binds its conserved 3'-UTR
region and suppresses both USP10 mRNA and protein expression.
Similarly, Ma et al (106) reported that miR-574-5p
upregulation reduces USP10 expression. Conversely, several ncRNAs,
including long ncRNA (lncRNA)-NR3C (107), Mirt2 (108) and GASAL1 (109), have been shown to increase
USP10 expression.
Together, these findings represent only part of the
regulatory mechanisms governing USP10 (Table I). With further investigation,
future studies are expected to integrate transcriptional,
translational and post-translational layers to construct a more
comprehensive network of USP10 regulation.
Due to its central role in regulating stress
responses, genome stability, immune activation and protein
homeostasis, dysregulation of USP10 has been associated with the
pathogenesis of several human diseases. The present section
highlights the major disease systems in which USP10 has
demonstrated pathogenic relevance.
Growing data suggest that dysregulation of USP10 is
closely related to cancer initiation, cell proliferation and
metastasis. Li et al (50) demonstrated that USP10 promotes
CRC progression and tumor-associated macrophage polarization
through deubiquitination of NLR family pyrin domain containing
(NLRP)7. Similarly, USP10 stabilizes Musashi-2 via deubiquitination
to enhance tumor proliferation in colon cancer (110). PLAGL2, a zinc finger
transcription factor implicated in multiple cancers (111,112), is stabilized by USP10-mediated
deubiquitination. Adrenergic signaling through the adrenergic
β-receptor 2-c-Myc axis upregulates USP10, which stabilizes PLAGL2
and drives hepatocellular carcinoma (HCC) progression (97). In pancreatic cancer, Quan et
al (41) reported that USP10
deubiquitinates phosphoglycerate kinase 1 (PGK1), enhancing its
protein stability and promoting aerobic glycolysis. Kruppel-like
factor (KLF)15 acts as a scaffold to improve USP-PGK1 interaction,
further supporting proliferation and metastasis. Wang et al
(113) showed that tribbles
pseudokinase 3 binds USP10 to increase structure specific
recognition protein 1 expression, promoting multiple myeloma (MM)
progression. USP10 also deubiquitinates KLF4, and loss of USP10
downregulates KLF4 to facilitate lung tumorigenesis (114). Additionally, LINC00240
stabilizes DEAD-box helicase 21 (DDX21) and accelerates the
development of gastric cancer (GC) by suppressing USP10-mediated
deubiquitination of DDX21 (115).
USP10 further contributes to tumor proliferation by
preventing degradation of key cell-cycle regulators such as c-Myc
and cyclins D1/D3. For instance, USP10 antagonizes transient c-Myc
activation through SIRT6 and TP53, suppressing cell-cycle
progression, tumor growth and oncogenesis (116). SIRT6, a tumor suppressor, is
stabilized by USP10-mediated deubiquitination. In HCT1 colon cancer
cells, overexpression of either USP10 or SIRT6 decreases the
proportion of cells in G2/M phases while increasing the
G0/G1 phase, with combined overexpression
showing an enhanced effect (116). USP10 also stabilizes cyclin D1
(117) and cyclin D3 (118), promoting GBM and MM
progression. Furthermore, USP10 increases TNF receptor superfamily
member 10b stability to inhibit epithelial-mesenchymal transition
(EMT) in GC (119), and
stabilizes Yes1 associated transcriptional regulator (YAP1) to
promote osteosarcoma metastasis and EMT (120).
As previously noted, USP10 regulates multiple
signaling pathways, including Wnt, NF-κB and TGF-β. Wang et
al (89) identified a
positive feedback loop between USP10 and FOXC1, which activates Wnt
signaling and drives PDAC malignancy. USP10 stabilizes K(lysine)
acetyltransferase 8 (KAT8, also known as MOF) to epigenetically
activate ANXA2/Wnt signaling, enhancing proliferation and
metastasis in ESCC (121).
USP10 also represses NF-κB activation by promoting monocyte
chemotactic protein-1-induced protein-1-mediated deubiquitination
of K63-linked linear ubiquitin chains on NF-κB essential modulator
(122). By directly engaging
with Smad4 and inhibiting its proteasomal degradation, USP10
facilitates HCC metastasis (86). Liu et al (123) demonstrated that USP10
suppresses YAP1 ubiquitination and degradation, promoting cysteine
rich angiogenic inducer 61 (Cyr61) expression, immune evasion,
tumor growth and metastasis. By contrast, USP10 can suppress HCC
progression by inhibiting mTOR activation (124). A study also showed that
circPOKE regulates USP10-mediated Snail deubiquitination,
influencing breast cancer metastasis and stemness (125). In summary, USP10 is pivotal in
modulating cell proliferation, metastasis and signaling across
multiple cancers, including CRC, PDAC, GC, GBM, ESCC, HCC and
breast cancer. Beyond promoting proliferation, USP10 also
contributes to the maintenance of cancer stem cells (CSCs).
USP10 is identified as a CSC marker that facilitates
the development and maintenance of CSCs by modulating the
expression of stemness-related genes, including CD44, a non-kinase
transmembrane glycoprotein intricately linked to CSC proliferation
and tumor progression (126).
USP10-mediated deubiquitination stabilizes CD44, enhancing breast
cancer proliferation, stemness and metastasis (127). Additionally, Shi et al
(128) reported that USP10
promotes CSC traits in head and neck squamous cell carcinoma by
stabilizing bromodomain adjacent to zinc finger domain protein 1A.
In CRC, USP10 drives CSCs and mediates 'super-competition'
signaling to support tumor aggressiveness (129).
USP10 contributes to immune evasion in multiple
ways. It inhibits YAP1 ubiquitination and degradation, upregulates
Cyr61 and promotes PDAC growth and metastasis (123). TANK, a TRAF family-related
NF-κB activator, mediates USP10-dependent deubiquitination of
TRAF6, suppressing NF-κB activation and IL-1R/TLR signaling
(134). These pathways suggest
that USP10 may influence tumor inflammation and immune responses.
Emerging evidence also implicates USP10 in regulating the
infiltration of diverse immune cells within malignancies, perhaps
modulating the abundance of specific immune cell types (135). USP10 may also function as a
regulator of PD-L1. It stabilizes PD-L1 by direct deubiquitination
and indirectly via the USP10/ nuclear Dbf2-related 1/PD-L1 axis
(136). In esophageal cancer,
FOXP4-antisense 1 promotes CD8+ T-cell exhaustion and
immune escape by stabilizing PD-L1 through USP10 (137). Furthermore, USP10 inhibits
autocrine motility factor receptor-mediated B7-H4 ubiquitination,
stabilizing B7-H4 and suppressing tumor immune activity, which
reduces the efficacy of SG-targeted therapies (138).
In conclusion, these data highlight USP10 as a key
modulator of CSC maintenance and tumor immune regulation, although
further studies are required to fully clarify its processes across
different cancer contexts.
Beyond its role in tumor progression, USP10
contributes to cancer drug resistance, often by enhancing DNA
damage repair. USP10 binds to XAB2 and removes K48-linked
polyubiquitin chains at K593, thereby stabilizing XAB2. XAB2
subsequently binds to the ANXA2 promoter, upregulating ANXA2
transcription, facilitating DNA damage repair, alleviating
oxaliplatin-induced DNA damage and boosting oxaliplatin resistance
(38). Similarly, USP10
deubiquitinates and stabilizes ATM interactor (ATMIN), leading to
its upregulation in nasopharyngeal carcinoma. ATMIN
transcriptionally activates its downstream target
lymphocyte-specific protein tyrosine kinase, promoting cell
proliferation and docetaxel resistance (139). Another study revealed that DDB1
and CUL4-associated factor 7 acts as a scaffold to recruit USP10 to
G3BP1, enhancing nasopharyngeal carcinoma chemoresistance and
metastasis (140). USP10 also
cooperates with protein phosphatase 1B (PPM1B) to regulate Y box
binding protein 1 (YBX1)-mediated anti-apoptotic signaling. PPM1B
directly interacts with YBX1, inducing dephosphorylation at serine
314, which affects USP10-mediated YBX1 deubiquitination, reduces
YBX1 protein levels and modulates apoptosis and oxaliplatin
resistance in GC cells (141).
Furthermore, Notch1 degradation-associated
regulatory polypeptide (N1DARP) exerts tumor-suppressive and
chemosensitizing effects by modulating the USP10-Notch1 oncogenic
signaling axis, suggesting a promising therapeutic strategy
targeting the N1DARP-Notch1 intracellular domain (N1ICD)
interaction in Notch1-activated pancreatic cancer (142). In ESCC, USP10 is essential for
cisplatin resistance and migration through deubiquitination and
stabilization of integrin β1/YAP, highlighting USP10 inhibition as
a potential therapeutic approach (143). Therefore, these studies
underscore that USP10 is pivotal in tumor invasion, metastasis,
immunological modulation and chemoresistance. Nevertheless, the
detailed molecular mechanisms underlying its diverse functions
remain to be fully elucidated.
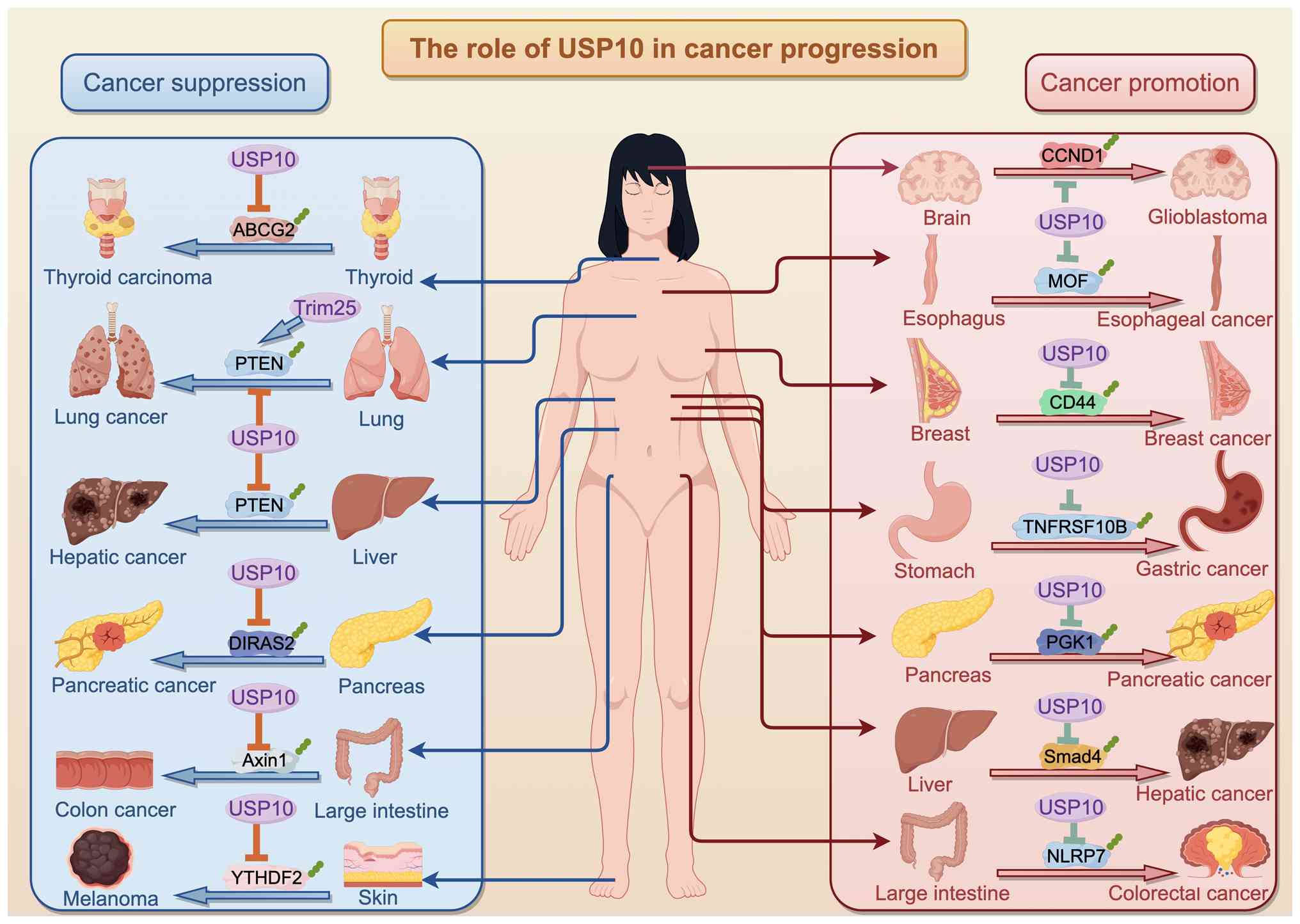
In cancer, USP10 primarily acts as an oncogene,
while it can also function as a tumor suppressor (Fig. 5). Lu et al (124) found that the expression of
USP10 was markedly downregulated in HCC tumor tissues compared with
adjacent non-cancer tissues, and in several independent cohorts,
low USP10 levels were associated with low patient survival.
Mechanistically, USP10 stabilizes AMPKα and phosphatase and tensin
homolog (PTEN) in HCC cells, thereby negatively regulating mTORC1
activation and AKT phosphorylation, ultimately inhibiting HCC
progression. In addition, this study also analyzed the correlation
between the high and low expression of USP10 and hepatitis B/C, TNM
stage, tumor size and Barcelona-Clinic Liver Cancer (BCLC) stage,
and found that only tumor size and BCLC staging were associated
with USP10 expression (124).
Additionally, as mentioned above, elevated USP10 expression has
been observed in metastatic or aggressive HCC, consistent with
experimental findings supporting a pro-metastatic role through the
stabilization of Smad4 (86).
Regrettably, current cohorts do not stratify USP10 expression by
HCC etiology [HBV/HCV/non-alcoholic steatohepatitis (NASH)] or by
molecular subtype, and no published study has systematically
evaluated USP10 across distinct TNM stages within defined etiologic
subgroups, which may be a novel direction for future research.
Similarly, USP10 activates PTEN by preventing tripartite
motif-containing 25-mediated K63-linked polyubiquitination,
suppressing the AKT/mTOR signaling pathway and inhibiting
proliferation in NSCLC (144).
Sun et al (145) further
demonstrated that USP10 suppresses lung cancer cell growth and
invasion by upregulating PTEN. In the thyroid cancer cell line
FTC133, low USP10 expression was observed; USP10 overexpression
activated PTEN, repressed PI3K/AKT signaling and downregulated
ABCG2, thereby suppressing doxorubicin-resistant thyroid cancer
invasion, migration and EMT (92).
Similarly, in CRC cells, USP10 suppresses metastasis
by stabilizing the zinc finger E-box binding homeobox 1 protein
(99). Luo et al
(146) showed that USP10
deubiquitinates and stabilizes YTH N6-methyladenosine RNA binding
protein F2 (YTHDF2), inhibiting melanoma proliferation and
migration. Disruption of the USP10-YTHDF2 interaction by lncRNA JPX
promotes YTHDF2 degradation, stabilizes BMP2 mRNA, activates AKT
phosphorylation and accelerates melanoma progression (146). Furthermore, N1DARP exerts
tumor-suppressive and chemosensitizing effects by modulating
USP10-Notch1 oncogenic signaling, suggesting a potential treatment
approach aimed at the N1DARP-N1ICD interaction in Notch1-activated
pancreatic cancer (142). In PC
cells, Chen et al (147)
indicated that USP10 regulated the stability of DIRAS family GTPase
2 through deubiquitylation, suppressing PC growth. In CRC, USP10
suppresses tumor growth primarily by facilitating phase separation,
and its expression is associated with clinical Wnt/β-catenin
signaling levels (148).
Overall, these results indicate that USP10 can act
as a context-dependent tumor suppressor, regulating multiple
pathways including PTEN, AKT/mTOR, Notch1 and Wnt/β-catenin to
inhibit tumor growth, invasion, metastasis and drug resistance.
In ischemic stroke, vagus nerve stimulation
alleviates neurological deficits, neuroinflammation and glial
activation by repressing the NF-κB signaling pathway, with USP10
likely playing a key role in this process (88). Additionally, USP10 suppresses
transforming growth factor β-activated kinase 1 (TAK1) signaling,
reducing inflammation and apoptosis, thereby mitigating brain
ischemic injury (74).
In neurodegenerative disorders such as Alzheimer's
disease (AD), characterized by tau protein aggregation in neurons,
USP10 participates in tau pathology. The RNA-binding protein TIA1
initiates tau aggregation by promoting SG formation. In the early
stages of AD and Parkinson's disease (PD), SG formation is common.
Piatnitskaia et al (149) demonstrated that USP10 is
essential for T-cell intracellular antigen 1 (TIA1)/tau-positive SG
assembly, and that catalytically inactive USP10 can still promote
tau/TIA1/USP10-positive SG formation, indicating a
deubiquitinase-independent, protein-interaction-driven role in SG
dynamics. By contrast, Wei et al (26) showed that USP10 directly
deubiquitinates tau, reducing its ubiquitination and turnover and
thereby promoting aggregation. Additionally, Cai et al
(150) suggested that USP10 may
also interact with β-site amyloid precursor protein cleaving enzyme
1, affecting hippocampal volume and contributing to AD
pathogenesis. In PD, α-synuclein is a major pathogenic factor.
Anisimov et al (151)
found that USP10 inhibits chaperone-mediated autophagy, suppressing
α-synuclein degradation and slowing PD progression.
In amyotrophic lateral sclerosis (ALS), USP10
facilitates the clearance of TAR DNA-binding protein 43
(TDP)-43/TDP-35-positive SGs while promoting proper aggregate
formation, thereby reducing cytoplasmic TDP-43/TDP-35
mis-accumulation and neuronal toxicity (152). Pang et al (107) further demonstrated that the
lncRNA NR3C2-8:1 upregulates USP10 and activates p53, promoting
p53-mediated apoptosis in ALS. Conversely, lnc-NR3C knockdown
suppresses the activation of p53 that is mediated by USP10,
therefore shielding cells from oxidative damage.
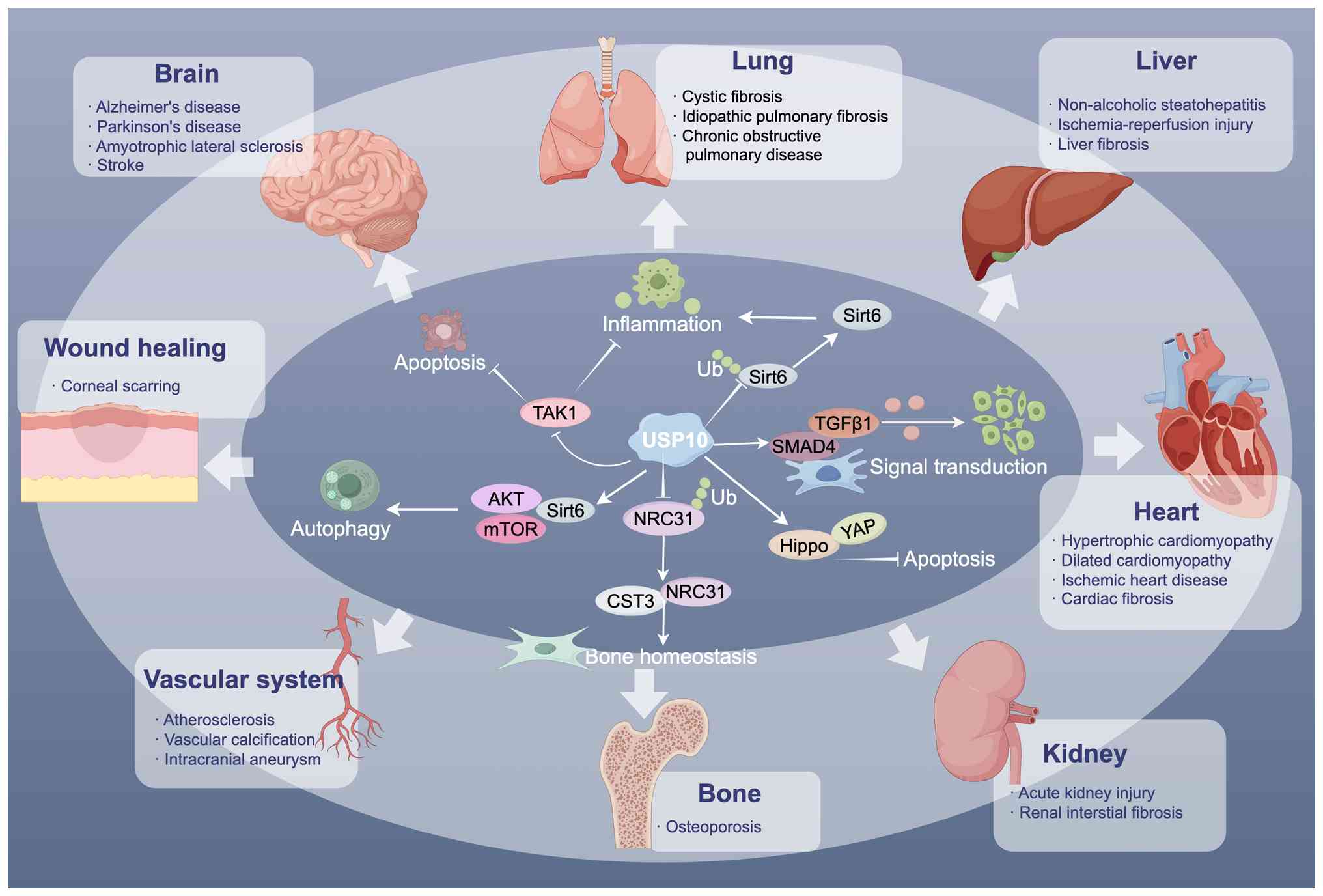
Beyond its roles in cancer, USP10 also serves
critical roles in cardiovascular, renal, hepatic and pulmonary
diseases (Fig. 6). This section
will examine the mechanisms and functions of USP10 in these
non-cancer pathologies.
Recently, growing findings have revealed that USP10
serves critical roles in various cardiovascular pathologies,
including hypertrophic cardiomyopathy (HCM), dilated cardiomyopathy
(DCM), ischemic heart disease and vascular remodeling. In HCM,
USP10 expression is upregulated and correlates with impaired
cardiac contractile function. Mechanistically, USP10
deubiquitinates Sirt6, thereby repressing the Akt signaling pathway
and attenuating cardiomyocyte hypertrophy (153). Li et al (95) further confirmed that USP10 exerts
protective effects in pressure overload-induced pathological
hypertrophy. Specifically, USP10 interacts with cytoplasmic Mfn2
via His-679 in its UCH domain, removing K11/K48-linked ubiquitin
chains to prevent proteasomal degradation, thereby maintaining
mitochondrial function and cellular homeostasis.
In diabetic cardiomyopathy, USP10 activates Notch1,
protecting against myocardial injury in type 2 diabetic mice
(154). In models of
hypoxia/reoxygenation, USP10 protects H9C2
cells from apoptosis and oxidative stress by activating the
Hippo/YAP pathway, while FOXO4 acts as a negative transcriptional
regulator of USP10 to suppress Hippo/YAP activation (91). By contrast, in DCM, USP10
promotes endothelial-to-mesenchymal transition (EndMT) via
SMAD4/TGF-β1 signaling, exacerbating disease progression, whereas
EGF like and discoidin domains 3 deficiency attenuates EndMT by
inhibiting USP10-dependent Smad4 deubiquitination (155). In ischemic cardiomyopathy,
USP10 in cardiac fibroblasts deubiquitinates SMAD4 to activate
TGF-β/SMAD4 signaling, promoting cardiac fibrosis (156). Additionally, USP10 expression
is upregulated in doxorubicin-treated mouse hearts, H9C2 and HL-1
cells when Ca2+/calmodulin-dependent protein kinase is
inhibited; this protective regulation against apoptosis and
ubiquitination is blocked by the USP10 inhibitor Spautin-1
(157). Heat shock protein A12A
acts as a scaffold for USP10 and p53, promoting USP10-mediated
deubiquitination of p53, stabilizing p53 and inhibiting
p53-mediated glycolysis, thereby alleviating cardiac fibrosis
(158).
USP10 also regulates vascular signaling pathways
beyond the heart. It deubiquitinates and stabilizes the
transcriptional coactivator Yorkie, thereby modulating the Hippo
pathway (90). In retinal
endothelial cells, USP10 stabilizes the intracellular domain of
Notch1, regulating Notch signaling and having an effect on retinal
angiogenesis and vascular homeostasis (85). In pulmonary vascular cells, USP10
inhibits BUB3 mitotic checkpoint protein (BUB3) ubiquitination,
increasing BUB3 stability, which drives cell growth and survival
via non-canonical Hippo/mammalian Ste20-like kinases signaling
involving BUB3 and FOXO (159).
Vascular smooth muscle cells (VSMCs) are also influenced by USP10.
In ligated carotid arteries, USP10 expression is elevated,
stabilizing Skp2 and promoting VSMC proliferation and migration,
thus exacerbating neointimal formation (160). In diabetes-associated vascular
calcification and atherosclerosis, USP10 mediates AMPKα
ubiquitination, increasing Thr172 phosphorylation and aggravating
vascular calcification (161).
Recently, a study revealed that lncRNAs interact with USP10 and
KLF4 to enhance NLRP3 transcription, driving pyroptosis in human
brain VSMCs and contributing to intracranial aneurysm progression
(162).
Renal interstitial fibrosis is a major determinant
of chronic kidney disease progression, with renal tubular
epithelial cells serving as key drivers of this pathological
process. Liu et al (167) reported that USP10 facilitates
renal interstitial fibrosis by deubiquitinating and stabilizing
p53, which enhances its nuclear translocation and upregulates p21
expression, ultimately driving fibrotic progression. Knockout of
USP10 or treatment with Spautin-1, an inhibitor of USP10
deubiquitinase activity, markedly reduces fibronectin expression
and ameliorates TGF-β1-induced tubular epithelial cell
dedifferentiation (167). USP10
also deubiquitinates and stabilizes FOXQ1, which exerts protective
effects against inflammation and apoptosis in sepsis-associated
acute kidney injury (AKI) via the cAMP response element binding
5/NF-κB axis (168).
Additionally, PR/SET domain 16 attenuates rhabdomyolysis-induced
AKI by upregulating USP10 to inhibit ferroptosis (169). Similarly, USP10 modulates
Sirt6-mediated Nrf2/antioxidant response element signaling, thereby
alleviating sepsis-induced AKI (170).
Idiopathic pulmonary fibrosis is a progressive
pulmonary disorder marked by fibroblast activation and collagen
accumulation, with limited effective therapeutic interventions. Mao
et al (171) reported
that USP10 was markedly downregulated in bleomycin-induced
pulmonary fibroblasts. Overexpression of USP10 mitigates lung
injury and reduces collagen deposition. Mechanistically, USP10
integrates with Sirt6 to promote Sirt6/AKT/mTOR-mediated autophagy,
thereby alleviating lung fibrosis.
Cystic fibrosis transmembrane conductance regulator
(CFTR) is a cyclic AMP-regulated chloride channel critical for
regulating airway surface liquid volume, facilitating mucociliary
clearance and pathogen elimination. USP10 has been shown to
regulate CFTR deubiquitination, affecting CFTR endocytic recycling
in human airway epithelial cells (172,173). Cif (PA2934), a bacterial toxin
secreted by Pseudomonas aeruginosa in outer membrane
vesicles, reduces CFTR-mediated chloride secretion, which is
essential for mucociliary clearance (174). Bomberger et al (175) demonstrated that Cif enhanced
the interaction between G3BP1 and USP10, inhibiting USP10-mediated
CFTR deubiquitination and increasing CFTR lysosomal degradation.
These studies suggest that USP10 exerts a dual effect in lung
disease, both by modulating autophagy in fibrotic processes and by
regulating CFTR stability to maintain airway epithelial
function.
USP10 has been shown to promote fibrotic wound
healing by regulating integrin β1 and β5 (176). Conversely, USP10 overexpression
can block TGF-β signaling or αv integrins on the cell surface,
preventing or reducing the expression of fibrotic markers (176). In injured corneal fibroblasts,
Boumil et al (177)
demonstrated that USP10 knockdown can prevent corneal scarring.
Beyond wound healing, USP10 exerts an important function in viral
infection. Kaposi's sarcoma-related herpesvirus encodes viral
interferon regulatory factor 1 (vIRF1), which undergoes lysine
acetylation necessary to effectively inhibit IFN-β production and
antiviral signaling. vIRF1 blocks the interaction between SIRT6 and
USP10, resulting in vIRF1 degradation via the ubiquitin-proteasome
pathway and promoting its own acetylation. Notably, vIRF1
acetylation is required for its ability to block IRF3-CREB-binding
protein/p300 recruitment and suppress the stimulator of
IFN-dependent genes DNA-sensing pathway (178). These results point to USP10 as
a crucial modulator of tissue repair and viral immune evasion,
suggesting potential therapeutic applications in fibrosis and viral
infections.
Over the past decades, numerous small-molecule
inhibitors that target DUBs have been developed and tested in
preclinical studies, showing notable therapeutic potential,
particularly in oncology (179,180). As previously discussed, USP10
is markedly upregulated in various malignancies and is associated
with poor survival outcomes, making it a potential target for
oncological treatment. Other USP10 inhibitors have recently been
developed for human cancers, including Spautin-1, P22077, HBX19818,
Wu-5 and D1.
Spautin-1 is a small-molecule inhibitor
non-selectively targeting USP10 and USP13, which was reported by
Liu et al (68) in 2011.
Furthermore, Liu et al (68) also confirmed that Spautin-1 had
no significant inhibitory effect on USP14 and CYLD. Notably, the
impact of other DUBs (such as USP7) has not been systematically
evaluated, which may cause the risk of off-target effects. A recent
study showed that Spautin-1 can attenuate GBM progression by
independently modulating RAF-ERK-mediated glycolysis and SKP2
(181). Similarly, it also
inhibits EGFR phosphorylation and downstream signaling, suppressing
prostate cancer progression (182). Furthermore, in combination with
the chemotherapeutic agent cisplatin, Spautin-1 notably enhances
antitumor activity and reduces osteosarcoma development (36). Of note, a recent study indicated
that Spautin-1 promotes mitophagy via the PTEN induced kinase
1-parkin RBR E3 ubiquitin protein ligase pathway, improving
associative learning in an AD C. elegans model (183).
Other inhibitors include Ly-2, a high-affinity
USP10 inhibitor that promotes apoptosis through cyclin-dependent
kinase 4 downregulation in HCC. A study by Lu et al
(185) revealed that Ly-2 is
the first USP10 inhibitor to achieve nanomolar-level binding
affinity in cells and in vitro. In this study, a Chai-1
model and sites mutation experiment confirmed that Ly2 binds to
USP10 through the interaction between Ly-2 with Phe629 and Thr630.
Compared with D1, Ly-2 showed notable affinity. Regrettably, the
selectivity of Ly-2 for other DUBs was not evaluated; therefore,
its specificity remains to be determined. Weisberg et al
(186) found that HBX19818 and
P22077 could inhibit USP10 (IC50=6 μM), but also inhibit
USP7 (IC50=10 μM). In patient-derived xenograft models,
P22077 exhibits selective antiproliferative effects against mutant
FMS-like tyrosine kinase-3 (FLT3). Wu-5, another novel USP10
inhibitor, shows high selectivity for USP10. Yu et al
(187) demonstrated that Wu-5
targets FLT3 and AMPKα pathways to overcome FLT3 inhibitor
resistance and synergistically enhance the anti-AML effects of
crenolanib in vitro. Furthermore, the study confirmed that
Wu-5 inhibited the binding of HA-Ub-VS to USP10 but not to USP5
through DUB labeling. These findings indicate that Wu-5 directly
interacts with USP10 in vitro. Notably, this study did not
carry out in vivo experiments, and its toxic effects (such
as hepatoxicity) need to be further clarified.
Several natural compounds have also been identified
to modulate USP10, offering alternative therapeutic strategies. For
instance, ginkgolic acid stabilizes YAP1 via USP10, enhancing
YAP1/TEA domain transcription factor 4-mediated transcription of
prolyl 4-hydroxylase subunit alpha 1 and suppressing HCC
progression (188). The
macrolide compound FW-04-806 (F806), with potential against ESCC,
targets USP10 to promote ANLN degradation and inhibit mitosis
(189). Acevaltrate inhibits
USP10-mediated cyclin D (CCND1) demethylation, inducing CCND1
degradation and suppressing GBM progression (117). Acacetin enhances cardiac
remodeling by inhibiting USP10-mediated Beclin1 ubiquitination and
autophagy in cardiomyocytes (190), whereas limonin activates
USP10-mediated SIRT6 ubiquitination and degradation, offering a
potential target for cardiac hypertrophy (191). Tetrahydroxy stilbene glucoside
promotes USP10-mediated YBX1 stabilization, enhancing mitophagy and
alleviating neuronal injury after cerebral I/R (192). Additionally, melatonin
suppresses ESCC proliferation by attenuating the histone
deacetylase HDAC7/β-catenin/c-Myc positive feedback loop and
reducing USP10-stabilized HDAC7 protein (54). Notably, whether these natural
compounds have selective inhibitory effects on USP10 has not been
studied, yet.
The development of selective USP10 inhibitors
remains challenging owing to the high structural conservation of
catalytic domains across the USP family. Most compounds reported to
modulate USP10 activity are therefore regarded as broad-spectrum
USP inhibitors or indirect regulators. Despite the challenges in
DUB inhibitor development, two DUB-targeting small molecules,
VLX1570 and KSQ-4279, have been advanced to phase I clinical trials
for refractory MM (NCT02372240 and NCT05240898) (193,194). Collectively, these findings
underscore the therapeutic promise of DUB inhibitors, particularly
USP10-targeted agents, in treating cancer and other diseases
(Table II).
USP10, a multifunctional deubiquitinase,
participates in a wide range of physiological processes, including
DNA damage repair, immune and inflammatory regulation,
environmental adaptation and autophagy. Its ability to modulate
diverse substrates, such as p53, AMPK, Beclin1 and ATM, highlights
its critical role as a dynamic regulator of cellular stress
responses. Furthermore, USP10 acts as a pivotal node in multiple
signaling pathways, contributing to both tumor progression and
suppression. A broad spectrum of diseases, including cancer,
neurodegenerative and infectious diseases and organ fibrosis, have
been linked to USP10 dysregulation.
Decades of research have driven the development of
numerous USP10-targeting inhibitors. As a result of the notable
therapeutic potential that has been established in preclinical
research conducted on illness models, there is a rising interest in
techniques that target USP10. Nevertheless, the context-dependent
functions of USP10, particularly its dual roles in tumor promotion
and suppression, necessitate careful consideration of disease
states and compensatory signaling mechanisms. Furthermore, for a
specific cancer, the association between USP10 and the etiology
should be further clarified, which may be beneficial to cancer
prevention and diagnosis. At present, numerous inhibitors targeting
DUBs are still in preclinical research, while there is a lack of
in vivo experiments, and the evaluation of the toxic and
side effects of inhibitors is still lacking. Broad USP10 inhibition
may be beneficial in certain cancers or fibrotic conditions but
could be detrimental in scenarios where USP10 supports genome
integrity or immune defense. Several key challenges must be
addressed to translate USP10-targeted therapies into clinical
practice: i) USP10 shares high structural similarity with other
USPs, particularly USP13. Dual inhibition may confound mechanistic
interpretation and therapeutic outcomes. Developing highly
selective USP10 inhibitors remains a priority, necessitating
further structural and mechanistic studies to map its molecular
interactions comprehensively. Notably, there are no reports on the
differences between USP10 and USP13 in catalytic cleft,
thumb-finger interface and ubiquitin binding surface; therefore,
further research should highlight these differences so that
specific USP10 inhibitors are designed. While high-resolution
experimental structures of full-length USP10 or its catalytic
domain remain inaccessible, recent advances in structural modeling,
including AlphaFold-derived predictions, may provide useful help
for the structural elucidation of USP10 (195). These differences may form
unique inhibitor-binding pockets, offering an opportunity for
structure-guided development of next-generation USP10-selective
inhibitors; ii) embedded within complex signaling networks, USP10
inhibition could trigger compensatory upregulation of other DUBs or
pathway rewiring, potentially reducing efficacy or promoting
resistance; iii) current inhibitors, such as Spautin-1, exhibit
moderate potency, suboptimal pharmacokinetics and poor tissue
distribution. Optimizing drug delivery, bioavailability and
metabolic stability is essential; and iv) the cell-type specificity
of USP10 across disease contexts is still unclear.
Future directions should focus on developing highly
selective and potent USP10 inhibitors, elucidating its substrate
landscape and interactions in specific disease contexts, and
integrating USP10 modulation into rational combination therapies.
For instance, pairing USP10 inhibitors with chemotherapy,
immunotherapy or targeted agents may enhance antitumor efficacy and
overcome compensatory resistance mechanisms. Additionally,
leveraging structural biology, single-cell proteomics and chemical
biology approaches will be crucial for fully delineating regulatory
complexity of USP10 and providing a robust foundation for drug
discovery and clinical translation.
Not applicable.
LZ was involved in literature search, visualization
and writing-original draft. HS provided resources and supervision
and performed visualization. ZiW was involved in visualization and
software. ZhW conceptualized the study and provided resources. QM
performed project administration and review & editing. YL
provided supervision, acquired funding and performed review &
editing. Data authentication is not applicable. All authors have
read and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
This work was supported by Hubei Chen Xiaoping Science and
Technology Development Foundation (grant no. CXPJJH122011-011) and
the Wu JiePing Medical Foundation (grant no.
320.6750.2023-18-21).
|
1
|
Swatek KN and Komander D: Ubiquitin
modifications. Cell Res. 26:399–422. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Popovic D, Vucic D and Dikic I:
Ubiquitination in disease pathogenesis and treatment. Nat Med.
20:1242–1253. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Pohl C and Dikic I: Cellular quality
control by the ubiquitin-proteasome system and autophagy. Science.
366:818–822. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhao J, Zhai B, Gygi SP and Goldberg AL:
mTOR inhibition activates overall protein degradation by the
ubiquitin proteasome system as well as by autophagy. Proc Natl Acad
Sci USA. 112:15790–15797. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ren J, Yu P, Liu S, Li R, Niu X, Chen Y,
Zhang Z, Zhou F and Zhang L: Deubiquitylating enzymes in cancer and
immunity. Adv Sci (Weinh). 10:e23038072023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chau V, Tobias JW, Bachmair A, Marriott D,
Ecker DJ, Gonda DK and Varshavsky A: A multiubiquitin chain is
confined to specific lysine in a targeted short-lived protein.
Science. 243:1576–1583. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Madiraju C, Novack JP, Reed JC and
Matsuzawa SI: K63 ubiquitination in immune signaling. Trends
Immunol. 43:148–162. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tracz M and Bialek W: Beyond K48 and K63:
Non-canonical protein ubiquitination. Cell Mol Biol Lett. 26:12021.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yuan WC, Lee YR, Lin SY, Chang LY, Tan YP,
Hung CC, Kuo JC, Liu CH, Lin MY, Xu M, et al: K33-linked
polyubiquitination of coronin 7 by Cul3-KLHL20 ubiquitin E3 ligase
regulates protein trafficking. Mol Cell. 54:586–600. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen YJ, Wu H and Shen XZ: The
ubiquitin-proteasome system and its potential application in
hepatocellular carcinoma therapy. Cancer Lett. 379:245–252. 2016.
View Article : Google Scholar
|
|
11
|
Mevissen TET and Komander D: Mechanisms of
deubiquitinase specificity and regulation. Annu Rev Biochem.
86:159–192. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim SY and Baek KH: TGF-β signaling
pathway mediated by deubiquitinating enzymes. Cell Mol Life Sci.
76:653–665. 2019. View Article : Google Scholar
|
|
13
|
Serratore V, Lucibello M, Malanga D,
Viglietto G and De Marco C: AKT and DUBs: A bidirectional
relationship. Cell Mol Biol Lett. 30:772025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dewson G, Eichhorn PJA and Komander D:
Deubiquitinases in cancer. Nat Rev Cancer. 23:842–862. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sharma A, Alswillah T, Singh K, Chatterjee
P, Willard B, Venere M, Summers MK and Almasan A: USP14 regulates
DNA damage repair by targeting RNF168-dependent ubiquitination.
Autophagy. 14:1976–1990. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhu X, Wang P, Zhan X, Zhang Y, Sheng J,
He S, Chen Y, Nie D, You X, Mai H, et al: USP1-regulated reciprocal
differentiation of Th17 cells and Treg cells by deubiquitinating
and stabilizing TAZ. Cell Mol Immunol. 20:252–263. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Reyes-Turcu FE, Ventii KH and Wilkinson
KD: Regulation and cellular roles of ubiquitin-specific
deubiquitinating enzymes. Annu Rev Biochem. 78:363–397. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kim GW, Cha M, Ong HTM, Yoo J, Jeon YH,
Lee SW, Oh SY, Kang MJ, Kim Y and Kwon SH: HDAC6 and USP9X control
glutamine metabolism by stabilizing GS to promote glioblastoma
tumorigenesis. Adv Sci (Weinh). 12:e25015532025. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qian C, Hu C, Xu Y, Xu W, Wang Z, Gan W,
Tang W, Huang L, Samorodov AV and Wang Y: Intestinal
epithelial-derived USP13 alleviates colonic inflammation by
suppressing GRP78-mediated endoplasmic reticulum stress. Adv Sci
(Weinh). 12:e007412025. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kang S, Liu S, Dong X, Li H, Qian Y, Dai
A, He W, Li X, Chen Q, Wang H and Ding PH: USP4 depletion-driven
RAB7A ubiquitylation impairs autophagosome-lysosome fusion and
aggravates periodontitis. Autophagy. 21:771–788. 2025. View Article : Google Scholar :
|
|
21
|
Zhao Y, Wang F, Gao L, Xu L, Tong R, Lin
N, Su Y, Yan Y, Gao Y, He J, et al: Ubiquitin-specific protease 4
is an endogenous negative regulator of metabolic dysfunctions in
nonalcoholic fatty liver disease in mice. Hepatology. 68:897–917.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li D and Ma Q: Ubiquitin-specific
protease: An emerging key player in cardiomyopathy. Cell Commun
Signal. 23:1432025. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ye B, Zhou H, Chen Y, Luo W, Lin W, Zhao
Y, Han J, Han X, Huang W, Wu G, et al: USP25 ameliorates
pathological cardiac hypertrophy by stabilizing SERCA2a in
cardiomyocytes. Circ Res. 132:465–480. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Su JB, Chen G, Sun QY, Fu X, Wu LX, Qiu
HB, Lv ZL, Hu JY, Wang Y, Zhuang YY, et al: USP48 protects against
myocardial ischemia-reperfusion injury by stabilizing and
upregulating CNN1 in type 1 diabetes mice. Metabolism.
170:1563262025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gao B, Jing Y, Li X and Cong S: Ubiquitin
specific peptidase 11 knockdown slows Huntington's disease
progression via regulating mitochondrial dysfunction and neuronal
damage depending on PTEN-mediated AKT pathway. Mol Med. 31:72025.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wei Z, Zeng K, Hu J, Li X, Huang F, Zhang
B, Wang JZ, Liu R, Li HL and Wang X: USP10 deubiquitinates tau,
mediating its aggregation. Cell Death Dis. 13:7262022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang XW, Feng N, Liu YC, Guo Q, Wang JK,
Bai YZ, Ye XM, Yang Z, Yang H, Liu Y, et al: Neuroinflammation
inhibition by small-molecule targeting USP7 noncatalytic domain for
neurodegenerative disease therapy. Sci Adv. 8:eabo07892022.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu F, Chen J, Li K, Li H, Zhu Y, Zhai Y,
Lu B, Fan Y, Liu Z, Chen X, et al: Ubiquitination and
deubiquitination in cancer: From mechanisms to novel therapeutic
approaches. Mol Cancer. 23:1482024. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Young MJ, Hsu KC, Lin TE, Chang WC and
Hung JJ: The role of ubiquitin-specific peptidases in cancer
progression. J Biomed Sci. 26:422019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Feng T, Ling S, Xu C, Ying L, Su D and Xu
X: Ubiquitin-specific peptidase 22 in cancer. Cancer Lett.
514:30–37. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li S, Song Y, Wang K, Liu G, Dong X, Yang
F, Chen G, Cao C, Zhang H, Wang M, et al: USP32 deubiquitinase:
Cellular functions, regulatory mechanisms, and potential as a
cancer therapy target. Cell Death Discov. 9:3382023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Soncini C, Berdo I and Draetta G: Ras-GAP
SH3 domain binding protein (G3BP) is a modulator of USP10, a novel
human ubiquitin specific protease. Oncogene. 20:3869–3879. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li H, Li C, Zhai W, Zhang X, Li L, Wu B,
Yu B, Zhang P, Li J, Cui CP and Zhang L: Destabilization of TP53 by
USP10 is essential for neonatal autophagy and survival. Cell Rep.
41:1114352022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yuan J, Luo K, Zhang L, Cheville JC and
Lou Z: USP10 regulates p53 localization and stability by
deubiquitinating p53. Cell. 140:384–396. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jia R and Bonifacino JS: The ubiquitin
isopeptidase USP10 deubiquitinates LC3B to increase LC3B levels and
autophagic activity. J Biol Chem. 296:1004052021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Feng Z, Ou Y, Deng X, Deng M, Yan X, Chen
L, Zhou F and Hao L: Deubiquitinase USP10 promotes osteosarcoma
autophagy and progression through regulating GSK3β-ULK1 axis. Cell
Biosci. 14:1112024. View Article : Google Scholar
|
|
37
|
Zhang M, Hu C, Tong D, Xiang S, Williams
K, Bai W, Li GM, Bepler G and Zhang X: Ubiquitin-specific peptidase
10 (USP10) deubiquitinates and stabilizes MutS homolog 2 (MSH2) to
regulate cellular sensitivity to DNA damage. J Biol Chem.
291:10783–10791. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu X, Zhang S, An Y, Xu B, Yan G and Sun
M: USP10/XAB2/ANXA2 axis promotes DNA damage repair to enhance
chemoresistance to oxaliplatin in colorectal cancer. J Exp Clin
Cancer Res. 44:942025. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liu J, Zhang S, Cao L, Zhang N, Guo Q, Zou
Y, Yang R, Dong S, Zheng L, Xiao Y, et al: The
deubiquitination-PARylation positive feedback loop of the
USP10-PARP1 axis promotes DNA damage repair and affects therapeutic
efficacy of PARP1 inhibitor. Oncogene. 44:2515–2529. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Deng M, Yang X, Qin B, Liu T, Zhang H, Guo
W, Lee SB, Kim JJ, Yuan J, Pei H, et al: Deubiquitination and
activation of AMPK by USP10. Mol Cell. 61:614–624. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Quan G, Xu J, Wang J, Liu X, Xu J and
Jiang J: KIF15 is essential for USP10-mediated PGK1
deubiquitination during the glycolysis of pancreatic cancer. Cell
Death Dis. 14:1372023. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zeng Z, Wu HX, Zhan N, Huang YB, Wang ZS,
Yang GF, Wang P and Fu GH: Prognostic significance of USP10 as a
tumor-associated marker in gastric carcinoma. Tumour Biol.
35:3845–3853. 2014. View Article : Google Scholar
|
|
43
|
Jumper J, Evans R, Pritzel A, Green T,
Figurnov M, Ronneberger O, Tunyasuvunakool K, Bates R, Žídek A,
Potapenko A, et al: Highly accurate protein structure prediction
with AlphaFold. Nature. 596:583–589. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Varadi M, Anyango S, Deshpande M, Nair S,
Natassia C, Yordanova G, Yuan D, Stroe O, Wood G, Laydon A, et al:
AlphaFold protein structure database: Massively expanding the
structural coverage of protein-sequence space with high-accuracy
models. Nucleic Acids Res. 50(D1): D439–D444. 2022. View Article : Google Scholar :
|
|
45
|
Komander D, Clague MJ and Urbé S: Breaking
the chains: Structure and function of the deubiquitinases. Nat Rev
Mol Cell Biol. 10:550–563. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ye Y, Scheel H, Hofmann K and Komander D:
Dissection of USP catalytic domains reveals five common insertion
points. Mol Biosyst. 5:1797–1808. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zeng Z, Zhou Z, Zhan N, Yuan J, Ye B, Gu
L, Wang J, Jian Z and Xiong X: USP10 expression in normal adrenal
gland and various adrenal tumors. Endocr Pathol. 26:302–308. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yang JR, Lu YB, Su HX, Xiao Y, Pan Q, Su
F, Zhang XB, Zhu KL, Guan QL and Ling XL: USP10 promotes the
progression of triple-negative breast cancer by enhancing the
stability of TCF4 protein. Biochem Pharmacol. 218:1158642023.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Du X, Yu R, Yan C, Dong P, Wei C, Wang B,
Zhang C, He Y, Wei Y, Han L and Sun J: USP10 promotes the
progression and attenuates gemcitabine chemotherapy sensitivity via
stabilizing PLK1 in PDAC. Cell Death Dis. 16:4492025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Li B, Qi ZP, He DL, Chen ZH, Liu JY, Wong
MW, Zhang JW, Xu EP, Shi Q, Cai SL, et al: NLRP7 deubiquitination
by USP10 promotes tumor progression and tumor-associated macrophage
polarization in colorectal cancer. J Exp Clin Cancer Res.
40:1262021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Takayama KI, Suzuki T, Fujimura T,
Takahashi S and Inoue S: Association of USP10 with G3BP2 inhibits
p53 signaling and contributes to poor outcome in prostate cancer.
Mol Cancer Res. 16:846–856. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hu C, Zhang M, Moses N, Hu CL, Polin L,
Chen W, Jang H, Heyza J, Malysa A, Caruso JA, et al: The
USP10-HDAC6 axis confers cisplatin resistance in non-small cell
lung cancer lacking wild-type p53. Cell Death Dis. 11:3282020.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Qiu W, Xiao Z, Yang Y, Jiang L, Song S, Qi
X, Chen Y, Yang H, Liu J and Chu L: USP10 deubiquitinates RUNX1 and
promotes proneural-to-mesenchymal transition in glioblastoma. Cell
Death Dis. 14:2072023. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Ma ZQ, Feng YT, Guo K, Liu D, Shao CJ, Pan
MH, Zhang YM, Zhang YX, Lu D, Huang D, et al: Melatonin inhibits
ESCC tumor growth by mitigating the HDAC7/β-catenin/c-Myc positive
feedback loop and suppressing the USP10-maintained HDAC7 protein
stability. Mil Med Res. 9:542022.
|
|
55
|
Zhang D, Wang X, Lu S, Gao Y, Zhu G, Li G,
Yu Z, Hou J, Yan H, Yuan W, et al: USP10 inhibits ferroptosis via
deubiquinating POLR2A in head and neck squamous cell carcinoma. Adv
Sci (Weinh). 12:e122712025. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Dodgson SE, Santaguida S, Kim S, Sheltzer
J and Amon A: The pleiotropic deubiquitinase Ubp3 confers
aneuploidy tolerance. Genes Dev. 30:2259–2271. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Bhattacharya U, Neizer-Ashun F, Mukherjee
P and Bhattacharya R: When the chains do not break: The role of
USP10 in physiology and pathology. Cell Death Dis. 11:10332020.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Luo Y, Zhang X, Chen R, Li R, Liu Y, Zhang
J, Liu Q, Si M, Liu J, Wu B, et al: USP10 regulates B cell response
to SARS-CoV-2 or HIV-1 nanoparticle vaccines through
deubiquitinating AID. Signal Transduct Target Ther. 7:72022.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Drew Y, Zenke FT and Curtin NJ: DNA damage
response inhibitors in cancer therapy: Lessons from the past,
current status and future implications. Nat Rev Drug Discov.
24:19–39. 2025. View Article : Google Scholar
|
|
60
|
Li S, Xiong S, Li Z, Yang L, Yang H, Xiong
J, Pan W, Guo J, Xu S and Fu B: USP3 promotes DNA damage response
and chemotherapy resistance through stabilizing and
deubiquitinating SMARCA5 in prostate cancer. Cell Death Dis.
15:7902024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Su D, Ma S, Shan L, Wang Y, Wang Y, Cao C,
Liu B, Yang C, Wang L, Tian S, et al: Ubiquitin-specific protease 7
sustains DNA damage response and promotes cervical carcinogenesis.
J Clin Invest. 128:4280–4296. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Li Y, Luo K, Yin Y, Wu C, Deng M, Li L,
Chen Y, Nowsheen S, Lou Z and Yuan J: USP13 regulates the
RAP80-BRCA1 complex dependent DNA damage response. Nat Commun.
8:157522017. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Sharma A and Almasan A: USP14 regulates
DNA damage response and is a target for radiosensitization in
non-small cell lung cancer. Int J Mol Sci. 21:63832020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Zhang D, Zaugg K, Mak TW and Elledge SJ: A
role for the deubiquitinating enzyme USP28 in control of the
DNA-damage response. Cell. 126:529–542. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Zhu Y, Chen Z, Niu K, Li M, Deng Y, Zhang
J, Wei D, Wang J and Zhao Y: USP33 regulates DNA damage response
and carcinogenesis through deubiquitylating and stabilising p53.
Cell Prolif. 58:e137932025. View Article : Google Scholar :
|
|
66
|
Wu J, Chen Y, Geng G, Li L, Yin P,
Nowsheen S, Li Y, Wu C, Liu J, Zhao F, et al: USP39 regulates DNA
damage response and chemo-radiation resistance by deubiquitinating
and stabilizing CHK2. Cancer Lett. 449:114–124. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Zhao X, Ma Y, Li J, Sun X, Sun Y, Qu F,
Shi X, Xie Y, Liu S, Ma Y, et al: The AEG-1-USP10-PARP1 axis
confers radioresistance in esophageal squamous cell carcinoma via
facilitating homologous recombination-dependent DNA damage repair.
Cancer Lett. 577:2164402023. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Liu J, Xia H, Kim M, Xu L, Li Y, Zhang L,
Cai Y, Norberg HV, Zhang T, Furuya T, et al: Beclin1 controls the
levels of p53 by regulating the deubiquitination activity of USP10
and USP13. Cell. 147:223–234. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Song D, Kuang L, Yang L, Wang L, Li H, Li
X, Zhu Z, Shi C, Zhu H and Gong W: Yin and yang regulation of
stress granules by Caprin-1. Proc Natl Acad Sci USA.
119:e22079751192022. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Tahmasebinia F, Tang Y, Tang R, Zhang Y,
Bonderer W, de Oliveira M, Laboret B, Chen S, Jian R, Jiang L, et
al: The 40S ribosomal subunit recycling complex modulates
mitochondrial dynamics and endoplasmic reticulum-mitochondria
tethering at mitochondrial fission/fusion hotspots. Nat Commun.
16:10212025. View Article : Google Scholar
|
|
71
|
Faus H, Meyer HA, Huber M, Bahr I and
Haendler B: The ubiquitin-specific protease USP10 modulates
androgen receptor function. Mol Cell Endocrinol. 245:138–146. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Liu YL, Liu JY, Zhu XX, Wei JH, Mi SL, Liu
SY, Li XL, Zhang WW, Zhao LL, Wang H, et al: Pubertal exposure to
Microcystin-LR arrests spermatogonia proliferation by inducing DSB
and inhibiting SIRT6 dependent DNA repair in vivo and in vitro.
Ecotoxicol Environ Saf. 274:1161912024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Wang X, Fan X, Zhang J, Wang F, Chen J,
Wen Y, Wang L, Li T, Li H, Gu H, et al: hnRNPA2B1 represses the
disassembly of arsenite-induced stress granules and is essential
for male fertility. Cell Rep. 43:1137692024. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Wang L, Wu D and Xu Z: USP10 protects
against cerebral ischemia injury by suppressing inflammation and
apoptosis through the inhibition of TAK1 signaling. Biochem Biophys
Res Commun. 516:1272–1278. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Luo P, Qin C, Zhu L, Fang C, Zhang Y,
Zhang H, Pei F, Tian S, Zhu XY, Gong J, et al: Ubiquitin-specific
peptidase 10 (USP10) inhibits hepatic steatosis, insulin
resistance, and inflammation through Sirt6. Hepatology.
68:1786–1803. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Pan L, Chen Z, Wang L, Chen C, Li D, Wan
H, Li B and Shi G: Deubiquitination and stabilization of T-bet by
USP10. Biochem Biophys Res Commun. 449:289–294. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Shi Y, Yuan B, Zhu W, Zhang R, Li L, Hao
X, Chen S and Hou F: Ube2D3 and Ube2N are essential for
RIG-I-mediated MAVS aggregation in antiviral innate immunity. Nat
Commun. 8:151382017. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Liu F, Zhuang W, Song B, Yang Y, Liu J,
Zheng Y, Liu B, Zheng J, Zhao W and Gao C: MAVS-loaded unanchored
Lys63-linked polyubiquitin chains activate the RIG-I-MAVS signaling
cascade. Cell Mol Immunol. 20:1186–1202. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Zhou L, Mu S, Zhang Y and Song H:
USP10-mediated deubiquitination of NR3C1 regulates bone homeostasis
by controlling CST3 expression. Biochem Pharmacol. 229:1165192024.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Liu Y and Zhou Y: Circ_0087960 stabilizes
KDM5B by reducing SKP2 mediated ubiquitination degradation and
promotes osteogenic differentiation in periodontal ligament stem
cells. Regen Ther. 19:122–130. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wang H, Liu Z, Niu D, Li H, Han Y, Peng J
and Qian Q: Carbamazepine regulates USP10 through miR-20a-5p to
affect the deubiquitination of SKP2 and inhibit osteogenic
differentiation. J Orthop Surg Res. 18:8202023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Huang Y, Zhang J, Zhu Y, Zhao R, Xie Z, Qu
X, Duan Y, Li N, Tang D and Luo X: BMP9 alleviates iron
accumulation-induced osteoporosis via the USP10/FOXO1/GPX4 axis. J
Adv Res. 79:953–972. 2026. View Article : Google Scholar :
|
|
83
|
Higuchi M, Kawamura H, Matsuki H, Hara T,
Takahashi M, Saito S, Saito K, Jiang S, Naito M, Kiyonari H and
Fujii M: usp10 is an essential deubiquitinase for hematopoiesis and
inhibits apoptosis of long-term hematopoietic stem cells. Stem Cell
Reports. 7:1116–1129. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Sever R and Brugge JS: Signal transduction
in cancer. Cold Spring Harb Perspect Med. 5:a0060982015. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Lim R, Sugino T, Nolte H, Andrade J,
Zimmermann B, Shi C, Doddaballapur A, Ong YT, Wilhelm K, Fasse JWD,
et al: Deubiquitinase USP10 regulates Notch signaling in the
endothelium. Science. 364:188–193. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Yuan T, Chen Z, Yan F, Qian M, Luo H, Ye
S, Cao J, Ying M, Dai X, Gai R, et al: Deubiquitinating enzyme
USP10 promotes hepatocellular carcinoma metastasis through
deubiquitinating and stabilizing Smad4 protein. Mol Oncol.
14:197–210. 2020. View Article : Google Scholar :
|
|
87
|
Teng S, Zhang Y, Jin X, Zhu Y, Li L, Huang
X, Wang D and Lin Z: Structure and hepatoprotective activity of
Usp10/NF-κB/Nrf2 pathway-related Morchella esculenta
polysaccharide. Carbohydr Polym. 303:1204532023. View Article : Google Scholar
|
|
88
|
Xie C, Gao X, Liu G, Tang H and Li C:
USP10 is a potential mediator for vagus nerve stimulation to
alleviate neuroinflammation in ischaemic stroke by inhibiting NF-κB
signalling pathway. Front Immunol. 14:11306972023. View Article : Google Scholar
|
|
89
|
Wang J, Gan L, Liu F, Yang Q, Deng Q,
Jiang D, Zhang C, Zhang L and Wang X: USP10 promotes pancreatic
ductal adenocarcinoma progression by attenuating FOXC1 protein
degradation to activate the WNT signaling pathway. Int J Biol Sci.
20:5343–5362. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Gao Y, Zhang X, Xiao L, Zhai C, Yi T, Wang
G, Wang E, Ji X, Hu L, Shen G and Wu S: Usp10 modulates the hippo
pathway by deubiquitinating and stabilizing the transcriptional
coactivator Yorkie. Int J Mol Sci. 20:60132019. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Huang J, Liu Y, Wang M, Wang R, Ling H and
Yang Y: FoxO4 negatively modulates USP10 transcription to aggravate
the apoptosis and oxidative stress of hypoxia/reoxygenation-induced
cardiomyocytes by regulating the Hippo/YAP pathway. J Bioenerg
Biomembr. 53:541–551. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Sun J, Xiang Q, Ding D and Yan N: USP10
suppresses ABCG2-induced malignant characteristics of
doxorubicin-resistant thyroid cancer by inhibiting PI3K/AKT
pathway. J Bioenerg Biomembr. 55:457–466. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Phillips AT, Boumil EF, Castro N,
Venkatesan A, Gallo E, Adams JJ, Sidhu SS and Bernstein AM: USP10
promotes fibronectin recycling, secretion, and organization. Invest
Ophthalmol Vis Sci. 62:152021. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Chen X, Ma Y, Liu H and Wang Y:
Multifunctional regulation and treatment of ubiquitin specific
protease 10. Biochem Pharmacol. 242:1172512025. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Li R, Gao F, Chen Y, Zhao J, Shi R, Li M,
Zuo Z, Chang P, De D, Chen L, et al: USP10 protects against
pressure overload-induced mitochondrial morphofunctional defects
and pathological cardiac hypertrophy through stabilizing
cytoplasmic Mfn2. Redox Biol. 85:1037452025. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Ko A, Han SY, Choi CH, Cho H, Lee MS, Kim
SY, Song JS, Hong KM, Lee HW, Hewitt SM, et al: Oncogene-induced
senescence mediated by c-Myc requires USP10 dependent
deubiquitination and stabilization of p14ARF. Cell Death Differ.
25:1050–1062. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wang C, Ni J, Zhai D, Xu Y, Wu Z, Chen Y,
Liu N, Du J, Shen Y, Liu G, et al: Stress-induced epinephrine
promotes hepatocellular carcinoma progression via the USP10-PLAGL2
signaling loop. Exp Mol Med. 56:1150–1163. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Wang Y, Liu J, Zheng S, Cao L, Li Y and
Sheng R: The deubiquitinase USP10 mediates crosstalk between the
LKB1/AMPK axis and Wnt/β-catenin signaling in cancer. FEBS Lett.
597:3061–3071. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Sun L, Yu J, Guinney J, Qin B and
Sinicrope FA: USP10 regulates ZEB1 ubiquitination and protein
stability to inhibit ZEB1-mediated colorectal cancer metastasis.
Mol Cancer Res. 21:578–590. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Duan R, Du W and Guo W: EZH2: A novel
target for cancer treatment. J Hematol Oncol. 13:1042020.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Lei A, Chen L, Zhang M, Yang X, Xu L, Cao
N, Zhang Z and Cao Y: EZH2 regulates protein stability via
recruiting USP7 to mediate neuronal gene expression in cancer
cells. Front Genet. 10:4222019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Huang J, Yin Q, Wang Y, Zhou X, Guo Y,
Tang Y, Cheng R, Yu X, Zhang J, Huang C, et al: EZH2 inhibition
enhances PD-L1 protein stability through USP22-mediated
deubiquitination in colorectal cancer. Adv Sci (Weinh).
11:e23080452024. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Dai Y, Chen J and Duan Q: Epigenetic
mechanism of EZH2-mediated histone methylation modification in
regulating ferroptosis of alveolar epithelial cells in
sepsis-induced acute lung injury. Drug Dev Res. 85:e222632024.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Alemasova EE and Lavrik OI:
Poly(ADP-ribosyl)ation by PARP1: Reaction mechanism and regulatory
proteins. Nucleic Acids Res. 47:3811–3827. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Peng Q, Wang S, Huang S, Deng Y, Li Z, Liu
C, Hong Y, Duan R, Xue X and Ge P: FTO/miR-503-5p/USP10 axis
regulates neuronal endoplasmic reticulum stress-mediated apoptosis
in ischemic stroke. Int Immunopharmacol. 149:1141502025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Ma B, Wang L, Li J, Zhang B, Wang Y, Li N,
Shen D and Han C: Brusatol suppresses malignant progression of
glioma via modulating the miR-574-5p/USP10/PLK1 axis.
Phytomedicine. 146:1571422025. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Pang D, Yu Y, Zhao B, Huang J, Cui Y, Li
T, Li C and Shang H: The long non-coding RNA NR3C2-8:1 promotes
p53-mediated apoptosis through the miR-129-5p/USP10 axis in
amyotrophic lateral sclerosis. Mol Neurobiol. 61:7466–7480. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Zhang B, Li H, Li D, Sun H, Li M and Hu H:
Long noncoding RNA Mirt2 upregulates USP10 expression to suppress
hepatic steatosis by sponging miR-34a-5p. Gene. 700:139–148. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Shen C, Li J, Zhang Q, Tao Y, Li R, Ma Z
and Wang Z: LncRNA GASAL1 promotes hepatocellular carcinoma
progression by up-regulating USP10-stabilized PCNA. Exp Cell Res.
415:1129732022. View Article : Google Scholar
|
|
110
|
Ouyang SW, Liu TT, Liu XS, Zhu FX, Zhu FM,
Liu XN and Peng ZH: USP10 regulates Musashi-2 stability via
deubiquitination and promotes tumour proliferation in colon cancer.
FEBS Lett. 593:406–413. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wu L, Zhao N, Zhou Z, Chen J, Han S, Zhang
X, Bao H, Yuan W and Shu X: PLAGL2 promotes the proliferation and
migration of gastric cancer cells via USP37-mediated
deubiquitination of Snail1. Theranostics. 11:700–714. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Sekiya R, Maeda M, Yuan H, Asano E, Hyodo
T, Hasegawa H, Ito S, Shibata K, Hamaguchi M, Kikkawa F, et al:
PLAGL2 regulates actin cytoskeletal architecture and cell
migration. Carcinogenesis. 35:1993–2001. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Wang H, Liang L, Xie Y, Gong H, Fan F, Wen
C, Jiang Y, Lei S, Qiu X, Peng H, et al: Pseudokinase TRIB3
stabilizes SSRP1 via USP10-mediated deubiquitination to promote
multiple myeloma progression. Oncogene. 44:694–708. 2025.
View Article : Google Scholar
|
|
114
|
Wang X, Xia S, Li H, Wang X, Li C, Chao Y,
Zhang L and Han C: The deubiquitinase USP10 regulates KLF4
stability and suppresses lung tumorigenesis. Cell Death Differ.
27:1747–1764. 2020. View Article : Google Scholar
|
|
115
|
Zhang N, Wang B, Ma C, Zeng J, Wang T, Han
L and Yang M: LINC00240 in the 6p22.1 risk locus promotes gastric
cancer progression through USP10-mediated DDX21 stabilization. J
Exp Clin Cancer Res. 42:892023. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Lin Z, Yang H, Tan C, Li J, Liu Z, Quan Q,
Kong S, Ye J, Gao B and Fang D: USP10 antagonizes c-Myc
transcriptional activation through SIRT6 stabilization to suppress
tumor formation. Cell Rep. 5:1639–1649. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Sun T, Xu YJ, Jiang SY, Xu Z, Cao BY,
Sethi G, Zeng YY, Kong Y and Mao XL: Suppression of the USP10/CCND1
axis induces glioblastoma cell apoptosis. Acta Pharmacol Sin.
42:1338–1346. 2021. View Article : Google Scholar
|
|
118
|
Xu YJ, Zeng K, Ren Y, Mao CY, Ye YH, Zhu
XT, Sun ZY, Cao BY, Zhang ZB, Xu GQ, et al: Inhibition of USP10
induces myeloma cell apoptosis by promoting cyclin D3 degradation.
Acta Pharmacol Sin. 44:1920–1931. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Zeng Z, Li Y, Zhou H, Li M, Ye J, Li D,
Zhu Y, Zhang Y, Zhang X, Deng Y, et al: System-wide identification
of novel de-ubiquitination targets for USP10 in gastric cancer
metastasis through multi-omics screening. BMC Cancer. 24:7732024.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Deng J, Yi X, Feng Z, Peng J, Li D, Li C,
Deng B, Liu S, Sahu S and Hao L: Deubiquitinating enzyme USP10
promotes osteosarcoma metastasis and epithelial-mesenchymal
transition by stabilizing YAP1. Cancer Med. 12:14452–14467. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Li P, Yang L, Park SY, Liu F, Li AH, Zhu
Y, Sui H, Gao F, Li L, Ye L, et al: Stabilization of MOF (KAT8) by
USP10 promotes esophageal squamous cell carcinoma proliferation and
metastasis through epigenetic activation of ANXA2/Wnt signaling.
Oncogene. 43:899–917. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Niu J, Shi Y, Xue J, Miao R, Huang S, Wang
T, Wu J, Fu M and Wu ZH: USP10 inhibits genotoxic NF-κB activation
by MCPIP1-facilitated deubiquitination of NEMO. EMBO J.
32:3206–3219. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Liu X, Chen B, Chen J, Su Z and Sun S:
Deubiquitinase ubiquitin-specific peptidase 10 maintains cysteine
rich angiogenic inducer 61 expression via Yes1 associated
transcriptional regulator to augment immune escape and metastasis
of pancreatic adenocarcinoma. Cancer Sci. 113:1868–1879. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Lu C, Ning Z, Wang A, Chen D, Liu X, Xia
T, Tekcham DS, Wang W, Li T, Liu X, et al: USP10 suppresses tumor
progression by inhibiting mTOR activation in hepatocellular
carcinoma. Cancer Lett. 436:139–148. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Luo Y, Zhu Q, Xiang S, Wang Q, Li J, Chen
X, Yan W, Feng J and Zu X: Downregulated circPOKE promotes breast
cancer metastasis through activation of the USP10-Snail axis.
Oncogene. 42:3236–3251. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Chen C, Zhao S, Karnad A and Freeman JW:
The biology and role of CD44 in cancer progression: Therapeutic
implications. J Hematol Oncol. 11:642018. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Sethi A, Mishra S, Upadhyay V, Dubey P,
Siddiqui S, Singh AK, Chowdhury S, Srivastava S, Srivastava P,
Sahoo P, et al: USP10 deubiquitinates and stabilizes CD44 leading
to enhanced breast cancer cell proliferation, stemness and
metastasis. Biochem J. 481:1877–1900. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Shi Y, Ding J, Ling X, Xu D, Shen Y and
Qin X: USP10 stabilizes BAZ1A to drive tumor stemness via an
epigenetic mechanism in head and neck squamous cell carcinoma. Cell
Death Dis. 16:2702025. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Reissland M, Hartmann O, Tauch S, Bugter
JM, Prieto-Garcia C, Schulte C, Loebbert S, Solvie D, Bitman-Lotan
E, Narain A, et al: USP10 drives cancer stemness and enables
super-competitor signalling in colorectal cancer. Oncogene.
43:3645–3659. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Sun SC: Deubiquitylation and regulation of
the immune response. Nat Rev Immunol. 8:501–511. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Dufner A, Kisser A, Niendorf S, Basters A,
Reissig S, Schönle A, Aichem A, Kurz T, Schlosser A, Yablonski D,
et al: The ubiquitin-specific protease USP8 is critical for the
development and homeostasis of T cells. Nat Immunol. 16:950–960.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Gao Y, Lin F, Xu P, Nie J, Chen Z, Su J,
Tang J, Wu Q, Li Y, Guo Z, et al: USP22 is a positive regulator of
NFATc2 on promoting IL2 expression. FEBS Lett. 588:878–883. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Kuang Z, Liu X, Zhang N, Dong J, Sun C,
Yin M, Wang Y, Liu L, Xiao D, Zhou X, et al: USP2 promotes tumor
immune evasion via deubiquitination and stabilization of PD-L1.
Cell Death Differ. 30:2249–2264. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Wang W, Huang X, Xin HB, Fu M, Xue A and
Wu ZH: TRAF family member-associated NF-κB activator (TANK)
inhibits genotoxic nuclear factor κB activation by facilitating
deubiquitinase USP10-dependent deubiquitination of TRAF6 ligase. J
Biol Chem. 290:13372–13385. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Ye Z, Chen J, Huang P, Xuan Z and Zheng S:
Ubiquitin-specific peptidase 10, a deubiquitinating enzyme:
Assessing its role in tumor prognosis and immune response. Front
Oncol. 12:9901952022. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Fu M, Li J, Xuan Z, Zheng Z, Liu Y, Zhang
Z, Zheng J, Zhong M, Liu B, Du Y, et al: NDR1 mediates PD-L1
deubiquitination to promote prostate cancer immune escape via
USP10. Cell Commun Signal. 22:4292024. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Shen GY, Zhang Y, Huang RZ, Huang ZY, Yang
LY, Chen DZ and Yang SB: FOXP4-AS1 promotes CD8+ T cell
exhaustion and esophageal cancer immune escape through
USP10-stabilized PD-L1. Immunol Res. 72:766–775. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Zeng L, Zhu Y, Cui X, Chi J, Uddin A, Zhou
Z, Song X, Dai M, Cristofanilli M, Kalinsky K and Wan Y: Tuning
immune-cold tumor by suppressing USP10/B7-H4 proteolytic axis
reinvigorates therapeutic efficacy of ADCs. Adv Sci (Weinh).
11:e24007572024. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Fang XL, Li QJ, Lin JY, Huang CL, Huang
SY, Tan XR, He SW, Zhu XH, Li JY, Gong S, et al: Transcription
factor ATMIN facilitates chemoresistance in nasopharyngeal
carcinoma. Cell Death Dis. 15:1122024. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Li QJ, Fang XL, Li YQ, Lin JY, Huang CL,
He SW, Huang SY, Li JY, Gong S, Liu N, et al: DCAF7 acts as a
scaffold to Recruit USP10 for G3BP1 deubiquitylation and
facilitates chemoresistance and metastasis in nasopharyngeal
carcinoma. Adv Sci (Weinh). 11:e24032622024. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Lin C, Lin P, Yao H, Liu S, Lin X, He R,
Teng Z, Zuo X, Li Y, Ye J and Zhu G: Modulation of YBX1-mediated
PANoptosis inhibition by PPM1B and USP10 confers chemoresistance to
oxaliplatin in gastric cancer. Cancer Lett. 587:2167122024.
View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Zhai S, Lin J, Ji Y, Zhang R, Zhang Z, Cao
Y, Liu Y, Tang X, Liu J, Liu P, et al: A microprotein N1DARP
encoded by LINC00261 promotes Notch1 intracellular domain (N1ICD)
degradation via disrupting USP10-N1ICD interaction to inhibit
chemoresistance in Notch1-hyperactivated pancreatic cancer. Cell
Discov. 9:952023. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Hou S, Zhao T, Deng B, Li C, Li W, Huang H
and Hang Q: USP10 promotes migration and cisplatin resistance in
esophageal squamous cell carcinoma cells. Med Oncol. 41:332023.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
He Y, Jiang S, Mao C, Zheng H, Cao B,
Zhang Z, Zhao J, Zeng Y and Mao X: The deubiquitinase USP10
restores PTEN activity and inhibits non-small cell lung cancer cell
proliferation. J Biol Chem. 297:1010882021. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Sun J, Li T, Zhao Y, Huang L, Sun H, Wu H
and Jiang X: USP10 inhibits lung cancer cell growth and invasion by
upregulating PTEN. Mol Cell Biochem. 441:1–7. 2018. View Article : Google Scholar
|
|
146
|
Luo D, Tang H, Tan L, Zhang L, Wang L,
Cheng Q, Lei X and Wu J: lncRNA JPX promotes tumor progression by
interacting with and destabilizing YTHDF2 in cutaneous melanoma.
Mol Cancer Res. 22:524–537. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Chen Q, Xiong X and Sun Y: USP10
deubiquitylates and stabilizes DIRAS2 to suppress the growth of
pancreatic cancer cells. MedComm (2020). 5:e7512024. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Wang Y, Mao A, Liu J, Li P, Zheng S, Tong
T, Li Z, Zhang H, Ma L, Lin J, et al: USP10 strikes down β-catenin
by dual-wielding deubiquitinase activity and phase separation
potential. Cell Chem Biol. 30:1436–1452.e10. 2023. View Article : Google Scholar
|
|
149
|
Piatnitskaia S, Takahashi M, Kitaura H,
Katsuragi Y, Kakihana T, Zhang L, Kakita A, Iwakura Y, Nawa H,
Miura T, et al: USP10 is a critical factor for tau-positive stress
granule formation in neuronal cells. Sci Rep. 9:105912019.
View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Cai J, Xiong W, Wang X and Tan H;
Alzheimer's Disease Neuroimaging Initiative (ADNI) Database:
Genetic architecture of hippocampus subfields volumes in
Alzheimer's disease. CNS Neurosci Ther. 30:e141102024. View Article : Google Scholar :
|
|
151
|
Anisimov S, Takahashi M, Kakihana T,
Katsuragi Y, Sango J, Abe T and Fujii M: USP10 inhibits the
degradation of α-synuclein, a pathogenic factor associated with
Parkinson's disease, by inhibiting chaperone-mediated autophagy. J
Biol Chem. 301:1102922025. View Article : Google Scholar
|
|
152
|
Takahashi M, Kitaura H, Kakita A, Kakihana
T, Katsuragi Y, Onodera O, Iwakura Y, Nawa H, Komatsu M and Fujii
M: USP10 inhibits aberrant cytoplasmic aggregation of TDP-43 by
promoting stress granule clearance. Mol Cell Biol. 42:e00393212022.
View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Zhang DH, Zhang JL, Huang Z, Wu LM, Wang
ZM, Li YP, Tian XY, Kong LY, Yao R and Zhang YZ: Deubiquitinase
ubiquitin-specific protease 10 deficiency regulates Sirt6 signaling
and exacerbates cardiac hypertrophy. J Am Heart Assoc.
9:e0177512020. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Lu L, Ma J, Liu Y, Shao Y, Xiong X, Duan
W, Gao E, Yang Q, Chen S, Yang J, et al: FSTL1-USP10-Notch1
signaling axis protects against cardiac dysfunction through
inhibition of myocardial fibrosis in diabetic mice. Front Cell Dev
Biol. 9:7570682021. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Zhao M, Zheng Z, Peng S, Xu Y, Zhang J,
Liu J, Pan W, Yin Z, Xu S, Wei C, et al: Epidermal growth
factor-like repeats and discoidin I-like domains 3 deficiency
attenuates dilated cardiomyopathy by inhibiting ubiquitin specific
peptidase 10 dependent Smad4 deubiquitination. J Am Heart Assoc.
13:e0312832024. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Xie S, Xing Y, Shi W, Zhang M, Chen M,
Fang W, Liu S, Zhang T, Zeng X, Chen S, et al: Cardiac fibroblast
heat shock protein 47 aggravates cardiac fibrosis post myocardial
ischemia-reperfusion injury by encouraging ubiquitin specific
peptidase 10 dependent Smad4 deubiquitination. Acta Pharm Sin B.
12:4138–4153. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Yang Y, Wang Z, Wang N, Yang J and Yang L:
CaMKII exacerbates doxorubicin-induced cardiotoxicity by promoting
ubiquitination through USP10 inhibition. Cancer Med. 13:e702862024.
View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Mao Q, Zhang X, Yang J, Kong Q, Cheng H,
Yu W, Cao X, Li Y, Li C, Liu L and Ding Z: HSPA12A acts as a
scaffolding protein to inhibit cardiac fibroblast activation and
cardiac fibrosis. J Adv Res. 67:217–229. 2025. View Article : Google Scholar :
|
|
159
|
Kudryashova TV, Dabral S, Nayakanti S, Ray
A, Goncharov DA, Avolio T, Shen Y, Rode A, Pena A, Jiang L, et al:
Noncanonical HIPPO/MST signaling via BUB3 and FOXO drives pulmonary
vascular cell growth and survival. Circ Res. 130:760–778. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Xia X, Liu X, Chai R, Xu Q, Luo Z, Gu J,
Jin Y, Hu T, Yu C, Du B, et al: USP10 exacerbates neointima
formation by stabilizing Skp2 protein in vascular smooth muscle
cells. J Biol Chem. 297:1012582021. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Mushajiang M, Li Y, Sun Z, Liu J, Zhang L
and Wang Z: USP10 alleviates Nε-carboxymethyl-lysine-induced
vascular calcification and atherogenesis in diabetes mellitus by
promoting AMPK activation. Cell Signal. 120:1112112024. View Article : Google Scholar
|
|
162
|
Chen M, Li Z, Da L, Liu J, Huang C and Zha
Z: LncRNA PVT1 promotes intracranial aneurysm development via
USP10/KLF4/NLRP3 axis-mediated pyroptosis in human cerebral smooth
muscle cells. J Biochem Mol Toxicol. 39:e701022025. View Article : Google Scholar
|
|
163
|
Jiangqiao Z, Tianyu W, Zhongbao C, Long Z,
Jilin Z, Xiaoxiong M and Tao Q: Ubiquitin-specific peptidase 10
protects against hepatic ischaemic/reperfusion injury via TAK1
signalling. Front Immunol. 11:5062752020. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Xin SL and Yu YY: Ubiquitin-specific
peptidase 10 ameliorates hepatic steatosis in nonalcoholic
steatohepatitis model by restoring autophagic activity. Dig Liver
Dis. 54:1021–1029. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Xin SL, Pan XL, Xu XY and Yu YY: USP10
alleviates palmitic acid-induced steatosis through autophagy in
HepG2 cells. J Clin Transl Hepatol. 11:45–57. 2023.
|
|
166
|
Tian S, Zhou X, Zheng L, Liu J, Zhang M,
Ma S, Zheng X, Guo G, Ju R, Yang F, et al: Engineered mesenchymal
stem cell-derived small extracellular vesicles mitigate liver
fibrosis by delivering USP10 to reprogram macrophage phenotype.
Biomater Res. 29:02442025. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Liu S, Yang Y, Li Q, Yu L, Zong Z, Zang R,
Ji W and Sun S: Ubiquitin-specific peptidase 10 promotes renal
interstitial fibrosis progression through deubiquitinating and
stabilizing P53 protein. Biochim Biophys Acta Mol Basis Dis.
1871:1676602025. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Zhao Q, Zhang R, Wang Y, Li T, Xue J and
Chen Z: FOXQ1, deubiquitinated by USP10, alleviates sepsis-induced
acute kidney injury by targeting the CREB5/NF-κB signaling axis.
Biochim Biophys Acta Mol Basis Dis. 1870:1673312024. View Article : Google Scholar
|
|
169
|
Han B, Zheng Q, Li H, Wang Y and Zhang D:
PRDM16 suppresses pyroptosis to attenuate the progression of AKI
caused by rhabdomyolysis via upregulation of USP10. Cell Mol Life
Sci. 82:1382025. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Gao F, Qian M, Liu G, Ao W, Dai D and Yin
C: USP10 alleviates sepsis-induced acute kidney injury by
regulating Sirt6-mediated Nrf2/ARE signaling pathway. J Inflamm
(Lond). 18:252021. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Mao S, Yu N, Wang W, Mao Y, Du Y, Zhao Q,
Gu X and Kang J: Ubiquitin-specific peptidase 10 attenuates
bleomycin-induced pulmonary fibrosis via modulating autophagy
depending on sirtuin 6-mediated AKT/mTOR. Cell Biol Toxicol.
41:732025. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Bomberger JM, Barnaby RL and Stanton BA:
The deubiquitinating enzyme USP10 regulates the post-endocytic
sorting of cystic fibrosis transmembrane conductance regulator in
airway epithelial cells. J Biol Chem. 284:18778–18789. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Bomberger JM, Barnaby RL and Stanton BA:
The deubiquitinating enzyme USP10 regulates the endocytic recycling
of CFTR in airway epithelial cells. Channels (Austin). 4:150–154.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Swiatecka-Urban A, Moreau-Marquis S,
Maceachran DP, Connolly JP, Stanton CR, Su JR, Barnaby R, O'toole
GA and Stanton BA: Pseudomonas aeruginosa inhibits endocytic
recycling of CFTR in polarized human airway epithelial cells. Am J
Physiol Cell Physiol. 290:C862–C872. 2006. View Article : Google Scholar
|
|
175
|
Bomberger JM, Ye S, Maceachran DP, Koeppen
K, Barnaby RL, O'Toole GA and Stanton BA: A Pseudomonas aeruginosa
toxin that hijacks the host ubiquitin proteolytic system. PLoS
Pathog. 7:e10013252011. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Gillespie SR, Tedesco LJ, Wang L and
Bernstein AM: The deubiquitylase USP10 regulates integrin β1 and β5
and fibrotic wound healing. J Cell Sci. 130:3481–3495. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Boumil EF, Castro N, Phillips AT,
Chatterton JE, McCauley SM, Wolfson AD, Shmushkovich T, Ridilla M
and Bernstein AM: USP10 targeted self-deliverable siRNA to prevent
scarring in the Cornea. Mol Ther Nucleic Acids. 21:1029–1043. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Shi J, Jia X, He Y, Ma X, Qi X, Li W, Gao
SJ, Yan Q and Lu C: Immune evasion strategy involving
propionylation by the KSHV interferon regulatory factor 1 (vIRF1).
PLoS Pathog. 19:e10113242023. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Gao H, Xi Z, Dai J, Xue J, Guan X, Zhao L,
Chen Z and Xing F: Drug resistance mechanisms and treatment
strategies mediated by ubiquitin-specific proteases (USPs) in
cancers: New directions and therapeutic options. Mol Cancer.
23:882024. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Harrigan JA, Jacq X, Martin NM and Jackson
SP: Deubiquitylating enzymes and drug discovery: Emerging
opportunities. Nat Rev Drug Discov. 17:57–78. 2018. View Article : Google Scholar
|
|
181
|
Kona SV and Kalivendi SV: The USP10/13
inhibitor, spautin-1, attenuates the progression of glioblastoma by
independently regulating RAF-ERK mediated glycolysis and SKP2.
Biochim Biophys Acta Mol Basis Dis. 1870:1672912024. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Liao Y, Guo Z, Xia X, Liu Y, Huang C,
Jiang L, Wang X, Liu J and Huang H: Inhibition of EGFR signaling
with Spautin-1 represents a novel therapeutics for prostate cancer.
J Exp Clin Cancer Res. 38:1572019. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Yi J, Wang HL, Lu G, Zhang H, Wang L, Li
ZY, Wang L, Wu Y, Xia D, Fang EF and Shen HM: Spautin-1 promotes
PINK1-PRKN-dependent mitophagy and improves associative learning
capability in an alzheimer disease animal model. Autophagy.
20:2655–2676. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Lu Y, Gao J, Wang P, Chen H, He X, Luo M,
Guo Y, Li L, Zhuang W, Zhang B, et al: Discovery of potent small
molecule ubiquitin-specific protease 10 inhibitors with
anti-hepatocellular carcinoma activity through regulating YAP
expression. Eur J Med Chem. 272:1164682024. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Lu Y, Wang W, Yuan C, Luo M, Wang P, Li B,
Hu L, Shen Z, Zhou Y, Che J and Dong X: Discovery of a
ubiquitin-specific protease 10 inhibitor with high binding affinity
promoting cell apoptosis via down-regulated CDK4 for the treatment
of hepatocellular carcinoma. Bioorg Chem. 163:1087752025.
View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Weisberg EL, Schauer NJ, Yang J, Lamberto
I, Doherty L, Bhatt S, Nonami A, Meng C, Letai A, Wright R, et al:
Inhibition of USP10 induces degradation of oncogenic FLT3. Nat Chem
Biol. 13:1207–1215. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Yu M, Fang ZX, Wang WW, Zhang Y, Bu ZL,
Liu M, Xiao XH, Zhang ZL, Zhang XM, Cao Y, et al: Wu-5, a novel
USP10 inhibitor, enhances crenolanib-induced FLT3-ITD-positive AML
cell death via inhibiting FLT3 and AMPK pathways. Acta Pharmacol
Sin. 42:604–612. 2021. View Article : Google Scholar
|
|
188
|
Han Y, Kong W, Shang Q, Liu Y, Ni X, Yang
L and Lei J: Discovery of targeting USP10-mediated proline
metabolism arrangement to inhibit hepatocellular carcinoma
progression. Biochem Pharmacol. 236:1169042025. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Cao YF, Xie L, Tong BB, Chu MY, Shi WQ, Li
X, He JZ, Wang SH, Wu ZY, Deng DX, et al: Targeting USP10 induces
degradation of oncogenic ANLN in esophageal squamous cell
carcinoma. Cell Death Differ. 30:527–543. 2023. View Article : Google Scholar :
|
|
190
|
Ge G, Zhao W, Zhong Z, Huang Y, Hua Y,
Chen K, Yu Y, Wu T, Lu Y, Yadav N and Zhang F: Acacetin ameliorates
pressure overload-induced cardiac remodeling by targeting USP10 and
inhibiting maladaptive cardiomyocyte autophagy. Phytomedicine.
141:1565882025. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Liu LB, Huang SH, Qiu HL, Cen XF, Guo YY,
Li D, Ma YL, Xu M and Tang QZ: Limonin stabilises sirtuin 6 (SIRT6)
by activating ubiquitin specific peptidase 10 (USP10) in cardiac
hypertrophy. Br J Pharmacol. 179:4516–4533. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Li Y, Hu K, Li J, Yang X, Wu X, Liu Q,
Chen Y, Ding Y, Liu L, Yang Q and Wang G: Tetrahydroxy stilbene
glucoside promotes mitophagy and ameliorates neuronal injury after
cerebral ischemia reperfusion via promoting USP10-mediated YBX1
stability. eNeuro. 11:ENEURO.0269–24.2024. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Rowinsky EK, Paner A, Berdeja JG,
Paba-Prada C, Venugopal P, Porkka K, Gullbo J, Linder S, Loskog A,
Richardson PG and Landgren O: Phase 1 study of the protein
deubiquitinase inhibitor VLX1570 in patients with relapsed and/or
refractory multiple myeloma. Invest New Drugs. 38:1448–1453. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Torrado C, Ashton NW, D'Andrea AD and Yap
TA: USP1 inhibition: A journey from target discovery to clinical
translation. Pharmacol Ther. 271:1088652025. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Fleming J, Magana P, Nair S, Tsenkov M,
Bertoni D, Pidruchna I, Lima Afonso MQ, Midlik A, Paramval U, Žídek
A, et al: AlphaFold protein structure database and 3D-beacons: New
data and capabilities. J Mol Biol. 437:1689672025. View Article : Google Scholar : PubMed/NCBI
|