An aortic aneurysm (AA) is a life-threatening
condition characterized by localized or diffuse dilation of the
aortic wall, >50% of the normal diameter (1). The high associated mortality rate
is primarily due to rupture, with more than one-half of untreated
patients succumbing within 5 years (2). Post-rupture mortality rates range
from 35 to 67%, underscoring the grave prognosis (3). Often asymptomatic, AAs are
frequently undetected until rupture, which is fatal for most
patients before they reach medical care, earning them the moniker
'vascular time bombs'. Current management relies exclusively on
surgical interventions, such as endovascular or open repair, while
effective pharmacotherapies to prevent expansion or rupture remain
elusive (4). This urgency drives
research into the molecular pathophysiology of AAs to identify
novel, druggable targets.
The pathology of AAs features vascular smooth muscle
cell (VSMC) depletion, elastic fiber degradation and pervasive
inflammatory cell infiltration. These processes collectively
undermine aortic wall integrity, leading to dilation and
ultimately, dissection or rupture. Persistent VSMC loss is a
central hallmark, driven by mechanisms such as senescence,
phenotypic switching and regulated cell death, with the latter
being a major contributor (5).
Emerging evidence also implicates endothelial cell (EC) dysfunction
and death in aortic disease development (6). Simultaneously, inflammatory and
autoimmune responses at the medial-adventitial junction are
critical triggers of aortic destruction and dilation (7). Infiltration of immune cells (for
example, macrophages and lymphocytes) into the aortic wall is a
hallmark of aneurysm formation (8). Infiltrating macrophages,
particularly classically activated M1-type cells, release large
amounts of proinflammatory cytokines, sustaining a proinflammatory
microenvironment (9). This
persistent vascular inflammation not only initiates aneurysm
development but also profoundly accelerates pathological aortic
remodeling by triggering VSMC death, inducing pathological
phenotypic switching (for example, to a synthetic state), and
promoting the secretion of extracellular matrix (ECM)-degrading
proteases (10). The suppression
of inflammation has been shown to be effective in attenuating
aneurysm progression (11).
Multiple types of regulated cell death have been
reported, such as necroptosis, ferroptosis, cuproptosis,
disulfidptosis and pyroptosis (12). Among these, pyroptosis, which is
an exceedingly inflammatory type, has undergone a revolutionary
decade of mechanistic elucidation, revealing its dual role in
disease: Moderate activation mediates host defense, whereas
excessive activation exacerbates tissue damage (13). However, research into specific
pyroptosis mechanisms in AAs remains limited. A deeper
understanding of pyroptosis in AAs may reveal new therapeutic
opportunities to slow disease progression.
The present review summarizes recent advances in the
understanding of pyroptosis and its relevance to AAs. A focus is
placed on the pyroptotic pathway, the role of pyroptosis in diverse
aneurysmal pathologies and the underlying molecular mechanisms,
with a particular emphasis on the key effector protein, gasdermin D
(GSDMD). Furthermore, a detailed discussion of potential
therapeutic strategies targeting pyroptosis for AA intervention is
provided.
Understanding the multilayered molecular signaling
pathways that regulate pyroptosis is fundamental to the development
of targeted therapies.
Pyroptosis is now characterized as a type of
regulated, lytic cell death executed by the pore-forming activity
of the N-terminal domains of GSDM family proteins in the plasma
membrane (14). This process is
triggered by infectious stimuli [such as pathogen-associated
molecular patterns (PAMPs)] or non-infectious danger signals [such
as damage-associated molecular patterns (DAMPs)] (15). Hallmark features include robust
proinflammatory activity and pore-dependent execution (16).
Morphologically, pyroptotic cells undergo
characteristic changes. The N-terminal domain of GSDM proteins
oligomerizes to form 10- to 20-nm pores in the plasma membrane
(17). This process induces
ionic imbalances (K+ efflux and
Na+/Ca2+ influx) and osmotic lysis, resulting
in cellular swelling, organellar edema and eventual plasma membrane
rupture with the release of intracellular contents and inflammatory
mediators (18,19). The nucleus exhibits chromatin
condensation (often marginalized) but lacks the DNA laddering
characteristic of apoptosis (20). Molecularly, pyroptosis crucially
depends on activated inflammatory caspases, which cleave GSDM
proteins to release the active, pore-forming N-terminal domain from
autoinhibition (21,22). Caspase activation is typically
driven by specific inflammasome complexes upon recognition of PAMPs
or DAMPs (23). These caspases
also process proinflammatory cytokine precursors [pro-interleukin
(IL)-1β and pro-IL-18] into their mature, active forms (24).
Functionally, pyroptosis essentially initiates and
amplifies inflammatory responses and plays pivotal roles in immune
defense and disease pathogenesis. Membrane rupture leads to the
massive release of cellular contents [DAMPs, such as high mobility
group box 1 (HMGB1) protein, adenosine triphosphate (ATP), IL-1α
and IL-33] and mature IL-1β and IL-18 (25). These alarmins potently activate
and recruit neighboring immune cells, amplify the inflammatory
cascade and establish an immune defense mechanism for intracellular
pathogen clearance (26).
However, uncontrolled pyroptosis can drive intense inflammation and
serve as a central pathogenic driver in diseases such as
autoinflammatory disorders and atherosclerosis (27,28).
The execution of pyroptosis across all known
pathways ultimately relies on the GSDM protein family, whose pore
formation is the indispensable final common step. In humans, this
family comprises GSDMA, GSDMB, GSDMC, GSDMD, GSDME and deafness
autosomal recessive 59 (29).
GSDM family members A-E consist of a highly differentiated peptide
linker domain, a conserved cytotoxic N-terminus (GSDM-N) that forms
pores and a conserved C-terminus (GSDM-C) that inhibits GSDM-N
(30). A self-locking regulatory
mechanism that consists of GSDM-C and GSDM-N ensures that the
active GSDM-N domain is released only after specific cleavage by
inflammatory caspases, thereby preventing cell damage caused by
spontaneous activation (31).
GSDM pores use an 'electrostatic sieve' mechanism, orchestrating
the selective and efficient release of IL-1β/IL-18. Concurrently,
pore-driven K+ efflux activates the NOD-like receptor
family-pyrin domain-containing 3 (NLRP3) inflammasome, initiating a
positive feedback loop that amplifies inflammatory responses
(32).
GSDM family members exhibit distinct expression
patterns and functions. GSDMD is widely expressed and
constitutively highly expressed in myeloid cells (for example,
neutrophils, macrophages and dendritic cells) (33). GSDMD serves as the most specific
and efficient substrate for caspase-1/4/5/11 and is considered the
most versatile executor (22).
GSDME is widely distributed across the placenta, intestines,
thyroid gland and brain (34);
it can switch apoptotic stimuli to pyroptotic death stimuli
(35). GSDMB is enriched in the
gastrointestinal epithelium and in colorectal cancer, and can be
cleaved by granzyme A (GzmA) (36,37). GSDMA and GSDMC are predominantly
restricted to epithelial surfaces and the skin (38).
The multipathway nature of pyroptosis initiation
reflects the flexibility of cells in response to various threats,
including PAMPs, DAMPs, cytosolic disturbances and bacterial
components, such as lipopolysaccharide (LPS) (Fig. 1).
The canonical caspase-1-dependent pathway is the
most extensively studied pyroptosis pathway, and is primarily
activated in immune cells during host defense against pathogens.
PAMPs, DAMPs and cytosolic disturbances activate pattern
recognition receptors (PRRs) (39). Subsequently, PRRs trigger
oligomeric sensors, the adaptor protein apoptosis-associated
speck-like protein (ASC) and the effector inactive pro-caspase-1 to
assemble canonical inflammasomes (40). Five sensor types, NLRP3, absent
in melanoma 2 (AIM2), NOD-like receptor family-pyrin domain
containing 1, pyrin and NOD-like receptor c4 (NLRC4), primarily
mediate the canonical pathway. The caspase recruitment domain
(CARD) domain of ASC subsequently engages the CARD domain of
pro-caspase-1, triggering self-cleavage of pro-caspase-1 to
generate mature caspase-1 (41).
Activated caspase-1 then cleaves both GSDMD to generate the
pore-forming GSDMD N-terminal (GSDMD-NT) and pro-IL-1β/pro-IL-18
into their mature forms (42).
After that, pores in the cell membrane form and pyroptosis
occurs.
The non-canonical caspase-4/5-dependent pathway in
humans and the non-canonical caspase-11-dependent pathway mice
responds directly to cytosolicLPSfrom Gram-negative bacteria
(30). These caspases are
upregulated by type I interferon signaling and are activated upon
LPS binding without inflammasome assembly to directly cleave GSDMD
to induce pyroptosis (42).
Moreover, K+ efflux mediated by GSDMD pores indirectly
activates the NLRP3 inflammasome-caspase-1 canonical pyroptosis
pathway (22). This indirect
mechanism is recognized as central to non-canonical pathway-induced
mature cytokine release and secondary inflammatory amplification.
Caspase-4/5/11 cannot directly process pro-IL-1β/pro-IL-18
(43,44). However, recent evidence has
demonstrated that LPS-activated human caspase-4 and -5 can cleave
pro-IL-18 at sites identical to those targeted by caspase-1
(24). These findings challenge
conventional perception, suggesting that non-canonical caspases may
have the ability to directly address specific cytokine precursors
under specific circumstances.
In the caspase-3-mediated pathway, caspase-3,
traditionally designated as the key executioner of apoptosis, can
initiate pyroptotic switching under specific stimuli. In
GSDME-expressing cells, activated caspase-3 mediates specific
cleavage of GSDME, generating its N-terminal pore-forming domain
fragment. Subsequent plasma membrane pores and pyroptosis occur
thereby executing a modality shift from apoptosis to pyroptosis
(35). This process plays a
particularly important role in tumor cell killing by
chemotherapeutic agents.
In the caspase-8-mediated pathway, caspase-8 is a
multifunctional initiator of caspase that promotes pyroptosis
through multiple mechanisms; it can directly cleave GSDMD or GSDMC,
or activate GSDME via caspase-3, leading to pyroptosis. This
process, characterized by the absence of canonical IL-18 release
coupled with compensatory IL-1α secretion, has been described as
'incomplete pyroptosis' (45).
For instance, Yersinia infection triggers the canonical
pyroptosis pathway through direct caspase-8 activation, where
caspase-8 cleaves GSDMD (46).
Under hypoxic conditions in the tumor microenvironment, tumor
necrosis factor-α (TNFα) stimulation causes caspase-8 to cleave
GSDMC, shifting the mode of cell death from apoptosis to pyroptosis
(47). In caspase-1/11-deficient
macrophages, the NLRP3 inflammasome activates caspase-3/8, which
subsequently cleaves GSDME to induce pyroptosis. GSDMB can be
activated by caspase-3/6/7.
The Gzm-mediated pathway represents a mechanism
through which adaptive immune cells (cytotoxic T lymphocytes and
natural killer cells) directly induce pyroptosis in target cells.
Gzms, serine proteases delivered into target cells via perforin,
can directly cleave GSDM proteins. The Gzm family comprises five
known human members: GzmA, GzmB, GzmH, GzmK and GzmM (48). Among them, GzmA is the most
abundant, while GzmB is distinguished by its potent proapoptotic
activity (49,50). GzmB induces pyroptosis through
both caspase-3-dependent and caspase-3-independent mechanisms: It
rapidly activates caspase-3, which cleaves GSDME, and can also
directly cleave GSDME itself (51). By contrast, GzmA directly
hydrolyzes and cleaves GSDMB, triggering its pore-forming activity
(37). This Gzm-mediated
pyroptosis effectively amplifies inflammation in contexts such as
antitumor immunity (52).
Multiple aspects of pyroptosis are involved in the
pathology of Aas, as described in Fig. 2 and Table I (53-73).
Accumulating evidence directly implicates pyroptosis
activation in aneurysmal lesions, particularly in VSMCs. Both human
AA tissues and mouse models consistently exhibit elevated levels of
activated pyroptotic executors, including GSDMD-NT accumulation and
mature IL-1β and IL-18, specifically in VSMCs within the lesion
wall (53,74). Furthermore, components of the
NLRP3 inflammasome are upregulated in AA tissues, with activation
markers such as ASC speck formation and cleaved caspase-1 detected
specifically in VSMCs (54,58). Functional studies using NLRP3- or
caspase-1/11-deficient mice have revealed markedly attenuated
vascular dilation, reduced VSMC death and decreased disease
incidence across multiple aneurysm models (57,58,75,76). Crucially, abdominal AAs (AAAs)
are associated with increased TNFα levels. Wang et al
(53) observed cell swelling and
pore formation in the VSMC membrane under TNFα stimulation by
scanning electron microscopy, indicating the pyroptotic lytic death
of VSMCs during mouse AAAs. This direct and rapid mode of cell
death leads to substantial and irreversible loss of VSMCs,
disrupting the cellular foundation required for maintaining aortic
wall tension and structural integrity. Thus, pyroptosis represents
a key mechanism driving vascular wall thinning, dilation and
eventual rupture.
The profound impact of pyroptosis on aneurysmal
pathology extends far beyond the direct killing of VSMCs; it
triggers and perpetuates a storm of inflammation within the aortic
wall.
First, as aforementioned, cellular components and
mature and highly bioactive IL-1β and IL-18 secreted from VSMCs
undergoing pyroptosis function as potent alarm signals and
chemoattractants, robustly recruiting circulating monocytes,
neutrophils, T lymphocytes and other inflammatory cells into the
vascular wall, particularly at the medial-adventitial junction
(77,78). IL-1β and IL-18 directly activate
ECs to generate adhesion molecules, facilitating leukocyte rolling,
adhesion and transmigration (79). Released ATP acts as an agonist
for the purinergic receptor p2x-ligand-gated ion channel 7 (P2X7)
receptor on macrophages, thereby activating them, whereas HMGB1
strongly promotes proinflammatory effects via receptor for advanced
glycation end product/toll-like receptor (TLR) signaling (80,81).
Second, pyroptosis establishes a positive feedback
loop that aggravates vascular inflammation. Infiltrating immune
cells, particularly macrophages, are strongly activated by
pyroptosis-derived factors (especially IL-1β and IL-18) and DAMPs,
polarizing toward a proinflammatory M1 phenotype (82). These cells subsequently produce
and release large amounts of proinflammatory cytokines (such as
TNFα, interleukin-6 and IL-1β), chemokines (such as monocyte
chemotactic factor/c-c motif chemokine ligand 2) and reactive
oxygen species (ROS) (83,84). Crucially, they may undergo
pyroptosis themselves, leading to further release of DAMPs and
cytokines. This creates a self-sustaining and amplifying vicious
cycle of 'pyroptosis-inflammation-more pyroptosis/inflammation'
(72,85). This escalation of the
inflammatory milieu not only further damages and kills additional
VSMCs (through pyroptosis or other death modalities) but also
profoundly affects the other aforementioned key pathological
processes, ultimately driving pathological vascular remodeling
through the sustained release of proinflammatory cytokines and
proteases (86). As shown by
Zhang et al (85),
exosomal lncRNA plasmacytoma variant translocation 1 (PVT1), which
originates from M1 macrophages, promotes human aortic vascular
smooth muscle cell (HA-VSMC) pyroptosis and inflammation via the
miR-186-5p/HMGB1 pathway, upregulating NLRP3 and GSDMD expression,
thereby exacerbating AAA progression (85).
Pyroptosis does not occur in isolation but is
intricately interwoven with other pathological processes in AAs,
collectively driving disease progression. The first of these
processes is VSMC phenotypic switching. The study by Sun et
al (87) demonstrated that
NLRP3 inhibition prevents platelet-derived growth factor subunit
B-induced phenotypic modulation in HA-VSMCs (87). Similarly, the genetic deficiency
of NLRP3 attenuates angiotensin II (Ang II)-induced phenotypic
switching and vascular remodeling in mouse VSMCs (88). NLRP3 activation contributes to
the phenotypic transition and proliferation of rat VSMCs in
hypertension (89). Inflammatory
mediators and cytokines released during pyroptosis can induce
surviving VSMCs to shift from a contractile to a synthetic
phenotype (87). This transition
impairs contractile function and promotes the secretion of
proinflammatory factors and matrix metalloproteinases (MMPs),
further disrupting vascular homeostasis and accelerating
inflammation and matrix degradation. The second process is ECM
degradation. The inflammatory microenvironment triggered by
pyroptosis plays a central role in promoting ECM degradation. High
levels of cytokines (such as IL-1β and TNFα) upregulate multiple
MMPs (including MMP-2 and MMP-9), disrupting the balance between
MMPs and their endogenous inhibitors (57). The NLRP3 inflammasome may further
directly activate pro-enzymes through cleavage of inhibitory
domains (53). Studies have
demonstrated that NLRP3 or caspase-1 deficiency significantly
reduces elastin degradation and MMP activation during early mouse
AAA formation (57,76,90). Activated MMPs degrade critical
structural components of the aortic wall, namely, elastic fibers
and collagen, resulting in a precipitous loss of tensile strength,
decreased wall compliance, and ultimately constituting the
biomechanical basis for dilation and rupture. Infiltrating
inflammatory cells, such as neutrophils and macrophages, are also
major sources of MMPs (91). The
third pathological process in AAs is EC dysfunction.
Pyroptosis-induced EC death and associated inflammatory factors
compromise endothelial integrity, increase vascular permeability,
and facilitate plasma leakage and leukocyte extravasation,
exacerbating vascular inflammation and edema. Furthermore,
pyroptosis-related EC injury may cause dysregulation of the
secretion of vasoactive factors (92). A fourth process is crosstalk with
other programmed cell death pathways. VSMCs within the aneurysmal
microenvironment often undergo multiple forms of programmed cell
death simultaneously or sequentially. For instance, in Ang
II-induced mouse AAA models, markers of pyroptosis (GSDMD
activation), apoptosis (caspase-3 cleavage) and necroptosis (mixed
lineage kinase domain-like protein phosphorylation) have been
detected concurrently in VSMCs (53,93,94). Complex molecular crosstalk occurs
among these pathways. The coexistence and interaction of these
death mechanisms likely synergistically amplify VSMC loss and
inflammatory responses, thereby accelerating aneurysmal
progression. The differences and connections between them will be
elaborated later in the review.
Studies have indicated that cell-specific deletion
of GSDMD in either VSMCs or macrophages ameliorates AAA development
and progression (74,86). In various AAA induction models,
Gsdmd−/− mice consistently exhibited significant and
concordant protective effects. Specifically, VSMC-specific GSDMD
knockout substantially attenuated aneurysm formation, aortic
dilation, VSMC death, inflammatory responses and ECM degradation
(74). Research by Ye et
al (86) further confirmed
that macrophage-specific GSDMD deletion also delayed mouse AAA
progression (86). These genetic
findings are strongly supported by pharmacological interventions:
Compounds that specifically inhibit GSDMD pore formation or
oligomerization confer therapeutic benefits in AAA mouse models
(65). Together, these results
underscore GSDMD as a highly attractive and precise therapeutic
target for inhibiting pyroptosis in aneurysms.
GSDMD plays a uniquely destructive role in
aneurysms, as its ability to mediate pyroptosis is rapid and lytic.
Although VSMCs in AAs may be exposed to multiple cell death
signals, GSDMD-driven pyroptosis is rapid and lytic, causing
complete cellular disintegration and the rapid release of cellular
contents. By contrast, apoptosis is generally slower and non-lytic
(via phagocytic clearance) and tends to be anti-inflammatory or
immunologically silent (12,95). Thus, pyroptosis poses a greater
threat due to its acute and inflammatory nature, leading to a
substantial net loss of structural cells. Furthermore, as a 'master
switch' that initiates and amplifies lethal inflammatory storms,
the GSDMD pore-mediated release of inflammatory mediators occurs
explosively and at high local concentrations, resulting in a
proinflammatory effect far exceeding that of other secretory
mechanisms (96). As previously
discussed, these mediators further amplify inflammatory signaling
and cellular damage (97).
Consequently, GSDMD pores essentially control the release of the
most potent initial triggers (IL-1β/IL-18) and critical DAMPs in
the pro-aneurysmal inflammatory cascade (86). Without GSDMD pore formation, even
upon upstream inflammasome activation, the release efficiency of
these key factors is substantially compromised, resulting in a
failure to ignite or sustain the intense inflammatory storm
necessary for driving aneurysm progression (98,99). This establishes GSDMD as an
indispensable hub and amplifier connecting upstream danger sensing
(inflammasome activation) with widespread downstream inflammatory
destruction (immune cell infiltration, activation and cytokine
storm).
In addition to GSDMD, other GSDM family members,
notably GSDMB and GSDME, and potentially GSDMA/GSDMC, have been
implicated as pyroptotic executors in aneurysmal vascular
remodeling processes (64,100,101).
These paralogs share a common execution mechanism,
yet their upstream activating signals are markedly different. GSDMB
is cleaved by lymphocyte-derived GzmA at Lys244 and is upregulated
in human AAA tissues in parallel with caspase-4 (102). A functional study has
demonstrated that GSDMB knockdown attenuates macrophage pyroptosis
via the mitochondrial ROS (mtROS)-dynamin-related protein 1
(Drp1)-caspase-4 axis, subsequently suppressing VSMC phenotypic
switching and apoptosis, and implicating GSDMB in AA pathogenesis
through immune-VSMC crosstalk (64). By contrast, GSDME activation is
intimately linked to the apoptosis-pyroptosis continuum: In both
human and murine aneurysmal tissues, GSDME mediates non-canonical
pyroptosis and modulates inflammatory responses and senescence
following VSMC phenotypic transition (100). Moreover, AMP-activated protein
kinase-mediated phosphorylation of GSDME suppresses its
proinflammatory activity, revealing a regulatory interface between
metabolic homeostasis and cell death (103). Although direct evidence linking
GSDMA or GSDMC to AAs is currently lacking, their non-canonical
activation mechanisms in other systems, including streptococcal
toxin-mediated GSDMA cleavage and GzmB-induced GSDMC pyroptosis in
melanoma cells, offer exciting clues for potential alternative
activation pathways in the vasculature (104,105). Furthermore, the non-proteolytic
activation modes observed in lower eukaryotes, such as dimer
dissociation-dependent activation of Tricho-GSDMs in Trichoplax
adhaerens, suggest at an even broader diversity of GSDM
regulatory mechanisms that may extend beyond current paradigms
(106).
At the cellular level, emerging evidence has
revealed a preliminary division of labor among GSDM family members
within the aortic wall. GSDMD is ubiquitously expressed in
macrophages, T lymphocytes, VSMCs and ECs, and functions as a
multilineage pyroptosis executor (65,74,86,107). GSDMB is predominantly enriched
in macrophages, indirectly shaping VSMC fate through immune
modulation (64), and GSDME is
preferentially upregulated in VSMCs, potentially serving as a
molecular switch that governs the apoptosis-pyroptosis transition
(100). This functional
segregation suggests that GSDMD and GSDMB may synergistically
orchestrate immune defense in AAs, whereas GSDME governs VSMC death
modality decisions. Although direct evidence for functional
redundancy or complementarity among GSDM members remains elusive,
these observations provide a conceptual framework for deciphering
the spatiotemporal coordination of multicellular and multipathway
death programs during AA progression.
As aforementioned, AA progression involves the
concurrent or competing activation of multiple regulated cell death
pathways, among which pyroptosis, apoptosis, necroptosis and
ferroptosis exhibit distinct triggers, execution mechanisms,
inflammatory consequences and cell-type preferences, collectively
constituting a complex regulatory network that governs vascular
cell fate.
Pyroptosis, the central focus of this review, has
previously been characterized in detail in AAs. By contrast,
apoptosis plays a dual role in AA pathobiology that is highly
context dependent. As a non-lytic death modality, apoptosis is
initiated by intrinsic mitochondrial pathways (Bcl-2 associated X
protein/Bcl-2 antagonist/killer 1-caspase-9) or extrinsic death
receptor pathways [fas cell surface death receptor (Fas)-fas
ligand-caspase-8], culminating in caspase-3/7-mediated cellular
dismantling and phagocytic clearance without eliciting inflammation
(108-110). During early AA formation,
physiological apoptosis contributes to vascular remodeling;
however, in progressive stages, excessive VSMC apoptosis leads to
medial thinning and elastic fiber depletion, whereas uncleared
macrophage apoptosis may undergo secondary necrosis, exacerbating
local inflammation (111).
Notably, apoptosis-pyroptosis transformation has recently garnered
attention. For instance, caspase-8 can initiate apoptosis and under
certain conditions, cleave GSDMD or activate NLRP3. Apoptotic
bodies generated during apoptosis may be phagocytosed by
macrophages, triggering further inflammation and pyroptosis. In
addition, cytokines released during pyroptosis can induce apoptosis
in neighboring cells (112,113). This mechanism was preliminarily
validated in aneurysmal VSMCs (64).
Necroptosis, a prototypical type of necrosis
pathway, is triggered by death receptors (tumor necrosis factor
receptor 1 and Fas) or PRRs (TLR3/4) under conditions of caspase-8
inhibition, which are executed via the receptor interacting
serine/threonine kinase 1 (RIPK1)-RIPK3-mixed lineage kinase
domain-like cascade to form plasma membrane pores and release
cellular contents, with the intensity of inflammation intermediate
between that of apoptosis and pyroptosis (114,115). In AAs, necroptosis has been
documented in both macrophages and VSMCs and is associated with MMP
activation and vascular wall remodeling (116). However, its shared upstream
signals with pyroptosis (for example, RIPK1 involvement in
inflammasome regulation) and similar lytic morphology suggest
potential functional overlap or compensatory mechanisms.
Ferroptosis is characterized by iron-dependent lipid
peroxidation, resulting from inactivation of the system
Xc−/glutathione/glutathione peroxidase 4
antioxidant axis or disruption of the ferroptosis suppressor
protein 1/CoEnzyme Q10 pathway (117-120). Morphologically, it features
mitochondrial shrinkage with initially preserved plasma membrane
integrity, culminating in membrane rupture and the release of DAMPs
(121). In AAs, iron deposition
and lipid peroxidation products are markedly elevated; VSMC
ferroptosis has been shown to be involved in medial degeneration
and potentially cross talks with pyroptosis. Ferroptosis-derived
lipid peroxides activate the NLRP3 inflammasome, while
pyroptosis-released heme exacerbates iron overload (122).
In summary, in the aneurysmal context, pyroptosis,
apoptosis, necroptosis and ferroptosis do not occur in isolation
but instead form an integrated programmed cell death network
through shared triggers, competition for caspase resources and
reciprocal modulation of inflammatory outputs. Pyroptosis is a
network hub with a potent proinflammatory capacity; apoptosis
promotes basal cellular turnover but can be switched to pyroptosis
to amplify pathology, while necroptosis serves as an alternative
lytic pathway when caspases are compromised, and ferroptosis is
involved in positive feedback with pyroptosis through lipid
peroxidation and oxidative stress. The cell-type preferences,
temporal dynamics and functional consequences of these four death
modalities in AAs are summarized comparatively in Table II (38,86,122-132).
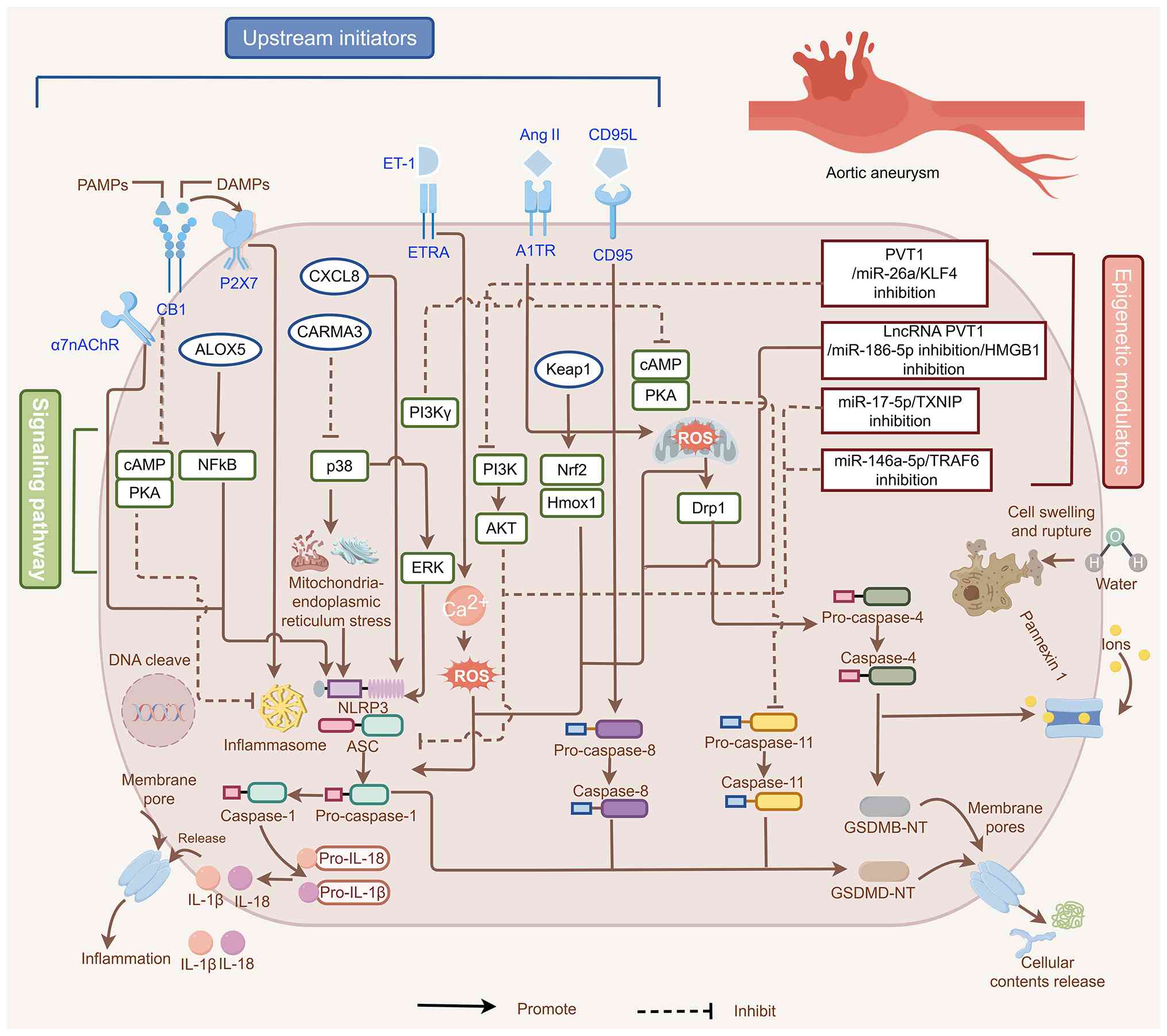
The initiation of pyroptosis in AAs is governed by a
multilayered network of upstream signaling cascades (Fig. 3). This architecture can be
deconstructed into four hierarchical tiers, including the
triggering level, transduction level, effector level and regulatory
level. At the triggering level, a repertoire of membrane receptors,
including α7 nicotinic acetylcholine receptor (α7nAChR),
cannabinoid receptor type 1, P2X7, endothelin receptor type A and
Ang II's type 1 receptor, senses danger signals within the
aneurysmal microenvironment (55,57,62,67,72). At the transduction level, signals
from receptors are transduced to the intracellular level via the
modulation of key second messengers, such as cyclic adenosine
monophosphate (cAMP)/protein kinase A, nuclear factor
κ-light-chain-enhancer of activated B cells (NF-κB), p38
mitogen-activated protein kinase (p38)/extracellularly regulated
protein kinases (ERK), phosphatidylinositol 3-kinase (PI3K)/protein
kinase B (AKT), nuclear factor erythroid 2-related factor 2
(Nrf2)/heme oxygenase 1 and Ca2+ influx. Next, these
intracellular signals converge on the effector level, namely the
NLRP3 inflammasome assembly (53-56,67,69,73). Parallel kinase pathways, such as
p38/ERK and PI3K/AKT, are engaged by effectors such as caspase
recruitment domain family member 10 deficiency, further amplifying
inflammasome assembly and crosstalk with the endoplasmic reticulum
and mitochondrial stress (71).
Non-canonical pyroptosis in AAs involves caspase-4/11-mediated
GSDMB/GSDMD cleavage, which is driven by the mtROS-Drp1 or
PI3Kγ-cAMP axis (64,69). Superimposed upon this core
cascade is the regulatory level mediated by epigenetic mechanisms,
where lncRNAs (such as PVT1) and miRNAs (such as miR-186-5p,
miR-146a-5p, miR-17-5p and miR-26a) fine-tune signaling intensity
by targeting key nodal molecules, including tumor necrosis factor
receptor-associated factor 6 (TRAF6), thioredoxin interacting
protein, HMGB1 and krüppel-like factor 4 (63,70,73,85). Furthermore, the cluster of
differentiation 95 ligand-cluster of differentiation 95 axis
induces caspase-8-mediated pyroptosis, whereas the kelch-like
ECH-associated protein 1-Nrf2 pathway provides antioxidative
counter-regulation (53,66).
The aforementioned levels are illustrated by
specific examples in AA; for example, at the triggering and signal
transduction level, TRAF6 promotes macrophage pyroptosis and
accelerates mouse AAAs through NLRP3 inflammasome engagement
(61). The
endothelin-1/Ca2+ axis similarly drives VSMC pyroptosis
and mouse AAA formation via NLRP3 and GSDMD activation (55). Conversely, α7nAChR activation
suppresses NLRP3 and GSDMD activity, limiting pyroptosis and
attenuating mouse AAAs (62). At
the regulatory level, exosomal miR-17-5p from adipose-derived
mesenchymal stem cells alleviates mouse AAAs by inhibiting
thioredoxin-interacting protein-NLRP3 signaling and GSDMD-NT
expression (63).
Collectively, these intertwined upstream signals
precisely orchestrate canonical and non-canonical pyroptosis
pathways in vascular cells, fueling the sustained inflammation and
matrix degradation that characterize aneurysm progression. Related
studies are shown in Table I
(53-73).
Given the central role of pyroptosis in AAs, the
development of specific inhibitors has emerged as a promising
therapeutic frontier. The pathway offers multiple nodes for
pharmacological intervention, from upstream sensors to downstream
inflammatory effectors (Fig. 2;
Table III) (54,65,66,133-151). This section will systematically
review these strategies and evaluate the mechanisms, advantages and
limitations of their main representative compounds from the
perspective of medicinal chemistry.
Inflammasomes, particularly NLRP3, are attractive
upstream targets.
MCC950 is a potent and selective small-molecule
inhibitor of NLRP3; it binds to the NACHT domain of NLRP3,
inhibiting its ATPase activity and thereby preventing inflammasome
oligomerization and activation (152). Notably, MCC950 suppresses NLRP3
activation through both canonical and non-canonical pathways
(153). In apolipoprotein
E-deficient (ApoE−/−) mice fed a Western diet, MCC950
delayed atherosclerosis progression and reduced IL-1β and IL-18
secretion (133). Although
direct evidence in AA models needs accumulation, its potent
anti-inflammatory effects suggest considerable therapeutic
potential in AAs, including the alleviation of vascular
inflammation and VSMC dysfunction. MCC950 has entered clinical
trials for several autoinflammatory diseases; however, its
development was halted in one trial due to hepatotoxicity, and
structural optimization efforts are ongoing to improve its safety
profile (154-156). Similar to MCC950, CY-09 and
3,4-meth ylenedioxy-β-nitrostyrene (MNS) also bind directly to the
NACHT domain of NLRP3, competitively inhibiting ATP binding and
blocking NLRP3 activation (134,157). CY-09 has been shown to
alleviate aortic valve stenosis and calcification by suppressing
proinflammatory cytokine expression through NLRP3 inhibition,
whereas MNS improved outcomes in murine colitis (134,157). The natural compound curcumin
modulates NLRP3 inflammasome activation by regulating NF-κB
signaling, attenuating K+ efflux, blocking
Ca2+ influx and preventing ASC oligomerization with
NLRP3, and has been demonstrated to reduce endothelial damage and
atherosclerosis in mice (136,158).
Beyond NLRP3, specific inhibitors for AIM2 or NLRC4
are less developed but represent a future direction. For instance,
pristimerin has been shown to inhibit AIM2 and ameliorate
tendonopathy in mice (137).
Direct caspase inhibition provides a strategy to
block pyroptosis execution.
VX-765 is an orally bioavailable, reversible and
selective caspase-1 inhibitor; it is a prodrug that is hydrolyzed
in vivo to its active form, VRT-043198 (159). By inhibiting caspase-1, VX-765
reduces the production of IL-1β and IL-18, thereby mitigating
inflammatory tissue damage. Li et al (138) demonstrated that VX-765
attenuated plaque formation and inflammation in an atherosclerosis
model. In a vascular calcification model, it alleviated VSMC
calcification via suppression of STAT3 signaling (139). Moreover, VX-765 protects
against acute myocardial infarction by mitigating pyroptosis,
hypoxia-induced cardiomyocyte injury, oxidative stress, apoptosis
and inflammation (140). These
findings suggest its potential benefit in AA-related vascular
remodeling. Notably, VX-765 has been evaluated in clinical trials
for psoriasis (NCT00205465) and epilepsy (NCT01048255; EudraCT
2011-004156-19), although its long-term use may raise safety
concerns.
Other caspase-1 inhibitors also represent promising
therapeutic candidates. For instance, Ac-YVAD-CHO is a
peptide-based irreversible caspase-1 inhibitor widely used in basic
research; it reduces the plasma levels of the caspase-1-dependent
cytokines IL-1β and IL-18 in endotoxemic rats. Inhalation of
Ac-YVAD-CHO concurrently downregulates inducible nitric oxide
synthase gene expression in alveolar macrophages and
cyclooxygenase-2 expression in lung tissues, supporting the
potential of inhaled caspase inhibitors for treating inflammatory
diseases (141). However, owing
to its peptide nature, Ac-YVAD-CHO has poor pharmacokinetic
properties and is unsuitable for systemic administration, although
it serves as a valuable lead compound for inhibitor design.
Targeting caspases involved in the non-canonical
pyroptosis pathway is an emerging area of research. Several
molecules have been reported to directly bind and inhibit
caspase-4/11. For instance, oxidized
1-palmitoyl-2-arachidonyl-phosphatidylcholine binds directly to
caspase-4/11, competing with LPS to suppress pyroptosis, IL-1β
release and septic shock (142). However, its potential as a drug
candidate remains limited. Serine protease inhibitor B1 (SERPINB1)
functions as a critical endogenous inhibitor of pyroptosis; its
suppression promotes spontaneous activation of caspase-1/4/5/11,
while its presence helps minimize inflammatory damage (160). Specifically, SERPINB1 restricts
caspase activity by inhibiting CARD oligomerization and enzymatic
activation. Choi et al (160) demonstrated that its C-terminal
CARD-binding motif suppresses the activation of
pro-caspase-1/4/5/10. Furthermore, a study has indicated that
SERPINB1 attenuates pathological cardiac hypertrophy and remodeling
through its antipyroptotic effects (143). These findings offer a rationale
for the development of novel inhibitors that mimic this
mechanism.
Additional inhibitors targeting caspases implicated
in pyroptosis include Ac-DEVD-CHO, caspase-6b and Z-IETD-FMK.
Ac-DEVD-CHO is a specific caspase-3 inhibitor that
suppresses endothelial apoptosis and promotes cerebrovascular
spasms. Zhou et al (161) demonstrated that selective
caspase-3 inhibition blocks downstream signaling of caspase-8, even
when caspase-8 is activated within the caspase cascade. Caspase-6b,
an isoform generated by alternative splicing of the
melanin-concentrating hormone receptor 2 gene, acts as a natural
caspase-6-specific inhibitor; it selectively prevents caspase-6
autoactivation without affecting its preactivated form (162).
Z-IETD-FMK is a broad-spectrum, irreversible
caspase inhibitor commonly used in scientific research; it
mitigates the interplay between NLRP3 inflammasome activation,
pyroptosis and apoptosis, thereby conferring protection against
oxidative stress-induced lung injury (163). In a CaCl2-induced
mouse AAA model, intraperitoneal administration of Z-IETD-FMK
reduced the incidence of AAAs. This protective effect is attributed
to the role of the CD95/CD95L system in recruiting inflammatory
cells to injury sites and promoting the phenotypic switching of
aortic smooth muscle cells via caspase-8 signaling, processes that
are involved in AAA pathogenesis (66). However, owing to its lack of
selectivity and associated toxicity, Z-IETD-FMK is unsuitable for
clinical use.
Directly inhibiting GSDMD pore formation is
considered a highly specific strategy that potentially blocks
pathological inflammation while sparing upstream immune
surveillance.
These inhibitors function by covalently modifying
specific cysteine residues on GSDMD, thereby blocking its
oligomerization and pore formation.
Necrosulfonamide (NSA) covalently binds to GSDMD
(Cys191/Cys192 in humans/mice) to inhibit oligomerization and pore
formation, thereby inhibiting inflammasome-dependent pyroptosis
(164). A distinctive feature
of NSA is its ability to directly target GSDMD while preserving
other cell death pathways, such as those involved in TLR
signaling-mediated cell death (165). However, studies indicate that
it also impedes inflammasome priming and caspase-1 activation
(166-168). Owing to its extreme
hydrophobicity and immunocyte toxicity, NSA exhibits unfavorable
pharmacokinetics in vivo. To address this limitation,
Boersma et al (169)
developed a porous nanoparticle-based delivery system for targeted
NSA delivery to phagocytes, improving its targeting and safety.
Recent evidence has demonstrated that NSA ameliorates Ang
II-induced AAA progression in ApoE−/− mice (65).
Disulfiram (DSF), a Food and Drug
Administration-approved drug for alcohol dependence, is metabolized
in vivo to S-methyl-N, N-diethylthiocarbamate sulfoxide,
which covalently modifies GSDMD at Cys191/Cys192, effectively
inhibiting pore formation (170). DSF significantly reduces IL-1β
secretion from activated macrophages and attenuates systemic
inflammatory responses (171).
Liao et al (54) reported
that oral DSF administration in Ang II-infused mice reduces AAA
incidence while ameliorating collagen deposition and elastin
degradation within the aortic wall. The repurposing potential of
DSF supports its rapid clinical translation. However, its
multitarget nature, including inhibition of aldehyde dehydrogenase,
may lead to off-target effects, necessitating careful dose and
indication exploration.
Dimethyl fumarate (DMF), a drug used for multiple
sclerosis, inhibits pyroptosis by succinylating GSDMD at Cys192,
thereby blocking its processing and oligomerization (172). Wang et al (144) reported that DMF restored v-src
sarcoma/focal adhesion kinase signaling and collagen transport in
aortic VSMCs, and mitigated aortic injury in mouse aneurysm and
dissection models. As an approved drug, DMF has a well-established
safety profile, facilitating its translational research in AAs.
Additional non-covalent inhibitors and natural
compounds suppress pyroptosis by blocking GSDMD pore formation.
C202-2729, a small molecule identified through high-throughput
screening, non-covalently binds to the GSDMD N-terminal domain,
preventing its membrane association and pore formation. C202-2729
has demonstrated efficacy in a sepsis model (146). Caffeic acid, a natural phenolic
compound found in coffee and various fruits and vegetables,
inhibits lipoxygenase activity and has antioxidant and
anti-inflammatory effects (175); it has been shown to bind GSDMD
and inhibit its cleavage by caspase-1, thereby reducing pyroptosis
(147).
The two strategies targeting GSDMD possess distinct
pharmacological profiles and clinical applications (176). Covalent inhibitors irreversibly
modify active site cysteines to block pore formation, offering
potent and sustained inhibition suitable for acute inflammatory
conditions (165,177). However, their broad-spectrum
electrophilic nature confers off-target effects, and irreversible
modification may increase their immunogenicity (176,178). Non-covalent inhibitors
reversibly bind the GSDMD N-terminal domain through conformational
complementarity, theoretically providing higher selectivity and
lower off-target toxicity, making them preferable for chronic
disease intervention (179,180). However, their efficacy is
highly dependent on target conformational stability;
activation-induced conformational rearrangements may diminish
binding affinity, posing challenges for in vivo persistence
(181).
From a translational perspective, strategy
selection should align with therapeutic windows. Acute progressive
stages in AAs warrant covalent inhibitors with targeted delivery to
mitigate off-target effects. Chronic settings favor non-covalent
inhibitors with optimized pharmacokinetics through medicinal
chemistry. Future directions include integrating both approaches,
namely, the development of reversible covalent modifiers or
non-covalent inhibitors targeting intermediate GSDMD
conformations.
Given that the detrimental effects of pyroptosis
largely result from the release of IL-1β and IL-18, directly
neutralizing these cytokines represents another effective
therapeutic strategy.
Anakinra, a recombinant IL-1 receptor antagonist,
blocks the signaling of both IL-1α and IL-1β; its efficacy has been
demonstrated in macrophage activation syndrome with markedly
elevated IL-18 levels, and it is already used to treat
autoinflammatory diseases (148). Canakinumab is a humanized
monoclonal antibody against IL-1β. In a randomized trial involving
postmyocardial infarction patients conducted by Ridker et al
(149), canakinumab markedly
reduced cardiovascular events, providing strong clinical evidence
for the role of IL-1β in human vascular disease. Additionally, the
IL-1 inhibitor goflikicept notably reduced recurrence rates in
idiopathic recurrent pericarditis and improved patient quality of
life (150). These findings
indirectly support the therapeutic potential of IL-1-targeting
agents in AAs. IL-18 binding protein, a natural inhibitor of IL-18,
is also under development as a recombinant therapeutic.
Each of the aforementioned strategies presents
distinct advantages and limitations. Upstream inhibitors (for
example, MCC950) may broadly suppress multiple downstream effects
but risk interfering with essential host defense mechanisms.
Downstream cytokine inhibitors (for example, canakinumab) have
well-defined targets but are often costly and require parenteral
administration. Direct targeting of GSDMD is regarded as a strategy
to precisely 'defuse the bomb', blocking pathological lytic cell
death and inflammatory release while potentially preserving
upstream immune sensing functions. This approach has thus become a
major focus in contemporary medicinal chemistry.
Targeting pyroptosis in AAs presents translational
potential, yet several challenges remain. The predominant focus on
GSDMD has also left other GSDM family members largely unexplored.
Furthermore, the extensive crosstalk among pyroptosis, apoptosis
and necroptosis is poorly understood; inhibiting one cell death
pathway may trigger compensatory activation of another. The
relative contributions of pyroptosis in VSMCs, macrophages and ECs
to AA progression also require clarification to enable
cell-specific targeting. Finally, the specificity and long-term
safety of pyroptosis inhibitors, particularly the risk of
immunosuppression, warrant careful evaluation.
Current research is limited by an over-reliance on
murine models and a paucity of human data for validation.
Throughout this review, findings supported by direct evidence from
human aneurysmal tissues have been distinguished from those
inferred solely from animal studies. For instance, while GSDMD
upregulation in macrophages within human AA tissues has been
corroborated by multiple studies, the mechanism by which GSDMB
regulates macrophage pyroptosis via the mtROS-Drp1-caspase-4 axis
is currently largely determined through cellular experiments and
murine models, with limited direct confirmation in human tissues.
Similarly, although preliminary evidence has shown that
GSDME-mediated apoptosis-to-pyroptosis switching occurs in both
human and murine specimens, its upstream regulatory networks are
derived predominantly from mouse genetic studies. The vast majority
of mechanistic studies have employed Ang II-infused murine AAA
models. While these models effectively recapitulate the
inflammatory infiltration and VSMC loss observed in human AA, their
disease progression timeline (days to weeks) fundamentally differs
from that of the chronic, insidious course of human AAs (years to
decades). Moreover, murine vascular wall biomechanics and immune
microenvironments differ substantially from those of humans.
Critically, intrinsic species differences in pyroptosis pathways
must be acknowledged: In humans, non-canonical pyroptosis is
mediated by caspase-4/5, whereas in mice, it relies on caspase-11;
moreover, the tissue distribution and regulatory mechanisms of GSDM
family members also exhibit interspecies variation. These
differences suggest that the efficacy of pyroptosis inhibitors
identified through murine screens (such as compounds targeting
caspase-11) may show attenuated efficacy in humans due to the
structural divergence in target proteins, and that the cell
type-specific pyroptosis phenotypes observed in mice may not
faithfully recapitulate human pathology. Therefore, future
investigations must explicitly acknowledge the inferential nature
of animal-derived data and incorporate human tissue validation as
an essential component of mechanistic studies.
Future efforts should focus on the following: i)
Deepening mechanistic insight. Single-cell and spatial
transcriptomics of human AA specimens can be used to map all GSDMs
and identify novel targets and subtypes. Proteomic approaches may
identify specific pyroptosis-related biomarkers (for example,
circulating GSDMD-NT) for diagnosis and monitoring. ii) Innovative
drug development. Structure-based design should aim to develop more
potent and selective GSDM inhibitors, such as non-covalent
compounds or proteolysis-targeting chimeras. Chemical optimization
of existing inhibitors (such as NSA) can improve pharmacokinetics,
whereas nanoparticle-based or microenvironment-responsive delivery
systems may enhance targeting and reduce systemic exposure. iii)
Exploring combination therapies. Combining pyroptosis inhibitors
with existing treatments (such as Ang II receptor blockers) may
yield synergistic benefits. iv) Advancing clinical translation. The
efficacy and safety of pyroptosis-targeting strategies should be
rigorously evaluated in large animal models (for example, porcine
or canine aneurysm models), whose vascular dimensions, hemodynamics
and immune backgrounds more closely approximate human conditions.
Concurrently, proof-of-concept clinical trials for repurposed drugs
(such as DSF and DMF) incorporating pyroptosis biomarkers for
patient stratification should be conducted, paving the way for
precision medicine approaches.
Pyroptosis has firmly established its core role in
the pathogenesis of AAs. By directly mediating the lytic death of
VSMCs and initiating a powerful, self-amplifying inflammatory
response, it acts as a key pathological engine driving AA
initiation and progression. Multiple nodes in this signaling
pathway, from the NLRP3 inflammasome to the final GSDMD pore, have
been validated as effective intervention targets in preclinical
models.
The present review, while summarizing existing
knowledge, critically highlights the limited understanding of GSDM
members beyond GSDMD, non-canonical pathways and complex
intercellular cross-talk. From a medicinal chemistry perspective,
various strategies have been systematically evaluated, with direct
inhibition of the terminal executor GSDMD (for example, by DSF and
DMF) emerging as a particularly attractive and precise therapeutic
direction.
However, translating these promising targets into
clinical reality faces hurdles, including understanding human
disease heterogeneity, ensuring inhibitor specificity and safety,
and managing long-term risks. Future research, integrating
multiomics technologies, structural biology, nanotechnology and
intelligent drug design, holds the promise of developing safer and
more effective pyroptosis-targeted therapies. Ultimately, through
well-designed clinical trials, these novel agents, either alone or
in combination with existing strategies, may enable a paradigm
shift from purely surgical repair to active pharmacological
intervention, potentially improving the prognosis and reducing the
societal burden of this devastating disease.
Not applicable.
JM drafted the original manuscript. ML
conceptualized and designed the paper, provided the methodology,
and reviewed and edited the manuscript. JM and ML used Figdraw.com and Biorender.com to create the figures. LR and CZ
proposed the study concept, provided funding support, contributed
to the interpretation of the literature and were involved in
revising the manuscript critically for important intellectual
content. All authors have read and approved the final manuscript.
Data authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present study was supported by the National Natural Science
Foundation of China (grant no. 82571792), the Major Technology
Innovation of Hubei Province (grant no. 2022BCA001), the National
Key Research and Development Program of China (grant no.
2020YFC2008000), the National Natural Science Foundation Regional
Innovation and Development Joint Fund (grant no. U24A20741) and the
Zhang Cuntai Expert Workstation of Yunnan Province (grant no.
202405AF140057).
|
1
|
Gong W, Tian Y and Li L: T cells in
abdominal aortic aneurysm: Immunomodulation and clinical
application. Front Immunol. 14:12401322023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Talvitie M, Stenman M, Roy J, Leander K
and Hultgren R: Sex differences in rupture risk and mortality in
untreated patients with intact abdominal aortic aneurysms. J Am
Heart Assoc. 10:e0195922021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sidloff D, Stather P, Dattani N, Bown M,
Thompson J, Sayers R and Choke E: Aneurysm global epidemiology
study: Public health measures can further reduce abdominal aortic
aneurysm mortality. Circulation. 129:747–753. 2014. View Article : Google Scholar
|
|
4
|
Baman JR and Eskandari MK: What is an
abdominal aortic aneurysm? JAMA. 328:22802022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chakraborty A, Li Y, Zhang C, Li Y,
LeMaire SA and Shen YH: Programmed cell death in aortic aneurysm
and dissection: A potential therapeutic target. J Mol Cell Cardiol.
163:67–80. 2022. View Article : Google Scholar :
|
|
6
|
Luo S, Kong C, Zhao S, Tang X, Wang Y,
Zhou X, Li R, Liu X, Tang X, Sun S, et al: Endothelial
HDAC1-ZEB2-NuRD complex drives aortic aneurysm and dissection
through regulation of protein S-sulfhydration. Circulation.
147:1382–1403. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tilson MD: Decline of the atherogenic
theory of the etiology of the abdominal aortic aneurysm and rise of
the autoimmune hypothesis. J Vasc Surg. 64:1523–1525. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sun P, Zhang L, Gu Y, Wei S, Wang Z, Li M,
Wang W, Wang Z and Bai H: Immune checkpoint programmed death-1
mediates abdominal aortic aneurysm and pseudoaneurysm progression.
Biomed Pharmacother. 142:1119552021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hsieh CC, Yen MH, Yen CH and Lau YT:
Oxidized low density lipoprotein induces apoptosis via generation
of reactive oxygen species in vascular smooth muscle cells.
Cardiovasc Res. 49:135–145. 2001. View Article : Google Scholar
|
|
10
|
Golledge J: Abdominal aortic aneurysm:
Update on pathogenesis and medical treatments. Nat Rev Cardiol.
16:225–242. 2019. View Article : Google Scholar
|
|
11
|
Ding YN, Wang TT, Lv SJ, Tang X, Wei ZY,
Yao F, Xu HS, Chen YN, Wang XM, Wang HY, et al: SIRT6 is an
epigenetic repressor of thoracic aortic aneurysms via inhibiting
inflammation and senescence. Signal Transduct Target Ther.
8:2552023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yuan J and Ofengeim D: A guide to cell
death pathways. Nat Rev Mol Cell Biol. 25:379–395. 2024. View Article : Google Scholar
|
|
13
|
Pandey A, Li Z, Gautam M, Ghosh A and Man
SM: Molecular mechanisms of emerging inflammasome complexes and
their activation and signaling in inflammation and pyroptosis.
Immunol Rev. 329:e134062025. View Article : Google Scholar :
|
|
14
|
Shi J, Gao W and Shao F: Pyroptosis:
Gasdermin-mediated programmed necrotic cell death. Trends Biochem
Sci. 42:245–254. 2017. View Article : Google Scholar
|
|
15
|
Vasudevan SO, Behl B and Rathinam VA:
Pyroptosis-induced inflammation and tissue damage. Semin Immunol.
69:1017812023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Elias EE, Lyons B and Muruve DA:
Gasdermins and pyroptosis in the kidney. Nat Rev Nephrol.
19:337–350. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ding J, Wang K, Liu W, She Y, Sun Q, Shi
J, Sun H, Wang DC and Shao F: Pore-forming activity and structural
autoinhibition of the gasdermin family. Nature. 535:111–116. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rühl S, Shkarina K, Demarco B, Heilig R,
Santos JC and Broz P: ESCRT-dependent membrane repair negatively
regulates pyroptosis downstream of GSDMD activation. Science.
362:956–960. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yu P, Zhang X, Liu N, Tang L, Peng C and
Chen X: Pyroptosis: Mechanisms and diseases. Signal Transduct
Target Ther. 6:1282021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fang Y, Tian S, Pan Y, Li W, Wang Q, Tang
Y, Yu T, Wu X, Shi Y, Ma P and Shu Y: Pyroptosis: A new frontier in
cancer. Biomed Pharmacother. 121:1095952020. View Article : Google Scholar
|
|
21
|
Vanaja SK, Rathinam VA and Fitzgerald KA:
Mechanisms of inflammasome activation: Recent advances and novel
insights. Trends Cell Biol. 25:308–315. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shi J, Zhao Y, Wang K, Shi X, Wang Y,
Huang H, Zhuang Y, Cai T, Wang F and Shao F: Cleavage of GSDMD by
inflammatory caspases determines pyroptotic cell death. Nature.
526:660–665. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xia X, Wang X, Cheng Z, Qin W, Lei L,
Jiang J and Hu J: The role of pyroptosis in cancer: Pro-cancer or
pro-'host'? Cell Death Dis. 10:6502019. View Article : Google Scholar
|
|
24
|
Shi X, Sun Q, Hou Y, Zeng H, Cao Y, Dong
M, Ding J and Shao F: Recognition and maturation of IL-18 by
caspase-4 non-canonical inflammasome. Nature. 624:442–450. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lamkanfi M, Sarkar A, Vande Walle L,
Vitari AC, Amer AO, Wewers MD, Tracey KJ, Kanneganti TD and Dixit
VM: Inflammasome-dependent release of the alarmin HMGB1 in
endotoxemia. J Immunol. 185:4385–4392. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Man SM, Karki R and Kanneganti TD:
Molecular mechanisms and functions of pyroptosis, inflammatory
caspases and inflammasomes in infectious diseases. Immunol Rev.
277:61–75. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wei Y, Lan B, Zheng T, Yang L, Zhang X,
Cheng L, Tuerhongjiang G, Yuan Z and Wu Y: GSDME-mediated
pyroptosis promotes the progression and associated inflammation of
atherosclerosis. Nat Commun. 14:9292023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wei Y, Yang L, Pandeya A, Cui J, Zhang Y
and Li Z: Pyroptosis-induced inflammation and tissue damage. J Mol
Biol. 434:1673012022. View Article : Google Scholar
|
|
29
|
Liu Z, Wang C, Yang J, Zhou B, Yang R,
Ramachandran R, Abbott DW and Xiao TS: Crystal structures of the
Full-length murine and human gasdermin D reveal mechanisms of
autoinhibition, lipid binding, and oligomerization. Immunity.
51:43–49.e4. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Burdette BE, Esparza AN, Zhu H and Wang S:
Gasdermin D in pyroptosis. Acta Pharm Sin B. 11:2768–2782. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang K, Sun Q, Zhong X, Zeng M, Zeng H,
Shi X, Li Z, Wang Y, Zhao Q, Shao F and Ding J: Structural
mechanism for GSDMD targeting by autoprocessed caspases in
pyroptosis. Cell. 180:941–955.e20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Coll RC, Schroder K and Pelegrín P: NLRP3
and pyroptosis blockers for treating inflammatory diseases. Trends
Pharmacol Sci. 43:653–668. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Saeki N, Usui T, Aoyagi K, Kim DH, Sato M,
Mabuchi T, Yanagihara K, Ogawa K, Sakamoto H, Yoshida T and Sasaki
H: Distinctive expression and function of four GSDM family genes
(GSDMA-D) in normal and malignant upper gastrointestinal
epithelium. Genes Chromosomes Cancer. 48:261–271. 2009. View Article : Google Scholar
|
|
34
|
Cadena C, Kornfeld OS, Lee BL and Kayagaki
N: Epigenetic and transcriptional control of gasdermins. Semin
Immunol. 70:1018412023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang Y, Gao W, Shi X, Ding J, Liu W, He H,
Wang K and Shao F: Chemotherapy drugs induce pyroptosis through
caspase-3 cleavage of a gasdermin. Nature. 547:99–103. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jiang H, Deng L, Lin Z, Yang K, Yang J,
Zhao W and Gong W: GSDMB interacts with IGF2BP1 to suppress
colorectal cancer progression by modulating DUSP6-ERK pathway. In
Immunopharmacol. 143:1132802024. View Article : Google Scholar
|
|
37
|
Zhou Z, He H, Wang K, Shi X, Wang Y, Su Y,
Wang Y, Li D, Liu W and Zhang Y: Granzyme A from cytotoxic
lymphocytes cleaves GSDMB to trigger pyroptosis in target cells.
Science. 368:eaaz75482020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu X, Xia S, Zhang Z, Wu H and Lieberman
J: Channelling inflammation: Gasdermins in physiology and disease.
Nat Rev Drug Discov. 20:384–405. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liston A and Masters SL:
Homeostasis-altering molecular processes as mechanisms of
inflammasome activation. Nat Rev Immunol. 17:208–214. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zheng D, Liwinski T and Elinav E:
Inflammasome activation and regulation: Toward a better
understanding of complex mechanisms. Cell Discov. 6:362020.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Franchi L, Warner N, Viani K and Nuñez G:
Function of Nod-like receptors in microbial recognition and host
defense. Immunol Rev. 227:106–128. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wei X, Xie F, Zhou X, Wu Y, Yan H, Liu T,
Huang J, Wang F, Zhou F and Zhang L: Role of pyroptosis in
inflammation and cancer. Cell Mol Immunol. 19:971–992. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kayagaki N, Stowe IB, Lee BL, O'Rourke K,
Anderson K, Warming S, Cuellar T, Haley B, Roose-Girma M, Phung QT,
et al: Caspase-11 cleaves gasdermin D for non-canonical
inflammasome signalling. Nature. 526:666–671. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wang Y, Zhang Y, Gao X, Qian J, Yang J,
Sun W, Wang H and Yang Y: Resistin-like molecule beta augments
phenotypic modulation of human aortic smooth muscle cell triggered
by high glucose. Endocr J. 68:461–468. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Aizawa E, Karasawa T, Watanabe S, Komada
T, Kimura H, Kamata R, Ito H, Hishida E, Yamada N, Kasahara T, et
al: GSDME-Dependent Incomplete Pyroptosis Permits Selective IL-1α
Release under Caspase-1 Inhibition. iScience. 23:1010702020.
View Article : Google Scholar
|
|
46
|
Sarhan J, Liu BC, Muendlein HI, Li P,
Nilson R, Tang AY, Rongvaux A, Bunnell SC, Shao F, Green DR and
Poltorak A: Caspase-8 induces cleavage of gasdermin D to elicit
pyroptosis during Yersinia infection. Proc Natl Acad Sci USA.
115:E10888–E10897. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Hou J, Zhao R, Xia W, Chang CW, You Y, Hsu
JM, Nie L, Chen Y, Wang YC, Liu C, et al: PD-L1-mediated gasdermin
C expression switches apoptosis to pyroptosis in cancer cells and
facilitates tumour necrosis. Nat Cell Biol. 22:1264–1275. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Cigalotto L and Martinvalet D: Granzymes
in health and diseases: The good, the bad and the ugly. Front
Immunol. 15:13717432024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chowdhury D and Lieberman J: Death by a
thousand cuts: Granzyme pathways of programmed cell death. Annu Rev
Immunol. 26:389–420. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Li Z, Ma R, Tang H, Guo J, Shah Z, Zhang
J, Liu N, Cao S, Marcucci G, Artis D, et al: Therapeutic
application of human type 2 innate lymphoid cells via induction of
granzyme B-mediated tumor cell death. Cell. 187:624–641.e23. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Long Y, Jia X and Chu L: Insight into the
structure, function and the tumor suppression effect of gasdermin
E. Biochem Pharmacol. 226:1163482024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Rao Z, Zhu Y, Yang P, Chen Z, Xia Y, Qiao
C, Liu W, Deng H, Li J, Ning P and Wang Z: Pyroptosis in
inflammatory diseases and cancer. Theranostics. 12:4310–4329. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wang J, Ye W, Zou J, Yang P, Jin M, Zheng
Z, Zhou C, Qiu W, Lu J, Li C, et al: Targeting the smooth muscle
cell Keap1-Nrf2-GSDMD-pyroptosis axis by cryptotanshinone prevents
abdominal aortic aneurysm formation. Theranostics. 14:6516–6542.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Liao F, Wang L, Wu Z, Luo G, Qian Y, He X,
Ding S and Pu J: Disulfiram protects against abdominal aortic
aneurysm by ameliorating vascular smooth muscle cells pyroptosis.
Cardiovasc Drugs Ther. 37:1–14. 2023. View Article : Google Scholar
|
|
55
|
Liu S, Xue YJ, Yin RP, Wu BS, Yu YW, Zhou
YY, Wang J and Ji KT: 3,4-Benzopyrene (Bap) aggravated abdominal
aortic aneurysm formation by targeting pyroptosis in smooth muscle
cells through ET-1 mediated NLRP3-inflammasome activation. Int
Immunopharmacol. 124:1108512023. View Article : Google Scholar
|
|
56
|
Hu J, Xu J, Zhao J, Liu Y, Huang R, Yao D,
Xie J and Lei Y: Colchicine ameliorates short-term abdominal aortic
aneurysms by inhibiting the expression of NLRP3 inflammasome
components in mice. Eur J Pharmacol. 964:1762972024. View Article : Google Scholar
|
|
57
|
Usui F, Shirasuna K, Kimura H, Tatsumi K,
Kawashima A, Karasawa T, Yoshimura K, Aoki H, Tsutsui H, Noda T, et
al: Inflammasome activation by mitochondrial oxidative stress in
macrophages leads to the development of angiotensin II-induced
aortic aneurysm. Arterioscler Thromb Vasc Biol. 35:127–136. 2015.
View Article : Google Scholar
|
|
58
|
Wu D, Ren P, Zheng Y, Zhang L, Xu G, Xie
W, Lloyd EE, Zhang S, Zhang Q, Curci JA, et al: NLRP3 (Nucleotide
oligomerization domain-like receptor family, pyrin domain
containing 3)-Caspase-1 inflammasome degrades contractile proteins:
Implications for aortic biomechanical dysfunction and aneurysm and
dissection formation. Arterioscler Thromb Vasc Biol. 37:694–706.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Dihlmann S, Erhart P, Mehrabi A,
Nickkholgh A, Lasitschka F, Böckler D and Hakimi M: Increased
expression and activation of absent in melanoma 2 inflammasome
components in lymphocytic infiltrates of abdominal aortic
aneurysms. Mol Med. 20:230–237. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang JH, Zhang XL, Liu XQ, Chen HJ, Wang
JF and Ji M: M1 Macrophage-derived exosome LncRNA PVT1 promotes
inflammation and pyroptosis of vascular smooth muscle cells in
abdominal aortic aneurysm by inhibiting miR-186-5p and regulating
HMGB1. Cardiovasc Toxicol. 24:302–320. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Cai H, Li H, Xiao X, Wang S, Liu R, Qin Y,
Zhou Y and Yao C: TRAF6 promotes abdominal aortic aneurysm
development by activating macrophage pyroptosis via the
NLRP3/Caspase1/GSDMD pathway. Faseb J. 39:e703182025. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Fu H, Shen QR, Zhao Y, Ni M, Zhou CC, Chen
JK, Chi C, Li DJ, Liang G and Shen FM: Activating α7nAChR
ameliorates abdominal aortic aneurysm through inhibiting pyroptosis
mediated by NLRP3 inflammasome. Acta Pharmacol Sin. 43:2585–2595.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Hu J, Jiang Y, Wu X, Wu Z, Qin J, Zhao Z,
Li B, Xu Z, Lu X, Wang X and Liu X: Exosomal miR-17-5p from
adipose-derived mesenchymal stem cells inhibits abdominal aortic
aneurysm by suppressing TXNIP-NLRP3 inflammasome. Stem Cell Res
Ther. 13:3492022. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Xie X, Shen X, Liu Y, Zuo Y, Wang S, Zhou
Y, Li X, Wang K, Li B and Wang Z: GSDMB involvement in the
pathogenesis of abdominal aortic aneurysm through regulation of
macrophage non-canonical pyroptosis. Arch Biochem Biophys.
759:1101022024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Guo J, Zhang Q, Li Z, Qin M, Shi J, Wang
Y, Ai W, Ju J, Samura M, Tsao PS and Xu B: Gasdermin D inhibitor
necrosulfonamide alleviates angiotensin II-Induced abdominal aortic
aneurysms in apolipoprotein E-Deficient mice. Biomolecules.
14:7262024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Liu Z, Fitzgerald M, Meisinger T, Batra R,
Suh M, Greene H, Penrice AJ, Sun L, Baxter BT and Xiong W:
CD95-ligand contributes to abdominal aortic aneurysm progression by
modulating inflammation. Cardiovasc Res. 115:807–818. 2019.
View Article : Google Scholar
|
|
67
|
Chi Y, Huang T, Zhao J, Huang Y, Zhou Z,
Xing B, Qu D, Sun J, Fu Y, Yu L, et al: Genistein ameliorates
thoracic aortic dissection by inhibiting CB1 receptor
hyperactivation and modulating its-mediated cAMP-PKA signaling.
Phytomedicine. 147:1571782025. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Huang Y, Xie X, Huang G, Hong X, Lu W, Fu
W and Wang L: CXCL8 upregulation mediates inflammatory cell
infiltration and accelerates abdominal aortic aneurysm progression.
Sci Prog. 108:3685042513287542025. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Xiong Y, Liu S, Liu Y, Zhao J, Sun J, Li
Y, Pan B and Wang W: PI3Kγ promotes neutrophil extracellular trap
formation by non-canonical pyroptosis in abdominal aortic aneurysm.
JCI Insight. 9:e1832372024.
|
|
70
|
Cai H, Huang L, Wang M, Liu R, Qiu J, Qin
Y, Yao X, Wang S, Yao C, Hu Z and Zhou Y: Pterostilbene alleviates
abdominal aortic aneurysm via inhibiting macrophage pyroptosis by
activating the miR-146a-5p/TRAF6 axis. Food Funct. 15:139–157.
2024. View Article : Google Scholar
|
|
71
|
Yao Y, Cao Y, Xu Y, Chen G, Liu Y, Jiang
H, Fan R, Qin W, Wang X, Chai H, et al: CARMA3 deficiency
aggravates angiotensin II-Induced abdominal aortic aneurysm
development interacting between endoplasmic reticulum and
mitochondria. Can J Cardiol. 39:1449–1462. 2023.PubMed/NCBI
|
|
72
|
Sun L, Li X, Luo Z, Li M, Liu H, Zhu Z,
Wang J, Lu P, Wang L, Yang C, et al: Purinergic receptor P2X7
contributes to abdominal aortic aneurysm development via modulating
macrophage pyroptosis and inflammation. Transl Res. 258:72–85.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Xiong JM, Liu H, Chen J, Zou QQ, Wang YY
and Bi GS: Curcumin nicotinate suppresses abdominal aortic aneurysm
pyroptosis via lncRNA PVT1/miR-26a/KLF4 axis through regulating the
PI3K/AKT signaling pathway. Toxicol Res (Camb). 10:651–661. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Gao J, Chen Y, Wang H, Li X, Li K, Xu Y,
Xie X, Guo Y, Yang N, Zhang X, et al: Gasdermin D deficiency in
vascular smooth muscle cells ameliorates abdominal aortic aneurysm
through reducing putrescine synthesis. Adv Sci (Weinh).
10:e22040382023. View Article : Google Scholar :
|
|
75
|
Ren P, Wu D, Appel R, Zhang L, Zhang C,
Luo W, Robertson AAB, Cooper MA, Coselli JS, Milewicz DM, et al:
Targeting the NLRP3 inflammasome with inhibitor MCC950 prevents
aortic aneurysms and dissections in mice. J Am Heart Assoc.
9:e0140442020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Wakita D, Kurashima Y, Crother TR, Noval
Rivas M, Lee Y, Chen S, Fury W, Bai Y, Wagner S, Li D, et al: Role
of Interleukin-1 signaling in a mouse model of kawasaki
Disease-associated abdominal aortic aneurysm. Arterioscler Thromb
Vasc Biol. 36:886–897. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Xu XD, Chen JX, Zhu L, Xu ST, Jiang J and
Ren K: The emerging role of pyroptosis-related inflammasome pathway
in atherosclerosis. Mol Med. 28:1602022. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Zheng K, Wang S, Deng M, Luo Y, Li W, Zeng
L and Wang Y: Mechanisms and therapeutic strategies of macrophage
polarization in intervertebral disc degeneration. JOR Spine.
8:e700652025. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Hsieh PC, Wu YK, Yang MC, Su WL, Kuo CY
and Lan CC: Deciphering the role of damage-associated molecular
patterns and inflammatory responses in acute lung injury. Life Sci.
305:1207822022. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Roberts E, Charras A, Hahn G and Hedrich
CM: An improved understanding of pediatric chronic nonbacterial
osteomyelitis pathophysiology informs current and future treatment.
J Bone Miner Res. 39:1523–1538. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Habotta OA, Abdeen A, El-Hanafy AA, Yassin
N, Elgameel D, Ibrahim SF, Abdelrahaman D, Hasan T, Imbrea F,
Ghamry HI, et al: Sesquiterpene nootkatone counteracted the
melamine-induced neurotoxicity via repressing of oxidative stress,
inflammatory, and apoptotic trajectories. Biomed Pharmacother.
165:1151332023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Yang G, Yang Y, Liu Y and Liu X:
Regulation of alveolar macrophage death in pulmonary fibrosis: A
review. Apoptosis. 28:1505–1519. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zheng J, Yi Y, Tian T, Luo S, Liang X and
Bai Y: ICI-induced cardiovascular toxicity: Mechanisms and immune
reprogramming therapeutic strategies. Front Immunol.
16:15504002025. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Drożdżal S, Gomółka A, Opara-Bajerowicz M,
Lisak M, Sielicka U, Bąk K, Przybyciński J and Feret-Adrabińska W:
Neutrophil extracellular traps in Anti-neutrophil cytoplasmic
Antibody-associated vasculitis: Diagnostic and clinical
Significance-A review of the current literature. J Clin Med.
14:36392025. View Article : Google Scholar
|
|
85
|
Zhang J, Zhang X, Liu X, Chen H, Wang J
and Ji M: M1 Macrophage-derived exosome LncRNA PVT1 promotes
inflammation and pyroptosis of vascular smooth muscle cells in
abdominal aortic aneurysm by inhibiting miR-186-5p and Regulating
HMGB1. Cardiovasc Toxicol. 24:302–320. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Ye B, Fan X, Fang Z, Mao C, Lin L, Wu J,
Zheng W, Cai X, Huang W, Lv Y, et al: Macrophage-derived GSDMD
promotes abdominal aortic aneurysm and aortic smooth muscle cells
pyroptosis. Int Immunopharmacol. 128:1115542024. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Sun J, Zhu Q, Yu X, Liang X, Guan H, Zhao
H and Yao W: RhoGDI3 at the trans-Golgi network participates in
NLRP3 inflammasome activation, VSMC phenotypic modulation, and
neointima formation. Atherosclerosis. 387:1173912023. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Ren XS, Tong Y, Ling L, Chen D, Sun HJ,
Zhou H, Qi XH, Chen Q, Li YH, Kang YM and Zhu GQ: NLRP3 gene
deletion attenuates angiotensin II-Induced phenotypic
transformation of vascular smooth muscle cells and vascular
remodeling. Cell Physiol Biochem. 44:2269–2280. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Sun HJ, Ren XS, Xiong XQ, Chen YZ, Zhao
MX, Wang JJ, Zhou YB, Han Y, Chen Q, Li YH, et al: NLRP3
inflammasome activation contributes to VSMC phenotypic
transformation and proliferation in hypertension. Cell Death Dis.
8:e30742017. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Sun W, Pang Y, Liu Z, Sun L, Liu B, Xu M,
Dong Y, Feng J, Jiang C, Kong W and Wang X: Macrophage inflammasome
mediates hyperhomocysteinemia-aggravated abdominal aortic aneurysm.
J Mol Cell Cardiol. 81:96–106. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Wang X and Khalil RA: Matrix
metalloproteinases, vascular remodeling, and vascular disease. Adv
Pharmacol. 81:241–330. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Chen R, Zhou Z, Song Z, Feng W, Ding X,
Zhang H and Wang Y: Tangzhiqing exacerbates oxidized Low-density
Lipoprotein-induced cell pyroptosis through activation of NLRP3
inflammasome in human umbilical vein endothelial cells. Cell Biol
Int. 49:1173–1183. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Lu H, Sun J, Liang W, Chang Z, Rom O, Zhao
Y, Zhao G, Xiong W, Wang H, Zhu T, et al: Cyclodextrin prevents
abdominal aortic aneurysm via activation of vascular smooth muscle
cell transcription factor EB. Circulation. 142:483–498. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Li K, Zhang D, Zhai S, Wu H and Liu H:
METTL3-METTL14 complex induces necroptosis and inflammation of
vascular smooth muscle cells via promoting N6 methyladenosine mRNA
methylation of receptor-interacting protein 3 in abdominal aortic
aneurysms. J Cell Commun Signal. 17:897–914. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Bertheloot D, Latz E and Franklin BS:
Necroptosis, pyroptosis and apoptosis: An intricate game of cell
death. Cell Mol Immunol. 18:1106–1121. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Chang W, Lin J, Dong J and Li D:
Pyroptosis: An inflammatory cell death implicates in
atherosclerosis. Med Hypotheses. 81:484–486. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wortmann M, Peters AS, Erhart P, Körfer D,
Böckler D and Dihlmann S: Inflammasomes in the pathophysiology of
aortic disease. Cells. 10:24332021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Xia S, Zhang Z, Magupalli VG, Pablo JL,
Dong Y, Vora SM, Wang L, Fu TM, Jacobson MP, Greka A, et al:
Gasdermin D pore structure reveals preferential release of mature
interleukin-1. Nature. 593:607–611. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Mitra S and Sarkar A: Microparticulate
P2X7 and GSDM-D mediated regulation of functional IL-1β release.
Purinergic Signal. 15:119–123. 2019. View Article : Google Scholar :
|
|
100
|
Sun SJ, Zhang Z, Zhang GY, Wu JJ, Zhang
YX, Wu WB, Zhang Y, Yin H, Zhang JB, Cao Q, et al: GSDME-dependent
pyroptosis drives abdominal aortic aneurysm via promoting vascular
senescence. Nat Commun. 16:112482025. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Wang Q, Wu J, Zeng Y, Chen K, Wang C, Yang
S, Sun N, Chen H, Duan K and Zeng G: Pyroptosis: A pro-inflammatory
type of cell death in cardiovascular disease. Clin Chim Acta.
510:62–72. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Gong WB, Fu HY, Yang K, Zheng T, Guo K and
Zhao W: 4-Octyl itaconate blocks GSDMB-mediated pyroptosis and
restricts inflammation by inactivating granzyme A. Cell Prolif.
57:e137112024. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Ai YL, Wang WJ, Liu FJ, Fang W, Chen HZ,
Wu LZ, Hong X, Zhu Y, Zhang CX, Liu LY, et al: Mannose antagonizes
GSDME-mediated pyroptosis through AMPK activated by metabolite
GlcNAc-6P. Cell Res. 33:904–922. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Deng W, Bai Y, Deng F, Pan Y, Mei S, Zheng
Z, Min R, Wu Z, Li W, Miao R, et al: Streptococcal pyrogenic
exotoxin B cleaves GSDMA and triggers pyroptosis. Nature.
602:496–502. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Wu LZ, Huang YY, Hu HJ, Hong WB, Yan H, Ai
YL, Mi XY, Feng DY, Guo JY, Ding Y, et al: Disruption of heme
homeostasis by nuclear receptor Nur77 induces pyroptosis through
granzyme B-dependent GSDMC cleavage. Signal Transduct Target Ther.
10:4132025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Li Y, Hou Y, Sun Q, Zeng H, Meng F, Tian
X, He Q, Shao F and Ding J: Cleavage-independent activation of
ancient eukaryotic gasdermins and structural mechanisms. Science.
384:adm91902024. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Ke TT, Yuan C, Yuan Y, Liu JL, Zhou XL,
Liu JC, Xiong WF and Liu ZB: CD95 ligand drives abdominal aortic
aneurysm progression through Caspase-8-mediated GSDMD-dependent
endothelial pyroptosis: Modulation by SRC kinase. Apoptosis.
31:602026. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Singh P and Lim B: Targeting apoptosis in
cancer. Curr Oncol Rep. 24:273–284. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Zhang W, Zhu C, Liao Y, Zhou M, Xu W and
Zou Z: Caspase-8 in inflammatory diseases: A potential therapeutic
target. Cell Mol Biol Lett. 29:1302024. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Sahoo G, Samal D, Khandayataray P and
Murthy MK: A review on caspases: Key regulators of biological
activities and apoptosis. Mol Neurobiol. 60:5805–5837. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Green DR: The coming decade of cell death
research: Five riddles. Cell. 177:1094–1107. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Newton K, Strasser A, Kayagaki N and Dixit
VM: Cell death. Cell. 187:235–256. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Tsuchiya K: Switching from apoptosis to
pyroptosis: Gasdermin-elicited inflammation and antitumor immunity.
Int J Mol Sci. 22:4262021. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Zhang DW, Shao J, Lin J, Zhang N, Lu BJ,
Lin SC, Dong MQ and Han J: RIP3, an energy metabolism regulator
that switches TNF-induced cell death from apoptosis to necrosis.
Science. 325:332–336. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Wang H, Sun L, Su L, Rizo J, Liu L, Wang
LF, Wang FS and Wang X: Mixed lineage kinase domain-like protein
MLKL causes necrotic membrane disruption upon phosphorylation by
RIP3. Mol Cell. 54:133–146. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Rocca A, Tafuri D, Paccone M, Giuliani A,
Zamboli AGI, Surfaro G, Paccone A, Compagna R, Amato M, Serra R and
Amato B: Cell based therapeutic approach in vascular surgery:
Application and review. Open Med (Wars). 12:308–322. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Kitakata H, Endo J, Ikura H, Moriyama H,
Shirakawa K, Katsumata Y and Sano M: Therapeutic targets for
DOX-induced cardiomyopathy: Role of apoptosis vs. Ferroptosis. Int
J Mol Sci. 23:14142022. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Bedoui S, Herold MJ and Strasser A:
Emerging connectivity of programmed cell death pathways and its
physiological implications. Nat Rev Mol Cell Biol. 21:678–695.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Yang WS, SriRamaratnam R, Welsch ME,
Shimada K, Skouta R, Viswanathan VS, Cheah JH, Clemons PA, Shamji
AF, Clish CB, et al: Regulation of ferroptotic cancer cell death by
GPX4. Cell. 156:317–331. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Mao C, Liu X, Zhang Y, Lei G, Yan Y, Lee
H, Koppula P, Wu S, Zhuang L, Fang B, et al: DHODH-mediated
ferroptosis defence is a targetable vulnerability in cancer.
Nature. 593:586–590. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Dixon SJ and Olzmann JA: The cell biology
of ferroptosis. Nat Rev Mol Cell Biol. 25:424–442. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Fu H, Huang Q and Xie J: Targeting
regulated cell death pathways in COPD: Mechanisms and therapeutic
strategies. Cells. 14:18742025. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Kist M and Vucic D: Cell death pathways:
Intricate connections and disease implications. EMBO J.
40:e1067002021. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
D'Amico M and De Amicis F: Challenges of
regulated cell death: Implications for therapy resistance in
cancer. Cells. 13:10832024. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Xu AP, Xu LB, Smith ER, Fleishman JS, Chen
ZS and Xu XX: Cell death in cancer chemotherapy using taxanes.
Front Pharmacol. 14:13386332023. View Article : Google Scholar
|
|
126
|
Zhou SY, Cui GZ, Yan XL, Wang X, Qu Y, Guo
ZN and Jin H: Mechanism of ferroptosis and its relationships with
other types of programmed cell death: Insights for potential
interventions after intracerebral hemorrhage. Front Neurosci.
14:5890422020. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Tang D, Kang R, Berghe TV, Vandenabeele P
and Kroemer G: The molecular machinery of regulated cell death.
Cell Res. 29:347–364. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Jia LX, Zhang WM, Zhang HJ, Li TT, Wang
YL, Qin YW, Gu H and Du J: Mechanical stretch-induced endoplasmic
reticulum stress, apoptosis and inflammation contribute to thoracic
aortic aneurysm and dissection. J Pathol. 236:373–383. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Bian S, Yang L, Zhao DF, Lv LZ, Wang TZ
and Yuan H: HMGB1/TLR4 signaling pathway enhances abdominal aortic
aneurysm progression in mice by upregulating necroptosis. Inflamm
Res. 72:703–713. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Shi Y, Zhao Y, Sun SJ, Lan XT, Wu WB,
Zhang Z, Chen YX, Yan YY, Xu YP, Li DJ, et al: Targeting GPX4
alleviates ferroptosis and retards abdominal aortic aneurysm
formation. Biochem Pharmacol. 234:1168002025. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Chen Y, Wang J, Dai Q, Chen P, Wang H, Li
R, Gu Y, Liu H, Xiao H, Qin S, et al: Harnessing the death switch:
Empowering cancer therapy by modulating the apoptosis-pyroptosis
transition. Biomaterials. 329:1239732026. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Theofilis P, Sagris M, Oikonomou E,
Antonopoulos AS, Siasos G, Tsioufis C and Tousoulis D: Inflammatory
mechanisms contributing to endothelial dysfunction. Biomedicines.
9:7812021. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
van der Heijden T, Kritikou E, Venema W,
van Duijn J, van Santbrink PJ, Slütter B, Foks AC, Bot I and Kuiper
J: NLRP3 inflammasome inhibition by MCC950 reduces atherosclerotic
lesion development in apolipoprotein E-Deficient Mice-Brief report.
Arterioscler Thromb Vasc Biol. 37:1457–1461. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Lu J, Xie S, Deng Y, Xie X and Liu Y:
Blocking the NLRP3 inflammasome reduces osteogenic calcification
and M1 macrophage polarization in a mouse model of calcified aortic
valve stenosis. Atherosclerosis. 347:28–38. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Xiao M, Li L, Li C, Liu L, Yu Y and Ma L:
3,4-Methylenedioxy-β-Nitrostyrene ameliorates experimental burn
wound progression by inhibiting the NLRP3 inflammasome activation.
Plast Reconstr Surg. 137:566e–575e. 2016. View Article : Google Scholar
|
|
136
|
Ruan H, Zeng X and Shen S: Mechanism of
curcumin inhibiting NLRP3 inflammatory body and improving
atherosclerotic endothelial cell injury. Discov Med. 36:121–128.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Jiang H, Xie Y, Lu J, Li H, Zeng K, Hu Z,
Wu D, Yang J, Yao Z, Chen H, et al: Pristimerin suppresses AIM2
inflammasome by modulating AIM2-PYCARD/ASC stability via selective
autophagy to alleviate tendinopathy. Autophagy. 20:76–93. 2024.
View Article : Google Scholar :
|
|
138
|
Li Y, Niu X, Xu H, Li Q, Meng L, He M,
Zhang J and Zhang Z and Zhang Z: VX-765 attenuates atherosclerosis
in ApoE deficient mice by modulating VSMCs pyroptosis. Exp Cell
Res. 389:1118472020. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Duan Y, Peng Z, Zhong S, Zhou P, Huang H,
Li J and He Z: VX-765 ameliorates CKD VSMC calcification by
regulating STAT3 activation. Eur J Pharmacol. 945:1756102023.
View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Ye X, Lin ZJ, Hong GH, Wang ZM, Dou RT,
Lin JY, Xie JH and Shen YW: Pyroptosis inhibitors MCC950 and VX-765
mitigate myocardial injury by alleviating oxidative stress,
inflammation, and apoptosis in acute myocardial hypoxia. Exp Cell
Res. 438:1140612024. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Boost KA, Hoegl S, Hofstetter C, Flondor
M, Stegewerth K, Platacis I, Pfeilschifter J, Muhl H and Zwissler
B: Targeting caspase-1 by inhalation-therapy: Effects of
Ac-YVAD-CHO on IL-1 beta, IL-18 and downstream proinflammatory
parameters as detected in rat endotoxaemia. Intensive Care Med.
33:863–871. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Chu LH, Indramohan M, Ratsimandresy RA,
Gangopadhyay A, Morris EP, Monack DM, Dorfleutner A and Stehlik C:
The oxidized phospholipid oxPAPC protects from septic shock by
targeting the non-canonical inflammasome in macrophages. Nature
Commun. 9:9962018. View Article : Google Scholar
|
|
143
|
Lan C, Fang G, Li X, Chen X, Chen Y, Hu T,
Wang X, Cai H, Hao J, Li H, et al: SerpinB1 targeting safeguards
against pathological cardiac hypertrophy and remodelling by
suppressing cardiomyocyte pyroptosis and inflammation initiation.
Cardiovasc Res. 121:113–127. 2025. View Article : Google Scholar
|
|
144
|
Wang X, Kuang J, Li XT, Hu X, Liu YH, Hu
CP, Wang M, Wang Q and Zhang Z: Dimethyl fumarate is repurposed to
ameliorate aortic aneurysm and dissection in mice. Eur J Pharmacol.
988:1772152025. View Article : Google Scholar
|
|
145
|
Song H, Xu T, Feng X, Lai Y, Yang Y, Zheng
H, He X, Wei G, Liao W, Liao Y, et al: Itaconate prevents abdominal
aortic aneurysm formation through inhibiting inflammation via
activation of Nrf2. EBioMedicine. 57:1028322020. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Cao R, Li Z, Wu C, Ji S, Li Y, Cao X, Dong
X, Jiang M, Pang T, Wang C, et al: Identification of a small
molecule with strong Anti-Inflammatory activity in experimental
autoimmune encephalomyelitis and sepsis through blocking Gasdermin
D activation. J Immunol. 209:820–828. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Liu M, Liu D, Yu C, Fan HH, Zhao X, Wang
H, Zhang C, Zhang M, Bo R, He S, et al: Caffeic acid, but not
ferulic acid, inhibits macrophage pyroptosis by directly blocking
gasdermin D activation. MedComm (2020). 4:e2552023. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Shakoory B, Carcillo JA, Chatham WW, Amdur
RL, Zhao H, Dinarello CA, Cron RQ and Opal SM: Interleukin-1
receptor blockade is associated with reduced mortality in sepsis
patients with features of macrophage activation syndrome:
Reanalysis of a prior phase III trial. Crit Care Med. 44:275–281.
2016. View Article : Google Scholar
|
|
149
|
Ridker PM, Everett BM, Thuren T, MacFadyen
JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker
SD, et al: Antiinflammatory therapy with canakinumab for
atherosclerotic disease. N Engl J Med. 377:1119–1131. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Myachikova VY, Maslyanskiy AL, Moiseeva
OM, Vinogradova OV, Gleykina EV, Lavrovsky Y, Abbate A, Grishin SA,
Egorova AN, Schedrova ML and Samsonov MY: Treatment of idiopathic
recurrent pericarditis with goflikicept: Phase II/III study
results. J Am Coll Cardiol. 82:30–40. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Zaccone P, Phillips J, Conget I, Cooke A
and Nicoletti F: IL-18 binding protein fusion construct delays the
development of diabetes in adoptive transfer and
cyclophosphamide-induced diabetes in NOD mouse. Clin Immunol.
115:74–79. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Coll RC, Hill JR, Day CJ, Zamoshnikova A,
Boucher D, Massey NL, Chitty JL, Fraser JA, Jennings MP, Robertson
AAB and Schroder K: MCC950 directly targets the NLRP3
ATP-hydrolysis motif for inflammasome inhibition. Nat Chem Biol.
15:556–559. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Coll RC, Robertson AA, Chae JJ, Higgins
SC, Muñoz-Planillo R, Inserra MC, Vetter I, Dungan LS, Monks BG,
Stutz A, et al: A small-molecule inhibitor of the NLRP3
inflammasome for the treatment of inflammatory diseases. Nat Med.
21:248–255. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Li H, Guan Y, Liang B, Ding P, Hou X, Wei
W and Ma Y: Therapeutic potential of MCC950, a specific inhibitor
of NLRP3 inflammasome. Eur J Pharmacol. 928:1750912022. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Mangan MSJ, Olhava EJ, Roush WR, Seidel
HM, Glick GD and Latz E: Targeting the NLRP3 inflammasome in
inflammatory diseases. Nat Rev Drug Discov. 17:6882018. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Wu J, Wang Z, Shao W and Mo J: Bridging
inflammation and venous thrombosis: The NLRP3 inflammasome
connection. Front Cardiovasc Med. 12:15847452025. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Zheng J, Jiang Z, Song Y, Huang S, Du Y,
Yang X, Xiao Y, Ma Z, Xu D and Li J:
3,4-Methylenedioxy-β-Nitrostyrene alleviates dextran sulfate
Sodium-induced mouse colitis by inhibiting the NLRP3 inflammasome.
Front Pharmacol. 13:8662282022. View Article : Google Scholar
|
|
158
|
Hasanzadeh S, Read MI, Bland AR, Majeed M,
Jamialahmadi T and Sahebkar A: Curcumin: An inflammasome silencer.
Pharmacol Res. 159:1049212020. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Li F, Xu M, Wang M, Wang L, Wang H, Zhang
H, Chen Y, Gong J, Zhang JJ, Adcock IM, et al: Roles of
mitochondrial ROS and NLRP3 inflammasome in multiple ozone-induced
lung inflammation and emphysema. Respir Res. 19:2302018. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Choi YJ, Kim S, Choi Y, Nielsen TB, Yan J,
Lu A, Ruan J, Lee HR, Wu H, Spellberg B, et al: SERPINB1-mediated
checkpoint of inflammatory caspase activation. Nat Immunol.
20:276–287. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Zhou C, Yamaguchi M, Kusaka G, Schonholz
C, Nanda A and Zhang JH: Caspase inhibitors prevent endothelial
apoptosis and cerebral vasospasm in dog model of experimental
subarachnoid hemorrhage. J Cereb Blood Flow Metab. 24:419–431.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Wang XJ, Cao Q, Zhang Y and Su XD:
Activation and regulation of caspase-6 and its role in
neurodegenerative diseases. Annu Rev Pharmacol Toxicol. 55:553–572.
2015. View Article : Google Scholar
|
|
163
|
Huang D, Xu D, You C, Li B, Li L, Yang Q,
Zhou W, Meng Q and Liang Z: Caspase-8-driven NLRP3 inflammasome
activation exacerbates bronchopulmonary dysplasia by increasing the
apoptosis and pyroptosis crosstalk of alveolar epithelial cells.
Int Immunopharmacol. 161:1150252025. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Zhu C, Xu S, Jiang R, Yu Y, Bian J and Zou
Z: The gasdermin family: Emerging therapeutic targets in diseases.
Signal Transduct Target Ther. 9:872024. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Rathkey JK, Zhao J, Liu Z, Chen Y, Yang J,
Kondolf HC, Benson BL, Chirieleison SM, Huang AY, Dubyak GR, et al:
Chemical disruption of the pyroptotic pore-forming protein
gasdermin D inhibits inflammatory cell death and sepsis. Sci
Immunol. 3:eaat27382018. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Rashidi M, Simpson DS, Hempel A, Frank D,
Petrie E, Vince A, Feltham R, Murphy J, Chatfield SM, Salvesen GS,
et al: The pyroptotic cell death effector gasdermin D is activated
by gout-associated uric acid crystals but is dispensable for cell
death and IL-1β release. J Immunol. 203:736–748. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Qiao YL, Zhu MW, Xu S, Jiao WE, Ni HF, Tao
ZZ and Chen SM: Allergen-induced CD11c + dendritic cell pyroptosis
aggravates allergic rhinitis. Cell Commun Signal. 21:2812023.
View Article : Google Scholar
|
|
168
|
Yao F, Jin Z, Zheng Z, Lv X, Ren L, Yang
J, Chen D, Wang B, Yang W, Chen L, et al: HDAC11 promotes both
NLRP3/caspase-1/GSDMD and caspase-3/GSDME pathways causing
pyroptosis via ERG in vascular endothelial cells. Cell Death
Discov. 8:1122022. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Boersma B, Möller K, Wehl L, Puddinu V,
Huard A, Fauteux-Daniel S, Bourquin C, Palmer G and Bein T:
Inhibition of IL-1β release from macrophages targeted with
necrosulfonamide-loaded porous nanoparticles. J Control Release.
351:989–1002. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Guo W, Chen S, Li C, Xu J and Wang L:
Application of disulfiram and its metabolites in treatment of
inflammatory disorders. Front Oharmacol. 12:7950782021.
|
|
171
|
Guo J, Shi J, Qin M, Wang Y, Li Z, Shoji
T, Ikezoe T, Ge Y and Xu B: Pharmacological inhibition of gasdermin
D suppresses angiotensin II-induced experimental abdominal aortic
aneurysms. Biomolecules. 13:8992023. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Humphries F, Shmuel-Galia L,
Ketelut-Carneiro N, Li S, Wang B, Nemmara VV, Wilson R, Jiang Z,
Khalighinejad F, Muneeruddin K, et al: Succination inactivates
gasdermin D and blocks pyroptosis. Science. 369:1633–1637. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Runtsch MC, Angiari S, Hooftman A, Wadhwa
R, Zhang Y, Zheng Y, Spina JS, Ruzek MC, Argiriadi MA, McGettrick
AF, et al: Itaconate and itaconate derivatives target JAK1 to
suppress alternative activation of macrophages. Cell Metab.
34:487–501.e8. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Bambouskova M, Potuckova L, Paulenda T,
Kerndl M, Mogilenko DA, Lizotte K, Swain A, Hayes S, Sheldon RD,
Kim H, et al: Itaconate confers tolerance to late NLRP3
inflammasome activation. Cell Rep. 34:1087562021. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
El-Seedi HR, El-Said AM, Khalifa SA,
Göransson U, Bohlin L, Borg-Karlson AK and Verpoorte R:
Biosynthesis, natural sources, dietary intake, pharmacokinetic
properties, and biological activities of hydroxycinnamic acids. J
Agric Food Chem. 60:10877–10895. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Chauhan D, Vande Walle L and Lamkanfi M:
Therapeutic modulation of inflammasome pathways. Immunol Rev.
297:123–138. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Hu JJ, Liu X, Xia S, Zhang Z, Zhang Y,
Zhao J, Ruan J, Luo X, Lou X, Bai Y, et al: FDA-approved disulfiram
inhibits pyroptosis by blocking gasdermin D pore formation. Nat
Immunol. 21:736–745. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Orning P, Lien E and Fitzgerald KA:
Gasdermins and their role in immunity and inflammation. J Exp Med.
216:2453–2465. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Shua Y, Wang Q, Wang Y and Jiang C:
Progress in small-molecule inhibitors of gasdermin D. Eur J Med
Chem. 300:1181712025. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Hu Y, Li H, Zhang X, Song Y, Liu J, Pu J,
Wen S, Xu H, Xin H, Wang B and Yang S: Identification of two
repurposed drugs targeting GSDMD oligomerization interface I to
block pyroptosis. Cell Chem Biol. 31:2024–2038.e7. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Schiffelers LDJ, Tesfamariam YM, Jenster
LM, Diehl S, Binder SC, Normann S, Mayr J, Pritzl S, Hagelauer E,
Kopp A, et al: Antagonistic nanobodies implicate mechanism of GSDMD
pore formation and potential therapeutic application. Nat Commun.
15:82662024. View Article : Google Scholar : PubMed/NCBI
|















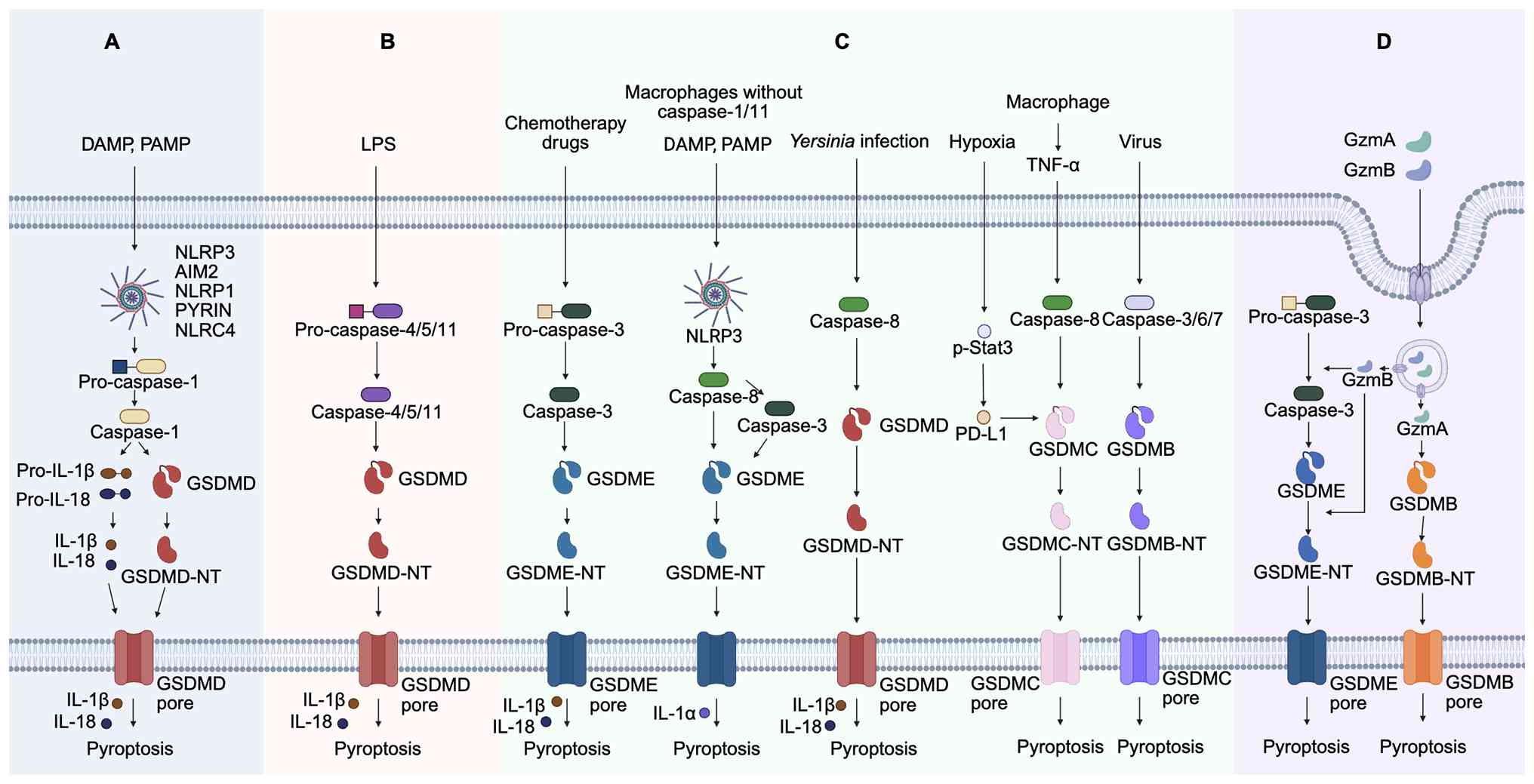
![Potential therapeutic strategies
targeting pyroptotic pathway for AA and the link between pytoptosis
and AA. Inhibitors of NLRP3 inflammasomes: MCC950, CY-09, MNS and
curcumin. Inhibitors of caspase-1: VX-765 and Ac-YYAD-CHO.
Inhibitors of caspase-4/5/11: ox-PAPC and SERPINB1. Inhibitors of
IL-1: Anakinra, Canakinumab and Goflikicept; Inhibitors of IL-18:
IL-18BP. Inhibitors that inhibit GSDMD cleavage: Caffeic acid.
Inhibitors of GSDMD pore formation: NSA, DSF, DMF, Itaconate and
C-202-2729. Created with Biorender.com. AA, aortic aneurysm; NLRP3, NOD-like
receptor family pyrin domain-containing 3; MCC950,
1-(1,2,3,5,6,7-hexahydro-s-indacen-4-yl)-3-[4-(2-hydroxypropan-2-yl)furan-2-yl]sulfonylurea;
CY-09,
4-[[4-Oxo-2-thioxo-3-[[3-(trifluoromethyl)phenyl]methyl]-5-thiazolidinylidene]methyl]benzoic
acid; MNS, 3,4-methylenedioxy-β-nitrostyrene; VSMC, vascular smooth
muscle cells; GSDMD, gasdermin D; Caspase-1/4/5/11,
cysteine-aspartic protease 1/4/5/11; IL-1β, interleukin-1β; IL-18,
interleukin-18; DAMPs, damage-associated molecular patterns; α-SMA,
α-smooth muscle actin; SM22α, smooth muscle protein 22 α; SMMHC,
smooth muscle myosin heavy chain; OPN, osteopontin; BMP-2, bone
morphogenetic protein 2; OCN, osteocalcin; VX-765,
(S)-1-((S)-2-{[1-(4-Amino-3-chlorophenyl)methanoyl]amino}-3,3-dimethylbutanoyl)pyrrolidine-2-carboxylic
acid ((2R,3S)-2-ethoxy-5-oxotetrahydrofuran-3-yl)amide;
Ac-YVAD-CHO,
N-Acetyl-L-tyrosyl-L-valyl-N-[(1S)-2-carboxy-1-formylethyl]-L-alaninamide;
EC, endothelial cell; MMP, matrix metalloproteinase; ECM,
extracellular matrix; ox-PAPC, oxidized
1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphorylcholine; -NT,
N-terminal; IL-18BP, interleukin-18 binding protein; NSA,
necrosulfonamide; DSF, disulfiram; DMF, dimethyl fumarate;
SERPINB1, serine protease inhibitor clade B member 1; C202-2729, a
GSDMD N-terminal inhibitor.](/article_images/ijmm/57/6/ijmm-57-06-05818-g01.jpg)