Introduction
Diabetes mellitus (DM), a chronic metabolic disorder
characterized by hyperglycemia, poses a severe global health burden
and leads to multi-organ complications (1,2).
Despite advances in glucose-lowering therapies, the progressive
nature of diabetic complications underscores an unmet need for
strategies that move beyond glycemic control to directly protect
target organs (3,4).
The apelin/elabela-apelin receptor (APJ) signaling
system, composed of the G protein-coupled receptor APJ and its two
endogenous ligands, apelin and elabela (ELA), plays a fundamental
role in maintaining metabolic homeostasis (5,6).
While it modulates crucial processes such as insulin secretion,
vascular tone, and cell survival, its net effect is notoriously
context-dependent, shifting from protective to pathogenic across
different disease stages, tissue types and even between its two
ligands (7-11). Currently, it remains unclear how
the APJ system integrates signals from apelin and ELA to produce
divergent outcomes in complications such as retinopathy and
nephropathy. These knowledge gaps, particularly regarding
ligand-specific biased signaling and tissue-specific receptor
behavior, represent major obstacles to clinical application.
Through a systematic review of the literature, the
present review first provided a concise overview of the
Apelin/ELA-APJ system. Its complex and often contradictory, roles
in systemic glucose metabolism and in the pathogenesis of diabetic
kidney disease, cardiomyopathy and retinopathy were then dissected.
Finally, these insights were synthesized to evaluate critically
emerging therapeutic strategies, arguing that the future of
targeting this system lies in context-guided precision
intervention.
A brief description of the apelin/ELA
system
Understanding the multifaceted Apelin/ELA-APJ system
is important for deciphering its context-dependent roles in
diabetes. This section outlined its core components, expression and
key signaling mechanisms that form the basis for its complex
physiological and pathological functions. For a comprehensive
treatise on the biochemistry and broad physiology of this system,
readers are referred to excellent dedicated reviews (12-15).
Molecular components of the system
APJ
APJ is a class of G protein-coupled receptor (GPCR),
initially identified as an orphan receptor with high sequence
homology to the angiotensin II type 1 receptor (AT1R) (16-18). Despite this structural
similarity, APJ does not bind angiotensin II. Instead, it forms a
critical counter-regulatory axis against AT1R signaling,
antagonizing a number of angiotensin II's deleterious effects in
cardiovascular and renal tissues (19,20). APJ is found to be widely
expressed in heart, blood vessels, kidney, ovary and human early
embryonic tissues (13,21). This broad distribution, coupled
with its evolutionary kinship to the AT1R, positions the APJ system
as a key counter-regulatory module to the renin-angiotensin system
(RAS), a relationship with profound implications for vascular and
metabolic homeostasis in diabetes.
Apelin
Apelin was isolated through reverse pharmacology
approaches as its first cognate ligand in 1998 (15). It is derived from a preproprotein
and enzymatically processed into multiple active isoforms,
including apelin-36, -17, and -13 (Fig. 1A) (22). Among these, apelin-36 is the most
widely distributed subtype, with high expression levels
particularly observed in the lungs, testes and uterus (23). Apelin-13, though less abundant in
a number of tissues, is a highly potent isoform (24,25). (Pyr1) apelin-13, which
contains an N-terminal pyroglutamate modification, exhibits
increased stability against aminopeptidase degradation and
represents the most abundant Apelin isoform in human plasma and the
cardiovascular system (24,26). Apelin-17 has been identified as a
potential biomarker for idiopathic pulmonary arterial hypertension
(27). It also contributes to
diuresis by modulating the arginine-vasopressin system, thereby
aiding in the correction of hyponatremia (28,29). All apelin isoforms bind to the
APJ, albeit with varying affinities, leading to distinct downstream
biological effects (30,31). The interaction between apelin and
APJ depends critically on specific structural motifs within the
peptide. The RPRL sequence, in particular, facilitates the
formation of a stable beta-sheet structure that is essential for
receptor binding (32). The
existence of multiple bioactive isoforms with distinct
pharmacokinetic profiles and tissue distributions forms a molecular
basis for the nuanced and context-dependent regulation exerted by
the apelin arm of the system.
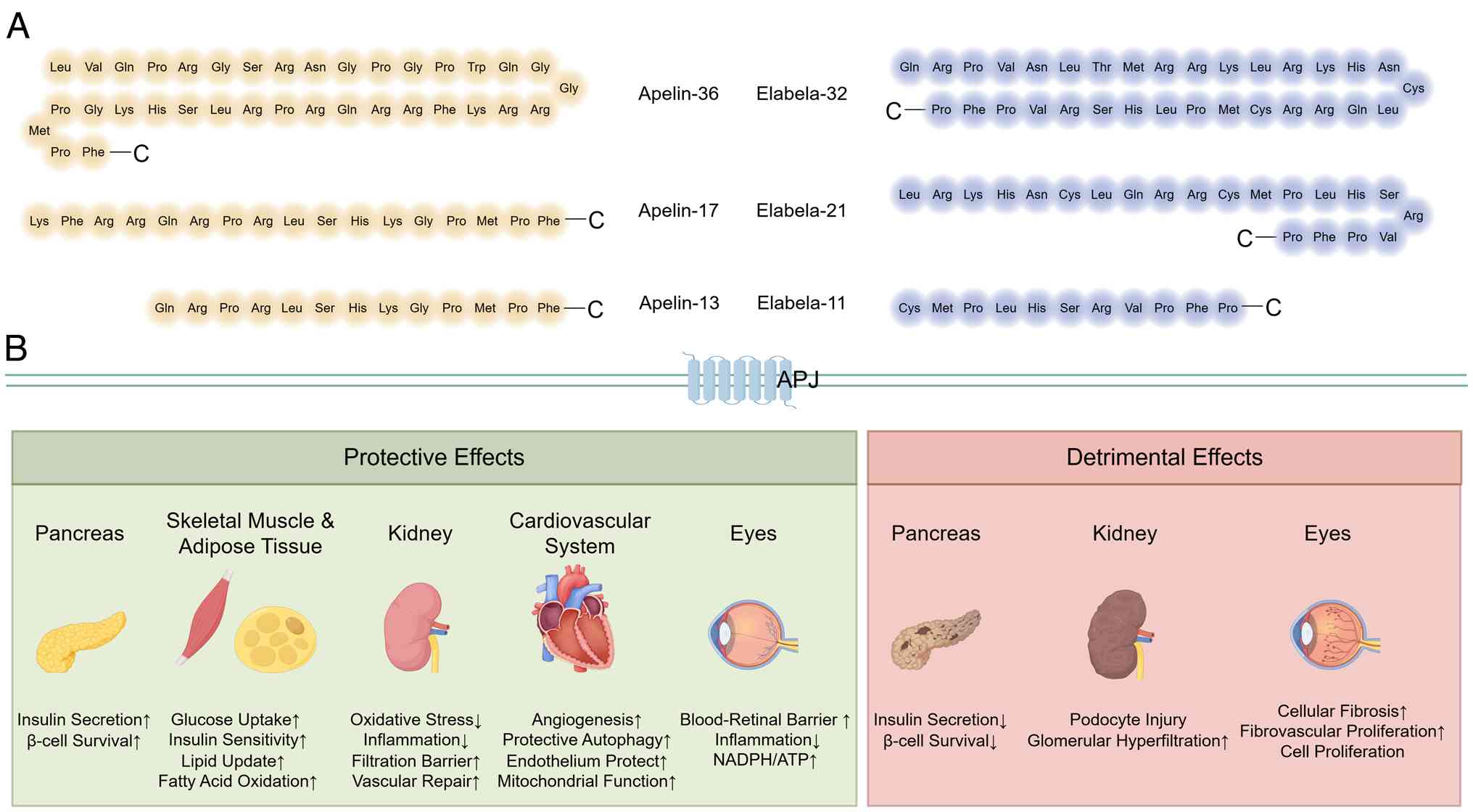 | Figure 1Biosynthesis and duality role of the
apelin/ELA-APJ axis. (A) The APLN gene encodes a 77-amino acid
preproapelin, which undergoes sequential proteolytic cleavage to
generate bioactive isoforms, primarily apelin-36, apelin-17 and
apelin-13. Similarly, ELA is processed from its 54-amino-acid
precursor into active fragments, such as ELA-32, ELA-21, and
ELA-11. (B) The APJ system exhibits functional divergence across
organ microenvironments. ELA-APJ. ELA, elabela; APJ, apelin
receptor; NADPH, nicotinamide adenine dinucleotide phosphate. |
ELA
ELA or Toddler, is a distinct peptide encoded by the
APELA gene, discovered as a second endogenous ligand for APJ
in 2013 (33). It is cleaved to
produce the mature ELA-32 peptide, which is cleaved by furin to
produce ELA-21 and ELA-11 (Fig.
1A) (33,34). Its identification resolved the
phenotypic disparity between APJ and apelin gene knockout models,
with APJ deficiency causing more severe developmental defects,
underscoring ELA's non-redundant role (35,36). Unlike apelin, ELA is critically
expressed during embryogenesis and maintains prominent expression
in adult vascular endothelium and kidney (37,38). Despite minimal sequence homology
with apelin, ELA binds APJ with high affinity but is proposed to
engage distinct downstream signaling profiles (39,40). The differential spatiotemporal
expression and signaling bias of these two ligands form the
molecular basis for their potentially divergent, and occasionally
opposing, roles in diabetic pathophysiology.
Expression patterns and physiological
roles
The evolutionarily conserved expression profile of
APJ across mammalian species reflects its pan-tissue regulatory
functions in homeostatic maintenance and pathological mechanisms
(16). Spatiotemporal expression
profiling reveals conserved receptor distribution patterns, being
expressed in key metabolic tissues such as adipose, skeletal
muscle, liver and pancreatic islets; in the cardiovascular-renal
system, spanning the heart, vasculature and kidney; and throughout
the central nervous system (16,18,41,42). This ubiquitous tissue
distribution phylogenetically correlates with APJ's pleiotropic
regulatory capacities, spanning neurohumoral modulation,
angiogenesis, and energy metabolism.
Functionally, this ligand-receptor system engages in
pleiotropic regulation. It is a key determinant of cardiovascular
homeostasis, modulating vascular tone, cardiac contractility and
angiogenesis (6,26,43). In metabolic regulation, it
enhances insulin sensitivity, promotes glucose uptake and regulates
lipid handling, with apelin itself being an insulin-responsive
adipokine (7,44). APJ activation could enhance
mitochondrial biogenesis and autophagy, highlighting its potential
role in metabolic diseases such as DM (45,46). It also contributes to fluid
balance and hormonal secretion, including in the hypothalamus and
pituitary (47). The system
promotes vasodilation, reduces cardiac afterload, inhibits
angiotensin II-induced hypertrophy and protects endothelial cells
from apoptosis. In the kidney, it modulates glomerular hemodynamics
and tubular function (38,48-51). The very pervasiveness of this
system means that its dysregulation in diabetes has the potential
to disrupt multiple organ systems simultaneously. Conversely, its
protective functions position it as an endogenous compensatory
mechanism against hyperglycemic injury (Fig. 1B). The ensuing sections will
explore how this delicate balance tips toward protection or
pathology in specific diabetic complications.
Signaling mechanisms and functional
diversity
The APJ functions as a sophisticated molecular
switch, where the physiological outcome is determined not by a
single pathway, but by the type of the receptor activation. APJ
predominantly couples with Gαi/o and Gαq/11
subfamilies, with distinct activation mechanisms (52). Real-time bioluminescence
resonance energy transfer/Förster resonance energy transfer imaging
has revealed that Gαi2 and Gαi3 undergo
molecular rearrangement without subunit dissociation, whereas
Gαo and Gαq follow the classical dissociation
model (53). This diversity in
G-protein engagement enables APJ to simultaneously modulate cyclic
adenosine monophosphate inhibition, intracellular Ca2+
release and phosphatidylinositol 3-kinase (PI3K)/Akt activation,
depending on the cellular context (52). The termination and
diversification of these signals depend on the phosphorylation
barcode at the APJ C-terminus. Research has revealed a specific
phosphorylation barcode on APJ, where residues such as Ser339 are
important for β-arrestin recruitment and sustained ERK signaling,
while Ser335 phosphorylation specifically mediates ELA-induced
β-arrestin interactions (54).
This ligand-influenced phosphorylation pattern underlies the
emerging concept of ligand-specific signaling bias. Apelin and ELA
stabilize different APJ conformations, leading to biased engagement
of downstream G proteins and β-arrestin (34,55,56).
While GPCRs function as monomers, they also
dynamically form homodimers, heterodimers and higher-order
oligomers with unique functions. APJ constitutively heterodimerizes
with AT1R, as well as with other GPCRs, including the κ-opioid
receptor, neurotensin receptor-1 and bradykinin receptors (55,57). Within the APJ-AT1R heterodimer,
APJ engagement selectively attenuates AT1R-mediated β-arrestin
recruitment while preserving Gq-dependent calcium mobilization, a
pattern of biased antagonism that cannot be explained by simple
pathway crosstalk. Apelin-13 increases APJ-AT1R heterodimer
abundance, thereby potentiating cardioprotective signaling outputs,
whereas angiotensin II itself does not affect dimer stability
(55). APJ signaling antagonizes
angiotensin II/AT1R actions and may upregulate
angiotensin-converting enzyme 2 (ACE2), thereby shifting the RAS
balance toward the protective angiotensin-(1-7)
axis (50,51). This physical interaction explains
how the apelin system counteracts the pathological pressure of the
RAS even when AT1R expression is high (58).
These signaling events converge on PI3K/Akt and
AMP-activated protein kinase (AMPK) activation, promoting glucose
transporter 4 (GLUT4) translocation, nitric oxide (NO) production
and cell survival (44,59,60). Under physiological conditions,
the MAPK and STAT3 pathways contribute to adaptive cellular growth
and repair. However, in sustained chronic hyperglycemia or hypoxia,
these same signals can drive excessive cell proliferation,
migration and extracellular matrix deposition, linking the system
to fibrosis and pathological remodeling observed in advanced
complications (61,62). Therefore, the net biological
effect is not intrinsic to the receptor, but an emergent property
of ligand identity, phosphorylation state, oligomeric configuration
and metabolic milieu. This principle shapes the context-dependent
duality of the system in diabetic complications.
Apelin/ELA-APJ system in diabetes
The apelin/ELA-APJ system plays a multifaceted role
in glucose metabolism. Its effect is not universally beneficial or
detrimental but is dictated by specific physiological and
pathological contexts. Understanding this ligand-specific
functional duality is key to appreciating the system's role in
diabetes.
Apelin: A multipotent and
context-dependent metabolic regulator
Apelin transcends the simplistic definition of an
insulin secretion modulator, emerging instead as a sophisticated
systemic regulator of glucose metabolism whose actions are finely
tuned by concentration, metabolic status and tissue environment.
Its role in diabetes is best understood not as uniformly beneficial
or detrimental, but as a series of adaptive and maladaptive
responses to specific metabolic stresses across different organs
(7,44,63,64).
Biphasic guardian of pancreatic
β-cells
Initial investigation has positioned apelin as an
inhibitory modulator of insulin secretion in vivo and in
isolated islets (65). This
acute suppression appears to be mediated through a PI3K-dependent
phosphodiesterase 3B pathway, a mechanism potentiated by
co-administration with glucagon-like peptide-1 (63). However, higher concentrations or
chronic exposure improve overall glucose metabolism and normalize
insulin levels (66). Genetic
ablation of apelin results in fasting hyperinsulinemia and systemic
insulin resistance, whereas chronic apelin-13 improves overall
glucose metabolism and normalizes insulin levels (67). This long-term benefit is linked
to enhanced insulin sensitivity and direct β-cell protection,
including mitigation of endoplasmic reticulum stress in type 1
diabetes (T1D) and promotion of β-cell mass expansion in type 2
diabetes (T2D) models (45,68). Conditional knockout of APJ in
islets impairs compensatory β-cell hyperplasia, confirming the
essential role of the receptor in adaptation to metabolic demand
(69). These findings reposition
the long-term benefit of apelin as stemming more from its ability
to enhance insulin sensitivity and preserve functional β-cell mass,
rather than from its direct and variable effects on acute insulin
secretion.
Insulin-sensitizing hormone in peripheral
tissues
The primary benefit of apelin in diabetes appears to
be its potent insulin-sensitizing effect in key metabolic tissues.
In adipocytes, apelin-13 enhances glucose uptake via the PI3K/Akt
pathway, promoting GLUT4 translocation and membrane expression, and
upregulates aquaporin 7 to ameliorate lipid accumulation (60,70). Enhancement of glucose uptake
mediated by apelin occurs through dose-dependent activation of AMPK
phosphorylation; however, no significant effect on lipolysis has
been observed (71). In skeletal
muscle, chronic apelin-13 treatment promotes complete fatty acid
oxidation, enhances mitochondrial oxidative capacity and biogenesis
and reduces the accumulation of toxic lipid intermediates, thereby
ameliorating insulin resistance at the muscular level (72). Differing from findings in mouse
models, the baseline expression levels of apelin and its receptor
APJ in adipose tissue and skeletal muscle do not differ markedly
between patients with T2D and healthy controls (73). This highlights the
tissue-specific and species-specific regulation of the apelin
system, which may vary with the degree of insulin resistance
(Fig. 2A).
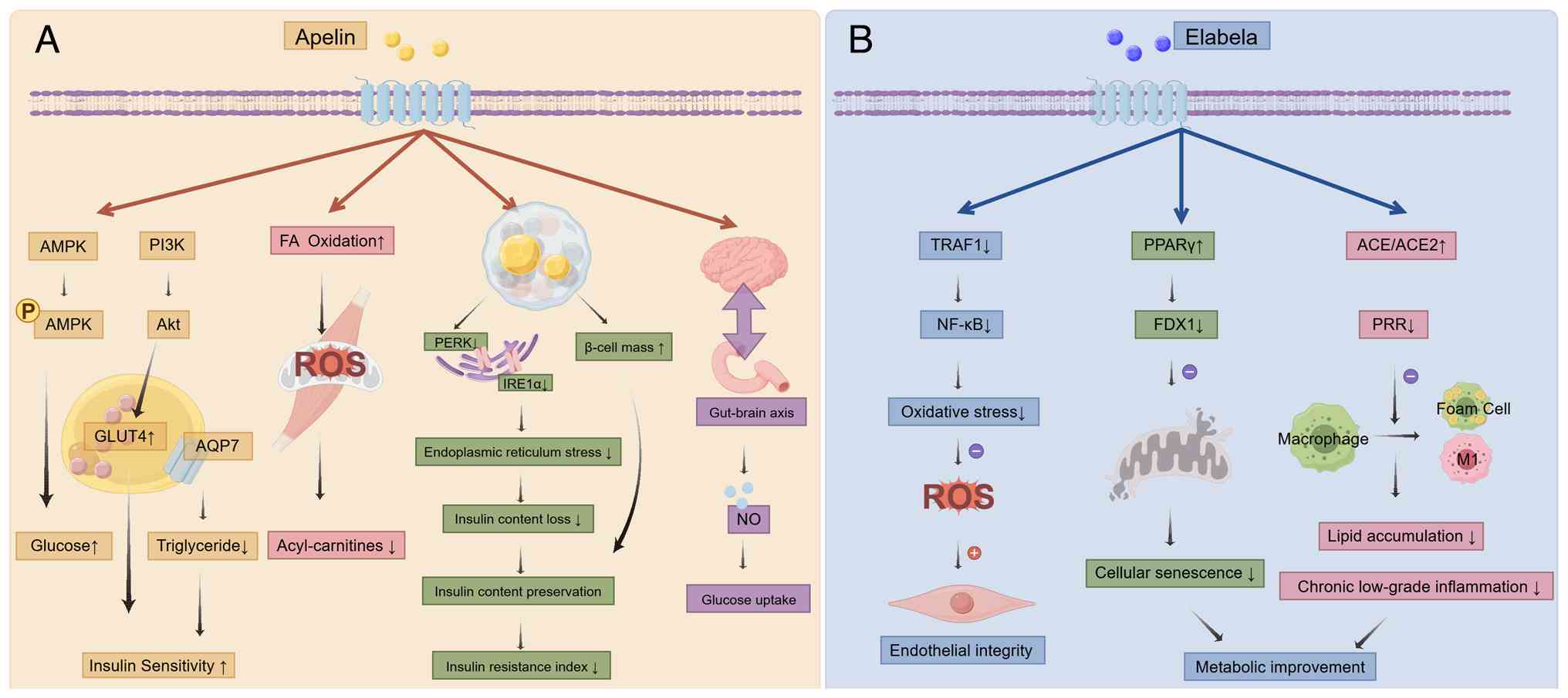 | Figure 2Role of the apelin/ELA-APJ axis in
diabetic homeostasis. (A) The Apelin system affects glucose
absorption and the survival of pancreatic islet cells by regulating
fat, β-cell endoplasmic reticulum, skeletal muscle, brain-gut axis,
etc. (B) The ELA system maintains homeostasis by preserving the
integrity of endothelial cells, regulating oxidative stress, and
controlling lipid deposition. ELA, elabela; APJ, apelin receptor;
AMPK, AMP-activated protein kinase; FAA, free fatty acid; NO,
nitric oxide; TRAF-1, tumor necrosis factor receptor-associated
factor 1; PPAR, peroxisome proliferator-activated receptor; ACE,
angiotensin-converting enzyme; FDX1, ferredoxin 1; PPR, (Pro)renin
receptor; ROS, Reactive oxygen species. |
These pre-clinical findings are supported by
clinical evidence. Serum apelin levels are elevated in patients
with both T1D and T2D compared with healthy controls, with the
highest levels found in patients with T1D (74). Critically, a proof-of-concept
clinical trial has provided the direct evidence in humans,
demonstrating that apelin infusion acutely improved glucose
tolerance and insulin sensitivity in patients with T2D (75). This collective evidence
solidifies the hypothesis that increased circulating apelin levels
are a counter-regulatory response to insulin resistance and
hyperglycemia, primarily acting to enhance insulin sensitivity in
the periphery.
Central-peripheral dichotomy
Central administration of low-dose apelin lowers
blood glucose levels, while higher doses induce insulin resistance
(59). However, in obese mice,
apelin promotes hypothalamic inflammation, suppresses brown adipose
tissue thermogenesis and contributes to the pathogenesis of T2D
(59,76). An additional layer of complexity
involves the gut-brain axis. Apelin can modulate duodenal
contraction rhythms, which in turn influence hypothalamic NO levels
to fine-tune skeletal muscle glucose uptake (47). This central-peripheral conflict
necessitates therapeutic strategies that can selectively engage the
beneficial peripheral actions while avoiding potential central
adverse effects.
ELA in diabetes: A promising but still
unexplored guardian peptide
Within the apelinergic system, structural divergence
dictates functional specialization. Apelin isoforms share a
conserved C-terminal receptor, while ELA possesses a distinct
N-terminal architecture, promoting a unique active conformation of
APJ (53,55). Coupled with its persistent
expression in adult vascular endothelium and kidneys, this
structural and signaling distinction positions ELA as a separate,
developmentally-rooted guardian within glucose homeostasis and
diabetic complication pathways, diverging from the primary roles of
apelin in metabolic adaptation (10,52).
Clinical and mechanistic foundations
Consistently reduced plasma ELA levels have been
observed in individuals with T2D (77,78). This is not an isolated
phenomenon. Our previous findings further underscored the clinical
relevance of ELA, demonstrating a close relationship between serum
ELA levels and the development and progression of diabetic
nephropathy (77,79). Consequently, it is hypothesized
that insufficient ELA expression might be a critical factor
underlying systemic glycemic dysregulation and insulin
resistance.
To elucidate the pathophysiological consequences of
diminished ELA levels, researchers have employed various models to
investigate their protective mechanisms from multiple perspectives
(Fig. 2B). Regarding vascular
endothelial protection, under high-glucose conditions, ELA
specifically inhibits the tumor necrosis factor receptor-associated
factor 1/NF-κB axis, mitigating oxidative DNA damage and preserving
vascular integrity (80). In
terms of ameliorating cellular metabolic status, synergistic
effects have been uncovered. ELA acts synergistically with the
classic insulin sensitizer rosiglitazone by specifically activating
peroxisome proliferator-activated receptor γ and suppressing the
expression of its downstream target ferredoxin 1, ultimately
alleviating mitochondrial damage and delaying cellular senescence
(81). Tang et al
(82) offer a novel perspective
on the regulation of lipid metabolism and inflammation. ELA
enhanced the expression levels of ACE and ACE2 in macrophages,
while inhibiting the (pro) renin receptor system, modulating
macrophage polarization to suppress inflammation.
Discrepant therapeutic outcomes in
preclinical research
Despite promising mechanisms, the therapeutic
translation of ELA has yielded contradictory results across models
(10,83). In streptozotocin-induced T1D,
prolonged exogenous administration of ELA-32 reduced peripheral
glucose levels and restored insulin concentrations (83). By contrast, our research using
db/db mice yielded divergent outcomes: Extended ELA-21 treatment
failed to improve fasting blood glucose and glycated hemoglobin
levels (10). It was
hypothesized that the efficacy of ELA is contingent upon the
underlying metabolic milieu. In the insulinopenic environment of
T1D, the actions of ELA may directly counter serious
glucose-induced damage. Conversely, in the db/db model, which is
characterized by profound leptin receptor deficiency, rampant
obesity and systemic insulin resistance, this dominant metabolic
dysregulation may create a signaling environment that overwhelms or
fundamentally alters ELA-APJ signaling. This hypothesis predicts
that the therapeutic window of ELA is narrower than initially
assumed, favoring earlier or less metabolically chaotic disease
stages.
Apelin/ELAAPJ system and diabetic kidney
disease
Diabetic kidney disease (DKD) progression is driven
by a confluence of metabolic, hemodynamic and inflammatory injuries
(84). Hemodynamic imbalance,
tubule-glomerular feedback changes, renal hypoxia, lipotoxicity,
podocyte injury, inflammation, mitochondrial dysfunction, impaired
autophagy and increased sodium and hydrogen exchanger activity are
involved in the occurrence and development of diabetic nephropathy
(84,85). Within this milieu, the
apelin/ELA-APJ system plays a decisive yet divergent role. Apelin
exhibits a stark functional duality, swinging from renoprotection
to injury, while ELA consistently signals toward protection, though
the underlying metabolic environment may modulate its efficacy.
The dual faces of apelin in diabetic
kidney disease
The role of Apelin in DKD remains controversial and
inadequately defined by large-scale clinical studies. Current
evidence reveals a system whose function pivots markedly based on
the pathological context, particularly the diabetes type and the
stage of renal involvement.
Circulating apelin as a marker of
disease
In the human kidney, the apelin system is widely
expressed across renal vasculature, glomeruli and tubules (86). Plasma apelin concentrations in
patients with DKD increased with declining renal function and
correlated positively with albuminuria severity. This elevation may
represent compensatory upregulation, reduced renal clearance, or
both, creating ambiguity for biomarker interpretation. Apelin
levels were independently associated with a decline in eGFR,
positioning it as both a disease monitor and a potential
intervention point (86,87).
Evidence from experimental models
Substantial evidence from animal models supports the
renoprotective role of apelin, particularly under specific
conditions (Fig. 3A). In
insulin-deficient mice, apelin-13 treatment prevents pathological
renal hypertrophy and reduces albuminuria through antioxidant
enzyme upregulation and histone deacetylation-mediated inflammation
suppression (88,89). Even in T2D models, where its role
is more controversial, apelin exhibits protective effects. Apelin
inhibits epithelial-mesenchymal transition (EMT) in podocytes by
suppressing the immunoproteasome subunit β5i, and attenuates
glomerular endothelial fibrosis through a sirtuin 3
(SIRT3)-Krüppel-like factor 15 (KLF15)-dependent pathway (90,91). One report suggested that apelin
increased the activity of large-conductance calcium-activated
potassium (BKCa) channels, promoting the nuclear translocation of
the SP1 transcription factor. This enhanced the expression of
Dickkopf-related protein 1, which inhibited β-catenin nuclear
translocation and subsequent EMT (92). Huang et al (93) proposed that apelin upregulated
the α and β4 subunits of the BKCa channel via the PI3K/AKT axis,
leading to increased NO bioavailability and reduced endothelin-1
expression, thereby mitigating glomerular hypertension. Conditioned
medium from human Wharton's jelly-derived mesenchymal stem cells
improves renal function by upregulating apelin mRNA levels while
downregulating transforming growth factor beta (TGF-β) mRNA levels
(94), whereas quercetin
treatment achieves similar benefits by reducing the expression of
both molecules (95). These
findings collectively established the renoprotective potential of
apelin under hyperglycemic conditions.
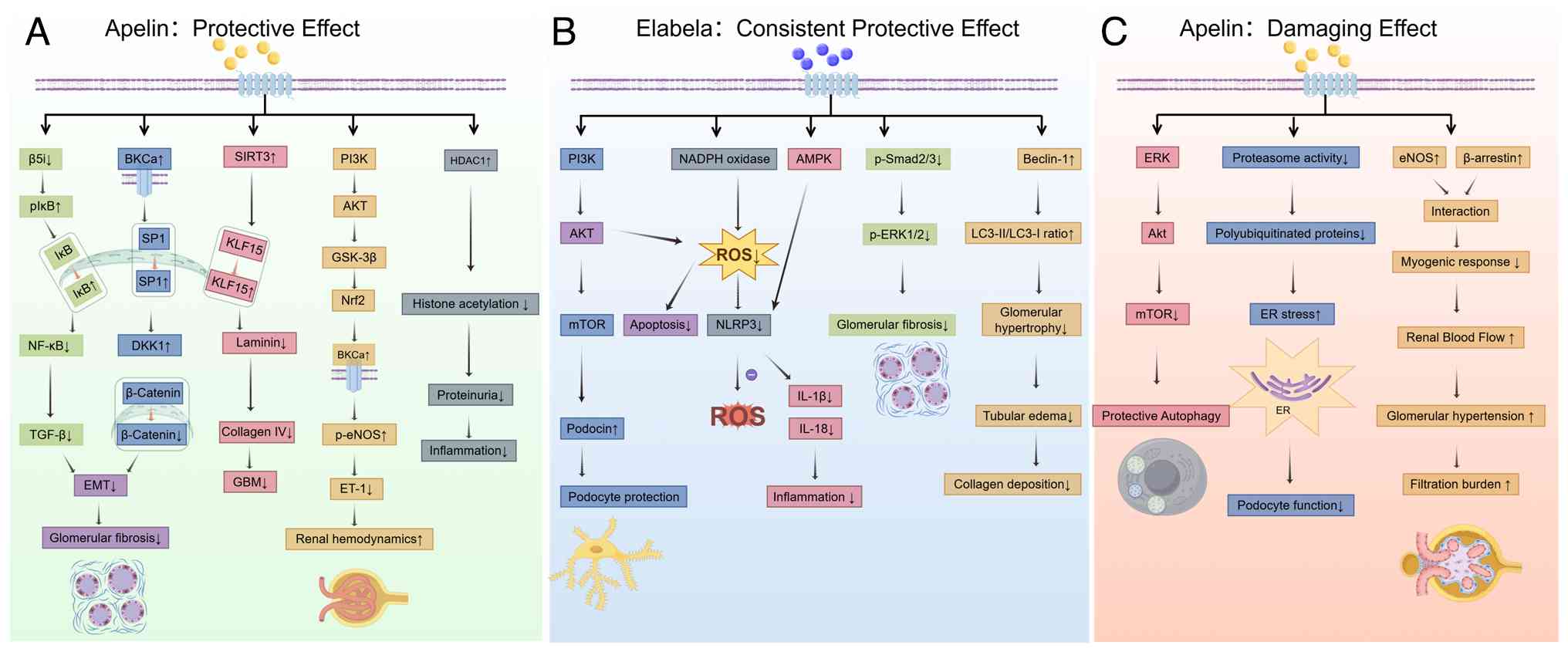 | Figure 3Dual role of apelin and the
protective role of ELA in diabetic kidney disease. (A) Protective
effects of apelin. Apelin can exert both protective effects by
upregulating antioxidant defenses, inhibiting podocyte injury and
fibrosis and improving endothelial function. (B) ELA consistently
demonstrates renoprotective properties by preserving podocytes,
reactivating autophagy in renal tubules, and mitigating
inflammation and oxidative stress. (C) Detrimental effects of
apelin in the diabetic kidney. In certain contexts, apelin exerts
damaging effects by impairing proteasome activity and autophagic
flux, leading to endoplasmic reticulum stress, podocyte dysfunction
and accumulation of polyubiquitinated proteins. Apelin also
disrupts vascular homeostasis through eNOS/β-arrestin interaction,
reducing myogenic response while increasing renal blood flow,
glomerular hypertension and filtration burden. (p)IkB,
(phosphorylated) inhibitor of NF-κB; NF-κB, nuclear factor κB;
TGF-β, transforming growth factor-β; EMT, epithelial-mesenchymal
transition; BKCa, large-conductance calcium-activated potassium
channel; SP1, specificity protein 1; DKK1, dickkopf-related protein
1; SIRT3, sirtuin 3; KLF15, krüppel-like factor 15; GBM, glomerular
basement membrane; PI3K, phosphoinositide 3-kinase; AKT, protein
kinase B; GSK-3β, glycogen synthase kinase-3β; Nrf2, nuclear factor
erythroid 2-related factor 2; p-eNOS, phosphorylated endothelial
nitric oxide synthase; ET-1, endothelin-1; HDAC1, histone
deacetylase 1; mTOR, mammalian target of rapamycin; NADPH,
nicotinamide adenine dinucleotide phosphate; ROS, reactive oxygen
species; AMPK, AMP-activated protein kinase; NLRP3, NOD-like
receptor family pyrin domain containing 3; IL-1β, interleukin-1β;
IL-18, interleukin-18; p-Smad2/3, phosphorylated Smad2/3; p-ERK1/2,
phosphorylated extracellular signal-regulated kinase 1/2;
LC3-II/LC3-I, microtubule-associated protein 1 light chain
3-II/I. |
By contrast, evidence from models of advanced T2D
has revealed the pathogenic potential of apelin (Fig. 3C). In these settings,
characterized by profound insulin resistance and metabolic
dysregulation, apelin signaling appears to become maladaptive. It
impairs podocyte function by inhibiting proteasome activity,
leading to toxic accumulation of polyubiquitinated proteins and
endoplasmic reticulum stress (96). Liu et al (8) further demonstrated that apelin
suppressed protective autophagy in glomeruli via activation of the
Akt and mammalian target of rapamycin (mTOR) pathways, exacerbating
cellular injury. Apelin could disrupt the intrinsic myogenic
response of renal resistance vessels, causing inappropriate
vasodilation that may exacerbate glomerular hyperfiltration and
capillary pressure, and driving DKD progression (97). Paradoxically, in cultured
podocytes, apelin was observed to reduce mTOR phosphorylation and
enhance autophagic activity (8).
The role of Elabela in diabetic kidney
disease
In contrast to the context-dependent duality of
Apelin, accumulating evidence positions ELA as a more uniformly
protective agent in DKD. Its role extends beyond that of a passive
biomarker to an active guardian peptide, with deficiency
contributing to disease progression.
A marker of renal health
Clinical observations consistently associate ELA
with renal protection. Our previous study demonstrated a
progressive decline in serum ELA concentrations throughout DKD
progression, suggesting the potential utility of ELA as a
prognostic biomarker for disease monitoring (77). This relationship has been
independently validated in a Turkish observational study (79). In a previous study, across
different stages of chronic kidney disease, a gradual reduction in
ELA concentrations paralleling eGFR decline was observed, with
multivariate linear regression confirming eGFR as an independent
predictor of serum ELA levels (98). These findings indicate that
reduced ELA levels are not merely a consequence of renal impairment
but may signify a loss of an endogenous protective mechanism.
Mechanistic foundation of
renoprotection
Mechanistic studies have elucidated multiple
protective pathways through which ELA exerts its beneficial effects
(Fig. 3B). In T1D models,
exogenous ELA administration restored the expression of
podocyte-specific proteins, including synaptopodin and podocin, via
activation of the PI3K/Akt/mTOR pathway, thereby stabilizing the
glomerular filtration barrier (83). Tubular injury is now recognized
as a crucial factor in the development of DKD. ELA treatment
reactivates high glucose-inhibited renal tubular autophagy,
promoting survival in diabetic tubules (10). Its anti-fibrotic potential is
linked to predominant renal tubular expression, with specific
knockout of tubular ELA exacerbating renal injury and fibrosis
(99), mediated by the
suppression of Smad2/3 phosphorylation (100). Regarding oxidative stress
regulation, ELA reduces renal reactive oxygen species (ROS)
generation by blocking the NADPH oxidase (NOX2)/ROS/NOD-like
receptor family pyrin domain containing 3 (NLRP3) pathway and
through PI3K/Akt-mediated survival signaling, thereby alleviating
oxidative damage (101,102). Additionally, ELA modulates the
AMPK/NLRP3 signaling axis to inhibit inflammasome activation and
the subsequent release of pro-inflammatory cytokines (103).
Current evidence is predominantly derived from
animal studies, with limited human data available. Existing
clinical investigations are constrained by small sample sizes and
insufficient long-term follow-up. While ELA exhibits clear efficacy
in T1D, in db/db mice has yielded divergent outcomes, with no
statistically significant improvements in core metabolic parameters
(10,83). This discrepancy underscores a
contingency: The efficacy of ELA may be constrained in profoundly
insulin-resistant and dysmetabolic environments, suggesting its
therapeutic window may be favorable in earlier or less
metabolically chaotic stages of disease.
Comparative implications: Ligand
selection based on disease stage
Due to the context-dependent role of apelin,
patients with early disease may be more likely to benefit from
apelin treatment than patients with advanced T2D with severe
insulin resistance (90,94,97). Its signaling flexibility, rooted
in multiple phosphorylation sites and G protein coupling options,
creates opportunity but also the risk of unintended pathway
activation (53,54). By contrast, the protective action
of ELA likely traces back to the receptor level. As aforementioned,
ELA induces distinct phosphorylation patterns on the receptor's
C-terminus (54). ELA may exert
its effects mainly via transient PI3K/Akt-driven survival signaling
and preserve autophagy (10,83,102). Rather than examining whether
the APJ system is protective or harmful, it could be explored under
what conditions, and through which ligand, it assumes one role or
the other. The convergence of both ligands on shared downstream
effectors, such as SIRT3, also suggests that the most durable
strategy may lie not in choosing one ligand over the other, but in
identifying nodes common to both protective pathways (11,90).
Apelin/ELA-APJ system and diabetic
cardiomyopathy
Diabetic cardiomyopathy (DCM) is characterized by
myocardial dysfunction independent of coronary artery disease or
hypertension, driven by metabolic disturbances, oxidative stress,
inflammation, and microvascular impairment (104). Within this pathological
framework, the Apelin/ELA-APJ system, widely expressed in
cardiomyocytes, vascular endothelium, and smooth muscle, emerges as
a critical endogenous regulator (11,14,49,64). In contrast to its more
paradoxical roles in renal and retinal complications, evidence in
DCM predominantly points toward a protective adaptation, though
nuanced by clinical context and therapeutic modulation.
The role of Apelin in diabetic
cardiomyopathy
Apelin orchestrates a multifaceted defense against
DCM through coordinated modulation of metabolism, redox balance,
and microvascular function. Its clinical relevance, however, is
shaped by a complex interplay of disease subtype, comorbidities,
and pharmacologic interventions.
Fundamental protective mechanisms
The mitochondrial deacetylase SIRT3 emerges as a
pivotal mediator of the antioxidant and anti-inflammatory effects
of apelin (Fig. 4A). Apelin
treatment elevates myocardial SIRT3 expression in DCM models,
concurrently promoting the expression of angiogenesis-related
factors (105,106). In diabetic myocardial
infarction models, apelin activates protective autophagy through
SIRT3-dependent mechanisms, which in turn inhibits NADPH
oxidase-driven ROS generation and blocks the inflammatory pathway
(106,107). Genetic ablation of SIRT3
completely abolishes these effects, unequivocally establishing its
indispensable role (105-107).
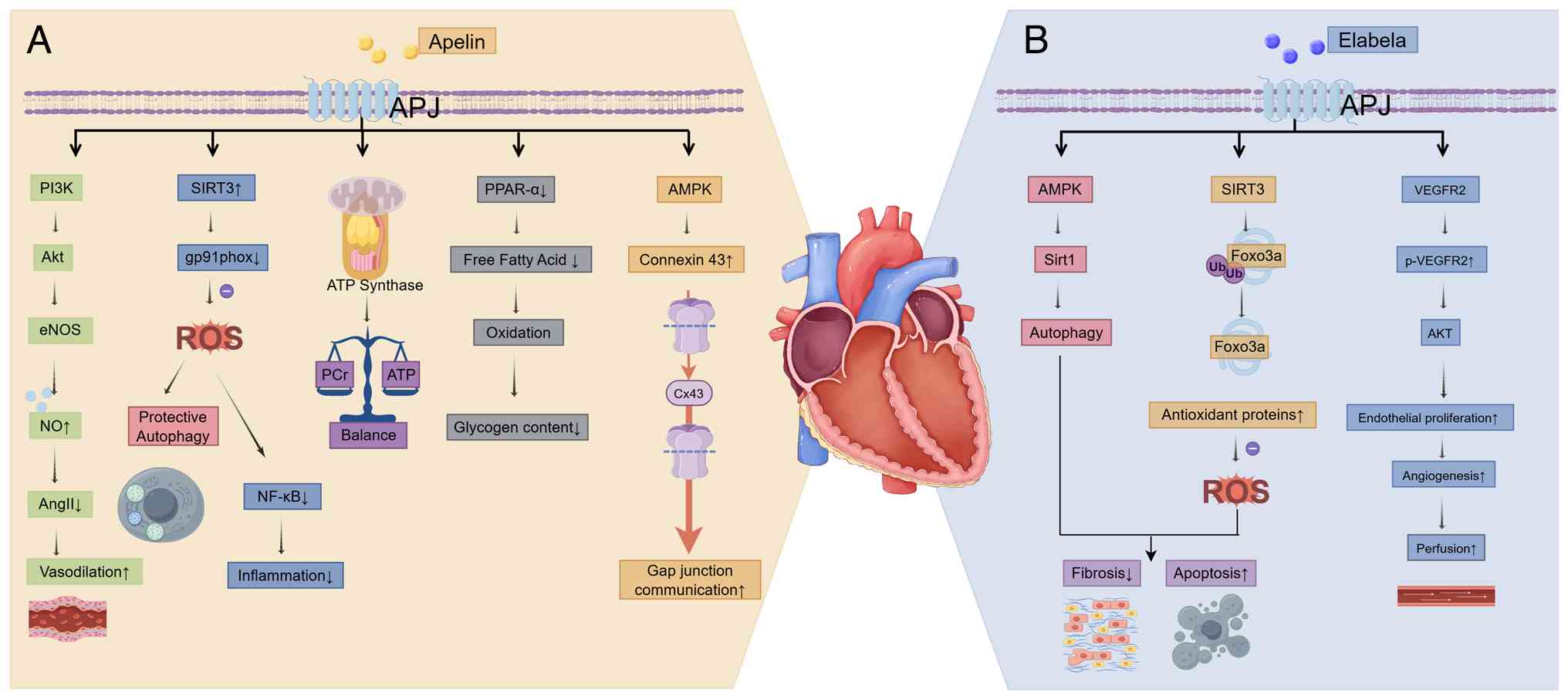 | Figure 4Protective mechanisms of the
apelin/ELA-APJ system against diabetic cardiomyopathy. In the
diabetic heart, both (A) apelin and (B) ELA mitigate key
pathological processes, including metabolic derangement, oxidative
stress, inflammation, fibrosis and microvascular rarefaction. APJ,
apelin receptor; PI3K, phosphoinositide 3-kinase; Akt, protein
kinase B; eNOS, endothelial nitric oxide synthase; AngII,
angiotensin II; SIRT3, sirtuin 3; gp91phox, glycoprotein 91
phagocyte oxidase; ATP, adenosine triphosphate; PCr,
phosphocreatine; PPAR-α, peroxisome proliferator-activated receptor
α; AMPK, AMP-activated protein kinase; Cx43, connexin 43; SIRT1/3,
sirtuin 1/3; Foxo3a, forkhead box O3a; Ub, ubiquitin; VEGFR2,
vascular endothelial growth factor receptor 2. |
Beyond the SIRT3 pathway, apelin protects vascular
function in diabetes by counteracting angiotensin II-induced
vasoconstriction and enhancing endothelium-dependent vasodilation
via the PI3K/Akt-endothelial nitric oxide synthase pathway
(108). Apelin also attenuates
cardiac microvascular inflammation and adhesion molecule expression
in T2D mice via suppression of the NF-κB pathway (64). At the cellular level, it
upregulates connexin 43 in cardiomyocytes under high glucose
conditions, thereby improving gap junctional communication and
electrical stability (109).
Apelin also mediates pro-angiogenic signals from regulatory T
cells, promoting microvascular density and perfusion in DCM
(110).
From a metabolic perspective, chronic apelin-13
treatment reduces myocardial free fatty acid and glycogen content
in T2D rats, suppressing excessive fatty acid oxidation (45). Complementing these findings,
apelin-12 restores the phosphocreatine/ATP ratio, indicating
improved energy reserve (111).
Furthermore, apelin restores erythrocyte deformability and enhances
the myocardial antioxidant defense capacity (9,112,113). These preclinical findings paint
a compelling picture of comprehensive cardioprotection. However,
the transition to human disease reveals complexities that temper
enthusiasm for uncomplicated therapeutic translation.
Clinical complexities and translational
insights
Population studies have produced more nuanced and
notably inconsistent results. While reduced apelin levels are
associated with cardiac remodeling severity in hypertensive
patients with diabetes, differences between diabetic patients with
and without complications frequently fail to reach statistical
significance (114,115). Associations have been observed
in children with T1D and Egyptian diabetic patients, while apelin
levels exhibit no association with carotid intima-media thickness
in populations predisposed to T2D (116,117). These discrepancies likely
reflect differences in diabetes duration, the metabolic profile of
T1D vs. T2D and the confounding effects of concurrent
medications.
Therapeutic interventions further complicate this
narrative. Pioglitazone suppresses apelin transcriptional activity
by inducing KLF4 expression (118), while dapagliflozin increases
serum apelin levels (119).
Non-pharmacologic interventions such as physical exercise could
elevate apelin levels, as well as improve cardiovascular risk
markers (120). These
diametrically opposed drug effects not only reveal differential
regulation of the apelin system by various glucose-lowering agents
but may partially explain their distinct cardiovascular protective
profiles. The association between increased apelin levels following
dapagliflozin treatment and improved left ventricular function
offers novel insights into the cardiovascular benefits of this drug
class.
In biomarker development, the apelin/N-terminal
pro-brain natriuretic peptide ratio has demonstrated superior
predictive value for heart failure with preserved ejection fraction
in diabetic patients compared with either biomarker alone,
suggesting that single biomarkers may be insufficient to reflect
complex cardiovascular pathological states (121). The lack of significant apelin
level differences between patients with tight compared with poor
glycemic control suggests system regulation relating more
fundamentally to the presence of diabetes itself rather than
glycemic management specifically (122).
Role of ELA in DCM
Direct research on ELA in the specific context of
DCM is nascent, but a compelling protective role may be inferred
from its well-established actions in related cardiovascular
pathologies and is supported by emerging direct evidence. These
findings collectively position ELA as a promising endogenous
guardian against diabetic heart injury.
Mechanistic insights from broad
cardiovascular protection
The core pathological processes of DCM, including
severe oxidative stress, inflammatory responses, endothelial
dysfunction and cellular apoptosis, are also central to other
cardiovascular conditions (85,104). In non-diabetic models, ELA has
been demonstrated to intervene in these processes effectively
(14,18). In myocardial ischemia-reperfusion
injury, ELA exerts cardioprotective effects by activating PI3K/AKT
signaling to enhance cell survival and mitochondrial function
(123), while suppressing
TGFβ1-Smad2/3 and ERK/hypoxia-inducible factor-1 signaling to
inhibit fibroblast migration (124,125). ELA also modulates the
AMPK-Sirt1 axis to regulate apoptosis and autophagy (124). Regarding atherosclerosis,
clinical observations have indicated reduced circulating levels of
ELA in patients with the condition, and ELA itself possessed
functions that stabilized the endothelium and inhibited smooth
muscle cell proliferation (14,126). The related peptide apelin-13
has also been shown to inhibit macrophage foam cell formation and
promote cholesterol efflux via the APJ receptor (127). Given the exacerbation of these
same pathways in diabetes, dysregulation of the ELA-APJ axis likely
contributes to accelerated cardiovascular damage in DCM, while ELA
supplementation represents a rational therapeutic strategy.
Emerging direct evidence in diabetic
contexts
Preliminary research directly investigating ELA in
diabetic heart disease have begun to illuminate its protective
mechanisms (Fig. 4B). In the
diabetic heart, ELA activates SIRT3, promoting the deacetylation of
the transcription factor Forkhead Box O3a (Foxo3a) and subsequent
upregulation of antioxidant defenses. This pathway mitigates
oxidative stress, thereby attenuating cardiomyocyte fibrosis and
apoptosis (11). ELA expression
is upregulated in diabetic ischemic tissues, where it promotes
endothelial cell proliferation by upregulating vascular endothelial
growth factor receptor 2 (VEGFR2) and its phosphorylation levels,
activating the downstream AKT signaling pathway (128). Functional experiments have
confirmed that specific knockout of ELA in endothelial cells
impaired blood flow recovery and capillary density in ischemic
tissues, highlighting its essential role in vascular repair
(129). This inducibility
contrasts with the relatively constitutive expression of ELA in
healthy endothelium and may represent an attempt to activate
protective signaling pathways that becomes inadequate in sustained
diabetes.
Complementary rather than redundant
The cardiac APJ narrative, while less dichotomous
than renal or retinal presentations, reveals equally important
principles for therapeutic development. The two ligands engage
SIRT3 as a common protective node, yet arrive through distinct
signaling architectures with different context sensitivities
(11,107,130). Circulating apelin levels rise
in advanced disease, but whether this represents exhausted
compensation or active pathogenesis remains unclear (114). Divergent effects of
glucose-lowering approaches on apelin levels also caution against
inferring causality from cross-sectional associations (119,120). For ELA, the protective signal
is consistent and the evidence base remains thin; whether its
efficacy becomes contextually constrained in the severely
insulin-resistant or chronically remodeled heart has not been
systematically tested (11,128). The challenge in DCM is
therefore not whether to activate or inhibit, but how to engage the
right pathways with precision. Whether through apelin analogs in
metabolically normalized patients, ELA supplementation for vascular
protection or synergistic pairing, represents a unifying
therapeutic goal.
Apelin/ELA-APJ system and diabetic retinal
disease
Diabetic retinopathy (DR), a leading cause of
vision loss, evolves from early neurovascular dysfunction to
advanced proliferative stages characterized by pathological
angiogenesis and fibrosis (131,132). Expanding beyond the traditional
focus on vascular endothelial cells, recent studies reveal that
diabetes affects neurons, glial cells and other retinal cell types
(133,134), prompting the term 'diabetic
retinal disease' (DRD) to describe these broader structural and
functional alterations (135).
Research on the apelin/ELA-APJ system in this context reveals a
paradigm of context-dependency. Its role is not static but
undergoes a dramatic reversal contingent upon disease stage,
offering a unique lens through which to understand how a single
signaling axis can be both a protector and a perpetrator within the
same organ.
The role of apelin in diabetic retinal
disease
Apelin's function in DRD is not merely dualistic
but dynamically inverted across the disease continuum. It acts as a
protective guardian in early stages, yet transforms into a
pathological driver in proliferative diabetic retinopathy (PDR).
This switch represents one of the striking examples of
context-dependent signaling in diabetic complications.
Pathological driver in proliferative
DRD
In PDR, apelin and its receptor are consistently
upregulated in vitreous, fibrovascular and epiretinal membranes
(136-138). Immunohistochemistry has
localized apelin to endothelial, glial and epithelial components of
these pathological tissues, implicating it directly in
neovascularization and fibrosis (138).
At the molecular and cellular level, high glucose
conditions upregulate apelin expression in human retinal pigment
epithelial (RPE) cells and apelin subsequently promotes RPE cell
proliferation, migration and collagen I expression through
activation of the PI3K/Akt and MEK/Erk signaling pathways (62). Similarly, in diabetic models,
apelin activates the Janus kinase 2/STAT3 pathway in Müller glial
cells to drive fibrosis (61,139). These findings establish apelin
as an active participant in the fibrovascular proliferation that
characterizes advanced disease.
A protective guardian in early-stage
DRD
In contrast to its apparently detrimental role in
advanced PDR, apelin exhibits protective effects in early DRD
stages or specific contexts. In T2D mouse models, apelin enhances
blood-retinal barrier integrity by upregulating tight junction
proteins via the PI3K/Akt pathway, thereby reducing vascular
leakage. Apelin concurrently mitigates retinal inflammation by
downregulating adhesion molecules and suppressing NF-κB activation
(140). Additionally, apelin-13
exhibits protective effects on retinal ganglion cells by activating
the PI3K/Akt pathway to inhibit apoptosis, enhancing the pentose
phosphate pathway enzyme activity to increase NADPH and ATP
production while reducing oxidative stress, and inhibiting
mitochondrial cytochrome c release (141). These actions collectively
preserve the neurovascular unit in the face of early diabetic
insult.
The Role of ELA in DRD
In contrast to the stage-dependent functional
reversal of Apelin, emerging research on ELA in DRD suggests a
profile that is more uniformly associated with vascular protection,
particularly against oxidative injury. While direct evidence in
diabetic models remains less extensive, convergent findings from
clinical observations and related disease models position ELA as a
compelling candidate for stabilizing the retinal vasculature.
Evidence supporting the vascular
protective role
Clinical studies indicated that serum ELA levels
were elevated in patients with PDR compared to those without
retinopathy, a finding that coincided with longer diabetes duration
in the PDR group (142). This
correlation invites investigation into whether elevated ELA
represents a compensatory protective response to severe vascular
stress.
Mechanistic insights were primarily derived from
the oxygen-induced retinopathy model, which replicates the
obliterative and ischemic phases relevant to DRD. During the
obliterative phase, exogenous ELA reduced avascular area and
promoted physiological retinal vessel regrowth by inhibiting
ferroptosis through modulation of the xCT/GPX4 axis, thereby
preserving mitochondrial function and endothelial cell survival
under ischemic stress (143).
Given that oxidative stress is a cornerstone of DRD
pathophysiology, these findings provide a strong rationale for
ELA's potential to protect retinal microvessels from
hyperglycemia-induced degeneration.
Reasons why ligands diverge
The functional shift of apelin from a guardian to a
driver likely reflects changes in the retinal cellular environment
and signaling landscape as DRD progresses. In early stages,
characterized by metabolic stress and oxidative damage, apelin
signaling through pro-survival pathways in endothelial cells and
neurons may predominate (140).
In the hypoxic, inflammatory milieu of PDR, its signaling may be
shunted toward fibrotic pathways (61). ELA does not undergo a similar
inversion and instead appears to be consistently protective. ELA
induces a distinct APJ phosphorylation barcode and transient signal
kinetics, whereas apelin favors sustained β-arrestin signaling
(54).
This bias may render ELA less susceptible to
pathological rerouting, even in a hostile microenvironment. Whether
the protective capacity of ELA is also overwhelmed in late stages,
or whether its elevation in patient vitreous remains functionally
protective, has not been directly tested. This staged, personalized
approach to APJ modulation, informed by mechanistic understanding
of ligand-specific signaling, offers a path toward therapeutic
utility in a disease where previous attempts at pathway
manipulation have often failed due to oversimplification.
Current challenges and future
perspectives
While the present review focused on the classic
triad of diabetic complications (cardiomyopathy, nephropathy and
retinopathy), emerging evidence suggests that the APJ axis also
influences diabetic neuropathy and wound healing (80,144). Preliminary studies have
indicated that apelin may promote neuron survival and vascular
regeneration, potentially mitigating diabetic neuropathy (145,146). Apelin may alleviate
diabetes-associated hearing loss by reducing cochlear endoplasmic
reticulum stress and mitochondrial dysfunction (147). However, compared with the
robust data in the heart and kidneys, the mechanisms in these
tissues remain under-characterized. Future research should
determine whether the protective efficacy observed in
cardiovascular tissues translates to the nervous system or if the
pro-angiogenic risks, as seen in the retina, outweigh the benefits
(61,137,140,141).
Apelin epitomizes this duality, acting as an
insulin sensitizer yet a potential central disruptor, a guardian in
early diabetic complications but a driver in advanced proliferative
states (65,76,90,97). By contrast, ELA exhibits a more
consistent protective profile in diabetic kidney, heart and retinal
models, although its efficacy may be attenuated in profound
insulin-resistant states, highlighting its own context-dependency
(90,93,97). The divergent roles of apelin and
ELA likely arise from ligand-specific signaling bias, differential
cellular expression and distinct interactions with the
phosphorylation of the receptor.
Despite therapeutic promise, several hurdles remain
for clinical translation. To overcome the rapid degradation of
native apelin peptides, researchers have developed stabilized
analogues with extended half-lives and enhanced in vivo
efficacy (148-150). More advanced strategies include
gene and cell-based therapies, such as engineering mesenchymal stem
cells to serve as sustained Apelin delivery platforms, which have
shown promise in rodent models (7,151). However, validation has largely
been confined to rodent models, and a loss of therapeutic efficacy
may be observed during chronic exposure. Given the
heterodimerization between APJ and AT1R, therapeutic strategies
must account for the patient's concurrent use of ACE inhibitors or
angiotensin receptor blockers, which are standard-of-care in
diabetes (55,58).
In summary, the apelin/ELA-APJ system represents a
double-edged sword in diabetes management. The complexity of the
apelin/ELA-APJ system constitutes its great strength as a
multifaceted modulator. Embracing this complexity and guiding
research with a principle of precision intervention will be
essential to fully unlock its potential and offer a novel,
integrative strategy to combat the multifaceted diabetic
syndrome.
Availability of data and materials
Not applicable.
Authors' contributions
ZH and JZ were responsible for writing the text and
completing the figures. MS, JC, AL and YY were responsible for
researching the data, discussing the content. HZ was responsible
for reviewing and editing the manuscript prior to submission. Data
authentication is not applicable. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Abbreviations:
|
ACE2
|
angiotensin-converting enzyme 2
|
|
AMPK
|
AMP-activated protein kinase
|
|
BKCa
|
large conductance calcium-activated
potassium
|
|
DM
|
diabetes mellitus
|
|
ELA
|
elabela
|
|
EMT
|
epithelial-mesenchymal transition
|
|
Foxo3a
|
forkhead box O3a
|
|
GLUT4
|
glucose transporter 4
|
|
GPCR
|
G protein-coupled receptor
|
|
KLF15
|
krüppel-like factor 15
|
|
mTOR
|
mammalian target of rapamycin
|
|
NADPH
|
nicotinamide adenine dinucleotide
phosphate
|
|
NF-κB
|
nuclear factor-k-gene binding
|
|
NLRP3
|
NOD-like receptor family pyrin domain
containing 3
|
|
NO
|
nitric oxide
|
|
NOX2
|
NADPH oxidase 2/gp91phox
|
|
PI3K
|
phosphatidylinositol 3-kinase
|
|
RAS
|
renin-angiotensin system
|
|
ROS
|
reactive oxygen species
|
|
SIRT3
|
sirtuin 3
|
|
TGF-β
|
transforming growth factor β
|
|
VEGFR2
|
vascular endothelial growth factor
receptor 2
|
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the Science and
Technology Department of Jiangsu Province (grant no. BE2023745) and
Health Commission of Jiangsu Province (grant no. H2023137) to
HZ.
References
|
1
|
Lytrivi M, Tong Y, Virgilio E, Yi X and
Cnop M: Diabetes mellitus and the key role of endoplasmic reticulum
stress in pancreatic β cells. Nat Rev Endocrinol. 21:546–563. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen X, Zhang L and Chen W: Global,
regional, and national burdens of type 1 and type 2 diabetes
mellitus in adolescents from 1990 to 2021, with forecasts to 2030:
A systematic analysis of the global burden of disease study 2021.
BMC Med. 23:482025. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ali MK, Pearson-Stuttard J, Selvin E and
Gregg EW: Interpreting global trends in type 2 diabetes
complications and mortality. Diabetologia. 65:3–13. 2022.
View Article : Google Scholar
|
|
4
|
Zhang Y, Ni Q, Chen Y, Pei Z and Xu X: The
molecular mechanisms of disulfidptosis and its role in diabetes
mellitus and its complications. Biomed Pharmacother.
191:1184992025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
García-Hermoso A, Ramírez-Vélez R, Díez J,
González A and Izquierdo M: Exercise training-induced changes in
exerkine concentrations may be relevant to the metabolic control of
type 2 diabetes mellitus patients: A systematic review and
meta-analysis of randomized controlled trials. J Sport Health Sci.
12:147–157. 2023. View Article : Google Scholar :
|
|
6
|
Song K, Yang X, An G, Xia X, Zhao J, Xu X,
Wan C, Liu T, Zheng Y, Ren S, et al: Targeting APLN/APJ restores
blood-testis barrier and improves spermatogenesis in murine and
human diabetic models. Nat Commun. 13:73352022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cui J, Wang M, Zhang W, Sun J, Zhang Y,
Zhao L, Hong Z, Li D, Huang YX, Zhang N and Chen Y: Enhancing
insulin sensitivity in type 2 diabetes mellitus using apelin-loaded
small extracellular vesicles from Wharton's jelly-derived
mesenchymal stem cells: A novel therapeutic approach. Diabetol
Metab Syndr. 16:842024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu Y, Zhang J, Wang Y and Zeng X: Apelin
involved in progression of diabetic nephropathy by inhibiting
autophagy in podocytes. Cell Death Dis. 8:e30062017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
An S, Wang X, Shi H, Zhang X, Meng H, Li
W, Chen D and Ge J: Apelin protects against ischemia-reperfusion
injury in diabetic myocardium via inhibiting apoptosis and
oxidative stress through PI3K and p38-MAPK signaling pathways.
Aging (Albany N Y). 12:25120–25137. 2020.
|
|
10
|
Zheng X, Yin L, Song J, Chen J, Gu W, Shi
M and Zhang H: ELABELA protects against diabetic kidney disease by
activating high glucose-inhibited renal tubular autophagy. J Biomed
Res. 37:460–469. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li C, Miao X, Wang S, Liu Y, Sun J, Liu Q,
Cai L and Wang Y: Elabela may regulate SIRT3-mediated inhibition of
oxidative stress through Foxo3a deacetylation preventing
diabetic-induced myocardial injury. J Cell Mol Med. 25:323–332.
2021. View Article : Google Scholar :
|
|
12
|
Murali S and Aradhyam GK:
Structure-function relationship and physiological role of apelin
and its G protein coupled receptor. Biophys Rev. 15:127–143. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mughal A and O'Rourke ST: Vascular effects
of apelin: Mechanisms and therapeutic potential. Pharmacol Ther.
190:139–147. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chapman FA, Maguire JJ, Newby DE,
Davenport AP and Dhaun N: Targeting the apelin system for the
treatment of cardiovascular diseases. Cardiovasc Res.
119:2683–2696. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tatemoto K, Hosoya M, Habata Y, Fujii R,
Kakegawa T, Zou MX, Kawamata Y, Fukusumi S, Hinuma S, Kitada C, et
al: Isolation and characterization of a novel endogenous peptide
ligand for the human APJ receptor. Biochem Biophys Res Commun.
251:471–476. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
O'Dowd BF, Heiber M, Chan A, Heng HH, Tsui
LC, Kennedy JL, Shi X, Petronis A, George SR and Nguyen T: A human
gene that shows identity with the gene encoding the angiotensin
receptor is located on chromosome 11. Gene. 136:355–360. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kleinz MJ and Davenport AP: Emerging roles
of apelin in biology and medicine. Pharmacol Ther. 107:198–211.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Williams TL, Verdon G, Kuc RE, Currinn H,
Bender B, Solcan N, Schlenker O, Macrae RGC, Brown J, Schütz M, et
al: Structural and functional determination of peptide versus small
molecule ligand binding at the apelin receptor. Nat Commun.
15:107142024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Than A, Tee WT and Chen P: Apelin
secretion and expression of apelin receptors in 3T3-L1 adipocytes
are differentially regulated by angiotensin type 1 and type 2
receptors. Mol Cell Endocrinol. 351:296–305. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
De Mota N, Lenkei Z and Llorens-Cortès C:
Cloning, pharmacological characterization and brain distribution of
the rat apelin receptor. Neuroendocrinology. 72:400–407. 2000.
View Article : Google Scholar
|
|
21
|
Ishida J, Hashimoto T, Hashimoto Y,
Nishiwaki S, Iguchi T, Harada S, Sugaya T, Matsuzaki H, Yamamoto R,
Shiota N, et al: Regulatory roles for APJ, a seven-transmembrane
receptor related to angiotensin-type 1 receptor in blood pressure
in vivo. J Biol Chem. 279:26274–26279. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hosoya M, Kawamata Y, Fukusumi S, Fujii R,
Habata Y, Hinuma S, Kitada C, Honda S, Kurokawa T, Onda H, et al:
Molecular and functional characteristics of APJ. Tissue
distribution of mRNA and interaction with the endogenous ligand
apelin. J Biol Chem. 275:21061–21067. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kawamata Y, Habata Y, Fukusumi S, Hosoya
M, Fujii R, Hinuma S, Nishizawa N, Kitada C, Onda H, Nishimura O
and Fujino M: Molecular properties of apelin: Tissue distribution
and receptor binding. Biochim Biophys Acta. 1538:162–171. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhen EY, Higgs RE and Gutierrez JA:
Pyroglutamyl apelin-13 identified as the major apelin isoform in
human plasma. Anal Biochem. 442:1–9. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gerbier R, Leroux V, Couvineau P,
Alvear-Perez R, Maigret B, Llorens-Cortes C and Iturrioz X: New
structural insights into the apelin receptor: Identification of key
residues for apelin binding. FASEB J. 29:314–322. 2015. View Article : Google Scholar
|
|
26
|
Chapman FA, Melville V, Godden E, Morrison
B, Bruce L, Maguire JJ, Davenport AP, Newby DE and Dhaun N:
Cardiovascular and renal effects of apelin in chronic kidney
disease: A randomised, double-blind, placebo-controlled, crossover
study. Nat Commun. 15:83872024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Foris V, Kovacs G, Avian A, Bálint Z,
Douschan P, Ghanim B, Klepetko W, Olschewski A and Olschewski H:
Apelin-17 to diagnose idiopathic pulmonary arterial hypertension: A
biomarker study. Front Physiol. 13:9862952023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Flahault A, Girault-Sotias PE, Keck M,
Alvear-Perez R, De Mota N, Estéoulle L, Ramanoudjame SM, Iturrioz
X, Bonnet D and Llorens-Cortes C: A metabolically stable apelin-17
analog decreases AVP-induced antidiuresis and improves
hyponatremia. Nat Commun. 12:3052021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
De Mota N, Reaux-Le Goazigo A, El Messari
S, Chartrel N, Roesch D, Dujardin C, Kordon C, Vaudry H, Moos F and
Llorens-Cortes C: Apelin, a potent diuretic neuropeptide
counteracting vasopressin actions through inhibition of vasopressin
neuron activity and vasopressin release. Proc Natl Acad Sci USA.
101:10464–10469. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Saiki H, Hayashi Y, Yoshii S, Kimura E,
Nakagawa K, Kato M, Uema R, Inoue T, Sakatani A, Yoshihara T, et
al: The apelin-apelin receptor signaling pathway in fibroblasts is
involved in tumor growth via p53 expression of cancer cells. Int J
Oncol. 63:1392023. View Article : Google Scholar
|
|
31
|
Bai B, Tang J, Liu H, Chen J, Li Y and
Song W: Apelin-13 induces ERK1/2 but not p38 MAPK activation
through coupling of the human apelin receptor to the Gi2 pathway.
Acta Biochim Biophys Sin (Shanghai). 40:311–318. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Macaluso NJ and Glen RC: Exploring the
'RPRL' motif of apelin-13 through molecular simulation and
biological evaluation of cyclic peptide analogues. ChemMedChem.
5:1247–1253. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chng SC, Ho L, Tian J and Reversade B:
ELABELA: A hormone essential for heart development signals via the
apelin receptor. Dev Cell. 27:672–680. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Read C, Nyimanu D, Williams TL, Huggins
DJ, Sulentic P, Macrae RGC, Yang P, Glen RC, Maguire JJ and
Davenport AP: International union of basic and clinical
pharmacology. CVII. Structure and pharmacology of the apelin
receptor with a recommendation that elabela/toddler is a second
endogenous peptide ligand. Pharmacol Rev. 71:467–502. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Charo DN, Ho M, Fajardo G, Kawana M, Kundu
RK, Sheikh AY, Finsterbach TP, Leeper NJ, Ernst KV, Chen MM, et al:
Endogenous regulation of cardiovascular function by apelin-APJ. Am
J Physiol Heart Circ Physiol. 297:H1904–H1913. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang J, Zhou Y, Wu C, Wan Y, Fang C, Li
J, Fang W, Yi R, Zhu G, Li J and Wang Y: Characterization of the
apelin/elabela receptors (APLNR) in chickens, turtles, and
zebrafish: Identification of a novel apelin-specific receptor in
teleosts. Front Endocrinol (Lausanne). 9:7562018. View Article : Google Scholar
|
|
37
|
Eberlé D, Marousez L, Hanssens S, Knauf C,
Breton C, Deruelle P and Lesage J: Elabela and Apelin actions in
healthy and pathological pregnancies. Cytokine Growth Factor Rev.
46:45–53. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang Z, Tang J, Song J, Xie M, Liu Y,
Dong Z, Liu X, Li X, Zhang M, Chen Y, et al: Elabela alleviates
ferroptosis, myocardial remodeling, fibrosis and heart dysfunction
in hypertensive mice by modulating the IL-6/STAT3/GPX4 signaling.
Free Radic Biol Med. 181:130–142. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Couvineau P, Llorens-Cortes C and Iturrioz
X: Elabela/toddler and apelin bind differently to the apelin
receptor. FASEB J. 34:7989–8000. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Murza A, Sainsily X, Coquerel D, Côté J,
Marx P, Besserer-Offroy É, Longpré JM, Lainé J, Reversade B,
Salvail D, et al: Discovery and structure-activity relationship of
a bioactive fragment of ELABELA that modulates vascular and cardiac
functions. J Med Chem. 59:2962–2972. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kleinz MJ and Davenport AP:
Immunocytochemical localization of the endogenous vasoactive
peptide apelin to human vascular and endocardial endothelial cells.
Regul Pept. 118:119–125. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kang Y, Kim J, Anderson JP, Wu J, Gleim
SR, Kundu RK, McLean DL, Kim JD, Park H, Jin SW, et al: Apelin-APJ
signaling is a critical regulator of endothelial MEF2 activation in
cardiovascular development. Circ Res. 113:22–31. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Habibian M, Biniaz S and Moosavi SJ:
Protective role of short-term aerobic exercise against zinc oxide
nanoparticles-induced cardiac oxidative stress via possible changes
of apelin, angiotensin II/angiotensin II type I signalling pathway.
Cardiovasc Toxicol. 23:177–184. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tanday N, Irwin N, Moffett RC, Flatt PR
and O'Harte FPM: Beneficial actions of a long-acting apelin
analogue in diabetes are related to positive effects on islet cell
turnover and transdifferentiation. Diabetes Obes Metab.
22:2468–2478. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Feng J, Zhao H, Du M and Wu X: The effect
of apelin-13 on pancreatic islet beta cell mass and myocardial
fatty acid and glucose metabolism of experimental type 2 diabetic
rats. Peptides. 114:1–7. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chae SA, Du M, Son JS and Zhu MJ: Exercise
improves homeostasis of the intestinal epithelium by activation of
apelin receptor-AMP-activated protein kinase signalling. J Physiol.
601:2371–2389. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Fournel A, Drougard A, Duparc T, Marlin A,
Brierley SM, Castro J, Le-Gonidec S, Masri B, Colom A, Lucas A, et
al: Apelin targets gut contraction to control glucose metabolism
via the brain. Gut. 66:258–269. 2017. View Article : Google Scholar :
|
|
48
|
Shao ZQ, Dou SS, Zhu JG, Wang HQ, Wang CM,
Cheng BH and Bai B: Apelin-13 inhibits apoptosis and excessive
autophagy in cerebral ischemia/reperfusion injury. Neural Regen
Res. 16:1044–1051. 2021. View Article : Google Scholar :
|
|
49
|
Frump AL, Albrecht M, Yakubov B,
Breuils-Bonnet S, Nadeau V, Tremblay E, Potus F, Omura J, Cook T,
Fisher A, et al: 17β-Estradiol and estrogen receptor α protect
right ventricular function in pulmonary hypertension via BMPR2 and
apelin. J Clin Invest. 131:e1294332021. View Article : Google Scholar
|
|
50
|
Fischer C, Lamer T, Wang W, McKinnie SMK,
Iturrioz X, Llorens-Cortes C, Oudit GY and Vederas JC: Plasma
kallikrein cleaves and inactivates apelin-17: Palmitoyl- and
PEG-extended apelin-17 analogs as metabolically stable blood
pressure-lowering agents. Eur J Med Chem. 166:119–124. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang W, McKinnie SM, Farhan M, Paul M,
McDonald T, McLean B, Llorens-Cortes C, Hazra S, Murray AG, Vederas
JC and Oudit GY: Angiotensin-converting enzyme 2 metabolizes and
partially inactivates Pyr-apelin-13 and apelin-17: Physiological
effects in the cardiovascular system. Hypertension. 68:365–377.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chapman NA, Dupré DJ and Rainey JK: The
apelin receptor: Physiology, pathology, cell signalling, and ligand
modulation of a peptide-activated class A GPCR. Biochem Cell Biol.
92:431–440. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Bai B, Jiang Y, Cai X and Chen J: Dynamics
of apelin receptor/G protein coupling in living cells. Exp Cell
Res. 328:401–409. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Chen J, Chen X, Li S, Jiang Y, Mao H,
Zhang R, Ji B, Yan M, Cai X and Wang C: Individual phosphorylation
sites at the C-terminus of the apelin receptor play different roles
in signal transduction. Redox Biol. 36:1016292020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hu S, Wang D, Liu W, Wang Y, Chen J and
Cai X: Apelin receptor dimer: Classification, future prospects, and
pathophysiological perspectives. Biochim Biophys Acta Mol Basis
Dis. 1870:1672572024. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Jiang Y, Yan M, Wang C, Wang Q, Chen X,
Zhang R, Wan L, Ji B, Dong B, Wang H and Chen J: The effects of
apelin and elabela ligands on apelin receptor distinct signaling
profiles. Front Pharmacol. 12:6305482021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Chen J, Wang Z, Zhang R, Yin H, Wang P,
Wang C and Jiang Y: Heterodimerization of apelin and opioid
receptor-like 1 receptors mediates apelin-13-induced G protein
biased signaling. Life Sci. 328:1218922023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zhang X, Zhang S, Wang M, Chen H and Liu
H: Advances in the allostery of angiotensin II type 1 receptor.
Cell Biosci. 13:1102023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Duparc T, Colom A, Cani PD, Massaly N,
Rastrelli S, Drougard A, Le Gonidec S, Moulédous L, Frances B,
Leclercq I, et al: Central apelin controls glucose homeostasis via
a nitric oxide-dependent pathway in mice. Antioxid Redox Signal.
15:1477–1496. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhu S, Sun F, Li W, Cao Y, Wang C, Wang Y,
Liang D, Zhang R, Zhang S, Wang H and Cao F: Apelin stimulates
glucose uptake through the PI3K/Akt pathway and improves insulin
resistance in 3T3-L1 adipocytes. Mol Cell Biochem. 353:305–313.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Li Y, Hu Q and Wang B: Effects of Apelin
on the fibrosis of retinal tissues and Müller cells in diabetes
retinopathy through the JAK2/STAT3 signalling pathway.
Autoimmunity. 56:22591292023. View Article : Google Scholar
|
|
62
|
Qin D, Zheng XX and Jiang YR: Apelin-13
induces proliferation, migration, and collagen I mRNA expression in
human RPE cells via PI3K/Akt and MEK/Erk signaling pathways. Mol
Vis. 19:2227–2236. 2013.PubMed/NCBI
|
|
63
|
Guo L, Li Q, Wang W, Yu P, Pan H, Li P,
Sun Y and Zhang J: Apelin inhibits insulin secretion in pancreatic
beta-cells by activation of PI3-kinase-phosphodiesterase 3B. Endocr
Res. 34:142–154. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Li B, Yin J, Chang J, Zhang J, Wang Y,
Huang H, Wang W and Zeng X: Apelin/APJ relieve diabetic
cardiomyopathy by reducing microvascular dysfunction. J Endocrinol.
249:1–18. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Sörhede Winzell M, Magnusson C and Ahrén
B: The apj receptor is expressed in pancreatic islets and its
ligand, apelin, inhibits insulin secretion in mice. Regul Pept.
131:12–17. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Ringström C, Nitert MD, Bennet H, Fex M,
Valet P, Rehfeld JF, Friis-Hansen L and Wierup N: Apelin is a novel
islet peptide. Regul Pept. 162:44–51. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Yue P, Jin H, Aillaud M, Deng AC, Azuma J,
Asagami T, Kundu RK, Reaven GM, Quertermous T and Tsao PS: Apelin
is necessary for the maintenance of insulin sensitivity. Am J
Physiol Endocrinol Metab. 298:E59–E67. 2010. View Article : Google Scholar
|
|
68
|
Chen H, Zheng C, Zhang X, Li J, Li J,
Zheng L and Huang K: Apelin alleviates diabetes-associated
endoplasmic reticulum stress in the pancreas of Akita mice.
Peptides. 32:1634–1639. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Han S, Englander EW, Gomez GA, Rastellini
C, Quertermous T, Kundu RK and Greeley GH Jr: Pancreatic Islet APJ
deletion reduces Islet density and glucose tolerance in mice.
Endocrinology. 156:2451–2460. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Guo M, Chen F, Lin T, Peng Y, Li W, Zhu X,
Lin L and Chen Y: Apelin-13 decreases lipid storage in hypertrophic
adipocytes in vitro through the upregulation of AQP7 expression by
the PI3K signaling pathway. Med Sci Monit. 20:1345–1352. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Attané C, Daviaud D, Dray C, Dusaulcy R,
Masseboeuf M, Prévot D, Carpéné C, Castan-Laurell I and Valet P:
Apelin stimulates glucose uptake but not lipolysis in human adipose
tissue ex vivo. J Mol Endocrinol. 46:21–28. 2011. View Article : Google Scholar
|
|
72
|
Attané C, Foussal C, Le Gonidec S, Benani
A, Daviaud D, Wanecq E, Guzmán-Ruiz R, Dray C, Bezaire V, Rancoule
C, et al: Apelin treatment increases complete Fatty Acid oxidation,
mitochondrial oxidative capacity, and biogenesis in muscle of
insulin-resistant mice. Diabetes. 61:310–320. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Dray C, Debard C, Jager J, Disse E,
Daviaud D, Martin P, Attané C, Wanecq E, Guigné C, Bost F, et al:
Apelin and APJ regulation in adipose tissue and skeletal muscle of
type 2 diabetic mice and humans. Am J Physiol Endocrinol Metab.
298:E1161–E1169. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Habchi M, Duvillard L, Cottet V, Brindisi
MC, Bouillet B, Beacco M, Crevisy E, Buffier P, Baillot-Rudoni S,
Verges B and Petit JM: Circulating apelin is increased in patients
with type 1 or type 2 diabetes and is associated with better
glycaemic control. Clin Endocrinol (Oxf). 81:696–701. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Gourdy P, Cazals L, Thalamas C, Sommet A,
Calvas F, Galitzky M, Vinel C, Dray C, Hanaire H, Castan-Laurell I
and Valet P: Apelin administration improves insulin sensitivity in
overweight men during hyperinsulinaemic-euglycaemic clamp. Diabetes
Obes Metab. 20:157–164. 2018. View Article : Google Scholar
|
|
76
|
Drougard A, Fournel A, Marlin A, Meunier
E, Abot A, Bautzova T, Duparc T, Louche K, Batut A, Lucas A, et al:
Central chronic apelin infusion decreases energy expenditure and
thermogenesis in mice. Sci Rep. 6:318492016. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Zhang H, Gong D, Ni L, Shi L, Xu W, Shi M,
Chen J, Ai Y and Zhang X: Serum elabela/toddler levels are
associated with albuminuria in patients with type 2 diabetes. Cell
Physiol Biochem. 48:1347–1354. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Karagoz ZK and Aydin S, Ugur K, Tigli A,
Deniz R, Baykus Y, Sahin I, Yalcin MH, Yavuz A, Aksoy A and Aydin
S: Molecular communication between apelin-13, apelin-36, elabela,
and nitric oxide in gestational diabetes mellitus. Eur Rev Med
Pharmacol Sci. 26:3289–3300. 2022.PubMed/NCBI
|
|
79
|
Onalan E, Doğan Y, Onalan E, Gozel N,
Buran I and Donder E: Elabela levels in patients with type 2
diabetes: Can it be a marker for diabetic nephropathy? Afr Health
Sci. 20:833–840. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Hong Y, Li J, Zhong Y, Yang S, Pei L,
Huang Z, Chen X, Wu H, Zheng G, Zeng C, et al: Elabela inhibits
TRAF1/NF-κB induced oxidative DNA damage to promote diabetic foot
ulcer wound healing. iScience. 26:1076012023. View Article : Google Scholar
|
|
81
|
Qi RQ, Chen YF, Cheng J, Song JW, Chen YH,
Wang SY, Liu Y, Yan KX, Liu XY, Li J and Zhong JC: Elabela
alleviates cuproptosis and vascular calcification in
vitaminD3-overloaded mice via regulation of the PPAR-γ/FDX1
signaling. Mol Med. 30:2232024. View Article : Google Scholar
|
|
82
|
Tang L, Yi X, Tan W, Yang H, Song S, Xiong
J, Liu C, Zhang Y, Wang M, Zhu M, et al: ELABELA ameliorates
atherosclerosis through restoring the M1/M2 macrophage balance in
ApoE−/− mice. J Am Heart Assoc. 14:e0412612025.
View Article : Google Scholar
|
|
83
|
Zhang Y, Wang Y, Luo M, Xu F, Lu Y, Zhou
X, Cui W and Miao L: Elabela protects against podocyte injury in
mice with streptozocin-induced diabetes by associating with the
PI3K/Akt/mTOR pathway. Peptides. 114:29–37. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Rayego-Mateos S, Rodrigues-Diez RR,
Fernandez-Fernandez B, Mora-Fernández C, Marchant V, Donate-Correa
J, Navarro-González JF, Ortiz A and Ruiz-Ortega M: Targeting
inflammation to treat diabetic kidney disease: The road to 2030.
Kidney Int. 103:282–296. 2023. View Article : Google Scholar
|
|
85
|
Cleveland KH and Schnellmann RG:
Pharmacological targeting of mitochondria in diabetic kidney
disease. Pharmacol Rev. 75:250–262. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Nyimanu D, Chapman FA, Gallacher PJ, Kuc
RE, Williams TL, Newby DE, Maguire JJ, Davenport AP and Dhaun N:
Apelin is expressed throughout the human kidney, is elevated in
chronic kidney disease & associates independently with decline
in kidney function. Br J Clin Pharmacol. 88:5295–5306. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Ozdemir N, Toraman A, Taneli F, Yurekli BS
and Hekimsoy Z: An evaluation of both serum Klotho/FGF-23 and
apelin-13 for detection of diabetic nephropathy. Hormones (Athens).
22:413–423. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Day RT, Cavaglieri RC and Feliers D:
Apelin retards the progression of diabetic nephropathy. Am J
Physiol Renal Physiol. 304:F788–F800. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Chen H, Li J, Jiao L, Petersen RB, Li J,
Peng A, Zheng L and Huang K: Apelin inhibits the development of
diabetic nephropathy by regulating histone acetylation in Akita
mouse. J Physiol. 592:505–521. 2014. View Article : Google Scholar
|
|
90
|
Yin J, Wang Y, Chang J, Li B, Zhang J, Liu
Y, Lai S, Jiang Y, Li H and Zeng X: Apelin inhibited
epithelial-mesenchymal transition of podocytes in diabetic mice
through downregulating immunoproteasome subunits β5i. Cell Death
Dis. 9:10312018. View Article : Google Scholar
|
|
91
|
Huang M, Chang J, Liu Y, Yin J and Zeng X:
Apelin/APJ alleviates diabetic nephropathy by improving glomerular
endothelial cells dysfunction via SIRT3-KLF15. Mol Med Rep.
31:1222025. View Article : Google Scholar
|
|
92
|
Fan C, Liu Y, Chang J, Huang M, Gao Z,
Wang H, Zheng X, Li M, Xu J, Yin J and Zeng X: Apelin alleviated
endothelial-to-mesenchymal transition via increasing BKCa in
diabetic nephropathy. FASEB J. 39:e707472025. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Huang M, Chang J, Liu Y, Yin J and Zeng X:
Apelin/APJ increased renal blood flow through endothelial BKCa
channel induced p-eNOS and ET-1 in diabetic conditions. Peptides.
183:1713332025. View Article : Google Scholar
|
|
94
|
Karimi Z, Daryabor G and Masjedi F:
Effects of conditioned media derived from human Wharton's jelly
mesenchymal stem cells on diabetic nephropathy and hepatopathy via
modulating TGF-β and apelin signaling pathways in male rats. BMC
Endocr Disord. 24:62024. View Article : Google Scholar
|
|
95
|
Hossein A, Firouzeh G, Zeinab K and
Gholamreza D: Quercetin prevents kidney against diabetes mellitus
(type 1) in rats by inhibiting TGF-β/apelin gene expression. Mol
Biol Rep. 51:6772024. View Article : Google Scholar
|
|
96
|
Guo C, Liu Y, Zhao W, Wei S, Zhang X, Wang
W and Zeng X: Apelin promotes diabetic nephropathy by inducing
podocyte dysfunction via inhibiting proteasome activities. J Cell
Mol Med. 19:2273–2285. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zhang J, Yin J, Wang Y, Li B and Zeng X:
Apelin impairs myogenic response to induce diabetic nephropathy in
mice. FASEB J. 32:4315–4327. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Lu X, Liu S, Luan R, Cui W, Chen Y, Zhang
Y, Lu Y, Zhang H, Shi L, Miao L and Xu F: Serum elabela and apelin
levels during different stages of chronic kidney disease. Ren Fail.
42:667–672. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Xiong M, Chen H, Fan Y, Jin M, Yang D,
Chen Y, Zhang Y, Petersen RB, Su H, Peng A, et al: Tubular
Elabela-APJ axis attenuates ischemia-reperfusion induced acute
kidney injury and the following AKI-CKD transition by protecting
renal microcirculation. Theranostics. 13:3387–3401. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Yan Z, Shi Y, Yang R, Xue J and Fu C:
ELABELA-derived peptide ELA13 attenuates kidney fibrosis by
inhibiting the Smad and ERK signaling pathways. J Zhejiang Univ Sci
B. 25:341–353. 2024.In English, Chinese. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Xu F, Zhou H, Wu M, Zhang H, Zhang Y, Zhao
Q, Brown R, Gong DW and Miao L: Fc-Elabela fusion protein
attenuates lipopolysaccharide-induced kidney injury in mice. Biosci
Rep. 40:BSR201923972020. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Chen Z, Wu C, Liu Y, Li H, Zhu Y, Huang C,
Lin H, Qiao Q, Huang M, Zhu Q and Wang L: ELABELA attenuates
deoxycorticosterone acetate/salt-induced hypertension and renal
injury by inhibition of NADPH oxidase/ROS/NLRP3 inflammasome
pathway. Cell Death Dis. 11:6982020. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Chen Z, Wang Z, Hu Y, Lin H, Yin L, Kong
J, Zhang Y, Hu B, Li T, Zheng X, et al: ELABELA/APJ axis prevents
diabetic glomerular endothelial injury by regulating AMPK/NLRP3
pathway. Inflammation. 46:2343–2358. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Ng ACT, Delgado V, Borlaug BA and Bax JJ:
Diabesity: The combined burden of obesity and diabetes on heart
disease and the role of imaging. Nat Rev Cardiol. 18:291–304. 2021.
View Article : Google Scholar
|
|
105
|
Zeng H, He X, Hou X, Li L and Chen JX:
Apelin gene therapy increases myocardial vascular density and
ameliorates diabetic cardiomyopathy via upregulation of sirtuin 3.
Am J Physiol Heart Circ Physiol. 306:H585–H597. 2014. View Article : Google Scholar
|
|
106
|
Hou X, Zeng H, He X and Chen JX: Sirt3 is
essential for apelin-induced angiogenesis in post-myocardial
infarction of diabetes. J Cell Mol Med. 19:53–61. 2015. View Article : Google Scholar :
|
|
107
|
Hou X, Zeng H, Tuo QH, Liao DF and Chen
JX: Apelin gene therapy increases autophagy via activation of
sirtuin 3 in diabetic heart. Diabetes Res (Fairfax). 1:84–91.
2015.PubMed/NCBI
|
|
108
|
Zhong JC, Yu XY, Huang Y, Yung LM, Lau CW
and Lin SG: Apelin modulates aortic vascular tone via endothelial
nitric oxide synthase phosphorylation pathway in diabetic mice.
Cardiovasc Res. 74:388–395. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Li X, Yu L, Gao J, Bi X, Zhang J, Xu S,
Wang M, Chen M, Qiu F and Fu G: Apelin ameliorates high
glucose-induced downregulation of connexin 43 via AMPK-dependent
pathway in neonatal rat cardiomyocytes. Aging Dis. 9:66–76. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Leung OM, Li J, Li X, Li X, Chan VW, Yang
KY, Ku M, Ji L, Sun H, Waldmann H, et al: Regulatory T cells
promote apelin-mediated sprouting angiogenesis in type 2 diabetes.
Cell Rep. 24:1610–1626. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Studneva IM, Veselova OM, Dobrokhotov IV,
Serebryakova LI, Palkeeva ME, Avdeev DV, Molokoedov AS, Sidorova MV
and Pisarenko OI: The structural analogue of apelin-12 prevents
energy disorders in the heart in experimental type 1 diabetes
mellitus. Biomed Khim. 70:135–144. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Kartal H, Comu FM, Kucuk A, Polat Y,
Dursun AD and Arslan M: Effect of apelin-13 on erythrocyte
deformability during ischaemia-reperfusion injury of heart in
diabetic rats. Bratisl Lek Listy. 118:133–136. 2017.PubMed/NCBI
|
|
113
|
Gunes I, Kartal H, Dursun AD, Sungu N,
Polat YS, Erkent FD, Arslan M and Kucuk A: Effects of apelin-13 on
myocardial ischemia reperfusion injury in streptozotocine induced
diabetic rats. Bratisl. Lek Listy. 119:348–354. 2018.PubMed/NCBI
|
|
114
|
Koval S, Iushko K and Starchenko T:
Relations of apelin with cardiac remodeling in patients with
hypertension and type 2 diabetes. Folia Med (Plovdiv). 60:117–123.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Karakoç A, Sahin A, Polat ES, Aliyev E,
Yildirim A, Bakan N and Dokumacioglu E: Serum apelin and ADMA
levels in type 2 diabetics with and without vascular complications.
Diabetes Metab Syndr. 10(2 Suppl 1): S106–S109. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Rittig K, Hildebrandt U, Thamer C, Staiger
H, Peter A, Stefan N, Fritsche A, Häring HU, Balletshofer BM and
Siegel-Axel D: Apelin serum levels are not associated with early
atherosclerosis or fat distribution in young subjects with
increased risk for type 2 diabetes. Exp Clin Endocrinol Diabetes.
119:358–361. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Sabry RN, El Wakeel MA, El-Kassas GM, Amer
AF, El Batal WH, El-Zayat SR and Abou-El-Asrar M: Serum apelin: A
new marker of early atherosclerosis in children with type 1
diabetes mellitus. Open Access Maced J Med Sci. 6:613–617. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Wang Y, Zhang R, Shen H, Kong J and Lv X:
Pioglitazone protects blood vessels through inhibition of the
apelin signaling pathway by promoting KLF4 expression in rat models
of T2DM. Biosci Rep. 39:BSR201903172019. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Berezin AA, Fushtey IM and Berezin AE: The
Effect of SGLT2 inhibitor dapagliflozin on serum levels of apelin
in T2DM patients with heart failure. Biomedicines. 10:17512022.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Kadoglou NPE, Fotiadis G, Kapelouzou A,
Kostakis A, Liapis CD and Vrabas IS: The differential
anti-inflammatory effects of exercise modalities and their
association with early carotid atherosclerosis progression in
patients with type 2 diabetes. Diabet Med. 30:e41–e50. 2013.
View Article : Google Scholar
|
|
121
|
Berezin AA, Fushtey IM and Berezin AE:
Discriminative utility of apelin-to-NT-pro-brain natriuretic
peptide ratio for heart failure with preserved ejection fraction
among type 2 diabetes mellitus patients. J Cardiovasc Dev Dis.
9:232022.PubMed/NCBI
|
|
122
|
Berezin AA, Obradovic Z, Novikov EV,
Boxhammer E, Lichtenauer M and Berezin AE: Interplay between
myokine profile and glycemic control in type 2 diabetes mellitus
patients with heart failure. Diagnostics (Basel). 12:29402022.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Yu P, Ma S, Dai X and Cao F: Elabela
alleviates myocardial ischemia reperfusion-induced apoptosis,
fibrosis and mitochondrial dysfunction through PI3K/AKT signaling.
Am J Transl Res. 12:4467–4477. 2020.PubMed/NCBI
|
|
124
|
Ren W, Xi Y, Ma Y, Duan X, Wang T, Yu M,
Qin S, Gong DW and Tian Z: Aerobic exercise upregulates
renal-derived ELABELA to improve myocardial fibrosis by activating
APJ-AMPK-Sirt1 and inhibiting TGFβ1-Smad2/3 signaling pathways in
myocardial infarction mice. FASEB J. 39:e707342025. View Article : Google Scholar
|
|
125
|
Rakhshan K, Sharifi M, Ramezani F, Azizi Y
and Aboutaleb N: ERK/HIF-1α/VEGF pathway: A molecular target of
ELABELA (ELA) peptide for attenuating cardiac ischemia-reperfusion
injury in rats by promoting angiogenesis. Mol Biol Rep.
49:10509–10519. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Hendrianus, Adiarto S, Prakoso R, Firdaus
I, Indriani S, Rudiktyo E, Widyantoro B, Taofan, Ambari AM and
Sukmawan R: A novel peptide elabela is associated with
hypertension-related subclinical atherosclerosis. High Blood Press
Cardiovasc Prev. 30:37–44. 2023. View Article : Google Scholar
|
|
127
|
Yao F, Lv YC, Zhang M, Xie W, Tan YL, Gong
D, Cheng HP, Liu D, Li L, Liu XY, et al: Apelin-13 impedes foam
cell formation by activating class III PI3K/Beclin-1-mediated
autophagic pathway. Biochem Biophys Res Commun. 466:637–643. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Peng JY, Fu X, Luo XY, Liu F, Zhang B,
Zhou B, Sun K and Chen AF: Endothelial ELABELA improves
post-ischemic angiogenesis by upregulating VEGFR2 expression.
Transl Res. 270:13–23. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Li W, Xu P, Kong L, Feng S, Shen N, Huang
H, Wang W, Xu X, Wang X, Wang G, et al: Elabela-APJ axis mediates
angiogenesis via YAP/TAZ pathway in cerebral ischemia/reperfusion
injury. Transl Res. 257:78–92. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Ni T, Lin N, Huang X, Lu W, Sun Z, Zhang
J, Lin H, Chi J and Guo H: Icariin ameliorates diabetic
cardiomyopathy through apelin/Sirt3 signalling to improve
mitochondrial dysfunction. Front Pharmacol. 11:2562020. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Seo H, Park SJ and Song M: Diabetic
retinopathy (DR): Mechanisms, current therapies, and emerging
strategies. Cells. 14:3762025. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Little K, Llorián-Salvador M, Scullion S,
Hernández C, Simó-Servat O, Del Marco A, Bosma E, Vargas-Soria M,
Carranza-Naval MJ, Van Bergen T, et al: Common pathways in dementia
and diabetic retinopathy: Understanding the mechanisms of
diabetes-related cognitive decline. Trends Endocrinol Metab.
33:50–71. 2022. View Article : Google Scholar
|
|
133
|
Lessieur EM, Liu H, Saadane A, Du Y, Kiser
J and Kern TS: ICAM-1 on the luminal surface of endothelial cells
is induced to a greater extent in mouse retina than in other
tissues in diabetes. Diabetologia. 65:1734–1744. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Church KA, Rodriguez D, Vanegas D,
Gutierrez IL, Cardona SM, Madrigal JLM, Kaur T and Cardona AE:
Models of microglia depletion and replenishment elicit protective
effects to alleviate vascular and neuronal damage in the diabetic
murine retina. J Neuroinflammation. 19:3002022. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Sivaprasad S, Wong TY, Gardner TW, Sun JK
and Bressler NM: Diabetic retinal disease. Nat Rev Dis Primers.
11:622025. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Du JH, Li X, Li R, Xu L, Ma RR, Liu SF,
Zhang Z and Sun HZ: Elevation of serum apelin-13 associated with
proliferative diabetic retinopathy in type 2 diabetic patients. Int
J Ophthalmol. 7:968–973. 2014.PubMed/NCBI
|
|
137
|
Tao Y, Lu Q, Jiang YR, Qian J, Wang JY,
Gao L and Jonas JB: Apelin in plasma and vitreous and in
fibrovascular retinal membranes of patients with proliferative
diabetic retinopathy. Invest Ophthalmol Vis Sci. 51:4237–4242.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Lu Q, Ma Y, Xu YS and Jiang YR: Apelin in
epiretinal membranes of patients with proliferative diabetic
retinopathy. Mol Vis. 20:1122–1131. 2014.PubMed/NCBI
|
|
139
|
Lu Q, Feng J and Jiang YR: The role of
apelin in the retina of diabetic rats. PLoS One. 8:e697032013.
View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Feng J, Yang W, Luan F, Ma F, Wang Y,
Zhang Y, Liu X, Chen L, Hu X and Tao Y: The protective role of
apelin in the early stages of diabetic retinopathy. Int J Mol Sci.
23:146802022. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Dai L, Luo L, Zhang Y, Fu M and Yu L:
Protective effect of apelin-13 on oxygen and glucose deprivation
induced-damage in retinal ganglion cells cultured in vitro. J Mol
Histol. 56:252024. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Seyithanoğlu M, Meşen S, Comez A, Meşen A,
Beyoğlu A, Baykişi Y and Alkan Baylan F: The potential of serum
elabela levels as a marker of diabetic retinopathy: Results from a
pilot cross-sectional study. PeerJ. 13:e188412025. View Article : Google Scholar
|
|
143
|
Wang J, Zhang Q, Chen E, Zhao P and Xu Y:
Elabela promotes the retinal angiogenesis by inhibiting ferroptosis
during the vaso-obliteration phase in mouse oxygen-induced
retinopathy model. FASEB J. 36:e222572022. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Hosny SS, Nasr MS, Ibrahim RH and
YousryHelal Z: Relation between plasma apelin level and peripheral
neuropathy in type 2 diabetic patients. Diabetes Metab Syndr.
13:626–629. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Cai X, Hu S, Liu W, Yin Y, Jiang Y, Wang
Y, Lu B, Wang Y, Wang D and Chen J: Apelin receptor
homodimerisation inhibits hippocampal neuronal autophagy via G
protein-dependent signalling in vascular dementia. Mol Neurobiol.
62:1826–1839. 2025. View Article : Google Scholar
|
|
146
|
Robillard S, Trân K, Lachance MS, Brazeau
T, Boisvert E, Lizotte F, Auger-Messier M, Boudreault PL, Marsault
É and Geraldes P: Apelin prevents diabetes-induced poor collateral
vessel formation and blood flow reperfusion in ischemic limb. Front
Cardiovasc Med. 10:11918912023. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Kılınç S, Ölçüoğlu R, Arzu Yiğit A,
Güneşer Ö and Eylül Aydemir B: Effects of apelin-13 on auditory
system in STZ-induced diabetic rats. Neurosci Lett. 842:1379962024.
View Article : Google Scholar
|
|
148
|
O'Harte FPM, Parthsarathy V, Hogg C and
Flatt PR: Acylated apelin-13 amide analogues exhibit enzyme
resistance and prolonged insulin releasing, glucose lowering and
anorexic properties. Biochem Pharmacol. 146:165–173. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
O'Harte FPM, Parthsarathy V, Hogg C and
Flatt PR: Apelin-13 analogues show potent in vitro and in vivo
insulinotropic and glucose lowering actions. Peptides. 100:219–228.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
O'Harte FPM, Parthsarathy V, Hogg C and
Flatt PR: Long-term treatment with acylated analogues of apelin-13
amide ameliorates diabetes and improves lipid profile of high-fat
fed mice. PLoS One. 13:e02023502018. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Gao LR, Zhang NK, Zhang Y, Chen Y, Wang L,
Zhu Y and Tang HH: Overexpression of apelin in Wharton' jelly
mesenchymal stem cell reverses insulin resistance and promotes
pancreatic β cell proliferation in type 2 diabetic rats. Stem Cell
Res Ther. 9:3392018. View Article : Google Scholar
|


















