Introduction
Herpes simplex virus type 1 (HSV-1) is a neurotropic
human pathogen that has infected over 67% of the global population
(1,2). When it infects the central nervous
system (CNS), it can lead to Herpes simplex encephalitis (HSE),
posing significant risks to the health and survival of neonates and
immunocompromised individuals (3). Furthermore, HSV-1 infection has
been associated with an elevated risk of HIV-1 infection and
transmission (4,5) and is closely linked to the
pathogenesis and progression of neurodegenerative disorders,
including Alzheimer's disease (6-8).
Notably, there is currently no clinically approved vaccine
available to prevent HSV-1 infection (9). Presently, nucleoside analogs such
as acyclovir (ACV) and valacyclovir are primarily employed to
mitigate the symptoms associated with HSV-1 infection and to
suppress viral reactivation. However, the prolonged use of these
antiviral agents has led to the emergence of drug-resistant strains
(10), which pose a significant
threat to the health of immunocompromised patients (11,12). Consequently, the development of
novel therapeutic agents effective against HSV-1 infection is of
paramount importance.
Viruses, as parasitic entities, typically depend on
the host's cellular synthesis and metabolic pathways to complete
their lifecycle (13). Among
these pathways, heat shock protein 90 (HSP90), a highly conserved
molecular chaperone, emerges as a critical host factor essential
for the lifecycle of numerous viruses (14). This includes processes such as
viral entry, transport, assembly, and the release of viral
particles, all of which necessitate host molecular chaperones. The
RAF/MEK/ERK signaling pathway, a component of the classical MAPK
cascade, plays a pivotal role in cell proliferation and development
(15). RAF family proteins,
which serve as client proteins of HSP90, are exploited by viruses
such as the influenza virus, SARS-CoV-2 and HSV-1 to co-opt the
host's Raf/MEK/ERK pathway, thereby enhancing their replication and
proliferation (13,16). The interaction between host and
pathogen results in substantial reorganization of the host protein
network, leading to dysregulation of signaling pathways and the
establishment of a pathological environment (17). Consequently, the RAF/MEK/ERK
pathway represents a promising target for antiviral intervention
(18). A comprehensive
understanding of the intricate dynamics of host-pathogen
interactions is imperative for the development of strategies aimed
at inhibiting viral replication. In contrast to antiviral agents
targeting viral components, those designed to interact with host
factors demonstrate a reduced susceptibility to the emergence of
drug-resistant viral strains (19,20). While HSP90 inhibitors have shown
broad-spectrum antiviral efficacy, their approval by the FDA
remains pending due to concerns regarding toxicity and limited
efficacy (21). Consequently,
advancing the development of safe and effective HSP90 inhibitors
represents a critical focus for current antiviral drug research and
development.
The present study assessed JD-02's potential as an
anti-HSV-1 treatment. Among six benzamide-based compounds, JD-02
showed anti-HSV-1 activity in Vero cell assays. As a novel HSP90
inhibitor, it blocks HSV-1 infection by targeting the Raf/MEK/ERK
pathway. In vivo mouse model studies confirmed JD-02's
effectiveness in reducing HSV-1-related disease symptoms.
Materials and methods
Compounds, antibodies and reagents
Six benzamide derivatives were synthesized by
Professor Daohua Xu from the School of Pharmacy, Guangdong Medical
University (22). The compounds
were purified by silica gel column, and the purity was tested by
High Performance Liquid Chromatography analysis [Agilent 1260
Infinity II HPLC system (Agilent Technologies, Inc.) with a Zorbax
Eclipse Plus C18 column (4.6×150 mm, 5 μm; Agilent
Technologies, Inc.). Conditions included a 30°C column temperature,
20 μl injection volume, mobile phase consisting of solvent A
(water/0.1% formic acid) and solvent B (acetonitrile/0.1% formic
acid) at 1.0 ml/min flow rate]. All compounds were dissolved in
dimethyl sulfoxide at a concentration of 50 mM.
Antibodies against ICP0 (cat. no. ab6513), gD
(ab6507), ICP27 (cat. no. ab53480) and Anti-HSV-1 (cat. no. ab9533)
were obtained from Abcam. Antibodies against GAPDH (cat. no.
GTX100118) were purchased from GeneTex, Inc. Antibodies against
phospho-ERK1/2 (cat. no. 4370S), ERK1/2 (cat. no. 4695S),
phospho-MEK1/2 (cat. no. 9154T), phospho-B-Raf (cat. no. 2696T), HA
(cat. no. 3724S) and Flag (cat. no. 14793S) were obtained from Cell
Signaling Technology, Inc. Antibodies against VP5 (cat. no.
sc-13525), gB (cat. no. sc-56987) and Raf-B (C-19) (cat. no.
sc-166) were obtained from Santa Cruz Biotechnology, Inc.
Antibodies against MEK1/2 (cat. no. AF6385) were obtained from
Affinity Biosciences.
ACV was purchased from MilliporeSigma, which was
dissolved in dimethyl sulfoxide (DMSO) with a concentration of 20
mM. The MEK1/2-inhibitor U0126 was purchased from Cell Signaling
Technology, Inc. and was dissolved in DMSO with a concentration of
10 mM.
Cell lines and virus
SH-SY5Y [cat. no. CRL226; American Type Culture
Collection (ATCC)] and HaCaT (cat. no. 300493; CLS Cell Lines
Service GmbH) and were maintained in Dulbecco's modified Eagle's
medium (DMEM; Gibco; Thermo Fisher Scientific, Inc.) with 10% fetal
bovine serum (FBS; Thermo Fisher Scientific, Inc.). Vero cells
(cat. no. CCL81; ATCC) were cultured in DMEM with 10% FBS (Hangzhou
Biology Engineering Materials Co., Ltd.). All the cells were
cultured at 37°C in a humid atmosphere with 5% CO2.
HSV-1 strain F was acquired from the Hong Kong
University and propagated in Vero cells. The reporter virus
EGFP-HSV-1, which expresses EGFP-tagged viral protein Us11, was
acquired from the Anti-Stress and Health Research Center, College
of Pharmacy, Jinan University. ACV resistant HSV-1 strains,
HSV-1/Blue and HSV-1/153, were kind gifts from Prof Tao Peng
(Guangzhou Institutes of Biomedicine and Health, Chinese Academy of
Sciences) and prepared as previously methods (23).
Cytotoxicity assay
The cytotoxic effects of JD-02 were assessed
utilizing the Cell Counting Kit (CCK-8) assay kit (cat. no. 96992;
MilliporeSigma). Initially, Vero, HaCaT, SH-SY5Y and BV2 were
seeded in triplicate into 96-well plates at a density of 10,000
cells per well and incubated for 24 h. Following this incubation
period, the cells were exposed to varying concentrations of JD-02
(0.125, 0.25, 0.5, 1, 2, 4, 8 and 16 μM). Following a 24-h
incubation at 37°C, 10 μl of CCK-8 reagent was added to each
well, and the optical density at 490 nm (OD490) was measured using
a microplate reader (24). The
experiment utilized non-cytotoxic compound concentrations.
Viral plaque assay
Viral plaque assay was performed in Vero cells
(25). Briefly, once the Vero
cells formed a confluent monolayer, they were infected with HSV-1
[multiplicity of infection (MOI)=1] and treated with drugs or ACV
at 37°C to facilitate optimal viral particle absorption. Following
this, the medium was replaced with a maintenance medium containing
1% methylcellulose (cat. no. M0512; MilliporeSigma) with or without
JD-02 and ACV in each well. The cells were incubated for 72 h
before the cells were fixed with 4% polyoxymethylene for 20 min at
room temperature and stained with 1% crystal violet (cat. no.
C0121; Beyotime Institute of Biotechnology) for 30 min at room
temperature. The total number of plaques was counted, and the
inhibition rate was determined.
Virus titration assay
Virus titers were quantified by assessing the
cytopathic effects (CPEs) induced by HSV-1. Viral titration was
conducted by assessing the CPEs in Vero cells infected with HSV-1.
The cells were cultured in 96-well plates at a density of
1.5×105 cells per well and were exposed to a culture
medium containing serial 10-fold dilutions of HSV-1 viral particles
(26). Following a 72-h
incubation period, the CPEs were evaluated to determine the 50%
tissue culture infectious dose (TCID50), which was
subsequently converted to plaque-forming units (PFU) per ml using
the formula: PFU/ml=TCID50/ml × 0.7. CPE grade: '-' for
no CPE; '+' for 1-25% CPE; '++' for 26-50% CPE; '++' for 51-75%
cytopathic as '++++'; 76-100% cytopathic as '++++'.
Viral inactivation, attachment,
penetration assay
For virus inactivation analysis, HSV-1 was incubated
with JD-02 (0.5 μM) at 37°C for 2 h, then diluted and used
to infect Vero cells at 37°C for another 2 h. After replacing the
solution, incubation continued for 72 h (27). Empty spot reduction assays were
conducted. For the virus-attached vacuole assay, HaCaT cells were
chilled at 4°C for 1 h, exposed to HSV-1 (30 PFUs/well) with or
without JD-02 (0.5 μM) at 4°C for 2 h, then washed with cold
PBS and incubated for 48 h with a complete solution for empty
plaque experiments. For the viral DNA copy number assay, HSV-1
(MOI=5) and JD-02 (0.5 μM) were applied to HaCaT cells at
4°C for 2 h, washed with cold PBS, subjected to three freeze-thaw
cycles. For the virus-penetration empty plaque assay, HaCaT cells
were infected with HSV-1 at 4°C for 2 h, treated with JD-02 at 37°C
for 10 min, then exposed to acidic and alkaline PBS for 1 min each,
and incubated with an overlay solution for 72 h. For the viral DNA
copy number assay, HaCaT cells were treated with HSV-1 and JD-02 at
4°C for 2 h, followed by JD-02 at 37°C for 10 min, and exposure to
acidic and alkaline PBS for 1 min each (28).
Immunofluorescence assay
The cells to be treated were fixed with 4%
paraformaldehyde (PFA), penetrated with 0.1% NP-40 for 4 min, and
then blocked with 5% bovine serum albumin (BSA) for 90 min before
staining with VP5 (1:100; cat. no. sc-13525; Santa Cruz
Biotechnology, Inc.) overnight at 4°C. The cells were then
incubated with Alexa Fluor-conjugated secondary antibody (1:1,000;
cat. no. A32723; Thermo Fisher Scientific, Inc.) at room
temperature. Nuclei were next labeled with DAPI (cat. no. C1006;
Beyotime Institute of Biotechnology) for 15 min at room
temperature. It should be noted that each step of these processes
was washed with PBS for 3 min. Finally, fluorescence images were
acquired using a Zeiss LSM510 Meta confocal system (Zeiss
GmbH).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from the indicated treated
cultured cells using TRIzol Reagent (TRIzol; cat. no. DP424;
Tiangen Biotech Co., Ltd.), and a PrimeScript RT kit (cat. no.
RR036A-1; Takara Bio, Inc.) was used for cDNA synthesis according
to the manufacturer's instructions. The RT-qPCR assay was performed
in a CFX96 Touch-Real-Time PCR detection system (Bio-Rad
Laboratories, Inc.) using the TB Green Premix Ex Taq II kit (cat.
no. RR820; Takara Bio, Inc.) according to the manufacturer's
instructions (28). Gene
expression levels were normalized to the internal regulatory gene
GAPDH. To determine the viral genomic DNA, HaCaT cells were
infected with HSV-1 (MOI=0.1) while incubated with the addition of
JD-02. After 24 h, samples were placed in −80°C refrigerator and
repeatedly frozen and thawed three times, after which the
supernatant and cell pellet were collected. The viral genomic DNA
was obtained using EasyPure® Viral DNA/RNA Kit (TransGen
Biotech Co., Ltd.), which were subjected to RT-qPCR for
quantification. The thermal cycling conditions were set as follows:
Initial denaturation at 95°C for 30 sec; 40 cycles of 95°C for 5
sec and 60°C for 30 sec. The 2-ΔΔCq method was applied
for mRNA analysis and GAPDH was used as the reference gene
(29). All primer sequences are
shown in Table SI.
Transfection of plasmids or small
interfering RNA (siRNA)
The HA-UL42 plasmids and Flag-UL30 plasmids were
synthesized and constructed by Shanghai GenePharma Co., Ltd. All
siRNAs were synthesized and constructed by Integrated Biotech
Solutions. Cells were transfected with plasmid (2 μg) or
siRNA (100 nM) using jetPRIME®transfection reagent (cat. no.
PT114-15; Polyplus-transfection). After 24 or 48 h, the cells were
infected with HSV-1 with or without chemicals. All primer sequences
of siRNA are displayed in Table
SII.
Western blot assay
Samples were extracted from HSV-1-infected cells
using RIPA buffer (cat. no. P0013B; Beyotime Institute of
Biotechnology) supplemented with 1% PMSF (cat. no. ST506; Beyotime
Institute of Biotechnology). The lysates were clarified by
centrifugation at 12,000 × g for 10 min at 4°C. Protein
concentrations were normalized using the bicinchoninic acid (BCA)
protein assay kit (Beyotime Institute of Biotechnology). Proteins
(30 μg total per lane) were separated on an 8 to 12%
gradient SDS-PAGE gel and transferred onto polyvinylidene fluoride
membranes (MilliporeSigma). Membranes were blocked with 5% BSA for
1 h at room temperature and incubated with primary antibodies at
4°C overnight, followed by incubation with secondary antibodies
(1:5,000; cat. nos. 31430 and 31460; Invitrogen; Thermo Fisher
Scientific, Inc.,) for 60 to 90 min at room temperature. These
primary antibodies included ICP0 (1:1,000; cat. no. ab6513; Abcam),
ICP27 (1:1,000; cat. no. ab53480; Abcam), gD (1:1,000; cat. no.
ab6507; Abcam), gB (1:500; cat. no. sc-56987; Santa Cruz
Biotechnology, Inc.), Raf-B (1:500; cat. no. sc-166; Santa Cruz
Biotechnology, Inc.), p-BRAF (1:1,000; cat. no. 2696T; Cell
Signaling Technology, Inc.), ERK (1:1,000; cat. no. 4695S; Cell
Signaling Technology, Inc.), p-ERK (1:1,000; cat. no. 4370S; Cell
Signaling Technology, Inc.), MEK1/2 (1:1,000; cat. no. AF6385;
Affinity Biosciences), p-MEK1/2 (1:1,000; cat. no. 9154T; Cell
Signaling Technology, Inc.), Flag (1:1,000; cat. no. 14793S; Cell
Signaling Technology, Inc.), HA (1:1,000; cat. no. 3724S; Cell
Signaling Technology, Inc.). Target proteins were visualized using
enhanced chemiluminescence (ECL) solution (cat. no. 36208ES60;
Shanghai Yeasen Biotechnology Co., Ltd.) and images were captured
with a Tanon 5200 Image Analysis System (Tanon Science and
Technology Co., Ltd.). ImageJ software (version 1.54f; National
Institutes of Health) was used for densitometric analysis.
HSE mice model
All animal experiments were approved (approval no.
20200402-07) by the Animal Care and Use Committee of Jinan
University (Guangzhou, China). A total 50 male BALB/c mice, aged 5
weeks and weighing between 20-22 g, were procured from the
Guangdong Medical Laboratory Animal Center. The mice were
maintained under a controlled environmental conditions, with a
standardize temperature range of 20-26°C. They were housed to a
regulated 12/12-h reverse light/dark cycle and were provided with
ad libitum access to food and water. Following an
acclimatization period, the mice were randomly allocated into
different experimental groups and subsequently inoculated
intranasally with HSV-1 at a concentration of 2×106 PFU
per mouse. Infected mice were injected intraperitoneally with JD-02
(10 or 20 mg/kg/day) or ACV (20 mg/kg/day) for 5 consecutive days.
Correspondingly, mice in the mock and HSV-1 control groups were
treated with saline (0.9%). Body weight and HSE symptoms were
recorded daily for all mice. The scoring rules for HSE symptoms
were based on previous studies: Eye swelling/lesions (0: no
symptoms, 1: mild eyelid swelling, 2: moderate eyelid swelling with
crusting > 50% 3: severe eyelid swelling with crusting), hair
loss (0: none hair loss, 1: minimal periocular hair loss, 2:
moderate periocular hair loss, 3: severe hair loss limited to the
periocular region); hydrocephalus (0: none, 1: minor bump, 2:
moderate bump, 3: large bump). Mice were sacrificed for
histological analysis and viral quantification as soon as they lost
~20% of their body weight (the 6th day after infection). The mice
were euthanized using isoflurane (5% induction), followed by
cervical dislocation. Death was confirmed by the absence of
respiration and heartbeat. Whole brains were primarily dissected
and promptly stored at −80°C for subsequent viral titer and gene
expression analysis. Parts of the heart, liver, spleen, lung,
kidney and intestine were fixed in 4% PFA solution, at room
temperature for 24 h, embedded in paraffin, sectioned, stained with
hematoxylin-eosin (H&E) solution or anti-HSV-1 antibody, and
examined under the light microscope. In addition, total RNA was
extracted from brain tissues, and the samples were subjected to
RT-qPCR.
Histology assay
The primary organs from mice infected with HSV-1 and
subsequently treated with JD-02 at six days post-infection (n=4)
were preserved in 4% PFA, embedded in paraffin, deparaffinized,
stained with H&E (cat. no. G1120; Beijing Solarbio Science
& Technology Co., Ltd.) The specimen was stained with
hematoxylin solution for 15 min and eosin for 3 min, both at room
temperature and subsequently examined using light microscopy.
Statistical analysis
Data are presented as the mean ± SD of the results
from at least 2 independent experiments. The unpaired Student's
t-test analysis was executed to compare the means of two groups.
*P<0.05 was considered to indicate a statistically
significant difference. Statistical analysis was carried out using
GraphPad Prism 8.0 software (Dotmatics).
Results
Anti-HSV-1 activity and cytotoxicity of
JD-02
The structures of the six novel benzamide
derivatives are detailed in Table
I. Initially, the antiviral efficacy of these derivatives
against HSV-1 was assessed in Vero cells using a CPE. Observations
via light microscopy revealed that JD-02 significantly inhibited
HSV-1-induced CPE in Vero cells more effectively than the other
compounds tested (Table II).
Consequently, JD-02 was selected for further experimentation.
 | Table IStructures of 6 novel benzamide
derivatives. |
Table I
Structures of 6 novel benzamide
derivatives.
 | Table IIAntiviral screening based on the CPE
assay. |
Table II
Antiviral screening based on the CPE
assay.
| Compound | 1 μM | 2 μM | 4 μM |
|---|
| JD-01 | ++++ | +++ | +++ |
| JD-02 | + | + | + |
| JD-03 | ++++ | ++++ | ++++ |
| JD-04 | +++ | ++ | ++ |
| JD-05 | ++++ | +++ | +++ |
| JD-06 | ++++ | +++ | +++ |
| Cell | - | - | - |
| HSV-1 | ++++ | ++++ | ++++ |
The cytotoxicity of JD-02 was compared with that of
the commercial benzamide Hsp90 inhibitor AT533 across various cell
lines, including Vero, HaCaT, SH-SY5Y and BV2, utilizing the CCK-8
assay (Fig. S1). The findings
indicated that JD-02 as Hsp90 inhibitor exhibited relatively low
toxicity in Vero, HaCaT and BV2 cells compared with AT533. The 50%
cytotoxic concentration (CC50) values for JD-02 exceeded
32 μM in Vero cells, 5.503 μM in HaCaT cells, 10.75
μM in BV2 cells and 3.374 μM in SH-SY5Y cells.
Therefore, it was chosen to use a concentration below its
CC50 for subsequent studies.
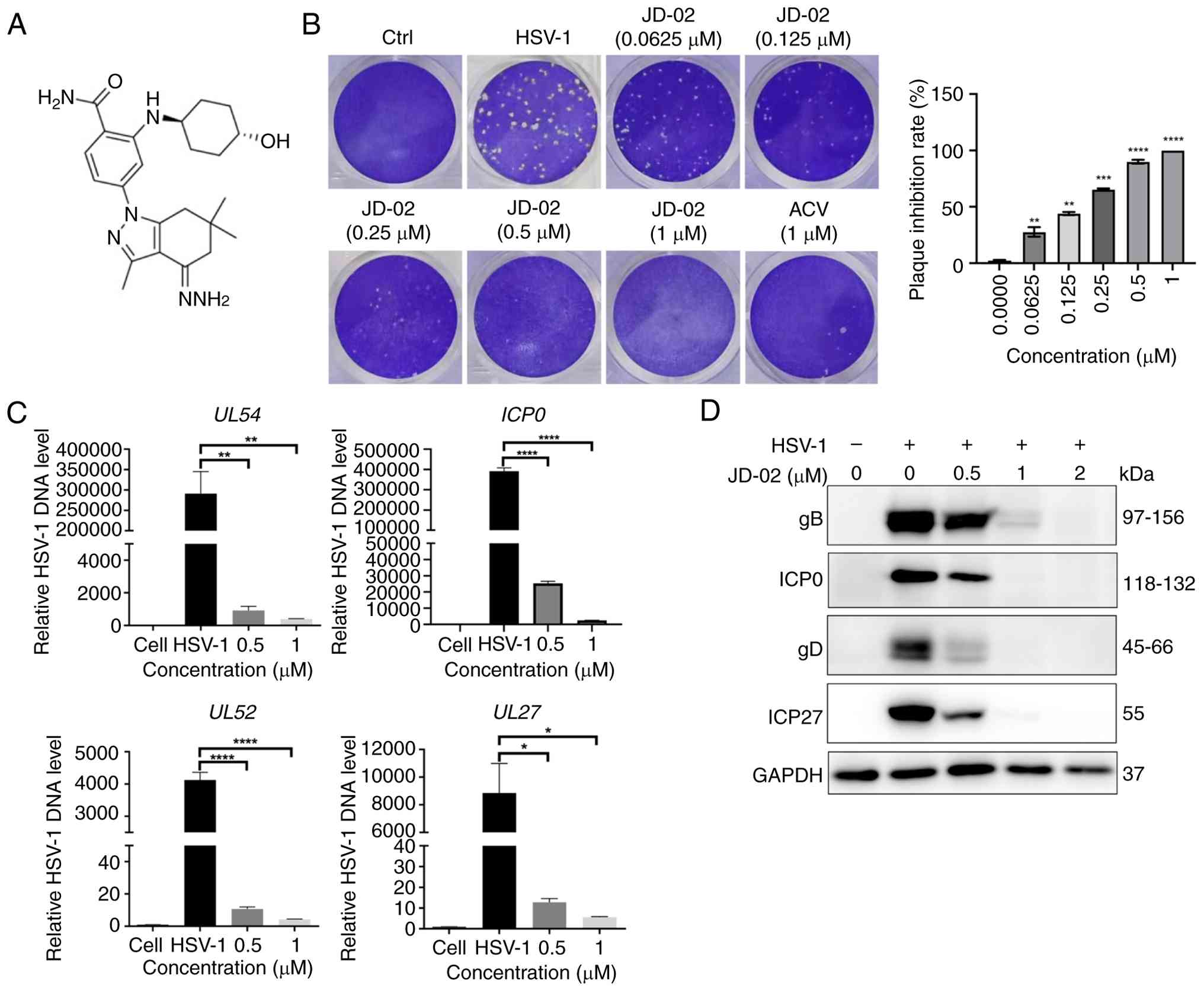
JD-02 suppresses HSV-1 replication
Among the compounds evaluated, JD-02 (Fig. 1A) demonstrated significant
inhibitory activity against HSV-1 infection across all tested
concentrations. The anti-HSV-1 efficacy of JD-02 was assessed using
viral plaque assays, which revealed a dose-dependent decrease in
the number of viral plaques formed in Vero cells infected with
HSV-1 and the half-maximal effective concentration was determined
to be 0.1396 μM (Fig.
1B). JD-02 exhibited inhibition rates against HSV-1 infection
that were comparable to those of ACV at equivalent concentrations.
Additionally, RT-qPCR analysis indicated that JD-02 significantly
reduced the DNA copy number of several viral genes, including UL54,
ICP0, UL52 and UL27, at concentrations of 0.5 and 1 μM
(Fig. 1C). Besides, western blot
analysis further confirmed a dose-dependent inhibitory effect of
JD-02 on the expression of the viral immediate-early protein ICP0,
early proteins ICP27 and gD, as well as the late protein gB
(Fig. 1D). Collectively, these
findings suggest that JD-02 possesses a potent capacity to inhibit
HSV-1 infection.
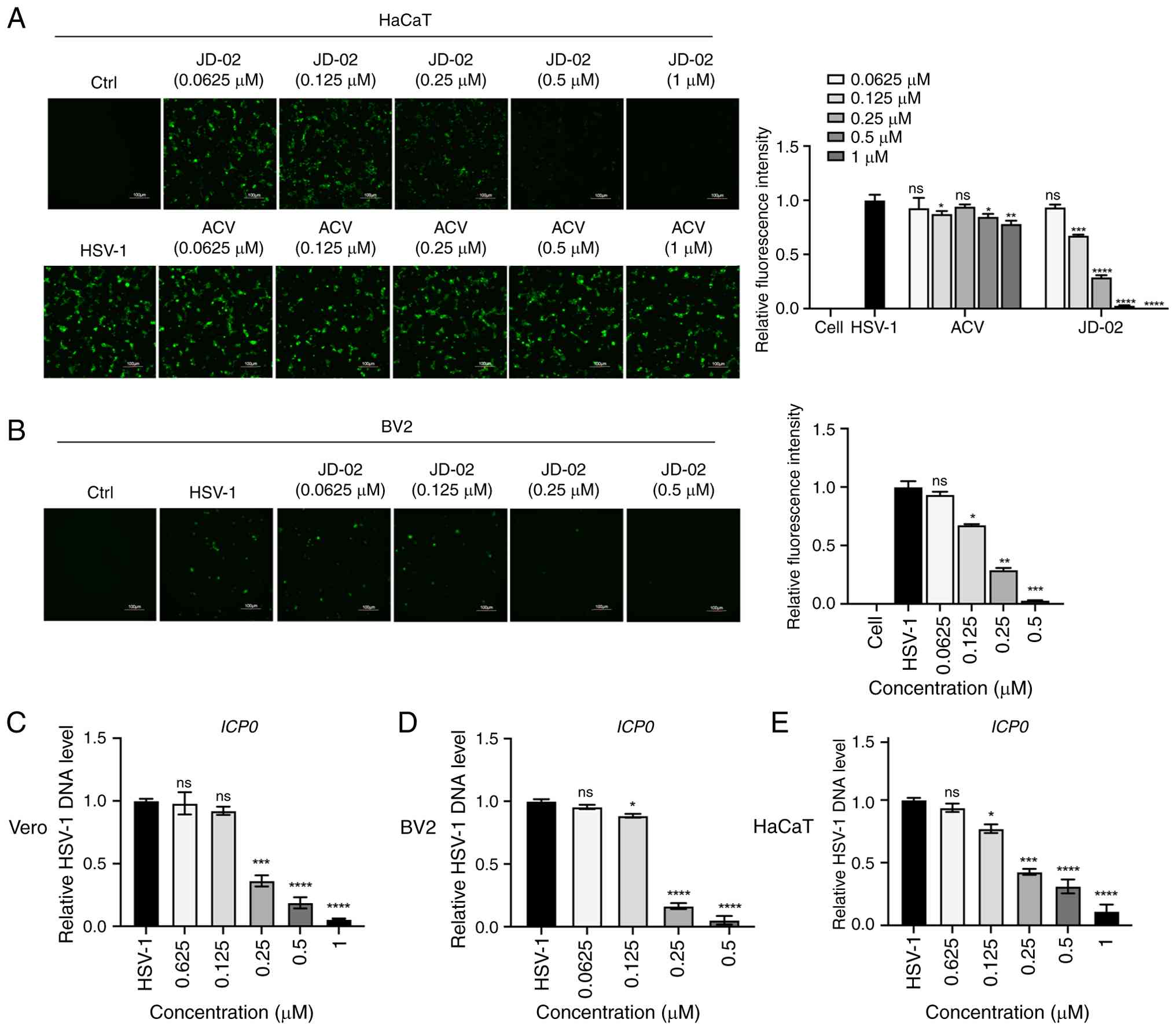
To further investigate the antiviral efficacy of
JD-02, an EGFP-tagged HSV-1 was employed. Fluorescence analysis
revealed a dose-dependent reduction in fluorescence intensity in
Vero cells treated with JD-02. Moreover, JD-02 demonstrated greater
effectiveness in inhibiting fluorescence intensity compared with
ACV (Fig. 2A). Considering that
HSV-1 is a neurotropic virus capable of accessing the CNS via
epithelial cells and potentially inducing severe HSE (3), and acknowledging that microglia
function as the primary immune cells within the brain, the effects
of JD-02 on HSV-1 infection in microglia were investigated.
Fluorescence data revealed a concentration-dependent decrease in
fluorescence intensity with the treatment of JD-02 (Fig. 2B). Additionally, RT-qPCR analysis
showed a significant, concentration-dependent reduction in the copy
number of the viral gene ICP0 following JD-02 administration in
Vero, BV2 and HaCaT cells (Fig.
2C-E). These findings substantiate the efficacy of JD-02 in
inhibiting HSV-1 infection across various cell types.
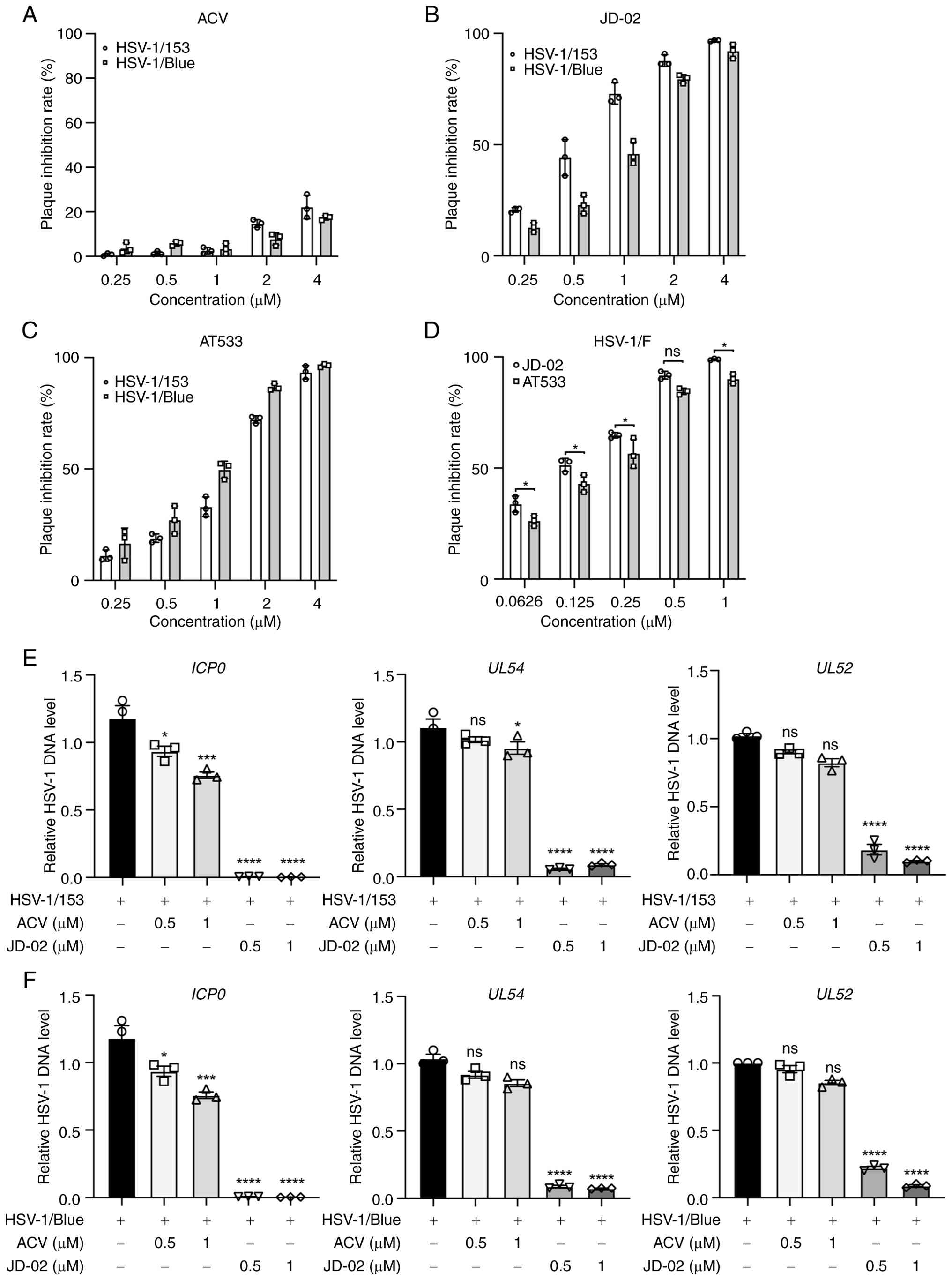
JD-02 inhibits normal and ACV-resistant
HSV-1 strains infection
The emergence of ACV-resistant strains poses a
considerable challenge in the clinical management of diseases
associated with HSV-1 (11,30). In the present study, the
antiviral efficacy of JD-02 was evaluated against two ACV-resistant
strains, HSV-1/153 and HSV-1/Blue, in HaCaT cells. A viral plaque
assay was performed to determine the antiviral efficacy of JD-2
against these ACV-resistant strains. The results indicated that ACV
was ineffective in inhibiting the infection efficiency of these two
viral strains (Fig. 3A).
Conversely, JD-02 significantly reduced the infection efficiency of
HSV-1/153 and HSV-1/Blue (Fig.
3B) in a dose-dependent manner, with half-maximal inhibitory
concentration (IC50) of 1.52 μM (HSV-1/153) and
0.72 μM (HSV-1/Blue). Additionally, AT533 also inhibited the
infection of HSV-1/153 and HSV-1/Blue (Fig. 3C), with IC50 values of
1.74 μM (HSV-1/153) and 1.39 μM (HSV-1/Blue),
although its efficacy was inferior to that of JD-02. Similarly,
JD-02 (IC50=0.11 μM) exhibited superior
inhibitory effects on HSV-1/F strains' infection compared with
AT533 (IC50=0.23 μM) (Fig. 3D). The DNA copy number analysis
further confirmed that JD-02 actively suppresses HSV-1/153
(Fig. 3E) and HSV-1/Blue
(Fig. 3F) in HaCaT cells. These
findings suggest that JD-02 is capable of inhibiting both normal
and ACV-resistant HSV-1 infection, indicating that its mechanism of
action against HSV-1 may differ from that of ACV.
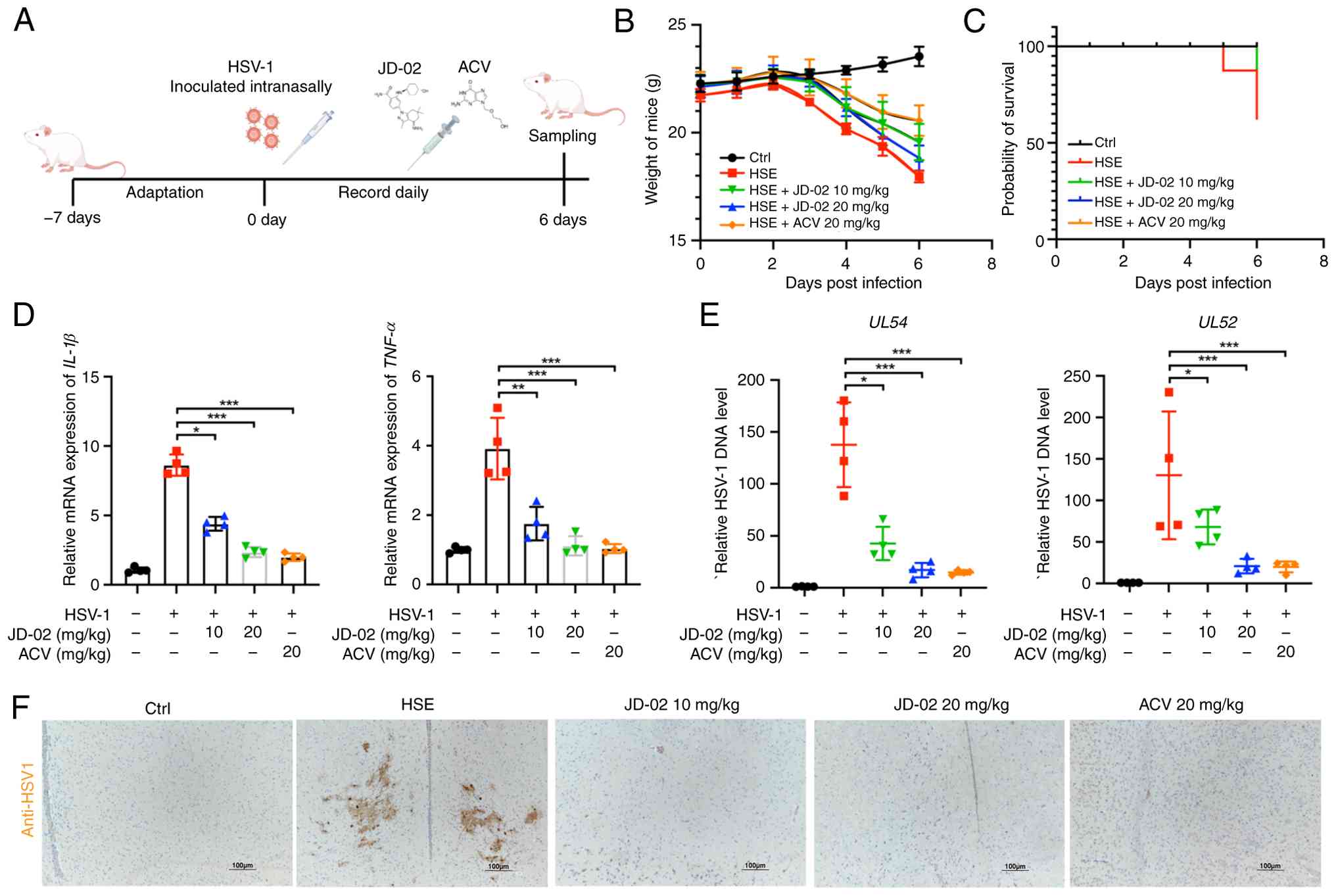
JD-02 alleviation of HSV-1 induced HSE in
vivo
To assess the anti-HSV-1 efficacy of JD-02 in
vivo, male BALB/c mice aged 5 weeks-old were nasally infected
with HSV-1 (Fig. 4A). JD-02, ACV
and a placebo (0.9% saline) were administered intraperitoneally.
The results indicated that both JD-02 and ACV effectively reversed
weight loss in mice (Fig. 4B),
enhanced survival rates (Fig.
4C), and mitigated symptoms associated with HSE, such as eye
swell (Fig. S2A), hair loss
(Fig. S2B) and hydrocephalus
(Fig. S2C). Treatment with
JD-02 significantly reduced the expression levels of inflammatory
cytokines, specifically IL-1β and TNF-α, in brain tissue (Fig. 4D) and decreased the DNA copy
number of the viral genes UL52 and UL54 in brain tissue (Fig. 4E). Additionally,
immunohistochemistry staining of brain tissues revealed a minimal
presence of viral particles in the JD-02 and ACV treatment groups
(Fig. 4F). H&E staining of
various tissues and organs showed that JD-02 administration did not
induce any damage to mouse organs (Fig. S3). In conclusion, JD-02
demonstrated potent anti-HSV-1 activity in vivo and
effectively ameliorated HSV-1-induced neurotropic infection and
neuroinflammation.
JD-02 plays a role in the early phase of
the HSV-1 life cycle
Subsequently, the potential antiviral mechanisms of
JD-02 were investigated. Given that the host's primary defense
against viral infections involves the activation of the antiviral
immune response, it was assessed whether JD-02 could activate this
response to inhibit HSV-1 infection. The RT-qPCR analyses indicated
that JD-02 administration led to a suppression of IFNB1 expression
(Fig. S4A) as well as CXCL10
expression (Fig. S4B). JD-02
also significantly reduced the expression levels of inflammatory
cytokines, including IL-1β, IL-6, and TNF-α (Fig. S4C-E). These findings suggest
that the antiviral activity of JD-02 is not mediated through
modulation of the host immune response.
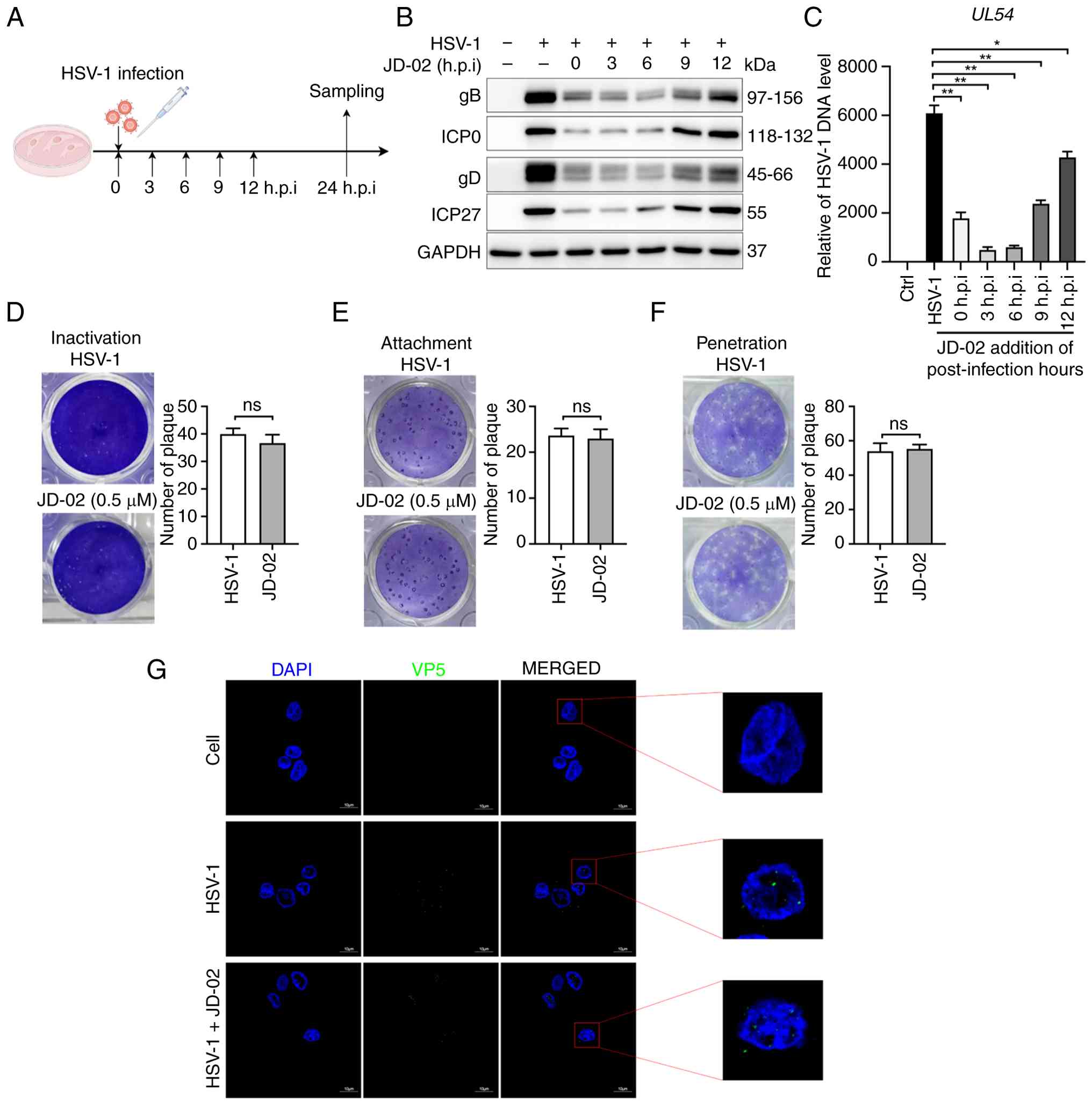
To evaluate JD-02 impact on viral protein expression
and viral gene copy number, time-of-addition experiments were
utilized (Fig. 5A). The western
blot indicated that JD-02 significantly inhibited viral protein
expression within 6 h (Fig. 5B)
corroborated by RT-qPCR results (Fig. 5C). Furthermore, consistent
findings were obtained in the evaluation of JD-02's impact on the
infection efficiency of EGFP-labeled HSV-1, as assessed through
fluorescence microscopy (Fig.
S5A) and virus plaque assays (Fig. S5B). These results suggest that
JD-02 exerts its antiviral effects during the initial stages of
HSV-1 infection. To investigate whether JD-02 directly targets
viral particles, viral plaque assay was performed. It was found
that JD-02 neither directly inactivated HSV-1 virions (Fig. 5D) nor affected the viral
adsorption (Fig. 5E) and
penetration processes (Fig. 5F).
Moreover, immunofluorescence analysis showed that JD-02 treatment
did not alter HSV-1 nuclear entry (Fig. 5G). Consequently, JD-02 exerts its
antiviral effect not by modulating the host immune response but by
affecting the early stages of HSV-1 infection.
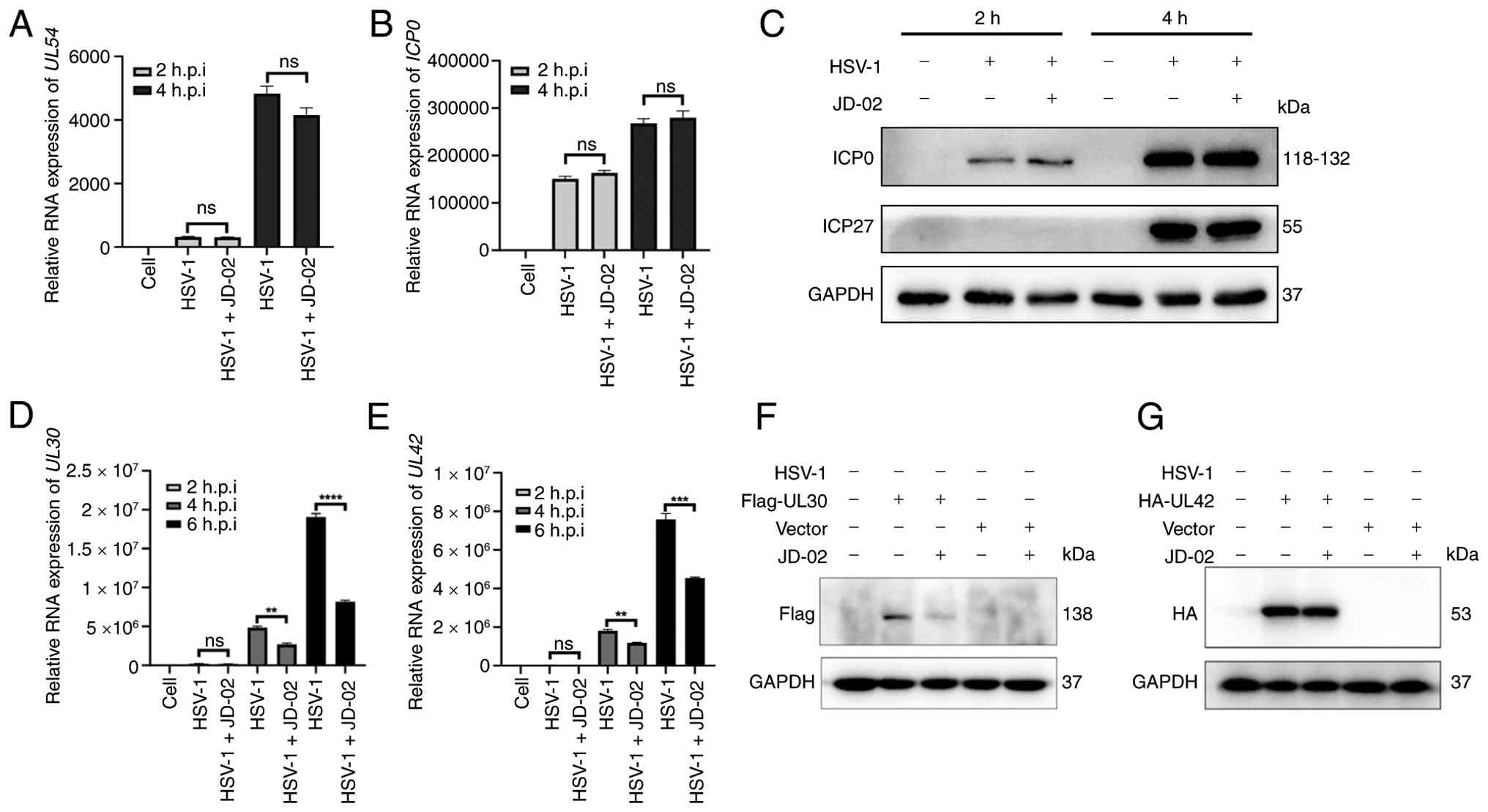
JD-02 suppresses genes linked to HSV-1
replication
Next, the potential impact of JD-02 on viral
immediate-early genes and the expression of genes associated with
early replication of HSV-1 was assessed. The expression levels of
the viral immediate-early genes UL54 (Fig. 6A) and ICP0 (Fig. 6B) were quantified and the
findings indicated that JD-02 did not influence the expression of
these immediate-early genes within 2- or 4-h post-treatment.
Western blot analysis further demonstrated that JD-02 did not alter
the expression of viral proteins ICP27 and ICP0 (Fig. 6C). However, additional analysis
revealed that JD-02 significantly suppressed the expression of
viral replication-related genes UL30 (Fig. 6D) and UL42 (Fig. 6E). To further investigate,
Flag-tagged UL30 and HA-tagged UL42 plasmids were overexpressed in
HaCaT cells, followed by treatment with JD-02. Western blot
analysis indicated that JD-02 effectively inhibited the protein
expression level of UL30 (Fig.
6F). It is noteworthy that JD-02 did not decrease the protein
levels of UL42 (Fig. 6G). This
observation suggests that the regulatory mechanism of JD-02 on UL42
requires further investigation. The aforementioned results further
prove that JD-02 exerts its anti-HSV-1 function by interfering with
the early viral infection events.
JD-02 inhibits HSV-1 replication by
suppressing Raf/MEK/ERK signaling pathway
Viruses, as obligate parasites, necessitate a host
for successful infection and replication. HSV-1, a DNA virus, has
been demonstrated to exploit the MAPK-ERK signaling pathway to
facilitate its replication and proliferation (31). Given that BRAF is a client
protein of Hsp90, the potential impact of JD-02 on the RAF-MEK-ERK
signaling cascade was explored. Through western blot analysis, both
in the presence and absence of JD-02 treatment, it was observed
that HSV-1 infection activates the BRAF/MEK/ERK signaling pathway,
resulting in a significant increase in the phosphorylation levels
of MEK and ERK (Fig. 7A).
Treatment with JD-02 significantly inhibited the activation of the
BRAF/MEK/ERK pathway induced by HSV-1 infection. Additionally, when
various concentrations of JD-02 were applied to HaCaT cells not
infected with HSV-1, JD-02 was found to downregulate the
phosphorylation levels of BRAF, MEK and ERK, without affecting ERK
protein expression (Fig. 7B).
These findings suggest that JD-02 may inhibit HSV-1 infection in
host cells by suppressing the activation of the RAF/MEK/ERK
signaling pathway.
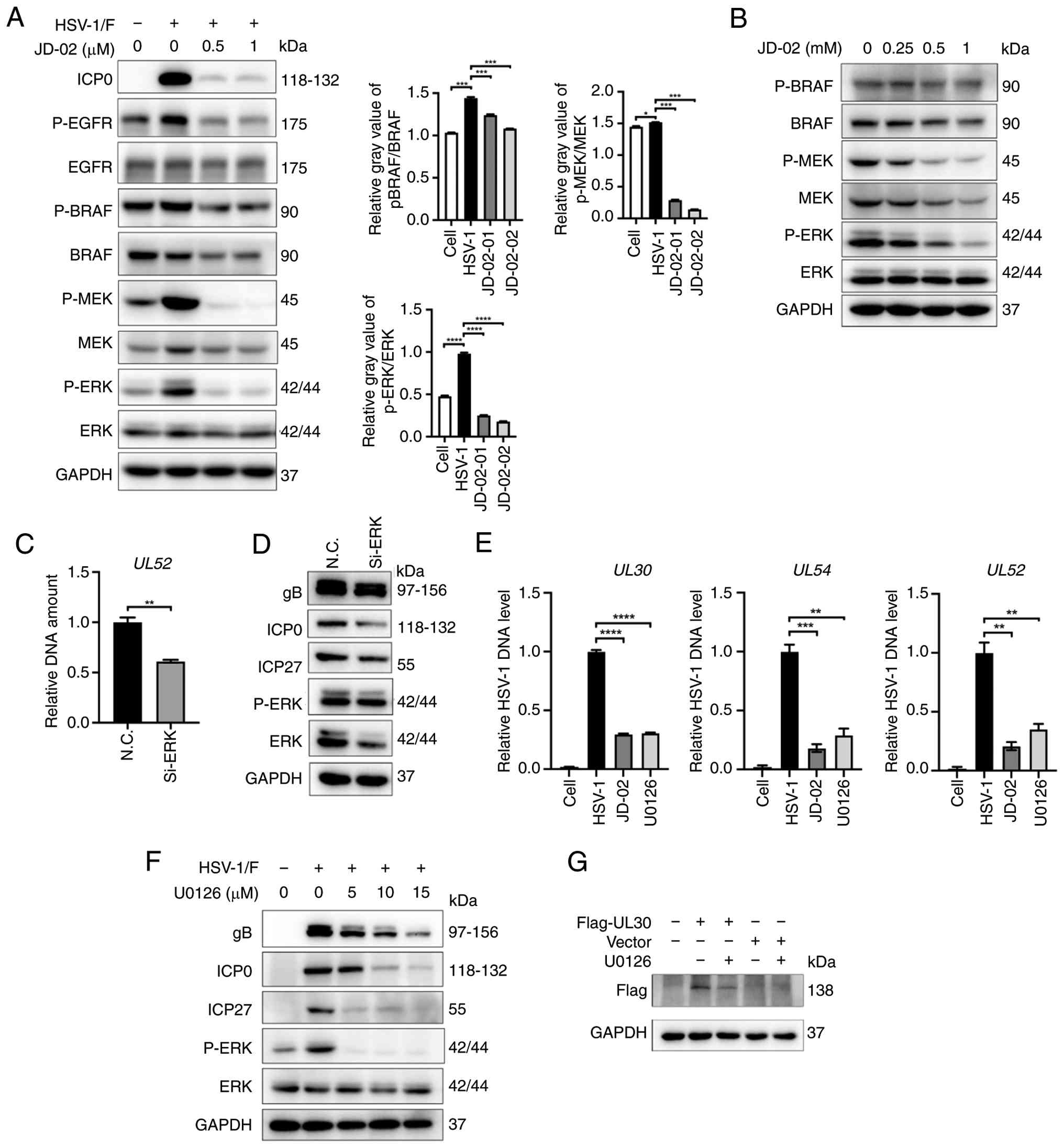 | Figure 7JD-02 inhibits HSV-1 replication by
suppressing the Raf/MEK/ERK signaling pathway. (A) Western blot
analysis was conducted to assess the effects of HSV-1 (MOI=0.1)
infection on the protein levels of BRAF, MEK and ERK, with and
without treatment using JD-02. (B) Treatment with JD-02 at the
specified concentration influences the protein levels of BRAF, MEK
and ERK in HaCaT cells over a 12-h period. (C and D) HaCaT cells
were subjected to transfection with either N.C. siRNA or ERK siRNA
for a period of 48 h. Subsequently, the cells were infected with
HSV-1 (MOI=0.1) for an additional 24 h. The DNA copy number of the
viral gene UL54, as well as the viral protein expression of gB,
ICP0, ICP27, ERK and p-ERK were evaluated. (E) The DNA copy numbers
of the viral genes UL30, UL52 and UL54 in
HaCaT cells infected with HSV-1 (MOI=0.1) and subsequently treated
with either JD-02 (1 μM) or U0126 (10 μM) for 24 h,
were quantified using reverse transcription-quantitative PCR. (F)
Western blot analysis of viral proteins (gB, ICP0 and ICP27), ERK
and p-ERK expression in HaCaT cell infected with HSV-1 (MOI=0.1)
and treated with U0126 for indicated concentration. (G) Western
blot analysis of UL30 overexpression in HaCaT cells treated with
U0126. Data are presented as the mean ± SD (n=3).
*P<0.05, **P<0.01,
***P<0.01 and ****P<0.0001 compared
with the HSV-1 group. HSV, Herpes Simplex Virus; MOI, multiplicity
of infection; N.C., negative control; siRNA, small interfering RNA;
p-, phosphorylated. |
To further elucidate the role of the ERK signaling
pathway in HSV-1 infection, siRNA was utilized to suppress ERK
expression and western blot analyses were conducted to assess the
efficacy of siRNA in suppressing ERK expression in HaCaT cells
(Fig. S6). Subsequently,
RT-qPCR analysis demonstrated that ERK inhibition led to a
significant reduction in the DNA copy number of the HSV-1 viral
gene UL52 (Fig. 7C), a
result that was corroborated by western blotting (Fig. 7D). Furthermore, HSV-1-infected
HaCaT cells were treated with the MEK/ERK inhibitors U0126 and
JD-02, respectively, which resulted in a significant decrease in
the expression levels of the viral genes UL30, UL54
and UL52 (Fig. 7E).
Additionally, western blotting indicated that treatment with U0126
significantly diminished the expression of viral proteins as well
as phosphorylated ERK (p-ERK) (Fig.
7F). Furthermore, the viral gene UL30 was exogenously
overexpressed and using U0126 still markedly inhibited viral
protein expression (Fig. 7G).
These findings provide compelling evidence supporting the
hypothesis that JD-02 exerts its antiviral effects against HSV-1
through the inhibition of the RAF/MEK/ERK signaling pathway.
Discussion
Considering the high prevalence of HSV-1 infection,
the absence of an effective vaccine, and the frequent emergence of
drug-resistant strains, the development of novel and efficacious
therapeutic agents against HSV-1 is imperative for enhancing
current clinical treatments for HSV-1-associated diseases (10,32). The present study explored the
efficacy of a novel HSP90 inhibitor (33), JD-02, a benzamide derivative,
which demonstrated significant inhibitory effects on both standard
HSV-1 and ACV-resistant strains. Moreover, JD-02 exhibited reduced
cytotoxicity compared with the conventional HSP90 inhibitor AT533
and mitigated disease symptoms in HSE mouse models. These findings
suggest its potential as a promising HSP90 inhibitor for the
development of therapeutic agents targeting HSV-1-related
diseases.
HSV-1, a prototypical neurotropic virus, can cause
HSE and establishing lifelong latent infections by targeting the
CNS (34). This poses a
considerable threat to the health and survival of neonates and
immunocompromised individuals (12). Presently, nucleoside analogs that
inhibit viral DNA polymerases constitute the mainstay of
therapeutic interventions. Nonetheless, the extensive utilization
of these antiviral agents has led to an increase in the prevalence
of drug-resistant mutations (35), underscoring the urgent need for
the development of novel therapeutic compounds (36). HSP90 is a crucial molecular
chaperone implicated in various stages of the herpesvirus life
cycle (14,37). Numerous studies have validated
HSP90 as a viable target for anti-herpesvirus therapies (14,27,38,39). However, challenges such as the
toxicity of inhibitors have resulted in clinical trials falling
short of expectations. Previous advancements in research have led
to the progressive refinement of HSP90 inhibitors. Notably, the
orally bioavailable HSP90 inhibitor SNX-5422 has demonstrated
efficacy in inhibiting the replication of SARS-CoV-2 (40). The HSP90 inhibitor Pimitespib has
received clinical approval in Japan (41), as documented in the literature,
underscoring the significance of HSP90 as a critical drug target.
In the authors' prior research, a series of benzoyl analogs were
synthesized, including SNX-2112 and AT533, which demonstrated
efficacy against HSV-1 infection, thereby suggesting the promising
potential of HSP90 inhibitors as antiviral agents (27,42). In the present study, six
compounds were designed based on the benzoyl core structure. These
compounds have been previously identified as novel HSP90 inhibitors
capable of inhibiting colorectal cancer growth (33). Furthermore, the findings of the
present study revealed that JD-02 exhibits superior cytotoxicity
compared with the HSP90 inhibitor AT533. In the HSE model mice, the
in vivo antiviral efficacy of JD-02 is comparable to that of
the nucleoside analog ACV.
In the present study, JD-02, a novel inhibitor of
Hsp90, demonstrated significant inhibitory effects against both
common HSV-1 and ACV-resistant strains, HSV-1-blue and HSV-1-153.
At equivalent concentrations, JD-02 exhibited superior in
vitro inhibitory activity against HSV-1 compared with ACV.
JD-02 significantly reduced the copy numbers of virus-related genes
(UL54, UL52, ICP0 and UL27) as well as
the expression levels of virus-related proteins (gB, ICP0, gD,
ICP27 and UL30). To elucidate the mechanism underlying JD-02's
antiviral action, its potential antiviral pathways were explored.
The experimental results indicated that JD-02 does not directly
target HSV-1 and does not significantly influence HSV-1 adsorption
or penetration. Further investigation revealed that JD-02 exerts
its anti-HSV-1 activity by inhibiting the RAF/MEK/ERK signaling
pathway, which is crucial for cell proliferation, differentiation
and survival (43). BRAF, a
client protein of HSP90, plays a significant role in HSV-1
infection of the host (44).
Consistent with previous findings, HSV-1 infection significantly
enhances the activation of this signaling pathway (17,45). The present research demonstrated
that JD-02 effectively inhibits the activation of the RAF/MEK/ERK
signaling pathway, as evidenced by a significant reduction in the
phosphorylation levels of BRAF, MEK and ERK following JD-02
treatment. Furthermore, the current findings revealed that the ERK
inhibitor U0126 also suppresses HSV-1 infection, suggesting that
ERK may serve as a critical target for anti-HSV-1 strategies. This
warrants further investigation.
It is noteworthy that therapeutics targeting host
factors present several advantages over those that directly target
viral components. Such drugs often exhibit broad-spectrum antiviral
activity and are less prone to inducing drug resistance. This is
particularly significant given the frequent emergence of resistance
in viruses subjected to treatments targeting specific viral
elements, posing a substantial challenge in clinical settings. The
documented resistance of HSV and the resistance to remdesivir or
nirmatrelvir caused by SARS-CoV-2 mutations both demonstrate this
issue (46,47). However, the likelihood of HSV-1
developing resistance to JD-02 through mutation is remarkably low.
The absence of drug-resistant viral strains following the use of
HSP90 inhibitors in HSV-1 infections underscores the critical role
of HSP90 as a host factor essential for the viral lifecycle
(38), rendering it a promising
therapeutic target. Consequently, the development of novel HSP90
inhibitors with reduced toxicity, building upon traditional HSP90
inhibitors, holds significant potential for HSV-1 treatment and
merits further investigation.
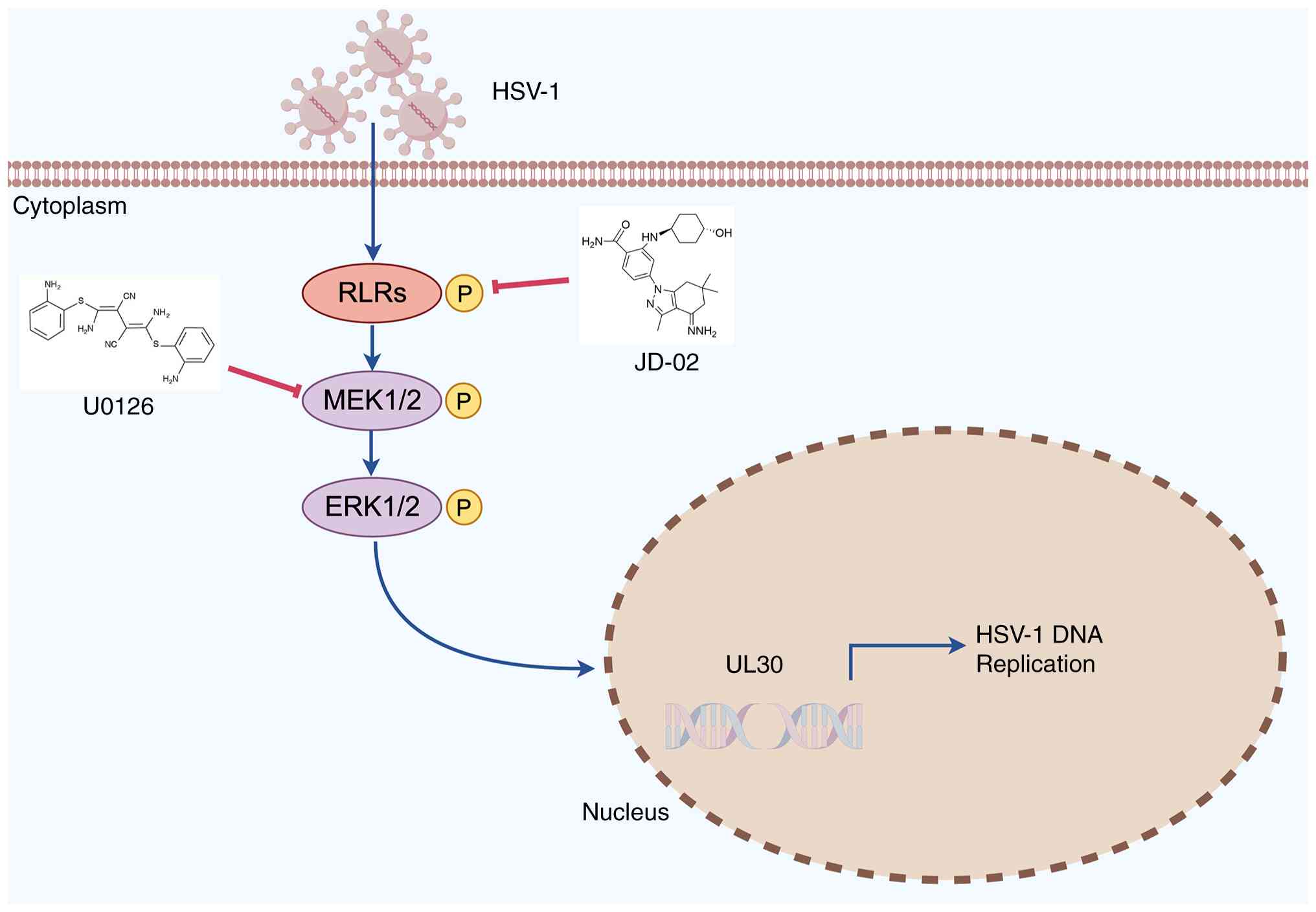
In conclusion, the present research demonstrated
that JD-02, a novel HSP90 inhibitor, effectively suppresses HSV-1
replication and mitigates HSE symptoms via the RAF/MEK/ERK
signaling pathway (Fig. 8).
Notably, JD-02 shows reduced toxicity and enhanced viral inhibition
efficacy compared with the conventional HSP 90 inhibitor AT533.
Furthermore, JD-02 exhibits the capacity to inhibit HSV-1 infection
in ACV-resistant strains. These findings propose that JD-02, as a
novel HSP90 inhibitor, holds promise for development as an
antiviral therapeutic agent for HSV-1 infection-related
diseases.
Supplementary Data
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YW and JX conceptualized the study. XiaohW, YZ and
JL performed the experimental studies. XiaohW, JL and XiaoW
validated data. YZ and JX wrote the manuscript. JL and XiaoW
revised the manuscript. KZ and ZR were involved in the conduction
of animal experiments. YW and JX was responsible for study
supervision and funding acquisition. YZ, YW and JX confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
Animal experiments were conducted in accordance with
laboratory animal management guidelines. This study was conducted
according to the ethical policies and procedures approved by the
Animal Care and Use Committee of Jinan University (approval no.
20200402-07; Guangzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that have no competing
interests.
Abbreviations:
|
ACV
|
acyclovir
|
|
CPE
|
cytopathic effect
|
|
MOI
|
multiplicity of infection
|
|
HSV-1
|
Herpes simplex virus 1
|
|
HSE
|
Herpes simplex virus encephalitis
|
|
IFN
|
interferon
|
|
RT-qPCR
|
reverse transcription-quantitative
PCR
|
|
DMEM
|
Dulbecco's modified Eagle's medium
|
|
FBS
|
fetal bovine serum
|
|
PBS
|
phosphate-buffered saline
|
|
PFU
|
plaque-forming units
|
|
PVDF
|
polyvinylidene difluoride
|
Acknowledgements
The authors would like to thank Professor Yunsheng
Huang (Guangdong Medical University) for providing six benzamide
derivatives.
Funding
The present study was supported by the National Natural Science
Foundation of China (grant nos. 82473972 and 82373917), the Key
Basic Research Project of Shenzhen (grant no.
JCYJ20220818102605011) and Key Scientific Research Project of the
Health System in Zigong City (grant no. 24zd001].
References
|
1
|
Whitley RJ and Roizman B: Herpes simplex
virus infections. Lancet. 357:1513–1518. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wang Z, Liu J, Han J, Zhang T, Li S, Hou
Y, Su H, Han F and Zhang C: Herpes simplex virus 1 accelerates the
progression of Alzheimer's disease by modulating microglial
phagocytosis and activating NLRP3 pathway. J Neuroinflammation.
21:1762024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Marcocci ME, Napoletani G, Protto V,
Kolesova O, Piacentini R, Li Puma DD, Lomonte P, Grassi C, Palamara
AT and De Chiara G: Herpes simplex virus-1 in the brain: The dark
side of a sneaky infection. Trends Microbiol. 28:808–820. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Diaz JJ, Dodon MD, Schaerer-Uthurralt N,
Simonin D, Kindbeiter K, Gazzolo L and Madjar JJ:
Post-transcriptional transactivation of human retroviral envelope
glycoprotein expression by herpes simplex virus Us11 protein.
Nature. 379:273–277. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Andrade VM, Pereira-Dutra F, Abrantes JL,
Miranda MD and Souza TML: HSV1-induced enhancement of productive
HIV-1 replication is associated with interferon pathway
downregulation in human macrophages. Mem Inst Oswaldo Cruz.
119:e2401022024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Linard M, Letenneur L, Garrigue I, Doize
A, Dartigues JF and Helmer C: Interaction between APOE4 and herpes
simplex virus type 1 in Alzheimer's disease. Alzheimers Dement.
16:200–208. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Baggen J, Vanstreels E, Jansen S and
Daelemans D: Cellular host factors for SARS-CoV-2 infection. Nat
Microbiol. 6:1219–1232. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Protto V, Marcocci ME, Miteva MT,
Piacentini R, Li Puma DD, Grassi C, Palamara AT and De Chiara G:
Role of HSV-1 in Alzheimer's disease pathogenesis: A challenge for
novel preventive/therapeutic strategies. Curr Opin Pharmacol.
63:1022002022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Coleman JL and Shukla D: Recent advances
in vaccine development for herpes simplex virus types I and II. Hum
Vaccin Immunother. 9:729–735. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sadowski LA, Upadhyay R, Greeley ZW and
Margulies BJ: Current drugs to treat infections with herpes simplex
viruses-1 and -2. Viruses. 13:12282021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bacon TH, Levin MJ, Leary JJ, Sarisky RT
and Sutton D: Herpes simplex virus resistance to acyclovir and
penciclovir after two decades of antiviral therapy. Clin Microbiol
Rev. 16:114–128. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Piret J and Boivin G: Antiviral resistance
in herpes simplex virus and varicella-zoster virus infections:
Diagnosis and management. Curr Opin Infect Dis. 29:654–662. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Berrington William R, Jerome Keith R, Cook
L, Wald A, Corey L and Casper C: Clinical correlates of herpes
simplex virus viremia among hospitalized adults. Clin Infect Dis.
49:1295–1301. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chakraborty A, Roos-Mattjus P and
Gramolelli S: Therapeutic targeting of HSP90 in herpesvirus
infections, past and future challenges. Trans R Soc S Afr.
80:47–52. 2025. View Article : Google Scholar
|
|
15
|
Ullah R, Yin Q, Snell AH and Wan L:
RAF-MEK-ERK pathway in cancer evolution and treatment. Semin Cancer
Biol. 85:123–154. 2022. View Article : Google Scholar
|
|
16
|
Higgins CA, Nilsson-Payant BE, Bonaventure
B, Kurland AP, Ye C, Yaron TM, Johnson JL, Adhikary P, Golynker I,
Panis M, et al: SARS-CoV-2 hijacks p38β/MAPK11 to promote virus
replication. mBio. 14:e01007232023. View Article : Google Scholar
|
|
17
|
DuShane JK and Maginnis MS: Human DNA
virus exploitation of the MAPK-ERK cascade. Int J Mol Sci.
20:34272019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Watanabe M, Arii J, Takeshima K, Fukui A,
Shimojima M, Kozuka-Hata H, Oyama M, Minamitani T, Yasui T, Kubota
Y, et al: Prohibitin-1 contributes to cell-to-cell transmission of
herpes simplex virus 1 via the MAPK/ERK signaling pathway. J Virol.
95:e01413202021. View Article : Google Scholar :
|
|
19
|
Lv W, Zhou L, Wu J, Cheng J, Duan Y and
Qian W: Anti-HSV-1 agents: An update. Front Pharmacol.
15:14510832025. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Koujah L, Madavaraju K, Agelidis AM, Patil
CD and Shukla D: Heparanase-induced activation of AKT stabilizes
β-catenin and modulates Wnt/β-catenin signaling during herpes
simplex virus 1 infection. mBio. 12:e02792212021. View Article : Google Scholar
|
|
21
|
Kim Y, Lim SY, Kim HO, Ha SJ, Park JA, Won
YW, Chae S and Lim KS: Combination strategies with HSP90 inhibitors
in cancer therapy: Mechanisms, challenges, and future perspectives.
Pharmaceuticals (Basel). 18. pp. 10832025, View Article : Google Scholar
|
|
22
|
Jiang H, Lan N, Ma W, Zhang Z, Zhao Z, Hu
Y, Su Y, Huang Y, Wang Y, Xu D and Liu K: Synthesis and evaluation
of the antitumor activity of
2-amino-4-tetrahydroindazole-substituted benzamide derivatives as
HSP90 inhibitors. J Mol Struct. 1300:1372662024. View Article : Google Scholar
|
|
23
|
Huang Z, Li S, Zhong L, Su Y, Li M, Wang
X, Wang Z, Wang Z, Ye C, Ren Z, et al: Effect of resveratrol on
herpesvirus encephalitis: Evidences for its mechanisms of action.
Phytomedicine. 127:1554762024. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Song X, Wang Y, Zou W, Wang Z, Cao W,
Liang M, Li F, Zeng Q, Ren Z, Wang Y and Zheng K: Inhibition of
mitophagy via the EIF2S1-ATF4-PRKN pathway contributes to viral
encephalitis. J Adv Res. 73:199–217. 2025. View Article : Google Scholar :
|
|
25
|
Li F, Wang Y, Song X, Wang Z, Jia J, Qing
S, Huang L, Wang Y, Wang S, Ren Z, et al: The intestinal microbial
metabolite nicotinamide n-oxide prevents herpes simplex
encephalitis via activating mitophagy in microglia. Gut Microbes.
14:20969892022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang Y, Luo W, Wang X, Ma Y, Huang L and
Wang Y: MAMDC2, a gene highly expressed in microglia in
experimental models of Alzheimers disease, positively regulates the
innate antiviral response during neurotropic virus infection. J
Infect. 84:187–204. 2022. View Article : Google Scholar
|
|
27
|
Li F, Song X, Su G and Wang Y, Wang Z,
Qing S, Jia J and Wang Y, Huang L, Zheng K and Wang Y: AT-533, a
Hsp90 inhibitor, attenuates HSV-1-induced inflammation. Biochem
Pharmacol. 166:82–92. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang YL, Luo WS, Huang LZ, Xiao J, Song X,
Li F, Ma Y, Wang X, Jin F, Liu P, et al: A novel lncRNA linc-AhRA
negatively regulates innate antiviral response in murine microglia
upon neurotropic herpesvirus infection. Theranostics. 11:9623–9651.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
30
|
Jiang YC, Feng H, Lin YC and Guo XR: New
strategies against drug resistance to herpes simplex virus. Int J
Oral Sci. 8:1–6. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ludwig S, Pleschka S and Planz O: MEK
inhibitors as novel host-targeted antivirals with a dual-benefit
mode of action against hyperinflammatory respiratory viral
diseases. Curr Opin Virol. 59:1013042023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Duarte LF, Farías MA, Alvarez DM, Bueno
SM, Ríedel CA and González PA: Herpes simplex virus type 1
infection of the central nervous system: Insights into proposed
interrelationships with neurodegenerative disorders. Front Cell
Neurosci. 13:462019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lan N, Su Y, Zeng Q, Zhou P, Hu Y, Zhang
Z, Wang Y and Liu K: JD-02, a novel Hsp90 inhibitor, induces
ROS/SRC axis-dependent cytoprotective autophagy in colorectal
cancer cells. Mol Carcinog. 63:1038–1050. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
James C, Harfouche M, Welton NJ, Turner
KM, Abu-Raddad LJ, Gottlieb SL and Looker KJ: Herpes simplex virus:
Global infection prevalence and incidence estimates, 2016. Bull
World Health Organ. 98:315–329. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shankar S, Pan J, Yang P, Bian Y, Oroszlán
G, Yu Z, Mukherjee P, Filman DJ, Hogle JM, Shekhar M, et al: Viral
DNA polymerase structures reveal mechanisms of antiviral drug
resistance. Cell. 187:5572–5586.e15. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cakir M, Obernier K, Forget A and Krogan
NJ: Target discovery for host-directed antiviral therapies:
Application of proteomics approaches. mSystems. 6:e00388212021.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang Y, Wang R, Li F and Wang Y, Zhang Z,
Wang Q, Ren Z, Jin F, Kitazato K and Wang Y: Heat-shock protein 90α
is involved in maintaining the stability of VP16 and VP16-mediated
transactivation of α genes from herpes simplex virus-1. Mol Med.
24:652018. View Article : Google Scholar
|
|
38
|
Lubkowska A, Pluta W, Strońska A and Lalko
A: Role of heat shock proteins (HSP70 and HSP90) in viral
infection. Int J Mol Sci. 22:93662021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Qin S, Hu X, Lin S, Xiao J, Wang Z, Jia J,
Song X, Liu K, Ren Z and Wang Y: Hsp90 inhibitors prevent HSV-1
replication by directly targeting UL42-Hsp90 complex. Front
Microbiol. 12:7972792022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Goswami R, Russell VS, Tu JJ, Thomas C,
Hughes P, Kelly F, Langel SN, Steppe J, Palmer SM, Haystead T, et
al: Oral Hsp90 inhibitor SNX-5422 attenuates SARS-CoV-2 replication
and dampens inflammation in airway cells. iScience. 24:1034122021.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kurokawa Y, Honma Y, Sawaki A, Naito Y,
Iwagami S, Komatsu Y, Takahashi T, Nishida T and Doi T: Pimitespib
in patients with advanced gastrointestinal stromal tumor
(CHAPTER-GIST-301): A randomized, double-blind, placebo-controlled
phase III trial. Ann Oncol. 33:959–967. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Xiang YF, Qian CW, Xing GW, Hao J, Xia M
and Wang YF: Anti-herpes simplex virus efficacies of
2-aminobenzamide derivatives as novel HSP90 inhibitors. Bioorg Med
Chem Lett. 22:4703–4706. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li Q, Li Z, Luo T and Shi H: Targeting the
PI3K/AKT/mTOR and RAF/MEK/ERK pathways for cancer therapy. Mol
Biomed. 3:472022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Qin D, Feng N, Fan W, Ma X, Yan Q, Lv Z,
Zeng Y, Zhu J and Lu C: Activation of PI3K/AKT and ERK MAPK signal
pathways is required for the induction of lytic cycle replication
of Kaposi's sarcoma-associated herpesvirus by herpes simplex virus
type 1. BMC Microbiol. 11:2402011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Cheng Y, Sun F, Wang L, Gao M, Xie Y, Sun
Y, Liu H, Yuan Y, Yi W, Huang Z, et al: Virus-induced p38 MAPK
activation facilitates viral infection. Theranostics.
10:12223–12240. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hu Y, Lewandowski EM, Tan H, Zhang X,
Morgan RT, Zhang X, Jacobs LMC, Butler SG, Gongora MV, Choy J, et
al: Naturally occurring mutations of SARS-CoV-2 main protease
confer drug resistance to nirmatrelvir. ACS Cent Sci. 9:1658–1669.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Vitiello A: Sars-Cov-2 and risk of
antiviral drug resistance. Ir J Med Sci. 191:2367–2368. 2022.
View Article : Google Scholar
|