Diabetes mellitus (DM) is the most common chronic
metabolic disease worldwide, with its global incidence and
prevalence rising continuously over the past 30 years: The number
of adults living with diabetes has quadrupled from 198 million in
1990 to 828 million in 2022, and the age-standardized prevalence
has doubled from 7.0 to 14.0%, with sustained growth observed in
the vast majority of countries worldwide (1,2).
Clinically, complications of DM mainly include coronary heart
disease, peripheral artery disease, retinopathy, neuropathy and
nephropathy (3). In recent
years, the incidence of diabetic nephropathy (DN), a serious kidney
disease, has continued to rise, 20 to 40% of patients with diabetes
may develop kidney disease (4).
Long-term hyperglycemia can alter the physiological
microenvironment, triggering reactions such as oxidative stress
(OS), pyroptosis, immune responses, mitochondrial dysfunction and
lysosomal damage. These reactions can cause a series of abnormal
adaptations, including glomerular hypertrophy, podocyte loss and
mesangial matrix dilation. In the development of DN, activation of
the NOD-like Receptor family Pyrin domain containing 3 (NLRP3)
inflammasomes is a key contributory factor in a variety of renal
injury mechanisms (5,6).
The NLRP3 inflammasome is an essential intracellular
multiprotein complex in innate immunity, predominantly expressed in
the cytoplasm of innate immune cells such as macrophages, dendritic
cells and mast cells, and is currently the most well-established
inflammasome complex (7). The
NLRP3 inflammasome recognizes multiple pathogen molecular patterns
and endogenous danger signals, recruits and activates caspase-1 via
the apoptosis-associated speck-like protein (ASC) aptamer, induces
cleavage of downstream Gasdermin D (GSDMD) and regulates the
release of IL-1β and IL-18, ultimately leading to pyroptosis
(8). The NLRP3 inflammasome can
be activated not only by various pathogenic and environmental
stimuli, such as microbial cell wall components, nucleic acids and
alum and silica, but also by endogenous hazard signals, such as
lipopolysaccharides, adenosine triphosphate, hyaluronic acid, islet
amyloid polypeptide, heme, oxidized mitochondrial DNA and the
membrane attack complex (MAC) (9,10). In autoimmune diseases,
arteriosclerosis and diabetic complications, abnormal activation of
the NLRP3 inflammasome can trigger excessive inflammatory
responses, acts as one of the key inflammatory mediators, rather
than a sole dominant driver of tissue damage (11-13).
In the progression of DN, alterations in the renal
microenvironment lead to excessive activation of NLRP3
inflammasomes, which trigger apoptosis and pyroptosis in glomerular
mesangial cells, endothelial cells and tubular epithelial cells,
ultimately resulting in chronic inflammation and renal fibrosis
(14,15). Targeted inhibition of
NLRP3-mediated pyroptosis is considered to be a potential
therapeutic approach (16);
NLRP3 knockout in diabetic mice was shown to alleviate
streptozotocin-induced glomerular hypertrophy, glomerular sclerosis
and mesangial matrix dilation (17). The present review introduces the
structure and function of the NLRP3 inflammasome and examines the
various pathways involved in its activation. The potential roles
and mechanisms of the NLRP3 inflammasome in DN are discussed and
finally, current therapeutic strategies targeting the NLRP3
inflammasome for the treatment of DN are summarized.
DN is a specific form of kidney injury and the
leading cause of end-stage kidney disease (18). The chronic renal failure
resulting from DN is also a major cause of mortality among patients
with DM (19,20). DN involves both structural
changes in the kidneys and changes in kidney function (21). Structurally, DN includes
glomerular mesangial dilation, thickening of the basement membrane,
podocyte reduction, nodular glomerulosclerosis and endothelial cell
destruction (22,23). Functionally, DN is characterized
by increased albumin excretion and impaired glomerular filtration
(24). In addition, a key
pathogenic factor for the development of DN is persistent
hyperglycemia and it has been shown to induce marked renal
structural damage through advanced glycation end-product (AGE)
accumulation, activation of the polyol and hexosamine pathways,
stimulation of the renin-angiotensin-aldosterone and sympathetic
nervous systems, and the onset of insulin resistance and
endothelial dysfunction (25,26). Studies have shown that factors
such as OS, immune response, mitochondrial damage, pyroptosis and
podocyte autophagy can all cause damage to these cells (27,28), which disrupts the filtration
barrier and leads to elevated proteinuria, abnormal glomerular
filtration rate and elevated creatinine levels (29,30), thereby promoting the occurrence
and development of DN (Fig. 1).
During the development the DN, all of these factors trigger an
inflammatory response, the core of which is the inflammasome. This
cytoplasmic multiprotein complex activates the inflammatory protein
caspase-1 and serves a key role in the innate immune system
(31). The NLRP3 inflammasome,
as a key participant, is widely involved in the inflammatory
response by forming and releasing inflammatory cytokines, thereby
exacerbating the development of DN (32,33).
Inflammasomes are large multiprotein complexes
belonging to the pattern recognition receptor (PRR) family and
serve a key role in the innate immune system (34). They are composed of inflammatory
caspases and various sensors, including the nucleotide-binding
domain and leucine-rich repeat-containing receptors (NLRs) family,
pyrin and absent in melanoma 2 (AIM2) (35,36). Members of the NLR family
typically contain three conserved structural domains: A C-terminal
leucine-rich repeat (LRR) region, a central nucleotide-binding site
and an NACHT domain responsible for oligomerization and an
N-terminal effector domain (37,38). The NLR comprises several members,
including NLRP1, NLRP3, NLRP6 and NLRC4, among which the NLRP3
inflammasome is the most well-characterized (39,40).
The NLRP3 inflammasome is composed of NLRP3, a
multiprotein complex consisting of ASC and caspase-1 (41). This complex serves a key role in
the immune system, recognizing pathogen-associated molecular
patterns (PAMPs) and damage-associated molecular patterns (DAMPs)
to initiate immune responses (42). Upon activation, the NLRP3
inflammasome recruits ASC to form speck-like aggregates, which
facilitate the conversion of pro-caspase-1 into its active p10/p20
subunits (43). Activated
caspase-1 subsequently processes the inactive precursors pro-IL-1
and pro-IL-18 into their mature, bioactive forms, thereby promoting
the secretion of these key pro-inflammatory cytokines. Furthermore,
caspase-1 cleaves the pyroptosis executioner protein GSDMD into its
N- and C-terminal fragments, leading to membrane pore formation,
cellular lysis and pyroptotic cell death (44,45).
NLRP3 functions as a pivotal sensor in sterile
inflammatory signaling and acts as a central regulator of chronic
inflammatory disorders. Under homeostatic conditions, the NLRP3
inflammasome exerts beneficial effects by facilitating pathogen
clearance, promoting tissue repair and preserving physiological
balance. By contrast, its aberrant or sustained activation drives
chronic inflammation and tissue damage (Table I) (59-87).
Pyroptosis of inflammatory and renal intrinsic cells
mediated by the NLRP3 inflammasome is a key contributory cell death
mode in renal inflammation and injury during DN. The canonical
NLRP3 inflammasome-mediated pyroptosis pathway is the predominant
pathway in DN, with considerable in vivo evidence, while the
non-canonical (caspases-4/5/11-mediated) and
caspase-3/GSDME-mediated pathways have presently been only
validated in vitro and lack direct in vivo
verification in DN models. The pyroptosis pathway is characterized
by the formation of pores in the cell membrane, leading to cell
rupture and the release of pro-inflammatory factors, exacerbating
renal inflammation and fibrosis (88). This process is initiated by
stimuli including hyperglycemia, OS, AGEs and lipotoxicity, in
which activation of the NLRP3 inflammasome represents a pivotal
event (89). Following NLRP3
inflammasome activation, Caspase-1 cleaves GSDMD and
pro-IL-1β/IL-18, leading to the release of inflammatory mediators
and the induction of pyroptosis (90).
In the atypical inflammasome pathway, caspases-4, -5
and -11 directly detect pathogen molecules via their caspase
recruitment domains (CARDs), leading to caspase-1 activation, IL-1
and IL-18 maturation, GSDMD cleavage and IL-1 release (95,96). GSDMB does not induce pyroptosis
via its N-terminal domain, as other Gasdermin family members do,
but instead promotes caspase-4 activity by directly binding to the
caspase-4 CARD domain (97).
Subsequently, activated caspase-4 can form membrane pores via GSDMD
activity and potassium influx can activate NLRP3. Another study
demonstrated that the downregulation of caspase-4 inhibits
TNF-α-induced pyroptosis in human pulmonary artery endothelial
cells, as well as the activation of GSDMD and GSDME (98). Furthermore, caspase-11 recognizes
its substrates, predominantly GSDMD, via the P1'-P4' region of the
target protein (99,100).
Finally, the caspase-3-mediated inflammasome pathway
is also associated with pyroptosis as caspase-3 is activated during
LPS-induced pyroptosis (101).
GSDME can also be cleaved by caspase-3, leading to a transition
from apoptosis to pyroptosis (102). When NLRP3-specific inhibitors
were used to suppress NLRP3-mediated pyroptosis, ATP induced
pyroptosis in macrophages via the caspase-3/GSDME axis (103). Meanwhile, activated caspase-3
also inactivates the pore-forming domain of GSDMD, which inhibits
GSDMD-mediated pyroptosis (104). Notably, both GSDMD-NT
(N-terminal) and GSDME-NT can act on mitochondria, leading to
increased ROS production, inducing apoptosis, further stimulating
the release of inflammatory substances and exacerbating tissue
damage (105,106). Notably, the non-canonical
inflammasome pathway and caspase-3/GSDME-mediated pyroptosis
pathway have not been validated using in vivo DN models,
whereas the canonical NLRP3/caspase-1/GSDMD pathway is fully
supported by multiple genetic and pharmacological in vivo
studies (88,90,107).
The progression of DN is associated with pyroptosis,
which is regulated by five interacting hierarchical core signaling
pathways (NF-κB/NLRP3, TXNIP/NLRP3, Nrf2/HO-1/NLRP3, HIF-1α/NLRP3
and PTEN/PI3K/Akt) (TXNIP, thioredoxin-interacting protein; Nrf2,
nuclear factor erythroid 2-related Factor 2; HO-1, heme
oxygenase-1; HIF-1α, hypoxia-inducible factor-1α) rather than
independent mechanisms (Fig. 4).
These pathways form an upstream-downstream regulatory hierarchy
with extensive crosstalk and are divided into three modules: NF-κB
acts as the top upstream module, priming NLRP3 inflammasome by
transcriptionally upregulating NLRP3, pro-IL-1, pro-IL-18, and
activating TXNIP/HIF-1α; the midstream module (TXNIP/NLRP3,
Nrf2/HO-1/NLRP3, HIF-1/NLRP3) mediates NLRP3 activation (TXNIP
binds NLRP3 via ROS, Nrf2/HO-1 negatively regulates ROS/TXNIP/NF-κB
but is impaired in DN and HIF-1α forms a positive feedback loop);
the PTEN/PI3K/Akt pathway serves as the final facilitator, as
hyperglycemia inactivates PTEN to hyperactivate Akt, amplifying the
cascade and promoting NLRP3-ASC-caspase-1 assembly (108-110). Collectively, these pathways
converge to three core nodes (NF-κB-mediated priming,
ROS-TXNIP-induced NLRP3 oligomerization and Akt-mediated execution)
to drive aberrant NLRP3 activation and pyroptosis in DN, indicating
multi-pathway synergistic targeting as a rational therapeutic
strategy.
OS refers to a pathological state in which the
generation rate of ROS and reactive nitrogen species (RNS) in the
body exceeds the clearance capacity of the endogenous antioxidant
system, resulting in an imbalance in oxidative and antioxidant
homeostasis (111). OS in DN is
predominantly caused by the overproduction of ROS and RNS, induced
by factors such as hyperglycemia and AGEs (112). A previous study has shown that
hyperglycemia can induce OS and podocyte damage by mediating the
Von Hippel-Lindau E3 ubiquitin ligase, which promotes the
ubiquitin-mediated degradation of glucose-6-phosphate dehydrogenase
(113). Advanced oxidation
protein products (AOPPs) are oxidation-modified products formed
after plasma proteins are attacked by ROS (114); AOPPs can synergize with AGEs to
activate NADPH oxidase (NOX), leading to excessive ROS production
and NF-κB activation, thereby triggering the Wnt/β-catenin
signaling pathway and ultimately mediating podocyte
dedifferentiation and epithelial-mesenchymal transition (EMT)
(115). This oxidative
imbalance mediates kidney injury through multiple pathways,
including glomerular endothelial cell damage, mesangial cell
proliferation, increased extracellular matrix (ECM) deposition,
induction of podocyte apoptosis, suppression of podocyte protein
expression, EMT-related pathological changes, inflammatory
factor-driven injury, renal vascular endothelial cell impairment
and mitochondrial dysfunction (116).
In the pathological progression of DN, the NLRP3
inflammasome serves a pivotal role by driving the
OS-inflammation-cell injury cascade. Under hyperglycemic
conditions, aberrant NOX activation and dysfunction of the
mitochondrial electron transport chain in renal tissues lead to
excessive ROS production, which acts as a key initiating signal for
NLRP3 inflammasome activation (117-119).
ROS can induce NLRP3 oligomerization through
multiple mechanisms. First, ROS directly oxidize TXNIP, promoting
its binding to NLRP3 and thereby relieving Trx-mediated inhibition
of NLRP3 (120). Second, ROS
disrupt lysosomal membrane stability, leading to the release of
cathepsin B and subsequent proteolytic signaling (121). Third, ROS trigger the release
of mitochondrial (mt) DNA and the accumulation of mtROS, which
activate innate immune responses through DAMPs (122). After NLRP3 activation, ASC is
recruited and caspase-1 is cleaved, promoting the conversion of
IL-1β and IL-18 precursors into mature inflammatory factors
(123). IL-1β activates the
NF-κB pathway in renal interstitial fibroblasts, induces expression
of monocyte chemoattractant protein-1 (MCP-1) and intercellular
adhesion molecule-1 (ICAM-1), which exacerbates macrophage
infiltration and renal interstitial fibrosis. IL-18, together with
IFN-γ, promotes M1-type polarization in renal tubular epithelial
cells and amplifies the local inflammatory response (124-126). Meanwhile, caspase-1-mediated
cleavage of GSDMD forms pore-forming proteins, causing pyroptosis
in podocytes and renal tubular epithelial cells, disrupting the
glomerular filtration barrier and inducing tubulointerstitial
injury (127,128). Notably, inflammatory factors
form a positive feedback loop with OS signaling. IL-1β and TNF-α
enhance ROS production by upregulating NOX subunits, while ROS
further amplify the inflammatory cascade by activating NLRP3. This
cycle ultimately contributes to mesangial matrix expansion,
basement membrane thickening and tubular atrophy, thereby
accelerating the progression of DN (129,130). OS-driven NLRP3 inflammasome
abnormal activation not only serves as the core hub of DN
inflammatory damage, but also acts as a key molecular node
connecting glycolipid metabolism disorder and renal tissue fibrosis
(Fig. 5).
Mitochondria, as the prominent energy-metabolism
organelles within cells, play an indispensable role in aerobic
respiration (131). Podocytes
are rich in mitochondria, which are decisive for the normal
physiological function and energy supply of podocytes (132). In the pathological process of
DN, mitochondrial dysfunction is considered a key mechanism of
podocyte damage, involving abnormalities in oxidative
phosphorylation (OXPHOS), dysregulation of mitochondrial dynamics,
defects in biosynthesis, dysregulation of autophagy and
interactions among multiple signaling pathways (133-135). Hyperglycemia promotes the
formation of AGEs from glucose, which then bind to RAGE on the
surface of renal cells, generating a large amount of ROS via NOX.
ROS can directly attack mitochondria, disrupt mitochondrial
membrane structure, activate MAPK pathway, phosphorylate the
subunit of the mitochondrial respiratory chain complex and inhibit
OXPHOS efficiency. Furthermore, this downregulates the expression
of mitochondrial antioxidant enzymes, weakens the clearance ability
of mtROS and intensifies OS (136,137). The novel molecular mechanism by
which mitochondrial damage activates the NLRP3 inflammasome mainly
involves the regulatory role of mitochondrial metabolites and the
participation and synergy of the cGAS-STING pathway (138). It first manifests as the dual
effects of succinic acid; under hyperglycemic conditions, the
mitochondrial tricarboxylic acid cycle becomes impaired, leading to
succinic acid accumulation. Whilst excess succinic acid upregulates
NLRP3 transcription by stabilizing HIF-1α, it also inhibits
mitochondrial succinate dehydrogenase, which promotes electron
leakage from the electron transport chain and enhances mtROS
production. Together, this creates a positive feedback loop
(139). A further mechanism of
action is the mitochondrial targeting effect of ceramides, with
ceramide synthase 6 catalyzing mitochondrial ceramides, which leads
to mtDNA release and NLRP3 activation (140). In addition,
hyperglycemia-induced enhanced glycolysis leads to intracellular
lactic acid accumulation. Lactic acid directly binds to the NLRP3
promoter region by modifying histone H3 lysine 18 to promote its
transcriptional activation (141). Regarding the cGAS-STING
pathway, hyperglycemia-induced mitochondrial damage leads to mtDNA
leakage into the cytoplasm; cGAS recognizes mtDNA to generate
cGAMP, which activates STING and recruits TBK1, which
phosphorylates IRF3 and NF-κB to promote the expression of
pro-inflammatory factors such as Interferon-beta (IFN-β), IL-6 and
TNF-α. At the same time, the cGAS-STING pathway can act
synergistically with NLRP3 by upregulating NLRP3 transcription and
enhancing mtROS production, thereby facilitating inflammasome
assembly and further exacerbating the progression of DN (Fig. 6) (142,143).
The immune-mediated damage of DN centers on the
overactivation of innate immune pathways, which work in synergy to
drive the inflammatory cascade and fibrosis in the kidneys
(144). The initial triggers of
injury include hyperglycemia, glycotoxicity, AGEs and OS, all of
which cause cellular damage and lead to the release of DAMPs and
mtDNA. These danger signals subsequently activate TLRs (such as
TLR2 or TLR4) and the NLRP3 inflammasome (145,146). TLR signaling induces the
expression of pro-inflammatory cytokines (such as IL-6 and TNF-α)
and ICAM-1 through the NF-κB pathway, which promotes the
infiltration of macrophages and T cells. Meanwhile, the NLRP3
inflammasome mediates the maturation and release of IL-1β and IL-18
via caspase-1, amplifying the inflammatory response and triggering
podocyte apoptosis (147,148). In terms of immune cell effects,
macrophages recruit and release fibrotic factors through CCL2-CCR2
signaling, and T cells enhance the inflammatory response through
IFN-γ, both of which form a pro-inflammatory and pro-fibrotic
positive feedback loop with innate cells (149,150). Ultimately, these immune
mechanisms lead to the thickening of the glomerular basement
membrane, mesangial dilation, interstitial fibrosis and loss of
podocytes, resulting in pathological features such as proteinuria
and a gradual decline in renal function, which in turn cause
glomerular sclerosis and tubulointerstitial fibrosis driven by
chronic inflammation, eventually developing into end-stage renal
disease (151,152).
In DN, the activation of NLRP3 inflammasomes drives
renal immunopathological damage through multiple mechanisms. The
core effect is the amplification of the inflammatory cascade. Upon
activation of the NLRP3/caspase-1 pathway, mature IL-1β and IL-18
are released through cleavage of their precursors, triggering
infiltration of immune cells such as macrophages and T cells into
the renal interstitium and inducing the secretion of chemokines
such as MCP-1 and IL-6, thereby forming a positive feedback
inflammatory loop. Additionally, IL-1β activates the MAC via the
classical complement pathway, directly damaging the glomerular
basement membrane (153-155).
At the level of intrinsic kidney cell damage, NLRP3 activation
results in the loss of podocyte slit diaphragm proteins and fusion
of podocyte foot processes, leading to proteinuria. NLRP3
activation also induces pyroptosis of renal tubular epithelial
cells through the CD36-mtROS-NLRP3 axis, releasing pro-inflammatory
contents. Simultaneously, NLRP3 activation promotes mesangial cell
proliferation and ECM secretion, accelerating glomerular sclerosis
(156-158). During fibrosis, IL-1β and IL-18
promote fibroblast activation and collagen deposition by
upregulating TGF-β, inducing EMT and causing mitochondrial
dysfunction through sustained mtROS production, which leads to
renal interstitial fibrosis and an irreversible decline in renal
function (159-161). The multifaceted effects of this
pathological network indicate that the NLRP3 inflammasome serves as
a central regulatory node for both immune-mediated injury and
fibrotic progression in DN (Fig.
7).
Autophagy is a highly conserved process in
eukaryotic cells, in which autophagosomes enclose damaged
organelles, misfolded proteins or pathogens and fuse with lysosomes
to degrade their contents (162). It is mainly classified into
three types based on substrate handling and function:
Macroautophagy, microautophagy and chaperone-mediated autophagy.
Podocyte autophagy is the process by which glomerular podocytes
selectively or non-selectively degrade damaged intracellular
components through pathways primarily mediated by macroautophagy,
and is supplemented by chaperone-mediated autophagy. This process
regulates cellular metabolism and stress responses, ultimately
preserving podocyte survival, structural integrity and filtration
function (163).
In DN, hyperglycemia directly inhibits podocyte
autophagy through three main mechanisms. First, DN activates
negative regulatory pathways of autophagy via the PI3K-AKT-mTOR and
PKC-mTOR pathways, where mTORC1 suppresses the assembly and
activation of the autophagy initiation complex, which blocks
autophagy initiation. Second, hyperglycemia-induced NF-κB
translocation into the nucleus allows NF-κB to bind the negative
regulatory element in the Beclin1 promoter, downregulating Beclin1
expression and activity and inhibiting autophagy nucleation.
Finally, excessive ROS and mtROS production damages lysosomal
membranes, resulting in lysosomal membrane permeabilization (LMP)
(164,165). LMP causes lysosomal hydrolases
to leak into the cytoplasm, degrading autophagy-related proteins,
blocking autophagosome degradation, disrupting autophagic flux and
leading to autophagosome accumulation (166). Blocked autophagy initiation,
defective nucleation and disrupted flux lead to a marked reduction
in podocyte autophagic activity, preventing effective clearance of
damaged intracellular components, particularly mitochondria. This
causes mitochondrial dysfunction, activates the NLRP3 inflammasome
and aggravates cellular injury (132,167).
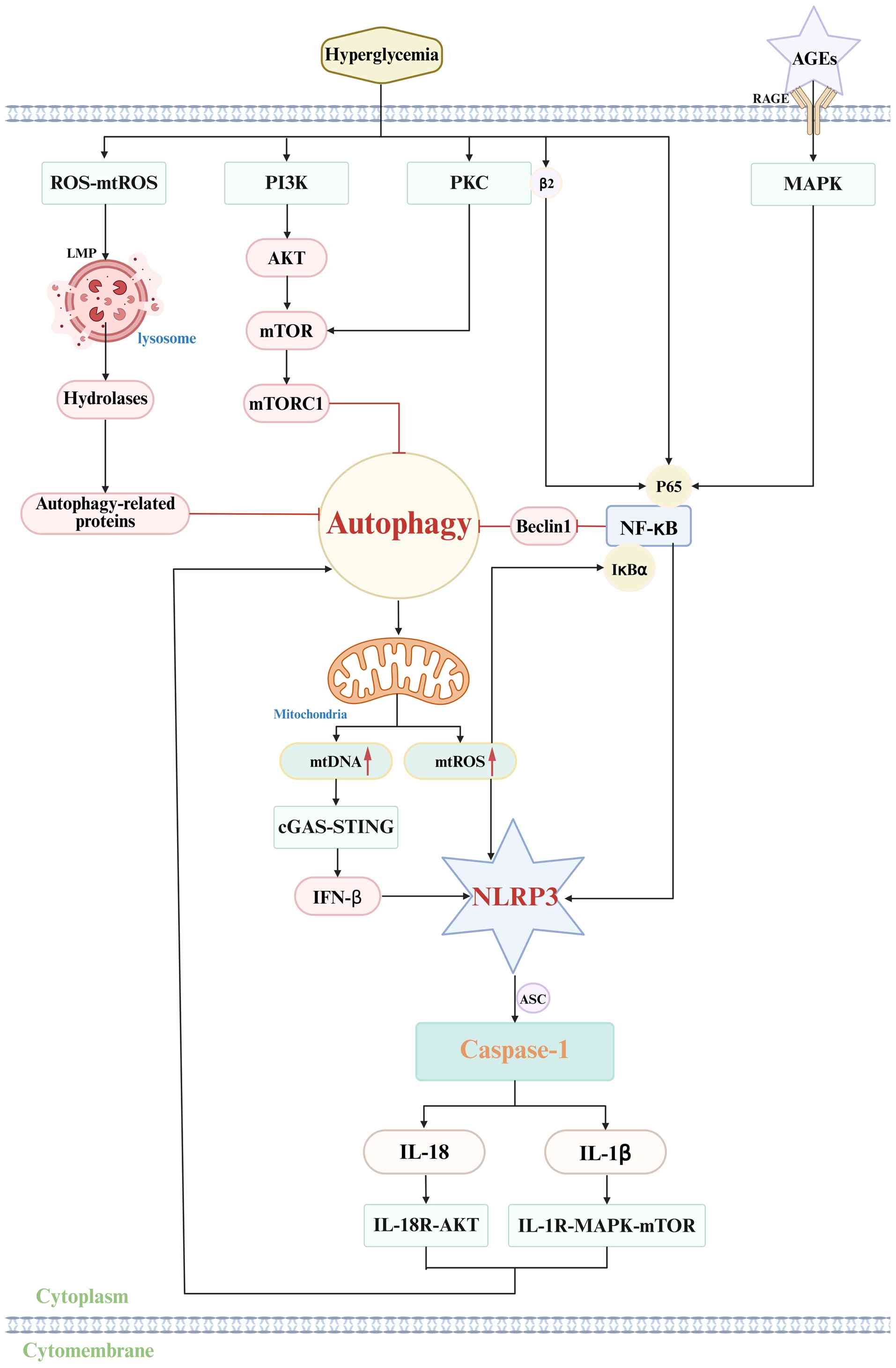
The activation of the NLRP3 inflammasome involves
two stages, initiation and activation, followed by the subsequent
release of effector molecules. During the activation phase,
hyperglycemia and autophagy deficiency synergistically activate the
NF-κB pathway. Hyperglycemia triggers NF-κB through PKC-b2 and the
AGEs-RAGE pathway, with AGEs binding to RAGE to activate MAPK and
NF-κB, leading to phosphorylation and nuclear translocation of the
NF-κB subunit. At the same time, impaired autophagy causes
accumulation of mtROS from damaged mitochondria, which promotes
oxidative modification and ubiquitin-mediated degradation of IκBα
and further enhances NF-κB activity (168,169). Activated NF-κB binds to the κB
binding site in the promoter region of NLRP3, pro-IL-1β and
pro-IL-18 genes after entering the nucleus, which markedly
upregulates their mRNA transcription and protein expression levels,
thus providing the necessary components for inflammasome assembly
(170). Mitochondrial
dysfunction caused by autophagy defects leads to the release of
multiple DAMPs in the activation phase. mtROS oxidize cysteine
residues in NLRP3, which alters the conformation of NLRP3 and
converts it into an active state. The loss of the mitochondrial
outer membrane potential triggers the opening of the mitochondrial
permeability transition pore, causing mtDNA to leak into the
cytoplasm, where it either directly binds to the LRR domain of
NLRP3 or indirectly promotes NLRP3 oligomerization via the
cGAS-STING pathway. Once activated, NLRP3 oligomerizes through the
NACHT domain, recruits the adaptor protein ASC via the CARD domain,
and induces ASC self-polymerization through the PYD domain to form
the ASC speck, which subsequently recruits pro-caspase-1 and drives
its autocatalytic activation (171,172). In the effector molecule release
phase, activated caspase-1 generates mature cytokines by
specifically cleaving pro-IL-1β and pro-IL-18. At the same time,
caspase-1 cleaves the pyroptosis effector protein GSDMD to produce
the GSDMD-N terminal fragment, which inserts into the podocyte
membrane to form pores, leading to cytoplasmic content leakage and
triggering podocyte pyroptosis, thereby exacerbating DN progression
(Fig. 8) (173).
DN is driven by a complex, interconnected network of
inflammatory and fibrotic signaling cascades, in which the NLRP3
inflammasome acts as a key synergistic hub rather than an exclusive
dominant pathogenic mechanism. The canonical TGF-/Smad pathway, as
the core fibrotic driver that mediates excessive ECM deposition and
tubular EMT in DN, engages in bidirectional crosstalk with the
NLRP3 inflammasome: TGF-upregulates TXNIP expression to potentiate
NLRP3 activation, while IL-1 generated by NLRP3 inflammasome
signaling in turn amplifies TGF-mediated renal fibrosis, forming a
pro-fibrotic positive feedback loop (174,175). The TLR4/MAPK/NF-κB pathway
serves as a pivotal upstream priming signal for the global
inflammatory cascade in DN and operates in parallel with the NLRP3
axis, transcriptionally inducing the expression of NLRP3, pro-IL-1β
and pro-IL-18 and cooperating with the NLRP3 inflammasome to
promote renal macrophage infiltration and sustained inflammatory
responses, thereby collectively exacerbating inflammatory renal
injury (7,144). The AGEs/RAGE pathway
constitutes a core metabolic-immune linkage in DN, whereby
hyperglycemia-induced AGEs bind to their receptor RAGE
simultaneously and activate the NLRP3 inflammasome and the
MAPK/NF-κB signaling cascade, acting as a common upstream trigger
that bridges metabolic disorders and inflammatory activation across
multiple DN-related pathways (21,176). The JAK/STAT pathway primarily
mediates the transduction of pro-inflammatory cytokines including
IL-6 and IFN-γ in DN; its activation facilitates the assembly of
the NLRP3 inflammasome, whereas inflammatory cytokines released
upon NLRP3 activation further stimulate the JAK/STAT cascade,
establishing a signal amplification loop that aggravates renal
inflammatory damage (144,177,178). The Wnt/β-catenin pathway
promotes podocyte dedifferentiation and renal fibrosis, with its
activation closely associated with OS, a key initiator of NLRP3
inflammasome activation. The Wnt/β-catenin pathway acts in parallel
with NLRP3-mediated pyroptosis to jointly induce renal structural
lesions and functional deterioration (179-181). Collectively, these pathways
interact and synergize to propel the pathological progression of
DN, and the NLRP3 inflammasome functions as a key contributory
component within this multi-pathway regulatory network.
Targeting the NLRP3 inflammasome represents a
promising contributory therapeutic strategy for DN, complementing
interventions against other major inflammatory and fibrotic
pathways. Notable progress has been made in developing
NLRP3-specific inhibitors. MCC950, the first selective NLRP3
inhibitor to enter preclinical studies, has demonstrated marked
efficacy in db/db mouse models of DN. By suppressing the
NLRP3/caspase-1/IL-1β pathway, MCC950 reduced renal inflammation
and fibrosis, improved renal function parameters and alleviated
glomerular basement membrane thickening as well as podocyte injury
(182,183). BAY11-7082, a compound initially
identified as an NF-κB inhibitor, has been rigorously verified to
suppress NLRP3 inflammasome activation specifically by inhibiting
NLRP3 ATPase activity in DN models, and this renoprotective effect
is independent of its canonical NF-κB inhibitory function. The
specificity of this mechanism in DN is supported by multiple lines
of evidence: i) BAY11-7082 directly binds to the NACHT (ATPase)
domain of NLRP3 in renal intrinsic cells and diabetic mouse
kidneys, without targeting the ATPase of other NLR family
inflammasomes (such as NLRC4 and AIM2) (184); ii) the anti-inflammatory and
renal protective effects of BAY11-7082 are completely lost in
NLRP3-knockout diabetic mice and NLRP3-silenced renal cells,
excluding off-target anti-inflammatory interference (183); and iii) the low concentration
used in DN studies exerts no notable cytotoxicity on renal cells
(normal cell viability and apoptosis rate), ruling out non-specific
cytotoxic effects as a confounding factor (185). Other studies have shown that
dapagliflozin inhibits the miR-155-5p/HO-1/NLRP3 axis in diabetic
models, reduces pyroptosis and delays DN development (186). In addition, multiple pathways
exist to reduce DN by inhibiting NLRP3 (187-189) (Table II) (190-201).
With the development of traditional Chinese
medicine, certain monomers of Chinese herbs have been identified
for use in the treatment of DN. Astragaloside IV (AS-IV), the main
active ingredient of Astragalus membranaceus, has
pharmacological effects including anti-inflammatory, antioxidant,
hypoglycemic, lipid-lowering and anti-fibrotic effects (202). Studies have shown that AS-IV
inhibits hyperglycemia-induced NLRP3 activation in mouse podocytes
and reduces podocyte death by upregulating SIRT6 and downregulating
HIF-1α, thereby suppressing the expression of NLRP3, caspase-1,
GSDMD, IL-1β and IL-18. In addition, AS-IV increases endogenous
Klotho expression levels by modulating the NF-κB/NLRP3 signaling
pathway while improving OS, protecting glomerular podocytes,
reducing inflammation and alleviating endoplasmic reticulum stress
(203-205). Triptolide, an essential active
ingredient extracted from the traditional Chinese medicine
Tripterygium wilfordii, reduces hyperglycemia-induced EMT in
podocytes, inhibits NLRP3 inflammasome activation and prevents
pyroptosis of podocytes (206).
The mechanism is associated with upregulation of Nrf2 and HO-1
protein expression and a reduction in ROS levels (207,208). Ginsenosides are active monomer
components of ginseng and have a wide range of pharmacological
properties, including antioxidant, anti-inflammatory, hypoglycemic
and anti-aging effects (209).
Different ginsenosides exert their effects through distinct
mechanisms. Compound K inhibits TXNIP-mediated NLRP3 inflammasome
activation, thereby reducing the expression of inflammatory
cytokines such as IL-1β and IL-18, improving renal function and
mitigating inflammatory injury (210). Ginsenoside Rg1 protects DN
podocytes from hyperlipidemic-induced damage through the
mTOR/NF-κB/NLRP3 signaling axis (211). Ginsenoside Rg3 inhibits NLRP3
inflammasome activation in human and mouse macrophages, blocks
IL-1β secretion and caspase-1 activation and improves renal injury
(212). Ginsenoside Rg5
inhibits NLRP3 inflammasome activation, reduces inflammatory
response, lowers blood glucose, creatinine and urea nitrogen
levels, and improves renal pathology (213). Other monomers of traditional
Chinese medicine have been reported to delay the progression of DN
by inhibiting NLRP3 (Table SI)
(203-251).
The application of traditional Chinese medicine
prescriptions has also demonstrated notable therapeutic benefits in
DN. Buzhong yiqi decoction is reported to inhibit activation of the
NLRP3 inflammasome pathway, restore the Th1/Th2 immune balance and
alleviate DN progression (237). The Yi shen pai du formula,
composed of Astragalus membranaceus, rhubarb, leeches and
silkworm pupae, can reduce OS and the release of inflammatory
factors, relieve renal fibrosis, prevent EMT in renal tubular
epithelial cells and inhibit the formation of NLRP3 inflammasomes,
mainly by activating the Nrf2 pathway (238,239). Zuogui-jiangtang-yishen is
derived from the classic Chinese medicine formula Zuogui pills and
targets the mROS-NLRP3 axis, inhibiting intestinal-origin
Trimethylamine N-oxide-induced pyroptosis, improving glucose and
lipid metabolism disorders, protecting renal function and delaying
DN progression (240,241). The Yiqi-huoxue-jiangzhuo
formula has been shown to improve chronic kidney disease (CKD) and
its associated complications. Its cardioprotective effects in CKD
mice are associated with modulation of the gut microbiota and
inhibition of the NLRP3 inflammasome (242,251). Tongluo yishen decoction,
composed of Salvia miltiorrhiza and Stephania
tetrandra, can improve renal fibrosis by regulating
NLRP3-mediated cell death and had a notable effect in rat models
after 2 weeks of intervention (243,244). Numerous traditional Chinese
medicine prescriptions have also been reported to reduce DN by
inhibiting NLRP3 (Table SI)
(203-251).
Gene therapy refers to introducing functional
exogenous genes into target cells to correct or compensate for
diseases caused by defective or abnormal genes. It also encompasses
genetic modification techniques in which exogenous genes are
delivered into appropriate recipient cells so that their gene
products exert therapeutic effects. In a broader sense, gene
therapy includes various strategies and emerging technologies that
intervene at the DNA level to treat human diseases. Specifically,
in the context of the present topic, gene therapy can be applied as
a novel strategy for NLRP3 inflammasome-mediated therapeutic
targeting in DN, by regulating the expression of NLRP3
inflammasome-related genes or delivering functional genes to
inhibit NLRP3-mediated neuronal pyroptosis and neuroinflammation,
thereby achieving therapeutic effects on diabetic neuropathy
(252,253).
MicroRNAs (miRNAs/miRs) are key post-transcriptional
regulators involved in metabolic and inflammatory processes,
offering new insights into the molecular mechanisms underlying DN.
Non-coding RNAs modulate gene expression through epigenetic
pathways and thus present opportunities for the development of
preventive therapeutics. Experimental studies have shown that
specific miRNAs, such as miR-192, can alleviate kidney injury
(254,255). Long non-coding RNAs (lncRNA),
such as lncRNA MALAT1, can indirectly regulate the NLRP3 pathway by
adsorbing miRNA. This non-coding RNA-targeted intervention offers a
new direction for gene therapy (256,257) (Table III) (258-263). Silencing the NLRP3 or ASC gene
using RNA interference may effectively reduce inflammasome
expression in renal tissue, thereby attenuating the inflammatory
response.
The CRISPR-Cas9 system has emerged as a powerful
gene-editing tool capable of precisely cutting, inserting or
deleting DNA sequences. Knocking out the NLRP3 gene with CRISPR can
directly block inflammasome assembly and activation, reduce IL-1β
and IL-18 secretion, and thereby alleviate renal inflammation
(264,265). Although the application of the
technology in kidney disease is still in its early stages, its
precision offers a promising direction for targeted therapy
(Table IV) (266-270).
The present review outlines the multifactorial
pathogenesis of DN. It should be emphasized that the NLRP3
inflammasome does not operate as the sole dominant mechanism in DN;
rather, it functions in concert with other pathways. In contrast to
existing reviews, the present study proposes a novel hierarchical
regulatory framework for NLRP3 inflammasome activation in DN,
systematically dissects the mitochondrial metabolic-cGAS-STING
synergy mechanism, and provides evidence-stratified therapeutic
strategies for clinical translation as a complementary approach to
other anti-inflammatory and anti-fibrotic therapies. Several
pathways involved in NLRP3 inflammasome activation were discussed,
including key signaling molecules such as NF-κB, TXNIP, Nrf2 and
PI3K/Akt, which act via interconnecting mechanisms and contribute
to the complexity of inflammasome activation. In addition,
therapeutic strategies targeting NLRP3 inflammasomes to alleviate
renal injury in DN were summarized. Overall, the multifactorial
pathogenesis of DN remains incompletely understood. The NLRP3
inflammasome is a key contributory hub within the inflammatory
network of DN, acting in synergy with TGF-β/Smad, TLR4/NF-κB and
other core pathways. Further investigation into the crosstalk
between NLRP3 and parallel pathways will facilitate the development
of combination therapeutic strategies for DN, with the expectation
that more effective and precise clinical interventions will be
developed in the future.
Not applicable.
HY and XW conceived and supervised the present
study; HY, XW, ShLi, XG, TX, SaLi, RY and TL analyzed data; XW
wrote the manuscript. All authors reviewed the results and approved
the final version of the manuscript. Data authentication not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
This research is supported by the Doctoral Research Start-up
Fund Project of Mudanjiang Medical University (grant no.
2021-MYBSKY-027); Heilongjiang Province Natural Science Foundation
Project (grant no. SS2023H003); Heilongjiang Province Traditional
Chinese Medicine Research Project (grant no. ZHY2024-212); Basic
Scientific Research Business Research Project of Heilongjiang
(grant no. 2024-KYYWF-0502); Program for Young Talents of Basic
Research in Universities of Heilongjiang Province (grant no.
YQJH2024254).
|
1
|
Gheith O, Farouk N, Nampoory N, Halim MA
and Al-Otaibi T: Diabetic kidney disease: World wide difference of
prevalence and risk factors. J Nephropharmacol. 5:49–56.
2015.PubMed/NCBI
|
|
2
|
NCD Risk Factor Collaboration (NCD-RisC):
Worldwide trends in diabetes prevalence and treatment from 1990 to
2022: A pooled analysis of 1108 population-representative studies
with 141 million participants. Lancet. 404:2077–2093. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Harding JL, Pavkov ME, Magliano DJ, Shaw
JE and Gregg EW: Global trends in diabetes complications: A review
of current evidence. Diabetologia. 62:3–16. 2019. View Article : Google Scholar
|
|
4
|
Wal P, Tyagi S, Pal RS, Yadav A and
Jaiswal R: A strategic investigation on diabetic nephropathy; Its
conceptual model and clinical manifestations: A review. Curr
Diabetes Rev. 19:e2604222040362023. View Article : Google Scholar
|
|
5
|
Donate-Correa J, Martín-Núñez E,
Muros-de-Fuentes M, Mora-Fernández C and Navarro-González JF:
Inflammatory cytokines in diabetic nephropathy. J Diabetes Res.
2015:9484172015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li Y, Huang H, Liu B, Zhang Y, Pan X, Yu
XY, Shen Z and Song YH: Inflammasomes as therapeutic targets in
human diseases. Signal Transduct Target Ther. 6:2472021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tang SCW and Yiu WH: Innate immunity in
diabetic kidney disease. Nat Rev Nephrol. 16:206–222. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jo EK, Kim JK, Shin DM and Sasakawa C:
Molecular mechanisms regulating NLRP3 inflammasome activation. Cell
Mol Immunol. 13:148–159. 2016. View Article : Google Scholar :
|
|
9
|
Duncan JA, Bergstralh DT, Wang Y,
Willingham SB, Ye Z, Zimmermann AG and Ting JP: Cryopyrin/NALP3
binds ATP/dATP, is an ATPase, and requires ATP binding to mediate
inflammatory signaling. Proc Natl Acad Sci USA. 104:8041–8046.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Swanson KV, Deng M and Ting JP: The NLRP3
inflammasome: Molecular activation and regulation to therapeutics.
Nat Rev Immunol. 19:477–489. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cabral JE, Wu A, Zhou H, Pham MA, Lin S
and McNulty R: Targeting the NLRP3 inflammasome for inflammatory
disease therapy. Trends Pharmacol Sci. 46:503–519. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Duewell P, Kono H, Rayner KJ, Sirois CM,
Vladimer G, Bauernfeind FG, Abela GS, Franchi L, Nuñez G, Schnurr
M, et al: NLRP3 inflammasomes are required for atherogenesis and
activated by cholesterol crystals. Nature. 464:1357–1361. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Grebe A, Hoss F and Latz E: NLRP3
inflammasome and the IL-1 pathway in atherosclerosis. Circ Res.
122:1722–1740. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fu Y, Wu N and Zhao D: Function of NLRP3
in the pathogenesis and development of diabetic nephropathy. Med
Sci Monit. 23:3878–3884. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Williams BM, Cliff CL, Lee K, Squires PE
and Hills CE: The role of the NLRP3 inflammasome in mediating
glomerular and tubular injury in diabetic nephropathy. Front
Physiol. 13:9075042022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Newton K, Dixit VM and Kayagaki N: Dying
cells fan the flames of inflammation. Science. 374:1076–1080. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu M, Han W, Song S, Du Y, Liu C, Chen N,
Wu H, Shi Y and Duan H: NLRP3 deficiency ameliorates renal
inflammation and fibrosis in diabetic mice. Mol Cell Endocrinol.
478:115–125. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Selby NM and Taal MW: An updated overview
of diabetic nephropathy: Diagnosis, prognosis, treatment goals and
latest guidelines. Diabetes Obes Metab. 22(Suppl 1): S3–S15. 2020.
View Article : Google Scholar
|
|
19
|
Li KX, Ji MJ and Sun HJ: An updated
pharmacological insight of resveratrol in the treatment of diabetic
nephropathy. Gene. 780:1455322021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang L, Jiang L, Xu R, Zhang X, Zhang B
and Yue R: Epidemiological research on diabetic nephropathy at
global, regional, and national levels from 1990 to 2021: An
analysis derived from the global burden of disease 2021 study.
Front Endocrinol (Lausanne). 16:16470642025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wada J and Makino H: Inflammation and the
pathogenesis of diabetic nephropathy. Clin Sci (Lond). 124:139–152.
2013. View Article : Google Scholar
|
|
22
|
Barrera-Chimal J and Jaisser F:
Pathophysiologic mechanisms in diabetic kidney disease: A focus on
current and future therapeutic targets. Diabetes Obes Metab.
22(Suppl 1): S16–S31. 2020. View Article : Google Scholar
|
|
23
|
Samsu N: Diabetic nephropathy: Challenges
in pathogenesis, diagnosis, and treatment. Biomed Res Int.
2021:14974492021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen J, Liu Q, He J and Li Y: Immune
responses in diabetic nephropathy: Pathogenic mechanisms and
therapeutic target. Front Immunol. 13:9587902022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Warren AM, Knudsen ST and Cooper ME:
Diabetic nephropathy: An insight into molecular mechanisms and
emerging therapies. Expert Opin Ther Targets. 23:579–591. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang N and Zhang C: Recent advances in the
management of diabetic kidney disease: Slowing progression. Int J
Mol Sci. 25:30862024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Han YP, Liu LJ, Yan JL, Chen MY, Meng XF,
Zhou XR and Qian LB: Autophagy and its therapeutic potential in
diabetic nephropathy. Front Endocrinol (Lausanne). 14:11394442023.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Barutta F, Bellini S, Kimura S, Hase K,
Corbetta B, Corbelli A, Fiordaliso F, Bruno S, Biancone L, Barreca
A, et al: Protective effect of the tunneling nanotube-TNFAIP2/M-sec
system on podocyte autophagy in diabetic nephropathy. Autophagy.
19:505–524. 2023. View Article : Google Scholar :
|
|
29
|
Barutta F, Bellini S and Gruden G:
Mechanisms of podocyte injury and implications for diabetic
nephropathy. Clin Sci (Lond). 136:493–520. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li X, Zhang Y, Xing X, Li M, Liu Y, Xu A
and Zhang J: Podocyte injury of diabetic nephropathy: Novel
mechanism discovery and therapeutic prospects. Biomed Pharmacother.
168:1156702023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen Y, Chen R, Ji X, Zeng Z and Guan C:
NLRP3 inflammasome-mediated pyroptosis in diabetic nephropathy:
Pathogenic mechanisms and therapeutic targets. J Inflamm Res.
18:8399–8418. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Vodošek Hojs N, Bevc S, Ekart R and Hojs
R: Oxidative stress markers in chronic kidney disease with emphasis
on diabetic nephropathy. Antioxidants (Basel). 9:9252020.
View Article : Google Scholar
|
|
33
|
Yang M and Zhang C: The role of innate
immunity in diabetic nephropathy and their therapeutic
consequences. J Pharm Anal. 14:39–51. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Takeuchi O and Akira S: Pattern
recognition receptors and inflammation. Cell. 140:805–820. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Scarfe L, Mackie GM and Maslowski KM:
Inflammasome-independent functions of NAIPs and NLRs in the
intestinal epithelium. Biochem Soc Trans. 49:2601–2610. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chou WC, Jha S, Linhoff MW and Ting JP:
The NLR gene family: from discovery to present day. Nat Rev
Immunol. 23:635–654. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Inoue M and Shinohara ML: NLRP3
Inflammasome and MS/EAE. Autoimmune Dis. 2013:8591452013.PubMed/NCBI
|
|
38
|
Clay GM, Sutterwala FS and Wilson ME: NLR
proteins and parasitic disease. Immunol Res. 59:142–152. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sundaram B, Tweedell RE, Prasanth Kumar S
and Kanneganti TD: The NLR family of innate immune and cell death
sensors. Immunity. 57:674–699. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kodi T, Sankhe R, Gopinathan A, Nandakumar
K and Kishore A: New insights on NLRP3 inflammasome: Mechanisms of
activation, inhibition, and epigenetic regulation. J Neuroimmune
Pharmacol. 19:72024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhen Y and Zhang H: NLRP3 inflammasome and
inflammatory bowel disease. Front Immunol. 10:2762019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Blevins HM, Xu Y, Biby S and Zhang S: The
NLRP3 inflammasome pathway: A review of mechanisms and inhibitors
for the treatment of inflammatory diseases. Front Aging Neurosci.
14:8790212022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Boucher D, Monteleone M, Coll RC, Chen KW,
Ross CM, Teo JL, Gomez GA, Holley CL, Bierschenk D, Stacey KJ, et
al: Caspase-1 self-cleavage is an intrinsic mechanism to terminate
inflammasome activity. J Exp Med. 215:827–840. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wang K, Sun Q, Zhong X, Zeng M, Zeng H,
Shi X, Li Z, Wang Y, Zhao Q, Shao F and Ding J: Structural
mechanism for GSDMD targeting by autoprocessed caspases in
pyroptosis. Cell. 180:941–955.e20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Vande Walle L and Lamkanfi M: Snapshot of
a deadly embrace: The Caspase-1-GSDMD interface. Immunity. 53:6–8.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Christgen S, Place DE and Kanneganti TD:
Toward targeting inflammasomes: Insights into their regulation and
activation. Cell Res. 30:315–327. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Huang Y, Xu W and Zhou R: NLRP3
inflammasome activation and cell death. Cell Mol Immunol.
18:2114–2127. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Maharana J, Vats A, Gautam S, Nayak BP,
Kumar S, Sendha J and De S: POP1 might be recruiting its type-Ia
interface for NLRP3-mediated PYD-PYD interaction: Insights from MD
simulation. J Mol Recognit. 30:2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Biasizzo M and Kopitar-Jerala N: Interplay
between NLRP3 inflammasome and autophagy. Front Immunol.
11:5918032020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Andreeva L, David L, Rawson S, Shen C,
Pasricha T, Pelegrin P and Wu H: NLRP3 cages revealed by
full-length mouse NLRP3 structure control pathway activation. Cell.
184:6299–6312.e22. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Chung C, Park SY, Huh JY, Kim NH, Shon C,
Oh EY, Park YJ, Lee SJ, Kim HC and Lee SW: Fine particulate matter
aggravates smoking induced lung injury via NLRP3/caspase-1 pathway
in COPD. J Inflamm (Lond). 21:132024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Adinolfi E, Giuliani AL, De Marchi E,
Pegoraro A, Orioli E and Di Virgilio F: The P2X7 receptor: A main
player in inflammation. Biochem Pharmacol. 151:234–244. 2018.
View Article : Google Scholar
|
|
53
|
Coll RC, Schroder K and Pelegrín P: NLRP3
and pyroptosis blockers for treating inflammatory diseases. Trends
Pharmacol Sci. 43:653–668. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Hoseini Z, Sepahvand F, Rashidi B,
Sahebkar A, Masoudifar A and Mirzaei H: NLRP3 inflammasome: Its
regulation and involvement in atherosclerosis. J Cell Physiol.
233:2116–2132. 2018. View Article : Google Scholar
|
|
55
|
Zhou K, Enkhjargal B, Xie Z, Sun C, Wu L,
Malaguit J, Chen S, Tang J, Zhang J and Zhang JH: Dihydrolipoic
acid inhibits lysosomal rupture and NLRP3 through
lysosome-associated membrane protein-1/calcium/calmodulin-dependent
protein kinase II/TAK1 pathways after subarachnoid hemorrhage in
rat. Stroke. 49:175–183. 2018. View Article : Google Scholar
|
|
56
|
Elliott EI, Miller AN, Banoth B, Iyer SS,
Stotland A, Weiss JP, Gottlieb RA, Sutterwala FS and Cassel SL:
Cutting edge: Mitochondrial assembly of the NLRP3 inflammasome
complex is initiated at priming. J Immunol. 200:3047–3052. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Liu Q, Zhang D, Hu D, Zhou X and Zhou Y:
The role of mitochondria in NLRP3 inflammasome activation. Mol
Immunol. 103:115–124. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Ichinohe T, Yamazaki T, Koshiba T and
Yanagi Y: Mitochondrial protein mitofusin 2 is required for NLRP3
inflammasome activation after RNA virus infection. Proc Natl Acad
Sci USA. 110:17963–17968. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Li R, Deng H, Han Y, Tong Y, Hou Y, Huang
T, Xiao M, Deng L, Zhao X, Chen Y, et al: Therapeutic effects of
Lianhua Qingke on COPD and influenza virus-induced exacerbation of
COPD are associated with the inhibition of NF-κB signaling and
NLRP3 inflammasome responses. Int Immunopharmacol. 142:1132132024.
View Article : Google Scholar
|
|
60
|
Kim K, Kim HJ, Binas B, Kang JH and Chung
IY: Inflammatory mediators ATP and S100A12 activate the NLRP3
inflammasome to induce MUC5AC production in airway epithelial
cells. Biochem Biophys Res Commun. 503:657–664. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yang J, Zhang M, Luo Y, Xu F, Gao F, Sun
Y, Yang B and Kuang H: Protopine ameliorates OVA-induced asthma
through modulatingTLR4/MyD88/NF-κB pathway and NLRP3
inflammasome-mediated pyroptosis. Phytomedicine. 126:1554102024.
View Article : Google Scholar
|
|
62
|
Song MY, Wang JX, Sun YL, Han ZF, Zhou YT,
Liu Y, Fan TH, Li ZG, Qi XM, Luo Y, et al: Tetrandrine alleviates
silicosis by inhibiting canonical and non-canonical NLRP3
inflammasome activation in lung macrophages. Acta Pharmacol Sin.
43:1274–1284. 2022. View Article : Google Scholar :
|
|
63
|
Wu S and Huang J: Resveratrol alleviates
Staphylococcus aureus pneumonia by inhibition of the NLRP3
inflammasome. Exp Ther Med. 14:6099–6104. 2017.PubMed/NCBI
|
|
64
|
Chen L and Liu Z: Downregulation of FSTL-1
attenuates the inflammation injury during Streptococcus pneumoniae
infection by inhibiting the NLRP3 and TLR4/NF-κB signaling pathway.
Mol Med Rep. 20:5345–5352. 2019.PubMed/NCBI
|
|
65
|
Fei X, Chen S, Li L, Xu X, Wang H, Ke H,
He C, Xie C, Wu X, Liu J, et al: Helicobacter pylori infection
promotes M1 macrophage polarization and gastric inflammation by
activation of NLRP3 inflammasome via TNF/TNFR1 axis. Cell Commun
Signal. 23:62025. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Mridha AR, Wree A, Robertson AAB, Yeh MM,
Johnson CD, Van Rooyen DM, Haczeyni F, Teoh NC, Savard C, Ioannou
GN, et al: NLRP3 inflammasome blockade reduces liver inflammation
and fibrosis in experimental NASH in mice. J Hepatol. 66:1037–1046.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Arrè V, Scialpi R, Centonze M, Giannelli
G, Scavo MP and Negro R: The 'speck'-tacular oversight of the
NLRP3-pyroptosis pathway on gastrointestinal inflammatory diseases
and tumorigenesis. J Biomed Sci. 30:902023. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Rong J, Han C, Huang Y, Wang Y, Qiu Q,
Wang M, Wang S, Wang R, Yang J, Li X, et al: Inhibition of xanthine
oxidase alleviated pancreatic necrosis via HIF-1α-regulated LDHA
and NLRP3 signaling pathway in acute pancreatitis. Acta Pharm Sin
B. 14:3591–3604. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Li W, Wang K, Liu Y, Wu H, He Y, Li C,
Wang Q, Su X, Yan S, Su W, et al: A novel drug combination of
mangiferin and cinnamic acid alleviates rheumatoid arthritis by
inhibiting TLR4/NFκB/NLRP3 activation-induced pyroptosis. Front
Immunol. 13:9129332022. View Article : Google Scholar
|
|
70
|
Liu J, Jia S, Yang Y, Piao L, Wang Z, Jin
Z and Bai L: Exercise induced meteorin-like protects chondrocytes
against inflammation and pyroptosis in osteoarthritis by inhibiting
PI3K/Akt/NF-κB and NLRP3/caspase-1/GSDMD signaling. Biomed
Pharmacother. 158:1141182023. View Article : Google Scholar
|
|
71
|
Fan J, Du G, Ba T and Sun H: NLRP3
inflammasome-mediated pyroptosis in osteoporosis: Osteoimmune
mechanisms and therapeutic targeting. J Cell Mol Med.
29:e707982025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Liu YR, Wang JQ and Li J: Role of NLRP3 in
the pathogenesis and treatment of gout arthritis. Front Immunol.
14:11378222023. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhao Y, Qiu C, Wang W, Peng J, Cheng X,
Shangguan Y, Xu M, Li J, Qu R, Chen X, et al: Cortistatin protects
against intervertebral disc degeneration through targeting
mitochondrial ROS-dependent NLRP3 inflammasome activation.
Theranostics. 10:7015–7033. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhao Y, Shao C, Zhou H, Yu L, Bao Y, Mao
Q, Yang J and Wan H: Salvianolic acid B inhibits atherosclerosis
and TNF-α-induced inflammation by regulating NF-κB/NLRP3 signaling
pathway. Phytomedicine. 119:1550022023. View Article : Google Scholar
|
|
75
|
Wang D, Yu X, Gao K, Li F, Li X, Pu H,
Zhang P, Guo S and Wang W: Sweroside alleviates pressure
overload-induced heart failure through targeting CaMKIIδ to inhibit
ROS-mediated NF-κB/NLRP3 in cardiomyocytes. Redox Biol.
74:1032232024. View Article : Google Scholar
|
|
76
|
Liu S, Bi Y, Han T, Li YE, Wang Q, Wu NN,
Xu C, Ge J, Hu R and Zhang Y: The E3 ubiquitin ligase MARCH2
protects against myocardial ischemia-reperfusion injury through
inhibiting pyroptosis via negative regulation of PGAM5/MAVS/NLRP3
axis. Cell Discov. 10:242024. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Chakraborty R, Tabassum H and Parvez S:
NLRP3 inflammasome in traumatic brain injury: Its implication in
the disease pathophysiology and potential as a therapeutic target.
Life Sci. 314:1213522023. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Yan YQ, Zheng R, Liu Y, Ruan Y, Lin ZH,
Xue NJ, Chen Y, Zhang BR and Pu JL: Parkin regulates microglial
NLRP3 and represses neurodegeneration in Parkinson's disease. Aging
Cell. 22:e138342023. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Moonen S, Koper MJ, Van Schoor E,
Schaeverbeke JM, Vandenberghe R, von Arnim CAF, Tousseyn T, De
Strooper B and Thal DR: Pyroptosis in Alzheimer's disease: Cell
type-specific activation in microglia, astrocytes and neurons. Acta
Neuropathol. 145:175–195. 2023. View Article : Google Scholar
|
|
80
|
Brint A, Greene S, Fennig-Victor AR and
Wang S: Multiple sclerosis: The NLRP3 inflammasome, gasdermin D,
and therapeutics. Ann Transl Med. 12:622024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Li J, Xu P, Hong Y, Xie Y, Peng M, Sun R,
Guo H, Zhang X, Zhu W, Wang J and Liu X: Lipocalin-2-mediated
astrocyte pyroptosis promotes neuroinflammatory injury via NLRP3
inflammasome activation in cerebral ischemia/reperfusion injury. J
Neuroinflammation. 20:1482023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Kuemmerle-Deschner JB, Lohse P, Koetter I,
Dannecker GE, Reess F, Ummenhofer K, Koch S, Tzaribachev N,
Bialkowski A and Benseler SM: NLRP3 E311K mutation in a large
family with Muckle-Wells syndrome-description of a heterogeneous
phenotype and response to treatment. Arthritis Res Ther.
13:R1962011. View
Article : Google Scholar
|
|
83
|
Neven B, Callebaut I, Prieur AM, Feldmann
J, Bodemer C, Lepore L, Derfalvi B, Benjaponpitak S, Vesely R,
Sauvain MJ, et al: Molecular basis of the spectral expression of
CIAS1 mutations associated with phagocytic cell-mediated
autoinflammatory disorders CINCA/NOMID, MWS, and FCU. Blood.
103:2809–2815. 2004. View Article : Google Scholar
|
|
84
|
Chen S, Li Z, Hu X, Zhang H, Chen W, Xu Q,
Tang L, Ge H, Zhen Q, Yong L, et al: Rare mutations in NLRP3 and
NLRP12 associated with familial cold autoinflammatory syndrome: Two
Chinese pedigrees. Clin Rheumatol. 41:3461–3470. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Theodoropoulou K, Spel L, Zaffalon L,
Delacrétaz M, Hofer M and Martinon F: NLRP3 leucine-rich repeats
control induced and spontaneous inflammasome activation in
cryopyrin-associated periodic syndrome. J Allergy Clin Immunol.
151:222–232.e9. 2023. View Article : Google Scholar
|
|
86
|
Povero D, Lazic M, McBride C,
Ambrus-Aikelin G, Stansfield R, Johnson CD, Santini AM, Pranadinata
RF, McGeough MD, Stafford JA, et al: Pharmacology of a potent and
novel inhibitor of the NOD-Like receptor pyrin domain-containing
protein 3 (NLRP3) inflammasome that attenuates development of
nonalcoholic steatohepatitis and liver fibrosis. J Pharmacol Exp
Ther. 386:242–258. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Meier DT, de Paula Souza J and Donath MY:
Targeting the NLRP3 inflammasome-IL-1β pathway in type 2 diabetes
and obesity. Diabetologia. 68:3–16. 2025. View Article : Google Scholar
|
|
88
|
Zhang L, Ai C, Bai M, Niu J and Zhang Z:
NLRP3 inflammasome/pyroptosis: A key driving force in diabetic
cardiomyopathy. Int J Mol Sci. 23:106322022. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Qiu YY and Tang LQ: Roles of the NLRP3
inflammasome in the pathogenesis of diabetic nephropathy. Pharmacol
Res. 114:251–264. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Shahzad K, Fatima S, Khawaja H, Elwakiel
A, Gadi I, Ambreen S, Zimmermann S, Mertens PR, Biemann R and
Isermann B: Podocyte-specific Nlrp3 inflammasome activation
promotes diabetic kidney disease. Kidney Int. 102:766–779. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lin J, Cheng A, Cheng K, Deng Q, Zhang S,
Lan Z, Wang W and Chen J: New insights into the mechanisms of
pyroptosis and implications for diabetic kidney disease. Int J Mol
Sci. 21:70572020. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Wan J, Liu D, Pan S, Zhou S and Liu Z:
NLRP3-mediated pyroptosis in diabetic nephropathy. Front Pharmacol.
13:9985742022. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Shi J, Gao W and Shao F: Pyroptosis:
Gasdermin-mediated programmed necrotic cell death. Trends Biochem
Sci. 42:245–254. 2017. View Article : Google Scholar
|
|
94
|
Liu X, Zhang Z, Ruan J, Pan Y, Magupalli
VG, Wu H and Lieberman J: Inflammasome-activated gasdermin D causes
pyroptosis by forming membrane pores. Nature. 535:153–158. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Rathinam VAK, Zhao Y and Shao F: Innate
immunity to intracellular LPS. Nat Immunol. 20:527–533. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Zamyatina A and Heine H:
Lipopolysaccharide recognition in the crossroads of TLR4 and
Caspase-4/11 mediated inflammatory pathways. Front Immunol.
11:5851462020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Chen Q, Shi P, Wang Y, Zou D, Wu X, Wang
D, Hu Q, Zou Y, Huang Z, Ren J, et al: GSDMB promotes non-canonical
pyroptosis by enhancing caspase-4 activity. J Mol Cell Biol.
11:496–508. 2019. View Article : Google Scholar :
|
|
98
|
Chen A, Liu J, Zhu J, Wang X, Xu Z, Cui Z,
Yao D, Huang Z, Xu M, Chen M, et al: FGF21 attenuates
hypoxia-induced dysfunction and apoptosis in HPAECs through
alleviating endoplasmic reticulum stress. Int J Mol Med.
42:1684–1694. 2018.PubMed/NCBI
|
|
99
|
Kayagaki N, Stowe IB, Lee BL, O'Rourke K,
Anderson K, Warming S, Cuellar T, Haley B, Roose-Girma M, Phung QT,
et al: Caspase-11 cleaves gasdermin D for non-canonical
inflammasome signalling. Nature. 526:666–671. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Bibo-Verdugo B, Snipas SJ, Kolt S, Poreba
M and Salvesen GS: Extended subsite profiling of the pyroptosis
effector protein gasdermin D reveals a region recognized by
inflammatory caspase-11. J Biol Chem. 295:11292–11302. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Rogers C, Erkes DA, Nardone A, Aplin AE,
Fernandes-Alnemri T and Alnemri ES: Gasdermin pores permeabilize
mitochondria to augment caspase-3 activation during apoptosis and
inflammasome activation. Nat Commun. 10:16892019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Jiang M, Qi L, Li L and Li Y: The
caspase-3/GSDME signal pathway as a switch between apoptosis and
pyroptosis in cancer. Cell Death Discov. 6:1122020. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Zeng CY, Li CG, Shu JX, Xu LH, Ouyang DY,
Mai FY, Zeng QZ, Zhang CC, Li RM and He XH: ATP induces
caspase-3/gasdermin E-mediated pyroptosis in NLRP3 pathway-blocked
murine macrophages. Apoptosis. 24:703–717. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Taabazuing CY, Okondo MC and Bachovchin
DA: Pyroptosis and apoptosis pathways engage in bidirectional
crosstalk in monocytes and macrophages. Cell Chem Biol.
24:507–514.e4. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Miao R, Jiang C, Chang WY, Zhang H, An J,
Ho F, Chen P, Zhang H, Junqueira C, Amgalan D, et al: Gasdermin D
permeabilization of mitochondrial inner and outer membranes
accelerates and enhances pyroptosis. Immunity. 56:2523–2541.e8.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Yuan T, Yang HY, Li YP, Shi ZJ, Zhou ZY,
You YP, Ke HY, Yan L, Xu LH, Ouyang DY, et al: Scutellarin inhibits
inflammatory PANoptosis by diminishing mitochondrial ROS generation
and blocking PANoptosome formation. Int Immunopharmacol.
139:1127102024. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Han Y, Xu X, Tang C, Gao P, Chen X, Xiong
X, Yang M, Yang S, Zhu X, Yuan S, et al: Reactive oxygen species
promote tubular injury in diabetic nephropathy: The role of the
mitochondrial ros-txnip-nlrp3 biological axis. Redox Biol.
16:32–46. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Liu Y, Sun R, Lin X, Wu L, Chen H, Shen S,
Li Y, Wei Y and Deng G: Procyanidins and its metabolites by gut
microbiome improves insulin resistance in gestational diabetes
mellitus mice model via regulating NF-κB and NLRP3 inflammasome
pathway. Biomed Pharmacother. 151:1130782022. View Article : Google Scholar
|
|
109
|
Venditti M, Romano MZ, Boccella S, Haddadi
A, Biasi A, Maione S and Minucci S: Type 1 diabetes impairs the
activity of rat testicular somatic and germ cells through
NRF2/NLRP3 pathway-mediated oxidative stress. Front Endocrinol
(Lausanne). 15:13992562024. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Zhang WJ, Chen SJ, Zhou SC, Wu SZ and Wang
H: Inflammasomes and fibrosis. Front Immunol. 12:6431492021.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Rotariu D, Babes EE, Tit DM, Moisi M,
Bustea C, Stoicescu M, Radu AF, Vesa CM, Behl T, Bungau AF and
Bungau SG: Oxidative stress-complex pathological issues concerning
the hallmark of cardiovascular and metabolic disorders. Biomed
Pharmacother. 152:1132382022. View Article : Google Scholar
|
|
112
|
Bigagli E and Lodovici M: Circulating
oxidative stress biomarkers in clinical studies on type 2 diabetes
and its complications. Oxid Med Cell Longev. 2019:59536852019.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Wang M, Hu J, Yan L, Yang Y, He M, Wu M,
Li Q, Gong W, Yang Y, Wang Y, et al: High glucose-induced
ubiquitination of G6PD leads to the injury of podocytes. FASEB J.
33:6296–6310. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Kato H, Watanabe H, Imafuku T, Arimura N,
Fujita I, Noguchi I, Tanaka S, Nakano T, Tokumaru K, Enoki Y, et
al: Advanced oxidation protein products contribute to chronic
kidney disease-induced muscle atrophy by inducing oxidative stress
via CD36/NADPH oxidase pathway. J Cachexia Sarcopenia Muscle.
12:1832–1847. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Liu Z, Yao X, Jiang W, Li W, Zhu S, Liao
C, Zou L, Ding R and Chen J: Advanced oxidation protein products
induce microglia-mediated neuroinflammation via MAPKs-NF-κB
signaling pathway and pyroptosis after secondary spinal cord
injury. J Neuroinflammation. 17:902020. View Article : Google Scholar
|
|
116
|
Jin Q, Liu T, Qiao Y, Liu D, Yang L, Mao
H, Ma F, Wang Y, Peng L and Zhan Y: Oxidative stress and
inflammation in diabetic nephropathy: Role of polyphenols. Front
Immunol. 14:11853172023. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Fan Q, Li R, Wei H, Xue W, Li X, Xia Z,
Zhao L, Qiu Y and Cui D: Research Progress of Pyroptosis in
Diabetic Kidney Disease. Int J Mol Sci. 25:2024. View Article : Google Scholar
|
|
118
|
Su L, Zhang J, Gomez H, Kellum JA and Peng
Z: Mitochondria ROS and mitophagy in acute kidney injury.
Autophagy. 19:401–414. 2023. View Article : Google Scholar :
|
|
119
|
Liu S, Tao J, Duan F, Li H and Tan H: HHcy
induces pyroptosis and atherosclerosis via the lipid raft-mediated
NOX-ROS-NLRP3 inflammasome pathway in apoE(-/-) mice. Cells.
11:24382022. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Wang J and Zhao SM: LncRNA-antisense
non-coding RNA in the INK4 locus promotes pyroptosis via
miR-497/thioredoxin-interacting protein axis in diabetic
nephropathy. Life Sci. 264:1187282021. View Article : Google Scholar
|
|
121
|
Lin Q, Li S, Jiang N, Jin H, Shao X, Zhu
X, Wu J, Zhang M, Zhang Z, Shen J, et al: Inhibiting NLRP3
inflammasome attenuates apoptosis in contrast-induced acute kidney
injury through the upregulation of HIF1A and BNIP3-mediated
mitophagy. Autophagy. 17:2975–2990. 2021. View Article : Google Scholar :
|
|
122
|
Chen Y, Ye X, Escames G, Lei W, Zhang X,
Li M, Jing T, Yao Y, Qiu Z, Wang Z, et al: The NLRP3 inflammasome:
contributions to inflammation-related diseases. Cell Mol Biol Lett.
28:512023. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Yu C, Chen P, Miao L and Di G: The role of
the NLRP3 inflammasome and programmed cell death in acute liver
injury. Int J Mol Sci. 24:30672023. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Peng L, Wen L, Shi QF, Gao F, Huang B,
Meng J, Hu CP and Wang CM: Scutellarin ameliorates pulmonary
fibrosis through inhibiting NF-κB/NLRP3-mediated
epithelial-mesenchymal transition and inflammation. Cell Death Dis.
11:9782020. View Article : Google Scholar
|
|
125
|
Zhao W, Ma L, Cai C and Gong X: Caffeine
inhibits NLRP3 inflammasome activation by suppressing MAPK/NF-κB
and A2aR signaling in LPS-Induced THP-1 macrophages. Int J Biol
Sci. 15:1571–1581. 2019. View Article : Google Scholar
|
|
126
|
Alehashemi S and Goldbach-Mansky R: Human
autoinflammatory diseases mediated by NLRP3-, Pyrin-, NLRP1-, and
NLRC4-inflammasome dysregulation updates on diagnosis, treatment,
and the respective roles of IL-1 and IL-18. Front Immunol.
11:18402020. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Xu J and Núñez G: The NLRP3 inflammasome:
Activation and regulation. Trends Biochem Sci. 48:331–344. 2023.
View Article : Google Scholar
|
|
128
|
Jia Y, Cui R, Wang C, Feng Y, Li Z, Tong
Y, Qu K, Liu C and Zhang J: Metformin protects against intestinal
ischemia-reperfusion injury and cell pyroptosis via
TXNIP-NLRP3-GSDMD pathway. Redox Biol. 32:1015342020. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Vermot A, Petit-Härtlein I, Smith SME and
Fieschi F: NADPH Oxidases (NOX): An overview from discovery,
molecular mechanisms to physiology and pathology. Antioxidants
(Basel). 10:8902021. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Lin Q, Li S, Jiang N, Shao X, Zhang M, Jin
H, Zhang Z, Shen J, Zhou Y, Zhou W, et al: PINK1-parkin pathway of
mitophagy protects against contrast-induced acute kidney injury via
decreasing mitochondrial ROS and NLRP3 inflammasome activation.
Redox Biol. 26:1012542019. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Ploumi C, Daskalaki I and Tavernarakis N:
Mitochondrial biogenesis and clearance: A balancing act. FEBS J.
284:183–195. 2017. View Article : Google Scholar
|
|
132
|
Liu S, Yuan Y, Xue Y, Xing C and Zhang B:
Podocyte injury in diabetic kidney disease: A focus on
mitochondrial dysfunction. Front Cell Dev Biol. 10:8328872022.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Audzeyenka I, Bierżyńska A and Lay AC:
Podocyte bioenergetics in the development of diabetic nephropathy:
The role of mitochondria. Endocrinology. 163:bqab2342022.
View Article : Google Scholar
|
|
134
|
Qi W, Keenan HA, Li Q, Ishikado A, Kannt
A, Sadowski T, Yorek MA, Wu IH, Lockhart S, Coppey LJ, et al:
Pyruvate kinase M2 activation may protect against the progression
of diabetic glomerular pathology and mitochondrial dysfunction. Nat
Med. 23:753–762. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Qin X, Jiang M, Zhao Y, Gong J, Su H, Yuan
F, Fang K, Yuan X, Yu X, Dong H and Lu F: Berberine protects
against diabetic kidney disease via promoting PGC-1α-regulated
mitochondrial energy homeostasis. Br J Pharmacol. 177:3646–3661.
2020. View Article : Google Scholar :
|
|
136
|
Kang Q and Yang C: Oxidative stress and
diabetic retinopathy: Molecular mechanisms, pathogenetic role and
therapeutic implications. Redox Biol. 37:1017992020. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Ham J, Song J, Song G and Lim W: Oryzalin
impairs maternal-fetal interaction during early pregnancy via
ROS-mediated P38 MAPK/AKT and OXPHOS downregulation. Food Chem
Toxicol. 174:1136652023. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Kim J, Kim HS and Chung JH: Molecular
mechanisms of mitochondrial DNA release and activation of the
cGAS-STING pathway. Exp Mol Med. 55:510–519. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Yang JX, Chuang YC, Tseng JC, Liu YL, Lai
CY, Lee AY, Huang CF, Hong YR and Chuang TH: Tumor promoting effect
of PDLIM2 downregulation involves mitochondrial ROS, oncometabolite
accumulations and HIF-1α activation. J Exp Clin Cancer Res.
43:1692024. View Article : Google Scholar
|
|
140
|
Hu H, Liang W and Ding G: Podocyte
metabolic reprogramming and targeted therapy. J Am Soc Nephrol.
37:619–633. 2026. View Article : Google Scholar
|
|
141
|
An Z, Hu H, Wang Q, Qiu Y, Chu J, Xia Y
and Li S: Dietary Selenium deficiency activates the NLRP3
inflammasome to induce gallbladder pyroptosis by regulating
glycolysis and histone lactylation through ROS/HIF-1α pathway. J
Nutr Biochem. 147:1101182026. View Article : Google Scholar
|
|
142
|
Li YX, Cui SF, Meng W, Hu HY and Wang C:
Mitochondrial DNA and cGAS-STING innate immune signaling pathway:
Latest research progress. Sichuan Da Xue Xue Bao Yi Xue Ban.
52:387–395. 2021.In Chinese. PubMed/NCBI
|
|
143
|
Xian H, Watari K, Sanchez-Lopez E,
Offenberger J, Onyuru J, Sampath H, Ying W, Hoffman HM, Shadel GS
and Karin M: Oxidized DNA fragments exit mitochondria via mPTP- and
VDAC-dependent channels to activate NLRP3 inflammasome and
interferon signaling. Immunity. 55:1370–1385.e8. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Wan S, Wan S, Jiao X, Cao H, Gu Y, Yan L,
Zheng Y, Niu P and Shao F: Advances in understanding the innate
immune-associated diabetic kidney disease. FASEB J. 35:e213672021.
View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Rock KL, Latz E, Ontiveros F and Kono H:
The sterile inflammatory response. Annu Rev Immunol. 28:321–342.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Caruso R, Warner N, Inohara N and Núñez G:
NOD1 and NOD2: Signaling, host defense, and inflammatory disease.
Immunity. 41:898–908. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Ferrara F, Cordone V, Pecorelli A,
Benedusi M, Pambianchi E, Guiotto A, Vallese A, Cervellati F and
Valacchi G: Ubiquitination as a key regulatory mechanism for
O(3)-induced cutaneous redox inflammasome activation. Redox Biol.
56:1024402022. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
La Bonte LR, Pavlov VI, Tan YS, Takahashi
K, Takahashi M, Banda NK, Zou C, Fujita T and Stahl GL:
Mannose-binding lectin-associated serine protease-1 is a
significant contributor to coagulation in a murine model of
occlusive thrombosis. J Immunol. 188:885–891. 2012. View Article : Google Scholar
|
|
149
|
Moon JY, Jeong KH, Lee TW, Ihm CG, Lim SJ
and Lee SH: Aberrant recruitment and activation of T cells in
diabetic nephropathy. Am J Nephrol. 35:164–174. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Pichler R, Afkarian M, Dieter BP and
Tuttle KR: Immunity and inflammation in diabetic kidney disease:
Translating mechanisms to biomarkers and treatment targets. Am J
Physiol Renal Physiol. 312:F716–F731. 2017. View Article : Google Scholar
|
|
151
|
Komada T and Muruve DA: The role of
inflammasomes in kidney disease. Nat Rev Nephrol. 15:501–520. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Kong L, Andrikopoulos S, MacIsaac RJ,
Mackay LK, Nikolic-Paterson DJ, Torkamani N, Zafari N, Marin ECS
and Ekinci EI: Role of the adaptive immune system in diabetic
kidney disease. J Diabetes Investig. 13:213–226. 2022. View Article : Google Scholar :
|
|
153
|
Gupta A, Singh K, Fatima S, Ambreen S,
Zimmermann S, Younis R, Krishnan S, Rana R, Gadi I, Schwab C, et
al: Correction: Gupta et al. Neutrophil extracellular traps promote
NLRP3 inflammasome activation and glomerular endothelial
dysfunction in diabetic kidney disease. Nutrients. 14:2965Nutrients
15: 2965, 2023.
|
|
154
|
Diaz-Del-Olmo I, Worboys J, Martin-Sanchez
F, Gritsenko A, Ambrose AR, Tannahill GM, Nichols EM,
Lopez-Castejon G and Davis DM: Internalization of the membrane
attack complex triggers NLRP3 inflammasome activation and IL-1β
secretion in human macrophages. Front Immunol. 12:7206552021.
View Article : Google Scholar
|
|
155
|
Huang XR, Ye L, An N, Wu CY, Wu HL, Li HY,
Huang YH, Ye QR, Liu MD, Yang LW, et al: Macrophage autophagy
protects against acute kidney injury by inhibiting renal
inflammation through the degradation of TARM1. Autophagy.
21:120–140. 2025. View Article : Google Scholar :
|
|
156
|
Zhao J, Rui HL, Yang M, Sun LJ, Dong HR
and Cheng H: CD36-mediated lipid accumulation and activation of
NLRP3 inflammasome lead to podocyte injury in obesity-related
glomerulopathy. Mediators Inflamm. 2019:31726472019.PubMed/NCBI
|
|
157
|
Hou Y, Wang Q, Han B, Chen Y, Qiao X and
Wang L: CD36 promotes NLRP3 inflammasome activation via the mtROS
pathway in renal tubular epithelial cells of diabetic kidneys. Cell
Death Dis. 12:5232021. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Huang R, Fu P and Ma L: Kidney fibrosis:
From mechanisms to therapeutic medicines. Signal Transduct Target
Ther. 8:1292023. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Zhang S, Fan Y, Qin L, Fang X, Zhang C,
Yue J, Bai W, Wang G, Chen Z, Renz H, et al: IL-1beta augments
TGF-beta inducing epithelial-mesenchymal transition of epithelial
cells and associates with poor pulmonary function improvement in
neutrophilic asthmatics. Respir Res. 22:2162021. View Article : Google Scholar
|
|
160
|
Qin D and Jiang YR: Tangeretin inhibition
of high-glucose-induced IL-1β, IL-6, TGF-β1, and VEGF expression in
human RPE cells. J Diabetes Res. 2020:94906422020. View Article : Google Scholar
|
|
161
|
You D, Nie K, Wu X, Weng M, Yang L, Chen
Y, Cui J and Wan J: C3a/C3aR synergies with TGF-β to promote
epithelial-mesenchymal transition of renal tubular epithelial cells
via the activation of the NLRP3 inflammasome. J Transl Med.
21:9042023. View Article : Google Scholar
|
|
162
|
Liu S, Yao S, Yang H, Liu S and Wang Y:
Autophagy: Regulator of cell death. Cell Death Dis. 14:6482023.
View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Lin Q, Banu K, Ni Z, Leventhal JS and
Menon MC: Podocyte autophagy in homeostasis and disease. J Clin
Med. 10:11842021. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Xu J, Shan X, Chen C, Gao Y, Zou D, Wang
X, Wang T and Shi Y: Tangshenning attenuates high glucose-induced
podocyte injury via restoring autophagy activity through inhibiting
mTORC1 activation. J Diabetes Res. 2022:16104162022. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Zhou L, Zhang L, Lv Y, Qian J, Huang L and
Qu C: YTHDC1 inhibits autophagy-dependent NF-κB signaling by
stabilizing Beclin1 mRNA in macrophages. J Inflamm (Lond).
21:222024. View Article : Google Scholar
|
|
166
|
Qi Z, Yang W, Xue B, Chen T, Lu X, Zhang
R, Li Z, Zhao X, Zhang Y, Han F, et al: ROS-mediated lysosomal
membrane permeabilization and autophagy inhibition regulate
bleomycin-induced cellular senescence. Autophagy. 20:2000–2016.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Li Y, Fan J, Zhu W, Niu Y, Wu M and Zhang
A: Therapeutic potential targeting podocyte mitochondrial
dysfunction in focal segmental glomerulosclerosis. Kidney Dis
(Basel). 9:254–264. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Abdelkader NF, Ibrahim SM, Moustafa PE and
Elbaset MA: Inosine mitigated diabetic peripheral neuropathy via
modulating GLO1/AGEs/RAGE/NF-κB/Nrf2 and TGF-β/PKC/TRPV1 signaling
pathways. Biomed Pharmacother. 145:1123952022. View Article : Google Scholar
|
|
169
|
Chen J, Zhou Z, Wu N, Li J, Xi N, Xu M, Wu
F, Fu Q, Yan G, Liu Y and Xu X: Chlorogenic acid attenuates
deoxynivalenol-induced apoptosis and pyroptosis in human
keratinocytes via activating Nrf2/HO-1 and inhibiting
MAPK/NF-κB/NLRP3 pathways. Biomed Pharmacother. 170:1160032024.
View Article : Google Scholar
|
|
170
|
Guo Y, Zhang H, Lv Z, Du Y, Li D, Fang H,
You J, Yu L and Li R: Up-regulated CD38 by daphnetin alleviates
lipopolysaccharide-induced lung injury via inhibiting
MAPK/NF-κB/NLRP3 pathway. Cell Commun Signal. 21:662023. View Article : Google Scholar
|
|
171
|
Ding R, Li H, Liu Y, Ou W, Zhang X, Chai
H, Huang X, Yang W and Wang Q: Activating cGAS-STING axis
contributes to neuroinflammation in CVST mouse model and induces
inflammasome activation and microglia pyroptosis. J
Neuroinflammation. 19:1372022. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Vande Walle L and Lamkanfi M: Drugging the
NLRP3 inflammasome: From signalling mechanisms to therapeutic
targets. Nat Rev Drug Discov. 23:43–66. 2024. View Article : Google Scholar
|
|
173
|
Zhang C, Gong Y, Li N, Liu X, Zhang Y, Ye
F, Guo Q and Zheng J: Long noncoding RNA Kcnq1ot1 promotes
sC5b-9-induced podocyte pyroptosis by inhibiting miR-486a-3p and
upregulating NLRP3. Am J Physiol Cell Physiol. 320:C355–C364. 2021.
View Article : Google Scholar
|
|
174
|
Li G, Liu C, Yang L, Feng L, Zhang S, An
J, Li J, Gao Y, Pan Z, Xu Y, et al: Syringaresinol protects against
diabetic nephropathy by inhibiting pyroptosis via NRF2-mediated
antioxidant pathway. Cell Biol Toxicol. 39:621–639. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Sharma R, Mali Y, Agrawal YO, Agnihotri VV
and Goyal SN: Repurposing nano curcumin: Unveiling its therapeutic
potential in diabetic nephropathy. Curr Drug Targets. 26:298–319.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Shahzad K, Bock F, Dong W, Wang H, Kopf S,
Kohli S, Al-Dabet MM, Ranjan S, Wolter J, Wacker C, et al:
Nlrp3-inflammasome activation in non-myeloid-derived cells
aggravates diabetic nephropathy. Kidney Int. 87:74–84. 2015.
View Article : Google Scholar :
|
|
177
|
Lu M, Yin J, Xu T, Dai X, Liu T, Zhang Y,
Wang S, Liu Y, Shi H, Zhang Y, et al: Fuling-Zexie formula
attenuates hyperuricemia-induced nephropathy and inhibits
JAK2/STAT3 signaling and NLRP3 inflammasome activation in mice. J
Ethnopharmacol. 319:1172622024. View Article : Google Scholar
|
|
178
|
Zhang Y, Wang S, Dai X, Liu T, Liu Y, Shi
H, Yin J, Xu T, Zhang Y, Zhao D, et al: Simiao San alleviates
hyperuricemia and kidney inflammation by inhibiting NLRP3
inflammasome and JAK2/STAT3 signaling in hyperuricemia mice. J
Ethnopharmacol. 312:1165302023. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Wang P, Yang J, Dai S, Gao P, Qi Y, Zhao
X, Liu J, Wang Y and Gao Y: miRNA-193a-mediated WT1 suppression
triggers podocyte injury through activation of the
EZH2/β-catenin/NLRP3 pathway in children with diabetic nephropathy.
Exp Cell Res. 442:1142382024. View Article : Google Scholar
|
|
180
|
Zhang X, Li B, Huo S, Du J, Zhang J, Song
M, Cui Y and Li Y: T-2 toxin induces kidney fibrosis via the
mtROS-NLRP3-Wnt/β-catenin axis. J Agric Food Chem. 70:13765–13777.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Ansari Z, Chaurasia A, Neha, Sharma N,
Bachheti RK and Gupta PC: Exploring inflammatory and fibrotic
mechanisms driving diabetic nephropathy progression. Cytokine
Growth Factor Rev. 84:120–134. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Zhang C, Zhu X, Li L, Ma T, Shi M, Yang Y
and Fan Q: A small molecule inhibitor MCC950 ameliorates kidney
injury in diabetic nephropathy by inhibiting NLRP3 inflammasome
activation. Diabetes Metab Syndr Obes. 12:1297–1309. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Wu M, Yang Z, Zhang C and Shi Y, Han W,
Song S, Mu L, Du C and Shi Y: Inhibition of NLRP3 inflammasome
ameliorates podocyte damage by suppressing lipid accumulation in
diabetic nephropathy. Metabolism. 118:1547482021. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Juliana C, Fernandes-Alnemri T, Wu J,
Datta P, Solorzano L, Yu JW, Meng R, Quong AA, Latz E, Scott CP and
Alnemri ES: Anti-inflammatory compounds parthenolide and Bay
11-7082 are direct inhibitors of the inflammasome. J Biol Chem.
285:9792–9802. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Kolati SR, Kasala ER, Bodduluru LN,
Mahareddy JR, Uppulapu SK, Gogoi R, Barua CC and Lahkar M: BAY
11-7082 ameliorates diabetic nephropathy by attenuating
hyperglycemia-mediated oxidative stress and renal inflammation via
NF-κB pathway. Environ Toxicol Pharmacol. 39:690–699. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Shi X, Zou W, Li X, Liu S, Hu T, Li Q,
Zhang T, Chen L, Wu S, Wang C and Jin Y: SGLT2 inhibition
attenuates diabetic tubulopathy by suppressing SGK1-mediated
pyroptosis. Front Endocrinol (Lausanne). 16:16202302025. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Qiu Z, Lei S, Zhao B, Wu Y, Su W, Liu M,
Meng Q, Zhou B, Leng Y and Xia ZY: NLRP3 inflammasome
activation-mediated pyroptosis aggravates myocardial
ischemia/reperfusion injury in diabetic rats. Oxid Med Cell Longev.
2017:97432802017. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Qiu Z, He Y, Ming H, Lei S, Leng Y and Xia
ZY: Lipopolysaccharide (LPS) aggravates high glucose- and
hypoxia/reoxygenation-induced injury through activating
ROS-Dependent NLRP3 inflammasome-mediated pyroptosis in H9C2
cardiomyocytes. J Diabetes Res. 2019:81518362019. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Zhang ZW, Tang MQ, Liu W, Song Y, Gao MJ,
Ni P, Zhang DD, Mo QG and Zhao BQ: Dapagliflozin prevents kidney
podocytes pyroptosis via miR-155-5p/HO-1/NLRP3 axis modulation. Int
Immunopharmacol. 131:1117852024. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Birnbaum Y, Bajaj M, Qian J and Ye Y:
Dipeptidyl peptidase-4 inhibition by Saxagliptin prevents
inflammation and renal injury by targeting the Nlrp3/ASC
inflammasome. BMJ Open Diabetes Res Care. 4:e0002272016. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Chen A, Chen Z, Xia Y, Lu D, Yang X, Sun
A, Zou Y, Qian J and Ge J: Liraglutide attenuates NLRP3
inflammasome-dependent pyroptosis via regulating SIRT1/NOX4/ROS
pathway in H9c2 cells. Biochem Biophys Res Commun. 499:267–272.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Gao Y, Ma Y, Xie D and Jiang H: ManNAc
protects against podocyte pyroptosis via inhibiting mitochondrial
damage and ROS/NLRP3 signaling pathway in diabetic kidney injury
model. Int Immunopharmacol. 107:1087112022. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Li R, Zeng X, Yang M, Feng J, Xu X, Bao L,
Ye T, Wang X, Xue B and Huang Y: Antidiabetic DPP-4 inhibitors
reprogram tumor microenvironment that facilitates murine breast
cancer metastasis through interaction with cancer cells via a
ROS-NF-кB-NLRP3 axis. Front Oncol. 11:7280472021. View Article : Google Scholar
|
|
194
|
Tang PM, Zhang YY, Hung JS, Chung JY,
Huang XR, To KF and Lan HY: DPP4/CD32b/NF-κB Circuit: A novel
druggable target for inhibiting CRP-Driven diabetic nephropathy.
Mol Ther. 29:365–375. 2021. View Article : Google Scholar
|
|
195
|
Wang F, Liu C, Ren L, Li Y, Yang H, Yu Y
and Xu W: Sanziguben polysaccharides improve diabetic nephropathy
in mice by regulating gut microbiota to inhibit the
TLR4/NF-κB/NLRP3 signalling pathway. Pharm Biol. 61:427–436. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Du L, Wang J, Chen Y, Li X, Wang L, Li Y,
Jin X, Gu X, Hao M, Zhu X, et al: Novel biphenyl diester derivative
AB-38b inhibits NLRP3 inflammasome through Nrf2 activation in
diabetic nephropathy. Cell Biol Toxicol. 36:243–260. 2020.
View Article : Google Scholar
|
|
197
|
Liu XQ, Jiang L, Lei L, Nie ZY, Zhu W,
Wang S, Zeng HX, Zhang SQ, Zhang Q, Yard B and Wu YG: Carnosine
alleviates diabetic nephropathy by targeting GNMT, a key enzyme
mediating renal inflammation and fibrosis. Clin Sci (Lond).
134:3175–3193. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Ram C, Gairola S, Verma S, Mugale MN,
Bonam SR, Murty US and Sahu BD: Biochanin a ameliorates nephropathy
in high-fat diet/streptozotocin-induced diabetic rats: Effects on
NF-kB/NLRP3 axis, pyroptosis, and fibrosis. Antioxidants (Basel).
12:10522023. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Li Y, Long W, Zhang H, Zhao M, Gao M, Guo
W and Yu L: Irbesartan ameliorates diabetic nephropathy by
activating the Nrf2/Keap1 pathway and suppressing NLRP3
inflammasomes in vivo and in vitro. Int Immunopharmacol.
131:1118442024. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Azar YO, Ibrahim SM, Zaki HF, Elshazly SM
and Badawi GA: Targeting α-klotho protein by agmatine and
pioglitazone is a new avenue against diabetic nephropathy. ACS
Pharmacol Transl Sci. 8:2493–2506. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Awad AM, Elshaer SL, Gangaraju R,
Abdelaziz RR and Nader MA: Ameliorative effect of montelukast
against STZ induced diabetic nephropathy: Targeting HMGB1, TLR4,
NF-κB, NLRP3 inflammasome, and autophagy pathways.
Inflammopharmacology. 32:495–508. 2024. View Article : Google Scholar :
|
|
202
|
Zhou R, Guo T and Li J: Research progress
on the antitumor effects of astragaloside IV. Eur J Pharmacol.
938:1754492023. View Article : Google Scholar
|
|
203
|
Gao Y, Su X, Xue T and Zhang N: The
beneficial effects of astragaloside IV on ameliorating diabetic
kidney disease. Biomed Pharmacother. 163:1145982023. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Zhang M, Liu W, Liu Y, Zhang Z, Hu Y, Sun
D, Li S and Fang J: Astragaloside IV inhibited podocyte pyroptosis
in diabetic kidney disease by regulating SIRT6/HIF-1α axis. DNA
Cell Biol. 42:594–607. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
He J, Cui J, Shi Y, Wang T, Xin J, Li Y,
Shan X, Zhu Z and Gao Y: Astragaloside IV attenuates
high-glucose-induced impairment in diabetic nephropathy by
increasing klotho expression via the NF-κB/NLRP3 Axis. J Diabetes
Res. 2023:74236612023. View Article : Google Scholar
|
|
206
|
Tong L, Zhao Q, Datan E, Lin GQ, Minn I,
Pomper MG, Yu B, Romo D, He QL and Liu JO: Triptolide: Reflections
on two decades of research and prospects for the future. Nat Prod
Rep. 38:843–860. 2021. View Article : Google Scholar
|
|
207
|
Wu W, Liu BH, Wan YG, Sun W, Liu YL, Wang
WW, Fang QJ, Tu Y, Yee HY, Yuan CC and Wan ZY: Triptolide inhibits
NLRP3 inflammasome activation and ameliorates podocyte
epithelial-mesenchymal transition induced by high glucose. Zhongguo
Zhong Yao Za Zhi. 44:5457–5464. 2019.In Chinese.
|
|
208
|
Lv C, Cheng T, Zhang B, Sun K and Lu K:
Triptolide protects against podocyte injury in diabetic nephropathy
by activating the Nrf2/HO-1 pathway and inhibiting the NLRP3
inflammasome pathway. Ren Fail. 45:21651032023. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Zhou P, Xie W, He S, Sun Y, Meng X, Sun G
and Sun X: Ginsenoside Rb1 as an Anti-diabetic agent and its
underlying mechanism analysis. Cells. 8:2042019. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Song W, Wei L, Du Y, Wang Y and Jiang S:
Protective effect of ginsenoside metabolite compound K against
diabetic nephropathy by inhibiting NLRP3 inflammasome activation
and NF-κB/p38 signaling pathway in high-fat
diet/streptozotocin-induced diabetic mice. Int Immunopharmacol.
63:227–238. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Wang T, Gao Y, Yue R, Wang X, Shi Y, Xu J,
Wu B and Li Y: Ginsenoside Rg1 alleviates podocyte injury induced
by hyperlipidemia via targeting the mTOR/NF-κB/NLRP3 axis. Evid
Based Complement Alternat Med. 2020:27357142020. View Article : Google Scholar
|
|
212
|
Shi Y, Wang H, Zheng M, Xu W, Yang Y and
Shi F: Ginsenoside Rg3 suppresses the NLRP3 inflammasome activation
through inhibition of its assembly. FASEB J. 34:208–221. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Zhu Y, Zhu C, Yang H, Deng J and Fan D:
Protective effect of ginsenoside Rg5 against kidney injury via
inhibition of NLRP3 inflammasome activation and the MAPK signaling
pathway in high-fat diet/streptozotocin-induced diabetic mice.
Pharmacol Res. 155:1047462020. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Wang J, Geng L, Yue Y and Zhang Q: Use of
fucoidan to treat renal diseases: A review of 15 years of clinic
studies. Prog Mol Biol Transl Sci. 163:95–111. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Wang MZ, Wang J, Cao DW, Tu Y, Liu BH,
Yuan CC, Li H, Fang QJ, Chen JX, Fu Y, et al: Fucoidan alleviates
renal fibrosis in diabetic kidney disease via inhibition of NLRP3
inflammasome-mediated podocyte pyroptosis. Front Pharmacol.
13:7909372022. View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Ding T, Wang S, Zhang X, Zai W, Fan J,
Chen W, Bian Q, Luan J, Shen Y, Zhang Y, et al: Kidney protection
effects of dihydroquercetin on diabetic nephropathy through
suppressing ROS and NLRP3 inflammasome. Phytomedicine. 41:45–53.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Liu Y, Lei H, Zhang W, Xing Q, Liu R, Wu
S, Liu Z, Yan Q, Li W, Liu X and Hu Y: Pyroptosis in renal
inflammation and fibrosis: Current knowledge and clinical
significance. Cell Death Dis. 14:4722023. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Wang C, Hou XX, Rui HL, Li LJ, Zhao J,
Yang M, Sun LJ, Dong HR, Cheng H and Chen YP: Artificially
cultivated ophiocordyceps sinensis alleviates diabetic nephropathy
and its podocyte injury via inhibiting P2X7R expression and NLRP3
inflammasome activation. J Diabetes Res. 2018:13904182018.
View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Han W, Ma Q, Liu Y, Wu W, Tu Y, Huang L,
Long Y, Wang W, Yee H, Wan Z, et al: Huangkui capsule alleviates
renal tubular epithelial-mesenchymal transition in diabetic
nephropathy via inhibiting NLRP3 inflammasome activation and
TLR4/NF-κB signaling. Phytomedicine. 57:203–214. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Liu BH, Tu Y, Ni GX, Yan J, Yue L, Li ZL,
Wu JJ, Cao YT, Wan ZY, Sun W and Wan YG: Total flavones of
abelmoschus manihot ameliorates podocyte pyroptosis and injury in
high glucose conditions by targeting METTL3-dependent m(6)A
modification-mediated NLRP3-Inflammasome activation and
PTEN/PI3K/Akt signaling. Front Pharmacol. 12:6676442021. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Li F, Chen Y, Li Y, Huang M and Zhao W:
Geniposide alleviates diabetic nephropathy of mice through
AMPK/SIRT1/NF-κB pathway. Eur J Pharmacol. 886:1734492020.
View Article : Google Scholar
|
|
222
|
Wu Q, Guan YB, Zhang KJ, Li L and Zhou Y:
Tanshinone IIA mediates protection from diabetes kidney disease by
inhibiting oxidative stress induced pyroptosis. J Ethnopharmacol.
316:1166672023. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Yosri H, El-Kashef DH, El-Sherbiny M, Said
E and Salem HA: Calycosin modulates NLRP3 and TXNIP-mediated
pyroptotic signaling and attenuates diabetic nephropathy
progression in diabetic rats; An insight. Biomed Pharmacother.
155:1137582022. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Liu W, Liang L, Zhang Q, Li Y, Yan S, Tang
T, Ren Y, Mo J, Liu F, Chen X and Lan T: Effects of andrographolide
on renal tubulointersticial injury and fibrosis. Evidence of its
mechanism of action. Phytomedicine. 91:1536502021. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
An X, Zhang Y, Cao Y, Chen J, Qin H and
Yang L: Punicalagin protects diabetic nephropathy by inhibiting
pyroptosis based on TXNIP/NLRP3 pathway. Nutrients. 12:15162020.
View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Pan S, Jiang SS, Li R, Tian B, Huang CY,
Wang R, Li YY, Zhu H, Yuan YF and Hu X: Hong Guo Ginseng Guo (HGGG)
protects against kidney injury in diabetic nephropathy by
inhibiting NLRP3 inflammasome and regulating intestinal flora.
Phytomedicine. 132:1558612024. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Huang R, Zeng J, Yu X, Shi Y, Song N,
Zhang J, Wang P, Luo M, Ma Y, Xiao C, et al: Luteolin alleviates
diabetic nephropathy fibrosis involving AMPK/NLRP3/TGF-β pathway.
Diabetes Metab Syndr Obes. 17:2855–2867. 2024. View Article : Google Scholar
|
|
228
|
Tsai IN, Tsai YC, Huang HP, Chen CC, Hung
TW and Wang CJ: Protective effect of neochlorogenic acid on
diabetic nephropathy via inflammation and pyroptosis suppression. J
Agric Food Chem. 73:26763–26779. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Tang L, Yuan L, Ren D, Huang J, Liu R, Xia
Z, Huang N and Zhang S: Tetrandrine improves oxidative stress and
pyroptosis of podocytes in diabetic kidney disease by regulating
TXNIP/NLRP3/GSDMD signaling pathway. J Mol Histol. 56:3272025.
View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Cui Y, Lin L, Yu B, Wu M and Teng J:
Chelerythrine ameliorates renal injury in diabetic nephropathy rats
by enhancing autophagy and modulating AMPK/mTOR signaling with
concurrent anti-inflammatory and anti-fibrotic actions. Biochem
Biophys Res Commun. 778:1523622025. View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Shanshan J, Shu P, Xiao H, Kuerban K, Hao
Z, Yujie W, Rong W, Yuhuan S and Yongfang Y: Modulating the gut
microbiota and inflammation is involved in the effect of diosgenin
against diabetic nephropathy in rat. Front Pharmacol.
16:15558492025. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Wang L, Xie X, Chen Q, Chen Y, Xu X and
Liang T: Puerarin reduces diabetic nephropathy-induced podocyte
pyroptosis by modulating the SIRT1/NLRP3/caspase-1 pathway. Mol
Cell Endocrinol. 595:1124092025. View Article : Google Scholar
|
|
233
|
Xiao T, Zhao H, Wang Y, Chen M, Wang C and
Qiao C: Shionone inhibits glomerular fibirosis by suppressing NLRP3
related inflammasome though SESN2-NRF2/HO-1 pathway. Diabetes Metab
J. 49:34–48. 2025. View Article : Google Scholar :
|
|
234
|
Wang Y, Yang J, Chang X, Xue Y, Liu G,
Zhang T, Chen W, Fan W, Tian J and Ren X: Isoliquiritigenin
alleviates diabetic kidney disease via oxidative stress and the
TLR4/NF-κB/NLRP3 inflammasome pathway. Mol Nutr Food Res.
68:e24002152024. View Article : Google Scholar
|
|
235
|
Kong X, Zhao Y and Wang X, Yu Y, Meng Y,
Yan G, Yu M, Jiang L, Song W, Wang B and Wang X: Loganin reduces
diabetic kidney injury by inhibiting the activation of NLRP3
inflammasome-mediated pyroptosis. Chem Biol Interact.
382:1106402023. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Huang G, Zhang Y, Zhang Y, Zhou X, Xu Y,
Wei H, Chen X and Ma Y: Oridonin attenuates diabetes-induced renal
fibrosis via the inhibition of TXNIP/NLRP3 and NF-κB pathways by
activating PPARγ in rats. Exp Clin Endocrinol Diabetes.
132:536–544. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Zhang X, Wei W, Liu Z, Gao H, Guo F, Liu
D, Yin Y and Yang X: Comprehensive transcriptomic and bioinformatic
analysis of the mechanism of Buzhong Yiqi decoction in the
improvement of diabetic nephropathy. Endocr Metab Immune Disord
Drug Targets. 25:1099–1109. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Zhang Q, Liu X, Sullivan MA, Shi C and
Deng B: Protective effect of Yi Shen Pai Du Formula against
diabetic kidney injury via inhibition of oxidative stress,
inflammation, and epithelial-to-mesenchymal transition in db/db
Mice. Oxid Med Cell Longev. 2021:79580212021. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Lin Q, Long C, Wang Z, Wang R, Shi W, Qiu
J, Mo J and Xie Y: Hirudin, a thrombin inhibitor, attenuates
TGF-β-induced fibrosis in renal proximal tubular epithelial cells
by inhibition of protease-activated receptor 1 expression via
S1P/S1PR2/S1PR3 signaling. Exp Ther Med. 23:32022. View Article : Google Scholar
|
|
240
|
Liu XJ, Hu XK, Yang H, Gui LM, Cai ZX, Qi
MS and Dai CM: A review of traditional Chinese medicine on
treatment of diabetic nephropathy and the involved mechanisms. Am J
Chin Med. 50:1739–1779. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Yi ZY, Peng YJ, Hui BP, Liu Z, Lin QX,
Zhao D, Wang Y, Liu X, Xie J, Zhang SH, et al:
Zuogui-Jiangtang-Yishen decoction prevents diabetic kidney disease:
Intervene pyroptosis induced by trimethylamine n-oxide through the
mROS-NLRP3 axis. Phytomedicine. 114:1547752023. View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Liu T, Lu X, Gao W, Zhai Y, Li H, Li S,
Yang L, Ma F, Zhan Y and Mao H: Cardioprotection effect of
Yiqi-Huoxue-Jiangzhuo formula in a chronic kidney disease mouse
model associated with gut microbiota modulation and NLRP3
inflammasome inhibition. Biomed Pharmacother. 152:1131592022.
View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Jia Q, Zhang X, Hao G, Zhao Y, Lowe S, Han
L and Qin J: Tongluo Yishen decoction ameliorates renal fibrosis
via NLRP3-Mediated pyroptosis in vivo and in vitro. Front
Pharmacol. 13:9368532022. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Jia Q, Han L, Zhang X, Yang W, Gao Y, Shen
Y, Li B, Wang S, Qin M, Lowe S, et al: Tongluo Yishen decoction
ameliorates renal fibrosis via regulating mitochondrial dysfunction
induced by oxidative stress in unilateral ureteral obstruction
rats. Front Pharmacol. 12:7627562021. View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Ma ZA, Wang LX, Zhang H, Li HZ, Dong L,
Wang QH, Wang YS, Pan BC, Zhang SF, Cui HT and Lv SQ: Jianpi Gushen
Huayu decoction ameliorated diabetic nephropathy through modulating
metabolites in kidney, and inhibiting TLR4/NF-κB/NLRP3 and JNK/P38
pathways. World J Diabetes. 15:502–518. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
246
|
Li N, Zhao T, Cao Y, Zhang H, Peng L, Wang
Y, Zhou X, Wang Q, Li J, Yan M, et al: Tangshen formula attenuates
diabetic kidney injury by imparting anti-pyroptotic effects via the
TXNIP-NLRP3-GSDMD axis. Front Pharmacol. 11:6234892020. View Article : Google Scholar
|
|
247
|
Yang YJ, Ye BH, Qiu C, Wu HQ, Huang BW,
Wang T, Ruan SW, Guo F, Wang JT and Jiang MQ: Mechanism of Yishen
Jiangtang Decoction in regulating endoplasmic reticulum
stress-mediated NLRP3 inflammasome to improve renal damage in
diabetic nephropathy db/db mice. Zhongguo Zhong Yao Za Zhi.
50:2740–2749. 2025.In Chinese. PubMed/NCBI
|
|
248
|
Li J, Wang B, Zhou G, Yan X and Zhang Y:
Tetrahydroxy stilbene glucoside alleviates high glucose-induced
MPC5 podocytes injury through suppression of NLRP3 inflammasome. Am
J Med Sci. 355:588–596. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
249
|
Fan W, Chen S, Wu X, Zhu J and Li J:
Resveratrol relieves gouty arthritis by promoting mitophagy to
inhibit activation of NLRP3 inflammasomes. J Inflamm Res.
14:3523–3536. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Wang S, Zhao X, Yang S, Chen B and Shi J:
Salidroside alleviates high glucose-induced oxidative stress and
extracellular matrix accumulation in rat glomerular mesangial cells
by the TXNIP-NLRP3 inflammasome pathway. Chem Biol Interact.
278:48–53. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Li S, Rao XR, Dai XW, Pei K, Wang L, Huo
BM, Wang XJ, Kong LX, Zhang NN and Lian FM: Beneficial effects of
Fu-Zheng-Qu-Zhuo oral liquid combined with standard integrated
therapy in patients with chronic kidney disease (stage 3-4): A
randomized placebo-controlled clinical trial. Medicine (Baltimore).
96:e74482017. View Article : Google Scholar : PubMed/NCBI
|
|
252
|
Bhagat M, Kamal R, Sharma J, Kaur K,
Sharma A, Singh TG, Bhatia R and Awasthi A: Gene Therapy: Towards a
new era of medicine. AAPS PharmSciTech. 26:172024. View Article : Google Scholar : PubMed/NCBI
|
|
253
|
Lao A, Li W, Sun Y, Cao Y, Zhuang Y, Wu J,
Li D, Lin K, Mao J and Liu J: Metabolic and immunomodulatory
control of type 2 diabetes via generating cellular itaconate
reservoirs by inflammatory-targeting gene-therapy nanovesicles.
Trends Biotechnol. 44:170–192. 2026. View Article : Google Scholar
|
|
254
|
Tang J, Yao D, Yan H, Chen X, Wang L and
Zhan H: The role of MicroRNAs in the pathogenesis of diabetic
nephropathy. Int J Endocrinol. 2019:87190602019. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Szostak J, Gorący A, Durys D, Dec P,
Modrzejewski A and Pawlik A: The role of MicroRNA in the
pathogenesis of diabetic nephropathy. Int J Mol Sci. 24:62142023.
View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Yan H, Luo B, Wu X, Guan F, Yu X, Zhao L,
Ke X, Wu J and Yuan J: Cisplatin induces pyroptosis via activation
of MEG3/NLRP3/caspase-1/GSDMD pathway in triple-negative breast
cancer. Int J Biol Sci. 17:2606–2621. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
257
|
Xu Y, Fang H, Xu Q, Xu C, Yang L and Huang
C: LncRNA GAS5 inhibits NLRP3 inflammasome activation-mediated
pyroptosis in diabetic cardiomyopathy by targeting miR-34b-3p/AHR.
Cell Cycle. 19:3054–3065. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Zhang M, Xue SJ, Yang F, Xiao M, Tang YB
and Wu Y: LncRNA SNHG7/miR-181b-5p/TLR4 activates inflammation and
promotes pyroptosis through NF-κB signaling in diabetic
nephropathy. Inflammation. 48:3828–3840. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Liu Y, Zhang M, Zhong H, Xie N, Wang Y,
Ding S and Su X: LncRNA SNHG16 regulates RAS and NF-κB
pathway-mediated NLRP3 inflammasome activation to aggravate
diabetes nephropathy through stabilizing TLR4. Acta Diabetol.
60:563–577. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Zhuang L, Jin G, Wang Q, Ge X and Pei X:
Long Non-coding RNA ZFAS1 regulates fibrosis and scortosis in the
cell model of diabetic nephropathy through miR-525-5p/SGK1 axis.
Appl Biochem Biotechnol. 196:3731–3746. 2024. View Article : Google Scholar
|
|
261
|
Xu J, Wang Q, Song YF, Xu XH, Zhu H, Chen
PD and Ren YP: Long noncoding RNA X-inactive specific transcript
regulates NLR family pyrin domain containing 3/caspase-1-mediated
pyroptosis in diabetic nephropathy. World J Diabetes. 13:358–375.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
262
|
El-Lateef AEA, El-Shemi AGA, Alhammady MS,
Yuan R and Zhang Y: LncRNA NEAT2 modulates pyroptosis of renal
tubular cells induced by high glucose in diabetic nephropathy (DN)
by via miR-206 regulation. Biochem Genet. 60:1733–1747. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
263
|
Zhan JF, Huang HW, Huang C, Hu LL and Xu
WW: Long Non-Coding RNA NEAT1 regulates pyroptosis in diabetic
nephropathy via mediating the miR-34c/NLRP3 axis. Kidney Blood
Press Res. 45:589–602. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
264
|
Xu C, Lu Z, Luo Y, Liu Y, Cao Z, Shen S,
Li H, Liu J, Chen K, Chen Z, et al: Targeting of NLRP3 inflammasome
with gene editing for the amelioration of inflammatory diseases.
Nat Commun. 9:40922018. View Article : Google Scholar : PubMed/NCBI
|
|
265
|
Wan T, Pan Q and Ping Y:
Microneedle-assisted genome editing: A transdermal strategy of
targeting NLRP3 by CRISPR-Cas9 for synergistic therapy of
inflammatory skin disorders. Sci Adv. 7:eabe28882021. View Article : Google Scholar : PubMed/NCBI
|
|
266
|
Lv F, He Y, Xu H, Li Y, Han L, Yan L, Lang
H, Zhao Y, Zhao Z and Qi Y: CD36 aggravates podocyte injury by
activating NLRP3 inflammasome and inhibiting autophagy in lupus
nephritis. Cell Death Dis. 13:7292022. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Kadowaki A, Wheeler MA, Li Z, Andersen BM,
Lee HG, Illouz T, Lee JH, Ndayisaba A, Zandee SEJ, Basu H, et al:
CLEC16A in astrocytes promotes mitophagy and limits pathology in a
multiple sclerosis mouse model. Nat Neurosci. 28:470–486. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
268
|
Tian C, Min X, Zhao Y, Wang Y, Wu X, Liu
S, Dou W, Zhou T, Liu Y, Luo R, et al: MRG15 aggravates
non-alcoholic steatohepatitis progression by regulating the
mitochondrial proteolytic degradation of TUFM. J Hepatol.
77:1491–1503. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
269
|
Zhuo X, Wu Y, Fu X, Li J, Xiang Y, Liang
X, Mao C and Jiang Y: Genome editing of PAR2 through targeted
delivery of CRISPR-Cas9 system for alleviating acute lung
inflammation via ERK/NLRP3/IL-1β and NO/iNOS signalling. Acta Pharm
Sin B. 14:1441–1456. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Monjarret B, Shour S, Benyoucef A,
Benyoucef A, Heckel E, Marchitto L, Leiding JW, Cros G, Fernandez
I, Joyal JS and Touzot F: NOX2 deficiency enhances priming and
activation of the NLRP3 inflammasome. J Allergy Clin Immunol.
157:711–721.e5. 2026. View Article : Google Scholar
|















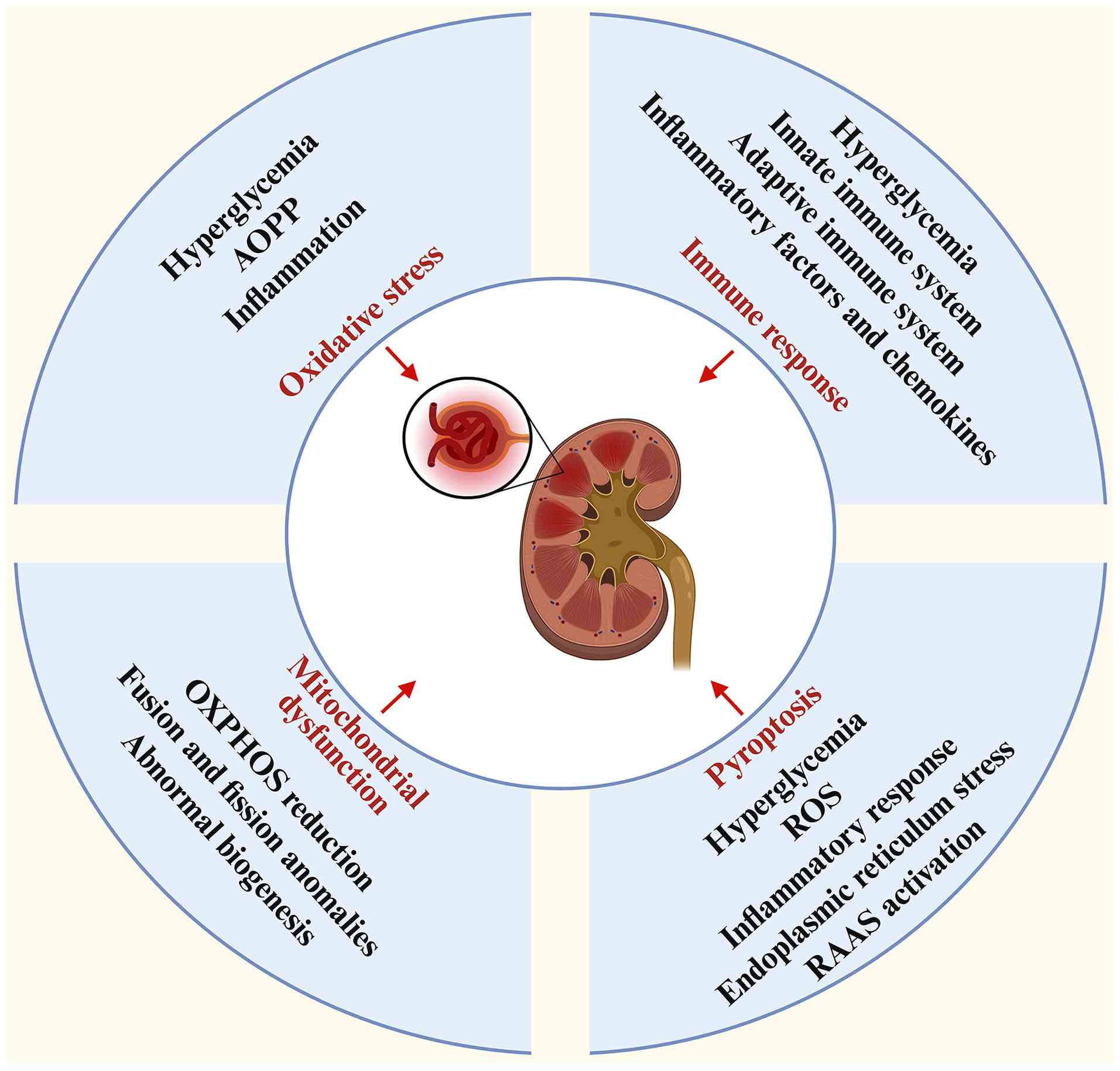
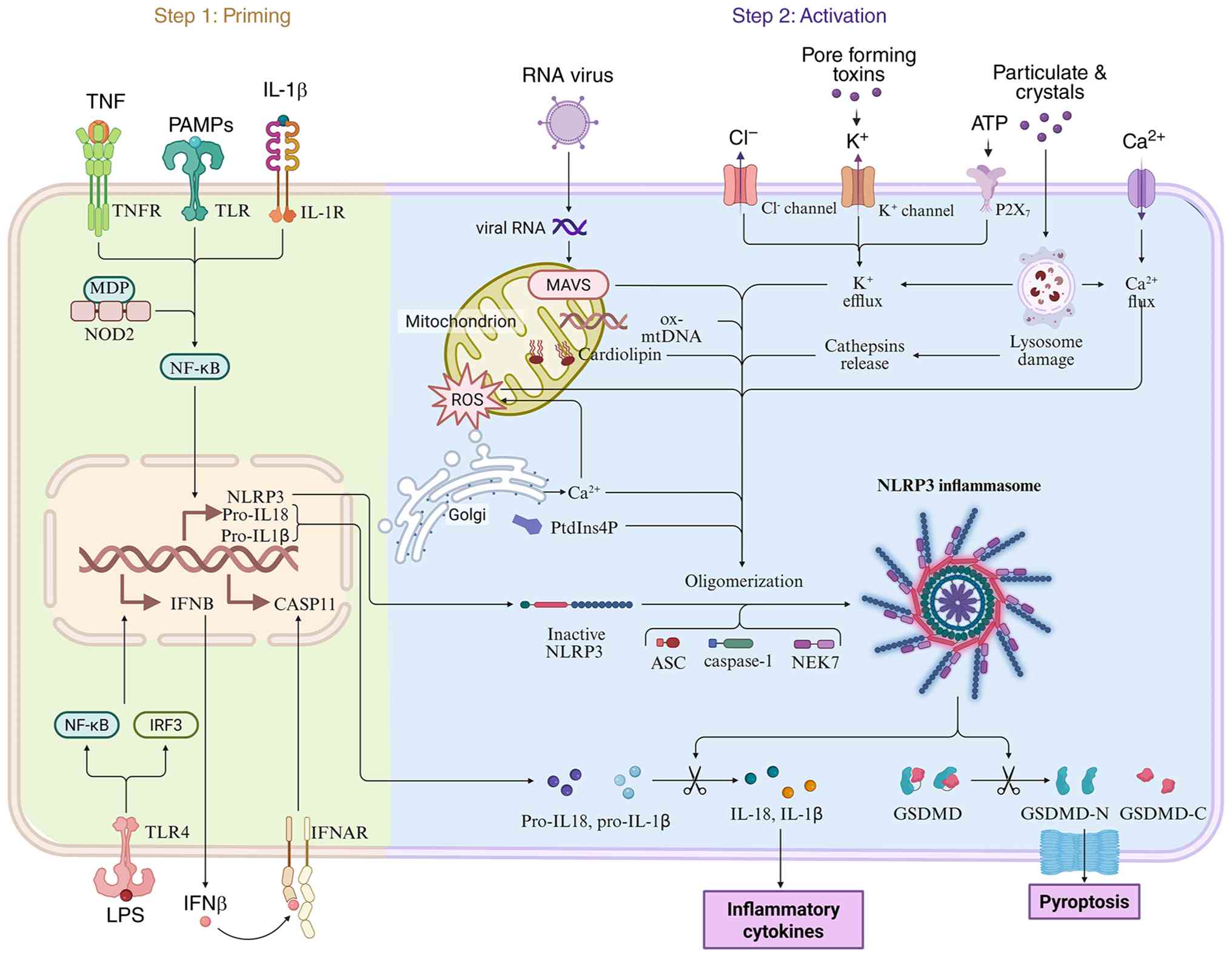


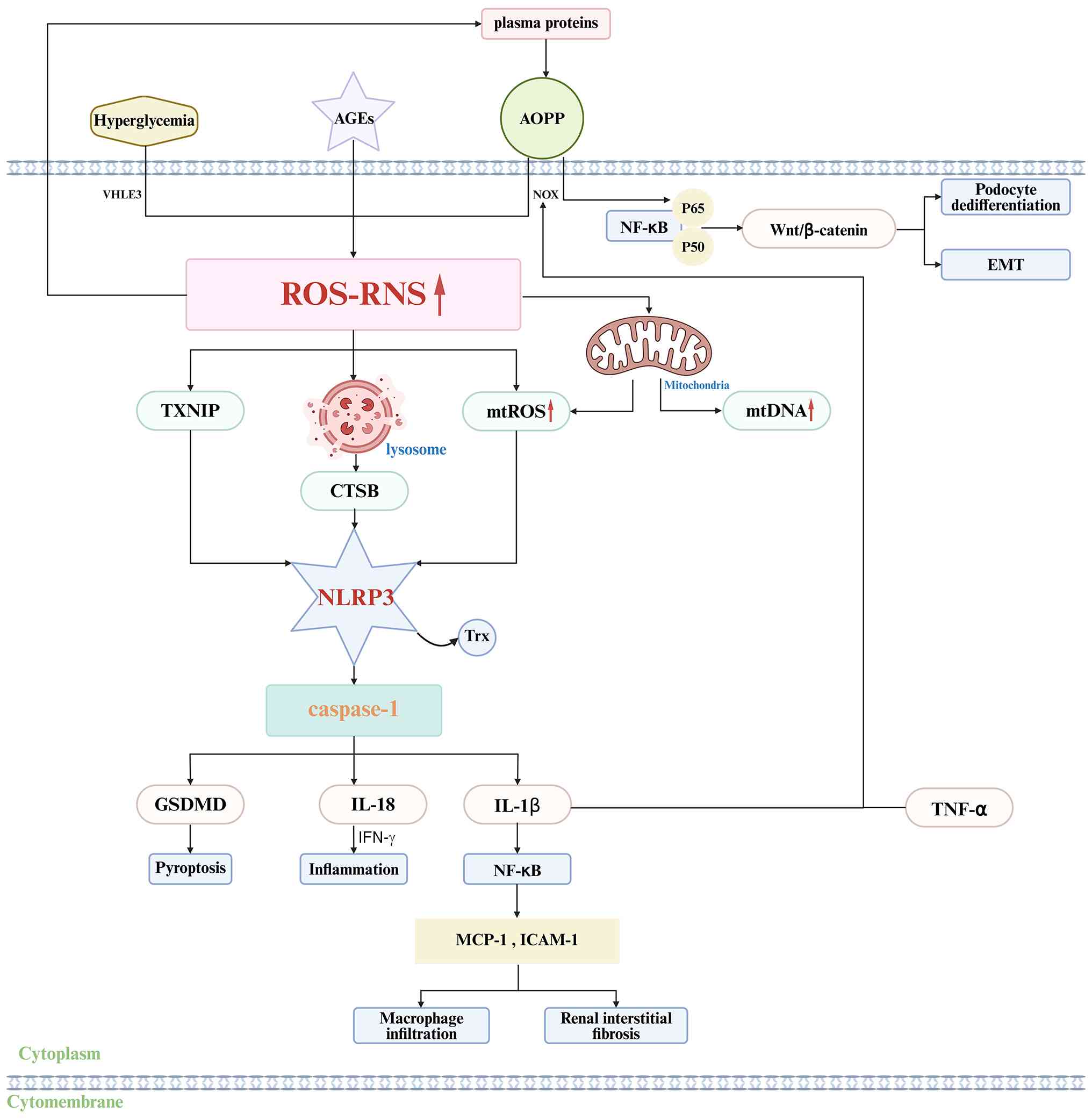
![Schematic diagram illustrating the
molecular mechanism through which hyperglycemia and AGEs regulate
NLRP3 inflammasome activation via mitochondrial pathways. This
figure delineates the molecular cascades whereby hyperglycemia and
AGEs (acting via receptor for AGEs [RAGE] binding) modulate
mitochondrial function, subsequently activating the NLRP3
inflammasome and triggering downstream inflammatory responses and
cell death pathways. Within this schematic, red arrows denote
increased activity or levels, whereas black arrows signify
molecular promotion or activation events. AGEs, advanced glycation
end products; RAGE, receptor for advanced glycation end products;
ROS, reactive oxygen species; CerS6, Ceramide Synthase 6; SDH,
succinate dehydrogenase; OXPHOS, oxidative phosphorylation; mtROS,
mitochondrial reactive oxygen species; mtDNA, mitochondrial DNA;
cGAS, cyclic GMP-AMP synthase; cGAMP, cyclic guanosine
monophosphate-adenosine monophosphate; SITNG, stimulator of
interferon genes; TBK1, TANK-binding kinase 1; NLRP3, NOD-like
receptor family pyrin domain containing 3; IRF3, interferon
regulatory factor 3; GSDM, Gasdermin; H3K18la, Histone H3 Lysine 18
Lactylation; HIF-1α, hypoxia inducible factor 1 subunit α.](/article_images/ijmm/58/1/ijmm-58-01-05847-g05.jpg)