Introduction
Spinal cord injury (SCI) is a traumatic condition
that imposes notable physical, emotional and economic burdens on
patients and their families. An epidemiological meta-analysis
showed that the incidence of traumatic SCI in developing countries
was 22.55 per million individuals per year (1). The pathogenesis of SCI is well
understood and involves primary and secondary injuries. Primary
injury occurs immediately following trauma and typically involves
mechanical damage, either permanent or temporary compression
(2,3). Secondary injury, characterized by
destructive biochemical changes within the spinal cord, including
neuroinflammation, cell death, blood vessel injury, disruption of
the blood-spinal cord barrier and oxidative stress, further
exacerbates tissue damage and functional impairment (4,5).
Despite extensive research into various treatment modalities such
as pharmacotherapy, stem cell therapy, biomaterials and
rehabilitation, effective treatments for SCI remain elusive
(6-8).
Cell death, particularly neuronal death, is a
critical factor contributing to functional deficits following SCI.
Therefore, strategies aimed at preventing neuronal death may hold
promise for ameliorating post-SCI pathology. Various types of cell
death have been reported in SCI, including necroptosis, apoptosis,
autophagy, ferroptosis, pyroptosis and paraptosis (9). Ferroptosis is a recently discovered
form of regulated cell death defined by iron-dependent lipid
peroxidation (10). Glutathione
peroxidase 4 (GPX4), cystine/glutamate antiporter (System Xc-) and
glutathione (GSH) collectively constitute the System Xc-/GSH/GPX4
axis, which is crucial in mitigating excessive lipid peroxidation
and reducing reactive oxygen species generation in ferroptosis
(11). Our previous studies have
revealed the engagement of ferroptosis in SCI pathogenesis and
highlighted the therapeutic potential of ferroptosis inhibitors in
promoting functional recovery in SCI animal models (12,13). Moreover, previous studies have
underscored the significance of the System Xc-/GSH/GPX4 axis in
regulating ferroptosis after SCI, consistent with findings from
other research groups (14-16).
Deferoxamine (DFO), an Food and Drug Administration
(FDA)-approved iron-chelating agent, has been shown in our previous
study to prevent ferroptosis and promote functional recovery in SCI
mice (12). Additionally, DFO
has been demonstrated to reverse erastin-induced neuronal
ferroptosis in primary cortical neurons (17). Although the efficacy of DFO in
treating SCI by inhibiting ferroptosis has been established, the
underlying molecular mechanisms remain incompletely understood.
Previous studies have suggested that various
compounds, including zinc, trehalose and resveratrol, promote
functional recovery after SCI by inhibiting ferroptosis through
upregulation of the Nrf2 gene. Specifically, Ge et al
(18) explored the role of zinc
in SCI and found that it attenuates ferroptosis and promotes
functional recovery by activating the Nrf2/GPX4 defense pathway.
This study underscored the importance of the Nrf2 pathway in
mitigating ferroptosis and aiding recovery after SCI. Gong et
al (19) also reported that
trehalose inhibits ferroptosis in an SCI mouse model by activating
the Nrf2/heme oxygenase-1 (HO-1) pathway. Their findings suggested
that the Nrf2/HO-1 axis plays a crucial role in protecting neurons
from ferroptosis and enhancing functional recovery. Ni et al
(20) found that resveratrol
inhibits ferroptosis via activation of the Nrf2/GPX4 pathway in
mice with SCI. This study further supported the involvement of the
Nrf2 pathway in reducing ferroptosis and promoting recovery
post-SCI. These studies collectively highlight the Nrf2 signaling
pathway as a key regulator in inhibiting ferroptosis and promoting
functional recovery in SCI. By activating components of the Nrf2
pathway, such as GPX4 and HO-1, various compounds, including zinc,
trehalose and resveratrol, offer neuroprotection and facilitate
recovery following SCI. This suggests that targeting the Nrf2
pathway could be a promising therapeutic strategy for mitigating
ferroptosis and enhancing outcomes in SCI. Despite the
well-documented role of the Nrf2/HO-1 signaling pathway in
modulating oxidative stress and ferroptosis, the specific
interaction between DFO and this pathway in the context of SCI
remains largely unexplored. Previous studies have primarily focused
on the general effects of Nrf2 activation in oxidative stress and
ferroptosis, but the direct involvement of the Nrf2/HO-1 axis in
mediating the neuroprotective effects of DFO after SCI has not been
fully elucidated (21,22). This gap in knowledge presents an
opportunity to explore the intricate molecular crosstalk between
DFO and the Nrf2/HO-1 pathway, thereby advancing our understanding
of the mechanistic basis of the therapeutic potential of DFO in
SCI.
The present study aimed to elucidate the role of the
Nrf2/HO-1 signaling pathway in mediating the inhibitory effects of
DFO on neuronal ferroptosis after SCI. To achieve this goal, an SCI
mouse model was employed to investigate alterations in Nrf2
expression following DFO administration. Additionally, the
Nrf2-specific inhibitor, ML385, was utilized to elucidate the
engagement of the Nrf2/HO-1 signaling pathway in the molecular
mechanisms underlying DFO treatment.
Materials and methods
Animals
Female C57BL/6 J Nifdc mice weighing 20-25 g and
aged 6-8 weeks were procured from Beijing Vital River Laboratory
Animal Technology Co., Ltd. [License SCXK (Jing) 2021-0011]. To
eliminate potential sex-related variations and reduce the risk of
urinary tract infections, only female mice were included in the
present study. The mice were kept in standard cages with a 12-h
light-dark cycle, with 5 mice per cage. Adequate water and food
were supplied ad libitum and the ambient temperature was
maintained between 22 and 24°C. Prior to the commencement of the
experimental procedures, the mice were housed to acclimatize to
their surroundings for 1 week. Ethical approval for the study was
obtained from the Animal Experiments and Experimental Animal
Welfare Committee of Capital Medical University (Beijing, China;
approval no. AEEI-2023-029, granted on March 8, 2023). All
procedures were performed according to the guidelines stipulated by
the Animal Research: Reporting of in vivo Experiments
(ARRIVE) (23).
SCI model and treatment
The SCI model was established following the protocol
outlined in a previous study (24). In brief, mice were anesthetized
with a combination of isoflurane (Lunan Pharmaceutical Group
Limited) and air (4% isoflurane for induction and 2% for
maintenance). A total of 60 mice were randomly assigned to one of
four groups: The Sham, SCI, SCI + DFO injection (DFO group) and SCI
+ DFO with ML385 injection (ML385 group) groups (n=15 per group).
To establish the SCI model, mice were anesthetized and maintained
with isoflurane. Subsequently, each mouse was positioned on the
operating table in a prone position. A laminectomy was performed to
expose the T9/T10 vertebral region and the dorsal tissue of the
spinal cord was fully exposed. SCI was induced using a
weight-dropping method with an IMPACTOR MODEL III (procured from
State University of New Jersey, New Brunswick, NJ, USA), utilizing
a 30 mm high, 5 g weight and 2 mm diameter rod. The incision was
then closed with sterile sutures. Mice in the Sham group only
underwent laminectomy without spinal cord damage. Successful
induction of SCI was confirmed by bilateral hind limb paralysis in
the mice. Throughout the experiment, the animals were monitored
every day for general health, cage activity and signs of infection.
Bladder urination was completed twice daily during the first week
post-surgery. Mice in the DFO and ML385 groups received
intraperitoneal injections of DFO (100 mg/kg; once daily for 3
consecutive days) and mice in the ML385 group additionally received
ML385 (30 mg/kg; once daily for 3 consecutive days). The doses of
DFO and ML385 were selected based on previous studies in which
these regimens effectively reduced ferroptosis and modulated Nrf2
signaling in vivo without causing notable toxicity in mice
(12,25). Mice in the SCI and Sham groups
received equivalent intraperitoneal injections of 0.9% NaCl, also
administered once daily until the third day post-modeling. Animals
were monitored at least once daily throughout the study and more
frequently during the first 72 h post-injury. Predefined humane
endpoints for early euthanasia included: i) >20% loss of
preoperative body weight or progressive weight loss with
dehydration despite supportive care; ii) inability to reach
food/water or failure to thrive; iii) severe or unrelieved
pain/distress not responsive to analgesia/supportive care; iv)
severe wound complications (such as dehiscence, uncontrolled
bleeding, infection, pressure sores or autophagia); and v) moribund
condition (such as minimal responsiveness or other signs of
impending death as assessed by trained personnel). If any criterion
had been met, animals would have been euthanized immediately in
accordance with institutional guidelines. However, no animals
reached the predefined humane endpoints before completion of the
planned study period.
Behavioral testing
For behavioral assessment, 6 mice per group were
followed longitudinally, and Basso Mouse Scale (BMS) scores and
subscores were recorded at 1, 3, 5, 7, 14, 21 and 28 days
post-injury. The BMS hindlimb locomotor rating system, as described
by Basso et al (26), was
employed to assess the functional recovery of the mice. The BMS
scores range from 0 to 9 points, with 0 indicating complete
paralysis and 9 representing normal function. Assessment criteria
encompassed observations of range of motion, frequency of hindlimb
plantar stepping, rear ankle joint activities, coordination, touch
response of the soles of the feet and insteps, foot position, trunk
stability and tail position. Additionally, BMS subscores ranging
from 0 to 11 were assessed concurrently. To minimize the potential
influence of bladder fullness, manual bladder expression was
performed 2 h prior to BMS evaluations. The evaluators were blinded
to the experimental conditions of the mice and assessments were
conducted three times with immediate recording. All personnel
involved underwent rigorous training to ensure consistency and
accuracy in the assessments.
Tissue preparation
At 2 (n=6 per group) and 28 (n=9 per group) days
post-surgery, the spinal cord tissue was harvested from the mice of
each experimental group. Animals were euthanized by isoflurane
overdose (5% isoflurane in room air until cessation of breathing)
and death was confirmed by the absence of heartbeat and the loss of
corneal and pedal withdrawal reflexes prior to perfusion. Day 2
post-SCI was selected as an early acute-phase time point to assess
rapid activation of oxidative stress responsive pathways (including
Nrf2/HO-1) and the early effect of DFO, whereas day 28 was used to
evaluate longer-term outcomes. This was followed by cardiac
perfusion with 0.9% NaCl and 4% paraformaldehyde (PFA).
Subsequently, spinal cord samples measuring 0.5 cm in length,
containing the epicenter of the injury site, were carefully
harvested. Samples designated for western blot analysis were washed
with phosphate-buffered saline (PBS) and lysed using RIPA lysis
buffer (Beijing Aoqing Biotechnology Co., Ltd.). Samples intended
for immunofluorescence staining or histology staining were fixed in
4% PFA solution. Following fixation, a gradient dehydration process
was carried out using sucrose solutions. The spinal cord tissues
were embedded in Optimal Cutting Temperature Compound (Beijing
Aoqing Biotechnology Co., Ltd.), frozen and cut into 10-µm
longitudinal cryosections in a cryostat (CM1950; Leica Microsystems
GmbH) with the chamber temperature set to -20°C. These frozen
sections were stored at -20°C until further analysis.
Western blotting
Western blotting was performed according to our
previous study (27). For each
time point, spinal cord samples from 3 mice per group were pooled
and analyzed. For each sample, 20-30 µg of protein was mixed with
5X SDS-PAGE loading buffer (Beyotime Biotechnology) and boiled at
95°C for 5 min. The samples were then loaded onto a 10% SDS-PAGE
gel and separated by electrophoresis at 80 V for stacking and 120 V
for resolving. After electrophoresis, the proteins were transferred
to PVDF membranes (MilliporeSigma) at 300 mA for 90 min using a wet
transfer system (Bio-Rad Laboratories, Inc.). Following the
transfer, membranes were blocked with 5% non-fat milk in
Tris-buffered saline with 0.1% Tween-20 (TBST) for 1 h at room
temperature. The membranes were then incubated with primary
antibodies overnight at 4°C. The primary antibodies used were:
Anti-Nrf2 (1:1,000; CST Biological Reagents Co., Ltd.; cat. no.
12721S), anti-HO-1 (1:1,000; Abcam; cat. no. ab68477), anti-GPX4
(1:1,000; Abcam; cat. no. ab41787), anti-xCT (1:5,000; Abcam; cat.
no. ab175186) and β-actin (1:1,000; Abcam; cat. no. ab8227). After
primary antibody incubation, the membranes were washed three times
with TBST for 10 min each and then incubated with horseradish
peroxidase-conjugated secondary antibodies [1:5,000; Beijing
Huaxing Bocheng Gene Technology Co., Ltd.; cat. nos. HX2031
(rabbit) and HX2032 (mouse)] for 1 h at room temperature. The
membranes were washed again with TBST three times for 10 min each.
Immunoreactive bands were visualized using BeyoECL Plus (Beyotime
Biotechnology) according to the manufacturer's instructions. The
chemiluminescent signals were detected and captured using the
ChemiDoc XRS+ imaging system (Bio-Rad Laboratories, Inc.).
Densitometric analysis of the bands was performed using ImageJ2X
software (National Institutes of Health). The expression levels of
target proteins were normalized to β-actin as the loading
control.
VSC4.1 cell culture
The effects of DFO on the expression of the
Nrf2/HO-1 signaling pathway were validated with an in vitro
erastin-induced neuronal ferroptosis model using the rat VSC4.1
cell line, created as previously described (18). VSC4.1 is an immortalized motor
neuron-like hybrid cell line that has been described as a
motoneuron-neuroblastoma fusion product (28). The cells were purchased from
Hunan Fenghui Biotechnology Co., Ltd. (cat. no. CL0674) and
cultured at 37°C with 5% CO2 in Dulbecco's Modified
Eagle Medium (Procell Life Science & Technology Co., Ltd.; cat.
no. PM150220) supplemented with 10% fetal bovine serum (Gibco;
Thermo Fisher Scientific, Inc.) and 1% penicillin-streptomycin. The
VSC4.1 cells were divided into four groups: The Control, Erastin,
DFO and ML385 groups. In the Erastin group, VSC4.1 cells were
treated with erastin (10 µM) for 24 h. In the DFO group,
VSC4.1 cells were treated with a combination of erastin (10
µM) and DFO (10 µM) for 24 h. For the ML385 group, an
inhibitor of the Nrf2 signaling pathway, ML385 (5 µM), was
also added to DFO treatment for 24 h (29). The Control group was cultured in
culture medium without any additional treatment. The Control group
served as a baseline to evaluate the effects of erastin-induced
ferroptosis and the subsequent protective effects of DFO and the
impact of Nrf2 inhibition by ML385. All other culture conditions
and parameters remained consistent across all experimental
groups.
Immunofluorescence staining
Immunofluorescence staining of spinal cord tissue
was performed on sections obtained from 6 mice per group at the
indicated time points. Frozen spinal cord sections were allowed to
equilibrate to room temperature for 1 h. Sections were then washed
three times with PBS for 5 min each to remove any residual
embedding medium. The sections were permeabilized and blocked by
incubating them in a solution containing 0.5% Triton X-100 and 1%
bovine serum albumin (BSA) (Thermo Fisher Scientific, Inc.) in PBS
for 2 h at room temperature. This step reduces non-specific
antibody binding and permeabilizes the tissue to allow antibody
penetration. After blocking, the sections were incubated with the
following primary antibodies diluted in PBS containing 1% BSA:
Anti-Nrf2 (1:250; CST Biological Reagents Co., Ltd.; cat. no.
12721S), anti-GPX4 (1:300; Abcam; cat. no. ab41787) and anti-Tuj1
(1:250; Abcam; cat. no. ab78078). The sections were incubated with
the primary antibodies overnight at 4°C to ensure optimal binding.
The next day, sections were washed three times in PBS (5 min each)
and then incubated with fluorophore-conjugated secondary antibodies
diluted in PBS containing 1% BSA for 2 h at room temperature in the
dark, including Alexa Fluor 488-conjugated goat anti-rabbit IgG
(H+L) (1:500; Abcam; cat. no. ab150077) and Rhodamine
(TRITC)–conjugated goat anti-mouse IgG (H+L) (1:500; Proteintech
Group, Inc.; cat. no. SA00007-1). Following secondary antibody
incubation, the sections were washed three times with PBS for 5 min
each. The sections were then stained with
4′,6-diamidino-2-phenylindole (DAPI) for 10 min at room temperature
to label cell nuclei. After the final wash with PBS, the sections
were mounted with an antifade mounting medium to preserve
fluorescence and minimize photobleaching. The stained sections were
visualized and imaged using a laser scanning confocal microscope
(Nikon Corporation). Z-stack images were captured for
three-dimensional analysis where necessary. Representative images
were analyzed using ImageJ software (National Institutes of
Health), and fluorescence intensity was quantified to assess
protein expression levels.
For cellular experiments, the ventral spinal cord
4.1 (VSC4.1) cells were seeded into 24-well plates pre-coated with
poly-D-lysine (Sigma-Aldrich; Merck KGaA). After the experimental
treatments, the cells were washed three times with 1X PBS, 5 min
per wash, to remove any residual medium and treatment agents. Cells
were fixed with 4% PFA for 15 min at room temperature to preserve
cellular structure. Following fixation, cells were permeabilized
and blocked with a solution containing 0.1% Triton X-100 and 0.5%
BSA in PBS for 30 min at room temperature to reduce non-specific
binding. The fixed and permeabilized cells were incubated with the
aforementioned primary antibodies diluted in PBS containing 1% BSA.
The cells were incubated overnight at 4°C to allow adequate
antibody binding. After three washes with PBS, the cells were
incubated with the aforementioned fluorophore-conjugated secondary
antibodies diluted in PBS with 1% BSA for 1 h at room temperature
in the dark. Cells were stained with DAPI for 10 min at room
temperature to label nuclei. After the final washes, the cells were
kept in PBS for imaging. Images were captured using a confocal
microscope and representative fields were analyzed with ImageJ
software for fluorescence intensity quantification.
Hematoxylin and eosin (H&E) and Nissl
staining
At 28 days post-injury, spinal cord tissues (n=6 per
group) were harvested and subjected to standard histopathological
processing for H&E and Nissl staining according to established
protocols.
For H&E staining, first, the cryopreserved
spinal cord sections were equilibrated to room temperature for
thawing. Nuclear staining was then performed by incubation in
hematoxylin solution (Beijing Solarbio Science & Technology
Co., Ltd.) (3 min at room temperature), resulting in characteristic
blue-purple nuclear chromatin visualization. Following
differentiation, cytoplasmic counterstaining was achieved using
eosin solution for 1 min at room temperature to demonstrate
pink-red cytoplasmic components. Subsequent dehydration was
performed through a graded ethanol series, with xylene clearing for
tissue transparency. Sections were permanently mounted using
neutral resin mounting medium prior to brightfield microscopic
examination. Histological damage scoring based on edema, neutrophil
infiltration and hemorrhage was as follows: 0 points, asymptomatic
or mild; 1 point, limited; 2 points, moderate; 3 points, prominent;
and 4 points, extensive. For each section, the three components
were scored separately and then averaged (range 0-4) to obtain an
overall damage score. Scores were independently calculated by two
pathologists and averaged to obtain the final values.
For Nissl staining, after similar thawing
procedures, sections were processed using standardized Nissl
staining reagents (Wuhan Servicebio Technology Co., Ltd.). The
staining protocol included differentiation in glacial acetic acid
solution to optimize Nissl staining contrast. Nissl staining was
performed by incubating sections in cresyl violet for 5 min at room
temperature. Following air-drying, sections were similarly mounted
with neutral resin for microscopic evaluation of neuronal
architecture and Nissl staining distribution.
Source of the gene datasets, data
collection and analyses
The present study performed bioinformatics analyses
using the publicly available dataset GSE162610 (https://www.ncbi.nlm.nih.gov/geo/) deposited in
the NCBI Gene Expression Omnibus (GEO) database, which contains
single-cell RNA sequencing data from uninjured and injured mouse
spinal cords at 1, 3, and 7 days post-injury, originally generated
by Milich et al (30).
The raw expression matrix was processed and analyzed in the R
programming environment (version 4.3.1; R Core Team; https://www.r-project.org/) for subsequent statistical
analysis. The following programs and their respective versions were
used for each analysis: readxl (version 1.4.5, for reading Excel
files), ggplot2 (version 4.0.1, for data visualization), cluster
Profiler (version 4.2.2, Bioconductor Release 3.13, for
differential expression analysis and functional enrichment), enrich
plot (version 1.30.4, Bioconductor, for visualizing enrichment
analysis results) and org.Mm.eg.db (version 3.22.0, Bioconductor,
for mouse genome annotation). Differentially expressed genes (DEGs)
were identified by comparing the gene expression levels between the
injury and control groups using a pairwise quasi-likelihood F-test.
A significance threshold of P<0.05 was applied for statistical
analysis and the pseudo-bulk method was used for differential
analysis. Based on the selected DEGs, Gene Ontology (GO) and Kyoto
Encyclopedia of Genes and Genomes (KEGG) pathway enrichment
analyses were conducted and volcano plots were generated,
highlighting genes specifically associated with ferroptosis.
Furthermore, a protein-protein interaction (PPI) network was
constructed using the STRING database (https://string-db.org/) (limited to Mus
musculus) to obtain interaction data, which was then imported
into Cytoscape for network visualization and topological analysis.
Key node proteins were selected for further analysis. All figures
were generated using GraphPad Prism 9.4.0 (Dotmatics).
Statistical analysis
Data were analyzed using GraphPad Prism (version
9.3; Dotamatics). All data are presented as the mean ± standard
deviation. For single time-point outcomes involving more than two
groups, one-way ANOVA was applied when assumptions of normality and
homogeneity of variance were met, followed by Bonferroni post hoc
testing for multiple comparisons. For two-group comparisons at a
single time point in which the data were not normally distributed,
the Mann-Whitney U test was used. For locomotor recovery, BMS
scores were analyzed using a two-way repeated-measures ANOVA across
the full time course, with treatment group as the between-subject
factor and time (post-injury day) as the within-subject repeated
factor, including assessment of the group × time interaction. When
significant effects were detected, Tukey's post hoc test was
performed to compare treatment groups at the same post-injury day.
P<0.05 was considered to indicate a statistically significant
difference.
Results
DFO protects neurons and activates the
Nrf2/HO-1 signaling pathway after SCI
To assess changes in Nrf2/HO-1 expression following
SCI and treatment with DFO in mice, western blotting was employed.
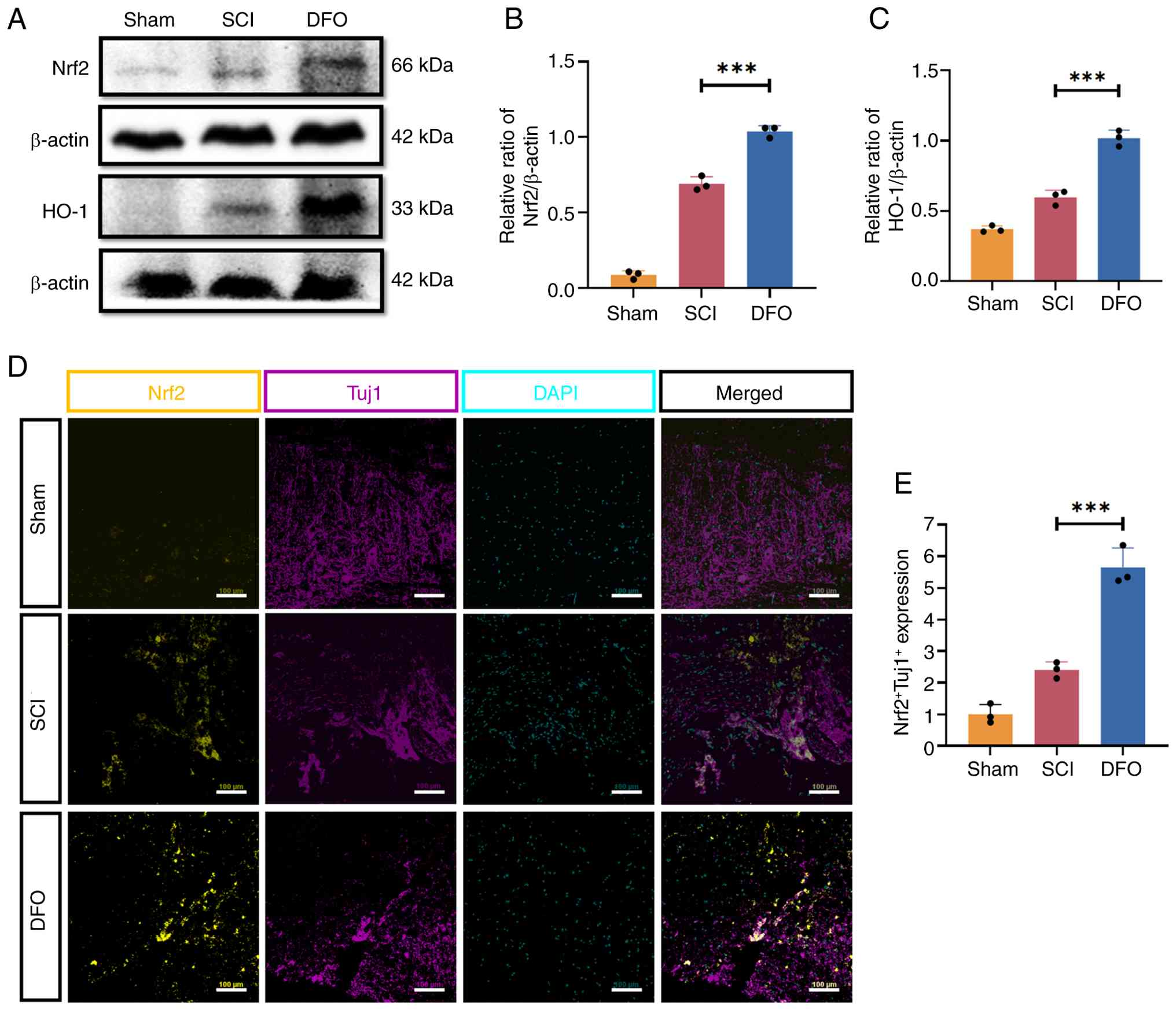
As depicted in Fig. 1A-C, the
expression of Nrf2 and HO-1 in the DFO-treated group significantly
increased compared with the SCI group. Notably, the Nrf2/HO-1
expression levels showed a trend toward upregulation in the SCI
group compared with the Sham group (P>0.05), suggesting a
possible endogenous compensatory response to injury-associated
oxidative stress. However, this trend was not statistically
significant. Moreover, immunofluorescence staining demonstrated a
significant increase in neuronal Nrf2 expression (Tuj1+
cells) in the DFO-treated group compared with the SCI group at 2
days post-SCI (P<0.05; Fig. 1D
and E).
DFO confers neuroprotection and enhances
the expression of the Nrf2/HO-1 signaling pathway in an
erastin-induced neuronal ferroptosis model
Neurons play a pivotal role in the pathogenesis of
SCI and represent a potential target for treatment strategies.
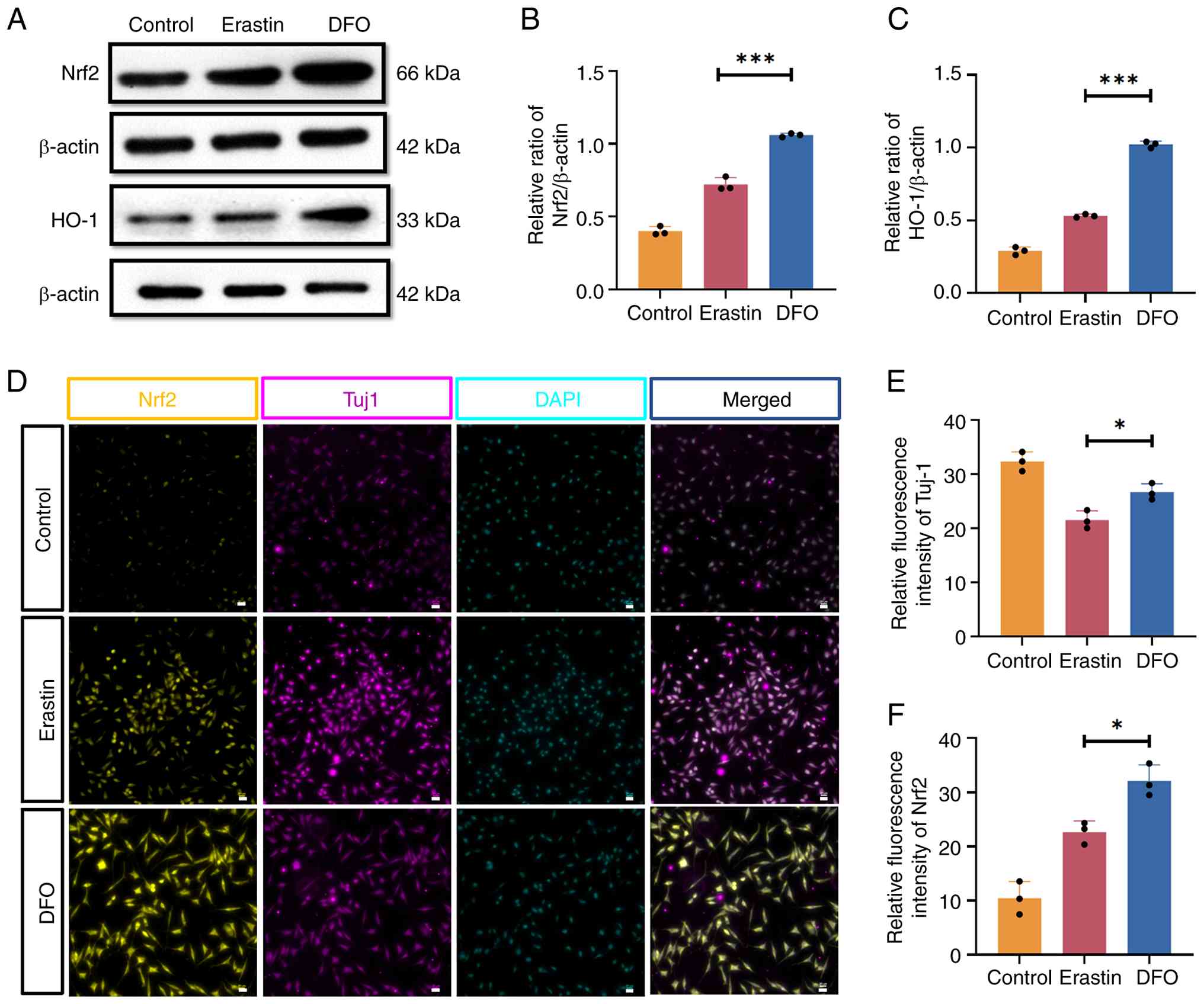
Western blotting analysis of VSC4.1 cells revealed that the
expression levels of Nrf2 and HO-1 were significantly elevated in
the DFO treatment group compared with the Erastin group (Fig. 2A-C). Similarly, erastin treatment
tended to increase Nrf2 and HO-1 expression compared with the
Control group; however, the differences did not reach statistical
significance (P>0.05) (Fig.
2A-C). Immunofluorescence staining further demonstrated the
neuroprotective effect of DFO on VSC4.1 cells. Specifically, Tuj1
(βIII-tubulin), a marker of neuronal phenotype and
cytoskeletal/neurite integrity, was markedly reduced after Erastin
exposure, whereas DFO treatment preserved Tuj1 signal (and/or the
proportion of Tuj1+ cells), indicating improved
maintenance of neuronal structural identity (Fig. 2D-F).
DFO attenuates neuronal ferroptosis by
modulating the xCT/GPX4 antioxidant system via activating the
Nrf2/HO-1 signaling pathway in SCI mice
Previous investigations from our laboratory have
demonstrated the efficacy of DFO in preventing neuronal ferroptosis
by governing the xCT/GPX4 antioxidant system (12). However, the participation of the
Nrf2/HO-1 signaling pathway in this process remains elusive. To
address this question, ML385, an Nrf2 inhibitor, was employed to
elucidate the role of Nrf2 in the DFO-mediated treatment of SCI in
mice. The experimental design is outlined in Fig. 3A.
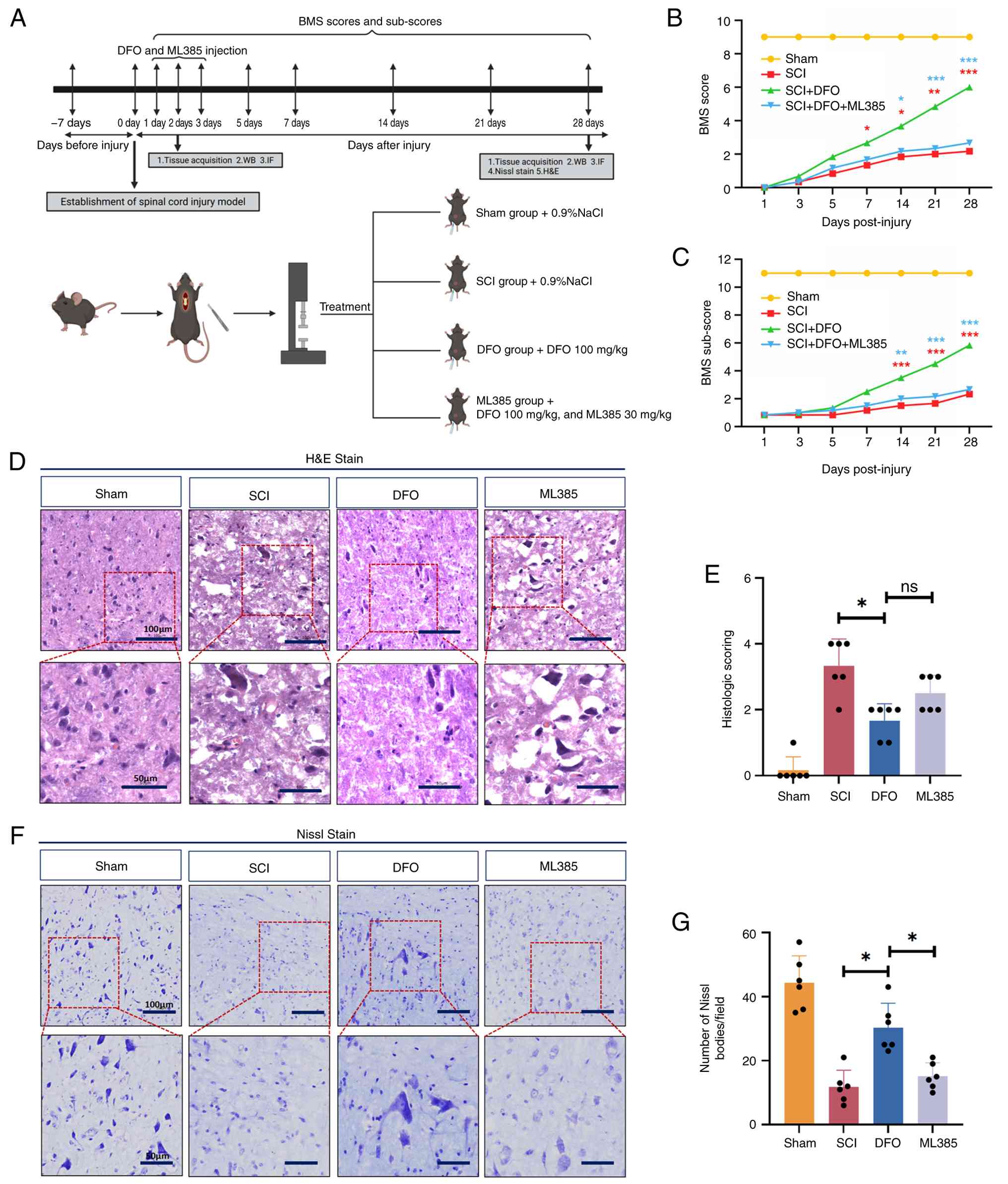 | Figure 3Nrf2 inhibitor ML385 reverses the
effects of DFO on the functional and histological recovery of SCI
mice. (A) Schematic diagram of the in vivo experimental
design. (B) BMS score and (C) sub-score were applied to evaluate
the recovery of motor function in each group at 1-, 3-, 5-, 7-,
14-, 21- and 28-days post-injury. Data are presented as the mean ±
SD (n=6 per group). (D) Representative H&E-stained longitudinal
sections of spinal cords from different groups (scale bars, 100 and
50 µm). (E) Quantification of the histopathological changes
scored based on edema, neutrophil infiltration and hemorrhage. Data
are presented as the mean ± SD (n=6 per group) and were analyzed
using one-way ANOVA followed by Bonferroni post hoc test. (F) Nissl
staining to observe the number of motor neurons in the anterior
horn of the spinal cord (scale bars, 100 and 50 µm). (G)
Quantitative analysis of the Nissl body counts. Data are presented
as the mean ± SD (n=6 per group) and were analyzed using one-way
ANOVA followed by Bonferroni post hoc test. *P<0.05,
**P<0.01, ***P<0.001. ns, not
significant; DFO, deferoxamine; SCI, spinal cord injury; BMS, Basso
Mouse Scale; H&E, hematoxylin and eosin; WB, western blotting;
IF, immunofluorescence. |
The BMS was utilized to assess open-field locomotion
in SCI mice with assessments conducted at various time points
post-injury. At 1 day post-injury, which was the first behavioral
assessment time point, the BMS score was 0, supporting reliable
establishment of the SCI model. Subsequent assessment revealed that
the scores and sub-scores of mice in the DFO group were
significantly higher than those in the SCI group and ML385 group at
days 14, 21 and 28 post-injury (P<0.05). However, no significant
differences were observed between the SCI group and ML385 group
(Fig. 3B and C), suggesting that
ML385 administration reversed the beneficial effects of DFO on
functional recovery in SCI mice.
H&E staining showed that the SCI group had
significant spinal cord damage, hemorrhage and edema compared with
the DFO group at 28 days post-injury (P<0.05). ML385
administration appeared to attenuate spinal cord recovery compared
with the DFO group, but this effect did not reach statistical
significance (Fig. 3D and E).
Nissl staining revealed that the SCI group exhibited significant
tissue loss and neuronal damage. By contrast, the DFO group
exhibited improved tissue integrity and reduced neuronal damage.
However, these beneficial effects were diminished in the
ML385-treated group (P<0.05; Fig.
3F and G). These findings suggest that DFO-Nrf2 signaling
pathway may mitigate tissue loss and neuronal injury following
SCI.
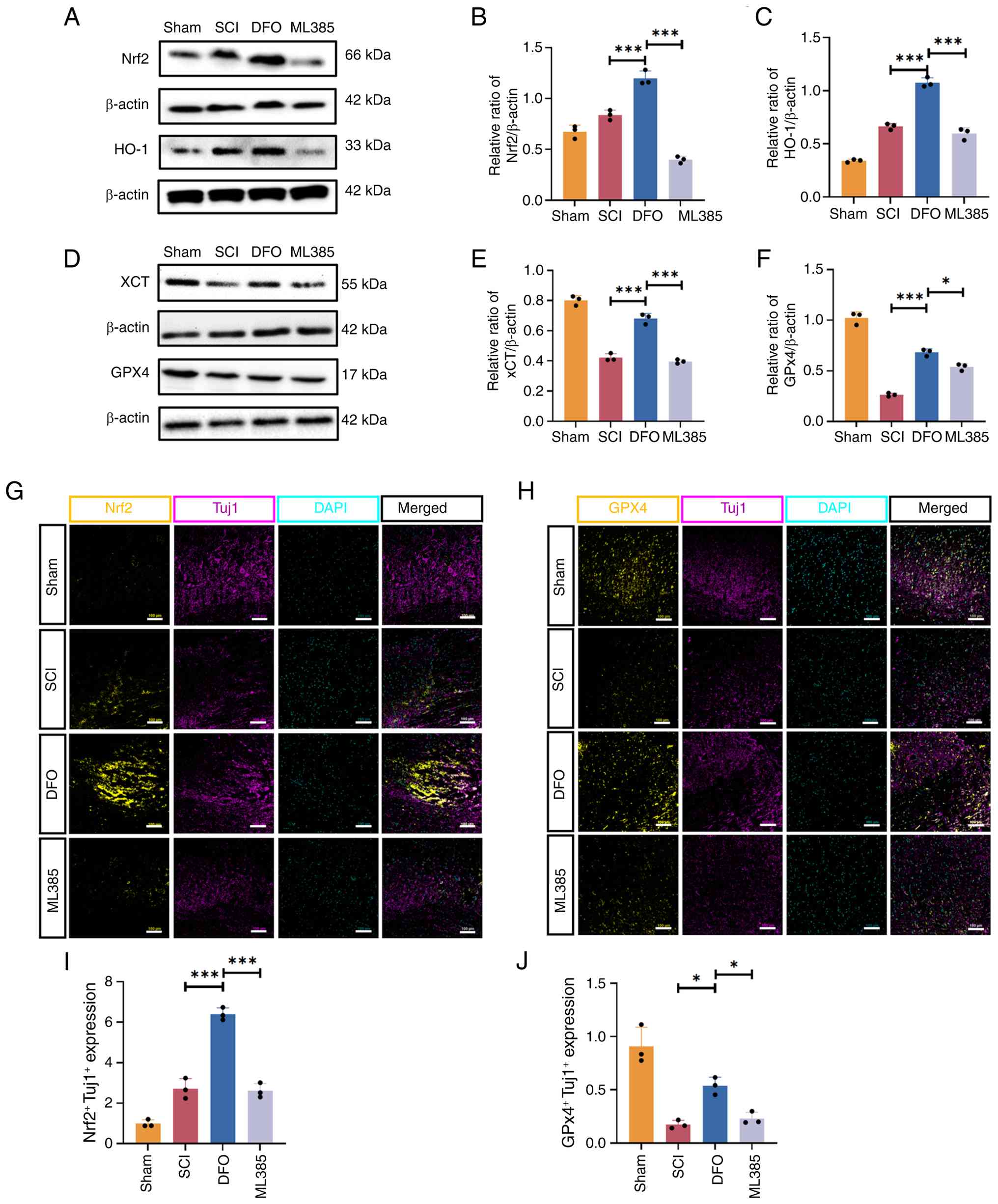
Further examination via western blotting revealed
that the expression levels of Nrf2 and HO-1 were significantly
downregulated in the ML385 group compared with the DFO group
(Fig. 4A-C). Conversely, DFO
treatment led to a significant upregulation in the expression of
xCT and GPX4 compared with the SCI group. However, the protein
levels of xCT and GPX4 in the ML385 group were reduced compared
with the DFO group (Fig. 4D-F),
indicating a reversal of the effects of DFO by ML385.
Immunofluorescence analysis further supported these
findings, evidenced by a significant decrease in the number of
Nrf2+Tuj1+ neurons in the ML385 group
compared with the DFO group (Fig. 4G
and I). Additionally, DFO treatment significantly upregulated
the expression of GPX4 in Tuj1+ neurons in spinal cord
tissue, while ML385 administration attenuated this effect (Fig. 4H and J), further highlighting the
role of Nrf2 in regulating the neuroprotective effects of DFO.
DFO inhibits erastin-induced neuronal
ferroptosis by regulating the xCT/GPX4 antioxidant system via
activation of the Nrf2/HO-1 signaling pathway
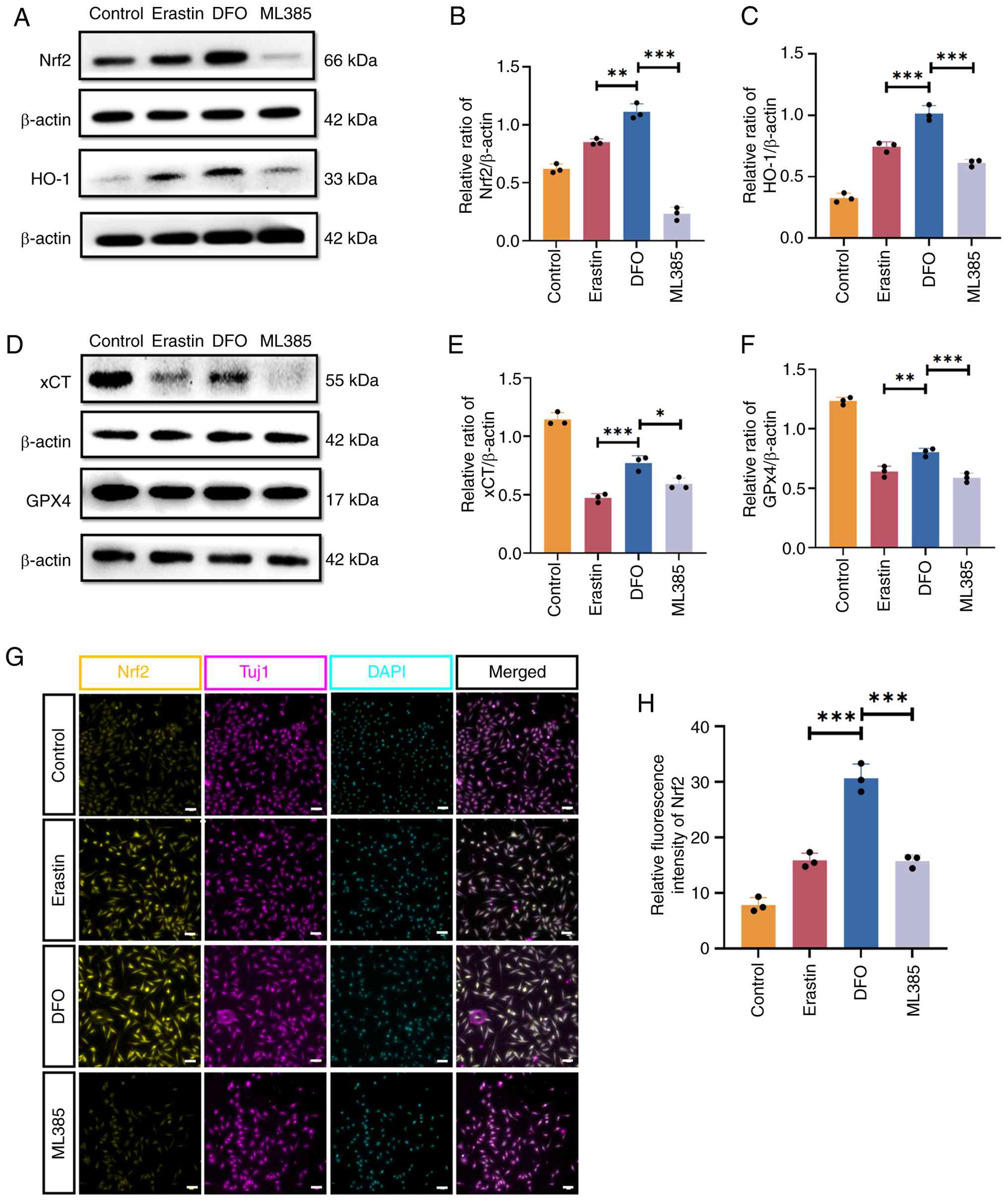
To elucidate the involvement of the Nrf2/HO-1
signaling pathway in DFO intervention in vitro, ML385 was
introduced into VSC4.1 cells. Western blotting analysis revealed
that the expression levels of Nrf2 and HO-1 were downregulated in
the ML385 group compared with the DFO group (Fig. 5A-C). Additionally, the levels of
the ferroptosis markers xCT and GPX4 were significantly higher in
the DFO group compared with the Erastin group. However, following
ML385 treatment, the expression of xCT and GPX4 was notably
downregulated compared with the DFO group (Fig. 5D-F). Immunofluorescence analysis
further corroborated these findings, demonstrating a significant
upregulation of Nrf2 expression in VSC4.1 cells after DFO treatment
compared with the Erastin group, while ML385 administration
attenuated the levels of Nrf2 compared with the DFO group (Fig. 5G and H).
Analysis of a single-cell RNA sequencing
dataset reveals dynamic activation of ferroptosis-related pathways
after SCI
An unbiased overview of transcriptional alterations
associated with SCI was obtained by analyzing the public dataset
GSE162610, in which spinal cord tissues collected at 1, 3 and 7
days post-injury were compared with those from uninjured controls.
KEGG pathway enrichment analysis demonstrated a clear
time-dependent shift in biological programs following SCI. At 1 day
post-injury, DEGs were mainly enriched in the MAPK, PI3K-AKT, TNF
and NF-κB signaling pathways, together with pathways associated
with apoptosis and HIF-1 signaling, reflecting an acute stress and
inflammatory response (Fig.
S1D). At 3 and 7 days post-SCI, enrichment progressively
shifted toward immune-related pathways, including
'Cytokine-cytokine receptor interaction', 'Chemokine signaling
pathway' and 'Leukocyte transendothelial migration'. Notably, the
KEGG pathway 'Ferroptosis' was significantly enriched at 3 days
(subacute phase) and 7 days (early chronic phase) post-SCI,
suggesting persistent involvement of iron-dependent lipid
peroxidation in SCI pathophysiology (Fig. 6A and B).
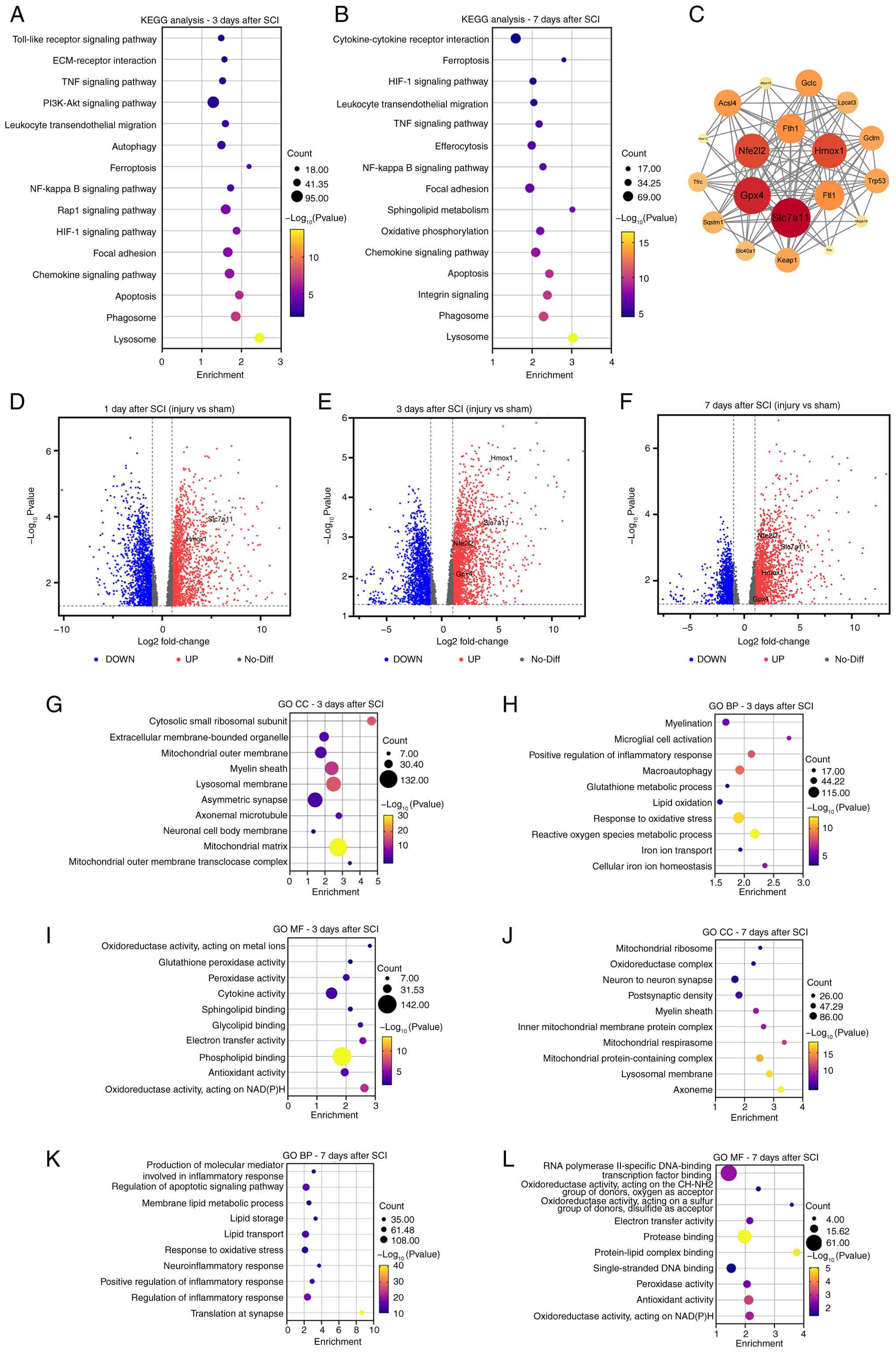 | Figure 6Bioinformatics analysis of DEGs in
mouse SCI models. KEGG pathway enrichment analysis for (A) 3 and
(B) 7 days post-SCI. (C) Protein-protein interaction network of key
ferroptosis-related proteins. Volcano plots showing the DEGs at (D)
1-, (E) 3- and (F) 7-days post-SCI. (G-L) GO enrichment analysis of
CC, BP and MF at 3- and 7-days post-SCI. Statistical analysis was
performed using a pairwise quasi-likelihood F-test, with a
significance threshold of P<0.05, and the pseudo-bulk method was
used for differential analysis. DEGs, differentially expressed
genes; SCI, spinal cord injury; GO, Gene Ontology; KEGG, Kyoto
Encyclopedia of Genes and Genomes; CC, cellular components; BP,
biological processes; MF, molecular functions. |
To further functionally characterize
ferroptosis-associated transcriptional alterations after SCI, GO
enrichment analysis was performed for DEGs at 1, 3 and 7 days
post-injury. The predominant acute stress response at 1 day
post-SCI was evidenced by the significant enrichment of GO
biological process terms including 'response to oxidative stress'
and 'regulation of inflammatory response' (Fig. S1B), together with molecular
function terms such as 'ferrous iron binding' and 'peroxidase
activity' (Fig. S1A and C). At
3 and 7 days post-SCI, GO enrichment revealed a more notable and
consistent involvement of ferroptosis-related biological processes
(Fig. 6G-L). In the biological
processes category, enriched terms included 'response to oxidative
stress', 'lipid oxidation', 'glutathione metabolic process' and
'cellular iron ion homeostasis'. In the cellular components
category, DEGs were predominantly localized to 'mitochondrial
matrix' and 'lysosomal membrane', suggesting sustained
mitochondrial dysfunction and lipid turnover. Correspondingly,
molecular functions analysis highlighted enrichment of 'glutathione
peroxidase activity', 'antioxidant activity' and 'oxidoreductase
activity, acting on metal ions'. Collectively, these GO enrichment
patterns indicate persistent disruption of redox balance, iron
metabolism and lipid peroxidation processes at 3 days (subacute
phase) and 7 days (early chronic phase) post-SCI, which are
hallmark features of ferroptosis.
Volcano plot analysis illustrated the differential
gene expression profiles between injured and uninjured spinal cord
tissues at 1, 3 and 7 days after SCI (Fig. 6D-F). At 1 day post-injury,
widespread transcriptional changes were observed, reflecting an
acute injury response, whereas ferroptosis-related genes showed
relatively limited changes. At 3 and 7 days post-SCI, several
ferroptosis-related genes showed robust and consistent
upregulation. Intersection of the DEGs with curated ferroptosis
gene sets identified four representative markers: xCT (Slc7a11),
GPX4, Hmox1 and Nfe2l2 (Nrf2), all of which were significantly
upregulated at these later time points, reflecting coordinated
activation of antioxidant defense and iron metabolism at 3 and 7
days post-SCI (Fig. 6D-F).
Therefore, these genes were selected for further analysis. Temporal
expression analysis demonstrated dynamic regulation of these genes
after SCI (Fig. S1E). Slc7a11,
encoding the cystine/glutamate antiporter xCT, was markedly
upregulated at 1 day post-SCI, suggesting an early compensatory
response aimed at maintaining GSH synthesis. GPX4, a key lipid
peroxide scavenger, showed sustained induction at 3 days post-SCI.
By contrast, Hmox1 exhibited notable upregulation at later time
points, reflecting persistent oxidative stress and iron metabolism
dysregulation. Nfe2l2 showed time-dependent activation, consistent
with its role as a master regulator of antioxidant and
anti-ferroptotic responses. To further explore the regulatory
relationships among ferroptosis-associated genes, a PPI interaction
network was constructed (Fig.
6C). Network analysis revealed a highly interconnected
structure centered on Nfe2l2, Slc7a11 and GPX4, with extensive
interactions involving proteins related to iron storage and
transport (such as Fth1, Ftl1 and Tfrc), lipid peroxidation (such
as Acsl4, Alox12 and Alox15) and GSH metabolism (such as Gclc, Gclm
and Gss). After SCI, ferroptosis-related proteins showed close
functional associations, centering on the Nrf2/xCT/GPX4 pathway
rather than acting in isolation.
Discussion
The present study explored the role of the Nrf2/HO-1
signaling pathway in mediating the inhibitory effects of DFO on
neuronal ferroptosis following SCI. The findings revealed an
upregulation of Nrf2 and HO-1 expression after SCI and DFO
treatment in mice, corroborated by in vitro experiments
demonstrating increased Nrf2 and HO-1 levels following DFO
administration in an erastin-induced neuronal ferroptosis model.
Employing the Nrf2 inhibitor ML385, it was demonstrated that
blocking Nrf2 reversed the inhibitory effects of DFO on ferroptosis
in both SCI mice and VSC4.1 cells. Notably, ML385 also attenuated
the functional recovery of SCI mice under DFO treatment.
Collectively, these findings underscore the role of DFO in
promoting functional recovery after SCI by mitigating neuronal
ferroptosis through activation of the Nrf2/HO-1 signaling pathway
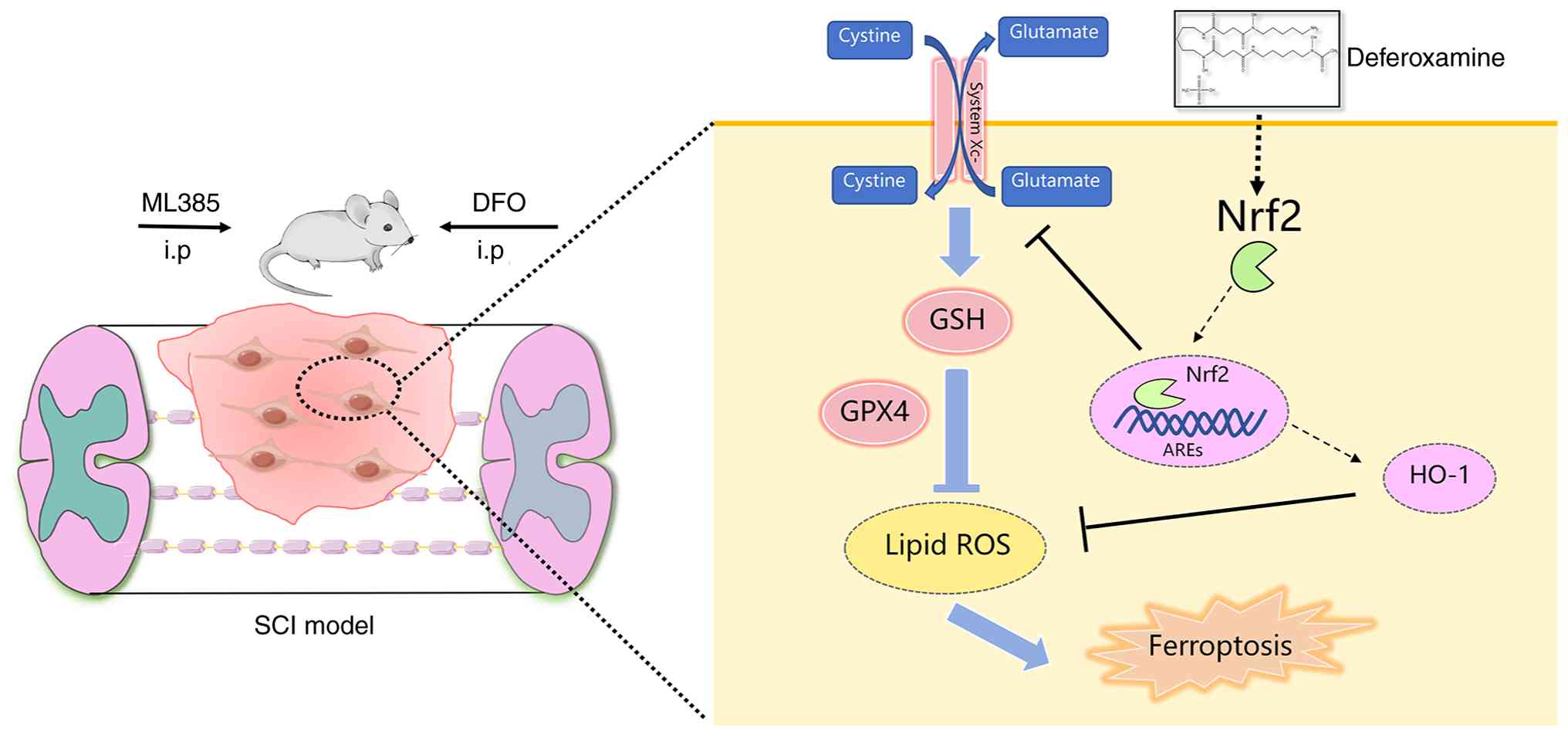
(Fig. 7).
Ferroptosis, a recently identified type of cell
death, has emerged as a critical contributor to SCI pathology, as
substantiated by our prior research (12) and other investigations in the
field (6,14). DFO, an iron chelator, has shown
potential in enhancing functional and histological recovery after
SCI by mitigating ferroptosis (12). Expanding upon these findings, our
previous study further delineated the inhibitory effects of DFO on
erastin-induced neuronal ferroptosis in vitro (17). The role of ferroptosis in SCI has
gained increasing attention. Ni et al (20) demonstrated that resveratrol
promotes functional recovery and attenuates ferroptosis post-SCI.
Zou et al (16) reported
the promotion of neuronal ferroptosis and M1 macrophage
polarization in SCI, highlighting the potential inhibitory role of
protein arginine methyltransferase 8 via regulation of glial
cell-derived neurotrophic factor expression. Additionally, Li et
al (31) provided evidence
suggesting that dihydroorotate dehydrogenase, a potent inhibitor of
ferroptosis in tumor cells, may also play a role in mitigating
neuronal ferroptosis following SCI (32). Given the mounting evidence
implicating ferroptosis in SCI pathogenesis, targeting this pathway
presents a promising avenue for therapeutic intervention.
To advance the clinical translation of drug therapy
for SCI, it is imperative to elucidate the molecular mechanisms
comprehensively. The present study provides evidence that DFO
fosters functional recovery and mitigates neuronal ferroptosis by
modulating the Nrf2/HO-1 signaling pathway following SCI. Notably,
the Nrf2/HO-1 pathway is well recognized in SCI research. For
instance, curcumin, a medicinal plant agent, has been reported to
mitigate inflammatory damage by scavenging free radicals and
activating Nrf2/HO-1 to facilitate SCI repair (33). Moreover, deficiency in the
Pregnane X receptor has been shown to activate the Nrf2/HO-1
pathway, conferring protection against SCI (34). Several studies have explored the
role of Nrf2 in regulating ferroptosis (19,35). Gong et al (19) concluded that trehalose inhibits
ferroptosis through the Nrf2/HO-1 pathway, promoting functional
recovery in SCI mice. In addition to its involvement in
ferroptosis, Nrf2 also modulates other biological processes in SCI,
such as microglial polarization, neuroinflammation and oxidative
stress (36,37). Notably, the expression of Nrf2
and HO-1 were upregulated following SCI in the present study. This
might be attributed to the body's response to increased oxidative
stress and inflammation, which are key components of the secondary
injury phase after SCI. Hence, the upregulation of Nrf2 and HO-1
after SCI might represent the body's attempt to limit the extent of
damage and promote recovery. Although the present study did not
directly investigate these processes, it highlights the need for
further research into the regulatory effects of DFO on various
biological processes through the activation of Nrf2/HO-1. Such
investigations could provide deeper insights into the multifaceted
therapeutic mechanisms underlying DFO treatment in SCI. Besides,
although Nrf2/HO-1 is a well-known pathway involved in oxidative
stress responses, its precise role in the context of the inhibitory
effects of DFO on ferroptosis after SCI has not been fully
elucidated. The present study advances the current understanding by
providing novel insights into how DFO modulates this critical
pathway to mitigate neuronal damage in SCI. To the best of our
knowledge, the present study is the first to demonstrate that the
efficacy of DFO in reducing ferroptosis may be closely linked to
the activation of Nrf2/HO-1, which may subsequently lead to
enhanced antioxidant defense mechanisms and improved functional
recovery. This novel mechanistic insight not only deepens our
understanding of the therapeutic action of DFO but also opens new
avenues for targeted interventions in SCI treatment, making the
findings both innovative and notable in the field of
neuroprotection.
The spinal cord tissue comprises several types of
cells, including neurons, astrocytes, oligodendrocytes and
microglia. Notably, all these cell types are susceptible to
ferroptosis, a form of regulated cell death characterized by lipid
peroxidation. For instance, in Alzheimer's disease, NADPH oxidase 4
induces ferroptosis in astrocytes through oxidative stress-induced
lipid peroxidation, leading to mitochondrial dysfunction (38). Similarly, ferroptosis in
oligodendrocyte progenitor cells contributes to white matter injury
following hemorrhagic stroke, suggesting that targeting ferroptotic
oligodendrocytes could be a therapeutic approach for stroke and
related conditions (39). Spinal
cord neurons play crucial roles as cellular connections between the
brain and the body, influencing behavior regulation and
pathological conditions. Given their importance, neuronal survival
or loss directly impacts functional recovery in SCI. Therefore, the
present study primarily focused on investigating ferroptosis in
neurons. To assess ferroptosis in spinal cord neurons, Tuj1 was
utilized as a marker for neuronal labeling. The findings revealed
an increase in GPX4+Tuj1+ neurons in spinal
cord tissue following DFO administration, indicative of a
protective effect against ferroptosis. Several studies have shown
that neuronal ferroptosis plays a vital role in SCI pathology
(40,41). Conversely, interference with Nrf2
using ML385 led to a decrease in GPX4+Tuj1+
neurons, suggesting a disruption of the protective mechanism. In
the present study, the VSC4.1 cell line, a hybrid motor neuron cell
line created through the fusion of a mice ventral spinal cord
neuron with a mouse neuroblastoma cell line (42), was employed to establish a
neuronal ferroptosis model using erastin. The results demonstrated
that under the experimental conditions used in the present study
(Control and erastin ± DFO ± ML385), VSC4.1 cells were
Tuj1-positive and showed detectable Nrf2 and GPX4 expression,
supporting their use as an in vitro neuronal model for
ferroptosis-related analyses. However, it is essential to
acknowledge that the focus of the present study on neuronal
ferroptosis has limited the comprehensive understanding of
ferroptosis processes in other cell types following SCI. In
addition to neurons, accumulating data indicate that ferroptosis in
glial cells also contributes to the complex pathology of SCI
(43-45). Oligodendrocytes, which are
enriched in polyunsaturated phospholipids and display relatively
high intracellular iron levels, are particularly susceptible to
ferroptotic injury after central nervous system trauma (46). Experimental SCI models have
demonstrated that iron accumulation and lipid peroxidation within
oligodendrocytes are closely associated with secondary white-matter
damage, and pharmacological inhibition of ferroptosis (such as by
ferrostatin-1 or liproxstatin-1) reduces oligodendrocyte loss,
alleviates white-matter injury and improves locomotor recovery
(47,48). Moreover, astrocytes and microglia
exhibit ferroptosis-related changes after SCI, and
astrocyte-specific activation of Nrf2 has been shown to attenuate
oxidative stress, demyelination and neurological dysfunction,
underscoring the importance of glial antioxidant and
ferroptosis-regulating pathways in shaping the injury milieu
(49). Given that DFO is a
potent iron chelator and ferroptosis inhibitor, its beneficial
effects in the SCI model established in the present study are
likely not restricted to neurons. Previous work has demonstrated
that systemic DFO treatment after SCI decreases iron overload,
restores GPX4/xCT/GSH expression, improves behavioral recovery and
concurrently enhances neuronal survival while inhibiting gliosis,
suggesting coordinated protection of both neuronal and glial
compartments (12). Together
with the present findings, these observations support the notion
that DFO may modulate ferroptosis across multiple spinal cord cell
types, which including neurons, oligodendrocytes and astrocytes,
through iron chelation and Nrf2-dependent antioxidant mechanisms.
Future studies should consider investigating ferroptosis across
various cell types to provide a more comprehensive acknowledgement
of its role in SCI pathogenesis.
The demonstration by the present study that DFO can
effectively mitigate neuronal ferroptosis and promote functional
recovery by activating the Nrf2/HO-1 pathway suggests that DFO or
similar agents could be integrated into current SCI treatment
paradigms. Given that DFO is an FDA-approved iron chelator
indicated for acute iron intoxication and transfusional iron
overload in patients with chronic anemia, its repurposing for SCI
treatment could offer a more expedited pathway toward clinical
translation. Beyond ferroptosis inhibition, DFO has also been
reported to attenuate key components of secondary injury after SCI,
including oxidative/nitrosative stress (such as reduced lipid
peroxidation/MDA and nitrotyrosine formation) and inflammation
(such as reduced neutrophil infiltration, NF-κB activation and iNOS
expression), thereby contributing to tissue protection and improved
functional outcomes in preclinical models (50). However, the clinical translation
of these findings will require careful consideration of several
challenges. First, the optimal dosing and timing of DFO
administration must be established to maximize therapeutic efficacy
while minimizing potential side effects, such as infusion-related
hypotension/hypersensitivity and dose-dependent auditory/ocular
toxicities, which warrant careful safety monitoring in
translational studies (51).
Additionally, the long-term effects of DFO treatment on
neuroregeneration and functional outcomes in patients with SCI need
to be thoroughly evaluated through clinical trials. Another
challenge lies in understanding the interaction of DFO with other
cell types within the spinal cord, such as astrocytes and
microglia, which also play crucial roles in SCI pathology.
Addressing these challenges through rigorous preclinical and
clinical studies will be essential for translating the findings of
the present study into effective clinical interventions.
Previous transcriptomic analyses of public datasets
have shown that multiple regulated cell death pathways, including
pyroptosis, necroptosis and hypoxia-related injury, are actively
involved in shaping the post-injury microenvironment and
inflammatory response after SCI (52-55). To further characterize
ferroptosis over time, the present study applied a time-resolved,
ferroptosis-centered transcriptomic analysis based on a publicly
available GEO dataset (GSE162610), including differential
expression analysis, KEGG enrichment, GO annotation and PPI network
construction. At 1 day (acute phase), 3 days (subacute phase) and 7
days (early chronic phase) post-SCI, ferroptosis-associated
signaling was consistently linked to oxidative stress, lipid
peroxidation, iron metabolism dysregulation and mitochondrial
dysfunction, which are hallmark features of secondary injury
following SCI. KEGG analysis showed a progressive enrichment of
ferroptosis-related pathways after injury, while GO analysis
further linked these changes to GSH metabolism, antioxidant
defense, iron homeostasis and mitochondrial and lysosomal
functions. Notably, the PPI network further revealed that these
diverse processes are integrated through a tightly connected
regulatory core centered on the Nrf2/xCT/GPX4 axis. Within this
network, Nfe2l2 (Nrf2) occupies a central transcriptional position
linking upstream redox sensing (Keap1) with downstream antioxidant
and ferroptosis-suppressing targets (Slc7a11, Gpx4 and Hmox1). This
systems-level organization suggests that activation of
Nrf2-dependent signaling represents an endogenous compensatory
mechanism aimed at counteracting ferroptosis after SCI. However, in
the time-resolved analyses, despite upregulation of
antioxidant/anti-ferroptotic genes (such as Nfe2l2/Nrf2,
Slc7a11/xCT, Gpx4 and Hmox1), the KEGG term 'Ferroptosis' remained
significantly enriched at 3 and 7 days post-SCI and GO enrichment
continued to highlight 'lipid oxidation' and 'cellular iron ion
homeostasis'. Together with the PPI network implicating
iron-handling and lipid-peroxidation nodes (such as Fth1/Ftl1/Tfrc,
Acsl4 and Alox12/15), these findings suggest that endogenous
defenses are insufficient to fully restrain persistent
ferroptosis-associated injury. Given that DFO is a potent iron
chelator and ferroptosis inhibitor, we hypothesized that its
neuroprotective effects after SCI might be mediated through
activation of the Nrf2/HO-1/xCT/GPX4 axis. Accordingly,
complementary in vivo and in vitro experiments were
performed to test the causal involvement of ferroptosis and Nrf2
signaling in neuronal protection after SCI. When integrated with
the transcriptomic evidence of regulated cell-death programs, the
present study supports a mechanistic model in which DFO confers
neuroprotection primarily by suppressing ferroptotic injury through
Nrf2-dependent antioxidant regulation, with downstream responses
becoming particularly prominent during the subacute phase (3 days
post-SCI). Although the xCT/GPX4 axis was validated experimentally,
the relative contributions and interactions of additional
Nrf2-regulated targets remain to be clarified. Future work using
cell-type-resolved profiling (such as single-cell or spatial
transcriptomics) together with targeted perturbation of prioritized
downstream nodes will help define the regulatory hierarchy of
Nrf2-controlled ferroptosis and refine therapeutic strategies.
Several limitations of the present study should be
acknowledged. First, the in vitro experiments performed in
the present study relied on the VSC4.1 neuronal cell line, which
may not fully recapitulate the morphological and functional
complexity of primary neurons, potentially limiting translational
relevance. Second, the in vivo experiments were conducted
exclusively in female mice, which may introduce sex-specific bias
and limit generalizability across sexes. In addition, despite
efforts to standardize the injury procedure, inherent variability
in animal models of SCI may contribute to outcome heterogeneity.
Furthermore, in the present study, the experimental interventions
primarily assessed the necessity of Nrf2 signaling through
pharmacological inhibition and complementary activation strategies
using Nrf2 agonists were not explored. Finally, the present study
focused mainly on neuronal responses, without fully addressing
interactions with other spinal cord cell types, such as astrocytes
and microglia, which are known to influence ferroptosis and
secondary injury after SCI. Future studies incorporating
cell-specific omics approaches, pathway activation strategies and
multicellular interaction analyses will be important to further
refine the translational potential of targeting Nrf2-regulated
ferroptosis in SCI.
In summary, the present study demonstrated that DFO
mitigated neuronal ferroptosis and improved functional recovery
after SCI in part by activating the Nrf2/HO-1 signaling pathway and
enhancing the xCT/GPX4 antioxidant system. These results support
DFO as a potential therapeutic agent for SCI and highlight
Nrf2-dependent anti-ferroptotic mechanisms as promising targets for
future therapeutic strategies.
Supplementary Data
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YZ, LL and XMC conceived and designed the study,
supervised the project and acquired funding. ZQM, XWZ, XY and TL
established the SCI model, conducted the animal experiments and
drug interventions and performed the primary data acquisition. BQT
carried out the behavioral assessments (including BMS scoring) and
contributed to the acquisition of functional outcome data. ZQM and
XWZ performed the molecular assays (including protein and/or gene
expression analyses), acquired the associated data and participated
in data processing. TL, GHL and HYZ conducted the histological and
immunofluorescence analyses and performed the quantitative
image-based measurements. YZ, LL and XMC performed the statistical
analyses and assisted with data interpretation. XWZ and ZQM
contributed to experimental procedures, data collection and
prepared the figures. YZ, XY and BQT analyzed and interpreted the
data and drafted the initial manuscript. XMC, ZQM and XWZ
critically revised the manuscript for important intellectual
content. YZ and XMC confirm the authenticity of all the raw data.
All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
Animal experiments were conducted in accordance with
the laboratory animal management guidelines. All animal
experimental procedures were approved by the Animal Experiments and
Experimental Animal Welfare Committee of Capital Medical University
(approval no. AEEI-2023-029).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
ORCID: Xueming Chen, 0009-0001-8681-9660; Xinwei
Zhang, 0009-0003-3182-4758; Ziqian Ma, 0000-0003-1245-378X
Acknowledgments
Not applicable.
Funding
This work was supported by the Beijing Tongzhou District Science
and Technology Project (grant no. KJ2023SS013), the Youth
Scientific Research Incubation Program of Beijing Luhe Hospital,
Capital Medical University (grant no. LHYY-JC104) and the
Beijing-Tianjin-Hebei Basic Research Cooperation Project (grant no.
J230012).
References
|
1
|
Golestani A, Shobeiri P, Sadeghi-Naini M,
Jazayeri SB, Maroufi SF, Ghodsi Z, Dabbagh Ohadi MA, Mohammadi E,
Rahimi-Movaghar V and Ghodsi SM: Epidemiology of traumatic spinal
cord injury in developing countries from 2009 to 2020: A systematic
review and meta-analysis. Neuroepidemiology. 56:219–239. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tubbs RS, Blouir MC, Romeo AK, Mortazavi
MM and Cohen-Gadol AA: Spinal cord ischemia and atherosclerosis: A
review of the literature. Br J Neurosurg. 25:666–670. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Quadri SA, Farooqui M, Ikram A, Zafar A,
Khan MA, Suriya SS, Claus CF, Fiani B, Rahman M, Ramachandran A, et
al: Recent update on basic mechanisms of spinal cord injury.
Neurosurg Rev. 43:425–441. 2020. View Article : Google Scholar
|
|
4
|
Choo AM, Liu J, Lam CK, Dvorak M, Tetzlaff
W and Oxland TR: Contusion, dislocation, and distraction: Primary
hemorrhage and membrane permeability in distinct mechanisms of
spinal cord injury. J Neurosurg Spine. 6:255–266. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ahuja CS, Wilson JR, Nori S, Kotter MRN,
Druschel C, Curt A and Fehlings MG: Traumatic spinal cord injury.
Nat Rev Dis Primers. 3:170182017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li S, Gao S, Hu Y, Sun G, Feng J and Sheng
W: Ferroptosis in spinal cord injury: Research progress and novel
insights. J Cell Biochem. 126:e700672025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yu T, Yang LL, Zhou Y, Wu MF and Jiao JH:
Exosome-mediated repair of spinal cord injury: A promising
therapeutic strategy. Stem Cell Res Ther. 15:62024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhiguo F, Ji W, Shenyuan C, Guoyou Z, Chen
K, Hui Q, Wenrong X and Zhai X: A swift expanding trend of
extracellular vesicles in spinal cord injury research: A
bibliometric analysis. J Nanobiotechnology. 21:2892023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shi Z, Yuan S, Shi L, Li J, Ning G, Kong X
and Feng S: Programmed cell death in spinal cord injury
pathogenesis and therapy. Cell Prolif. 54:e129922021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta
R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS,
et al: Ferroptosis: An iron-dependent form of nonapoptotic cell
death. Cell. 149:1060–1072. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li FJ, Long HZ, Zhou ZW, Luo HY, Xu SG and
Gao LC: System Xc-/GSH/GPX4 axis: An
important antioxidant system for the ferroptosis in drug-resistant
solid tumor therapy. Front Pharmacol. 13:9102922022. View Article : Google Scholar
|
|
12
|
Yao X, Zhang Y, Hao J, Duan HQ, Zhao CX,
Sun C, Li B, Fan BY, Wang X, Li WX, et al: Deferoxamine promotes
recovery of traumatic spinal cord injury by inhibiting ferroptosis.
Neural Regen Res. 14:532–541. 2019. View Article : Google Scholar :
|
|
13
|
Zhang Y, Sun C, Zhao C, Hao J, Zhang Y,
Fan B, Li B, Duan H, Liu C, Kong X, et al: Ferroptosis inhibitor
SRS 16-86 attenuates ferroptosis and promotes functional recovery
in contusion spinal cord injury. Brain Res. 1706:48–57. 2019.
View Article : Google Scholar
|
|
14
|
Bai XY, Liu XL, Deng ZZ, Wei DM, Zhang D,
Xi HL, Wang QY, He MZ and Yang YL: Ferroptosis is a new therapeutic
target for spinal cord injury. Front Neurosci. 17:11361432023.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang C, Zhu Y, Zhu X, Chen R, Zhang X and
Lian N: USP7 regulates HMOX-1 via deubiquitination to suppress
ferroptosis and ameliorate spinal cord injury in rats. Neurochem
Int. 168:1055542023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zou Z, Liu R, Wang Y, Tan H, An G, Zhang
B, Wang Y and Dong D: Protein arginine methyltransferase 8
regulates ferroptosis and macrophage polarization in spinal cord
injury via glial cell-derived neurotrophic factor. CNS Neurosci
Ther. 29:2145–2161. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang Y, Fan BY, Pang YL, Shen WY, Wang X,
Zhao CX, Li WX, Liu C, Kong XH, Ning GZ, et al: Neuroprotective
effect of deferoxamine on erastininduced ferroptosis in primary
cortical neurons. Neural Regen Res. 15:1539–1545. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ge MH, Tian H, Mao L, Li DY, Lin JQ, Hu
HS, Huang SC, Zhang CJ and Mei XF: Zinc attenuates ferroptosis and
promotes functional recovery in contusion spinal cord injury by
activating Nrf2/GPX4 defense pathway. CNS Neurosci Ther.
27:1023–1040. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gong F, Ge T, Liu J, Xiao J, Wu X, Wang H,
Zhu Y, Xia D and Hu B: Trehalose inhibits ferroptosis via NRF2/HO-1
pathway and promotes functional recovery in mice with spinal cord
injury. Aging (Albany NY). 14:3216–3232. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ni C, Ye Q, Mi X, Jiao D, Zhang S, Cheng
R, Fang Z, Fang M and Ye X: Resveratrol inhibits ferroptosis via
activating NRF2/GPX4 pathway in mice with spinal cord injury.
Microsc Res Tech. 86:1378–1390. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li Z, Cheng P, Xi H, Jiang T, Zheng X, Qiu
J, Gong Y, Wu X, Mi S, Hong Y, et al: Tomatidine alleviates
intervertebral disc degeneration by activating the Nrf2/HO-1/GPX4
signaling pathway. Drug Des Devel Ther. 18:6313–6329. 2024.
View Article : Google Scholar :
|
|
22
|
Liu D, Yang S and Yu S: Interactions
between ferroptosis and oxidative stress in ischemic stroke.
Antioxidants (Basel). 13:13292024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Percie du Sert N, Hurst V, Ahluwalia A,
Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl
U, et al: The ARRIVE guidelines 2.0: Updated guidelines for
reporting animal research. BMJ Open Sci. 4:e1001152020.
|
|
24
|
Zhang X, Liu T, Ma Z, Li G, Ding N, Wang
Z, Guan Y, Zhang Y, Liu L and Chen X: VEGF secreted by human dental
pulp stem cell promotes spinal cord injury repair by inhibiting
microglial pyroptosis through the PI3K/AKT pathway. J Transl Med.
23:4372025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xian P, Hei Y, Wang R, Wang T, Yang J, Li
J, Di Z, Liu Z, Baskys A, Liu W, et al: Mesenchymal stem
cell-derived exosomes as a nanotherapeutic agent for amelioration
of inflammation-induced astrocyte alterations in mice.
Theranostics. 9:5956–5975. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Basso DM, Fisher LC, Anderson AJ, Jakeman
LB, McTigue DM and Popovich PG: Basso mouse scale for locomotion
detects differences in recovery after spinal cord injury in five
common mouse strains. J Neurotrauma. 23:635–659. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu T, Ma Z, Liu L, Pei Y, Wu Q, Xu S, Liu
Y, Ding N, Guan Y, Zhang Y and Chen X: Conditioned medium from
human dental pulp stem cells treats spinal cord injury by
inhibiting microglial pyroptosis. Neural Regen Res. 19:1105–1111.
2024. View Article : Google Scholar
|
|
28
|
Kim HJ, Im W, Kim S, Kim SH, Sung JJ, Kim
M and Lee KW: Calcium-influx increases SOD1 aggregates via nitric
oxide in cultured motor neurons. Exp Mol Med. 39:574–582. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Choi JH, Jang M, Lee JI, Chung WS and Cho
IH: Neuroprotective effects of a traditional multi-herbal medicine
kyung-ok-ko in an animal model of Parkinson's disease: Inhibition
of MAPKs and NF-κB pathways and activation of keap1-Nrf2 pathway.
Front Pharmacol. 9:14442018. View Article : Google Scholar
|
|
30
|
Milich LM, Choi JS, Ryan C, Cerqueira SR,
Benavides S, Yahn SL, Tsoulfas P and Lee JK: Single-cell analysis
of the cellular heterogeneity and interactions in the injured mouse
spinal cord. J Exp Med. 218:e202100402021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li D, Lu X, Xu G, Liu S, Gong Z, Lu F, Xia
X, Jiang J, Wang H, Zou F and Ma X: Dihydroorotate dehydrogenase
regulates ferroptosis in neurons after spinal cord injury via the
P53-ALOX15 signaling pathway. CNS Neurosci Ther. 29:1923–1939.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zavvarian MM, Hong J and Fehlings MG: The
functional role of spinal interneurons following traumatic spinal
cord injury. Front Cell Neurosci. 14:1272020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jin W, Botchway BOA and Liu X: Curcumin
can activate the Nrf2/HO-1 signaling pathway and scavenge free
radicals in spinal cord injury treatment. Neurorehabil Neural
Repair. 35:576–584. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Xuan LN, Hu ZX, Jiang ZF, Zhang C, Sun XW,
Ming WH, Liu HT, Qiao RF, Shen LJ, Liu SB, et al: Pregnane X
receptor (PXR) deficiency protects against spinal cord injury by
activating NRF2/HO-1 pathway. CNS Neurosci Ther. 29:3460–3478.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Song X and Long D: Nrf2 and ferroptosis: A
new research direction for neurodegenerative diseases. Front
Neurosci. 14:2672020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Luo Y, He YZ, Wang YF, Xu YX and Yang L:
Adipose-derived mesenchymal stem cell exosomes ameliorate spinal
cord injury in rats by activating the Nrf2/HO-1 pathway and
regulating microglial polarization. Folia Neuropathol. 61:326–335.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhang X, Xu L, Chen X, Zhou X and Cao L:
Acacetin alleviates neuroinflammation and oxidative stress injury
via the Nrf2/HO-1 pathway in a mouse model of spinal cord injury.
Transl Neurosci. 13:483–494. 2022. View Article : Google Scholar :
|
|
38
|
Park MW, Cha HW, Kim J, Kim JH, Yang H,
Yoon S, Boonpraman N, Yi SS, Yoo ID and Moon JS: NOX4 promotes
ferroptosis of astrocytes by oxidative stress-induced lipid
peroxidation via the impairment of mitochondrial metabolism in
Alzheimer's diseases. Redox Biol. 41:1019472021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shen D, Wu W, Liu J, Lan T, Xiao Z, Gai K,
Hu L, Luo Z, Wei C, Wang X, et al: Ferroptosis in oligodendrocyte
progenitor cells mediates white matter injury after hemorrhagic
stroke. Cell Death Dis. 13:2592022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xu T, Zhu Q, Huang Q, Gu Q, Zhu Y, Tang M,
Tian S, Wang L, Yan F, Ge J, et al: FGF21 prevents neuronal cell
ferroptosis after spinal cord injury by activating the
FGFR1/β-Klotho pathway. Brain Res Bull. 202:1107532023. View Article : Google Scholar
|
|
41
|
Xiao S, Zhang Y, Wang S, Liu J, Dan F,
Yang F, Hong S, Liu N, Zeng Y, Huang K, et al: The Syvn1 inhibits
neuronal cell ferroptosis by activating Stat3/Gpx4 axis in rat with
spinal cord injury. Cell Prolif. 57:e136582024. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Alexianu ME, Mohamed AH, Smith RG, Colom
LV and Appel SH: Apoptotic cell death of a hybrid motoneuron cell
line induced by immunoglobulins from patients with amyotrophic
lateral sclerosis. J Neurochem. 63:2365–2368. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Song Q, Cui Q, Sun S, Wang Y, Yuan Y and
Zhang L: Crosstalk between cell death and spinal cord injury:
Neurology and therapy. Mol Neurobiol. 61:10271–10287. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li L, Cao Y, Zhang X, Guo J, Lin Z, Zhou
P, Chen C, Chen J, Liu Y, Luo D, et al: Injectable ROS homeostasis
protective hydrogel inhibiting microglial ferroptosis through the
Nrf2/Slc7a11/Gpx4 to alleviate neuropathic pain and promote spinal
cord injury repair. Redox Biol. 86:1038162025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Liang X, Qin R, Qin Q, Xu W, Xu H, Lai X,
Shao L, Li C, Xie M, Xiong X, et al: Therapeutic potential of
astrocyte transdifferentiated neurons. Neural Regen Res. Dec
30–2025.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Li F and Wang H, Chen H, Guo J, Dang X, Ru
Y and Wang H: Mechanism of ferroptosis and its role in spinal cord
injury. Front Neurol. 13:9267802022. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ge H, Xue X, Xian J, Yuan L, Wang L, Zou
Y, Zhong J, Jiang Z, Shi J, Chen T, et al: Ferrostatin-1 Alleviates
white matter injury via decreasing ferroptosis following spinal
cord injury. Mol Neurobiol. 59:161–176. 2022. View Article : Google Scholar
|
|
48
|
Li Y, Cao Y, Xiao J, Shang J, Tan Q, Ping
F, Huang W, Wu F, Zhang H and Zhang X: Inhibitor of
apoptosis-stimulating protein of p53 inhibits ferroptosis and
alleviates intestinal ischemia/reperfusion-induced acute lung
injury. Cell Death Differ. 27:2635–2650. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhao W, Gasterich N, Clarner T, Voelz C,
Behrens V, Beyer C, Fragoulis A and Zendedel A: Astrocytic Nrf2
expression protects spinal cord from oxidative stress following
spinal cord injury in a male mouse model. J Neuroinflammation.
19:1342022. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Paterniti I, Mazzon E, Emanuela E, Paola
RD, Galuppo M, Bramanti P and Cuzzocrea S: Modulation of
inflammatory response after spinal cord trauma with deferoxamine,
an iron chelator. Free Radic Res. 44:694–709. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yi Y, Jia P, Xie P, Peng X, Zhu X, Yin S,
Luo Y, Deng Y and Wan L: Beyond oxidative stress: Ferroptosis as a
novel orchestrator in neurodegenerative disorders. Front Immunol.
16:16838762025. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Cheng S, Li L, Xu M, Ma N and Zheng Y:
Exploring hypoxia-related genes in spinal cord injury: A pathway to
new therapeutic targets. Front Mol Neurosci. 18:15654302025.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Liu J, Cao J, Yu X, Chang J, Sui T and Cao
X: Necroptosis pathway emerged as potential diagnosis markers in
spinal cord injury. J Cell Mol Med. 28:e182192024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Lu F, Liu Y, Chen Z, Chen S, Liang W, Hua
F, Zhong M and Wang L: Identification and validation of
glucocorticoid receptor and programmed cell death-related genes in
spinal cord injury using machine learning. Sci Rep. 15:242022025.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Xiang-Xia, Li KX, Li QW, Wu ZM, Guo RC and
Shen CL: Identifying pyroptosis- and inflammation-related genes in
spinal cord injury based on bioinformatics analysis. Sci Rep.
15:254242025. View Article : Google Scholar : PubMed/NCBI
|