Introduction
Intestinal diseases are common clinical conditions with persistently high incidence and mortality rates, making them global health issues (1,2). Intestinal diseases, such as inflammatory bowel disease (IBD), colorectal cancer (CRC) and intestinal inflammation, affect the small and large intestine and rectum (3). The pathogenesis of these diseases is complex and involves multiple factors, such as immune dysregulation, oxidative stress and gut microbiota dysbiosis (4). Intestinal diseases not only impact patient quality of life but also impose a socioeconomic burden on healthcare systems (5). In recent years, treatment for intestinal diseases has relied primarily on anti-inflammatory drugs (5-aminosalicylic acid and glucocorticoids) (6), immunosuppressants (azathioprine and biological agents) (7) and surgical interventions (8). However, certain patients face challenges such as inadequate efficacy, drug resistance or adverse effects (6-8). Further elucidation of the mechanisms underlying disease onset and progression is needed to identify novel therapeutic targets.
Itaconate (ITA) is a metabolite synthesized by immune cells such as macrophages under inflammatory or infectious conditions. It is catalysed by cis-dihydroconiferyl acid decarboxylase (CAD), which is encoded by immune response gene 1 (IRG1) (9,10). A derivative of the tricarboxylic acid (TCA) cycle, ITA was initially discovered for its antibacterial properties (11). Subsequent research revealed its involvement not only in energy metabolism but also in immune regulation and oxidative stress modulation (11-14). ITA exerts its effects by covalently modifying proteins, such as Kelch-like ECH-associated protein 1 (Keap1) and GAPDH, regulating the antioxidant pathway of Nrf2, inhibiting the activation of nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) inflammasomes and influencing immune cell function (14,15). As the largest immune and microbial ecosystem in the human body, gastrointestinal homeostasis is associated with metabolic regulation (16). Increasing evidence suggests that ITA may serve a pivotal role in the onset or progression of various intestinal diseases, including IBD, CRC and intestinal inflammation, by modulating immune responses, maintaining intestinal barrier function and influencing microbial composition (17-19). Therefore, elucidating the mechanisms of ITA in intestinal diseases may provide key insights for the development of novel metabolic immunotherapies. The present review summarizes the biosynthesis, regulatory mechanisms and potential roles of ITA in several intestinal disorders while exploring the feasibility of targeting ITA and its derivatives as novel therapeutic agents.
Discovery and molecular structure of ITA
ITA was first discovered by Samuel Baup during his research on the thermal decomposition of citric acid (20). Subsequently, Kinoshita (21) identified a strain of Aspergillus terreus that is capable of producing and secreting ITA in large quantities. The biosynthetic method used by the fungus, in which glucose is fermented to generate ITA, is the primary approach for industrial production (22). ITA is present in the lungs of mice infected with Mycobacterium tuberculosis (23). ITA is formed during the TCA cycle as a mammalian metabolic byproduct and is produced in large quantities in lipopolysaccharide (LPS)-activated macrophages (9). This implies that ITA serves a crucial role in macrophage immunity.
The molecular structure of ITA is distinct. Its chemical formula is C5H6O4 and it features a polymerizable double bond and two carboxyl groups. These carboxyl groups confer the properties of a dicarboxylic acid, enabling it to undergo typical carboxylic acid reactions, such as neutralization, esterification and salt formation (24). The direct linkage between the double bond and one carboxyl group results in the formation of a highly reactive α,β-unsaturated carbonyl compound. This linkage allows ITA to serve as an acid in reactions and a vinyl monomer for polymerization, and participate in reactions such as Michael addition (25). ITA is widely used in the synthesis of resin, plastic and chemical intermediates (24,25). Owing to its chemical structure, ITA has poor cell permeability, potentially limiting the transport of exogenous ITA into the cytoplasm (26). The mechanism underlying the transmembrane transport of ITA remains unclear, limiting direct studies on its function. Researchers have therefore developed membrane-permeable derivatives such as 4-octyl ITA (4OI) (12) and dimethyl ITA (DI) (27), which serve as key research tools for exploring the functions and mechanisms of action of ITA (12,27).
Synthesis and metabolism of ITA
ITA is synthesized in both mammals and microorganisms (12,22) (Fig. 1). In mammals, ITA biosynthesis is associated with citrate metabolism within the TCA cycle (9). In the classical TCA cycle, citrate is converted to cis-aconitate by aconitase, which is metabolised to cis-aconitate and enters oxidative pathways (28). However, when macrophages are stimulated by LPS or proinflammatory cytokines, such as IFN-γ or tumour necrosis factor-α (TNF-α) (29,30), a portion of cis-aconitate enters the ITA synthesis pathway (31). ITA synthesis consumes substantial amounts of cis-aconitate, disrupting the TCA cycle and inhibiting the mitochondrial respiratory chain function. This promotes increased glycolytic metabolism, a phenomenon termed 'immune metabolic reprogramming' (32). The key regulator of this metabolic reprogramming is IRG1 and its encoded CAD (11). IRG1 resides within mitochondria and is expressed at low levels in resting macrophages, however, its expression significantly increases upon inflammatory signal activation (33-35). IRG1 catalyses the decarboxylation of cis-aconitate, generating ITA and releasing CO2 (36). This reaction relies on Fe2+ as a cofactor and may be influenced by the intracellular redox status (37). Furthermore, ITA synthesis is associated with macrophage polarisation. Under external stimulation, proinflammatory macrophages (M1 type) highly express IRG1 and produce substantial amounts of ITA, whereas anti-inflammatory/reparative macrophages (M2 type) synthesise negligible amounts of ITA (38). This selective expression pattern suggests that ITA may serve a crucial role in regulating inflammatory responses.
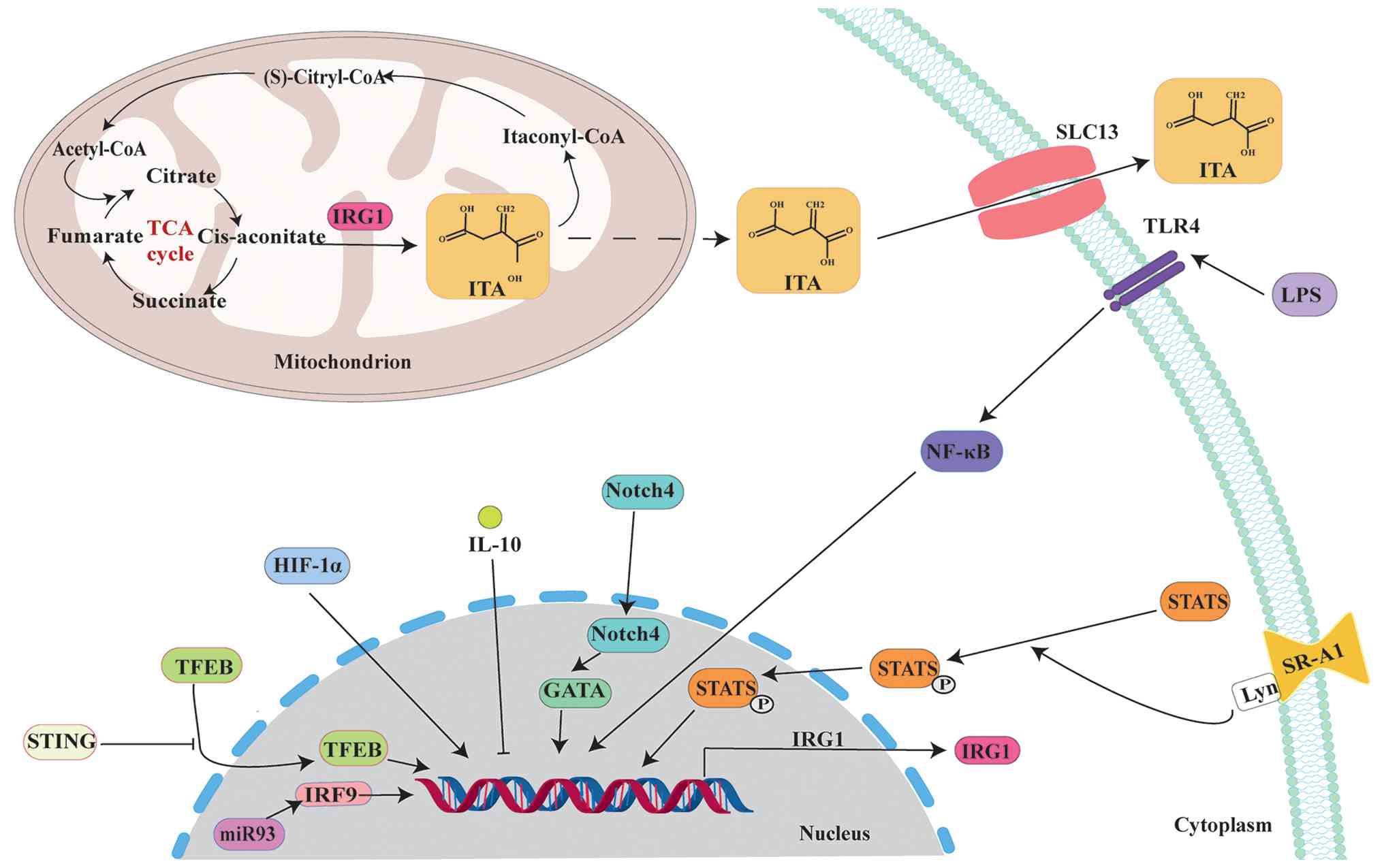 |
Figure 1
Synthesis and metabolism of ITA. ITA biosynthesis occurs via the decarboxylation of citrate in the TCA cycle and is subject to multi-level regulation. Following its production within the mitochondria, ITA is shuttled to the extracellular space by specific membrane transporters to exert its effects. ITA, itaconate; STING, stimulator of interferon genes; TFEB, transcription factor EB; miR, microRNA; IRF9, interferon regulatory factor 9; HIF-1α, hypoxia inducible factor-1α; Notch4, neurogenic locus notch homolog 4; LPS, lipopolysaccharide; TLR4, toll-like receptor 4; SR-A1, scavenger receptor A1; TCA, tricarboxylic acid; SLC, solute carrier family.
|
As a key regulator of ITA synthesis, IRG1 expression is modulated by multiple signalling pathways. LPS activates NF-κB signalling via toll-like receptor 4, directly inducing IRG1 transcription and promoting ITA production (35). Hypoxia-inducible factor-1α (HIF-1α) also increases IRG1 expression, further stimulating ITA generation (39). Type I IFN suppresses IRG1 expression via IL-10, reducing ITA synthesis in macrophages (40). Absolute or relative deficiency of microRNA-93 in hypoxic, serum-starved macrophages leads to increased IFN regulatory factor 9-IRG1 expression, resulting in elevated ITA production (41). Additionally, the neurogenic locus notch homologue 4 (NOTCH4)/GATA binding protein (GATA)/IRG1, scavenger receptor A1/STAT3/IRG1 and lysosomal biogenesis factor transcription factor EB (TFEB)/IRG1 pathways (42-44) also regulate ITA synthesis.
In addition to mammals, certain pathogens (Aspergillus fumigatus) and engineered microorganisms also synthesise ITA, although their synthesis pathways differ from those in mammalian cells (21,45). Studies indicate that fungi directly convert cis-aconitate via homologous CAD enzymes, whereas certain bacteria utilise the isocitrate lyase (ICL) pathway to indirectly generate ITA (46,47).
ITA accumulates at relatively high levels within cells but is present in plasma at micromolar concentrations (48). Research has indicated that ITA is primarily excreted via the kidney but is also taken up and metabolised by tissues such as the liver (49). ITA is a metabolite that is produced when energy generation bypasses the TCA cycle (31). ITA is first catalysed by succinyl-CoA synthase to form itaconyl-CoA. Itaconyl-CoA undergoes hydration catalysed by methyl-5-enyl-2-pyruvate hydratase to form (S)-citryl-CoA. (S)-citryl-CoA is cleaved by citraldehyde decarboxylase to yield acetyl-CoA and pyruvate (50,51). Additionally, ITA is secreted into the extracellular microenvironment via specific carboxylate transporters (members of the solute carrier 13 family) following synthesis (52).
Role of ITA
Immunoregulatory function of ITA
ITA has been demonstrated to act as an immunoregulator in macrophages (53). After ITA is activated by IRG1, which is located in the mitochondrial matrix of macrophages, it traverses the inner mitochondrial membrane to affect cytoplasmic Nrf2 (14). Nrf2 is a transcription factor whose N-terminal domain binds Keap1, leading to Nrf2 degradation via the proteasome and suppression of its activity (54). ITA directly alkylates the cysteine residues of the Keap1 protein, preventing the Keap1-Nrf2 interaction. This facilitates Nrf2 translocation into the nucleus, where it activates the transcription of genes associated with anti-inflammatory and antioxidant responses and promotes the expression of multiple antioxidant and anti-inflammatory proteins, including heme oxygenase-1 (HO-1), NAD (P) H:quinone oxidoreductase 1 and cyclooxygenase-2 (14), thereby exerting anti-inflammatory and antioxidant effects.
The activation of immune cells and production of inflammatory mediators depend on the reprogramming of energy metabolism, particularly the shift towards glycolysis (55). ITA serves as an allosteric inhibitor of pyruvate kinase M2 (PKM2), a key glycolytic enzyme (56). By inhibiting PKM2, ITA suppresses glycolysis (56,57), decreasing the energy and biomolecular substrates that are required for macrophage activation. This limits their ability to produce proinflammatory factors (TNF-α, IL-1β, and IL-6) (58,59). Furthermore, the low-activity dimeric form of PKM2 enters the nucleus and serves as a transcriptional coactivator to increase the expressions of genes such as IL-1β (60). ITA indirectly inhibits this process by suppressing PKM2.
ITA influences the mitochondrial respiratory chain function by inhibiting succinate dehydrogenase (SDH), thereby decreasing the production of proinflammatory factors such as IL-1β and IL-6 (61,62). Further research has indicated that ITA, by inhibiting SDH activity, attenuates HIF-1α/NLRP3 signalling, promoting macrophage polarisation from the M1 to the M2 phenotype, thereby alleviating inflammation (63). ITA inhibits NLRP3 inflammasome activation, reducing the maturation and release of the proinflammatory factors IL-1β and IL-18 (15). ITA also inhibits JAK1 activity, decreasing the signalling of cytokines such as IFN-γ (38). ITA alkylates the transcription coactivator IκBζ, suppressing its transcriptional activity and inhibiting the expressions of genes such as IL-6 (64). In summary, ITA simultaneously targets multiple key inflammatory signalling nodes, including IκBζ, JAK1 and NLRP3, thereby suppressing the expression and functions of several proinflammatory cytokines. This provides the molecular basis for its potent anti-inflammatory activity. In macrophages, low doses (0-120 μM) of 4-OI suppress inflammation, whereas high doses (480 μM) promote inflammation (65), demonstrating the dual regulation of inflammation by ITA.
ITA disrupts mitochondrial function in dendritic cells, inducing the release of mitochondrial DNA into the cytoplasm. This activates the stimulator of IFN genes (STING)/IFN regulatory factor 3/7 signalling pathway, promoting the upregulation of programmed death receptor-ligand 1 and other immune checkpoint molecules, thereby suppressing CD8+ T cell activation and function. This ultimately weakens the immune response to eliminate pathogens (66). ITA also inhibits the formation of neutrophil extracellular traps (NETs) (67). Excessive NET formation damages self-tissue and is associated with multiple types of autoimmune disease, such as systemic lupus erythematosus and rheumatoid arthritis (68). These findings suggest that ITA may be involved in autoimmune disorder. T helper (Th) 1 and Th17 cells are key drivers of autoimmunity (69). ITA suppresses Th1 and Th17 cell differentiation (70) while potentially promoting regulatory T cell production, thereby enhancing immune tolerance (57).
Antimicrobial activity of ITA
ITA has been demonstrated to inhibit proliferation of several bacterial species, including M. tuberculosis, Pseudomonas indigofera, Salmonella enterica, Yersinia pestis and Staphylococcus aureus (11,71-74). ICL is an essential enzyme for bacteria during infection (75). Studies indicate that ITA inhibits ICL activity by disrupting the glycolate cycle, which is key for bacterial survival within host cells (11,76). This leads to bacterial death because of insufficient energy and carbon sources (11,76). Beyond ICL, ITA also inhibits fructose-1,6-bisphosphate aldolase A (ALDOA) during glycolysis, impairing glycolytic flux and suppressing bacterial growth (56). Additionally, ITA suppresses guanosine monophosphate synthase B2 (GuaB2) enzyme activity in the purine biosynthesis pathway, further inhibiting bacterial growth (77).
In addition to directly inhibiting enzyme activities, ITA accumulation depletes other key substances that are required for bacterial growth. ITA is generated through the decarboxylation of cis-aconitic acid, a reaction that consumes the metabolic intermediates pyruvate and acetyl-CoA (50,51). Pyruvate and acetyl-CoA serve as cornerstones for maintaining energy supply and recycling metabolites in bacteria (78). Increased ITA synthesis competes with bacteria for these key metabolic resources, further enhancing its antibacterial effect. ITA disrupts key energy metabolism pathways in S. aureus, including the TCA cycle, glycolysis, pyruvate metabolism and arginine biosynthesis, by inducing transcriptomic and metabolomic changes associated with decreased energy metabolism, thereby slowing S. aureus growth (79). As an organic acid, ITA exhibits bactericidal activity when it is added at supraphysiological concentrations to cultures of Streptococcus faecalis, M. tuberculosis and Lactobacillus pneumophilus (11,62).
The autoinducer-2 (AI-2)/S-ribosylcysteine synthase (LuxS) regulatory system governs multiple key bacterial functions, including virulence expression, biofilm formation and motility (80). ITA decreases bacterial pathogenicity by inhibiting the AI-2/LuxS system and virulence-associated gene expression (81,82). Notably, ITA also disrupts bacterial membrane integrity and exacerbates oxidative damage, further demonstrating its antibacterial effects (81). ITA depletes the antioxidant glutathione (GSH) within bacteria; reduced GSH levels impair bacterial resistance to host-generated reactive oxygen species (ROS) attacks (83), increasing bacterial susceptibility to oxidative stress damage. Lysosomes serve as the primary organelles by which macrophages eliminate invading bacteria (84). As a lysosome inducer, ITA enhances the antimicrobial capacity of macrophages (85). Furthermore, ITA impairs the cytotoxicity of Plasmodium parasites (66), further demonstrating its antibacterial effects.
Antioxidant effects of ITA
Within the TCA cycle, SDH catalyses the conversion of succinate to fumarate; subsequent oxidation of succinate further promotes the production of mitochondrial ROS in vivo (86). Like succinate, ITA competitively inhibits mitochondrial SDH (87), thereby decreasing ROS generation at its source. SDH inhibition induces succinate accumulation and alters mitochondrial electron transport. This metabolic reprogramming decreases mitochondrial ROS production (85), resulting in antioxidant effects (87). ITA has been demonstrated to activate the Keap1/Nrf2 pathway (14). Nrf2 activation not only has anti-inflammatory effects but also upregulates the expression of a series of antioxidant genes (88), including HO-1, NADPH:oxidoreductase 1 and γ-glutamylcysteinyl ligase catalytic subunit (14). This enhances the cell antioxidant capacity, thereby mitigating oxidative stress-induced cell damage (Fig. 2). Additionally, ITA alleviates perfluorooctanoic acid-induced oxidative stress and intestinal injury by modulating the Keap1/Nrf2/HO-1 pathway and reshaping the gut microbiota (89). This provides a potential mechanistic explanation for the protective effects of ITA in gastrointestinal disease.
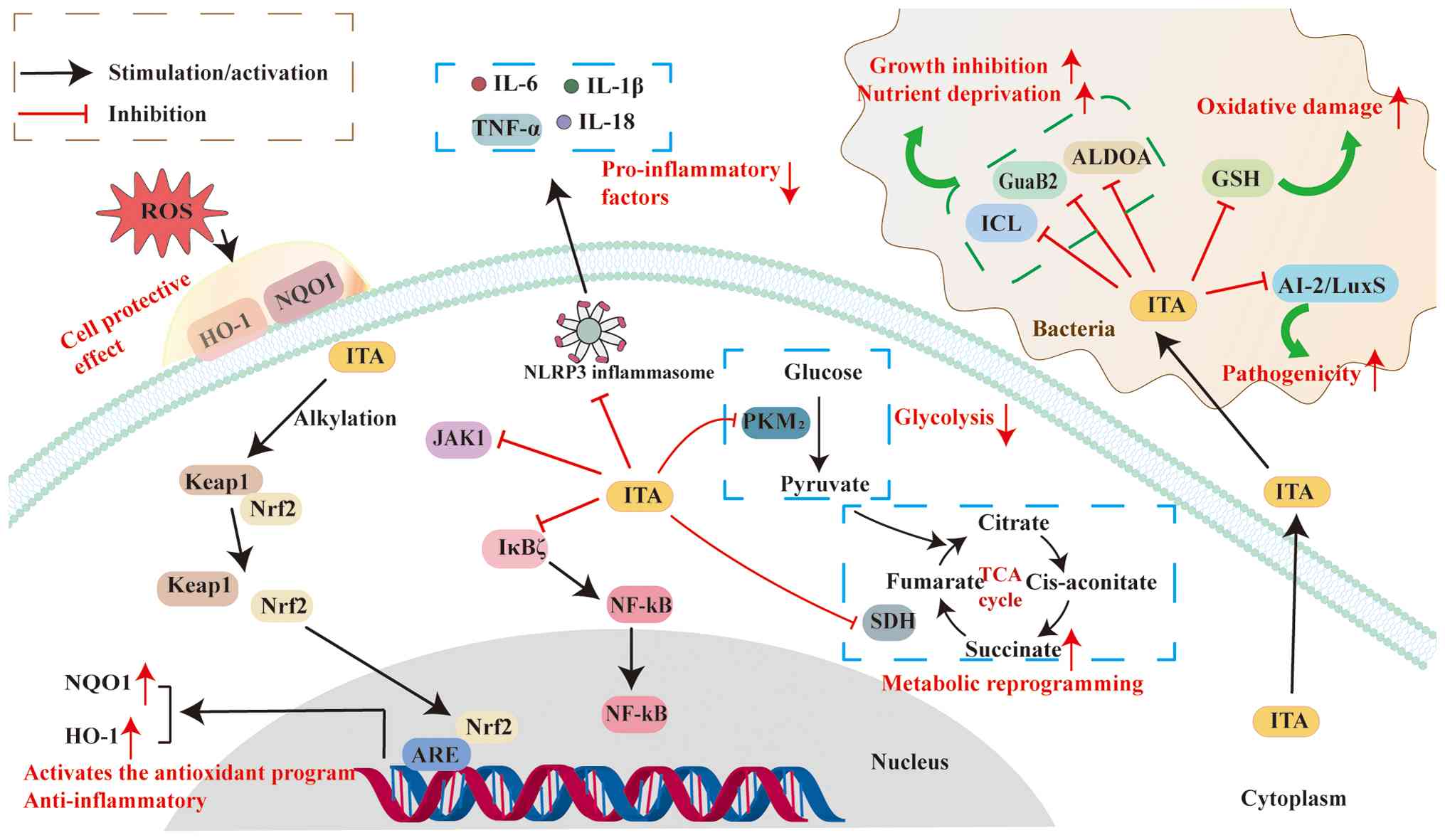 |
Figure 2
Multidimensional biological functions of ITA and its molecular mechanisms of action. ITA activates the Nrf2 pathway by alkylating Keap1, thereby driving the expression of antioxidant genes. ITA suppresses inflammation by inhibiting PKM2 and SDH to reprogramme metabolism, whilst also blocking NLRP3 and NF-κB. ITA exerts antimicrobial effects by inhibiting bacterial ICL, GuaB2, ALDOA and AI-2/LuxS. ITA, itaconate; ROS, reactive oxygen species; HO-1, haem oxygenase-1; NQO1, (NADPH): oxidoreductase 1; Keap1, kelch-like ECH-associated protein 1; ARE, anti-oxidative response element; TNF-α, tumour necrosis factor-α; NLRP3 NOD-like receptor thermal protein domain-associated protein 3; PKM2, pyruvate kinase M2; SDH, succinate dehydrogenase; ICL, isocitrate lyase; ALDOA, aldolase A; GSH, glutathione; AI-2/LuxS, autoinducer-2/S-ribosylcysteine synthase; GuaB2, guanosine monophosphate synthase B2; TCA, tricarboxylic acid.
|
Role of ITA in intestinal disease
Role of ITA in IBD
IBD is a chronic, non-specific disorder that is characterised by gastrointestinal inflammation, with Crohn's disease and ulcerative colitis (UC) representing its primary forms (90). Macrophages serve as key regulators of intestinal immune homeostasis, and their dysregulated polarisation constitutes a core mechanism in IBD pathogenesis (91). Within the healthy intestine, the lamina propria harbours abundant levels of M2 macrophages with anti-inflammatory and tissue-repair functions. These cells maintain immune tolerance by producing factors such as IL-10 and transforming growth factor-β (TGF-β; Table I) (92,93). However, during active IBD, the gut becomes saturated with M1-polarised macrophages that release tumour necrosis factor-α (TNF-α), IL-1β, IL-6 and nitric oxide, directly causing tissue damage (94).
 |
Table I
Effects of ITA and its derivatives on macrophage polarization.
|
Table I
Effects of ITA and its derivatives on macrophage polarization.
| Stimulus/polarization context |
Intervention |
Dose and timing |
Model system |
Polarization and mechanism |
(Refs.) |
| DSS-induced UC |
Exogenous ITA |
In vivo, 0.12 g/kg; in vitro, 200.00 μM; daily during |
DSS-induced UC mice |
Inhibition of M1 polarisation and TET2/STAT1 and TET2/NF-κB signaling |
(99) |
| Tumor microenvironment (CRC liver metastasis) |
Exogenous 4-OI/STING/IRG1 pathway activation |
In vivo, 50.00 mg/kg, i.p. for 3 weeks; in vitro, 150.00 μM, 4 h |
CRC liver metastasis mice; BMDM-tumor cell co-culture |
Promotion of M1 and inhibition of M2 polarisation; STING-IRG1-ITA axis activation drives TFEB nuclear translocation |
(113) |
| NEC model |
IRG1 knockout |
Not applicable |
NEC mice |
Promotion of M1 polarisation and glycolysis; inhibition of OXPHOS |
(129) |
| Exogenous 4-OI |
40.00 mg/kg/day i.p., 3 consecutive days |
|
Inhibition of M1 polarisation and glycolysis; promotion of OXPHOS |
|
| Pathogen infection (hvKP) |
Exogenous ITA |
In vivo: ITA, 25.00 mg/kg, i.p., twice daily; in vitro: ITA: 2.50 mM, 2 h |
Infected mice; peritoneal macrophages |
Inhibition of M1 polarisation and SYK activity through alkylation; suppressed inflammation |
(18) |
| Exogenous 4-OI |
In vivo: 4-OI, 12.50 mg/kg, i.p., twice daily; in vitro: 4-OI, 250.00 μM, 2 h |
|
|
|
| IL-4/IL-13 |
Exogenous 4-OI |
250.00 μM 4-OI pre-treatment for 2 h, followed by IL-4 (20.00 ng/ml) 24 h or IL-13 (20.00 ng/ml) 24 h |
Mouse BMDMs |
Inhibition of M2 polarisation and JAK1/STAT6 signaling |
(38) |
| LPS/IFN-β |
Exogenous 4-OI |
125.00 μM 4-OI pre-treatment for 3 h, followed by LPS (100.00 ng/ml, 24 h) or IFN-β (1,000.00 U/ml, 27 h) |
Mouse BMDMs |
Inhibition of M1 polarisation; Keap1 alkylation activates Nrf2 and suppresses IκBζ |
(14,65,66) |
Baseline concentration of ITA in normal mouse colonic tissue is ~0.15 μg/g, whereas in mice with diarrhoea-induced colitis, modelled by 3% dextran sulfate sodium (DSS), the ITA concentration in diseased colonic tissue range from 0.07 to 0.12 μg/g (95). When exogenous ITA (0.12 g/kg) is administered in this model, it significantly ameliorates pathological intestinal damage and decreases inflammatory infiltration (96). Similar results are obtained in an in vitro model using the murine macrophage RAW264.7 cells (96). Notably, the administered dose (0.12 g/kg) at this experimental concentration notably exceeds the endogenous ITA levels in the model animals (~0.15 μg/g in normal and 0.07-0.12 μg/g in UC model colon tissue) (95-97). Mechanistic analysis indicates that this ITA dose remodels macrophage phenotypes, inhibiting proinflammatory M1 polarisation while promoting reparative M2 conversion (95). This decreases the release of proinflammatory cytokines such as TNF-α and IL-6, thereby alleviating colitis (95) (Fig. 3).
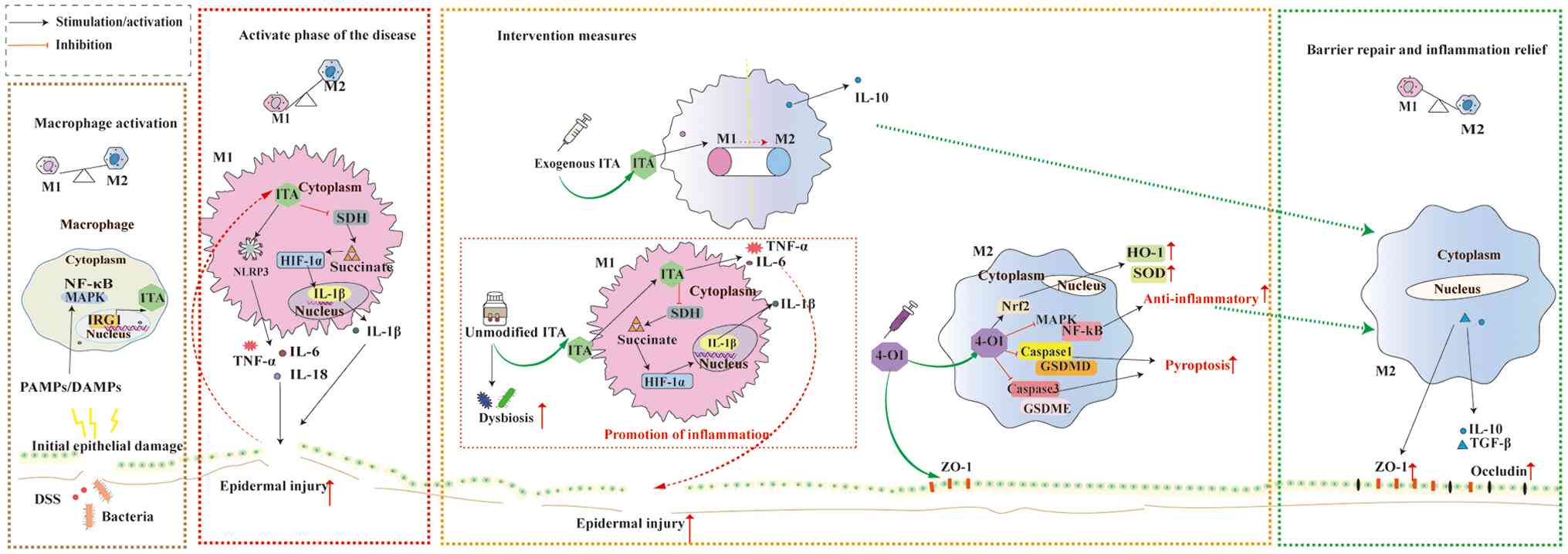 |
Figure 3
Schematic of the dynamic role of ITA in the progression and repair of inflammatory bowel disease. Following stimulation by injurious factors such as DSS or bacteria, the epithelial barrier integrity is disrupted, leading to the release of PAMPs/DAMPs and activation of the MAPK/NF-κB signaling pathway. Activated macrophages upregulate IRG1 expression, which drives the production of ITA. Macrophages polarise toward the pro-inflammatory M1 phenotype and release cytokines, including TNF-α, IL-1β, IL-6 and IL-18, thereby exacerbating tissue damage. Intraperitoneal administration of exogenous ITA promotes the transition of macrophages from the M1 to the M2 phenotype, leading to IL-10 release and supporting tissue repair. Gastric lavage with unmodified exogenous ITA sustains an M1-dominant macrophage response. This activates the ITA/SDH/HIF-α/IL-1β pathway, elevating IL-1β secretion. ITA stimulates the release of TNF-α and IL-6, aggravating tissue injury, and may disrupt gut microbiota balance. Intraperitoneal injection of exogenous 4-OI activates the Nrf2 antioxidant pathway while inhibiting pro-inflammatory signals such as NF-κB. This reprograms macrophages toward the M2 phenotype and suppresses pyroptosis. Following effective intervention with 4-OI derivatives, pro-inflammatory responses are suppressed and M2 macrophages become predominant. Anti-inflammatory and reparative mechanisms are enhanced, characterized by increased expression of intestinal tight junction proteins. Epithelial barrier function is restored, inflammation is alleviated and tissue homeostasis is reestablished. ITA, itaconate; PAMP, pathogen-associated molecular pattern; DAMP, damage-associated molecular pattern; DSS, dextran sulphate sodium; NLRP3, NOD-like receptor thermal protein domain-associated protein 3; SDH, succinate dehydrogenase; HIF-1α, hypoxia-inducible factor-1α; ROS, reactive oxygen species; HO-1, haem oxygenase-1; ZO-1, zonula occludens-1; SOD, superoxide dismutase; 4-OI, 4-octyl itaconate; TGF, transforming growth factor.
|
In UC induced by DSS, compared with wild-type mice, IRG1−/− mice exhibit more severe clinical symptoms (such as weight loss, diarrhoea and bloody stool) and more pronounced histological damage, as well as elevated levels of proinflammatory cytokines in colonic tissue (96). Mechanistically, decreased ITA due to IRG1 deficiency activates the NF-κB/MAPK signalling pathway and promotes GSDMD/GSDME-mediated pyroptosis, thereby exacerbating colonic inflammation (17). By contrast, the ITA derivative 4-OI has potent anti-inflammatory effects (12). In IRG1-deficient mice, 4-OI (25 mg/kg/day, intravenous injection for 7 days) effectively alleviates colitis symptoms (96). Mechanistically, 4-OI mitigates inflammation by increasing Nrf2 expression, suppressing ROS production and inhibiting MAPK/NF-κB signalling in conjunction with caspase1/GSDMD- and caspase3/GSDME-mediated pyroptosis (17). Furthermore, 4-OI potentiates the therapeutic effects of mesalazine through mechanisms involving the activation of the Keap1-Nrf2 pathway, upregulation of antioxidant enzyme expression, alleviation of oxidative stress and apoptosis and enhancement of intestinal barrier function by increasing tight junction protein expression, ultimately ameliorating UC (98). However, another study demonstrated that oral administration of unmodified ITA to mice exacerbates DSS-induced UC symptoms and upregulates the mRNA expression of proinflammatory factors, such as TNF-α, IL-1β and IL-6, in macrophages (99). These findings reveal the proinflammatory and disease-aggravating effects of ITA in UC (98). This contrasts with the anti-inflammatory action of 4-OI observed in the aforementioned study (12). Reports indicate that ITA, which is highly hydrophilic and weakly electrophilic, struggles to activate the KEAP1-NRF2 antioxidant pathway, fails to suppress oxidative stress and results in poor mucosal permeability (64,99). Under specific conditions (such as prolonged pretreatment), it may enhance proinflammatory signalling (99). 4-OI, enhanced by its octyl side chain, increases hydrophobicity and electrophilicity, facilitating penetration of damaged mucosa while efficiently activating antioxidant pathways (100). ITA is directly phagocytosed and accumulated by macrophages, where it inhibits SDH intracellularly, leading to succinate accumulation. This promotes the expression of inflammatory mediators such as IL-1β by stabilising HIF-1α (99,100). Conversely, 4-OI targets mitochondrial metabolism and the NF-κB pathway for inhibition, thereby downregulating proinflammatory factors (101,102). Furthermore, ITA significantly enhances LPS-induced IFN-β secretion by macrophages (100), whereas IFN-β exerts proinflammatory effects in UC (103). Concurrently, oral ITA exacerbates dysbiosis (104), intensifying intestinal inflammation. Conversely, 4-OI remodels the microbiota structure by promoting beneficial bacterial proliferation and inhibiting proinflammatory metabolite production (98). Moreover, the local pH environment has indirect effects. While inflammation-induced intestinal pH reduction does not impair ITA uptake (100,105), it alters its ionisation state to increase the metabolic activation of the microbiota (106). Conversely, 4-OI maintains stable activity across a broad range of pH values (100). The diverse effects of ITA and its derivatives on UC stem not only from chemical structural differences between the parent compound and its derivatives but also from the complex interplay between their intrinsic properties and the pathological microenvironment. This highlights the complexity of their therapeutic application, necessitating further experimental studies to elucidate these mechanisms.
Role of ITA in CRC
CRC ranks as the third most common cancer globally, with an estimated 1.9 million new cases and 935,000 deaths in 2020 (107). Despite an overall decline in incidence, CRC mortality rates remain high, with a 5-year survival rate <15% for metastatic disease (107-109). Increasing evidence indicates that CRC is associated with chronic inflammation, with inflammatory cytokines produced by cancer cells or within the tumour microenvironment (TME) playing a notable role in CRC progression (110,111). Attenuating inflammatory responses is an effective therapeutic strategy to prevent CRC progression (112). Tumour-associated macrophages serve as key mediators of inflammatory signalling within the TME and serve a crucial role in CRC progression (111). OI acid ester, a cell-permeable ITA derivative, undergoes esterase hydrolysis in vivo to release ITA (14). An in vivo study demonstrated that 3 consecutive weeks of OI treatment in mice with CRC liver metastases significantly decreases tumour burden in the liver while markedly alleviating hepatic injury (113). Mechanistic analysis has revealed that ITA inhibits TME polarisation towards M2-phenotype macrophages, thereby suppressing tumour progression (113). The aforementioned study revealed that in coculture experiments, STING protein agonists inhibit CRC cell migration and invasion via the IRG1/ITA pathway in macrophages. Conversely, direct treatment of macrophages with OI (regardless of IRG1 knockdown status) effectively suppresses the migration and invasion of cocultured tumour cells (113). Subsequent analysis demonstrated that STING activation induces IRG1 upregulation, increases ITA expression and facilitates nuclear translocation of TFEB. This stimulates lysosomal biosynthesis and enhances macrophage invasiveness, thereby bolstering the pathogen clearance capacity. Concurrently, it suppresses macrophage polarisation towards the M2 phenotype while promoting M1 macrophage polarisation, further inhibiting CRC migration and invasion (113,114). Administration of DI to mice decreases the risk of colitis-associated CRC (27). Mechanistic analysis has revealed that DI suppresses the secretion of the cytokines IL-1β and CCL2 by intestinal epithelial cells, thereby decreasing macrophage recruitment to the TME (27). The aforementioned studies indicate that ITA, as a key signalling mediator in immunometabolic regulation, inhibits CRC metastasis by reprogramming tumour-associated macrophage function and enhancing the tumour-killing capacity. However, existing studies have not explicitly reported the direct effects of altered ITA levels on CRC (27,113). Previous research has positioned it as a key signalling mediator in immunometabolic regulation, indirectly influencing tumour progression through functions such as macrophage reprogramming (113,114). Therefore, elucidating the direct effects of ITA on CRC cells represents an important future research direction.
Chemotherapy is a key therapeutic modality following surgery for advanced CRC, with sensitivity to chemotherapeutic agents serving as a primary determinant of treatment efficacy (115). Nrf2 modulates multiple antioxidant and detoxification pathways, thereby mitigating cell damage induced by chemotherapeutic drugs (116). 4-OI is an Nrf2 activator (117). Huang et al (19) demonstrated that treating HCT-116 and LOVO cells with 200 μM 4-OI in combination with oxaliplatin (30 μM) or lobaplatin (16 μg/ml) for 48 h significantly attenuates the cytotoxic effects of these chemotherapeutic agents, resulting in enhanced CRC cell survival. Mechanistic analysis has revealed that under these conditions, 4-OI effectively decreases chemotherapy-induced ROS levels by activating the Nrf2 pathway, thereby inhibiting apoptosis and ferroptosis and protecting tumour cells (19,116). Yang et al (118) reported that treatment of HCT116 and LOVO cells with 4-OI at 100, 200 or 300 μM alone does not affect cell viability. However, when 200 or 300 μM 4-OI is combined with copper death inducers such as elesclomol-Cu, cell death is promoted. The higher the 4-OI concentration, the stronger the sensitising effect on copper-induced death, with the 300 μM 4-OI + elesclomol-Cu group exhibiting significantly higher cell death rate than the 200 μM combination group. This effect has been validated in vivo: In a nude mouse xenograft model, the tumour suppression efficacy of the combined treatment with elesclomol-Cu (10 mg/kg) and 4-OI (50 mg/kg) surpasses that of elesclomol-Cu monotherapy; mechanistic analysis has revealed that 4-OI targets and inhibits the expression of GAPDH, a key enzyme in glycolysis, thereby blocking the energy supply of tumour cells (19,118). This sensitises cells to copper-based death inducers (such as elesclomol-Cu), directly inducing metabolic crisis and ultimately inhibiting tumour growth (118).
The aforementioned studies indicate that the effects of 4-OI (protective and inhibitory) are dependent on its microenvironment (Fig. 4). When 4-OI is co-administered with chemotherapeutic agents (30 μM oxaliplatin/16 μg/ml lobaplatin), the Nrf2-mediated cell protection mechanism is predominant only at concentrations ≥200 μM and after 48 h coculture (19). In the absence of chemotherapy intervention, 4-OI alone (100-300 μM) has no significant tumour-suppressive or proliferative effects (118). Its copper death-sensitising effect, mediated by GAPDH inhibition, is pronounced only at concentrations ≥200 μM in combination with copper death inducers such as elesclomol-Cu, with greater inhibition at 300 μM (118). In vivo, nude mouse xenograft model of CRC has demonstrated that coadministration of 4-OI (50 mg/kg) with elesclomol-Cu (10 mg/kg) effectively exerts tumour-suppressive effects (118). These seemingly contradictory phenomena collectively establish a framework of effect dominance: The ultimate outcome of 4-OI depends on the presence or absence of chemotherapeutic agents/causes of copper death. A distinct concentration threshold (200 μM) serves as the critical transition point from no discernible effect to either protective or inhibitory effect, with the specific direction determined by the type of co-administered drug (19,118).
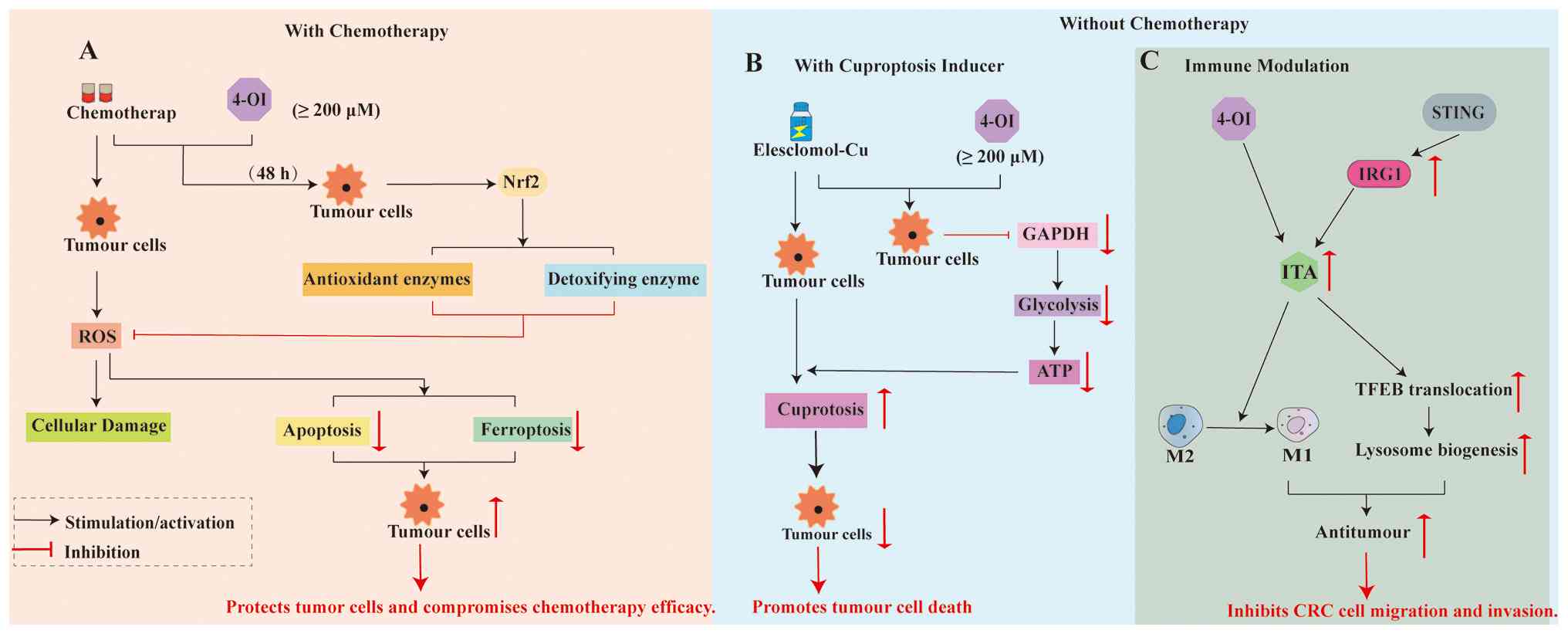 |
Figure 4
Dual roles of ITA and its derivatives in CRC. (A) When combined with conventional chemotherapeutic agents such as oxaliplatin, 4-OI at concentrations ≥200 μM activates the Nrf2 pathway, enhancing cell antioxidant and detoxification capacity. This leads to the clearance of chemotherapy-induced ROS and suppresses apoptosis and ferroptosis, protecting tumour cells and reducing chemotherapy efficacy. (B) In combination with copper-death inducers (elesclomol-Cu), 4-OI ≥200 μM targets the glycolytic enzyme GAPDH, triggering an energy supply crisis in tumour cells. This metabolic disruption sensitises cells to copper-death, resulting in enhanced tumour-suppressive effects. (C) Immunomodulatory role in the tumour microenvironment. STING agonists can upregulate endogenous ITA via IRG1. Both exogenous 4-OI and endogenous ITA promote TFEB nuclear translocation, which enhances lysosomal biogenesis and function. ITA reprograms tumour-associated macrophages by inhibiting M2-type (pro-tumour) polarisation and promoting M1-type (anti-tumour) polarisation, thereby strengthening anti-tumour immunity and suppressing CRC cell migration and invasion. ITA, itaconate; 4-OI, 4-octyl itaconate; ROS, reactive oxygen species; TFEB, transcription factor EB; CRC, colorectal cancer; IRG, immune response gene.
|
Early-onset CRC (EOCRC) is defined as CRC diagnosed in individuals aged <50 years (119). Its pathogenesis may be associated with abnormal activation of the ITA pathway. High expressions of genes associated with the NOTCH4/GATA4/IRG1 signalling pathway are associated with reduced overall survival in patients (120). ITA is a metabolite with immunomodulatory functions and serves as a key effector molecule within this pathway. Consequently, targeting ITA or its signalling pathways may offer a unique therapeutic strategy for developing novel immunotherapies for patients with EOCRC. To the best of our knowledge, however, no experimental studies have elucidated the mechanism of action of ITA in EOCRC, necessitating further investigation.
Role of ITA in necrotising enterocolitis (NEC)
NEC is the most lethal gastrointestinal disorder among preterm infants (121,122), affecting 5-10% of very low birth weight infants with a mortality rate of 20-30% among those requiring surgical intervention (123,124). Despite decades of progress in neonatal intensive care that have improved outcomes for other complications, NEC incidence has remained high: Globally, 7 out of every 100 ELBW infants admitted to neonatal units develop the disease (122,125). The pathogenesis of NEC remains unclear, and treatment encompasses bowel rest, antibiotics, supportive care and surgical intervention (126,127). Consequently, there is need to identify novel therapeutic approaches for NEC. Research has revealed an immune system association, with dysregulated inflammation serving a key role in NEC onset and progression (128). Huangfu et al (129) revealed elevated IRG1 expression in the intestinal tissue of both patients with NEC and mice relative to normal controls. Metabolite analysis has revealed significantly lower ITA levels (~11 ng/ml) in the peripheral blood of patients with NEC compared with healthy controls (~18 ng/ml), with ITA levels negatively associated with NEC severity. Further investigations have revealed that in NEC models, compared with control mice, IRG1 knockout mice exhibit more severe intestinal epithelial damage, higher mortality rate, elevated proinflammatory cytokine levels (IL-6, IL-1β and TNF-α) and increased ROS levels (129,130). Intraperitoneal injection of 4-OI (40 mg/kg/day for 3 consecutive days) reverses these phenotypes. Mechanistic analysis has indicated that ITA deficiency due to IRG1 knockout exacerbates NEC, whereas 4-OI supplementation reverses this process, confirming the critical protective role of the ITA pathway in regulating intestinal inflammation (131). The aforementioned study demonstrated that ITA suppresses neonatal intestinal inflammation via metabolic reprogramming of M1 macrophages, suggesting ITA may represent a potential therapeutic target for NEC.
Role of ITA in intestinal infection
The gut serves as the primary gateway through which numerous pathogens invade the body (132). Macrophages recognise, phagocytose and digest invading pathogens, forming the cornerstone of infection control (133). Within the complex immune environment of intestinal infection, ITA, as an immune metabolite that is predominantly produced by macrophages, serves an indispensable role (134). When the intestinal mucosa encounters pathogens such as Salmonella or Shigella, macrophages rapidly initiate metabolic reprogramming to increase antimicrobial activities by recognising pathogen-associated molecular patterns via pattern recognition receptors, including toll-like receptors (89,135). This process is centrally driven by notable upregulation of IRG1-encoded proteins (11). ITA has potent antibacterial effects, directly inhibiting bacterial growth and proliferation in the gut (11,76). Beyond direct pathogen suppression, ITA also exerts protective effects against intestinal infection by modulating the host immune response. Through its inherent immunomodulatory and antioxidant mechanisms (15), ITA maintains immune homeostasis and safeguards the intestinal barrier during infection (89).
Intestinal infection disrupt the gut microbiota (136). ITA may regulate the equilibrium of the intestinal microenvironment (137). Compared with control mice, IRG−/− mice exhibit significantly increased levels of the order Bacillales (137). ITA supplementation increases the abundances of beneficial gut bacteria, promoting postinfection recovery of the microbiota towards a healthy state (18). ITA also enhances the host defence against Vibrio cholerae by impairing its ability to metabolise fatty acids in the gut, thereby mitigating intestinal injury (129). Li et al (18) demonstrated that intravenous administration of ITA (25 mg/kg) or 4-OI (12.5 mg/kg) in a mouse model of infection with highly virulent Klebsiella pneumoniae (hvKP) mitigates intestinal injury, restores impaired intestinal barrier function and alleviates induced dysbiosis. Mechanistic analysis has revealed that ITA inhibits spleen tyrosine kinase (SYK) activation by alkylating SYK proteins, thereby suppressing the production of inflammatory mediators such as IL-6, IL-1β and TNF-α (18). ITA inhibits macrophage activation towards the M1 phenotype while suppressing macrophage death, thereby curbing inflammation induced by dysbiosis (18) and mitigating intestinal injury. Moreover, treatment with ITA and 4-OI prevents hvKP-induced depletion of zonula occludens-1 and occludin-1 proteins in the intestine, preserving epithelial barrier integrity. These findings contradict those of Runtsch et al (38), who reported that ITA and its derivatives effectively inhibit M2 macrophage polarisation following stimulation with the classical activator IL-4. Notably, Li et al (18) directly stimulated macrophages with the hvKP pathogen and reported that ITA and its derivatives suppress M1 polarisation while promoting M2 polarisation. This discrepancy may stem from differences in experimental design and stimulus type.
During intestinal infections, ITA produced by macrophages directly suppresses pathogen metabolism to inhibit infection. It also maintains intestinal barrier integrity and immune homeostasis by activating antioxidant pathways and suppressing excessive inflammation, thereby exerting protective effects on the gut (15,18,38,89).
Clinical prospects and challenges of ITA derivatives
Despite serving a pivotal role in immunometabolism, itaconic acid salts face limitations in direct clinical application because of their inherent chemical instability and poor cell membrane permeability (12). Consequently, researchers have synthesised numerous itaconic acid salt derivatives, with the most investigations focusing on 4-OI (12) and DI (27). Studies indicate that 4-OI is converted to ITA intracellularly via esterases, whereas DI does not undergo methylation to ITA (136,137). Both compounds demonstrate therapeutic efficacy in preclinical models of intestinal disease (14,98,138). In a DSS-induced UC mouse model, intraperitoneal administration of 4-OI (50-100 mg/kg/day for 7 consecutive days) significantly decreases intestinal mucosal inflammatory infiltration and ulcer formation in a dose-dependent manner (98). This effect is associated with the activation of the KEAP1/NRF2 pathway and the upregulation of cell-protective proteins (98). Similarly, intraperitoneal administration of DI (40 mg/kg/day, initiated 3 days prior to DSS exposure and continued for 11 days) exerts comparable intestinal protective effects via Nrf2 pathway activation (138). The key mechanism of these derivatives is the enhanced electrophilicity of both 4-OI and DI compared with natural ITA. This enables efficient alkylation modification of the cysteine residues in key signalling proteins such as KEAP1 (137) and SYK (18), thereby mimicking and amplifying the biological effects of natural ITA, including anti-inflammatory and antioxidant actions (83).
However, the translation of experimental findings into clinical applications is constrained by translational challenges, with species differences constituting the primary translational gap. Current research predominantly relies on animal models (113,118,129). While both human and mouse macrophages upregulate IRG1 expression in response to inflammatory stimuli, the intensity and dynamics of this response differ (11,35,39). For example, LPS-stimulated itaconic acid is present at millimolar concentrations in mouse macrophages, with LPS-stimulated mouse macrophage itaconic acid concentrations two orders of magnitude higher than those in human macrophages (8 mM vs. 60 μM) (11). These findings suggest potential differences in the intensity of the IRG1-mediated immunometabolic responses between mice and humans. Whether human macrophages respond to itaconic acid and its derivatives in a manner consistent with that observed in mice remains unknown. This prevents direct extrapolation of the effective doses from animal studies to humans. To better predict human responses, future preclinical research should employ more predictive systems, such as humanised mouse models or patient-derived organoids, to evaluate the therapeutic potential and elucidate the underlying mechanisms. Data derived from these models, which more closely approximate human physiology, may more reliable foundations for clinical trial dose design and protocol development.
In addition to species differences, key data on human pharmacokinetics and safety are generally lacking, constituting a core obstacle to clinical translation. Reports indicate that DI is converted intracellularly into a mixture of 1- and 4-methyl ITA (MI) (83,136). To address the poor permeability of ITA and 4-MI, Lee et al (48) designed prodrug-based derivatives P2 and P13. Oral administration of prodrug P13 (100 mg/kg 4-MI equivalent) in mice results in rapid hydrolysis, yielding peak plasma concentrations of the active molecule 4-MI of 349 μM, with a half-life of 0.7-0.9 h, maintaining effective concentrations in the target skin tissue. Similarly, oral administration of prodrug P2 (100 mg/kg ITA equivalent) results in peak plasma concentrations of the active molecule ITA of 83.8 μM, with a half-life of 1.1-1.7 h. By contrast, the half-life in skin is 3.32±1.23 h, indicating prolonged retention in the target skin tissue. These data corroborate the rapid clearance of exogenous ITA in vivo, which is consistent with findings by Willenbockel et al (49): In a rat intravenous infusion model (15 mg/kg/min ITA), the half-life of ITA ranges from 53 to 85 min, whereas in a mouse model, a single high-dose (400 mg/kg ITA) yields a half-life of 10.9 min, demonstrating notable species differences. Elimination primarily occurs via renal excretion, while the compound may also be taken up and metabolised by tissues such as the liver and kidney. Although animal models provide key insights, these data cannot be directly extrapolated to humans. In existing studies, human pharmacokinetic data regarding the absorption, distribution, metabolism and excretion characteristics of 4-OI and DI, including half-life, volume of distribution, clearance and bioavailability across different routes of administration (particularly oral), remain unknown (24,100). Current evidence supporting the efficacy of 4-OI and DI in intestinal disease models relies primarily on intraperitoneal injection (96,129). While this route ensures rapid systemic entry and onset of action, it differs from the conventional oral administration pathway that is used in clinical intestinal disease treatment. Furthermore, the oral bioavailability and stability of these compounds within the human gastrointestinal tract remain unclear, constituting a key bottleneck for their translation into clinical applications. Although prodrug strategies have improved the oral stability, the water solubility and chemical stability of the active molecule remain fundamental constraints in formulation development (48). Research has indicated that compared with DI alone, DI-loaded liposomes containing dodecyl isovalerate as an activator demonstrate superior efficacy in treating acute liver failure (139). This suggests that optimising the pharmaceutical formulation process for relevant derivatives represents a crucial direction for advancing clinical translation. Enhancing delivery systems and improving the bioavailability of such compounds are central research priorities in this field.
ITA originates from the immune responses and may exhibit low toxicity, providing a potential basis for its clinical application (24). A preliminary in vitro toxicity study indicated that neither ITA nor DI significantly affects macrophage viability at a concentration of 10 mM (98). Further a short-term animal study reveals no marked pathological alterations in liver or kidney tissue following intraperitoneal administration of 4-OI (40 mg/kg/day for 3 consecutive days) in mice (139). However, these data are insufficient to define organ-specific toxicity, maximum tolerated dose or risk of interactions with other drugs during long-term administration (12,140,141). Given the extensive involvement of the ITA pathway in cell metabolism, the risk of interactions between its derivatives and other drugs, particularly those utilising similar metabolic pathways or affecting immune function, warrants urgent evaluation. Coadministration of 4-OI significantly enhances the therapeutic efficacy of mesalazine in UC (96), offering a novel option for patients who are resistant to conventional therapies. Future research should focus on elucidating the regulatory role of 4-OI in the gut microbiota-immune axis and exploring synergistic effects when combined with traditional drugs or biological agents, thereby pioneering new avenues for precision treatment of IBD.
The ITA pathway constitutes a natural immunosuppressive mechanism. Prolonged or inappropriate exogenous enhancement of this pathway may, under certain conditions, such as during secondary bacterial infection following influenza or during chronic M. tuberculosis infection (142,143), compromise the ability to clear pathogens. While this risk may be outweighed by benefits in conditions of excessive inflammation, persistent ITA administration-induced systemic immunosuppression may weaken immune surveillance, increasing susceptibility to opportunistic infections (such as cytomegalovirus reactivation) or potentially malignant tumours (144). ITA serves as a potent metabolic modulator by regulating immune cell metabolism through the inhibition of the activity of key glycolytic enzymes (57). Short-term 4-OI intervention (50 mg/kg for 12 weeks) has demonstrated benefits in metabolic disease models, including high-fat diet-induced obese mouse models, by improving insulin resistance (145,146). However, prolonged systemic administration may exceed physiological compensatory limits, potentially disrupting systemic energy homeostasis. Whether the sustained inhibition of glycolysis in immune and potentially other cell types affects normal immune surveillance, tissue repair and other processes that require a rapid energy supply remains unexplored, as do its long-term effects on metabolic organs such as the liver and muscle. Although most research focuses on short-term therapeutic effects (14,95,98,139,145,146), potential long-term metabolic maladaptation or energy imbalance necessitates systematic evaluation as a key safety concern in prospective preclinical toxicology studies.
To the best of our knowledge, no phase I/II clinical trials of 4-OI or DI for treating intestinal disorders have been registered on publicly accessible clinical trial platforms. Consequently, future research must fill the gaps in human pharmacokinetic and safety data, developing stable formulations suitable for clinical administration and cautiously designing human trials on the basis of more comprehensive preclinical toxicology data to assess their clinical applicability.
Conclusion
ITA and its derivatives target immunometabolic pathways, offering novel therapeutic approaches for intestinal disorders. However, translating these promising experimental findings into clinically viable therapies requires systematically addressing the critical knowledge gaps and methodological limitations in current research. Future studies should prioritise establishing comprehensive human pharmacology and safety profiles, adopting more predictive preclinical experimental systems, and developing minimum standards for experimental reporting.
Establishing comprehensive human pharmacology and safety profiles is a key step in bridging the translational gap. The most pressing requirement is obtaining systemic pharmacokinetic data for the primary ITA derivatives, such as 4-OI and DI, in humans, encompassing oral bioavailability, elimination half-life, metabolic pathways and tissue distribution characteristics. Equally key is conducting long-term toxicology studies that are compliant with drug development standards and extend beyond short-term cell viability and organ pathology observations. These studies must specifically evaluate the risk of immunosuppression-associated infections under chronic dosing conditions and potential systemic energy homeostasis imbalances arising from its extensive metabolic regulatory properties.
As preclinical research relies primarily on animal models and notable interspecies variations exist in ITA production, adopting more predictive experimental systems is key. In mechanistic studies, humanised immune system models, patient-derived organoids or primary immune cells are prioritised to validate targets and define dose-response relationships in settings closer to human physiology. To address the high context dependency of ITA effects, experimental designs must precisely control and standardise the reporting of key microenvironmental variables, such as coexisting cell types, local cytokine profiles, redox states and specific conditions of co-administered medication.
To facilitate the comparison, integration and ultimate translation of disparate research findings, minimum standards for experimental reporting are required. This must encompass several core elements: Explicit chemical structure and purity of the ITA derivative; precise concentrations, treatment duration and solvent information for in vitro experiments; detailed administration protocols for in vivo studies (including route, dosage, formulation and conversion basis); specific strains; sex, age and activation status of the animal or cell models employed and detection results for key biological markers such as IRG1 expression and endogenous ITA concentrations.
In summary, advancing ITA research to its next phase requires shifting focus from describing its complex biological effects towards systematic, engineering-driven approaches aimed at clinical translation. By collectively addressing these knowledge gaps and adopting standardised practices, ITA may be transformed from a potent endogenous metabolite into a reliable therapeutic agent for the precise treatment of intestinal disorder.
Availability of data and materials
Not applicable.
Authors' contributions
XL wrote the manuscript. QL, JL, LZ, SY, LT, BY and YW performed the literature review. GW and JA designed the study and constructed figures. HJ and BT edited the manuscript. All authors have read and approved the final manuscript. Data authentication is not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Natural Science Foundation of China (grant nos. 81960507, 82073087 and 82160112), the Collaborative Innovation Center of the Chinese Ministry of Education (grant no. 2020-39), the Science and Technology Bureau fund of Zunyi City [grant no. ZUN SHI KE HE HZ ZI (2019) 93-Hao], the Science and Technology Plan Project of Guizhou Province [grant no. QIAN KE HE JI CHU-ZK (2024)YI BAN 323] and Medical Research Union Fund for High-quality health development of Guizhou Province (grant no. 2024GZYXKYJJXM0019).
References
|
1
|
Wang Y, Huang Y, Chase RC, Li T, Ramai D, Li S, Huang X, Antwi SO, Keaveny AP and Pang M: Global burden of digestive diseases: A systematic analysis of the global burden of diseases study, 1990 to 2019. Gastroenterology. 165:773–783.e15. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen HY, Ge P, Liu JY, Qu JL, Bao F, Xu CM, Chen HL, Shang D and Zhang GX: Artificial intelligence: Emerging player in the diagnosis and treatment of digestive disease. World J Gastroenterol. 28:2152–2162. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Edgington-Mitchell LE: Pathophysiological roles of proteases in gastrointestinal disease. Am J Physiol Gastrointest Liver Physiol. 310:G234–G239. 2016. View Article : Google Scholar
|
|
4
|
Shinomura Y: Preface. Recent advances in the diagnosis and treatment of gastrointestinal diseases. Digestion. 91:5–6. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Peery AF, Crockett SD, Murphy CC, Lund JL, Dellon ES, Williams JL, Jensen ET, Shaheen NJ, Barritt AS, Lieber SR, et al: Burden and cost of gastrointestinal, liver, and pancreatic diseases in the United States: Update 2018. Gastroenterology. 156:254–272.e11. 2019. View Article : Google Scholar
|
|
6
|
Ivány E, Farkas B, Bacsur P, Rutka M, Gálfalvi N, Farkas K, Szepes Z and Molnár T: A narrative review on the frequency, pathophysiology and management of bowel urgency associated with ulcerative colitis. Best Pract Res Clin Gastroenterol. 78:1020612025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Singh M, Morris VK, Bandey IN, Hong DS and Kopetz S: Advancements in combining targeted therapy and immunotherapy for colorectal cancer. Trends Cancer. 10:598–609. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Reichert M, Willis F, Post S, Schneider M, Vilz T, Willis M and Hecker A: Pharmacologic prevention and therapy of postoperative paralytic ileus after gastrointestinal cancer surgery: Systematic review and meta-analysis. Int J Surg. 110:4329–4341. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Strelko CL, Lu W, Dufort FJ, Seyfried TN, Chiles TC, Rabinowitz JD and Roberts MF: Itaconic acid is a mammalian metabolite induced during macrophage activation. J Am Chem Soc. 133:16386–16389. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shi J and Cai C: Research progress on the mechanism of itaconate regulating macrophage immunometabolism. Front Immunol. 13:9372472022. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Michelucci A, Cordes T, Ghelfi J, Pailot A, Reiling N, Goldmann O, Binz T, Wegner A, Tallam A, Rausell A, et al: Immune-responsive gene 1 protein links metabolism to immunity by catalyzing itaconic acid production. Proc Natl Acad Sci USA. 110:7820–7825. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Peace CG and O'Neill LA: The role of itaconate in host defense and inflammation. J Clin Invest. 132:e1485482022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zasłona Z and O'Neill LAJ: Cytokine-like roles for metabolites in immunity. Mol Cell. 78:814–823. 2020. View Article : Google Scholar
|
|
14
|
Mills EL, Ryan DG, Prag HA, Dikovskaya D, Menon D, Zaslona Z, Jedrychowski MP, Costa ASH, Higgins M, Hams E, et al: Itaconate is an anti-inflammatory metabolite that activates Nrf2 via alkylation of KEAP1. Nature. 556:113–117. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hooftman A, Angiari S, Hester S, Corcoran SE, Runtsch MC, Ling C, Ruzek MC, Slivka PF, McGettrick AF, Banahan K, et al: The immunomodulatory metabolite itaconate modifies NLRP3 and inhibits inflammasome activation. Cell Metab. 32:468–478.e7. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Farré R, Fiorani M, Abdu Rahiman S and Matteoli G: Intestinal permeability, inflammation and the role of nutrients. Nutrients. 12:11852020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang W, Wang Y, Wang T, Li C, Shi L, Zhang P, Yin Y, Tao K and Li R: Protective effects of IRG1/itaconate on acute colitis through the inhibition of gasdermins-mediated pyroptosis and inflammation response. Genes Dis. 10:1552–1563. 2022. View Article : Google Scholar
|
|
18
|
Li Y, Xu Y, Li W, Li J, Wu W, Kang J, Jiang H, Liu P, Liu J, Gong W, et al: Itaconate inhibits SYK through alkylation and suppresses inflammation against hvKP induced intestinal dysbiosis. Cell Mol Life Sci. 80:3372023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Huang Y, Yang W, Yang L, Wang T, Li C, Yu J, Zhang P, Yin Y, Li R and Tao K: Nrf2 inhibition increases sensitivity to chemotherapy of colorectal cancer by promoting ferroptosis and pyroptosis. Sci Rep. 13:143592023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Baup S: Ueber eine neue Pyrocitronsäure, und über Zusammensetzung der Citronensäure [On a new pyrocitric acid, and on the composition of citric acid]. Ann Pharmacie. 19:29–38. 1836.In German. View Article : Google Scholar
|
|
21
|
Kinoshita K: Über eine neue Aspergillus-Art, Asp. itaconicus nov. spec. [About a new Aspergillus species, Asp. itaconicus nov. spec.]. Shokubutsugaku Zasshi (Botanical Magazine Tokyo). 45:45–61. 1931.In German, Japanese. View Article : Google Scholar
|
|
22
|
Willke T and Vorlop KD: Biotechnological production of itaconic acid. Appl Microbiol Biotechnol. 56:289–295. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shin JH, Yang JY, Jeon BY, Yoon YJ, Cho SN, Kang YH, Ryu DH and Hwang GS: 1H NMR-based metabolomic profiling in mice infected with Mycobacterium tuberculosis. J Proteome Res. 10:2238–2247. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
O'Neill LAJ and Artyomov MN: Itaconate: The poster child of metabolic reprogramming in macrophage function. Nat Rev Immunol. 19:273–281. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Teleky BE and Vodnar DC: Biomass-derived production of itaconic acid as a building block in specialty polymers. Polymers (Basel). 11:10352019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shi X, Zhou H, Wei J, Mo W, Li Q and Lv X: The signaling pathways and therapeutic potential of itaconate to alleviate inflammation and oxidative stress in inflammatory diseases. Redox Biol. 58:1025532022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Q, Li XL, Mei Y, Ye JC, Fan W, Cheng GH, Zeng MS and Feng GK: The anti-inflammatory drug dimethyl itaconate protects against colitis-associated colorectal cancer. J Mol Med (Berl). 98:1457–1466. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Anderson NM, Mucka P, Kern JG and Feng H: The emerging role and targetability of the TCA cycle in cancer metabolism. Protein Cell. 9:216–237. 2018. View Article : Google Scholar :
|
|
29
|
Orecchioni M, Ghosheh Y, Pramod AB and Ley K: Macrophage polarization: Different gene signatures in M1(LPS+) vs classically and M2(LPS-) vs alternatively activated macrophages. Front Immunol. 10:10842019. View Article : Google Scholar
|
|
30
|
Gordon S and Martinez FO: Alternative activation of macrophages: Mechanism and functions. Immunity. 32:593–604. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Feng J, Li C, He H, Xu S, Wang X and Chen K: Construction of cell factory through combinatorial metabolic engineering for efficient production of itaconic acid. Microb Cell Fact. 21:2752022. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li Y, Gong W, Li W, Liu P, Liu J, Jiang H, Zheng T, Wu J, Wu X, Zhao Y and Ren J: The IRG1-itaconate axis: A regulatory hub for immunity and metabolism in macrophages. Int Rev Immunol. 42:364–378. 2023. View Article : Google Scholar
|
|
33
|
Wu R, Chen F, Wang N, Tang D and Kang R: ACOD1 in immunometabolism and disease. Cell Mol Immunol. 17:822–833. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lee CG, Jenkins NA, Gilbert DJ, Copeland NG and O'Brien WE: Cloning and analysis of gene regulation of a novel LPS-inducible cDNA. Immunogenetics. 41:263–270. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Degrandi D, Hoffmann R, Beuter-Gunia C and Pfeffer K: The proinflammatory cytokine-induced IRG1 protein associates with mitochondria. J Interferon Cytokine Res. 29:55–67. 2009. View Article : Google Scholar
|
|
36
|
Kang H, Liu T, Wang Y, Bai W, Luo Y and Wang J: Neutrophil-macrophage communication via extracellular vesicle transfer promotes itaconate accumulation and ameliorates cytokine storm syndrome. Cell Mol Immunol. 21:689–706. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hall CJ, Boyle RH, Astin JW, Flores MV, Oehlers SH, Sanderson LE, Ellett F, Lieschke GJ, Crosier KE and Crosier PS: Immunoresponsive gene 1 augments bactericidal activity of macrophage-lineage cells by regulating β-oxidation-dependent mitochondrial ROS production. Cell Metab. 18:265–278. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Runtsch MC, Angiari S, Hooftman A, Wadhwa R, Zhang Y, Zheng Y, Spina JS, Ruzek MC, Argiriadi MA, McGettrick AF, et al: Itaconate and itaconate derivatives target JAK1 to suppress alternative activation of macrophages. Cell Metab. 34:487–501.e8. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li Y, Li YC, Liu XT, Zhang L, Chen YH, Zhao Q, Gao W, Liu B, Yang H and Li P: Blockage of citrate export prevents TCA cycle fragmentation via Irg1 inactivation. Cell Rep. 38:1103912022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
De Souza DP, Achuthan A, Lee MK, Binger KJ, Lee MC, Davidson S, Tull DL, McConville MJ, Cook AD, Murphy AJ, et al: Autocrine IFN-I inhibits isocitrate dehydrogenase in the TCA cycle of LPS-stimulated macrophages. J Clin Invest. 129:4239–4244. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ganta VC, Choi MH, Kutateladze A, Fox TE, Farber CR and Annex BH: A MicroRNA93-interferon regulatory factor-9-immunoresponsive gene-1-itaconic acid pathway modulates M2-like macrophage polarization to revascularize ischemic muscle. Circulation. 135:2403–2425. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Huang J, Jiang Y, Ji R, Jia Y, Wang S, Zhou Z, Wang S, Wang J, Yang Q, Bai H, et al: Macrophage scavenger receptor A1 antagonizes abdominal aortic aneurysm via upregulating IRG1. Biochem Pharmacol. 213:1156312023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Scheurlen KM, Chariker JH, Kanaan Z, Littlefield AB, George JB, Seraphine C, Rochet A, Rouchka EC and Galandiuk S: The NOTCH4-GATA4-IRG1 axis as a novel target in early-onset colorectal cancer. Cytokine Growth Factor Rev. 67:25–34. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Schuster EM, Epple MW, Glaser KM, Mihlan M, Lucht K, Zimmermann JA, Bremser A, Polyzou A, Obier N, Cabezas-Wallscheid N, et al: TFEB induces mitochondrial itaconate synthesis to suppress bacterial growth in macrophages. Nat Metab. 4:856–866. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Larsen H and Eimhjellen KE: The mechanism of itaconic acid formation by Aspergillus terreus. 1. The effect of acidity. Biochem J. 60:135–139. 1955. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cheah HL, Lim V and Sandai D: Inhibitors of the glyoxylate cycle enzyme ICL1 in Candida albicans for potential use as antifungal agents. PLoS One. 9:e959512014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Otten A, Brocker M and Bott M: Metabolic engineering of Corynebacterium glutamicum for the production of itaconate. Metab Eng. 30:156–165. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lee CB, Šnajdr I, Tenora L, Alt J, Gori S, Krečmerová M, Maragakis RM, Paule J, Tiwari S, Iyer J, et al: Discovery of orally available prodrugs of itaconate and derivatives. J Med Chem. 68:3433–3444. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Willenbockel HF, Williams AT, Lucas A, Reynolds MB, Joulia E, Ruchhoeft ML, Dowerg B, Cabrales P, Metallo CM and Cordes T: In vivo itaconate tracing reveals degradation pathway and turnover kinetics. Nat Metab. 7:1781–1790. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Cooper RA and Kornberg HL: The utilization of itaconate by Pseudomonas sp. Biochem J. 91:82–91. 1964. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Adler J, Wang SF and Lardy HA: The metabolism of itaconic acid by liver mitochondria. J Biol Chem. 229:865–879. 1957. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Fan Y, Dan W, Wang Y, Ma Z, Jian Y, Liu T, Li M, Wang Z, Wei Y, Liu B, et al: Itaconate transporter SLC13A3 confers immunotherapy resistance via alkylation-mediated stabilization of PD-L1. Cell Metab. 37:514–526.e5. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Liu X, Shi B, Suo R, Xiong S, Wang X, Liang X, Li X and Li G: Itaconate regulates macrophage function through stressful iron-sulfur cluster disrupting and iron metabolism rebalancing. FASEB J. 35:e219362021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Itoh K, Wakabayashi N, Katoh Y, Ishii T, Igarashi K, Engel JD and Yamamoto M: Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 13:76–86. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Rodríguez-Prados JC, Través PG, Cuenca J, Rico D, Aragonés J, Martín-Sanz P, Cascante M and Boscá L: Substrate fate in activated macrophages: A comparison between innate, classic, and alternative activation. J Immunol. 185:605–614. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Qin W, Qin K, Zhang Y, Jia W, Chen Y, Cheng B, Peng L, Chen N, Liu Y, Zhou W, et al: S-glycosylation-based cysteine profiling reveals regulation of glycolysis by itaconate. Nat Chem Biol. 15:983–991. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Aso K, Kono M, Kanda M, Kudo Y, Sakiyama K, Hisada R, Karino K, Ueda Y, Nakazawa D, Fujieda Y, et al: Itaconate ameliorates autoimmunity by modulating T cell imbalance via metabolic and epigenetic reprogramming. Nat Commun. 14:9842023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Bardi GT, Smith MA and Hood JL: Melanoma exosomes promote mixed M1 and M2 macrophage polarization. Cytokine. 105:63–72. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Soto-Heredero G, Gómez de Las Heras MM, Gabandé-Rodríguez E, Oller J and Mittelbrunn M: Glycolysis-a key player in the inflammatory response. FEBS J. 287:3350–3369. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Wang F, Wang K, Xu W, Zhao S, Ye D, Wang Y, Xu Y, Zhou L, Chu Y, Zhang C, et al: SIRT5 desuccinylates and activates pyruvate kinase M2 to block macrophage IL-1β production and to prevent DSS-induced colitis in mice. Cell Rep. 19:2331–2344. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Tannahill GM, Curtis AM, Adamik J, Palsson-McDermott EM, McGettrick AF, Goel G, Frezza C, Bernard NJ, Kelly B, Foley NH, et al: Succinate is a danger signal that induces IL-1β via HIF-1α. Nature. 496:238–242. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Lampropoulou V, Sergushichev A, Bambouskova M, Nair S, Vincent EE, Loginicheva E, Cervantes-Barragan L, Ma X, Huang SC, Griss T, et al: Itaconate links inhibition of succinate dehydrogenase with macrophage metabolic remodeling and regulation of inflammation. Cell Metab. 24:158–166. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
An L, Zhai Q, Tao K, Xiong Y, Ou W, Yu Z, Yang X, Ji J and Lu M: Quercetin induces itaconic acid-mediated M1/M2 alveolar macrophages polarization in respiratory syncytial virus infection. Phytomedicine. 130:1557612024. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Blanco LP, Patino-Martinez E, Nakabo S, Zhang M, Pedersen HL, Wang X, Carmona-Rivera C, Claybaugh D, Yu ZX, Desta E and Kaplan MJ: Modulation of the itaconate pathway attenuates murine lupus. Arthritis Rheumatol. 74:1971–1983. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Muri J, Wolleb H, Broz P, Carreira EM and Kopf M: Electrophilic Nrf2 activators and itaconate inhibit inflammation at low dose and promote IL-1β production and inflammatory apoptosis at high dose. Redox Biol. 36:1016472020. View Article : Google Scholar
|
|
66
|
Ramalho T, Assis PA, Ojelabi O, Tan L, Carvalho B, Gardinassi L, Campos O, Lorenzi PL, Fitzgerald KA, Haynes C, et al: Itaconate impairs immune control of Plasmodium by enhancing mtDNA-mediated PD-L1 expression in monocyte-derived dendritic cells. Cell Metab. 36:484–497.e6. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Burczyk G, Cichon I and Kolaczkowska E: Itaconate suppresses formation of neutrophil extracellular traps (NETs): Involvement of hypoxia-inducible factor 1α (Hif-1α) and heme oxygenase (HO-1). Front Immunol. 13:8646382022. View Article : Google Scholar
|
|
68
|
Fousert E, Toes R and Desai J: Neutrophil extracellular traps (NETs) take the central stage in driving autoimmune responses. Cells. 9:9152020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Romagnani S: Th1/Th2 cells. Inflamm Bowel Dis. 5:285–294. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Kuo PC, Weng WT, Scofield BA, Paraiso HC, Brown DA, Wang PY, Yu IC and Yen JH: Dimethyl itaconate, an itaconate derivative, exhibits immunomodulatory effects on neuroinflammation in experimental autoimmune encephalomyelitis. J Neuroinflammation. 17:1382020. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Tomlinson KL, Lung TWF, Dach F, Annavajhala MK, Gabryszewski SJ, Groves RA, Drikic M, Francoeur NJ, Sridhar SH, Smith ML, et al: Staphylococcus aureus induces an itaconate-dominated immunometabolic response that drives biofilm formation. Nat Commun. 12:13992021. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Mcfadden BA and Purohit S: Itaconate, an isocitrate lyase-directed inhibitor in Pseudomonas indigofera. J Bacteriol. 131:136–144. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Hillier S and Charnetzky WT: Glyoxylate bypass enzymes in Yersinia species and multiple forms of isocitrate lyase in Yersinia pestis. J Bacteriol. 145:452–458. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Breen P, Zimbric M and Caverly LJ: Itaconic acid inhibits nontuberculous mycobacterial growth in pH dependent manner while 4-octyl-itaconic acid enhances THP-1 clearance of nontuberculous mycobacteria in vitro. PLoS One. 19:e03035162024. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Huang EY, Kwai BXC, Bhusal RP, Bashiri G and Leung IKH: Mycobacterium tuberculosis Rv1916 is an Acetyl-CoA-binding protein. Chembiochem. 24:e2023001622023. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Priya M, Gupta SK, Koundal A, Kapoor S, Tiwari S, Kidwai S, Sorio de Carvalho LP, Thakur KG, Mahajan D, Sharma D, et al: Itaconate mechanism of action and dissimilation in Mycobacterium tuberculosis. Proc Natl Acad Sci USA. 122:e24231141222025. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Liu Z, Liu D and Wang C: In situ chemoproteomic profiling reveals itaconate inhibits de novo purine biosynthesis in pathogens. Cell Rep. 43:1147372024. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Wang X, Menezes CJ, Jia Y, Xiao Y, Venigalla SSK, Cai F, Hsieh MH, Gu W, Du L, Sudderth J, et al: Metabolic inflexibility promotes mitochondrial health during liver regeneration. Science. 384:eadj43012024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Zhao R, Xu L, Chen J, Yang Y, Guo X, Dai M, Tian GB and Qin LN: Itaconate induces tolerance of Staphylococcus aureus to aminoglycoside antibiotics. Front Microbiol. 15:14500852024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Zhang B, Ku X, Zhang X, Zhang Y, Chen G, Chen F, Zeng W, Li J, Zhu L and He Q: The AI-2/luxS quorum sensing system affects the growth characteristics, biofilm formation, and virulence of haemophilus parasuis. Front Cell Infect Microbiol. 9:622019. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Xie LY, Xu YB, Ding XQ, Liang S, Li DL, Fu AK and Zhan XA: Itaconic acid and dimethyl itaconate exert antibacterial activity in carbon-enriched environments through the TCA cycle. Biomed Pharmacother. 167:1154872023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Che C, Fan X, Wang H, Wang Z, Bao Y, Jiang W, Nawaz S, Yin H and Han X: 4-octyl itaconate modulates virulence-associated phenotypes and oxidative stress resistance in avian pathogenic Escherichia coli by targeting menB and wza. Poult Sci. Dec 6–2025.Epub ahead of print. PubMed/NCBI
|
|
83
|
Bambouskova M, Gorvel L, Lampropoulou V, Sergushichev A, Loginicheva E, Johnson K, Korenfeld D, Mathyer ME, Kim H, Huang LH, et al: Electrophilic properties of itaconate and derivatives regulate the IκBζ-ATF3 inflammatory axis. Nature. 556:501–504. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Yang C and Wang X: Lysosome biogenesis: Regulation and functions. J Cell Biol. 220:e2021020012021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Zhang Z, Chen C, Yang F, Zeng YX, Sun P, Liu P and Li X: Itaconate is a lysosomal inducer that promotes antibacterial innate immunity. Mol Cell. 82:2844–2857.e10. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Murphy MP: How mitochondria produce reactive oxygen species. Biochem J. 417:1–13. 2009. View Article : Google Scholar
|
|
87
|
O'Carroll SM, Peace CG, Toller-Kawahisa JE, Min Y, Hooftman A, Charki S, Kehoe L, O'Sullivan MJ, Zoller A, Mcgettrick AF, et al: Itaconate drives mtRNA-mediated type I interferon production through inhibition of succinate dehydrogenase. Nat Metab. 6:2060–2069. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Luchkova A, Mata A and Cadenas S: Nrf2 as a regulator of energy metabolism and mitochondrial function. FEBS Lett. 598:2092–2105. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Wu L, Hu Z, Luo X, Ge C, Lv Y, Zhan S, Huang W, Shen X, Yu D and Liu B: Itaconic acid alleviates perfluorooctanoic acid-induced oxidative stress and intestinal damage by regulating the keap1/Nrf2/Ho-1 pathway and reshaping the gut microbiota. Int J Mol Sci. 25:98262024. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Singh N and Bernstein CN: Environmental risk factors for inflammatory bowel disease. United European Gastroenterol J. 10:1047–1053. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Pan X, Zhu Q, Pan LL and Sun J: Macrophage immunometabolism in inflammatory bowel diseases: From pathogenesis to therapy. Pharmacol Ther. 238:1081762022. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Chikina AS, Nadalin F, Maurin M, San-Roman M, Thomas-Bonafos T, Li XV, Lameiras S, Baulande S, Henri S, Malissen B, et al: Macrophages maintain epithelium integrity by limiting fungal product absorption. Cell. 183:411–428.e16. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Castro-Dopico T, Fleming A, Dennison TW, Ferdinand JR, Harcourt K, Stewart BJ, Cader Z, Tuong ZK, Jing C, Lok LSC, et al: GM-CSF calibrates macrophage defense and wound healing programs during intestinal infection and inflammation. Cell Rep. 32:1078572020. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Zhao X, Di Q, Liu H, Quan J, Ling J, Zhao Z, Xiao Y, Wu H, Wu Z, Song W, et al: MEF2C promotes M1 macrophage polarization and Th1 responses. Cell Mol Immunol. 19:540–553. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhang JX, Hu YX, Liu Y, Chen ZZ, Zheng JT, Qu XT, Zhang Y, Tang WY, Huang SC and Liu CS: Xianglian pill alleviates ulcerative colitis by inhibiting M1 macrophage polarization via modulation of energy metabolite itaconate. Phytomedicine. 135:1561792024. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kim HW, Yu AR, Lee JW, Yoon HS, Lee BS, Park HW, Lee SK, Lee YI, Whang J and Kim JS: Aconitate decarboxylase 1 deficiency exacerbates mouse colitis induced by dextran sodium sulfate. Int J Mol Sci. 23:43922022. View Article : Google Scholar :
|
|
97
|
Reagan-Shaw S, Nihal M and Ahmad N: Dose translation from animal to human studies revisited. FASEB J. 22:659–661. 2008. View Article : Google Scholar
|
|
98
|
Wang Y, Zhao X, Gao Y, Zhao C, Li J, Wang S, Xue B, Liu C and Ma X: 4-Octyl itaconate alleviates dextran sulfate sodium-induced ulcerative colitis in mice via activating the KEAP1-NRF2 pathway. Inflammopharmacology. 32:2555–2574. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Wang HG, Zhang MN, Wen X and Yang XZ: Itaconate aggravates experimental colitis. Clin Res Hepatol Gastroenterol. 45:1016292021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Swain A, Bambouskova M, Kim H, Andhey PS, Duncan D, Auclair K, Chubukov V, Simons DM, Roddy TP, Stewart KM and Artyomov MN: Comparative evaluation of itaconate and its derivatives reveals divergent inflammasome and type I interferon regulation in macrophages. Nat Metab. 2:594–602. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Tang C, Tan S, Zhang Y, Dong L and Xu Y: Activation of Keap1-Nrf2 signaling by 4-octyl itaconate protects human umbilical vein endothelial cells from high glucose. Biochem Biophys Res Commun. 508:921–927. 2019. View Article : Google Scholar
|
|
102
|
Li R, Ma Y, Wu H, Zhang X, Ding N, Li Z, Hu X, Rao J, Zhou Y, Wang L, et al: 4-Octyl itaconate alleviates endothelial cell inflammation and barrier dysfunction in LPS-induced sepsis via modulating TLR4/MAPK/NF-κB signaling: 4-Octyl itaconate alleviates endothelial dysfunction. Mol Med. 31:2402025. View Article : Google Scholar
|
|
103
|
Guo H, Guo H, Xie Y, Chen Y, Lu C, Yang Z, Zhu Y, Ouyang Y, Zhang Y and Wang X: Mo3Se4 nanoparticle with ROS scavenging and multi-enzyme activity for the treatment of DSS-induced colitis in mice. Redox Biol. 56:1024412022. View Article : Google Scholar
|
|
104
|
Wu R, Liu J, Wang N, Zeng L, Yu C, Chen F, Wang H, Billiar TR, Jiang J, Tang D and Kang R: Aconitate decarboxylase 1 is a mediator of polymicrobial sepsis. Sci Transl Med. 14:eabo20282022. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Nugent SG, Kumar D, Rampton DS and Evans DF: Intestinal luminal pH in inflammatory bowel disease: Possible determinants and implications for therapy with aminosalicylates and other drugs. Gut. 48:571–577. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Brinck JE, Sinha AK, Laursen MF, Dragsted LO, Raes J, Uribe RV, Walter J, Roager HM and Licht TR: Intestinal pH: A major driver of human gut microbiota composition and metabolism. Nat Rev Gastroenterol Hepatol. 22:639–656. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Klimeck L, Heisser T, Hoffmeister M and Brenner H: Colorectal cancer: A health and economic problem. Best Pract Res Clin Gastroenterol. 66:1018392023. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM and Wallace MB: Colorectal cancer. Lancet. 394:1467–1480. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Baidoun F, Elshiwy K, Elkeraie Y, Merjaneh Z, Khoudari G, Sarmini MT, Gad M, Al-Husseini M and Saad A: Colorectal cancer epidemiology: Recent trends and impact on outcomes. Curr Drug Targets. 22:998–1009. 2021. View Article : Google Scholar
|
|
110
|
Lasry A, Zinger A and Ben-Neriah Y: Inflammatory networks underlying colorectal cancer. Nat Immunol. 17:230–240. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wang H, Tian T and Zhang J: Tumor-associated macrophages (TAMs) in colorectal cancer (CRC): From mechanism to therapy and prognosis. Int J Mol Sci. 22:84702021. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Schmitt M and Greten FR: The inflammatory pathogenesis of colorectal cancer. Nat Rev Immunol. 21:653–667. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Liu Y, Sun Q, Zhang C, Ding M, Wang C, Zheng Q, Ma Z, Xu H, Zhou G, Wang X, et al: STING-IRG1 inhibits liver metastasis of colorectal cancer by regulating the polarization of tumor-associated macrophages. iScience. 26:1073762023. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Martina JA and Puertollano R: The IRG1/itaconate/TFEB axis: A new weapon in macrophage antibacterial defense. Mol Cell. 82:2732–2734. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Zhou S, Peng J, Xiao L, Zhou C, Fang Y, Ou Q, Qin J, Liu M, Pan Z and Hou Z: TRIM25 regulates oxaliplatin resistance in colorectal cancer by promoting EZH2 stability. Cell Death Dis. 12:4632021. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Payandeh Z, Pirpour Tazehkand A, Barati G, Pouremamali F, Kahroba H, Baradaran B and Samadi N: Role of Nrf2 and mitochondria in cancer stem cells; in carcinogenesis, tumor progression, and chemoresistance. Biochimie. 179:32–45. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Zheng Y, Chen Z, She C, Lin Y, Hong Y, Shi L, Zhang Y, Cao P and Xu X: Four-octyl itaconate activates Nrf2 cascade to protect osteoblasts from hydrogen peroxide-induced oxidative injury. Cell Death Dis. 11:7722020. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Yang W, Wang Y, Huang Y, Yu J, Wang T, Li C, Yang L, Zhang P, Shi L, Yin Y, et al: 4-Octyl itaconate inhibits aerobic glycolysis by targeting GAPDH to promote cuproptosis in colorectal cancer. Biomed Pharmacother. 159:1143012023. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Patel SG, Karlitz JJ, Yen T, Lieu CH and Boland CR: The rising tide of early-onset colorectal cancer: A comprehensive review of epidemiology, clinical features, biology, risk factors, prevention, and early detection. Lancet Gastroenterol Hepatol. 7:262–274. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Scheurlen KM, Hallion J, Snook DL, MacLeod A, Beal RJ, Parks MA, Littlefield AB, Hiken E, Billeter AT, Bensen J, et al: Itaconate and obesity-related hormones promote tumor progression-new insights on metabolic dysfunction in early-onset colon cancer. Front Immunol. 16:15729852025. View Article : Google Scholar
|
|
121
|
Niño DF, Sodhi CP and Hackam DJ: Necrotizing enterocolitis: New insights into pathogenesis and mechanisms. Nat Rev Gastroenterol Hepatol. 13:590–600. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Alsaied A, Islam N and Thalib L: Global incidence of necrotizing enterocolitis: A systematic review and meta-analysis. BMC Pediat. 20:3442020. View Article : Google Scholar
|
|
123
|
Neu J and Walker WA: Necrotizing enterocolitis. N Engl J Med. 364:255–264. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Neu J, Modi N and Caplan M: Necrotizing enterocolitis comes in different forms: Historical perspectives and defining the disease. Semin Fetal Neonatal Med. 23:370–373. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Apostolidi DM, Kapetanidi M, Vretou E, Sarantaki A, Lykeridou K, Karampas G, Diamanti A, Vlachou M, Pantelaki N, Deltsidou A, et al: Morbidity and mortality trends in preterm neonates at the limits of viability: Retrospective observations from one greek hospital. Life (Basel). 15:7082025.PubMed/NCBI
|
|
126
|
Hall NJ, Eaton S and Pierro A: Royal australasia of surgeons guest lecture. Necrotizing enterocolitis: Prevention, treatment, and outcome. J Pediatr Surg. 48:2359–2367. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC, Hale EC, Newman NS, Schibler K, Carlo WA, et al: Neonatal outcomes of extremely preterm infants from the NICHD neonatal research network. Pediatrics. 126:443–456. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Cho SX, Rudloff I, Lao JC, Pang MA, Goldberg R, Bui CB, McLean CA, Stock M, Klassert TE, Slevogt H, et al: Characterization of the pathoimmunology of necrotizing enterocolitis reveals novel therapeutic opportunities. Nat Commun. 11:57942020. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Huangfu S, Lan C, Li S, Wang H, Yan C, Yang Y, Tian B, Mu Y, Zhao P, Tian Y, et al: Itaconate suppresses neonatal intestinal inflammation via metabolic reprogramming of M1 macrophage. Clin Transl Med. 15:e704192025. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Donskey CJ: The role of the intestinal tract as a reservoir and source for transmission of nosocomial pathogens. Clin Infect Dis. 39:219–226. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Rougerie P, Miskolci V and Cox D: Generation of membrane structures during phagocytosis and chemotaxis of macrophages: Role and regulation of the actin cytoskeleton. Immunol Rev. 256:222–239. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
O'Callaghan AA, Dempsey E, Iyer N, Stiegeler S, Mercurio K and Corr SC: Intestinal metabolites influence macrophage phagocytosis and clearance of bacterial infection. Front Cell Infect Microbiol. 11:6224912021. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Joerger RD: Salmonella enterica's 'choice': Itaconic acid degradation or bacteriocin immunity genes. Genes (Basel). 11:7972020. View Article : Google Scholar
|
|
134
|
Hopkins EGD, Roumeliotis TI, Mullineaux-Sanders C, Choudhary JS and Frankel G: Intestinal epithelial cells and the microbiome undergo swift reprogramming at the inception of colonic citrobacter rodentium infection. mBio. 10:e00062–19. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Zhang T, Hasegawa Y and Waldor MK: Enteric bacterial infection stimulates remodelling of bile metabolites to promote intestinal homeostasis. Nat Microbiol. 9:3376–3390. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
ElAzzouny M, Tom CT, Evans CR, Olson LL, Tanga MJ, Gallagher KA, Martin BR and Burant CF: Dimethyl itaconate is not metabolized into itaconate intracellularly. J Biol Chem. 292:4766–4769. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Liao ST, Han C, Xu DQ, Fu XW, Wang JS and Kong LY: 4-Octyl itaconate inhibits aerobic glycolysis by targeting GAPDH to exert anti-inflammatory effects. Nat Commun. 10:50912019. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Xu S, Zhao JY, Ma JY, Cui XL, Lin JH, Sun SY, Liu SJ, Zhou GK, Zhang JT, Kang P and Liu T: Desensitization of TRPA1 by dimethyl itaconate attenuates acute and chronic pain in mice. Front Pharmacol. 16:16714612025. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Yin N, Zhang W, Wei R, Yang Q, He F, Guo L and Feng M: Liposome cocktail activator modulates hepatocytes and remodels the microenvironment to mitigate acute liver failure. Asian J Pharm Sci. 17:867–879. 2022.
|
|
140
|
Rezazadeh Ardabili A, Van Esser D, Wintjens D, Cilissen M, Deben D, Mujagic Z, Russ F, Stassen L, Van Bodegraven A, Wong D, et al: The risk of mild, moderate, and severe infections in IBD patients-A prospective, multicentre observational cohort study (PRIQ). J Crohns Colitis. Jul 9–2025.Epub ahead of print. View Article : Google Scholar
|
|
141
|
Ryan DG and O'Neill LAJ: Krebs cycle rewired for macrophage and dendritic cell effector functions. FEBS Lett. 591:2992–3006. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Sohail A, Iqbal AA, Sahini N, Chen F, Tantawy M, Waqas SFH, Winterhoff M, Ebensen T, Schultz K, Geffers R, et al: Itaconate and derivatives reduce interferon responses and inflammation in influenza A virus infection. PLoS Pathog. 18:e10102192022. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Yang Z, Zhang L, Ottavi S, Geri JB, Perkowski A, Jiang X, Pfau D, Bryk R, Aubé J, Zimmerman M, et al: ACOD1-mediated lysosomal membrane permeabilization contributes to Mycobacterium tuberculosis-induced macrophage death. Proc Natl Acad Sci USA. 122:e24253091222025. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Kang M, Tang D, Huang W, Zhou M, Ma Q, Lan Y, Chen D, Li Y and Han B: Emerging role of Itaconate in inflammatory bowel disease: From multitarget Immunometabolic mechanisms to therapeutic translation. Int Immunopharmacol. 166:1156322025. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Weiss JM, Palmieri EM, Gonzalez-Cotto M, Bettencourt IA, Megill EL, Snyder NW and McVicar DW: Itaconic acid underpins hepatocyte lipid metabolism in non-alcoholic fatty liver disease in male mice. Nat Metab. 5:981–995. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Zhang X, Zhi Y, Zan X, Fan K, Chen K, Zhao S, Dai X, Li L, Yang Y, Hu K, et al: Immune response gene 1 deficiency aggravates high fat diet-induced nonalcoholic fatty liver disease via promotion of redox-sensitive AKT suppression. Biochim Biophys Acta Mol Basis Dis. 1869:1666562023. View Article : Google Scholar : PubMed/NCBI
|


















